User login
For MD-IQ use only
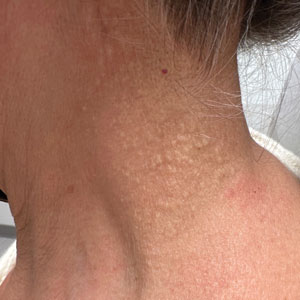
Cobblestonelike Papules on the Neck
The Diagnosis: Fibroelastolytic Papulosis
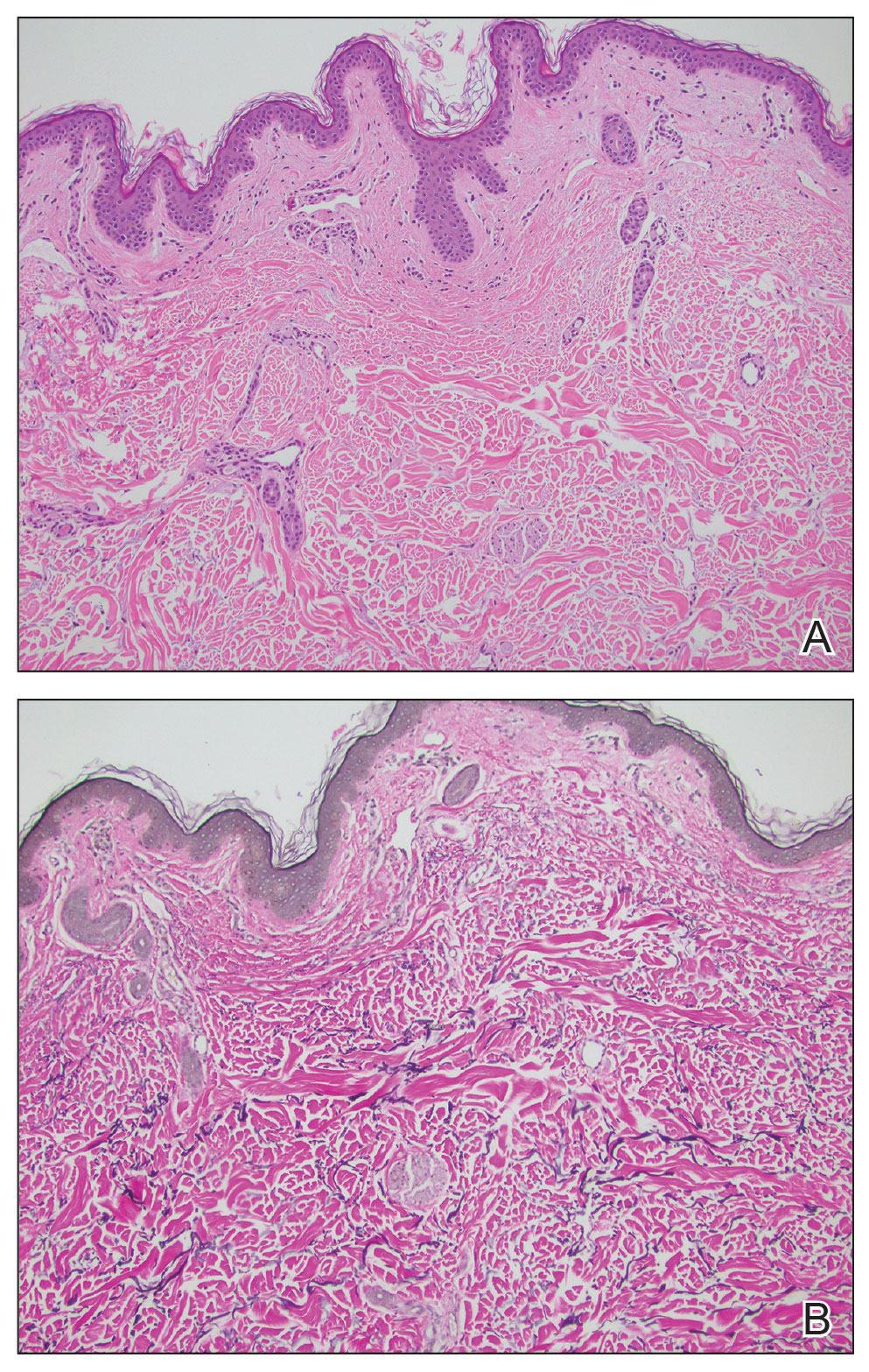
Histopathology demonstrated decreased density and fragmentation of elastic fibers in the superficial reticular and papillary dermis consistent with an elastolytic disease process (Figure). Of note, elastolysis typically is visualized with Verhoeff-van Gieson stain but cannot be visualized well with standard hematoxylin and eosin staining. Additional staining with Congo red was negative for amyloid, and colloidal iron did not show any increase in dermal mucin, ruling out amyloidosis and scleromyxedema, respectively. Based on the histopathologic findings and the clinical history, a diagnosis of fibroelastolytic papulosis (FP) was made. Given the benign nature of the condition, the patient was prescribed a topical steroid (clobetasol 0.05%) for symptomatic relief.
Cutaneous conditions can arise from abnormalities in the elastin composition of connective tissue due to abnormal elastin formation or degradation (elastolysis).1 Fibroelastolytic papulosis is a distinct elastolytic disorder diagnosed histologically by a notable loss of elastic fibers localized to the papillary dermis.2 Fibroelastolytic papulosis is an acquired condition linked to exposure to UV radiation, abnormal elastogenesis, and hormonal factors that commonly involves the neck, supraclavicular area, and upper back.1-3 Predominantly affecting elderly women, FP is characterized by soft white papules that often coalesce into a cobblestonelike plaque.2 Because the condition rarely is seen in men, there is speculation that it may involve genetic, hereditary, and hormonal factors that have yet to be identified.1
Fibroelastolytic papulosis can be classified as either pseudoxanthoma elasticum–like papillary dermal elastolysis or white fibrous papulosis.2,3 White fibrous papulosis manifests with haphazardly arranged collagen fibers in the reticular and deep dermis with papillary dermal elastolysis and most commonly develops on the neck.3 Although our patient’s lesion was on the neck, the absence of thickened collagen bands on histology supported classification as the pseudoxanthoma elasticum– like papillary dermal elastolysis subtype.
Fibroelastolytic papulosis can be distinguished from other elastic abnormalities by its characteristic clinical appearance, demographic distribution, and associated histopathologic findings. The differential diagnosis of FP includes pseudoxanthoma elasticum (PXE), anetoderma, scleromyxedema, and lichen amyloidosis.
Pseudoxanthoma elasticum is a hereditary or acquired multisystem disease characterized by fragmentation and calcification of elastic fibers in the mid dermis.1,4 Its clinical presentation resembles that of FP, appearing as small, asymptomatic, yellowish or flesh-colored papules in a reticular pattern that progressively coalesce into larger plaques with a cobblestonelike appearance.1 Like FP, PXE commonly affects the flexural creases in women but in contrast may manifest earlier (ie, second or third decades of life). Additionally, the pathogenesis of PXE is not related to UV radiation exposure. The hereditary form develops due to a gene variation, whereas the acquired form may be due to conditions associated with physiologic and/or mechanical stress.1
Anetoderma, also known as macular atrophy, is another condition that demonstrates elastic tissue loss in the dermis on histopathology.1 Anetoderma commonly is seen in younger patients and can be differentiated from FP by the antecedent presence of an inflammatory process. Anetoderma is classified as primary or secondary. Primary anetoderma is associated with prothrombotic abnormalities, while secondary anetoderma is associated with systemic disease including but not limited to sarcoidosis, systemic lupus erythematous, and Graves disease.1
Neither lichen myxedematosus (LM) nor lichen amyloidosis (LA) are true elastolytic conditions. Lichen myxedematosus is considered in the differential diagnosis of FP due to the associated loss of elastin observed with disease progression. An idiopathic cutaneous mucinosis, LM is a localized form of scleromyxedema, which is characterized by small, firm, waxy papules; mucin deposition in the skin; fibroblast proliferation; and fibrosis. On histologic analysis, typical findings of LM include irregularly arranged fibroblasts, diffuse mucin deposition within the upper and mid reticular dermis, increased collagen deposition, and a decrease in elastin fibers.5
Lichen amyloidosis is a subtype of primary localized cutaneous amyloidosis, a rare condition characterized by the extracellular deposition of amyloid proteins in the skin and a lack of systemic involvement. Although it is not an elastolytic condition, LA is clinically similar to FP, often manifesting as multiple localized, pruritic, hyperpigmented papules that can coalesce into larger plaques; it tends to develop on the shins, calves, ankles, and thighs.6,7 The condition commonly manifests in the fifth and sixth decades of life; however, in contrast to FP, LA is more prevalent in men and individuals from Central and South American as well as Middle Eastern and non-Chinese Asian populations.8 Lichen amyloidosis is a keratin-derived amyloidosis with cytokeratin-based amyloid precursors that only deposit in the dermis.6 Histopathology reveals colloid bodies due to the presence of apoptotic basal keratinocytes. The etiology of LA is unknown, but on rare occasions it has been associated with multiple endocrine neoplasia 2A rearranged during transfection mutations.6
In summary, FP is an uncommonly diagnosed elastolytic condition that often is asymptomatic or associated with mild pruritus. Biopsy is warranted to help differentiate it from mimicker conditions that may be associated with systemic disease. Currently, there is no established therapy that provides successful treatment. Research suggests unsatisfactory results with the use of topical tretinoin or topical antioxidants.3 More recently, nonablative fractional resurfacing lasers have been evaluated as a possible therapeutic strategy of promise for elastic disorders.9
- Andrés-Ramos I, Alegría-Landa V, Gimeno I, et al. Cutaneous elastic tissue anomalies. Am J Dermatopathol. 2019;41:85-117. doi:10.1097/DAD.0000000000001275
- Valbuena V, Assaad D, Yeung J. Pseudoxanthoma elasticum-like papillary dermal elastolysis: a single case report. J Cutan Med Surg. 2017;21:345-347. doi:10.1177/1203475417699407
- Dokic Y, Tschen J. White fibrous papulosis of the axillae and neck. Cureus. 2020;12:E7635. doi:10.7759/cureus.7635
- Recio-Monescillo M, Torre-Castro J, Manzanas C, et al. Papillary dermal elastolysis histopathology mimicking folliculotropic mycosis fungoides. J Cutan Pathol. 2023;50:430-433. doi:10.1111/cup.14402
- Cokonis Georgakis CD, Falasca G, Georgakis A, et al. Scleromyxedema. Clin Dermatol. 2006;24:493-497. doi:10.1016/j.clindermatol.2006.07.011
- Weidner T, Illing T, Elsner P. Primary localized cutaneous amyloidosis: a systematic treatment review. Am J Clin Dermatol. 2017;18:629-642. doi:10.1007/s40257-017-0278-9
- Ladizinski B, Lee KC. Lichen amyloidosis. CMAJ. 2014;186:532. doi:10.1503/cmaj.130698
- Chen JF, Chen YF. Answer: can you identify this condition? Can Fam Physician. 2012;58:1234-1235.
- Foering K, Torbeck RL, Frank MP, et al. Treatment of pseudoxanthoma elasticum-like papillary dermal elastolysis with nonablative fractional resurfacing laser resulting in clinical and histologic improvement in elastin and collagen. J Cosmet Laser Ther. 2018;20:382-384. doi:10.1080/14764172.2017.1358457
The Diagnosis: Fibroelastolytic Papulosis
Histopathology demonstrated decreased density and fragmentation of elastic fibers in the superficial reticular and papillary dermis consistent with an elastolytic disease process (Figure). Of note, elastolysis typically is visualized with Verhoeff-van Gieson stain but cannot be visualized well with standard hematoxylin and eosin staining. Additional staining with Congo red was negative for amyloid, and colloidal iron did not show any increase in dermal mucin, ruling out amyloidosis and scleromyxedema, respectively. Based on the histopathologic findings and the clinical history, a diagnosis of fibroelastolytic papulosis (FP) was made. Given the benign nature of the condition, the patient was prescribed a topical steroid (clobetasol 0.05%) for symptomatic relief.

Cutaneous conditions can arise from abnormalities in the elastin composition of connective tissue due to abnormal elastin formation or degradation (elastolysis).1 Fibroelastolytic papulosis is a distinct elastolytic disorder diagnosed histologically by a notable loss of elastic fibers localized to the papillary dermis.2 Fibroelastolytic papulosis is an acquired condition linked to exposure to UV radiation, abnormal elastogenesis, and hormonal factors that commonly involves the neck, supraclavicular area, and upper back.1-3 Predominantly affecting elderly women, FP is characterized by soft white papules that often coalesce into a cobblestonelike plaque.2 Because the condition rarely is seen in men, there is speculation that it may involve genetic, hereditary, and hormonal factors that have yet to be identified.1
Fibroelastolytic papulosis can be classified as either pseudoxanthoma elasticum–like papillary dermal elastolysis or white fibrous papulosis.2,3 White fibrous papulosis manifests with haphazardly arranged collagen fibers in the reticular and deep dermis with papillary dermal elastolysis and most commonly develops on the neck.3 Although our patient’s lesion was on the neck, the absence of thickened collagen bands on histology supported classification as the pseudoxanthoma elasticum– like papillary dermal elastolysis subtype.
Fibroelastolytic papulosis can be distinguished from other elastic abnormalities by its characteristic clinical appearance, demographic distribution, and associated histopathologic findings. The differential diagnosis of FP includes pseudoxanthoma elasticum (PXE), anetoderma, scleromyxedema, and lichen amyloidosis.
Pseudoxanthoma elasticum is a hereditary or acquired multisystem disease characterized by fragmentation and calcification of elastic fibers in the mid dermis.1,4 Its clinical presentation resembles that of FP, appearing as small, asymptomatic, yellowish or flesh-colored papules in a reticular pattern that progressively coalesce into larger plaques with a cobblestonelike appearance.1 Like FP, PXE commonly affects the flexural creases in women but in contrast may manifest earlier (ie, second or third decades of life). Additionally, the pathogenesis of PXE is not related to UV radiation exposure. The hereditary form develops due to a gene variation, whereas the acquired form may be due to conditions associated with physiologic and/or mechanical stress.1
Anetoderma, also known as macular atrophy, is another condition that demonstrates elastic tissue loss in the dermis on histopathology.1 Anetoderma commonly is seen in younger patients and can be differentiated from FP by the antecedent presence of an inflammatory process. Anetoderma is classified as primary or secondary. Primary anetoderma is associated with prothrombotic abnormalities, while secondary anetoderma is associated with systemic disease including but not limited to sarcoidosis, systemic lupus erythematous, and Graves disease.1
Neither lichen myxedematosus (LM) nor lichen amyloidosis (LA) are true elastolytic conditions. Lichen myxedematosus is considered in the differential diagnosis of FP due to the associated loss of elastin observed with disease progression. An idiopathic cutaneous mucinosis, LM is a localized form of scleromyxedema, which is characterized by small, firm, waxy papules; mucin deposition in the skin; fibroblast proliferation; and fibrosis. On histologic analysis, typical findings of LM include irregularly arranged fibroblasts, diffuse mucin deposition within the upper and mid reticular dermis, increased collagen deposition, and a decrease in elastin fibers.5
Lichen amyloidosis is a subtype of primary localized cutaneous amyloidosis, a rare condition characterized by the extracellular deposition of amyloid proteins in the skin and a lack of systemic involvement. Although it is not an elastolytic condition, LA is clinically similar to FP, often manifesting as multiple localized, pruritic, hyperpigmented papules that can coalesce into larger plaques; it tends to develop on the shins, calves, ankles, and thighs.6,7 The condition commonly manifests in the fifth and sixth decades of life; however, in contrast to FP, LA is more prevalent in men and individuals from Central and South American as well as Middle Eastern and non-Chinese Asian populations.8 Lichen amyloidosis is a keratin-derived amyloidosis with cytokeratin-based amyloid precursors that only deposit in the dermis.6 Histopathology reveals colloid bodies due to the presence of apoptotic basal keratinocytes. The etiology of LA is unknown, but on rare occasions it has been associated with multiple endocrine neoplasia 2A rearranged during transfection mutations.6
In summary, FP is an uncommonly diagnosed elastolytic condition that often is asymptomatic or associated with mild pruritus. Biopsy is warranted to help differentiate it from mimicker conditions that may be associated with systemic disease. Currently, there is no established therapy that provides successful treatment. Research suggests unsatisfactory results with the use of topical tretinoin or topical antioxidants.3 More recently, nonablative fractional resurfacing lasers have been evaluated as a possible therapeutic strategy of promise for elastic disorders.9
The Diagnosis: Fibroelastolytic Papulosis
Histopathology demonstrated decreased density and fragmentation of elastic fibers in the superficial reticular and papillary dermis consistent with an elastolytic disease process (Figure). Of note, elastolysis typically is visualized with Verhoeff-van Gieson stain but cannot be visualized well with standard hematoxylin and eosin staining. Additional staining with Congo red was negative for amyloid, and colloidal iron did not show any increase in dermal mucin, ruling out amyloidosis and scleromyxedema, respectively. Based on the histopathologic findings and the clinical history, a diagnosis of fibroelastolytic papulosis (FP) was made. Given the benign nature of the condition, the patient was prescribed a topical steroid (clobetasol 0.05%) for symptomatic relief.

Cutaneous conditions can arise from abnormalities in the elastin composition of connective tissue due to abnormal elastin formation or degradation (elastolysis).1 Fibroelastolytic papulosis is a distinct elastolytic disorder diagnosed histologically by a notable loss of elastic fibers localized to the papillary dermis.2 Fibroelastolytic papulosis is an acquired condition linked to exposure to UV radiation, abnormal elastogenesis, and hormonal factors that commonly involves the neck, supraclavicular area, and upper back.1-3 Predominantly affecting elderly women, FP is characterized by soft white papules that often coalesce into a cobblestonelike plaque.2 Because the condition rarely is seen in men, there is speculation that it may involve genetic, hereditary, and hormonal factors that have yet to be identified.1
Fibroelastolytic papulosis can be classified as either pseudoxanthoma elasticum–like papillary dermal elastolysis or white fibrous papulosis.2,3 White fibrous papulosis manifests with haphazardly arranged collagen fibers in the reticular and deep dermis with papillary dermal elastolysis and most commonly develops on the neck.3 Although our patient’s lesion was on the neck, the absence of thickened collagen bands on histology supported classification as the pseudoxanthoma elasticum– like papillary dermal elastolysis subtype.
Fibroelastolytic papulosis can be distinguished from other elastic abnormalities by its characteristic clinical appearance, demographic distribution, and associated histopathologic findings. The differential diagnosis of FP includes pseudoxanthoma elasticum (PXE), anetoderma, scleromyxedema, and lichen amyloidosis.
Pseudoxanthoma elasticum is a hereditary or acquired multisystem disease characterized by fragmentation and calcification of elastic fibers in the mid dermis.1,4 Its clinical presentation resembles that of FP, appearing as small, asymptomatic, yellowish or flesh-colored papules in a reticular pattern that progressively coalesce into larger plaques with a cobblestonelike appearance.1 Like FP, PXE commonly affects the flexural creases in women but in contrast may manifest earlier (ie, second or third decades of life). Additionally, the pathogenesis of PXE is not related to UV radiation exposure. The hereditary form develops due to a gene variation, whereas the acquired form may be due to conditions associated with physiologic and/or mechanical stress.1
Anetoderma, also known as macular atrophy, is another condition that demonstrates elastic tissue loss in the dermis on histopathology.1 Anetoderma commonly is seen in younger patients and can be differentiated from FP by the antecedent presence of an inflammatory process. Anetoderma is classified as primary or secondary. Primary anetoderma is associated with prothrombotic abnormalities, while secondary anetoderma is associated with systemic disease including but not limited to sarcoidosis, systemic lupus erythematous, and Graves disease.1
Neither lichen myxedematosus (LM) nor lichen amyloidosis (LA) are true elastolytic conditions. Lichen myxedematosus is considered in the differential diagnosis of FP due to the associated loss of elastin observed with disease progression. An idiopathic cutaneous mucinosis, LM is a localized form of scleromyxedema, which is characterized by small, firm, waxy papules; mucin deposition in the skin; fibroblast proliferation; and fibrosis. On histologic analysis, typical findings of LM include irregularly arranged fibroblasts, diffuse mucin deposition within the upper and mid reticular dermis, increased collagen deposition, and a decrease in elastin fibers.5
Lichen amyloidosis is a subtype of primary localized cutaneous amyloidosis, a rare condition characterized by the extracellular deposition of amyloid proteins in the skin and a lack of systemic involvement. Although it is not an elastolytic condition, LA is clinically similar to FP, often manifesting as multiple localized, pruritic, hyperpigmented papules that can coalesce into larger plaques; it tends to develop on the shins, calves, ankles, and thighs.6,7 The condition commonly manifests in the fifth and sixth decades of life; however, in contrast to FP, LA is more prevalent in men and individuals from Central and South American as well as Middle Eastern and non-Chinese Asian populations.8 Lichen amyloidosis is a keratin-derived amyloidosis with cytokeratin-based amyloid precursors that only deposit in the dermis.6 Histopathology reveals colloid bodies due to the presence of apoptotic basal keratinocytes. The etiology of LA is unknown, but on rare occasions it has been associated with multiple endocrine neoplasia 2A rearranged during transfection mutations.6
In summary, FP is an uncommonly diagnosed elastolytic condition that often is asymptomatic or associated with mild pruritus. Biopsy is warranted to help differentiate it from mimicker conditions that may be associated with systemic disease. Currently, there is no established therapy that provides successful treatment. Research suggests unsatisfactory results with the use of topical tretinoin or topical antioxidants.3 More recently, nonablative fractional resurfacing lasers have been evaluated as a possible therapeutic strategy of promise for elastic disorders.9
- Andrés-Ramos I, Alegría-Landa V, Gimeno I, et al. Cutaneous elastic tissue anomalies. Am J Dermatopathol. 2019;41:85-117. doi:10.1097/DAD.0000000000001275
- Valbuena V, Assaad D, Yeung J. Pseudoxanthoma elasticum-like papillary dermal elastolysis: a single case report. J Cutan Med Surg. 2017;21:345-347. doi:10.1177/1203475417699407
- Dokic Y, Tschen J. White fibrous papulosis of the axillae and neck. Cureus. 2020;12:E7635. doi:10.7759/cureus.7635
- Recio-Monescillo M, Torre-Castro J, Manzanas C, et al. Papillary dermal elastolysis histopathology mimicking folliculotropic mycosis fungoides. J Cutan Pathol. 2023;50:430-433. doi:10.1111/cup.14402
- Cokonis Georgakis CD, Falasca G, Georgakis A, et al. Scleromyxedema. Clin Dermatol. 2006;24:493-497. doi:10.1016/j.clindermatol.2006.07.011
- Weidner T, Illing T, Elsner P. Primary localized cutaneous amyloidosis: a systematic treatment review. Am J Clin Dermatol. 2017;18:629-642. doi:10.1007/s40257-017-0278-9
- Ladizinski B, Lee KC. Lichen amyloidosis. CMAJ. 2014;186:532. doi:10.1503/cmaj.130698
- Chen JF, Chen YF. Answer: can you identify this condition? Can Fam Physician. 2012;58:1234-1235.
- Foering K, Torbeck RL, Frank MP, et al. Treatment of pseudoxanthoma elasticum-like papillary dermal elastolysis with nonablative fractional resurfacing laser resulting in clinical and histologic improvement in elastin and collagen. J Cosmet Laser Ther. 2018;20:382-384. doi:10.1080/14764172.2017.1358457
- Andrés-Ramos I, Alegría-Landa V, Gimeno I, et al. Cutaneous elastic tissue anomalies. Am J Dermatopathol. 2019;41:85-117. doi:10.1097/DAD.0000000000001275
- Valbuena V, Assaad D, Yeung J. Pseudoxanthoma elasticum-like papillary dermal elastolysis: a single case report. J Cutan Med Surg. 2017;21:345-347. doi:10.1177/1203475417699407
- Dokic Y, Tschen J. White fibrous papulosis of the axillae and neck. Cureus. 2020;12:E7635. doi:10.7759/cureus.7635
- Recio-Monescillo M, Torre-Castro J, Manzanas C, et al. Papillary dermal elastolysis histopathology mimicking folliculotropic mycosis fungoides. J Cutan Pathol. 2023;50:430-433. doi:10.1111/cup.14402
- Cokonis Georgakis CD, Falasca G, Georgakis A, et al. Scleromyxedema. Clin Dermatol. 2006;24:493-497. doi:10.1016/j.clindermatol.2006.07.011
- Weidner T, Illing T, Elsner P. Primary localized cutaneous amyloidosis: a systematic treatment review. Am J Clin Dermatol. 2017;18:629-642. doi:10.1007/s40257-017-0278-9
- Ladizinski B, Lee KC. Lichen amyloidosis. CMAJ. 2014;186:532. doi:10.1503/cmaj.130698
- Chen JF, Chen YF. Answer: can you identify this condition? Can Fam Physician. 2012;58:1234-1235.
- Foering K, Torbeck RL, Frank MP, et al. Treatment of pseudoxanthoma elasticum-like papillary dermal elastolysis with nonablative fractional resurfacing laser resulting in clinical and histologic improvement in elastin and collagen. J Cosmet Laser Ther. 2018;20:382-384. doi:10.1080/14764172.2017.1358457
A 76-year-old woman presented to the dermatology clinic for evaluation of a pruritic rash on the posterior lateral neck of several years’ duration. The rash had been slowly worsening and was intermittently symptomatic. Physical examination revealed monomorphous flesh-colored papules coalescing on the neck, yielding a cobblestonelike texture. The patient had been treated previously by dermatology with topical steroids, but symptoms persisted. A punch biopsy of the left lateral neck was performed.
Introduction: Health Professions Education Evaluation and Research (HPEER) Advanced Fellowship Abstracts
The original four HPEER Advanced Fellowship sites were established by the Department of Veterans Affairs (VA) Office of Academic Affiliation in 2014, and expanded in 2020 to include 8 sites and a national coordinating center with leadership shared between VA facilities in Houston and White River Junction. The VA invests heavily in training the nation’s healthcare professionals. The mission of HPEER is to develop leaders who can educate, evaluate, and innovate in Health Professions Education for the VA and the nation. All HPEER sites take part in a nationally coordinated curriculum covering topics in curriculum design, learner assessment, leadership, interprofessional education, as well as scholarship and educational research.
As part of the national HPEER curriculum covering scholarship and educational research, and in concert with Wednesday, May 14, 2025 VA Research Week 2025, HPEER organized a joint conference with the Center for Health Professions Education at the Uniformed Services University of the Health Sciences (USUHS). This interagency online event included poster sessions and oral presentations from HPEER fellows and students in USUHS certificate and graduate degree programs.
Education scholarship is broad, ranging from descriptions of curricular innovations and works in progress to advanced research using techniques drawn from psychology, sociology, anthropology, economics, and other scientific disciplines. The abstracts presented here summarize some of the work being done by HPEER fellows. Dougherty et al (Boston) described a project to create a primer outlining methodology for conducting and interpreting cost-effectiveness evaluations in the context of proposed HPE innovations. Cohen et al (Cleveland) found reduction in potentially problematic orders in the context of life-sustaining treatment following a multifaceted intervention program. Sorenson (Dublin, Georgia) reported an expanded Tai Chi program that included modifications allowing seated positions for veterans with mobility limitations. Young et al (Dublin) described an interprofessional curriculum to strengthen communication between nurses and social workers in their conversations with women veterans living in rural settings. Misedah-Robinson et al (Houston) showed that a new training program strengthened coordinators’ self-reports of preparedness and confidence in their ability to support veterans who have experienced human trafficking. Tovar et al (Salt Lake City) describe a methodology for using data from the VHA Corporate Data Warehouse to optimize schedules of HPE students assigned to VA clinical rotations. Yanez et al (San Francisco) presented initial observations of learner-centered outcomes following participation in a new multidisciplinary integrative health elective. Resto et al (West Haven) reported that implementation of self-serve kiosks increased distribution of substance use harm reduction resources beyond usual clinical care.
A second joint conference between VA HPEER and USUHS is planned for VA Research Week 2026; we look forward to the abstracts that will be produced by this new cohort of fellows, as well as to the future scholarship and contributions to the field that will be made by alumni of the HPEER Advanced Fellowship.
The original four HPEER Advanced Fellowship sites were established by the Department of Veterans Affairs (VA) Office of Academic Affiliation in 2014, and expanded in 2020 to include 8 sites and a national coordinating center with leadership shared between VA facilities in Houston and White River Junction. The VA invests heavily in training the nation’s healthcare professionals. The mission of HPEER is to develop leaders who can educate, evaluate, and innovate in Health Professions Education for the VA and the nation. All HPEER sites take part in a nationally coordinated curriculum covering topics in curriculum design, learner assessment, leadership, interprofessional education, as well as scholarship and educational research.
As part of the national HPEER curriculum covering scholarship and educational research, and in concert with Wednesday, May 14, 2025 VA Research Week 2025, HPEER organized a joint conference with the Center for Health Professions Education at the Uniformed Services University of the Health Sciences (USUHS). This interagency online event included poster sessions and oral presentations from HPEER fellows and students in USUHS certificate and graduate degree programs.
Education scholarship is broad, ranging from descriptions of curricular innovations and works in progress to advanced research using techniques drawn from psychology, sociology, anthropology, economics, and other scientific disciplines. The abstracts presented here summarize some of the work being done by HPEER fellows. Dougherty et al (Boston) described a project to create a primer outlining methodology for conducting and interpreting cost-effectiveness evaluations in the context of proposed HPE innovations. Cohen et al (Cleveland) found reduction in potentially problematic orders in the context of life-sustaining treatment following a multifaceted intervention program. Sorenson (Dublin, Georgia) reported an expanded Tai Chi program that included modifications allowing seated positions for veterans with mobility limitations. Young et al (Dublin) described an interprofessional curriculum to strengthen communication between nurses and social workers in their conversations with women veterans living in rural settings. Misedah-Robinson et al (Houston) showed that a new training program strengthened coordinators’ self-reports of preparedness and confidence in their ability to support veterans who have experienced human trafficking. Tovar et al (Salt Lake City) describe a methodology for using data from the VHA Corporate Data Warehouse to optimize schedules of HPE students assigned to VA clinical rotations. Yanez et al (San Francisco) presented initial observations of learner-centered outcomes following participation in a new multidisciplinary integrative health elective. Resto et al (West Haven) reported that implementation of self-serve kiosks increased distribution of substance use harm reduction resources beyond usual clinical care.
A second joint conference between VA HPEER and USUHS is planned for VA Research Week 2026; we look forward to the abstracts that will be produced by this new cohort of fellows, as well as to the future scholarship and contributions to the field that will be made by alumni of the HPEER Advanced Fellowship.
The original four HPEER Advanced Fellowship sites were established by the Department of Veterans Affairs (VA) Office of Academic Affiliation in 2014, and expanded in 2020 to include 8 sites and a national coordinating center with leadership shared between VA facilities in Houston and White River Junction. The VA invests heavily in training the nation’s healthcare professionals. The mission of HPEER is to develop leaders who can educate, evaluate, and innovate in Health Professions Education for the VA and the nation. All HPEER sites take part in a nationally coordinated curriculum covering topics in curriculum design, learner assessment, leadership, interprofessional education, as well as scholarship and educational research.
As part of the national HPEER curriculum covering scholarship and educational research, and in concert with Wednesday, May 14, 2025 VA Research Week 2025, HPEER organized a joint conference with the Center for Health Professions Education at the Uniformed Services University of the Health Sciences (USUHS). This interagency online event included poster sessions and oral presentations from HPEER fellows and students in USUHS certificate and graduate degree programs.
Education scholarship is broad, ranging from descriptions of curricular innovations and works in progress to advanced research using techniques drawn from psychology, sociology, anthropology, economics, and other scientific disciplines. The abstracts presented here summarize some of the work being done by HPEER fellows. Dougherty et al (Boston) described a project to create a primer outlining methodology for conducting and interpreting cost-effectiveness evaluations in the context of proposed HPE innovations. Cohen et al (Cleveland) found reduction in potentially problematic orders in the context of life-sustaining treatment following a multifaceted intervention program. Sorenson (Dublin, Georgia) reported an expanded Tai Chi program that included modifications allowing seated positions for veterans with mobility limitations. Young et al (Dublin) described an interprofessional curriculum to strengthen communication between nurses and social workers in their conversations with women veterans living in rural settings. Misedah-Robinson et al (Houston) showed that a new training program strengthened coordinators’ self-reports of preparedness and confidence in their ability to support veterans who have experienced human trafficking. Tovar et al (Salt Lake City) describe a methodology for using data from the VHA Corporate Data Warehouse to optimize schedules of HPE students assigned to VA clinical rotations. Yanez et al (San Francisco) presented initial observations of learner-centered outcomes following participation in a new multidisciplinary integrative health elective. Resto et al (West Haven) reported that implementation of self-serve kiosks increased distribution of substance use harm reduction resources beyond usual clinical care.
A second joint conference between VA HPEER and USUHS is planned for VA Research Week 2026; we look forward to the abstracts that will be produced by this new cohort of fellows, as well as to the future scholarship and contributions to the field that will be made by alumni of the HPEER Advanced Fellowship.
Development and Implementation of an Anti-Human Trafficking Education for Veterans and Clinicians
Background
Veterans may have a greater risk of experiencing human trafficking (HT) than the general population because of social aspects of health, including housing insecurity, justice involvement, food insecurity, and adverse childhood events.1-4 Since 2023, the U.S. Department of Veterans Affairs (VA) has explored veterans’ experiences of HT through the Anti-Human Trafficking (AHT) Pilot Project. This quality improvement project evaluated: 1) development of clinician AHT training materials to enhance identification and response to Veterans experiencing HT, and 2) educational resources aimed at raising awareness tailored to veterans and clinicians.
Methods
South Central Mental Illness Research, Education and Clinical Center (SCMIRECC) facilitated two focus group discussions with AHT coordinators implementing the pilot at six sites. Based on discussions and leadership input, SCMIRECC developed a training curriculum, with bi-weekly readings culminating in a two-hour workshop. Training evaluation followed Kirkpatrick’s model using questions adapted from the Provider Responses, Treatment, and Care for Trafficked People (PROTECT) Survey.5,6 Veteran-facing materials, including a brochure and whiteboard video, were reviewed by two Veteran Consumer Advisory Boards (CAB). The brochures, whiteboard video, and awareness modules were developed and revised based on feedback from focus group discussions. VA Central Office cleared all materials.
Results
Coordinators were satisfied with the training (mean, 4.20). After the training, none of the coordinators (n = 6) felt unprepared to assist Veterans (pre-training mean, 2.25; post-training mean, 1.40), and confidence in documentation improved (pre-training mean, 3.00; post-training mean, 3.40). Veteran CAB members recommended simplified language and veteran-centered messaging. The coordinators found the brochures and training useful. Recommendations included adding more representation to brochure covers, advanced training, a list of commonly asked questions, and a simplified screening tool. Barriers included delays in material development due to language guidance under recent executive orders.
Conclusions
The AHT training improved coordinators’ preparedness and confidence in supporting Veterans with trafficking experiences. Feedback emphasized the value of concise, Veteran-centered materials and a practical HT screening tool. These findings support the continued implementation of AHT education across VA settings to enhance identification and response for Veterans at risk of HT.
- US Department of Veterans Affairs, Veterans Health Administration. Annual Report 2023 Veterans Health Administration Homeless Programs Office.
- Tsai J, Kasprow WJ, Rosenheck RA. Alcohol and drug use disorders among homeless veterans: prevalence and association with supported housing outcomes. Addict Behav. 2014;39(2):455-460. doi:10.1016/j.addbeh.2013.02.002
- Wang EA, McGinnis KA, Goulet J, et al. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268. doi:10.1177/003335491513000313
- Blosnich JR, Garfin DR, Maguen S, et al. Differences in childhood adversity, suicidal ideation, and suicide attempt among veterans and nonveterans. Am Psychol. 2021;76(2):284-299. doi:10.1037/amp0000755
- Kirkpatrick D. Great ideas revisited. Training & Development. 1996;50(1):54-60.
- Ross C, Dimitrova S, Howard LM, Dewey M, Zimmerman C, Oram S. Human trafficking and health: a cross-sectional survey of NHS professionals' contact with victims of human trafficking. BMJ Open. 2015;5(8):e008682. Published 2015 Aug 20. doi:10.1136/bmjopen-2015-008682
Background
Veterans may have a greater risk of experiencing human trafficking (HT) than the general population because of social aspects of health, including housing insecurity, justice involvement, food insecurity, and adverse childhood events.1-4 Since 2023, the U.S. Department of Veterans Affairs (VA) has explored veterans’ experiences of HT through the Anti-Human Trafficking (AHT) Pilot Project. This quality improvement project evaluated: 1) development of clinician AHT training materials to enhance identification and response to Veterans experiencing HT, and 2) educational resources aimed at raising awareness tailored to veterans and clinicians.
Methods
South Central Mental Illness Research, Education and Clinical Center (SCMIRECC) facilitated two focus group discussions with AHT coordinators implementing the pilot at six sites. Based on discussions and leadership input, SCMIRECC developed a training curriculum, with bi-weekly readings culminating in a two-hour workshop. Training evaluation followed Kirkpatrick’s model using questions adapted from the Provider Responses, Treatment, and Care for Trafficked People (PROTECT) Survey.5,6 Veteran-facing materials, including a brochure and whiteboard video, were reviewed by two Veteran Consumer Advisory Boards (CAB). The brochures, whiteboard video, and awareness modules were developed and revised based on feedback from focus group discussions. VA Central Office cleared all materials.
Results
Coordinators were satisfied with the training (mean, 4.20). After the training, none of the coordinators (n = 6) felt unprepared to assist Veterans (pre-training mean, 2.25; post-training mean, 1.40), and confidence in documentation improved (pre-training mean, 3.00; post-training mean, 3.40). Veteran CAB members recommended simplified language and veteran-centered messaging. The coordinators found the brochures and training useful. Recommendations included adding more representation to brochure covers, advanced training, a list of commonly asked questions, and a simplified screening tool. Barriers included delays in material development due to language guidance under recent executive orders.
Conclusions
The AHT training improved coordinators’ preparedness and confidence in supporting Veterans with trafficking experiences. Feedback emphasized the value of concise, Veteran-centered materials and a practical HT screening tool. These findings support the continued implementation of AHT education across VA settings to enhance identification and response for Veterans at risk of HT.
Background
Veterans may have a greater risk of experiencing human trafficking (HT) than the general population because of social aspects of health, including housing insecurity, justice involvement, food insecurity, and adverse childhood events.1-4 Since 2023, the U.S. Department of Veterans Affairs (VA) has explored veterans’ experiences of HT through the Anti-Human Trafficking (AHT) Pilot Project. This quality improvement project evaluated: 1) development of clinician AHT training materials to enhance identification and response to Veterans experiencing HT, and 2) educational resources aimed at raising awareness tailored to veterans and clinicians.
Methods
South Central Mental Illness Research, Education and Clinical Center (SCMIRECC) facilitated two focus group discussions with AHT coordinators implementing the pilot at six sites. Based on discussions and leadership input, SCMIRECC developed a training curriculum, with bi-weekly readings culminating in a two-hour workshop. Training evaluation followed Kirkpatrick’s model using questions adapted from the Provider Responses, Treatment, and Care for Trafficked People (PROTECT) Survey.5,6 Veteran-facing materials, including a brochure and whiteboard video, were reviewed by two Veteran Consumer Advisory Boards (CAB). The brochures, whiteboard video, and awareness modules were developed and revised based on feedback from focus group discussions. VA Central Office cleared all materials.
Results
Coordinators were satisfied with the training (mean, 4.20). After the training, none of the coordinators (n = 6) felt unprepared to assist Veterans (pre-training mean, 2.25; post-training mean, 1.40), and confidence in documentation improved (pre-training mean, 3.00; post-training mean, 3.40). Veteran CAB members recommended simplified language and veteran-centered messaging. The coordinators found the brochures and training useful. Recommendations included adding more representation to brochure covers, advanced training, a list of commonly asked questions, and a simplified screening tool. Barriers included delays in material development due to language guidance under recent executive orders.
Conclusions
The AHT training improved coordinators’ preparedness and confidence in supporting Veterans with trafficking experiences. Feedback emphasized the value of concise, Veteran-centered materials and a practical HT screening tool. These findings support the continued implementation of AHT education across VA settings to enhance identification and response for Veterans at risk of HT.
- US Department of Veterans Affairs, Veterans Health Administration. Annual Report 2023 Veterans Health Administration Homeless Programs Office.
- Tsai J, Kasprow WJ, Rosenheck RA. Alcohol and drug use disorders among homeless veterans: prevalence and association with supported housing outcomes. Addict Behav. 2014;39(2):455-460. doi:10.1016/j.addbeh.2013.02.002
- Wang EA, McGinnis KA, Goulet J, et al. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268. doi:10.1177/003335491513000313
- Blosnich JR, Garfin DR, Maguen S, et al. Differences in childhood adversity, suicidal ideation, and suicide attempt among veterans and nonveterans. Am Psychol. 2021;76(2):284-299. doi:10.1037/amp0000755
- Kirkpatrick D. Great ideas revisited. Training & Development. 1996;50(1):54-60.
- Ross C, Dimitrova S, Howard LM, Dewey M, Zimmerman C, Oram S. Human trafficking and health: a cross-sectional survey of NHS professionals' contact with victims of human trafficking. BMJ Open. 2015;5(8):e008682. Published 2015 Aug 20. doi:10.1136/bmjopen-2015-008682
- US Department of Veterans Affairs, Veterans Health Administration. Annual Report 2023 Veterans Health Administration Homeless Programs Office.
- Tsai J, Kasprow WJ, Rosenheck RA. Alcohol and drug use disorders among homeless veterans: prevalence and association with supported housing outcomes. Addict Behav. 2014;39(2):455-460. doi:10.1016/j.addbeh.2013.02.002
- Wang EA, McGinnis KA, Goulet J, et al. Food insecurity and health: data from the Veterans Aging Cohort Study. Public Health Rep. 2015;130(3):261-268. doi:10.1177/003335491513000313
- Blosnich JR, Garfin DR, Maguen S, et al. Differences in childhood adversity, suicidal ideation, and suicide attempt among veterans and nonveterans. Am Psychol. 2021;76(2):284-299. doi:10.1037/amp0000755
- Kirkpatrick D. Great ideas revisited. Training & Development. 1996;50(1):54-60.
- Ross C, Dimitrova S, Howard LM, Dewey M, Zimmerman C, Oram S. Human trafficking and health: a cross-sectional survey of NHS professionals' contact with victims of human trafficking. BMJ Open. 2015;5(8):e008682. Published 2015 Aug 20. doi:10.1136/bmjopen-2015-008682
Developing a Multi-Disciplinary Integrative Health Elective at the San Francisco VA
Background
Integrative health (IH) combines conventional and complementary medicine in a coordinated, evidence-based approach to treat the whole person. Nearly 40% of American adults have used complementary health approaches,1 yet IH exposure in medical training is limited. In 2022, the San Francisco VA Health Care Center launched a multidisciplinary clinical IH elective for University of California San Francisco (UCSF) internal medicine and SFVA nurse practitioner residents. Based on findings from a general and targeted needs assessment, including faculty and learner feedback, we found that the elective was well-received, but relied on one-on-one patient-based teaching. This structure created variable learning experiences and high faculty burden. Our project aims to formalize and evaluate the IH elective curriculum to better address the needs of both faculty and learners.
Methods
We used Kern’s six-step framework for curriculum development. To reduce variability, we sought to formalize the core curricular content by: 1) reviewing existing elective components, comparing them to similar curricula nationwide, and outlining foundational knowledge based on the exam domains of the American Board of Integrative Medicine (ABOIM);2 2) creating eleven learning objectives across three themes: patient-centered care, systems-based practice, and IH-specific knowledge; 3) developing IH subspecialty experience guides to standardize clinical teaching with suggested takeaways, guided reflection, and curated resources. To reduce faculty burden, we consolidated elective resources into a centralized e-learning hub. Trainees complete a pre/post self-assessment and evaluation at the end of the elective.
Results
We identified key learning opportunities in each IH shadowing experience to enhance learners’ knowledge. We developed an IH e-Learning Hub to provide easy access to elective materials and IH clinical tools. Evaluations from the first two learners who completed the elective indicate that the learning objectives were met and that learners gained increased knowledge of lifestyle medicine, mind-body medicine, manual medicine, and botanicals/dietary supplements. Learners valued increased IH subspecialty familiarity and reported high likelihood of future practice change.
Discussion
The project is ongoing. Next steps include collecting faculty evaluations about their experience, continuing to create and refine experience guides, promoting clinical tools for learner’s future practice, and developing strategies to recruit more learners to the elective.
- Nahin RL, Rhee A, Stussman B. Use of Complementary Health Approaches Overall and for Pain Management by US Adults. JAMA. 2024;331(7):613-615. doi:10.1001/jama.2023.26775
- Integrative medicine exam description. American Board of Physician Specialties. Updated July 2021. Accessed December 12, 2025. https://www.abpsus.org/integrative-medicine-description
Background
Integrative health (IH) combines conventional and complementary medicine in a coordinated, evidence-based approach to treat the whole person. Nearly 40% of American adults have used complementary health approaches,1 yet IH exposure in medical training is limited. In 2022, the San Francisco VA Health Care Center launched a multidisciplinary clinical IH elective for University of California San Francisco (UCSF) internal medicine and SFVA nurse practitioner residents. Based on findings from a general and targeted needs assessment, including faculty and learner feedback, we found that the elective was well-received, but relied on one-on-one patient-based teaching. This structure created variable learning experiences and high faculty burden. Our project aims to formalize and evaluate the IH elective curriculum to better address the needs of both faculty and learners.
Methods
We used Kern’s six-step framework for curriculum development. To reduce variability, we sought to formalize the core curricular content by: 1) reviewing existing elective components, comparing them to similar curricula nationwide, and outlining foundational knowledge based on the exam domains of the American Board of Integrative Medicine (ABOIM);2 2) creating eleven learning objectives across three themes: patient-centered care, systems-based practice, and IH-specific knowledge; 3) developing IH subspecialty experience guides to standardize clinical teaching with suggested takeaways, guided reflection, and curated resources. To reduce faculty burden, we consolidated elective resources into a centralized e-learning hub. Trainees complete a pre/post self-assessment and evaluation at the end of the elective.
Results
We identified key learning opportunities in each IH shadowing experience to enhance learners’ knowledge. We developed an IH e-Learning Hub to provide easy access to elective materials and IH clinical tools. Evaluations from the first two learners who completed the elective indicate that the learning objectives were met and that learners gained increased knowledge of lifestyle medicine, mind-body medicine, manual medicine, and botanicals/dietary supplements. Learners valued increased IH subspecialty familiarity and reported high likelihood of future practice change.
Discussion
The project is ongoing. Next steps include collecting faculty evaluations about their experience, continuing to create and refine experience guides, promoting clinical tools for learner’s future practice, and developing strategies to recruit more learners to the elective.
Background
Integrative health (IH) combines conventional and complementary medicine in a coordinated, evidence-based approach to treat the whole person. Nearly 40% of American adults have used complementary health approaches,1 yet IH exposure in medical training is limited. In 2022, the San Francisco VA Health Care Center launched a multidisciplinary clinical IH elective for University of California San Francisco (UCSF) internal medicine and SFVA nurse practitioner residents. Based on findings from a general and targeted needs assessment, including faculty and learner feedback, we found that the elective was well-received, but relied on one-on-one patient-based teaching. This structure created variable learning experiences and high faculty burden. Our project aims to formalize and evaluate the IH elective curriculum to better address the needs of both faculty and learners.
Methods
We used Kern’s six-step framework for curriculum development. To reduce variability, we sought to formalize the core curricular content by: 1) reviewing existing elective components, comparing them to similar curricula nationwide, and outlining foundational knowledge based on the exam domains of the American Board of Integrative Medicine (ABOIM);2 2) creating eleven learning objectives across three themes: patient-centered care, systems-based practice, and IH-specific knowledge; 3) developing IH subspecialty experience guides to standardize clinical teaching with suggested takeaways, guided reflection, and curated resources. To reduce faculty burden, we consolidated elective resources into a centralized e-learning hub. Trainees complete a pre/post self-assessment and evaluation at the end of the elective.
Results
We identified key learning opportunities in each IH shadowing experience to enhance learners’ knowledge. We developed an IH e-Learning Hub to provide easy access to elective materials and IH clinical tools. Evaluations from the first two learners who completed the elective indicate that the learning objectives were met and that learners gained increased knowledge of lifestyle medicine, mind-body medicine, manual medicine, and botanicals/dietary supplements. Learners valued increased IH subspecialty familiarity and reported high likelihood of future practice change.
Discussion
The project is ongoing. Next steps include collecting faculty evaluations about their experience, continuing to create and refine experience guides, promoting clinical tools for learner’s future practice, and developing strategies to recruit more learners to the elective.
- Nahin RL, Rhee A, Stussman B. Use of Complementary Health Approaches Overall and for Pain Management by US Adults. JAMA. 2024;331(7):613-615. doi:10.1001/jama.2023.26775
- Integrative medicine exam description. American Board of Physician Specialties. Updated July 2021. Accessed December 12, 2025. https://www.abpsus.org/integrative-medicine-description
- Nahin RL, Rhee A, Stussman B. Use of Complementary Health Approaches Overall and for Pain Management by US Adults. JAMA. 2024;331(7):613-615. doi:10.1001/jama.2023.26775
- Integrative medicine exam description. American Board of Physician Specialties. Updated July 2021. Accessed December 12, 2025. https://www.abpsus.org/integrative-medicine-description
Tai Chi Modification and Supplemental Movements Quality Improvement Program
Background
The original program consisted of 12 movements that were to be split up between 3 weeks teaching 4 movements each week. Range of mobility was the main consideration for developing this HPE quality improvement project. Veterans who wanted to participate in Tai Chi were not able to engage in the activity due to the range of movement traditional Tai Chi required.
Innovation
The HPE Quality Improvement program developed a 15-movement warm-up, 12 co-ordinational movements consistent with the original program, 18 supplemental Tai Chi movements that were not included in the original program all of which focus on movements remaining below the shoulders and can be done standing or sitting. Four advanced exercises including “hip over heel” were included to target participants balance if able and to improve their hip strength, knee tendon/ligament strength. Tai Chi loses its potential to increase balance when performed in a sitting position.1 The movements drew upon Fu style Tai Chi and the program developer was given permission from Tommy Kirchoff to use his DVD Healing Exercises. The HPE program consisted of four 30–60-minute weekly sessions of learning the movements with another 4 weekly sessions of demonstrating the movements. Instructors were given written and visual documents to learn from and were evaluated by the developer during the last 4 weeks.
.
Results
Qualitative Data: Instructors notice a difference in how they feel, and appreciate having another option to offer veterans with mobility/standing issues. Patients expressed improvement in mobility relating to bending, arm extension, arm raising, muscle strengthening, hip strengthening and rotation.
Discussion
Future research will want to look at taking measurements before and after patient implementation to determine quantitative data related to balance, strength and range of movement including grip strength, stand up and go, and one-legged stands.
- Skelton DA, Mavroeidi A. How do muscle and bone strengthening and balance activities (MBSBA) vary across the life course, and are there particular ages where MBSBA are most important?. J Frailty Sarcopenia Falls. 2018;3(2):74-84. Published 2018 Jun 1. doi:10.22540/JFSF-03-074
Background
The original program consisted of 12 movements that were to be split up between 3 weeks teaching 4 movements each week. Range of mobility was the main consideration for developing this HPE quality improvement project. Veterans who wanted to participate in Tai Chi were not able to engage in the activity due to the range of movement traditional Tai Chi required.
Innovation
The HPE Quality Improvement program developed a 15-movement warm-up, 12 co-ordinational movements consistent with the original program, 18 supplemental Tai Chi movements that were not included in the original program all of which focus on movements remaining below the shoulders and can be done standing or sitting. Four advanced exercises including “hip over heel” were included to target participants balance if able and to improve their hip strength, knee tendon/ligament strength. Tai Chi loses its potential to increase balance when performed in a sitting position.1 The movements drew upon Fu style Tai Chi and the program developer was given permission from Tommy Kirchoff to use his DVD Healing Exercises. The HPE program consisted of four 30–60-minute weekly sessions of learning the movements with another 4 weekly sessions of demonstrating the movements. Instructors were given written and visual documents to learn from and were evaluated by the developer during the last 4 weeks.
.
Results
Qualitative Data: Instructors notice a difference in how they feel, and appreciate having another option to offer veterans with mobility/standing issues. Patients expressed improvement in mobility relating to bending, arm extension, arm raising, muscle strengthening, hip strengthening and rotation.
Discussion
Future research will want to look at taking measurements before and after patient implementation to determine quantitative data related to balance, strength and range of movement including grip strength, stand up and go, and one-legged stands.
Background
The original program consisted of 12 movements that were to be split up between 3 weeks teaching 4 movements each week. Range of mobility was the main consideration for developing this HPE quality improvement project. Veterans who wanted to participate in Tai Chi were not able to engage in the activity due to the range of movement traditional Tai Chi required.
Innovation
The HPE Quality Improvement program developed a 15-movement warm-up, 12 co-ordinational movements consistent with the original program, 18 supplemental Tai Chi movements that were not included in the original program all of which focus on movements remaining below the shoulders and can be done standing or sitting. Four advanced exercises including “hip over heel” were included to target participants balance if able and to improve their hip strength, knee tendon/ligament strength. Tai Chi loses its potential to increase balance when performed in a sitting position.1 The movements drew upon Fu style Tai Chi and the program developer was given permission from Tommy Kirchoff to use his DVD Healing Exercises. The HPE program consisted of four 30–60-minute weekly sessions of learning the movements with another 4 weekly sessions of demonstrating the movements. Instructors were given written and visual documents to learn from and were evaluated by the developer during the last 4 weeks.
.
Results
Qualitative Data: Instructors notice a difference in how they feel, and appreciate having another option to offer veterans with mobility/standing issues. Patients expressed improvement in mobility relating to bending, arm extension, arm raising, muscle strengthening, hip strengthening and rotation.
Discussion
Future research will want to look at taking measurements before and after patient implementation to determine quantitative data related to balance, strength and range of movement including grip strength, stand up and go, and one-legged stands.
- Skelton DA, Mavroeidi A. How do muscle and bone strengthening and balance activities (MBSBA) vary across the life course, and are there particular ages where MBSBA are most important?. J Frailty Sarcopenia Falls. 2018;3(2):74-84. Published 2018 Jun 1. doi:10.22540/JFSF-03-074
- Skelton DA, Mavroeidi A. How do muscle and bone strengthening and balance activities (MBSBA) vary across the life course, and are there particular ages where MBSBA are most important?. J Frailty Sarcopenia Falls. 2018;3(2):74-84. Published 2018 Jun 1. doi:10.22540/JFSF-03-074
A Health Educator’s Primer to Cost-Effectiveness in Health Professions Education
Background
Cost-effectiveness (CE) evaluations, for existing and anticipated programs, are common in healthcare, but are rarely used in health professions education (HPE). A systematic review of HPE literature found not only few examples of CE evaluations, but also unclear and inconsistent methodology.1 One proposed reason HPE has been slow to adopt CE evaluations is uncertainty over terminology and how to adapt this methodology to HPE.2 CE evaluations present further challenges for HPE since educational outcomes are often not easily monetized. However, given the reality of constrained budgets and limited resources, CE evaluations can be a powerful tool for educators to strengthen arguments for proposed innovations, and for scholars seeking to conduct rigorous work that sustains critical review.
Innovation
This project aims to make CE evaluations more understandable to HPE educators, using a one-page infographic and glossary. This will provide a primer, operationalizing the steps involved in CE evaluations and addressing why and when CE evaluations might be considered in HPE. To improve comprehension, this is being developed collaboratively with health professions educators and an economist. This infographic will be submitted for publication, as a resource to facilitate educators’ scholarly work and conversations with fiscal administrators.
Results
The infographic includes 1) an overview of CE evaluations, 2) information about inputs required for CE evaluations, 3) guidance on interpreting results, 4) a glossary of key terminology, and 5) considerations for why educators might consider this type of analysis. A final draft will be pilot tested with a focus group to assess interdisciplinary accessibility.
Discussion
Discussions between health professions educators and an economist on this infographic uncovered concepts that were poorly understood or defined differently across disciplines, determining specific knowledge gaps and misunderstandings. For example, facilitating conversation between educators and economists highlighted key terms that were a source of misunderstanding. These were then added to the glossary, creating a shared vocabulary. This also helped clarify the steps and information necessary for conducting CE evaluations in HPE, particularly the issue of perspective choice for the analysis (educator, patient, learner, etc.). Overall, this collaboration aimed at making CE evaluations more approachable and understandable for HPE professionals through this infographic.
- Foo J, Cook DA, Walsh K, et al. Cost evaluations in health professions education: a systematic review of methods and reporting quality. Med Educ. 2019;53(12):1196-1208. doi:10.1111/medu.13936
- Maloney S, Reeves S, Rivers G, Ilic D, Foo J, Walsh K. The Prato Statement on cost and value in professional and interprofessional education. J Interprof Care. 2017;31(1):1-4. doi:10.1080/13561820.2016.1257255
Background
Cost-effectiveness (CE) evaluations, for existing and anticipated programs, are common in healthcare, but are rarely used in health professions education (HPE). A systematic review of HPE literature found not only few examples of CE evaluations, but also unclear and inconsistent methodology.1 One proposed reason HPE has been slow to adopt CE evaluations is uncertainty over terminology and how to adapt this methodology to HPE.2 CE evaluations present further challenges for HPE since educational outcomes are often not easily monetized. However, given the reality of constrained budgets and limited resources, CE evaluations can be a powerful tool for educators to strengthen arguments for proposed innovations, and for scholars seeking to conduct rigorous work that sustains critical review.
Innovation
This project aims to make CE evaluations more understandable to HPE educators, using a one-page infographic and glossary. This will provide a primer, operationalizing the steps involved in CE evaluations and addressing why and when CE evaluations might be considered in HPE. To improve comprehension, this is being developed collaboratively with health professions educators and an economist. This infographic will be submitted for publication, as a resource to facilitate educators’ scholarly work and conversations with fiscal administrators.
Results
The infographic includes 1) an overview of CE evaluations, 2) information about inputs required for CE evaluations, 3) guidance on interpreting results, 4) a glossary of key terminology, and 5) considerations for why educators might consider this type of analysis. A final draft will be pilot tested with a focus group to assess interdisciplinary accessibility.
Discussion
Discussions between health professions educators and an economist on this infographic uncovered concepts that were poorly understood or defined differently across disciplines, determining specific knowledge gaps and misunderstandings. For example, facilitating conversation between educators and economists highlighted key terms that were a source of misunderstanding. These were then added to the glossary, creating a shared vocabulary. This also helped clarify the steps and information necessary for conducting CE evaluations in HPE, particularly the issue of perspective choice for the analysis (educator, patient, learner, etc.). Overall, this collaboration aimed at making CE evaluations more approachable and understandable for HPE professionals through this infographic.
Background
Cost-effectiveness (CE) evaluations, for existing and anticipated programs, are common in healthcare, but are rarely used in health professions education (HPE). A systematic review of HPE literature found not only few examples of CE evaluations, but also unclear and inconsistent methodology.1 One proposed reason HPE has been slow to adopt CE evaluations is uncertainty over terminology and how to adapt this methodology to HPE.2 CE evaluations present further challenges for HPE since educational outcomes are often not easily monetized. However, given the reality of constrained budgets and limited resources, CE evaluations can be a powerful tool for educators to strengthen arguments for proposed innovations, and for scholars seeking to conduct rigorous work that sustains critical review.
Innovation
This project aims to make CE evaluations more understandable to HPE educators, using a one-page infographic and glossary. This will provide a primer, operationalizing the steps involved in CE evaluations and addressing why and when CE evaluations might be considered in HPE. To improve comprehension, this is being developed collaboratively with health professions educators and an economist. This infographic will be submitted for publication, as a resource to facilitate educators’ scholarly work and conversations with fiscal administrators.
Results
The infographic includes 1) an overview of CE evaluations, 2) information about inputs required for CE evaluations, 3) guidance on interpreting results, 4) a glossary of key terminology, and 5) considerations for why educators might consider this type of analysis. A final draft will be pilot tested with a focus group to assess interdisciplinary accessibility.
Discussion
Discussions between health professions educators and an economist on this infographic uncovered concepts that were poorly understood or defined differently across disciplines, determining specific knowledge gaps and misunderstandings. For example, facilitating conversation between educators and economists highlighted key terms that were a source of misunderstanding. These were then added to the glossary, creating a shared vocabulary. This also helped clarify the steps and information necessary for conducting CE evaluations in HPE, particularly the issue of perspective choice for the analysis (educator, patient, learner, etc.). Overall, this collaboration aimed at making CE evaluations more approachable and understandable for HPE professionals through this infographic.
- Foo J, Cook DA, Walsh K, et al. Cost evaluations in health professions education: a systematic review of methods and reporting quality. Med Educ. 2019;53(12):1196-1208. doi:10.1111/medu.13936
- Maloney S, Reeves S, Rivers G, Ilic D, Foo J, Walsh K. The Prato Statement on cost and value in professional and interprofessional education. J Interprof Care. 2017;31(1):1-4. doi:10.1080/13561820.2016.1257255
- Foo J, Cook DA, Walsh K, et al. Cost evaluations in health professions education: a systematic review of methods and reporting quality. Med Educ. 2019;53(12):1196-1208. doi:10.1111/medu.13936
- Maloney S, Reeves S, Rivers G, Ilic D, Foo J, Walsh K. The Prato Statement on cost and value in professional and interprofessional education. J Interprof Care. 2017;31(1):1-4. doi:10.1080/13561820.2016.1257255
Not a Professional Degree? A New Federal Policy Could Exacerbate the Nursing Shortage
The statistics are shocking: 138,000 registered nurses (RNs) have left the workforce since 2022 and at least 40% plan to retire or leave the profession in the next 5 years — and new updates from the Department of Education could make the national nursing crisis even worse.
The reason? Nursing is no longer considered a professional degree.
A recent Department of Education rulemaking session omitted advanced nursing programs (as well as physician assistance programs, physical therapy, occupational therapy, audiology, social work, and public health programs) from the definition of professional degrees and limited the amount of student loan funding available to pursue advanced practice degrees like Master of Science in Nursing and Doctor of Nursing Practice.
“We have a primary care crisis in this country,” said Deborah Trautman PhD, RN, president and chief executive officer of the American Association of Colleges of Nursing (AACN). “The omission is not only harmful for nursing; the omission is not good for anyone who needs healthcare.”
Limiting Loan Access
The One Big, Beautiful Bill Act eliminated the Grad PLUS student loan program and amended the list of professional degrees to exclude advanced practice nursing. Although the change doesn’t affect the licensure or legal standing of nurses, it alters access to financial aid and limits advanced education opportunities.
Starting on July 1, 2026, graduate students will be limited to a total of $100,000 in federal student loans, a decrease from the previous cap of $138,500 but loan caps for graduate students in professional degree programs will increase to $200,000. The changes led the National Association of Student Financial Aid Administrators to declare, “Many will be shut out of graduate education.”
“It would force people who need loan support and don’t have a sufficient amount through a federal loan to seek [private loans], but federal loans have better interest rates and/or other conditions, and some students may not qualify for the private loans,” Trautman said. “The risk then is that students may not pursue these advanced nursing degrees because of the financial barriers that they will face.”
The Department of Education disagrees. In a statement, the federal department said, “Placing a cap on loans will push the remaining graduate nursing programs to reduce their program costs, ensuring that nurses will not be saddled with unmanageable student loan debt.” So far, Trautman has seen “no evidence” that limiting access to advanced nursing programs would reduce tuition costs.
Industry-Wide Impacts
Trautman worries that omitting nursing from the list of professional degrees will reduce access to care.
Nurse practitioners are providing primary care in rural and underserved areas; certified registered nurse anesthetists make up more than 50% of anesthesia providers in the US (a number that jumps to 80% in rural areas); and the percentage of births attended by certified nurse midwives is growing fast.
“These are nurses…who are working to achieve better patient outcomes and to make the health system work better for all of us,” Trautman said. “And we would be compromising this workforce that is so critical to our nation.”
Limiting the federal student loan borrowing cap for advanced nursing degrees could also exacerbate the nursing faculty shortage. In 2023, more than 65,000 qualified applicants were denied admission to baccalaureate and graduate nursing programs; insufficient number of faculty was the top reason.
Colleges depend on nurses with advanced degrees to fill faculty vacancies. In fact, more than 80% of open positions required or preferred a doctoral degree, according to AACN. Removing nursing from the list of professional degree programs and limiting access to student loans will make it even harder to fill vacancies, limiting the number of new nurses entering the profession.
“We’re finalizing the results of [a new national survey] that showed overwhelming feedback from our member deans and students who believe enrollment in advanced nursing programs is going be impacted,” said Trautman. “We’re going to see the faculty shortage worsen; we’re going see increased financial burdens to our students, and we believe it’s going to undermine the stability of the healthcare workforce.”
Industry associations, including the American Nurses Association, American Academy of Nursing, and American Organization for Nursing Leadership have released statements opposing the change and advocating for graduate nursing degrees to be added to the list of professional programs. Trautman hopes that public pressure and cross-sector support will lead the Department of Education to reverse its current position.
“It’s the wrong decision,” she said. “There is an opportunity to make this right, and that is to include nursing on that professional list.”
A version of this article first appeared on Medscape.com.
The statistics are shocking: 138,000 registered nurses (RNs) have left the workforce since 2022 and at least 40% plan to retire or leave the profession in the next 5 years — and new updates from the Department of Education could make the national nursing crisis even worse.
The reason? Nursing is no longer considered a professional degree.
A recent Department of Education rulemaking session omitted advanced nursing programs (as well as physician assistance programs, physical therapy, occupational therapy, audiology, social work, and public health programs) from the definition of professional degrees and limited the amount of student loan funding available to pursue advanced practice degrees like Master of Science in Nursing and Doctor of Nursing Practice.
“We have a primary care crisis in this country,” said Deborah Trautman PhD, RN, president and chief executive officer of the American Association of Colleges of Nursing (AACN). “The omission is not only harmful for nursing; the omission is not good for anyone who needs healthcare.”
Limiting Loan Access
The One Big, Beautiful Bill Act eliminated the Grad PLUS student loan program and amended the list of professional degrees to exclude advanced practice nursing. Although the change doesn’t affect the licensure or legal standing of nurses, it alters access to financial aid and limits advanced education opportunities.
Starting on July 1, 2026, graduate students will be limited to a total of $100,000 in federal student loans, a decrease from the previous cap of $138,500 but loan caps for graduate students in professional degree programs will increase to $200,000. The changes led the National Association of Student Financial Aid Administrators to declare, “Many will be shut out of graduate education.”
“It would force people who need loan support and don’t have a sufficient amount through a federal loan to seek [private loans], but federal loans have better interest rates and/or other conditions, and some students may not qualify for the private loans,” Trautman said. “The risk then is that students may not pursue these advanced nursing degrees because of the financial barriers that they will face.”
The Department of Education disagrees. In a statement, the federal department said, “Placing a cap on loans will push the remaining graduate nursing programs to reduce their program costs, ensuring that nurses will not be saddled with unmanageable student loan debt.” So far, Trautman has seen “no evidence” that limiting access to advanced nursing programs would reduce tuition costs.
Industry-Wide Impacts
Trautman worries that omitting nursing from the list of professional degrees will reduce access to care.
Nurse practitioners are providing primary care in rural and underserved areas; certified registered nurse anesthetists make up more than 50% of anesthesia providers in the US (a number that jumps to 80% in rural areas); and the percentage of births attended by certified nurse midwives is growing fast.
“These are nurses…who are working to achieve better patient outcomes and to make the health system work better for all of us,” Trautman said. “And we would be compromising this workforce that is so critical to our nation.”
Limiting the federal student loan borrowing cap for advanced nursing degrees could also exacerbate the nursing faculty shortage. In 2023, more than 65,000 qualified applicants were denied admission to baccalaureate and graduate nursing programs; insufficient number of faculty was the top reason.
Colleges depend on nurses with advanced degrees to fill faculty vacancies. In fact, more than 80% of open positions required or preferred a doctoral degree, according to AACN. Removing nursing from the list of professional degree programs and limiting access to student loans will make it even harder to fill vacancies, limiting the number of new nurses entering the profession.
“We’re finalizing the results of [a new national survey] that showed overwhelming feedback from our member deans and students who believe enrollment in advanced nursing programs is going be impacted,” said Trautman. “We’re going to see the faculty shortage worsen; we’re going see increased financial burdens to our students, and we believe it’s going to undermine the stability of the healthcare workforce.”
Industry associations, including the American Nurses Association, American Academy of Nursing, and American Organization for Nursing Leadership have released statements opposing the change and advocating for graduate nursing degrees to be added to the list of professional programs. Trautman hopes that public pressure and cross-sector support will lead the Department of Education to reverse its current position.
“It’s the wrong decision,” she said. “There is an opportunity to make this right, and that is to include nursing on that professional list.”
A version of this article first appeared on Medscape.com.
The statistics are shocking: 138,000 registered nurses (RNs) have left the workforce since 2022 and at least 40% plan to retire or leave the profession in the next 5 years — and new updates from the Department of Education could make the national nursing crisis even worse.
The reason? Nursing is no longer considered a professional degree.
A recent Department of Education rulemaking session omitted advanced nursing programs (as well as physician assistance programs, physical therapy, occupational therapy, audiology, social work, and public health programs) from the definition of professional degrees and limited the amount of student loan funding available to pursue advanced practice degrees like Master of Science in Nursing and Doctor of Nursing Practice.
“We have a primary care crisis in this country,” said Deborah Trautman PhD, RN, president and chief executive officer of the American Association of Colleges of Nursing (AACN). “The omission is not only harmful for nursing; the omission is not good for anyone who needs healthcare.”
Limiting Loan Access
The One Big, Beautiful Bill Act eliminated the Grad PLUS student loan program and amended the list of professional degrees to exclude advanced practice nursing. Although the change doesn’t affect the licensure or legal standing of nurses, it alters access to financial aid and limits advanced education opportunities.
Starting on July 1, 2026, graduate students will be limited to a total of $100,000 in federal student loans, a decrease from the previous cap of $138,500 but loan caps for graduate students in professional degree programs will increase to $200,000. The changes led the National Association of Student Financial Aid Administrators to declare, “Many will be shut out of graduate education.”
“It would force people who need loan support and don’t have a sufficient amount through a federal loan to seek [private loans], but federal loans have better interest rates and/or other conditions, and some students may not qualify for the private loans,” Trautman said. “The risk then is that students may not pursue these advanced nursing degrees because of the financial barriers that they will face.”
The Department of Education disagrees. In a statement, the federal department said, “Placing a cap on loans will push the remaining graduate nursing programs to reduce their program costs, ensuring that nurses will not be saddled with unmanageable student loan debt.” So far, Trautman has seen “no evidence” that limiting access to advanced nursing programs would reduce tuition costs.
Industry-Wide Impacts
Trautman worries that omitting nursing from the list of professional degrees will reduce access to care.
Nurse practitioners are providing primary care in rural and underserved areas; certified registered nurse anesthetists make up more than 50% of anesthesia providers in the US (a number that jumps to 80% in rural areas); and the percentage of births attended by certified nurse midwives is growing fast.
“These are nurses…who are working to achieve better patient outcomes and to make the health system work better for all of us,” Trautman said. “And we would be compromising this workforce that is so critical to our nation.”
Limiting the federal student loan borrowing cap for advanced nursing degrees could also exacerbate the nursing faculty shortage. In 2023, more than 65,000 qualified applicants were denied admission to baccalaureate and graduate nursing programs; insufficient number of faculty was the top reason.
Colleges depend on nurses with advanced degrees to fill faculty vacancies. In fact, more than 80% of open positions required or preferred a doctoral degree, according to AACN. Removing nursing from the list of professional degree programs and limiting access to student loans will make it even harder to fill vacancies, limiting the number of new nurses entering the profession.
“We’re finalizing the results of [a new national survey] that showed overwhelming feedback from our member deans and students who believe enrollment in advanced nursing programs is going be impacted,” said Trautman. “We’re going to see the faculty shortage worsen; we’re going see increased financial burdens to our students, and we believe it’s going to undermine the stability of the healthcare workforce.”
Industry associations, including the American Nurses Association, American Academy of Nursing, and American Organization for Nursing Leadership have released statements opposing the change and advocating for graduate nursing degrees to be added to the list of professional programs. Trautman hopes that public pressure and cross-sector support will lead the Department of Education to reverse its current position.
“It’s the wrong decision,” she said. “There is an opportunity to make this right, and that is to include nursing on that professional list.”
A version of this article first appeared on Medscape.com.
Interview Tips for Dermatology Applicants From Dr. Scott Worswick
What qualities are dermatology programs looking for that may be different from 5 years ago?
DR. WORSWICK: Every dermatology residency program is different, and as a result, each program is looking for different qualities in its applicants. Overall, I don’t think there has been a huge change in what programs are generally looking for, though. While each program may have a particular trait it values more than another, in general, programs are looking to find residents who will be competent and caring doctors, who work well in teams, and who could be future leaders in our field.
What are common mistakes you see in dermatology residency interviews, and how can applicants avoid them?
DR. WORSWICK: Most dermatology applicants are highly accomplished and empathic soon-to-be physicians, so I haven’t found a lot of “mistakes” from this incredible group of people that we have the privilege of interviewing. From time to time, an applicant will lie in an interview, usually out of a desire to appear to be a certain way, and occasionally, they may be nervous and stumble over their words. The former is a really big problem when it happens, and I would recommend that applicants be honest in all their encounters. The latter is not a major problem, and in some cases, might be avoided by lots of practice in advance.
What types of questions do you recommend applicants ask their interviewers to demonstrate genuine interest in the program?
DR. WORSWICK: Because of the signaling system, I think that programs assume interest at baseline once an applicant has sent the signal. So, “demonstrating interest” is generally not something I would recommend to applicants during the interview day. It is important for applicants to determine on interview day if a program is a fit for them, so applicants should showcase their unique strengths and skills and find out about what makes any given program different from another. The match generally works well and gets applicants into a program that closely aligns with their strengths and interests. So, think of interview day as your time to figure out how good a fit a program is for you, and not the other way around.
How can applicants who feel they don't have standout research or leadership credentials differentiate themselves in the interview?
DR. WORSWICK: While leadership, and less so research experience, is a trait valued highly by most if not all dermatology programs, it is only a part of what an applicant can offer a program. Most programs employ holistic review and consider several factors, probably most commonly grades in medical school, leadership experience, mentorship, teaching, volunteering, Step 2 scores, and letters of recommendation. Any given applicant does not need to excel in all of these. If an applicant has not done a lot of research, they may not match into a research-heavy program, but it doesn’t mean they won’t match. They should determine in which areas they shine and signal the programs that align with those interests/strengths.
How should applicants discuss nontraditional experiences in a way that adds value rather than raising red flags?
DR. WORSWICK: In general, my recommendation would be to explain what happened leading up to the change or challenge so that someone reading the application clearly understands the circumstances of the experience, then add value to the description by explaining what was learned and how this might relate to the applicant being a dermatology resident. For example, if a resident took time off for financial reasons and had to work as a medical assitant for a year, a concise description that explains the need for the leave (financial) as well as what value was gained (a year of hands-on patient care experience that validated their choice of going into medicine) could be very helpful.
What qualities are dermatology programs looking for that may be different from 5 years ago?
DR. WORSWICK: Every dermatology residency program is different, and as a result, each program is looking for different qualities in its applicants. Overall, I don’t think there has been a huge change in what programs are generally looking for, though. While each program may have a particular trait it values more than another, in general, programs are looking to find residents who will be competent and caring doctors, who work well in teams, and who could be future leaders in our field.
What are common mistakes you see in dermatology residency interviews, and how can applicants avoid them?
DR. WORSWICK: Most dermatology applicants are highly accomplished and empathic soon-to-be physicians, so I haven’t found a lot of “mistakes” from this incredible group of people that we have the privilege of interviewing. From time to time, an applicant will lie in an interview, usually out of a desire to appear to be a certain way, and occasionally, they may be nervous and stumble over their words. The former is a really big problem when it happens, and I would recommend that applicants be honest in all their encounters. The latter is not a major problem, and in some cases, might be avoided by lots of practice in advance.
What types of questions do you recommend applicants ask their interviewers to demonstrate genuine interest in the program?
DR. WORSWICK: Because of the signaling system, I think that programs assume interest at baseline once an applicant has sent the signal. So, “demonstrating interest” is generally not something I would recommend to applicants during the interview day. It is important for applicants to determine on interview day if a program is a fit for them, so applicants should showcase their unique strengths and skills and find out about what makes any given program different from another. The match generally works well and gets applicants into a program that closely aligns with their strengths and interests. So, think of interview day as your time to figure out how good a fit a program is for you, and not the other way around.
How can applicants who feel they don't have standout research or leadership credentials differentiate themselves in the interview?
DR. WORSWICK: While leadership, and less so research experience, is a trait valued highly by most if not all dermatology programs, it is only a part of what an applicant can offer a program. Most programs employ holistic review and consider several factors, probably most commonly grades in medical school, leadership experience, mentorship, teaching, volunteering, Step 2 scores, and letters of recommendation. Any given applicant does not need to excel in all of these. If an applicant has not done a lot of research, they may not match into a research-heavy program, but it doesn’t mean they won’t match. They should determine in which areas they shine and signal the programs that align with those interests/strengths.
How should applicants discuss nontraditional experiences in a way that adds value rather than raising red flags?
DR. WORSWICK: In general, my recommendation would be to explain what happened leading up to the change or challenge so that someone reading the application clearly understands the circumstances of the experience, then add value to the description by explaining what was learned and how this might relate to the applicant being a dermatology resident. For example, if a resident took time off for financial reasons and had to work as a medical assitant for a year, a concise description that explains the need for the leave (financial) as well as what value was gained (a year of hands-on patient care experience that validated their choice of going into medicine) could be very helpful.
What qualities are dermatology programs looking for that may be different from 5 years ago?
DR. WORSWICK: Every dermatology residency program is different, and as a result, each program is looking for different qualities in its applicants. Overall, I don’t think there has been a huge change in what programs are generally looking for, though. While each program may have a particular trait it values more than another, in general, programs are looking to find residents who will be competent and caring doctors, who work well in teams, and who could be future leaders in our field.
What are common mistakes you see in dermatology residency interviews, and how can applicants avoid them?
DR. WORSWICK: Most dermatology applicants are highly accomplished and empathic soon-to-be physicians, so I haven’t found a lot of “mistakes” from this incredible group of people that we have the privilege of interviewing. From time to time, an applicant will lie in an interview, usually out of a desire to appear to be a certain way, and occasionally, they may be nervous and stumble over their words. The former is a really big problem when it happens, and I would recommend that applicants be honest in all their encounters. The latter is not a major problem, and in some cases, might be avoided by lots of practice in advance.
What types of questions do you recommend applicants ask their interviewers to demonstrate genuine interest in the program?
DR. WORSWICK: Because of the signaling system, I think that programs assume interest at baseline once an applicant has sent the signal. So, “demonstrating interest” is generally not something I would recommend to applicants during the interview day. It is important for applicants to determine on interview day if a program is a fit for them, so applicants should showcase their unique strengths and skills and find out about what makes any given program different from another. The match generally works well and gets applicants into a program that closely aligns with their strengths and interests. So, think of interview day as your time to figure out how good a fit a program is for you, and not the other way around.
How can applicants who feel they don't have standout research or leadership credentials differentiate themselves in the interview?
DR. WORSWICK: While leadership, and less so research experience, is a trait valued highly by most if not all dermatology programs, it is only a part of what an applicant can offer a program. Most programs employ holistic review and consider several factors, probably most commonly grades in medical school, leadership experience, mentorship, teaching, volunteering, Step 2 scores, and letters of recommendation. Any given applicant does not need to excel in all of these. If an applicant has not done a lot of research, they may not match into a research-heavy program, but it doesn’t mean they won’t match. They should determine in which areas they shine and signal the programs that align with those interests/strengths.
How should applicants discuss nontraditional experiences in a way that adds value rather than raising red flags?
DR. WORSWICK: In general, my recommendation would be to explain what happened leading up to the change or challenge so that someone reading the application clearly understands the circumstances of the experience, then add value to the description by explaining what was learned and how this might relate to the applicant being a dermatology resident. For example, if a resident took time off for financial reasons and had to work as a medical assitant for a year, a concise description that explains the need for the leave (financial) as well as what value was gained (a year of hands-on patient care experience that validated their choice of going into medicine) could be very helpful.
Millipede Burns: An Unusual Cause of Purplish Toes
To the Editor:
Millipedes do not have nearly as many feet as their name would suggest; most have fewer than 100.1 They are not actually insects; they are a wormlike arthropod in the Diplopoda class. Generally these harmless animals can be a welcome resident in gardens because they break down decaying plant material and rejuvenate the soil.1 However, they are less welcome in the home or underfoot because of what happens when these invertebrates are threatened or crushed.2
Millipedes, which typically have at least 30 pairs of legs, have 2 defense mechanisms: (1) body coiling to withstand external pressure, and (2) secretion of fluids with insecticidal properties from specialized glands distributed along their body.3 These secretions, which are used by the millipede to defend against predators, contain organic compounds including benzoquinone. When these secretions come into contact with skin, pigmentary changes resembling a burn or necrosis and irritation to the skin (pain, burning, itching) occur.4,5
Millipedes typically are found in tropical and temperate regions worldwide, such as the Amazon rainforest, Southeast Asia, tropical areas of Africa, forests, grasslands, and gardens in North America and Europe.6 They also are found in every US state as well as Puerto Rico.1 Millipedes are nocturnal, favor dark places, and can make their way into residential areas, including homes, basements, gardens, and yards.2,6 Although millipede burns commonly are reported in tropical regions, we present a case in China.6A 33-year-old woman presented with purplish-red discoloration on all 5 toes on the left foot. The patient recounted that she discovered a millipede in her shoe earlier in the day, removed it, and crushed it with her bare foot. That night, while taking a bath, she noticed that the toes had turned purplish-red (Figure 1). The patient brought the crushed millipede with her to the emergency department where she sought treatment. The dermatologist confirmed that it was a millipede; however, the team was unable to determine the specific species because it had been crushed (Figure 2).

Physical examination of the affected toes showed a clear boundary and iodinelike staining. The patient did not report pain. The stained skin had a normal temperature, pulse, texture, and sensation. Dermoscopy revealed multiple black-brown patches on the toes (Figure 3). The pigmented area gradually faded over a 1-month period. Superficial damage to the toenail revealed evidence of black-brown pigmentation on both the nail and the skin underneath. The diagnosis in the dermoscopy report suggested exogenous pigmentation of the toes. The patient was advised that no treatment was needed and that the condition would resolve on its own. At 1-month follow-up, the patient’s toes had returned to their normal color (Figure 4).
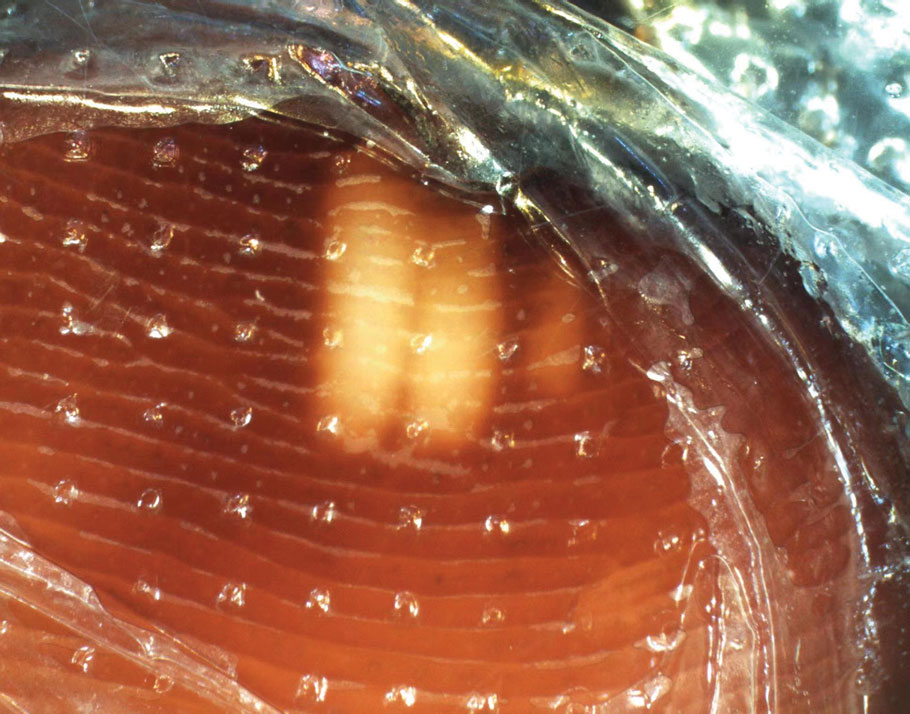
The feet are common sites of millipede burns; other exposed areas, such as the arms, face, and eyes, also are potential sites of involvement.5 The cutaneous pigmentary changes seen on our patient’s foot were a result of the millipede’s defense mechanism—secreted toxic chemicals that stained the foot. It is important to note that the pigmentation was not associated with the death of the millipede, as the millipede was still alive upon initial contact with the patient’s foot in her shoe.
When a patient presents with pigmentary changes, several conditions must be ruled out—notably acute arterial thrombosis. Patients with this condition will describe acute pain and weakness in the area of involvement. Physicians inspecting the area will note coldness and pallor in the affected limb as well as a diminished or absent pulse. In severe cases, the skin may exhibit a purplish-red appearance.5 Millipede burns also should be distinguished from bacterial endocarditis and cryoglobulinemia.7 All 3 conditions can manifest with redness, swelling, blisters, and purpuralike changes. Positive blood culture is an important diagnostic basis for bacterial endocarditis; in addition, routine blood tests will demonstrate a decrease in red blood cells and hemoglobin, and routine urinalysis may show proteinuria and microscopic hematuria. Patients with cryoglobulinemia will have a positive cryoglobulin assay, increased IgM, and often decreased complement.7 It also is worth noting that millipede burns might resemble child abuse in pediatric patients, necessitating further evaluation.5
It is unusual to see a millipede burn in nontropical regions. Therefore, the identification of our patient’s millipede burn was notable and serves as a reminder to keep this diagnosis in the differential when caring for patients with pigmentary changes. An accurate diagnosis hinges on being alert to a millipede exposure history and recognizing the clinical manifestations. For affected patients, it may be beneficial to recommend they advise friends and relatives to avoid skin contact with millipedes and most importantly to avoid stepping on them with bare feet.
Millipedes. National Wildlife Federation. Accessed October 15, 2025. https://www.nwf.org/Educational-Resources/Wildlife-Guide/Invertebrates/Millipedes
Pennini SN, Rebello PFB, Guerra MdGVB, et al. Millipede accident with unusual dermatological lesion. An Bras Dermatol. 2019;94:765-767. doi:10.1016/j.abd.2019.10.003
Lima CAJ, Cardoso JLC, Magela A, et al. Exogenous pigmentation in toes feigning ischemia of the extremities: a diagnostic challenge brought by arthropods of the Diplopoda Class (“millipedes“). An Bras Dermatol. 2010;85:391-392. doi:10.1590/s0365-05962910000300018
De Capitani EM, Vieira RJ, Bucaretchi F, et al. Human accidents involving Rhinocricus spp., a common millipede genus observed in urban areas of Brazil. Clin Toxicol (Phila). 2011;49:187-190. doi:10.3109/15563650.2011.560855
Lacy FA, Elston DM. What’s eating you? millipede burns. Cutis. 2019;103:195-196.
Neto ASH, Filho FB, Martins G. Skin lesions simulating blue toe syndrome caused by prolonged contact with a millipede. Rev Soc Bras Med Trop. 2014;47:257-258. doi:10.1590/0037-8682-0212-2013
Sampaio FMS, Valviesse VRGdA, Lyra-da-Silva JO, et al. Pain and hyperpigmentation of the toes: a quiz. hyperpigmentation of the toes caused by millipedes. Acta Derm Venereol. 2014;94:253-254. doi:10.2340/00015555-1645
To the Editor:
Millipedes do not have nearly as many feet as their name would suggest; most have fewer than 100.1 They are not actually insects; they are a wormlike arthropod in the Diplopoda class. Generally these harmless animals can be a welcome resident in gardens because they break down decaying plant material and rejuvenate the soil.1 However, they are less welcome in the home or underfoot because of what happens when these invertebrates are threatened or crushed.2
Millipedes, which typically have at least 30 pairs of legs, have 2 defense mechanisms: (1) body coiling to withstand external pressure, and (2) secretion of fluids with insecticidal properties from specialized glands distributed along their body.3 These secretions, which are used by the millipede to defend against predators, contain organic compounds including benzoquinone. When these secretions come into contact with skin, pigmentary changes resembling a burn or necrosis and irritation to the skin (pain, burning, itching) occur.4,5
Millipedes typically are found in tropical and temperate regions worldwide, such as the Amazon rainforest, Southeast Asia, tropical areas of Africa, forests, grasslands, and gardens in North America and Europe.6 They also are found in every US state as well as Puerto Rico.1 Millipedes are nocturnal, favor dark places, and can make their way into residential areas, including homes, basements, gardens, and yards.2,6 Although millipede burns commonly are reported in tropical regions, we present a case in China.6A 33-year-old woman presented with purplish-red discoloration on all 5 toes on the left foot. The patient recounted that she discovered a millipede in her shoe earlier in the day, removed it, and crushed it with her bare foot. That night, while taking a bath, she noticed that the toes had turned purplish-red (Figure 1). The patient brought the crushed millipede with her to the emergency department where she sought treatment. The dermatologist confirmed that it was a millipede; however, the team was unable to determine the specific species because it had been crushed (Figure 2).


Physical examination of the affected toes showed a clear boundary and iodinelike staining. The patient did not report pain. The stained skin had a normal temperature, pulse, texture, and sensation. Dermoscopy revealed multiple black-brown patches on the toes (Figure 3). The pigmented area gradually faded over a 1-month period. Superficial damage to the toenail revealed evidence of black-brown pigmentation on both the nail and the skin underneath. The diagnosis in the dermoscopy report suggested exogenous pigmentation of the toes. The patient was advised that no treatment was needed and that the condition would resolve on its own. At 1-month follow-up, the patient’s toes had returned to their normal color (Figure 4).


The feet are common sites of millipede burns; other exposed areas, such as the arms, face, and eyes, also are potential sites of involvement.5 The cutaneous pigmentary changes seen on our patient’s foot were a result of the millipede’s defense mechanism—secreted toxic chemicals that stained the foot. It is important to note that the pigmentation was not associated with the death of the millipede, as the millipede was still alive upon initial contact with the patient’s foot in her shoe.
When a patient presents with pigmentary changes, several conditions must be ruled out—notably acute arterial thrombosis. Patients with this condition will describe acute pain and weakness in the area of involvement. Physicians inspecting the area will note coldness and pallor in the affected limb as well as a diminished or absent pulse. In severe cases, the skin may exhibit a purplish-red appearance.5 Millipede burns also should be distinguished from bacterial endocarditis and cryoglobulinemia.7 All 3 conditions can manifest with redness, swelling, blisters, and purpuralike changes. Positive blood culture is an important diagnostic basis for bacterial endocarditis; in addition, routine blood tests will demonstrate a decrease in red blood cells and hemoglobin, and routine urinalysis may show proteinuria and microscopic hematuria. Patients with cryoglobulinemia will have a positive cryoglobulin assay, increased IgM, and often decreased complement.7 It also is worth noting that millipede burns might resemble child abuse in pediatric patients, necessitating further evaluation.5
It is unusual to see a millipede burn in nontropical regions. Therefore, the identification of our patient’s millipede burn was notable and serves as a reminder to keep this diagnosis in the differential when caring for patients with pigmentary changes. An accurate diagnosis hinges on being alert to a millipede exposure history and recognizing the clinical manifestations. For affected patients, it may be beneficial to recommend they advise friends and relatives to avoid skin contact with millipedes and most importantly to avoid stepping on them with bare feet.
To the Editor:
Millipedes do not have nearly as many feet as their name would suggest; most have fewer than 100.1 They are not actually insects; they are a wormlike arthropod in the Diplopoda class. Generally these harmless animals can be a welcome resident in gardens because they break down decaying plant material and rejuvenate the soil.1 However, they are less welcome in the home or underfoot because of what happens when these invertebrates are threatened or crushed.2
Millipedes, which typically have at least 30 pairs of legs, have 2 defense mechanisms: (1) body coiling to withstand external pressure, and (2) secretion of fluids with insecticidal properties from specialized glands distributed along their body.3 These secretions, which are used by the millipede to defend against predators, contain organic compounds including benzoquinone. When these secretions come into contact with skin, pigmentary changes resembling a burn or necrosis and irritation to the skin (pain, burning, itching) occur.4,5
Millipedes typically are found in tropical and temperate regions worldwide, such as the Amazon rainforest, Southeast Asia, tropical areas of Africa, forests, grasslands, and gardens in North America and Europe.6 They also are found in every US state as well as Puerto Rico.1 Millipedes are nocturnal, favor dark places, and can make their way into residential areas, including homes, basements, gardens, and yards.2,6 Although millipede burns commonly are reported in tropical regions, we present a case in China.6A 33-year-old woman presented with purplish-red discoloration on all 5 toes on the left foot. The patient recounted that she discovered a millipede in her shoe earlier in the day, removed it, and crushed it with her bare foot. That night, while taking a bath, she noticed that the toes had turned purplish-red (Figure 1). The patient brought the crushed millipede with her to the emergency department where she sought treatment. The dermatologist confirmed that it was a millipede; however, the team was unable to determine the specific species because it had been crushed (Figure 2).


Physical examination of the affected toes showed a clear boundary and iodinelike staining. The patient did not report pain. The stained skin had a normal temperature, pulse, texture, and sensation. Dermoscopy revealed multiple black-brown patches on the toes (Figure 3). The pigmented area gradually faded over a 1-month period. Superficial damage to the toenail revealed evidence of black-brown pigmentation on both the nail and the skin underneath. The diagnosis in the dermoscopy report suggested exogenous pigmentation of the toes. The patient was advised that no treatment was needed and that the condition would resolve on its own. At 1-month follow-up, the patient’s toes had returned to their normal color (Figure 4).


The feet are common sites of millipede burns; other exposed areas, such as the arms, face, and eyes, also are potential sites of involvement.5 The cutaneous pigmentary changes seen on our patient’s foot were a result of the millipede’s defense mechanism—secreted toxic chemicals that stained the foot. It is important to note that the pigmentation was not associated with the death of the millipede, as the millipede was still alive upon initial contact with the patient’s foot in her shoe.
When a patient presents with pigmentary changes, several conditions must be ruled out—notably acute arterial thrombosis. Patients with this condition will describe acute pain and weakness in the area of involvement. Physicians inspecting the area will note coldness and pallor in the affected limb as well as a diminished or absent pulse. In severe cases, the skin may exhibit a purplish-red appearance.5 Millipede burns also should be distinguished from bacterial endocarditis and cryoglobulinemia.7 All 3 conditions can manifest with redness, swelling, blisters, and purpuralike changes. Positive blood culture is an important diagnostic basis for bacterial endocarditis; in addition, routine blood tests will demonstrate a decrease in red blood cells and hemoglobin, and routine urinalysis may show proteinuria and microscopic hematuria. Patients with cryoglobulinemia will have a positive cryoglobulin assay, increased IgM, and often decreased complement.7 It also is worth noting that millipede burns might resemble child abuse in pediatric patients, necessitating further evaluation.5
It is unusual to see a millipede burn in nontropical regions. Therefore, the identification of our patient’s millipede burn was notable and serves as a reminder to keep this diagnosis in the differential when caring for patients with pigmentary changes. An accurate diagnosis hinges on being alert to a millipede exposure history and recognizing the clinical manifestations. For affected patients, it may be beneficial to recommend they advise friends and relatives to avoid skin contact with millipedes and most importantly to avoid stepping on them with bare feet.
Millipedes. National Wildlife Federation. Accessed October 15, 2025. https://www.nwf.org/Educational-Resources/Wildlife-Guide/Invertebrates/Millipedes
Pennini SN, Rebello PFB, Guerra MdGVB, et al. Millipede accident with unusual dermatological lesion. An Bras Dermatol. 2019;94:765-767. doi:10.1016/j.abd.2019.10.003
Lima CAJ, Cardoso JLC, Magela A, et al. Exogenous pigmentation in toes feigning ischemia of the extremities: a diagnostic challenge brought by arthropods of the Diplopoda Class (“millipedes“). An Bras Dermatol. 2010;85:391-392. doi:10.1590/s0365-05962910000300018
De Capitani EM, Vieira RJ, Bucaretchi F, et al. Human accidents involving Rhinocricus spp., a common millipede genus observed in urban areas of Brazil. Clin Toxicol (Phila). 2011;49:187-190. doi:10.3109/15563650.2011.560855
Lacy FA, Elston DM. What’s eating you? millipede burns. Cutis. 2019;103:195-196.
Neto ASH, Filho FB, Martins G. Skin lesions simulating blue toe syndrome caused by prolonged contact with a millipede. Rev Soc Bras Med Trop. 2014;47:257-258. doi:10.1590/0037-8682-0212-2013
Sampaio FMS, Valviesse VRGdA, Lyra-da-Silva JO, et al. Pain and hyperpigmentation of the toes: a quiz. hyperpigmentation of the toes caused by millipedes. Acta Derm Venereol. 2014;94:253-254. doi:10.2340/00015555-1645
Millipedes. National Wildlife Federation. Accessed October 15, 2025. https://www.nwf.org/Educational-Resources/Wildlife-Guide/Invertebrates/Millipedes
Pennini SN, Rebello PFB, Guerra MdGVB, et al. Millipede accident with unusual dermatological lesion. An Bras Dermatol. 2019;94:765-767. doi:10.1016/j.abd.2019.10.003
Lima CAJ, Cardoso JLC, Magela A, et al. Exogenous pigmentation in toes feigning ischemia of the extremities: a diagnostic challenge brought by arthropods of the Diplopoda Class (“millipedes“). An Bras Dermatol. 2010;85:391-392. doi:10.1590/s0365-05962910000300018
De Capitani EM, Vieira RJ, Bucaretchi F, et al. Human accidents involving Rhinocricus spp., a common millipede genus observed in urban areas of Brazil. Clin Toxicol (Phila). 2011;49:187-190. doi:10.3109/15563650.2011.560855
Lacy FA, Elston DM. What’s eating you? millipede burns. Cutis. 2019;103:195-196.
Neto ASH, Filho FB, Martins G. Skin lesions simulating blue toe syndrome caused by prolonged contact with a millipede. Rev Soc Bras Med Trop. 2014;47:257-258. doi:10.1590/0037-8682-0212-2013
Sampaio FMS, Valviesse VRGdA, Lyra-da-Silva JO, et al. Pain and hyperpigmentation of the toes: a quiz. hyperpigmentation of the toes caused by millipedes. Acta Derm Venereol. 2014;94:253-254. doi:10.2340/00015555-1645
PRACTICE POINTS
- Millipede burns can resemble ischemia. The most common site of a millipede burn is the feet.
- Diagnosing a millipede burn hinges on obtaining a detailed history, viewing the site under a dermatoscope, and carefully assessing the temperature and pulse of the affected area.
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.

Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.


Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
Practice Gap
Poly-L-lactic acid is approved by the US Food and Drug Administration for addressing fat loss due to HAART in patients with HIV.2,3 When used as a dermal filler for correction of facial lipoatrophy, PLLA is well tolerated and has been shown to improve quality of life.2,3 Poly-L-lactic acid is available for clinical use as microparticles of lyophilized alpha hydroxy acid polymers. Once injected (after the carrier substance is absorbed), PLLA induces an inflammatory response that ultimately leads to the production of new collagen.3 Unfortunately, PLLA microparticles often obstruct needles and make the product difficult to use, potentially hindering effective injection; thus, it is in the best interest of the patient to mitigate needle obstruction during this procedure. In this article, we describe a simple and effective way to mitigate this problem by utilizing a water bath to warm the filler prior to injection.
Technique
The required supplies include a thermostatic water bath, reconstituted PLLA, a syringe, and a 26-gauge injection needle. Because laboratory-grade heated water baths typically cost between $300 and $3000,4 we recommend using a more affordable, commercially available thermostatic water bath (eg, baby bottle warmer)(Figure 1) to warm the filler prior to injection, as the optimal temperature for this technique can still be achieved while remaining cost effective. Vials of PLLA reconstituted with 7 mL of sterile water and 2 mL lidocaine hydrochloride 1% should be labeled with the date of reconstitution and manually agitated for 30 seconds. The reconstituted product should be stored for 24 hours to ensure even suspension and powder saturation.5 On the day of the procedure, the vial should be placed into the water bath (heated to 100 °C) for 10 minutes prior to injection (Figure 2) and agitated again immediately before withdrawal into the syringe. The clinician then should sterilize the rubber top and draw the product from the warmed vial using the same size needle that will be used for injection. Although a larger gauge needle may make drawing up the product easier in typical practice, drawing and injecting with the same gauge needle helps prevent larger particles from clogging a smaller injection needle. Using a 26-gauge injection needle for withdrawal further reduces clogging by serving as a filter to prevent larger product particles from entering the injection syringe. The vials of PLLA can be kept in the water bath throughout the procedure between uses to keep the filler at a consistent temperature.


Practice Implications
Although many clinicians reduce needle obstructions by warming PLLA before injection, a published protocol currently is not available. One consideration when utilizing this technique is the limited data on the clinical stability and efficacy of PLLA at varying temperatures. Two studies recommend bringing the reconstituted vial to room temperature prior to injection, while others have documented an endothermic melting point in the range of 120 °C to 180
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
- James J, Carruthers A, Carruthers J. HIV-associated facial lipoatrophy. Dermatol Surg. 2002;28:979-986. doi:10.1046/j.1524-4725.2002.02099.x
- Duracinsky M, Leclercq P, Herrmann S, et al. Safety of poly-L-lactic acid (New-Fill®) in the treatment of facial lipoatrophy: a large observational study among HIV-positive patients. BMC Infect Dis. 2014;14:474. doi:10.1186/1471-2334-14-474
- Sickles CK, Nassereddin A, Patel P, et al. Poly-L-lactic acid. StatPearls [Internet]. Updated February 28, 2024. Accessed October 31, 2025. https://www.ncbi.nlm.nih.gov/books/NBK507871/
- Laboratory equipment: Water bath. Global Lab Supply. (n.d.). http://www.globallabsupply.com/Water-Bath-s/2122.htm
- Lin MJ, Dubin DP, Goldberg DJ, et al. Practices in the usage and reconstitution of poly-L-lactic acid. J Drugs Dermatol. 2019;18:880-886.
- Vleggaar D, Fitzgerald R, Lorenc ZP, et al. Consensus recommendations on the use of injectable poly-L-lactic acid for facial and nonfacial volumization. J Drugs Dermatol. 2014;13:s44-51.
- Sedush NG, Kalinin KT, Azarkevich PN, et al. Physicochemical characteristics and hydrolytic degradation of polylactic acid dermal fillers: a comparative study. Cosmetics. 2023;10:110. doi:10.3390/cosmetics10040110
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction
Poly-L-Lactic Acid Reconstitution Technique to Reduce Needle Obstruction