User login
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
A professional is someone who can do his best work when he doesn’t feel like it.
Alistair Cooke
When I was a young person with no idea about growing up to be something, my father used to tell me there were 4 learned professions: medicine to heal the body, law to protect the body politic, teaching to nurture the mind, and the clergy to care for the soul.1 That adage, or some version of it, is attributed to a variety of sources, likely because it captures something essential and timeless about the learned professions. I write this as a much older person, and it has been my privilege to have worked in some capacity in all 4 of these venerable vocations.
There are many more recognized professions now than in my father’s time with new ones still emerging as the world becomes more complicated and specialized. In November 2025, however, the growth of the professions was dealt a serious blow when the US Department of Education (DOE) redefined what constitutes a profession for the purpose of federal funding of graduate degrees.2 The internet is understandably abuzz with opinions across the political spectrum. What is missing from many of these discussions is an understanding of the criteria for a profession and, even more importantly, who has the authority to decide when an individual or a group has met that standard.
But first, what and why did the DOE make this change? The One Big Beautiful Bill Act charged the DOE with reducing what it claims is massive overspending on graduate education by limiting the programs that meet the definition of a “professional degree” eligible for higher funding. Of my father’s 4, medicine (including dentistry) and law made the cut with students in those professions able to borrow up to $200,000 in direct unsubsidized student loans while those in other programs would be limited to $100,000.2
As one of the oldest and most respected professions in America, nursing has received the most media attention, yet there are also other important and valued professions that are missing from the DOE list.3 The excluded professions also include: physician assistants, physical therapists, audiologists, architects, accountants, educators, and social workers. The proposed regulatory changes are not yet finalized and Congressional representatives, health care experts, and a myriad of professional associations have rightly objected the reclassification will only worsen the critical shortage of nurses, teachers, and other helping professions the country is already facing.4
There are thousands of federal health care professionals who worked long and hard to achieve their goals whom this Act undervalues. Moreover, the regulatory change leaves many students enrolled in education and training programs under federal practice auspices confused and overwhelmed. Perhaps they can take some hope and inspiration from the recognition that historically and philosophically, no agency or administration can unilaterally define what is a profession.
The literature on professionalism is voluminous, in large part because it has been surprisingly difficult to reach a consensus definition. A proposed definition from scholars captures most of the key aspects of a profession. While it is drawn from the medical literature, it applies to most of the caring professions the DOE disqualified. For pedagogic purposes, the definition is parsed into discrete criteria in the Table.5

Even this simple summary makes it obvious that a government agency alone could not possibly have the competence to determine who meets these complex technical and moral criteria. The members of the profession must assume a primary role in that determination. The complicated history of the professions shows that the locus of these decisions has resided in various combinations of educational institutions, such as nursing schools,6 professional societies (eg, National Association of Social Workers),7 and certifying boards (eg, National Commission on Certification of Physician Assistants).8 States, not the federal government, have long played a key part in defining professions in the US, through their authority to grant licenses to practice.9
In response to criticism, the DOE has stated that “the definition of a ‘professional degree’ is an internal definition used by the Department of Education to distinguish among programs that qualify for higher loan limits, not a value judgment about the importance of programs. It has no bearing on whether a program is professional in nature or not.”2 Given the ancient compact between society and the professions in which the government subsidizes the training of professionals dedicated to public service, it is hard to see how these changes can be dismissed as merely semantic and not a promissory breach.10
I recognize that this abstract editorial is little comfort to beleaguered and demoralized professionals and students. Still, it offers a voice of support for each federal practitioner or trainee who fulfills the epigraph’s description of a professional day after day. The nurse who works the extra shift without complaint or resentment so that veterans receive the care they deserve, the social worker who responds on a weekend night to an active duty family without food so they do not spend another night hungry, and the physician assistant who makes it into the isolated public health clinic despite the terrible weather so there is someone ready to take care for patients in need. The proposed policy shift cannot in any meaningful sense rob them of their identity as individuals committed to a code of caring. However, without an intact social compact, it may well remove their practical ability to remain and enter the helping professions to the detriment of us all.
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
- Wade JW. Public responsibilities of the learned professions. Louisiana Law Rev. 1960;21:130-148
- US Department of Education. Myth vs. fact: the definition of professional degrees. Press Release. November 24, 2025. Accessed December 22, 2025. https://www.ed.gov/about/news/press-release/myth-vs-fact-definition-of-professional-degrees
- Laws J. Full list of degrees not classed as “professional” by Trump admin. Newsweek. Updated November 26, 2025. Accessed December 22, 2025. https://www.newsweek.com/full-list-degrees-professional-trump-administration-11085695
- New York Academy of Medicine. Response to stripping “professional status” as proposed by the Department of Education. New York Academy of Medicine. November 24, 2025. Accessed December 22, 2025. https://nyam.org/article/response-to-stripping-professional-status-as-proposed-by-the-department-of-education
- Cruess SR, Johnston S, Cruess RL. “Profession”: a working definition for medical educators. Teach Learn Med. 2004;16:74-76. doi:10.1207/s15328015tlm1601_15
- American Association of Colleges of Nursing. Nursing is a professional degree. American Association of Colleges of Nursing. Accessed December 20, 2025. https://www.aacnnursing.org/policy-advocacy/take-action/nursing-is-a-professional-degree
- National Association of Social Workers. Social work is a profession. Social Workers. Accessed December 20, 2025. https://www.socialworkers.org
- National Commission on Certification of Physician Assistants. Accessed December 20, 2025. https://www.nccpa.net/about-nccpa/#who-we-are
- The Federation of State Boards of Physical Therapy. Accessed December 20, 2025. https://www.fsbpt.org/About-Us/Staff-Home
- Cruess SR, Cruess RL. Professionalism and medicine’s contract with social contract with society. Virtual Mentor. 2004;6:185-188. doi:10.1001/virtualmentor.2004.6.4.msoc1-040
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Codes, Contracts, and Commitments: Who Defines What is a Profession?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
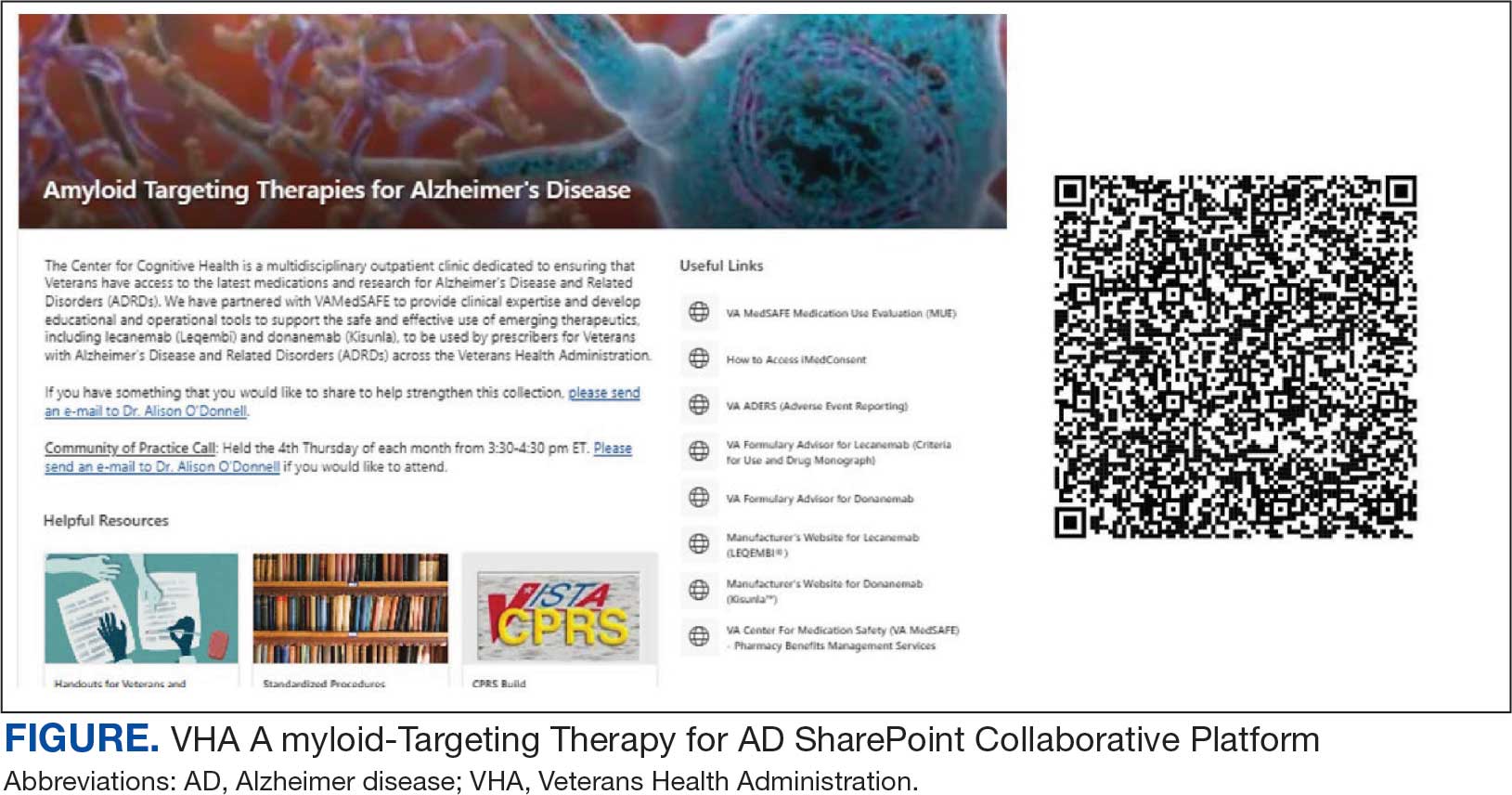
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.
Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
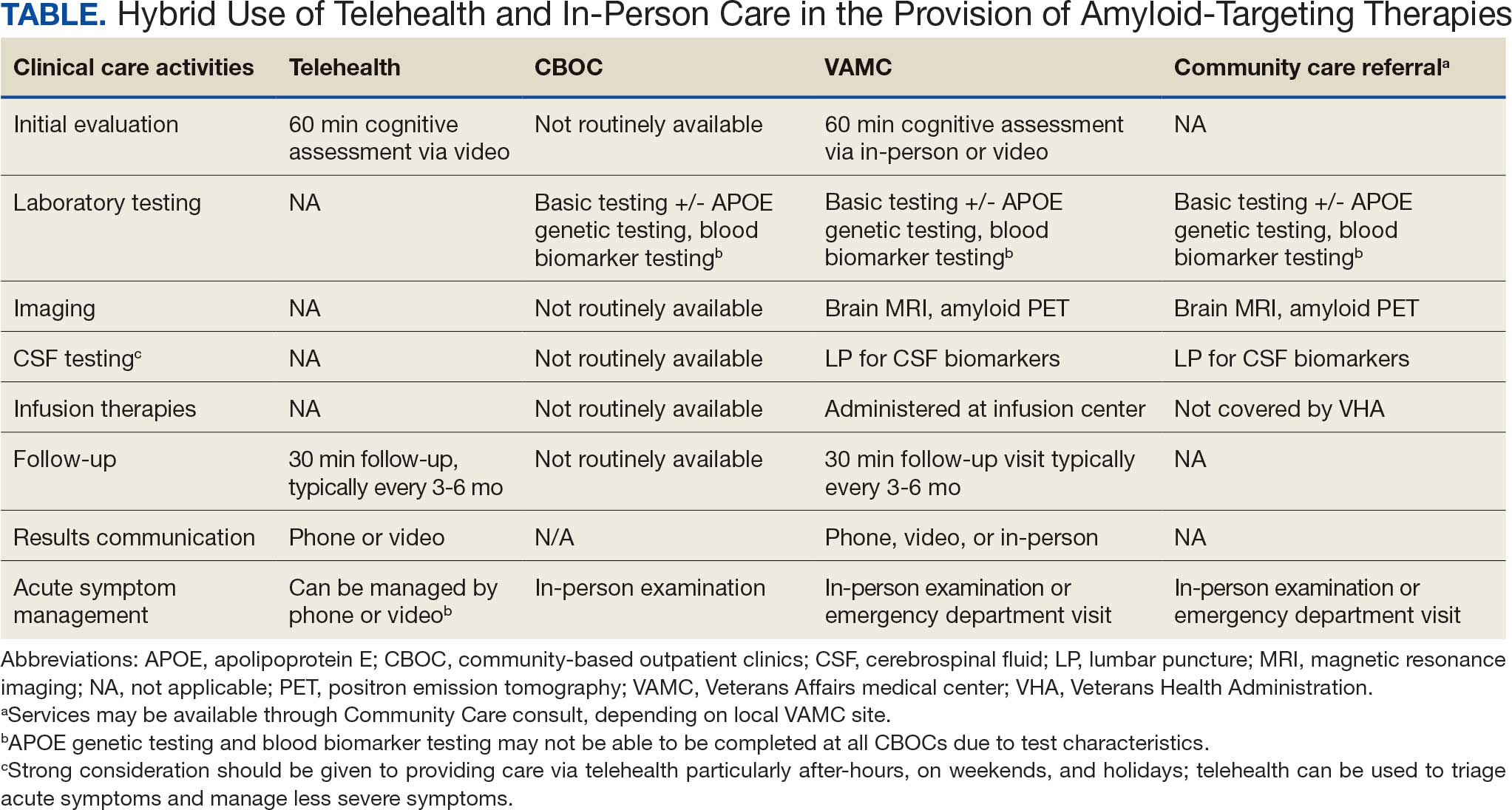
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.
The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
The Veterans Health Administration (VHA) is the largest US integrated health care system, providing health care to > 9 million veterans annually. Dementia affects > 7.2 million Americans, and an estimated 450,000 veterans live with Alzheimer disease (AD).1,2 Compared with the general population, veterans have a higher burden of chronic medical conditions and are disproportionately affected by AD due to exposure to military-related risk factors (eg, traumatic brain injury and posttraumatic stress disorder) and the high prevalence of nonmilitary risk factors, such as cardiovascular disease. The VHA is a pioneer in dementia care, having established a Dementia System of Care to provide primary and specialty care to veterans with dementia. The VHA also is leading the way in implementing the Institute for Healthcare Improvement Age-Friendly Health Systems (AFHS) framework for providing goal-concordant care in > 100 VHA medical centers. The VHA aims to be the largest AFHS in the country.
AD profoundly affects individuals and their families. The progressive nature of the most common form of dementia diminishes the quality of life for patients as well as their care partners in an ongoing fashion, often leading to emotional, physical, and financial strain. Costs for health and long-term care for people living with AD and other dementias were projected at $360 billion in 2024, largely due to the need for nursing home care.1 Although several oral medications are available, their capacity to effectively mitigate the negative effects of AD is limited. Cholinesterase inhibitors and memantine may offer temporary symptomatic relief, but they do not alter disease progression.3 The use of these agents is relatively low, with about one-third of patients diagnosed with AD receiving these medications.4
Amyloid-Targeting Therapies
Recent advancements in biologics, particularly amyloid-targeting therapies, such as lecanemab and donanemab, offer new hope for managing AD. Older adults treated with these medications show less decline on measures of cognition and function than those receiving a placebo at 18 months.5,6 However, accessing and using these medications is challenging.
Use of amyloid-targeting therapies poses challenges. The medications are expensive, potentially placing a financial burden on patients, families, and health care systems.7 Determining initial eligibility for treatment requires a battery of cognitive assessments, laboratory tests, advanced radiologic studies (eg, magnetic resonance imaging [MRI] of the brain and amyloid positron emission tomography [PET] scans), and possible cerebrospinal fluid (CSF) testing. Frequent ongoing assessments are necessary to monitor safety and efficacy. These treatments carry substantial risks, particularly amyloid-related imaging abnormalities (ARIA) such as cerebral edema, microhemorrhages, and superficial siderosis. Therefore, follow-up assessments typically occur around months 2, 3, 4, and 7, depending on which medication is selected. Finally, at present, both agents must be intravenous (IV)-administered in a monitored clinical setting, which requires additional coordination, transportation, and cost.
Ongoing evaluations and in-person administration particularly affect patients and care partners with limitations regarding transportation, time off work, and navigating complex health care systems.8 VHA clinicians at sites that have implemented or are interested in implementing amyloid-targeting therapy programs endorse similar challenges when implementing these therapies in their US Department of Veterans Affairs (VA) medical centers (VAMCs).9
The VHA was one of the first health care systems to use amyloid-targeting therapies, covering the cost of lecanemab and donanemab, in addition to costs associated with concomitant evaluation and testing. However, given the safety concerns with this novel class of medications, the VHA National Formulary Committee developed criteria for use and recommended the VA Center for Medication Safety (VAMedSAFE) conduct a mandatory real-time medication use evaluation (MUE). VAMedSAFE developed the MUE to monitor the safe and appropriate use of amyloid-targeting therapy for AD. Two authors (AJO, SMH) partnered with VAMedSAFE through the VA Pittsburgh Healthcare System Technology Enhancing Cognition and Health–Geriatric Research, Education, and Clinical Center (TECH-GRECC) to provide clinical expertise, substantive feedback for the development of the MUE, and guidance for VHA sites starting amyloid targeting-therapy programs. We started a VHA Amyloid-Targeting Therapy for AD SharePoint collaborative platform and VHA AD Therapeutics Community of Practice (CoP) for shared learning (Figure). The private SharePoint platform houses an array of implementation materials for VAMCs starting programs: key documents and links; educational materials; sample guidelines; note templates; and electronic health record screenshots. The CoP allows VHAs to share best practices and discuss challenges.

Even with these advantages, we found that ensuring the safe and appropriate use of amyloid-targeting therapies did not overcome the barriers associated with their complexity. This was especially true for veterans living in rural areas. Only 4 VAMCs had administered amyloid-targeting therapies in the first year they were available. Preliminary data demonstrated that 27 (84%) of 32 veterans who initiated lecanemab in the VHA between October 2023 and September 2024 resided in urban areas.10 To address the underutilization of amyloid-targeting therapy, we propose leveraging the strengths of VHA telehealth to facilitate expansion of access to these medications for veterans with early AD. Telehealth may substantially increase access to evaluation for veterans with early dementia and, when medically appropriate, to receive amyloid-targeting therapies by reducing transportation needs and mitigating costs while ensuring appropriate monitoring through ongoing clinical assessments.
Using Telehealth
The VHA is a pioneer in telehealth, with programs dating back to 2003.11 Between October 1, 2018, and September 30, 2019, the VHA served > 900,000 veterans through the provision of > 2.6 million episodes of care via telehealth.12 The COVID-19 pandemic further cemented the role of telemedicine as an essential component of health care. Telehealth has demonstrated success in the assessment and management of individuals living with dementia. At the VHA, the GRECC-Connect Project is a partnership between 9 urban GRECC sites that seek to provide consultative geriatric and dementia care to rural veterans through telehealth.13 Additional evidence supports the potential to leverage telehealth to effectively communicate results of amyloid PET scans.14
This approach is not without limitations such as the digital divide, or the gap that separates technology-enabled individuals and those unprepared to adopt technology due to limited digital literacy levels or access to needed hardware, software, and connectivity. The VHA has taken steps to address these digital divide barriers by broadly providing tools—such as tablets and broadband connectivity—to veterans. Specifically, the VHA has instituted digital divide consults to determine whether telehealth could be a potential solution for appropriate veterans and to provide an iPad (if eligible) to connect with VA clinicians. Complementary to the digital divide consult, a VHA-specific telehealth preparedness assessment tool is under development and being tested by 2 authors (JF, SMH). This telehealth preparedness assessment tool is designed to aid in the seamless integration of telehealth services with the support of tailored education materials specific to gaps in digital literacy that a veteran might experience.
Building on these initiatives, there is an opportunity to expand access to amyloid-targeting therapies, regardless of distance to large VAMCs, by leveraging telehealth as an alternative method of connecting patients with specialty care. Specifically, a hybrid approach could be used to accomplish the myriad initial and follow-up tasks involved in the provision of amyloid-targeting therapies (Table). Not all VHA facilities possess the specialty expertise to prescribe these medications, and local clinicians may not have sufficient knowledge and clinical support to prescribe and monitor these therapies.

The first step is identifying local and regional subject matter experts, followed by the development and expansion of these networks. The National TeleNeurology Program is a good example of a national telehealth program that leverages technology to bring specialty services to rural areas with limited access to care. Although amyloid-targeting therapies often require more complex logistics, such as laboratory tests and imaging, these initial hurdles can be overcome through localized services and collaboration between VAMCs.
While treatment and imaging will most likely need to occur at a VAMC, most basic laboratory studies can be performed at community-based outpatient clinics (CBOCs). Some CBOCs may not be able to process more specialized laboratory tests such as apolipoprotein E genetic testing. Samples for these tests can be collected and processed at VAMCs, which usually have contracts with outside laboratories capable of performing these studies. Most, although not all, VAMCs offer advanced imaging, including MRI of the brain and amyloid PETs. VAMCs without those modalities may need to coordinate with other regional VAMCs. Additionally, a pilot program is already underway whereby VAMCs without the ability to quantify the amount of amyloid on PETs are able to leverage technology and collaborations with other VAMCs to obtain these data.
Once the initial phases of evaluation and care are completed, telemedicine can be leveraged for follow-up and ongoing management. Interdisciplinary teams can help facilitate care related to amyloid-targeting therapies, including the close monitoring of veterans for development of ARIA.15 To achieve this monitoring, specialty clinic teams prescribing amyloid-targeting therapies, which may be geographically distant, need to coordinate with local primary care clinical teams and emergency clinicians. All of these health care team members, along with neurologists and neurosurgeons, should be involved in the development and implementation of protocols in the event that patients present to their local primary or specialty care clinics or emergency department with ARIA symptoms.
If amyloid-targeting therapies are to be provided along with other emerging treatments for rural veterans, telehealth must be part of the solution. There is a pressing need to explore innovative evaluation and delivery models for these therapies, particularly as we expect additional diagnostics and therapeutics to be available in the future. With the advent of commercially available blood tests (ie, blood biomarkers) for AD, there is hope for a transition away from PETs and CSF testing given their cost, limited access, and invasiveness for diagnosis and monitoring of AD. These advances will increase the utility of telehealth to help rural veterans access amyloid-targeting therapies.
Additionally, administering the drug at home or at local clinics, supported by a dedicated health care team or home health agency, could further improve accessibility. Telehealth can be leveraged in this scenario, allowing specialty clinics and specialists to connect with patients and clinicians based out of local clinics or even home health agencies. In this scenario, specialists can provide hands-on care guidance and oversight even though they may be geographically distant from care recipients. Transitioning from IV administration to subcutaneous formulations would further enhance convenience and reduce barriers; these formulations may be available soon.16 Addressing logistical challenges to care and access through technology-based solutions will require coordinated efforts and continued VHA investment.
Conclusions
The VHA has a large population of veterans with dementia, and the costs to care for these veterans will only increase. While the current benefits of amyloid-targeting therapies are modest, now is the time to establish care processes that will support future innovations in amyloid-targeting therapies and other treatments and diagnostics. We are developing better ways to detect AD using clinical decision support tools, improving care pathways and the management of AD, and leveraging telehealth to improve access. The VA is conducting research to investigate whether a cognitive screening and laboratory evaluation that includes a telehealth preparedness assessment will be feasible and effective for improving the detection of AD and access to treatment, and we plan to publish the results.
The lessons learned can be extended to non-VHA care settings to help achieve potential benefits for other patients with early AD. Emerging therapies have the potential to improve the quality of life for both patients and care partners, adding life to years and not just years to life. Policymakers and payors must prioritize research funding to evaluate the safety and efficacy of these approaches to the delivery of health services, ensuring that emerging therapies are accessible for all individuals affected by AD.
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
- Alzheimer’s Association. 2025 Alzheimer’s disease facts and figures. Alzheimers Dement. 2025;21(4):e70235. doi:10.1002/alz.70235
- US Department of Veterans Affairs. Statistical Projections of Alzheimer’s Dementia for VA Patients, VA Enrollees, and US Veterans. December 18, 2020. Accessed November 2, 2025. https://www.va.gov/GERIATRICS/docs/VHA_ALZHEIMERS_DEMENTIA_Statistical_Projections_FY21_and_FY33_sgc121820.pdf
- Casey DA, Antimisiaris D, O’Brien J. Drugs for Alzheimer’s disease: are they effective? P T. 2010;35(4):208-211.
- Barthold D, Joyce G, Ferido P, et al. Pharmaceutical treatment for Alzheimer’s disease and related dementias: utilization and disparities. J Alzheimers Dis. 2020;76(2):579-589. doi:10.3233/JAD-200133
- Sims JR, Zimmer JA, Evans CD, et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA. 2023;330(6):512-527. doi:10.1001/jama.2023.13239
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
- Tanne JH. Lecanemab: US Veterans Health Administration will cover cost of new Alzheimer’s drug. BMJ. 2023;380:p628. doi:10.1136/bmj.p628
- Nadeau SE. Lecanemab questions. Neurology. 2024;102(7):e209320. doi:10.1212/WNL.0000000000209320 9. O’Donnell AJ, Fortunato AT, Spitznogle BL, et al. Implementation of lecanemab for Alzheimer’s disease: facilitators and barriers. Presented at: American Geriatrics Society 2025 Annual Scientific Meeting, Chicago. May 2025.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- O’Donnell AJ, Zhao X, Parr A, et al. Use of lecanemab for Alzheimer’s disease within the Veteran’s Health Foundation: early findings. Abstract presented at: Alzheimer’s Association International Conference 2025; July 27, 2025; Toronto, Canada.
- Hopp F, Whitten P, Subramanian U, et al. Perspectives from the Veterans Health Administration about opportunities and barriers in telemedicine. J Telemed Telecare. 2006;12(8):404-409. doi:10.1258/135763306779378717
- VA reports significant increase in veteran use of telehealth services. News release. US Department of Veterans Affairs. November 22, 2019. Accessed November 19, 2025. https://news.va.gov/press-room/va-reports-significant-increase-in-veteran-use-of-telehealth-services/
- Powers BB, Homer MC, Morone N, et al. Creation of an interprofessional teledementia clinic for rural veterans: preliminary data. J Am Geriatr Soc. 2017;65(5):1092-1099. doi:10.1111/jgs.14839
- Erickson CM, Chin NA, Rosario HL, et al. Feasibility of virtual Alzheimer’s biomarker disclosure: findings from an observational cohort. Alzheimers Dement (N Y). 2023;9(3):e12413. doi:10.1002/trc2.12413
- Turk KW, Knobel MD, Nothern A, et al. An interprofessional team for disease-modifying therapy in Alzheimer disease implementation. Neurol Clin Pract. 2024;14(6):e200346. doi:10.1212/CPJ.0000000000200346
- FDA accepts LEQEMBI® (lecanemab-irmb) biologics license application for subcutaneous maintenance dosing for the treatment of early Alzheimer’s disease. News release. Elsai US. January 13, 2025. Accessed November 2, 2025. https://media-us.eisai.com/2025-01-13-FDA-Accepts-LEQEMBI-R-lecanemab-irmb-Biologics-License-Application-for-Subcutaneous-Maintenance-Dosing-for-the-Treatment-of-Early-Alzheimers-Disease
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
Can Telehealth Improve Access to Amyloid-Targeting Therapies for Veterans Living With Alzheimer Disease?
The Once and Future Veterans Health Administration
The Once and Future Veterans Health Administration
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
He who thus considers things in their first growth and origin ... will obtain the clearest view of them. Politics, Book I, Part II by Aristotle
Many seasoned observers of federal practice have signaled that the future of US Department of Veterans Affairs (VA) health care is threatened as never before. Political forces and economic interests are siphoning Veterans Health Administration (VHA) capital and human resources into the community with an ineluctable push toward privatization.1
This Veterans Day, the vitality, if not the very viability of veteran health care, is in serious jeopardy, so it seems fitting to review the rationale for having institutions dedicated to the specialized medical treatment of veterans. Aristotle advises us on how to undertake this intellectual exercise in the epigraph. This column will revisit the historical origins of VA medicine to better appreciate the justification of an agency committed to this unique purpose and what may be sacrificed if it is decimated.
The provision of medical care focused on the injuries and illnesses of warriors is as old as war. The ancient Romans had among the first veterans’ hospital, named a valetudinarium. Sick and injured members of the Roman legions received state-of-the-art medical and surgical care from military doctors inside these facilities.2
In the United States, federal practice emerged almost simultaneously with the birth of a nation. Wounded troops and families of slain soldiers required rehabilitation and support from the fledgling federal government. This began a pattern of development in which each war generated novel injuries and disorders that required the VA to evolve (Table).3

Many arguments can be marshalled to demonstrate the importance of not just ensuring VA health care survives but also has the resources needed to thrive. I will highlight what I argue are the most important justifications for its existence.
The ethical argument: President Abraham Lincoln and a long line of government officials for more than 2 centuries have called the provision of high-quality health care focused on veterans a sacred trust. Failing to fulfill that promise is a violation of the deepest principles of veracity and fidelity that those who govern owe to the citizens who selflessly sacrificed time, health, and even in some cases life, for the safety and well-being of their country.4
The quality argument: Dozens of studies have found that compared to the community, many areas of veteran medical care are just plain better. Two surveys particularly salient in the aging veteran population illustrate this growing body of positive research. The most recent and largest survey of Medicare patients found that VHA hospitals surpassed community-based hospitals on all 10 metrics.5 A retrospective cohort study of mortality compared veterans transported by ambulance to VHA or community-based hospitals. The researchers found that those taken to VHA facilities had a 30-day all cause adjustment mortality 20 times lower than those taken to civilian hospitals, especially among minoritized populations who generally have higher mortality.6
The cultural argument: Glance at almost any form of communication from veterans or about their health care and you will apprehend common cultural themes. Even when frustrated that the system has not lived up to their expectations, and perhaps because of their sense of belonging, they voice ownership of VHA as their medical home. Surveys of veteran experiences have shown many feel more comfortable receiving care in the company of comrades in arms and from health care professionals with expertise and experience with veterans’ distinctive medical problems and the military values that inform their preferences for care.7
The complexity argument: Anyone who has worked even a short time in a VHA hospital or clinic knows the patients are in general more complicated than similar patients in the community. Multiple medical, geriatric, neuropsychiatric, substance use, and social comorbidities are the expectation, not the exception, as in some civilian systems. Many of the conditions common in the VHA such as traumatic brain injury, service-connected cancers, suicidal ideation, environmental exposures, and posttraumatic stress disorder would be encountered in community health care settings. The differences between VHA and community care led the RAND Corporation to caution that “Community care providers might not be equipped to handle the needs of veterans.”8
Let me bring this 1000-foot view of the crisis facing federal practice down to the literal level of my own home. For many years I have had a wonderful mechanic who has a mobile bike service. I was talking to him as he fixed my trike. I never knew he was a Vietnam era veteran, and he didn’t realize that I was a career VA health care professional at the very VHA hospital where he received care. He spontaneously told me that, “when I first got out, the VA was awful, but now it is wonderful and they are so good to me. I would not go anywhere else.” For the many veterans of that era who would echo his sentiments, we must not allow the VA to lose all it has gained since that painful time
Another philosopher, Søren Kierkegaard, wrote that “life must be understood backwards but lived forwards.”9 Our own brief back to the future journey in this editorial has, I hope, shown that VHA medical institutions and health professionals cannot be replaced with or replicated by civilian systems and clinicians. Continued attempts to do so betray the trust and risks the health and well-being of veterans. It also would deprive the country of research, innovation, and education that make unparalleled contributions to public health. Ultimately, these efforts to diminish VHA compromise the solidarity of service members with each other and with their federal practitioners. If this trend to dismantle an organization that originated with the sole purpose of caring for veterans continues, then the public expressions of respect and gratitude will sound shallower and more tentative with each passing Veterans Day.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
- Quil L. Hundreds of VA clinicians warn that cuts threaten vet’s health care. National Public Radio. October 1, 2025. Accessed October 27, 2025. https://www.npr.org/2025/10/01/nx-s1-5554394/hundreds-of-va-clinicians-warn-that-cuts-threaten-vets-health-care
- Nutton V. Ancient Medicine. 2nd ed. Routledge; 2012.
- US Department of Veterans Affairs. VA History Summary. Updated June 13, 2025. Accessed October 27, 2025. https://department.va.gov/history/history-overview/
- Geppert CMA. Learning from history: the ethical foundation of VA health care. Fed Pract. 2016;33:6-7.
- US Department of Veterans Affairs. Nationwide patient survey shows VA hospitals outperform non-VA hospitals. News release. June 14, 2023. Accessed October 27, 2025. https://news.va.gov/press-room/nationwide-patient-survey-shows-va-hospitals-outperform-non-va-hospitals
- Chan DC, Danesh K, Costantini S, Card D, Taylor L, Studdert DM. Mortality among US veterans after emergency visits to Veterans Affairs and other hospitals: retrospective cohort study. BMJ. 2022;376:e068099. doi:10.1136/bmj-2021-068099
- Vigilante K, Batten SV, Shang Q, et al. Camaraderie among US veterans and their preferences for health care systems and practitioners. JAMA Netw Open. 2025;8(4):e255253. doi:10.1001/jamanetworkopen.2025.5253
- Rasmussen P, Farmer CM. The promise and challenges of VA community care: veterans’ issues in focus. Rand Health Q. 2023;10:9.
- Kierkegaard S. Journalen JJ:167 (1843) in: Søren Kierkegaards Skrifter. Vol 18. Copenhagen; 1997:306.
The Once and Future Veterans Health Administration
The Once and Future Veterans Health Administration
Special Report II: Tackling Burnout
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Last month, we introduced the epidemic of burnout and the adverse consequences for both our vascular surgery patients and ourselves. Today we will outline a framework for addressing these issues. The foundation of this framework is informed by the social and neurosciences.
From the perspective of the social scientist: Christina Maslach, the originator of the widely used Maslach Burnout Inventory, theorized that burnout arises from a chronic mismatch between people and their work setting in some or all of the following domains: Workload (too much, wrong kind); control (lack of autonomy, or insufficient control over resources); reward (insufficient financial or social rewards commensurate with achievements); community (loss of positive connection with others); fairness (lack of perceived fairness, inequity of work, pay, or promotion); and values (conflict of personal and organizational values). The reality of practicing medicine in today’s business milieu – of achieving service efficiencies by meeting performance targets – brings many of these mismatches into sharp focus.
From the perspective of the neuroscientist: Recent advances, including functional MRI, have demonstrated that the human brain is hard wired for compassion. Compassion is the deep feeling that arises when confronted with another’s suffering, coupled with a strong desire to alleviate that suffering. There are at least two neural pathways: one activated during empathy, having us experience another’s pain; and the other activated during compassion, resulting in our sense of reward. Thus, burnout is thought to occur when you know what your patient needs but you are unable to deliver it. Compassionate medical care is purposeful work, which promotes a sense of reward and mitigates burnout.
Because burnout affects all caregivers (anyone who touches the patient), a successful program addressing workforce well-being must be comprehensive and organization wide, similar to successful patient safety, CPI [continuous process improvement] and LEAN [Six Sigma] initiatives.
There are no shortcuts. Creating a culture of compassionate, collaborative care requires an understanding of the interrelationships between the individual provider, the unit or team, and organizational leadership.
1) The individual provider: There is evidence to support the use of programs that build personal resilience. A recently published meta-analysis by West and colleagues concluded that while no specific physician burnout intervention has been shown to be better than other types of interventions, mindfulness, stress management, and small-group discussions can be effective approaches to reducing burnout scores. Strategies to build individual resilience, such as mindfulness and meditation, are easy to teach but place the burden for success on the individual. No amount of resilience can withstand an unsupportive or toxic workplace environment, so both individual and organizational strategies in combination are necessary.
2) The unit or team: Scheduling time for open and honest discussion of social and emotional issues that arise in caring for patients helps nourish caregiver to caregiver compassion. The Schwartz Center for Compassionate Healthcare is a national nonprofit leading the movement to bring compassion to every patient-caregiver interaction. More than 425 health care organization are Schwartz Center members and conduct Schwartz Rounds™ to bring doctors, nurses, and other caregivers together to discuss the human side of health care. (www.theschwartzcenter.org). Team member to team member support is essential for navigating the stressors of practice. With having lunch in front of your computer being the norm, and the disappearance of traditional spaces for colleagues to connect (for example, nurses’ lounge, physician dining rooms), the opportunity for caregivers to have a safe place to escape, a place to have their own humanity reaffirmed, a place to offer support to their peers, has been eliminated.
3) Organizational Leadership: Making compassion a core value, articulating it, and establishing metrics whereby it can be measured, is a good start. The barriers to a culture of compassion are related to our systems of care. There are burgeoning administrative and documentation tasks to be performed, and productivity expectations that turn our clinics and hospitals into assembly lines. No, we cannot expect the EMR [electronic medical records] to be eliminated, but workforce well-being cannot be sustainable in the context of inadequate resources. A culture of compassionate collaborative care requires programs and policies that are implemented by the organization itself. Examples of organization-wide initiatives that support workforce well-being and provider engagement include: screening for caregiver burnout, establishing policies for managing adverse events with an eye toward the second victim, and most importantly, supporting systems that preserve work control autonomy of physicians and nurses in clinical settings. The business sector has long recognized that workplace stress is a function of how demanding a person’s job is and how much control that person has over his or her responsibilities. The business community has also recognized that the experience of the worker (provider) drives the experience of the customer (patient). In a study of hospital compassionate practices and HCAHPS [the Hospital Consumer Assessment of Healthcare Providers and Systems], McClelland and Vogus reported that how well a hospital compassionately supports it employees and rewards compassionate acts is significantly and positively is associated with that hospital’s ratings and likelihood of patients recommending it.
How does the Society of Vascular Surgery, or any professional medical/nursing society for that matter, fit into this model?
We propose that the SVS find ways to empower their members to be agents for culture change within their own health care organizations. How might this be done:
- Teach organizational leadership skills, starting with the SVS Board of Directors, the presidential line, and the chairs of committees. Offer leadership courses at the Annual Meeting.
- Develop a community of caregivers committed to creating a compassionate collaborative culture. The SVS is a founding member of the Schwartz Center Healthcare Society Leadership Council, and you, as members of the SVS benefit from reduced registration at the Annual Compassion in Action Healthcare Conference, June 24-27, 2017 in Boston. (http://compassioninactionconference.org) This conference is designed to be highly experiential, using a hands-on “how to do it” model.
- The SVS should make improving the overall wellness of its members a specific goal and find specific metrics to monitor our progress towards this goal. Members can be provided with the tools to identify, monitor, and measure burnout and compassion. Each committee and council of the SVS can reexamine their objectives through the lens of reducing burnout and improving the wellness of vascular surgeons.
- Provide members with evidence-based programs that build personal resilience. This will not be a successful initiative unless our surgeons recognize and acknowledge the symptoms of burnout, and are willing to admit vulnerability. Without doing so, it is difficult to reach out for help.
- Redesign postgraduate resident and fellowship education. Standardizing clinical care may reduce variation and promote efficiency. However, when processes such as time-limited appointment scheduling, EMR templates, and protocols that drive physician-patient interactions are embedded in Resident and Fellowship education, the result may well be inflexibility in practice, reduced face time with patients, and interactions that lack compassion; all leading to burnout. Graduate Medical Education leaders must develop programs that support the learner’s ability to connect with patients and families, cultivate and role-model skills and behaviors that strengthen compassionate interactions, and strive to develop clinical practice models that increase Resident and Fellow work control autonomy.
The SVS should work proactively to optimize workload, fairness, and reward on a larger scale for its members as it relates to the EMR, reimbursement, and systems coverage. While we may be relatively small in size, as leaders, we are perfectly poised to address these larger, global issues. Perhaps working within the current system (i.e., PAC and APM task force) and considering innovative solutions at a national leadership scale, the SVS can direct real change!
Changing culture is not easy, nor quick, nor does it have an easy-to-follow blueprint. The first step is recognizing the need. The second is taking a leadership role. The third is thinking deeply about implementation.
*The authors extend their thanks and appreciation for the guidance, resources and support of Michael Goldberg, MD, scholar in residence, Schwartz Center for Compassionate Care, Boston and clinical professor of orthopedics at Seattle Children’s Hospital.
REFERENCES
1. J Managerial Psychol. (2007) 22:309-28
2. Annu Rev Neurosci. (2012) 35:1-23
3. Medicine. (2016) 44:583-5
4. J Health Organization Manag. (2015) 29:973-87
5. De Zulueta P Developing compassionate leadership in health care: an integrative review. J Healthcare Leadership. (2016) 8:1-10
6. Dolan ED, Morh D, Lempa M et al. Using a single item to measure burnout in primary care staff: A psychometry evaluation. J Gen Intern Med. (2015) 30:582-7
7. Karasek RA Job demands, job decision latitude, and mental strain: implications for job design. Administrative Sciences Quarterly (1979) 24: 285-308
8. Lee VS, Miller T, Daniels C, et al. Creating the exceptional patient experience in one academic health system. Acad Med. (2016) 91:338-44
9. Linzer M, Levine R, Meltzer D, et al. 10 bold steps to prevent burnout in general internal medicine. J Gen Intern Med. (2013) 29:18-20
10. Lown BA, Manning CF The Schwartz Center Rounds: Evaluation of an interdisciplinary approach to enhancing patient-centered communication, teamwork, and provider support. Acad Med. (2010) 85:1073-81
11. Lown BA, Muncer SJ, Chadwick R Can compassionate healthcare be measured? The Schwartz Center Compassionate Care Scale. Patient Education and Counseling (2015) 98:1005-10
12. Lown BA, McIntosh S, Gaines ME, et. al. Integrating compassionate collaborative care (“the Triple C”) into health professional education to advance the triple aim of health care. Acad Med (2016) 91:1-7
13. Lown BA A social neuroscience-informed model for teaching and practicing compassion in health care. Medical Education (2016) 50: 332-342
14. Maslach C, Schaufeli WG, Leiter MP Job burnout. Annu Rev Psychol (2001) 52:397-422
15. McClelland LE, Vogus TJ Compassion practices and HCAHPS: Does rewarding and supporting workplace compassion influence patient perceptions? HSR: Health Serv Res. (2014) 49:1670-83
16. Shanafelt TD, Noseworthy JH Executive leadership and physician well-being: Nine organizational strategies to promote engagement and reduce burnout. Mayo Clin Proc. (2016) 6:1-18
17. Shanafelt TD, Dyrbye LN, West CP Addressing physician burnout: the way forward. JAMA (2017) 317:901-2
18. Singer T, Klimecki OM Empathy and compassion Curr Biol. (2014) 24: R875-8
19. West CP, Dyrbye LN, Satele DV et. al. Concurrent validity of single-item measures of emotional exhaustion and depersonalization in burnout assessment. J Gen Intern Med. (2012) 27:1445-52
20. West CP, Dyrbye LN, Erwin PJ, et al. Interventions to address and reduce physician burnout: a systematic review and meta-analysis. Lancet. (2016) 388:2272-81
21. Wuest TK, Goldberg MJ, Kelly JD Clinical faceoff: Physician burnout-Fact, fantasy, or the fourth component of the triple aim? Clin Orthop Relat Res. (2016) doi: 10.1007/5-11999-016-5193-5
Transplantation palliative care: The time is ripe
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
Exogenously, hypochlorous acid (HOCl) is a powerful oxidizing agent formed from chlorine dissolved in water. Within the body, it is part of the immune response, created by activated leukocytes, which form HOCl from hydrogen peroxide and chloride. HOCl has been used as a disinfectant in wound care due to its antimicrobial properties via inhibition of DNA synthesis, protein synthesis, and decreased adenosine triphosphate production. It specifically targets bacteria by blocking bacterial cell wall synthesis and decreasing DNA replication.1
During the COVID-19 pandemic, HOCl was recommended by the US Environmental Protection Agency as a disinfectant.2 HOCl can be purchased from a supplier, though its major limitation is its shelf life. The main environmental factors affecting its stability are sunlight exposure, temperatures > 25 °C, and air exposure. HOCl is stable and most potent when the pH falls between 3.5 and 5.5.3 It is best stored in a cool, dark environment to maintain efficacy for 2 weeks. Rossi-Fedele et al found that when exposed to sunlight, chlorine reduction starts on day 4, whereas solutions kept in dark storage remained more stable, with this process starting after day 14.4
HOCl can also be made on-site via a machine, which ranges in price from a portable version costing < $200 to a large commercial option that can cost $7000 to $25,000. HOCl is produced by mixing noniodinated salt and water, and using electrolysis, which generally takes less than 10 minutes before it is ready for use.2 Given the cost and nonreusable nature of disinfecting wipes, HOCl may be more worthwhile for economic and disposal purposes in the long term.
Different concentrations of HOCl are readily available commercially. Because topical application of 1% HOCl may cause skin irritation, solutions with lower concentrations have been developed including Vashe (0.03% HOCl; SteadMed), PhaseOne (0.025% HOCl solution; IHT), OCuSOFT (0.02% HOCl; OCuSOFT), Bruder (0.02% HOCl; Bruder Healthcare), Acuicyn (0.01% HOCl solution in dilute saline; Sonoma Pharmaceuticals), and Avenova (0.01% HOCl solution; NovaBay Pharmaceuticals).5
Aside from its surface utility, HOCl has been researched for its beneficial effects on skin. HOCl has been shown to be helpful intraoperatively and postoperatively in improving adverse effects (AEs) after hair restoration, including erythema and pruritus, and in optimizing healing by reducing inflammation, likely due to its antimicrobial properties and ability to promote oxygenation.6 Bucko et al demonstrated that Microcyn scar gel (with HOCl) was a superior nonirritating, nontoxic method of not only improving scar appearance (vascularity, scar height, and pliability) but also reducing scar symptoms of pain and pruritus in comparison to 100% silicone scar gel (traditional application used to improve scarring).7 Zhang et al demonstrated that HOCl consistently improved symptom relief of blepharitis, including meibomian gland, eyelash, and eyelid redness, irritation, and appearance in comparison as well as were better tolerated in comparison to traditional recommendations of eyelid compresses and wash (tea tree oil, diluted baby shampoo, and topical antibiotics).8 In children with moderate to severe atopic dermatitis, Majewski et al compared a traditional bleach bath with a body wash containing hypochlorite (NaOCl; hypochlorous acid in alkaline aqueous solution). The body wash proved to be more convenient (showering vs 10-minute bath) and significantly improved symptoms while reducing the need for topical corticosteroids (common treatment modality for atopic dermatitis).9
The skin is the body’s primary defense against both dermatologic and respiratory infections. The face is especially vulnerable to microbes via airborne or environmental transmission, mechanical irritation, and touch. In the military environment, personal protective equipment (PPE) or uniform items may increase the risk of dermatologic conditions such as allergic or irritant dermatitis, infection, and friction blisters.
In a literature review of 312 dermatologic articles published between 2002 and 2022, Singal and Lipner found that among deployed soldiers serving in hot and dry climates, dermatitis and eczematous conditions were the most common, whereas bacterial and fungal conditions were most common in hot and humid settings. In the nondeployed setting, dermatitis and eczematous, acne, and fungal infections were the most common skin conditions. This is reflected by the unique circumstances that service members face at home and while deployed, when they may be more vulnerable to developing new or worsening chronic skin conditions depending on the environment (access to shelter, humid vs dry environments), and decreased access or time for hygiene (shared quarters at home in barracks or on deployment). Occupation-related conditions also play a large role in military dermatologic conditions.10
Dever et al noted the unique risks and exposures in the environment itself (plants, arthropods) as well as uniform items (protective gear) that carry an increased risk of friction irritation and dermatitis. Occupational exposures commonly associated with irritant contact dermatitis include alcohols, oils, fuel, disinfectants, and solvents. Chemicals in military uniforms themselves (eg, formaldehyde resins, disperse dyes, and chromate-containing dyes) also have the potential to cause allergic contact dermatitis, which can be challenging to address given the emphasis on uniformity and standards.11 PPE also may exacerbate rosacea and acne.
Some pathologies are associated specifically with bacteria, such as Cutibacterium acnes, as seen in acne vulgaris. Colonization of bacteria on the face may create biofilms that are difficult to detect, may be resistant to antibiotic therapy, and are implicated in other dermatologic conditions, such as persistent wounds, atopic dermatitis, and candidiasis.12
Biofilm and antibiotic resistance already pose a risk to patient care, but the unique environmental conditions and exposures of military settings can amplify this risk in the military population.13 Using HOCl in austere environments or the field for wound care may help reduce microbial load and the subsequent need for systemic antibiotics which carry the risk of gastrointestinal AEs and resistance.1
An optimized healing rate would support operational objectives by enabling service members to remain on full duty and avoid medications, which may prevent them from special duty, such as aviation. Sakarya et al found that HOCl solution enhanced wound healing in contrast with povidone-iodine (PI), while a study by Dharap et al discussed how HOCl provided major improvement in ulcer wound size (and infection), as well as significant reduction of inflammation.13
Anagnostopoulos et al studied the efficacy of 0.01% HOCl vs other disinfectants (5% PI, 4% chlorhexidine gluconate [CHG] and 70% isopropyl alcohol [IPA]) against common skin organisms, including methicillin-susceptible Staphylococcus aureus and methicillin-susceptible Staphylococcus epidermidis. The study found that HOCl had at least equal if not greater efficacy to PI, CHG, and IPA depending on the bacterial strain, demonstrating immediate bactericidal effects.14
Furthermore, HOCl has been shown to be useful in suturing and wound closure by reducing microbial load when soaked gauze is placed in wound beds prior to closure, while not harming surrounding tissue.15 This would be especially advantageous for military health care when specialist follow-up would be delayed or to prevent infection risk while en route to higher care. Aside from its disinfectant strength, it’s also well tolerated. HOCl studies on human tissue demonstrate its efficacy to prevent irritation and AEs while also preventing infection and promoting wound healing.
Gozukucuk and Cakiroglu studied the use of HOCl as a skin disinfectant before neonatal circumcision and demonstrated fewer adverse effects compared with the more commonly used PI. Neonates treated with PI prior to circumcision resulted in greater postoperative edema and increased duration of wound healing compared with infants treated with HOCl.16 Furthermore, studies have shown that PI can lead to irritant dermatitis or chemical burns if not properly dried or if it becomes pooled because of occlusion dressings.17
Aside from its indicated use for infection or wound care, anti-inflammatory properties of HOCl also may be beneficial for off-label use in preventing flareups of chronic conditions as well as for treating symptoms while awaiting specialist evaluation. This might be the case during US-based training exercises, in remote locations without nearby dermatologists, or during virtual care because of internet constraints. For chronic conditions such as rosacea or atopic dermatitis, which research has shown are related to mast cell activation and degranulation and cytokine release, HOCl has been shown to reduce histamine, neutrophil-generated leukotrienes, in addition to interleukin-6 and interleukin-2 to improve symptoms by reducing inflammation.18
Limitations of HOCl to explore would be extending its shelf life, exploring its various forms (eg, spray, topical) and storage limitations, and training of the machine and materials needed to be made in-house if not purchased. There are also no official guidelines for clinicians to recommend HOCl to patients, and research should be expanded on its use in humans, though it generally is well tolerated without AEs. HOCl has the potential to be a potent, nontoxic, inexpensive tool in med bags or at austere clinics to help maintain a sterile space for procedures, prevent infection while rendering care, and help with exacerbations or prevent flare-ups of chronic conditions such as psoriasis, acne, and atopic dermatitis while specialist care is pending.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
- Natarelli N, et al. Hypochlorous acid: applications in dermatology. J Integr Dermatol. December 22, 2022. Accessed March 2, 2026. https://www.jintegrativederm.org/article/56663-hypochlorous-acid-applications-in-dermatology
- Block MS, Rowan BG. Hypochlorous acid: a review. J Oral Maxillofac Surg. 2020;78:1461-1466. doi:10.1016/j.joms.2020.06.029
- Menta N, Vidal SI, Friedman A. Hypochlorous acid: a blast from the past. J Drugs Dermatol. 2024;23:909-910.
- Rossi-Fedele G, Dogramaci E, Steier L, et al. Some factors influencing the stability of Sterilox®, a super-oxidised water. Br Dent J. 2011;210:E23. doi:10.1038/sj.bdj.2011.143
- Tran AQ, Topilow N, Rong A, et al. Comparison of skin antiseptic agents and the role of 0.01% hypochlorous acid. Aesthet Surg J. 2021;41:1170-1175. doi:10.1093/asj/sjaa322
- Stough D. Topical stabilized super-oxidized hypochlorous acid for wound healing in hair restoration surgery: a real-time usage-controlled trial evaluating safety, efficacy, and tolerability. J Drugs Dermatol. 2023;22:1191-1196. doi:10.36849/JDD.7172
- Bucko AD, Draelos Z, Dubois JC, Jones TM. A doubleblind, randomized study to compare Microcyn scar management hydrogel, K103163, and Kelo-cote scar gel for hypertrophic or keloid scars. Dermatologist. 2015;23:113-122.
- Zhang H, Wu Y, Wan X, et al. Effect of hypochlorous acid on blepharitis through ultrasonic atomization: a randomized clinical trial. J Clin Med. 2023;12(3):1164. doi:10.3390/jcm12031164
- Majewski S, Bhattacharya T, Asztalos M, et al. Sodium hypochlorite body wash in the management of Staphylococcus aureus-colonized moderate-to-severe atopic dermatitis in infants, children, and adolescents. Pediatr Dermatol. 2019;36:442-447. doi:10.1111/pde.13842
- Singal A, Lipner SR. A review of skin disease in military soldiers: challenges and potential solutions. Ann Med. 2023;55:2267425. doi:10.1080/07853890.2023.2267425
- Dever TT, Walters M, Jacob S. Contact dermatitis in military personnel. Dermatitis. 2011;22:313-319. doi:10.2310/6620.2011.11024
- Nowbuth AA, Armstrong J, Cloete T, et al. A potential benefit of hypochlorous acid-facial sanitisation: a review. Preprints. 2021. doi:10.20944/preprints202107.0129.v2
- Gold MH, Andriessen A, Bhatia AC, et al. Topical stabilized hypochlorous acid: the future gold standard for wound care and scar management in dermatologic and plastic surgery procedures. J Cosmet Dermatol. 2020;19:270-277. doi:10.1111/jocd.13280
- Anagnostopoulos AG, Rong A, Miller D, et al. 0.01% hypochlorous acid as an alternative skin antiseptic: an in vitro comparison. Dermatol Surg. 2018;44:1489-1493. doi:10.1097/DSS.0000000000001594
- Odom EB, Mundschenk MB, Hard KA, et al. The utility of hypochlorous acid wound therapy in wound bed preparation and skin graft salvage. Plast Reconstr Surg. 2019;143:677e-678e. doi:10.1097/PRS.0000000000005359
- Gozukucuk A, Cakiroglu B. Comparison of hypochlorous acid and povidone-iodine as a disinfectant in neonatal circumcision. J Pediatr Urol. 2022;18:341.e1-341.e5. doi:10.1016/j.jpurol.2022.03.011
- Borrego L, Hernández N, Hernández Z, et al. Povidoneiodine-induced postsurgical irritant contact dermatitis localized outside of the surgical incision area: report of 27 cases and a literature review. Int J Dermatol. 2016;55:540- 545. doi:10.1111/ijd.12957
- Del Rosso JQ, Bhatia N. Status report on topical hypochlorous acid: clinical relevance of specific formulations, potential modes of action, and study outcomes. J Clin Aesthet Dermatol. 2018;11:36-39.
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
Hypochlorous Acid: A Multipurpose New Addition to the Military Med Bag?
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
The Cutis editorial staff is proud to announce that Vincent A. DeLeo, MD, Editor-in-Chief, was honored with the Master Dermatologist Award at the 2026 Annual Meeting of the American Academy of Dermatology (AAD) in Denver, Colorado.
Presented as part of the AAD’s “Stars of the Academy” program, this award is reserved for physicians whose careers have advanced dermatology through leadership, service, and meaningful contributions to patient care, education, and research. The award reflects Dr. DeLeo’s impact across the specialty.

“Vince’s passion for dermatology has impacted all aspects of our specialty. He has been at the forefront of basic science research, clinical dermatology, education, mentoring, and leadership of specialty organizations and societies.” –Susan C. Taylor, MD
During the presentation, outgoing AAD president Susan C. Taylor, MD, emphasized Dr. DeLeo’s wide-ranging influence, noting his reputation as a researcher, compassionate physician, and skilled diagnostician. He is adept at managing complex cases and improving patient outcomes. Dr. DeLeo is widely recognized for his expertise in contact dermatitis, photomedicine, and photoprotection, as well as for his contributions to dermatologic education.
Beyond his clinical and editorial leadership of Cutis for the past 25 years, Dr. DeLeo is committed to mentorship and leadership by serving on the AAD Board of Directors as well as other specialty organizations such as the American Contact Dermatitis Society.
We congratulate Dr. DeLeo on this well-deserved distinction and thank him for his continued vision and dedication to our readers and the specialty at large.
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
A Legacy in Dermatology: Dr. Vincent A. DeLeo Named AAD Master Dermatologist
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Practice Points
- Military service members face increased UV exposure from occupational and deployment conditions, but sunscreen use is limited by logistical, educational, and behavioral barriers.
- Sunscreen availability in military exchanges partially meets dermatologic guidelines, highlighting opportunities to expand tailored options and integrate sun protection into operational readiness policy.
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO