User login
AVAHO Regional Meeting Addresses Complex World of Peripheral T-Cell Lymphoma
AVAHO Regional Meeting Addresses Complex World of Peripheral T-Cell Lymphoma
SAN FRANCISCO – Peripheral T-cell lymphoma (PTCL) accounts for 4% of mature non-Hodgkin lymphoma cases in the US, or only about 4000 cases a year. While the number of patients is small, however, treatment for PTCL is complex due to wide variations in subtypes and survival rates, a hematologist-oncologist said at the March 21 Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
Weiyun Ai, MD, PhD, a clinical professor of medicine at University of California, San Francisco who specializes in lymphoma, explained that there are multiple subtypes of PTCL based on their location within the body. Ai discussed a 2008 analysis of North American cases of PTCL and natural killer/T-cell lymphoma from 1990-2002, of which:
34% were PTCL, not otherwise specified;
16% were angioimmunoblastic T-cell lymphoma (AITL);
16% were anaplastic large cell lymphoma (ALCL), anaplastic lymphoma kinase (ALK)-positive;
7.8% were ALCL, ALK-negative;
5.8% were enteropathy-type;
5.4% were primary cutaneous ALCL; and
5.1% were extranodal natural killer/T-cell lymphoma, nasal type.
The remaining cases were adult T-cell leukemia/lymphoma, hepatosplenic, subcutaneous panniculitis-like, and unclassified.
International Prognostic Index Predicts Outcomes
“The subtype with the best outcome is ALCL, ALK-positive with a 5-year overall survival rate of 70% followed by ALK-negative ALCL at 50%, and all the other common subtypes at 30%,” Ai said.
Ai outlined the International Prognostic Index (IPI), a tool to predict clinical outcomes in patients with aggressive non-Hodgkin lymphoma based on risk factors. IPI assigns worse scores to patients aged > 60 years; patients who have higher (worse) performance scores, higher lactate dehydrogenase (LDH) levels, and more extranodal sites; and patients at stages III-IV.
First-Line Therapy: Consider Subtypes and CD30 Levels
Subtypes and CD30 expression levels are important factors in choosing therapy, Ai said, and 2019’s landmark ECHELON-2 study (updated in 2022) defines the standard.
Newly diagnosed patients who strongly express CD30 (ie, those with both types of ALCL) are recommended to be treated with A+CHP (brentuximab vedotin [BV] plus cyclophosphamide, doxorubicin, and prednisone).
Combination therapy of cyclophosphamide, doxorubicin, hydroxydaunorubicin, vincristine, and prednisone (CHOP) was the prior standard of care until the ECHELON-2 study, Ai said.
That trial, which randomized 452 patients with untreated PTCL (CD30 ≥ 10%) to A+CHP or CHOP, found that 5-year progression-free rates were 51.4% vs 43.0%, respectively (hazard ratio [HR], 0.70; 95% CI, 0.53-0.91). Five-year overall survival rates were 70.1% vs. 61.0%, respectively (HR, 0.72; 95% CI, 0.53-0.99).
The threshold CD30 level at which to turn to A+CHP—1%, 5%, or 10%—“is kind of a dealer’s choice,” Ai said. Her own cutoff is 1%.
“If they're < 1%, I tend not to do it,” Ai said. “It's usually much more expensive, as you can imagine.”
If CD30 < 1%, Ai recommends CHOP or, in younger patients, CHOP plus etoposide (CHOEP).
Follow-up treatments include autologous stem cell transplant (ASCT) and observation/maintenance, depending on factors such as subtype, fitness, and remission.
Transplant: Still Relevant
When ECHELON-2 was released, some clinicians wondered if ASCT was still warranted, Ai said. A posthoc exploratory analysis found a 62% reduction in relative risk for progression in patients who underwent transplants after reaching complete remission with A+CHP.
The findings provide support for transplant, she said.
For transplant-ineligible patients, a small analysis of BV and CHP followed by BV maintenance showed a progression-free survival curve that appeared to plateau after 18-24 months.
“You don't see this kind of curve very often. I was quite impressed,” Ai said. “If the patient is willing and able, I will give them BV cycles.”
Ai discloses relationships with ADC, AbbVie, Acrotech, Kite, and Kyowa Kirin.
SAN FRANCISCO – Peripheral T-cell lymphoma (PTCL) accounts for 4% of mature non-Hodgkin lymphoma cases in the US, or only about 4000 cases a year. While the number of patients is small, however, treatment for PTCL is complex due to wide variations in subtypes and survival rates, a hematologist-oncologist said at the March 21 Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
Weiyun Ai, MD, PhD, a clinical professor of medicine at University of California, San Francisco who specializes in lymphoma, explained that there are multiple subtypes of PTCL based on their location within the body. Ai discussed a 2008 analysis of North American cases of PTCL and natural killer/T-cell lymphoma from 1990-2002, of which:
34% were PTCL, not otherwise specified;
16% were angioimmunoblastic T-cell lymphoma (AITL);
16% were anaplastic large cell lymphoma (ALCL), anaplastic lymphoma kinase (ALK)-positive;
7.8% were ALCL, ALK-negative;
5.8% were enteropathy-type;
5.4% were primary cutaneous ALCL; and
5.1% were extranodal natural killer/T-cell lymphoma, nasal type.
The remaining cases were adult T-cell leukemia/lymphoma, hepatosplenic, subcutaneous panniculitis-like, and unclassified.
International Prognostic Index Predicts Outcomes
“The subtype with the best outcome is ALCL, ALK-positive with a 5-year overall survival rate of 70% followed by ALK-negative ALCL at 50%, and all the other common subtypes at 30%,” Ai said.
Ai outlined the International Prognostic Index (IPI), a tool to predict clinical outcomes in patients with aggressive non-Hodgkin lymphoma based on risk factors. IPI assigns worse scores to patients aged > 60 years; patients who have higher (worse) performance scores, higher lactate dehydrogenase (LDH) levels, and more extranodal sites; and patients at stages III-IV.
First-Line Therapy: Consider Subtypes and CD30 Levels
Subtypes and CD30 expression levels are important factors in choosing therapy, Ai said, and 2019’s landmark ECHELON-2 study (updated in 2022) defines the standard.
Newly diagnosed patients who strongly express CD30 (ie, those with both types of ALCL) are recommended to be treated with A+CHP (brentuximab vedotin [BV] plus cyclophosphamide, doxorubicin, and prednisone).
Combination therapy of cyclophosphamide, doxorubicin, hydroxydaunorubicin, vincristine, and prednisone (CHOP) was the prior standard of care until the ECHELON-2 study, Ai said.
That trial, which randomized 452 patients with untreated PTCL (CD30 ≥ 10%) to A+CHP or CHOP, found that 5-year progression-free rates were 51.4% vs 43.0%, respectively (hazard ratio [HR], 0.70; 95% CI, 0.53-0.91). Five-year overall survival rates were 70.1% vs. 61.0%, respectively (HR, 0.72; 95% CI, 0.53-0.99).
The threshold CD30 level at which to turn to A+CHP—1%, 5%, or 10%—“is kind of a dealer’s choice,” Ai said. Her own cutoff is 1%.
“If they're < 1%, I tend not to do it,” Ai said. “It's usually much more expensive, as you can imagine.”
If CD30 < 1%, Ai recommends CHOP or, in younger patients, CHOP plus etoposide (CHOEP).
Follow-up treatments include autologous stem cell transplant (ASCT) and observation/maintenance, depending on factors such as subtype, fitness, and remission.
Transplant: Still Relevant
When ECHELON-2 was released, some clinicians wondered if ASCT was still warranted, Ai said. A posthoc exploratory analysis found a 62% reduction in relative risk for progression in patients who underwent transplants after reaching complete remission with A+CHP.
The findings provide support for transplant, she said.
For transplant-ineligible patients, a small analysis of BV and CHP followed by BV maintenance showed a progression-free survival curve that appeared to plateau after 18-24 months.
“You don't see this kind of curve very often. I was quite impressed,” Ai said. “If the patient is willing and able, I will give them BV cycles.”
Ai discloses relationships with ADC, AbbVie, Acrotech, Kite, and Kyowa Kirin.
SAN FRANCISCO – Peripheral T-cell lymphoma (PTCL) accounts for 4% of mature non-Hodgkin lymphoma cases in the US, or only about 4000 cases a year. While the number of patients is small, however, treatment for PTCL is complex due to wide variations in subtypes and survival rates, a hematologist-oncologist said at the March 21 Association of VA Hematology/Oncology (AVAHO) regional meeting on lymphoma.
Weiyun Ai, MD, PhD, a clinical professor of medicine at University of California, San Francisco who specializes in lymphoma, explained that there are multiple subtypes of PTCL based on their location within the body. Ai discussed a 2008 analysis of North American cases of PTCL and natural killer/T-cell lymphoma from 1990-2002, of which:
34% were PTCL, not otherwise specified;
16% were angioimmunoblastic T-cell lymphoma (AITL);
16% were anaplastic large cell lymphoma (ALCL), anaplastic lymphoma kinase (ALK)-positive;
7.8% were ALCL, ALK-negative;
5.8% were enteropathy-type;
5.4% were primary cutaneous ALCL; and
5.1% were extranodal natural killer/T-cell lymphoma, nasal type.
The remaining cases were adult T-cell leukemia/lymphoma, hepatosplenic, subcutaneous panniculitis-like, and unclassified.
International Prognostic Index Predicts Outcomes
“The subtype with the best outcome is ALCL, ALK-positive with a 5-year overall survival rate of 70% followed by ALK-negative ALCL at 50%, and all the other common subtypes at 30%,” Ai said.
Ai outlined the International Prognostic Index (IPI), a tool to predict clinical outcomes in patients with aggressive non-Hodgkin lymphoma based on risk factors. IPI assigns worse scores to patients aged > 60 years; patients who have higher (worse) performance scores, higher lactate dehydrogenase (LDH) levels, and more extranodal sites; and patients at stages III-IV.
First-Line Therapy: Consider Subtypes and CD30 Levels
Subtypes and CD30 expression levels are important factors in choosing therapy, Ai said, and 2019’s landmark ECHELON-2 study (updated in 2022) defines the standard.
Newly diagnosed patients who strongly express CD30 (ie, those with both types of ALCL) are recommended to be treated with A+CHP (brentuximab vedotin [BV] plus cyclophosphamide, doxorubicin, and prednisone).
Combination therapy of cyclophosphamide, doxorubicin, hydroxydaunorubicin, vincristine, and prednisone (CHOP) was the prior standard of care until the ECHELON-2 study, Ai said.
That trial, which randomized 452 patients with untreated PTCL (CD30 ≥ 10%) to A+CHP or CHOP, found that 5-year progression-free rates were 51.4% vs 43.0%, respectively (hazard ratio [HR], 0.70; 95% CI, 0.53-0.91). Five-year overall survival rates were 70.1% vs. 61.0%, respectively (HR, 0.72; 95% CI, 0.53-0.99).
The threshold CD30 level at which to turn to A+CHP—1%, 5%, or 10%—“is kind of a dealer’s choice,” Ai said. Her own cutoff is 1%.
“If they're < 1%, I tend not to do it,” Ai said. “It's usually much more expensive, as you can imagine.”
If CD30 < 1%, Ai recommends CHOP or, in younger patients, CHOP plus etoposide (CHOEP).
Follow-up treatments include autologous stem cell transplant (ASCT) and observation/maintenance, depending on factors such as subtype, fitness, and remission.
Transplant: Still Relevant
When ECHELON-2 was released, some clinicians wondered if ASCT was still warranted, Ai said. A posthoc exploratory analysis found a 62% reduction in relative risk for progression in patients who underwent transplants after reaching complete remission with A+CHP.
The findings provide support for transplant, she said.
For transplant-ineligible patients, a small analysis of BV and CHP followed by BV maintenance showed a progression-free survival curve that appeared to plateau after 18-24 months.
“You don't see this kind of curve very often. I was quite impressed,” Ai said. “If the patient is willing and able, I will give them BV cycles.”
Ai discloses relationships with ADC, AbbVie, Acrotech, Kite, and Kyowa Kirin.
AVAHO Regional Meeting Addresses Complex World of Peripheral T-Cell Lymphoma
AVAHO Regional Meeting Addresses Complex World of Peripheral T-Cell Lymphoma
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
Introduction
Cutaneous T-cell lymphomas (CTCL) are a heterogenous group of rare extranodal non-Hodgkin lymphomas that are caused by the accumulation of neoplastic lymphocytes in the skin.1,2 According to the Surveillance, Epidemiology, and End Results database, a total of 14,942 CTCL cases were recorded between 2000 and 2018.3 The incidence rate for all CTCLs is 8.55 per million and appears to be rising. The causes of such an increase are multifactorial and may be related to better diagnostic tools and increased physician awareness.
The incidence of CTCLs also increases with age. The median age at diagnosis is mid-50s but the incidence of CTCLs is 4-fold greater in patients aged 70 years and older.2 Furthermore, men and Black individuals have the highest incidence rates for CTCLs.2,3 More than 10 types of CTCLs have been identified based on biology, histopathology, and clinical features. CTCL types can be either indolent or aggressive.1,4 Approximately 75% of all primary cutaneous lymphomas consist of CTCLs, including mycosis fungoides (MF), Sézary syndrome (SS), or CD30+ lymphoproliferative disorders (lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma).
The most common CTCL is MF, a clinically heterogeneous, often indolent disease that tends to progress over years or decades.1 This condition classically presents as cutaneous erythematous patches or plaques in sun-protected areas, ie, demonstrating a bathing suit distribution.5 Rarely, MF can present as or progress to more aggressive disease, with infiltrative plaques or tumors. For MF, 5- and 10-year survival ranges from 49% to 100% depending on the stage at diagnosis.1
The most common aggressive CTCL is SS, characterized by erythroderma, intractable pruritis, and the presence of neoplastic clonal T cells (eg, Sézary cells) in the skin, peripheral blood, and/or lymph nodes, with a Sézary cell absolute count of ≥ 1,000 cells/mm3.1,2 SS tends to progress more rapidly than MF and has a worse prognosis, with 5-year survival ranging from 10% to 50%.1,4
Definitive Diagnosis
Diagnosis of CTCL requires the neoplastic T cells be confined to the skin.2 Thus, diagnostic evaluation should involve a comprehensive physical examination, skin biopsy, and staging blood tests including a peripheral blood flow cytometry if indicated. Sometimes, radiologic imaging is needed, and if there are any abnormalities found on staging blood tests or imaging, lymph node and bone marrow biopsy may be necessary.1
MF
MF mimics a wide variety of dermatological diseases, with nearly 50 different clinical entities in the differential, making diagnosis challenging.5 Clinical findings are heterogenous, and symptoms may be attributed to benign diseases, eg, eczema, or psoriasis. Pathological features may be nonspecific and subtle in the early stages of the disease and overlap with reactive processes; therefore, multiple biopsies performed during the disease course may be required to reach a definitive diagnosis. Creating a further challenge is the potential for skin-directed therapies (such as topical steroids) to interfere with pathological assessment at the time of biopsy.2 Thus, obtaining a definitive diagnosis for MF, particularly in the patch or plaque stage, could take a median of 4 years but can take up to 4 decades.2,5
A definitive diagnosis for MF can be made using clinical and histopathological features. Possible ancillary studies (if indicated) include determination of T-cell clonality by polymerase chain reaction or next-generation sequencing methods, and assessment for aberrant loss of T-cell antigen expression by immunohistochemical staining.2
SS
Clinical features of SS may be similar to erythrodermic inflammatory dermatoses, and thus the gold standard for diagnosis is peripheral blood involvement and assessing for clonally related neoplastic T-cell populations.1 Histopathological findings on skin biopsy are often nonspecific.4 The currently proposed International Society for Cutaneous Lymphomas criteria for SS integrate clinical, histopathological, immunophenotyping, and molecular studies.2
Benefits of a Multidisciplinary Team Care Approach
Early-stage MF with limited disease can be managed by a dermatologist, but advanced cases often benefit from a multidisciplinary team care model, including hematology-oncology, dermatology, and radiation oncology.5,6 Several different CTCL care models exist that incorporate resource allocation, staffing availability, and institutional practices developed over time. Regardless of whether care is delivered in a specialized CTCL clinic or a community practice setting, a multidisciplinary team care approach is crucial for patients with advanced-stage CTCL. Dermatologists, hematologist-oncologists, and radiation oncologists may see a patient together or separately, depending on clinical context, and collaborate to formulate the assessment, treatment plan, and address the patient’s questions and concerns. In addition, supportive staff including patient assistance coordinators, pharmacists, behavior health specialists, and palliative care specialists may be included to address the patients’ mental health needs as considerable morbidity from pain, itching, and disfigurement occurs with MF and SS—putting patients at a greater risk for social isolation and depression.7
There are several benefits to using a multidisciplinary team care model for managing CTCLs. Different specialties can provide various services and treatment options for patients to consider. Dermatologists perform skin biopsies to monitor disease progression and can administer skin-directed treatments such as phototherapy; radiation oncologists can administer radiation treatment; and oncologists can administer systemic therapies that are outside the scope of dermatology.8 The coordination of specialty visits can improve patient satisfaction.
Treatment Goals and Disease Management
Goals for treatment include delaying progression, reducing disease burden, and improving or preserving quality of life.5 Decision-making for treating CTCLs should involve preserving potential active treatments for when they are needed during an extended disease course, and mitigating associated burdens of logistical, financial, and physical toxicity.1
A variety of therapeutic modalities are available for CTCL that target tumor cells and boost antitumor responses, including topical therapies, phototherapy, radiation, chemotherapy, retinoids, and immune-modulating drugs (Table). Because no specific driver mutations have been identified for CTCLs, recent targeted therapy development has focused on various immunomodulators, small molecule inhibitors, monoclonal antibodies, and antibody-drug conjugates.1 Lastly, for high-risk patients with persistent disease or disease that is refractory to multiple previous therapies, allogenic hematopoietic stem cell transplantation as a potential therapy to induce durable remission may be considered, with careful attention paid to the timing of its use as well as disease and patient characteristics.9
Table. Therapies for CTCL Care9,10,a
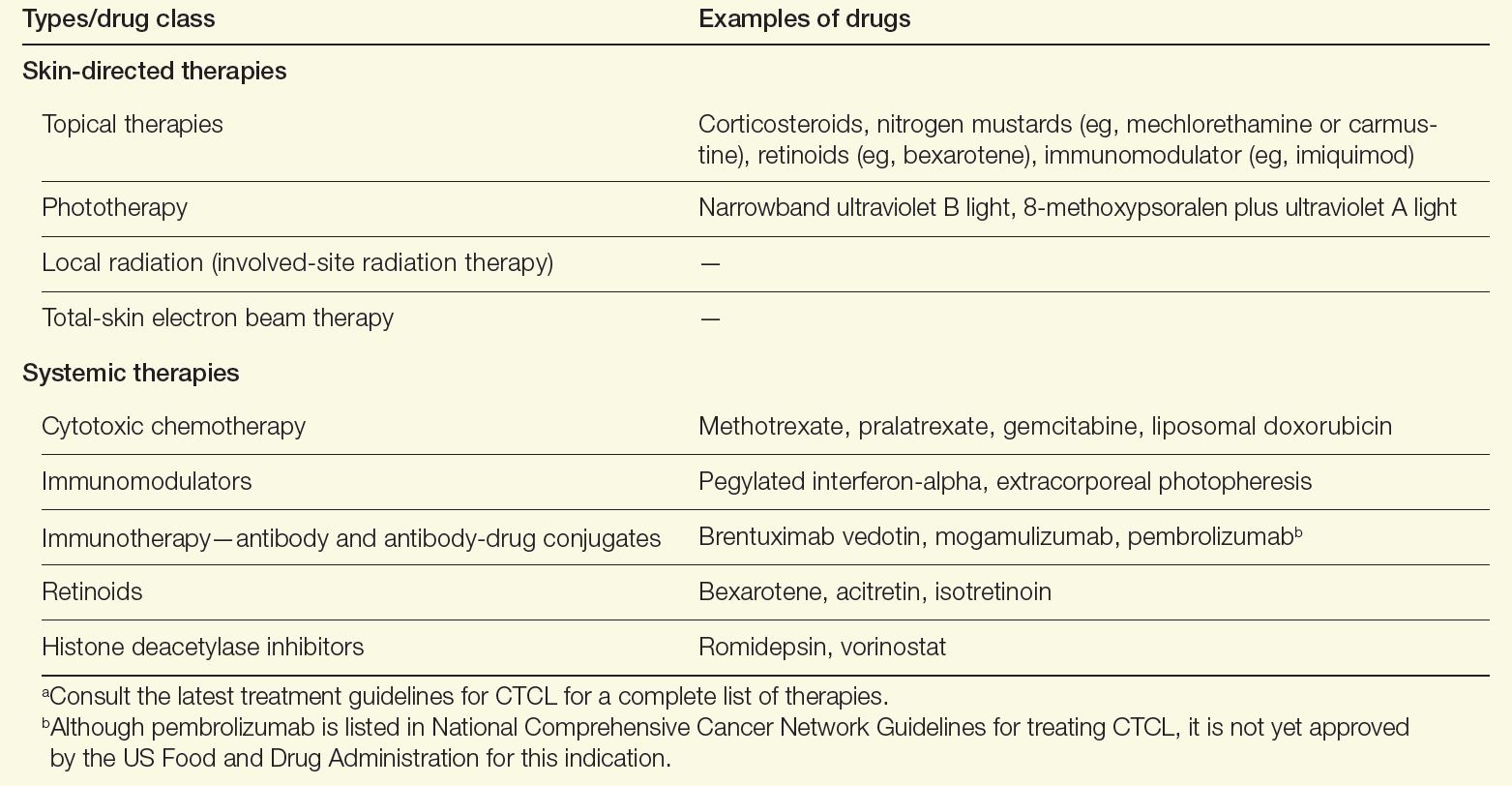
Alternatively for early-stage MF, a “watch-and-wait” approach depending on the site of lesions and disease evolution may be an option, as this approach is not associated with a worsening of the disease course or survival.1 Furthermore, aggressive treatments during early stages have not been found to modify the disease course or survival, emphasizing the need for tailoring treatments based on the extent of involvement of the skin and extracutaneous sites.1,10 New strategies in development to treat CTCL include immune-checkpoint inhibitors and chimeric antigen receptor T-cell therapies. Both strategies focus on engaging the immune system to better combat lymphoma.11,12
Outlook for Patients With CTCL
Using a multidisciplinary care approach is the optimal way to deliver the complex care required for CTCL.5 Such an approach can reduce the time to a definitive diagnosis and accurately stage and risk-stratify the disease. A stage-based treatment approach using sequential therapies in an escalated fashion can help reserve active treatments for advanced disease management and maintain quality of life for patients with CTCL.1,2
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
- Dummer R, Vermeer MH, Scarisbrick JJ, et al. Cutaneous T cell lymphoma. Nat Rev Dis Primers. 2021;7(1):61. doi:10.1038/s41572-021-00296-9
- Hristov AC, Tejasvi T, Wilcox RA. Cutaneous T-cell lymphomas: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023;98(1):193-209. doi:10.1002/ajh.26760
- Cai ZR, Chen ML, Weinstock MA, Kim YH, Novoa RA, Linos E. Incidence trends of primary cutaneous T-cell lymphoma in the US from 2000 to 2018: a SEER population data analysis. JAMA Oncol. 2022;8(11):1690-1692. doi:10.1001/jamaoncol.2022.3236
- Saleh JS, Subtil A, Hristov AC. Primary cutaneous T-cell lymphoma: a review of the most common entities with focus on recent updates. Hum Pathol. 2023;140:75-100. doi:10.1016/j.humpath.2023.09.009
- Vitiello P, Sagnelli C, Ronchi A, et al. Multidisciplinary approach to the diagnosis and therapy of mycosis fungoides. Healthcare (Basel). 2023;11(4):614. doi:10.3390/healthcare11040614
- Morgenroth S, Roggo A, Pawlik L, Dummer R, Ramelyte E. What is new in cutaneous T cell lymphoma? Curr Oncol Rep. 2023;25(11):1397-1408. doi:10.1007/s11912-023-01464-8
- Molloy K, Jonak C, Woei-A-Jin FJSH, et al. Characteristics associated with significantly worse quality of life in mycosis fungoides/Sézary syndrome from the Prospective Cutaneous Lymphoma International Prognostic Index (PROCLIPI) study. Br J Dermatol. 2020;182(3):770-779. doi:10.1111/bjd.18089
- Tyler KH, Haverkos BM, Hastings J, et al. The role of an integrated multidisciplinary clinic in the management of patients with cutaneous lymphoma. Front Oncol. 2015;5:136. doi:10.3389/fonc.2015.00136
- National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: primary cutaneous lymphomas. Version 3.2024. August 22, 2024. Accessed October 6, 2024. https://www.nccn.org/professionals/physician_gls/pdf/primary_cutaneous.pdf
- Goel RR, Rook AH. Immunobiology and treatment of cutaneous T-cell lymphoma. Expert Rev Clin Immunol. 2024;20(8):985-996. doi:10.1080/1744666X.2024.2326035
- Iyer SP, Sica RA, Ho PJ, et al. S262: The COBALT-LYM study of CTX130: a phase 1 dose escalation study of CD70-targeted allogeneic CRISPR-Cas9–engineered CAR T cells in patients with relapsed/refractory (R/R) T-cell malignancies. HemaSphere. 2022;6(S3):163-164. doi:10.1097/01.HS9.0000843940.96598.e2
- Khodadoust MS, Rook AH, Porcu P, et al. Pembrolizumab in relapsed and refractory mycosis fungoides and Sézary syndrome: a multicenter phase II study. J Clin Oncol. 2020;38(1):20-28. doi:10.1200/JCO.19.01056
Introduction
Cutaneous T-cell lymphomas (CTCL) are a heterogenous group of rare extranodal non-Hodgkin lymphomas that are caused by the accumulation of neoplastic lymphocytes in the skin.1,2 According to the Surveillance, Epidemiology, and End Results database, a total of 14,942 CTCL cases were recorded between 2000 and 2018.3 The incidence rate for all CTCLs is 8.55 per million and appears to be rising. The causes of such an increase are multifactorial and may be related to better diagnostic tools and increased physician awareness.
The incidence of CTCLs also increases with age. The median age at diagnosis is mid-50s but the incidence of CTCLs is 4-fold greater in patients aged 70 years and older.2 Furthermore, men and Black individuals have the highest incidence rates for CTCLs.2,3 More than 10 types of CTCLs have been identified based on biology, histopathology, and clinical features. CTCL types can be either indolent or aggressive.1,4 Approximately 75% of all primary cutaneous lymphomas consist of CTCLs, including mycosis fungoides (MF), Sézary syndrome (SS), or CD30+ lymphoproliferative disorders (lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma).
The most common CTCL is MF, a clinically heterogeneous, often indolent disease that tends to progress over years or decades.1 This condition classically presents as cutaneous erythematous patches or plaques in sun-protected areas, ie, demonstrating a bathing suit distribution.5 Rarely, MF can present as or progress to more aggressive disease, with infiltrative plaques or tumors. For MF, 5- and 10-year survival ranges from 49% to 100% depending on the stage at diagnosis.1
The most common aggressive CTCL is SS, characterized by erythroderma, intractable pruritis, and the presence of neoplastic clonal T cells (eg, Sézary cells) in the skin, peripheral blood, and/or lymph nodes, with a Sézary cell absolute count of ≥ 1,000 cells/mm3.1,2 SS tends to progress more rapidly than MF and has a worse prognosis, with 5-year survival ranging from 10% to 50%.1,4
Definitive Diagnosis
Diagnosis of CTCL requires the neoplastic T cells be confined to the skin.2 Thus, diagnostic evaluation should involve a comprehensive physical examination, skin biopsy, and staging blood tests including a peripheral blood flow cytometry if indicated. Sometimes, radiologic imaging is needed, and if there are any abnormalities found on staging blood tests or imaging, lymph node and bone marrow biopsy may be necessary.1
MF
MF mimics a wide variety of dermatological diseases, with nearly 50 different clinical entities in the differential, making diagnosis challenging.5 Clinical findings are heterogenous, and symptoms may be attributed to benign diseases, eg, eczema, or psoriasis. Pathological features may be nonspecific and subtle in the early stages of the disease and overlap with reactive processes; therefore, multiple biopsies performed during the disease course may be required to reach a definitive diagnosis. Creating a further challenge is the potential for skin-directed therapies (such as topical steroids) to interfere with pathological assessment at the time of biopsy.2 Thus, obtaining a definitive diagnosis for MF, particularly in the patch or plaque stage, could take a median of 4 years but can take up to 4 decades.2,5
A definitive diagnosis for MF can be made using clinical and histopathological features. Possible ancillary studies (if indicated) include determination of T-cell clonality by polymerase chain reaction or next-generation sequencing methods, and assessment for aberrant loss of T-cell antigen expression by immunohistochemical staining.2
SS
Clinical features of SS may be similar to erythrodermic inflammatory dermatoses, and thus the gold standard for diagnosis is peripheral blood involvement and assessing for clonally related neoplastic T-cell populations.1 Histopathological findings on skin biopsy are often nonspecific.4 The currently proposed International Society for Cutaneous Lymphomas criteria for SS integrate clinical, histopathological, immunophenotyping, and molecular studies.2
Benefits of a Multidisciplinary Team Care Approach
Early-stage MF with limited disease can be managed by a dermatologist, but advanced cases often benefit from a multidisciplinary team care model, including hematology-oncology, dermatology, and radiation oncology.5,6 Several different CTCL care models exist that incorporate resource allocation, staffing availability, and institutional practices developed over time. Regardless of whether care is delivered in a specialized CTCL clinic or a community practice setting, a multidisciplinary team care approach is crucial for patients with advanced-stage CTCL. Dermatologists, hematologist-oncologists, and radiation oncologists may see a patient together or separately, depending on clinical context, and collaborate to formulate the assessment, treatment plan, and address the patient’s questions and concerns. In addition, supportive staff including patient assistance coordinators, pharmacists, behavior health specialists, and palliative care specialists may be included to address the patients’ mental health needs as considerable morbidity from pain, itching, and disfigurement occurs with MF and SS—putting patients at a greater risk for social isolation and depression.7
There are several benefits to using a multidisciplinary team care model for managing CTCLs. Different specialties can provide various services and treatment options for patients to consider. Dermatologists perform skin biopsies to monitor disease progression and can administer skin-directed treatments such as phototherapy; radiation oncologists can administer radiation treatment; and oncologists can administer systemic therapies that are outside the scope of dermatology.8 The coordination of specialty visits can improve patient satisfaction.
Treatment Goals and Disease Management
Goals for treatment include delaying progression, reducing disease burden, and improving or preserving quality of life.5 Decision-making for treating CTCLs should involve preserving potential active treatments for when they are needed during an extended disease course, and mitigating associated burdens of logistical, financial, and physical toxicity.1
A variety of therapeutic modalities are available for CTCL that target tumor cells and boost antitumor responses, including topical therapies, phototherapy, radiation, chemotherapy, retinoids, and immune-modulating drugs (Table). Because no specific driver mutations have been identified for CTCLs, recent targeted therapy development has focused on various immunomodulators, small molecule inhibitors, monoclonal antibodies, and antibody-drug conjugates.1 Lastly, for high-risk patients with persistent disease or disease that is refractory to multiple previous therapies, allogenic hematopoietic stem cell transplantation as a potential therapy to induce durable remission may be considered, with careful attention paid to the timing of its use as well as disease and patient characteristics.9
Table. Therapies for CTCL Care9,10,a

Alternatively for early-stage MF, a “watch-and-wait” approach depending on the site of lesions and disease evolution may be an option, as this approach is not associated with a worsening of the disease course or survival.1 Furthermore, aggressive treatments during early stages have not been found to modify the disease course or survival, emphasizing the need for tailoring treatments based on the extent of involvement of the skin and extracutaneous sites.1,10 New strategies in development to treat CTCL include immune-checkpoint inhibitors and chimeric antigen receptor T-cell therapies. Both strategies focus on engaging the immune system to better combat lymphoma.11,12
Outlook for Patients With CTCL
Using a multidisciplinary care approach is the optimal way to deliver the complex care required for CTCL.5 Such an approach can reduce the time to a definitive diagnosis and accurately stage and risk-stratify the disease. A stage-based treatment approach using sequential therapies in an escalated fashion can help reserve active treatments for advanced disease management and maintain quality of life for patients with CTCL.1,2
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
Introduction
Cutaneous T-cell lymphomas (CTCL) are a heterogenous group of rare extranodal non-Hodgkin lymphomas that are caused by the accumulation of neoplastic lymphocytes in the skin.1,2 According to the Surveillance, Epidemiology, and End Results database, a total of 14,942 CTCL cases were recorded between 2000 and 2018.3 The incidence rate for all CTCLs is 8.55 per million and appears to be rising. The causes of such an increase are multifactorial and may be related to better diagnostic tools and increased physician awareness.
The incidence of CTCLs also increases with age. The median age at diagnosis is mid-50s but the incidence of CTCLs is 4-fold greater in patients aged 70 years and older.2 Furthermore, men and Black individuals have the highest incidence rates for CTCLs.2,3 More than 10 types of CTCLs have been identified based on biology, histopathology, and clinical features. CTCL types can be either indolent or aggressive.1,4 Approximately 75% of all primary cutaneous lymphomas consist of CTCLs, including mycosis fungoides (MF), Sézary syndrome (SS), or CD30+ lymphoproliferative disorders (lymphomatoid papulosis and primary cutaneous anaplastic large cell lymphoma).
The most common CTCL is MF, a clinically heterogeneous, often indolent disease that tends to progress over years or decades.1 This condition classically presents as cutaneous erythematous patches or plaques in sun-protected areas, ie, demonstrating a bathing suit distribution.5 Rarely, MF can present as or progress to more aggressive disease, with infiltrative plaques or tumors. For MF, 5- and 10-year survival ranges from 49% to 100% depending on the stage at diagnosis.1
The most common aggressive CTCL is SS, characterized by erythroderma, intractable pruritis, and the presence of neoplastic clonal T cells (eg, Sézary cells) in the skin, peripheral blood, and/or lymph nodes, with a Sézary cell absolute count of ≥ 1,000 cells/mm3.1,2 SS tends to progress more rapidly than MF and has a worse prognosis, with 5-year survival ranging from 10% to 50%.1,4
Definitive Diagnosis
Diagnosis of CTCL requires the neoplastic T cells be confined to the skin.2 Thus, diagnostic evaluation should involve a comprehensive physical examination, skin biopsy, and staging blood tests including a peripheral blood flow cytometry if indicated. Sometimes, radiologic imaging is needed, and if there are any abnormalities found on staging blood tests or imaging, lymph node and bone marrow biopsy may be necessary.1
MF
MF mimics a wide variety of dermatological diseases, with nearly 50 different clinical entities in the differential, making diagnosis challenging.5 Clinical findings are heterogenous, and symptoms may be attributed to benign diseases, eg, eczema, or psoriasis. Pathological features may be nonspecific and subtle in the early stages of the disease and overlap with reactive processes; therefore, multiple biopsies performed during the disease course may be required to reach a definitive diagnosis. Creating a further challenge is the potential for skin-directed therapies (such as topical steroids) to interfere with pathological assessment at the time of biopsy.2 Thus, obtaining a definitive diagnosis for MF, particularly in the patch or plaque stage, could take a median of 4 years but can take up to 4 decades.2,5
A definitive diagnosis for MF can be made using clinical and histopathological features. Possible ancillary studies (if indicated) include determination of T-cell clonality by polymerase chain reaction or next-generation sequencing methods, and assessment for aberrant loss of T-cell antigen expression by immunohistochemical staining.2
SS
Clinical features of SS may be similar to erythrodermic inflammatory dermatoses, and thus the gold standard for diagnosis is peripheral blood involvement and assessing for clonally related neoplastic T-cell populations.1 Histopathological findings on skin biopsy are often nonspecific.4 The currently proposed International Society for Cutaneous Lymphomas criteria for SS integrate clinical, histopathological, immunophenotyping, and molecular studies.2
Benefits of a Multidisciplinary Team Care Approach
Early-stage MF with limited disease can be managed by a dermatologist, but advanced cases often benefit from a multidisciplinary team care model, including hematology-oncology, dermatology, and radiation oncology.5,6 Several different CTCL care models exist that incorporate resource allocation, staffing availability, and institutional practices developed over time. Regardless of whether care is delivered in a specialized CTCL clinic or a community practice setting, a multidisciplinary team care approach is crucial for patients with advanced-stage CTCL. Dermatologists, hematologist-oncologists, and radiation oncologists may see a patient together or separately, depending on clinical context, and collaborate to formulate the assessment, treatment plan, and address the patient’s questions and concerns. In addition, supportive staff including patient assistance coordinators, pharmacists, behavior health specialists, and palliative care specialists may be included to address the patients’ mental health needs as considerable morbidity from pain, itching, and disfigurement occurs with MF and SS—putting patients at a greater risk for social isolation and depression.7
There are several benefits to using a multidisciplinary team care model for managing CTCLs. Different specialties can provide various services and treatment options for patients to consider. Dermatologists perform skin biopsies to monitor disease progression and can administer skin-directed treatments such as phototherapy; radiation oncologists can administer radiation treatment; and oncologists can administer systemic therapies that are outside the scope of dermatology.8 The coordination of specialty visits can improve patient satisfaction.
Treatment Goals and Disease Management
Goals for treatment include delaying progression, reducing disease burden, and improving or preserving quality of life.5 Decision-making for treating CTCLs should involve preserving potential active treatments for when they are needed during an extended disease course, and mitigating associated burdens of logistical, financial, and physical toxicity.1
A variety of therapeutic modalities are available for CTCL that target tumor cells and boost antitumor responses, including topical therapies, phototherapy, radiation, chemotherapy, retinoids, and immune-modulating drugs (Table). Because no specific driver mutations have been identified for CTCLs, recent targeted therapy development has focused on various immunomodulators, small molecule inhibitors, monoclonal antibodies, and antibody-drug conjugates.1 Lastly, for high-risk patients with persistent disease or disease that is refractory to multiple previous therapies, allogenic hematopoietic stem cell transplantation as a potential therapy to induce durable remission may be considered, with careful attention paid to the timing of its use as well as disease and patient characteristics.9
Table. Therapies for CTCL Care9,10,a

Alternatively for early-stage MF, a “watch-and-wait” approach depending on the site of lesions and disease evolution may be an option, as this approach is not associated with a worsening of the disease course or survival.1 Furthermore, aggressive treatments during early stages have not been found to modify the disease course or survival, emphasizing the need for tailoring treatments based on the extent of involvement of the skin and extracutaneous sites.1,10 New strategies in development to treat CTCL include immune-checkpoint inhibitors and chimeric antigen receptor T-cell therapies. Both strategies focus on engaging the immune system to better combat lymphoma.11,12
Outlook for Patients With CTCL
Using a multidisciplinary care approach is the optimal way to deliver the complex care required for CTCL.5 Such an approach can reduce the time to a definitive diagnosis and accurately stage and risk-stratify the disease. A stage-based treatment approach using sequential therapies in an escalated fashion can help reserve active treatments for advanced disease management and maintain quality of life for patients with CTCL.1,2
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
- Dummer R, Vermeer MH, Scarisbrick JJ, et al. Cutaneous T cell lymphoma. Nat Rev Dis Primers. 2021;7(1):61. doi:10.1038/s41572-021-00296-9
- Hristov AC, Tejasvi T, Wilcox RA. Cutaneous T-cell lymphomas: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023;98(1):193-209. doi:10.1002/ajh.26760
- Cai ZR, Chen ML, Weinstock MA, Kim YH, Novoa RA, Linos E. Incidence trends of primary cutaneous T-cell lymphoma in the US from 2000 to 2018: a SEER population data analysis. JAMA Oncol. 2022;8(11):1690-1692. doi:10.1001/jamaoncol.2022.3236
- Saleh JS, Subtil A, Hristov AC. Primary cutaneous T-cell lymphoma: a review of the most common entities with focus on recent updates. Hum Pathol. 2023;140:75-100. doi:10.1016/j.humpath.2023.09.009
- Vitiello P, Sagnelli C, Ronchi A, et al. Multidisciplinary approach to the diagnosis and therapy of mycosis fungoides. Healthcare (Basel). 2023;11(4):614. doi:10.3390/healthcare11040614
- Morgenroth S, Roggo A, Pawlik L, Dummer R, Ramelyte E. What is new in cutaneous T cell lymphoma? Curr Oncol Rep. 2023;25(11):1397-1408. doi:10.1007/s11912-023-01464-8
- Molloy K, Jonak C, Woei-A-Jin FJSH, et al. Characteristics associated with significantly worse quality of life in mycosis fungoides/Sézary syndrome from the Prospective Cutaneous Lymphoma International Prognostic Index (PROCLIPI) study. Br J Dermatol. 2020;182(3):770-779. doi:10.1111/bjd.18089
- Tyler KH, Haverkos BM, Hastings J, et al. The role of an integrated multidisciplinary clinic in the management of patients with cutaneous lymphoma. Front Oncol. 2015;5:136. doi:10.3389/fonc.2015.00136
- National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: primary cutaneous lymphomas. Version 3.2024. August 22, 2024. Accessed October 6, 2024. https://www.nccn.org/professionals/physician_gls/pdf/primary_cutaneous.pdf
- Goel RR, Rook AH. Immunobiology and treatment of cutaneous T-cell lymphoma. Expert Rev Clin Immunol. 2024;20(8):985-996. doi:10.1080/1744666X.2024.2326035
- Iyer SP, Sica RA, Ho PJ, et al. S262: The COBALT-LYM study of CTX130: a phase 1 dose escalation study of CD70-targeted allogeneic CRISPR-Cas9–engineered CAR T cells in patients with relapsed/refractory (R/R) T-cell malignancies. HemaSphere. 2022;6(S3):163-164. doi:10.1097/01.HS9.0000843940.96598.e2
- Khodadoust MS, Rook AH, Porcu P, et al. Pembrolizumab in relapsed and refractory mycosis fungoides and Sézary syndrome: a multicenter phase II study. J Clin Oncol. 2020;38(1):20-28. doi:10.1200/JCO.19.01056
- Dummer R, Vermeer MH, Scarisbrick JJ, et al. Cutaneous T cell lymphoma. Nat Rev Dis Primers. 2021;7(1):61. doi:10.1038/s41572-021-00296-9
- Hristov AC, Tejasvi T, Wilcox RA. Cutaneous T-cell lymphomas: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023;98(1):193-209. doi:10.1002/ajh.26760
- Cai ZR, Chen ML, Weinstock MA, Kim YH, Novoa RA, Linos E. Incidence trends of primary cutaneous T-cell lymphoma in the US from 2000 to 2018: a SEER population data analysis. JAMA Oncol. 2022;8(11):1690-1692. doi:10.1001/jamaoncol.2022.3236
- Saleh JS, Subtil A, Hristov AC. Primary cutaneous T-cell lymphoma: a review of the most common entities with focus on recent updates. Hum Pathol. 2023;140:75-100. doi:10.1016/j.humpath.2023.09.009
- Vitiello P, Sagnelli C, Ronchi A, et al. Multidisciplinary approach to the diagnosis and therapy of mycosis fungoides. Healthcare (Basel). 2023;11(4):614. doi:10.3390/healthcare11040614
- Morgenroth S, Roggo A, Pawlik L, Dummer R, Ramelyte E. What is new in cutaneous T cell lymphoma? Curr Oncol Rep. 2023;25(11):1397-1408. doi:10.1007/s11912-023-01464-8
- Molloy K, Jonak C, Woei-A-Jin FJSH, et al. Characteristics associated with significantly worse quality of life in mycosis fungoides/Sézary syndrome from the Prospective Cutaneous Lymphoma International Prognostic Index (PROCLIPI) study. Br J Dermatol. 2020;182(3):770-779. doi:10.1111/bjd.18089
- Tyler KH, Haverkos BM, Hastings J, et al. The role of an integrated multidisciplinary clinic in the management of patients with cutaneous lymphoma. Front Oncol. 2015;5:136. doi:10.3389/fonc.2015.00136
- National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: primary cutaneous lymphomas. Version 3.2024. August 22, 2024. Accessed October 6, 2024. https://www.nccn.org/professionals/physician_gls/pdf/primary_cutaneous.pdf
- Goel RR, Rook AH. Immunobiology and treatment of cutaneous T-cell lymphoma. Expert Rev Clin Immunol. 2024;20(8):985-996. doi:10.1080/1744666X.2024.2326035
- Iyer SP, Sica RA, Ho PJ, et al. S262: The COBALT-LYM study of CTX130: a phase 1 dose escalation study of CD70-targeted allogeneic CRISPR-Cas9–engineered CAR T cells in patients with relapsed/refractory (R/R) T-cell malignancies. HemaSphere. 2022;6(S3):163-164. doi:10.1097/01.HS9.0000843940.96598.e2
- Khodadoust MS, Rook AH, Porcu P, et al. Pembrolizumab in relapsed and refractory mycosis fungoides and Sézary syndrome: a multicenter phase II study. J Clin Oncol. 2020;38(1):20-28. doi:10.1200/JCO.19.01056
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
2024 Rare Diseases Report: Hematology and Oncology
2024 Rare Diseases Report: Hematology and Oncology
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
2024 Rare Diseases Report: Hematology and Oncology
2024 Rare Diseases Report: Hematology and Oncology
New Cancer Drugs: Do Patients Prefer Faster Access or Clinical Benefit?
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
FROM THE LANCET ONCOLOGY
New Cancer Vaccines on the Horizon: Renewed Hope or Hype?
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Inside the Patient-Oncologist Bond: Why It’s Often So Strong
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Michigan Oncologist Charged in Scheme to Illegally Sell Cancer Drugs
In late October, a federal grand jury charged a Detroit-area medical oncologist Naveed Aslam, MD, in an indictment for his part in a scheme to illegally sell cancer drugs.
According to the indictment, Aslam acquired and sold more than $17 million in cancer drugs and personally netted more than $2.5 million during the scheme.
The charges against Aslam, filed on October 23 in the US District Court for the Eastern District of Michigan, include 10 counts of illegally selling or trading prescription drugs and one count of conspiring to do so.
“Dr. Aslam’s alleged participation in this scheme not only allowed him to profit unlawfully from the sale of cancer drugs but it also posed a serious threat by potentially placing these medications into the wrong hands,” Cheyvoryea Gibson, special agent in charge of the FBI in Michigan, said in a press release announcing the indictment.
The investigation is being conducted jointly by the FBI, the US Food and Drug Administration (FDA), the US Department of Health and Human Services Office of Inspector General, and Homeland Security Investigations.
The indictment alleges that Aslam was recruited by an unnamed operator of a Michigan corporation that engaged in business as a retail pharmacy and in the wholesale distribution of expensive prescription drugs, largely oncology drugs.
According to the indictment, Aslam and the operator came to an agreement where Aslam would purchase these expensive drugs from an authorized distributor under the false pretense that he was going to prescribe them to patients.
Instead, Aslam allegedly “sold and transferred the prescription drugs” to or through the Michigan business, with involvement from the unnamed operator and a second unnamed individual.
The unnamed individuals “identified customers interested in buying prescription cancer drugs” and “communicated with Dr. Aslam about what cancer drugs were requested,” according to the press release. “Dr. Aslam used his access to certain cancer drugs through his medical practice, Somerset Hematology and Oncology, P.C., to order and purchase the cancer drugs from his supplier.”
The indictment lays out that Aslam allegedly profited from this scheme in several ways, which included charging the Michigan business more than he paid the distributor for the drugs, sharing the profits when the business resold the drugs at a markup, and receiving rebates and discounts from the distributor “based on the amount of qualifying drugs he purchased and resold.”
According to the indictment, the scheme ran from early 2019 to mid-2023 and included four antibody drug conjugates — trastuzumab deruxtecan (Enhertu), enfortumab vedotin (Padcev), tisotumab vedotin (Tivdak), and sacituzumab govitecan (Trodelvy) — and the monoclonal antibody mogamulizumab (Poteligeo) for cutaneous T-cell lymphoma.
By working with Aslam, the operatives “obtained prescription drugs from an authorized distributor that they would not otherwise have been permitted to purchase, and which they were able to sell at a profit,” according to the indictment.
Both the prosecuting assistant US attorney, Andrew Lievense, and Aslam’s defense lawyer, Daniel Dena, declined to comment for this news organization.
The prosecutor is seeking to recoup the more than $2.5 million Aslam allegedly pocketed, according to the indictment. The press release also noted that an “indictment is only a charge and is not evidence of guilt.”
A version of this article first appeared on Medscape.com.
In late October, a federal grand jury charged a Detroit-area medical oncologist Naveed Aslam, MD, in an indictment for his part in a scheme to illegally sell cancer drugs.
According to the indictment, Aslam acquired and sold more than $17 million in cancer drugs and personally netted more than $2.5 million during the scheme.
The charges against Aslam, filed on October 23 in the US District Court for the Eastern District of Michigan, include 10 counts of illegally selling or trading prescription drugs and one count of conspiring to do so.
“Dr. Aslam’s alleged participation in this scheme not only allowed him to profit unlawfully from the sale of cancer drugs but it also posed a serious threat by potentially placing these medications into the wrong hands,” Cheyvoryea Gibson, special agent in charge of the FBI in Michigan, said in a press release announcing the indictment.
The investigation is being conducted jointly by the FBI, the US Food and Drug Administration (FDA), the US Department of Health and Human Services Office of Inspector General, and Homeland Security Investigations.
The indictment alleges that Aslam was recruited by an unnamed operator of a Michigan corporation that engaged in business as a retail pharmacy and in the wholesale distribution of expensive prescription drugs, largely oncology drugs.
According to the indictment, Aslam and the operator came to an agreement where Aslam would purchase these expensive drugs from an authorized distributor under the false pretense that he was going to prescribe them to patients.
Instead, Aslam allegedly “sold and transferred the prescription drugs” to or through the Michigan business, with involvement from the unnamed operator and a second unnamed individual.
The unnamed individuals “identified customers interested in buying prescription cancer drugs” and “communicated with Dr. Aslam about what cancer drugs were requested,” according to the press release. “Dr. Aslam used his access to certain cancer drugs through his medical practice, Somerset Hematology and Oncology, P.C., to order and purchase the cancer drugs from his supplier.”
The indictment lays out that Aslam allegedly profited from this scheme in several ways, which included charging the Michigan business more than he paid the distributor for the drugs, sharing the profits when the business resold the drugs at a markup, and receiving rebates and discounts from the distributor “based on the amount of qualifying drugs he purchased and resold.”
According to the indictment, the scheme ran from early 2019 to mid-2023 and included four antibody drug conjugates — trastuzumab deruxtecan (Enhertu), enfortumab vedotin (Padcev), tisotumab vedotin (Tivdak), and sacituzumab govitecan (Trodelvy) — and the monoclonal antibody mogamulizumab (Poteligeo) for cutaneous T-cell lymphoma.
By working with Aslam, the operatives “obtained prescription drugs from an authorized distributor that they would not otherwise have been permitted to purchase, and which they were able to sell at a profit,” according to the indictment.
Both the prosecuting assistant US attorney, Andrew Lievense, and Aslam’s defense lawyer, Daniel Dena, declined to comment for this news organization.
The prosecutor is seeking to recoup the more than $2.5 million Aslam allegedly pocketed, according to the indictment. The press release also noted that an “indictment is only a charge and is not evidence of guilt.”
A version of this article first appeared on Medscape.com.
In late October, a federal grand jury charged a Detroit-area medical oncologist Naveed Aslam, MD, in an indictment for his part in a scheme to illegally sell cancer drugs.
According to the indictment, Aslam acquired and sold more than $17 million in cancer drugs and personally netted more than $2.5 million during the scheme.
The charges against Aslam, filed on October 23 in the US District Court for the Eastern District of Michigan, include 10 counts of illegally selling or trading prescription drugs and one count of conspiring to do so.
“Dr. Aslam’s alleged participation in this scheme not only allowed him to profit unlawfully from the sale of cancer drugs but it also posed a serious threat by potentially placing these medications into the wrong hands,” Cheyvoryea Gibson, special agent in charge of the FBI in Michigan, said in a press release announcing the indictment.
The investigation is being conducted jointly by the FBI, the US Food and Drug Administration (FDA), the US Department of Health and Human Services Office of Inspector General, and Homeland Security Investigations.
The indictment alleges that Aslam was recruited by an unnamed operator of a Michigan corporation that engaged in business as a retail pharmacy and in the wholesale distribution of expensive prescription drugs, largely oncology drugs.
According to the indictment, Aslam and the operator came to an agreement where Aslam would purchase these expensive drugs from an authorized distributor under the false pretense that he was going to prescribe them to patients.
Instead, Aslam allegedly “sold and transferred the prescription drugs” to or through the Michigan business, with involvement from the unnamed operator and a second unnamed individual.
The unnamed individuals “identified customers interested in buying prescription cancer drugs” and “communicated with Dr. Aslam about what cancer drugs were requested,” according to the press release. “Dr. Aslam used his access to certain cancer drugs through his medical practice, Somerset Hematology and Oncology, P.C., to order and purchase the cancer drugs from his supplier.”
The indictment lays out that Aslam allegedly profited from this scheme in several ways, which included charging the Michigan business more than he paid the distributor for the drugs, sharing the profits when the business resold the drugs at a markup, and receiving rebates and discounts from the distributor “based on the amount of qualifying drugs he purchased and resold.”
According to the indictment, the scheme ran from early 2019 to mid-2023 and included four antibody drug conjugates — trastuzumab deruxtecan (Enhertu), enfortumab vedotin (Padcev), tisotumab vedotin (Tivdak), and sacituzumab govitecan (Trodelvy) — and the monoclonal antibody mogamulizumab (Poteligeo) for cutaneous T-cell lymphoma.
By working with Aslam, the operatives “obtained prescription drugs from an authorized distributor that they would not otherwise have been permitted to purchase, and which they were able to sell at a profit,” according to the indictment.
Both the prosecuting assistant US attorney, Andrew Lievense, and Aslam’s defense lawyer, Daniel Dena, declined to comment for this news organization.
The prosecutor is seeking to recoup the more than $2.5 million Aslam allegedly pocketed, according to the indictment. The press release also noted that an “indictment is only a charge and is not evidence of guilt.”
A version of this article first appeared on Medscape.com.
Mycosis Fungoides: Measured Approach Key to Treatment
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
HUNTINGTON BEACH, CALIFORNIA — When patients of Aaron Mangold, MD, first learn they have mycosis fungoides (MF), the most common form of primary cutaneous T-cell lymphoma (CTCL), some are concerned about whether the diagnosis means a shortened life expectancy.
Dr. Mangold, codirector of the multidisciplinary cutaneous lymphoma clinic at Mayo Clinic, Scottsdale, Arizona, said at the annual meeting of the Pacific Dermatologic Association. “For early-stage disease, I think of it more like diabetes; this is really a chronic disease” that unlikely will be fatal but may be associated with increased morbidity as the disease progresses, and “the overall goal of therapy should be disease control to increase quality of life.”
Patient- and lymphoma-specific factors drive the choice of therapy. The focus for patients with early-stage disease, Dr. Mangold said, is to treat comorbidities and symptoms, such as itch or skin pain, maximize their quality of life, and consider the potential for associated toxicities of therapy as the disease progresses. Start with the least toxic, targeted, nonimmunosuppressive therapy, “then work toward more toxic immunosuppressive therapies,” he advised. “Use toxic agents just long enough to control the disease, then transition to a maintenance regimen with less toxic immunosuppressive agents.”
When Close Follow-Up Is Advised
According to unpublished data from PROCLIPI (the Prospective Cutaneous Lymphoma International Prognostic Index) study presented at the fifth World Congress of Cutaneous Lymphomas earlier in 2024, the following factors warrant consideration for close follow-up and more aggressive treatment: Nodal enlargement greater than 15 mm, age over 60 years, presence of plaques, and large-cell transformation in skin. “These are some of the stigmata in early disease that might guide you toward referring” a patient to a CTCL expert, Dr. Mangold said. (Consensus-based recommendations on the management of MF in children were published in August of 2024.)
According to Dr. Mangold, topical/skin-directed therapies are best for early-stage disease or in combination with systemic therapies in advanced disease. For early-stage disease, one of his preferred options is daily application of a skin moisturizer plus a topical corticosteroid such as clobetasol, halobetasol, or augmented betamethasone, then evaluating the response at 3 months. “This is a cheap option, and we see response rates as high as 90%,” he said. “I don’t often see steroid atrophy when treating patients with active MF. There’s a tendency to think, ‘I don’t want to overtreat.’ I think you can be aggressive. If you look in the literature, people typically pulse twice daily for a couple of weeks with a 1-week break.”
Mechlorethamine, a topical alkylating gel approved in 2013 for the treatment of early-stage MF, is an option when patients fail to respond to topical steroids, prefer to avoid steroids, or have thick, plaque-like disease. With mechlorethamine, it is important to “start slow and be patient,” Dr. Mangold said. “Real-world data shows that it takes 12-18 months to get a good response. Counsel patients that they are likely to get a rash, and that the risk of rash is dose dependent.”
Other treatment options to consider include imiquimod, which can be used for single refractory spots. He typically recommends application 5 days per week with titration up to daily if tolerated for up to 3 months. “Treat until you get a brisk immune response,” he said. “We’ve seen patients with durable, long-term responses.”
UVB Phototherapy Effective
For patients with stage IB disease, topical therapies are less practical and may be focused on refractory areas of disease. Narrow-band UVB phototherapy is the most practical and cost-effective treatment, Dr. Mangold said. Earlier-stage patch disease responds to phototherapy in up to 80% of cases, while plaque-stage disease responds in up to half of cases. “More frequent use of phototherapy may decrease time to clearance, but overall response is similar.”
Dr. Mangold recommends phototherapy 2-3 days per week, titrating up to a maximal response dose, and maintaining that dose for about 3 months. Maintenance involves tapering the phototherapy dose to a minimal dose with continued response. “The goal is to prevent relapse,” he said.
For patients with MF of stage IIB and higher, he considers total skin electron beam therapy, an oral retinoid with phototherapy, systemic agents, and focal radiation with systemic treatment. One of his go-to systemic options is bexarotene, which he uses for early-stage disease refractory to treatment or for less aggressive advanced disease. “We typically use a low dose ... and about half of patients respond,” Dr. Mangold said. The time to response is about 6 months. Bexarotene causes elevated lipids and low thyroid function, so he initiates patients on fenofibrate and levothyroxine at baseline.
Another systemic option is brentuximab vedotin, a monoclonal antibody that targets cells with CD30 expression, which is typically administered in a specialty center every 3 weeks for up to 16 cycles. “In practice, we often use six to eight cycles to avoid neuropathy,” he said. “It’s a good debulking agent, the time to response is 6-9 weeks, and it has a sustained response of 60%.” Neuropathy can occur with treatment, but improves over time.
Other systemic options for MF include romidepsin, mogamulizumab, and extracorporeal photopheresis used in erythrodermic disease.
Radiation An Option in Some Cases
Dr. Mangold noted that low doses of radiation therapy can effectively treat MF lesions in as little as one dose. “We can use it as a cure for a single spot or to temporarily treat the disease while other therapies are being started,” he said. Long-term side effects need to be considered when using radiation. “The more radiation, the more side effects.”
Dr. Mangold disclosed that he is an investigator for Sun Pharmaceutical, Solagenix, Elorac, miRagen, Kyowa Kirin, the National Clinical Trials Network, and CRISPR Therapeutics. He has also received consulting fees/honoraria from Kirin and Solagenix.
A version of this article first appeared on Medscape.com.
FROM PDA 2024
Popular Weight Loss Drugs Now for Patients With Cancer?
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Demand for new weight loss drugs has surged over the past few years.
Led by the antiobesity drugs semaglutide (Wegovy) and tirzepatide (Zepbound), these popular medications — more commonly known as glucagon-like peptide 1 (GLP-1) agonists — have become game changers for shedding excess pounds.
Aside from obesity indications, both drugs have been approved to treat type 2 diabetes under different brand names and have a growing list of other potential benefits, such as reducing inflammation and depression.
While there’s limited data to support the use of GLP-1 agonists for weight loss in cancer, some oncologists have begun carefully integrating the antiobesity agents into care and studying their effects in this patient population.
The reason: Research suggests that obesity can reduce the effectiveness of cancer therapies, especially in patients with breast cancer, and can increase the risk for treatment-related side effects.
The idea is that managing patients’ weight will improve their cancer outcomes, explained Lajos Pusztai, MD, PhD, a breast cancer specialist and professor of medicine at Yale School of Medicine in New Haven, Connecticut.
Although Dr. Pusztai and his oncology peers at Yale don’t yet use GPL-1 agonists, Neil Iyengar, MD, and colleagues have begun doing so to help some patients with breast cancer manage their weight. Dr. Iyengar estimates that a few hundred — almost 40% — of his patients are on the antiobesity drugs.
“For a patient who has really tried to reduce their weight and who is in the obese range, that’s where I think the use of these medications can be considered,” said Dr. Iyengar, a breast cancer oncologist at Memorial Sloan Kettering Cancer Center in New York City.
Why GLP-1s in Cancer?
GLP-1 is a hormone that the small intestine releases after eating. GLP-1 agonists work by mimicking GLP-1 to trigger the release of insulin and reduce the production of glucagon — two processes that help regulate blood sugar.
These agents, such as Wegovy (or Ozempic when prescribed for diabetes), also slow gastric emptying and can make people feel fuller longer.
Zebound (or Mounjaro for type 2 diabetes) is considered a dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, which may enhance its weight loss benefits.
In practice, however, these drugs can increase nausea and vomiting from chemotherapy, so Dr. Iyengar typically has patients use them afterwards, during maintenance treatment.
Oncologists don’t prescribe the drugs themselves but instead refer patients to endocrinologists or weight management centers that then write the prescriptions. Taking these drugs involves weekly subcutaneous injections patients can administer themselves.
Endocrinologist Emily Gallagher, MD, PhD, of Mount Sinai Hospital in New York City, estimates she has prescribed the antiobesity drugs to a few hundred patients with cancer and, like Dr. Iyengar, uses the drugs during maintenance treatment with hormone therapy for breast cancer. She also has used these agents in patients with prostate and endometrial cancers and has found the drugs can help counter steroid weight gain in multiple myeloma.
But, to date, the evidence for using GPL-1 agonists in cancer remains limited and the practice has not yet become widespread.
Research largely comes down to a few small retrospective studies in patients with breast cancer receiving aromatase inhibitors. Although no safety issues have emerged so far, these initial reports suggest that the drugs lead to significantly less weight loss in patients with cancer compared to the general population.
Dr. Iyengar led one recent study, presented at the 2024 annual meeting of the American Society of Clinical Oncology, in which he and his team assessed outcomes in 75 women with breast cancer who received a GLP-1 agonist. Almost 80% of patients had diabetes, and 60% received hormone therapy, most commonly an aromatase inhibitor. Patients’ median body mass index (BMI) at baseline was 34 kg/m2 (range, 23-50 kg/m2).
From baseline, patients lost 6.2 kg, on average, or about 5% of their total body weight, 12 months after initiating GLP-1 therapy.
In contrast, phase 3 trials show much higher mean weight loss — about two times — in patients without cancer.
Another recent study also reported modest weight loss results in patients with breast cancer undergoing endocrine therapy. The researchers reported that, at 12 months, Wegovy led to 4.34% reduction in BMI, compared with a 14% change reported in the general population. Zebound, however, was associated with a 2.31% BMI increase overall — though some patients did experience a decrease — compared with a 15% reduction in the general population.
“These findings indicate a substantially reduced weight loss efficacy in breast cancer patients on endocrine therapy compared to the general population,” the authors concluded.
It’s unclear why the drugs appear to not work as well in patients with cancer. It’s possible that hormone therapy or metabolic changes interfere with their effectiveness, given that some cancer therapies lead to weight gain. Steroids and hormone therapies, for instance, often increase appetite, and some treatments can slow patients’ metabolism or lead to fatigue, which can make it harder to exercise.
Patients with cancer may need a higher dose of GLP-1 agonists to achieve similar weight loss to the general population, Dr. Iyengar noted.
However, Dr. Gallagher said, in her own experience, she hasn’t found the drugs to be less effective in patients with cancer, especially the newer agents, like Wegovy and Zepbound.
As for safety, Wegovy and Zepbound both carry a black box warning for thyroid C-cell tumors, including medullary thyroid carcinoma. (Recent research, however, has found that GLP-1 agonists do not increase thyroid cancer risk).
These antiobesity agents are also contraindicated in patients with a personal or family history of medullary thyroid carcinoma and in patients who have multiple endocrine neoplasia syndrome type 2, which is associated with medullary thyroid carcinoma.
Dr. Gallagher hasn’t seen any secondary tumors — thyroid or otherwise — in her patients with cancer, but she follows the labeling contraindications. Dr. Iyengar also noted that more recent and larger data sets have shown no impact on this risk, which may not actually exist, he said
Dr. Gallagher remains cautious about using GPL-1 agonists in patients who have had bariatric surgery because these agents can compound the slower gastric emptying and intestinal transit from surgery, potentially leading to gastrointestinal obstructions.
Looking ahead, GPL-1 manufacturers are interested in adding cancer indications to the drug labeling. Both Dr. Iyengar and Dr. Gallagher said their institutions are in talks with companies to participate in large, multicenter, global phase 3 trials.
Dr. Iyengar welcomes the efforts, not only to test the effectiveness of GPL-1 agonists in oncology but also to “nail down” their safety in cancer.
“I don’t think that there’s mechanistically anything that’s particularly worrisome,” and current observations suggest that these drugs are likely to be safe, Dr. Iyengar said. Even so, “GLP-1 agonists do a lot of things that we don’t fully understand yet.”
The bigger challenge, Dr. Iyengar noted, is that companies will have to show a sizable benefit to using these drugs in patients with cancer to get the Food and Drug Administration’s approval. And to move the needle on cancer-specific outcomes, these antiobesity drugs will need to demonstrate significant, durable weight loss in patients with cancer.
But if these drugs can do that, “I think it’s going to be one of the biggest advances in medicine and oncology given the obesity and cancer epidemic,” Dr. Iyengar said.
Dr. Iyengar has adviser and/or researcher ties with companies that make or are developing GPL-1 agonists, including AstraZeneca, Novartis, Gilead, and Pfizer. Dr. Gallagher is a consultant for Novartis, Flare Therapeutics, Reactive Biosciences, and Seagen.
A version of this article first appeared on Medscape.com.
Does Medicare Advantage Offer Higher-Value Chemotherapy?
TOPLINE:
METHODOLOGY:
- Private Medicare Advantage plans enroll more than half of the Medicare population, but it is unknown if or how the cost restrictions they impose affect chemotherapy, which accounts for a large portion of cancer care costs.
- Researchers conducted a cohort study using national Medicare data from January 2015 to December 2019 to look at Medicare Advantage enrollment and treatment patterns for patients with cancer receiving chemotherapy.
- The study included 96,501 Medicare Advantage enrollees and 206,274 traditional Medicare beneficiaries who initiated chemotherapy between January 2016 and December 2019 (mean age, ~73 years; ~56% women; Hispanic individuals, 15% and 8%; Black individuals, 15% and 8%; and White individuals, 75% and 86%, respectively).
- Resource use and care quality were measured during a 6-month period following chemotherapy initiation, and survival days were measured 18 months after beginning chemotherapy.
- Resource use measures included hospital inpatient services, outpatient care, prescription drugs, hospice services, and chemotherapy services. Quality measures included chemotherapy-related emergency visits and hospital admissions, as well as avoidable emergency visits and preventable hospitalizations.
TAKEAWAY:
- Medicare Advantage plans had lower resource use than traditional Medicare per enrollee with cancer undergoing chemotherapy ($8718 lower; 95% CI, $8343-$9094).
- The lower resource use was largely caused by fewer chemotherapy visits and less expensive chemotherapy per visit in Medicare Advantage plans ($5032 lower; 95% CI, $4772-$5293).
- Medicare Advantage enrollees had 2.5 percentage points fewer chemotherapy-related emergency department visits and 0.7 percentage points fewer chemotherapy-related hospitalizations than traditional Medicare beneficiaries.
- There was no clinically meaningful difference in survival between Medicare Advantage and traditional Medicare beneficiaries during the 18 months following chemotherapy initiation.
IN PRACTICE:
“Our new finding is that MA [Medicare Advantage] plans had lower resource use than TM [traditional Medicare] among enrollees with cancer undergoing chemotherapy — a serious condition managed by specialists and requiring expensive treatments. This suggests that MA’s cost advantages over TM are not limited to conditions for which low-cost primary care management can avoid costly services,” the authors wrote.
SOURCE:
The study was led by Yamini Kalidindi, PhD, McDermott+ Consulting, Washington, DC. It was published online on September 20, 2024, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2024.34707), with a commentary.
LIMITATIONS:
The study’s findings may be affected by unobserved patient characteristics despite the use of inverse-probability weighting. The exclusion of Medicare Advantage enrollees in contracts with incomplete encounter data limits the generalizability of the results. The study does not apply to beneficiaries without Part D drug coverage. Quality measures were limited to those available from claims and encounter data, lacking information on patients’ cancer stage. The 18-month measure of survival might not adequately capture survival differences associated with early-stage cancers. The study did not measure whether patient care followed recommended guidelines.
DISCLOSURES:
Various authors reported grants from the National Institute on Aging, the National Institutes of Health, The Commonwealth Fund, Arnold Ventures, the National Cancer Institute, the Department of Defense, and the National Institute of Health Care Management. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Private Medicare Advantage plans enroll more than half of the Medicare population, but it is unknown if or how the cost restrictions they impose affect chemotherapy, which accounts for a large portion of cancer care costs.
- Researchers conducted a cohort study using national Medicare data from January 2015 to December 2019 to look at Medicare Advantage enrollment and treatment patterns for patients with cancer receiving chemotherapy.
- The study included 96,501 Medicare Advantage enrollees and 206,274 traditional Medicare beneficiaries who initiated chemotherapy between January 2016 and December 2019 (mean age, ~73 years; ~56% women; Hispanic individuals, 15% and 8%; Black individuals, 15% and 8%; and White individuals, 75% and 86%, respectively).
- Resource use and care quality were measured during a 6-month period following chemotherapy initiation, and survival days were measured 18 months after beginning chemotherapy.
- Resource use measures included hospital inpatient services, outpatient care, prescription drugs, hospice services, and chemotherapy services. Quality measures included chemotherapy-related emergency visits and hospital admissions, as well as avoidable emergency visits and preventable hospitalizations.
TAKEAWAY:
- Medicare Advantage plans had lower resource use than traditional Medicare per enrollee with cancer undergoing chemotherapy ($8718 lower; 95% CI, $8343-$9094).
- The lower resource use was largely caused by fewer chemotherapy visits and less expensive chemotherapy per visit in Medicare Advantage plans ($5032 lower; 95% CI, $4772-$5293).
- Medicare Advantage enrollees had 2.5 percentage points fewer chemotherapy-related emergency department visits and 0.7 percentage points fewer chemotherapy-related hospitalizations than traditional Medicare beneficiaries.
- There was no clinically meaningful difference in survival between Medicare Advantage and traditional Medicare beneficiaries during the 18 months following chemotherapy initiation.
IN PRACTICE:
“Our new finding is that MA [Medicare Advantage] plans had lower resource use than TM [traditional Medicare] among enrollees with cancer undergoing chemotherapy — a serious condition managed by specialists and requiring expensive treatments. This suggests that MA’s cost advantages over TM are not limited to conditions for which low-cost primary care management can avoid costly services,” the authors wrote.
SOURCE:
The study was led by Yamini Kalidindi, PhD, McDermott+ Consulting, Washington, DC. It was published online on September 20, 2024, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2024.34707), with a commentary.
LIMITATIONS:
The study’s findings may be affected by unobserved patient characteristics despite the use of inverse-probability weighting. The exclusion of Medicare Advantage enrollees in contracts with incomplete encounter data limits the generalizability of the results. The study does not apply to beneficiaries without Part D drug coverage. Quality measures were limited to those available from claims and encounter data, lacking information on patients’ cancer stage. The 18-month measure of survival might not adequately capture survival differences associated with early-stage cancers. The study did not measure whether patient care followed recommended guidelines.
DISCLOSURES:
Various authors reported grants from the National Institute on Aging, the National Institutes of Health, The Commonwealth Fund, Arnold Ventures, the National Cancer Institute, the Department of Defense, and the National Institute of Health Care Management. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Private Medicare Advantage plans enroll more than half of the Medicare population, but it is unknown if or how the cost restrictions they impose affect chemotherapy, which accounts for a large portion of cancer care costs.
- Researchers conducted a cohort study using national Medicare data from January 2015 to December 2019 to look at Medicare Advantage enrollment and treatment patterns for patients with cancer receiving chemotherapy.
- The study included 96,501 Medicare Advantage enrollees and 206,274 traditional Medicare beneficiaries who initiated chemotherapy between January 2016 and December 2019 (mean age, ~73 years; ~56% women; Hispanic individuals, 15% and 8%; Black individuals, 15% and 8%; and White individuals, 75% and 86%, respectively).
- Resource use and care quality were measured during a 6-month period following chemotherapy initiation, and survival days were measured 18 months after beginning chemotherapy.
- Resource use measures included hospital inpatient services, outpatient care, prescription drugs, hospice services, and chemotherapy services. Quality measures included chemotherapy-related emergency visits and hospital admissions, as well as avoidable emergency visits and preventable hospitalizations.
TAKEAWAY:
- Medicare Advantage plans had lower resource use than traditional Medicare per enrollee with cancer undergoing chemotherapy ($8718 lower; 95% CI, $8343-$9094).
- The lower resource use was largely caused by fewer chemotherapy visits and less expensive chemotherapy per visit in Medicare Advantage plans ($5032 lower; 95% CI, $4772-$5293).
- Medicare Advantage enrollees had 2.5 percentage points fewer chemotherapy-related emergency department visits and 0.7 percentage points fewer chemotherapy-related hospitalizations than traditional Medicare beneficiaries.
- There was no clinically meaningful difference in survival between Medicare Advantage and traditional Medicare beneficiaries during the 18 months following chemotherapy initiation.
IN PRACTICE:
“Our new finding is that MA [Medicare Advantage] plans had lower resource use than TM [traditional Medicare] among enrollees with cancer undergoing chemotherapy — a serious condition managed by specialists and requiring expensive treatments. This suggests that MA’s cost advantages over TM are not limited to conditions for which low-cost primary care management can avoid costly services,” the authors wrote.
SOURCE:
The study was led by Yamini Kalidindi, PhD, McDermott+ Consulting, Washington, DC. It was published online on September 20, 2024, in JAMA Network Open (doi: 10.1001/jamanetworkopen.2024.34707), with a commentary.
LIMITATIONS:
The study’s findings may be affected by unobserved patient characteristics despite the use of inverse-probability weighting. The exclusion of Medicare Advantage enrollees in contracts with incomplete encounter data limits the generalizability of the results. The study does not apply to beneficiaries without Part D drug coverage. Quality measures were limited to those available from claims and encounter data, lacking information on patients’ cancer stage. The 18-month measure of survival might not adequately capture survival differences associated with early-stage cancers. The study did not measure whether patient care followed recommended guidelines.
DISCLOSURES:
Various authors reported grants from the National Institute on Aging, the National Institutes of Health, The Commonwealth Fund, Arnold Ventures, the National Cancer Institute, the Department of Defense, and the National Institute of Health Care Management. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.




