User login
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).
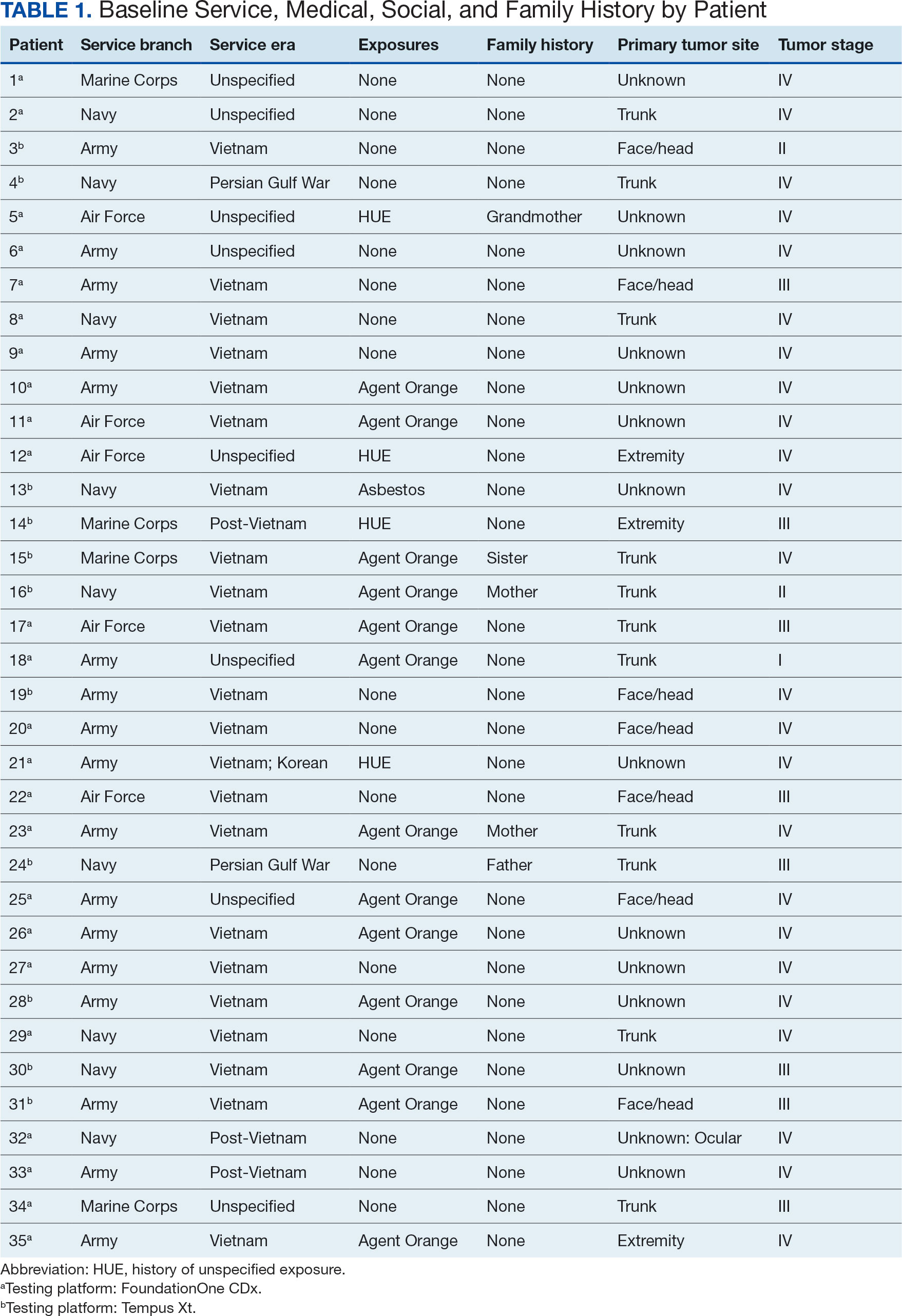
Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
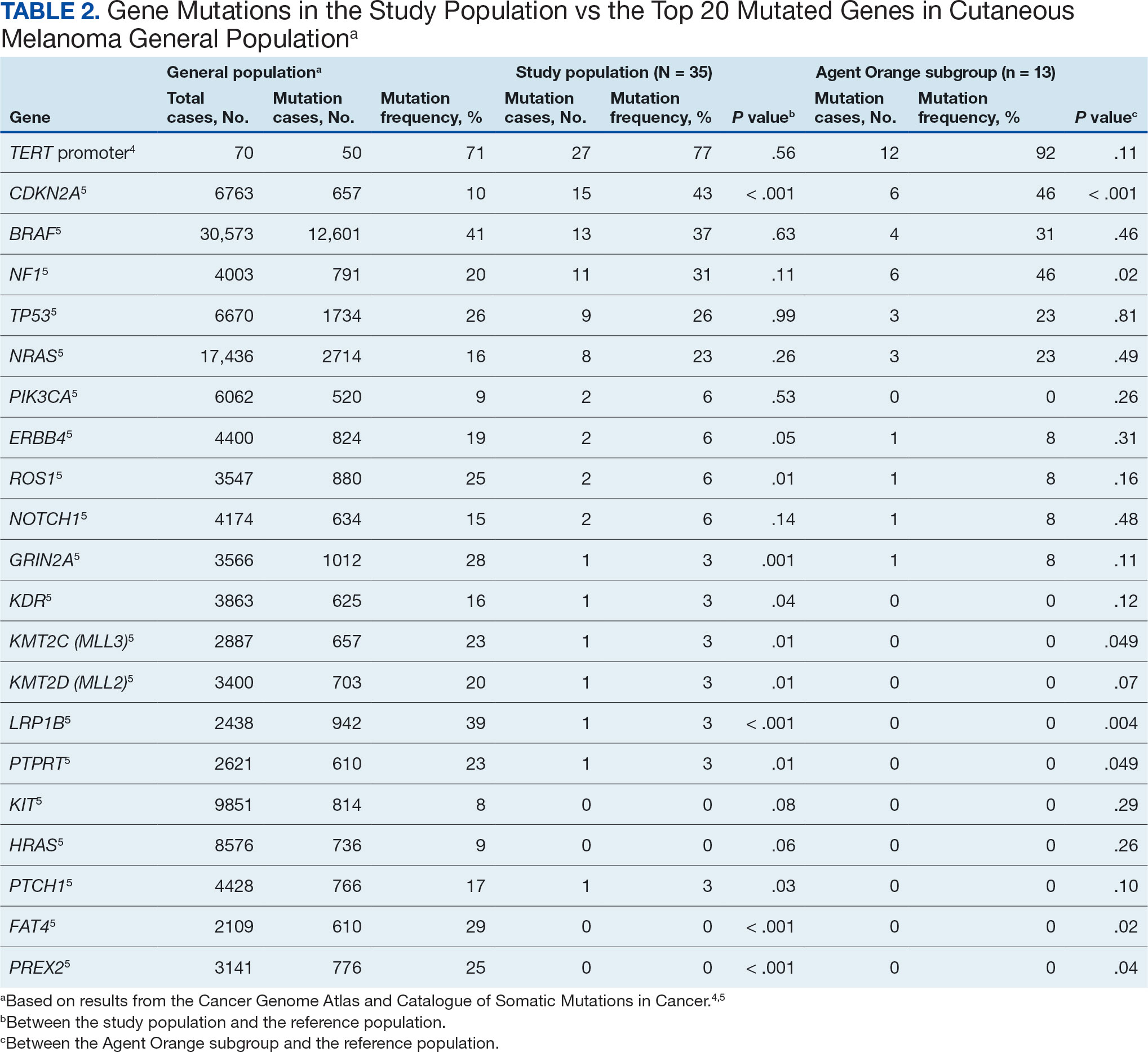
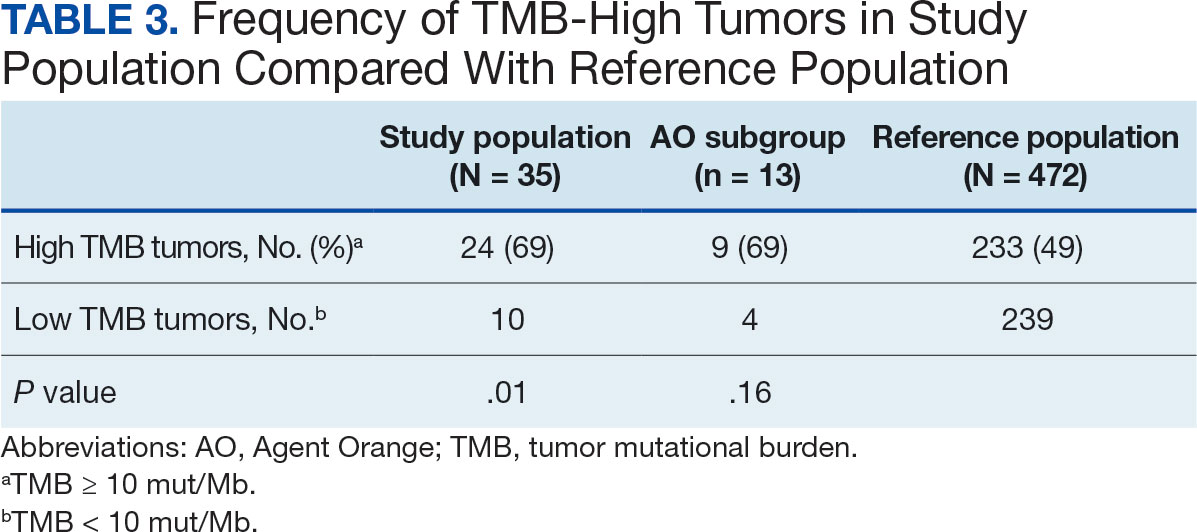
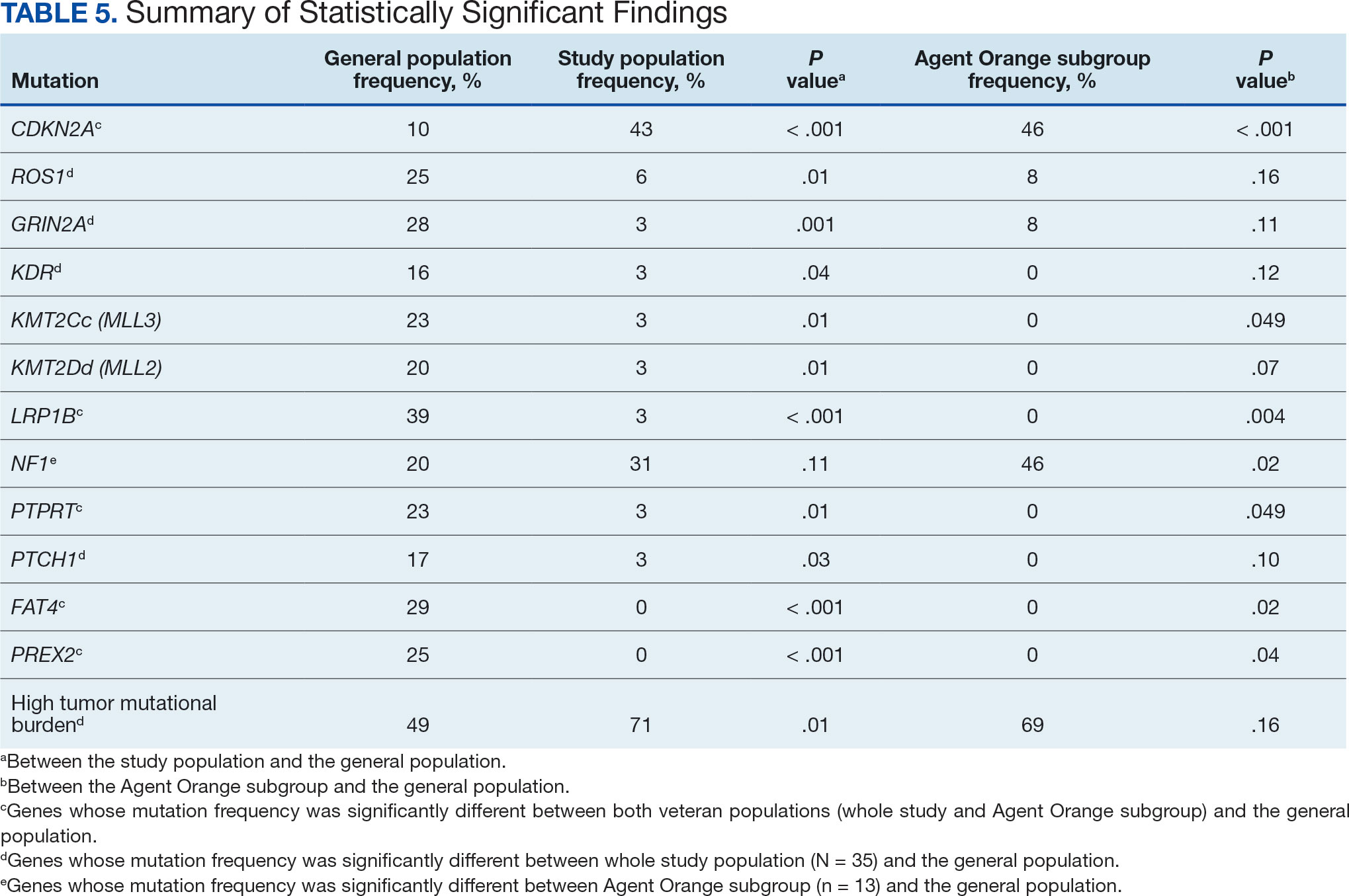
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.

Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).

Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.




Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
The veteran population, with its unique and diverse types of exposure and military service experiences, faces distinct health factors compared with the general population. These factors can be categorized into exposures during military service and those occurring postservice. While the latter phase incorporates psychological issues that may arise while transitioning to civilian life, the service period is associated with major physical, chemical, and psychological exposures that can impact veterans’ health. Carcinogenesis related to military exposures is concerning, and different types of malignancies have been associated with military exposures.1 The 2022 introduction of the Cancer Moonshot initiative served as a breeding ground for multiple projects aimed at investigation of exposure-related carcinogenesis, prompting increased attention and efforts to linking specific exposures to specific malignancies.2
Melanoma is the deadliest skin cancer, accounting for 1.3% of all cancer deaths.3 Although it may only account for 1% to 5% of skin cancer diagnoses, its incidence in the United States’ population has been increasing.4,5 There were 97,610 estimated new cases of melanoma in 2023, according to the National Cancer Institute.6
The incidence of melanoma may be higher in the military population compared with the general population.7 Melanoma is the fourth-most common cancer diagnosed in veterans.8
Several demographic characteristics of the US military population are associated with higher melanoma incidence and poorer prognosis, including male sex, older age, and White race. Apart from sun exposure—a known risk factor for melanoma development—other factors, such as service branch, seem to contribute to risk, with the highest melanoma rates noted in the Air Force.9 According to a study by Chang et al, veterans have a higher risk of stage III (18%) or stage IV (13%) melanoma at initial diagnosis.8
Molecular testing of metastatic melanoma is currently the standard of care for guiding the use of US Food and Drug Administration-approved targeted therapies such as BRAF, MEK, and KIT inhibitors. This comparative analysis details the melanoma comprehensive genomic profiles observed at a large US Department of Veterans Affairs (VA) medical center (VAMC) and those reported in reference databases.
Methods
A query to select all metastatic melanomas sent for comprehensive genomic profiling from the Kansas City VAMC (KCVAMC), identified 35 cases from 2019 through 2023 as the study population. The health records of these patients were reviewed to collect demographic information, military service history, melanoma history, other medical, social, and family histories. The comprehensive genomic profiling reports were reviewed to collect the reported pathogenic variants, microsatellite instability (MSI) status, and tumor mutational burden (TMB) for each case.
The Catalogue of Somatic Mutations in Cancer (COSMIC) was used to identify the most commonly mutated genes in melanomas from The Cancer Genome Atlas for the general population.4,5 The literature was consulted to determine the MSI status and TMB in melanomas from The Cancer Genome Atlas for separate reference populations.6,7 The frequency of MSI-high (MSI-H) status, TMB ≥ 10 mutations/megabase (mut/Mb), and mutations in each of the 20 most commonly mutated genes was determined and compared between melanomas from The Cancer Genome Atlas and KCVAMC cases. Corresponding P values were calculated to identify significant differences. Values were calculated for the entire sample as well as a subgroup with Agent Orange (AO) exposure. The study was approved by the KCVAMC Institutional Review Board.
Results
The mean (SD) age of study participants was 72.9 (9.4) years (range, 39-90 years). The mean (SD) duration of military service was 1654 (1421) days (about 4 years, 6 months, and 10 days). Of the 35 patients included, 22 (63%) served during the Vietnam era (November 1, 1965, to April 30, 1975) and 2 (6%) served during the Persian Gulf War era (August 2, 1990, to February 28, 1991). Seventeen veterans (49%) served in the Army, 9 in the Navy (26%), 5 in the Air Force (14%), and 4 in the Marine Corps (11%). Definitive AO exposure was noted in 13 patients (37%) (Table 1).

Of the 35 patients, 24 (69%) had metastatic disease and the primary site of melanoma was unknown in 14 patients (40%). One patient (Patient 32) had an intraocular melanoma. The primary site was the trunk for 11 patients (31%), the face/head for 7 patients (20%) and extremities for 3 patients (9%). Eight patients (23%) were pT3 stage (thickness > 2 mm but < 4 mm), 7 patients (20%) were pT4 stage (thickness > 4 mm), and 5 patients (14%) were pT1 (thickness ≥ 1 mm). One patient had a primary lesion at pT2 stage, and 1 had a Tis stage lesion. Three patients (9%) had a family history of melanoma in a first-degree relative.
The list of genes mutated in melanoma cells in the study population is provided in the eAppendix.10,11 Twenty-seven patients (77%) had mutations in TERT promoter, 15 (43%) in CDKN2A/B, 13 (37%) in BRAF, 11 (31%) in NF1, 9 (26%) in TP53, and 8 (23%) in NRAS (Table 2). The majority of mutations in TERT promoter were c.- 146C>T (18 of 27 patients [67%]), whereas c.-124C>T was the second-most common (8 of 27 patients [30%]). The 2 observed mutations in the 13 patients with BRAF mutations were V600E and V600K, with almost equal distribution (54% and 46%, respectively). The mean (SD) TMB was 33.2 (39) mut/Mb (range, 1-203 mut/Mb). Ten patients (29%) had a TMB < 10 mut/Mb, whereas 24 (69%) had a TMB > 10 mut/Mb. The TMB could not be determined in 1 case. The frequency of TMB-high tumors in the study population compared with frequency in the reference population is shown in Table 3.12 Only 3 patients (0.64%) in the reference population had MSI-H tumors, and the microsatellite status could not be determined in those tumors (Table 4).13 Table 5 outlines statistically significant findings.




Agent Orange Subgroup
AO was a tactical herbicide used by the US military, named for the orange band around the storage barrels. Possible mutagenic properties of AO have been attributed to its byproduct, dioxin. Among the most common cancers known to be associated with AO exposure are bladder and prostate carcinoma and hematopoietic neoplasms. The association between genetic alterations and AO exposure was studied in veterans with prostate cancer.14 However, to our knowledge, insufficient information is available to determine whether an association exists between exposure to herbicides used in Vietnam or the contaminant dioxin and melanoma. Because a significant proportion of this study population had a well-documented history of AO exposure (37.1%), we were able to analyze them as a subgroup and to separately compare their mutation frequency with the general population.
Results were notable for different distributions of the most frequently mutated genes in the AO subgroup compared with the whole study population. As such, TERT promoter remained the most frequently mutated gene (92%), followed by CDKN2A/B (46%); however, frequency of mutations in NF1 (46%) outnumbered those of BRAF (31%), the fourth-most common mutation. Moreover, when compared with the general melanoma population, a significantly higher frequency of mutations in the NF1 gene was observed in the AO subgroup—not the entire study population.
Discussion
Given that veterans constitute a distinct population, there is reasonable interest in investigating characteristic health issues related to military service. Skin cancer—melanoma in particular—has been researched recently in a veteran population. The differences in demographics, tumor characteristics, and melanoma- specific survival in veterans compared with the general population have already been assessed. According to Chang et al, compared with the general population, veterans are more likely to present with metastatic disease and have lower 5-year survival rates.8
Melanoma is one of the most highly mutated malignancies.15 Fortunately, the most common mutation in melanoma, BRAF V600E, is now considered therapeutically targetable. However, there are still many mutations that are less often discussed and not well understood. Regardless of therapeutic implications, all mutations observed in melanoma are worth investigating because a tumor’s genomic profile also can provide prognostic and etiologic information. Developing comprehensive descriptions of melanoma mutational profiles in specific populations is critical to advancing etiologic understanding and informing prevention strategies.
Our results demonstrate the high prevalence of TERT promoter mutations with characteristic ultraviolet signature (C>T) in the study population. This aligns with general evidence that TERT promoter mutations are common in cutaneous melanomas: 77% of this study sample and up to 86% of all mutations are TERT promoter mutations, according to Davis et al.15 TERT promoter mutations are positively associated with the initiation, invasion, and metastasis of melanoma. In certain subtypes, there is evidence that the presence of TERT promoter mutations is significantly associated with risk for extranodal metastasis and death.16 The second-most common mutated gene in the veteran study population was CDKN2A/B (43%), and the third-most mutated gene was BRAF (37%).
In chronically sun-exposed skin NF1, NRAS, and occasionally BRAF V600K mutations tend to predominate. BRAF V600E mutations, on the other hand, are rare in these melanomas.15 In our study population, the most prevalent melanoma site was the trunk (31%), which is considered a location with an intermittent pattern of sun exposure.17
This study population also had a higher frequency of CDKN2A/B mutations. High frequencies of CDKN2A/B mutations have been reported in familial melanomas, but only 1 patient with CDKN2A/B mutations had a known family history of melanoma.15 Tumors in the study population showed significantly lower frequency of mutations in ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 (P < .05).
In this study the subgroup of veterans with AO exposure differed from the whole study population. As such, CDKN2A/B mutations were observed with the same frequency as NF1 mutations (46% each); however, BRAF mutations constituted only 31% of the mutations. In addition, the frequency of NF1 mutations was significantly higher in the AO subgroup compared with the general population, but not in the whole study population.
Our sample also differed from the reference population by showing a significantly higher frequency of TMB-high (ie, ≥ 10 mut/Mb) tumors (71% vs 49%; P = .01).12 Interestingly, no significant difference in the frequency of TMB-high tumors was observed between the AO subgroup and the reference population (69% vs 49%; P = .16). There also was no statistically significant difference between the frequency of MSI-H tumors in our study population and the reference population (P = .64).13
One patient in the study population had uveal melanoma. Mutations encountered in this patient’s tumor differed from the general mutational profile of tumors. None of the 21 mutations depicted in Table 2 were present in this sample.10,11 On the other hand, those mutations frequently observed in intraocular melanomas, BAP1 and GNA11, were present in this patient.18 Additionally, this particular melanoma possessed mutations in genes RICTOR, RAD21, and PIK3R1.
Limitations
This study population consisted exclusively of male patients, introducing sex as a potential confounder in analyzing differences between the study population and the general population. As noted in a 2020 systematic review, there were no sex-based differences in the frequency of mutations in BRAF, NRAS, and KIT genes.19
Regarding NF1 mutations, only NF1-mutated acral and mucosal melanomas were more frequently observed in female patients, whereas nonacral NF1-mutated melanomas were more frequently observed in male patients.20 However, there is currently no clear evidence of whether the mutational landscapes of cutaneous melanoma differ by sex.21 Among the 11 cases with NF1-mutatation, site of origin was known in 6, 5 of which originated at nonacral sites. Although the AO subgroup also consisted entirely of male patients, this does not explain the observed increased frequency of NF1 mutations relative to the general population. No such difference was observed between the whole study population, which also consisted exclusively of male patients, and the general population. The similar frequencies of nonacral location in the whole study population (3 acral, 18 nonacral, 14 unknown site of origin) and AO subgroup (1 acral, 7 nonacral, 5 unknown site of origin) preclude location as an explanation.
The Cancer Genome Atlas Network proposed a framework for genomic classification of melanoma into 4 subtypes based on the pattern of the most prevalent significantly mutated genes: mutant BRAF, mutant RAS, mutant NF1, and triple–wild-type. According to that study, BRAF mutations were indeed associated with younger age, in contrast to the NF1-mutant genomic subtype, which was more prevalent in older individuals with higher TMB.22 This emphasizes the need to interpret the potential association of AO exposure and NF1 mutation in melanoma with caution, although additional studies are required to observe the difference between the veteran population and age-matched general population.
On the other hand, Yu et al reported no significant differences of TMB values between patients aged < 60 and ≥ 60 years with melanoma.23 In short, the observed differences we report in our limited study warrant additional investigation with larger sample sizes, sex-matched controlling, and age-matched controlling. The study was limited by its small sample size and the single location.
Conclusion
The genomic profile of melanomas in the veteran population appears to be similar to that of the general population with a few possible differences. Melanomas in the veteran study population showed a higher frequency of CDKN2A/B mutations; lower frequency of ROS1, GRIN2A, KDR, KMT2C (MLL3), KMT2D (MLL2), LRP1B, PTPRT, PTCH1, FAT4, and PREX2 mutations; and higher TMB. In addition, melanomas in the AO subgroup showed higher frequencies of NF1 mutations. The significance of such findings remains to be determined by further investigation.
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
- Bytnar JA, McGlynn KA, et al. Cancer incidence in the US military: An updated analysis. Cancer. 2024;130(1):96-106. doi:10.1002/cncr.34978
- Singer DS. A new phase of the Cancer Moonshot to end cancer as we know it. Nat Med. 2022;28(7):1345-1347. doi:10.1038/s41591-022-01881-5
- Koczkodaj P, Sulkowska U, Didkowska J, et al. Melanoma mortality trends in 28 European countries: a retrospective analysis for the years 1960-2020. Cancers (Basel). 2023;15(5):1514. Published 2023 Feb 28. doi:10.3390/cancers15051514
- Okobi OE, Abreo E, Sams NP, et al. Trends in melanoma incidence, prevalence, stage at diagnosis, and survival: an analysis of the United States Cancer Statistics (USCS) database. Cureus. 2024;16(10):e70697. doi:10.7759/cureus.70697
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:e2034-e2039. doi:10.7205/MILMED-D-17-00127
- American Cancer Society. Cancer facts & figures 2023. American Cancer Society; 2023. Accessed June 20, 2025. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
- Rezaei SJ, Kim J, Onyeka S, et al. Skin cancer and other dermatologic conditions among US veterans. JAMA Dermatol. 2024;160(10):1107-1111. doi:10.1001/jamadermatol.2024.3043
- Chang MS, La J, Trepanowski N, et al. Increased relative proportions of advanced melanoma among veterans: a comparative analysis with the Surveillance, Epidemiology, and End Results registry. J Am Acad Dermatol. 2022;87:72-79. doi:10.1016/j.jaad.2022.02.063
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Huang FW, Hodis E, Xu MJ, et al. Highly recurrent TERT promoter mutations in human melanoma. Science. 2013;339:957-959. doi:10.1126/science.1229259
- Tate JG, Bamford S, Jubb HC, et al. COSMIC: the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2019;47:D941-D947. doi:10.1093/nar/gky1015
- Li M, Gao X, Wang X. Identification of tumor mutation burden-associated molecular and clinical features in cancer by analyzing multi-omics data. Front Immunol. 2023;14:1090838. doi:10.3389/fimmu.2023.1090838
- Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017:PO.17.00073. doi:10.1200/PO.17.00073
- Lui AJ, Pagadala MS, Zhong AY, et al. Agent Orange exposure and prostate cancer risk in the Million Veteran Program. medRxiv [Preprint]. 2023:2023.06.14.23291413. doi:10.1101/2023.06.14.23291413
- Davis EJ, Johnson DB, Sosman JA, et al. Melanoma: what do all the mutations mean? Cancer. 2018;124:3490-3499. doi:10.1002/cncr.31345
- Guo Y, Chen Y, Zhang L, et al. TERT promoter mutations and telomerase in melanoma. J Oncol. 2022;2022:6300329. doi:10.1155/2022/6300329
- Whiteman DC, Stickley M, Watt P, et al. Anatomic site, sun exposure, and risk of cutaneous melanoma. J Clin Oncol. 2006;24:3172-3177. doi:10.1200/JCO.2006.06.1325
- Decatur CL, Ong E, Garg N, et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 2016;134:728-733. doi:10.1001/jamaophthalmol.2016.0903
- Gutiérrez-Castañeda LD, Nova JA, Tovar-Parra JD. Frequency of mutations in BRAF, NRAS, and KIT in different populations and histological subtypes of melanoma: a systemic review. Melanoma Res. 2020;30:62- 70. doi:10.1097/CMR.0000000000000628
- Thielmann CM, Chorti E, Matull J, et al. NF1-mutated melanomas reveal distinct clinical characteristics depending on tumour origin and respond favourably to immune checkpoint inhibitors. Eur J Cancer. 2021;159:113-124. doi:10.1016/j.ejca.2021.09.035
- D’Ecclesiis O, Caini S, Martinoli C, et al. Gender-dependent specificities in cutaneous melanoma predisposition, risk factors, somatic mutations, prognostic and predictive factors: a systematic review. Int J Environ Res Public Health. 2021;18:7945. doi:10.3390/ijerph18157945
- Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell. 2015;161:1681-1696. doi:10.1016/j.cell.2015.05.044
- Yu Z, Wang J, Feng L, et al. Association of tumor mutational burden with age in solid tumors. J Clin Oncol. 2020;38:e13590-e13590. doi:10.1200/JCO.2020.38.15_suppl.e13590
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
Comprehensive Genomic Profiles of Melanoma in Veterans Compared to Reference Databases
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
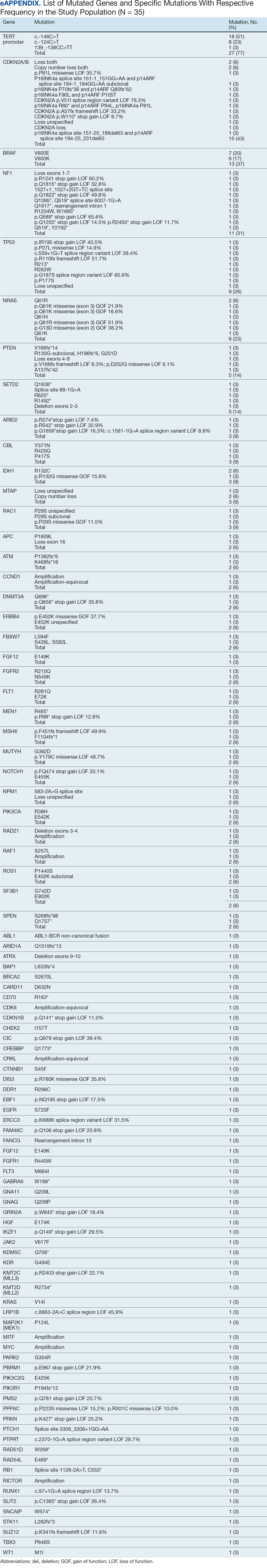
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).
The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.
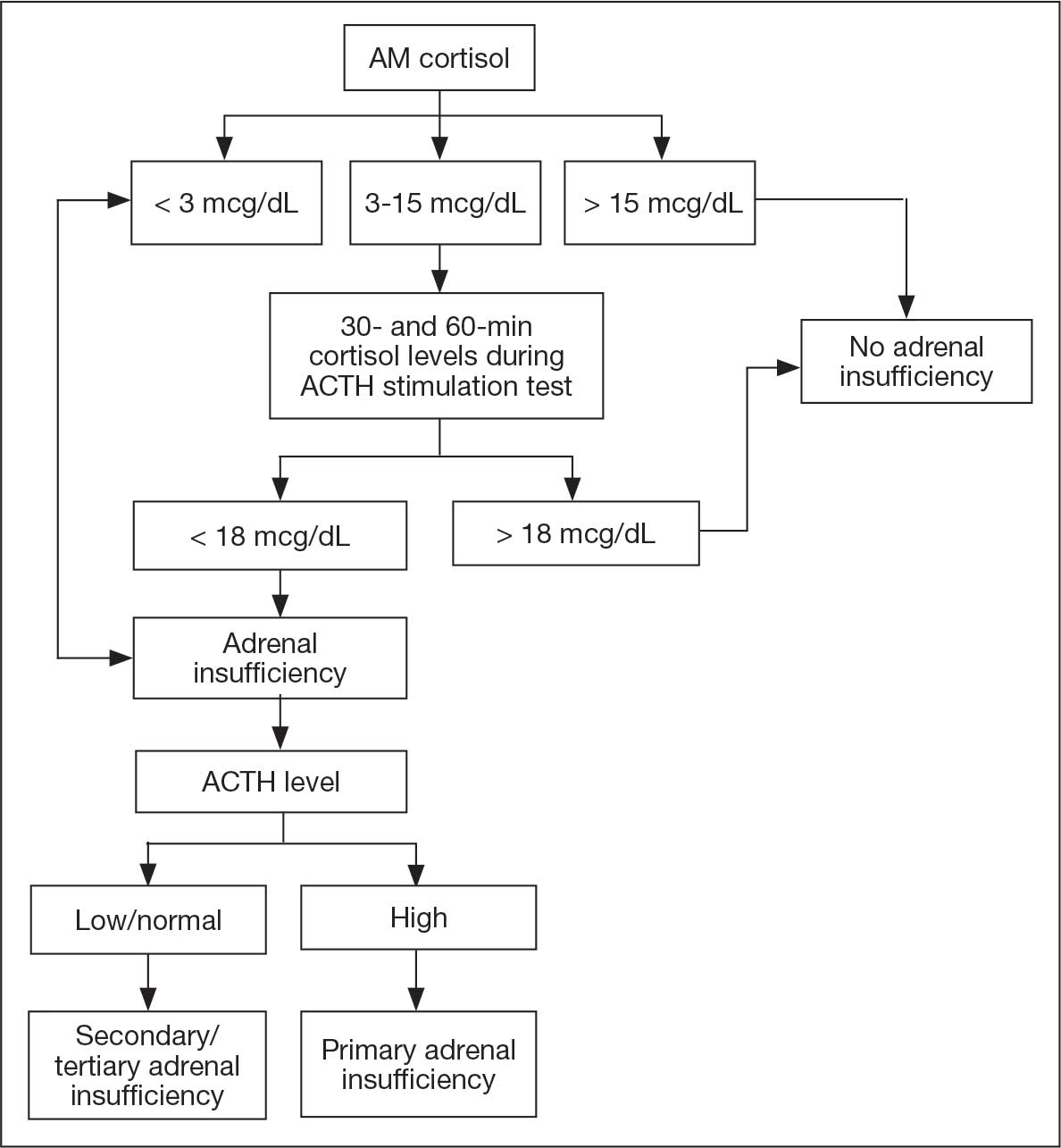
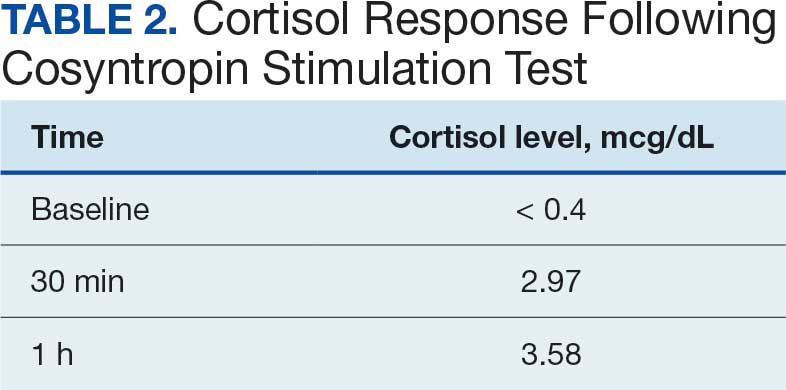
One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).
The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).
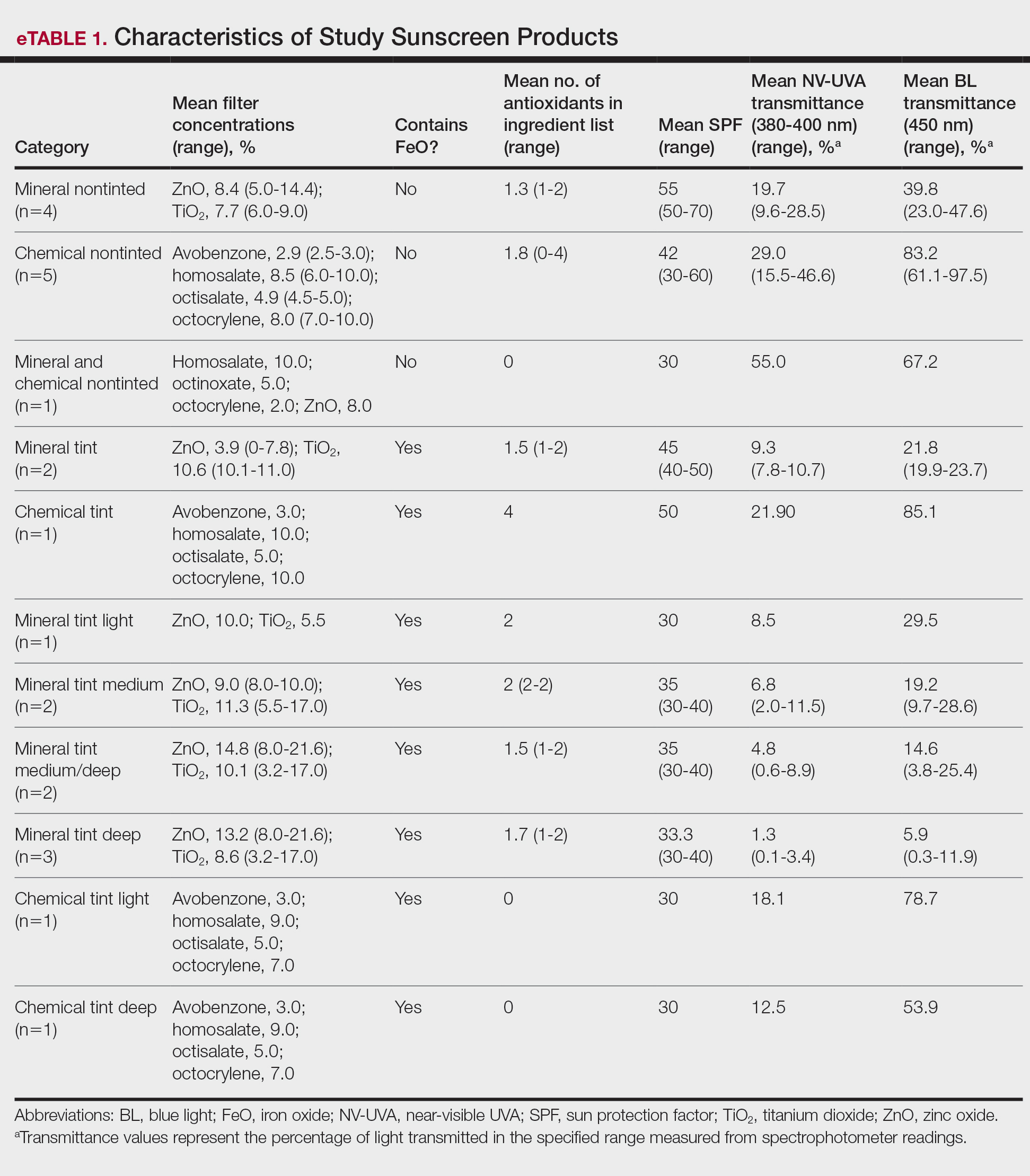
The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).
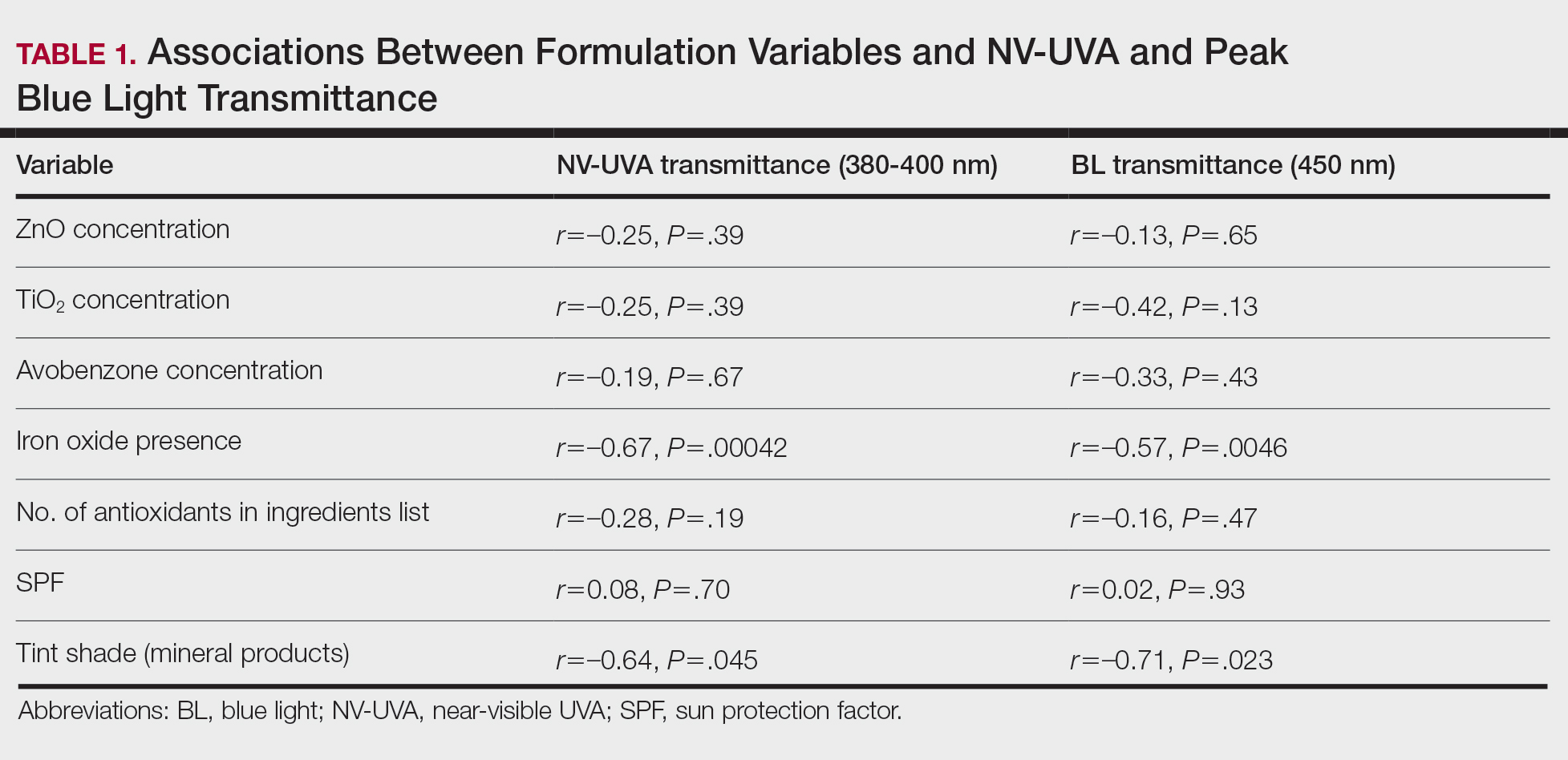
Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).
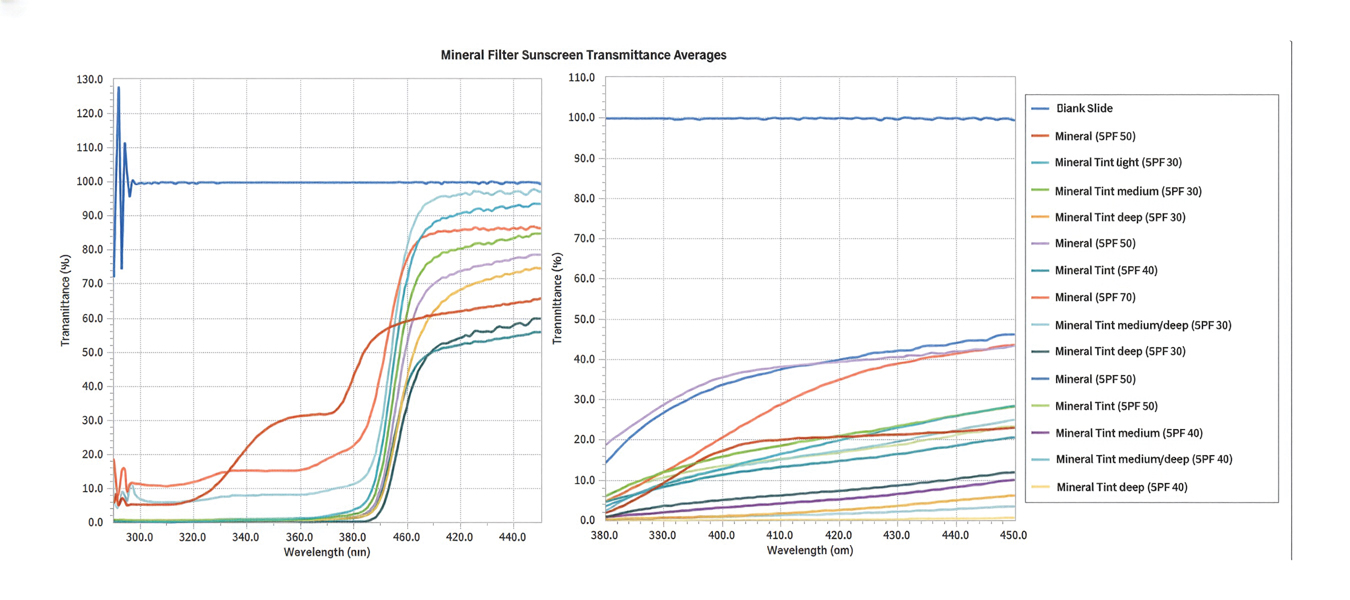
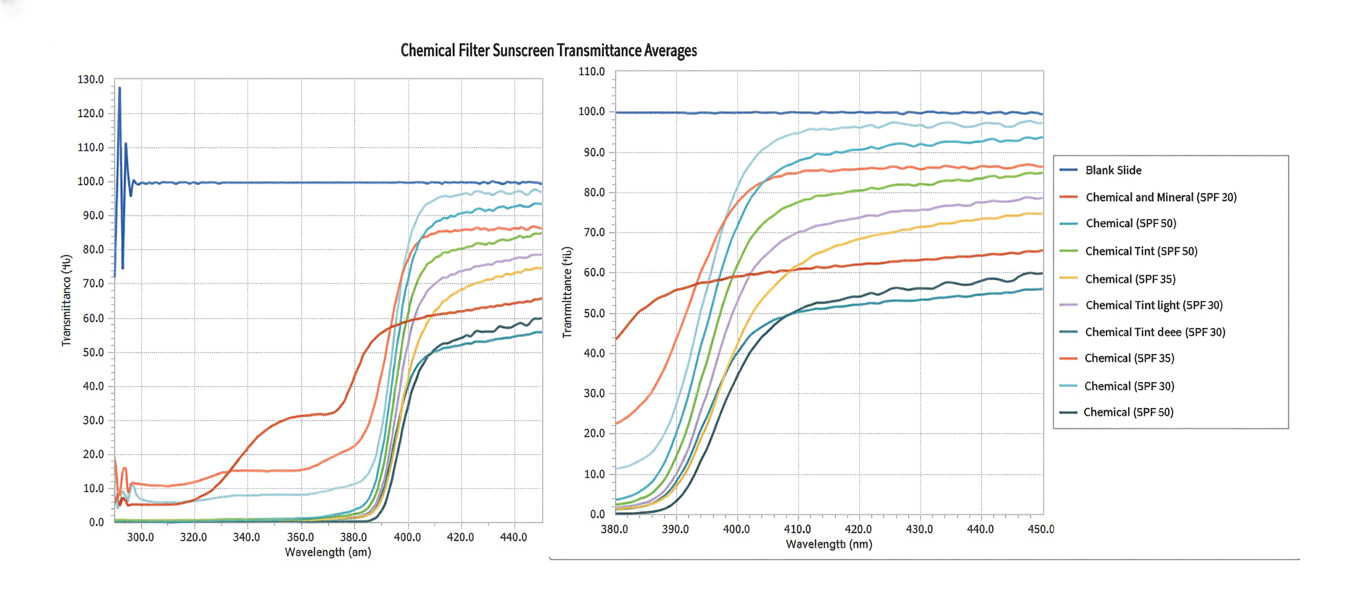
After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).
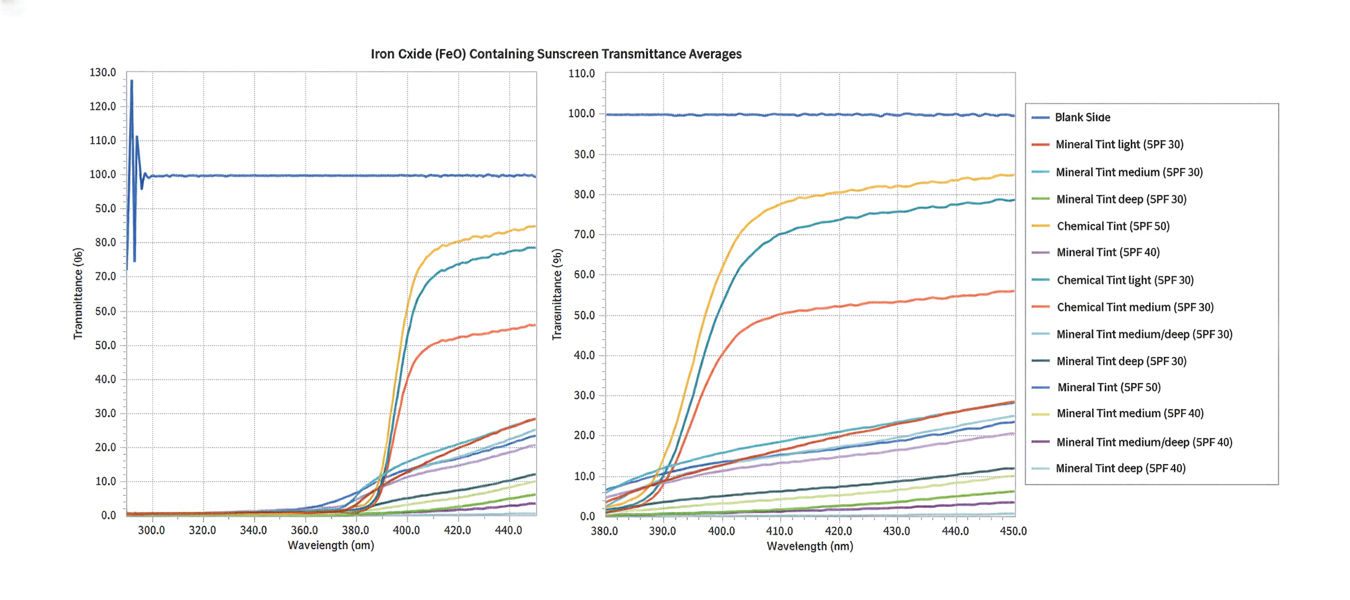
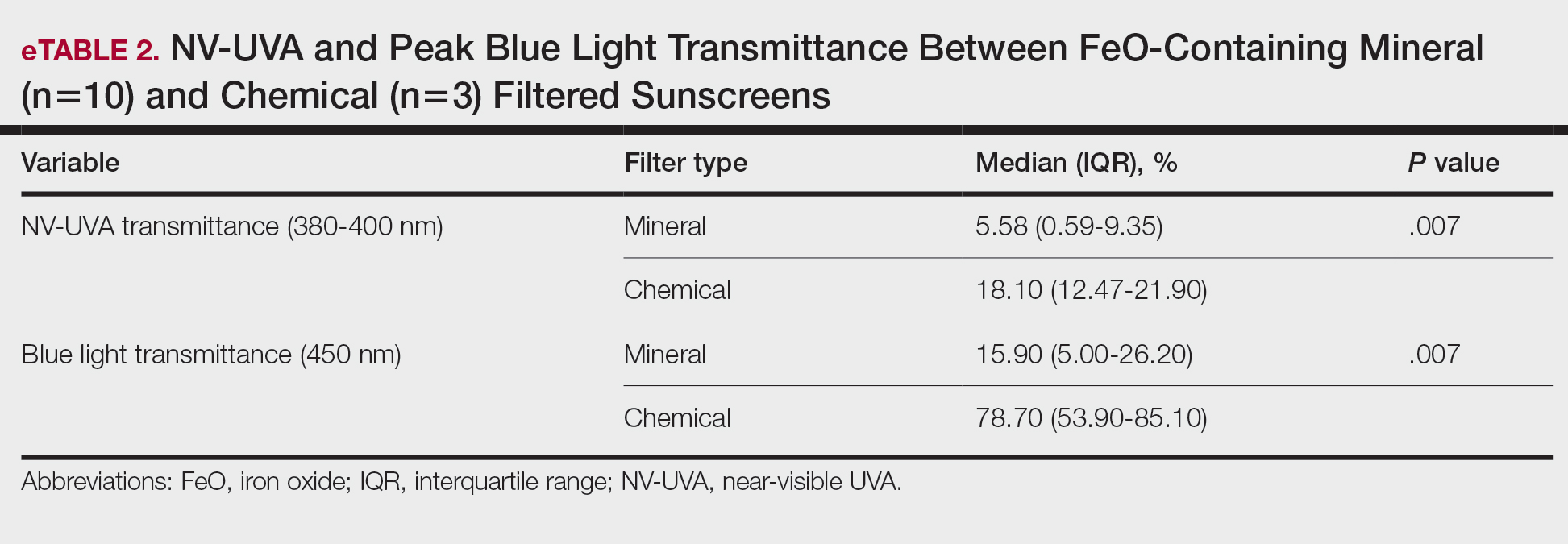
Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
Individuals with skin of color (SOC) are disproportionately affected by hyperpigmentation disorders such as melasma and postinflammatory hyperpigmentation following sun exposure. Although epidermal melanin provides UVB protection, susceptibility to pigmentary responses from longer UVA wavelengths and visible light (VL) remains, particularly the highest energy wavelengths of blue light (BL) between 400 and 450 nm.1 Blue light can induce immediate and persistent pigment darkening in those with Fitzpatrick skin types IV to VI, and trace amounts of near-visible UVA (NV-UVA) between 370 and 400 nm can synergize with VL to amplify pigmentation and erythema responses.2
Current photoprotection recommendations emphasize sun protection factor (SPF) ratings of 30+ and broad-spectrum labeling; however, under the US Food and Drug Administration standards, the broad-spectrum designation is based solely on achieving a mean critical wavelength of 370 nm or higher, which does not ensure meaningful attenuation of NV-UVA or VL wavelengths.3 Tinted sunscreens containing iron oxides (FeO) have been shown to improve protection against these pigment-inducing wavelengths,4 yet quantitative comparisons between tinted and nontinted commercial sunscreen products remain limited.
To address the gap in understanding about tinted vs nontinted commercial sunscreen products, we conducted an in vitro spectrophotometric comparative analysis. The objectives were to quantify NV-UVA and BL attenuation across products and evaluate whether formulation characteristics (eg, SPF rating, filter types and concentration, the presence and depth of tint, antioxidant content) would correlate with improved photoprotection in pigment-sensitive wavelengths. We hypothesized that formulation features such as higher SPF, inorganic filters, and the presence of tint antioxidants would be associated with superior NV-UVA and BL attenuation compared with nontinted formulations.
Methods
Sunscreen Selection—A convenience sample of 23 broad-spectrum sunscreens commercially available at drugstores was selected to reflect easily accessible options. Six sunscreen brands with tinted (n=13) and nontinted (n=10) counterpart formulations were included. Filter category (mineral and/or chemical), SPF, UV filter type and concentration, tint shade (light, medium, medium/deep, deep), number of photoprotective antioxidants (diethylhexyl syringylidenemalonate, vitamin E, vitamin C, licochalcone A, and glycyrrhetinic acid), and presence of FeO were recorded.
Substrate Preparation—Testing was performed using standardized polymethyl methacrylate (PMMA) plates. Sunscreens were mixed prior to application and applied at 1.3 mg/cm² per the European Cosmetic and Perfumery Association (COLIPA) UVA testing guidelines.5 Plates were reweighed to confirm dosing and dried in a dark environment for at least 15 minutes prior to testing.
Spectrophotometric Measurements—Spectral transmittance was measured from 250 to 450 nm using a spectrophotometer equipped with a xenon flash lamp (energy <0.2 J/cm²). Baseline transmission was recorded using untreated PMMA plates. Five scans were averaged per plate. Analyses focused on NV-UVA transmittance from 380 to 400 nm and peak BL transmission at 450 nm.
Mean NV-UVA transmittance was calculated as the arithmetic mean of percent transmittance measured at 1-nm increments from 380 to 400 nm (n=21). Because of the steep rise in transmittance between 380 and 400 nm and subsequent plateau into the visible range, this approach was used to approximate the area under the transmittance-wavelength curve over the specified interval, enabling direct comparison of NV-UVA penetration between formulations.
Statistical Analysis—Descriptive statistics were used to summarize transmittance values. Spearman rank correlation was used to assess associations between formulation variables and spectral attenuation. Analysis of covariance was used to evaluate the effect of FeO on transmittance while adjusting for SPF or filter type. The Mann-Whitney U test was used to compare NV-UVA and blue light transmittance between FeO-containing mineral and chemical formulations. Statistical significance was set at P<.05.
Results
Across broad-spectrum sunscreen formulations (N=23), mean SPF values were 40.4 (range, 30-70), and the mean number of antioxidants in the ingredient list was 1.5 (range, 0-4). Mean NV-UVA transmittance was 16.7% (range, 0.1%-55.0%) and mean BL transmittance was 44.3% (range, 0.3%-97.5%)(eTable 1).

The mean labeled zinc oxide (ZnO) concentration among ZnO-containing formulations (n=14) was 10.5% (range, 5.0%-21.6%), with mean NV-UVA and BL transmittance of 12.6% (range, 0.1%-55.0%) and 25.8% (range, 0.3%-67.2%), respectively. Mean NV-UVA and BL transmittance were 26.7% (range, 9.6%-55.0%) and 45.6% (range, 23.0%-67.2%) among ZnO formulations without FeO (n=5), compared with lower transmittance of 4.8% (range, 0.1%-11.5%) and 14.9% (range, 0.3%-29.5%) in ZnO formulations containing FeO (n=9).
The mean labeled titanium dioxide (TiO2) concentration among TiO2-containing formulations (n=14) was 9.0% (range, 3.2%-17.0%), with corresponding mean NV-UVA and BL transmittance of 9.5% (range, 0.1%-28.5%) and 22.7% (range, 0.3%-47.6%), respectively. Among TiO2 formulations without FeO (n=4), mean NV-UVA and BL transmittance was 19.7% (range, 9.6%-28.5%) and 39.8% (range, 23.0%-47.6%), while FeO-containing TiO2 formulations (n=10) showed lower mean NV-UVA and BL transmittance of 5.4% (range, 0.1%-11.5%) and 15.8% (range, 0.3%-29.5%), respectively. The mean labeled avobenzone concentration among avobenzone-containing formulations (n=8) was 2.9% (range, 2.5%-3%), with mean NV-UVA and BL transmittance of 24.7% (range, 10.2%-46.6%) and 79.2% (range, 53.9%-97.5%). Formulations without FeO (n=5) had mean NV-UVA and BL transmittance of 29.0% (range, 10.2%-46.6%) and 83.2% (range, 61.1%-97.5%), whereas FeO-containing products (n=3) demonstrated lower mean NV-UVA and BL transmittance of 17.5% (range, 12.5%-21.9%) and 72.6% (range, 53.9%-85.1%), respectively.
Among products containing ZnO, TiO2, and avobenzone, the specific UV filter concentrations showed no statistically significant correlation with NV-UVA or BL transmittance (all P>.05). Iron oxide presence significantly correlated with lower NV-UVA (r=–0.67; P=.00042) and lower BL transmittance (r=–0.57; P=.0046). The number of antioxidants in the ingredient list did not correlate with NV-UVA transmittance (r=–0.28; P=.19) or BL transmittance (r=–0.16; P=.47). Sun protection factor was not significantly correlated with either wavelength range (Table 1).

Tint shade was treated as an ordinal variable (light, medium, medium/deep, and deep; medium was considered the universal shade). Increasing tint shade depth was significantly associated with reduced NV-UVA (r=–0.64; P=.045) and BL (r=–0.71; P=.023), suggesting a dose-response relationship wherein darker tints exhibited greater attenuation of pigment-relevant wavelengths. Among mineral filter formulations, tinted products demonstrated lower NV-UVA and BL transmittance compared with their nontinted counterparts, with deeper tints providing the greatest reduction in transmittance (eFigure 1). Similar results were observed for chemical filter formulations with greater attenuation in the NV-UVA and BL range for tinted versus nontinted products with greater variability across shade depths (eFigure 2).


After adjusting for SPF, FeO presence remained significantly associated with reduced NV-UVA (F[1,20]=26.9; P<.001) and BL transmittance (F[1,20]=11.7; P=.003). After adjusting for filter type (mineral vs chemical), FeO remained significantly associated with NV-UVA (F[1,19]=10.1; P=.004) and BL transmittance (F[1,19]=10.4; P=.005)(Table 2).

Among FeO-containing products, mineral filters demonstrated significantly lower NV-UVA transmittance compared with chemical filters (median, 5.58% [interquartile range (IQR), 0.59%-9.35%] vs 18.10% [IQR, 12.47%-21.90%]; U=0.00; P=.007). The same was true for BL transmittance (median, 15.90% [IQR, 5.00%-26.20%] vs 78.70% [IQR, 53.90%-85.10%]; U=0.00; P=.007). The differences in spectral transmittance between FeO-containing mineral and chemical filter formulations are illustrated in eFigure 3, with mineral-based products demonstrating lower transmittance, particularly across the upper NV-UVA range and across the BL range. These results indicated greater pigment-relevant photoprotection with mineral vs chemical filters (eTable 2).


Comment
Our initial hypothesis proposed that tinted sunscreens would provide greater NV-UVA and BL attenuation than nontinted formulations, and that characteristics such as inorganic filter content, SPF rating, and antioxidants would correlate with improved protection in pigment-sensitive wavelengths. Our findings partially supported this hypothesis. In this analysis, substantial variability in the NV-UVA and BL transmittance was observed despite all products meeting broad-spectrum criteria. Nontinted mineral and chemical sunscreens exhibited high transmittance in these pigment-related wavelengths, reaching values as high as 55.0% for NV-UVA and 97.5% for BL. These findings align with prior analysis demonstrating that while broad-spectrum sunscreens available in the United States may meet the current critical wavelength criteria for protection in the UVA range, they still may transmit 30% to 66% of available UVA over 2 hours between formulations with equivalent SPF label values.6
Recent analyses show that sunscreen recommendations in lay media rarely incorporate input from board-certified dermatologists for individuals with SOC and disproportionately favor nontinted chemical formulations, despite the high prevalence of pigmentary disorders in this population.7 Near-visible UVA and BL have been demonstrated to be biologically relevant pigment-inducing wavelengths, both in vitro and in vivo, particularly in individuals with SOC, yet broad-spectrum labeling does not ensure protection against these spectra.8 Pigmentary tints such as FeO have demonstrated enhanced attenuation in this spectral region in vivo and may provide more reliable coverage than products with broad-spectrum designation alone.4,9 Treatment options for pigmentary disorders such as melasma tend to be palliative and costly, making optimized photoprotection a critical component of care to reduce ongoing pigmentary stimuli.10
Formulations containing FeO demonstrated significantly lower NV-UVA (P<.001) and BL transmittance (P=.003) on average; however, transmittance values ranged widely (NV-UVA: 0.10%-21.90%, BL: 0.30%-85.10%), indicating that FeO presence alone does not determine the magnitude of attenuation. Notably, among FeO-containing products, mineral filters provided significantly lower NV-UVA and BL transmittance compared with chemical filters (P=.007 for both), suggesting that filter type further modulates pigment-relevant photoprotection. Tinted formulations may improve compliance with product use by reducing the white cast and improve shade matching to find suitable options for deeper skin tones,11 but the highly variable photoprotection offered raises concerns about clinical benefit. Although deeper tints showed greater attenuation, pigment concentrations and combinations are not disclosed by manufacturers as FeO is not considered an active ingredient. Darker shades are not practical across all skin tones in individuals with SOC, which underscores the need for standardized pigment metrics and shade-inclusive options.
While avobenzone and ZnO are the only US Food and Drug Administration–approved sunscreen active ingredients that extend protection beyond 360 nm,12 both exhibited reduced attenuation beyond the longer end of the UVA spectrum. Formulation characteristics, including the concentration of ZnO, TiO2, and/or avobenzone as well as SPF, did not correlate with NV-UVA or BL attenuation. In the adjusted analysis, FeO presence remained significantly associated with reduced transmittance after adjusting for SPF (NV-UVA: P<.001, BL: P=.003) or filter type (NV-UVA: P=.004, BL: P=.005). These findings suggest that the presence of FeO, rather than UV filters or SPF ratings, supports attenuation in the 380 to 450–nm range, indicating a functional benefit in addition to improved cosmesis.13
Although antioxidants in specific combinations have shown promise in vivo, no association was observed between the number of antioxidants present and NV-UVA or BL attenuation compared with added tint.14 This suggests that specific antioxidant combinations and their concentrations may be more relevant than the total count.
Several study limitations need to be considered in interpreting our results, including a modest number of products, controlled in vitro testing conditions, and an incomplete representation of products with pigment concentrations and shade ranges marketed to individuals with SOC across all price categories, despite our focus on affordable, commercially available options. Moreover, PMMA-based spectrophotometry does not account for skin surface heterogeneity, photodegradation, sweat, oil, friction, or application variability, which may alter real-world performance. Additionally, FeO concentrations could not be quantified beyond labeling of tint shade depth, preventing a true assessment of dose-response effects. These limitations may reduce generalizability and highlight the need for complementary in vivo studies to assess clinically relevant outcomes such as persistent pigment darkening. For this reason, caution is warranted in extrapolating these spectral findings to clinical efficacy.
Conclusion
Given the susceptibility of individuals with SOC to pigmentary disorders driven by NV-UVA and BL, our findings support further development and study of FeO-containing sunscreens that address clinically relevant wavelengths. Wide variability in photo-attenuation among tinted formulations underscores the need for evidence-based recommendations, with further studies needed to guide photoprotection strategies for populations with SOC.
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
- Marionnet C, Piffaut V, Sasai J, et al. A precise analysis of the relative contribution of UVA1 and visible light colour domains in solar light-induced skin pigmentation. J Eur Acad Dermatol Venereol. 2023;37(suppl 4):3-11. doi:10.1111/jdv.18948
- Kohli I, Chaowattanapanit S, Mohammad TF, et al. Synergistic effects of long-wavelength ultraviolet A1 and visible light on pigmentation and erythema. Br J Dermatol. 2018;178:1173-1180. doi: 10.1111/bjd.15940
- US Food and Drug Administration. Over-the-counter monograph M020: sunscreen drug products for over-the-counter human use. September 24, 2021. Accessed April 7, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTCMonograph_M020-SunscreenDrugProductsforOTCHumanUse09242021.pdf
- Grimes PE, Paturi J, Chen Y, et al. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage. J Drugs Dermatol. 2025;24:662-667. doi:10.36849/JDD.9240
- Moyal D, Alard V, Bertin C, et al. The revised COLIPA in vitro UVA method. Int J Cosmet Sci. 2013;35:35-40. doi:10.1111/j.1468-2494.2012.00748.x
- Coelho SG, Rua D, Miller SA, et al. Suboptimal UVA attenuation by broad spectrum sunscreens under outdoor solar conditions contributes to lifetime UVA burden. Photodermatol Photoimmunol Photomed. 2020;36:42-52. doi:10.1111/phpp.12503
- Song H, Beckles A, Salian P, et al. Sunscreen recommendations for patients with skin of color in the popular press and in the dermatology clinic. Int J Womens Dermatol. 2020;7:165-170. doi:10.1016/j.ijwd.2020.10.008
- Lawrence KP, Douki T, Sarkany RPE, et al. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722. doi:10.1038/s41598-018-30738-6
- Ezekwe N, Pourang A, Lyons AB, et al. Evaluation of the protection of sunscreen products against long wavelength ultraviolet A1 and visible light-induced biological effects. Photodermatol Photoimmunol Photomed. 2024;40:E12937. doi:10.1111/phpp.12937
- Mpofana N, Chibi B, Gqaleni N, et al. Melasma in people with darker skin types: a scoping review protocol on prevalence, treatment options for melasma and impact on quality of life. Syst Rev. 2023;12:139. doi:10.1186/s13643-023-02300-7
- Wang JY, Patel P, Philip R, et al. Sunscreen practices and preferences of skin of color patients. J Drugs Dermatol. 2024;23:456-462. doi:10.36849/JDD.8268
- Beasley DG, Meyer TA. Characterization of the UVA protection provided by avobenzone, zinc oxide, and titanium dioxide in broad-spectrum sunscreen products. Am J Clin Dermatol. 2010;11:413-421. doi:10.2165/11537050-000000000-00000
- Morgado-Carrasco D, Delgado J, Prudkin-Silva Let al. Sunscreens prescribed to patients with skin of color and/or with melasma: a survey of 221 dermatologists and dermatology residents in Spain. Photodermatol Photoimmunol Photomed. 2024;40:E12996. doi:10.1111/phpp.12996
- Ruvolo E, Boothby-Shoemaker W, Kumar N, et al. Evaluation of efficacy of antioxidant-enriched sunscreen prodcuts against long wavelength ultraviolet A1 and visible light. Int J Cosmet Sci. 2022;44:394-402. doi:10.1111/ics.12785
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
Tinted vs Nontinted Sunscreens: Attenuation of Near-Visible UVA and Visible Light and Implications for Skin of Color
PRACTICE POINTS
- There is substantial variability in spectral attenuation among tinted sunscreens, highlighting the need for standardized pigment metrics and evidence-based photoprotection guidance for individuals with skin of color (SOC).
- Broad-spectrum labeling, sun protection factor values, UV filter type (mineral vs chemical), and antioxidant inclusion do not reliably predict protection against pigment-inducing UV and visible light wavelengths for individuals with SOC.
- Iron oxide–containing tinted sunscreens demonstrate lower near-visible UVA and blue light transmittance than nontinted formulations, with greater attenuation in mineral vs chemical products. Both pigment and filter type should inform photoprotection recommendations in individuals with SOC.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).

Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
To the Editor:
ChatGPT (OpenAI), a popular large language model that generates responses to user queries, has attracted substantial attention as a potential resource for patient education.1 While prior studies have shown that ChatGPT can provide reliable and general patient information, its alignment with the American Academy of Dermatology’s (AAD’s) guidelines for primary cutaneous melanoma (CM) compared to evidence in the recent literature has not been evaluated.2,3 In this study, we compared ChatGPT’s responses to the 25 evidence-based questions utilized by the AAD to establish its 2019 recommendations for primary CM. Because the 2019 AAD guidelines included literature only through April 2017, we conducted an additional search (May 2017–February 2024) to assess ChatGPT’s alignment with more recent evidence not captured in the guidelines.
On April 17, 2024, 2 authors (D.P. and A.F.) prompted ChatGPT with 25 evidence-based questions from the 2019 AAD guidelines for the management of primary CM.4 ChatGPT’s responses were compared with the AAD’s published recommendations and were cross-referenced with responses gathered from our own search of PubMed articles indexed for MEDLINE using the phrase melanoma (cutaneous) and treatment, which included studies from May 2017 to February 2024.
ChatGPT’s answers to 23 of the questions aligned with the AAD’s guidelines (Table 1); in instances when the guidelines were inconclusive regarding pathology, the model provided recommendations supported by our contemporary PubMed literature search. Of the 3 questions related to CM pathology, the AAD guidelines had sufficient evidence to provide recommendations for 2 questions. The first question evaluated the clinical information necessary to help the pathologist improve diagnosis (Table 2). ChatGPT’s response to one question about staged excision and Mohs micrographic surgery for melanoma in situ did not align with the AAD guidelines (Table 3).



Our results showed that ChatGPT provided comprehensive responses aligned with current evidence on CM treatment, except for one surgery question for which its response differed from the AAD guidelines. Our findings are consistent with an observational study that reported board-certified dermatologists rated ChatGPT’s responses on melanoma-related questions as 4.88 on a scale of 1 to 5 (1 indicated completely inaccurate information, 5 indicated complete accuracy for clinical sufficiency in practice). The authors also found that ChatGPT gave vague advice, such as to “get regular skin exams,” which is less specific than dermatologists’ recommendations for annual, biannual, or more frequent examinations.5 ChatGPT’s limitations in offering comprehensive answers for some questions aligned with our findings, specifically the omission of key information in the surgical-related question, highlighting the challenge of relying on AI for nuanced clinical guidance.
We found that ChatGPT considered immunosuppression an important risk factor for CM. Similarly, a 2023 cohort study of 93 patients with melanoma and a history of immunosuppression reported that these patients had a higher risk for CM compared with a control group from the National Cancer Institute’s Surveillance, Epidemiology and End Results Program (standardized incidence ratio, 1.53; 95% CI, 1.12-2.04), indicating that incidence of CM in immunocompromised patients was 53% higher than an age- and sex-matched population cohort.6
Our findings also demonstrated that both ChatGPT’s responses and the AAD guidelines aligned in indicating that evidence linking pregnancy to an increased risk for CM remains inconclusive and that pregnant women should still undergo surveillance. A 2022 retrospective cohort study of 1406 women comparing pregnancy-associated melanoma to non–pregnancy-associated CM had no difference in overall survival (hazard ratio, 0.75; 95% CI, 0.54-1.05).7 However, tumor thickness (2.01-4.00 mm) was greater in postpartum cases compared with cases in nonpregnant women (odds ratio, 1.75; 95% CI, 1.03-2.98), suggesting that pregnancy may affect tumor characteristics.7 These findings underscore the importance of using AI tools such as ChatGPT as a supplement to—rather than as a replacement for—expert clinical judgment and up-to-date medical guidelines.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
- Kung TH, Cheatham M, Medenilla A, et al. Performance of ChatGPT on USMLE: potential for AI-assisted medical education using large language models. PLOS Digit Health. 2023;2:E0000198.
- Roster K, Kann RB, Farabi B, et al. Readability and health literacy scores for ChatGPT-generated dermatology public education materials: cross-sectional analysis of sunscreen and melanoma questions. JMIR Dermatol. 2024;7:E50163.
- Dihan Q, Chauhan M, Eleiwa T, et al. Using large language models to generate educational materials on childhood glaucoma. Am J Ophthalmol. 2024;265:28-38.
- Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
- Young JN, O’Hagan R, Poplausky D, et al. The utility of ChatGPT in generating patient-facing and clinical responses for melanoma. J Am Acad Dermatol. 2023;89:602-604.
- Killeen TF, Shanley R, Ramesh V, et al. Malignant melanoma in a retrospective cohort of immunocompromised patients: a statistical and pathologic analysis. Cancers (Basel). 2023;15:3600.
- Kiuru M, Li Q, Zhu G, et al. Melanoma in women of childbearing age and in pregnancy in California, 1994-2015: a population-based cohort study. J Eur Acad Dermatol Venereol. 2022;36:2025-2035.
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
Alignment of ChatGPT Responses With AAD Guidelines for Cutaneous Melanoma
PRACTICE POINTS
- ChatGPT provides structured, educational-style responses with broad contextual detail but may omit key clinical nuances such as specific surgical considerations, including staged excision or Mohs micrographic surgery for melanoma in situ.
- Large language models should be viewed as a tool to supplement expert clinical judgment and established guidelines rather than as a standalone replacement for dermatologic decision-making.
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
Military service members are at high risk for skin cancer due to unique occupational and environmental exposures, particularly in the aviation community, in which high-altitude flying, prolonged outdoor aircraft maintenance, physical training, field exercises, and deployments limit access to shade and opportunities for sunscreen reapplication. During deployment or field operations, service members may operate in environments with limited access to SPF products, particularly if sunscreen is not included among personal items.
Research on sun protection strategies and skin cancer risk factors in military personnel is critical to improving prevention, particularly given the higher incidence of melanoma in this population. A 2010 retrospective tumor registry review from the Department of Defense and the National Cancer Institute found higher melanoma rates in military personnel compared with the general population among individuals aged 45 to 49 years (33.62 vs 27.49), 50 to 54 years (49.76 vs 32.18), and 55 to 59 years (178.48 vs 39.17).1
This article discusses barriers to sun protection in military populations, evaluates sunscreen availability in military exchanges, and considers implications for policy and prevention.
Barriers to Sun Protection and Sunscreen Use
According to Rosenberg et al,2 the cause of higher rates of skin cancer among military service members may be multifactorial, including financial barriers to sunscreen use, limited education on photodamage, and insufficient emphasis on sun protection during demanding operational or training activities. Veterans of Operation Enduring Freedom and Operation Iraqi Freedom who were surveyed about UV exposure and sunscreen indicated that 23% (49/211) received education about skin cancer but less than 30% (60/211) used sunscreen consistently during deployment due to lack of access, which has been reported previously.3 Sunscreen adherence also may be reduced in this population due to factors such as skin irritation, cost, poor cosmetic acceptability, and lower utilization among male service members. In their literature review of 9 publications pertaining to skin cancer risk through December 2016, Riemenschneider et al1 noted that male service members comprised 85% of the US military in 2014, and men statistically have lower rates of sunscreen use than women.
Sunscreen Availability and Product Analysis in Military Exchanges
Sunscreen is an important component of skin care for skin cancer prevention. More consistent use has been noted in households with annual incomes of $60,000 or higher.4 Sunscreen product availability has not been evaluated in the military community. Exchange stores are military equivalents of commercial chain stores where service members can purchase tax-free items. The Marine Corps Exchange (MCX) operates on 18 large active-duty bases worldwide. Patrons include active-duty service members from any branch, veterans, and family members. Officials from the MCX headquarters approve and maintain items sold on base. Although product availability may vary by location, standardization is maintained through vendor agreements influenced by customer demand and includes both exchange-branded and private-label products.5
In a review of 96 sunscreen products at Marine Corps Air Station Cherry Point MCX, 62.5% (60/96) met American Academy of Dermatology guideline criteria (SPF ≥30, broad-spectrum UVA/UVB protection, and water resistance of 40-80 minutes).6 Of all products, 79.1% (76/96) were SPF 30 or higher, 76.0% (73/96) were water-resistant, and all provided broad-spectrum protection. Lotion formulations comprised 62.5% (60/96), and the mean price per ounce was $11.96. Opportunities for product expansion include increased availability of options for sensitive skin, as mineral sunscreens comprised 14.6% (14/96) of products; greater variety of products marketed to men, which accounted for 5.2% (5/96); and improved representation for service members with skin of color, as tinted formulations comprised 2.1% (2/96).6
Implications for Policy and Operational Readiness
Given these data, future studies should evaluate sunscreen purchasing behaviors among US service members to determine MCX utilization and whether product selection is driven by active-duty demand or broader consumer purchasing patterns. If product offerings are driven by the civilian customer base, this may result in a lack of tailored options for military service members who are most at risk for high UV exposure. If the MCX does not meet the needs of service members adequately or is inaccessible due to cost or inventory limitations, it highlights a weakness in skin cancer prevention.
Future research should explore not only sunscreen purchasing behavior among service members but also barriers to access and compliance with sun protection measures, as these insights are critical for informing effective policy that balances personal responsibility with institutional support. This could help with advocacy efforts for more effective, readily available options on base. It also could strengthen the argument for alternative strategies to complement sunscreen use, such as a sunscreen allowance, inclusion of sunscreen with provided uniforms and equipment, patient education, work breaks, sun-protective uniform items, and designated shade areas at work.6
Final Thoughts
Policy changes such as routine provision of sunscreen through supply chains, issuing sunscreen with uniforms, or providing a sunscreen stipend could remove financial and logistical barriers to consistent use of sunscreen in military populations. These measures could be impactful during field operations, deployments, and training in austere environments, where commercial purchasing options are limited and UV exposure is high. A proactive approach to sun safety could demonstrate a commitment to preserving the current health and operational readiness of active-duty service members while reducing future financial burdens of skin disease and helping promote wellness in this population during retirement. As with ear protection, uniforms, and eyewear, sunscreen should be considered a standard component of operational readiness.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192. doi:10.1016/j.jaad.2017.11.062
- Rosenberg A, Cho S. We can do better at protecting our service members from skin cancer. Mil Med. 2022;187:311-313. doi:10.1093/milmed/usac198
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873. doi:10.1038/jid.2015.238
- Tahir S, Ihebom D, Garcia E, et al. Sunscreen access, availability, and quality in dollar store chains. J Am Acad of Dermatol. 2024;90:1284-1285. doi:10.1016/j.jaad.2024.02.018
- mymcx.com. Published 2025. Accessed May 15, 2025. https://www.mymcx.com/
- American Academy of Dermatology Association. How to select a sunscreen. Accessed April 23, 2026. https://www.aad.org/public/everyday-care/sun-protection/shade-clothing-sunscreen/how-to-select-sunscreen.
- Anderson S, Zhang S, Crotty A, et al. Bridging the knowledge-action gap in skin cancer prevention among US military personnel. Cutis. 2025;115:146-149. doi:10.12788/cutis.1207
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Sunscreen Access and Skin Cancer Prevention: Availability at the Marine Corps Exchange
Practice Points
- Military service members face increased UV exposure from occupational and deployment conditions, but sunscreen use is limited by logistical, educational, and behavioral barriers.
- Sunscreen availability in military exchanges partially meets dermatologic guidelines, highlighting opportunities to expand tailored options and integrate sun protection into operational readiness policy.
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
Sunscreen continues to be the foundation of successful skin cancer prevention. Daily sunscreen application and reapplication are recommended to prevent all types of skin cancer, with the strongest body of evidence supporting prevention of squamous cell carcinoma.1 Sunscreens have been used safely for decades; since my last update in 2024,2 no evidence of harm has emerged, despite the fact that organic (chemical) sunscreen filters are absorbed systemically.3
So, what’s happening with sunscreen in 2026? Let’s review some hot news and fresh controversies.
Sunscreen Doping Is Pervasive
Sunscreen “doping” signifies the sneaky addition of UV filters that have not been approved by the US Food and Drug Administration (FDA) into sunscreens under the guise of inactive ingredients. Why would a manufacturer do such a thing? To enhance a sunscreen’s UV absorption without having to increase the concentration of zinc oxide/titanium dioxide (which creates an undesirable white cast) or exceeding the maximum permitted concentration of chemical active ingredients.4,5 In a 2025 analysis of the top 150 sunscreens sold on Amazon, 48.3% contained these covert UV filters, including almost half of those marketed as mineral-only products.6 The most prevalent doping ingredient was butyloctyl salicylate, which is chemically and functionally related to the FDA-approved chemical UV filter octisalate (ethylhexyl salicylate).5
The practice of sunscreen doping is deceptive. Can a product be accurately marketed as mineral sunscreen if it contains ingredients that function as chemical UV filters but are not classified as active ingredients by the FDA? The bigger picture is that sunscreen doping is a symptom of regulatory malaise specific to the United States. Regulation of sunscreens as over-the-counter drugs plus the FDA’s stringent requirements for UV filters to be generally recognized as safe and effective (GRASE) have stymied the approval process to the extent that no new active ingredients have been approved since 1999.2 The FDA allows 16 active ingredients compared to about 30 in Europe and Asia—not for lack of safety evaluations prior to approval in those regions.7 In the United States, getting a new active sunscreen ingredient approved is far more onerous and costly than the streamlined processes that are in place abroad. This restricts sunscreen innovation; in particular, the US market lacks the wide variety of international options for protection against long-wave UVA radiation, remaining limited to just avobenzone and zinc oxide. Since long-wave UVA plays a major role in photoaging, this represents a gap in protection compared to international sunscreen offerings.1,7 Due to domestic sunscreen limitations, some Americans have turned to purchasing non–FDA-approved sunscreens abroad or through online channels.8
New Sunscreen Filter Pending Approval, and Hope for Regulatory Changes
Let’s move on to a more positive development. A new sunscreen filter is actually nearing approval in the United States! Bemotrizinol, also known as bis-ethylhexyloxyphenol methoxyphenyl triazine, is a broad-spectrum chemical UVA/UVB blocker that would represent the first new active ingredient to become available in the United States since 1999.9 It satisfies the FDA requirement for minimal systemic absorption and GRASE status and has been used with a clean safety record since 2000 in Europe.10 The icing on top is that bemotrizinol seems to be minimally allergenic, with only a few published reports of contact dermatitis over several decades of use.11,12
Yes, as I write, the FDA is on the cusp of approving bemotrizinol, a great broad-spectrum sunscreen ingredient, to one day be added to the products on our shelves. The cynic in me can’t help but point out that it took more than 20 years of effort and an estimated $20 million to get us to this point of near-approval of one new sunscreen filter.13
Perhaps things won’t be so difficult in the future. In late 2025, the bipartisan Supporting Accessible, Flexible, and Effective (SAFE) Sunscreen Standards Act was signed into law.14 The SAFE Sunscreen Standards Act calls on the FDA to be more flexible and allow for the use of real-world evidence and observational studies to demonstrate safety and effectiveness of active ingredients used in sunscreens. We can only hope that real change is forthcoming and that future sunscreen approvals won’t require decades of work and millions of dollars, as in the case of bemotrizinol.
Daily Sunscreen Use Linked to Reduction in Vitamin D Levels
The UVB wavelengths that cause sunburn overlap with those that initiate vitamin D production in the skin, generating concerns about sunscreen use reducing vitamin D levels. Nevertheless, in 2019, expert opinion and a systematic literature review determined that routine use of sunscreen was unlikely to be associated with a reduction in vitamin D levels.15,16 However, a major limitation at that time was a lack of studies examining vitamin D status in individuals using high–sun protection factor (SPF) sunscreens.
Now we have results from the first field study assessing the impact of long-term daily application of higher SPF sunscreen on vitamin D levels. In the Australian Sun-D Trial, Tran et al17 randomly assigned 639 participants to either an intervention group (routine application of SPF 50+ sunscreen on days forecasted to have a UV index ≥3) or a control group (discretionary sunscreen use). Vitamin D levels were measured at baseline in the winter/spring, at the end of summer, and then at the end of the following winter. At the end of summer, vitamin D levels increased in both groups but less in the intervention group, then decreased similarly in both groups by winter. Routine sunscreen application was associated with a decrease of 5.2 nmol/L (2.1 ng/mL) in vitamin D levels, which the authors rightfully considered to be modest. Additionally, vitamin D deficiency (defined as <50 nmol/L [<20 ng/mL]) was detected in more of the intervention group compared to the control group (45.7% vs 36.9%). The study reasonably concluded that sunscreen continues to be essential in preventing skin cancers but regular users may require vitamin D testing and/or supplementation.17
Looking Ahead
In this update, I discussed several important pieces of sunscreen news. If you check your favorite mineral sunscreen’s ingredients list, odds are you will find it also contains inactive doping ingredients shown to secretly enhance UV protection. Perhaps manufacturers won’t have to dope sunscreens in the United States forever if regulatory reforms facilitate the approval of active ingredients such as bemotrizinol used safely in other countries without huge investments of time and money. For daily sunscreen users, consider checking and/or empirically supplementing vitamin D.
None of this should discourage us from recommending regular consistent sunscreen application and reapplication to our patients. There continues to be a lack of evidence of harms associated with systemic absorption of chemical UV filters in humans, and sunscreen will continue to function as an indispensable component of skin cancer prevention for the foreseeable future.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 1: mechanisms and efficacy. J Am Acad Dermatol. 2025;92:677-686.
- Adler BL. Sunscreen safety: 2024 updates. Cutis. 2024;113:195-196.
- Abdel Azim S, Bainvoll L, Vecerek N, et al. Sunscreens part 2: regulation and safety. J Am Acad Dermatol. 2025;92:689-698.
- Gawey L, Gunnell L, Shi VY. Sunscreen doping: a misnomer. J Invest Dermatol. 2025;145:2941-2942.
- Moradi Tuchayi S, Wang Z, Yan J, et al. Sunscreens: misconceptions and misinformation. J Invest Dermatol. 2023;143:1406-1411.
- Gawey L, Gunnell L, Joshi A, et al. Hidden UV filters: an analysis of the 150 most popular sunscreens. J Am Acad Dermatol. 2025;93:1594-1596.
- Pantelic MN, Wong N, Kwa M, et al. Ultraviolet filters in the United States and European Union: a review of safety and implications for the future of US sunscreens. J Am Acad Dermatol. 2023;88:632-646.
- Scaturro M. Other countries have better sunscreens. here’s why we can’t get them in the U.S. NPR. May 18, 2024. Accessed April 9, 2026. https://www.npr.org/sections/health-shots/2024/05/18/1251919831/sunscreen-effective-better-ingredients-fda
- US Food & Drug Administration. FDA proposes expanding sunscreen active ingredient list [news release]. December 11, 2025. Accessed February 24, 2026. https://www.fda.gov/news-events/press-announcements/fda-proposes-expanding-sunscreen-active-ingredient-list
- D’Ruiz CD, Plautz JR, Schuetz R, et al. Preliminary clinical pharmacokinetic evaluation of bemotrizinol—a new sunscreen active ingredient being considered for inclusion under FDA’s over-the-counter (OTC) sunscreen monograph. Regul Toxicol Pharmacol. 2023;139:105344.
- Badaoui A. Allergic contact dermatitis to bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb S) in a sunscreen: an emerging allergen? Contact Dermatitis. 2024;91:443-445.
- European Multicentre Photopatch Test Study (EMCPPTS) Taskforce. A European multicentre photopatch test study. Br J Dermatol. 2012;166:1002-1009.
- Nesvig K. The FDA finally proposed the approval of a new sunscreen filter. December 11, 2025. Accessed February 22, 2026. https://www.allure.com/story/fda-proposes-bemotrizinol-suncreen-filter-approval
- SAFE Sunscreen Standards Act, HR 3686, 119th Congress (2025-2026). Accessed February 23, 2026. https://www.congress.gov/bill/119th-congress/house-bill/3686/text
- Neale RE, Khan SR, Lucas RM, et al. The effect of sunscreen on vitamin D: a review. Br J Dermatol. 2019;181:907-915.
- Passeron T, Bouillon R, Callender V, et al. Sunscreen photoprotection and vitamin D status. Br J Dermatol. 2019;181:916-931.
- Tran V, Duarte Romero BL, Andersen H, et al. Effect of daily sunscreen application on vitamin D: findings from the open-label randomized controlled Sun-D Trial. Br J Dermatol. 2025;193:1128-1137.
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
Sunscreen in 2026: Doping, Regulatory Changes, and Impact on Vitamin D Levels
No Survival Gain With Adjuvant Therapy in Stage III Melanoma
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
AI Skin Cancer Apps: Do They Work?
An AI-based skin assessment app may drive up healthcare visits for benign lesions, with unclear benefits for skin cancer detection, a Dutch clinical trial has found.
The trial, of nearly 20,000 patients in one health insurance plan, found that those given free access to the app were no more likely to be diagnosed with skin cancer over 1 year than participants assigned to a control group with no app access. They were, however, more likely to make healthcare visits for benign skin lesions.
The results came as a surprise, lead researcher Marlies Wakkee, MD, PhD, said during a presentation at the European Association of Dermato-Oncology (EADO) Congress 2026, held in Prague, Czech Republic.
“We were a bit flabbergasted,” said Wakkee, of Erasmus MC in Rotterdam, Netherlands. “We were, of course, expecting that those who would use this intervention app would have more skin cancer diagnoses than those who did not.”
She did, however, point to a potential reason for the lack of benefit: A deeper look at the data suggested that participants in the control group might have been particularly motivated to see their doctor for suspicious skin growths.
Can AI Apps Fill a Gap?
Wakkee pointed out that routine skin cancer screening via clinical skin examination is considered infeasible in many countries. Current guidance from the US Preventive Services Task Force says there is insufficient evidence to assess the balance of benefits and harms from widespread screening.
A plethora of AI-based skin assessment apps have entered the market in recent years, Wakkee said, and in theory, they have the potential to aid in earlier skin cancer diagnosis. But, she added, the technology also comes with potential harms, ranging from spurring healthcare visits for benign lesions to missing true cancers.
The current trial focused on the SkinVision app. It relies on a convolutional neural network to analyze images of skin lesions captured by the user’s smartphone and provides risk assessments of low, medium or high; a tele-dermatology team is available for support.
The app has been reimbursed in Netherlands via health insurance companies since 2019, and by 2021, it was available to 2.2 million insurees, with an uptake of about 1%, according to Wakkee.
In a previous study, the researchers used insurance claims data to study 18,960 app users and compare them with 56,880 nonusers. They found that app use was associated with an increased likelihood of being diagnosed with cutaneous malignancies and premalignancies but also benign tumors and nevi.
“So there’s a group in there that just is very worried about their skin,” Wakkee said.
To investigate further, her team conducted the SPOT-study, a randomized controlled trial in which roughly 226,000 adults covered by a Dutch nonprofit health insurance provider were invited to take part.
Of those, just over 19,000 agreed and were randomly assigned to either an intervention group that had free access to the skin app for 12 months or a control group that had no access. They were told that if they had any skin lesions they were worried about, they should visit their general practitioner.
During that period, the study found there was no significant difference in rates of histologically verified melanoma between the intervention and control groups, at 0.26% vs 0.31% — a risk difference of -0.05% (P = .68).
Similarly, the groups showed no difference in rates of any type of skin cancer, including squamous cell and basal cell carcinomas, at 2.66% in the intervention group vs 2.27% in the control group (P = .10). Rates of premalignant lesions were also comparable (6.9% vs 6.3%; P = .23).
The researchers then examined participants’ claims data to look at healthcare visits for benign skin lesions. There, app users did have a significantly higher rate, at 3.9% vs 2.6% (P < .001).
A Case of Inherent Bias?
The lack of benefit for skin cancer detection prompted the researchers to view the data from a different angle. They compared their trial participants with over 200,000 nonresponders from the health insurance plan. And that’s when a difference emerged.
Overall, trial participants were nearly three times more likely to have a skin premalignancy or malignancy diagnosed during that period, at 6.7% vs 2.4% (P < .001).
Wakkee said that because trial participants were told that the study aimed to gauge “the potential impact of this technology” in assessing skin lesions, that might have created an inherent bias. Participants assigned to the control group may have been motivated to have any worrisome skin growth checked out by their general practitioners.
In addition, Wakkee cautioned that the 12-month results are based on a small number of cancer cases, making it difficult to draw firm conclusions about the app’s performance. The trial has a second phase, where both groups were given free access to the app for 12 months, then followed for an additional 24 months.
Longer-term data are needed, Wakkee noted, in part to see whether people’s app usage changes over time.
Future Questions
Audience members at the presentation raised questions about how AI-based apps could be best deployed for skin cancer detection — including whether they might work better in the hands of clinicians rather than patients.
Wakkee said that clinicians would need a more advanced technology than that included in the app used in this trial. But future studies, she said, will look at whether the app can be used in a more targeted way, specifically, as a triage tool for people who are already concerned about something on their skin, to help them decide if they need to visit their doctor.
One presentation attendee wondered whether people given a low-risk result by the app were likely to be reassured or still make an appointment.
Wakkee said her team has begun to dig into that question. In a pilot study, 50 patients who wanted to see their general practitioner for a skin lesion were asked: If you received a low-risk rating on the skin app, would you still visit your doctor?
“Half of them said they would stay at home,” Wakkee said. She added, however, that her team is conducting a follow-up study to see what people actually do.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
An AI-based skin assessment app may drive up healthcare visits for benign lesions, with unclear benefits for skin cancer detection, a Dutch clinical trial has found.
The trial, of nearly 20,000 patients in one health insurance plan, found that those given free access to the app were no more likely to be diagnosed with skin cancer over 1 year than participants assigned to a control group with no app access. They were, however, more likely to make healthcare visits for benign skin lesions.
The results came as a surprise, lead researcher Marlies Wakkee, MD, PhD, said during a presentation at the European Association of Dermato-Oncology (EADO) Congress 2026, held in Prague, Czech Republic.
“We were a bit flabbergasted,” said Wakkee, of Erasmus MC in Rotterdam, Netherlands. “We were, of course, expecting that those who would use this intervention app would have more skin cancer diagnoses than those who did not.”
She did, however, point to a potential reason for the lack of benefit: A deeper look at the data suggested that participants in the control group might have been particularly motivated to see their doctor for suspicious skin growths.
Can AI Apps Fill a Gap?
Wakkee pointed out that routine skin cancer screening via clinical skin examination is considered infeasible in many countries. Current guidance from the US Preventive Services Task Force says there is insufficient evidence to assess the balance of benefits and harms from widespread screening.
A plethora of AI-based skin assessment apps have entered the market in recent years, Wakkee said, and in theory, they have the potential to aid in earlier skin cancer diagnosis. But, she added, the technology also comes with potential harms, ranging from spurring healthcare visits for benign lesions to missing true cancers.
The current trial focused on the SkinVision app. It relies on a convolutional neural network to analyze images of skin lesions captured by the user’s smartphone and provides risk assessments of low, medium or high; a tele-dermatology team is available for support.
The app has been reimbursed in Netherlands via health insurance companies since 2019, and by 2021, it was available to 2.2 million insurees, with an uptake of about 1%, according to Wakkee.
In a previous study, the researchers used insurance claims data to study 18,960 app users and compare them with 56,880 nonusers. They found that app use was associated with an increased likelihood of being diagnosed with cutaneous malignancies and premalignancies but also benign tumors and nevi.
“So there’s a group in there that just is very worried about their skin,” Wakkee said.
To investigate further, her team conducted the SPOT-study, a randomized controlled trial in which roughly 226,000 adults covered by a Dutch nonprofit health insurance provider were invited to take part.
Of those, just over 19,000 agreed and were randomly assigned to either an intervention group that had free access to the skin app for 12 months or a control group that had no access. They were told that if they had any skin lesions they were worried about, they should visit their general practitioner.
During that period, the study found there was no significant difference in rates of histologically verified melanoma between the intervention and control groups, at 0.26% vs 0.31% — a risk difference of -0.05% (P = .68).
Similarly, the groups showed no difference in rates of any type of skin cancer, including squamous cell and basal cell carcinomas, at 2.66% in the intervention group vs 2.27% in the control group (P = .10). Rates of premalignant lesions were also comparable (6.9% vs 6.3%; P = .23).
The researchers then examined participants’ claims data to look at healthcare visits for benign skin lesions. There, app users did have a significantly higher rate, at 3.9% vs 2.6% (P < .001).
A Case of Inherent Bias?
The lack of benefit for skin cancer detection prompted the researchers to view the data from a different angle. They compared their trial participants with over 200,000 nonresponders from the health insurance plan. And that’s when a difference emerged.
Overall, trial participants were nearly three times more likely to have a skin premalignancy or malignancy diagnosed during that period, at 6.7% vs 2.4% (P < .001).
Wakkee said that because trial participants were told that the study aimed to gauge “the potential impact of this technology” in assessing skin lesions, that might have created an inherent bias. Participants assigned to the control group may have been motivated to have any worrisome skin growth checked out by their general practitioners.
In addition, Wakkee cautioned that the 12-month results are based on a small number of cancer cases, making it difficult to draw firm conclusions about the app’s performance. The trial has a second phase, where both groups were given free access to the app for 12 months, then followed for an additional 24 months.
Longer-term data are needed, Wakkee noted, in part to see whether people’s app usage changes over time.
Future Questions
Audience members at the presentation raised questions about how AI-based apps could be best deployed for skin cancer detection — including whether they might work better in the hands of clinicians rather than patients.
Wakkee said that clinicians would need a more advanced technology than that included in the app used in this trial. But future studies, she said, will look at whether the app can be used in a more targeted way, specifically, as a triage tool for people who are already concerned about something on their skin, to help them decide if they need to visit their doctor.
One presentation attendee wondered whether people given a low-risk result by the app were likely to be reassured or still make an appointment.
Wakkee said her team has begun to dig into that question. In a pilot study, 50 patients who wanted to see their general practitioner for a skin lesion were asked: If you received a low-risk rating on the skin app, would you still visit your doctor?
“Half of them said they would stay at home,” Wakkee said. She added, however, that her team is conducting a follow-up study to see what people actually do.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
An AI-based skin assessment app may drive up healthcare visits for benign lesions, with unclear benefits for skin cancer detection, a Dutch clinical trial has found.
The trial, of nearly 20,000 patients in one health insurance plan, found that those given free access to the app were no more likely to be diagnosed with skin cancer over 1 year than participants assigned to a control group with no app access. They were, however, more likely to make healthcare visits for benign skin lesions.
The results came as a surprise, lead researcher Marlies Wakkee, MD, PhD, said during a presentation at the European Association of Dermato-Oncology (EADO) Congress 2026, held in Prague, Czech Republic.
“We were a bit flabbergasted,” said Wakkee, of Erasmus MC in Rotterdam, Netherlands. “We were, of course, expecting that those who would use this intervention app would have more skin cancer diagnoses than those who did not.”
She did, however, point to a potential reason for the lack of benefit: A deeper look at the data suggested that participants in the control group might have been particularly motivated to see their doctor for suspicious skin growths.
Can AI Apps Fill a Gap?
Wakkee pointed out that routine skin cancer screening via clinical skin examination is considered infeasible in many countries. Current guidance from the US Preventive Services Task Force says there is insufficient evidence to assess the balance of benefits and harms from widespread screening.
A plethora of AI-based skin assessment apps have entered the market in recent years, Wakkee said, and in theory, they have the potential to aid in earlier skin cancer diagnosis. But, she added, the technology also comes with potential harms, ranging from spurring healthcare visits for benign lesions to missing true cancers.
The current trial focused on the SkinVision app. It relies on a convolutional neural network to analyze images of skin lesions captured by the user’s smartphone and provides risk assessments of low, medium or high; a tele-dermatology team is available for support.
The app has been reimbursed in Netherlands via health insurance companies since 2019, and by 2021, it was available to 2.2 million insurees, with an uptake of about 1%, according to Wakkee.
In a previous study, the researchers used insurance claims data to study 18,960 app users and compare them with 56,880 nonusers. They found that app use was associated with an increased likelihood of being diagnosed with cutaneous malignancies and premalignancies but also benign tumors and nevi.
“So there’s a group in there that just is very worried about their skin,” Wakkee said.
To investigate further, her team conducted the SPOT-study, a randomized controlled trial in which roughly 226,000 adults covered by a Dutch nonprofit health insurance provider were invited to take part.
Of those, just over 19,000 agreed and were randomly assigned to either an intervention group that had free access to the skin app for 12 months or a control group that had no access. They were told that if they had any skin lesions they were worried about, they should visit their general practitioner.
During that period, the study found there was no significant difference in rates of histologically verified melanoma between the intervention and control groups, at 0.26% vs 0.31% — a risk difference of -0.05% (P = .68).
Similarly, the groups showed no difference in rates of any type of skin cancer, including squamous cell and basal cell carcinomas, at 2.66% in the intervention group vs 2.27% in the control group (P = .10). Rates of premalignant lesions were also comparable (6.9% vs 6.3%; P = .23).
The researchers then examined participants’ claims data to look at healthcare visits for benign skin lesions. There, app users did have a significantly higher rate, at 3.9% vs 2.6% (P < .001).
A Case of Inherent Bias?
The lack of benefit for skin cancer detection prompted the researchers to view the data from a different angle. They compared their trial participants with over 200,000 nonresponders from the health insurance plan. And that’s when a difference emerged.
Overall, trial participants were nearly three times more likely to have a skin premalignancy or malignancy diagnosed during that period, at 6.7% vs 2.4% (P < .001).
Wakkee said that because trial participants were told that the study aimed to gauge “the potential impact of this technology” in assessing skin lesions, that might have created an inherent bias. Participants assigned to the control group may have been motivated to have any worrisome skin growth checked out by their general practitioners.
In addition, Wakkee cautioned that the 12-month results are based on a small number of cancer cases, making it difficult to draw firm conclusions about the app’s performance. The trial has a second phase, where both groups were given free access to the app for 12 months, then followed for an additional 24 months.
Longer-term data are needed, Wakkee noted, in part to see whether people’s app usage changes over time.
Future Questions
Audience members at the presentation raised questions about how AI-based apps could be best deployed for skin cancer detection — including whether they might work better in the hands of clinicians rather than patients.
Wakkee said that clinicians would need a more advanced technology than that included in the app used in this trial. But future studies, she said, will look at whether the app can be used in a more targeted way, specifically, as a triage tool for people who are already concerned about something on their skin, to help them decide if they need to visit their doctor.
One presentation attendee wondered whether people given a low-risk result by the app were likely to be reassured or still make an appointment.
Wakkee said her team has begun to dig into that question. In a pilot study, 50 patients who wanted to see their general practitioner for a skin lesion were asked: If you received a low-risk rating on the skin app, would you still visit your doctor?
“Half of them said they would stay at home,” Wakkee said. She added, however, that her team is conducting a follow-up study to see what people actually do.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
“Noteworthy” Link Between Agent Orange and Acral Melanoma Found
Recent research has revealed potential links between Agent Orange (AO) exposure and risk of acral melanoma (AM) among Vietnam War-era veterans, providing strong evidence of a relationship between the chemical and this type of cancer.
Localized to the palms, soles, and nail units, AM is a melanoma subtype less associated with UV radiation. From 1962 to 1971, the US military sprayed an estimated 18 million gallons of herbicides, including AO, over the fields and forests of Vietnam. Those herbicides have since been connected to numerous health issues, including cancer, though evidence of a relationship between AO and skin cancers has been weak.
Vietnam War-era veterans have a higher melanoma burden than the general population, with the disease being the fourth-most common cancer among those who served. AM, however, is rare, representing about 2% to 3% of all melanomas.
In a nested case-control study, Hwang et al used US Department of Veterans Affairs (VA) health care system data, including the VA Cancer Registry. The authors compared 1292 patients with AM and 2 pair-matched control groups: a group matched 4:1 to nonacral cutaneous melanoma controls, and a group without a melanoma diagnosis.
Hwang et al found AO exposure was associated with increased odds of AM compared with each control group. In an accompanying editorial, Andrew Olshan, PhD, from Department of Epidemiology at the University of North Carolina Gillings School of Global Public Health, wrote, “The magnitude of the effects was modest (about 30%) but noteworthy.”
A limitation of the study was that presumptive AOE status was based on whether the veteran filed a disability claim with evidence of officially recognized service in a period and place where Agent Orange was used—not on an assessment of the veteran’s individual AOE potential, including level of exposure. Because melanoma has never been included on the VA list of cancers presumed to be related to AO exposure, veterans do not automatically gain benefits by filing AOE claims after diagnosis. Even so, Olshan says, the reported study findings may underestimate the true effect of AO exposure on the risk of AM.
Given the rarity of AM, the association (if causal) would translate to 0.4 to 0.8 new annual cases of AM per 1,000,000 veterans, according to the study. Narrowed down to Vietnam War-era veterans—who are dwindling in number—the attributable cases would be scarce.
Nevertheless, the search for a better understanding of a potential link between AOE and melanomas among Vietnam War-era veterans is important, Olshan wrote.
“The Hwang et al study provides a strong impetus to further these research goals and contribute to the investigation of the legacy of the Vietnam War and honor a commitment to the veterans community.”
Recent research has revealed potential links between Agent Orange (AO) exposure and risk of acral melanoma (AM) among Vietnam War-era veterans, providing strong evidence of a relationship between the chemical and this type of cancer.
Localized to the palms, soles, and nail units, AM is a melanoma subtype less associated with UV radiation. From 1962 to 1971, the US military sprayed an estimated 18 million gallons of herbicides, including AO, over the fields and forests of Vietnam. Those herbicides have since been connected to numerous health issues, including cancer, though evidence of a relationship between AO and skin cancers has been weak.
Vietnam War-era veterans have a higher melanoma burden than the general population, with the disease being the fourth-most common cancer among those who served. AM, however, is rare, representing about 2% to 3% of all melanomas.
In a nested case-control study, Hwang et al used US Department of Veterans Affairs (VA) health care system data, including the VA Cancer Registry. The authors compared 1292 patients with AM and 2 pair-matched control groups: a group matched 4:1 to nonacral cutaneous melanoma controls, and a group without a melanoma diagnosis.
Hwang et al found AO exposure was associated with increased odds of AM compared with each control group. In an accompanying editorial, Andrew Olshan, PhD, from Department of Epidemiology at the University of North Carolina Gillings School of Global Public Health, wrote, “The magnitude of the effects was modest (about 30%) but noteworthy.”
A limitation of the study was that presumptive AOE status was based on whether the veteran filed a disability claim with evidence of officially recognized service in a period and place where Agent Orange was used—not on an assessment of the veteran’s individual AOE potential, including level of exposure. Because melanoma has never been included on the VA list of cancers presumed to be related to AO exposure, veterans do not automatically gain benefits by filing AOE claims after diagnosis. Even so, Olshan says, the reported study findings may underestimate the true effect of AO exposure on the risk of AM.
Given the rarity of AM, the association (if causal) would translate to 0.4 to 0.8 new annual cases of AM per 1,000,000 veterans, according to the study. Narrowed down to Vietnam War-era veterans—who are dwindling in number—the attributable cases would be scarce.
Nevertheless, the search for a better understanding of a potential link between AOE and melanomas among Vietnam War-era veterans is important, Olshan wrote.
“The Hwang et al study provides a strong impetus to further these research goals and contribute to the investigation of the legacy of the Vietnam War and honor a commitment to the veterans community.”
Recent research has revealed potential links between Agent Orange (AO) exposure and risk of acral melanoma (AM) among Vietnam War-era veterans, providing strong evidence of a relationship between the chemical and this type of cancer.
Localized to the palms, soles, and nail units, AM is a melanoma subtype less associated with UV radiation. From 1962 to 1971, the US military sprayed an estimated 18 million gallons of herbicides, including AO, over the fields and forests of Vietnam. Those herbicides have since been connected to numerous health issues, including cancer, though evidence of a relationship between AO and skin cancers has been weak.
Vietnam War-era veterans have a higher melanoma burden than the general population, with the disease being the fourth-most common cancer among those who served. AM, however, is rare, representing about 2% to 3% of all melanomas.
In a nested case-control study, Hwang et al used US Department of Veterans Affairs (VA) health care system data, including the VA Cancer Registry. The authors compared 1292 patients with AM and 2 pair-matched control groups: a group matched 4:1 to nonacral cutaneous melanoma controls, and a group without a melanoma diagnosis.
Hwang et al found AO exposure was associated with increased odds of AM compared with each control group. In an accompanying editorial, Andrew Olshan, PhD, from Department of Epidemiology at the University of North Carolina Gillings School of Global Public Health, wrote, “The magnitude of the effects was modest (about 30%) but noteworthy.”
A limitation of the study was that presumptive AOE status was based on whether the veteran filed a disability claim with evidence of officially recognized service in a period and place where Agent Orange was used—not on an assessment of the veteran’s individual AOE potential, including level of exposure. Because melanoma has never been included on the VA list of cancers presumed to be related to AO exposure, veterans do not automatically gain benefits by filing AOE claims after diagnosis. Even so, Olshan says, the reported study findings may underestimate the true effect of AO exposure on the risk of AM.
Given the rarity of AM, the association (if causal) would translate to 0.4 to 0.8 new annual cases of AM per 1,000,000 veterans, according to the study. Narrowed down to Vietnam War-era veterans—who are dwindling in number—the attributable cases would be scarce.
Nevertheless, the search for a better understanding of a potential link between AOE and melanomas among Vietnam War-era veterans is important, Olshan wrote.
“The Hwang et al study provides a strong impetus to further these research goals and contribute to the investigation of the legacy of the Vietnam War and honor a commitment to the veterans community.”
Can Fecal Transplants Enhance Immunotherapy? New Evidence and Cautions
Can Fecal Transplants Enhance Immunotherapy? New Evidence and Cautions
A trio of new studies, published simultaneously in February in Nature Medicine, add to growing evidence that manipulating the gut microbiome may enhance responses to immunotherapy in selected patients with cancer.
In these small, early-phase studies involving patients with metastatic renal cell carcinoma (RCC), non-small cell lung cancer (NSCLC), and melanoma receiving immune checkpoint inhibitor (ICI) therapy, fecal microbiota transplantation (FMT) was associated with objective response rates that compared favorably with historical or prespecified benchmarks.
The idea that microbiome modulation via FMT “can augment immunotherapy efficacy is probably a good one and these studies certainly support that hypothesis,” said Diwakar Davar, MD, assistant professor of medicine and an oncologist/hematologist at the University of Pittsburgh, Pennsylvania, who wasn’t part of the new work.While “an intriguing approach and certainly worthy of further evaluation,” Davar cautioned that the latest studies are not robust enough to answer the question conclusively.
Although ICIs have improved outcomes for patients with melanoma, NSCLC, and RCC, many patients still do not respond or eventually develop resistance. A growing body of evidence suggests that the gut microbiome can influence the effectiveness of ICI therapy. However, much of this evidence comes from preclinical studies showing that modulating the microbiome via FMT can alter responses to immunotherapy, along with small proof-of-concept human studies — predominantly in melanoma — suggesting this approach may help overcome primary or acquired resistance to anti-PD-1 therapy.
The new studies aimed to build on this foundation by exploring whether FMT could improve ICI responses and clinical outcomes in patients with NSCLC, melanoma, and RCC.
In the phase 2, open-label FMT-LUMINate trial, researchers tested a healthy-donor FMT delivered as oral capsules before patients began immunotherapy. FMT capsules were produced using 80-100 g of feces per dose from screened healthy donors, and patients consumed 30-40 capsules while under supervision. The study included 20 patients with NSCLC and high PD-L1 tumor expression receiving FMT before standard first-line pembrolizumab monotherapy and 20 patients with cutaneous melanoma receiving FMT before ipilimumab plus nivolumab.
In the NSCLC cohort, 16 patients (80%) achieved an objective response. The 80% objective response rate exceeded the prespecified efficacy threshold of 64% and was higher than previously described historical data, which ranged from 39% to 46%, the study team noted.
In the melanoma cohort, FMT before nivolumab and ipilimumab yielded an objective response rate of 75%, also exceeding the historical expected response rates of 50% to 58% among patients receiving this ICI combination.
In patients with NSCLC, no grade 3 or higher adverse events were reported. However, grade 3 or higher adverse events were reported in 13 (65%) patients in the melanoma group, suggesting a potentially accelerated onset of immune-related adverse events. Researchers also observed a higher-than-expected frequency of myocarditis in melanoma patients (15%). These toxicities clustered among patients who had FMT donors enriched in Prevotella spp, highlighting the importance of donor selection for future trials, the researchers explained.
The team plans to assess the potential of FMT to overcome primary resistance to ICI as part of the phase 2 CanBiome2 randomized trial, which aims to enroll 128 patients.
The RCC Data
The other two studies focused on FMT in patients with metastatic RCC. In the phase 1 PERFORM study, 20 treatment-naive patients with metastatic RCC added encapsulated healthy-donor FMT to standard ICI-based regimens — most commonly ipilimumab plus nivolumab, with some patients receiving pembrolizumab plus axitinib or pembrolizumab plus lenvatinib.
The primary endpoint was safety defined by the incidence and severity of immune-related adverse events. The safety endpoint was met; 50% of patients (10 of 20) experienced grade 3 immune-related adverse events, and there were no serious FMT-related toxicities and no grade 4 or 5 events.
Among 18 evaluable patients, nine (50%) achieved an objective response, including two who had complete responses (11%). Notably, most treatment responders did not develop any grade 3 or higher immune-related adverse events, the researchers reported.
Finally, in the phase 2a TACITO trial, 45 patients with treatment-naive metastatic RCC were randomly allocated to receive donor FMT or placebo FMT. Patients received three administrations over 6 months — first via colonoscopy then as capsulized doses, alongside pembrolizumab plus axitinib.
The primary endpoint of 12-month progression-free survival narrowly missed statistical significance — 70% vs 41% (P = .053) — but suggested a benefit in the donor FMT group.
“We need more than 1 year to appreciate statistical significance in terms of progression-free survival,” study investigator Gianluca Ianiro, MD, PhD, with Catholic University of the Sacred Heart, Rome, told Medscape Medical News.
As for secondary endpoints, median progression-free survival was significantly longer with donor FMT (24.0 vs 9.0 months; hazard ratio, 0.50; P = .035) and the objective response rate was higher with donor FMT (52% vs 32%).
Why Might FMT Boost ICI Response?
Conceptually, FMT is intended to reshape the gut ecosystem in ways that favor antitumor immunity, and possibly reduce immune dysregulation.
Across these new studies, the mechanistic story is moving beyond the idea that more diversity is good and toward a model that suggests a benefit to removing or suppressing taxa associated with resistance or inflammatory toxicity.
For example, in the TACITO trial, microbiome analysis confirmed that acquisition or loss of specific bacterial strains was associated with 12-month progression-free survival.
Additionally, results of the FMT-LUMINate trial hinted that the therapeutic benefit of FMT may be driven by eliminating harmful bacteria present at baseline, most notably Enterocloster, Clostridium and Streptococcus spp.
“This bacterial depletion was associated with a favorable immunometabolic milieu,” the FMT-LUMINate researchers wrote. Additionally, the results suggest that “failure to eliminate baseline deleterious taxa may sustain an immunosuppressive metabolic and systemic immune milieu that compromises ICI responses.”
Is FMT Ready for Prime Time?
Ianiro told Medscape Medical News he “definitely” thinks microbiome modulation could eventually become part of standard immunotherapy regimens.
Although the “signal” of benefit is clearly there, Davar cautioned that it’s too early to justify routine, off-trial use of FMT specifically to improve ICI response.
“These remain small, proof-of-concept studies. They are not adequately powered trials of fecal transplants and multiple different covariates haven’t been considered,” Davar said.
The study researchers noted that issues around donor selection and availability, dosing schedules, product standardization, and safety risk stratification need to be resolved.
For example, TACITO’s real-world experience shows logistics can matter. Delays occurred due to capsule unavailability and other scheduling barriers, which led to late dosing and missed or shifted treatments in some patients.
That’s a reminder that scaling FMT for oncology would require robust manufacturing, distribution, and time-sensitive coordination with ICI start dates.
More broadly, “whether FMT is the most suitable method of essentially changing the gut microbiome remains unclear,” explained Davar, who suggested that engineered microbiome therapeutics or tailored therapies may be a preferable, more scalable and tailored long-term solution.
Overall, does this new research provide impetus to develop stool banks? “Probably not,” Davar said.
But is it a call for interested parties to think about clinical trials and experimental products that could influence the gut microbiome? “Those are all probably good ideas,” he said.
The PERFORM, TACITO and FMT-LUMINate trials had no commercial funding. Saman Maleki Vareki, PhD, of the PERFORM trial, is a cofounder of LND Therapeutics Inc and has submitted a US patent application related to FMT donor screening. Ianiro has received personal fees for acting as a speaker for Biocodex and Illumina and for acting as a consultant/advisor for Ferring Therapeutics. Arielle Elkrief, MD, of the FMT-LUMINate trial, has received honoraria from AstraZeneca, Merck, Bristol Myers Squibb, and EMD Serono; consulting fees from EverImmune, NECBio, and Sanofi-Pasteur; and is an inventor on a patent regarding the microbiome and immunotherapy response. Davar had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A trio of new studies, published simultaneously in February in Nature Medicine, add to growing evidence that manipulating the gut microbiome may enhance responses to immunotherapy in selected patients with cancer.
In these small, early-phase studies involving patients with metastatic renal cell carcinoma (RCC), non-small cell lung cancer (NSCLC), and melanoma receiving immune checkpoint inhibitor (ICI) therapy, fecal microbiota transplantation (FMT) was associated with objective response rates that compared favorably with historical or prespecified benchmarks.
The idea that microbiome modulation via FMT “can augment immunotherapy efficacy is probably a good one and these studies certainly support that hypothesis,” said Diwakar Davar, MD, assistant professor of medicine and an oncologist/hematologist at the University of Pittsburgh, Pennsylvania, who wasn’t part of the new work.While “an intriguing approach and certainly worthy of further evaluation,” Davar cautioned that the latest studies are not robust enough to answer the question conclusively.
Although ICIs have improved outcomes for patients with melanoma, NSCLC, and RCC, many patients still do not respond or eventually develop resistance. A growing body of evidence suggests that the gut microbiome can influence the effectiveness of ICI therapy. However, much of this evidence comes from preclinical studies showing that modulating the microbiome via FMT can alter responses to immunotherapy, along with small proof-of-concept human studies — predominantly in melanoma — suggesting this approach may help overcome primary or acquired resistance to anti-PD-1 therapy.
The new studies aimed to build on this foundation by exploring whether FMT could improve ICI responses and clinical outcomes in patients with NSCLC, melanoma, and RCC.
In the phase 2, open-label FMT-LUMINate trial, researchers tested a healthy-donor FMT delivered as oral capsules before patients began immunotherapy. FMT capsules were produced using 80-100 g of feces per dose from screened healthy donors, and patients consumed 30-40 capsules while under supervision. The study included 20 patients with NSCLC and high PD-L1 tumor expression receiving FMT before standard first-line pembrolizumab monotherapy and 20 patients with cutaneous melanoma receiving FMT before ipilimumab plus nivolumab.
In the NSCLC cohort, 16 patients (80%) achieved an objective response. The 80% objective response rate exceeded the prespecified efficacy threshold of 64% and was higher than previously described historical data, which ranged from 39% to 46%, the study team noted.
In the melanoma cohort, FMT before nivolumab and ipilimumab yielded an objective response rate of 75%, also exceeding the historical expected response rates of 50% to 58% among patients receiving this ICI combination.
In patients with NSCLC, no grade 3 or higher adverse events were reported. However, grade 3 or higher adverse events were reported in 13 (65%) patients in the melanoma group, suggesting a potentially accelerated onset of immune-related adverse events. Researchers also observed a higher-than-expected frequency of myocarditis in melanoma patients (15%). These toxicities clustered among patients who had FMT donors enriched in Prevotella spp, highlighting the importance of donor selection for future trials, the researchers explained.
The team plans to assess the potential of FMT to overcome primary resistance to ICI as part of the phase 2 CanBiome2 randomized trial, which aims to enroll 128 patients.
The RCC Data
The other two studies focused on FMT in patients with metastatic RCC. In the phase 1 PERFORM study, 20 treatment-naive patients with metastatic RCC added encapsulated healthy-donor FMT to standard ICI-based regimens — most commonly ipilimumab plus nivolumab, with some patients receiving pembrolizumab plus axitinib or pembrolizumab plus lenvatinib.
The primary endpoint was safety defined by the incidence and severity of immune-related adverse events. The safety endpoint was met; 50% of patients (10 of 20) experienced grade 3 immune-related adverse events, and there were no serious FMT-related toxicities and no grade 4 or 5 events.
Among 18 evaluable patients, nine (50%) achieved an objective response, including two who had complete responses (11%). Notably, most treatment responders did not develop any grade 3 or higher immune-related adverse events, the researchers reported.
Finally, in the phase 2a TACITO trial, 45 patients with treatment-naive metastatic RCC were randomly allocated to receive donor FMT or placebo FMT. Patients received three administrations over 6 months — first via colonoscopy then as capsulized doses, alongside pembrolizumab plus axitinib.
The primary endpoint of 12-month progression-free survival narrowly missed statistical significance — 70% vs 41% (P = .053) — but suggested a benefit in the donor FMT group.
“We need more than 1 year to appreciate statistical significance in terms of progression-free survival,” study investigator Gianluca Ianiro, MD, PhD, with Catholic University of the Sacred Heart, Rome, told Medscape Medical News.
As for secondary endpoints, median progression-free survival was significantly longer with donor FMT (24.0 vs 9.0 months; hazard ratio, 0.50; P = .035) and the objective response rate was higher with donor FMT (52% vs 32%).
Why Might FMT Boost ICI Response?
Conceptually, FMT is intended to reshape the gut ecosystem in ways that favor antitumor immunity, and possibly reduce immune dysregulation.
Across these new studies, the mechanistic story is moving beyond the idea that more diversity is good and toward a model that suggests a benefit to removing or suppressing taxa associated with resistance or inflammatory toxicity.
For example, in the TACITO trial, microbiome analysis confirmed that acquisition or loss of specific bacterial strains was associated with 12-month progression-free survival.
Additionally, results of the FMT-LUMINate trial hinted that the therapeutic benefit of FMT may be driven by eliminating harmful bacteria present at baseline, most notably Enterocloster, Clostridium and Streptococcus spp.
“This bacterial depletion was associated with a favorable immunometabolic milieu,” the FMT-LUMINate researchers wrote. Additionally, the results suggest that “failure to eliminate baseline deleterious taxa may sustain an immunosuppressive metabolic and systemic immune milieu that compromises ICI responses.”
Is FMT Ready for Prime Time?
Ianiro told Medscape Medical News he “definitely” thinks microbiome modulation could eventually become part of standard immunotherapy regimens.
Although the “signal” of benefit is clearly there, Davar cautioned that it’s too early to justify routine, off-trial use of FMT specifically to improve ICI response.
“These remain small, proof-of-concept studies. They are not adequately powered trials of fecal transplants and multiple different covariates haven’t been considered,” Davar said.
The study researchers noted that issues around donor selection and availability, dosing schedules, product standardization, and safety risk stratification need to be resolved.
For example, TACITO’s real-world experience shows logistics can matter. Delays occurred due to capsule unavailability and other scheduling barriers, which led to late dosing and missed or shifted treatments in some patients.
That’s a reminder that scaling FMT for oncology would require robust manufacturing, distribution, and time-sensitive coordination with ICI start dates.
More broadly, “whether FMT is the most suitable method of essentially changing the gut microbiome remains unclear,” explained Davar, who suggested that engineered microbiome therapeutics or tailored therapies may be a preferable, more scalable and tailored long-term solution.
Overall, does this new research provide impetus to develop stool banks? “Probably not,” Davar said.
But is it a call for interested parties to think about clinical trials and experimental products that could influence the gut microbiome? “Those are all probably good ideas,” he said.
The PERFORM, TACITO and FMT-LUMINate trials had no commercial funding. Saman Maleki Vareki, PhD, of the PERFORM trial, is a cofounder of LND Therapeutics Inc and has submitted a US patent application related to FMT donor screening. Ianiro has received personal fees for acting as a speaker for Biocodex and Illumina and for acting as a consultant/advisor for Ferring Therapeutics. Arielle Elkrief, MD, of the FMT-LUMINate trial, has received honoraria from AstraZeneca, Merck, Bristol Myers Squibb, and EMD Serono; consulting fees from EverImmune, NECBio, and Sanofi-Pasteur; and is an inventor on a patent regarding the microbiome and immunotherapy response. Davar had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A trio of new studies, published simultaneously in February in Nature Medicine, add to growing evidence that manipulating the gut microbiome may enhance responses to immunotherapy in selected patients with cancer.
In these small, early-phase studies involving patients with metastatic renal cell carcinoma (RCC), non-small cell lung cancer (NSCLC), and melanoma receiving immune checkpoint inhibitor (ICI) therapy, fecal microbiota transplantation (FMT) was associated with objective response rates that compared favorably with historical or prespecified benchmarks.
The idea that microbiome modulation via FMT “can augment immunotherapy efficacy is probably a good one and these studies certainly support that hypothesis,” said Diwakar Davar, MD, assistant professor of medicine and an oncologist/hematologist at the University of Pittsburgh, Pennsylvania, who wasn’t part of the new work.While “an intriguing approach and certainly worthy of further evaluation,” Davar cautioned that the latest studies are not robust enough to answer the question conclusively.
Although ICIs have improved outcomes for patients with melanoma, NSCLC, and RCC, many patients still do not respond or eventually develop resistance. A growing body of evidence suggests that the gut microbiome can influence the effectiveness of ICI therapy. However, much of this evidence comes from preclinical studies showing that modulating the microbiome via FMT can alter responses to immunotherapy, along with small proof-of-concept human studies — predominantly in melanoma — suggesting this approach may help overcome primary or acquired resistance to anti-PD-1 therapy.
The new studies aimed to build on this foundation by exploring whether FMT could improve ICI responses and clinical outcomes in patients with NSCLC, melanoma, and RCC.
In the phase 2, open-label FMT-LUMINate trial, researchers tested a healthy-donor FMT delivered as oral capsules before patients began immunotherapy. FMT capsules were produced using 80-100 g of feces per dose from screened healthy donors, and patients consumed 30-40 capsules while under supervision. The study included 20 patients with NSCLC and high PD-L1 tumor expression receiving FMT before standard first-line pembrolizumab monotherapy and 20 patients with cutaneous melanoma receiving FMT before ipilimumab plus nivolumab.
In the NSCLC cohort, 16 patients (80%) achieved an objective response. The 80% objective response rate exceeded the prespecified efficacy threshold of 64% and was higher than previously described historical data, which ranged from 39% to 46%, the study team noted.
In the melanoma cohort, FMT before nivolumab and ipilimumab yielded an objective response rate of 75%, also exceeding the historical expected response rates of 50% to 58% among patients receiving this ICI combination.
In patients with NSCLC, no grade 3 or higher adverse events were reported. However, grade 3 or higher adverse events were reported in 13 (65%) patients in the melanoma group, suggesting a potentially accelerated onset of immune-related adverse events. Researchers also observed a higher-than-expected frequency of myocarditis in melanoma patients (15%). These toxicities clustered among patients who had FMT donors enriched in Prevotella spp, highlighting the importance of donor selection for future trials, the researchers explained.
The team plans to assess the potential of FMT to overcome primary resistance to ICI as part of the phase 2 CanBiome2 randomized trial, which aims to enroll 128 patients.
The RCC Data
The other two studies focused on FMT in patients with metastatic RCC. In the phase 1 PERFORM study, 20 treatment-naive patients with metastatic RCC added encapsulated healthy-donor FMT to standard ICI-based regimens — most commonly ipilimumab plus nivolumab, with some patients receiving pembrolizumab plus axitinib or pembrolizumab plus lenvatinib.
The primary endpoint was safety defined by the incidence and severity of immune-related adverse events. The safety endpoint was met; 50% of patients (10 of 20) experienced grade 3 immune-related adverse events, and there were no serious FMT-related toxicities and no grade 4 or 5 events.
Among 18 evaluable patients, nine (50%) achieved an objective response, including two who had complete responses (11%). Notably, most treatment responders did not develop any grade 3 or higher immune-related adverse events, the researchers reported.
Finally, in the phase 2a TACITO trial, 45 patients with treatment-naive metastatic RCC were randomly allocated to receive donor FMT or placebo FMT. Patients received three administrations over 6 months — first via colonoscopy then as capsulized doses, alongside pembrolizumab plus axitinib.
The primary endpoint of 12-month progression-free survival narrowly missed statistical significance — 70% vs 41% (P = .053) — but suggested a benefit in the donor FMT group.
“We need more than 1 year to appreciate statistical significance in terms of progression-free survival,” study investigator Gianluca Ianiro, MD, PhD, with Catholic University of the Sacred Heart, Rome, told Medscape Medical News.
As for secondary endpoints, median progression-free survival was significantly longer with donor FMT (24.0 vs 9.0 months; hazard ratio, 0.50; P = .035) and the objective response rate was higher with donor FMT (52% vs 32%).
Why Might FMT Boost ICI Response?
Conceptually, FMT is intended to reshape the gut ecosystem in ways that favor antitumor immunity, and possibly reduce immune dysregulation.
Across these new studies, the mechanistic story is moving beyond the idea that more diversity is good and toward a model that suggests a benefit to removing or suppressing taxa associated with resistance or inflammatory toxicity.
For example, in the TACITO trial, microbiome analysis confirmed that acquisition or loss of specific bacterial strains was associated with 12-month progression-free survival.
Additionally, results of the FMT-LUMINate trial hinted that the therapeutic benefit of FMT may be driven by eliminating harmful bacteria present at baseline, most notably Enterocloster, Clostridium and Streptococcus spp.
“This bacterial depletion was associated with a favorable immunometabolic milieu,” the FMT-LUMINate researchers wrote. Additionally, the results suggest that “failure to eliminate baseline deleterious taxa may sustain an immunosuppressive metabolic and systemic immune milieu that compromises ICI responses.”
Is FMT Ready for Prime Time?
Ianiro told Medscape Medical News he “definitely” thinks microbiome modulation could eventually become part of standard immunotherapy regimens.
Although the “signal” of benefit is clearly there, Davar cautioned that it’s too early to justify routine, off-trial use of FMT specifically to improve ICI response.
“These remain small, proof-of-concept studies. They are not adequately powered trials of fecal transplants and multiple different covariates haven’t been considered,” Davar said.
The study researchers noted that issues around donor selection and availability, dosing schedules, product standardization, and safety risk stratification need to be resolved.
For example, TACITO’s real-world experience shows logistics can matter. Delays occurred due to capsule unavailability and other scheduling barriers, which led to late dosing and missed or shifted treatments in some patients.
That’s a reminder that scaling FMT for oncology would require robust manufacturing, distribution, and time-sensitive coordination with ICI start dates.
More broadly, “whether FMT is the most suitable method of essentially changing the gut microbiome remains unclear,” explained Davar, who suggested that engineered microbiome therapeutics or tailored therapies may be a preferable, more scalable and tailored long-term solution.
Overall, does this new research provide impetus to develop stool banks? “Probably not,” Davar said.
But is it a call for interested parties to think about clinical trials and experimental products that could influence the gut microbiome? “Those are all probably good ideas,” he said.
The PERFORM, TACITO and FMT-LUMINate trials had no commercial funding. Saman Maleki Vareki, PhD, of the PERFORM trial, is a cofounder of LND Therapeutics Inc and has submitted a US patent application related to FMT donor screening. Ianiro has received personal fees for acting as a speaker for Biocodex and Illumina and for acting as a consultant/advisor for Ferring Therapeutics. Arielle Elkrief, MD, of the FMT-LUMINate trial, has received honoraria from AstraZeneca, Merck, Bristol Myers Squibb, and EMD Serono; consulting fees from EverImmune, NECBio, and Sanofi-Pasteur; and is an inventor on a patent regarding the microbiome and immunotherapy response. Davar had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Can Fecal Transplants Enhance Immunotherapy? New Evidence and Cautions
Can Fecal Transplants Enhance Immunotherapy? New Evidence and Cautions




