User login
Children with uncontrolled asthma at higher risk of being bullied
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
The risk of bullying and teasing is higher in children and young people with poorer asthma control, an international study reported. Published online in the Archives of Disease in Childhood, the Room to Breathe survey of 943 children in six countries found 9.9% had experienced asthma-related bullying or teasing (n = 93).
Children with well-controlled disease, however, were less likely to report being victimized by asthma-related bullying/teasing: odds ratio, 0.51; 95% confidence interval, 0.23-0.84; P = .006).
“It’s important for pediatricians to recognize that children and young people with asthma commonly report bullying or teasing as a result of their condition,” Will Carroll, MD, of the Paediatric Respiratory Service at Staffordshire Children’s Hospital at Royal Stoke, Stoke-on-Trent, England, told this news organization. “Pediatricians should talk to children themselves with asthma about this and not just their parents, and efforts should be made to improve asthma control whenever possible.”
Though common and potentially long-lasting in its effects, bullying is rarely addressed by health care professionals, the U.K. authors said.
But things may differ in the United States. According to Mark Welles, MD, a pediatrician at Cohen Children’s Medical Center at Northwell Health in Queen’s, N.Y., and regional cochair of the American Academy of Pediatrics antibullying committee, young doctors here are trained to ask about bullying when seeing a child, no matter what the reason for the visit. “It’s important to build a rapport with the child, and you need to ask about the disease they may have but also generally ask, ‘How are things at school? Is everyone nice to you?’ It is becoming more common practice to ask this,” said Dr. Welles, who was not involved with the U.K. research.
The U.K. study drew on unpublished data from the Room to Breathe survey conducted by Dr. Carroll’s group during 2008-2009 in Canada, the United Kingdom, Greece, Hungary, South Africa, and the Netherlands. Only 358 of 930 (38.5%) children were found to be well controlled according to current Global Initiative for Asthma symptom-control criteria.
The analysis also found a highly significant association (P < .0001) between Childhood Asthma Control Test (C-ACT) score and reported bullying/teasing, with bullied children having lower scores. C-ACT–defined controlled asthma scores of 20 or higher were significantly associated with a lower risk of bullying (OR, 0.46; 95% CI, 0.28-0.76; P = .001).
In other study findings, harassment was more common in children whose asthma was serious enough to entail activity restriction (OR, 1.74; 95% CI, 1.11-2.75; P = .010) and who described their asthma as “bad” (OR, 3.02; 95% CI, 1.86-4.85; P < .001), as well as those whose parents reported ongoing asthma-related health worries (OR, 1.64; 95% CI, 1.04-2.58; P = .024).
“When a child is clearly different from others, such as having bad asthma or being limited in activities due to asthma, they stand out more and are more frequently bullied,” said Tracy Evian Waasdorp, PhD, MSEd, director of research for school-based bullying and social-emotional learning at Children’s Hospital of Philadelphia, and also not a participant in the U.K. study.
In contrast to the 10% bullying rate in Dr. Carroll’s study, Dr. Waasdorp referred to a CHOP analysis of more than 64,000 youth from a Northeastern state in which those with asthma were 40% more likely to be victims of in-person bullying and 70% were more likely to be cyberbullied than youth without asthma. “Having a medical condition can therefore put you at risk of being bullied regardless of what country you live in,” she said.
CHOP policy encourages practitioners to routinely ask about bullying and to provide handouts and resources for parents, she added.
Interestingly, the U.K. investigators found that open public use of spacers was not associated with asthma-related bullying, nor was parental worry at diagnosis or parental concern about steroid use.
But according to Dr. Welles, “Kids may be using the inhaler in front of other kids, and they may be embarrassed and not want to be seen as different. So they may not use the inhaler when needed for gym class or sports, forcing them to sit out and then potentially be bullied again. It’s a vicious cycle.”
Previous research has identified the bullying and teasing of children with food allergies.
Behaviors have included allergy-specific harassment such as smearing peanut butter on a youngster’s forehead or putting peanut butter cookie crumbs in a child’s lunch box.
“In our survey we asked the question ‘Have you been teased or bullied because of your asthma?’ but we didn’t ask what form this took,” Dr. Carroll said. “But we were surprised at just how many children said yes. It’s time for more research, I think.”
“There are never enough studies around this,” added Dr. Welles. “Bullying, whether because of asthma or otherwise, has the potential for long-term effects well into adulthood.”
In the meantime, asthma consultations should incorporate specific questions about bullying. They should also be child focused in order to gain a representative appreciation of asthma control and its effect on the child’s life.
“As pediatricians, we need to be continuously supporting parents and find the help they need to address any mental health issues,” Dr. Welles said. “Every pediatrician and parent needs to be aware and recognize when something is different in their child’s life. Please don’t ignore it.”
Dr. Waasdorp stressed that school and other communities should be aware that children with asthma may be at increased risk for aggression and harmful interactions related to their asthma. “Programming to reduce bullying should focus broadly on shifting the climate so that bullying is not perceived to be normative and on improving ‘upstander,’ or positive bystander, responses.” she said.
The original survey was funded by Nycomed (Zurich). No additional funding was requested for the current analysis. Dr. Carroll reported personal fees from GlaxoSmithKline, Novartis, and Trudell Medical International outside the submitted work. Dr. Welles and Dr. Waasdorp disclosed no competing interests relevant to their comments.
FROM ARCHIVES OF DISEASE IN CHILDHOOD
Ten changes that could keep clinicians in the workforce in a pandemic
Indeed, a recent poll of 1,000 health care workers conducted Sept. 2-8 by Morning Consult, showed that 18% of medical workers polled quit their jobs during the pandemic. Additionally, 31% said they had at least thought about leaving their work.
“As physicians, educators, peers and friends of COVID-19 responders, we are gravely concerned about our colleagues’ exhaustion, burnout, and disillusionment,” wrote lead author Eileen Barrett, MD, and coauthors of the new action plan, which was published in the Annals of Internal Medicine.
The 10-point, one-page checklist includes providing “practical support in the areas that clinicians identify as causing emotional stress or moral injury,” such as managing anger and grief when patients have chosen not to be vaccinated or confronting misinformation.
“Those are the things that are making people’s mental health worse” psychiatrist Jessi Gold, MD, MS, said in an interview. “I don’t think I’ve seen that mentioned other places.”
Among the other action items are:
- Reduce administrative tasks that are not “mission critical,” such as mandatory training that has no evidence of improving patient outcomes and meetings that could be skipped.
- Offer free and confidential resources to support clinicians’ mental health, such as easy access to crisis hotlines and peer support groups.
- Maintain transparency about personal protective equipment and contingency plans when there are shortages to restore trust.
- Encourage clinicians to use vacation time; leaders should model this.
- Implement suicide prevention strategies, including wellness check-ins for clinicians in hard-hit areas.
The action plan was based on the authors’ own experiences and the stories of colleagues and information in literature. It includes 10 changes health care leaders could make to help retain providers who may be on the brink of leaving their jobs or leaving medicine
Action items intended to be easily achievable, low cost
Dr. Barrett, who is a hospitalist in Albuquerque, said the goal was to present easily achievable and low-cost action items that clinicians and health care leaders could use as a starting point when change seems insurmountable and evidence on what works is slow to come.
She said one of the things that spurred her to coauthor the list was becoming aware of other clinicians’ “secret shame” in thinking about leaving medicine.
“Maybe a person who is not being listened to could take this journal article and say ‘we don’t know where to start. It looks like we can start here,’ ” said Dr. Barrett, who is also an associate professor in the division of hospital medicine, department of internal medicine, at the University of New Mexico, Albuquerque.
She noted that some of the good ideas floated around did not make the list, because they required daunting budget commitments and too much time to put into place.
Numerous other proposed solutions were of the wrong tone, according to Dr. Barrett.
“It’s not just about a hug or a piece of pizza,” she said.
Dr. Gold, who is an assistant professor at Washington University, St. Louis, and specializes in the mental health of health care workers, noted that, even though the list was pared to 10 action items, it is still hard for health care organizations to prioritize mental health.
“Many hospitals are still struggling with the active bleed of the pandemic and financially recovering,” she said. “If you’re dealing with a full ER and people are still dying of COVID and you don’t have the resources to support them, it’s really hard to then find magic money to deal with mental health. I’d love for that to be true.”
Every organization, however, can start with removing questions about mental and physical health diagnoses from credentialing and employment applications, which is one of the items on the list, she said.
“It’s the lowest-bar thing that you can fix for making people in crisis not fear getting help,” she said. That change must come on a state-by-state and individual hospital level.
Favorable reactions to list
Dr. Barrett, who also serves on the editorial advisory board of Internal Medicine News, said the reactions to the checklist have been “overwhelmingly favorable and appreciative.”
Eric J. Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News, tweeted about this list: “For COVID-19, more than ever before, it’s vital to keep clinicians in the U.S. health care workforce. These are 10 steps that will help.” The tweet was retweeted more than 100 times.
Lotte Dyrbye, MD, MHPE, a primary care physician and codirector of the program on physician well-being at the Mayo Clinic in Rochester, Minn., said in an interview that managing the anger around patients who choose to be unvaccinated is critical and something that has gotten little notice since the vaccines became available.
“Physicians and nurses are working extremely hard and seeing a lot of suffering and are taking care of patients very sick with COVID-19, knowing they had access to the vaccine. That is causing anger and frustration. We haven’t prepared health care workers to deal with that,” she said.
Outside expert: Not all items may be easy to implement
Dr. Dyrbye said that, though she found adding time to address COVID misinformation questions in appointments is very important, it may be wishful thinking.
The authors suggested training other members of the care team to answer those questions to free up time, but she said, for patients who have been swayed by misinformation, hearing information from someone other than the physician they have a relationship with won’t be convincing.
According to Dr. Dyrbye, the items on the list are not easy to implement, but the action plan is worthwhile to consider adopting as a multipronged approach.
“Most of these things are hard and we need to be in it for the long run,” she said.
The need is clear for efforts to address the mental health of not just experienced clinicians but those in training as well, she noted.
Related research
A study that was also recently published in the Annals of Internal Medicine suggested that making a few simple changes can help improve the mental health of residents. The research, which included nearly 17,000 first-year residents who started training between 2007 and 2019, addressed indicators of mental health in light of interventions such as limiting residents’ work hours and providing more services.
The investigators found that, though depression remains high among residents, depressive symptoms among first-year residents dropped 24.4% from 2007 to 2019 in parallel with four main factors: an increase in mental health services; restrictions on work hours for residents; more sleep hours; and higher-quality feedback from faculty.
Dr. Barrett said she hopes her colleagues and health care workers everywhere will find some solace in seeing that the new checklist she coauthored was published in a prominent journal.
The message Dr. Barrett said she hopes they see is: “Someone is validating it is not in their head. They are validating we can do better. They are validating that we must.”
Dr. Barrett and coauthors had no conflicts of interest. Dr. Gold and Dr. Dyrbye also disclosed having no relevant financial relationships.
Indeed, a recent poll of 1,000 health care workers conducted Sept. 2-8 by Morning Consult, showed that 18% of medical workers polled quit their jobs during the pandemic. Additionally, 31% said they had at least thought about leaving their work.
“As physicians, educators, peers and friends of COVID-19 responders, we are gravely concerned about our colleagues’ exhaustion, burnout, and disillusionment,” wrote lead author Eileen Barrett, MD, and coauthors of the new action plan, which was published in the Annals of Internal Medicine.
The 10-point, one-page checklist includes providing “practical support in the areas that clinicians identify as causing emotional stress or moral injury,” such as managing anger and grief when patients have chosen not to be vaccinated or confronting misinformation.
“Those are the things that are making people’s mental health worse” psychiatrist Jessi Gold, MD, MS, said in an interview. “I don’t think I’ve seen that mentioned other places.”
Among the other action items are:
- Reduce administrative tasks that are not “mission critical,” such as mandatory training that has no evidence of improving patient outcomes and meetings that could be skipped.
- Offer free and confidential resources to support clinicians’ mental health, such as easy access to crisis hotlines and peer support groups.
- Maintain transparency about personal protective equipment and contingency plans when there are shortages to restore trust.
- Encourage clinicians to use vacation time; leaders should model this.
- Implement suicide prevention strategies, including wellness check-ins for clinicians in hard-hit areas.
The action plan was based on the authors’ own experiences and the stories of colleagues and information in literature. It includes 10 changes health care leaders could make to help retain providers who may be on the brink of leaving their jobs or leaving medicine
Action items intended to be easily achievable, low cost
Dr. Barrett, who is a hospitalist in Albuquerque, said the goal was to present easily achievable and low-cost action items that clinicians and health care leaders could use as a starting point when change seems insurmountable and evidence on what works is slow to come.
She said one of the things that spurred her to coauthor the list was becoming aware of other clinicians’ “secret shame” in thinking about leaving medicine.
“Maybe a person who is not being listened to could take this journal article and say ‘we don’t know where to start. It looks like we can start here,’ ” said Dr. Barrett, who is also an associate professor in the division of hospital medicine, department of internal medicine, at the University of New Mexico, Albuquerque.
She noted that some of the good ideas floated around did not make the list, because they required daunting budget commitments and too much time to put into place.
Numerous other proposed solutions were of the wrong tone, according to Dr. Barrett.
“It’s not just about a hug or a piece of pizza,” she said.
Dr. Gold, who is an assistant professor at Washington University, St. Louis, and specializes in the mental health of health care workers, noted that, even though the list was pared to 10 action items, it is still hard for health care organizations to prioritize mental health.
“Many hospitals are still struggling with the active bleed of the pandemic and financially recovering,” she said. “If you’re dealing with a full ER and people are still dying of COVID and you don’t have the resources to support them, it’s really hard to then find magic money to deal with mental health. I’d love for that to be true.”
Every organization, however, can start with removing questions about mental and physical health diagnoses from credentialing and employment applications, which is one of the items on the list, she said.
“It’s the lowest-bar thing that you can fix for making people in crisis not fear getting help,” she said. That change must come on a state-by-state and individual hospital level.
Favorable reactions to list
Dr. Barrett, who also serves on the editorial advisory board of Internal Medicine News, said the reactions to the checklist have been “overwhelmingly favorable and appreciative.”
Eric J. Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News, tweeted about this list: “For COVID-19, more than ever before, it’s vital to keep clinicians in the U.S. health care workforce. These are 10 steps that will help.” The tweet was retweeted more than 100 times.
Lotte Dyrbye, MD, MHPE, a primary care physician and codirector of the program on physician well-being at the Mayo Clinic in Rochester, Minn., said in an interview that managing the anger around patients who choose to be unvaccinated is critical and something that has gotten little notice since the vaccines became available.
“Physicians and nurses are working extremely hard and seeing a lot of suffering and are taking care of patients very sick with COVID-19, knowing they had access to the vaccine. That is causing anger and frustration. We haven’t prepared health care workers to deal with that,” she said.
Outside expert: Not all items may be easy to implement
Dr. Dyrbye said that, though she found adding time to address COVID misinformation questions in appointments is very important, it may be wishful thinking.
The authors suggested training other members of the care team to answer those questions to free up time, but she said, for patients who have been swayed by misinformation, hearing information from someone other than the physician they have a relationship with won’t be convincing.
According to Dr. Dyrbye, the items on the list are not easy to implement, but the action plan is worthwhile to consider adopting as a multipronged approach.
“Most of these things are hard and we need to be in it for the long run,” she said.
The need is clear for efforts to address the mental health of not just experienced clinicians but those in training as well, she noted.
Related research
A study that was also recently published in the Annals of Internal Medicine suggested that making a few simple changes can help improve the mental health of residents. The research, which included nearly 17,000 first-year residents who started training between 2007 and 2019, addressed indicators of mental health in light of interventions such as limiting residents’ work hours and providing more services.
The investigators found that, though depression remains high among residents, depressive symptoms among first-year residents dropped 24.4% from 2007 to 2019 in parallel with four main factors: an increase in mental health services; restrictions on work hours for residents; more sleep hours; and higher-quality feedback from faculty.
Dr. Barrett said she hopes her colleagues and health care workers everywhere will find some solace in seeing that the new checklist she coauthored was published in a prominent journal.
The message Dr. Barrett said she hopes they see is: “Someone is validating it is not in their head. They are validating we can do better. They are validating that we must.”
Dr. Barrett and coauthors had no conflicts of interest. Dr. Gold and Dr. Dyrbye also disclosed having no relevant financial relationships.
Indeed, a recent poll of 1,000 health care workers conducted Sept. 2-8 by Morning Consult, showed that 18% of medical workers polled quit their jobs during the pandemic. Additionally, 31% said they had at least thought about leaving their work.
“As physicians, educators, peers and friends of COVID-19 responders, we are gravely concerned about our colleagues’ exhaustion, burnout, and disillusionment,” wrote lead author Eileen Barrett, MD, and coauthors of the new action plan, which was published in the Annals of Internal Medicine.
The 10-point, one-page checklist includes providing “practical support in the areas that clinicians identify as causing emotional stress or moral injury,” such as managing anger and grief when patients have chosen not to be vaccinated or confronting misinformation.
“Those are the things that are making people’s mental health worse” psychiatrist Jessi Gold, MD, MS, said in an interview. “I don’t think I’ve seen that mentioned other places.”
Among the other action items are:
- Reduce administrative tasks that are not “mission critical,” such as mandatory training that has no evidence of improving patient outcomes and meetings that could be skipped.
- Offer free and confidential resources to support clinicians’ mental health, such as easy access to crisis hotlines and peer support groups.
- Maintain transparency about personal protective equipment and contingency plans when there are shortages to restore trust.
- Encourage clinicians to use vacation time; leaders should model this.
- Implement suicide prevention strategies, including wellness check-ins for clinicians in hard-hit areas.
The action plan was based on the authors’ own experiences and the stories of colleagues and information in literature. It includes 10 changes health care leaders could make to help retain providers who may be on the brink of leaving their jobs or leaving medicine
Action items intended to be easily achievable, low cost
Dr. Barrett, who is a hospitalist in Albuquerque, said the goal was to present easily achievable and low-cost action items that clinicians and health care leaders could use as a starting point when change seems insurmountable and evidence on what works is slow to come.
She said one of the things that spurred her to coauthor the list was becoming aware of other clinicians’ “secret shame” in thinking about leaving medicine.
“Maybe a person who is not being listened to could take this journal article and say ‘we don’t know where to start. It looks like we can start here,’ ” said Dr. Barrett, who is also an associate professor in the division of hospital medicine, department of internal medicine, at the University of New Mexico, Albuquerque.
She noted that some of the good ideas floated around did not make the list, because they required daunting budget commitments and too much time to put into place.
Numerous other proposed solutions were of the wrong tone, according to Dr. Barrett.
“It’s not just about a hug or a piece of pizza,” she said.
Dr. Gold, who is an assistant professor at Washington University, St. Louis, and specializes in the mental health of health care workers, noted that, even though the list was pared to 10 action items, it is still hard for health care organizations to prioritize mental health.
“Many hospitals are still struggling with the active bleed of the pandemic and financially recovering,” she said. “If you’re dealing with a full ER and people are still dying of COVID and you don’t have the resources to support them, it’s really hard to then find magic money to deal with mental health. I’d love for that to be true.”
Every organization, however, can start with removing questions about mental and physical health diagnoses from credentialing and employment applications, which is one of the items on the list, she said.
“It’s the lowest-bar thing that you can fix for making people in crisis not fear getting help,” she said. That change must come on a state-by-state and individual hospital level.
Favorable reactions to list
Dr. Barrett, who also serves on the editorial advisory board of Internal Medicine News, said the reactions to the checklist have been “overwhelmingly favorable and appreciative.”
Eric J. Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News, tweeted about this list: “For COVID-19, more than ever before, it’s vital to keep clinicians in the U.S. health care workforce. These are 10 steps that will help.” The tweet was retweeted more than 100 times.
Lotte Dyrbye, MD, MHPE, a primary care physician and codirector of the program on physician well-being at the Mayo Clinic in Rochester, Minn., said in an interview that managing the anger around patients who choose to be unvaccinated is critical and something that has gotten little notice since the vaccines became available.
“Physicians and nurses are working extremely hard and seeing a lot of suffering and are taking care of patients very sick with COVID-19, knowing they had access to the vaccine. That is causing anger and frustration. We haven’t prepared health care workers to deal with that,” she said.
Outside expert: Not all items may be easy to implement
Dr. Dyrbye said that, though she found adding time to address COVID misinformation questions in appointments is very important, it may be wishful thinking.
The authors suggested training other members of the care team to answer those questions to free up time, but she said, for patients who have been swayed by misinformation, hearing information from someone other than the physician they have a relationship with won’t be convincing.
According to Dr. Dyrbye, the items on the list are not easy to implement, but the action plan is worthwhile to consider adopting as a multipronged approach.
“Most of these things are hard and we need to be in it for the long run,” she said.
The need is clear for efforts to address the mental health of not just experienced clinicians but those in training as well, she noted.
Related research
A study that was also recently published in the Annals of Internal Medicine suggested that making a few simple changes can help improve the mental health of residents. The research, which included nearly 17,000 first-year residents who started training between 2007 and 2019, addressed indicators of mental health in light of interventions such as limiting residents’ work hours and providing more services.
The investigators found that, though depression remains high among residents, depressive symptoms among first-year residents dropped 24.4% from 2007 to 2019 in parallel with four main factors: an increase in mental health services; restrictions on work hours for residents; more sleep hours; and higher-quality feedback from faculty.
Dr. Barrett said she hopes her colleagues and health care workers everywhere will find some solace in seeing that the new checklist she coauthored was published in a prominent journal.
The message Dr. Barrett said she hopes they see is: “Someone is validating it is not in their head. They are validating we can do better. They are validating that we must.”
Dr. Barrett and coauthors had no conflicts of interest. Dr. Gold and Dr. Dyrbye also disclosed having no relevant financial relationships.
FROM ANNALS OF INTERNAL MEDICINE
‘I Can Go Anywhere’: How Service Dogs Help Veterans With PTSD
It was supper time in the Whittier, California, home of Air Force veteran Danyelle Clark-Gutierrez, and eagerly awaiting a bowl of kibble and canned dog food was Lisa, a 3-year-old yellow Labrador retriever.
Her nails clicking on the kitchen floor as she danced about, Lisa looked more like an exuberant puppy than the highly trained service animal that helps Clark-Gutierrez manage the symptoms of post-traumatic stress disorder.
“Having her now, it’s like I can go anywhere,” Clark-Gutierrez said. “And, yes, if somebody did come at me, I’d have warning — I could run.”
A growing body of research into PTSD and service animals paved the way for President Joe Biden to sign into law the Puppies Assisting Wounded Servicemembers (PAWS) for Veterans Therapy Act. The legislation, enacted in August, requires the Department of Veterans Affairs to open its service dog referral program to veterans with PTSD and to launch a five-year pilot program in which veterans with PTSD train service dogs for other veterans.
Clark-Gutierrez, 33, is among the 25 percent of female veterans who have reported experiencing military sexual trauma while serving in the U.S. armed services.
Military sexual trauma, combat violence and brain injuries are some of the experiences that increase the risk that service members will develop PTSD. Symptoms include flashbacks to the traumatic event, severe anxiety, nightmares and hypervigilance — all normal reactions to experiencing or witnessing violence, according to psychologists. Someone receives a PTSD diagnosis when symptoms worsen or remain for months or years.
That’s what Clark-Gutierrez said happened to her after ongoing sexual harassment by a fellow airman escalated to a physical attack about a decade ago. A lawyer with three children, she said that to feel safe leaving her home she needed her husband by her side. After diagnosing Clark-Gutierrez with PTSD, doctors at VA hospitals prescribed a cascade of medications for her. At one point, Clark-Gutierrez said, her prescriptions added up to more than a dozen pills a day.
“I had medication, and then I had medication for the two or three side effects for each medication,” she said. “And every time they gave me a new med, they had to give me three more. I just couldn’t do it anymore. I was just getting so tired. So we started looking at other therapies.”
And that’s how she got her service dog, Lisa. Clark-Gutierrez’s husband, also an Air Force veteran, discovered the nonprofit group K9s for Warriors, which rescues dogs — many from kill shelters — and trains them to be service animals for veterans with PTSD. Lisa is one of about 700 dogs the group has paired with veterans dealing with symptoms caused by traumatic experiences.
“Now with Lisa we take bike rides, we go down to the park, we go to Home Depot,” said Clark-Gutierrez. “I go grocery shopping — normal-people things that I get to do that I didn’t get to do before Lisa.”
That comes as no surprise to Maggie O’Haire, an associate professor of human-animal interaction at Purdue University. Her research suggests that while service dogs aren’t necessarily a cure for PTSD, they do ease its symptoms. Among her published studies is one showing that veterans partnered with these dogs experience less anger and anxiety and get better sleep than those without a service dog. Another of her studies suggests that service dogs lower cortisol levels in veterans who have been traumatized.
“We actually saw patterns of that stress hormone that were more similar to healthy adults who don’t have post-traumatic stress disorder,” O’Haire said.
A congressionally mandated VA study that focuses on service dogs’ impact on veterans with PTSD and was published this year suggests that those partnered with the animals experience less suicidal ideation and more improvement to their symptoms than those without them.
Until now, the federal dog referral program — which relies on nonprofit service dog organizations to pay for the dogs and to provide them to veterans for free — required that participating veterans have a physical mobility issue, such as a lost limb, paralysis or blindness. Veterans like Clark-Gutierrez who have PTSD but no physical disability were on their own in arranging for a service dog.
The pilot program created by the new federal law will give veterans with PTSD the chance to train mental health service dogs for other veterans. It’s modeled on a program at the VA hospital in Palo Alto, California, and will be offered at five VA medical centers nationwide in partnership with accredited service dog training organizations.
“This bill is really about therapeutic, on-the-job training, or ‘training the trainer,’” said Adam Webb, a spokesperson for Sen. Thom Tillis (R-N.C.), who introduced the legislation in the Senate. “We don’t anticipate VA will start prescribing PTSD service dogs, but the data we generate from this pilot program will likely be useful in making that case in the future.”
The Congressional Budget Office estimates the pilot program will cost the VA about $19 million. The law stops short of requiring the VA to pay for the dogs. Instead, the agency will partner with accredited service dog organizations that use private money to cover the cost of adopting, training and pairing the dogs with veterans.
Still, the law represents a welcome about-face in VA policy, said Rory Diamond, CEO of K9s for Warriors.
“For the last 10 years, the VA has essentially told us that they don’t recognize service dogs as helping a veteran with post-traumatic stress,” Diamond said.
PTSD service dogs are often confused with emotional support dogs, Diamond said. The latter provide companionship and are not trained to support someone with a disability. PTSD service dogs cost about $25,000 to adopt and train, he said.
Diamond explained that the command “cover” means “the dog will sit next to the warrior, look behind them and alert them if someone comes up from behind.” The command “block” means the dog will “stand perpendicular and give them some space from whatever’s in front of them.”
Retired Army Master Sgt. David Crenshaw of Kearny, New Jersey, said his service dog, Doc, has changed his life.
“We teach in the military to have a battle buddy,” Crenshaw said. “And these service animals act as a battle buddy.”
A few months ago, Crenshaw experienced this firsthand. He had generally avoided large gatherings because persistent hypervigilance is one symptom of his combat-caused PTSD. But this summer, Doc, a pointer and Labrador mix, helped Crenshaw navigate the crowds at Disney World — a significant first for Crenshaw and his family of five.
“I was not agitated. I was not anxious. I was not upset,” said Crenshaw, 39. “It was truly, truly amazing and so much so that I didn’t even have to even stop to think about it in the moment. It just happened naturally.”
Thanks to Doc, Crenshaw said, he no longer takes PTSD drugs or self-medicates with alcohol. Clark-Gutierrez said Lisa, too, has helped her quit using alcohol and stop taking VA-prescribed medications for panic attacks, nightmares and periods of disassociation.
The dogs actually save the VA money over time, Diamond said. “Our warriors are far less likely to be on expensive prescription drugs, are far less likely to use other VA services and far more likely to go to school or go to work. So it’s a win-win-win across the board.”
It was supper time in the Whittier, California, home of Air Force veteran Danyelle Clark-Gutierrez, and eagerly awaiting a bowl of kibble and canned dog food was Lisa, a 3-year-old yellow Labrador retriever.
Her nails clicking on the kitchen floor as she danced about, Lisa looked more like an exuberant puppy than the highly trained service animal that helps Clark-Gutierrez manage the symptoms of post-traumatic stress disorder.
“Having her now, it’s like I can go anywhere,” Clark-Gutierrez said. “And, yes, if somebody did come at me, I’d have warning — I could run.”
A growing body of research into PTSD and service animals paved the way for President Joe Biden to sign into law the Puppies Assisting Wounded Servicemembers (PAWS) for Veterans Therapy Act. The legislation, enacted in August, requires the Department of Veterans Affairs to open its service dog referral program to veterans with PTSD and to launch a five-year pilot program in which veterans with PTSD train service dogs for other veterans.
Clark-Gutierrez, 33, is among the 25 percent of female veterans who have reported experiencing military sexual trauma while serving in the U.S. armed services.
Military sexual trauma, combat violence and brain injuries are some of the experiences that increase the risk that service members will develop PTSD. Symptoms include flashbacks to the traumatic event, severe anxiety, nightmares and hypervigilance — all normal reactions to experiencing or witnessing violence, according to psychologists. Someone receives a PTSD diagnosis when symptoms worsen or remain for months or years.
That’s what Clark-Gutierrez said happened to her after ongoing sexual harassment by a fellow airman escalated to a physical attack about a decade ago. A lawyer with three children, she said that to feel safe leaving her home she needed her husband by her side. After diagnosing Clark-Gutierrez with PTSD, doctors at VA hospitals prescribed a cascade of medications for her. At one point, Clark-Gutierrez said, her prescriptions added up to more than a dozen pills a day.
“I had medication, and then I had medication for the two or three side effects for each medication,” she said. “And every time they gave me a new med, they had to give me three more. I just couldn’t do it anymore. I was just getting so tired. So we started looking at other therapies.”
And that’s how she got her service dog, Lisa. Clark-Gutierrez’s husband, also an Air Force veteran, discovered the nonprofit group K9s for Warriors, which rescues dogs — many from kill shelters — and trains them to be service animals for veterans with PTSD. Lisa is one of about 700 dogs the group has paired with veterans dealing with symptoms caused by traumatic experiences.
“Now with Lisa we take bike rides, we go down to the park, we go to Home Depot,” said Clark-Gutierrez. “I go grocery shopping — normal-people things that I get to do that I didn’t get to do before Lisa.”
That comes as no surprise to Maggie O’Haire, an associate professor of human-animal interaction at Purdue University. Her research suggests that while service dogs aren’t necessarily a cure for PTSD, they do ease its symptoms. Among her published studies is one showing that veterans partnered with these dogs experience less anger and anxiety and get better sleep than those without a service dog. Another of her studies suggests that service dogs lower cortisol levels in veterans who have been traumatized.
“We actually saw patterns of that stress hormone that were more similar to healthy adults who don’t have post-traumatic stress disorder,” O’Haire said.
A congressionally mandated VA study that focuses on service dogs’ impact on veterans with PTSD and was published this year suggests that those partnered with the animals experience less suicidal ideation and more improvement to their symptoms than those without them.
Until now, the federal dog referral program — which relies on nonprofit service dog organizations to pay for the dogs and to provide them to veterans for free — required that participating veterans have a physical mobility issue, such as a lost limb, paralysis or blindness. Veterans like Clark-Gutierrez who have PTSD but no physical disability were on their own in arranging for a service dog.
The pilot program created by the new federal law will give veterans with PTSD the chance to train mental health service dogs for other veterans. It’s modeled on a program at the VA hospital in Palo Alto, California, and will be offered at five VA medical centers nationwide in partnership with accredited service dog training organizations.
“This bill is really about therapeutic, on-the-job training, or ‘training the trainer,’” said Adam Webb, a spokesperson for Sen. Thom Tillis (R-N.C.), who introduced the legislation in the Senate. “We don’t anticipate VA will start prescribing PTSD service dogs, but the data we generate from this pilot program will likely be useful in making that case in the future.”
The Congressional Budget Office estimates the pilot program will cost the VA about $19 million. The law stops short of requiring the VA to pay for the dogs. Instead, the agency will partner with accredited service dog organizations that use private money to cover the cost of adopting, training and pairing the dogs with veterans.
Still, the law represents a welcome about-face in VA policy, said Rory Diamond, CEO of K9s for Warriors.
“For the last 10 years, the VA has essentially told us that they don’t recognize service dogs as helping a veteran with post-traumatic stress,” Diamond said.
PTSD service dogs are often confused with emotional support dogs, Diamond said. The latter provide companionship and are not trained to support someone with a disability. PTSD service dogs cost about $25,000 to adopt and train, he said.
Diamond explained that the command “cover” means “the dog will sit next to the warrior, look behind them and alert them if someone comes up from behind.” The command “block” means the dog will “stand perpendicular and give them some space from whatever’s in front of them.”
Retired Army Master Sgt. David Crenshaw of Kearny, New Jersey, said his service dog, Doc, has changed his life.
“We teach in the military to have a battle buddy,” Crenshaw said. “And these service animals act as a battle buddy.”
A few months ago, Crenshaw experienced this firsthand. He had generally avoided large gatherings because persistent hypervigilance is one symptom of his combat-caused PTSD. But this summer, Doc, a pointer and Labrador mix, helped Crenshaw navigate the crowds at Disney World — a significant first for Crenshaw and his family of five.
“I was not agitated. I was not anxious. I was not upset,” said Crenshaw, 39. “It was truly, truly amazing and so much so that I didn’t even have to even stop to think about it in the moment. It just happened naturally.”
Thanks to Doc, Crenshaw said, he no longer takes PTSD drugs or self-medicates with alcohol. Clark-Gutierrez said Lisa, too, has helped her quit using alcohol and stop taking VA-prescribed medications for panic attacks, nightmares and periods of disassociation.
The dogs actually save the VA money over time, Diamond said. “Our warriors are far less likely to be on expensive prescription drugs, are far less likely to use other VA services and far more likely to go to school or go to work. So it’s a win-win-win across the board.”
It was supper time in the Whittier, California, home of Air Force veteran Danyelle Clark-Gutierrez, and eagerly awaiting a bowl of kibble and canned dog food was Lisa, a 3-year-old yellow Labrador retriever.
Her nails clicking on the kitchen floor as she danced about, Lisa looked more like an exuberant puppy than the highly trained service animal that helps Clark-Gutierrez manage the symptoms of post-traumatic stress disorder.
“Having her now, it’s like I can go anywhere,” Clark-Gutierrez said. “And, yes, if somebody did come at me, I’d have warning — I could run.”
A growing body of research into PTSD and service animals paved the way for President Joe Biden to sign into law the Puppies Assisting Wounded Servicemembers (PAWS) for Veterans Therapy Act. The legislation, enacted in August, requires the Department of Veterans Affairs to open its service dog referral program to veterans with PTSD and to launch a five-year pilot program in which veterans with PTSD train service dogs for other veterans.
Clark-Gutierrez, 33, is among the 25 percent of female veterans who have reported experiencing military sexual trauma while serving in the U.S. armed services.
Military sexual trauma, combat violence and brain injuries are some of the experiences that increase the risk that service members will develop PTSD. Symptoms include flashbacks to the traumatic event, severe anxiety, nightmares and hypervigilance — all normal reactions to experiencing or witnessing violence, according to psychologists. Someone receives a PTSD diagnosis when symptoms worsen or remain for months or years.
That’s what Clark-Gutierrez said happened to her after ongoing sexual harassment by a fellow airman escalated to a physical attack about a decade ago. A lawyer with three children, she said that to feel safe leaving her home she needed her husband by her side. After diagnosing Clark-Gutierrez with PTSD, doctors at VA hospitals prescribed a cascade of medications for her. At one point, Clark-Gutierrez said, her prescriptions added up to more than a dozen pills a day.
“I had medication, and then I had medication for the two or three side effects for each medication,” she said. “And every time they gave me a new med, they had to give me three more. I just couldn’t do it anymore. I was just getting so tired. So we started looking at other therapies.”
And that’s how she got her service dog, Lisa. Clark-Gutierrez’s husband, also an Air Force veteran, discovered the nonprofit group K9s for Warriors, which rescues dogs — many from kill shelters — and trains them to be service animals for veterans with PTSD. Lisa is one of about 700 dogs the group has paired with veterans dealing with symptoms caused by traumatic experiences.
“Now with Lisa we take bike rides, we go down to the park, we go to Home Depot,” said Clark-Gutierrez. “I go grocery shopping — normal-people things that I get to do that I didn’t get to do before Lisa.”
That comes as no surprise to Maggie O’Haire, an associate professor of human-animal interaction at Purdue University. Her research suggests that while service dogs aren’t necessarily a cure for PTSD, they do ease its symptoms. Among her published studies is one showing that veterans partnered with these dogs experience less anger and anxiety and get better sleep than those without a service dog. Another of her studies suggests that service dogs lower cortisol levels in veterans who have been traumatized.
“We actually saw patterns of that stress hormone that were more similar to healthy adults who don’t have post-traumatic stress disorder,” O’Haire said.
A congressionally mandated VA study that focuses on service dogs’ impact on veterans with PTSD and was published this year suggests that those partnered with the animals experience less suicidal ideation and more improvement to their symptoms than those without them.
Until now, the federal dog referral program — which relies on nonprofit service dog organizations to pay for the dogs and to provide them to veterans for free — required that participating veterans have a physical mobility issue, such as a lost limb, paralysis or blindness. Veterans like Clark-Gutierrez who have PTSD but no physical disability were on their own in arranging for a service dog.
The pilot program created by the new federal law will give veterans with PTSD the chance to train mental health service dogs for other veterans. It’s modeled on a program at the VA hospital in Palo Alto, California, and will be offered at five VA medical centers nationwide in partnership with accredited service dog training organizations.
“This bill is really about therapeutic, on-the-job training, or ‘training the trainer,’” said Adam Webb, a spokesperson for Sen. Thom Tillis (R-N.C.), who introduced the legislation in the Senate. “We don’t anticipate VA will start prescribing PTSD service dogs, but the data we generate from this pilot program will likely be useful in making that case in the future.”
The Congressional Budget Office estimates the pilot program will cost the VA about $19 million. The law stops short of requiring the VA to pay for the dogs. Instead, the agency will partner with accredited service dog organizations that use private money to cover the cost of adopting, training and pairing the dogs with veterans.
Still, the law represents a welcome about-face in VA policy, said Rory Diamond, CEO of K9s for Warriors.
“For the last 10 years, the VA has essentially told us that they don’t recognize service dogs as helping a veteran with post-traumatic stress,” Diamond said.
PTSD service dogs are often confused with emotional support dogs, Diamond said. The latter provide companionship and are not trained to support someone with a disability. PTSD service dogs cost about $25,000 to adopt and train, he said.
Diamond explained that the command “cover” means “the dog will sit next to the warrior, look behind them and alert them if someone comes up from behind.” The command “block” means the dog will “stand perpendicular and give them some space from whatever’s in front of them.”
Retired Army Master Sgt. David Crenshaw of Kearny, New Jersey, said his service dog, Doc, has changed his life.
“We teach in the military to have a battle buddy,” Crenshaw said. “And these service animals act as a battle buddy.”
A few months ago, Crenshaw experienced this firsthand. He had generally avoided large gatherings because persistent hypervigilance is one symptom of his combat-caused PTSD. But this summer, Doc, a pointer and Labrador mix, helped Crenshaw navigate the crowds at Disney World — a significant first for Crenshaw and his family of five.
“I was not agitated. I was not anxious. I was not upset,” said Crenshaw, 39. “It was truly, truly amazing and so much so that I didn’t even have to even stop to think about it in the moment. It just happened naturally.”
Thanks to Doc, Crenshaw said, he no longer takes PTSD drugs or self-medicates with alcohol. Clark-Gutierrez said Lisa, too, has helped her quit using alcohol and stop taking VA-prescribed medications for panic attacks, nightmares and periods of disassociation.
The dogs actually save the VA money over time, Diamond said. “Our warriors are far less likely to be on expensive prescription drugs, are far less likely to use other VA services and far more likely to go to school or go to work. So it’s a win-win-win across the board.”
We physicians must pull together as a knowledge community
The COVID-19 pandemic is a biosocial phenomenon. Patients and doctors alike find themselves assigned to groups designated as responsible and wise, or selfish and irrational, based strictly upon their personal assessments of medical risk. This trend in our culture is represented by threats of disciplinary action issued by medical regulators against physicians who are perceived to be undermining the public health message by spreading “misinformation.”
Our review of the literature reveals many references to “misinformation” but no definition narrow and precise enough to be interpreted consistently in a disciplinary environment. More pressing, this ambiguous word’s use is correlated with negative meaning and innuendo, often discrediting valuable information a priori without actual data points.
The most basic definition available is Merriam Webster’s: “incorrect or misleading information.” This definition includes no point of reference against which competing scientific claims can be measured.
Claudia E. Haupt, PhD, a political scientist and law professor, articulates a useful framework for understanding the relationship between medicine and state regulators. In the Yale Law Journal, Dr. Haupt wrote: “Knowledge communities have specialized expertise and are closest to those affected; they must have the freedom to work things out for themselves. The professions as knowledge communities have a fundamental interest in not having the state (or anyone else, for that matter) corrupt or distort what amounts to the state of the art in their respective fields.”
Injecting the artificial term “misinformation” into the science information ecosystem obfuscates and impedes the very ability of this vital knowledge community to perform its raison d’être. , rather than attending to healing or promoting progress.
Time has certainly shown us that science is anything but settled on all things COVID. If the scientific community accepts disrespect as the response of choice to difference of opinion and practice, we lose the trust in one another as colleagues; we need to keep scientific inquiry and exploration alive. Curiosity, equanimity, and tolerance are key components of the professional attitude as we deftly maneuver against the virus together.
In the face of deadly disease, it is especially imperative that intelligent, thoughtful, highly respected scientists, researchers, and physicians have room to safely share their knowledge and clinical experience. The Association of American Physicians and Surgeons has published a statement on scientific integrity that can be used as a measuring stick for claims about misinformation in medicine. We call on physicians to pull together as a knowledge community. Kindness and respect for patients starts with kindness and respect for one another as colleagues.
Dr. Kohanski is in private practice in Somerset, N.J., and is a diplomate of the American Board of Psychiatry & Neurology. She disclosed no relevant financial relationships. Dr. Emmons is part-time clinical associate professor in the department of psychiatry at the University of Vermont, Burlington, and is a past chair of the Ethics Committee for the Vermont District Branch of the American Psychiatric Association. He is in private practice in Moretown, Vt., and disclosed no relevant financial relationships.
The COVID-19 pandemic is a biosocial phenomenon. Patients and doctors alike find themselves assigned to groups designated as responsible and wise, or selfish and irrational, based strictly upon their personal assessments of medical risk. This trend in our culture is represented by threats of disciplinary action issued by medical regulators against physicians who are perceived to be undermining the public health message by spreading “misinformation.”
Our review of the literature reveals many references to “misinformation” but no definition narrow and precise enough to be interpreted consistently in a disciplinary environment. More pressing, this ambiguous word’s use is correlated with negative meaning and innuendo, often discrediting valuable information a priori without actual data points.
The most basic definition available is Merriam Webster’s: “incorrect or misleading information.” This definition includes no point of reference against which competing scientific claims can be measured.
Claudia E. Haupt, PhD, a political scientist and law professor, articulates a useful framework for understanding the relationship between medicine and state regulators. In the Yale Law Journal, Dr. Haupt wrote: “Knowledge communities have specialized expertise and are closest to those affected; they must have the freedom to work things out for themselves. The professions as knowledge communities have a fundamental interest in not having the state (or anyone else, for that matter) corrupt or distort what amounts to the state of the art in their respective fields.”
Injecting the artificial term “misinformation” into the science information ecosystem obfuscates and impedes the very ability of this vital knowledge community to perform its raison d’être. , rather than attending to healing or promoting progress.
Time has certainly shown us that science is anything but settled on all things COVID. If the scientific community accepts disrespect as the response of choice to difference of opinion and practice, we lose the trust in one another as colleagues; we need to keep scientific inquiry and exploration alive. Curiosity, equanimity, and tolerance are key components of the professional attitude as we deftly maneuver against the virus together.
In the face of deadly disease, it is especially imperative that intelligent, thoughtful, highly respected scientists, researchers, and physicians have room to safely share their knowledge and clinical experience. The Association of American Physicians and Surgeons has published a statement on scientific integrity that can be used as a measuring stick for claims about misinformation in medicine. We call on physicians to pull together as a knowledge community. Kindness and respect for patients starts with kindness and respect for one another as colleagues.
Dr. Kohanski is in private practice in Somerset, N.J., and is a diplomate of the American Board of Psychiatry & Neurology. She disclosed no relevant financial relationships. Dr. Emmons is part-time clinical associate professor in the department of psychiatry at the University of Vermont, Burlington, and is a past chair of the Ethics Committee for the Vermont District Branch of the American Psychiatric Association. He is in private practice in Moretown, Vt., and disclosed no relevant financial relationships.
The COVID-19 pandemic is a biosocial phenomenon. Patients and doctors alike find themselves assigned to groups designated as responsible and wise, or selfish and irrational, based strictly upon their personal assessments of medical risk. This trend in our culture is represented by threats of disciplinary action issued by medical regulators against physicians who are perceived to be undermining the public health message by spreading “misinformation.”
Our review of the literature reveals many references to “misinformation” but no definition narrow and precise enough to be interpreted consistently in a disciplinary environment. More pressing, this ambiguous word’s use is correlated with negative meaning and innuendo, often discrediting valuable information a priori without actual data points.
The most basic definition available is Merriam Webster’s: “incorrect or misleading information.” This definition includes no point of reference against which competing scientific claims can be measured.
Claudia E. Haupt, PhD, a political scientist and law professor, articulates a useful framework for understanding the relationship between medicine and state regulators. In the Yale Law Journal, Dr. Haupt wrote: “Knowledge communities have specialized expertise and are closest to those affected; they must have the freedom to work things out for themselves. The professions as knowledge communities have a fundamental interest in not having the state (or anyone else, for that matter) corrupt or distort what amounts to the state of the art in their respective fields.”
Injecting the artificial term “misinformation” into the science information ecosystem obfuscates and impedes the very ability of this vital knowledge community to perform its raison d’être. , rather than attending to healing or promoting progress.
Time has certainly shown us that science is anything but settled on all things COVID. If the scientific community accepts disrespect as the response of choice to difference of opinion and practice, we lose the trust in one another as colleagues; we need to keep scientific inquiry and exploration alive. Curiosity, equanimity, and tolerance are key components of the professional attitude as we deftly maneuver against the virus together.
In the face of deadly disease, it is especially imperative that intelligent, thoughtful, highly respected scientists, researchers, and physicians have room to safely share their knowledge and clinical experience. The Association of American Physicians and Surgeons has published a statement on scientific integrity that can be used as a measuring stick for claims about misinformation in medicine. We call on physicians to pull together as a knowledge community. Kindness and respect for patients starts with kindness and respect for one another as colleagues.
Dr. Kohanski is in private practice in Somerset, N.J., and is a diplomate of the American Board of Psychiatry & Neurology. She disclosed no relevant financial relationships. Dr. Emmons is part-time clinical associate professor in the department of psychiatry at the University of Vermont, Burlington, and is a past chair of the Ethics Committee for the Vermont District Branch of the American Psychiatric Association. He is in private practice in Moretown, Vt., and disclosed no relevant financial relationships.
CBT prevents depression in up to 50% of patients with insomnia
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
Cognitive-behavioral therapy (CBT) is linked to a significantly reduced risk of depression in patients with insomnia, new research shows.
Insomnia affects over 50% of older adults, and insomnia contributes to a twofold greater risk for major depression, investigators noted.
“We show that by treating insomnia with a simple behavioral approach called Cognitive Behavioral Therapy for Insomnia, or CBT-I, you can reduce the likelihood of developing depression by over 50%,” lead author Michael R. Irwin, MD, Cousins Distinguished Professor of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, said in an interview.
The study is unique in that the treatment “is not just reducing depression, it’s preventing depression,” Dr. Irwin added.
The findings were published online Nov. 24 in JAMA Psychiatry.
Primary outcome met
within the previous 12 months.
All were randomly assigned to receive either CBT-I or Sleep Education Therapy (SET).
CBT-I is a first-line treatment for insomnia that includes five components: cognitive therapy targeting dysfunctional thoughts about sleep, stimulus control, sleep restriction, sleep hygiene, and relaxation.
SET provides information on behavioral and environmental factors contributing to poor sleep. While sleep education provides tips on improving sleep, CBT-I helps patients implement those changes and behaviors, Dr. Irwin noted.
Both interventions were delivered by trained personnel in weekly 120-minute group sessions for 2 months, consistent with the format and duration of most CBT-I trials.
The primary outcome was time to incident or recurrent major depressive disorder as diagnosed by the Structured Clinical Interview of the DSM-5 every 6 months during 36 months of follow-up. A monthly Patient Health Questionnaire 9 (PHQ-9) was used to screen for depressive symptoms.
Results showed depression occurred in 12.2% of the CBT-I group versus 25.9% of the SET group. The hazard ratio (HR) for depression in the CBT-I group compared with the SET group was 0.51 (95% confidence interval, 0.29-0.88; P = .02). The number needed to treat to prevent incident or recurrent depression was 7.3.
After adjustment for factors affecting depression risk such as sex, educational level, income, comorbidity, and history of depression, the HR for depression in the CBT-I group versus the SET group was 0.45 (95% CI, 0.23-0.86; P = .02).
Treatment with CBT-I yielded an annual 4.1% incidence of depression, which is similar to the population rate and half the rate in SET, which was 8.6%.
‘Remission is key’
The secondary outcome was sustained remission of insomnia disorder. The investigators found a greater proportion of the CBT-I group than the SET group achieved remission after treatment (50.7% vs. 37.7%; 95% CI, 0.10-0.93; P = .02).
“Remission is really key to the benefits that we’re seeing,” said Dr. Irwin.
Inflammation may explain why insomnia raises the risk for depression, he noted. “We know sleep disturbance can lead to inflammation and we also know inflammation can produce depression,” Dr. Irwin said.
It is also possible insomnia leads to an impaired pleasure or reward system, which is linked to depression, he added.
The authors noted that because insomnia is associated with suicidal ideation and dementia, CBT-I may reduce risk for suicide or cognitive decline.
While 8-week CBT-I treatments are readily available, “unfortunately, most clinicians will prescribe medications,” said Dr. Irwin. He noted that in older adults, drugs are linked to adverse events such as falls and cognitive problems.
These new results “really argue that psychology and psychiatry need to be fully integrated into what we call collaborative care models,” Dr. Irwin said.
There were no adverse events during treatment, and none of the serious events that occurred during follow-up were attributed to the trial.
Convincing argument?
Commenting on the findings for this news organization, Philip R. Muskin, MD, professor of psychiatry at Columbia University Irving Medical Center, New York, said the study was “nicely written” and the authors put forward “a very convincing argument” for CBT-I to prevent depression.
“It’s eye opening in that it’s a robust study; it’s carefully done; subjects were followed for a long period of time, and it’s an accessible treatment,” said Dr. Muskin, who was not involved with the research.
The study also shows “it’s possible to intervene in something we know is a risk factor in elderly people,” he added. “We think of older people as being less malleable to these kinds of things, but they’re not. They clearly participated, and there wasn’t a huge dropout rate.”
Dr. Muskin noted that less than half of the older participants were married or had a partner. He would have liked more information on this status because being widowed or divorced, as well as when this life change occurred, could affect vulnerability to depression.
The authors of an accompanying editorial called the study “seminal,” and noted that insomnia treatment possibly preventing depressive disorders is a “major finding.”
Proving this preventive strategy is effective in older adults will be important because “insomnia and depression are highly prevalent in this population and the uptake of both preventive and treatment services is low,” wrote Pim Cuijpers, PhD, department of clinical, neuro, and developmental psychology, Amsterdam Public Health Research Institute, and Charles F. Reynolds III, MD, department of psychiatry, University of Pittsburgh.
If the reduced rates of depression observed in the study could be generalized to the total population with insomnia, “the incidence of major depression could be reduced considerably,” they wrote.
“Can we prevent depression through interventions aimed at procrastination in college students, interventions aimed at perfectionism in perinatal women, stress management training for employees, social skills training in adolescents?” they asked.
This approach to preventing depressive disorders “offers all kinds of new opportunities to develop and test indirect interventions” for problems that are significantly associated with the onset of depression, the editorialists wrote.
The study was funded by a grant from the National Institute on Aging to the University of California, which partially supported the authors’ salaries. Dr. Irwin, Dr. Muskin, and Dr. Cuijpers have reported no relevant financial relationships. Dr. Reynolds reported being coinventor of the Pittsburgh Sleep Quality Index, for which he receives royalties.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
New tool guides nutrition counseling in schizophrenia patients
A new tool designed by psychiatrists to help guide nutritional counseling in patients with schizophrenia spectrum disorders (SSD) has been released.
The worksheet and clinician guide were developed using results from a recent scoping review on the relationship between diet and mental health in patients with SSD, and a feedback process involving a focus group with psychiatrists and individuals who had lived experience with psychosis.
“Mental health clinicians already have the training to help our patients make behavioral changes,” lead author Laura LaChance, MD, lecturer, department of psychiatry, and a psychiatrist at St. Mary’s Hospital Centre, McGill University, Montreal, said in an interview.
“We work every day with patients to help them to reduce their substance use, improve their sleep, take medications, etc., and nutrition should be added to the radar [since] eating well for mental health is part of self-care and can be included in mental health treatment plans,” she said.
The paper was published online Nov. 10 in BMC Psychiatry.
Nutrition frequently ignored
Dr. LaChance noted that “nutrition is largely absent from mental health training programs and often ignored in clinical practice.”
The investigators “wanted to create a tool to help incorporate basic nutritional counseling into the care of individuals with severe mental illness.” They wanted the tool “to be simple enough to understand for patients and simple enough to use for mental health care professionals who don’t have any official nutrition training.”
The team developed a worksheet that includes dietary recommendations, the majority of which are supported by the scoping review and consistent with Canada’s Food Guide. The review “identified all of the published literature related to the relationship between diet and psychiatric symptoms of SSD,” synthesizing the results of 822 prior articles.
and does not contradict generally accepted recommendations for weight management. It is suitable for all patients including those with low or normal body mass index and provides psychoeducation about the importance of quality nutrition as a determinant of mental health.
Positive tone
The worksheet was informed by social cognitive theory, which “highlights the important role of goal setting and behavior contracting, reinforcement, self-control, social norms, attitudes, and self-efficacy.”
It provides “basic education about important nutrition principles” as well as “very simple recommendations to increase knowledge about healthy eating” and “actionable tips for individuals to incorporate.” The researchers used a “positive” tone and included motivational interviewing questions.
“Delivery of the intervention by the patient’s mental health care provider is by design, in an attempt to address the widely held misbelief that nutrition intervention is of limited importance to mental health care and begin to change norms,” Dr. LaChance said.
The worksheet addresses monetary barriers to healthy eating; offers practical tips to “increase perceived control and self-efficacy”; is written in simple, accessible, nontechnical language; and includes foods from a range of cultural backgrounds.
To ensure that the worksheet and clinical guide met the needs of the target population, the researchers conducted a focus group with five psychiatrists and individual phone interviews with people who live with psychosis (n = 6).
Participants with psychosis were evenly divided between male and female and six age groups were represented: younger than 20 years; 21-30 years; 31-40 years; 41-50 years; 51-50 years; and older than 60 years. Of these participants, half scored in the “limited literacy” range, based on a nutritional literacy assessment tool (the Newest Vital Sign [NVS]).
A revised version of the worksheet, taking participants’ feedback into account, was mailed to all participants, who then provided further feedback on the revised version.
‘Unspoken area’
The clinician guide contains not only an overview and a suggested agenda to steer discussion, but also a sample visual representation of the recommended relative proportions of different food categories in an ideal meal as well as sample meals, a budgeting discussion, and a list of goals.
A closing statement encourages the clinician to “keep the messaging positive, celebrate small victories, and provide encouragement.”
Specific dietary recommendations include choosing complex carbohydrates and healthy fats, reducing highly processed foods and sugar, adding vegetables and fruits to meals and snacks, and eating protein-rich foods throughout the day.
A “noteworthy theme” that emerged in discussions with psychiatrists as well as participants with SSD was “the lack of nutrition training in medical education and psychiatric residency and the general absence of nutritional counseling in this field of medicine.”
One participant described nutrition as “definitely an unspoken area” in schizophrenia – especially in institutional settings, where “you are overloaded with sugars, not healthy grain, not complex grain. You get white bread sandwiches, shitty juice.”
Powerful tool
Commenting on the paper for this news organization, Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and a nutrition educator at Harvard Medical School, both in Boston, said she appreciates that this paper “is seeking methods to expand treatment options for those with SSD and improve provider understanding/knowledge of therapeutic foods.”
She called the pilot evaluation “notably small,” but added that it “provides results to suggest that scaling this worksheet/guide may hold promise to better provide nutritional counseling to those with psychiatric illness.”
Dr. Naidoo, also a chef and the author of “This Is Your Brain on Food,” who was not involved in the study said, “I’ve seen the power of food as medicine in my own hospital practice and do believe that food is one of the most powerful tools we have in supporting mental fitness and emotional well-being.”
The project was funded by the Canadian CAM Research Fund. Dr. LaChance and Dr. Naidoo have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new tool designed by psychiatrists to help guide nutritional counseling in patients with schizophrenia spectrum disorders (SSD) has been released.
The worksheet and clinician guide were developed using results from a recent scoping review on the relationship between diet and mental health in patients with SSD, and a feedback process involving a focus group with psychiatrists and individuals who had lived experience with psychosis.
“Mental health clinicians already have the training to help our patients make behavioral changes,” lead author Laura LaChance, MD, lecturer, department of psychiatry, and a psychiatrist at St. Mary’s Hospital Centre, McGill University, Montreal, said in an interview.
“We work every day with patients to help them to reduce their substance use, improve their sleep, take medications, etc., and nutrition should be added to the radar [since] eating well for mental health is part of self-care and can be included in mental health treatment plans,” she said.
The paper was published online Nov. 10 in BMC Psychiatry.
Nutrition frequently ignored
Dr. LaChance noted that “nutrition is largely absent from mental health training programs and often ignored in clinical practice.”
The investigators “wanted to create a tool to help incorporate basic nutritional counseling into the care of individuals with severe mental illness.” They wanted the tool “to be simple enough to understand for patients and simple enough to use for mental health care professionals who don’t have any official nutrition training.”
The team developed a worksheet that includes dietary recommendations, the majority of which are supported by the scoping review and consistent with Canada’s Food Guide. The review “identified all of the published literature related to the relationship between diet and psychiatric symptoms of SSD,” synthesizing the results of 822 prior articles.
and does not contradict generally accepted recommendations for weight management. It is suitable for all patients including those with low or normal body mass index and provides psychoeducation about the importance of quality nutrition as a determinant of mental health.
Positive tone
The worksheet was informed by social cognitive theory, which “highlights the important role of goal setting and behavior contracting, reinforcement, self-control, social norms, attitudes, and self-efficacy.”
It provides “basic education about important nutrition principles” as well as “very simple recommendations to increase knowledge about healthy eating” and “actionable tips for individuals to incorporate.” The researchers used a “positive” tone and included motivational interviewing questions.
“Delivery of the intervention by the patient’s mental health care provider is by design, in an attempt to address the widely held misbelief that nutrition intervention is of limited importance to mental health care and begin to change norms,” Dr. LaChance said.
The worksheet addresses monetary barriers to healthy eating; offers practical tips to “increase perceived control and self-efficacy”; is written in simple, accessible, nontechnical language; and includes foods from a range of cultural backgrounds.
To ensure that the worksheet and clinical guide met the needs of the target population, the researchers conducted a focus group with five psychiatrists and individual phone interviews with people who live with psychosis (n = 6).
Participants with psychosis were evenly divided between male and female and six age groups were represented: younger than 20 years; 21-30 years; 31-40 years; 41-50 years; 51-50 years; and older than 60 years. Of these participants, half scored in the “limited literacy” range, based on a nutritional literacy assessment tool (the Newest Vital Sign [NVS]).
A revised version of the worksheet, taking participants’ feedback into account, was mailed to all participants, who then provided further feedback on the revised version.
‘Unspoken area’
The clinician guide contains not only an overview and a suggested agenda to steer discussion, but also a sample visual representation of the recommended relative proportions of different food categories in an ideal meal as well as sample meals, a budgeting discussion, and a list of goals.
A closing statement encourages the clinician to “keep the messaging positive, celebrate small victories, and provide encouragement.”
Specific dietary recommendations include choosing complex carbohydrates and healthy fats, reducing highly processed foods and sugar, adding vegetables and fruits to meals and snacks, and eating protein-rich foods throughout the day.
A “noteworthy theme” that emerged in discussions with psychiatrists as well as participants with SSD was “the lack of nutrition training in medical education and psychiatric residency and the general absence of nutritional counseling in this field of medicine.”
One participant described nutrition as “definitely an unspoken area” in schizophrenia – especially in institutional settings, where “you are overloaded with sugars, not healthy grain, not complex grain. You get white bread sandwiches, shitty juice.”
Powerful tool
Commenting on the paper for this news organization, Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and a nutrition educator at Harvard Medical School, both in Boston, said she appreciates that this paper “is seeking methods to expand treatment options for those with SSD and improve provider understanding/knowledge of therapeutic foods.”
She called the pilot evaluation “notably small,” but added that it “provides results to suggest that scaling this worksheet/guide may hold promise to better provide nutritional counseling to those with psychiatric illness.”
Dr. Naidoo, also a chef and the author of “This Is Your Brain on Food,” who was not involved in the study said, “I’ve seen the power of food as medicine in my own hospital practice and do believe that food is one of the most powerful tools we have in supporting mental fitness and emotional well-being.”
The project was funded by the Canadian CAM Research Fund. Dr. LaChance and Dr. Naidoo have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new tool designed by psychiatrists to help guide nutritional counseling in patients with schizophrenia spectrum disorders (SSD) has been released.
The worksheet and clinician guide were developed using results from a recent scoping review on the relationship between diet and mental health in patients with SSD, and a feedback process involving a focus group with psychiatrists and individuals who had lived experience with psychosis.
“Mental health clinicians already have the training to help our patients make behavioral changes,” lead author Laura LaChance, MD, lecturer, department of psychiatry, and a psychiatrist at St. Mary’s Hospital Centre, McGill University, Montreal, said in an interview.
“We work every day with patients to help them to reduce their substance use, improve their sleep, take medications, etc., and nutrition should be added to the radar [since] eating well for mental health is part of self-care and can be included in mental health treatment plans,” she said.
The paper was published online Nov. 10 in BMC Psychiatry.
Nutrition frequently ignored
Dr. LaChance noted that “nutrition is largely absent from mental health training programs and often ignored in clinical practice.”
The investigators “wanted to create a tool to help incorporate basic nutritional counseling into the care of individuals with severe mental illness.” They wanted the tool “to be simple enough to understand for patients and simple enough to use for mental health care professionals who don’t have any official nutrition training.”
The team developed a worksheet that includes dietary recommendations, the majority of which are supported by the scoping review and consistent with Canada’s Food Guide. The review “identified all of the published literature related to the relationship between diet and psychiatric symptoms of SSD,” synthesizing the results of 822 prior articles.
and does not contradict generally accepted recommendations for weight management. It is suitable for all patients including those with low or normal body mass index and provides psychoeducation about the importance of quality nutrition as a determinant of mental health.
Positive tone
The worksheet was informed by social cognitive theory, which “highlights the important role of goal setting and behavior contracting, reinforcement, self-control, social norms, attitudes, and self-efficacy.”
It provides “basic education about important nutrition principles” as well as “very simple recommendations to increase knowledge about healthy eating” and “actionable tips for individuals to incorporate.” The researchers used a “positive” tone and included motivational interviewing questions.
“Delivery of the intervention by the patient’s mental health care provider is by design, in an attempt to address the widely held misbelief that nutrition intervention is of limited importance to mental health care and begin to change norms,” Dr. LaChance said.
The worksheet addresses monetary barriers to healthy eating; offers practical tips to “increase perceived control and self-efficacy”; is written in simple, accessible, nontechnical language; and includes foods from a range of cultural backgrounds.
To ensure that the worksheet and clinical guide met the needs of the target population, the researchers conducted a focus group with five psychiatrists and individual phone interviews with people who live with psychosis (n = 6).
Participants with psychosis were evenly divided between male and female and six age groups were represented: younger than 20 years; 21-30 years; 31-40 years; 41-50 years; 51-50 years; and older than 60 years. Of these participants, half scored in the “limited literacy” range, based on a nutritional literacy assessment tool (the Newest Vital Sign [NVS]).
A revised version of the worksheet, taking participants’ feedback into account, was mailed to all participants, who then provided further feedback on the revised version.
‘Unspoken area’
The clinician guide contains not only an overview and a suggested agenda to steer discussion, but also a sample visual representation of the recommended relative proportions of different food categories in an ideal meal as well as sample meals, a budgeting discussion, and a list of goals.
A closing statement encourages the clinician to “keep the messaging positive, celebrate small victories, and provide encouragement.”
Specific dietary recommendations include choosing complex carbohydrates and healthy fats, reducing highly processed foods and sugar, adding vegetables and fruits to meals and snacks, and eating protein-rich foods throughout the day.
A “noteworthy theme” that emerged in discussions with psychiatrists as well as participants with SSD was “the lack of nutrition training in medical education and psychiatric residency and the general absence of nutritional counseling in this field of medicine.”
One participant described nutrition as “definitely an unspoken area” in schizophrenia – especially in institutional settings, where “you are overloaded with sugars, not healthy grain, not complex grain. You get white bread sandwiches, shitty juice.”
Powerful tool
Commenting on the paper for this news organization, Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and a nutrition educator at Harvard Medical School, both in Boston, said she appreciates that this paper “is seeking methods to expand treatment options for those with SSD and improve provider understanding/knowledge of therapeutic foods.”
She called the pilot evaluation “notably small,” but added that it “provides results to suggest that scaling this worksheet/guide may hold promise to better provide nutritional counseling to those with psychiatric illness.”
Dr. Naidoo, also a chef and the author of “This Is Your Brain on Food,” who was not involved in the study said, “I’ve seen the power of food as medicine in my own hospital practice and do believe that food is one of the most powerful tools we have in supporting mental fitness and emotional well-being.”
The project was funded by the Canadian CAM Research Fund. Dr. LaChance and Dr. Naidoo have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BMC PSYCHIATRY
Swell in off-label antipsychotic prescribing ‘not harmless’
A growing trend of off-label, low-dose antipsychotic prescribing to treat disorders such as anxiety and insomnia has been tied to an increased risk of cardiometabolic death, new research shows.
Investigators studied data from large Swedish registries on over 420,000 individuals without previous psychotic, bipolar, or cardiometabolic disorders and found that off-label treatment with olanzapine or quetiapine for 6 to 12 months – even at a low dose – was associated with an almost twofold higher risk of cardiometabolic mortality, compared to no treatment. The risk remained elevated after 12 months, but the finding was not deemed significant.
“Clinicians should be made aware that low-dose treatment with these drugs is probably not a harmless choice for insomnia and anxiety, and while they have the benefit of not being addictive and [are] seemingly effective, they might come at a cost of shortening patients’ life span,” study investigator Jonas Berge, MD, PhD, associate professor and resident psychiatrist, Lund University, Sweden, said in an interview.
“Clinicians should take this information into account when prescribing the drugs and also monitor the patients with regular physical examinations and blood tests in the same way as when treating patients with psychosis with higher doses of these drugs,” he said.
The study was published online Nov. 9 in the Journal of Psychiatric Research.
A growing trend
Use of low-dose antipsychotics to treat a variety of psychiatric and behavioral disturbances, including anxiety, insomnia, and agitation, has “surged in popularity,” the authors wrote.
Quetiapine and olanzapine “rank as two of the most frequently prescribed second-generation antipsychotics and, next to clozapine, are considered to exhort the highest risk for cardiometabolic sequelae, including components of metabolic syndrome,” they added.
Previous research examining the association between second-generation antipsychotics and placebo has either not focused on cardiometabolic-specific causes or has examined only cohorts with severe mental illness, so those findings “do not necessarily generalize to others treated off-label,” they noted.
“The motivation for the study came from my work as a psychiatrist, in which I’ve noticed that the off-label use of these medications [olanzapine and quetiapine] for anxiety and insomnia seems highly prevalent, and that many patients seem to gain a lot of weight, despite low doses,” Dr. Berge said.
There is “evidence to suggest that clinicians may underappreciate cardiometabolic risks owing to antipsychotic treatment, as routine screening is often incomplete or inconsistent,” the authors noted.
“To do a risk-benefit analysis of these drugs in low doses, the risks involved – as well as the effects, of course – need to be studied,” Dr. Berge stated.
To investigate the question, the researchers turned to three large cross-linked Swedish registers: the National Patient Register, containing demographic and medical data; the Prescribed Drug Register; and the Cause of Death Register.
They identified all individuals aged 18 years and older with at least one psychiatric visit (inpatient or outpatient) between July 1, 2006, and Dec. 31, 2016, to see how many were prescribed low-dose olanzapine or quetiapine (defined as ≤ 5 mg/day of olanzapine or olanzapine equivalent [OE]), which was used as a proxy marker for off-label treatment, since this dose is considered subtherapeutic for severe mental illness.
They calculated two time-dependent variables – cumulative dose and past annual average dose – and then used those to compute three different exposure valuables: those treated with low-dose OE; cumulative exposure (i.e., period treated with an average 5 mg/day); and a continuous variable “corresponding to each year exposed OE 5 mg/day.”
The primary outcome was set as mortality from cardiometabolic-related disorders, while secondary outcomes were disease-specific and all-cause mortality.
‘Weak’ association
The final cohort consisted of 428,525 individuals (mean [SD] age, 36.8 [15.4] years, 52.7% female) at baseline, with observation taking place over a mean of 4.8 years [range, 1 day to 10.5 years]) or a total of over 2 million (2,062,241) person-years.
Of the cohort, 4.3% (n = 18,317) had at least two prescriptions for either olanzapine or quetiapine (although subsequently, 86.5% were censored for exceeding the average OE dose of 5 mg/day).
By the end of the study, 3.1% of the cohort had died during the observation time, and of these, 69.5% were from disease-specific causes, while close to one-fifth (19.5%) were from cardiometabolic-specific causes.
On the whole, treatment status (i.e., treated vs. untreated) was not significantly associated with cardiometabolic mortality (adjusted hazard ratio [HR], .86 [95% confidence interval, 0.64-1.15]; P = .307).
Compared to no treatment, treatment with olanzapine or quetiapine for less than 6 months was significantly associated with a reduced risk of cardiovascular mortality (adjusted HR, .56 [.37 – .87]; P = .010). On the other hand, treatment for 6-12 months was significantly associated with an almost twofold increased risk (adjusted HR, 1.89 [1.22-2.92]; P = .004). The increased risk continued beyond 12 months, although the difference no longer remained significant.
“In the subgroup analysis consisting of individuals who had ever been treated with olanzapine/quetiapine, starting at the date of their first prescription, the hazard for cardiometabolic mortality increased significantly by 45% (6%-99%; P = .019) for every year exposed to an average 5 mg/day,” the authors reported.
The authors concluded that the association between low-dose olanzapine/quetiapine treatment and cardiometabolic mortality was present, but “weak.”
The hazard for disease-specific mortality also significantly increased with each year exposed to an average of 5 mg/day of OE (HR, 1.24 [1.03-1.50]; P = .026).
Treatment status similarly was associated with all-cause mortality (HR, 1.16 [1.03-1.30]; P = .012), although the increased hazard for all-cause mortality with each year of exposure was not considered significant.
“The findings of this study are consistent with the hypothesis that continuous low-dose treatment with these drugs is associated with increased cardiometabolic mortality, but ,” Dr. Berge said.
Seek alternatives
Commenting on the study for this news organization, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, called it a “timely paper” and “an important concept [because] low-doses of these antipsychotics are frequently prescribed across America and there has been less data on the safety [of these antipsychotics at lower doses].”
Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto, who was not involved with the study, said that this “important report reminds us that there are metabolic safety concerns, even at low doses, where these medications are often used off label.”
He advised clinicians to “seek alternatives, and alternatives that are on-label, for conditions like anxiety and sleep disturbances.”
This work was supported by the South Region Board ALF, Sweden. Dr. Berge and coauthors have disclosed no relevant financial relationships. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; and speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and AbbVie. Dr. McIntyre is CEO of AltMed.
A version of this article first appeared on Medscape.com.
A growing trend of off-label, low-dose antipsychotic prescribing to treat disorders such as anxiety and insomnia has been tied to an increased risk of cardiometabolic death, new research shows.
Investigators studied data from large Swedish registries on over 420,000 individuals without previous psychotic, bipolar, or cardiometabolic disorders and found that off-label treatment with olanzapine or quetiapine for 6 to 12 months – even at a low dose – was associated with an almost twofold higher risk of cardiometabolic mortality, compared to no treatment. The risk remained elevated after 12 months, but the finding was not deemed significant.
“Clinicians should be made aware that low-dose treatment with these drugs is probably not a harmless choice for insomnia and anxiety, and while they have the benefit of not being addictive and [are] seemingly effective, they might come at a cost of shortening patients’ life span,” study investigator Jonas Berge, MD, PhD, associate professor and resident psychiatrist, Lund University, Sweden, said in an interview.
“Clinicians should take this information into account when prescribing the drugs and also monitor the patients with regular physical examinations and blood tests in the same way as when treating patients with psychosis with higher doses of these drugs,” he said.
The study was published online Nov. 9 in the Journal of Psychiatric Research.
A growing trend
Use of low-dose antipsychotics to treat a variety of psychiatric and behavioral disturbances, including anxiety, insomnia, and agitation, has “surged in popularity,” the authors wrote.
Quetiapine and olanzapine “rank as two of the most frequently prescribed second-generation antipsychotics and, next to clozapine, are considered to exhort the highest risk for cardiometabolic sequelae, including components of metabolic syndrome,” they added.
Previous research examining the association between second-generation antipsychotics and placebo has either not focused on cardiometabolic-specific causes or has examined only cohorts with severe mental illness, so those findings “do not necessarily generalize to others treated off-label,” they noted.
“The motivation for the study came from my work as a psychiatrist, in which I’ve noticed that the off-label use of these medications [olanzapine and quetiapine] for anxiety and insomnia seems highly prevalent, and that many patients seem to gain a lot of weight, despite low doses,” Dr. Berge said.
There is “evidence to suggest that clinicians may underappreciate cardiometabolic risks owing to antipsychotic treatment, as routine screening is often incomplete or inconsistent,” the authors noted.
“To do a risk-benefit analysis of these drugs in low doses, the risks involved – as well as the effects, of course – need to be studied,” Dr. Berge stated.
To investigate the question, the researchers turned to three large cross-linked Swedish registers: the National Patient Register, containing demographic and medical data; the Prescribed Drug Register; and the Cause of Death Register.
They identified all individuals aged 18 years and older with at least one psychiatric visit (inpatient or outpatient) between July 1, 2006, and Dec. 31, 2016, to see how many were prescribed low-dose olanzapine or quetiapine (defined as ≤ 5 mg/day of olanzapine or olanzapine equivalent [OE]), which was used as a proxy marker for off-label treatment, since this dose is considered subtherapeutic for severe mental illness.
They calculated two time-dependent variables – cumulative dose and past annual average dose – and then used those to compute three different exposure valuables: those treated with low-dose OE; cumulative exposure (i.e., period treated with an average 5 mg/day); and a continuous variable “corresponding to each year exposed OE 5 mg/day.”
The primary outcome was set as mortality from cardiometabolic-related disorders, while secondary outcomes were disease-specific and all-cause mortality.
‘Weak’ association
The final cohort consisted of 428,525 individuals (mean [SD] age, 36.8 [15.4] years, 52.7% female) at baseline, with observation taking place over a mean of 4.8 years [range, 1 day to 10.5 years]) or a total of over 2 million (2,062,241) person-years.
Of the cohort, 4.3% (n = 18,317) had at least two prescriptions for either olanzapine or quetiapine (although subsequently, 86.5% were censored for exceeding the average OE dose of 5 mg/day).
By the end of the study, 3.1% of the cohort had died during the observation time, and of these, 69.5% were from disease-specific causes, while close to one-fifth (19.5%) were from cardiometabolic-specific causes.
On the whole, treatment status (i.e., treated vs. untreated) was not significantly associated with cardiometabolic mortality (adjusted hazard ratio [HR], .86 [95% confidence interval, 0.64-1.15]; P = .307).
Compared to no treatment, treatment with olanzapine or quetiapine for less than 6 months was significantly associated with a reduced risk of cardiovascular mortality (adjusted HR, .56 [.37 – .87]; P = .010). On the other hand, treatment for 6-12 months was significantly associated with an almost twofold increased risk (adjusted HR, 1.89 [1.22-2.92]; P = .004). The increased risk continued beyond 12 months, although the difference no longer remained significant.
“In the subgroup analysis consisting of individuals who had ever been treated with olanzapine/quetiapine, starting at the date of their first prescription, the hazard for cardiometabolic mortality increased significantly by 45% (6%-99%; P = .019) for every year exposed to an average 5 mg/day,” the authors reported.
The authors concluded that the association between low-dose olanzapine/quetiapine treatment and cardiometabolic mortality was present, but “weak.”
The hazard for disease-specific mortality also significantly increased with each year exposed to an average of 5 mg/day of OE (HR, 1.24 [1.03-1.50]; P = .026).
Treatment status similarly was associated with all-cause mortality (HR, 1.16 [1.03-1.30]; P = .012), although the increased hazard for all-cause mortality with each year of exposure was not considered significant.
“The findings of this study are consistent with the hypothesis that continuous low-dose treatment with these drugs is associated with increased cardiometabolic mortality, but ,” Dr. Berge said.
Seek alternatives
Commenting on the study for this news organization, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, called it a “timely paper” and “an important concept [because] low-doses of these antipsychotics are frequently prescribed across America and there has been less data on the safety [of these antipsychotics at lower doses].”
Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto, who was not involved with the study, said that this “important report reminds us that there are metabolic safety concerns, even at low doses, where these medications are often used off label.”
He advised clinicians to “seek alternatives, and alternatives that are on-label, for conditions like anxiety and sleep disturbances.”
This work was supported by the South Region Board ALF, Sweden. Dr. Berge and coauthors have disclosed no relevant financial relationships. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; and speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and AbbVie. Dr. McIntyre is CEO of AltMed.
A version of this article first appeared on Medscape.com.
A growing trend of off-label, low-dose antipsychotic prescribing to treat disorders such as anxiety and insomnia has been tied to an increased risk of cardiometabolic death, new research shows.
Investigators studied data from large Swedish registries on over 420,000 individuals without previous psychotic, bipolar, or cardiometabolic disorders and found that off-label treatment with olanzapine or quetiapine for 6 to 12 months – even at a low dose – was associated with an almost twofold higher risk of cardiometabolic mortality, compared to no treatment. The risk remained elevated after 12 months, but the finding was not deemed significant.
“Clinicians should be made aware that low-dose treatment with these drugs is probably not a harmless choice for insomnia and anxiety, and while they have the benefit of not being addictive and [are] seemingly effective, they might come at a cost of shortening patients’ life span,” study investigator Jonas Berge, MD, PhD, associate professor and resident psychiatrist, Lund University, Sweden, said in an interview.
“Clinicians should take this information into account when prescribing the drugs and also monitor the patients with regular physical examinations and blood tests in the same way as when treating patients with psychosis with higher doses of these drugs,” he said.
The study was published online Nov. 9 in the Journal of Psychiatric Research.
A growing trend
Use of low-dose antipsychotics to treat a variety of psychiatric and behavioral disturbances, including anxiety, insomnia, and agitation, has “surged in popularity,” the authors wrote.
Quetiapine and olanzapine “rank as two of the most frequently prescribed second-generation antipsychotics and, next to clozapine, are considered to exhort the highest risk for cardiometabolic sequelae, including components of metabolic syndrome,” they added.
Previous research examining the association between second-generation antipsychotics and placebo has either not focused on cardiometabolic-specific causes or has examined only cohorts with severe mental illness, so those findings “do not necessarily generalize to others treated off-label,” they noted.
“The motivation for the study came from my work as a psychiatrist, in which I’ve noticed that the off-label use of these medications [olanzapine and quetiapine] for anxiety and insomnia seems highly prevalent, and that many patients seem to gain a lot of weight, despite low doses,” Dr. Berge said.
There is “evidence to suggest that clinicians may underappreciate cardiometabolic risks owing to antipsychotic treatment, as routine screening is often incomplete or inconsistent,” the authors noted.
“To do a risk-benefit analysis of these drugs in low doses, the risks involved – as well as the effects, of course – need to be studied,” Dr. Berge stated.
To investigate the question, the researchers turned to three large cross-linked Swedish registers: the National Patient Register, containing demographic and medical data; the Prescribed Drug Register; and the Cause of Death Register.
They identified all individuals aged 18 years and older with at least one psychiatric visit (inpatient or outpatient) between July 1, 2006, and Dec. 31, 2016, to see how many were prescribed low-dose olanzapine or quetiapine (defined as ≤ 5 mg/day of olanzapine or olanzapine equivalent [OE]), which was used as a proxy marker for off-label treatment, since this dose is considered subtherapeutic for severe mental illness.
They calculated two time-dependent variables – cumulative dose and past annual average dose – and then used those to compute three different exposure valuables: those treated with low-dose OE; cumulative exposure (i.e., period treated with an average 5 mg/day); and a continuous variable “corresponding to each year exposed OE 5 mg/day.”
The primary outcome was set as mortality from cardiometabolic-related disorders, while secondary outcomes were disease-specific and all-cause mortality.
‘Weak’ association
The final cohort consisted of 428,525 individuals (mean [SD] age, 36.8 [15.4] years, 52.7% female) at baseline, with observation taking place over a mean of 4.8 years [range, 1 day to 10.5 years]) or a total of over 2 million (2,062,241) person-years.
Of the cohort, 4.3% (n = 18,317) had at least two prescriptions for either olanzapine or quetiapine (although subsequently, 86.5% were censored for exceeding the average OE dose of 5 mg/day).
By the end of the study, 3.1% of the cohort had died during the observation time, and of these, 69.5% were from disease-specific causes, while close to one-fifth (19.5%) were from cardiometabolic-specific causes.
On the whole, treatment status (i.e., treated vs. untreated) was not significantly associated with cardiometabolic mortality (adjusted hazard ratio [HR], .86 [95% confidence interval, 0.64-1.15]; P = .307).
Compared to no treatment, treatment with olanzapine or quetiapine for less than 6 months was significantly associated with a reduced risk of cardiovascular mortality (adjusted HR, .56 [.37 – .87]; P = .010). On the other hand, treatment for 6-12 months was significantly associated with an almost twofold increased risk (adjusted HR, 1.89 [1.22-2.92]; P = .004). The increased risk continued beyond 12 months, although the difference no longer remained significant.
“In the subgroup analysis consisting of individuals who had ever been treated with olanzapine/quetiapine, starting at the date of their first prescription, the hazard for cardiometabolic mortality increased significantly by 45% (6%-99%; P = .019) for every year exposed to an average 5 mg/day,” the authors reported.
The authors concluded that the association between low-dose olanzapine/quetiapine treatment and cardiometabolic mortality was present, but “weak.”
The hazard for disease-specific mortality also significantly increased with each year exposed to an average of 5 mg/day of OE (HR, 1.24 [1.03-1.50]; P = .026).
Treatment status similarly was associated with all-cause mortality (HR, 1.16 [1.03-1.30]; P = .012), although the increased hazard for all-cause mortality with each year of exposure was not considered significant.
“The findings of this study are consistent with the hypothesis that continuous low-dose treatment with these drugs is associated with increased cardiometabolic mortality, but ,” Dr. Berge said.
Seek alternatives
Commenting on the study for this news organization, Roger S. McIntyre, MD, professor of psychiatry and pharmacology, University of Toronto, and head of the Mood Disorders Psychopharmacology Unit, called it a “timely paper” and “an important concept [because] low-doses of these antipsychotics are frequently prescribed across America and there has been less data on the safety [of these antipsychotics at lower doses].”
Dr. McIntyre, chairman and executive director of the Brain and Cognitive Discover Foundation, Toronto, who was not involved with the study, said that this “important report reminds us that there are metabolic safety concerns, even at low doses, where these medications are often used off label.”
He advised clinicians to “seek alternatives, and alternatives that are on-label, for conditions like anxiety and sleep disturbances.”
This work was supported by the South Region Board ALF, Sweden. Dr. Berge and coauthors have disclosed no relevant financial relationships. Dr. McIntyre has received research grant support from CIHR/GACD/Chinese National Natural Research Foundation; and speaker/consultation fees from Lundbeck, Janssen, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, Eisai, Minerva, Intra-Cellular, and AbbVie. Dr. McIntyre is CEO of AltMed.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF PSYCHIATRIC RESEARCH
COVID-19 mortality risk factors: An unexpected finding
Schizophrenia and severe mood and anxiety disorders are associated with a significantly lower risk of COVID-19 but are tied to a two- to fourfold increased risk of death from the virus, new research shows.
The study results held after the researchers controlled for other risk factors, and they contradict an earlier study that showed no increased mortality risk associated with mood or anxiety disorders. The findings come as the overall number of deaths in the United States approaches 800,000.
“These patients were less likely to be infected because they were probably less exposed, but once they have the infection, they are more prone to worse outcomes,” lead author Antonio L. Teixeira, MD, PhD, professor of psychiatry with McGovern Medical School at the University of Texas Health Science Center at Houston, said in an interview.
The study was published online Nov. 23 in JAMA Network Open.
Unexpected finding
Researchers analyzed electronic health records for 2.5 million adults with private health insurance who were tested for COVID-19 in 2020.
The overall positivity rate for the entire cohort was 11.91%, and patients with severe psychiatric illness fell below that rate. Positivity rates were 9.86% for people with schizophrenia or mood disorders and 11.17% among those with anxiety disorder.
Despite their lower positivity rate, patients with schizophrenia had the highest odds of death from COVID-19 after adjustment for age, race, body mass index, and comorbidities (aOR, 3.74; 95% confidence interval, 2.66-5.24).
Those results were not very surprising, Dr. Teixeira said, as earlier studies have reported similar findings. However,
Patients with mood disorders were nearly three times as likely to die (aOR, 2.76; 95% CI, 2.00-3.81), and those with anxiety disorders had more than double the mortality risk (aOR, 2.34; 95% CI, 1.68-3.27).
“We were expecting some increase, but there was strong evidence in those populations as well,” he said. “We were especially surprised at the data on patients with anxiety disorders.”
An outstanding question
These findings contradict a study published Jan. 27, 2021, in JAMA Psychiatry, that showed no significant increase in mortality risk among those with mood or anxiety disorders.
Study methodology and timing might explain some of the differences, Katlyn Nemani, MD, a research assistant professor of psychiatry at New York University, who led that earlier study, said in an interview.
Dr. Nemani’s study had a smaller study sample, examined mortality over a 30-day period after a positive COVID-19 test, and was limited to the peak of the pandemic in New York, between March and May 2020. Dr. Teixeira’s team examined a full year of data and assessed mortality for 7 days following a positive test.
“It is possible patients with some psychiatric disorders were less likely to receive or successfully respond to treatment for severe COVD-19 which evolved during the course of the pandemic,” Dr. Nemani said, adding that it’s also possible that differences in mortality in the days following infection became attenuated over time.
While a meta-analysis published in July and reported by this news organization at that time did show higher COVID-19 mortality among patients with mood disorders, the risk was far lower than that reported in this new study. That report, which included 33 studies in 22 countries, also found no increase in risk among those with anxiety disorder.
In October, the Centers for Disease Control and Prevention added mood disorders to the list of medical conditions that increase the risk for more severe COVID-19. Schizophrenia was already on that list.
“The outstanding question is what underlies this increased risk,” Dr. Nemani said. “Future studies focused on immune-mediated mechanisms and other potential explanations will help guide targeted interventions to reduce morbidity and mortality in this vulnerable population.”
Funding for the study was not disclosed. Dr. Teixeira and Dr. Nemani report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Schizophrenia and severe mood and anxiety disorders are associated with a significantly lower risk of COVID-19 but are tied to a two- to fourfold increased risk of death from the virus, new research shows.
The study results held after the researchers controlled for other risk factors, and they contradict an earlier study that showed no increased mortality risk associated with mood or anxiety disorders. The findings come as the overall number of deaths in the United States approaches 800,000.
“These patients were less likely to be infected because they were probably less exposed, but once they have the infection, they are more prone to worse outcomes,” lead author Antonio L. Teixeira, MD, PhD, professor of psychiatry with McGovern Medical School at the University of Texas Health Science Center at Houston, said in an interview.
The study was published online Nov. 23 in JAMA Network Open.
Unexpected finding
Researchers analyzed electronic health records for 2.5 million adults with private health insurance who were tested for COVID-19 in 2020.
The overall positivity rate for the entire cohort was 11.91%, and patients with severe psychiatric illness fell below that rate. Positivity rates were 9.86% for people with schizophrenia or mood disorders and 11.17% among those with anxiety disorder.
Despite their lower positivity rate, patients with schizophrenia had the highest odds of death from COVID-19 after adjustment for age, race, body mass index, and comorbidities (aOR, 3.74; 95% confidence interval, 2.66-5.24).
Those results were not very surprising, Dr. Teixeira said, as earlier studies have reported similar findings. However,
Patients with mood disorders were nearly three times as likely to die (aOR, 2.76; 95% CI, 2.00-3.81), and those with anxiety disorders had more than double the mortality risk (aOR, 2.34; 95% CI, 1.68-3.27).
“We were expecting some increase, but there was strong evidence in those populations as well,” he said. “We were especially surprised at the data on patients with anxiety disorders.”
An outstanding question
These findings contradict a study published Jan. 27, 2021, in JAMA Psychiatry, that showed no significant increase in mortality risk among those with mood or anxiety disorders.
Study methodology and timing might explain some of the differences, Katlyn Nemani, MD, a research assistant professor of psychiatry at New York University, who led that earlier study, said in an interview.
Dr. Nemani’s study had a smaller study sample, examined mortality over a 30-day period after a positive COVID-19 test, and was limited to the peak of the pandemic in New York, between March and May 2020. Dr. Teixeira’s team examined a full year of data and assessed mortality for 7 days following a positive test.
“It is possible patients with some psychiatric disorders were less likely to receive or successfully respond to treatment for severe COVD-19 which evolved during the course of the pandemic,” Dr. Nemani said, adding that it’s also possible that differences in mortality in the days following infection became attenuated over time.
While a meta-analysis published in July and reported by this news organization at that time did show higher COVID-19 mortality among patients with mood disorders, the risk was far lower than that reported in this new study. That report, which included 33 studies in 22 countries, also found no increase in risk among those with anxiety disorder.
In October, the Centers for Disease Control and Prevention added mood disorders to the list of medical conditions that increase the risk for more severe COVID-19. Schizophrenia was already on that list.
“The outstanding question is what underlies this increased risk,” Dr. Nemani said. “Future studies focused on immune-mediated mechanisms and other potential explanations will help guide targeted interventions to reduce morbidity and mortality in this vulnerable population.”
Funding for the study was not disclosed. Dr. Teixeira and Dr. Nemani report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Schizophrenia and severe mood and anxiety disorders are associated with a significantly lower risk of COVID-19 but are tied to a two- to fourfold increased risk of death from the virus, new research shows.
The study results held after the researchers controlled for other risk factors, and they contradict an earlier study that showed no increased mortality risk associated with mood or anxiety disorders. The findings come as the overall number of deaths in the United States approaches 800,000.
“These patients were less likely to be infected because they were probably less exposed, but once they have the infection, they are more prone to worse outcomes,” lead author Antonio L. Teixeira, MD, PhD, professor of psychiatry with McGovern Medical School at the University of Texas Health Science Center at Houston, said in an interview.
The study was published online Nov. 23 in JAMA Network Open.
Unexpected finding
Researchers analyzed electronic health records for 2.5 million adults with private health insurance who were tested for COVID-19 in 2020.
The overall positivity rate for the entire cohort was 11.91%, and patients with severe psychiatric illness fell below that rate. Positivity rates were 9.86% for people with schizophrenia or mood disorders and 11.17% among those with anxiety disorder.
Despite their lower positivity rate, patients with schizophrenia had the highest odds of death from COVID-19 after adjustment for age, race, body mass index, and comorbidities (aOR, 3.74; 95% confidence interval, 2.66-5.24).
Those results were not very surprising, Dr. Teixeira said, as earlier studies have reported similar findings. However,
Patients with mood disorders were nearly three times as likely to die (aOR, 2.76; 95% CI, 2.00-3.81), and those with anxiety disorders had more than double the mortality risk (aOR, 2.34; 95% CI, 1.68-3.27).
“We were expecting some increase, but there was strong evidence in those populations as well,” he said. “We were especially surprised at the data on patients with anxiety disorders.”
An outstanding question
These findings contradict a study published Jan. 27, 2021, in JAMA Psychiatry, that showed no significant increase in mortality risk among those with mood or anxiety disorders.
Study methodology and timing might explain some of the differences, Katlyn Nemani, MD, a research assistant professor of psychiatry at New York University, who led that earlier study, said in an interview.
Dr. Nemani’s study had a smaller study sample, examined mortality over a 30-day period after a positive COVID-19 test, and was limited to the peak of the pandemic in New York, between March and May 2020. Dr. Teixeira’s team examined a full year of data and assessed mortality for 7 days following a positive test.
“It is possible patients with some psychiatric disorders were less likely to receive or successfully respond to treatment for severe COVD-19 which evolved during the course of the pandemic,” Dr. Nemani said, adding that it’s also possible that differences in mortality in the days following infection became attenuated over time.
While a meta-analysis published in July and reported by this news organization at that time did show higher COVID-19 mortality among patients with mood disorders, the risk was far lower than that reported in this new study. That report, which included 33 studies in 22 countries, also found no increase in risk among those with anxiety disorder.
In October, the Centers for Disease Control and Prevention added mood disorders to the list of medical conditions that increase the risk for more severe COVID-19. Schizophrenia was already on that list.
“The outstanding question is what underlies this increased risk,” Dr. Nemani said. “Future studies focused on immune-mediated mechanisms and other potential explanations will help guide targeted interventions to reduce morbidity and mortality in this vulnerable population.”
Funding for the study was not disclosed. Dr. Teixeira and Dr. Nemani report no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
High-poverty areas host more firearm-related youth deaths
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
Higher poverty concentration at the county level significantly increased the risk of firearm-related deaths in children and youth aged 5-24 years in the United States, based on a review of approximately 67,000 fatalities.
Firearms are the second-leading cause of death in children and young adults in the United States, according to data from the Centers for Disease Control and Prevention, wrote Jefferson T. Barrett, MD, of The Children’s Hospital at Montefiore, New York, and colleagues. County-level poverty has been associated with increased injury mortality in children, but the association between county-level poverty and firearm-related mortality in particular has not been well studied.
In a cross-sectional study published in JAMA Pediatrics, 67,905 firearm-related deaths in children and youth aged 5-24 years that occurred between Jan. 1, 2007, and Dec. 31, 2016 were analyzed. The deaths included 42,512 homicides (62.6%), 23,034 suicides (33.9%), and 1,627 unintentional deaths (2.4%).
County poverty data were acquired from the U.S. Census Bureau. County-level poverty was divided into five categories based on percentage of the population living below the federal poverty level: 0%-4.9%, 5%-9.9%, 10%-14.9%, 15%-19.9%, and 20% or more.
Overall, 88.6% of the total deaths were in males. Notably, 44.8% of total firearm-related deaths and 63.9% of homicides occurred in non-Hispanic Blacks, who make up only 14% of the youth population in the United States, the researchers wrote.
The total number of firearm-related deaths was 248 in the lowest quintile of poverty concentration, followed by 6,841, 18,551, 27,305, and 14,960 in the remaining quintiles.
In a multivariate regression model that included demographics, urban versus rural, and statewide firearm prevalence, youth in counties with the highest quintile of poverty concentration had an increased rate of total firearm-related deaths (adjusted incidence rate ratio, 2.29), as well as increased rates of homicides, suicides, and unintentional deaths (aIRR, 3.55, 1.45, and 9.32, respectively), compared with those living in the lowest quintile of poverty concentration. Individuals in the highest poverty quintile accounted for 22.0% of total firearm-related deaths, 25.5% of homicides, 15.3% of suicides, and 25.1% of unintentional deaths.
The researchers also calculated the population-attributable fraction (PAF) and years of potential life lost. “The PAF represents the proportion of deaths associated with a particular exposure, which was concentrated county poverty in this study,” they explained. The PAF for all firearm-related deaths was 0.51, PAFs for homicides, suicides, and unintentional deaths were 0.66, 0.30, and 0.86, respectively. The PAF calculation translated to 34,292 firearm-related deaths that may not have occurred if youth in all counties had the same risk as those in counties with the lowest poverty concentration.
“Over the 10-year study period, we observed 3,833,105 years of potential life lost in youth aged 5-24 years from firearm-related deaths,” the researchers wrote.
The study findings were limited by several factors including the potential bias of a cross-section design, and inability to account for all the ways that county-level poverty might increase the risk of firearm-related death in children and teens, the researchers noted. Other potential limitations include possible misclassification of death, lack of data on individual family incomes, shifts in counties in the poverty categories over time, and the use of statewide, rather than countywide, estimates of firearm ownership.
However, the results are consistent with those of previous studies, and add that “mortality rates were consistent even after controlling for demographic variables, county urbanicity, and statewide firearm prevalence,” the researchers concluded.
Address structural racism to reduce disparities
“Firearm-related homicides among youth aged 5-24 years are among the causes of death with the greatest disparities,” based on CDC fatal injury reports, wrote Alice M. Ellyson, PhD, Frederick P. Rivara, MD, and Ali Rowhani-Rahbar, MD, all of the University of Washington, Seattle, in an accompanying editorial.
The current study builds on previous research, including studies showing an association between income inequality and firearm-related homicide, they said. More research is needed to determine how to intervene in the pathways between poverty and firearm-related death. For example, if access to high-quality health care is a factor, programs to increase access to health insurance, such as the Affordable Care Act and Children’s Health Insurance Program, or to increase access to high-quality trauma care may help reduce firearm-related death in youth.
“The study of where, how, and why racism operates as a factor in both poverty and firearm-related death must continue, especially considering the disparities consistently documented in Alaska Native or American Indian, Black, and Hispanic communities,” the editorialists wrote.
“Key potential mechanisms for reducing the consequences of poverty for firearm-related death are often denied to racial and ethnic minority groups through a variety of structures, policies, and systems in health care, employment, housing, transportation, and education,” they emphasized, and the impact of racism, not only on the pathways to poverty, but also on mediators between poverty and firearm-related death, must be explored.
Findings spotlight need to for poverty programs
The study was an interesting look at the specific relationship between poverty and firearm-related deaths in people aged younger than 25 years in the United States, Tim Joos, MD, of Seattle said in an interview.
“Although America is not a poor country, the combination of poverty within America and its unique gun culture seems to prove deadly for its youth,” Dr. Joos said. “The strongest relationship is between firearm-related homicide and poverty, but unintentional firearm deaths and poverty also are clearly linked, whereas the link between firearm-related suicide and poverty appears to be present, but small.”.
In the current study, “the authors note that firearm deaths are the second-leading cause of death among all people ages 15-24 years,” said Dr. Joos. “Many of us have followed children from infancy just to have them meet this untimely end as adolescents, wishing we had a vaccine or other remedy in our toolbelt for this particular scourge.
“As our country currently debates the size of the social safety net, this study is one of many that suggests government programs aimed at poverty alleviation would substantially contribute to the health of American youth,” Dr. Joos added.
The study received no outside funding. Lead author Dr. Barrett had no financial conflicts to disclose. Dr. Ellyson disclosed funds from the CDC, the state of Washington, and the Grandmothers Against Gun Violence Foundation for research outside the submitted work. Dr. Rivara disclosed funds from the National Institutes of Health, the State of Washington, and the National Collaborative on Gun Violence Research for research outside the submitted work. Dr. Rowhani-Rahbar disclosed funds from the CDC, National Institutes of Health, National Collaborative on Gun Violence Research, Fund for a Safer Future, and state of Washington for research outside the submitted work. Dr. Joos had no financial conflicts to disclose, but serves on the editorial advisory board of Pediatric News.
FROM JAMA PEDIATRICS
Positive Outcomes Following a Multidisciplinary Approach in the Diagnosis and Prevention of Hospital Delirium
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens
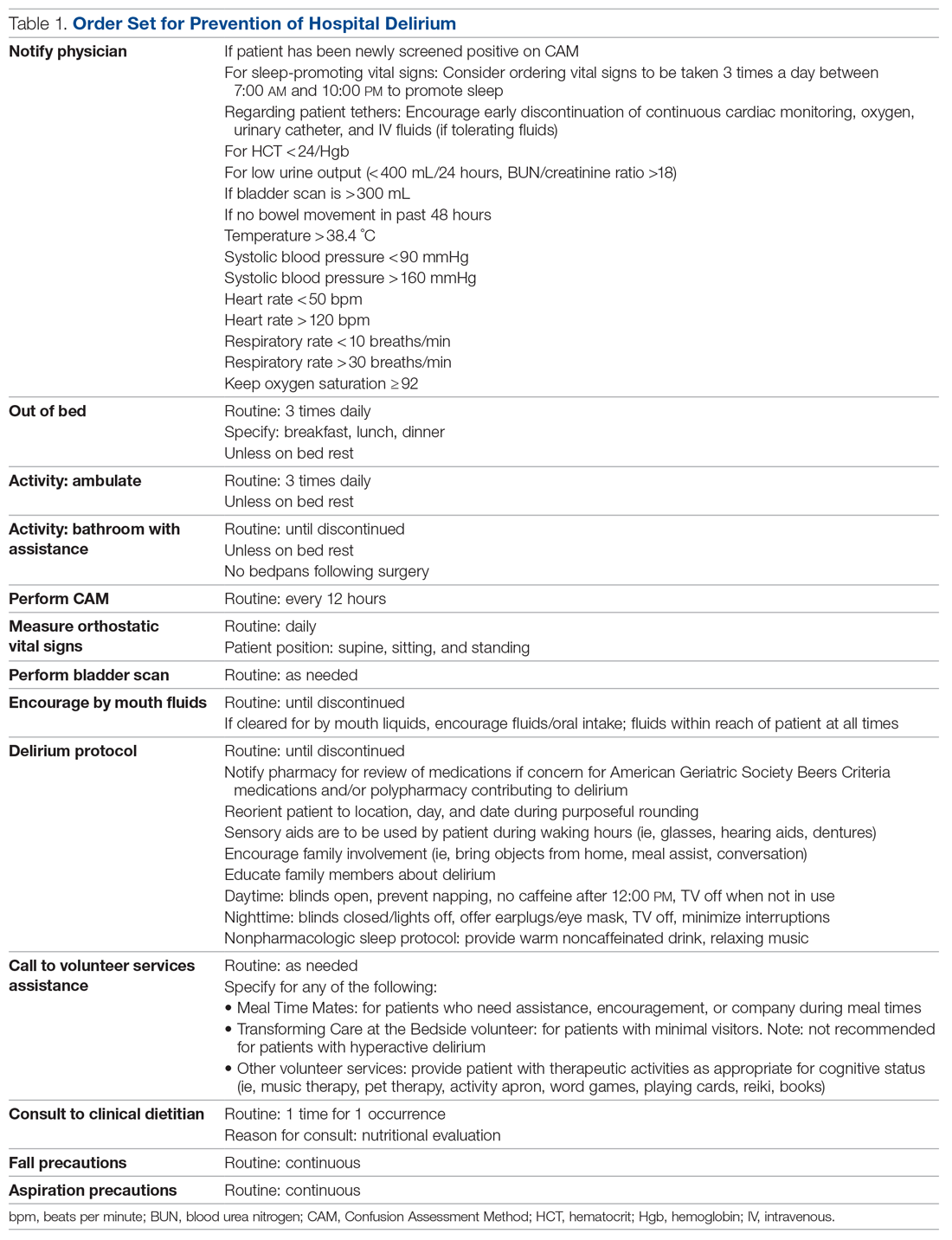
Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.
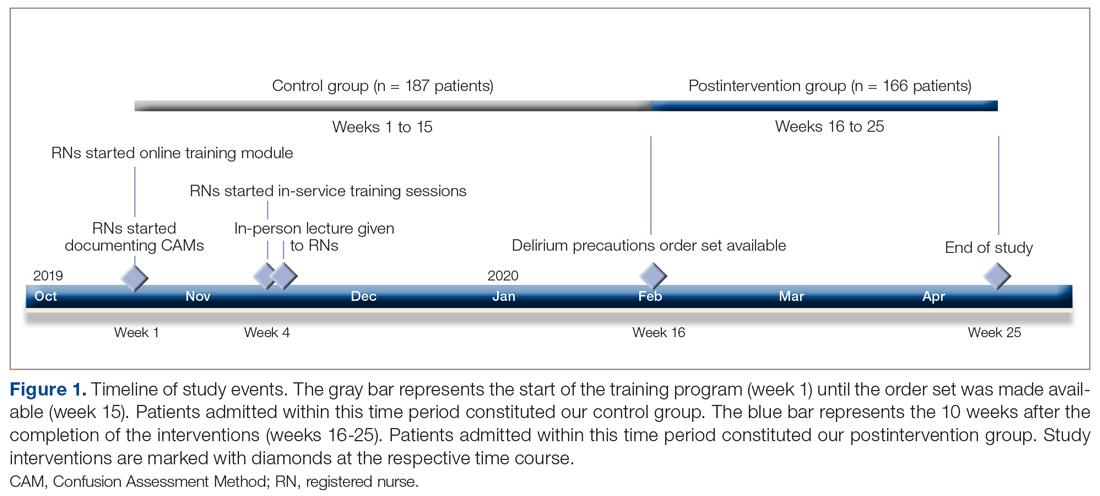
Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.
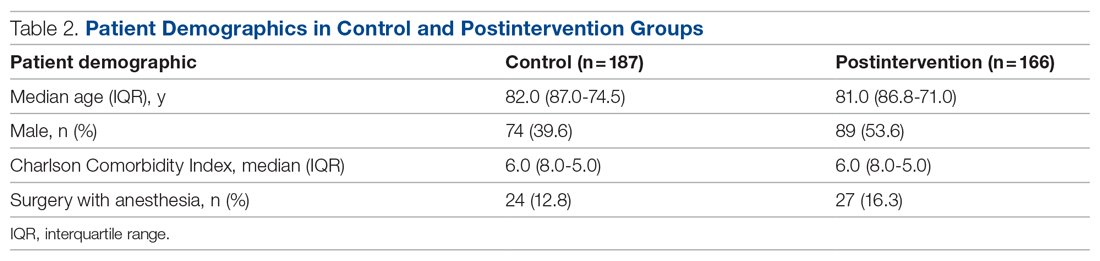
In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.
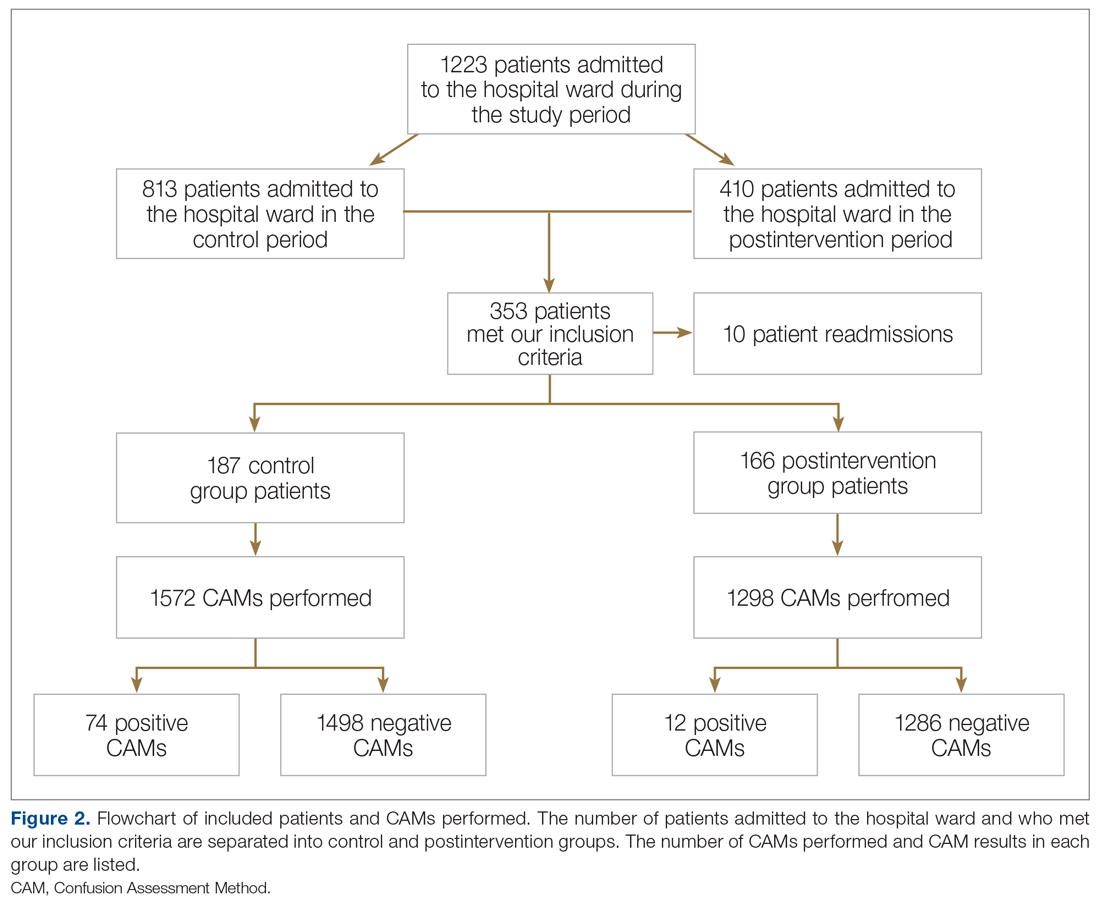
Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.
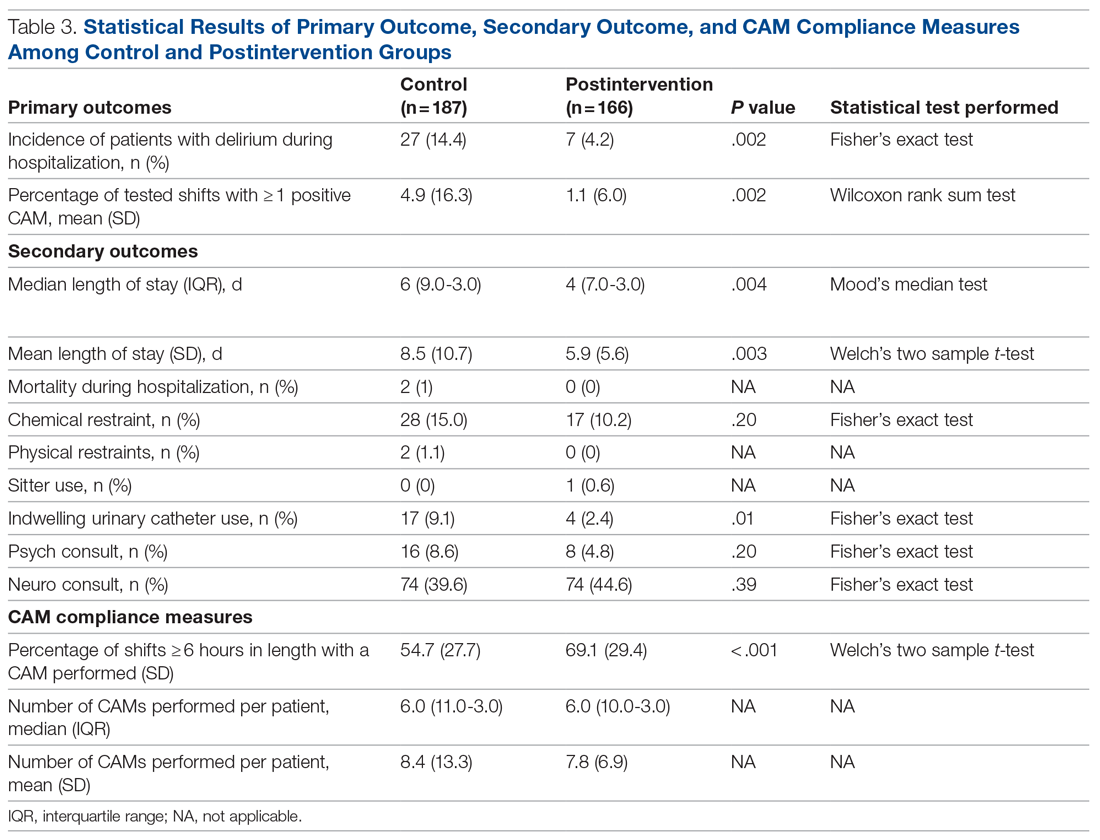
Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.
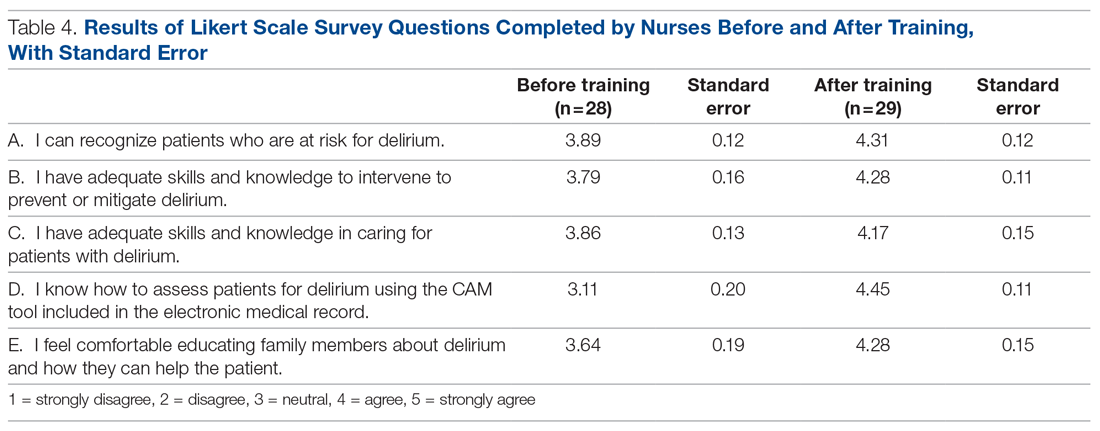
Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens

Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.

Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.

In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.

Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.

Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.

Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
From the Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA (Drs. Ching, Darwish, Li, Wong, Simpson, and Funk), the Department of Anesthesia, Cedars-Sinai Medical Center, Los Angeles, CA (Keith Siegel), and the Department of Psychiatry, Cedars-Sinai Medical Center, Los Angeles, CA (Dr. Bamgbose).
Objectives: To reduce the incidence and duration of delirium among patients in a hospital ward through standardized delirium screening tools and nonpharmacologic interventions. To advance nursing-focused education on delirium-prevention strategies. To measure the efficacy of the interventions with the aim of reproducing best practices.
Background: Delirium is associated with poor patient outcomes but may be preventable in a significant percentage of hospitalized patients.
Methods: Following nursing-focused education to prevent delirium, we prospectively evaluated patient care outcomes in a consecutive series of patients who were admitted to a hospital medical-surgical ward within a 25-week period. All patients who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria (N = 353). Standards for Quality Improvement Reporting Excellence guidelines were adhered to.
Results: There were 187 patients in the control group, and 166 in the postintervention group. Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days), mean length of stay (8.5 days vs 5.9 days), and use of an indwelling urinary catheter (9.1% vs 2.4%).
Conclusion: A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs.
Delirium is a disorder characterized by inattention and acute changes in cognition. It is defined by the American Psychiatric Association’s fifth edition of the Diagnostic and Statistical Manual of Mental Disorders as a disturbance in attention, awareness, and cognition over hours to a few days that is not better explained by a preexisting, established, or other evolving neurocognitive disorder.1 Delirium is common yet often under-recognized among hospitalized patients, particularly in the elderly. The incidence of delirium in elderly patients on admission is estimated to be 11% to 25%, and an additional 29% to 31% of elderly patients will develop delirium during the hospitalization.2 Delirium costs the health care system an estimated $38 billion to $152 billion per year.3 It is associated with negative outcomes, such as increased new placements to nursing homes, increased mortality, increased risk of dementia, and further cognitive deterioration among patients with dementia.4-6
Despite its prevalence, delirium may be preventable in a significant percentage of hospitalized patients. Targeted intervention strategies, such as frequent reorientation, maximizing sleep, early mobilization, restricting use of psychoactive medications, and addressing hearing or vision impairment, have been demonstrated to significantly reduce the incidence of hospital delirium.7,8 To achieve these goals, we explored the use of a multimodal strategy centered on nursing education. We integrated consistent, standardized delirium screening and nonpharmacologic interventions as part of a preventative protocol to reduce the incidence of delirium in the hospital ward.
Methods
We evaluated a consecutive series of patients who were admitted to a designated hospital medical-surgical ward within a 25-week period between October 2019 and April 2020. All patients during this period who had at least 1 Confusion Assessment Method (CAM) documented by a nurse during hospitalization met our inclusion criteria. Patients who did not have a CAM documented were excluded from the analysis. Delirium was defined according to the CAM diagnostic algorithm.9
Core nursing staff regularly assigned to the ward completed a multimodal training program designed to improve recognition, documentation, and prevention of hospital delirium. Prior to the training, the nurses completed a 5-point Likert scale survey assessing their level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium. Nurses completed the same survey after the study period ended.
The training curriculum for nurses began with an online module reviewing the epidemiology and risk factors for delirium. Nurses then participated in a series of in-service training sessions led by a team of physicians, during which the CAM and nonpharmacologic delirium prevention measures were reviewed then practiced first-hand. Nursing staff attended an in-person lecture reviewing the current body of literature on delirium risk factors and effective nursing interventions. After formal training was completed, nurses were instructed to document CAM screens

Patients admitted to the hospital unit from the start of the training program (week 1) until the order set was made available (week 15) constituted our control group. The postintervention study group consisted of patients admitted for 10 weeks after the completion of the interventions (weeks 16-25). A timeline of the study events is shown in Figure 1.

Patient demographics and hospital-stay metrics determined a priori were attained via the Cedars-Sinai Enterprise Information Services core. Age, sex, medical history, and incidence of surgery with anesthesia during hospitalization were recorded. The Charlson Comorbidity Index was calculated from patients’ listed diagnoses following discharge. Primary outcomes included incidence of patients with delirium during hospitalization, percentage of tested shifts with positive CAM screens, length of hospital stay, and survival. Secondary outcomes included measures associated with delirium, including the use of chemical restraints, physical restraints, sitters, indwelling urinary catheters, and new psychiatry and neurology consults. Chemical restraints were defined as administration of a new antipsychotic medication or benzodiazepine for the specific indication of hyperactive delirium or agitation.
Statistical analysis was conducted by a statistician, using R version 3.6.3.10P values of < .05 were considered significant. Categorical variables were analyzed using Fisher’s exact test. Continuous variables were analyzed with Welch’s t-test or, for highly skewed continuous variables, with Wilcoxon rank-sum test or Mood’s median test. All patient data were anonymized and stored securely in accordance with institutional guidelines.
Our project was deemed to represent nonhuman subject research and therefore did not require Institutional Review Board (IRB) approval upon review by our institution’s IRB committee and Office of Research Compliance and Quality Improvement. Standards for Quality Improvement Reporting Excellence (SQUIRE 2.0) guidelines were adhered to (Supplementary File can be found at mdedge.com/jcomjournal).
Results
We evaluated 353 patients who met our inclusion criteria: 187 in the control group, and 166 in the postintervention group. Ten patients were readmitted to the ward after their initial discharge; only the initial admission encounters were included in our analysis. Median age, sex, median Charlson Comorbidity Index, and incidence of surgery with anesthesia during hospitalization were comparable between the control and postintervention groups and are summarized in Table 2.

In the control group, 1572 CAMs were performed, with 74 positive CAMs recorded among 27 patients with delirium. In the postintervention group, 1298 CAMs were performed, with 12 positive CAMs recorded among 7 patients with delirium (Figure 2). Primary and secondary outcomes, as well as CAM compliance measures, are summarized in Table 3.

Compared to the control group, the postintervention group had a significant decrease in the incidence of delirium during hospitalization (14.4% vs 4.2%, P = .002) and a significant decrease in the mean percentage of tested nursing shifts with 1 or more positive CAM (4.9% vs 1.1%, P = .002). Significant differences in secondary outcomes between the control and postintervention groups included median length of stay (6 days vs 4 days, P = .004), mean length of stay (8.5 days vs 5.9 days, P = .003), and use of an indwelling urinary catheter (9.1% vs 2.4%, P = .012). There was a trend towards decreased incidence of chemical restraints and psychiatry consults, which did not reach statistical significance. Differences in mortality during hospitalization, physical restraint use, and sitter use could not be assessed due to low incidence.

Compliance with nursing CAM assessments was evaluated. Compared to the control group, the postintervention group saw a significant increase in the percentage of shifts with a CAM performed (54.7% vs 69.1%, P < .001). The median and mean number of CAMs performed per patient were similar between the control and postintervention groups.
Results of nursing surveys completed before and after the training program are listed in Table 4. After training, nurses had a greater level of confidence with recognizing delirium risk factors, preventing delirium, addressing delirium, utilizing the CAM tool, and educating others about delirium.

Discussion
Our study utilized a standardized delirium assessment tool to compare patient cohorts before and after nurse-targeted training interventions on delirium recognition and prevention. Our interventions emphasized nonpharmacologic intervention strategies, which are recommended as first-line in the management of patients with delirium.11 Patients were not excluded from the analysis based on preexisting medical conditions or recent surgery with anesthesia, to allow for conditions that are representative of community hospitals. We also did not use an inclusion criterion based on age; however, the majority of our patients were greater than 70 years old, representing those at highest risk for delirium.2 Significant outcomes among patients in the postintervention group include decreased incidence of delirium, lower average length of stay, decreased indwelling urinary catheter use, and increased compliance with delirium screening by nursing staff.
While the study’s focus was primarily on delirium prevention rather than treatment, these strategies may also have conferred the benefit of reversing delirium symptoms. In addition to measuring incidence of delirium, our primary outcome of percentage of tested shifts with 1 or more positive CAM was intended to assess the overall duration in which patients had delirium during their hospitalization. The reduction in shifts with positive CAMs observed in the postintervention group is notable, given that a significant percentage of patients with hospital delirium have the potential for symptom reversibility.12
Multiple studies have shown that admitted patients who develop delirium experience prolonged hospital stays, often up to 5 to 10 days longer.12-14 The decreased incidence and duration of delirium in our postintervention group is a reasonable explanation for the observed decrease in average length of stay. Our study is in line with previously documented initiatives that show that nonpharmacologic interventions can effectively address downstream health and fiscal sequelae of hospital delirium. For example, a volunteer-based initiative named the Hospital Elder Life Program, from which elements in our order set were modeled after, demonstrated significant reductions in delirium incidence, length of stay, and health care costs.14-16 Other initiatives that focused on educational training for nurses to assess and prevent delirium have also demonstrated similar positive results.17-19 Our study provides a model for effective nursing-focused education that can be reproduced in the hospital setting.
Unlike some other studies, which identified delirium based only on physician assessments, our initiative utilized the CAM performed by floor nurses to identify delirium. While this method
Our study demonstrated an increase in the overall compliance with the CAM screening during the postintervention period, which is significant given the under-recognition of delirium by health care professionals.20 We attribute this increase to greater realized importance and a higher level of confidence from nursing staff in recognizing and addressing delirium, as supported by survey data. While the increased screening of patients should be considered a positive outcome, it also poses the possibility that the observed decrease in delirium incidence in the postintervention group was in fact due to more CAMs performed on patients without delirium. Likewise, nurses may have become more adept at recognizing true delirium, as opposed to delirium mimics, in the latter period of the study.
Perhaps the greatest limitation of our study is the variability in performing and recording CAMs, as some patients had multiple CAMs recorded while others did not have any CAMs recorded. This may have been affected in part by the increase in COVID-19 cases in our hospital towards the latter half of the study, which resulted in changes in nursing assignments as well as patient comorbidities in ways that cannot be easily quantified. Given the limited size of our patient cohorts, certain outcomes, such as the use of sitters, physical restraints, and in-hospital mortality, were unable to be assessed for changes statistically. Causative relationships between our interventions and associated outcome measures are necessarily limited in a binary comparison between control and postintervention groups.
Within these limitations, our study demonstrates promising results in core dimensions of patient care. We anticipate further quality improvement initiatives involving greater numbers of nursing staff and patients to better quantify the impact of nonpharmacologic nursing-centered interventions for preventing hospital delirium.
Conclusion
A multimodal strategy involving nursing-focused training and nonpharmacologic interventions to address hospital delirium is associated with improved patient care outcomes and nursing confidence. Nurses play an integral role in the early recognition and prevention of hospital delirium, which directly translates to reducing burdens in both patient functionality and health care costs. Education and tools to equip nurses to perform standardized delirium screening and interventions should be prioritized.
Acknowledgment: The authors thanks Olena Svetlov, NP, Oscar Abarca, Jose Chavez, and Jenita Gutierrez.
Corresponding author: Jason Ching, MD, Department of Neurology, Cedars-Sinai Medical Center, 8700 Beverly Blvd, Los Angeles, CA 90048; jason.ching@cshs.org.
Financial disclosures: None.
Funding: This research was supported by NIH National Center for Advancing Translational Science (NCATS) UCLA CTSI Grant Number UL1TR001881.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th edition. American Psychiatric Association; 2013.
2. Vasilevskis EE, Han JH, Hughes CG, et al. Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol. 2012;26(3):277-287. doi:10.1016/j.bpa.2012.07003
3. Leslie DL, Marcantonio ER, Zhang Y, et al. One-year health care costs associated with delirium in the elderly population. Arch Intern Med. 2008;168(1):27-32. doi:10.1001/archinternmed.2007.4
4. McCusker J, Cole M, Abrahamowicz M, et al. Delirium predicts 12-month mortality. Arch Intern Med. 2002;162(4):457-463. doi:10.1001/archinte.162.4.457
5. Witlox J, Eurelings LS, de Jonghe JF, et al. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304(4):443-451. doi:10.1001/jama.2010.1013
6. Gross AL, Jones RN, Habtemariam DA, et al. Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324-1331. doi:10.1001/archinternmed.2012.3203
7. Inouye SK. Prevention of delirium in hospitalized older patients: risk factors and targeted intervention strategies. Ann Med. 2000;32(4):257-263. doi:10.3109/07853890009011770
8. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3
9. Inouye SK, van Dyck CH, Alessi CA, et al. Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Intern Med. 1990;113(12):941-948. doi:10.7326/0003-4819-113-12-941
10. R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2017.
11. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. 2009;5(4):210-220. doi:10.1038/nrneurol.2009.24
12. Siddiqi N, House AO, Holmes JD. Occurrence and outcome of delirium in medical in-patients: a systematic literature review. Age Ageing. 2006;35(4):350-364. doi:10.1093/ageing/afl005
13. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291(14):1753-1762. doi:10.1001/jama.291.14.1753
14. Chen CC, Lin MT, Tien YW, et al. Modified Hospital Elder Life Program: effects on abdominal surgery patients. J Am Coll Surg. 2011;213(2):245-252. doi:10.1016/j.jamcollsurg.2011.05.004
15. Zaubler TS, Murphy K, Rizzuto L, et al. Quality improvement and cost savings with multicomponent delirium interventions: replication of the Hospital Elder Life Program in a community hospital. Psychosomatics. 2013;54(3):219-226. doi:10.1016/j.psym.2013.01.010
16. Rubin FH, Neal K, Fenlon K, et al. Sustainability and scalability of the Hospital Elder Life Program at a community hospital. J Am Geriatr Soc. 2011;59(2):359-365. doi:10.1111/j.1532-5415.2010.03243.x
17. Milisen K, Foreman MD, Abraham IL, et al. A nurse-led interdisciplinary intervention program for delirium in elderly hip-fracture patients. J Am Geriatr Soc. 2001;49(5):523-532. doi:10.1046/j.1532-5415.2001.49109.x
18. Lundström M, Edlund A, Karlsson S, et al. A multifactorial intervention program reduces the duration of delirium, length of hospitalization, and mortality in delirious patients. J Am Geriatr Soc. 2005;53(4):622-628. doi:10.1111/j.1532-5415.2005.53210.x
19. Tabet N, Hudson S, Sweeney V, et al. An educational intervention can prevent delirium on acute medical wards. Age Ageing. 2005;34(2):152-156. doi:10.1093/ageing/afi0320. Han JH, Zimmerman EE, Cutler N, et al. Delirium in older emergency department patients: recognition, risk factors, and psychomotor subtypes. Acad Emerg Med. 2009;16(3):193-200. doi:10.1111/j.1553-2712.2008.00339.x