User login
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.
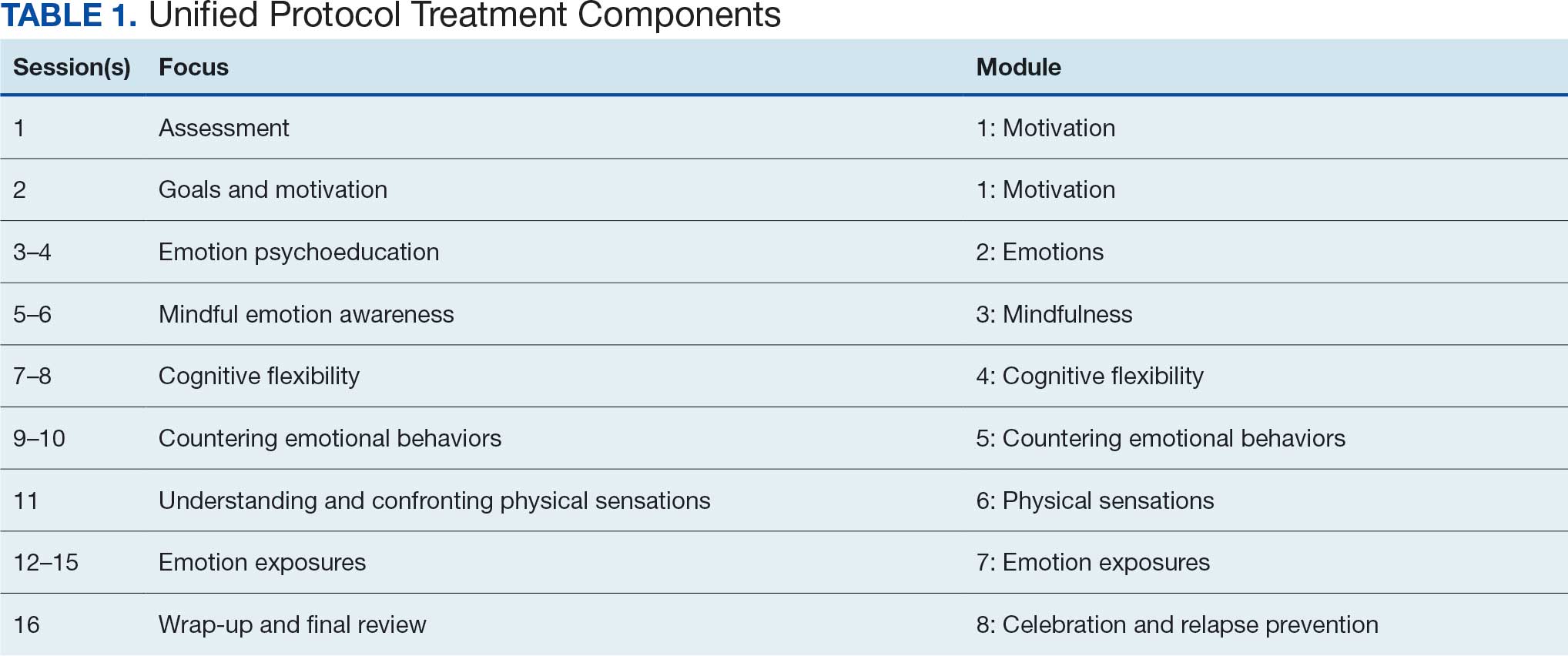
UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
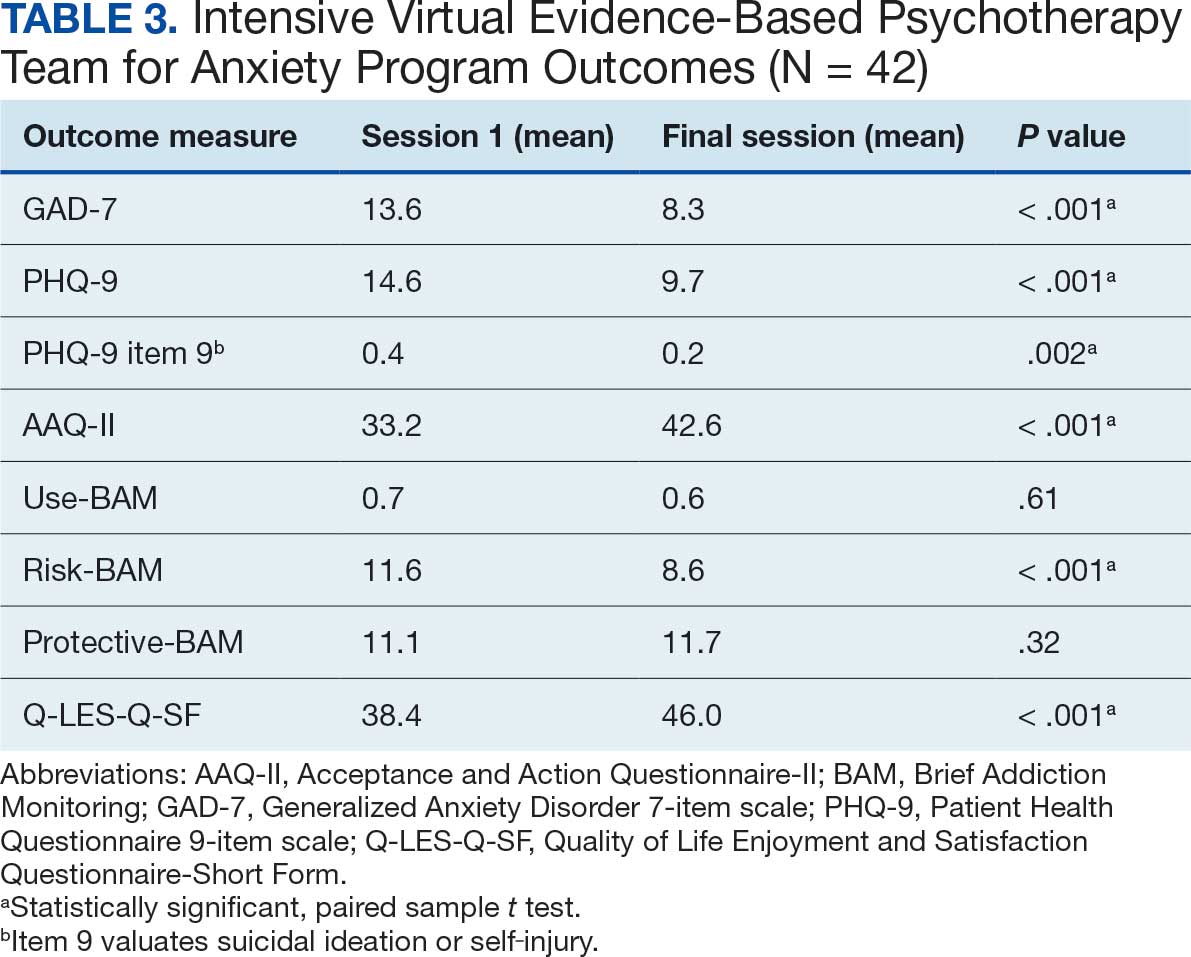
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
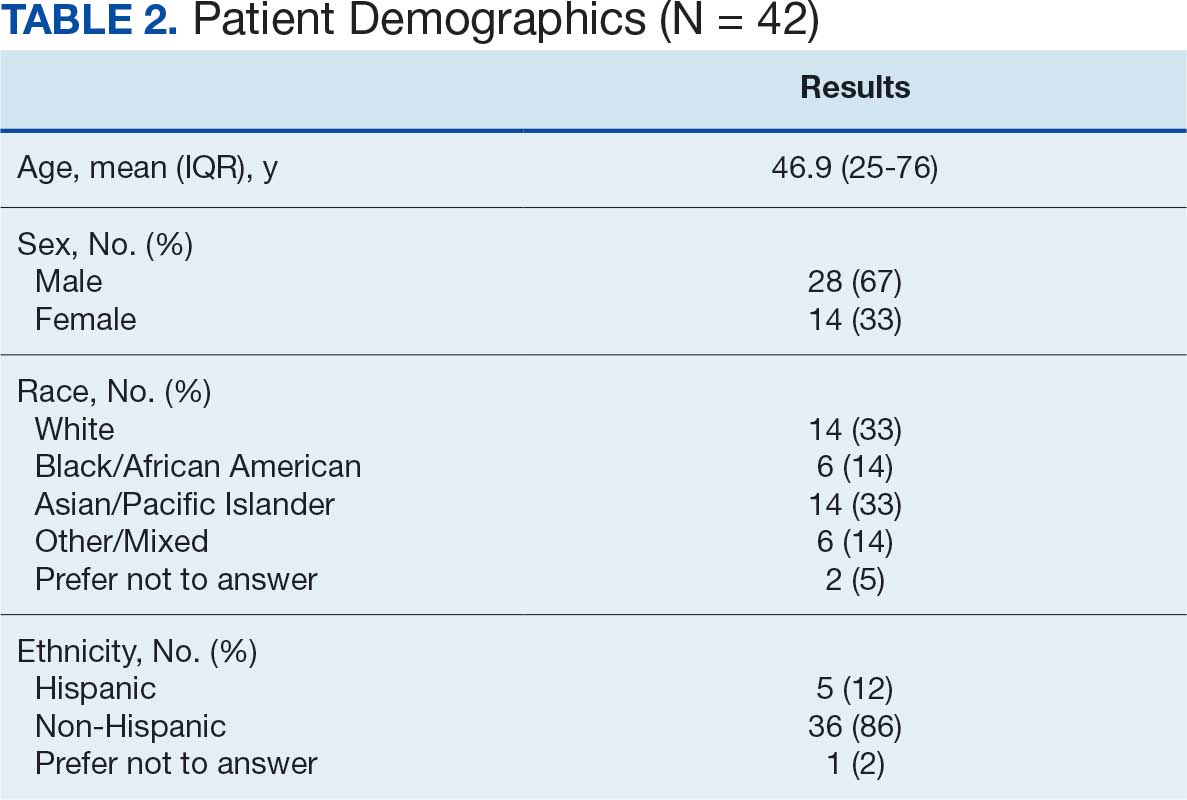
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.
Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).
Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.

UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.

Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).

Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
Cognitive behavioral therapy (CBT) is supported as an effective treatment for depression by clinical practice guidelines from the US Department of Veterans Affairs (VA) and US Department of Defense.1-3 Despite research supporting the use of evidence-based CBT for anxiety, mood, and emotional disorders, patient access to these interventions is limited.4 One barrier to CBT for anxiety, mood, and emotional disorders is the traditional use of single-disorder protocols (SDPs) to separately treat each disorder (eg, major depression, generalized anxiety disorder, panic disorder).
Use of SDPs places a high burden on clinicians, requiring them to learn and competently implement multiple different manualized interventions for each anxiety, mood, or emotional disorder encountered in practice.4 It is common for individuals who present with an anxiety, mood, or emotional disorder to experience co-occurring disorders.5 Traditional SDP-based CBT may require multiple SDPs to address co-occurring disorders, extending time in treatment and increasing training burden. There is evidence that even when an SDP is used, co-occurring difficulties may decrease in intensity.6 Thus, evidence-based CBT for 1 presenting concern may positively affect co-occurring difficulties that are not the primary treatment target.6
Unified Protocol (UP) is a transdiagnostic CBT intervention for anxiety and mood disorders. UP targets emotional experiences (eg, negative affect and emotional distress) that are present in multiple anxiety, depression, and emotional disorders. UP is organized into 8 modules and uses interventions present in other CBT SDPs, such as teaching objective assessment methods to clients, motivational enhancement and goal setting, emotion psychoeducation, mindful awareness, cognitive reframing, and exposure principles as mechanisms of change (Table 1).4 UP is an ideal intervention for addressing a number of anxiety, mood, and emotional disorders as well as addressing cooccurring disorders within the same course of treatment.

UP has been compared to SDPs; studies illustrate that UP is equivalent to SDPs at the end of treatment and at 6 months posttreatment.4,7 Additionally, patients who received UP experienced symptom reduction for multiple co-occurring disorders.7 Furthermore, patients were less likely to drop out of UP when compared with SDPs.4
Studies have reported positive impacts on affective disorders with UP. A 2019 meta-analysis found significant reductions in depression and anxiety-related disorders.8 Additionally, UP has been shown to be effective when delivered in person and via telehealth.9,10 UP has also been successfully used in veterans.11 While traditional models of UP (1-2 sessions weekly) have a lower dropout rate than SDPs, UP and CBT dropout rates still leave room for improvement.12-14 Specifically, rates of attrition from SDP CBT and transdiagnostic CBT protocols range from 9% to 35%, and dropout reduces the likelihood of attaining a full therapeutic dose of any course of CBT. Notably, accelerated delivery of CBT (ie, ≥ 3 sessions/wk) has been shown to reduce risk of dropout.14,15
Veterans are at increased risk for suicide, and anxiety and mood disorders are associated with increased risk of suicide attempt and death.16,17 Very few veterans who could benefit from high-quality CBT interventions, whether SDP or UP, are able to access them. Only 6.3% of veterans received ≥1 session of an evidence-based CBT SDP for posttraumatic stress disorder (PTSD) at 6 clinics evaluated in 2010.18
We identified 2 strategies to increase access to and completion of an evidence-based CBT course. First, the use of transdiagnostic UP instead of SDPs reduced burden on therapists and allowed them to address presenting and co-occurring disorders within the same course of care. Second, the use of an accelerated model of service delivery reduced dropout risk. Training clinicians to deliver UP is efficient and cost-effective, considering clinicians use core strategies that can be tailored and flexibly applied to a range of emotional difficulties. Thus, implementing UP may decrease barriers to receiving an optimal dose of an evidence-based CBT delivered with fidelity in a time- and cost-efficient manner.4 Two studies have found no evidence of differences in outcomes between UP and SDPs, suggesting that training and supervising clinicians in a single transdiagnostic UP intervention may prepare them to treat heterogeneous and co-occurring anxiety, mood, and emotional disorders with less burden than learning multiple SDPs.7,19
Delivering UP in an accelerated or massed format (≥4 sessions/wk) instead of the traditional spaced model (1 session/wk) has empirical support but has not been widely implemented. This approach, sometimes referred to as a UP-intensive outpatient program (UP-IOP) or UP-intensive outpatient track (UP-IOT), has been shown to be feasible, acceptable, and effective, with increased completion rates compared with traditional UP delivery (1-2 sessions/wk).20-22
Ragsdale et al describe a 2-week IOP with multiple treatment tracks, including a general track.20 The general track includes massed UP and additional standard services, including case management, wellness services, family services, and a single session effective behaviors group. Additional augmentation services are available when clinically indicated (eg, repetitive transcranial magnetic stimulation, transcranial direct current stimulation, psychoeducation, motivational interviewing, relapse prevention). In other words, this was an UP-IOP.20
Thompson-Brenner et al described a successful implementation of massed UP applied in intensive treatment settings, such as residential and day-hospital programs, for eating disorders. Patients reported improvements in 3 UP targets: experiential avoidance, mindfulness, and anxiety.21 Watkins et al evaluated a 2-week IOP using CBT for comorbid substance use and mental health disorders, including prolonged exposure, UP, and relapse prevention for substance use disorders. Participants were post-9/11 veterans and activeduty personnel. Results indicated that UP reduced PTSD and depressive symptoms following treatment. Furthermore, the retention rate (91%) was higher than retention in outpatient treatment (39%-65%), supporting the IOP model as a strategy to reduce dropout.22
Massed psychotherapy has been extended to IOP programs for PTSD treatment within the Veterans Health Administration (VHA). Yamokoski et al found that patients who completed an IOP that included massed CBT for PTSD had high retention, high satisfaction, and significant reduction in self-reported co-occurring depression symptoms. The authors also found that this model of care could be implemented and sustained within a VHA facility using minimal staffing resources.23
The UP-IOP models described by Ragsdale et al and Watkins et al included massed UP as the primary evidence-based practice (EBP) with adjunctive groups (eg, wellness and educational groups and access to complementary interventions such as mindfulness and yoga); they found that adding these groups increased retention and patient- reported satisfaction (ie, UP-IOP).20,22 The addition of wellness education alongside a primary EBP aligns with the VHA focus on whole health well-being and wellness. This includes understanding factors that motivate a patient toward health and well-being, providing health education, and offering access to complementary interventions such as mindfulness.24
Dryden et al described the whole health transformation within VHA as a proactive approach to addressing both employee and patient wellness. Their research found that the whole health model promoted wellbeing in patients and staff and these improvements were sustained during the COVID-19 pandemic. Dryden et al also noted that virtual technologies facilitated continued whole health implementation.25
The literature illustrates that (1) massed UP can be delivered with complementary education and wellness offerings that may increase retention and satisfaction by enriching treatment (eg, delivering UP-IOP); (2) whole health, including wellness education and complementary interventions (eg, mindfulness, motivational enhancement), promotes well-being in patients and clinicians; and (3) whole health education and complementary interventions can be delivered virtually.
IVET
Health Care Need
Veterans Affairs Pacific Islands Health Care System (VAPIHCS) provides medical services to veterans in the Hawaiian Islands, American Samoa, Guam, and Saipan spanning nearly 4000 miles across the Pacific Ocean. Prior to implementation of this program, veterans who received care at VAPIHCS had little to no access to UP in outpatient settings and no access to UP in residential settings. Access to UP depended on the presence of a therapist trained in UP within a given clinic and was geographically limited to the location of the UP-trained therapist. The limited outpatient access to UP was restricted to the traditional UP delivery model (eg, about 1 session/wk); thus, there was no access to accelerated UP for veterans served at VAPIHCS. In the fiscal year prior to implementation of the massed UP program, > 1000 VAPIHCS veterans had been diagnosed with obsessive-compulsive related disorder or anxiety. A massed-UP program with weekly rolling admissions would support access to UP for more veterans over 12 months, and the virtual treatment modality would reduce barriers for diverse and underserved veterans, making care more equitable and inclusive.
Successful implementation and sustainment of an EBP prompted the establishment of this UP program. In 2022, VAPIHCS launched the Intensive Virtual Evidence- Based Psychotherapy Team (iVET) for treatment of PTSD. This clinic and associated EBP project demonstrated that massed (≥3 individual sessions/wk) of prolonged exposure (PE) therapy, delivered virtually to a geographically diverse veteran population with PTSD, resulted in significant reductions in PTSD, depression, and anxiety symptoms and substance use risk factors, with very high retention rates. The iVET for PTSD program was feasible, acceptable, and effective, with veterans reporting significant improvement in quality of life and high satisfaction with their mental health services.15 Given the known benefits of transdiagnostic UP treatment (vs SDPs), the need for accelerated UP, and success with accelerated PE, our goal was to spread the EBP of massed (≥3 sessions/ wk) virtual psychotherapy to other presenting problems (eg, anxiety disorders with or without co-occurring unipolar depression) using transdiagnostic UP.
Program Description
The program implemented within outpatient mental health services at VAPIHCS was iVET for the treatment of anxiety with or without co-occurring depression. The program model consists of an accelerated course of UP and whole health education provided via VA Video Connect (VVC), the VA video telehealth platform. iVET is a 2- to 4-week program and consists of 3 parts: (1) massed individual UP for transdiagnostic treatment of anxiety and co-occurring depression, (2) group whole health and wellness classes, and (3) individual health coaching to address personal wellness goals. Programming is offered over 10-hour days to increase access across multiple time zones, especially to allow participation from Guam and Saipan.
When a patient is referred to iVET, the first contact is a video (or telephone) appointment with a registered nurse (RN) for a screening session. The screening session is designed to provide education about the program (including interventions, time commitment, and resources required for participation). Following education, the RN completes a safety screening, including screening for suicidal ideation and risk, as well as intimate partner violence risk. If urgent safety concerns are present, a licensed social worker or psychologist joins the screening to complete further risk assessment and address any safety concerns.
Following screening, patients are scheduled for a VVC telehealth intake with a licensed therapist (social worker or psychologist) to complete a diagnostic interview. Patients are sent a secure link to complete a measurement-based care (MBC) battery of self-report measures, including assessments of demographics, anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care. The results of the diagnostic interview and self-report measures are discussed with the patient during the intake session to plan next steps and support shared decision-making. This initial VVC intake not only allows for fit assessment but also serves to troubleshoot technical difficulties with the virtual platforms.
Notably, there are minimal exclusion criteria for participation in iVET. These include active unmanaged psychosis or manic symptoms, recent suicidal crises (attempt within 8 weeks), active nonsuicidal self-injury (within 8 weeks), and moderate to severe cognitive impairment. Following intake, patients are scheduled to begin their course of care with iVET. Upon completion of intake, patients receive program materials for individual and group classes and are told they will receive email links for all VVC telehealth appointments. Patients are admitted to the iVET on a rolling basis, thereby increasing access compared with closed group and/or cohort models of care.
Patients receiving iVET attend 2 to 4 telehealth appointments daily and complete exercises between sessions. The primary iVET for Anxiety program EBP intervention is a massed or accelerated individual course of UP, which includes 8 primary components: assessment, goals and motivation, understanding emotions, mindful emotion awareness, cognitive flexibility, countering emotional behaviors, understanding and confronting physical sensations, and emotional exposures. UP is delivered in 4 to 8 individual sessions weekly (60-90 minutes each), allowing completion of the full UP protocol with fidelity in 2 to 4 weeks. In addition to primary EBP intervention, patients participate in 4 group sessions weekly (50 minutes each) of a whole health and wellness education class and have access to 1 individual health coaching session weekly (30- 60 minutes) to set wellness goals and receive coaching. During iVET, patients are invited to complete MBC batteries of self-report measures assessing anxiety symptoms, depression symptoms, substance use, psychological flexibility, quality of life, and satisfaction with mental health care at sessions 1, 5, 9, 13, and the final UP session. Following discharge from the iVET, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with a therapist, during which they are invited to complete MBC measures and review relapse prevention and maintenance of treatment gains. Likewise, patients are offered 1-, 3-, and 6-month individual postdischarge check-up sessions with an RN focused on maintaining wellness gains.
The iVET for Anxiety staff has 3 therapists (psychologists or social workers) and 1 RN. Additionally, the iVET for Anxiety is supported by a program manager and a program support assistant who support 2 programs total (the iVET for Anxiety plus another mental health program). The primary cost of the program is staff salary. Additional resources included computer equipment for staff and supplies (eg, printed materials for patients and office supplies). Due to the virtual environment of care, iVET staff telework and do not require physical space within VAPIHCS.
Outcomes
Veterans receiving iVET for Anxiety are invited to complete MBC multiple times, including pretreatment, during UP treatment (sessions 1, 5, 9, 13, and the final session), and posttreatment (1, 3, and 6 months). MBC measures include self-reported demographics; a 2-item measure of satisfaction with mental health services; the Acceptance and Action Questionnaire II,26 the Brief Addiction Monitor-Intensive Outpatient Program, 27 the Generalized Anxiety Disorder-7,28 the Patient Health Questionnaire (PHQ-9),29 and the Quality of Life Enjoyment and Satisfaction Questionnaire-Short Form.30
Forty-two veterans completed the iVET for Anxiety program, with a retention rate of 87.5% completing . 16 sessions of massed UP (Table 2). Veterans reported reduced anxiety (P < .001), depression (P < .001), and substance use risk (P < .001). Veterans experienced improved acceptance of emotional experiences (P < .001) and quality of life (P < .001), based on paired sample t tests comparing session 1 vs final session scores on the self-reported measures. Veterans also reported high satisfaction with mental health care in iVET for Anxiety.

Veterans experienced reduced thoughts of death or suicidal ideation (SI) based on responses to item 9 of PHQ-9. When SI was categorically analyzed (presence vs absence) using PHQ-9 item 9, there was an association observed between absence of SI and completion of a course of massed UP that does not appear to be due to chance, (X2 [1, N = 42) = 3.94; P = .047). In addition, veterans who completed the program showed a significant decrease in SI severity measured continuously (range, 0-3) on PHQ-9 item 9 (P < .05) (Table 3).

Future Directions
The aim of this program is to see sustained patient outcomes as iVET continues to serve more veterans. Another line of inquiry is longer- term follow-up, given that long-term follow- up was not addressed in this project. We also hope that the accelerated model of care can be applied to treatment of other presenting concerns (eg, relationship difficulties, insomnia). Expansion of accelerated mental health treatment into other federal and nonfederal health care settings is another area worthy of future inquiry. Exploration of staff satisfaction and burnout related to providing accelerated UP is another important future direction. Relatedly, assessment of the staff burden to learn 1 transdiagnostic EBP vs learning multiple SDPs is another future direction. Likewise, exploration of institutional benefits of investment in transdiagnostic training, supervision, and consultation for UP vs multiple SDPs may be important. These areas could also result in insightful, beneficial evidence of the effectiveness of massed UP to add to the existing literature.
Conclusions
UP for transdiagnostic treatment of anxiety, depressive, and emotional disorders has demonstrated reduced suffering and improved functioning and is supported by multiple clinical practice guidelines.1-4 Federal practitioners are positioned to improve access to this intervention, thereby reducing pain and improving lives. Indeed, it is crucial to envision a future state in which access to UP for a range of anxiety and depressive disorders is improved and broad, retention rates are dramatically improved, and clinicians providing UP do not experience the high burden and burnout associated with needing to learn and implement a variety of SDPs. Development of these programs, or similar tracks within existing programs, that provide massed or accelerated UP for transdiagnostic treatment of a range of anxiety and depressive disorders with virtual delivery options, is imperative to advance improved care for patients and clinicians.
Federal health care settings treating patients with anxiety and depression, such as those within the US Department of Defense, Indian Health Services, Bureau of Prisons, and VHA, are positioned to implement programs like iVET. Moreover, at the institutional level, investment in training and supervision in the transdiagnostic UP as opposed to multiple SDPs warrants consideration. We believe this model of care has great merit and foresee a future where all patients seeking treatment for anxiety and depression have the option to complete an accelerated or massed course of transdiagnostic care with UP if they so desire. Our experiences with iVET illustrate the feasibility, acceptability, and sustainability of such programs without requiring substantial staffing and financial resources.
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
- US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the management of major depressive disorder. Version 4. 2022. Accessed February 1, 2026. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFinal508.pdf
- American Psychological Association. Clinical practice guideline for the treatment of depression across three age cohorts. February 2019. Accessed February 4, 2026. https://www.apa.org/depression-guideline/guideline.pdf
- Katzman MA, Bleau P, Blier P, et al. Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive compulsive disorders. BMC Psychiatry. 2014;14:S1. doi:10.1186/1471-244X-14-S1-S1
- Barlow DH, Farchione TJ, Bullis JR, et al. The unified protocol for transdiagnostic treatment of emotional disorders compared with diagnosis-specific protocols for anxiety disorders: a randomized clinical trial. JAMA Psychiatry. 2017;74:875-884. doi:10.1001/jamapsychiatry.2017.2164
- Calkins AW, et al. Comorbidity of anxiety and depression. In: Ressler KJ, Pine DS, Rothbaum BO, eds. Anxiety Disorders, Primer On. Oxford Academic; 2015. https://doi.org/10.1093/med/9780199395125.003.0021
- Manber R, Edinger JD, Gress JL, et al. Cognitive behavioral therapy for insomnia enhances depression outcome in patients with comorbid major depressive disorder and insomnia. Sleep. 2008;31:489-495. doi:10.1093/sleep/31.4.489
- Steele SJ, Farchione TJ, Cassiello-Robbins C, et al. Efficacy of the Unified Protocol for transdiagnostic treatment of comorbid psychopathology accompanying emotional disorders compared to treatments targeting single disorders. J Psychiatr Res. 2018;104:211-216. doi:10.1016/j.jpsychires.2018.08.005
- Sakiris N, Berle D. A systematic review and meta-analysis of the Unified Protocol as a transdiagnostic emotion regulation based intervention. Clin Psychol Rev. 2019;72:101751. doi:10.1016/j.cpr.2019.101751
- Cassiello-Robbins C, Rosenthal MZ, Ammirati RJ. Delivering transdiagnostic treatment over telehealth during the COVID-19 pandemic: application of the unified protocol. Cogn Behav Pract. 2021;28:555-572. doi:10.1016/j.cbpra.2021.04.007
- Meyer EC, Coe E, Pennington ML, et al. The unified protocol for transdiagnostic treatment of emotional disorders delivered to firefighters via videoconferencing: pilot outcomes highlighting improvements in alcohol use disorder and posttraumatic stress disorder symptoms. Cogn Behav Pract. 2024;31:215-229. doi:10.1016/j.cbpra.2022.08.004
- Varkovitzky RL, Sherrill AM, Reger GM. Effectiveness of the unified protocol for transdiagnostic treatment of emotional disorders among veterans with posttraumatic stress disorder: a pilot study. Behav Modif. 2018;42:210-230. doi:10.1177/0145445517724539
- Oliveira JT, Sousa I, Ribeiro AP, et al. Premature termination of the unified protocol for the transdiagnostic treatment of emotional disorders: The role of ambivalence towards change. Clin Psychol Psychother. 2022;29:1089-1100. doi:10.1002/cpp.2694
- Schaeuffele C, Homeyer S, Perea L, et al. The unified protocol as an internet-based intervention for emotional disorders: Randomized controlled trial. PLoS One. 2022;17:e0270178. doi:10.1371/journal.pone.0270178
- Bentley KH, Cohen ZD, Kim T, et al. The nature, timing, and symptom trajectories of dropout from transdiagnostic and single-diagnosis cognitive-behavioral therapy for anxiety disorders. Behav Ther. 2021;52:1364-1376. doi:10.1016/j.beth.2021.03.007
- Aosved AC, Brown TB, Bell JC, et al. Accelerated prolonged exposure therapy for posttraumatic stress disorder in a Veterans Health Administration system. Fed Pract. 2025;42:S6-S11. doi:10.12788/fp.0568
- Nepon J, Belik SL, Bolton J, et al. The relationship between anxiety disorders and suicide attempts: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. Depress Anxiety. 2010;27:791-798. doi:10.1002/da.20674
- Shiner B, D’Avolio LW, Nguyen TM, et al. Measuring use of evidence based psychotherapy for posttraumatic stress disorder. Adm Policy Ment Health. 2013;40:311-318. doi:10.1007/s10488-012-0421-0
- Nichter B, Stein MB, Monteith LL, et al. Risk factors for suicide attempts among U.S. military veterans: A 7-year population-based, longitudinal cohort study. Suicide Life Threat Behav. 2022;52:303-316. doi:10.1111/sltb.12822
- McHugh RK, Barlow DH. The dissemination and implementation of evidence-based psychological treatments. A review of current efforts. Am Psychol. 2010;65:73-84. doi:10.1037/a0018121
- Ragsdale KA, Nichols AA, Mehta M, et al. Comorbid treatment of traumatic brain injury and mental health disorders. NeuroRehabilitation. 2024;55:375-384. doi:10.3233/NRE-230235
- Thompson-Brenner H, Brooks GE, Boswell JF, et al. Evidence-based implementation practices applied to the intensive treatment of eating disorders: summary of research and illustration of principles using a case example. Clin Psychol Sci Pract. 2018;25:e12221. doi:10.1111/cpsp.12221
- Watkins LE, Patton SC, Drexler K, et al. Clinical effectiveness of an intensive outpatient program for integrated treatment of comorbid substance abuse and mental health disorders. Cog Behav Pract. 2023;30:354-366.
- Yamokoski C, Flores H, Facemire V, et al. Feasibility of an intensive outpatient treatment program for post-traumatic stress disorder within the veterans health care administration. Psychol Serv. 2023;20:506-515. doi:10.1037/ser0000628
- Gaudet T, Kligler B. Whole health in the whole system of the Veterans Administration: how will we know we have reached this future state?. J Altern Complement Med. 2019;25:S7-S11. doi:10.1089/acm.2018.29061.gau
- Dryden EM, Bolton RE, Bokhour BG, et al. Leaning into whole health: sustaining system transformation while supporting patients and employees during COVID-19. Glob Adv Health Med. 2021;10:21649561211021047. doi:10.1177/21649561211021047
- Bond FW, Hayes SC, Baer RA, et al. Preliminary psychometric properties of the Acceptance and Action Questionnaire-II: a revised measure of psychological inflexibility and experiential avoidance. Behav Ther. 2011;42:676-688. doi:10.1016/j.beth.2011.03.007
- Cacciola JS, Alterman AI, Dephilippis D, et al. Development and initial evaluation of the Brief Addiction Monitor (BAM). J Subst Abuse Treat. 2013;44:256-263. doi:10.1016/j.jsat.2012.07.013
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097. doi:10.1001/archinte.166.10.1092
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613. doi:10.1046/j.1525-1497.2001.016009606.x
- Stevanovic D. Quality of life enjoyment and satisfaction questionnaire-short form for quality of life assessments in clinical practice: a psychometric study. J Psychiatr Ment Health Nurs. 2011;18:744-750. doi:10.1111/j.1365-2850.2011.01735.x
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
Accelerated Unified Protocol for Transdiagnostic Treatment of Anxiety Disorders in a VHA System
'Energy Insecurity' Tied to Anxiety, Depression Risk
'Energy Insecurity' Tied to Anxiety, Depression Risk
TOPLINE:
Energy insecurity, the inability to meet household energy needs, was associated with more than twice the odds of having depression and anxiety symptoms than energy security in US adults, a new cross-sectional study showed.
METHODOLOGY:
- Using data from the US Census Bureau's online Household Pulse Survey, administered between 2022 and 2024, researchers conducted a cross-sectional study with a weighted population of > 187 million US adults (51% women; 64% White, 16% Hispanic, 10% Black, and 5% Asian). About a quarter of the population was in each of 4 age groups: 18-34 years, 35-49 years, 50-64 years, and ≥ 65 years.
- Three indicators of energy insecurity—inability to pay energy bills, maintaining unsafe/unhealthy home temperatures, and forgoing expenses on basic necessities to pay energy bills—were assessed individually and as a composite measure.
- Mental health was assessed using modified versions of the 2-item Patient Health Questionnaire for depression and the 2-item Generalized Anxiety Disorder scale for anxiety.
- The analysis was adjusted for other social determinants of health, including unemployment, housing instability, and food insecurity. Covariates included a wide range of factors, such as age, educational level, sex, and annual household income.
TAKEAWAY:
- In all, > 43% of the population reported having ≥ 1 form of energy security; around 22% reported being unable to pay energy bills, 22% maintained unsafe home temperatures, and nearly 34% forewent spending on basic necessities to pay energy bills.
- Individuals who gave up spending on basic necessities to pay energy bills had higher odds of anxiety (adjusted odds ratio [aOR], 1.79) and depression (aOR, 1.74) than those who did not.
- Adults with energy insecurity on the composite measure had higher odds for anxiety (aOR, 2.29) and depression (aOR, 2.31) than those with energy security.
- Food insecurity was also associated with poorer mental health, with higher odds for symptoms of depression (aOR, 2.05) and anxiety (aOR, 2.07).
IN PRACTICE:
"Despite its high prevalence, energy insecurity remains underrecognized in public health and policy intervention strategies," the investigators wrote.
"These findings suggest that energy insecurity is a widespread and important factor associated with mental health symptoms and may warrant consideration in efforts to reduce adverse mental health outcomes," they added.
SOURCE:
This study was led by Michelle Graf, PhD, Carter School of Public Policy, Georgia Institute of Technology, Atlanta. It was published online on October 27 in JAMA Network Open.
LIMITATIONS:
The cross-sectional nature of the data limited causal interference and increased the possibility of reverse causality. The questionnaire captured subjective interpretations of unsafe and unhealthy indoor temperatures, which may have varied among respondents. Additionally, the recall periods for energy insecurity and mental health outcomes were different.
DISCLOSURES:
The investigators reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Energy insecurity, the inability to meet household energy needs, was associated with more than twice the odds of having depression and anxiety symptoms than energy security in US adults, a new cross-sectional study showed.
METHODOLOGY:
- Using data from the US Census Bureau's online Household Pulse Survey, administered between 2022 and 2024, researchers conducted a cross-sectional study with a weighted population of > 187 million US adults (51% women; 64% White, 16% Hispanic, 10% Black, and 5% Asian). About a quarter of the population was in each of 4 age groups: 18-34 years, 35-49 years, 50-64 years, and ≥ 65 years.
- Three indicators of energy insecurity—inability to pay energy bills, maintaining unsafe/unhealthy home temperatures, and forgoing expenses on basic necessities to pay energy bills—were assessed individually and as a composite measure.
- Mental health was assessed using modified versions of the 2-item Patient Health Questionnaire for depression and the 2-item Generalized Anxiety Disorder scale for anxiety.
- The analysis was adjusted for other social determinants of health, including unemployment, housing instability, and food insecurity. Covariates included a wide range of factors, such as age, educational level, sex, and annual household income.
TAKEAWAY:
- In all, > 43% of the population reported having ≥ 1 form of energy security; around 22% reported being unable to pay energy bills, 22% maintained unsafe home temperatures, and nearly 34% forewent spending on basic necessities to pay energy bills.
- Individuals who gave up spending on basic necessities to pay energy bills had higher odds of anxiety (adjusted odds ratio [aOR], 1.79) and depression (aOR, 1.74) than those who did not.
- Adults with energy insecurity on the composite measure had higher odds for anxiety (aOR, 2.29) and depression (aOR, 2.31) than those with energy security.
- Food insecurity was also associated with poorer mental health, with higher odds for symptoms of depression (aOR, 2.05) and anxiety (aOR, 2.07).
IN PRACTICE:
"Despite its high prevalence, energy insecurity remains underrecognized in public health and policy intervention strategies," the investigators wrote.
"These findings suggest that energy insecurity is a widespread and important factor associated with mental health symptoms and may warrant consideration in efforts to reduce adverse mental health outcomes," they added.
SOURCE:
This study was led by Michelle Graf, PhD, Carter School of Public Policy, Georgia Institute of Technology, Atlanta. It was published online on October 27 in JAMA Network Open.
LIMITATIONS:
The cross-sectional nature of the data limited causal interference and increased the possibility of reverse causality. The questionnaire captured subjective interpretations of unsafe and unhealthy indoor temperatures, which may have varied among respondents. Additionally, the recall periods for energy insecurity and mental health outcomes were different.
DISCLOSURES:
The investigators reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
Energy insecurity, the inability to meet household energy needs, was associated with more than twice the odds of having depression and anxiety symptoms than energy security in US adults, a new cross-sectional study showed.
METHODOLOGY:
- Using data from the US Census Bureau's online Household Pulse Survey, administered between 2022 and 2024, researchers conducted a cross-sectional study with a weighted population of > 187 million US adults (51% women; 64% White, 16% Hispanic, 10% Black, and 5% Asian). About a quarter of the population was in each of 4 age groups: 18-34 years, 35-49 years, 50-64 years, and ≥ 65 years.
- Three indicators of energy insecurity—inability to pay energy bills, maintaining unsafe/unhealthy home temperatures, and forgoing expenses on basic necessities to pay energy bills—were assessed individually and as a composite measure.
- Mental health was assessed using modified versions of the 2-item Patient Health Questionnaire for depression and the 2-item Generalized Anxiety Disorder scale for anxiety.
- The analysis was adjusted for other social determinants of health, including unemployment, housing instability, and food insecurity. Covariates included a wide range of factors, such as age, educational level, sex, and annual household income.
TAKEAWAY:
- In all, > 43% of the population reported having ≥ 1 form of energy security; around 22% reported being unable to pay energy bills, 22% maintained unsafe home temperatures, and nearly 34% forewent spending on basic necessities to pay energy bills.
- Individuals who gave up spending on basic necessities to pay energy bills had higher odds of anxiety (adjusted odds ratio [aOR], 1.79) and depression (aOR, 1.74) than those who did not.
- Adults with energy insecurity on the composite measure had higher odds for anxiety (aOR, 2.29) and depression (aOR, 2.31) than those with energy security.
- Food insecurity was also associated with poorer mental health, with higher odds for symptoms of depression (aOR, 2.05) and anxiety (aOR, 2.07).
IN PRACTICE:
"Despite its high prevalence, energy insecurity remains underrecognized in public health and policy intervention strategies," the investigators wrote.
"These findings suggest that energy insecurity is a widespread and important factor associated with mental health symptoms and may warrant consideration in efforts to reduce adverse mental health outcomes," they added.
SOURCE:
This study was led by Michelle Graf, PhD, Carter School of Public Policy, Georgia Institute of Technology, Atlanta. It was published online on October 27 in JAMA Network Open.
LIMITATIONS:
The cross-sectional nature of the data limited causal interference and increased the possibility of reverse causality. The questionnaire captured subjective interpretations of unsafe and unhealthy indoor temperatures, which may have varied among respondents. Additionally, the recall periods for energy insecurity and mental health outcomes were different.
DISCLOSURES:
The investigators reported no relevant conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
'Energy Insecurity' Tied to Anxiety, Depression Risk
'Energy Insecurity' Tied to Anxiety, Depression Risk
Americans’ Top Causes of Anxiety Revealed
What current events are keeping Americans up at night? The economy, gun violence, and hate crimes top the list, results from a newly released American Psychiatric Association (APA) survey showed.
Anxiety about international conflicts — namely, the Russia-Ukraine and Israel-Hamas wars — also remains high.
“While we like to stay informed, the news can also impact our mental health, and being mindful of that impact is important. If current events seem overwhelming it may be time to limit your news consumption,” APA CEO and Medical Director Marketa M. Wills, MD, MBD, said in a statement.
Survey results also revealed the election and the holidays were common sources of stress.
“Election stress is common, and it’s important to recognize that, as we’re spending more time with family around the holidays, we might need to have a strategy to manage our own mental health during these times,” Howard Liu, MD, MBA, chair of the Department of Psychiatry, University of Nebraska Medical Center, Omaha, told this news organization.
“As with any difficult topic, we all have different levels of avoidance or desire to engage, and it’s okay to set boundaries based on past conversations with family. I think sometimes we get drawn into arguments that we don’t want to have or may not be productive for either side,” said Liu, who chairs the APA Council on Communications.
In line with trends throughout 2024, adults polled by the APA in November were most anxious about the economy (75%), gun violence (64%), and hate crimes (60%). The survey included 2200 US adults as part of the APA’s Healthy Minds monthly series.
Anxiety about international conflicts remained high in November at 57% — but was down from 65% in August.
Election anxiety remained high in mid-November but not as high as before the election. In August, 72% of Americans said they were anxious about the 2024 election. In November, just after the election, 50% reported anxiety over the election outcome.
“I think the anticipation of change can sometimes be worse than the change itself. So I think a lot of people are now taking the attitude of — let’s wait and see what actually happens,” said Liu.
Half the adults (50%) anticipate the same amount of stress as the 2023 holiday season, while almost one third expect more stress (28%), and one fourth anticipate less stress (23%).
When asked how the holidays generally affect their mental health, 38% said it has positive effects, and 21% said the opposite was true.
Anxiety About the Future
After a divisive election, most Americans were ready to avoid politics at holiday gatherings, results of a separate poll conducted by the American Psychological Association in late November showed.
That poll, which included 2000 US adults, showed that more than 7 in 10 (72%) said they wanted to avoid talking about politics with family and friends over the holidays.
In addition, nearly two in five adults (39%) reported they were stressed by the thought of politics being raised at holiday gatherings and would try to avoid family members they disagree with. Younger adults were significantly more likely than were their older counterparts to report they planned to avoid family over the holidays.
The future of the nation also weighs on the minds of many Americans.
Another poll conducted by the American Psychological Association in August prior to the 2024 US presidential election showed that 77% of respondents said the future of the nation was a significant source of stress for them.
In the postelection poll, more than one third of adults (35%) said they are more stressed about the future of the nation now than they were leading up to the election, and another third reported they are now less stressed (32%). A quarter of adults (24%) said their stress about the future of the nation was unchanged, and 9% said they were not stressed about the future of the nation then or now.
“There’s still clearly a lot of uncertainty, and there’s a lot of activity right now for the president-elect,” which can contribute to anxiety, C. Vaile Wright, PhD, psychologist, researcher and spokesperson for the American Psychological Association, told this news organization.
These data also show that many Americans have little or no trust in the government, with some wanting to leave the United States.
“It’s a reflection of the lack of strong leadership across the board in this country. We have a governmental system in place that does not seem to serve the people, but to serve corporations and maintenance of power. I think people are disillusioned with it and that creates a lack of trust and hopelessness,” Wright noted.
Liu and Wright reported no relevant disclosures.
A version of this article appeared on Medscape.com.
What current events are keeping Americans up at night? The economy, gun violence, and hate crimes top the list, results from a newly released American Psychiatric Association (APA) survey showed.
Anxiety about international conflicts — namely, the Russia-Ukraine and Israel-Hamas wars — also remains high.
“While we like to stay informed, the news can also impact our mental health, and being mindful of that impact is important. If current events seem overwhelming it may be time to limit your news consumption,” APA CEO and Medical Director Marketa M. Wills, MD, MBD, said in a statement.
Survey results also revealed the election and the holidays were common sources of stress.
“Election stress is common, and it’s important to recognize that, as we’re spending more time with family around the holidays, we might need to have a strategy to manage our own mental health during these times,” Howard Liu, MD, MBA, chair of the Department of Psychiatry, University of Nebraska Medical Center, Omaha, told this news organization.
“As with any difficult topic, we all have different levels of avoidance or desire to engage, and it’s okay to set boundaries based on past conversations with family. I think sometimes we get drawn into arguments that we don’t want to have or may not be productive for either side,” said Liu, who chairs the APA Council on Communications.
In line with trends throughout 2024, adults polled by the APA in November were most anxious about the economy (75%), gun violence (64%), and hate crimes (60%). The survey included 2200 US adults as part of the APA’s Healthy Minds monthly series.
Anxiety about international conflicts remained high in November at 57% — but was down from 65% in August.
Election anxiety remained high in mid-November but not as high as before the election. In August, 72% of Americans said they were anxious about the 2024 election. In November, just after the election, 50% reported anxiety over the election outcome.
“I think the anticipation of change can sometimes be worse than the change itself. So I think a lot of people are now taking the attitude of — let’s wait and see what actually happens,” said Liu.
Half the adults (50%) anticipate the same amount of stress as the 2023 holiday season, while almost one third expect more stress (28%), and one fourth anticipate less stress (23%).
When asked how the holidays generally affect their mental health, 38% said it has positive effects, and 21% said the opposite was true.
Anxiety About the Future
After a divisive election, most Americans were ready to avoid politics at holiday gatherings, results of a separate poll conducted by the American Psychological Association in late November showed.
That poll, which included 2000 US adults, showed that more than 7 in 10 (72%) said they wanted to avoid talking about politics with family and friends over the holidays.
In addition, nearly two in five adults (39%) reported they were stressed by the thought of politics being raised at holiday gatherings and would try to avoid family members they disagree with. Younger adults were significantly more likely than were their older counterparts to report they planned to avoid family over the holidays.
The future of the nation also weighs on the minds of many Americans.
Another poll conducted by the American Psychological Association in August prior to the 2024 US presidential election showed that 77% of respondents said the future of the nation was a significant source of stress for them.
In the postelection poll, more than one third of adults (35%) said they are more stressed about the future of the nation now than they were leading up to the election, and another third reported they are now less stressed (32%). A quarter of adults (24%) said their stress about the future of the nation was unchanged, and 9% said they were not stressed about the future of the nation then or now.
“There’s still clearly a lot of uncertainty, and there’s a lot of activity right now for the president-elect,” which can contribute to anxiety, C. Vaile Wright, PhD, psychologist, researcher and spokesperson for the American Psychological Association, told this news organization.
These data also show that many Americans have little or no trust in the government, with some wanting to leave the United States.
“It’s a reflection of the lack of strong leadership across the board in this country. We have a governmental system in place that does not seem to serve the people, but to serve corporations and maintenance of power. I think people are disillusioned with it and that creates a lack of trust and hopelessness,” Wright noted.
Liu and Wright reported no relevant disclosures.
A version of this article appeared on Medscape.com.
What current events are keeping Americans up at night? The economy, gun violence, and hate crimes top the list, results from a newly released American Psychiatric Association (APA) survey showed.
Anxiety about international conflicts — namely, the Russia-Ukraine and Israel-Hamas wars — also remains high.
“While we like to stay informed, the news can also impact our mental health, and being mindful of that impact is important. If current events seem overwhelming it may be time to limit your news consumption,” APA CEO and Medical Director Marketa M. Wills, MD, MBD, said in a statement.
Survey results also revealed the election and the holidays were common sources of stress.
“Election stress is common, and it’s important to recognize that, as we’re spending more time with family around the holidays, we might need to have a strategy to manage our own mental health during these times,” Howard Liu, MD, MBA, chair of the Department of Psychiatry, University of Nebraska Medical Center, Omaha, told this news organization.
“As with any difficult topic, we all have different levels of avoidance or desire to engage, and it’s okay to set boundaries based on past conversations with family. I think sometimes we get drawn into arguments that we don’t want to have or may not be productive for either side,” said Liu, who chairs the APA Council on Communications.
In line with trends throughout 2024, adults polled by the APA in November were most anxious about the economy (75%), gun violence (64%), and hate crimes (60%). The survey included 2200 US adults as part of the APA’s Healthy Minds monthly series.
Anxiety about international conflicts remained high in November at 57% — but was down from 65% in August.
Election anxiety remained high in mid-November but not as high as before the election. In August, 72% of Americans said they were anxious about the 2024 election. In November, just after the election, 50% reported anxiety over the election outcome.
“I think the anticipation of change can sometimes be worse than the change itself. So I think a lot of people are now taking the attitude of — let’s wait and see what actually happens,” said Liu.
Half the adults (50%) anticipate the same amount of stress as the 2023 holiday season, while almost one third expect more stress (28%), and one fourth anticipate less stress (23%).
When asked how the holidays generally affect their mental health, 38% said it has positive effects, and 21% said the opposite was true.
Anxiety About the Future
After a divisive election, most Americans were ready to avoid politics at holiday gatherings, results of a separate poll conducted by the American Psychological Association in late November showed.
That poll, which included 2000 US adults, showed that more than 7 in 10 (72%) said they wanted to avoid talking about politics with family and friends over the holidays.
In addition, nearly two in five adults (39%) reported they were stressed by the thought of politics being raised at holiday gatherings and would try to avoid family members they disagree with. Younger adults were significantly more likely than were their older counterparts to report they planned to avoid family over the holidays.
The future of the nation also weighs on the minds of many Americans.
Another poll conducted by the American Psychological Association in August prior to the 2024 US presidential election showed that 77% of respondents said the future of the nation was a significant source of stress for them.
In the postelection poll, more than one third of adults (35%) said they are more stressed about the future of the nation now than they were leading up to the election, and another third reported they are now less stressed (32%). A quarter of adults (24%) said their stress about the future of the nation was unchanged, and 9% said they were not stressed about the future of the nation then or now.
“There’s still clearly a lot of uncertainty, and there’s a lot of activity right now for the president-elect,” which can contribute to anxiety, C. Vaile Wright, PhD, psychologist, researcher and spokesperson for the American Psychological Association, told this news organization.
These data also show that many Americans have little or no trust in the government, with some wanting to leave the United States.
“It’s a reflection of the lack of strong leadership across the board in this country. We have a governmental system in place that does not seem to serve the people, but to serve corporations and maintenance of power. I think people are disillusioned with it and that creates a lack of trust and hopelessness,” Wright noted.
Liu and Wright reported no relevant disclosures.
A version of this article appeared on Medscape.com.
California Seeks Mental Health Warning Labels on Social Media
In the latest effort to address the mental health crisis among adolescents, legislation in California would require social media platforms to come with a “black box” mental health warning label.
Despite growing evidence linking young people’s use of social media to significant health risks including depression, anxiety, and suicidal thoughts, social media companies have failed to be transparent about the risks, said Assembly member Rebecca Bauer-Kahan (D-Orinda), who introduced Assembly Bill (AB) 56.
“AB 56 ensures that families are armed with clear, actionable information to understand these dangers and make decisions that prioritizes their children’s well-being,” she said in a press release.
Bauer-Kahan noted that 95% of teens report using at least one social media platform and that more than one third say they use social media almost constantly.
“There is a powerful profit motive to keep our young people hooked online and engaged and it is exploiting the human psychology with notifications, likes, endless scrolling, and algorithmic amplification that is harming our children every day,” she said at a press conference on December 9 announcing the bill.
, a sponsor of AB 56, said in a press release.
Speaking at the press conference, Bonta said social media has many “incredible benefits” from giving people an outlet of expression to providing access to critical information but “there is no disputing the fact, it can have an enormously detrimental and dangerous impact on our young people. You cannot debate that. Our children are suffering.”
If AB 56 is successful, he said social media platforms would be required to display a “black box warning” for all users that would appear upon the first use of a platform and weekly thereafter.
The proposed language for the warning label is: “The Surgeon General has advised that there are ample indicators that social media can have a profound risk of harm to the mental health and well-being of children and adolescents.”
“This warning label isn’t a panacea, we know that, but it is another tool in our toolbox. It’s one prong in what has to be a multi-pronged continued, coordinated effort to address this public health crisis,” Bonta said.
Reached for comment, Bonta’s office said sponsorship of the bill was informed by their ongoing work to create a safer online space for children and teens and by the US Surgeon General’s call to Congress to add warning labels to social media.
In June, US Surgeon General Vivek Murthy, MD, said a Surgeon General’s warning label is needed to address the mental health emergency among adolescents and noted that evidence from tobacco studies shows warning labels can increase awareness and change behavior. In September, the attorneys general of 42 states announced their support of the proposal.
Also in September, US Senators John Fetterman (D-PA) and Katie Britt (R-AL) introduced the Stop the Scroll Act to create a mental health warning label requirement for social media platforms.
In a controversial move in November, Australia passed the world’s first law banning social media for children younger than 16 years. The law gives platforms such as TikTok, Facebook, X, Snapchat, and Instagram 1 year to figure out how to implement the ban before facing fines of up 50 million Australian dollars ($33 million) for systemic failures to prevent children younger than 16 years from holding accounts.
‘A Broken Fire Alarm’
“Slapping a warning label on social media is like a broken fire alarm going off with no evidence of smoke. It ignores the reality that most teens view social media as an important outlet for social connection,” Todd O’Boyle, with the tech industry policy group Chamber of Progress, said in a statement on AB 56.
He highlighted a 2022 Pew Research Center survey reporting that most teens credit social media with deepening connections and providing a support network and a 2020 study reporting that social media is not a strong or consistent risk factor for depressive symptoms in US adolescents.
O’Boyle predicted that, without strong evidence, AB 56 will run into the same “First Amendment buzzsaw” that has doomed other California kids’ bills.
Pediatrician Jason Nagata, MD, University of California San Francisco, points out in Bonta’s press release that social media can displace time for other healthful activities including sleep, exercise, and in-person socialization.
“While social media can provide educational content, it can also provide misinformation about health and expose children to content that damages their mental well-being. These are risks that adolescents and their parents should be aware of,” Nagata said.
Indeed, a tearful Victoria Hinks of Larkspur, California, spoke at the press conference of her 16-year-old daughter, Alexandra, who committed suicide 4 months ago after being sucked into social media and served content on self-harm, eating disorders, suicidal ideation, and glamorization of suicide.
“She was led down dark rabbit holes she had no hope of escaping,” Hinks said. “There is not a bone in my body that doubts social media played a role leading her to that final irreversible decision.”
Jim Steyer, CEO and founder of Common Sense Media, applauded California for being the first state to introduce social media warning label legislation. The group plans to lobby for similar proposals in other states he said at the press conference, noting that there are “tens of thousands of Alexandras out there.”
“We have seat belt laws, we have warning labels on cigarettes and alcohol, and that’s what we’re doing here,” Steyer said. “It’s a straightforward simple proposition, which is put your kids and teenagers first, put their self-interest first and hold the largest, most powerful, and wealthy companies in all of our lifetimes accountable for the harms that have happened on their platforms.”
A version of this article first appeared on Medscape.com.
In the latest effort to address the mental health crisis among adolescents, legislation in California would require social media platforms to come with a “black box” mental health warning label.
Despite growing evidence linking young people’s use of social media to significant health risks including depression, anxiety, and suicidal thoughts, social media companies have failed to be transparent about the risks, said Assembly member Rebecca Bauer-Kahan (D-Orinda), who introduced Assembly Bill (AB) 56.
“AB 56 ensures that families are armed with clear, actionable information to understand these dangers and make decisions that prioritizes their children’s well-being,” she said in a press release.
Bauer-Kahan noted that 95% of teens report using at least one social media platform and that more than one third say they use social media almost constantly.
“There is a powerful profit motive to keep our young people hooked online and engaged and it is exploiting the human psychology with notifications, likes, endless scrolling, and algorithmic amplification that is harming our children every day,” she said at a press conference on December 9 announcing the bill.
, a sponsor of AB 56, said in a press release.
Speaking at the press conference, Bonta said social media has many “incredible benefits” from giving people an outlet of expression to providing access to critical information but “there is no disputing the fact, it can have an enormously detrimental and dangerous impact on our young people. You cannot debate that. Our children are suffering.”
If AB 56 is successful, he said social media platforms would be required to display a “black box warning” for all users that would appear upon the first use of a platform and weekly thereafter.
The proposed language for the warning label is: “The Surgeon General has advised that there are ample indicators that social media can have a profound risk of harm to the mental health and well-being of children and adolescents.”
“This warning label isn’t a panacea, we know that, but it is another tool in our toolbox. It’s one prong in what has to be a multi-pronged continued, coordinated effort to address this public health crisis,” Bonta said.
Reached for comment, Bonta’s office said sponsorship of the bill was informed by their ongoing work to create a safer online space for children and teens and by the US Surgeon General’s call to Congress to add warning labels to social media.
In June, US Surgeon General Vivek Murthy, MD, said a Surgeon General’s warning label is needed to address the mental health emergency among adolescents and noted that evidence from tobacco studies shows warning labels can increase awareness and change behavior. In September, the attorneys general of 42 states announced their support of the proposal.
Also in September, US Senators John Fetterman (D-PA) and Katie Britt (R-AL) introduced the Stop the Scroll Act to create a mental health warning label requirement for social media platforms.
In a controversial move in November, Australia passed the world’s first law banning social media for children younger than 16 years. The law gives platforms such as TikTok, Facebook, X, Snapchat, and Instagram 1 year to figure out how to implement the ban before facing fines of up 50 million Australian dollars ($33 million) for systemic failures to prevent children younger than 16 years from holding accounts.
‘A Broken Fire Alarm’
“Slapping a warning label on social media is like a broken fire alarm going off with no evidence of smoke. It ignores the reality that most teens view social media as an important outlet for social connection,” Todd O’Boyle, with the tech industry policy group Chamber of Progress, said in a statement on AB 56.
He highlighted a 2022 Pew Research Center survey reporting that most teens credit social media with deepening connections and providing a support network and a 2020 study reporting that social media is not a strong or consistent risk factor for depressive symptoms in US adolescents.
O’Boyle predicted that, without strong evidence, AB 56 will run into the same “First Amendment buzzsaw” that has doomed other California kids’ bills.
Pediatrician Jason Nagata, MD, University of California San Francisco, points out in Bonta’s press release that social media can displace time for other healthful activities including sleep, exercise, and in-person socialization.
“While social media can provide educational content, it can also provide misinformation about health and expose children to content that damages their mental well-being. These are risks that adolescents and their parents should be aware of,” Nagata said.
Indeed, a tearful Victoria Hinks of Larkspur, California, spoke at the press conference of her 16-year-old daughter, Alexandra, who committed suicide 4 months ago after being sucked into social media and served content on self-harm, eating disorders, suicidal ideation, and glamorization of suicide.
“She was led down dark rabbit holes she had no hope of escaping,” Hinks said. “There is not a bone in my body that doubts social media played a role leading her to that final irreversible decision.”
Jim Steyer, CEO and founder of Common Sense Media, applauded California for being the first state to introduce social media warning label legislation. The group plans to lobby for similar proposals in other states he said at the press conference, noting that there are “tens of thousands of Alexandras out there.”
“We have seat belt laws, we have warning labels on cigarettes and alcohol, and that’s what we’re doing here,” Steyer said. “It’s a straightforward simple proposition, which is put your kids and teenagers first, put their self-interest first and hold the largest, most powerful, and wealthy companies in all of our lifetimes accountable for the harms that have happened on their platforms.”
A version of this article first appeared on Medscape.com.
In the latest effort to address the mental health crisis among adolescents, legislation in California would require social media platforms to come with a “black box” mental health warning label.
Despite growing evidence linking young people’s use of social media to significant health risks including depression, anxiety, and suicidal thoughts, social media companies have failed to be transparent about the risks, said Assembly member Rebecca Bauer-Kahan (D-Orinda), who introduced Assembly Bill (AB) 56.
“AB 56 ensures that families are armed with clear, actionable information to understand these dangers and make decisions that prioritizes their children’s well-being,” she said in a press release.
Bauer-Kahan noted that 95% of teens report using at least one social media platform and that more than one third say they use social media almost constantly.
“There is a powerful profit motive to keep our young people hooked online and engaged and it is exploiting the human psychology with notifications, likes, endless scrolling, and algorithmic amplification that is harming our children every day,” she said at a press conference on December 9 announcing the bill.
, a sponsor of AB 56, said in a press release.
Speaking at the press conference, Bonta said social media has many “incredible benefits” from giving people an outlet of expression to providing access to critical information but “there is no disputing the fact, it can have an enormously detrimental and dangerous impact on our young people. You cannot debate that. Our children are suffering.”
If AB 56 is successful, he said social media platforms would be required to display a “black box warning” for all users that would appear upon the first use of a platform and weekly thereafter.
The proposed language for the warning label is: “The Surgeon General has advised that there are ample indicators that social media can have a profound risk of harm to the mental health and well-being of children and adolescents.”
“This warning label isn’t a panacea, we know that, but it is another tool in our toolbox. It’s one prong in what has to be a multi-pronged continued, coordinated effort to address this public health crisis,” Bonta said.
Reached for comment, Bonta’s office said sponsorship of the bill was informed by their ongoing work to create a safer online space for children and teens and by the US Surgeon General’s call to Congress to add warning labels to social media.
In June, US Surgeon General Vivek Murthy, MD, said a Surgeon General’s warning label is needed to address the mental health emergency among adolescents and noted that evidence from tobacco studies shows warning labels can increase awareness and change behavior. In September, the attorneys general of 42 states announced their support of the proposal.
Also in September, US Senators John Fetterman (D-PA) and Katie Britt (R-AL) introduced the Stop the Scroll Act to create a mental health warning label requirement for social media platforms.
In a controversial move in November, Australia passed the world’s first law banning social media for children younger than 16 years. The law gives platforms such as TikTok, Facebook, X, Snapchat, and Instagram 1 year to figure out how to implement the ban before facing fines of up 50 million Australian dollars ($33 million) for systemic failures to prevent children younger than 16 years from holding accounts.
‘A Broken Fire Alarm’
“Slapping a warning label on social media is like a broken fire alarm going off with no evidence of smoke. It ignores the reality that most teens view social media as an important outlet for social connection,” Todd O’Boyle, with the tech industry policy group Chamber of Progress, said in a statement on AB 56.
He highlighted a 2022 Pew Research Center survey reporting that most teens credit social media with deepening connections and providing a support network and a 2020 study reporting that social media is not a strong or consistent risk factor for depressive symptoms in US adolescents.
O’Boyle predicted that, without strong evidence, AB 56 will run into the same “First Amendment buzzsaw” that has doomed other California kids’ bills.
Pediatrician Jason Nagata, MD, University of California San Francisco, points out in Bonta’s press release that social media can displace time for other healthful activities including sleep, exercise, and in-person socialization.
“While social media can provide educational content, it can also provide misinformation about health and expose children to content that damages their mental well-being. These are risks that adolescents and their parents should be aware of,” Nagata said.
Indeed, a tearful Victoria Hinks of Larkspur, California, spoke at the press conference of her 16-year-old daughter, Alexandra, who committed suicide 4 months ago after being sucked into social media and served content on self-harm, eating disorders, suicidal ideation, and glamorization of suicide.
“She was led down dark rabbit holes she had no hope of escaping,” Hinks said. “There is not a bone in my body that doubts social media played a role leading her to that final irreversible decision.”
Jim Steyer, CEO and founder of Common Sense Media, applauded California for being the first state to introduce social media warning label legislation. The group plans to lobby for similar proposals in other states he said at the press conference, noting that there are “tens of thousands of Alexandras out there.”
“We have seat belt laws, we have warning labels on cigarettes and alcohol, and that’s what we’re doing here,” Steyer said. “It’s a straightforward simple proposition, which is put your kids and teenagers first, put their self-interest first and hold the largest, most powerful, and wealthy companies in all of our lifetimes accountable for the harms that have happened on their platforms.”
A version of this article first appeared on Medscape.com.
Managing Return-to-Work Barriers for People With Long COVID
Although some patients experience symptoms so severe that they cannot work under any conditions, medical providers and employers can help ensure many patients with long COVID can stay in the workforce.
Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems. By the end of 2023, at least 400 million people worldwide were estimated to have long COVID.
As members of the Patient-Led Research Collaborative, an international group of more than 60 researchers and health advocates living with long COVID and other infection-associated chronic conditions, we have published one of the first research studies of people with long COVID and their desire to work, the specific needs they have, and what doctors and employers can do to create a path for returning to the workforce.
In our recent paper, we document the barriers and facilitators that individuals living with long COVID experience when attempting to return to work. Our recommendations are based on these findings and include recommendations for both medical providers and employers.
If you are a medical provider:
- Ensure you adequately document your patients’ COVID cases, any long COVID diagnoses, and the functional impairment that long COVID causes. Remember that you can diagnose a patient with long COVID on the basis of their symptoms, and clinical guidelines do not require a record of a positive COVID-19 test, which many patients lack owing to testing barriers.
- Keep up to date on research on long COVID and related infection-associated chronic conditions — for example, through the Project ECHO Long COVID and Fatiguing Illness Recovery Program — and learn about pacing and other treatment options.
If you are an employer:
- Utilize a return-to-work model in which any worker with suspected or confirmed COVID discusses support they may need with their employer when they return to work, with additional check-in dates scheduled to reevaluate supports as needed. Planning for this collaborative and iterative evaluation of return-to-work supports for all workers with COVID-19 is important because it may not be immediately clear to a worker whether they have developed long COVID or are generally recuperating from the illness.
- Do not require medical documentation of a SARS-CoV-2 infection or a Long COVID diagnosis to access accommodations — this is owing to disparities in accessing documentation.
- Tailor job responsibilities, provide remote options, allow flexible hours, and provide longer-range deadlines to account for symptoms for people with long COVID and other infection-associated chronic conditions.
- Provide accommodations to any caregivers of people with long COVID in your workplace.
- If requiring in-person work, make the workplace as safe as possible through ventilation and masking requirements, which will help ensure fewer of your workers develop long COVID, and those already with infection-associated chronic conditions will not get worse.
Our findings and recommendations are specific to long COVID, but they can and should apply to other disabilities. Given that our study’s sample was predominately White and working in jobs that did not require substantial physical labor, additional recommendations may be needed for other populations and workers who have labor-intensive jobs.
510 Study Participants
Long COVID is characterized as a relapsing-remitting illness, often described as episodic, in which an individual’s symptoms may fluctuate. Symptoms can become more or less severe depending on tasks, exertion, and social support in addition to physiologic processes and medical intervention. In our paper, we illustrate how the long COVID return-to-work experience and individuals’ symptoms can be shaped by workplace, home, and medical environments.
We randomly selected 510 participants from a global survey of people living with long COVID and systematically analyzed their open-ended responses using established qualitative analysis methods. In this study, we specifically analyzed what patients wrote about their return-to-work experiences, considering how work experiences and relapsing and remitting long COVID symptoms intersected with personal lives and medical care.
Most of the study participants identified as White, were 30-60 years old (ie, in their key earning years), and had at least a baccalaureate degree. Participants lived in the United States (38%), United Kingdom (25%), continental Europe (8%), Canada (4%), or other countries (25%). Most participants worked in professions that did not require substantial physical labor, and individuals in those fields may experience even greater return-to-work barriers than are reported in this study.
Key Findings
Through our qualitative analysis, we identified four primary return-to-work themes:
1. People living with long COVID have a strong desire and financial need to return to work.
The participants in our study described how they had experienced financial hardship because they could not successfully return to work and may have incurred new expenses with long COVID. They also often wrote how they wanted to return to work because their jobs provided meaning and structure for their lives. Some people in this study shared how they had tried to return to their jobs but relapsed. As a result, they considered leaving the workforce.
2. Workers’ long COVID symptoms intersect with organization of work and home life.
Most of the people in our study were employed in positions that did not require substantial physical labor. Even so, workers described how their long COVID symptoms were exacerbated by some job tasks. Computer screen time; reading dense material or writing (including emails); and conversations and meetings, regardless of whether they were in-person or via phone or video conferencing, could trigger or make symptoms worse. Workers who needed to stand for long periods of time, such as teachers and healthcare workers, and workers who needed to do lifting as part of their jobs described how these requirements were too taxing and could lead to relapses.
Because of the relapsing and remitting nature of many long COVID symptoms, people reported how it could be difficult to predict how job tasks, long hours, or pressing deadlines may exacerbate symptoms, which would require them to take time off work. For these participants, “pushing through” symptoms only made the symptoms worse. However, people in the study who were allowed to work from home reported how pacing, elevating their legs, and conserving energy (especially by not commuting) was key to doing their jobs well.
Some people in the study described how they were only able to return to work because they had substantial support from family or partners at home. These individuals shared how the people they lived with did most of the cooking, cleaning, and other household tasks so that the person living with long COVID could conserve their energy for work. This reorganization of home life notably shifted household tasks and caregiving to other people in the household, but without this shift, the individual’s long COVID symptoms may be too severe to work.
3. People with long COVID experience disbelief and stigma at work and healthcare settings.
Some people in our study described how their colleagues, supervisors, and human resource managers insinuated that they were fabricating or exaggerating their symptoms. This made it hard for workers to communicate what support they needed and could limit access to necessary work accommodations.
Many people in our study also described how medical providers did not believe that they had long COVID despite experiencing debilitating symptoms, often because they did not have a positive COVID-19 test to prove they had had an acute infection. Many people with long COVID may not have a positive COVID-19 test because:
- They could not access a test because testing access was limited at the start of the COVID-19 pandemic, there are transportation and cost barriers to tests, many health insurance providers no longer cover tests; and there are fewer public testing sites since the World Health Organization declared an end to the public health emergency;
- There is a high probability of false-negative results for viral and antibody tests (especially during the first wave of the pandemic and for individuals with limited immune response); and
- People can develop long COVID after asymptomatic acute infection.
Although healthcare providers can provide a long COVID diagnosis without a positive COVID-19 test on the basis of a patient’s presentation of symptoms and clinical history, many people in our study said that their providers would not provide this diagnosis, which restricted access to worker’s compensation, paid time off, and job accommodations.
Many people in the study also reported that their medical providers misdiagnosed them with a mental health disorder, such as anxiety, instead of long COVID. Although some people with long COVID may experience poor mental health as a natural consequence of dealing with a debilitating medical condition or may have neuropsychiatric symptoms as part of their long COVID, long COVID is not caused by an underlying psychiatric illness.
4. Support of medical providers is key to successful return to work for people living with long COVID.
Some people in our study described how they were able to get workplace accommodations or access workers’ compensation or sick leave because their medical providers recognized they had long COVID and provided them with this documentation. Some of these participants did not have a positive COVID-19 test, but their medical providers were able to diagnose them with long COVID on the basis of symptom presentation and clinical history. This documentation was critical for helping workers remain financially stable and able to return to work.
Conclusion
While we continue to search for treatment and cures for long COVID and work to provide a robust social safety net, it is crucial to address the stigma, inaccessibility, and lack of support often experienced by patients in their workplaces. Disabled people have long faced these issues; long COVID may be an opportunity to revolutionize the workplace to ensure an inclusive and accessible environment that can improve the lives of all workers.
For more on how to best be inclusive of employees with long COVID, read Harvard Business Review’s “Long Covid at Work: A Manager’s Guide” and visit the Job Accommodation Network webpage dedicated to long COVID.
Additional discussion about our study and applying the findings to improve work and medical care can be found by listening to the Healthy Work podcast episode titled “Supporting Long COVID at Work.”
Elisabeth Stelson, Gina Assaf, and Lisa McCorkell are members of the Patient-Led Research Collaborative, an international group of more than 60 researchers. Dr Stelson, Postdoctoral Research Fellow, Department of Epidemiology, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, has disclosed no relevant financial relationships. Gina Assaf is Research Lead, Patient-Led Research Collaborative, Washington, DC. Lisa McCorkell is a long COVID patient; Cofounder, Team Lead, Researcher, Patient-Led Research Collaborative, Washington, DC.
A version of this article appeared on Medscape.com.
Although some patients experience symptoms so severe that they cannot work under any conditions, medical providers and employers can help ensure many patients with long COVID can stay in the workforce.
Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems. By the end of 2023, at least 400 million people worldwide were estimated to have long COVID.
As members of the Patient-Led Research Collaborative, an international group of more than 60 researchers and health advocates living with long COVID and other infection-associated chronic conditions, we have published one of the first research studies of people with long COVID and their desire to work, the specific needs they have, and what doctors and employers can do to create a path for returning to the workforce.
In our recent paper, we document the barriers and facilitators that individuals living with long COVID experience when attempting to return to work. Our recommendations are based on these findings and include recommendations for both medical providers and employers.
If you are a medical provider:
- Ensure you adequately document your patients’ COVID cases, any long COVID diagnoses, and the functional impairment that long COVID causes. Remember that you can diagnose a patient with long COVID on the basis of their symptoms, and clinical guidelines do not require a record of a positive COVID-19 test, which many patients lack owing to testing barriers.
- Keep up to date on research on long COVID and related infection-associated chronic conditions — for example, through the Project ECHO Long COVID and Fatiguing Illness Recovery Program — and learn about pacing and other treatment options.
If you are an employer:
- Utilize a return-to-work model in which any worker with suspected or confirmed COVID discusses support they may need with their employer when they return to work, with additional check-in dates scheduled to reevaluate supports as needed. Planning for this collaborative and iterative evaluation of return-to-work supports for all workers with COVID-19 is important because it may not be immediately clear to a worker whether they have developed long COVID or are generally recuperating from the illness.
- Do not require medical documentation of a SARS-CoV-2 infection or a Long COVID diagnosis to access accommodations — this is owing to disparities in accessing documentation.
- Tailor job responsibilities, provide remote options, allow flexible hours, and provide longer-range deadlines to account for symptoms for people with long COVID and other infection-associated chronic conditions.
- Provide accommodations to any caregivers of people with long COVID in your workplace.
- If requiring in-person work, make the workplace as safe as possible through ventilation and masking requirements, which will help ensure fewer of your workers develop long COVID, and those already with infection-associated chronic conditions will not get worse.
Our findings and recommendations are specific to long COVID, but they can and should apply to other disabilities. Given that our study’s sample was predominately White and working in jobs that did not require substantial physical labor, additional recommendations may be needed for other populations and workers who have labor-intensive jobs.
510 Study Participants
Long COVID is characterized as a relapsing-remitting illness, often described as episodic, in which an individual’s symptoms may fluctuate. Symptoms can become more or less severe depending on tasks, exertion, and social support in addition to physiologic processes and medical intervention. In our paper, we illustrate how the long COVID return-to-work experience and individuals’ symptoms can be shaped by workplace, home, and medical environments.
We randomly selected 510 participants from a global survey of people living with long COVID and systematically analyzed their open-ended responses using established qualitative analysis methods. In this study, we specifically analyzed what patients wrote about their return-to-work experiences, considering how work experiences and relapsing and remitting long COVID symptoms intersected with personal lives and medical care.
Most of the study participants identified as White, were 30-60 years old (ie, in their key earning years), and had at least a baccalaureate degree. Participants lived in the United States (38%), United Kingdom (25%), continental Europe (8%), Canada (4%), or other countries (25%). Most participants worked in professions that did not require substantial physical labor, and individuals in those fields may experience even greater return-to-work barriers than are reported in this study.
Key Findings
Through our qualitative analysis, we identified four primary return-to-work themes:
1. People living with long COVID have a strong desire and financial need to return to work.
The participants in our study described how they had experienced financial hardship because they could not successfully return to work and may have incurred new expenses with long COVID. They also often wrote how they wanted to return to work because their jobs provided meaning and structure for their lives. Some people in this study shared how they had tried to return to their jobs but relapsed. As a result, they considered leaving the workforce.
2. Workers’ long COVID symptoms intersect with organization of work and home life.
Most of the people in our study were employed in positions that did not require substantial physical labor. Even so, workers described how their long COVID symptoms were exacerbated by some job tasks. Computer screen time; reading dense material or writing (including emails); and conversations and meetings, regardless of whether they were in-person or via phone or video conferencing, could trigger or make symptoms worse. Workers who needed to stand for long periods of time, such as teachers and healthcare workers, and workers who needed to do lifting as part of their jobs described how these requirements were too taxing and could lead to relapses.
Because of the relapsing and remitting nature of many long COVID symptoms, people reported how it could be difficult to predict how job tasks, long hours, or pressing deadlines may exacerbate symptoms, which would require them to take time off work. For these participants, “pushing through” symptoms only made the symptoms worse. However, people in the study who were allowed to work from home reported how pacing, elevating their legs, and conserving energy (especially by not commuting) was key to doing their jobs well.
Some people in the study described how they were only able to return to work because they had substantial support from family or partners at home. These individuals shared how the people they lived with did most of the cooking, cleaning, and other household tasks so that the person living with long COVID could conserve their energy for work. This reorganization of home life notably shifted household tasks and caregiving to other people in the household, but without this shift, the individual’s long COVID symptoms may be too severe to work.
3. People with long COVID experience disbelief and stigma at work and healthcare settings.
Some people in our study described how their colleagues, supervisors, and human resource managers insinuated that they were fabricating or exaggerating their symptoms. This made it hard for workers to communicate what support they needed and could limit access to necessary work accommodations.
Many people in our study also described how medical providers did not believe that they had long COVID despite experiencing debilitating symptoms, often because they did not have a positive COVID-19 test to prove they had had an acute infection. Many people with long COVID may not have a positive COVID-19 test because:
- They could not access a test because testing access was limited at the start of the COVID-19 pandemic, there are transportation and cost barriers to tests, many health insurance providers no longer cover tests; and there are fewer public testing sites since the World Health Organization declared an end to the public health emergency;
- There is a high probability of false-negative results for viral and antibody tests (especially during the first wave of the pandemic and for individuals with limited immune response); and
- People can develop long COVID after asymptomatic acute infection.
Although healthcare providers can provide a long COVID diagnosis without a positive COVID-19 test on the basis of a patient’s presentation of symptoms and clinical history, many people in our study said that their providers would not provide this diagnosis, which restricted access to worker’s compensation, paid time off, and job accommodations.
Many people in the study also reported that their medical providers misdiagnosed them with a mental health disorder, such as anxiety, instead of long COVID. Although some people with long COVID may experience poor mental health as a natural consequence of dealing with a debilitating medical condition or may have neuropsychiatric symptoms as part of their long COVID, long COVID is not caused by an underlying psychiatric illness.
4. Support of medical providers is key to successful return to work for people living with long COVID.
Some people in our study described how they were able to get workplace accommodations or access workers’ compensation or sick leave because their medical providers recognized they had long COVID and provided them with this documentation. Some of these participants did not have a positive COVID-19 test, but their medical providers were able to diagnose them with long COVID on the basis of symptom presentation and clinical history. This documentation was critical for helping workers remain financially stable and able to return to work.
Conclusion
While we continue to search for treatment and cures for long COVID and work to provide a robust social safety net, it is crucial to address the stigma, inaccessibility, and lack of support often experienced by patients in their workplaces. Disabled people have long faced these issues; long COVID may be an opportunity to revolutionize the workplace to ensure an inclusive and accessible environment that can improve the lives of all workers.
For more on how to best be inclusive of employees with long COVID, read Harvard Business Review’s “Long Covid at Work: A Manager’s Guide” and visit the Job Accommodation Network webpage dedicated to long COVID.
Additional discussion about our study and applying the findings to improve work and medical care can be found by listening to the Healthy Work podcast episode titled “Supporting Long COVID at Work.”
Elisabeth Stelson, Gina Assaf, and Lisa McCorkell are members of the Patient-Led Research Collaborative, an international group of more than 60 researchers. Dr Stelson, Postdoctoral Research Fellow, Department of Epidemiology, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, has disclosed no relevant financial relationships. Gina Assaf is Research Lead, Patient-Led Research Collaborative, Washington, DC. Lisa McCorkell is a long COVID patient; Cofounder, Team Lead, Researcher, Patient-Led Research Collaborative, Washington, DC.
A version of this article appeared on Medscape.com.
Although some patients experience symptoms so severe that they cannot work under any conditions, medical providers and employers can help ensure many patients with long COVID can stay in the workforce.
Long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems. By the end of 2023, at least 400 million people worldwide were estimated to have long COVID.
As members of the Patient-Led Research Collaborative, an international group of more than 60 researchers and health advocates living with long COVID and other infection-associated chronic conditions, we have published one of the first research studies of people with long COVID and their desire to work, the specific needs they have, and what doctors and employers can do to create a path for returning to the workforce.
In our recent paper, we document the barriers and facilitators that individuals living with long COVID experience when attempting to return to work. Our recommendations are based on these findings and include recommendations for both medical providers and employers.
If you are a medical provider:
- Ensure you adequately document your patients’ COVID cases, any long COVID diagnoses, and the functional impairment that long COVID causes. Remember that you can diagnose a patient with long COVID on the basis of their symptoms, and clinical guidelines do not require a record of a positive COVID-19 test, which many patients lack owing to testing barriers.
- Keep up to date on research on long COVID and related infection-associated chronic conditions — for example, through the Project ECHO Long COVID and Fatiguing Illness Recovery Program — and learn about pacing and other treatment options.
If you are an employer:
- Utilize a return-to-work model in which any worker with suspected or confirmed COVID discusses support they may need with their employer when they return to work, with additional check-in dates scheduled to reevaluate supports as needed. Planning for this collaborative and iterative evaluation of return-to-work supports for all workers with COVID-19 is important because it may not be immediately clear to a worker whether they have developed long COVID or are generally recuperating from the illness.
- Do not require medical documentation of a SARS-CoV-2 infection or a Long COVID diagnosis to access accommodations — this is owing to disparities in accessing documentation.
- Tailor job responsibilities, provide remote options, allow flexible hours, and provide longer-range deadlines to account for symptoms for people with long COVID and other infection-associated chronic conditions.
- Provide accommodations to any caregivers of people with long COVID in your workplace.
- If requiring in-person work, make the workplace as safe as possible through ventilation and masking requirements, which will help ensure fewer of your workers develop long COVID, and those already with infection-associated chronic conditions will not get worse.
Our findings and recommendations are specific to long COVID, but they can and should apply to other disabilities. Given that our study’s sample was predominately White and working in jobs that did not require substantial physical labor, additional recommendations may be needed for other populations and workers who have labor-intensive jobs.
510 Study Participants
Long COVID is characterized as a relapsing-remitting illness, often described as episodic, in which an individual’s symptoms may fluctuate. Symptoms can become more or less severe depending on tasks, exertion, and social support in addition to physiologic processes and medical intervention. In our paper, we illustrate how the long COVID return-to-work experience and individuals’ symptoms can be shaped by workplace, home, and medical environments.
We randomly selected 510 participants from a global survey of people living with long COVID and systematically analyzed their open-ended responses using established qualitative analysis methods. In this study, we specifically analyzed what patients wrote about their return-to-work experiences, considering how work experiences and relapsing and remitting long COVID symptoms intersected with personal lives and medical care.
Most of the study participants identified as White, were 30-60 years old (ie, in their key earning years), and had at least a baccalaureate degree. Participants lived in the United States (38%), United Kingdom (25%), continental Europe (8%), Canada (4%), or other countries (25%). Most participants worked in professions that did not require substantial physical labor, and individuals in those fields may experience even greater return-to-work barriers than are reported in this study.
Key Findings
Through our qualitative analysis, we identified four primary return-to-work themes:
1. People living with long COVID have a strong desire and financial need to return to work.
The participants in our study described how they had experienced financial hardship because they could not successfully return to work and may have incurred new expenses with long COVID. They also often wrote how they wanted to return to work because their jobs provided meaning and structure for their lives. Some people in this study shared how they had tried to return to their jobs but relapsed. As a result, they considered leaving the workforce.
2. Workers’ long COVID symptoms intersect with organization of work and home life.
Most of the people in our study were employed in positions that did not require substantial physical labor. Even so, workers described how their long COVID symptoms were exacerbated by some job tasks. Computer screen time; reading dense material or writing (including emails); and conversations and meetings, regardless of whether they were in-person or via phone or video conferencing, could trigger or make symptoms worse. Workers who needed to stand for long periods of time, such as teachers and healthcare workers, and workers who needed to do lifting as part of their jobs described how these requirements were too taxing and could lead to relapses.
Because of the relapsing and remitting nature of many long COVID symptoms, people reported how it could be difficult to predict how job tasks, long hours, or pressing deadlines may exacerbate symptoms, which would require them to take time off work. For these participants, “pushing through” symptoms only made the symptoms worse. However, people in the study who were allowed to work from home reported how pacing, elevating their legs, and conserving energy (especially by not commuting) was key to doing their jobs well.
Some people in the study described how they were only able to return to work because they had substantial support from family or partners at home. These individuals shared how the people they lived with did most of the cooking, cleaning, and other household tasks so that the person living with long COVID could conserve their energy for work. This reorganization of home life notably shifted household tasks and caregiving to other people in the household, but without this shift, the individual’s long COVID symptoms may be too severe to work.
3. People with long COVID experience disbelief and stigma at work and healthcare settings.
Some people in our study described how their colleagues, supervisors, and human resource managers insinuated that they were fabricating or exaggerating their symptoms. This made it hard for workers to communicate what support they needed and could limit access to necessary work accommodations.
Many people in our study also described how medical providers did not believe that they had long COVID despite experiencing debilitating symptoms, often because they did not have a positive COVID-19 test to prove they had had an acute infection. Many people with long COVID may not have a positive COVID-19 test because:
- They could not access a test because testing access was limited at the start of the COVID-19 pandemic, there are transportation and cost barriers to tests, many health insurance providers no longer cover tests; and there are fewer public testing sites since the World Health Organization declared an end to the public health emergency;
- There is a high probability of false-negative results for viral and antibody tests (especially during the first wave of the pandemic and for individuals with limited immune response); and
- People can develop long COVID after asymptomatic acute infection.
Although healthcare providers can provide a long COVID diagnosis without a positive COVID-19 test on the basis of a patient’s presentation of symptoms and clinical history, many people in our study said that their providers would not provide this diagnosis, which restricted access to worker’s compensation, paid time off, and job accommodations.
Many people in the study also reported that their medical providers misdiagnosed them with a mental health disorder, such as anxiety, instead of long COVID. Although some people with long COVID may experience poor mental health as a natural consequence of dealing with a debilitating medical condition or may have neuropsychiatric symptoms as part of their long COVID, long COVID is not caused by an underlying psychiatric illness.
4. Support of medical providers is key to successful return to work for people living with long COVID.
Some people in our study described how they were able to get workplace accommodations or access workers’ compensation or sick leave because their medical providers recognized they had long COVID and provided them with this documentation. Some of these participants did not have a positive COVID-19 test, but their medical providers were able to diagnose them with long COVID on the basis of symptom presentation and clinical history. This documentation was critical for helping workers remain financially stable and able to return to work.
Conclusion
While we continue to search for treatment and cures for long COVID and work to provide a robust social safety net, it is crucial to address the stigma, inaccessibility, and lack of support often experienced by patients in their workplaces. Disabled people have long faced these issues; long COVID may be an opportunity to revolutionize the workplace to ensure an inclusive and accessible environment that can improve the lives of all workers.
For more on how to best be inclusive of employees with long COVID, read Harvard Business Review’s “Long Covid at Work: A Manager’s Guide” and visit the Job Accommodation Network webpage dedicated to long COVID.
Additional discussion about our study and applying the findings to improve work and medical care can be found by listening to the Healthy Work podcast episode titled “Supporting Long COVID at Work.”
Elisabeth Stelson, Gina Assaf, and Lisa McCorkell are members of the Patient-Led Research Collaborative, an international group of more than 60 researchers. Dr Stelson, Postdoctoral Research Fellow, Department of Epidemiology, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, has disclosed no relevant financial relationships. Gina Assaf is Research Lead, Patient-Led Research Collaborative, Washington, DC. Lisa McCorkell is a long COVID patient; Cofounder, Team Lead, Researcher, Patient-Led Research Collaborative, Washington, DC.
A version of this article appeared on Medscape.com.
Psychologists and Psychotropic Prescribing: An Old Debate Heats Up
Earlier in 2024, Utah became the seventh state to allow psychologists with the proper training to prescribe psychotropic medications, giving supporters reason to hope that more states might support expanding this scope of practice.
However, the American Psychiatric Association — and some psychologists — oppose granting psychologists this privilege, arguing that the training offered is insufficient and could jeopardize patient safety.
The controversy over whether psychologists should be allowed to prescribe is as old as the so-called RxP movement itself, which began in the early 1990s.
Psychologists have not rushed to become licensed prescribers. After three decades, an estimated 226 psychologists — representing just 0.14% of all those licensed in the United States — have been authorized to prescribe in the six states and one territory where it has been legalized, according to a just-published study in Clinical Psychology.
These are Colorado, Idaho, Illinois, Iowa, Louisiana, New Mexico, and Guam. Data from the study show that only 73 psychologists are prescribing in New Mexico, which authorized it in 2002.
Less is known about the number of psychologists who are prescribing under allowances in the Department of Defense, Indian Health Service and US Public Health Service.
Some psychologists — and the American Psychological Association (APA) — believe that the persistence of the opioid epidemic coupled with a continued lack of access to mental health care for millions of Americans will bring more legislators on-side.
“I feel like we’re on an upswing again,” Deborah Baker, director of legal and regulatory policy for the APA, told Medscape Medical News. “The access issue continues to be a perennial kind of driver.” She noted that at least six states pursued expanding privileges this year.
Robert L. Trestman, MD, chair of the American Psychiatric Association’s Council on Healthcare Systems and Financing, said he doesn’t see new momentum. The interest in having psychologists prescribe “continues to trickle based on just the frustration that people have about not getting adequate access to psychiatry,” he told Medscape Medical News.
While states may be trying to increase access to care, granting psychologists privileges is “not a very effective way of doing it,” said Trestman, Chair of Psychiatry and Behavioral Medicine at Virginia Tech Carilion School of Medicine in Roanoke. Psychologists are needed to deliver psychotherapy, he said. “It makes almost no sense to try to make them into pseudo medical professionals,” said Trestman. “It just exposes people to risks.”
William Robiner, PhD — author of Clinical Psychology study — is a long-time opponent of RxP. The psychologist told Medscape Medical News he’s concerned about patient safety and “about some of the disingenuous reasons” that psychologists want to prescribe. Among these are the ability to increase status and income, said Robiner, a professor of medicine at the University of Minnesota Medical School, Minneapolis, and a board member of Psychologists Opposed to Prescription Privileges for Psychologists.
Adequate Training?
Only PhD and PsyD psychologists are eligible for RxP training, which entails a master’s in clinical psychopharmacology. After receiving the master’s, they must pass the Psychopharmacology Examination for Psychologists and then are only permitted to prescribe medications for mental health disorders.
They must also obtain a Drug Enforcement Administration license but can’t write prescriptions for schedule 2 medications. In some states, psychologists can prescribe buprenorphine and other opioid use disorder medications.
The APA has developed guidance for master’s programs, which currently number just over a handful in the United States.
At Fairleigh Dickinson University in New Jersey, students enrolled in the master’s program — a distance-learning format — complete 10 courses over five 15-week semesters. The curriculum spans a range of topics, from foundational sciences and legal and ethical considerations to strategies for treating specific disorders.
Derek Phillips, PhD, the program’s executive director, said that when he took the position in 2020, enrollment was capped at 45 students, but “we were not routinely enrolling the maximum.” Now, even with class size increased to 60 “we are consistently full and have a waitlist,” he told Medscape Medical News.
Interest is being driven in part by new laws in Colorado (2023) and Utah, said Phillips. But many are enrolling without intending to write a prescription, he said. The degree gives graduates the ability to better collaborate with other clinicians, teach clinical psychopharmacology, and be expert witnesses in medico-legal cases, he said.
In addition, the training gives students “a balanced and thorough biopsychosocial understanding of our patients,” he said. Students also see the “potential of being able to be a ‘one-stop-stop’ of mental health services,” said Phillips.
The American Board of Professional Psychology is developing a board certification in clinical psychopharmacology.
The APA states on its website that prescribing psychologists have “more training in diagnosing and treating (including prescribing) mental health disorders than primary care physicians.”
However, critics argue that the training falls short. Most psychologists, said Robiner, have not completed the undergraduate prerequisites — such as anatomy, physiology, and chemistry — that are required for other prescribing professionals.
In a 2019 article comparing the training of prescribing professionals, Robiner and colleagues reported that psychiatrists undergo 4- to 6-week rotations during medical school and accumulate 8000 clinical hours focused on psychiatric conditions over the course of their 4-year residencies.
States set requirements for clinical hours for prescribing psychologists, but they are generally elective and completed after individuals receive a master’s degree.
Robiner said psychologists aren’t trained in evaluating drug-related adverse events. “If you show a psychologist a rash, they have no idea whether that rash might be a medication adverse effect or poison ivy,” he said.
Trestman pointed out that many psychotropic medications have black box warnings. “The risk of toxicity is by no means trivial, and the majority of people who are seeking care in psychiatry have multiple comorbidities,” he said. “Giving people the equivalent of more or less 10 weeks of training is just woefully inappropriate,” Trestman said.
Increase in Access?
Psychology’s main argument for expanding its scope of practice is that it will increase the number of clinicians available to provide behavioral and mental health care.
Critics said that is a failed experiment, in part because so few psychologists have become prescribers, but also because most psychologists practice in the same areas as psychiatrists. Both specialists tend to cluster in urban regions, which already have high clinician density, said Trestman.
Psychologists are not practicing in underserved rural areas, as even APA data show. A 2018 APA snapshot of the workforce found that the highest density of psychologists was in Washington, DC, Massachusetts, and New York. South Carolina, West Virginia and Mississippi had the fewest number of psychologists per 100,000 people.
The University of Washington Rural Health Research Center reported in 2022 that in 2021, almost half of rural counties did not have a psychologist compared with 15.7% of urban counties.
Psychiatrists also are concentrated on the coasts and New England, according to a study by Ohio State researchers. The highest densities were in Washington, DC (79 per 100,000), Massachusetts (45.3), Rhode Island (42.6), Connecticut (38.6) and Vermont (37.7), whereas the lowest densities were in Idaho (11.8), Mississippi (11.8), Wyoming (12.4), Alabama (13.1), and Indiana (13.5). The study estimated that there were 57,163 psychiatrists responsible for the care of 333,287,557 Americans. “Clinical psychologists, psychotherapists, and counselors can provide alternative forms of intervention, though access to such services is also poor in rural areas,” wrote the authors.
The APA counters with data it says shows that RxP may have increased access. Using the number of psychology practices as a proxy for supply, the authors reported that practices grew in New Mexico, Illinois, Iowa, and Idaho — states that have implemented prescription privileges. Overall, there was an increase of 0.8047 practices per 100,000 residents per county.
However, the access argument “is seriously challenged by the reality of the limited number of psychologists who complete the pathway to prescribing,” Robiner and his colleague Tanya Tompkins countered in Clinical Psychology. They note that in Idaho — a state with shortages of psychologists and psychiatrists — just 10 of the state’s 615 psychologists had prescriptive authority. An estimated 5131 nonpsychologists are prescribers.
Robiner and Tompkins noted that it’s not clear why so few psychologists are pursuing RxP but that many seem to be unaware of the possibility.
Do Benefits Outweigh the Harms?
There is not a large body of literature assessing the harms or benefits of prescribing privileges for psychologists.
Baker shared several studies by Phillip Hughes, PhD, an outcomes researcher at the University of North Carolina Eshelman School of Pharmacy, Chapel Hill, North Carolina. In one study, Hughes found that patients of prescribing psychologists had a 24% lower rate of adverse drug events than patients of psychiatrists. Psychologists’ patients had lower rates of psychotropic polypharmacy but similar rates of emergency room use.
In another paper Hughes suggested that deaths attributable to mental illness had declined in New Mexico after it passed its law. There was no change in Louisiana.
With little evidence of harm — and ongoing provider shortages — making use of nonphysician prescribers is gaining traction with policymakers, claims the psychology association’s Baker, adding that in Utah, the Republican governor was the biggest supporter.
But psychiatrists argue that it’s more important to increase their numbers. Congress agreed in 2021 and 2023 to add 1200 new residency slots — in every specialty — to ease physician shortages. The Centers for Medicare & Medicaid Services recently announced that 70% of the new slots for July 2025 will go to primary care and psychiatry.
“Once those positions are in place, it will be four more years before the first crop of new psychiatrists come out,” noted Trestman. “None of these fixes are quick,” he said.
Baker, Robiner, and Trestman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Earlier in 2024, Utah became the seventh state to allow psychologists with the proper training to prescribe psychotropic medications, giving supporters reason to hope that more states might support expanding this scope of practice.
However, the American Psychiatric Association — and some psychologists — oppose granting psychologists this privilege, arguing that the training offered is insufficient and could jeopardize patient safety.
The controversy over whether psychologists should be allowed to prescribe is as old as the so-called RxP movement itself, which began in the early 1990s.
Psychologists have not rushed to become licensed prescribers. After three decades, an estimated 226 psychologists — representing just 0.14% of all those licensed in the United States — have been authorized to prescribe in the six states and one territory where it has been legalized, according to a just-published study in Clinical Psychology.
These are Colorado, Idaho, Illinois, Iowa, Louisiana, New Mexico, and Guam. Data from the study show that only 73 psychologists are prescribing in New Mexico, which authorized it in 2002.
Less is known about the number of psychologists who are prescribing under allowances in the Department of Defense, Indian Health Service and US Public Health Service.
Some psychologists — and the American Psychological Association (APA) — believe that the persistence of the opioid epidemic coupled with a continued lack of access to mental health care for millions of Americans will bring more legislators on-side.
“I feel like we’re on an upswing again,” Deborah Baker, director of legal and regulatory policy for the APA, told Medscape Medical News. “The access issue continues to be a perennial kind of driver.” She noted that at least six states pursued expanding privileges this year.
Robert L. Trestman, MD, chair of the American Psychiatric Association’s Council on Healthcare Systems and Financing, said he doesn’t see new momentum. The interest in having psychologists prescribe “continues to trickle based on just the frustration that people have about not getting adequate access to psychiatry,” he told Medscape Medical News.
While states may be trying to increase access to care, granting psychologists privileges is “not a very effective way of doing it,” said Trestman, Chair of Psychiatry and Behavioral Medicine at Virginia Tech Carilion School of Medicine in Roanoke. Psychologists are needed to deliver psychotherapy, he said. “It makes almost no sense to try to make them into pseudo medical professionals,” said Trestman. “It just exposes people to risks.”
William Robiner, PhD — author of Clinical Psychology study — is a long-time opponent of RxP. The psychologist told Medscape Medical News he’s concerned about patient safety and “about some of the disingenuous reasons” that psychologists want to prescribe. Among these are the ability to increase status and income, said Robiner, a professor of medicine at the University of Minnesota Medical School, Minneapolis, and a board member of Psychologists Opposed to Prescription Privileges for Psychologists.
Adequate Training?
Only PhD and PsyD psychologists are eligible for RxP training, which entails a master’s in clinical psychopharmacology. After receiving the master’s, they must pass the Psychopharmacology Examination for Psychologists and then are only permitted to prescribe medications for mental health disorders.
They must also obtain a Drug Enforcement Administration license but can’t write prescriptions for schedule 2 medications. In some states, psychologists can prescribe buprenorphine and other opioid use disorder medications.
The APA has developed guidance for master’s programs, which currently number just over a handful in the United States.
At Fairleigh Dickinson University in New Jersey, students enrolled in the master’s program — a distance-learning format — complete 10 courses over five 15-week semesters. The curriculum spans a range of topics, from foundational sciences and legal and ethical considerations to strategies for treating specific disorders.
Derek Phillips, PhD, the program’s executive director, said that when he took the position in 2020, enrollment was capped at 45 students, but “we were not routinely enrolling the maximum.” Now, even with class size increased to 60 “we are consistently full and have a waitlist,” he told Medscape Medical News.
Interest is being driven in part by new laws in Colorado (2023) and Utah, said Phillips. But many are enrolling without intending to write a prescription, he said. The degree gives graduates the ability to better collaborate with other clinicians, teach clinical psychopharmacology, and be expert witnesses in medico-legal cases, he said.
In addition, the training gives students “a balanced and thorough biopsychosocial understanding of our patients,” he said. Students also see the “potential of being able to be a ‘one-stop-stop’ of mental health services,” said Phillips.
The American Board of Professional Psychology is developing a board certification in clinical psychopharmacology.
The APA states on its website that prescribing psychologists have “more training in diagnosing and treating (including prescribing) mental health disorders than primary care physicians.”
However, critics argue that the training falls short. Most psychologists, said Robiner, have not completed the undergraduate prerequisites — such as anatomy, physiology, and chemistry — that are required for other prescribing professionals.
In a 2019 article comparing the training of prescribing professionals, Robiner and colleagues reported that psychiatrists undergo 4- to 6-week rotations during medical school and accumulate 8000 clinical hours focused on psychiatric conditions over the course of their 4-year residencies.
States set requirements for clinical hours for prescribing psychologists, but they are generally elective and completed after individuals receive a master’s degree.
Robiner said psychologists aren’t trained in evaluating drug-related adverse events. “If you show a psychologist a rash, they have no idea whether that rash might be a medication adverse effect or poison ivy,” he said.
Trestman pointed out that many psychotropic medications have black box warnings. “The risk of toxicity is by no means trivial, and the majority of people who are seeking care in psychiatry have multiple comorbidities,” he said. “Giving people the equivalent of more or less 10 weeks of training is just woefully inappropriate,” Trestman said.
Increase in Access?
Psychology’s main argument for expanding its scope of practice is that it will increase the number of clinicians available to provide behavioral and mental health care.
Critics said that is a failed experiment, in part because so few psychologists have become prescribers, but also because most psychologists practice in the same areas as psychiatrists. Both specialists tend to cluster in urban regions, which already have high clinician density, said Trestman.
Psychologists are not practicing in underserved rural areas, as even APA data show. A 2018 APA snapshot of the workforce found that the highest density of psychologists was in Washington, DC, Massachusetts, and New York. South Carolina, West Virginia and Mississippi had the fewest number of psychologists per 100,000 people.
The University of Washington Rural Health Research Center reported in 2022 that in 2021, almost half of rural counties did not have a psychologist compared with 15.7% of urban counties.
Psychiatrists also are concentrated on the coasts and New England, according to a study by Ohio State researchers. The highest densities were in Washington, DC (79 per 100,000), Massachusetts (45.3), Rhode Island (42.6), Connecticut (38.6) and Vermont (37.7), whereas the lowest densities were in Idaho (11.8), Mississippi (11.8), Wyoming (12.4), Alabama (13.1), and Indiana (13.5). The study estimated that there were 57,163 psychiatrists responsible for the care of 333,287,557 Americans. “Clinical psychologists, psychotherapists, and counselors can provide alternative forms of intervention, though access to such services is also poor in rural areas,” wrote the authors.
The APA counters with data it says shows that RxP may have increased access. Using the number of psychology practices as a proxy for supply, the authors reported that practices grew in New Mexico, Illinois, Iowa, and Idaho — states that have implemented prescription privileges. Overall, there was an increase of 0.8047 practices per 100,000 residents per county.
However, the access argument “is seriously challenged by the reality of the limited number of psychologists who complete the pathway to prescribing,” Robiner and his colleague Tanya Tompkins countered in Clinical Psychology. They note that in Idaho — a state with shortages of psychologists and psychiatrists — just 10 of the state’s 615 psychologists had prescriptive authority. An estimated 5131 nonpsychologists are prescribers.
Robiner and Tompkins noted that it’s not clear why so few psychologists are pursuing RxP but that many seem to be unaware of the possibility.
Do Benefits Outweigh the Harms?
There is not a large body of literature assessing the harms or benefits of prescribing privileges for psychologists.
Baker shared several studies by Phillip Hughes, PhD, an outcomes researcher at the University of North Carolina Eshelman School of Pharmacy, Chapel Hill, North Carolina. In one study, Hughes found that patients of prescribing psychologists had a 24% lower rate of adverse drug events than patients of psychiatrists. Psychologists’ patients had lower rates of psychotropic polypharmacy but similar rates of emergency room use.
In another paper Hughes suggested that deaths attributable to mental illness had declined in New Mexico after it passed its law. There was no change in Louisiana.
With little evidence of harm — and ongoing provider shortages — making use of nonphysician prescribers is gaining traction with policymakers, claims the psychology association’s Baker, adding that in Utah, the Republican governor was the biggest supporter.
But psychiatrists argue that it’s more important to increase their numbers. Congress agreed in 2021 and 2023 to add 1200 new residency slots — in every specialty — to ease physician shortages. The Centers for Medicare & Medicaid Services recently announced that 70% of the new slots for July 2025 will go to primary care and psychiatry.
“Once those positions are in place, it will be four more years before the first crop of new psychiatrists come out,” noted Trestman. “None of these fixes are quick,” he said.
Baker, Robiner, and Trestman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Earlier in 2024, Utah became the seventh state to allow psychologists with the proper training to prescribe psychotropic medications, giving supporters reason to hope that more states might support expanding this scope of practice.
However, the American Psychiatric Association — and some psychologists — oppose granting psychologists this privilege, arguing that the training offered is insufficient and could jeopardize patient safety.
The controversy over whether psychologists should be allowed to prescribe is as old as the so-called RxP movement itself, which began in the early 1990s.
Psychologists have not rushed to become licensed prescribers. After three decades, an estimated 226 psychologists — representing just 0.14% of all those licensed in the United States — have been authorized to prescribe in the six states and one territory where it has been legalized, according to a just-published study in Clinical Psychology.
These are Colorado, Idaho, Illinois, Iowa, Louisiana, New Mexico, and Guam. Data from the study show that only 73 psychologists are prescribing in New Mexico, which authorized it in 2002.
Less is known about the number of psychologists who are prescribing under allowances in the Department of Defense, Indian Health Service and US Public Health Service.
Some psychologists — and the American Psychological Association (APA) — believe that the persistence of the opioid epidemic coupled with a continued lack of access to mental health care for millions of Americans will bring more legislators on-side.
“I feel like we’re on an upswing again,” Deborah Baker, director of legal and regulatory policy for the APA, told Medscape Medical News. “The access issue continues to be a perennial kind of driver.” She noted that at least six states pursued expanding privileges this year.
Robert L. Trestman, MD, chair of the American Psychiatric Association’s Council on Healthcare Systems and Financing, said he doesn’t see new momentum. The interest in having psychologists prescribe “continues to trickle based on just the frustration that people have about not getting adequate access to psychiatry,” he told Medscape Medical News.
While states may be trying to increase access to care, granting psychologists privileges is “not a very effective way of doing it,” said Trestman, Chair of Psychiatry and Behavioral Medicine at Virginia Tech Carilion School of Medicine in Roanoke. Psychologists are needed to deliver psychotherapy, he said. “It makes almost no sense to try to make them into pseudo medical professionals,” said Trestman. “It just exposes people to risks.”
William Robiner, PhD — author of Clinical Psychology study — is a long-time opponent of RxP. The psychologist told Medscape Medical News he’s concerned about patient safety and “about some of the disingenuous reasons” that psychologists want to prescribe. Among these are the ability to increase status and income, said Robiner, a professor of medicine at the University of Minnesota Medical School, Minneapolis, and a board member of Psychologists Opposed to Prescription Privileges for Psychologists.
Adequate Training?
Only PhD and PsyD psychologists are eligible for RxP training, which entails a master’s in clinical psychopharmacology. After receiving the master’s, they must pass the Psychopharmacology Examination for Psychologists and then are only permitted to prescribe medications for mental health disorders.
They must also obtain a Drug Enforcement Administration license but can’t write prescriptions for schedule 2 medications. In some states, psychologists can prescribe buprenorphine and other opioid use disorder medications.
The APA has developed guidance for master’s programs, which currently number just over a handful in the United States.
At Fairleigh Dickinson University in New Jersey, students enrolled in the master’s program — a distance-learning format — complete 10 courses over five 15-week semesters. The curriculum spans a range of topics, from foundational sciences and legal and ethical considerations to strategies for treating specific disorders.
Derek Phillips, PhD, the program’s executive director, said that when he took the position in 2020, enrollment was capped at 45 students, but “we were not routinely enrolling the maximum.” Now, even with class size increased to 60 “we are consistently full and have a waitlist,” he told Medscape Medical News.
Interest is being driven in part by new laws in Colorado (2023) and Utah, said Phillips. But many are enrolling without intending to write a prescription, he said. The degree gives graduates the ability to better collaborate with other clinicians, teach clinical psychopharmacology, and be expert witnesses in medico-legal cases, he said.
In addition, the training gives students “a balanced and thorough biopsychosocial understanding of our patients,” he said. Students also see the “potential of being able to be a ‘one-stop-stop’ of mental health services,” said Phillips.
The American Board of Professional Psychology is developing a board certification in clinical psychopharmacology.
The APA states on its website that prescribing psychologists have “more training in diagnosing and treating (including prescribing) mental health disorders than primary care physicians.”
However, critics argue that the training falls short. Most psychologists, said Robiner, have not completed the undergraduate prerequisites — such as anatomy, physiology, and chemistry — that are required for other prescribing professionals.
In a 2019 article comparing the training of prescribing professionals, Robiner and colleagues reported that psychiatrists undergo 4- to 6-week rotations during medical school and accumulate 8000 clinical hours focused on psychiatric conditions over the course of their 4-year residencies.
States set requirements for clinical hours for prescribing psychologists, but they are generally elective and completed after individuals receive a master’s degree.
Robiner said psychologists aren’t trained in evaluating drug-related adverse events. “If you show a psychologist a rash, they have no idea whether that rash might be a medication adverse effect or poison ivy,” he said.
Trestman pointed out that many psychotropic medications have black box warnings. “The risk of toxicity is by no means trivial, and the majority of people who are seeking care in psychiatry have multiple comorbidities,” he said. “Giving people the equivalent of more or less 10 weeks of training is just woefully inappropriate,” Trestman said.
Increase in Access?
Psychology’s main argument for expanding its scope of practice is that it will increase the number of clinicians available to provide behavioral and mental health care.
Critics said that is a failed experiment, in part because so few psychologists have become prescribers, but also because most psychologists practice in the same areas as psychiatrists. Both specialists tend to cluster in urban regions, which already have high clinician density, said Trestman.
Psychologists are not practicing in underserved rural areas, as even APA data show. A 2018 APA snapshot of the workforce found that the highest density of psychologists was in Washington, DC, Massachusetts, and New York. South Carolina, West Virginia and Mississippi had the fewest number of psychologists per 100,000 people.
The University of Washington Rural Health Research Center reported in 2022 that in 2021, almost half of rural counties did not have a psychologist compared with 15.7% of urban counties.
Psychiatrists also are concentrated on the coasts and New England, according to a study by Ohio State researchers. The highest densities were in Washington, DC (79 per 100,000), Massachusetts (45.3), Rhode Island (42.6), Connecticut (38.6) and Vermont (37.7), whereas the lowest densities were in Idaho (11.8), Mississippi (11.8), Wyoming (12.4), Alabama (13.1), and Indiana (13.5). The study estimated that there were 57,163 psychiatrists responsible for the care of 333,287,557 Americans. “Clinical psychologists, psychotherapists, and counselors can provide alternative forms of intervention, though access to such services is also poor in rural areas,” wrote the authors.
The APA counters with data it says shows that RxP may have increased access. Using the number of psychology practices as a proxy for supply, the authors reported that practices grew in New Mexico, Illinois, Iowa, and Idaho — states that have implemented prescription privileges. Overall, there was an increase of 0.8047 practices per 100,000 residents per county.
However, the access argument “is seriously challenged by the reality of the limited number of psychologists who complete the pathway to prescribing,” Robiner and his colleague Tanya Tompkins countered in Clinical Psychology. They note that in Idaho — a state with shortages of psychologists and psychiatrists — just 10 of the state’s 615 psychologists had prescriptive authority. An estimated 5131 nonpsychologists are prescribers.
Robiner and Tompkins noted that it’s not clear why so few psychologists are pursuing RxP but that many seem to be unaware of the possibility.
Do Benefits Outweigh the Harms?
There is not a large body of literature assessing the harms or benefits of prescribing privileges for psychologists.
Baker shared several studies by Phillip Hughes, PhD, an outcomes researcher at the University of North Carolina Eshelman School of Pharmacy, Chapel Hill, North Carolina. In one study, Hughes found that patients of prescribing psychologists had a 24% lower rate of adverse drug events than patients of psychiatrists. Psychologists’ patients had lower rates of psychotropic polypharmacy but similar rates of emergency room use.
In another paper Hughes suggested that deaths attributable to mental illness had declined in New Mexico after it passed its law. There was no change in Louisiana.
With little evidence of harm — and ongoing provider shortages — making use of nonphysician prescribers is gaining traction with policymakers, claims the psychology association’s Baker, adding that in Utah, the Republican governor was the biggest supporter.
But psychiatrists argue that it’s more important to increase their numbers. Congress agreed in 2021 and 2023 to add 1200 new residency slots — in every specialty — to ease physician shortages. The Centers for Medicare & Medicaid Services recently announced that 70% of the new slots for July 2025 will go to primary care and psychiatry.
“Once those positions are in place, it will be four more years before the first crop of new psychiatrists come out,” noted Trestman. “None of these fixes are quick,” he said.
Baker, Robiner, and Trestman reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Leaded Gas Exposure Tied to 151 Million Excess Cases of Mental Illness Cases
Childhood exposure to leaded gasoline via car exhaust over the past 75 years is linked to 151 million excess cases of psychiatric disorders in the United States, new research suggested.
The data revealed that the group most heavily exposed to lead — individuals born between 1966 and 1986, commonly known as Generation X — experienced the biggest increases in mental health issues.
Within this cohort, those born between 1966 and 1970 were affected the most. This timeline, the investigators noted, aligns with the peak use of leaded gasoline during the mid-1960s and 1970s. Specifically for this group, overall mental health issues increase by 0.35 times the average, anxiety and depression by 1.75 times, and attention-deficit/hyperactivity disorder (ADHD) symptoms by 1.17 times.
“Lead exposure across the country has probably played a significant role in worsening mental health in ways that had previously been unappreciated and invisible,” study investigator Aaron Reuben, PhD, assistant professor of clinical neuropsychology at the University of Virginia, Charlottesville, said in an interview.
However, the investigators emphasized that the study only establishes an association and not a causal relationship between leaded gas exposure and subsequent psychopathology.
The findings were published online on December 4 in The Journal of Child Psychology and Psychiatry.
Lead Astray?
Leaded gasoline was first used in the 1920s to stop engine knocking and improve performance. Despite early evidence of neurotoxicity its use continued until 1996 when it was banned.
The investigators noted that over half of the current US population was exposed to adverse lead levels from gasoline in childhood. However, they added the total contribution of childhood lead exposure to the population’s mental health and personality has not previously been evaluated.
For the study, the researchers combined serial, cross-sectional data on blood lead levels (BLLs) from the National Health and Nutrition Examination Survey and integrated it with historical data on gasoline usage, allowing them to estimate childhood BLLs in the United States from 1940 to 2015.
They calculated the impact of lead exposure on mental health using general psychopathology factor points, also referred to as mental illness points, which function similarly to IQ points.
These measures used were based on a prior study led by Reuben in 2019 from a New Zealand cohort and a study by a different group that followed a longitudinal birth cohort in Chicago .
Using these data, the researchers calculated population-level elevations in mental health symptoms on the basis of lead exposure and five key psychiatric outcomes in the US population.
These included general psychopathology, which reflects an individual’s overall liability to mental disorders and was scaled to match IQ scores with a mean of 100 and an SD of 15.
They also evaluated symptoms of internalizing disorders including anxiety and depression and ADHD and standardized them to a mean of 0 and an SD of 1.
The researchers also looked at differences in the personality traits of neuroticism, which relate to emotional instability and conscientiousness and reflect organization and responsibility. Both of these were assessed using similar standardized scales.
151 Million Excess Cases of Mental Illness
Using this approach, the researchers were able to assess the historical and long-term implications of lead exposure on mental health and personality traits in the US population over time.
Results showed that during the peak era of leaded gasoline in the United States, children were routinely exposed to lead levels three to six times higher than the current reference point for clinical concern (3.5 μg/dL of blood), the authors noted.
While the United States banned leaded gasoline in 1996, lead can still be present in water pipes, old paint, and soil. The Centers for Disease Control and Prevention cautions that no BLLs are safe.
The investigators found that between 1940 and 2015, the US population gained 602 million general psychopathology points, which the investigators said equates to 151 million excess instances of mental disorders that are primarily, but not completely, attributable to early life exposure to leaded gasoline.
“Assuming that published lead-psychopathology associations are causal and not purely correlational, we estimate that by 2015, the US population had gained 602 million General Psychopathology factor points because of exposure arising from leaded gasoline, reflecting a 0.13 standard deviation increase in overall liability to mental illness in the population and an estimated 151 million excess mental disorders attributable to lead exposure,” the researchers wrote.
Specific effects included a 0.64 SD increase in anxiety and depression symptoms, a 0.42 SD increase in ADHD symptoms, a 0.14 SD increase in ADHD symptoms, a 0.14 SD increase in emotional instability (neuroticism), and a 0.20 SD decrease in traits like organization and responsibility (conscientiousness).
These mental health and personality changes were not distributed evenly among the generations, most significant in those born between 1966 and 1986, the investigators reported.
For example, children between 1966 and 1970, the period when leaded gasoline use was at its peak, had BLLs > 5 μg/dL and experienced a 1.75 SD increase in internalizing symptoms and a 1.17 SD increase in ADHD symptoms.
Assess Lead Risk
The study had several limitations. Causality could not be established, and the accuracy of the estimates relied on findings from the researchers’ two previous key studies.
However, the investigators noted that these findings have been replicated across multiple cohorts and settings. Additionally, the results may not be fully generalizable to the entire US population, as one study was based in New Zealand and the other in urban Chicago.
Reuben cautioned that even though gasoline and new paint no longer contain lead, exposure is still possible.
“We saw this most acutely in Flint, Michigan,” when aging water pipes exposed more than 100,000 residents to high lead levels in 2014. “This situation made us aware that thousands of communities are exposed to lead service lines.”
He recommended that physicians consider screening patients for lead exposure — both new and old. Experts estimate that 90% of lead in the body is stored in the skeleton and can be released back into the bloodstream over time, particularly in cases of calcium deficiency, pregnancy, or osteoporosis.
While reversing childhood lead exposure is not possible, Reuben noted that healthy lifestyle choices and multimodal interventions such as medication and therapy can effectively address and alleviate mental illness.
‘Legacy of Lead’
In a comment, Terrie Moffitt, PhD, who was not involved in the research, said the study “is important because it gives us a crystal ball to see into the future of children living with lead today.”
“It’s called the ‘legacy of lead,’ and what a legacy,” said Moffitt, professor in the Department of Psychology and Neuroscience, Nannerl O. Keohane University at Duke University in Durham, North Carolina.
Moffitt emphasized that children exposed to lead today often live in areas of poverty and disadvantage, making it difficult to disentangle the potential effects of lead exposure from those of childhood adversity that predispose individuals to mental illness.
“This study tells us about lead’s damage in an era when it was everywhere, not just in poor communities,” she said.
The study was funded by a fellowship from the National Institute of Environmental Health Sciences. Reuben and Moffitt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Childhood exposure to leaded gasoline via car exhaust over the past 75 years is linked to 151 million excess cases of psychiatric disorders in the United States, new research suggested.
The data revealed that the group most heavily exposed to lead — individuals born between 1966 and 1986, commonly known as Generation X — experienced the biggest increases in mental health issues.
Within this cohort, those born between 1966 and 1970 were affected the most. This timeline, the investigators noted, aligns with the peak use of leaded gasoline during the mid-1960s and 1970s. Specifically for this group, overall mental health issues increase by 0.35 times the average, anxiety and depression by 1.75 times, and attention-deficit/hyperactivity disorder (ADHD) symptoms by 1.17 times.
“Lead exposure across the country has probably played a significant role in worsening mental health in ways that had previously been unappreciated and invisible,” study investigator Aaron Reuben, PhD, assistant professor of clinical neuropsychology at the University of Virginia, Charlottesville, said in an interview.
However, the investigators emphasized that the study only establishes an association and not a causal relationship between leaded gas exposure and subsequent psychopathology.
The findings were published online on December 4 in The Journal of Child Psychology and Psychiatry.
Lead Astray?
Leaded gasoline was first used in the 1920s to stop engine knocking and improve performance. Despite early evidence of neurotoxicity its use continued until 1996 when it was banned.
The investigators noted that over half of the current US population was exposed to adverse lead levels from gasoline in childhood. However, they added the total contribution of childhood lead exposure to the population’s mental health and personality has not previously been evaluated.
For the study, the researchers combined serial, cross-sectional data on blood lead levels (BLLs) from the National Health and Nutrition Examination Survey and integrated it with historical data on gasoline usage, allowing them to estimate childhood BLLs in the United States from 1940 to 2015.
They calculated the impact of lead exposure on mental health using general psychopathology factor points, also referred to as mental illness points, which function similarly to IQ points.
These measures used were based on a prior study led by Reuben in 2019 from a New Zealand cohort and a study by a different group that followed a longitudinal birth cohort in Chicago .
Using these data, the researchers calculated population-level elevations in mental health symptoms on the basis of lead exposure and five key psychiatric outcomes in the US population.
These included general psychopathology, which reflects an individual’s overall liability to mental disorders and was scaled to match IQ scores with a mean of 100 and an SD of 15.
They also evaluated symptoms of internalizing disorders including anxiety and depression and ADHD and standardized them to a mean of 0 and an SD of 1.
The researchers also looked at differences in the personality traits of neuroticism, which relate to emotional instability and conscientiousness and reflect organization and responsibility. Both of these were assessed using similar standardized scales.
151 Million Excess Cases of Mental Illness
Using this approach, the researchers were able to assess the historical and long-term implications of lead exposure on mental health and personality traits in the US population over time.
Results showed that during the peak era of leaded gasoline in the United States, children were routinely exposed to lead levels three to six times higher than the current reference point for clinical concern (3.5 μg/dL of blood), the authors noted.
While the United States banned leaded gasoline in 1996, lead can still be present in water pipes, old paint, and soil. The Centers for Disease Control and Prevention cautions that no BLLs are safe.
The investigators found that between 1940 and 2015, the US population gained 602 million general psychopathology points, which the investigators said equates to 151 million excess instances of mental disorders that are primarily, but not completely, attributable to early life exposure to leaded gasoline.
“Assuming that published lead-psychopathology associations are causal and not purely correlational, we estimate that by 2015, the US population had gained 602 million General Psychopathology factor points because of exposure arising from leaded gasoline, reflecting a 0.13 standard deviation increase in overall liability to mental illness in the population and an estimated 151 million excess mental disorders attributable to lead exposure,” the researchers wrote.
Specific effects included a 0.64 SD increase in anxiety and depression symptoms, a 0.42 SD increase in ADHD symptoms, a 0.14 SD increase in ADHD symptoms, a 0.14 SD increase in emotional instability (neuroticism), and a 0.20 SD decrease in traits like organization and responsibility (conscientiousness).
These mental health and personality changes were not distributed evenly among the generations, most significant in those born between 1966 and 1986, the investigators reported.
For example, children between 1966 and 1970, the period when leaded gasoline use was at its peak, had BLLs > 5 μg/dL and experienced a 1.75 SD increase in internalizing symptoms and a 1.17 SD increase in ADHD symptoms.
Assess Lead Risk
The study had several limitations. Causality could not be established, and the accuracy of the estimates relied on findings from the researchers’ two previous key studies.
However, the investigators noted that these findings have been replicated across multiple cohorts and settings. Additionally, the results may not be fully generalizable to the entire US population, as one study was based in New Zealand and the other in urban Chicago.
Reuben cautioned that even though gasoline and new paint no longer contain lead, exposure is still possible.
“We saw this most acutely in Flint, Michigan,” when aging water pipes exposed more than 100,000 residents to high lead levels in 2014. “This situation made us aware that thousands of communities are exposed to lead service lines.”
He recommended that physicians consider screening patients for lead exposure — both new and old. Experts estimate that 90% of lead in the body is stored in the skeleton and can be released back into the bloodstream over time, particularly in cases of calcium deficiency, pregnancy, or osteoporosis.
While reversing childhood lead exposure is not possible, Reuben noted that healthy lifestyle choices and multimodal interventions such as medication and therapy can effectively address and alleviate mental illness.
‘Legacy of Lead’
In a comment, Terrie Moffitt, PhD, who was not involved in the research, said the study “is important because it gives us a crystal ball to see into the future of children living with lead today.”
“It’s called the ‘legacy of lead,’ and what a legacy,” said Moffitt, professor in the Department of Psychology and Neuroscience, Nannerl O. Keohane University at Duke University in Durham, North Carolina.
Moffitt emphasized that children exposed to lead today often live in areas of poverty and disadvantage, making it difficult to disentangle the potential effects of lead exposure from those of childhood adversity that predispose individuals to mental illness.
“This study tells us about lead’s damage in an era when it was everywhere, not just in poor communities,” she said.
The study was funded by a fellowship from the National Institute of Environmental Health Sciences. Reuben and Moffitt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Childhood exposure to leaded gasoline via car exhaust over the past 75 years is linked to 151 million excess cases of psychiatric disorders in the United States, new research suggested.
The data revealed that the group most heavily exposed to lead — individuals born between 1966 and 1986, commonly known as Generation X — experienced the biggest increases in mental health issues.
Within this cohort, those born between 1966 and 1970 were affected the most. This timeline, the investigators noted, aligns with the peak use of leaded gasoline during the mid-1960s and 1970s. Specifically for this group, overall mental health issues increase by 0.35 times the average, anxiety and depression by 1.75 times, and attention-deficit/hyperactivity disorder (ADHD) symptoms by 1.17 times.
“Lead exposure across the country has probably played a significant role in worsening mental health in ways that had previously been unappreciated and invisible,” study investigator Aaron Reuben, PhD, assistant professor of clinical neuropsychology at the University of Virginia, Charlottesville, said in an interview.
However, the investigators emphasized that the study only establishes an association and not a causal relationship between leaded gas exposure and subsequent psychopathology.
The findings were published online on December 4 in The Journal of Child Psychology and Psychiatry.
Lead Astray?
Leaded gasoline was first used in the 1920s to stop engine knocking and improve performance. Despite early evidence of neurotoxicity its use continued until 1996 when it was banned.
The investigators noted that over half of the current US population was exposed to adverse lead levels from gasoline in childhood. However, they added the total contribution of childhood lead exposure to the population’s mental health and personality has not previously been evaluated.
For the study, the researchers combined serial, cross-sectional data on blood lead levels (BLLs) from the National Health and Nutrition Examination Survey and integrated it with historical data on gasoline usage, allowing them to estimate childhood BLLs in the United States from 1940 to 2015.
They calculated the impact of lead exposure on mental health using general psychopathology factor points, also referred to as mental illness points, which function similarly to IQ points.
These measures used were based on a prior study led by Reuben in 2019 from a New Zealand cohort and a study by a different group that followed a longitudinal birth cohort in Chicago .
Using these data, the researchers calculated population-level elevations in mental health symptoms on the basis of lead exposure and five key psychiatric outcomes in the US population.
These included general psychopathology, which reflects an individual’s overall liability to mental disorders and was scaled to match IQ scores with a mean of 100 and an SD of 15.
They also evaluated symptoms of internalizing disorders including anxiety and depression and ADHD and standardized them to a mean of 0 and an SD of 1.
The researchers also looked at differences in the personality traits of neuroticism, which relate to emotional instability and conscientiousness and reflect organization and responsibility. Both of these were assessed using similar standardized scales.
151 Million Excess Cases of Mental Illness
Using this approach, the researchers were able to assess the historical and long-term implications of lead exposure on mental health and personality traits in the US population over time.
Results showed that during the peak era of leaded gasoline in the United States, children were routinely exposed to lead levels three to six times higher than the current reference point for clinical concern (3.5 μg/dL of blood), the authors noted.
While the United States banned leaded gasoline in 1996, lead can still be present in water pipes, old paint, and soil. The Centers for Disease Control and Prevention cautions that no BLLs are safe.
The investigators found that between 1940 and 2015, the US population gained 602 million general psychopathology points, which the investigators said equates to 151 million excess instances of mental disorders that are primarily, but not completely, attributable to early life exposure to leaded gasoline.
“Assuming that published lead-psychopathology associations are causal and not purely correlational, we estimate that by 2015, the US population had gained 602 million General Psychopathology factor points because of exposure arising from leaded gasoline, reflecting a 0.13 standard deviation increase in overall liability to mental illness in the population and an estimated 151 million excess mental disorders attributable to lead exposure,” the researchers wrote.
Specific effects included a 0.64 SD increase in anxiety and depression symptoms, a 0.42 SD increase in ADHD symptoms, a 0.14 SD increase in ADHD symptoms, a 0.14 SD increase in emotional instability (neuroticism), and a 0.20 SD decrease in traits like organization and responsibility (conscientiousness).
These mental health and personality changes were not distributed evenly among the generations, most significant in those born between 1966 and 1986, the investigators reported.
For example, children between 1966 and 1970, the period when leaded gasoline use was at its peak, had BLLs > 5 μg/dL and experienced a 1.75 SD increase in internalizing symptoms and a 1.17 SD increase in ADHD symptoms.
Assess Lead Risk
The study had several limitations. Causality could not be established, and the accuracy of the estimates relied on findings from the researchers’ two previous key studies.
However, the investigators noted that these findings have been replicated across multiple cohorts and settings. Additionally, the results may not be fully generalizable to the entire US population, as one study was based in New Zealand and the other in urban Chicago.
Reuben cautioned that even though gasoline and new paint no longer contain lead, exposure is still possible.
“We saw this most acutely in Flint, Michigan,” when aging water pipes exposed more than 100,000 residents to high lead levels in 2014. “This situation made us aware that thousands of communities are exposed to lead service lines.”
He recommended that physicians consider screening patients for lead exposure — both new and old. Experts estimate that 90% of lead in the body is stored in the skeleton and can be released back into the bloodstream over time, particularly in cases of calcium deficiency, pregnancy, or osteoporosis.
While reversing childhood lead exposure is not possible, Reuben noted that healthy lifestyle choices and multimodal interventions such as medication and therapy can effectively address and alleviate mental illness.
‘Legacy of Lead’
In a comment, Terrie Moffitt, PhD, who was not involved in the research, said the study “is important because it gives us a crystal ball to see into the future of children living with lead today.”
“It’s called the ‘legacy of lead,’ and what a legacy,” said Moffitt, professor in the Department of Psychology and Neuroscience, Nannerl O. Keohane University at Duke University in Durham, North Carolina.
Moffitt emphasized that children exposed to lead today often live in areas of poverty and disadvantage, making it difficult to disentangle the potential effects of lead exposure from those of childhood adversity that predispose individuals to mental illness.
“This study tells us about lead’s damage in an era when it was everywhere, not just in poor communities,” she said.
The study was funded by a fellowship from the National Institute of Environmental Health Sciences. Reuben and Moffitt reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CHILD PSYCHOLOGY AND PSYCHIATRY
Rise in Psychotherapy Use Exposes Access Inequities
Outpatient psychotherapy use in the United States rose sharply between 2018 and 2021, an increase that was driven primarily by young, urban professionals with higher family incomes, new data exposed significant disparities in access to this treatment type.
Results of a large population-based repeated cross-sectional study revealed that psychotherapy use increased significantly faster for women vs men, younger individuals vs their older counterparts, college graduates than those without a high school diploma, and privately insured vs publicly insured individuals.
Overall, psychotherapy use increased significantly faster among several socioeconomically advantaged groups, and inequalities were evident in teletherapy access. These trends and patterns highlight a need for clinical interventions and healthcare policies to broaden access to psychotherapy, including teletherapy, the authors noted.
“While psychotherapy access has expanded in the US, there’s concern that recent gains may not be equally distributed, despite or maybe because of the growth of teletherapy,” study author Mark Olfson, MD, MPH, Department of Psychiatry, Mailman School of Public Health, Columbia University, New York City, said in a press release.
“This increase in psychotherapy use, driven by the rise of teletherapy, has largely benefited socioeconomically advantaged adults with mild to moderate distress,” he added.
The findings were published online in JAMA Psychiatry.
Psychotherapy Uptick
Psychotherapy is among the most widely used methods for delivering mental health care in the United States. A recent study conducted by Olfson and colleagues showed that the percentage of US adults receiving psychotherapy increased from 6.5% in 2018 to 8.5% in 2021. However, it was unclear how this overall increase varied across different sociodemographic groups or levels of psychological distress.
Analyzing population-level trends in psychotherapy use can identify sociodemographic groups with declining access to services, providing valuable insights for developing initiatives to improve accessibility, the investigators noted.
To evaluate national trends in psychotherapy use, the researchers analyzed data from the 2018-2021 Medical Expenditure Panel Survey (MEPS). These are yearly surveys representing noninstitutionalized adults across the United States.
The study included 89,619 adults. Of these, 51.5% were women, nearly half were aged 35-64 years, and 62.2% were White individuals. The study used a repeated cross-sectional design with new, nationally representative samples of about 22,000 participants each year.
The investigators tracked the overall increase in psychotherapy use, especially among groups at higher risk for untreated mental health conditions. They also examined how video-based therapy (teletherapy) was being used, paying particular attention to differences in access among various demographic groups and levels of psychological distress, given ongoing concerns about equity in telehealth access.
Psychological distress was measured using the Kessler-6 scale, with scores ≥ 13 defining serious psychological distress, 1-12 defining mild to moderate distress, and 0 defining no distress.
Psychotherapy use increased across all racial and ethnic groups, with rates rising among Black (5.4% to 7.1%), Hispanic (4.1% to 5.8%), White (7.5% to 9.8%), and other, non-Hispanic (4.8% to 6.6%) individuals.
Participants with mild to moderate distress experienced the greatest increases in psychotherapy use (8.6% to 11.2%, respectively).
After adjusting for age, sex, and level of psychological distress, investigators found that psychotherapy use increased to a greater degree among women (7.7% to 10.5%) vs men (5.2% to 6.3%), younger adults aged 18-34 years (8% to 11.9%) vs adults aged 65 years or older (3.6% to 4.6%), and college graduates (7.6% to 11.4%) than those without a high school diploma (5.5% to 7%).
A National Priority
Adults with higher incomes — defined as two to four times the federal poverty level — had greater increases in psychotherapy use (5.7% to 8.2%) than those below the poverty level (9.7% to 10%).
Unsurprisingly, privately insured individuals saw more significant increases (6.1% to 8.9%) than publicly insured individuals (8.8% to 8.8%). Also, there was a larger increase in psychotherapy use among employed individuals (5.7% to 8.9%) than among unemployed individuals (10.8% to 10.5%).
In addition, there was a significantly greater increase in psychotherapy use among urban residents (6.5% to 8.7%), whereas it declined among rural residents (6.4% to 5.9%).
Data on teletherapy use from 2021 revealed that 39.9% of adults receiving psychotherapy had one or more teletherapy visits.
Teletherapy use was higher among younger adults, women, college-educated individuals, those with higher incomes, those with private insurance, and those who lived in urban areas.
The authors noted that while teletherapy is intended to remove transportation and time barriers and was widely adopted during the pandemic, the findings show that those who were older, less educated, and with lower incomes were less likely to use it.
Notably, urban residents were more than twice as likely to use teletherapy than rural residents. Prior to the COVID-19 pandemic, teletherapy was viewed as a potential solution for individuals living in rural areas facing a shortage of mental health professionals, but study results showed that “teletherapy does not appear to have addressed this public health challenge,” the investigators wrote.
“The trends we are seeing underscore the need for targeted interventions and health policies that expand psychotherapy access to underserved groups,” said Olfson.
“Ensuring that individuals in psychological distress can access care is a national priority. Addressing technical and financial barriers to teletherapy could help bridge the gap in access and promote equity in mental health care,” he added.
Study limitations included a possible underreporting of psychotherapy use by participants. In addition, MEPS does not include nursing home residents, incarcerated, and unhoused individuals.
Study funding was not disclosed. Olfson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Outpatient psychotherapy use in the United States rose sharply between 2018 and 2021, an increase that was driven primarily by young, urban professionals with higher family incomes, new data exposed significant disparities in access to this treatment type.
Results of a large population-based repeated cross-sectional study revealed that psychotherapy use increased significantly faster for women vs men, younger individuals vs their older counterparts, college graduates than those without a high school diploma, and privately insured vs publicly insured individuals.
Overall, psychotherapy use increased significantly faster among several socioeconomically advantaged groups, and inequalities were evident in teletherapy access. These trends and patterns highlight a need for clinical interventions and healthcare policies to broaden access to psychotherapy, including teletherapy, the authors noted.
“While psychotherapy access has expanded in the US, there’s concern that recent gains may not be equally distributed, despite or maybe because of the growth of teletherapy,” study author Mark Olfson, MD, MPH, Department of Psychiatry, Mailman School of Public Health, Columbia University, New York City, said in a press release.
“This increase in psychotherapy use, driven by the rise of teletherapy, has largely benefited socioeconomically advantaged adults with mild to moderate distress,” he added.
The findings were published online in JAMA Psychiatry.
Psychotherapy Uptick
Psychotherapy is among the most widely used methods for delivering mental health care in the United States. A recent study conducted by Olfson and colleagues showed that the percentage of US adults receiving psychotherapy increased from 6.5% in 2018 to 8.5% in 2021. However, it was unclear how this overall increase varied across different sociodemographic groups or levels of psychological distress.
Analyzing population-level trends in psychotherapy use can identify sociodemographic groups with declining access to services, providing valuable insights for developing initiatives to improve accessibility, the investigators noted.
To evaluate national trends in psychotherapy use, the researchers analyzed data from the 2018-2021 Medical Expenditure Panel Survey (MEPS). These are yearly surveys representing noninstitutionalized adults across the United States.
The study included 89,619 adults. Of these, 51.5% were women, nearly half were aged 35-64 years, and 62.2% were White individuals. The study used a repeated cross-sectional design with new, nationally representative samples of about 22,000 participants each year.
The investigators tracked the overall increase in psychotherapy use, especially among groups at higher risk for untreated mental health conditions. They also examined how video-based therapy (teletherapy) was being used, paying particular attention to differences in access among various demographic groups and levels of psychological distress, given ongoing concerns about equity in telehealth access.
Psychological distress was measured using the Kessler-6 scale, with scores ≥ 13 defining serious psychological distress, 1-12 defining mild to moderate distress, and 0 defining no distress.
Psychotherapy use increased across all racial and ethnic groups, with rates rising among Black (5.4% to 7.1%), Hispanic (4.1% to 5.8%), White (7.5% to 9.8%), and other, non-Hispanic (4.8% to 6.6%) individuals.
Participants with mild to moderate distress experienced the greatest increases in psychotherapy use (8.6% to 11.2%, respectively).
After adjusting for age, sex, and level of psychological distress, investigators found that psychotherapy use increased to a greater degree among women (7.7% to 10.5%) vs men (5.2% to 6.3%), younger adults aged 18-34 years (8% to 11.9%) vs adults aged 65 years or older (3.6% to 4.6%), and college graduates (7.6% to 11.4%) than those without a high school diploma (5.5% to 7%).
A National Priority
Adults with higher incomes — defined as two to four times the federal poverty level — had greater increases in psychotherapy use (5.7% to 8.2%) than those below the poverty level (9.7% to 10%).
Unsurprisingly, privately insured individuals saw more significant increases (6.1% to 8.9%) than publicly insured individuals (8.8% to 8.8%). Also, there was a larger increase in psychotherapy use among employed individuals (5.7% to 8.9%) than among unemployed individuals (10.8% to 10.5%).
In addition, there was a significantly greater increase in psychotherapy use among urban residents (6.5% to 8.7%), whereas it declined among rural residents (6.4% to 5.9%).
Data on teletherapy use from 2021 revealed that 39.9% of adults receiving psychotherapy had one or more teletherapy visits.
Teletherapy use was higher among younger adults, women, college-educated individuals, those with higher incomes, those with private insurance, and those who lived in urban areas.
The authors noted that while teletherapy is intended to remove transportation and time barriers and was widely adopted during the pandemic, the findings show that those who were older, less educated, and with lower incomes were less likely to use it.
Notably, urban residents were more than twice as likely to use teletherapy than rural residents. Prior to the COVID-19 pandemic, teletherapy was viewed as a potential solution for individuals living in rural areas facing a shortage of mental health professionals, but study results showed that “teletherapy does not appear to have addressed this public health challenge,” the investigators wrote.
“The trends we are seeing underscore the need for targeted interventions and health policies that expand psychotherapy access to underserved groups,” said Olfson.
“Ensuring that individuals in psychological distress can access care is a national priority. Addressing technical and financial barriers to teletherapy could help bridge the gap in access and promote equity in mental health care,” he added.
Study limitations included a possible underreporting of psychotherapy use by participants. In addition, MEPS does not include nursing home residents, incarcerated, and unhoused individuals.
Study funding was not disclosed. Olfson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Outpatient psychotherapy use in the United States rose sharply between 2018 and 2021, an increase that was driven primarily by young, urban professionals with higher family incomes, new data exposed significant disparities in access to this treatment type.
Results of a large population-based repeated cross-sectional study revealed that psychotherapy use increased significantly faster for women vs men, younger individuals vs their older counterparts, college graduates than those without a high school diploma, and privately insured vs publicly insured individuals.
Overall, psychotherapy use increased significantly faster among several socioeconomically advantaged groups, and inequalities were evident in teletherapy access. These trends and patterns highlight a need for clinical interventions and healthcare policies to broaden access to psychotherapy, including teletherapy, the authors noted.
“While psychotherapy access has expanded in the US, there’s concern that recent gains may not be equally distributed, despite or maybe because of the growth of teletherapy,” study author Mark Olfson, MD, MPH, Department of Psychiatry, Mailman School of Public Health, Columbia University, New York City, said in a press release.
“This increase in psychotherapy use, driven by the rise of teletherapy, has largely benefited socioeconomically advantaged adults with mild to moderate distress,” he added.
The findings were published online in JAMA Psychiatry.
Psychotherapy Uptick
Psychotherapy is among the most widely used methods for delivering mental health care in the United States. A recent study conducted by Olfson and colleagues showed that the percentage of US adults receiving psychotherapy increased from 6.5% in 2018 to 8.5% in 2021. However, it was unclear how this overall increase varied across different sociodemographic groups or levels of psychological distress.
Analyzing population-level trends in psychotherapy use can identify sociodemographic groups with declining access to services, providing valuable insights for developing initiatives to improve accessibility, the investigators noted.
To evaluate national trends in psychotherapy use, the researchers analyzed data from the 2018-2021 Medical Expenditure Panel Survey (MEPS). These are yearly surveys representing noninstitutionalized adults across the United States.
The study included 89,619 adults. Of these, 51.5% were women, nearly half were aged 35-64 years, and 62.2% were White individuals. The study used a repeated cross-sectional design with new, nationally representative samples of about 22,000 participants each year.
The investigators tracked the overall increase in psychotherapy use, especially among groups at higher risk for untreated mental health conditions. They also examined how video-based therapy (teletherapy) was being used, paying particular attention to differences in access among various demographic groups and levels of psychological distress, given ongoing concerns about equity in telehealth access.
Psychological distress was measured using the Kessler-6 scale, with scores ≥ 13 defining serious psychological distress, 1-12 defining mild to moderate distress, and 0 defining no distress.
Psychotherapy use increased across all racial and ethnic groups, with rates rising among Black (5.4% to 7.1%), Hispanic (4.1% to 5.8%), White (7.5% to 9.8%), and other, non-Hispanic (4.8% to 6.6%) individuals.
Participants with mild to moderate distress experienced the greatest increases in psychotherapy use (8.6% to 11.2%, respectively).
After adjusting for age, sex, and level of psychological distress, investigators found that psychotherapy use increased to a greater degree among women (7.7% to 10.5%) vs men (5.2% to 6.3%), younger adults aged 18-34 years (8% to 11.9%) vs adults aged 65 years or older (3.6% to 4.6%), and college graduates (7.6% to 11.4%) than those without a high school diploma (5.5% to 7%).
A National Priority
Adults with higher incomes — defined as two to four times the federal poverty level — had greater increases in psychotherapy use (5.7% to 8.2%) than those below the poverty level (9.7% to 10%).
Unsurprisingly, privately insured individuals saw more significant increases (6.1% to 8.9%) than publicly insured individuals (8.8% to 8.8%). Also, there was a larger increase in psychotherapy use among employed individuals (5.7% to 8.9%) than among unemployed individuals (10.8% to 10.5%).
In addition, there was a significantly greater increase in psychotherapy use among urban residents (6.5% to 8.7%), whereas it declined among rural residents (6.4% to 5.9%).
Data on teletherapy use from 2021 revealed that 39.9% of adults receiving psychotherapy had one or more teletherapy visits.
Teletherapy use was higher among younger adults, women, college-educated individuals, those with higher incomes, those with private insurance, and those who lived in urban areas.
The authors noted that while teletherapy is intended to remove transportation and time barriers and was widely adopted during the pandemic, the findings show that those who were older, less educated, and with lower incomes were less likely to use it.
Notably, urban residents were more than twice as likely to use teletherapy than rural residents. Prior to the COVID-19 pandemic, teletherapy was viewed as a potential solution for individuals living in rural areas facing a shortage of mental health professionals, but study results showed that “teletherapy does not appear to have addressed this public health challenge,” the investigators wrote.
“The trends we are seeing underscore the need for targeted interventions and health policies that expand psychotherapy access to underserved groups,” said Olfson.
“Ensuring that individuals in psychological distress can access care is a national priority. Addressing technical and financial barriers to teletherapy could help bridge the gap in access and promote equity in mental health care,” he added.
Study limitations included a possible underreporting of psychotherapy use by participants. In addition, MEPS does not include nursing home residents, incarcerated, and unhoused individuals.
Study funding was not disclosed. Olfson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM JAMA PSYCHIATRY
Hoarding Disorder: A Looming National Crisis?
A report published in July 2024 by the US Senate Special Committee on Aging is calling for a national coordinated response to what the authors claim may be an emerging hoarding disorder (HD) crisis.
While millions of US adults are estimated to have HD, it is the disorder’s prevalence and severity among older adults that sounded the alarm for the Committee Chair Sen. Bob Casey (D-PA).
the report stated. Older adults made up about 16% of the US population in 2019. By 2060, that proportion is projected to soar to 25%.
The country’s aging population alone “could fuel a rise in hoarding in the coming decades,” the report authors noted.
These findings underscore the pressing need for a deeper understanding of HD, particularly as reports of its impact continue to rise. The Senate report also raises critical questions about the nature of HD: What is known about the condition? What evidence-based treatments are currently available, and are there national strategies that will prevent it from becoming a systemic crisis?
Why the Urgency?
An increase in anecdotal reports of HD in his home state prompted Casey, chair of the Senate Committee on Aging, to launch the investigation into the incidence and consequences of HD. Soon after the committee began its work, it became evident that the problem was not unique to communities in Pennsylvania. It was a nationwide issue.
“Communities throughout the United States are already grappling with HD,” the report noted.
HD is characterized by persistent difficulty discarding possessions, regardless of their monetary value. For individuals with HD, such items frequently hold meaningful reminders of past events and provide a sense of security. Difficulties with emotional regulation, executive functioning, and impulse control all contribute to the excessive buildup of clutter. Problems with attention, organization, and problem-solving are also common.
As individuals with HD age, physical limitations or disabilities may hinder their ability to discard clutter. As the accumulation increases, it can pose serious risks not only to their safety but also to public health.
Dozens of statements submitted to the Senate committee by those with HD, clinicians and social workers, first responders, social service organizations, state and federal agencies, and professional societies paint a concerning picture about the impact of hoarding on emergency and community services.
Data from the National Fire Incident Reporting System show the number of hoarding-related residential structural fires increased 26% between 2014 and 2022. Some 5242 residential fires connected to cluttered environments during that time resulted in 1367 fire service injuries, 1119 civilian injuries, and over $396 million in damages.
“For older adults, those consequences include health and safety risks, social isolation, eviction, and homelessness,” the report authors noted. “For communities, those consequences include public health concerns, increased risk of fire, and dangers to emergency responders.”
What Causes HD?
HD was once classified as a symptom of obsessive-compulsive personality disorder, with extreme causes meeting the diagnostic criteria for obsessive-compulsive disorder. That changed in 2010 when a working group recommended that HD be added to the Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition, as a stand-alone disorder. That recommendation was approved in 2012.
However, a decade later, much about HD’s etiology remains unknown.
Often beginning in early adolescence, HD is a chronic and progressive condition, with genetics and trauma playing a role in its onset and course, Sanjaya Saxena, MD, director of Clinical and Research Affairs at the International OCD Foundation, said in an interview.
Between 50% and 85% of people with HD symptoms have family members with similar behavior. HD is often comorbid with other psychiatric and medical disorders, which can complicate treatment.
Results of a 2022 study showed that, compared with healthy control individuals, people with HD had widespread abnormalities in the prefrontal white matter tract which connects cortical regions involved in executive functioning, including working memory, attention, reward processing, and decision-making.
Some research also suggests that dysregulation of serotonin transmission may contribute to compulsive behaviors and the difficulty in letting go of possessions.
“We do know that there are factors that contribute to worsening of hoarding symptoms, but that’s not the same thing as what really causes it. So unfortunately, it’s still very understudied, and we don’t have great knowledge of what causes it,” Saxena said.
What Treatments Are Available?
There are currently no Food and Drug Administration–approved medications to treat HD, although some research has shown antidepressants paroxetine and venlafaxine may have some benefit. Methylphenidate and atomoxetine are also under study for HD.
Nonpharmacological therapies have shown more promising results. Among the first was a specialized cognitive-behavioral therapy (CBT) program developed by Randy Frost, PhD, professor emeritus of psychology at Smith College in Northampton, Massachusetts, and Gail Steketee, PhD, dean emerita and professor emerita of social work at Boston University in Massachusetts.
First published in 2007 and the subject of many clinical trials and studies since, the 26-session program has served as a model for psychosocial treatments for HD. The evidence-based therapy addresses various symptoms, including impulse control. One module encourages participants to develop a set of questions to consider before acquiring new items, gradually helping them build resistance to the urge to accumulate more possessions, said Frost, whose early work on HD was cited by those who supported adding the condition to the DSM in 2012.
“There are several features that I think are important including exercises in resisting acquiring and processing information when making decisions about discarding,” Frost said in an interview.
A number of studies have demonstrated the efficacy of CBT for HD, including a 2015 meta-analysis coauthored by Frost. The research showed symptom severity decreased significantly following CBT, with the largest gains in difficulty discarding and moderate improvements in clutter and acquiring.
Responses were better among women and younger patients, and although symptoms improved, posttreatment scores remained closer to the clinical range, researchers noted. It’s possible that more intervention beyond what is usually included in clinical trials — such as more sessions or adding home decluttering visits — could improve treatment response, they added.
A workshop based on the specialized CBT program has expanded the reach of the treatment. The group therapy project, Buried in Treasures (BiT), was developed by Frost, Steketee, and David Tolin, PhD, founder and director of the Anxiety Disorders Center at the Institute of Living, Hartford, and an adjunct professor of psychiatry at Yale School of Medicine, New Haven, Connecticut. The workshop is designed as a facilitated treatment that can be delivered by clinicians or trained nonclinician facilitators.
A study published in May found that more than half the participants with HD responded to the treatment, and of those, 39% reported significant reductions in HD symptoms. BiT sessions were led by trained facilitators, and the study included in-home decluttering sessions, also led by trained volunteers. Researchers said adding the home intervention could increase engagement with the group therapy.
Another study of a modified version of BiT found a 32% decrease in HD symptoms after 15 weeks of treatment delivered via video teleconference.
“The BiT workshop has been expanding around the world and has the advantage of being relatively inexpensive,” Frost said. Another advantage is that it can be run by nonclinicians, which expands treatment options in areas where mental health professionals trained to treat HD are in short supply.
However, the workshop “is not perfect, and clients usually still have symptoms at the end of the workshop,” Frost noted.
“The point is that the BiT workshop is the first step in changing a lifestyle related to possessions,” he continued. “We do certainly need to train more people in how to treat hoarding, and we need to facilitate research to make our treatments more effective.”
What’s New in the Field?
One novel program currently under study combines CBT with a cognitive rehabilitation protocol. Called Cognitive Rehabilitation and Exposure/Sorting Therapy (CREST), the program has been shown to help older adults with HD who don’t respond to traditional CBT for HD.
The program, led by Catherine Ayers, PhD, professor of clinical psychiatry at University of California, San Diego, involves memory training and problem-solving combined with exposure therapy to help participants learn how to tolerate distress associated with discarding their possessions.
Early findings pointed to symptom improvement in older adults following 24 sessions with CREST. The program fared better than geriatric case management in a 2018 study — the first randomized controlled trial of a treatment for HD in older adults — and offered additional benefits compared with exposure therapy in a study published in February 2024.
Virtual reality is also helping people with HD. A program developed at Stanford University in California, allows people with HD to work with a therapist as they practice decluttering in a three-dimensional virtual environment created using photographs and videos of actual hoarded objects and cluttered rooms in patients’ homes.
In a small pilot study, nine people older than 55 years with HD attended 16 weeks of online facilitated therapy where they learned to better understand their attachment to those items. They practiced decluttering by selecting virtual items for recycling, donation, or trash. A virtual garbage truck even hauled away the items they had placed in the trash.
Participants were then asked to discard the actual items at home. Most participants reported a decrease in hoarding symptoms, which was confirmed following a home assessment by a clinician.
“When you pick up an object from a loved one, it still maybe has the scent of the loved one. It has these tactile cues, colors. But in the virtual world, you can take a little bit of a step back,” lead researchers Carolyn Rodriguez, MD, PhD, director of Stanford’s Hoarding Disorders Research Program, said in an interview.
“It’s a little ramp to help people practice these skills. And then what we find is that it actually translated really well. They were able to go home and actually do the real uncluttering,” Rodriguez added.
What Else Can Be Done?
While researchers like Rodriguez continue studies of new and existing treatments, the Senate report draws attention to other responses that could aid people with HD. Because of its significant impact on emergency responders, adult protective services, aging services, and housing providers, the report recommends a nationwide response to older adults with HD.
Currently, federal agencies in charge of mental and community health are not doing enough to address HD, the report’s authors noted.
The report demonstrates “the scope and severity of these challenges and offers a path forward for how we can help people, communities, and local governments contend with this condition,” Casey said.
Specifically, the document cites a lack of HD services and tracking by the Substance Abuse and Mental Health Services Administration, the Administration for Community Living, and the Centers for Disease Control and Prevention.
The committee recommended these agencies collaborate to improve HD data collection, which will be critical to managing a potential spike in cases as the population ages. The committee also suggested awareness and training campaigns to better educate clinicians, social service providers, court officials, and first responders about HD.
Further, the report’s authors called for the Department of Housing and Urban Development to provide guidance and technical assistance on HD for landlords and housing assistance programs and urged Congress to collaborate with the Centers for Medicare & Medicaid Services to expand coverage for hoarding treatments.
Finally, the committee encouraged policymakers to engage directly with individuals affected by HD and their families to better understand the impact of the disorder and inform policy development.
“I think the Senate report focuses on education, not just for therapists, but other stakeholders too,” Frost said. “There are lots of other professionals who have a stake in this process, housing specialists, elder service folks, health and human services. Awareness of this problem is something that’s important for them as well.”
Rodriguez characterized the report’s recommendations as “potentially lifesaving” for individuals with HD. She added that it represents the first step in an ongoing effort to address an impending public health crisis related to HD in older adults and its broader impact on communities.
A spokesperson with Casey’s office said it’s unclear whether any federal agencies have acted on the report recommendations since it was released in June. It’s also unknown whether the Senate Committee on Aging will pursue any additional work on HD when new committee leaders are appointed in 2025.
“Although some federal agencies have taken steps to address HD, those steps are frequently limited. Other relevant agencies have not addressed HD at all in recent years,” report authors wrote. “The federal government can, and should, do more to bolster the response to HD.”
Frost agreed.
“I think federal agencies can have a positive effect by promoting, supporting, and tracking local efforts in dealing with this problem,” he said.
With reporting from Eve Bender.
A version of this article appeared on Medscape.com.
A report published in July 2024 by the US Senate Special Committee on Aging is calling for a national coordinated response to what the authors claim may be an emerging hoarding disorder (HD) crisis.
While millions of US adults are estimated to have HD, it is the disorder’s prevalence and severity among older adults that sounded the alarm for the Committee Chair Sen. Bob Casey (D-PA).
the report stated. Older adults made up about 16% of the US population in 2019. By 2060, that proportion is projected to soar to 25%.
The country’s aging population alone “could fuel a rise in hoarding in the coming decades,” the report authors noted.
These findings underscore the pressing need for a deeper understanding of HD, particularly as reports of its impact continue to rise. The Senate report also raises critical questions about the nature of HD: What is known about the condition? What evidence-based treatments are currently available, and are there national strategies that will prevent it from becoming a systemic crisis?
Why the Urgency?
An increase in anecdotal reports of HD in his home state prompted Casey, chair of the Senate Committee on Aging, to launch the investigation into the incidence and consequences of HD. Soon after the committee began its work, it became evident that the problem was not unique to communities in Pennsylvania. It was a nationwide issue.
“Communities throughout the United States are already grappling with HD,” the report noted.
HD is characterized by persistent difficulty discarding possessions, regardless of their monetary value. For individuals with HD, such items frequently hold meaningful reminders of past events and provide a sense of security. Difficulties with emotional regulation, executive functioning, and impulse control all contribute to the excessive buildup of clutter. Problems with attention, organization, and problem-solving are also common.
As individuals with HD age, physical limitations or disabilities may hinder their ability to discard clutter. As the accumulation increases, it can pose serious risks not only to their safety but also to public health.
Dozens of statements submitted to the Senate committee by those with HD, clinicians and social workers, first responders, social service organizations, state and federal agencies, and professional societies paint a concerning picture about the impact of hoarding on emergency and community services.
Data from the National Fire Incident Reporting System show the number of hoarding-related residential structural fires increased 26% between 2014 and 2022. Some 5242 residential fires connected to cluttered environments during that time resulted in 1367 fire service injuries, 1119 civilian injuries, and over $396 million in damages.
“For older adults, those consequences include health and safety risks, social isolation, eviction, and homelessness,” the report authors noted. “For communities, those consequences include public health concerns, increased risk of fire, and dangers to emergency responders.”
What Causes HD?
HD was once classified as a symptom of obsessive-compulsive personality disorder, with extreme causes meeting the diagnostic criteria for obsessive-compulsive disorder. That changed in 2010 when a working group recommended that HD be added to the Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition, as a stand-alone disorder. That recommendation was approved in 2012.
However, a decade later, much about HD’s etiology remains unknown.
Often beginning in early adolescence, HD is a chronic and progressive condition, with genetics and trauma playing a role in its onset and course, Sanjaya Saxena, MD, director of Clinical and Research Affairs at the International OCD Foundation, said in an interview.
Between 50% and 85% of people with HD symptoms have family members with similar behavior. HD is often comorbid with other psychiatric and medical disorders, which can complicate treatment.
Results of a 2022 study showed that, compared with healthy control individuals, people with HD had widespread abnormalities in the prefrontal white matter tract which connects cortical regions involved in executive functioning, including working memory, attention, reward processing, and decision-making.
Some research also suggests that dysregulation of serotonin transmission may contribute to compulsive behaviors and the difficulty in letting go of possessions.
“We do know that there are factors that contribute to worsening of hoarding symptoms, but that’s not the same thing as what really causes it. So unfortunately, it’s still very understudied, and we don’t have great knowledge of what causes it,” Saxena said.
What Treatments Are Available?
There are currently no Food and Drug Administration–approved medications to treat HD, although some research has shown antidepressants paroxetine and venlafaxine may have some benefit. Methylphenidate and atomoxetine are also under study for HD.
Nonpharmacological therapies have shown more promising results. Among the first was a specialized cognitive-behavioral therapy (CBT) program developed by Randy Frost, PhD, professor emeritus of psychology at Smith College in Northampton, Massachusetts, and Gail Steketee, PhD, dean emerita and professor emerita of social work at Boston University in Massachusetts.
First published in 2007 and the subject of many clinical trials and studies since, the 26-session program has served as a model for psychosocial treatments for HD. The evidence-based therapy addresses various symptoms, including impulse control. One module encourages participants to develop a set of questions to consider before acquiring new items, gradually helping them build resistance to the urge to accumulate more possessions, said Frost, whose early work on HD was cited by those who supported adding the condition to the DSM in 2012.
“There are several features that I think are important including exercises in resisting acquiring and processing information when making decisions about discarding,” Frost said in an interview.
A number of studies have demonstrated the efficacy of CBT for HD, including a 2015 meta-analysis coauthored by Frost. The research showed symptom severity decreased significantly following CBT, with the largest gains in difficulty discarding and moderate improvements in clutter and acquiring.
Responses were better among women and younger patients, and although symptoms improved, posttreatment scores remained closer to the clinical range, researchers noted. It’s possible that more intervention beyond what is usually included in clinical trials — such as more sessions or adding home decluttering visits — could improve treatment response, they added.
A workshop based on the specialized CBT program has expanded the reach of the treatment. The group therapy project, Buried in Treasures (BiT), was developed by Frost, Steketee, and David Tolin, PhD, founder and director of the Anxiety Disorders Center at the Institute of Living, Hartford, and an adjunct professor of psychiatry at Yale School of Medicine, New Haven, Connecticut. The workshop is designed as a facilitated treatment that can be delivered by clinicians or trained nonclinician facilitators.
A study published in May found that more than half the participants with HD responded to the treatment, and of those, 39% reported significant reductions in HD symptoms. BiT sessions were led by trained facilitators, and the study included in-home decluttering sessions, also led by trained volunteers. Researchers said adding the home intervention could increase engagement with the group therapy.
Another study of a modified version of BiT found a 32% decrease in HD symptoms after 15 weeks of treatment delivered via video teleconference.
“The BiT workshop has been expanding around the world and has the advantage of being relatively inexpensive,” Frost said. Another advantage is that it can be run by nonclinicians, which expands treatment options in areas where mental health professionals trained to treat HD are in short supply.
However, the workshop “is not perfect, and clients usually still have symptoms at the end of the workshop,” Frost noted.
“The point is that the BiT workshop is the first step in changing a lifestyle related to possessions,” he continued. “We do certainly need to train more people in how to treat hoarding, and we need to facilitate research to make our treatments more effective.”
What’s New in the Field?
One novel program currently under study combines CBT with a cognitive rehabilitation protocol. Called Cognitive Rehabilitation and Exposure/Sorting Therapy (CREST), the program has been shown to help older adults with HD who don’t respond to traditional CBT for HD.
The program, led by Catherine Ayers, PhD, professor of clinical psychiatry at University of California, San Diego, involves memory training and problem-solving combined with exposure therapy to help participants learn how to tolerate distress associated with discarding their possessions.
Early findings pointed to symptom improvement in older adults following 24 sessions with CREST. The program fared better than geriatric case management in a 2018 study — the first randomized controlled trial of a treatment for HD in older adults — and offered additional benefits compared with exposure therapy in a study published in February 2024.
Virtual reality is also helping people with HD. A program developed at Stanford University in California, allows people with HD to work with a therapist as they practice decluttering in a three-dimensional virtual environment created using photographs and videos of actual hoarded objects and cluttered rooms in patients’ homes.
In a small pilot study, nine people older than 55 years with HD attended 16 weeks of online facilitated therapy where they learned to better understand their attachment to those items. They practiced decluttering by selecting virtual items for recycling, donation, or trash. A virtual garbage truck even hauled away the items they had placed in the trash.
Participants were then asked to discard the actual items at home. Most participants reported a decrease in hoarding symptoms, which was confirmed following a home assessment by a clinician.
“When you pick up an object from a loved one, it still maybe has the scent of the loved one. It has these tactile cues, colors. But in the virtual world, you can take a little bit of a step back,” lead researchers Carolyn Rodriguez, MD, PhD, director of Stanford’s Hoarding Disorders Research Program, said in an interview.
“It’s a little ramp to help people practice these skills. And then what we find is that it actually translated really well. They were able to go home and actually do the real uncluttering,” Rodriguez added.
What Else Can Be Done?
While researchers like Rodriguez continue studies of new and existing treatments, the Senate report draws attention to other responses that could aid people with HD. Because of its significant impact on emergency responders, adult protective services, aging services, and housing providers, the report recommends a nationwide response to older adults with HD.
Currently, federal agencies in charge of mental and community health are not doing enough to address HD, the report’s authors noted.
The report demonstrates “the scope and severity of these challenges and offers a path forward for how we can help people, communities, and local governments contend with this condition,” Casey said.
Specifically, the document cites a lack of HD services and tracking by the Substance Abuse and Mental Health Services Administration, the Administration for Community Living, and the Centers for Disease Control and Prevention.
The committee recommended these agencies collaborate to improve HD data collection, which will be critical to managing a potential spike in cases as the population ages. The committee also suggested awareness and training campaigns to better educate clinicians, social service providers, court officials, and first responders about HD.
Further, the report’s authors called for the Department of Housing and Urban Development to provide guidance and technical assistance on HD for landlords and housing assistance programs and urged Congress to collaborate with the Centers for Medicare & Medicaid Services to expand coverage for hoarding treatments.
Finally, the committee encouraged policymakers to engage directly with individuals affected by HD and their families to better understand the impact of the disorder and inform policy development.
“I think the Senate report focuses on education, not just for therapists, but other stakeholders too,” Frost said. “There are lots of other professionals who have a stake in this process, housing specialists, elder service folks, health and human services. Awareness of this problem is something that’s important for them as well.”
Rodriguez characterized the report’s recommendations as “potentially lifesaving” for individuals with HD. She added that it represents the first step in an ongoing effort to address an impending public health crisis related to HD in older adults and its broader impact on communities.
A spokesperson with Casey’s office said it’s unclear whether any federal agencies have acted on the report recommendations since it was released in June. It’s also unknown whether the Senate Committee on Aging will pursue any additional work on HD when new committee leaders are appointed in 2025.
“Although some federal agencies have taken steps to address HD, those steps are frequently limited. Other relevant agencies have not addressed HD at all in recent years,” report authors wrote. “The federal government can, and should, do more to bolster the response to HD.”
Frost agreed.
“I think federal agencies can have a positive effect by promoting, supporting, and tracking local efforts in dealing with this problem,” he said.
With reporting from Eve Bender.
A version of this article appeared on Medscape.com.
A report published in July 2024 by the US Senate Special Committee on Aging is calling for a national coordinated response to what the authors claim may be an emerging hoarding disorder (HD) crisis.
While millions of US adults are estimated to have HD, it is the disorder’s prevalence and severity among older adults that sounded the alarm for the Committee Chair Sen. Bob Casey (D-PA).
the report stated. Older adults made up about 16% of the US population in 2019. By 2060, that proportion is projected to soar to 25%.
The country’s aging population alone “could fuel a rise in hoarding in the coming decades,” the report authors noted.
These findings underscore the pressing need for a deeper understanding of HD, particularly as reports of its impact continue to rise. The Senate report also raises critical questions about the nature of HD: What is known about the condition? What evidence-based treatments are currently available, and are there national strategies that will prevent it from becoming a systemic crisis?
Why the Urgency?
An increase in anecdotal reports of HD in his home state prompted Casey, chair of the Senate Committee on Aging, to launch the investigation into the incidence and consequences of HD. Soon after the committee began its work, it became evident that the problem was not unique to communities in Pennsylvania. It was a nationwide issue.
“Communities throughout the United States are already grappling with HD,” the report noted.
HD is characterized by persistent difficulty discarding possessions, regardless of their monetary value. For individuals with HD, such items frequently hold meaningful reminders of past events and provide a sense of security. Difficulties with emotional regulation, executive functioning, and impulse control all contribute to the excessive buildup of clutter. Problems with attention, organization, and problem-solving are also common.
As individuals with HD age, physical limitations or disabilities may hinder their ability to discard clutter. As the accumulation increases, it can pose serious risks not only to their safety but also to public health.
Dozens of statements submitted to the Senate committee by those with HD, clinicians and social workers, first responders, social service organizations, state and federal agencies, and professional societies paint a concerning picture about the impact of hoarding on emergency and community services.
Data from the National Fire Incident Reporting System show the number of hoarding-related residential structural fires increased 26% between 2014 and 2022. Some 5242 residential fires connected to cluttered environments during that time resulted in 1367 fire service injuries, 1119 civilian injuries, and over $396 million in damages.
“For older adults, those consequences include health and safety risks, social isolation, eviction, and homelessness,” the report authors noted. “For communities, those consequences include public health concerns, increased risk of fire, and dangers to emergency responders.”
What Causes HD?
HD was once classified as a symptom of obsessive-compulsive personality disorder, with extreme causes meeting the diagnostic criteria for obsessive-compulsive disorder. That changed in 2010 when a working group recommended that HD be added to the Diagnostic and Statistical Manual of Mental Disorders (DSM), Fifth Edition, as a stand-alone disorder. That recommendation was approved in 2012.
However, a decade later, much about HD’s etiology remains unknown.
Often beginning in early adolescence, HD is a chronic and progressive condition, with genetics and trauma playing a role in its onset and course, Sanjaya Saxena, MD, director of Clinical and Research Affairs at the International OCD Foundation, said in an interview.
Between 50% and 85% of people with HD symptoms have family members with similar behavior. HD is often comorbid with other psychiatric and medical disorders, which can complicate treatment.
Results of a 2022 study showed that, compared with healthy control individuals, people with HD had widespread abnormalities in the prefrontal white matter tract which connects cortical regions involved in executive functioning, including working memory, attention, reward processing, and decision-making.
Some research also suggests that dysregulation of serotonin transmission may contribute to compulsive behaviors and the difficulty in letting go of possessions.
“We do know that there are factors that contribute to worsening of hoarding symptoms, but that’s not the same thing as what really causes it. So unfortunately, it’s still very understudied, and we don’t have great knowledge of what causes it,” Saxena said.
What Treatments Are Available?
There are currently no Food and Drug Administration–approved medications to treat HD, although some research has shown antidepressants paroxetine and venlafaxine may have some benefit. Methylphenidate and atomoxetine are also under study for HD.
Nonpharmacological therapies have shown more promising results. Among the first was a specialized cognitive-behavioral therapy (CBT) program developed by Randy Frost, PhD, professor emeritus of psychology at Smith College in Northampton, Massachusetts, and Gail Steketee, PhD, dean emerita and professor emerita of social work at Boston University in Massachusetts.
First published in 2007 and the subject of many clinical trials and studies since, the 26-session program has served as a model for psychosocial treatments for HD. The evidence-based therapy addresses various symptoms, including impulse control. One module encourages participants to develop a set of questions to consider before acquiring new items, gradually helping them build resistance to the urge to accumulate more possessions, said Frost, whose early work on HD was cited by those who supported adding the condition to the DSM in 2012.
“There are several features that I think are important including exercises in resisting acquiring and processing information when making decisions about discarding,” Frost said in an interview.
A number of studies have demonstrated the efficacy of CBT for HD, including a 2015 meta-analysis coauthored by Frost. The research showed symptom severity decreased significantly following CBT, with the largest gains in difficulty discarding and moderate improvements in clutter and acquiring.
Responses were better among women and younger patients, and although symptoms improved, posttreatment scores remained closer to the clinical range, researchers noted. It’s possible that more intervention beyond what is usually included in clinical trials — such as more sessions or adding home decluttering visits — could improve treatment response, they added.
A workshop based on the specialized CBT program has expanded the reach of the treatment. The group therapy project, Buried in Treasures (BiT), was developed by Frost, Steketee, and David Tolin, PhD, founder and director of the Anxiety Disorders Center at the Institute of Living, Hartford, and an adjunct professor of psychiatry at Yale School of Medicine, New Haven, Connecticut. The workshop is designed as a facilitated treatment that can be delivered by clinicians or trained nonclinician facilitators.
A study published in May found that more than half the participants with HD responded to the treatment, and of those, 39% reported significant reductions in HD symptoms. BiT sessions were led by trained facilitators, and the study included in-home decluttering sessions, also led by trained volunteers. Researchers said adding the home intervention could increase engagement with the group therapy.
Another study of a modified version of BiT found a 32% decrease in HD symptoms after 15 weeks of treatment delivered via video teleconference.
“The BiT workshop has been expanding around the world and has the advantage of being relatively inexpensive,” Frost said. Another advantage is that it can be run by nonclinicians, which expands treatment options in areas where mental health professionals trained to treat HD are in short supply.
However, the workshop “is not perfect, and clients usually still have symptoms at the end of the workshop,” Frost noted.
“The point is that the BiT workshop is the first step in changing a lifestyle related to possessions,” he continued. “We do certainly need to train more people in how to treat hoarding, and we need to facilitate research to make our treatments more effective.”
What’s New in the Field?
One novel program currently under study combines CBT with a cognitive rehabilitation protocol. Called Cognitive Rehabilitation and Exposure/Sorting Therapy (CREST), the program has been shown to help older adults with HD who don’t respond to traditional CBT for HD.
The program, led by Catherine Ayers, PhD, professor of clinical psychiatry at University of California, San Diego, involves memory training and problem-solving combined with exposure therapy to help participants learn how to tolerate distress associated with discarding their possessions.
Early findings pointed to symptom improvement in older adults following 24 sessions with CREST. The program fared better than geriatric case management in a 2018 study — the first randomized controlled trial of a treatment for HD in older adults — and offered additional benefits compared with exposure therapy in a study published in February 2024.
Virtual reality is also helping people with HD. A program developed at Stanford University in California, allows people with HD to work with a therapist as they practice decluttering in a three-dimensional virtual environment created using photographs and videos of actual hoarded objects and cluttered rooms in patients’ homes.
In a small pilot study, nine people older than 55 years with HD attended 16 weeks of online facilitated therapy where they learned to better understand their attachment to those items. They practiced decluttering by selecting virtual items for recycling, donation, or trash. A virtual garbage truck even hauled away the items they had placed in the trash.
Participants were then asked to discard the actual items at home. Most participants reported a decrease in hoarding symptoms, which was confirmed following a home assessment by a clinician.
“When you pick up an object from a loved one, it still maybe has the scent of the loved one. It has these tactile cues, colors. But in the virtual world, you can take a little bit of a step back,” lead researchers Carolyn Rodriguez, MD, PhD, director of Stanford’s Hoarding Disorders Research Program, said in an interview.
“It’s a little ramp to help people practice these skills. And then what we find is that it actually translated really well. They were able to go home and actually do the real uncluttering,” Rodriguez added.
What Else Can Be Done?
While researchers like Rodriguez continue studies of new and existing treatments, the Senate report draws attention to other responses that could aid people with HD. Because of its significant impact on emergency responders, adult protective services, aging services, and housing providers, the report recommends a nationwide response to older adults with HD.
Currently, federal agencies in charge of mental and community health are not doing enough to address HD, the report’s authors noted.
The report demonstrates “the scope and severity of these challenges and offers a path forward for how we can help people, communities, and local governments contend with this condition,” Casey said.
Specifically, the document cites a lack of HD services and tracking by the Substance Abuse and Mental Health Services Administration, the Administration for Community Living, and the Centers for Disease Control and Prevention.
The committee recommended these agencies collaborate to improve HD data collection, which will be critical to managing a potential spike in cases as the population ages. The committee also suggested awareness and training campaigns to better educate clinicians, social service providers, court officials, and first responders about HD.
Further, the report’s authors called for the Department of Housing and Urban Development to provide guidance and technical assistance on HD for landlords and housing assistance programs and urged Congress to collaborate with the Centers for Medicare & Medicaid Services to expand coverage for hoarding treatments.
Finally, the committee encouraged policymakers to engage directly with individuals affected by HD and their families to better understand the impact of the disorder and inform policy development.
“I think the Senate report focuses on education, not just for therapists, but other stakeholders too,” Frost said. “There are lots of other professionals who have a stake in this process, housing specialists, elder service folks, health and human services. Awareness of this problem is something that’s important for them as well.”
Rodriguez characterized the report’s recommendations as “potentially lifesaving” for individuals with HD. She added that it represents the first step in an ongoing effort to address an impending public health crisis related to HD in older adults and its broader impact on communities.
A spokesperson with Casey’s office said it’s unclear whether any federal agencies have acted on the report recommendations since it was released in June. It’s also unknown whether the Senate Committee on Aging will pursue any additional work on HD when new committee leaders are appointed in 2025.
“Although some federal agencies have taken steps to address HD, those steps are frequently limited. Other relevant agencies have not addressed HD at all in recent years,” report authors wrote. “The federal government can, and should, do more to bolster the response to HD.”
Frost agreed.
“I think federal agencies can have a positive effect by promoting, supporting, and tracking local efforts in dealing with this problem,” he said.
With reporting from Eve Bender.
A version of this article appeared on Medscape.com.
Veterans’ Well-Being Tools Aim to Improve Quality of Life
Could assessing the well-being of older patients create better treatment plans?
Researchers with the US Department of Veterans Affairs posit that doing so just might improve patient quality of life.
In an article in Medical Care, Dawne Vogt, PhD, and her colleagues described two surveys of well-being developed for use in clinical settings.
“Well-Being Signs” (WBS), a 1-minute screening, asks patients about how satisfied they are with the most important parts of their daily life, which could include time with family. It also asks how regularly involved they are in the activities and their level of functioning.
“Well-Being Brief” (WBB) is self-administered and asks more in-depth questions about finances, health, social relationships, and vocation. Clinicians can use the tool to make referrals to appropriate services like counseling or resources like senior centers.
“They’re not things that we’ve historically paid a lot of attention to, at least in the healthcare setting,” said Vogt, a research psychologist in the Women’s Health Sciences Division of the VA Boston Healthcare System in Massachusetts. “A growing body of research shows that they have really big implications for health.”
The two approaches stem from an increased awareness of the relationship between social determinants of health and outcomes. Both screenings can be implemented more effectively in a clinical setting than other measures because of their brevity and ease of use, she said.
Vogt shared that anecdotally, she finds patients are pleasantly surprised by the questionnaires “because they’re being seen in a way that they don’t always feel like they’re seen.”
Vogt said that the two well-being measurements are more nuanced than standard screenings for depression.
“A measure of depression tells you something much more narrow than a measure of well-being tells you,” she said, adding that identifying problem areas early can help prevent developing mental health disorders. For example, Vogt said that veterans with higher well-being are less likely to develop posttraumatic stress disorder when exposed to trauma.
The WBS has been validated, while the WBB questionnaire awaits final testing.
James Michail, MD, a family and geriatric physician with Providence Health & Services in Los Angeles, California, said he views the well-being screeners as launching points into discussing whether a treatment is enhancing or inhibiting a patient’s life.
“We have screenings for everything else but not for wellness, and the goal of care isn’t necessarily always treatment,” Michail said. “It’s taking the whole person into consideration. There’s a person behind the disease.”
Kendra Segura, MD, an obstetrician-gynecologist in Los Angeles, said she is open to using a well-being screener. Usually, building repertoire with a patient takes time, and sometimes only then can it allow for a more candid assessment of well-being.
“Over the course of several visits, that is when patients open up,” she said. “It’s when that starts to happen where they start to tell you about their well-being. It’s not an easy thing to establish.”
The authors of the article reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Could assessing the well-being of older patients create better treatment plans?
Researchers with the US Department of Veterans Affairs posit that doing so just might improve patient quality of life.
In an article in Medical Care, Dawne Vogt, PhD, and her colleagues described two surveys of well-being developed for use in clinical settings.
“Well-Being Signs” (WBS), a 1-minute screening, asks patients about how satisfied they are with the most important parts of their daily life, which could include time with family. It also asks how regularly involved they are in the activities and their level of functioning.
“Well-Being Brief” (WBB) is self-administered and asks more in-depth questions about finances, health, social relationships, and vocation. Clinicians can use the tool to make referrals to appropriate services like counseling or resources like senior centers.
“They’re not things that we’ve historically paid a lot of attention to, at least in the healthcare setting,” said Vogt, a research psychologist in the Women’s Health Sciences Division of the VA Boston Healthcare System in Massachusetts. “A growing body of research shows that they have really big implications for health.”
The two approaches stem from an increased awareness of the relationship between social determinants of health and outcomes. Both screenings can be implemented more effectively in a clinical setting than other measures because of their brevity and ease of use, she said.
Vogt shared that anecdotally, she finds patients are pleasantly surprised by the questionnaires “because they’re being seen in a way that they don’t always feel like they’re seen.”
Vogt said that the two well-being measurements are more nuanced than standard screenings for depression.
“A measure of depression tells you something much more narrow than a measure of well-being tells you,” she said, adding that identifying problem areas early can help prevent developing mental health disorders. For example, Vogt said that veterans with higher well-being are less likely to develop posttraumatic stress disorder when exposed to trauma.
The WBS has been validated, while the WBB questionnaire awaits final testing.
James Michail, MD, a family and geriatric physician with Providence Health & Services in Los Angeles, California, said he views the well-being screeners as launching points into discussing whether a treatment is enhancing or inhibiting a patient’s life.
“We have screenings for everything else but not for wellness, and the goal of care isn’t necessarily always treatment,” Michail said. “It’s taking the whole person into consideration. There’s a person behind the disease.”
Kendra Segura, MD, an obstetrician-gynecologist in Los Angeles, said she is open to using a well-being screener. Usually, building repertoire with a patient takes time, and sometimes only then can it allow for a more candid assessment of well-being.
“Over the course of several visits, that is when patients open up,” she said. “It’s when that starts to happen where they start to tell you about their well-being. It’s not an easy thing to establish.”
The authors of the article reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Could assessing the well-being of older patients create better treatment plans?
Researchers with the US Department of Veterans Affairs posit that doing so just might improve patient quality of life.
In an article in Medical Care, Dawne Vogt, PhD, and her colleagues described two surveys of well-being developed for use in clinical settings.
“Well-Being Signs” (WBS), a 1-minute screening, asks patients about how satisfied they are with the most important parts of their daily life, which could include time with family. It also asks how regularly involved they are in the activities and their level of functioning.
“Well-Being Brief” (WBB) is self-administered and asks more in-depth questions about finances, health, social relationships, and vocation. Clinicians can use the tool to make referrals to appropriate services like counseling or resources like senior centers.
“They’re not things that we’ve historically paid a lot of attention to, at least in the healthcare setting,” said Vogt, a research psychologist in the Women’s Health Sciences Division of the VA Boston Healthcare System in Massachusetts. “A growing body of research shows that they have really big implications for health.”
The two approaches stem from an increased awareness of the relationship between social determinants of health and outcomes. Both screenings can be implemented more effectively in a clinical setting than other measures because of their brevity and ease of use, she said.
Vogt shared that anecdotally, she finds patients are pleasantly surprised by the questionnaires “because they’re being seen in a way that they don’t always feel like they’re seen.”
Vogt said that the two well-being measurements are more nuanced than standard screenings for depression.
“A measure of depression tells you something much more narrow than a measure of well-being tells you,” she said, adding that identifying problem areas early can help prevent developing mental health disorders. For example, Vogt said that veterans with higher well-being are less likely to develop posttraumatic stress disorder when exposed to trauma.
The WBS has been validated, while the WBB questionnaire awaits final testing.
James Michail, MD, a family and geriatric physician with Providence Health & Services in Los Angeles, California, said he views the well-being screeners as launching points into discussing whether a treatment is enhancing or inhibiting a patient’s life.
“We have screenings for everything else but not for wellness, and the goal of care isn’t necessarily always treatment,” Michail said. “It’s taking the whole person into consideration. There’s a person behind the disease.”
Kendra Segura, MD, an obstetrician-gynecologist in Los Angeles, said she is open to using a well-being screener. Usually, building repertoire with a patient takes time, and sometimes only then can it allow for a more candid assessment of well-being.
“Over the course of several visits, that is when patients open up,” she said. “It’s when that starts to happen where they start to tell you about their well-being. It’s not an easy thing to establish.”
The authors of the article reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MEDICAL CARE