User login
Dramatic improvements reported after surgery for hidradenitis suppurativa
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY.
Key clinical point: Patients with hidradenitis suppurativa (HS) report dramatic improvement after radical surgery.
Major finding: The percentage reporting strong or very strong impairment of private life fell from 77% before surgery to 20% afterward.
Study details: A retrospective survey of 255 patients who had undergone surgery for HS (Hurley stage III).
Disclosures: The study had no funding source. The authors had no conflicts to disclose.
Source: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
A Case of Pustular Psoriasis of Pregnancy With Positive Maternal-Fetal Outcomes
Pustular psoriasis of pregnancy (PPP), also known as impetigo herpetiformis, is a relatively rare cutaneous disorder of pregnancy wherein lesions typically appear in the third trimester and resolve after delivery; however, lesions may persist through the postpartum period. Pustular psoriasis of pregnancy may be considered a fifth dermatosis of pregnancy, alongside the classic dermatoses of atopic eruption of pregnancy, intrahepatic cholestasis of pregnancy, pemphigoid gestationis, and pruritic urticarial papules and plaques of pregnancy.1
As PPP is a rare disease, its effects on maternal-fetal health outcomes and management remain to be elucidated. Though maternal mortality is rare in PPP, it is a unique dermatosis of pregnancy because it may be associated with severe systemic maternal symptoms.2 Fetal morbidity and mortality are less predictable in PPP, with reported cases of stillbirth, fetal anomalies, and neonatal death thought to be due largely to placental insufficiency, even with control of symptoms.1,3 Given the risk of serious harm to the fetus, reporting of cases and discussion of PPP management is critical.
Case Report
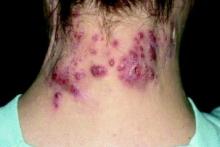
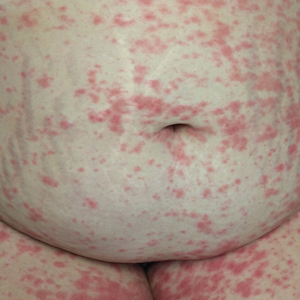
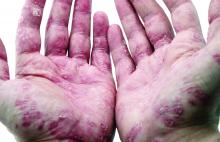
An otherwise healthy 29-year-old G2P1 woman at 32 weeks’ gestation presented to our emergency department with a 1-week history of a pruritic, burning rash that started on the thighs then spread diffusely. She denied any similar rash in her prior pregnancy. She was not currently taking any medications except for prenatal vitamins and denied any systemic symptoms. The patient’s obstetrician initiated treatment with methylprednisolone 50 mg once daily for the rash 3 days prior to the current presentation, which had not seemed to help. On physical examination, edematous pink plaques studded with 1- to 2-mm collarettes of scaling and sparse 1-mm pustules involving the arms, chest, abdomen, back, groin, buttocks, and legs were noted. The plaques on the back and inner thighs had a peripheral rim of desquamative scaling. There were pink macules on the palms, and superficial desquamation was noted on the lips. The oral mucosa was otherwise spared (Figure 1).
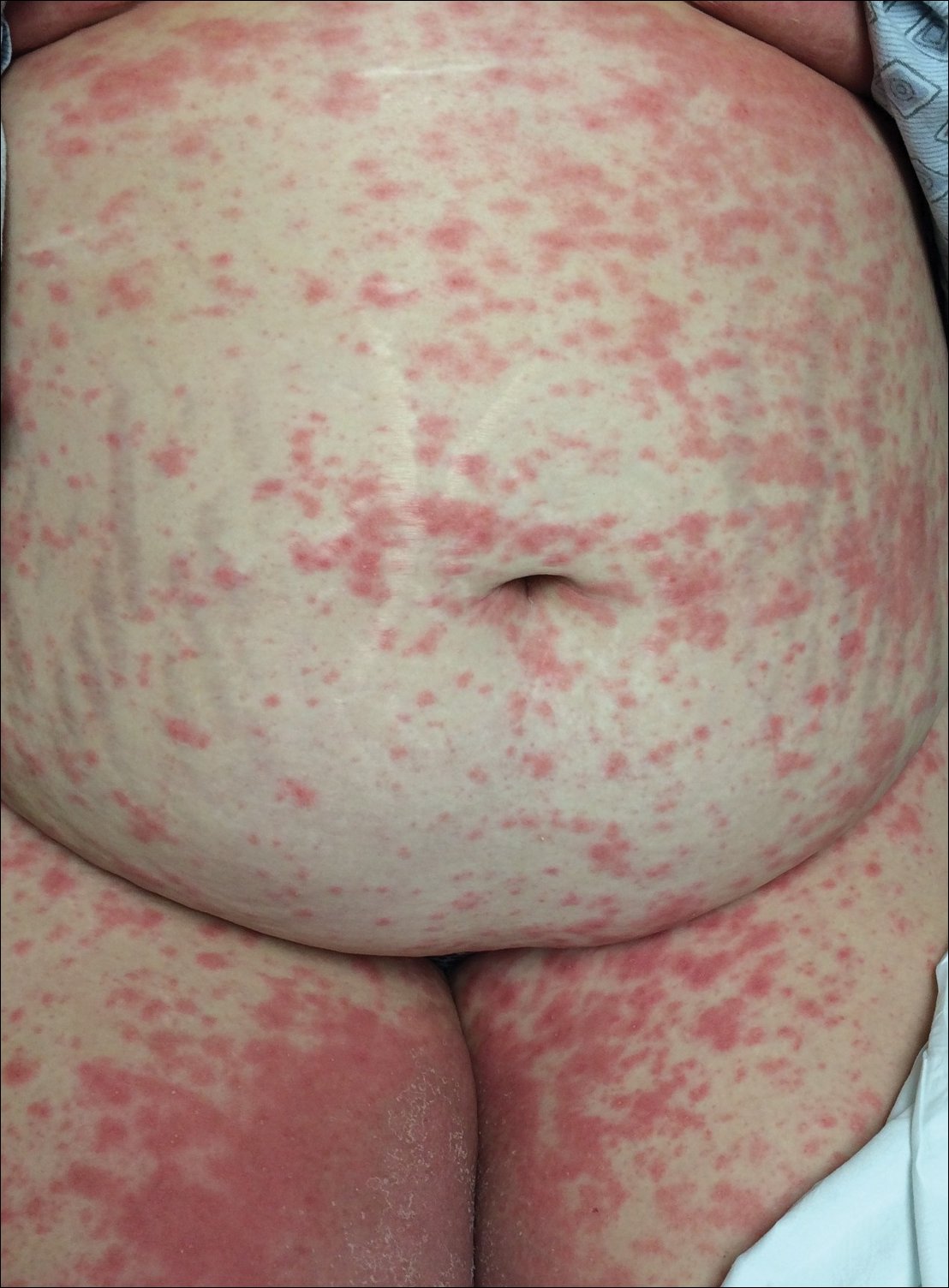
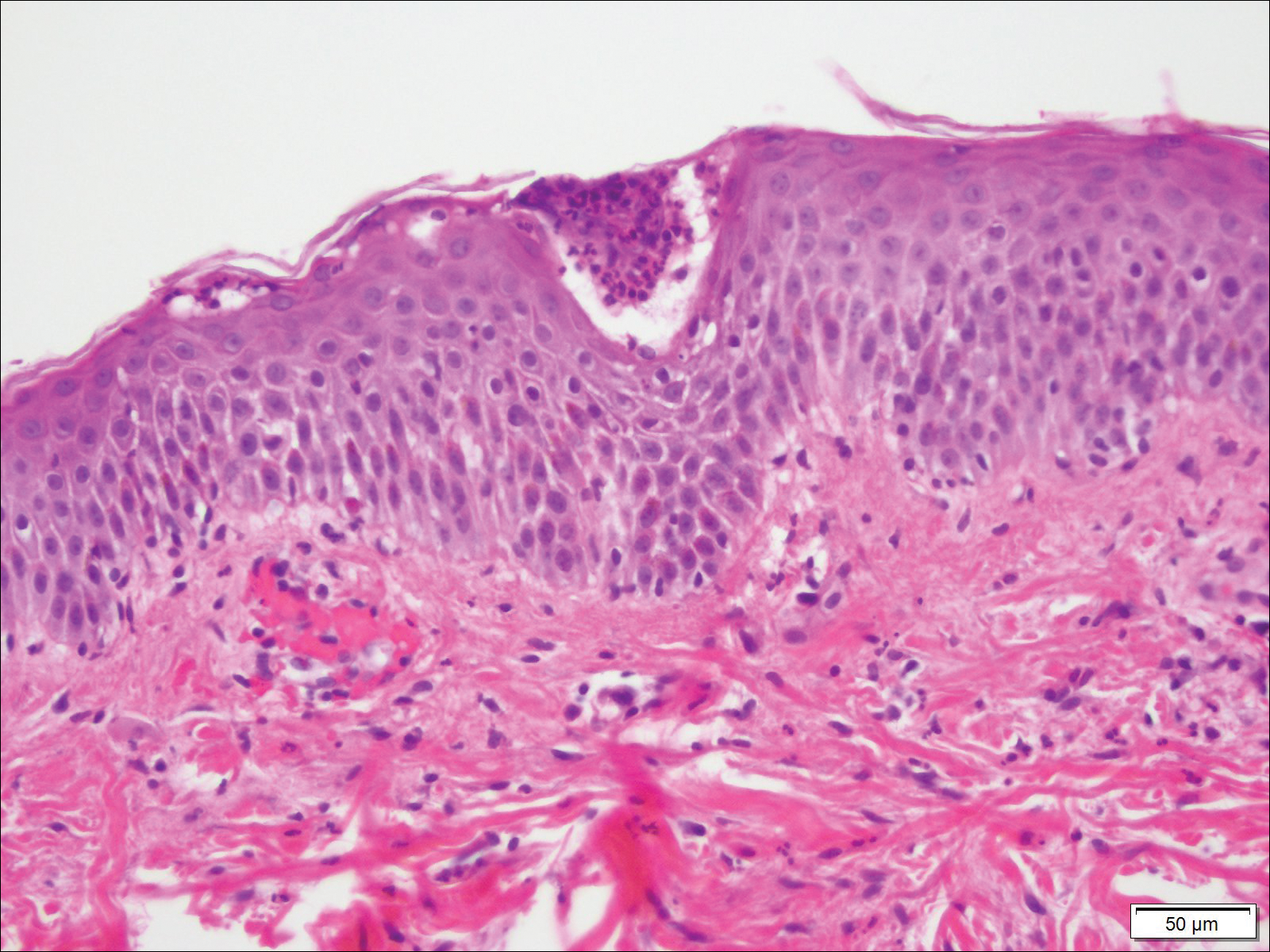
Biopsy specimens from the left arm revealed discrete subcorneal pustules with mild acanthosis of the epidermis with spongiosis (Figure 2). The papillary dermis showed a sparse infiltrate of neutrophils with many marginated neutrophils within vessels. Direct immunofluorescence was negative for human IgG, IgA, IgM, complement component 3, and fibrinogen. Laboratory workup revealed leukocytosis of 21.5×109/L (reference range, 4.5–11.0×109/L) with neutrophilic predominance of 73.6% (reference range, 56%), an elevated erythrocyte sedimentation rate (ESR) of 40 mm/h (reference range, 0–20 mm/h), and a mild hypocalcemia of 8.6 mg/dL (reference range, 8.2–10.2 mg/dL). The patient was started on methylprednisone 40 mg once daily with a plan to taper the dose by 8 mg every 5 days.
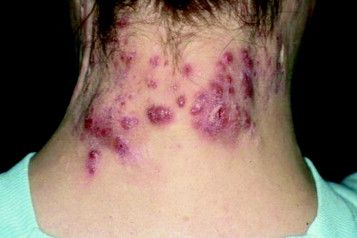
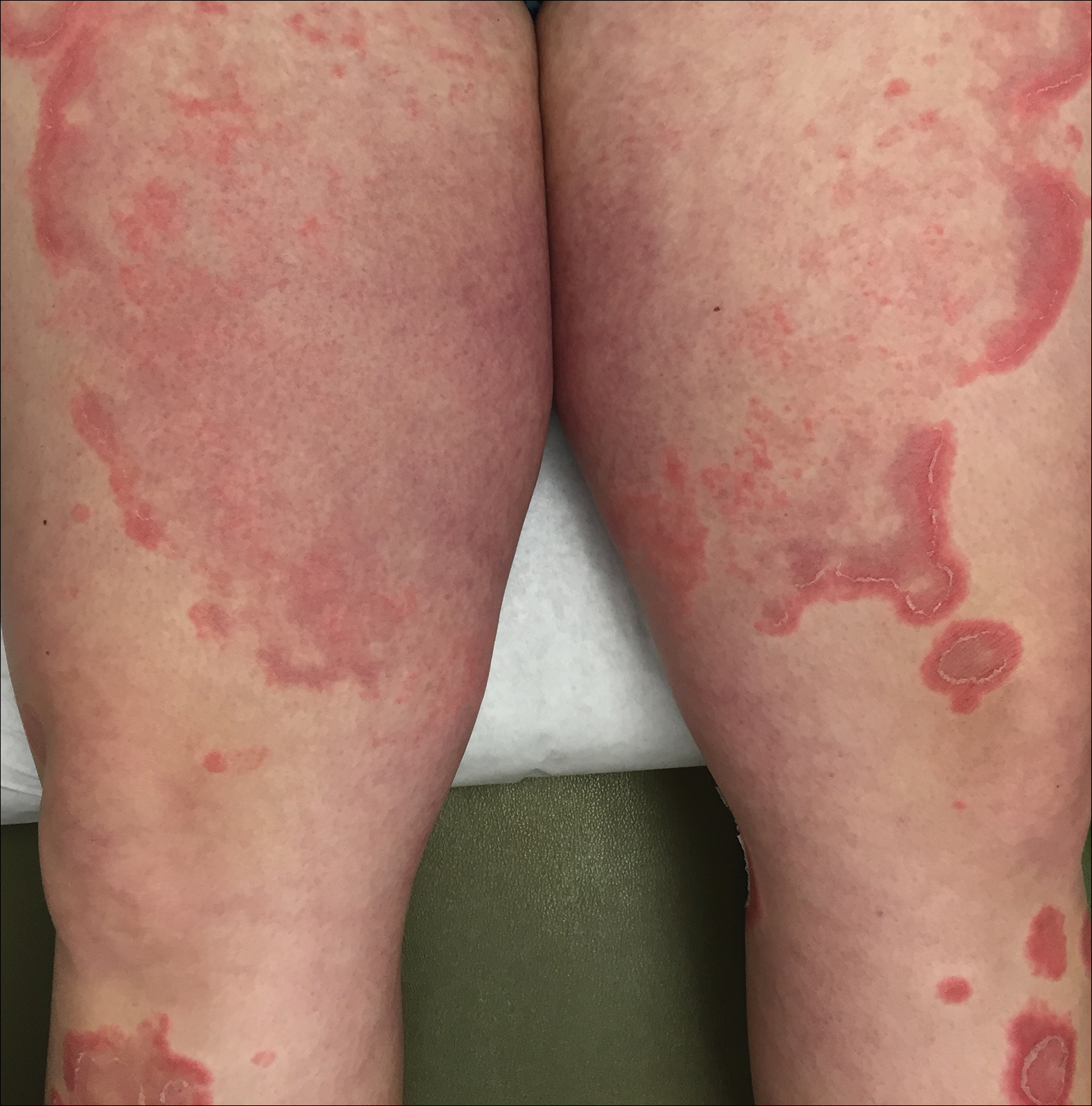
At 35 weeks’ gestation, the patient continued to report pruritus and burning in the areas where the rash had developed. The morphology of the rash had changed considerably, as she now had prominent, annular, pink plaques with central clearing, trailing scaling, and a border of subtle pustules on the legs. There also were rings of desquamative scaling on the palms. During follow-up at 37 weeks’ gestation, the back, chest, and abdomen were improved from the initial presentation, and annular pink plaques with central clearing were noted on the legs (Figure 3). Given the clinical and histopathologic findings, a diagnosis of PPP was made. It was recommended that she undergo increased fetal surveillance with close obstetric follow-up. Weekly office visits with obstetrics and twice-weekly Doppler ultrasounds and fetal nonstress tests were deemed appropriate management. The patient was scheduled for induction at 39 weeks’ gestation given the risk for potential harm to the fetus. She was maintained on low-dose methylprednisolone 4 mg once daily for the duration of the pregnancy. The patient continued to have gradual improvement of the rash at the low treatment dose.
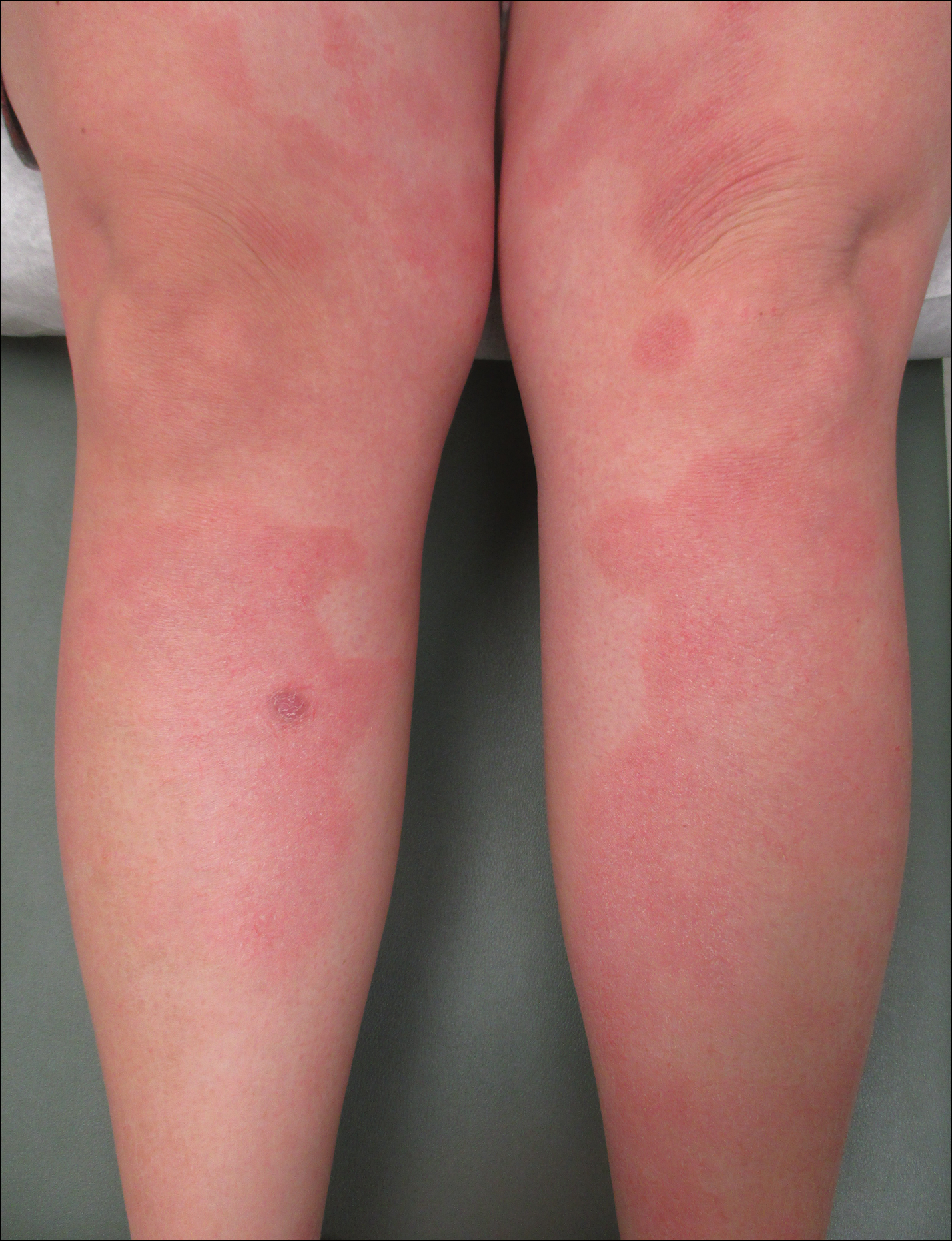
Following induction at 39 weeks’ gestation, the patient vaginally delivered a healthy, 6-lb male neonate at an outside hospital. She reported that the burning sensation improved within hours of delivery, and systemic steroids were stopped after delivery. At a follow-up visit 3 weeks postpartum, considerable improvement of the rash was noted with no evidence of pustules. Fading pink patches with a superficial scaling were noted on the back, chest, abdomen, arms, legs (Figure 4), and fingertips. The patient was counseled that PPP could recur in subsequent pregnancies and that she should be aware of the potential risks to the fetus.
Comment
In our patient, the diagnosis of PPP was supported by the presence of erythematous, coalescent plaques with small pustules at the margins and central erosions as well as the histologic findings of subcorneal pustules with mild acanthosis of the epidermis with spongiosis and a sparse neutrophilic infiltrate into the dermis.
The typical presentation of PPP is characterized by lesions that initially develop in skin folds with centrifugal spread.3 The lesions usually begin as erythematous plaques with a pustular ring with a central erosion. The face, palms, and soles of the feet typically are spared with occasional involvement of oral and esophageal mucosae. Biopsy findings typically include spongiform pustules with neutrophil invasion into the epidermis. Typical laboratory findings include electrolyte derangements with elevated ESR and leukocytosis.1
Diagnosis of PPP is critical given the potential for associated fetal morbidity and mortality.4 Anticipatory guidance for the patient also is necessary, as PPP can recur with subsequent pregnancies or even use of oral contraceptive pills (OCPs). Notably, a patient with recurrences of PPP with each of 9 pregnancies also experienced a recurrence when taking a combination estrogen/progesterone OCP, but not with an estrogen-only diethylstilbestrol OCP.5 Although the pathophysiology is not entirely understood, the development of PPP is thought to be related to the hormonal changes that occur in the third trimester, most notably due to elevated progesterone levels.2 The presence of progesterone in OCPs and recurrences associated with their use supports this altered hormonal state, contributing to the underlying pathophysiology of PPP.
Pustular psoriasis of pregnancy can occur in women without any personal or family history of psoriasis, and as such, it is unclear whether PPP is a separate entity or a hormonally induced variation of generalized pustular psoriasis. Recent evidence included reports of women with PPP who had a mutation in the IL-36 receptor antagonist, leading to a relative abundance of IL-36 inflammatory cytokines.6
The mainstay of treatment for PPP is oral corticosteroids. Cases of PPP that are unresponsive to systemic steroids have been documented, requiring treatment with cyclosporine.9 Antitumor necrosis factors also have been used safely during pregnancy.10 Narrowband UVB phototherapy also has been proposed as a treatment alternative for patients who do not respond to oral corticosteroids.11
Conclusion
Pustular psoriasis of pregnancy is a rare dermatosis of pregnancy that, unlike most other common dermatoses of pregnancy, is associated with adverse fetal outcomes. Diagnosis and management of PPP are critical to ensure the best care and outcomes for the patient and fetus and for a successful delivery of a healthy neonate. Our patient with PPP presented with involvement of the body, palms, and oral mucosa in the absence of systemic symptoms. Close follow-up and comanagement with the patient’s obstetrician ensured safe outcomes for the patient and the neonate.
- Lehrhoff S, Pomeranz MK. Specific dermatoses of pregnancy and their treatment. Dermatol Ther. 2013;26:274-284.
- Kar S, Krishnan A, Shivkumar PV. Pregnancy and skin [published online August 28, 2012]. J Obstet Gynaecol India. 2012;62:268-275.
- Kondo RN, Araújo FM, Pereira AM, et al. Pustular psoriasis of pregnancy (impetigo herpetiformis)—case report. An Bras Dermatol. 2013;88(6 suppl 1):186-189.
- Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24:101-104.
- Oumeish OY, Farraj SE, Bataineh AS. Some aspects of impetigo herpetiformis. Arch Dermatol. 1982;118:103-105.
- Sugiura K, Oiso N, Iinuma S, et al. IL36RN mutations underlie impetigo herpetiformis. J Invest Dermatol. 2014;134:2472-2474.
- Sugiura K. The genetic background of generalized pustular psoriasis: IL36RN mutations and CARD14 gain-of-function variants [published online March 5, 2014]. J Dermatol Sci. 2014;74:187-192.
- Li X, Chen M, Fu X, et al. Mutation analysis of the IL36RN gene in Chinese patients with generalized pustular psoriasis with/without psoriasis vulgaris. J Dermatol Sci. 2014;76:132-138.
- Hazarika D. Generalized pustular psoriasis of pregnancy successfully treated with cyclosporine. Indian J Dermatol Venereol Leprol. 2009;75:638.
- Puig L, Barco D, Alomar A. Treatment of psoriasis with anti-TNF drugs during pregnancy: case report and review of the literature. Dermatology. 2010;220:71-76.
- Bozdag K, Ozturk S, Ermete M. A case of recurrent impetigo herpetiformis treated with systemic corticosteroids and narrowband UVB [published online January 20, 2012]. Cutan Ocul Toxicol. 2012;31:67-69.
Pustular psoriasis of pregnancy (PPP), also known as impetigo herpetiformis, is a relatively rare cutaneous disorder of pregnancy wherein lesions typically appear in the third trimester and resolve after delivery; however, lesions may persist through the postpartum period. Pustular psoriasis of pregnancy may be considered a fifth dermatosis of pregnancy, alongside the classic dermatoses of atopic eruption of pregnancy, intrahepatic cholestasis of pregnancy, pemphigoid gestationis, and pruritic urticarial papules and plaques of pregnancy.1
As PPP is a rare disease, its effects on maternal-fetal health outcomes and management remain to be elucidated. Though maternal mortality is rare in PPP, it is a unique dermatosis of pregnancy because it may be associated with severe systemic maternal symptoms.2 Fetal morbidity and mortality are less predictable in PPP, with reported cases of stillbirth, fetal anomalies, and neonatal death thought to be due largely to placental insufficiency, even with control of symptoms.1,3 Given the risk of serious harm to the fetus, reporting of cases and discussion of PPP management is critical.
Case Report
An otherwise healthy 29-year-old G2P1 woman at 32 weeks’ gestation presented to our emergency department with a 1-week history of a pruritic, burning rash that started on the thighs then spread diffusely. She denied any similar rash in her prior pregnancy. She was not currently taking any medications except for prenatal vitamins and denied any systemic symptoms. The patient’s obstetrician initiated treatment with methylprednisolone 50 mg once daily for the rash 3 days prior to the current presentation, which had not seemed to help. On physical examination, edematous pink plaques studded with 1- to 2-mm collarettes of scaling and sparse 1-mm pustules involving the arms, chest, abdomen, back, groin, buttocks, and legs were noted. The plaques on the back and inner thighs had a peripheral rim of desquamative scaling. There were pink macules on the palms, and superficial desquamation was noted on the lips. The oral mucosa was otherwise spared (Figure 1).

Biopsy specimens from the left arm revealed discrete subcorneal pustules with mild acanthosis of the epidermis with spongiosis (Figure 2). The papillary dermis showed a sparse infiltrate of neutrophils with many marginated neutrophils within vessels. Direct immunofluorescence was negative for human IgG, IgA, IgM, complement component 3, and fibrinogen. Laboratory workup revealed leukocytosis of 21.5×109/L (reference range, 4.5–11.0×109/L) with neutrophilic predominance of 73.6% (reference range, 56%), an elevated erythrocyte sedimentation rate (ESR) of 40 mm/h (reference range, 0–20 mm/h), and a mild hypocalcemia of 8.6 mg/dL (reference range, 8.2–10.2 mg/dL). The patient was started on methylprednisone 40 mg once daily with a plan to taper the dose by 8 mg every 5 days.

At 35 weeks’ gestation, the patient continued to report pruritus and burning in the areas where the rash had developed. The morphology of the rash had changed considerably, as she now had prominent, annular, pink plaques with central clearing, trailing scaling, and a border of subtle pustules on the legs. There also were rings of desquamative scaling on the palms. During follow-up at 37 weeks’ gestation, the back, chest, and abdomen were improved from the initial presentation, and annular pink plaques with central clearing were noted on the legs (Figure 3). Given the clinical and histopathologic findings, a diagnosis of PPP was made. It was recommended that she undergo increased fetal surveillance with close obstetric follow-up. Weekly office visits with obstetrics and twice-weekly Doppler ultrasounds and fetal nonstress tests were deemed appropriate management. The patient was scheduled for induction at 39 weeks’ gestation given the risk for potential harm to the fetus. She was maintained on low-dose methylprednisolone 4 mg once daily for the duration of the pregnancy. The patient continued to have gradual improvement of the rash at the low treatment dose.

Following induction at 39 weeks’ gestation, the patient vaginally delivered a healthy, 6-lb male neonate at an outside hospital. She reported that the burning sensation improved within hours of delivery, and systemic steroids were stopped after delivery. At a follow-up visit 3 weeks postpartum, considerable improvement of the rash was noted with no evidence of pustules. Fading pink patches with a superficial scaling were noted on the back, chest, abdomen, arms, legs (Figure 4), and fingertips. The patient was counseled that PPP could recur in subsequent pregnancies and that she should be aware of the potential risks to the fetus.

Comment
In our patient, the diagnosis of PPP was supported by the presence of erythematous, coalescent plaques with small pustules at the margins and central erosions as well as the histologic findings of subcorneal pustules with mild acanthosis of the epidermis with spongiosis and a sparse neutrophilic infiltrate into the dermis.
The typical presentation of PPP is characterized by lesions that initially develop in skin folds with centrifugal spread.3 The lesions usually begin as erythematous plaques with a pustular ring with a central erosion. The face, palms, and soles of the feet typically are spared with occasional involvement of oral and esophageal mucosae. Biopsy findings typically include spongiform pustules with neutrophil invasion into the epidermis. Typical laboratory findings include electrolyte derangements with elevated ESR and leukocytosis.1
Diagnosis of PPP is critical given the potential for associated fetal morbidity and mortality.4 Anticipatory guidance for the patient also is necessary, as PPP can recur with subsequent pregnancies or even use of oral contraceptive pills (OCPs). Notably, a patient with recurrences of PPP with each of 9 pregnancies also experienced a recurrence when taking a combination estrogen/progesterone OCP, but not with an estrogen-only diethylstilbestrol OCP.5 Although the pathophysiology is not entirely understood, the development of PPP is thought to be related to the hormonal changes that occur in the third trimester, most notably due to elevated progesterone levels.2 The presence of progesterone in OCPs and recurrences associated with their use supports this altered hormonal state, contributing to the underlying pathophysiology of PPP.
Pustular psoriasis of pregnancy can occur in women without any personal or family history of psoriasis, and as such, it is unclear whether PPP is a separate entity or a hormonally induced variation of generalized pustular psoriasis. Recent evidence included reports of women with PPP who had a mutation in the IL-36 receptor antagonist, leading to a relative abundance of IL-36 inflammatory cytokines.6
The mainstay of treatment for PPP is oral corticosteroids. Cases of PPP that are unresponsive to systemic steroids have been documented, requiring treatment with cyclosporine.9 Antitumor necrosis factors also have been used safely during pregnancy.10 Narrowband UVB phototherapy also has been proposed as a treatment alternative for patients who do not respond to oral corticosteroids.11
Conclusion
Pustular psoriasis of pregnancy is a rare dermatosis of pregnancy that, unlike most other common dermatoses of pregnancy, is associated with adverse fetal outcomes. Diagnosis and management of PPP are critical to ensure the best care and outcomes for the patient and fetus and for a successful delivery of a healthy neonate. Our patient with PPP presented with involvement of the body, palms, and oral mucosa in the absence of systemic symptoms. Close follow-up and comanagement with the patient’s obstetrician ensured safe outcomes for the patient and the neonate.
Pustular psoriasis of pregnancy (PPP), also known as impetigo herpetiformis, is a relatively rare cutaneous disorder of pregnancy wherein lesions typically appear in the third trimester and resolve after delivery; however, lesions may persist through the postpartum period. Pustular psoriasis of pregnancy may be considered a fifth dermatosis of pregnancy, alongside the classic dermatoses of atopic eruption of pregnancy, intrahepatic cholestasis of pregnancy, pemphigoid gestationis, and pruritic urticarial papules and plaques of pregnancy.1
As PPP is a rare disease, its effects on maternal-fetal health outcomes and management remain to be elucidated. Though maternal mortality is rare in PPP, it is a unique dermatosis of pregnancy because it may be associated with severe systemic maternal symptoms.2 Fetal morbidity and mortality are less predictable in PPP, with reported cases of stillbirth, fetal anomalies, and neonatal death thought to be due largely to placental insufficiency, even with control of symptoms.1,3 Given the risk of serious harm to the fetus, reporting of cases and discussion of PPP management is critical.
Case Report
An otherwise healthy 29-year-old G2P1 woman at 32 weeks’ gestation presented to our emergency department with a 1-week history of a pruritic, burning rash that started on the thighs then spread diffusely. She denied any similar rash in her prior pregnancy. She was not currently taking any medications except for prenatal vitamins and denied any systemic symptoms. The patient’s obstetrician initiated treatment with methylprednisolone 50 mg once daily for the rash 3 days prior to the current presentation, which had not seemed to help. On physical examination, edematous pink plaques studded with 1- to 2-mm collarettes of scaling and sparse 1-mm pustules involving the arms, chest, abdomen, back, groin, buttocks, and legs were noted. The plaques on the back and inner thighs had a peripheral rim of desquamative scaling. There were pink macules on the palms, and superficial desquamation was noted on the lips. The oral mucosa was otherwise spared (Figure 1).

Biopsy specimens from the left arm revealed discrete subcorneal pustules with mild acanthosis of the epidermis with spongiosis (Figure 2). The papillary dermis showed a sparse infiltrate of neutrophils with many marginated neutrophils within vessels. Direct immunofluorescence was negative for human IgG, IgA, IgM, complement component 3, and fibrinogen. Laboratory workup revealed leukocytosis of 21.5×109/L (reference range, 4.5–11.0×109/L) with neutrophilic predominance of 73.6% (reference range, 56%), an elevated erythrocyte sedimentation rate (ESR) of 40 mm/h (reference range, 0–20 mm/h), and a mild hypocalcemia of 8.6 mg/dL (reference range, 8.2–10.2 mg/dL). The patient was started on methylprednisone 40 mg once daily with a plan to taper the dose by 8 mg every 5 days.

At 35 weeks’ gestation, the patient continued to report pruritus and burning in the areas where the rash had developed. The morphology of the rash had changed considerably, as she now had prominent, annular, pink plaques with central clearing, trailing scaling, and a border of subtle pustules on the legs. There also were rings of desquamative scaling on the palms. During follow-up at 37 weeks’ gestation, the back, chest, and abdomen were improved from the initial presentation, and annular pink plaques with central clearing were noted on the legs (Figure 3). Given the clinical and histopathologic findings, a diagnosis of PPP was made. It was recommended that she undergo increased fetal surveillance with close obstetric follow-up. Weekly office visits with obstetrics and twice-weekly Doppler ultrasounds and fetal nonstress tests were deemed appropriate management. The patient was scheduled for induction at 39 weeks’ gestation given the risk for potential harm to the fetus. She was maintained on low-dose methylprednisolone 4 mg once daily for the duration of the pregnancy. The patient continued to have gradual improvement of the rash at the low treatment dose.

Following induction at 39 weeks’ gestation, the patient vaginally delivered a healthy, 6-lb male neonate at an outside hospital. She reported that the burning sensation improved within hours of delivery, and systemic steroids were stopped after delivery. At a follow-up visit 3 weeks postpartum, considerable improvement of the rash was noted with no evidence of pustules. Fading pink patches with a superficial scaling were noted on the back, chest, abdomen, arms, legs (Figure 4), and fingertips. The patient was counseled that PPP could recur in subsequent pregnancies and that she should be aware of the potential risks to the fetus.

Comment
In our patient, the diagnosis of PPP was supported by the presence of erythematous, coalescent plaques with small pustules at the margins and central erosions as well as the histologic findings of subcorneal pustules with mild acanthosis of the epidermis with spongiosis and a sparse neutrophilic infiltrate into the dermis.
The typical presentation of PPP is characterized by lesions that initially develop in skin folds with centrifugal spread.3 The lesions usually begin as erythematous plaques with a pustular ring with a central erosion. The face, palms, and soles of the feet typically are spared with occasional involvement of oral and esophageal mucosae. Biopsy findings typically include spongiform pustules with neutrophil invasion into the epidermis. Typical laboratory findings include electrolyte derangements with elevated ESR and leukocytosis.1
Diagnosis of PPP is critical given the potential for associated fetal morbidity and mortality.4 Anticipatory guidance for the patient also is necessary, as PPP can recur with subsequent pregnancies or even use of oral contraceptive pills (OCPs). Notably, a patient with recurrences of PPP with each of 9 pregnancies also experienced a recurrence when taking a combination estrogen/progesterone OCP, but not with an estrogen-only diethylstilbestrol OCP.5 Although the pathophysiology is not entirely understood, the development of PPP is thought to be related to the hormonal changes that occur in the third trimester, most notably due to elevated progesterone levels.2 The presence of progesterone in OCPs and recurrences associated with their use supports this altered hormonal state, contributing to the underlying pathophysiology of PPP.
Pustular psoriasis of pregnancy can occur in women without any personal or family history of psoriasis, and as such, it is unclear whether PPP is a separate entity or a hormonally induced variation of generalized pustular psoriasis. Recent evidence included reports of women with PPP who had a mutation in the IL-36 receptor antagonist, leading to a relative abundance of IL-36 inflammatory cytokines.6
The mainstay of treatment for PPP is oral corticosteroids. Cases of PPP that are unresponsive to systemic steroids have been documented, requiring treatment with cyclosporine.9 Antitumor necrosis factors also have been used safely during pregnancy.10 Narrowband UVB phototherapy also has been proposed as a treatment alternative for patients who do not respond to oral corticosteroids.11
Conclusion
Pustular psoriasis of pregnancy is a rare dermatosis of pregnancy that, unlike most other common dermatoses of pregnancy, is associated with adverse fetal outcomes. Diagnosis and management of PPP are critical to ensure the best care and outcomes for the patient and fetus and for a successful delivery of a healthy neonate. Our patient with PPP presented with involvement of the body, palms, and oral mucosa in the absence of systemic symptoms. Close follow-up and comanagement with the patient’s obstetrician ensured safe outcomes for the patient and the neonate.
- Lehrhoff S, Pomeranz MK. Specific dermatoses of pregnancy and their treatment. Dermatol Ther. 2013;26:274-284.
- Kar S, Krishnan A, Shivkumar PV. Pregnancy and skin [published online August 28, 2012]. J Obstet Gynaecol India. 2012;62:268-275.
- Kondo RN, Araújo FM, Pereira AM, et al. Pustular psoriasis of pregnancy (impetigo herpetiformis)—case report. An Bras Dermatol. 2013;88(6 suppl 1):186-189.
- Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24:101-104.
- Oumeish OY, Farraj SE, Bataineh AS. Some aspects of impetigo herpetiformis. Arch Dermatol. 1982;118:103-105.
- Sugiura K, Oiso N, Iinuma S, et al. IL36RN mutations underlie impetigo herpetiformis. J Invest Dermatol. 2014;134:2472-2474.
- Sugiura K. The genetic background of generalized pustular psoriasis: IL36RN mutations and CARD14 gain-of-function variants [published online March 5, 2014]. J Dermatol Sci. 2014;74:187-192.
- Li X, Chen M, Fu X, et al. Mutation analysis of the IL36RN gene in Chinese patients with generalized pustular psoriasis with/without psoriasis vulgaris. J Dermatol Sci. 2014;76:132-138.
- Hazarika D. Generalized pustular psoriasis of pregnancy successfully treated with cyclosporine. Indian J Dermatol Venereol Leprol. 2009;75:638.
- Puig L, Barco D, Alomar A. Treatment of psoriasis with anti-TNF drugs during pregnancy: case report and review of the literature. Dermatology. 2010;220:71-76.
- Bozdag K, Ozturk S, Ermete M. A case of recurrent impetigo herpetiformis treated with systemic corticosteroids and narrowband UVB [published online January 20, 2012]. Cutan Ocul Toxicol. 2012;31:67-69.
- Lehrhoff S, Pomeranz MK. Specific dermatoses of pregnancy and their treatment. Dermatol Ther. 2013;26:274-284.
- Kar S, Krishnan A, Shivkumar PV. Pregnancy and skin [published online August 28, 2012]. J Obstet Gynaecol India. 2012;62:268-275.
- Kondo RN, Araújo FM, Pereira AM, et al. Pustular psoriasis of pregnancy (impetigo herpetiformis)—case report. An Bras Dermatol. 2013;88(6 suppl 1):186-189.
- Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24:101-104.
- Oumeish OY, Farraj SE, Bataineh AS. Some aspects of impetigo herpetiformis. Arch Dermatol. 1982;118:103-105.
- Sugiura K, Oiso N, Iinuma S, et al. IL36RN mutations underlie impetigo herpetiformis. J Invest Dermatol. 2014;134:2472-2474.
- Sugiura K. The genetic background of generalized pustular psoriasis: IL36RN mutations and CARD14 gain-of-function variants [published online March 5, 2014]. J Dermatol Sci. 2014;74:187-192.
- Li X, Chen M, Fu X, et al. Mutation analysis of the IL36RN gene in Chinese patients with generalized pustular psoriasis with/without psoriasis vulgaris. J Dermatol Sci. 2014;76:132-138.
- Hazarika D. Generalized pustular psoriasis of pregnancy successfully treated with cyclosporine. Indian J Dermatol Venereol Leprol. 2009;75:638.
- Puig L, Barco D, Alomar A. Treatment of psoriasis with anti-TNF drugs during pregnancy: case report and review of the literature. Dermatology. 2010;220:71-76.
- Bozdag K, Ozturk S, Ermete M. A case of recurrent impetigo herpetiformis treated with systemic corticosteroids and narrowband UVB [published online January 20, 2012]. Cutan Ocul Toxicol. 2012;31:67-69.
Practice Points
- Given its association with maternal and fetal morbidity/mortality, it is important for physicians to have a high suspicion for pustular psoriasis of pregnancy (PPP) in pregnant women with widespread cutaneous eruptions.
- Oral corticosteroids and close involvement of obstetric care is the mainstay of treatment for PPP.
Study using U.K. data quantifies infection risk associated with psoriasis
Psoriasis was linked to increased risk of serious infection, with more severe disease associated with increased infection risk, in a study that used electronic medical records of patients in the United Kingdom.
The most common serious infections were lower respiratory tract, skin and soft tissue, and upper respiratory tract infections; and the most common opportunistic infection was tuberculosis, reported Junko Takeshita, MD, PhD, of the departments of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, and her coauthors.
They identified 199,700 psoriasis patients and 954,315 healthy patients from THIN (the Health Improvement Network), a medical records database in the United Kingdom. Of the psoriasis patients, 187,258 had mild disease and 12,442 had moderate to severe disease; almost 70% of patients with moderate to severe disease were treated with methotrexate.
Adjusted hazard ratios for serious infection were 1.21 (95% confidence interval, 1.18-1.23) for psoriasis patients overall, 1.18 (95% confidence interval, 1.16-1.21) for those with mild psoriasis, and 1.63 (95% CI, 1.52-1.75) for those with moderate to severe psoriasis, Dr. Takeshita and her coauthors wrote in the Journal of Investigative Dermatology.
Among all psoriasis patients, the attributable risk of serious infection was 16.2 per 10,000 person-years, compared with 14.4 per 10,000 person-years among those with mild psoriasis, and 49.5 per 10,000 person-years, among those with moderate to severe disease.
The investigators also analyzed data from a nested cohort – the iHOPE (Incident Health Outcomes and Psoriasis Events) study – of 8,569 psoriasis patients, with mild (less than 3% of body surface area involvement) or moderate to severe disease (3% or greater BSA), and 83,540 matched patients without psoriasis.
The adjusted HR for serious infection was 1.21 (95% CI, 1.09-1.35) for all psoriasis patients, 1.16 (95% CI, 0.99-1.35) for those with mild disease, and 1.27 (95% CI, 1.10-1.47) for those with moderate to severe disease. When patients who had received immunosuppressive treatment were excluded from the analysis, hazard ratios were similar among the different psoriasis groups, at 1.18 for all psoriasis patients (95% CI, 1.05-1.32), 1.15 among those with mild disease (95% CI, 0.99-1.34), and 1.21 for those with moderate to severe disease (95% CI, 1.03-1.42).
“Importantly, the risk of serious infection was observed to be similar in both the full THIN and iHOPE cohorts with the exception of the moderate to severe psoriasis subgroup among whom the risk of serious infection was attenuated but still significantly elevated in the iHOPE versus full THIN cohort,” they observed.
In the THIN cohort, the most common opportunistic infection “by far” was tuberculosis, with incidence rates of 1.05, 0.94, and 3.00 per 10,000 person-years among all psoriasis patients, patients with mild disease, and patients with moderate to severe disease, respectively, compared with 1.15 for those without psoriasis.
Patients with moderate to severe disease had an increased risk of opportunistic infection (HR, 1.57; 95% CI, 1.06-2.34), but rates were similar among those with mild disease and those without psoriasis, Dr. Takeshita and her colleagues reported. But the opportunistic infection risk was “substantially attenuated” when patients who had received immunosuppressive treatment were excluded (HR, 1.17; 95% CI, 0.44-3.12).
Patients with moderate to severe disease also had the greatest risk of herpes zoster (HR, 1.17; 95% CI, 1.06-1.30). While the increased risk of herpes zoster was smaller in patients with mild psoriasis, it was still significant (HR, 1.07; 95% CI, 1.05-1.10). Again, when exclusion of patients who had received immunosuppressive therapies, the risk for herpes zoster associated with moderate to severe psoriasis no longer was elevated (HR, 0.97; 95% CI, 0.76-1.23).
“Our findings suggest that psoriasis is associated with an increased risk of serious infection, and more severe psoriasis, whether defined by treatment pattern or by BSA involvement, is a predictor of greater serious infection risk,” the authors wrote. Clinicians should ensure that patients, especially those with severe disease and those who receive immunosuppressive treatment, are vaccinated against influenza and pneumonia, and “should also consider herpes zoster vaccination with the new nonlive vaccine.”
“Future studies will be important to further characterize the risk of various infections among patients with psoriasis, compare the risk of infection associated with psoriasis to that of other chronic diseases, and delineate the pathophysiologic mechanisms that contribute to the increased risk of infections associated with psoriasis and its therapies,” they concluded.
The study was funded by an unrestricted Pfizer grant. Dr. Takeshita has received a research grant (to the Trustees of the University of Pennsylvania) from Pfizer for unrelated work payment for continuing medical education work related to psoriasis supported indirectly by Eli Lilly and Novartis. Other authors’ disclosures included servings as a consultant for Bristol-Myers Squibb, Novartis, Pfizer, Coherus, and other pharmaceutical companies.
dermnews@mdedge.com
SOURCE: Takeshita J et al. J Invest Dermatol. 2018 Mar 2. doi: 10.1016/j.jid.2018.01.039.
Psoriasis was linked to increased risk of serious infection, with more severe disease associated with increased infection risk, in a study that used electronic medical records of patients in the United Kingdom.
The most common serious infections were lower respiratory tract, skin and soft tissue, and upper respiratory tract infections; and the most common opportunistic infection was tuberculosis, reported Junko Takeshita, MD, PhD, of the departments of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, and her coauthors.
They identified 199,700 psoriasis patients and 954,315 healthy patients from THIN (the Health Improvement Network), a medical records database in the United Kingdom. Of the psoriasis patients, 187,258 had mild disease and 12,442 had moderate to severe disease; almost 70% of patients with moderate to severe disease were treated with methotrexate.
Adjusted hazard ratios for serious infection were 1.21 (95% confidence interval, 1.18-1.23) for psoriasis patients overall, 1.18 (95% confidence interval, 1.16-1.21) for those with mild psoriasis, and 1.63 (95% CI, 1.52-1.75) for those with moderate to severe psoriasis, Dr. Takeshita and her coauthors wrote in the Journal of Investigative Dermatology.
Among all psoriasis patients, the attributable risk of serious infection was 16.2 per 10,000 person-years, compared with 14.4 per 10,000 person-years among those with mild psoriasis, and 49.5 per 10,000 person-years, among those with moderate to severe disease.
The investigators also analyzed data from a nested cohort – the iHOPE (Incident Health Outcomes and Psoriasis Events) study – of 8,569 psoriasis patients, with mild (less than 3% of body surface area involvement) or moderate to severe disease (3% or greater BSA), and 83,540 matched patients without psoriasis.
The adjusted HR for serious infection was 1.21 (95% CI, 1.09-1.35) for all psoriasis patients, 1.16 (95% CI, 0.99-1.35) for those with mild disease, and 1.27 (95% CI, 1.10-1.47) for those with moderate to severe disease. When patients who had received immunosuppressive treatment were excluded from the analysis, hazard ratios were similar among the different psoriasis groups, at 1.18 for all psoriasis patients (95% CI, 1.05-1.32), 1.15 among those with mild disease (95% CI, 0.99-1.34), and 1.21 for those with moderate to severe disease (95% CI, 1.03-1.42).
“Importantly, the risk of serious infection was observed to be similar in both the full THIN and iHOPE cohorts with the exception of the moderate to severe psoriasis subgroup among whom the risk of serious infection was attenuated but still significantly elevated in the iHOPE versus full THIN cohort,” they observed.
In the THIN cohort, the most common opportunistic infection “by far” was tuberculosis, with incidence rates of 1.05, 0.94, and 3.00 per 10,000 person-years among all psoriasis patients, patients with mild disease, and patients with moderate to severe disease, respectively, compared with 1.15 for those without psoriasis.
Patients with moderate to severe disease had an increased risk of opportunistic infection (HR, 1.57; 95% CI, 1.06-2.34), but rates were similar among those with mild disease and those without psoriasis, Dr. Takeshita and her colleagues reported. But the opportunistic infection risk was “substantially attenuated” when patients who had received immunosuppressive treatment were excluded (HR, 1.17; 95% CI, 0.44-3.12).
Patients with moderate to severe disease also had the greatest risk of herpes zoster (HR, 1.17; 95% CI, 1.06-1.30). While the increased risk of herpes zoster was smaller in patients with mild psoriasis, it was still significant (HR, 1.07; 95% CI, 1.05-1.10). Again, when exclusion of patients who had received immunosuppressive therapies, the risk for herpes zoster associated with moderate to severe psoriasis no longer was elevated (HR, 0.97; 95% CI, 0.76-1.23).
“Our findings suggest that psoriasis is associated with an increased risk of serious infection, and more severe psoriasis, whether defined by treatment pattern or by BSA involvement, is a predictor of greater serious infection risk,” the authors wrote. Clinicians should ensure that patients, especially those with severe disease and those who receive immunosuppressive treatment, are vaccinated against influenza and pneumonia, and “should also consider herpes zoster vaccination with the new nonlive vaccine.”
“Future studies will be important to further characterize the risk of various infections among patients with psoriasis, compare the risk of infection associated with psoriasis to that of other chronic diseases, and delineate the pathophysiologic mechanisms that contribute to the increased risk of infections associated with psoriasis and its therapies,” they concluded.
The study was funded by an unrestricted Pfizer grant. Dr. Takeshita has received a research grant (to the Trustees of the University of Pennsylvania) from Pfizer for unrelated work payment for continuing medical education work related to psoriasis supported indirectly by Eli Lilly and Novartis. Other authors’ disclosures included servings as a consultant for Bristol-Myers Squibb, Novartis, Pfizer, Coherus, and other pharmaceutical companies.
dermnews@mdedge.com
SOURCE: Takeshita J et al. J Invest Dermatol. 2018 Mar 2. doi: 10.1016/j.jid.2018.01.039.
Psoriasis was linked to increased risk of serious infection, with more severe disease associated with increased infection risk, in a study that used electronic medical records of patients in the United Kingdom.
The most common serious infections were lower respiratory tract, skin and soft tissue, and upper respiratory tract infections; and the most common opportunistic infection was tuberculosis, reported Junko Takeshita, MD, PhD, of the departments of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, and her coauthors.
They identified 199,700 psoriasis patients and 954,315 healthy patients from THIN (the Health Improvement Network), a medical records database in the United Kingdom. Of the psoriasis patients, 187,258 had mild disease and 12,442 had moderate to severe disease; almost 70% of patients with moderate to severe disease were treated with methotrexate.
Adjusted hazard ratios for serious infection were 1.21 (95% confidence interval, 1.18-1.23) for psoriasis patients overall, 1.18 (95% confidence interval, 1.16-1.21) for those with mild psoriasis, and 1.63 (95% CI, 1.52-1.75) for those with moderate to severe psoriasis, Dr. Takeshita and her coauthors wrote in the Journal of Investigative Dermatology.
Among all psoriasis patients, the attributable risk of serious infection was 16.2 per 10,000 person-years, compared with 14.4 per 10,000 person-years among those with mild psoriasis, and 49.5 per 10,000 person-years, among those with moderate to severe disease.
The investigators also analyzed data from a nested cohort – the iHOPE (Incident Health Outcomes and Psoriasis Events) study – of 8,569 psoriasis patients, with mild (less than 3% of body surface area involvement) or moderate to severe disease (3% or greater BSA), and 83,540 matched patients without psoriasis.
The adjusted HR for serious infection was 1.21 (95% CI, 1.09-1.35) for all psoriasis patients, 1.16 (95% CI, 0.99-1.35) for those with mild disease, and 1.27 (95% CI, 1.10-1.47) for those with moderate to severe disease. When patients who had received immunosuppressive treatment were excluded from the analysis, hazard ratios were similar among the different psoriasis groups, at 1.18 for all psoriasis patients (95% CI, 1.05-1.32), 1.15 among those with mild disease (95% CI, 0.99-1.34), and 1.21 for those with moderate to severe disease (95% CI, 1.03-1.42).
“Importantly, the risk of serious infection was observed to be similar in both the full THIN and iHOPE cohorts with the exception of the moderate to severe psoriasis subgroup among whom the risk of serious infection was attenuated but still significantly elevated in the iHOPE versus full THIN cohort,” they observed.
In the THIN cohort, the most common opportunistic infection “by far” was tuberculosis, with incidence rates of 1.05, 0.94, and 3.00 per 10,000 person-years among all psoriasis patients, patients with mild disease, and patients with moderate to severe disease, respectively, compared with 1.15 for those without psoriasis.
Patients with moderate to severe disease had an increased risk of opportunistic infection (HR, 1.57; 95% CI, 1.06-2.34), but rates were similar among those with mild disease and those without psoriasis, Dr. Takeshita and her colleagues reported. But the opportunistic infection risk was “substantially attenuated” when patients who had received immunosuppressive treatment were excluded (HR, 1.17; 95% CI, 0.44-3.12).
Patients with moderate to severe disease also had the greatest risk of herpes zoster (HR, 1.17; 95% CI, 1.06-1.30). While the increased risk of herpes zoster was smaller in patients with mild psoriasis, it was still significant (HR, 1.07; 95% CI, 1.05-1.10). Again, when exclusion of patients who had received immunosuppressive therapies, the risk for herpes zoster associated with moderate to severe psoriasis no longer was elevated (HR, 0.97; 95% CI, 0.76-1.23).
“Our findings suggest that psoriasis is associated with an increased risk of serious infection, and more severe psoriasis, whether defined by treatment pattern or by BSA involvement, is a predictor of greater serious infection risk,” the authors wrote. Clinicians should ensure that patients, especially those with severe disease and those who receive immunosuppressive treatment, are vaccinated against influenza and pneumonia, and “should also consider herpes zoster vaccination with the new nonlive vaccine.”
“Future studies will be important to further characterize the risk of various infections among patients with psoriasis, compare the risk of infection associated with psoriasis to that of other chronic diseases, and delineate the pathophysiologic mechanisms that contribute to the increased risk of infections associated with psoriasis and its therapies,” they concluded.
The study was funded by an unrestricted Pfizer grant. Dr. Takeshita has received a research grant (to the Trustees of the University of Pennsylvania) from Pfizer for unrelated work payment for continuing medical education work related to psoriasis supported indirectly by Eli Lilly and Novartis. Other authors’ disclosures included servings as a consultant for Bristol-Myers Squibb, Novartis, Pfizer, Coherus, and other pharmaceutical companies.
dermnews@mdedge.com
SOURCE: Takeshita J et al. J Invest Dermatol. 2018 Mar 2. doi: 10.1016/j.jid.2018.01.039.
Key clinical point: Psoriasis is linked to risk of serious infection, with increased risk in more severe disease.
Major finding: Hazard ratios for serious infection were 1.21 (95% CI, 1.18-1.23) for psoriasis overall, 1.18 (95% CI, 1.16-1.21) for mild psoriasis, and 1.63 (95% CI, 1.52-1.75) for moderate to severe psoriasis.
Study details: The evaluation included data on 199,700 patients with psoriasis and 954,315 patients without psoriasis in a U.K. electronic medical records database.
Disclosures: The study was funded by an unrestricted Pfizer grant. Dr. Takeshita has received a research grant (to the Trustees of the University of Pennsylvania) from Pfizer for unrelated work payment for continuing medical education work related to psoriasis supported indirectly by Eli Lilly and Novartis. Other authors’ disclosures included servings as a consultant for Bristol-Myers Squibb, Novartis, Pfizer, Coherus, and other pharmaceutical companies.
Source: Takeshita J et al. J Invest Dermatol. 2018 Mar 2. doi: 10.1016/j.jid.2018.01.039.
mainbar
Psoriasis was linked to increased risk of serious infection, with more severe disease associated with increased infection risk, in a study that used electronic medical records of patients in the United Kingdom.
The most common serious infections were lower respiratory tract, skin and soft tissue, and upper respiratory tract infections; and the most common opportunistic infection was tuberculosis, reported Junko Takeshita, MD, PhD, of the departments of dermatology and epidemiology at the University of Pennsylvania, Philadelphia, and her coauthors.
Adjusted hazard ratios for serious infection were 1.21 (95% confidence interval, 1.18-1.23) for psoriasis patients overall, 1.18 (95% confidence interval, 1.16-1.21) for those with mild psoriasis, and 1.63 (95% CI, 1.52-1.75) for those with moderate to severe psoriasis, Dr. Takeshita and her coauthors wrote in the Journal of Investigative Dermatology.
Among all psoriasis patients, the attributable risk of serious infection was 16.2 per 10,000 person-years, compared with 14.4 per 10,000 person-years among those with mild psoriasis, and 49.5 per 10,000 person-years, among those with moderate to severe disease.
The investigators also analyzed data from a nested cohort – the iHOPE (Incident Health Outcomes and Psoriasis Events) study – of 8,569 psoriasis patients, with mild (less than 3% of body surface area involvement) or moderate to severe disease (3% or greater BSA), and 83,540 matched patients without psoriasis. The adjusted HR for serious infection was 1.21 (95% CI, 1.09-1.35) for all psoriasis patients, 1.16 (95% CI, 0.99-1.35) for those with mild disease, and 1.27 (95% CI, 1.10-1.47) for those with moderate to severe disease. When patients who had received immunosuppressive treatment were excluded from the analysis, hazard ratios were similar among the different psoriasis groups, at 1.18 for all psoriasis patients (95% CI, 1.05-1.32), 1.15 among those with mild disease (95% CI, 0.99-1.34), and 1.21 for those with moderate to severe disease (95% CI, 1.03-1.42).
“Importantly, the risk of serious infection was observed to be similar in both the full THIN and iHOPE cohorts with the exception of the moderate to severe psoriasis subgroup among whom the risk of serious infection was attenuated but still significantly elevated in the iHOPE versus full THIN cohort,” they observed.
In the THIN cohort, the most common opportunistic infection “by far” was tuberculosis, with incidence rates of 1.05, 0.94, and 3.00 per 10,000 person-years among all psoriasis patients, patients with mild disease, and patients with moderate to severe disease, respectively, compared with 1.15 for those without psoriasis.
Patients with moderate to severe disease had an increased risk of opportunistic infection (HR, 1.57; 95% CI, 1.06-2.34), but rates were similar among those with mild disease and those without psoriasis, Dr. Takeshita and her colleagues reported. But the opportunistic infection risk was “substantially attenuated” when patients who had received immunosuppressive treatment were excluded (HR, 1.17; 95% CI, 0.44-3.12).
Patients with moderate to severe disease also had the greatest risk of herpes zoster (HR, 1.17; 95% CI, 1.06-1.30). While the increased risk of herpes zoster was smaller in patients with mild psoriasis, it was still significant (HR, 1.07; 95% CI, 1.05-1.10). Again, when exclusion of patients who had received immunosuppressive therapies, the risk for herpes zoster associated with moderate to severe psoriasis no longer was elevated (HR, 0.97; 95% CI, 0.76-1.23).
“Our findings suggest that psoriasis is associated with an increased risk of serious infection, and more severe psoriasis, whether defined by treatment pattern or by BSA involvement, is a predictor of greater serious infection risk,” the authors wrote. Clinicians should ensure that patients, especially those with severe disease and those who receive immunosuppressive treatment, are vaccinated against influenza and pneumonia, and “should also consider herpes zoster vaccination with the new nonlive vaccine.”
“Future studies will be important to further characterize the risk of various infections among patients with psoriasis, compare the risk of infection associated with psoriasis to that of other chronic diseases, and delineate the pathophysiologic mechanisms that contribute to the increased risk of infections associated with psoriasis and its therapies,” they concluded.
The study was funded by an unrestricted Pfizer grant. Dr. Takeshita has received a research grant (to the Trustees of the University of Pennsylvania) from Pfizer for unrelated work payment for continuing medical education work related to psoriasis supported indirectly by Eli Lilly and Novartis. Other authors’ disclosures included servings as a consultant for Bristol-Myers Squibb, Novartis, Pfizer, Coherus, and other pharmaceutical companies.
SOURCE: Takeshita J et al. J Invest Dermatol. 2018 Mar 2. doi: 10.1016/j.jid.2018.01.039.
Psoriasis patients often have history of childhood trauma
reported Maria Luigia Crosta, of Catholic University of the Sacred Heart, Rome, and her associates.
Other studies have shown that among dermatologic disorders, psoriasis has the highest link to psychiatric illness such as mood, anxiety, and personality disorders, and that patients with psoriasis have an increased risk of suicidal ideation, the investigators said.
“Improving resilience with a multidisciplinary approach and an early psychological intervention could facilitate the management of psoriasis, by promoting the establishment of a stronger therapeutic alliance and a better acceptance of disease. Programs for psoriasis patients should focus on self-motivation and strengthening of self-efficacy,” Dr. Crosta and her associates concluded.
SOURCE: Crosta ML et al. J Psychosom Res. 2018;106:25-8.
reported Maria Luigia Crosta, of Catholic University of the Sacred Heart, Rome, and her associates.
Other studies have shown that among dermatologic disorders, psoriasis has the highest link to psychiatric illness such as mood, anxiety, and personality disorders, and that patients with psoriasis have an increased risk of suicidal ideation, the investigators said.
“Improving resilience with a multidisciplinary approach and an early psychological intervention could facilitate the management of psoriasis, by promoting the establishment of a stronger therapeutic alliance and a better acceptance of disease. Programs for psoriasis patients should focus on self-motivation and strengthening of self-efficacy,” Dr. Crosta and her associates concluded.
SOURCE: Crosta ML et al. J Psychosom Res. 2018;106:25-8.
reported Maria Luigia Crosta, of Catholic University of the Sacred Heart, Rome, and her associates.
Other studies have shown that among dermatologic disorders, psoriasis has the highest link to psychiatric illness such as mood, anxiety, and personality disorders, and that patients with psoriasis have an increased risk of suicidal ideation, the investigators said.
“Improving resilience with a multidisciplinary approach and an early psychological intervention could facilitate the management of psoriasis, by promoting the establishment of a stronger therapeutic alliance and a better acceptance of disease. Programs for psoriasis patients should focus on self-motivation and strengthening of self-efficacy,” Dr. Crosta and her associates concluded.
SOURCE: Crosta ML et al. J Psychosom Res. 2018;106:25-8.
FROM THE JOURNAL OF PSYCHOSOMATIC RESEARCH
FDA approves certolizumab label update for pregnancy, breastfeeding
The manufacturer of certolizumab pegol, UCB, announced March 22 that the Food and Drug Administration approved a label update to the biologic that includes pharmacokinetic data showing negligible to low transfer of the biologic through the placenta and minimal mother-to-infant transfer from breast milk.
In the CRIB study, certolizumab levels were below the lower limit of quantification (defined as 0.032 mcg/mL) in 13 out of 15 infant blood samples at birth and in all samples at weeks 4 and 8. No anticertolizumab antibodies were detected in mothers, umbilical cords, or infants.
In the CRADLE study, 56% of 137 breast milk samples from 17 mothers had no measurable certolizumab, and the remaining samples showed minimal levels of the biologic. No serious adverse reactions were noted in the 17 infants in the study.
“It is well recognized that women with chronic inflammatory disease face uncertainty during motherhood given the lack of information on treatment during pregnancy and breastfeeding. Many women with chronic inflammatory disease discontinue their biologic treatment during pregnancy, often when they need disease control the most,” said CRADLE lead study author Megan E. B. Clowse, MD, of Duke University, Durham, N.C., in a press release issued by UCB. “These data for Cimzia provide important information to empower women and healthcare providers making decisions about treatment during pregnancy and breastfeeding.”
UCB said that limited data from an ongoing pregnancy registry regarding the use of certolizumab in pregnant women are not sufficient to inform a risk of major birth defects or other adverse pregnancy outcomes.
The manufacturer of certolizumab pegol, UCB, announced March 22 that the Food and Drug Administration approved a label update to the biologic that includes pharmacokinetic data showing negligible to low transfer of the biologic through the placenta and minimal mother-to-infant transfer from breast milk.
In the CRIB study, certolizumab levels were below the lower limit of quantification (defined as 0.032 mcg/mL) in 13 out of 15 infant blood samples at birth and in all samples at weeks 4 and 8. No anticertolizumab antibodies were detected in mothers, umbilical cords, or infants.
In the CRADLE study, 56% of 137 breast milk samples from 17 mothers had no measurable certolizumab, and the remaining samples showed minimal levels of the biologic. No serious adverse reactions were noted in the 17 infants in the study.
“It is well recognized that women with chronic inflammatory disease face uncertainty during motherhood given the lack of information on treatment during pregnancy and breastfeeding. Many women with chronic inflammatory disease discontinue their biologic treatment during pregnancy, often when they need disease control the most,” said CRADLE lead study author Megan E. B. Clowse, MD, of Duke University, Durham, N.C., in a press release issued by UCB. “These data for Cimzia provide important information to empower women and healthcare providers making decisions about treatment during pregnancy and breastfeeding.”
UCB said that limited data from an ongoing pregnancy registry regarding the use of certolizumab in pregnant women are not sufficient to inform a risk of major birth defects or other adverse pregnancy outcomes.
The manufacturer of certolizumab pegol, UCB, announced March 22 that the Food and Drug Administration approved a label update to the biologic that includes pharmacokinetic data showing negligible to low transfer of the biologic through the placenta and minimal mother-to-infant transfer from breast milk.
In the CRIB study, certolizumab levels were below the lower limit of quantification (defined as 0.032 mcg/mL) in 13 out of 15 infant blood samples at birth and in all samples at weeks 4 and 8. No anticertolizumab antibodies were detected in mothers, umbilical cords, or infants.
In the CRADLE study, 56% of 137 breast milk samples from 17 mothers had no measurable certolizumab, and the remaining samples showed minimal levels of the biologic. No serious adverse reactions were noted in the 17 infants in the study.
“It is well recognized that women with chronic inflammatory disease face uncertainty during motherhood given the lack of information on treatment during pregnancy and breastfeeding. Many women with chronic inflammatory disease discontinue their biologic treatment during pregnancy, often when they need disease control the most,” said CRADLE lead study author Megan E. B. Clowse, MD, of Duke University, Durham, N.C., in a press release issued by UCB. “These data for Cimzia provide important information to empower women and healthcare providers making decisions about treatment during pregnancy and breastfeeding.”
UCB said that limited data from an ongoing pregnancy registry regarding the use of certolizumab in pregnant women are not sufficient to inform a risk of major birth defects or other adverse pregnancy outcomes.
FDA approves IL-23 antagonist for plaque psoriasis
in adults who are eligible for systemic therapy or phototherapy, according to a statement from Sun Pharma.
Tildrakizumab is administered at a dose of 100 mg, subcutaneously, at weeks 0 and 4, then every 12 weeks. Approval is based on data from two phase 3, identically designed clinical trials, reSURFACE1 and reSURFACE2. Both studies were multicenter, randomized, double-blind, and placebo controlled. In the studies, 926 patients received tildrakizumab (616 patients) or placebo (310 patients).
The effectiveness of tildrakizumab extended beyond 12 weeks, with 74% of patients achieving a PASI 75 at 28 weeks after three doses. This percentage grew to 84% at week 64 in patients who continued treatment. Similar results were observed with PGA scores, with 69% of patients who had a PGA score of 0 or 1 at 12 weeks maintaining that score at week 28.
Tildrakizumab has been associated with serious side effects, including serious allergic reactions including skin rash, swelling of the face and mouth, trouble breathing, and chest tightness. It may also increase patient susceptibility to infection. It is approved with a Medication Guide for patients, explaining the potential risks associated with treatment.
Tildrakizumab will be marketed as Ilumya.
Sun Pharma is working with the FDA on postapproval commitments, and once that has been completed, they will have a better idea of when it will become available, according to a spokesperson for the manufacturer. The cost is not yet available.
in adults who are eligible for systemic therapy or phototherapy, according to a statement from Sun Pharma.
Tildrakizumab is administered at a dose of 100 mg, subcutaneously, at weeks 0 and 4, then every 12 weeks. Approval is based on data from two phase 3, identically designed clinical trials, reSURFACE1 and reSURFACE2. Both studies were multicenter, randomized, double-blind, and placebo controlled. In the studies, 926 patients received tildrakizumab (616 patients) or placebo (310 patients).
The effectiveness of tildrakizumab extended beyond 12 weeks, with 74% of patients achieving a PASI 75 at 28 weeks after three doses. This percentage grew to 84% at week 64 in patients who continued treatment. Similar results were observed with PGA scores, with 69% of patients who had a PGA score of 0 or 1 at 12 weeks maintaining that score at week 28.
Tildrakizumab has been associated with serious side effects, including serious allergic reactions including skin rash, swelling of the face and mouth, trouble breathing, and chest tightness. It may also increase patient susceptibility to infection. It is approved with a Medication Guide for patients, explaining the potential risks associated with treatment.
Tildrakizumab will be marketed as Ilumya.
Sun Pharma is working with the FDA on postapproval commitments, and once that has been completed, they will have a better idea of when it will become available, according to a spokesperson for the manufacturer. The cost is not yet available.
in adults who are eligible for systemic therapy or phototherapy, according to a statement from Sun Pharma.
Tildrakizumab is administered at a dose of 100 mg, subcutaneously, at weeks 0 and 4, then every 12 weeks. Approval is based on data from two phase 3, identically designed clinical trials, reSURFACE1 and reSURFACE2. Both studies were multicenter, randomized, double-blind, and placebo controlled. In the studies, 926 patients received tildrakizumab (616 patients) or placebo (310 patients).
The effectiveness of tildrakizumab extended beyond 12 weeks, with 74% of patients achieving a PASI 75 at 28 weeks after three doses. This percentage grew to 84% at week 64 in patients who continued treatment. Similar results were observed with PGA scores, with 69% of patients who had a PGA score of 0 or 1 at 12 weeks maintaining that score at week 28.
Tildrakizumab has been associated with serious side effects, including serious allergic reactions including skin rash, swelling of the face and mouth, trouble breathing, and chest tightness. It may also increase patient susceptibility to infection. It is approved with a Medication Guide for patients, explaining the potential risks associated with treatment.
Tildrakizumab will be marketed as Ilumya.
Sun Pharma is working with the FDA on postapproval commitments, and once that has been completed, they will have a better idea of when it will become available, according to a spokesperson for the manufacturer. The cost is not yet available.
PASI responses with biologics similar among white, nonwhite individuals, study finds
MIAMI – Skin clearance rates among people with moderate to severe plaque psoriasis treated with brodalumab were superior to clearance rates among those treated with ustekinumab in a study that also provided comparisons between white and nonwhite patients.
In the study, presented in a poster at the 2018 Orlando Dermatology Aesthetic and Clinical Conference, there were no significant difference in overall efficacy, safety, or health-related quality of life outcomes between white and nonwhite patients treated with either biologic.
Additional analyses specific to patients with skin of color can be beneficial, Amy McMichael, MD, one of the investigators, said in an interview. “Patients with skin of color experience differences in psoriasis-related symptoms,” noted Dr. McMichael, chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C. “Greater degrees of skin involvement have been shown in African-American patients, as have differences in erythema, scaling, dyspigmentation, and plaque thickness.”
She and her colleagues evaluated 1,849 participants in phase 3 brodalumab clinical trials, which included ustekinumab-treated patients as a comparison group. Approximately 10% of the AMAGINE-2 and AMAGINE-3 study populations were skin of color participants. The results reported at the meeting were from their ad hoc study of 12-week induction findings from the 52-week clinical trials.
At week 12, 70% of white and 63% of nonwhite participants treated with ustekinumab achieved a Psoriasis Area and Severity Index (PASI) 75. At the same time, 86% of white and 88% of nonwhite patients treated with brodalumab achieved the same outcome. Similarly, PASI 90 and PASI 100 scores did not differ significantly between the 1,667 white and 182 skin of color participants.
The two biologics act on different aspects of the molecular pathway involved in psoriasis. Brodalumab (Siliq) specifically blocks the interleukin-17 receptor, whereas other biologics used to treat psoriasis, including ustekinumab (Stelara), an IL-12 and -23 antagonist, target upstream molecules in the inflammatory pathway. Dr. McMichael said, “The superior skin clearance rates seen in patients treated with brodalumab may be due to its target being a receptor as opposed to a ligand.”
Treatment-emergent adverse event rates were similar between the white and nonwhite patients. Treatment-emergent adverse events were reported in 58% and 57% of the white brodalumab and ustekinumab groups, respectively, and in 53% and 47% of the nonwhite brodalumab and ustekinumab groups, respectively. Serious adverse events occurred in 1.2% and 1.1% of the white brodalumab and ustekinumab cohorts, respectively, and in 1.7% and 0% of the nonwhite participants, respectively.
The investigators also assessed health-related quality of life and again found outcomes were similar between white and nonwhite participants. For example, among those treated with brodalumab, 80% of white and 78% of nonwhite patients achieved a score improvement of 5 or greater on the Dermatology Life Quality Index. Of those randomized to ustekinumab, 76% of white patients and 73% of nonwhite patients achieved the same outcome.
“We plan to perform a further analysis evaluating the Dr. McMichael said. Additionally, a population-based study to investigate treatment patterns in patients with psoriasis across racial and socioeconomic groups could also shed light on how patients with skin of color manage their psoriasis, she added.
Dr. McMichael’s disclosures include having been an investigator for Allergan, Incyte, and Samumed and a consultant to Aclaris, Galderma, IntraDerm, Johnson & Johnson, Merz, Pfizer, and Procter & Gamble.
SOURCE: McMichael A et al. ODAC 2018.
MIAMI – Skin clearance rates among people with moderate to severe plaque psoriasis treated with brodalumab were superior to clearance rates among those treated with ustekinumab in a study that also provided comparisons between white and nonwhite patients.
In the study, presented in a poster at the 2018 Orlando Dermatology Aesthetic and Clinical Conference, there were no significant difference in overall efficacy, safety, or health-related quality of life outcomes between white and nonwhite patients treated with either biologic.
Additional analyses specific to patients with skin of color can be beneficial, Amy McMichael, MD, one of the investigators, said in an interview. “Patients with skin of color experience differences in psoriasis-related symptoms,” noted Dr. McMichael, chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C. “Greater degrees of skin involvement have been shown in African-American patients, as have differences in erythema, scaling, dyspigmentation, and plaque thickness.”
She and her colleagues evaluated 1,849 participants in phase 3 brodalumab clinical trials, which included ustekinumab-treated patients as a comparison group. Approximately 10% of the AMAGINE-2 and AMAGINE-3 study populations were skin of color participants. The results reported at the meeting were from their ad hoc study of 12-week induction findings from the 52-week clinical trials.
At week 12, 70% of white and 63% of nonwhite participants treated with ustekinumab achieved a Psoriasis Area and Severity Index (PASI) 75. At the same time, 86% of white and 88% of nonwhite patients treated with brodalumab achieved the same outcome. Similarly, PASI 90 and PASI 100 scores did not differ significantly between the 1,667 white and 182 skin of color participants.
The two biologics act on different aspects of the molecular pathway involved in psoriasis. Brodalumab (Siliq) specifically blocks the interleukin-17 receptor, whereas other biologics used to treat psoriasis, including ustekinumab (Stelara), an IL-12 and -23 antagonist, target upstream molecules in the inflammatory pathway. Dr. McMichael said, “The superior skin clearance rates seen in patients treated with brodalumab may be due to its target being a receptor as opposed to a ligand.”
Treatment-emergent adverse event rates were similar between the white and nonwhite patients. Treatment-emergent adverse events were reported in 58% and 57% of the white brodalumab and ustekinumab groups, respectively, and in 53% and 47% of the nonwhite brodalumab and ustekinumab groups, respectively. Serious adverse events occurred in 1.2% and 1.1% of the white brodalumab and ustekinumab cohorts, respectively, and in 1.7% and 0% of the nonwhite participants, respectively.
The investigators also assessed health-related quality of life and again found outcomes were similar between white and nonwhite participants. For example, among those treated with brodalumab, 80% of white and 78% of nonwhite patients achieved a score improvement of 5 or greater on the Dermatology Life Quality Index. Of those randomized to ustekinumab, 76% of white patients and 73% of nonwhite patients achieved the same outcome.
“We plan to perform a further analysis evaluating the Dr. McMichael said. Additionally, a population-based study to investigate treatment patterns in patients with psoriasis across racial and socioeconomic groups could also shed light on how patients with skin of color manage their psoriasis, she added.
Dr. McMichael’s disclosures include having been an investigator for Allergan, Incyte, and Samumed and a consultant to Aclaris, Galderma, IntraDerm, Johnson & Johnson, Merz, Pfizer, and Procter & Gamble.
SOURCE: McMichael A et al. ODAC 2018.
MIAMI – Skin clearance rates among people with moderate to severe plaque psoriasis treated with brodalumab were superior to clearance rates among those treated with ustekinumab in a study that also provided comparisons between white and nonwhite patients.
In the study, presented in a poster at the 2018 Orlando Dermatology Aesthetic and Clinical Conference, there were no significant difference in overall efficacy, safety, or health-related quality of life outcomes between white and nonwhite patients treated with either biologic.
Additional analyses specific to patients with skin of color can be beneficial, Amy McMichael, MD, one of the investigators, said in an interview. “Patients with skin of color experience differences in psoriasis-related symptoms,” noted Dr. McMichael, chair of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C. “Greater degrees of skin involvement have been shown in African-American patients, as have differences in erythema, scaling, dyspigmentation, and plaque thickness.”
She and her colleagues evaluated 1,849 participants in phase 3 brodalumab clinical trials, which included ustekinumab-treated patients as a comparison group. Approximately 10% of the AMAGINE-2 and AMAGINE-3 study populations were skin of color participants. The results reported at the meeting were from their ad hoc study of 12-week induction findings from the 52-week clinical trials.
At week 12, 70% of white and 63% of nonwhite participants treated with ustekinumab achieved a Psoriasis Area and Severity Index (PASI) 75. At the same time, 86% of white and 88% of nonwhite patients treated with brodalumab achieved the same outcome. Similarly, PASI 90 and PASI 100 scores did not differ significantly between the 1,667 white and 182 skin of color participants.
The two biologics act on different aspects of the molecular pathway involved in psoriasis. Brodalumab (Siliq) specifically blocks the interleukin-17 receptor, whereas other biologics used to treat psoriasis, including ustekinumab (Stelara), an IL-12 and -23 antagonist, target upstream molecules in the inflammatory pathway. Dr. McMichael said, “The superior skin clearance rates seen in patients treated with brodalumab may be due to its target being a receptor as opposed to a ligand.”
Treatment-emergent adverse event rates were similar between the white and nonwhite patients. Treatment-emergent adverse events were reported in 58% and 57% of the white brodalumab and ustekinumab groups, respectively, and in 53% and 47% of the nonwhite brodalumab and ustekinumab groups, respectively. Serious adverse events occurred in 1.2% and 1.1% of the white brodalumab and ustekinumab cohorts, respectively, and in 1.7% and 0% of the nonwhite participants, respectively.
The investigators also assessed health-related quality of life and again found outcomes were similar between white and nonwhite participants. For example, among those treated with brodalumab, 80% of white and 78% of nonwhite patients achieved a score improvement of 5 or greater on the Dermatology Life Quality Index. Of those randomized to ustekinumab, 76% of white patients and 73% of nonwhite patients achieved the same outcome.
“We plan to perform a further analysis evaluating the Dr. McMichael said. Additionally, a population-based study to investigate treatment patterns in patients with psoriasis across racial and socioeconomic groups could also shed light on how patients with skin of color manage their psoriasis, she added.
Dr. McMichael’s disclosures include having been an investigator for Allergan, Incyte, and Samumed and a consultant to Aclaris, Galderma, IntraDerm, Johnson & Johnson, Merz, Pfizer, and Procter & Gamble.
SOURCE: McMichael A et al. ODAC 2018.
REPORTING FROM ODAC 2018
Key clinical point: Responses to brodalumab and ustekinumab were comparable in nonwhite and white patients with psoriasis.
Major finding: At week 12, 70% of white and 63% of nonwhite participants treated with ustekinumab achieved PASI 75, a nonsignificant difference.
Study details: An ad hoc comparison of week 12 phase 3 study data in 1,849 patients including 182 patients with skin of color.
Disclosures: Dr. McMichael’s disclosures include having been an investigator for Allergan, Incyte, and Samumed; and a consultant to Aclaris, Galderma, IntraDerm, Johnson & Johnson, Merz, Pfizer, and Procter & Gamble.
Source: McMichael A et al. ODAC 2018.
Study highlights need to investigate psoriasis treatment outcomes in skin of color patients
MIAMI – Psoriasis often presents differently in skin of color patients, but an unanswered question remains: Does response to treatment with an agent like a fixed-dose combination foam also differ by ethnicity?
Researchers at Mount Sinai St. Luke’s Hospital in New York addressed this question using phase 2 and 3 study data for 1,104 people with psoriasis, about half of whom were randomized to topical treatment with calcipotriene and betamethasone dipropionate foam 0.005%/0.064% (Enstilar); the rest received a single component or vehicle only. The data were obtained from LEO Pharma, the product’s manufacturer.
“We were very interested in knowing if there was any difference in efficacy between the specific ethnic groups – the skin of color and non–skin of color patients,” said Bridget Kaufman, MD, a dermatopharmacology fellow at Mount Sinai St. Luke’s Hospital. “So we went back to look at the data to see if there was any difference in side effects or efficacy between ethnic groups.”
Strength in numbers?
The three randomized, pooled clinical studies included many ethnic groups. However, only 6.5% of participants were black and even fewer were Asian, American Indian, or native Hawaiian, Dr. Kaufman said. “It’s hard to see meaningful differences when you don’t have a substantial skin of color population.”
As a result, no significant associations emerged from the pooled data. “That is the main take-home message of this study: We don’t have a great understanding now of the difference in efficacy between white and nonwhite ethnic groups,” Dr. Kaufman said.
The researchers defined treatment success at 4 weeks as a two-point improvement to “clear” or “almost clear” on the Investigator Global Assessment of psoriasis. Of the adult participants with chronic plaque psoriasis randomized to the combination foam product, 54% of the white patients; 30% of black patients; 69% of Asian patients; and one of the two Hawaiian/Pacific Islander patients achieved treatment success after 4 weeks of topical treatment.
“All subgroups analyzed had a good response to treatment at 4 weeks. Numerically it appears African Americans in particular did not do quite as well, but we can’t [definitively] draw that conclusion,” Dr. Kaufman said in an interview.
More data, please
The study is just the first step in investigating the efficacy of this particular product in diverse ethnic groups, Dr. Kaufman added. “That really emphasizes the importance of studying these medications in skin of color populations in particular.”
More guidance on psoriasis in skin of color patients is available in a published review article by Dr. Kaufman and Andrew F. Alexis, MD, director of the Skin of Color Center at Mount Sinai St. Luke’s and Mount Sinai West, New York (Am J Clin Dermatol. 2017 Dec 5. doi: 10.1007/s40257-017-0332-7).
Enstilar manufacturer LEO Pharma supplied the clinical data but did not fund the study. Dr. Kaufman had no relevant financial disclosures.
dermnews@frontlinemedcom.com
SOURCE: Kaufman B et al. ODAC 2018
MIAMI – Psoriasis often presents differently in skin of color patients, but an unanswered question remains: Does response to treatment with an agent like a fixed-dose combination foam also differ by ethnicity?
Researchers at Mount Sinai St. Luke’s Hospital in New York addressed this question using phase 2 and 3 study data for 1,104 people with psoriasis, about half of whom were randomized to topical treatment with calcipotriene and betamethasone dipropionate foam 0.005%/0.064% (Enstilar); the rest received a single component or vehicle only. The data were obtained from LEO Pharma, the product’s manufacturer.
“We were very interested in knowing if there was any difference in efficacy between the specific ethnic groups – the skin of color and non–skin of color patients,” said Bridget Kaufman, MD, a dermatopharmacology fellow at Mount Sinai St. Luke’s Hospital. “So we went back to look at the data to see if there was any difference in side effects or efficacy between ethnic groups.”
Strength in numbers?
The three randomized, pooled clinical studies included many ethnic groups. However, only 6.5% of participants were black and even fewer were Asian, American Indian, or native Hawaiian, Dr. Kaufman said. “It’s hard to see meaningful differences when you don’t have a substantial skin of color population.”
As a result, no significant associations emerged from the pooled data. “That is the main take-home message of this study: We don’t have a great understanding now of the difference in efficacy between white and nonwhite ethnic groups,” Dr. Kaufman said.
The researchers defined treatment success at 4 weeks as a two-point improvement to “clear” or “almost clear” on the Investigator Global Assessment of psoriasis. Of the adult participants with chronic plaque psoriasis randomized to the combination foam product, 54% of the white patients; 30% of black patients; 69% of Asian patients; and one of the two Hawaiian/Pacific Islander patients achieved treatment success after 4 weeks of topical treatment.
“All subgroups analyzed had a good response to treatment at 4 weeks. Numerically it appears African Americans in particular did not do quite as well, but we can’t [definitively] draw that conclusion,” Dr. Kaufman said in an interview.
More data, please
The study is just the first step in investigating the efficacy of this particular product in diverse ethnic groups, Dr. Kaufman added. “That really emphasizes the importance of studying these medications in skin of color populations in particular.”
More guidance on psoriasis in skin of color patients is available in a published review article by Dr. Kaufman and Andrew F. Alexis, MD, director of the Skin of Color Center at Mount Sinai St. Luke’s and Mount Sinai West, New York (Am J Clin Dermatol. 2017 Dec 5. doi: 10.1007/s40257-017-0332-7).
Enstilar manufacturer LEO Pharma supplied the clinical data but did not fund the study. Dr. Kaufman had no relevant financial disclosures.
dermnews@frontlinemedcom.com
SOURCE: Kaufman B et al. ODAC 2018
MIAMI – Psoriasis often presents differently in skin of color patients, but an unanswered question remains: Does response to treatment with an agent like a fixed-dose combination foam also differ by ethnicity?
Researchers at Mount Sinai St. Luke’s Hospital in New York addressed this question using phase 2 and 3 study data for 1,104 people with psoriasis, about half of whom were randomized to topical treatment with calcipotriene and betamethasone dipropionate foam 0.005%/0.064% (Enstilar); the rest received a single component or vehicle only. The data were obtained from LEO Pharma, the product’s manufacturer.
“We were very interested in knowing if there was any difference in efficacy between the specific ethnic groups – the skin of color and non–skin of color patients,” said Bridget Kaufman, MD, a dermatopharmacology fellow at Mount Sinai St. Luke’s Hospital. “So we went back to look at the data to see if there was any difference in side effects or efficacy between ethnic groups.”
Strength in numbers?
The three randomized, pooled clinical studies included many ethnic groups. However, only 6.5% of participants were black and even fewer were Asian, American Indian, or native Hawaiian, Dr. Kaufman said. “It’s hard to see meaningful differences when you don’t have a substantial skin of color population.”
As a result, no significant associations emerged from the pooled data. “That is the main take-home message of this study: We don’t have a great understanding now of the difference in efficacy between white and nonwhite ethnic groups,” Dr. Kaufman said.
The researchers defined treatment success at 4 weeks as a two-point improvement to “clear” or “almost clear” on the Investigator Global Assessment of psoriasis. Of the adult participants with chronic plaque psoriasis randomized to the combination foam product, 54% of the white patients; 30% of black patients; 69% of Asian patients; and one of the two Hawaiian/Pacific Islander patients achieved treatment success after 4 weeks of topical treatment.
“All subgroups analyzed had a good response to treatment at 4 weeks. Numerically it appears African Americans in particular did not do quite as well, but we can’t [definitively] draw that conclusion,” Dr. Kaufman said in an interview.
More data, please
The study is just the first step in investigating the efficacy of this particular product in diverse ethnic groups, Dr. Kaufman added. “That really emphasizes the importance of studying these medications in skin of color populations in particular.”
More guidance on psoriasis in skin of color patients is available in a published review article by Dr. Kaufman and Andrew F. Alexis, MD, director of the Skin of Color Center at Mount Sinai St. Luke’s and Mount Sinai West, New York (Am J Clin Dermatol. 2017 Dec 5. doi: 10.1007/s40257-017-0332-7).
Enstilar manufacturer LEO Pharma supplied the clinical data but did not fund the study. Dr. Kaufman had no relevant financial disclosures.
dermnews@frontlinemedcom.com
SOURCE: Kaufman B et al. ODAC 2018
REPORTING FROM ODAC 2018
Key clinical point: There were no significant differences in response to calcipotriene and betamethasone dipropionate foam in phase 2 and 3 trials, possibly because of small percentages of skin of color participants in these studies.
Major finding: Treatment success rates at 4 weeks were 30% among black patients, 54% among white patients, and 69% among Asian patients, but not enough skin of color patients were enrolled for differences to reach statistical significance.
Study details: A retrospective analysis of pooled phase 2 and phase 3 studies with 1,104 participants with psoriasis.
Disclosures: LEO Pharma supplied the clinical data but did not fund the study. Dr. Kaufman had no relevant financial disclosures.
Source: Kaufman B et al. ODAC 2018.
Xeljanz: FDA panel recommends ulcerative colitis indication
SILVER SPRING, MD. – Federal advisors to the Food and Drug Administration on March 8 voted unanimously to recommend approval of an additional indication for tofacitinib (Xeljanz), this time for ulcerative colitis.
Members of the Gastrointestinal Drugs Advisory Committee unanimously voted to recommend two different dosing regimens: 10 mg twice daily for 16 weeks in patients who have not experienced a therapeutic benefit after 8 weeks of treatment, as well as 10 mg twice daily for patients who have an inadequate or loss of response to TNF-blocker therapy, based on the results of several phase 3 clinical trials.
The recommended ulcerative colitis (UC) indication was based on the OCTAVE trials (N Engl J Med 2017;376:1723-36), including a phase 2 study, two identical phase 3 induction trials (OCTAVE Induction 1 and OCTAVE Induction 2), a 53-week, phase 3 maintenance trial (OCTAVE Sustain), and an open-label extension study.
The induction trials enrolled a total of 1,139 patients with moderate to severe UC. Patients in both studies were administered tofacitinib 10 mg twice daily or placebo and were assessed after 8 weeks to judge clinical response. Patients in both studies displayed notable remission rates (18.5% and 16.6%), compared with placebo, according to Eric Maller, MD, executive director of the UC development program at Pfizer.*
Patients who did not achieve remission, but showed some clinical response (decrease in Mayo score of at least 3 points), were then enrolled in the 53-week OCTAVE Sustain, where they were randomized to receive tofacitinib 10 mg twice daily, 5 mg twice daily, or placebo.
During maintenance treatment, both 5 mg and 10 mg doses demonstrated substantial treatment benefits, with 32.4% and 41.0% of patients achieving remission, an increase of 22.0% and 30.7%, compared with placebo, respectively.
As part of the maintenance study, Pfizer analyzed patients with or without prior TNF-blocker failure. This analysis revealed that patients who had previously failed TNF-blocker therapy experienced a greater treatment benefit than those who had not. While the benefit was noticeable in both dosage groups, patients taking the 10-mg dose experienced the greatest benefit, with 70% increase in remission rates, 39% increase in mucosal healing, and 75% increase in steroid-free remission among baseline remitters, compared with patients in the 5-mg group, Dr. Maller said.
Researchers also looked at a subgroup of 295 patients as part of an open-label extension study who had no clinical response to tofacitinib 10 mg twice daily after 8 weeks and subsequently treated them for an additional 8 weeks. After the additional 8 weeks of treatment, over half (51.2%) displayed clinical responses and 8.6% were in remission.
“This is a desperate patient population. These are impressive results,” stated Darrell Pardi, MD, vice chair of the advisory committee and professor of medicine at the Mayo Clinic, Rochester, Minn.
Serious adverse events were seen in 4% of tofacitinib-treated patients in the induction trials, compared with 6% of placebo-treated, according to Lesley Hanes, MD, medical officer with the FDA Center for Drug Evaluation and Research.
Adverse events appeared to be dose dependent, with risk of deaths and malignancies (excluding nonmelanoma skin cancer), opportunistic infections, herpes zoster infection (HZ), “possible” drug-induced liver injury, and cardiovascular and thromboembolic events more common with the 10-mg dose, Dr. Hanes said.
“I think the safety concerns, though, they are dose dependent, the difference between the 5 [mg] and 10 [mg] were not large,” according to Dr. Pardi. “Several of these are mitigatable by dermatologic exam or, hopefully, a vaccine.”
Several of the advisory committee members submitted conflict of interest waivers. Chair and vice chair Jean-Pierre Raufman, MD, and Darrell Pardi, MD, disclosed funding from competing pharmaceutical manufacturers.
*This article was updated on March 12, 2018.
SILVER SPRING, MD. – Federal advisors to the Food and Drug Administration on March 8 voted unanimously to recommend approval of an additional indication for tofacitinib (Xeljanz), this time for ulcerative colitis.
Members of the Gastrointestinal Drugs Advisory Committee unanimously voted to recommend two different dosing regimens: 10 mg twice daily for 16 weeks in patients who have not experienced a therapeutic benefit after 8 weeks of treatment, as well as 10 mg twice daily for patients who have an inadequate or loss of response to TNF-blocker therapy, based on the results of several phase 3 clinical trials.
The recommended ulcerative colitis (UC) indication was based on the OCTAVE trials (N Engl J Med 2017;376:1723-36), including a phase 2 study, two identical phase 3 induction trials (OCTAVE Induction 1 and OCTAVE Induction 2), a 53-week, phase 3 maintenance trial (OCTAVE Sustain), and an open-label extension study.
The induction trials enrolled a total of 1,139 patients with moderate to severe UC. Patients in both studies were administered tofacitinib 10 mg twice daily or placebo and were assessed after 8 weeks to judge clinical response. Patients in both studies displayed notable remission rates (18.5% and 16.6%), compared with placebo, according to Eric Maller, MD, executive director of the UC development program at Pfizer.*
Patients who did not achieve remission, but showed some clinical response (decrease in Mayo score of at least 3 points), were then enrolled in the 53-week OCTAVE Sustain, where they were randomized to receive tofacitinib 10 mg twice daily, 5 mg twice daily, or placebo.
During maintenance treatment, both 5 mg and 10 mg doses demonstrated substantial treatment benefits, with 32.4% and 41.0% of patients achieving remission, an increase of 22.0% and 30.7%, compared with placebo, respectively.
As part of the maintenance study, Pfizer analyzed patients with or without prior TNF-blocker failure. This analysis revealed that patients who had previously failed TNF-blocker therapy experienced a greater treatment benefit than those who had not. While the benefit was noticeable in both dosage groups, patients taking the 10-mg dose experienced the greatest benefit, with 70% increase in remission rates, 39% increase in mucosal healing, and 75% increase in steroid-free remission among baseline remitters, compared with patients in the 5-mg group, Dr. Maller said.
Researchers also looked at a subgroup of 295 patients as part of an open-label extension study who had no clinical response to tofacitinib 10 mg twice daily after 8 weeks and subsequently treated them for an additional 8 weeks. After the additional 8 weeks of treatment, over half (51.2%) displayed clinical responses and 8.6% were in remission.
“This is a desperate patient population. These are impressive results,” stated Darrell Pardi, MD, vice chair of the advisory committee and professor of medicine at the Mayo Clinic, Rochester, Minn.
Serious adverse events were seen in 4% of tofacitinib-treated patients in the induction trials, compared with 6% of placebo-treated, according to Lesley Hanes, MD, medical officer with the FDA Center for Drug Evaluation and Research.
Adverse events appeared to be dose dependent, with risk of deaths and malignancies (excluding nonmelanoma skin cancer), opportunistic infections, herpes zoster infection (HZ), “possible” drug-induced liver injury, and cardiovascular and thromboembolic events more common with the 10-mg dose, Dr. Hanes said.
“I think the safety concerns, though, they are dose dependent, the difference between the 5 [mg] and 10 [mg] were not large,” according to Dr. Pardi. “Several of these are mitigatable by dermatologic exam or, hopefully, a vaccine.”
Several of the advisory committee members submitted conflict of interest waivers. Chair and vice chair Jean-Pierre Raufman, MD, and Darrell Pardi, MD, disclosed funding from competing pharmaceutical manufacturers.
*This article was updated on March 12, 2018.
SILVER SPRING, MD. – Federal advisors to the Food and Drug Administration on March 8 voted unanimously to recommend approval of an additional indication for tofacitinib (Xeljanz), this time for ulcerative colitis.
Members of the Gastrointestinal Drugs Advisory Committee unanimously voted to recommend two different dosing regimens: 10 mg twice daily for 16 weeks in patients who have not experienced a therapeutic benefit after 8 weeks of treatment, as well as 10 mg twice daily for patients who have an inadequate or loss of response to TNF-blocker therapy, based on the results of several phase 3 clinical trials.
The recommended ulcerative colitis (UC) indication was based on the OCTAVE trials (N Engl J Med 2017;376:1723-36), including a phase 2 study, two identical phase 3 induction trials (OCTAVE Induction 1 and OCTAVE Induction 2), a 53-week, phase 3 maintenance trial (OCTAVE Sustain), and an open-label extension study.
The induction trials enrolled a total of 1,139 patients with moderate to severe UC. Patients in both studies were administered tofacitinib 10 mg twice daily or placebo and were assessed after 8 weeks to judge clinical response. Patients in both studies displayed notable remission rates (18.5% and 16.6%), compared with placebo, according to Eric Maller, MD, executive director of the UC development program at Pfizer.*
Patients who did not achieve remission, but showed some clinical response (decrease in Mayo score of at least 3 points), were then enrolled in the 53-week OCTAVE Sustain, where they were randomized to receive tofacitinib 10 mg twice daily, 5 mg twice daily, or placebo.
During maintenance treatment, both 5 mg and 10 mg doses demonstrated substantial treatment benefits, with 32.4% and 41.0% of patients achieving remission, an increase of 22.0% and 30.7%, compared with placebo, respectively.
As part of the maintenance study, Pfizer analyzed patients with or without prior TNF-blocker failure. This analysis revealed that patients who had previously failed TNF-blocker therapy experienced a greater treatment benefit than those who had not. While the benefit was noticeable in both dosage groups, patients taking the 10-mg dose experienced the greatest benefit, with 70% increase in remission rates, 39% increase in mucosal healing, and 75% increase in steroid-free remission among baseline remitters, compared with patients in the 5-mg group, Dr. Maller said.
Researchers also looked at a subgroup of 295 patients as part of an open-label extension study who had no clinical response to tofacitinib 10 mg twice daily after 8 weeks and subsequently treated them for an additional 8 weeks. After the additional 8 weeks of treatment, over half (51.2%) displayed clinical responses and 8.6% were in remission.
“This is a desperate patient population. These are impressive results,” stated Darrell Pardi, MD, vice chair of the advisory committee and professor of medicine at the Mayo Clinic, Rochester, Minn.
Serious adverse events were seen in 4% of tofacitinib-treated patients in the induction trials, compared with 6% of placebo-treated, according to Lesley Hanes, MD, medical officer with the FDA Center for Drug Evaluation and Research.
Adverse events appeared to be dose dependent, with risk of deaths and malignancies (excluding nonmelanoma skin cancer), opportunistic infections, herpes zoster infection (HZ), “possible” drug-induced liver injury, and cardiovascular and thromboembolic events more common with the 10-mg dose, Dr. Hanes said.
“I think the safety concerns, though, they are dose dependent, the difference between the 5 [mg] and 10 [mg] were not large,” according to Dr. Pardi. “Several of these are mitigatable by dermatologic exam or, hopefully, a vaccine.”
Several of the advisory committee members submitted conflict of interest waivers. Chair and vice chair Jean-Pierre Raufman, MD, and Darrell Pardi, MD, disclosed funding from competing pharmaceutical manufacturers.
*This article was updated on March 12, 2018.
REPORTING FROM AN FDA ADVISORY COMMITTEE MEETING
Pediatric Psoriasis: An Interview With Nanette B. Silverberg, MD
What causes psoriasis in children?
Psoriasis is a chronic immune-mediated inflammatory skin disease with a genetic predisposition (Eichenfield et al). Similar to many inflammatory skin diseases, school-aged children have a greater predisposition before or in early adolescence. As with adult disease, pediatric psoriasis has a complex pathogenesis largely related to aberrant immune response to triggers such as infections (eg, streptococcal pharyngitis, perianal streptococcal dermatitis, upper respiratory viral infections), trauma (ie, Koebner phenomenon), stress, and obesity.
What are the emerging data and recommendations on screening for comorbidities in children with psoriasis?
Similar to psoriasis in adults, obesity and the metabolic syndrome are a true association with pediatric psoriasis that has been discussed in the literature (Eichenfield et al). Although many children with psoriasis have obesity as a potential comorbidity, the risk of cardiovascular comorbidities independent of obesity is high in pediatric psoriasis including elevated lipids, hypertension, polycystic ovaries, nonalcoholic liver disease, and elevated liver enzymes (Tollefson et al). Children with psoriasis have greater central obesity and adiposity, often accompanied by a family history of obesity. Interventions in this direction may be needed for long-term disease control and general health (Mercy and Paller). One target population is hospitalized children with psoriasis, particularly black and Hispanic children aged 0 to 9 years. This population has been identified to have a greater risk for obesity, diabetes mellitus, hypertension, arrhythmia, and valvular heart disease (Kwa et al). Therefore, it can be said that dermatologists can help to improve the overall health and lifestyle long-term in children with psoriasis.
Early-onset disease also is associated with greater risk for lifetime quality-of-life impairments including poor lifetime dermatology life quality index scores, depression and psoriasis-induced depression, social discrimination, sleep problems, and recreational drug usage (Kim et al).
How does psoriasis in children differ from adults?
Children have a variety of features that differ from adult disease. First, they are more likely to have an infectious trigger and therefore may have an identifiable treatable source. Second, they are more likely to have a family history of disease, with one-third having a relative with psoriasis, therefore, identifying the child at risk for long-standing disease. Third, children have far more visible head and neck disease, especially facial involvement including eyelids (Raychaudhuri and Gross), which increases the risk of bullying, social stigma, and negative effects on self-image. Of course, site is affected by age, and in infancy diaper dermatitis and inverse disease with maceration and overlying candidal diaper dermatitis can occur. Although children have less joint disease, it can be dramatic and crippling to the developing child.
What treatments are available for children?
In childhood, identification of precipitating infections such as streptococcal infection is ideal with appropriate intervention thereafter. Topical therapies are appropriate for limited disease with minimal disability; however, phototherapy and systemic agents can be used in pediatric psoriasis in extensive cases. Topical therapies can include corticosteroids, calcineurin inhibitors often used in sensitive skin such as the face and intertriginous areas, and calcipotriene (Eichenfield et al). Additional agents such as tar and salicylic acid can be used, with limitations on the latter due to risk for absorption in smaller children. Systemic interventions often are introduced after years of disease. A recent study identified practitioners with special interest in pediatric psoriasis and determined that systemic interventions were on average introduced 3 years after psoriasis was diagnosed and most commonly included methotrexate followed by etanercept, the latter having fewer gastrointestinal tract side effects. The panel found that usage of folic acid 6 days weekly minimized gastrointestinal tract side effects with methotrexate. Acitretin and cyclosporine were alternatives (Bronckers et al; Psoriasis Investigator Group [PsIG] of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis [EWGPP]).
Recently, dermatologists have become aware of the dramatic benefits of immune response modifiers and some biologics on pediatric psoriasis. In the setting of joint and skin involvement, I allow the rheumatologist to make the choice of agents for the child's best outcome. However, for pediatric and adolescent psoriasis, we now have 2 US Food and Drug Administration-approved agents and more rapid and thorough testing of adult-approved agents in children, with a hope of greater ability to modify disease course at a younger age, both now and in the future.
Which biologics are approved for the pediatric patient population?
Currently, in the United States 2 biologics have been approved: (1) etanercept, a fusion protein of tumor necrosis factor receptor extracellular domain linked to the Fc portion of human IgG, for moderate to severe plaque psoriasis in patients 4 years and older, and (2) ustekinumab, a human IgG1κ monoclonal antibody against the shared p40 subunit of the IL-12 and IL-23 cytokines, for moderate to severe plaque psoriasis in patients 12 years and older based on the encouraging data of the CADMUS trial (Kellen et al; Landells et al). In Europe, adalimumab has been approved as a first-line therapy in pediatric psoriasis (age ≥4 years), and etanercept (age ≥6 years) and ustekinumab (age ≥12 years) have been approved as second-line agents, all with grade A evidence, according to a recent Italian panel (Fortina et al). (A thorough review of the guidelines on screening, administration, and vaccination is available from Eichenfield et al.)
What treatments are in the pipeline?
In the United States we have clinical trials ongoing of adult-approved topical and immune response-modifying agents such as apremilast. These agents, as they become available and the data are gathered, will be added to what I refer to as our "pharmamentarium" of agents we can use to combat a difficult and disabling illness.
What gaps are there in the pediatric psoriasis research?
Currently, there is poor awareness that there is research for pediatric psoriasis, and there is a need for pediatric groups and the National Psoriasis Foundation to allow children, adolescents, and their families to know that clinical trials are available looking into newer, more targeted, and less immunosuppressive agents. There is new hope on the horizon!
Suggested Readings
Bronckers IMGJ, Seyger MMB, West DP, et al; Psoriasis Investigator Group (PsIG) of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis (EWGPP). Safety of systemic agents for the treatment of pediatric psoriasis. JAMA Dermatol. 2017;153:1147-1157.
Eichenfield LF, Paller AS, Tom WL, et al. Pediatric psoriasis: evolving perspectives [published online January 4, 2018]. Pediatr Dermatol. doi:10.1111/pde.13382.
Fortina AB, Bardazzi F, Berti S, et al. Treatment of severe psoriasis in children: recommendations of an Italian expert group [published online August 23, 2017]. Eur J Pediatr. 2017;176:1339-1354.
Kellen R, Silverberg NB, Lebwohl M. Efficacy and safety of ustekinumab in adolescents. Pediatric Health Med Ther. 2016;7:109-120.
Kim GE, Seidler E, Kimball AB. Effect of age at diagnosis on chronic quality of life and long-term outcomes of individuals with psoriasis [published online December 29, 2014]. Pediatr Dermatol. 2015;32:656-662.
Kwa L, Kwa MC, Silverberg JI. Cardiovascular comorbidities of pediatric psoriasis among hospitalized children in the United States. J Am Acad Dermatol. 2017;77:1023-1029.
Landells I, Marano C, Hsu MC, et al. Ustekinumab in adolescent patients age 12 to 17 years with moderate-to-severe plaque psoriasis: results of the randomized phase 3 CADMUS study [published online August 7, 2015]. J Am Acad Dermatol. 2015;73:594-603.
Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
Raychaudhuri SP, Gross J. A comparative study of pediatric onset psoriasis with adult onset psoriasis. Pediatr Dermatol. 2000;17:174-178.
Tollefson MM, Van Houten HK, Asante D, et al. Association of psoriasis with comorbidity development in children with psoriasis [published online January 10, 2018]. JAMA Dermatol. doi:10.1001/jamadermatol.2017.5417.
What causes psoriasis in children?
Psoriasis is a chronic immune-mediated inflammatory skin disease with a genetic predisposition (Eichenfield et al). Similar to many inflammatory skin diseases, school-aged children have a greater predisposition before or in early adolescence. As with adult disease, pediatric psoriasis has a complex pathogenesis largely related to aberrant immune response to triggers such as infections (eg, streptococcal pharyngitis, perianal streptococcal dermatitis, upper respiratory viral infections), trauma (ie, Koebner phenomenon), stress, and obesity.
What are the emerging data and recommendations on screening for comorbidities in children with psoriasis?
Similar to psoriasis in adults, obesity and the metabolic syndrome are a true association with pediatric psoriasis that has been discussed in the literature (Eichenfield et al). Although many children with psoriasis have obesity as a potential comorbidity, the risk of cardiovascular comorbidities independent of obesity is high in pediatric psoriasis including elevated lipids, hypertension, polycystic ovaries, nonalcoholic liver disease, and elevated liver enzymes (Tollefson et al). Children with psoriasis have greater central obesity and adiposity, often accompanied by a family history of obesity. Interventions in this direction may be needed for long-term disease control and general health (Mercy and Paller). One target population is hospitalized children with psoriasis, particularly black and Hispanic children aged 0 to 9 years. This population has been identified to have a greater risk for obesity, diabetes mellitus, hypertension, arrhythmia, and valvular heart disease (Kwa et al). Therefore, it can be said that dermatologists can help to improve the overall health and lifestyle long-term in children with psoriasis.
Early-onset disease also is associated with greater risk for lifetime quality-of-life impairments including poor lifetime dermatology life quality index scores, depression and psoriasis-induced depression, social discrimination, sleep problems, and recreational drug usage (Kim et al).
How does psoriasis in children differ from adults?
Children have a variety of features that differ from adult disease. First, they are more likely to have an infectious trigger and therefore may have an identifiable treatable source. Second, they are more likely to have a family history of disease, with one-third having a relative with psoriasis, therefore, identifying the child at risk for long-standing disease. Third, children have far more visible head and neck disease, especially facial involvement including eyelids (Raychaudhuri and Gross), which increases the risk of bullying, social stigma, and negative effects on self-image. Of course, site is affected by age, and in infancy diaper dermatitis and inverse disease with maceration and overlying candidal diaper dermatitis can occur. Although children have less joint disease, it can be dramatic and crippling to the developing child.
What treatments are available for children?
In childhood, identification of precipitating infections such as streptococcal infection is ideal with appropriate intervention thereafter. Topical therapies are appropriate for limited disease with minimal disability; however, phototherapy and systemic agents can be used in pediatric psoriasis in extensive cases. Topical therapies can include corticosteroids, calcineurin inhibitors often used in sensitive skin such as the face and intertriginous areas, and calcipotriene (Eichenfield et al). Additional agents such as tar and salicylic acid can be used, with limitations on the latter due to risk for absorption in smaller children. Systemic interventions often are introduced after years of disease. A recent study identified practitioners with special interest in pediatric psoriasis and determined that systemic interventions were on average introduced 3 years after psoriasis was diagnosed and most commonly included methotrexate followed by etanercept, the latter having fewer gastrointestinal tract side effects. The panel found that usage of folic acid 6 days weekly minimized gastrointestinal tract side effects with methotrexate. Acitretin and cyclosporine were alternatives (Bronckers et al; Psoriasis Investigator Group [PsIG] of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis [EWGPP]).
Recently, dermatologists have become aware of the dramatic benefits of immune response modifiers and some biologics on pediatric psoriasis. In the setting of joint and skin involvement, I allow the rheumatologist to make the choice of agents for the child's best outcome. However, for pediatric and adolescent psoriasis, we now have 2 US Food and Drug Administration-approved agents and more rapid and thorough testing of adult-approved agents in children, with a hope of greater ability to modify disease course at a younger age, both now and in the future.
Which biologics are approved for the pediatric patient population?
Currently, in the United States 2 biologics have been approved: (1) etanercept, a fusion protein of tumor necrosis factor receptor extracellular domain linked to the Fc portion of human IgG, for moderate to severe plaque psoriasis in patients 4 years and older, and (2) ustekinumab, a human IgG1κ monoclonal antibody against the shared p40 subunit of the IL-12 and IL-23 cytokines, for moderate to severe plaque psoriasis in patients 12 years and older based on the encouraging data of the CADMUS trial (Kellen et al; Landells et al). In Europe, adalimumab has been approved as a first-line therapy in pediatric psoriasis (age ≥4 years), and etanercept (age ≥6 years) and ustekinumab (age ≥12 years) have been approved as second-line agents, all with grade A evidence, according to a recent Italian panel (Fortina et al). (A thorough review of the guidelines on screening, administration, and vaccination is available from Eichenfield et al.)
What treatments are in the pipeline?
In the United States we have clinical trials ongoing of adult-approved topical and immune response-modifying agents such as apremilast. These agents, as they become available and the data are gathered, will be added to what I refer to as our "pharmamentarium" of agents we can use to combat a difficult and disabling illness.
What gaps are there in the pediatric psoriasis research?
Currently, there is poor awareness that there is research for pediatric psoriasis, and there is a need for pediatric groups and the National Psoriasis Foundation to allow children, adolescents, and their families to know that clinical trials are available looking into newer, more targeted, and less immunosuppressive agents. There is new hope on the horizon!
Suggested Readings
Bronckers IMGJ, Seyger MMB, West DP, et al; Psoriasis Investigator Group (PsIG) of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis (EWGPP). Safety of systemic agents for the treatment of pediatric psoriasis. JAMA Dermatol. 2017;153:1147-1157.
Eichenfield LF, Paller AS, Tom WL, et al. Pediatric psoriasis: evolving perspectives [published online January 4, 2018]. Pediatr Dermatol. doi:10.1111/pde.13382.
Fortina AB, Bardazzi F, Berti S, et al. Treatment of severe psoriasis in children: recommendations of an Italian expert group [published online August 23, 2017]. Eur J Pediatr. 2017;176:1339-1354.
Kellen R, Silverberg NB, Lebwohl M. Efficacy and safety of ustekinumab in adolescents. Pediatric Health Med Ther. 2016;7:109-120.
Kim GE, Seidler E, Kimball AB. Effect of age at diagnosis on chronic quality of life and long-term outcomes of individuals with psoriasis [published online December 29, 2014]. Pediatr Dermatol. 2015;32:656-662.
Kwa L, Kwa MC, Silverberg JI. Cardiovascular comorbidities of pediatric psoriasis among hospitalized children in the United States. J Am Acad Dermatol. 2017;77:1023-1029.
Landells I, Marano C, Hsu MC, et al. Ustekinumab in adolescent patients age 12 to 17 years with moderate-to-severe plaque psoriasis: results of the randomized phase 3 CADMUS study [published online August 7, 2015]. J Am Acad Dermatol. 2015;73:594-603.
Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
Raychaudhuri SP, Gross J. A comparative study of pediatric onset psoriasis with adult onset psoriasis. Pediatr Dermatol. 2000;17:174-178.
Tollefson MM, Van Houten HK, Asante D, et al. Association of psoriasis with comorbidity development in children with psoriasis [published online January 10, 2018]. JAMA Dermatol. doi:10.1001/jamadermatol.2017.5417.
What causes psoriasis in children?
Psoriasis is a chronic immune-mediated inflammatory skin disease with a genetic predisposition (Eichenfield et al). Similar to many inflammatory skin diseases, school-aged children have a greater predisposition before or in early adolescence. As with adult disease, pediatric psoriasis has a complex pathogenesis largely related to aberrant immune response to triggers such as infections (eg, streptococcal pharyngitis, perianal streptococcal dermatitis, upper respiratory viral infections), trauma (ie, Koebner phenomenon), stress, and obesity.
What are the emerging data and recommendations on screening for comorbidities in children with psoriasis?
Similar to psoriasis in adults, obesity and the metabolic syndrome are a true association with pediatric psoriasis that has been discussed in the literature (Eichenfield et al). Although many children with psoriasis have obesity as a potential comorbidity, the risk of cardiovascular comorbidities independent of obesity is high in pediatric psoriasis including elevated lipids, hypertension, polycystic ovaries, nonalcoholic liver disease, and elevated liver enzymes (Tollefson et al). Children with psoriasis have greater central obesity and adiposity, often accompanied by a family history of obesity. Interventions in this direction may be needed for long-term disease control and general health (Mercy and Paller). One target population is hospitalized children with psoriasis, particularly black and Hispanic children aged 0 to 9 years. This population has been identified to have a greater risk for obesity, diabetes mellitus, hypertension, arrhythmia, and valvular heart disease (Kwa et al). Therefore, it can be said that dermatologists can help to improve the overall health and lifestyle long-term in children with psoriasis.
Early-onset disease also is associated with greater risk for lifetime quality-of-life impairments including poor lifetime dermatology life quality index scores, depression and psoriasis-induced depression, social discrimination, sleep problems, and recreational drug usage (Kim et al).
How does psoriasis in children differ from adults?
Children have a variety of features that differ from adult disease. First, they are more likely to have an infectious trigger and therefore may have an identifiable treatable source. Second, they are more likely to have a family history of disease, with one-third having a relative with psoriasis, therefore, identifying the child at risk for long-standing disease. Third, children have far more visible head and neck disease, especially facial involvement including eyelids (Raychaudhuri and Gross), which increases the risk of bullying, social stigma, and negative effects on self-image. Of course, site is affected by age, and in infancy diaper dermatitis and inverse disease with maceration and overlying candidal diaper dermatitis can occur. Although children have less joint disease, it can be dramatic and crippling to the developing child.
What treatments are available for children?
In childhood, identification of precipitating infections such as streptococcal infection is ideal with appropriate intervention thereafter. Topical therapies are appropriate for limited disease with minimal disability; however, phototherapy and systemic agents can be used in pediatric psoriasis in extensive cases. Topical therapies can include corticosteroids, calcineurin inhibitors often used in sensitive skin such as the face and intertriginous areas, and calcipotriene (Eichenfield et al). Additional agents such as tar and salicylic acid can be used, with limitations on the latter due to risk for absorption in smaller children. Systemic interventions often are introduced after years of disease. A recent study identified practitioners with special interest in pediatric psoriasis and determined that systemic interventions were on average introduced 3 years after psoriasis was diagnosed and most commonly included methotrexate followed by etanercept, the latter having fewer gastrointestinal tract side effects. The panel found that usage of folic acid 6 days weekly minimized gastrointestinal tract side effects with methotrexate. Acitretin and cyclosporine were alternatives (Bronckers et al; Psoriasis Investigator Group [PsIG] of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis [EWGPP]).
Recently, dermatologists have become aware of the dramatic benefits of immune response modifiers and some biologics on pediatric psoriasis. In the setting of joint and skin involvement, I allow the rheumatologist to make the choice of agents for the child's best outcome. However, for pediatric and adolescent psoriasis, we now have 2 US Food and Drug Administration-approved agents and more rapid and thorough testing of adult-approved agents in children, with a hope of greater ability to modify disease course at a younger age, both now and in the future.
Which biologics are approved for the pediatric patient population?
Currently, in the United States 2 biologics have been approved: (1) etanercept, a fusion protein of tumor necrosis factor receptor extracellular domain linked to the Fc portion of human IgG, for moderate to severe plaque psoriasis in patients 4 years and older, and (2) ustekinumab, a human IgG1κ monoclonal antibody against the shared p40 subunit of the IL-12 and IL-23 cytokines, for moderate to severe plaque psoriasis in patients 12 years and older based on the encouraging data of the CADMUS trial (Kellen et al; Landells et al). In Europe, adalimumab has been approved as a first-line therapy in pediatric psoriasis (age ≥4 years), and etanercept (age ≥6 years) and ustekinumab (age ≥12 years) have been approved as second-line agents, all with grade A evidence, according to a recent Italian panel (Fortina et al). (A thorough review of the guidelines on screening, administration, and vaccination is available from Eichenfield et al.)
What treatments are in the pipeline?
In the United States we have clinical trials ongoing of adult-approved topical and immune response-modifying agents such as apremilast. These agents, as they become available and the data are gathered, will be added to what I refer to as our "pharmamentarium" of agents we can use to combat a difficult and disabling illness.
What gaps are there in the pediatric psoriasis research?
Currently, there is poor awareness that there is research for pediatric psoriasis, and there is a need for pediatric groups and the National Psoriasis Foundation to allow children, adolescents, and their families to know that clinical trials are available looking into newer, more targeted, and less immunosuppressive agents. There is new hope on the horizon!
Suggested Readings
Bronckers IMGJ, Seyger MMB, West DP, et al; Psoriasis Investigator Group (PsIG) of the Pediatric Dermatology Research Alliance and the European Working Group on Pediatric Psoriasis (EWGPP). Safety of systemic agents for the treatment of pediatric psoriasis. JAMA Dermatol. 2017;153:1147-1157.
Eichenfield LF, Paller AS, Tom WL, et al. Pediatric psoriasis: evolving perspectives [published online January 4, 2018]. Pediatr Dermatol. doi:10.1111/pde.13382.
Fortina AB, Bardazzi F, Berti S, et al. Treatment of severe psoriasis in children: recommendations of an Italian expert group [published online August 23, 2017]. Eur J Pediatr. 2017;176:1339-1354.
Kellen R, Silverberg NB, Lebwohl M. Efficacy and safety of ustekinumab in adolescents. Pediatric Health Med Ther. 2016;7:109-120.
Kim GE, Seidler E, Kimball AB. Effect of age at diagnosis on chronic quality of life and long-term outcomes of individuals with psoriasis [published online December 29, 2014]. Pediatr Dermatol. 2015;32:656-662.
Kwa L, Kwa MC, Silverberg JI. Cardiovascular comorbidities of pediatric psoriasis among hospitalized children in the United States. J Am Acad Dermatol. 2017;77:1023-1029.
Landells I, Marano C, Hsu MC, et al. Ustekinumab in adolescent patients age 12 to 17 years with moderate-to-severe plaque psoriasis: results of the randomized phase 3 CADMUS study [published online August 7, 2015]. J Am Acad Dermatol. 2015;73:594-603.
Mercy KM, Paller AS. The relationship between obesity and psoriasis in the pediatric population: implications and future directions. Cutis. 2013;92:107-109.
Raychaudhuri SP, Gross J. A comparative study of pediatric onset psoriasis with adult onset psoriasis. Pediatr Dermatol. 2000;17:174-178.
Tollefson MM, Van Houten HK, Asante D, et al. Association of psoriasis with comorbidity development in children with psoriasis [published online January 10, 2018]. JAMA Dermatol. doi:10.1001/jamadermatol.2017.5417.