User login
CBC values linked to CVD risk in psoriasis
ORLANDO – conducted by researchers at Case Western Reserve University, Cleveland.*
It’s generally accepted that psoriasis increases the risk of cardiovascular disease (CVD), but it’s not clear who’s most at risk. “We really wanted to find something that is cheap and easy to risk stratify these patients” said lead investigator Rosalynn Conic, MD, of Case Western’s department of dermatology.
What they found was “very impressive, for sure,” Dr. Conic said at the International Investigative Dermatology meeting.
The incidence of MI was highest among the 1,920 patients (5%) with elevated RDW and MPV (odds ratio, 3.4; 95% confidence interval, 2.7-4.2; P less than .001), followed by the 7,060 (18%) patients with high RDW and normal MPV (OR, 2.4; 95% CI, 2.1-2.8; P less than .001), as compared with normal/low MPV and RDW patients.
Elevated RDW or elevated RDW plus MPV increased the odds of atrial fibrillation, coronary artery disease, heart failure, and peripheral vascular disease anywhere from 2 to 8.3 times (P less than .001). Among psoriatic arthritis patients, elevated RDW almost doubled the risk of MI (OR, 1.8; P less than .001). Results were adjusted for age, gender, and hypertension.
In a subanalysis of treatment effects, 4 of 23 psoriasis patients at Case Western had elevated RDWs at baseline. Values normalized in the three patients who achieved a 75% reduction in the Psoriasis Area and Severity Index score after about a year of systemic treatment.
“We aim to validate [the study results] with a Veterans Administration data set,” Dr. Conic said. If it pans out, “one use would be to send [patients with elevated values] to a cardiologist earlier” so other CVD risk factors can be monitored and treated. The findings also add to the case for good control, she noted.
Systemic inflammation is the common denominator between the blood value elevations and CVD. The same inflammatory cytokines that cause skin problems in psoriasis also stimulate bone marrow to release immature red blood cells, which are larger than mature cells, leading to an increased RDW. Similarly, elevated MPV indicates a higher number of larger, younger platelets in the blood.
“It’s probably something along those lines, but I think we need to go back to basic science and really figure it out,” Dr. Conic said.
Patients were 18-65 years old. The study excluded patients with diabetes, Crohn’s disease, RA, and generalized atherosclerosis.
The National Institutes of Health funded the work. Dr. Conic reported no relevant financial disclosures.
*This article was updated on May 30, 2018.
SOURCE: Conic R et al. IID 2018, Abstract 550.
ORLANDO – conducted by researchers at Case Western Reserve University, Cleveland.*
It’s generally accepted that psoriasis increases the risk of cardiovascular disease (CVD), but it’s not clear who’s most at risk. “We really wanted to find something that is cheap and easy to risk stratify these patients” said lead investigator Rosalynn Conic, MD, of Case Western’s department of dermatology.
What they found was “very impressive, for sure,” Dr. Conic said at the International Investigative Dermatology meeting.
The incidence of MI was highest among the 1,920 patients (5%) with elevated RDW and MPV (odds ratio, 3.4; 95% confidence interval, 2.7-4.2; P less than .001), followed by the 7,060 (18%) patients with high RDW and normal MPV (OR, 2.4; 95% CI, 2.1-2.8; P less than .001), as compared with normal/low MPV and RDW patients.
Elevated RDW or elevated RDW plus MPV increased the odds of atrial fibrillation, coronary artery disease, heart failure, and peripheral vascular disease anywhere from 2 to 8.3 times (P less than .001). Among psoriatic arthritis patients, elevated RDW almost doubled the risk of MI (OR, 1.8; P less than .001). Results were adjusted for age, gender, and hypertension.
In a subanalysis of treatment effects, 4 of 23 psoriasis patients at Case Western had elevated RDWs at baseline. Values normalized in the three patients who achieved a 75% reduction in the Psoriasis Area and Severity Index score after about a year of systemic treatment.
“We aim to validate [the study results] with a Veterans Administration data set,” Dr. Conic said. If it pans out, “one use would be to send [patients with elevated values] to a cardiologist earlier” so other CVD risk factors can be monitored and treated. The findings also add to the case for good control, she noted.
Systemic inflammation is the common denominator between the blood value elevations and CVD. The same inflammatory cytokines that cause skin problems in psoriasis also stimulate bone marrow to release immature red blood cells, which are larger than mature cells, leading to an increased RDW. Similarly, elevated MPV indicates a higher number of larger, younger platelets in the blood.
“It’s probably something along those lines, but I think we need to go back to basic science and really figure it out,” Dr. Conic said.
Patients were 18-65 years old. The study excluded patients with diabetes, Crohn’s disease, RA, and generalized atherosclerosis.
The National Institutes of Health funded the work. Dr. Conic reported no relevant financial disclosures.
*This article was updated on May 30, 2018.
SOURCE: Conic R et al. IID 2018, Abstract 550.
ORLANDO – conducted by researchers at Case Western Reserve University, Cleveland.*
It’s generally accepted that psoriasis increases the risk of cardiovascular disease (CVD), but it’s not clear who’s most at risk. “We really wanted to find something that is cheap and easy to risk stratify these patients” said lead investigator Rosalynn Conic, MD, of Case Western’s department of dermatology.
What they found was “very impressive, for sure,” Dr. Conic said at the International Investigative Dermatology meeting.
The incidence of MI was highest among the 1,920 patients (5%) with elevated RDW and MPV (odds ratio, 3.4; 95% confidence interval, 2.7-4.2; P less than .001), followed by the 7,060 (18%) patients with high RDW and normal MPV (OR, 2.4; 95% CI, 2.1-2.8; P less than .001), as compared with normal/low MPV and RDW patients.
Elevated RDW or elevated RDW plus MPV increased the odds of atrial fibrillation, coronary artery disease, heart failure, and peripheral vascular disease anywhere from 2 to 8.3 times (P less than .001). Among psoriatic arthritis patients, elevated RDW almost doubled the risk of MI (OR, 1.8; P less than .001). Results were adjusted for age, gender, and hypertension.
In a subanalysis of treatment effects, 4 of 23 psoriasis patients at Case Western had elevated RDWs at baseline. Values normalized in the three patients who achieved a 75% reduction in the Psoriasis Area and Severity Index score after about a year of systemic treatment.
“We aim to validate [the study results] with a Veterans Administration data set,” Dr. Conic said. If it pans out, “one use would be to send [patients with elevated values] to a cardiologist earlier” so other CVD risk factors can be monitored and treated. The findings also add to the case for good control, she noted.
Systemic inflammation is the common denominator between the blood value elevations and CVD. The same inflammatory cytokines that cause skin problems in psoriasis also stimulate bone marrow to release immature red blood cells, which are larger than mature cells, leading to an increased RDW. Similarly, elevated MPV indicates a higher number of larger, younger platelets in the blood.
“It’s probably something along those lines, but I think we need to go back to basic science and really figure it out,” Dr. Conic said.
Patients were 18-65 years old. The study excluded patients with diabetes, Crohn’s disease, RA, and generalized atherosclerosis.
The National Institutes of Health funded the work. Dr. Conic reported no relevant financial disclosures.
*This article was updated on May 30, 2018.
SOURCE: Conic R et al. IID 2018, Abstract 550.
REPORTING FROM IID 2018
Key clinical point: Elevated red blood cell distribution width and mean platelet volume might identify psoriasis patients at risk for cardiovascular disease.
Major finding: The incidence of MI was highest among the 1,920 patients with elevated red cell distribution width and mean platelet volume (odds ratio, 3.4; 95% confidence interval, 2.7-4.2; P less than .001).
Study details: A database review of 39,510 patients with psoriasis.
Disclosures: The National Institutes of Health funded the work. The lead investigator had no disclosures to report.
Source: Conic R et al. IID 2018, Abstract 550.
Topical corticosteroid-retinoid combination effective in moderate to severe psoriasis
The combination of a topical corticosteroid and a topical retinoid for the treatment of plaque psoriasis resulted in significant improvements in clinical signs, in two multicenter, double-blind, vehicle-controlled phase 3 studies.
In the two studies, investigators randomized a total of 418 or vehicle lotion, applied once a day to affected areas. After 8 weeks of treatment, 35.8% of adults in the first study and 45.3% of those in the second study had achieved the primary outcome of at least a two-grade improvement in the Investigator’s Global Assessment score and reaching “clear” or “almost clear,” compared with 7.0% and 12.5%, respectively, of patients treated with the vehicle (P less than .001). The report was published online in the Journal of the American Academy of Dermatology.
At 8 weeks, reduction in erythema was achieved by 44.2% and 49.6% of patients in the treatment arms, compared with 10% and 18.7% of patients in the control arms. Plaque elevation was reduced in 59.3% and 59.7% of patients in the treatment arms, compared with 17.9% and 21.3% of patients in the control arms; and scaling was reduced in 59.4% and 62.9% of those on treatment, compared with 20.6% and 21.0%, respectively. All differences between the treatment and control groups were statistically significant (P less than .001).
Participants who received the treatment also reported significantly lower scores for itching, dryness, and burning or stinging compared with those who received the vehicle lotion.
Dr. Linda Stein Gold of Henry Ford Hospital in Detroit, and her coauthors, wrote that while clinical studies have established the benefit of using a topical corticosteroid as an adjunct to tazarotene for plaque psoriasis, data on their combined use was limited. This combination “was consistently more effective than vehicle in achieving treatment success; effectively reducing affected area and psoriasis signs at the target lesion, and improving QoL [quality of life],” they wrote.
Most patients maintained these improvements over the 4-week posttreatment period.
Patients who received the halobetasol propionate/tazarotene lotion reported more adverse events than did those who received the control lotion, but most were mild to moderate and included contact dermatitis (6.3%), pruritus (2.2%) and application site pain (2.6%). Three serious adverse events were not related to treatment.
The studies were funded by Dow Pharmaceutical Sciences, a division of Valeant Pharmaceuticals North America. Four authors disclosed advisory, consultancy and speaking positions and other funding from the pharmaceutical industry, including with Valeant Pharmaceuticals. Five authors are employees of the company.
SOURCE: Gold L et al. J Am Acad Dermatol. 2018 Mar 31. pii: S0190-9622(18)30494-8. doi: 10.1016/j.jaad.2018.03.040.
The combination of a topical corticosteroid and a topical retinoid for the treatment of plaque psoriasis resulted in significant improvements in clinical signs, in two multicenter, double-blind, vehicle-controlled phase 3 studies.
In the two studies, investigators randomized a total of 418 or vehicle lotion, applied once a day to affected areas. After 8 weeks of treatment, 35.8% of adults in the first study and 45.3% of those in the second study had achieved the primary outcome of at least a two-grade improvement in the Investigator’s Global Assessment score and reaching “clear” or “almost clear,” compared with 7.0% and 12.5%, respectively, of patients treated with the vehicle (P less than .001). The report was published online in the Journal of the American Academy of Dermatology.
At 8 weeks, reduction in erythema was achieved by 44.2% and 49.6% of patients in the treatment arms, compared with 10% and 18.7% of patients in the control arms. Plaque elevation was reduced in 59.3% and 59.7% of patients in the treatment arms, compared with 17.9% and 21.3% of patients in the control arms; and scaling was reduced in 59.4% and 62.9% of those on treatment, compared with 20.6% and 21.0%, respectively. All differences between the treatment and control groups were statistically significant (P less than .001).
Participants who received the treatment also reported significantly lower scores for itching, dryness, and burning or stinging compared with those who received the vehicle lotion.
Dr. Linda Stein Gold of Henry Ford Hospital in Detroit, and her coauthors, wrote that while clinical studies have established the benefit of using a topical corticosteroid as an adjunct to tazarotene for plaque psoriasis, data on their combined use was limited. This combination “was consistently more effective than vehicle in achieving treatment success; effectively reducing affected area and psoriasis signs at the target lesion, and improving QoL [quality of life],” they wrote.
Most patients maintained these improvements over the 4-week posttreatment period.
Patients who received the halobetasol propionate/tazarotene lotion reported more adverse events than did those who received the control lotion, but most were mild to moderate and included contact dermatitis (6.3%), pruritus (2.2%) and application site pain (2.6%). Three serious adverse events were not related to treatment.
The studies were funded by Dow Pharmaceutical Sciences, a division of Valeant Pharmaceuticals North America. Four authors disclosed advisory, consultancy and speaking positions and other funding from the pharmaceutical industry, including with Valeant Pharmaceuticals. Five authors are employees of the company.
SOURCE: Gold L et al. J Am Acad Dermatol. 2018 Mar 31. pii: S0190-9622(18)30494-8. doi: 10.1016/j.jaad.2018.03.040.
The combination of a topical corticosteroid and a topical retinoid for the treatment of plaque psoriasis resulted in significant improvements in clinical signs, in two multicenter, double-blind, vehicle-controlled phase 3 studies.
In the two studies, investigators randomized a total of 418 or vehicle lotion, applied once a day to affected areas. After 8 weeks of treatment, 35.8% of adults in the first study and 45.3% of those in the second study had achieved the primary outcome of at least a two-grade improvement in the Investigator’s Global Assessment score and reaching “clear” or “almost clear,” compared with 7.0% and 12.5%, respectively, of patients treated with the vehicle (P less than .001). The report was published online in the Journal of the American Academy of Dermatology.
At 8 weeks, reduction in erythema was achieved by 44.2% and 49.6% of patients in the treatment arms, compared with 10% and 18.7% of patients in the control arms. Plaque elevation was reduced in 59.3% and 59.7% of patients in the treatment arms, compared with 17.9% and 21.3% of patients in the control arms; and scaling was reduced in 59.4% and 62.9% of those on treatment, compared with 20.6% and 21.0%, respectively. All differences between the treatment and control groups were statistically significant (P less than .001).
Participants who received the treatment also reported significantly lower scores for itching, dryness, and burning or stinging compared with those who received the vehicle lotion.
Dr. Linda Stein Gold of Henry Ford Hospital in Detroit, and her coauthors, wrote that while clinical studies have established the benefit of using a topical corticosteroid as an adjunct to tazarotene for plaque psoriasis, data on their combined use was limited. This combination “was consistently more effective than vehicle in achieving treatment success; effectively reducing affected area and psoriasis signs at the target lesion, and improving QoL [quality of life],” they wrote.
Most patients maintained these improvements over the 4-week posttreatment period.
Patients who received the halobetasol propionate/tazarotene lotion reported more adverse events than did those who received the control lotion, but most were mild to moderate and included contact dermatitis (6.3%), pruritus (2.2%) and application site pain (2.6%). Three serious adverse events were not related to treatment.
The studies were funded by Dow Pharmaceutical Sciences, a division of Valeant Pharmaceuticals North America. Four authors disclosed advisory, consultancy and speaking positions and other funding from the pharmaceutical industry, including with Valeant Pharmaceuticals. Five authors are employees of the company.
SOURCE: Gold L et al. J Am Acad Dermatol. 2018 Mar 31. pii: S0190-9622(18)30494-8. doi: 10.1016/j.jaad.2018.03.040.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Topical halobetasol propionate and tazarotene can significantly improve plaque psoriasis symptoms.
Major finding: Nearly half of adults treated with topical halobetasol propionate and tazarotene were “clear” or “almost clear” after 8 weeks.
Study details: Two multicenter, double-blind, vehicle-controlled phase 3 studies of 418 adults with psoriasis.
Disclosures: The studies were funded by Dow Pharmaceutical Sciences, a division of Valeant Pharmaceuticals North America. Four authors disclosed advisory, consultancy and speaking positions and other funding from the pharmaceutical industry, including Valeant Pharmaceuticals. Five authors are employees of the company.
Source: Gold L et al. J Am Acad Dermatol. 2018 Mar 31. pii: S0190-9622(18)30494-8. doi: 10.1016/j.jaad.2018.03.040.
Secukinumab Emerges as a Rapidly Effective Therapy for Pityriasis Rubra Pilaris
Although there currently are no formal guidelines for the treatment of refractory pityriasis rubra pilaris (PRP), successful off-label treatment of the condition with multiple biologics approved for psoriasis has been reported.1,2 Secukinumab, an IL-17A antagonist, has shown particularly striking results in the treatment of PRP in 2 recent case reports.3,4 We report 2 additional cases of severe refractory PRP that responded rapidly to treatment with secukinumab. In both cases, the patients’ erythematous plaques resolved or had nearly resolved by week 4 of treatment. Our findings suggest that IL-17 plays an important role in PRP pathogenesis and support future clinical trials of anti–IL-17 agents for treatment of this entity.
Case Reports
Patient 1
A 60-year-old man with a history of biopsy-proven PRP presented with persistent generalized erythema, scattered patches of normal skin, and hyperkeratotic plaques on the bilateral palms of 1 year’s duration. Previous therapies included topical steroids, topical calcipotriene, adalimumab 40 mg once every other week, infliximab 5 mg/kg once every 8 weeks, ustekinumab 90 mg once every 12 weeks, acitretin 25 mg once daily, and most recently cyclosporine 200 mg twice daily. Of these treatments, infliximab was the
At 4 weeks’ follow-up, there was a marked decrease in erythema and scaling. The body surface area affected had decreased to 5%, and improvement of palmar keratoderma was noted. The patient continued with maintenance dosing of secukinumab 300 mg once every 4 weeks. By week 8, the erythema had fully resolved (Figure 1B), and he remained clear at week 24. No adverse events were noted since initiation of therapy.
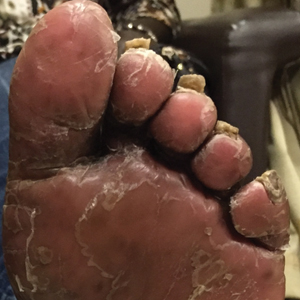
Patient 2
A 74-year-old woman with a history of PRP that had previously been misdiagnosed as psoriasis by an outside physician presented for evaluation of palmoplantar keratoderma (Figure 2A), follicular hyperkeratosis, and erythematous plaques on the trunk and arms of 5 years’ duration. Previous therapies included topical steroids, topical urea, methotrexate 20 mg once weekly, adalimumab 40 mg once every other week, infliximab 10 mg/kg once every 4 weeks, ustekinumab 90 mg once every 12 weeks, and most recently acitretin 50 mg once daily.
The patient had been maintained on ustekinumab and acitretin for 2 years with only mild improvement. Ustekinumab was then discontinued, and after 3 months treatment with secukinumab was added to the once-daily acitretin. Similar to Patient 1, loading doses of secukinumab 300 mg were administered once weekly for 5 weeks. The plaques on the trunk and arms had resolved by week 4, but the palmoplantar keratoderma persisted. The patient continued with the maintenance dose of secukinumab 300 mg once every 4 weeks and reported an increase in peeling of the palms and soles at week 8.
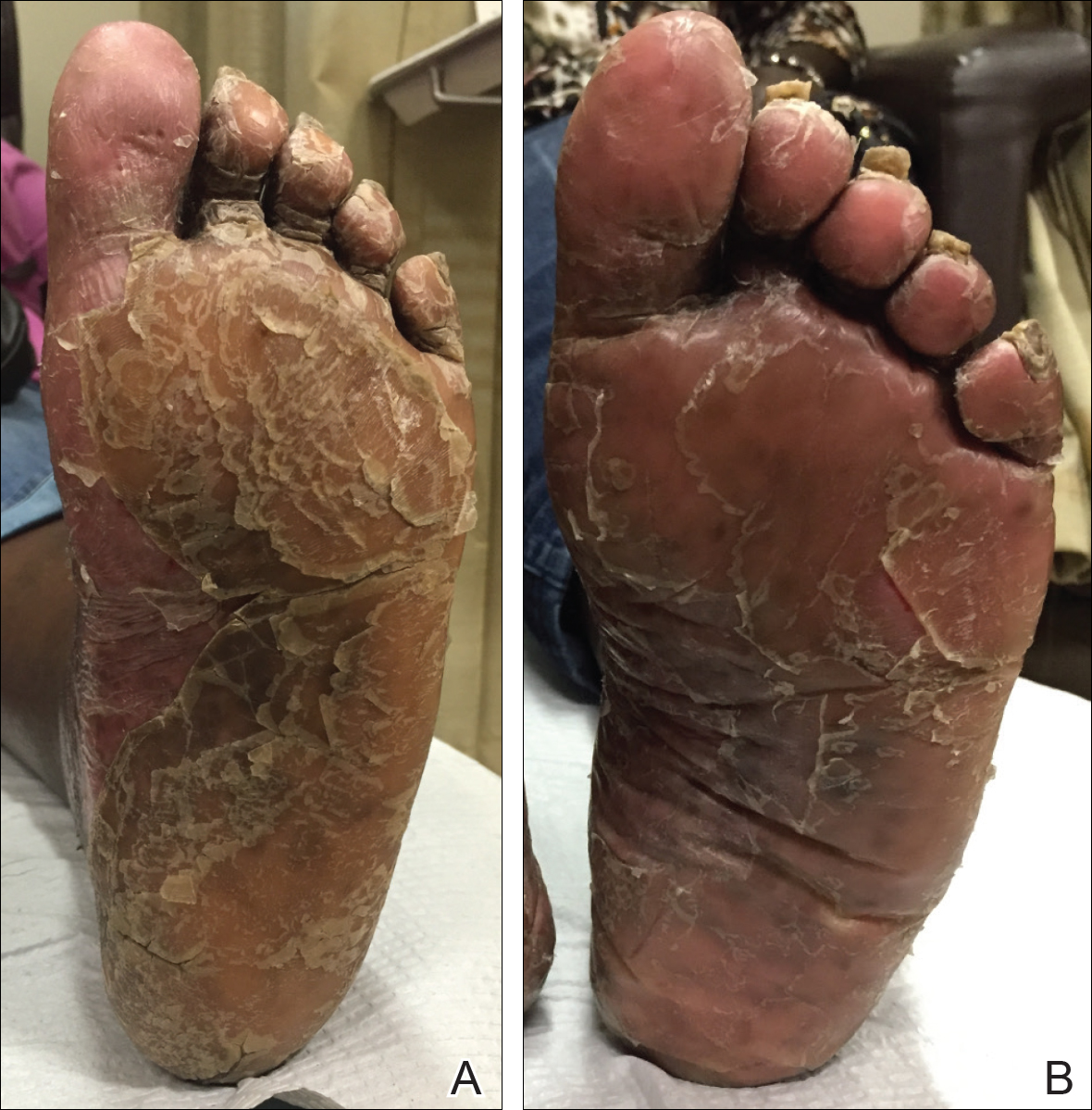
By week 12 of treatment, the palmar keratoderma had resolved, and debridement of the soles revealed patches of normal skin (Figure 2B). By week 52, no adverse events had been noted. The patient continued to experience mild keratoderma of the soles, making us reluctant to discontinue acitretin; however, she has maintained her maximal response, and her quality of life has significantly improved. The patient was continued on acitretin and secukinumab, and her condition remained stable.
Comment
Because there are no formal treatment guidelines for refractory PRP, case reports play an important role in clinical decision-making. When a patient is unresponsive to topical medications and first-line traditional systemic therapies (eg, methotrexate, cyclosporine, acitretin), biologic drugs effective in the treatment of psoriasis are widely accepted as the next therapeutic step.1 The biologic medications that are most often reported in the treatment of PRP are the TNF-α antagonists, as they have been available the longest.1-2 In a systematic review of 15 patients with PRP who were treated with TNF-α antagonists,2 80% of patients achieved complete response (mean time to maximal response, 5 months). There also are a number of reports of successful treatment of PRP with the IL-12/23 antagonist ustekinumab, which has been commercially available since 2009.5-9 Although improvement was noted in most of these patients at the time of the second injection (week 4 of therapy), maximal response with ustekinumab typically occurs between weeks 12 and 28.10
In our cases of PRP treated with secukinumab as well as 2 others that were recently reported in the literature, resolution of erythema and plaques was rapid. This superiority of the response rate parallels the performance of secukinumab relative to ustekinumab in patients with psoriasis11 In one case of a 67-year-old man with PRP treated with secukinumab, scaling and pruritus were reduced by week 3 of treatment and erythema had cleared by week 8.3 In another case of a 33-year-old woman with PRP, pruritus resolved after 1 week of treatment and erythematous plaques and palmoplantar keratoderma improved by week 2.4 In both of our cases, plaques had resolved or nearly resolved by week 4 of follow-up. Patient 1 achieved complete response at week 8 of therapy. Patient 2 never attained complete response, but by week 12 she achieved maximal response, which still resulted in markedly increased quality of life. We do not intend to make additions to her treatment plan because she is currently the clearest she has been since onset of symptoms and is happy with her present condition.
Although it is difficult to predict the long-term prognosis in our 2 patients, we will continue their current regimens indefinitely—as long as the response persists and no adverse events are experienced. This approach is consistent with guidelines for management of plaque psoriasis with secukinumab.12
This accumulation of evidence suggests the importance of the role of IL-17 in the pathogenesis of PRP. The serum level of IL-17 was not evaluated in our patients, but elevation of IL-17 has been reported in a case of PRP.13 Further studies are needed to clarify the role of IL-17 in this disease entity.
Conclusion
Given the refractory nature of PRP and the relative safety of targeted immunotherapy, trials of new biologics and potent small molecules approved for psoriasis treatment are worth exploring for PRP. In light of our reports and those in the literature and given the relative safety of anti–IL-17 agents, it may be reasonable to consider such agents as a first-line therapy for this predictably refractory disease.
- Klein A, Landthaler M, Karrer S. Pityriasis rubra pilaris. Am J Clin Dermatol. 2010;11:157-170.
- Petrof G, Almaani N, Archer CB, et al. A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists. J Eur Acad Dermatol Venereol. 2013;27:E131-E135.
- Schuster D, Pfister-Wartha A, Bruckner-Tuderman L, et al. Successful treatment of refractory pityriasis rubra pilaris with secukinumab. JAMA Dermatol. 2016;152:1278-1280.
- Gauci ML, Jachiet M, Gottlieb J, et al. Successful treatment of type II pityriasis rubra pilaris with secukinumab. JAAD Case Rep. 2016;2:462-264.
- Chowdhary M, Davila U, Cohen DJ. Ustekinumab as an alternative treatment option for chronic pityriasis rubra pilaris. Case Rep Dermatol. 2015;7:46-50.
- Wohlrab J, Kreft B. Treatment of pityriasis rubra pilaris with ustekinumab. Br J Dermatol. 2010;163:655-656.
- Villaverde RR, Cano DS. Successful treatment of type 1 pityriasis rubra pilaris with ustekinumab therapy. Eur J Dermatol. 2010;20:630-631.
- Di Stefani A, Galluzzo M, Talamonti M, et al. Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris. J Dermatol Case Rep. 2013;7:5-9.
- Eytan O, Sarig O, Sprecher E, et al. Clinical response to ustekinumab in familial pityriasis rubra pilaris caused by a novel mutation in CARD14. Br J Dermatol. 2014;171:420-422.
- Papp KA, Langley RG, Lebwohl M, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet. 2008;371:1675-1684.
- Thaçi D, Blauvelt A, Reich K, et al. Secukinumab is superior to ustekinumab in clearing skin of subjects with moderate to severe plaque psoriasis: CLEAR, a randomized controlled trial. J Am Acad Dermatol. 2015;73:400-409.
- van de Kerkhof PC, Griffiths CE, Reich K, et al. Secukinumab long-term safety experience: a pooled analysis of 10 phase II and III clinical studies in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2016;75:83-98.
- Adnot-Desanlis L, Antonicelli F, Tabary T, et al. Effectiveness of infliximab in pityriasis rubra pilaris is associated with pro-inflammatory cytokine inhibition. Dermatology. 2013;226:41-46.
Although there currently are no formal guidelines for the treatment of refractory pityriasis rubra pilaris (PRP), successful off-label treatment of the condition with multiple biologics approved for psoriasis has been reported.1,2 Secukinumab, an IL-17A antagonist, has shown particularly striking results in the treatment of PRP in 2 recent case reports.3,4 We report 2 additional cases of severe refractory PRP that responded rapidly to treatment with secukinumab. In both cases, the patients’ erythematous plaques resolved or had nearly resolved by week 4 of treatment. Our findings suggest that IL-17 plays an important role in PRP pathogenesis and support future clinical trials of anti–IL-17 agents for treatment of this entity.
Case Reports
Patient 1
A 60-year-old man with a history of biopsy-proven PRP presented with persistent generalized erythema, scattered patches of normal skin, and hyperkeratotic plaques on the bilateral palms of 1 year’s duration. Previous therapies included topical steroids, topical calcipotriene, adalimumab 40 mg once every other week, infliximab 5 mg/kg once every 8 weeks, ustekinumab 90 mg once every 12 weeks, acitretin 25 mg once daily, and most recently cyclosporine 200 mg twice daily. Of these treatments, infliximab was the
At 4 weeks’ follow-up, there was a marked decrease in erythema and scaling. The body surface area affected had decreased to 5%, and improvement of palmar keratoderma was noted. The patient continued with maintenance dosing of secukinumab 300 mg once every 4 weeks. By week 8, the erythema had fully resolved (Figure 1B), and he remained clear at week 24. No adverse events were noted since initiation of therapy.

Patient 2
A 74-year-old woman with a history of PRP that had previously been misdiagnosed as psoriasis by an outside physician presented for evaluation of palmoplantar keratoderma (Figure 2A), follicular hyperkeratosis, and erythematous plaques on the trunk and arms of 5 years’ duration. Previous therapies included topical steroids, topical urea, methotrexate 20 mg once weekly, adalimumab 40 mg once every other week, infliximab 10 mg/kg once every 4 weeks, ustekinumab 90 mg once every 12 weeks, and most recently acitretin 50 mg once daily.
The patient had been maintained on ustekinumab and acitretin for 2 years with only mild improvement. Ustekinumab was then discontinued, and after 3 months treatment with secukinumab was added to the once-daily acitretin. Similar to Patient 1, loading doses of secukinumab 300 mg were administered once weekly for 5 weeks. The plaques on the trunk and arms had resolved by week 4, but the palmoplantar keratoderma persisted. The patient continued with the maintenance dose of secukinumab 300 mg once every 4 weeks and reported an increase in peeling of the palms and soles at week 8.

By week 12 of treatment, the palmar keratoderma had resolved, and debridement of the soles revealed patches of normal skin (Figure 2B). By week 52, no adverse events had been noted. The patient continued to experience mild keratoderma of the soles, making us reluctant to discontinue acitretin; however, she has maintained her maximal response, and her quality of life has significantly improved. The patient was continued on acitretin and secukinumab, and her condition remained stable.
Comment
Because there are no formal treatment guidelines for refractory PRP, case reports play an important role in clinical decision-making. When a patient is unresponsive to topical medications and first-line traditional systemic therapies (eg, methotrexate, cyclosporine, acitretin), biologic drugs effective in the treatment of psoriasis are widely accepted as the next therapeutic step.1 The biologic medications that are most often reported in the treatment of PRP are the TNF-α antagonists, as they have been available the longest.1-2 In a systematic review of 15 patients with PRP who were treated with TNF-α antagonists,2 80% of patients achieved complete response (mean time to maximal response, 5 months). There also are a number of reports of successful treatment of PRP with the IL-12/23 antagonist ustekinumab, which has been commercially available since 2009.5-9 Although improvement was noted in most of these patients at the time of the second injection (week 4 of therapy), maximal response with ustekinumab typically occurs between weeks 12 and 28.10
In our cases of PRP treated with secukinumab as well as 2 others that were recently reported in the literature, resolution of erythema and plaques was rapid. This superiority of the response rate parallels the performance of secukinumab relative to ustekinumab in patients with psoriasis11 In one case of a 67-year-old man with PRP treated with secukinumab, scaling and pruritus were reduced by week 3 of treatment and erythema had cleared by week 8.3 In another case of a 33-year-old woman with PRP, pruritus resolved after 1 week of treatment and erythematous plaques and palmoplantar keratoderma improved by week 2.4 In both of our cases, plaques had resolved or nearly resolved by week 4 of follow-up. Patient 1 achieved complete response at week 8 of therapy. Patient 2 never attained complete response, but by week 12 she achieved maximal response, which still resulted in markedly increased quality of life. We do not intend to make additions to her treatment plan because she is currently the clearest she has been since onset of symptoms and is happy with her present condition.
Although it is difficult to predict the long-term prognosis in our 2 patients, we will continue their current regimens indefinitely—as long as the response persists and no adverse events are experienced. This approach is consistent with guidelines for management of plaque psoriasis with secukinumab.12
This accumulation of evidence suggests the importance of the role of IL-17 in the pathogenesis of PRP. The serum level of IL-17 was not evaluated in our patients, but elevation of IL-17 has been reported in a case of PRP.13 Further studies are needed to clarify the role of IL-17 in this disease entity.
Conclusion
Given the refractory nature of PRP and the relative safety of targeted immunotherapy, trials of new biologics and potent small molecules approved for psoriasis treatment are worth exploring for PRP. In light of our reports and those in the literature and given the relative safety of anti–IL-17 agents, it may be reasonable to consider such agents as a first-line therapy for this predictably refractory disease.
Although there currently are no formal guidelines for the treatment of refractory pityriasis rubra pilaris (PRP), successful off-label treatment of the condition with multiple biologics approved for psoriasis has been reported.1,2 Secukinumab, an IL-17A antagonist, has shown particularly striking results in the treatment of PRP in 2 recent case reports.3,4 We report 2 additional cases of severe refractory PRP that responded rapidly to treatment with secukinumab. In both cases, the patients’ erythematous plaques resolved or had nearly resolved by week 4 of treatment. Our findings suggest that IL-17 plays an important role in PRP pathogenesis and support future clinical trials of anti–IL-17 agents for treatment of this entity.
Case Reports
Patient 1
A 60-year-old man with a history of biopsy-proven PRP presented with persistent generalized erythema, scattered patches of normal skin, and hyperkeratotic plaques on the bilateral palms of 1 year’s duration. Previous therapies included topical steroids, topical calcipotriene, adalimumab 40 mg once every other week, infliximab 5 mg/kg once every 8 weeks, ustekinumab 90 mg once every 12 weeks, acitretin 25 mg once daily, and most recently cyclosporine 200 mg twice daily. Of these treatments, infliximab was the
At 4 weeks’ follow-up, there was a marked decrease in erythema and scaling. The body surface area affected had decreased to 5%, and improvement of palmar keratoderma was noted. The patient continued with maintenance dosing of secukinumab 300 mg once every 4 weeks. By week 8, the erythema had fully resolved (Figure 1B), and he remained clear at week 24. No adverse events were noted since initiation of therapy.

Patient 2
A 74-year-old woman with a history of PRP that had previously been misdiagnosed as psoriasis by an outside physician presented for evaluation of palmoplantar keratoderma (Figure 2A), follicular hyperkeratosis, and erythematous plaques on the trunk and arms of 5 years’ duration. Previous therapies included topical steroids, topical urea, methotrexate 20 mg once weekly, adalimumab 40 mg once every other week, infliximab 10 mg/kg once every 4 weeks, ustekinumab 90 mg once every 12 weeks, and most recently acitretin 50 mg once daily.
The patient had been maintained on ustekinumab and acitretin for 2 years with only mild improvement. Ustekinumab was then discontinued, and after 3 months treatment with secukinumab was added to the once-daily acitretin. Similar to Patient 1, loading doses of secukinumab 300 mg were administered once weekly for 5 weeks. The plaques on the trunk and arms had resolved by week 4, but the palmoplantar keratoderma persisted. The patient continued with the maintenance dose of secukinumab 300 mg once every 4 weeks and reported an increase in peeling of the palms and soles at week 8.

By week 12 of treatment, the palmar keratoderma had resolved, and debridement of the soles revealed patches of normal skin (Figure 2B). By week 52, no adverse events had been noted. The patient continued to experience mild keratoderma of the soles, making us reluctant to discontinue acitretin; however, she has maintained her maximal response, and her quality of life has significantly improved. The patient was continued on acitretin and secukinumab, and her condition remained stable.
Comment
Because there are no formal treatment guidelines for refractory PRP, case reports play an important role in clinical decision-making. When a patient is unresponsive to topical medications and first-line traditional systemic therapies (eg, methotrexate, cyclosporine, acitretin), biologic drugs effective in the treatment of psoriasis are widely accepted as the next therapeutic step.1 The biologic medications that are most often reported in the treatment of PRP are the TNF-α antagonists, as they have been available the longest.1-2 In a systematic review of 15 patients with PRP who were treated with TNF-α antagonists,2 80% of patients achieved complete response (mean time to maximal response, 5 months). There also are a number of reports of successful treatment of PRP with the IL-12/23 antagonist ustekinumab, which has been commercially available since 2009.5-9 Although improvement was noted in most of these patients at the time of the second injection (week 4 of therapy), maximal response with ustekinumab typically occurs between weeks 12 and 28.10
In our cases of PRP treated with secukinumab as well as 2 others that were recently reported in the literature, resolution of erythema and plaques was rapid. This superiority of the response rate parallels the performance of secukinumab relative to ustekinumab in patients with psoriasis11 In one case of a 67-year-old man with PRP treated with secukinumab, scaling and pruritus were reduced by week 3 of treatment and erythema had cleared by week 8.3 In another case of a 33-year-old woman with PRP, pruritus resolved after 1 week of treatment and erythematous plaques and palmoplantar keratoderma improved by week 2.4 In both of our cases, plaques had resolved or nearly resolved by week 4 of follow-up. Patient 1 achieved complete response at week 8 of therapy. Patient 2 never attained complete response, but by week 12 she achieved maximal response, which still resulted in markedly increased quality of life. We do not intend to make additions to her treatment plan because she is currently the clearest she has been since onset of symptoms and is happy with her present condition.
Although it is difficult to predict the long-term prognosis in our 2 patients, we will continue their current regimens indefinitely—as long as the response persists and no adverse events are experienced. This approach is consistent with guidelines for management of plaque psoriasis with secukinumab.12
This accumulation of evidence suggests the importance of the role of IL-17 in the pathogenesis of PRP. The serum level of IL-17 was not evaluated in our patients, but elevation of IL-17 has been reported in a case of PRP.13 Further studies are needed to clarify the role of IL-17 in this disease entity.
Conclusion
Given the refractory nature of PRP and the relative safety of targeted immunotherapy, trials of new biologics and potent small molecules approved for psoriasis treatment are worth exploring for PRP. In light of our reports and those in the literature and given the relative safety of anti–IL-17 agents, it may be reasonable to consider such agents as a first-line therapy for this predictably refractory disease.
- Klein A, Landthaler M, Karrer S. Pityriasis rubra pilaris. Am J Clin Dermatol. 2010;11:157-170.
- Petrof G, Almaani N, Archer CB, et al. A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists. J Eur Acad Dermatol Venereol. 2013;27:E131-E135.
- Schuster D, Pfister-Wartha A, Bruckner-Tuderman L, et al. Successful treatment of refractory pityriasis rubra pilaris with secukinumab. JAMA Dermatol. 2016;152:1278-1280.
- Gauci ML, Jachiet M, Gottlieb J, et al. Successful treatment of type II pityriasis rubra pilaris with secukinumab. JAAD Case Rep. 2016;2:462-264.
- Chowdhary M, Davila U, Cohen DJ. Ustekinumab as an alternative treatment option for chronic pityriasis rubra pilaris. Case Rep Dermatol. 2015;7:46-50.
- Wohlrab J, Kreft B. Treatment of pityriasis rubra pilaris with ustekinumab. Br J Dermatol. 2010;163:655-656.
- Villaverde RR, Cano DS. Successful treatment of type 1 pityriasis rubra pilaris with ustekinumab therapy. Eur J Dermatol. 2010;20:630-631.
- Di Stefani A, Galluzzo M, Talamonti M, et al. Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris. J Dermatol Case Rep. 2013;7:5-9.
- Eytan O, Sarig O, Sprecher E, et al. Clinical response to ustekinumab in familial pityriasis rubra pilaris caused by a novel mutation in CARD14. Br J Dermatol. 2014;171:420-422.
- Papp KA, Langley RG, Lebwohl M, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet. 2008;371:1675-1684.
- Thaçi D, Blauvelt A, Reich K, et al. Secukinumab is superior to ustekinumab in clearing skin of subjects with moderate to severe plaque psoriasis: CLEAR, a randomized controlled trial. J Am Acad Dermatol. 2015;73:400-409.
- van de Kerkhof PC, Griffiths CE, Reich K, et al. Secukinumab long-term safety experience: a pooled analysis of 10 phase II and III clinical studies in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2016;75:83-98.
- Adnot-Desanlis L, Antonicelli F, Tabary T, et al. Effectiveness of infliximab in pityriasis rubra pilaris is associated with pro-inflammatory cytokine inhibition. Dermatology. 2013;226:41-46.
- Klein A, Landthaler M, Karrer S. Pityriasis rubra pilaris. Am J Clin Dermatol. 2010;11:157-170.
- Petrof G, Almaani N, Archer CB, et al. A systematic review of the literature on the treatment of pityriasis rubra pilaris type 1 with TNF-antagonists. J Eur Acad Dermatol Venereol. 2013;27:E131-E135.
- Schuster D, Pfister-Wartha A, Bruckner-Tuderman L, et al. Successful treatment of refractory pityriasis rubra pilaris with secukinumab. JAMA Dermatol. 2016;152:1278-1280.
- Gauci ML, Jachiet M, Gottlieb J, et al. Successful treatment of type II pityriasis rubra pilaris with secukinumab. JAAD Case Rep. 2016;2:462-264.
- Chowdhary M, Davila U, Cohen DJ. Ustekinumab as an alternative treatment option for chronic pityriasis rubra pilaris. Case Rep Dermatol. 2015;7:46-50.
- Wohlrab J, Kreft B. Treatment of pityriasis rubra pilaris with ustekinumab. Br J Dermatol. 2010;163:655-656.
- Villaverde RR, Cano DS. Successful treatment of type 1 pityriasis rubra pilaris with ustekinumab therapy. Eur J Dermatol. 2010;20:630-631.
- Di Stefani A, Galluzzo M, Talamonti M, et al. Long-term ustekinumab treatment for refractory type I pityriasis rubra pilaris. J Dermatol Case Rep. 2013;7:5-9.
- Eytan O, Sarig O, Sprecher E, et al. Clinical response to ustekinumab in familial pityriasis rubra pilaris caused by a novel mutation in CARD14. Br J Dermatol. 2014;171:420-422.
- Papp KA, Langley RG, Lebwohl M, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet. 2008;371:1675-1684.
- Thaçi D, Blauvelt A, Reich K, et al. Secukinumab is superior to ustekinumab in clearing skin of subjects with moderate to severe plaque psoriasis: CLEAR, a randomized controlled trial. J Am Acad Dermatol. 2015;73:400-409.
- van de Kerkhof PC, Griffiths CE, Reich K, et al. Secukinumab long-term safety experience: a pooled analysis of 10 phase II and III clinical studies in patients with moderate to severe plaque psoriasis. J Am Acad Dermatol. 2016;75:83-98.
- Adnot-Desanlis L, Antonicelli F, Tabary T, et al. Effectiveness of infliximab in pityriasis rubra pilaris is associated with pro-inflammatory cytokine inhibition. Dermatology. 2013;226:41-46.
Practice Points
- In patients with pityriasis rubra pilaris (PRP) who have not responded to topical treatments, off-label treatment with systemic therapies approved for plaque psoriasis can be considered.
- Secukinumab, an IL-17A antagonist, has shown particularly striking results in the treatment of PRP.
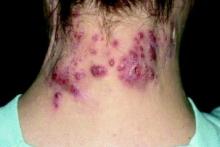
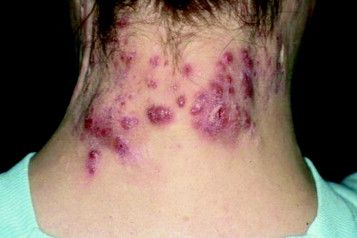
Hidradenitis suppurativa packs mighty QOL impact
KAUAI, HAWAII – Anyone who has treated patients with hidradenitis suppurativa (HS) recognizes that this can be a debilitating disease. Helping put that into fuller perspective, recent evidence has shown that the quality of life effects of moderate to severe HS are objectively worse than those of moderate to severe psoriasis, according to Iltefat H. Hamzavi, MD, president of the Hidradenitis Suppurativa Foundation and a dermatologist at Henry Ford Hospital in Detroit.
He was lead author of a study in which he and his coinvestigators compared weighted averages of a variety of quality of life measures in patients with moderate to severe HS or psoriasis who participated in five AbbVie-sponsored randomized clinical trials of biologic agents (J Am Acad Dermatol. 2017 Dec;77[6]:1038-46).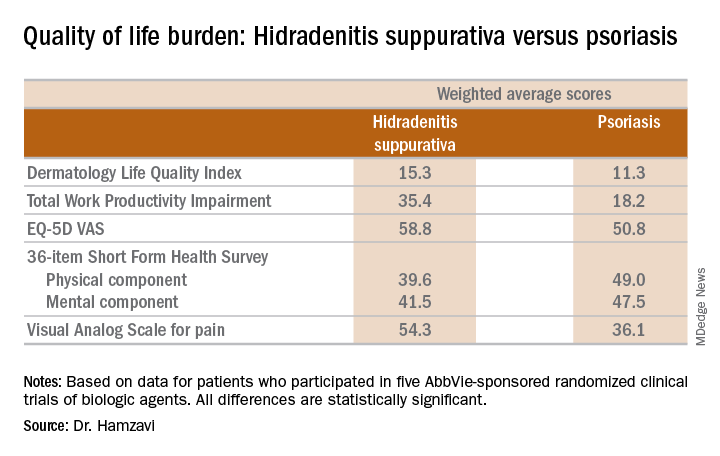
“The number of HS patients who experience downward drift – losing their job and their health insurance and ultimately being unable to move out of a lower socioeconomic group – is staggering,” the dermatologist said.
which underscores the importance of a psychiatric evaluation as part of routine care for patients with this dermatologic disease. “Suicide is much more common in the HS population than in almost any other dermatologic disease,” Dr. Hamzavi added.
He reported serving as a consultant for AbbVie, Incyte, and UCB.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Anyone who has treated patients with hidradenitis suppurativa (HS) recognizes that this can be a debilitating disease. Helping put that into fuller perspective, recent evidence has shown that the quality of life effects of moderate to severe HS are objectively worse than those of moderate to severe psoriasis, according to Iltefat H. Hamzavi, MD, president of the Hidradenitis Suppurativa Foundation and a dermatologist at Henry Ford Hospital in Detroit.
He was lead author of a study in which he and his coinvestigators compared weighted averages of a variety of quality of life measures in patients with moderate to severe HS or psoriasis who participated in five AbbVie-sponsored randomized clinical trials of biologic agents (J Am Acad Dermatol. 2017 Dec;77[6]:1038-46).
“The number of HS patients who experience downward drift – losing their job and their health insurance and ultimately being unable to move out of a lower socioeconomic group – is staggering,” the dermatologist said.
which underscores the importance of a psychiatric evaluation as part of routine care for patients with this dermatologic disease. “Suicide is much more common in the HS population than in almost any other dermatologic disease,” Dr. Hamzavi added.
He reported serving as a consultant for AbbVie, Incyte, and UCB.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Anyone who has treated patients with hidradenitis suppurativa (HS) recognizes that this can be a debilitating disease. Helping put that into fuller perspective, recent evidence has shown that the quality of life effects of moderate to severe HS are objectively worse than those of moderate to severe psoriasis, according to Iltefat H. Hamzavi, MD, president of the Hidradenitis Suppurativa Foundation and a dermatologist at Henry Ford Hospital in Detroit.
He was lead author of a study in which he and his coinvestigators compared weighted averages of a variety of quality of life measures in patients with moderate to severe HS or psoriasis who participated in five AbbVie-sponsored randomized clinical trials of biologic agents (J Am Acad Dermatol. 2017 Dec;77[6]:1038-46).
“The number of HS patients who experience downward drift – losing their job and their health insurance and ultimately being unable to move out of a lower socioeconomic group – is staggering,” the dermatologist said.
which underscores the importance of a psychiatric evaluation as part of routine care for patients with this dermatologic disease. “Suicide is much more common in the HS population than in almost any other dermatologic disease,” Dr. Hamzavi added.
He reported serving as a consultant for AbbVie, Incyte, and UCB.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Psoriasis duration reflects cardiovascular event risk
KAUAI, HAWAII – The recent report that the risk of a major adverse cardiovascular event increases by 1% more than in the general population for each additional year of psoriasis duration is sobering news for physicians who treat pediatric psoriasis.
“If I have a 16-year-old who has a 5-year history of psoriasis, what does that mean for when she’s 30 or 40? And should we be intervening more aggressively?” Lawrence F. Eichenfield, MD, asked at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even though there’s not a great deal of evidence, there’s some evidence to rationalize early screening in psoriasis,” according to Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
Psoriasis develops during childhood in almost one-third of patients.
The pediatric psoriasis screening guidelines describe a simple routine screening program and timeline for early identification of overweight or obesity, type 2 diabetes, hypertension, nonalcoholic fatty liver disease, anxiety, depression, substance abuse, inflammatory bowel disease, and quality of life issues, all of which are encountered with increased frequency in pediatric psoriasis patients. A fasting lipid panel is recommended in children aged 9-11 years with psoriasis and again at age 17-21 years.
“Don’t forget arthritis. For a kid with psoriasis, at every office visit, I ask about morning stiffness or limp. Those are probably the two most sensitive questions in screening for psoriatic arthritis,” according to Dr. Eichenfield.
It has been clear for some time that the skin is not the only organ affected by psoriatic inflammation. The study that quantified the relationship between psoriasis duration and cardiovascular risk – a 1% increase for each year of psoriasis – was a collaboration between investigators at the University of Copenhagen and the University of Pennsylvania, Philadelphia.
The two-part project included aortal imaging of 190 psoriasis patients using fludeoxyglucose F 18 PET/CT scan, which showed a strong relationship between duration of psoriasis and the degree of vascular inflammation. This was bolstered by a population-based study using Danish national registry data on 87,161 psoriasis patients and 4.2 million controls from the general Danish population (J Am Acad Dermatol. 2017 Oct;77[4]:650-56.e3).
Dr. Eichenfield reported serving as a consultant to and/or recipient of research grants from more than a dozen pharmaceutical companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – The recent report that the risk of a major adverse cardiovascular event increases by 1% more than in the general population for each additional year of psoriasis duration is sobering news for physicians who treat pediatric psoriasis.
“If I have a 16-year-old who has a 5-year history of psoriasis, what does that mean for when she’s 30 or 40? And should we be intervening more aggressively?” Lawrence F. Eichenfield, MD, asked at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even though there’s not a great deal of evidence, there’s some evidence to rationalize early screening in psoriasis,” according to Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
Psoriasis develops during childhood in almost one-third of patients.
The pediatric psoriasis screening guidelines describe a simple routine screening program and timeline for early identification of overweight or obesity, type 2 diabetes, hypertension, nonalcoholic fatty liver disease, anxiety, depression, substance abuse, inflammatory bowel disease, and quality of life issues, all of which are encountered with increased frequency in pediatric psoriasis patients. A fasting lipid panel is recommended in children aged 9-11 years with psoriasis and again at age 17-21 years.
“Don’t forget arthritis. For a kid with psoriasis, at every office visit, I ask about morning stiffness or limp. Those are probably the two most sensitive questions in screening for psoriatic arthritis,” according to Dr. Eichenfield.
It has been clear for some time that the skin is not the only organ affected by psoriatic inflammation. The study that quantified the relationship between psoriasis duration and cardiovascular risk – a 1% increase for each year of psoriasis – was a collaboration between investigators at the University of Copenhagen and the University of Pennsylvania, Philadelphia.
The two-part project included aortal imaging of 190 psoriasis patients using fludeoxyglucose F 18 PET/CT scan, which showed a strong relationship between duration of psoriasis and the degree of vascular inflammation. This was bolstered by a population-based study using Danish national registry data on 87,161 psoriasis patients and 4.2 million controls from the general Danish population (J Am Acad Dermatol. 2017 Oct;77[4]:650-56.e3).
Dr. Eichenfield reported serving as a consultant to and/or recipient of research grants from more than a dozen pharmaceutical companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – The recent report that the risk of a major adverse cardiovascular event increases by 1% more than in the general population for each additional year of psoriasis duration is sobering news for physicians who treat pediatric psoriasis.
“If I have a 16-year-old who has a 5-year history of psoriasis, what does that mean for when she’s 30 or 40? And should we be intervening more aggressively?” Lawrence F. Eichenfield, MD, asked at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even though there’s not a great deal of evidence, there’s some evidence to rationalize early screening in psoriasis,” according to Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego and professor of dermatology and pediatrics at the University of California, San Diego.
Psoriasis develops during childhood in almost one-third of patients.
The pediatric psoriasis screening guidelines describe a simple routine screening program and timeline for early identification of overweight or obesity, type 2 diabetes, hypertension, nonalcoholic fatty liver disease, anxiety, depression, substance abuse, inflammatory bowel disease, and quality of life issues, all of which are encountered with increased frequency in pediatric psoriasis patients. A fasting lipid panel is recommended in children aged 9-11 years with psoriasis and again at age 17-21 years.
“Don’t forget arthritis. For a kid with psoriasis, at every office visit, I ask about morning stiffness or limp. Those are probably the two most sensitive questions in screening for psoriatic arthritis,” according to Dr. Eichenfield.
It has been clear for some time that the skin is not the only organ affected by psoriatic inflammation. The study that quantified the relationship between psoriasis duration and cardiovascular risk – a 1% increase for each year of psoriasis – was a collaboration between investigators at the University of Copenhagen and the University of Pennsylvania, Philadelphia.
The two-part project included aortal imaging of 190 psoriasis patients using fludeoxyglucose F 18 PET/CT scan, which showed a strong relationship between duration of psoriasis and the degree of vascular inflammation. This was bolstered by a population-based study using Danish national registry data on 87,161 psoriasis patients and 4.2 million controls from the general Danish population (J Am Acad Dermatol. 2017 Oct;77[4]:650-56.e3).
Dr. Eichenfield reported serving as a consultant to and/or recipient of research grants from more than a dozen pharmaceutical companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Is PASI 100 the new benchmark in psoriasis?
KAUAI, HAWAII – I think we should just do away with PASI 90 [90% improvement in Psoriasis Area and Severity Index score] and look at how well our drugs do against the metric of PASI 100. The whole ball of wax. Let’s just go for complete clearance,” Craig L. Leonardi, MD, declared in a provocative presentation at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
He advocates using number needed to treat (NNT) as a performance yardstick. He finds it helpful in translating sometimes-arcane clinical trial results into useful information to guide everyday practice. The NNT is the average number of patients who need to be treated with a drug or procedure in order to achieve one additional good outcome, compared with a control intervention or placebo. It’s the inverse of the absolute risk reduction. The lower the NNT, the better an intervention is performing.
He presented a chart that summarized the NNTs to achieve a PASI 100 response for various systemic agents commonly used in treating moderate to severe psoriasis. He obtained the data from Food and Drug Administration–regulated product labeling and phase 3 clinical trials.
Dr. Leonardi drew attention to the worst performers on the list: methotrexate, with an NNT of 25 to achieve a PASI 100 response, and etanercept, with an NNT of 23.3.
“Methotrexate is a drug that the insurance industry says we have to flow through on our way to biologic drugs. But if complete clearance is your goal, this is an exercise in futility. These patients will never, ever get to complete clearance – or it’s at least very unlikely. We shouldn’t be asked to go through methotrexate on our way to anything. We shouldn’t be asked to use methotrexate at all. We should be bypassing it. And some of us are working on this,” he said.
Ustekinumab and adalimumab are the current market leaders in biologic therapy for psoriasis, but they don’t stack up so well when viewed through the filter of PASI 100 response, with NNTs of 9.2 and 5.3, respectively.
“These market leaders may not be the most relevant drugs in the current era,” according to the dermatologist.
In contrast, the high-performance biologics – the interleukin-17 inhibitors secukinumab, ixekizumab, and brodalumab and the interleukin-23 antagonist guselkumab – have impressively low NNTs of 2.4-3.6 in order to achieve complete clearance.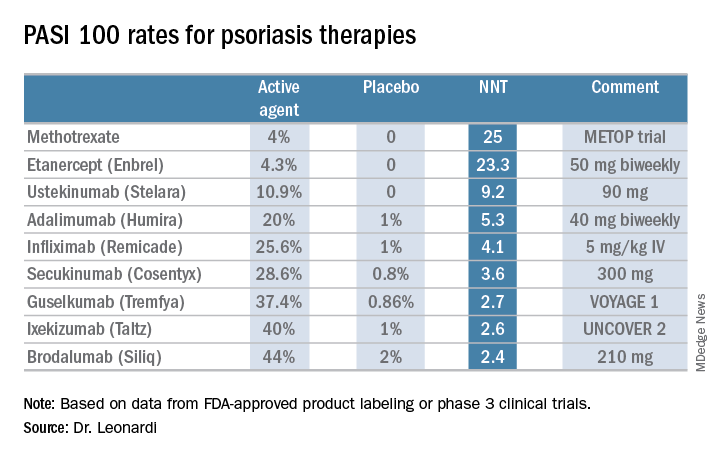
“But our IL-17 and IL-23 antagonists are markedly different from all other therapies, with NNTs of 1.3-1.1. With an NNT of 1.1, if you treated 11 patients with ixekizumab, 10 of them would achieve a PASI 75,” he explained.
“This is really quite remarkable,” Dr. Leonardi commented. “Our first drug back in 2002 was alefacept, and that drug was a ‘twenty-one percenter’: 21% of patients achieved a PASI 75. And quite frankly, we thought that was rocking voodoo science back in the day. Well, we’re really out there now. This is utterly amazing data: a PASI 75 of 81.6% for secukinumab, 86% for brodalumab, 90% for ixekizumab, and 91.2% for guselkumab. This is why we’re publishing this stuff in the best medical journals, because these results are absolutely amazing. So many different medical specialties are interested in what we’re doing with these drugs.”
He reported serving as a consultant to AbbVie, Amgen, Boehringer Ingelheim, Dermira, Eli Lilly, Janssen, Leo, Pfizer, Sandoz, and UCB and receiving research funding from 21 pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – I think we should just do away with PASI 90 [90% improvement in Psoriasis Area and Severity Index score] and look at how well our drugs do against the metric of PASI 100. The whole ball of wax. Let’s just go for complete clearance,” Craig L. Leonardi, MD, declared in a provocative presentation at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
He advocates using number needed to treat (NNT) as a performance yardstick. He finds it helpful in translating sometimes-arcane clinical trial results into useful information to guide everyday practice. The NNT is the average number of patients who need to be treated with a drug or procedure in order to achieve one additional good outcome, compared with a control intervention or placebo. It’s the inverse of the absolute risk reduction. The lower the NNT, the better an intervention is performing.
He presented a chart that summarized the NNTs to achieve a PASI 100 response for various systemic agents commonly used in treating moderate to severe psoriasis. He obtained the data from Food and Drug Administration–regulated product labeling and phase 3 clinical trials.
Dr. Leonardi drew attention to the worst performers on the list: methotrexate, with an NNT of 25 to achieve a PASI 100 response, and etanercept, with an NNT of 23.3.
“Methotrexate is a drug that the insurance industry says we have to flow through on our way to biologic drugs. But if complete clearance is your goal, this is an exercise in futility. These patients will never, ever get to complete clearance – or it’s at least very unlikely. We shouldn’t be asked to go through methotrexate on our way to anything. We shouldn’t be asked to use methotrexate at all. We should be bypassing it. And some of us are working on this,” he said.
Ustekinumab and adalimumab are the current market leaders in biologic therapy for psoriasis, but they don’t stack up so well when viewed through the filter of PASI 100 response, with NNTs of 9.2 and 5.3, respectively.
“These market leaders may not be the most relevant drugs in the current era,” according to the dermatologist.
In contrast, the high-performance biologics – the interleukin-17 inhibitors secukinumab, ixekizumab, and brodalumab and the interleukin-23 antagonist guselkumab – have impressively low NNTs of 2.4-3.6 in order to achieve complete clearance.
“But our IL-17 and IL-23 antagonists are markedly different from all other therapies, with NNTs of 1.3-1.1. With an NNT of 1.1, if you treated 11 patients with ixekizumab, 10 of them would achieve a PASI 75,” he explained.
“This is really quite remarkable,” Dr. Leonardi commented. “Our first drug back in 2002 was alefacept, and that drug was a ‘twenty-one percenter’: 21% of patients achieved a PASI 75. And quite frankly, we thought that was rocking voodoo science back in the day. Well, we’re really out there now. This is utterly amazing data: a PASI 75 of 81.6% for secukinumab, 86% for brodalumab, 90% for ixekizumab, and 91.2% for guselkumab. This is why we’re publishing this stuff in the best medical journals, because these results are absolutely amazing. So many different medical specialties are interested in what we’re doing with these drugs.”
He reported serving as a consultant to AbbVie, Amgen, Boehringer Ingelheim, Dermira, Eli Lilly, Janssen, Leo, Pfizer, Sandoz, and UCB and receiving research funding from 21 pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – I think we should just do away with PASI 90 [90% improvement in Psoriasis Area and Severity Index score] and look at how well our drugs do against the metric of PASI 100. The whole ball of wax. Let’s just go for complete clearance,” Craig L. Leonardi, MD, declared in a provocative presentation at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
He advocates using number needed to treat (NNT) as a performance yardstick. He finds it helpful in translating sometimes-arcane clinical trial results into useful information to guide everyday practice. The NNT is the average number of patients who need to be treated with a drug or procedure in order to achieve one additional good outcome, compared with a control intervention or placebo. It’s the inverse of the absolute risk reduction. The lower the NNT, the better an intervention is performing.
He presented a chart that summarized the NNTs to achieve a PASI 100 response for various systemic agents commonly used in treating moderate to severe psoriasis. He obtained the data from Food and Drug Administration–regulated product labeling and phase 3 clinical trials.
Dr. Leonardi drew attention to the worst performers on the list: methotrexate, with an NNT of 25 to achieve a PASI 100 response, and etanercept, with an NNT of 23.3.
“Methotrexate is a drug that the insurance industry says we have to flow through on our way to biologic drugs. But if complete clearance is your goal, this is an exercise in futility. These patients will never, ever get to complete clearance – or it’s at least very unlikely. We shouldn’t be asked to go through methotrexate on our way to anything. We shouldn’t be asked to use methotrexate at all. We should be bypassing it. And some of us are working on this,” he said.
Ustekinumab and adalimumab are the current market leaders in biologic therapy for psoriasis, but they don’t stack up so well when viewed through the filter of PASI 100 response, with NNTs of 9.2 and 5.3, respectively.
“These market leaders may not be the most relevant drugs in the current era,” according to the dermatologist.
In contrast, the high-performance biologics – the interleukin-17 inhibitors secukinumab, ixekizumab, and brodalumab and the interleukin-23 antagonist guselkumab – have impressively low NNTs of 2.4-3.6 in order to achieve complete clearance.
“But our IL-17 and IL-23 antagonists are markedly different from all other therapies, with NNTs of 1.3-1.1. With an NNT of 1.1, if you treated 11 patients with ixekizumab, 10 of them would achieve a PASI 75,” he explained.
“This is really quite remarkable,” Dr. Leonardi commented. “Our first drug back in 2002 was alefacept, and that drug was a ‘twenty-one percenter’: 21% of patients achieved a PASI 75. And quite frankly, we thought that was rocking voodoo science back in the day. Well, we’re really out there now. This is utterly amazing data: a PASI 75 of 81.6% for secukinumab, 86% for brodalumab, 90% for ixekizumab, and 91.2% for guselkumab. This is why we’re publishing this stuff in the best medical journals, because these results are absolutely amazing. So many different medical specialties are interested in what we’re doing with these drugs.”
He reported serving as a consultant to AbbVie, Amgen, Boehringer Ingelheim, Dermira, Eli Lilly, Janssen, Leo, Pfizer, Sandoz, and UCB and receiving research funding from 21 pharmaceutical companies.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Surgical excision essential in severe hidradenitis suppurativa
KAUAI, HAWAII – Medical therapy alone is never sufficient in Hurley stage III hidradenitis suppurativa (HS), Iltefat H. Hamzavi, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even with the advances in biologics and antibiotic therapy, you still have to excise once you’re in full-blown Hurley stage III disease. Surgery has to be part of your protocol,” according to Dr. Hamzavi, a dermatologist at Henry Ford Hospital in Detroit, which runs one of the nation’s largest hidradenitis suppurativa clinics, with roughly 1,600 patients.
“Of course we’re biased. But until the data can set us free, you’re stuck with me,” the dermatologist quipped.
A core principle of the Henry Ford algorithm is this: “Medical therapy [for patients with advanced HS] stabilizes them and reduces their draining and pain, then you try to bring them back to a lower stage with surgical options,” he explained.
Although other HS staging systems exist, Dr. Hamzavi and his colleagues rely on the Hurley staging system to guide their treatment. Basically, Hurley stage I consists of follicular nodules and abscesses. When the nodules connect to form sinus tracts with scarring, that’s stage II. And if the sinus tracts interconnect throughout an entire area, that’s stage III.
Hurley stage I
First-line treatment of localized Hurley stage I disease at Henry Ford is a 10% topical benzoyl peroxide wash left on for 5 minutes before bathing, followed by postbathing topical clindamycin 1% lotion or solution applied to the nodules. If this maintenance regimen isn’t sufficient to prevent formation of new and worsening nodules, Dr. Hamzavi supplements it with up to three once-monthly 1064-nm Nd:YAG laser sessions aimed at follicular ablation. It’s a laser application he and his colleagues pioneered (Dermatol Surg. 2009 Aug;35[8]:1188-98). They subsequently documented the histopathologic basis of the procedure’s efficacy, which entails selective thermolysis of follicles, destruction of inflammatory lesions in the superficial to mid-dermis, followed by fibrosis and scarring (Arch Dermatol. 2011 Jan;147[1]:21-8).
In generalized Hurley stage I HS, the Henry Ford approach is to supplement the topical regimen and laser sessions with oral doxycycline at 100-150 mg daily for 1-6 months.
“The theory here is this is a dysbiotic event. The antibiotics reduce commensal bacteria, which ultimately reduces the reactive inflammatory response. But when you stop the antibiotics, the inflammatory response returns. So antibiotics can help stabilize the disease state but really can’t reverse the disease state. For that we have to turn to ablative treatment options: laser, surgery,” the dermatologist continued.
Hurley stage II
“At this point you’re looking at procedures,” according to Dr. Hamzavi. “Once you have sinus tracts it’s critical to remove them.”
The treatment backbone in stage II disease is 8-10 weeks of oral clindamycin and rifampin, both at 300 mg twice daily.
“This is one of the fundamental building blocks of HS clinics throughout the world,” he noted.
Clostridium difficile infection is exceedingly rare in HS patients on this regimen, for reasons still unclear.
If this dual-antibiotic regimen doesn’t dramatically reduce drainage and pain, he adds levofloxacin at 500 mg twice daily for up to 2 weeks in an effort to calm down unstable, decompensating disease.
Dapsone at 50-150 mg/day for up to 12 weeks is an additional option. It’s most useful in patients with nodules that are disproportionately painful, in Dr. Hamzavi’s experience.
Deroofing is a simple procedure that should be considered for all sinus tracts. It entails numbing the area with a ring block then introducing a curette or surgical probe into the sinus tract to open it up and get rid of the gelatinous material within. Dutch investigators have detailed the technique (J Am Acad Dermatol. 2010 Sep;63[3]:475-80).
Tumor necrosis factor–inhibitor therapy has been a major advance in Hurley stage II and III disease. “It doesn’t work in everybody, but a lot of patients can be stabilized,” Dr. Hamzavi observed.
Efficacy has been amply demonstrated for adalimumab (Humira) and infliximab (Remicade). In Dr. Hamzavi’s experience infliximab works better, probably because it offers more dosing options.
Assuming medical therapy has resulted in disease stabilization, CO2 laser excision of sinus tracts under local anesthesia can then be employed as an office procedure to turn back the clock and return to an earlier stage of disease. Dermatologists at the Cleveland Clinic have described the technique in detail (Dermatol Surg. 2010 Feb;36[2]:208-13).
Hurley stage III
If biologic therapy doesn’t bring disease stabilization, the patient is likely headed for surgical excision using the CO2 laser. The Henry Ford team favors a specific regimen of surgical preparation using wide-spectrum antibiotics. The program begins with 6 weeks of IV ertapenem at 1 g/day delivered by a peripherally inserted central catheter managed by infectious disease colleagues.
“IV ertapenem is a drug you may not know much about. We find it works really well as a great way to bridge patients towards surgery,” the dermatologist explained.
The IV ertapenem is followed by 6 weeks of oral triple therapy with rifampin, moxifloxacin, and metronidazole then another 6 weeks of rifampin plus moxifloxacin. Next it’s surgical excision time.
Lifestyle modification
Lifestyle modification deserves to be a major priority in all HS patients, regardless of Hurley stage. Smoking cessation results in significantly greater likelihood of favorable response to first-line therapy. In obese patients, greater than 15% weight loss has been associated with significant reduction in disease severity. A sartorial shift to loose-fitting clothing can quiet down skin lesions through decreased friction and pressure. And proper utilization of warm compression will rapidly decrease acute lesional pain.
Dr. Hamzavi and his coinvestigators have described the Henry Ford Hospital treatment algorithm in a review of HS published in an open-access journal meant to serve as a resource for patients and physicians alike (F1000Res. 2017 Jul 28;6:1272. doi: 10.12688/f1000research.11337.1. eCollection 2017).
He reported serving as a consultant to AbbVie, Incyte, and UCB.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
KAUAI, HAWAII – Medical therapy alone is never sufficient in Hurley stage III hidradenitis suppurativa (HS), Iltefat H. Hamzavi, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even with the advances in biologics and antibiotic therapy, you still have to excise once you’re in full-blown Hurley stage III disease. Surgery has to be part of your protocol,” according to Dr. Hamzavi, a dermatologist at Henry Ford Hospital in Detroit, which runs one of the nation’s largest hidradenitis suppurativa clinics, with roughly 1,600 patients.
“Of course we’re biased. But until the data can set us free, you’re stuck with me,” the dermatologist quipped.
A core principle of the Henry Ford algorithm is this: “Medical therapy [for patients with advanced HS] stabilizes them and reduces their draining and pain, then you try to bring them back to a lower stage with surgical options,” he explained.
Although other HS staging systems exist, Dr. Hamzavi and his colleagues rely on the Hurley staging system to guide their treatment. Basically, Hurley stage I consists of follicular nodules and abscesses. When the nodules connect to form sinus tracts with scarring, that’s stage II. And if the sinus tracts interconnect throughout an entire area, that’s stage III.
Hurley stage I
First-line treatment of localized Hurley stage I disease at Henry Ford is a 10% topical benzoyl peroxide wash left on for 5 minutes before bathing, followed by postbathing topical clindamycin 1% lotion or solution applied to the nodules. If this maintenance regimen isn’t sufficient to prevent formation of new and worsening nodules, Dr. Hamzavi supplements it with up to three once-monthly 1064-nm Nd:YAG laser sessions aimed at follicular ablation. It’s a laser application he and his colleagues pioneered (Dermatol Surg. 2009 Aug;35[8]:1188-98). They subsequently documented the histopathologic basis of the procedure’s efficacy, which entails selective thermolysis of follicles, destruction of inflammatory lesions in the superficial to mid-dermis, followed by fibrosis and scarring (Arch Dermatol. 2011 Jan;147[1]:21-8).
In generalized Hurley stage I HS, the Henry Ford approach is to supplement the topical regimen and laser sessions with oral doxycycline at 100-150 mg daily for 1-6 months.
“The theory here is this is a dysbiotic event. The antibiotics reduce commensal bacteria, which ultimately reduces the reactive inflammatory response. But when you stop the antibiotics, the inflammatory response returns. So antibiotics can help stabilize the disease state but really can’t reverse the disease state. For that we have to turn to ablative treatment options: laser, surgery,” the dermatologist continued.
Hurley stage II
“At this point you’re looking at procedures,” according to Dr. Hamzavi. “Once you have sinus tracts it’s critical to remove them.”
The treatment backbone in stage II disease is 8-10 weeks of oral clindamycin and rifampin, both at 300 mg twice daily.
“This is one of the fundamental building blocks of HS clinics throughout the world,” he noted.
Clostridium difficile infection is exceedingly rare in HS patients on this regimen, for reasons still unclear.
If this dual-antibiotic regimen doesn’t dramatically reduce drainage and pain, he adds levofloxacin at 500 mg twice daily for up to 2 weeks in an effort to calm down unstable, decompensating disease.
Dapsone at 50-150 mg/day for up to 12 weeks is an additional option. It’s most useful in patients with nodules that are disproportionately painful, in Dr. Hamzavi’s experience.
Deroofing is a simple procedure that should be considered for all sinus tracts. It entails numbing the area with a ring block then introducing a curette or surgical probe into the sinus tract to open it up and get rid of the gelatinous material within. Dutch investigators have detailed the technique (J Am Acad Dermatol. 2010 Sep;63[3]:475-80).
Tumor necrosis factor–inhibitor therapy has been a major advance in Hurley stage II and III disease. “It doesn’t work in everybody, but a lot of patients can be stabilized,” Dr. Hamzavi observed.
Efficacy has been amply demonstrated for adalimumab (Humira) and infliximab (Remicade). In Dr. Hamzavi’s experience infliximab works better, probably because it offers more dosing options.
Assuming medical therapy has resulted in disease stabilization, CO2 laser excision of sinus tracts under local anesthesia can then be employed as an office procedure to turn back the clock and return to an earlier stage of disease. Dermatologists at the Cleveland Clinic have described the technique in detail (Dermatol Surg. 2010 Feb;36[2]:208-13).
Hurley stage III
If biologic therapy doesn’t bring disease stabilization, the patient is likely headed for surgical excision using the CO2 laser. The Henry Ford team favors a specific regimen of surgical preparation using wide-spectrum antibiotics. The program begins with 6 weeks of IV ertapenem at 1 g/day delivered by a peripherally inserted central catheter managed by infectious disease colleagues.
“IV ertapenem is a drug you may not know much about. We find it works really well as a great way to bridge patients towards surgery,” the dermatologist explained.
The IV ertapenem is followed by 6 weeks of oral triple therapy with rifampin, moxifloxacin, and metronidazole then another 6 weeks of rifampin plus moxifloxacin. Next it’s surgical excision time.
Lifestyle modification
Lifestyle modification deserves to be a major priority in all HS patients, regardless of Hurley stage. Smoking cessation results in significantly greater likelihood of favorable response to first-line therapy. In obese patients, greater than 15% weight loss has been associated with significant reduction in disease severity. A sartorial shift to loose-fitting clothing can quiet down skin lesions through decreased friction and pressure. And proper utilization of warm compression will rapidly decrease acute lesional pain.
Dr. Hamzavi and his coinvestigators have described the Henry Ford Hospital treatment algorithm in a review of HS published in an open-access journal meant to serve as a resource for patients and physicians alike (F1000Res. 2017 Jul 28;6:1272. doi: 10.12688/f1000research.11337.1. eCollection 2017).
He reported serving as a consultant to AbbVie, Incyte, and UCB.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
KAUAI, HAWAII – Medical therapy alone is never sufficient in Hurley stage III hidradenitis suppurativa (HS), Iltefat H. Hamzavi, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
“Even with the advances in biologics and antibiotic therapy, you still have to excise once you’re in full-blown Hurley stage III disease. Surgery has to be part of your protocol,” according to Dr. Hamzavi, a dermatologist at Henry Ford Hospital in Detroit, which runs one of the nation’s largest hidradenitis suppurativa clinics, with roughly 1,600 patients.
“Of course we’re biased. But until the data can set us free, you’re stuck with me,” the dermatologist quipped.
A core principle of the Henry Ford algorithm is this: “Medical therapy [for patients with advanced HS] stabilizes them and reduces their draining and pain, then you try to bring them back to a lower stage with surgical options,” he explained.
Although other HS staging systems exist, Dr. Hamzavi and his colleagues rely on the Hurley staging system to guide their treatment. Basically, Hurley stage I consists of follicular nodules and abscesses. When the nodules connect to form sinus tracts with scarring, that’s stage II. And if the sinus tracts interconnect throughout an entire area, that’s stage III.
Hurley stage I
First-line treatment of localized Hurley stage I disease at Henry Ford is a 10% topical benzoyl peroxide wash left on for 5 minutes before bathing, followed by postbathing topical clindamycin 1% lotion or solution applied to the nodules. If this maintenance regimen isn’t sufficient to prevent formation of new and worsening nodules, Dr. Hamzavi supplements it with up to three once-monthly 1064-nm Nd:YAG laser sessions aimed at follicular ablation. It’s a laser application he and his colleagues pioneered (Dermatol Surg. 2009 Aug;35[8]:1188-98). They subsequently documented the histopathologic basis of the procedure’s efficacy, which entails selective thermolysis of follicles, destruction of inflammatory lesions in the superficial to mid-dermis, followed by fibrosis and scarring (Arch Dermatol. 2011 Jan;147[1]:21-8).
In generalized Hurley stage I HS, the Henry Ford approach is to supplement the topical regimen and laser sessions with oral doxycycline at 100-150 mg daily for 1-6 months.
“The theory here is this is a dysbiotic event. The antibiotics reduce commensal bacteria, which ultimately reduces the reactive inflammatory response. But when you stop the antibiotics, the inflammatory response returns. So antibiotics can help stabilize the disease state but really can’t reverse the disease state. For that we have to turn to ablative treatment options: laser, surgery,” the dermatologist continued.
Hurley stage II
“At this point you’re looking at procedures,” according to Dr. Hamzavi. “Once you have sinus tracts it’s critical to remove them.”
The treatment backbone in stage II disease is 8-10 weeks of oral clindamycin and rifampin, both at 300 mg twice daily.
“This is one of the fundamental building blocks of HS clinics throughout the world,” he noted.
Clostridium difficile infection is exceedingly rare in HS patients on this regimen, for reasons still unclear.
If this dual-antibiotic regimen doesn’t dramatically reduce drainage and pain, he adds levofloxacin at 500 mg twice daily for up to 2 weeks in an effort to calm down unstable, decompensating disease.
Dapsone at 50-150 mg/day for up to 12 weeks is an additional option. It’s most useful in patients with nodules that are disproportionately painful, in Dr. Hamzavi’s experience.
Deroofing is a simple procedure that should be considered for all sinus tracts. It entails numbing the area with a ring block then introducing a curette or surgical probe into the sinus tract to open it up and get rid of the gelatinous material within. Dutch investigators have detailed the technique (J Am Acad Dermatol. 2010 Sep;63[3]:475-80).
Tumor necrosis factor–inhibitor therapy has been a major advance in Hurley stage II and III disease. “It doesn’t work in everybody, but a lot of patients can be stabilized,” Dr. Hamzavi observed.
Efficacy has been amply demonstrated for adalimumab (Humira) and infliximab (Remicade). In Dr. Hamzavi’s experience infliximab works better, probably because it offers more dosing options.
Assuming medical therapy has resulted in disease stabilization, CO2 laser excision of sinus tracts under local anesthesia can then be employed as an office procedure to turn back the clock and return to an earlier stage of disease. Dermatologists at the Cleveland Clinic have described the technique in detail (Dermatol Surg. 2010 Feb;36[2]:208-13).
Hurley stage III
If biologic therapy doesn’t bring disease stabilization, the patient is likely headed for surgical excision using the CO2 laser. The Henry Ford team favors a specific regimen of surgical preparation using wide-spectrum antibiotics. The program begins with 6 weeks of IV ertapenem at 1 g/day delivered by a peripherally inserted central catheter managed by infectious disease colleagues.
“IV ertapenem is a drug you may not know much about. We find it works really well as a great way to bridge patients towards surgery,” the dermatologist explained.
The IV ertapenem is followed by 6 weeks of oral triple therapy with rifampin, moxifloxacin, and metronidazole then another 6 weeks of rifampin plus moxifloxacin. Next it’s surgical excision time.
Lifestyle modification
Lifestyle modification deserves to be a major priority in all HS patients, regardless of Hurley stage. Smoking cessation results in significantly greater likelihood of favorable response to first-line therapy. In obese patients, greater than 15% weight loss has been associated with significant reduction in disease severity. A sartorial shift to loose-fitting clothing can quiet down skin lesions through decreased friction and pressure. And proper utilization of warm compression will rapidly decrease acute lesional pain.
Dr. Hamzavi and his coinvestigators have described the Henry Ford Hospital treatment algorithm in a review of HS published in an open-access journal meant to serve as a resource for patients and physicians alike (F1000Res. 2017 Jul 28;6:1272. doi: 10.12688/f1000research.11337.1. eCollection 2017).
He reported serving as a consultant to AbbVie, Incyte, and UCB.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
How to avoid severe diarrhea from apremilast
KAUAI, HAWAII – Physicians have become much more cognizant of severe diarrhea and nausea as potential side effects of apremilast since the Food and Drug Administration–approved change in the warnings and precautions section of the drug’s labeling in June 2017. Jashin J. Wu, MD, director of the psoriasis clinic at Kaiser Permanente Los Angeles Medical Center, has a tip for avoiding these problems: Delay up-titrating.
“In my opinion, that may be too quick of an up-titration. I tell patients that, if they feel the GI issues are still a problem for them on day 6, they should take 30 mg just once a day for the first 1-2 months. After that we’ll see how they’re doing, and if they feel they can make the jump to twice a day, then they can go for it. Of course, I also tell them that maybe their psoriasis will not clear as well as if they’d been on apremilast twice a day right from day 6, but if they’re able to tolerate it and can continue to take it, they can improve while they’re on it,” the dermatologist said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wu presented an update on recent developments regarding the newest oral drugs for psoriasis and one of the oldest: apremilast and methotrexate, respectively.
Apremilast
The revised warning label highlighting the risks of severe diarrhea and nausea associated with the oral phosphodiesterase-4 inhibitor says that most such events have occurred within the first few weeks of therapy. The guidance also notes that patients who reduced the dosage or discontinued treatment outright generally improved rapidly.
“I see this in a lot of my patients. They have to go to the bathroom pretty often. It’s actually unusual for me for a patient not to have any GI issues at all,” according to Dr. Wu.
He shared a number of other fresh insights into apremilast’s safety and efficacy derived from recent studies.
Efficacy appears to increase at least out to 1 year
A report from the phase 3b, randomized, placebo-controlled, 250-patient LIBERATE trial showed that the week 16 PASI 75 response rate was 39.8% with apremilast, 48.2% with etanercept (Enbrel), and 11.9% with placebo. After week 16, everyone switched to apremilast. The PASI 75 rate in patients on apremilast all along climbed from 39.8% at week 16 to 52.7% at week 52. That result was in the same ballpark as the 57% rate in those switched from etanercept to apremilast and the 53.4% PASI 75 rate at week 52 in patients switched from placebo to apremilast (J Eur Acad Dermatol Venereol. 2017 Mar;31[3]:507-17).
“It was interesting to see that, as the study continued on for 1 year, the PASI 75 rate continued to improve. That’s worth noting: In general, I tell patients you have to be on a drug for about 3 months before we’re going to say if it worked or not, and that’s true even with drugs for other conditions, like doxycycline for acne. But this study seems to indicate that you have much better improvement at the 1-year point, and that’s not so much true for the biologics,” the dermatologist observed.
Safety to 3 years looks reassuringly good: 3-year follow-up of the 1,184-patient, phase 3, randomized, controlled ESTEEM 1 and 2 trials provided by far the longest look to date at apremilast’s safety. There were no surprises, no serious opportunistic infections, and no significant changes in laboratory values (J Am Acad Dermatol. 2017 Aug;77[2]:310-17.e1).
Of note, 21.9% of patients on apremilast lost more than 5% of their baseline body weight. Most of the weight loss occurred during year 1 of treatment and mostly in patients with a higher baseline body mass index.
“It seems like apremilast is definitely a good option if patients can tolerate the GI upset,” Dr. Wu said.
Apremilast can safely and effectively be combined with other psoriasis therapies: Dermatologists at the University of Toronto reported on a retrospective analysis of 81 biologic-naive psoriasis patients treated with apremilast in combination with methotrexate, acitretin (Soriatane), cyclosporine, narrowband UVB, etanercept, infliximab (Remicade), adalimumab (Humira), and/or ustekinumab (Stelara). Of these patients, 81% achieved a PASI 75 response at week 12 (J Cutan Med Surg. 2016 Jul;20[4]:313-6).
“That’s pretty good. It’s certainly better than apremilast by itself. So if you can get the payer to cover a combination of apremilast and something else, it may help get to PASI 75,” Dr. Wu noted.
Session chair Craig L. Leonardi, MD, said he hasn’t had any luck in going that route.
“The insurance industry just won’t give me apremilast in combination with a biologic drug. Even though it makes complete sense to use it in place of methotrexate with a biologic, I just can’t get it,” according to Dr. Leonardi, a dermatologist at Saint Louis University.
“I don’t have those limitations at Kaiser,” according to Dr. Wu. “I personally have only used apremilast and methotrexate and apremilast and acitretin in combination. I just want to be kind to Kaiser and not give two branded medications to a patient, but I certainly think it’s a feasible option.”
Methotrexate
A simple response prediction rule: Kenneth B. Gordon, MD, professor and chair of dermatology at the Medical College of Wisconsin in Milwaukee, and his coinvestigators at AbbVie developed a methotrexate response/nonresponse prediction rule using data on 110 participants in the phase 3 CHAMPION trial. Then they validated the rule in the phase 3 M10-255 trial. They found that a PASI 25 response to methotrexate at week 4 was associated with an 8.9-fold increased likelihood of a week-16 PASI 75 response. Patients with a predicted response probability of less than 30% were asked to discontinue the drug; their week 16 PASI 75 rate was only 21.1%, compared with a 65.8% response rate in patients with a prediction rating (J Am Acad Dermatol. 2017 Dec;77[6]:1030-7).
“Four weeks of methotrexate may be sufficient to determine the long-term response. It may not be necessary to put them on the drug for 3 months,” Dr. Wu commented.
Subcutaneous methotrexate: European investigators demonstrated that an intensified dosing schedule of subcutaneous methotrexate was safe and effective for treatment of moderate to severe plaque psoriasis in the 52-week, phase 3, randomized, 16-center, double-blind, 120-patient, placebo-controlled METOP study.
The intensified subcutaneous regimen consisted of 17.5 mg/week initially, escalated to 22.5 mg/week after 8 weeks if a patient hadn’t achieved at least a PASI 50 response at that point. The primary outcome, the PASI 75 response at week 16, was 41% in the subcutaneous methotrexate group and 10% in controls, with a maximum PASI 75 rate of 51% seen beginning at week 24. The week 4 and 8 PASI 50 rates were 50% and 58%, respectively, with methotrexate versus 3% and 17% in placebo-treated controls. The subcutaneous regimen was generally well tolerated, with no serious infections or malignancies arising during 52 weeks (Lancet. 2017 Feb 4;389[10068]:528-37).
Dr. Wu reported receiving research funding from AbbVie, Amgen, Eli Lilly, Janssen, Novartis, and Regeneron.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
KAUAI, HAWAII – Physicians have become much more cognizant of severe diarrhea and nausea as potential side effects of apremilast since the Food and Drug Administration–approved change in the warnings and precautions section of the drug’s labeling in June 2017. Jashin J. Wu, MD, director of the psoriasis clinic at Kaiser Permanente Los Angeles Medical Center, has a tip for avoiding these problems: Delay up-titrating.
“In my opinion, that may be too quick of an up-titration. I tell patients that, if they feel the GI issues are still a problem for them on day 6, they should take 30 mg just once a day for the first 1-2 months. After that we’ll see how they’re doing, and if they feel they can make the jump to twice a day, then they can go for it. Of course, I also tell them that maybe their psoriasis will not clear as well as if they’d been on apremilast twice a day right from day 6, but if they’re able to tolerate it and can continue to take it, they can improve while they’re on it,” the dermatologist said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wu presented an update on recent developments regarding the newest oral drugs for psoriasis and one of the oldest: apremilast and methotrexate, respectively.
Apremilast
The revised warning label highlighting the risks of severe diarrhea and nausea associated with the oral phosphodiesterase-4 inhibitor says that most such events have occurred within the first few weeks of therapy. The guidance also notes that patients who reduced the dosage or discontinued treatment outright generally improved rapidly.
“I see this in a lot of my patients. They have to go to the bathroom pretty often. It’s actually unusual for me for a patient not to have any GI issues at all,” according to Dr. Wu.
He shared a number of other fresh insights into apremilast’s safety and efficacy derived from recent studies.
Efficacy appears to increase at least out to 1 year
A report from the phase 3b, randomized, placebo-controlled, 250-patient LIBERATE trial showed that the week 16 PASI 75 response rate was 39.8% with apremilast, 48.2% with etanercept (Enbrel), and 11.9% with placebo. After week 16, everyone switched to apremilast. The PASI 75 rate in patients on apremilast all along climbed from 39.8% at week 16 to 52.7% at week 52. That result was in the same ballpark as the 57% rate in those switched from etanercept to apremilast and the 53.4% PASI 75 rate at week 52 in patients switched from placebo to apremilast (J Eur Acad Dermatol Venereol. 2017 Mar;31[3]:507-17).
“It was interesting to see that, as the study continued on for 1 year, the PASI 75 rate continued to improve. That’s worth noting: In general, I tell patients you have to be on a drug for about 3 months before we’re going to say if it worked or not, and that’s true even with drugs for other conditions, like doxycycline for acne. But this study seems to indicate that you have much better improvement at the 1-year point, and that’s not so much true for the biologics,” the dermatologist observed.
Safety to 3 years looks reassuringly good: 3-year follow-up of the 1,184-patient, phase 3, randomized, controlled ESTEEM 1 and 2 trials provided by far the longest look to date at apremilast’s safety. There were no surprises, no serious opportunistic infections, and no significant changes in laboratory values (J Am Acad Dermatol. 2017 Aug;77[2]:310-17.e1).
Of note, 21.9% of patients on apremilast lost more than 5% of their baseline body weight. Most of the weight loss occurred during year 1 of treatment and mostly in patients with a higher baseline body mass index.
“It seems like apremilast is definitely a good option if patients can tolerate the GI upset,” Dr. Wu said.
Apremilast can safely and effectively be combined with other psoriasis therapies: Dermatologists at the University of Toronto reported on a retrospective analysis of 81 biologic-naive psoriasis patients treated with apremilast in combination with methotrexate, acitretin (Soriatane), cyclosporine, narrowband UVB, etanercept, infliximab (Remicade), adalimumab (Humira), and/or ustekinumab (Stelara). Of these patients, 81% achieved a PASI 75 response at week 12 (J Cutan Med Surg. 2016 Jul;20[4]:313-6).
“That’s pretty good. It’s certainly better than apremilast by itself. So if you can get the payer to cover a combination of apremilast and something else, it may help get to PASI 75,” Dr. Wu noted.
Session chair Craig L. Leonardi, MD, said he hasn’t had any luck in going that route.
“The insurance industry just won’t give me apremilast in combination with a biologic drug. Even though it makes complete sense to use it in place of methotrexate with a biologic, I just can’t get it,” according to Dr. Leonardi, a dermatologist at Saint Louis University.
“I don’t have those limitations at Kaiser,” according to Dr. Wu. “I personally have only used apremilast and methotrexate and apremilast and acitretin in combination. I just want to be kind to Kaiser and not give two branded medications to a patient, but I certainly think it’s a feasible option.”
Methotrexate
A simple response prediction rule: Kenneth B. Gordon, MD, professor and chair of dermatology at the Medical College of Wisconsin in Milwaukee, and his coinvestigators at AbbVie developed a methotrexate response/nonresponse prediction rule using data on 110 participants in the phase 3 CHAMPION trial. Then they validated the rule in the phase 3 M10-255 trial. They found that a PASI 25 response to methotrexate at week 4 was associated with an 8.9-fold increased likelihood of a week-16 PASI 75 response. Patients with a predicted response probability of less than 30% were asked to discontinue the drug; their week 16 PASI 75 rate was only 21.1%, compared with a 65.8% response rate in patients with a prediction rating (J Am Acad Dermatol. 2017 Dec;77[6]:1030-7).
“Four weeks of methotrexate may be sufficient to determine the long-term response. It may not be necessary to put them on the drug for 3 months,” Dr. Wu commented.
Subcutaneous methotrexate: European investigators demonstrated that an intensified dosing schedule of subcutaneous methotrexate was safe and effective for treatment of moderate to severe plaque psoriasis in the 52-week, phase 3, randomized, 16-center, double-blind, 120-patient, placebo-controlled METOP study.
The intensified subcutaneous regimen consisted of 17.5 mg/week initially, escalated to 22.5 mg/week after 8 weeks if a patient hadn’t achieved at least a PASI 50 response at that point. The primary outcome, the PASI 75 response at week 16, was 41% in the subcutaneous methotrexate group and 10% in controls, with a maximum PASI 75 rate of 51% seen beginning at week 24. The week 4 and 8 PASI 50 rates were 50% and 58%, respectively, with methotrexate versus 3% and 17% in placebo-treated controls. The subcutaneous regimen was generally well tolerated, with no serious infections or malignancies arising during 52 weeks (Lancet. 2017 Feb 4;389[10068]:528-37).
Dr. Wu reported receiving research funding from AbbVie, Amgen, Eli Lilly, Janssen, Novartis, and Regeneron.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
KAUAI, HAWAII – Physicians have become much more cognizant of severe diarrhea and nausea as potential side effects of apremilast since the Food and Drug Administration–approved change in the warnings and precautions section of the drug’s labeling in June 2017. Jashin J. Wu, MD, director of the psoriasis clinic at Kaiser Permanente Los Angeles Medical Center, has a tip for avoiding these problems: Delay up-titrating.
“In my opinion, that may be too quick of an up-titration. I tell patients that, if they feel the GI issues are still a problem for them on day 6, they should take 30 mg just once a day for the first 1-2 months. After that we’ll see how they’re doing, and if they feel they can make the jump to twice a day, then they can go for it. Of course, I also tell them that maybe their psoriasis will not clear as well as if they’d been on apremilast twice a day right from day 6, but if they’re able to tolerate it and can continue to take it, they can improve while they’re on it,” the dermatologist said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Dr. Wu presented an update on recent developments regarding the newest oral drugs for psoriasis and one of the oldest: apremilast and methotrexate, respectively.
Apremilast
The revised warning label highlighting the risks of severe diarrhea and nausea associated with the oral phosphodiesterase-4 inhibitor says that most such events have occurred within the first few weeks of therapy. The guidance also notes that patients who reduced the dosage or discontinued treatment outright generally improved rapidly.
“I see this in a lot of my patients. They have to go to the bathroom pretty often. It’s actually unusual for me for a patient not to have any GI issues at all,” according to Dr. Wu.
He shared a number of other fresh insights into apremilast’s safety and efficacy derived from recent studies.
Efficacy appears to increase at least out to 1 year
A report from the phase 3b, randomized, placebo-controlled, 250-patient LIBERATE trial showed that the week 16 PASI 75 response rate was 39.8% with apremilast, 48.2% with etanercept (Enbrel), and 11.9% with placebo. After week 16, everyone switched to apremilast. The PASI 75 rate in patients on apremilast all along climbed from 39.8% at week 16 to 52.7% at week 52. That result was in the same ballpark as the 57% rate in those switched from etanercept to apremilast and the 53.4% PASI 75 rate at week 52 in patients switched from placebo to apremilast (J Eur Acad Dermatol Venereol. 2017 Mar;31[3]:507-17).
“It was interesting to see that, as the study continued on for 1 year, the PASI 75 rate continued to improve. That’s worth noting: In general, I tell patients you have to be on a drug for about 3 months before we’re going to say if it worked or not, and that’s true even with drugs for other conditions, like doxycycline for acne. But this study seems to indicate that you have much better improvement at the 1-year point, and that’s not so much true for the biologics,” the dermatologist observed.
Safety to 3 years looks reassuringly good: 3-year follow-up of the 1,184-patient, phase 3, randomized, controlled ESTEEM 1 and 2 trials provided by far the longest look to date at apremilast’s safety. There were no surprises, no serious opportunistic infections, and no significant changes in laboratory values (J Am Acad Dermatol. 2017 Aug;77[2]:310-17.e1).
Of note, 21.9% of patients on apremilast lost more than 5% of their baseline body weight. Most of the weight loss occurred during year 1 of treatment and mostly in patients with a higher baseline body mass index.
“It seems like apremilast is definitely a good option if patients can tolerate the GI upset,” Dr. Wu said.
Apremilast can safely and effectively be combined with other psoriasis therapies: Dermatologists at the University of Toronto reported on a retrospective analysis of 81 biologic-naive psoriasis patients treated with apremilast in combination with methotrexate, acitretin (Soriatane), cyclosporine, narrowband UVB, etanercept, infliximab (Remicade), adalimumab (Humira), and/or ustekinumab (Stelara). Of these patients, 81% achieved a PASI 75 response at week 12 (J Cutan Med Surg. 2016 Jul;20[4]:313-6).
“That’s pretty good. It’s certainly better than apremilast by itself. So if you can get the payer to cover a combination of apremilast and something else, it may help get to PASI 75,” Dr. Wu noted.
Session chair Craig L. Leonardi, MD, said he hasn’t had any luck in going that route.
“The insurance industry just won’t give me apremilast in combination with a biologic drug. Even though it makes complete sense to use it in place of methotrexate with a biologic, I just can’t get it,” according to Dr. Leonardi, a dermatologist at Saint Louis University.
“I don’t have those limitations at Kaiser,” according to Dr. Wu. “I personally have only used apremilast and methotrexate and apremilast and acitretin in combination. I just want to be kind to Kaiser and not give two branded medications to a patient, but I certainly think it’s a feasible option.”
Methotrexate
A simple response prediction rule: Kenneth B. Gordon, MD, professor and chair of dermatology at the Medical College of Wisconsin in Milwaukee, and his coinvestigators at AbbVie developed a methotrexate response/nonresponse prediction rule using data on 110 participants in the phase 3 CHAMPION trial. Then they validated the rule in the phase 3 M10-255 trial. They found that a PASI 25 response to methotrexate at week 4 was associated with an 8.9-fold increased likelihood of a week-16 PASI 75 response. Patients with a predicted response probability of less than 30% were asked to discontinue the drug; their week 16 PASI 75 rate was only 21.1%, compared with a 65.8% response rate in patients with a prediction rating (J Am Acad Dermatol. 2017 Dec;77[6]:1030-7).
“Four weeks of methotrexate may be sufficient to determine the long-term response. It may not be necessary to put them on the drug for 3 months,” Dr. Wu commented.
Subcutaneous methotrexate: European investigators demonstrated that an intensified dosing schedule of subcutaneous methotrexate was safe and effective for treatment of moderate to severe plaque psoriasis in the 52-week, phase 3, randomized, 16-center, double-blind, 120-patient, placebo-controlled METOP study.
The intensified subcutaneous regimen consisted of 17.5 mg/week initially, escalated to 22.5 mg/week after 8 weeks if a patient hadn’t achieved at least a PASI 50 response at that point. The primary outcome, the PASI 75 response at week 16, was 41% in the subcutaneous methotrexate group and 10% in controls, with a maximum PASI 75 rate of 51% seen beginning at week 24. The week 4 and 8 PASI 50 rates were 50% and 58%, respectively, with methotrexate versus 3% and 17% in placebo-treated controls. The subcutaneous regimen was generally well tolerated, with no serious infections or malignancies arising during 52 weeks (Lancet. 2017 Feb 4;389[10068]:528-37).
Dr. Wu reported receiving research funding from AbbVie, Amgen, Eli Lilly, Janssen, Novartis, and Regeneron.
The Global Academy for Medical Education/SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
New PsA questionnaire fails to beat existing early screening methods
A new screening tool for detecting early psoriatic arthritis in patients with psoriasis that combined the “most discriminative questions” from several other questionnaires performed as well as the Psoriasis Epidemiology Screening Tool (PEST) for detecting the disease.
“The CONTEST questionnaire was developed using the best performing items from three other screening questionnaires in the hope that it would perform better than its originators,” Laura Coates, MBChB, PhD, of the Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences at the University of Oxford, England, and her coauthors wrote in the Journal of the European Academy of Dermatology and Venereology. “In development this was partly correct but the current study does not support this – statistically there was no difference between PEST and CONTEST in terms of ability to detect psoriatic arthritis [PsA] in patients with psoriasis.”
The researchers found 27 patients (17%; 95% confidence interval, 12.3%-21.7%) with previously undiagnosed PsA, 71 with a different musculoskeletal disease, and 61 without musculoskeletal disease. Patients with PsA tended to be male, older, with “worse functional ability,” a similar age at onset of psoriasis, and had similar skin and nail disease severity. The sensitivity for PEST was 0.60 (95% CI, 0.42-0.78) and the specificity was 0.76 (95% CI, 0.69-0.83), while for CONTEST, the sensitivity was 0.53 (95% CI, 0.34-0.72) and the specificity was 0.71 (95% CI, 0.63-0.79). The area under the receiver operating curve confidence intervals for both screening tools were similar, with PEST having an AUC of 0.72 (95% CI, 0.61-0.84) and CONTEST having an AUC of 0.66 (95% CI, 0.54-0.77).
“The relative simplicity of the PEST questionnaire has raised concerns that the tool is not able to detect pure axial forms of the disease,” Dr. Coates and her colleagues wrote. “The CONTEST questionnaire includes items specific to back and neck pain, and so it was hoped it would better detect this subgroup. In this study this is not the case, although the numbers were small and imaging of the spine was not part of the study.”
AbbVie supported this study with an educational grant. The study was supported by the National Institute for Health Research Leeds Biomedical Research Centre. Some of the authors reported potential conflicts of interest.
SOURCE: Coates L et al. J Eur Acad Dermatol Venereol. 2018 Mar 26. doi: 10.1111/jdv.14971.
A new screening tool for detecting early psoriatic arthritis in patients with psoriasis that combined the “most discriminative questions” from several other questionnaires performed as well as the Psoriasis Epidemiology Screening Tool (PEST) for detecting the disease.
“The CONTEST questionnaire was developed using the best performing items from three other screening questionnaires in the hope that it would perform better than its originators,” Laura Coates, MBChB, PhD, of the Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences at the University of Oxford, England, and her coauthors wrote in the Journal of the European Academy of Dermatology and Venereology. “In development this was partly correct but the current study does not support this – statistically there was no difference between PEST and CONTEST in terms of ability to detect psoriatic arthritis [PsA] in patients with psoriasis.”
The researchers found 27 patients (17%; 95% confidence interval, 12.3%-21.7%) with previously undiagnosed PsA, 71 with a different musculoskeletal disease, and 61 without musculoskeletal disease. Patients with PsA tended to be male, older, with “worse functional ability,” a similar age at onset of psoriasis, and had similar skin and nail disease severity. The sensitivity for PEST was 0.60 (95% CI, 0.42-0.78) and the specificity was 0.76 (95% CI, 0.69-0.83), while for CONTEST, the sensitivity was 0.53 (95% CI, 0.34-0.72) and the specificity was 0.71 (95% CI, 0.63-0.79). The area under the receiver operating curve confidence intervals for both screening tools were similar, with PEST having an AUC of 0.72 (95% CI, 0.61-0.84) and CONTEST having an AUC of 0.66 (95% CI, 0.54-0.77).
“The relative simplicity of the PEST questionnaire has raised concerns that the tool is not able to detect pure axial forms of the disease,” Dr. Coates and her colleagues wrote. “The CONTEST questionnaire includes items specific to back and neck pain, and so it was hoped it would better detect this subgroup. In this study this is not the case, although the numbers were small and imaging of the spine was not part of the study.”
AbbVie supported this study with an educational grant. The study was supported by the National Institute for Health Research Leeds Biomedical Research Centre. Some of the authors reported potential conflicts of interest.
SOURCE: Coates L et al. J Eur Acad Dermatol Venereol. 2018 Mar 26. doi: 10.1111/jdv.14971.
A new screening tool for detecting early psoriatic arthritis in patients with psoriasis that combined the “most discriminative questions” from several other questionnaires performed as well as the Psoriasis Epidemiology Screening Tool (PEST) for detecting the disease.
“The CONTEST questionnaire was developed using the best performing items from three other screening questionnaires in the hope that it would perform better than its originators,” Laura Coates, MBChB, PhD, of the Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences at the University of Oxford, England, and her coauthors wrote in the Journal of the European Academy of Dermatology and Venereology. “In development this was partly correct but the current study does not support this – statistically there was no difference between PEST and CONTEST in terms of ability to detect psoriatic arthritis [PsA] in patients with psoriasis.”
The researchers found 27 patients (17%; 95% confidence interval, 12.3%-21.7%) with previously undiagnosed PsA, 71 with a different musculoskeletal disease, and 61 without musculoskeletal disease. Patients with PsA tended to be male, older, with “worse functional ability,” a similar age at onset of psoriasis, and had similar skin and nail disease severity. The sensitivity for PEST was 0.60 (95% CI, 0.42-0.78) and the specificity was 0.76 (95% CI, 0.69-0.83), while for CONTEST, the sensitivity was 0.53 (95% CI, 0.34-0.72) and the specificity was 0.71 (95% CI, 0.63-0.79). The area under the receiver operating curve confidence intervals for both screening tools were similar, with PEST having an AUC of 0.72 (95% CI, 0.61-0.84) and CONTEST having an AUC of 0.66 (95% CI, 0.54-0.77).
“The relative simplicity of the PEST questionnaire has raised concerns that the tool is not able to detect pure axial forms of the disease,” Dr. Coates and her colleagues wrote. “The CONTEST questionnaire includes items specific to back and neck pain, and so it was hoped it would better detect this subgroup. In this study this is not the case, although the numbers were small and imaging of the spine was not part of the study.”
AbbVie supported this study with an educational grant. The study was supported by the National Institute for Health Research Leeds Biomedical Research Centre. Some of the authors reported potential conflicts of interest.
SOURCE: Coates L et al. J Eur Acad Dermatol Venereol. 2018 Mar 26. doi: 10.1111/jdv.14971.
FROM JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Key clinical point: The CONTEST screening tool detected psoriatic arthritis in psoriasis patients as accurately as the Psoriasis Epidemiology Screening Tool.
Major finding: The sensitivity and specificity of CONTEST was 0.53 and 0.71, respectively, while PEST had a sensitivity of 0.60 and specificity of 0.76.
Study details: An observational, cross-sectional study of 159 psoriasis patients at four secondary care dermatology centers in the United Kingdom from November 2013 to March 2017.
Disclosures: AbbVie supported this study with an educational grant. The study was supported by the National Institute for Health Research Leeds Biomedical Research Centre. Some of the authors reported potential conflicts of interest.
Source: Coates L et al. J Eur Acad Dermatol Venereol. 2018 Mar 26. doi: 10.1111/jdv.14971.
AbbVie, Samsung Bioepis settle suits with delayed U.S. entry for adalimumab biosimilar
A new adalimumab biosimilar will become available in the European Union later this year, but a court settlement will keep Samsung Bioepis’ competitor off U.S. shelves until 2023.
Under the settlement, AbbVie, which manufactures adalimumab (Humira), will grant Bioepis and its partner, Biogen, a nonexclusive license to the intellectual property relating to the antibody. Bioepis’ version, dubbed SB5 (Imraldi), will enter global markets in a staggered fashion, according to an AbbVie press statement. In most countries in the European Union, the license period will begin on Oct. 16, 2018. In the United States, Samsung Bioepis’ license period will begin on June 30, 2023, according to the Abbvie statement.
Biogen and Bioepis hailed the settlement as a victory, but Imraldi won’t be the first Humira biosimilar to break into the U.S. market. Last September, AbbVie settled a similar suit with Amgen, granting patent licenses for the global use and sale of its anti–tumor necrosis factor–alpha antibody, Amgevita/Amjevita. Amgen expects to launch Amgevita in Europe on Oct. 16, 2018, and Amjevita in the United States on Jan. 31, 2023. Samsung Bioepis’ U.S. license date will not be accelerated upon Amgen’s entry.
Ian Henshaw, Biogen’s global head of biosimilars, said the deal further strengthens the company’s European biosimilars reach.
“Biogen is a leader in the emerging field of biosimilars through Samsung Bioepis, our joint venture with Samsung BioLogics,” Mr. Henshaw said in a press statement. “Biogen already markets two biosimilars in Europe and the planned introduction of Imraldi on Oct. 16 could potentially expand patient choice by offering physicians more options to meet the needs of patients while delivering significant savings to healthcare systems.”
AbbVie focused on the settlement as a global recognition of its leadership role in developing the anti-TNF-alpha antibody.
“The Samsung Bioepis settlement reflects the strength and breadth of AbbVie’s intellectual property,” Laura Schumacher, the company’s general counsel, said in the Abbvie statement. “We continue to believe biosimilars will play an important role in our healthcare system, but we also believe it is important to protect our investment in innovation. This agreement accomplishes both objectives.”
Samsung Bioepis will pay royalties to AbbVie for licensing its adalimumab patents once its biosimilar product is launched. As is the case with the prior Amgen resolution, AbbVie will not make any payments to Samsung Bioepis. “All litigation pending between the parties, as well as all litigation with Samsung Bioepis’ European partner, Biogen, will be dismissed. The precise terms of the agreements are confidential,” the Abbvie statement said.
The settlement brings to a closing a flurry of lawsuits Samsung Bioepis filed against AbbVie in 2017.
A new adalimumab biosimilar will become available in the European Union later this year, but a court settlement will keep Samsung Bioepis’ competitor off U.S. shelves until 2023.
Under the settlement, AbbVie, which manufactures adalimumab (Humira), will grant Bioepis and its partner, Biogen, a nonexclusive license to the intellectual property relating to the antibody. Bioepis’ version, dubbed SB5 (Imraldi), will enter global markets in a staggered fashion, according to an AbbVie press statement. In most countries in the European Union, the license period will begin on Oct. 16, 2018. In the United States, Samsung Bioepis’ license period will begin on June 30, 2023, according to the Abbvie statement.
Biogen and Bioepis hailed the settlement as a victory, but Imraldi won’t be the first Humira biosimilar to break into the U.S. market. Last September, AbbVie settled a similar suit with Amgen, granting patent licenses for the global use and sale of its anti–tumor necrosis factor–alpha antibody, Amgevita/Amjevita. Amgen expects to launch Amgevita in Europe on Oct. 16, 2018, and Amjevita in the United States on Jan. 31, 2023. Samsung Bioepis’ U.S. license date will not be accelerated upon Amgen’s entry.
Ian Henshaw, Biogen’s global head of biosimilars, said the deal further strengthens the company’s European biosimilars reach.
“Biogen is a leader in the emerging field of biosimilars through Samsung Bioepis, our joint venture with Samsung BioLogics,” Mr. Henshaw said in a press statement. “Biogen already markets two biosimilars in Europe and the planned introduction of Imraldi on Oct. 16 could potentially expand patient choice by offering physicians more options to meet the needs of patients while delivering significant savings to healthcare systems.”
AbbVie focused on the settlement as a global recognition of its leadership role in developing the anti-TNF-alpha antibody.
“The Samsung Bioepis settlement reflects the strength and breadth of AbbVie’s intellectual property,” Laura Schumacher, the company’s general counsel, said in the Abbvie statement. “We continue to believe biosimilars will play an important role in our healthcare system, but we also believe it is important to protect our investment in innovation. This agreement accomplishes both objectives.”
Samsung Bioepis will pay royalties to AbbVie for licensing its adalimumab patents once its biosimilar product is launched. As is the case with the prior Amgen resolution, AbbVie will not make any payments to Samsung Bioepis. “All litigation pending between the parties, as well as all litigation with Samsung Bioepis’ European partner, Biogen, will be dismissed. The precise terms of the agreements are confidential,” the Abbvie statement said.
The settlement brings to a closing a flurry of lawsuits Samsung Bioepis filed against AbbVie in 2017.
A new adalimumab biosimilar will become available in the European Union later this year, but a court settlement will keep Samsung Bioepis’ competitor off U.S. shelves until 2023.
Under the settlement, AbbVie, which manufactures adalimumab (Humira), will grant Bioepis and its partner, Biogen, a nonexclusive license to the intellectual property relating to the antibody. Bioepis’ version, dubbed SB5 (Imraldi), will enter global markets in a staggered fashion, according to an AbbVie press statement. In most countries in the European Union, the license period will begin on Oct. 16, 2018. In the United States, Samsung Bioepis’ license period will begin on June 30, 2023, according to the Abbvie statement.
Biogen and Bioepis hailed the settlement as a victory, but Imraldi won’t be the first Humira biosimilar to break into the U.S. market. Last September, AbbVie settled a similar suit with Amgen, granting patent licenses for the global use and sale of its anti–tumor necrosis factor–alpha antibody, Amgevita/Amjevita. Amgen expects to launch Amgevita in Europe on Oct. 16, 2018, and Amjevita in the United States on Jan. 31, 2023. Samsung Bioepis’ U.S. license date will not be accelerated upon Amgen’s entry.
Ian Henshaw, Biogen’s global head of biosimilars, said the deal further strengthens the company’s European biosimilars reach.
“Biogen is a leader in the emerging field of biosimilars through Samsung Bioepis, our joint venture with Samsung BioLogics,” Mr. Henshaw said in a press statement. “Biogen already markets two biosimilars in Europe and the planned introduction of Imraldi on Oct. 16 could potentially expand patient choice by offering physicians more options to meet the needs of patients while delivering significant savings to healthcare systems.”
AbbVie focused on the settlement as a global recognition of its leadership role in developing the anti-TNF-alpha antibody.
“The Samsung Bioepis settlement reflects the strength and breadth of AbbVie’s intellectual property,” Laura Schumacher, the company’s general counsel, said in the Abbvie statement. “We continue to believe biosimilars will play an important role in our healthcare system, but we also believe it is important to protect our investment in innovation. This agreement accomplishes both objectives.”
Samsung Bioepis will pay royalties to AbbVie for licensing its adalimumab patents once its biosimilar product is launched. As is the case with the prior Amgen resolution, AbbVie will not make any payments to Samsung Bioepis. “All litigation pending between the parties, as well as all litigation with Samsung Bioepis’ European partner, Biogen, will be dismissed. The precise terms of the agreements are confidential,” the Abbvie statement said.
The settlement brings to a closing a flurry of lawsuits Samsung Bioepis filed against AbbVie in 2017.