User login
PTSD Disability Examination Reports: A Comparison of Veterans Health Administration and Contract Examiners
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
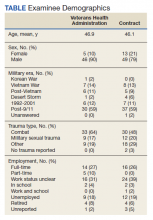
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
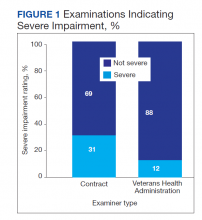
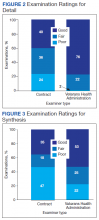
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
Updated June 17, 2022
The US Department of Veterans Affairs (VA) provides health care for > 9 million military veterans, nearly half of all former service members.1 Over the past 15 years, there has been a steady and substantial increase in the frequency of disability awards for veterans with post-9/11 military service. Recent data from the Bureau of Labor Statistics indicate that 41% of veterans who served after 9/11 receive service-connected disability benefits compared with 28% of veterans overall.2 More than 5 million veterans receive VA service-related disability benefits.2,3 More than half of the VA $243 billion budget for fiscal year (FY) 2021 ($135.5 billion) was allocated to the Veterans Benefits Administration (VBA), of which $115.7 billion (85%) was allocated specifically for service-related compensation claims payments.4
The VA predicted that VBA will have completed 1.4 million ratings for disability claims in 2021.5 A substantial percentage of these claims will be for mental disorders, specifically posttraumatic stress disorder (PTSD). VA officials testifying before Congress in 2017 noted that the number of PTSD claims had nearly tripled in the previous 10 years.6 As far back as 2013, McNally and Frueh analyzed “the skyrocketing of disability claims,” particularly for PTSD, among veterans who served in Iraq and Afghanistan.7
This large increase has placed an unprecedented burden on the VBA to expand its capacity to conduct initial PTSD disability evaluations that by regulations are completed by psychologists or psychiatrists. This need has led the VBA to make significant changes in the compensation and pension (C&P) process, including a reduced role for Veterans Health Administration (VHA) examiners and increased reliance on non-VA (contract) examiners through the Contract Medical Disability Examination (MDE) program. In 2019, the MDE budget was $1.23 billion; in 2020, it was increased to $1.79 billion, and for 2021, it was $2.23 billion, reflecting the increasing investment of resources in non-VA examiners, ostensibly to both increase capacity and save costs.5
Anecdotally, concerns have been raised regarding inadequate training of contract examiners as well as inadequate reports by these examiners. A 2018 Government Accountability Office (GAO) report concluded that VA lacked the data to determine whether contract examiners were meeting standards for quality, timeliness, and accuracy.8 The GAO report noted that VA required 92% of contractor reports contained no obvious errors, a relatively low target; however, in the first half of 2017, only 1 contractor group met that target. The report noted further that “VBA does not verify if examiners have completed training nor does it collect information to assess training effectiveness in preparing examiners.”8 A subsequent analysis of contract examinations completed by the VA Office of the Inspector General (OIG) in 2019 concluded that the MDE program was “hampered in their ability to provide oversight because of limitations with VBA’s electronic examination management systems, the lack of reliable data, and inadequate staffing of the program.”9
These reports have focused almost exclusively on simple performance metrics, such as timeliness of examination completion. However, the 2018 GAO report referenced isolated “focused reviews” of complaints about the quality of examinations by contract examiners and gave as an example an isolated “review of one contracted examiner who had high rates of diagnosing severe posttraumatic stress disorder.”8 After review indicated the examiner’s reports were of poor quality, the VBA discontinued the examiner’s contract.
Unfortunately, despite such anecdotal reports and isolated actions, to date there are no published reports examining and comparing the quality of PTSD examination reports completed by VHA and contract examiners or the subsequent disability determinations made by the VBA as a result of these evaluations. In a November 2020 letter to the VA Secretary, 11 US Senators expressed “grave concerns” regarding the VA decision to privatize C&P programs noting, among other concerns, that there were “no clinical quality measurement for, or evaluation of, contractor examinations.”10 The letter cited anecdotal evidence of contract examiners not reviewing veteran’s medical records and diagnosing conditions “without supporting evidence.”10
The purpose of the present evaluation was to provide a systematic comparison of the content and quality of initial PTSD disability examinations conducted by VHA and non-VA contract examiners. In addition, this study compared the disability rating decisions resulting from VHA and contract examinations.
Methods
A random sample of 100 Initial PTSD Disability Benefits Questionnaires (DBQs)—structured forms completed by all examiners—were obtained from a list supplied by the VA Office of Performance Analysis and Integrity. All examinations were from the Veterans Integrated Service Network (VISN) 1, encompassing the New England region and were conducted in 2019 and 2020. Two of the 100 cases were excluded for technical reasons, resulting in 98 examination reports. However, the final pool yielded 62 contract examinations and only 36 VHA examinations. To make the sample sizes more comparable, an additional 15 examinations were randomly selected from the local examination database (also VISN 1) to complement the original examination pool.
Once DBQs were retrieved, all identifying information was deleted, and cases were analyzed using assigned record numbers. All coding was completed by the 2 principal investigators, both VA psychologists with extensive training and experience in C&P evaluation and treatment of veterans with PTSD. Due to inherent structural differences between the forms used for VA and contract examinations, raters could not be masked/blinded to the source of the report.
A number of measures were taken to reduce bias and enhance objectivity of rating. First, objectively coded variables (eg, age and sex of veteran, period of service, trauma type, diagnoses rendered by the examiner, impairment category endorsed, number and type of symptoms) were transcribed directly from the DBQ as recorded by the examiner. Second, to rate report quality, an initial categorical rating scale was developed based on predetermined elements of examination quality that were considered essential. After refinement and preliminary analysis of interrater reliabilities, 3 quality-related indices were identified: (1) level of detail in description of key content areas (history before service, service trauma, after service social and vocational history, mental health history, substance use); (2) synthesis of history and findings in explaining opinion rendered; and (3) clarity of opinion regarding causation required “at least as likely as not” degree of confidence. The first 2 quality ratings were based on a 3-point scale (poor, fair, good), and the third variable was coded as yes or no. (eAppendix available at doi:10.12788/fp.0225). Interrater reliabilities calculated based on a subsample of 18 cases, randomly selected and rated by both raters, yielded Cohen κ in the acceptable range (.61, .72, and .89 for detail, synthesis, and clarity, respectively). Finally, for information regarding VBA decision making, rating decision documents contained in the Veterans Benefit Management System database were reviewed to determine whether the veteran was granted service connection for PTSD or another mental disorder based on the examination report in question and, if so, the disability rating percentage awarded. These were recorded independently after all other coding had been completed.
Results
Comparison of VHA and contract examinations revealed no significant differences between groups on relevant sociodemographic and other measures (Table). Missing data were not obtained from other records or sources, and for this study, reflect only what is recorded in the examination reports except for age, which was calculated using veteran’s date of birth and the date of examination.
To examine differences between VHA and contract examinations, the groups were first compared on a set of predetermined objectively coded variables taken directly from the DBQ. The frequency of PTSD diagnoses by VHA (57%) and contract (71%) examiners was not significantly different nor were rates of non-PTSD diagnoses by VHA (51%) and contract (73%) examiners. There also was no difference in the mean number of PTSD symptoms endorsed across PTSD diagnostic criteria B, C, D, and E (maximum of 20) recorded by VHA (9.4) and contract (10.9) examiners.
Contract examiners recorded a significantly greater mean number of “other symptoms” on a checklist of 31 possible symptoms as compared to VHA examiners: 7.3 vs 5.8, respectively (t[104] = 2.27, P < .05). An initial analysis of overall social/vocational impairment ratings coded by examiners did not reveal significant differences between examiner groups. However, when the 2 most severe impairment categories were combined to create a pooled “severe” category, 31% of contract examiners rated veterans as severely impaired compared with only 12% of VHA examiners (χ2 = 5.79, 1 df, P < .05) (Figure 1).
VHA and contract examinations were compared on 3 measures of report quality. Significant differences were found for both level of detail (χ2 = 16.44, 2 df, P < .01) and synthesis (χ2 = 6.68, 2 df, P < .05). Contract examinations were more likely to be rated as poor and less likely to be rated good, with a similar proportion of fair ratings for the 2 examination types (Figures 2 and 3). There was no significant difference in the proportion of VHA and contract examinations providing clear statement of opinion regarding causation (ie, whether or not the diagnosed condition was service related), with the majority rendering an adequate opinion in both examiner groups (VHA, 78%; contract, 79%).
Qualitative review revealed examples of markedly deficient examinations among contract examinations, including several reports that contained no review of records, no report of relevant background, and no mention or assessment of social and vocational function needed to inform opinions about diagnosis and impairment.
Finally, the VBA database was used to compare the resulting disability award decisions made by VBA based on the examination reports in question. Examination by contractors resulted in significantly higher mean service-connected disability ratings for examinees compared with VHA examiners (46.8 vs 33.5, respectively; t[108] = 2.3, P < .05).
Discussion
The present study provides the first reported systematic comparison of VA disability examinations for PTSD completed by examiners employed by the VHA and those hired as contract examiners through the MDE program. Although the frequency of PTSD diagnoses by contract examiners was higher than that of VHA examiners (71% vs 57%, respectively), the difference was not statistically significant. However, contract examiners recorded significantly more symptoms for examinees and rated them as severely impaired more frequently than did their VHA counterparts. In keeping with rating guidelines used by the VBA, these differences in examination content resulted in higher disability ratings for veterans seen by contract examiners.
Along with these elevated reports in symptom and severity ratings, contract examiners were less likely to provide adequate detail in the narrative sections of their reports and less frequently provided a satisfactory explanation and synthesis of relevant history and findings in support of their conclusions. Although not reflected in the statistical analysis, case-by-case review revealed some startlingly inadequate examination reports by contract examiners, several of which contained no review of records, no report or discussion of relevant background, and no discussion or analysis of social and vocational function to inform and support their opinion about level of impairment. None of the VHA examination reports reviewed lacked information to that degree.
Such deficiencies in detail and synthesis run counter to accepted guidelines for the adequate assessment of psychological injury in general and in VA disability claims specifically.11,12 For example, Watson and colleagues proposed that a minimum of 3 hours was required to conduct an initial PTSD examination, with more complex cases possibly taking longer.11 There is no information available about how long contract examiners take to complete their examinations and how that compares with the time taken by VA examiners. The VBA failure to monitor whether or not examiners follow accepted guidelines for PTSD examination has not previously been evaluated. Historically, a large number of clinicians, researchers, and policy critics have raised concerns about the potential for exaggeration or malingering among VA PTSD disability claimants and have urged the need to adequately assess for unreliable reporting and presentation.13,14 However, the possibility of systematic examiner deficiency and/or bias increasing the frequency of false or inflated claims being approved has received little empirical attention.
Although contract examiners did not diagnose PTSD significantly more frequently than VHA examiners (71% vs 57%, respectively), the overall frequency of PTSD diagnosis across both groups (65%) was substantially higher than previous figures that have, on average, estimated the lifetime prevalence of PTSD in trauma-exposed veterans to be about 31%.15 A re-analysis of the same National Vietnam Veterans Readjustment Survey data, but applying more conservative diagnostic criteria, reduced the lifetime prevalence to just under 19%, with point prevalence estimates even lower.15,16
In a study of concordance rates between service connection for PTSD and both current and lifetime diagnosis by independent, structured assessment, Marx and colleagues found that a “significant minority” of veterans who were already receiving service-connected disability for PTSD did not meet lifetime and/or current diagnostic criteria.17 Although it is possible that the group of veterans who were applying for disability benefits in our study had a higher rate of PTSD, it also is possible if not likely that the PTSD examination process overall yields inflated rates of diagnosis and levels of impairment. This speaks to the concern raised by Marx and colleagues who found that veterans with service connection for PTSD who received related benefits “may not have the disorder.”17
Limitations
A methodological limitation of the present study was that, due to structural differences in the DBQ forms used for VHA and contract examinations, the reports could not be de-identified as to examiner type and thus raters could not be masked/blinded. To mitigate bias, a predetermined, piloted, and refined coding and rating plan for report quality metrics was adhered to strictly, and interrater reliabilities were acceptable. Future study is suggested in which all report content is standardized for coding using the same format, which at present would require a complete rewriting of the entire report; this problem could be resolved by having the VBA adopt a more coherent system in which all reports, regardless of examiner type, use a single, standardized template. Further study using larger data sets and expanding to other VA regions also is needed.
Conclusions
The present study suggests that poor examination and report quality—by contract examiners and to a lesser degree VHA examiners—are not uncommon. The findings confirm and extend previous anecdotal reports of deficiencies in PTSD examinations performed by contract examiners and provide empirical support for concerns raised of global deficiencies in the VBA oversight of the MDE program. Such deficiencies have significant implications for the quality and integrity of the VA disability determination process for veterans claiming PTSD related to military service.
The current findings support and strengthen the call for development and management of a structured and enforced training and quality assurance/improvement program for VA PTSD disability examinations. Such training and oversight will be critical to improve the quality and integrity of these examinations, reduce error and waste in VBA’s Compensation and Pension process, and in doing so optimize VA financial resources to best serve veterans’ benefits and health care needs.
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
1. US Department of Veterans Affairs, Veterans Health Adminstration. About VHA. Updated April 23, 2021. Accessed January 6, 2022. www.va.gov/health/aboutvha.asp
2. US Department of Labor, Bureau of Labor Statistics. News release. Employment situation of veterans—2020. Published March 18, 2020. Accessed January 6, 2022. https://www.bls.gov/news.release/pdf/vet.pdf
3. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Department of Veterans Affairs statistics at a glance. Updated December 31, 2020. Accessed January 6, 2022. https://www.va.gov/vetdata/docs/Quickfacts/Stats_at_a_glance_12_31_20.PDF
4. US Department of Veterans Affairs. FY 2021 Budget submission: budget in brief. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
5. US Department of Veterans Affairs. FY 2021 budget submission: benefits and burial programs and Departmental Administration volume 3 of 4:178. Published February 2020. Accessed January 6, 2022. https://www.va.gov/budget/docs/summary/archive/FY-2021-VA-BudgetSubmission.zip
6. Statement of Ronald Burke, assistant deputy under secretary, office of field operations Veterans Benefits Administration before the Subcommittee on Disability And Memorial Affairs of the House Committee on Veterans’ Affairs. Published July 25, 2017. Accessed January 6, 2022. https://www.congress.gov/115/meeting/house/106322/witnesses/HHRG-115-VR09-Wstate-BurkeR-20170725.pdf
7. McNally RJ, Frueh BC. Why are Iraq and Afghanistan War veterans seeking PTSD disability compensation at unprecedented rates? J Anxiety Disord. 2013;27(5):520-526. doi:10.1016/j.janxdis.2013.07.002
8. US Government Accountability Office. VA disability exams: improved performance analysis and training oversight needed for contracted exams. GAO-19-13. Published October 2018. Accessed January 6, 2022. https://www.gao.gov/assets/gao-19-13.pdf
9. US Department of Veterans Affairs, Office of Inspector General. Inadequate oversight of contracted disability exam cancellations. Report #18-04266-115. Published June 10, 2019. Accessed January 6, 2022. https://www.va.gov/oig/pubs/VAOIG-18-04266-115.pdf
10. Letter to VA Secretary Wilkie. Published November 11, 2020. Accessed January 6, 2022. https://www.veterans.senate.gov/download/candp-exam-va-letter
11. Watson PW, McFall M, McBrine C, Schnurr PP, Friedman MJ, Keane TM, Hamblen JL (2005). Best practice manual for posttraumatic stress disorder (PTSD) compensation and pension examinations. Portland, OR: Northwest Network Mental Illness Research, Education, and Clinical Center, VA Puget Sound Healthcare System.
12. Worthen MD, Moering RG. A practical guide to conducting VA compensation and pension exams for PTSD and other mental disorders. Psychol Inj and Law. 2011;4:187-216. doi:10.1007/s12207-011-9115-2
13. DeViva JC, Bloem WD. Symptom exaggeration and compensation seeking among combat veterans with posttraumatic stress disorder. J Trauma Stress. 2003;16(5):503-507. doi:10.1023/A:1025766713188
14. Ray CL. Feigning screeners in VA PTSD compensation and pension examinations. Psychol Inj and Law. 2014;7:370-387. doi:10.1007/s12207-014-9210-2
15. Kulka RA, Schlenger WE, Fairbank JA, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. Brunner Mazel Publishers; 1990.
16. Dohrenwend BP, Turner JB, Turse NA, Adams BG, Koenen KC, Marshall R. The psychological risks of Vietnam for U.S. veterans: a revisit with new data and methods. Science. 2006;313(5789):979-982. doi:10.1126/science.1128944
17. Marx BP, Bovin MJ, Szafranski DD, et al. Validity of posttraumatic stress disorder service connection status in Veterans Affairs electronic records of Iraq and Afghanistan Veterans. J Clin Psychiatry. 2016;77(4):517-522. doi:10.4088/JCP.14m09666
Chronic stress accelerates aging: Epigenetic evidence
The increase in cardiovascular disease caused by chronic stress is related to biologic mechanisms (metabolic, hormonal, inflammatory) and to behavioral mechanisms (lifestyle). There is a popular saying that “stress speeds up aging,” which makes sense if we consider the age-old idea that “our age corresponds to that of our arteries.”
The study of the mechanisms of psychosocial risk factors is of major relevance to the creation of the individual and communal preventive strategies that ensure longevity and maintain quality of life.
The following hypotheses were proposed by a group of researchers from Yale University, in New Haven, Conn., in a recent study:
1. Stress is positively associated with accelerated biologic aging, and this relationship will be mediated by stress-related physiologic changes, such as insulin and hypothalamic-pituitary-adrenal (HPA) signaling.
2. Strong factors associated with psychologic resilience will be protective against the negative consequences of stress on aging. (These relationships are predictive, not causative, as this study is cross-sectional.)
The study
In their study, the team assessed 444 adults with no chronic medical conditions or psychiatric disorders who were 18-50 years of age and living in the greater New Haven area. Levels of obesity and alcohol consumption in the study cohort were generally in line with those in a community population, so alcohol use and body mass index were used as covariates to account for their impact on the results.
The team also used the latest “epigenetic clock,” known as GrimAge. In recent years, several methods of determining biologic age have been developed that trace chemical changes in the DNA that are natural to the aging process but occur at different moments in different people. The epigenetic clocks have proved to be better predictors of longevity and health than chronologic age, and GrimAge predicts mortality better than other epigenetic clocks.
Results
1. Cumulative stress was associated with the acceleration of GrimAge and stress-related physiologic measures of adrenal sensitivity (cortisol/ACTH ratio) and insulin resistance (HOMA). After the researchers controlled for demographic and behavioral factors, HOMA was correlated with GrimAge acceleration.
2. Psychologic resilience factors moderated the association between stress and aging, such that with worse regulation of emotions, there was greater stress-related age acceleration, and with stronger regulation of emotions, any significant effect of stress on GrimAge was prevented. Self-control moderated the relationship between stress and insulin resistance, with high self-control blunting this relationship.
3. In the final model, in those with poor emotion regulation, cumulative stress continued to predict additional GrimAge acceleration, even when demographic, physiologic, and behavioral covariates were accounted for.
Implications
These results elegantly demonstrate that cumulative stress is associated with epigenetic aging in a healthy population, and these associations are modified by biobehavioral resilience factors.
Even after adjustment for demographic and behavioral factors – such as smoking, body mass index, race, and income – people with high chronic stress scores showed markers of accelerated aging and physiologic changes, such as increased insulin resistance.
However, individuals with high scores on two psychologic resilience measures – emotion regulation and self-control – were more resilient to the effects of stress on aging and insulin resistance.
These results support the popular notion that
In other words, the greater the psychologic resilience, the more likely the individual is to live a long and healthy life. “We like to feel as if we have some sovereignty over our destiny and, therefore, it is worth emphasizing to people (and healthcare providers) that it is important to invest in mental health,” said one of the study researchers.
With all the stress we face these days, it is essential to remember that there is no health without mental health. Above all, if we can achieve greater psychologic resilience, we will have a better chance of delaying aging.
A version of this article first appeared on Medscape.com.
The increase in cardiovascular disease caused by chronic stress is related to biologic mechanisms (metabolic, hormonal, inflammatory) and to behavioral mechanisms (lifestyle). There is a popular saying that “stress speeds up aging,” which makes sense if we consider the age-old idea that “our age corresponds to that of our arteries.”
The study of the mechanisms of psychosocial risk factors is of major relevance to the creation of the individual and communal preventive strategies that ensure longevity and maintain quality of life.
The following hypotheses were proposed by a group of researchers from Yale University, in New Haven, Conn., in a recent study:
1. Stress is positively associated with accelerated biologic aging, and this relationship will be mediated by stress-related physiologic changes, such as insulin and hypothalamic-pituitary-adrenal (HPA) signaling.
2. Strong factors associated with psychologic resilience will be protective against the negative consequences of stress on aging. (These relationships are predictive, not causative, as this study is cross-sectional.)
The study
In their study, the team assessed 444 adults with no chronic medical conditions or psychiatric disorders who were 18-50 years of age and living in the greater New Haven area. Levels of obesity and alcohol consumption in the study cohort were generally in line with those in a community population, so alcohol use and body mass index were used as covariates to account for their impact on the results.
The team also used the latest “epigenetic clock,” known as GrimAge. In recent years, several methods of determining biologic age have been developed that trace chemical changes in the DNA that are natural to the aging process but occur at different moments in different people. The epigenetic clocks have proved to be better predictors of longevity and health than chronologic age, and GrimAge predicts mortality better than other epigenetic clocks.
Results
1. Cumulative stress was associated with the acceleration of GrimAge and stress-related physiologic measures of adrenal sensitivity (cortisol/ACTH ratio) and insulin resistance (HOMA). After the researchers controlled for demographic and behavioral factors, HOMA was correlated with GrimAge acceleration.
2. Psychologic resilience factors moderated the association between stress and aging, such that with worse regulation of emotions, there was greater stress-related age acceleration, and with stronger regulation of emotions, any significant effect of stress on GrimAge was prevented. Self-control moderated the relationship between stress and insulin resistance, with high self-control blunting this relationship.
3. In the final model, in those with poor emotion regulation, cumulative stress continued to predict additional GrimAge acceleration, even when demographic, physiologic, and behavioral covariates were accounted for.
Implications
These results elegantly demonstrate that cumulative stress is associated with epigenetic aging in a healthy population, and these associations are modified by biobehavioral resilience factors.
Even after adjustment for demographic and behavioral factors – such as smoking, body mass index, race, and income – people with high chronic stress scores showed markers of accelerated aging and physiologic changes, such as increased insulin resistance.
However, individuals with high scores on two psychologic resilience measures – emotion regulation and self-control – were more resilient to the effects of stress on aging and insulin resistance.
These results support the popular notion that
In other words, the greater the psychologic resilience, the more likely the individual is to live a long and healthy life. “We like to feel as if we have some sovereignty over our destiny and, therefore, it is worth emphasizing to people (and healthcare providers) that it is important to invest in mental health,” said one of the study researchers.
With all the stress we face these days, it is essential to remember that there is no health without mental health. Above all, if we can achieve greater psychologic resilience, we will have a better chance of delaying aging.
A version of this article first appeared on Medscape.com.
The increase in cardiovascular disease caused by chronic stress is related to biologic mechanisms (metabolic, hormonal, inflammatory) and to behavioral mechanisms (lifestyle). There is a popular saying that “stress speeds up aging,” which makes sense if we consider the age-old idea that “our age corresponds to that of our arteries.”
The study of the mechanisms of psychosocial risk factors is of major relevance to the creation of the individual and communal preventive strategies that ensure longevity and maintain quality of life.
The following hypotheses were proposed by a group of researchers from Yale University, in New Haven, Conn., in a recent study:
1. Stress is positively associated with accelerated biologic aging, and this relationship will be mediated by stress-related physiologic changes, such as insulin and hypothalamic-pituitary-adrenal (HPA) signaling.
2. Strong factors associated with psychologic resilience will be protective against the negative consequences of stress on aging. (These relationships are predictive, not causative, as this study is cross-sectional.)
The study
In their study, the team assessed 444 adults with no chronic medical conditions or psychiatric disorders who were 18-50 years of age and living in the greater New Haven area. Levels of obesity and alcohol consumption in the study cohort were generally in line with those in a community population, so alcohol use and body mass index were used as covariates to account for their impact on the results.
The team also used the latest “epigenetic clock,” known as GrimAge. In recent years, several methods of determining biologic age have been developed that trace chemical changes in the DNA that are natural to the aging process but occur at different moments in different people. The epigenetic clocks have proved to be better predictors of longevity and health than chronologic age, and GrimAge predicts mortality better than other epigenetic clocks.
Results
1. Cumulative stress was associated with the acceleration of GrimAge and stress-related physiologic measures of adrenal sensitivity (cortisol/ACTH ratio) and insulin resistance (HOMA). After the researchers controlled for demographic and behavioral factors, HOMA was correlated with GrimAge acceleration.
2. Psychologic resilience factors moderated the association between stress and aging, such that with worse regulation of emotions, there was greater stress-related age acceleration, and with stronger regulation of emotions, any significant effect of stress on GrimAge was prevented. Self-control moderated the relationship between stress and insulin resistance, with high self-control blunting this relationship.
3. In the final model, in those with poor emotion regulation, cumulative stress continued to predict additional GrimAge acceleration, even when demographic, physiologic, and behavioral covariates were accounted for.
Implications
These results elegantly demonstrate that cumulative stress is associated with epigenetic aging in a healthy population, and these associations are modified by biobehavioral resilience factors.
Even after adjustment for demographic and behavioral factors – such as smoking, body mass index, race, and income – people with high chronic stress scores showed markers of accelerated aging and physiologic changes, such as increased insulin resistance.
However, individuals with high scores on two psychologic resilience measures – emotion regulation and self-control – were more resilient to the effects of stress on aging and insulin resistance.
These results support the popular notion that
In other words, the greater the psychologic resilience, the more likely the individual is to live a long and healthy life. “We like to feel as if we have some sovereignty over our destiny and, therefore, it is worth emphasizing to people (and healthcare providers) that it is important to invest in mental health,” said one of the study researchers.
With all the stress we face these days, it is essential to remember that there is no health without mental health. Above all, if we can achieve greater psychologic resilience, we will have a better chance of delaying aging.
A version of this article first appeared on Medscape.com.
‘Deaths of despair’ rising, but only in the U.S.
In the United States,
This is not the case in 16 other industrialized nations, however, including Canada, Australia, and Japan, where mortality rates are actually decreasing.
One likely reason is that other countries take better care of their citizens from cradle to grave, authors Peter Sterling, PhD, and Michael Platt, PhD, of the University of Pennsylvania, Philadelphia, wrote in a special communication in JAMA Psychiatry published online Feb. 2.
In the United States, individuals and families often struggle in isolation to navigate the life cycle, whereas other countries offer communal assistance to every life stage, and this support protects individuals and families in the long term, they noted.
The United States could solve this “health crisis” by adopting the best practices of these other nations, they wrote.
U.S. is an outlier
From an anthropological perspective, Dr. Sterling and Dr. Platt point out that “hunter-gatherers” prioritized food, comfort, and companionship. When one of these needs is unexpectedly met, the surprise triggers a pulse of the feel-good hormone dopamine.
However, much of modern life offers few opportunities for surprise and dopamine pulses.
“It is the difference between a day’s hard walk to finally encounter and kill a wild pig to feed the family and community versus a quick trip to aisle 7 to select a pork roast in plastic wrap,” Dr. Sterling and Dr. Platt noted.
The hunter-gatherers were far more physically active, and cardiovascular disease, diabetes, obesity, and hypertension were virtually unknown.
The small-scale societies of hunters and gatherers depended on strong family bonds and cooperation with community members.
Modern life is more isolating, often with hours spent alone in front of a computer screen.
Yet the lack of natural dopamine producers in modern society and the increased social isolation is not unique to the United States but holds across the board for industrialized nations.
So why has the United States suffered more deaths of despair?
Dr. Sterling and Dr. Platt assert that it comes down to public support other countries provide their citizens across the life span, from prenatal care and quality preschool and elementary school to affordable (or free) education beyond high school.
This support did not require “bloody revolutions, just simple agreements to prepay basic human needs from public funds collected as taxes,” Dr. Sterling and Dr. Platt noted.
By adopting some of the best practices pioneered by other wealthy nations, the United States could reduce despair and restore to many the will to live, they added.
However, they caution against the “medicalization” of every identified cause of rising death rates.
“Every symptom of despair has been defined as a disorder or dysregulation within the individual. This incorrectly frames the problem, forcing individuals to grapple on their own,” they wrote.
“It also emphasizes treatment by pharmacology, providing innumerable drugs for anxiety, depression, anger, psychosis, and obesity, plus new drugs to treat addictions to the old drugs. We cannot defeat despair solely with pills – to the contrary, pills will only deepen it,” they added.
Dr. Platt reported receiving grant support from the National Institutes of Health, the National Science Foundation, and the Charles E. Kaufman Foundation. He is cofounder of Cogwear and a scientific adviser to Neuroflow, Amplio, Blue Horizon International, and Progenity. Dr. Sterling has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the United States,
This is not the case in 16 other industrialized nations, however, including Canada, Australia, and Japan, where mortality rates are actually decreasing.
One likely reason is that other countries take better care of their citizens from cradle to grave, authors Peter Sterling, PhD, and Michael Platt, PhD, of the University of Pennsylvania, Philadelphia, wrote in a special communication in JAMA Psychiatry published online Feb. 2.
In the United States, individuals and families often struggle in isolation to navigate the life cycle, whereas other countries offer communal assistance to every life stage, and this support protects individuals and families in the long term, they noted.
The United States could solve this “health crisis” by adopting the best practices of these other nations, they wrote.
U.S. is an outlier
From an anthropological perspective, Dr. Sterling and Dr. Platt point out that “hunter-gatherers” prioritized food, comfort, and companionship. When one of these needs is unexpectedly met, the surprise triggers a pulse of the feel-good hormone dopamine.
However, much of modern life offers few opportunities for surprise and dopamine pulses.
“It is the difference between a day’s hard walk to finally encounter and kill a wild pig to feed the family and community versus a quick trip to aisle 7 to select a pork roast in plastic wrap,” Dr. Sterling and Dr. Platt noted.
The hunter-gatherers were far more physically active, and cardiovascular disease, diabetes, obesity, and hypertension were virtually unknown.
The small-scale societies of hunters and gatherers depended on strong family bonds and cooperation with community members.
Modern life is more isolating, often with hours spent alone in front of a computer screen.
Yet the lack of natural dopamine producers in modern society and the increased social isolation is not unique to the United States but holds across the board for industrialized nations.
So why has the United States suffered more deaths of despair?
Dr. Sterling and Dr. Platt assert that it comes down to public support other countries provide their citizens across the life span, from prenatal care and quality preschool and elementary school to affordable (or free) education beyond high school.
This support did not require “bloody revolutions, just simple agreements to prepay basic human needs from public funds collected as taxes,” Dr. Sterling and Dr. Platt noted.
By adopting some of the best practices pioneered by other wealthy nations, the United States could reduce despair and restore to many the will to live, they added.
However, they caution against the “medicalization” of every identified cause of rising death rates.
“Every symptom of despair has been defined as a disorder or dysregulation within the individual. This incorrectly frames the problem, forcing individuals to grapple on their own,” they wrote.
“It also emphasizes treatment by pharmacology, providing innumerable drugs for anxiety, depression, anger, psychosis, and obesity, plus new drugs to treat addictions to the old drugs. We cannot defeat despair solely with pills – to the contrary, pills will only deepen it,” they added.
Dr. Platt reported receiving grant support from the National Institutes of Health, the National Science Foundation, and the Charles E. Kaufman Foundation. He is cofounder of Cogwear and a scientific adviser to Neuroflow, Amplio, Blue Horizon International, and Progenity. Dr. Sterling has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the United States,
This is not the case in 16 other industrialized nations, however, including Canada, Australia, and Japan, where mortality rates are actually decreasing.
One likely reason is that other countries take better care of their citizens from cradle to grave, authors Peter Sterling, PhD, and Michael Platt, PhD, of the University of Pennsylvania, Philadelphia, wrote in a special communication in JAMA Psychiatry published online Feb. 2.
In the United States, individuals and families often struggle in isolation to navigate the life cycle, whereas other countries offer communal assistance to every life stage, and this support protects individuals and families in the long term, they noted.
The United States could solve this “health crisis” by adopting the best practices of these other nations, they wrote.
U.S. is an outlier
From an anthropological perspective, Dr. Sterling and Dr. Platt point out that “hunter-gatherers” prioritized food, comfort, and companionship. When one of these needs is unexpectedly met, the surprise triggers a pulse of the feel-good hormone dopamine.
However, much of modern life offers few opportunities for surprise and dopamine pulses.
“It is the difference between a day’s hard walk to finally encounter and kill a wild pig to feed the family and community versus a quick trip to aisle 7 to select a pork roast in plastic wrap,” Dr. Sterling and Dr. Platt noted.
The hunter-gatherers were far more physically active, and cardiovascular disease, diabetes, obesity, and hypertension were virtually unknown.
The small-scale societies of hunters and gatherers depended on strong family bonds and cooperation with community members.
Modern life is more isolating, often with hours spent alone in front of a computer screen.
Yet the lack of natural dopamine producers in modern society and the increased social isolation is not unique to the United States but holds across the board for industrialized nations.
So why has the United States suffered more deaths of despair?
Dr. Sterling and Dr. Platt assert that it comes down to public support other countries provide their citizens across the life span, from prenatal care and quality preschool and elementary school to affordable (or free) education beyond high school.
This support did not require “bloody revolutions, just simple agreements to prepay basic human needs from public funds collected as taxes,” Dr. Sterling and Dr. Platt noted.
By adopting some of the best practices pioneered by other wealthy nations, the United States could reduce despair and restore to many the will to live, they added.
However, they caution against the “medicalization” of every identified cause of rising death rates.
“Every symptom of despair has been defined as a disorder or dysregulation within the individual. This incorrectly frames the problem, forcing individuals to grapple on their own,” they wrote.
“It also emphasizes treatment by pharmacology, providing innumerable drugs for anxiety, depression, anger, psychosis, and obesity, plus new drugs to treat addictions to the old drugs. We cannot defeat despair solely with pills – to the contrary, pills will only deepen it,” they added.
Dr. Platt reported receiving grant support from the National Institutes of Health, the National Science Foundation, and the Charles E. Kaufman Foundation. He is cofounder of Cogwear and a scientific adviser to Neuroflow, Amplio, Blue Horizon International, and Progenity. Dr. Sterling has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Case report: Male with acute new-onset suicidal ideation tied to SARS-CoV-2
An otherwise healthy 55-year-old male, with no previous psychiatric or medical history, sought care with a family medicine physician for the first time in decades.
Medical symptoms began Oct. 9, 2021, with “some leg weakness and mild sniffles.” Since he was going to be at a public event, he decided to take a PCR test for the SARS-CoV-2 virus on Oct. 13. The patient tested positive.
His symptoms continued to worsen, and he experienced severe body fatigue, sleep disturbance, and lethargy. “A few days after my positive test, the cognitive and physical symptoms dramatically ramped up,” the patient recalled.
Because of those worsening symptoms, on Oct. 20, the patient obtained a new patient appointment with a family medicine physician. After a telemedicine evaluation, the family medicine physician began a multifaceted early outpatient COVID-19 treatment protocol,1 as I (C.M.W.) and colleagues wrote about late last year. However, this treatment began late in the course because of the patient’s initial resistance to seek care.
This early outpatient treatment protocol for COVID-19 included vitamin D3 125 mcg (5,000 ICU), N-acetylcysteine (NAC) 600 mg every day x 30 days; acetylsalicylic acid 325 mg every day x 30 days; azithromycin 250 mg b.i.d. before every meal x 10 days; hydroxychloroquine sulfate 200 mg b.i.d. x 10 days; ivermectin 3 mg, 5 pills daily x 10 days; zinc sulfate 220 mg (50 mg elemental) every day x 30 days; and a prednisone taper (30 mg daily x 3 days, tapering down 5 mg every 3 days). Hydroxyzine 50 mg at bedtime as needed was added for sleep. The patient did not comment to the family physician on any of the psychological or psychiatric symptoms and responded appropriately to questions during the Oct. 20 initial evaluation.
However, he later described that around the time the PCR was positive, For example, he was watching a simple YouTube video for work and “everything was confusing me ... it rattled me, and I couldn’t understand it.” He described his COVID-19 mind as: “The words in my head would come out in a jumbled order, like the message from the words in my brain to my mouth would get crossed. I had trouble spelling and texting. Total cognitive breakdown. I couldn’t do simple mathematics.”
Despite his physical exhaustion, he endured a 3-day period of sleep deprivation. During this time, he recalled looking up at the roof and thinking, “I need to jump off the roof” or thinking, “I might want to throw myself under a bus.” He did not initially reveal his suicidal thoughts to his family medicine physician. After beginning COVID-19 treatment, the patient had two nights of sleep and felt notably improved, and his physical symptoms began to remit. However, the sleeplessness quickly returned “with a vengeance” along with “silly suicidal thoughts.” The thoughts took on a more obsessional quality. For example, he repeatedly thought of jumping out of his second-story bedroom to the living room below and was preoccupied by continually looking at people’s roofs and thinking about jumping. Those thoughts intensified and culminated in his “going missing,” leading his wife to call the police. It was discovered that he had driven to a local bridge and was contemplating jumping off.
After that “going missing” incident, the patient and his wife reached out to their family medicine physician. He reevaluated the patient and, given the new information about the psychiatric symptoms, strongly recommended stat crisis and psychiatric consultation. After discussing the case on the same day, both the family medicine physician and the psychiatrist recommended stat hospital emergency department (ED) assessment on Oct. 29. In the ED, a head CT without contrast at the recommendation of both psychiatrist and family physician, routine electrolytes, CBC with differential, and EKG all were within normal limits. The ED initially discharged him home after crisis evaluation, deciding he was not an imminent risk to himself or others.
The next day, the psychiatrist spoke on the phone with the patient, family medicine physician, and the patient’s wife to arrange an initial assessment. At that time, it remained unclear to all whether the obsessional thoughts had resolved to such a degree that the patient could resist acting upon them. Further, the patient’s sleep architecture had not returned to normal. All agreed another emergency ED assessment was indicated. Ultimately, after voluntary re-evaluation and a difficult hold in the crisis unit, the patient was admitted for psychiatric hospitalization on Oct. 29 and discharged on Nov. 4.
In the psychiatric hospital, venlafaxine XR was started and titrated to 75 mg. The patient was discovered to be hypertensive, and hydrochlorothiazide was started. The discharge diagnosis was major depressive disorder, single episode, severe, without psychotic features.
Posthospitalization course
He was seen for his initial psychiatric outpatient assessment postpsychiatric hospitalization on Nov. 9, as he had not yet been formally evaluated by the psychiatrist because of the emergency situation.
Gabapentin 300 mg by mouth at bedtime was started, and his sleep architecture was restored. The initial plan to titrate venlafaxine XR into dual selective norepinephrine reuptake inhibitor dose range was terminated, and his psychiatrist considered tapering and discontinuing the venlafaxine XR. A clinical examination, additional history, and collateral data no longer necessarily pointed to an active major depressive disorder or even unspecified depressive disorder, though to be sure, the patient was taking 75 mg of venlafaxine XR. While there were seasonal stressors, historically, nothing had risen to the level of MDD.
The obsessions driving his thoughts to jump off buildings and bridges had completely remitted. His cognitive ability returned to baseline with an ability to focus and perform the complicated tasks of his high-intensity work by the Dec. 8 psychiatric examination, where he was accompanied by his wife. He described feeling like, “I snapped back to like I was before this crazy stuff happened.” His mood was reported as, “Very good; like my old self” and this was confirmed by his wife. His affect was calmer and less tense. He was now using gabapentin sparingly for sleep. We continued to entertain discontinuing the venlafaxine XR, considering this recent severe episode likely driven by the COVID-19 virus. The decision was made to continue venlafaxine XR through the winter rather than discontinuing, remaining on the conservative side of treatment. The patient’s diagnosis was changed from “MDD, single episode,” to “mood disorder due to known physiologic condition (COVID-19) (F06.31) with depressive features; resolving.” At the patient’s follow-up examination on Jan. 5, 2022, he was continuing to do well, stating, “The whole series of crazy events happened to someone else.” The hydrochlorothiazide had been discontinued, and the patient’s blood pressure and pulse were normal at 119/81 and 69, respectively. He had made strategic changes at work to lessen stressors during the typically difficult months.
Discussion
Literature has discussed neuropsychiatric sequelae of COVID-19.2 The cited example questions whether psychiatric symptoms are tied directly to the viral infection or to the “host’s immune response.” We believe our case represents a direct neurocognitive/neuropsychiatric insult due to the COVID-19 infection.
This case presents a 55-year-old male with no previous psychiatric or medical history with new onset significant and debilitating cognitive impairment and obsessive thoughts of throwing himself from his bedroom balcony ending up at a bridge struggling with an irrational thought of jumping; ultimately requiring psychiatric hospitalization for acute suicidal thoughts. The patient’s psychiatric symptoms arose prior to any and all medication treatment. The obsessive thoughts correlated both with the onset of SARS-CoV-2 infection and a period of sleep deprivation subsequent to the infection. A course of steroid treatment and taper were started after the onset of neurocognitive-psychiatric symptoms, though there is close timing. We submit that the patient experienced, as part of the initial neurocognitive psychiatric initiating cascade, a COVID-19–induced sleep deprivation that was not etiologic but part of the process; since, even when sleep returned to normal, it was still several weeks before full cognitive function returned to baseline.
An argument could be made for possible MDD or unspecified depressive disorder, as historically there had been work-related stressors for the patient at this time of year because of the chronological nature of his work; though previously nothing presented with obsessional suicidal thinking and nothing with any cognitive impairment – let alone to this incapacitating degree.
The patient describes his seasonal work much like an accountant’s work at the beginning of each year. In the patient’s case, the months of September and October are historically “nonstop, working days,” which then slow down in the winter months for a period of recuperation. In gathering his past history of symptoms, he denied neurovegetative symptoms to meet full diagnostic criteria for MDD or unspecified depressive disorder, absent this episode in the presence of SARS-CoV-2 infection.
We could also consider a contributory negative “organic push” by the viral load and prednisone helping to express an underlying unspecified depression or MDD, but for the profound and unusual presentation. There was little prodrome of depressive symptoms (again, he reported his “typical” extraordinary work burden for this time of year, which is common in his industry).
In this patient, the symptoms have remitted completely. However, the patient is currently taking venlafaxine XR 75 mg. We have considered tapering and discontinuing the venlafaxine – since it is not entirely clear that he needs to be on this medication – so this question remains an open one. We did decide, however, to continue the venlafaxine until after the winter months and to reassess at that time.
Conclusion
The patient presented with new onset psychological and psychiatric symptoms in addition to physiologic symptoms; the former symptoms were not revealed prior to initial family medicine evaluation. As the symptoms worsened, he and his wife sought additional consultation with family physician, psychiatrists, and ED. Steroid treatment may have played a part in exacerbation of symptoms, but the neuropsychiatric cognitive symptoms were present prior to initiation of all pharmacologic and medical treatment. The successful outcome of this case was based upon quick action and collaboration between the family medicine physician, the psychiatrist, and the ED physician. The value of communication, assessment, and action via phone call and text cannot be overstated. Future considerations include further large-scale evaluation of multifaceted early treatment of patients with COVID-19 within the first 72 hours of symptoms to prevent not only hospitalization, morbidity, and mortality, but newly recognized psychological and psychiatric syndromes.3,4
Lastly, fluvoxamine might have been a better choice for adjunctive early treatment of COVID-19.5 As a matter of distinction, if a lingering mood disorder or obsessive-compulsive disorder remain a result of SARS-CoV-2 or if one were to start an antidepressant during the course of illness, it would be reasonable to consider fluvoxamine as a potential first-line agent.
Dr. Kohanski is a fellowship trained forensic psychiatrist and a diplomate of the American Board of Psychiatry & Neurology. She maintains a private practice in Somerset, N.J., and is a frequent media commentator and medical podcaster. Dr. Kohanski has no conflicts of interest. Dr. Wax is a residency-trained osteopathic family medicine physician in independent private practice in Mullica Hill, N.J. He has authored multiple papers over 2 decades on topics such as SARS-CoV-2 and COVID-19 early treatment. He has been a speaker and media host over 2 decades and served on the National Physicians Council on Healthcare Policy’s congressional subcommittee. Dr. Wax has no conflicts of interest.
References
1. Rev Cardiovasc Med. 2020 Dec 30;21(4):517-30.
2. Brain Behav Immun. 2020 Jul;87:34-9.
3. Trav Med Infect Dis. 2020 May-Jun 35;10738.
4. Kirsch S. “Early treatment for COVID is key to better outcomes.” Times of India. 2021 May 21.
5. Lancet. 2022 Jan 1;10(1):E42-E51.
An otherwise healthy 55-year-old male, with no previous psychiatric or medical history, sought care with a family medicine physician for the first time in decades.
Medical symptoms began Oct. 9, 2021, with “some leg weakness and mild sniffles.” Since he was going to be at a public event, he decided to take a PCR test for the SARS-CoV-2 virus on Oct. 13. The patient tested positive.
His symptoms continued to worsen, and he experienced severe body fatigue, sleep disturbance, and lethargy. “A few days after my positive test, the cognitive and physical symptoms dramatically ramped up,” the patient recalled.
Because of those worsening symptoms, on Oct. 20, the patient obtained a new patient appointment with a family medicine physician. After a telemedicine evaluation, the family medicine physician began a multifaceted early outpatient COVID-19 treatment protocol,1 as I (C.M.W.) and colleagues wrote about late last year. However, this treatment began late in the course because of the patient’s initial resistance to seek care.
This early outpatient treatment protocol for COVID-19 included vitamin D3 125 mcg (5,000 ICU), N-acetylcysteine (NAC) 600 mg every day x 30 days; acetylsalicylic acid 325 mg every day x 30 days; azithromycin 250 mg b.i.d. before every meal x 10 days; hydroxychloroquine sulfate 200 mg b.i.d. x 10 days; ivermectin 3 mg, 5 pills daily x 10 days; zinc sulfate 220 mg (50 mg elemental) every day x 30 days; and a prednisone taper (30 mg daily x 3 days, tapering down 5 mg every 3 days). Hydroxyzine 50 mg at bedtime as needed was added for sleep. The patient did not comment to the family physician on any of the psychological or psychiatric symptoms and responded appropriately to questions during the Oct. 20 initial evaluation.
However, he later described that around the time the PCR was positive, For example, he was watching a simple YouTube video for work and “everything was confusing me ... it rattled me, and I couldn’t understand it.” He described his COVID-19 mind as: “The words in my head would come out in a jumbled order, like the message from the words in my brain to my mouth would get crossed. I had trouble spelling and texting. Total cognitive breakdown. I couldn’t do simple mathematics.”
Despite his physical exhaustion, he endured a 3-day period of sleep deprivation. During this time, he recalled looking up at the roof and thinking, “I need to jump off the roof” or thinking, “I might want to throw myself under a bus.” He did not initially reveal his suicidal thoughts to his family medicine physician. After beginning COVID-19 treatment, the patient had two nights of sleep and felt notably improved, and his physical symptoms began to remit. However, the sleeplessness quickly returned “with a vengeance” along with “silly suicidal thoughts.” The thoughts took on a more obsessional quality. For example, he repeatedly thought of jumping out of his second-story bedroom to the living room below and was preoccupied by continually looking at people’s roofs and thinking about jumping. Those thoughts intensified and culminated in his “going missing,” leading his wife to call the police. It was discovered that he had driven to a local bridge and was contemplating jumping off.
After that “going missing” incident, the patient and his wife reached out to their family medicine physician. He reevaluated the patient and, given the new information about the psychiatric symptoms, strongly recommended stat crisis and psychiatric consultation. After discussing the case on the same day, both the family medicine physician and the psychiatrist recommended stat hospital emergency department (ED) assessment on Oct. 29. In the ED, a head CT without contrast at the recommendation of both psychiatrist and family physician, routine electrolytes, CBC with differential, and EKG all were within normal limits. The ED initially discharged him home after crisis evaluation, deciding he was not an imminent risk to himself or others.
The next day, the psychiatrist spoke on the phone with the patient, family medicine physician, and the patient’s wife to arrange an initial assessment. At that time, it remained unclear to all whether the obsessional thoughts had resolved to such a degree that the patient could resist acting upon them. Further, the patient’s sleep architecture had not returned to normal. All agreed another emergency ED assessment was indicated. Ultimately, after voluntary re-evaluation and a difficult hold in the crisis unit, the patient was admitted for psychiatric hospitalization on Oct. 29 and discharged on Nov. 4.
In the psychiatric hospital, venlafaxine XR was started and titrated to 75 mg. The patient was discovered to be hypertensive, and hydrochlorothiazide was started. The discharge diagnosis was major depressive disorder, single episode, severe, without psychotic features.
Posthospitalization course
He was seen for his initial psychiatric outpatient assessment postpsychiatric hospitalization on Nov. 9, as he had not yet been formally evaluated by the psychiatrist because of the emergency situation.
Gabapentin 300 mg by mouth at bedtime was started, and his sleep architecture was restored. The initial plan to titrate venlafaxine XR into dual selective norepinephrine reuptake inhibitor dose range was terminated, and his psychiatrist considered tapering and discontinuing the venlafaxine XR. A clinical examination, additional history, and collateral data no longer necessarily pointed to an active major depressive disorder or even unspecified depressive disorder, though to be sure, the patient was taking 75 mg of venlafaxine XR. While there were seasonal stressors, historically, nothing had risen to the level of MDD.
The obsessions driving his thoughts to jump off buildings and bridges had completely remitted. His cognitive ability returned to baseline with an ability to focus and perform the complicated tasks of his high-intensity work by the Dec. 8 psychiatric examination, where he was accompanied by his wife. He described feeling like, “I snapped back to like I was before this crazy stuff happened.” His mood was reported as, “Very good; like my old self” and this was confirmed by his wife. His affect was calmer and less tense. He was now using gabapentin sparingly for sleep. We continued to entertain discontinuing the venlafaxine XR, considering this recent severe episode likely driven by the COVID-19 virus. The decision was made to continue venlafaxine XR through the winter rather than discontinuing, remaining on the conservative side of treatment. The patient’s diagnosis was changed from “MDD, single episode,” to “mood disorder due to known physiologic condition (COVID-19) (F06.31) with depressive features; resolving.” At the patient’s follow-up examination on Jan. 5, 2022, he was continuing to do well, stating, “The whole series of crazy events happened to someone else.” The hydrochlorothiazide had been discontinued, and the patient’s blood pressure and pulse were normal at 119/81 and 69, respectively. He had made strategic changes at work to lessen stressors during the typically difficult months.
Discussion
Literature has discussed neuropsychiatric sequelae of COVID-19.2 The cited example questions whether psychiatric symptoms are tied directly to the viral infection or to the “host’s immune response.” We believe our case represents a direct neurocognitive/neuropsychiatric insult due to the COVID-19 infection.
This case presents a 55-year-old male with no previous psychiatric or medical history with new onset significant and debilitating cognitive impairment and obsessive thoughts of throwing himself from his bedroom balcony ending up at a bridge struggling with an irrational thought of jumping; ultimately requiring psychiatric hospitalization for acute suicidal thoughts. The patient’s psychiatric symptoms arose prior to any and all medication treatment. The obsessive thoughts correlated both with the onset of SARS-CoV-2 infection and a period of sleep deprivation subsequent to the infection. A course of steroid treatment and taper were started after the onset of neurocognitive-psychiatric symptoms, though there is close timing. We submit that the patient experienced, as part of the initial neurocognitive psychiatric initiating cascade, a COVID-19–induced sleep deprivation that was not etiologic but part of the process; since, even when sleep returned to normal, it was still several weeks before full cognitive function returned to baseline.
An argument could be made for possible MDD or unspecified depressive disorder, as historically there had been work-related stressors for the patient at this time of year because of the chronological nature of his work; though previously nothing presented with obsessional suicidal thinking and nothing with any cognitive impairment – let alone to this incapacitating degree.
The patient describes his seasonal work much like an accountant’s work at the beginning of each year. In the patient’s case, the months of September and October are historically “nonstop, working days,” which then slow down in the winter months for a period of recuperation. In gathering his past history of symptoms, he denied neurovegetative symptoms to meet full diagnostic criteria for MDD or unspecified depressive disorder, absent this episode in the presence of SARS-CoV-2 infection.
We could also consider a contributory negative “organic push” by the viral load and prednisone helping to express an underlying unspecified depression or MDD, but for the profound and unusual presentation. There was little prodrome of depressive symptoms (again, he reported his “typical” extraordinary work burden for this time of year, which is common in his industry).
In this patient, the symptoms have remitted completely. However, the patient is currently taking venlafaxine XR 75 mg. We have considered tapering and discontinuing the venlafaxine – since it is not entirely clear that he needs to be on this medication – so this question remains an open one. We did decide, however, to continue the venlafaxine until after the winter months and to reassess at that time.
Conclusion
The patient presented with new onset psychological and psychiatric symptoms in addition to physiologic symptoms; the former symptoms were not revealed prior to initial family medicine evaluation. As the symptoms worsened, he and his wife sought additional consultation with family physician, psychiatrists, and ED. Steroid treatment may have played a part in exacerbation of symptoms, but the neuropsychiatric cognitive symptoms were present prior to initiation of all pharmacologic and medical treatment. The successful outcome of this case was based upon quick action and collaboration between the family medicine physician, the psychiatrist, and the ED physician. The value of communication, assessment, and action via phone call and text cannot be overstated. Future considerations include further large-scale evaluation of multifaceted early treatment of patients with COVID-19 within the first 72 hours of symptoms to prevent not only hospitalization, morbidity, and mortality, but newly recognized psychological and psychiatric syndromes.3,4
Lastly, fluvoxamine might have been a better choice for adjunctive early treatment of COVID-19.5 As a matter of distinction, if a lingering mood disorder or obsessive-compulsive disorder remain a result of SARS-CoV-2 or if one were to start an antidepressant during the course of illness, it would be reasonable to consider fluvoxamine as a potential first-line agent.
Dr. Kohanski is a fellowship trained forensic psychiatrist and a diplomate of the American Board of Psychiatry & Neurology. She maintains a private practice in Somerset, N.J., and is a frequent media commentator and medical podcaster. Dr. Kohanski has no conflicts of interest. Dr. Wax is a residency-trained osteopathic family medicine physician in independent private practice in Mullica Hill, N.J. He has authored multiple papers over 2 decades on topics such as SARS-CoV-2 and COVID-19 early treatment. He has been a speaker and media host over 2 decades and served on the National Physicians Council on Healthcare Policy’s congressional subcommittee. Dr. Wax has no conflicts of interest.
References
1. Rev Cardiovasc Med. 2020 Dec 30;21(4):517-30.
2. Brain Behav Immun. 2020 Jul;87:34-9.
3. Trav Med Infect Dis. 2020 May-Jun 35;10738.
4. Kirsch S. “Early treatment for COVID is key to better outcomes.” Times of India. 2021 May 21.
5. Lancet. 2022 Jan 1;10(1):E42-E51.
An otherwise healthy 55-year-old male, with no previous psychiatric or medical history, sought care with a family medicine physician for the first time in decades.
Medical symptoms began Oct. 9, 2021, with “some leg weakness and mild sniffles.” Since he was going to be at a public event, he decided to take a PCR test for the SARS-CoV-2 virus on Oct. 13. The patient tested positive.
His symptoms continued to worsen, and he experienced severe body fatigue, sleep disturbance, and lethargy. “A few days after my positive test, the cognitive and physical symptoms dramatically ramped up,” the patient recalled.
Because of those worsening symptoms, on Oct. 20, the patient obtained a new patient appointment with a family medicine physician. After a telemedicine evaluation, the family medicine physician began a multifaceted early outpatient COVID-19 treatment protocol,1 as I (C.M.W.) and colleagues wrote about late last year. However, this treatment began late in the course because of the patient’s initial resistance to seek care.
This early outpatient treatment protocol for COVID-19 included vitamin D3 125 mcg (5,000 ICU), N-acetylcysteine (NAC) 600 mg every day x 30 days; acetylsalicylic acid 325 mg every day x 30 days; azithromycin 250 mg b.i.d. before every meal x 10 days; hydroxychloroquine sulfate 200 mg b.i.d. x 10 days; ivermectin 3 mg, 5 pills daily x 10 days; zinc sulfate 220 mg (50 mg elemental) every day x 30 days; and a prednisone taper (30 mg daily x 3 days, tapering down 5 mg every 3 days). Hydroxyzine 50 mg at bedtime as needed was added for sleep. The patient did not comment to the family physician on any of the psychological or psychiatric symptoms and responded appropriately to questions during the Oct. 20 initial evaluation.
However, he later described that around the time the PCR was positive, For example, he was watching a simple YouTube video for work and “everything was confusing me ... it rattled me, and I couldn’t understand it.” He described his COVID-19 mind as: “The words in my head would come out in a jumbled order, like the message from the words in my brain to my mouth would get crossed. I had trouble spelling and texting. Total cognitive breakdown. I couldn’t do simple mathematics.”
Despite his physical exhaustion, he endured a 3-day period of sleep deprivation. During this time, he recalled looking up at the roof and thinking, “I need to jump off the roof” or thinking, “I might want to throw myself under a bus.” He did not initially reveal his suicidal thoughts to his family medicine physician. After beginning COVID-19 treatment, the patient had two nights of sleep and felt notably improved, and his physical symptoms began to remit. However, the sleeplessness quickly returned “with a vengeance” along with “silly suicidal thoughts.” The thoughts took on a more obsessional quality. For example, he repeatedly thought of jumping out of his second-story bedroom to the living room below and was preoccupied by continually looking at people’s roofs and thinking about jumping. Those thoughts intensified and culminated in his “going missing,” leading his wife to call the police. It was discovered that he had driven to a local bridge and was contemplating jumping off.
After that “going missing” incident, the patient and his wife reached out to their family medicine physician. He reevaluated the patient and, given the new information about the psychiatric symptoms, strongly recommended stat crisis and psychiatric consultation. After discussing the case on the same day, both the family medicine physician and the psychiatrist recommended stat hospital emergency department (ED) assessment on Oct. 29. In the ED, a head CT without contrast at the recommendation of both psychiatrist and family physician, routine electrolytes, CBC with differential, and EKG all were within normal limits. The ED initially discharged him home after crisis evaluation, deciding he was not an imminent risk to himself or others.
The next day, the psychiatrist spoke on the phone with the patient, family medicine physician, and the patient’s wife to arrange an initial assessment. At that time, it remained unclear to all whether the obsessional thoughts had resolved to such a degree that the patient could resist acting upon them. Further, the patient’s sleep architecture had not returned to normal. All agreed another emergency ED assessment was indicated. Ultimately, after voluntary re-evaluation and a difficult hold in the crisis unit, the patient was admitted for psychiatric hospitalization on Oct. 29 and discharged on Nov. 4.
In the psychiatric hospital, venlafaxine XR was started and titrated to 75 mg. The patient was discovered to be hypertensive, and hydrochlorothiazide was started. The discharge diagnosis was major depressive disorder, single episode, severe, without psychotic features.
Posthospitalization course
He was seen for his initial psychiatric outpatient assessment postpsychiatric hospitalization on Nov. 9, as he had not yet been formally evaluated by the psychiatrist because of the emergency situation.
Gabapentin 300 mg by mouth at bedtime was started, and his sleep architecture was restored. The initial plan to titrate venlafaxine XR into dual selective norepinephrine reuptake inhibitor dose range was terminated, and his psychiatrist considered tapering and discontinuing the venlafaxine XR. A clinical examination, additional history, and collateral data no longer necessarily pointed to an active major depressive disorder or even unspecified depressive disorder, though to be sure, the patient was taking 75 mg of venlafaxine XR. While there were seasonal stressors, historically, nothing had risen to the level of MDD.
The obsessions driving his thoughts to jump off buildings and bridges had completely remitted. His cognitive ability returned to baseline with an ability to focus and perform the complicated tasks of his high-intensity work by the Dec. 8 psychiatric examination, where he was accompanied by his wife. He described feeling like, “I snapped back to like I was before this crazy stuff happened.” His mood was reported as, “Very good; like my old self” and this was confirmed by his wife. His affect was calmer and less tense. He was now using gabapentin sparingly for sleep. We continued to entertain discontinuing the venlafaxine XR, considering this recent severe episode likely driven by the COVID-19 virus. The decision was made to continue venlafaxine XR through the winter rather than discontinuing, remaining on the conservative side of treatment. The patient’s diagnosis was changed from “MDD, single episode,” to “mood disorder due to known physiologic condition (COVID-19) (F06.31) with depressive features; resolving.” At the patient’s follow-up examination on Jan. 5, 2022, he was continuing to do well, stating, “The whole series of crazy events happened to someone else.” The hydrochlorothiazide had been discontinued, and the patient’s blood pressure and pulse were normal at 119/81 and 69, respectively. He had made strategic changes at work to lessen stressors during the typically difficult months.
Discussion
Literature has discussed neuropsychiatric sequelae of COVID-19.2 The cited example questions whether psychiatric symptoms are tied directly to the viral infection or to the “host’s immune response.” We believe our case represents a direct neurocognitive/neuropsychiatric insult due to the COVID-19 infection.
This case presents a 55-year-old male with no previous psychiatric or medical history with new onset significant and debilitating cognitive impairment and obsessive thoughts of throwing himself from his bedroom balcony ending up at a bridge struggling with an irrational thought of jumping; ultimately requiring psychiatric hospitalization for acute suicidal thoughts. The patient’s psychiatric symptoms arose prior to any and all medication treatment. The obsessive thoughts correlated both with the onset of SARS-CoV-2 infection and a period of sleep deprivation subsequent to the infection. A course of steroid treatment and taper were started after the onset of neurocognitive-psychiatric symptoms, though there is close timing. We submit that the patient experienced, as part of the initial neurocognitive psychiatric initiating cascade, a COVID-19–induced sleep deprivation that was not etiologic but part of the process; since, even when sleep returned to normal, it was still several weeks before full cognitive function returned to baseline.
An argument could be made for possible MDD or unspecified depressive disorder, as historically there had been work-related stressors for the patient at this time of year because of the chronological nature of his work; though previously nothing presented with obsessional suicidal thinking and nothing with any cognitive impairment – let alone to this incapacitating degree.
The patient describes his seasonal work much like an accountant’s work at the beginning of each year. In the patient’s case, the months of September and October are historically “nonstop, working days,” which then slow down in the winter months for a period of recuperation. In gathering his past history of symptoms, he denied neurovegetative symptoms to meet full diagnostic criteria for MDD or unspecified depressive disorder, absent this episode in the presence of SARS-CoV-2 infection.
We could also consider a contributory negative “organic push” by the viral load and prednisone helping to express an underlying unspecified depression or MDD, but for the profound and unusual presentation. There was little prodrome of depressive symptoms (again, he reported his “typical” extraordinary work burden for this time of year, which is common in his industry).
In this patient, the symptoms have remitted completely. However, the patient is currently taking venlafaxine XR 75 mg. We have considered tapering and discontinuing the venlafaxine – since it is not entirely clear that he needs to be on this medication – so this question remains an open one. We did decide, however, to continue the venlafaxine until after the winter months and to reassess at that time.
Conclusion
The patient presented with new onset psychological and psychiatric symptoms in addition to physiologic symptoms; the former symptoms were not revealed prior to initial family medicine evaluation. As the symptoms worsened, he and his wife sought additional consultation with family physician, psychiatrists, and ED. Steroid treatment may have played a part in exacerbation of symptoms, but the neuropsychiatric cognitive symptoms were present prior to initiation of all pharmacologic and medical treatment. The successful outcome of this case was based upon quick action and collaboration between the family medicine physician, the psychiatrist, and the ED physician. The value of communication, assessment, and action via phone call and text cannot be overstated. Future considerations include further large-scale evaluation of multifaceted early treatment of patients with COVID-19 within the first 72 hours of symptoms to prevent not only hospitalization, morbidity, and mortality, but newly recognized psychological and psychiatric syndromes.3,4
Lastly, fluvoxamine might have been a better choice for adjunctive early treatment of COVID-19.5 As a matter of distinction, if a lingering mood disorder or obsessive-compulsive disorder remain a result of SARS-CoV-2 or if one were to start an antidepressant during the course of illness, it would be reasonable to consider fluvoxamine as a potential first-line agent.
Dr. Kohanski is a fellowship trained forensic psychiatrist and a diplomate of the American Board of Psychiatry & Neurology. She maintains a private practice in Somerset, N.J., and is a frequent media commentator and medical podcaster. Dr. Kohanski has no conflicts of interest. Dr. Wax is a residency-trained osteopathic family medicine physician in independent private practice in Mullica Hill, N.J. He has authored multiple papers over 2 decades on topics such as SARS-CoV-2 and COVID-19 early treatment. He has been a speaker and media host over 2 decades and served on the National Physicians Council on Healthcare Policy’s congressional subcommittee. Dr. Wax has no conflicts of interest.
References
1. Rev Cardiovasc Med. 2020 Dec 30;21(4):517-30.
2. Brain Behav Immun. 2020 Jul;87:34-9.
3. Trav Med Infect Dis. 2020 May-Jun 35;10738.
4. Kirsch S. “Early treatment for COVID is key to better outcomes.” Times of India. 2021 May 21.
5. Lancet. 2022 Jan 1;10(1):E42-E51.
HIV stigma persists globally, according to Harris poll
Four decades into the AIDS epidemic and for some, it’s as if gains in awareness, advances in prevention and treatment, and the concept of undetected equals untransmissable (U=U) never happened. In its place,
Accordingly, findings from a Harris poll conducted Oct. 13-18, 2021, among 5,047 adults (18 and older) residing in Australia, Portugal, the United Kingdom, and the United States, reveal that 88% of those surveyed believe that negative perceptions toward people living with HIV persist even though HIV infection can be effectively managed with antiretroviral therapy (ART). Conversely, three-quarters (76%) are unaware of U=U, and the fact that someone with HIV who is taking effective treatment cannot pass it on to their partner. Two-thirds incorrectly believe that a person living with HIV can pass it onto their baby, even when they are ART adherent.
“The survey made me think of people who work in HIV clinics, and how much of a bubble I think that we in the HIV field live in,” Nneka Nwokolo, MBBS, senior global medical director at ViiV Healthcare, London, and practicing consultant in sexual health and HIV medicine, told this news organization. “I think that we generally feel that everyone knows as much as we do or feels the way that we do.”
Misconceptions abound across the globe
The online survey, which was commissioned by ViiV Healthcare, also highlights that one in five adults do not know that anyone can acquire HIV regardless of lifestyle, thereby perpetuating the stereotype that HIV is a disease that only affects certain populations, such as men who have sex with men (MSM) or transgender women (TGW).
Pervasive stereotypes and stigmatization only serve to magnify preexisting social inequities that affect access to appropriate care. A recent editorial published in the journal AIDS and Behavior underscores that stigma experienced by marginalized populations in particular (for example, Black MSM, TGW) is directly linked to decreased access to and use of effective HIV prevention and treatment services. Additionally, once stigma becomes internalized, it might further affect overall well-being, mental health, and social support.
“One of the most significant consequences of the ongoing stigma is that people are scared to test and then they end up coming to services late [when] they’re really ill,” explained Dr. Nwokolo. “It goes back to the early days when HIV was a death sentence ... it’s still there. I have one patient who to this day hates the fact that he has HIV, that he has to come to the clinic – it’s a reminder of why he hates himself.”
Great strides in testing and advances in treatment might be helping to reframe HIV as a chronic but treatable and preventable disease. Nevertheless, survey findings also revealed that nearly three out of five adults incorrectly believe that a person living with HIV will have a shorter lifespan than someone who is HIV negative, even if they are on effective treatment.
These beliefs are especially true among Dr. Nwokolo’s patient base, most of whom are Africans who’ve immigrated to the United Kingdom from countries that have been devastated by the HIV epidemic. “Those who’ve never tested are reluctant to do so because they are afraid that they will have the same outcome as the people that they know that they’ve left behind,” she said.
HIV stigma in the era of 90-90-90
While there has been progress toward achieving UN AID’s 90-90-90 targets (that is, 90% living with HIV know their status, 90% who know their status are on ART, and 90% of people on ART are virally suppressed), exclusion and isolation – the key hallmarks of stigma – may ultimately be the most important barriers preventing a lofty goal to end the AIDS epidemic by the year 2030.
“Here we are, 40 years in and we are still facing such ignorance, some stigma,” Carl Schmid, MBA, former cochair of the Presidential Advisory Council on HIV/AIDS, and executive director of HIV+Policy Institute, told this news organization. “It’s gotten better, but it is really putting a damper on people being tested, getting treated, getting access to PrEP.” Mr. Schmid was not involved in the Harris Poll.
Mr. Schmid also said that, in addition to broader outreach and education as well as dissemination of information about HIV and AIDS from the White House and other government leaders, physician involvement is essential.
“They’re the ones that need to step up. They have to talk about sex with their patients, [but] they don’t do that, especially in the South among certain populations,” he noted.
Data support the unique challenges faced by at-risk individuals living in the southern United States. Not only do Southern states account for roughly half of all new HIV cases annually, but Black MSM and Black women account for the majority of new diagnoses, according to the Centers for Disease Control and Prevention. Data have also demonstrated discrimination and prejudice toward people with HIV persist among many medical professionals in the South (especially those working in rural areas).
But this is not only a Southern problem; a 2018 review of studies in clinicians across the United States published in AIDS Patient Care and STDs linked provider fear of acquiring HIV through occupational exposure to reduced quality of care, refusal of care, and anxiety, especially among providers with limited awareness of PrEP. Discordant attitudes around making a priority to address HIV-related stigma versus other health care needs also reduced overall care delivery and patient experience.
“I think that the first thing that we as HIV clinicians can and should do – and is definitely within our power to do – is to educate our peers about HIV,” Dr. Nwokolo said, “HIV has gone off the radar, but it’s still out there.”
The study was commissioned by Viiv Healthcare. Dr. Nwokolo is an employee of ViiV Healthcare. Mr. Schmid disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Four decades into the AIDS epidemic and for some, it’s as if gains in awareness, advances in prevention and treatment, and the concept of undetected equals untransmissable (U=U) never happened. In its place,
Accordingly, findings from a Harris poll conducted Oct. 13-18, 2021, among 5,047 adults (18 and older) residing in Australia, Portugal, the United Kingdom, and the United States, reveal that 88% of those surveyed believe that negative perceptions toward people living with HIV persist even though HIV infection can be effectively managed with antiretroviral therapy (ART). Conversely, three-quarters (76%) are unaware of U=U, and the fact that someone with HIV who is taking effective treatment cannot pass it on to their partner. Two-thirds incorrectly believe that a person living with HIV can pass it onto their baby, even when they are ART adherent.
“The survey made me think of people who work in HIV clinics, and how much of a bubble I think that we in the HIV field live in,” Nneka Nwokolo, MBBS, senior global medical director at ViiV Healthcare, London, and practicing consultant in sexual health and HIV medicine, told this news organization. “I think that we generally feel that everyone knows as much as we do or feels the way that we do.”
Misconceptions abound across the globe
The online survey, which was commissioned by ViiV Healthcare, also highlights that one in five adults do not know that anyone can acquire HIV regardless of lifestyle, thereby perpetuating the stereotype that HIV is a disease that only affects certain populations, such as men who have sex with men (MSM) or transgender women (TGW).
Pervasive stereotypes and stigmatization only serve to magnify preexisting social inequities that affect access to appropriate care. A recent editorial published in the journal AIDS and Behavior underscores that stigma experienced by marginalized populations in particular (for example, Black MSM, TGW) is directly linked to decreased access to and use of effective HIV prevention and treatment services. Additionally, once stigma becomes internalized, it might further affect overall well-being, mental health, and social support.
“One of the most significant consequences of the ongoing stigma is that people are scared to test and then they end up coming to services late [when] they’re really ill,” explained Dr. Nwokolo. “It goes back to the early days when HIV was a death sentence ... it’s still there. I have one patient who to this day hates the fact that he has HIV, that he has to come to the clinic – it’s a reminder of why he hates himself.”
Great strides in testing and advances in treatment might be helping to reframe HIV as a chronic but treatable and preventable disease. Nevertheless, survey findings also revealed that nearly three out of five adults incorrectly believe that a person living with HIV will have a shorter lifespan than someone who is HIV negative, even if they are on effective treatment.
These beliefs are especially true among Dr. Nwokolo’s patient base, most of whom are Africans who’ve immigrated to the United Kingdom from countries that have been devastated by the HIV epidemic. “Those who’ve never tested are reluctant to do so because they are afraid that they will have the same outcome as the people that they know that they’ve left behind,” she said.
HIV stigma in the era of 90-90-90
While there has been progress toward achieving UN AID’s 90-90-90 targets (that is, 90% living with HIV know their status, 90% who know their status are on ART, and 90% of people on ART are virally suppressed), exclusion and isolation – the key hallmarks of stigma – may ultimately be the most important barriers preventing a lofty goal to end the AIDS epidemic by the year 2030.
“Here we are, 40 years in and we are still facing such ignorance, some stigma,” Carl Schmid, MBA, former cochair of the Presidential Advisory Council on HIV/AIDS, and executive director of HIV+Policy Institute, told this news organization. “It’s gotten better, but it is really putting a damper on people being tested, getting treated, getting access to PrEP.” Mr. Schmid was not involved in the Harris Poll.
Mr. Schmid also said that, in addition to broader outreach and education as well as dissemination of information about HIV and AIDS from the White House and other government leaders, physician involvement is essential.
“They’re the ones that need to step up. They have to talk about sex with their patients, [but] they don’t do that, especially in the South among certain populations,” he noted.
Data support the unique challenges faced by at-risk individuals living in the southern United States. Not only do Southern states account for roughly half of all new HIV cases annually, but Black MSM and Black women account for the majority of new diagnoses, according to the Centers for Disease Control and Prevention. Data have also demonstrated discrimination and prejudice toward people with HIV persist among many medical professionals in the South (especially those working in rural areas).
But this is not only a Southern problem; a 2018 review of studies in clinicians across the United States published in AIDS Patient Care and STDs linked provider fear of acquiring HIV through occupational exposure to reduced quality of care, refusal of care, and anxiety, especially among providers with limited awareness of PrEP. Discordant attitudes around making a priority to address HIV-related stigma versus other health care needs also reduced overall care delivery and patient experience.
“I think that the first thing that we as HIV clinicians can and should do – and is definitely within our power to do – is to educate our peers about HIV,” Dr. Nwokolo said, “HIV has gone off the radar, but it’s still out there.”
The study was commissioned by Viiv Healthcare. Dr. Nwokolo is an employee of ViiV Healthcare. Mr. Schmid disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Four decades into the AIDS epidemic and for some, it’s as if gains in awareness, advances in prevention and treatment, and the concept of undetected equals untransmissable (U=U) never happened. In its place,
Accordingly, findings from a Harris poll conducted Oct. 13-18, 2021, among 5,047 adults (18 and older) residing in Australia, Portugal, the United Kingdom, and the United States, reveal that 88% of those surveyed believe that negative perceptions toward people living with HIV persist even though HIV infection can be effectively managed with antiretroviral therapy (ART). Conversely, three-quarters (76%) are unaware of U=U, and the fact that someone with HIV who is taking effective treatment cannot pass it on to their partner. Two-thirds incorrectly believe that a person living with HIV can pass it onto their baby, even when they are ART adherent.
“The survey made me think of people who work in HIV clinics, and how much of a bubble I think that we in the HIV field live in,” Nneka Nwokolo, MBBS, senior global medical director at ViiV Healthcare, London, and practicing consultant in sexual health and HIV medicine, told this news organization. “I think that we generally feel that everyone knows as much as we do or feels the way that we do.”
Misconceptions abound across the globe
The online survey, which was commissioned by ViiV Healthcare, also highlights that one in five adults do not know that anyone can acquire HIV regardless of lifestyle, thereby perpetuating the stereotype that HIV is a disease that only affects certain populations, such as men who have sex with men (MSM) or transgender women (TGW).
Pervasive stereotypes and stigmatization only serve to magnify preexisting social inequities that affect access to appropriate care. A recent editorial published in the journal AIDS and Behavior underscores that stigma experienced by marginalized populations in particular (for example, Black MSM, TGW) is directly linked to decreased access to and use of effective HIV prevention and treatment services. Additionally, once stigma becomes internalized, it might further affect overall well-being, mental health, and social support.
“One of the most significant consequences of the ongoing stigma is that people are scared to test and then they end up coming to services late [when] they’re really ill,” explained Dr. Nwokolo. “It goes back to the early days when HIV was a death sentence ... it’s still there. I have one patient who to this day hates the fact that he has HIV, that he has to come to the clinic – it’s a reminder of why he hates himself.”
Great strides in testing and advances in treatment might be helping to reframe HIV as a chronic but treatable and preventable disease. Nevertheless, survey findings also revealed that nearly three out of five adults incorrectly believe that a person living with HIV will have a shorter lifespan than someone who is HIV negative, even if they are on effective treatment.
These beliefs are especially true among Dr. Nwokolo’s patient base, most of whom are Africans who’ve immigrated to the United Kingdom from countries that have been devastated by the HIV epidemic. “Those who’ve never tested are reluctant to do so because they are afraid that they will have the same outcome as the people that they know that they’ve left behind,” she said.
HIV stigma in the era of 90-90-90
While there has been progress toward achieving UN AID’s 90-90-90 targets (that is, 90% living with HIV know their status, 90% who know their status are on ART, and 90% of people on ART are virally suppressed), exclusion and isolation – the key hallmarks of stigma – may ultimately be the most important barriers preventing a lofty goal to end the AIDS epidemic by the year 2030.
“Here we are, 40 years in and we are still facing such ignorance, some stigma,” Carl Schmid, MBA, former cochair of the Presidential Advisory Council on HIV/AIDS, and executive director of HIV+Policy Institute, told this news organization. “It’s gotten better, but it is really putting a damper on people being tested, getting treated, getting access to PrEP.” Mr. Schmid was not involved in the Harris Poll.
Mr. Schmid also said that, in addition to broader outreach and education as well as dissemination of information about HIV and AIDS from the White House and other government leaders, physician involvement is essential.
“They’re the ones that need to step up. They have to talk about sex with their patients, [but] they don’t do that, especially in the South among certain populations,” he noted.
Data support the unique challenges faced by at-risk individuals living in the southern United States. Not only do Southern states account for roughly half of all new HIV cases annually, but Black MSM and Black women account for the majority of new diagnoses, according to the Centers for Disease Control and Prevention. Data have also demonstrated discrimination and prejudice toward people with HIV persist among many medical professionals in the South (especially those working in rural areas).
But this is not only a Southern problem; a 2018 review of studies in clinicians across the United States published in AIDS Patient Care and STDs linked provider fear of acquiring HIV through occupational exposure to reduced quality of care, refusal of care, and anxiety, especially among providers with limited awareness of PrEP. Discordant attitudes around making a priority to address HIV-related stigma versus other health care needs also reduced overall care delivery and patient experience.
“I think that the first thing that we as HIV clinicians can and should do – and is definitely within our power to do – is to educate our peers about HIV,” Dr. Nwokolo said, “HIV has gone off the radar, but it’s still out there.”
The study was commissioned by Viiv Healthcare. Dr. Nwokolo is an employee of ViiV Healthcare. Mr. Schmid disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Open-label placebo improves symptoms in pediatric IBS and functional abdominal pain
A spoonful of sugar helps the medicine go down – but what if the sugar is the medicine?
Nearly three in four children with irritable bowel syndrome (IBS) or unexplained abdominal pain reported at least a 30% improvement in discomfort after taking a regimen of sugar water they knew had no medicinal properties.
The findings, published online in JAMA Pediatrics on Jan. 31, 2022, also revealed that participants used significantly less rescue medications when taking the so-called “open-label placebo.” The magnitude of the effect was enough to meet one of the criteria from the Food and Drug Administration to approve drugs to treat IBS, which affects between 10% and 15% of U.S. children.
Although open-label placebo is not ready for clinical use, IBS expert Miranda van Tilburg, PhD, said she is “glad we have evidence” of a strong response in this patient population and that the results “may make clinicians rethink how they introduce treatments.
“By emphasizing their belief that a treatment may work, clinicians can harness the placebo effect,” Dr. van Tilburg, professor of medicine and vice chair of research at Marshall University, Huntington, W.Va., told this news organization.
Study leader Samuel Nurko, MD, MPH, the director of the functional abdominal pain program at Harvard Medical School, Boston, said placebo-controlled trials in patients with IBS and functional abdominal pain consistently show a “very high placebo response.” The question his group set out to answer, he said, was: “Can we get the pain symptoms of these children better by giving them placebo with no deception?”
Between 2015 and 2018, Dr. Nurko and colleagues randomly assigned 30 children and adolescents, aged 8-18 years, with IBS or functional abdominal pain to receive either an open-label inert liquid placebo – consisting of 85% sucrose, citric acid, purified water, and the preservative methyl paraben – twice daily for 3 weeks followed by 3 weeks with no placebo, or to follow the reverse sequence. Roughly half (53%) of the children had functional abdominal pain, and 47% had IBS as defined by Rome III criteria.
Researchers at the three participating clinical sites followed a standardized protocol for explaining the nature of placebo (“like sugar pills without medication”), telling participants that adults with conditions like theirs often benefit from placebo when they receive it as part of blinded, randomized clinical trials. Participants in the study were allowed to use hyoscyamine, an anticholinergic medication, as rescue treatment during the trial.
Dr. Nurko’s team reported that patients had a mean pain score of 39.9 on a 100-point visual analogue scale during the open-label placebo phase of the trial and a mean score of 45 during the control period. That difference was statistically significant (P = .03).
Participants took an average of two hyoscyamine pills during the placebo phase, compared with 3.8 pills during the 3-week period when they did not receive placebo (P < .001).
Nearly three-fourths (73.3%) of children in the study reported that open-label placebo improved their pain by over 30%, thus meeting one of the FDA’s criteria for clinical evaluation of drugs for IBS. Half said the placebo liquid cut their pain by more than 50%.
Dr. Nurko said the findings highlight the need to address “mind-body connections” in the management of gut-brain disorders. Like Dr. van Tilburg, he cautioned that open-label placebo “is not ready for widespread use. Placebo is complicated, and we need to understand the mechanism” underlying its efficacy.
“The idea is eventually we will be able to sort out the exact mechanism and harness it for clinical practice,” he added.
However, Dr. van Tilburg expressed that using placebo therapy to treat children and adolescents with these conditions could send the message that “the pain is not real or all in their heads. Children with chronic pain encounter a lot of stigma, and this kind of treatment may increase the feeling of not being believed. We should be careful to avoid this.”
The study was funded by the National Institutes of Health, the Swiss National Science Foundation, the Schwartz family fund, the Foundation for the Science of the Therapeutic Relationship, and the Morgan Family Foundation.
A version of this article first appeared on Medscape.com.
A spoonful of sugar helps the medicine go down – but what if the sugar is the medicine?
Nearly three in four children with irritable bowel syndrome (IBS) or unexplained abdominal pain reported at least a 30% improvement in discomfort after taking a regimen of sugar water they knew had no medicinal properties.
The findings, published online in JAMA Pediatrics on Jan. 31, 2022, also revealed that participants used significantly less rescue medications when taking the so-called “open-label placebo.” The magnitude of the effect was enough to meet one of the criteria from the Food and Drug Administration to approve drugs to treat IBS, which affects between 10% and 15% of U.S. children.
Although open-label placebo is not ready for clinical use, IBS expert Miranda van Tilburg, PhD, said she is “glad we have evidence” of a strong response in this patient population and that the results “may make clinicians rethink how they introduce treatments.
“By emphasizing their belief that a treatment may work, clinicians can harness the placebo effect,” Dr. van Tilburg, professor of medicine and vice chair of research at Marshall University, Huntington, W.Va., told this news organization.
Study leader Samuel Nurko, MD, MPH, the director of the functional abdominal pain program at Harvard Medical School, Boston, said placebo-controlled trials in patients with IBS and functional abdominal pain consistently show a “very high placebo response.” The question his group set out to answer, he said, was: “Can we get the pain symptoms of these children better by giving them placebo with no deception?”
Between 2015 and 2018, Dr. Nurko and colleagues randomly assigned 30 children and adolescents, aged 8-18 years, with IBS or functional abdominal pain to receive either an open-label inert liquid placebo – consisting of 85% sucrose, citric acid, purified water, and the preservative methyl paraben – twice daily for 3 weeks followed by 3 weeks with no placebo, or to follow the reverse sequence. Roughly half (53%) of the children had functional abdominal pain, and 47% had IBS as defined by Rome III criteria.
Researchers at the three participating clinical sites followed a standardized protocol for explaining the nature of placebo (“like sugar pills without medication”), telling participants that adults with conditions like theirs often benefit from placebo when they receive it as part of blinded, randomized clinical trials. Participants in the study were allowed to use hyoscyamine, an anticholinergic medication, as rescue treatment during the trial.
Dr. Nurko’s team reported that patients had a mean pain score of 39.9 on a 100-point visual analogue scale during the open-label placebo phase of the trial and a mean score of 45 during the control period. That difference was statistically significant (P = .03).
Participants took an average of two hyoscyamine pills during the placebo phase, compared with 3.8 pills during the 3-week period when they did not receive placebo (P < .001).
Nearly three-fourths (73.3%) of children in the study reported that open-label placebo improved their pain by over 30%, thus meeting one of the FDA’s criteria for clinical evaluation of drugs for IBS. Half said the placebo liquid cut their pain by more than 50%.
Dr. Nurko said the findings highlight the need to address “mind-body connections” in the management of gut-brain disorders. Like Dr. van Tilburg, he cautioned that open-label placebo “is not ready for widespread use. Placebo is complicated, and we need to understand the mechanism” underlying its efficacy.
“The idea is eventually we will be able to sort out the exact mechanism and harness it for clinical practice,” he added.
However, Dr. van Tilburg expressed that using placebo therapy to treat children and adolescents with these conditions could send the message that “the pain is not real or all in their heads. Children with chronic pain encounter a lot of stigma, and this kind of treatment may increase the feeling of not being believed. We should be careful to avoid this.”
The study was funded by the National Institutes of Health, the Swiss National Science Foundation, the Schwartz family fund, the Foundation for the Science of the Therapeutic Relationship, and the Morgan Family Foundation.
A version of this article first appeared on Medscape.com.
A spoonful of sugar helps the medicine go down – but what if the sugar is the medicine?
Nearly three in four children with irritable bowel syndrome (IBS) or unexplained abdominal pain reported at least a 30% improvement in discomfort after taking a regimen of sugar water they knew had no medicinal properties.
The findings, published online in JAMA Pediatrics on Jan. 31, 2022, also revealed that participants used significantly less rescue medications when taking the so-called “open-label placebo.” The magnitude of the effect was enough to meet one of the criteria from the Food and Drug Administration to approve drugs to treat IBS, which affects between 10% and 15% of U.S. children.
Although open-label placebo is not ready for clinical use, IBS expert Miranda van Tilburg, PhD, said she is “glad we have evidence” of a strong response in this patient population and that the results “may make clinicians rethink how they introduce treatments.
“By emphasizing their belief that a treatment may work, clinicians can harness the placebo effect,” Dr. van Tilburg, professor of medicine and vice chair of research at Marshall University, Huntington, W.Va., told this news organization.
Study leader Samuel Nurko, MD, MPH, the director of the functional abdominal pain program at Harvard Medical School, Boston, said placebo-controlled trials in patients with IBS and functional abdominal pain consistently show a “very high placebo response.” The question his group set out to answer, he said, was: “Can we get the pain symptoms of these children better by giving them placebo with no deception?”
Between 2015 and 2018, Dr. Nurko and colleagues randomly assigned 30 children and adolescents, aged 8-18 years, with IBS or functional abdominal pain to receive either an open-label inert liquid placebo – consisting of 85% sucrose, citric acid, purified water, and the preservative methyl paraben – twice daily for 3 weeks followed by 3 weeks with no placebo, or to follow the reverse sequence. Roughly half (53%) of the children had functional abdominal pain, and 47% had IBS as defined by Rome III criteria.
Researchers at the three participating clinical sites followed a standardized protocol for explaining the nature of placebo (“like sugar pills without medication”), telling participants that adults with conditions like theirs often benefit from placebo when they receive it as part of blinded, randomized clinical trials. Participants in the study were allowed to use hyoscyamine, an anticholinergic medication, as rescue treatment during the trial.
Dr. Nurko’s team reported that patients had a mean pain score of 39.9 on a 100-point visual analogue scale during the open-label placebo phase of the trial and a mean score of 45 during the control period. That difference was statistically significant (P = .03).
Participants took an average of two hyoscyamine pills during the placebo phase, compared with 3.8 pills during the 3-week period when they did not receive placebo (P < .001).
Nearly three-fourths (73.3%) of children in the study reported that open-label placebo improved their pain by over 30%, thus meeting one of the FDA’s criteria for clinical evaluation of drugs for IBS. Half said the placebo liquid cut their pain by more than 50%.
Dr. Nurko said the findings highlight the need to address “mind-body connections” in the management of gut-brain disorders. Like Dr. van Tilburg, he cautioned that open-label placebo “is not ready for widespread use. Placebo is complicated, and we need to understand the mechanism” underlying its efficacy.
“The idea is eventually we will be able to sort out the exact mechanism and harness it for clinical practice,” he added.
However, Dr. van Tilburg expressed that using placebo therapy to treat children and adolescents with these conditions could send the message that “the pain is not real or all in their heads. Children with chronic pain encounter a lot of stigma, and this kind of treatment may increase the feeling of not being believed. We should be careful to avoid this.”
The study was funded by the National Institutes of Health, the Swiss National Science Foundation, the Schwartz family fund, the Foundation for the Science of the Therapeutic Relationship, and the Morgan Family Foundation.
A version of this article first appeared on Medscape.com.
FROM JAMA PEDIATRICS
Childhood trauma may influence vaccine hesitancy
, data published Feb. 1 suggest.
The findings by Mark A. Bellis, DSc, College of Human Sciences, Bangor (Wales) University, and colleagues were published online in BMJ Open.
The results are especially significant, the authors say, because of the prevalence of adverse childhood experiences (ACEs) globally, with proportions of people having multiple traumas in some countries at 10% or more of the population.
The authors wrote that hesitancy or refusal to get the vaccine increased with the number of traumas reported.
For example, hesitancy was three times higher among people who had experienced four or more types of childhood trauma than among those who did not report any traumatic events.
Dr. Bellis told this news organization that though their work suggests that higher levels of ACEs are linked with higher vaccine hesitancy, it is by no means the only reason people choose not to get vaccinated.
However, he said, the association they found may have key messages for clinicians.
“For clinicians, simply being trauma informed can help,” Dr. Bellis said. “Understanding how such childhood adversity can affect people may help them when discussing vaccines, and in understanding resistance to what is a complex medical issue and one that requires considerable trust. What can appear routine to a clinician may be a difficult leap of faith especially for those who have poorer experiences of trusting even within family settings.”
More trauma, less trust
The authors used responses to a nationally representative telephone survey of adults in Wales taken between December 2020 and March 2021, when COVID-19 restrictions were in force. Out of 6,763 people contacted, 2,285 met all criteria and answered all the questions and were included in the final analysis.
The survey asked about nine types of ACEs before the age of 18, including: parental separation; physical, verbal, and sexual abuse; exposure to domestic violence; and living with a household member who has mental illness, misuses alcohol and/or drugs, or who was incarcerated.
It also included personal details and long-term health information.
About half of the respondents said they hadn’t experienced any childhood trauma. Of those who did, one in five said they had experienced one type, 17% reported two to three types, and 10% reported four or more.
According to the authors, prevalence of ACEs reported was consistent with other comparable population surveys, including those conducted face to face.
They also investigated measures of trust and preference for different health regulations.
People with more ACEs were more likely to have low trust in National Health Service COVID-19 information.
“Other sociodemographics and a history of either chronic disease or COVID-19 infection were not significantly associated with low trust,” the authors pointed out.
People reporting higher ACEs also were more likely to report that they felt they were unfairly restricted by the government. People with four or more ACEs were twice as likely than were those with no ACEs to say they felt unfairly restricted and wanted rules such as mandatory masking to stop.
People with four or more types of trauma were almost twice as likely to ignore the restrictions as were those who hadn’t experienced any – 38% versus 21% – to ignore the restrictions, even after the researchers accounted for associations with sociodemographic factors and previous COVID-19 infection or a history of long-term conditions.
“Clinicians can be a powerful voice to counter more alarmist or even conspiratorial messages that might otherwise resonate with those who find trust difficult,” Dr. Bellis said.
He said that the effect of childhood adversity needs to be considered at all levels in health systems. Overarching public health strategists should include ways to earn trust to counter resistance in some of the most vulnerable communities where ACEs can be higher.
It will also be important in the short-term to “provide reassurance, build community champions, and understand the low base from which trust needs to be built,” he said.
Loss of control
“Past traumatic experiences can predispose someone to avoid things that remind them of that trauma. This avoidance protects them from re-experiencing the negative symptoms and behaviors that come with it. Whether this results into hesitancy of something that would benefit their health is not well known,” Consuelo Cagande, MD, senior associate program director and fellowship adviser in the department of child and adolescent psychiatry and behavioral sciences, Children’s Hospital of Philadelphia, told this news organization.
She pointed out a limitation the authors mention that is common when using ACEs as a measure linking to future negative behaviors – that people self-report them and may misremember or misreport them.
Another limitation is the potential for self-selection bias, as participation level was 36.4%, though the authors noted that is not unusual for unsolicited telephone surveys.
Dr. Cagande said that fearing loss of control may be another factor at play in having to follow restrictions, such as quarantining and masking, social distancing, or mandated vaccinations.
She said it’s important to understand a person’s reason for hesitancy to vaccines and work with the person with the help of the community, to help them trust and feel safe.
Young adults of particular concern
The 18- to 29-year-old age group is of particular concern, Dr. Bellis said.
The researchers estimated the likely rates of vaccine hesitancy according to childhood trauma and age, and the numbers ranged from around 3.5% among those aged 70 and older with no experience of childhood adversity to 38% among 18- to 29-year-olds who had experienced four or more types of childhood trauma.
“Childhood adversity can be an especially raw issue in this group,” he explained. “Some have already been obliged to sacrifice substantial proportions of their teenage lives and some will have suffered greater exposure to adverse childhood experiences as a result of being isolated during the pandemic, sometimes in difficult home environments. Our results suggest that this age group and especially those with high levels of ACEs are some of the most likely to be vaccine hesitant.”
This work was supported by Public Health Wales. The study authors and Dr. Cagande reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, data published Feb. 1 suggest.
The findings by Mark A. Bellis, DSc, College of Human Sciences, Bangor (Wales) University, and colleagues were published online in BMJ Open.
The results are especially significant, the authors say, because of the prevalence of adverse childhood experiences (ACEs) globally, with proportions of people having multiple traumas in some countries at 10% or more of the population.
The authors wrote that hesitancy or refusal to get the vaccine increased with the number of traumas reported.
For example, hesitancy was three times higher among people who had experienced four or more types of childhood trauma than among those who did not report any traumatic events.
Dr. Bellis told this news organization that though their work suggests that higher levels of ACEs are linked with higher vaccine hesitancy, it is by no means the only reason people choose not to get vaccinated.
However, he said, the association they found may have key messages for clinicians.
“For clinicians, simply being trauma informed can help,” Dr. Bellis said. “Understanding how such childhood adversity can affect people may help them when discussing vaccines, and in understanding resistance to what is a complex medical issue and one that requires considerable trust. What can appear routine to a clinician may be a difficult leap of faith especially for those who have poorer experiences of trusting even within family settings.”
More trauma, less trust
The authors used responses to a nationally representative telephone survey of adults in Wales taken between December 2020 and March 2021, when COVID-19 restrictions were in force. Out of 6,763 people contacted, 2,285 met all criteria and answered all the questions and were included in the final analysis.
The survey asked about nine types of ACEs before the age of 18, including: parental separation; physical, verbal, and sexual abuse; exposure to domestic violence; and living with a household member who has mental illness, misuses alcohol and/or drugs, or who was incarcerated.
It also included personal details and long-term health information.
About half of the respondents said they hadn’t experienced any childhood trauma. Of those who did, one in five said they had experienced one type, 17% reported two to three types, and 10% reported four or more.
According to the authors, prevalence of ACEs reported was consistent with other comparable population surveys, including those conducted face to face.
They also investigated measures of trust and preference for different health regulations.
People with more ACEs were more likely to have low trust in National Health Service COVID-19 information.
“Other sociodemographics and a history of either chronic disease or COVID-19 infection were not significantly associated with low trust,” the authors pointed out.
People reporting higher ACEs also were more likely to report that they felt they were unfairly restricted by the government. People with four or more ACEs were twice as likely than were those with no ACEs to say they felt unfairly restricted and wanted rules such as mandatory masking to stop.
People with four or more types of trauma were almost twice as likely to ignore the restrictions as were those who hadn’t experienced any – 38% versus 21% – to ignore the restrictions, even after the researchers accounted for associations with sociodemographic factors and previous COVID-19 infection or a history of long-term conditions.
“Clinicians can be a powerful voice to counter more alarmist or even conspiratorial messages that might otherwise resonate with those who find trust difficult,” Dr. Bellis said.
He said that the effect of childhood adversity needs to be considered at all levels in health systems. Overarching public health strategists should include ways to earn trust to counter resistance in some of the most vulnerable communities where ACEs can be higher.
It will also be important in the short-term to “provide reassurance, build community champions, and understand the low base from which trust needs to be built,” he said.
Loss of control
“Past traumatic experiences can predispose someone to avoid things that remind them of that trauma. This avoidance protects them from re-experiencing the negative symptoms and behaviors that come with it. Whether this results into hesitancy of something that would benefit their health is not well known,” Consuelo Cagande, MD, senior associate program director and fellowship adviser in the department of child and adolescent psychiatry and behavioral sciences, Children’s Hospital of Philadelphia, told this news organization.
She pointed out a limitation the authors mention that is common when using ACEs as a measure linking to future negative behaviors – that people self-report them and may misremember or misreport them.
Another limitation is the potential for self-selection bias, as participation level was 36.4%, though the authors noted that is not unusual for unsolicited telephone surveys.
Dr. Cagande said that fearing loss of control may be another factor at play in having to follow restrictions, such as quarantining and masking, social distancing, or mandated vaccinations.
She said it’s important to understand a person’s reason for hesitancy to vaccines and work with the person with the help of the community, to help them trust and feel safe.
Young adults of particular concern
The 18- to 29-year-old age group is of particular concern, Dr. Bellis said.
The researchers estimated the likely rates of vaccine hesitancy according to childhood trauma and age, and the numbers ranged from around 3.5% among those aged 70 and older with no experience of childhood adversity to 38% among 18- to 29-year-olds who had experienced four or more types of childhood trauma.
“Childhood adversity can be an especially raw issue in this group,” he explained. “Some have already been obliged to sacrifice substantial proportions of their teenage lives and some will have suffered greater exposure to adverse childhood experiences as a result of being isolated during the pandemic, sometimes in difficult home environments. Our results suggest that this age group and especially those with high levels of ACEs are some of the most likely to be vaccine hesitant.”
This work was supported by Public Health Wales. The study authors and Dr. Cagande reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, data published Feb. 1 suggest.
The findings by Mark A. Bellis, DSc, College of Human Sciences, Bangor (Wales) University, and colleagues were published online in BMJ Open.
The results are especially significant, the authors say, because of the prevalence of adverse childhood experiences (ACEs) globally, with proportions of people having multiple traumas in some countries at 10% or more of the population.
The authors wrote that hesitancy or refusal to get the vaccine increased with the number of traumas reported.
For example, hesitancy was three times higher among people who had experienced four or more types of childhood trauma than among those who did not report any traumatic events.
Dr. Bellis told this news organization that though their work suggests that higher levels of ACEs are linked with higher vaccine hesitancy, it is by no means the only reason people choose not to get vaccinated.
However, he said, the association they found may have key messages for clinicians.
“For clinicians, simply being trauma informed can help,” Dr. Bellis said. “Understanding how such childhood adversity can affect people may help them when discussing vaccines, and in understanding resistance to what is a complex medical issue and one that requires considerable trust. What can appear routine to a clinician may be a difficult leap of faith especially for those who have poorer experiences of trusting even within family settings.”
More trauma, less trust
The authors used responses to a nationally representative telephone survey of adults in Wales taken between December 2020 and March 2021, when COVID-19 restrictions were in force. Out of 6,763 people contacted, 2,285 met all criteria and answered all the questions and were included in the final analysis.
The survey asked about nine types of ACEs before the age of 18, including: parental separation; physical, verbal, and sexual abuse; exposure to domestic violence; and living with a household member who has mental illness, misuses alcohol and/or drugs, or who was incarcerated.
It also included personal details and long-term health information.
About half of the respondents said they hadn’t experienced any childhood trauma. Of those who did, one in five said they had experienced one type, 17% reported two to three types, and 10% reported four or more.
According to the authors, prevalence of ACEs reported was consistent with other comparable population surveys, including those conducted face to face.
They also investigated measures of trust and preference for different health regulations.
People with more ACEs were more likely to have low trust in National Health Service COVID-19 information.
“Other sociodemographics and a history of either chronic disease or COVID-19 infection were not significantly associated with low trust,” the authors pointed out.
People reporting higher ACEs also were more likely to report that they felt they were unfairly restricted by the government. People with four or more ACEs were twice as likely than were those with no ACEs to say they felt unfairly restricted and wanted rules such as mandatory masking to stop.
People with four or more types of trauma were almost twice as likely to ignore the restrictions as were those who hadn’t experienced any – 38% versus 21% – to ignore the restrictions, even after the researchers accounted for associations with sociodemographic factors and previous COVID-19 infection or a history of long-term conditions.
“Clinicians can be a powerful voice to counter more alarmist or even conspiratorial messages that might otherwise resonate with those who find trust difficult,” Dr. Bellis said.
He said that the effect of childhood adversity needs to be considered at all levels in health systems. Overarching public health strategists should include ways to earn trust to counter resistance in some of the most vulnerable communities where ACEs can be higher.
It will also be important in the short-term to “provide reassurance, build community champions, and understand the low base from which trust needs to be built,” he said.
Loss of control
“Past traumatic experiences can predispose someone to avoid things that remind them of that trauma. This avoidance protects them from re-experiencing the negative symptoms and behaviors that come with it. Whether this results into hesitancy of something that would benefit their health is not well known,” Consuelo Cagande, MD, senior associate program director and fellowship adviser in the department of child and adolescent psychiatry and behavioral sciences, Children’s Hospital of Philadelphia, told this news organization.
She pointed out a limitation the authors mention that is common when using ACEs as a measure linking to future negative behaviors – that people self-report them and may misremember or misreport them.
Another limitation is the potential for self-selection bias, as participation level was 36.4%, though the authors noted that is not unusual for unsolicited telephone surveys.
Dr. Cagande said that fearing loss of control may be another factor at play in having to follow restrictions, such as quarantining and masking, social distancing, or mandated vaccinations.
She said it’s important to understand a person’s reason for hesitancy to vaccines and work with the person with the help of the community, to help them trust and feel safe.
Young adults of particular concern
The 18- to 29-year-old age group is of particular concern, Dr. Bellis said.
The researchers estimated the likely rates of vaccine hesitancy according to childhood trauma and age, and the numbers ranged from around 3.5% among those aged 70 and older with no experience of childhood adversity to 38% among 18- to 29-year-olds who had experienced four or more types of childhood trauma.
“Childhood adversity can be an especially raw issue in this group,” he explained. “Some have already been obliged to sacrifice substantial proportions of their teenage lives and some will have suffered greater exposure to adverse childhood experiences as a result of being isolated during the pandemic, sometimes in difficult home environments. Our results suggest that this age group and especially those with high levels of ACEs are some of the most likely to be vaccine hesitant.”
This work was supported by Public Health Wales. The study authors and Dr. Cagande reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM BMJ OPEN
Intervention in Acute Hospital Unit Reduces Delirium Incidence for Older Adults, Has No Effect on Length of Stay, Other Complications
Study Overview
Objective: To examine the effect of the intervention “Eat Walk Engage,” a program that is designed to more consistently deliver age-friendly principles of care to older individuals in acute medical and surgical wards.
Design: This cluster randomized trial to examine the effect of an intervention in acute medical and surgical wards on older adults was conducted in 8 acute medical and surgical wards in 4 public hospitals in Australia from 2016 to 2017. To be eligible to participate in this trial, wards had to have the following: a patient population with 50% of patients aged 65 years and older; perceived alignment with hospital priorities; and nurse manager agreement to participation. Randomization was stratified by hospital, resulting in 4 wards with the intervention (a general medicine ward, an orthopedic ward, a general surgery ward, and a respiratory medicine ward) and 4 control wards (2 general medicine wards, a respiratory medicine ward, and a general surgery ward). Participants were consecutive inpatients aged 65 years or older who were admitted to the ward for at least 3 consecutive days during the study time period. Exclusion criteria included terminal or critical illness, severe cognitive impairment without a surrogate decision-maker, non-English speaking, or previously enrolled in the trial. Of a total of 453 patients who were eligible from the intervention wards, 188 were excluded and 6 died, yielding 259 participants in the intervention group. There were 413 patients eligible from the control wards, with 139 excluded and 3 deaths, yielding 271 participants in the control group.
Intervention: The intervention, called “Eat Walk Engage,” was developed to target older adults at risk for hospital-associated complications of delirium, functional decline, pressure injuries, falls, and incontinence, and aimed to improve care practices, environment, and culture to support age-friendly principles. This ward-based program delivered a structured improvement intervention through a site facilitator who is a nurse or allied health professional. The site facilitator identified opportunities for improvement using structured assessments of context, patient-experience interviews, and audits of care processes, and engaged an interdisciplinary working group from the intervention wards to participate in an hour-per-month meeting to develop plans for iterative improvements. Each site developed their own intervention plan; examples of interventions include shifting priorities to enable staff to increase the proportion of patients sitting in a chair for meals; designating the patient lounge as a walking destination to increase the proportion of time patients spend mobile; and using orientation boards and small groups to engage older patients in meaningful activities.
Main outcome measures: Study outcome measures included hospital-associated complications for older people, which is a composite of hospital-associated delirium, hospital-associated disability, hospital-associated incontinence, and fall or pressure injury during hospitalization. Delirium was assessed using the 3-minute diagnostic interview for Confusion Assessment Method (3D-CAM); hospital-associated disability was defined as new disability at discharge compared to 2 weeks prior to hospitalization. The primary outcome was defined as incidence of any complications and hospital length of stay. Secondary outcomes included incidence of individual complications, hospital discharge to facility, mortality at 6 months, and readmission for any cause at 6 months.
Main results: Patient characteristics for the intervention and control groups, respectively, were: 47% women with a mean age of 75.9 years (SD, 7.3), and 53% women with a mean age of 78.0 years (SD, 8.2). For the primary outcome, 46.4% of participants in the intervention group experienced any hospital complications compared with 51.8% in the control group (odds ratio [OR], 1.07; 95% CI, 0.71-1.61). The incidence of delirium was lower in the intervention group as compared with the control group (15.9% vs 31.4%; OR, 0.53; 95% CI, 0.31-0.90), while there were no other differences in the incidence rates of other complications. There was also no difference in hospital length of stay; median length of stay in the intervention group was 6 days (interquartile range [IQR], 4-9 days) compared with 7 days in the control group (IQR, 5-10), with an estimated mean difference in length of stay of 0.16 days (95% CI, –0.43 to 0.78 days). There was also no significant difference in mortality or all-cause readmission at 6 months.
Conclusion: The intervention “Eat Walk Engage” did not reduce hospital-associated complications overall or hospital length of stay, but it did reduce the incidence of hospital-associated delirium.
Commentary
Older adults, often with reduced physiologic reserve, when admitted to the hospital with an acute illness may be vulnerable to potential hazards of hospitalization, such as complications from prolonged periods of immobility, pressure injury, and delirium.1 Models of care in the inpatient setting to reduce these hazards, including the Acute Care for the Elderly model and the Mobile Acute Care for the Elderly Team model, have been examined in clinical trials.2,3 Specifically, models of care to prevent and treat delirium have been developed and tested over the past decade.4 The effect of these models in improving function, reducing complications, and reducing delirium incidence has been well documented. The present study adds to the literature by testing a model that utilizes implementation science methods to take into account real-world settings. In contrast with prior models-of-care studies, the implementation of the intervention at each ward was not prescriptive, but rather was developed in each ward in an iterative manner with stakeholder input. The advantage of this approach is that engagement of stakeholders at each intervention ward obtains buy-in from staff, mobilizing staff in a way that a prescriptive model of care may not; this ultimately may lead to longer-lasting change. The iterative approach also allows for the intervention to be adapted to conditions and settings over time. Other studies have taken this approach of using implementation science to drive change.5 Although the intervention in the present study failed to improve the primary outcome, it did reduce the incidence of delirium, which is a significant outcome and one that may confer considerable benefits to older adults under the model’s care.
A limitation of the intervention’s nonprescriptive approach is that, because of the variation of the interventions across sites, it is difficult to discern what elements drove the clinical outcomes. In addition, it would be challenging to consider what aspects of the intervention did not work should refinement or changes be needed. How one may measure fidelity to the intervention or how well a site implements the intervention and its relationship with clinical outcomes will need to be examined further.
Application for Clinical Practice
Clinicians look to effective models of care to improve clinical outcomes for older adults in the hospital. The intervention described in this study offers a real-world approach that may need less upfront investment than other recently studied models, such as the Acute Care for the Elderly model, which requires structural and staffing enhancements. Clinicians and health system leaders may consider implementing this model to improve the care delivered to older adults in the hospital as it may help reduce the incidence of delirium among the older adults they serve.
–William W. Hung, MD, MPH
Disclosures: None.
1. Creditor MC. Hazards of hospitalization of the elderly. Ann Intern Med. 1993;118(3):219-223. doi:10.7326/0003-4819-118-3-199302010-00011
2. Fox MT, Persaud M, Maimets I, et al. Effectiveness of acute geriatric unit care using acute care for elders components: a systematic review and meta-analysis. J Am Geriatr Soc. 2012;60(12):2237-2245. doi:10.1111/jgs.12028
3. Hung WW, Ross JS, Farber J, Siu AL. Evaluation of the Mobile Acute Care of the Elderly (MACE) service. JAMA Intern Med. 2013;173(11):990-996. doi:10.1001/jamainternmed.2013.478
4. Hshieh TT, Yang T, Gartaganis SL, Yue J, Inouye SK. Hospital Elder Life Program: systematic review and meta-analysis of effectiveness. Am J Geriatr Psychiatry. 2018;26(10):1015-1033. doi:10.1016/j.jagp.2018.06.007
5. Naughton C, Cummins H, de Foubert M, et al. Implementation of the Frailty Care Bundle (FCB) to promote mobilisation, nutrition and cognitive engagement in older people in acute care settings: protocol for an implementation science study. [version 1; peer review: 1 approved]. HRB Open Res. 2022;5:3. doi:10.12688/hrbopenres.134731
Study Overview
Objective: To examine the effect of the intervention “Eat Walk Engage,” a program that is designed to more consistently deliver age-friendly principles of care to older individuals in acute medical and surgical wards.
Design: This cluster randomized trial to examine the effect of an intervention in acute medical and surgical wards on older adults was conducted in 8 acute medical and surgical wards in 4 public hospitals in Australia from 2016 to 2017. To be eligible to participate in this trial, wards had to have the following: a patient population with 50% of patients aged 65 years and older; perceived alignment with hospital priorities; and nurse manager agreement to participation. Randomization was stratified by hospital, resulting in 4 wards with the intervention (a general medicine ward, an orthopedic ward, a general surgery ward, and a respiratory medicine ward) and 4 control wards (2 general medicine wards, a respiratory medicine ward, and a general surgery ward). Participants were consecutive inpatients aged 65 years or older who were admitted to the ward for at least 3 consecutive days during the study time period. Exclusion criteria included terminal or critical illness, severe cognitive impairment without a surrogate decision-maker, non-English speaking, or previously enrolled in the trial. Of a total of 453 patients who were eligible from the intervention wards, 188 were excluded and 6 died, yielding 259 participants in the intervention group. There were 413 patients eligible from the control wards, with 139 excluded and 3 deaths, yielding 271 participants in the control group.
Intervention: The intervention, called “Eat Walk Engage,” was developed to target older adults at risk for hospital-associated complications of delirium, functional decline, pressure injuries, falls, and incontinence, and aimed to improve care practices, environment, and culture to support age-friendly principles. This ward-based program delivered a structured improvement intervention through a site facilitator who is a nurse or allied health professional. The site facilitator identified opportunities for improvement using structured assessments of context, patient-experience interviews, and audits of care processes, and engaged an interdisciplinary working group from the intervention wards to participate in an hour-per-month meeting to develop plans for iterative improvements. Each site developed their own intervention plan; examples of interventions include shifting priorities to enable staff to increase the proportion of patients sitting in a chair for meals; designating the patient lounge as a walking destination to increase the proportion of time patients spend mobile; and using orientation boards and small groups to engage older patients in meaningful activities.
Main outcome measures: Study outcome measures included hospital-associated complications for older people, which is a composite of hospital-associated delirium, hospital-associated disability, hospital-associated incontinence, and fall or pressure injury during hospitalization. Delirium was assessed using the 3-minute diagnostic interview for Confusion Assessment Method (3D-CAM); hospital-associated disability was defined as new disability at discharge compared to 2 weeks prior to hospitalization. The primary outcome was defined as incidence of any complications and hospital length of stay. Secondary outcomes included incidence of individual complications, hospital discharge to facility, mortality at 6 months, and readmission for any cause at 6 months.
Main results: Patient characteristics for the intervention and control groups, respectively, were: 47% women with a mean age of 75.9 years (SD, 7.3), and 53% women with a mean age of 78.0 years (SD, 8.2). For the primary outcome, 46.4% of participants in the intervention group experienced any hospital complications compared with 51.8% in the control group (odds ratio [OR], 1.07; 95% CI, 0.71-1.61). The incidence of delirium was lower in the intervention group as compared with the control group (15.9% vs 31.4%; OR, 0.53; 95% CI, 0.31-0.90), while there were no other differences in the incidence rates of other complications. There was also no difference in hospital length of stay; median length of stay in the intervention group was 6 days (interquartile range [IQR], 4-9 days) compared with 7 days in the control group (IQR, 5-10), with an estimated mean difference in length of stay of 0.16 days (95% CI, –0.43 to 0.78 days). There was also no significant difference in mortality or all-cause readmission at 6 months.
Conclusion: The intervention “Eat Walk Engage” did not reduce hospital-associated complications overall or hospital length of stay, but it did reduce the incidence of hospital-associated delirium.
Commentary
Older adults, often with reduced physiologic reserve, when admitted to the hospital with an acute illness may be vulnerable to potential hazards of hospitalization, such as complications from prolonged periods of immobility, pressure injury, and delirium.1 Models of care in the inpatient setting to reduce these hazards, including the Acute Care for the Elderly model and the Mobile Acute Care for the Elderly Team model, have been examined in clinical trials.2,3 Specifically, models of care to prevent and treat delirium have been developed and tested over the past decade.4 The effect of these models in improving function, reducing complications, and reducing delirium incidence has been well documented. The present study adds to the literature by testing a model that utilizes implementation science methods to take into account real-world settings. In contrast with prior models-of-care studies, the implementation of the intervention at each ward was not prescriptive, but rather was developed in each ward in an iterative manner with stakeholder input. The advantage of this approach is that engagement of stakeholders at each intervention ward obtains buy-in from staff, mobilizing staff in a way that a prescriptive model of care may not; this ultimately may lead to longer-lasting change. The iterative approach also allows for the intervention to be adapted to conditions and settings over time. Other studies have taken this approach of using implementation science to drive change.5 Although the intervention in the present study failed to improve the primary outcome, it did reduce the incidence of delirium, which is a significant outcome and one that may confer considerable benefits to older adults under the model’s care.
A limitation of the intervention’s nonprescriptive approach is that, because of the variation of the interventions across sites, it is difficult to discern what elements drove the clinical outcomes. In addition, it would be challenging to consider what aspects of the intervention did not work should refinement or changes be needed. How one may measure fidelity to the intervention or how well a site implements the intervention and its relationship with clinical outcomes will need to be examined further.
Application for Clinical Practice
Clinicians look to effective models of care to improve clinical outcomes for older adults in the hospital. The intervention described in this study offers a real-world approach that may need less upfront investment than other recently studied models, such as the Acute Care for the Elderly model, which requires structural and staffing enhancements. Clinicians and health system leaders may consider implementing this model to improve the care delivered to older adults in the hospital as it may help reduce the incidence of delirium among the older adults they serve.
–William W. Hung, MD, MPH
Disclosures: None.
Study Overview
Objective: To examine the effect of the intervention “Eat Walk Engage,” a program that is designed to more consistently deliver age-friendly principles of care to older individuals in acute medical and surgical wards.
Design: This cluster randomized trial to examine the effect of an intervention in acute medical and surgical wards on older adults was conducted in 8 acute medical and surgical wards in 4 public hospitals in Australia from 2016 to 2017. To be eligible to participate in this trial, wards had to have the following: a patient population with 50% of patients aged 65 years and older; perceived alignment with hospital priorities; and nurse manager agreement to participation. Randomization was stratified by hospital, resulting in 4 wards with the intervention (a general medicine ward, an orthopedic ward, a general surgery ward, and a respiratory medicine ward) and 4 control wards (2 general medicine wards, a respiratory medicine ward, and a general surgery ward). Participants were consecutive inpatients aged 65 years or older who were admitted to the ward for at least 3 consecutive days during the study time period. Exclusion criteria included terminal or critical illness, severe cognitive impairment without a surrogate decision-maker, non-English speaking, or previously enrolled in the trial. Of a total of 453 patients who were eligible from the intervention wards, 188 were excluded and 6 died, yielding 259 participants in the intervention group. There were 413 patients eligible from the control wards, with 139 excluded and 3 deaths, yielding 271 participants in the control group.
Intervention: The intervention, called “Eat Walk Engage,” was developed to target older adults at risk for hospital-associated complications of delirium, functional decline, pressure injuries, falls, and incontinence, and aimed to improve care practices, environment, and culture to support age-friendly principles. This ward-based program delivered a structured improvement intervention through a site facilitator who is a nurse or allied health professional. The site facilitator identified opportunities for improvement using structured assessments of context, patient-experience interviews, and audits of care processes, and engaged an interdisciplinary working group from the intervention wards to participate in an hour-per-month meeting to develop plans for iterative improvements. Each site developed their own intervention plan; examples of interventions include shifting priorities to enable staff to increase the proportion of patients sitting in a chair for meals; designating the patient lounge as a walking destination to increase the proportion of time patients spend mobile; and using orientation boards and small groups to engage older patients in meaningful activities.
Main outcome measures: Study outcome measures included hospital-associated complications for older people, which is a composite of hospital-associated delirium, hospital-associated disability, hospital-associated incontinence, and fall or pressure injury during hospitalization. Delirium was assessed using the 3-minute diagnostic interview for Confusion Assessment Method (3D-CAM); hospital-associated disability was defined as new disability at discharge compared to 2 weeks prior to hospitalization. The primary outcome was defined as incidence of any complications and hospital length of stay. Secondary outcomes included incidence of individual complications, hospital discharge to facility, mortality at 6 months, and readmission for any cause at 6 months.
Main results: Patient characteristics for the intervention and control groups, respectively, were: 47% women with a mean age of 75.9 years (SD, 7.3), and 53% women with a mean age of 78.0 years (SD, 8.2). For the primary outcome, 46.4% of participants in the intervention group experienced any hospital complications compared with 51.8% in the control group (odds ratio [OR], 1.07; 95% CI, 0.71-1.61). The incidence of delirium was lower in the intervention group as compared with the control group (15.9% vs 31.4%; OR, 0.53; 95% CI, 0.31-0.90), while there were no other differences in the incidence rates of other complications. There was also no difference in hospital length of stay; median length of stay in the intervention group was 6 days (interquartile range [IQR], 4-9 days) compared with 7 days in the control group (IQR, 5-10), with an estimated mean difference in length of stay of 0.16 days (95% CI, –0.43 to 0.78 days). There was also no significant difference in mortality or all-cause readmission at 6 months.
Conclusion: The intervention “Eat Walk Engage” did not reduce hospital-associated complications overall or hospital length of stay, but it did reduce the incidence of hospital-associated delirium.
Commentary
Older adults, often with reduced physiologic reserve, when admitted to the hospital with an acute illness may be vulnerable to potential hazards of hospitalization, such as complications from prolonged periods of immobility, pressure injury, and delirium.1 Models of care in the inpatient setting to reduce these hazards, including the Acute Care for the Elderly model and the Mobile Acute Care for the Elderly Team model, have been examined in clinical trials.2,3 Specifically, models of care to prevent and treat delirium have been developed and tested over the past decade.4 The effect of these models in improving function, reducing complications, and reducing delirium incidence has been well documented. The present study adds to the literature by testing a model that utilizes implementation science methods to take into account real-world settings. In contrast with prior models-of-care studies, the implementation of the intervention at each ward was not prescriptive, but rather was developed in each ward in an iterative manner with stakeholder input. The advantage of this approach is that engagement of stakeholders at each intervention ward obtains buy-in from staff, mobilizing staff in a way that a prescriptive model of care may not; this ultimately may lead to longer-lasting change. The iterative approach also allows for the intervention to be adapted to conditions and settings over time. Other studies have taken this approach of using implementation science to drive change.5 Although the intervention in the present study failed to improve the primary outcome, it did reduce the incidence of delirium, which is a significant outcome and one that may confer considerable benefits to older adults under the model’s care.
A limitation of the intervention’s nonprescriptive approach is that, because of the variation of the interventions across sites, it is difficult to discern what elements drove the clinical outcomes. In addition, it would be challenging to consider what aspects of the intervention did not work should refinement or changes be needed. How one may measure fidelity to the intervention or how well a site implements the intervention and its relationship with clinical outcomes will need to be examined further.
Application for Clinical Practice
Clinicians look to effective models of care to improve clinical outcomes for older adults in the hospital. The intervention described in this study offers a real-world approach that may need less upfront investment than other recently studied models, such as the Acute Care for the Elderly model, which requires structural and staffing enhancements. Clinicians and health system leaders may consider implementing this model to improve the care delivered to older adults in the hospital as it may help reduce the incidence of delirium among the older adults they serve.
–William W. Hung, MD, MPH
Disclosures: None.
1. Creditor MC. Hazards of hospitalization of the elderly. Ann Intern Med. 1993;118(3):219-223. doi:10.7326/0003-4819-118-3-199302010-00011
2. Fox MT, Persaud M, Maimets I, et al. Effectiveness of acute geriatric unit care using acute care for elders components: a systematic review and meta-analysis. J Am Geriatr Soc. 2012;60(12):2237-2245. doi:10.1111/jgs.12028
3. Hung WW, Ross JS, Farber J, Siu AL. Evaluation of the Mobile Acute Care of the Elderly (MACE) service. JAMA Intern Med. 2013;173(11):990-996. doi:10.1001/jamainternmed.2013.478
4. Hshieh TT, Yang T, Gartaganis SL, Yue J, Inouye SK. Hospital Elder Life Program: systematic review and meta-analysis of effectiveness. Am J Geriatr Psychiatry. 2018;26(10):1015-1033. doi:10.1016/j.jagp.2018.06.007
5. Naughton C, Cummins H, de Foubert M, et al. Implementation of the Frailty Care Bundle (FCB) to promote mobilisation, nutrition and cognitive engagement in older people in acute care settings: protocol for an implementation science study. [version 1; peer review: 1 approved]. HRB Open Res. 2022;5:3. doi:10.12688/hrbopenres.134731
1. Creditor MC. Hazards of hospitalization of the elderly. Ann Intern Med. 1993;118(3):219-223. doi:10.7326/0003-4819-118-3-199302010-00011
2. Fox MT, Persaud M, Maimets I, et al. Effectiveness of acute geriatric unit care using acute care for elders components: a systematic review and meta-analysis. J Am Geriatr Soc. 2012;60(12):2237-2245. doi:10.1111/jgs.12028
3. Hung WW, Ross JS, Farber J, Siu AL. Evaluation of the Mobile Acute Care of the Elderly (MACE) service. JAMA Intern Med. 2013;173(11):990-996. doi:10.1001/jamainternmed.2013.478
4. Hshieh TT, Yang T, Gartaganis SL, Yue J, Inouye SK. Hospital Elder Life Program: systematic review and meta-analysis of effectiveness. Am J Geriatr Psychiatry. 2018;26(10):1015-1033. doi:10.1016/j.jagp.2018.06.007
5. Naughton C, Cummins H, de Foubert M, et al. Implementation of the Frailty Care Bundle (FCB) to promote mobilisation, nutrition and cognitive engagement in older people in acute care settings: protocol for an implementation science study. [version 1; peer review: 1 approved]. HRB Open Res. 2022;5:3. doi:10.12688/hrbopenres.134731
Psychedelic therapy and suicide: A myth busted?
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
Motherhood and mortality: Navigating miscarriages as a physician
One clinic morning in an office visit, I stood next to the door talking, hand on the doorknob ready to exit. My elderly patient was sitting in the chair next to the door, family member in another, as I attempted my exit. Suddenly, as if looking for something, my patient locked her gaze to my abdomen and began to slowly advance herself forward, eyes squinting for a better view. She had found something. Poke, poke, poke. Three pokes in quick succession into my apparently protruding abdomen stoked an internal horror that I dared not release onto my face. How in the hell could she know? My heart sank – the signs were still there.
“There’s something in there,” she said with a seasoned certainty.
“No there’s not,” I said trying hard to hide any emotion.
“Yes, there is,” she said flatly.
“Grannie, no there isn’t,” her family member interrupted, unknowingly saving me. I thanked them again and quickly left the room.
My patient had the ongoings of slowly progressing dementia. Little did she know she was right. Maybe she had known something in another time and space. Either way, I wasn’t prepared to tell the story. She wasn’t prepared to fully understand.
I tried to forge on to see the next patient. Tears began welling in both eyes. I tilted my head back slightly to prevent the water from falling. I wanted to feel offended, but she couldn’t have known the war my body was fighting at the time. I had not yet shared the pregnancy news with this particular patient, and yet her knowing was telling in a sense. I’m learning that the old folks always know.
I was at work, actively having yet another miscarriage. This was the second of two. This most recent time, we found out at 9 weeks that our baby had stopped growing about a week or so earlier. Cue the denial. Cue the rage. Cue the devastation.
Thinking back, with each pregnancy discovery, we did not wait the customary 3 months before telling anyone. Just about everyone knew. We were immediately excited to start sharing with friends, family, coworkers, and even patients early on. We knew the risks in my 40-something age group but were quintessentially optimistic.
I am a family medicine physician with expert-level knowledge and clinical experiences in women’s health counseling, contraception, conception, and pregnancy. In my training, I’ve delivered babies, been elbow-deep searching for wayward tissue from bleeding uteri, and sutured gaping vaginal lacerations. I’ve cried with new mothers at the end of long labors. I’ve been bear-hugged by doting new fathers. I have an abundance of medical knowledge, and yet the pain and struggle of miscarriage over the past 2.5 years has twice reduced me to absolute pieces. There was no course to teach me how to navigate loss within my own body, no textbook to study so that I could test out of the experience. Life hit us dead-on, and I was broken.
I can say that the experience of a miscarriage does not get easier with each subsequent loss. At least for me, the emotions were always raw and tender. Each one was a new gash to my emotional and physical health. My sanity bled out. I was physically exhausted. The struggles of being a health care worker in the midst of a global pandemic I’m sure did not help the situation. My first miscarriage was just before the start of the pandemic. I was in New York visiting family and after dinner at Tavern on the Green, of all places, when I began showing signs. Two days later, I was at the coffee station in our clinic cafeteria adding my cream and sugar when my ob.gyn.’s office called. The hCG levels were probably too low; a miscarriage was likely. I kept my composure, walked out of the cafeteria, got my car keys, went to my car, and proceeded to scream at the top of my lungs for a few minutes. Afterward, I went back to finish up my work and canceled my clinic for the rest of the day.
For my second miscarriage, I was laying in my doctor’s office getting an ultrasound. I had started bleeding the previous day but thought that the subchorionic hemorrhage noted on the last ultrasound might be the culprit. The bleeding was light. That’s the thing about being a pregnant physician: We know too much. The image on the screen looked abnormal, the remnants a ghost of its former self. I knew something was wrong but held out some hope. She searched and turned and pressed the transducer into my belly for a seemingly better view. She apologized for not finding the heartbeat. How is this happening again?
So how does one get through the loss of multiple pregnancies? I know my husband and I worked hard to get through each loss. We did all the right things a good therapist would recommend: Be present in the moment, go with your feelings, allow yourself to feel everything. There were no wrong emotions. Little by little we grieved and healed, grieved and healed. Having a successful pregnancy did help. Miracles are not promised but I believe we were sent one, and her name is Giavonna Barbara. Bookended by miscarriages, she has made me realize just how precious and delicate life really is. She is our absolute world and joy.
I’ve learned twice now that men mourn differently than women. Not any less, just in a different way. There is a pain in the silence that often goes unvocalized, but it is of no less value. My husband and I allowed each other to heal in our own unique ways, and that has made all of the difference. I think I knew I was doing okay when one day I found something funny and I let out the heartiest laugh my belly could muster. A different purpose was renewed. Tears were harder to come by. Hope for the future again sprung eternal. Life went on and so did we.
Looking back, I realize that having a miscarriage and working as a physician in the middle of a global pandemic pushed me to my emotional and physical limits. There is a second-guessing of sorts that occurs. Did the miscarriage happen because I was under so much stress at work? It had happened in the past, was this going to continue to happen?
I can say that I was great at compartmentalizing emotions. I’d try and box them away until I got off of work and then turn them on like a switch once I hit the driver’s seat. It’s easy as a busy physician with so many patients to see, messages to return, notes to write, students and residents to teach, and programs to run to completely tune out the thought of mourning. Temporarily anyway. Work was actually a welcome distraction at times. A purpose. The journey to healing is individualized and can’t be rushed. I like to think that I heal a little bit more every day thinking about the losses and gains that I’ve had. I’m grateful for the experience and growth.
In 2022, I’m looking forward to continuing my healing journey among the twists and turns of the pandemic. I now bring a different level of understanding and empathy to my patients who are undergoing or who have undergone a miscarriage. There will always be a piece of me that viscerally mourns with them. We have a hidden shared experience. I believe I am a better physician because of those lessons learned from my own personal tragedy. Now, I look forward to sharing big belly laughs with my family and friends and savoring the small, quiet moments with my husband and daughter.
A version of this article first appeared on Medscape.com.
One clinic morning in an office visit, I stood next to the door talking, hand on the doorknob ready to exit. My elderly patient was sitting in the chair next to the door, family member in another, as I attempted my exit. Suddenly, as if looking for something, my patient locked her gaze to my abdomen and began to slowly advance herself forward, eyes squinting for a better view. She had found something. Poke, poke, poke. Three pokes in quick succession into my apparently protruding abdomen stoked an internal horror that I dared not release onto my face. How in the hell could she know? My heart sank – the signs were still there.
“There’s something in there,” she said with a seasoned certainty.
“No there’s not,” I said trying hard to hide any emotion.
“Yes, there is,” she said flatly.
“Grannie, no there isn’t,” her family member interrupted, unknowingly saving me. I thanked them again and quickly left the room.
My patient had the ongoings of slowly progressing dementia. Little did she know she was right. Maybe she had known something in another time and space. Either way, I wasn’t prepared to tell the story. She wasn’t prepared to fully understand.
I tried to forge on to see the next patient. Tears began welling in both eyes. I tilted my head back slightly to prevent the water from falling. I wanted to feel offended, but she couldn’t have known the war my body was fighting at the time. I had not yet shared the pregnancy news with this particular patient, and yet her knowing was telling in a sense. I’m learning that the old folks always know.
I was at work, actively having yet another miscarriage. This was the second of two. This most recent time, we found out at 9 weeks that our baby had stopped growing about a week or so earlier. Cue the denial. Cue the rage. Cue the devastation.
Thinking back, with each pregnancy discovery, we did not wait the customary 3 months before telling anyone. Just about everyone knew. We were immediately excited to start sharing with friends, family, coworkers, and even patients early on. We knew the risks in my 40-something age group but were quintessentially optimistic.
I am a family medicine physician with expert-level knowledge and clinical experiences in women’s health counseling, contraception, conception, and pregnancy. In my training, I’ve delivered babies, been elbow-deep searching for wayward tissue from bleeding uteri, and sutured gaping vaginal lacerations. I’ve cried with new mothers at the end of long labors. I’ve been bear-hugged by doting new fathers. I have an abundance of medical knowledge, and yet the pain and struggle of miscarriage over the past 2.5 years has twice reduced me to absolute pieces. There was no course to teach me how to navigate loss within my own body, no textbook to study so that I could test out of the experience. Life hit us dead-on, and I was broken.
I can say that the experience of a miscarriage does not get easier with each subsequent loss. At least for me, the emotions were always raw and tender. Each one was a new gash to my emotional and physical health. My sanity bled out. I was physically exhausted. The struggles of being a health care worker in the midst of a global pandemic I’m sure did not help the situation. My first miscarriage was just before the start of the pandemic. I was in New York visiting family and after dinner at Tavern on the Green, of all places, when I began showing signs. Two days later, I was at the coffee station in our clinic cafeteria adding my cream and sugar when my ob.gyn.’s office called. The hCG levels were probably too low; a miscarriage was likely. I kept my composure, walked out of the cafeteria, got my car keys, went to my car, and proceeded to scream at the top of my lungs for a few minutes. Afterward, I went back to finish up my work and canceled my clinic for the rest of the day.
For my second miscarriage, I was laying in my doctor’s office getting an ultrasound. I had started bleeding the previous day but thought that the subchorionic hemorrhage noted on the last ultrasound might be the culprit. The bleeding was light. That’s the thing about being a pregnant physician: We know too much. The image on the screen looked abnormal, the remnants a ghost of its former self. I knew something was wrong but held out some hope. She searched and turned and pressed the transducer into my belly for a seemingly better view. She apologized for not finding the heartbeat. How is this happening again?
So how does one get through the loss of multiple pregnancies? I know my husband and I worked hard to get through each loss. We did all the right things a good therapist would recommend: Be present in the moment, go with your feelings, allow yourself to feel everything. There were no wrong emotions. Little by little we grieved and healed, grieved and healed. Having a successful pregnancy did help. Miracles are not promised but I believe we were sent one, and her name is Giavonna Barbara. Bookended by miscarriages, she has made me realize just how precious and delicate life really is. She is our absolute world and joy.
I’ve learned twice now that men mourn differently than women. Not any less, just in a different way. There is a pain in the silence that often goes unvocalized, but it is of no less value. My husband and I allowed each other to heal in our own unique ways, and that has made all of the difference. I think I knew I was doing okay when one day I found something funny and I let out the heartiest laugh my belly could muster. A different purpose was renewed. Tears were harder to come by. Hope for the future again sprung eternal. Life went on and so did we.
Looking back, I realize that having a miscarriage and working as a physician in the middle of a global pandemic pushed me to my emotional and physical limits. There is a second-guessing of sorts that occurs. Did the miscarriage happen because I was under so much stress at work? It had happened in the past, was this going to continue to happen?
I can say that I was great at compartmentalizing emotions. I’d try and box them away until I got off of work and then turn them on like a switch once I hit the driver’s seat. It’s easy as a busy physician with so many patients to see, messages to return, notes to write, students and residents to teach, and programs to run to completely tune out the thought of mourning. Temporarily anyway. Work was actually a welcome distraction at times. A purpose. The journey to healing is individualized and can’t be rushed. I like to think that I heal a little bit more every day thinking about the losses and gains that I’ve had. I’m grateful for the experience and growth.
In 2022, I’m looking forward to continuing my healing journey among the twists and turns of the pandemic. I now bring a different level of understanding and empathy to my patients who are undergoing or who have undergone a miscarriage. There will always be a piece of me that viscerally mourns with them. We have a hidden shared experience. I believe I am a better physician because of those lessons learned from my own personal tragedy. Now, I look forward to sharing big belly laughs with my family and friends and savoring the small, quiet moments with my husband and daughter.
A version of this article first appeared on Medscape.com.
One clinic morning in an office visit, I stood next to the door talking, hand on the doorknob ready to exit. My elderly patient was sitting in the chair next to the door, family member in another, as I attempted my exit. Suddenly, as if looking for something, my patient locked her gaze to my abdomen and began to slowly advance herself forward, eyes squinting for a better view. She had found something. Poke, poke, poke. Three pokes in quick succession into my apparently protruding abdomen stoked an internal horror that I dared not release onto my face. How in the hell could she know? My heart sank – the signs were still there.
“There’s something in there,” she said with a seasoned certainty.
“No there’s not,” I said trying hard to hide any emotion.
“Yes, there is,” she said flatly.
“Grannie, no there isn’t,” her family member interrupted, unknowingly saving me. I thanked them again and quickly left the room.
My patient had the ongoings of slowly progressing dementia. Little did she know she was right. Maybe she had known something in another time and space. Either way, I wasn’t prepared to tell the story. She wasn’t prepared to fully understand.
I tried to forge on to see the next patient. Tears began welling in both eyes. I tilted my head back slightly to prevent the water from falling. I wanted to feel offended, but she couldn’t have known the war my body was fighting at the time. I had not yet shared the pregnancy news with this particular patient, and yet her knowing was telling in a sense. I’m learning that the old folks always know.
I was at work, actively having yet another miscarriage. This was the second of two. This most recent time, we found out at 9 weeks that our baby had stopped growing about a week or so earlier. Cue the denial. Cue the rage. Cue the devastation.
Thinking back, with each pregnancy discovery, we did not wait the customary 3 months before telling anyone. Just about everyone knew. We were immediately excited to start sharing with friends, family, coworkers, and even patients early on. We knew the risks in my 40-something age group but were quintessentially optimistic.
I am a family medicine physician with expert-level knowledge and clinical experiences in women’s health counseling, contraception, conception, and pregnancy. In my training, I’ve delivered babies, been elbow-deep searching for wayward tissue from bleeding uteri, and sutured gaping vaginal lacerations. I’ve cried with new mothers at the end of long labors. I’ve been bear-hugged by doting new fathers. I have an abundance of medical knowledge, and yet the pain and struggle of miscarriage over the past 2.5 years has twice reduced me to absolute pieces. There was no course to teach me how to navigate loss within my own body, no textbook to study so that I could test out of the experience. Life hit us dead-on, and I was broken.
I can say that the experience of a miscarriage does not get easier with each subsequent loss. At least for me, the emotions were always raw and tender. Each one was a new gash to my emotional and physical health. My sanity bled out. I was physically exhausted. The struggles of being a health care worker in the midst of a global pandemic I’m sure did not help the situation. My first miscarriage was just before the start of the pandemic. I was in New York visiting family and after dinner at Tavern on the Green, of all places, when I began showing signs. Two days later, I was at the coffee station in our clinic cafeteria adding my cream and sugar when my ob.gyn.’s office called. The hCG levels were probably too low; a miscarriage was likely. I kept my composure, walked out of the cafeteria, got my car keys, went to my car, and proceeded to scream at the top of my lungs for a few minutes. Afterward, I went back to finish up my work and canceled my clinic for the rest of the day.
For my second miscarriage, I was laying in my doctor’s office getting an ultrasound. I had started bleeding the previous day but thought that the subchorionic hemorrhage noted on the last ultrasound might be the culprit. The bleeding was light. That’s the thing about being a pregnant physician: We know too much. The image on the screen looked abnormal, the remnants a ghost of its former self. I knew something was wrong but held out some hope. She searched and turned and pressed the transducer into my belly for a seemingly better view. She apologized for not finding the heartbeat. How is this happening again?
So how does one get through the loss of multiple pregnancies? I know my husband and I worked hard to get through each loss. We did all the right things a good therapist would recommend: Be present in the moment, go with your feelings, allow yourself to feel everything. There were no wrong emotions. Little by little we grieved and healed, grieved and healed. Having a successful pregnancy did help. Miracles are not promised but I believe we were sent one, and her name is Giavonna Barbara. Bookended by miscarriages, she has made me realize just how precious and delicate life really is. She is our absolute world and joy.
I’ve learned twice now that men mourn differently than women. Not any less, just in a different way. There is a pain in the silence that often goes unvocalized, but it is of no less value. My husband and I allowed each other to heal in our own unique ways, and that has made all of the difference. I think I knew I was doing okay when one day I found something funny and I let out the heartiest laugh my belly could muster. A different purpose was renewed. Tears were harder to come by. Hope for the future again sprung eternal. Life went on and so did we.
Looking back, I realize that having a miscarriage and working as a physician in the middle of a global pandemic pushed me to my emotional and physical limits. There is a second-guessing of sorts that occurs. Did the miscarriage happen because I was under so much stress at work? It had happened in the past, was this going to continue to happen?
I can say that I was great at compartmentalizing emotions. I’d try and box them away until I got off of work and then turn them on like a switch once I hit the driver’s seat. It’s easy as a busy physician with so many patients to see, messages to return, notes to write, students and residents to teach, and programs to run to completely tune out the thought of mourning. Temporarily anyway. Work was actually a welcome distraction at times. A purpose. The journey to healing is individualized and can’t be rushed. I like to think that I heal a little bit more every day thinking about the losses and gains that I’ve had. I’m grateful for the experience and growth.
In 2022, I’m looking forward to continuing my healing journey among the twists and turns of the pandemic. I now bring a different level of understanding and empathy to my patients who are undergoing or who have undergone a miscarriage. There will always be a piece of me that viscerally mourns with them. We have a hidden shared experience. I believe I am a better physician because of those lessons learned from my own personal tragedy. Now, I look forward to sharing big belly laughs with my family and friends and savoring the small, quiet moments with my husband and daughter.
A version of this article first appeared on Medscape.com.