User login
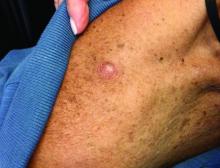
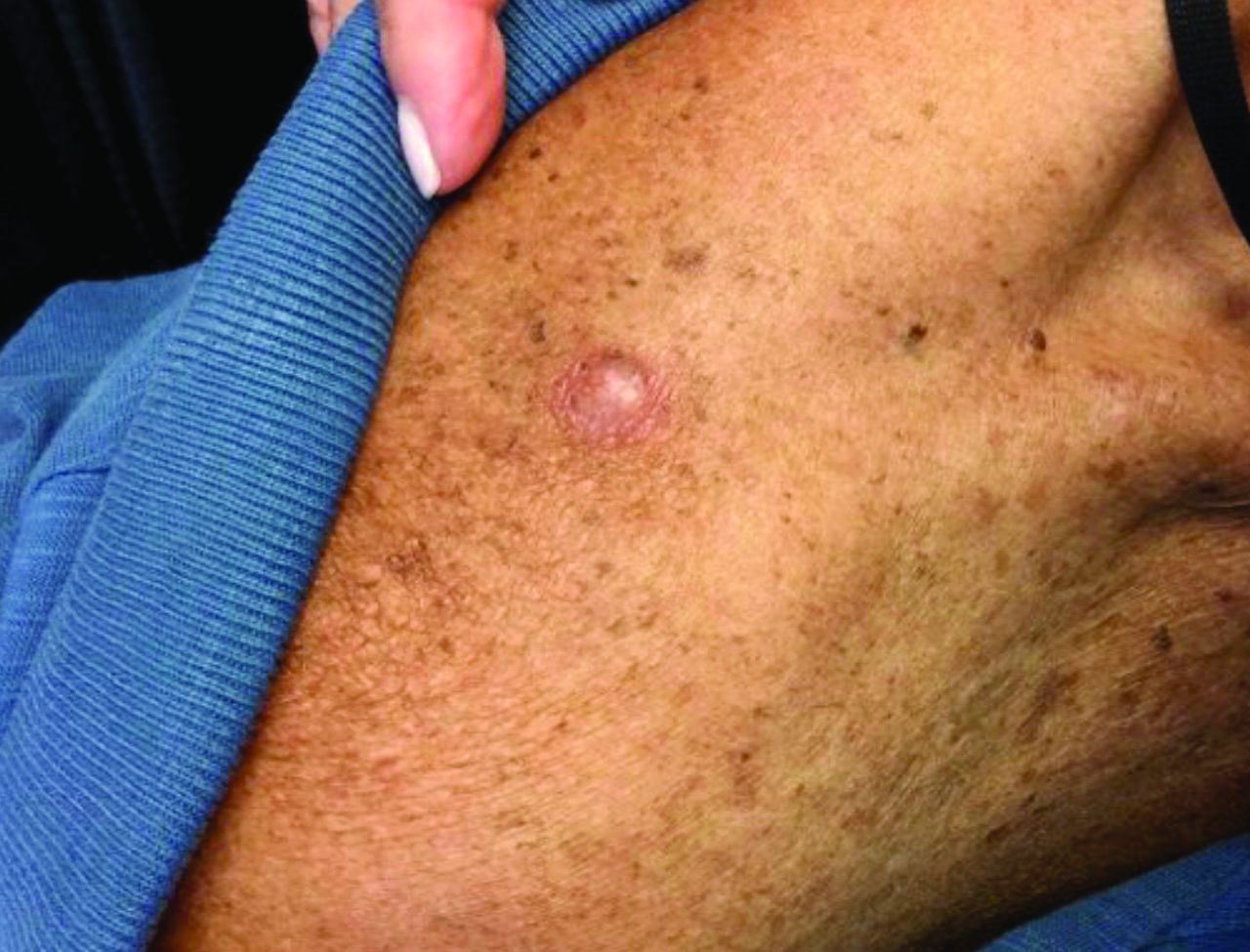
An 80-year-old patient presents with an asymptomatic firm pink plaque on his shoulder
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
Melanoma is a type of skin cancer that arises from melanocytes. According to the American Cancer Society, about 106,110 new melanomas will be diagnosed in the United States in 2021.The risk for developing melanoma increases with age. There are multiple clinical forms of cutaneous melanoma. The four main types are superficial spreading melanoma, nodular melanoma, melanoma in situ (lentigo maligna), and acral lentiginous melanoma. Rare variants include amelanotic melanoma, nevoid melanoma, spitzoid melanoma, and desmoplastic melanoma (DM). Melanoma can also rarely affect parts of the eye and mucosa.
, according to the Memorial Sloan Kettering Cancer Center. It typically presents as a subtle pigmented, pink, red, or skin colored patch, papule or plaque on sun-exposed skin (head and neck most frequently). Chronic UV exposure has been linked to DM. It may be mistaken for a scar or dermatofibroma. DM tends to grow locally and has less risk for nodal metastasis.1
Histologic diagnosis may be challenging. Two histologic variants in desmoplastic melanoma have been described: pure and mixed, depending on the degree of desmoplasia and cellularity present in the tumor.1 Pure DM tends to have a less aggressive course. Melanocytes can appear spindled in a fibrotic stroma. Patchy lymphocyte aggregates may be seen. Perineural invasion is more common in desmoplastic melanoma. Histologically, the differential includes spindle cell carcinoma and sarcoma. Immunostaining is helpful in differentiation.
Our patient had no lymphadenopathy on physical examination. Biopsy revealed a desmoplastic melanoma, 3.6 mm in depth, no ulceration, no regression, mitotic rate 1/mm2. He was referred to surgical oncology. The patient underwent wide excision. Sentinel lymph node biopsy was deferred.
It is imperative for dermatologists to be cognizant of this challenging subtype of melanoma when evaluating patients.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Chen L et al. J Am Acad Dermatol. 2013 May;68(5):825-33.
Don’t delay: Cancer patients need both doses of COVID vaccine
The new findings, which are soon to be published as a preprint, cast doubt on the current U.K. policy of delaying the second dose of the vaccine.
Delaying the second dose can leave most patients with cancer wholly or partially unprotected, according to the researchers. Moreover, such a delay has implications for transmission of SARS-CoV-2 in the cancer patient’s environs as well as for the evolution of virus variants that could be of concern, the researchers concluded.
The data come from a British study that included 151 patients with cancer and 54 healthy control persons. All participants received the COVID-19 mRNA BNT162b2 vaccine (Pfizer-BioNTech).
This vaccine requires two doses. The first few participants in this study were given the second dose 21 days after they had received the first dose, but then national guidelines changed, and the remaining participants had to wait 12 weeks to receive their second dose.
The researchers reported that, among health controls, the immune efficacy of the first dose was very high (97% efficacious). By contrast, among patients with solid tumors, the immune efficacy of a single dose was strikingly low (39%), and it was even lower in patients with hematologic malignancies (13%).
The second dose of vaccine greatly and rapidly increased the immune efficacy in patients with solid tumors (95% within 2 weeks of receiving the second dose), the researchers added.
Too few patients with hematologic cancers had received the second dose before the study ended for clear conclusions to be drawn. Nevertheless, the available data suggest that 50% of patients with hematologic cancers who had received the booster at day 21 were seropositive at 5 weeks vs. only 8% of those who had not received the booster.
“Our data provide the first real-world evidence of immune efficacy following one dose of the Pfizer vaccine in immunocompromised patient populations [and] clearly show that the poor one-dose efficacy in cancer patients can be rescued with an early booster at day 21,” commented senior author Sheeba Irshad, MD, senior clinical lecturer, King’s College London.
“Based on our findings, we would recommend an urgent review of the vaccine strategy for clinically extremely vulnerable groups. Until then, it is important that cancer patients continue to observe all public health measures in place, such as social distancing and shielding when attending hospitals, even after vaccination,” Dr. Irshad added.
The paper, with first author Leticia Monin-Aldama, PhD, is scheduled to appear on the preprint server medRxiv. It has not undergone peer review. The paper was distributed to journalists, with comments from experts not involved in the study, by the UK Science Media Centre.
These data are “of immediate importance” to patients with cancer, commented Shoba Amarnath, PhD, Newcastle University research fellow, Laboratory of T-cell Regulation, Newcastle University Center for Cancer, Newcastle upon Tyne, England.
“These findings are consistent with our understanding. … We know that the immune system within cancer patients is compromised as compared to healthy controls,” Dr. Amarnath said. “The data in the study support the notion that, in solid cancer patients, a considerable delay in second dose will extend the period when cancer patients are at risk of SARS-CoV-2 infection.”
Although more data are required, “this study does raise the issue of whether patients with cancer, other diseases, or those undergoing therapies that affect the body’s immune response should be fast-tracked for their second vaccine dose,” commented Lawrence Young, PhD, professor of molecular oncology and director of the Warwick Cancer Research Center, University of Warwick, Coventry, England.
Stephen Evans, MSc, professor of pharmacoepidemiology, London School of Hygiene and Tropical Medicine, underlined that the study is “essentially” observational and “inevitable limitations must be taken into account.
“Nevertheless, these results do suggest that the vaccines may well not protect those patients with cancer as well as those without cancer,” Mr. Evans said. He added that it is “important that this population continues to observe all COVID-19–associated measures, such as social distancing and shielding when attending hospitals, even after vaccination.”
Study details
Previous studies have shown that some patients with cancer have prolonged responses to SARS-CoV-2 infection, with ongoing immune dysregulation, inefficient seroconversion, and prolonged viral shedding.
There are few data, however, on how these patients respond to COVID-19 vaccination. The authors point out that, among the 18,860 individuals who received the Pfizer vaccine during its development trials, “none with an active oncological diagnosis was included.”
To investigate this issue, they launched the SARS-CoV-2 for Cancer Patients (SOAP-02) study.
The 151 patients with cancer who participated in this study were mostly elderly, the authors noted (75% were older than 65 years; the median age was 73 years). The majority (63%) had solid-tumor malignancies. Of those, 8% had late-stage disease and had been living with their cancer for more than 24 months.
The healthy control persons were vaccine-eligible primary health care workers who were not age matched to the cancer patients.
All participants received the first dose of vaccine; 31 (of 151) patients with cancer and 16 (of 54) healthy control persons received the second dose on day 21.
The remaining participants were scheduled to receive their second dose 12 weeks later (after the study ended), in line with the changes in the national guidelines.
The team reported that, approximately 21 days after receiving the first vaccine dose, the immune efficacy of the vaccine was estimated to be 97% among healthy control persons vs. 39% for patients with solid tumors and only 13% for those with hematologic malignancies (P < .0001 for both).
T-cell responses, as assessed via interferon-gamma and/or interleukin-2 production, were observed in 82% of healthy control persons, 71% of patients with solid tumors, and 50% of those with hematologic cancers.
Vaccine boosting at day 21 resulted in immune efficacy of 100% for healthy control persons and 95% for patients with solid tumors. In contrast, only 43% of those who did not receive the second dose were seropositive 2 weeks later.
Further analysis suggested that participants who did not have a serologic response were “spread evenly” across different cancer types, but the reduced responses were more frequent among patients who had received the vaccine within 15 days of cancer treatment, especially chemotherapy, and had undergone intensive treatments.
The SOAP study is sponsored by King’s College London and Guy’s and St. Thomas Trust Foundation NHS Trust. It is funded from grants from the KCL Charity, Cancer Research UK, and program grants from Breast Cancer Now. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The new findings, which are soon to be published as a preprint, cast doubt on the current U.K. policy of delaying the second dose of the vaccine.
Delaying the second dose can leave most patients with cancer wholly or partially unprotected, according to the researchers. Moreover, such a delay has implications for transmission of SARS-CoV-2 in the cancer patient’s environs as well as for the evolution of virus variants that could be of concern, the researchers concluded.
The data come from a British study that included 151 patients with cancer and 54 healthy control persons. All participants received the COVID-19 mRNA BNT162b2 vaccine (Pfizer-BioNTech).
This vaccine requires two doses. The first few participants in this study were given the second dose 21 days after they had received the first dose, but then national guidelines changed, and the remaining participants had to wait 12 weeks to receive their second dose.
The researchers reported that, among health controls, the immune efficacy of the first dose was very high (97% efficacious). By contrast, among patients with solid tumors, the immune efficacy of a single dose was strikingly low (39%), and it was even lower in patients with hematologic malignancies (13%).
The second dose of vaccine greatly and rapidly increased the immune efficacy in patients with solid tumors (95% within 2 weeks of receiving the second dose), the researchers added.
Too few patients with hematologic cancers had received the second dose before the study ended for clear conclusions to be drawn. Nevertheless, the available data suggest that 50% of patients with hematologic cancers who had received the booster at day 21 were seropositive at 5 weeks vs. only 8% of those who had not received the booster.
“Our data provide the first real-world evidence of immune efficacy following one dose of the Pfizer vaccine in immunocompromised patient populations [and] clearly show that the poor one-dose efficacy in cancer patients can be rescued with an early booster at day 21,” commented senior author Sheeba Irshad, MD, senior clinical lecturer, King’s College London.
“Based on our findings, we would recommend an urgent review of the vaccine strategy for clinically extremely vulnerable groups. Until then, it is important that cancer patients continue to observe all public health measures in place, such as social distancing and shielding when attending hospitals, even after vaccination,” Dr. Irshad added.
The paper, with first author Leticia Monin-Aldama, PhD, is scheduled to appear on the preprint server medRxiv. It has not undergone peer review. The paper was distributed to journalists, with comments from experts not involved in the study, by the UK Science Media Centre.
These data are “of immediate importance” to patients with cancer, commented Shoba Amarnath, PhD, Newcastle University research fellow, Laboratory of T-cell Regulation, Newcastle University Center for Cancer, Newcastle upon Tyne, England.
“These findings are consistent with our understanding. … We know that the immune system within cancer patients is compromised as compared to healthy controls,” Dr. Amarnath said. “The data in the study support the notion that, in solid cancer patients, a considerable delay in second dose will extend the period when cancer patients are at risk of SARS-CoV-2 infection.”
Although more data are required, “this study does raise the issue of whether patients with cancer, other diseases, or those undergoing therapies that affect the body’s immune response should be fast-tracked for their second vaccine dose,” commented Lawrence Young, PhD, professor of molecular oncology and director of the Warwick Cancer Research Center, University of Warwick, Coventry, England.
Stephen Evans, MSc, professor of pharmacoepidemiology, London School of Hygiene and Tropical Medicine, underlined that the study is “essentially” observational and “inevitable limitations must be taken into account.
“Nevertheless, these results do suggest that the vaccines may well not protect those patients with cancer as well as those without cancer,” Mr. Evans said. He added that it is “important that this population continues to observe all COVID-19–associated measures, such as social distancing and shielding when attending hospitals, even after vaccination.”
Study details
Previous studies have shown that some patients with cancer have prolonged responses to SARS-CoV-2 infection, with ongoing immune dysregulation, inefficient seroconversion, and prolonged viral shedding.
There are few data, however, on how these patients respond to COVID-19 vaccination. The authors point out that, among the 18,860 individuals who received the Pfizer vaccine during its development trials, “none with an active oncological diagnosis was included.”
To investigate this issue, they launched the SARS-CoV-2 for Cancer Patients (SOAP-02) study.
The 151 patients with cancer who participated in this study were mostly elderly, the authors noted (75% were older than 65 years; the median age was 73 years). The majority (63%) had solid-tumor malignancies. Of those, 8% had late-stage disease and had been living with their cancer for more than 24 months.
The healthy control persons were vaccine-eligible primary health care workers who were not age matched to the cancer patients.
All participants received the first dose of vaccine; 31 (of 151) patients with cancer and 16 (of 54) healthy control persons received the second dose on day 21.
The remaining participants were scheduled to receive their second dose 12 weeks later (after the study ended), in line with the changes in the national guidelines.
The team reported that, approximately 21 days after receiving the first vaccine dose, the immune efficacy of the vaccine was estimated to be 97% among healthy control persons vs. 39% for patients with solid tumors and only 13% for those with hematologic malignancies (P < .0001 for both).
T-cell responses, as assessed via interferon-gamma and/or interleukin-2 production, were observed in 82% of healthy control persons, 71% of patients with solid tumors, and 50% of those with hematologic cancers.
Vaccine boosting at day 21 resulted in immune efficacy of 100% for healthy control persons and 95% for patients with solid tumors. In contrast, only 43% of those who did not receive the second dose were seropositive 2 weeks later.
Further analysis suggested that participants who did not have a serologic response were “spread evenly” across different cancer types, but the reduced responses were more frequent among patients who had received the vaccine within 15 days of cancer treatment, especially chemotherapy, and had undergone intensive treatments.
The SOAP study is sponsored by King’s College London and Guy’s and St. Thomas Trust Foundation NHS Trust. It is funded from grants from the KCL Charity, Cancer Research UK, and program grants from Breast Cancer Now. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The new findings, which are soon to be published as a preprint, cast doubt on the current U.K. policy of delaying the second dose of the vaccine.
Delaying the second dose can leave most patients with cancer wholly or partially unprotected, according to the researchers. Moreover, such a delay has implications for transmission of SARS-CoV-2 in the cancer patient’s environs as well as for the evolution of virus variants that could be of concern, the researchers concluded.
The data come from a British study that included 151 patients with cancer and 54 healthy control persons. All participants received the COVID-19 mRNA BNT162b2 vaccine (Pfizer-BioNTech).
This vaccine requires two doses. The first few participants in this study were given the second dose 21 days after they had received the first dose, but then national guidelines changed, and the remaining participants had to wait 12 weeks to receive their second dose.
The researchers reported that, among health controls, the immune efficacy of the first dose was very high (97% efficacious). By contrast, among patients with solid tumors, the immune efficacy of a single dose was strikingly low (39%), and it was even lower in patients with hematologic malignancies (13%).
The second dose of vaccine greatly and rapidly increased the immune efficacy in patients with solid tumors (95% within 2 weeks of receiving the second dose), the researchers added.
Too few patients with hematologic cancers had received the second dose before the study ended for clear conclusions to be drawn. Nevertheless, the available data suggest that 50% of patients with hematologic cancers who had received the booster at day 21 were seropositive at 5 weeks vs. only 8% of those who had not received the booster.
“Our data provide the first real-world evidence of immune efficacy following one dose of the Pfizer vaccine in immunocompromised patient populations [and] clearly show that the poor one-dose efficacy in cancer patients can be rescued with an early booster at day 21,” commented senior author Sheeba Irshad, MD, senior clinical lecturer, King’s College London.
“Based on our findings, we would recommend an urgent review of the vaccine strategy for clinically extremely vulnerable groups. Until then, it is important that cancer patients continue to observe all public health measures in place, such as social distancing and shielding when attending hospitals, even after vaccination,” Dr. Irshad added.
The paper, with first author Leticia Monin-Aldama, PhD, is scheduled to appear on the preprint server medRxiv. It has not undergone peer review. The paper was distributed to journalists, with comments from experts not involved in the study, by the UK Science Media Centre.
These data are “of immediate importance” to patients with cancer, commented Shoba Amarnath, PhD, Newcastle University research fellow, Laboratory of T-cell Regulation, Newcastle University Center for Cancer, Newcastle upon Tyne, England.
“These findings are consistent with our understanding. … We know that the immune system within cancer patients is compromised as compared to healthy controls,” Dr. Amarnath said. “The data in the study support the notion that, in solid cancer patients, a considerable delay in second dose will extend the period when cancer patients are at risk of SARS-CoV-2 infection.”
Although more data are required, “this study does raise the issue of whether patients with cancer, other diseases, or those undergoing therapies that affect the body’s immune response should be fast-tracked for their second vaccine dose,” commented Lawrence Young, PhD, professor of molecular oncology and director of the Warwick Cancer Research Center, University of Warwick, Coventry, England.
Stephen Evans, MSc, professor of pharmacoepidemiology, London School of Hygiene and Tropical Medicine, underlined that the study is “essentially” observational and “inevitable limitations must be taken into account.
“Nevertheless, these results do suggest that the vaccines may well not protect those patients with cancer as well as those without cancer,” Mr. Evans said. He added that it is “important that this population continues to observe all COVID-19–associated measures, such as social distancing and shielding when attending hospitals, even after vaccination.”
Study details
Previous studies have shown that some patients with cancer have prolonged responses to SARS-CoV-2 infection, with ongoing immune dysregulation, inefficient seroconversion, and prolonged viral shedding.
There are few data, however, on how these patients respond to COVID-19 vaccination. The authors point out that, among the 18,860 individuals who received the Pfizer vaccine during its development trials, “none with an active oncological diagnosis was included.”
To investigate this issue, they launched the SARS-CoV-2 for Cancer Patients (SOAP-02) study.
The 151 patients with cancer who participated in this study were mostly elderly, the authors noted (75% were older than 65 years; the median age was 73 years). The majority (63%) had solid-tumor malignancies. Of those, 8% had late-stage disease and had been living with their cancer for more than 24 months.
The healthy control persons were vaccine-eligible primary health care workers who were not age matched to the cancer patients.
All participants received the first dose of vaccine; 31 (of 151) patients with cancer and 16 (of 54) healthy control persons received the second dose on day 21.
The remaining participants were scheduled to receive their second dose 12 weeks later (after the study ended), in line with the changes in the national guidelines.
The team reported that, approximately 21 days after receiving the first vaccine dose, the immune efficacy of the vaccine was estimated to be 97% among healthy control persons vs. 39% for patients with solid tumors and only 13% for those with hematologic malignancies (P < .0001 for both).
T-cell responses, as assessed via interferon-gamma and/or interleukin-2 production, were observed in 82% of healthy control persons, 71% of patients with solid tumors, and 50% of those with hematologic cancers.
Vaccine boosting at day 21 resulted in immune efficacy of 100% for healthy control persons and 95% for patients with solid tumors. In contrast, only 43% of those who did not receive the second dose were seropositive 2 weeks later.
Further analysis suggested that participants who did not have a serologic response were “spread evenly” across different cancer types, but the reduced responses were more frequent among patients who had received the vaccine within 15 days of cancer treatment, especially chemotherapy, and had undergone intensive treatments.
The SOAP study is sponsored by King’s College London and Guy’s and St. Thomas Trust Foundation NHS Trust. It is funded from grants from the KCL Charity, Cancer Research UK, and program grants from Breast Cancer Now. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
mCODE: Improving data sharing to enhance cancer care
An initiative designed to improve sharing of patient data may provide “tremendous benefits” in cancer care and research, according to authors of a review article.
The goals of the initiative, called Minimal Common Oncology Data Elements (mCODE), were to identify the data elements in electronic health records that are “essential” for making treatment decisions and create “a standardized computable data format” that would improve the exchange of data across EHRs, according to the mCODE website.
Travis J. Osterman, DO, of Vanderbilt University Medical Center in Nashville, Tenn., and colleagues described the mCODE initiative in a review published in JCO Clinical Cancer Informatics.
At present, commercially available EHRs are poorly designed to support modern oncology workflow, requiring laborious data entry and lacking a common library of oncology-specific discrete data elements. As an example, most EHRs poorly support the needs of precision oncology and clinical genetics, since next-generation sequencing and genetic test results are almost universally reported in PDF files.
In addition, basic, operational oncology data (e.g., cancer staging, adverse event documentation, response to treatment, etc.) are captured in EHRs primarily as an unstructured narrative.
Computable, analytical data are found for only the small percentage of patients in clinical trials. Even then, some degree of manual data abstraction is regularly required.
Interoperability of EHRs between practices and health care institutions is often so poor that the transfer of basic cancer-related information as analyzable data is difficult or even impossible.
Making progress: The 21st Century Cures Act
The American Society of Clinical Oncology has a more than 15-year history of developing oncology data standards. Unfortunately, progress in implementing these standards has been glacially slow. Impediments have included:
- A lack of conformance with clinical workflows.
- Failure to test standards on specific-use cases during pilot testing.
- A focus on data exchange, rather than the practical impediments to data entry.
- Poor engagement with EHR vendors in distributing clinical information modules with an oncology-specific focus
- Instability of data interoperability technologies.
The 21st Century Cures Act, which became law in December 2016, mandated improvement in the interoperability of health information through the development of data standards and application programming interfaces.
In early 2020, final rules for implementation required technology vendors to employ application programming interfaces using a single interoperability resource. In addition, payers were required to use the United States Core Data for Interoperability Standard for data exchange. These requirements were intended to provide patients with access to their own health care data “without special effort.”
As a fortunate byproduct, since EHR vendors are required to implement application program interfaces using the Health Level Seven International (HL7) Fast Healthcare Interoperability Resource (FHIR) Specification, the final rules could enable systems like mCODE to be more easily integrated with existing EHRs.
Lessons from CancerLinQ
ASCO created the health technology platform CancerLinQ in 2014, envisioning that it could become an oncology-focused learning health system – a system in which internal data and experience are systematically integrated with external evidence, allowing knowledge to be put into practice.
CancerLinQ extracts data from EHRs and other sources via direct software connections. CancerLinQ then aggregates, harmonizes, and normalizes the data in a cloud-based environment.
The data are available to participating practices for quality improvement in patient care and secondary research. In 2020, records of cancer patients in the CancerLinQ database surpassed 2 million.
CancerLinQ has been successful. However, because of the nature of the EHR ecosystem and the scope and variability of data capture by clinicians, supporting a true learning health system has proven to be a formidable task. Postprocessing manual review using trained human curators is laborious and unsustainable.
The CancerLinQ experience illustrated that basic cancer-pertinent data should be standardized in the EHR and collected prospectively.
The mCODE model
The mCODE initiative seeks to facilitate progress in care quality, clinical research, and health care policy by developing and maintaining a standard, computable, interoperable data format.
Guiding principles that were adopted early in mCODE’s development included:
- A collaborative, noncommercial, use case–driven developmental model.
- Iterative processes.
- User-driven development, refinement, and maintenance.
- Low ongoing maintenance requirements.
A foundational moment in mCODE’s development involved achieving consensus among stakeholders that the project would fail if EHR vendors required additional data entry by users.
After pilot work, a real-world endpoints project, working-group deliberation, public comment, and refinement, the final data standard included six primary domains: patient, disease, laboratory data/vital signs, genomics, treatment, and outcome.
Each domain is further divided into several concepts with specific associated data elements. The data elements are modeled into value sets that specify the possible values for the data element.
To test mCODE, eight organizations representing oncology EHR vendors, standards developers, and research organizations participated in a cancer interoperability track. The comments helped refine mCODE version 1.0, which was released in March 2020 and is accessible via the mCODE website.
Additions will likely be reviewed by a technical review group after external piloting of new use cases.
Innovation, not regulation
Every interaction between a patient and care provider yields information that could lead to improved safety and better outcomes. To be successful, the information must be collected in a computable format so it can be aggregated with data from other patients, analyzed without manual curation, and shared through interoperable systems. Those data should also be secure enough to protect the privacy of individual patients.
mCODE is a consensus data standard for oncology that provides an infrastructure to share patient data between oncology practices and health care systems while promising little to no additional data entry on the part of clinicians. Adoption by sites will be critical, however.
Publishing the standard through the HL7 FHIR technology demonstrated to EHR vendors and regulatory agencies the stability of HL7, an essential requirement for its incorporation into software.
EHR vendors and others are engaged in the CodeX HL7 FHIR Accelerator to design projects to expand and/or modify mCODE. Their creativity and innovativeness via the external advisory mCODE council and/or CodeX will be encouraged to help mCODE reach its full potential.
As part of CodeX, the Community of Practice, an open forum for end users, was established to provide regular updates about mCODE-related initiatives and use cases to solicit in-progress input, according to Robert S. Miller, MD, medical director of CancerLinQ and an author of the mCODE review.
For mCODE to be embraced by all stakeholders, there should be no additional regulations. By engaging stakeholders in an enterprise that supports innovation and collaboration – without additional regulation – mCODE could maximize the potential of EHRs that, until now, have assisted us only marginally in accomplishing those goals.
mCODE is a joint venture of ASCO/CancerLinQ, the Alliance for Clinical Trials in Oncology Foundation, the MITRE Corporation, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Osterman disclosed a grant from the National Cancer Institute and relationships with Infostratix, eHealth, AstraZeneca, Outcomes Insights, Biodesix, MD Outlook, GenomOncology, Cota Healthcare, GE Healthcare, and Microsoft. Dr. Miller and the third review author disclosed no conflicts of interest.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
An initiative designed to improve sharing of patient data may provide “tremendous benefits” in cancer care and research, according to authors of a review article.
The goals of the initiative, called Minimal Common Oncology Data Elements (mCODE), were to identify the data elements in electronic health records that are “essential” for making treatment decisions and create “a standardized computable data format” that would improve the exchange of data across EHRs, according to the mCODE website.
Travis J. Osterman, DO, of Vanderbilt University Medical Center in Nashville, Tenn., and colleagues described the mCODE initiative in a review published in JCO Clinical Cancer Informatics.
At present, commercially available EHRs are poorly designed to support modern oncology workflow, requiring laborious data entry and lacking a common library of oncology-specific discrete data elements. As an example, most EHRs poorly support the needs of precision oncology and clinical genetics, since next-generation sequencing and genetic test results are almost universally reported in PDF files.
In addition, basic, operational oncology data (e.g., cancer staging, adverse event documentation, response to treatment, etc.) are captured in EHRs primarily as an unstructured narrative.
Computable, analytical data are found for only the small percentage of patients in clinical trials. Even then, some degree of manual data abstraction is regularly required.
Interoperability of EHRs between practices and health care institutions is often so poor that the transfer of basic cancer-related information as analyzable data is difficult or even impossible.
Making progress: The 21st Century Cures Act
The American Society of Clinical Oncology has a more than 15-year history of developing oncology data standards. Unfortunately, progress in implementing these standards has been glacially slow. Impediments have included:
- A lack of conformance with clinical workflows.
- Failure to test standards on specific-use cases during pilot testing.
- A focus on data exchange, rather than the practical impediments to data entry.
- Poor engagement with EHR vendors in distributing clinical information modules with an oncology-specific focus
- Instability of data interoperability technologies.
The 21st Century Cures Act, which became law in December 2016, mandated improvement in the interoperability of health information through the development of data standards and application programming interfaces.
In early 2020, final rules for implementation required technology vendors to employ application programming interfaces using a single interoperability resource. In addition, payers were required to use the United States Core Data for Interoperability Standard for data exchange. These requirements were intended to provide patients with access to their own health care data “without special effort.”
As a fortunate byproduct, since EHR vendors are required to implement application program interfaces using the Health Level Seven International (HL7) Fast Healthcare Interoperability Resource (FHIR) Specification, the final rules could enable systems like mCODE to be more easily integrated with existing EHRs.
Lessons from CancerLinQ
ASCO created the health technology platform CancerLinQ in 2014, envisioning that it could become an oncology-focused learning health system – a system in which internal data and experience are systematically integrated with external evidence, allowing knowledge to be put into practice.
CancerLinQ extracts data from EHRs and other sources via direct software connections. CancerLinQ then aggregates, harmonizes, and normalizes the data in a cloud-based environment.
The data are available to participating practices for quality improvement in patient care and secondary research. In 2020, records of cancer patients in the CancerLinQ database surpassed 2 million.
CancerLinQ has been successful. However, because of the nature of the EHR ecosystem and the scope and variability of data capture by clinicians, supporting a true learning health system has proven to be a formidable task. Postprocessing manual review using trained human curators is laborious and unsustainable.
The CancerLinQ experience illustrated that basic cancer-pertinent data should be standardized in the EHR and collected prospectively.
The mCODE model
The mCODE initiative seeks to facilitate progress in care quality, clinical research, and health care policy by developing and maintaining a standard, computable, interoperable data format.
Guiding principles that were adopted early in mCODE’s development included:
- A collaborative, noncommercial, use case–driven developmental model.
- Iterative processes.
- User-driven development, refinement, and maintenance.
- Low ongoing maintenance requirements.
A foundational moment in mCODE’s development involved achieving consensus among stakeholders that the project would fail if EHR vendors required additional data entry by users.
After pilot work, a real-world endpoints project, working-group deliberation, public comment, and refinement, the final data standard included six primary domains: patient, disease, laboratory data/vital signs, genomics, treatment, and outcome.
Each domain is further divided into several concepts with specific associated data elements. The data elements are modeled into value sets that specify the possible values for the data element.
To test mCODE, eight organizations representing oncology EHR vendors, standards developers, and research organizations participated in a cancer interoperability track. The comments helped refine mCODE version 1.0, which was released in March 2020 and is accessible via the mCODE website.
Additions will likely be reviewed by a technical review group after external piloting of new use cases.
Innovation, not regulation
Every interaction between a patient and care provider yields information that could lead to improved safety and better outcomes. To be successful, the information must be collected in a computable format so it can be aggregated with data from other patients, analyzed without manual curation, and shared through interoperable systems. Those data should also be secure enough to protect the privacy of individual patients.
mCODE is a consensus data standard for oncology that provides an infrastructure to share patient data between oncology practices and health care systems while promising little to no additional data entry on the part of clinicians. Adoption by sites will be critical, however.
Publishing the standard through the HL7 FHIR technology demonstrated to EHR vendors and regulatory agencies the stability of HL7, an essential requirement for its incorporation into software.
EHR vendors and others are engaged in the CodeX HL7 FHIR Accelerator to design projects to expand and/or modify mCODE. Their creativity and innovativeness via the external advisory mCODE council and/or CodeX will be encouraged to help mCODE reach its full potential.
As part of CodeX, the Community of Practice, an open forum for end users, was established to provide regular updates about mCODE-related initiatives and use cases to solicit in-progress input, according to Robert S. Miller, MD, medical director of CancerLinQ and an author of the mCODE review.
For mCODE to be embraced by all stakeholders, there should be no additional regulations. By engaging stakeholders in an enterprise that supports innovation and collaboration – without additional regulation – mCODE could maximize the potential of EHRs that, until now, have assisted us only marginally in accomplishing those goals.
mCODE is a joint venture of ASCO/CancerLinQ, the Alliance for Clinical Trials in Oncology Foundation, the MITRE Corporation, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Osterman disclosed a grant from the National Cancer Institute and relationships with Infostratix, eHealth, AstraZeneca, Outcomes Insights, Biodesix, MD Outlook, GenomOncology, Cota Healthcare, GE Healthcare, and Microsoft. Dr. Miller and the third review author disclosed no conflicts of interest.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
An initiative designed to improve sharing of patient data may provide “tremendous benefits” in cancer care and research, according to authors of a review article.
The goals of the initiative, called Minimal Common Oncology Data Elements (mCODE), were to identify the data elements in electronic health records that are “essential” for making treatment decisions and create “a standardized computable data format” that would improve the exchange of data across EHRs, according to the mCODE website.
Travis J. Osterman, DO, of Vanderbilt University Medical Center in Nashville, Tenn., and colleagues described the mCODE initiative in a review published in JCO Clinical Cancer Informatics.
At present, commercially available EHRs are poorly designed to support modern oncology workflow, requiring laborious data entry and lacking a common library of oncology-specific discrete data elements. As an example, most EHRs poorly support the needs of precision oncology and clinical genetics, since next-generation sequencing and genetic test results are almost universally reported in PDF files.
In addition, basic, operational oncology data (e.g., cancer staging, adverse event documentation, response to treatment, etc.) are captured in EHRs primarily as an unstructured narrative.
Computable, analytical data are found for only the small percentage of patients in clinical trials. Even then, some degree of manual data abstraction is regularly required.
Interoperability of EHRs between practices and health care institutions is often so poor that the transfer of basic cancer-related information as analyzable data is difficult or even impossible.
Making progress: The 21st Century Cures Act
The American Society of Clinical Oncology has a more than 15-year history of developing oncology data standards. Unfortunately, progress in implementing these standards has been glacially slow. Impediments have included:
- A lack of conformance with clinical workflows.
- Failure to test standards on specific-use cases during pilot testing.
- A focus on data exchange, rather than the practical impediments to data entry.
- Poor engagement with EHR vendors in distributing clinical information modules with an oncology-specific focus
- Instability of data interoperability technologies.
The 21st Century Cures Act, which became law in December 2016, mandated improvement in the interoperability of health information through the development of data standards and application programming interfaces.
In early 2020, final rules for implementation required technology vendors to employ application programming interfaces using a single interoperability resource. In addition, payers were required to use the United States Core Data for Interoperability Standard for data exchange. These requirements were intended to provide patients with access to their own health care data “without special effort.”
As a fortunate byproduct, since EHR vendors are required to implement application program interfaces using the Health Level Seven International (HL7) Fast Healthcare Interoperability Resource (FHIR) Specification, the final rules could enable systems like mCODE to be more easily integrated with existing EHRs.
Lessons from CancerLinQ
ASCO created the health technology platform CancerLinQ in 2014, envisioning that it could become an oncology-focused learning health system – a system in which internal data and experience are systematically integrated with external evidence, allowing knowledge to be put into practice.
CancerLinQ extracts data from EHRs and other sources via direct software connections. CancerLinQ then aggregates, harmonizes, and normalizes the data in a cloud-based environment.
The data are available to participating practices for quality improvement in patient care and secondary research. In 2020, records of cancer patients in the CancerLinQ database surpassed 2 million.
CancerLinQ has been successful. However, because of the nature of the EHR ecosystem and the scope and variability of data capture by clinicians, supporting a true learning health system has proven to be a formidable task. Postprocessing manual review using trained human curators is laborious and unsustainable.
The CancerLinQ experience illustrated that basic cancer-pertinent data should be standardized in the EHR and collected prospectively.
The mCODE model
The mCODE initiative seeks to facilitate progress in care quality, clinical research, and health care policy by developing and maintaining a standard, computable, interoperable data format.
Guiding principles that were adopted early in mCODE’s development included:
- A collaborative, noncommercial, use case–driven developmental model.
- Iterative processes.
- User-driven development, refinement, and maintenance.
- Low ongoing maintenance requirements.
A foundational moment in mCODE’s development involved achieving consensus among stakeholders that the project would fail if EHR vendors required additional data entry by users.
After pilot work, a real-world endpoints project, working-group deliberation, public comment, and refinement, the final data standard included six primary domains: patient, disease, laboratory data/vital signs, genomics, treatment, and outcome.
Each domain is further divided into several concepts with specific associated data elements. The data elements are modeled into value sets that specify the possible values for the data element.
To test mCODE, eight organizations representing oncology EHR vendors, standards developers, and research organizations participated in a cancer interoperability track. The comments helped refine mCODE version 1.0, which was released in March 2020 and is accessible via the mCODE website.
Additions will likely be reviewed by a technical review group after external piloting of new use cases.
Innovation, not regulation
Every interaction between a patient and care provider yields information that could lead to improved safety and better outcomes. To be successful, the information must be collected in a computable format so it can be aggregated with data from other patients, analyzed without manual curation, and shared through interoperable systems. Those data should also be secure enough to protect the privacy of individual patients.
mCODE is a consensus data standard for oncology that provides an infrastructure to share patient data between oncology practices and health care systems while promising little to no additional data entry on the part of clinicians. Adoption by sites will be critical, however.
Publishing the standard through the HL7 FHIR technology demonstrated to EHR vendors and regulatory agencies the stability of HL7, an essential requirement for its incorporation into software.
EHR vendors and others are engaged in the CodeX HL7 FHIR Accelerator to design projects to expand and/or modify mCODE. Their creativity and innovativeness via the external advisory mCODE council and/or CodeX will be encouraged to help mCODE reach its full potential.
As part of CodeX, the Community of Practice, an open forum for end users, was established to provide regular updates about mCODE-related initiatives and use cases to solicit in-progress input, according to Robert S. Miller, MD, medical director of CancerLinQ and an author of the mCODE review.
For mCODE to be embraced by all stakeholders, there should be no additional regulations. By engaging stakeholders in an enterprise that supports innovation and collaboration – without additional regulation – mCODE could maximize the potential of EHRs that, until now, have assisted us only marginally in accomplishing those goals.
mCODE is a joint venture of ASCO/CancerLinQ, the Alliance for Clinical Trials in Oncology Foundation, the MITRE Corporation, the American Society for Radiation Oncology, and the Society of Surgical Oncology.
Dr. Osterman disclosed a grant from the National Cancer Institute and relationships with Infostratix, eHealth, AstraZeneca, Outcomes Insights, Biodesix, MD Outlook, GenomOncology, Cota Healthcare, GE Healthcare, and Microsoft. Dr. Miller and the third review author disclosed no conflicts of interest.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
FROM JCO CLINICAL CANCER INFORMATICS
The Genital Examination in Dermatologic Practice
A casual survey of my dermatology co-residents yielded overwhelmingly unanimous results: A complete skin check goes from head to toe but does not routinely include an examination of the genital area. This observation contrasts starkly with the American Academy of Dermatology’s Basic Dermatology Curriculum, which recommends inspection of the entire skin surface including the mucous membranes (ie, eyes, mouth, anus, genital area) as part of the total-body skin examination (TBSE).1 It even draws attention to so-called hidden areas where lesions easily can be missed, such as the perianal skin. My observation seems far from anecdotal; even a recent attempt at optimizing movements in the TBSE neglected to include examination of the genitalia in the proposed method,2-4 and many practicing dermatologists seem to agree. A survey of international dermatologists at high-risk skin cancer clinics found male and female genitalia were the least frequently examined anatomy sites during the TBSE. Additionally, female genitalia were examined less frequently than male genitalia (labia majora, 28%; penis, 52%; P=.003).5 Another survey of US academic dermatologists (23 dermatologists, 1 nurse practitioner) found that only 4% always visually inspected the vulva during routine annual examinations, and 50% did not think that vulvar examination was the dermatologist’s responsibility.6 Similar findings were reported in a survey of US dermatology residents.7
Why is the genital area routinely omitted from the dermatologic TBSE? Based on the surveys of dermatologists and dermatology residents, the most common reason cited for not examining these sites was patient discomfort, but there also was a dominant belief that other specialties, such as gynecologists, urologists, or primary care providers, routinely examine these areas.5,7 Time constraints also were a concern.
Although examination of sensitive areas can be uncomfortable,8 most patients still expect these locations to be examined during the TBSE. In a survey of 500 adults presenting for TBSE at an academic dermatology clinic, 84% of respondents expected the dermatologist to examine the genital area.9 Similarly, another survey of patient preferences (N=443) for the TBSE found that only 31.3% of women and 12.5% of men preferred not to have their genital area examined.10 As providers, we may be uncomfortable examining the genital area; however, our patients mostly expect it as part of routine practice. There are a number of barriers that may prevent incorporating the genital examination into daily dermatologic practice.
Training in Genital Examinations
Adequate training may be an issue for provider comfort when examining the genital skin. In a survey of dermatology residency program directors (n=38) and residents (n=91), 61.7% reported receiving formal instruction on TBSE technique and 38.3% reported being self-taught. Examination of the genital skin was included only 40% of the time.11 Even vulvar disorder experts have admitted to receiving their training by self-teaching, with only 19% receiving vulvar training during residency and 11% during fellowship.12 Improving this training appears to be an ongoing effort.2
Passing the Buck
It may be easier to think that another provider is routinely examining genital skin based on the relative absence of this area in dermatologic training; however, that does not appear to be the case. In a 1999 survey of primary care providers, only 31% reported performing skin cancer screenings on their adult patients, citing lack of confidence in this clinical skill as the biggest hurdle.13 Similarly, changes in recommendations for the utility of the screening pelvic examination in asymptomatic, average-risk, nonpregnant adult women have decreased the performance of this examination in actual practice.14 Reviews of resident training in vulvovaginal disease also have shown that although dermatology residents receive slightly less formal training hours on vulvar skin disease, they see more than double the number of patients with vulvar disease per year when compared to obstetrics and gynecology residents.15 In practice, dermatologists generally are more confident when evaluating vulvar pigmented lesions than gynecologists.6
The Importance of the Genital Examination
Looking past these barriers seems essential to providing the best dermatologic care, as there are a multitude of neoplastic and inflammatory dermatoses that can affect the genital skin. Furthermore, early diagnosis and treatment of these conditions potentially can limit morbidity and mortality as well as improve quality of life. Genital melanomas are a good example. Although they may be rare, it is well known that genital melanomas are associated with an aggressive disease course and have worse outcomes than melanomas found elsewhere on the body.16,17 Increasing rates of genital and perianal keratinocyte carcinomas make including this as part of the TBSE even more important.18
We also should not forget that inflammatory conditions can routinely involve the genitals.19-21 Although robust data are lacking, chronic vulvar concerns frequently are seen in the primary care setting. In one study in the United Kingdom, 52% of general practitioners surveyed saw more than 3 patients per month with vulvar concerns.22 Even in common dermatologic conditions such as psoriasis and lichen planus, genital involvement often is overlooked despite its relative frequency.23-27 In one study, 60% of psoriasis patients with genital involvement had not had these lesions examined by a physician.28
Theoretically, TBSEs that include genital examination would yield higher and earlier detection rates of neoplasms as well as inflammatory dermatoses.29-32 Thus, there is real value in diagnosing ailments of the genital skin, and dermatologists are well prepared to manage these conditions. Consistently incorporating a genital examination within the TBSE is the first step.
An Approach to the Genital Skin Examination
As with the TBSE, no standardized protocol for the genital skin examination exists, and there is no consensus for how best to perform this evaluation. Ideally, both male and female patients should remove all clothing, including undergarments, though one study found patients preferred to keep undergarments on during the genital examination.10,33,34
In general, adult female genital anatomy is best viewed with the patient in the supine position.6,33,35 There is no clear agreement on the use of stirrups, and the decision to use these may be left to the discretion of the patient. One randomized clinical trial found that women undergoing routine gynecologic examination without stirrups reported less physical discomfort and had a reduced sense of vulnerability than women examined in stirrups.36 During the female genital examination, the head of the bed ideally should be positioned at a 30° to 45° angle to allow the provider to maintain eye contact and face-to-face communication with the patient.33 This positioning also facilitates the use of a handheld mirror to instruct patients on techniques for medication application as well as to point out sites of disease.
For adult males, the genital examination can be performed with the patient standing facing a seated examiner.35 The patient’s gown should be raised to the level of the umbilicus to expose the entire genital region. Good lighting is essential. These recommendations apply mainly to adults, but helpful tips on how to approach evaluating prepubertal children in the dermatology clinic are available.37
The presence of a chaperone also is optional for maximizing patient comfort but also may be helpful for providing medicolegal protection for the provider. It always should be offered regardless of patient gender. A dermatology study found that when patients were examined by a same-gender physician, women and men were more comfortable without a chaperone than with a chaperone, and patients generally preferred fewer bodies in the room during sensitive examinations.9
Educating Patients About the TBSE
The most helpful recommendation for successfully incorporating and performing the genital skin examination as part of the TBSE appears to be patient education. In a randomized double-arm study, patients who received pre-education consisting of written information explaining the need for a TBSE were less likely to be concerned about a genital examination compared to patients who received no information.38 Discussing that skin diseases, including melanoma, can arise in all areas of the body including the genital skin and encouraging patients to perform genital self-examinations is critical.35 In the age of the electronic health record and virtual communication, disseminating this information has become even easier.39 It may be beneficial to explore patients’ TBSE expectations at the outset through these varied avenues to help establish a trusted physician-patient relationship.40
Final Thoughts
Dermatologists should consistently offer a genital examination to all patients who present for a routine TBSE. Patients should be provided with adequate education to assess their comfort level for the skin examination. If a patient declines this examination, the dermatologist should ensure that another physician—be it a gynecologist, primary care provider, or other specialist—is routinely examining the area.6,7
- The skin exam. American Academy of Dermatology. https://digital-catalog.aad.org/diweb/catalog/launch/package/4/did/327974/iid/327974
- Helm MF, Hallock KK, Bisbee E, et al. Optimizing the total-body skin exam: an observational cohort study. J Am Acad Dermatol. 2019;81:1115-1119.
- Nielson CB, Grant-Kels JM. Commentary on “optimizing the total-body skin exam: an observational cohort study.” J Am Acad Dermatol. 2019;81:E131.
- Helm MF, Hallock KK, Bisbee E, et al. Reply to: “commentary on ‘optimizing the total-body skin exam: an observational cohort study.’” J Am Acad Dermatol. 2019;81:E133.
- Bajaj S, Wolner ZJ, Dusza SW, et al. Total body skin examination practices: a survey study amongst dermatologists at high-risk skin cancer clinics. Dermatol Pract Concept. 2019;9:132-138.
- Krathen MS, Liu CL, Loo DS. Vulvar melanoma: a missed opportunity for early intervention? J Am Acad Dermatol. 2012;66:697-698.
- Hosking AM, Chapman L, Zachary CB, et al. Anogenital examination practices among U.S. dermatology residents [published online January 9, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2019.12.061
- Grundström H, Wallin K, Berterö C. ‘You expose yourself in so many ways’: young women’s experiences of pelvic examination. J Psychosom Obstet Gynaecol. 2011;32:59-64.
- McClatchey Connors T, Reddy P, Weiss E, et al. Patient comfort and expectations for total body skin examinations: a cross-sectional study. J Am Acad Dermatol. 2019;81:615-617.
- Houston NA, Secrest AM, Harris RJ, et al. Patient preferences during skin cancer screening examination. JAMA Dermatol. 2016;152:1052-1054.
- Milchak M, Miller J, Dellasega C, et al. Education on total body skin examination in dermatology residency. Poster presented at: Association of Professors of Dermatology Annual Meeting; September 25-26, 2015; Chicago, IL.
- Venkatesan A, Farsani T, O’Sullivan P, et al. Identifying competencies in vulvar disorder management for medical students and residents: a survey of US vulvar disorder experts. J Low Genit Tract Dis. 2012;16:398-402.
- Kirsner RS, Muhkerjee S, Federman DG. Skin cancer screening in primary care: prevalence and barriers. J Am Acad Dermatol. 1999;41:564-566.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for gynecologic conditions with pelvic examination: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:947-953.
- Comstock JR, Endo JO, Kornik RI. Adequacy of dermatology and ob-gyn graduate medical education for inflammatory vulvovaginal skin disease: a nationwide needs assessment survey. Int J Womens Dermatol. 2020;6:182-185.
- Sanchez A, Rodríguez D, Allard CB, et al. Primary genitourinary melanoma: epidemiology and disease-specific survival in a large population-based cohort. Urol Oncol. 2016;34:E7-E14.
- Vyas R, Thompson CL, Zargar H, et al. Epidemiology of genitourinary melanoma in the United States: 1992 through 2012. J Am Acad Dermatol. 2016;75:144-150.
- Misitzis A, Beatson M, Weinstock MA. Keratinocyte carcinoma mortality in the United States as reported in death certificates, 2011-2017. Dermatol Surg. 2020;46:1135-1140.
- Sullivan AK, Straughair GJ, Marwood RP, et al. A multidisciplinary vulva clinic: the role of genito-urinary medicine. J Eur Acad Dermatol Venereol. 1999;13:36-40.
- Goncalves DLM, Romero RL, Ferreira PL, et al. Clinical and epidemiological profile of patients attended in a vulvar clinic of the dermatology outpatient unit of a tertiary hospital during a 4-year period. Int J Dermatol. 2019;58:1311-1316.
- Bauer A, Greif C, Vollandt R, et al. Vulval diseases need an interdisciplinary approach. Dermatology. 1999;199:223-226.
- Nunns D, Mandal D. The chronically symptomatic vulva: prevalence in primary health care. Genitourin Med. 1996;72:343-344.
- Meeuwis KA, de Hullu JA, de Jager ME, et al. Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands. J Eur Acad Dermatol Venereol. 2010;24:1425-1430.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Fouéré S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;(19 suppl 3):2-6.
- Eisen D. The evaluation of cutaneous, genital, scalp, nail, esophageal, and ocular involvement in patients with oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88:431-436.
- Meeuwis KAP, Potts Bleakman A, van de Kerkhof PCM, et al. Prevalence of genital psoriasis in patients with psoriasis. J Dermatolog Treat. 2018;29:754-760.
- Larsabal M, Ly S, Sbidian E, et al. GENIPSO: a French prospective study assessing instantaneous prevalence, clinical features and impact on quality of life of genital psoriasis among patients consulting for psoriasis. Br J Dermatol. 2019;180:647-656.
- Rigel DS, Friedman RJ, Kopf AW, et al. Importance of complete cutaneous examination for the detection of malignant melanoma. J Am Acad Dermatol. 1986;14(5 pt 1):857-860.
- De Rooij MJ, Rampen FH, Schouten LJ, et al. Total skin examination during screening for malignant melanoma does not increase the detection rate. Br J Dermatol. 1996;135:42-45.
- Johansson M, Brodersen J, Gøtzsche PC, et al. Screening for reducing morbidity and mortality in malignant melanoma. Cochrane Database Syst Rev. 2019;6:CD012352.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:429-435.
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases: approach to the patient. J Am Acad Dermatol. 2020;82:1277-1284.
- Chen C. How full is a full body skin exam? investigation into the practice of the full body skin exam as conducted by board-certified and board-eligibile dermatologists. Michigan State University. Published April 24, 2015. Accessed February 4, 2021. https://cdn.ymaws.com/www.aocd.org/resource/resmgr/2015SpringMeeting/ChenSpr15.pdf
- Zikry J, Chapman LW, Korta DZ, et al. Genital melanoma: are we adequately screening our patients? Dermatol Online J. 2017;23:13030/qt7zk476vn.
- Seehusen DA, Johnson DR, Earwood JS, et al. Improving women’s experience during speculum examinations at routine gynaecological visits: randomised clinical trial [published online June 27, 2006]. BMJ. 2006;333:171.
- Habeshian K, Fowler K, Gomez-Lobo V, et al. Guidelines for pediatric anogenital examination: insights from our vulvar dermatology clinic. Pediatr Dermatol. 2018;35:693-695.
- Leffell DJ, Berwick M, Bolognia J. The effect of pre-education on patient compliance with full-body examination in a public skin cancer screening. J Dermatol Surg Oncol. 1993;19:660-663.
- Hong J, Nguyen TV, Prose NS. Compassionate care: enhancing physician-patient communication and education in dermatology: part II: patient education. J Am Acad Dermatol. 2013;68:364.e361-310.
- Rosamilia LL. The naked truth about total body skin examination: a lesson from Goldilocks and the Three Bears. American Academy of Dermatology. Published November 13, 2019. Accessed February 4, 2021. https://www.aad.org/dw/dw-insights-and-inquiries/2019-archive/november/dwii-11-13-19-the-naked-truth-about-total-body-skin-examination-a-lesson-from-goldilocks-and-the-three-bears
A casual survey of my dermatology co-residents yielded overwhelmingly unanimous results: A complete skin check goes from head to toe but does not routinely include an examination of the genital area. This observation contrasts starkly with the American Academy of Dermatology’s Basic Dermatology Curriculum, which recommends inspection of the entire skin surface including the mucous membranes (ie, eyes, mouth, anus, genital area) as part of the total-body skin examination (TBSE).1 It even draws attention to so-called hidden areas where lesions easily can be missed, such as the perianal skin. My observation seems far from anecdotal; even a recent attempt at optimizing movements in the TBSE neglected to include examination of the genitalia in the proposed method,2-4 and many practicing dermatologists seem to agree. A survey of international dermatologists at high-risk skin cancer clinics found male and female genitalia were the least frequently examined anatomy sites during the TBSE. Additionally, female genitalia were examined less frequently than male genitalia (labia majora, 28%; penis, 52%; P=.003).5 Another survey of US academic dermatologists (23 dermatologists, 1 nurse practitioner) found that only 4% always visually inspected the vulva during routine annual examinations, and 50% did not think that vulvar examination was the dermatologist’s responsibility.6 Similar findings were reported in a survey of US dermatology residents.7
Why is the genital area routinely omitted from the dermatologic TBSE? Based on the surveys of dermatologists and dermatology residents, the most common reason cited for not examining these sites was patient discomfort, but there also was a dominant belief that other specialties, such as gynecologists, urologists, or primary care providers, routinely examine these areas.5,7 Time constraints also were a concern.
Although examination of sensitive areas can be uncomfortable,8 most patients still expect these locations to be examined during the TBSE. In a survey of 500 adults presenting for TBSE at an academic dermatology clinic, 84% of respondents expected the dermatologist to examine the genital area.9 Similarly, another survey of patient preferences (N=443) for the TBSE found that only 31.3% of women and 12.5% of men preferred not to have their genital area examined.10 As providers, we may be uncomfortable examining the genital area; however, our patients mostly expect it as part of routine practice. There are a number of barriers that may prevent incorporating the genital examination into daily dermatologic practice.
Training in Genital Examinations
Adequate training may be an issue for provider comfort when examining the genital skin. In a survey of dermatology residency program directors (n=38) and residents (n=91), 61.7% reported receiving formal instruction on TBSE technique and 38.3% reported being self-taught. Examination of the genital skin was included only 40% of the time.11 Even vulvar disorder experts have admitted to receiving their training by self-teaching, with only 19% receiving vulvar training during residency and 11% during fellowship.12 Improving this training appears to be an ongoing effort.2
Passing the Buck
It may be easier to think that another provider is routinely examining genital skin based on the relative absence of this area in dermatologic training; however, that does not appear to be the case. In a 1999 survey of primary care providers, only 31% reported performing skin cancer screenings on their adult patients, citing lack of confidence in this clinical skill as the biggest hurdle.13 Similarly, changes in recommendations for the utility of the screening pelvic examination in asymptomatic, average-risk, nonpregnant adult women have decreased the performance of this examination in actual practice.14 Reviews of resident training in vulvovaginal disease also have shown that although dermatology residents receive slightly less formal training hours on vulvar skin disease, they see more than double the number of patients with vulvar disease per year when compared to obstetrics and gynecology residents.15 In practice, dermatologists generally are more confident when evaluating vulvar pigmented lesions than gynecologists.6
The Importance of the Genital Examination
Looking past these barriers seems essential to providing the best dermatologic care, as there are a multitude of neoplastic and inflammatory dermatoses that can affect the genital skin. Furthermore, early diagnosis and treatment of these conditions potentially can limit morbidity and mortality as well as improve quality of life. Genital melanomas are a good example. Although they may be rare, it is well known that genital melanomas are associated with an aggressive disease course and have worse outcomes than melanomas found elsewhere on the body.16,17 Increasing rates of genital and perianal keratinocyte carcinomas make including this as part of the TBSE even more important.18
We also should not forget that inflammatory conditions can routinely involve the genitals.19-21 Although robust data are lacking, chronic vulvar concerns frequently are seen in the primary care setting. In one study in the United Kingdom, 52% of general practitioners surveyed saw more than 3 patients per month with vulvar concerns.22 Even in common dermatologic conditions such as psoriasis and lichen planus, genital involvement often is overlooked despite its relative frequency.23-27 In one study, 60% of psoriasis patients with genital involvement had not had these lesions examined by a physician.28
Theoretically, TBSEs that include genital examination would yield higher and earlier detection rates of neoplasms as well as inflammatory dermatoses.29-32 Thus, there is real value in diagnosing ailments of the genital skin, and dermatologists are well prepared to manage these conditions. Consistently incorporating a genital examination within the TBSE is the first step.
An Approach to the Genital Skin Examination
As with the TBSE, no standardized protocol for the genital skin examination exists, and there is no consensus for how best to perform this evaluation. Ideally, both male and female patients should remove all clothing, including undergarments, though one study found patients preferred to keep undergarments on during the genital examination.10,33,34
In general, adult female genital anatomy is best viewed with the patient in the supine position.6,33,35 There is no clear agreement on the use of stirrups, and the decision to use these may be left to the discretion of the patient. One randomized clinical trial found that women undergoing routine gynecologic examination without stirrups reported less physical discomfort and had a reduced sense of vulnerability than women examined in stirrups.36 During the female genital examination, the head of the bed ideally should be positioned at a 30° to 45° angle to allow the provider to maintain eye contact and face-to-face communication with the patient.33 This positioning also facilitates the use of a handheld mirror to instruct patients on techniques for medication application as well as to point out sites of disease.
For adult males, the genital examination can be performed with the patient standing facing a seated examiner.35 The patient’s gown should be raised to the level of the umbilicus to expose the entire genital region. Good lighting is essential. These recommendations apply mainly to adults, but helpful tips on how to approach evaluating prepubertal children in the dermatology clinic are available.37
The presence of a chaperone also is optional for maximizing patient comfort but also may be helpful for providing medicolegal protection for the provider. It always should be offered regardless of patient gender. A dermatology study found that when patients were examined by a same-gender physician, women and men were more comfortable without a chaperone than with a chaperone, and patients generally preferred fewer bodies in the room during sensitive examinations.9
Educating Patients About the TBSE
The most helpful recommendation for successfully incorporating and performing the genital skin examination as part of the TBSE appears to be patient education. In a randomized double-arm study, patients who received pre-education consisting of written information explaining the need for a TBSE were less likely to be concerned about a genital examination compared to patients who received no information.38 Discussing that skin diseases, including melanoma, can arise in all areas of the body including the genital skin and encouraging patients to perform genital self-examinations is critical.35 In the age of the electronic health record and virtual communication, disseminating this information has become even easier.39 It may be beneficial to explore patients’ TBSE expectations at the outset through these varied avenues to help establish a trusted physician-patient relationship.40
Final Thoughts
Dermatologists should consistently offer a genital examination to all patients who present for a routine TBSE. Patients should be provided with adequate education to assess their comfort level for the skin examination. If a patient declines this examination, the dermatologist should ensure that another physician—be it a gynecologist, primary care provider, or other specialist—is routinely examining the area.6,7
A casual survey of my dermatology co-residents yielded overwhelmingly unanimous results: A complete skin check goes from head to toe but does not routinely include an examination of the genital area. This observation contrasts starkly with the American Academy of Dermatology’s Basic Dermatology Curriculum, which recommends inspection of the entire skin surface including the mucous membranes (ie, eyes, mouth, anus, genital area) as part of the total-body skin examination (TBSE).1 It even draws attention to so-called hidden areas where lesions easily can be missed, such as the perianal skin. My observation seems far from anecdotal; even a recent attempt at optimizing movements in the TBSE neglected to include examination of the genitalia in the proposed method,2-4 and many practicing dermatologists seem to agree. A survey of international dermatologists at high-risk skin cancer clinics found male and female genitalia were the least frequently examined anatomy sites during the TBSE. Additionally, female genitalia were examined less frequently than male genitalia (labia majora, 28%; penis, 52%; P=.003).5 Another survey of US academic dermatologists (23 dermatologists, 1 nurse practitioner) found that only 4% always visually inspected the vulva during routine annual examinations, and 50% did not think that vulvar examination was the dermatologist’s responsibility.6 Similar findings were reported in a survey of US dermatology residents.7
Why is the genital area routinely omitted from the dermatologic TBSE? Based on the surveys of dermatologists and dermatology residents, the most common reason cited for not examining these sites was patient discomfort, but there also was a dominant belief that other specialties, such as gynecologists, urologists, or primary care providers, routinely examine these areas.5,7 Time constraints also were a concern.
Although examination of sensitive areas can be uncomfortable,8 most patients still expect these locations to be examined during the TBSE. In a survey of 500 adults presenting for TBSE at an academic dermatology clinic, 84% of respondents expected the dermatologist to examine the genital area.9 Similarly, another survey of patient preferences (N=443) for the TBSE found that only 31.3% of women and 12.5% of men preferred not to have their genital area examined.10 As providers, we may be uncomfortable examining the genital area; however, our patients mostly expect it as part of routine practice. There are a number of barriers that may prevent incorporating the genital examination into daily dermatologic practice.
Training in Genital Examinations
Adequate training may be an issue for provider comfort when examining the genital skin. In a survey of dermatology residency program directors (n=38) and residents (n=91), 61.7% reported receiving formal instruction on TBSE technique and 38.3% reported being self-taught. Examination of the genital skin was included only 40% of the time.11 Even vulvar disorder experts have admitted to receiving their training by self-teaching, with only 19% receiving vulvar training during residency and 11% during fellowship.12 Improving this training appears to be an ongoing effort.2
Passing the Buck
It may be easier to think that another provider is routinely examining genital skin based on the relative absence of this area in dermatologic training; however, that does not appear to be the case. In a 1999 survey of primary care providers, only 31% reported performing skin cancer screenings on their adult patients, citing lack of confidence in this clinical skill as the biggest hurdle.13 Similarly, changes in recommendations for the utility of the screening pelvic examination in asymptomatic, average-risk, nonpregnant adult women have decreased the performance of this examination in actual practice.14 Reviews of resident training in vulvovaginal disease also have shown that although dermatology residents receive slightly less formal training hours on vulvar skin disease, they see more than double the number of patients with vulvar disease per year when compared to obstetrics and gynecology residents.15 In practice, dermatologists generally are more confident when evaluating vulvar pigmented lesions than gynecologists.6
The Importance of the Genital Examination
Looking past these barriers seems essential to providing the best dermatologic care, as there are a multitude of neoplastic and inflammatory dermatoses that can affect the genital skin. Furthermore, early diagnosis and treatment of these conditions potentially can limit morbidity and mortality as well as improve quality of life. Genital melanomas are a good example. Although they may be rare, it is well known that genital melanomas are associated with an aggressive disease course and have worse outcomes than melanomas found elsewhere on the body.16,17 Increasing rates of genital and perianal keratinocyte carcinomas make including this as part of the TBSE even more important.18
We also should not forget that inflammatory conditions can routinely involve the genitals.19-21 Although robust data are lacking, chronic vulvar concerns frequently are seen in the primary care setting. In one study in the United Kingdom, 52% of general practitioners surveyed saw more than 3 patients per month with vulvar concerns.22 Even in common dermatologic conditions such as psoriasis and lichen planus, genital involvement often is overlooked despite its relative frequency.23-27 In one study, 60% of psoriasis patients with genital involvement had not had these lesions examined by a physician.28
Theoretically, TBSEs that include genital examination would yield higher and earlier detection rates of neoplasms as well as inflammatory dermatoses.29-32 Thus, there is real value in diagnosing ailments of the genital skin, and dermatologists are well prepared to manage these conditions. Consistently incorporating a genital examination within the TBSE is the first step.
An Approach to the Genital Skin Examination
As with the TBSE, no standardized protocol for the genital skin examination exists, and there is no consensus for how best to perform this evaluation. Ideally, both male and female patients should remove all clothing, including undergarments, though one study found patients preferred to keep undergarments on during the genital examination.10,33,34
In general, adult female genital anatomy is best viewed with the patient in the supine position.6,33,35 There is no clear agreement on the use of stirrups, and the decision to use these may be left to the discretion of the patient. One randomized clinical trial found that women undergoing routine gynecologic examination without stirrups reported less physical discomfort and had a reduced sense of vulnerability than women examined in stirrups.36 During the female genital examination, the head of the bed ideally should be positioned at a 30° to 45° angle to allow the provider to maintain eye contact and face-to-face communication with the patient.33 This positioning also facilitates the use of a handheld mirror to instruct patients on techniques for medication application as well as to point out sites of disease.
For adult males, the genital examination can be performed with the patient standing facing a seated examiner.35 The patient’s gown should be raised to the level of the umbilicus to expose the entire genital region. Good lighting is essential. These recommendations apply mainly to adults, but helpful tips on how to approach evaluating prepubertal children in the dermatology clinic are available.37
The presence of a chaperone also is optional for maximizing patient comfort but also may be helpful for providing medicolegal protection for the provider. It always should be offered regardless of patient gender. A dermatology study found that when patients were examined by a same-gender physician, women and men were more comfortable without a chaperone than with a chaperone, and patients generally preferred fewer bodies in the room during sensitive examinations.9
Educating Patients About the TBSE
The most helpful recommendation for successfully incorporating and performing the genital skin examination as part of the TBSE appears to be patient education. In a randomized double-arm study, patients who received pre-education consisting of written information explaining the need for a TBSE were less likely to be concerned about a genital examination compared to patients who received no information.38 Discussing that skin diseases, including melanoma, can arise in all areas of the body including the genital skin and encouraging patients to perform genital self-examinations is critical.35 In the age of the electronic health record and virtual communication, disseminating this information has become even easier.39 It may be beneficial to explore patients’ TBSE expectations at the outset through these varied avenues to help establish a trusted physician-patient relationship.40
Final Thoughts
Dermatologists should consistently offer a genital examination to all patients who present for a routine TBSE. Patients should be provided with adequate education to assess their comfort level for the skin examination. If a patient declines this examination, the dermatologist should ensure that another physician—be it a gynecologist, primary care provider, or other specialist—is routinely examining the area.6,7
- The skin exam. American Academy of Dermatology. https://digital-catalog.aad.org/diweb/catalog/launch/package/4/did/327974/iid/327974
- Helm MF, Hallock KK, Bisbee E, et al. Optimizing the total-body skin exam: an observational cohort study. J Am Acad Dermatol. 2019;81:1115-1119.
- Nielson CB, Grant-Kels JM. Commentary on “optimizing the total-body skin exam: an observational cohort study.” J Am Acad Dermatol. 2019;81:E131.
- Helm MF, Hallock KK, Bisbee E, et al. Reply to: “commentary on ‘optimizing the total-body skin exam: an observational cohort study.’” J Am Acad Dermatol. 2019;81:E133.
- Bajaj S, Wolner ZJ, Dusza SW, et al. Total body skin examination practices: a survey study amongst dermatologists at high-risk skin cancer clinics. Dermatol Pract Concept. 2019;9:132-138.
- Krathen MS, Liu CL, Loo DS. Vulvar melanoma: a missed opportunity for early intervention? J Am Acad Dermatol. 2012;66:697-698.
- Hosking AM, Chapman L, Zachary CB, et al. Anogenital examination practices among U.S. dermatology residents [published online January 9, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2019.12.061
- Grundström H, Wallin K, Berterö C. ‘You expose yourself in so many ways’: young women’s experiences of pelvic examination. J Psychosom Obstet Gynaecol. 2011;32:59-64.
- McClatchey Connors T, Reddy P, Weiss E, et al. Patient comfort and expectations for total body skin examinations: a cross-sectional study. J Am Acad Dermatol. 2019;81:615-617.
- Houston NA, Secrest AM, Harris RJ, et al. Patient preferences during skin cancer screening examination. JAMA Dermatol. 2016;152:1052-1054.
- Milchak M, Miller J, Dellasega C, et al. Education on total body skin examination in dermatology residency. Poster presented at: Association of Professors of Dermatology Annual Meeting; September 25-26, 2015; Chicago, IL.
- Venkatesan A, Farsani T, O’Sullivan P, et al. Identifying competencies in vulvar disorder management for medical students and residents: a survey of US vulvar disorder experts. J Low Genit Tract Dis. 2012;16:398-402.
- Kirsner RS, Muhkerjee S, Federman DG. Skin cancer screening in primary care: prevalence and barriers. J Am Acad Dermatol. 1999;41:564-566.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for gynecologic conditions with pelvic examination: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:947-953.
- Comstock JR, Endo JO, Kornik RI. Adequacy of dermatology and ob-gyn graduate medical education for inflammatory vulvovaginal skin disease: a nationwide needs assessment survey. Int J Womens Dermatol. 2020;6:182-185.
- Sanchez A, Rodríguez D, Allard CB, et al. Primary genitourinary melanoma: epidemiology and disease-specific survival in a large population-based cohort. Urol Oncol. 2016;34:E7-E14.
- Vyas R, Thompson CL, Zargar H, et al. Epidemiology of genitourinary melanoma in the United States: 1992 through 2012. J Am Acad Dermatol. 2016;75:144-150.
- Misitzis A, Beatson M, Weinstock MA. Keratinocyte carcinoma mortality in the United States as reported in death certificates, 2011-2017. Dermatol Surg. 2020;46:1135-1140.
- Sullivan AK, Straughair GJ, Marwood RP, et al. A multidisciplinary vulva clinic: the role of genito-urinary medicine. J Eur Acad Dermatol Venereol. 1999;13:36-40.
- Goncalves DLM, Romero RL, Ferreira PL, et al. Clinical and epidemiological profile of patients attended in a vulvar clinic of the dermatology outpatient unit of a tertiary hospital during a 4-year period. Int J Dermatol. 2019;58:1311-1316.
- Bauer A, Greif C, Vollandt R, et al. Vulval diseases need an interdisciplinary approach. Dermatology. 1999;199:223-226.
- Nunns D, Mandal D. The chronically symptomatic vulva: prevalence in primary health care. Genitourin Med. 1996;72:343-344.
- Meeuwis KA, de Hullu JA, de Jager ME, et al. Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands. J Eur Acad Dermatol Venereol. 2010;24:1425-1430.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Fouéré S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;(19 suppl 3):2-6.
- Eisen D. The evaluation of cutaneous, genital, scalp, nail, esophageal, and ocular involvement in patients with oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88:431-436.
- Meeuwis KAP, Potts Bleakman A, van de Kerkhof PCM, et al. Prevalence of genital psoriasis in patients with psoriasis. J Dermatolog Treat. 2018;29:754-760.
- Larsabal M, Ly S, Sbidian E, et al. GENIPSO: a French prospective study assessing instantaneous prevalence, clinical features and impact on quality of life of genital psoriasis among patients consulting for psoriasis. Br J Dermatol. 2019;180:647-656.
- Rigel DS, Friedman RJ, Kopf AW, et al. Importance of complete cutaneous examination for the detection of malignant melanoma. J Am Acad Dermatol. 1986;14(5 pt 1):857-860.
- De Rooij MJ, Rampen FH, Schouten LJ, et al. Total skin examination during screening for malignant melanoma does not increase the detection rate. Br J Dermatol. 1996;135:42-45.
- Johansson M, Brodersen J, Gøtzsche PC, et al. Screening for reducing morbidity and mortality in malignant melanoma. Cochrane Database Syst Rev. 2019;6:CD012352.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:429-435.
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases: approach to the patient. J Am Acad Dermatol. 2020;82:1277-1284.
- Chen C. How full is a full body skin exam? investigation into the practice of the full body skin exam as conducted by board-certified and board-eligibile dermatologists. Michigan State University. Published April 24, 2015. Accessed February 4, 2021. https://cdn.ymaws.com/www.aocd.org/resource/resmgr/2015SpringMeeting/ChenSpr15.pdf
- Zikry J, Chapman LW, Korta DZ, et al. Genital melanoma: are we adequately screening our patients? Dermatol Online J. 2017;23:13030/qt7zk476vn.
- Seehusen DA, Johnson DR, Earwood JS, et al. Improving women’s experience during speculum examinations at routine gynaecological visits: randomised clinical trial [published online June 27, 2006]. BMJ. 2006;333:171.
- Habeshian K, Fowler K, Gomez-Lobo V, et al. Guidelines for pediatric anogenital examination: insights from our vulvar dermatology clinic. Pediatr Dermatol. 2018;35:693-695.
- Leffell DJ, Berwick M, Bolognia J. The effect of pre-education on patient compliance with full-body examination in a public skin cancer screening. J Dermatol Surg Oncol. 1993;19:660-663.
- Hong J, Nguyen TV, Prose NS. Compassionate care: enhancing physician-patient communication and education in dermatology: part II: patient education. J Am Acad Dermatol. 2013;68:364.e361-310.
- Rosamilia LL. The naked truth about total body skin examination: a lesson from Goldilocks and the Three Bears. American Academy of Dermatology. Published November 13, 2019. Accessed February 4, 2021. https://www.aad.org/dw/dw-insights-and-inquiries/2019-archive/november/dwii-11-13-19-the-naked-truth-about-total-body-skin-examination-a-lesson-from-goldilocks-and-the-three-bears
- The skin exam. American Academy of Dermatology. https://digital-catalog.aad.org/diweb/catalog/launch/package/4/did/327974/iid/327974
- Helm MF, Hallock KK, Bisbee E, et al. Optimizing the total-body skin exam: an observational cohort study. J Am Acad Dermatol. 2019;81:1115-1119.
- Nielson CB, Grant-Kels JM. Commentary on “optimizing the total-body skin exam: an observational cohort study.” J Am Acad Dermatol. 2019;81:E131.
- Helm MF, Hallock KK, Bisbee E, et al. Reply to: “commentary on ‘optimizing the total-body skin exam: an observational cohort study.’” J Am Acad Dermatol. 2019;81:E133.
- Bajaj S, Wolner ZJ, Dusza SW, et al. Total body skin examination practices: a survey study amongst dermatologists at high-risk skin cancer clinics. Dermatol Pract Concept. 2019;9:132-138.
- Krathen MS, Liu CL, Loo DS. Vulvar melanoma: a missed opportunity for early intervention? J Am Acad Dermatol. 2012;66:697-698.
- Hosking AM, Chapman L, Zachary CB, et al. Anogenital examination practices among U.S. dermatology residents [published online January 9, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2019.12.061
- Grundström H, Wallin K, Berterö C. ‘You expose yourself in so many ways’: young women’s experiences of pelvic examination. J Psychosom Obstet Gynaecol. 2011;32:59-64.
- McClatchey Connors T, Reddy P, Weiss E, et al. Patient comfort and expectations for total body skin examinations: a cross-sectional study. J Am Acad Dermatol. 2019;81:615-617.
- Houston NA, Secrest AM, Harris RJ, et al. Patient preferences during skin cancer screening examination. JAMA Dermatol. 2016;152:1052-1054.
- Milchak M, Miller J, Dellasega C, et al. Education on total body skin examination in dermatology residency. Poster presented at: Association of Professors of Dermatology Annual Meeting; September 25-26, 2015; Chicago, IL.
- Venkatesan A, Farsani T, O’Sullivan P, et al. Identifying competencies in vulvar disorder management for medical students and residents: a survey of US vulvar disorder experts. J Low Genit Tract Dis. 2012;16:398-402.
- Kirsner RS, Muhkerjee S, Federman DG. Skin cancer screening in primary care: prevalence and barriers. J Am Acad Dermatol. 1999;41:564-566.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for gynecologic conditions with pelvic examination: US Preventive Services Task Force recommendation statement. JAMA. 2017;317:947-953.
- Comstock JR, Endo JO, Kornik RI. Adequacy of dermatology and ob-gyn graduate medical education for inflammatory vulvovaginal skin disease: a nationwide needs assessment survey. Int J Womens Dermatol. 2020;6:182-185.
- Sanchez A, Rodríguez D, Allard CB, et al. Primary genitourinary melanoma: epidemiology and disease-specific survival in a large population-based cohort. Urol Oncol. 2016;34:E7-E14.
- Vyas R, Thompson CL, Zargar H, et al. Epidemiology of genitourinary melanoma in the United States: 1992 through 2012. J Am Acad Dermatol. 2016;75:144-150.
- Misitzis A, Beatson M, Weinstock MA. Keratinocyte carcinoma mortality in the United States as reported in death certificates, 2011-2017. Dermatol Surg. 2020;46:1135-1140.
- Sullivan AK, Straughair GJ, Marwood RP, et al. A multidisciplinary vulva clinic: the role of genito-urinary medicine. J Eur Acad Dermatol Venereol. 1999;13:36-40.
- Goncalves DLM, Romero RL, Ferreira PL, et al. Clinical and epidemiological profile of patients attended in a vulvar clinic of the dermatology outpatient unit of a tertiary hospital during a 4-year period. Int J Dermatol. 2019;58:1311-1316.
- Bauer A, Greif C, Vollandt R, et al. Vulval diseases need an interdisciplinary approach. Dermatology. 1999;199:223-226.
- Nunns D, Mandal D. The chronically symptomatic vulva: prevalence in primary health care. Genitourin Med. 1996;72:343-344.
- Meeuwis KA, de Hullu JA, de Jager ME, et al. Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands. J Eur Acad Dermatol Venereol. 2010;24:1425-1430.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Fouéré S, Adjadj L, Pawin H. How patients experience psoriasis: results from a European survey. J Eur Acad Dermatol Venereol. 2005;(19 suppl 3):2-6.
- Eisen D. The evaluation of cutaneous, genital, scalp, nail, esophageal, and ocular involvement in patients with oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88:431-436.
- Meeuwis KAP, Potts Bleakman A, van de Kerkhof PCM, et al. Prevalence of genital psoriasis in patients with psoriasis. J Dermatolog Treat. 2018;29:754-760.
- Larsabal M, Ly S, Sbidian E, et al. GENIPSO: a French prospective study assessing instantaneous prevalence, clinical features and impact on quality of life of genital psoriasis among patients consulting for psoriasis. Br J Dermatol. 2019;180:647-656.
- Rigel DS, Friedman RJ, Kopf AW, et al. Importance of complete cutaneous examination for the detection of malignant melanoma. J Am Acad Dermatol. 1986;14(5 pt 1):857-860.
- De Rooij MJ, Rampen FH, Schouten LJ, et al. Total skin examination during screening for malignant melanoma does not increase the detection rate. Br J Dermatol. 1996;135:42-45.
- Johansson M, Brodersen J, Gøtzsche PC, et al. Screening for reducing morbidity and mortality in malignant melanoma. Cochrane Database Syst Rev. 2019;6:CD012352.
- Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2016;316:429-435.
- Mauskar MM, Marathe K, Venkatesan A, et al. Vulvar diseases: approach to the patient. J Am Acad Dermatol. 2020;82:1277-1284.
- Chen C. How full is a full body skin exam? investigation into the practice of the full body skin exam as conducted by board-certified and board-eligibile dermatologists. Michigan State University. Published April 24, 2015. Accessed February 4, 2021. https://cdn.ymaws.com/www.aocd.org/resource/resmgr/2015SpringMeeting/ChenSpr15.pdf
- Zikry J, Chapman LW, Korta DZ, et al. Genital melanoma: are we adequately screening our patients? Dermatol Online J. 2017;23:13030/qt7zk476vn.
- Seehusen DA, Johnson DR, Earwood JS, et al. Improving women’s experience during speculum examinations at routine gynaecological visits: randomised clinical trial [published online June 27, 2006]. BMJ. 2006;333:171.
- Habeshian K, Fowler K, Gomez-Lobo V, et al. Guidelines for pediatric anogenital examination: insights from our vulvar dermatology clinic. Pediatr Dermatol. 2018;35:693-695.
- Leffell DJ, Berwick M, Bolognia J. The effect of pre-education on patient compliance with full-body examination in a public skin cancer screening. J Dermatol Surg Oncol. 1993;19:660-663.
- Hong J, Nguyen TV, Prose NS. Compassionate care: enhancing physician-patient communication and education in dermatology: part II: patient education. J Am Acad Dermatol. 2013;68:364.e361-310.
- Rosamilia LL. The naked truth about total body skin examination: a lesson from Goldilocks and the Three Bears. American Academy of Dermatology. Published November 13, 2019. Accessed February 4, 2021. https://www.aad.org/dw/dw-insights-and-inquiries/2019-archive/november/dwii-11-13-19-the-naked-truth-about-total-body-skin-examination-a-lesson-from-goldilocks-and-the-three-bears
Resident Pearls
- Dermatologists should offer a genital examination to all patients who present for a routine total-body skin examination.
- It is critical to educate patients about the importance of examining the genital skin by discussing that skin diseases can arise in all areas of the body including the genital area. Encouraging genital self-examination also is helpful.
- If a patient declines, the dermatologist should strive to ensure that another provider is examining the genital skin.
AI detects ugly-duckling skin lesions for melanoma follow-up
.
The system could use photographs of large areas of patients’ bodies taken with ordinary cameras in primary care or by the patients themselves to screen for early-stage melanoma, said Luis R. Soenksen, PhD, a postdoctoral associate and venture builder at Massachusetts Institute of Technology in Cambridge, Mass.
“We believe we’re providing technology for that to happen at a massive scale, which is what is needed to reduce mortality rates,” he said in an interview.
He and his colleagues published their findings in Science Translational Medicine.
Diagnosing skin lesions has already proved one of the most promising medical applications of AI. In a 2017 paper, researchers reported that a deep neural network had classified skin lesions more accurately than did dermatologists. But so far, most such programs depend on experts to preselect the lesions worthy of analysis. And they use images from dermoscopy or single-lesion near-field photography.
Dr. Soenksen and colleagues wanted a system that could use a variety of cameras such as those in smartphones under a variety of conditions to assess lesions over wide areas of anatomy.
So they programmed their convolutional neural network to simultaneously use two approaches for screening lesions. Like the earlier systems, theirs looks for characteristics of individual lesions, such as asymmetry, border unevenness, color distribution, diameter, and evolution (ABCDE.) But it also looks for lesion saliency, a comparison of the lesions on the skin of one individual to identify the “ugly ducklings” that stand out from the rest.
They trained the system using 20,388 wide-field images from 133 patients at the Hospital Gregorio Marañón in Madrid, as well as publicly available images. The images were taken with a variety of consumer-grade cameras, about half of them nondermoscopy, and included backgrounds, skin edges, bare skin sections, nonsuspicious pigmented lesions, and suspicious pigmented lesions. The lesions in the images were visually classified by a consensus of three board-certified dermatologists.
Once they trained the system, the researchers tested it on another 6,796 images from the same patients, using the dermatologists’ classification as the gold standard. The system distinguished the suspicious lesions with 90.3% sensitivity (true positive), 89.9% specificity (true negative), and 86.56% accuracy.
Dr. Soenksen said he could envision photos acquired for screening in three scenarios. First, people could photograph themselves, or someone else at their homes could photograph them. These photos could even include whole nude bodies.
Second, clinicians could photograph patients’ body parts during medical visits for other purposes. “It makes sense to do these evaluations in the point of care where a referral can actually happen, like the primary care office,” said Dr. Soenksen.
Third, photos could be taken at places where people show up in bathing suits.
In each scenario, the system would then tell patients whether any lesions needed evaluation by a dermatologist.
To ensure privacy, Dr. Soenksen envisions using devices that do not transmit all the data to the cloud but instead do at least some of the calculations on their own. High-end smartphones have sufficient computing capacity for that, he said.
In their next phase of this work, the researchers would like to test the system on more skin of color cases and in more varied conditions, said Dr. Soenksen. And they would like to put it through randomized clinical trials, potentially using biopsies to validate the results.
That’s a key step, said Veronica Rotemberg, MD, PhD, director of the dermatology imaging informatics program at Memorial Sloan Kettering Cancer Center, New York.
“Usually when we think about melanoma, we think of histology as the gold standard, or specific subtypes of melanoma as a gold standard,” she said in an interview.
The technology also raises the question of excessive screening, she said. “Identifying the ugly duckling could be extremely important in finding more melanoma,” she said. “But in a patient who doesn’t have melanoma, it could lead to a lot of unnecessary biopsies.”
The sheer number of referrals generated by such a system could overwhelm the dermatologists assigned to follow up on them, she added.
Still, Dr. Rotemberg said, the study is “a good proof of concept.” Ugly duckling analysis is a very active area of AI research with thousands of teams of researchers worldwide working on systems similar to this one, she added. “I’m so excited for the authors.”
Neither Dr. Soenksen nor Dr. Rotemberg disclosed any relevant financial interests.
.
The system could use photographs of large areas of patients’ bodies taken with ordinary cameras in primary care or by the patients themselves to screen for early-stage melanoma, said Luis R. Soenksen, PhD, a postdoctoral associate and venture builder at Massachusetts Institute of Technology in Cambridge, Mass.
“We believe we’re providing technology for that to happen at a massive scale, which is what is needed to reduce mortality rates,” he said in an interview.
He and his colleagues published their findings in Science Translational Medicine.
Diagnosing skin lesions has already proved one of the most promising medical applications of AI. In a 2017 paper, researchers reported that a deep neural network had classified skin lesions more accurately than did dermatologists. But so far, most such programs depend on experts to preselect the lesions worthy of analysis. And they use images from dermoscopy or single-lesion near-field photography.
Dr. Soenksen and colleagues wanted a system that could use a variety of cameras such as those in smartphones under a variety of conditions to assess lesions over wide areas of anatomy.
So they programmed their convolutional neural network to simultaneously use two approaches for screening lesions. Like the earlier systems, theirs looks for characteristics of individual lesions, such as asymmetry, border unevenness, color distribution, diameter, and evolution (ABCDE.) But it also looks for lesion saliency, a comparison of the lesions on the skin of one individual to identify the “ugly ducklings” that stand out from the rest.
They trained the system using 20,388 wide-field images from 133 patients at the Hospital Gregorio Marañón in Madrid, as well as publicly available images. The images were taken with a variety of consumer-grade cameras, about half of them nondermoscopy, and included backgrounds, skin edges, bare skin sections, nonsuspicious pigmented lesions, and suspicious pigmented lesions. The lesions in the images were visually classified by a consensus of three board-certified dermatologists.
Once they trained the system, the researchers tested it on another 6,796 images from the same patients, using the dermatologists’ classification as the gold standard. The system distinguished the suspicious lesions with 90.3% sensitivity (true positive), 89.9% specificity (true negative), and 86.56% accuracy.
Dr. Soenksen said he could envision photos acquired for screening in three scenarios. First, people could photograph themselves, or someone else at their homes could photograph them. These photos could even include whole nude bodies.
Second, clinicians could photograph patients’ body parts during medical visits for other purposes. “It makes sense to do these evaluations in the point of care where a referral can actually happen, like the primary care office,” said Dr. Soenksen.
Third, photos could be taken at places where people show up in bathing suits.
In each scenario, the system would then tell patients whether any lesions needed evaluation by a dermatologist.
To ensure privacy, Dr. Soenksen envisions using devices that do not transmit all the data to the cloud but instead do at least some of the calculations on their own. High-end smartphones have sufficient computing capacity for that, he said.
In their next phase of this work, the researchers would like to test the system on more skin of color cases and in more varied conditions, said Dr. Soenksen. And they would like to put it through randomized clinical trials, potentially using biopsies to validate the results.
That’s a key step, said Veronica Rotemberg, MD, PhD, director of the dermatology imaging informatics program at Memorial Sloan Kettering Cancer Center, New York.
“Usually when we think about melanoma, we think of histology as the gold standard, or specific subtypes of melanoma as a gold standard,” she said in an interview.
The technology also raises the question of excessive screening, she said. “Identifying the ugly duckling could be extremely important in finding more melanoma,” she said. “But in a patient who doesn’t have melanoma, it could lead to a lot of unnecessary biopsies.”
The sheer number of referrals generated by such a system could overwhelm the dermatologists assigned to follow up on them, she added.
Still, Dr. Rotemberg said, the study is “a good proof of concept.” Ugly duckling analysis is a very active area of AI research with thousands of teams of researchers worldwide working on systems similar to this one, she added. “I’m so excited for the authors.”
Neither Dr. Soenksen nor Dr. Rotemberg disclosed any relevant financial interests.
.
The system could use photographs of large areas of patients’ bodies taken with ordinary cameras in primary care or by the patients themselves to screen for early-stage melanoma, said Luis R. Soenksen, PhD, a postdoctoral associate and venture builder at Massachusetts Institute of Technology in Cambridge, Mass.
“We believe we’re providing technology for that to happen at a massive scale, which is what is needed to reduce mortality rates,” he said in an interview.
He and his colleagues published their findings in Science Translational Medicine.
Diagnosing skin lesions has already proved one of the most promising medical applications of AI. In a 2017 paper, researchers reported that a deep neural network had classified skin lesions more accurately than did dermatologists. But so far, most such programs depend on experts to preselect the lesions worthy of analysis. And they use images from dermoscopy or single-lesion near-field photography.
Dr. Soenksen and colleagues wanted a system that could use a variety of cameras such as those in smartphones under a variety of conditions to assess lesions over wide areas of anatomy.
So they programmed their convolutional neural network to simultaneously use two approaches for screening lesions. Like the earlier systems, theirs looks for characteristics of individual lesions, such as asymmetry, border unevenness, color distribution, diameter, and evolution (ABCDE.) But it also looks for lesion saliency, a comparison of the lesions on the skin of one individual to identify the “ugly ducklings” that stand out from the rest.
They trained the system using 20,388 wide-field images from 133 patients at the Hospital Gregorio Marañón in Madrid, as well as publicly available images. The images were taken with a variety of consumer-grade cameras, about half of them nondermoscopy, and included backgrounds, skin edges, bare skin sections, nonsuspicious pigmented lesions, and suspicious pigmented lesions. The lesions in the images were visually classified by a consensus of three board-certified dermatologists.
Once they trained the system, the researchers tested it on another 6,796 images from the same patients, using the dermatologists’ classification as the gold standard. The system distinguished the suspicious lesions with 90.3% sensitivity (true positive), 89.9% specificity (true negative), and 86.56% accuracy.
Dr. Soenksen said he could envision photos acquired for screening in three scenarios. First, people could photograph themselves, or someone else at their homes could photograph them. These photos could even include whole nude bodies.
Second, clinicians could photograph patients’ body parts during medical visits for other purposes. “It makes sense to do these evaluations in the point of care where a referral can actually happen, like the primary care office,” said Dr. Soenksen.
Third, photos could be taken at places where people show up in bathing suits.
In each scenario, the system would then tell patients whether any lesions needed evaluation by a dermatologist.
To ensure privacy, Dr. Soenksen envisions using devices that do not transmit all the data to the cloud but instead do at least some of the calculations on their own. High-end smartphones have sufficient computing capacity for that, he said.
In their next phase of this work, the researchers would like to test the system on more skin of color cases and in more varied conditions, said Dr. Soenksen. And they would like to put it through randomized clinical trials, potentially using biopsies to validate the results.
That’s a key step, said Veronica Rotemberg, MD, PhD, director of the dermatology imaging informatics program at Memorial Sloan Kettering Cancer Center, New York.
“Usually when we think about melanoma, we think of histology as the gold standard, or specific subtypes of melanoma as a gold standard,” she said in an interview.
The technology also raises the question of excessive screening, she said. “Identifying the ugly duckling could be extremely important in finding more melanoma,” she said. “But in a patient who doesn’t have melanoma, it could lead to a lot of unnecessary biopsies.”
The sheer number of referrals generated by such a system could overwhelm the dermatologists assigned to follow up on them, she added.
Still, Dr. Rotemberg said, the study is “a good proof of concept.” Ugly duckling analysis is a very active area of AI research with thousands of teams of researchers worldwide working on systems similar to this one, she added. “I’m so excited for the authors.”
Neither Dr. Soenksen nor Dr. Rotemberg disclosed any relevant financial interests.
How has the pandemic affected rural and urban cancer patients?
Research has shown that, compared with their urban counterparts, rural cancer patients have higher cancer-related mortality and other negative treatment outcomes.
Among other explanations, the disparity has been attributed to lower education and income levels, medical and behavioral risk factors, differences in health literacy, and lower confidence in the medical system among rural residents (JCO Oncol Pract. 2020 Jul;16(7):422-30).
A new survey has provided some insight into how the COVID-19 pandemic has impacted rural and urban cancer patients differently.
The survey showed that urban patients were more likely to report changes to their daily lives, thought themselves more likely to become infected with SARS-CoV-2, and were more likely to take measures to mitigate the risk of infection. However, there were no major differences between urban and rural patients with regard to changes in social interaction.
Bailee Daniels of the University of Utah in Salt Lake City, presented these results at the AACR Virtual Meeting: COVID-19 and Cancer (Abstract S04-03).
The COVID-19 and Oncology Patient Experience Consortium
Ms. Daniels explained that the COVID-19 and Oncology Patient Experience (COPES) Consortium was created to investigate various aspects of the patient experience during the pandemic. Three cancer centers – Moffitt Cancer Center, Huntsman Cancer Institute, and the Sylvester Comprehensive Cancer Center – participate in COPES.
At Huntsman, investigators studied social and health behaviors of cancer patients to assess whether there was a difference between those from rural and urban areas. The researchers looked at the impact of the pandemic on psychosocial outcomes, preventive measures patients implemented, and their perceptions of the risk of SARS-CoV-2 infection.
The team’s hypothesis was that rural patients might be more vulnerable than urban patients to the effects of social isolation, emotional distress, and health-adverse behaviors, but the investigators noted that there has been no prior research on the topic.
Assessing behaviors, attitudes, and outcomes
Between August and September 2020, the researchers surveyed 1,328 adult cancer patients who had visited Huntsman in the previous 4 years and who were enrolled in Huntsman’s Total Cancer Care or Precision Exercise Prescription studies.
Patients completed questionnaires that encompassed demographic and clinical factors, employment status, health behaviors, and infection preventive measures. Questionnaires were provided in electronic, paper, or phone-based formats. Information regarding age, race, ethnicity, and tumor stage was abstracted from Huntsman’s electronic health record.
Modifications in daily life and social interaction were assessed on a 5-point scale. Changes in exercise habits and alcohol consumption were assessed on a 3-point scale. Infection mitigation measures (the use of face masks and hand sanitizer) and perceptions about the likelihood of SARS-CoV-2 infection were measured.
The rural-urban community area codes system, which classifies U.S. census tracts by measures of population density, urbanization, and daily commuting, was utilized to categorize patients into rural and urban residences.
Characteristics of urban and rural cancer patients
There were 997 urban and 331 rural participants. The mean age was 60.1 years in the urban population and 62.6 years in the rural population (P = .01). There were no urban-rural differences in sex, ethnicity, cancer stage, or body mass index.
More urban than rural participants were employed full- or part-time (45% vs. 37%; P = .045). The rural counties had more patients who were not currently employed, primarily due to retirement (77% vs. 69% urban; P < .001).
“No health insurance coverage” was reported by 2% of urban and 4% of rural participants (P = .009), and 85% of all patients reported “good” to “excellent” overall health. Cancer patients in rural counties were significantly more likely to have ever smoked (37% vs. 25% urban; P = .001). In addition, alcohol consumption in the previous year was higher in rural patients. “Every day to less than once monthly” alcohol usage was reported by 44% of urban and 60% of rural patients (P < .001).
Changes in daily life and health-related behavior during the pandemic
Urban patients were more likely to report changes in their daily lives due to the pandemic. Specifically, 35% of urban patients and 26% of rural patients said the pandemic had changed their daily life “a lot” (P = .001).
However, there were no major differences between urban and rural patients when it came to changes in social interaction in the past month or feeling lonely in the past month (P = .45 and P = .88, respectively). Similarly, there were no significant differences for changes in alcohol consumption between the groups (P = .90).
Changes in exercise habits due to the pandemic were more common among patients in urban counties (51% vs. 39% rural; P < .001), though similar percentages of patients reported exercising less (44% urban vs. 45% rural) or more frequently (24% urban vs. 20% rural).
In terms of infection mitigation measures, urban patients were more likely to use face masks “very often” (83% vs. 66% rural; P < .001), while hand sanitizer was used “very often” among 66% of urban and 57% of rural participants (P = .05).
Urban participants were more likely than were their rural counterparts to think themselves “somewhat” or “very” likely to develop COVID-19 (22% vs. 14%; P = .04).
It might be short-sighted for oncology and public health specialists to be dismissive of differences in infection mitigation behaviors and perceptions of vulnerability to SARS-CoV-2 infection. Those behaviors and perceptions of risk could lead to lower vaccination rates in rural areas. If that occurs, there would be major negative consequences for the long-term health of rural communities and their medically vulnerable residents.
Future directions
Although the first 6 months of the COVID-19 pandemic had disparate effects on cancer patients living in rural and urban counties, the reasons for the disparities are complex and not easily explained by this study.
It is possible that sequential administration of the survey during the pandemic would have uncovered greater variances in attitude and health-related behaviors.
As Ms. Daniels noted, when the survey was performed, Utah had not experienced a high frequency of COVID-19 cases. Furthermore, different levels of restrictions were implemented on a county-by-county basis, potentially influencing patients’ behaviors, psychosocial adjustment, and perceptions of risk.
In addition, there may have been differences in unmeasured endpoints (infection rates, medical care utilization via telemedicine, hospitalization rates, late effects, and mortality) between the urban and rural populations.
As the investigators concluded, further research is needed to better characterize the pandemic’s short- and long-term effects on cancer patients in rural and urban settings and appropriate interventions. Such studies may yield insights into the various facets of the well-documented “rural health gap” in cancer outcomes and interventions that could narrow the gap in spheres beyond the COVID-19 pandemic.
Ms. Daniels reported having no relevant disclosures.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Research has shown that, compared with their urban counterparts, rural cancer patients have higher cancer-related mortality and other negative treatment outcomes.
Among other explanations, the disparity has been attributed to lower education and income levels, medical and behavioral risk factors, differences in health literacy, and lower confidence in the medical system among rural residents (JCO Oncol Pract. 2020 Jul;16(7):422-30).
A new survey has provided some insight into how the COVID-19 pandemic has impacted rural and urban cancer patients differently.
The survey showed that urban patients were more likely to report changes to their daily lives, thought themselves more likely to become infected with SARS-CoV-2, and were more likely to take measures to mitigate the risk of infection. However, there were no major differences between urban and rural patients with regard to changes in social interaction.
Bailee Daniels of the University of Utah in Salt Lake City, presented these results at the AACR Virtual Meeting: COVID-19 and Cancer (Abstract S04-03).
The COVID-19 and Oncology Patient Experience Consortium
Ms. Daniels explained that the COVID-19 and Oncology Patient Experience (COPES) Consortium was created to investigate various aspects of the patient experience during the pandemic. Three cancer centers – Moffitt Cancer Center, Huntsman Cancer Institute, and the Sylvester Comprehensive Cancer Center – participate in COPES.
At Huntsman, investigators studied social and health behaviors of cancer patients to assess whether there was a difference between those from rural and urban areas. The researchers looked at the impact of the pandemic on psychosocial outcomes, preventive measures patients implemented, and their perceptions of the risk of SARS-CoV-2 infection.
The team’s hypothesis was that rural patients might be more vulnerable than urban patients to the effects of social isolation, emotional distress, and health-adverse behaviors, but the investigators noted that there has been no prior research on the topic.
Assessing behaviors, attitudes, and outcomes
Between August and September 2020, the researchers surveyed 1,328 adult cancer patients who had visited Huntsman in the previous 4 years and who were enrolled in Huntsman’s Total Cancer Care or Precision Exercise Prescription studies.
Patients completed questionnaires that encompassed demographic and clinical factors, employment status, health behaviors, and infection preventive measures. Questionnaires were provided in electronic, paper, or phone-based formats. Information regarding age, race, ethnicity, and tumor stage was abstracted from Huntsman’s electronic health record.
Modifications in daily life and social interaction were assessed on a 5-point scale. Changes in exercise habits and alcohol consumption were assessed on a 3-point scale. Infection mitigation measures (the use of face masks and hand sanitizer) and perceptions about the likelihood of SARS-CoV-2 infection were measured.
The rural-urban community area codes system, which classifies U.S. census tracts by measures of population density, urbanization, and daily commuting, was utilized to categorize patients into rural and urban residences.
Characteristics of urban and rural cancer patients
There were 997 urban and 331 rural participants. The mean age was 60.1 years in the urban population and 62.6 years in the rural population (P = .01). There were no urban-rural differences in sex, ethnicity, cancer stage, or body mass index.
More urban than rural participants were employed full- or part-time (45% vs. 37%; P = .045). The rural counties had more patients who were not currently employed, primarily due to retirement (77% vs. 69% urban; P < .001).
“No health insurance coverage” was reported by 2% of urban and 4% of rural participants (P = .009), and 85% of all patients reported “good” to “excellent” overall health. Cancer patients in rural counties were significantly more likely to have ever smoked (37% vs. 25% urban; P = .001). In addition, alcohol consumption in the previous year was higher in rural patients. “Every day to less than once monthly” alcohol usage was reported by 44% of urban and 60% of rural patients (P < .001).
Changes in daily life and health-related behavior during the pandemic
Urban patients were more likely to report changes in their daily lives due to the pandemic. Specifically, 35% of urban patients and 26% of rural patients said the pandemic had changed their daily life “a lot” (P = .001).
However, there were no major differences between urban and rural patients when it came to changes in social interaction in the past month or feeling lonely in the past month (P = .45 and P = .88, respectively). Similarly, there were no significant differences for changes in alcohol consumption between the groups (P = .90).
Changes in exercise habits due to the pandemic were more common among patients in urban counties (51% vs. 39% rural; P < .001), though similar percentages of patients reported exercising less (44% urban vs. 45% rural) or more frequently (24% urban vs. 20% rural).
In terms of infection mitigation measures, urban patients were more likely to use face masks “very often” (83% vs. 66% rural; P < .001), while hand sanitizer was used “very often” among 66% of urban and 57% of rural participants (P = .05).
Urban participants were more likely than were their rural counterparts to think themselves “somewhat” or “very” likely to develop COVID-19 (22% vs. 14%; P = .04).
It might be short-sighted for oncology and public health specialists to be dismissive of differences in infection mitigation behaviors and perceptions of vulnerability to SARS-CoV-2 infection. Those behaviors and perceptions of risk could lead to lower vaccination rates in rural areas. If that occurs, there would be major negative consequences for the long-term health of rural communities and their medically vulnerable residents.
Future directions
Although the first 6 months of the COVID-19 pandemic had disparate effects on cancer patients living in rural and urban counties, the reasons for the disparities are complex and not easily explained by this study.
It is possible that sequential administration of the survey during the pandemic would have uncovered greater variances in attitude and health-related behaviors.
As Ms. Daniels noted, when the survey was performed, Utah had not experienced a high frequency of COVID-19 cases. Furthermore, different levels of restrictions were implemented on a county-by-county basis, potentially influencing patients’ behaviors, psychosocial adjustment, and perceptions of risk.
In addition, there may have been differences in unmeasured endpoints (infection rates, medical care utilization via telemedicine, hospitalization rates, late effects, and mortality) between the urban and rural populations.
As the investigators concluded, further research is needed to better characterize the pandemic’s short- and long-term effects on cancer patients in rural and urban settings and appropriate interventions. Such studies may yield insights into the various facets of the well-documented “rural health gap” in cancer outcomes and interventions that could narrow the gap in spheres beyond the COVID-19 pandemic.
Ms. Daniels reported having no relevant disclosures.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Research has shown that, compared with their urban counterparts, rural cancer patients have higher cancer-related mortality and other negative treatment outcomes.
Among other explanations, the disparity has been attributed to lower education and income levels, medical and behavioral risk factors, differences in health literacy, and lower confidence in the medical system among rural residents (JCO Oncol Pract. 2020 Jul;16(7):422-30).
A new survey has provided some insight into how the COVID-19 pandemic has impacted rural and urban cancer patients differently.
The survey showed that urban patients were more likely to report changes to their daily lives, thought themselves more likely to become infected with SARS-CoV-2, and were more likely to take measures to mitigate the risk of infection. However, there were no major differences between urban and rural patients with regard to changes in social interaction.
Bailee Daniels of the University of Utah in Salt Lake City, presented these results at the AACR Virtual Meeting: COVID-19 and Cancer (Abstract S04-03).
The COVID-19 and Oncology Patient Experience Consortium
Ms. Daniels explained that the COVID-19 and Oncology Patient Experience (COPES) Consortium was created to investigate various aspects of the patient experience during the pandemic. Three cancer centers – Moffitt Cancer Center, Huntsman Cancer Institute, and the Sylvester Comprehensive Cancer Center – participate in COPES.
At Huntsman, investigators studied social and health behaviors of cancer patients to assess whether there was a difference between those from rural and urban areas. The researchers looked at the impact of the pandemic on psychosocial outcomes, preventive measures patients implemented, and their perceptions of the risk of SARS-CoV-2 infection.
The team’s hypothesis was that rural patients might be more vulnerable than urban patients to the effects of social isolation, emotional distress, and health-adverse behaviors, but the investigators noted that there has been no prior research on the topic.
Assessing behaviors, attitudes, and outcomes
Between August and September 2020, the researchers surveyed 1,328 adult cancer patients who had visited Huntsman in the previous 4 years and who were enrolled in Huntsman’s Total Cancer Care or Precision Exercise Prescription studies.
Patients completed questionnaires that encompassed demographic and clinical factors, employment status, health behaviors, and infection preventive measures. Questionnaires were provided in electronic, paper, or phone-based formats. Information regarding age, race, ethnicity, and tumor stage was abstracted from Huntsman’s electronic health record.
Modifications in daily life and social interaction were assessed on a 5-point scale. Changes in exercise habits and alcohol consumption were assessed on a 3-point scale. Infection mitigation measures (the use of face masks and hand sanitizer) and perceptions about the likelihood of SARS-CoV-2 infection were measured.
The rural-urban community area codes system, which classifies U.S. census tracts by measures of population density, urbanization, and daily commuting, was utilized to categorize patients into rural and urban residences.
Characteristics of urban and rural cancer patients
There were 997 urban and 331 rural participants. The mean age was 60.1 years in the urban population and 62.6 years in the rural population (P = .01). There were no urban-rural differences in sex, ethnicity, cancer stage, or body mass index.
More urban than rural participants were employed full- or part-time (45% vs. 37%; P = .045). The rural counties had more patients who were not currently employed, primarily due to retirement (77% vs. 69% urban; P < .001).
“No health insurance coverage” was reported by 2% of urban and 4% of rural participants (P = .009), and 85% of all patients reported “good” to “excellent” overall health. Cancer patients in rural counties were significantly more likely to have ever smoked (37% vs. 25% urban; P = .001). In addition, alcohol consumption in the previous year was higher in rural patients. “Every day to less than once monthly” alcohol usage was reported by 44% of urban and 60% of rural patients (P < .001).
Changes in daily life and health-related behavior during the pandemic
Urban patients were more likely to report changes in their daily lives due to the pandemic. Specifically, 35% of urban patients and 26% of rural patients said the pandemic had changed their daily life “a lot” (P = .001).
However, there were no major differences between urban and rural patients when it came to changes in social interaction in the past month or feeling lonely in the past month (P = .45 and P = .88, respectively). Similarly, there were no significant differences for changes in alcohol consumption between the groups (P = .90).
Changes in exercise habits due to the pandemic were more common among patients in urban counties (51% vs. 39% rural; P < .001), though similar percentages of patients reported exercising less (44% urban vs. 45% rural) or more frequently (24% urban vs. 20% rural).
In terms of infection mitigation measures, urban patients were more likely to use face masks “very often” (83% vs. 66% rural; P < .001), while hand sanitizer was used “very often” among 66% of urban and 57% of rural participants (P = .05).
Urban participants were more likely than were their rural counterparts to think themselves “somewhat” or “very” likely to develop COVID-19 (22% vs. 14%; P = .04).
It might be short-sighted for oncology and public health specialists to be dismissive of differences in infection mitigation behaviors and perceptions of vulnerability to SARS-CoV-2 infection. Those behaviors and perceptions of risk could lead to lower vaccination rates in rural areas. If that occurs, there would be major negative consequences for the long-term health of rural communities and their medically vulnerable residents.
Future directions
Although the first 6 months of the COVID-19 pandemic had disparate effects on cancer patients living in rural and urban counties, the reasons for the disparities are complex and not easily explained by this study.
It is possible that sequential administration of the survey during the pandemic would have uncovered greater variances in attitude and health-related behaviors.
As Ms. Daniels noted, when the survey was performed, Utah had not experienced a high frequency of COVID-19 cases. Furthermore, different levels of restrictions were implemented on a county-by-county basis, potentially influencing patients’ behaviors, psychosocial adjustment, and perceptions of risk.
In addition, there may have been differences in unmeasured endpoints (infection rates, medical care utilization via telemedicine, hospitalization rates, late effects, and mortality) between the urban and rural populations.
As the investigators concluded, further research is needed to better characterize the pandemic’s short- and long-term effects on cancer patients in rural and urban settings and appropriate interventions. Such studies may yield insights into the various facets of the well-documented “rural health gap” in cancer outcomes and interventions that could narrow the gap in spheres beyond the COVID-19 pandemic.
Ms. Daniels reported having no relevant disclosures.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
FROM AACR: COVID-19 AND CANCER 2021
Checkpoint inhibitors’ ‘big picture’ safety shown with preexisting autoimmune diseases
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
FROM ANNALS OF INTERNAL MEDICINE
Neoadjuvant immunotherapy shows promise in stage III melanoma
The next dramatic , John M. Kirkwood, MD, predicted at a virtual forum on cutaneous malignancies jointly presented by the Postgraduate Institute for Medicine and Global Academy for Medical Education.
These agents have already demonstrated profound efficacy, first in stage IV metastatic disease and more recently as adjuvant therapy for resected stage III melanoma. Now, there is a great interest in learning whether by prescribing them preoperatively, patients might reduce their risk of advancing to metastatic disease. And neoadjuvant therapy offers an extremely attractive feature: It yields results in an accelerated fashion.
“The major problem with postoperative adjuvant trials in melanoma since 1984 is the long time to maturity. Many of us don’t want to wait the full 9 or 10 years for a full-bore, phase 3 adjuvant trial in stage III melanoma to mature,” explained Dr. Kirkwood, professor of medicine, dermatology, and translational science and coleader of the melanoma and skin cancer program at the University of Pittsburgh. “The opportunity to treat a patient who presents with a bulky lymph node, has a biopsy, and then can be treated for 3 or 6 weeks or sometimes even longer periods with a therapy that’s promising allows us to ask what’s going on in the tumor tissue, what’s going on in the clinical response at 3 or 6 weeks, and if there’s pathological complete or near-complete response under the microscope.”
Because pathological complete response is a strong predictor of relapse-free survival, this neoadjuvant-forward therapeutic strategy has the potential to provide patients and their physicians with an early forecast of likely clinical outcome only 4-6 weeks into treatment. Also, there is both preclinical and clinical evidence that neoadjuvant therapy may offer a survival advantage over adjuvant therapy, perhaps as a result of early treatment of micrometastatic disease. Another benefit of neoadjuvant therapy for melanoma is the resultant tumor shrinkage, which can permit less extensive surgery.
Dr. Kirkwood highlighted a phase 2 clinical trial conducted at the University of Pittsburgh to illustrate the potential of neoadjuvant therapy in melanoma. The ongoing single-arm study includes 32 patients with stage IIIB or IIIC resectable melanoma along with accessible tumor for biopsy and intratumoral injections of CMP-001, a toll-like receptor 9 agonist. According to the Eighth Edition of the American Joint Committee on Cancer staging manual, stage IIIB melanoma has a 10-year mortality of 23%, and stage IIIC disease has 40%.
CMP-001 triggers type 1 interferon production through activation of plasmacytoid dendritic cells. The resultant inflammatory response draws T cells into the tumor to enhance the response to immunotherapy, which in this study was nivolumab (Opdivo), a human programmed death ligand 1 (PD-L1)–blocking antibody. The neoadjuvant regimen consisted of seven once-weekly intratumoral injections of CMP-001, plus three 240-mg doses of nivolumab given at 2-week intervals. This was followed by resection, then 1 year of adjuvant therapy with nivolumab at 480 mg every 4 weeks and intratumoral CMP-001 every 4 weeks.
In an interim analysis, a major pathologic response occurred in an impressive 15 of 21 patients (71%) after 6 weeks of neoadjuvant therapy. Thirteen of the 15 had a pathologic complete response. Encouragingly, no one with a pathologic complete or near-complete response has relapsed to date.
“A pathologic complete response or near-complete response with neoadjuvant therapy appears to be a biomarker of durable disease control and is associated with excellent outcomes,” Dr. Kirkwood observed, adding that the Pittsburgh experience has been mirrored in reports from the Netherlands, Australia, and University of Texas M.D. Anderson Cancer Center, Houston, involving other neoadjuvant agents.
Other potential early biomarkers of favorable outcome with neoadjuvant therapy include CD8+ T cells in the tumor at baseline, tumor mutational burden, T-cell clonality, and a T-cell–inflamed gene-expression profile.
There were no dose-limiting toxicities or delays in surgery related to the neoadjuvant treatment.
Of note, imaging often inaccurately showed only a partial response in patients who actually had a pathologic complete response, meaning totally devoid of tumor, Dr. Kirkwood said.
Corroboration of these findings is planned in the national multicenter ECOG-ACRIN neoadjuvant trial EA6194.
“Consider referring to this trial any patients who present with bulky nodal disease for whom a treatment assessment at 4-6 weeks is desired in order to predict what the outcome may be,” he suggested.
Dr. Kirkwood reported receiving research grants from Amgen, BMS, Castle Biosciences, Checkmate, Immunocore, Iovance, and Novartis and serving as a consultant to a handful of companies.
Global Academy for Medical Education and this news organization are owned by the same company.
The next dramatic , John M. Kirkwood, MD, predicted at a virtual forum on cutaneous malignancies jointly presented by the Postgraduate Institute for Medicine and Global Academy for Medical Education.
These agents have already demonstrated profound efficacy, first in stage IV metastatic disease and more recently as adjuvant therapy for resected stage III melanoma. Now, there is a great interest in learning whether by prescribing them preoperatively, patients might reduce their risk of advancing to metastatic disease. And neoadjuvant therapy offers an extremely attractive feature: It yields results in an accelerated fashion.
“The major problem with postoperative adjuvant trials in melanoma since 1984 is the long time to maturity. Many of us don’t want to wait the full 9 or 10 years for a full-bore, phase 3 adjuvant trial in stage III melanoma to mature,” explained Dr. Kirkwood, professor of medicine, dermatology, and translational science and coleader of the melanoma and skin cancer program at the University of Pittsburgh. “The opportunity to treat a patient who presents with a bulky lymph node, has a biopsy, and then can be treated for 3 or 6 weeks or sometimes even longer periods with a therapy that’s promising allows us to ask what’s going on in the tumor tissue, what’s going on in the clinical response at 3 or 6 weeks, and if there’s pathological complete or near-complete response under the microscope.”
Because pathological complete response is a strong predictor of relapse-free survival, this neoadjuvant-forward therapeutic strategy has the potential to provide patients and their physicians with an early forecast of likely clinical outcome only 4-6 weeks into treatment. Also, there is both preclinical and clinical evidence that neoadjuvant therapy may offer a survival advantage over adjuvant therapy, perhaps as a result of early treatment of micrometastatic disease. Another benefit of neoadjuvant therapy for melanoma is the resultant tumor shrinkage, which can permit less extensive surgery.
Dr. Kirkwood highlighted a phase 2 clinical trial conducted at the University of Pittsburgh to illustrate the potential of neoadjuvant therapy in melanoma. The ongoing single-arm study includes 32 patients with stage IIIB or IIIC resectable melanoma along with accessible tumor for biopsy and intratumoral injections of CMP-001, a toll-like receptor 9 agonist. According to the Eighth Edition of the American Joint Committee on Cancer staging manual, stage IIIB melanoma has a 10-year mortality of 23%, and stage IIIC disease has 40%.
CMP-001 triggers type 1 interferon production through activation of plasmacytoid dendritic cells. The resultant inflammatory response draws T cells into the tumor to enhance the response to immunotherapy, which in this study was nivolumab (Opdivo), a human programmed death ligand 1 (PD-L1)–blocking antibody. The neoadjuvant regimen consisted of seven once-weekly intratumoral injections of CMP-001, plus three 240-mg doses of nivolumab given at 2-week intervals. This was followed by resection, then 1 year of adjuvant therapy with nivolumab at 480 mg every 4 weeks and intratumoral CMP-001 every 4 weeks.
In an interim analysis, a major pathologic response occurred in an impressive 15 of 21 patients (71%) after 6 weeks of neoadjuvant therapy. Thirteen of the 15 had a pathologic complete response. Encouragingly, no one with a pathologic complete or near-complete response has relapsed to date.
“A pathologic complete response or near-complete response with neoadjuvant therapy appears to be a biomarker of durable disease control and is associated with excellent outcomes,” Dr. Kirkwood observed, adding that the Pittsburgh experience has been mirrored in reports from the Netherlands, Australia, and University of Texas M.D. Anderson Cancer Center, Houston, involving other neoadjuvant agents.
Other potential early biomarkers of favorable outcome with neoadjuvant therapy include CD8+ T cells in the tumor at baseline, tumor mutational burden, T-cell clonality, and a T-cell–inflamed gene-expression profile.
There were no dose-limiting toxicities or delays in surgery related to the neoadjuvant treatment.
Of note, imaging often inaccurately showed only a partial response in patients who actually had a pathologic complete response, meaning totally devoid of tumor, Dr. Kirkwood said.
Corroboration of these findings is planned in the national multicenter ECOG-ACRIN neoadjuvant trial EA6194.
“Consider referring to this trial any patients who present with bulky nodal disease for whom a treatment assessment at 4-6 weeks is desired in order to predict what the outcome may be,” he suggested.
Dr. Kirkwood reported receiving research grants from Amgen, BMS, Castle Biosciences, Checkmate, Immunocore, Iovance, and Novartis and serving as a consultant to a handful of companies.
Global Academy for Medical Education and this news organization are owned by the same company.
The next dramatic , John M. Kirkwood, MD, predicted at a virtual forum on cutaneous malignancies jointly presented by the Postgraduate Institute for Medicine and Global Academy for Medical Education.
These agents have already demonstrated profound efficacy, first in stage IV metastatic disease and more recently as adjuvant therapy for resected stage III melanoma. Now, there is a great interest in learning whether by prescribing them preoperatively, patients might reduce their risk of advancing to metastatic disease. And neoadjuvant therapy offers an extremely attractive feature: It yields results in an accelerated fashion.
“The major problem with postoperative adjuvant trials in melanoma since 1984 is the long time to maturity. Many of us don’t want to wait the full 9 or 10 years for a full-bore, phase 3 adjuvant trial in stage III melanoma to mature,” explained Dr. Kirkwood, professor of medicine, dermatology, and translational science and coleader of the melanoma and skin cancer program at the University of Pittsburgh. “The opportunity to treat a patient who presents with a bulky lymph node, has a biopsy, and then can be treated for 3 or 6 weeks or sometimes even longer periods with a therapy that’s promising allows us to ask what’s going on in the tumor tissue, what’s going on in the clinical response at 3 or 6 weeks, and if there’s pathological complete or near-complete response under the microscope.”
Because pathological complete response is a strong predictor of relapse-free survival, this neoadjuvant-forward therapeutic strategy has the potential to provide patients and their physicians with an early forecast of likely clinical outcome only 4-6 weeks into treatment. Also, there is both preclinical and clinical evidence that neoadjuvant therapy may offer a survival advantage over adjuvant therapy, perhaps as a result of early treatment of micrometastatic disease. Another benefit of neoadjuvant therapy for melanoma is the resultant tumor shrinkage, which can permit less extensive surgery.
Dr. Kirkwood highlighted a phase 2 clinical trial conducted at the University of Pittsburgh to illustrate the potential of neoadjuvant therapy in melanoma. The ongoing single-arm study includes 32 patients with stage IIIB or IIIC resectable melanoma along with accessible tumor for biopsy and intratumoral injections of CMP-001, a toll-like receptor 9 agonist. According to the Eighth Edition of the American Joint Committee on Cancer staging manual, stage IIIB melanoma has a 10-year mortality of 23%, and stage IIIC disease has 40%.
CMP-001 triggers type 1 interferon production through activation of plasmacytoid dendritic cells. The resultant inflammatory response draws T cells into the tumor to enhance the response to immunotherapy, which in this study was nivolumab (Opdivo), a human programmed death ligand 1 (PD-L1)–blocking antibody. The neoadjuvant regimen consisted of seven once-weekly intratumoral injections of CMP-001, plus three 240-mg doses of nivolumab given at 2-week intervals. This was followed by resection, then 1 year of adjuvant therapy with nivolumab at 480 mg every 4 weeks and intratumoral CMP-001 every 4 weeks.
In an interim analysis, a major pathologic response occurred in an impressive 15 of 21 patients (71%) after 6 weeks of neoadjuvant therapy. Thirteen of the 15 had a pathologic complete response. Encouragingly, no one with a pathologic complete or near-complete response has relapsed to date.
“A pathologic complete response or near-complete response with neoadjuvant therapy appears to be a biomarker of durable disease control and is associated with excellent outcomes,” Dr. Kirkwood observed, adding that the Pittsburgh experience has been mirrored in reports from the Netherlands, Australia, and University of Texas M.D. Anderson Cancer Center, Houston, involving other neoadjuvant agents.
Other potential early biomarkers of favorable outcome with neoadjuvant therapy include CD8+ T cells in the tumor at baseline, tumor mutational burden, T-cell clonality, and a T-cell–inflamed gene-expression profile.
There were no dose-limiting toxicities or delays in surgery related to the neoadjuvant treatment.
Of note, imaging often inaccurately showed only a partial response in patients who actually had a pathologic complete response, meaning totally devoid of tumor, Dr. Kirkwood said.
Corroboration of these findings is planned in the national multicenter ECOG-ACRIN neoadjuvant trial EA6194.
“Consider referring to this trial any patients who present with bulky nodal disease for whom a treatment assessment at 4-6 weeks is desired in order to predict what the outcome may be,” he suggested.
Dr. Kirkwood reported receiving research grants from Amgen, BMS, Castle Biosciences, Checkmate, Immunocore, Iovance, and Novartis and serving as a consultant to a handful of companies.
Global Academy for Medical Education and this news organization are owned by the same company.
FROM THE CUTANEOUS MALIGNANCIES FORUM
Managing cancer outpatients during the pandemic: Tips from MSKCC
“We’ve tried a lot of new things to ensure optimal care for our patients,” said Tiffany A. Traina, MD, of Memorial Sloan Kettering Cancer Center (MSKCC) in New York. “We need to effectively utilize all resources at our disposal to keep in touch with our patients during this time.”
Dr. Traina described the approach to outpatient management used at MSKCC during a presentation at the AACR Virtual Meeting: COVID-19 and Cancer.
Four guiding principles
MSKCC has established four guiding principles on how to manage cancer patients during the pandemic: openness, safety, technology, and staffing.
Openness ensures that decisions are guided by clinical priorities to provide optimal patient care and allow for prioritization of clinical research and education, Dr. Traina said.
The safety of patients and staff is of the utmost importance, she added. To ensure safety in the context of outpatient care, several operational levers were developed, including COVID surge planning, universal masking and personal protective equipment guidelines, remote work, clinical levers, and new dashboards and communications.
Dr. Traina said data analytics and dashboards have been key technological tools used to support evidence-based decision-making and deliver care remotely for patients during the pandemic.
Staffing resources have also shifted to support demand at different health system locations.
Screening, cohorting, and telemedicine
One measure MSKCC adopted is the MSK Engage Questionnaire, a COVID-19 screening questionnaire assigned to every patient with a scheduled outpatient visit. After completing the questionnaire, patients receive a response denoting whether they need to come into the outpatient setting.
On the staffing side, clinic coordinators prepare appointments accordingly, based on the risk level for each patient.
“We also try to cohort COVID-positive patients into particular areas within the outpatient setting,” Dr. Traina explained. “In addition, we control flow through ambulatory care locations by having separate patient entrances and use other tools to make flow as efficient as possible.”
On the technology side, interactive dashboards are being used to model traffic through different buildings.
“These data and analytics are useful for operational engineering, answering questions such as (1) Are there backups in chemotherapy? and (2) Are patients seeing one particular physician?” Dr. Traina explained. “One important key takeaway is the importance of frequently communicating simple messages through multiple mechanisms, including signage, websites, and dedicated resources.”
Other key technological measures are leveraging telemedicine to convert inpatient appointments to a virtual setting, as well as developing and deploying a system for centralized outpatient follow-up of COVID-19-positive patients.
“We saw a 3,000% increase in telemedicine utilization from February 2020 to June 2020,” Dr. Traina reported. “In a given month, we have approximately 230,000 outpatient visits, and a substantial proportion of these are now done via telemedicine.”
Dr. Traina also noted that multiple organizations have released guidelines addressing when to resume anticancer therapy in patients who have been COVID-19 positive. Adherence is important, as unnecessary COVID-19 testing may delay cancer therapy and is not recommended.
During a live discussion, Louis P. Voigt, MD, of MSKCC, said Dr. Traina’s presentation provided “a lot of good ideas for other institutions who may be facing similar challenges.”
Dr. Traina and Dr. Voigt disclosed no conflicts of interest. No funding sources were reported.
“We’ve tried a lot of new things to ensure optimal care for our patients,” said Tiffany A. Traina, MD, of Memorial Sloan Kettering Cancer Center (MSKCC) in New York. “We need to effectively utilize all resources at our disposal to keep in touch with our patients during this time.”
Dr. Traina described the approach to outpatient management used at MSKCC during a presentation at the AACR Virtual Meeting: COVID-19 and Cancer.
Four guiding principles
MSKCC has established four guiding principles on how to manage cancer patients during the pandemic: openness, safety, technology, and staffing.
Openness ensures that decisions are guided by clinical priorities to provide optimal patient care and allow for prioritization of clinical research and education, Dr. Traina said.
The safety of patients and staff is of the utmost importance, she added. To ensure safety in the context of outpatient care, several operational levers were developed, including COVID surge planning, universal masking and personal protective equipment guidelines, remote work, clinical levers, and new dashboards and communications.
Dr. Traina said data analytics and dashboards have been key technological tools used to support evidence-based decision-making and deliver care remotely for patients during the pandemic.
Staffing resources have also shifted to support demand at different health system locations.
Screening, cohorting, and telemedicine
One measure MSKCC adopted is the MSK Engage Questionnaire, a COVID-19 screening questionnaire assigned to every patient with a scheduled outpatient visit. After completing the questionnaire, patients receive a response denoting whether they need to come into the outpatient setting.
On the staffing side, clinic coordinators prepare appointments accordingly, based on the risk level for each patient.
“We also try to cohort COVID-positive patients into particular areas within the outpatient setting,” Dr. Traina explained. “In addition, we control flow through ambulatory care locations by having separate patient entrances and use other tools to make flow as efficient as possible.”
On the technology side, interactive dashboards are being used to model traffic through different buildings.
“These data and analytics are useful for operational engineering, answering questions such as (1) Are there backups in chemotherapy? and (2) Are patients seeing one particular physician?” Dr. Traina explained. “One important key takeaway is the importance of frequently communicating simple messages through multiple mechanisms, including signage, websites, and dedicated resources.”
Other key technological measures are leveraging telemedicine to convert inpatient appointments to a virtual setting, as well as developing and deploying a system for centralized outpatient follow-up of COVID-19-positive patients.
“We saw a 3,000% increase in telemedicine utilization from February 2020 to June 2020,” Dr. Traina reported. “In a given month, we have approximately 230,000 outpatient visits, and a substantial proportion of these are now done via telemedicine.”
Dr. Traina also noted that multiple organizations have released guidelines addressing when to resume anticancer therapy in patients who have been COVID-19 positive. Adherence is important, as unnecessary COVID-19 testing may delay cancer therapy and is not recommended.
During a live discussion, Louis P. Voigt, MD, of MSKCC, said Dr. Traina’s presentation provided “a lot of good ideas for other institutions who may be facing similar challenges.”
Dr. Traina and Dr. Voigt disclosed no conflicts of interest. No funding sources were reported.
“We’ve tried a lot of new things to ensure optimal care for our patients,” said Tiffany A. Traina, MD, of Memorial Sloan Kettering Cancer Center (MSKCC) in New York. “We need to effectively utilize all resources at our disposal to keep in touch with our patients during this time.”
Dr. Traina described the approach to outpatient management used at MSKCC during a presentation at the AACR Virtual Meeting: COVID-19 and Cancer.
Four guiding principles
MSKCC has established four guiding principles on how to manage cancer patients during the pandemic: openness, safety, technology, and staffing.
Openness ensures that decisions are guided by clinical priorities to provide optimal patient care and allow for prioritization of clinical research and education, Dr. Traina said.
The safety of patients and staff is of the utmost importance, she added. To ensure safety in the context of outpatient care, several operational levers were developed, including COVID surge planning, universal masking and personal protective equipment guidelines, remote work, clinical levers, and new dashboards and communications.
Dr. Traina said data analytics and dashboards have been key technological tools used to support evidence-based decision-making and deliver care remotely for patients during the pandemic.
Staffing resources have also shifted to support demand at different health system locations.
Screening, cohorting, and telemedicine
One measure MSKCC adopted is the MSK Engage Questionnaire, a COVID-19 screening questionnaire assigned to every patient with a scheduled outpatient visit. After completing the questionnaire, patients receive a response denoting whether they need to come into the outpatient setting.
On the staffing side, clinic coordinators prepare appointments accordingly, based on the risk level for each patient.
“We also try to cohort COVID-positive patients into particular areas within the outpatient setting,” Dr. Traina explained. “In addition, we control flow through ambulatory care locations by having separate patient entrances and use other tools to make flow as efficient as possible.”
On the technology side, interactive dashboards are being used to model traffic through different buildings.
“These data and analytics are useful for operational engineering, answering questions such as (1) Are there backups in chemotherapy? and (2) Are patients seeing one particular physician?” Dr. Traina explained. “One important key takeaway is the importance of frequently communicating simple messages through multiple mechanisms, including signage, websites, and dedicated resources.”
Other key technological measures are leveraging telemedicine to convert inpatient appointments to a virtual setting, as well as developing and deploying a system for centralized outpatient follow-up of COVID-19-positive patients.
“We saw a 3,000% increase in telemedicine utilization from February 2020 to June 2020,” Dr. Traina reported. “In a given month, we have approximately 230,000 outpatient visits, and a substantial proportion of these are now done via telemedicine.”
Dr. Traina also noted that multiple organizations have released guidelines addressing when to resume anticancer therapy in patients who have been COVID-19 positive. Adherence is important, as unnecessary COVID-19 testing may delay cancer therapy and is not recommended.
During a live discussion, Louis P. Voigt, MD, of MSKCC, said Dr. Traina’s presentation provided “a lot of good ideas for other institutions who may be facing similar challenges.”
Dr. Traina and Dr. Voigt disclosed no conflicts of interest. No funding sources were reported.
FROM AACR: COVID-19 AND CANCER 2021
COVID-19 vaccination in cancer patients: NCCN outlines priorities
Vaccination timing considerations vary based on factors such as cancer and treatment type, and reasons for delaying vaccination in the general public also apply to cancer patients (recent COVID-19 exposure, for example).
In general, however, patients with cancer should be assigned to Centers for Disease Control and Prevention priority group 1 b/c and immunized when vaccination is available to them, the guidelines state. Exceptions to this recommendation include:
- Patients undergoing hematopoietic stem cell transplant or receiving engineered cellular therapy such as chimeric antigen receptor T-cell therapy. Vaccination should be delayed for at least 3 months in these patients to maximize vaccine efficacy. Caregivers of these patients, however, should be immunized when possible.
- Patients with hematologic malignancies who are receiving intensive cytotoxic chemotherapy, such as cytarabine- or anthracycline-based regimens for acute myeloid leukemia. Vaccination in these patients should be delayed until absolute neutrophil count recovery.
- Patients undergoing major surgery. Vaccination should occur at least a few days before or after surgery.
- Patients who have experienced a severe or immediate adverse reaction to any of the ingredients in the mRNA COVID-19 vaccines.
Conversely, vaccination should occur when available in patients with hematologic malignancies and marrow failure who are expected to have limited or no recovery, patients with hematologic malignancies who are on long-term maintenance therapy, and patients with solid tumors who are receiving cytotoxic chemotherapy, targeted therapy, checkpoint inhibitors and other immunotherapy, or radiotherapy.
Caregivers, household contacts, and other close contacts who are 16 years of age and older should be vaccinated whenever they are eligible.
Unique concerns in patients with cancer
The NCCN recommendations were developed to address the unique issues and concerns with respect to patients with cancer, who have an increased risk of severe illness from SARS-CoV-2 infection. But the guidelines come with a caveat: “[t]here are limited safety and efficacy data in these patients,” the NCCN emphasized in a press statement.
“Right now, there is urgent need and limited data,” Steven Pergam, MD, co-leader of the NCCN COVID-19 Vaccination Committee, said in the statement.
“Our number one goal is helping to get the vaccine to as many people as we can,” Dr. Pergam said. “That means following existing national and regional directions for prioritizing people who are more likely to face death or severe illness from COVID-19.”
Dr. Pergam, associate professor at Fred Hutchinson Cancer Research Center in Seattle, further explained that “people receiving active cancer treatment are at greater risk for worse outcomes from COVID-19, particularly if they are older and have additional comorbidities, like immunosuppression.”
NCCN’s recommendations couldn’t have come at a better time for patients with cancer, according to Nora Disis, MD, a professor at the University of Washington in Seattle.
“The NCCN’s recommendations to prioritize COVID vaccinations for cancer patients on active treatment is an important step forward in protecting our patients from the infection,” Dr. Disis said in an interview.
“Cancer patients may be at higher risk for the complications seen with infection. In addition, cancer is a disease of older people, and a good number of our patients have the comorbidities that would predict a poorer outcome if they should become sick,” Dr. Disis added. “With the correct treatment, many patients with cancer will be long-term survivors. It is important that they be protected from infection with COVID to realize their best outcome.”
Additional vaccine considerations
The NCCN recommendations also address several other issues of importance for cancer patients, including:
- Deprioritizing other vaccines. COVID-19 vaccines should take precedence over other vaccines because data on dual vaccination are lacking. The NCCN recommends waiting 14 days after COVID-19 vaccination to deliver other vaccines.
- Vaccinating clinical trial participants. Trial leads should be consulted to prevent protocol violations or exclusions.
- Decision-making in the setting of limited vaccine availability. The NCCN noted that decisions on allocation must be made in accordance with state and local vaccine guidance but suggests prioritizing appropriate patients on active treatment, those planning to start treatment, and those who have just completed treatment. Additional risk factors for these patients, as well as other factors associated with risk for adverse COVID-19 outcomes, should also be considered. These include advanced age, comorbidities, and adverse social and demographic factors such as poverty and limited health care access.
- The need for ongoing prevention measures. Vaccines have been shown to decrease the incidence of COVID-19 and related complications, but it remains unclear whether vaccines prevent infection and subsequent transmission. This means everyone should continue following prevention recommendations, such as wearing masks and avoiding crowds.
The NCCN stressed that these recommendations are “intended to be a living document that is constantly evolving – it will be updated rapidly whenever new data comes out, as well as any potential new vaccines that may get approved in the future.” The NCCN also noted that the advisory committee will meet regularly to refine the recommendations as needed.
Dr. Pergam disclosed relationships with Chimerix Inc., Merck & Co., Global Life Technologies Inc., and Sanofi-Aventis. Dr. Disis disclosed grants from Pfizer, Bavarian Nordisk, Janssen, and Precigen. She is the founder of EpiThany and editor-in-chief of JAMA Oncology.
Vaccination timing considerations vary based on factors such as cancer and treatment type, and reasons for delaying vaccination in the general public also apply to cancer patients (recent COVID-19 exposure, for example).
In general, however, patients with cancer should be assigned to Centers for Disease Control and Prevention priority group 1 b/c and immunized when vaccination is available to them, the guidelines state. Exceptions to this recommendation include:
- Patients undergoing hematopoietic stem cell transplant or receiving engineered cellular therapy such as chimeric antigen receptor T-cell therapy. Vaccination should be delayed for at least 3 months in these patients to maximize vaccine efficacy. Caregivers of these patients, however, should be immunized when possible.
- Patients with hematologic malignancies who are receiving intensive cytotoxic chemotherapy, such as cytarabine- or anthracycline-based regimens for acute myeloid leukemia. Vaccination in these patients should be delayed until absolute neutrophil count recovery.
- Patients undergoing major surgery. Vaccination should occur at least a few days before or after surgery.
- Patients who have experienced a severe or immediate adverse reaction to any of the ingredients in the mRNA COVID-19 vaccines.
Conversely, vaccination should occur when available in patients with hematologic malignancies and marrow failure who are expected to have limited or no recovery, patients with hematologic malignancies who are on long-term maintenance therapy, and patients with solid tumors who are receiving cytotoxic chemotherapy, targeted therapy, checkpoint inhibitors and other immunotherapy, or radiotherapy.
Caregivers, household contacts, and other close contacts who are 16 years of age and older should be vaccinated whenever they are eligible.
Unique concerns in patients with cancer
The NCCN recommendations were developed to address the unique issues and concerns with respect to patients with cancer, who have an increased risk of severe illness from SARS-CoV-2 infection. But the guidelines come with a caveat: “[t]here are limited safety and efficacy data in these patients,” the NCCN emphasized in a press statement.
“Right now, there is urgent need and limited data,” Steven Pergam, MD, co-leader of the NCCN COVID-19 Vaccination Committee, said in the statement.
“Our number one goal is helping to get the vaccine to as many people as we can,” Dr. Pergam said. “That means following existing national and regional directions for prioritizing people who are more likely to face death or severe illness from COVID-19.”
Dr. Pergam, associate professor at Fred Hutchinson Cancer Research Center in Seattle, further explained that “people receiving active cancer treatment are at greater risk for worse outcomes from COVID-19, particularly if they are older and have additional comorbidities, like immunosuppression.”
NCCN’s recommendations couldn’t have come at a better time for patients with cancer, according to Nora Disis, MD, a professor at the University of Washington in Seattle.
“The NCCN’s recommendations to prioritize COVID vaccinations for cancer patients on active treatment is an important step forward in protecting our patients from the infection,” Dr. Disis said in an interview.
“Cancer patients may be at higher risk for the complications seen with infection. In addition, cancer is a disease of older people, and a good number of our patients have the comorbidities that would predict a poorer outcome if they should become sick,” Dr. Disis added. “With the correct treatment, many patients with cancer will be long-term survivors. It is important that they be protected from infection with COVID to realize their best outcome.”
Additional vaccine considerations
The NCCN recommendations also address several other issues of importance for cancer patients, including:
- Deprioritizing other vaccines. COVID-19 vaccines should take precedence over other vaccines because data on dual vaccination are lacking. The NCCN recommends waiting 14 days after COVID-19 vaccination to deliver other vaccines.
- Vaccinating clinical trial participants. Trial leads should be consulted to prevent protocol violations or exclusions.
- Decision-making in the setting of limited vaccine availability. The NCCN noted that decisions on allocation must be made in accordance with state and local vaccine guidance but suggests prioritizing appropriate patients on active treatment, those planning to start treatment, and those who have just completed treatment. Additional risk factors for these patients, as well as other factors associated with risk for adverse COVID-19 outcomes, should also be considered. These include advanced age, comorbidities, and adverse social and demographic factors such as poverty and limited health care access.
- The need for ongoing prevention measures. Vaccines have been shown to decrease the incidence of COVID-19 and related complications, but it remains unclear whether vaccines prevent infection and subsequent transmission. This means everyone should continue following prevention recommendations, such as wearing masks and avoiding crowds.
The NCCN stressed that these recommendations are “intended to be a living document that is constantly evolving – it will be updated rapidly whenever new data comes out, as well as any potential new vaccines that may get approved in the future.” The NCCN also noted that the advisory committee will meet regularly to refine the recommendations as needed.
Dr. Pergam disclosed relationships with Chimerix Inc., Merck & Co., Global Life Technologies Inc., and Sanofi-Aventis. Dr. Disis disclosed grants from Pfizer, Bavarian Nordisk, Janssen, and Precigen. She is the founder of EpiThany and editor-in-chief of JAMA Oncology.
Vaccination timing considerations vary based on factors such as cancer and treatment type, and reasons for delaying vaccination in the general public also apply to cancer patients (recent COVID-19 exposure, for example).
In general, however, patients with cancer should be assigned to Centers for Disease Control and Prevention priority group 1 b/c and immunized when vaccination is available to them, the guidelines state. Exceptions to this recommendation include:
- Patients undergoing hematopoietic stem cell transplant or receiving engineered cellular therapy such as chimeric antigen receptor T-cell therapy. Vaccination should be delayed for at least 3 months in these patients to maximize vaccine efficacy. Caregivers of these patients, however, should be immunized when possible.
- Patients with hematologic malignancies who are receiving intensive cytotoxic chemotherapy, such as cytarabine- or anthracycline-based regimens for acute myeloid leukemia. Vaccination in these patients should be delayed until absolute neutrophil count recovery.
- Patients undergoing major surgery. Vaccination should occur at least a few days before or after surgery.
- Patients who have experienced a severe or immediate adverse reaction to any of the ingredients in the mRNA COVID-19 vaccines.
Conversely, vaccination should occur when available in patients with hematologic malignancies and marrow failure who are expected to have limited or no recovery, patients with hematologic malignancies who are on long-term maintenance therapy, and patients with solid tumors who are receiving cytotoxic chemotherapy, targeted therapy, checkpoint inhibitors and other immunotherapy, or radiotherapy.
Caregivers, household contacts, and other close contacts who are 16 years of age and older should be vaccinated whenever they are eligible.
Unique concerns in patients with cancer
The NCCN recommendations were developed to address the unique issues and concerns with respect to patients with cancer, who have an increased risk of severe illness from SARS-CoV-2 infection. But the guidelines come with a caveat: “[t]here are limited safety and efficacy data in these patients,” the NCCN emphasized in a press statement.
“Right now, there is urgent need and limited data,” Steven Pergam, MD, co-leader of the NCCN COVID-19 Vaccination Committee, said in the statement.
“Our number one goal is helping to get the vaccine to as many people as we can,” Dr. Pergam said. “That means following existing national and regional directions for prioritizing people who are more likely to face death or severe illness from COVID-19.”
Dr. Pergam, associate professor at Fred Hutchinson Cancer Research Center in Seattle, further explained that “people receiving active cancer treatment are at greater risk for worse outcomes from COVID-19, particularly if they are older and have additional comorbidities, like immunosuppression.”
NCCN’s recommendations couldn’t have come at a better time for patients with cancer, according to Nora Disis, MD, a professor at the University of Washington in Seattle.
“The NCCN’s recommendations to prioritize COVID vaccinations for cancer patients on active treatment is an important step forward in protecting our patients from the infection,” Dr. Disis said in an interview.
“Cancer patients may be at higher risk for the complications seen with infection. In addition, cancer is a disease of older people, and a good number of our patients have the comorbidities that would predict a poorer outcome if they should become sick,” Dr. Disis added. “With the correct treatment, many patients with cancer will be long-term survivors. It is important that they be protected from infection with COVID to realize their best outcome.”
Additional vaccine considerations
The NCCN recommendations also address several other issues of importance for cancer patients, including:
- Deprioritizing other vaccines. COVID-19 vaccines should take precedence over other vaccines because data on dual vaccination are lacking. The NCCN recommends waiting 14 days after COVID-19 vaccination to deliver other vaccines.
- Vaccinating clinical trial participants. Trial leads should be consulted to prevent protocol violations or exclusions.
- Decision-making in the setting of limited vaccine availability. The NCCN noted that decisions on allocation must be made in accordance with state and local vaccine guidance but suggests prioritizing appropriate patients on active treatment, those planning to start treatment, and those who have just completed treatment. Additional risk factors for these patients, as well as other factors associated with risk for adverse COVID-19 outcomes, should also be considered. These include advanced age, comorbidities, and adverse social and demographic factors such as poverty and limited health care access.
- The need for ongoing prevention measures. Vaccines have been shown to decrease the incidence of COVID-19 and related complications, but it remains unclear whether vaccines prevent infection and subsequent transmission. This means everyone should continue following prevention recommendations, such as wearing masks and avoiding crowds.
The NCCN stressed that these recommendations are “intended to be a living document that is constantly evolving – it will be updated rapidly whenever new data comes out, as well as any potential new vaccines that may get approved in the future.” The NCCN also noted that the advisory committee will meet regularly to refine the recommendations as needed.
Dr. Pergam disclosed relationships with Chimerix Inc., Merck & Co., Global Life Technologies Inc., and Sanofi-Aventis. Dr. Disis disclosed grants from Pfizer, Bavarian Nordisk, Janssen, and Precigen. She is the founder of EpiThany and editor-in-chief of JAMA Oncology.