User login
Expert offers tips for sorting out pink lesions on dermoscopy
Even in the most experienced hands, .
“For me, pink lesions are challenging,” Jennifer A. Stein, MD, PhD, said during the virtual Orlando Dermatology Aesthetic and Clinical Conference. “How can dermoscopy help us distinguish between Spitz nevus, melanoma, clear cell acanthoma, psoriasis, basal cell carcinoma, and squamous cell carcinoma?”
Dr. Stein, professor of dermatology at New York University, offered four tips. First, look for the shiny white perpendicular lines, otherwise known as the chrysalis or crystalline pattern. “You can only see this feature when you’re looking with polarized light,” she said. “This is why you want a dermatoscope that has polarized light, and better yet, one that you’re able to turn on and off, the hybrid kind, because then you can convince yourself that you’re looking at this feature, because it blinks on and off.”
The differential diagnosis for white shiny perpendicular lines includes dermatofibroma/scars (which is most common), Spitz and atypical genital nevi, BCC, and melanoma. “Dermatofibromas sometimes have white circles or rings in the center,” Dr. Stein said. “In BCC, the lines aren’t always perpendicular. Sometimes it’s more of a blotch or strands.”
A second tip for managing a pink lesion on dermoscopy is to look for any brown color. “When you see that combo together you have to worry,” she said. “When you see pigment network on dermoscopy, you have to put melanoma in your differential. If you see shiny white lines in something that is melanocytic, there’s a 98% specificity for melanoma.”
A third tip she offered for managing pink lesions is to check the blood vessels for clues. “For years, I was just naming the vessels based on making the diagnosis and then deciding, ‘that’s a basal cell carcinoma; those must be branching vessels,’ ” said Dr. Stein, who manages NYU’s medical dermatology faculty group practice.
However, blood vessel patterns differ. For example, branching or arborizing vessels are suggestive of BCC. “These vessels are very crisp-looking on dermoscopy,” she said. “They’re all in the same plane of focus and they look like they were drawn in with a fine point marker. That’s different from other blood vessel patterns.” She also pointed out that superficial basal cells have short, fine telangiectasias. “When you put on the polarized light, the clue is the white, shiny structures,” she said.
Dotted vessels, meanwhile, appear on dermoscopy as small red dots aligned perpendicular to the skin surface. The differential includes inflammatory lesions like psoriasis, stasis, and trauma; clear cell acanthoma (characterized by a “string of pearls” arrangement), nevi, and melanoma. “I find dermoscopy most useful in diagnosing SCC – especially squamous cell in situ,” she said. “Important clinical clues suggestive of SCC or melanoma include a solitary lesion, it’s new, it’s growing, and it’s not going away with a topical steroid.”
An additional pattern to be aware of are hairpin vessels, which are looped and feature a sharp bend at one end. These are often seen in seborrheic keratoses. “You can’t count on the hairpin vessels alone, because you can see this in anything keratotic, such as in keratoacanthoma (at the periphery with a yellow keratotic center), warts, SCC, BCC, as well as in dermal nevi and Spitz nevi,” said Dr. Stein, who recommended dermoscopedia.org as resource.
Comma vessels, meanwhile, appear in dermal or compound nevi. She described these as “slightly curved vessels that are much less in focus than branched vessels, because they come in and out of the plane of focus,” she said. “If you put your dermatoscope on top of the nevus and wobble it around you can appreciate the curve. If you look at it from the side, it looks like a curve. If you look at it straight on it will look more like a line. If you look at from the end it will look like a dot.”
Another vessel type she discussed are linear irregular and polymorphous vessels, which she described as “any combination of different types of vessels. We get most worried when we see dotted and linear irregular vessels together. In that case, you worry about melanoma. These can also be seen in nevi and other tumors, such as BCC.”
Dr. Stein’s fourth tip of the presentation was a reminder to consider dermoscopy as one piece of the clinical exam. “Always think about the lesion in context of the rest of the clinical picture and history,” she said. “Don’t get discouraged if it’s hard; just keep practicing. Look for any brown and use your clinical clues to put together to make the right decision.”
She disclosed that NYU receives compensation from MoleSafe for her telemedicine dermoscopic diagnoses.
Even in the most experienced hands, .

“For me, pink lesions are challenging,” Jennifer A. Stein, MD, PhD, said during the virtual Orlando Dermatology Aesthetic and Clinical Conference. “How can dermoscopy help us distinguish between Spitz nevus, melanoma, clear cell acanthoma, psoriasis, basal cell carcinoma, and squamous cell carcinoma?”
Dr. Stein, professor of dermatology at New York University, offered four tips. First, look for the shiny white perpendicular lines, otherwise known as the chrysalis or crystalline pattern. “You can only see this feature when you’re looking with polarized light,” she said. “This is why you want a dermatoscope that has polarized light, and better yet, one that you’re able to turn on and off, the hybrid kind, because then you can convince yourself that you’re looking at this feature, because it blinks on and off.”
The differential diagnosis for white shiny perpendicular lines includes dermatofibroma/scars (which is most common), Spitz and atypical genital nevi, BCC, and melanoma. “Dermatofibromas sometimes have white circles or rings in the center,” Dr. Stein said. “In BCC, the lines aren’t always perpendicular. Sometimes it’s more of a blotch or strands.”
A second tip for managing a pink lesion on dermoscopy is to look for any brown color. “When you see that combo together you have to worry,” she said. “When you see pigment network on dermoscopy, you have to put melanoma in your differential. If you see shiny white lines in something that is melanocytic, there’s a 98% specificity for melanoma.”
A third tip she offered for managing pink lesions is to check the blood vessels for clues. “For years, I was just naming the vessels based on making the diagnosis and then deciding, ‘that’s a basal cell carcinoma; those must be branching vessels,’ ” said Dr. Stein, who manages NYU’s medical dermatology faculty group practice.
However, blood vessel patterns differ. For example, branching or arborizing vessels are suggestive of BCC. “These vessels are very crisp-looking on dermoscopy,” she said. “They’re all in the same plane of focus and they look like they were drawn in with a fine point marker. That’s different from other blood vessel patterns.” She also pointed out that superficial basal cells have short, fine telangiectasias. “When you put on the polarized light, the clue is the white, shiny structures,” she said.
Dotted vessels, meanwhile, appear on dermoscopy as small red dots aligned perpendicular to the skin surface. The differential includes inflammatory lesions like psoriasis, stasis, and trauma; clear cell acanthoma (characterized by a “string of pearls” arrangement), nevi, and melanoma. “I find dermoscopy most useful in diagnosing SCC – especially squamous cell in situ,” she said. “Important clinical clues suggestive of SCC or melanoma include a solitary lesion, it’s new, it’s growing, and it’s not going away with a topical steroid.”
An additional pattern to be aware of are hairpin vessels, which are looped and feature a sharp bend at one end. These are often seen in seborrheic keratoses. “You can’t count on the hairpin vessels alone, because you can see this in anything keratotic, such as in keratoacanthoma (at the periphery with a yellow keratotic center), warts, SCC, BCC, as well as in dermal nevi and Spitz nevi,” said Dr. Stein, who recommended dermoscopedia.org as resource.
Comma vessels, meanwhile, appear in dermal or compound nevi. She described these as “slightly curved vessels that are much less in focus than branched vessels, because they come in and out of the plane of focus,” she said. “If you put your dermatoscope on top of the nevus and wobble it around you can appreciate the curve. If you look at it from the side, it looks like a curve. If you look at it straight on it will look more like a line. If you look at from the end it will look like a dot.”
Another vessel type she discussed are linear irregular and polymorphous vessels, which she described as “any combination of different types of vessels. We get most worried when we see dotted and linear irregular vessels together. In that case, you worry about melanoma. These can also be seen in nevi and other tumors, such as BCC.”
Dr. Stein’s fourth tip of the presentation was a reminder to consider dermoscopy as one piece of the clinical exam. “Always think about the lesion in context of the rest of the clinical picture and history,” she said. “Don’t get discouraged if it’s hard; just keep practicing. Look for any brown and use your clinical clues to put together to make the right decision.”
She disclosed that NYU receives compensation from MoleSafe for her telemedicine dermoscopic diagnoses.
Even in the most experienced hands, .

“For me, pink lesions are challenging,” Jennifer A. Stein, MD, PhD, said during the virtual Orlando Dermatology Aesthetic and Clinical Conference. “How can dermoscopy help us distinguish between Spitz nevus, melanoma, clear cell acanthoma, psoriasis, basal cell carcinoma, and squamous cell carcinoma?”
Dr. Stein, professor of dermatology at New York University, offered four tips. First, look for the shiny white perpendicular lines, otherwise known as the chrysalis or crystalline pattern. “You can only see this feature when you’re looking with polarized light,” she said. “This is why you want a dermatoscope that has polarized light, and better yet, one that you’re able to turn on and off, the hybrid kind, because then you can convince yourself that you’re looking at this feature, because it blinks on and off.”
The differential diagnosis for white shiny perpendicular lines includes dermatofibroma/scars (which is most common), Spitz and atypical genital nevi, BCC, and melanoma. “Dermatofibromas sometimes have white circles or rings in the center,” Dr. Stein said. “In BCC, the lines aren’t always perpendicular. Sometimes it’s more of a blotch or strands.”
A second tip for managing a pink lesion on dermoscopy is to look for any brown color. “When you see that combo together you have to worry,” she said. “When you see pigment network on dermoscopy, you have to put melanoma in your differential. If you see shiny white lines in something that is melanocytic, there’s a 98% specificity for melanoma.”
A third tip she offered for managing pink lesions is to check the blood vessels for clues. “For years, I was just naming the vessels based on making the diagnosis and then deciding, ‘that’s a basal cell carcinoma; those must be branching vessels,’ ” said Dr. Stein, who manages NYU’s medical dermatology faculty group practice.
However, blood vessel patterns differ. For example, branching or arborizing vessels are suggestive of BCC. “These vessels are very crisp-looking on dermoscopy,” she said. “They’re all in the same plane of focus and they look like they were drawn in with a fine point marker. That’s different from other blood vessel patterns.” She also pointed out that superficial basal cells have short, fine telangiectasias. “When you put on the polarized light, the clue is the white, shiny structures,” she said.
Dotted vessels, meanwhile, appear on dermoscopy as small red dots aligned perpendicular to the skin surface. The differential includes inflammatory lesions like psoriasis, stasis, and trauma; clear cell acanthoma (characterized by a “string of pearls” arrangement), nevi, and melanoma. “I find dermoscopy most useful in diagnosing SCC – especially squamous cell in situ,” she said. “Important clinical clues suggestive of SCC or melanoma include a solitary lesion, it’s new, it’s growing, and it’s not going away with a topical steroid.”
An additional pattern to be aware of are hairpin vessels, which are looped and feature a sharp bend at one end. These are often seen in seborrheic keratoses. “You can’t count on the hairpin vessels alone, because you can see this in anything keratotic, such as in keratoacanthoma (at the periphery with a yellow keratotic center), warts, SCC, BCC, as well as in dermal nevi and Spitz nevi,” said Dr. Stein, who recommended dermoscopedia.org as resource.
Comma vessels, meanwhile, appear in dermal or compound nevi. She described these as “slightly curved vessels that are much less in focus than branched vessels, because they come in and out of the plane of focus,” she said. “If you put your dermatoscope on top of the nevus and wobble it around you can appreciate the curve. If you look at it from the side, it looks like a curve. If you look at it straight on it will look more like a line. If you look at from the end it will look like a dot.”
Another vessel type she discussed are linear irregular and polymorphous vessels, which she described as “any combination of different types of vessels. We get most worried when we see dotted and linear irregular vessels together. In that case, you worry about melanoma. These can also be seen in nevi and other tumors, such as BCC.”
Dr. Stein’s fourth tip of the presentation was a reminder to consider dermoscopy as one piece of the clinical exam. “Always think about the lesion in context of the rest of the clinical picture and history,” she said. “Don’t get discouraged if it’s hard; just keep practicing. Look for any brown and use your clinical clues to put together to make the right decision.”
She disclosed that NYU receives compensation from MoleSafe for her telemedicine dermoscopic diagnoses.
FROM ODAC 2021
Male Genital Examinations: Special Considerations and Pearls for Dermatologists
Men have unique dermatologic needs yet are significantly less likely than women to visit a dermatologist’s office.1 Male patients might have preconceived notions about the nature of dermatology visits and necessary areas of the body to be examined: For example, male patients might associate the genital examination with a urologist and not expect a dermatologist to complete such a seemingly private examination.2
Genital examinations are currently underperformed: Only one-quarter of dermatologists report examining a male patient’s genitals at most or all visits.3 In this commentary, we discuss the importance of genital examinations in men’s dermatology, specific issues that can arise, and strategies to enhance the quality and frequency of genital examinations in male patients.
Invaluable Aspect of Care
Thorough inspection of a male patient’s genital region is an important part of conducting a total-body skin examination (TBSE) for routine surveillance and evaluation of genital dermatoses. Sexually transmitted infections, warts, and other common lesions can be missed in diagnosis without careful inspection of the genital region. Additionally, scrotal malignancies, such as primary and metastatic melanoma and basal cell carcinoma, though rare, might be overlooked until symptoms become severe.4,5
There is no substitute for a physical examination but, in certain circumstances, it might be appropriate for a dermatologist to ask a patient if he has concerning lesions on his genitals. However, patients often are unsure of worrisome signs, and areas of the perineum might not be easily visible to a patient. Genital inspection during the physical examination allows for a teachable moment, during which the dermatologist can educate the patient about benign lesions and variants, such as pearly penile papules, seborrheic keratoses, and sebaceous cysts.6 These lesions might not require intervention but should be monitored for atypical features or infection.6
Also, the dermatologist might incidentally discover transmissible lesions, such as condylomata caused by human papillomavirus, which has been shown to be present in approximately 50% of men in the United States7—many of whom are unaware. Inflammatory dermatoses, such as psoriasis, often affect the genitals and go unnoticed; prompt intervention can decrease the likelihood of complications.6
Protocol for Genital Examinations
To examine the genitals, all surfaces of the penis, scrotum, and perineum should be evaluated, with anatomic and pathologic variants noted. The patient or physician should stretch the penis, maneuvering it in multiple directions so that all aspects can be examined. In uncircumcised men, the foreskin should be retracted so that the head of the penis can be examined, followed by replacement of the foreskin by the patient.8 The scrotum also should be examined and lifted to fully view the perineum.
Providers should not grasp the penis with the whole hand but use the thumb and first finger to hold the head of the penis to maneuver it.8 Similarly, using the back of the hand and fingers to manipulate the genitals establishes boundaries and sets a clinical tone for the examination.
Unintentional Erection
Unique to the male dermatologic examination is the unintentional patient erection; a physician might be unsure of how to approach such a potentially awkward situation. An erection is not always an indication of sexual arousal; rather, it can reflect an autonomic reflex in response to physical stimulation. Erections occur commonly in health care settings, especially if the genitals are being manipulated.9
Generally, the course of action here depends on the patient’s response.10 For patients who appear unbothered, it might be appropriate to ignore the erection and proceed with the examination, especially if the physician is not actively examining the genital region. If the patient appears embarrassed, the physician can say “This is completely normal” or “Random erections are common” to normalize the situation. Joking or laughing should be avoided. For a patient who appears upset, the physician can step outside the room for a brief period to give the patient privacy, then re-enter and ask him if he is comfortable continuing with the examination.
When a patient develops an erection, the physician might become uncomfortable and, consciously or subconsciously, increase the pace of the examination, which is a natural tendency, but expediency at the expense of comprehensive care is inappropriate.
Examiner’s Body Language and Tone
Throughout the genital examination, the physician should be mindful of their comments and body language to avoid exacerbating patient vulnerability. Using anatomic terms, rather than colloquial ones, to describe the genitalia is advised to prevent misunderstanding and maintain a professional clinical environment. Providers should be prepared to explain anatomic terms because some patients are not familiar with medical terminology.
Presence of a Chaperone
Involving a chaperone, as recommended by the American Medical Association, might make a patient more comfortable and alleviate potential misunderstanding. Still, physicians should be aware that some patients might feel uncomfortable with a chaperone, interpreting their presence as an expectation of impropriety.11 Universal offering of a chaperone to all patients, regardless of the gender of the physician, as well as appropriate signage in the clinical environment, normalizes chaperone invitation and use.
Other Helpful Considerations
Various strategies in the male genital examination can increase patient and physician comfort and improve care:
- The patient should be offered a gown before a TBSE or any skin examination during which the genitals will be examined.
- The patient should be allowed to keep his shorts or underwear on to avoid the feeling of being naked, which can provoke anxiety. Prior to beginning the examination, disclose that it will include “under the covered areas.”
- Ask the patient for permission to conduct the examination, enumerate the steps, and provide a rationale for a genital examination. These steps help gain cooperation, alleviate anticipation, and prevent surprise.
- To increase the patient’s comfort level, he can be asked whether he prefers to be examined supine or standing.
- Consider allowing the patient, himself, to expose and manipulate his genitals during the examination to increase his involvement and sense of autonomy.
- For genital examinations, patients often prefer that the examiner be a physician of the same gender. Accommodating a patient’s request regarding the examiner’s gender might not always be possible, but the medical practice should make an honest attempt to oblige.
Lastly, providers should be cognizant of the needs of male sexual and gender minority populations (ie, gay, bisexual, transgender/gender diverse, queer or questioning, intersex, and asexual persons). For example, transgender women might retain male anatomy or have surgical alteration of the genital region that also requires evaluation. In such patient populations, the genital examination is equally important to evaluate for dermatologic conditions that require treatment.
Final Thoughts
The male genital examination is an important component of the TBSE, as dermatologists can recognize lesions before symptoms present. Robust educational methods for trainees and practitioners in dermatology are lacking, and development of curricula might be beneficial to increase comfort in performing the genital examination. Still, use of the procedures described in this commentary can normalize the men’s genital examination, optimize the physical examination, and improve men’s overall dermatologic health.
- Tripathi R, Knusel KD, Ezaldein HH, et al. Association of demographic and socioeconomic characteristics with differences in use of outpatient dermatology services in the United States. JAMA Dermatol. 2018;154:1286-1291.
- Brezinski EA, Harskamp CT, Ledo L, et al. Public perception of dermatologists and comparison with other medical specialties: results from a national survey. J Am Acad Dermatol. 2014;71:875-881.
- Rieder EA, Mu EW, Wang J, et al. Dermatologist practices during total body skin examinations: a survey study. J Drugs Dermatol. 2018;17:516-520.
- Gonzalez CD, Hawkes JE, Bowles TL. Malignant melanoma scrotal metastasis: the importance of the genital examination. JAAD Case Rep. 2017;3:10-12.
- Solimani F, Juratli H, Hoch M, et al. Basal cell carcinoma of the scrotum: an important but easily overlooked entity. J Eur Acad Dermatol Venereol. 2018;32:E254-E255.
- Gabrielson AT, Le TV, Fontenot C, et al. Male genital dermatology: a primer for the sexual medicine physician. Sex Med Rev. 2019;7:71-83.
- Han JJ, Beltran TH, Song JW, et al. Prevalence of genital human papillomavirus infection and human papillomavirus vaccination rates among US adult men: National Health and Nutrition Examination Survey (NHANES) 2013-2014. JAMA Oncol. 2017;3:810-816.
- Albaugh JA, Kellogg-Spadt S. Genital and dermatologic examination. part II: the male patient. Urol Nurs. 2003;23:366-367.
- Dean RC, Lue TF. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol Clin North Am. 2005;32:379-395.
- Norwick P, Weston GK, Grant-Kels JM. Erection ethics. J Am Acad Dermatol. 2019;81:1225.
- Vogel L. Chaperones: friend or foe, and to whom? CMAJ. 2012;184:642-643.
Men have unique dermatologic needs yet are significantly less likely than women to visit a dermatologist’s office.1 Male patients might have preconceived notions about the nature of dermatology visits and necessary areas of the body to be examined: For example, male patients might associate the genital examination with a urologist and not expect a dermatologist to complete such a seemingly private examination.2
Genital examinations are currently underperformed: Only one-quarter of dermatologists report examining a male patient’s genitals at most or all visits.3 In this commentary, we discuss the importance of genital examinations in men’s dermatology, specific issues that can arise, and strategies to enhance the quality and frequency of genital examinations in male patients.
Invaluable Aspect of Care
Thorough inspection of a male patient’s genital region is an important part of conducting a total-body skin examination (TBSE) for routine surveillance and evaluation of genital dermatoses. Sexually transmitted infections, warts, and other common lesions can be missed in diagnosis without careful inspection of the genital region. Additionally, scrotal malignancies, such as primary and metastatic melanoma and basal cell carcinoma, though rare, might be overlooked until symptoms become severe.4,5
There is no substitute for a physical examination but, in certain circumstances, it might be appropriate for a dermatologist to ask a patient if he has concerning lesions on his genitals. However, patients often are unsure of worrisome signs, and areas of the perineum might not be easily visible to a patient. Genital inspection during the physical examination allows for a teachable moment, during which the dermatologist can educate the patient about benign lesions and variants, such as pearly penile papules, seborrheic keratoses, and sebaceous cysts.6 These lesions might not require intervention but should be monitored for atypical features or infection.6
Also, the dermatologist might incidentally discover transmissible lesions, such as condylomata caused by human papillomavirus, which has been shown to be present in approximately 50% of men in the United States7—many of whom are unaware. Inflammatory dermatoses, such as psoriasis, often affect the genitals and go unnoticed; prompt intervention can decrease the likelihood of complications.6
Protocol for Genital Examinations
To examine the genitals, all surfaces of the penis, scrotum, and perineum should be evaluated, with anatomic and pathologic variants noted. The patient or physician should stretch the penis, maneuvering it in multiple directions so that all aspects can be examined. In uncircumcised men, the foreskin should be retracted so that the head of the penis can be examined, followed by replacement of the foreskin by the patient.8 The scrotum also should be examined and lifted to fully view the perineum.
Providers should not grasp the penis with the whole hand but use the thumb and first finger to hold the head of the penis to maneuver it.8 Similarly, using the back of the hand and fingers to manipulate the genitals establishes boundaries and sets a clinical tone for the examination.
Unintentional Erection
Unique to the male dermatologic examination is the unintentional patient erection; a physician might be unsure of how to approach such a potentially awkward situation. An erection is not always an indication of sexual arousal; rather, it can reflect an autonomic reflex in response to physical stimulation. Erections occur commonly in health care settings, especially if the genitals are being manipulated.9
Generally, the course of action here depends on the patient’s response.10 For patients who appear unbothered, it might be appropriate to ignore the erection and proceed with the examination, especially if the physician is not actively examining the genital region. If the patient appears embarrassed, the physician can say “This is completely normal” or “Random erections are common” to normalize the situation. Joking or laughing should be avoided. For a patient who appears upset, the physician can step outside the room for a brief period to give the patient privacy, then re-enter and ask him if he is comfortable continuing with the examination.
When a patient develops an erection, the physician might become uncomfortable and, consciously or subconsciously, increase the pace of the examination, which is a natural tendency, but expediency at the expense of comprehensive care is inappropriate.
Examiner’s Body Language and Tone
Throughout the genital examination, the physician should be mindful of their comments and body language to avoid exacerbating patient vulnerability. Using anatomic terms, rather than colloquial ones, to describe the genitalia is advised to prevent misunderstanding and maintain a professional clinical environment. Providers should be prepared to explain anatomic terms because some patients are not familiar with medical terminology.
Presence of a Chaperone
Involving a chaperone, as recommended by the American Medical Association, might make a patient more comfortable and alleviate potential misunderstanding. Still, physicians should be aware that some patients might feel uncomfortable with a chaperone, interpreting their presence as an expectation of impropriety.11 Universal offering of a chaperone to all patients, regardless of the gender of the physician, as well as appropriate signage in the clinical environment, normalizes chaperone invitation and use.
Other Helpful Considerations
Various strategies in the male genital examination can increase patient and physician comfort and improve care:
- The patient should be offered a gown before a TBSE or any skin examination during which the genitals will be examined.
- The patient should be allowed to keep his shorts or underwear on to avoid the feeling of being naked, which can provoke anxiety. Prior to beginning the examination, disclose that it will include “under the covered areas.”
- Ask the patient for permission to conduct the examination, enumerate the steps, and provide a rationale for a genital examination. These steps help gain cooperation, alleviate anticipation, and prevent surprise.
- To increase the patient’s comfort level, he can be asked whether he prefers to be examined supine or standing.
- Consider allowing the patient, himself, to expose and manipulate his genitals during the examination to increase his involvement and sense of autonomy.
- For genital examinations, patients often prefer that the examiner be a physician of the same gender. Accommodating a patient’s request regarding the examiner’s gender might not always be possible, but the medical practice should make an honest attempt to oblige.
Lastly, providers should be cognizant of the needs of male sexual and gender minority populations (ie, gay, bisexual, transgender/gender diverse, queer or questioning, intersex, and asexual persons). For example, transgender women might retain male anatomy or have surgical alteration of the genital region that also requires evaluation. In such patient populations, the genital examination is equally important to evaluate for dermatologic conditions that require treatment.
Final Thoughts
The male genital examination is an important component of the TBSE, as dermatologists can recognize lesions before symptoms present. Robust educational methods for trainees and practitioners in dermatology are lacking, and development of curricula might be beneficial to increase comfort in performing the genital examination. Still, use of the procedures described in this commentary can normalize the men’s genital examination, optimize the physical examination, and improve men’s overall dermatologic health.
Men have unique dermatologic needs yet are significantly less likely than women to visit a dermatologist’s office.1 Male patients might have preconceived notions about the nature of dermatology visits and necessary areas of the body to be examined: For example, male patients might associate the genital examination with a urologist and not expect a dermatologist to complete such a seemingly private examination.2
Genital examinations are currently underperformed: Only one-quarter of dermatologists report examining a male patient’s genitals at most or all visits.3 In this commentary, we discuss the importance of genital examinations in men’s dermatology, specific issues that can arise, and strategies to enhance the quality and frequency of genital examinations in male patients.
Invaluable Aspect of Care
Thorough inspection of a male patient’s genital region is an important part of conducting a total-body skin examination (TBSE) for routine surveillance and evaluation of genital dermatoses. Sexually transmitted infections, warts, and other common lesions can be missed in diagnosis without careful inspection of the genital region. Additionally, scrotal malignancies, such as primary and metastatic melanoma and basal cell carcinoma, though rare, might be overlooked until symptoms become severe.4,5
There is no substitute for a physical examination but, in certain circumstances, it might be appropriate for a dermatologist to ask a patient if he has concerning lesions on his genitals. However, patients often are unsure of worrisome signs, and areas of the perineum might not be easily visible to a patient. Genital inspection during the physical examination allows for a teachable moment, during which the dermatologist can educate the patient about benign lesions and variants, such as pearly penile papules, seborrheic keratoses, and sebaceous cysts.6 These lesions might not require intervention but should be monitored for atypical features or infection.6
Also, the dermatologist might incidentally discover transmissible lesions, such as condylomata caused by human papillomavirus, which has been shown to be present in approximately 50% of men in the United States7—many of whom are unaware. Inflammatory dermatoses, such as psoriasis, often affect the genitals and go unnoticed; prompt intervention can decrease the likelihood of complications.6
Protocol for Genital Examinations
To examine the genitals, all surfaces of the penis, scrotum, and perineum should be evaluated, with anatomic and pathologic variants noted. The patient or physician should stretch the penis, maneuvering it in multiple directions so that all aspects can be examined. In uncircumcised men, the foreskin should be retracted so that the head of the penis can be examined, followed by replacement of the foreskin by the patient.8 The scrotum also should be examined and lifted to fully view the perineum.
Providers should not grasp the penis with the whole hand but use the thumb and first finger to hold the head of the penis to maneuver it.8 Similarly, using the back of the hand and fingers to manipulate the genitals establishes boundaries and sets a clinical tone for the examination.
Unintentional Erection
Unique to the male dermatologic examination is the unintentional patient erection; a physician might be unsure of how to approach such a potentially awkward situation. An erection is not always an indication of sexual arousal; rather, it can reflect an autonomic reflex in response to physical stimulation. Erections occur commonly in health care settings, especially if the genitals are being manipulated.9
Generally, the course of action here depends on the patient’s response.10 For patients who appear unbothered, it might be appropriate to ignore the erection and proceed with the examination, especially if the physician is not actively examining the genital region. If the patient appears embarrassed, the physician can say “This is completely normal” or “Random erections are common” to normalize the situation. Joking or laughing should be avoided. For a patient who appears upset, the physician can step outside the room for a brief period to give the patient privacy, then re-enter and ask him if he is comfortable continuing with the examination.
When a patient develops an erection, the physician might become uncomfortable and, consciously or subconsciously, increase the pace of the examination, which is a natural tendency, but expediency at the expense of comprehensive care is inappropriate.
Examiner’s Body Language and Tone
Throughout the genital examination, the physician should be mindful of their comments and body language to avoid exacerbating patient vulnerability. Using anatomic terms, rather than colloquial ones, to describe the genitalia is advised to prevent misunderstanding and maintain a professional clinical environment. Providers should be prepared to explain anatomic terms because some patients are not familiar with medical terminology.
Presence of a Chaperone
Involving a chaperone, as recommended by the American Medical Association, might make a patient more comfortable and alleviate potential misunderstanding. Still, physicians should be aware that some patients might feel uncomfortable with a chaperone, interpreting their presence as an expectation of impropriety.11 Universal offering of a chaperone to all patients, regardless of the gender of the physician, as well as appropriate signage in the clinical environment, normalizes chaperone invitation and use.
Other Helpful Considerations
Various strategies in the male genital examination can increase patient and physician comfort and improve care:
- The patient should be offered a gown before a TBSE or any skin examination during which the genitals will be examined.
- The patient should be allowed to keep his shorts or underwear on to avoid the feeling of being naked, which can provoke anxiety. Prior to beginning the examination, disclose that it will include “under the covered areas.”
- Ask the patient for permission to conduct the examination, enumerate the steps, and provide a rationale for a genital examination. These steps help gain cooperation, alleviate anticipation, and prevent surprise.
- To increase the patient’s comfort level, he can be asked whether he prefers to be examined supine or standing.
- Consider allowing the patient, himself, to expose and manipulate his genitals during the examination to increase his involvement and sense of autonomy.
- For genital examinations, patients often prefer that the examiner be a physician of the same gender. Accommodating a patient’s request regarding the examiner’s gender might not always be possible, but the medical practice should make an honest attempt to oblige.
Lastly, providers should be cognizant of the needs of male sexual and gender minority populations (ie, gay, bisexual, transgender/gender diverse, queer or questioning, intersex, and asexual persons). For example, transgender women might retain male anatomy or have surgical alteration of the genital region that also requires evaluation. In such patient populations, the genital examination is equally important to evaluate for dermatologic conditions that require treatment.
Final Thoughts
The male genital examination is an important component of the TBSE, as dermatologists can recognize lesions before symptoms present. Robust educational methods for trainees and practitioners in dermatology are lacking, and development of curricula might be beneficial to increase comfort in performing the genital examination. Still, use of the procedures described in this commentary can normalize the men’s genital examination, optimize the physical examination, and improve men’s overall dermatologic health.
- Tripathi R, Knusel KD, Ezaldein HH, et al. Association of demographic and socioeconomic characteristics with differences in use of outpatient dermatology services in the United States. JAMA Dermatol. 2018;154:1286-1291.
- Brezinski EA, Harskamp CT, Ledo L, et al. Public perception of dermatologists and comparison with other medical specialties: results from a national survey. J Am Acad Dermatol. 2014;71:875-881.
- Rieder EA, Mu EW, Wang J, et al. Dermatologist practices during total body skin examinations: a survey study. J Drugs Dermatol. 2018;17:516-520.
- Gonzalez CD, Hawkes JE, Bowles TL. Malignant melanoma scrotal metastasis: the importance of the genital examination. JAAD Case Rep. 2017;3:10-12.
- Solimani F, Juratli H, Hoch M, et al. Basal cell carcinoma of the scrotum: an important but easily overlooked entity. J Eur Acad Dermatol Venereol. 2018;32:E254-E255.
- Gabrielson AT, Le TV, Fontenot C, et al. Male genital dermatology: a primer for the sexual medicine physician. Sex Med Rev. 2019;7:71-83.
- Han JJ, Beltran TH, Song JW, et al. Prevalence of genital human papillomavirus infection and human papillomavirus vaccination rates among US adult men: National Health and Nutrition Examination Survey (NHANES) 2013-2014. JAMA Oncol. 2017;3:810-816.
- Albaugh JA, Kellogg-Spadt S. Genital and dermatologic examination. part II: the male patient. Urol Nurs. 2003;23:366-367.
- Dean RC, Lue TF. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol Clin North Am. 2005;32:379-395.
- Norwick P, Weston GK, Grant-Kels JM. Erection ethics. J Am Acad Dermatol. 2019;81:1225.
- Vogel L. Chaperones: friend or foe, and to whom? CMAJ. 2012;184:642-643.
- Tripathi R, Knusel KD, Ezaldein HH, et al. Association of demographic and socioeconomic characteristics with differences in use of outpatient dermatology services in the United States. JAMA Dermatol. 2018;154:1286-1291.
- Brezinski EA, Harskamp CT, Ledo L, et al. Public perception of dermatologists and comparison with other medical specialties: results from a national survey. J Am Acad Dermatol. 2014;71:875-881.
- Rieder EA, Mu EW, Wang J, et al. Dermatologist practices during total body skin examinations: a survey study. J Drugs Dermatol. 2018;17:516-520.
- Gonzalez CD, Hawkes JE, Bowles TL. Malignant melanoma scrotal metastasis: the importance of the genital examination. JAAD Case Rep. 2017;3:10-12.
- Solimani F, Juratli H, Hoch M, et al. Basal cell carcinoma of the scrotum: an important but easily overlooked entity. J Eur Acad Dermatol Venereol. 2018;32:E254-E255.
- Gabrielson AT, Le TV, Fontenot C, et al. Male genital dermatology: a primer for the sexual medicine physician. Sex Med Rev. 2019;7:71-83.
- Han JJ, Beltran TH, Song JW, et al. Prevalence of genital human papillomavirus infection and human papillomavirus vaccination rates among US adult men: National Health and Nutrition Examination Survey (NHANES) 2013-2014. JAMA Oncol. 2017;3:810-816.
- Albaugh JA, Kellogg-Spadt S. Genital and dermatologic examination. part II: the male patient. Urol Nurs. 2003;23:366-367.
- Dean RC, Lue TF. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol Clin North Am. 2005;32:379-395.
- Norwick P, Weston GK, Grant-Kels JM. Erection ethics. J Am Acad Dermatol. 2019;81:1225.
- Vogel L. Chaperones: friend or foe, and to whom? CMAJ. 2012;184:642-643.
Practice Points
- Genital examinations are an important aspect of comprehensive dermatologic care for male patients.
- Unintentional patient erections are unique to male patients and should be addressed professionally, depending on the patient’s reaction.
- In addition to being mindful of body language and tone, dermatologists may consider involving a chaperone when performing genital examinations to optimize patient experience.
Hypertrophic Lichen Planus–like Eruption Following Pembrolizumab
To the Editor:
Pembrolizumab, a humanized monoclonal anti–programmed cell death protein 1 (PD-1) antibody, acts by blocking negative immune regulators such as PD-1.1 Since its approval by the US Food and Drug Administration in 2014, the use of PD-1 inhibitors such as pembrolizumab has dramatically increased, and they are now the standard of care for cancers such as melanoma, lung cancer, and renal cell carcinoma.2,3 With increased use comes a better understanding of the cutaneous adverse effects that may occur. To date, almost 50% of patients treated with PD-1 inhibitors will develop an adverse cutaneous reaction.4 Thus far, cases of patients developing vitiligo, bullous pemphigoid, psoriasis, granulomatous skin reactions, severe cutaneous reactions (ie, toxic epidermal necrolysis), lupus erythematosus, and lichenoid reactions have been described.3,5,6 There are fewer than 30 documented cases of lichenoid reactions due to anti–PD-1 treatment described in the literature, increasing the importance of case reports to demonstrate a full range of cutaneous findings.3 We present a case of a reaction to pembrolizumab with an eruption of lichenoid papules predominantly involving the hands and feet as well as nail changes.
A 60-year-old man with ocular melanoma metastatic to the right lung, transverse colon, and right axillary lymph nodes presented with a chief concern of growing skin lesions present for 6 weeks on the hands and feet. The lesions were tender to the touch and occasionally drained a clear fluid. He also reported nail fragility. Of note, the patient was being treated for metastatic melanoma with pembrolizumab infusions every 3 weeks, which started 6 weeks prior to the onset of the eruption.
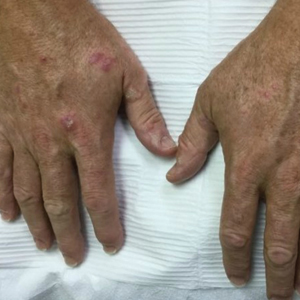
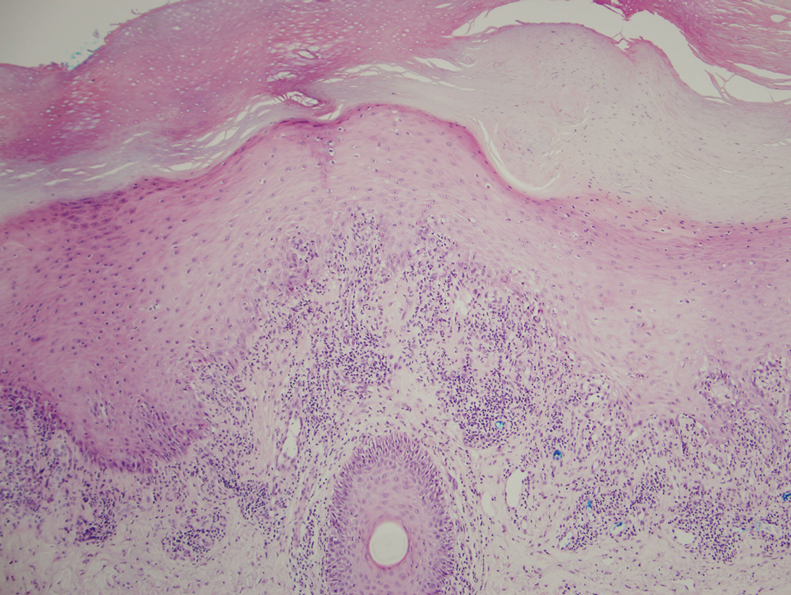
Physical examination demonstrated lichenoid papules on the dorsal and ventral aspects of the hands and feet (Figure 1), as well as longitudinal ridging on numerous fingernails and mild koilonychia. A punch biopsy revealed lichenoid interface dermatitis with irregular epidermal hyperplasia (Figure 2). A diagnosis of hypertrophic lichen planus–like drug eruption in response to pembrolizumab was made and clobetasol cream was prescribed.
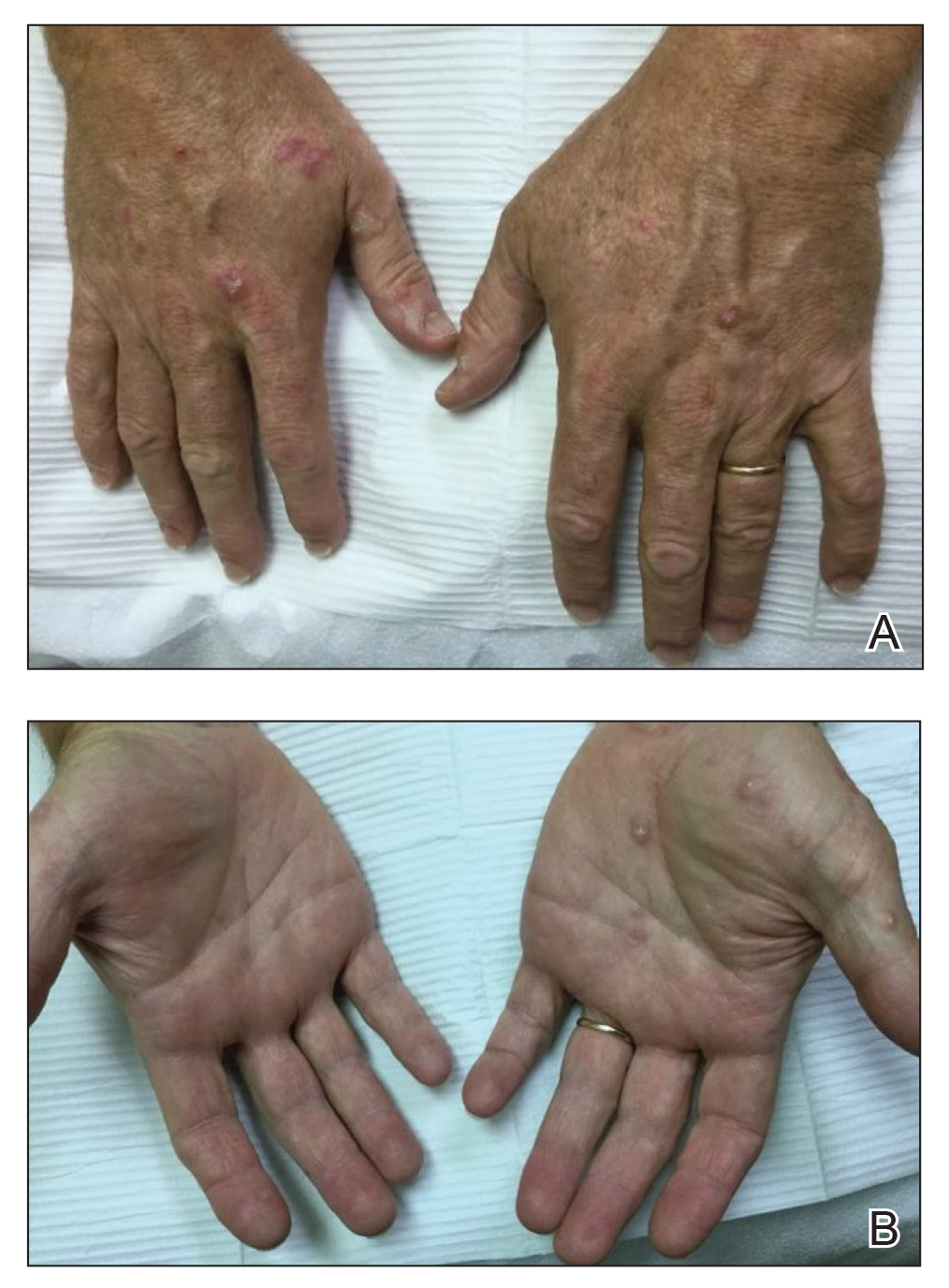
At 1-month follow-up, the patient reported notable improvement with clobetasol, and he was transitioned to tacrolimus ointment 0.1%. He continued to improve until a month later when he reported new lesions arising a week after a pembrolizumab infusion. He continued to use clobetasol cream for flares and tacrolimus ointment for maintenance.
Almost 3 months after the initial visit, the patient presented with inflammation around his right third fingernail of 1 week’s duration, with more notable fragility than his other nails. No trauma was described, and the nail abnormality was attributed to pembrolizumab. Clobetasol cream and biotin 3 mg daily resulted in improvement, and no other nails were affected in a similar way.
Programmed cell death protein 1 blockers are associated with a variety of adverse events including hypothyroidism, gastrointestinal abnormalities, fatigue, and skin disorders.7 In one study (N=83), cutaneous adverse drug events were found to occur in 42% (35/83) of patients following pembrolizumab therapy, with the most common cutaneous lesions being maculopapular eruptions (29% [24/83]), pruritus (12% [10/83]), and hypopigmentation (8% [7/83]).5
A total of 29 cases of lichenoid dermatitis following anti–PD-1 therapy have been described in the literature.3 Cases range from an eruption of photodistributed hyperkeratotic papules and plaques to hypertrophic vesiculobullous lesions.3,6 Suggested pathophysiology involves blocking the interaction of programmed death ligand 1 on keratinocytes with PD-1 on T cells.3 Management typically includes topical or systemic steroids. Cyclosporine and acitretin also have been successful in a small number of patients. Most patients continue anti–PD-1 treatment with systemic therapy.3
Our patient represents a similar lichenoid eruption; however, the distribution on the dorsal and ventral aspects of the hands and feet as well as nail dystrophy make the presentation unique. Anticancer drugs that increase the T-cell immune response by altering the complex signaling among T cells, antigen-presenting cells, and tumor cells have been associated with cutaneous eruptions. Although the exact mechanism is still not fully understood, clinical suspicion of a pembrolizumab reaction should remain high on the differential in the setting of hyperkeratotic papules in association with anti–PD-1 therapy.
- Homet Moreno B, Ribas A. Anti-programmed cell death protein-1/ligand-1 therapy in different cancers. Br J Cancer. 2015;112:1421-1427.
- Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384:1109-1117.
- Simonsen AB, Kaae J, Elleback E, et al. Cutaneous adverse reactions to anti-PD-1 treatment: a systematic review. J Am Acad Dermatol. 2020;83:1415-1424.
- Hwang SJ, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;151:1206-1212.
- Joseph RW, Cappel M, Goedjen B, et al. Lichenoid dermatitis in three patients with metastatic melanoma treated with anti-PD-1 therapy. Cancer Immunol Res. 2015;3:18-22.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
To the Editor:
Pembrolizumab, a humanized monoclonal anti–programmed cell death protein 1 (PD-1) antibody, acts by blocking negative immune regulators such as PD-1.1 Since its approval by the US Food and Drug Administration in 2014, the use of PD-1 inhibitors such as pembrolizumab has dramatically increased, and they are now the standard of care for cancers such as melanoma, lung cancer, and renal cell carcinoma.2,3 With increased use comes a better understanding of the cutaneous adverse effects that may occur. To date, almost 50% of patients treated with PD-1 inhibitors will develop an adverse cutaneous reaction.4 Thus far, cases of patients developing vitiligo, bullous pemphigoid, psoriasis, granulomatous skin reactions, severe cutaneous reactions (ie, toxic epidermal necrolysis), lupus erythematosus, and lichenoid reactions have been described.3,5,6 There are fewer than 30 documented cases of lichenoid reactions due to anti–PD-1 treatment described in the literature, increasing the importance of case reports to demonstrate a full range of cutaneous findings.3 We present a case of a reaction to pembrolizumab with an eruption of lichenoid papules predominantly involving the hands and feet as well as nail changes.
A 60-year-old man with ocular melanoma metastatic to the right lung, transverse colon, and right axillary lymph nodes presented with a chief concern of growing skin lesions present for 6 weeks on the hands and feet. The lesions were tender to the touch and occasionally drained a clear fluid. He also reported nail fragility. Of note, the patient was being treated for metastatic melanoma with pembrolizumab infusions every 3 weeks, which started 6 weeks prior to the onset of the eruption.
Physical examination demonstrated lichenoid papules on the dorsal and ventral aspects of the hands and feet (Figure 1), as well as longitudinal ridging on numerous fingernails and mild koilonychia. A punch biopsy revealed lichenoid interface dermatitis with irregular epidermal hyperplasia (Figure 2). A diagnosis of hypertrophic lichen planus–like drug eruption in response to pembrolizumab was made and clobetasol cream was prescribed.


At 1-month follow-up, the patient reported notable improvement with clobetasol, and he was transitioned to tacrolimus ointment 0.1%. He continued to improve until a month later when he reported new lesions arising a week after a pembrolizumab infusion. He continued to use clobetasol cream for flares and tacrolimus ointment for maintenance.
Almost 3 months after the initial visit, the patient presented with inflammation around his right third fingernail of 1 week’s duration, with more notable fragility than his other nails. No trauma was described, and the nail abnormality was attributed to pembrolizumab. Clobetasol cream and biotin 3 mg daily resulted in improvement, and no other nails were affected in a similar way.
Programmed cell death protein 1 blockers are associated with a variety of adverse events including hypothyroidism, gastrointestinal abnormalities, fatigue, and skin disorders.7 In one study (N=83), cutaneous adverse drug events were found to occur in 42% (35/83) of patients following pembrolizumab therapy, with the most common cutaneous lesions being maculopapular eruptions (29% [24/83]), pruritus (12% [10/83]), and hypopigmentation (8% [7/83]).5
A total of 29 cases of lichenoid dermatitis following anti–PD-1 therapy have been described in the literature.3 Cases range from an eruption of photodistributed hyperkeratotic papules and plaques to hypertrophic vesiculobullous lesions.3,6 Suggested pathophysiology involves blocking the interaction of programmed death ligand 1 on keratinocytes with PD-1 on T cells.3 Management typically includes topical or systemic steroids. Cyclosporine and acitretin also have been successful in a small number of patients. Most patients continue anti–PD-1 treatment with systemic therapy.3
Our patient represents a similar lichenoid eruption; however, the distribution on the dorsal and ventral aspects of the hands and feet as well as nail dystrophy make the presentation unique. Anticancer drugs that increase the T-cell immune response by altering the complex signaling among T cells, antigen-presenting cells, and tumor cells have been associated with cutaneous eruptions. Although the exact mechanism is still not fully understood, clinical suspicion of a pembrolizumab reaction should remain high on the differential in the setting of hyperkeratotic papules in association with anti–PD-1 therapy.
To the Editor:
Pembrolizumab, a humanized monoclonal anti–programmed cell death protein 1 (PD-1) antibody, acts by blocking negative immune regulators such as PD-1.1 Since its approval by the US Food and Drug Administration in 2014, the use of PD-1 inhibitors such as pembrolizumab has dramatically increased, and they are now the standard of care for cancers such as melanoma, lung cancer, and renal cell carcinoma.2,3 With increased use comes a better understanding of the cutaneous adverse effects that may occur. To date, almost 50% of patients treated with PD-1 inhibitors will develop an adverse cutaneous reaction.4 Thus far, cases of patients developing vitiligo, bullous pemphigoid, psoriasis, granulomatous skin reactions, severe cutaneous reactions (ie, toxic epidermal necrolysis), lupus erythematosus, and lichenoid reactions have been described.3,5,6 There are fewer than 30 documented cases of lichenoid reactions due to anti–PD-1 treatment described in the literature, increasing the importance of case reports to demonstrate a full range of cutaneous findings.3 We present a case of a reaction to pembrolizumab with an eruption of lichenoid papules predominantly involving the hands and feet as well as nail changes.
A 60-year-old man with ocular melanoma metastatic to the right lung, transverse colon, and right axillary lymph nodes presented with a chief concern of growing skin lesions present for 6 weeks on the hands and feet. The lesions were tender to the touch and occasionally drained a clear fluid. He also reported nail fragility. Of note, the patient was being treated for metastatic melanoma with pembrolizumab infusions every 3 weeks, which started 6 weeks prior to the onset of the eruption.
Physical examination demonstrated lichenoid papules on the dorsal and ventral aspects of the hands and feet (Figure 1), as well as longitudinal ridging on numerous fingernails and mild koilonychia. A punch biopsy revealed lichenoid interface dermatitis with irregular epidermal hyperplasia (Figure 2). A diagnosis of hypertrophic lichen planus–like drug eruption in response to pembrolizumab was made and clobetasol cream was prescribed.


At 1-month follow-up, the patient reported notable improvement with clobetasol, and he was transitioned to tacrolimus ointment 0.1%. He continued to improve until a month later when he reported new lesions arising a week after a pembrolizumab infusion. He continued to use clobetasol cream for flares and tacrolimus ointment for maintenance.
Almost 3 months after the initial visit, the patient presented with inflammation around his right third fingernail of 1 week’s duration, with more notable fragility than his other nails. No trauma was described, and the nail abnormality was attributed to pembrolizumab. Clobetasol cream and biotin 3 mg daily resulted in improvement, and no other nails were affected in a similar way.
Programmed cell death protein 1 blockers are associated with a variety of adverse events including hypothyroidism, gastrointestinal abnormalities, fatigue, and skin disorders.7 In one study (N=83), cutaneous adverse drug events were found to occur in 42% (35/83) of patients following pembrolizumab therapy, with the most common cutaneous lesions being maculopapular eruptions (29% [24/83]), pruritus (12% [10/83]), and hypopigmentation (8% [7/83]).5
A total of 29 cases of lichenoid dermatitis following anti–PD-1 therapy have been described in the literature.3 Cases range from an eruption of photodistributed hyperkeratotic papules and plaques to hypertrophic vesiculobullous lesions.3,6 Suggested pathophysiology involves blocking the interaction of programmed death ligand 1 on keratinocytes with PD-1 on T cells.3 Management typically includes topical or systemic steroids. Cyclosporine and acitretin also have been successful in a small number of patients. Most patients continue anti–PD-1 treatment with systemic therapy.3
Our patient represents a similar lichenoid eruption; however, the distribution on the dorsal and ventral aspects of the hands and feet as well as nail dystrophy make the presentation unique. Anticancer drugs that increase the T-cell immune response by altering the complex signaling among T cells, antigen-presenting cells, and tumor cells have been associated with cutaneous eruptions. Although the exact mechanism is still not fully understood, clinical suspicion of a pembrolizumab reaction should remain high on the differential in the setting of hyperkeratotic papules in association with anti–PD-1 therapy.
- Homet Moreno B, Ribas A. Anti-programmed cell death protein-1/ligand-1 therapy in different cancers. Br J Cancer. 2015;112:1421-1427.
- Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384:1109-1117.
- Simonsen AB, Kaae J, Elleback E, et al. Cutaneous adverse reactions to anti-PD-1 treatment: a systematic review. J Am Acad Dermatol. 2020;83:1415-1424.
- Hwang SJ, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;151:1206-1212.
- Joseph RW, Cappel M, Goedjen B, et al. Lichenoid dermatitis in three patients with metastatic melanoma treated with anti-PD-1 therapy. Cancer Immunol Res. 2015;3:18-22.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
- Homet Moreno B, Ribas A. Anti-programmed cell death protein-1/ligand-1 therapy in different cancers. Br J Cancer. 2015;112:1421-1427.
- Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384:1109-1117.
- Simonsen AB, Kaae J, Elleback E, et al. Cutaneous adverse reactions to anti-PD-1 treatment: a systematic review. J Am Acad Dermatol. 2020;83:1415-1424.
- Hwang SJ, Carlos G, Wakade D, et al. Cutaneous adverse events (AEs) of anti-programmed cell death (PD)-1 therapy in patients with metastatic melanoma: a single-institution cohort. J Am Acad Dermatol. 2016;74:455-461.
- Sanlorenzo M, Vujic I, Daud A, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;151:1206-1212.
- Joseph RW, Cappel M, Goedjen B, et al. Lichenoid dermatitis in three patients with metastatic melanoma treated with anti-PD-1 therapy. Cancer Immunol Res. 2015;3:18-22.
- Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144.
Practice Points
- With an increased use of immunotherapy medications such as pembrolizumab for various cancers, it is important that dermatologists are aware of the wide range of adverse cutaneous reactions that can occur, including lichenoid reactions.
- Hypertrophic lichen planus should be considered in the differential diagnosis of patients with cutaneous lesions in addition to nail findings developing after starting programmed cell death protein 1 inhibitor therapy.
Does screening for skin cancer result in melanoma overdiagnosis?
When the COVID-19 pandemic first hit, cancer screening in the United States came to an abrupt halt. That experience, coupled with the financial fallout of the pandemic, has led some doctors to reassess business as usual.
In particular, a trio has taken aim at skin cancer screening – arguing that it should stop – in a ‘sounding board’ commentary published online Jan. 7 in the New England Journal of Medicine.
“The COVID-19 pandemic has functionally stopped skin cancer screening; what is important is not to restart it,” wrote the authors, led by H. Gilbert Welch, MD, MPH, at Brigham and Women’s Hospital, Boston, Massachusetts. Dr. Welch has often raised questions about cancer screening and highlighted the issue of overdiagnosis.
In this latest essay, Dr. Welch teamed up with pathologist Benjamin Mazer, MD, Yale University, New Haven, Conn., who writes commentaries for this news organization, and dermatologist Adewole S. Adamson, MD, University of Texas, Austin, to argue that screening for skin cancer has led to an overdiagnosis of melanoma.
However, two melanoma experts pointed out flaws in some of their arguments, and said the issue is more nuanced than they present.
Arguing that melanoma is overdiagnosed
The incidence of melanoma is six times as high as it was 40 years ago, making it the third most common cancer in the United States, the investigators pointed out. However, while case rates have skyrocketed, death rates from melanoma have remained about the same, which points to overdiagnosis.
They described a cycle of increased diagnostic scrutiny that is driving overdiagnosis of melanoma. This includes heightened awareness (perhaps overly) among patients, widespread skin screenings, lower clinical thresholds for biopsy, and lower thresholds among pathologists for diagnosis of melanoma. Fear of missing cancer, legal concerns, and financial incentives may all contribute.
“We view the rise in the incidence of melanoma as a sentinel event, a warning that an epidemic of inspection, surveillance, and biopsy of pigmented skin lesions is permeating through the general population,” they wrote.
Furthermore, overdiagnosis could contribute to unnecessary intervention.
Between 2004 and 2017, rates of biopsy among fee-for-service Medicare recipients almost doubled (from 5% to 8%), according to coding trends data cited in the article. Overdiagnosis and unnecessary intervention could cause psychological, financial, and physical harm to the patient, and the authors argued for interrupting the cycle.
“The most important step to break the cycle of melanoma overdiagnosis is to stop population-wide screening for skin cancer,” they wrote.
The U.S. Preventive Services Task Force currently states that there is insufficient evidence to weigh the balances versus the harms of skin cancer screening, leaving it open to interpretation.
“[T]he increase in melanoma diagnoses by a factor of 6, with at least an order of magnitude more persons undergoing a biopsy and no apparent effect on mortality, is more than enough to recommend against population-wide screening,” Dr. Welch and colleagues concluded.
But the issue may be more nuanced, argued a melanoma expert.
“Everyone agrees that screening high-risk groups has the greatest chance of reducing cancer mortality. In melanoma, the strongest risk factor is the number of moles and presence of clinically atypical moles,” David Polsky, MD, PhD, commented in an interview. Dr. Polsky is a professor of dermatologic oncology at the Perlmutter Cancer Center at New York University Langone Health.
However, population-based studies have shown that at least half of melanoma patients are not considered high risk based on the appearance of the mole, he explained.
“Studies to identify genetic risk factors for melanoma have not yet progressed to the point where these can be tested in the clinic. We clearly have a knowledge gap that needs to be addressed,” he said.
Moreover, it’s not easy to predict which early melanomas will metastasize, said dermatologist Jennifer Stein, MD, PhD, who specializes in treating patients at high risk for melanoma at NYU Langone.
“This paper suggests that it may not be important to detect and treat melanoma in situ, and that the increase in diagnosis of melanoma in situ has led to more harms than good,” she said. “There is evidence that most melanomas do originate as in situ lesions. Unfortunately, we cannot predict which ones will become more aggressive. For this reason, we treat melanoma in situ.”
Taking issue with some of the arguments
Both Dr. Polsky and Dr. Stein took issue with several of the arguments put forward by Dr. Welch and colleagues.
For instance, Dr. Welch and colleagues cited research suggesting that UV light is a weak risk factor for melanoma, but Dr. Polsky disagreed. “There are many lines of evidence ranging from epidemiological, clinical, and biological studies that prove the causative association between ultraviolet light and melanoma, while acknowledging that other factors, such as genetic predisposition, play an important role,” he said. “Since ultraviolet light in the form of outdoor sunburns or indoor tanning exposure are modifiable risk factors, it is important that we continue with our current public messaging on their causal role in the development of melanoma.”
Furthermore, the 2012 study that the authors cited to support their argument that pathologists today are more likely to diagnose melanoma than in years past is flawed, according to Dr. Stein. The study was very small and included just nine contemporary pathologists. Unlike in real life, pathologists in the study could not diagnose lesions as “atypical,” and may have erred on the side of caution by calling them malignant.
“There were multiple limitations to this study that were acknowledged by its authors, who stated that it was a hypothesis-generating study and may not be generalizable,” Dr. Stein said.
In addition, Dr. Polsky took issue with the suggestion that awareness about melanoma among the general public is overly heightened.
“Reducing melanoma awareness would not be wise,” he said. “Studies have shown that awareness of melanoma is associated with the diagnosis of earlier-stage lesions that can be cured by simple skin surgery, without the need for more costly interventions utilized for more advanced melanomas.”
Dr. Mazer reported receiving travel compensation from Hillcrest Healthcare Systems, and is a commentator for this new organization. Dr. Welch has written three books on the subjects of overdiagnosis and testing for cancer. Dr. Adamson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When the COVID-19 pandemic first hit, cancer screening in the United States came to an abrupt halt. That experience, coupled with the financial fallout of the pandemic, has led some doctors to reassess business as usual.
In particular, a trio has taken aim at skin cancer screening – arguing that it should stop – in a ‘sounding board’ commentary published online Jan. 7 in the New England Journal of Medicine.
“The COVID-19 pandemic has functionally stopped skin cancer screening; what is important is not to restart it,” wrote the authors, led by H. Gilbert Welch, MD, MPH, at Brigham and Women’s Hospital, Boston, Massachusetts. Dr. Welch has often raised questions about cancer screening and highlighted the issue of overdiagnosis.
In this latest essay, Dr. Welch teamed up with pathologist Benjamin Mazer, MD, Yale University, New Haven, Conn., who writes commentaries for this news organization, and dermatologist Adewole S. Adamson, MD, University of Texas, Austin, to argue that screening for skin cancer has led to an overdiagnosis of melanoma.
However, two melanoma experts pointed out flaws in some of their arguments, and said the issue is more nuanced than they present.
Arguing that melanoma is overdiagnosed
The incidence of melanoma is six times as high as it was 40 years ago, making it the third most common cancer in the United States, the investigators pointed out. However, while case rates have skyrocketed, death rates from melanoma have remained about the same, which points to overdiagnosis.
They described a cycle of increased diagnostic scrutiny that is driving overdiagnosis of melanoma. This includes heightened awareness (perhaps overly) among patients, widespread skin screenings, lower clinical thresholds for biopsy, and lower thresholds among pathologists for diagnosis of melanoma. Fear of missing cancer, legal concerns, and financial incentives may all contribute.
“We view the rise in the incidence of melanoma as a sentinel event, a warning that an epidemic of inspection, surveillance, and biopsy of pigmented skin lesions is permeating through the general population,” they wrote.
Furthermore, overdiagnosis could contribute to unnecessary intervention.
Between 2004 and 2017, rates of biopsy among fee-for-service Medicare recipients almost doubled (from 5% to 8%), according to coding trends data cited in the article. Overdiagnosis and unnecessary intervention could cause psychological, financial, and physical harm to the patient, and the authors argued for interrupting the cycle.
“The most important step to break the cycle of melanoma overdiagnosis is to stop population-wide screening for skin cancer,” they wrote.
The U.S. Preventive Services Task Force currently states that there is insufficient evidence to weigh the balances versus the harms of skin cancer screening, leaving it open to interpretation.
“[T]he increase in melanoma diagnoses by a factor of 6, with at least an order of magnitude more persons undergoing a biopsy and no apparent effect on mortality, is more than enough to recommend against population-wide screening,” Dr. Welch and colleagues concluded.
But the issue may be more nuanced, argued a melanoma expert.
“Everyone agrees that screening high-risk groups has the greatest chance of reducing cancer mortality. In melanoma, the strongest risk factor is the number of moles and presence of clinically atypical moles,” David Polsky, MD, PhD, commented in an interview. Dr. Polsky is a professor of dermatologic oncology at the Perlmutter Cancer Center at New York University Langone Health.
However, population-based studies have shown that at least half of melanoma patients are not considered high risk based on the appearance of the mole, he explained.
“Studies to identify genetic risk factors for melanoma have not yet progressed to the point where these can be tested in the clinic. We clearly have a knowledge gap that needs to be addressed,” he said.
Moreover, it’s not easy to predict which early melanomas will metastasize, said dermatologist Jennifer Stein, MD, PhD, who specializes in treating patients at high risk for melanoma at NYU Langone.
“This paper suggests that it may not be important to detect and treat melanoma in situ, and that the increase in diagnosis of melanoma in situ has led to more harms than good,” she said. “There is evidence that most melanomas do originate as in situ lesions. Unfortunately, we cannot predict which ones will become more aggressive. For this reason, we treat melanoma in situ.”
Taking issue with some of the arguments
Both Dr. Polsky and Dr. Stein took issue with several of the arguments put forward by Dr. Welch and colleagues.
For instance, Dr. Welch and colleagues cited research suggesting that UV light is a weak risk factor for melanoma, but Dr. Polsky disagreed. “There are many lines of evidence ranging from epidemiological, clinical, and biological studies that prove the causative association between ultraviolet light and melanoma, while acknowledging that other factors, such as genetic predisposition, play an important role,” he said. “Since ultraviolet light in the form of outdoor sunburns or indoor tanning exposure are modifiable risk factors, it is important that we continue with our current public messaging on their causal role in the development of melanoma.”
Furthermore, the 2012 study that the authors cited to support their argument that pathologists today are more likely to diagnose melanoma than in years past is flawed, according to Dr. Stein. The study was very small and included just nine contemporary pathologists. Unlike in real life, pathologists in the study could not diagnose lesions as “atypical,” and may have erred on the side of caution by calling them malignant.
“There were multiple limitations to this study that were acknowledged by its authors, who stated that it was a hypothesis-generating study and may not be generalizable,” Dr. Stein said.
In addition, Dr. Polsky took issue with the suggestion that awareness about melanoma among the general public is overly heightened.
“Reducing melanoma awareness would not be wise,” he said. “Studies have shown that awareness of melanoma is associated with the diagnosis of earlier-stage lesions that can be cured by simple skin surgery, without the need for more costly interventions utilized for more advanced melanomas.”
Dr. Mazer reported receiving travel compensation from Hillcrest Healthcare Systems, and is a commentator for this new organization. Dr. Welch has written three books on the subjects of overdiagnosis and testing for cancer. Dr. Adamson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When the COVID-19 pandemic first hit, cancer screening in the United States came to an abrupt halt. That experience, coupled with the financial fallout of the pandemic, has led some doctors to reassess business as usual.
In particular, a trio has taken aim at skin cancer screening – arguing that it should stop – in a ‘sounding board’ commentary published online Jan. 7 in the New England Journal of Medicine.
“The COVID-19 pandemic has functionally stopped skin cancer screening; what is important is not to restart it,” wrote the authors, led by H. Gilbert Welch, MD, MPH, at Brigham and Women’s Hospital, Boston, Massachusetts. Dr. Welch has often raised questions about cancer screening and highlighted the issue of overdiagnosis.
In this latest essay, Dr. Welch teamed up with pathologist Benjamin Mazer, MD, Yale University, New Haven, Conn., who writes commentaries for this news organization, and dermatologist Adewole S. Adamson, MD, University of Texas, Austin, to argue that screening for skin cancer has led to an overdiagnosis of melanoma.
However, two melanoma experts pointed out flaws in some of their arguments, and said the issue is more nuanced than they present.
Arguing that melanoma is overdiagnosed
The incidence of melanoma is six times as high as it was 40 years ago, making it the third most common cancer in the United States, the investigators pointed out. However, while case rates have skyrocketed, death rates from melanoma have remained about the same, which points to overdiagnosis.
They described a cycle of increased diagnostic scrutiny that is driving overdiagnosis of melanoma. This includes heightened awareness (perhaps overly) among patients, widespread skin screenings, lower clinical thresholds for biopsy, and lower thresholds among pathologists for diagnosis of melanoma. Fear of missing cancer, legal concerns, and financial incentives may all contribute.
“We view the rise in the incidence of melanoma as a sentinel event, a warning that an epidemic of inspection, surveillance, and biopsy of pigmented skin lesions is permeating through the general population,” they wrote.
Furthermore, overdiagnosis could contribute to unnecessary intervention.
Between 2004 and 2017, rates of biopsy among fee-for-service Medicare recipients almost doubled (from 5% to 8%), according to coding trends data cited in the article. Overdiagnosis and unnecessary intervention could cause psychological, financial, and physical harm to the patient, and the authors argued for interrupting the cycle.
“The most important step to break the cycle of melanoma overdiagnosis is to stop population-wide screening for skin cancer,” they wrote.
The U.S. Preventive Services Task Force currently states that there is insufficient evidence to weigh the balances versus the harms of skin cancer screening, leaving it open to interpretation.
“[T]he increase in melanoma diagnoses by a factor of 6, with at least an order of magnitude more persons undergoing a biopsy and no apparent effect on mortality, is more than enough to recommend against population-wide screening,” Dr. Welch and colleagues concluded.
But the issue may be more nuanced, argued a melanoma expert.
“Everyone agrees that screening high-risk groups has the greatest chance of reducing cancer mortality. In melanoma, the strongest risk factor is the number of moles and presence of clinically atypical moles,” David Polsky, MD, PhD, commented in an interview. Dr. Polsky is a professor of dermatologic oncology at the Perlmutter Cancer Center at New York University Langone Health.
However, population-based studies have shown that at least half of melanoma patients are not considered high risk based on the appearance of the mole, he explained.
“Studies to identify genetic risk factors for melanoma have not yet progressed to the point where these can be tested in the clinic. We clearly have a knowledge gap that needs to be addressed,” he said.
Moreover, it’s not easy to predict which early melanomas will metastasize, said dermatologist Jennifer Stein, MD, PhD, who specializes in treating patients at high risk for melanoma at NYU Langone.
“This paper suggests that it may not be important to detect and treat melanoma in situ, and that the increase in diagnosis of melanoma in situ has led to more harms than good,” she said. “There is evidence that most melanomas do originate as in situ lesions. Unfortunately, we cannot predict which ones will become more aggressive. For this reason, we treat melanoma in situ.”
Taking issue with some of the arguments
Both Dr. Polsky and Dr. Stein took issue with several of the arguments put forward by Dr. Welch and colleagues.
For instance, Dr. Welch and colleagues cited research suggesting that UV light is a weak risk factor for melanoma, but Dr. Polsky disagreed. “There are many lines of evidence ranging from epidemiological, clinical, and biological studies that prove the causative association between ultraviolet light and melanoma, while acknowledging that other factors, such as genetic predisposition, play an important role,” he said. “Since ultraviolet light in the form of outdoor sunburns or indoor tanning exposure are modifiable risk factors, it is important that we continue with our current public messaging on their causal role in the development of melanoma.”
Furthermore, the 2012 study that the authors cited to support their argument that pathologists today are more likely to diagnose melanoma than in years past is flawed, according to Dr. Stein. The study was very small and included just nine contemporary pathologists. Unlike in real life, pathologists in the study could not diagnose lesions as “atypical,” and may have erred on the side of caution by calling them malignant.
“There were multiple limitations to this study that were acknowledged by its authors, who stated that it was a hypothesis-generating study and may not be generalizable,” Dr. Stein said.
In addition, Dr. Polsky took issue with the suggestion that awareness about melanoma among the general public is overly heightened.
“Reducing melanoma awareness would not be wise,” he said. “Studies have shown that awareness of melanoma is associated with the diagnosis of earlier-stage lesions that can be cured by simple skin surgery, without the need for more costly interventions utilized for more advanced melanomas.”
Dr. Mazer reported receiving travel compensation from Hillcrest Healthcare Systems, and is a commentator for this new organization. Dr. Welch has written three books on the subjects of overdiagnosis and testing for cancer. Dr. Adamson disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adjuvant nivolumab plus ipilimumab shows strong results in resected stage IV melanoma
Results of the
IMMUNED was a multicenter German double-blind, placebo-controlled, phase 2 trial conducted by the Dermatologic Cooperative Oncology Group. It included 167 patients with resected stage IV melanoma and no evidence of disease who were randomized to adjuvant nivolumab (Opdivo) plus placebo, nivolumab plus ipilimumab (Yervoy), or double placebo, with relapse-free survival as the primary outcome, Merrick I. Ross, MD, explained at a forum on cutaneous malignancies jointly presented by Postgraduate Institute for Medicine and Global Academy for Medical Education.
“The patients who received adjuvant ipilimumab and nivolumab had amazing 24-month outcomes: a relapse-free survival of 70% versus 42% with nivolumab and 14% with placebo,” observed Dr. Ross, professor of surgical oncology and chief of the melanoma section at the University of Texas M.D. Anderson Cancer Center, Houston.
“It’s not a long-term survival outcome, but we’ll see what happens long term. This could be a very interesting approach to move forward with,” he commented.
By way of background, the cancer surgeon noted that nivolumab has achieved standard-of-care status as adjuvant immunotherapy in patients with resected stage IIIB-C and stage IV melanoma, largely on the strength of the CheckMate-238 trial, which randomized 906 such patients at 130 academic centers in 25 countries to 1 year of adjuvant therapy with either intravenous nivolumab or ipilimumab. In the study, nivolumab emerged as the clear winner, with a 4-year recurrence-free survival of 51.7%, compared with 41.2% for ipilimumab, for a 29% relative risk reduction. Ipilimumab was associated with greater toxicity.
The between-group difference in relapse-free survival in the overall study population also held true in the subgroup comprised of 169 CheckMate 238 participants with resected stage IV melanoma and no evidence of disease at enrollment, Dr. Ross noted.
In the IMMUNED trial, the superior outcome achieved with adjuvant nivolumab plus ipilimumab came at the cost of significantly greater toxicity than with nivolumab alone. Treatment-related adverse events led to medication discontinuation in 62% of the dual-adjuvant therapy group, compared with 13% of those on adjuvant nivolumab.
IMMUNED was funded by Bristol-Myers Squibb.
Dr. Ross reported having no financial conflicts regarding his presentation.
Global Academy for Medical Education and this news organization are owned by the same company.
Results of the
IMMUNED was a multicenter German double-blind, placebo-controlled, phase 2 trial conducted by the Dermatologic Cooperative Oncology Group. It included 167 patients with resected stage IV melanoma and no evidence of disease who were randomized to adjuvant nivolumab (Opdivo) plus placebo, nivolumab plus ipilimumab (Yervoy), or double placebo, with relapse-free survival as the primary outcome, Merrick I. Ross, MD, explained at a forum on cutaneous malignancies jointly presented by Postgraduate Institute for Medicine and Global Academy for Medical Education.
“The patients who received adjuvant ipilimumab and nivolumab had amazing 24-month outcomes: a relapse-free survival of 70% versus 42% with nivolumab and 14% with placebo,” observed Dr. Ross, professor of surgical oncology and chief of the melanoma section at the University of Texas M.D. Anderson Cancer Center, Houston.
“It’s not a long-term survival outcome, but we’ll see what happens long term. This could be a very interesting approach to move forward with,” he commented.
By way of background, the cancer surgeon noted that nivolumab has achieved standard-of-care status as adjuvant immunotherapy in patients with resected stage IIIB-C and stage IV melanoma, largely on the strength of the CheckMate-238 trial, which randomized 906 such patients at 130 academic centers in 25 countries to 1 year of adjuvant therapy with either intravenous nivolumab or ipilimumab. In the study, nivolumab emerged as the clear winner, with a 4-year recurrence-free survival of 51.7%, compared with 41.2% for ipilimumab, for a 29% relative risk reduction. Ipilimumab was associated with greater toxicity.
The between-group difference in relapse-free survival in the overall study population also held true in the subgroup comprised of 169 CheckMate 238 participants with resected stage IV melanoma and no evidence of disease at enrollment, Dr. Ross noted.
In the IMMUNED trial, the superior outcome achieved with adjuvant nivolumab plus ipilimumab came at the cost of significantly greater toxicity than with nivolumab alone. Treatment-related adverse events led to medication discontinuation in 62% of the dual-adjuvant therapy group, compared with 13% of those on adjuvant nivolumab.
IMMUNED was funded by Bristol-Myers Squibb.
Dr. Ross reported having no financial conflicts regarding his presentation.
Global Academy for Medical Education and this news organization are owned by the same company.
Results of the
IMMUNED was a multicenter German double-blind, placebo-controlled, phase 2 trial conducted by the Dermatologic Cooperative Oncology Group. It included 167 patients with resected stage IV melanoma and no evidence of disease who were randomized to adjuvant nivolumab (Opdivo) plus placebo, nivolumab plus ipilimumab (Yervoy), or double placebo, with relapse-free survival as the primary outcome, Merrick I. Ross, MD, explained at a forum on cutaneous malignancies jointly presented by Postgraduate Institute for Medicine and Global Academy for Medical Education.
“The patients who received adjuvant ipilimumab and nivolumab had amazing 24-month outcomes: a relapse-free survival of 70% versus 42% with nivolumab and 14% with placebo,” observed Dr. Ross, professor of surgical oncology and chief of the melanoma section at the University of Texas M.D. Anderson Cancer Center, Houston.
“It’s not a long-term survival outcome, but we’ll see what happens long term. This could be a very interesting approach to move forward with,” he commented.
By way of background, the cancer surgeon noted that nivolumab has achieved standard-of-care status as adjuvant immunotherapy in patients with resected stage IIIB-C and stage IV melanoma, largely on the strength of the CheckMate-238 trial, which randomized 906 such patients at 130 academic centers in 25 countries to 1 year of adjuvant therapy with either intravenous nivolumab or ipilimumab. In the study, nivolumab emerged as the clear winner, with a 4-year recurrence-free survival of 51.7%, compared with 41.2% for ipilimumab, for a 29% relative risk reduction. Ipilimumab was associated with greater toxicity.
The between-group difference in relapse-free survival in the overall study population also held true in the subgroup comprised of 169 CheckMate 238 participants with resected stage IV melanoma and no evidence of disease at enrollment, Dr. Ross noted.
In the IMMUNED trial, the superior outcome achieved with adjuvant nivolumab plus ipilimumab came at the cost of significantly greater toxicity than with nivolumab alone. Treatment-related adverse events led to medication discontinuation in 62% of the dual-adjuvant therapy group, compared with 13% of those on adjuvant nivolumab.
IMMUNED was funded by Bristol-Myers Squibb.
Dr. Ross reported having no financial conflicts regarding his presentation.
Global Academy for Medical Education and this news organization are owned by the same company.
Telltale dermoscopic features of melanomas lacking pigment reviewed
by familiarity with a handful of dermoscopic features specific to melanomas lacking significant pigment, Steven Q. Wang, MD, said at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
These features emerged from a major study conducted on five continents by members of the International Dermoscopy Society. The investigators developed a simple, eight-variable model, which demonstrated a sensitivity of 70% and specificity of 56% for diagnosis of melanoma. And while that’s a markedly worse performance than when dermoscopy is used for detection of pigmented melanomas, where sensitivities in excess of 90% and specificities greater than 70% are typical, it’s nonetheless a significant improvement over naked-eye evaluation of these challenging pigment-deprived melanomas, noted Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan Kettering Basking Ridge (N.J.)
Using the predictive model developed in the international study to evaluate lesions lacking pigment, a diagnosis of melanoma is made provided two conditions are met: The lesion can have no more than three milia-like cysts, and it has to possess one or more of seven positive dermoscopic findings. The strongest predictor of melanoma in the study was the presence of a blue-white veil, which in univariate analysis was associated with a 13-fold increased likelihood of melanoma.
The other positive predictors were irregularly shaped depigmentation, more than one shade of pink, predominant central vessels, irregularly sized or distributed brown dots or globules, multiple blue-gray dots, and dotted and linear irregular vessels.
Dr. Wang emphasized that, when dermoscopy and clinical skin examination of a featureless hypomelanotic or amelanotic lesion yield ambiguous findings, frequent vigilant follow-up is a viable strategy to detect early melanoma – provided the lesion is superficial.
“The reality is not all melanomas are the same. The superficial spreading melanomas and lentigo melanomas grow very, very slowly: less than 0.1 mm per month. Those are the types of lesions you can monitor. But there is one type of lesion you should never, ever monitor: nodular lesions. They are the type of lesions that can do your patient harm because nodular melanomas can grow really fast. So my key takeaway message is, if you see a nodule and you don’t know what it is, take it off,” the dermatologist said.
Dermoscopy in the hands of experienced users has repeatedly been shown to improve diagnostic accuracy by more than 25%. But there is an additional very important reason to embrace dermoscopy in daily clinical practice, according to Dr. Wang: “When you put the scope on an individual, you slow down the exam and patients feels like you’re paying more attention to them.”
That’s worthwhile because the No. 1 complaint voiced by patients who make their way to Sloan Kettering for a second opinion is that their prior skin examination by an outside physician wasn’t thorough. They’re often angry about it. And while it’s true that incorporating dermoscopy does make for a lengthier skin examination, the additional time involved is actually minimal. Dr. Wang cited a randomized, prospective, multicenter study which documented that the median time required to conduct a thorough complete skin examination without dermoscopy was 70 seconds versus 142 seconds with dermoscopy.
Dr. Wang reported having no financial conflicts regarding his presentation.
MedscapeLive and this news organization are owned by the same parent company.
by familiarity with a handful of dermoscopic features specific to melanomas lacking significant pigment, Steven Q. Wang, MD, said at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
These features emerged from a major study conducted on five continents by members of the International Dermoscopy Society. The investigators developed a simple, eight-variable model, which demonstrated a sensitivity of 70% and specificity of 56% for diagnosis of melanoma. And while that’s a markedly worse performance than when dermoscopy is used for detection of pigmented melanomas, where sensitivities in excess of 90% and specificities greater than 70% are typical, it’s nonetheless a significant improvement over naked-eye evaluation of these challenging pigment-deprived melanomas, noted Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan Kettering Basking Ridge (N.J.)
Using the predictive model developed in the international study to evaluate lesions lacking pigment, a diagnosis of melanoma is made provided two conditions are met: The lesion can have no more than three milia-like cysts, and it has to possess one or more of seven positive dermoscopic findings. The strongest predictor of melanoma in the study was the presence of a blue-white veil, which in univariate analysis was associated with a 13-fold increased likelihood of melanoma.
The other positive predictors were irregularly shaped depigmentation, more than one shade of pink, predominant central vessels, irregularly sized or distributed brown dots or globules, multiple blue-gray dots, and dotted and linear irregular vessels.
Dr. Wang emphasized that, when dermoscopy and clinical skin examination of a featureless hypomelanotic or amelanotic lesion yield ambiguous findings, frequent vigilant follow-up is a viable strategy to detect early melanoma – provided the lesion is superficial.
“The reality is not all melanomas are the same. The superficial spreading melanomas and lentigo melanomas grow very, very slowly: less than 0.1 mm per month. Those are the types of lesions you can monitor. But there is one type of lesion you should never, ever monitor: nodular lesions. They are the type of lesions that can do your patient harm because nodular melanomas can grow really fast. So my key takeaway message is, if you see a nodule and you don’t know what it is, take it off,” the dermatologist said.
Dermoscopy in the hands of experienced users has repeatedly been shown to improve diagnostic accuracy by more than 25%. But there is an additional very important reason to embrace dermoscopy in daily clinical practice, according to Dr. Wang: “When you put the scope on an individual, you slow down the exam and patients feels like you’re paying more attention to them.”
That’s worthwhile because the No. 1 complaint voiced by patients who make their way to Sloan Kettering for a second opinion is that their prior skin examination by an outside physician wasn’t thorough. They’re often angry about it. And while it’s true that incorporating dermoscopy does make for a lengthier skin examination, the additional time involved is actually minimal. Dr. Wang cited a randomized, prospective, multicenter study which documented that the median time required to conduct a thorough complete skin examination without dermoscopy was 70 seconds versus 142 seconds with dermoscopy.
Dr. Wang reported having no financial conflicts regarding his presentation.
MedscapeLive and this news organization are owned by the same parent company.
by familiarity with a handful of dermoscopic features specific to melanomas lacking significant pigment, Steven Q. Wang, MD, said at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
These features emerged from a major study conducted on five continents by members of the International Dermoscopy Society. The investigators developed a simple, eight-variable model, which demonstrated a sensitivity of 70% and specificity of 56% for diagnosis of melanoma. And while that’s a markedly worse performance than when dermoscopy is used for detection of pigmented melanomas, where sensitivities in excess of 90% and specificities greater than 70% are typical, it’s nonetheless a significant improvement over naked-eye evaluation of these challenging pigment-deprived melanomas, noted Dr. Wang, director of dermatologic surgery and dermatology at Memorial Sloan Kettering Basking Ridge (N.J.)
Using the predictive model developed in the international study to evaluate lesions lacking pigment, a diagnosis of melanoma is made provided two conditions are met: The lesion can have no more than three milia-like cysts, and it has to possess one or more of seven positive dermoscopic findings. The strongest predictor of melanoma in the study was the presence of a blue-white veil, which in univariate analysis was associated with a 13-fold increased likelihood of melanoma.
The other positive predictors were irregularly shaped depigmentation, more than one shade of pink, predominant central vessels, irregularly sized or distributed brown dots or globules, multiple blue-gray dots, and dotted and linear irregular vessels.
Dr. Wang emphasized that, when dermoscopy and clinical skin examination of a featureless hypomelanotic or amelanotic lesion yield ambiguous findings, frequent vigilant follow-up is a viable strategy to detect early melanoma – provided the lesion is superficial.
“The reality is not all melanomas are the same. The superficial spreading melanomas and lentigo melanomas grow very, very slowly: less than 0.1 mm per month. Those are the types of lesions you can monitor. But there is one type of lesion you should never, ever monitor: nodular lesions. They are the type of lesions that can do your patient harm because nodular melanomas can grow really fast. So my key takeaway message is, if you see a nodule and you don’t know what it is, take it off,” the dermatologist said.
Dermoscopy in the hands of experienced users has repeatedly been shown to improve diagnostic accuracy by more than 25%. But there is an additional very important reason to embrace dermoscopy in daily clinical practice, according to Dr. Wang: “When you put the scope on an individual, you slow down the exam and patients feels like you’re paying more attention to them.”
That’s worthwhile because the No. 1 complaint voiced by patients who make their way to Sloan Kettering for a second opinion is that their prior skin examination by an outside physician wasn’t thorough. They’re often angry about it. And while it’s true that incorporating dermoscopy does make for a lengthier skin examination, the additional time involved is actually minimal. Dr. Wang cited a randomized, prospective, multicenter study which documented that the median time required to conduct a thorough complete skin examination without dermoscopy was 70 seconds versus 142 seconds with dermoscopy.
Dr. Wang reported having no financial conflicts regarding his presentation.
MedscapeLive and this news organization are owned by the same parent company.
FROM MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR
Skin Cancer Management During the COVID-19 Pandemic
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome novel coronavirus 2 (SARS-CoV-2), has presented a unique challenge to providing essential care to patients. Increased demand for health care workers and medical supplies, in addition to the risk for COVID-19 infection and asymptomatic transmission of SARS-CoV-2 among health care workers and patients, prompted the delay of nonessential services during the surge of cases this summer.1 Key considerations for continuing operation included current and projected COVID-19 cases in the region, ability to implement telehealth, staffing availability, personal protective equipment availability, and office capacity.2 Providing care that is deemed essential often was determined by the urgency of the treatment or service.
The Centers for Medicare & Medicaid Services outlined a strategy to stratify patients, based on level of acuity, during the COVID-19 surge3:
- Low-acuity treatments or services: includes routine primary, specialty, or preventive care visits. They should be postponed; telehealth follow-ups should be considered.
- Intermediate-acuity treatments or services: includes pediatric and neonatal care, follow-up visits for existing conditions, and evaluation of new symptoms (including those consistent with COVID-19). These services should initially be evaluated using telehealth, then triaged to the appropriate site and level of care.
- High-acuity treatments or services: address symptoms consistent with COVID-19 or other severe disease, of which the lack of in-person evaluation would result in harm to the patient.
Employees in hospitals and health care clinics were classified as essential, but dermatologists were not given explicit direction regarding clinic operation. Many practices have restricted services, especially those in an area of higher COVID-19 prevalence. However, the challenge of determining day-to-day operation may have been left to the provider in most cases.4 As many states in the United States continue to relax restrictions, total cases and the rate of positivity of COVID-19 have been sharply rising again, after months of decline,5 which suggests increased transmission of SARS-CoV-2 and potential resurgence of the high case burden on our health care system. Furthermore, a lack of a widely distributed vaccine or herd immunity suggests we will need to take many of the same precautions as in the first surge.6
In general, patients with cancer have been found to be at greater risk for adverse outcomes and mortality after COVID-19.7 Therefore, resource rationing is particularly concerning for patients with skin cancer, including melanoma, Merkel cell carcinoma, mycosis fungoides, and keratinocyte carcinoma. Triaging patients based on level of acuity, type of skin cancer, disease burden, host immunosuppression, and risk for progression must be carefully considered in this population.2 Treatment and follow-up present additional challenges.
Guidelines provided by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) elaborated on key considerations for the treatment of melanoma, keratinocyte carcinoma, and Merkel cell carcinoma during the COVID-19 pandemic.8-10 Guidelines from the NCCN concentrated on clear divisions between disease stages to determine provider response. Guidelines for melanoma patients proposed by the ESMO assign tiers by value-based priority in various treatment settings, which offered flexibility to providers as the COVID-19 landscape continued to change. Recommendations from the NCCN and ESMO are summarized in Tables 1 to 5.



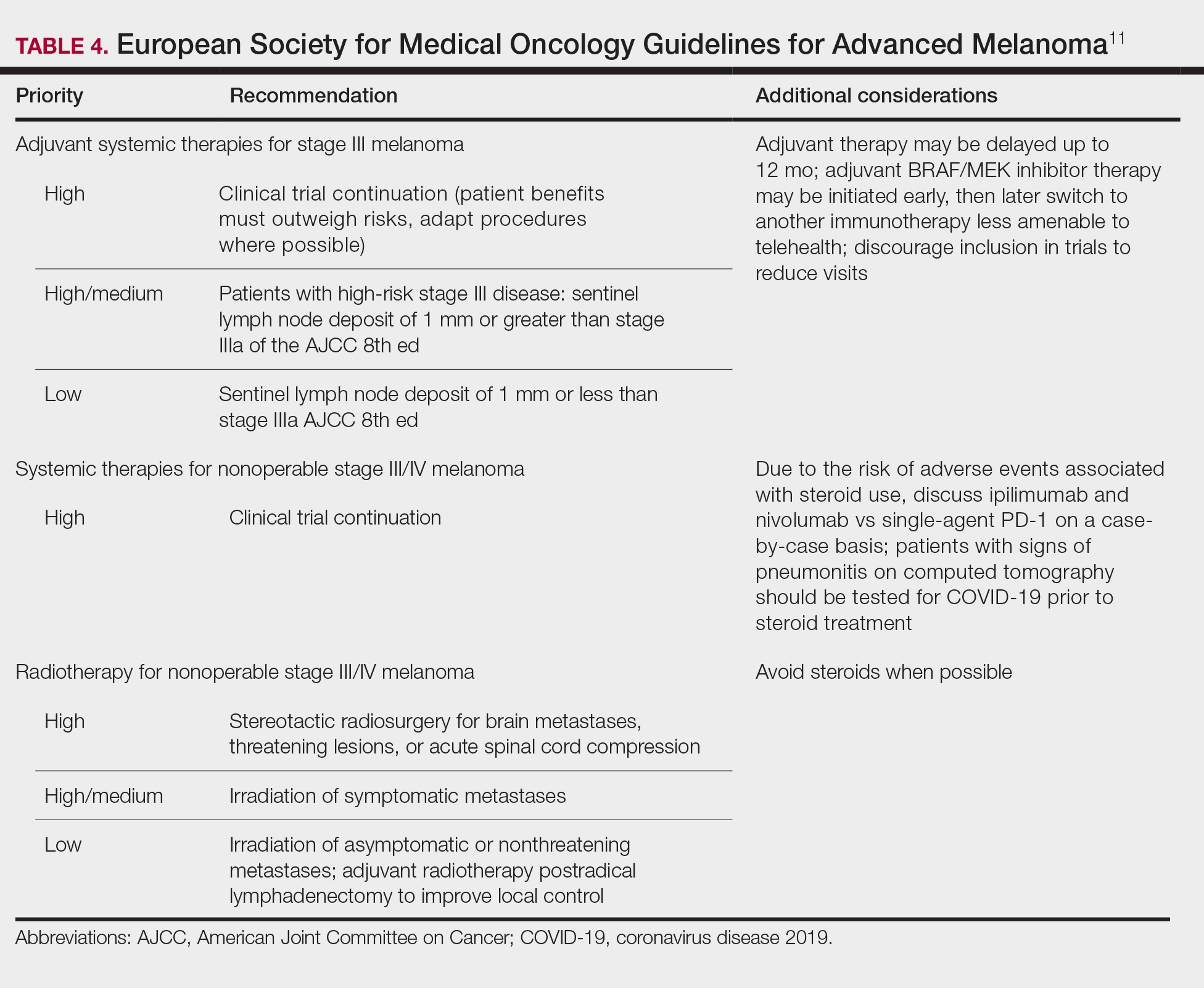

Although these guidelines initially may have been proposed to delay treatment of lower-acuity tumors, such delay might not be feasible given the unknown duration of this pandemic and future disease waves. One review of several studies, which addressed the outcomes on melanoma survival following the surgical delay recommended by the NCCN, revealed contradictory evidence.12 Further, sufficiently powered studies will be needed to better understand the impact of delaying treatment during the summer COVID-19 surge on patients with skin cancer. Therefore, physicians must triage patients accordingly to manage and treat while also preventing disease spread.
Tips for Performing Dermatologic Surgery
Careful consideration should be made to protect both the patient and staff during office-based excisional surgery during the COVID-19 pandemic. To minimize the risk of transmission of SARS-CoV-2, patients and staff should (1) be screened for symptoms of COVID-19 at least 48 hours prior to entering the office via telephone screening questions, and (2) follow proper hygiene and contact procedures once entering the office. Consider obtaining a nasal polymerase chain reaction swab or saliva test 48 hours prior to the procedure if the patient is undergoing a head and neck procedure or there is risk for transmission.
Guidelines from the ESMO recommended that all patients undergoing surgery or therapy should be swabbed for SARS-CoV-2 before each treatment.11 Patients should wear a mask, remain 6-feet apart in the waiting room, and avoid touching objects until they enter the procedure room. Objects that the patient must touch, such as pens, should be cleaned immediately after such contact with either alcohol or soap and water for 20 seconds.
Office capacity should be reduced by allowing no more than 1 person to accompany the patient and ensuring the presence of only the minimum staff needed for the procedure. Staff who are deemed necessary should wear a mask continuously and gloves during patient contact.
Once in the procedure room, providers might be at elevated risk of contracting COVID-19 or transmitting SARS-CoV-2. A properly fitted N95 respirator and a face shield are recommended, especially for facial cases. N95 respirators can be reused by following the latest Centers for Disease Control and Prevention recommendations for reuse and decontamination techniques,13 which may include protecting the N95 respirator with a surgical mask and storing it in a paper bag when not in use. Consider testing asymptomatic patients in facial cases when they cannot wear a mask.
Steps should be taken to reduce in-person visits. Dissolving sutures can help avoid return visits. Follow-up visits and postprocedural questions should be managed by telehealth. However, patients with a high-risk underlying conditions (eg, posttransplantation, immunosuppressed) should continue to obtain regular skin checks because they are at higher risk for more aggressive malignancies, such as Merkel cell carcinoma.
Conclusion
The future trajectory of the COVID-19 pandemic is uncertain. Dermatologists should continue providing care for patients with skin cancer while mitigating the risk for COVID-19 infection and transmission of SARS-CoV-2. Guidelines provided by the NCCN and ESMO should help providers triage patients. Decisions should be made case by case, keeping in mind the availability of resources and practicing in compliance with local guidance.
- Moletta L, Pierobon ES, Capovilla G, et al. International guidelines and recommendations for surgery during COVID-19 pandemic: a systematic review. Int J Surg. 2020;79:180-188.
- Ueda M, Martins R, Hendrie PC, et al. Managing cancer care during the COVID-19 pandemic: agility and collaboration toward common goal. J Natl Compr Canc Netw. 2020:1-4.
- Center for Medicare & Medicaid Services. Non-emergent, elective medical services, and treatment recommendations. Published April 7, 2020. Accessed October 15, 2020. https://www.cms.gov/files/document/cms-non-emergent-elective-medical-recommendations.pdf
- Muddasani S, Housholder A, Fleischer AB. An assessment of United States dermatology practices during the COVID-19 outbreak. J Dermatolog Treat. 2020;31:436-438.
- Coronavirus Resource Center, Johns Hopkins University & Medicine. Rate of positive tests in the US and states over time. Updated December 11, 2020. Accessed December 11, 2020. https://coronavirus.jhu.edu/testing/individual-states
- Middleton J, Lopes H, Michelson K, et al. Planning for a second wave pandemic of COVID-19 and planning for winter: a statement from the Association of Schools of Public Health in the European Region. Int J Public Health. 2020;65:1525-1527.
- Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335-337.
- National Comprehensive Cancer Network. Advisory statement for non-melanoma skin cancer care during the COVID-19 pandemic (version 4). Published May 22, 2020. Accessed December 11, 2020. https://www.nccn.org/covid-19/pdf/NCCN-NMSC.pdf
National Comprehensive Cancer Network. Short-term recommendations for cutaneous melanoma management during COVID-19 pandemic (version 3). Published May 6, 2020. Accessed December 11, 2020. www.nccn.org/covid-19/pdf/Melanoma.pdf - Conforti C, Giuffrida R, Di Meo N, et al. Management of advanced melanoma in the COVID-19 era. Dermatol Ther. 2020;33:e13444.
- ESMO [European Society for Medical Oncology]. Cancer patient management during the COVID-19 pandemic. Accessed Decemeber 11, 2020. https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic?hit=ehp
- Guhan S, Boland G, Tanabe K, et al. Surgical delay and mortality for primary cutaneous melanoma [published online July 22, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.07.078
- Centers for Disease Control and Prevention. Implementing filtering facepiece respirator (FFR) reuse, including reuse after decontamination, when there are known shortages of N95 respirators. Updated October 19, 2020. Accessed December 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/decontamination-reuse-respirators.html
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome novel coronavirus 2 (SARS-CoV-2), has presented a unique challenge to providing essential care to patients. Increased demand for health care workers and medical supplies, in addition to the risk for COVID-19 infection and asymptomatic transmission of SARS-CoV-2 among health care workers and patients, prompted the delay of nonessential services during the surge of cases this summer.1 Key considerations for continuing operation included current and projected COVID-19 cases in the region, ability to implement telehealth, staffing availability, personal protective equipment availability, and office capacity.2 Providing care that is deemed essential often was determined by the urgency of the treatment or service.
The Centers for Medicare & Medicaid Services outlined a strategy to stratify patients, based on level of acuity, during the COVID-19 surge3:
- Low-acuity treatments or services: includes routine primary, specialty, or preventive care visits. They should be postponed; telehealth follow-ups should be considered.
- Intermediate-acuity treatments or services: includes pediatric and neonatal care, follow-up visits for existing conditions, and evaluation of new symptoms (including those consistent with COVID-19). These services should initially be evaluated using telehealth, then triaged to the appropriate site and level of care.
- High-acuity treatments or services: address symptoms consistent with COVID-19 or other severe disease, of which the lack of in-person evaluation would result in harm to the patient.
Employees in hospitals and health care clinics were classified as essential, but dermatologists were not given explicit direction regarding clinic operation. Many practices have restricted services, especially those in an area of higher COVID-19 prevalence. However, the challenge of determining day-to-day operation may have been left to the provider in most cases.4 As many states in the United States continue to relax restrictions, total cases and the rate of positivity of COVID-19 have been sharply rising again, after months of decline,5 which suggests increased transmission of SARS-CoV-2 and potential resurgence of the high case burden on our health care system. Furthermore, a lack of a widely distributed vaccine or herd immunity suggests we will need to take many of the same precautions as in the first surge.6
In general, patients with cancer have been found to be at greater risk for adverse outcomes and mortality after COVID-19.7 Therefore, resource rationing is particularly concerning for patients with skin cancer, including melanoma, Merkel cell carcinoma, mycosis fungoides, and keratinocyte carcinoma. Triaging patients based on level of acuity, type of skin cancer, disease burden, host immunosuppression, and risk for progression must be carefully considered in this population.2 Treatment and follow-up present additional challenges.
Guidelines provided by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) elaborated on key considerations for the treatment of melanoma, keratinocyte carcinoma, and Merkel cell carcinoma during the COVID-19 pandemic.8-10 Guidelines from the NCCN concentrated on clear divisions between disease stages to determine provider response. Guidelines for melanoma patients proposed by the ESMO assign tiers by value-based priority in various treatment settings, which offered flexibility to providers as the COVID-19 landscape continued to change. Recommendations from the NCCN and ESMO are summarized in Tables 1 to 5.





Although these guidelines initially may have been proposed to delay treatment of lower-acuity tumors, such delay might not be feasible given the unknown duration of this pandemic and future disease waves. One review of several studies, which addressed the outcomes on melanoma survival following the surgical delay recommended by the NCCN, revealed contradictory evidence.12 Further, sufficiently powered studies will be needed to better understand the impact of delaying treatment during the summer COVID-19 surge on patients with skin cancer. Therefore, physicians must triage patients accordingly to manage and treat while also preventing disease spread.
Tips for Performing Dermatologic Surgery
Careful consideration should be made to protect both the patient and staff during office-based excisional surgery during the COVID-19 pandemic. To minimize the risk of transmission of SARS-CoV-2, patients and staff should (1) be screened for symptoms of COVID-19 at least 48 hours prior to entering the office via telephone screening questions, and (2) follow proper hygiene and contact procedures once entering the office. Consider obtaining a nasal polymerase chain reaction swab or saliva test 48 hours prior to the procedure if the patient is undergoing a head and neck procedure or there is risk for transmission.
Guidelines from the ESMO recommended that all patients undergoing surgery or therapy should be swabbed for SARS-CoV-2 before each treatment.11 Patients should wear a mask, remain 6-feet apart in the waiting room, and avoid touching objects until they enter the procedure room. Objects that the patient must touch, such as pens, should be cleaned immediately after such contact with either alcohol or soap and water for 20 seconds.
Office capacity should be reduced by allowing no more than 1 person to accompany the patient and ensuring the presence of only the minimum staff needed for the procedure. Staff who are deemed necessary should wear a mask continuously and gloves during patient contact.
Once in the procedure room, providers might be at elevated risk of contracting COVID-19 or transmitting SARS-CoV-2. A properly fitted N95 respirator and a face shield are recommended, especially for facial cases. N95 respirators can be reused by following the latest Centers for Disease Control and Prevention recommendations for reuse and decontamination techniques,13 which may include protecting the N95 respirator with a surgical mask and storing it in a paper bag when not in use. Consider testing asymptomatic patients in facial cases when they cannot wear a mask.
Steps should be taken to reduce in-person visits. Dissolving sutures can help avoid return visits. Follow-up visits and postprocedural questions should be managed by telehealth. However, patients with a high-risk underlying conditions (eg, posttransplantation, immunosuppressed) should continue to obtain regular skin checks because they are at higher risk for more aggressive malignancies, such as Merkel cell carcinoma.
Conclusion
The future trajectory of the COVID-19 pandemic is uncertain. Dermatologists should continue providing care for patients with skin cancer while mitigating the risk for COVID-19 infection and transmission of SARS-CoV-2. Guidelines provided by the NCCN and ESMO should help providers triage patients. Decisions should be made case by case, keeping in mind the availability of resources and practicing in compliance with local guidance.
The coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome novel coronavirus 2 (SARS-CoV-2), has presented a unique challenge to providing essential care to patients. Increased demand for health care workers and medical supplies, in addition to the risk for COVID-19 infection and asymptomatic transmission of SARS-CoV-2 among health care workers and patients, prompted the delay of nonessential services during the surge of cases this summer.1 Key considerations for continuing operation included current and projected COVID-19 cases in the region, ability to implement telehealth, staffing availability, personal protective equipment availability, and office capacity.2 Providing care that is deemed essential often was determined by the urgency of the treatment or service.
The Centers for Medicare & Medicaid Services outlined a strategy to stratify patients, based on level of acuity, during the COVID-19 surge3:
- Low-acuity treatments or services: includes routine primary, specialty, or preventive care visits. They should be postponed; telehealth follow-ups should be considered.
- Intermediate-acuity treatments or services: includes pediatric and neonatal care, follow-up visits for existing conditions, and evaluation of new symptoms (including those consistent with COVID-19). These services should initially be evaluated using telehealth, then triaged to the appropriate site and level of care.
- High-acuity treatments or services: address symptoms consistent with COVID-19 or other severe disease, of which the lack of in-person evaluation would result in harm to the patient.
Employees in hospitals and health care clinics were classified as essential, but dermatologists were not given explicit direction regarding clinic operation. Many practices have restricted services, especially those in an area of higher COVID-19 prevalence. However, the challenge of determining day-to-day operation may have been left to the provider in most cases.4 As many states in the United States continue to relax restrictions, total cases and the rate of positivity of COVID-19 have been sharply rising again, after months of decline,5 which suggests increased transmission of SARS-CoV-2 and potential resurgence of the high case burden on our health care system. Furthermore, a lack of a widely distributed vaccine or herd immunity suggests we will need to take many of the same precautions as in the first surge.6
In general, patients with cancer have been found to be at greater risk for adverse outcomes and mortality after COVID-19.7 Therefore, resource rationing is particularly concerning for patients with skin cancer, including melanoma, Merkel cell carcinoma, mycosis fungoides, and keratinocyte carcinoma. Triaging patients based on level of acuity, type of skin cancer, disease burden, host immunosuppression, and risk for progression must be carefully considered in this population.2 Treatment and follow-up present additional challenges.
Guidelines provided by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) elaborated on key considerations for the treatment of melanoma, keratinocyte carcinoma, and Merkel cell carcinoma during the COVID-19 pandemic.8-10 Guidelines from the NCCN concentrated on clear divisions between disease stages to determine provider response. Guidelines for melanoma patients proposed by the ESMO assign tiers by value-based priority in various treatment settings, which offered flexibility to providers as the COVID-19 landscape continued to change. Recommendations from the NCCN and ESMO are summarized in Tables 1 to 5.





Although these guidelines initially may have been proposed to delay treatment of lower-acuity tumors, such delay might not be feasible given the unknown duration of this pandemic and future disease waves. One review of several studies, which addressed the outcomes on melanoma survival following the surgical delay recommended by the NCCN, revealed contradictory evidence.12 Further, sufficiently powered studies will be needed to better understand the impact of delaying treatment during the summer COVID-19 surge on patients with skin cancer. Therefore, physicians must triage patients accordingly to manage and treat while also preventing disease spread.
Tips for Performing Dermatologic Surgery
Careful consideration should be made to protect both the patient and staff during office-based excisional surgery during the COVID-19 pandemic. To minimize the risk of transmission of SARS-CoV-2, patients and staff should (1) be screened for symptoms of COVID-19 at least 48 hours prior to entering the office via telephone screening questions, and (2) follow proper hygiene and contact procedures once entering the office. Consider obtaining a nasal polymerase chain reaction swab or saliva test 48 hours prior to the procedure if the patient is undergoing a head and neck procedure or there is risk for transmission.
Guidelines from the ESMO recommended that all patients undergoing surgery or therapy should be swabbed for SARS-CoV-2 before each treatment.11 Patients should wear a mask, remain 6-feet apart in the waiting room, and avoid touching objects until they enter the procedure room. Objects that the patient must touch, such as pens, should be cleaned immediately after such contact with either alcohol or soap and water for 20 seconds.
Office capacity should be reduced by allowing no more than 1 person to accompany the patient and ensuring the presence of only the minimum staff needed for the procedure. Staff who are deemed necessary should wear a mask continuously and gloves during patient contact.
Once in the procedure room, providers might be at elevated risk of contracting COVID-19 or transmitting SARS-CoV-2. A properly fitted N95 respirator and a face shield are recommended, especially for facial cases. N95 respirators can be reused by following the latest Centers for Disease Control and Prevention recommendations for reuse and decontamination techniques,13 which may include protecting the N95 respirator with a surgical mask and storing it in a paper bag when not in use. Consider testing asymptomatic patients in facial cases when they cannot wear a mask.
Steps should be taken to reduce in-person visits. Dissolving sutures can help avoid return visits. Follow-up visits and postprocedural questions should be managed by telehealth. However, patients with a high-risk underlying conditions (eg, posttransplantation, immunosuppressed) should continue to obtain regular skin checks because they are at higher risk for more aggressive malignancies, such as Merkel cell carcinoma.
Conclusion
The future trajectory of the COVID-19 pandemic is uncertain. Dermatologists should continue providing care for patients with skin cancer while mitigating the risk for COVID-19 infection and transmission of SARS-CoV-2. Guidelines provided by the NCCN and ESMO should help providers triage patients. Decisions should be made case by case, keeping in mind the availability of resources and practicing in compliance with local guidance.
- Moletta L, Pierobon ES, Capovilla G, et al. International guidelines and recommendations for surgery during COVID-19 pandemic: a systematic review. Int J Surg. 2020;79:180-188.
- Ueda M, Martins R, Hendrie PC, et al. Managing cancer care during the COVID-19 pandemic: agility and collaboration toward common goal. J Natl Compr Canc Netw. 2020:1-4.
- Center for Medicare & Medicaid Services. Non-emergent, elective medical services, and treatment recommendations. Published April 7, 2020. Accessed October 15, 2020. https://www.cms.gov/files/document/cms-non-emergent-elective-medical-recommendations.pdf
- Muddasani S, Housholder A, Fleischer AB. An assessment of United States dermatology practices during the COVID-19 outbreak. J Dermatolog Treat. 2020;31:436-438.
- Coronavirus Resource Center, Johns Hopkins University & Medicine. Rate of positive tests in the US and states over time. Updated December 11, 2020. Accessed December 11, 2020. https://coronavirus.jhu.edu/testing/individual-states
- Middleton J, Lopes H, Michelson K, et al. Planning for a second wave pandemic of COVID-19 and planning for winter: a statement from the Association of Schools of Public Health in the European Region. Int J Public Health. 2020;65:1525-1527.
- Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335-337.
- National Comprehensive Cancer Network. Advisory statement for non-melanoma skin cancer care during the COVID-19 pandemic (version 4). Published May 22, 2020. Accessed December 11, 2020. https://www.nccn.org/covid-19/pdf/NCCN-NMSC.pdf
National Comprehensive Cancer Network. Short-term recommendations for cutaneous melanoma management during COVID-19 pandemic (version 3). Published May 6, 2020. Accessed December 11, 2020. www.nccn.org/covid-19/pdf/Melanoma.pdf - Conforti C, Giuffrida R, Di Meo N, et al. Management of advanced melanoma in the COVID-19 era. Dermatol Ther. 2020;33:e13444.
- ESMO [European Society for Medical Oncology]. Cancer patient management during the COVID-19 pandemic. Accessed Decemeber 11, 2020. https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic?hit=ehp
- Guhan S, Boland G, Tanabe K, et al. Surgical delay and mortality for primary cutaneous melanoma [published online July 22, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.07.078
- Centers for Disease Control and Prevention. Implementing filtering facepiece respirator (FFR) reuse, including reuse after decontamination, when there are known shortages of N95 respirators. Updated October 19, 2020. Accessed December 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/decontamination-reuse-respirators.html
- Moletta L, Pierobon ES, Capovilla G, et al. International guidelines and recommendations for surgery during COVID-19 pandemic: a systematic review. Int J Surg. 2020;79:180-188.
- Ueda M, Martins R, Hendrie PC, et al. Managing cancer care during the COVID-19 pandemic: agility and collaboration toward common goal. J Natl Compr Canc Netw. 2020:1-4.
- Center for Medicare & Medicaid Services. Non-emergent, elective medical services, and treatment recommendations. Published April 7, 2020. Accessed October 15, 2020. https://www.cms.gov/files/document/cms-non-emergent-elective-medical-recommendations.pdf
- Muddasani S, Housholder A, Fleischer AB. An assessment of United States dermatology practices during the COVID-19 outbreak. J Dermatolog Treat. 2020;31:436-438.
- Coronavirus Resource Center, Johns Hopkins University & Medicine. Rate of positive tests in the US and states over time. Updated December 11, 2020. Accessed December 11, 2020. https://coronavirus.jhu.edu/testing/individual-states
- Middleton J, Lopes H, Michelson K, et al. Planning for a second wave pandemic of COVID-19 and planning for winter: a statement from the Association of Schools of Public Health in the European Region. Int J Public Health. 2020;65:1525-1527.
- Liang W, Guan W, Chen R, et al. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335-337.
- National Comprehensive Cancer Network. Advisory statement for non-melanoma skin cancer care during the COVID-19 pandemic (version 4). Published May 22, 2020. Accessed December 11, 2020. https://www.nccn.org/covid-19/pdf/NCCN-NMSC.pdf
National Comprehensive Cancer Network. Short-term recommendations for cutaneous melanoma management during COVID-19 pandemic (version 3). Published May 6, 2020. Accessed December 11, 2020. www.nccn.org/covid-19/pdf/Melanoma.pdf - Conforti C, Giuffrida R, Di Meo N, et al. Management of advanced melanoma in the COVID-19 era. Dermatol Ther. 2020;33:e13444.
- ESMO [European Society for Medical Oncology]. Cancer patient management during the COVID-19 pandemic. Accessed Decemeber 11, 2020. https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic?hit=ehp
- Guhan S, Boland G, Tanabe K, et al. Surgical delay and mortality for primary cutaneous melanoma [published online July 22, 2020]. J Am Acad Dermatol. doi:10.1016/j.jaad.2020.07.078
- Centers for Disease Control and Prevention. Implementing filtering facepiece respirator (FFR) reuse, including reuse after decontamination, when there are known shortages of N95 respirators. Updated October 19, 2020. Accessed December 11, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/ppe-strategy/decontamination-reuse-respirators.html
Practice Points
- Consider the rate of cases and transmission in your area during a pandemic surge when triaging surgical and nonsurgical cases.
- If performing head and neck surgical procedures or cosmetic procedures in which the patient cannot wear a mask, consider testing them 24 to 48 hours before the procedure.
- Follow Centers for Disease Control and Prevention (CDC) guidelines concerning screening asymptomatic patients. Also, follow CDC guidelines on testing patients who have had prior infections.
- Ensure proper personal protective equipment for yourself and staff, including the use of properly fitting N95 respirators and face shields.
Skin Cancer Screening and Prevention During the COVID-19 Pandemic
On March 11, 2020, the World Health Organization declared the outbreak of coronavirus disease 2019 (COVID-19) a pandemic, leading to an abrupt widespread shift to teledermatology, with postponement of nonessential in-office medical and surgical services, according to American Academy of Dermatology (AAD) recommendations.1 Perspectives have been offered regarding skin cancer management during the pandemic2; however, the current literature is lacking guidance on skin cancer screening and prevention during the COVID-19 era.
Preliminary data show a 34.3% reduction in skin cancer referrals from February to April 2020 compared to the same period in 2019. The authors also presented a subsequent reduction in the number of skin cancer diagnoses in March 2020 compared to March 2019.3 Although the COVID-19 public health emergency should be prioritized by all health care workers, the duty to maintain disease prevention remains.
We aim to provide recommendations for this urgent topic. Our goal is finding balance in preventing an increase in the incidence of and mortality from skin cancer that results from delayed detection, while conserving personalprotective equipment and minimizing exposure, by patients and clinical personnel, to the severe acute respiratory syndrome coronavirus 2. A primary benefit of skin cancer screening lies in the ability to detect melanoma, which is associated with higher mortality than the more common nonmelanoma skin cancers, basal and cutaneous squamous cell carcinomas. We place preeminence on screening directed toward detecting melanoma. The main screening method that dermatologists employ is the total-body skin examination (TBSE). Another widely encouraged and utilized component in skin cancer prevention is patient education, emphasizing avoidance of risk factors, undertaking protective factors, and providing clear instructions for performing the patient-led skin self-examination (SSE).
Teledermatology Essentials for Skin Cancer Screening
Arguably, dermatology possesses the most potential for successfully utilizing telemedicine. Teledermatology has become widely implemented across the United States, secondary to the implications of the current pandemic. A report by Perkins and colleagues4 provided a positive outlook in the preliminary transition to teledermatology beginning in March 2020, though reported time of use was relatively short (3 weeks). A May 2020 article in Dermatology News provided tips for implementing telemedicine for practices.5
We agree with the comprehensive screening algorithm for teledermatology presented by Perkins and colleagues4 (Figure 1A in their report) and recommend the following for the screening and prevention of skin cancer:
• Patients with any characteristics of increased risk, including a personal or family history of melanoma, large congenital nevi, many melanotic nevi, dysplastic nevi, and Fitzpatrick skin types I and II,6 should be prioritized for an in-person visit for TBSE.
• Immunosuppressed patients, particularly organ transplant recipients and those with a history of skin cancer, should be prioritized for an in-person visit for TBSE.
• Established patients evaluated and determined to be at average risk for skin cancer should be offered a teledermatology visit. Suspicious findings during these visits should be prioritized for an in-person visit, with subsequent biopsy and follow-up.
• New patients should be offered a teledermatology visit.
These recommendations must be reviewed alongside each patient’s risk for travel and being present in person as well as other factors that might place the patient at increased risk for COVID-19.
Total-body skin examination, a widely used tool in the dermatologist’s tool kit, presents minimal risk to patients while providing important data for each dermatology patient’s profile, ultimately directing patient care. The role of TBSE in skin cancer screening and prevention has been in discussion even prior to the current pandemic. The US Preventive Services Task Force (USPSTF) has not declared a role for TBSE in recent years; however, USPSTF recommendations are formulated using data from all forms of screening, not only dermatologist-led interventions. Accordingly, USPSTF recommendations target primary care. The AAD has released statements addressing the role of TBSE and skin cancer prevention in the past, when necessary, to provide clarity.7
There is no clear definition of SSE or guidelines on how to educate a patient to perform regular SSE; however, the AAD provides patients with resources on how to perform an SSE.8 Just as dermatologists would provide education, advice, and guidance by directing patients to the AAD website for the SSE during an in-person visit, we encourage dermatologists to continue this practice during all teledermatology visits.
The role of teledermatology in skin cancer screening and prevention is limited; dermatologists will not be able to adequately perform TBSE as it would be done at in-person visits. Furthermore, the true implications of teledermatology compared to in-person visits during the COVID-19 pandemic have yet to be realized and analyzed. It is nonetheless important to appreciate that teledermatology holds great promise of benefit in skin cancer prevention, especially in the form of patient education by dermatologists. Practices in the realm of screening and prevention by health care professionals should be continually addressed during the pandemic; it is important to consider the implications associated with delays in diagnosis and treatment.
Teledermatology Limitations and Recommendations for High-Quality Visits
A benefit of video consultation (VC) vs telephone visits is visual interaction—the crux of dermatology. A 2019 study investigated VC experiences among providers and patients in the primary care setting. Benefits of VC were reported to include convenience for working patients and patients with mobility or mental health problems, visual cues, building rapport, and improving communication.9
Despite these benefits, VC is not without limitations. Many technical factors create variability in the quality of teledermatology VCs for a melanocytic lesion, including patient environment and lighting, color distortion, video resolution, and Internet connection. We make the following recommendations:
• Environment: Locate or create a dedicated space for teledermatology visits that is well lit, private, and has minimal background noise. Place the device on a level surface, center yourself in the frame, and keep the camera at eye level.
• Lighting: Use neutral lighting, placing the light source in front of you but behind the camera of the device. Avoid placing light sources, such as a window, behind you.
• Video resolution: Regardless of the type of camera (eg, integrated webcam, external camera), close out all other running software programs to optimize bandwidth during the visit.
• Internet connection: Use a wired connection (via an Ethernet cable) instead of a Wi-Fi connection to greatly decrease the chance of losing the connection during the visit. It also is faster than Wi-Fi.
• Addressing specific lesions: Patients should keep the device in place, repositioning themselves to show the lesions rather than moving the device by hand.
• Video capacity: Test your device’s video capacity beforehand, which can be as simple as video-calling a family member or friend from your designated space. Feedback regarding video and audio quality will help fine-tune your setup.
• Instructions to the patient: Provide clear instructions to the patient when photographs of specific lesions are needed for further review. Specify what view(s) you need and whether size or bilateral comparison is needed. A web post by VisualDx10 provides advice to patients on taking high-quality photographs.
Final Thoughts
Teledermatology indubitably presents a learning curve for dermatologists and patients. As with other technological advances in society, we are optimistic that, first, the confidence level in teledermatology use will increase, and, second, evidence-based data will pave the way to enhance this experience. We realize the inherent limitation of accessibility to certain technologies, which is regrettably far from equitable. Patients need a personal device equipped with audio and video; access to a high-quality Internet connection; some degree of technological literacy; and a quiet private location.
We hope to learn from all experiences during the current pandemic. Future innovation in teledermatology and in telemedicine generally should aim to address technological inequities to allow for the delivery of quality care to as many patients as possible.
- American Academy of Dermatology. Everyday health and preparedness steps in clinic Updated April 4, 2020. Accessed December 17, 2020. https://assets.ctfassets.net/1ny4yoiyrqia/4LNCNjucOonbQx7aC970x/b56b540957ddad94dcc61949b8e3acc9/COVID-19_Preparedness_30Apr2020.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:295-296.
- Earnshaw CH, Hunter HJA, McMullen E, et al. Reduction in skin cancer diagnosis, and overall cancer referrals, during the COVID-19 pandemic. Br J Dermatol. 2020;183:792-794.
- Perkins S, Cohen JM, Nelson CA, et al. Teledermatology in the era of COVID-19: experience of an academic department of dermatology. J Am Acad Dermatol. 2020;83:E43-E44.
- Marina F. COVID-19: telehealth at the forefront of the pandemic. Dermatology News. May 12, 2020. Accessed December 17, 2020. www.mdedge.com/dermatology/article/222089/coronavirus-updates/covid-19-telehealth-forefront-pandemic?channel=52
- Watts CG, Dieng M, Morton RL, et al. Clinical practice guidelines for identification, screening and follow-up of individuals at high risk of primary cutaneous melanoma: a systematic review. Br J Dermatol. 2015;172:33-47.
- Rosamilia LL. “Doctor, do I need a skin check?” Cutis. 2019;103:290-291.
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology. Accessed December 17, 2020. www.aad.org/public/diseases/skin-cancer/find/check-skin
- Donaghy E, Atherton H, Hammersley V, et al. Acceptability, benefits, and challenges of video consulting: a qualitative study in primary care. Br J Gen Pract. 2019;69:E586-E594.
- How to take the best photos for teledermatology. VisualDx. Accessed December 17, 2020. https://info.visualdx.com/l/11412/2020-03-31/6h4hdz
On March 11, 2020, the World Health Organization declared the outbreak of coronavirus disease 2019 (COVID-19) a pandemic, leading to an abrupt widespread shift to teledermatology, with postponement of nonessential in-office medical and surgical services, according to American Academy of Dermatology (AAD) recommendations.1 Perspectives have been offered regarding skin cancer management during the pandemic2; however, the current literature is lacking guidance on skin cancer screening and prevention during the COVID-19 era.
Preliminary data show a 34.3% reduction in skin cancer referrals from February to April 2020 compared to the same period in 2019. The authors also presented a subsequent reduction in the number of skin cancer diagnoses in March 2020 compared to March 2019.3 Although the COVID-19 public health emergency should be prioritized by all health care workers, the duty to maintain disease prevention remains.
We aim to provide recommendations for this urgent topic. Our goal is finding balance in preventing an increase in the incidence of and mortality from skin cancer that results from delayed detection, while conserving personalprotective equipment and minimizing exposure, by patients and clinical personnel, to the severe acute respiratory syndrome coronavirus 2. A primary benefit of skin cancer screening lies in the ability to detect melanoma, which is associated with higher mortality than the more common nonmelanoma skin cancers, basal and cutaneous squamous cell carcinomas. We place preeminence on screening directed toward detecting melanoma. The main screening method that dermatologists employ is the total-body skin examination (TBSE). Another widely encouraged and utilized component in skin cancer prevention is patient education, emphasizing avoidance of risk factors, undertaking protective factors, and providing clear instructions for performing the patient-led skin self-examination (SSE).
Teledermatology Essentials for Skin Cancer Screening
Arguably, dermatology possesses the most potential for successfully utilizing telemedicine. Teledermatology has become widely implemented across the United States, secondary to the implications of the current pandemic. A report by Perkins and colleagues4 provided a positive outlook in the preliminary transition to teledermatology beginning in March 2020, though reported time of use was relatively short (3 weeks). A May 2020 article in Dermatology News provided tips for implementing telemedicine for practices.5
We agree with the comprehensive screening algorithm for teledermatology presented by Perkins and colleagues4 (Figure 1A in their report) and recommend the following for the screening and prevention of skin cancer:
• Patients with any characteristics of increased risk, including a personal or family history of melanoma, large congenital nevi, many melanotic nevi, dysplastic nevi, and Fitzpatrick skin types I and II,6 should be prioritized for an in-person visit for TBSE.
• Immunosuppressed patients, particularly organ transplant recipients and those with a history of skin cancer, should be prioritized for an in-person visit for TBSE.
• Established patients evaluated and determined to be at average risk for skin cancer should be offered a teledermatology visit. Suspicious findings during these visits should be prioritized for an in-person visit, with subsequent biopsy and follow-up.
• New patients should be offered a teledermatology visit.
These recommendations must be reviewed alongside each patient’s risk for travel and being present in person as well as other factors that might place the patient at increased risk for COVID-19.
Total-body skin examination, a widely used tool in the dermatologist’s tool kit, presents minimal risk to patients while providing important data for each dermatology patient’s profile, ultimately directing patient care. The role of TBSE in skin cancer screening and prevention has been in discussion even prior to the current pandemic. The US Preventive Services Task Force (USPSTF) has not declared a role for TBSE in recent years; however, USPSTF recommendations are formulated using data from all forms of screening, not only dermatologist-led interventions. Accordingly, USPSTF recommendations target primary care. The AAD has released statements addressing the role of TBSE and skin cancer prevention in the past, when necessary, to provide clarity.7
There is no clear definition of SSE or guidelines on how to educate a patient to perform regular SSE; however, the AAD provides patients with resources on how to perform an SSE.8 Just as dermatologists would provide education, advice, and guidance by directing patients to the AAD website for the SSE during an in-person visit, we encourage dermatologists to continue this practice during all teledermatology visits.
The role of teledermatology in skin cancer screening and prevention is limited; dermatologists will not be able to adequately perform TBSE as it would be done at in-person visits. Furthermore, the true implications of teledermatology compared to in-person visits during the COVID-19 pandemic have yet to be realized and analyzed. It is nonetheless important to appreciate that teledermatology holds great promise of benefit in skin cancer prevention, especially in the form of patient education by dermatologists. Practices in the realm of screening and prevention by health care professionals should be continually addressed during the pandemic; it is important to consider the implications associated with delays in diagnosis and treatment.
Teledermatology Limitations and Recommendations for High-Quality Visits
A benefit of video consultation (VC) vs telephone visits is visual interaction—the crux of dermatology. A 2019 study investigated VC experiences among providers and patients in the primary care setting. Benefits of VC were reported to include convenience for working patients and patients with mobility or mental health problems, visual cues, building rapport, and improving communication.9
Despite these benefits, VC is not without limitations. Many technical factors create variability in the quality of teledermatology VCs for a melanocytic lesion, including patient environment and lighting, color distortion, video resolution, and Internet connection. We make the following recommendations:
• Environment: Locate or create a dedicated space for teledermatology visits that is well lit, private, and has minimal background noise. Place the device on a level surface, center yourself in the frame, and keep the camera at eye level.
• Lighting: Use neutral lighting, placing the light source in front of you but behind the camera of the device. Avoid placing light sources, such as a window, behind you.
• Video resolution: Regardless of the type of camera (eg, integrated webcam, external camera), close out all other running software programs to optimize bandwidth during the visit.
• Internet connection: Use a wired connection (via an Ethernet cable) instead of a Wi-Fi connection to greatly decrease the chance of losing the connection during the visit. It also is faster than Wi-Fi.
• Addressing specific lesions: Patients should keep the device in place, repositioning themselves to show the lesions rather than moving the device by hand.
• Video capacity: Test your device’s video capacity beforehand, which can be as simple as video-calling a family member or friend from your designated space. Feedback regarding video and audio quality will help fine-tune your setup.
• Instructions to the patient: Provide clear instructions to the patient when photographs of specific lesions are needed for further review. Specify what view(s) you need and whether size or bilateral comparison is needed. A web post by VisualDx10 provides advice to patients on taking high-quality photographs.
Final Thoughts
Teledermatology indubitably presents a learning curve for dermatologists and patients. As with other technological advances in society, we are optimistic that, first, the confidence level in teledermatology use will increase, and, second, evidence-based data will pave the way to enhance this experience. We realize the inherent limitation of accessibility to certain technologies, which is regrettably far from equitable. Patients need a personal device equipped with audio and video; access to a high-quality Internet connection; some degree of technological literacy; and a quiet private location.
We hope to learn from all experiences during the current pandemic. Future innovation in teledermatology and in telemedicine generally should aim to address technological inequities to allow for the delivery of quality care to as many patients as possible.
On March 11, 2020, the World Health Organization declared the outbreak of coronavirus disease 2019 (COVID-19) a pandemic, leading to an abrupt widespread shift to teledermatology, with postponement of nonessential in-office medical and surgical services, according to American Academy of Dermatology (AAD) recommendations.1 Perspectives have been offered regarding skin cancer management during the pandemic2; however, the current literature is lacking guidance on skin cancer screening and prevention during the COVID-19 era.
Preliminary data show a 34.3% reduction in skin cancer referrals from February to April 2020 compared to the same period in 2019. The authors also presented a subsequent reduction in the number of skin cancer diagnoses in March 2020 compared to March 2019.3 Although the COVID-19 public health emergency should be prioritized by all health care workers, the duty to maintain disease prevention remains.
We aim to provide recommendations for this urgent topic. Our goal is finding balance in preventing an increase in the incidence of and mortality from skin cancer that results from delayed detection, while conserving personalprotective equipment and minimizing exposure, by patients and clinical personnel, to the severe acute respiratory syndrome coronavirus 2. A primary benefit of skin cancer screening lies in the ability to detect melanoma, which is associated with higher mortality than the more common nonmelanoma skin cancers, basal and cutaneous squamous cell carcinomas. We place preeminence on screening directed toward detecting melanoma. The main screening method that dermatologists employ is the total-body skin examination (TBSE). Another widely encouraged and utilized component in skin cancer prevention is patient education, emphasizing avoidance of risk factors, undertaking protective factors, and providing clear instructions for performing the patient-led skin self-examination (SSE).
Teledermatology Essentials for Skin Cancer Screening
Arguably, dermatology possesses the most potential for successfully utilizing telemedicine. Teledermatology has become widely implemented across the United States, secondary to the implications of the current pandemic. A report by Perkins and colleagues4 provided a positive outlook in the preliminary transition to teledermatology beginning in March 2020, though reported time of use was relatively short (3 weeks). A May 2020 article in Dermatology News provided tips for implementing telemedicine for practices.5
We agree with the comprehensive screening algorithm for teledermatology presented by Perkins and colleagues4 (Figure 1A in their report) and recommend the following for the screening and prevention of skin cancer:
• Patients with any characteristics of increased risk, including a personal or family history of melanoma, large congenital nevi, many melanotic nevi, dysplastic nevi, and Fitzpatrick skin types I and II,6 should be prioritized for an in-person visit for TBSE.
• Immunosuppressed patients, particularly organ transplant recipients and those with a history of skin cancer, should be prioritized for an in-person visit for TBSE.
• Established patients evaluated and determined to be at average risk for skin cancer should be offered a teledermatology visit. Suspicious findings during these visits should be prioritized for an in-person visit, with subsequent biopsy and follow-up.
• New patients should be offered a teledermatology visit.
These recommendations must be reviewed alongside each patient’s risk for travel and being present in person as well as other factors that might place the patient at increased risk for COVID-19.
Total-body skin examination, a widely used tool in the dermatologist’s tool kit, presents minimal risk to patients while providing important data for each dermatology patient’s profile, ultimately directing patient care. The role of TBSE in skin cancer screening and prevention has been in discussion even prior to the current pandemic. The US Preventive Services Task Force (USPSTF) has not declared a role for TBSE in recent years; however, USPSTF recommendations are formulated using data from all forms of screening, not only dermatologist-led interventions. Accordingly, USPSTF recommendations target primary care. The AAD has released statements addressing the role of TBSE and skin cancer prevention in the past, when necessary, to provide clarity.7
There is no clear definition of SSE or guidelines on how to educate a patient to perform regular SSE; however, the AAD provides patients with resources on how to perform an SSE.8 Just as dermatologists would provide education, advice, and guidance by directing patients to the AAD website for the SSE during an in-person visit, we encourage dermatologists to continue this practice during all teledermatology visits.
The role of teledermatology in skin cancer screening and prevention is limited; dermatologists will not be able to adequately perform TBSE as it would be done at in-person visits. Furthermore, the true implications of teledermatology compared to in-person visits during the COVID-19 pandemic have yet to be realized and analyzed. It is nonetheless important to appreciate that teledermatology holds great promise of benefit in skin cancer prevention, especially in the form of patient education by dermatologists. Practices in the realm of screening and prevention by health care professionals should be continually addressed during the pandemic; it is important to consider the implications associated with delays in diagnosis and treatment.
Teledermatology Limitations and Recommendations for High-Quality Visits
A benefit of video consultation (VC) vs telephone visits is visual interaction—the crux of dermatology. A 2019 study investigated VC experiences among providers and patients in the primary care setting. Benefits of VC were reported to include convenience for working patients and patients with mobility or mental health problems, visual cues, building rapport, and improving communication.9
Despite these benefits, VC is not without limitations. Many technical factors create variability in the quality of teledermatology VCs for a melanocytic lesion, including patient environment and lighting, color distortion, video resolution, and Internet connection. We make the following recommendations:
• Environment: Locate or create a dedicated space for teledermatology visits that is well lit, private, and has minimal background noise. Place the device on a level surface, center yourself in the frame, and keep the camera at eye level.
• Lighting: Use neutral lighting, placing the light source in front of you but behind the camera of the device. Avoid placing light sources, such as a window, behind you.
• Video resolution: Regardless of the type of camera (eg, integrated webcam, external camera), close out all other running software programs to optimize bandwidth during the visit.
• Internet connection: Use a wired connection (via an Ethernet cable) instead of a Wi-Fi connection to greatly decrease the chance of losing the connection during the visit. It also is faster than Wi-Fi.
• Addressing specific lesions: Patients should keep the device in place, repositioning themselves to show the lesions rather than moving the device by hand.
• Video capacity: Test your device’s video capacity beforehand, which can be as simple as video-calling a family member or friend from your designated space. Feedback regarding video and audio quality will help fine-tune your setup.
• Instructions to the patient: Provide clear instructions to the patient when photographs of specific lesions are needed for further review. Specify what view(s) you need and whether size or bilateral comparison is needed. A web post by VisualDx10 provides advice to patients on taking high-quality photographs.
Final Thoughts
Teledermatology indubitably presents a learning curve for dermatologists and patients. As with other technological advances in society, we are optimistic that, first, the confidence level in teledermatology use will increase, and, second, evidence-based data will pave the way to enhance this experience. We realize the inherent limitation of accessibility to certain technologies, which is regrettably far from equitable. Patients need a personal device equipped with audio and video; access to a high-quality Internet connection; some degree of technological literacy; and a quiet private location.
We hope to learn from all experiences during the current pandemic. Future innovation in teledermatology and in telemedicine generally should aim to address technological inequities to allow for the delivery of quality care to as many patients as possible.
- American Academy of Dermatology. Everyday health and preparedness steps in clinic Updated April 4, 2020. Accessed December 17, 2020. https://assets.ctfassets.net/1ny4yoiyrqia/4LNCNjucOonbQx7aC970x/b56b540957ddad94dcc61949b8e3acc9/COVID-19_Preparedness_30Apr2020.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:295-296.
- Earnshaw CH, Hunter HJA, McMullen E, et al. Reduction in skin cancer diagnosis, and overall cancer referrals, during the COVID-19 pandemic. Br J Dermatol. 2020;183:792-794.
- Perkins S, Cohen JM, Nelson CA, et al. Teledermatology in the era of COVID-19: experience of an academic department of dermatology. J Am Acad Dermatol. 2020;83:E43-E44.
- Marina F. COVID-19: telehealth at the forefront of the pandemic. Dermatology News. May 12, 2020. Accessed December 17, 2020. www.mdedge.com/dermatology/article/222089/coronavirus-updates/covid-19-telehealth-forefront-pandemic?channel=52
- Watts CG, Dieng M, Morton RL, et al. Clinical practice guidelines for identification, screening and follow-up of individuals at high risk of primary cutaneous melanoma: a systematic review. Br J Dermatol. 2015;172:33-47.
- Rosamilia LL. “Doctor, do I need a skin check?” Cutis. 2019;103:290-291.
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology. Accessed December 17, 2020. www.aad.org/public/diseases/skin-cancer/find/check-skin
- Donaghy E, Atherton H, Hammersley V, et al. Acceptability, benefits, and challenges of video consulting: a qualitative study in primary care. Br J Gen Pract. 2019;69:E586-E594.
- How to take the best photos for teledermatology. VisualDx. Accessed December 17, 2020. https://info.visualdx.com/l/11412/2020-03-31/6h4hdz
- American Academy of Dermatology. Everyday health and preparedness steps in clinic Updated April 4, 2020. Accessed December 17, 2020. https://assets.ctfassets.net/1ny4yoiyrqia/4LNCNjucOonbQx7aC970x/b56b540957ddad94dcc61949b8e3acc9/COVID-19_Preparedness_30Apr2020.pdf
- Geskin LJ, Trager MH, Aasi SZ, et al. Perspectives on the recommendations for skin cancer management during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:295-296.
- Earnshaw CH, Hunter HJA, McMullen E, et al. Reduction in skin cancer diagnosis, and overall cancer referrals, during the COVID-19 pandemic. Br J Dermatol. 2020;183:792-794.
- Perkins S, Cohen JM, Nelson CA, et al. Teledermatology in the era of COVID-19: experience of an academic department of dermatology. J Am Acad Dermatol. 2020;83:E43-E44.
- Marina F. COVID-19: telehealth at the forefront of the pandemic. Dermatology News. May 12, 2020. Accessed December 17, 2020. www.mdedge.com/dermatology/article/222089/coronavirus-updates/covid-19-telehealth-forefront-pandemic?channel=52
- Watts CG, Dieng M, Morton RL, et al. Clinical practice guidelines for identification, screening and follow-up of individuals at high risk of primary cutaneous melanoma: a systematic review. Br J Dermatol. 2015;172:33-47.
- Rosamilia LL. “Doctor, do I need a skin check?” Cutis. 2019;103:290-291.
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology. Accessed December 17, 2020. www.aad.org/public/diseases/skin-cancer/find/check-skin
- Donaghy E, Atherton H, Hammersley V, et al. Acceptability, benefits, and challenges of video consulting: a qualitative study in primary care. Br J Gen Pract. 2019;69:E586-E594.
- How to take the best photos for teledermatology. VisualDx. Accessed December 17, 2020. https://info.visualdx.com/l/11412/2020-03-31/6h4hdz
Practice Points
- It is important for dermatologists to maintain skin cancer screening and prevention efforts during the coronavirus disease 2019 pandemic.
- Patient populations at increased risk for skin cancer should be prioritized for in-person evaluations, but teledermatology should be considered for initial examination in new patients and patients at average risk for skin cancer.
- Teledermatology presents a learning curve for dermatologists and patients, but the confidence level will increase, and evidence-based data will pave the way to enhance this experience.
Skin Cancer in the US Military
There are numerous intrinsic risks that military servicemembers face, such as the dangers of combat, handling firearms, operating ships and heavy machinery, undersea diving, and aircraft operations. Multiple studies also have identified an increased risk for melanomas and keratinocyte cancers in those who have served on active duty.
Epidemiology
Differences in demographics are important to consider given the differences among races in the risks of skin cancers. Important racial demographic differences exist between the US Military and the general US population. Racial demographic differences also exist among the various military branches themselves. The US population is 61.0% White, 20.7% racial minorities (defined as Black or African American, Asian, American Indian or Alaska native, Native Hawaiian or other Pacific Islander, multiracial, or unknown), and 18.3% Hispanic or Latino (Hispanic or Latino was not listed as a component of racial minorities).1 According to 2018 data, the US Military population is 52.9% White, 31.0% racial minorities, and 16.1% Hispanic or Latino.2 The percentage of White military members was highest in the US Marine Corps (58.4%) and lowest in the US Navy (46.5%). The percentage of racial minorities was highest in the US Navy (38.0%) and lowest in the US Marine Corps (20.0%).2 The percentage of Hispanic and Latino military members was highest in the US Marine Corps (21.6%) and lowest in the US Air Force (14.5%).2
Melanoma in Military Members
It is estimated that the annual incidence rate of melanoma in the United States is 27 per 100,000 individuals for non-Hispanic Whites, 5 per 100,000 for Hispanics, and 1 per 100,000 for Black individuals and Asians/Pacific Islanders.3 Three studies have reviewed melanoma incidence in relation to service in the US Military.
A 2011 retrospective tumor registries study of US veterans aged 45 years or older demonstrated increased incidences of melanoma compared with the general population.4 With age, the melanoma incidence per 100,000 person-years increased in White veterans compared to their civilian counterparts (aged 45 to 49 years, 33.62 vs 27.49; aged 50 to 54 years, 49.76 vs 32.18; aged 55 to 59 years, 178.48 vs 39.17).4 An increased melanoma incidence of 62% also was seen in active-duty servicemembers aged 18 to 56 years compared to their age-matched civilian peers in a 2014 retrospective cohort study.5
Melanoma rates also vary depending on military service branch. Across 3 separate studies, service in the US Air Force was associated with the highest risk for melanoma development. A surveillance report of cancer incidence in active-duty US Armed Forces personnel between 2000 and 2011 conducted by the Defense Medical Surveillance System showed an incidence rate (per 100,000 person-years) for melanoma of 10.5 in all services, and a rate of 15.5 in the US Air Force vs 8.6 in the US Army, further highlighting the disparity between the services.6 The 2014 study also demonstrated a melanoma incidence rate of 17.80 in active-duty
Keratinocyte Cancers in Military Members
Although less well studied than melanoma, keratinocyte-derived skin cancers represent a major source of disease burden both during and after active-duty service. In a retrospective chart review of dermatology patients seen at the 86th Combat Support Hospital at Ibn Sina Hospital in Baghdad, Iraq, during a 6-month period in 2008, 8% of 2696 total visits were identified to be due to skin cancer, with the overwhelming majority being for keratinocyte cancers.7 A 1993 retrospective chart review of World War II veterans referred for Mohs micrographic surgery showed a considerably higher incidence in those who served in the Pacific Theater compared to those who served in the European Theater. Despite having approximately equal characteristics—age, skin type, and cumulative time spent outdoors—between the 2 groups, military servicemembers deployed to the Pacific represented 66% of the patients with basal cell carcinoma and 68% of the patients with squamous cell carcinoma.8
Contributing Factors
There are many factors related to military service that are likely to contribute to the increased risk for skin cancer. Based on a review of the literature, we have found an increased exposure to UV radiation, low utilization of sun-protective strategies, and low overall education regarding the risks for UV exposure to be the primary contributors to increased risks for skin cancer.
UV exposure is the primary mitigatable risk factor for developing melanoma and keratinocyte cancers.9,10 In a 2015 study of 212 military servicemembers returning from deployments in Iraq and Afghanistan, 77% reported spending more than 4 hours per day working directly in the bright sun, with 64% spending more than 75% of the average day in the bright sun.11 A 1984 study of World War II veterans diagnosed with melanoma also showed that 34% of those with melanoma had prior deployments to the tropics compared to 6% in age-matched controls.12
Even in those not deployed to overseas locations, military work still frequently involves prolonged sun exposure. In a 2015 cross-sectional study of US Air Force maintenance squadrons at Travis Air Force Base in Fairfield, California (N=356), 67% of those surveyed reported having careers that frequently involved direct sun exposure.13 This occupational sun exposure may be worsened by increased UV exposure during recreational activities, as active-duty military servicemembers may reasonably be expected to engage in more outdoor exercise and leisure activities than their civilian counterparts.
Other occupation-specific risk factors also may affect skin cancer rates in certain populations. In a study of aircraft personnel that included male military and civilian pilots, a meta-standardized incidence ratio for melanoma of 3.42 was identified compared to controls not involved in aircraft work.14 Theories to explain this increased incidence of melanoma include increased exposure to ionizing radiation at high altitudes, exposure to aviation-related chemicals, and alterations in circadian rhythm.14,15
This increased sun exposure is compounded by the overall low rates of sun protection among military members. Of those returning from Iraq and Afghanistan in the 2015 study, less than 30% of servicemembers reported routine access to sunscreen, and only 13% stated that they routinely applied sunscreen when exposed to the sun. Of this same group, only 23% endorsed that the military made them very aware of their risk for skin cancer.11 The low rates of sunscreen usage by those deployed to an active combat zone may partially be explained by the assumption that those individuals placed more emphasis on the acute dangers of combat rather than the perceived future dangers of skin cancer. A decreased availability of sunscreen for deployed military servicemembers, particularly those located at small austere bases where supplies are likely to be limited, likely makes the use of sunscreen even more difficult.
However, even within the continental United States, active-duty military servicemembers still exhibit low rates of sunscreen usage. In the 2015 study of US Air Force personnel in maintenance squadrons in California, less than 11% of those surveyed reported using sunscreen most of the time despite high rates of outdoor work.13
Another factor likely contributing to increased sun exposure and decreased sun-protection practices is the so-called invincibility complex, which is a common set of egocentric beliefs that leads to a perception that an individual is not likely to suffer the consequences of engaging in risky behaviors. Despite knowledge of the dangers associated with risky activity, individuals with an invincibility complex are more likely to view potential consequences as relevant only to others, not to themselves.16 A study of adolescent smokers in the Netherlands examined why subjects continue to smoke, despite knowledge of the potentially deadly consequences of smoking. Three common rationalizing beliefs were found: trivialization of the immediate consequences, that their smoking is only temporary and they have time in the future to stop, and that they have control over how much they smoke and can prevent fatal consequences with moderation.17 Such an invincibility complex is thought to directly run counter to the efforts of public health and educational campaigns. This belief set is thought to at least partially explain why adolescents in Australia are the most knowledgeable age cohort regarding the dangers of UV exposure but the least likely to engage in skin-protective measures.18 This inflated sense of invincibility may be leading active-duty military servicemembers to engage in unhealthy sun-exposure practices regardless of knowledge of the associated risks.
Members of the military may be uniquely susceptible to this invincibility complex. Growing evidence suggests that exposure to life-threatening circumstances may lead to long-lasting alterations in threat assessment.19,20 A 2008 study of Iraq veterans returning from deployment found that direct exposure to violent combat and human trauma was associated with an increased perceived degree of invincibility and a higher propensity to engage in risky behaviors after returning from deployment.19 Additionally, it has been speculated that individuals with a higher degree of perceived invincibility may be more likely to pursue military service, as a higher degree of self-confidence in the face of the often dangerous circumstances of military operations may be advantageous.20
In addition to scarce use of sun-protective strategies, military servicemembers also tend to lack awareness of the potential short-term and long-term harm from UV radiation. In a 2016 study of veterans undergoing treatment for skin cancer, patients reported inadequate education about skin cancer risks and strategies to decrease their chances of developing it.21 Sunscreen is less frequently used in males, specifically those aged 18 to 30 years; this demographic makes up 55.7% of the active-duty population.2,22 Low income also has been associated with decreased sunscreen use; junior enlisted military servicemembers (ranks E1-E4) make up 43.8% of the military’s ranks and make less than the average annual American household income.2,23,24
Prevention and Risk-Mitigation Strategies
Although many of the risk factors in the US Military promoting skin cancer are intrinsic to the occupation, certain steps could help minimize servicemembers’ risks. To be effective, any attempt to decrease the risk for skin cancer in the US Military must take into consideration the environment in which the military operates. To complete their mission, military personnel often are required to operate for extended periods outdoors in areas of high UV exposure, such as the deserts of Iraq or the mountains of Afghanistan. Outdoor work at times of peak sunlight often is required for successful mission completion, thus it would be ineffective to simply give blanket advice to avoid sun exposure.
Another important factor is the impact that official policy plays in shaping the daily actions of individual military servicemembers. In a hierarchical organization such as the US Military, unit commanders have substantial authority over the behaviors of their subordinates. Thus, strategies to mitigate skin cancer risks should be aimed at the individual servicemembers and unit commanders and at a policy level. Ultimately, a 3-pronged approach built on education, access to sun-protective gear, and increased availability to sunscreen is recommended.
Education
The foundation for any skin cancer prevention strategies should be built on the education of individual military servicemembers. The majority of active-duty members and veterans did not believe the military did enough to actively educate them on the risks for developing skin cancer.21 An effective educational program should focus on prevention and detection. Prevention programs should explain the role of UV exposure in the development of skin cancer, the intrinsic risks of UV exposure associated with outdoor activities, and strategies that can be implemented to reduce UV exposure and lifetime risk of skin cancer development. In a study of German outdoor workers, displays of support and concern by management regarding UV protection were associated with increases in sun-protective behaviors among the employees.25
Because patient self-examinations have been shown to be associated with earlier melanoma diagnosis and a more superficial depth at diagnosis, detection programs also should focus on the identification of suspicious skin lesions, such as by teaching the ABCDEs of melanoma.26 Among the general population, educational campaigns have been shown to be effective at reducing melanoma mortality.27,28
Access to Sun-Protective Gear
The second aspect of reducing skin cancer risk should be aiming to protect military servicemembers from UV exposure. Any prevention strategy must fit within the military’s broader tactical and strategic framework.
The use of photoprotective strategies rather than the outright avoidance of sun exposure should be implemented to minimize the deleterious effects of outdoor work. The most recent study of the UV-protective properties of US Military uniforms found all tested uniforms to have either very good or excellent UV protection, with UV protection factors (UPFs) ranging from 35 to 50+.29 However, this study was performed in 2002, and the majority of the uniforms tested are no longer in service. More up-to-date UPF information for existing military uniforms is not currently available. Most military commands wear baseball hat–style covers when operating outdoors, which generally provide good photoprotection with UPF ratings of 35 to 50 over the protected areas.29 Unfortunately, these types of headgear offer less photoprotection than do wide-brimmed hats, which have demonstrated improved photoprotection, particularly of the neck, cheeks, ears, and chin.30 A wide-brimmed hat, known as the boonie hat, was originally proposed for military use in 1966 to provide protection of servicemembers’ faces and necks from the intense sun of Vietnam. Currently, the use of the boonie hat typically is prohibited for units not engaged in combat or combat-support roles and requires authorization by the unit-level commander.31 Because of its perception as “unmilitary appearing” by many unit commanders and its restriction of use to combat-related units, the boonie hat is not consistently used. Increasing the use of this type of wide-brimmed hat would be an important asset in decreasing chronic UV exposure in military servicemembers, particularly on those parts of the body where skin cancer occurrence is the greatest.32 Policies should be aimed at increasing the use of the boonie hat, both through expanding its availability to troops in non–combat-related fields and by encouraging unit commanders to authorize its use in their units.
Sunscreen Availability
Improving the use of sunscreen is another impactful strategy that could be undertaken to decrease the risk for skin cancer in military servicemembers. The use of sunscreen is low in both those deployed overseas and those stationed within the United States. Improving access to sunscreen, particularly in the deployed setting, also could reduce barriers to use. Providing sunscreen directly to servicemembers, either when issuing gear or integrated within Meals Ready to Eat, could remove both the financial and logistical barriers to sunscreen utilization. Centralized troop-gathering locations, such as dining facilities, could be utilized both for the mass distribution of sunscreen and to display educational material. Unit commanders also could mandate times for servicemembers to stop work and apply sunscreen at regularly scheduled intervals.
The composition and delivery vehicle of sunscreen may have an impact on its efficacy and ease of use in the field. The American Academy of Dermatology (AAD) recommends using sunscreen that is broad spectrum, sun protection factor (SPF) 30 or greater, and water resistant.33 However, the AAD does not make a recommendation of whether to use a physical sunscreen (such as titanium dioxide) or a chemical sunscreen. If applied in equal amounts, a chemical sunscreen and a physical sunscreen with an equal SPF should offer the same UV protection. However, a study in the British Journal of Dermatology showed that subjects applied only two-thirds the quantity of physical sunscreen compared to those applying chemical sunscreen, achieving approximately only one-half the SPF as provided by the chemical sunscreen.34 Because sunscreen is only effective when it is used, consideration should be given to the preferences of the military population when selecting sunscreens. A review of consumer preferences of sunscreen qualities showed that sunscreens that were nongreasy and did not leave a residue were given the most favorable rankings.35 In recent years, sunscreen sprays have become increasingly popular. When adequately applied, sprays have been shown to be equally effective as sunscreen lotions.36 However, although recommendations have been issued by both the AAD and the US Food and Drug Administration on the application of sunscreen lotion to adequately cover exposed skin, no such recommendations have been given for sunscreen sprays.33 Some safety concerns also remain regarding the flammability of aerosol sunscreens, which could be exacerbated in a combat situation.37
However, there are some obvious downsides to sunscreen use. During certain operational tasks, particularly in combat settings, it may not be feasible or even safe to stop working to apply sunscreen at the 2-hour intervals required for effective UV protection.38 Water exposure or large amounts of perspiration also would cause sunscreen to lose effectiveness earlier than expected. Logistically, it may be challenging to regularly supply sunscreen to small austere bases in remote locations.
Final Thoughts
The men and women of our armed forces already undertake great risk in the defense of our country. It should be ensured that their risk for developing skin cancer is made as low as possible, while still allowing them to successfully accomplish their mission. Multiple studies have shown servicemembers to be at an increased risk for skin cancer, particularly melanoma. We believe the primary factor behind this increased risk is occupational UV exposure, which is compounded by the suboptimal use of sun-protective strategies. By educating our servicemembers about their risk for skin cancer and promoting increased UV protection, we can effectively reduce the burden of skin cancer on our active-duty servicemembers and veterans.
- QuickFacts. United States Census Bureau. Accessed December 15, 2020. https://www.census.gov/quickfacts/fact/table/US/PST045219
- 2018 Demographics Profile. Military OneSource. Accessed December 15, 2020. https://www.militaryonesource.mil/reports-and-surveys/infographics/active-duty-member-and-family-demographics
- Cancer Facts & Figures 2019. American Cancer Society. Accessed December 15, 2020. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2019.html
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Military Med. 2014;179:247-253.
- Armed Forces Health Surveillance Center. Incident diagnoses of cancers and cancer-related deaths, active component, US Armed Forces, 2000-2011. MSMR. 2012;19:18-22.
- Henning JS, Firoz BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Ramani ML, Bennett RG. High prevalence of skin-cancer in World-War-II servicemen stationed in the Pacific Theater. J Am Acad Dermatol. 1993;28:733-737.
- Schmitt J, Seidler A, Diepgen TL, et al. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: a systematic review and meta-analysis. Br J Dermatol. 2011;164:291-307.
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. J Photochem Photobiol B. 2001;63:8-18.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Brown J, Kopf AW, Rica DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Parker G, Williams B, Driggers P. Sun exposure knowledge and practices survey of maintenance squadrons at Travis AFB. Military Med. 2015;180:26-31.
- Buja A, Lange JH, Perissinotto E, et al. Cancer incidence among male military and civil pilots and flight attendants: an analysis on published data. Toxicol Ind Health. 2005;21:273-282.
- Wilkison BD, Wong EB. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Wickman ME, Anderson NLR, Smith Greenberg C. The adolescent perception of invincibility and its influence on teen acceptance of health promotion strategies. J Pediatr Nurs. 2008;23:460-468.
- Schreuders M, Krooneman NT, van den Putte B, et al. Boy smokers’ rationalisations for engaging in potentially fatal behaviour: in-depth interviews in the Netherlands. Int J Environ Res Public Health. 2018;15:767.
- Eastabrook S, Chang P, Taylor MF. Melanoma risk: adolescent females’ perspectives on skin protection pre/post-viewing a ultraviolet photoaged photograph of their own facial sun damage. Glob Health Promot. 2018;25:23-32.
- Killgore WD, Cotting DI, Thomas JL, et al. Post-combat invincibility: violent combat experiences are associated with increased risk-taking propensity following deployment. J Psychiatr Res. 2008;42:1112-1121.
- Killgore WD, Kelley A, Balkin TJ. So you think you’re bulletproof: development and validation of the Invincibility Belief Index (IBI). Military Med. 2010;175:499-508.
- McGrath JM, Fisher V, Krejci-Manwaring J. Skin cancer warnings and the need for new preventive campaigns: a pilot study. Am J Prevent Med. 2016;50:E62-E63.
- Thieden E, Philipsen PA, Sandby-Moller J, et al. Sunscreen use related to UV exposure, age, sex, and occupation based on personal dosimeter readings and sun-exposure behavior diaries. Arch Dermatol. 2005;141:967-973.
- Holman DM, Berkowitz Z, Guy GP Jr, et al. Patterns of sunscreen use on the face and other exposed skin among US adults. J Am Acad Dermatol. 2015;73:83-92.e1.
- Military Pay Tables & Information. Defense Finance and Accounting Service website. Accessed December 21, 2020. https://www.dfas.mil/militarymembers/payentitlements/Pay-Tables.html
- Schilling L, Schneider S, Gorig T, et al. “Lost in the sun”—the key role of perceived workplace support for sun-protective behavior in outdoor workers. Am J Ind Med. 2018;61:929-938.
- Uliasz A, Lebwohl M. Patient education and regular surveillance results in earlier diagnosis of second primary melanoma. Int J Dermatol. 2007;46:575-577.
- MacKie RM, Hole D. Audit of public education campaign to encourage earlier detection of malignant melanoma. BMJ. 1992;304:1012-1015.
- Berwick M, Begg CB, Fine JA, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
- Winterhalter C, DiLuna K, Bide M. Characterization of the ultraviolet protection of combat uniform fabrics. US Army Soldier and Biological Chemical Command Soldier Systems Center technical report Natick/TR-02/006. Published January 21, 2002. Accessed December 21, 2021. https://apps.dtic.mil/dtic/tr/fulltext/u2/a398572.pdf
- Gies P, Javorniczky J, Roy C, et al. Measurements of the UVR protection provided by hats used at school. Photochem Photobiol. 2006;82:750-754.
- Stanton S. Headgear. In: Stanton S. US Army Uniforms of the Vietnam War. Stackpole Books; 1992:26-61.
- Richmond-Sinclair NM, Pandeya N, Ware RS, et al. Incidence of basal cell carcinoma multiplicity and detailed anatomic distribution: longitudinal study of an Australian population. J Invest Dermatol. 2009;129:323-328.
- How to select a sunscreen. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/sun-protection/how-to-select-sunscreen
- Diffey BL, Grice J. The influence of sunscreen type on photoprotection. Br J Dermatol. 1997;137:103-105.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Ou-Yang H, Stanfield J, Cole C, et al. High-SPF sunscreens (SPF ≥ 70) may provide ultraviolet protection above minimal recommended levels by adequately compensating for lower sunscreen user application amounts. J Am Acad Dermatol. 2012;67:1220-1227.
- O’Connor A. Is sunscreen flammable? The New York Times. June 6, 2012. Accessed December 15, 2020. https://well.blogs.nytimes.com/2012/06/06/is-sunscreen-flammable/
- Prevent skin cancer. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/public/spot-skin-cancer/learn-about-skin-cancer/prevent
There are numerous intrinsic risks that military servicemembers face, such as the dangers of combat, handling firearms, operating ships and heavy machinery, undersea diving, and aircraft operations. Multiple studies also have identified an increased risk for melanomas and keratinocyte cancers in those who have served on active duty.
Epidemiology
Differences in demographics are important to consider given the differences among races in the risks of skin cancers. Important racial demographic differences exist between the US Military and the general US population. Racial demographic differences also exist among the various military branches themselves. The US population is 61.0% White, 20.7% racial minorities (defined as Black or African American, Asian, American Indian or Alaska native, Native Hawaiian or other Pacific Islander, multiracial, or unknown), and 18.3% Hispanic or Latino (Hispanic or Latino was not listed as a component of racial minorities).1 According to 2018 data, the US Military population is 52.9% White, 31.0% racial minorities, and 16.1% Hispanic or Latino.2 The percentage of White military members was highest in the US Marine Corps (58.4%) and lowest in the US Navy (46.5%). The percentage of racial minorities was highest in the US Navy (38.0%) and lowest in the US Marine Corps (20.0%).2 The percentage of Hispanic and Latino military members was highest in the US Marine Corps (21.6%) and lowest in the US Air Force (14.5%).2
Melanoma in Military Members
It is estimated that the annual incidence rate of melanoma in the United States is 27 per 100,000 individuals for non-Hispanic Whites, 5 per 100,000 for Hispanics, and 1 per 100,000 for Black individuals and Asians/Pacific Islanders.3 Three studies have reviewed melanoma incidence in relation to service in the US Military.
A 2011 retrospective tumor registries study of US veterans aged 45 years or older demonstrated increased incidences of melanoma compared with the general population.4 With age, the melanoma incidence per 100,000 person-years increased in White veterans compared to their civilian counterparts (aged 45 to 49 years, 33.62 vs 27.49; aged 50 to 54 years, 49.76 vs 32.18; aged 55 to 59 years, 178.48 vs 39.17).4 An increased melanoma incidence of 62% also was seen in active-duty servicemembers aged 18 to 56 years compared to their age-matched civilian peers in a 2014 retrospective cohort study.5
Melanoma rates also vary depending on military service branch. Across 3 separate studies, service in the US Air Force was associated with the highest risk for melanoma development. A surveillance report of cancer incidence in active-duty US Armed Forces personnel between 2000 and 2011 conducted by the Defense Medical Surveillance System showed an incidence rate (per 100,000 person-years) for melanoma of 10.5 in all services, and a rate of 15.5 in the US Air Force vs 8.6 in the US Army, further highlighting the disparity between the services.6 The 2014 study also demonstrated a melanoma incidence rate of 17.80 in active-duty
Keratinocyte Cancers in Military Members
Although less well studied than melanoma, keratinocyte-derived skin cancers represent a major source of disease burden both during and after active-duty service. In a retrospective chart review of dermatology patients seen at the 86th Combat Support Hospital at Ibn Sina Hospital in Baghdad, Iraq, during a 6-month period in 2008, 8% of 2696 total visits were identified to be due to skin cancer, with the overwhelming majority being for keratinocyte cancers.7 A 1993 retrospective chart review of World War II veterans referred for Mohs micrographic surgery showed a considerably higher incidence in those who served in the Pacific Theater compared to those who served in the European Theater. Despite having approximately equal characteristics—age, skin type, and cumulative time spent outdoors—between the 2 groups, military servicemembers deployed to the Pacific represented 66% of the patients with basal cell carcinoma and 68% of the patients with squamous cell carcinoma.8
Contributing Factors
There are many factors related to military service that are likely to contribute to the increased risk for skin cancer. Based on a review of the literature, we have found an increased exposure to UV radiation, low utilization of sun-protective strategies, and low overall education regarding the risks for UV exposure to be the primary contributors to increased risks for skin cancer.
UV exposure is the primary mitigatable risk factor for developing melanoma and keratinocyte cancers.9,10 In a 2015 study of 212 military servicemembers returning from deployments in Iraq and Afghanistan, 77% reported spending more than 4 hours per day working directly in the bright sun, with 64% spending more than 75% of the average day in the bright sun.11 A 1984 study of World War II veterans diagnosed with melanoma also showed that 34% of those with melanoma had prior deployments to the tropics compared to 6% in age-matched controls.12
Even in those not deployed to overseas locations, military work still frequently involves prolonged sun exposure. In a 2015 cross-sectional study of US Air Force maintenance squadrons at Travis Air Force Base in Fairfield, California (N=356), 67% of those surveyed reported having careers that frequently involved direct sun exposure.13 This occupational sun exposure may be worsened by increased UV exposure during recreational activities, as active-duty military servicemembers may reasonably be expected to engage in more outdoor exercise and leisure activities than their civilian counterparts.
Other occupation-specific risk factors also may affect skin cancer rates in certain populations. In a study of aircraft personnel that included male military and civilian pilots, a meta-standardized incidence ratio for melanoma of 3.42 was identified compared to controls not involved in aircraft work.14 Theories to explain this increased incidence of melanoma include increased exposure to ionizing radiation at high altitudes, exposure to aviation-related chemicals, and alterations in circadian rhythm.14,15
This increased sun exposure is compounded by the overall low rates of sun protection among military members. Of those returning from Iraq and Afghanistan in the 2015 study, less than 30% of servicemembers reported routine access to sunscreen, and only 13% stated that they routinely applied sunscreen when exposed to the sun. Of this same group, only 23% endorsed that the military made them very aware of their risk for skin cancer.11 The low rates of sunscreen usage by those deployed to an active combat zone may partially be explained by the assumption that those individuals placed more emphasis on the acute dangers of combat rather than the perceived future dangers of skin cancer. A decreased availability of sunscreen for deployed military servicemembers, particularly those located at small austere bases where supplies are likely to be limited, likely makes the use of sunscreen even more difficult.
However, even within the continental United States, active-duty military servicemembers still exhibit low rates of sunscreen usage. In the 2015 study of US Air Force personnel in maintenance squadrons in California, less than 11% of those surveyed reported using sunscreen most of the time despite high rates of outdoor work.13
Another factor likely contributing to increased sun exposure and decreased sun-protection practices is the so-called invincibility complex, which is a common set of egocentric beliefs that leads to a perception that an individual is not likely to suffer the consequences of engaging in risky behaviors. Despite knowledge of the dangers associated with risky activity, individuals with an invincibility complex are more likely to view potential consequences as relevant only to others, not to themselves.16 A study of adolescent smokers in the Netherlands examined why subjects continue to smoke, despite knowledge of the potentially deadly consequences of smoking. Three common rationalizing beliefs were found: trivialization of the immediate consequences, that their smoking is only temporary and they have time in the future to stop, and that they have control over how much they smoke and can prevent fatal consequences with moderation.17 Such an invincibility complex is thought to directly run counter to the efforts of public health and educational campaigns. This belief set is thought to at least partially explain why adolescents in Australia are the most knowledgeable age cohort regarding the dangers of UV exposure but the least likely to engage in skin-protective measures.18 This inflated sense of invincibility may be leading active-duty military servicemembers to engage in unhealthy sun-exposure practices regardless of knowledge of the associated risks.
Members of the military may be uniquely susceptible to this invincibility complex. Growing evidence suggests that exposure to life-threatening circumstances may lead to long-lasting alterations in threat assessment.19,20 A 2008 study of Iraq veterans returning from deployment found that direct exposure to violent combat and human trauma was associated with an increased perceived degree of invincibility and a higher propensity to engage in risky behaviors after returning from deployment.19 Additionally, it has been speculated that individuals with a higher degree of perceived invincibility may be more likely to pursue military service, as a higher degree of self-confidence in the face of the often dangerous circumstances of military operations may be advantageous.20
In addition to scarce use of sun-protective strategies, military servicemembers also tend to lack awareness of the potential short-term and long-term harm from UV radiation. In a 2016 study of veterans undergoing treatment for skin cancer, patients reported inadequate education about skin cancer risks and strategies to decrease their chances of developing it.21 Sunscreen is less frequently used in males, specifically those aged 18 to 30 years; this demographic makes up 55.7% of the active-duty population.2,22 Low income also has been associated with decreased sunscreen use; junior enlisted military servicemembers (ranks E1-E4) make up 43.8% of the military’s ranks and make less than the average annual American household income.2,23,24
Prevention and Risk-Mitigation Strategies
Although many of the risk factors in the US Military promoting skin cancer are intrinsic to the occupation, certain steps could help minimize servicemembers’ risks. To be effective, any attempt to decrease the risk for skin cancer in the US Military must take into consideration the environment in which the military operates. To complete their mission, military personnel often are required to operate for extended periods outdoors in areas of high UV exposure, such as the deserts of Iraq or the mountains of Afghanistan. Outdoor work at times of peak sunlight often is required for successful mission completion, thus it would be ineffective to simply give blanket advice to avoid sun exposure.
Another important factor is the impact that official policy plays in shaping the daily actions of individual military servicemembers. In a hierarchical organization such as the US Military, unit commanders have substantial authority over the behaviors of their subordinates. Thus, strategies to mitigate skin cancer risks should be aimed at the individual servicemembers and unit commanders and at a policy level. Ultimately, a 3-pronged approach built on education, access to sun-protective gear, and increased availability to sunscreen is recommended.
Education
The foundation for any skin cancer prevention strategies should be built on the education of individual military servicemembers. The majority of active-duty members and veterans did not believe the military did enough to actively educate them on the risks for developing skin cancer.21 An effective educational program should focus on prevention and detection. Prevention programs should explain the role of UV exposure in the development of skin cancer, the intrinsic risks of UV exposure associated with outdoor activities, and strategies that can be implemented to reduce UV exposure and lifetime risk of skin cancer development. In a study of German outdoor workers, displays of support and concern by management regarding UV protection were associated with increases in sun-protective behaviors among the employees.25
Because patient self-examinations have been shown to be associated with earlier melanoma diagnosis and a more superficial depth at diagnosis, detection programs also should focus on the identification of suspicious skin lesions, such as by teaching the ABCDEs of melanoma.26 Among the general population, educational campaigns have been shown to be effective at reducing melanoma mortality.27,28
Access to Sun-Protective Gear
The second aspect of reducing skin cancer risk should be aiming to protect military servicemembers from UV exposure. Any prevention strategy must fit within the military’s broader tactical and strategic framework.
The use of photoprotective strategies rather than the outright avoidance of sun exposure should be implemented to minimize the deleterious effects of outdoor work. The most recent study of the UV-protective properties of US Military uniforms found all tested uniforms to have either very good or excellent UV protection, with UV protection factors (UPFs) ranging from 35 to 50+.29 However, this study was performed in 2002, and the majority of the uniforms tested are no longer in service. More up-to-date UPF information for existing military uniforms is not currently available. Most military commands wear baseball hat–style covers when operating outdoors, which generally provide good photoprotection with UPF ratings of 35 to 50 over the protected areas.29 Unfortunately, these types of headgear offer less photoprotection than do wide-brimmed hats, which have demonstrated improved photoprotection, particularly of the neck, cheeks, ears, and chin.30 A wide-brimmed hat, known as the boonie hat, was originally proposed for military use in 1966 to provide protection of servicemembers’ faces and necks from the intense sun of Vietnam. Currently, the use of the boonie hat typically is prohibited for units not engaged in combat or combat-support roles and requires authorization by the unit-level commander.31 Because of its perception as “unmilitary appearing” by many unit commanders and its restriction of use to combat-related units, the boonie hat is not consistently used. Increasing the use of this type of wide-brimmed hat would be an important asset in decreasing chronic UV exposure in military servicemembers, particularly on those parts of the body where skin cancer occurrence is the greatest.32 Policies should be aimed at increasing the use of the boonie hat, both through expanding its availability to troops in non–combat-related fields and by encouraging unit commanders to authorize its use in their units.
Sunscreen Availability
Improving the use of sunscreen is another impactful strategy that could be undertaken to decrease the risk for skin cancer in military servicemembers. The use of sunscreen is low in both those deployed overseas and those stationed within the United States. Improving access to sunscreen, particularly in the deployed setting, also could reduce barriers to use. Providing sunscreen directly to servicemembers, either when issuing gear or integrated within Meals Ready to Eat, could remove both the financial and logistical barriers to sunscreen utilization. Centralized troop-gathering locations, such as dining facilities, could be utilized both for the mass distribution of sunscreen and to display educational material. Unit commanders also could mandate times for servicemembers to stop work and apply sunscreen at regularly scheduled intervals.
The composition and delivery vehicle of sunscreen may have an impact on its efficacy and ease of use in the field. The American Academy of Dermatology (AAD) recommends using sunscreen that is broad spectrum, sun protection factor (SPF) 30 or greater, and water resistant.33 However, the AAD does not make a recommendation of whether to use a physical sunscreen (such as titanium dioxide) or a chemical sunscreen. If applied in equal amounts, a chemical sunscreen and a physical sunscreen with an equal SPF should offer the same UV protection. However, a study in the British Journal of Dermatology showed that subjects applied only two-thirds the quantity of physical sunscreen compared to those applying chemical sunscreen, achieving approximately only one-half the SPF as provided by the chemical sunscreen.34 Because sunscreen is only effective when it is used, consideration should be given to the preferences of the military population when selecting sunscreens. A review of consumer preferences of sunscreen qualities showed that sunscreens that were nongreasy and did not leave a residue were given the most favorable rankings.35 In recent years, sunscreen sprays have become increasingly popular. When adequately applied, sprays have been shown to be equally effective as sunscreen lotions.36 However, although recommendations have been issued by both the AAD and the US Food and Drug Administration on the application of sunscreen lotion to adequately cover exposed skin, no such recommendations have been given for sunscreen sprays.33 Some safety concerns also remain regarding the flammability of aerosol sunscreens, which could be exacerbated in a combat situation.37
However, there are some obvious downsides to sunscreen use. During certain operational tasks, particularly in combat settings, it may not be feasible or even safe to stop working to apply sunscreen at the 2-hour intervals required for effective UV protection.38 Water exposure or large amounts of perspiration also would cause sunscreen to lose effectiveness earlier than expected. Logistically, it may be challenging to regularly supply sunscreen to small austere bases in remote locations.
Final Thoughts
The men and women of our armed forces already undertake great risk in the defense of our country. It should be ensured that their risk for developing skin cancer is made as low as possible, while still allowing them to successfully accomplish their mission. Multiple studies have shown servicemembers to be at an increased risk for skin cancer, particularly melanoma. We believe the primary factor behind this increased risk is occupational UV exposure, which is compounded by the suboptimal use of sun-protective strategies. By educating our servicemembers about their risk for skin cancer and promoting increased UV protection, we can effectively reduce the burden of skin cancer on our active-duty servicemembers and veterans.
There are numerous intrinsic risks that military servicemembers face, such as the dangers of combat, handling firearms, operating ships and heavy machinery, undersea diving, and aircraft operations. Multiple studies also have identified an increased risk for melanomas and keratinocyte cancers in those who have served on active duty.
Epidemiology
Differences in demographics are important to consider given the differences among races in the risks of skin cancers. Important racial demographic differences exist between the US Military and the general US population. Racial demographic differences also exist among the various military branches themselves. The US population is 61.0% White, 20.7% racial minorities (defined as Black or African American, Asian, American Indian or Alaska native, Native Hawaiian or other Pacific Islander, multiracial, or unknown), and 18.3% Hispanic or Latino (Hispanic or Latino was not listed as a component of racial minorities).1 According to 2018 data, the US Military population is 52.9% White, 31.0% racial minorities, and 16.1% Hispanic or Latino.2 The percentage of White military members was highest in the US Marine Corps (58.4%) and lowest in the US Navy (46.5%). The percentage of racial minorities was highest in the US Navy (38.0%) and lowest in the US Marine Corps (20.0%).2 The percentage of Hispanic and Latino military members was highest in the US Marine Corps (21.6%) and lowest in the US Air Force (14.5%).2
Melanoma in Military Members
It is estimated that the annual incidence rate of melanoma in the United States is 27 per 100,000 individuals for non-Hispanic Whites, 5 per 100,000 for Hispanics, and 1 per 100,000 for Black individuals and Asians/Pacific Islanders.3 Three studies have reviewed melanoma incidence in relation to service in the US Military.
A 2011 retrospective tumor registries study of US veterans aged 45 years or older demonstrated increased incidences of melanoma compared with the general population.4 With age, the melanoma incidence per 100,000 person-years increased in White veterans compared to their civilian counterparts (aged 45 to 49 years, 33.62 vs 27.49; aged 50 to 54 years, 49.76 vs 32.18; aged 55 to 59 years, 178.48 vs 39.17).4 An increased melanoma incidence of 62% also was seen in active-duty servicemembers aged 18 to 56 years compared to their age-matched civilian peers in a 2014 retrospective cohort study.5
Melanoma rates also vary depending on military service branch. Across 3 separate studies, service in the US Air Force was associated with the highest risk for melanoma development. A surveillance report of cancer incidence in active-duty US Armed Forces personnel between 2000 and 2011 conducted by the Defense Medical Surveillance System showed an incidence rate (per 100,000 person-years) for melanoma of 10.5 in all services, and a rate of 15.5 in the US Air Force vs 8.6 in the US Army, further highlighting the disparity between the services.6 The 2014 study also demonstrated a melanoma incidence rate of 17.80 in active-duty
Keratinocyte Cancers in Military Members
Although less well studied than melanoma, keratinocyte-derived skin cancers represent a major source of disease burden both during and after active-duty service. In a retrospective chart review of dermatology patients seen at the 86th Combat Support Hospital at Ibn Sina Hospital in Baghdad, Iraq, during a 6-month period in 2008, 8% of 2696 total visits were identified to be due to skin cancer, with the overwhelming majority being for keratinocyte cancers.7 A 1993 retrospective chart review of World War II veterans referred for Mohs micrographic surgery showed a considerably higher incidence in those who served in the Pacific Theater compared to those who served in the European Theater. Despite having approximately equal characteristics—age, skin type, and cumulative time spent outdoors—between the 2 groups, military servicemembers deployed to the Pacific represented 66% of the patients with basal cell carcinoma and 68% of the patients with squamous cell carcinoma.8
Contributing Factors
There are many factors related to military service that are likely to contribute to the increased risk for skin cancer. Based on a review of the literature, we have found an increased exposure to UV radiation, low utilization of sun-protective strategies, and low overall education regarding the risks for UV exposure to be the primary contributors to increased risks for skin cancer.
UV exposure is the primary mitigatable risk factor for developing melanoma and keratinocyte cancers.9,10 In a 2015 study of 212 military servicemembers returning from deployments in Iraq and Afghanistan, 77% reported spending more than 4 hours per day working directly in the bright sun, with 64% spending more than 75% of the average day in the bright sun.11 A 1984 study of World War II veterans diagnosed with melanoma also showed that 34% of those with melanoma had prior deployments to the tropics compared to 6% in age-matched controls.12
Even in those not deployed to overseas locations, military work still frequently involves prolonged sun exposure. In a 2015 cross-sectional study of US Air Force maintenance squadrons at Travis Air Force Base in Fairfield, California (N=356), 67% of those surveyed reported having careers that frequently involved direct sun exposure.13 This occupational sun exposure may be worsened by increased UV exposure during recreational activities, as active-duty military servicemembers may reasonably be expected to engage in more outdoor exercise and leisure activities than their civilian counterparts.
Other occupation-specific risk factors also may affect skin cancer rates in certain populations. In a study of aircraft personnel that included male military and civilian pilots, a meta-standardized incidence ratio for melanoma of 3.42 was identified compared to controls not involved in aircraft work.14 Theories to explain this increased incidence of melanoma include increased exposure to ionizing radiation at high altitudes, exposure to aviation-related chemicals, and alterations in circadian rhythm.14,15
This increased sun exposure is compounded by the overall low rates of sun protection among military members. Of those returning from Iraq and Afghanistan in the 2015 study, less than 30% of servicemembers reported routine access to sunscreen, and only 13% stated that they routinely applied sunscreen when exposed to the sun. Of this same group, only 23% endorsed that the military made them very aware of their risk for skin cancer.11 The low rates of sunscreen usage by those deployed to an active combat zone may partially be explained by the assumption that those individuals placed more emphasis on the acute dangers of combat rather than the perceived future dangers of skin cancer. A decreased availability of sunscreen for deployed military servicemembers, particularly those located at small austere bases where supplies are likely to be limited, likely makes the use of sunscreen even more difficult.
However, even within the continental United States, active-duty military servicemembers still exhibit low rates of sunscreen usage. In the 2015 study of US Air Force personnel in maintenance squadrons in California, less than 11% of those surveyed reported using sunscreen most of the time despite high rates of outdoor work.13
Another factor likely contributing to increased sun exposure and decreased sun-protection practices is the so-called invincibility complex, which is a common set of egocentric beliefs that leads to a perception that an individual is not likely to suffer the consequences of engaging in risky behaviors. Despite knowledge of the dangers associated with risky activity, individuals with an invincibility complex are more likely to view potential consequences as relevant only to others, not to themselves.16 A study of adolescent smokers in the Netherlands examined why subjects continue to smoke, despite knowledge of the potentially deadly consequences of smoking. Three common rationalizing beliefs were found: trivialization of the immediate consequences, that their smoking is only temporary and they have time in the future to stop, and that they have control over how much they smoke and can prevent fatal consequences with moderation.17 Such an invincibility complex is thought to directly run counter to the efforts of public health and educational campaigns. This belief set is thought to at least partially explain why adolescents in Australia are the most knowledgeable age cohort regarding the dangers of UV exposure but the least likely to engage in skin-protective measures.18 This inflated sense of invincibility may be leading active-duty military servicemembers to engage in unhealthy sun-exposure practices regardless of knowledge of the associated risks.
Members of the military may be uniquely susceptible to this invincibility complex. Growing evidence suggests that exposure to life-threatening circumstances may lead to long-lasting alterations in threat assessment.19,20 A 2008 study of Iraq veterans returning from deployment found that direct exposure to violent combat and human trauma was associated with an increased perceived degree of invincibility and a higher propensity to engage in risky behaviors after returning from deployment.19 Additionally, it has been speculated that individuals with a higher degree of perceived invincibility may be more likely to pursue military service, as a higher degree of self-confidence in the face of the often dangerous circumstances of military operations may be advantageous.20
In addition to scarce use of sun-protective strategies, military servicemembers also tend to lack awareness of the potential short-term and long-term harm from UV radiation. In a 2016 study of veterans undergoing treatment for skin cancer, patients reported inadequate education about skin cancer risks and strategies to decrease their chances of developing it.21 Sunscreen is less frequently used in males, specifically those aged 18 to 30 years; this demographic makes up 55.7% of the active-duty population.2,22 Low income also has been associated with decreased sunscreen use; junior enlisted military servicemembers (ranks E1-E4) make up 43.8% of the military’s ranks and make less than the average annual American household income.2,23,24
Prevention and Risk-Mitigation Strategies
Although many of the risk factors in the US Military promoting skin cancer are intrinsic to the occupation, certain steps could help minimize servicemembers’ risks. To be effective, any attempt to decrease the risk for skin cancer in the US Military must take into consideration the environment in which the military operates. To complete their mission, military personnel often are required to operate for extended periods outdoors in areas of high UV exposure, such as the deserts of Iraq or the mountains of Afghanistan. Outdoor work at times of peak sunlight often is required for successful mission completion, thus it would be ineffective to simply give blanket advice to avoid sun exposure.
Another important factor is the impact that official policy plays in shaping the daily actions of individual military servicemembers. In a hierarchical organization such as the US Military, unit commanders have substantial authority over the behaviors of their subordinates. Thus, strategies to mitigate skin cancer risks should be aimed at the individual servicemembers and unit commanders and at a policy level. Ultimately, a 3-pronged approach built on education, access to sun-protective gear, and increased availability to sunscreen is recommended.
Education
The foundation for any skin cancer prevention strategies should be built on the education of individual military servicemembers. The majority of active-duty members and veterans did not believe the military did enough to actively educate them on the risks for developing skin cancer.21 An effective educational program should focus on prevention and detection. Prevention programs should explain the role of UV exposure in the development of skin cancer, the intrinsic risks of UV exposure associated with outdoor activities, and strategies that can be implemented to reduce UV exposure and lifetime risk of skin cancer development. In a study of German outdoor workers, displays of support and concern by management regarding UV protection were associated with increases in sun-protective behaviors among the employees.25
Because patient self-examinations have been shown to be associated with earlier melanoma diagnosis and a more superficial depth at diagnosis, detection programs also should focus on the identification of suspicious skin lesions, such as by teaching the ABCDEs of melanoma.26 Among the general population, educational campaigns have been shown to be effective at reducing melanoma mortality.27,28
Access to Sun-Protective Gear
The second aspect of reducing skin cancer risk should be aiming to protect military servicemembers from UV exposure. Any prevention strategy must fit within the military’s broader tactical and strategic framework.
The use of photoprotective strategies rather than the outright avoidance of sun exposure should be implemented to minimize the deleterious effects of outdoor work. The most recent study of the UV-protective properties of US Military uniforms found all tested uniforms to have either very good or excellent UV protection, with UV protection factors (UPFs) ranging from 35 to 50+.29 However, this study was performed in 2002, and the majority of the uniforms tested are no longer in service. More up-to-date UPF information for existing military uniforms is not currently available. Most military commands wear baseball hat–style covers when operating outdoors, which generally provide good photoprotection with UPF ratings of 35 to 50 over the protected areas.29 Unfortunately, these types of headgear offer less photoprotection than do wide-brimmed hats, which have demonstrated improved photoprotection, particularly of the neck, cheeks, ears, and chin.30 A wide-brimmed hat, known as the boonie hat, was originally proposed for military use in 1966 to provide protection of servicemembers’ faces and necks from the intense sun of Vietnam. Currently, the use of the boonie hat typically is prohibited for units not engaged in combat or combat-support roles and requires authorization by the unit-level commander.31 Because of its perception as “unmilitary appearing” by many unit commanders and its restriction of use to combat-related units, the boonie hat is not consistently used. Increasing the use of this type of wide-brimmed hat would be an important asset in decreasing chronic UV exposure in military servicemembers, particularly on those parts of the body where skin cancer occurrence is the greatest.32 Policies should be aimed at increasing the use of the boonie hat, both through expanding its availability to troops in non–combat-related fields and by encouraging unit commanders to authorize its use in their units.
Sunscreen Availability
Improving the use of sunscreen is another impactful strategy that could be undertaken to decrease the risk for skin cancer in military servicemembers. The use of sunscreen is low in both those deployed overseas and those stationed within the United States. Improving access to sunscreen, particularly in the deployed setting, also could reduce barriers to use. Providing sunscreen directly to servicemembers, either when issuing gear or integrated within Meals Ready to Eat, could remove both the financial and logistical barriers to sunscreen utilization. Centralized troop-gathering locations, such as dining facilities, could be utilized both for the mass distribution of sunscreen and to display educational material. Unit commanders also could mandate times for servicemembers to stop work and apply sunscreen at regularly scheduled intervals.
The composition and delivery vehicle of sunscreen may have an impact on its efficacy and ease of use in the field. The American Academy of Dermatology (AAD) recommends using sunscreen that is broad spectrum, sun protection factor (SPF) 30 or greater, and water resistant.33 However, the AAD does not make a recommendation of whether to use a physical sunscreen (such as titanium dioxide) or a chemical sunscreen. If applied in equal amounts, a chemical sunscreen and a physical sunscreen with an equal SPF should offer the same UV protection. However, a study in the British Journal of Dermatology showed that subjects applied only two-thirds the quantity of physical sunscreen compared to those applying chemical sunscreen, achieving approximately only one-half the SPF as provided by the chemical sunscreen.34 Because sunscreen is only effective when it is used, consideration should be given to the preferences of the military population when selecting sunscreens. A review of consumer preferences of sunscreen qualities showed that sunscreens that were nongreasy and did not leave a residue were given the most favorable rankings.35 In recent years, sunscreen sprays have become increasingly popular. When adequately applied, sprays have been shown to be equally effective as sunscreen lotions.36 However, although recommendations have been issued by both the AAD and the US Food and Drug Administration on the application of sunscreen lotion to adequately cover exposed skin, no such recommendations have been given for sunscreen sprays.33 Some safety concerns also remain regarding the flammability of aerosol sunscreens, which could be exacerbated in a combat situation.37
However, there are some obvious downsides to sunscreen use. During certain operational tasks, particularly in combat settings, it may not be feasible or even safe to stop working to apply sunscreen at the 2-hour intervals required for effective UV protection.38 Water exposure or large amounts of perspiration also would cause sunscreen to lose effectiveness earlier than expected. Logistically, it may be challenging to regularly supply sunscreen to small austere bases in remote locations.
Final Thoughts
The men and women of our armed forces already undertake great risk in the defense of our country. It should be ensured that their risk for developing skin cancer is made as low as possible, while still allowing them to successfully accomplish their mission. Multiple studies have shown servicemembers to be at an increased risk for skin cancer, particularly melanoma. We believe the primary factor behind this increased risk is occupational UV exposure, which is compounded by the suboptimal use of sun-protective strategies. By educating our servicemembers about their risk for skin cancer and promoting increased UV protection, we can effectively reduce the burden of skin cancer on our active-duty servicemembers and veterans.
- QuickFacts. United States Census Bureau. Accessed December 15, 2020. https://www.census.gov/quickfacts/fact/table/US/PST045219
- 2018 Demographics Profile. Military OneSource. Accessed December 15, 2020. https://www.militaryonesource.mil/reports-and-surveys/infographics/active-duty-member-and-family-demographics
- Cancer Facts & Figures 2019. American Cancer Society. Accessed December 15, 2020. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2019.html
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Military Med. 2014;179:247-253.
- Armed Forces Health Surveillance Center. Incident diagnoses of cancers and cancer-related deaths, active component, US Armed Forces, 2000-2011. MSMR. 2012;19:18-22.
- Henning JS, Firoz BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Ramani ML, Bennett RG. High prevalence of skin-cancer in World-War-II servicemen stationed in the Pacific Theater. J Am Acad Dermatol. 1993;28:733-737.
- Schmitt J, Seidler A, Diepgen TL, et al. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: a systematic review and meta-analysis. Br J Dermatol. 2011;164:291-307.
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. J Photochem Photobiol B. 2001;63:8-18.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Brown J, Kopf AW, Rica DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Parker G, Williams B, Driggers P. Sun exposure knowledge and practices survey of maintenance squadrons at Travis AFB. Military Med. 2015;180:26-31.
- Buja A, Lange JH, Perissinotto E, et al. Cancer incidence among male military and civil pilots and flight attendants: an analysis on published data. Toxicol Ind Health. 2005;21:273-282.
- Wilkison BD, Wong EB. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Wickman ME, Anderson NLR, Smith Greenberg C. The adolescent perception of invincibility and its influence on teen acceptance of health promotion strategies. J Pediatr Nurs. 2008;23:460-468.
- Schreuders M, Krooneman NT, van den Putte B, et al. Boy smokers’ rationalisations for engaging in potentially fatal behaviour: in-depth interviews in the Netherlands. Int J Environ Res Public Health. 2018;15:767.
- Eastabrook S, Chang P, Taylor MF. Melanoma risk: adolescent females’ perspectives on skin protection pre/post-viewing a ultraviolet photoaged photograph of their own facial sun damage. Glob Health Promot. 2018;25:23-32.
- Killgore WD, Cotting DI, Thomas JL, et al. Post-combat invincibility: violent combat experiences are associated with increased risk-taking propensity following deployment. J Psychiatr Res. 2008;42:1112-1121.
- Killgore WD, Kelley A, Balkin TJ. So you think you’re bulletproof: development and validation of the Invincibility Belief Index (IBI). Military Med. 2010;175:499-508.
- McGrath JM, Fisher V, Krejci-Manwaring J. Skin cancer warnings and the need for new preventive campaigns: a pilot study. Am J Prevent Med. 2016;50:E62-E63.
- Thieden E, Philipsen PA, Sandby-Moller J, et al. Sunscreen use related to UV exposure, age, sex, and occupation based on personal dosimeter readings and sun-exposure behavior diaries. Arch Dermatol. 2005;141:967-973.
- Holman DM, Berkowitz Z, Guy GP Jr, et al. Patterns of sunscreen use on the face and other exposed skin among US adults. J Am Acad Dermatol. 2015;73:83-92.e1.
- Military Pay Tables & Information. Defense Finance and Accounting Service website. Accessed December 21, 2020. https://www.dfas.mil/militarymembers/payentitlements/Pay-Tables.html
- Schilling L, Schneider S, Gorig T, et al. “Lost in the sun”—the key role of perceived workplace support for sun-protective behavior in outdoor workers. Am J Ind Med. 2018;61:929-938.
- Uliasz A, Lebwohl M. Patient education and regular surveillance results in earlier diagnosis of second primary melanoma. Int J Dermatol. 2007;46:575-577.
- MacKie RM, Hole D. Audit of public education campaign to encourage earlier detection of malignant melanoma. BMJ. 1992;304:1012-1015.
- Berwick M, Begg CB, Fine JA, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
- Winterhalter C, DiLuna K, Bide M. Characterization of the ultraviolet protection of combat uniform fabrics. US Army Soldier and Biological Chemical Command Soldier Systems Center technical report Natick/TR-02/006. Published January 21, 2002. Accessed December 21, 2021. https://apps.dtic.mil/dtic/tr/fulltext/u2/a398572.pdf
- Gies P, Javorniczky J, Roy C, et al. Measurements of the UVR protection provided by hats used at school. Photochem Photobiol. 2006;82:750-754.
- Stanton S. Headgear. In: Stanton S. US Army Uniforms of the Vietnam War. Stackpole Books; 1992:26-61.
- Richmond-Sinclair NM, Pandeya N, Ware RS, et al. Incidence of basal cell carcinoma multiplicity and detailed anatomic distribution: longitudinal study of an Australian population. J Invest Dermatol. 2009;129:323-328.
- How to select a sunscreen. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/sun-protection/how-to-select-sunscreen
- Diffey BL, Grice J. The influence of sunscreen type on photoprotection. Br J Dermatol. 1997;137:103-105.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Ou-Yang H, Stanfield J, Cole C, et al. High-SPF sunscreens (SPF ≥ 70) may provide ultraviolet protection above minimal recommended levels by adequately compensating for lower sunscreen user application amounts. J Am Acad Dermatol. 2012;67:1220-1227.
- O’Connor A. Is sunscreen flammable? The New York Times. June 6, 2012. Accessed December 15, 2020. https://well.blogs.nytimes.com/2012/06/06/is-sunscreen-flammable/
- Prevent skin cancer. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/public/spot-skin-cancer/learn-about-skin-cancer/prevent
- QuickFacts. United States Census Bureau. Accessed December 15, 2020. https://www.census.gov/quickfacts/fact/table/US/PST045219
- 2018 Demographics Profile. Military OneSource. Accessed December 15, 2020. https://www.militaryonesource.mil/reports-and-surveys/infographics/active-duty-member-and-family-demographics
- Cancer Facts & Figures 2019. American Cancer Society. Accessed December 15, 2020. https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2019.html
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Military Med. 2014;179:247-253.
- Armed Forces Health Surveillance Center. Incident diagnoses of cancers and cancer-related deaths, active component, US Armed Forces, 2000-2011. MSMR. 2012;19:18-22.
- Henning JS, Firoz BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Ramani ML, Bennett RG. High prevalence of skin-cancer in World-War-II servicemen stationed in the Pacific Theater. J Am Acad Dermatol. 1993;28:733-737.
- Schmitt J, Seidler A, Diepgen TL, et al. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: a systematic review and meta-analysis. Br J Dermatol. 2011;164:291-307.
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. J Photochem Photobiol B. 2001;63:8-18.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Brown J, Kopf AW, Rica DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Parker G, Williams B, Driggers P. Sun exposure knowledge and practices survey of maintenance squadrons at Travis AFB. Military Med. 2015;180:26-31.
- Buja A, Lange JH, Perissinotto E, et al. Cancer incidence among male military and civil pilots and flight attendants: an analysis on published data. Toxicol Ind Health. 2005;21:273-282.
- Wilkison BD, Wong EB. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Wickman ME, Anderson NLR, Smith Greenberg C. The adolescent perception of invincibility and its influence on teen acceptance of health promotion strategies. J Pediatr Nurs. 2008;23:460-468.
- Schreuders M, Krooneman NT, van den Putte B, et al. Boy smokers’ rationalisations for engaging in potentially fatal behaviour: in-depth interviews in the Netherlands. Int J Environ Res Public Health. 2018;15:767.
- Eastabrook S, Chang P, Taylor MF. Melanoma risk: adolescent females’ perspectives on skin protection pre/post-viewing a ultraviolet photoaged photograph of their own facial sun damage. Glob Health Promot. 2018;25:23-32.
- Killgore WD, Cotting DI, Thomas JL, et al. Post-combat invincibility: violent combat experiences are associated with increased risk-taking propensity following deployment. J Psychiatr Res. 2008;42:1112-1121.
- Killgore WD, Kelley A, Balkin TJ. So you think you’re bulletproof: development and validation of the Invincibility Belief Index (IBI). Military Med. 2010;175:499-508.
- McGrath JM, Fisher V, Krejci-Manwaring J. Skin cancer warnings and the need for new preventive campaigns: a pilot study. Am J Prevent Med. 2016;50:E62-E63.
- Thieden E, Philipsen PA, Sandby-Moller J, et al. Sunscreen use related to UV exposure, age, sex, and occupation based on personal dosimeter readings and sun-exposure behavior diaries. Arch Dermatol. 2005;141:967-973.
- Holman DM, Berkowitz Z, Guy GP Jr, et al. Patterns of sunscreen use on the face and other exposed skin among US adults. J Am Acad Dermatol. 2015;73:83-92.e1.
- Military Pay Tables & Information. Defense Finance and Accounting Service website. Accessed December 21, 2020. https://www.dfas.mil/militarymembers/payentitlements/Pay-Tables.html
- Schilling L, Schneider S, Gorig T, et al. “Lost in the sun”—the key role of perceived workplace support for sun-protective behavior in outdoor workers. Am J Ind Med. 2018;61:929-938.
- Uliasz A, Lebwohl M. Patient education and regular surveillance results in earlier diagnosis of second primary melanoma. Int J Dermatol. 2007;46:575-577.
- MacKie RM, Hole D. Audit of public education campaign to encourage earlier detection of malignant melanoma. BMJ. 1992;304:1012-1015.
- Berwick M, Begg CB, Fine JA, et al. Screening for cutaneous melanoma by skin self-examination. J Natl Cancer Inst. 1996;88:17-23.
- Winterhalter C, DiLuna K, Bide M. Characterization of the ultraviolet protection of combat uniform fabrics. US Army Soldier and Biological Chemical Command Soldier Systems Center technical report Natick/TR-02/006. Published January 21, 2002. Accessed December 21, 2021. https://apps.dtic.mil/dtic/tr/fulltext/u2/a398572.pdf
- Gies P, Javorniczky J, Roy C, et al. Measurements of the UVR protection provided by hats used at school. Photochem Photobiol. 2006;82:750-754.
- Stanton S. Headgear. In: Stanton S. US Army Uniforms of the Vietnam War. Stackpole Books; 1992:26-61.
- Richmond-Sinclair NM, Pandeya N, Ware RS, et al. Incidence of basal cell carcinoma multiplicity and detailed anatomic distribution: longitudinal study of an Australian population. J Invest Dermatol. 2009;129:323-328.
- How to select a sunscreen. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/sun-protection/how-to-select-sunscreen
- Diffey BL, Grice J. The influence of sunscreen type on photoprotection. Br J Dermatol. 1997;137:103-105.
- Xu S, Kwa M, Agarwal A, et al. Sunscreen product performance and other determinants of consumer preferences. JAMA Dermatol. 2016;152:920-927.
- Ou-Yang H, Stanfield J, Cole C, et al. High-SPF sunscreens (SPF ≥ 70) may provide ultraviolet protection above minimal recommended levels by adequately compensating for lower sunscreen user application amounts. J Am Acad Dermatol. 2012;67:1220-1227.
- O’Connor A. Is sunscreen flammable? The New York Times. June 6, 2012. Accessed December 15, 2020. https://well.blogs.nytimes.com/2012/06/06/is-sunscreen-flammable/
- Prevent skin cancer. American Academy of Dermatology. Accessed December 15, 2020. https://www.aad.org/public/spot-skin-cancer/learn-about-skin-cancer/prevent
Practice Points
- An increased risk for melanoma and keratinocyte carcinomas has been identified in those who have served in the US Military.
- UV radiation exposure, low utilization of sun-protective strategies, and low overall education regarding the risks of UV exposure appear to be the primary contributors to increased risks of skin cancer in this population.
- Improving education for military servicemembers on the risks of UV exposure, increasing utilization of sun-protective clothing, and improving access and utilization of sunscreen are viable options to decrease the risk for cutaneous malignancies in US Military servicemembers.
COVID-19 vaccines and cancer patients: 4 things to know
Earlier this week, Medscape spoke with Nora Disis, MD, about vaccinating cancer patients. Disis is a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, the University of Washington, Seattle, Washington. As editor-in-chief of JAMA Oncology, she has watched COVID-19 developments in the oncology community over the past year.
Here are a few themes that Disis said oncologists should be aware of as vaccines eventually begin reaching cancer patients.
We should expect cancer patients to respond to vaccines. Historically, some believed that cancer patients would be unable to mount an immune response to vaccines. Data on other viral vaccines have shown otherwise. For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection. Likewise for pneumococcal vaccine, which, generally speaking, cancer patients should receive.
Special cases may include hematologic malignancies in which the immune system has been destroyed and profound immunosuppression occurs. Data on immunization during this immunosuppressed period are scarce, but what data are available suggest that once cancer patients are through this immunosuppressed period, they can be vaccinated successfully.
The type of vaccine will probably be important for cancer patients. Currently, there are 61 coronavirus vaccines in human clinical trials, and 17 have reached the final stages of testing. At least 85 preclinical vaccines are under active investigation in animals.
Both the Pfizer-BioNTech and Moderna COVID vaccines are mRNA type. There are many other types, including protein-based vaccines, viral vector vaccines based on adenoviruses, and inactivated or attenuated coronavirus vaccines.
The latter vaccines, particularly attenuated live virus vaccines, may not be a good choice for cancer patients. Especially in those with rapidly progressing disease or on chemotherapy, attenuated live viruses may cause a low-grade infection.
Incidentally, the technology used in the genetic, or mRNA, vaccines developed by both Pfizer-BioNTech and Moderna was initially developed for fighting cancer, and studies have shown that patients can generate immune responses to cancer-associated proteins with this type of vaccine.
These genetic vaccines could turn out to be the most effective for cancer patients, especially those with solid tumors.
Our understanding is very limited right now. Neither the Pfizer-BioNTech nor the Moderna early data discuss cancer patients. Two of the most important questions for cancer patients are dosing and booster scheduling. Potential defects in lymphocyte function among cancer patients may require unique initial dosing and booster schedules. In terms of timing, it is unclear how active therapy might affect a patient’s immune response to vaccination and whether vaccines should be timed with therapy cycles.
Vaccine access may depend on whether cancer patients are viewed as a vulnerable population. Those at higher risk for severe COVID-19 clearly have a greater need for vaccination. While there are data suggesting that cancer patients are at higher risk, they are a bit murky, in part because cancer patients are a heterogeneous group. For example, there are data suggesting that lung and blood cancer patients fare worse. There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.
It is likely, then, that personalized risk factors such as type of cancer therapy, site of disease, and comorbidities will shape individual choices about vaccination among cancer patients.
A version of this article first appeared on Medscape.com.
Earlier this week, Medscape spoke with Nora Disis, MD, about vaccinating cancer patients. Disis is a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, the University of Washington, Seattle, Washington. As editor-in-chief of JAMA Oncology, she has watched COVID-19 developments in the oncology community over the past year.
Here are a few themes that Disis said oncologists should be aware of as vaccines eventually begin reaching cancer patients.
We should expect cancer patients to respond to vaccines. Historically, some believed that cancer patients would be unable to mount an immune response to vaccines. Data on other viral vaccines have shown otherwise. For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection. Likewise for pneumococcal vaccine, which, generally speaking, cancer patients should receive.
Special cases may include hematologic malignancies in which the immune system has been destroyed and profound immunosuppression occurs. Data on immunization during this immunosuppressed period are scarce, but what data are available suggest that once cancer patients are through this immunosuppressed period, they can be vaccinated successfully.
The type of vaccine will probably be important for cancer patients. Currently, there are 61 coronavirus vaccines in human clinical trials, and 17 have reached the final stages of testing. At least 85 preclinical vaccines are under active investigation in animals.
Both the Pfizer-BioNTech and Moderna COVID vaccines are mRNA type. There are many other types, including protein-based vaccines, viral vector vaccines based on adenoviruses, and inactivated or attenuated coronavirus vaccines.
The latter vaccines, particularly attenuated live virus vaccines, may not be a good choice for cancer patients. Especially in those with rapidly progressing disease or on chemotherapy, attenuated live viruses may cause a low-grade infection.
Incidentally, the technology used in the genetic, or mRNA, vaccines developed by both Pfizer-BioNTech and Moderna was initially developed for fighting cancer, and studies have shown that patients can generate immune responses to cancer-associated proteins with this type of vaccine.
These genetic vaccines could turn out to be the most effective for cancer patients, especially those with solid tumors.
Our understanding is very limited right now. Neither the Pfizer-BioNTech nor the Moderna early data discuss cancer patients. Two of the most important questions for cancer patients are dosing and booster scheduling. Potential defects in lymphocyte function among cancer patients may require unique initial dosing and booster schedules. In terms of timing, it is unclear how active therapy might affect a patient’s immune response to vaccination and whether vaccines should be timed with therapy cycles.
Vaccine access may depend on whether cancer patients are viewed as a vulnerable population. Those at higher risk for severe COVID-19 clearly have a greater need for vaccination. While there are data suggesting that cancer patients are at higher risk, they are a bit murky, in part because cancer patients are a heterogeneous group. For example, there are data suggesting that lung and blood cancer patients fare worse. There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.
It is likely, then, that personalized risk factors such as type of cancer therapy, site of disease, and comorbidities will shape individual choices about vaccination among cancer patients.
A version of this article first appeared on Medscape.com.
Earlier this week, Medscape spoke with Nora Disis, MD, about vaccinating cancer patients. Disis is a medical oncologist and director of both the Institute of Translational Health Sciences and the Cancer Vaccine Institute, the University of Washington, Seattle, Washington. As editor-in-chief of JAMA Oncology, she has watched COVID-19 developments in the oncology community over the past year.
Here are a few themes that Disis said oncologists should be aware of as vaccines eventually begin reaching cancer patients.
We should expect cancer patients to respond to vaccines. Historically, some believed that cancer patients would be unable to mount an immune response to vaccines. Data on other viral vaccines have shown otherwise. For example, there has been a long history of studies of flu vaccination in cancer patients, and in general, those vaccines confer protection. Likewise for pneumococcal vaccine, which, generally speaking, cancer patients should receive.
Special cases may include hematologic malignancies in which the immune system has been destroyed and profound immunosuppression occurs. Data on immunization during this immunosuppressed period are scarce, but what data are available suggest that once cancer patients are through this immunosuppressed period, they can be vaccinated successfully.
The type of vaccine will probably be important for cancer patients. Currently, there are 61 coronavirus vaccines in human clinical trials, and 17 have reached the final stages of testing. At least 85 preclinical vaccines are under active investigation in animals.
Both the Pfizer-BioNTech and Moderna COVID vaccines are mRNA type. There are many other types, including protein-based vaccines, viral vector vaccines based on adenoviruses, and inactivated or attenuated coronavirus vaccines.
The latter vaccines, particularly attenuated live virus vaccines, may not be a good choice for cancer patients. Especially in those with rapidly progressing disease or on chemotherapy, attenuated live viruses may cause a low-grade infection.
Incidentally, the technology used in the genetic, or mRNA, vaccines developed by both Pfizer-BioNTech and Moderna was initially developed for fighting cancer, and studies have shown that patients can generate immune responses to cancer-associated proteins with this type of vaccine.
These genetic vaccines could turn out to be the most effective for cancer patients, especially those with solid tumors.
Our understanding is very limited right now. Neither the Pfizer-BioNTech nor the Moderna early data discuss cancer patients. Two of the most important questions for cancer patients are dosing and booster scheduling. Potential defects in lymphocyte function among cancer patients may require unique initial dosing and booster schedules. In terms of timing, it is unclear how active therapy might affect a patient’s immune response to vaccination and whether vaccines should be timed with therapy cycles.
Vaccine access may depend on whether cancer patients are viewed as a vulnerable population. Those at higher risk for severe COVID-19 clearly have a greater need for vaccination. While there are data suggesting that cancer patients are at higher risk, they are a bit murky, in part because cancer patients are a heterogeneous group. For example, there are data suggesting that lung and blood cancer patients fare worse. There is also a suggestion that, like in the general population, COVID risk in cancer patients remains driven by comorbidities.
It is likely, then, that personalized risk factors such as type of cancer therapy, site of disease, and comorbidities will shape individual choices about vaccination among cancer patients.
A version of this article first appeared on Medscape.com.