User login
Benefits of bremelanotide to women with HSDD questioned in analysis paper
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
Dr. Spielmans, professor of psychology at Metropolitan State University in Saint Paul, Minn., examined data from the FDA application for bremelanotide, clinicaltrials.gov entries for two phase 3 trials of the drug, and a 2019 article published in Obstetrics & Gynecology that described results from the 24-week trials.
In Dr. Speilman’s analysis, which was published online March 7 in the Journal of Sex Research, he notes that 42.1% of trial participants who received bremelanotide did not complete the trial, compared with 20.48% of participants who received placebo.
Of those who completed the study, 87.22% who received placebo wanted to continue treatment in an open-label extension, compared with 69.97% who received bremelanotide, he wrote.
Women “should be aware of the small degree of bremelanotide’s efficacy, that the protocol-specified outcomes of bremelanotide are mostly unknown, and that participants would rather take a placebo than bremelanotide,” Dr. Spielmans said.
Anita H. Clayton, MD, an author of the Obstetrics & Gynecology paper addressed in Dr. Spielmans’ analysis, says the Journal of Sex Research article does not provide new information and is a disservice to women because it questions accurate scientific data.
Measuring outcomes in HSDD is an evolving field, Dr. Clayton, a psychiatrist at the University of Virginia in Charlottesville, said in an interview. Initial FDA guidance relied on satisfying sexual events as an outcome measure, but this measure was derived from erectile dysfunction studies and is not necessarily adequate for assessing HSDD, she said. The FDA and drug developers agreed to use the desire subscale of the Female Sexual Function Index (FSFI-D) as a coprimary outcome measure instead, she noted.
Dr. Spielmans’ critique of Obstetrics & Gynecology paper
The article published in Obstetrics & Gynecology reporting bremelanotide trial results was noteworthy, although the various issues involved can be seen in reports about other drug trials, Dr. Spielmans said in an interview.
“It is well-established that journal articles reporting clinical trial data overstate benefits and understate harms,” he continued. In this case, “the very incomplete data reporting, reliance on many post-hoc measures of questionable validity, hiding the concerning number of dropouts due to adverse events, and putting a positive spin on efficacy and tolerability is both remarkable and highly problematic,” Dr. Spielmans said.
Dr. Clayton’s reaction
Data about dropout rates due to adverse events have been reported and presented at national meetings, she said in an interview. In addition, a questionnaire found that bremelanotide was superior to placebo in terms of patients feeling that the treatment had provided clinically meaningful benefit, Dr. Clayton said.
The available information enables patients to make informed treatment decisions, Dr. Clayton continued. “There is really this sexist attitude of women needing protection from their own decisions,” she said.
Diagnosing and treating HSDD
Eight of 11 efficacy outcomes in the clinicaltrials.gov study protocols for bremelanotide were not reported in the Obstetrics & Gynecology article in a way that was consistent with the protocols, Dr. Spielmans said. Changing a coprimary outcome to the key secondary outcome “occurred over a year after the trials had begun,” and the authors of the journal article “did not mention that this change occurred,” Dr. Spielmans wrote.
For the coprimary outcome measures of mean change on FSFI-D and Female Sexual Distress Scale–Desire/Arousal/Orgasm #13, “bremelanotide offers modest benefits over placebo,” Dr. Spielmans reported.
In addition to outlining his concerns about transparency in the reporting of trial data and raising questions about the outcome measures used in the Obstetrics & Gynecology article, Dr. Spielmans wrote that the diagnosis of HSDD is problematic.
“The lack of specifying symptom duration, questionable validity for the lack of sexual fantasies as a diagnostic criterion, difficulty in disentangling individual sexual problems from relational problems, and the failure to consider cultural influence (including the pressure on women to satisfy the sexual desires of their male partners) in the experience of sexuality all render HSDD as a problematic entity,” Dr. Spielmans wrote.
The fifth edition of the Diagnostic and Statistical Manual of Mental Disorders replaced HSDD and female sexual arousal disorder with the combined condition female sexual interest/arousal disorder. HSDD is in the 11th edition of the International Classification of Diseases and can be applied to men or women, Dr. Spielmans said.
FDA acknowledged HSDD as an unmet medical need
Dr. Clayton pointed out that HSDD was described decades ago and the FDA acknowledged it as an unmet medical need, and she expressed dissatisfaction with the fact the hypoactive sexual desire disorder appears with quotation marks around it in the title of Dr. Spielmans’ article. This way of presenting HSDD indicates that “the author has no concept of sexual health or sexual dysfunction,” Dr. Clayton said. “Basically this is sort of a dramatic tool, I think, to act like this is not a real disorder,” she added.
Carl Spana, PhD, CEO and president of Palatin Technologies, the developer of bremelanotide, defined the article in the Journal of Sex Research as a “retrospective meta-analysis, and not a re-analysis of the data.
“As a meta-analysis, it is open to various interpretations and reflects the author’s interpretations, which appear to have clear biases,” Dr. Spana said in an interview. “We believe several of this author’s interpretations are contrary to the FDA’s positive assessment that led to Vyleesi’s approval as a safe and effective treatment for women suffering from hypoactive sexual desire disorder.”
The author is unaware of the validation that was conducted at the direction of the FDA to establish clinically meaningful cutoffs for patient-reported outcomes and to establish metrics that define clinical benefit, Dr. Spana said
“Vyleesi was approved by the FDA after a thorough analysis of data from two well-controlled phase 3 clinical studies and multiple clinical and preclinical safety studies,” he said. “The analyses in the New Drug Application were prespecified and conducted according to a statistical analysis plan that the sponsor and FDA agreed to prior to database lock.”
Dr. Spielmans disclosed holdings in Vanguard Healthcare, a mutual fund that invests in pharmaceutical firms. Dr. Clayton has received financial support from Palatin and AMAG Pharmaceuticals, the companies that developed bremelanotide, in previous years.
FROM THE JOURNAL OF SEX RESEARCH
AAP report aims to educate providers on female genital mutilation/cutting
Although female genital mutilation or cutting (FGM/C) is outlawed in much of the world, it still occurs for cultural reasons despite having no medical benefit, according to a clinical report from the American Academy of Pediatrics.
FGM/C is mainly performed on children and adolescents, but most of the research and teaching to date has addressed the impact of FGM/C on women of childbearing age and management during pregnancy and post partum, wrote Janine Young, MD, of the University of Colorado Denver in Aurora and colleagues. They are members of the AAP section on global health, committee on medical liability and risk management, or the committee on bioethics.
Published in Pediatrics, the report provides “the first comprehensive summary of FGM/C in children and includes education regarding a standard-of-care approach for examination of external female genitalia at all health supervision examinations, diagnosis, complications, management, treatment, culturally sensitive discussion and counseling approaches, and legal and ethical considerations,” they wrote.
The World Health Organization categorizes FGM/C into four subtypes. “Type I includes cutting of the glans or part of the body of the clitoris and/or prepuce; type II includes excision of the clitoris and labia minora, with or without excision of the labia majora; type III, infibulation, includes cutting and apposing the labia minora and/or majora over the urethral meatus and vaginal opening to significantly narrow it and may include clitoral excision; and type IV includes piercing, scraping, nicking, stretching, or otherwise injuring the external female genitalia without removing any genital tissue and includes practices that do not fall into the other three categories,” the authors wrote. Of these, type III is associated with the greatest long-term morbidity.
Data suggest that the prevalence and type of FGM/C varies by region, with the highest prevalence of type III in East Africa, where 82%-99% of girls reported FGM/C and 34%-79% of these cases involved type III, the authors reported.
Generally, pediatric health care providers in the United States have limited knowledge of FGM/C in the absence of any required courses on diagnosis or treatment for most primary care specialties. However, clinicians should be aware of possible risk factors, including a mother or sibling with a history of FGM/C, or patients with a country of origin, birth country, or travel history to a country where FGM/C is practiced, Dr. Young and associates noted.
They recommend that but acknowledged the challenges in raising the topic and addressing it in a culturally sensitive way. “Experts suggest that health care providers ask the patient or parent the term they use to name female genital cutting” and avoid the term mutilation, which may be offensive or misunderstood.
Many girls who have undergone FGM/C were too young to remember, the authors note. “Instead, it is advisable that the FGM/C clinical history taking include both the girl and parent or guardian once rapport has been established.”
Review potential medical complications if FGM/C is identified, and plans should be made for follow-up visits to monitor development of complications, the authors said. In addition, engage in a culturally sensitive discussion with teenagers, who may or may not have known about their FGM/C. In some cases, parents and caregivers may not have known about the FGM/C, which may be a community practice in some cultures with decisions made by other family members or authority figures.
“It is important for health care providers to assess each patient individually and make no assumptions about her and her parents’ beliefs regarding FGM/C,” Dr. Young and associates emphasized. “Mothers and fathers may or may not hold discordant views about FGM/C, and some clinical experts suggest that mothers who have themselves undergone FGM/C may nonetheless oppose subjecting their daughters to this practice. Instead, treating patients and caregivers with respect, sensitivity, and professionalism will encourage them to return and supports health-seeking behavior.”
The report presents 11 specific recommendations, including that health care providers should not perform any type of FGM/C and actively counsel families against such practices. In addition, children should have external genitalia checked at all health supervision examinations (with the consent of the guardian and/or child), and an assessment for FGM/C should be documented in the health records of patients with risk factors.
Notably, “[i]f genital examination findings are equivocal for the presence of FGM/C and risk factors for FGM/ C are present, a specialist trained in identification of FGM/C should be consulted,” Dr. Young and associates recommended. They also recommended defibulation for all girls and teenagers with type III FGM/C, especially for those with complications, and the procedure should be performed by an experienced pediatric gynecologist, gynecologist, urologist, or urogynecologist.
Finally, “[i]f FGM/C is suspected to have occurred in the United States, or as vacation cutting after immigration to the United States, the child should be evaluated for potential abuse. ... Expressed intention to engage in FGM/C, either in the United States or abroad, should also prompt a report to CPS [child protective services] if the child’s parent or caregiver cannot be dissuaded,” the authors wrote.
The report also includes case examples and expert analyses from legal and medical ethics experts to provide additional guidance for clinicians.
“This work seeks to educate pediatric health care providers on the occurrence of FGM/C, and the broader applications to the patients/population it impacts as well as the intersecting issues of diagnosis, complications, treatment, counseling needs, and the ethical and legal implications,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
However, challenges in implementing the recommendations “relate to the complexity of the issue and also the need for greater education of primary providers,” Dr. Jay said. “The overall message for providers, I believe, is a greater understanding of the practice [of FGM/C] as most providers have limited knowledge of this practice in the United States.”
“I believe the case-based presentations allow for a better understanding of how best to approach patients and families,” she added.
Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said, “I think one of largest barriers to implementing the strategies [from] this report is the limited knowledge of FGM/C by most clinicians.”
“In general, many pediatricians are uncomfortable with genital examinations,” she said in an interview. “I suspect most feel uncomfortable with identifying FGM/C versus other genital pathology and may not have ready access to FGM/C experts. Additionally, having these difficult conversations with families about this sensitive topic may be challenging,” said Dr. Curran. “Fortunately, this report is incredibly comprehensive, providing extensive background into FGM/C, effectively using diagrams and pictures, and explaining the legal and ethical issues that arise in the care of these patients.”
“Ultimately, I think there will need to be more education within medical training and further research into FGM/C,” Dr. Curran added. “Clinicians should be knowledgeable about FGM/C, including prevalence, identification, health complications, and treatment, as well as legal and ethical implications.” However, “in addition to knowledge, clinicians must be able to navigate counseling patients and their families around this culturally sensitive topic.”
The report is thorough and well written, yet “there still remains significant gaps in knowledge about FGM/C in children and adolescents,” she said. “I think future research into prevalence, along with the health effects of FGM/C, including its impact on mental and sexual health, in the pediatric population will be essential.”
The study received no outside funding. Coauthor Christa Johnson-Agbakwu, MD, disclosed a grant relationship with Arizona State University from the 2018 copyright of “Female Genital Mutilation/Cutting (FGM/C): A Visual Reference and Learning Tool for Health Care Professionals.” The other researchers had no financial conflicts to disclose. Dr. Jay and Dr. Curran had no relevant financial conflicts to disclose. They are members of the Pediatric News editorial advisory board.
SOURCE: Young J et al. Pediatrics. 2020 Jul 27. doi: 10.1542/peds.2020-1012.
Although female genital mutilation or cutting (FGM/C) is outlawed in much of the world, it still occurs for cultural reasons despite having no medical benefit, according to a clinical report from the American Academy of Pediatrics.
FGM/C is mainly performed on children and adolescents, but most of the research and teaching to date has addressed the impact of FGM/C on women of childbearing age and management during pregnancy and post partum, wrote Janine Young, MD, of the University of Colorado Denver in Aurora and colleagues. They are members of the AAP section on global health, committee on medical liability and risk management, or the committee on bioethics.
Published in Pediatrics, the report provides “the first comprehensive summary of FGM/C in children and includes education regarding a standard-of-care approach for examination of external female genitalia at all health supervision examinations, diagnosis, complications, management, treatment, culturally sensitive discussion and counseling approaches, and legal and ethical considerations,” they wrote.
The World Health Organization categorizes FGM/C into four subtypes. “Type I includes cutting of the glans or part of the body of the clitoris and/or prepuce; type II includes excision of the clitoris and labia minora, with or without excision of the labia majora; type III, infibulation, includes cutting and apposing the labia minora and/or majora over the urethral meatus and vaginal opening to significantly narrow it and may include clitoral excision; and type IV includes piercing, scraping, nicking, stretching, or otherwise injuring the external female genitalia without removing any genital tissue and includes practices that do not fall into the other three categories,” the authors wrote. Of these, type III is associated with the greatest long-term morbidity.
Data suggest that the prevalence and type of FGM/C varies by region, with the highest prevalence of type III in East Africa, where 82%-99% of girls reported FGM/C and 34%-79% of these cases involved type III, the authors reported.
Generally, pediatric health care providers in the United States have limited knowledge of FGM/C in the absence of any required courses on diagnosis or treatment for most primary care specialties. However, clinicians should be aware of possible risk factors, including a mother or sibling with a history of FGM/C, or patients with a country of origin, birth country, or travel history to a country where FGM/C is practiced, Dr. Young and associates noted.
They recommend that but acknowledged the challenges in raising the topic and addressing it in a culturally sensitive way. “Experts suggest that health care providers ask the patient or parent the term they use to name female genital cutting” and avoid the term mutilation, which may be offensive or misunderstood.
Many girls who have undergone FGM/C were too young to remember, the authors note. “Instead, it is advisable that the FGM/C clinical history taking include both the girl and parent or guardian once rapport has been established.”
Review potential medical complications if FGM/C is identified, and plans should be made for follow-up visits to monitor development of complications, the authors said. In addition, engage in a culturally sensitive discussion with teenagers, who may or may not have known about their FGM/C. In some cases, parents and caregivers may not have known about the FGM/C, which may be a community practice in some cultures with decisions made by other family members or authority figures.
“It is important for health care providers to assess each patient individually and make no assumptions about her and her parents’ beliefs regarding FGM/C,” Dr. Young and associates emphasized. “Mothers and fathers may or may not hold discordant views about FGM/C, and some clinical experts suggest that mothers who have themselves undergone FGM/C may nonetheless oppose subjecting their daughters to this practice. Instead, treating patients and caregivers with respect, sensitivity, and professionalism will encourage them to return and supports health-seeking behavior.”
The report presents 11 specific recommendations, including that health care providers should not perform any type of FGM/C and actively counsel families against such practices. In addition, children should have external genitalia checked at all health supervision examinations (with the consent of the guardian and/or child), and an assessment for FGM/C should be documented in the health records of patients with risk factors.
Notably, “[i]f genital examination findings are equivocal for the presence of FGM/C and risk factors for FGM/ C are present, a specialist trained in identification of FGM/C should be consulted,” Dr. Young and associates recommended. They also recommended defibulation for all girls and teenagers with type III FGM/C, especially for those with complications, and the procedure should be performed by an experienced pediatric gynecologist, gynecologist, urologist, or urogynecologist.
Finally, “[i]f FGM/C is suspected to have occurred in the United States, or as vacation cutting after immigration to the United States, the child should be evaluated for potential abuse. ... Expressed intention to engage in FGM/C, either in the United States or abroad, should also prompt a report to CPS [child protective services] if the child’s parent or caregiver cannot be dissuaded,” the authors wrote.
The report also includes case examples and expert analyses from legal and medical ethics experts to provide additional guidance for clinicians.
“This work seeks to educate pediatric health care providers on the occurrence of FGM/C, and the broader applications to the patients/population it impacts as well as the intersecting issues of diagnosis, complications, treatment, counseling needs, and the ethical and legal implications,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
However, challenges in implementing the recommendations “relate to the complexity of the issue and also the need for greater education of primary providers,” Dr. Jay said. “The overall message for providers, I believe, is a greater understanding of the practice [of FGM/C] as most providers have limited knowledge of this practice in the United States.”
“I believe the case-based presentations allow for a better understanding of how best to approach patients and families,” she added.
Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said, “I think one of largest barriers to implementing the strategies [from] this report is the limited knowledge of FGM/C by most clinicians.”
“In general, many pediatricians are uncomfortable with genital examinations,” she said in an interview. “I suspect most feel uncomfortable with identifying FGM/C versus other genital pathology and may not have ready access to FGM/C experts. Additionally, having these difficult conversations with families about this sensitive topic may be challenging,” said Dr. Curran. “Fortunately, this report is incredibly comprehensive, providing extensive background into FGM/C, effectively using diagrams and pictures, and explaining the legal and ethical issues that arise in the care of these patients.”
“Ultimately, I think there will need to be more education within medical training and further research into FGM/C,” Dr. Curran added. “Clinicians should be knowledgeable about FGM/C, including prevalence, identification, health complications, and treatment, as well as legal and ethical implications.” However, “in addition to knowledge, clinicians must be able to navigate counseling patients and their families around this culturally sensitive topic.”
The report is thorough and well written, yet “there still remains significant gaps in knowledge about FGM/C in children and adolescents,” she said. “I think future research into prevalence, along with the health effects of FGM/C, including its impact on mental and sexual health, in the pediatric population will be essential.”
The study received no outside funding. Coauthor Christa Johnson-Agbakwu, MD, disclosed a grant relationship with Arizona State University from the 2018 copyright of “Female Genital Mutilation/Cutting (FGM/C): A Visual Reference and Learning Tool for Health Care Professionals.” The other researchers had no financial conflicts to disclose. Dr. Jay and Dr. Curran had no relevant financial conflicts to disclose. They are members of the Pediatric News editorial advisory board.
SOURCE: Young J et al. Pediatrics. 2020 Jul 27. doi: 10.1542/peds.2020-1012.
Although female genital mutilation or cutting (FGM/C) is outlawed in much of the world, it still occurs for cultural reasons despite having no medical benefit, according to a clinical report from the American Academy of Pediatrics.
FGM/C is mainly performed on children and adolescents, but most of the research and teaching to date has addressed the impact of FGM/C on women of childbearing age and management during pregnancy and post partum, wrote Janine Young, MD, of the University of Colorado Denver in Aurora and colleagues. They are members of the AAP section on global health, committee on medical liability and risk management, or the committee on bioethics.
Published in Pediatrics, the report provides “the first comprehensive summary of FGM/C in children and includes education regarding a standard-of-care approach for examination of external female genitalia at all health supervision examinations, diagnosis, complications, management, treatment, culturally sensitive discussion and counseling approaches, and legal and ethical considerations,” they wrote.
The World Health Organization categorizes FGM/C into four subtypes. “Type I includes cutting of the glans or part of the body of the clitoris and/or prepuce; type II includes excision of the clitoris and labia minora, with or without excision of the labia majora; type III, infibulation, includes cutting and apposing the labia minora and/or majora over the urethral meatus and vaginal opening to significantly narrow it and may include clitoral excision; and type IV includes piercing, scraping, nicking, stretching, or otherwise injuring the external female genitalia without removing any genital tissue and includes practices that do not fall into the other three categories,” the authors wrote. Of these, type III is associated with the greatest long-term morbidity.
Data suggest that the prevalence and type of FGM/C varies by region, with the highest prevalence of type III in East Africa, where 82%-99% of girls reported FGM/C and 34%-79% of these cases involved type III, the authors reported.
Generally, pediatric health care providers in the United States have limited knowledge of FGM/C in the absence of any required courses on diagnosis or treatment for most primary care specialties. However, clinicians should be aware of possible risk factors, including a mother or sibling with a history of FGM/C, or patients with a country of origin, birth country, or travel history to a country where FGM/C is practiced, Dr. Young and associates noted.
They recommend that but acknowledged the challenges in raising the topic and addressing it in a culturally sensitive way. “Experts suggest that health care providers ask the patient or parent the term they use to name female genital cutting” and avoid the term mutilation, which may be offensive or misunderstood.
Many girls who have undergone FGM/C were too young to remember, the authors note. “Instead, it is advisable that the FGM/C clinical history taking include both the girl and parent or guardian once rapport has been established.”
Review potential medical complications if FGM/C is identified, and plans should be made for follow-up visits to monitor development of complications, the authors said. In addition, engage in a culturally sensitive discussion with teenagers, who may or may not have known about their FGM/C. In some cases, parents and caregivers may not have known about the FGM/C, which may be a community practice in some cultures with decisions made by other family members or authority figures.
“It is important for health care providers to assess each patient individually and make no assumptions about her and her parents’ beliefs regarding FGM/C,” Dr. Young and associates emphasized. “Mothers and fathers may or may not hold discordant views about FGM/C, and some clinical experts suggest that mothers who have themselves undergone FGM/C may nonetheless oppose subjecting their daughters to this practice. Instead, treating patients and caregivers with respect, sensitivity, and professionalism will encourage them to return and supports health-seeking behavior.”
The report presents 11 specific recommendations, including that health care providers should not perform any type of FGM/C and actively counsel families against such practices. In addition, children should have external genitalia checked at all health supervision examinations (with the consent of the guardian and/or child), and an assessment for FGM/C should be documented in the health records of patients with risk factors.
Notably, “[i]f genital examination findings are equivocal for the presence of FGM/C and risk factors for FGM/ C are present, a specialist trained in identification of FGM/C should be consulted,” Dr. Young and associates recommended. They also recommended defibulation for all girls and teenagers with type III FGM/C, especially for those with complications, and the procedure should be performed by an experienced pediatric gynecologist, gynecologist, urologist, or urogynecologist.
Finally, “[i]f FGM/C is suspected to have occurred in the United States, or as vacation cutting after immigration to the United States, the child should be evaluated for potential abuse. ... Expressed intention to engage in FGM/C, either in the United States or abroad, should also prompt a report to CPS [child protective services] if the child’s parent or caregiver cannot be dissuaded,” the authors wrote.
The report also includes case examples and expert analyses from legal and medical ethics experts to provide additional guidance for clinicians.
“This work seeks to educate pediatric health care providers on the occurrence of FGM/C, and the broader applications to the patients/population it impacts as well as the intersecting issues of diagnosis, complications, treatment, counseling needs, and the ethical and legal implications,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
However, challenges in implementing the recommendations “relate to the complexity of the issue and also the need for greater education of primary providers,” Dr. Jay said. “The overall message for providers, I believe, is a greater understanding of the practice [of FGM/C] as most providers have limited knowledge of this practice in the United States.”
“I believe the case-based presentations allow for a better understanding of how best to approach patients and families,” she added.
Kelly Curran, MD, of the University of Oklahoma Health Sciences Center, Oklahoma City, said, “I think one of largest barriers to implementing the strategies [from] this report is the limited knowledge of FGM/C by most clinicians.”
“In general, many pediatricians are uncomfortable with genital examinations,” she said in an interview. “I suspect most feel uncomfortable with identifying FGM/C versus other genital pathology and may not have ready access to FGM/C experts. Additionally, having these difficult conversations with families about this sensitive topic may be challenging,” said Dr. Curran. “Fortunately, this report is incredibly comprehensive, providing extensive background into FGM/C, effectively using diagrams and pictures, and explaining the legal and ethical issues that arise in the care of these patients.”
“Ultimately, I think there will need to be more education within medical training and further research into FGM/C,” Dr. Curran added. “Clinicians should be knowledgeable about FGM/C, including prevalence, identification, health complications, and treatment, as well as legal and ethical implications.” However, “in addition to knowledge, clinicians must be able to navigate counseling patients and their families around this culturally sensitive topic.”
The report is thorough and well written, yet “there still remains significant gaps in knowledge about FGM/C in children and adolescents,” she said. “I think future research into prevalence, along with the health effects of FGM/C, including its impact on mental and sexual health, in the pediatric population will be essential.”
The study received no outside funding. Coauthor Christa Johnson-Agbakwu, MD, disclosed a grant relationship with Arizona State University from the 2018 copyright of “Female Genital Mutilation/Cutting (FGM/C): A Visual Reference and Learning Tool for Health Care Professionals.” The other researchers had no financial conflicts to disclose. Dr. Jay and Dr. Curran had no relevant financial conflicts to disclose. They are members of the Pediatric News editorial advisory board.
SOURCE: Young J et al. Pediatrics. 2020 Jul 27. doi: 10.1542/peds.2020-1012.
FROM PEDIATRICS
Does BSO status affect health outcomes for women taking estrogen for menopause?
Do health effects of menopausal estrogen therapy differ between women with bilateral oophorectomy versus those with conserved ovaries? To answer this question a group of investigators performed a subanalysis of the Women’s Health Initiative (WHI) Estrogen-Alone Trial,1 which included 40 clinical centers across the United States. They examined estrogen therapy outcomes by bilateral salpingo-oophorectomy (BSO) status, with additional stratification by 10-year age groups in 9,939 women aged 50 to 79 years with prior hysterectomy and known oophorectomy status. In the WHI trial, women were randomly assigned to conjugated equine estrogens (CEE) 0.625 mg/d or placebo for a median of 7.2 years. Investigators assessed the incidence of coronary heart disease and invasive breast cancer (the trial’s 2 primary end points), all-cause mortality, and a “global index”—these end points plus stroke, pulmonary embolism, colorectal cancer, and hip fracture—during the intervention phase and 18-year cumulative follow-up.
OBG Management caught up with lead author JoAnn E. Manson, MD, DrPH, NCMP, to discuss the study’s results.
OBG Management : How many women undergo BSO with their hysterectomy?
Dr. JoAnn E. Manson, MD, DrPH, NCMP: Of the 425,000 women who undergo hysterectomy in the United States for benign reasons each year,2,3 about 40% of them undergo BSO—so between 150,000 and 200,000 women per year undergo BSO with their hysterectomy.4,5
OBG Management : Although BSO is performed with hysterectomy to minimize patients’ future ovarian cancer risk, does BSO have health risks of its own, and how has estrogen been shown to affect these risks?
Dr. Manson: First, yes, BSO has been associated with health risks, especially when it is performed at a young age, such as before age 45. It has been linked to an increased risk of heart disease, osteoporosis, cognitive decline, and all-cause mortality. According to observational studies, estrogen therapy appears to offset many of these risks, particularly those related to heart disease and osteoporosis (the evidence is less clear on cognitive deficits).5
OBG Management : What did you find in your trial when you randomly assigned women in the age groups of 50 to 79 who underwent hysterectomy with and without BSO to estrogen therapy or placebo?
Dr. Manson: The WHI is the first study to be conducted in a randomized trial setting to analyze the health risks and benefits of estrogen therapy according to whether or not women had their ovaries removed. What we found was that the woman’s age had a strong influence on the effects of estrogen therapy among women who had BSO but only a negligible effect among women who had conserved ovaries. Overall, across the full age range, the effects of estrogen therapy did not differ substantially between women who had a BSO and those who had their ovaries conserved.
However, there were major differences by age group among the women who had BSO. A significant 32% reduction in all-cause mortality emerged during the 18-year follow-up period among the younger women (below age 60) who had BSO when they received estrogen therapy as compared with placebo. By contrast, the women who had conserved ovaries did not have this significant reduction in all-cause mortality, or in most of the other outcomes on estrogen compared with placebo. Overall, the effects of estrogen therapy tended to be relatively neutral in the women with conserved ovaries.
Now, the reduction in all-cause mortality with estrogen therapy was particularly pronounced among women who had BSO before age 45. They had a 40% statistically significant reduction in all-cause mortality with estrogen therapy compared with placebo. Also, among the women with BSO, there was a strong association between the timing of estrogen initiation and the magnitude of reduction in mortality. Women who started the estrogen therapy within 10 years of having the BSO had a 34% significant reduction in all-cause mortality, and those who started estrogen more than 20 years after having their ovaries removed had no reduction in mortality.
Continue to:
OBG Management : Do your data give support to the timing hypothesis?
Dr. Manson: Yes, our findings do support a timing hypothesis that was particularly pronounced for women who underwent BSO. It was the women who had early surgical menopause (before age 45) and those who started the estrogen therapy within 10 years of having their ovaries removed who had the greatest reduction in all-cause mortality and the most favorable benefit-risk profile from hormone therapy. So, the results do lend support to the timing hypothesis.
By contrast, women who had BSO at hysterectomy and began hormone therapy at age 70 or older had net adverse effects from hormone therapy. They posted a 40% increase in the global index—which is a summary measure of adverse effects on cardiovascular disease, cancer, and other major health outcomes. So, the women with BSO who were randomized in the trial at age 70 and older, had unfavorable results from estrogen therapy and an increase in the global index, in contrast to the women who were below age 60 or within 10 years of menopause.
OBG Management : Given your study findings, in which women would you recommend estrogen therapy? And are there groups of women in which you would advise avoiding estrogen therapy?
Dr. Manson: Current guidelines6,7 recommend estrogen therapy for women who have early menopause, particularly an early surgical menopause and BSO prior to the average age at natural menopause. Unless the woman has contraindications to estrogen therapy, the recommendations are to treat with estrogen until the average age of menopause—until about age 50 to 51.
Our study findings provide reassurance that, if a woman continues to have indications for estrogen (vasomotor symptoms, or other indications for estrogen therapy), there is relative safety of continuing estrogen-alone therapy through her 50s, until age 60. For example, a woman who, after the average age of menopause continues to have vasomotor symptoms, or if she has bone health problems, our study would suggest that estrogen therapy would continue to have a favorable benefit-risk profile until at least the age of 60. Decisions would have to be individualized, especially after age 60, with shared decision-making particularly important for those decisions. (Some women, depending on their risk profile, may continue to be candidates for estrogen therapy past age 60.)
So, this study provides reassurance regarding use of estrogen therapy for women in their 50s if they have had BSO. Actually, the women who had conserved ovaries also had relative safety with estrogen therapy until age 60. They just didn’t show the significant benefits for all-cause mortality. Overall, their pattern of health-related benefits and risks was neutral. Thus, if vasomotor symptom management, quality of life benefits, or bone health effects are sought, taking hormone therapy is a quite reasonable choice for these women.
By contrast, women who have had a BSO and are age 70 or older should really avoid initiating estrogen therapy because it would follow a prolonged period of estrogen deficiency, or very low estrogen levels, and these women appeared to have a net adverse effect from initiating hormone therapy (with increases in the global index found).
Continue to:
OBG Management : Did taking estrogen therapy prior to trial enrollment make a difference when it came to study outcomes?
Dr. Manson: We found minimal if any effect in our analyses. In fact, even the women who did not have prior (pre-randomization) use of estrogen therapy tended to do well on estrogen-alone therapy if they were younger than age 60. This was particularly true for the women who had BSO. Even if they had not used estrogen previously, and they were many years past the BSO, they still did well on estrogen therapy if they were below age 60.
1. Manson JE, Aragaki AK, Bassuk SS. Menopausal estrogen-alone therapy and health outcomes in women with and without bilateral oophorectomy: a randomized trial. Ann Intern Med. 2019 September 10. doi:10.7326/M19-0274.
2. Einarsson J. Are hysterectomy volumes in the US really falling? Contemporary OB/GYN. 1 September 2017. www.contemporaryobgyn.net/gynecology/are-hysterectomy-volumes-us-really-falling. November 4, 2019.
3. Temkin SM, Minasian L, Noone AM. The end of the hysterectomy epidemic and endometrial cancer incidence: what are the unintended consequences of declining hysterectomy rates? Front Oncol. 2016;6:89.
4. Doll KM, Dusetzina SB, Robinson W. Trends in inpatient and outpatient hysterectomy and oophorectomy rates among commercially insured women in the United States, 2000-2014. JAMA Surg. 2016;151:876-877.
5. Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279.
6. ACOG Practice Bulletin No. 141: management of menopausal symptoms [published corrections appear in: Obstet Gynecol. 2016;127(1):166. and Obstet Gynecol. 2018;131(3):604]. Obstet Gynecol. 2014;123:202-216.
7. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753.
Do health effects of menopausal estrogen therapy differ between women with bilateral oophorectomy versus those with conserved ovaries? To answer this question a group of investigators performed a subanalysis of the Women’s Health Initiative (WHI) Estrogen-Alone Trial,1 which included 40 clinical centers across the United States. They examined estrogen therapy outcomes by bilateral salpingo-oophorectomy (BSO) status, with additional stratification by 10-year age groups in 9,939 women aged 50 to 79 years with prior hysterectomy and known oophorectomy status. In the WHI trial, women were randomly assigned to conjugated equine estrogens (CEE) 0.625 mg/d or placebo for a median of 7.2 years. Investigators assessed the incidence of coronary heart disease and invasive breast cancer (the trial’s 2 primary end points), all-cause mortality, and a “global index”—these end points plus stroke, pulmonary embolism, colorectal cancer, and hip fracture—during the intervention phase and 18-year cumulative follow-up.
OBG Management caught up with lead author JoAnn E. Manson, MD, DrPH, NCMP, to discuss the study’s results.
OBG Management : How many women undergo BSO with their hysterectomy?
Dr. JoAnn E. Manson, MD, DrPH, NCMP: Of the 425,000 women who undergo hysterectomy in the United States for benign reasons each year,2,3 about 40% of them undergo BSO—so between 150,000 and 200,000 women per year undergo BSO with their hysterectomy.4,5
OBG Management : Although BSO is performed with hysterectomy to minimize patients’ future ovarian cancer risk, does BSO have health risks of its own, and how has estrogen been shown to affect these risks?
Dr. Manson: First, yes, BSO has been associated with health risks, especially when it is performed at a young age, such as before age 45. It has been linked to an increased risk of heart disease, osteoporosis, cognitive decline, and all-cause mortality. According to observational studies, estrogen therapy appears to offset many of these risks, particularly those related to heart disease and osteoporosis (the evidence is less clear on cognitive deficits).5
OBG Management : What did you find in your trial when you randomly assigned women in the age groups of 50 to 79 who underwent hysterectomy with and without BSO to estrogen therapy or placebo?
Dr. Manson: The WHI is the first study to be conducted in a randomized trial setting to analyze the health risks and benefits of estrogen therapy according to whether or not women had their ovaries removed. What we found was that the woman’s age had a strong influence on the effects of estrogen therapy among women who had BSO but only a negligible effect among women who had conserved ovaries. Overall, across the full age range, the effects of estrogen therapy did not differ substantially between women who had a BSO and those who had their ovaries conserved.
However, there were major differences by age group among the women who had BSO. A significant 32% reduction in all-cause mortality emerged during the 18-year follow-up period among the younger women (below age 60) who had BSO when they received estrogen therapy as compared with placebo. By contrast, the women who had conserved ovaries did not have this significant reduction in all-cause mortality, or in most of the other outcomes on estrogen compared with placebo. Overall, the effects of estrogen therapy tended to be relatively neutral in the women with conserved ovaries.
Now, the reduction in all-cause mortality with estrogen therapy was particularly pronounced among women who had BSO before age 45. They had a 40% statistically significant reduction in all-cause mortality with estrogen therapy compared with placebo. Also, among the women with BSO, there was a strong association between the timing of estrogen initiation and the magnitude of reduction in mortality. Women who started the estrogen therapy within 10 years of having the BSO had a 34% significant reduction in all-cause mortality, and those who started estrogen more than 20 years after having their ovaries removed had no reduction in mortality.
Continue to:
OBG Management : Do your data give support to the timing hypothesis?
Dr. Manson: Yes, our findings do support a timing hypothesis that was particularly pronounced for women who underwent BSO. It was the women who had early surgical menopause (before age 45) and those who started the estrogen therapy within 10 years of having their ovaries removed who had the greatest reduction in all-cause mortality and the most favorable benefit-risk profile from hormone therapy. So, the results do lend support to the timing hypothesis.
By contrast, women who had BSO at hysterectomy and began hormone therapy at age 70 or older had net adverse effects from hormone therapy. They posted a 40% increase in the global index—which is a summary measure of adverse effects on cardiovascular disease, cancer, and other major health outcomes. So, the women with BSO who were randomized in the trial at age 70 and older, had unfavorable results from estrogen therapy and an increase in the global index, in contrast to the women who were below age 60 or within 10 years of menopause.
OBG Management : Given your study findings, in which women would you recommend estrogen therapy? And are there groups of women in which you would advise avoiding estrogen therapy?
Dr. Manson: Current guidelines6,7 recommend estrogen therapy for women who have early menopause, particularly an early surgical menopause and BSO prior to the average age at natural menopause. Unless the woman has contraindications to estrogen therapy, the recommendations are to treat with estrogen until the average age of menopause—until about age 50 to 51.
Our study findings provide reassurance that, if a woman continues to have indications for estrogen (vasomotor symptoms, or other indications for estrogen therapy), there is relative safety of continuing estrogen-alone therapy through her 50s, until age 60. For example, a woman who, after the average age of menopause continues to have vasomotor symptoms, or if she has bone health problems, our study would suggest that estrogen therapy would continue to have a favorable benefit-risk profile until at least the age of 60. Decisions would have to be individualized, especially after age 60, with shared decision-making particularly important for those decisions. (Some women, depending on their risk profile, may continue to be candidates for estrogen therapy past age 60.)
So, this study provides reassurance regarding use of estrogen therapy for women in their 50s if they have had BSO. Actually, the women who had conserved ovaries also had relative safety with estrogen therapy until age 60. They just didn’t show the significant benefits for all-cause mortality. Overall, their pattern of health-related benefits and risks was neutral. Thus, if vasomotor symptom management, quality of life benefits, or bone health effects are sought, taking hormone therapy is a quite reasonable choice for these women.
By contrast, women who have had a BSO and are age 70 or older should really avoid initiating estrogen therapy because it would follow a prolonged period of estrogen deficiency, or very low estrogen levels, and these women appeared to have a net adverse effect from initiating hormone therapy (with increases in the global index found).
Continue to:
OBG Management : Did taking estrogen therapy prior to trial enrollment make a difference when it came to study outcomes?
Dr. Manson: We found minimal if any effect in our analyses. In fact, even the women who did not have prior (pre-randomization) use of estrogen therapy tended to do well on estrogen-alone therapy if they were younger than age 60. This was particularly true for the women who had BSO. Even if they had not used estrogen previously, and they were many years past the BSO, they still did well on estrogen therapy if they were below age 60.
Do health effects of menopausal estrogen therapy differ between women with bilateral oophorectomy versus those with conserved ovaries? To answer this question a group of investigators performed a subanalysis of the Women’s Health Initiative (WHI) Estrogen-Alone Trial,1 which included 40 clinical centers across the United States. They examined estrogen therapy outcomes by bilateral salpingo-oophorectomy (BSO) status, with additional stratification by 10-year age groups in 9,939 women aged 50 to 79 years with prior hysterectomy and known oophorectomy status. In the WHI trial, women were randomly assigned to conjugated equine estrogens (CEE) 0.625 mg/d or placebo for a median of 7.2 years. Investigators assessed the incidence of coronary heart disease and invasive breast cancer (the trial’s 2 primary end points), all-cause mortality, and a “global index”—these end points plus stroke, pulmonary embolism, colorectal cancer, and hip fracture—during the intervention phase and 18-year cumulative follow-up.
OBG Management caught up with lead author JoAnn E. Manson, MD, DrPH, NCMP, to discuss the study’s results.
OBG Management : How many women undergo BSO with their hysterectomy?
Dr. JoAnn E. Manson, MD, DrPH, NCMP: Of the 425,000 women who undergo hysterectomy in the United States for benign reasons each year,2,3 about 40% of them undergo BSO—so between 150,000 and 200,000 women per year undergo BSO with their hysterectomy.4,5
OBG Management : Although BSO is performed with hysterectomy to minimize patients’ future ovarian cancer risk, does BSO have health risks of its own, and how has estrogen been shown to affect these risks?
Dr. Manson: First, yes, BSO has been associated with health risks, especially when it is performed at a young age, such as before age 45. It has been linked to an increased risk of heart disease, osteoporosis, cognitive decline, and all-cause mortality. According to observational studies, estrogen therapy appears to offset many of these risks, particularly those related to heart disease and osteoporosis (the evidence is less clear on cognitive deficits).5
OBG Management : What did you find in your trial when you randomly assigned women in the age groups of 50 to 79 who underwent hysterectomy with and without BSO to estrogen therapy or placebo?
Dr. Manson: The WHI is the first study to be conducted in a randomized trial setting to analyze the health risks and benefits of estrogen therapy according to whether or not women had their ovaries removed. What we found was that the woman’s age had a strong influence on the effects of estrogen therapy among women who had BSO but only a negligible effect among women who had conserved ovaries. Overall, across the full age range, the effects of estrogen therapy did not differ substantially between women who had a BSO and those who had their ovaries conserved.
However, there were major differences by age group among the women who had BSO. A significant 32% reduction in all-cause mortality emerged during the 18-year follow-up period among the younger women (below age 60) who had BSO when they received estrogen therapy as compared with placebo. By contrast, the women who had conserved ovaries did not have this significant reduction in all-cause mortality, or in most of the other outcomes on estrogen compared with placebo. Overall, the effects of estrogen therapy tended to be relatively neutral in the women with conserved ovaries.
Now, the reduction in all-cause mortality with estrogen therapy was particularly pronounced among women who had BSO before age 45. They had a 40% statistically significant reduction in all-cause mortality with estrogen therapy compared with placebo. Also, among the women with BSO, there was a strong association between the timing of estrogen initiation and the magnitude of reduction in mortality. Women who started the estrogen therapy within 10 years of having the BSO had a 34% significant reduction in all-cause mortality, and those who started estrogen more than 20 years after having their ovaries removed had no reduction in mortality.
Continue to:
OBG Management : Do your data give support to the timing hypothesis?
Dr. Manson: Yes, our findings do support a timing hypothesis that was particularly pronounced for women who underwent BSO. It was the women who had early surgical menopause (before age 45) and those who started the estrogen therapy within 10 years of having their ovaries removed who had the greatest reduction in all-cause mortality and the most favorable benefit-risk profile from hormone therapy. So, the results do lend support to the timing hypothesis.
By contrast, women who had BSO at hysterectomy and began hormone therapy at age 70 or older had net adverse effects from hormone therapy. They posted a 40% increase in the global index—which is a summary measure of adverse effects on cardiovascular disease, cancer, and other major health outcomes. So, the women with BSO who were randomized in the trial at age 70 and older, had unfavorable results from estrogen therapy and an increase in the global index, in contrast to the women who were below age 60 or within 10 years of menopause.
OBG Management : Given your study findings, in which women would you recommend estrogen therapy? And are there groups of women in which you would advise avoiding estrogen therapy?
Dr. Manson: Current guidelines6,7 recommend estrogen therapy for women who have early menopause, particularly an early surgical menopause and BSO prior to the average age at natural menopause. Unless the woman has contraindications to estrogen therapy, the recommendations are to treat with estrogen until the average age of menopause—until about age 50 to 51.
Our study findings provide reassurance that, if a woman continues to have indications for estrogen (vasomotor symptoms, or other indications for estrogen therapy), there is relative safety of continuing estrogen-alone therapy through her 50s, until age 60. For example, a woman who, after the average age of menopause continues to have vasomotor symptoms, or if she has bone health problems, our study would suggest that estrogen therapy would continue to have a favorable benefit-risk profile until at least the age of 60. Decisions would have to be individualized, especially after age 60, with shared decision-making particularly important for those decisions. (Some women, depending on their risk profile, may continue to be candidates for estrogen therapy past age 60.)
So, this study provides reassurance regarding use of estrogen therapy for women in their 50s if they have had BSO. Actually, the women who had conserved ovaries also had relative safety with estrogen therapy until age 60. They just didn’t show the significant benefits for all-cause mortality. Overall, their pattern of health-related benefits and risks was neutral. Thus, if vasomotor symptom management, quality of life benefits, or bone health effects are sought, taking hormone therapy is a quite reasonable choice for these women.
By contrast, women who have had a BSO and are age 70 or older should really avoid initiating estrogen therapy because it would follow a prolonged period of estrogen deficiency, or very low estrogen levels, and these women appeared to have a net adverse effect from initiating hormone therapy (with increases in the global index found).
Continue to:
OBG Management : Did taking estrogen therapy prior to trial enrollment make a difference when it came to study outcomes?
Dr. Manson: We found minimal if any effect in our analyses. In fact, even the women who did not have prior (pre-randomization) use of estrogen therapy tended to do well on estrogen-alone therapy if they were younger than age 60. This was particularly true for the women who had BSO. Even if they had not used estrogen previously, and they were many years past the BSO, they still did well on estrogen therapy if they were below age 60.
1. Manson JE, Aragaki AK, Bassuk SS. Menopausal estrogen-alone therapy and health outcomes in women with and without bilateral oophorectomy: a randomized trial. Ann Intern Med. 2019 September 10. doi:10.7326/M19-0274.
2. Einarsson J. Are hysterectomy volumes in the US really falling? Contemporary OB/GYN. 1 September 2017. www.contemporaryobgyn.net/gynecology/are-hysterectomy-volumes-us-really-falling. November 4, 2019.
3. Temkin SM, Minasian L, Noone AM. The end of the hysterectomy epidemic and endometrial cancer incidence: what are the unintended consequences of declining hysterectomy rates? Front Oncol. 2016;6:89.
4. Doll KM, Dusetzina SB, Robinson W. Trends in inpatient and outpatient hysterectomy and oophorectomy rates among commercially insured women in the United States, 2000-2014. JAMA Surg. 2016;151:876-877.
5. Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279.
6. ACOG Practice Bulletin No. 141: management of menopausal symptoms [published corrections appear in: Obstet Gynecol. 2016;127(1):166. and Obstet Gynecol. 2018;131(3):604]. Obstet Gynecol. 2014;123:202-216.
7. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753.
1. Manson JE, Aragaki AK, Bassuk SS. Menopausal estrogen-alone therapy and health outcomes in women with and without bilateral oophorectomy: a randomized trial. Ann Intern Med. 2019 September 10. doi:10.7326/M19-0274.
2. Einarsson J. Are hysterectomy volumes in the US really falling? Contemporary OB/GYN. 1 September 2017. www.contemporaryobgyn.net/gynecology/are-hysterectomy-volumes-us-really-falling. November 4, 2019.
3. Temkin SM, Minasian L, Noone AM. The end of the hysterectomy epidemic and endometrial cancer incidence: what are the unintended consequences of declining hysterectomy rates? Front Oncol. 2016;6:89.
4. Doll KM, Dusetzina SB, Robinson W. Trends in inpatient and outpatient hysterectomy and oophorectomy rates among commercially insured women in the United States, 2000-2014. JAMA Surg. 2016;151:876-877.
5. Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279.
6. ACOG Practice Bulletin No. 141: management of menopausal symptoms [published corrections appear in: Obstet Gynecol. 2016;127(1):166. and Obstet Gynecol. 2018;131(3):604]. Obstet Gynecol. 2014;123:202-216.
7. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause. 2017;24:728-753.
Product Update: Addyi alcohol ban lifted, fezolinetant trial, outcomes tracker, comfort gown
FDA REMOVES ALCOHOL BAN WITH ADDYI
Sprout Pharmaceuticals announced that the US Food and Drug Administration (FDA) has removed their contraindication on alcohol use with Addyi® (flibanserin). Addyi was approved in 2015 and is an oral nonhormonal pill for acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women. Patients are advised to discontinue drinking alcohol at least 2 hours before taking Addyi at bedtime or skip the Addyi dose that evening.
The FDA also removed the requirement, under its Risk Evaluation and Mitigation Strategy (REMS) program, for health care practitioners or pharmacies to be certified to prescribe or dispense Addyi. Sprout says that to make all labeling elements consistent with the FDA’s findings the boxed warning will change and the medication guide will be updated and included under the REMS.
The most commonly reported adverse events among patients taking Addyi are dizziness, sleepiness, nausea, fatigue, insomnia, and dry mouth. Addyi is contraindicated in patients taking moderate or strong cytochrome P450 3A4 (CYP3A4) inhibitors and in those with hepatic impairment.
FOR MORE INFORMATION AND THE FULL PRESCRIBING INFORMATION AND MEDICATION GUIDE, VISIT: www.addyi.com
FEZOLINETANT FOR VMS
FOR MORE INFORMATION, VISIT: http://www.clinicaltrials.gov, TRIAL IDENTIFIERS NCT04003155, NCT04003142, AND NCT04003389
SOLUTIONS FOR OUTCOME TRACKING
DrChrono and OutcomeMD announce a partnership to track and analyze patient outcome data and confounding factors. DrChrono is an electronic health record (EHR) system, and OutcomeMD is a software solution that uses literature-validated patient-reported outcome instruments to score and track a patient’s symptom severity and inform treatment decisions for users.
Via a HIPAA compliant process, patients answer a list of questions that are accessed through a web link on their mobile or desktop devices. OutcomeMD summarizes the symptoms into a score that displays to both the physician and patient. Patients’ answers and scores are pushed to the clinician’s DrChrono EHR medical note.
FOR MORE INFORMATION, VISIT: www.outcomemd.com
Continue to: NEW MATERNITY GOWN...
NEW MATERNITY GOWN
ImageFIRST launched a new maternity gown for expecting mothers. The Comfort Care® Maternity Gown is a lightweight, premium polyester/nylon fabric that front snaps to allow for skin-to-skin access and optional breastfeeding. The gown also includes shoulder snaps and a full cut for extra coverage and to accommodate a variety of body types, says ImageFIRST.
ImageFIRST is a national linen rental provider. It developed the Comfort Care® Maternity Gown with input from labor and delivery departments to best meet the needs of expecting mothers. It also says that a portion of the proceeds from each gown rental will be donated to the National Pediatric Cancer Foundation.
FOR MORE INFORMATION, VISIT: www.imagefirst.com
FDA REMOVES ALCOHOL BAN WITH ADDYI
Sprout Pharmaceuticals announced that the US Food and Drug Administration (FDA) has removed their contraindication on alcohol use with Addyi® (flibanserin). Addyi was approved in 2015 and is an oral nonhormonal pill for acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women. Patients are advised to discontinue drinking alcohol at least 2 hours before taking Addyi at bedtime or skip the Addyi dose that evening.
The FDA also removed the requirement, under its Risk Evaluation and Mitigation Strategy (REMS) program, for health care practitioners or pharmacies to be certified to prescribe or dispense Addyi. Sprout says that to make all labeling elements consistent with the FDA’s findings the boxed warning will change and the medication guide will be updated and included under the REMS.
The most commonly reported adverse events among patients taking Addyi are dizziness, sleepiness, nausea, fatigue, insomnia, and dry mouth. Addyi is contraindicated in patients taking moderate or strong cytochrome P450 3A4 (CYP3A4) inhibitors and in those with hepatic impairment.
FOR MORE INFORMATION AND THE FULL PRESCRIBING INFORMATION AND MEDICATION GUIDE, VISIT: www.addyi.com
FEZOLINETANT FOR VMS
FOR MORE INFORMATION, VISIT: http://www.clinicaltrials.gov, TRIAL IDENTIFIERS NCT04003155, NCT04003142, AND NCT04003389
SOLUTIONS FOR OUTCOME TRACKING
DrChrono and OutcomeMD announce a partnership to track and analyze patient outcome data and confounding factors. DrChrono is an electronic health record (EHR) system, and OutcomeMD is a software solution that uses literature-validated patient-reported outcome instruments to score and track a patient’s symptom severity and inform treatment decisions for users.
Via a HIPAA compliant process, patients answer a list of questions that are accessed through a web link on their mobile or desktop devices. OutcomeMD summarizes the symptoms into a score that displays to both the physician and patient. Patients’ answers and scores are pushed to the clinician’s DrChrono EHR medical note.
FOR MORE INFORMATION, VISIT: www.outcomemd.com
Continue to: NEW MATERNITY GOWN...
NEW MATERNITY GOWN
ImageFIRST launched a new maternity gown for expecting mothers. The Comfort Care® Maternity Gown is a lightweight, premium polyester/nylon fabric that front snaps to allow for skin-to-skin access and optional breastfeeding. The gown also includes shoulder snaps and a full cut for extra coverage and to accommodate a variety of body types, says ImageFIRST.
ImageFIRST is a national linen rental provider. It developed the Comfort Care® Maternity Gown with input from labor and delivery departments to best meet the needs of expecting mothers. It also says that a portion of the proceeds from each gown rental will be donated to the National Pediatric Cancer Foundation.
FOR MORE INFORMATION, VISIT: www.imagefirst.com
FDA REMOVES ALCOHOL BAN WITH ADDYI
Sprout Pharmaceuticals announced that the US Food and Drug Administration (FDA) has removed their contraindication on alcohol use with Addyi® (flibanserin). Addyi was approved in 2015 and is an oral nonhormonal pill for acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women. Patients are advised to discontinue drinking alcohol at least 2 hours before taking Addyi at bedtime or skip the Addyi dose that evening.
The FDA also removed the requirement, under its Risk Evaluation and Mitigation Strategy (REMS) program, for health care practitioners or pharmacies to be certified to prescribe or dispense Addyi. Sprout says that to make all labeling elements consistent with the FDA’s findings the boxed warning will change and the medication guide will be updated and included under the REMS.
The most commonly reported adverse events among patients taking Addyi are dizziness, sleepiness, nausea, fatigue, insomnia, and dry mouth. Addyi is contraindicated in patients taking moderate or strong cytochrome P450 3A4 (CYP3A4) inhibitors and in those with hepatic impairment.
FOR MORE INFORMATION AND THE FULL PRESCRIBING INFORMATION AND MEDICATION GUIDE, VISIT: www.addyi.com
FEZOLINETANT FOR VMS
FOR MORE INFORMATION, VISIT: http://www.clinicaltrials.gov, TRIAL IDENTIFIERS NCT04003155, NCT04003142, AND NCT04003389
SOLUTIONS FOR OUTCOME TRACKING
DrChrono and OutcomeMD announce a partnership to track and analyze patient outcome data and confounding factors. DrChrono is an electronic health record (EHR) system, and OutcomeMD is a software solution that uses literature-validated patient-reported outcome instruments to score and track a patient’s symptom severity and inform treatment decisions for users.
Via a HIPAA compliant process, patients answer a list of questions that are accessed through a web link on their mobile or desktop devices. OutcomeMD summarizes the symptoms into a score that displays to both the physician and patient. Patients’ answers and scores are pushed to the clinician’s DrChrono EHR medical note.
FOR MORE INFORMATION, VISIT: www.outcomemd.com
Continue to: NEW MATERNITY GOWN...
NEW MATERNITY GOWN
ImageFIRST launched a new maternity gown for expecting mothers. The Comfort Care® Maternity Gown is a lightweight, premium polyester/nylon fabric that front snaps to allow for skin-to-skin access and optional breastfeeding. The gown also includes shoulder snaps and a full cut for extra coverage and to accommodate a variety of body types, says ImageFIRST.
ImageFIRST is a national linen rental provider. It developed the Comfort Care® Maternity Gown with input from labor and delivery departments to best meet the needs of expecting mothers. It also says that a portion of the proceeds from each gown rental will be donated to the National Pediatric Cancer Foundation.
FOR MORE INFORMATION, VISIT: www.imagefirst.com
Try testosterone for some women with sexual dysfunction, but not others
A new international position statement on testosterone therapy for women concludes that a trial of testosterone is appropriate for postmenopausal women with hypoactive sexual desire dysfunction (HSDD) and that its use for any other condition, symptom, or reason is not supported by available evidence.
The seven-page position statement, developed by an international task force of experts from the Endocrine Society, the American College of Gynecologists and Obstetricians, and multiple other medical societies, also emphasized that blood concentrations of testosterone should approximate premenopausal physiological conditions.
“When testosterone therapy is given, the resultant blood levels should not be above those seen in healthy young women,” said lead author Susan Ruth Davis, PhD, MBBS, of Monash University in Melbourne, Australia, in a press release issued by the Endocrine Society. Dr. Davis is president of the International Menopause Society, which coordinated the panel.
The statement was published in the Journal of Clinical Endocrinology & Metabolism and three other medical journals.
Margaret E. Wierman, MD, who represented the Endocrine Society on the task force, said in an interview that there has been “growing concern about testosterone being prescribed for a variety of signs and symptoms without data to support” such use. At the same time, there is significant concern about the ongoing lack of approved formulations licensed specifically for women, she said.
In part, the statement is about a renewed “call to industry to make some [female-specific] formulations so that we can examine other potential roles of testosterone in women,” said Dr. Wierman, professor of medicine and physiology at the University of Colorado at Denver, Aurora, and chief of endocrinology at the Rocky Mountain Regional Veterans Affairs Medical Center in Aurora.
“Testosterone may be useful [for indications other than HSDD], but we don’t know. There may be no [breast or cardiovascular disease risk], but we don’t know,” she said. “And without a formulation to study potential benefits and risks, it’s good to be cautious. It’s good to really outline where we have data and where we don’t.”
The Endocrine Society’s 2014 clinical practice guideline on androgen therapy in women, for which Dr. Wierman was the lead author, also recommended against the off-label use of testosterone for sexual dysfunction other than HSDD or for any other reason, such as cognitive, cardiovascular, metabolic, or bone health. As with the new statement, the society’s position statement was guided by an international, multisociety task force, albeit a smaller one.
For the new global position statement, the task force’s review of evidence includes a recently published systematic review and meta-analysis of randomized controlled trial data – of at least 12 weeks’ duration – on the use of testosterone for sexual function, cardiometabolic variables, cognitive measures, and musculoskeletal health. Some of the data from the randomized controlled trials were unpublished.
The meta-analysis, led by Dr. Davis and published in July in the Lancet Diabetes & Endocrinology, found that, compared with placebo or a comparator (such as estrogen, with or without progesterone), testosterone in either oral or transdermal form significantly improved sexual function in postmenopausal women. However, data about the effects of testosterone for other indications, its long-term safety, and its use in premenopausal women, were insufficient for drawing any conclusions (Lancet Diabetes Endocrinol. 2019 Jul 25. doi: 10.1016/S2213-8587[19]30189-5).
In addition, testosterone administered orally – but not nonorally (patch or cream) – was associated with adverse lipid profiles, Dr. Davis and her colleagues reported.
Another systematic review and meta-analysis, published in Fertility and Sterility in 2017 and included in the task force’s evidence review, focused specifically on transdermal testosterone for menopausal women with HSDD, with or without estrogen and progestin therapy. It also showed short-term efficacy in terms of improvement in sexual function, as well as short-term safety (Fertil Steril. 2017;107(2):475-82).
The new position statement warns about the lack of long-term safety data, stating that “safety data for testosterone in physiologic doses are not available beyond 24 months of treatment.”
In the short term, testosterone therapy for postmenopausal women (in doses approximating testosterone concentrations for premenopausal women), is associated with mild increases in acne and body/facial hair growth in some women, but not with alopecia, clitoromegaly, or voice change. Short-term transdermal therapy also does not seem to affect breast cancer risk or have any significant effects on lipid profiles, the statement says.
The panel points out, however, that randomized controlled trials with testosterone therapy have excluded women who are at high risk of cardiometabolic disease, and that women with a previous diagnosis of breast cancer have also been excluded from randomized trials of testosterone in women with HSDD. This is a “big issue,” said Dr. Wierman, and means that recommendations regarding the effect of testosterone in postmenopausal women with HSDD may not be generalizable to possible at-risk subpopulations.
The panel endorsed testosterone therapy specifically for women with HSDD because most of the studies reporting on sexual function have recruited women with diagnosed HSDD. Demonstrated benefits of testosterone in these cases include improved sexual desire, arousal, orgasm, and pleasure, and reduced concerns and distress about sex. HSDD should be diagnosed after formal biopsychosocial assessment, the statement notes.
“We don’t completely understand the control of sexual function in women, but it’s very dependent on estrogen status. And it’s also dependent on psychosocial factors, emotional health, relationship issues, and physical issues,” Dr. Wierman said in the interview.
“In practice, we look at all these issues, and we first optimize estrogen status. Once that’s done, and we’ve looked at all the other components of sexual function, then we can consider off-label use of testosterone,” she said. “If there’s no response in 3-6 months, we stop it.”
Testosterone levels do not correlate with sexual dysfunction, Dr. Wierman emphasized, and direct assays for the measurement of total and free testosterone are unreliable. The statement acknowledges that but still recommends measurement of testosterone using direct assays, in cases in which liquid/gas chromatography and tandem mass spectrometry assay (which has “high accuracy and reproducibility”) are not available. This is “to exclude high baseline concentrations and also to exclude supraphysiological concentrations during treatment,” the panel said.
Most endocrinologists and other experts who prescribe testosterone therapy for women use an approved male formulation off label and adjust it – an approach that the panel says is reasonable as long as hormone concentrations are “maintained in the physiologic female range.”
Compounded “bioidentical” testosterone therapy “cannot be recommended for the treatment of HSDD because of the lack of evidence for safety and efficacy,” the statement says.
“A big concern of many endocrinologists,” Dr. Wierman added, “is the recent explosion of using pharmacological levels of both estrogen and testosterone in either [injections] or pellets.” The Endocrine Society and other societies have alerted the Food and Drug Administration to “this new cottage industry, which may have significant side effects and risks for our patients,” she said.
Dr. Wierman reported received funding from Corcept Therapeutics, Novartis, and the Cancer League of Colorado, and honoraria or consultation fees from Pfizer to review ASPIRE grant applications for studies of acromegaly as well as Endocrine Society honorarium for teaching in the Endocrine Board Review and Clinical Endocrine Update. Dr. Davis reported receiving funding from a National Health and Medical Research Council Project Grant, a National Breast Foundation accelerator grant, and the Grollo-Ruzenne Foundation, as well as honoraria from Besins and Pfizer Australia. She has been a consultant to Besins Healthcare, Mayne Pharmaceuticals, Lawley Pharmaceuticals, and Que Oncology. Disclosures for other authors of the position statement are listed with the statement.
SOURCE: Davis SR et al. J Clin Endocrinol Metab. 2019 Sep 2. doi: 10.1210/jc.2019-01603.
A new international position statement on testosterone therapy for women concludes that a trial of testosterone is appropriate for postmenopausal women with hypoactive sexual desire dysfunction (HSDD) and that its use for any other condition, symptom, or reason is not supported by available evidence.
The seven-page position statement, developed by an international task force of experts from the Endocrine Society, the American College of Gynecologists and Obstetricians, and multiple other medical societies, also emphasized that blood concentrations of testosterone should approximate premenopausal physiological conditions.
“When testosterone therapy is given, the resultant blood levels should not be above those seen in healthy young women,” said lead author Susan Ruth Davis, PhD, MBBS, of Monash University in Melbourne, Australia, in a press release issued by the Endocrine Society. Dr. Davis is president of the International Menopause Society, which coordinated the panel.
The statement was published in the Journal of Clinical Endocrinology & Metabolism and three other medical journals.
Margaret E. Wierman, MD, who represented the Endocrine Society on the task force, said in an interview that there has been “growing concern about testosterone being prescribed for a variety of signs and symptoms without data to support” such use. At the same time, there is significant concern about the ongoing lack of approved formulations licensed specifically for women, she said.
In part, the statement is about a renewed “call to industry to make some [female-specific] formulations so that we can examine other potential roles of testosterone in women,” said Dr. Wierman, professor of medicine and physiology at the University of Colorado at Denver, Aurora, and chief of endocrinology at the Rocky Mountain Regional Veterans Affairs Medical Center in Aurora.
“Testosterone may be useful [for indications other than HSDD], but we don’t know. There may be no [breast or cardiovascular disease risk], but we don’t know,” she said. “And without a formulation to study potential benefits and risks, it’s good to be cautious. It’s good to really outline where we have data and where we don’t.”
The Endocrine Society’s 2014 clinical practice guideline on androgen therapy in women, for which Dr. Wierman was the lead author, also recommended against the off-label use of testosterone for sexual dysfunction other than HSDD or for any other reason, such as cognitive, cardiovascular, metabolic, or bone health. As with the new statement, the society’s position statement was guided by an international, multisociety task force, albeit a smaller one.
For the new global position statement, the task force’s review of evidence includes a recently published systematic review and meta-analysis of randomized controlled trial data – of at least 12 weeks’ duration – on the use of testosterone for sexual function, cardiometabolic variables, cognitive measures, and musculoskeletal health. Some of the data from the randomized controlled trials were unpublished.
The meta-analysis, led by Dr. Davis and published in July in the Lancet Diabetes & Endocrinology, found that, compared with placebo or a comparator (such as estrogen, with or without progesterone), testosterone in either oral or transdermal form significantly improved sexual function in postmenopausal women. However, data about the effects of testosterone for other indications, its long-term safety, and its use in premenopausal women, were insufficient for drawing any conclusions (Lancet Diabetes Endocrinol. 2019 Jul 25. doi: 10.1016/S2213-8587[19]30189-5).
In addition, testosterone administered orally – but not nonorally (patch or cream) – was associated with adverse lipid profiles, Dr. Davis and her colleagues reported.
Another systematic review and meta-analysis, published in Fertility and Sterility in 2017 and included in the task force’s evidence review, focused specifically on transdermal testosterone for menopausal women with HSDD, with or without estrogen and progestin therapy. It also showed short-term efficacy in terms of improvement in sexual function, as well as short-term safety (Fertil Steril. 2017;107(2):475-82).
The new position statement warns about the lack of long-term safety data, stating that “safety data for testosterone in physiologic doses are not available beyond 24 months of treatment.”
In the short term, testosterone therapy for postmenopausal women (in doses approximating testosterone concentrations for premenopausal women), is associated with mild increases in acne and body/facial hair growth in some women, but not with alopecia, clitoromegaly, or voice change. Short-term transdermal therapy also does not seem to affect breast cancer risk or have any significant effects on lipid profiles, the statement says.
The panel points out, however, that randomized controlled trials with testosterone therapy have excluded women who are at high risk of cardiometabolic disease, and that women with a previous diagnosis of breast cancer have also been excluded from randomized trials of testosterone in women with HSDD. This is a “big issue,” said Dr. Wierman, and means that recommendations regarding the effect of testosterone in postmenopausal women with HSDD may not be generalizable to possible at-risk subpopulations.
The panel endorsed testosterone therapy specifically for women with HSDD because most of the studies reporting on sexual function have recruited women with diagnosed HSDD. Demonstrated benefits of testosterone in these cases include improved sexual desire, arousal, orgasm, and pleasure, and reduced concerns and distress about sex. HSDD should be diagnosed after formal biopsychosocial assessment, the statement notes.
“We don’t completely understand the control of sexual function in women, but it’s very dependent on estrogen status. And it’s also dependent on psychosocial factors, emotional health, relationship issues, and physical issues,” Dr. Wierman said in the interview.
“In practice, we look at all these issues, and we first optimize estrogen status. Once that’s done, and we’ve looked at all the other components of sexual function, then we can consider off-label use of testosterone,” she said. “If there’s no response in 3-6 months, we stop it.”
Testosterone levels do not correlate with sexual dysfunction, Dr. Wierman emphasized, and direct assays for the measurement of total and free testosterone are unreliable. The statement acknowledges that but still recommends measurement of testosterone using direct assays, in cases in which liquid/gas chromatography and tandem mass spectrometry assay (which has “high accuracy and reproducibility”) are not available. This is “to exclude high baseline concentrations and also to exclude supraphysiological concentrations during treatment,” the panel said.
Most endocrinologists and other experts who prescribe testosterone therapy for women use an approved male formulation off label and adjust it – an approach that the panel says is reasonable as long as hormone concentrations are “maintained in the physiologic female range.”
Compounded “bioidentical” testosterone therapy “cannot be recommended for the treatment of HSDD because of the lack of evidence for safety and efficacy,” the statement says.
“A big concern of many endocrinologists,” Dr. Wierman added, “is the recent explosion of using pharmacological levels of both estrogen and testosterone in either [injections] or pellets.” The Endocrine Society and other societies have alerted the Food and Drug Administration to “this new cottage industry, which may have significant side effects and risks for our patients,” she said.
Dr. Wierman reported received funding from Corcept Therapeutics, Novartis, and the Cancer League of Colorado, and honoraria or consultation fees from Pfizer to review ASPIRE grant applications for studies of acromegaly as well as Endocrine Society honorarium for teaching in the Endocrine Board Review and Clinical Endocrine Update. Dr. Davis reported receiving funding from a National Health and Medical Research Council Project Grant, a National Breast Foundation accelerator grant, and the Grollo-Ruzenne Foundation, as well as honoraria from Besins and Pfizer Australia. She has been a consultant to Besins Healthcare, Mayne Pharmaceuticals, Lawley Pharmaceuticals, and Que Oncology. Disclosures for other authors of the position statement are listed with the statement.
SOURCE: Davis SR et al. J Clin Endocrinol Metab. 2019 Sep 2. doi: 10.1210/jc.2019-01603.
A new international position statement on testosterone therapy for women concludes that a trial of testosterone is appropriate for postmenopausal women with hypoactive sexual desire dysfunction (HSDD) and that its use for any other condition, symptom, or reason is not supported by available evidence.
The seven-page position statement, developed by an international task force of experts from the Endocrine Society, the American College of Gynecologists and Obstetricians, and multiple other medical societies, also emphasized that blood concentrations of testosterone should approximate premenopausal physiological conditions.
“When testosterone therapy is given, the resultant blood levels should not be above those seen in healthy young women,” said lead author Susan Ruth Davis, PhD, MBBS, of Monash University in Melbourne, Australia, in a press release issued by the Endocrine Society. Dr. Davis is president of the International Menopause Society, which coordinated the panel.
The statement was published in the Journal of Clinical Endocrinology & Metabolism and three other medical journals.
Margaret E. Wierman, MD, who represented the Endocrine Society on the task force, said in an interview that there has been “growing concern about testosterone being prescribed for a variety of signs and symptoms without data to support” such use. At the same time, there is significant concern about the ongoing lack of approved formulations licensed specifically for women, she said.
In part, the statement is about a renewed “call to industry to make some [female-specific] formulations so that we can examine other potential roles of testosterone in women,” said Dr. Wierman, professor of medicine and physiology at the University of Colorado at Denver, Aurora, and chief of endocrinology at the Rocky Mountain Regional Veterans Affairs Medical Center in Aurora.
“Testosterone may be useful [for indications other than HSDD], but we don’t know. There may be no [breast or cardiovascular disease risk], but we don’t know,” she said. “And without a formulation to study potential benefits and risks, it’s good to be cautious. It’s good to really outline where we have data and where we don’t.”
The Endocrine Society’s 2014 clinical practice guideline on androgen therapy in women, for which Dr. Wierman was the lead author, also recommended against the off-label use of testosterone for sexual dysfunction other than HSDD or for any other reason, such as cognitive, cardiovascular, metabolic, or bone health. As with the new statement, the society’s position statement was guided by an international, multisociety task force, albeit a smaller one.
For the new global position statement, the task force’s review of evidence includes a recently published systematic review and meta-analysis of randomized controlled trial data – of at least 12 weeks’ duration – on the use of testosterone for sexual function, cardiometabolic variables, cognitive measures, and musculoskeletal health. Some of the data from the randomized controlled trials were unpublished.
The meta-analysis, led by Dr. Davis and published in July in the Lancet Diabetes & Endocrinology, found that, compared with placebo or a comparator (such as estrogen, with or without progesterone), testosterone in either oral or transdermal form significantly improved sexual function in postmenopausal women. However, data about the effects of testosterone for other indications, its long-term safety, and its use in premenopausal women, were insufficient for drawing any conclusions (Lancet Diabetes Endocrinol. 2019 Jul 25. doi: 10.1016/S2213-8587[19]30189-5).
In addition, testosterone administered orally – but not nonorally (patch or cream) – was associated with adverse lipid profiles, Dr. Davis and her colleagues reported.
Another systematic review and meta-analysis, published in Fertility and Sterility in 2017 and included in the task force’s evidence review, focused specifically on transdermal testosterone for menopausal women with HSDD, with or without estrogen and progestin therapy. It also showed short-term efficacy in terms of improvement in sexual function, as well as short-term safety (Fertil Steril. 2017;107(2):475-82).
The new position statement warns about the lack of long-term safety data, stating that “safety data for testosterone in physiologic doses are not available beyond 24 months of treatment.”
In the short term, testosterone therapy for postmenopausal women (in doses approximating testosterone concentrations for premenopausal women), is associated with mild increases in acne and body/facial hair growth in some women, but not with alopecia, clitoromegaly, or voice change. Short-term transdermal therapy also does not seem to affect breast cancer risk or have any significant effects on lipid profiles, the statement says.
The panel points out, however, that randomized controlled trials with testosterone therapy have excluded women who are at high risk of cardiometabolic disease, and that women with a previous diagnosis of breast cancer have also been excluded from randomized trials of testosterone in women with HSDD. This is a “big issue,” said Dr. Wierman, and means that recommendations regarding the effect of testosterone in postmenopausal women with HSDD may not be generalizable to possible at-risk subpopulations.
The panel endorsed testosterone therapy specifically for women with HSDD because most of the studies reporting on sexual function have recruited women with diagnosed HSDD. Demonstrated benefits of testosterone in these cases include improved sexual desire, arousal, orgasm, and pleasure, and reduced concerns and distress about sex. HSDD should be diagnosed after formal biopsychosocial assessment, the statement notes.
“We don’t completely understand the control of sexual function in women, but it’s very dependent on estrogen status. And it’s also dependent on psychosocial factors, emotional health, relationship issues, and physical issues,” Dr. Wierman said in the interview.
“In practice, we look at all these issues, and we first optimize estrogen status. Once that’s done, and we’ve looked at all the other components of sexual function, then we can consider off-label use of testosterone,” she said. “If there’s no response in 3-6 months, we stop it.”
Testosterone levels do not correlate with sexual dysfunction, Dr. Wierman emphasized, and direct assays for the measurement of total and free testosterone are unreliable. The statement acknowledges that but still recommends measurement of testosterone using direct assays, in cases in which liquid/gas chromatography and tandem mass spectrometry assay (which has “high accuracy and reproducibility”) are not available. This is “to exclude high baseline concentrations and also to exclude supraphysiological concentrations during treatment,” the panel said.
Most endocrinologists and other experts who prescribe testosterone therapy for women use an approved male formulation off label and adjust it – an approach that the panel says is reasonable as long as hormone concentrations are “maintained in the physiologic female range.”
Compounded “bioidentical” testosterone therapy “cannot be recommended for the treatment of HSDD because of the lack of evidence for safety and efficacy,” the statement says.
“A big concern of many endocrinologists,” Dr. Wierman added, “is the recent explosion of using pharmacological levels of both estrogen and testosterone in either [injections] or pellets.” The Endocrine Society and other societies have alerted the Food and Drug Administration to “this new cottage industry, which may have significant side effects and risks for our patients,” she said.
Dr. Wierman reported received funding from Corcept Therapeutics, Novartis, and the Cancer League of Colorado, and honoraria or consultation fees from Pfizer to review ASPIRE grant applications for studies of acromegaly as well as Endocrine Society honorarium for teaching in the Endocrine Board Review and Clinical Endocrine Update. Dr. Davis reported receiving funding from a National Health and Medical Research Council Project Grant, a National Breast Foundation accelerator grant, and the Grollo-Ruzenne Foundation, as well as honoraria from Besins and Pfizer Australia. She has been a consultant to Besins Healthcare, Mayne Pharmaceuticals, Lawley Pharmaceuticals, and Que Oncology. Disclosures for other authors of the position statement are listed with the statement.
SOURCE: Davis SR et al. J Clin Endocrinol Metab. 2019 Sep 2. doi: 10.1210/jc.2019-01603.
FROM JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
2019 Update on female sexual dysfunction
Hypoactive sexual desire disorder (HSDD) is the most prevalent sexual health problem in women of all ages, with population-based studies showing that about 36% to 39% of women report low sexual desire, and 8% to 10% meet the diagnostic criteria of low sexual desire and associated distress.1,2 An expanded definition of HSDD may include3:
- lack of motivation for sexual activity (reduced or absent spontaneous desire or responsive desire to erotic cues and stimulation; inability to maintain desire or interest through sexual activity)
- loss of desire to initiate or participate in sexual activity (including avoiding situations that could lead to sexual activity) combined with significant personal distress (frustration, loss, sadness, worry) (FIGURE).4
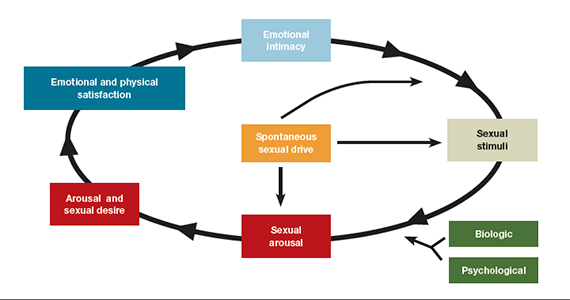
Despite the high prevalence of HSDD, patients often are uncomfortable and reluctant to voice concerns about low sexual desire to their ObGyn. Further, clinicians may feel ill equipped to diagnose and treat patients with HSDD. ObGyns, however, are well positioned to initiate a general discussion about sexual concerns with patients and use screening tools, such as the Decreased Sexual Desire Screener (DSDS), to facilitate a discussion and clarify a diagnosis of generalized acquired HSDD (TABLES 1 and 2).5 Helpful guidance on HSDD is available from the American College of Obstetricians and Gynecologists and the International Society for the Study of Women’s Sexual Health.6-8
Importantly, clinicians have a new treatment option they can offer to patients with HSDD. Bremelanotide was approved by the US Food and Drug Administration (FDA) on June 21, 2019, to treat acquired, generalized HSDD in premenopausal women. Up until this approval, flibanserin (approved in 2015) was the only drug FDA approved for the treatment of HSDD.
Assessing and treating HSDD today can be likened to managing depression 30 years ago, before selective serotonin receptor inhibitors were available. ObGyns would refer patients with depression to other health care providers, or not even ask patients about depressive symptoms because we had so little to offer. Once safe and effective antidepressants became available, knowing we could provide pharmacologic options made inquiring about depressive symptoms and the use of screening tools more readily incorporated into standard clinical practice. Depression is now recognized as a medical condition with biologic underpinnings, just like HSDD, and treatment options are available for both disorders.
For this Update, I had the opportunity to discuss the clinical trial experience with bremelanotide for HSDD with Dr. Sheryl Kingsberg, including efficacy and safety, dosage and administration, contraindications, and adverse events. She also details an ideal patient for treatment with bremelanotide, and we review pertinent aspects of flibanserin for comparative purposes.

Bremelanotide: A new therapeutic option
According to the product labeling for bremelanotide, the drug is indicated for the treatment of premenopausal women with acquired, generalized HSDD (low sexual desire that causes marked distress or interpersonal difficulty).9 This means that the HSDD developed in a woman who previously did not have problems with sexual desire, and that it occurred regardless of the type of stimulation, situation, or partner. In addition, the HSDD should not result from a coexisting medical or psychiatric condition, problems with the relationship, or the effects of a medication or drug substance.
Flibanserin also is indicated for the treatment of premenopausal women with HSDD. While both bremelanotide and flibanserin have indications only for premenopausal women, 2 studies of flibanserin in postmenopausal women have been published.10,11 Results from these studies in naturally menopausal women suggest that flibanserin may be efficacious in this population, with improvement in sexual desire, reduced distress associated with low desire, and improvement in the number of satisfying sexual events (SSEs).
No trials of bremelanotide in postmenopausal women have been published, but since this drug acts on central nervous system receptors, as does flibanserin, it may have similar effectiveness in postmenopausal women as well.

Continue to: Clinical trials show bremelanotide improves desire, reduces distress...
Clinical trials show bremelanotide improves desire, reduces distress
Two phase 3 clinical trials, dubbed the Reconnect studies, demonstrated that, compared with placebo, bremelanotide was associated with statistically significant improvements in sexual desire and levels of distress regarding sexual desire.
The 2 identical, randomized, placebo-controlled multicenter trials included 1,247 premenopausal women with HSDD of at least 6 months' duration.9,12 Bremelanotide 1.75 mg (or placebo) was self-administered subcutaneously with an autoinjector on an as-desired basis. The 24-week double-blind treatment period was followed by a 52-week open-label extension study.
The co-primary efficacy end points were the change from baseline to end-of-study (week 24 of the double-blind treatment period) in the 1) Female Sexual Function Index (FSFI) desire domain score and 2) feeling bothered by low sexual desire as measured by Question 13 on the Female Sexual Distress Scale (FSDS). An increase in the FSFI desire domain score over time denotes improvement in sexual desire, while a decrease in the FSDS Question 13 score over time indicates improvement in the level of distress associated with low sexual desire.
In the 2 clinical studies, the mean change from baseline (SD) in the FSFI desire domain score, which ranged from 1.2 to 6.0 at study outset (higher scores indicate greater desire), was:
- study 1: 0.5 (1.1) in the bremelanotide-treated women and 0.2 (1.0) in the placebo-treated women (P = .0002)
- study 2: 0.6 (1.0) in the bremelanotide group versus 0.2 (0.9) in the placebo group (P<.0001).
For FSDS Question 13, for which the score range was 0 to 4 (higher scores indicate greater bother), the mean change from baseline score was:
- study 1: -0.7 (1.2) in the bremelanotide-treated group compared with -0.4 (1.1) in the placebo-treated group (P<.0001)
- study 2: -0.7 (1.1) in the bremelanotide group and -0.4 (1.1) in the placebo group (P = .0053).
It should be noted that, in the past, SSEs were used as a primary end point in clinical studies. However, we have shifted from SSEs to desire and distress as an end point because SSEs have little to do with desire. Women worry about and are distressed by the fact that they no longer have sexual appetite. They no longer "want to want" even though their body will be responsive and they can have an orgasm. That is exemplified by the woman in our case scenario (see box, page 18), who very much wants the experience of being able to anticipate with pleasure the idea of having an enjoyable connection with her partner.
Continue to: Physiologic target: The melanocortin receptor...
Physiologic target: The melanocortin receptor
Bremelanotide's theorized mechanism of action is that it works to rebalance neurotransmitters that are implicated in causing HSDD, acting as an agonist on the melanocortin receptor to promote dopamine release and allow women to perceive sexual cues as rewarding. They can then respond to those cues the way they used to and therefore experience desire. Flibanserin has affinity for serotonin (5-hydroxytryptamine [5-HT]) receptors, with agonist and antagonist activity, as well as moderate antagonist activity on some dopamine receptors.
The bottom line is that we now have treatments to address the underlying biologic aspect of HSDD, which is a biopsychosocial disorder. Again, this has parallels to depression and its biologic mechanism, for which we have effective treatments.
Dosing is an as-needed injection
Unlike the daily nighttime oral dose required with flibanserin, bremelanotide is a 1.75-mg dose administered as a subcutaneous injection (in either the thigh or the abdomen) with a pen-like autoinjector, on an as-needed basis. It should be administered at least 45 minutes before anticipated sexual activity. That is a benefit for many women who do not want to take a daily pill when they know that their "desire to desire" may be once per week or once every other week.
Regarding the drug delivery mode, nobody dropped out of the bremelanotide clinical trials because of having to take an injection with an autoinjector, which employs a very thin needle and is virtually painless. A small number of bremelanotide-treated women, about 13%, had injection site reactions (compared with 8% in the placebo group), which is common with subcutaneous injection. Even in the phase 2 clinical trial, in which a syringe was used to administer the drug, no participants discontinued the study because of the injection mode.
There are no clear pharmacokinetic data on how long bremelanotide's effects last, but it may be anywhere from 8 to 16 hours. Patients should not take more than 1 dose within 24 hours--but since the effect may last up to 16 hours that should not be a problem--and use of more than 8 doses per month is not recommended.
While bremelanotide improves desire, certainly better than placebo, there is also some peripheral improvement in arousal, although women in the trials had only HSDD. We do not know whether bremelanotide would treat arousal disorder, but it will help women with or without arousal difficulties associated with their HSDD, as shown in a subgroup analysis in the trials.13
Counsel patients on treatment potentialities
Clinicians should be aware of several precautions with bremelanotide use.
Blood pressure increases. After each dose of bremelanotide, transient increases in blood pressure (6 mm Hg in systolic and 3 mm Hg in diastolic blood pressure) and reductions in heart rate (up to 5 beats per minute) occur; these measurements return to baseline usually within 12 hours postdose.9 When you think about whether having sexual desire will increase blood pressure, this may be physiologic. It is similar to walking up a flight of stairs.
The drug is not recommended, however, for use in patients at high risk for cardiovascular disease, and it is contraindicated in women with uncontrolled hypertension or known cardiovascular disease. Blood pressure should be well controlled before bremelanotide is initiated--use of antihypertensive agents is not contraindicated with bremelanotide as the drugs do not interact.
Clinicians are not required to participate in a Risk Evaluation and Mitigation Strategy (REMS) program to prescribe bremelanotide as they are with flibanserin (because of the increased risk of severe hypotension and syncope due to flibanserin's interaction with alcohol).
Drug interactions. Bremelanotide is a melanocortin receptor agonist--a unique compound. Antidepressants, other psychoactive medications, and oral contraceptives are not contraindicated with bremelanotide as there are no known interactions. Alcohol use also is not a contraindication or caution, in contrast to flibanserin. (In April, the FDA issued a labeling change order for flibanserin, specifying that alcohol does not have to be avoided completely when taking flibanserin, but that women should discontinue drinking alcohol at least 2 hours before taking the drug at bedtime, or skip the flibanserin dose that evening.14) Bremelanotide may slow gastric emptying, though, so when a patient is taking oral drugs that require threshold concentrations for efficacy, such as antibiotics, they should avoid bremelanotide. In addition, some drugs, such as indomethacin, may have a delayed onset of action with concomitant bremelanotide use.9
Importantly, patients should avoid using bremelanotide if they are taking an oral naltrexone product for treatment of alcohol or opioid addiction, because bremelanotide may decrease systemic exposure of oral naltrexone. That would potentially lead to naltrexone treatment failure and its consequences.9
Skin pigmentation changes. Hyperpigmentation occurred with bremelanotide use on the face, gingiva, and breasts, as reported in the clinical trials, in 1% of treated patients who received up to 8 doses per month, compared with no such occurrences in placebo-treated patients. In addition, 38% of patients who received bremelanotide daily for 8 days developed focal hyperpigmentation. It was not confirmed in all patients whether the hyperpigmentation resolved. Women with dark skin were more likely to develop hyperpigmentation.9
Common adverse reactions. The most common adverse reactions with bremelanotide treatment are nausea, flushing, injection site reactions, and headache, with most events being mild to moderate in intensity. In the clinical trials, 40% of the bremelanotide-treated women experienced nausea (compared with 1% of placebo-treated women), with most occurrences being mild; for most participants nausea improved with the second dose. Women had nausea that either went away or was intermittent, or it was mild enough that the drug benefits outweighed the tolerability costs--of women who experienced nausea, 92% continued in the trial, and 8% dropped out because of nausea.9
The following scenario describes the experience of HSDD in one of Dr. Kingsberg's patients.
CASE Woman avoids sex because of low desire; marriage is suffering
A 40-year-old woman, Sandra, who has been married for 19 years and has fraternal twins aged 8, presented to the behavioral medicine clinic with distressing symptoms of low sexual desire. For several years into the marriage the patient experienced excellent sex drive. After 6 to 7 years, she noticed that her desire had declined and that she was starting to avoid sex. She was irritated when her husband initiated sex, and she would make excuses as to why it was not the right time.
Her husband felt hurt, frustrated, and rejected. The couple was close to divorce because he was angry and resentful. Sandra recognized there was a problem but did not know how to fix it. She could not understand why her interest had waned since she still loved her husband and considered him objectively very attractive.
Sandra came to see Dr. Kingsberg at the behavioral medicine clinic. Using the 5-item validated diagnostic tool called the Decreased Sexual Desire Screener, Dr. Kingsberg diagnosed hypoactive sexual desire disorder (HSDD), a term Sandra had never heard of and did not know was a condition. The patient was relieved to know that she was one of several million women affected by HSDD and that the problem was not just that she was a "bad wife" or that she had some kind of psychological block. She emphasized how much she loved her husband and how she wanted desperately to "want to want desire," as she recalled feeling previously.
Sandra was treated with counseling and psychotherapy in which we addressed the relationship issues, the avoidance of sex, the comfort with being sexual, and the recognition that responsive desire can be helpful (as she was able to have arousal and orgasm and have a satisfying sexual event). The issue was that she had no motivation to seek out sex and had no interest in experiencing that pleasure. In subsequent couple's therapy, the husband recognized that his wife was not intentionally rejecting him, but that she had a real medical condition.
Although Sandra's relationship was now more stable and she and her husband were both working toward finding a solution to Sandra's loss of desire, she was still very distressed by her lack of desire. Sandra tried flibanserin for 3 months but unfortunately did not respond. Sandra heard about the recent approval of bremelanotide and is looking forward to the drug being available so that she can try it.
Final considerations
Asking patients about sexual function and using sexual function screening tools can help clinicians identify patients with the decreased sexual desire and associated distress characteristic of HSDD. ObGyns are the appropriate clinicians to treat these women and soon will have 2 pharmacologic options--bremelanotide (anticipated to be available in Fall 2019) and flibanserin--to offer patients with this biopsychosocial disorder that can adversely impact well-being and quality of life. Clinicians should individualize treatment, which may include psychotherapeutic counseling, and counsel patients on appropriate drug use and potential adverse effects.
AMAG Pharmaceuticals, Inc. has announced that they will have a copay assistance program for bremelanotide, where the first prescription of four autoinjectors will be a $0 copay, followed by a $99 copay or less for refills.15
- Shifren JL, Monz BU, Russo PA, et al. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112:970-978.
- West SL, D'Aloisio AA, Agans RP, et al. Prevalence of low sexual desire and hypoactive sexual desire disorder in a nationally representative sample of US women. Arch Intern Med. 2008;168:1441-1449.
- Parish SJ, Goldstein AT, Goldstein SW, et al. Toward a more evidence-based nosology and nomenclature for female sexual dysfunctions: part II. J Sex Med. 2016;13:1888-1906.
- Basson R. Using a different model for female sexual response to address women's problematic low sexual desire. J Sex Marital Ther. 2001;27:395-403.
- Clayton AH, Goldfischer ER, Goldstein I, et al. Validation of the Decreased Sexual Desire Screener (DSDS): a brief diagnostic instrument for generalized acquired female hypoactive sexual desire disorder (HSDD). J Sex Med. 2009;6:730-738.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins-Gynecology. ACOG practice bulletin no. 213: Female sexual dysfunction. Obstet Gynecol. 2019;134:e1-e18.
- Goldstein I, Kim NN, Clayton AH, et al. Hypoactive sexual desire disorder: International Society for the Study of Women's Sexual Health (ISSWSH) expert consensus panel review. Mayo Clin Proc. 2017;92:114-128.
- Clayton AH, Goldstein I, Kim NN, et al. The International Society for the Study of Women's Sexual Health process of care for management of hypoactive sexual desire disorder in women. Mayo Clin Proc. 2018;93:467-487.
- Vyleesi [package insert]. Waltham, MA: AMAG Pharmaceuticals; 2019.
- Simon JA, Kingsberg SA, Shumel B, et al. Efficacy and safety of flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the SNOWDROP trial. Menopause. 2014;21;633-640.
- Portman DJ, Brown L, Yuan, et al. Flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the PLUMERIA study. J Sex Med. 2017;14:834-842.
- Kingsberg SA, Clayton AH, Portman D, et al. Bremelanotide for the treatment of hypoactive sexual desire disorder: two randomized phase 3 trials. Obstet Gynecol. Forthcoming.
- Clayton AH, Lucas J, Jordon R, et al. Efficacy of the Investigational drug bremelanotide in the Reconnect studies. Poster presented at: 30th ECNP Congress of Applied and Translational Neuroscience; September 2-5, 2017, Paris, France.
- US Food and Drug Administration. FDA orders important safety labeling changes for Addyi [press release]. April 11, 2019. https://www.fda.gov/news-events/press-announcements/fda-orders-important-safety-labeling-changes-addyi. Accessed July 17, 2019.
- Vyleesi website (https://vyleesipro.com). Accessed August 5, 2019.
Hypoactive sexual desire disorder (HSDD) is the most prevalent sexual health problem in women of all ages, with population-based studies showing that about 36% to 39% of women report low sexual desire, and 8% to 10% meet the diagnostic criteria of low sexual desire and associated distress.1,2 An expanded definition of HSDD may include3:
- lack of motivation for sexual activity (reduced or absent spontaneous desire or responsive desire to erotic cues and stimulation; inability to maintain desire or interest through sexual activity)
- loss of desire to initiate or participate in sexual activity (including avoiding situations that could lead to sexual activity) combined with significant personal distress (frustration, loss, sadness, worry) (FIGURE).4

Despite the high prevalence of HSDD, patients often are uncomfortable and reluctant to voice concerns about low sexual desire to their ObGyn. Further, clinicians may feel ill equipped to diagnose and treat patients with HSDD. ObGyns, however, are well positioned to initiate a general discussion about sexual concerns with patients and use screening tools, such as the Decreased Sexual Desire Screener (DSDS), to facilitate a discussion and clarify a diagnosis of generalized acquired HSDD (TABLES 1 and 2).5 Helpful guidance on HSDD is available from the American College of Obstetricians and Gynecologists and the International Society for the Study of Women’s Sexual Health.6-8
Importantly, clinicians have a new treatment option they can offer to patients with HSDD. Bremelanotide was approved by the US Food and Drug Administration (FDA) on June 21, 2019, to treat acquired, generalized HSDD in premenopausal women. Up until this approval, flibanserin (approved in 2015) was the only drug FDA approved for the treatment of HSDD.
Assessing and treating HSDD today can be likened to managing depression 30 years ago, before selective serotonin receptor inhibitors were available. ObGyns would refer patients with depression to other health care providers, or not even ask patients about depressive symptoms because we had so little to offer. Once safe and effective antidepressants became available, knowing we could provide pharmacologic options made inquiring about depressive symptoms and the use of screening tools more readily incorporated into standard clinical practice. Depression is now recognized as a medical condition with biologic underpinnings, just like HSDD, and treatment options are available for both disorders.
For this Update, I had the opportunity to discuss the clinical trial experience with bremelanotide for HSDD with Dr. Sheryl Kingsberg, including efficacy and safety, dosage and administration, contraindications, and adverse events. She also details an ideal patient for treatment with bremelanotide, and we review pertinent aspects of flibanserin for comparative purposes.

Bremelanotide: A new therapeutic option
According to the product labeling for bremelanotide, the drug is indicated for the treatment of premenopausal women with acquired, generalized HSDD (low sexual desire that causes marked distress or interpersonal difficulty).9 This means that the HSDD developed in a woman who previously did not have problems with sexual desire, and that it occurred regardless of the type of stimulation, situation, or partner. In addition, the HSDD should not result from a coexisting medical or psychiatric condition, problems with the relationship, or the effects of a medication or drug substance.
Flibanserin also is indicated for the treatment of premenopausal women with HSDD. While both bremelanotide and flibanserin have indications only for premenopausal women, 2 studies of flibanserin in postmenopausal women have been published.10,11 Results from these studies in naturally menopausal women suggest that flibanserin may be efficacious in this population, with improvement in sexual desire, reduced distress associated with low desire, and improvement in the number of satisfying sexual events (SSEs).
No trials of bremelanotide in postmenopausal women have been published, but since this drug acts on central nervous system receptors, as does flibanserin, it may have similar effectiveness in postmenopausal women as well.

Continue to: Clinical trials show bremelanotide improves desire, reduces distress...
Clinical trials show bremelanotide improves desire, reduces distress
Two phase 3 clinical trials, dubbed the Reconnect studies, demonstrated that, compared with placebo, bremelanotide was associated with statistically significant improvements in sexual desire and levels of distress regarding sexual desire.
The 2 identical, randomized, placebo-controlled multicenter trials included 1,247 premenopausal women with HSDD of at least 6 months' duration.9,12 Bremelanotide 1.75 mg (or placebo) was self-administered subcutaneously with an autoinjector on an as-desired basis. The 24-week double-blind treatment period was followed by a 52-week open-label extension study.
The co-primary efficacy end points were the change from baseline to end-of-study (week 24 of the double-blind treatment period) in the 1) Female Sexual Function Index (FSFI) desire domain score and 2) feeling bothered by low sexual desire as measured by Question 13 on the Female Sexual Distress Scale (FSDS). An increase in the FSFI desire domain score over time denotes improvement in sexual desire, while a decrease in the FSDS Question 13 score over time indicates improvement in the level of distress associated with low sexual desire.
In the 2 clinical studies, the mean change from baseline (SD) in the FSFI desire domain score, which ranged from 1.2 to 6.0 at study outset (higher scores indicate greater desire), was:
- study 1: 0.5 (1.1) in the bremelanotide-treated women and 0.2 (1.0) in the placebo-treated women (P = .0002)
- study 2: 0.6 (1.0) in the bremelanotide group versus 0.2 (0.9) in the placebo group (P<.0001).
For FSDS Question 13, for which the score range was 0 to 4 (higher scores indicate greater bother), the mean change from baseline score was:
- study 1: -0.7 (1.2) in the bremelanotide-treated group compared with -0.4 (1.1) in the placebo-treated group (P<.0001)
- study 2: -0.7 (1.1) in the bremelanotide group and -0.4 (1.1) in the placebo group (P = .0053).
It should be noted that, in the past, SSEs were used as a primary end point in clinical studies. However, we have shifted from SSEs to desire and distress as an end point because SSEs have little to do with desire. Women worry about and are distressed by the fact that they no longer have sexual appetite. They no longer "want to want" even though their body will be responsive and they can have an orgasm. That is exemplified by the woman in our case scenario (see box, page 18), who very much wants the experience of being able to anticipate with pleasure the idea of having an enjoyable connection with her partner.
Continue to: Physiologic target: The melanocortin receptor...
Physiologic target: The melanocortin receptor
Bremelanotide's theorized mechanism of action is that it works to rebalance neurotransmitters that are implicated in causing HSDD, acting as an agonist on the melanocortin receptor to promote dopamine release and allow women to perceive sexual cues as rewarding. They can then respond to those cues the way they used to and therefore experience desire. Flibanserin has affinity for serotonin (5-hydroxytryptamine [5-HT]) receptors, with agonist and antagonist activity, as well as moderate antagonist activity on some dopamine receptors.
The bottom line is that we now have treatments to address the underlying biologic aspect of HSDD, which is a biopsychosocial disorder. Again, this has parallels to depression and its biologic mechanism, for which we have effective treatments.
Dosing is an as-needed injection
Unlike the daily nighttime oral dose required with flibanserin, bremelanotide is a 1.75-mg dose administered as a subcutaneous injection (in either the thigh or the abdomen) with a pen-like autoinjector, on an as-needed basis. It should be administered at least 45 minutes before anticipated sexual activity. That is a benefit for many women who do not want to take a daily pill when they know that their "desire to desire" may be once per week or once every other week.
Regarding the drug delivery mode, nobody dropped out of the bremelanotide clinical trials because of having to take an injection with an autoinjector, which employs a very thin needle and is virtually painless. A small number of bremelanotide-treated women, about 13%, had injection site reactions (compared with 8% in the placebo group), which is common with subcutaneous injection. Even in the phase 2 clinical trial, in which a syringe was used to administer the drug, no participants discontinued the study because of the injection mode.
There are no clear pharmacokinetic data on how long bremelanotide's effects last, but it may be anywhere from 8 to 16 hours. Patients should not take more than 1 dose within 24 hours--but since the effect may last up to 16 hours that should not be a problem--and use of more than 8 doses per month is not recommended.
While bremelanotide improves desire, certainly better than placebo, there is also some peripheral improvement in arousal, although women in the trials had only HSDD. We do not know whether bremelanotide would treat arousal disorder, but it will help women with or without arousal difficulties associated with their HSDD, as shown in a subgroup analysis in the trials.13
Counsel patients on treatment potentialities
Clinicians should be aware of several precautions with bremelanotide use.
Blood pressure increases. After each dose of bremelanotide, transient increases in blood pressure (6 mm Hg in systolic and 3 mm Hg in diastolic blood pressure) and reductions in heart rate (up to 5 beats per minute) occur; these measurements return to baseline usually within 12 hours postdose.9 When you think about whether having sexual desire will increase blood pressure, this may be physiologic. It is similar to walking up a flight of stairs.
The drug is not recommended, however, for use in patients at high risk for cardiovascular disease, and it is contraindicated in women with uncontrolled hypertension or known cardiovascular disease. Blood pressure should be well controlled before bremelanotide is initiated--use of antihypertensive agents is not contraindicated with bremelanotide as the drugs do not interact.
Clinicians are not required to participate in a Risk Evaluation and Mitigation Strategy (REMS) program to prescribe bremelanotide as they are with flibanserin (because of the increased risk of severe hypotension and syncope due to flibanserin's interaction with alcohol).
Drug interactions. Bremelanotide is a melanocortin receptor agonist--a unique compound. Antidepressants, other psychoactive medications, and oral contraceptives are not contraindicated with bremelanotide as there are no known interactions. Alcohol use also is not a contraindication or caution, in contrast to flibanserin. (In April, the FDA issued a labeling change order for flibanserin, specifying that alcohol does not have to be avoided completely when taking flibanserin, but that women should discontinue drinking alcohol at least 2 hours before taking the drug at bedtime, or skip the flibanserin dose that evening.14) Bremelanotide may slow gastric emptying, though, so when a patient is taking oral drugs that require threshold concentrations for efficacy, such as antibiotics, they should avoid bremelanotide. In addition, some drugs, such as indomethacin, may have a delayed onset of action with concomitant bremelanotide use.9
Importantly, patients should avoid using bremelanotide if they are taking an oral naltrexone product for treatment of alcohol or opioid addiction, because bremelanotide may decrease systemic exposure of oral naltrexone. That would potentially lead to naltrexone treatment failure and its consequences.9
Skin pigmentation changes. Hyperpigmentation occurred with bremelanotide use on the face, gingiva, and breasts, as reported in the clinical trials, in 1% of treated patients who received up to 8 doses per month, compared with no such occurrences in placebo-treated patients. In addition, 38% of patients who received bremelanotide daily for 8 days developed focal hyperpigmentation. It was not confirmed in all patients whether the hyperpigmentation resolved. Women with dark skin were more likely to develop hyperpigmentation.9
Common adverse reactions. The most common adverse reactions with bremelanotide treatment are nausea, flushing, injection site reactions, and headache, with most events being mild to moderate in intensity. In the clinical trials, 40% of the bremelanotide-treated women experienced nausea (compared with 1% of placebo-treated women), with most occurrences being mild; for most participants nausea improved with the second dose. Women had nausea that either went away or was intermittent, or it was mild enough that the drug benefits outweighed the tolerability costs--of women who experienced nausea, 92% continued in the trial, and 8% dropped out because of nausea.9
The following scenario describes the experience of HSDD in one of Dr. Kingsberg's patients.
CASE Woman avoids sex because of low desire; marriage is suffering
A 40-year-old woman, Sandra, who has been married for 19 years and has fraternal twins aged 8, presented to the behavioral medicine clinic with distressing symptoms of low sexual desire. For several years into the marriage the patient experienced excellent sex drive. After 6 to 7 years, she noticed that her desire had declined and that she was starting to avoid sex. She was irritated when her husband initiated sex, and she would make excuses as to why it was not the right time.
Her husband felt hurt, frustrated, and rejected. The couple was close to divorce because he was angry and resentful. Sandra recognized there was a problem but did not know how to fix it. She could not understand why her interest had waned since she still loved her husband and considered him objectively very attractive.
Sandra came to see Dr. Kingsberg at the behavioral medicine clinic. Using the 5-item validated diagnostic tool called the Decreased Sexual Desire Screener, Dr. Kingsberg diagnosed hypoactive sexual desire disorder (HSDD), a term Sandra had never heard of and did not know was a condition. The patient was relieved to know that she was one of several million women affected by HSDD and that the problem was not just that she was a "bad wife" or that she had some kind of psychological block. She emphasized how much she loved her husband and how she wanted desperately to "want to want desire," as she recalled feeling previously.
Sandra was treated with counseling and psychotherapy in which we addressed the relationship issues, the avoidance of sex, the comfort with being sexual, and the recognition that responsive desire can be helpful (as she was able to have arousal and orgasm and have a satisfying sexual event). The issue was that she had no motivation to seek out sex and had no interest in experiencing that pleasure. In subsequent couple's therapy, the husband recognized that his wife was not intentionally rejecting him, but that she had a real medical condition.
Although Sandra's relationship was now more stable and she and her husband were both working toward finding a solution to Sandra's loss of desire, she was still very distressed by her lack of desire. Sandra tried flibanserin for 3 months but unfortunately did not respond. Sandra heard about the recent approval of bremelanotide and is looking forward to the drug being available so that she can try it.
Final considerations
Asking patients about sexual function and using sexual function screening tools can help clinicians identify patients with the decreased sexual desire and associated distress characteristic of HSDD. ObGyns are the appropriate clinicians to treat these women and soon will have 2 pharmacologic options--bremelanotide (anticipated to be available in Fall 2019) and flibanserin--to offer patients with this biopsychosocial disorder that can adversely impact well-being and quality of life. Clinicians should individualize treatment, which may include psychotherapeutic counseling, and counsel patients on appropriate drug use and potential adverse effects.
AMAG Pharmaceuticals, Inc. has announced that they will have a copay assistance program for bremelanotide, where the first prescription of four autoinjectors will be a $0 copay, followed by a $99 copay or less for refills.15
Hypoactive sexual desire disorder (HSDD) is the most prevalent sexual health problem in women of all ages, with population-based studies showing that about 36% to 39% of women report low sexual desire, and 8% to 10% meet the diagnostic criteria of low sexual desire and associated distress.1,2 An expanded definition of HSDD may include3:
- lack of motivation for sexual activity (reduced or absent spontaneous desire or responsive desire to erotic cues and stimulation; inability to maintain desire or interest through sexual activity)
- loss of desire to initiate or participate in sexual activity (including avoiding situations that could lead to sexual activity) combined with significant personal distress (frustration, loss, sadness, worry) (FIGURE).4

Despite the high prevalence of HSDD, patients often are uncomfortable and reluctant to voice concerns about low sexual desire to their ObGyn. Further, clinicians may feel ill equipped to diagnose and treat patients with HSDD. ObGyns, however, are well positioned to initiate a general discussion about sexual concerns with patients and use screening tools, such as the Decreased Sexual Desire Screener (DSDS), to facilitate a discussion and clarify a diagnosis of generalized acquired HSDD (TABLES 1 and 2).5 Helpful guidance on HSDD is available from the American College of Obstetricians and Gynecologists and the International Society for the Study of Women’s Sexual Health.6-8
Importantly, clinicians have a new treatment option they can offer to patients with HSDD. Bremelanotide was approved by the US Food and Drug Administration (FDA) on June 21, 2019, to treat acquired, generalized HSDD in premenopausal women. Up until this approval, flibanserin (approved in 2015) was the only drug FDA approved for the treatment of HSDD.
Assessing and treating HSDD today can be likened to managing depression 30 years ago, before selective serotonin receptor inhibitors were available. ObGyns would refer patients with depression to other health care providers, or not even ask patients about depressive symptoms because we had so little to offer. Once safe and effective antidepressants became available, knowing we could provide pharmacologic options made inquiring about depressive symptoms and the use of screening tools more readily incorporated into standard clinical practice. Depression is now recognized as a medical condition with biologic underpinnings, just like HSDD, and treatment options are available for both disorders.
For this Update, I had the opportunity to discuss the clinical trial experience with bremelanotide for HSDD with Dr. Sheryl Kingsberg, including efficacy and safety, dosage and administration, contraindications, and adverse events. She also details an ideal patient for treatment with bremelanotide, and we review pertinent aspects of flibanserin for comparative purposes.

Bremelanotide: A new therapeutic option
According to the product labeling for bremelanotide, the drug is indicated for the treatment of premenopausal women with acquired, generalized HSDD (low sexual desire that causes marked distress or interpersonal difficulty).9 This means that the HSDD developed in a woman who previously did not have problems with sexual desire, and that it occurred regardless of the type of stimulation, situation, or partner. In addition, the HSDD should not result from a coexisting medical or psychiatric condition, problems with the relationship, or the effects of a medication or drug substance.
Flibanserin also is indicated for the treatment of premenopausal women with HSDD. While both bremelanotide and flibanserin have indications only for premenopausal women, 2 studies of flibanserin in postmenopausal women have been published.10,11 Results from these studies in naturally menopausal women suggest that flibanserin may be efficacious in this population, with improvement in sexual desire, reduced distress associated with low desire, and improvement in the number of satisfying sexual events (SSEs).
No trials of bremelanotide in postmenopausal women have been published, but since this drug acts on central nervous system receptors, as does flibanserin, it may have similar effectiveness in postmenopausal women as well.

Continue to: Clinical trials show bremelanotide improves desire, reduces distress...
Clinical trials show bremelanotide improves desire, reduces distress
Two phase 3 clinical trials, dubbed the Reconnect studies, demonstrated that, compared with placebo, bremelanotide was associated with statistically significant improvements in sexual desire and levels of distress regarding sexual desire.
The 2 identical, randomized, placebo-controlled multicenter trials included 1,247 premenopausal women with HSDD of at least 6 months' duration.9,12 Bremelanotide 1.75 mg (or placebo) was self-administered subcutaneously with an autoinjector on an as-desired basis. The 24-week double-blind treatment period was followed by a 52-week open-label extension study.
The co-primary efficacy end points were the change from baseline to end-of-study (week 24 of the double-blind treatment period) in the 1) Female Sexual Function Index (FSFI) desire domain score and 2) feeling bothered by low sexual desire as measured by Question 13 on the Female Sexual Distress Scale (FSDS). An increase in the FSFI desire domain score over time denotes improvement in sexual desire, while a decrease in the FSDS Question 13 score over time indicates improvement in the level of distress associated with low sexual desire.
In the 2 clinical studies, the mean change from baseline (SD) in the FSFI desire domain score, which ranged from 1.2 to 6.0 at study outset (higher scores indicate greater desire), was:
- study 1: 0.5 (1.1) in the bremelanotide-treated women and 0.2 (1.0) in the placebo-treated women (P = .0002)
- study 2: 0.6 (1.0) in the bremelanotide group versus 0.2 (0.9) in the placebo group (P<.0001).
For FSDS Question 13, for which the score range was 0 to 4 (higher scores indicate greater bother), the mean change from baseline score was:
- study 1: -0.7 (1.2) in the bremelanotide-treated group compared with -0.4 (1.1) in the placebo-treated group (P<.0001)
- study 2: -0.7 (1.1) in the bremelanotide group and -0.4 (1.1) in the placebo group (P = .0053).
It should be noted that, in the past, SSEs were used as a primary end point in clinical studies. However, we have shifted from SSEs to desire and distress as an end point because SSEs have little to do with desire. Women worry about and are distressed by the fact that they no longer have sexual appetite. They no longer "want to want" even though their body will be responsive and they can have an orgasm. That is exemplified by the woman in our case scenario (see box, page 18), who very much wants the experience of being able to anticipate with pleasure the idea of having an enjoyable connection with her partner.
Continue to: Physiologic target: The melanocortin receptor...
Physiologic target: The melanocortin receptor
Bremelanotide's theorized mechanism of action is that it works to rebalance neurotransmitters that are implicated in causing HSDD, acting as an agonist on the melanocortin receptor to promote dopamine release and allow women to perceive sexual cues as rewarding. They can then respond to those cues the way they used to and therefore experience desire. Flibanserin has affinity for serotonin (5-hydroxytryptamine [5-HT]) receptors, with agonist and antagonist activity, as well as moderate antagonist activity on some dopamine receptors.
The bottom line is that we now have treatments to address the underlying biologic aspect of HSDD, which is a biopsychosocial disorder. Again, this has parallels to depression and its biologic mechanism, for which we have effective treatments.
Dosing is an as-needed injection
Unlike the daily nighttime oral dose required with flibanserin, bremelanotide is a 1.75-mg dose administered as a subcutaneous injection (in either the thigh or the abdomen) with a pen-like autoinjector, on an as-needed basis. It should be administered at least 45 minutes before anticipated sexual activity. That is a benefit for many women who do not want to take a daily pill when they know that their "desire to desire" may be once per week or once every other week.
Regarding the drug delivery mode, nobody dropped out of the bremelanotide clinical trials because of having to take an injection with an autoinjector, which employs a very thin needle and is virtually painless. A small number of bremelanotide-treated women, about 13%, had injection site reactions (compared with 8% in the placebo group), which is common with subcutaneous injection. Even in the phase 2 clinical trial, in which a syringe was used to administer the drug, no participants discontinued the study because of the injection mode.
There are no clear pharmacokinetic data on how long bremelanotide's effects last, but it may be anywhere from 8 to 16 hours. Patients should not take more than 1 dose within 24 hours--but since the effect may last up to 16 hours that should not be a problem--and use of more than 8 doses per month is not recommended.
While bremelanotide improves desire, certainly better than placebo, there is also some peripheral improvement in arousal, although women in the trials had only HSDD. We do not know whether bremelanotide would treat arousal disorder, but it will help women with or without arousal difficulties associated with their HSDD, as shown in a subgroup analysis in the trials.13
Counsel patients on treatment potentialities
Clinicians should be aware of several precautions with bremelanotide use.
Blood pressure increases. After each dose of bremelanotide, transient increases in blood pressure (6 mm Hg in systolic and 3 mm Hg in diastolic blood pressure) and reductions in heart rate (up to 5 beats per minute) occur; these measurements return to baseline usually within 12 hours postdose.9 When you think about whether having sexual desire will increase blood pressure, this may be physiologic. It is similar to walking up a flight of stairs.
The drug is not recommended, however, for use in patients at high risk for cardiovascular disease, and it is contraindicated in women with uncontrolled hypertension or known cardiovascular disease. Blood pressure should be well controlled before bremelanotide is initiated--use of antihypertensive agents is not contraindicated with bremelanotide as the drugs do not interact.
Clinicians are not required to participate in a Risk Evaluation and Mitigation Strategy (REMS) program to prescribe bremelanotide as they are with flibanserin (because of the increased risk of severe hypotension and syncope due to flibanserin's interaction with alcohol).
Drug interactions. Bremelanotide is a melanocortin receptor agonist--a unique compound. Antidepressants, other psychoactive medications, and oral contraceptives are not contraindicated with bremelanotide as there are no known interactions. Alcohol use also is not a contraindication or caution, in contrast to flibanserin. (In April, the FDA issued a labeling change order for flibanserin, specifying that alcohol does not have to be avoided completely when taking flibanserin, but that women should discontinue drinking alcohol at least 2 hours before taking the drug at bedtime, or skip the flibanserin dose that evening.14) Bremelanotide may slow gastric emptying, though, so when a patient is taking oral drugs that require threshold concentrations for efficacy, such as antibiotics, they should avoid bremelanotide. In addition, some drugs, such as indomethacin, may have a delayed onset of action with concomitant bremelanotide use.9
Importantly, patients should avoid using bremelanotide if they are taking an oral naltrexone product for treatment of alcohol or opioid addiction, because bremelanotide may decrease systemic exposure of oral naltrexone. That would potentially lead to naltrexone treatment failure and its consequences.9
Skin pigmentation changes. Hyperpigmentation occurred with bremelanotide use on the face, gingiva, and breasts, as reported in the clinical trials, in 1% of treated patients who received up to 8 doses per month, compared with no such occurrences in placebo-treated patients. In addition, 38% of patients who received bremelanotide daily for 8 days developed focal hyperpigmentation. It was not confirmed in all patients whether the hyperpigmentation resolved. Women with dark skin were more likely to develop hyperpigmentation.9
Common adverse reactions. The most common adverse reactions with bremelanotide treatment are nausea, flushing, injection site reactions, and headache, with most events being mild to moderate in intensity. In the clinical trials, 40% of the bremelanotide-treated women experienced nausea (compared with 1% of placebo-treated women), with most occurrences being mild; for most participants nausea improved with the second dose. Women had nausea that either went away or was intermittent, or it was mild enough that the drug benefits outweighed the tolerability costs--of women who experienced nausea, 92% continued in the trial, and 8% dropped out because of nausea.9
The following scenario describes the experience of HSDD in one of Dr. Kingsberg's patients.
CASE Woman avoids sex because of low desire; marriage is suffering
A 40-year-old woman, Sandra, who has been married for 19 years and has fraternal twins aged 8, presented to the behavioral medicine clinic with distressing symptoms of low sexual desire. For several years into the marriage the patient experienced excellent sex drive. After 6 to 7 years, she noticed that her desire had declined and that she was starting to avoid sex. She was irritated when her husband initiated sex, and she would make excuses as to why it was not the right time.
Her husband felt hurt, frustrated, and rejected. The couple was close to divorce because he was angry and resentful. Sandra recognized there was a problem but did not know how to fix it. She could not understand why her interest had waned since she still loved her husband and considered him objectively very attractive.
Sandra came to see Dr. Kingsberg at the behavioral medicine clinic. Using the 5-item validated diagnostic tool called the Decreased Sexual Desire Screener, Dr. Kingsberg diagnosed hypoactive sexual desire disorder (HSDD), a term Sandra had never heard of and did not know was a condition. The patient was relieved to know that she was one of several million women affected by HSDD and that the problem was not just that she was a "bad wife" or that she had some kind of psychological block. She emphasized how much she loved her husband and how she wanted desperately to "want to want desire," as she recalled feeling previously.
Sandra was treated with counseling and psychotherapy in which we addressed the relationship issues, the avoidance of sex, the comfort with being sexual, and the recognition that responsive desire can be helpful (as she was able to have arousal and orgasm and have a satisfying sexual event). The issue was that she had no motivation to seek out sex and had no interest in experiencing that pleasure. In subsequent couple's therapy, the husband recognized that his wife was not intentionally rejecting him, but that she had a real medical condition.
Although Sandra's relationship was now more stable and she and her husband were both working toward finding a solution to Sandra's loss of desire, she was still very distressed by her lack of desire. Sandra tried flibanserin for 3 months but unfortunately did not respond. Sandra heard about the recent approval of bremelanotide and is looking forward to the drug being available so that she can try it.
Final considerations
Asking patients about sexual function and using sexual function screening tools can help clinicians identify patients with the decreased sexual desire and associated distress characteristic of HSDD. ObGyns are the appropriate clinicians to treat these women and soon will have 2 pharmacologic options--bremelanotide (anticipated to be available in Fall 2019) and flibanserin--to offer patients with this biopsychosocial disorder that can adversely impact well-being and quality of life. Clinicians should individualize treatment, which may include psychotherapeutic counseling, and counsel patients on appropriate drug use and potential adverse effects.
AMAG Pharmaceuticals, Inc. has announced that they will have a copay assistance program for bremelanotide, where the first prescription of four autoinjectors will be a $0 copay, followed by a $99 copay or less for refills.15
- Shifren JL, Monz BU, Russo PA, et al. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112:970-978.
- West SL, D'Aloisio AA, Agans RP, et al. Prevalence of low sexual desire and hypoactive sexual desire disorder in a nationally representative sample of US women. Arch Intern Med. 2008;168:1441-1449.
- Parish SJ, Goldstein AT, Goldstein SW, et al. Toward a more evidence-based nosology and nomenclature for female sexual dysfunctions: part II. J Sex Med. 2016;13:1888-1906.
- Basson R. Using a different model for female sexual response to address women's problematic low sexual desire. J Sex Marital Ther. 2001;27:395-403.
- Clayton AH, Goldfischer ER, Goldstein I, et al. Validation of the Decreased Sexual Desire Screener (DSDS): a brief diagnostic instrument for generalized acquired female hypoactive sexual desire disorder (HSDD). J Sex Med. 2009;6:730-738.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins-Gynecology. ACOG practice bulletin no. 213: Female sexual dysfunction. Obstet Gynecol. 2019;134:e1-e18.
- Goldstein I, Kim NN, Clayton AH, et al. Hypoactive sexual desire disorder: International Society for the Study of Women's Sexual Health (ISSWSH) expert consensus panel review. Mayo Clin Proc. 2017;92:114-128.
- Clayton AH, Goldstein I, Kim NN, et al. The International Society for the Study of Women's Sexual Health process of care for management of hypoactive sexual desire disorder in women. Mayo Clin Proc. 2018;93:467-487.
- Vyleesi [package insert]. Waltham, MA: AMAG Pharmaceuticals; 2019.
- Simon JA, Kingsberg SA, Shumel B, et al. Efficacy and safety of flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the SNOWDROP trial. Menopause. 2014;21;633-640.
- Portman DJ, Brown L, Yuan, et al. Flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the PLUMERIA study. J Sex Med. 2017;14:834-842.
- Kingsberg SA, Clayton AH, Portman D, et al. Bremelanotide for the treatment of hypoactive sexual desire disorder: two randomized phase 3 trials. Obstet Gynecol. Forthcoming.
- Clayton AH, Lucas J, Jordon R, et al. Efficacy of the Investigational drug bremelanotide in the Reconnect studies. Poster presented at: 30th ECNP Congress of Applied and Translational Neuroscience; September 2-5, 2017, Paris, France.
- US Food and Drug Administration. FDA orders important safety labeling changes for Addyi [press release]. April 11, 2019. https://www.fda.gov/news-events/press-announcements/fda-orders-important-safety-labeling-changes-addyi. Accessed July 17, 2019.
- Vyleesi website (https://vyleesipro.com). Accessed August 5, 2019.
- Shifren JL, Monz BU, Russo PA, et al. Sexual problems and distress in United States women: prevalence and correlates. Obstet Gynecol. 2008;112:970-978.
- West SL, D'Aloisio AA, Agans RP, et al. Prevalence of low sexual desire and hypoactive sexual desire disorder in a nationally representative sample of US women. Arch Intern Med. 2008;168:1441-1449.
- Parish SJ, Goldstein AT, Goldstein SW, et al. Toward a more evidence-based nosology and nomenclature for female sexual dysfunctions: part II. J Sex Med. 2016;13:1888-1906.
- Basson R. Using a different model for female sexual response to address women's problematic low sexual desire. J Sex Marital Ther. 2001;27:395-403.
- Clayton AH, Goldfischer ER, Goldstein I, et al. Validation of the Decreased Sexual Desire Screener (DSDS): a brief diagnostic instrument for generalized acquired female hypoactive sexual desire disorder (HSDD). J Sex Med. 2009;6:730-738.
- American College of Obstetricians and Gynecologists Committee on Practice Bulletins-Gynecology. ACOG practice bulletin no. 213: Female sexual dysfunction. Obstet Gynecol. 2019;134:e1-e18.
- Goldstein I, Kim NN, Clayton AH, et al. Hypoactive sexual desire disorder: International Society for the Study of Women's Sexual Health (ISSWSH) expert consensus panel review. Mayo Clin Proc. 2017;92:114-128.
- Clayton AH, Goldstein I, Kim NN, et al. The International Society for the Study of Women's Sexual Health process of care for management of hypoactive sexual desire disorder in women. Mayo Clin Proc. 2018;93:467-487.
- Vyleesi [package insert]. Waltham, MA: AMAG Pharmaceuticals; 2019.
- Simon JA, Kingsberg SA, Shumel B, et al. Efficacy and safety of flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the SNOWDROP trial. Menopause. 2014;21;633-640.
- Portman DJ, Brown L, Yuan, et al. Flibanserin in postmenopausal women with hypoactive sexual desire disorder: results of the PLUMERIA study. J Sex Med. 2017;14:834-842.
- Kingsberg SA, Clayton AH, Portman D, et al. Bremelanotide for the treatment of hypoactive sexual desire disorder: two randomized phase 3 trials. Obstet Gynecol. Forthcoming.
- Clayton AH, Lucas J, Jordon R, et al. Efficacy of the Investigational drug bremelanotide in the Reconnect studies. Poster presented at: 30th ECNP Congress of Applied and Translational Neuroscience; September 2-5, 2017, Paris, France.
- US Food and Drug Administration. FDA orders important safety labeling changes for Addyi [press release]. April 11, 2019. https://www.fda.gov/news-events/press-announcements/fda-orders-important-safety-labeling-changes-addyi. Accessed July 17, 2019.
- Vyleesi website (https://vyleesipro.com). Accessed August 5, 2019.
Survey: Patient-provider communication regarding dyspareunia disappoints
NASHVILLE, TENN. – Many women with endometriosis experience dyspareunia, but they are largely unsatisfied when it comes to discussions with health care providers about their symptoms, the results of an online survey suggest.
Of 638 women with self-reported endometriosis who responded to the survey, 81% said they always or usually experience pain during intercourse, 51% described their pain as severe, and 49% said they experience pain lasting more than 24 hours, Roberta Renzelli-Cain, DO, reported during a poster session at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists.
“The results of our survey suggest that said Dr. Renzelli-Cain, director of the West Virginia National Center of Excellence in Women’s Health and an ob.gyn. at West Virginia University, Morgantown.
In fact, survey responses suggested that dyspareunia has a marked impact on quality of life; 69% of respondents said they find sexual intercourse unpleasant, 31% said they always or usually avoid intercourse, 44% strongly agreed that dyspareunia has affected their relationship with their spouse or partner, 63% said they worry that their spouse or partner will leave, and 63% said they feel depressed because of their dyspareunia, she and her colleagues found.
Most respondents (88%) discussed their symptoms with health care providers (HCPs), and 85% did so with their ob.gyn. Among the other HCPs who respondents spoke with about their dyspareunia were primary care physicians, nurse practitioners, emergency department doctors, fertility specialists, and pain specialists.
Among the reasons given for avoiding discussions with HCPs about painful intercourse were embarrassment (34% of respondents), thinking nothing would help (26%), the physician was a man (5%), and a feeling that the provider was not understanding (3%).
Overall, 18% of respondents said they received no advice from their HCPs regarding how to deal with their dyspareunia, and 39% found nothing that their HCPs suggested to be effective.
Advice given by HCPs included surgery, lubricant use, over-the-counter pain medication, and trying different sexual positions. The percentages of respondents receiving this advice, and the percentages who considered the advice effective, respectively, were 46%, 25% for surgery; 32%, 21% for lubricant use; 36%, 18% for OTC medication; and 21%, 14% for trying different sexual positions, the investigators said.
Importantly, 42% of respondent said they felt it would be easier to discuss dyspareunia if their HCP initiated the subject.
The findings are notable given that 6%-10% of women of childbearing age are affected by endometriosis, and about 30% of those women have related dyspareunia – a “challenging symptom associated with lower sexual functioning, as well as lower self-esteem, and body image,” the investigators wrote.
The 24-question English-language survey was conducted online among women aged 19 years or older who reported having endometriosis and dyspareunia. Participants were recruited via a social network for women with endometriosis (MyEndometriosisTeam.com) and invited by e-mail to participate.
Of the 32,865 invited participants, 361 U.S.-based women and 277 women from outside the United States completed the survey. Most (83%) were aged 19-29 years.
In this online survey, the majority of women reported suboptimal communication with HCPs when seeking help for dyspareunia, the investigators said, concluding that “these results were similar between the U.S.- and non-U.S.–based women, highlighting the need for better medical communication between patients and HCPs, and better advice for patients regarding dyspareunia.”
Dr. Renzelli-Cain reported having no relevant financial disclosures.
NASHVILLE, TENN. – Many women with endometriosis experience dyspareunia, but they are largely unsatisfied when it comes to discussions with health care providers about their symptoms, the results of an online survey suggest.
Of 638 women with self-reported endometriosis who responded to the survey, 81% said they always or usually experience pain during intercourse, 51% described their pain as severe, and 49% said they experience pain lasting more than 24 hours, Roberta Renzelli-Cain, DO, reported during a poster session at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists.
“The results of our survey suggest that said Dr. Renzelli-Cain, director of the West Virginia National Center of Excellence in Women’s Health and an ob.gyn. at West Virginia University, Morgantown.
In fact, survey responses suggested that dyspareunia has a marked impact on quality of life; 69% of respondents said they find sexual intercourse unpleasant, 31% said they always or usually avoid intercourse, 44% strongly agreed that dyspareunia has affected their relationship with their spouse or partner, 63% said they worry that their spouse or partner will leave, and 63% said they feel depressed because of their dyspareunia, she and her colleagues found.
Most respondents (88%) discussed their symptoms with health care providers (HCPs), and 85% did so with their ob.gyn. Among the other HCPs who respondents spoke with about their dyspareunia were primary care physicians, nurse practitioners, emergency department doctors, fertility specialists, and pain specialists.
Among the reasons given for avoiding discussions with HCPs about painful intercourse were embarrassment (34% of respondents), thinking nothing would help (26%), the physician was a man (5%), and a feeling that the provider was not understanding (3%).
Overall, 18% of respondents said they received no advice from their HCPs regarding how to deal with their dyspareunia, and 39% found nothing that their HCPs suggested to be effective.
Advice given by HCPs included surgery, lubricant use, over-the-counter pain medication, and trying different sexual positions. The percentages of respondents receiving this advice, and the percentages who considered the advice effective, respectively, were 46%, 25% for surgery; 32%, 21% for lubricant use; 36%, 18% for OTC medication; and 21%, 14% for trying different sexual positions, the investigators said.
Importantly, 42% of respondent said they felt it would be easier to discuss dyspareunia if their HCP initiated the subject.
The findings are notable given that 6%-10% of women of childbearing age are affected by endometriosis, and about 30% of those women have related dyspareunia – a “challenging symptom associated with lower sexual functioning, as well as lower self-esteem, and body image,” the investigators wrote.
The 24-question English-language survey was conducted online among women aged 19 years or older who reported having endometriosis and dyspareunia. Participants were recruited via a social network for women with endometriosis (MyEndometriosisTeam.com) and invited by e-mail to participate.
Of the 32,865 invited participants, 361 U.S.-based women and 277 women from outside the United States completed the survey. Most (83%) were aged 19-29 years.
In this online survey, the majority of women reported suboptimal communication with HCPs when seeking help for dyspareunia, the investigators said, concluding that “these results were similar between the U.S.- and non-U.S.–based women, highlighting the need for better medical communication between patients and HCPs, and better advice for patients regarding dyspareunia.”
Dr. Renzelli-Cain reported having no relevant financial disclosures.
NASHVILLE, TENN. – Many women with endometriosis experience dyspareunia, but they are largely unsatisfied when it comes to discussions with health care providers about their symptoms, the results of an online survey suggest.
Of 638 women with self-reported endometriosis who responded to the survey, 81% said they always or usually experience pain during intercourse, 51% described their pain as severe, and 49% said they experience pain lasting more than 24 hours, Roberta Renzelli-Cain, DO, reported during a poster session at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists.
“The results of our survey suggest that said Dr. Renzelli-Cain, director of the West Virginia National Center of Excellence in Women’s Health and an ob.gyn. at West Virginia University, Morgantown.
In fact, survey responses suggested that dyspareunia has a marked impact on quality of life; 69% of respondents said they find sexual intercourse unpleasant, 31% said they always or usually avoid intercourse, 44% strongly agreed that dyspareunia has affected their relationship with their spouse or partner, 63% said they worry that their spouse or partner will leave, and 63% said they feel depressed because of their dyspareunia, she and her colleagues found.
Most respondents (88%) discussed their symptoms with health care providers (HCPs), and 85% did so with their ob.gyn. Among the other HCPs who respondents spoke with about their dyspareunia were primary care physicians, nurse practitioners, emergency department doctors, fertility specialists, and pain specialists.
Among the reasons given for avoiding discussions with HCPs about painful intercourse were embarrassment (34% of respondents), thinking nothing would help (26%), the physician was a man (5%), and a feeling that the provider was not understanding (3%).
Overall, 18% of respondents said they received no advice from their HCPs regarding how to deal with their dyspareunia, and 39% found nothing that their HCPs suggested to be effective.
Advice given by HCPs included surgery, lubricant use, over-the-counter pain medication, and trying different sexual positions. The percentages of respondents receiving this advice, and the percentages who considered the advice effective, respectively, were 46%, 25% for surgery; 32%, 21% for lubricant use; 36%, 18% for OTC medication; and 21%, 14% for trying different sexual positions, the investigators said.
Importantly, 42% of respondent said they felt it would be easier to discuss dyspareunia if their HCP initiated the subject.
The findings are notable given that 6%-10% of women of childbearing age are affected by endometriosis, and about 30% of those women have related dyspareunia – a “challenging symptom associated with lower sexual functioning, as well as lower self-esteem, and body image,” the investigators wrote.
The 24-question English-language survey was conducted online among women aged 19 years or older who reported having endometriosis and dyspareunia. Participants were recruited via a social network for women with endometriosis (MyEndometriosisTeam.com) and invited by e-mail to participate.
Of the 32,865 invited participants, 361 U.S.-based women and 277 women from outside the United States completed the survey. Most (83%) were aged 19-29 years.
In this online survey, the majority of women reported suboptimal communication with HCPs when seeking help for dyspareunia, the investigators said, concluding that “these results were similar between the U.S.- and non-U.S.–based women, highlighting the need for better medical communication between patients and HCPs, and better advice for patients regarding dyspareunia.”
Dr. Renzelli-Cain reported having no relevant financial disclosures.
REPORTING FROM ACOG 2019
Sexual assault in military linked to sexual pain
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
according to an observational study involving interviews with more than 1,000 military women.
Female veterans with histories of both childhood sexual abuse and sexual assault in the military were 4.33 times more likely to report sexual pain than female veterans with no history of sexual assault; women whose history of sexual assault occurred in the military only were 2.37 times more likely to report sexual pain. Those with histories of childhood sexual abuse but no military assaults were 1.75 times more likely to report sexual pain than those who had no history of sexual assault.
The findings suggest that sexual assault in the military is more detrimental to sexual function than childhood sexual abuse alone, which “is distinct from the pattern long observed in civilian women that childhood sexual abuse confers a greater risk for sexual pain than adulthood sexual assault,” Carey S. Pulverman, PhD, then of the Department of Veterans Affairs Center of Excellence for Research on Returning War Veterans in Waco, Tex., and coinvestigators wrote in Obstetrics & Gynecology.
The findings come from a secondary analysis of data collected for a larger project titled Sexual Violence and Women Veterans’ Gynecologic Health . The research team conducted telephone interviews with 1,004 female veterans younger than 52 years of age (mean, 38 years) who were enrolled at two large Midwestern VA medical centers and associated clinics. Sexual pain was assessed by one question: “Does it hurt you to have sexual intercourse or penetration?”
The study also identified high comorbidity between sexual pain and mental health concerns. As with sexual pain, rates of depression and PTSD were highest among female veterans with histories of both sexual abuse in childhood and sexual assault in the military, followed by women with histories of sexual assaults in the military alone, and then women with histories of childhood sexual abuse alone. Women with both histories were 6.35 times more likely to report PTSD, and 3.91 times more likely to report depression, compared with female veterans with no history of sexual assault.
Women who experienced sexual assault during their childhood and/or while serving in the military also may have been exposed to sexual assault during their pre- or postmilitary adulthood as well, but this was a small number and its effects were not evaluated, the authors noted.
Especially given the “growing numbers of women serving in the military and prevalence of sexual assault in this population,” there’s a need for more research on the sexual function of female veterans and development of “targeted treatments,” the investigators wrote.
For now, providers should be “more comprehensive in their assessment of sexual assault history” and should consider developing relationships with community providers who specialize in sexual health, they added.
The study was funded by the VA. The authors did not report any relevant financial disclosures.
SOURCE: Pulverman CS et al. Obstet Gynecol. 2019;134:63-71.
FROM OBSTETRICS & GYNECOLOGY
FDA approves drug to treat low sexual desire in women
The .
“There are women who, for no known reason, have reduced sexual desire that causes marked distress, and who can benefit from safe and effective pharmacologic treatment,” Hylton V. Joffe, MD, director of the Center for Drug Evaluation and Research’s Division of Bone, Reproductive, and Urologic Products, stated in a press release. “Today’s approval provides women with another treatment option for this condition.”
HSDD is characterized by low sexual desire that causes marked distress or interpersonal difficulty and is not caused by a medical or psychiatric condition. Acquired HSDD develops in a patient who previously experienced no problems with sexual desire, and generalized HSDD is a lack of desire that occurs regardless of the type of sexual activity, situation, or partner.
Vyleesi was studied in two 24-week, randomized, double-blind, placebo-controlled trials in 1,247 premenopausal women with acquired, generalized HSDD. The women used Vyleesi two or three times per month and no more than once a week. About one-quarter of patients treated with Vyleesi had an increase of 1.2 or more in their sexual desire score (scored on a range of 1.2 to 6.0, with higher scores indicating greater sexual desire), compared with about 17% of those who took placebo. About 35% of the patients treated with Vyleesi had a decrease of one or more in their distress score (scored on a range of 0-4, with higher scores indicating greater distress from low sexual desire) compared with about 31% of those who took placebo.
The drug is injected under the skin of the abdomen or thigh at least 45 minutes before anticipated sexual activity. Patients may decide the optimal time to use Vyleesi based on the duration of benefit and any side effects, such as nausea. Patients should not take more than one dose of Vyleesi within 24 hours, or more than eight doses per month. Patients should discontinue treatment after 8 weeks if they do not report an improvement in sexual desire and associated distress.
Vyleesi works by activating melanocortin receptors but the exact mechanism for improving sexual desire is unknown. Some side effects were reported. “The most common side effects of Vyleesi are nausea and vomiting, flushing, injection site reactions, and headache. About 40% of patients in the clinical trials experienced nausea, most commonly with the first Vyleesi injection, and 13% needed medications for the treatment of nausea. About 1% of patients treated with Vyleesi in the clinical trials reported darkening of the gums and parts of the skin, including the face and breasts, which did not go away in about half the patients after stopping treatment. Patients with dark skin were more likely to develop this side effect,” according to the press release.
A temporary increase in blood pressure in patients after dosing with Vyleesi was observed during the clinical trials and therefore the drug is not recommended in patients at high risk for cardiovascular disease. In addition, patients who take a naltrexone-containing medication by mouth to treat alcohol or opioid dependence should not use Vyleesi because it may significantly decrease the levels of naltrexone in the blood and could lead to naltrexone treatment failure.
The full press release can be found on the FDA website.
The .
“There are women who, for no known reason, have reduced sexual desire that causes marked distress, and who can benefit from safe and effective pharmacologic treatment,” Hylton V. Joffe, MD, director of the Center for Drug Evaluation and Research’s Division of Bone, Reproductive, and Urologic Products, stated in a press release. “Today’s approval provides women with another treatment option for this condition.”
HSDD is characterized by low sexual desire that causes marked distress or interpersonal difficulty and is not caused by a medical or psychiatric condition. Acquired HSDD develops in a patient who previously experienced no problems with sexual desire, and generalized HSDD is a lack of desire that occurs regardless of the type of sexual activity, situation, or partner.
Vyleesi was studied in two 24-week, randomized, double-blind, placebo-controlled trials in 1,247 premenopausal women with acquired, generalized HSDD. The women used Vyleesi two or three times per month and no more than once a week. About one-quarter of patients treated with Vyleesi had an increase of 1.2 or more in their sexual desire score (scored on a range of 1.2 to 6.0, with higher scores indicating greater sexual desire), compared with about 17% of those who took placebo. About 35% of the patients treated with Vyleesi had a decrease of one or more in their distress score (scored on a range of 0-4, with higher scores indicating greater distress from low sexual desire) compared with about 31% of those who took placebo.
The drug is injected under the skin of the abdomen or thigh at least 45 minutes before anticipated sexual activity. Patients may decide the optimal time to use Vyleesi based on the duration of benefit and any side effects, such as nausea. Patients should not take more than one dose of Vyleesi within 24 hours, or more than eight doses per month. Patients should discontinue treatment after 8 weeks if they do not report an improvement in sexual desire and associated distress.
Vyleesi works by activating melanocortin receptors but the exact mechanism for improving sexual desire is unknown. Some side effects were reported. “The most common side effects of Vyleesi are nausea and vomiting, flushing, injection site reactions, and headache. About 40% of patients in the clinical trials experienced nausea, most commonly with the first Vyleesi injection, and 13% needed medications for the treatment of nausea. About 1% of patients treated with Vyleesi in the clinical trials reported darkening of the gums and parts of the skin, including the face and breasts, which did not go away in about half the patients after stopping treatment. Patients with dark skin were more likely to develop this side effect,” according to the press release.
A temporary increase in blood pressure in patients after dosing with Vyleesi was observed during the clinical trials and therefore the drug is not recommended in patients at high risk for cardiovascular disease. In addition, patients who take a naltrexone-containing medication by mouth to treat alcohol or opioid dependence should not use Vyleesi because it may significantly decrease the levels of naltrexone in the blood and could lead to naltrexone treatment failure.
The full press release can be found on the FDA website.
The .
“There are women who, for no known reason, have reduced sexual desire that causes marked distress, and who can benefit from safe and effective pharmacologic treatment,” Hylton V. Joffe, MD, director of the Center for Drug Evaluation and Research’s Division of Bone, Reproductive, and Urologic Products, stated in a press release. “Today’s approval provides women with another treatment option for this condition.”
HSDD is characterized by low sexual desire that causes marked distress or interpersonal difficulty and is not caused by a medical or psychiatric condition. Acquired HSDD develops in a patient who previously experienced no problems with sexual desire, and generalized HSDD is a lack of desire that occurs regardless of the type of sexual activity, situation, or partner.
Vyleesi was studied in two 24-week, randomized, double-blind, placebo-controlled trials in 1,247 premenopausal women with acquired, generalized HSDD. The women used Vyleesi two or three times per month and no more than once a week. About one-quarter of patients treated with Vyleesi had an increase of 1.2 or more in their sexual desire score (scored on a range of 1.2 to 6.0, with higher scores indicating greater sexual desire), compared with about 17% of those who took placebo. About 35% of the patients treated with Vyleesi had a decrease of one or more in their distress score (scored on a range of 0-4, with higher scores indicating greater distress from low sexual desire) compared with about 31% of those who took placebo.
The drug is injected under the skin of the abdomen or thigh at least 45 minutes before anticipated sexual activity. Patients may decide the optimal time to use Vyleesi based on the duration of benefit and any side effects, such as nausea. Patients should not take more than one dose of Vyleesi within 24 hours, or more than eight doses per month. Patients should discontinue treatment after 8 weeks if they do not report an improvement in sexual desire and associated distress.
Vyleesi works by activating melanocortin receptors but the exact mechanism for improving sexual desire is unknown. Some side effects were reported. “The most common side effects of Vyleesi are nausea and vomiting, flushing, injection site reactions, and headache. About 40% of patients in the clinical trials experienced nausea, most commonly with the first Vyleesi injection, and 13% needed medications for the treatment of nausea. About 1% of patients treated with Vyleesi in the clinical trials reported darkening of the gums and parts of the skin, including the face and breasts, which did not go away in about half the patients after stopping treatment. Patients with dark skin were more likely to develop this side effect,” according to the press release.
A temporary increase in blood pressure in patients after dosing with Vyleesi was observed during the clinical trials and therefore the drug is not recommended in patients at high risk for cardiovascular disease. In addition, patients who take a naltrexone-containing medication by mouth to treat alcohol or opioid dependence should not use Vyleesi because it may significantly decrease the levels of naltrexone in the blood and could lead to naltrexone treatment failure.
The full press release can be found on the FDA website.
FDA grants marketing clearance for chlamydia and gonorrhea extragenital tests
The new tests, the Aptima Combo 2 Assay and Xpert CT/NG, use samples from the throat and rectum to test for chlamydia and gonorrhea, according to a statement from the FDA.
“It is best for patients if both [chlamydia and gonorrhea] are caught and treated right away, as significant complications can occur if left untreated,” noted Tim Stenzel, MD, in the statement.
“Today’s clearances provide a mechanism for more easily diagnosing these infections,” said Dr. Stenzel, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
The two tests were reviewed through the premarket notification – or 510(k) – pathway, which seeks to demonstrate to the FDA that the device to be marketed is equivalent or better in safety and effectiveness to the legally marketed device.
In the FDA’s evaluation of the tests, it reviewed clinical data from a multisite study of more than 2,500 patients. This study evaluated the diagnostic accuracy of multiple commercially available nucleic acid amplification tests for detection of Neisseria gonorrhoeae and Chlamydia trachomatis from throat and rectal sites. The results of this study and other information reviewed by the FDA demonstrated that the two tests “are safe and effective for extragenital testing for chlamydia and gonorrhea,” according to the statement.
The data were collected through a cross-sectional study coordinated by the Antibacterial Resistance Leadership Group, which is funded and supported by the National Institute of Allergy and Infectious Diseases.
The FDA granted marketing clearance to Hologic and Cepheid for the Aptima Combo 2 Assay and the Xpert CT/NG, respectively.
The new tests, the Aptima Combo 2 Assay and Xpert CT/NG, use samples from the throat and rectum to test for chlamydia and gonorrhea, according to a statement from the FDA.
“It is best for patients if both [chlamydia and gonorrhea] are caught and treated right away, as significant complications can occur if left untreated,” noted Tim Stenzel, MD, in the statement.
“Today’s clearances provide a mechanism for more easily diagnosing these infections,” said Dr. Stenzel, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
The two tests were reviewed through the premarket notification – or 510(k) – pathway, which seeks to demonstrate to the FDA that the device to be marketed is equivalent or better in safety and effectiveness to the legally marketed device.
In the FDA’s evaluation of the tests, it reviewed clinical data from a multisite study of more than 2,500 patients. This study evaluated the diagnostic accuracy of multiple commercially available nucleic acid amplification tests for detection of Neisseria gonorrhoeae and Chlamydia trachomatis from throat and rectal sites. The results of this study and other information reviewed by the FDA demonstrated that the two tests “are safe and effective for extragenital testing for chlamydia and gonorrhea,” according to the statement.
The data were collected through a cross-sectional study coordinated by the Antibacterial Resistance Leadership Group, which is funded and supported by the National Institute of Allergy and Infectious Diseases.
The FDA granted marketing clearance to Hologic and Cepheid for the Aptima Combo 2 Assay and the Xpert CT/NG, respectively.
The new tests, the Aptima Combo 2 Assay and Xpert CT/NG, use samples from the throat and rectum to test for chlamydia and gonorrhea, according to a statement from the FDA.
“It is best for patients if both [chlamydia and gonorrhea] are caught and treated right away, as significant complications can occur if left untreated,” noted Tim Stenzel, MD, in the statement.
“Today’s clearances provide a mechanism for more easily diagnosing these infections,” said Dr. Stenzel, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
The two tests were reviewed through the premarket notification – or 510(k) – pathway, which seeks to demonstrate to the FDA that the device to be marketed is equivalent or better in safety and effectiveness to the legally marketed device.
In the FDA’s evaluation of the tests, it reviewed clinical data from a multisite study of more than 2,500 patients. This study evaluated the diagnostic accuracy of multiple commercially available nucleic acid amplification tests for detection of Neisseria gonorrhoeae and Chlamydia trachomatis from throat and rectal sites. The results of this study and other information reviewed by the FDA demonstrated that the two tests “are safe and effective for extragenital testing for chlamydia and gonorrhea,” according to the statement.
The data were collected through a cross-sectional study coordinated by the Antibacterial Resistance Leadership Group, which is funded and supported by the National Institute of Allergy and Infectious Diseases.
The FDA granted marketing clearance to Hologic and Cepheid for the Aptima Combo 2 Assay and the Xpert CT/NG, respectively.