User login
Diagnosing and treating bipolar disorder in primary care
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
KEY POINTS
- Bipolar depression in its manifest and subthreshold forms is nearly as prevalent as unipolar depression and often occurs in successful professionals.
- A manic or hypomanic episode can make a patient highly productive, but it can also be severely disruptive, leading to loss of job, marriage, and financial savings.
- Identifying bipolar depression depends on asking about bipolar symptoms, using screening instruments, and being aware of clues from the patient’s history.
- A major depressive episode in patients with a history of mania or hypomania should be treated with a combination of an antidepressant and a mood stabilizer or a mood stabilizer alone.
How to prescribe effectively for opioid use disorder
NEW ORLEANS – Physicians committed to fighting the national opioid epidemic really need to take the 8-hour training course on addiction treatment required to obtain a Drug Enforcement Administration ‘X’ number, because it will enable them to prescribe buprenorphine, a drug with unique advantages for many affected patients, Ellie Grossman, MD, asserted at the annual meeting of the American College of Physicians.
Buprenorphine (Subutex) is one of the three medications approved for treatment of opioid use disorder (OUD), along with methadone and naltrexone (Revia). And for certain patients, it’s clearly the best choice, according to Dr. Grossman, a general internist at Harvard Medical School, Boston, and the primary care lead for behavioral health integration at the Cambridge (Mass.) Health Alliance.
The DEA X number certification process, which entails obtaining a waiver through SAMHSA – the Substance Abuse and Mental Health Services Administration – is bureaucratic. It’s unpopular with many physicians. But it’s well worth 8 hours of an internist’s time to get the waiver and gain the ability to prescribe buprenorphine.
“The requirement is admittedly clunky, and many people have strong feelings about whether this is a regulation that should exist,” according to Dr. Grossman. “I myself didn’t need to have special training to prescribe methadone, a full opioid agonist that my patients could easily die from. But I did have to undergo an 8-hour training to prescribe buprenorphine, and it’s much harder to die from that drug.”
She addressed which of the three medications for OUD is the best fit in a given patient, the appropriate treatment duration, and the role of adjunctive counseling, which – spoiler alert – has been cast into question by the results of a major government-funded randomized trial.
Dr. Grossman’s overriding message: “You are saving lives by getting people on medication.”
Indeed, studies have shown that patients with OUD who receive no treatment have a sixfold increase in the standardized mortality ratio, compared with the general population. Contrast that with the less than 2-fold increased risk with medication-assisted treatment and roughly a 2.5-fold increased risk when medication is given short term to cover withdrawal and then tapered and discontinued.
Other documented benefits of long-term medication-assisted treatment of patients with OUD as described in a 2014 Cochrane review include reductions in injection drug use, crime days, HIV-related risk behaviors and seroconversion, and improved health and social functioning.
Of note, those well-documented benefits apply only to methadone, a full opioid agonist, and buprenorphine, a partial agonist, because those two drugs have been around long enough to generate long-term outcome data. Naltrexone, which has a completely different mechanism of action – it’s a full opioid antagonist – has not as of yet.
Individualizing medical therapy for OUD
Physicians can’t write a prescription for methadone. The drug must be administered at a certified opioid treatment program, or OTP, otherwise known as a methadone clinic. Those clinics are highly regulated at both the federal and state levels, with lots of minutia involved. Patient counseling and drug screening are required.
In contrast, a physician with a DEA X number can write a prescription for buprenorphine and have a patient fill it at a pharmacy. There is inherently less structure surrounding buprenorphine therapy than that of methadone, Dr. Grossman noted. There are no hard and fast rules about how often a physician has to see the patient or do drug screens or counseling. Buprenorphine is available as once-daily oral sublingual therapy and, more recently, in long-acting injectable and implantable formulations, although Dr. Grossman believes the jury is still out about how these nonoral agents are best utilized.
“I’m often asked, ‘Which is better, methadone or buprenorphine?’ Really, the answer is they’re both pretty darn good,” according to Dr. Grossman.
The Cochrane review concluded that, in the studies that have used real-world dosing – that is, higher doses than in the initial studies – high-dose buprenorphine and high-dose methadone have similar rates of retention in treatment.
“What I tell patients is that a lot hinges on the structure of the treatment delivery system,” Dr. Grossman said. “If it’s methadone, they’re going to the OTP every day. Some people need more structure; they need a set of eyes on them every day. Or if they are at high risk for medication diversion – for example, someone else in their household might want to steal their medications – going to a methadone program gets around that. Also, when somebody has been on methadone in the past and did well on it and wants to go on it again, I’m likely to say, ‘That sounds like a good fit.’”
Buprenorphine is a good option for patients who don’t require close, structured supervision. It has fewer drug interactions than does methadone and is less prone to cause QTc prolongation. Also, it’s a more realistic option for patients who live so far from an OTP that daily attendance is impractical. And ob.gyns increasingly favor buprenorphine, because the problem of neonatal abstinence syndrome is less severe than when mothers are on methadone.
As for extended-release naltrexone (Vivitrol), the pivotal double-blind Russian trial that won FDA approval for treatment of OUD showed a dramatic improvement in opioid-free weeks (Lancet. 2011 Apr 30;377[9776]:1506-13).
More recently, the 24-week, multicenter, open-label X:BOT trial randomized 570 U.S. patients with OUD to once-monthly extended-release naltrexone or daily sublingual buprenorphine-naloxone (Suboxone). The dropout rate was higher in the extended-release naltrexone arm because patients had to be opioid free for 2 weeks before starting on the opioid antagonist. As a practical matter, that can be difficult to achieve unless a patient has just been released from jail or prison. But the per-protocol relapse rates were similar (Lancet. 2018 Jan 27;391[10118]:309-18).
“Many people interpret this study as saying, with the right patient who can get into an opioid-free state or, if you inherit an opioid-free state, the choice between extended-release naltrexone and buprenorphine-naloxone may be a bit of a wash in terms of clinical effectiveness, as best we can detect,” Dr. Grossman explained. “That said, they’re very different experiences: One is a shot in your butt once a month, the other is something you put in your mouth once a day. Patients typically have a strong point of view regarding what they’re up for.”
Extended-release naltrexone doesn’t require a DEA waiver or attendance at an OTP. But it costs roughly $800 per injection, although many insurers do cover it after additional paperwork is completed. While Dr. Grossman does use extended-release naltrexone in her own practice, it comes with some baggage. The drug comes in a powder, which is mixed with a diluent in the office, creating a thick, frothy substance that’s slow to inject. It has to be kept refrigerated, then warmed up in time for the patient visit.
“If you live somewhere where there’s no OTP and you don’t have a DEA X number, and you have a patient with OUD who’s interested in extended-release naltrexone, it’s not crazy to think about,” she noted.
Duration of medical therapy
Study after study demonstrates that, when treatment stops, the risk of relapse goes up.
“We as health care providers are used to the mentality of chronic diseases, like diabetes, where you’re probably on medicine for the rest of your life,” Dr. Grossman said. “OUD is another chronic disease where you might have a patient on medication for the rest of their life, although you may not want to drum that into their head right up front. It’s kind of scary. I don’t usually talk that way with my diabetic patients when I give them their diagnosis. So, I don’t push it.
“But the reality is, to give them the best chance of health, they should be on medication for a good long time,” she added. “And that’s true for all of the OUD medications.”
The role of counseling
The best evidence of the utility of adjunctive counseling in the treatment of OUD comes from the landmark Prescription Opioid Addiction Treatment Study (POATS), a 653-patient multicenter trial conducted by the National Drug Abuse Treatment Network and funded by the National Institute on Drug Abuse. Participants were randomized to standard medical management including medication and a meeting with a physician every 1-2 weeks, or to standard therapy plus individual counseling with a trained substance use counselor.
To the surprise of many, given that SAMHSA guidance strongly recommends counseling and other forms of behavioral therapy, there was no difference in outcomes between the two groups (Drug Alcohol Depend. 2015 May 1;150:112-9).
Subsequent parsing of the POATS data showed that the subgroup of people who were using heroin rather than prescription pills and who actually attended at least 60% of their counseling sessions did better than if they were randomized to no counseling.
“There’s still room for quibbling about the study, but many people would say, ‘You know, it’s not a slam dunk that everybody needs counseling,’ ” the internist commented.
“So, how do we pick the right treatment for our patients with OUD? It’s what feels right for them,” Dr. Grossman cautioned. “This gets back to what we do every day in managing chronic diseases: We nudge, we encourage, we use our motivational interviewing skills to help people figure out how they can change their lives and get healthier. There’s a long list of things going on in our patients’ lives that are going to help guide that decision.
“The message here: Medication is better than no medication, but it’s not a slam dunk which medication or how,” she concluded.
NEW ORLEANS – Physicians committed to fighting the national opioid epidemic really need to take the 8-hour training course on addiction treatment required to obtain a Drug Enforcement Administration ‘X’ number, because it will enable them to prescribe buprenorphine, a drug with unique advantages for many affected patients, Ellie Grossman, MD, asserted at the annual meeting of the American College of Physicians.
Buprenorphine (Subutex) is one of the three medications approved for treatment of opioid use disorder (OUD), along with methadone and naltrexone (Revia). And for certain patients, it’s clearly the best choice, according to Dr. Grossman, a general internist at Harvard Medical School, Boston, and the primary care lead for behavioral health integration at the Cambridge (Mass.) Health Alliance.
The DEA X number certification process, which entails obtaining a waiver through SAMHSA – the Substance Abuse and Mental Health Services Administration – is bureaucratic. It’s unpopular with many physicians. But it’s well worth 8 hours of an internist’s time to get the waiver and gain the ability to prescribe buprenorphine.
“The requirement is admittedly clunky, and many people have strong feelings about whether this is a regulation that should exist,” according to Dr. Grossman. “I myself didn’t need to have special training to prescribe methadone, a full opioid agonist that my patients could easily die from. But I did have to undergo an 8-hour training to prescribe buprenorphine, and it’s much harder to die from that drug.”
She addressed which of the three medications for OUD is the best fit in a given patient, the appropriate treatment duration, and the role of adjunctive counseling, which – spoiler alert – has been cast into question by the results of a major government-funded randomized trial.
Dr. Grossman’s overriding message: “You are saving lives by getting people on medication.”
Indeed, studies have shown that patients with OUD who receive no treatment have a sixfold increase in the standardized mortality ratio, compared with the general population. Contrast that with the less than 2-fold increased risk with medication-assisted treatment and roughly a 2.5-fold increased risk when medication is given short term to cover withdrawal and then tapered and discontinued.
Other documented benefits of long-term medication-assisted treatment of patients with OUD as described in a 2014 Cochrane review include reductions in injection drug use, crime days, HIV-related risk behaviors and seroconversion, and improved health and social functioning.
Of note, those well-documented benefits apply only to methadone, a full opioid agonist, and buprenorphine, a partial agonist, because those two drugs have been around long enough to generate long-term outcome data. Naltrexone, which has a completely different mechanism of action – it’s a full opioid antagonist – has not as of yet.
Individualizing medical therapy for OUD
Physicians can’t write a prescription for methadone. The drug must be administered at a certified opioid treatment program, or OTP, otherwise known as a methadone clinic. Those clinics are highly regulated at both the federal and state levels, with lots of minutia involved. Patient counseling and drug screening are required.
In contrast, a physician with a DEA X number can write a prescription for buprenorphine and have a patient fill it at a pharmacy. There is inherently less structure surrounding buprenorphine therapy than that of methadone, Dr. Grossman noted. There are no hard and fast rules about how often a physician has to see the patient or do drug screens or counseling. Buprenorphine is available as once-daily oral sublingual therapy and, more recently, in long-acting injectable and implantable formulations, although Dr. Grossman believes the jury is still out about how these nonoral agents are best utilized.
“I’m often asked, ‘Which is better, methadone or buprenorphine?’ Really, the answer is they’re both pretty darn good,” according to Dr. Grossman.
The Cochrane review concluded that, in the studies that have used real-world dosing – that is, higher doses than in the initial studies – high-dose buprenorphine and high-dose methadone have similar rates of retention in treatment.
“What I tell patients is that a lot hinges on the structure of the treatment delivery system,” Dr. Grossman said. “If it’s methadone, they’re going to the OTP every day. Some people need more structure; they need a set of eyes on them every day. Or if they are at high risk for medication diversion – for example, someone else in their household might want to steal their medications – going to a methadone program gets around that. Also, when somebody has been on methadone in the past and did well on it and wants to go on it again, I’m likely to say, ‘That sounds like a good fit.’”
Buprenorphine is a good option for patients who don’t require close, structured supervision. It has fewer drug interactions than does methadone and is less prone to cause QTc prolongation. Also, it’s a more realistic option for patients who live so far from an OTP that daily attendance is impractical. And ob.gyns increasingly favor buprenorphine, because the problem of neonatal abstinence syndrome is less severe than when mothers are on methadone.
As for extended-release naltrexone (Vivitrol), the pivotal double-blind Russian trial that won FDA approval for treatment of OUD showed a dramatic improvement in opioid-free weeks (Lancet. 2011 Apr 30;377[9776]:1506-13).
More recently, the 24-week, multicenter, open-label X:BOT trial randomized 570 U.S. patients with OUD to once-monthly extended-release naltrexone or daily sublingual buprenorphine-naloxone (Suboxone). The dropout rate was higher in the extended-release naltrexone arm because patients had to be opioid free for 2 weeks before starting on the opioid antagonist. As a practical matter, that can be difficult to achieve unless a patient has just been released from jail or prison. But the per-protocol relapse rates were similar (Lancet. 2018 Jan 27;391[10118]:309-18).
“Many people interpret this study as saying, with the right patient who can get into an opioid-free state or, if you inherit an opioid-free state, the choice between extended-release naltrexone and buprenorphine-naloxone may be a bit of a wash in terms of clinical effectiveness, as best we can detect,” Dr. Grossman explained. “That said, they’re very different experiences: One is a shot in your butt once a month, the other is something you put in your mouth once a day. Patients typically have a strong point of view regarding what they’re up for.”
Extended-release naltrexone doesn’t require a DEA waiver or attendance at an OTP. But it costs roughly $800 per injection, although many insurers do cover it after additional paperwork is completed. While Dr. Grossman does use extended-release naltrexone in her own practice, it comes with some baggage. The drug comes in a powder, which is mixed with a diluent in the office, creating a thick, frothy substance that’s slow to inject. It has to be kept refrigerated, then warmed up in time for the patient visit.
“If you live somewhere where there’s no OTP and you don’t have a DEA X number, and you have a patient with OUD who’s interested in extended-release naltrexone, it’s not crazy to think about,” she noted.
Duration of medical therapy
Study after study demonstrates that, when treatment stops, the risk of relapse goes up.
“We as health care providers are used to the mentality of chronic diseases, like diabetes, where you’re probably on medicine for the rest of your life,” Dr. Grossman said. “OUD is another chronic disease where you might have a patient on medication for the rest of their life, although you may not want to drum that into their head right up front. It’s kind of scary. I don’t usually talk that way with my diabetic patients when I give them their diagnosis. So, I don’t push it.
“But the reality is, to give them the best chance of health, they should be on medication for a good long time,” she added. “And that’s true for all of the OUD medications.”
The role of counseling
The best evidence of the utility of adjunctive counseling in the treatment of OUD comes from the landmark Prescription Opioid Addiction Treatment Study (POATS), a 653-patient multicenter trial conducted by the National Drug Abuse Treatment Network and funded by the National Institute on Drug Abuse. Participants were randomized to standard medical management including medication and a meeting with a physician every 1-2 weeks, or to standard therapy plus individual counseling with a trained substance use counselor.
To the surprise of many, given that SAMHSA guidance strongly recommends counseling and other forms of behavioral therapy, there was no difference in outcomes between the two groups (Drug Alcohol Depend. 2015 May 1;150:112-9).
Subsequent parsing of the POATS data showed that the subgroup of people who were using heroin rather than prescription pills and who actually attended at least 60% of their counseling sessions did better than if they were randomized to no counseling.
“There’s still room for quibbling about the study, but many people would say, ‘You know, it’s not a slam dunk that everybody needs counseling,’ ” the internist commented.
“So, how do we pick the right treatment for our patients with OUD? It’s what feels right for them,” Dr. Grossman cautioned. “This gets back to what we do every day in managing chronic diseases: We nudge, we encourage, we use our motivational interviewing skills to help people figure out how they can change their lives and get healthier. There’s a long list of things going on in our patients’ lives that are going to help guide that decision.
“The message here: Medication is better than no medication, but it’s not a slam dunk which medication or how,” she concluded.
NEW ORLEANS – Physicians committed to fighting the national opioid epidemic really need to take the 8-hour training course on addiction treatment required to obtain a Drug Enforcement Administration ‘X’ number, because it will enable them to prescribe buprenorphine, a drug with unique advantages for many affected patients, Ellie Grossman, MD, asserted at the annual meeting of the American College of Physicians.
Buprenorphine (Subutex) is one of the three medications approved for treatment of opioid use disorder (OUD), along with methadone and naltrexone (Revia). And for certain patients, it’s clearly the best choice, according to Dr. Grossman, a general internist at Harvard Medical School, Boston, and the primary care lead for behavioral health integration at the Cambridge (Mass.) Health Alliance.
The DEA X number certification process, which entails obtaining a waiver through SAMHSA – the Substance Abuse and Mental Health Services Administration – is bureaucratic. It’s unpopular with many physicians. But it’s well worth 8 hours of an internist’s time to get the waiver and gain the ability to prescribe buprenorphine.
“The requirement is admittedly clunky, and many people have strong feelings about whether this is a regulation that should exist,” according to Dr. Grossman. “I myself didn’t need to have special training to prescribe methadone, a full opioid agonist that my patients could easily die from. But I did have to undergo an 8-hour training to prescribe buprenorphine, and it’s much harder to die from that drug.”
She addressed which of the three medications for OUD is the best fit in a given patient, the appropriate treatment duration, and the role of adjunctive counseling, which – spoiler alert – has been cast into question by the results of a major government-funded randomized trial.
Dr. Grossman’s overriding message: “You are saving lives by getting people on medication.”
Indeed, studies have shown that patients with OUD who receive no treatment have a sixfold increase in the standardized mortality ratio, compared with the general population. Contrast that with the less than 2-fold increased risk with medication-assisted treatment and roughly a 2.5-fold increased risk when medication is given short term to cover withdrawal and then tapered and discontinued.
Other documented benefits of long-term medication-assisted treatment of patients with OUD as described in a 2014 Cochrane review include reductions in injection drug use, crime days, HIV-related risk behaviors and seroconversion, and improved health and social functioning.
Of note, those well-documented benefits apply only to methadone, a full opioid agonist, and buprenorphine, a partial agonist, because those two drugs have been around long enough to generate long-term outcome data. Naltrexone, which has a completely different mechanism of action – it’s a full opioid antagonist – has not as of yet.
Individualizing medical therapy for OUD
Physicians can’t write a prescription for methadone. The drug must be administered at a certified opioid treatment program, or OTP, otherwise known as a methadone clinic. Those clinics are highly regulated at both the federal and state levels, with lots of minutia involved. Patient counseling and drug screening are required.
In contrast, a physician with a DEA X number can write a prescription for buprenorphine and have a patient fill it at a pharmacy. There is inherently less structure surrounding buprenorphine therapy than that of methadone, Dr. Grossman noted. There are no hard and fast rules about how often a physician has to see the patient or do drug screens or counseling. Buprenorphine is available as once-daily oral sublingual therapy and, more recently, in long-acting injectable and implantable formulations, although Dr. Grossman believes the jury is still out about how these nonoral agents are best utilized.
“I’m often asked, ‘Which is better, methadone or buprenorphine?’ Really, the answer is they’re both pretty darn good,” according to Dr. Grossman.
The Cochrane review concluded that, in the studies that have used real-world dosing – that is, higher doses than in the initial studies – high-dose buprenorphine and high-dose methadone have similar rates of retention in treatment.
“What I tell patients is that a lot hinges on the structure of the treatment delivery system,” Dr. Grossman said. “If it’s methadone, they’re going to the OTP every day. Some people need more structure; they need a set of eyes on them every day. Or if they are at high risk for medication diversion – for example, someone else in their household might want to steal their medications – going to a methadone program gets around that. Also, when somebody has been on methadone in the past and did well on it and wants to go on it again, I’m likely to say, ‘That sounds like a good fit.’”
Buprenorphine is a good option for patients who don’t require close, structured supervision. It has fewer drug interactions than does methadone and is less prone to cause QTc prolongation. Also, it’s a more realistic option for patients who live so far from an OTP that daily attendance is impractical. And ob.gyns increasingly favor buprenorphine, because the problem of neonatal abstinence syndrome is less severe than when mothers are on methadone.
As for extended-release naltrexone (Vivitrol), the pivotal double-blind Russian trial that won FDA approval for treatment of OUD showed a dramatic improvement in opioid-free weeks (Lancet. 2011 Apr 30;377[9776]:1506-13).
More recently, the 24-week, multicenter, open-label X:BOT trial randomized 570 U.S. patients with OUD to once-monthly extended-release naltrexone or daily sublingual buprenorphine-naloxone (Suboxone). The dropout rate was higher in the extended-release naltrexone arm because patients had to be opioid free for 2 weeks before starting on the opioid antagonist. As a practical matter, that can be difficult to achieve unless a patient has just been released from jail or prison. But the per-protocol relapse rates were similar (Lancet. 2018 Jan 27;391[10118]:309-18).
“Many people interpret this study as saying, with the right patient who can get into an opioid-free state or, if you inherit an opioid-free state, the choice between extended-release naltrexone and buprenorphine-naloxone may be a bit of a wash in terms of clinical effectiveness, as best we can detect,” Dr. Grossman explained. “That said, they’re very different experiences: One is a shot in your butt once a month, the other is something you put in your mouth once a day. Patients typically have a strong point of view regarding what they’re up for.”
Extended-release naltrexone doesn’t require a DEA waiver or attendance at an OTP. But it costs roughly $800 per injection, although many insurers do cover it after additional paperwork is completed. While Dr. Grossman does use extended-release naltrexone in her own practice, it comes with some baggage. The drug comes in a powder, which is mixed with a diluent in the office, creating a thick, frothy substance that’s slow to inject. It has to be kept refrigerated, then warmed up in time for the patient visit.
“If you live somewhere where there’s no OTP and you don’t have a DEA X number, and you have a patient with OUD who’s interested in extended-release naltrexone, it’s not crazy to think about,” she noted.
Duration of medical therapy
Study after study demonstrates that, when treatment stops, the risk of relapse goes up.
“We as health care providers are used to the mentality of chronic diseases, like diabetes, where you’re probably on medicine for the rest of your life,” Dr. Grossman said. “OUD is another chronic disease where you might have a patient on medication for the rest of their life, although you may not want to drum that into their head right up front. It’s kind of scary. I don’t usually talk that way with my diabetic patients when I give them their diagnosis. So, I don’t push it.
“But the reality is, to give them the best chance of health, they should be on medication for a good long time,” she added. “And that’s true for all of the OUD medications.”
The role of counseling
The best evidence of the utility of adjunctive counseling in the treatment of OUD comes from the landmark Prescription Opioid Addiction Treatment Study (POATS), a 653-patient multicenter trial conducted by the National Drug Abuse Treatment Network and funded by the National Institute on Drug Abuse. Participants were randomized to standard medical management including medication and a meeting with a physician every 1-2 weeks, or to standard therapy plus individual counseling with a trained substance use counselor.
To the surprise of many, given that SAMHSA guidance strongly recommends counseling and other forms of behavioral therapy, there was no difference in outcomes between the two groups (Drug Alcohol Depend. 2015 May 1;150:112-9).
Subsequent parsing of the POATS data showed that the subgroup of people who were using heroin rather than prescription pills and who actually attended at least 60% of their counseling sessions did better than if they were randomized to no counseling.
“There’s still room for quibbling about the study, but many people would say, ‘You know, it’s not a slam dunk that everybody needs counseling,’ ” the internist commented.
“So, how do we pick the right treatment for our patients with OUD? It’s what feels right for them,” Dr. Grossman cautioned. “This gets back to what we do every day in managing chronic diseases: We nudge, we encourage, we use our motivational interviewing skills to help people figure out how they can change their lives and get healthier. There’s a long list of things going on in our patients’ lives that are going to help guide that decision.
“The message here: Medication is better than no medication, but it’s not a slam dunk which medication or how,” she concluded.
REPORTING FROM ACP INTERNAL MEDICINE
National Academies issues 5-step plan to address infections linked to opioid use disorder
Widespread opioid use disorder (OUD) has spawned new epidemics of hepatitis C virus (HCV) and HIV infections as well as increased hospitalizations for bacteremia, endocarditis, skin and soft tissue infections, and osteomyelitis, according to a report arising from a National Academies of Science, Engineering and Medicine (NASEM) workshop titled Integrating Infectious Disease Considerations with Response to the Opioid Epidemic.
Optimal treatment of these infections is often impeded by untreated OUD, Sandra A. Springer, MD, and her colleagues wrote in an article published online in the Annals of Internal Medicine. Failing to address OUD can result in longer hospital stays; frequent readmissions because of a lack of adherence to antibiotic regimens; or reinfection, morbidity, and high costs. “Medical settings that manage such infections offer a potential means of engaging people in treatment of OUD; however, few providers and hospitals treating such infections have the needed resources and capabilities,” Dr. Springer, director, infectious disease outpatient clinic, Veterans Administration, Newington, and of Yale University, New Haven, both in Conn., and her colleagues wrote.
The authors outlined five action steps resulting from the NASEM workshop:
- Implement screening for OUD in all relevant health care settings.
- For patients with positive screening results, immediately prescribe effective medication for OUD and/or opioid withdrawal symptoms.
- Develop hospital-based protocols that facilitate OUD treatment initiation and linkage to community-based treatment upon discharge.
- Hospitals, medical schools, physician assistant schools, nursing schools, and residency programs should increase training to identify and treat OUD.
- Increase access to addiction care and funding to states to provide effective medications to treat OUD.
Opioid withdrawal and pain syndromes should be addressed with opioid agonist therapies to optimize infectious disease (ID) treatment and relieve pain, according to Dr. Springer and her colleagues. In addition, “Because ID specialists are likely to be consulted for anyone requiring long-term antibiotic therapy or patients with HIV and HCV infection, OUD screening should be a standard part of an ID consult assessment,” the authors wrote.
“All health care providers have a role in combating the OUD epidemic and its ID consequences. Those who treat infectious complications of OUD are well suited to screen for OUD and begin treatment with effective FDA-approved medications,” the authors concluded.
The workshop was held in March 2018 in Washington and videos and slide presentations from the meeting are available.
Dr. Springer and her colleagues reported grant funding from the National Institutes of Health, but no commercial conflicts.
SOURCE: Springer SA et al. Ann Intern Med. 2018 Jul 13. doi: 10.7326/M18-1203.
Widespread opioid use disorder (OUD) has spawned new epidemics of hepatitis C virus (HCV) and HIV infections as well as increased hospitalizations for bacteremia, endocarditis, skin and soft tissue infections, and osteomyelitis, according to a report arising from a National Academies of Science, Engineering and Medicine (NASEM) workshop titled Integrating Infectious Disease Considerations with Response to the Opioid Epidemic.
Optimal treatment of these infections is often impeded by untreated OUD, Sandra A. Springer, MD, and her colleagues wrote in an article published online in the Annals of Internal Medicine. Failing to address OUD can result in longer hospital stays; frequent readmissions because of a lack of adherence to antibiotic regimens; or reinfection, morbidity, and high costs. “Medical settings that manage such infections offer a potential means of engaging people in treatment of OUD; however, few providers and hospitals treating such infections have the needed resources and capabilities,” Dr. Springer, director, infectious disease outpatient clinic, Veterans Administration, Newington, and of Yale University, New Haven, both in Conn., and her colleagues wrote.
The authors outlined five action steps resulting from the NASEM workshop:
- Implement screening for OUD in all relevant health care settings.
- For patients with positive screening results, immediately prescribe effective medication for OUD and/or opioid withdrawal symptoms.
- Develop hospital-based protocols that facilitate OUD treatment initiation and linkage to community-based treatment upon discharge.
- Hospitals, medical schools, physician assistant schools, nursing schools, and residency programs should increase training to identify and treat OUD.
- Increase access to addiction care and funding to states to provide effective medications to treat OUD.
Opioid withdrawal and pain syndromes should be addressed with opioid agonist therapies to optimize infectious disease (ID) treatment and relieve pain, according to Dr. Springer and her colleagues. In addition, “Because ID specialists are likely to be consulted for anyone requiring long-term antibiotic therapy or patients with HIV and HCV infection, OUD screening should be a standard part of an ID consult assessment,” the authors wrote.
“All health care providers have a role in combating the OUD epidemic and its ID consequences. Those who treat infectious complications of OUD are well suited to screen for OUD and begin treatment with effective FDA-approved medications,” the authors concluded.
The workshop was held in March 2018 in Washington and videos and slide presentations from the meeting are available.
Dr. Springer and her colleagues reported grant funding from the National Institutes of Health, but no commercial conflicts.
SOURCE: Springer SA et al. Ann Intern Med. 2018 Jul 13. doi: 10.7326/M18-1203.
Widespread opioid use disorder (OUD) has spawned new epidemics of hepatitis C virus (HCV) and HIV infections as well as increased hospitalizations for bacteremia, endocarditis, skin and soft tissue infections, and osteomyelitis, according to a report arising from a National Academies of Science, Engineering and Medicine (NASEM) workshop titled Integrating Infectious Disease Considerations with Response to the Opioid Epidemic.
Optimal treatment of these infections is often impeded by untreated OUD, Sandra A. Springer, MD, and her colleagues wrote in an article published online in the Annals of Internal Medicine. Failing to address OUD can result in longer hospital stays; frequent readmissions because of a lack of adherence to antibiotic regimens; or reinfection, morbidity, and high costs. “Medical settings that manage such infections offer a potential means of engaging people in treatment of OUD; however, few providers and hospitals treating such infections have the needed resources and capabilities,” Dr. Springer, director, infectious disease outpatient clinic, Veterans Administration, Newington, and of Yale University, New Haven, both in Conn., and her colleagues wrote.
The authors outlined five action steps resulting from the NASEM workshop:
- Implement screening for OUD in all relevant health care settings.
- For patients with positive screening results, immediately prescribe effective medication for OUD and/or opioid withdrawal symptoms.
- Develop hospital-based protocols that facilitate OUD treatment initiation and linkage to community-based treatment upon discharge.
- Hospitals, medical schools, physician assistant schools, nursing schools, and residency programs should increase training to identify and treat OUD.
- Increase access to addiction care and funding to states to provide effective medications to treat OUD.
Opioid withdrawal and pain syndromes should be addressed with opioid agonist therapies to optimize infectious disease (ID) treatment and relieve pain, according to Dr. Springer and her colleagues. In addition, “Because ID specialists are likely to be consulted for anyone requiring long-term antibiotic therapy or patients with HIV and HCV infection, OUD screening should be a standard part of an ID consult assessment,” the authors wrote.
“All health care providers have a role in combating the OUD epidemic and its ID consequences. Those who treat infectious complications of OUD are well suited to screen for OUD and begin treatment with effective FDA-approved medications,” the authors concluded.
The workshop was held in March 2018 in Washington and videos and slide presentations from the meeting are available.
Dr. Springer and her colleagues reported grant funding from the National Institutes of Health, but no commercial conflicts.
SOURCE: Springer SA et al. Ann Intern Med. 2018 Jul 13. doi: 10.7326/M18-1203.
FROM ANNALS OF INTERNAL MEDICINE
Usability and Patient Perceptions of the Sarilumab Pen for Treatment of RA
Study Overview
Objective. To assess usability and patient perceptions of the sarilumab auto-injector device (“sarilumab pen”) among patients with moderate-to-severe rheumatoid arthritis (RA).
Design. 12-week, randomized, parallel-group usability study.
Setting and participants. The study was conducted at 53 centers in 6 countries. Inclusion criteria were a diagnosis of RA (as defined by American College of Rheumatology/ European League Against Rheumatism 2010 Criteria) of ≥ 3-month disease duration, willing and able to self inject, continuous treatment with 1 or a combination of nonbiologic disease modifying antirheumatic drugs (except leflunomide in combination with methotrexate); and moderatly to severely active RA, defined as 4/66 swollen joint, 4/68 tender joints, and high-sensitivity C-reactive protein (hsCRP) measurement ≥ 4 mg/L. Exclusion criteria were age
Patients were randomized 1:1:1:1 to sarilumamb 150 or 200 mg every 2 weeks administered by single-use, disposable, prefilled pen or pre-filled syringe. Randomization method was not reported.
Main outcomes measures. The primary endpoint was number of “product technical failures” (PTFs). Patients randomized to the pen were given a diary that had questions related to their ability to remove the cap, start the injection, and complete the injection. Participants were asked to answer the questions each time they used the pen. If the response was “no” to any of the 3 questions, this was considered a “product technical complaint” (PTC). PTCs that had a validated technical cause based on pen evaluation and analysis were considered PTFs.
In addition, patient perceptions and satisfaction with the pen were assessed via questionnaire. At baseline, patients were asked about injections and prior experience with self-injection, and at 12 weeks they were asked about their experiences in using the pen. Other outcomes assessed included adverse events and pharmokinetic parameters.
Results. 217 participants were enrolled: 108 patients were in the pen group (56 randomized to 150 mg and 52 randomized to 200 mg) and 109 were in the syringe group (53 randomized to 150 mg and 56 randomized to 200 mg). Completion rates were similar among groups. Sixteen patients discontinued due to treatment-emergent adverse events. There were no PTFs. There was one PTC, in which the user accidently bumped the pen, which expelled the drug onto the floor.
At baseline, before the first injection, the majority of patients reported that they were not afraid of needles (58%), had past experience with self-injections (55%), and were either “very confident” or “extremely confident” regarding self-injections (55%). After the 12-week assessment phase, when asked about their overall level of satisfaction, 98% of patients reported they were “satisfied” or “very satisfied” with the sarilumab pen.
Treatment emergent adverse events occurred in 66% of patients, with no clinically meaningful differences leading to discontinuation in the pen and syringe groups. The most common adverse events were infections and neutropenia.
Conclusion. Patients successfully completed self-injections with the sarilumab pen and found it easy to use.
Commentary
Rheumatoid arthritis (RA) is a common immune-mediated disease characterized by chronically progressive inflammation and destruction of joints and associated structures, resulting in significant morbidity, mortality, and disability. Improved understanding of RA disease pathogenesis in recent years has led to the development of new biologic treatments designed to target specific elements of the RA inflammatory response.
Sarilumab is an interleukin-6 blocker that was approved in the US in 2017 for the treatment of adult patients with moderately to severely active RA who have had an inadequate response or intolerance to one or more disease-modifying antirheumatic drugs. While a syringe form of this drug is currently available, at the time of this writing the pen has not yet been released.
In this real-world usability study sponsored by Sanofi, there were no technical difficulties with using the pen. Most patients thought the pen was easy or very easy to use, and safety and effeicacy appeared to be generally comparable between the pen and syringe. The pen also offers safety protection features that prevent needlestick injury.
The authors of the current study noted that results from previous studies have shown that patients with RA favor treatment devices that are easy to use, convenient, less painful, and take less time to use, and patients have demonstrated a preference for autoinjector devices over more conventional methods of treatment administration [1–3], such as syringes. Pens have been well accepted for the treatment of other chronic health conditions, including diabetes mellitus, migraine headaches, and growth hormone deficiency, and subcutaneous administration of a tumor necrosis factor (TNF) inhibitor via pen has also been accepted for the treatment of RA [1]. As RA requires lifelong treatment, the use of a pen that is ergonomically designed to take into account the manual dexterity issues relevant to this patient population could potentially enhance compliance.
Applications for Clinical Practice
A prefilled pen was well accepted and associated with favorable patient perceptions,
1. Kivitz A, Cohen S, Dowd JE, et al. Clinical assessment of pain, tolerability, and preference of an autoinjection pen versus a prefilled syringe for patient self-administration of the fully human, monoclonal antibody adalimumab: the TOUCH trial. Clin Ther 2006;28:1619–29.
2. Demary W, Schwenke H, Rockwitz K, et al. Subcutaneously administered methotrexate for rheumatoid arthritis, by prefilled syringes versus prefilled pens: patient preference and comparison of the self-injection experience. Patient Prefer Adherence 2014;8:1061–71.
3. Thakur K, Biberger A, Handrich A, Rezk MF. Patient perceptions and preferences of two etanercept autoinjectors for rheumatoid arthritis: findings from a patient survey in Europe. Rheumatol Ther 2016;3:245–56.
Study Overview
Objective. To assess usability and patient perceptions of the sarilumab auto-injector device (“sarilumab pen”) among patients with moderate-to-severe rheumatoid arthritis (RA).
Design. 12-week, randomized, parallel-group usability study.
Setting and participants. The study was conducted at 53 centers in 6 countries. Inclusion criteria were a diagnosis of RA (as defined by American College of Rheumatology/ European League Against Rheumatism 2010 Criteria) of ≥ 3-month disease duration, willing and able to self inject, continuous treatment with 1 or a combination of nonbiologic disease modifying antirheumatic drugs (except leflunomide in combination with methotrexate); and moderatly to severely active RA, defined as 4/66 swollen joint, 4/68 tender joints, and high-sensitivity C-reactive protein (hsCRP) measurement ≥ 4 mg/L. Exclusion criteria were age
Patients were randomized 1:1:1:1 to sarilumamb 150 or 200 mg every 2 weeks administered by single-use, disposable, prefilled pen or pre-filled syringe. Randomization method was not reported.
Main outcomes measures. The primary endpoint was number of “product technical failures” (PTFs). Patients randomized to the pen were given a diary that had questions related to their ability to remove the cap, start the injection, and complete the injection. Participants were asked to answer the questions each time they used the pen. If the response was “no” to any of the 3 questions, this was considered a “product technical complaint” (PTC). PTCs that had a validated technical cause based on pen evaluation and analysis were considered PTFs.
In addition, patient perceptions and satisfaction with the pen were assessed via questionnaire. At baseline, patients were asked about injections and prior experience with self-injection, and at 12 weeks they were asked about their experiences in using the pen. Other outcomes assessed included adverse events and pharmokinetic parameters.
Results. 217 participants were enrolled: 108 patients were in the pen group (56 randomized to 150 mg and 52 randomized to 200 mg) and 109 were in the syringe group (53 randomized to 150 mg and 56 randomized to 200 mg). Completion rates were similar among groups. Sixteen patients discontinued due to treatment-emergent adverse events. There were no PTFs. There was one PTC, in which the user accidently bumped the pen, which expelled the drug onto the floor.
At baseline, before the first injection, the majority of patients reported that they were not afraid of needles (58%), had past experience with self-injections (55%), and were either “very confident” or “extremely confident” regarding self-injections (55%). After the 12-week assessment phase, when asked about their overall level of satisfaction, 98% of patients reported they were “satisfied” or “very satisfied” with the sarilumab pen.
Treatment emergent adverse events occurred in 66% of patients, with no clinically meaningful differences leading to discontinuation in the pen and syringe groups. The most common adverse events were infections and neutropenia.
Conclusion. Patients successfully completed self-injections with the sarilumab pen and found it easy to use.
Commentary
Rheumatoid arthritis (RA) is a common immune-mediated disease characterized by chronically progressive inflammation and destruction of joints and associated structures, resulting in significant morbidity, mortality, and disability. Improved understanding of RA disease pathogenesis in recent years has led to the development of new biologic treatments designed to target specific elements of the RA inflammatory response.
Sarilumab is an interleukin-6 blocker that was approved in the US in 2017 for the treatment of adult patients with moderately to severely active RA who have had an inadequate response or intolerance to one or more disease-modifying antirheumatic drugs. While a syringe form of this drug is currently available, at the time of this writing the pen has not yet been released.
In this real-world usability study sponsored by Sanofi, there were no technical difficulties with using the pen. Most patients thought the pen was easy or very easy to use, and safety and effeicacy appeared to be generally comparable between the pen and syringe. The pen also offers safety protection features that prevent needlestick injury.
The authors of the current study noted that results from previous studies have shown that patients with RA favor treatment devices that are easy to use, convenient, less painful, and take less time to use, and patients have demonstrated a preference for autoinjector devices over more conventional methods of treatment administration [1–3], such as syringes. Pens have been well accepted for the treatment of other chronic health conditions, including diabetes mellitus, migraine headaches, and growth hormone deficiency, and subcutaneous administration of a tumor necrosis factor (TNF) inhibitor via pen has also been accepted for the treatment of RA [1]. As RA requires lifelong treatment, the use of a pen that is ergonomically designed to take into account the manual dexterity issues relevant to this patient population could potentially enhance compliance.
Applications for Clinical Practice
A prefilled pen was well accepted and associated with favorable patient perceptions,
Study Overview
Objective. To assess usability and patient perceptions of the sarilumab auto-injector device (“sarilumab pen”) among patients with moderate-to-severe rheumatoid arthritis (RA).
Design. 12-week, randomized, parallel-group usability study.
Setting and participants. The study was conducted at 53 centers in 6 countries. Inclusion criteria were a diagnosis of RA (as defined by American College of Rheumatology/ European League Against Rheumatism 2010 Criteria) of ≥ 3-month disease duration, willing and able to self inject, continuous treatment with 1 or a combination of nonbiologic disease modifying antirheumatic drugs (except leflunomide in combination with methotrexate); and moderatly to severely active RA, defined as 4/66 swollen joint, 4/68 tender joints, and high-sensitivity C-reactive protein (hsCRP) measurement ≥ 4 mg/L. Exclusion criteria were age
Patients were randomized 1:1:1:1 to sarilumamb 150 or 200 mg every 2 weeks administered by single-use, disposable, prefilled pen or pre-filled syringe. Randomization method was not reported.
Main outcomes measures. The primary endpoint was number of “product technical failures” (PTFs). Patients randomized to the pen were given a diary that had questions related to their ability to remove the cap, start the injection, and complete the injection. Participants were asked to answer the questions each time they used the pen. If the response was “no” to any of the 3 questions, this was considered a “product technical complaint” (PTC). PTCs that had a validated technical cause based on pen evaluation and analysis were considered PTFs.
In addition, patient perceptions and satisfaction with the pen were assessed via questionnaire. At baseline, patients were asked about injections and prior experience with self-injection, and at 12 weeks they were asked about their experiences in using the pen. Other outcomes assessed included adverse events and pharmokinetic parameters.
Results. 217 participants were enrolled: 108 patients were in the pen group (56 randomized to 150 mg and 52 randomized to 200 mg) and 109 were in the syringe group (53 randomized to 150 mg and 56 randomized to 200 mg). Completion rates were similar among groups. Sixteen patients discontinued due to treatment-emergent adverse events. There were no PTFs. There was one PTC, in which the user accidently bumped the pen, which expelled the drug onto the floor.
At baseline, before the first injection, the majority of patients reported that they were not afraid of needles (58%), had past experience with self-injections (55%), and were either “very confident” or “extremely confident” regarding self-injections (55%). After the 12-week assessment phase, when asked about their overall level of satisfaction, 98% of patients reported they were “satisfied” or “very satisfied” with the sarilumab pen.
Treatment emergent adverse events occurred in 66% of patients, with no clinically meaningful differences leading to discontinuation in the pen and syringe groups. The most common adverse events were infections and neutropenia.
Conclusion. Patients successfully completed self-injections with the sarilumab pen and found it easy to use.
Commentary
Rheumatoid arthritis (RA) is a common immune-mediated disease characterized by chronically progressive inflammation and destruction of joints and associated structures, resulting in significant morbidity, mortality, and disability. Improved understanding of RA disease pathogenesis in recent years has led to the development of new biologic treatments designed to target specific elements of the RA inflammatory response.
Sarilumab is an interleukin-6 blocker that was approved in the US in 2017 for the treatment of adult patients with moderately to severely active RA who have had an inadequate response or intolerance to one or more disease-modifying antirheumatic drugs. While a syringe form of this drug is currently available, at the time of this writing the pen has not yet been released.
In this real-world usability study sponsored by Sanofi, there were no technical difficulties with using the pen. Most patients thought the pen was easy or very easy to use, and safety and effeicacy appeared to be generally comparable between the pen and syringe. The pen also offers safety protection features that prevent needlestick injury.
The authors of the current study noted that results from previous studies have shown that patients with RA favor treatment devices that are easy to use, convenient, less painful, and take less time to use, and patients have demonstrated a preference for autoinjector devices over more conventional methods of treatment administration [1–3], such as syringes. Pens have been well accepted for the treatment of other chronic health conditions, including diabetes mellitus, migraine headaches, and growth hormone deficiency, and subcutaneous administration of a tumor necrosis factor (TNF) inhibitor via pen has also been accepted for the treatment of RA [1]. As RA requires lifelong treatment, the use of a pen that is ergonomically designed to take into account the manual dexterity issues relevant to this patient population could potentially enhance compliance.
Applications for Clinical Practice
A prefilled pen was well accepted and associated with favorable patient perceptions,
1. Kivitz A, Cohen S, Dowd JE, et al. Clinical assessment of pain, tolerability, and preference of an autoinjection pen versus a prefilled syringe for patient self-administration of the fully human, monoclonal antibody adalimumab: the TOUCH trial. Clin Ther 2006;28:1619–29.
2. Demary W, Schwenke H, Rockwitz K, et al. Subcutaneously administered methotrexate for rheumatoid arthritis, by prefilled syringes versus prefilled pens: patient preference and comparison of the self-injection experience. Patient Prefer Adherence 2014;8:1061–71.
3. Thakur K, Biberger A, Handrich A, Rezk MF. Patient perceptions and preferences of two etanercept autoinjectors for rheumatoid arthritis: findings from a patient survey in Europe. Rheumatol Ther 2016;3:245–56.
1. Kivitz A, Cohen S, Dowd JE, et al. Clinical assessment of pain, tolerability, and preference of an autoinjection pen versus a prefilled syringe for patient self-administration of the fully human, monoclonal antibody adalimumab: the TOUCH trial. Clin Ther 2006;28:1619–29.
2. Demary W, Schwenke H, Rockwitz K, et al. Subcutaneously administered methotrexate for rheumatoid arthritis, by prefilled syringes versus prefilled pens: patient preference and comparison of the self-injection experience. Patient Prefer Adherence 2014;8:1061–71.
3. Thakur K, Biberger A, Handrich A, Rezk MF. Patient perceptions and preferences of two etanercept autoinjectors for rheumatoid arthritis: findings from a patient survey in Europe. Rheumatol Ther 2016;3:245–56.
Navigating travel with diabetes
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
KEY POINTS
- Patients should pack all diabetes medications and supplies in a carry-on bag and keep it in their possession at all times.
- A travel letter will facilitate easy transfer through security and customs.
- Patients should always take more supplies than needed to accommodate changes in travel plans.
- If patients will cross multiple time zones during their travel, they will likely need to adjust their medication and food schedules.
What inpatient treatments do we have for acute intractable migraine?
We recommend the following combination treatment:
Normal saline (0.9% NaCl) 1 to 2 L by intravenous (IV) infusion over 2 to 4 hours. This can be repeated every 6 to 12 hours.
Ketorolac 30-mg IV bolus, which can be repeated every 6 hours. However, patients with coronary artery disease, uncontrolled hypertension, acute renal failure, or cerebrovascular disease should instead receive acetaminophen 1,000 mg, naproxen sodium 550 mg, or aspirin 325 mg by mouth.
Prochlorperazine or metoclopramide 10-mg IV infusion. This can be repeated every 6 hours. However, to reduce the extrapyramidal adverse effects of these drugs, patients should first receive diphenhydramine 25- to 50-mg IV bolus, which can be repeated every 6 to 8 hours.
Sodium valproate 500 to 1,000 mg by IV infusion over 20 minutes. This can be repeated after 8 hours.
Dexamethasone 4-mg IV bolus every 6 hours, or 10-mg IV bolus once in 24 hours.
Magnesium sulfate 500 to 1,000 mg by IV infusion over 1 hour. This can be repeated every 6 to 12 hours.
If the migraine has not improved after 3 cycles of this regimen, a neurologic consultation should be considered. Other options include dihydroergotamine and occipital nerve blocks1 performed at the bedside.
GENERAL PRINCIPLES
Managing acute intractable migraine can be frustrating for both the practitioner and the patient. Some general principles are helpful.
Use a combination of drugs. Aborting a severe migraine attack often requires a combination of medications that work synergistically.
Use IV and intramuscular formulations rather than oral formulations: they are more rapidly absorbed, provide faster pain relief, and can be given when the nausea that often accompanies migraine precludes oral treatments.
Rule out secondary causes. The mnemonic SNOOP—systemic signs, neurologic signs, onset, older age, progression of existing headache disorder—is useful for assessing underlying causes.2 Any patient presenting with intractable migraine should also have a thorough neurologic examination.
Screening electrocardiography may be helpful, as the pretreatment QTc interval may direct the choice of intravenous treatment. If the patient has a prolonged QTc or is taking another drug that could prolong the QTc, certain medications, specifically dopamine receptor antagonists and diphenhydramine, should be avoided.
Ask the patient what has worked previously. A particular agent may have been effective in aborting the migraine; thus, a single dose of it could abort the headache, expediting discharge.
Establish if a triptan or ergot derivative has been used during the 24 hours before presentation, as repeated dosing within this interval is not recommended.3
Establish the baseline headache severity. Complete headache relief is difficult to achieve in a patient with chronic daily headache, and establishing a more realistic goal (eg, 50% relief) from the outset is useful.
OPTIONS FOR DRUG THERAPY
Antiemetics
Dopamine receptor antagonists are assumed to merely treat nausea in patients with migraine; however, they act independently to abort migraine and thus should be considered, irrespective of the presence of nausea.
The two most commonly used agents are prochlorperazine and metoclopramide. The American Academy of Neurology guidelines recommend prochlorperazine as first-line therapy for acute migraine. Metoclopramide is rated slightly lower and is considered to have moderate benefit.4 The Canadian Headache Society cites a high level of evidence supporting prochlorperazine and a moderate level of evidence supporting metoclopramide.5 The American Headache Society assessment of parenteral pharmacotherapies gives prochlorperazine and metoclopramide a level B recommendation of “should offer” (a recommendation only additionally assigned to subcutaneous sumatriptan).3 Hence, either agent can be used.
To reduce the risk of posttreatment akathisia, diphenhydramine or benztropine may be given before starting a dopamine receptor antagonist. Diphenhydramine may be independently effective in migraine treatment,6,7 but data on this are limited.
Ketorolac, ibuprofen
Ketorolac and ibuprofen are the only available nonsteroidal antiinflammatory drugs (NSAIDs) for IV administration. The Canadian Headache Society guidelines strongly recommend ketorolac for the treatment of migraine in emergency settings.5 Doses range from 30 mg to 60 mg.1 Ibuprofen 400 to 800 mg by IV infusion is an acceptable alternative. These medications should be avoided in patients with renal failure or severe coronary artery disease.
Oral naproxen sodium is a possible alternative in patients with cardiovascular disease, as it has been shown to carry a lower cardiovascular risk than other NSAIDs.8
The same concerns in patients with renal dysfunction apply to any NSAID, as the enzyme cyclooxygenase plays a constitutive role in glomerular function.
Antiepileptic drugs
The antiepileptic drugs sodium valproate and levetiracetam are available in IV formulations that have demonstrated efficacy in the treatment of status migrainosus1 (ie, migraine lasting more than 72 hours). Valproate has the strongest track record, is well tolerated, and is effective in rapidly aborting migraine.9
Volume repletion
Although its use is anecdotal and to date no trial has measured its efficacy, IV volume repletion is often used in acute migraine, as most headache experts surmise it to be highly effective, especially in patients with prolonged nausea or vomiting.1
Magnesium
IV magnesium is effective, particularly for migraine with aura.10 Hypotension is a common side effect, and pretreatment or concurrent treatment with IV fluids is advised. Doses from 500 mg to 1,000 mg have demonstrated efficacy.10
Corticosteroids
Corticosteroids can be used in the treatment of status migrainosus. Most studies have shown benefit in preventing recurrences rather than merely aborting migraine.11 A systematic review suggested that recurrent headaches are milder with corticosteroid treatment; 19 of 25 studies indicated favorable benefit, and 6 of 19 studies indicated noninferior outcomes.12
Both IV methylprednisolone and IV dexamethasone may be considered.12 Dexamethasone appears to be particularly effective in preventing headache recurrence when combined with other IV therapies.13 It can be given as a single dose of 10 mg, or as repeated doses of 4 mg up to 16 mg/day.1 Patients with active psychosis or uncontrolled diabetes should be closely monitored for these conditions, which corticosteroids can worsen.
Serotoninergic agents
Serotonin agonists including subcutaneous sumatriptan and IV dihydroergotamine are highly effective, with proven statistical and clinical benefit.4 They should be considered in patients with no known history of coronary artery disease or other vaso-occlusive vascular disorder.1
Ideally, IV dihydroergotamine should be administered after consultation with a neurologist or headache specialist, given the pretreatment and cotreatment requirements often necessary to suppress its side effects. Careful titration is important to prevent transient headache exacerbations during infusion, as well as abdominal cramping, nausea, and diarrhea.
Avoid opioids
Opioids should be avoided. Evidence supporting their use in acute migraine is extremely limited,3 and the risks of migraine becoming chronic and of addiction are high.14 Safer, more effective alternatives have been detailed above.
A detailed algorithm for the management of patients with acute migraine has been published14 and is aimed at decreasing acute treatment with opioids and barbiturates.
- Rozen TD. Emergency department and inpatient management of status migrainosus and intractable headache. Continuum (Minneap Minn) 2015; 21(4):1004–1017. doi:10.1212/CON.0000000000000191
- Dodick D. Headache as a symptom of ominous disease. What are the warning signals? Postgrad Med 1997; 101(5):46–50, 55–56, 62–64. doi:10.3810/pgm.1997.05.217
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache 2016; 56(6):911–940. doi:10.1111/head.12835
- Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55(6):754–762. doi:10.1212/WNL.55.6.754
- Orr SL, Aubé M, Becker WJ, et al. Canadian Headache Society systematic review and recommendations on the treatment of migraine pain in emergency settings. Cephalalgia 2015; 35(3):271–284. doi:10.1177/0333102414535997
- Swidan SZ, Lake AE 3rd, Saper JR. Efficacy of intravenous diphenhydramine versus intravenous DHE-45 in the treatment of severe migraine headache. Curr Pain Headache Rep 2005; 9(1):65–70. doi:10.1007/s11916-005-0077-5
- Marmura MJ, Goldberg SW. Inpatient management of migraine. Curr Neurol Neurosci Rep 2015; 15(4):13. doi:10.1007/s11910-015-0539-z
- Farkouh ME, Greenberg BP. An evidence-based review of the cardiovascular risks of nonsteroidal anti-inflammatory drugs. Am J Cardiol 2009; 103(9):1227–1237. doi:10.1016/j.amjcard.2009.01.014
- Stillman MJ, Zajac D, Rybicki LA. Treatment of primary headache disorders with intravenous valproate: initial outpatient experience. Headache 2004; 44(1):65–69. doi:10.1111/j.1526-4610.2004.04010.x
- Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache 2015; 55(1):3–20. doi:10.1111/head.12499
- Colman I, Friedman BW, Brown MD, et al. Parenteral dexamethasone for acute severe migraine headache: meta-analysis of randomised controlled trials for preventing recurrence. BMJ 2008; 336(7657):1359–1361. doi:10.1136/bmj.39566.806725.BE
- Woldeamanuel YW, Rapoport AM, Cowan RP. The place of corticosteroids in migraine attack management: a 65-year systematic review with pooled analysis and critical appraisal. Cephalalgia 2015; 35(1):996–1024. doi:10.1177/0333102414566200
- Singh A, Alter HJ, Zaia B. Does the addition of dexamethasone to standard therapy for acute migraine headache decrease the incidence of recurrent headache for patients treated in the emergency department? A meta-analysis and systematic review of the literature. Acad Emerg Med 2008; 15(12):1223–1233. doi:10.1111/j.1553-2712.2008.00283.x
- Ahmed ZA, Nacopoulos DA, John S, Papesh N, Levine D, Bamford CC. An algorithm for opioid and barbiturate reduction in the acute management of headache in the emergency department. Headache 2017; 57(1):71–79. doi:10.1111/head.12961
We recommend the following combination treatment:
Normal saline (0.9% NaCl) 1 to 2 L by intravenous (IV) infusion over 2 to 4 hours. This can be repeated every 6 to 12 hours.
Ketorolac 30-mg IV bolus, which can be repeated every 6 hours. However, patients with coronary artery disease, uncontrolled hypertension, acute renal failure, or cerebrovascular disease should instead receive acetaminophen 1,000 mg, naproxen sodium 550 mg, or aspirin 325 mg by mouth.
Prochlorperazine or metoclopramide 10-mg IV infusion. This can be repeated every 6 hours. However, to reduce the extrapyramidal adverse effects of these drugs, patients should first receive diphenhydramine 25- to 50-mg IV bolus, which can be repeated every 6 to 8 hours.
Sodium valproate 500 to 1,000 mg by IV infusion over 20 minutes. This can be repeated after 8 hours.
Dexamethasone 4-mg IV bolus every 6 hours, or 10-mg IV bolus once in 24 hours.
Magnesium sulfate 500 to 1,000 mg by IV infusion over 1 hour. This can be repeated every 6 to 12 hours.
If the migraine has not improved after 3 cycles of this regimen, a neurologic consultation should be considered. Other options include dihydroergotamine and occipital nerve blocks1 performed at the bedside.
GENERAL PRINCIPLES
Managing acute intractable migraine can be frustrating for both the practitioner and the patient. Some general principles are helpful.
Use a combination of drugs. Aborting a severe migraine attack often requires a combination of medications that work synergistically.
Use IV and intramuscular formulations rather than oral formulations: they are more rapidly absorbed, provide faster pain relief, and can be given when the nausea that often accompanies migraine precludes oral treatments.
Rule out secondary causes. The mnemonic SNOOP—systemic signs, neurologic signs, onset, older age, progression of existing headache disorder—is useful for assessing underlying causes.2 Any patient presenting with intractable migraine should also have a thorough neurologic examination.
Screening electrocardiography may be helpful, as the pretreatment QTc interval may direct the choice of intravenous treatment. If the patient has a prolonged QTc or is taking another drug that could prolong the QTc, certain medications, specifically dopamine receptor antagonists and diphenhydramine, should be avoided.
Ask the patient what has worked previously. A particular agent may have been effective in aborting the migraine; thus, a single dose of it could abort the headache, expediting discharge.
Establish if a triptan or ergot derivative has been used during the 24 hours before presentation, as repeated dosing within this interval is not recommended.3
Establish the baseline headache severity. Complete headache relief is difficult to achieve in a patient with chronic daily headache, and establishing a more realistic goal (eg, 50% relief) from the outset is useful.
OPTIONS FOR DRUG THERAPY
Antiemetics
Dopamine receptor antagonists are assumed to merely treat nausea in patients with migraine; however, they act independently to abort migraine and thus should be considered, irrespective of the presence of nausea.
The two most commonly used agents are prochlorperazine and metoclopramide. The American Academy of Neurology guidelines recommend prochlorperazine as first-line therapy for acute migraine. Metoclopramide is rated slightly lower and is considered to have moderate benefit.4 The Canadian Headache Society cites a high level of evidence supporting prochlorperazine and a moderate level of evidence supporting metoclopramide.5 The American Headache Society assessment of parenteral pharmacotherapies gives prochlorperazine and metoclopramide a level B recommendation of “should offer” (a recommendation only additionally assigned to subcutaneous sumatriptan).3 Hence, either agent can be used.
To reduce the risk of posttreatment akathisia, diphenhydramine or benztropine may be given before starting a dopamine receptor antagonist. Diphenhydramine may be independently effective in migraine treatment,6,7 but data on this are limited.
Ketorolac, ibuprofen
Ketorolac and ibuprofen are the only available nonsteroidal antiinflammatory drugs (NSAIDs) for IV administration. The Canadian Headache Society guidelines strongly recommend ketorolac for the treatment of migraine in emergency settings.5 Doses range from 30 mg to 60 mg.1 Ibuprofen 400 to 800 mg by IV infusion is an acceptable alternative. These medications should be avoided in patients with renal failure or severe coronary artery disease.
Oral naproxen sodium is a possible alternative in patients with cardiovascular disease, as it has been shown to carry a lower cardiovascular risk than other NSAIDs.8
The same concerns in patients with renal dysfunction apply to any NSAID, as the enzyme cyclooxygenase plays a constitutive role in glomerular function.
Antiepileptic drugs
The antiepileptic drugs sodium valproate and levetiracetam are available in IV formulations that have demonstrated efficacy in the treatment of status migrainosus1 (ie, migraine lasting more than 72 hours). Valproate has the strongest track record, is well tolerated, and is effective in rapidly aborting migraine.9
Volume repletion
Although its use is anecdotal and to date no trial has measured its efficacy, IV volume repletion is often used in acute migraine, as most headache experts surmise it to be highly effective, especially in patients with prolonged nausea or vomiting.1
Magnesium
IV magnesium is effective, particularly for migraine with aura.10 Hypotension is a common side effect, and pretreatment or concurrent treatment with IV fluids is advised. Doses from 500 mg to 1,000 mg have demonstrated efficacy.10
Corticosteroids
Corticosteroids can be used in the treatment of status migrainosus. Most studies have shown benefit in preventing recurrences rather than merely aborting migraine.11 A systematic review suggested that recurrent headaches are milder with corticosteroid treatment; 19 of 25 studies indicated favorable benefit, and 6 of 19 studies indicated noninferior outcomes.12
Both IV methylprednisolone and IV dexamethasone may be considered.12 Dexamethasone appears to be particularly effective in preventing headache recurrence when combined with other IV therapies.13 It can be given as a single dose of 10 mg, or as repeated doses of 4 mg up to 16 mg/day.1 Patients with active psychosis or uncontrolled diabetes should be closely monitored for these conditions, which corticosteroids can worsen.
Serotoninergic agents
Serotonin agonists including subcutaneous sumatriptan and IV dihydroergotamine are highly effective, with proven statistical and clinical benefit.4 They should be considered in patients with no known history of coronary artery disease or other vaso-occlusive vascular disorder.1
Ideally, IV dihydroergotamine should be administered after consultation with a neurologist or headache specialist, given the pretreatment and cotreatment requirements often necessary to suppress its side effects. Careful titration is important to prevent transient headache exacerbations during infusion, as well as abdominal cramping, nausea, and diarrhea.
Avoid opioids
Opioids should be avoided. Evidence supporting their use in acute migraine is extremely limited,3 and the risks of migraine becoming chronic and of addiction are high.14 Safer, more effective alternatives have been detailed above.
A detailed algorithm for the management of patients with acute migraine has been published14 and is aimed at decreasing acute treatment with opioids and barbiturates.
We recommend the following combination treatment:
Normal saline (0.9% NaCl) 1 to 2 L by intravenous (IV) infusion over 2 to 4 hours. This can be repeated every 6 to 12 hours.
Ketorolac 30-mg IV bolus, which can be repeated every 6 hours. However, patients with coronary artery disease, uncontrolled hypertension, acute renal failure, or cerebrovascular disease should instead receive acetaminophen 1,000 mg, naproxen sodium 550 mg, or aspirin 325 mg by mouth.
Prochlorperazine or metoclopramide 10-mg IV infusion. This can be repeated every 6 hours. However, to reduce the extrapyramidal adverse effects of these drugs, patients should first receive diphenhydramine 25- to 50-mg IV bolus, which can be repeated every 6 to 8 hours.
Sodium valproate 500 to 1,000 mg by IV infusion over 20 minutes. This can be repeated after 8 hours.
Dexamethasone 4-mg IV bolus every 6 hours, or 10-mg IV bolus once in 24 hours.
Magnesium sulfate 500 to 1,000 mg by IV infusion over 1 hour. This can be repeated every 6 to 12 hours.
If the migraine has not improved after 3 cycles of this regimen, a neurologic consultation should be considered. Other options include dihydroergotamine and occipital nerve blocks1 performed at the bedside.
GENERAL PRINCIPLES
Managing acute intractable migraine can be frustrating for both the practitioner and the patient. Some general principles are helpful.
Use a combination of drugs. Aborting a severe migraine attack often requires a combination of medications that work synergistically.
Use IV and intramuscular formulations rather than oral formulations: they are more rapidly absorbed, provide faster pain relief, and can be given when the nausea that often accompanies migraine precludes oral treatments.
Rule out secondary causes. The mnemonic SNOOP—systemic signs, neurologic signs, onset, older age, progression of existing headache disorder—is useful for assessing underlying causes.2 Any patient presenting with intractable migraine should also have a thorough neurologic examination.
Screening electrocardiography may be helpful, as the pretreatment QTc interval may direct the choice of intravenous treatment. If the patient has a prolonged QTc or is taking another drug that could prolong the QTc, certain medications, specifically dopamine receptor antagonists and diphenhydramine, should be avoided.
Ask the patient what has worked previously. A particular agent may have been effective in aborting the migraine; thus, a single dose of it could abort the headache, expediting discharge.
Establish if a triptan or ergot derivative has been used during the 24 hours before presentation, as repeated dosing within this interval is not recommended.3
Establish the baseline headache severity. Complete headache relief is difficult to achieve in a patient with chronic daily headache, and establishing a more realistic goal (eg, 50% relief) from the outset is useful.
OPTIONS FOR DRUG THERAPY
Antiemetics
Dopamine receptor antagonists are assumed to merely treat nausea in patients with migraine; however, they act independently to abort migraine and thus should be considered, irrespective of the presence of nausea.
The two most commonly used agents are prochlorperazine and metoclopramide. The American Academy of Neurology guidelines recommend prochlorperazine as first-line therapy for acute migraine. Metoclopramide is rated slightly lower and is considered to have moderate benefit.4 The Canadian Headache Society cites a high level of evidence supporting prochlorperazine and a moderate level of evidence supporting metoclopramide.5 The American Headache Society assessment of parenteral pharmacotherapies gives prochlorperazine and metoclopramide a level B recommendation of “should offer” (a recommendation only additionally assigned to subcutaneous sumatriptan).3 Hence, either agent can be used.
To reduce the risk of posttreatment akathisia, diphenhydramine or benztropine may be given before starting a dopamine receptor antagonist. Diphenhydramine may be independently effective in migraine treatment,6,7 but data on this are limited.
Ketorolac, ibuprofen
Ketorolac and ibuprofen are the only available nonsteroidal antiinflammatory drugs (NSAIDs) for IV administration. The Canadian Headache Society guidelines strongly recommend ketorolac for the treatment of migraine in emergency settings.5 Doses range from 30 mg to 60 mg.1 Ibuprofen 400 to 800 mg by IV infusion is an acceptable alternative. These medications should be avoided in patients with renal failure or severe coronary artery disease.
Oral naproxen sodium is a possible alternative in patients with cardiovascular disease, as it has been shown to carry a lower cardiovascular risk than other NSAIDs.8
The same concerns in patients with renal dysfunction apply to any NSAID, as the enzyme cyclooxygenase plays a constitutive role in glomerular function.
Antiepileptic drugs
The antiepileptic drugs sodium valproate and levetiracetam are available in IV formulations that have demonstrated efficacy in the treatment of status migrainosus1 (ie, migraine lasting more than 72 hours). Valproate has the strongest track record, is well tolerated, and is effective in rapidly aborting migraine.9
Volume repletion
Although its use is anecdotal and to date no trial has measured its efficacy, IV volume repletion is often used in acute migraine, as most headache experts surmise it to be highly effective, especially in patients with prolonged nausea or vomiting.1
Magnesium
IV magnesium is effective, particularly for migraine with aura.10 Hypotension is a common side effect, and pretreatment or concurrent treatment with IV fluids is advised. Doses from 500 mg to 1,000 mg have demonstrated efficacy.10
Corticosteroids
Corticosteroids can be used in the treatment of status migrainosus. Most studies have shown benefit in preventing recurrences rather than merely aborting migraine.11 A systematic review suggested that recurrent headaches are milder with corticosteroid treatment; 19 of 25 studies indicated favorable benefit, and 6 of 19 studies indicated noninferior outcomes.12
Both IV methylprednisolone and IV dexamethasone may be considered.12 Dexamethasone appears to be particularly effective in preventing headache recurrence when combined with other IV therapies.13 It can be given as a single dose of 10 mg, or as repeated doses of 4 mg up to 16 mg/day.1 Patients with active psychosis or uncontrolled diabetes should be closely monitored for these conditions, which corticosteroids can worsen.
Serotoninergic agents
Serotonin agonists including subcutaneous sumatriptan and IV dihydroergotamine are highly effective, with proven statistical and clinical benefit.4 They should be considered in patients with no known history of coronary artery disease or other vaso-occlusive vascular disorder.1
Ideally, IV dihydroergotamine should be administered after consultation with a neurologist or headache specialist, given the pretreatment and cotreatment requirements often necessary to suppress its side effects. Careful titration is important to prevent transient headache exacerbations during infusion, as well as abdominal cramping, nausea, and diarrhea.
Avoid opioids
Opioids should be avoided. Evidence supporting their use in acute migraine is extremely limited,3 and the risks of migraine becoming chronic and of addiction are high.14 Safer, more effective alternatives have been detailed above.
A detailed algorithm for the management of patients with acute migraine has been published14 and is aimed at decreasing acute treatment with opioids and barbiturates.
- Rozen TD. Emergency department and inpatient management of status migrainosus and intractable headache. Continuum (Minneap Minn) 2015; 21(4):1004–1017. doi:10.1212/CON.0000000000000191
- Dodick D. Headache as a symptom of ominous disease. What are the warning signals? Postgrad Med 1997; 101(5):46–50, 55–56, 62–64. doi:10.3810/pgm.1997.05.217
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache 2016; 56(6):911–940. doi:10.1111/head.12835
- Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55(6):754–762. doi:10.1212/WNL.55.6.754
- Orr SL, Aubé M, Becker WJ, et al. Canadian Headache Society systematic review and recommendations on the treatment of migraine pain in emergency settings. Cephalalgia 2015; 35(3):271–284. doi:10.1177/0333102414535997
- Swidan SZ, Lake AE 3rd, Saper JR. Efficacy of intravenous diphenhydramine versus intravenous DHE-45 in the treatment of severe migraine headache. Curr Pain Headache Rep 2005; 9(1):65–70. doi:10.1007/s11916-005-0077-5
- Marmura MJ, Goldberg SW. Inpatient management of migraine. Curr Neurol Neurosci Rep 2015; 15(4):13. doi:10.1007/s11910-015-0539-z
- Farkouh ME, Greenberg BP. An evidence-based review of the cardiovascular risks of nonsteroidal anti-inflammatory drugs. Am J Cardiol 2009; 103(9):1227–1237. doi:10.1016/j.amjcard.2009.01.014
- Stillman MJ, Zajac D, Rybicki LA. Treatment of primary headache disorders with intravenous valproate: initial outpatient experience. Headache 2004; 44(1):65–69. doi:10.1111/j.1526-4610.2004.04010.x
- Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache 2015; 55(1):3–20. doi:10.1111/head.12499
- Colman I, Friedman BW, Brown MD, et al. Parenteral dexamethasone for acute severe migraine headache: meta-analysis of randomised controlled trials for preventing recurrence. BMJ 2008; 336(7657):1359–1361. doi:10.1136/bmj.39566.806725.BE
- Woldeamanuel YW, Rapoport AM, Cowan RP. The place of corticosteroids in migraine attack management: a 65-year systematic review with pooled analysis and critical appraisal. Cephalalgia 2015; 35(1):996–1024. doi:10.1177/0333102414566200
- Singh A, Alter HJ, Zaia B. Does the addition of dexamethasone to standard therapy for acute migraine headache decrease the incidence of recurrent headache for patients treated in the emergency department? A meta-analysis and systematic review of the literature. Acad Emerg Med 2008; 15(12):1223–1233. doi:10.1111/j.1553-2712.2008.00283.x
- Ahmed ZA, Nacopoulos DA, John S, Papesh N, Levine D, Bamford CC. An algorithm for opioid and barbiturate reduction in the acute management of headache in the emergency department. Headache 2017; 57(1):71–79. doi:10.1111/head.12961
- Rozen TD. Emergency department and inpatient management of status migrainosus and intractable headache. Continuum (Minneap Minn) 2015; 21(4):1004–1017. doi:10.1212/CON.0000000000000191
- Dodick D. Headache as a symptom of ominous disease. What are the warning signals? Postgrad Med 1997; 101(5):46–50, 55–56, 62–64. doi:10.3810/pgm.1997.05.217
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache 2016; 56(6):911–940. doi:10.1111/head.12835
- Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55(6):754–762. doi:10.1212/WNL.55.6.754
- Orr SL, Aubé M, Becker WJ, et al. Canadian Headache Society systematic review and recommendations on the treatment of migraine pain in emergency settings. Cephalalgia 2015; 35(3):271–284. doi:10.1177/0333102414535997
- Swidan SZ, Lake AE 3rd, Saper JR. Efficacy of intravenous diphenhydramine versus intravenous DHE-45 in the treatment of severe migraine headache. Curr Pain Headache Rep 2005; 9(1):65–70. doi:10.1007/s11916-005-0077-5
- Marmura MJ, Goldberg SW. Inpatient management of migraine. Curr Neurol Neurosci Rep 2015; 15(4):13. doi:10.1007/s11910-015-0539-z
- Farkouh ME, Greenberg BP. An evidence-based review of the cardiovascular risks of nonsteroidal anti-inflammatory drugs. Am J Cardiol 2009; 103(9):1227–1237. doi:10.1016/j.amjcard.2009.01.014
- Stillman MJ, Zajac D, Rybicki LA. Treatment of primary headache disorders with intravenous valproate: initial outpatient experience. Headache 2004; 44(1):65–69. doi:10.1111/j.1526-4610.2004.04010.x
- Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: the American Headache Society evidence assessment of migraine pharmacotherapies. Headache 2015; 55(1):3–20. doi:10.1111/head.12499
- Colman I, Friedman BW, Brown MD, et al. Parenteral dexamethasone for acute severe migraine headache: meta-analysis of randomised controlled trials for preventing recurrence. BMJ 2008; 336(7657):1359–1361. doi:10.1136/bmj.39566.806725.BE
- Woldeamanuel YW, Rapoport AM, Cowan RP. The place of corticosteroids in migraine attack management: a 65-year systematic review with pooled analysis and critical appraisal. Cephalalgia 2015; 35(1):996–1024. doi:10.1177/0333102414566200
- Singh A, Alter HJ, Zaia B. Does the addition of dexamethasone to standard therapy for acute migraine headache decrease the incidence of recurrent headache for patients treated in the emergency department? A meta-analysis and systematic review of the literature. Acad Emerg Med 2008; 15(12):1223–1233. doi:10.1111/j.1553-2712.2008.00283.x
- Ahmed ZA, Nacopoulos DA, John S, Papesh N, Levine D, Bamford CC. An algorithm for opioid and barbiturate reduction in the acute management of headache in the emergency department. Headache 2017; 57(1):71–79. doi:10.1111/head.12961
Trump administration proposes changes to HHS, FDA
The Trump administration seeks to reorganize several federal agencies as part of a sweeping reform proposal, issued June 21.
“Government in the 21st century is fundamentally a services business, and modern information technology should be at the heart of the U.S. government service delivery model,” according to the administration’s reform proposal. “And yet, today’s Executive branch is still aligned to the stove-piped organizational constructs of the 20th century, which in many cases have grown inefficient and out of date. Consequently, the public and our workforce are frustrated with government’s ability to deliver its mission in an effective, efficient, and secure way.”
Under the proposal, nutrition assistance programs currently run out of the U.S. Department of Agriculture (USDA) including the Supplemental Nutrition Assistance Program (SNAP) and the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) would move to the Department of Health and Human Services, which would be rebranded the Department of Health and Public Welfare.
Moving these programs “would allow for better and easier coordination across programs that serve similar populations, ensuring consistent policies and a single point of administration for the major public assistance programs,” according to the proposal. “This single point of administration would lead to reduced duplication in state reporting requirements and other administrative burdens, and a more streamlined process for issuing guidance, writing regulations, and approving waivers.”
Food oversight functions would move from the Food and Drug Administration to the USDA; FDA would be rebranded the Federal Drug Administration and focus on drugs, devices, biologics, tobacco, dietary supplements, and cosmetics.
The administration also proposed to created a Council on Public Assistance comprised of “all federal agencies that administer public benefits, with a statutory authority to set cross-cutting program policies, including uniform work requirements.”
Other functions of the council would include approving service plans and waivers by states under Welfare-to-Work projects; resolving disputes when multiple agencies disagree on a particular policy; and recommending policy changes to eliminate barriers at the federal, state, and local level to getting welfare beneficiaries to work.
The proposal also calls for a restructuring of the National Institutes of Health “to ensure operations are effective and efficient,” with no detail provided. It would also place the Agency for Healthcare Research and Quality under the auspices of NIH.
The Strategic National Stockpile would be managed by the Assistant Secretary for Preparedness and Response “to consolidate strategic decision making around the development and procurement of medical countermeasures, and streamline operational decisions during responses to public health and other emergencies to improve responsiveness.”
The Trump administration seeks to reorganize several federal agencies as part of a sweeping reform proposal, issued June 21.
“Government in the 21st century is fundamentally a services business, and modern information technology should be at the heart of the U.S. government service delivery model,” according to the administration’s reform proposal. “And yet, today’s Executive branch is still aligned to the stove-piped organizational constructs of the 20th century, which in many cases have grown inefficient and out of date. Consequently, the public and our workforce are frustrated with government’s ability to deliver its mission in an effective, efficient, and secure way.”
Under the proposal, nutrition assistance programs currently run out of the U.S. Department of Agriculture (USDA) including the Supplemental Nutrition Assistance Program (SNAP) and the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) would move to the Department of Health and Human Services, which would be rebranded the Department of Health and Public Welfare.
Moving these programs “would allow for better and easier coordination across programs that serve similar populations, ensuring consistent policies and a single point of administration for the major public assistance programs,” according to the proposal. “This single point of administration would lead to reduced duplication in state reporting requirements and other administrative burdens, and a more streamlined process for issuing guidance, writing regulations, and approving waivers.”
Food oversight functions would move from the Food and Drug Administration to the USDA; FDA would be rebranded the Federal Drug Administration and focus on drugs, devices, biologics, tobacco, dietary supplements, and cosmetics.
The administration also proposed to created a Council on Public Assistance comprised of “all federal agencies that administer public benefits, with a statutory authority to set cross-cutting program policies, including uniform work requirements.”
Other functions of the council would include approving service plans and waivers by states under Welfare-to-Work projects; resolving disputes when multiple agencies disagree on a particular policy; and recommending policy changes to eliminate barriers at the federal, state, and local level to getting welfare beneficiaries to work.
The proposal also calls for a restructuring of the National Institutes of Health “to ensure operations are effective and efficient,” with no detail provided. It would also place the Agency for Healthcare Research and Quality under the auspices of NIH.
The Strategic National Stockpile would be managed by the Assistant Secretary for Preparedness and Response “to consolidate strategic decision making around the development and procurement of medical countermeasures, and streamline operational decisions during responses to public health and other emergencies to improve responsiveness.”
The Trump administration seeks to reorganize several federal agencies as part of a sweeping reform proposal, issued June 21.
“Government in the 21st century is fundamentally a services business, and modern information technology should be at the heart of the U.S. government service delivery model,” according to the administration’s reform proposal. “And yet, today’s Executive branch is still aligned to the stove-piped organizational constructs of the 20th century, which in many cases have grown inefficient and out of date. Consequently, the public and our workforce are frustrated with government’s ability to deliver its mission in an effective, efficient, and secure way.”
Under the proposal, nutrition assistance programs currently run out of the U.S. Department of Agriculture (USDA) including the Supplemental Nutrition Assistance Program (SNAP) and the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) would move to the Department of Health and Human Services, which would be rebranded the Department of Health and Public Welfare.
Moving these programs “would allow for better and easier coordination across programs that serve similar populations, ensuring consistent policies and a single point of administration for the major public assistance programs,” according to the proposal. “This single point of administration would lead to reduced duplication in state reporting requirements and other administrative burdens, and a more streamlined process for issuing guidance, writing regulations, and approving waivers.”
Food oversight functions would move from the Food and Drug Administration to the USDA; FDA would be rebranded the Federal Drug Administration and focus on drugs, devices, biologics, tobacco, dietary supplements, and cosmetics.
The administration also proposed to created a Council on Public Assistance comprised of “all federal agencies that administer public benefits, with a statutory authority to set cross-cutting program policies, including uniform work requirements.”
Other functions of the council would include approving service plans and waivers by states under Welfare-to-Work projects; resolving disputes when multiple agencies disagree on a particular policy; and recommending policy changes to eliminate barriers at the federal, state, and local level to getting welfare beneficiaries to work.
The proposal also calls for a restructuring of the National Institutes of Health “to ensure operations are effective and efficient,” with no detail provided. It would also place the Agency for Healthcare Research and Quality under the auspices of NIH.
The Strategic National Stockpile would be managed by the Assistant Secretary for Preparedness and Response “to consolidate strategic decision making around the development and procurement of medical countermeasures, and streamline operational decisions during responses to public health and other emergencies to improve responsiveness.”
Hydroxychloroquine: An old drug with new relevance
A 29-year-old African American woman presents with a photosensitive malar rash, fatigue, morning stiffness, and swelling in her hands. She is found to have elevated antinuclear antibody at a titer of 1:320. A complete blood cell count demonstrates leukopenia and thrombocytopenia. Results of renal function testing and urinalysis are within normal limits. She has no other medical problems and no history of blood clots or pregnancy loss.
Her arthritis and rash suggest systemic lupus erythematosus. She is counseled to avoid sun exposure, and treatment with hydroxychloroquine is considered.
WHAT IS HYDROXYCHLOROQUINE?
Hydroxychloroquine was developed to treat malaria but was later found to have immunomodulatory properties. It is now approved by the US Food and Drug Administration for treatment of discoid lupus, systemic lupus erythematosus, and rheumatoid arthritis. It is also approved to treat malaria; however, of the several malarial parasites, only Plasmodium falciparum can still be cured by hydroxychloroquine, and growing resistance limits the geographic locations where this drug can be used effectively.1,2
HISTORICAL BACKGROUND
Antimalarial drugs were discovered shortly before World War II. Their production was industrialized during the war because malaria was a leading cause of disease among soldiers, especially those deployed to the South Pacific.3
Atabrine (quinacrine), the first antimalarial widely used, had numerous side effects including yellowing of the skin. Aggressive research efforts to develop an alternative led to field testing of one of its derivative compounds, chloroquine, by the US Army in 1943. Continued chemical modification would create hydroxychloroquine, introduced in 1955.
A serendipitous consequence of the mass use of antimalarials during World War II was the discovery that they could be used to treat inflammatory arthritis and lupus. Eight years after the war ended, Shee4 reported that chloroquine had a beneficial effect on lupus and rheumatoid arthritis in US soldiers. Hydroxychloroquine is now the most commonly prescribed antimalarial for treatment of autoimmune disease.
HOW HYDROXYCHLOROQUINE WORKS
The primary mechanism by which hydroxychloroquine modulates systemic lupus erythematosus is by suppressing activation of Toll-like receptors, which exist on the surface of endosomes and play a significant role in the innate immune response and in autoimmune disease. Their activation is necessary for the expression of interferon-regulated genes and production of tumor necrosis factor alpha, which are key in the cell-mediated inflammatory response.
Antimalarial drugs such as hydroxychloroquine prevent Toll-like receptor activation by binding directly to nucleic acids in the activation pathway.5 In vitro studies show that blocking this pathway blunts the body’s primary cell-mediated inflammatory response; in vivo studies show that use of hydroxychloroquine is strongly correlated with a reduction in interferon alpha levels.6 The powerful effect of hydroxychloroquine on the cell-mediated pattern of inflammation found in lupus is consistent with this theory.
It was previously hypothesized that the immune-modulating effects of hydroxychloroquine were associated with a more general dysregulation of cellular lysosomes through inhibition of proteolysis or changes in cellular pH.7 This theory has since been displaced by the more specific and elegant mechanism described above.5
HOW WELL DOES IT WORK?
Benefit in systemic lupus erythematosus
Hydroxychloroquine has consistently demonstrated significant and multifaceted benefit in patients with systemic lupus erythematosus.
A systematic review of 95 articles8 concluded that this drug decreases lupus flares and decreases mortality rates in lupus patients by at least 50%, with a high level of evidence. Beneficial effects that had a moderate level of evidence were an increase in bone mineral density, fewer thrombotic events, and fewer cases of irreversible organ damage.
The preventive effect of hydroxychloroquine on thrombosis in lupus patients has been consistently demonstrated and is one of the key reasons the drug is considered a cornerstone of therapy in this disease.9 A nested case-control study of patients with lupus and thromboembolism demonstrated an odds ratio of 0.31 and relative risk reduction of 68% for those using antimalarials.10
Benefit in antiphospholipid antibody syndrome
Hydroxychloroquine prevents thrombosis in other diseases as well. For example, it has been shown to reduce the incidence of thrombotic events in patients with primary antiphospholipid syndrome.
In a retrospective cohort study in 114 patients with this disease, hydroxychloroquine significantly reduced the incidence of arterial thrombotic events over 10 years of follow-up (recurrence incidence 0 in those treated with hydroxychloroquine vs 1.14% in those not treated).11 The study also tracked levels of antiphospholipid antibodies and reported that hydroxychloroquine significantly reduced the levels of antibodies to cardiolipin and beta-2 glycoprotein 1, both implicated in the pathology of thrombosis.11
In vitro studies have also demonstrated that hydroxychloroquine can modulate a dysregulated inflammatory system to reduce thrombosis. For example, it has been shown that hydroxychloroquine can reverse platelet activation by antiphospholipid antibodies, prevent linking of antibody complexes to cell membranes, and promote proper membrane protein expression, thereby reducing the thrombotic qualities of antiphospholipid antibodies and even improving clearance times of antiphospholipid-related thrombi.12
Benefit in rheumatoid arthritis
Though there is less evidence, hydroxychloroquine has also shown benefit in rheumatoid arthritis, where it can be used by itself in mild disease or as part of combination therapy with active arthritis. Compared with biologic therapy in patients with early aggressive rheumatoid arthritis, triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine was nearly as effective in terms of quality of life, and it cost only one-third as much, saving $20,000 per year of therapy per patient.13
Hydroxychloroquine has also been compared directly with chloroquine, its closest relation, in a large study incorporating patients with rheumatoid arthritis and patients with systemic lupus erythematosus. Patients using chloroquine experienced significantly more side effects, though it did prove marginally more effective.14
No benefit shown in Sjögren syndrome
Unfortunately, despite widespread use, hydroxychloroquine has not demonstrated positive clinical effects when used to treat primary Sjögren syndrome. Most notably, a 2014 randomized controlled trial of hydroxychloroquine vs placebo in 120 Sjögren patients found no significant improvement in primary symptoms of dryness, pain, or fatigue after 6 months of therapy.15
Metabolic benefits
Unexpectedly, hydroxychloroquine is associated with multiple metabolic benefits including improved lipid profiles and lower blood glucose levels. These findings, in addition to a reduced incidence of thrombosis, were initially reported in the Baltimore Lupus Cohort in 1996.16 Specifically, longitudinal evaluation of a cohort of lupus patients showed that hydroxychloroquine use was associated with a 7.6% reduction in total cholesterol and a 13.7% reduction in low-density lipoprotein cholesterol (LDL-C) over 3 months of therapy.17
Similar findings, including a reduction in LDL-C and an increase in high-density lipoprotein cholesterol, were strongly associated with the addition of hydroxychloroquine to methotrexate or to methotrexate and etanercept in a large cohort of rheumatoid arthritis patients followed over 2 years of therapy.18
In nondiabetic women with systemic lupus erythematosus or rheumatoid arthritis, average blood glucose was significantly lower in those taking hydroxychloroquine than in nonusers. The incidence of insulin resistance was also lower, but the difference was not statistically significant.19
Some have suggested that hydroxychloroquine may prevent diabetes mellitus. In a retrospective case series, compared with rheumatoid arthritis patients not taking the drug, patients treated with hydroxychloroquine for more than 4 years had a 25% lower risk of developing diabetes mellitus.20
In view of these metabolic benefits, especially regarding lipid regulation, and the above described antithrombotic properties of hydroxychloroquine, some researchers have recently hypothesized that hydroxychloroquine may be of benefit in patients with coronary artery disease.21 They suggested that the inflammatory contribution to the mechanism of coronary artery disease could be lessened by hydroxychloroquine even in patients without lupus erythematosus or rheumatoid arthritis.
PHARMACOLOGIC PROPERTIES
Understanding the pharmacologic properties of hydroxychloroquine is key to using it appropriately in clinical practice.
The half-life of elimination of hydroxychloroquine is 40 to 50 days, with half of the drug excreted renally in a concentration-dependent fashion.22,23 The drug reaches 95% of its steady-state concentration by about 6 months of therapy. Shorter durations of therapy do not provide adequate time for the drug to achieve steady-state concentration and may not allow patients and providers time to see its full clinical results. Therefore, its manufacturers recommend a 6-month trial of therapy to adequately determine if the drug improves symptoms.1
The oral bioavailability of hydroxychloroquine is about 75%, but pharmacokinetics vary among individuals.22,23 It has been suggested that this variability affects the efficacy of hydroxychloroquine. In a study of 300 patients with cutaneous lupus erythematosus, those whose treatment failed had significantly lower blood concentrations of hydroxychloroquine, while those who achieved complete remission had significantly higher concentrations.24
Another study found that titrating doses to target therapeutic blood concentrations can reduce disease activity in cutaneous lupus erythematosus.25 Measuring the blood concentration of hydroxychloroquine is not common in clinical practice but may have a role in select patients in whom initial therapy using a standard dosing regimen does not produce the desired results.
HOW SAFE IS HYDROXYCHLOROQUINE?
Hydroxychloroquine has numerous adverse effects, necessitating vigilance on the part of the prescriber. Most commonly reported are retinopathy, hyperpigmentation, myopathy, and skin reactions.1
Retinopathy
Retinopathy’s irreversibility—the threat of permanent vision loss—and its substantial prevalence in patients with a large drug exposure history, have marked retinopathy as the most concerning potential toxicity. The risk of ocular toxicity increases with the cumulative hydroxychloroquine dose. The prevalence of retinopathy in those using the drug less than 10 years is less than 2%; in contrast, the prevalence in patients with more than 20 years of exposure is reported to be as high as 20%.26
The American Academy of Ophthalmology has long stated that retinopathy is a significant risk of hydroxychloroquine therapy and that patients taking hydroxychloroquine should therefore undergo routine retinal and visual field screening by an ophthalmologist.
Currently, initial screening followed by yearly screening beginning 5 years thereafter is recommended for patients at low risk of toxicity (Table 1).27 Patients determined by an ophthalmologist to be at higher risk of retinopathy should be screened yearly. As identified by the American Academy of Ophthalmology, major risk factors for retinopathy include duration of use, concomitant tamoxifen exposure, significant renal disease, and preexisting retinal and macular disease.26,28
Recommendations for hydroxychloroquine dosing and screening were recently revised, for 2 reasons. Initially, its manufacturers recommended that hydroxychloroquine dosage be no higher than 6.5 mg/kg of ideal body weight to prevent retinopathy.1,29,30 However, it has recently been demonstrated that real body weight is a better predictor of risk of retinopathy than ideal body weight when dosing hydroxychloroquine, perhaps because of the increasing variance of real body weight in our patient population.26
Further, an atypical pattern of retinopathy called pericentral retinopathy is more common in Asians. A study of about 200 patients with a history of hydroxychloroquine retinopathy, including 36 Asian patients, found that the pericentral pattern occurred in half the Asian patients but only 2% of the white patients.31 The mechanism for this finding is unclear, but because pericentral retinopathy spares the macula, it can be missed using standard screening methods. Therefore, the American Academy of Ophthalmology now recommends that the dose limit be reduced from 6.5 mg/kg of ideal body weight to no more than 5.0 mg/kg of real body weight (Table 2).28
It is also recommended that screening methods such as automated visual fields and optical coherence tomography extend their fields beyond the macula in Asian patients to ensure that pericentral retinopathy is not missed.28
Optical coherence tomography is a particularly useful tool in the ocular evaluation of patients taking hydroxychloroquine. It can detect subtle changes such as thinning of the foveal photoreceptor outer segment, thickening of the retinal pigment epithelium, and loss of the macular ganglion cell–inner plexiform layer before there are visible signs of retinopathy and before symptoms arise.32
Currently, these guidelines are underutilized in clinical practice. Physician adherence to ophthalmologic guidelines is reported at about 50%.33 This statistic is jarring, given the potential for permanent loss of vision in those with hydroxychloroquine-mediated retinopathy, and demonstrates the importance of reinforcing proper understanding of the use of hydroxychloroquine in clinical practice.
Other adverse effects
Cutaneous hyperpigmentation can occur with hydroxychloroquine use (Figure 1). The hyperpigmentation appears to be due to local bruising following deposition of iron in the soft tissue.
While the pigmentation may persist permanently and cause an undesirable cosmetic effect, it has not been associated with other adverse outcomes.
Myopathy is a rare adverse effect. In one case series, 3 of 214 patients treated with hydroxychloroquine developed hydroxychloroquine-induced myopathy.35 Over the duration of their therapy, this was equivalent to an incidence of 1 case of myopathy in 100 patient-years of therapy. Myopathy improves with discontinuation of therapy, though it can persist for weeks, likely because of hydroxychloroquine’s prolonged elimination half-life.
Cardiomyopathy, specifically neurocardiomyopathy, is also an extremely rare adverse effect of hydroxychloroquine use. The mechanism is believed to be associated with the effect of hydroxychloroquine on lysosomal action, leading to an acquired lysosomal storage disorder with the typical cardiac hypertrophy and conduction abnormalities associated with this family of diseases.36
Acute generalized exanthematous pustulosis is another rare complication of hydroxychloroquine therapy. The appearance of the reaction is similar to that of pustular psoriasis, with pustules overlying flaking and scaling skin. It usually resolves within 2 weeks after cessation of hydroxychloroquine therapy. In a select few cases, the reaction persists or waxes and wanes over a period of weeks to months, and longer durations of recovery are thought to be due to hydroxychloroquine’s long half-life, as in hydroxychloroquine-induced myopathy.37
In view of this rare reaction, manufacturers of hydroxychloroquine recommend caution when using the drug in patients with psoriasis.1
Hematologic abnormalities. In very rare cases, hydroxychloroquine is associated with hematologic abnormalities including agranulocytosis, anemia, aplastic anemia, leukopenia, and thrombocytopenia.1
While no specific guidelines exist, caution is warranted when using hydroxychloroquine in patients with porphyria. Additionally, hydroxychloroquine and other antimalarials including primaquine have been associated with hemolysis in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency. The risk of hemolysis is generally considered low except at high hydroxychloroquine doses in patients with severe G6PD deficiency.38
For the above reasons, manufacturers recommended baseline and routine blood counts, and some providers screen patients for G6PD deficiency when prescribing hydroxychloroquine (Table 3).
PREGNANCY
Hydroxychloroquine is in pregnancy category C. Information is limited, and in view of the risks, the manufacturer says that it should be avoided in pregnancy.1 Nevertheless, it is generally considered safe during pregnancy, and its benefits may make it acceptable to continue in a patient who becomes pregnant, in spite of the possible risks.
We favor continuing hydroxychloroquine. This drug has been associated with improved maternal and fetal outcomes in lupus patients. Its use during pregnancy has not been associated with congenital malformations. The adverse visual effects of long-term hydroxychloroquine use, namely retinopathy, have never been reported in children as a consequence of exposure in utero.
In addition, hydroxychloroquine is transmitted only in minute quantities in breast milk.39 In pregnant women with systemic lupus erythematosus, hydroxychloroquine was associated with a lower risk of adverse outcomes, including preterm delivery and intrauterine growth restriction.40 However, hydroxychloroquine is far more toxic when ingested directly by infants than in adults.1
Maternal outcomes are also improved with the use of hydroxychloroquine. Stopping hydroxychloroquine during pregnancy in women with systemic lupus erythematosus is associated with significantly higher disease activity—fully twice as high as in those who continue hydroxychloroquine.41 These study results were corroborated in a small randomized trial in which pregnant women with lupus on placebo had significantly higher lupus disease activity scores than those pregnant women who were given hydroxychloroquine.42 The women taking hydroxychloroquine experienced no severe lupus flares for the duration of their pregnancies.
These findings suggest not only that hydroxychloroquine is safe in pregnancy, but also that it should be continued in lupus patients during pregnancy to prevent disease flares and adverse fetal outcomes.
AREAS OF UNCERTAINTY
Benefit in preclinical lupus?
Hydroxychloroquine has a consistently profound effect on outcomes in systemic lupus erythematosus. These findings, in addition to the more widespread use of antibody screening, have led to suggestions that hydroxychloroquine could be of benefit even before systemic lupus erythematosus is diagnosed.
A study in US military personnel found that patients taking hydroxychloroquine experienced a significantly longer lag time between first reported clinical symptoms of lupus and official diagnosis compared with matched controls who also went on to develop the disease, averaging 1.08 vs 0.29 years to disease classification.43 Those who used hydroxychloroquine also had lower rates of autoantibody accumulation. Therefore, hydroxychloroquine could be of benefit in carefully selected candidates at high risk of developing systemic lupus erythematosus.
The beneficial effects of hydroxychloroquine on patients with lupus and rheumatoid arthritis, in terms of primary measures of disease activity and secondary outcomes, were discovered fortuitously and were not the original intended targets of the drug. Because of its versatility, there are numerous other disease states in which hydroxychloroquine has shown a degree of benefit or has shown a potential for benefit.
Antiviral activity?
It has been suggested that antimalarial drugs could serve as adjunctive therapies against filoviruses such as Marburg and Ebola. There is a small body of in vitro and in vivo evidence that hydroxychloroquine could temper severe systemic inflammatory responses to filoviruses both through dysregulation of lysosomes and lysosomal pH (filoviruses have a pH-dependent mechanism of action) and through decreased production of tumor necrosis factor alpha and interferons. Heavy burdens of interferons and tumor necrosis factor alpha are associated with increased mortality rates in those infected with filoviruses.44
Antineoplastic activity?
Hydroxychloroquine has undergone in vitro testing as an adjunct to cancer therapies. There are several mechanisms by which it is theorized that hydroxychloroquine could target malignant cells, including inhibition of multidrug resistance pumps or autophagy, improvement of chemotherapy cell penetration, potentiation of presentation of major histocompatibility complexes, or even intercalation directly into DNA.45,46 However, it can also impair natural anticancer immunity and may allow cancer cells better nutrient supply through vascular effects.
In vitro studies have shown tumoricidal effects in lymphoma and melanoma, and inhibition of growth in lung, colon, breast, cervix, larynx, liver, and prostate cancers. In vivo studies have shown that hydroxychloroquine in high doses can prolong survival in glioblastoma.45
Unfortunately, all of these theorized or observed effects are dose-dependent and likely require doses that exceed currently recommended maximums.
Negative chronotropic effect?
Hydroxychloroquine has been found to decrease the resting heart rate in a cumulative dose-dependent fashion.47 Further, hydroxychloroquine has been known to increase digoxin levels, and the medications should not be used in combination.1
Whether the decrease in resting heart rate is associated with harm or benefit and whether the effect is significant enough to be considered when implementing therapy remain unanswered and deserve further investigation, as does the primary use of hydroxychloroquine for beneficial lipid and glucose reduction in patients who are otherwise healthy.
CASE CONCLUSION
The patient described at the beginning of this article was provided with information on the risks and benefits of hydroxychloroquine for treatment of her arthritis and rash suggestive of mild systemic lupus, and she opted to begin therapy. Her baseline eye screening was within normal limits. Based on her weight of 62 kg, she was started on 300 mg of hydroxychloroquine daily.
She had no significant adverse effects from the medication and reported slow improvement in her rash and joint complaints over the next 2 months. She remained on hydroxychloroquine over the next year without adverse effects or new evidence of autoimmune disease.
- Sanofi-Aventis. Product monograph: Plaquenil. http://products.sanofi.ca/en/plaquenil.pdf. Accessed May 2, 2018.
- Centers for Disease Control and Prevention (CDC). Malaria information and prophylaxis, by country. www.cdc.gov/malaria/travelers/country_table/a.html. Accessed May 2, 2018.
- Wallace DJ. The history of antimalarials. Lupus 1996; 5(suppl 1):S2–S3. pmid:8803902
- Shee JC. Lupus erythematosus treated with chloroquine. Lancet 1953; 265(6778):201–202. pmid:13070595
- Kuznik A, Bencina M, Svajger U, Jeras M, Rozman B, Jerala R. Mechanism of endosomal TLR inhibition by antimalarial drugs and imidazoquinolines. J Immunol 2011; 186:4794–4804. doi:10.4049/jimmunol.1000702
- Willis R, Seif AM, McGwin G Jr, et al. Effect of hydroxychloroquine treatment on pro-inflammatory cytokines and disease activity in SLE patients: data from LUMINA, a multiethnic US cohort. Lupus 2012; 21(8):830–835. doi:10.1177/0961203312437270
- Fox R. Anti-malarial drugs: possible mechanisms of action in autoimmune disease and prospects for drug development. Lupus 1996; 5(suppl 1):S4–S10. pmid:8803903
- Ruiz-Irastorza G, Ramos-Casals M, Brito-Zeron P, Khamashta MA. Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann Rheum Dis 2010; 69(1):20–28. doi:10.1136/ard.2008.101766
- Lam NC, Ghetu MV, Bieniek ML. Systemic lupus erythematosus: primary care approach to diagnosis and management. Am Fam Physician 2016; 94(4):284–294. pmid:27548593
- Jung H, Bobba R, Su J, et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum 2010; 62(3):863–868. doi:10.1002/art.27289
- Nuri E, Taraborelli M, Andreoli L, et al. Long-term use of hydroxychloroquine reduces antiphospholipid antibodies levels in patients with primary antiphospholipid syndrome. Immunol Res 2017; 65(1):17–24. doi:10.1007/s12026-016-8812-z
- Erkan D, Aguiar CL, Andrade D, et al. 14th International Congress on Antiphospholipid Antibodies: task force report on antiphospholipid syndrome treatment trends. Autoimmun Rev 2014; 13(6):685–696. doi:10.1016/j.autrev.2014.01.053
- Jalal H, O’Dell JR, Bridges SL Jr, et al. Cost-effectiveness of triple therapy versus etanercept plus methotrexate in early aggressive rheumatoid arthritis. Arthritis Care Res (Hoboken) 2016; 68(12):1751–1757. doi:10.1002/acr.22895
- Avina-Zubieta JA, Galindo-Rodriguez G, Newman S, Suarez-Almazor ME, Russell AS. Long-term effectiveness of antimalarial drugs in rheumatic diseases. Ann Rheum Dis 1998; 57(10):582–587. pmid:9893568
- Gottenberg JE, Ravaud P, Puechal X, et al. Effects of hydroxychloroquine on symptomatic improvement in primary Sjogren syndrome. JAMA 2014; 312(3):249–258. doi:10.1001/jama.2014.7682
- Petri M. Hydroxychloroquine use in the Baltimore Lupus Cohort: effects on lipids, glucose and thrombosis. Lupus 1996; 5(suppl 1):S16–S22. pmid:8803905
- Cairoli E, Rebella M, Danese N, Garra V, Borba EF. Hydroxychloroquine reduces low-density lipoprotein cholesterol levels in systemic lupus erythematosus: a longitudinal evaluation of the lipid-lowering effect. Lupus 2012; 21(11):1178–1182. doi:10.1177/0961203312450084
- Charles-Schoeman C, Wang X, Lee YY, et al. Association of triple therapy with improvement in cholesterol profiles over two-year followup in the treatment of early aggressive rheumatoid arthritis trial. Arthritis Rheumatol 2016; 68(3):577–586. doi:10.1002/art.39502
- Penn SK, Kao AH, Schott LL, et al. Hydroxychloroquine and glycemia in women with rheumatoid arthritis and systemic lupus erythematosus. J Rheumatol 2010; 37(6):1136–1142. doi:10.3899/jrheum.090994
- Wasko MC, Hubert HB, Lingala VB, et al. Hydroxychloroquine and risk of diabetes in patients with rheumatoid arthritis. JAMA 2007; 298(2):187–193. doi:10.1001/jama.298.2.187
- Sun L, Liu M, Li R, et al. Hydroxychloroquine, a promising choice for coronary artery disease? Med Hypotheses 2016; 93:5–7. doi:10.1016/j.mehy.2016.04.045
- Tett SE, Cutler DJ, Day RO, Brown KF. Bioavailability of hydroxychloroquine tablets in healthy volunteers. Br J Clin Pharmacol 1989; 27(6):771–779. pmid:2757893
- Furst DE. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus 1996; 5(suppl 1):S11–S15. pmid:8803904
- Frances C, Cosnes A, Duhaut P, et al. Low blood concentration of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus. Arch Dermatol 2012; 148(4):479–484. doi:10.1001/archdermatol.2011.2558
- Chasset F, Arnaud L, Costedoat-Chalumeau N, Zahr N, Bessis D, Francès C. The effect of increasing the dose of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus: an open-label prospective pilot study. J Am Acad Dermatol 2016; 74(4):693–699.e3. doi:10.1016/j.jaad.2015.09.064
- Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol 2014; 132(12):1453–1460. doi:10.1001/jamaophthalmol.2014.3459
- Committee on Rheumatologic Care. American College of Rheumatology position statement. Screening for hydroxychloroquine retinopathy. www.rheumatology.org/Portals/0/Files/Screening-for-Hydroxychloroquine-Retinopathy-Position-Statement.pdf. Accessed April 2, 2018.
- Marmor MF, Kellner U, Lai TY, Melles RB, Mieler WF; American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology 2016; 123(6):1386–1394. doi:10.1016/j.ophtha.2016.01.058
- Mackenzie AH. Antimalarial drugs for rheumatoid arthritis. Am J Med 1983; 75(6A):48–58. pmid:6362406
- Mackenzie AH. Dose refinements in long-term therapy of rheumatoid arthritis with antimalarials. Am J Med 1983; 75(1A):40–45. pmid:6869410
- Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology 2015; 122(1):110–116. doi:10.1016/j.ophtha.2014.07.018
- Uslu H, Gurler B, Yildirim A, et al. Effect of hydroxychloroquine on the retinal layers: a quantitative evaluation with spectral-domain optical coherence tomography. J Ophthalmol 2016; 2016:8643174. doi:10.1155/2016/8643174
- Au A, Parikh V, Modi YS, Ehlers JP, Schachat AP, Singh RP. Hydroxychloroquine screening practice patterns within a large multispecialty ophthalmic practice. Am J Ophthalmol 2015; 160(3):561–568.e2. doi:10.1016/j.ajo.2015.06.009
- Jallouli M, Frances C, Plette JC, et al; Plaquenil Lupus Systemic Study Group. Hydroxychloroquine-induced pigmentation in patients with systemic lupus erythematosus. JAMA Dermatol 2013; 149(8):935–940. doi:10.1001/jamadermatol.2013.709
- Avina-Zubieta JA, Johnson ES, Suarez-Almazor ME, Russell AS. Incidence of myopathy in patients treated with antimalarials: a report of three cases and review of the literature. Br J Rheumatol 1995; 34(2):166–170. pmid:7704464
- Yogasundaram H, Putko BN, Tien J, et al. Hydroxychloroquine-induced cardiomyopathy: case report, pathophysiology, diagnosis, and treatment. Can J Cardiol 2014; 30:1706–1715. doi:10.1016/j.cjca.2014.08.016
- Pearson KC, Morrell DS, Runge SR, Jolly P. Prolonged pustular eruption from hydroxychloroquine: an unusual case of acute generalized exanthematous pustulosis. Cutis 2016; 97(3):212–216. pmid:27023083
- Youngster I, Arcavi L, Schechmaster R, et al. Medications and glucose-6-phosphate dehydrogenase deficiency: an evidence-based review. Drug Saf 2010; 33(9):713–726. doi:10.2165/11536520-000000000-00000
- Ostensen M, Khamashta M, Lockshin M, et al. Anti-inflammatory and immunosuppressive drugs and reproduction. Arthritis Res Ther 2006; 8(3):209. doi:10.1186/ar1957
- Leroux M, Desveaux C, Parcevaux M, et al. Impact of hydroxychloroquine on preterm delivery and intrauterine growth restriction in pregnant women with systemic lupus erythematosus: a descriptive cohort study. Lupus 2015; 24(13):1384–1391. doi:10.1177/0961203315591027
- Clowse MEB, Magder L, Witter F, Petri M. Hydroxychloroquine in lupus pregnancy. Arthritis Rheum 2006; 54(11):3640–3647. doi:10.1002/art.22159
- Levy RA, Vilela VS, Cataldo MJ, et al. Hydroxychloroquine in lupus pregnancy: double-blind and placebo-controlled study. Lupus 2001; 10(6):401–404. doi:10.1191/096120301678646137
- James JA, Kim-Howard XR, Bruner BF, et al. Hydroxychloroquine sulfate treatment is associated with later onset of systemic lupus erythematosus. Lupus 2007; 16(6):401–409. doi:10.1177/0961203307078579
- Akpovwa H. Chloroquine could be used for the treatment of filoviral infections and other viral infections that emerge or emerged from viruses requiring an acidic pH for infectivity. Cell Biochem Funct 2016; 34(4):191–196. doi:10.1002/cbf.3182
- Pascolo S. Time to use a dose of chloroquine as an adjuvant to anti-cancer chemotherapies. Eur J Pharmacol 2016; 771:139–144. doi:10.1016/j.ejphar.2015.12.017
- Furlong HC, Wessels JM, Guerra MT, Stämpfli MR, Foster WG. Hydroxychloroquine attenuates cigarette smoke induced autophagic signaling in the mouse ovary. Reprod Toxicol 2016; 61:105–113. doi:10.1016/j.reprotox.2016.03.044
- Cairoli E, Danese N, Teliz M, et al. Cumulative dose of hydroxychloroquine is associated with a decrease of resting heart rate in patients with systemic lupus erythematosus: a pilot study. Lupus 2015; 24(11):1204–1209. doi:10.1177/0961203315580870
A 29-year-old African American woman presents with a photosensitive malar rash, fatigue, morning stiffness, and swelling in her hands. She is found to have elevated antinuclear antibody at a titer of 1:320. A complete blood cell count demonstrates leukopenia and thrombocytopenia. Results of renal function testing and urinalysis are within normal limits. She has no other medical problems and no history of blood clots or pregnancy loss.
Her arthritis and rash suggest systemic lupus erythematosus. She is counseled to avoid sun exposure, and treatment with hydroxychloroquine is considered.
WHAT IS HYDROXYCHLOROQUINE?
Hydroxychloroquine was developed to treat malaria but was later found to have immunomodulatory properties. It is now approved by the US Food and Drug Administration for treatment of discoid lupus, systemic lupus erythematosus, and rheumatoid arthritis. It is also approved to treat malaria; however, of the several malarial parasites, only Plasmodium falciparum can still be cured by hydroxychloroquine, and growing resistance limits the geographic locations where this drug can be used effectively.1,2
HISTORICAL BACKGROUND
Antimalarial drugs were discovered shortly before World War II. Their production was industrialized during the war because malaria was a leading cause of disease among soldiers, especially those deployed to the South Pacific.3
Atabrine (quinacrine), the first antimalarial widely used, had numerous side effects including yellowing of the skin. Aggressive research efforts to develop an alternative led to field testing of one of its derivative compounds, chloroquine, by the US Army in 1943. Continued chemical modification would create hydroxychloroquine, introduced in 1955.
A serendipitous consequence of the mass use of antimalarials during World War II was the discovery that they could be used to treat inflammatory arthritis and lupus. Eight years after the war ended, Shee4 reported that chloroquine had a beneficial effect on lupus and rheumatoid arthritis in US soldiers. Hydroxychloroquine is now the most commonly prescribed antimalarial for treatment of autoimmune disease.
HOW HYDROXYCHLOROQUINE WORKS
The primary mechanism by which hydroxychloroquine modulates systemic lupus erythematosus is by suppressing activation of Toll-like receptors, which exist on the surface of endosomes and play a significant role in the innate immune response and in autoimmune disease. Their activation is necessary for the expression of interferon-regulated genes and production of tumor necrosis factor alpha, which are key in the cell-mediated inflammatory response.
Antimalarial drugs such as hydroxychloroquine prevent Toll-like receptor activation by binding directly to nucleic acids in the activation pathway.5 In vitro studies show that blocking this pathway blunts the body’s primary cell-mediated inflammatory response; in vivo studies show that use of hydroxychloroquine is strongly correlated with a reduction in interferon alpha levels.6 The powerful effect of hydroxychloroquine on the cell-mediated pattern of inflammation found in lupus is consistent with this theory.
It was previously hypothesized that the immune-modulating effects of hydroxychloroquine were associated with a more general dysregulation of cellular lysosomes through inhibition of proteolysis or changes in cellular pH.7 This theory has since been displaced by the more specific and elegant mechanism described above.5
HOW WELL DOES IT WORK?
Benefit in systemic lupus erythematosus
Hydroxychloroquine has consistently demonstrated significant and multifaceted benefit in patients with systemic lupus erythematosus.
A systematic review of 95 articles8 concluded that this drug decreases lupus flares and decreases mortality rates in lupus patients by at least 50%, with a high level of evidence. Beneficial effects that had a moderate level of evidence were an increase in bone mineral density, fewer thrombotic events, and fewer cases of irreversible organ damage.
The preventive effect of hydroxychloroquine on thrombosis in lupus patients has been consistently demonstrated and is one of the key reasons the drug is considered a cornerstone of therapy in this disease.9 A nested case-control study of patients with lupus and thromboembolism demonstrated an odds ratio of 0.31 and relative risk reduction of 68% for those using antimalarials.10
Benefit in antiphospholipid antibody syndrome
Hydroxychloroquine prevents thrombosis in other diseases as well. For example, it has been shown to reduce the incidence of thrombotic events in patients with primary antiphospholipid syndrome.
In a retrospective cohort study in 114 patients with this disease, hydroxychloroquine significantly reduced the incidence of arterial thrombotic events over 10 years of follow-up (recurrence incidence 0 in those treated with hydroxychloroquine vs 1.14% in those not treated).11 The study also tracked levels of antiphospholipid antibodies and reported that hydroxychloroquine significantly reduced the levels of antibodies to cardiolipin and beta-2 glycoprotein 1, both implicated in the pathology of thrombosis.11
In vitro studies have also demonstrated that hydroxychloroquine can modulate a dysregulated inflammatory system to reduce thrombosis. For example, it has been shown that hydroxychloroquine can reverse platelet activation by antiphospholipid antibodies, prevent linking of antibody complexes to cell membranes, and promote proper membrane protein expression, thereby reducing the thrombotic qualities of antiphospholipid antibodies and even improving clearance times of antiphospholipid-related thrombi.12
Benefit in rheumatoid arthritis
Though there is less evidence, hydroxychloroquine has also shown benefit in rheumatoid arthritis, where it can be used by itself in mild disease or as part of combination therapy with active arthritis. Compared with biologic therapy in patients with early aggressive rheumatoid arthritis, triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine was nearly as effective in terms of quality of life, and it cost only one-third as much, saving $20,000 per year of therapy per patient.13
Hydroxychloroquine has also been compared directly with chloroquine, its closest relation, in a large study incorporating patients with rheumatoid arthritis and patients with systemic lupus erythematosus. Patients using chloroquine experienced significantly more side effects, though it did prove marginally more effective.14
No benefit shown in Sjögren syndrome
Unfortunately, despite widespread use, hydroxychloroquine has not demonstrated positive clinical effects when used to treat primary Sjögren syndrome. Most notably, a 2014 randomized controlled trial of hydroxychloroquine vs placebo in 120 Sjögren patients found no significant improvement in primary symptoms of dryness, pain, or fatigue after 6 months of therapy.15
Metabolic benefits
Unexpectedly, hydroxychloroquine is associated with multiple metabolic benefits including improved lipid profiles and lower blood glucose levels. These findings, in addition to a reduced incidence of thrombosis, were initially reported in the Baltimore Lupus Cohort in 1996.16 Specifically, longitudinal evaluation of a cohort of lupus patients showed that hydroxychloroquine use was associated with a 7.6% reduction in total cholesterol and a 13.7% reduction in low-density lipoprotein cholesterol (LDL-C) over 3 months of therapy.17
Similar findings, including a reduction in LDL-C and an increase in high-density lipoprotein cholesterol, were strongly associated with the addition of hydroxychloroquine to methotrexate or to methotrexate and etanercept in a large cohort of rheumatoid arthritis patients followed over 2 years of therapy.18
In nondiabetic women with systemic lupus erythematosus or rheumatoid arthritis, average blood glucose was significantly lower in those taking hydroxychloroquine than in nonusers. The incidence of insulin resistance was also lower, but the difference was not statistically significant.19
Some have suggested that hydroxychloroquine may prevent diabetes mellitus. In a retrospective case series, compared with rheumatoid arthritis patients not taking the drug, patients treated with hydroxychloroquine for more than 4 years had a 25% lower risk of developing diabetes mellitus.20
In view of these metabolic benefits, especially regarding lipid regulation, and the above described antithrombotic properties of hydroxychloroquine, some researchers have recently hypothesized that hydroxychloroquine may be of benefit in patients with coronary artery disease.21 They suggested that the inflammatory contribution to the mechanism of coronary artery disease could be lessened by hydroxychloroquine even in patients without lupus erythematosus or rheumatoid arthritis.
PHARMACOLOGIC PROPERTIES
Understanding the pharmacologic properties of hydroxychloroquine is key to using it appropriately in clinical practice.
The half-life of elimination of hydroxychloroquine is 40 to 50 days, with half of the drug excreted renally in a concentration-dependent fashion.22,23 The drug reaches 95% of its steady-state concentration by about 6 months of therapy. Shorter durations of therapy do not provide adequate time for the drug to achieve steady-state concentration and may not allow patients and providers time to see its full clinical results. Therefore, its manufacturers recommend a 6-month trial of therapy to adequately determine if the drug improves symptoms.1
The oral bioavailability of hydroxychloroquine is about 75%, but pharmacokinetics vary among individuals.22,23 It has been suggested that this variability affects the efficacy of hydroxychloroquine. In a study of 300 patients with cutaneous lupus erythematosus, those whose treatment failed had significantly lower blood concentrations of hydroxychloroquine, while those who achieved complete remission had significantly higher concentrations.24
Another study found that titrating doses to target therapeutic blood concentrations can reduce disease activity in cutaneous lupus erythematosus.25 Measuring the blood concentration of hydroxychloroquine is not common in clinical practice but may have a role in select patients in whom initial therapy using a standard dosing regimen does not produce the desired results.
HOW SAFE IS HYDROXYCHLOROQUINE?
Hydroxychloroquine has numerous adverse effects, necessitating vigilance on the part of the prescriber. Most commonly reported are retinopathy, hyperpigmentation, myopathy, and skin reactions.1
Retinopathy
Retinopathy’s irreversibility—the threat of permanent vision loss—and its substantial prevalence in patients with a large drug exposure history, have marked retinopathy as the most concerning potential toxicity. The risk of ocular toxicity increases with the cumulative hydroxychloroquine dose. The prevalence of retinopathy in those using the drug less than 10 years is less than 2%; in contrast, the prevalence in patients with more than 20 years of exposure is reported to be as high as 20%.26
The American Academy of Ophthalmology has long stated that retinopathy is a significant risk of hydroxychloroquine therapy and that patients taking hydroxychloroquine should therefore undergo routine retinal and visual field screening by an ophthalmologist.
Currently, initial screening followed by yearly screening beginning 5 years thereafter is recommended for patients at low risk of toxicity (Table 1).27 Patients determined by an ophthalmologist to be at higher risk of retinopathy should be screened yearly. As identified by the American Academy of Ophthalmology, major risk factors for retinopathy include duration of use, concomitant tamoxifen exposure, significant renal disease, and preexisting retinal and macular disease.26,28
Recommendations for hydroxychloroquine dosing and screening were recently revised, for 2 reasons. Initially, its manufacturers recommended that hydroxychloroquine dosage be no higher than 6.5 mg/kg of ideal body weight to prevent retinopathy.1,29,30 However, it has recently been demonstrated that real body weight is a better predictor of risk of retinopathy than ideal body weight when dosing hydroxychloroquine, perhaps because of the increasing variance of real body weight in our patient population.26
Further, an atypical pattern of retinopathy called pericentral retinopathy is more common in Asians. A study of about 200 patients with a history of hydroxychloroquine retinopathy, including 36 Asian patients, found that the pericentral pattern occurred in half the Asian patients but only 2% of the white patients.31 The mechanism for this finding is unclear, but because pericentral retinopathy spares the macula, it can be missed using standard screening methods. Therefore, the American Academy of Ophthalmology now recommends that the dose limit be reduced from 6.5 mg/kg of ideal body weight to no more than 5.0 mg/kg of real body weight (Table 2).28
It is also recommended that screening methods such as automated visual fields and optical coherence tomography extend their fields beyond the macula in Asian patients to ensure that pericentral retinopathy is not missed.28
Optical coherence tomography is a particularly useful tool in the ocular evaluation of patients taking hydroxychloroquine. It can detect subtle changes such as thinning of the foveal photoreceptor outer segment, thickening of the retinal pigment epithelium, and loss of the macular ganglion cell–inner plexiform layer before there are visible signs of retinopathy and before symptoms arise.32
Currently, these guidelines are underutilized in clinical practice. Physician adherence to ophthalmologic guidelines is reported at about 50%.33 This statistic is jarring, given the potential for permanent loss of vision in those with hydroxychloroquine-mediated retinopathy, and demonstrates the importance of reinforcing proper understanding of the use of hydroxychloroquine in clinical practice.
Other adverse effects
Cutaneous hyperpigmentation can occur with hydroxychloroquine use (Figure 1). The hyperpigmentation appears to be due to local bruising following deposition of iron in the soft tissue.
While the pigmentation may persist permanently and cause an undesirable cosmetic effect, it has not been associated with other adverse outcomes.
Myopathy is a rare adverse effect. In one case series, 3 of 214 patients treated with hydroxychloroquine developed hydroxychloroquine-induced myopathy.35 Over the duration of their therapy, this was equivalent to an incidence of 1 case of myopathy in 100 patient-years of therapy. Myopathy improves with discontinuation of therapy, though it can persist for weeks, likely because of hydroxychloroquine’s prolonged elimination half-life.
Cardiomyopathy, specifically neurocardiomyopathy, is also an extremely rare adverse effect of hydroxychloroquine use. The mechanism is believed to be associated with the effect of hydroxychloroquine on lysosomal action, leading to an acquired lysosomal storage disorder with the typical cardiac hypertrophy and conduction abnormalities associated with this family of diseases.36
Acute generalized exanthematous pustulosis is another rare complication of hydroxychloroquine therapy. The appearance of the reaction is similar to that of pustular psoriasis, with pustules overlying flaking and scaling skin. It usually resolves within 2 weeks after cessation of hydroxychloroquine therapy. In a select few cases, the reaction persists or waxes and wanes over a period of weeks to months, and longer durations of recovery are thought to be due to hydroxychloroquine’s long half-life, as in hydroxychloroquine-induced myopathy.37
In view of this rare reaction, manufacturers of hydroxychloroquine recommend caution when using the drug in patients with psoriasis.1
Hematologic abnormalities. In very rare cases, hydroxychloroquine is associated with hematologic abnormalities including agranulocytosis, anemia, aplastic anemia, leukopenia, and thrombocytopenia.1
While no specific guidelines exist, caution is warranted when using hydroxychloroquine in patients with porphyria. Additionally, hydroxychloroquine and other antimalarials including primaquine have been associated with hemolysis in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency. The risk of hemolysis is generally considered low except at high hydroxychloroquine doses in patients with severe G6PD deficiency.38
For the above reasons, manufacturers recommended baseline and routine blood counts, and some providers screen patients for G6PD deficiency when prescribing hydroxychloroquine (Table 3).
PREGNANCY
Hydroxychloroquine is in pregnancy category C. Information is limited, and in view of the risks, the manufacturer says that it should be avoided in pregnancy.1 Nevertheless, it is generally considered safe during pregnancy, and its benefits may make it acceptable to continue in a patient who becomes pregnant, in spite of the possible risks.
We favor continuing hydroxychloroquine. This drug has been associated with improved maternal and fetal outcomes in lupus patients. Its use during pregnancy has not been associated with congenital malformations. The adverse visual effects of long-term hydroxychloroquine use, namely retinopathy, have never been reported in children as a consequence of exposure in utero.
In addition, hydroxychloroquine is transmitted only in minute quantities in breast milk.39 In pregnant women with systemic lupus erythematosus, hydroxychloroquine was associated with a lower risk of adverse outcomes, including preterm delivery and intrauterine growth restriction.40 However, hydroxychloroquine is far more toxic when ingested directly by infants than in adults.1
Maternal outcomes are also improved with the use of hydroxychloroquine. Stopping hydroxychloroquine during pregnancy in women with systemic lupus erythematosus is associated with significantly higher disease activity—fully twice as high as in those who continue hydroxychloroquine.41 These study results were corroborated in a small randomized trial in which pregnant women with lupus on placebo had significantly higher lupus disease activity scores than those pregnant women who were given hydroxychloroquine.42 The women taking hydroxychloroquine experienced no severe lupus flares for the duration of their pregnancies.
These findings suggest not only that hydroxychloroquine is safe in pregnancy, but also that it should be continued in lupus patients during pregnancy to prevent disease flares and adverse fetal outcomes.
AREAS OF UNCERTAINTY
Benefit in preclinical lupus?
Hydroxychloroquine has a consistently profound effect on outcomes in systemic lupus erythematosus. These findings, in addition to the more widespread use of antibody screening, have led to suggestions that hydroxychloroquine could be of benefit even before systemic lupus erythematosus is diagnosed.
A study in US military personnel found that patients taking hydroxychloroquine experienced a significantly longer lag time between first reported clinical symptoms of lupus and official diagnosis compared with matched controls who also went on to develop the disease, averaging 1.08 vs 0.29 years to disease classification.43 Those who used hydroxychloroquine also had lower rates of autoantibody accumulation. Therefore, hydroxychloroquine could be of benefit in carefully selected candidates at high risk of developing systemic lupus erythematosus.
The beneficial effects of hydroxychloroquine on patients with lupus and rheumatoid arthritis, in terms of primary measures of disease activity and secondary outcomes, were discovered fortuitously and were not the original intended targets of the drug. Because of its versatility, there are numerous other disease states in which hydroxychloroquine has shown a degree of benefit or has shown a potential for benefit.
Antiviral activity?
It has been suggested that antimalarial drugs could serve as adjunctive therapies against filoviruses such as Marburg and Ebola. There is a small body of in vitro and in vivo evidence that hydroxychloroquine could temper severe systemic inflammatory responses to filoviruses both through dysregulation of lysosomes and lysosomal pH (filoviruses have a pH-dependent mechanism of action) and through decreased production of tumor necrosis factor alpha and interferons. Heavy burdens of interferons and tumor necrosis factor alpha are associated with increased mortality rates in those infected with filoviruses.44
Antineoplastic activity?
Hydroxychloroquine has undergone in vitro testing as an adjunct to cancer therapies. There are several mechanisms by which it is theorized that hydroxychloroquine could target malignant cells, including inhibition of multidrug resistance pumps or autophagy, improvement of chemotherapy cell penetration, potentiation of presentation of major histocompatibility complexes, or even intercalation directly into DNA.45,46 However, it can also impair natural anticancer immunity and may allow cancer cells better nutrient supply through vascular effects.
In vitro studies have shown tumoricidal effects in lymphoma and melanoma, and inhibition of growth in lung, colon, breast, cervix, larynx, liver, and prostate cancers. In vivo studies have shown that hydroxychloroquine in high doses can prolong survival in glioblastoma.45
Unfortunately, all of these theorized or observed effects are dose-dependent and likely require doses that exceed currently recommended maximums.
Negative chronotropic effect?
Hydroxychloroquine has been found to decrease the resting heart rate in a cumulative dose-dependent fashion.47 Further, hydroxychloroquine has been known to increase digoxin levels, and the medications should not be used in combination.1
Whether the decrease in resting heart rate is associated with harm or benefit and whether the effect is significant enough to be considered when implementing therapy remain unanswered and deserve further investigation, as does the primary use of hydroxychloroquine for beneficial lipid and glucose reduction in patients who are otherwise healthy.
CASE CONCLUSION
The patient described at the beginning of this article was provided with information on the risks and benefits of hydroxychloroquine for treatment of her arthritis and rash suggestive of mild systemic lupus, and she opted to begin therapy. Her baseline eye screening was within normal limits. Based on her weight of 62 kg, she was started on 300 mg of hydroxychloroquine daily.
She had no significant adverse effects from the medication and reported slow improvement in her rash and joint complaints over the next 2 months. She remained on hydroxychloroquine over the next year without adverse effects or new evidence of autoimmune disease.
A 29-year-old African American woman presents with a photosensitive malar rash, fatigue, morning stiffness, and swelling in her hands. She is found to have elevated antinuclear antibody at a titer of 1:320. A complete blood cell count demonstrates leukopenia and thrombocytopenia. Results of renal function testing and urinalysis are within normal limits. She has no other medical problems and no history of blood clots or pregnancy loss.
Her arthritis and rash suggest systemic lupus erythematosus. She is counseled to avoid sun exposure, and treatment with hydroxychloroquine is considered.
WHAT IS HYDROXYCHLOROQUINE?
Hydroxychloroquine was developed to treat malaria but was later found to have immunomodulatory properties. It is now approved by the US Food and Drug Administration for treatment of discoid lupus, systemic lupus erythematosus, and rheumatoid arthritis. It is also approved to treat malaria; however, of the several malarial parasites, only Plasmodium falciparum can still be cured by hydroxychloroquine, and growing resistance limits the geographic locations where this drug can be used effectively.1,2
HISTORICAL BACKGROUND
Antimalarial drugs were discovered shortly before World War II. Their production was industrialized during the war because malaria was a leading cause of disease among soldiers, especially those deployed to the South Pacific.3
Atabrine (quinacrine), the first antimalarial widely used, had numerous side effects including yellowing of the skin. Aggressive research efforts to develop an alternative led to field testing of one of its derivative compounds, chloroquine, by the US Army in 1943. Continued chemical modification would create hydroxychloroquine, introduced in 1955.
A serendipitous consequence of the mass use of antimalarials during World War II was the discovery that they could be used to treat inflammatory arthritis and lupus. Eight years after the war ended, Shee4 reported that chloroquine had a beneficial effect on lupus and rheumatoid arthritis in US soldiers. Hydroxychloroquine is now the most commonly prescribed antimalarial for treatment of autoimmune disease.
HOW HYDROXYCHLOROQUINE WORKS
The primary mechanism by which hydroxychloroquine modulates systemic lupus erythematosus is by suppressing activation of Toll-like receptors, which exist on the surface of endosomes and play a significant role in the innate immune response and in autoimmune disease. Their activation is necessary for the expression of interferon-regulated genes and production of tumor necrosis factor alpha, which are key in the cell-mediated inflammatory response.
Antimalarial drugs such as hydroxychloroquine prevent Toll-like receptor activation by binding directly to nucleic acids in the activation pathway.5 In vitro studies show that blocking this pathway blunts the body’s primary cell-mediated inflammatory response; in vivo studies show that use of hydroxychloroquine is strongly correlated with a reduction in interferon alpha levels.6 The powerful effect of hydroxychloroquine on the cell-mediated pattern of inflammation found in lupus is consistent with this theory.
It was previously hypothesized that the immune-modulating effects of hydroxychloroquine were associated with a more general dysregulation of cellular lysosomes through inhibition of proteolysis or changes in cellular pH.7 This theory has since been displaced by the more specific and elegant mechanism described above.5
HOW WELL DOES IT WORK?
Benefit in systemic lupus erythematosus
Hydroxychloroquine has consistently demonstrated significant and multifaceted benefit in patients with systemic lupus erythematosus.
A systematic review of 95 articles8 concluded that this drug decreases lupus flares and decreases mortality rates in lupus patients by at least 50%, with a high level of evidence. Beneficial effects that had a moderate level of evidence were an increase in bone mineral density, fewer thrombotic events, and fewer cases of irreversible organ damage.
The preventive effect of hydroxychloroquine on thrombosis in lupus patients has been consistently demonstrated and is one of the key reasons the drug is considered a cornerstone of therapy in this disease.9 A nested case-control study of patients with lupus and thromboembolism demonstrated an odds ratio of 0.31 and relative risk reduction of 68% for those using antimalarials.10
Benefit in antiphospholipid antibody syndrome
Hydroxychloroquine prevents thrombosis in other diseases as well. For example, it has been shown to reduce the incidence of thrombotic events in patients with primary antiphospholipid syndrome.
In a retrospective cohort study in 114 patients with this disease, hydroxychloroquine significantly reduced the incidence of arterial thrombotic events over 10 years of follow-up (recurrence incidence 0 in those treated with hydroxychloroquine vs 1.14% in those not treated).11 The study also tracked levels of antiphospholipid antibodies and reported that hydroxychloroquine significantly reduced the levels of antibodies to cardiolipin and beta-2 glycoprotein 1, both implicated in the pathology of thrombosis.11
In vitro studies have also demonstrated that hydroxychloroquine can modulate a dysregulated inflammatory system to reduce thrombosis. For example, it has been shown that hydroxychloroquine can reverse platelet activation by antiphospholipid antibodies, prevent linking of antibody complexes to cell membranes, and promote proper membrane protein expression, thereby reducing the thrombotic qualities of antiphospholipid antibodies and even improving clearance times of antiphospholipid-related thrombi.12
Benefit in rheumatoid arthritis
Though there is less evidence, hydroxychloroquine has also shown benefit in rheumatoid arthritis, where it can be used by itself in mild disease or as part of combination therapy with active arthritis. Compared with biologic therapy in patients with early aggressive rheumatoid arthritis, triple therapy with methotrexate, sulfasalazine, and hydroxychloroquine was nearly as effective in terms of quality of life, and it cost only one-third as much, saving $20,000 per year of therapy per patient.13
Hydroxychloroquine has also been compared directly with chloroquine, its closest relation, in a large study incorporating patients with rheumatoid arthritis and patients with systemic lupus erythematosus. Patients using chloroquine experienced significantly more side effects, though it did prove marginally more effective.14
No benefit shown in Sjögren syndrome
Unfortunately, despite widespread use, hydroxychloroquine has not demonstrated positive clinical effects when used to treat primary Sjögren syndrome. Most notably, a 2014 randomized controlled trial of hydroxychloroquine vs placebo in 120 Sjögren patients found no significant improvement in primary symptoms of dryness, pain, or fatigue after 6 months of therapy.15
Metabolic benefits
Unexpectedly, hydroxychloroquine is associated with multiple metabolic benefits including improved lipid profiles and lower blood glucose levels. These findings, in addition to a reduced incidence of thrombosis, were initially reported in the Baltimore Lupus Cohort in 1996.16 Specifically, longitudinal evaluation of a cohort of lupus patients showed that hydroxychloroquine use was associated with a 7.6% reduction in total cholesterol and a 13.7% reduction in low-density lipoprotein cholesterol (LDL-C) over 3 months of therapy.17
Similar findings, including a reduction in LDL-C and an increase in high-density lipoprotein cholesterol, were strongly associated with the addition of hydroxychloroquine to methotrexate or to methotrexate and etanercept in a large cohort of rheumatoid arthritis patients followed over 2 years of therapy.18
In nondiabetic women with systemic lupus erythematosus or rheumatoid arthritis, average blood glucose was significantly lower in those taking hydroxychloroquine than in nonusers. The incidence of insulin resistance was also lower, but the difference was not statistically significant.19
Some have suggested that hydroxychloroquine may prevent diabetes mellitus. In a retrospective case series, compared with rheumatoid arthritis patients not taking the drug, patients treated with hydroxychloroquine for more than 4 years had a 25% lower risk of developing diabetes mellitus.20
In view of these metabolic benefits, especially regarding lipid regulation, and the above described antithrombotic properties of hydroxychloroquine, some researchers have recently hypothesized that hydroxychloroquine may be of benefit in patients with coronary artery disease.21 They suggested that the inflammatory contribution to the mechanism of coronary artery disease could be lessened by hydroxychloroquine even in patients without lupus erythematosus or rheumatoid arthritis.
PHARMACOLOGIC PROPERTIES
Understanding the pharmacologic properties of hydroxychloroquine is key to using it appropriately in clinical practice.
The half-life of elimination of hydroxychloroquine is 40 to 50 days, with half of the drug excreted renally in a concentration-dependent fashion.22,23 The drug reaches 95% of its steady-state concentration by about 6 months of therapy. Shorter durations of therapy do not provide adequate time for the drug to achieve steady-state concentration and may not allow patients and providers time to see its full clinical results. Therefore, its manufacturers recommend a 6-month trial of therapy to adequately determine if the drug improves symptoms.1
The oral bioavailability of hydroxychloroquine is about 75%, but pharmacokinetics vary among individuals.22,23 It has been suggested that this variability affects the efficacy of hydroxychloroquine. In a study of 300 patients with cutaneous lupus erythematosus, those whose treatment failed had significantly lower blood concentrations of hydroxychloroquine, while those who achieved complete remission had significantly higher concentrations.24
Another study found that titrating doses to target therapeutic blood concentrations can reduce disease activity in cutaneous lupus erythematosus.25 Measuring the blood concentration of hydroxychloroquine is not common in clinical practice but may have a role in select patients in whom initial therapy using a standard dosing regimen does not produce the desired results.
HOW SAFE IS HYDROXYCHLOROQUINE?
Hydroxychloroquine has numerous adverse effects, necessitating vigilance on the part of the prescriber. Most commonly reported are retinopathy, hyperpigmentation, myopathy, and skin reactions.1
Retinopathy
Retinopathy’s irreversibility—the threat of permanent vision loss—and its substantial prevalence in patients with a large drug exposure history, have marked retinopathy as the most concerning potential toxicity. The risk of ocular toxicity increases with the cumulative hydroxychloroquine dose. The prevalence of retinopathy in those using the drug less than 10 years is less than 2%; in contrast, the prevalence in patients with more than 20 years of exposure is reported to be as high as 20%.26
The American Academy of Ophthalmology has long stated that retinopathy is a significant risk of hydroxychloroquine therapy and that patients taking hydroxychloroquine should therefore undergo routine retinal and visual field screening by an ophthalmologist.
Currently, initial screening followed by yearly screening beginning 5 years thereafter is recommended for patients at low risk of toxicity (Table 1).27 Patients determined by an ophthalmologist to be at higher risk of retinopathy should be screened yearly. As identified by the American Academy of Ophthalmology, major risk factors for retinopathy include duration of use, concomitant tamoxifen exposure, significant renal disease, and preexisting retinal and macular disease.26,28
Recommendations for hydroxychloroquine dosing and screening were recently revised, for 2 reasons. Initially, its manufacturers recommended that hydroxychloroquine dosage be no higher than 6.5 mg/kg of ideal body weight to prevent retinopathy.1,29,30 However, it has recently been demonstrated that real body weight is a better predictor of risk of retinopathy than ideal body weight when dosing hydroxychloroquine, perhaps because of the increasing variance of real body weight in our patient population.26
Further, an atypical pattern of retinopathy called pericentral retinopathy is more common in Asians. A study of about 200 patients with a history of hydroxychloroquine retinopathy, including 36 Asian patients, found that the pericentral pattern occurred in half the Asian patients but only 2% of the white patients.31 The mechanism for this finding is unclear, but because pericentral retinopathy spares the macula, it can be missed using standard screening methods. Therefore, the American Academy of Ophthalmology now recommends that the dose limit be reduced from 6.5 mg/kg of ideal body weight to no more than 5.0 mg/kg of real body weight (Table 2).28
It is also recommended that screening methods such as automated visual fields and optical coherence tomography extend their fields beyond the macula in Asian patients to ensure that pericentral retinopathy is not missed.28
Optical coherence tomography is a particularly useful tool in the ocular evaluation of patients taking hydroxychloroquine. It can detect subtle changes such as thinning of the foveal photoreceptor outer segment, thickening of the retinal pigment epithelium, and loss of the macular ganglion cell–inner plexiform layer before there are visible signs of retinopathy and before symptoms arise.32
Currently, these guidelines are underutilized in clinical practice. Physician adherence to ophthalmologic guidelines is reported at about 50%.33 This statistic is jarring, given the potential for permanent loss of vision in those with hydroxychloroquine-mediated retinopathy, and demonstrates the importance of reinforcing proper understanding of the use of hydroxychloroquine in clinical practice.
Other adverse effects
Cutaneous hyperpigmentation can occur with hydroxychloroquine use (Figure 1). The hyperpigmentation appears to be due to local bruising following deposition of iron in the soft tissue.
While the pigmentation may persist permanently and cause an undesirable cosmetic effect, it has not been associated with other adverse outcomes.
Myopathy is a rare adverse effect. In one case series, 3 of 214 patients treated with hydroxychloroquine developed hydroxychloroquine-induced myopathy.35 Over the duration of their therapy, this was equivalent to an incidence of 1 case of myopathy in 100 patient-years of therapy. Myopathy improves with discontinuation of therapy, though it can persist for weeks, likely because of hydroxychloroquine’s prolonged elimination half-life.
Cardiomyopathy, specifically neurocardiomyopathy, is also an extremely rare adverse effect of hydroxychloroquine use. The mechanism is believed to be associated with the effect of hydroxychloroquine on lysosomal action, leading to an acquired lysosomal storage disorder with the typical cardiac hypertrophy and conduction abnormalities associated with this family of diseases.36
Acute generalized exanthematous pustulosis is another rare complication of hydroxychloroquine therapy. The appearance of the reaction is similar to that of pustular psoriasis, with pustules overlying flaking and scaling skin. It usually resolves within 2 weeks after cessation of hydroxychloroquine therapy. In a select few cases, the reaction persists or waxes and wanes over a period of weeks to months, and longer durations of recovery are thought to be due to hydroxychloroquine’s long half-life, as in hydroxychloroquine-induced myopathy.37
In view of this rare reaction, manufacturers of hydroxychloroquine recommend caution when using the drug in patients with psoriasis.1
Hematologic abnormalities. In very rare cases, hydroxychloroquine is associated with hematologic abnormalities including agranulocytosis, anemia, aplastic anemia, leukopenia, and thrombocytopenia.1
While no specific guidelines exist, caution is warranted when using hydroxychloroquine in patients with porphyria. Additionally, hydroxychloroquine and other antimalarials including primaquine have been associated with hemolysis in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency. The risk of hemolysis is generally considered low except at high hydroxychloroquine doses in patients with severe G6PD deficiency.38
For the above reasons, manufacturers recommended baseline and routine blood counts, and some providers screen patients for G6PD deficiency when prescribing hydroxychloroquine (Table 3).
PREGNANCY
Hydroxychloroquine is in pregnancy category C. Information is limited, and in view of the risks, the manufacturer says that it should be avoided in pregnancy.1 Nevertheless, it is generally considered safe during pregnancy, and its benefits may make it acceptable to continue in a patient who becomes pregnant, in spite of the possible risks.
We favor continuing hydroxychloroquine. This drug has been associated with improved maternal and fetal outcomes in lupus patients. Its use during pregnancy has not been associated with congenital malformations. The adverse visual effects of long-term hydroxychloroquine use, namely retinopathy, have never been reported in children as a consequence of exposure in utero.
In addition, hydroxychloroquine is transmitted only in minute quantities in breast milk.39 In pregnant women with systemic lupus erythematosus, hydroxychloroquine was associated with a lower risk of adverse outcomes, including preterm delivery and intrauterine growth restriction.40 However, hydroxychloroquine is far more toxic when ingested directly by infants than in adults.1
Maternal outcomes are also improved with the use of hydroxychloroquine. Stopping hydroxychloroquine during pregnancy in women with systemic lupus erythematosus is associated with significantly higher disease activity—fully twice as high as in those who continue hydroxychloroquine.41 These study results were corroborated in a small randomized trial in which pregnant women with lupus on placebo had significantly higher lupus disease activity scores than those pregnant women who were given hydroxychloroquine.42 The women taking hydroxychloroquine experienced no severe lupus flares for the duration of their pregnancies.
These findings suggest not only that hydroxychloroquine is safe in pregnancy, but also that it should be continued in lupus patients during pregnancy to prevent disease flares and adverse fetal outcomes.
AREAS OF UNCERTAINTY
Benefit in preclinical lupus?
Hydroxychloroquine has a consistently profound effect on outcomes in systemic lupus erythematosus. These findings, in addition to the more widespread use of antibody screening, have led to suggestions that hydroxychloroquine could be of benefit even before systemic lupus erythematosus is diagnosed.
A study in US military personnel found that patients taking hydroxychloroquine experienced a significantly longer lag time between first reported clinical symptoms of lupus and official diagnosis compared with matched controls who also went on to develop the disease, averaging 1.08 vs 0.29 years to disease classification.43 Those who used hydroxychloroquine also had lower rates of autoantibody accumulation. Therefore, hydroxychloroquine could be of benefit in carefully selected candidates at high risk of developing systemic lupus erythematosus.
The beneficial effects of hydroxychloroquine on patients with lupus and rheumatoid arthritis, in terms of primary measures of disease activity and secondary outcomes, were discovered fortuitously and were not the original intended targets of the drug. Because of its versatility, there are numerous other disease states in which hydroxychloroquine has shown a degree of benefit or has shown a potential for benefit.
Antiviral activity?
It has been suggested that antimalarial drugs could serve as adjunctive therapies against filoviruses such as Marburg and Ebola. There is a small body of in vitro and in vivo evidence that hydroxychloroquine could temper severe systemic inflammatory responses to filoviruses both through dysregulation of lysosomes and lysosomal pH (filoviruses have a pH-dependent mechanism of action) and through decreased production of tumor necrosis factor alpha and interferons. Heavy burdens of interferons and tumor necrosis factor alpha are associated with increased mortality rates in those infected with filoviruses.44
Antineoplastic activity?
Hydroxychloroquine has undergone in vitro testing as an adjunct to cancer therapies. There are several mechanisms by which it is theorized that hydroxychloroquine could target malignant cells, including inhibition of multidrug resistance pumps or autophagy, improvement of chemotherapy cell penetration, potentiation of presentation of major histocompatibility complexes, or even intercalation directly into DNA.45,46 However, it can also impair natural anticancer immunity and may allow cancer cells better nutrient supply through vascular effects.
In vitro studies have shown tumoricidal effects in lymphoma and melanoma, and inhibition of growth in lung, colon, breast, cervix, larynx, liver, and prostate cancers. In vivo studies have shown that hydroxychloroquine in high doses can prolong survival in glioblastoma.45
Unfortunately, all of these theorized or observed effects are dose-dependent and likely require doses that exceed currently recommended maximums.
Negative chronotropic effect?
Hydroxychloroquine has been found to decrease the resting heart rate in a cumulative dose-dependent fashion.47 Further, hydroxychloroquine has been known to increase digoxin levels, and the medications should not be used in combination.1
Whether the decrease in resting heart rate is associated with harm or benefit and whether the effect is significant enough to be considered when implementing therapy remain unanswered and deserve further investigation, as does the primary use of hydroxychloroquine for beneficial lipid and glucose reduction in patients who are otherwise healthy.
CASE CONCLUSION
The patient described at the beginning of this article was provided with information on the risks and benefits of hydroxychloroquine for treatment of her arthritis and rash suggestive of mild systemic lupus, and she opted to begin therapy. Her baseline eye screening was within normal limits. Based on her weight of 62 kg, she was started on 300 mg of hydroxychloroquine daily.
She had no significant adverse effects from the medication and reported slow improvement in her rash and joint complaints over the next 2 months. She remained on hydroxychloroquine over the next year without adverse effects or new evidence of autoimmune disease.
- Sanofi-Aventis. Product monograph: Plaquenil. http://products.sanofi.ca/en/plaquenil.pdf. Accessed May 2, 2018.
- Centers for Disease Control and Prevention (CDC). Malaria information and prophylaxis, by country. www.cdc.gov/malaria/travelers/country_table/a.html. Accessed May 2, 2018.
- Wallace DJ. The history of antimalarials. Lupus 1996; 5(suppl 1):S2–S3. pmid:8803902
- Shee JC. Lupus erythematosus treated with chloroquine. Lancet 1953; 265(6778):201–202. pmid:13070595
- Kuznik A, Bencina M, Svajger U, Jeras M, Rozman B, Jerala R. Mechanism of endosomal TLR inhibition by antimalarial drugs and imidazoquinolines. J Immunol 2011; 186:4794–4804. doi:10.4049/jimmunol.1000702
- Willis R, Seif AM, McGwin G Jr, et al. Effect of hydroxychloroquine treatment on pro-inflammatory cytokines and disease activity in SLE patients: data from LUMINA, a multiethnic US cohort. Lupus 2012; 21(8):830–835. doi:10.1177/0961203312437270
- Fox R. Anti-malarial drugs: possible mechanisms of action in autoimmune disease and prospects for drug development. Lupus 1996; 5(suppl 1):S4–S10. pmid:8803903
- Ruiz-Irastorza G, Ramos-Casals M, Brito-Zeron P, Khamashta MA. Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann Rheum Dis 2010; 69(1):20–28. doi:10.1136/ard.2008.101766
- Lam NC, Ghetu MV, Bieniek ML. Systemic lupus erythematosus: primary care approach to diagnosis and management. Am Fam Physician 2016; 94(4):284–294. pmid:27548593
- Jung H, Bobba R, Su J, et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum 2010; 62(3):863–868. doi:10.1002/art.27289
- Nuri E, Taraborelli M, Andreoli L, et al. Long-term use of hydroxychloroquine reduces antiphospholipid antibodies levels in patients with primary antiphospholipid syndrome. Immunol Res 2017; 65(1):17–24. doi:10.1007/s12026-016-8812-z
- Erkan D, Aguiar CL, Andrade D, et al. 14th International Congress on Antiphospholipid Antibodies: task force report on antiphospholipid syndrome treatment trends. Autoimmun Rev 2014; 13(6):685–696. doi:10.1016/j.autrev.2014.01.053
- Jalal H, O’Dell JR, Bridges SL Jr, et al. Cost-effectiveness of triple therapy versus etanercept plus methotrexate in early aggressive rheumatoid arthritis. Arthritis Care Res (Hoboken) 2016; 68(12):1751–1757. doi:10.1002/acr.22895
- Avina-Zubieta JA, Galindo-Rodriguez G, Newman S, Suarez-Almazor ME, Russell AS. Long-term effectiveness of antimalarial drugs in rheumatic diseases. Ann Rheum Dis 1998; 57(10):582–587. pmid:9893568
- Gottenberg JE, Ravaud P, Puechal X, et al. Effects of hydroxychloroquine on symptomatic improvement in primary Sjogren syndrome. JAMA 2014; 312(3):249–258. doi:10.1001/jama.2014.7682
- Petri M. Hydroxychloroquine use in the Baltimore Lupus Cohort: effects on lipids, glucose and thrombosis. Lupus 1996; 5(suppl 1):S16–S22. pmid:8803905
- Cairoli E, Rebella M, Danese N, Garra V, Borba EF. Hydroxychloroquine reduces low-density lipoprotein cholesterol levels in systemic lupus erythematosus: a longitudinal evaluation of the lipid-lowering effect. Lupus 2012; 21(11):1178–1182. doi:10.1177/0961203312450084
- Charles-Schoeman C, Wang X, Lee YY, et al. Association of triple therapy with improvement in cholesterol profiles over two-year followup in the treatment of early aggressive rheumatoid arthritis trial. Arthritis Rheumatol 2016; 68(3):577–586. doi:10.1002/art.39502
- Penn SK, Kao AH, Schott LL, et al. Hydroxychloroquine and glycemia in women with rheumatoid arthritis and systemic lupus erythematosus. J Rheumatol 2010; 37(6):1136–1142. doi:10.3899/jrheum.090994
- Wasko MC, Hubert HB, Lingala VB, et al. Hydroxychloroquine and risk of diabetes in patients with rheumatoid arthritis. JAMA 2007; 298(2):187–193. doi:10.1001/jama.298.2.187
- Sun L, Liu M, Li R, et al. Hydroxychloroquine, a promising choice for coronary artery disease? Med Hypotheses 2016; 93:5–7. doi:10.1016/j.mehy.2016.04.045
- Tett SE, Cutler DJ, Day RO, Brown KF. Bioavailability of hydroxychloroquine tablets in healthy volunteers. Br J Clin Pharmacol 1989; 27(6):771–779. pmid:2757893
- Furst DE. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus 1996; 5(suppl 1):S11–S15. pmid:8803904
- Frances C, Cosnes A, Duhaut P, et al. Low blood concentration of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus. Arch Dermatol 2012; 148(4):479–484. doi:10.1001/archdermatol.2011.2558
- Chasset F, Arnaud L, Costedoat-Chalumeau N, Zahr N, Bessis D, Francès C. The effect of increasing the dose of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus: an open-label prospective pilot study. J Am Acad Dermatol 2016; 74(4):693–699.e3. doi:10.1016/j.jaad.2015.09.064
- Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol 2014; 132(12):1453–1460. doi:10.1001/jamaophthalmol.2014.3459
- Committee on Rheumatologic Care. American College of Rheumatology position statement. Screening for hydroxychloroquine retinopathy. www.rheumatology.org/Portals/0/Files/Screening-for-Hydroxychloroquine-Retinopathy-Position-Statement.pdf. Accessed April 2, 2018.
- Marmor MF, Kellner U, Lai TY, Melles RB, Mieler WF; American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology 2016; 123(6):1386–1394. doi:10.1016/j.ophtha.2016.01.058
- Mackenzie AH. Antimalarial drugs for rheumatoid arthritis. Am J Med 1983; 75(6A):48–58. pmid:6362406
- Mackenzie AH. Dose refinements in long-term therapy of rheumatoid arthritis with antimalarials. Am J Med 1983; 75(1A):40–45. pmid:6869410
- Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology 2015; 122(1):110–116. doi:10.1016/j.ophtha.2014.07.018
- Uslu H, Gurler B, Yildirim A, et al. Effect of hydroxychloroquine on the retinal layers: a quantitative evaluation with spectral-domain optical coherence tomography. J Ophthalmol 2016; 2016:8643174. doi:10.1155/2016/8643174
- Au A, Parikh V, Modi YS, Ehlers JP, Schachat AP, Singh RP. Hydroxychloroquine screening practice patterns within a large multispecialty ophthalmic practice. Am J Ophthalmol 2015; 160(3):561–568.e2. doi:10.1016/j.ajo.2015.06.009
- Jallouli M, Frances C, Plette JC, et al; Plaquenil Lupus Systemic Study Group. Hydroxychloroquine-induced pigmentation in patients with systemic lupus erythematosus. JAMA Dermatol 2013; 149(8):935–940. doi:10.1001/jamadermatol.2013.709
- Avina-Zubieta JA, Johnson ES, Suarez-Almazor ME, Russell AS. Incidence of myopathy in patients treated with antimalarials: a report of three cases and review of the literature. Br J Rheumatol 1995; 34(2):166–170. pmid:7704464
- Yogasundaram H, Putko BN, Tien J, et al. Hydroxychloroquine-induced cardiomyopathy: case report, pathophysiology, diagnosis, and treatment. Can J Cardiol 2014; 30:1706–1715. doi:10.1016/j.cjca.2014.08.016
- Pearson KC, Morrell DS, Runge SR, Jolly P. Prolonged pustular eruption from hydroxychloroquine: an unusual case of acute generalized exanthematous pustulosis. Cutis 2016; 97(3):212–216. pmid:27023083
- Youngster I, Arcavi L, Schechmaster R, et al. Medications and glucose-6-phosphate dehydrogenase deficiency: an evidence-based review. Drug Saf 2010; 33(9):713–726. doi:10.2165/11536520-000000000-00000
- Ostensen M, Khamashta M, Lockshin M, et al. Anti-inflammatory and immunosuppressive drugs and reproduction. Arthritis Res Ther 2006; 8(3):209. doi:10.1186/ar1957
- Leroux M, Desveaux C, Parcevaux M, et al. Impact of hydroxychloroquine on preterm delivery and intrauterine growth restriction in pregnant women with systemic lupus erythematosus: a descriptive cohort study. Lupus 2015; 24(13):1384–1391. doi:10.1177/0961203315591027
- Clowse MEB, Magder L, Witter F, Petri M. Hydroxychloroquine in lupus pregnancy. Arthritis Rheum 2006; 54(11):3640–3647. doi:10.1002/art.22159
- Levy RA, Vilela VS, Cataldo MJ, et al. Hydroxychloroquine in lupus pregnancy: double-blind and placebo-controlled study. Lupus 2001; 10(6):401–404. doi:10.1191/096120301678646137
- James JA, Kim-Howard XR, Bruner BF, et al. Hydroxychloroquine sulfate treatment is associated with later onset of systemic lupus erythematosus. Lupus 2007; 16(6):401–409. doi:10.1177/0961203307078579
- Akpovwa H. Chloroquine could be used for the treatment of filoviral infections and other viral infections that emerge or emerged from viruses requiring an acidic pH for infectivity. Cell Biochem Funct 2016; 34(4):191–196. doi:10.1002/cbf.3182
- Pascolo S. Time to use a dose of chloroquine as an adjuvant to anti-cancer chemotherapies. Eur J Pharmacol 2016; 771:139–144. doi:10.1016/j.ejphar.2015.12.017
- Furlong HC, Wessels JM, Guerra MT, Stämpfli MR, Foster WG. Hydroxychloroquine attenuates cigarette smoke induced autophagic signaling in the mouse ovary. Reprod Toxicol 2016; 61:105–113. doi:10.1016/j.reprotox.2016.03.044
- Cairoli E, Danese N, Teliz M, et al. Cumulative dose of hydroxychloroquine is associated with a decrease of resting heart rate in patients with systemic lupus erythematosus: a pilot study. Lupus 2015; 24(11):1204–1209. doi:10.1177/0961203315580870
- Sanofi-Aventis. Product monograph: Plaquenil. http://products.sanofi.ca/en/plaquenil.pdf. Accessed May 2, 2018.
- Centers for Disease Control and Prevention (CDC). Malaria information and prophylaxis, by country. www.cdc.gov/malaria/travelers/country_table/a.html. Accessed May 2, 2018.
- Wallace DJ. The history of antimalarials. Lupus 1996; 5(suppl 1):S2–S3. pmid:8803902
- Shee JC. Lupus erythematosus treated with chloroquine. Lancet 1953; 265(6778):201–202. pmid:13070595
- Kuznik A, Bencina M, Svajger U, Jeras M, Rozman B, Jerala R. Mechanism of endosomal TLR inhibition by antimalarial drugs and imidazoquinolines. J Immunol 2011; 186:4794–4804. doi:10.4049/jimmunol.1000702
- Willis R, Seif AM, McGwin G Jr, et al. Effect of hydroxychloroquine treatment on pro-inflammatory cytokines and disease activity in SLE patients: data from LUMINA, a multiethnic US cohort. Lupus 2012; 21(8):830–835. doi:10.1177/0961203312437270
- Fox R. Anti-malarial drugs: possible mechanisms of action in autoimmune disease and prospects for drug development. Lupus 1996; 5(suppl 1):S4–S10. pmid:8803903
- Ruiz-Irastorza G, Ramos-Casals M, Brito-Zeron P, Khamashta MA. Clinical efficacy and side effects of antimalarials in systemic lupus erythematosus: a systematic review. Ann Rheum Dis 2010; 69(1):20–28. doi:10.1136/ard.2008.101766
- Lam NC, Ghetu MV, Bieniek ML. Systemic lupus erythematosus: primary care approach to diagnosis and management. Am Fam Physician 2016; 94(4):284–294. pmid:27548593
- Jung H, Bobba R, Su J, et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis Rheum 2010; 62(3):863–868. doi:10.1002/art.27289
- Nuri E, Taraborelli M, Andreoli L, et al. Long-term use of hydroxychloroquine reduces antiphospholipid antibodies levels in patients with primary antiphospholipid syndrome. Immunol Res 2017; 65(1):17–24. doi:10.1007/s12026-016-8812-z
- Erkan D, Aguiar CL, Andrade D, et al. 14th International Congress on Antiphospholipid Antibodies: task force report on antiphospholipid syndrome treatment trends. Autoimmun Rev 2014; 13(6):685–696. doi:10.1016/j.autrev.2014.01.053
- Jalal H, O’Dell JR, Bridges SL Jr, et al. Cost-effectiveness of triple therapy versus etanercept plus methotrexate in early aggressive rheumatoid arthritis. Arthritis Care Res (Hoboken) 2016; 68(12):1751–1757. doi:10.1002/acr.22895
- Avina-Zubieta JA, Galindo-Rodriguez G, Newman S, Suarez-Almazor ME, Russell AS. Long-term effectiveness of antimalarial drugs in rheumatic diseases. Ann Rheum Dis 1998; 57(10):582–587. pmid:9893568
- Gottenberg JE, Ravaud P, Puechal X, et al. Effects of hydroxychloroquine on symptomatic improvement in primary Sjogren syndrome. JAMA 2014; 312(3):249–258. doi:10.1001/jama.2014.7682
- Petri M. Hydroxychloroquine use in the Baltimore Lupus Cohort: effects on lipids, glucose and thrombosis. Lupus 1996; 5(suppl 1):S16–S22. pmid:8803905
- Cairoli E, Rebella M, Danese N, Garra V, Borba EF. Hydroxychloroquine reduces low-density lipoprotein cholesterol levels in systemic lupus erythematosus: a longitudinal evaluation of the lipid-lowering effect. Lupus 2012; 21(11):1178–1182. doi:10.1177/0961203312450084
- Charles-Schoeman C, Wang X, Lee YY, et al. Association of triple therapy with improvement in cholesterol profiles over two-year followup in the treatment of early aggressive rheumatoid arthritis trial. Arthritis Rheumatol 2016; 68(3):577–586. doi:10.1002/art.39502
- Penn SK, Kao AH, Schott LL, et al. Hydroxychloroquine and glycemia in women with rheumatoid arthritis and systemic lupus erythematosus. J Rheumatol 2010; 37(6):1136–1142. doi:10.3899/jrheum.090994
- Wasko MC, Hubert HB, Lingala VB, et al. Hydroxychloroquine and risk of diabetes in patients with rheumatoid arthritis. JAMA 2007; 298(2):187–193. doi:10.1001/jama.298.2.187
- Sun L, Liu M, Li R, et al. Hydroxychloroquine, a promising choice for coronary artery disease? Med Hypotheses 2016; 93:5–7. doi:10.1016/j.mehy.2016.04.045
- Tett SE, Cutler DJ, Day RO, Brown KF. Bioavailability of hydroxychloroquine tablets in healthy volunteers. Br J Clin Pharmacol 1989; 27(6):771–779. pmid:2757893
- Furst DE. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus 1996; 5(suppl 1):S11–S15. pmid:8803904
- Frances C, Cosnes A, Duhaut P, et al. Low blood concentration of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus. Arch Dermatol 2012; 148(4):479–484. doi:10.1001/archdermatol.2011.2558
- Chasset F, Arnaud L, Costedoat-Chalumeau N, Zahr N, Bessis D, Francès C. The effect of increasing the dose of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus: an open-label prospective pilot study. J Am Acad Dermatol 2016; 74(4):693–699.e3. doi:10.1016/j.jaad.2015.09.064
- Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol 2014; 132(12):1453–1460. doi:10.1001/jamaophthalmol.2014.3459
- Committee on Rheumatologic Care. American College of Rheumatology position statement. Screening for hydroxychloroquine retinopathy. www.rheumatology.org/Portals/0/Files/Screening-for-Hydroxychloroquine-Retinopathy-Position-Statement.pdf. Accessed April 2, 2018.
- Marmor MF, Kellner U, Lai TY, Melles RB, Mieler WF; American Academy of Ophthalmology. Recommendations on screening for chloroquine and hydroxychloroquine retinopathy (2016 revision). Ophthalmology 2016; 123(6):1386–1394. doi:10.1016/j.ophtha.2016.01.058
- Mackenzie AH. Antimalarial drugs for rheumatoid arthritis. Am J Med 1983; 75(6A):48–58. pmid:6362406
- Mackenzie AH. Dose refinements in long-term therapy of rheumatoid arthritis with antimalarials. Am J Med 1983; 75(1A):40–45. pmid:6869410
- Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology 2015; 122(1):110–116. doi:10.1016/j.ophtha.2014.07.018
- Uslu H, Gurler B, Yildirim A, et al. Effect of hydroxychloroquine on the retinal layers: a quantitative evaluation with spectral-domain optical coherence tomography. J Ophthalmol 2016; 2016:8643174. doi:10.1155/2016/8643174
- Au A, Parikh V, Modi YS, Ehlers JP, Schachat AP, Singh RP. Hydroxychloroquine screening practice patterns within a large multispecialty ophthalmic practice. Am J Ophthalmol 2015; 160(3):561–568.e2. doi:10.1016/j.ajo.2015.06.009
- Jallouli M, Frances C, Plette JC, et al; Plaquenil Lupus Systemic Study Group. Hydroxychloroquine-induced pigmentation in patients with systemic lupus erythematosus. JAMA Dermatol 2013; 149(8):935–940. doi:10.1001/jamadermatol.2013.709
- Avina-Zubieta JA, Johnson ES, Suarez-Almazor ME, Russell AS. Incidence of myopathy in patients treated with antimalarials: a report of three cases and review of the literature. Br J Rheumatol 1995; 34(2):166–170. pmid:7704464
- Yogasundaram H, Putko BN, Tien J, et al. Hydroxychloroquine-induced cardiomyopathy: case report, pathophysiology, diagnosis, and treatment. Can J Cardiol 2014; 30:1706–1715. doi:10.1016/j.cjca.2014.08.016
- Pearson KC, Morrell DS, Runge SR, Jolly P. Prolonged pustular eruption from hydroxychloroquine: an unusual case of acute generalized exanthematous pustulosis. Cutis 2016; 97(3):212–216. pmid:27023083
- Youngster I, Arcavi L, Schechmaster R, et al. Medications and glucose-6-phosphate dehydrogenase deficiency: an evidence-based review. Drug Saf 2010; 33(9):713–726. doi:10.2165/11536520-000000000-00000
- Ostensen M, Khamashta M, Lockshin M, et al. Anti-inflammatory and immunosuppressive drugs and reproduction. Arthritis Res Ther 2006; 8(3):209. doi:10.1186/ar1957
- Leroux M, Desveaux C, Parcevaux M, et al. Impact of hydroxychloroquine on preterm delivery and intrauterine growth restriction in pregnant women with systemic lupus erythematosus: a descriptive cohort study. Lupus 2015; 24(13):1384–1391. doi:10.1177/0961203315591027
- Clowse MEB, Magder L, Witter F, Petri M. Hydroxychloroquine in lupus pregnancy. Arthritis Rheum 2006; 54(11):3640–3647. doi:10.1002/art.22159
- Levy RA, Vilela VS, Cataldo MJ, et al. Hydroxychloroquine in lupus pregnancy: double-blind and placebo-controlled study. Lupus 2001; 10(6):401–404. doi:10.1191/096120301678646137
- James JA, Kim-Howard XR, Bruner BF, et al. Hydroxychloroquine sulfate treatment is associated with later onset of systemic lupus erythematosus. Lupus 2007; 16(6):401–409. doi:10.1177/0961203307078579
- Akpovwa H. Chloroquine could be used for the treatment of filoviral infections and other viral infections that emerge or emerged from viruses requiring an acidic pH for infectivity. Cell Biochem Funct 2016; 34(4):191–196. doi:10.1002/cbf.3182
- Pascolo S. Time to use a dose of chloroquine as an adjuvant to anti-cancer chemotherapies. Eur J Pharmacol 2016; 771:139–144. doi:10.1016/j.ejphar.2015.12.017
- Furlong HC, Wessels JM, Guerra MT, Stämpfli MR, Foster WG. Hydroxychloroquine attenuates cigarette smoke induced autophagic signaling in the mouse ovary. Reprod Toxicol 2016; 61:105–113. doi:10.1016/j.reprotox.2016.03.044
- Cairoli E, Danese N, Teliz M, et al. Cumulative dose of hydroxychloroquine is associated with a decrease of resting heart rate in patients with systemic lupus erythematosus: a pilot study. Lupus 2015; 24(11):1204–1209. doi:10.1177/0961203315580870
KEY POINTS
- Hydroxychloroquine acts by suppressing Toll-like receptors to trigger important immunomodulatory effects.
- Hydroxychloroquine is a well-established and effective therapy for systemic and cutaneous lupus and other autoimmune diseases.
- Patients with systemic lupus erythematosus treated with hydroxychloroquine have lower mortality rates and a lower risk of lupus nephritis.
- Retinal toxicity is the most serious potential complication of hydroxychloroquine therapy. Adherence to current ophthalmologic screening recommendations and proper dosing protocols lowers this risk.
N-acetylcysteine: A potential treatment for substance use disorders
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.
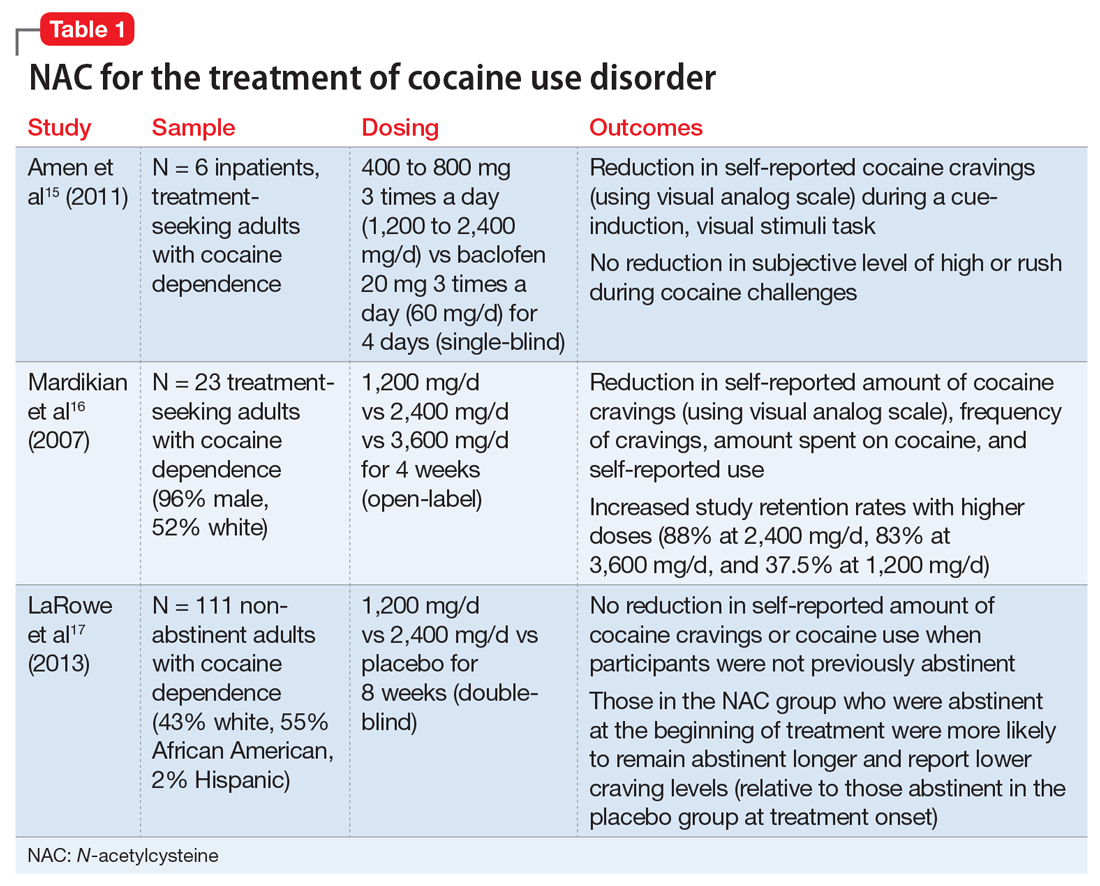
Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).
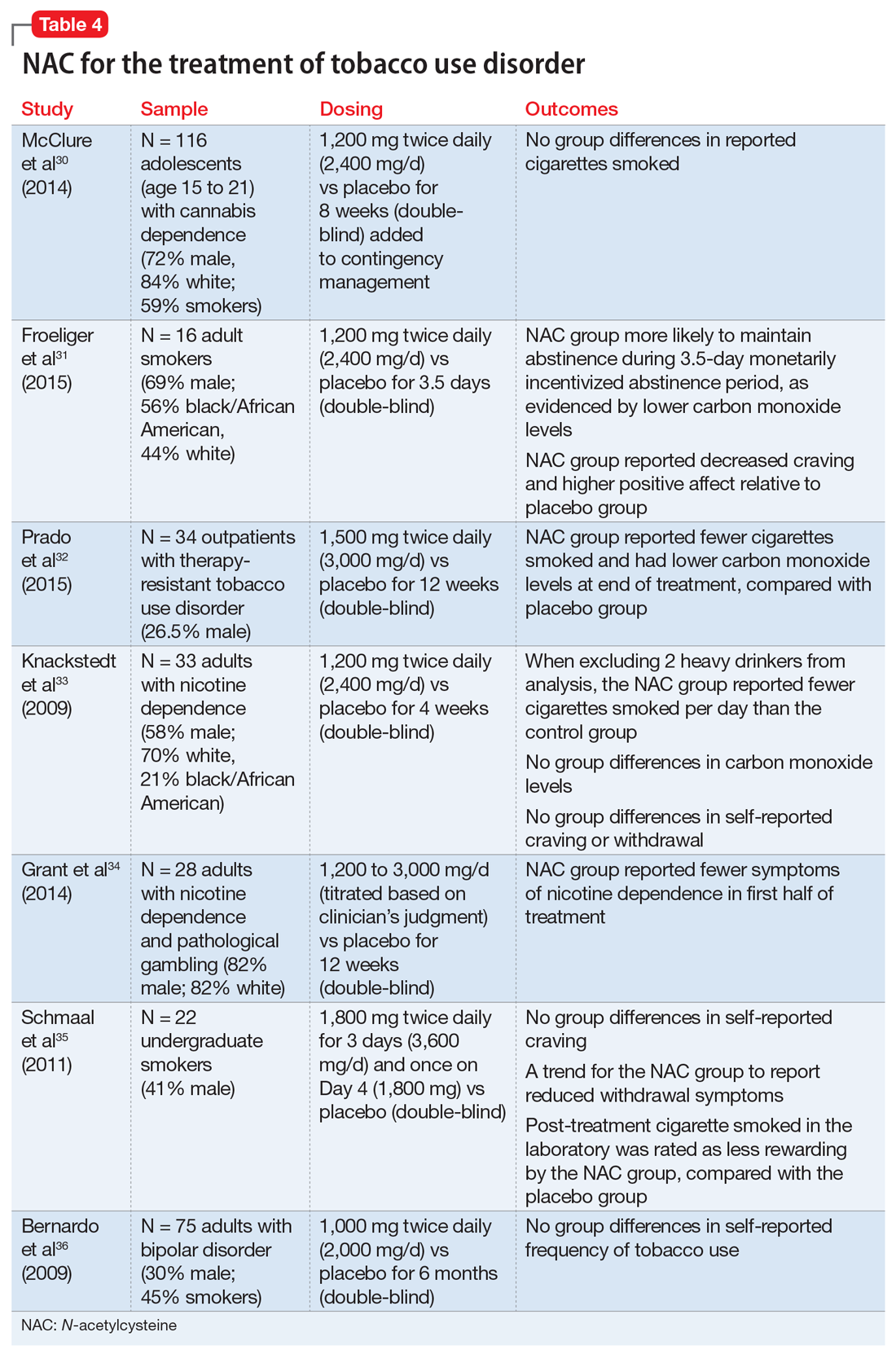
Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.
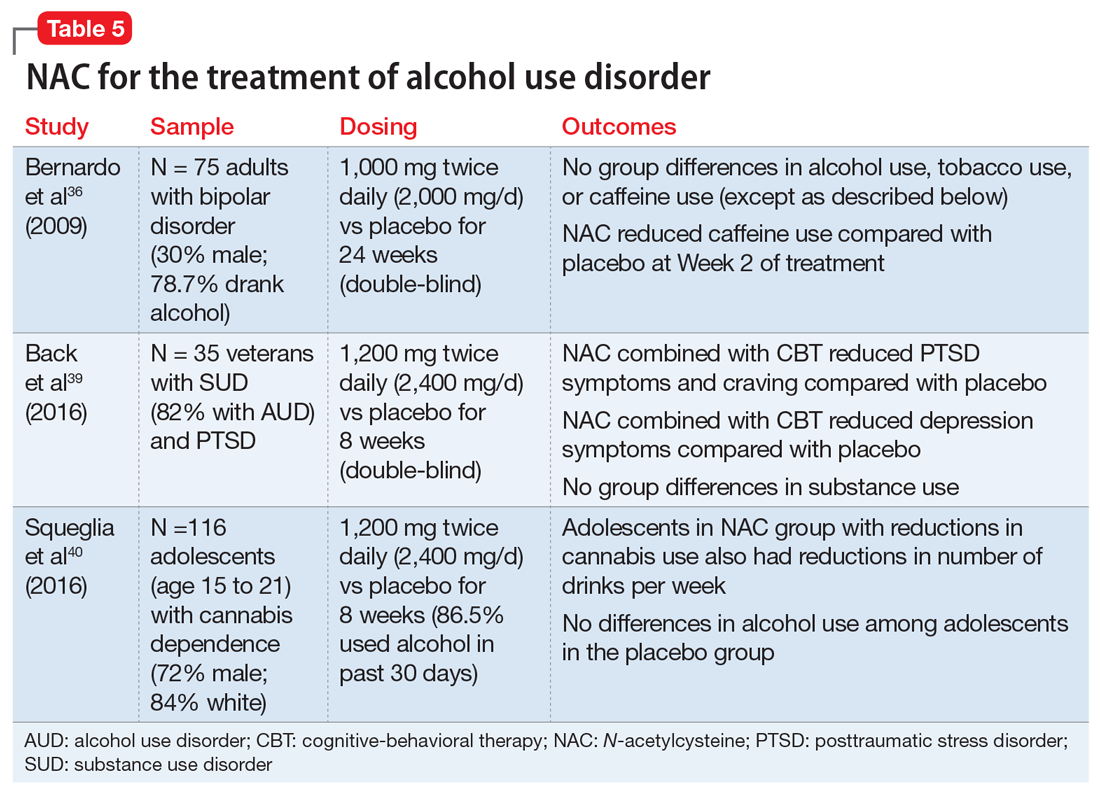
Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.

Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).

Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.

Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
Pharmacologic treatment options for many substance use disorders (SUDs) are limited. This is especially true for cocaine use disorder and cannabis use disorder, for which there are no FDA-approved medications. FDA-approved medications for other SUDs often take the form of replacement or agonist therapies (eg, nicotine replacement therapy) that substitute the effects of the substance to aid in cessation. Other pharmacotherapies treat symptoms of withdrawal, reduce craving, or provide aversive counter-conditioning if the patient consumes the substance while on the medication (eg, disulfiram).
The over-the-counter (OTC) antioxidant N-acetylcysteine (NAC) may be a potential treatment for SUDs. Although NAC is not approved by the FDA for treating SUDs, its proposed mechanism of action differs from that of current FDA-approved medications for SUDs. NAC’s potential for broad applicability, favorable adverse-effect profile, accessibility, and low cost make it an intriguing option for patients with multiple comorbidities, and potentially for individuals with polysubstance use. This article reviews the current evidence supporting NAC for treating SUDs, to provide insight about which patients may benefit from NAC and under which circumstances they are most likely to benefit.
NAC may correct glutamate dysregulation
Approximately 85% of individuals with an SUD do not seek treatment for it, and those who do are older, have a longer history of use, have more severe dependence, and have sought treatment numerous times before.1 By the time most people seek treatment, years of chronic substance use have likely led to significant brain-related adaptations. Individuals with SUDs often indicate that their substance use began as a pleasurable activity—the effects of the drug were enjoyable and they were motivated to use it again. With repeated substance use, they may begin to develop a stronger urge to use the drug, driven not necessarily by a desire for pleasure, but by compulsion.2
Numerous neural adaptations underlie the transition from “liking” a substance to engaging in the compulsive use that is characteristic of an SUD.2 For example, repeated use of an addictive substance may result in excess glutamate in the nucleus accumbens,3,4 an area of the brain that plays a critical role in motivation and learning. As a result, it has been proposed that pharmacotherapies that help correct glutamate dysregulation may be effective in promoting abstinence or preventing relapse to a substance.5,6
NAC may reverse the neural dysfunction seen in SUDs. As an OTC antioxidant that impacts glutamatergic functioning in the brain, NAC has long been used to treat acetaminophen overdose; however, in recent years, researchers have begun to tap its potential for treating substance use and psychiatric disorders. NAC is thought to upregulate the glutamate transporter (GLT-1) that removes excess glutamate from the nucleus accumbens.6 Several published reviews provide more in-depth information about the neurobiology of NAC.6-10
The adverse-effect profile of NAC is relatively benign. Nausea, vomiting, diarrhea, and sleepiness are relatively infrequent and mild.11,12 The bioavailability of NAC is about 4% to 9%, with an approximate half-life of 6.25 hours when orally administered.13 Because NAC is classified as an OTC supplement, the potency and preparation may vary by supplier. To maximize consistency, NAC should be obtained from a supplier that meets United States Pharmacopeia (USP) standards.
NAC for SUDs: Emerging evidence
Several recent reviews have described the efficacy of NAC for SUDs and other psychiatric disorders. Here we summarize the current research examining the efficacy of NAC for stimulant (ie, cocaine and methamphetamine), cannabis, tobacco, and alcohol use disorders.
Continue to: Stimulant use disorders
Stimulant use disorders. The United Nations Office for Drugs and Crime estimates that worldwide, more than 18 million people use cocaine and more than 35 million use amphetamines.14 There are currently no FDA-approved treatments for stimulant use disorders, and clinicians treating patients with cocaine or amphetamine dependence often are at a loss for how best to promote abstinence. Recent studies suggest that NAC may decrease drug-seeking behavior and cravings in adults who seek treatment. The results of studies examining NAC for treating cocaine use and methamphetamine use are summarized in Table 115-17 and Table 2,18,19 respectively.

Cocaine cessation and relapse prevention. Several small pilot projects15,16 found that compared with placebo, various doses of NAC reduced craving (as measured with a visual analog scale). However, in a double-blind, placebo-controlled study, NAC did not decrease cravings or use after 8 weeks of treatment in individuals with cocaine use disorder who were still using cocaine (ie, they had not yet become abstinent). Interestingly, those who were abstinent when treatment began reported lower craving and remained abstinent longer if they received NAC (vs placebo), which suggests that NAC may be useful for preventing relapse.17

Methamphetamine cessation and relapse prevention. One study (N = 32) that evaluated the use of NAC, 1,200 mg/d for 4 weeks, vs placebo found reduced cravings among methamphetamine users who were seeking treatment.18 In contrast, a study of 31 methamphetamine users who were not seeking treatment evaluated the use of NAC, 2,400 mg/d, plus naltrexone, 200 mg/d, vs placebo for 8 weeks.19 It found no significant differences in craving or use patterns. Further research is needed to optimize the use of NAC for stimulant use disorders, and to better understand the role that abstinence plays.
Appropriate populations. The most support for use of NAC has been as an anti-relapse agent in treatment-seeking adults.
Continue to: Safety and dosing
Safety and dosing. Suggested dosages for the treatment of cocaine use disorder range from 1,200 to 3,600 mg/d (typically 600 to 1,800 mg twice daily, due to NAC’s short half-life), with higher retention rates noted in individuals who received 2,400 mg/d and 3,600 mg/d.16
Clinical implications. NAC is thought to act as an anti-relapse agent, rather than an agent that can help someone who is actively using stimulants to stop. Consequently, NAC will likely be most helpful for patients who are motivated to quit and are abstinent when they start taking NAC; however, this hypothesis needs further testing.
Cannabis use disorder
There are no FDA-approved treatments for cannabis use disorder. Individuals who use marijuana or other forms of cannabis may be less likely to report negative consequences or seek treatment compared with those who use other substances. Approximately 9% of individuals who use marijuana develop cannabis use disorder20; those who begin using marijuana earlier in adolescence are at increased risk.21 Commonly reported reasons for wanting to stop using marijuana include being concerned about health consequences, regaining or demonstrating self-control, saving money, avoiding legal consequences, obtaining or keeping employment, and reducing interpersonal conflict.22,23 Table 324-27 summarizes initial evidence that suggests NAC may be particularly useful in reducing marijuana use among adolescents (age 15 to 21).24,25

Cessation. An open-label, pilot clinical trial found significant reductions in self-reported marijuana use and craving—but not in biomarkers of use—among 24 adolescents after 4 weeks of NAC, 1,200 mg twice daily.24 In an 8-week, double-blind, randomized controlled trial of 116 adolescents, NAC, 1,200 mg twice daily, plus contingency management doubled the odds of abstinence, but had no effect on self-reported craving or use.25,26 In a sample of 302 adults, a 12-week trial of NAC, 1,200 mg twice daily, plus contingency management was no more effective than contingency management alone in promoting abstinence.27
Continue to: Appropriate populations
Appropriate populations. Evidence is stronger for use of NAC among adolescents (age 15 to 21) than for individuals older than age 21.25,27 Further research is needed to explore potential reasons for age-specific effects.
Safety and dosing. A safe and potentially efficacious dosage for the treatment of cannabis use disorder is 2,400 mg/d (1,200 mg twice daily).24,25,27
Clinical implications. Combined with contingency management, NAC might be efficacious for adolescents with cannabis use disorder, with treatment gains evident by the fourth week of treatment.24,25 To date, no clinical trials have examined the efficacy of NAC for treating cannabis use disorder without adjunctive contingency management, and research is needed to isolate the clinical effect of NAC among adolescents.
Tobacco use disorder
Cigarette smoking remains a leading cause of preventable death in the United States,28 and nearly 70% of people who start using tobacco become dependent.20 Existing FDA-approved treatments include nicotine replacement products, varenicline, and bupropion. Even though efficacious treatments exist, successful and sustained quit attempts are infrequent.29 NAC may exert a complementary effect to existing tobacco cessation interventions, such as varenicline.30 While these medications promote abstinence, NAC may be particularly beneficial in preventing relapse after abstinence has been achieved (Table 430-36).

Continue to: Cessation and relapse prevention
Cessation and relapse prevention. Several pilot studies found that adult smokers who received NAC (alone or in combination with another treatment) had lower carbon monoxide levels,31,32 smoked fewer cigarettes,32,33 and had fewer self-reported symptoms of nicotine dependence34 and/or less craving for cigarettes.31 However, one study of 33 smokers did not find a reduction in craving or carbon monoxide for NAC compared with placebo.33 Another pilot study of 22 young adult smokers found that those who received NAC rated their first cigarette after treatment (smoked in the laboratory) as less rewarding, relative to smokers who received a placebo.35
Secondary analyses of adults with bipolar disorder36 and adolescents with cannabis use disorder37 found no decreases in tobacco use among those who received NAC compared with placebo. However, the studies in these analyses did not specifically recruit tobacco users, and participants who were tobacco users were not necessarily interested in quitting. This may partially explain discrepant findings.
Appropriate populations. NAC has been studied mostly in adult cigarette smokers.
Safety and dosing. Suggested dosages for treating tobacco use disorder range from 1,200 to 3,600 mg/d (600 to 1,800 mg twice daily).
Continue to: Clinical implications
Clinical implications. Data on NAC’s efficacy for tobacco use disorder come from small, pilot trials. Although initial evidence is promising, it is premature to suggest NAC for smoking cessation until a fully powered, randomized clinical trial provides evidence of efficacy.
Alcohol use disorder
Alcohol use disorders are widely prevalent; 13.9% of U.S. adults met criteria in the past year, and 29.1% of U.S. adults meet criteria in their lifetime.38 Alcohol use disorders can result in significant negative consequences, including relationship problems, violent behavior, medical problems, and death. Existing FDA-approved medications for alcohol use disorder include naltrexone, acamprosate, and disulfiram.
Due to the severe potential health consequences of alcohol, NAC has been examined as a possible aid in preventing relapse. However, most studies have been conducted using animals. Three studies have examined alcohol use in humans (Table 536,39,40). One was a pilot study,39 and the other 2 were secondary data analyses.36,40 None of them specifically focused on alcohol use disorders. A pilot study of 35 veterans with co-occurring posttraumautic stress disorder (PTSD) and SUDs (82% of whom had an alcohol use disorder) found that compared with placebo, NAC significantly decreased PTSD symptoms, craving, and depression.39 In a study of 75 adults with bipolar disorder, secondary alcohol use was not significantly reduced.36 However, one study suggested that NAC may decrease adolescent alcohol and marijuana co-use.40 Future work is needed to examine the potential clinical utility of NAC in individuals with alcohol use disorders.

Findings from animal studies indicate that NAC may:
- reduce alcohol-seeking41
- reduce withdrawal symptoms42
- reduce the teratogenic effects of alcohol43
- prevent alcohol toxicity44
- reduce health-related consequences of alcohol (eg, myocardial oxidative stress45 and alcohol-related steatohepatitis46).
Continue to: Appropriate populations
Appropriate populations. Pilot studies have suggested that appropriate populations may include veterans with SUD and PTSD39 and adolescents with marijuana dependence who use alcohol.40
Safety and dosing. Suggested dosages for the treatment of alcohol use disorder based on these studies range from 1,000 to 2,400 mg/d (500 to 1,200 mg twice daily).
Clinical implications. Future work is needed to determine if NAC is effective for treating alcohol use disorders. Ongoing randomized clinical trials are examining the efficacy of NAC in reducing alcohol use among individuals with alcohol use disorder. It is premature to recommend NAC for treatment of alcohol use disorders.
Other psychiatric uses
Although we have highlighted NAC’s effect on glutamatergic transmission, evidence suggests that NAC may have multiple mechanisms of action that could impact psychiatric functioning. For example, NAC may also reverse oxidative stress, which is frequently observed in psychiatric disorders such as schizophrenia and bipolar disorder.10,12 NAC also has anti-inflammatory properties. When inflammatory pathways of the CNS are dysregulated, production of neurotransmitters may be impaired, resulting in depression-like symptoms.10,12,47 Preliminary evidence suggests that NAC may be effective in treating mood-related symptoms (eg, irritability, depression) in individuals with psychiatric disorders (eg, bipolar and depressive disorders, PTSD, and SUDs) and general symptoms of schizophrenia, obsessive-compulsive disorder, and trichotillomania, although mixed findings in controlled studies suggest a need for further research.12,39
Continue to: NAC: A promising candidate
NAC: A promising candidate
Initial evidence suggests NAC may be helpful for treating patients with SUDs. A patient seeking SUD treatment who is treated with NAC may experience a decreased drive, craving, or compulsion to use. Notably, NAC may be particularly useful in preventing relapse after an individual has achieved abstinence. Evidence suggests that NAC may be useful in the treatment of adults with cocaine use disorders who have achieved abstinence, and adolescents with cannabis use disorders. Preliminary results for adult tobacco use disorder are also promising. Human data examining the efficacy of NAC for alcohol use disorder is limited. Researchers’ ongoing challenge is to identify which patients with which SUDs are most likely to benefit from NAC, and to create clear clinical guidelines for the provider.
Bottom Line
N-acetylcysteine is likely to have modest effects for some patients who have a substance use disorder, particularly adults who use cocaine and adolescents who use marijuana. It may be useful in preventing relapse to substance use after an individual has achieved abstinence.
Related Resources
- Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
- Roberts-Wolfe D, Kalivas P. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
- National Institute on Drug Abuse. Treatment approaches for drug addiction. https://www.drugabuse.gov/publications/drugfacts/treatment-approaches-drug-addiction.
Drug Brand Names
Acamprosate • Campral
Acetaminophen • Tylenol
Baclofen • Lioresal
Bupropion • Zyban
Disulfiram • Antabuse
Naltrexone • Revia,Vivitrol
Varenicline • Chantix
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
1. Grella CE, Karno MP, Warda US, et al. Perceptions of need and help received for substance dependence in a national probability survey. Psychiatr Serv. 2009;60(8):1068-1074.
2. Everitt BJ, Robbins TW. Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol. 2016;67:23-50.
3. McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23(8):3531-3537.
4. LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28(12):3170-3177.
5. Kalivas PW, Volkow ND. New medications for drug addiction hiding in glutamatergic neuroplasticity. Mol Psychiatry. 2011;16(10):974-986.
6. Roberts-Wolfe D, Kalivas PW. Glutamate transporter GLT-1 as a therapeutic target for substance use disorders. CNS Neurol Disord Drug Targets. 2015;14(6):745-756.
7. Berk M, Malhi GS, Gray LJ, et al. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34(3):167-177.
8. McClure EA, Gipson CD, Malcolm RJ, et al. Potential role of N-acetylcysteine in the management of substance use disorders. CNS drugs. 2014;28(2):95-106.
9. Deepmala, Slattery J, Kumar N, et al. Clinical trials of N-acetylcysteine in psychiatry and neurology: a systematic review. Neurosci Biobehav Rev. 2015;55:294-321.
10. Minarini A, Ferrari S, Galletti M, et al. N-acetylcysteine in the treatment of psychiatric disorders: current status and future prospects. Expert Opin Drug Metab Toxicol. 2017;13(3):279-292.
11. Grandjean EM, Berthet P, Ruffman R, et al. Efficacy of oral long-term N‑acetylcysteine in chronic bronchopulmonary disease: a meta-analysis of published double-blind, placebo-controlled clinical trials. Clin Ther. 2000;22(2):209‑221.
12. Rhodes K, Braakhuis A. Performance and side effects of supplementation with N-acetylcysteine: a systematic review and meta-analysis. Sports Med. 2017;47(8):1619-1636.
13. Olsson B, Johansson M, Gabrielsson J, et al. Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol. 1988;34(1):77-82.
14. United Nations Office on Drugs and Crime. World Drug Report 2016 (United Nations publication, Sales No. E.16.XI.7). https://www.unodc.org/doc/wdr2016/WORLD_DRUG_REPORT_2016_web.pdf. Published May 2016. Accessed April 26, 2018.
15. Amen SL, Piacentine LB, Ahmad ME, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871-878.
16. Mardikian PN, LaRowe SD, Hedden S, et al. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389-394.
17. LaRowe SD, Kalivas PW, Nicholas JS, et al. A double‐blind placebo‐controlled trial of N‐acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443-452.
18. Mousavi SG, Sharbafchi MR, Salehi M, et al. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28-33.
19. Grant JE, Odlaug BL, Kim SW. A double-blind, placebo-controlled study of N-acetyl cysteine plus naltrexone for methamphetamine dependence. Eur Neuropsychopharmacol. 2010;20(11):823-828.
20. Lopez-Quintero C, Pérez de los Cobos J, Hasin DS, et al. Probability and predictors of transition from first use to dependence on nicotine, alcohol, cannabis, and cocaine: results of the National Epidemiologic Survey on Alcohol and Related Conditions (NESARC). Drug Alcohol Depend. 2011;115(1-2):120-130.
21. Chen CY, O’Brien MS, Anthony JC. Who becomes cannabis dependent soon after onset of use? Epidemiological evidence from the United States: 2000-2001. Drug Alcohol Depend. 2005;79(1):11-22.
22. Copersino ML, Boyd SJ, Tashkin DP, et al. Quitting among non-treatment-seeking marijuana users: reasons and changes in other substance use. Am J Addict. 2006;15(4):297-302.
23. Weiner MD, Sussman S, McCuller WJ, et al. Factors in marijuana cessation among high-risk youth. J Drug Educ. 1999;29(4):337-357.
24. Gray KM, Watson NL, Carpenter MJ, et al. N-acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187-189.
25. Gray KM, Carpenter MJ, Baker NL, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805-812.
26. Roten AT, Baker NL, Gray KM. Marijuana craving trajectories in an adolescent marijuana cessation pharmacotherapy trial. Addict Behav. 2013;38(3):1788-1791.
27. Gray KM, Sonne SC, McClure EA, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249-257.
28. Rostron B. Mortality risks associated with environmental tobacco smoke exposure in the United States. Nicotine Tob Res. 2013;15(10):1722-1728.
29. Centers for Disease Control and Prevention. Quitting smoking among adults – United States, 2001–2010. MMWR. 2011;60(44):1513-1519.
30. McClure EA, Baker NL, Gipson CD, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52-56.
31. Froeliger B, McConnell P, Stankeviciute N, et al. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234-242.
32. Prado E, Maes M, Piccoli LG, et al. N-acetylcysteine for therapy-resistant tobacco use disorder: a pilot study. Redox Rep. 2015;20(5):215-222.
33. Knackstedt LA, LaRowe S, Mardikian P, et al. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841-845.
34. Grant JE, Odlaug BL, Chamberlain SR, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39-45.
35. Schmaal L, Berk L, Hulstijn KP, et al. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addiction Res. 2011;17(4):211-216.
36. Bernardo M, Dodd S, Gama CS, et al. Effects of N‐acetylcysteine on substance use in bipolar disorder: a randomised placebo‐controlled clinical trial. Acta Neuropsychiatr. 2009;21(5):239-245.
37. McClure EA, Baker NL, Gray KM. Cigarette smoking during an N-acetylcysteine-assisted cannabis cessation trial in adolescents. Am J Drug Alcohol Abuse. 2014;40(4):285-291.
38. Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 alcohol use disorder: Results from the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766.
39. Back SE, McCauley JL, Korte KJ, et al. A double-blind randomized controlled pilot trial of N-acetylcysteine in veterans with PTSD and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439-e1446.
40. Squeglia LM, Baker NL, McClure EA, et al. Alcohol use during a trial of N-acetylcysteine for adolescent marijuana cessation. Addict Behav. 2016;63:172-177.
41. Lebourgeois S, González-Marín MC, Jeanblanc J, et al. Effect of N-acetylcysteine on motivation, seeking and relapse to ethanol self-administration. Addict Biol. 2018;23(2):643-652.
42. Schneider R Jr, Santos CF, Clarimundo V, et al. N-acetylcysteine prevents behavioral and biochemical changes induced by alcohol cessation in rats. Alcohol. 2015;49(3):259-263.
43. Parnell SE, Sulik KK, Dehart DB, et al. Reduction of ethanol-induced ocular abnormalities in mice via dietary administration of N-acetylcysteine. Alcohol. 2010;44(7-8):699-705.
44. Ozkol H, Bulut G, Balahoroglu R, et al. Protective effects of Selenium, N-acetylcysteine and Vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res. 2017;175(1):177-185.
45. Seiva FR, Amauchi JF, Rocha KK, et al. Alcoholism and alcohol abstinence: N-acetylcysteine to improve energy expenditure, myocardial oxidative stress, and energy metabolism in alcoholic heart disease. Alcohol. 2009;43(8):649-656.
46. Setshedi M, Longato L, Petersen DR, et al. Limited therapeutic effect of N‐acetylcysteine on hepatic insulin resistance in an experimental model of alcohol‐induced steatohepatitis. Alcohol Clin Exp Res. 2011;35(12):2139-2151.
47. Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732-741.
CMS floats Medicare direct provider contracting
Under a direct provider contracting (DPC) arrangement, Medicare could pay physicians or physician groups a monthly fee to deliver a specific set of services to beneficiaries, who would gain greater access to the physicians. The physicians would be accountable for those Medicare patients’ costs and care quality.
CMS is looking at how to incorporate this concept into the Medicare ranks. On April 23, CMS issued a request for information (RFI) seeking input across a wide range of topics, including provider/state participation, beneficiary participation, payment, general model design, program integrity and beneficiary protection, and how such models would fit within the existing accountable care organization framework.
The RFI offered one possible vision on how a direct provider contracting model could work.
“Under a primary care–focused DPC model, CMS could enter into arrangements with primary care practices under which CMS would pay these participating practices a fixed per beneficiary per month (PBPM) payment to cover the primary care services the practice would be expected to furnish under the model, which may include office visits, certain office-based procedures, and other non–visit-based services covered under the physician fee schedule, and flexibility in how otherwise billable services are delivered,” the RFI states.
Physicians could also earn performance bonuses, depending on how the DPC is structured, through “performance-based incentives for total cost of care and quality.”
CMS noted it also “could test ways to reduce administrative burden though innovative changes to claims submission processes for services included in the PBPM payment under these models.”
The direct provider contracting idea grew out of a previous RFI issued in 2017 by CMS’s Center for Medicare and Medicaid Innovation to collect ideas on new ways to deliver patient-centered care. The agency released the more than 1,000 comments received from that request on the same day it issued the RFI on direct provider contracting.
In those comments, a number of physician groups offered support for a direct-contracting approach.
For example, the American Academy of Family Physicians wrote that it “sees continued growth and interest in family physicians adopting this practice model in all settings types, including rural and underserved communities.” And the AAFP suggested that the innovation center should work with DPC organizations to learn more about them.
The American College of Physicians reiterated its previous position that it “supports physician and patient choice of practice and delivery models that are accessible, ethical, and viable and that strengthen the patient-physician relationship.” But the ACP raised a number of issues that could impede access to care or result in lower quality care.
The American Medical Association offered support for “testing of models in which physicians have the ability to deliver more or different services to patients who need them and to be paid more for doing so.”
The AMA suggested that some of the models to be tested include allowing patients to contract directly with physicians, with Medicare paying its fee schedule rates and patients paying the difference; allowing patients to receive their care from DPC practices and get reimbursed by Medicare; or allowing “physicians to define a team of providers who will provide all of the treatment needed for an acute condition or management of a chronic condition, and then allowing patients who select the team to receive all of the services related to their condition from the team in return for a single predefined cost-sharing amount.”
Comments on the RFI are due May 25.
Under a direct provider contracting (DPC) arrangement, Medicare could pay physicians or physician groups a monthly fee to deliver a specific set of services to beneficiaries, who would gain greater access to the physicians. The physicians would be accountable for those Medicare patients’ costs and care quality.
CMS is looking at how to incorporate this concept into the Medicare ranks. On April 23, CMS issued a request for information (RFI) seeking input across a wide range of topics, including provider/state participation, beneficiary participation, payment, general model design, program integrity and beneficiary protection, and how such models would fit within the existing accountable care organization framework.
The RFI offered one possible vision on how a direct provider contracting model could work.
“Under a primary care–focused DPC model, CMS could enter into arrangements with primary care practices under which CMS would pay these participating practices a fixed per beneficiary per month (PBPM) payment to cover the primary care services the practice would be expected to furnish under the model, which may include office visits, certain office-based procedures, and other non–visit-based services covered under the physician fee schedule, and flexibility in how otherwise billable services are delivered,” the RFI states.
Physicians could also earn performance bonuses, depending on how the DPC is structured, through “performance-based incentives for total cost of care and quality.”
CMS noted it also “could test ways to reduce administrative burden though innovative changes to claims submission processes for services included in the PBPM payment under these models.”
The direct provider contracting idea grew out of a previous RFI issued in 2017 by CMS’s Center for Medicare and Medicaid Innovation to collect ideas on new ways to deliver patient-centered care. The agency released the more than 1,000 comments received from that request on the same day it issued the RFI on direct provider contracting.
In those comments, a number of physician groups offered support for a direct-contracting approach.
For example, the American Academy of Family Physicians wrote that it “sees continued growth and interest in family physicians adopting this practice model in all settings types, including rural and underserved communities.” And the AAFP suggested that the innovation center should work with DPC organizations to learn more about them.
The American College of Physicians reiterated its previous position that it “supports physician and patient choice of practice and delivery models that are accessible, ethical, and viable and that strengthen the patient-physician relationship.” But the ACP raised a number of issues that could impede access to care or result in lower quality care.
The American Medical Association offered support for “testing of models in which physicians have the ability to deliver more or different services to patients who need them and to be paid more for doing so.”
The AMA suggested that some of the models to be tested include allowing patients to contract directly with physicians, with Medicare paying its fee schedule rates and patients paying the difference; allowing patients to receive their care from DPC practices and get reimbursed by Medicare; or allowing “physicians to define a team of providers who will provide all of the treatment needed for an acute condition or management of a chronic condition, and then allowing patients who select the team to receive all of the services related to their condition from the team in return for a single predefined cost-sharing amount.”
Comments on the RFI are due May 25.
Under a direct provider contracting (DPC) arrangement, Medicare could pay physicians or physician groups a monthly fee to deliver a specific set of services to beneficiaries, who would gain greater access to the physicians. The physicians would be accountable for those Medicare patients’ costs and care quality.
CMS is looking at how to incorporate this concept into the Medicare ranks. On April 23, CMS issued a request for information (RFI) seeking input across a wide range of topics, including provider/state participation, beneficiary participation, payment, general model design, program integrity and beneficiary protection, and how such models would fit within the existing accountable care organization framework.
The RFI offered one possible vision on how a direct provider contracting model could work.
“Under a primary care–focused DPC model, CMS could enter into arrangements with primary care practices under which CMS would pay these participating practices a fixed per beneficiary per month (PBPM) payment to cover the primary care services the practice would be expected to furnish under the model, which may include office visits, certain office-based procedures, and other non–visit-based services covered under the physician fee schedule, and flexibility in how otherwise billable services are delivered,” the RFI states.
Physicians could also earn performance bonuses, depending on how the DPC is structured, through “performance-based incentives for total cost of care and quality.”
CMS noted it also “could test ways to reduce administrative burden though innovative changes to claims submission processes for services included in the PBPM payment under these models.”
The direct provider contracting idea grew out of a previous RFI issued in 2017 by CMS’s Center for Medicare and Medicaid Innovation to collect ideas on new ways to deliver patient-centered care. The agency released the more than 1,000 comments received from that request on the same day it issued the RFI on direct provider contracting.
In those comments, a number of physician groups offered support for a direct-contracting approach.
For example, the American Academy of Family Physicians wrote that it “sees continued growth and interest in family physicians adopting this practice model in all settings types, including rural and underserved communities.” And the AAFP suggested that the innovation center should work with DPC organizations to learn more about them.
The American College of Physicians reiterated its previous position that it “supports physician and patient choice of practice and delivery models that are accessible, ethical, and viable and that strengthen the patient-physician relationship.” But the ACP raised a number of issues that could impede access to care or result in lower quality care.
The American Medical Association offered support for “testing of models in which physicians have the ability to deliver more or different services to patients who need them and to be paid more for doing so.”
The AMA suggested that some of the models to be tested include allowing patients to contract directly with physicians, with Medicare paying its fee schedule rates and patients paying the difference; allowing patients to receive their care from DPC practices and get reimbursed by Medicare; or allowing “physicians to define a team of providers who will provide all of the treatment needed for an acute condition or management of a chronic condition, and then allowing patients who select the team to receive all of the services related to their condition from the team in return for a single predefined cost-sharing amount.”
Comments on the RFI are due May 25.