User login
Type 2 Diabetes in Adolescents: We Must Do Better
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172
What we know—and don’t—about non-nutritive sweeteners
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
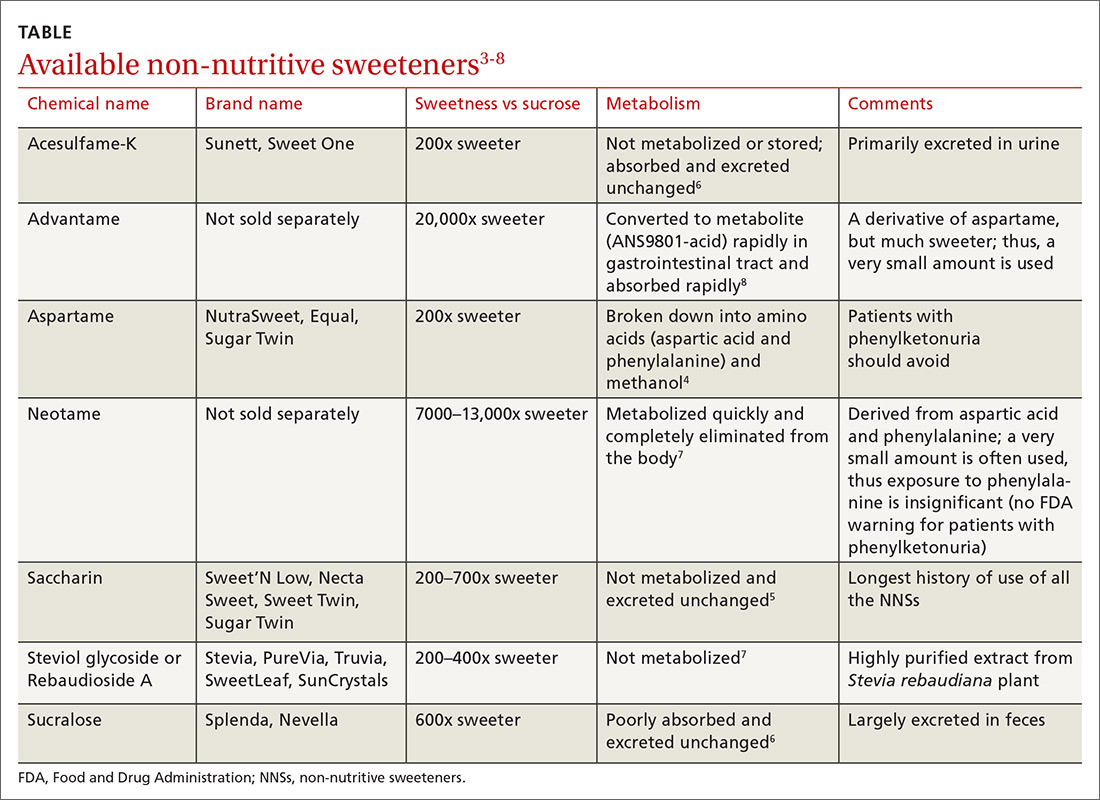
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.
Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; Clipper.young@tu.edu.
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.

Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; Clipper.young@tu.edu.
An estimated 93.3 million Americans (roughly 40% of the US population) were obese in 2015-2016, and most of them had at least 1 chronic disease.1 As a result, patient education focused on lifestyle modification, including healthy nutrition and physical activity, has become an integral part of our everyday practice.
At the same time, the most recent dietary guidelines recommend that added sugar make up < 10% of daily calories.2 In the United States, low-calorie food and beverages containing non-nutritive sweeteners (NNSs; TABLE3-8) have become a popular means of keeping the sweetness in our diet without the health ramifications associated with sugar. These NNSs (aka, artificial sweeteners, high-intensity sweeteners, and non-caloric sweeteners) are ubiquitous in soft drinks, processed grains (including breads, cereals, and granola bars), and dairy products (including yogurts, flavored milk, and ice cream). As examples, NNSs are present in 42% of flavored waters, 33% of yogurts, and all diet beverages.9,10 They can even be found in medications, multivitamins, toothpaste, and mouthwash.

Business is booming
Global NNS consumption has been growing more than 5% per year, meaning that by 2020, NNSs are expected to be a $2.2 billion industry.11 One study using data from the National Health and Nutrition Examination Survey (NHANES) found that the use of NNSs in the United States increased from 21.1% in 2003 to 24.9% in 2009-2010 among adults and increased from 7.8% to 18.9% over the same time period among children.12
The main increase in the consumption of NNSs across all age groups has been via the consumption of beverages. Approximately 11% of healthy weight, 19% of overweight, and 22% of obese adults consume diet beverages.13,14 Consumption of diet beverages or NNSs increases with age12 and is especially common among women with higher levels of education and income.15
However, concerns remain about the safety of these agents and their effect on weight, appetite, and the body’s glycemic response. This article reviews the available research and current recommendations regarding the use of NNSs.
WHAT EFFECT DO NNS s HAVE ON WEIGHT?
The data on NNSs and weight are inconsistent. One randomized controlled trial(RCT) compared weight loss over the course of 1 year (12-week weight loss phase; 9-month weight maintenance phase) when 303 participants consumed either water or drinks sweetened with NNSs.16 Weight loss was significantly greater in the NNS drink group when compared with the water group.16
Observational studies have revealed similar findings.17,18 Data from NHANES revealed that US adults (n = 14,098) during 2 nonconsecutive 24-hour dietary recall periods demonstrated lower total energy (calorie) intake if they consumed NNSs vs no NNSs.19 Another study using 2011-2016 NHANES data on adolescents (n = 7026) found no difference in energy intake between those who consumed beverages containing NNSs vs those who consumed beverages containing sugar.20
Continue to: Other lines of investigation...
Other lines of investigation, including animal studies, have shown that long-term use of NNSs is associated with numerous metabolic derangements including weight gain.21 The negative effects of NNSs appear to be the greatest in males and those who are obese and have high-calorie diets.21
A 2017 meta-analysis concluded that evidence from RCTs does not support a benefit of NNSs on weight management, and that routine consumption of NNSs may be associated with increased body mass index (BMI) and cardiometabolic risk.22 Another systematic review and meta-analysis found that there was a higher pooled risk for obesity among those who drank beverages containing NNSs vs those who drank sugar-containing beverages.23
Based on the most current literature, we conclude that NNSs are not beneficial for weight loss. While there is concern about weight gain through psychological effects (stimulation of sweetness receptors without satiety), further well-designed research is needed to explore whether this concern has merit.
WHAT IS THE EFFECT OF NNSs ON APPETITE?
There appears to be no effect. While original studies seemed to indicate there was an effect, later studies leaned to the contrary.
The notion that NNSs might enhance appetite and food intake was advanced in the 1980s by John Blundell and his research team.24 The hypothesis was that since NNSs uncouple sweet taste and calories, they do not exert the normal post-ingestive inhibitory influence that real sugar does. This, in turn, disrupts appetite control mechanisms.25-27
Continue to: However, subsequent research studies...
However, subsequent research studies found no relationship between the use of NNSs and appetite.28-30 Mattes and colleagues hypothesized that such a difference in findings could result from the fact that earlier studies focused on isolating NNSs from other energy-yielding products, which emphasized an association with heightened hunger.29 Subsequent studies showed that when NNSs were incorporated into energy-yielding products, there was no association between NNSs and increased hunger or appetite.
DO NNSs INCREASE THE RISK FOR TYPE 2 DIABETES MELLITUS?
The data are mixed. One study of women participating in the Nurses’ Health Study II showed that those who consumed caffeinated, artificially-sweetened beverages had a 35% higher risk of developing type 2 diabetes mellitus (T2DM); however, this risk was no longer significant after adjusting for BMI and energy intake.31
The Health Professionals Follow-Up Trial studied more than 40,000 men for more than 20 years and found that NNS consumption increased the risk of developing T2DM by 40%.32 However, this finding lost statistical significance after adjusting for BMI.32
These results make it difficult to determine whether there is any association between NNSs and T2DM; rather NNS-containing beverages are likely consumed more often by those who have higher BMIs and by those trying to lose weight.
A 2017 randomized crossover study involving 10 healthy men looked at the effects of a variety of caloric and non-caloric sweeteners on 24-hour glucose profiles and found no differences.33 Another study, a randomized, double-blind, crossover trial involving 60 non-obese adults without diabetes who did not consume NNSs, randomized the participants one-to-one to drink either 2 cans per day of either a beverage containing aspartame and acesulfame K or an unsweetened, no-calorie beverage for 12 weeks.34
Continue to: After a 4-week washout period...
After a 4-week washout period, the participants then switched to the opposite beverage for 12 weeks. The study concluded that consumption of 2 cans of a beverage containing aspartame and acesulfame K over 12 weeks had no significant effect on insulin sensitivity or secretion in nondiabetic adults.34
Similar results were obtained from a study involving 100 non-obese adults.35 The researchers found that aspartame ingested at 2 different doses (350 or 1050 mg/d) in beverages over 12 weeks had no effect on a 240-minute oral glucose tolerance test, blood pressure, appetite, or body weight.35
A 2016 systematic review critically evaluated the effect of NNSs on both glucose absorption and appetite.36 The review included 14 observational prospective trials, 28 RCTs, and 2 meta-analyses. The sweeteners studied included aspartame, sucralose, saccharin, acesulfame K, and stevia.36 The studies were focused largely on single-exposure outcomes (20 trials), but a minority of the studies (8 trials) looked at longer exposures from 1 to 18 weeks. Only some of the studies controlled for critical variables, such as BMI. In the end, there was no consistent pattern of increased or decreased risk for insulin resistance or diabetes.36
Two meta-analyses tried to determine if an association exists between consumption of beverages containing NNSs and the development of T2DM.37,38 The first meta-analysis with 4 studies showed a slight, but significant, relative risk (RR) of 1.13 (95% confidence interval [CI], 1.02-1.25) for those who consumed beverages containing NNSs.37 In the second meta-analysis (10 studies), NNS consumption had an RR of 1.48 (95% CI, 1.35-1.62), but the risk was lower (and no longer significant) after adjusting for BMI.38 A study of 98 Hispanic adolescents who were overweight or obese found that chronic users (n = 9) of NNSs had higher HbA1c levels 1 year later than did controls (n = 75) and people who initiated use of NNSs between the baseline and 1-year visit (n = 14).39
The American Diabetes Association (ADA) and American Heart Association joint position statement on NNSs, first published in 2012, says that NNSs can be utilized to reduce caloric and carbohydrate consumption for overall diabetes control and to obtain a healthy body weight.40 These principles were reaffirmed in the ADA Standards of Care in 2019.41
Continue to: The 2015 US Scientific Reports on Dietary Guidelines...
The 2015 US Scientific Reports on Dietary Guidelines provided a consensus statement saying, “Future experimental studies should examine the relationship between artificially sweetened soft drinks and biomarkers of insulin resistance and other diabetes markers.”42
DO NNSs HAVE ANY ADVERSE HEALTH EFFECTS?
Maybe. Many individuals avoid NNSs due to fear of developing cancer. While rat studies have previously shown a dose-dependent increased risk of developing cancer, epidemiologic studies in humans have not confirmed an association.43 The National Cancer Institute reports that carcinogenicity studies of NNSs have not shown an association with cancer in humans.44
A prospective study—the Nurses’ Health Study, which followed over 88,000 women for 24 years—found that consumption of > 2 diet sodas per day was associated with an increased risk for coronary heart disease (CHD) and chronic kidney disease (CKD) compared with consumption of < 1 diet soda per month.45 However, other prospective studies have shown that these specific negative health effects may not be present when controlling for weight.45,46
While the prospective studies found some associations between medical conditions (eg, CHD and CKD) and NNS consumption, the literature is limited to intake from beverages and does not include NNS-containing foods. More studies are needed to determine the relationship between NNSs and potential adverse health events, since the current literature is observational and cannot predict causation.
A 2019 study explored the associations between long-term consumption of sugar-sweetened beverages and artificially sweetened beverages (ASBs) and the risk of mortality in the United States.47 This study included 37,716 men from the Health Professionals Follow-up Study and 80,647 women from the Nurses’ Health Study. Subjects who had the highest consumption of ASBs had higher risks for total and cardiovascular disease mortality.47 Cohort-specific analyses showed that an association between ASB consumption and mortality was observed in the participants from the Nurses’ Health Study but not in those from the Health Professionals Follow-up Study, warranting further investigation.47 Cancer mortality and ASB consumption were not shown to have an association in this study.
Continue to: WHY ARE THE DATA INCONCLUSIVE?
WHY ARE THE DATA INCONCLUSIVE?
Nutritional studies are hard to complete accurately outside of the laboratory setting. Also, the science of NNSs is new and evolving.
With regard to obesity and NNSs, it is possible that findings have been due to reverse causation. People who are overweight or obese are more likely to consume low-calorie foods and beverages; they are also at greater risk for developing diseases, such as T2DM.48,49
HOW SAFE ARE NNSs?
They appear to be safe, but more data are needed. Each of the 7 FDA-approved NNSs has passed extensive laboratory, animal, and human testing, and appears to cause no harm in the human body when consumed.49 But clearly the data are incomplete. As we continue to gain a greater understanding of the metabolism of NNSs, we may need to revisit the issue of safety.
ARE THERE ANY NNSs THAT SOME PEOPLE SHOULD AVOID?
Yes. People with phenylketonuria, who have difficulty metabolizing phenylalanine (a component of aspartame), should avoid consumption of aspartame.50
In addition, NNSs have been found to be present in breast milk.51 While the significance of this finding is yet to be determined, we warn against the use of NNSs by women who are breastfeeding.51
WHAT EFFECT—IF ANY—DO NNSs HAVE ON GUT MICROBIOTA?
We don’t know. Disruptions in the gut microbiome have been linked to numerous metabolic abnormalities, including obesity, insulin resistance, and diabetes, as well as cardiovascular disorders.52,53 Diet is a main determinant of balance in the gut microbiota.54 The gut microbiota are centrally involved in energy harvest, and studies have suggested that low gut bacterial diversity is associated with increased adiposity, insulin resistance, and low-grade inflammation.55-60 Whether NNSs have a relationship with abnormal changes in gut microbiota requires further study.
CORRESPONDENCE
Clipper F. Young, PharmD, MPH, CDE, BC-ADM, BCGP, Touro University California, College of Osteopathic Medicine, 1310 Club Drive, Vallejo, CA 94592; Clipper.young@tu.edu.
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
1. Adult obesity facts. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/obesity/data/adult.html. Reviewed August 13, 2018. Accessed July 15, 2019.
2. Dietary guidelines for Americans 2015-2020: answers to your questions. USDA ChooseMyPlate.gov Web site. https://www.choosemyplate.gov/2015-2020-dietary-guidelines-answers-your-questions. Accessed July 15, 2019.
3. Additional information about high-intensity sweeteners permitted for use in food in the United States. US Food and Drug Administration Web site. https://www.fda.gov/food/food-additives-petitions/additional-information-about-high-intensity-sweeteners-permitted-use-food-united-states. Published February 8, 2018. Accessed July 15, 2019.
4. Magnuson B, for the Aspartame Expert Work Group. Nutritive and non-nutritive sweeteners. NNNS: aspartame, methanol and formaldehyde relationships (2011). https://www.foodsweeteners.com/wp-content/uploads/2015/08/Aspartame-Methanol-and-Formaldehyde-Relationships.pdf. Accessed July 15, 2019.
5. Jo JH, Kim S, Jeon TW, et al. Investigation of the regulatory effects of saccharin on cytochrome P450s in male ICR mice. Toxicol Res. 2017;33:25-30.
6. Shwide-Slavin C, Swift C, Ross T. Nonnutritive sweetener: where are we today? Diabetes Spectrum. 2012;25:104-110.
7. Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners – a review. J Food Sci Technol. 2014;51:611-621.
8. EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the safety of advantame for the proposed uses as a food additive. EFSA Journal. 2013;11:3301.
9. Fitch C, Keim KS; Academy of Nutrition and Dietetics. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
10. Ng SW, Slining MM, Popkin BM. Use of caloric and non-caloric sweeteners in US consumer packaged foods, 2005-2009. J Acad Nutr Diet. 2012;112:1828-1834.
11. Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164(Pt B):446-450.
12. Piernas C, Ng SW, Popkin B. Trends in purchases and intake of foods and beverages containing caloric and low-calorie sweeteners over the last decade in the United States. Pediatr Obes. 2013;8:294-306.
13. Sylvetsky AC, Welsh JA, Brown RJ, et al. Low-calorie sweetener consumption is increasing in the United States. Am J Clin Nutr. 2012;96:640-646.
14. Bleich SN, Wolfson JA, Vine S, et al. Diet-beverage consumption and caloric intake among US adults, overall and by body weight. Am J Public Health. 2014;104:e72-e78.
15. Drewnowski A, Rehm CD. Socio-demographic correlates and trends in low-calorie sweetener use among adults in the United States from 1999 to 2008. Eur J Clin Nutr. 2015;69:1035-1041.
16. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity 2014;22:1415-1421.
17. Peters JC, Wyatt HR, Foster GD, et al. The effects of water and non-nutritive sweetened beverages on weight loss during a 12-week weight loss treatment program. Obesity (Silver Spring). 2014;22:1415-1421.
18. Bellisle F, Drewnowski A. Intense sweeteners, energy intake and the control of body weight. Eur J Clin Nutr. 2007;61:691-700.
19. Malek AM, Hunt KJ, DellaValle DM, et al. Reported consumption of low-calorie sweetener in foods, beverages, and food and beverage additions by US adults: NHANES 2007-2012. Curr Dev Nutr. 2018;2:nzy054.
20. Sylvetsky AC, Figueroa J, Zimmerman T, et al. Consumption of low-calorie sweetened beverages is associated with higher total energy and sugar intake among children, NHANES 2011-2016. Pediatr Obes. 2019;2:e12535.
21. Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav. 2016;164(Pt B):517-523.
22. Azad MB, Abou-Setta AM, Chauhan BF, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. CMAJ. 2017;189: E929-E939.
23. Ruanpeng D, Thongprayoon C, Cheungpasitporn W, et al. Sugar and artificially-sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM. 2017;110:513-520.
24. Blundell JE, Rogers PJ, Hill AJ. Uncoupling sweetness and calories: methodological aspects of laboratory studies on appetite control. Appetite. 1988;11(Suppl 1):54-61.
25. Bellisle F. Intense sweeteners, appetite for the sweet taste, and relationship to weight management. Curr Obes Rep. 2015;4:106-110.
26. Bryant CE, Wasse LK, Astbury N, et al. Non-nutritive sweeteners: no class effect on the glycaemic or appetite responses to ingested glucose. Eur J Clin Nutr. 2014;68:629-631.
27. Canty DJ, Chan MM. Effects of consumption of caloric vs noncaloric sweet drinks on indices of hunger and food consumption in normal adults. Am J Clin Nutr. 1991;53:1159-1164.
28. Meyer-Gerspach AC, Wolnerhanssen B, Beglinger C. Functional roles of low calorie sweeteners on gut function. Physiol Behav. 2016;164(Pt B):479-481.
29. Mattes RD, Popkin BM. Nonnutritive sweetener consumption in humans: effects on appetite and food intake and their putative mechanisms. Am J Clin Nutr. 2009;89:1-14.
30. Bhupathiraju SN, Pan A, Malik VS, et al. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am J Clin Nutr. 2013;97:155-166.
31. Schulze MB, Manson JE, Ludwig DS, et al. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. JAMA. 2004;292:927-934.
32. de Koning L, Malik VS, Rimm EB, et al. Sugar-sweetened and artificially sweetened beverage consumption and the risk of type 2 diabetes in men. Am J Clin Nutr. 2011;93:1321-1327.
33. Tey SL, Salleh NB, Henry CJ, et al. Effect of non-nutritive (artificial vs natural) sweeteners on 24-hour glucose profile. Eur J Clin Nutr. 2017;71:1129-1132.
34. Bonnet F, Tavenard A, Esvan M, et al. Consumption of a carbonated beverage with high-intensity sweeteners has no effect on insulin sensitivity and secretion in nondiabetic adults. J Nutr. 2018;148:1293-1299.
35. Higgins KA, Considine RV, Mattes RD. Aspartame consumption for 12 weeks does not affect glycemia, appetite, or body weight of healthy, lean adults in a randomized controlled trial. J Nutr. 2018;148:650-657.
36. Romo-Romo A, Aguilar-Salinas CA, Brito-Cordova GX, et al. Effects of the non-nutritive sweeteners on glucose metabolism and appetite regulating hormones: systematic review of observational prospective studies and clinical trials. PloS One. 2016;11:e0161264.
37. Greenwood DC, Threspleton DE, Evans CE, et al. Association between sugar-sweetened and artificially sweetened soft drinks and type 2 diabetes: systematic review and dose-response meta-analysis of prospective studies. Br J Nutr. 2014;112:725-734.
38. Imamura F, O’Conner L, Ye M, et al. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ. 2015;351:h3576.
39. Davis JN, Asigbee FM, Markowitz AK, et al. Consumption of artificial sweetened beverages associated with adiposity and increasing HbA1c in Hispanic youth. Clin Obes. 2018;8:236-243.
40. Gardner C, Wylie-Rosett J, Gidding SS, et al. Nonnutritive sweeteners: current use and health perspectives. a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798-1808.
41. American Diabetes Association. Standards of Medical Care in Diabetes—2019. Diabetes Care. 2019;42(Suppl 1):S1-S183.
42. Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Health and Human Services and the Secretary of Agriculture. Office of Disease Prevention and Health Promotion Web site. https://health.gov/dietaryguidelines/2015-scientific-report/.Published February 2015. Accessed July 15, 2019.
43. Aune D. Soft drinks, aspartame, and the risk of cancer and cardiovascular disease. Am J Clin Nutr. 2012;96:1249-1251.
44. Artificial sweeteners and cancer. National Cancer Institute Web site. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/artificial-sweeteners-fact-sheet. Reviewed August 10, 2016. Accessed July 15, 2019.
45. Fung TT, Malik V, Rexrode KM, et al. Sweetened beverage consumption and risk of coronary heart disease in women. Am J Clin Nutr. 2009;89:1037-1042.
46. Lin J, Curhan GC. Associations of sugar and artificially sweetened soda with albuminuria and kidney function decline in women. Clin J Am Soc Nephrol. 2011;6:160-166.
47. Malik VS, Li Y, Pan A, et al. Long-term consumption of sugar-sweetened and artificially sweetened beverages and risk of mortality in US adults. Circulation. 2019;139:2113-2125.
48. Gardener H, Rundek T, Markert M, et al. Diet soft drink consumption is associated with an increased risk of vascular events in the Northern Manhattan Study. J Gen Inten Med. 2012;27:1120-1126.
49. Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739-758.
50. US Food and Drug Administration. Additional information about high-intensity sweeteners permitted for use in food in the United States. https://www.fda.gov/food/ingredientspackaginglabeling/foodadditivesingredients/ucm397725.htm#Aspartame. Accessed May 26, 2019.
51. Sylvetsky AC, Gardner AL, Bauman V, et al. Nonnutritive sweeteners in breast milk. J Toxicol Environ Health. 2015;78:1029-1032.
52. Rajani C, Jia W. Disruptions in gut microbial-host co-metabolism and the development of metabolic disorders. Clin Sci (Lond). 2018;132:791-811.
53. Kho ZY, Lal SK. The human gut microbiome—a potential controller of wellness and disease. Front Microbiol. 2018;9:1835.
54. Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance. Physiol Behav. 2016;164(Pt B):488-493.
55. Turnbaugh PJ, Hamady M, Yatsunenko T, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457:480-484.
56. Cotillard A, Kennedy SP, Kong LC, et al. Dietary intervention impact on gut microbial gene richness. Nature. 2013;500:585-588.
57. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546.
58. Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, et al. Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome P-450 in male rats. J Toxicol Environ Health A. 2008;71:1415-1429.
59. Anderson RL. Effect of saccharin ingestion on stool composition in relation to caecal enlargement and increased stool hydration. Food Chem Toxicol. 1983;21:255-257.
60. Suez J, Korem T, Zeevi D, et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186.
PRACTICE RECOMMENDATIONS
› Advise patients who are trying to lose weight that non-nutritive sweeteners (NNSs) are not beneficial for weight loss. A
› Reassure patients that NNSs do not appear to cause, or increase the risk of, developing type 2 diabetes mellitus. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Navigating travel with diabetes
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
Travel, once reserved for wealthy vacationers and high-level executives, has become a regular experience for many people. The US Travel and Tourism Overview reported that US domestic travel climbed to more than 2.25 billion person-trips in 2017.1 The US Centers for Disease Control and Prevention (CDC) and the US Travel Association suggest that, based on this frequency and the known rate of diabetes, 17 million people with diabetes travel annually for leisure and 5.6 million for business, and these numbers are expected to increase.2
It stands to reason that as the number of people who travel continues to increase, so too will the number of patients with diabetes seeking medical travel advice. Despite resources available to travelers with diabetes, researchers at the 2016 meeting of the American Diabetes Association noted that only 30% of patients with diabetes who responded to a survey reported being satisfied with the resources available to help them manage their diabetes while traveling.2 This article discusses how clinicians can help patients manage their diabetes while traveling, address common travel questions, and prepare patients for emergencies that may arise while traveling.
PRE-TRIP PREPARATION
Provider visit before travel: Checking the bases
Advise patients to schedule an appointment 4 to 6 weeks before their trip.3 At this appointment, give the patient a healthcare provider travel letter (Figure 1) and prescriptions that the patient can hand-carry en route.3 The provider letter should state that the patient has diabetes and should list all supplies the patient needs. The letter should also include specific medications used by the patient and the devices that deliver these medications, eg, Humalog insulin and U-100 syringes4 to administer insulin, as well as any food and medication allergies.
Prescriptions should be written for patients to use in the event of an emergency during travel. Prescriptions for diabetes medications should be written with generic names to minimize confusion for those traveling internationally. Additionally, all prescriptions should provide enough medication to last throughout the trip.4
Advise patients that rules for filling prescriptions may vary between states and countries.3 Also, the strength of insulin may vary between the United States and other countries. Patients should understand that if they fill their insulin prescription in a foreign country, they may need to purchase new syringes to match the insulin dose. For example, if patients use U-100 syringes and purchase U-40 insulin, they will need to buy U-40 syringes or risk taking too little of a dose.
Remind patients that prescriptions are not necessary for all diabetes supplies but are essential for coverage by insurance companies. Blood glucose testing supplies, ketone strips, and glucose tablets may be purchased in a pharmacy without a prescription. Human insulin may also be purchased over the counter. However, oral medications, glucagon, and analog insulins require a prescription. We suggest that patients who travel have their prescriptions on file at a chain pharmacy rather than an independent one. If they are in the United States, they can go to any branch of the chain pharmacy and easily fill a prescription.
Work with the patient to compile a separate document that details the medication dosing, correction-scale instructions, carbohydrate-to-insulin ratios, and pump settings (basal rates, insulin sensitivity, active insulin time).4 Patients who use an insulin pump should record all pump settings in the event that they need to convert to insulin injections during travel.4 We suggest that all patients with an insulin pump have an alternate insulin method (eg, pens, vials) and that they carry this with them along with basal insulin in case the pump fails. This level of preparation empowers the patient to assume responsibility for his or her own care if a healthcare provider is not available during travel.
Like all travelers, patients with diabetes should confirm that their immunizations are up to date. Encourage patients to the CDC’s page (wwwnc.cdc.gov/travel) to check the list of vaccines necessary for their region of travel.4,5 Many special immunizations can be acquired only from a public health department and not from a clinician’s office.
Additionally, depending on the region of travel, prescribing antibiotics or antidiarrheal medications may be necessary to ensure patient safety and comfort. We also recommend that patients with type 1 diabetes obtain a supply of antibiotics and antidiarrheals because they can become sick quickly.
Packing with diabetes: Double is better
The American Diabetes Association recommends that patients pack at least twice the medication and blood-testing supplies they anticipate needing.3 Reinforce to patients the need to pack all medications and supplies in their carry-on bag and to keep this bag in their possession at all times to avoid damage, loss, and extreme changes in temperature and air pressure, which can adversely affect the activity and stability of insulin.
Ask patients about the activities they plan to participate in and how many days they will be traveling, and then recommend shoes that will encourage appropriate foot care.4 Patients with diabetes should choose comfort over style when selecting footwear. All new shoes should be purchased and “broken in” 2 to 3 weeks before the trip. Alternating shoes decreases the risk of blisters and calluses.4
Emergency abroad: Planning to be prepared
It is crucial to counsel patients on how to respond in an emergency.
Encourage patients with diabetes, especially those who use insulin, to obtain a medical identification bracelet, necklace, or in some cases, a tattoo, that states they use insulin and discloses any allergies.3 This ensures that emergency medical personnel will be aware of the patient’s condition when providing care. Also suggest that your patients have emergency contact information available on their person and their cell phone to expedite assistance in an emergency (Table 2).
Urge patients to determine prior to their departure if their health coverage will change once they leave the state or the country. Some insurance companies require patients to go to a specific healthcare system while others regulate the amount of time a patient can be in the hospital before being transferred home. It is important for patients to be aware of these terms in the event of hospitalization.4 Travel insurance should be considered for international travel.
AIRPORT SECURITY: WHAT TO EXPECT WITH DIABETES
The American Diabetes Association works with the US Transportation Security Administration (TSA) to ensure that passengers with diabetes have access to supplies. Travelers with diabetes are allowed to apply for an optional disability notification card, which discreetly informs officers that the passenger has a condition or device that may affect screening procedures.6
The TSA suggests that, before going through airport screening, patients with diabetes separate their diabetes supplies from their luggage and declare all items.6 Including prescription labels for medications and medical devices helps speed up the security process. Advise patients to carry glucose tablets and other solid foods for treating hypoglycemia when passing through airport security checkpoints.7
Since 2016, the TSA has allowed all diabetes-related supplies, medications, and equipment, including liquids and devices, through security after they have been screened by the x-ray scanner or by hand.7 People with diabetes are allowed to carry insulin and other liquid medications in amounts greater than 3.4 ounces (100 mLs) through airport security checkpoints.
Insulin can pass safely through x-ray scanners, but if patients are concerned, they may request that their insulin be inspected by hand.7 Patients must inform airport security of this decision before the screening process begins. A hand inspection may include swabbing for explosives.
Patients with an insulin pump and a continuous glucose monitoring device may feel uncomfortable during x-ray screening and special security screenings. Remind patients that it is TSA policy that patients do not need to disconnect their devices and can request screening by pat-down rather than x-ray scanner.6 It is the responsibility of the patient to research whether the pump can pass through x-ray scanners.
All patients have the right to request a pat-down and can opt out of passing through the x-ray scanner.6 However, patients need to inform officers about a pump before screening and must understand that the pump may be subject to further inspection. Usually, this additional inspection includes swabbing the patient’s hands to check for explosive material and a simple pat-down of the insulin pump.7
IN-FLIGHT TIPS
Time zones and insulin dosing
Diabetes management is often based on a 24-hour medication schedule. Travel can disrupt this schedule, making it challenging for patients to determine the appropriate medication adjustments. With some assistance, the patient can determine the best course of action based on the direction of travel and the number of time zones crossed.
According to Chandran and Edelman,7 medication adjustments are needed only when the patient is traveling east or west, not north or south. As time zones change, day length changes and, consequently, so does the 24-hour regimen many patients follow. As a general rule, traveling east results in a shortened day, requiring a potential reduction in insulin, while traveling west results in a longer day, possibly requiring an increase in insulin dose.7 However, this is a guideline and may not be applicable to all patients.7
Advise patients to follow local time to administer medications beginning the morning after arrival.7 It is not uncommon, due to changes in meal schedules and dosing, for patients to experience hyperglycemia during travel. They should be prepared to correct this if necessary.
Patients using insulin injections should plan to adjust to the new time zone as soon as possible. If the time change is only 1 or 2 hours, they should take their medications before departure according to their normal home time.7 Upon arrival, they should resume their insulin regimen based on the local time.
Westward travel. If the patient is traveling west with a time change of 3 or more hours, additional changes may be necessary. Advise patients to take their insulin according to their normal home time before departure. The change in dosing and schedule will depend largely on current glucose control, time of travel, and availability of food and glucose during travel. Encourage patients to discuss these matters with you in advance of any long travel.
Eastward travel. When the patient is traveling east with a time change greater than 3 hours, the day will be consequently shortened. On the day of travel, patients should take their morning dose according to home time. If they are concerned about hypoglycemia, suggest that they decrease the dose by 10%.6 On arrival, they should adhere to the new time zone and base insulin dosing on local time.
Advice for insulin pump users. Patients with an insulin pump need make only minimal changes to their dosing schedule. They should continue their routine of basal and bolus doses and change the time on their insulin pump to local time when they arrive. Insulin pump users should bring insulin and syringes as backup; in the event of pump malfunction, the patient should continue to use the same amount of bolus insulin to correct glucose readings and to cover meals.7 As for the basal dose, patients can administer a once-daily injection of long-acting insulin, which can be calculated from their pump or accessed from the list they created as part of their pre-travel preparation.7
Advice for patients on oral diabetes medications
If a patient is taking an oral medication, it is less crucial to adhere to a time schedule. In fact, in some cases it may be preferable to skip a dose and risk slight hyperglycemia for a few hours rather than take medication too close in time and risk hypoglycemia.7
Remind patients to anticipate a change in their oral medication regimen if they travel farther than 5 time zones.7 Encourage patients to research time changes and discuss the necessary changes in medication dosage on the day of travel as well as the specific aspects of their trip. A time-zone converter can be found at www.timeanddate.com.8
WHAT TO EXPECT WHILE ON LAND
Insulin 101
Storing insulin at the appropriate temperature may be a concern. Insulin should be kept between 40°F and 86°F (4°C–30°C).4 Remind patients to carry their insulin with them at all times and to not store it in a car glove compartment or backpack where it can be exposed to excessive sun. The Frio cold pack (ReadyCare, Walnut Creek, CA) is a helpful alternative to refrigeration and can be used to cool insulin when hiking or participating in activities where insulin can overheat. These cooling gel packs are activated when exposed to cold water for 5 to 7 minutes5 and are reusable.
Alert patients that insulin names and concentrations may vary among countries. Most insulins are U-100 concentration, which means that for every 1 mL of liquid there are 100 units of insulin. This is the standard insulin concentration used in the United States. There are U-200, U-300, and U-500 insulins as well. In Europe, the standard concentration is U-40 insulin. Syringe sizes are designed to accommodate either U-100 or U-40 insulin. Review these differences with patients and explain the consequences of mixing insulin concentration with syringes of different sizes. Figure 2 shows how to calculate equivalent doses.
Resort tips: Food, drinks, and excursions
A large component of travel is indulging in local cuisine. Patients with diabetes need to be aware of how different foods can affect their diabetes control. Encourage them to research the foods common to the local cuisine. Websites such as Calorie King, MyFitnessPal, Lose it!, and Nutrition Data can help identify the caloric and nutritional makeup of foods.9
Advise patients to actively monitor how their blood glucose is affected by new foods by checking blood glucose levels before and after each meal.9 Opting for vegetables and protein sources minimizes glucose fluctuations. Remind patients that drinks at resorts may contain more sugar than advertised. Patients should continue to manage their blood glucose by checking levels and by making appropriate insulin adjustments based on the readings. We often advise patients to pack a jar of peanut butter when traveling to ensure a ready source of protein.
Patients who plan to participate in physically challenging activities while travelling should inform all relevant members of the activity staff of their condition. In case of an emergency, hotel staff and guides will be better equipped to help with situations such as hypoglycemia. As noted above, patients should always carry snacks and supplies to treat hypoglycemia in case no alternative food options are available during an excursion. Also, warn patients to avoid walking barefoot. Water shoes are a good alternative to protect feet from cuts and sores.
Patients should inquire about the safety of high-elevation activities. With many glucose meters, every 1,000 feet of elevation results in a 1% to 2% underestimation of blood glucose,10 which could result in an inaccurate reading. If high-altitude activities are planned, advise patients to bring multiple meters to cross-check glucose readings in cases where inaccuracies (due to elevation) are possible.
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
- US Travel Association. US travel and tourism overview. www.ustravel.org/system/files/media_root/document/Research_Fact-Sheet_US-Travel-and-Tourism-Overview.pdf. Accessed June 14, 2018.
- Brunk D. Long haul travel turbulent for many with type 1 diabetes. Clinical Endocrinology News 2016. www.mdedge.com/clinicalendocrinologynews/article/109866/diabetes/long-haul-travel-turbulent-many-type-1-diabetes. Accessed June 14, 2018.
- American Diabetes Association. When you travel. www.diabetes.org/living-with-diabetes/treatment-and-care/when-you-travel.html?utm_source=DSH_BLOG&utm_medium=BlogPost&utm_content=051514-travel&utm_campaign=CON. Accessed June 14, 2018.
- Kruger DF. The Diabetes Travel Guide. How to travel with diabetes-anywhere in the world. Arlington, VA: American Diabetes Association; 2000.
- Centers for Disease Control and Prevention. Travelers’ health. wwwnc.cdc.gov/travel/. Accessed June 14, 2018.
- American Diabetes Association. What special concerns may arise? www.diabetes.org/living-with-diabetes/know-your-rights/discrimination/public-accommodations/air-travel-and-diabetes/what-special-concerns-may.html. Accessed June 14, 2018.
- Chandran M, Edelman SV. Have insulin, will fly: diabetes management during air travel and time zone adjustment strategies. Clinical Diabetes 2003; 21(2):82–85. doi:10.2337/diaclin.21.2.82
- Time and Date AS. Time zone converter. timeanddate.com. Accessed March 19, 2018.
- Joslin Diabetes Center. Diabetes and travel—10 tips for a safe trip. www.joslin.org/info/diabetes_and_travel_10_tips_for_a_safe_trip.html. Accessed June 14, 2018.
- Jendle J, Adolfsson P. Impact of high altitudes on glucose control. J Diabetes Sci Technol 2011; 5(6):1621–1622. doi:10.1177/193229681100500642
KEY POINTS
- Patients should pack all diabetes medications and supplies in a carry-on bag and keep it in their possession at all times.
- A travel letter will facilitate easy transfer through security and customs.
- Patients should always take more supplies than needed to accommodate changes in travel plans.
- If patients will cross multiple time zones during their travel, they will likely need to adjust their medication and food schedules.
Insulin for type 2 diabetes: How and when to get started
› Initiate insulin for patients whose hemoglobin A1c ≥8% despite taking 2 or more oral agents. C
› Prescribe insulin for patients who have not reached their goal one year after diagnosis and initiation of oral therapy. C
› Consider reducing—but do not discontinue—oral agents, such as sulfonylureas and meglitinides, when you initiate insulin therapy. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
With type 2 diabetes now affecting 8.3% of the US population, most primary care physicians see patients with this disorder every day.1 Based on the concurrent obesity epidemic, aging population, and emergence of type 2 diabetes in children and adolescents, it is estimated that by 2050, the prevalence will have risen from one in 12 Americans to one in 3.1
Type 2 diabetes is a progressive disorder, with a relentless decline in beta cells. By the time of diagnosis, patients typically have lost at least 50% of insulin secretion; within 6 years of diagnosis, insulin secretion decreases to less than 25%.2
The American Association of Clinical Endocrinologists (AACE)3 and the American Diabetes Association/European Association for the Study of Diabetes (ADA/EASD)4 have recently published guidelines for the management of type 2 diabetes. While the AACE’s guidelines (available at https://www.aace.com/files/aace_algorithm.pdf) focus on different treatments at different stages of disease and both glycemic and nonglycemic benefits of treatment,3 the ADA/EASD’s guidelines (see http://care.diabetesjournals.org/content/early/2012/04/17/dc12-0413.full.pdf+html) emphasize a patient-centered approach, shared decision making, and individualization of treatment goals based on both patient preference and comorbid disease states.4
One thing both sets of guidelines have in common is a purposeful intensification of therapy every 2 to 3 months, as needed, and the introduction of insulin one year after diagnosis if the patient is still not at goal.3,4 But all too often, this does not occur, particularly in primary care settings.
This article will review the “when” and “how” of insulin initiation. But first, a look at barriers to insulin therapy and evidence in support of earlier use.
Clinical inertia and patient fear are associated with delays
Both the AACE and the ADA/EASD guidelines agree that metformin is best used as early as possible.5,6 With typical use, however, metformin fails to prevent the progression of diabetes, as measured by the climb of hemoglobin A1c (HbA1c), at a failure rate of about 17% of patients per year.5 Physicians have been slow to intensify treatment for type 2 diabetes6—a phenomenon referred to as clinical inertia.
Typically, physicians adopt a stepwise approach, which often results in patients spending more than 10 years with an HbA1c >7% and 5 years with an HbA1c >8% before insulin is started.5 In a recent Veterans Administration study, patients were out of control, with an HbA1c >8%, for an average of 4.6 years before insulin was initiated.7
Both patient and physician factors contribute to the delay. Patient factors include the fear of injection, the belief that insulin will interfere with their lifestyle, and the idea that the use of insulin signifies impending complications or even death.8 But such beliefs are starting to change. In a recent multinational study of patients with type 2 diabetes, less than 20% stated they were unwilling to start insulin.9
For their part, primary care physicians are much less likely to prescribe insulin than clinicians specializing in diabetes.6 Physician-reported barriers to insulin initiation include the time required to train patients to use it correctly; the lack of support, including access to diabetes educators; and the absence of clear guidelines on the use of insulin.10
A case for earlier insulin
There has been recent momentum in favor of earlier initiation of insulin. In fact, some researchers regard intensive insulin as an excellent first treatment for type 2 diabetes,11 based on the belief that early insulin (used for a brief time) can provide not only immediate improvement in glucose control, but also a lasting “legacy” effect. The ADA/EASD guidelines support the use of insulin as a first-line treatment for patients with symptoms of insulin deficiency,4 but do not recommend it for everyone with newly diagnosed type 2 diabetes.
There have also been a number of advances in insulin therapy over the past 2 decades. These include insulin analogs with physiologic profiles that better match daily schedules, as well as improvements in the way insulin is delivered. Insulin pens, smaller needles, disposable devices, and insulin pumps have made it easier to administer and fine-tune insulin delivery. Despite these improvements and recommendations for earlier implementation, the use of insulin in type 2 diabetes is significantly lower today than in the 1990s.12
When to introduce insulin
Insulin is indicated for patients with type 2 diabetes whose disease is not easily controlled. That includes individuals with decompensated type 2 diabetes, those whose HbA1c remains high even with 2 or more oral agents, and individuals who have not reached goal after a year of treatment.
Glucose toxicity. It is generally agreed that insulin is the most effective treatment for patients who present with decompensated type 2 diabetes4—ie, with significant hyperglycemia and catabolic symptoms such as polydipsia, polyuria, and weight loss. Initiation of insulin promotes reversal of glucose toxicity and stabilization of metabolic status. In such cases, insulin can be started at a low dose to expose the patient to the complexities of injection therapy (more about this in a bit), then titrated as needed for stabilization.
HbA1c ≥8% even with 2 or more drugs. In my experience, an oral diabetes drug will lead to a drop in HbA1c of about one percentage point. Generally, the further from goal the patient is, the greater the effect the medication will have. As HbA1c inches closer to 7%, the effect diminishes. And when 2 oral agents fail to lower a patient’s HbA1c adequately, the incremental change expected from the addition of a third, fourth, or fifth agent is small.
Thus, in a patient with an HbA1c ≥8%, there is still a significant fasting hyperglycemic component. In such a case, a basal insulin is likely the best treatment option.
Not at goal at one year. Both the AACE and the ADA/EASD guidelines agree that treatment titration should be considered every 2 to 3 months to achieve metabolic control and that if a patient is not at goal after a year, insulin should be started.3,4 However, traditionally this is not done. The delayed implementation of this recommendation is an example of clinical inertia, which can contribute to further misunderstandings about the role and effect of insulin therapy.
Getting started with basal insulin
Most patients who are started on insulin have global hyperglycemia. But because fasting hyperglycemia can affect pancreatic insulin secretion, it is important to get control of the fasting glucose first. This can often be done with insulin sensitizers (metformin, thiazolidinediones, and incretin-based agents).
Suppression of excessive hepatic glucose production, which is very common in type 2 diabetes, is one of the biggest challenges in normalizing fasting glucose. This is well managed with a basal insulin. When starting basal insulin, however, it is critical that current treatments not be stopped. Oral agents such as sulfonylureas and meglitinides can be reduced to lower the risk of hypoglycemia, but stopping them altogether will only prolong the time it takes to get to goal.
There are 3 insulin formulations that can serve as basal insulin (TABLE 1).13 Neutral protamine Hagedorn (NPH) is a human insulin that can be used 2 to 3 times daily to provide basal insulin coverage. But long-acting basal analog insulins glargine and detemir, typically administered once a day when used by patients with type 2 diabetes, are a better option.14
While all 3 formulations have similar efficacy for lowering HbA1c, the analog basal insulins have numerous advantages: less weight gain, less hypoglycemia for the same level of glucose control, and less frequent dosing. In addition, glargine and detemir are available in a pen or vial, while generic NPH is available only in a vial. The primary disadvantage of the analogs is cost: A month’s supply—one vial—of NPH sells for approximately $25 (generic) or $94 (brand name); in comparison, a month’s supply (one box of 5 3-mL pens) of detemir and glargine costs about $300 and $320, respectively.15 (Humulin N, a brand-name NPH, is available in a pen, at a cost of approximately $315 per box.)
Use a weight-based initial dose
The recommended starting dose is 0.1 to 0.2 U/kg daily for patients with an HbA1c <8%. If HbA1c is ≥8%, the ADA/EASD guidelines recommend a starting dose of 0.3 to 0.4 U/kg daily4(TABLE 2).3,4,16 While basal insulin is most commonly dosed at bedtime, in fact, basal analog insulins can be given at any time that’s convenient for the patient. Morning dosing may be preferable for individuals with a significant fear of hypoglycemia—a phobia that sometimes causes patients to skip insulin doses and engage in “defensive eating” (ie, eating in an attempt to prevent hypoglycemia rather than because of hunger or the need for nutrition).
Teach injection technique
It is critically important that patients get the first shot in the office, guided by a clinician who can teach proper injection technique. This also helps to dispel the apprehension of self-injection.
In addition to being surprised at how easy and painless injection can be, patients have the opportunity to observe the results and gain confidence in insulin’s efficacy. And, in my experience, adherence to an insulin regimen is much greater if the first injection is administered in an office setting.
(Tech-savvy patients may find it helpful to use a smartphone app, such as Glucose Buddy or Dbees.com, to help manage their diabetes. See “The 13 best diabetes iPhone & Android apps of 2013” at http://www.healthline.com/health-slideshow/top-iphone-android-apps-diabetes.)
Establish a titration schedule
It is important, too, to teach the patient how to titrate the insulin dose from the start, rather than waiting until the next visit to address this. Patient titration—facilitated by a clinician-provided titration schedule (available from the AACE and the ADA/EASD3,4)—has been shown to achieve target glucose levels faster than physician titration.17
I usually suggest that patients increase the basal insulin dose by 3 units every 3 days, with an upper limit of 0.5 U/kg/d, until fasting glucose is consistently between 100 and 150 mg/dL. I advise every patient who starts taking insulin to track morning readings and titrate the dose until one of 3 things occurs:
1) the 0.5 U/kg/d limit is reached;
2) the patient has a glucose reading <100 mg/dL; or
3) the patient achieves his or her HbA1c target (<7% for most patients).
In every case, I recommend that the patient call my office for further instruction.
If the patient has any low glucose readings, I reduce the basal insulin by 5 U/kg/d. If he or she is still above goal, I advise the patient to continue titration, but more slowly. If the patient is at goal, I advise continuing at the current dose.
Basal titration vs mealtime coverage. Most people with type 2 diabetes require between 0.2 and 1 U/kg of basal insulin daily. It is currently recommended that when a patient has titrated to a dose of 0.5 U/kg/d, it is time to look at the glucose pattern to determine whether further titrating basal insulin or addressing prandial hyperglycemia should be the next step.4,18 This requires a change in fingerstick pattern.
The patient can stop the first morning glucose check and start checking before meals and 90 to 120 minutes postmeal. This allows for exploration of the mealtime excursion. Generally, a difference of <50 mg/dL is preferred. If the morning glucose level is at target but HbA1c is high, it is likely that postprandial glucose is contributing to this difference. This is particularly true when the HbA1c is between 7% and 8%. If the glucose pattern shows high postmeal glucose readings, it is much safer to address mealtime insulin (not discussed in this article) than to continue to titrate the basal insulin.4,18
Avoid “overbasalization”—ie, titrating basal insulin beyond its normal role to suppress hepatic glucose production and get the fasting glucose to goal. Doing so puts the patient at risk for unexpected hypoglycemia, as the insulin will now try to overcome hyperglycemia with meals, as well. Basal insulins are not designed to meet insulin requirements at meals. If a patient misses a meal yet continues the same dose of basal insulin, the risk of a hypoglycemic episode increases substantially.
In the pipeline. There are a number of new basal insulins in development, including one that has a prolonged duration of action and the potential for every-other-day injections19 and another that uses an attached polyethylene glycol moiety to slow absorption and prolong its effect.20
The nuts and bolts of insulin prescribing
When you prescribe insulin, there are a number of components to consider.
Pen or vial? In addition to deciding whether to order pen or vial, it is essential to consider the volume of insulin needed. Glargine, detemir, and Humulin N are available in 10 mL vials (100 U/mL) and in 3 mL pens (100 U/mL). (Generic NPH is available in vials only.) Most patients prefer insulin pens, which are more convenient and easier to use than a vial and syringe.
The choice also depends on the dosage, however. A patient on a daily dose of 45 units would need one box of 5 pens (each prefilled pen has a 3 mL, or 300 unit, capacity) to have sufficient insulin for a month. Vials would be preferable for an individual who requires a larger single dose than a pen can dispense at one time (80 units of glargine, 60 units of detemir).
Syringe and needle size. If you are ordering insulin vials, you will also need to specify the correct syringe—available in 0.3 mL (which holds 30 units), 0.5 mL (50 units), and 1 mL (100 units) sizes. If the patient requires <50 units, order a small syringe to ensure that the unit markings are clear; a 1 mL syringe is preferable for those using a larger volume of insulin. Order the smallest syringe, which also has half-unit markings, if the patient is a child.
All needles are fine, with a 29 to 31 gauge, and available in regular (12.7 mm), short (8 mm), mini (5 mm), and nano (4 mm). Recent studies have shown that absorption, safety, and adverse events are similar for all needle lengths across a variety of patient factors,21 but patients generally prefer shorter needles.
Remember, too, to specify the maximum daily dose of insulin—a consideration that will be more important when prescribing mealtime insulin but is worth mentioning here.
Finally, tell patients who are getting started on insulin about www.accurateinsulin.org. Hosted by The Endocrine Society in partnership with the American Association of Diabetes Educators, ADA, American Pharmacists Association, and American College of Osteopathic Family Physicians, among other clinical groups, the Web site is designed to help patients (as well as providers) navigate the initiation and adjustment of insulin.18
CORRESPONDENCE
Jay Shubrook, DO, FAAFP, FACOFP, The Diabetes Institute at Ohio University, Athens, OH 45701; shubrook@ohio.edu
1. 2011 CDC National Diabetes Fact Sheet. Centers for Disease Control and Prevention Web site. Available at http://www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 3, 2014.
2. UKPDS Group. Intensive blood glucose control with sulphonylurea or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-853.
3. Garber AJ, Abrahamson MJ, Barzilay JI, et al. AACE Comprehensive Diabetes Management Algorithm 2013. Endocr Pract. 2013;19:327-336.
4. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: a patient centered approach. A position statement of the ADA and the EASD. Diabetes Care. 2012;35:1364-1379.
5. Brown JB, Nichols GA, Perry A. The burden of treatment failure in type 2 diabetes. Diabetes Care. 2004;27:1535-1540.
6. Shah BR, Hux JE, Laupacis A, et al. Clinical inertia in response to inadequate glycemic control. Diabetes Care. 2005;28:600-606.
7. Parchman ML, Wang CP. Initiation of insulin among veterans with type 2 diabetes and sustained elevation of HbA1c. Primary Care Diabetes. 2012;6:19-25.
8. Peyrot M, Rubin RR, Lauritzen T, et al. Resistance to insulin therapy among patients and providers: results of the cross-national Diabetes Attitudes, Wishes, and Needs (DAWN) study. Diabetes Care. 2005;28:2673-2679.
9. Polonsky WH, Hajos TR, Dain MP, et al. Are patients with type 2 diabetes reluctant to start insulin therapy? An examination of the scope and underpinnings of psychological insulin resistance in a large international trial. Curr Med Res Opin. 2011;27:1169-1174.
10. Kunt T, Snoek FJ. Barriers to insulin initiation and intensification and how to overcome them. Int J Clin Pract Suppl. 2009;(164):6-10.
11. Presswala LS, Shubrook JH. Intensive insulin therapy as the primary treatment of type 2 diabetes. Clin Diabetes. 2011;29:151-153.
12. Li, C, Ford ES, Zhao G, et al. Trends of insulin use among US adults with type 2 diabetes: the Behavioral Risk Factor Surveillance System, 1995-2007. J Diabetes Complications. 2012;12:17-22.
13. Monthly Prescribing Reference (MPR). Insulin. Available at: http://www.empr.com/insulins/article/123739/. Accessed January 10, 2014.
14. Monami M, Marchionni N, Mannucci E. Long acting insulin analogs vs. NPH human insulin in Type 1 diabetes. A metaanalysis. Diabetes Obes Metab. 2009;11:372-378.
15. Goodrx Web site. Available at: www.goodrx.com. Accessed January 4, 2014.
16. LaSalle JR, Berria R. Insulin therapy in type 2 diabetes: a practical approach for primary care physicians and other health professionals. J Am Osteopath Assoc. 2013;113:152-162.
17. Davies M, Storms F, Shutler S, et al; ATLANTUS Study Group. Improvement of glycemic control in subjects with poorly controlled type 2 diabetes: comparison of two treatment algorithms using insulin glargine. Diabetes Care. 2005;28:1282-1288.
18. Accurate Insulin Decisions. The Endocrine Society Web site. Available at: http://www.accurateinsulin.org/. Accessed January 4, 2014.
19. Keating GM. Insulin degludec and insulin degludec/insulin aspart: a review of their use in the management of diabetes mellitus. Drugs. 2013;73:575-593.
20. Bergenstal RM, Rosenstock J, Arakaki RF, et al. A randomized, controlled study of once –daily LY2605541, a novel long acting basal insulin, versus insulin glargine in basal Insulin treated with patients in type 2 diabetes. Diabetes Care. 2012;35:2140-2147.
21. Hirsch LJ, Gibney MA, Li L, et al. Glycemic control, reported pain and leakage with a 4 mm × 32 G pen needle in obese and non-obese adults with diabetes: a post hoc analysis. Curr Med Res Opin. 2012;28:1305-1311.
› Initiate insulin for patients whose hemoglobin A1c ≥8% despite taking 2 or more oral agents. C
› Prescribe insulin for patients who have not reached their goal one year after diagnosis and initiation of oral therapy. C
› Consider reducing—but do not discontinue—oral agents, such as sulfonylureas and meglitinides, when you initiate insulin therapy. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
With type 2 diabetes now affecting 8.3% of the US population, most primary care physicians see patients with this disorder every day.1 Based on the concurrent obesity epidemic, aging population, and emergence of type 2 diabetes in children and adolescents, it is estimated that by 2050, the prevalence will have risen from one in 12 Americans to one in 3.1
Type 2 diabetes is a progressive disorder, with a relentless decline in beta cells. By the time of diagnosis, patients typically have lost at least 50% of insulin secretion; within 6 years of diagnosis, insulin secretion decreases to less than 25%.2
The American Association of Clinical Endocrinologists (AACE)3 and the American Diabetes Association/European Association for the Study of Diabetes (ADA/EASD)4 have recently published guidelines for the management of type 2 diabetes. While the AACE’s guidelines (available at https://www.aace.com/files/aace_algorithm.pdf) focus on different treatments at different stages of disease and both glycemic and nonglycemic benefits of treatment,3 the ADA/EASD’s guidelines (see http://care.diabetesjournals.org/content/early/2012/04/17/dc12-0413.full.pdf+html) emphasize a patient-centered approach, shared decision making, and individualization of treatment goals based on both patient preference and comorbid disease states.4
One thing both sets of guidelines have in common is a purposeful intensification of therapy every 2 to 3 months, as needed, and the introduction of insulin one year after diagnosis if the patient is still not at goal.3,4 But all too often, this does not occur, particularly in primary care settings.
This article will review the “when” and “how” of insulin initiation. But first, a look at barriers to insulin therapy and evidence in support of earlier use.
Clinical inertia and patient fear are associated with delays
Both the AACE and the ADA/EASD guidelines agree that metformin is best used as early as possible.5,6 With typical use, however, metformin fails to prevent the progression of diabetes, as measured by the climb of hemoglobin A1c (HbA1c), at a failure rate of about 17% of patients per year.5 Physicians have been slow to intensify treatment for type 2 diabetes6—a phenomenon referred to as clinical inertia.
Typically, physicians adopt a stepwise approach, which often results in patients spending more than 10 years with an HbA1c >7% and 5 years with an HbA1c >8% before insulin is started.5 In a recent Veterans Administration study, patients were out of control, with an HbA1c >8%, for an average of 4.6 years before insulin was initiated.7
Both patient and physician factors contribute to the delay. Patient factors include the fear of injection, the belief that insulin will interfere with their lifestyle, and the idea that the use of insulin signifies impending complications or even death.8 But such beliefs are starting to change. In a recent multinational study of patients with type 2 diabetes, less than 20% stated they were unwilling to start insulin.9
For their part, primary care physicians are much less likely to prescribe insulin than clinicians specializing in diabetes.6 Physician-reported barriers to insulin initiation include the time required to train patients to use it correctly; the lack of support, including access to diabetes educators; and the absence of clear guidelines on the use of insulin.10
A case for earlier insulin
There has been recent momentum in favor of earlier initiation of insulin. In fact, some researchers regard intensive insulin as an excellent first treatment for type 2 diabetes,11 based on the belief that early insulin (used for a brief time) can provide not only immediate improvement in glucose control, but also a lasting “legacy” effect. The ADA/EASD guidelines support the use of insulin as a first-line treatment for patients with symptoms of insulin deficiency,4 but do not recommend it for everyone with newly diagnosed type 2 diabetes.
There have also been a number of advances in insulin therapy over the past 2 decades. These include insulin analogs with physiologic profiles that better match daily schedules, as well as improvements in the way insulin is delivered. Insulin pens, smaller needles, disposable devices, and insulin pumps have made it easier to administer and fine-tune insulin delivery. Despite these improvements and recommendations for earlier implementation, the use of insulin in type 2 diabetes is significantly lower today than in the 1990s.12
When to introduce insulin
Insulin is indicated for patients with type 2 diabetes whose disease is not easily controlled. That includes individuals with decompensated type 2 diabetes, those whose HbA1c remains high even with 2 or more oral agents, and individuals who have not reached goal after a year of treatment.
Glucose toxicity. It is generally agreed that insulin is the most effective treatment for patients who present with decompensated type 2 diabetes4—ie, with significant hyperglycemia and catabolic symptoms such as polydipsia, polyuria, and weight loss. Initiation of insulin promotes reversal of glucose toxicity and stabilization of metabolic status. In such cases, insulin can be started at a low dose to expose the patient to the complexities of injection therapy (more about this in a bit), then titrated as needed for stabilization.
HbA1c ≥8% even with 2 or more drugs. In my experience, an oral diabetes drug will lead to a drop in HbA1c of about one percentage point. Generally, the further from goal the patient is, the greater the effect the medication will have. As HbA1c inches closer to 7%, the effect diminishes. And when 2 oral agents fail to lower a patient’s HbA1c adequately, the incremental change expected from the addition of a third, fourth, or fifth agent is small.
Thus, in a patient with an HbA1c ≥8%, there is still a significant fasting hyperglycemic component. In such a case, a basal insulin is likely the best treatment option.
Not at goal at one year. Both the AACE and the ADA/EASD guidelines agree that treatment titration should be considered every 2 to 3 months to achieve metabolic control and that if a patient is not at goal after a year, insulin should be started.3,4 However, traditionally this is not done. The delayed implementation of this recommendation is an example of clinical inertia, which can contribute to further misunderstandings about the role and effect of insulin therapy.
Getting started with basal insulin
Most patients who are started on insulin have global hyperglycemia. But because fasting hyperglycemia can affect pancreatic insulin secretion, it is important to get control of the fasting glucose first. This can often be done with insulin sensitizers (metformin, thiazolidinediones, and incretin-based agents).
Suppression of excessive hepatic glucose production, which is very common in type 2 diabetes, is one of the biggest challenges in normalizing fasting glucose. This is well managed with a basal insulin. When starting basal insulin, however, it is critical that current treatments not be stopped. Oral agents such as sulfonylureas and meglitinides can be reduced to lower the risk of hypoglycemia, but stopping them altogether will only prolong the time it takes to get to goal.
There are 3 insulin formulations that can serve as basal insulin (TABLE 1).13 Neutral protamine Hagedorn (NPH) is a human insulin that can be used 2 to 3 times daily to provide basal insulin coverage. But long-acting basal analog insulins glargine and detemir, typically administered once a day when used by patients with type 2 diabetes, are a better option.14
While all 3 formulations have similar efficacy for lowering HbA1c, the analog basal insulins have numerous advantages: less weight gain, less hypoglycemia for the same level of glucose control, and less frequent dosing. In addition, glargine and detemir are available in a pen or vial, while generic NPH is available only in a vial. The primary disadvantage of the analogs is cost: A month’s supply—one vial—of NPH sells for approximately $25 (generic) or $94 (brand name); in comparison, a month’s supply (one box of 5 3-mL pens) of detemir and glargine costs about $300 and $320, respectively.15 (Humulin N, a brand-name NPH, is available in a pen, at a cost of approximately $315 per box.)
Use a weight-based initial dose
The recommended starting dose is 0.1 to 0.2 U/kg daily for patients with an HbA1c <8%. If HbA1c is ≥8%, the ADA/EASD guidelines recommend a starting dose of 0.3 to 0.4 U/kg daily4(TABLE 2).3,4,16 While basal insulin is most commonly dosed at bedtime, in fact, basal analog insulins can be given at any time that’s convenient for the patient. Morning dosing may be preferable for individuals with a significant fear of hypoglycemia—a phobia that sometimes causes patients to skip insulin doses and engage in “defensive eating” (ie, eating in an attempt to prevent hypoglycemia rather than because of hunger or the need for nutrition).
Teach injection technique
It is critically important that patients get the first shot in the office, guided by a clinician who can teach proper injection technique. This also helps to dispel the apprehension of self-injection.
In addition to being surprised at how easy and painless injection can be, patients have the opportunity to observe the results and gain confidence in insulin’s efficacy. And, in my experience, adherence to an insulin regimen is much greater if the first injection is administered in an office setting.
(Tech-savvy patients may find it helpful to use a smartphone app, such as Glucose Buddy or Dbees.com, to help manage their diabetes. See “The 13 best diabetes iPhone & Android apps of 2013” at http://www.healthline.com/health-slideshow/top-iphone-android-apps-diabetes.)
Establish a titration schedule
It is important, too, to teach the patient how to titrate the insulin dose from the start, rather than waiting until the next visit to address this. Patient titration—facilitated by a clinician-provided titration schedule (available from the AACE and the ADA/EASD3,4)—has been shown to achieve target glucose levels faster than physician titration.17
I usually suggest that patients increase the basal insulin dose by 3 units every 3 days, with an upper limit of 0.5 U/kg/d, until fasting glucose is consistently between 100 and 150 mg/dL. I advise every patient who starts taking insulin to track morning readings and titrate the dose until one of 3 things occurs:
1) the 0.5 U/kg/d limit is reached;
2) the patient has a glucose reading <100 mg/dL; or
3) the patient achieves his or her HbA1c target (<7% for most patients).
In every case, I recommend that the patient call my office for further instruction.
If the patient has any low glucose readings, I reduce the basal insulin by 5 U/kg/d. If he or she is still above goal, I advise the patient to continue titration, but more slowly. If the patient is at goal, I advise continuing at the current dose.
Basal titration vs mealtime coverage. Most people with type 2 diabetes require between 0.2 and 1 U/kg of basal insulin daily. It is currently recommended that when a patient has titrated to a dose of 0.5 U/kg/d, it is time to look at the glucose pattern to determine whether further titrating basal insulin or addressing prandial hyperglycemia should be the next step.4,18 This requires a change in fingerstick pattern.
The patient can stop the first morning glucose check and start checking before meals and 90 to 120 minutes postmeal. This allows for exploration of the mealtime excursion. Generally, a difference of <50 mg/dL is preferred. If the morning glucose level is at target but HbA1c is high, it is likely that postprandial glucose is contributing to this difference. This is particularly true when the HbA1c is between 7% and 8%. If the glucose pattern shows high postmeal glucose readings, it is much safer to address mealtime insulin (not discussed in this article) than to continue to titrate the basal insulin.4,18
Avoid “overbasalization”—ie, titrating basal insulin beyond its normal role to suppress hepatic glucose production and get the fasting glucose to goal. Doing so puts the patient at risk for unexpected hypoglycemia, as the insulin will now try to overcome hyperglycemia with meals, as well. Basal insulins are not designed to meet insulin requirements at meals. If a patient misses a meal yet continues the same dose of basal insulin, the risk of a hypoglycemic episode increases substantially.
In the pipeline. There are a number of new basal insulins in development, including one that has a prolonged duration of action and the potential for every-other-day injections19 and another that uses an attached polyethylene glycol moiety to slow absorption and prolong its effect.20
The nuts and bolts of insulin prescribing
When you prescribe insulin, there are a number of components to consider.
Pen or vial? In addition to deciding whether to order pen or vial, it is essential to consider the volume of insulin needed. Glargine, detemir, and Humulin N are available in 10 mL vials (100 U/mL) and in 3 mL pens (100 U/mL). (Generic NPH is available in vials only.) Most patients prefer insulin pens, which are more convenient and easier to use than a vial and syringe.
The choice also depends on the dosage, however. A patient on a daily dose of 45 units would need one box of 5 pens (each prefilled pen has a 3 mL, or 300 unit, capacity) to have sufficient insulin for a month. Vials would be preferable for an individual who requires a larger single dose than a pen can dispense at one time (80 units of glargine, 60 units of detemir).
Syringe and needle size. If you are ordering insulin vials, you will also need to specify the correct syringe—available in 0.3 mL (which holds 30 units), 0.5 mL (50 units), and 1 mL (100 units) sizes. If the patient requires <50 units, order a small syringe to ensure that the unit markings are clear; a 1 mL syringe is preferable for those using a larger volume of insulin. Order the smallest syringe, which also has half-unit markings, if the patient is a child.
All needles are fine, with a 29 to 31 gauge, and available in regular (12.7 mm), short (8 mm), mini (5 mm), and nano (4 mm). Recent studies have shown that absorption, safety, and adverse events are similar for all needle lengths across a variety of patient factors,21 but patients generally prefer shorter needles.
Remember, too, to specify the maximum daily dose of insulin—a consideration that will be more important when prescribing mealtime insulin but is worth mentioning here.
Finally, tell patients who are getting started on insulin about www.accurateinsulin.org. Hosted by The Endocrine Society in partnership with the American Association of Diabetes Educators, ADA, American Pharmacists Association, and American College of Osteopathic Family Physicians, among other clinical groups, the Web site is designed to help patients (as well as providers) navigate the initiation and adjustment of insulin.18
CORRESPONDENCE
Jay Shubrook, DO, FAAFP, FACOFP, The Diabetes Institute at Ohio University, Athens, OH 45701; shubrook@ohio.edu
› Initiate insulin for patients whose hemoglobin A1c ≥8% despite taking 2 or more oral agents. C
› Prescribe insulin for patients who have not reached their goal one year after diagnosis and initiation of oral therapy. C
› Consider reducing—but do not discontinue—oral agents, such as sulfonylureas and meglitinides, when you initiate insulin therapy. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
With type 2 diabetes now affecting 8.3% of the US population, most primary care physicians see patients with this disorder every day.1 Based on the concurrent obesity epidemic, aging population, and emergence of type 2 diabetes in children and adolescents, it is estimated that by 2050, the prevalence will have risen from one in 12 Americans to one in 3.1
Type 2 diabetes is a progressive disorder, with a relentless decline in beta cells. By the time of diagnosis, patients typically have lost at least 50% of insulin secretion; within 6 years of diagnosis, insulin secretion decreases to less than 25%.2
The American Association of Clinical Endocrinologists (AACE)3 and the American Diabetes Association/European Association for the Study of Diabetes (ADA/EASD)4 have recently published guidelines for the management of type 2 diabetes. While the AACE’s guidelines (available at https://www.aace.com/files/aace_algorithm.pdf) focus on different treatments at different stages of disease and both glycemic and nonglycemic benefits of treatment,3 the ADA/EASD’s guidelines (see http://care.diabetesjournals.org/content/early/2012/04/17/dc12-0413.full.pdf+html) emphasize a patient-centered approach, shared decision making, and individualization of treatment goals based on both patient preference and comorbid disease states.4
One thing both sets of guidelines have in common is a purposeful intensification of therapy every 2 to 3 months, as needed, and the introduction of insulin one year after diagnosis if the patient is still not at goal.3,4 But all too often, this does not occur, particularly in primary care settings.
This article will review the “when” and “how” of insulin initiation. But first, a look at barriers to insulin therapy and evidence in support of earlier use.
Clinical inertia and patient fear are associated with delays
Both the AACE and the ADA/EASD guidelines agree that metformin is best used as early as possible.5,6 With typical use, however, metformin fails to prevent the progression of diabetes, as measured by the climb of hemoglobin A1c (HbA1c), at a failure rate of about 17% of patients per year.5 Physicians have been slow to intensify treatment for type 2 diabetes6—a phenomenon referred to as clinical inertia.
Typically, physicians adopt a stepwise approach, which often results in patients spending more than 10 years with an HbA1c >7% and 5 years with an HbA1c >8% before insulin is started.5 In a recent Veterans Administration study, patients were out of control, with an HbA1c >8%, for an average of 4.6 years before insulin was initiated.7
Both patient and physician factors contribute to the delay. Patient factors include the fear of injection, the belief that insulin will interfere with their lifestyle, and the idea that the use of insulin signifies impending complications or even death.8 But such beliefs are starting to change. In a recent multinational study of patients with type 2 diabetes, less than 20% stated they were unwilling to start insulin.9
For their part, primary care physicians are much less likely to prescribe insulin than clinicians specializing in diabetes.6 Physician-reported barriers to insulin initiation include the time required to train patients to use it correctly; the lack of support, including access to diabetes educators; and the absence of clear guidelines on the use of insulin.10
A case for earlier insulin
There has been recent momentum in favor of earlier initiation of insulin. In fact, some researchers regard intensive insulin as an excellent first treatment for type 2 diabetes,11 based on the belief that early insulin (used for a brief time) can provide not only immediate improvement in glucose control, but also a lasting “legacy” effect. The ADA/EASD guidelines support the use of insulin as a first-line treatment for patients with symptoms of insulin deficiency,4 but do not recommend it for everyone with newly diagnosed type 2 diabetes.
There have also been a number of advances in insulin therapy over the past 2 decades. These include insulin analogs with physiologic profiles that better match daily schedules, as well as improvements in the way insulin is delivered. Insulin pens, smaller needles, disposable devices, and insulin pumps have made it easier to administer and fine-tune insulin delivery. Despite these improvements and recommendations for earlier implementation, the use of insulin in type 2 diabetes is significantly lower today than in the 1990s.12
When to introduce insulin
Insulin is indicated for patients with type 2 diabetes whose disease is not easily controlled. That includes individuals with decompensated type 2 diabetes, those whose HbA1c remains high even with 2 or more oral agents, and individuals who have not reached goal after a year of treatment.
Glucose toxicity. It is generally agreed that insulin is the most effective treatment for patients who present with decompensated type 2 diabetes4—ie, with significant hyperglycemia and catabolic symptoms such as polydipsia, polyuria, and weight loss. Initiation of insulin promotes reversal of glucose toxicity and stabilization of metabolic status. In such cases, insulin can be started at a low dose to expose the patient to the complexities of injection therapy (more about this in a bit), then titrated as needed for stabilization.
HbA1c ≥8% even with 2 or more drugs. In my experience, an oral diabetes drug will lead to a drop in HbA1c of about one percentage point. Generally, the further from goal the patient is, the greater the effect the medication will have. As HbA1c inches closer to 7%, the effect diminishes. And when 2 oral agents fail to lower a patient’s HbA1c adequately, the incremental change expected from the addition of a third, fourth, or fifth agent is small.
Thus, in a patient with an HbA1c ≥8%, there is still a significant fasting hyperglycemic component. In such a case, a basal insulin is likely the best treatment option.
Not at goal at one year. Both the AACE and the ADA/EASD guidelines agree that treatment titration should be considered every 2 to 3 months to achieve metabolic control and that if a patient is not at goal after a year, insulin should be started.3,4 However, traditionally this is not done. The delayed implementation of this recommendation is an example of clinical inertia, which can contribute to further misunderstandings about the role and effect of insulin therapy.
Getting started with basal insulin
Most patients who are started on insulin have global hyperglycemia. But because fasting hyperglycemia can affect pancreatic insulin secretion, it is important to get control of the fasting glucose first. This can often be done with insulin sensitizers (metformin, thiazolidinediones, and incretin-based agents).
Suppression of excessive hepatic glucose production, which is very common in type 2 diabetes, is one of the biggest challenges in normalizing fasting glucose. This is well managed with a basal insulin. When starting basal insulin, however, it is critical that current treatments not be stopped. Oral agents such as sulfonylureas and meglitinides can be reduced to lower the risk of hypoglycemia, but stopping them altogether will only prolong the time it takes to get to goal.
There are 3 insulin formulations that can serve as basal insulin (TABLE 1).13 Neutral protamine Hagedorn (NPH) is a human insulin that can be used 2 to 3 times daily to provide basal insulin coverage. But long-acting basal analog insulins glargine and detemir, typically administered once a day when used by patients with type 2 diabetes, are a better option.14
While all 3 formulations have similar efficacy for lowering HbA1c, the analog basal insulins have numerous advantages: less weight gain, less hypoglycemia for the same level of glucose control, and less frequent dosing. In addition, glargine and detemir are available in a pen or vial, while generic NPH is available only in a vial. The primary disadvantage of the analogs is cost: A month’s supply—one vial—of NPH sells for approximately $25 (generic) or $94 (brand name); in comparison, a month’s supply (one box of 5 3-mL pens) of detemir and glargine costs about $300 and $320, respectively.15 (Humulin N, a brand-name NPH, is available in a pen, at a cost of approximately $315 per box.)
Use a weight-based initial dose
The recommended starting dose is 0.1 to 0.2 U/kg daily for patients with an HbA1c <8%. If HbA1c is ≥8%, the ADA/EASD guidelines recommend a starting dose of 0.3 to 0.4 U/kg daily4(TABLE 2).3,4,16 While basal insulin is most commonly dosed at bedtime, in fact, basal analog insulins can be given at any time that’s convenient for the patient. Morning dosing may be preferable for individuals with a significant fear of hypoglycemia—a phobia that sometimes causes patients to skip insulin doses and engage in “defensive eating” (ie, eating in an attempt to prevent hypoglycemia rather than because of hunger or the need for nutrition).
Teach injection technique
It is critically important that patients get the first shot in the office, guided by a clinician who can teach proper injection technique. This also helps to dispel the apprehension of self-injection.
In addition to being surprised at how easy and painless injection can be, patients have the opportunity to observe the results and gain confidence in insulin’s efficacy. And, in my experience, adherence to an insulin regimen is much greater if the first injection is administered in an office setting.
(Tech-savvy patients may find it helpful to use a smartphone app, such as Glucose Buddy or Dbees.com, to help manage their diabetes. See “The 13 best diabetes iPhone & Android apps of 2013” at http://www.healthline.com/health-slideshow/top-iphone-android-apps-diabetes.)
Establish a titration schedule
It is important, too, to teach the patient how to titrate the insulin dose from the start, rather than waiting until the next visit to address this. Patient titration—facilitated by a clinician-provided titration schedule (available from the AACE and the ADA/EASD3,4)—has been shown to achieve target glucose levels faster than physician titration.17
I usually suggest that patients increase the basal insulin dose by 3 units every 3 days, with an upper limit of 0.5 U/kg/d, until fasting glucose is consistently between 100 and 150 mg/dL. I advise every patient who starts taking insulin to track morning readings and titrate the dose until one of 3 things occurs:
1) the 0.5 U/kg/d limit is reached;
2) the patient has a glucose reading <100 mg/dL; or
3) the patient achieves his or her HbA1c target (<7% for most patients).
In every case, I recommend that the patient call my office for further instruction.
If the patient has any low glucose readings, I reduce the basal insulin by 5 U/kg/d. If he or she is still above goal, I advise the patient to continue titration, but more slowly. If the patient is at goal, I advise continuing at the current dose.
Basal titration vs mealtime coverage. Most people with type 2 diabetes require between 0.2 and 1 U/kg of basal insulin daily. It is currently recommended that when a patient has titrated to a dose of 0.5 U/kg/d, it is time to look at the glucose pattern to determine whether further titrating basal insulin or addressing prandial hyperglycemia should be the next step.4,18 This requires a change in fingerstick pattern.
The patient can stop the first morning glucose check and start checking before meals and 90 to 120 minutes postmeal. This allows for exploration of the mealtime excursion. Generally, a difference of <50 mg/dL is preferred. If the morning glucose level is at target but HbA1c is high, it is likely that postprandial glucose is contributing to this difference. This is particularly true when the HbA1c is between 7% and 8%. If the glucose pattern shows high postmeal glucose readings, it is much safer to address mealtime insulin (not discussed in this article) than to continue to titrate the basal insulin.4,18
Avoid “overbasalization”—ie, titrating basal insulin beyond its normal role to suppress hepatic glucose production and get the fasting glucose to goal. Doing so puts the patient at risk for unexpected hypoglycemia, as the insulin will now try to overcome hyperglycemia with meals, as well. Basal insulins are not designed to meet insulin requirements at meals. If a patient misses a meal yet continues the same dose of basal insulin, the risk of a hypoglycemic episode increases substantially.
In the pipeline. There are a number of new basal insulins in development, including one that has a prolonged duration of action and the potential for every-other-day injections19 and another that uses an attached polyethylene glycol moiety to slow absorption and prolong its effect.20
The nuts and bolts of insulin prescribing
When you prescribe insulin, there are a number of components to consider.
Pen or vial? In addition to deciding whether to order pen or vial, it is essential to consider the volume of insulin needed. Glargine, detemir, and Humulin N are available in 10 mL vials (100 U/mL) and in 3 mL pens (100 U/mL). (Generic NPH is available in vials only.) Most patients prefer insulin pens, which are more convenient and easier to use than a vial and syringe.
The choice also depends on the dosage, however. A patient on a daily dose of 45 units would need one box of 5 pens (each prefilled pen has a 3 mL, or 300 unit, capacity) to have sufficient insulin for a month. Vials would be preferable for an individual who requires a larger single dose than a pen can dispense at one time (80 units of glargine, 60 units of detemir).
Syringe and needle size. If you are ordering insulin vials, you will also need to specify the correct syringe—available in 0.3 mL (which holds 30 units), 0.5 mL (50 units), and 1 mL (100 units) sizes. If the patient requires <50 units, order a small syringe to ensure that the unit markings are clear; a 1 mL syringe is preferable for those using a larger volume of insulin. Order the smallest syringe, which also has half-unit markings, if the patient is a child.
All needles are fine, with a 29 to 31 gauge, and available in regular (12.7 mm), short (8 mm), mini (5 mm), and nano (4 mm). Recent studies have shown that absorption, safety, and adverse events are similar for all needle lengths across a variety of patient factors,21 but patients generally prefer shorter needles.
Remember, too, to specify the maximum daily dose of insulin—a consideration that will be more important when prescribing mealtime insulin but is worth mentioning here.
Finally, tell patients who are getting started on insulin about www.accurateinsulin.org. Hosted by The Endocrine Society in partnership with the American Association of Diabetes Educators, ADA, American Pharmacists Association, and American College of Osteopathic Family Physicians, among other clinical groups, the Web site is designed to help patients (as well as providers) navigate the initiation and adjustment of insulin.18
CORRESPONDENCE
Jay Shubrook, DO, FAAFP, FACOFP, The Diabetes Institute at Ohio University, Athens, OH 45701; shubrook@ohio.edu
1. 2011 CDC National Diabetes Fact Sheet. Centers for Disease Control and Prevention Web site. Available at http://www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 3, 2014.
2. UKPDS Group. Intensive blood glucose control with sulphonylurea or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-853.
3. Garber AJ, Abrahamson MJ, Barzilay JI, et al. AACE Comprehensive Diabetes Management Algorithm 2013. Endocr Pract. 2013;19:327-336.
4. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: a patient centered approach. A position statement of the ADA and the EASD. Diabetes Care. 2012;35:1364-1379.
5. Brown JB, Nichols GA, Perry A. The burden of treatment failure in type 2 diabetes. Diabetes Care. 2004;27:1535-1540.
6. Shah BR, Hux JE, Laupacis A, et al. Clinical inertia in response to inadequate glycemic control. Diabetes Care. 2005;28:600-606.
7. Parchman ML, Wang CP. Initiation of insulin among veterans with type 2 diabetes and sustained elevation of HbA1c. Primary Care Diabetes. 2012;6:19-25.
8. Peyrot M, Rubin RR, Lauritzen T, et al. Resistance to insulin therapy among patients and providers: results of the cross-national Diabetes Attitudes, Wishes, and Needs (DAWN) study. Diabetes Care. 2005;28:2673-2679.
9. Polonsky WH, Hajos TR, Dain MP, et al. Are patients with type 2 diabetes reluctant to start insulin therapy? An examination of the scope and underpinnings of psychological insulin resistance in a large international trial. Curr Med Res Opin. 2011;27:1169-1174.
10. Kunt T, Snoek FJ. Barriers to insulin initiation and intensification and how to overcome them. Int J Clin Pract Suppl. 2009;(164):6-10.
11. Presswala LS, Shubrook JH. Intensive insulin therapy as the primary treatment of type 2 diabetes. Clin Diabetes. 2011;29:151-153.
12. Li, C, Ford ES, Zhao G, et al. Trends of insulin use among US adults with type 2 diabetes: the Behavioral Risk Factor Surveillance System, 1995-2007. J Diabetes Complications. 2012;12:17-22.
13. Monthly Prescribing Reference (MPR). Insulin. Available at: http://www.empr.com/insulins/article/123739/. Accessed January 10, 2014.
14. Monami M, Marchionni N, Mannucci E. Long acting insulin analogs vs. NPH human insulin in Type 1 diabetes. A metaanalysis. Diabetes Obes Metab. 2009;11:372-378.
15. Goodrx Web site. Available at: www.goodrx.com. Accessed January 4, 2014.
16. LaSalle JR, Berria R. Insulin therapy in type 2 diabetes: a practical approach for primary care physicians and other health professionals. J Am Osteopath Assoc. 2013;113:152-162.
17. Davies M, Storms F, Shutler S, et al; ATLANTUS Study Group. Improvement of glycemic control in subjects with poorly controlled type 2 diabetes: comparison of two treatment algorithms using insulin glargine. Diabetes Care. 2005;28:1282-1288.
18. Accurate Insulin Decisions. The Endocrine Society Web site. Available at: http://www.accurateinsulin.org/. Accessed January 4, 2014.
19. Keating GM. Insulin degludec and insulin degludec/insulin aspart: a review of their use in the management of diabetes mellitus. Drugs. 2013;73:575-593.
20. Bergenstal RM, Rosenstock J, Arakaki RF, et al. A randomized, controlled study of once –daily LY2605541, a novel long acting basal insulin, versus insulin glargine in basal Insulin treated with patients in type 2 diabetes. Diabetes Care. 2012;35:2140-2147.
21. Hirsch LJ, Gibney MA, Li L, et al. Glycemic control, reported pain and leakage with a 4 mm × 32 G pen needle in obese and non-obese adults with diabetes: a post hoc analysis. Curr Med Res Opin. 2012;28:1305-1311.
1. 2011 CDC National Diabetes Fact Sheet. Centers for Disease Control and Prevention Web site. Available at http://www.cdc.gov/diabetes/pubs/factsheet11.htm. Accessed January 3, 2014.
2. UKPDS Group. Intensive blood glucose control with sulphonylurea or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-853.
3. Garber AJ, Abrahamson MJ, Barzilay JI, et al. AACE Comprehensive Diabetes Management Algorithm 2013. Endocr Pract. 2013;19:327-336.
4. Inzucchi SE, Bergenstal RM, Buse JB, et al; American Diabetes Association (ADA); European Association for the Study of Diabetes (EASD). Management of hyperglycemia in type 2 diabetes: a patient centered approach. A position statement of the ADA and the EASD. Diabetes Care. 2012;35:1364-1379.
5. Brown JB, Nichols GA, Perry A. The burden of treatment failure in type 2 diabetes. Diabetes Care. 2004;27:1535-1540.
6. Shah BR, Hux JE, Laupacis A, et al. Clinical inertia in response to inadequate glycemic control. Diabetes Care. 2005;28:600-606.
7. Parchman ML, Wang CP. Initiation of insulin among veterans with type 2 diabetes and sustained elevation of HbA1c. Primary Care Diabetes. 2012;6:19-25.
8. Peyrot M, Rubin RR, Lauritzen T, et al. Resistance to insulin therapy among patients and providers: results of the cross-national Diabetes Attitudes, Wishes, and Needs (DAWN) study. Diabetes Care. 2005;28:2673-2679.
9. Polonsky WH, Hajos TR, Dain MP, et al. Are patients with type 2 diabetes reluctant to start insulin therapy? An examination of the scope and underpinnings of psychological insulin resistance in a large international trial. Curr Med Res Opin. 2011;27:1169-1174.
10. Kunt T, Snoek FJ. Barriers to insulin initiation and intensification and how to overcome them. Int J Clin Pract Suppl. 2009;(164):6-10.
11. Presswala LS, Shubrook JH. Intensive insulin therapy as the primary treatment of type 2 diabetes. Clin Diabetes. 2011;29:151-153.
12. Li, C, Ford ES, Zhao G, et al. Trends of insulin use among US adults with type 2 diabetes: the Behavioral Risk Factor Surveillance System, 1995-2007. J Diabetes Complications. 2012;12:17-22.
13. Monthly Prescribing Reference (MPR). Insulin. Available at: http://www.empr.com/insulins/article/123739/. Accessed January 10, 2014.
14. Monami M, Marchionni N, Mannucci E. Long acting insulin analogs vs. NPH human insulin in Type 1 diabetes. A metaanalysis. Diabetes Obes Metab. 2009;11:372-378.
15. Goodrx Web site. Available at: www.goodrx.com. Accessed January 4, 2014.
16. LaSalle JR, Berria R. Insulin therapy in type 2 diabetes: a practical approach for primary care physicians and other health professionals. J Am Osteopath Assoc. 2013;113:152-162.
17. Davies M, Storms F, Shutler S, et al; ATLANTUS Study Group. Improvement of glycemic control in subjects with poorly controlled type 2 diabetes: comparison of two treatment algorithms using insulin glargine. Diabetes Care. 2005;28:1282-1288.
18. Accurate Insulin Decisions. The Endocrine Society Web site. Available at: http://www.accurateinsulin.org/. Accessed January 4, 2014.
19. Keating GM. Insulin degludec and insulin degludec/insulin aspart: a review of their use in the management of diabetes mellitus. Drugs. 2013;73:575-593.
20. Bergenstal RM, Rosenstock J, Arakaki RF, et al. A randomized, controlled study of once –daily LY2605541, a novel long acting basal insulin, versus insulin glargine in basal Insulin treated with patients in type 2 diabetes. Diabetes Care. 2012;35:2140-2147.
21. Hirsch LJ, Gibney MA, Li L, et al. Glycemic control, reported pain and leakage with a 4 mm × 32 G pen needle in obese and non-obese adults with diabetes: a post hoc analysis. Curr Med Res Opin. 2012;28:1305-1311.