User login
Myopathy for the general internist: Statins and much more
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
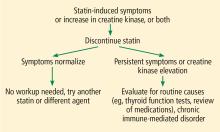
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
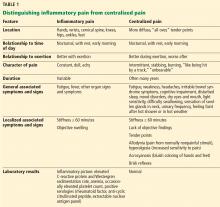
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
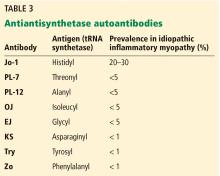
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
Myopathies can present with a wide variety of symptoms, so patients with muscle weakness are often seen initially by a general practitioner. Nonrheumatologists should be able to evaluate a patient presenting with muscle weakness or myalgia and be aware of red flags indicating potentially dangerous syndromes that require a prompt, thorough investigation.
This article reviews selected causes of muscle weakness, such as statin-induced and autoimmune disorders, and systemic features of inflammatory myopathies beyond myositis, such as dermatologic and pulmonary manifestations.
FOCUSING THE EVALUATION
The evaluation of a patient presenting with muscle weakness should include several assessments:
Temporal progression. Was the onset of symptoms rapid or insidious? Patterns of onset may give clues to etiology, including the possibility of an associated autoimmune condition.
Location of muscle weakness. Are symptoms global or localized? And if localized, are they proximal or distal? Proximal weakness can be manifested by difficulty rising from a chair (hip muscles) or combing one’s hair (shoulder muscles), whereas distal weakness can involve difficulty standing on toes (gastrocnemius and soleus muscles) or performing fine motor activities (intrinsic hand muscles).
Symmetry. A focal or asymmetric pattern often has a neurologic etiology, but this could also be consistent with inclusion body myositis.
Other symptoms. Arthritis, rash, and swallowing problems point to a possible underlying rheumatologic disease. Weight gain or loss may indicate a thyroid disorder.
Family history. Some patients report that others in their family have this pattern of weakness, indicating a likely genetic myopathy. If the patient reports a relative with multiple sclerosis, lupus erythematosus, rheumatoid arthritis, or another autoimmune disease, then an immune-mediated myopathy should be considered.
Medications should be reviewed, particularly statins.
CASE 1: SLOWLY PROGRESSIVE WEAKNESS
A 65-year-old man presented with the insidious onset of muscle weakness and episodes of falling. On review of his medical record, his serum creatine kinase (CK) levels were elevated at various periods at 2 to 4 times the upper limit of normal. Electromyography (EMG) previously showed a myopathic pattern, and a muscle biopsy was abnormal, consistent with endomysial inflammation (term is consistent with “polymyositis”). He was treated for polymyositis for several years with prednisone alone, with steroids plus methotrexate, and with combined immunosuppression including methotrexate and azathioprine, but with no improvement. Eventually, another muscle biopsy revealed inclusion bodies with rimmed vacuoles, consistent with inclusion body myositis.
Inclusion body myositis
Inclusion body myositis is the most common myopathy in middle-aged to elderly people, especially men. These patients are often told “You are just getting old,” but they have a defined condition. It should also be considered in patients failing to respond to treatment or with those with “refractory” polymyositis.
The onset of muscle weakness is insidious and painless, and the weakness progresses slowly. The pattern is distal and asymmetric (eg, foot drop), and muscle atrophy typically affects the forearm flexors, quadriceps, and intrinsic muscles of the hands.1
Magnetic resonance imaging may show marked muscle atrophy. Unfortunately, no treatment has shown efficacy, and most neuromuscular and rheumatology experts do not treat inclusion body myositis with immunosuppressive drugs.
CASE 2: MILD MYALGIA WITHOUT WEAKNESS
A black 52-year-old man was referred because of myalgia and a CK level of 862 U/L (reference range < 200). His physician wanted to start him on a statin but was hesitant to do so without first consulting a rheumatologist.
The patient had a long history of mild arthralgias and myalgias without muscle weakness. He had dyslipidemia and hypertension. He reported no family history of myopathy and no illicit drug use. He was formerly an athlete. Medications included a thiazide diuretic and a beta-blocker. On examination, his muscles were strong (rated 5 on a scale of 5) in the upper and lower extremities, without atrophy.
His records showed that his CK levels had risen and fallen repeatedly over the past few years, ranging from 600 to 1,100 U/L. On further questioning, he reported that when he had joined the army 30 years previously, a physician had recommended he undergo a liver biopsy in view of elevated liver function tests, but that he had refused because he felt fine.
Currently, his gamma-glutamyl transpeptidase levels were normal.
Idiopathic ‘hyperCKemia’
So-called idiopathic hyperCKemia is not a form of myositis but merely a laboratory result outside the “normal” range. Reference ranges are based predominantly on measurements in white people and on an assumption that the distribution is Gaussian (bell-shaped). A normal CK level is usually defined as less than 200 U/L. Using this standard, up to 20% of men and 5% of women have hyperCKemia.2
However, CK levels vary by sex and ethnicity, with mean levels highest in black men, followed by black women, white men, and white women. The mean level in black men is higher than the standard cutoff point for normal, and especially in this population, there is wide fluctuation around the mean, leading to hyperCKemia quite frequently in black men. Exercise and manual labor also drive up CK levels.3–5
Idiopathic hyperCKemia is benign. D’Adda et al6 followed 55 patients for a mean of 7.5 years. CK levels normalized in 12 patients or at least decreased in 24. Most remained symptom-free or had minimal symptoms.
Idiopathic hyperCKemia: Bottom line
Before prescribing a statin, determine the baseline CK level. If slightly elevated (ie, up to 3 to 5 times the upper limit of normal, or even higher) in the setting of normal muscle strength, there is no need for electromyography or muscle biopsy, and the patient can certainly receive a statin. Most of these patients do not need to see a rheumatologist but can simply have their CK and muscle strength monitored.
CLASSIFYING MYOSITIS
Myositis (idiopathic inflammatory myopathy) is a heterogeneous group of autoimmune syndromes of unknown cause characterized by chronic muscle weakness and inflammation of striated muscle. These syndromes likely arise as a result of genetic predisposition and an environmental or infectious “hit.”
Myositis is rare, with an incidence of 5 to 10 cases per million per year and an estimated prevalence of 50 to 90 cases per million. It has 2 incidence peaks: 1 in childhood (age 5–15) and another in adult midlife (age 30–50). Women are affected 2 to 3 times more often than men, with black women most commonly affected.
Myositis is traditionally classified as follows:
- Adult polymyositis
- Adult dermatomyositis
- Juvenile myositis (dermatomyositis much more frequent than polymyositis)
- Malignancy-associated myositis (usually dermatomyositis)
- Myositis overlapping with another autoimmune disease
- Inclusion body myositis.
However, polymyositis is less common than we originally thought, and the term necrotizing myopathy is now used in many patients, as noted in the case studies below. Further, myositis overlap syndromes are being increasingly diagnosed, likely related to the emergence of autoantibodies and clinical “syndromes” associated with these autoantibody subsets (discussed in cases below).
Dermatomyositis
Dermatomyositis is characterized by muscle weakness and a rash that can be obvious or subtle. Classic skin lesions are Gottron papules, which are raised, flat-topped red or purplish lesions over the knuckles, elbows, or knees.
Lesions may be confused with those of psoriasis. There can also be a V-neck rash over the anterior chest or upper back (“shawl sign”) or a rash over the lateral thigh (“holster sign”). A facial rash may occur, but unlike lupus, dermatomyositis does not spare the nasolabial area. However, the V-neck rash can be similar to that seen in lupus.
Dermatomyositis may cause muscle pain, perhaps related to muscle ischemia, whereas polymyositis and necrotizing myopathy are often painless. However, pain is also associated with fibromyalgia, which may be seen in many autoimmune conditions. It is important not to overtreat rheumatologic diseases with immunosuppression to try to control pain if the pain is actually caused by fibromyalgia.
Polymyositis mimics
Hypothyroid myopathy can present as classic polymyositis. The serum CK may be elevated, and there may be myalgias, muscle hypertrophy with stiffness, weakness, cramps, and even features of a proximal myopathy, and rhabdomyolysis. The electromyogram can be normal or myopathic. Results of muscle biopsy are often normal but may show focal necrosis and mild inflammatory infiltrates, thus mimicking that seen with inflammatory myopathy.7
Drug-induced or toxic myopathies can also mimic polymyositis. Statins are among the most commonly prescribed drugs in the United States, with more than 35 million people taking them. Statins are generally well tolerated but have a broad spectrum of toxicity, ranging from myalgias to life-threatening rhabdomyolysis. Myalgias lead to about 5% to 10% of patients refusing to take a statin or stopping it on their own.
Myalgias affect up to 20% of statin users in clinical practice.8,9 A small cross-sectional study10 of 1,000 patients in a primary care setting found that the risk of muscle complaints in statin users was 1.5 times higher than in nonstatin users, similar to findings in other studies.
My strategy for managing a patient with possible statin-induced myopathy is illustrated in Figure 1.
CASE 3: WEAKNESS, VERY HIGH CK ON A STATIN
In March 2010, a 67-year-old woman presented with muscle weakness. She had a history of hypertension, hyperlipidemia, and, more than 10 years previously, uterine cancer. In 2004, she was given atorvastatin for dyslipidemia. Four years later, she developed lower-extremity weakness, which her doctor attributed to normal aging. A year after that, she found it difficult to walk up steps and lift her arms overhead. In June 2009, she stopped taking the atorvastatin on her own, but the weakness did not improve.
In September 2009, she returned to her doctor, who found her CK level was 6,473 U/L but believed it to be an error, so the test was repeated, with a result of 9,375 U/L. She had no rash or joint involvement.
She was admitted to the hospital and underwent muscle biopsy, which showed myonecrosis with no inflammation or vasculitis. She was treated with prednisone 60 mg/day, and her elevated CK level and weakness improved.
Immune-mediated necrotizing myopathy associated with statins
The hallmark of necrotizing myopathy is myonecrosis without significant inflammation.12 This pattern contrasts with that of polymyositis, which is characterized by lymphocytic inflammation.
Although statins became available in the United States in 1987, immune-mediated necrotizing myopathy associated with statins was first described only in 2010. In that report, Grable-Esposito et al13 described 25 patients from 2 neuromuscular centers seen between 2000 and 2008 who had elevated CK and proximal weakness during or after statin use, both of which persisted despite stopping the statin. Patients improved with immunosuppressive agents but had a relapse when steroids were stopped or tapered, a pattern typical in autoimmune disease.
Autoantibody defines subgroup of necrotizing myopathy
Also in 2010, Christopher-Stine et al14 reported an antibody associated with necrotizing myopathy. Of 38 patients with the condition, 16 were found to have an abnormal “doublet” autoantibody recognizing 200- and 100-kDa proteins. All patients had weakness and a high CK level, and 63% had statin exposure before the weakness (this percentage increased to 83% in patients older than 50). All responded to immunosuppressive therapy, and many had a relapse when it was withdrawn.
Statins lower cholesterol by inhibiting 3-hydroxy-3-methylglutaryl-Co A reductase (HMGCR), and paradoxically, they also upregulate it. HMGCR has a molecular weight of 97 kDa. Mammen et al15 identified HMGCR as the 100-kDa target of the identified antibody and developed an enzyme-linked immunosorbent assay for it. Of 750 patients presenting to one center, only 45 (6%) had anti-HMGCR autoantibodies, but all 16 patients who had the abnormal doublet antibody tested positive for anti-HMGCR. Regenerating muscle cells express high levels of HMGCR, which may sustain the immune response after statins are discontinued.
Case 3 continued: Intravenous immunoglobulin brings improvement
In March 2010, when the 67-year-old patient presented to our myositis center, her CK level was 5,800 U/L, which increased as prednisone was tapered. She still felt weak. On examination, her muscle strength findings were deltoids 4+/5, neck flexors 4/5, and iliopsoas 3+/5. She was treated with methotrexate and azathioprine without benefit. She was next treated with intravenous immunoglobulin, and after 3 months, her strength normalized for the first time in years. Her CK level decreased but did not normalize. Testing showed that she was positive for anti-HMGCR autoantibody, as this test had become commercially available.
In 2015, Mammen and Tiniakou16 suggested using intravenous immunoglobulin as first-line therapy for statin-associated autoimmune necrotizing myopathy, based on experience at a single center with 3 patients who declined glucocorticoid treatment.
Necrotizing myopathy: Bottom line
Myositis overlap syndromes
Heterogeneity is the rule in myositis, and it can present with a wide variety of signs and symptoms as outlined in Table 2.
CASE 4: FEVER, NEW ‘RHEUMATOID ARTHRITIS,’ AND LUNG DISEASE
A 52-year-old woman with knee osteoarthritis saw her primary care physician in November 2013 for dyspnea and low-grade fever. The next month, she presented with polyarthritis, muscle weakness, and Raynaud phenomenon.
In January 2014, she developed acrocyanosis of her fingers. Examination revealed hyperkeratotic, cracked areas of her fingers. Her oxygen saturation by pulse oximetry was low. She was admitted to the hospital. Her doctor suspected new onset of rheumatoid arthritis, but blood tests revealed a negative antinuclear antibody, so an autoimmune condition was deemed unlikely. Her CK was mildly elevated at 350 U/L.
Because of her dyspnea, an open-lung biopsy was performed. High-resolution computed tomography (CT) revealed infiltrates and ground-glass opacities, leading to the diagnosis of nonspecific interstitial pneumonia. A rheumatologist was consulted and recommended pulse methylprednisolone, followed by prednisone 60 mg/day and mycophenolate mofetil. Testing for Jo-1 antibodies was positive.
Antisynthetase syndrome
The antisynthetase syndrome is a clinically heterogeneous condition that can occur with any or all of the following:
- Fever
- Myositis
- Arthritis (often misdiagnosed as rheumatoid arthritis)
- Raynaud phenomenon
- Mechanic’s hands (hyperkeratotic roughness with fissures on the lateral aspects of the fingers and finger pads)
- Interstitial lung disease.
The skin rashes and myositis may be subtle, making the presentation “lung-dominant,” and nonrheumatologists should be aware of this syndrome. Although in our patient the condition developed in a classic manner, with all of the aforementioned features of the antisynthetase syndrome, some patients will manifest one or a few of the features.
Clinically, patients with the Jo-1 antisynthetase syndrome often present differently than those with non-Jo-1 antisynthetase autoantibodies. When we compared 122 patients with Jo-1 vs 80 patients with a non-Jo-1 antisynthetase autoantibody, patients with Jo-1 antibodies were more likely to have initially received a diagnosis of myositis (83%), while myositis was the original diagnosis in only 17% of those possessing non-Jo-1 antisynthetase autoantibodies. In fact, many patients (approximately 50%) were diagnosed as having undifferentiated connective tissue disease or an overlap syndrome, and 13% had scleroderma as their first diagnosis.17
We also found that the survival rate was higher in patients with Jo-1 syndrome compared with patients with non-Jo-1 antisynthetase syndromes. We attributed the difference in survival rates to a delayed diagnosis in the non-Jo-1 group, perhaps due to their “nonclassic” presentations of the antisynthetase syndrome, delaying appropriate treatment. Patients received a diagnosis of Jo-1 antibody syndrome after a mean of 0.4 year (range 0.2–0.8), while those with a non-Jo-1 antisynthetase autoantibody had a delay in diagnosis of 1.0 year (range 0.4–5.1) (P < .01).17
In nearly half the cases in this cohort, pulmonary fibrosis was the cause of death, with primary pulmonary hypertension being the second leading cause (11%).
Antisynthetase syndrome: Bottom line
Antisynthetase syndrome is an often fatal disease that does not always present in a typical fashion with symptoms of myositis, as lung disease may be the predominant feature. A negative antinuclear antibody test result does not imply antibody negativity, as the autoantigen in these diseases is not located in the nucleus. Prompt diagnosis and appropriate immunosuppressive therapy are critical to improving outcomes.
CASE 5: FEVER, UNDIAGNOSED LUNG DISEASE, NO MYOSITIS
In January 2001, a 39-year-old woman was admitted to the hospital after 5 weeks of fever (temperatures 103°–104°F) and myalgias. An extensive workup was negative except for low-titer antinuclear antibody and for mild basilar fibrosis noted on chest radiography. She left the hospital against medical advice because of frustration with a lack of a specific diagnosis (“fever of unknown origin”).
Two months later, at a follow-up rheumatology consult, she reported more myalgias and arthralgias, as well as fever. Chest radiography now showed pleural effusions. Her fingers had color changes consistent with Raynaud phenomenon. At that time, I diagnosed an undifferentiated connective tissue disease and told her that I suspected an autoimmune condition that would need time to reveal itself. In the meantime, I treated her empirically with prednisone.
In April, she returned, much more short of breath and with more prominent diffuse pulmonary infiltrates. Physical examination revealed subtle Gottron changes. Testing revealed poor pulmonary function: forced vital capacity (FVC) 56%, forced expiratory volume in 1 second (FEV1) 52%, and diffusing capacity for carbon monoxide (Dlco) 40%. Blood testing was positive for anti-PL-12 antibody, one of the non-Jo-1 antisynthetase antibodies. At this time, we treated her with glucocorticoids and tacrolimus.
More than 15 years later, this patient is doing well. Her skin rash, joint symptoms, and fever have not returned, and interestingly, she never developed myositis. Her Raynaud symptoms are mild. Her most recent pulmonary function test results (January 2018) were FVC 75%, FEV1 87%, and Dlco 78%. Although these results are not normal, they are much improved and allow her to be completely functional without supplemental oxygen. Echocardiography showed normal pulmonary artery systolic pressure (25 mm Hg). She was still taking tacrolimus and prednisone. When we tried to stop tacrolimus after she had done well for many years, her condition flared.
Non-Jo-1 antisynthetase syndrome: Bottom line
Patients with a non-Jo-1 antisynthetase syndrome often present without myositis symptoms and may never manifest myositis symptoms. Likely because of this presentation, diagnosis of a specific connective tissue disorder is delayed, perhaps leading to increased mortality risk from pulmonary disease. Chronic immunosuppression is often required for these autoimmune conditions.
CASE 6: DERMATOMYOSITIS, RAPIDLY PROGRESSIVE INTERSTITIAL LUNG DISEASE
A 58-year-old woman presented in the summer of 2012 with a photosensitive rash. The following January, she returned with polyarthritis, mild muscle weakness, and a dermatomyositis-pattern rash. Her CK level was normal, and her antinuclear antibody and Sjögren syndrome antibody test results were negative. She improved on low-dose prednisone and methotrexate.
She was originally referred to me in May of that year for worsening rash and mild weakness. She denied pulmonary symptoms, but examination revealed faint basilar crackles. I increased her prednisone dosage to 20 mg/day and started mycophenolate mofetil mainly for the mild cutaneous and myositis features. I also recommended high-resolution CT of the lungs and pulmonary function tests, which she underwent in early June. High-resolution CT showed nonspecific mild infiltrates with minimal ground-glass opacities.
On July 1, she presented to her local emergency department with severe shortness of breath, requiring oxygen 12 L/min. She had a palmar rash. Repeat high-resolution CT showed dramatic worsening compared with the scan the previous month. Because of continued inadequate oxygenation, she was transferred to our center. A blood test later was positive for antimelanoma differentiation-associated gene 5 (MDA-5) autoantibody, previously known as anticlinically amyopathic dermatomyositis (anti-CADM)-140 antibody (based on immunoprecipitation results).
She died on the third day after transfer, just 2 months after I had originally seen her, at which time she had had no pulmonary symptoms.
Clinically amyopathic dermatomyositis
Anti-CADM-140, first reported from Asia,18–20 is an autoantibody-associated disease but not an antisynthetase. It is associated with dermatomyositis; patients often have a “vasculopathy” with cutaneous ulcerations and palmar papules.
MDA-5 is a cytoplasmic protein that “senses” viral RNA and induces production of type 1 interferon. It is involved in the innate immune defense against viruses.
Anti-MDA-5 positivity is associated with a poor pulmonary outcome.21 In our cohort from the University of Pittsburgh, many patients died within 3 years, compared with about a 40% survival rate in patients with dermatomyositis who tested negative for this antibody. That being said, many patients with anti-MDA-5 do not develop rapidly progressive interstitial lung disease.
Autoimmune interstitial lung disease: Bottom line
Autoimmune interstitial lung disease is easy to miss, especially in the case of a non-Jo-1 syndrome, for 3 important reasons:
- The autoimmune features may initially be subtle (eg, Raynaud phenomena, mild dermatomyositis rash, undifferentiated connective tissue disease)
- Autoantibody testing is not often ordered, is not standardized, or may be unavailable
- Providers are mistakenly reassured that a patient who tests negative for antinuclear antibody does not have an autoimmune condition.
To emphasize the last point, in a cohort of 202 patients who tested positive for an antisynthetase antibody, only half were antinuclear antibody-positive, but nearly three-quarters demonstrated anticytoplasmic staining on indirect immunofluorescence (due to the location of the autoantigen in the cytoplasm), making the latter a better screening test for an antisynthetase antibody. For scleroderma, 99% were antinculear antibody-positive, but for myositis, this test is much less sensitive.22
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
- Felice KJ, North WA. Inclusion body myositis in Connecticut: observations in 35 patients during an 8-year period. Medicine (Baltimore) 2001; 80(5):320–327. doi:10.1097/00005792-200109000-00006
- Lev EI, Tur-Kaspa I, Ashkenazy I, et al. Distribution of serum creatine kinase activity in young healthy persons. Clin Chim Acta 1999; 279(1-2):107–115. doi:10.1016/S0009-8981(98)00180-6
- Lilleng H, Abeler K, Johnsen SH, et al. Variation of serum creatine kinase (CK) levels and prevalence of persistent hyperCKemia in a Norwegian normal population. The Tromsø Study. Neuromuscul Disord 2011; 21(7):494–500. doi:10.1016/j.nmd.2011.04.007
- Johnston JD, Lloyd M, Mathews JA, Hawthorne SW. Racial variation in serum creatine kinase levels. J R Soc Med 1996; 89(8):462-464. pmid:8795501
- Prelle A, Tancredi L, Sciacco M, et al. Retrospective study of a large population of patients with asymptomatic or minimally symptomatic raised serum creatine kinase levels. J Neurol 2002; 249(3):305–311. pmid:11993531
- D’Adda E, Sciacco M, Fruguglietti ME, et al. Follow-up of a large population of asymptomatic/oligosymptomatic hyperckemic subjects. J Neurol 2006; 253(11):1399–1403. doi:10.1007/s00415-006-0223-y
- Madariaga MG. Polymyositis-like syndrome in hypothyroidism: review of cases reported over the past twenty-five years. Thyroid 2002; 12(4):331–336. doi:10.1089/10507250252949478
- de Sauvage Nolting PR, Buirma RJ, Hutten BA, Kastelein JJ; Dutch ExPRESS Investigator Group. Two-year efficacy and safety of simvastatin 80 mg in familial hypercholesterolemia (the Examination of Probands and Relatives in Statin Studies With Familial Hypercholesterolemia [ExPRESS FH]). Am J Cardiol 2002; 90(2):181–184. doi:10.1016/s0002-9149(02)02449-9
- Bruckert E, Hayem G, Dejager S, Yau C, Bégaud B. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005; 19(6):403–414. doi:10.1007/s10557-005-5686-z
- Mosshammer D, Lorenz G, Meznaric S, Schwarz J, Muche R, Mörike K. Statin use and its association with musculoskeletal symptoms—a cross-sectional study in primary care settings. Fam Pract 2009; 26(2):88–95. doi:10.1093/fampra/cmp006
- Nichols GA, Koro CE. Does statin therapy initiation increase the risk for myopathy? An observational study of 32,225 diabetic and nondiabetic patients. Clin Ther 2007; 29(8):1761–1770. doi:10.1016/j.clinthera.2007.08.022
- Kassardjian CD, Lennon VA, Alfugham NB, Mahler M, Milone M. Clinical features and treatment outcomes of necrotizing autoimmune myopathy. JAMA Neurol 2015; 72(9):996–1003. doi:10.1001/jamaneurol.2015.1207
- Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve 2010; 41(2):185–190. doi:10.1002/mus.21486
- Christopher-Stine L, Casciola-Rosen LA, Hong G, Chung T, Corse AM, Mammen AL. A novel autoantibody recognizing 200-kd and 100-kd proteins is associated with an immune-mediated necrotizing myopathy. Arthritis Rheum 2010; 62(9):2757–2766. doi:10.1002/art.27572
- Mammen AL, Chung T, Christopher-Stine L, et al. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum 2011; 63(3):713–721. doi:10.1002/art.30156
- Mammen AL, Tiniakou E. Intravenous immune globulin for statin-triggered autoimmune myopathy. N Engl J Med 2015; 373(17):1680–1682. doi:10.1056/NEJMc1506163
- Aggarwal R, Cassidy E, Fertig N, et al. Patients with non-Jo-1 anti-tRNA-synthetase autoantibodies have worse survival than Jo-1 positive patients. Ann Rheum Dis 2014; 73(1):227–232. doi:10.1136/annrheumdis-2012-201800
- Sato S, Hirakata M, Kuwana M, et al. Autoantibodies to a 140-kd polypeptide, CADM-140, in Japanese patients with clinically amyopathic dermatomyositis. Arthritis Rheum 2005; 52(5):1571–1576. doi:10.1002/art.21023
- Sato S, Hoshino K, Satoh T, et al. RNA helicase encoded by melanoma differentiation-associated gene 5 is a major autoantigen in patients with clinically amyopathic dermatomyositis: association with rapidly progressive interstitial lung disease. Arthritis Rheum 2009; 60(7):2193–2200. doi:10.1002/art.24621
- Chen F, Wang D, Shu X, Nakashima R, Wang G. Anti-MDA5 antibody is associated with A/SIP and decreased T cells in peripheral blood and predicts poor prognosis of ILD in Chinese patients with dermatomyositis. Rheumatol Int 2012; 32(12):3909–3915. doi:10.1007/s00296-011-2323-y
- Moghadam-Kia S, Oddis CV, Sato S, Kuwana M, Aggarwal R. Anti-melanoma differentiation-associated gene 5 is associated with rapidly progressive lung disease and poor survival in US patients with amyopathic and myopathic dermatomyositis. Arthritis Care Res (Hoboken) 2016; 68(5):689–694. doi:10.1002/acr.22728
- Aggarwal R, Dhillon N, Fertig N, Koontz D, Qi Z, Oddis CV. A negative antinuclear antibody does not indicate autoantibody negativity in myositis: role of anticytoplasmic antibody as a screening test for antisynthetase syndrome. J Rheumatol 2017; 44(2):223–229. doi:10.3899/jrheum.160618
KEY POINTS
- Inclusion body myositis affects older men more than women and is characterized by slowly progressive, asymmetric, distal and proximal weakness and atrophy.
- Statin-associated muscle complaints are common, whereas necrotizing myopathy, characterized by a very high CK plus weakness, is rare but must be recognized.
- Elevated CK does not necessarily indicate myositis, especially in African Americans or after heavy exercise.
- Dermatomyositis is characterized by muscle weakness and raised red or purple Gottron papules over the knuckles, elbows, or knees.
- Autoimmune interstitial lung disease may be caused by a variety of antibodies, the most common being anti-Jo-1 (directed against histidyl tRNA synthetase).
- The rarer non-Jo-1 antisynthetase autoantibodies may be associated with rapidly progressive interstitial lung disease, which is a challenge to recognize because associated rheumatologic symptoms may be minimal.
Progress in diagnosing and managing cardiac amyloidosis
From the Cleveland Clinic Journal of Medicine
This article has been removed from the website. The article was prepared by the editorial staff based on a transcript of the proceedings of a conference, and errors occurred during this process that were subsequently published. A clarification of the errors will be published in a future issue.
A review of this topic was published in the December 2017 issue of the Journal (Donnelly JP, Hanna M. Cardiac amyloidosis: An update on diagnosis and treatment. Cleve Clin J Med 2017;84[12suppl 3]:12–26). doi:10.3949/ccjm.84.s3.02
From the Cleveland Clinic Journal of Medicine
This article has been removed from the website. The article was prepared by the editorial staff based on a transcript of the proceedings of a conference, and errors occurred during this process that were subsequently published. A clarification of the errors will be published in a future issue.
A review of this topic was published in the December 2017 issue of the Journal (Donnelly JP, Hanna M. Cardiac amyloidosis: An update on diagnosis and treatment. Cleve Clin J Med 2017;84[12suppl 3]:12–26). doi:10.3949/ccjm.84.s3.02
From the Cleveland Clinic Journal of Medicine
This article has been removed from the website. The article was prepared by the editorial staff based on a transcript of the proceedings of a conference, and errors occurred during this process that were subsequently published. A clarification of the errors will be published in a future issue.
A review of this topic was published in the December 2017 issue of the Journal (Donnelly JP, Hanna M. Cardiac amyloidosis: An update on diagnosis and treatment. Cleve Clin J Med 2017;84[12suppl 3]:12–26). doi:10.3949/ccjm.84.s3.02
Our missing microbes: Short-term antibiotic courses have long-term consequences
Recent years have seen dramatic increases in the prevalences of chronic diseases such as type 1 diabetes,1 gastroesophageal reflux disease,2 asthma,3 inflammatory bowel disease,4 and, notably, obesity.5 I propose the hypothesis that much of this increase may be due to loss of diversity in the bacteria that make our guts their home.6 While multiple causes contribute, much of the blame may be attributed to the use—and overuse—of antibiotics.
FAT AND GETTING FATTER
Today, nearly 40% of US adults are obese, and nearly three-fourths are either obese or overweight.7 More alarming, the prevalence of obesity is also high and getting higher in children and adolescents,8 having increased from 10.0% in 1988–1994 to 17.8% in 2013–2016.
And not just in the United States. Trends in weight have been going up around the world, with a lag of about 30 years between developing countries and industrialized countries.5
OUR BACTERIA, OURSELVES
I believe that the bacteria we carry are not random, but rather have coevolved along with us, passed down from generation to generation in a state of dynamic equilibrium between microbes and host. Evidence supporting this comes from a study by Ochman et al,9 who analyzed the DNA from fecal samples from different hominid species (including Homo sapiens) and found that the phylogenic relationships among the bacteria mirrored those among the apes.
Interacting with each other and with us in complex ways, our bacteria are a diverse community to which we can apply the term microbiome. They are acquired in a standard, choreographed process,10 and their composition comes to resemble that of adults by the age of 3.11
Before modern times, microbes were transferred from mother to child during vaginal birth, from the mother’s breast during nursing, through skin-to-skin contact, and from the mother’s mouth by kissing. Now, widespread cesarean delivery, bottle-feeding, extensive bathing (especially with antibacterial soaps), and especially the use of antibiotics have changed the human ecology and altered transmission and maintenance of ancestral microbes, which affects the composition of the microbiota. The microbes, both good and bad, that are usually acquired early in life are especially important, since they affect a developmentally critical stage.12
Loss of microbial diversity in the mother appears to be cumulative over succeeding generations.13 For example, in a study in Japanese families, Urita et al14 found a decline in the prevalence of Helicobacter pylori colonization from 68.7% in the first generation to 43.4% in the second generation and 12.5% in the third. Clemente et al15 studied the intestinal microbiota in a previously uncontacted group of Yanomami people in the Amazon jungle and found they had the highest diversity of bacteria ever reported in a human group. By comparison, the research team calculated that we in the United States have already lost 50% of our microbial diversity, and 2 other groups, the Guahibo (another Amerindian group) and rural Malawians, were in between. More recent studies are confirming these observations.16,17
USE AND OVERUSE OF ANTIBIOTICS
More than 73 billion antibiotic doses are prescribed worldwide yearly,18 or about 10 doses for every man, woman, and child on Earth, and the numbers are rising. In the United States 262 million courses were prescribed in 2011, or 842 per 1,000 population.19 Children receive a mean of 2.7 courses by age 2, and 10.9 by age 10. More than 50% of women receive antibiotics during pregnancy or perinatally. This is in addition to an unknown level of exposure from agricultural use of antibiotics.
Repeated antibiotic exposure is common in early life, varies widely by country, and is often not medically justified.20 In the United States, antibiotic use varies by region, with the heaviest use in the South.19,21 It also varies widely among prescribers.22 Jones et al23 examined antibiotic prescribing for acute respiratory infections in US veterans and found that the top 10% of physicians gave an antibiotic more than 90% of the time. Physicians in Sweden prescribe about 60% fewer antibiotics than we do in the United States.21,24
Observational data indicate that people who receive antibiotics have a higher risk of chronic diseases later in life, eg:
- Type 2 diabetes (odds ratio 1.21, 95% confidence interval 1.19–1.23 with 2 to 4 courses, and odds ratio 1.53 (1.50–1.55) with 5 or more courses, up to 15 years after25
- Obesity: US states with the highest prevalence of antibiotic use also have the highest prevalence of obesity26
- Kidney stones: prior antibiotic exposure in a large UK study was associated with increased kidney stone risk, for exposures up to 5 years earlier.27
The meat industry has exploited the weight effect for decades, adding subtherapeutic doses of antibiotics to animals’ feed to make them gain weight.28
FINDINGS FROM STUDIES IN MICE
Laboratory studies of the relationship between antibiotic exposure and disease phenotypes in mice have yielded interesting findings.
Mice exposed to antibiotics had more body fat at 10 weeks (32.0%) than control mice (22.9%).29
Low-dose penicillin, started at birth, induces long-lasting effects on the expression of genes involved in immunity and enhances the effect of a high-fat diet in terms of weight gain.30 If the antibiotic exposure is limited to early life, the effect on the microbiota is transient, but the mice still gain weight. If the microbiota from the mice who received penicillin is transferred to germ-free mice, the recipients also become fat, indicating that the bacteria, not the antibiotics per se, cause the weight gain.
In other experiments,31 a series of short, therapeutic doses of antibiotics early in life modeled after those given to children to treat their acute infections caused long-term changes in the composition of the microbiome and in metabolism.
A single course of a macrolide antibiotic also had long-term effects on the microbial population and on the host’s ileal gene expression, T-cell populations, and secretory immunoglobulin A expression.32 These effects were seen only in mice that had a microbiome to begin with, not in germ-free mice, indicating that the antibiotics had their effect through the changes in the microbiome, not directly. But when germ-free mice received a fecal transplant of an impaired microbiome, it was sufficient to affect immunity.
In nonobese diabetic mice, treatment with antibiotics early in life altered the gut microbiome and its metabolic capacities, intestinal gene expression, and T-cell populations, accelerating the onset of type 1 diabetes.33
In a study in Danish children,34 the likelihood of inflammatory bowel disease increased with early-life antibiotic exposure: the more courses the child received, the greater the likelihood of disease. This observation led researchers to wonder if an antibiotic-altered microbiome affects the outcome of inflammatory bowel disease in the next generation.35 Germ-free female mice who received microbiota from mice who had received antibiotics passed the altered microbiome to their pups. Mice lacking the gene for interleukin 10 are genetically susceptible to colitis, and when this experiment was done in mice lacking this gene, the offspring developed markedly more colitis. This indicated the mothers could pass down their altered microbiome to the next generation and that it would affect their risk of disease.
WHAT CAN WE DO?
All physicians must adhere to the principles of antibiotic stewardship,36 not only to prevent the development of resistant strains of pathogens and the overgrowth of potentially dangerous species such as Clostridium difficile, but also, possibly, to prevent the loss of diversity in the human microbiome and thus discourage the development of chronic diseases.
In the future, as we discover more about the microbiome and the optimal mix of bacteria to carry, this information may find practical application in medicine. A pediatrician, for example, may want to analyze a child’s microbiome and, if it is abnormal, administer specific organisms to reshape it.
- TEDDY Study Group. The Environmental Determinants of Diabetes in the Young (TEDDY) study. Ann NY Acad Sci 2008; 1150:1–13. doi:10.1196/annals.1447.062
- El-Serag HB, Sonnenberg A. Associations between different forms of gastro-oesophageal reflux disease. Gut 1997; 41(5):594–599. pmid:9414963
- Eder W, Ege MJ, von Mutius E. The asthma epidemic. N Engl J Med 2006; 355(21):2226–2235. doi:10.1056/NEJMra054308
- Kaplan GG, Ng SC. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology 2017; 152(2):313–321. doi:10.1053/j.gastro.2016.10.020
- de Onis M, Blossner M, Borghi E. Global prevalence and trends of overweight and obesity among preschool children. Am J Clin Nutr 2010; 92(5):1257–1264. doi:10.3945/ajcn.2010.29786
- Blaser MJ. The theory of disappearing microbiota and the epidemics of chronic disease. Nat Rev Immunol 2017; 17(8):461–463. doi:10.1038/nri.2017.77
- Centers for Disease Control and Prevention. National Center for Health Statistics. Obesity and overweight. www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed November 6, 2018.
- Centers for Disease Control and Prevention. National Center for Health Statistics. Table 59. Obesity among children and adolescents aged 2-19 years, by selected characteristics: United States, selected years 1988–1994 through 2013–2016. www.cdc.gov/nchs/data/hus/2017/059.pdf. Accessed November 6, 2018.
- Ochman H, Worobey M, Kuo CH, et al. Evolutionary relationships of wild hominids recapitulated by gut microbial communities. PLoS Biology 2010; 8(11):e1000546. doi:10.1371/journal.pbio.1000546
- Bokulich NA, Chung J, Battaglia T, et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci Trans Med 2016; 8(343):343ra82. doi:10.1126/scitranslmed.aad7121
- Yatsunenko T, Rey FE, Manary MJ, et al. Human gut microbiome viewed across age and geography. Nature 2012; 486(7402):222–227. doi:10.1038/nature11053
- Blaser MJ. The past and future biology of the human microbiome in an age of extinctions. Cell 2018; 172(6):1173–1177. doi:10.1016/j.cell.2018.02.040
- Blaser MJ, Falkow S. What are the consequences of the disappearing human microbiota? Nat Rev Microbiol 2009; 7(12):887–894. doi:10.1038/nrmicro2245
- Urita Y, Watanabe T, Kawagoe N, et al. Role of infected grandmothers in transmission of Helicobacter pylori to children in a Japanese rural town. J Ped Child Health 2013; 49(5):394–398. doi:10.1111/jpc.12191
- Clemente JC, Pehrsson EC, Blaser MJ, et al. The microbiome of uncontacted Amerindians. Sci Adv 2015; 1(3). Pii:e1500183. doi:10.1126/sciadv.1500183
- Smits SA, Leach J, Sonnenburg ED, et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 2017; 357(6353):802-806. doi:10.1126/science.aan4834
- Vangay P, Johnson AJ, Ward TL, et al. US immigration westernizes the human gut microbiome. Cell 2018; 175(4):962–972. doi:10.1016/j.cell.2018.10.029
- Van Broeckel TP, Gandra S, Ashok A, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis 2014; 14(8):742–750. doi:10.1016/S1473-3099(14)70780-7
- Hicks LA, Bartoces MG, Roberts RM, et al. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis 2015; 60(9):1308–1316. doi:10.1093/cid/civ076
- Rogawski ET, Platts-Mills JA, Seidman JC, et al. Use of antibiotics in children younger than two years in eight countries: a prospective cohort study. Bull World Health Organ 2017; 95(1):49–61. doi:10.2471/BLT.16.176123
- Hicks LA, Taylor TH Jr, Hunkler RJ. U.S. outpatient antibiotic prescribing, 2010; N Engl J Med 2013; 368(15):1461–1462. doi:10.1056/NEJMc1212055
- Gerber JS, Prasad PA, Russell LA, et al. Variation in antibiotic prescribing across a pediatric primary care network. J Pediatric Infect Dis Soc 2015; 4(4):297–304. doi:10.1093/jpids/piu086
- Jones BE, Sauer B, Jones MM, et al. Variation in outpatient antibiotic prescribing for acute respiratory infections in the veteran population: a cross-sectional study. Ann Intern Med 2015; 163(2):73–80. doi:10.7326/M14-1933
- Ternhag A, Hellman J. More on U.S. outpatient antibiotic prescribing, 2010. N Engl J Med 2013; 369(12):1175. doi:10.1056/NEJMc1306863
- Mikkelsen KH, Knop FK, Frost M, Hallas J, Pottegard A. Use of antibiotics and risk of type 2 diabetes: a population-based case-control study. J Clin Endocrinol Metab 2015; 100(10):3633–3640. doi:10.1210/jc.2015-2696
- Petschow B, Dore J, Hibbert P, et al. Probiotics, prebiotics, and the host microbiome: the science of translation. Ann NY Acad Sci 2013; 1306:1–17. doi:10.1111/nyas.12303
- Tasian GE, Jemielita T, Goldfarb DS, et al. Oral antibiotic exposure and kidney stone disease. J Am Soc Nephrol 2018; 29(6):1731–1740. doi:10.1681/ASN.2017111213
- Zimmerman DR. Role of subtherapeutic levels of antimicrobials in pig production. J Anim Sci 1986; 62(suppl 3):6–16.
- Cho I, Yamanishi S, Cox L, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012; 488(7413):621–626. doi:10.1038/nature11400
- Cox LM, Yamanishi S, Sohn J, et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 2014; 158(4):705–721. doi:10.1016/j.cell.2014.05.052
- Nobel YR, Cox LM, Kirigin FF, et al. Metabolic and metagenomics outcomes from early-life pulsed antibiotic treatment. Nat Commun 2015; 6:7486. doi:10.1038/ncomms8486
- Ruiz VE, Battaglia T, Kurtz ZD, et al. A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity. Nat Commun 2017; 8(1):518. doi:10.1038/s41467-017-00531-6
- Livanos AE, Greiner TU, Vangay P, et al. Antibiotic-mediated gut microbiome perturbation accelerates development of type 1 diabetes in mice. Nat Microbiol 2016; 1(11):16149. doi:10.1038/nmicrobiol.2016.140
- Hvilid A, Svanström H, Frish M. Antibiotic use and inflammatory bowel disease in childhood. Gut 2011; 60(1):49–54. doi:10.1136/gut.2010.219683
- Schulfer AF, Battaglia T, Alvarez Y, et al. Intergenerational transfer of antibiotic-perturbed microbiota enhances colitis in susceptible mice. Nat Microbiol 2018; 3(2):234–242. doi:10.1038/s41564-017-0075-5
- Srinivasan A. Antibiotic stewardship: why we must, how we can. Cleve Clin J Med 2017; 84(9):673–679. doi:10.3949/ccjm.84gr.17003
Recent years have seen dramatic increases in the prevalences of chronic diseases such as type 1 diabetes,1 gastroesophageal reflux disease,2 asthma,3 inflammatory bowel disease,4 and, notably, obesity.5 I propose the hypothesis that much of this increase may be due to loss of diversity in the bacteria that make our guts their home.6 While multiple causes contribute, much of the blame may be attributed to the use—and overuse—of antibiotics.
FAT AND GETTING FATTER
Today, nearly 40% of US adults are obese, and nearly three-fourths are either obese or overweight.7 More alarming, the prevalence of obesity is also high and getting higher in children and adolescents,8 having increased from 10.0% in 1988–1994 to 17.8% in 2013–2016.
And not just in the United States. Trends in weight have been going up around the world, with a lag of about 30 years between developing countries and industrialized countries.5
OUR BACTERIA, OURSELVES
I believe that the bacteria we carry are not random, but rather have coevolved along with us, passed down from generation to generation in a state of dynamic equilibrium between microbes and host. Evidence supporting this comes from a study by Ochman et al,9 who analyzed the DNA from fecal samples from different hominid species (including Homo sapiens) and found that the phylogenic relationships among the bacteria mirrored those among the apes.
Interacting with each other and with us in complex ways, our bacteria are a diverse community to which we can apply the term microbiome. They are acquired in a standard, choreographed process,10 and their composition comes to resemble that of adults by the age of 3.11
Before modern times, microbes were transferred from mother to child during vaginal birth, from the mother’s breast during nursing, through skin-to-skin contact, and from the mother’s mouth by kissing. Now, widespread cesarean delivery, bottle-feeding, extensive bathing (especially with antibacterial soaps), and especially the use of antibiotics have changed the human ecology and altered transmission and maintenance of ancestral microbes, which affects the composition of the microbiota. The microbes, both good and bad, that are usually acquired early in life are especially important, since they affect a developmentally critical stage.12
Loss of microbial diversity in the mother appears to be cumulative over succeeding generations.13 For example, in a study in Japanese families, Urita et al14 found a decline in the prevalence of Helicobacter pylori colonization from 68.7% in the first generation to 43.4% in the second generation and 12.5% in the third. Clemente et al15 studied the intestinal microbiota in a previously uncontacted group of Yanomami people in the Amazon jungle and found they had the highest diversity of bacteria ever reported in a human group. By comparison, the research team calculated that we in the United States have already lost 50% of our microbial diversity, and 2 other groups, the Guahibo (another Amerindian group) and rural Malawians, were in between. More recent studies are confirming these observations.16,17
USE AND OVERUSE OF ANTIBIOTICS
More than 73 billion antibiotic doses are prescribed worldwide yearly,18 or about 10 doses for every man, woman, and child on Earth, and the numbers are rising. In the United States 262 million courses were prescribed in 2011, or 842 per 1,000 population.19 Children receive a mean of 2.7 courses by age 2, and 10.9 by age 10. More than 50% of women receive antibiotics during pregnancy or perinatally. This is in addition to an unknown level of exposure from agricultural use of antibiotics.
Repeated antibiotic exposure is common in early life, varies widely by country, and is often not medically justified.20 In the United States, antibiotic use varies by region, with the heaviest use in the South.19,21 It also varies widely among prescribers.22 Jones et al23 examined antibiotic prescribing for acute respiratory infections in US veterans and found that the top 10% of physicians gave an antibiotic more than 90% of the time. Physicians in Sweden prescribe about 60% fewer antibiotics than we do in the United States.21,24
Observational data indicate that people who receive antibiotics have a higher risk of chronic diseases later in life, eg:
- Type 2 diabetes (odds ratio 1.21, 95% confidence interval 1.19–1.23 with 2 to 4 courses, and odds ratio 1.53 (1.50–1.55) with 5 or more courses, up to 15 years after25
- Obesity: US states with the highest prevalence of antibiotic use also have the highest prevalence of obesity26
- Kidney stones: prior antibiotic exposure in a large UK study was associated with increased kidney stone risk, for exposures up to 5 years earlier.27
The meat industry has exploited the weight effect for decades, adding subtherapeutic doses of antibiotics to animals’ feed to make them gain weight.28
FINDINGS FROM STUDIES IN MICE
Laboratory studies of the relationship between antibiotic exposure and disease phenotypes in mice have yielded interesting findings.
Mice exposed to antibiotics had more body fat at 10 weeks (32.0%) than control mice (22.9%).29
Low-dose penicillin, started at birth, induces long-lasting effects on the expression of genes involved in immunity and enhances the effect of a high-fat diet in terms of weight gain.30 If the antibiotic exposure is limited to early life, the effect on the microbiota is transient, but the mice still gain weight. If the microbiota from the mice who received penicillin is transferred to germ-free mice, the recipients also become fat, indicating that the bacteria, not the antibiotics per se, cause the weight gain.
In other experiments,31 a series of short, therapeutic doses of antibiotics early in life modeled after those given to children to treat their acute infections caused long-term changes in the composition of the microbiome and in metabolism.
A single course of a macrolide antibiotic also had long-term effects on the microbial population and on the host’s ileal gene expression, T-cell populations, and secretory immunoglobulin A expression.32 These effects were seen only in mice that had a microbiome to begin with, not in germ-free mice, indicating that the antibiotics had their effect through the changes in the microbiome, not directly. But when germ-free mice received a fecal transplant of an impaired microbiome, it was sufficient to affect immunity.
In nonobese diabetic mice, treatment with antibiotics early in life altered the gut microbiome and its metabolic capacities, intestinal gene expression, and T-cell populations, accelerating the onset of type 1 diabetes.33
In a study in Danish children,34 the likelihood of inflammatory bowel disease increased with early-life antibiotic exposure: the more courses the child received, the greater the likelihood of disease. This observation led researchers to wonder if an antibiotic-altered microbiome affects the outcome of inflammatory bowel disease in the next generation.35 Germ-free female mice who received microbiota from mice who had received antibiotics passed the altered microbiome to their pups. Mice lacking the gene for interleukin 10 are genetically susceptible to colitis, and when this experiment was done in mice lacking this gene, the offspring developed markedly more colitis. This indicated the mothers could pass down their altered microbiome to the next generation and that it would affect their risk of disease.
WHAT CAN WE DO?
All physicians must adhere to the principles of antibiotic stewardship,36 not only to prevent the development of resistant strains of pathogens and the overgrowth of potentially dangerous species such as Clostridium difficile, but also, possibly, to prevent the loss of diversity in the human microbiome and thus discourage the development of chronic diseases.
In the future, as we discover more about the microbiome and the optimal mix of bacteria to carry, this information may find practical application in medicine. A pediatrician, for example, may want to analyze a child’s microbiome and, if it is abnormal, administer specific organisms to reshape it.
Recent years have seen dramatic increases in the prevalences of chronic diseases such as type 1 diabetes,1 gastroesophageal reflux disease,2 asthma,3 inflammatory bowel disease,4 and, notably, obesity.5 I propose the hypothesis that much of this increase may be due to loss of diversity in the bacteria that make our guts their home.6 While multiple causes contribute, much of the blame may be attributed to the use—and overuse—of antibiotics.
FAT AND GETTING FATTER
Today, nearly 40% of US adults are obese, and nearly three-fourths are either obese or overweight.7 More alarming, the prevalence of obesity is also high and getting higher in children and adolescents,8 having increased from 10.0% in 1988–1994 to 17.8% in 2013–2016.
And not just in the United States. Trends in weight have been going up around the world, with a lag of about 30 years between developing countries and industrialized countries.5
OUR BACTERIA, OURSELVES
I believe that the bacteria we carry are not random, but rather have coevolved along with us, passed down from generation to generation in a state of dynamic equilibrium between microbes and host. Evidence supporting this comes from a study by Ochman et al,9 who analyzed the DNA from fecal samples from different hominid species (including Homo sapiens) and found that the phylogenic relationships among the bacteria mirrored those among the apes.
Interacting with each other and with us in complex ways, our bacteria are a diverse community to which we can apply the term microbiome. They are acquired in a standard, choreographed process,10 and their composition comes to resemble that of adults by the age of 3.11
Before modern times, microbes were transferred from mother to child during vaginal birth, from the mother’s breast during nursing, through skin-to-skin contact, and from the mother’s mouth by kissing. Now, widespread cesarean delivery, bottle-feeding, extensive bathing (especially with antibacterial soaps), and especially the use of antibiotics have changed the human ecology and altered transmission and maintenance of ancestral microbes, which affects the composition of the microbiota. The microbes, both good and bad, that are usually acquired early in life are especially important, since they affect a developmentally critical stage.12
Loss of microbial diversity in the mother appears to be cumulative over succeeding generations.13 For example, in a study in Japanese families, Urita et al14 found a decline in the prevalence of Helicobacter pylori colonization from 68.7% in the first generation to 43.4% in the second generation and 12.5% in the third. Clemente et al15 studied the intestinal microbiota in a previously uncontacted group of Yanomami people in the Amazon jungle and found they had the highest diversity of bacteria ever reported in a human group. By comparison, the research team calculated that we in the United States have already lost 50% of our microbial diversity, and 2 other groups, the Guahibo (another Amerindian group) and rural Malawians, were in between. More recent studies are confirming these observations.16,17
USE AND OVERUSE OF ANTIBIOTICS
More than 73 billion antibiotic doses are prescribed worldwide yearly,18 or about 10 doses for every man, woman, and child on Earth, and the numbers are rising. In the United States 262 million courses were prescribed in 2011, or 842 per 1,000 population.19 Children receive a mean of 2.7 courses by age 2, and 10.9 by age 10. More than 50% of women receive antibiotics during pregnancy or perinatally. This is in addition to an unknown level of exposure from agricultural use of antibiotics.
Repeated antibiotic exposure is common in early life, varies widely by country, and is often not medically justified.20 In the United States, antibiotic use varies by region, with the heaviest use in the South.19,21 It also varies widely among prescribers.22 Jones et al23 examined antibiotic prescribing for acute respiratory infections in US veterans and found that the top 10% of physicians gave an antibiotic more than 90% of the time. Physicians in Sweden prescribe about 60% fewer antibiotics than we do in the United States.21,24
Observational data indicate that people who receive antibiotics have a higher risk of chronic diseases later in life, eg:
- Type 2 diabetes (odds ratio 1.21, 95% confidence interval 1.19–1.23 with 2 to 4 courses, and odds ratio 1.53 (1.50–1.55) with 5 or more courses, up to 15 years after25
- Obesity: US states with the highest prevalence of antibiotic use also have the highest prevalence of obesity26
- Kidney stones: prior antibiotic exposure in a large UK study was associated with increased kidney stone risk, for exposures up to 5 years earlier.27
The meat industry has exploited the weight effect for decades, adding subtherapeutic doses of antibiotics to animals’ feed to make them gain weight.28
FINDINGS FROM STUDIES IN MICE
Laboratory studies of the relationship between antibiotic exposure and disease phenotypes in mice have yielded interesting findings.
Mice exposed to antibiotics had more body fat at 10 weeks (32.0%) than control mice (22.9%).29
Low-dose penicillin, started at birth, induces long-lasting effects on the expression of genes involved in immunity and enhances the effect of a high-fat diet in terms of weight gain.30 If the antibiotic exposure is limited to early life, the effect on the microbiota is transient, but the mice still gain weight. If the microbiota from the mice who received penicillin is transferred to germ-free mice, the recipients also become fat, indicating that the bacteria, not the antibiotics per se, cause the weight gain.
In other experiments,31 a series of short, therapeutic doses of antibiotics early in life modeled after those given to children to treat their acute infections caused long-term changes in the composition of the microbiome and in metabolism.
A single course of a macrolide antibiotic also had long-term effects on the microbial population and on the host’s ileal gene expression, T-cell populations, and secretory immunoglobulin A expression.32 These effects were seen only in mice that had a microbiome to begin with, not in germ-free mice, indicating that the antibiotics had their effect through the changes in the microbiome, not directly. But when germ-free mice received a fecal transplant of an impaired microbiome, it was sufficient to affect immunity.
In nonobese diabetic mice, treatment with antibiotics early in life altered the gut microbiome and its metabolic capacities, intestinal gene expression, and T-cell populations, accelerating the onset of type 1 diabetes.33
In a study in Danish children,34 the likelihood of inflammatory bowel disease increased with early-life antibiotic exposure: the more courses the child received, the greater the likelihood of disease. This observation led researchers to wonder if an antibiotic-altered microbiome affects the outcome of inflammatory bowel disease in the next generation.35 Germ-free female mice who received microbiota from mice who had received antibiotics passed the altered microbiome to their pups. Mice lacking the gene for interleukin 10 are genetically susceptible to colitis, and when this experiment was done in mice lacking this gene, the offspring developed markedly more colitis. This indicated the mothers could pass down their altered microbiome to the next generation and that it would affect their risk of disease.
WHAT CAN WE DO?
All physicians must adhere to the principles of antibiotic stewardship,36 not only to prevent the development of resistant strains of pathogens and the overgrowth of potentially dangerous species such as Clostridium difficile, but also, possibly, to prevent the loss of diversity in the human microbiome and thus discourage the development of chronic diseases.
In the future, as we discover more about the microbiome and the optimal mix of bacteria to carry, this information may find practical application in medicine. A pediatrician, for example, may want to analyze a child’s microbiome and, if it is abnormal, administer specific organisms to reshape it.
- TEDDY Study Group. The Environmental Determinants of Diabetes in the Young (TEDDY) study. Ann NY Acad Sci 2008; 1150:1–13. doi:10.1196/annals.1447.062
- El-Serag HB, Sonnenberg A. Associations between different forms of gastro-oesophageal reflux disease. Gut 1997; 41(5):594–599. pmid:9414963
- Eder W, Ege MJ, von Mutius E. The asthma epidemic. N Engl J Med 2006; 355(21):2226–2235. doi:10.1056/NEJMra054308
- Kaplan GG, Ng SC. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology 2017; 152(2):313–321. doi:10.1053/j.gastro.2016.10.020
- de Onis M, Blossner M, Borghi E. Global prevalence and trends of overweight and obesity among preschool children. Am J Clin Nutr 2010; 92(5):1257–1264. doi:10.3945/ajcn.2010.29786
- Blaser MJ. The theory of disappearing microbiota and the epidemics of chronic disease. Nat Rev Immunol 2017; 17(8):461–463. doi:10.1038/nri.2017.77
- Centers for Disease Control and Prevention. National Center for Health Statistics. Obesity and overweight. www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed November 6, 2018.
- Centers for Disease Control and Prevention. National Center for Health Statistics. Table 59. Obesity among children and adolescents aged 2-19 years, by selected characteristics: United States, selected years 1988–1994 through 2013–2016. www.cdc.gov/nchs/data/hus/2017/059.pdf. Accessed November 6, 2018.
- Ochman H, Worobey M, Kuo CH, et al. Evolutionary relationships of wild hominids recapitulated by gut microbial communities. PLoS Biology 2010; 8(11):e1000546. doi:10.1371/journal.pbio.1000546
- Bokulich NA, Chung J, Battaglia T, et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci Trans Med 2016; 8(343):343ra82. doi:10.1126/scitranslmed.aad7121
- Yatsunenko T, Rey FE, Manary MJ, et al. Human gut microbiome viewed across age and geography. Nature 2012; 486(7402):222–227. doi:10.1038/nature11053
- Blaser MJ. The past and future biology of the human microbiome in an age of extinctions. Cell 2018; 172(6):1173–1177. doi:10.1016/j.cell.2018.02.040
- Blaser MJ, Falkow S. What are the consequences of the disappearing human microbiota? Nat Rev Microbiol 2009; 7(12):887–894. doi:10.1038/nrmicro2245
- Urita Y, Watanabe T, Kawagoe N, et al. Role of infected grandmothers in transmission of Helicobacter pylori to children in a Japanese rural town. J Ped Child Health 2013; 49(5):394–398. doi:10.1111/jpc.12191
- Clemente JC, Pehrsson EC, Blaser MJ, et al. The microbiome of uncontacted Amerindians. Sci Adv 2015; 1(3). Pii:e1500183. doi:10.1126/sciadv.1500183
- Smits SA, Leach J, Sonnenburg ED, et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 2017; 357(6353):802-806. doi:10.1126/science.aan4834
- Vangay P, Johnson AJ, Ward TL, et al. US immigration westernizes the human gut microbiome. Cell 2018; 175(4):962–972. doi:10.1016/j.cell.2018.10.029
- Van Broeckel TP, Gandra S, Ashok A, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis 2014; 14(8):742–750. doi:10.1016/S1473-3099(14)70780-7
- Hicks LA, Bartoces MG, Roberts RM, et al. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis 2015; 60(9):1308–1316. doi:10.1093/cid/civ076
- Rogawski ET, Platts-Mills JA, Seidman JC, et al. Use of antibiotics in children younger than two years in eight countries: a prospective cohort study. Bull World Health Organ 2017; 95(1):49–61. doi:10.2471/BLT.16.176123
- Hicks LA, Taylor TH Jr, Hunkler RJ. U.S. outpatient antibiotic prescribing, 2010; N Engl J Med 2013; 368(15):1461–1462. doi:10.1056/NEJMc1212055
- Gerber JS, Prasad PA, Russell LA, et al. Variation in antibiotic prescribing across a pediatric primary care network. J Pediatric Infect Dis Soc 2015; 4(4):297–304. doi:10.1093/jpids/piu086
- Jones BE, Sauer B, Jones MM, et al. Variation in outpatient antibiotic prescribing for acute respiratory infections in the veteran population: a cross-sectional study. Ann Intern Med 2015; 163(2):73–80. doi:10.7326/M14-1933
- Ternhag A, Hellman J. More on U.S. outpatient antibiotic prescribing, 2010. N Engl J Med 2013; 369(12):1175. doi:10.1056/NEJMc1306863
- Mikkelsen KH, Knop FK, Frost M, Hallas J, Pottegard A. Use of antibiotics and risk of type 2 diabetes: a population-based case-control study. J Clin Endocrinol Metab 2015; 100(10):3633–3640. doi:10.1210/jc.2015-2696
- Petschow B, Dore J, Hibbert P, et al. Probiotics, prebiotics, and the host microbiome: the science of translation. Ann NY Acad Sci 2013; 1306:1–17. doi:10.1111/nyas.12303
- Tasian GE, Jemielita T, Goldfarb DS, et al. Oral antibiotic exposure and kidney stone disease. J Am Soc Nephrol 2018; 29(6):1731–1740. doi:10.1681/ASN.2017111213
- Zimmerman DR. Role of subtherapeutic levels of antimicrobials in pig production. J Anim Sci 1986; 62(suppl 3):6–16.
- Cho I, Yamanishi S, Cox L, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012; 488(7413):621–626. doi:10.1038/nature11400
- Cox LM, Yamanishi S, Sohn J, et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 2014; 158(4):705–721. doi:10.1016/j.cell.2014.05.052
- Nobel YR, Cox LM, Kirigin FF, et al. Metabolic and metagenomics outcomes from early-life pulsed antibiotic treatment. Nat Commun 2015; 6:7486. doi:10.1038/ncomms8486
- Ruiz VE, Battaglia T, Kurtz ZD, et al. A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity. Nat Commun 2017; 8(1):518. doi:10.1038/s41467-017-00531-6
- Livanos AE, Greiner TU, Vangay P, et al. Antibiotic-mediated gut microbiome perturbation accelerates development of type 1 diabetes in mice. Nat Microbiol 2016; 1(11):16149. doi:10.1038/nmicrobiol.2016.140
- Hvilid A, Svanström H, Frish M. Antibiotic use and inflammatory bowel disease in childhood. Gut 2011; 60(1):49–54. doi:10.1136/gut.2010.219683
- Schulfer AF, Battaglia T, Alvarez Y, et al. Intergenerational transfer of antibiotic-perturbed microbiota enhances colitis in susceptible mice. Nat Microbiol 2018; 3(2):234–242. doi:10.1038/s41564-017-0075-5
- Srinivasan A. Antibiotic stewardship: why we must, how we can. Cleve Clin J Med 2017; 84(9):673–679. doi:10.3949/ccjm.84gr.17003
- TEDDY Study Group. The Environmental Determinants of Diabetes in the Young (TEDDY) study. Ann NY Acad Sci 2008; 1150:1–13. doi:10.1196/annals.1447.062
- El-Serag HB, Sonnenberg A. Associations between different forms of gastro-oesophageal reflux disease. Gut 1997; 41(5):594–599. pmid:9414963
- Eder W, Ege MJ, von Mutius E. The asthma epidemic. N Engl J Med 2006; 355(21):2226–2235. doi:10.1056/NEJMra054308
- Kaplan GG, Ng SC. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology 2017; 152(2):313–321. doi:10.1053/j.gastro.2016.10.020
- de Onis M, Blossner M, Borghi E. Global prevalence and trends of overweight and obesity among preschool children. Am J Clin Nutr 2010; 92(5):1257–1264. doi:10.3945/ajcn.2010.29786
- Blaser MJ. The theory of disappearing microbiota and the epidemics of chronic disease. Nat Rev Immunol 2017; 17(8):461–463. doi:10.1038/nri.2017.77
- Centers for Disease Control and Prevention. National Center for Health Statistics. Obesity and overweight. www.cdc.gov/nchs/fastats/obesity-overweight.htm. Accessed November 6, 2018.
- Centers for Disease Control and Prevention. National Center for Health Statistics. Table 59. Obesity among children and adolescents aged 2-19 years, by selected characteristics: United States, selected years 1988–1994 through 2013–2016. www.cdc.gov/nchs/data/hus/2017/059.pdf. Accessed November 6, 2018.
- Ochman H, Worobey M, Kuo CH, et al. Evolutionary relationships of wild hominids recapitulated by gut microbial communities. PLoS Biology 2010; 8(11):e1000546. doi:10.1371/journal.pbio.1000546
- Bokulich NA, Chung J, Battaglia T, et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci Trans Med 2016; 8(343):343ra82. doi:10.1126/scitranslmed.aad7121
- Yatsunenko T, Rey FE, Manary MJ, et al. Human gut microbiome viewed across age and geography. Nature 2012; 486(7402):222–227. doi:10.1038/nature11053
- Blaser MJ. The past and future biology of the human microbiome in an age of extinctions. Cell 2018; 172(6):1173–1177. doi:10.1016/j.cell.2018.02.040
- Blaser MJ, Falkow S. What are the consequences of the disappearing human microbiota? Nat Rev Microbiol 2009; 7(12):887–894. doi:10.1038/nrmicro2245
- Urita Y, Watanabe T, Kawagoe N, et al. Role of infected grandmothers in transmission of Helicobacter pylori to children in a Japanese rural town. J Ped Child Health 2013; 49(5):394–398. doi:10.1111/jpc.12191
- Clemente JC, Pehrsson EC, Blaser MJ, et al. The microbiome of uncontacted Amerindians. Sci Adv 2015; 1(3). Pii:e1500183. doi:10.1126/sciadv.1500183
- Smits SA, Leach J, Sonnenburg ED, et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 2017; 357(6353):802-806. doi:10.1126/science.aan4834
- Vangay P, Johnson AJ, Ward TL, et al. US immigration westernizes the human gut microbiome. Cell 2018; 175(4):962–972. doi:10.1016/j.cell.2018.10.029
- Van Broeckel TP, Gandra S, Ashok A, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis 2014; 14(8):742–750. doi:10.1016/S1473-3099(14)70780-7
- Hicks LA, Bartoces MG, Roberts RM, et al. US outpatient antibiotic prescribing variation according to geography, patient population, and provider specialty in 2011. Clin Infect Dis 2015; 60(9):1308–1316. doi:10.1093/cid/civ076
- Rogawski ET, Platts-Mills JA, Seidman JC, et al. Use of antibiotics in children younger than two years in eight countries: a prospective cohort study. Bull World Health Organ 2017; 95(1):49–61. doi:10.2471/BLT.16.176123
- Hicks LA, Taylor TH Jr, Hunkler RJ. U.S. outpatient antibiotic prescribing, 2010; N Engl J Med 2013; 368(15):1461–1462. doi:10.1056/NEJMc1212055
- Gerber JS, Prasad PA, Russell LA, et al. Variation in antibiotic prescribing across a pediatric primary care network. J Pediatric Infect Dis Soc 2015; 4(4):297–304. doi:10.1093/jpids/piu086
- Jones BE, Sauer B, Jones MM, et al. Variation in outpatient antibiotic prescribing for acute respiratory infections in the veteran population: a cross-sectional study. Ann Intern Med 2015; 163(2):73–80. doi:10.7326/M14-1933
- Ternhag A, Hellman J. More on U.S. outpatient antibiotic prescribing, 2010. N Engl J Med 2013; 369(12):1175. doi:10.1056/NEJMc1306863
- Mikkelsen KH, Knop FK, Frost M, Hallas J, Pottegard A. Use of antibiotics and risk of type 2 diabetes: a population-based case-control study. J Clin Endocrinol Metab 2015; 100(10):3633–3640. doi:10.1210/jc.2015-2696
- Petschow B, Dore J, Hibbert P, et al. Probiotics, prebiotics, and the host microbiome: the science of translation. Ann NY Acad Sci 2013; 1306:1–17. doi:10.1111/nyas.12303
- Tasian GE, Jemielita T, Goldfarb DS, et al. Oral antibiotic exposure and kidney stone disease. J Am Soc Nephrol 2018; 29(6):1731–1740. doi:10.1681/ASN.2017111213
- Zimmerman DR. Role of subtherapeutic levels of antimicrobials in pig production. J Anim Sci 1986; 62(suppl 3):6–16.
- Cho I, Yamanishi S, Cox L, et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012; 488(7413):621–626. doi:10.1038/nature11400
- Cox LM, Yamanishi S, Sohn J, et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 2014; 158(4):705–721. doi:10.1016/j.cell.2014.05.052
- Nobel YR, Cox LM, Kirigin FF, et al. Metabolic and metagenomics outcomes from early-life pulsed antibiotic treatment. Nat Commun 2015; 6:7486. doi:10.1038/ncomms8486
- Ruiz VE, Battaglia T, Kurtz ZD, et al. A single early-in-life macrolide course has lasting effects on murine microbial network topology and immunity. Nat Commun 2017; 8(1):518. doi:10.1038/s41467-017-00531-6
- Livanos AE, Greiner TU, Vangay P, et al. Antibiotic-mediated gut microbiome perturbation accelerates development of type 1 diabetes in mice. Nat Microbiol 2016; 1(11):16149. doi:10.1038/nmicrobiol.2016.140
- Hvilid A, Svanström H, Frish M. Antibiotic use and inflammatory bowel disease in childhood. Gut 2011; 60(1):49–54. doi:10.1136/gut.2010.219683
- Schulfer AF, Battaglia T, Alvarez Y, et al. Intergenerational transfer of antibiotic-perturbed microbiota enhances colitis in susceptible mice. Nat Microbiol 2018; 3(2):234–242. doi:10.1038/s41564-017-0075-5
- Srinivasan A. Antibiotic stewardship: why we must, how we can. Cleve Clin J Med 2017; 84(9):673–679. doi:10.3949/ccjm.84gr.17003
Diagnosing and treating bipolar disorder in primary care
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
Patients presenting with depression commonly have undiagnosed bipolar depression,1–7 that is, depression with shifts to periods of mania. During manic or hypomanic episodes, people feel energetic, need little sleep, and are often happy and charming.8 But too much of a good thing can also wreak havoc on their life.
Bipolar depression (ie, depression in patients with a diagnosis of bipolar disorder) is treated differently from unipolar depression,3,9–13 making it especially important that clinicians recognize if a patient who presents with depression has a history of (hypo)manic symptoms.
CASE 1: THE IMPULSIVE NURSE
A 32-year-old nurse presents to her primary care provider with depressed mood. She reports having had a single depressive episode when she was a college freshman. Her family history includes depression, bipolar disorder, and schizophrenia, and her paternal grandfather and a maternal aunt committed suicide. Upon questioning, she reveals that in the past, she has had 3 episodes lasting several weeks and characterized by insubordinate behavior at work, irritability, high energy, and decreased need for sleep. She regrets impulsive sexual and financial decisions that she made during these episodes and recently filed for personal bankruptcy. For the past month, her mood has been persistently low, with reduced sleep, appetite, energy, and concentration, and with passive thoughts of suicide.
A CAREFUL HISTORY IS CRITICAL
This case illustrates many typical features of bipolar depression that are revealed only by taking a thorough history. Although the patient is high-functioning, having attained a professional career, she has serious problems with sexual and financial impulsivity and at her job. She has a strong family history of mood disorder. And she describes episodes of depression and mania in the past.
Starts in young adulthood, strong heritability
Bipolar disorder can be a devastating condition with lifelong consequences,14–20 especially as it typically starts when patients are getting an education or embarking on a career. It usually first manifests in the late teenage years and progresses in the patient’s early 20s.21,22 The first hospitalization can occur soon thereafter.23,24
Bipolar disorder is one of the most heritable conditions in psychiatry, and about 13% of children who have an afflicted parent develop it.25 In identical twins, the concordance is about 50% to 75%, indicating the importance of genetics and environmental factors.26,27
Associated with migraine, other conditions
The disorder is associated with a variety of conditions (Table 1).28,29 Some conditions (eg, thyroid disease) can cause mood cycling, and some (eg, sexually transmitted infections, accidents) are the consequences of the lifestyle that may accompany mania. For unknown reasons, migraine is highly associated with bipolar disorder.
DEPRESSION AND MANIA: TWO SIDES OF THE SAME COIN
Symptoms of depression and mania are frequently viewed as opposite mood states, though many times patients report a mixture of them.17,30–35 For both states, the features of a distinct change from the patient’s normal condition and the sustained nature of the symptoms are important diagnostically and indicate a likely underlying biological cause.
Major depressive disorder: Slowing down
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5),8 defines major depressive disorder as having either depressed mood or markedly diminished pleasure in most activities for most days during at least 2 weeks.
In addition, at least 4 of the following must be present during the same period:
- Appetite disturbance
- Sleep disturbance
- Motor retardation or agitation
- Lack of energy
- Feelings of worthlessness or excessive guilt
- Decreased concentration
- Recurrent thoughts of death or suicide.
An estimated 20% of the population experience a major depressive episode over their lifetime. A surprisingly high proportion of people with depression—30% to 40%—also have had subthreshold symptoms of mania (symptoms not meeting the criteria for hypomania or mania in terms of number of symptoms or duration).21,22 Because of these odds, it is important to suspect bipolar disorder even in patients who present with depression but who may not yet have manifested episodes of mania or hypomania.
Mood disorders can be regarded as falling into a spectrum, ranging from unipolar or “pure” major depression without any features of hypomania to major depression and severe mania.17,31–36
Mania: Speeding up
The DSM-5 defines mania as the presence of persistently elevated, expansive, or irritable mood with increased activity for more than 1 week. In addition, at least 3 of the following features must be present, with impaired functioning (4 features are required if mood is only irritable)8:
- Inflated self-esteem or grandiosity
- Decreased need for sleep
- Pressured speech
- Racing thoughts
- Distractibility
- Excessive involvement in pleasurable, high-risk activities.
Hypomania: No functional impairment
Hypomania is a less severe condition, in which the abnormally elevated mood is of shorter duration (4–7 days) and meets the other criteria for mania but without significant functional impairment. People may, in fact, be very functional and productive during hypomanic episodes.8
CLASSIFYING BIPOLAR DISORDER
Bipolar disorder is categorized according to severity.24,37,38 The most severe form, bipolar I disorder, is marked by major depression and manic episodes. It affects up to 1.5% of the US population, with equal proportions of men and women.39 Bipolar II disorder is less severe. It affects 0.8% to 1.6% of the US population, predominantly women.21,40 In bipolar II disorder, depression is more prominent, with episodes of hypomania.
Subthreshold bipolar disorders are characterized by episodic symptoms that do not meet the threshold for depression or hypomania; the symptoms are fewer or of shorter duration. These minor types of bipolar disorder affect up to 6% of the US population.17
Other conditions within the spectrum of bipolar and depressive disorders include medication- and substance-induced mania, agitated or anxious depression, and mixed states.31,34–36
DISTINGUISHING UNIPOLAR FROM BIPOLAR DEPRESSION
Considerable research has focused on finding a clear-cut clinical or biological feature to differentiate unipolar from bipolar depression, but so far none has been discovered. Distinguishing the two conditions still depends on clinical judgment. There are important reasons to identify the distinction between unipolar depression and bipolar disorder.
Prognosis differs. Bipolar disorder tends to be a more severe condition. Young people, who may initially present with only mild symptoms of mania, may develop serious episodes over the years. People may lose their savings, their marriage, and their career during a manic episode. The more critical the occupation (eg, doctor, pilot), the greater the potential consequences of impaired judgment brought on by even mild hypomania.14–20
Treatment differs. Typical antidepressants given for depression can trigger a manic episode in patients with bipolar depression, with devastating consequences. Atypical neuroleptic drugs used to treat bipolar disorder can also have serious effects (eg, metabolic and neurologic effects, including irreversible tardive dyskinesia).3,13,40–43
Despite the good reasons to do so, many doctors (including some psychiatrists) do not ask their patients about a propensity to mania or hypomania.4–6 More stigma is attached to the diagnosis of bipolar disorder than to depression44–47; once it is in the medical record, the patient may have problems with employment and obtaining medical insurance.17,44 The old term “manic-depressive” is often associated in the public mind with a person on the streets displaying severely psychotic behavior; the condition is now understood to consist of a spectrum from mild to more severe illness.
Clinical indicators of bipolarity
There are many indicators that a person who presents with depression may be on the bipolar spectrum, but this is not always easily identified.48–53
History of hypomanic symptoms or subthreshold manic symptoms. Although directly asking the patient about the defining symptoms (eg, “Have you ever had episodes of being ‘hyper’ or too happy?”) may help elicit the diagnosis, many patients with bipolar disorder only report depression, as it is psychically painful. In contrast, hypomania and even mania can be perceived as positive, as patients may have less insight into the abnormality of the condition and feel that they are functioning extremely well.
Early age of onset of a mood disorder, such as severe depression in childhood or early adulthood, points toward bipolar disorder. Diagnosing mood disorders in childhood is difficult, as children are less able to recognize or verbalize many of their symptoms.
Postpartum mood disorder, particularly with psychotic symptoms, indicates a strong possibility of a diagnosis of bipolar disorder.
Drug-induced mania, hypomania, and periods of hyperactivity are key features of bipolar disorder. If asked, patients may report feeling a “buzz” when taking an antidepressant.
Erratic patterns in work and relationships are a red flag and are viewed as “soft signs” of bipolar depression. Akiskal54 described the “rule of three” that should make one consider bipolar disorder: for example, three failed marriages, three current jobs or frequent job changes, three distinct professions practiced at the same time, and simultaneously dating three people. Such features indicate both the hyperfunctioning and the disruptive aspects of mania.
Family history of bipolar disorder or severe psychiatric illness is a very important clue. A more subtle clue described by Akiskal54 may be that several members of the family are very high-functioning in several different fields: eg, one may be a highly accomplished doctor, another a famous lawyer, and another a prominent politician. Or several members of the family may have erratic patterns of work and relationships. However, these subtle clues have been derived from clinical experiences and have not been validated in large-scale studies.
CASE 2: THE FRIENDLY SURGEON
Dr. Z is a prominent surgical subspecialist who is part of a small group practice. His wife has become increasingly worried about his behavior changes at home, including sleeping only a few hours a night, spending sprees, and binge drinking. He reluctantly agrees to an outpatient psychiatric evaluation if she attends with him. He creates a disturbance in the waiting room by shaking everyone’s hands and trying to hug all the women. During his examination, he is loud and expansive, denying he has any problems and describing himself as “the greatest doctor in the world.” The psychiatrist recommends hospitalization, but Dr. Z refuses and becomes belligerent. He announces that he just needs a career change and that he will fly to Mexico to open a bar.
This case, from the Texas Medical Association Archives,55 is not unusual. In addition to many characteristics discussed above, this case is typical in that the spouse brought the patient in, reflecting that the patient lacked insight that his behavior was abnormal. The disinhibition (hugging women), grandiosity, and unrealistic plans are also typical.
DIFFERENTIAL DIAGNOSIS OF BIPOLAR DEPRESSION
Anxiety disorders may be associated with dissociative speech or racing thoughts, which can be confused with bipolar illness. Personality disorders (eg, borderline, narcissistic, sociopathic) can involve a tumultuous and impulsive lifestyle resembling episodes of depression and mania. Schizoaffective illness has features of schizophrenia and bipolar disorder.
It is also possible that, despite what may look like mild features of bipolar disorder, there is no psychiatric condition. Some people with mild mania—often successful professionals or politicians—have high energy and can function very well with only a few hours of sleep. Similarly, depressive symptoms for short periods of time can be adaptive, such as in the face of a serious setback when extreme reflection and a period of inactivity can be useful, leading to subsequent reorganization.
A psychiatric diagnosis is usually made only when there is an abnormality, ie, the behavior is beyond normal limits, the person cannot control his or her symptoms, or social or occupational functioning is impaired.
SCREENING INSTRUMENTS
A few tools help determine the likelihood of bipolar disorder.
The Patient Health Questionnaire (PHQ-9)59,60 is a good 9-item screening tool for depression.
The Mood Disorder Questionnaire60 is specific for bipolar disorder, and like the PHQ-9, it is a patient-reported, short questionnaire that is available free online. The Mood Disorder Questionnaire asks about the symptoms of mania in a yes-no format. The result is positive if all of the following are present:
- A “yes” response to 7 of the 13 features
- Several features occur simultaneously
- The features are a moderate or serious problem.
Unlike most screening instruments, the Mood Disorder Questionnaire is more specific than sensitive. It is 93% specific for bipolar disorder in patients treated for depression in a primary care setting, but only 58% sensitive.61–63
WHEN TO REFER TO PSYCHIATRY
Patients suspected of having bipolar disorder or who have been previously diagnosed with it should be referred to a psychiatrist if they have certain features, including:
- Bipolar I disorder
- Psychotic symptoms
- Suicide risk or in danger of harming others
- Significantly impaired functioning
- Unclear diagnosis.
CASE 3: A TELEVISION ANCHOR’S DREAM TURNS TO NIGHTMARE
According to a famous news anchor’s autobiography,64 the steroids prescribed for her hives “revved her up.” The next course left her depressed. Antidepressant medications propelled her into a manic state, and she was soon planning a book, a television show, and a magazine all at once. During that time, she bought a cottage online. Her shyness evaporated at parties. “I was suddenly the equal of my high-energy friends who move fast and talk fast and loud,” she wrote. “I told everyone that I could understand why men felt like they could run the world, because I felt like that. This was a new me, and I liked her!”64 She was soon diagnosed with bipolar disorder and admitted to a psychiatric clinic.
TREAT WITH ANTIDEPRESSANTS, MOOD STABILIZERS
In general, acute bipolar disorder should be treated with a combination of an antidepressant and a mood stabilizer, and possibly an antipsychotic drug. An antidepressant should not be used alone, particularly with patients with a diagnosis of bipolar I disorder, because of the risk of triggering mania or the risk of faster cycling between mania and depression.13
Mood stabilizers include lithium, lamotrigine, and valproate. Each can prevent episodes of depression and mania. Lithium, which has been used as a mood stabilizer for 60 years, is specific for bipolar disorder, and it remains the best mood stabilizer treatment.
Antidepressants. The first-line antidepressant medication is bupropion, which is thought to be less likely to precipitate a manic episode,65 though all antidepressants have been associated with this side effect in patients with bipolar disorder. Other antidepressants—for example, selective serotonin reuptake inhibitors such as fluoxetine and dual reuptake inhibitors such as venlafaxine and duloxetine—can also be used. The precipitation of mania and possible increased mood cycling was first described with tricylic antidepressants, so drugs of this class should be used with caution.
Neuroleptic drugs such as aripiprazole, quetiapine, and lurasidone may be the easiest drugs to use, as they have antidepressant effects and can also prevent the occurrence of mania. These medications are frequently classified as mood stabilizers. However, they may not have true mood stabilizing properties such as that of lithium. Importantly, their use tends to entail significant metabolic problems and can lead to hyperlipidemia and diabetes. In addition, Parkinson disease-like symptoms— and in some cases irreversible involuntary movements of the mouth and tongue, as well as the body (tardive dyskinesia)—are important possible side effects.
All psychiatric medications have potential side effects (Table 3). Newer antidepressants and neuroleptics may have fewer side effects than older medications but are not more effective.
Should milder forms of bipolar depression be treated?
A dilemma is whether we should treat milder forms of bipolar depression, such as bipolar II depression, depression with subthreshold hypomania symptoms, or depression in persons with a strong family history of bipolar disorder.
Many doctors are justifiably reluctant to prescribe antidepressants for depression because of the risk of triggering mania. Although mood stabilizers such as lithium would counteract possible mania emergence, physicians often do not prescribe them because of inexperience and fear of risks and possible side effects. Patients are likewise resistant because they feel that use of mood stabilizers is tantamount to being told they are “manic-depressive,” with its associated stigma.
Overuse of atypical neuroleptics such as aripiprazole, quetiapine, and olanzapine has led to an awareness of metabolic syndrome and tardive dyskinesia, also making doctors cautious about using these drugs.
Answer: Yes, but treat with caution
Not treating depression consigns a patient to suffer with untreated depression, sometimes for years. Outcomes for patients with depression and bipolar disorder are often poor because the conditions are not recognized, and even when the conditions are recognized, doctors and patients may be reluctant to medicate appropriately. Medications should be used as needed to treat depression, but with an awareness of the possible side effects and with close patient monitoring.
A truly sustained manic state (unlike the brief euphoria brought on by some drugs) is not actually so easy to induce. In an unpublished Cleveland Clinic study, we monitored peaks of hypomanic symptoms in young patients (ages 15–30) during antidepressant treatment without mood stabilizers. About 30% to 40% of patients had subthreshold manic symptoms or a family history of bipolar disorder; 3 patients out of 51 developed hypomania leading to a change of diagnosis to bipolar disorder. Even in patients who had no risk factors for bipolar disorder, 2 out of 53 converted to a bipolar diagnosis. So conversion rates in patients with subthreshold bipolar disorder seem to be low, and the disorder can be identified early by monitoring the patient closely.
NONPHARMACOLOGIC TREATMENTS FOR DEPRESSION
Psychotherapy is indicated for all patients on medications for depression, as both pharmacologic and nonpharmacologic treatments are more effective when combined.66 Other treatments include transcranial magnetic stimulation, electroconvulsive therapy, light therapy, and exercise. Having a consistent daily routine, particularly regarding the sleep-wake schedule, is also helpful, and patients should be educated about its importance.
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
- Cicchetti D, Toth SL. The development of depression in children and adolescents. Am Psychol 1998; 53(2):221–241. doi:10.1037/0003-066X.53.2.221
- Glick ID. Undiagnosed bipolar disorder: new syndromes and new treatments. Prim Care Companion J Clin Psychiatry 2004;6(1):27–33. pmid:15486598
- Ghaemi SN, Boiman EE, Goodwin FK. Diagnosing bipolar disorder and the effect of antidepressants: a naturalistic study. J Clin Psychiatry 2000; 61(10):804–808. pmid:11078046
- Singh T, Rajput M. Misdiagnosis of bipolar disorder. Psychiatry (Edgmont) 2006; 3(10):57–63. pmid: 20877548
- Lish JD, Dime-Meenan S, Whybrow PC, Price RA, Hirschfeld RM. The National Depressive and Manic-depressive Association (DMDA) survey of bipolar members. J Affect Disord 1994; 31(4):281–294. pmid:7989643
- Howes OD, Falkenberg I. Early detection and intervention in bipolar affective disorder: targeting the development of the disorder. Curr Psychiatry Rep 2011; 13(6):493–499. pmid:21850462
- Ghaemi SN, Sachs GS, Chiou AM, Pandurangi AK, Goodwin K. Is bipolar disorder still underdiagnosed? Are antidepressants overutilized? J Affect Disord 1999; 52(1–3):135–144. pmid:10357026
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-5. Washington, DC: American Psychiatric Publishing, 2013.
- Hlastala SA, Frank E, Mallinger AG, Thase ME, Ritenour AM, Kupfer DJ. Bipolar depression: an underestimated treatment challenge. Depress Anxiety 1997; 5(2):73–83. pmid:9262937
- Smith DJ, Craddock N. Unipolar and bipolar depression: different or the same? Br J Psychiatry 2011; 199(4):272–274. doi:10.1192/bjp.bp.111.092726
- Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry 2014; 171(10):1067–1073. doi:10.1176/appi.ajp.2014.13111501
- Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med 2007; 356(17):1711–1722. doi:10.1056/NEJMoa064135
- American Psychiatric Association. Practice guideline for the treatment of patients with bipolar disorder (revision). Am J Psychiatry 2002; 159(4 suppl):1–50. pmid:11958165
- Leonpacher AK, Liebers D, Pirooznia M, et al. Distinguishing bipolar from unipolar depression: the importance of clinical symptoms and illness features. Psychol Med 2015; 45(11):2437–2446. doi:10.1017/S0033291715000446
- Angst J, Gamma A, Benazzi F, Ajdacic V, Eich D, Rössler W. Toward a re-definition of subthreshold bipolarity: epidemiology and proposed criteria for bipolar-II, minor bipolar disorders and hypomania. J Affect Disord 2003; 73(1–2):133–146. pmid:12507746
- Faravelli C, Rosi S, Alessandra Scarpato M, Lampronti L, Amedei SG, Rana N. Threshold and subthreshold bipolar disorders in the Sesto Fiorentino Study. J Affect Disord 2006; 94(1–3):111–119. pmid:16701902
- Judd LL, Akiskal HS. The prevalence and disability of bipolar spectrum disorders in the US population: re-analysis of the ECA database taking into account subthreshold cases. J Affect Disord 2003; 73(1–2):123–131. pmid:12507745
- Park Y-M, Lee B-H. Treatment response in relation to subthreshold bipolarity in patients with major depressive disorder receiving antidepressant monotherapy: a post hoc data analysis (KOMDD study). Neuropsychiatr Dis Treat 2016; 12:1221–1227. doi:10.2147/NDT.S104188
- Perlis RH, Uher R, Ostacher M, Goldberg JF, et al. Association between bipolar spectrum features and treatment outcomes in outpatients with major depressive disorder. Arch Gen Psychiatry 2011; 68(4):351–360. doi:10.1001/archgenpsychiatry.2010.179
- Dudek D, Siwek M, Zielin´ska D, Jaeschke R, Rybakowski J. Diagnostic conversions from major depressive disorder into bipolar disorder in an outpatient setting: results of a retrospective chart review. J Affect Disord 2013; 144(1–2):112–115. doi:10.1016/j.jad.2012.06.014
- Angst J, Cui L, Swendsen J, et al. Major depressive disorder with subthreshold bipolarity in the National Comorbidity Survey Replication. Am J Psychiatry 2010; 167(10):1194–1201. doi:10.1176/appi.ajp.2010.09071011
- Zimmermann P, Brückl T, Nocon A, et al. Heterogeneity of DSM-IV major depressive disorder as a consequence of subthreshold bipolarity. Arch Gen Psychiatry 2009; 66(12):1341–1352. doi:10.1001/archgenpsychiatry.2009.158
- Patel NC, DelBello MP, Keck PE, Strakowski SM. Phenomenology associated with age at onset in patients with bipolar disorder at their first psychiatric hospitalization. Bipolar Disord 2006; 8(1):91–94. pmid:16411986
- Hirschfeld RMA, Lewis L, Vornik LA. Perceptions and impact of bipolar disorder: how far have we really come? Results of the National Depressive and Manic-Depressive Association 2000 survey of individuals with bipolar disorder. J Clin Psychiatry 2003; 64(2):161–174. pmid:12633125
- Craddock N, Jones I. Genetics of bipolar disorder. J Med Genet 1999; 36(8):585–594. pmid:10465107
- Griswold KS, Pessar LF. Management of bipolar disorder. Am Fam Physician 2000; 62(6):1343–1358. pmid:11011863
- Kerner B. Genetics of bipolar disorder. Appl Clin Genet 2014; 7:33–42. doi:10.2147/TACG.S39297
- Scheffer RE, Linden S. Concurrent medical conditions with pediatric bipolar disorder. Curr Opin Psychiatry 2007; 20(4):398–401. doi:10.1097/YCO.0b013e3281a305c3
- Carney CP, Jones LE. Medical comorbidity in women and men with bipolar disorders: a population-based controlled study. Psychosom Med 2006;68(5):684–691. doi:10.1097/01.psy.0000237316.09601.88
- Cassano GB, Akiskal HS, Savino M, Musetti L, Perugi G. Proposed subtypes of bipolar II and related disorders: with hypomanic episodes (or cyclothymia) and with hyperthymic temperament. J Affect Disord 1992; 26(2):127–140. pmid:1447430
- Akiskal HS, Mallya G. Criteria for the “soft” bipolar spectrum: treatment implications. Psychopharmacol Bull 1987; 23(1):68–73. pmid:3602332
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Maj M, Pirozzi R, Magliano L, Fiorillo A, Bartoli L. Agitated “unipolar” major depression: prevalence, phenomenology, and outcome. J Clin Psychiatry 2006; 67(5):712–719. pmid:16841620
- Akiskal HS. The bipolar spectrum: new concepts in classification and diagnosis. In: Psychiatry Update: the American Psychiatric Association Annual Review. Washington, DC: American Psychiatric Press; 1983:271–292.
- Akiskal HS, Pinto O. The evolving bipolar spectrum. Prototypes I, II, III, and IV. Psychiatr Clin North Am 1999; 22(3):517–534. pmid:10550853
- Cassano GB, Savino M, Perugi G, Musetti L, Akiskal HS. Major depressive episode: unipolar and bipolar II. Encephale 1992 Jan;18 Spec No:15–18. pmid:1600898
- Müller-Oerlinghausen B, Berghöfer A, Bauer M. Bipolar disorder. Lancet 2002; 359(9302):241–247. pmid:11812578
- Hirschfeld RMA, Calabrese JR, Weissman MM, et al. Screening for bipolar disorder in the community. J Clin Psychiatry 2003; 64(1):53–59. pmid:12590624
- Blanco C, Compton WM, Saha TD, et al. Epidemiology of DSM-5 bipolar I disorder: results from the National Epidemiologic survey on Alcohol and Related Conditions—III. J Psychiatr Res 2017; 84:310–317. doi:10.1016/j.jpsychires.2016.10.003
- McGirr A, Vöhringer PA, Ghaemi SN, Lam RW, Yatham LN. Safety and efficacy of adjunctive second-generation antidepressant therapy with a mood stabiliser or an atypical antipsychotic in acute bipolar depression: a systematic review and meta-analysis of randomised placebo-controlled trials. Lancet Psychiatry 2016; 3(12):1138–1146. doi:10.1016/S2215-0366(16)30264-4
- Gijsman HJ, Geddes JR, Rendell JM, Nolen WA, Goodwin GM. Antidepressants for bipolar depression: a systematic review of randomized, controlled trials. Am J Psychiatry 2004; 161(9):1537–1547. doi:10.1176/appi.ajp.161.9.1537
- Sidor MM, Macqueen GM. Antidepressants for the acute treatment of bipolar depression: a systematic review and meta-analysis. J Clin Psychiatry 2011; 72(2):156–167. doi:10.4088/JCP.09r05385gre
- Liu B, Zhang Y, Fang H, Liu J, Liu T, Li L. Efficacy and safety of long-term antidepressant treatment for bipolar disorders - A meta-analysis of randomized controlled trials. J Affect Disord 2017; 223(139):41–48. doi:10.1016/j.jad.2017.07.023
- Krupa T, Kirsh B, Cockburn L, Gewurtz R. Understanding the stigma of mental illness in employment. Work 2009; 33(4):413–425. doi:10.3233/WOR-2009-0890
- Hawke LD, Parikh SV, Michalak EE. Stigma and bipolar disorder: a review of the literature. J Affect Disord 2013; 150(2):181–191. doi:10.1016/j.jad.2013.05.030
- Cerit C, Filizer A, Tural Ü, Tufan AE. Stigma: a core factor on predicting functionality in bipolar disorder. Compr Psychiatry 2012; 53(5):484–489. doi:10.1016/j.comppsych.2011.08.010
- O’Donnell L, Himle JA, Ryan K, et al. Social aspects of the workplace among individuals with bipolar disorder. J Soc Social Work Res 2017; 8(3):379–398. doi:10.1086/693163
- Akiskal HS, Maser JD, Zeller PJ, et al. Switching from “unipolar” to bipolar II. An 11-year prospective study of clinical and temperamental predictors in 559 patients. Arch Gen Psychiatry 1995; 52(2):114–123. pmid:7848047
- Kroon JS, Wohlfarth TD, Dieleman J, et al. Incidence rates and risk factors of bipolar disorder in the general population: a population-based cohort study. Bipolar Disord 2013; 15(3):306–313. doi:10.1111/bdi.12058
- Fiedorowicz JG, Endicott J, Leon AC, Solomon DA, Keller MB, Coryell WH. Subthreshold hypomanic symptoms in progression from unipolar major depression to bipolar disorder. Am J Psychiatry 2011; 168(1):40–48. doi:10.1176/appi.ajp.2010.10030328
- Akiskal HS, Walker P, Puzantian VR, King D, Rosenthal TL, Dranon M. Bipolar outcome in the course of depressive illness. Phenomenologic, familial, and pharmacologic predictors. J Affect Disord 1983; 5(2):115–128. pmid:6222091
- Strober M, Carlson G. Bipolar illness in adolescents with major depression: clinical, genetic, and psychopharmacologic predictors in a three- to four-year prospective follow-up investigation. Arch Gen Psychiatry 1982; 39(5):549–555. pmid:7092488
- Goldberg JF, Harrow M, Whiteside JE. Risk for bipolar illness in patients initially hospitalized for unipolar depression. Am J Psychiatry 2001; 158(8):1265–1270. pmid:11481161
- Akiskal HS. Searching for behavioral indicators of bipolar II in patients presenting with major depressive episodes: the “red sign,” the “rule of three” and other biographic signs of temperamental extravagance, activation and hypomania. J Affect Disord 2005; 84(2–3):279–290. pmid:15708427
- Texas Medical Association. Mood disorders in physicians. www.texmed.org/Template.aspx?id=6833. Accessed June 7, 2018.
- Hirschfeld RM. Differential diagnosis of bipolar disorder and major depressive disorder. J Affect Disord 2014;169(suppl 1):S12–S16. doi:10.1016/S0165-0327(14)70004-7
- Dunner DL. Differential diagnosis of bipolar disorder. J Clin Psychopharmacol 1992; 12(1suppl):7S–12S. pmid:1541721
- Peet M, Peters S. Drug-induced mania. Drug Saf 1995; 12(2):146–153. pmid:7766338
- Spitzer RL, Kroenke K, Williams JBW. Validation and utility of a self-report version of PRIME-MD: the PHQ primary care study. Primary Care Evaluation of Mental Disorders. Patient Health Questionnaire. JAMA 1999; 282(18):1737–1744. pmid:10568646
- Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16(9):606–613. pmid:11556941
- Hirschfeld RMMA, Williams JBBW, Spitzer RL, et al. Development and validation of a screening instrument for bipolar spectrum disorder: the Mood Disorder Questionnaire. Am J Psychiatry 2000; 157(11)1873–1875. doi:10.1176/appi.ajp.157.11.1873
- Hirschfeld RMA. The Mood Disorder Questionnaire: a simple, patient-rated screening instrument for bipolar disorder. Prim Care Companion J Clin Psychiatry 2002; 4(1):9–11. pmid: 15014728
- Das AK, Olfson M, Gameroff MJ, et al. Screening for bipolar disorder in a primary care practice. JAMA 2005; 293(8):956–963. doi:10.1001/jama.293.8.956
- Pauley J. Skywriting: A Life Out of the Blue. New York: Random House, 2004.
- Goren JL, Levin GM. Mania with bupropion: a dose-related phenomenon? Ann Pharmacother 2000; 34(5):619–621. doi:10.1345/aph.19313
- Swann AC. Long-term treatment in bipolar disorder. J Clin Psychiatry 2005; 66(suppl 1):7–12. pmid:15693746
KEY POINTS
- Bipolar depression in its manifest and subthreshold forms is nearly as prevalent as unipolar depression and often occurs in successful professionals.
- A manic or hypomanic episode can make a patient highly productive, but it can also be severely disruptive, leading to loss of job, marriage, and financial savings.
- Identifying bipolar depression depends on asking about bipolar symptoms, using screening instruments, and being aware of clues from the patient’s history.
- A major depressive episode in patients with a history of mania or hypomania should be treated with a combination of an antidepressant and a mood stabilizer or a mood stabilizer alone.
What you can do for your fibromyalgia patient
Fibromyalgia may seem like a nebulous diagnosis, with its array of symptoms and pain refractory to medications. But fibromyalgia is a defined syndrome of neuronal dysregulation. It can be diagnosed from the history and physical examination and managed in a primary care setting.
CASE 1: MANY SYMPTOMS
A 43-year-old woman presents to her primary care physician with multiple complaints: pain in all joints and in her back and hips, swelling of her hands and feet, morning stiffness, chest pain and shortness of breath (not necessarily related to exertion), fatigue, generalized weakness, headaches, difficulty with memory and concentration, dry mouth and dry eyes, feeling weak and faint in the sun, cold intolerance with purple discoloration of her extremities, a self-described “butterfly” rash on the face, and hair that is thinning and falling out in clumps.
Because many of her symptoms could reflect an inflammatory process or an autoimmune disease,1 her primary care physician orders multiple tests. Her C-reactive protein level, Westergren sedimentation rate, complete blood cell count, and comprehensive metabolic panel are normal. Urinalysis shows trace leukocyte esterase. Indirect immunofluorescence assay on human laryngeal tumor (HEp-2) cells is positive for antinuclear antibody (ANA), with a titer of 1:320 (reference range ≤ 1:40) and a nuclear dense fine-speckled pattern.
In view of the positive ANA test, the patient is informed that she may have systemic lupus erythematosus (SLE) and will be referred to a rheumatologist. In the days before her rheumatology appointment, she becomes extremely anxious. Obsessively researching SLE online, she becomes convinced that SLE is the correct diagnosis.
Rheumatology evaluation
The rheumatologist assesses the patient’s pain and reports the following:
Location and duration: Hands, wrists, elbows, shoulders, upper and lower back, sides of hips, knees, and feet; has been ongoing for 10 years, but worse in the past 3 months.
Character: The patient describes her pain as “like an ice pick being driven through my joints,” “sometimes unbearable,” and “like being hit by a truck.” She also reports numb, tingly, burning pain in her upper neck and back.
Variation with time, activity, and weather: Worse at night, causing her to wake and toss and turn all night; better with exertion, but after activity or exercise, she is exhausted for the rest of the day and sometimes for up to a week; worse with weather changes, especially during cold or humid weather.
Associated symptoms: Occasional perceived swelling of hands and feet, especially upon wakening in the morning, and 2 to 3 hours of stiffness in the morning that sometimes lasts all day.
Physical examination. Her findings are inconsistent with her symptoms.
The patient exhibits limited range of motion. When asked to bend forward, rotate her neck, or flex and extend her neck and back, she does so only slightly. However, passive range of motion is normal in all joints.
When her joints are examined, she anticipates pain and withdraws her hands. But when she is distracted, examination reveals no evidence of swollen joints or synovitis. She has tenderness in 12 of 18 tender points. Her peripheral pulses are good, strength is normal, and reflexes are brisk.
Her facial rash looks more like rosacea than the butterfly rash of SLE. There is no indication of patchy hair loss. Heart and lung examinations are normal. She appears to have a good salivary pool without glossitis.
History reveals long-standing psychological issues
The patient reports a history of panic attacks and a prior diagnosis of anxiety. She is tested with the Generalized Anxiety Disorder 7-item scale (www.mdcalc.com/gad-7-general-anxiety-disorder-7) and scores 17 out of 21, indicating severe anxiety.
DISCUSSION: CHARACTERIZING PAIN
Understanding categories of pain syndromes can help us understand fibromyalgia. Pain can be categorized into 3 types that sometimes overlap2:
Nociceptive or peripheral pain is related to damage of tissue by trauma or inflammation. Syndromes include osteoarthritis, rheumatoid arthritis, and SLE.
Neuropathic pain is associated with damage of peripheral or central nerves. Examples are neuropathy from herpes, diabetes, or spinal stenosis.
Centralized pain has no identifiable nerve or tissue damage and is thought to result from persistent neuronal dysregulation, overactive ascending pain pathways, and a deficiency of descending inhibitory pain pathways. There is evidence of biochemical changes in muscles, possibly related to chronic ischemia and an overactive sympathetic nervous system. Dysregulation of the sympathoadrenal system and hypothalamic-pituitary axis has also been implicated. And genetic predisposition is possible. Examples of centralized pain syndromes include fibromyalgia, irritable bowel syndrome, pelvic pain syndrome, neurogenic bladder, and interstitial cystitis.
Clues to a centralized pain syndrome
For patients with suspected fibromyalgia, distinguishing peripheral pain from centralized pain can be a challenge (Table 1). For example, SLE does not cause inflammation of the spine, so neck or back pain is not typical. Although both nociceptive and centralized pain syndromes improve with exertion, only patients with centralized pain are typically exhausted and bedbound for days after activity. Patients with centralized pain tend to describe their pain in much more dramatic language than do patients with inflammatory pain. Centralized pain tends to be intermittent; inflammatory pain tends to be constant. Patients with centralized pain often have had pain for many years without a diagnosis, but this is rare for patients with an inflammatory condition.
A patient with fibromyalgia typically has a normal physical examination, although allodynia (experiencing pain from normally nonpainful stimulation), hyperalgesia (increased pain perception), and brisk reflexes may be present. Fibromyalgia may involve discoloration of the fingertips resulting from an overactive sympathetic nervous system. Laboratory results are typically normal with fibromyalgia.
Patients with either nociceptive or centralized pain report stiffness, but the cause likely differs. We typically think of stiffness as arising from swollen joints caused by inflammation, but stiffness can also be caused by abnormally tight muscles, as occurs in fibromyalgia.
FIBROMYALGIA IS A CLINICAL DIAGNOSIS
Diagnosing fibromyalgia does not require multiple laboratory and imaging tests. The key indicators are derived from the patient history and physical examination.
Diagnostic criteria for fibromyalgia published by the American College of Rheumatology have evolved over the years. The 2011 criteria, in the form of a self-reported patient questionnaire, have 2 components3:
- The Widespread Pain Index measures the extent of pain in 19 areas.
- The Symptom Severity scale assesses 3 key symptoms associated with fibromyalgia, ie, fatigue, cognitive problems, and nonrestorative sleep (scale of 0–3 for each symptom).
There are also questions about symptoms of irritable bowel syndrome, depression, and headache.
Fibromyalgia is diagnosed if a patient reports at least 7 painful areas and has a symptom severity score of at least 5. A patient may also meet the 20113 and the 2016 criteria4 if he or she has 4 painful areas and the pain is perceived in 4 of 5 areas and the Symptom Severity Scale score is 9 or higher.4 This questionnaire is not only a rapid diagnostic tool for fibromyalgia, it also helps identify and focus on specific issues—for example, having severe pain in a few localized areas, or having headache as a predominant problem.
These criteria are useful for a variety of patients, eg, a patient with hip arthritis may score high on the questionnaire, indicating that a component of centralized pain is also present. Also, people who have undergone orthopedic surgery who score high tend to require more narcotics to meet the goals of pain improvement.
The 2016 criteria,4 the most recent, maintain that pain must be generalized, ie, present in at least 4 of 5 body areas. They also emphasize that fibromyalgia is a valid diagnosis irrespective of other conditions.
CASE 1 CONTINUED: THE PATIENT REJECTS THE DIAGNOSIS
Our patient meets the definition of fibromyalgia by each iteration of the American College of Rheumatology clinical criteria. She also has generalized anxiety disorder and a positive ANA test. She is advised to participate in a fibromyalgia educational program, start an aerobic exercise program, and consider taking an antidepressant medication with anxiolytic effects.
However, the patient rejects the diagnosis of fibromyalgia. She believes that the diagnosis of SLE has been overlooked and that her symptom severity is being discounted.
In response, the rheumatologist orders additional tests to evaluate for an autoimmune disorder: extractable nuclear antigen panel, complement C3 and C4, double-stranded DNA antibodies, and protein electrophoresis. Results are all in the normal range. The patient is still concerned that she has SLE or another autoimmune disease because of her abnormal ANA test result and remains dissatisfied with her evaluation. She states that she will complain to the clinic ombudsman.
SIGNIFICANCE OF ANA TESTING
Patients with positive test results increasingly go online to get information. The significance of ANA testing can be confusing, and it is critical to understand and be able to explain abnormal results to worried patients. Following are answers to some common questions about ANA testing:
Is ANA positivity specific for SLE or another autoimmune disease?
No. ANA is usually tested by indirect immunofluorescence assay on HEp-2 cells. The test can identify about 150 antigens targeted by antibodies, but only a small percentage are associated with an autoimmune disease, and the others do not have a known clinical association. Enzyme-linked immunosorbent assay (ELISA) ANA testing is also available but is considered less sensitive.
Abeles and Abeles5 retrospectively assessed 232 patients between 2007 and 2009 who were referred to rheumatologists for evaluation because of an elevated ANA test result. No ANA-associated rheumatic disease was found in patients who had a result of less than 1:160, and more than 90% of referrals for a positive ANA test had no evidence of ANA-associated disease. The positive predictive value was 9% for any connective tissue disease, and only 2% for SLE. The most common reason for ordering the test was widespread pain (23%). The authors concluded that ANA testing is often ordered inappropriately for patients with a low pretest probability of an ANA-associated rheumatic disease.
Screening with ANA testing generates many false-positive results and unnecessary anxiety for patients. The prevalence of SLE in the general population is about 0.1%, and other autoimmune diseases total about 5% to 7%. By indirect immunofluorescence assay, using a cutoff of 1:80 (the standard at Cleveland Clinic), about 15% of the general population test positive. By ELISA, with a cutoff of 20 ELISA units, 25% of healthy controls test positive.
It is true that ANA positivity may precede the onset of SLE.6,7 Arbuckle et al8 evaluated serum samples from the US Department of Defense Serum Repository obtained from 130 people before they received a diagnosis of SLE; in 115 (88%), at least 1 SLE-related autoantibody was present before diagnosis (up to 9.4 years earlier). However, in those who test positive for ANA, the percentage who eventually develop autoimmune disease is small.5
Is the titer of ANA significant and of diagnostic value?
The likelihood of autoimmune disease increases with increasing titer. But high titers can be seen in healthy people. Mariz et al9 examined ANA test results from 918 healthy controls and 153 patients with an autoimmune rheumatic disease. Of these, ANA was positive in 13% of healthy people and 90% of patients with an autoimmune disease. High titers were more likely in patients with an autoimmune disease, but also occurred in healthy controls.
Does the immunofluorescence pattern provide diagnostic information?
It can. There are 28 identified patterns of ANA, including nuclear, cytoplasmic, and mitotic patterns. The most common, the nuclear fine-speckled pattern, is seen in healthy controls and patients with an autoimmune disease. But other patterns are either characteristic of an autoimmune disease or, conversely, of not having an autoimmune disease (Table 2).9
Our patient has a nuclear dense fine-speckled pattern, further reducing the likelihood that she has an autoimmune disease.
CASE 2: POORLY CONTROLLED,
LONG-STANDING FIBROMYALGIA
A 43-year-old woman who has had fibromyalgia for 15 years is referred to a new primary care provider. She reports severe pain all over, low back pain, fatigue, nonrefreshing sleep, chronic migraine, constipation alternating with diarrhea, heartburn, intermittent numbness and tingling in her hands and feet, and depression. At this time, she rates her pain on a visual analog scale as 9 out of 10, and her fatigue as 8 out of 10.
During the past 6 months, she has made 25 visits to specialists in 8 departments: spine, pain management, anesthesia, neurology, headache clinic, gastroenterology, sleep medicine, and physical therapy.
Her daily medications are duloxetine 120 mg, bupropion 300 mg, pregabalin 450 mg, cyclobenzaprine 30 mg, tramadol 200 mg, zolpidem 10 mg, nortriptyline 50 mg, acetaminophen 3,000 mg, and oxycodone 30 mg. She has also tried gabapentin and milnacipran without success. She reported previously taking different selective serotonin reuptake inhibitors and tricyclic antidepressants but cannot remember why they were stopped.
How should this complex patient be managed?
BIOPSYCHOSOCIAL MANAGEMENT
Managing the pain of fibromyalgia requires a different model than used for peripheral pain from injury, in which the source of pain can be identified and treated with injections or oral therapy.
Neuronal dysregulation is not amenable to clinical measurement or treatment by medications at this time. But fortunately, many factors associated with fibromyalgia can be addressed: stressful life events, sleep disturbance, physical deconditioning, mood disorders, and maladaptive pain responses, including “catastrophizing” behavior (coping with pain in a highly dramatic and obsessive way). Modifying these factors can be much more productive than focusing on treating pain.
The goal for care providers is to change the focus from reducing pain to a biopsychosocial model of pain control aimed at increasing function.10
Mood modification
Not only are mood disorders common in patients with fibromyalgia, but the prevalence of complex psychiatric conditions is also elevated. Up to 80% of patients with fibromyalgia meet criteria for axis I (clinical psychological) disorders, and up to about 30% of patients meet criteria for axis II (personality) disorders. About 22% of patients have existing major depression, and about 58% develop it during their lifetime. In a study of 678 patients with fibromyalgia, 21% had bipolar disorder.11–15
The severity of fibromyalgia increases linearly with the severity of depression.16 Patients with fibromyalgia and a “depressive affect balance style” have worse outcomes across all Outcome Measures in Rheumatology (OMERACT) core symptom domains, reporting more pain, fatigue, insomnia, anxiety, depression, and function.17,18
Fibromyalgia combined with mood disorders can also be costly. In one study, the mean annual employer payments (direct and indirect costs) per patient were $5,200 for patients with fibromyalgia only, $8,100 for patients with depression only, and $11,900 for patients with both.19
Obtaining a psychiatric history is important when evaluating a patient with fibromyalgia symptoms. Patients should be asked if they have a history of depression, anxiety, posttraumatic stress disorder, or other conditions. The Patient Health Questionnaire – Depression 9 and the Generalized Anxiety Disorder Assessment (GAD-7) (both available at www.mdcalc.com) can be useful for evaluating mood disorders.
Patients with moderate depression and fibromyalgia who have not yet been treated should be prescribed duloxetine for its potential benefits for both conditions.
Patients who have already been treated with multiple drugs at high doses without benefit, such as our patient, should be referred to a psychiatrist. There is no additional benefit to referring this patient to a rheumatologist or spine clinic.
Addressing sleep problems
Sleep problems are not easy to manage but can often be helped. Epidemiologic studies indicate that poor sleep quality leads to chronic widespread pain in otherwise healthy people.20–22 In addition, experimental sleep deprivation leads to fatigue, cognitive difficulty, and a reduced pain threshold.23 In our patients with fibromyalgia, we have observed an inverse relationship between the number of hours slept and the severity of depression.
Sleep quantity and quality can be assessed by asking patients whether they have trouble sleeping, how many hours they sleep, and whether they have been diagnosed with a sleep disorder.
Because many patients with fibromyalgia are overweight or obese, they should also be evaluated for sleep apnea, narcolepsy, and restless leg syndrome.24,25
Medications shown to improve sleep include pregabalin or gabapentin (taken at bedtime), low-dose amitriptyline, trazodone, cyclobenzaprine, melatonin, and nabilone.26–29
Patients should be counseled about sleep hygiene.30 Exercise can also help sleep.
Targeting maladaptive pain responses
Patients who catastrophize tend to have higher tender point counts, a hyperalgesic response, more depression and anxiety, and more self-reported disability. They are also less likely to return to work.31 They usually respond poorly to medications and are good candidates for cognitive behavioral therapy.
A high score on a self-reported Pain Catastrophizing Scale32 can help determine whether a multidisciplinary approach is advisable, although no threshold defines an abnormal score.
Educating patients about the neurobiology underlying their pain can be therapeutic.33–37 Cognitive behavioral therapy can help patients recognize their faulty thought processes and the relationship between pain and stress, and learn better coping mechanisms.38,39 Patients who achieve the highest improvements in pain catastrophizing tend to derive the greatest benefit from cognitive behavioral therapy.40
Exercise improves symptoms
Exercise improves fibromyalgia on many fronts and is associated with a host of positive effects in the brain and peripheral muscles. Exercise improves Fibromyalgia Impact Questionnaire scores, increases physical function and fitness, and reduces tender point counts, depression, and catastrophizing.41–52 There is no consensus on the best type of exercise, but both strengthening and aerobic exercises appear to be important.
I tell patients that fibromyalgia is an exercise-deprivation syndrome. Many are afraid to exercise because they associate it with pain and exhaustion afterwards. Patients should be encouraged to start with something very low-impact, such as gentle exercise in a warm-water pool. It should be emphasized that exercise is a lifelong treatment.
Drug therapy
The US Food and Drug Administration has approved 3 drugs for fibromyalgia management: 2 serotonin-norepinephrine reuptake inhibitors (duloxetine and milnacipran) and 1 gabapentinoid (pregabalin). Our patient in Case 2 is taking 2 of them without apparent benefit and has previously had no success with the third. This is not surprising. A summary of published treatment research on these drugs found that only 50% to 60% of patients tested reported more than 30% pain reduction.53 The studies also showed a placebo response of 30% to 40%. Depending on the study, the number needed to treat to see a benefit from these drugs is 8 to 14.53
EVALUATING THE SEVERITY OF FIBROMYALGIA
Focusing on key characteristics of the patient’s history can help evaluate fibromyalgia and determine a treatment strategy (Table 3). The Fibromyalgia Impact Questionnaire is also a useful evaluation tool.
It is important to assess the severity of fibromyalgia because patients with severe fibromyalgia are not good candidates for further referral to other specialists. They instead need chronic rehabilitation services, where they can learn to better function with a chronic pain syndrome.
In general, patients with the following features have conditions with high severity:
Symptoms: High burden and intensity
Function: Disabled, unemployed, interference with activities of daily living
Mood: Severe depression, bipolar disorder, axis II disorder, posttraumatic stress disorder
Medications: Polypharmacy, opioid drugs, multiple failed interventions
Maladaptive attitudes: High catastrophizing, refusal to accept diagnosis
Fibromyalgia Impact Questionnaire score: 60 or above.
The fibromyalgia of our patient in Case 2 would be categorized as severe.
MULTIFACETED MANAGEMENT
Patients with fibromyalgia are a heterogeneous group, and the syndrome does not lend itself to a single management strategy.54 Multiple guidelines have been published for managing fibromyalgia.55–57 Thieme et al58 reviewed existing guidelines and the strength of their recommendations. The guidelines unanimously strongly favor exercise, and most also strongly favor cognitive behavioral therapy. Most favor treating with amitriptyline and duloxetine; recommendations for other antidepressants vary. Nonsteroidal anti-inflammatory drugs, opioid drugs, and benzodiazepines are not recommended.
We offer a monthly 1-day clinic for patients and family members to provide education about fibromyalgia, discuss the importance of exercise, counsel on maladaptive responses, and demonstrate mindfulness techniques. We focus on function rather than pain. Interactive online-based interventions using cognitive behavioral techniques, such as FibroGuide: A Symptom Management Program for People Living With Fibromyalgia, developed at the University of Michigan, have proven helpful.59
RECOMMENDATIONS
For most patients, do not focus on pain reduction, as that is ineffective. Instead, target reversible factors, eg, mood, sleep, exercise status, stressors, and maladaptive attitudes toward pain. Possible treatment combinations include:
- A serotonin and norepinephrine reuptake inhibitor (eg, duloxetine)
- A low-dose tricyclic antidepressant at bedtime (eg, amitriptyline)
- A gabapentinoid (pregabalin or gabapentin).
If a medication within a class does not work, stop it and try another rather than add on.
Treat mild to moderate fibromyalgia with multidisciplinary interventions, with or without centrally acting medications. Treat severe fibromyalgia with more intensive psychiatric or psychologic interventions, multidisciplinary care, and medications targeted at comorbidities. Provide all patients with education and advice on exercise.
Keep laboratory tests and imaging studies to a minimum: a complete blood cell count with differential, comprehensive metabolic panel, thyroid-stimulating hormone, C-reactive protein, and Westergren sedimentation rate. Do not test for ANA unless the patient has objective features suggesting SLE.
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012; 64(8):2677–2686. doi:10.1002/art.34473
- Stanos S, Brodsky M, Argoff C, et al. Rethinking chronic pain in primary care setting. Postgraduate Med 2016; 128(5):502–513. doi:10.1080/00325481.2016.1188319
- Wolfe F, Clauw DJ, Fitzcharles M, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol 2011; 38(6):1113–1122. doi:10.3899/jrheum.100594
- Wolfe F, Clauw DJ, Fitzcharles MA, et al. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin Arthritis Rheum 2016; 46(3):319–329. doi:10.1016/j.semarthrit.2016.08.012
- Abeles AM, Abeles M. The clinical utility of a positive antinuclear antibody test result. Am J Med 2013; 126(4):342–348. doi:10.1016/j.amjmed.2012.09.014
- Li QZ, Karp DR, Quan J, et al. Risk factors for ANA positivity in healthy persons. Arthritis Res Ther 2011; 13(2):R38. doi:10.1186/ar3271
- Slight-Webb S, Lu R, Ritterhouse LL, et al. Autoantibody-positive healthy individuals display unique immune profiles that may regulate autoimmunity. Arthritis Rheumatol 2016; 68(1):2492–2502. doi:10.1002/art.39706
- Arbuckle MR, McClain MT, Rubertone MV, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med 2003; 349(16):1526–1533. doi:10.1056/NEJMoa021933
- Mariz HA, Sato EI, Barbosa SH, Rodrigues SH, Dellavance A, Andrade LE. Pattern of antinuclear antibody-HEp 2 test is a critical parameter for discriminating antinuclear antibody-positive healthy individuals and patients with autoimmune diseases. Arthritis Rheum 2011; 63(1):191–200. doi:10.1002/art.30084
- Häuser W, Clauw DJ, Fitzcharles MA. Treat to target in fibromyalgia: opening the dialogue. Arthritis Care Res (Hoboken) 2017; 69(4):462–466. doi:10.1002/acr.22970
- Forseth KO, Husby G, Gran JT, Fórre O. Prognostic factors for the development of fibromyalgia in women with self-reported musculoskeletal pain. A prospective study. J Rheumatol 1999; 26(11):2458–2567. pmid:10555910
- Thieme K, Turk DC, Flor H. Comorbid depression and anxiety in fibromyalgia syndrome: relationship to somatic and psychosocial variables. Psychosom Med 2004; 66(6):837–844. doi:10.1097/01.psy.0000146329.63158.40
- Fuller-Thomson E, Mimigon-Young J, Brennenstuhl S. Individuals with fibromyalgia and depression: findings from a nationally representative Canadian survey. Rheumatol Int 2012; 32(4):853–862. doi:10.1007/s00296-010-1713-x
- Kudlow PA, Rosenblat JD, Weissman CR, et al. Prevalence of fibromyalgia and co-morbid bipolar disorder: a systematic review and meta-analysis. J Affect Disord 2015; 188:134–142. doi:10.1016/j.jad.2015.08.030
- Corruble E, Guelfi JD. Pain complaints in depressed inpatients. Psychopathology 2000; 33(6):307–309. doi:10.1159/000029163
- Gota CE, Kaouk S, Wilke WS. The impact of depressive and bipolar symptoms on socioeconomic status, core symptoms, function and severity of fibromyalgia. Int J Rheum Dis 2017; 20(3):326–339. doi:10.1111/1756-185X.12603
- Toussaint LL, Vincent A, McAlister SJ, Oh TH, Hassett AL. A comparison of fibromyalgia symptoms in patients with healthy versus depressive, low and reactive affect balance styles. Scand J Pain 2014; 5(3):161–166. doi:10.1016/j.sjpain.2014.05.001
- Vincent A, Hoskin TL, Whipple MO, et al. OMERACT-based fibromyalgia symptom subgroups: an exploratory cluster analysis. Arthritis Res Ther 2014; 16(5):463. doi:10.1186/s13075-014-0463-7
- Robinson RL, Birnbaum HG, Morley MA, Sisitsky T, Greenberg PE, Wolfe F. Depression and fibromyalgia: treatment and cost when diagnosed separately or concurrently. J Rheumatol 2004; 31(8):1621–1629. pmid:5290744
- Choy EH. The role of sleep in pain and fibromyalgia. Nature Rev Rheumatol 2015; 11(9):513–520. doi:10.1038/nrrheum.2015.56
- Mork PJ, Nilsen TI. Sleep problems and risk of fibromyalgia: longitudinal data of an adult female population in Norway. Arthritis Rheum 2012; 64(1):281–284. doi:10.1002/art.33346
- McBeth J, Lacey RJ, Wilkie R. Predictors of new-onset widespread pain in older adults: results from a population-based prospective cohort study in the UK. Arthritis Rheumatol 2014; 66(3):757–767. doi:10.1002/art.38284
- Ablin JN, Clauw DJ, Lyden AK, et al. Effects of sleep restriction and exercise deprivation on somatic symptoms and mood in healthy adults. Clin Exp Rheumatol 2013; 31(6 suppl 79):S53–S59. pmid:24373363
- Russell IJ, Perkins AT, Michalek JE; Oxybate SXB-26 Fibromyalgia Syndrome Study Group. Sodium oxybate relieves pain and improves function in fibromyalgia syndrome: a randomized, double-blind, placebo-controlled, multicenter clinical trial. Arthritis Rheum 2009; 60(1):299–309. doi:10.1002/art.24142
- Spaeth M, Bennett RM, Benson BA, Wang YG, Lai C, Choy EH. Sodium oxybate therapy provides multidimensional improvement in fibromyalgia: results of an international phase 3 trial. Ann Rheum Dis 2012; 71(6):935–942. doi:10.1136/annrheumdis-2011-200418
- Roth T, Bhadra-Brown P, Pitman VW, Resnick EM. Pregabalin improves fibromyalgia-related sleep disturbance. Clin J Pain 2016; 32(4):308–312. doi:10.1097/AJP.0000000000000262
- Moldofsky H, Harris HW, Archambault WT, Kwong T, Lederman S. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol 2011; 38(12):2653–2663. doi:10.3899/jrheum.110194
- Feracioli-Oda E, Qawasmi A, Bloch MH. Meta-analysis: melatonin for the treatment of primary sleep disorders. PLoS One 2013; 8(5):e63773. doi:10.1371/journal.pone.0063773
- Ware MA, Fitzcharles MA, Joseph L, Shir Y. The effects of nabilone on sleep in fibromyalgia: results of a randomized controlled trial. Anesth Analg 2010; 110(2):604–610. doi:10.1213/ANE.0b013e3181c76f70
- Martínez M, Miró E, Sánchez AI, et al. Cognitive-behavioral therapy for insomnia and sleep hygiene in fibromyalgia: a randomized controlled trial. J Behav Med 2014; 37(4):683–697. doi:10.1007/s10865-013-9520-y
- Edwards RP, Cahalan C, Mensin G, Smith M, Haythornwaite JA. Pain, catastrophizing, and depression in the rheumatic diseases. Nature Rev Rheumatol 2011; 7(4):216–224. doi:10.1038/nrrheum.2011.2
- Sullivan MJL. The pain catastrophizing scale. User manual. McGill University, Montreal, Quebec, 2009. http://sullivan-painresearch.mcgill.ca/pdf/pcs/PCSManual_English.pdf. Accessed April 10, 2018.
- Louw A, Zimney K, O’Hotto C, Hilton S. The clinical application of teaching people about pain. Physiother Theory Pract 2016; 32(5):385–395. doi:10.1080/09593985.2016.1194652
- Burckhardt CS, Mannerkorpi K, Hedenberg L, Bjelle A. A randomized, controlled clinical trial of education and physical training for women with fibromyalgia. J Rheumatol 1994; 21(4):714–720. pmid:8035399
- King SJ, Wessel J, Bhambhani Y, Sholter D, Maksymowych W. The effects of exercise and education, individually or combined, in women with fibromyalgia. J Rheumatol 2002; 29(12):2620–2627. pmid:12465163
- Rooks DS, Gautam S, Romeling M, et al. Group exercise, education, and combination self-management in women with fibromyalgia: a randomized trial. Arch Intern Med 2007; 167(20):2192–2200. doi:10.1001/archinte.167.20.2192
- White KP, Nielson WR, Harth M, Ostbye T, Speechley M. Does the label ”fibromyalgia” alter health care status, function, and health service utilization? A prospective, within-group comparison in a community cohort of adults with chronic widespread pain. Arthritis Rheum 2002; 47(3):260–265. doi:10.1002/art.10400
- Bernardy K, Klose P, Busch AJ, Choy EH, Häuser W. Cognitive behavioural therapies for fibromyalgia. Cochrane Database Syst Rev 2013; 9:CD009796. doi:10.1002/14651858.CD009796.pub2
- Lazridou A, Kim J, Cahalan CM, et al. Effects of cognitive-behavioral therapy on brain connectivity supporting catastrophizing in fibromyalgia. Clin J Pain 2017; 33(3):215–221. doi:10.1097/AJP.0000000000000422
- Campbell CM, McCauley L, Bounds SC, et al. Changes in pain catastrophizing predict later changes in fibromyalgia clinical and experimental pain report: cross-lagged panel analyses of dispositional and situational catastrophizing. Arthritis Res Ther 2012; 14(5):R231. doi:10.1186/ar4073
- Vierck CJ. A mechanism-based approach to prevention of and therapy for fibromyalgia. Pain Res Treat 2012; 2012:951354. doi:10.1155/2012/951354
- Gerdle B, Ernberg M, Mannerkorpi K, et al. Increased interstitial concentrations of glutamate and pyruvate in vastus lateralis of women with fibromyalgia syndrome are normalized after exercise intervention—a case-control study. PLoS One 2016; 11(10):e0162010. doi:10.1371/journal.pone.0162010
- Jones J, Rutledge DN, Jones KD, Matallana L, Rooks DS. Self-assessed physical function levels of women with fibromyalgia: a national survey. Womens Health Issues 2008; 18(5):406–412. doi:10.1016/j.whi.2008.04.005
- Kelley GA, Kelley KS, Jones DL. Efficacy and effectiveness of exercise on tender points in adults with fibromyalgia: a meta-analysis of randomized controlled trials. Arthritis 2011; 2011:125485. doi:10.1155/2011/125485
- Valim V, Natour J, Ziao Y, et al. Effects of physical exercise on serum levels of serotonin and its metabolite in fibromyalgia: a randomized pilot study. Rev Bras Reumatol 2013; 53(6):538–541. doi:10.1016/j.rbr.2013.02.001
- Bidonde J, Busch AJ, Webber SC, et al. Aquatic exercise training for fibromyalgia. Cochrane Database Syst Rev 2014; 10:CD011336. doi:10.1002/14651858.CD011336
- Busch AJ, Webber SC, Richards RS, et al. Resistance exercise training for fibromyalgia. Cochrane Database Syst Rev 2013; 12:CD010884. doi:10.1002/14651858.CD010884
- Soriano-Maldonado A, Ruiz JR, Aparicio VA, et al. Association of physical fitness with pain in women with fibromyalgia: the al-Ándalus Project. Arthritis Care Res (Hoboken) 2015; 67(11):1561–1570. doi:10.1002/acr.22610
- Häuser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomized controlled trials. Arthritis Res Ther 2010; 12(3):R79. doi:10.1186/ar3002
- Häuser W, Perrot S, Clauw DJ, Fitzcharles MA. Unraveling fibromyalgia—steps towards individualized management. J Pain 2018; 19(2):125–134. doi:10.1016/j.jpain.2017.08.009
- Flodin P, Martinsen S, Mannerkorpi K, et al. Normalization of aberrant resting state functional connectivity in fibromyalgia patients following a three month physical exercise therapy. Neuroimage Clin 2015; 9:134–139. doi:10.1016/j.nicl.2015.08.004
- Dinoff A, Herrmann N, Swardfager W, et al. The effect of exercise training on resting concentrations of peripheral brain-derived neurotrophic factor (BDNF): a meta-analysis. PLOS One 2016. 11(9):e0163037. doi:10.1371/journal.pone.0163037
- Okifuji A, Gao J, Bokat C, Hare BD. Management of fibromyalgia syndrome in 2016. Pain Manag 2016; 6(4):383–400. doi:10.2217/pmt-2016-0006
- Masi AT, Vincent A. A historical and clinical perspective endorsing person-centered management of fibromyalgia syndrome. Curr Rheumatol Rev 2015; 11(2):86–95. pmid:26088217
- Fitzcharles MA, Ste-Marie PA, Goldenberg DL, et al; National Fibromyalgia Guideline Advisory Panel. 2012 Canadian guidelines for the diagnosis and management of fibromyalgia syndrome: executive summary. Pain Res Manag 2013: 18(3):119–126. pmid:23748251
- Ablin J, Fitzcharles MA, Buskila D, Shir Y, Sommer C, Häuser W. Treatment of fibromyalgia syndrome: recommendations of recent evidence-based interdisciplinary guidelines with special emphasis on complementary and alternative therapies. Evid Based Complement Alternat Med 2013; 2013:485272. doi:10.1155/2013/485272
- MacFarlane GJ, Kronisch C, Dean LE, et al. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis 2017; 76(2)318–328. doi:10.1136/annrheumdis-2016-209724
- Thieme K, Mathys M, Turk DC. Evidence-based guidelines on the treatment of fibromyalgia patients: are they consistent and if not, why not? Have effective psychological treatments been overlooked? J Pain 2017; 18(7):747–756. doi:10.1016/j.jpain.2016.12.006
- Williams DA, Kuper D, Segar M, Mohan N, Sheth M, Clauw DJ. Internet-enhanced management of fibromyalgia: a randomized controlled trial. Pain 2010; 151(3):694–702. doi:10.1016/j.pain.2010.08.034
Fibromyalgia may seem like a nebulous diagnosis, with its array of symptoms and pain refractory to medications. But fibromyalgia is a defined syndrome of neuronal dysregulation. It can be diagnosed from the history and physical examination and managed in a primary care setting.
CASE 1: MANY SYMPTOMS
A 43-year-old woman presents to her primary care physician with multiple complaints: pain in all joints and in her back and hips, swelling of her hands and feet, morning stiffness, chest pain and shortness of breath (not necessarily related to exertion), fatigue, generalized weakness, headaches, difficulty with memory and concentration, dry mouth and dry eyes, feeling weak and faint in the sun, cold intolerance with purple discoloration of her extremities, a self-described “butterfly” rash on the face, and hair that is thinning and falling out in clumps.
Because many of her symptoms could reflect an inflammatory process or an autoimmune disease,1 her primary care physician orders multiple tests. Her C-reactive protein level, Westergren sedimentation rate, complete blood cell count, and comprehensive metabolic panel are normal. Urinalysis shows trace leukocyte esterase. Indirect immunofluorescence assay on human laryngeal tumor (HEp-2) cells is positive for antinuclear antibody (ANA), with a titer of 1:320 (reference range ≤ 1:40) and a nuclear dense fine-speckled pattern.
In view of the positive ANA test, the patient is informed that she may have systemic lupus erythematosus (SLE) and will be referred to a rheumatologist. In the days before her rheumatology appointment, she becomes extremely anxious. Obsessively researching SLE online, she becomes convinced that SLE is the correct diagnosis.
Rheumatology evaluation
The rheumatologist assesses the patient’s pain and reports the following:
Location and duration: Hands, wrists, elbows, shoulders, upper and lower back, sides of hips, knees, and feet; has been ongoing for 10 years, but worse in the past 3 months.
Character: The patient describes her pain as “like an ice pick being driven through my joints,” “sometimes unbearable,” and “like being hit by a truck.” She also reports numb, tingly, burning pain in her upper neck and back.
Variation with time, activity, and weather: Worse at night, causing her to wake and toss and turn all night; better with exertion, but after activity or exercise, she is exhausted for the rest of the day and sometimes for up to a week; worse with weather changes, especially during cold or humid weather.
Associated symptoms: Occasional perceived swelling of hands and feet, especially upon wakening in the morning, and 2 to 3 hours of stiffness in the morning that sometimes lasts all day.
Physical examination. Her findings are inconsistent with her symptoms.
The patient exhibits limited range of motion. When asked to bend forward, rotate her neck, or flex and extend her neck and back, she does so only slightly. However, passive range of motion is normal in all joints.
When her joints are examined, she anticipates pain and withdraws her hands. But when she is distracted, examination reveals no evidence of swollen joints or synovitis. She has tenderness in 12 of 18 tender points. Her peripheral pulses are good, strength is normal, and reflexes are brisk.
Her facial rash looks more like rosacea than the butterfly rash of SLE. There is no indication of patchy hair loss. Heart and lung examinations are normal. She appears to have a good salivary pool without glossitis.
History reveals long-standing psychological issues
The patient reports a history of panic attacks and a prior diagnosis of anxiety. She is tested with the Generalized Anxiety Disorder 7-item scale (www.mdcalc.com/gad-7-general-anxiety-disorder-7) and scores 17 out of 21, indicating severe anxiety.
DISCUSSION: CHARACTERIZING PAIN
Understanding categories of pain syndromes can help us understand fibromyalgia. Pain can be categorized into 3 types that sometimes overlap2:
Nociceptive or peripheral pain is related to damage of tissue by trauma or inflammation. Syndromes include osteoarthritis, rheumatoid arthritis, and SLE.
Neuropathic pain is associated with damage of peripheral or central nerves. Examples are neuropathy from herpes, diabetes, or spinal stenosis.
Centralized pain has no identifiable nerve or tissue damage and is thought to result from persistent neuronal dysregulation, overactive ascending pain pathways, and a deficiency of descending inhibitory pain pathways. There is evidence of biochemical changes in muscles, possibly related to chronic ischemia and an overactive sympathetic nervous system. Dysregulation of the sympathoadrenal system and hypothalamic-pituitary axis has also been implicated. And genetic predisposition is possible. Examples of centralized pain syndromes include fibromyalgia, irritable bowel syndrome, pelvic pain syndrome, neurogenic bladder, and interstitial cystitis.
Clues to a centralized pain syndrome
For patients with suspected fibromyalgia, distinguishing peripheral pain from centralized pain can be a challenge (Table 1). For example, SLE does not cause inflammation of the spine, so neck or back pain is not typical. Although both nociceptive and centralized pain syndromes improve with exertion, only patients with centralized pain are typically exhausted and bedbound for days after activity. Patients with centralized pain tend to describe their pain in much more dramatic language than do patients with inflammatory pain. Centralized pain tends to be intermittent; inflammatory pain tends to be constant. Patients with centralized pain often have had pain for many years without a diagnosis, but this is rare for patients with an inflammatory condition.
A patient with fibromyalgia typically has a normal physical examination, although allodynia (experiencing pain from normally nonpainful stimulation), hyperalgesia (increased pain perception), and brisk reflexes may be present. Fibromyalgia may involve discoloration of the fingertips resulting from an overactive sympathetic nervous system. Laboratory results are typically normal with fibromyalgia.
Patients with either nociceptive or centralized pain report stiffness, but the cause likely differs. We typically think of stiffness as arising from swollen joints caused by inflammation, but stiffness can also be caused by abnormally tight muscles, as occurs in fibromyalgia.
FIBROMYALGIA IS A CLINICAL DIAGNOSIS
Diagnosing fibromyalgia does not require multiple laboratory and imaging tests. The key indicators are derived from the patient history and physical examination.
Diagnostic criteria for fibromyalgia published by the American College of Rheumatology have evolved over the years. The 2011 criteria, in the form of a self-reported patient questionnaire, have 2 components3:
- The Widespread Pain Index measures the extent of pain in 19 areas.
- The Symptom Severity scale assesses 3 key symptoms associated with fibromyalgia, ie, fatigue, cognitive problems, and nonrestorative sleep (scale of 0–3 for each symptom).
There are also questions about symptoms of irritable bowel syndrome, depression, and headache.
Fibromyalgia is diagnosed if a patient reports at least 7 painful areas and has a symptom severity score of at least 5. A patient may also meet the 20113 and the 2016 criteria4 if he or she has 4 painful areas and the pain is perceived in 4 of 5 areas and the Symptom Severity Scale score is 9 or higher.4 This questionnaire is not only a rapid diagnostic tool for fibromyalgia, it also helps identify and focus on specific issues—for example, having severe pain in a few localized areas, or having headache as a predominant problem.
These criteria are useful for a variety of patients, eg, a patient with hip arthritis may score high on the questionnaire, indicating that a component of centralized pain is also present. Also, people who have undergone orthopedic surgery who score high tend to require more narcotics to meet the goals of pain improvement.
The 2016 criteria,4 the most recent, maintain that pain must be generalized, ie, present in at least 4 of 5 body areas. They also emphasize that fibromyalgia is a valid diagnosis irrespective of other conditions.
CASE 1 CONTINUED: THE PATIENT REJECTS THE DIAGNOSIS
Our patient meets the definition of fibromyalgia by each iteration of the American College of Rheumatology clinical criteria. She also has generalized anxiety disorder and a positive ANA test. She is advised to participate in a fibromyalgia educational program, start an aerobic exercise program, and consider taking an antidepressant medication with anxiolytic effects.
However, the patient rejects the diagnosis of fibromyalgia. She believes that the diagnosis of SLE has been overlooked and that her symptom severity is being discounted.
In response, the rheumatologist orders additional tests to evaluate for an autoimmune disorder: extractable nuclear antigen panel, complement C3 and C4, double-stranded DNA antibodies, and protein electrophoresis. Results are all in the normal range. The patient is still concerned that she has SLE or another autoimmune disease because of her abnormal ANA test result and remains dissatisfied with her evaluation. She states that she will complain to the clinic ombudsman.
SIGNIFICANCE OF ANA TESTING
Patients with positive test results increasingly go online to get information. The significance of ANA testing can be confusing, and it is critical to understand and be able to explain abnormal results to worried patients. Following are answers to some common questions about ANA testing:
Is ANA positivity specific for SLE or another autoimmune disease?
No. ANA is usually tested by indirect immunofluorescence assay on HEp-2 cells. The test can identify about 150 antigens targeted by antibodies, but only a small percentage are associated with an autoimmune disease, and the others do not have a known clinical association. Enzyme-linked immunosorbent assay (ELISA) ANA testing is also available but is considered less sensitive.
Abeles and Abeles5 retrospectively assessed 232 patients between 2007 and 2009 who were referred to rheumatologists for evaluation because of an elevated ANA test result. No ANA-associated rheumatic disease was found in patients who had a result of less than 1:160, and more than 90% of referrals for a positive ANA test had no evidence of ANA-associated disease. The positive predictive value was 9% for any connective tissue disease, and only 2% for SLE. The most common reason for ordering the test was widespread pain (23%). The authors concluded that ANA testing is often ordered inappropriately for patients with a low pretest probability of an ANA-associated rheumatic disease.
Screening with ANA testing generates many false-positive results and unnecessary anxiety for patients. The prevalence of SLE in the general population is about 0.1%, and other autoimmune diseases total about 5% to 7%. By indirect immunofluorescence assay, using a cutoff of 1:80 (the standard at Cleveland Clinic), about 15% of the general population test positive. By ELISA, with a cutoff of 20 ELISA units, 25% of healthy controls test positive.
It is true that ANA positivity may precede the onset of SLE.6,7 Arbuckle et al8 evaluated serum samples from the US Department of Defense Serum Repository obtained from 130 people before they received a diagnosis of SLE; in 115 (88%), at least 1 SLE-related autoantibody was present before diagnosis (up to 9.4 years earlier). However, in those who test positive for ANA, the percentage who eventually develop autoimmune disease is small.5
Is the titer of ANA significant and of diagnostic value?
The likelihood of autoimmune disease increases with increasing titer. But high titers can be seen in healthy people. Mariz et al9 examined ANA test results from 918 healthy controls and 153 patients with an autoimmune rheumatic disease. Of these, ANA was positive in 13% of healthy people and 90% of patients with an autoimmune disease. High titers were more likely in patients with an autoimmune disease, but also occurred in healthy controls.
Does the immunofluorescence pattern provide diagnostic information?
It can. There are 28 identified patterns of ANA, including nuclear, cytoplasmic, and mitotic patterns. The most common, the nuclear fine-speckled pattern, is seen in healthy controls and patients with an autoimmune disease. But other patterns are either characteristic of an autoimmune disease or, conversely, of not having an autoimmune disease (Table 2).9
Our patient has a nuclear dense fine-speckled pattern, further reducing the likelihood that she has an autoimmune disease.
CASE 2: POORLY CONTROLLED,
LONG-STANDING FIBROMYALGIA
A 43-year-old woman who has had fibromyalgia for 15 years is referred to a new primary care provider. She reports severe pain all over, low back pain, fatigue, nonrefreshing sleep, chronic migraine, constipation alternating with diarrhea, heartburn, intermittent numbness and tingling in her hands and feet, and depression. At this time, she rates her pain on a visual analog scale as 9 out of 10, and her fatigue as 8 out of 10.
During the past 6 months, she has made 25 visits to specialists in 8 departments: spine, pain management, anesthesia, neurology, headache clinic, gastroenterology, sleep medicine, and physical therapy.
Her daily medications are duloxetine 120 mg, bupropion 300 mg, pregabalin 450 mg, cyclobenzaprine 30 mg, tramadol 200 mg, zolpidem 10 mg, nortriptyline 50 mg, acetaminophen 3,000 mg, and oxycodone 30 mg. She has also tried gabapentin and milnacipran without success. She reported previously taking different selective serotonin reuptake inhibitors and tricyclic antidepressants but cannot remember why they were stopped.
How should this complex patient be managed?
BIOPSYCHOSOCIAL MANAGEMENT
Managing the pain of fibromyalgia requires a different model than used for peripheral pain from injury, in which the source of pain can be identified and treated with injections or oral therapy.
Neuronal dysregulation is not amenable to clinical measurement or treatment by medications at this time. But fortunately, many factors associated with fibromyalgia can be addressed: stressful life events, sleep disturbance, physical deconditioning, mood disorders, and maladaptive pain responses, including “catastrophizing” behavior (coping with pain in a highly dramatic and obsessive way). Modifying these factors can be much more productive than focusing on treating pain.
The goal for care providers is to change the focus from reducing pain to a biopsychosocial model of pain control aimed at increasing function.10
Mood modification
Not only are mood disorders common in patients with fibromyalgia, but the prevalence of complex psychiatric conditions is also elevated. Up to 80% of patients with fibromyalgia meet criteria for axis I (clinical psychological) disorders, and up to about 30% of patients meet criteria for axis II (personality) disorders. About 22% of patients have existing major depression, and about 58% develop it during their lifetime. In a study of 678 patients with fibromyalgia, 21% had bipolar disorder.11–15
The severity of fibromyalgia increases linearly with the severity of depression.16 Patients with fibromyalgia and a “depressive affect balance style” have worse outcomes across all Outcome Measures in Rheumatology (OMERACT) core symptom domains, reporting more pain, fatigue, insomnia, anxiety, depression, and function.17,18
Fibromyalgia combined with mood disorders can also be costly. In one study, the mean annual employer payments (direct and indirect costs) per patient were $5,200 for patients with fibromyalgia only, $8,100 for patients with depression only, and $11,900 for patients with both.19
Obtaining a psychiatric history is important when evaluating a patient with fibromyalgia symptoms. Patients should be asked if they have a history of depression, anxiety, posttraumatic stress disorder, or other conditions. The Patient Health Questionnaire – Depression 9 and the Generalized Anxiety Disorder Assessment (GAD-7) (both available at www.mdcalc.com) can be useful for evaluating mood disorders.
Patients with moderate depression and fibromyalgia who have not yet been treated should be prescribed duloxetine for its potential benefits for both conditions.
Patients who have already been treated with multiple drugs at high doses without benefit, such as our patient, should be referred to a psychiatrist. There is no additional benefit to referring this patient to a rheumatologist or spine clinic.
Addressing sleep problems
Sleep problems are not easy to manage but can often be helped. Epidemiologic studies indicate that poor sleep quality leads to chronic widespread pain in otherwise healthy people.20–22 In addition, experimental sleep deprivation leads to fatigue, cognitive difficulty, and a reduced pain threshold.23 In our patients with fibromyalgia, we have observed an inverse relationship between the number of hours slept and the severity of depression.
Sleep quantity and quality can be assessed by asking patients whether they have trouble sleeping, how many hours they sleep, and whether they have been diagnosed with a sleep disorder.
Because many patients with fibromyalgia are overweight or obese, they should also be evaluated for sleep apnea, narcolepsy, and restless leg syndrome.24,25
Medications shown to improve sleep include pregabalin or gabapentin (taken at bedtime), low-dose amitriptyline, trazodone, cyclobenzaprine, melatonin, and nabilone.26–29
Patients should be counseled about sleep hygiene.30 Exercise can also help sleep.
Targeting maladaptive pain responses
Patients who catastrophize tend to have higher tender point counts, a hyperalgesic response, more depression and anxiety, and more self-reported disability. They are also less likely to return to work.31 They usually respond poorly to medications and are good candidates for cognitive behavioral therapy.
A high score on a self-reported Pain Catastrophizing Scale32 can help determine whether a multidisciplinary approach is advisable, although no threshold defines an abnormal score.
Educating patients about the neurobiology underlying their pain can be therapeutic.33–37 Cognitive behavioral therapy can help patients recognize their faulty thought processes and the relationship between pain and stress, and learn better coping mechanisms.38,39 Patients who achieve the highest improvements in pain catastrophizing tend to derive the greatest benefit from cognitive behavioral therapy.40
Exercise improves symptoms
Exercise improves fibromyalgia on many fronts and is associated with a host of positive effects in the brain and peripheral muscles. Exercise improves Fibromyalgia Impact Questionnaire scores, increases physical function and fitness, and reduces tender point counts, depression, and catastrophizing.41–52 There is no consensus on the best type of exercise, but both strengthening and aerobic exercises appear to be important.
I tell patients that fibromyalgia is an exercise-deprivation syndrome. Many are afraid to exercise because they associate it with pain and exhaustion afterwards. Patients should be encouraged to start with something very low-impact, such as gentle exercise in a warm-water pool. It should be emphasized that exercise is a lifelong treatment.
Drug therapy
The US Food and Drug Administration has approved 3 drugs for fibromyalgia management: 2 serotonin-norepinephrine reuptake inhibitors (duloxetine and milnacipran) and 1 gabapentinoid (pregabalin). Our patient in Case 2 is taking 2 of them without apparent benefit and has previously had no success with the third. This is not surprising. A summary of published treatment research on these drugs found that only 50% to 60% of patients tested reported more than 30% pain reduction.53 The studies also showed a placebo response of 30% to 40%. Depending on the study, the number needed to treat to see a benefit from these drugs is 8 to 14.53
EVALUATING THE SEVERITY OF FIBROMYALGIA
Focusing on key characteristics of the patient’s history can help evaluate fibromyalgia and determine a treatment strategy (Table 3). The Fibromyalgia Impact Questionnaire is also a useful evaluation tool.
It is important to assess the severity of fibromyalgia because patients with severe fibromyalgia are not good candidates for further referral to other specialists. They instead need chronic rehabilitation services, where they can learn to better function with a chronic pain syndrome.
In general, patients with the following features have conditions with high severity:
Symptoms: High burden and intensity
Function: Disabled, unemployed, interference with activities of daily living
Mood: Severe depression, bipolar disorder, axis II disorder, posttraumatic stress disorder
Medications: Polypharmacy, opioid drugs, multiple failed interventions
Maladaptive attitudes: High catastrophizing, refusal to accept diagnosis
Fibromyalgia Impact Questionnaire score: 60 or above.
The fibromyalgia of our patient in Case 2 would be categorized as severe.
MULTIFACETED MANAGEMENT
Patients with fibromyalgia are a heterogeneous group, and the syndrome does not lend itself to a single management strategy.54 Multiple guidelines have been published for managing fibromyalgia.55–57 Thieme et al58 reviewed existing guidelines and the strength of their recommendations. The guidelines unanimously strongly favor exercise, and most also strongly favor cognitive behavioral therapy. Most favor treating with amitriptyline and duloxetine; recommendations for other antidepressants vary. Nonsteroidal anti-inflammatory drugs, opioid drugs, and benzodiazepines are not recommended.
We offer a monthly 1-day clinic for patients and family members to provide education about fibromyalgia, discuss the importance of exercise, counsel on maladaptive responses, and demonstrate mindfulness techniques. We focus on function rather than pain. Interactive online-based interventions using cognitive behavioral techniques, such as FibroGuide: A Symptom Management Program for People Living With Fibromyalgia, developed at the University of Michigan, have proven helpful.59
RECOMMENDATIONS
For most patients, do not focus on pain reduction, as that is ineffective. Instead, target reversible factors, eg, mood, sleep, exercise status, stressors, and maladaptive attitudes toward pain. Possible treatment combinations include:
- A serotonin and norepinephrine reuptake inhibitor (eg, duloxetine)
- A low-dose tricyclic antidepressant at bedtime (eg, amitriptyline)
- A gabapentinoid (pregabalin or gabapentin).
If a medication within a class does not work, stop it and try another rather than add on.
Treat mild to moderate fibromyalgia with multidisciplinary interventions, with or without centrally acting medications. Treat severe fibromyalgia with more intensive psychiatric or psychologic interventions, multidisciplinary care, and medications targeted at comorbidities. Provide all patients with education and advice on exercise.
Keep laboratory tests and imaging studies to a minimum: a complete blood cell count with differential, comprehensive metabolic panel, thyroid-stimulating hormone, C-reactive protein, and Westergren sedimentation rate. Do not test for ANA unless the patient has objective features suggesting SLE.
Fibromyalgia may seem like a nebulous diagnosis, with its array of symptoms and pain refractory to medications. But fibromyalgia is a defined syndrome of neuronal dysregulation. It can be diagnosed from the history and physical examination and managed in a primary care setting.
CASE 1: MANY SYMPTOMS
A 43-year-old woman presents to her primary care physician with multiple complaints: pain in all joints and in her back and hips, swelling of her hands and feet, morning stiffness, chest pain and shortness of breath (not necessarily related to exertion), fatigue, generalized weakness, headaches, difficulty with memory and concentration, dry mouth and dry eyes, feeling weak and faint in the sun, cold intolerance with purple discoloration of her extremities, a self-described “butterfly” rash on the face, and hair that is thinning and falling out in clumps.
Because many of her symptoms could reflect an inflammatory process or an autoimmune disease,1 her primary care physician orders multiple tests. Her C-reactive protein level, Westergren sedimentation rate, complete blood cell count, and comprehensive metabolic panel are normal. Urinalysis shows trace leukocyte esterase. Indirect immunofluorescence assay on human laryngeal tumor (HEp-2) cells is positive for antinuclear antibody (ANA), with a titer of 1:320 (reference range ≤ 1:40) and a nuclear dense fine-speckled pattern.
In view of the positive ANA test, the patient is informed that she may have systemic lupus erythematosus (SLE) and will be referred to a rheumatologist. In the days before her rheumatology appointment, she becomes extremely anxious. Obsessively researching SLE online, she becomes convinced that SLE is the correct diagnosis.
Rheumatology evaluation
The rheumatologist assesses the patient’s pain and reports the following:
Location and duration: Hands, wrists, elbows, shoulders, upper and lower back, sides of hips, knees, and feet; has been ongoing for 10 years, but worse in the past 3 months.
Character: The patient describes her pain as “like an ice pick being driven through my joints,” “sometimes unbearable,” and “like being hit by a truck.” She also reports numb, tingly, burning pain in her upper neck and back.
Variation with time, activity, and weather: Worse at night, causing her to wake and toss and turn all night; better with exertion, but after activity or exercise, she is exhausted for the rest of the day and sometimes for up to a week; worse with weather changes, especially during cold or humid weather.
Associated symptoms: Occasional perceived swelling of hands and feet, especially upon wakening in the morning, and 2 to 3 hours of stiffness in the morning that sometimes lasts all day.
Physical examination. Her findings are inconsistent with her symptoms.
The patient exhibits limited range of motion. When asked to bend forward, rotate her neck, or flex and extend her neck and back, she does so only slightly. However, passive range of motion is normal in all joints.
When her joints are examined, she anticipates pain and withdraws her hands. But when she is distracted, examination reveals no evidence of swollen joints or synovitis. She has tenderness in 12 of 18 tender points. Her peripheral pulses are good, strength is normal, and reflexes are brisk.
Her facial rash looks more like rosacea than the butterfly rash of SLE. There is no indication of patchy hair loss. Heart and lung examinations are normal. She appears to have a good salivary pool without glossitis.
History reveals long-standing psychological issues
The patient reports a history of panic attacks and a prior diagnosis of anxiety. She is tested with the Generalized Anxiety Disorder 7-item scale (www.mdcalc.com/gad-7-general-anxiety-disorder-7) and scores 17 out of 21, indicating severe anxiety.
DISCUSSION: CHARACTERIZING PAIN
Understanding categories of pain syndromes can help us understand fibromyalgia. Pain can be categorized into 3 types that sometimes overlap2:
Nociceptive or peripheral pain is related to damage of tissue by trauma or inflammation. Syndromes include osteoarthritis, rheumatoid arthritis, and SLE.
Neuropathic pain is associated with damage of peripheral or central nerves. Examples are neuropathy from herpes, diabetes, or spinal stenosis.
Centralized pain has no identifiable nerve or tissue damage and is thought to result from persistent neuronal dysregulation, overactive ascending pain pathways, and a deficiency of descending inhibitory pain pathways. There is evidence of biochemical changes in muscles, possibly related to chronic ischemia and an overactive sympathetic nervous system. Dysregulation of the sympathoadrenal system and hypothalamic-pituitary axis has also been implicated. And genetic predisposition is possible. Examples of centralized pain syndromes include fibromyalgia, irritable bowel syndrome, pelvic pain syndrome, neurogenic bladder, and interstitial cystitis.
Clues to a centralized pain syndrome
For patients with suspected fibromyalgia, distinguishing peripheral pain from centralized pain can be a challenge (Table 1). For example, SLE does not cause inflammation of the spine, so neck or back pain is not typical. Although both nociceptive and centralized pain syndromes improve with exertion, only patients with centralized pain are typically exhausted and bedbound for days after activity. Patients with centralized pain tend to describe their pain in much more dramatic language than do patients with inflammatory pain. Centralized pain tends to be intermittent; inflammatory pain tends to be constant. Patients with centralized pain often have had pain for many years without a diagnosis, but this is rare for patients with an inflammatory condition.
A patient with fibromyalgia typically has a normal physical examination, although allodynia (experiencing pain from normally nonpainful stimulation), hyperalgesia (increased pain perception), and brisk reflexes may be present. Fibromyalgia may involve discoloration of the fingertips resulting from an overactive sympathetic nervous system. Laboratory results are typically normal with fibromyalgia.
Patients with either nociceptive or centralized pain report stiffness, but the cause likely differs. We typically think of stiffness as arising from swollen joints caused by inflammation, but stiffness can also be caused by abnormally tight muscles, as occurs in fibromyalgia.
FIBROMYALGIA IS A CLINICAL DIAGNOSIS
Diagnosing fibromyalgia does not require multiple laboratory and imaging tests. The key indicators are derived from the patient history and physical examination.
Diagnostic criteria for fibromyalgia published by the American College of Rheumatology have evolved over the years. The 2011 criteria, in the form of a self-reported patient questionnaire, have 2 components3:
- The Widespread Pain Index measures the extent of pain in 19 areas.
- The Symptom Severity scale assesses 3 key symptoms associated with fibromyalgia, ie, fatigue, cognitive problems, and nonrestorative sleep (scale of 0–3 for each symptom).
There are also questions about symptoms of irritable bowel syndrome, depression, and headache.
Fibromyalgia is diagnosed if a patient reports at least 7 painful areas and has a symptom severity score of at least 5. A patient may also meet the 20113 and the 2016 criteria4 if he or she has 4 painful areas and the pain is perceived in 4 of 5 areas and the Symptom Severity Scale score is 9 or higher.4 This questionnaire is not only a rapid diagnostic tool for fibromyalgia, it also helps identify and focus on specific issues—for example, having severe pain in a few localized areas, or having headache as a predominant problem.
These criteria are useful for a variety of patients, eg, a patient with hip arthritis may score high on the questionnaire, indicating that a component of centralized pain is also present. Also, people who have undergone orthopedic surgery who score high tend to require more narcotics to meet the goals of pain improvement.
The 2016 criteria,4 the most recent, maintain that pain must be generalized, ie, present in at least 4 of 5 body areas. They also emphasize that fibromyalgia is a valid diagnosis irrespective of other conditions.
CASE 1 CONTINUED: THE PATIENT REJECTS THE DIAGNOSIS
Our patient meets the definition of fibromyalgia by each iteration of the American College of Rheumatology clinical criteria. She also has generalized anxiety disorder and a positive ANA test. She is advised to participate in a fibromyalgia educational program, start an aerobic exercise program, and consider taking an antidepressant medication with anxiolytic effects.
However, the patient rejects the diagnosis of fibromyalgia. She believes that the diagnosis of SLE has been overlooked and that her symptom severity is being discounted.
In response, the rheumatologist orders additional tests to evaluate for an autoimmune disorder: extractable nuclear antigen panel, complement C3 and C4, double-stranded DNA antibodies, and protein electrophoresis. Results are all in the normal range. The patient is still concerned that she has SLE or another autoimmune disease because of her abnormal ANA test result and remains dissatisfied with her evaluation. She states that she will complain to the clinic ombudsman.
SIGNIFICANCE OF ANA TESTING
Patients with positive test results increasingly go online to get information. The significance of ANA testing can be confusing, and it is critical to understand and be able to explain abnormal results to worried patients. Following are answers to some common questions about ANA testing:
Is ANA positivity specific for SLE or another autoimmune disease?
No. ANA is usually tested by indirect immunofluorescence assay on HEp-2 cells. The test can identify about 150 antigens targeted by antibodies, but only a small percentage are associated with an autoimmune disease, and the others do not have a known clinical association. Enzyme-linked immunosorbent assay (ELISA) ANA testing is also available but is considered less sensitive.
Abeles and Abeles5 retrospectively assessed 232 patients between 2007 and 2009 who were referred to rheumatologists for evaluation because of an elevated ANA test result. No ANA-associated rheumatic disease was found in patients who had a result of less than 1:160, and more than 90% of referrals for a positive ANA test had no evidence of ANA-associated disease. The positive predictive value was 9% for any connective tissue disease, and only 2% for SLE. The most common reason for ordering the test was widespread pain (23%). The authors concluded that ANA testing is often ordered inappropriately for patients with a low pretest probability of an ANA-associated rheumatic disease.
Screening with ANA testing generates many false-positive results and unnecessary anxiety for patients. The prevalence of SLE in the general population is about 0.1%, and other autoimmune diseases total about 5% to 7%. By indirect immunofluorescence assay, using a cutoff of 1:80 (the standard at Cleveland Clinic), about 15% of the general population test positive. By ELISA, with a cutoff of 20 ELISA units, 25% of healthy controls test positive.
It is true that ANA positivity may precede the onset of SLE.6,7 Arbuckle et al8 evaluated serum samples from the US Department of Defense Serum Repository obtained from 130 people before they received a diagnosis of SLE; in 115 (88%), at least 1 SLE-related autoantibody was present before diagnosis (up to 9.4 years earlier). However, in those who test positive for ANA, the percentage who eventually develop autoimmune disease is small.5
Is the titer of ANA significant and of diagnostic value?
The likelihood of autoimmune disease increases with increasing titer. But high titers can be seen in healthy people. Mariz et al9 examined ANA test results from 918 healthy controls and 153 patients with an autoimmune rheumatic disease. Of these, ANA was positive in 13% of healthy people and 90% of patients with an autoimmune disease. High titers were more likely in patients with an autoimmune disease, but also occurred in healthy controls.
Does the immunofluorescence pattern provide diagnostic information?
It can. There are 28 identified patterns of ANA, including nuclear, cytoplasmic, and mitotic patterns. The most common, the nuclear fine-speckled pattern, is seen in healthy controls and patients with an autoimmune disease. But other patterns are either characteristic of an autoimmune disease or, conversely, of not having an autoimmune disease (Table 2).9
Our patient has a nuclear dense fine-speckled pattern, further reducing the likelihood that she has an autoimmune disease.
CASE 2: POORLY CONTROLLED,
LONG-STANDING FIBROMYALGIA
A 43-year-old woman who has had fibromyalgia for 15 years is referred to a new primary care provider. She reports severe pain all over, low back pain, fatigue, nonrefreshing sleep, chronic migraine, constipation alternating with diarrhea, heartburn, intermittent numbness and tingling in her hands and feet, and depression. At this time, she rates her pain on a visual analog scale as 9 out of 10, and her fatigue as 8 out of 10.
During the past 6 months, she has made 25 visits to specialists in 8 departments: spine, pain management, anesthesia, neurology, headache clinic, gastroenterology, sleep medicine, and physical therapy.
Her daily medications are duloxetine 120 mg, bupropion 300 mg, pregabalin 450 mg, cyclobenzaprine 30 mg, tramadol 200 mg, zolpidem 10 mg, nortriptyline 50 mg, acetaminophen 3,000 mg, and oxycodone 30 mg. She has also tried gabapentin and milnacipran without success. She reported previously taking different selective serotonin reuptake inhibitors and tricyclic antidepressants but cannot remember why they were stopped.
How should this complex patient be managed?
BIOPSYCHOSOCIAL MANAGEMENT
Managing the pain of fibromyalgia requires a different model than used for peripheral pain from injury, in which the source of pain can be identified and treated with injections or oral therapy.
Neuronal dysregulation is not amenable to clinical measurement or treatment by medications at this time. But fortunately, many factors associated with fibromyalgia can be addressed: stressful life events, sleep disturbance, physical deconditioning, mood disorders, and maladaptive pain responses, including “catastrophizing” behavior (coping with pain in a highly dramatic and obsessive way). Modifying these factors can be much more productive than focusing on treating pain.
The goal for care providers is to change the focus from reducing pain to a biopsychosocial model of pain control aimed at increasing function.10
Mood modification
Not only are mood disorders common in patients with fibromyalgia, but the prevalence of complex psychiatric conditions is also elevated. Up to 80% of patients with fibromyalgia meet criteria for axis I (clinical psychological) disorders, and up to about 30% of patients meet criteria for axis II (personality) disorders. About 22% of patients have existing major depression, and about 58% develop it during their lifetime. In a study of 678 patients with fibromyalgia, 21% had bipolar disorder.11–15
The severity of fibromyalgia increases linearly with the severity of depression.16 Patients with fibromyalgia and a “depressive affect balance style” have worse outcomes across all Outcome Measures in Rheumatology (OMERACT) core symptom domains, reporting more pain, fatigue, insomnia, anxiety, depression, and function.17,18
Fibromyalgia combined with mood disorders can also be costly. In one study, the mean annual employer payments (direct and indirect costs) per patient were $5,200 for patients with fibromyalgia only, $8,100 for patients with depression only, and $11,900 for patients with both.19
Obtaining a psychiatric history is important when evaluating a patient with fibromyalgia symptoms. Patients should be asked if they have a history of depression, anxiety, posttraumatic stress disorder, or other conditions. The Patient Health Questionnaire – Depression 9 and the Generalized Anxiety Disorder Assessment (GAD-7) (both available at www.mdcalc.com) can be useful for evaluating mood disorders.
Patients with moderate depression and fibromyalgia who have not yet been treated should be prescribed duloxetine for its potential benefits for both conditions.
Patients who have already been treated with multiple drugs at high doses without benefit, such as our patient, should be referred to a psychiatrist. There is no additional benefit to referring this patient to a rheumatologist or spine clinic.
Addressing sleep problems
Sleep problems are not easy to manage but can often be helped. Epidemiologic studies indicate that poor sleep quality leads to chronic widespread pain in otherwise healthy people.20–22 In addition, experimental sleep deprivation leads to fatigue, cognitive difficulty, and a reduced pain threshold.23 In our patients with fibromyalgia, we have observed an inverse relationship between the number of hours slept and the severity of depression.
Sleep quantity and quality can be assessed by asking patients whether they have trouble sleeping, how many hours they sleep, and whether they have been diagnosed with a sleep disorder.
Because many patients with fibromyalgia are overweight or obese, they should also be evaluated for sleep apnea, narcolepsy, and restless leg syndrome.24,25
Medications shown to improve sleep include pregabalin or gabapentin (taken at bedtime), low-dose amitriptyline, trazodone, cyclobenzaprine, melatonin, and nabilone.26–29
Patients should be counseled about sleep hygiene.30 Exercise can also help sleep.
Targeting maladaptive pain responses
Patients who catastrophize tend to have higher tender point counts, a hyperalgesic response, more depression and anxiety, and more self-reported disability. They are also less likely to return to work.31 They usually respond poorly to medications and are good candidates for cognitive behavioral therapy.
A high score on a self-reported Pain Catastrophizing Scale32 can help determine whether a multidisciplinary approach is advisable, although no threshold defines an abnormal score.
Educating patients about the neurobiology underlying their pain can be therapeutic.33–37 Cognitive behavioral therapy can help patients recognize their faulty thought processes and the relationship between pain and stress, and learn better coping mechanisms.38,39 Patients who achieve the highest improvements in pain catastrophizing tend to derive the greatest benefit from cognitive behavioral therapy.40
Exercise improves symptoms
Exercise improves fibromyalgia on many fronts and is associated with a host of positive effects in the brain and peripheral muscles. Exercise improves Fibromyalgia Impact Questionnaire scores, increases physical function and fitness, and reduces tender point counts, depression, and catastrophizing.41–52 There is no consensus on the best type of exercise, but both strengthening and aerobic exercises appear to be important.
I tell patients that fibromyalgia is an exercise-deprivation syndrome. Many are afraid to exercise because they associate it with pain and exhaustion afterwards. Patients should be encouraged to start with something very low-impact, such as gentle exercise in a warm-water pool. It should be emphasized that exercise is a lifelong treatment.
Drug therapy
The US Food and Drug Administration has approved 3 drugs for fibromyalgia management: 2 serotonin-norepinephrine reuptake inhibitors (duloxetine and milnacipran) and 1 gabapentinoid (pregabalin). Our patient in Case 2 is taking 2 of them without apparent benefit and has previously had no success with the third. This is not surprising. A summary of published treatment research on these drugs found that only 50% to 60% of patients tested reported more than 30% pain reduction.53 The studies also showed a placebo response of 30% to 40%. Depending on the study, the number needed to treat to see a benefit from these drugs is 8 to 14.53
EVALUATING THE SEVERITY OF FIBROMYALGIA
Focusing on key characteristics of the patient’s history can help evaluate fibromyalgia and determine a treatment strategy (Table 3). The Fibromyalgia Impact Questionnaire is also a useful evaluation tool.
It is important to assess the severity of fibromyalgia because patients with severe fibromyalgia are not good candidates for further referral to other specialists. They instead need chronic rehabilitation services, where they can learn to better function with a chronic pain syndrome.
In general, patients with the following features have conditions with high severity:
Symptoms: High burden and intensity
Function: Disabled, unemployed, interference with activities of daily living
Mood: Severe depression, bipolar disorder, axis II disorder, posttraumatic stress disorder
Medications: Polypharmacy, opioid drugs, multiple failed interventions
Maladaptive attitudes: High catastrophizing, refusal to accept diagnosis
Fibromyalgia Impact Questionnaire score: 60 or above.
The fibromyalgia of our patient in Case 2 would be categorized as severe.
MULTIFACETED MANAGEMENT
Patients with fibromyalgia are a heterogeneous group, and the syndrome does not lend itself to a single management strategy.54 Multiple guidelines have been published for managing fibromyalgia.55–57 Thieme et al58 reviewed existing guidelines and the strength of their recommendations. The guidelines unanimously strongly favor exercise, and most also strongly favor cognitive behavioral therapy. Most favor treating with amitriptyline and duloxetine; recommendations for other antidepressants vary. Nonsteroidal anti-inflammatory drugs, opioid drugs, and benzodiazepines are not recommended.
We offer a monthly 1-day clinic for patients and family members to provide education about fibromyalgia, discuss the importance of exercise, counsel on maladaptive responses, and demonstrate mindfulness techniques. We focus on function rather than pain. Interactive online-based interventions using cognitive behavioral techniques, such as FibroGuide: A Symptom Management Program for People Living With Fibromyalgia, developed at the University of Michigan, have proven helpful.59
RECOMMENDATIONS
For most patients, do not focus on pain reduction, as that is ineffective. Instead, target reversible factors, eg, mood, sleep, exercise status, stressors, and maladaptive attitudes toward pain. Possible treatment combinations include:
- A serotonin and norepinephrine reuptake inhibitor (eg, duloxetine)
- A low-dose tricyclic antidepressant at bedtime (eg, amitriptyline)
- A gabapentinoid (pregabalin or gabapentin).
If a medication within a class does not work, stop it and try another rather than add on.
Treat mild to moderate fibromyalgia with multidisciplinary interventions, with or without centrally acting medications. Treat severe fibromyalgia with more intensive psychiatric or psychologic interventions, multidisciplinary care, and medications targeted at comorbidities. Provide all patients with education and advice on exercise.
Keep laboratory tests and imaging studies to a minimum: a complete blood cell count with differential, comprehensive metabolic panel, thyroid-stimulating hormone, C-reactive protein, and Westergren sedimentation rate. Do not test for ANA unless the patient has objective features suggesting SLE.
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012; 64(8):2677–2686. doi:10.1002/art.34473
- Stanos S, Brodsky M, Argoff C, et al. Rethinking chronic pain in primary care setting. Postgraduate Med 2016; 128(5):502–513. doi:10.1080/00325481.2016.1188319
- Wolfe F, Clauw DJ, Fitzcharles M, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol 2011; 38(6):1113–1122. doi:10.3899/jrheum.100594
- Wolfe F, Clauw DJ, Fitzcharles MA, et al. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin Arthritis Rheum 2016; 46(3):319–329. doi:10.1016/j.semarthrit.2016.08.012
- Abeles AM, Abeles M. The clinical utility of a positive antinuclear antibody test result. Am J Med 2013; 126(4):342–348. doi:10.1016/j.amjmed.2012.09.014
- Li QZ, Karp DR, Quan J, et al. Risk factors for ANA positivity in healthy persons. Arthritis Res Ther 2011; 13(2):R38. doi:10.1186/ar3271
- Slight-Webb S, Lu R, Ritterhouse LL, et al. Autoantibody-positive healthy individuals display unique immune profiles that may regulate autoimmunity. Arthritis Rheumatol 2016; 68(1):2492–2502. doi:10.1002/art.39706
- Arbuckle MR, McClain MT, Rubertone MV, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med 2003; 349(16):1526–1533. doi:10.1056/NEJMoa021933
- Mariz HA, Sato EI, Barbosa SH, Rodrigues SH, Dellavance A, Andrade LE. Pattern of antinuclear antibody-HEp 2 test is a critical parameter for discriminating antinuclear antibody-positive healthy individuals and patients with autoimmune diseases. Arthritis Rheum 2011; 63(1):191–200. doi:10.1002/art.30084
- Häuser W, Clauw DJ, Fitzcharles MA. Treat to target in fibromyalgia: opening the dialogue. Arthritis Care Res (Hoboken) 2017; 69(4):462–466. doi:10.1002/acr.22970
- Forseth KO, Husby G, Gran JT, Fórre O. Prognostic factors for the development of fibromyalgia in women with self-reported musculoskeletal pain. A prospective study. J Rheumatol 1999; 26(11):2458–2567. pmid:10555910
- Thieme K, Turk DC, Flor H. Comorbid depression and anxiety in fibromyalgia syndrome: relationship to somatic and psychosocial variables. Psychosom Med 2004; 66(6):837–844. doi:10.1097/01.psy.0000146329.63158.40
- Fuller-Thomson E, Mimigon-Young J, Brennenstuhl S. Individuals with fibromyalgia and depression: findings from a nationally representative Canadian survey. Rheumatol Int 2012; 32(4):853–862. doi:10.1007/s00296-010-1713-x
- Kudlow PA, Rosenblat JD, Weissman CR, et al. Prevalence of fibromyalgia and co-morbid bipolar disorder: a systematic review and meta-analysis. J Affect Disord 2015; 188:134–142. doi:10.1016/j.jad.2015.08.030
- Corruble E, Guelfi JD. Pain complaints in depressed inpatients. Psychopathology 2000; 33(6):307–309. doi:10.1159/000029163
- Gota CE, Kaouk S, Wilke WS. The impact of depressive and bipolar symptoms on socioeconomic status, core symptoms, function and severity of fibromyalgia. Int J Rheum Dis 2017; 20(3):326–339. doi:10.1111/1756-185X.12603
- Toussaint LL, Vincent A, McAlister SJ, Oh TH, Hassett AL. A comparison of fibromyalgia symptoms in patients with healthy versus depressive, low and reactive affect balance styles. Scand J Pain 2014; 5(3):161–166. doi:10.1016/j.sjpain.2014.05.001
- Vincent A, Hoskin TL, Whipple MO, et al. OMERACT-based fibromyalgia symptom subgroups: an exploratory cluster analysis. Arthritis Res Ther 2014; 16(5):463. doi:10.1186/s13075-014-0463-7
- Robinson RL, Birnbaum HG, Morley MA, Sisitsky T, Greenberg PE, Wolfe F. Depression and fibromyalgia: treatment and cost when diagnosed separately or concurrently. J Rheumatol 2004; 31(8):1621–1629. pmid:5290744
- Choy EH. The role of sleep in pain and fibromyalgia. Nature Rev Rheumatol 2015; 11(9):513–520. doi:10.1038/nrrheum.2015.56
- Mork PJ, Nilsen TI. Sleep problems and risk of fibromyalgia: longitudinal data of an adult female population in Norway. Arthritis Rheum 2012; 64(1):281–284. doi:10.1002/art.33346
- McBeth J, Lacey RJ, Wilkie R. Predictors of new-onset widespread pain in older adults: results from a population-based prospective cohort study in the UK. Arthritis Rheumatol 2014; 66(3):757–767. doi:10.1002/art.38284
- Ablin JN, Clauw DJ, Lyden AK, et al. Effects of sleep restriction and exercise deprivation on somatic symptoms and mood in healthy adults. Clin Exp Rheumatol 2013; 31(6 suppl 79):S53–S59. pmid:24373363
- Russell IJ, Perkins AT, Michalek JE; Oxybate SXB-26 Fibromyalgia Syndrome Study Group. Sodium oxybate relieves pain and improves function in fibromyalgia syndrome: a randomized, double-blind, placebo-controlled, multicenter clinical trial. Arthritis Rheum 2009; 60(1):299–309. doi:10.1002/art.24142
- Spaeth M, Bennett RM, Benson BA, Wang YG, Lai C, Choy EH. Sodium oxybate therapy provides multidimensional improvement in fibromyalgia: results of an international phase 3 trial. Ann Rheum Dis 2012; 71(6):935–942. doi:10.1136/annrheumdis-2011-200418
- Roth T, Bhadra-Brown P, Pitman VW, Resnick EM. Pregabalin improves fibromyalgia-related sleep disturbance. Clin J Pain 2016; 32(4):308–312. doi:10.1097/AJP.0000000000000262
- Moldofsky H, Harris HW, Archambault WT, Kwong T, Lederman S. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol 2011; 38(12):2653–2663. doi:10.3899/jrheum.110194
- Feracioli-Oda E, Qawasmi A, Bloch MH. Meta-analysis: melatonin for the treatment of primary sleep disorders. PLoS One 2013; 8(5):e63773. doi:10.1371/journal.pone.0063773
- Ware MA, Fitzcharles MA, Joseph L, Shir Y. The effects of nabilone on sleep in fibromyalgia: results of a randomized controlled trial. Anesth Analg 2010; 110(2):604–610. doi:10.1213/ANE.0b013e3181c76f70
- Martínez M, Miró E, Sánchez AI, et al. Cognitive-behavioral therapy for insomnia and sleep hygiene in fibromyalgia: a randomized controlled trial. J Behav Med 2014; 37(4):683–697. doi:10.1007/s10865-013-9520-y
- Edwards RP, Cahalan C, Mensin G, Smith M, Haythornwaite JA. Pain, catastrophizing, and depression in the rheumatic diseases. Nature Rev Rheumatol 2011; 7(4):216–224. doi:10.1038/nrrheum.2011.2
- Sullivan MJL. The pain catastrophizing scale. User manual. McGill University, Montreal, Quebec, 2009. http://sullivan-painresearch.mcgill.ca/pdf/pcs/PCSManual_English.pdf. Accessed April 10, 2018.
- Louw A, Zimney K, O’Hotto C, Hilton S. The clinical application of teaching people about pain. Physiother Theory Pract 2016; 32(5):385–395. doi:10.1080/09593985.2016.1194652
- Burckhardt CS, Mannerkorpi K, Hedenberg L, Bjelle A. A randomized, controlled clinical trial of education and physical training for women with fibromyalgia. J Rheumatol 1994; 21(4):714–720. pmid:8035399
- King SJ, Wessel J, Bhambhani Y, Sholter D, Maksymowych W. The effects of exercise and education, individually or combined, in women with fibromyalgia. J Rheumatol 2002; 29(12):2620–2627. pmid:12465163
- Rooks DS, Gautam S, Romeling M, et al. Group exercise, education, and combination self-management in women with fibromyalgia: a randomized trial. Arch Intern Med 2007; 167(20):2192–2200. doi:10.1001/archinte.167.20.2192
- White KP, Nielson WR, Harth M, Ostbye T, Speechley M. Does the label ”fibromyalgia” alter health care status, function, and health service utilization? A prospective, within-group comparison in a community cohort of adults with chronic widespread pain. Arthritis Rheum 2002; 47(3):260–265. doi:10.1002/art.10400
- Bernardy K, Klose P, Busch AJ, Choy EH, Häuser W. Cognitive behavioural therapies for fibromyalgia. Cochrane Database Syst Rev 2013; 9:CD009796. doi:10.1002/14651858.CD009796.pub2
- Lazridou A, Kim J, Cahalan CM, et al. Effects of cognitive-behavioral therapy on brain connectivity supporting catastrophizing in fibromyalgia. Clin J Pain 2017; 33(3):215–221. doi:10.1097/AJP.0000000000000422
- Campbell CM, McCauley L, Bounds SC, et al. Changes in pain catastrophizing predict later changes in fibromyalgia clinical and experimental pain report: cross-lagged panel analyses of dispositional and situational catastrophizing. Arthritis Res Ther 2012; 14(5):R231. doi:10.1186/ar4073
- Vierck CJ. A mechanism-based approach to prevention of and therapy for fibromyalgia. Pain Res Treat 2012; 2012:951354. doi:10.1155/2012/951354
- Gerdle B, Ernberg M, Mannerkorpi K, et al. Increased interstitial concentrations of glutamate and pyruvate in vastus lateralis of women with fibromyalgia syndrome are normalized after exercise intervention—a case-control study. PLoS One 2016; 11(10):e0162010. doi:10.1371/journal.pone.0162010
- Jones J, Rutledge DN, Jones KD, Matallana L, Rooks DS. Self-assessed physical function levels of women with fibromyalgia: a national survey. Womens Health Issues 2008; 18(5):406–412. doi:10.1016/j.whi.2008.04.005
- Kelley GA, Kelley KS, Jones DL. Efficacy and effectiveness of exercise on tender points in adults with fibromyalgia: a meta-analysis of randomized controlled trials. Arthritis 2011; 2011:125485. doi:10.1155/2011/125485
- Valim V, Natour J, Ziao Y, et al. Effects of physical exercise on serum levels of serotonin and its metabolite in fibromyalgia: a randomized pilot study. Rev Bras Reumatol 2013; 53(6):538–541. doi:10.1016/j.rbr.2013.02.001
- Bidonde J, Busch AJ, Webber SC, et al. Aquatic exercise training for fibromyalgia. Cochrane Database Syst Rev 2014; 10:CD011336. doi:10.1002/14651858.CD011336
- Busch AJ, Webber SC, Richards RS, et al. Resistance exercise training for fibromyalgia. Cochrane Database Syst Rev 2013; 12:CD010884. doi:10.1002/14651858.CD010884
- Soriano-Maldonado A, Ruiz JR, Aparicio VA, et al. Association of physical fitness with pain in women with fibromyalgia: the al-Ándalus Project. Arthritis Care Res (Hoboken) 2015; 67(11):1561–1570. doi:10.1002/acr.22610
- Häuser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomized controlled trials. Arthritis Res Ther 2010; 12(3):R79. doi:10.1186/ar3002
- Häuser W, Perrot S, Clauw DJ, Fitzcharles MA. Unraveling fibromyalgia—steps towards individualized management. J Pain 2018; 19(2):125–134. doi:10.1016/j.jpain.2017.08.009
- Flodin P, Martinsen S, Mannerkorpi K, et al. Normalization of aberrant resting state functional connectivity in fibromyalgia patients following a three month physical exercise therapy. Neuroimage Clin 2015; 9:134–139. doi:10.1016/j.nicl.2015.08.004
- Dinoff A, Herrmann N, Swardfager W, et al. The effect of exercise training on resting concentrations of peripheral brain-derived neurotrophic factor (BDNF): a meta-analysis. PLOS One 2016. 11(9):e0163037. doi:10.1371/journal.pone.0163037
- Okifuji A, Gao J, Bokat C, Hare BD. Management of fibromyalgia syndrome in 2016. Pain Manag 2016; 6(4):383–400. doi:10.2217/pmt-2016-0006
- Masi AT, Vincent A. A historical and clinical perspective endorsing person-centered management of fibromyalgia syndrome. Curr Rheumatol Rev 2015; 11(2):86–95. pmid:26088217
- Fitzcharles MA, Ste-Marie PA, Goldenberg DL, et al; National Fibromyalgia Guideline Advisory Panel. 2012 Canadian guidelines for the diagnosis and management of fibromyalgia syndrome: executive summary. Pain Res Manag 2013: 18(3):119–126. pmid:23748251
- Ablin J, Fitzcharles MA, Buskila D, Shir Y, Sommer C, Häuser W. Treatment of fibromyalgia syndrome: recommendations of recent evidence-based interdisciplinary guidelines with special emphasis on complementary and alternative therapies. Evid Based Complement Alternat Med 2013; 2013:485272. doi:10.1155/2013/485272
- MacFarlane GJ, Kronisch C, Dean LE, et al. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis 2017; 76(2)318–328. doi:10.1136/annrheumdis-2016-209724
- Thieme K, Mathys M, Turk DC. Evidence-based guidelines on the treatment of fibromyalgia patients: are they consistent and if not, why not? Have effective psychological treatments been overlooked? J Pain 2017; 18(7):747–756. doi:10.1016/j.jpain.2016.12.006
- Williams DA, Kuper D, Segar M, Mohan N, Sheth M, Clauw DJ. Internet-enhanced management of fibromyalgia: a randomized controlled trial. Pain 2010; 151(3):694–702. doi:10.1016/j.pain.2010.08.034
- Petri M, Orbai AM, Alarcón GS, et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012; 64(8):2677–2686. doi:10.1002/art.34473
- Stanos S, Brodsky M, Argoff C, et al. Rethinking chronic pain in primary care setting. Postgraduate Med 2016; 128(5):502–513. doi:10.1080/00325481.2016.1188319
- Wolfe F, Clauw DJ, Fitzcharles M, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J Rheumatol 2011; 38(6):1113–1122. doi:10.3899/jrheum.100594
- Wolfe F, Clauw DJ, Fitzcharles MA, et al. 2016 Revisions to the 2010/2011 fibromyalgia diagnostic criteria. Semin Arthritis Rheum 2016; 46(3):319–329. doi:10.1016/j.semarthrit.2016.08.012
- Abeles AM, Abeles M. The clinical utility of a positive antinuclear antibody test result. Am J Med 2013; 126(4):342–348. doi:10.1016/j.amjmed.2012.09.014
- Li QZ, Karp DR, Quan J, et al. Risk factors for ANA positivity in healthy persons. Arthritis Res Ther 2011; 13(2):R38. doi:10.1186/ar3271
- Slight-Webb S, Lu R, Ritterhouse LL, et al. Autoantibody-positive healthy individuals display unique immune profiles that may regulate autoimmunity. Arthritis Rheumatol 2016; 68(1):2492–2502. doi:10.1002/art.39706
- Arbuckle MR, McClain MT, Rubertone MV, et al. Development of autoantibodies before the clinical onset of systemic lupus erythematosus. N Engl J Med 2003; 349(16):1526–1533. doi:10.1056/NEJMoa021933
- Mariz HA, Sato EI, Barbosa SH, Rodrigues SH, Dellavance A, Andrade LE. Pattern of antinuclear antibody-HEp 2 test is a critical parameter for discriminating antinuclear antibody-positive healthy individuals and patients with autoimmune diseases. Arthritis Rheum 2011; 63(1):191–200. doi:10.1002/art.30084
- Häuser W, Clauw DJ, Fitzcharles MA. Treat to target in fibromyalgia: opening the dialogue. Arthritis Care Res (Hoboken) 2017; 69(4):462–466. doi:10.1002/acr.22970
- Forseth KO, Husby G, Gran JT, Fórre O. Prognostic factors for the development of fibromyalgia in women with self-reported musculoskeletal pain. A prospective study. J Rheumatol 1999; 26(11):2458–2567. pmid:10555910
- Thieme K, Turk DC, Flor H. Comorbid depression and anxiety in fibromyalgia syndrome: relationship to somatic and psychosocial variables. Psychosom Med 2004; 66(6):837–844. doi:10.1097/01.psy.0000146329.63158.40
- Fuller-Thomson E, Mimigon-Young J, Brennenstuhl S. Individuals with fibromyalgia and depression: findings from a nationally representative Canadian survey. Rheumatol Int 2012; 32(4):853–862. doi:10.1007/s00296-010-1713-x
- Kudlow PA, Rosenblat JD, Weissman CR, et al. Prevalence of fibromyalgia and co-morbid bipolar disorder: a systematic review and meta-analysis. J Affect Disord 2015; 188:134–142. doi:10.1016/j.jad.2015.08.030
- Corruble E, Guelfi JD. Pain complaints in depressed inpatients. Psychopathology 2000; 33(6):307–309. doi:10.1159/000029163
- Gota CE, Kaouk S, Wilke WS. The impact of depressive and bipolar symptoms on socioeconomic status, core symptoms, function and severity of fibromyalgia. Int J Rheum Dis 2017; 20(3):326–339. doi:10.1111/1756-185X.12603
- Toussaint LL, Vincent A, McAlister SJ, Oh TH, Hassett AL. A comparison of fibromyalgia symptoms in patients with healthy versus depressive, low and reactive affect balance styles. Scand J Pain 2014; 5(3):161–166. doi:10.1016/j.sjpain.2014.05.001
- Vincent A, Hoskin TL, Whipple MO, et al. OMERACT-based fibromyalgia symptom subgroups: an exploratory cluster analysis. Arthritis Res Ther 2014; 16(5):463. doi:10.1186/s13075-014-0463-7
- Robinson RL, Birnbaum HG, Morley MA, Sisitsky T, Greenberg PE, Wolfe F. Depression and fibromyalgia: treatment and cost when diagnosed separately or concurrently. J Rheumatol 2004; 31(8):1621–1629. pmid:5290744
- Choy EH. The role of sleep in pain and fibromyalgia. Nature Rev Rheumatol 2015; 11(9):513–520. doi:10.1038/nrrheum.2015.56
- Mork PJ, Nilsen TI. Sleep problems and risk of fibromyalgia: longitudinal data of an adult female population in Norway. Arthritis Rheum 2012; 64(1):281–284. doi:10.1002/art.33346
- McBeth J, Lacey RJ, Wilkie R. Predictors of new-onset widespread pain in older adults: results from a population-based prospective cohort study in the UK. Arthritis Rheumatol 2014; 66(3):757–767. doi:10.1002/art.38284
- Ablin JN, Clauw DJ, Lyden AK, et al. Effects of sleep restriction and exercise deprivation on somatic symptoms and mood in healthy adults. Clin Exp Rheumatol 2013; 31(6 suppl 79):S53–S59. pmid:24373363
- Russell IJ, Perkins AT, Michalek JE; Oxybate SXB-26 Fibromyalgia Syndrome Study Group. Sodium oxybate relieves pain and improves function in fibromyalgia syndrome: a randomized, double-blind, placebo-controlled, multicenter clinical trial. Arthritis Rheum 2009; 60(1):299–309. doi:10.1002/art.24142
- Spaeth M, Bennett RM, Benson BA, Wang YG, Lai C, Choy EH. Sodium oxybate therapy provides multidimensional improvement in fibromyalgia: results of an international phase 3 trial. Ann Rheum Dis 2012; 71(6):935–942. doi:10.1136/annrheumdis-2011-200418
- Roth T, Bhadra-Brown P, Pitman VW, Resnick EM. Pregabalin improves fibromyalgia-related sleep disturbance. Clin J Pain 2016; 32(4):308–312. doi:10.1097/AJP.0000000000000262
- Moldofsky H, Harris HW, Archambault WT, Kwong T, Lederman S. Effects of bedtime very low dose cyclobenzaprine on symptoms and sleep physiology in patients with fibromyalgia syndrome: a double-blind randomized placebo-controlled study. J Rheumatol 2011; 38(12):2653–2663. doi:10.3899/jrheum.110194
- Feracioli-Oda E, Qawasmi A, Bloch MH. Meta-analysis: melatonin for the treatment of primary sleep disorders. PLoS One 2013; 8(5):e63773. doi:10.1371/journal.pone.0063773
- Ware MA, Fitzcharles MA, Joseph L, Shir Y. The effects of nabilone on sleep in fibromyalgia: results of a randomized controlled trial. Anesth Analg 2010; 110(2):604–610. doi:10.1213/ANE.0b013e3181c76f70
- Martínez M, Miró E, Sánchez AI, et al. Cognitive-behavioral therapy for insomnia and sleep hygiene in fibromyalgia: a randomized controlled trial. J Behav Med 2014; 37(4):683–697. doi:10.1007/s10865-013-9520-y
- Edwards RP, Cahalan C, Mensin G, Smith M, Haythornwaite JA. Pain, catastrophizing, and depression in the rheumatic diseases. Nature Rev Rheumatol 2011; 7(4):216–224. doi:10.1038/nrrheum.2011.2
- Sullivan MJL. The pain catastrophizing scale. User manual. McGill University, Montreal, Quebec, 2009. http://sullivan-painresearch.mcgill.ca/pdf/pcs/PCSManual_English.pdf. Accessed April 10, 2018.
- Louw A, Zimney K, O’Hotto C, Hilton S. The clinical application of teaching people about pain. Physiother Theory Pract 2016; 32(5):385–395. doi:10.1080/09593985.2016.1194652
- Burckhardt CS, Mannerkorpi K, Hedenberg L, Bjelle A. A randomized, controlled clinical trial of education and physical training for women with fibromyalgia. J Rheumatol 1994; 21(4):714–720. pmid:8035399
- King SJ, Wessel J, Bhambhani Y, Sholter D, Maksymowych W. The effects of exercise and education, individually or combined, in women with fibromyalgia. J Rheumatol 2002; 29(12):2620–2627. pmid:12465163
- Rooks DS, Gautam S, Romeling M, et al. Group exercise, education, and combination self-management in women with fibromyalgia: a randomized trial. Arch Intern Med 2007; 167(20):2192–2200. doi:10.1001/archinte.167.20.2192
- White KP, Nielson WR, Harth M, Ostbye T, Speechley M. Does the label ”fibromyalgia” alter health care status, function, and health service utilization? A prospective, within-group comparison in a community cohort of adults with chronic widespread pain. Arthritis Rheum 2002; 47(3):260–265. doi:10.1002/art.10400
- Bernardy K, Klose P, Busch AJ, Choy EH, Häuser W. Cognitive behavioural therapies for fibromyalgia. Cochrane Database Syst Rev 2013; 9:CD009796. doi:10.1002/14651858.CD009796.pub2
- Lazridou A, Kim J, Cahalan CM, et al. Effects of cognitive-behavioral therapy on brain connectivity supporting catastrophizing in fibromyalgia. Clin J Pain 2017; 33(3):215–221. doi:10.1097/AJP.0000000000000422
- Campbell CM, McCauley L, Bounds SC, et al. Changes in pain catastrophizing predict later changes in fibromyalgia clinical and experimental pain report: cross-lagged panel analyses of dispositional and situational catastrophizing. Arthritis Res Ther 2012; 14(5):R231. doi:10.1186/ar4073
- Vierck CJ. A mechanism-based approach to prevention of and therapy for fibromyalgia. Pain Res Treat 2012; 2012:951354. doi:10.1155/2012/951354
- Gerdle B, Ernberg M, Mannerkorpi K, et al. Increased interstitial concentrations of glutamate and pyruvate in vastus lateralis of women with fibromyalgia syndrome are normalized after exercise intervention—a case-control study. PLoS One 2016; 11(10):e0162010. doi:10.1371/journal.pone.0162010
- Jones J, Rutledge DN, Jones KD, Matallana L, Rooks DS. Self-assessed physical function levels of women with fibromyalgia: a national survey. Womens Health Issues 2008; 18(5):406–412. doi:10.1016/j.whi.2008.04.005
- Kelley GA, Kelley KS, Jones DL. Efficacy and effectiveness of exercise on tender points in adults with fibromyalgia: a meta-analysis of randomized controlled trials. Arthritis 2011; 2011:125485. doi:10.1155/2011/125485
- Valim V, Natour J, Ziao Y, et al. Effects of physical exercise on serum levels of serotonin and its metabolite in fibromyalgia: a randomized pilot study. Rev Bras Reumatol 2013; 53(6):538–541. doi:10.1016/j.rbr.2013.02.001
- Bidonde J, Busch AJ, Webber SC, et al. Aquatic exercise training for fibromyalgia. Cochrane Database Syst Rev 2014; 10:CD011336. doi:10.1002/14651858.CD011336
- Busch AJ, Webber SC, Richards RS, et al. Resistance exercise training for fibromyalgia. Cochrane Database Syst Rev 2013; 12:CD010884. doi:10.1002/14651858.CD010884
- Soriano-Maldonado A, Ruiz JR, Aparicio VA, et al. Association of physical fitness with pain in women with fibromyalgia: the al-Ándalus Project. Arthritis Care Res (Hoboken) 2015; 67(11):1561–1570. doi:10.1002/acr.22610
- Häuser W, Klose P, Langhorst J, et al. Efficacy of different types of aerobic exercise in fibromyalgia syndrome: a systematic review and meta-analysis of randomized controlled trials. Arthritis Res Ther 2010; 12(3):R79. doi:10.1186/ar3002
- Häuser W, Perrot S, Clauw DJ, Fitzcharles MA. Unraveling fibromyalgia—steps towards individualized management. J Pain 2018; 19(2):125–134. doi:10.1016/j.jpain.2017.08.009
- Flodin P, Martinsen S, Mannerkorpi K, et al. Normalization of aberrant resting state functional connectivity in fibromyalgia patients following a three month physical exercise therapy. Neuroimage Clin 2015; 9:134–139. doi:10.1016/j.nicl.2015.08.004
- Dinoff A, Herrmann N, Swardfager W, et al. The effect of exercise training on resting concentrations of peripheral brain-derived neurotrophic factor (BDNF): a meta-analysis. PLOS One 2016. 11(9):e0163037. doi:10.1371/journal.pone.0163037
- Okifuji A, Gao J, Bokat C, Hare BD. Management of fibromyalgia syndrome in 2016. Pain Manag 2016; 6(4):383–400. doi:10.2217/pmt-2016-0006
- Masi AT, Vincent A. A historical and clinical perspective endorsing person-centered management of fibromyalgia syndrome. Curr Rheumatol Rev 2015; 11(2):86–95. pmid:26088217
- Fitzcharles MA, Ste-Marie PA, Goldenberg DL, et al; National Fibromyalgia Guideline Advisory Panel. 2012 Canadian guidelines for the diagnosis and management of fibromyalgia syndrome: executive summary. Pain Res Manag 2013: 18(3):119–126. pmid:23748251
- Ablin J, Fitzcharles MA, Buskila D, Shir Y, Sommer C, Häuser W. Treatment of fibromyalgia syndrome: recommendations of recent evidence-based interdisciplinary guidelines with special emphasis on complementary and alternative therapies. Evid Based Complement Alternat Med 2013; 2013:485272. doi:10.1155/2013/485272
- MacFarlane GJ, Kronisch C, Dean LE, et al. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis 2017; 76(2)318–328. doi:10.1136/annrheumdis-2016-209724
- Thieme K, Mathys M, Turk DC. Evidence-based guidelines on the treatment of fibromyalgia patients: are they consistent and if not, why not? Have effective psychological treatments been overlooked? J Pain 2017; 18(7):747–756. doi:10.1016/j.jpain.2016.12.006
- Williams DA, Kuper D, Segar M, Mohan N, Sheth M, Clauw DJ. Internet-enhanced management of fibromyalgia: a randomized controlled trial. Pain 2010; 151(3):694–702. doi:10.1016/j.pain.2010.08.034
KEY POINTS
- Fibromyalgia is a clinical diagnosis, and specialized testing beyond basic laboratory tests is not indicated.
- Antinuclear antibody test results can be confusing, and the test should not be ordered unless a patient has objective features suggesting systemic lupus erythematosus.
- Treatment should be tailored to comorbidities such as depression, anxiety, and sleep disturbance. Options include serotonin-norepinephrine reuptake inhibitors (eg, duloxetine), selective serotonin reuptake inhibitors, low-dose tricyclic antidepressants (eg, amitriptyline), and gabapentinoids (pregabalin or gabapentin). These drugs can be used singly or in combination.
- Medications that do not work should be discontinued.
- “Catastrophizing” by the patient is common in fibromyalgia and can be addressed by education, cognitive behavioral therapy, and anxiolytic or antidepressant drugs.
- Sustained, lifelong exercise is the treatment strategy most associated with improvement.