User login
Fall Prevention Strategies
Inpatient falls are the most common type of inpatient adverse event,1 persist as a significant problem nationally, and result in patient injury, increased length of stay, healthcare costs, and litigation.27 Inpatient falls remain a main focus of patient safety and a measure of quality in this era of healthcare reform and quality improvement.8 Inpatient fall rates per 1000 patient‐days range from 1.4 to 18.2.4, 9 The absolute percentage of inpatients that fall ranges from 1.3% to 7%.4, 5, 9, 10 Of inpatient falls, almost all data suggest that roughly one‐third result in some type of injury while 3%‐8% result in serious injury or death.9, 1113
Fall prevention interventions have largely been aimed at modifiable risk factors such as getting out of bed with bed alarms, toileting needs with bedside commodes, and reducing delirium through reorientation techniques. There have been several attempts at decreasing fall rates in hospitals surrounding a multidisciplinary, team‐based approach. Two Cochrane reviews and 2 meta‐analyses have partially examined this issue with mixed results.1417 However, none of these reviews focused on the acute care inpatient population. In fact, the majority of the data analyzed for inpatients was from rehabilitation wards and long‐term care wards. Additionally, there exists almost no data examining fall prevention with single interventions in the acute inpatient population, likely due to the belief that falls are multifactorial in etiology and require more comprehensive interventions.
The aim of this article is to determine the impact of team‐based, multidisciplinary quality improvement efforts to reduce inpatient falls in acute care inpatient hospitals and identify key features that determine their effectiveness.
METHODS
Data Sources and Searches
A search of MEDLINE, CINAHL, EMBASE, and the Cochrane Library was done using the medical subject heading (MeSH) terms accidental falls, accident prevention, inpatients, and prevention and control. Non‐English language publications were included in the search. The search encompassed all published literature through December 1, 2011. In addition, reference lists of all systematic reviews and meta‐analyses were searched to identify all possible studies available.1416
Study Selection
Only primary research studies relating to acute care inpatient hospital fall prevention were included. Data generated exclusively or partially from psychiatric wards, rehabilitation units, subacute facilities, and long‐term facilities were excluded from the review.
Data Extraction and Quality Assessment
Each selected study was carefully hand searched by 2 authors for the purposes of data extraction. Data were collected for the following study characteristics and outcome measures: details of the fall prevention intervention used (allowing for all interventions used to be recorded in Table 3), markers of study quality, study period, study population, mean age of participants, sample size (in 1000 patient‐days), and fall rates (in 1000 patient‐days). In certain cases, sample size was converted to patient‐days using reported data points of total number of patients and average length of stay.
Two authors with experience in fall literature discussed methodological quality and reached a consensus regarding scores using a 20‐point scale previously described in fall literature for all studies included.14, 15 Ten individual criteria were scored on a 0‐2 point scale. No points were awarded when the criteria were not met, not clearly mentioned, or not mentioned at all. One point was awarded when the criterion was partially met, and both points awarded when it was fully met.
Data Synthesis and Analysis
Fall rate per 1000‐patient days was derived from reported data in both intervention and non‐intervention groups within each study. Effect sizes (odds ratios [OR]) and 95% confidence intervals (CI) were derived for individual studies and then combined across research reports using an inverse weighted random‐effects meta‐analysis.18 Random effects methodology was chosen to account for within‐study and between‐study variation. Statistical heterogeneity between trials was assessed using the Cochrane Q statistic and reported as I2, which estimates the percentage of variability across studies that is not due to chance.19 Due to the low number of included studies in our analysis, a formal statistical test on publication bias was not meaningful.20 Statistical significance was defined as P 0.05. Data analyses were done using Comprehensive Meta‐Analysis, Version 2 (Biostat, Englewood, NJ).
RESULTS
Selected Studies
Electronic search produced 259 results on MEDLINE, 2 results from the Cochrane Library, 94 from CINAHL, and 4 from EMBASE. Each result was hand searched to exclude duplicates, and irrelevant studies. Once such data were excluded, the above inclusion and exclusion criteria identified 6 primary articles for review.9, 2125 Additionally, a cluster randomized fall prevention trial in a mixed inpatient population was published by Cumming et al26 in 2008. The study was excluded, as the participants were pooled between rehabilitation wards and acute inpatient wards, and only incomplete data were reported separately for the acute inpatient wards. We were unsuccessful at obtaining necessary data to analyze the acute inpatient wards.
Study Quality
The quality assessment results scores ranged from 11 to 14 out of a possible 20 (Table 1). None of the studies explicitly used an intention‐to‐treat statistical model, as the nature of inpatient care largely prevents drop‐out or crossover, and all patients were included in individual study results.
| Included Study | Clearly Defined Inclusion and Exclusion Criteria | Randomization | Comparable Treatment Groups at Entry | Identical Standard Program for Both Groups | Fall Incident Clearly Defined and Staff Trained in Definition | Blinded Treatment Providers | Blinded Outcome Assessors | Blinded Patients | Identical Appraisal of Outcomes* | Intention‐to‐ Treat Analysis | Total Score (0‐20) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| Dykes et al22 | 2 | 2 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 14 |
| Krauss et al23 | 2 | 0 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 12 |
| Brandis21 | 1 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 10 |
| Mitchell and Jones25 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Schwendimann et al9 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Williams et al24 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
Study Characteristics
The available data are skewed towards elderly patients being hospitalized in general medicine or geriatric units (Table 2). All but 1 study had a large sample size, with 1000‐patient days ranging from 11.1 to 160.3.9, 2124
| Included Study | Study Design | Study Period | Study Wards | Mean Age | Sample Size With Intervention (1000 Patient‐Days) | Sample Size in Control (1000 Patient‐Days) | Fall Rate With Intervention (Falls per 1000 Patient‐Days) | Fall Rate in Control (Falls per 1000 Patient‐Days) |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| Dykes et al22 | RCT | 6 mo | 2 Medical units | 50% 65‐17% 65‐74 33% 75 | 24.1 | 24.1 | 4.18 | 4.64 |
| Krauss et al23 | Quasi‐experimental | 9 mo | General Medicine wards | 65.5 | 11.2 | 11.39 | 5.09 | 6.85 |
| Brandis21 | Pre/post | 12 mo | 500‐Bed acute care hospital | Not reported | 160.3 | 155.2 | 1.61 | 1.74 |
| Mitchell and Jones25 | Pre/post | 6 mo | Acute care hospital | 76.23 (Pre) 72.1 (Post) | 4.3 | 5 | 4.42 | 7.77 |
| Schwendimann et al9 | Pre/post | 4 yr | Internal Med, Surgery, and Geriatrics | 67.3 | 46.8 | 41.9 | 8.6 | 9.1 |
| Williams et al24 | Pre/post | 6 mo | 3 Medical wards and a Geriatrics ward | 79 | 15.88 | 12.53 | 8 | 9.5 |
Components of the Intervention
Multidisciplinary interventions were complex, and formulated based on available evidence for individual interventions and modifiable fall risk factors (Table 3). Each study reviewed included a fall risk assessment to risk‐stratify participants and modulate intervention according to risk.9, 2125
| Included Study | Fall Risk Assessment Used | Mobility Assessment and Assistance if Necessary | Mobility Aid Provided if Necessary | Medication Modification | Education About Risk Factors | Fall Risk Sign/Warning in Chart | Bedside Interventions (eg, Bed Alarm, Rail Adjustment, Bed Location/ Position, etc) | Toileting Schedule | Exercise Program | Other(s) |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Dykes et al22* | + | + | + | + | + | + | + | + | Frequent bed checks, documented fall prevention plan | |
| Krauss et al23 | + | + | + | + | + | + | + | + | Use of bedside interventions was done based on discretion on a case‐by‐case basis | |
| Brandis21 | + | + | + | Ward modifications after OT assessment of patient rooms and bathrooms; hip protectors | ||||||
| Mitchell and Jones25 | + | + | + | + | Introduced detailed system to track fall details; used other preventive actions not specified | |||||
| Schwendimann et al9 | + | + | + | + | + | + | + | + | + | Reassessment of patients who did fall; hip protectors |
| Williams et al24 | + | + | + | + | + | + | Possible sitter | |||
Each study implemented fall prevention programs in a slightly different way. Krauss et al23 used nurses to complete a Morse Fall Scale and subsequently implement several standard interventions based on risk. Staff was then authorized to employ bedside interventions as necessary without systematic data collection. Schwendimann et al9 had nurses complete a simple fall risk assessment (based on history of falls, impaired mobility, and impaired cognition) that prompted the examination by a physician if risk was determined to be high. A subsequent team‐based intervention was employed with nursing, physiotherapy, and the physician. Brandis21 employed a team of nurses and the aid of the Director of Occupational Therapy to assess risk (using an undisclosed system) and carry out an intervention. Dykes et al22 examined an electronic fall prevention tool kit (FPTK) using the electronic medical record (EMR). This intervention began with the Morse Fall Score, which triggered automatically ordered interventions that did not require personal oversight. In fact, the multidisciplinary interventions in the intervention group were also used in the control arm. The difference was the automatic nature in which the interventions were ordered in the interventions arm. Williams et al24 used nurses and physiotherapists, who were specifically trained for the study, to carry out study interventions. The Mitchell and Jones25 study focused on nursing care alone to carry out intervention and used a novel risk assessment tool.
Fall Rates
Dykes et al22 and Williams et al24 found a statistically significant reduction in fall rate with falls reduced by 1.16 per 1000‐patient days and 1.5 per 1000‐patient days, respectively. Mitchell and Jones25 demonstrated a large fall reduction but had an extremely small sample size. Brandis21 found an extremely small reduction in fall rates and failed to report a P‐value. Krauss et al23 showed a trend towards reducing falls, and even showed a statistically significant reduction over the first 5 months of the study, but lost significance in the final 4 months. Similarly, Schwendimann et al9 saw more impressive fall reductions in the first year of the study that dissipated in the final 3 years of data collection.
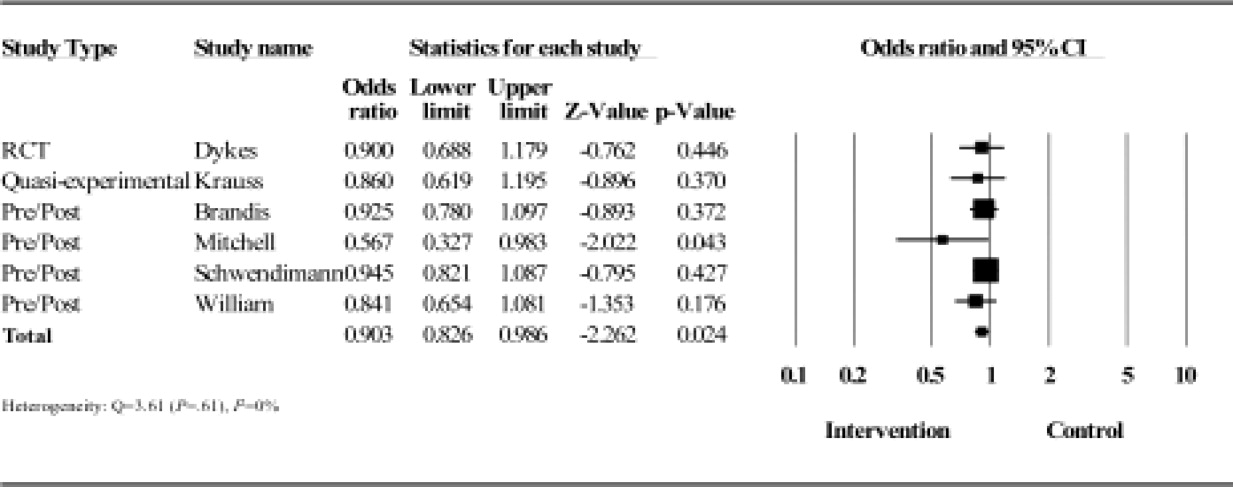
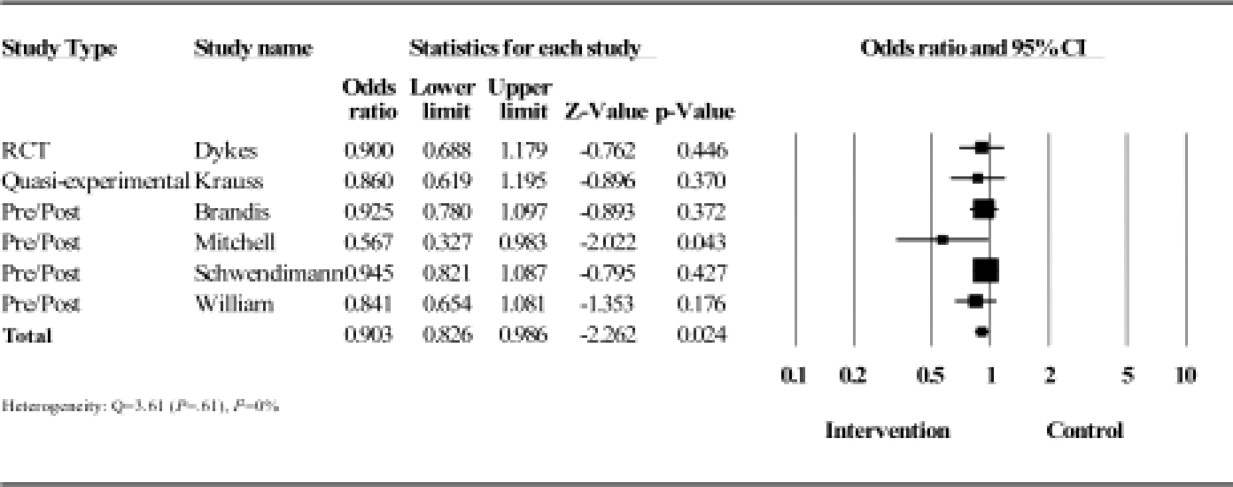
Results from the meta‐analysis of the 6 studies comparing odds ratios are displayed quantitatively and as a forest plot in Figure 1. The figure shows results with 95% CI for each individual study and overall. There was no statistical evidence of heterogeneity between the studies or study designs. Although, due to the small number of studies included, there is poor power to detect true heterogeneity among studies. The magnitude of boxes shown is a relative sample size indicator. Using the random‐effects model, the summary odds ratio is 0.90 (95% CI, 0.83 to 0.99) (P = 0.02) (I2 = 0%).27
DISCUSSION
The frequency and morbidity associated with inpatient falls is well established, based on reproduced epidemiologic data. Reducing these adverse events could reduce morbidity, mortality, and healthcare costs, and has become the focus of most hospitals quality and patient safety initiatives. The focus of this review was to examine multidisciplinary efforts to reduce falls in acute care inpatient hospitals. Despite the importance and scope of the problem, there is a paucity of research available on this topic, with a wide literature search yielding only 6 primary research studies.
Our major finding is that multidisciplinary fall prevention strategies have a statistically significant impact on fall rates with a combined OR of 0.90. While this review demonstrates a significant benefit to multidisciplinary fall prevention strategies in the acute inpatient population, the clinical impact of these efforts may be limited. Based on rates ranging from 1.7 to 9.5 falls per 1000‐patient days, multidisciplinary interventions would reduce falls by 1 to 10 falls per 10,000‐patient days using the combined OR calculated of 0.9. Using other available incidence data regarding inpatient falls,4, 9 a reasonable baseline frequency to consider would be 8 falls per 1000 patient‐days. Assuming that prevalence, the number needed to treat (NNT) to prevent a single inpatient fall is 1250 patient days. Furthermore, based on available data, only approximately one‐third of these falls result in injury and only a minor fraction of these results in serious injury.9, 1113 The magnitude of this apparent benefit in the context of fall incidence rates raises some concerns about cost‐effectiveness given the high staffing and systems needs that multidisciplinary prevention programs require. This also suggests that there are limitations when using inpatient falls as a measure of healthcare quality given the absence of high‐quality evidence demonstrating a viable solution to the problem. At present, the Center for Medicare and Medicaid services limit reimbursement for fall‐related injuries if they occur during an acute inpatient hospitalization.28
The complexity of the interventions used may help explain the limited impact. Krauss et al23 examined compliance to their interventions and found less than ideal results. They found only 36.4% of intervention floor patients had maintained a toileting schedule compared to 24.6% on control floors. Additionally, a greater proportion of patients on control floors had a physical or occupational therapy consult, and only 1.8% more patients on intervention floors had walking aids provided. These were all strategies emphasized on the intervention floors. Similarly, Schwendimann et al9 questioned their staff's adherence to protocol after fall prevention committee audits. This may help explain why a potential benefit lost statistical significance with time, based on a natural tendency towards more participation at the beginning of a new policy. Williams et al24 reported only a 64% compliance rate with fall care plan forms and 77% rate of missing information on fall care plans. A multidisciplinary fall prevention study that did not meet inclusion criteria (based on study population) yielded strongly positive results for which the authors commented mostly on changing of the hospital culture surrounding fall prevention as a key to their success.29 Adoptability of a multidisciplinary intervention will clearly impact adherence and the intervention's ultimate effectiveness.
Single intervention strategies, not analyzed in this review, are simpler to execute and adhere to. While these types of interventions may be superior, there is extremely limited data supporting or refuting patient fall benefits in the acute care inpatient population when using simple single interventions. However, some data generated partially on acute care geriatrics wards targeting patient education only showed benefit.30
Dykes et al22 was able to improve compliance rates by removing steps in the process of executing interventions with the FPTK built into the EMR. Importantly, the FPTK was compared against very similar fall prevention strategies, the difference being that patients randomized to the FPTK arm had the assessment and interventions automatically prompted on admission in the EMR. Adherence was measured through Morse Fall Scale completion rates (81% in control units versus 94% in intervention units).22 In many ways, the utility of this study was displaying a fall risk reduction by simply enhancing compliance using health information technology with automated alerts. Additionally, both arms of the study reported low fall rates compared to previously reported data, and there may have been larger benefit seen if the FPTK was compared against no fall prevention strategy. This diminishing of effect size may have been present in all studies reviewed, as usual hospital care commonly includes basic patient safety measures.
Another potential problem with the multidisciplinary fall prevention programs included in the meta‐analysis is the inability to target interventions. Each study employed a fall risk score in an attempt to focus resources on a select group of high‐risk patients. This method is problematic given that countless risk factors for inpatient falls have been identified in the literature. Factors that have been described range from clinical characteristics to laboratory tests.31 The most consistently reproducible patient‐related risks are altered mental status (including cognitive impairment and depression), altered mobility (particularly lower limb weakness), a history of falls, and toileting needs.13, 3236 Less consistency is seen with other traditional risk factors such as age, sedating medication, and length of stay.5, 13, 32, 3638 Attempting to risk‐stratify patients using simple and accurate assessment tools developed from these risk factors has proven to be very difficult. Many tools have been developed based on identified risk factors, but perform very poorly when trying to identify patients who will fall with reasonable specificity and positive predictive value.34, 3944 In fact, it has been demonstrated that using a nurse's judgment, a physician's opinion based on a patient's likelihood to wander or a simple 2‐question tool have all performed better than sophisticated risk calculators.33, 45, 46 Therefore, it is possible that interventions could benefit from including all patients, with de‐emphasis on unproven risk stratification tools.
In contrast to our findings, a modest risk reduction has been demonstrated in several primary articles and meta‐analyses in the subacute, rehabilitation, and long‐term care populations.15, 16, 4750 Additionally, a recent study has described a 63.9% risk reduction in a population that included medical, surgical, psychiatric, and rehabilitation wards.29 One important difference between these settings and the acute inpatient populations may be the amount of time and energy that can be dedicated to fall prevention and overall care planning. Another likely factor is the added challenge of preventing falls in patients with more active medical illnesses. In the acute care setting, a patient's chief complaint may not be completely addressed at the time of first mobilization and ambulation. This may be most relevant in patients who are admitted with syncope, seizure, vertigo, and dehydration.
Our study has several limitations; most notably, the available evidence is limited in quality and quantity. Furthermore, omission of unpublished data may also lead to effect bias, though this would likely be in the direction of ineffective interventions supporting a conclusion that multidisciplinary efforts have had only a small impact on fall rates. Ideally, future studies can limit confounding variables through randomization. However, it is difficult to adequately blind when studying a multidisciplinary fall intervention that depends on patient and provider participation. As a result, none of the papers reviewed met criteria for high quality. However, almost all available data examined in this review came from large sample sizes in which thoughtful interventions were used. Since an inpatient fall will not affect the majority of patients, it was crucial for these studies to recruit a large sample size to have adequate power to detect a difference in fall rates. However, each study used risk assessment tools, which are poor indicators of who will and will not fall in the hospital.34, 39, 42 This may suggest a need for improved risk assessment tools, or be further evidence to include all patients in fall prevention regardless of risk. Quantitative synthesis of multidisciplinary fall interventions has the added limitation of comparing complex, multifaceted treatments that are not perfectly uniform. It is our opinion that interventions are semi‐standardized using the grouping methods employed in Table 3.
Preventing inpatient falls remains a difficult issue to address while convincing data is lacking. Based on current evidence, multidisciplinary fall prevention efforts on acutely ill inpatients show a possible small benefit and should be explored from a cost‐effectiveness standpoint to ensure they garner appropriate investment. Many resources are required to run such teams including nursing staff, equipment, physical and occupational therapy staff, pharmacists, and specialized staff training. We are unaware of any such cost‐effectiveness data available. Effective interventions may be those that maximize compliance through health information technology, maintain staff dedication, increase staff availability, improve risk assessment, or include all patients regardless of calculated fall risk, and take the patient's chief complaint into account in the fall prevention strategy. Where resources are limited, it appears most reasonable to focus on major risk factors for inpatient falls that have independently been shown to be detrimental to outcomes, such as delirium.51 Additionally, using inpatient fall rates as a hospital quality measure may be premature, given the lack of proven efforts to lower fall rates. Multidisciplinary fall prevention efforts on acutely ill inpatients should be further studied using high‐quality, randomized trials. It remains to be seen whether these large programs are cost‐effective, or on balance clinically effective.
- ,,.Patient accidents in hospital: incidence, documentation and significance.Br J Clin Pract.1994;48(2):63–66.
- ,,,.Serious falls in hospitalized patients: correlates and resource utilization.Am J Med.1995;99(2):137–143.
- ,,,.Hospital falls: a persistent problem.Am J Public Health.1985;75(7):775–777.
- ,,.Falls in the acute hospital setting—impact on resource utilisation.Aust Health Rev.2007;31(3):471–477.
- ,,.Incidence and risk factors for inpatient falls in an academic acute‐care hospital.J Nippon Med Sch.2006;73(5):265–270.
- ,,,.Do falls and falls‐injuries in hospital indicate negligent care—and how big is the risk? A retrospective analysis of the NHS Litigation Authority Database of clinical negligence claims, resulting from falls in hospitals in England 1995 to 2006.Qual Saf Health Care.2008;17(6):431–436.
- ,,,.Hospital falls: development of a predictive model for clinical practice.Appl Nurs Res.1995;8(3):129–139.
- ,.Centers for Medicare and Medicaid Services' “never events”: an analysis and recommendations to hospitals.Health Care Manag (Frederick).2008;27(4):338–349.
- ,,,.Falls and consequent injuries in hospitalized patients: effects of an interdisciplinary falls prevention program.BMC Health Serv Res.2006;6:69.
- .Immobility and falls.Clin Geriatr Med.1998;14(4):699–726.
- ,,.A case control study of falls in the hospital setting.J Gerontol Nurs.1998;24(12):7–15.
- ,,,.A retrospective analysis of patient falls.Can J Public Health.1985;76(2):116–118.
- ,,, et al.Characteristics and circumstances of falls in a hospital setting: a prospective analysis.J Gen Intern Med.2004;19(7):732–739.
- ,,, et al.Interventions for preventing falls in older people in nursing care facilities and hospitals.Cochrane Database Syst Rev.2010(1):CD005465.
- , L,,,,.Interventions for preventing falls in acute‐ and chronic‐care hospitals: a systematic review and meta‐analysis.J Am Geriatr Soc.2008;56(1):29–36.
- ,,, et al.Strategies to prevent falls and fractures in hospitals and care homes and effect of cognitive impairment: systematic review and meta‐analyses.BMJ.2007;334(7584):82.
- ,,,,,.Interventions for preventing falls in elderly people.Cochrane Database Syst Rev.2003(4):CD000340.
- ,,,.Introduction to Meta‐Analysis.Chichester, UK:John Wiley 2009.
- ,.Quantifying heterogeneity in a meta‐analysis.Stat Med.2002;21(11):1539–1558.
- .The power of the standard test for the presence of heterogeneity in meta‐analysis.Stat Med.2006;25(15):2688–2699.
- .A collaborative occupational therapy and nursing approach to falls prevention in hospital inpatients.J Qual Clin Pract.1999;19(4):215–220.
- ,,, et al.Fall prevention in acute care hospitals: a randomized trial.JAMA.2010;304(17):1912–1918.
- ,,,,,.Intervention to prevent falls on the medical service in a teaching hospital.Infect Control Hosp Epidemiol.2008;29(6):539–545.
- ,,, et al.Evaluation of a falls prevention programme in an acute tertiary care hospital.J Clin Nurs.2007;16(2):316–324.
- ,.Striving to prevent falls in an acute care setting—action to enhance quality.J Clin Nurs.1996;5(4):213–220.
- ,,, et al.Cluster randomised trial of a targeted multifactorial intervention to prevent falls among older people in hospital.BMJ.2008;336(7647):758–760.
- ,.Graphical displays for meta‐analysis: an overview with suggestions for practice.Res Syn Meth.2010(1):66–80.
- Centers for Medicare 37(7):317–325.
- ,‐M,, et al.Patient education to prevent falls among older hospital inpatients: a randomized controlled trial.Arch Intern Med.2011;117:516–524.
- ,.The relationship between patient blood pathology values and patient falls in an acute‐care setting: a retrospective analysis.Int J Nurs Pract.2005;11(4):161–168.
- ,,,.Falls risk factors in the hospital setting: a systematic review.Int J Nurs Pract.2001;7(1):38–45.
- ,,.A simplified fall‐risk assessment tool for patients hospitalized in medical wards.Isr Med Assoc J.2008;10(2):125–129.
- ,,,.Risk factors and risk assessment tools for falls in hospital in‐patients: a systematic review.Age Ageing.2004;33(2):122–130.
- ,,, et al.Circumstances of patient falls and injuries in 9 hospitals in a midwestern healthcare system.Infect Control Hosp Epidemiol.2007;28(5):544–550.
- ,,, et al.A case‐control study of patient, medication, and care‐related risk factors for inpatient falls.J Gen Intern Med.2005;20(2):116–122.
- .The experience of a community hospital in quantifying and reducing patient falls.J Nurs Care Qual.2000;14(3):43–53.
- ,,,.Characteristics of hospital inpatient falls across clinical departments.Gerontology.2008;54(6):342–348.
- ,.Falls risk prediction tools for hospital inpatients: do they work?Nurs Times.2009;105(7):18–21.
- ,,,.Accidental falls in hospital inpatients: evaluation of sensitivity and specificity of two risk assessment tools.J Adv Nurs.2010;66(3):690–696.
- ,,,,,.A systematic review and meta‐analysis of studies using the STRATIFY tool for prediction of falls in hospital patients: how well does it work?Age Ageing.2008;37(6):621–627.
- .Falls risk‐prediction tools for hospital inpatients. Time to put them to bed?Age Ageing.2008;37(3):248–250.
- ,,,,.Evaluation of three fall‐risk assessment tools in an acute care setting.J Adv Nurs.2007;60(4):427–435.
- ,,, et al.Fall prediction in inpatients by bedside nurses using the St. Thomas's Risk Assessment Tool in Falling Elderly Inpatients (STRATIFY) instrument: a multicenter study.J Am Geriatr Soc.2007;55(5):725–733.
- ,,,.Comparison of a fall risk assessment tool with nurses' judgement alone: a cluster‐randomised controlled trial.Age Ageing.2009;38(4):417–423.
- ,,,,.Fall risk‐assessment tools compared with clinical judgment: an evaluation in a rehabilitation ward.Age Ageing.2008;37(3):277–281.
- ,.Incidence of in‐hospital falls in geriatric patients before and after the introduction of an interdisciplinary team‐based fall‐prevention intervention.J Am Geriatr Soc.2007;55(12):2068–2074.
- ,,,.Sustained reduction in serious fall‐related injuries in older people in hospital.Med J Aust.2006;184(8):379–382.
- ,,, et al.The effect of changing practice on fall prevention in a rehabilitative hospital: the Hospital Injury Prevention Study.J Am Geriatr Soc.2004;52(3):335–339.
- ,,,.Effectiveness of targeted falls prevention programme in subacute hospital setting: randomised controlled trial.BMJ.2004;328(7441):676.
- ,,,,.Does delirium contribute to poor hospital outcomes? A three‐site epidemiologic study.J Gen Intern Med.1998;13(4):234–242.
Inpatient falls are the most common type of inpatient adverse event,1 persist as a significant problem nationally, and result in patient injury, increased length of stay, healthcare costs, and litigation.27 Inpatient falls remain a main focus of patient safety and a measure of quality in this era of healthcare reform and quality improvement.8 Inpatient fall rates per 1000 patient‐days range from 1.4 to 18.2.4, 9 The absolute percentage of inpatients that fall ranges from 1.3% to 7%.4, 5, 9, 10 Of inpatient falls, almost all data suggest that roughly one‐third result in some type of injury while 3%‐8% result in serious injury or death.9, 1113
Fall prevention interventions have largely been aimed at modifiable risk factors such as getting out of bed with bed alarms, toileting needs with bedside commodes, and reducing delirium through reorientation techniques. There have been several attempts at decreasing fall rates in hospitals surrounding a multidisciplinary, team‐based approach. Two Cochrane reviews and 2 meta‐analyses have partially examined this issue with mixed results.1417 However, none of these reviews focused on the acute care inpatient population. In fact, the majority of the data analyzed for inpatients was from rehabilitation wards and long‐term care wards. Additionally, there exists almost no data examining fall prevention with single interventions in the acute inpatient population, likely due to the belief that falls are multifactorial in etiology and require more comprehensive interventions.
The aim of this article is to determine the impact of team‐based, multidisciplinary quality improvement efforts to reduce inpatient falls in acute care inpatient hospitals and identify key features that determine their effectiveness.
METHODS
Data Sources and Searches
A search of MEDLINE, CINAHL, EMBASE, and the Cochrane Library was done using the medical subject heading (MeSH) terms accidental falls, accident prevention, inpatients, and prevention and control. Non‐English language publications were included in the search. The search encompassed all published literature through December 1, 2011. In addition, reference lists of all systematic reviews and meta‐analyses were searched to identify all possible studies available.1416
Study Selection
Only primary research studies relating to acute care inpatient hospital fall prevention were included. Data generated exclusively or partially from psychiatric wards, rehabilitation units, subacute facilities, and long‐term facilities were excluded from the review.
Data Extraction and Quality Assessment
Each selected study was carefully hand searched by 2 authors for the purposes of data extraction. Data were collected for the following study characteristics and outcome measures: details of the fall prevention intervention used (allowing for all interventions used to be recorded in Table 3), markers of study quality, study period, study population, mean age of participants, sample size (in 1000 patient‐days), and fall rates (in 1000 patient‐days). In certain cases, sample size was converted to patient‐days using reported data points of total number of patients and average length of stay.
Two authors with experience in fall literature discussed methodological quality and reached a consensus regarding scores using a 20‐point scale previously described in fall literature for all studies included.14, 15 Ten individual criteria were scored on a 0‐2 point scale. No points were awarded when the criteria were not met, not clearly mentioned, or not mentioned at all. One point was awarded when the criterion was partially met, and both points awarded when it was fully met.
Data Synthesis and Analysis
Fall rate per 1000‐patient days was derived from reported data in both intervention and non‐intervention groups within each study. Effect sizes (odds ratios [OR]) and 95% confidence intervals (CI) were derived for individual studies and then combined across research reports using an inverse weighted random‐effects meta‐analysis.18 Random effects methodology was chosen to account for within‐study and between‐study variation. Statistical heterogeneity between trials was assessed using the Cochrane Q statistic and reported as I2, which estimates the percentage of variability across studies that is not due to chance.19 Due to the low number of included studies in our analysis, a formal statistical test on publication bias was not meaningful.20 Statistical significance was defined as P 0.05. Data analyses were done using Comprehensive Meta‐Analysis, Version 2 (Biostat, Englewood, NJ).
RESULTS
Selected Studies
Electronic search produced 259 results on MEDLINE, 2 results from the Cochrane Library, 94 from CINAHL, and 4 from EMBASE. Each result was hand searched to exclude duplicates, and irrelevant studies. Once such data were excluded, the above inclusion and exclusion criteria identified 6 primary articles for review.9, 2125 Additionally, a cluster randomized fall prevention trial in a mixed inpatient population was published by Cumming et al26 in 2008. The study was excluded, as the participants were pooled between rehabilitation wards and acute inpatient wards, and only incomplete data were reported separately for the acute inpatient wards. We were unsuccessful at obtaining necessary data to analyze the acute inpatient wards.
Study Quality
The quality assessment results scores ranged from 11 to 14 out of a possible 20 (Table 1). None of the studies explicitly used an intention‐to‐treat statistical model, as the nature of inpatient care largely prevents drop‐out or crossover, and all patients were included in individual study results.
| Included Study | Clearly Defined Inclusion and Exclusion Criteria | Randomization | Comparable Treatment Groups at Entry | Identical Standard Program for Both Groups | Fall Incident Clearly Defined and Staff Trained in Definition | Blinded Treatment Providers | Blinded Outcome Assessors | Blinded Patients | Identical Appraisal of Outcomes* | Intention‐to‐ Treat Analysis | Total Score (0‐20) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| Dykes et al22 | 2 | 2 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 14 |
| Krauss et al23 | 2 | 0 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 12 |
| Brandis21 | 1 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 10 |
| Mitchell and Jones25 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Schwendimann et al9 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Williams et al24 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
Study Characteristics
The available data are skewed towards elderly patients being hospitalized in general medicine or geriatric units (Table 2). All but 1 study had a large sample size, with 1000‐patient days ranging from 11.1 to 160.3.9, 2124
| Included Study | Study Design | Study Period | Study Wards | Mean Age | Sample Size With Intervention (1000 Patient‐Days) | Sample Size in Control (1000 Patient‐Days) | Fall Rate With Intervention (Falls per 1000 Patient‐Days) | Fall Rate in Control (Falls per 1000 Patient‐Days) |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| Dykes et al22 | RCT | 6 mo | 2 Medical units | 50% 65‐17% 65‐74 33% 75 | 24.1 | 24.1 | 4.18 | 4.64 |
| Krauss et al23 | Quasi‐experimental | 9 mo | General Medicine wards | 65.5 | 11.2 | 11.39 | 5.09 | 6.85 |
| Brandis21 | Pre/post | 12 mo | 500‐Bed acute care hospital | Not reported | 160.3 | 155.2 | 1.61 | 1.74 |
| Mitchell and Jones25 | Pre/post | 6 mo | Acute care hospital | 76.23 (Pre) 72.1 (Post) | 4.3 | 5 | 4.42 | 7.77 |
| Schwendimann et al9 | Pre/post | 4 yr | Internal Med, Surgery, and Geriatrics | 67.3 | 46.8 | 41.9 | 8.6 | 9.1 |
| Williams et al24 | Pre/post | 6 mo | 3 Medical wards and a Geriatrics ward | 79 | 15.88 | 12.53 | 8 | 9.5 |
Components of the Intervention
Multidisciplinary interventions were complex, and formulated based on available evidence for individual interventions and modifiable fall risk factors (Table 3). Each study reviewed included a fall risk assessment to risk‐stratify participants and modulate intervention according to risk.9, 2125
| Included Study | Fall Risk Assessment Used | Mobility Assessment and Assistance if Necessary | Mobility Aid Provided if Necessary | Medication Modification | Education About Risk Factors | Fall Risk Sign/Warning in Chart | Bedside Interventions (eg, Bed Alarm, Rail Adjustment, Bed Location/ Position, etc) | Toileting Schedule | Exercise Program | Other(s) |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Dykes et al22* | + | + | + | + | + | + | + | + | Frequent bed checks, documented fall prevention plan | |
| Krauss et al23 | + | + | + | + | + | + | + | + | Use of bedside interventions was done based on discretion on a case‐by‐case basis | |
| Brandis21 | + | + | + | Ward modifications after OT assessment of patient rooms and bathrooms; hip protectors | ||||||
| Mitchell and Jones25 | + | + | + | + | Introduced detailed system to track fall details; used other preventive actions not specified | |||||
| Schwendimann et al9 | + | + | + | + | + | + | + | + | + | Reassessment of patients who did fall; hip protectors |
| Williams et al24 | + | + | + | + | + | + | Possible sitter | |||
Each study implemented fall prevention programs in a slightly different way. Krauss et al23 used nurses to complete a Morse Fall Scale and subsequently implement several standard interventions based on risk. Staff was then authorized to employ bedside interventions as necessary without systematic data collection. Schwendimann et al9 had nurses complete a simple fall risk assessment (based on history of falls, impaired mobility, and impaired cognition) that prompted the examination by a physician if risk was determined to be high. A subsequent team‐based intervention was employed with nursing, physiotherapy, and the physician. Brandis21 employed a team of nurses and the aid of the Director of Occupational Therapy to assess risk (using an undisclosed system) and carry out an intervention. Dykes et al22 examined an electronic fall prevention tool kit (FPTK) using the electronic medical record (EMR). This intervention began with the Morse Fall Score, which triggered automatically ordered interventions that did not require personal oversight. In fact, the multidisciplinary interventions in the intervention group were also used in the control arm. The difference was the automatic nature in which the interventions were ordered in the interventions arm. Williams et al24 used nurses and physiotherapists, who were specifically trained for the study, to carry out study interventions. The Mitchell and Jones25 study focused on nursing care alone to carry out intervention and used a novel risk assessment tool.
Fall Rates
Dykes et al22 and Williams et al24 found a statistically significant reduction in fall rate with falls reduced by 1.16 per 1000‐patient days and 1.5 per 1000‐patient days, respectively. Mitchell and Jones25 demonstrated a large fall reduction but had an extremely small sample size. Brandis21 found an extremely small reduction in fall rates and failed to report a P‐value. Krauss et al23 showed a trend towards reducing falls, and even showed a statistically significant reduction over the first 5 months of the study, but lost significance in the final 4 months. Similarly, Schwendimann et al9 saw more impressive fall reductions in the first year of the study that dissipated in the final 3 years of data collection.
Results from the meta‐analysis of the 6 studies comparing odds ratios are displayed quantitatively and as a forest plot in Figure 1. The figure shows results with 95% CI for each individual study and overall. There was no statistical evidence of heterogeneity between the studies or study designs. Although, due to the small number of studies included, there is poor power to detect true heterogeneity among studies. The magnitude of boxes shown is a relative sample size indicator. Using the random‐effects model, the summary odds ratio is 0.90 (95% CI, 0.83 to 0.99) (P = 0.02) (I2 = 0%).27

DISCUSSION
The frequency and morbidity associated with inpatient falls is well established, based on reproduced epidemiologic data. Reducing these adverse events could reduce morbidity, mortality, and healthcare costs, and has become the focus of most hospitals quality and patient safety initiatives. The focus of this review was to examine multidisciplinary efforts to reduce falls in acute care inpatient hospitals. Despite the importance and scope of the problem, there is a paucity of research available on this topic, with a wide literature search yielding only 6 primary research studies.
Our major finding is that multidisciplinary fall prevention strategies have a statistically significant impact on fall rates with a combined OR of 0.90. While this review demonstrates a significant benefit to multidisciplinary fall prevention strategies in the acute inpatient population, the clinical impact of these efforts may be limited. Based on rates ranging from 1.7 to 9.5 falls per 1000‐patient days, multidisciplinary interventions would reduce falls by 1 to 10 falls per 10,000‐patient days using the combined OR calculated of 0.9. Using other available incidence data regarding inpatient falls,4, 9 a reasonable baseline frequency to consider would be 8 falls per 1000 patient‐days. Assuming that prevalence, the number needed to treat (NNT) to prevent a single inpatient fall is 1250 patient days. Furthermore, based on available data, only approximately one‐third of these falls result in injury and only a minor fraction of these results in serious injury.9, 1113 The magnitude of this apparent benefit in the context of fall incidence rates raises some concerns about cost‐effectiveness given the high staffing and systems needs that multidisciplinary prevention programs require. This also suggests that there are limitations when using inpatient falls as a measure of healthcare quality given the absence of high‐quality evidence demonstrating a viable solution to the problem. At present, the Center for Medicare and Medicaid services limit reimbursement for fall‐related injuries if they occur during an acute inpatient hospitalization.28
The complexity of the interventions used may help explain the limited impact. Krauss et al23 examined compliance to their interventions and found less than ideal results. They found only 36.4% of intervention floor patients had maintained a toileting schedule compared to 24.6% on control floors. Additionally, a greater proportion of patients on control floors had a physical or occupational therapy consult, and only 1.8% more patients on intervention floors had walking aids provided. These were all strategies emphasized on the intervention floors. Similarly, Schwendimann et al9 questioned their staff's adherence to protocol after fall prevention committee audits. This may help explain why a potential benefit lost statistical significance with time, based on a natural tendency towards more participation at the beginning of a new policy. Williams et al24 reported only a 64% compliance rate with fall care plan forms and 77% rate of missing information on fall care plans. A multidisciplinary fall prevention study that did not meet inclusion criteria (based on study population) yielded strongly positive results for which the authors commented mostly on changing of the hospital culture surrounding fall prevention as a key to their success.29 Adoptability of a multidisciplinary intervention will clearly impact adherence and the intervention's ultimate effectiveness.
Single intervention strategies, not analyzed in this review, are simpler to execute and adhere to. While these types of interventions may be superior, there is extremely limited data supporting or refuting patient fall benefits in the acute care inpatient population when using simple single interventions. However, some data generated partially on acute care geriatrics wards targeting patient education only showed benefit.30
Dykes et al22 was able to improve compliance rates by removing steps in the process of executing interventions with the FPTK built into the EMR. Importantly, the FPTK was compared against very similar fall prevention strategies, the difference being that patients randomized to the FPTK arm had the assessment and interventions automatically prompted on admission in the EMR. Adherence was measured through Morse Fall Scale completion rates (81% in control units versus 94% in intervention units).22 In many ways, the utility of this study was displaying a fall risk reduction by simply enhancing compliance using health information technology with automated alerts. Additionally, both arms of the study reported low fall rates compared to previously reported data, and there may have been larger benefit seen if the FPTK was compared against no fall prevention strategy. This diminishing of effect size may have been present in all studies reviewed, as usual hospital care commonly includes basic patient safety measures.
Another potential problem with the multidisciplinary fall prevention programs included in the meta‐analysis is the inability to target interventions. Each study employed a fall risk score in an attempt to focus resources on a select group of high‐risk patients. This method is problematic given that countless risk factors for inpatient falls have been identified in the literature. Factors that have been described range from clinical characteristics to laboratory tests.31 The most consistently reproducible patient‐related risks are altered mental status (including cognitive impairment and depression), altered mobility (particularly lower limb weakness), a history of falls, and toileting needs.13, 3236 Less consistency is seen with other traditional risk factors such as age, sedating medication, and length of stay.5, 13, 32, 3638 Attempting to risk‐stratify patients using simple and accurate assessment tools developed from these risk factors has proven to be very difficult. Many tools have been developed based on identified risk factors, but perform very poorly when trying to identify patients who will fall with reasonable specificity and positive predictive value.34, 3944 In fact, it has been demonstrated that using a nurse's judgment, a physician's opinion based on a patient's likelihood to wander or a simple 2‐question tool have all performed better than sophisticated risk calculators.33, 45, 46 Therefore, it is possible that interventions could benefit from including all patients, with de‐emphasis on unproven risk stratification tools.
In contrast to our findings, a modest risk reduction has been demonstrated in several primary articles and meta‐analyses in the subacute, rehabilitation, and long‐term care populations.15, 16, 4750 Additionally, a recent study has described a 63.9% risk reduction in a population that included medical, surgical, psychiatric, and rehabilitation wards.29 One important difference between these settings and the acute inpatient populations may be the amount of time and energy that can be dedicated to fall prevention and overall care planning. Another likely factor is the added challenge of preventing falls in patients with more active medical illnesses. In the acute care setting, a patient's chief complaint may not be completely addressed at the time of first mobilization and ambulation. This may be most relevant in patients who are admitted with syncope, seizure, vertigo, and dehydration.
Our study has several limitations; most notably, the available evidence is limited in quality and quantity. Furthermore, omission of unpublished data may also lead to effect bias, though this would likely be in the direction of ineffective interventions supporting a conclusion that multidisciplinary efforts have had only a small impact on fall rates. Ideally, future studies can limit confounding variables through randomization. However, it is difficult to adequately blind when studying a multidisciplinary fall intervention that depends on patient and provider participation. As a result, none of the papers reviewed met criteria for high quality. However, almost all available data examined in this review came from large sample sizes in which thoughtful interventions were used. Since an inpatient fall will not affect the majority of patients, it was crucial for these studies to recruit a large sample size to have adequate power to detect a difference in fall rates. However, each study used risk assessment tools, which are poor indicators of who will and will not fall in the hospital.34, 39, 42 This may suggest a need for improved risk assessment tools, or be further evidence to include all patients in fall prevention regardless of risk. Quantitative synthesis of multidisciplinary fall interventions has the added limitation of comparing complex, multifaceted treatments that are not perfectly uniform. It is our opinion that interventions are semi‐standardized using the grouping methods employed in Table 3.
Preventing inpatient falls remains a difficult issue to address while convincing data is lacking. Based on current evidence, multidisciplinary fall prevention efforts on acutely ill inpatients show a possible small benefit and should be explored from a cost‐effectiveness standpoint to ensure they garner appropriate investment. Many resources are required to run such teams including nursing staff, equipment, physical and occupational therapy staff, pharmacists, and specialized staff training. We are unaware of any such cost‐effectiveness data available. Effective interventions may be those that maximize compliance through health information technology, maintain staff dedication, increase staff availability, improve risk assessment, or include all patients regardless of calculated fall risk, and take the patient's chief complaint into account in the fall prevention strategy. Where resources are limited, it appears most reasonable to focus on major risk factors for inpatient falls that have independently been shown to be detrimental to outcomes, such as delirium.51 Additionally, using inpatient fall rates as a hospital quality measure may be premature, given the lack of proven efforts to lower fall rates. Multidisciplinary fall prevention efforts on acutely ill inpatients should be further studied using high‐quality, randomized trials. It remains to be seen whether these large programs are cost‐effective, or on balance clinically effective.
Inpatient falls are the most common type of inpatient adverse event,1 persist as a significant problem nationally, and result in patient injury, increased length of stay, healthcare costs, and litigation.27 Inpatient falls remain a main focus of patient safety and a measure of quality in this era of healthcare reform and quality improvement.8 Inpatient fall rates per 1000 patient‐days range from 1.4 to 18.2.4, 9 The absolute percentage of inpatients that fall ranges from 1.3% to 7%.4, 5, 9, 10 Of inpatient falls, almost all data suggest that roughly one‐third result in some type of injury while 3%‐8% result in serious injury or death.9, 1113
Fall prevention interventions have largely been aimed at modifiable risk factors such as getting out of bed with bed alarms, toileting needs with bedside commodes, and reducing delirium through reorientation techniques. There have been several attempts at decreasing fall rates in hospitals surrounding a multidisciplinary, team‐based approach. Two Cochrane reviews and 2 meta‐analyses have partially examined this issue with mixed results.1417 However, none of these reviews focused on the acute care inpatient population. In fact, the majority of the data analyzed for inpatients was from rehabilitation wards and long‐term care wards. Additionally, there exists almost no data examining fall prevention with single interventions in the acute inpatient population, likely due to the belief that falls are multifactorial in etiology and require more comprehensive interventions.
The aim of this article is to determine the impact of team‐based, multidisciplinary quality improvement efforts to reduce inpatient falls in acute care inpatient hospitals and identify key features that determine their effectiveness.
METHODS
Data Sources and Searches
A search of MEDLINE, CINAHL, EMBASE, and the Cochrane Library was done using the medical subject heading (MeSH) terms accidental falls, accident prevention, inpatients, and prevention and control. Non‐English language publications were included in the search. The search encompassed all published literature through December 1, 2011. In addition, reference lists of all systematic reviews and meta‐analyses were searched to identify all possible studies available.1416
Study Selection
Only primary research studies relating to acute care inpatient hospital fall prevention were included. Data generated exclusively or partially from psychiatric wards, rehabilitation units, subacute facilities, and long‐term facilities were excluded from the review.
Data Extraction and Quality Assessment
Each selected study was carefully hand searched by 2 authors for the purposes of data extraction. Data were collected for the following study characteristics and outcome measures: details of the fall prevention intervention used (allowing for all interventions used to be recorded in Table 3), markers of study quality, study period, study population, mean age of participants, sample size (in 1000 patient‐days), and fall rates (in 1000 patient‐days). In certain cases, sample size was converted to patient‐days using reported data points of total number of patients and average length of stay.
Two authors with experience in fall literature discussed methodological quality and reached a consensus regarding scores using a 20‐point scale previously described in fall literature for all studies included.14, 15 Ten individual criteria were scored on a 0‐2 point scale. No points were awarded when the criteria were not met, not clearly mentioned, or not mentioned at all. One point was awarded when the criterion was partially met, and both points awarded when it was fully met.
Data Synthesis and Analysis
Fall rate per 1000‐patient days was derived from reported data in both intervention and non‐intervention groups within each study. Effect sizes (odds ratios [OR]) and 95% confidence intervals (CI) were derived for individual studies and then combined across research reports using an inverse weighted random‐effects meta‐analysis.18 Random effects methodology was chosen to account for within‐study and between‐study variation. Statistical heterogeneity between trials was assessed using the Cochrane Q statistic and reported as I2, which estimates the percentage of variability across studies that is not due to chance.19 Due to the low number of included studies in our analysis, a formal statistical test on publication bias was not meaningful.20 Statistical significance was defined as P 0.05. Data analyses were done using Comprehensive Meta‐Analysis, Version 2 (Biostat, Englewood, NJ).
RESULTS
Selected Studies
Electronic search produced 259 results on MEDLINE, 2 results from the Cochrane Library, 94 from CINAHL, and 4 from EMBASE. Each result was hand searched to exclude duplicates, and irrelevant studies. Once such data were excluded, the above inclusion and exclusion criteria identified 6 primary articles for review.9, 2125 Additionally, a cluster randomized fall prevention trial in a mixed inpatient population was published by Cumming et al26 in 2008. The study was excluded, as the participants were pooled between rehabilitation wards and acute inpatient wards, and only incomplete data were reported separately for the acute inpatient wards. We were unsuccessful at obtaining necessary data to analyze the acute inpatient wards.
Study Quality
The quality assessment results scores ranged from 11 to 14 out of a possible 20 (Table 1). None of the studies explicitly used an intention‐to‐treat statistical model, as the nature of inpatient care largely prevents drop‐out or crossover, and all patients were included in individual study results.
| Included Study | Clearly Defined Inclusion and Exclusion Criteria | Randomization | Comparable Treatment Groups at Entry | Identical Standard Program for Both Groups | Fall Incident Clearly Defined and Staff Trained in Definition | Blinded Treatment Providers | Blinded Outcome Assessors | Blinded Patients | Identical Appraisal of Outcomes* | Intention‐to‐ Treat Analysis | Total Score (0‐20) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
| Dykes et al22 | 2 | 2 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 14 |
| Krauss et al23 | 2 | 0 | 2 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 12 |
| Brandis21 | 1 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 10 |
| Mitchell and Jones25 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Schwendimann et al9 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
| Williams et al24 | 2 | 0 | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 2 | 11 |
Study Characteristics
The available data are skewed towards elderly patients being hospitalized in general medicine or geriatric units (Table 2). All but 1 study had a large sample size, with 1000‐patient days ranging from 11.1 to 160.3.9, 2124
| Included Study | Study Design | Study Period | Study Wards | Mean Age | Sample Size With Intervention (1000 Patient‐Days) | Sample Size in Control (1000 Patient‐Days) | Fall Rate With Intervention (Falls per 1000 Patient‐Days) | Fall Rate in Control (Falls per 1000 Patient‐Days) |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| Dykes et al22 | RCT | 6 mo | 2 Medical units | 50% 65‐17% 65‐74 33% 75 | 24.1 | 24.1 | 4.18 | 4.64 |
| Krauss et al23 | Quasi‐experimental | 9 mo | General Medicine wards | 65.5 | 11.2 | 11.39 | 5.09 | 6.85 |
| Brandis21 | Pre/post | 12 mo | 500‐Bed acute care hospital | Not reported | 160.3 | 155.2 | 1.61 | 1.74 |
| Mitchell and Jones25 | Pre/post | 6 mo | Acute care hospital | 76.23 (Pre) 72.1 (Post) | 4.3 | 5 | 4.42 | 7.77 |
| Schwendimann et al9 | Pre/post | 4 yr | Internal Med, Surgery, and Geriatrics | 67.3 | 46.8 | 41.9 | 8.6 | 9.1 |
| Williams et al24 | Pre/post | 6 mo | 3 Medical wards and a Geriatrics ward | 79 | 15.88 | 12.53 | 8 | 9.5 |
Components of the Intervention
Multidisciplinary interventions were complex, and formulated based on available evidence for individual interventions and modifiable fall risk factors (Table 3). Each study reviewed included a fall risk assessment to risk‐stratify participants and modulate intervention according to risk.9, 2125
| Included Study | Fall Risk Assessment Used | Mobility Assessment and Assistance if Necessary | Mobility Aid Provided if Necessary | Medication Modification | Education About Risk Factors | Fall Risk Sign/Warning in Chart | Bedside Interventions (eg, Bed Alarm, Rail Adjustment, Bed Location/ Position, etc) | Toileting Schedule | Exercise Program | Other(s) |
|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||
| Dykes et al22* | + | + | + | + | + | + | + | + | Frequent bed checks, documented fall prevention plan | |
| Krauss et al23 | + | + | + | + | + | + | + | + | Use of bedside interventions was done based on discretion on a case‐by‐case basis | |
| Brandis21 | + | + | + | Ward modifications after OT assessment of patient rooms and bathrooms; hip protectors | ||||||
| Mitchell and Jones25 | + | + | + | + | Introduced detailed system to track fall details; used other preventive actions not specified | |||||
| Schwendimann et al9 | + | + | + | + | + | + | + | + | + | Reassessment of patients who did fall; hip protectors |
| Williams et al24 | + | + | + | + | + | + | Possible sitter | |||
Each study implemented fall prevention programs in a slightly different way. Krauss et al23 used nurses to complete a Morse Fall Scale and subsequently implement several standard interventions based on risk. Staff was then authorized to employ bedside interventions as necessary without systematic data collection. Schwendimann et al9 had nurses complete a simple fall risk assessment (based on history of falls, impaired mobility, and impaired cognition) that prompted the examination by a physician if risk was determined to be high. A subsequent team‐based intervention was employed with nursing, physiotherapy, and the physician. Brandis21 employed a team of nurses and the aid of the Director of Occupational Therapy to assess risk (using an undisclosed system) and carry out an intervention. Dykes et al22 examined an electronic fall prevention tool kit (FPTK) using the electronic medical record (EMR). This intervention began with the Morse Fall Score, which triggered automatically ordered interventions that did not require personal oversight. In fact, the multidisciplinary interventions in the intervention group were also used in the control arm. The difference was the automatic nature in which the interventions were ordered in the interventions arm. Williams et al24 used nurses and physiotherapists, who were specifically trained for the study, to carry out study interventions. The Mitchell and Jones25 study focused on nursing care alone to carry out intervention and used a novel risk assessment tool.
Fall Rates
Dykes et al22 and Williams et al24 found a statistically significant reduction in fall rate with falls reduced by 1.16 per 1000‐patient days and 1.5 per 1000‐patient days, respectively. Mitchell and Jones25 demonstrated a large fall reduction but had an extremely small sample size. Brandis21 found an extremely small reduction in fall rates and failed to report a P‐value. Krauss et al23 showed a trend towards reducing falls, and even showed a statistically significant reduction over the first 5 months of the study, but lost significance in the final 4 months. Similarly, Schwendimann et al9 saw more impressive fall reductions in the first year of the study that dissipated in the final 3 years of data collection.
Results from the meta‐analysis of the 6 studies comparing odds ratios are displayed quantitatively and as a forest plot in Figure 1. The figure shows results with 95% CI for each individual study and overall. There was no statistical evidence of heterogeneity between the studies or study designs. Although, due to the small number of studies included, there is poor power to detect true heterogeneity among studies. The magnitude of boxes shown is a relative sample size indicator. Using the random‐effects model, the summary odds ratio is 0.90 (95% CI, 0.83 to 0.99) (P = 0.02) (I2 = 0%).27

DISCUSSION
The frequency and morbidity associated with inpatient falls is well established, based on reproduced epidemiologic data. Reducing these adverse events could reduce morbidity, mortality, and healthcare costs, and has become the focus of most hospitals quality and patient safety initiatives. The focus of this review was to examine multidisciplinary efforts to reduce falls in acute care inpatient hospitals. Despite the importance and scope of the problem, there is a paucity of research available on this topic, with a wide literature search yielding only 6 primary research studies.
Our major finding is that multidisciplinary fall prevention strategies have a statistically significant impact on fall rates with a combined OR of 0.90. While this review demonstrates a significant benefit to multidisciplinary fall prevention strategies in the acute inpatient population, the clinical impact of these efforts may be limited. Based on rates ranging from 1.7 to 9.5 falls per 1000‐patient days, multidisciplinary interventions would reduce falls by 1 to 10 falls per 10,000‐patient days using the combined OR calculated of 0.9. Using other available incidence data regarding inpatient falls,4, 9 a reasonable baseline frequency to consider would be 8 falls per 1000 patient‐days. Assuming that prevalence, the number needed to treat (NNT) to prevent a single inpatient fall is 1250 patient days. Furthermore, based on available data, only approximately one‐third of these falls result in injury and only a minor fraction of these results in serious injury.9, 1113 The magnitude of this apparent benefit in the context of fall incidence rates raises some concerns about cost‐effectiveness given the high staffing and systems needs that multidisciplinary prevention programs require. This also suggests that there are limitations when using inpatient falls as a measure of healthcare quality given the absence of high‐quality evidence demonstrating a viable solution to the problem. At present, the Center for Medicare and Medicaid services limit reimbursement for fall‐related injuries if they occur during an acute inpatient hospitalization.28
The complexity of the interventions used may help explain the limited impact. Krauss et al23 examined compliance to their interventions and found less than ideal results. They found only 36.4% of intervention floor patients had maintained a toileting schedule compared to 24.6% on control floors. Additionally, a greater proportion of patients on control floors had a physical or occupational therapy consult, and only 1.8% more patients on intervention floors had walking aids provided. These were all strategies emphasized on the intervention floors. Similarly, Schwendimann et al9 questioned their staff's adherence to protocol after fall prevention committee audits. This may help explain why a potential benefit lost statistical significance with time, based on a natural tendency towards more participation at the beginning of a new policy. Williams et al24 reported only a 64% compliance rate with fall care plan forms and 77% rate of missing information on fall care plans. A multidisciplinary fall prevention study that did not meet inclusion criteria (based on study population) yielded strongly positive results for which the authors commented mostly on changing of the hospital culture surrounding fall prevention as a key to their success.29 Adoptability of a multidisciplinary intervention will clearly impact adherence and the intervention's ultimate effectiveness.
Single intervention strategies, not analyzed in this review, are simpler to execute and adhere to. While these types of interventions may be superior, there is extremely limited data supporting or refuting patient fall benefits in the acute care inpatient population when using simple single interventions. However, some data generated partially on acute care geriatrics wards targeting patient education only showed benefit.30
Dykes et al22 was able to improve compliance rates by removing steps in the process of executing interventions with the FPTK built into the EMR. Importantly, the FPTK was compared against very similar fall prevention strategies, the difference being that patients randomized to the FPTK arm had the assessment and interventions automatically prompted on admission in the EMR. Adherence was measured through Morse Fall Scale completion rates (81% in control units versus 94% in intervention units).22 In many ways, the utility of this study was displaying a fall risk reduction by simply enhancing compliance using health information technology with automated alerts. Additionally, both arms of the study reported low fall rates compared to previously reported data, and there may have been larger benefit seen if the FPTK was compared against no fall prevention strategy. This diminishing of effect size may have been present in all studies reviewed, as usual hospital care commonly includes basic patient safety measures.
Another potential problem with the multidisciplinary fall prevention programs included in the meta‐analysis is the inability to target interventions. Each study employed a fall risk score in an attempt to focus resources on a select group of high‐risk patients. This method is problematic given that countless risk factors for inpatient falls have been identified in the literature. Factors that have been described range from clinical characteristics to laboratory tests.31 The most consistently reproducible patient‐related risks are altered mental status (including cognitive impairment and depression), altered mobility (particularly lower limb weakness), a history of falls, and toileting needs.13, 3236 Less consistency is seen with other traditional risk factors such as age, sedating medication, and length of stay.5, 13, 32, 3638 Attempting to risk‐stratify patients using simple and accurate assessment tools developed from these risk factors has proven to be very difficult. Many tools have been developed based on identified risk factors, but perform very poorly when trying to identify patients who will fall with reasonable specificity and positive predictive value.34, 3944 In fact, it has been demonstrated that using a nurse's judgment, a physician's opinion based on a patient's likelihood to wander or a simple 2‐question tool have all performed better than sophisticated risk calculators.33, 45, 46 Therefore, it is possible that interventions could benefit from including all patients, with de‐emphasis on unproven risk stratification tools.
In contrast to our findings, a modest risk reduction has been demonstrated in several primary articles and meta‐analyses in the subacute, rehabilitation, and long‐term care populations.15, 16, 4750 Additionally, a recent study has described a 63.9% risk reduction in a population that included medical, surgical, psychiatric, and rehabilitation wards.29 One important difference between these settings and the acute inpatient populations may be the amount of time and energy that can be dedicated to fall prevention and overall care planning. Another likely factor is the added challenge of preventing falls in patients with more active medical illnesses. In the acute care setting, a patient's chief complaint may not be completely addressed at the time of first mobilization and ambulation. This may be most relevant in patients who are admitted with syncope, seizure, vertigo, and dehydration.
Our study has several limitations; most notably, the available evidence is limited in quality and quantity. Furthermore, omission of unpublished data may also lead to effect bias, though this would likely be in the direction of ineffective interventions supporting a conclusion that multidisciplinary efforts have had only a small impact on fall rates. Ideally, future studies can limit confounding variables through randomization. However, it is difficult to adequately blind when studying a multidisciplinary fall intervention that depends on patient and provider participation. As a result, none of the papers reviewed met criteria for high quality. However, almost all available data examined in this review came from large sample sizes in which thoughtful interventions were used. Since an inpatient fall will not affect the majority of patients, it was crucial for these studies to recruit a large sample size to have adequate power to detect a difference in fall rates. However, each study used risk assessment tools, which are poor indicators of who will and will not fall in the hospital.34, 39, 42 This may suggest a need for improved risk assessment tools, or be further evidence to include all patients in fall prevention regardless of risk. Quantitative synthesis of multidisciplinary fall interventions has the added limitation of comparing complex, multifaceted treatments that are not perfectly uniform. It is our opinion that interventions are semi‐standardized using the grouping methods employed in Table 3.
Preventing inpatient falls remains a difficult issue to address while convincing data is lacking. Based on current evidence, multidisciplinary fall prevention efforts on acutely ill inpatients show a possible small benefit and should be explored from a cost‐effectiveness standpoint to ensure they garner appropriate investment. Many resources are required to run such teams including nursing staff, equipment, physical and occupational therapy staff, pharmacists, and specialized staff training. We are unaware of any such cost‐effectiveness data available. Effective interventions may be those that maximize compliance through health information technology, maintain staff dedication, increase staff availability, improve risk assessment, or include all patients regardless of calculated fall risk, and take the patient's chief complaint into account in the fall prevention strategy. Where resources are limited, it appears most reasonable to focus on major risk factors for inpatient falls that have independently been shown to be detrimental to outcomes, such as delirium.51 Additionally, using inpatient fall rates as a hospital quality measure may be premature, given the lack of proven efforts to lower fall rates. Multidisciplinary fall prevention efforts on acutely ill inpatients should be further studied using high‐quality, randomized trials. It remains to be seen whether these large programs are cost‐effective, or on balance clinically effective.
- ,,.Patient accidents in hospital: incidence, documentation and significance.Br J Clin Pract.1994;48(2):63–66.
- ,,,.Serious falls in hospitalized patients: correlates and resource utilization.Am J Med.1995;99(2):137–143.
- ,,,.Hospital falls: a persistent problem.Am J Public Health.1985;75(7):775–777.
- ,,.Falls in the acute hospital setting—impact on resource utilisation.Aust Health Rev.2007;31(3):471–477.
- ,,.Incidence and risk factors for inpatient falls in an academic acute‐care hospital.J Nippon Med Sch.2006;73(5):265–270.
- ,,,.Do falls and falls‐injuries in hospital indicate negligent care—and how big is the risk? A retrospective analysis of the NHS Litigation Authority Database of clinical negligence claims, resulting from falls in hospitals in England 1995 to 2006.Qual Saf Health Care.2008;17(6):431–436.
- ,,,.Hospital falls: development of a predictive model for clinical practice.Appl Nurs Res.1995;8(3):129–139.
- ,.Centers for Medicare and Medicaid Services' “never events”: an analysis and recommendations to hospitals.Health Care Manag (Frederick).2008;27(4):338–349.
- ,,,.Falls and consequent injuries in hospitalized patients: effects of an interdisciplinary falls prevention program.BMC Health Serv Res.2006;6:69.
- .Immobility and falls.Clin Geriatr Med.1998;14(4):699–726.
- ,,.A case control study of falls in the hospital setting.J Gerontol Nurs.1998;24(12):7–15.
- ,,,.A retrospective analysis of patient falls.Can J Public Health.1985;76(2):116–118.
- ,,, et al.Characteristics and circumstances of falls in a hospital setting: a prospective analysis.J Gen Intern Med.2004;19(7):732–739.
- ,,, et al.Interventions for preventing falls in older people in nursing care facilities and hospitals.Cochrane Database Syst Rev.2010(1):CD005465.
- , L,,,,.Interventions for preventing falls in acute‐ and chronic‐care hospitals: a systematic review and meta‐analysis.J Am Geriatr Soc.2008;56(1):29–36.
- ,,, et al.Strategies to prevent falls and fractures in hospitals and care homes and effect of cognitive impairment: systematic review and meta‐analyses.BMJ.2007;334(7584):82.
- ,,,,,.Interventions for preventing falls in elderly people.Cochrane Database Syst Rev.2003(4):CD000340.
- ,,,.Introduction to Meta‐Analysis.Chichester, UK:John Wiley 2009.
- ,.Quantifying heterogeneity in a meta‐analysis.Stat Med.2002;21(11):1539–1558.
- .The power of the standard test for the presence of heterogeneity in meta‐analysis.Stat Med.2006;25(15):2688–2699.
- .A collaborative occupational therapy and nursing approach to falls prevention in hospital inpatients.J Qual Clin Pract.1999;19(4):215–220.
- ,,, et al.Fall prevention in acute care hospitals: a randomized trial.JAMA.2010;304(17):1912–1918.
- ,,,,,.Intervention to prevent falls on the medical service in a teaching hospital.Infect Control Hosp Epidemiol.2008;29(6):539–545.
- ,,, et al.Evaluation of a falls prevention programme in an acute tertiary care hospital.J Clin Nurs.2007;16(2):316–324.
- ,.Striving to prevent falls in an acute care setting—action to enhance quality.J Clin Nurs.1996;5(4):213–220.
- ,,, et al.Cluster randomised trial of a targeted multifactorial intervention to prevent falls among older people in hospital.BMJ.2008;336(7647):758–760.
- ,.Graphical displays for meta‐analysis: an overview with suggestions for practice.Res Syn Meth.2010(1):66–80.
- Centers for Medicare 37(7):317–325.
- ,‐M,, et al.Patient education to prevent falls among older hospital inpatients: a randomized controlled trial.Arch Intern Med.2011;117:516–524.
- ,.The relationship between patient blood pathology values and patient falls in an acute‐care setting: a retrospective analysis.Int J Nurs Pract.2005;11(4):161–168.
- ,,,.Falls risk factors in the hospital setting: a systematic review.Int J Nurs Pract.2001;7(1):38–45.
- ,,.A simplified fall‐risk assessment tool for patients hospitalized in medical wards.Isr Med Assoc J.2008;10(2):125–129.
- ,,,.Risk factors and risk assessment tools for falls in hospital in‐patients: a systematic review.Age Ageing.2004;33(2):122–130.
- ,,, et al.Circumstances of patient falls and injuries in 9 hospitals in a midwestern healthcare system.Infect Control Hosp Epidemiol.2007;28(5):544–550.
- ,,, et al.A case‐control study of patient, medication, and care‐related risk factors for inpatient falls.J Gen Intern Med.2005;20(2):116–122.
- .The experience of a community hospital in quantifying and reducing patient falls.J Nurs Care Qual.2000;14(3):43–53.
- ,,,.Characteristics of hospital inpatient falls across clinical departments.Gerontology.2008;54(6):342–348.
- ,.Falls risk prediction tools for hospital inpatients: do they work?Nurs Times.2009;105(7):18–21.
- ,,,.Accidental falls in hospital inpatients: evaluation of sensitivity and specificity of two risk assessment tools.J Adv Nurs.2010;66(3):690–696.
- ,,,,,.A systematic review and meta‐analysis of studies using the STRATIFY tool for prediction of falls in hospital patients: how well does it work?Age Ageing.2008;37(6):621–627.
- .Falls risk‐prediction tools for hospital inpatients. Time to put them to bed?Age Ageing.2008;37(3):248–250.
- ,,,,.Evaluation of three fall‐risk assessment tools in an acute care setting.J Adv Nurs.2007;60(4):427–435.
- ,,, et al.Fall prediction in inpatients by bedside nurses using the St. Thomas's Risk Assessment Tool in Falling Elderly Inpatients (STRATIFY) instrument: a multicenter study.J Am Geriatr Soc.2007;55(5):725–733.
- ,,,.Comparison of a fall risk assessment tool with nurses' judgement alone: a cluster‐randomised controlled trial.Age Ageing.2009;38(4):417–423.
- ,,,,.Fall risk‐assessment tools compared with clinical judgment: an evaluation in a rehabilitation ward.Age Ageing.2008;37(3):277–281.
- ,.Incidence of in‐hospital falls in geriatric patients before and after the introduction of an interdisciplinary team‐based fall‐prevention intervention.J Am Geriatr Soc.2007;55(12):2068–2074.
- ,,,.Sustained reduction in serious fall‐related injuries in older people in hospital.Med J Aust.2006;184(8):379–382.
- ,,, et al.The effect of changing practice on fall prevention in a rehabilitative hospital: the Hospital Injury Prevention Study.J Am Geriatr Soc.2004;52(3):335–339.
- ,,,.Effectiveness of targeted falls prevention programme in subacute hospital setting: randomised controlled trial.BMJ.2004;328(7441):676.
- ,,,,.Does delirium contribute to poor hospital outcomes? A three‐site epidemiologic study.J Gen Intern Med.1998;13(4):234–242.
- ,,.Patient accidents in hospital: incidence, documentation and significance.Br J Clin Pract.1994;48(2):63–66.
- ,,,.Serious falls in hospitalized patients: correlates and resource utilization.Am J Med.1995;99(2):137–143.
- ,,,.Hospital falls: a persistent problem.Am J Public Health.1985;75(7):775–777.
- ,,.Falls in the acute hospital setting—impact on resource utilisation.Aust Health Rev.2007;31(3):471–477.
- ,,.Incidence and risk factors for inpatient falls in an academic acute‐care hospital.J Nippon Med Sch.2006;73(5):265–270.
- ,,,.Do falls and falls‐injuries in hospital indicate negligent care—and how big is the risk? A retrospective analysis of the NHS Litigation Authority Database of clinical negligence claims, resulting from falls in hospitals in England 1995 to 2006.Qual Saf Health Care.2008;17(6):431–436.
- ,,,.Hospital falls: development of a predictive model for clinical practice.Appl Nurs Res.1995;8(3):129–139.
- ,.Centers for Medicare and Medicaid Services' “never events”: an analysis and recommendations to hospitals.Health Care Manag (Frederick).2008;27(4):338–349.
- ,,,.Falls and consequent injuries in hospitalized patients: effects of an interdisciplinary falls prevention program.BMC Health Serv Res.2006;6:69.
- .Immobility and falls.Clin Geriatr Med.1998;14(4):699–726.
- ,,.A case control study of falls in the hospital setting.J Gerontol Nurs.1998;24(12):7–15.
- ,,,.A retrospective analysis of patient falls.Can J Public Health.1985;76(2):116–118.
- ,,, et al.Characteristics and circumstances of falls in a hospital setting: a prospective analysis.J Gen Intern Med.2004;19(7):732–739.
- ,,, et al.Interventions for preventing falls in older people in nursing care facilities and hospitals.Cochrane Database Syst Rev.2010(1):CD005465.
- , L,,,,.Interventions for preventing falls in acute‐ and chronic‐care hospitals: a systematic review and meta‐analysis.J Am Geriatr Soc.2008;56(1):29–36.
- ,,, et al.Strategies to prevent falls and fractures in hospitals and care homes and effect of cognitive impairment: systematic review and meta‐analyses.BMJ.2007;334(7584):82.
- ,,,,,.Interventions for preventing falls in elderly people.Cochrane Database Syst Rev.2003(4):CD000340.
- ,,,.Introduction to Meta‐Analysis.Chichester, UK:John Wiley 2009.
- ,.Quantifying heterogeneity in a meta‐analysis.Stat Med.2002;21(11):1539–1558.
- .The power of the standard test for the presence of heterogeneity in meta‐analysis.Stat Med.2006;25(15):2688–2699.
- .A collaborative occupational therapy and nursing approach to falls prevention in hospital inpatients.J Qual Clin Pract.1999;19(4):215–220.
- ,,, et al.Fall prevention in acute care hospitals: a randomized trial.JAMA.2010;304(17):1912–1918.
- ,,,,,.Intervention to prevent falls on the medical service in a teaching hospital.Infect Control Hosp Epidemiol.2008;29(6):539–545.
- ,,, et al.Evaluation of a falls prevention programme in an acute tertiary care hospital.J Clin Nurs.2007;16(2):316–324.
- ,.Striving to prevent falls in an acute care setting—action to enhance quality.J Clin Nurs.1996;5(4):213–220.
- ,,, et al.Cluster randomised trial of a targeted multifactorial intervention to prevent falls among older people in hospital.BMJ.2008;336(7647):758–760.
- ,.Graphical displays for meta‐analysis: an overview with suggestions for practice.Res Syn Meth.2010(1):66–80.
- Centers for Medicare 37(7):317–325.
- ,‐M,, et al.Patient education to prevent falls among older hospital inpatients: a randomized controlled trial.Arch Intern Med.2011;117:516–524.
- ,.The relationship between patient blood pathology values and patient falls in an acute‐care setting: a retrospective analysis.Int J Nurs Pract.2005;11(4):161–168.
- ,,,.Falls risk factors in the hospital setting: a systematic review.Int J Nurs Pract.2001;7(1):38–45.
- ,,.A simplified fall‐risk assessment tool for patients hospitalized in medical wards.Isr Med Assoc J.2008;10(2):125–129.
- ,,,.Risk factors and risk assessment tools for falls in hospital in‐patients: a systematic review.Age Ageing.2004;33(2):122–130.
- ,,, et al.Circumstances of patient falls and injuries in 9 hospitals in a midwestern healthcare system.Infect Control Hosp Epidemiol.2007;28(5):544–550.
- ,,, et al.A case‐control study of patient, medication, and care‐related risk factors for inpatient falls.J Gen Intern Med.2005;20(2):116–122.
- .The experience of a community hospital in quantifying and reducing patient falls.J Nurs Care Qual.2000;14(3):43–53.
- ,,,.Characteristics of hospital inpatient falls across clinical departments.Gerontology.2008;54(6):342–348.
- ,.Falls risk prediction tools for hospital inpatients: do they work?Nurs Times.2009;105(7):18–21.
- ,,,.Accidental falls in hospital inpatients: evaluation of sensitivity and specificity of two risk assessment tools.J Adv Nurs.2010;66(3):690–696.
- ,,,,,.A systematic review and meta‐analysis of studies using the STRATIFY tool for prediction of falls in hospital patients: how well does it work?Age Ageing.2008;37(6):621–627.
- .Falls risk‐prediction tools for hospital inpatients. Time to put them to bed?Age Ageing.2008;37(3):248–250.
- ,,,,.Evaluation of three fall‐risk assessment tools in an acute care setting.J Adv Nurs.2007;60(4):427–435.
- ,,, et al.Fall prediction in inpatients by bedside nurses using the St. Thomas's Risk Assessment Tool in Falling Elderly Inpatients (STRATIFY) instrument: a multicenter study.J Am Geriatr Soc.2007;55(5):725–733.
- ,,,.Comparison of a fall risk assessment tool with nurses' judgement alone: a cluster‐randomised controlled trial.Age Ageing.2009;38(4):417–423.
- ,,,,.Fall risk‐assessment tools compared with clinical judgment: an evaluation in a rehabilitation ward.Age Ageing.2008;37(3):277–281.
- ,.Incidence of in‐hospital falls in geriatric patients before and after the introduction of an interdisciplinary team‐based fall‐prevention intervention.J Am Geriatr Soc.2007;55(12):2068–2074.
- ,,,.Sustained reduction in serious fall‐related injuries in older people in hospital.Med J Aust.2006;184(8):379–382.
- ,,, et al.The effect of changing practice on fall prevention in a rehabilitative hospital: the Hospital Injury Prevention Study.J Am Geriatr Soc.2004;52(3):335–339.
- ,,,.Effectiveness of targeted falls prevention programme in subacute hospital setting: randomised controlled trial.BMJ.2004;328(7441):676.
- ,,,,.Does delirium contribute to poor hospital outcomes? A three‐site epidemiologic study.J Gen Intern Med.1998;13(4):234–242.
RN‐Pharmacist Medication Reconciliation
Adverse drug events (ADE), of which medication errors are one form, refer to harm caused by use of a drug. ADEs occur frequently and are associated with an increased length of stay, economic burden, and risk of death.1, 2 Classen et al and Bates et al estimate, respectively, that there are 1.2 to 1.8 preventable ADEs per 100 inpatient admissions.1, 3 Adjusting these data to current levels of yearly admissions, 380,000 to 400,000 preventable ADEs occur each year, and are projected to cost upwards of $3.5 billion annually in 2006 dollars.4
Medication reconciliation is an active process that occurs at transitions in care (admissions, transfers in level of care, and discharge) and is designed to prevent medication errors as the patient moves across the continuum of care. Medications used by the patient prior to hospitalization are considered when developing the inpatient therapeutic regimen.
Medications are ordered on admission based in part on what providers believe is the patient's home medication list (HML). A systematic review revealed that errors in medication history taking, including errors of omission and commission, are extremely common and clinically important.5 Such inaccuracies lead to unintended discrepancies between the hospital medication orders and the patient's true home medication regimen, and can result in patient harm.
Numerous studies have documented that inpatient discrepancies are common.610 From September 2004 to July 2005, data from the United States Pharmacopeia MEDMARX voluntary medication error reporting program revealed over 2000 medication errors associated with reconciliation failures: 22% occurred during admission and 12% occurred at time of discharge.11 A Canadian study demonstrated that 81 of 151 enrolled patients, who were prescribed 4 or more medications and were admitted to a medicine service, had at least 1 unintended discrepancy.6 Of those discrepancies, 38.6% were thought to have the potential to cause moderate or severe discomfort or clinical deterioration. Bates et al found that 0.9% of all inpatient medication errors lead to harm.12
The Joint Commission highlighted the importance of this problem by creating National Patient Safety Goal (NPSG) 8 in 2005, Accurately and completely reconcile medications across the continuum of care.13 This goal was modified and became effective on July 1, 2011.14 As a response, organizations have been developing physician‐led, nurse‐led, or pharmacist‐led medication reconciliation processes.8, 1522 Typically, these teams have time dedicated to producing the most accurate home list possible, a gold standard list. Examples of successful pharmacist‐led interventions to address this goal are described by investigators at Northwestern Memorial Hospital16 and Duke University Medical Center.23 Other interventions implemented to improve the reconciliation process include computerized provider order entry (CPOE) systems24 and combining information technology (IT) with process redesign involving physicians, pharmacists, and nurses.20 While the literature shows that there are multiple interventions that can reduce medication reconciliation errors, there is a dearth of evidence for interventions that are low‐cost and easily replicable.
Given that unintended medication discrepancies are common and harmful, we sought to develop a generalizable intervention. Our prospective pilot study explored whether an easily replicable nurse‐pharmacist led medication reconciliation process could efficiently and inexpensively identify unintended medication discrepancies, thereby preventing potential adverse drug events (PADEs).
METHODS
Patient Selection
The study was conducted at a 1000 bed urban, tertiary care hospital that serves a diverse patient population. We enrolled eligible patients over a 15‐month period, from January 2008 to March 2009, admitted to 2 resident‐covered general medicine teams. Each team is composed of an attending physician, 2 senior residents, 4 interns, a case manager, a pharmacist, and a social worker. Patients were excluded only if they did not consent or were discharged from the hospital in less than 24 hours. Patients were interviewed Monday through Friday, and those admitted over the weekend were interviewed the following Monday. The study was approved by The Johns Hopkins institutional review board (IRB).
Intervention Team
Baccalaureate‐prepared registered nurses (RNs) provided the primary intervention in this model. Both nurses had practiced as bedside clinicians at the hospital and had knowledge of hospital systems and structures. No additional training was provided.
The study pharmacist, who is board certified in pharmacotherapy, has a doctor of pharmacy degree and completed 1 year of Pharmacy Practice residency, as well as a 1‐year specialty residency in Internal Medicine. She spends the majority of her clinical time rounding with the inpatient medicine teams where she provides medication management recommendations.
Home Medication List Compilation
Informed consent was obtained by the study nurse 24‐48 hours after admission. The nurse completed an initial patient interview to determine the HML or preadmission medication list. The patient‐reported HML was compared to the history obtained by the physician. If both lists matched, the HML was considered complete. If a patient was not able to provide a written HML or recall medications, the nurses reviewed the electronic patient record (EPR), which documents previous discharge medication lists and Hopkins outpatient medication lists. If not convinced that the HML was accurate or complete, the nurses could use other sources of information, including patients' families, primary care physicians, and community pharmacies. Patients were then asked to verify the HML. At the start of the study, the nurses created a handwritten HML that they placed in the chart. As functionality of the CPOE improved during the study, the nurses entered the lists into the CPOE instead.
Reconciling the HML with the Admission Orders and Discharge Medication List
The nurses created the HML during the first 24‐48 hours of a patient's admission, so admission orders were entered before the resident physicians were aware of the nurse‐complied HML. By comparing the active medication orders to the HML, the nurses created a list of admission discrepancies. The nurse evaluated the discrepancies in the context of the treatment plan to determine if they appeared to be intended or unintended. The nurses consulted the study pharmacist if they were unsure if a discrepancy was intended. Questions about specific drug substitutions were clarified with the study pharmacist. For example, the nurses consulted the pharmacist about a patient who was taking carvedilol at home but was changed to metoprolol during the hospitalization.
After consultation with the pharmacist, the nurse reviewed all remaining potential unintended admission discrepancies with the physician team. A similar process was repeated on the day of discharge. For all patients transitioning out of the hospital, the nurses compared the active medication list and the HML to the discharge worksheet medication list and patient instructions. The nurses contacted the physician team when potential unintended discrepancies were identified. If unintended discrepancies were confirmed for a patient who had already been discharged earlier that day, a resolution plan was determined and the patient was contacted.
Intended Versus Unintended Discrepancies
After completing the nurse‐pharmacist review, the nurse presented the admission discrepancies that were thought to be unintended to the prescriber. If the medication order was not changed, the discrepancy was considered intended. If the prescriber changed the order, the discrepancy was considered unintended. Unintended discrepancies were identified within 48 hours of admission and also upon discharge. If an unintended discrepancy was identified during admission, it only counted as an admission discrepancy. However, if the same mistake occurred again during discharge, the unintended discrepancy was also counted as a discharge discrepancy. The nurses classified the discrepancy by type: medication omission, frequency error, route error, wrong dose, and/or wrong drug.
Rating Potential Harm From the Unintended Discrepancies
Adjudicators assessed the potential harm of unintended admission discrepancies that could occur during an average 4‐day hospitalization. Similarly, raters assessed the potential harm of unintended discharge medication discrepancies. Each of the 4 adjudicators, 2 physicians and 2 pharmacists, were blinded and independently adjudicated all unintended admission and discharge discrepancies, rating the potential harm from the discrepancy on a scale6 from 1 to 3:
Rank 1: unlikely to cause any harm or discomfort.
Rank 2: potential to cause moderate discomfort or clinical deterioration.
Rank 3: potential for severe discomfort or clinical deterioration.
To rate the potential harm of the discrepancies, the raters were provided with the patients' diagnoses, the medications in question, the types of discrepancies, and whether the discrepancy occurred at admission or discharge. The final potential harm level was determined by the majority rating unless a rating spread of Rank 1 to Rank 3 existed. In that case, consensus was reached by discussion. If the 4 raters were evenly split, the mean value was used.
Cost Analysis: Resources, Valuation, and Cost Savings
The time involved in implementing the protocol was recorded in minutes on an Excel spreadsheet. The time records included: participant interview, contact with secondary sources for medication history (a primary care physician or pharmacy), consultation with the study pharmacist, patient education, discharge activities, and consultation with the prescribers. The study pharmacist submitted additional time for follow‐up of issues after the initial consultation with the nurse.
The cost of an ADE in our study was estimated based on the Bates et al study, which reported an average cost of $5857.00 per inpatient ADE in 1996 dollars.2 Using an inflation adjustment ratio from 1996 to 2008 of 1.595, we calculated the cost of an ADE in 2008 dollars to be an average of $9344.12.
We compared the cost of the program with potential cost savings. We performed a threshold analysis to determine the minimum proportion of Rank 2‐3 potential ADEs that would actually need to result in harm in order for the intervention to yield a cost savings. We also estimated the costs of harm based on the Bates et al study which found that 0.9% of all inpatient medication errors results in harm.12
Statistical Analysis
We used logistic regression to test for associations between discrepancies and patient characteristics including age, race, length of stay, education, marital status, primary payor, severity of illness, and number of medications. The outcome measure was at least 1 unintended discrepancy on admission or discharge. A paired‐samples t test was calculated to compare the mean number of discrepancies on admission to the mean discrepancies at discharge.
RESULTS
We enrolled 563 patients who were admitted a total of 698 times. Only the first admission for each patient was analyzed. Patient demographics are presented in Table 1. Almost 70% of our enrolled patients were less than 65 years old, 65% of the patients were black, 58% lived within 5 miles of the Johns Hopkins Hospital, and a plurality of the patients were single and received Medicare. The mean number of medications was 7.8 (SD 4.9).
| Demographic Variable | Percentage |
|---|---|
| Demographic Variable | Mean SD |
| |
| Sex | |
| Male | 49.2 |
| Female | 50.8 |
| Age | |
| <65 | 69.5 |
| 65 to <85 | 27.5 |
| 85 | 3.0 |
| Marital status | |
| Single | 47.0 |
| Married | 30.0 |
| Divorced | 10.5 |
| Widowed | 10.7 |
| Payor | |
| Medicare | 42.3 |
| Medicaid | 25.0 |
| HMO | 8.0 |
| Self‐pay | 9.2 |
| Race | |
| White | 33.0 |
| Black | 65.1 |
| Other | 1.9 |
| APDRG complexity 1‐4 | |
| 1 | 5.2 |
| 2 | 28.5 |
| 3 | 47.4 |
| 4 | 18.9 |
| Education | |
| Less than eighth grade | 9.2 |
| Some high school | 21.7 |
| High school or GED | 28.9 |
| Some college | 15.1 |
| College degree or greater | 19.5 |
| Chose not to answer | 5.5 |
| ICD‐9 codes | |
| Diseases of the circulatory system | 25.5 |
| Diseases of the respiratory system | 11.1 |
| Diseases of the digestive system | 10.9 |
| Symptoms, signs, and ill‐defined conditions | 10.7 |
| Admission from the Emergency Room | 87 |
| Patient lives within 5 miles of the hospital | 58 |
| Age (years) | 55.4 16.6 |
| Number of medications per patient | 7.76 4.9 |
| Length of stay | 5.72 7.28 |
The most frequent source of the home medication list was from patient verbal recall (52%). Few patients had lists of their current medications when admitted. The second most commonly used source was the electronic patient record, 36.6%, which was used to verify and complete the home list. The patient's community pharmacist, 12.5%, was contacted when other sources did not result in a complete home list. The primary care site was contacted in 6.0% of the cases. Patients were then asked to verify the HML.
Of the 563 patients, 225 (40%; 95% confidence interval [CI], 36%‐44%) had at least 1 unintended discrepancy on admission or discharge. On admission and discharge, 28% (95% CI, 25%‐30%) and 25% (95% CI, 21%‐29%) of the patients, respectively, had an unintended discrepancy. Of those 225 patients who had an unintended discrepancy, 162 (72%) had a discrepancy ranked 2 or 3 on the potential harm scale.
Overall, there were more unintended discrepancies on admission (364) than at discharge (167) (Figure 1). The paired t test showed a significant decrease (t[562] = 2.066, P = 0.039) between the number of discrepancies on admission to discharge. However, the majority of these discrepancies on admission (55%) were Rank 1 on the potential harm scale, while the majority of the discharge discrepancies (85%) were likely to cause harm (Rank 2‐3). There were many more Rank 3 discrepancies upon discharge, 39, than on admission, 13. The percentage of Rank 2‐3 discrepancies on admission and discharge were 45% versus 85%, respectively. Interclass correlation of ratings before consensus was 0.58.
The most common unintended discrepancies were omissions of medications at admission, 74%, and discharge, 62%, followed by discrepancies in dosing (Table 2). The majority of omission discrepancies were categorized as Rank 1. Discrepancies in frequency and dosing were most likely to be adjudicated as Rank 2 or 3. Table 3 gives examples of how discrepancies were ranked.
| a. Type of Discrepancy on Admission | Total | Rank 1 | Rank 2 | Rank 3 |
|---|---|---|---|---|
| 364 (%) | 199 (%) | 152 (%) | 13 (%) | |
| ||||
| Omission | 270 (74) | 157 (79) | 102 (67) | 11 (85) |
| Frequency | 19 (5) | 7 (4) | 12 (8) | 0 |
| Route | 3 (1) | 1 (1) | 2 (1) | 0 |
| Dose | 54 (15) | 23 (12) | 29 (19) | 2 (15) |
| Drug | 18 (5) | 11 (5) | 7 (5) | 0 |
| b. Type of Discrepancy on Discharge | Total | Rank 1 | Rank 2 | Rank 3 |
| 167 (%) | 43 (%) | 85 (%) | 39 (%) | |
| Omission | 104 (62) | 37 (86) | 46 (54) | 21 (54) |
| Frequency | 15 (9) | 3 (7) | 10 (12) | 2 (5) |
| Route | 12 (7) | 2 (5) | 6 (7) | 4 (10) |
| Dose | 22 (13) | 0 | 14 (16) | 8 (21) |
| Drug | 14 (8) | 1 (2) | 9 (11) | 4 (10) |
| Rank | Time of Discrepancy | Clinical Information |
|---|---|---|
| ||
| 1 | Discharge | Elderly patient with sepsis from acute cystitis. Centrum Silver, part of the HML, was not on the discharge orders. |
| 2 | Discharge | Patient admitted with UTI. Metoprolol XL 100 mg was on the patient's HML but not on the discharge orders. |
| 3 | Admission | Patient admitted with hypertensive urgency. Clonidine 0.2 mg by mouth 3 times daily, which was on the patient's HML, was omitted. |
The only statistically significantly variable associated with the presence of discrepancies was the number of medications (odds ratio, 1.087; 95% CI, 1.044‐1.132). Each additional medication increased the odds of a discrepancy by 8.7%. Other variables, including age, race, length of stay, level of education, marital status, primary payor, and severity of illness, were not associated with prevalent discrepancies.
Cost Analysis: Resources, Utilization, and Cost Savings
On average, the nurses spent 11.2 minutes (SD 8.0 minutes) of their time conducting the admitting patient interview. The average total time for the protocol excluding the initial interview was 29.3 minutes (SD 30.2 minutes). The clinical pharmacist was consulted in 30% of the cases. The average consultation time was 7.5 minutes (SD 4.4). We determined the hospital's cost of the intervention by adding hourly wages plus benefits for the nurse, pharmacist, and physician multiplied by the time required of each team member. The intervention cost $31.82 per patient. Given (1) the total of 40.5 minutes per patient‐admission spent by the nurse for each of 563 patients admitted a total of 698 times over 15 months, (2) the assumption of 2000 hours of work in a 12‐month period, and (3) the assumption that these patients and all their admissions were representative of the 15‐month period, the estimated full‐time equivalents was 0.19.
Since Rank 1 discrepancies do not cause harm, we considered only Rank 2‐3 discrepancies. One hundred sixty‐two of the 563 (29%) patients had a discrepancy categorized as Rank 2‐3. Since the cost of the intervention per patient was $31.82, it cost $113.64 to find 1 discrepancy that could cause harm. If each ADE cost a hospital approximately $9344 in 2008, then preventing 1 discrepancy in every 290 patient encounters would offset the intervention costs. For every 290 patients, our data suggest that we would prevent 81 discrepancies. Every potential ADE does not result in an actual harm. Only 1.2% of the potential ADEs would have to result in harm for the cost of the intervention to be offset. Bates el al found that 0.9% of all inpatient medication errors lead to harm.12 Applying this rate to the total of 531 discrepancies found in the current study, 4.8 of them would have caused harm. Applying the inflation‐adjusted cost to these 4.8 harmful discrepancies, the total estimated cost averted would be $44,607; this compares favorably with the $17,915 cost of the nurse‐pharmacist intervention.
DISCUSSION
Inpatient medication reconciliation, an essential patient safety process, prevents potential ADEs and is mandated by The Joint Commission. Previous studies have shown that discrepancies are common occurrences for patients treated in tertiary centers,68 and those discrepancies can lead to patient discomfort or clinical deterioration.6, 8 Our current study supports this body of literature, as 40% of patients had at least 1 discrepancy on admission or discharge, and 29% of those discrepancies had the potential to result in moderate or severe discomfort or clinical deterioration. Although consistent with some findings,8 these numbers are generally lower than other studies,6, 25 where anywhere from 39% to 64% of the discrepancies were classified as Rank 2‐3.
Consistent with other studies, we found that omission was the most common type of discrepancy at admission as well as discharge.6, 8, 9, 21 In recent studies, omissions accounted for 46.5%10, 21 to 60%9 of the discrepancies. Further analysis in our study showed that the more medications a patient took, the higher the likelihood of discrepancya correlation also seen in other studies.9, 10 As the number of medications that a patient takes increases, the more difficult it becomes for all parties involved, including patients, families, and physicians, to keep an accurate recordleading to more opportunities for discrepancies.
Unintended medication discrepancies do not just occur on admission. While we identified many fewer discharge discrepancies, they were more likely to be categorized as Rank 2‐3. This is in contrast with other research that has found more discrepancies at discharge than admission.9, 11 In the current study, active medication reconciliation on admission likely led to a decrease in the number of discharge discrepancies. Even though there were fewer discharge discrepancies, the potential for harm was great and should not be underestimated.
Although many different types of interventions have been tried, this pilot study demonstrated a remarkably easy, generalizable, and inexpensive method. Other interventions have depended on wholesale reengineering of complicated processes,20, 26 pharmacists,10, 15, 16, 18, 19, 21, 22 or particular IT systems.20 Our intervention employed a nursing‐pharmacist model, which may either reduce the cost of healthcare, or at the very least, pay for itself. Each ADE is projected to cost $9300. The nurse‐pharmacist collaboration costs approximately $32 per patient. Thus, preventing only 1 ADE in 290 patient admissions would constitute a breakeven point for the interventiona goal that is likely achievable according to our study results. Even more cost‐effective would be to target only those patients at highest risk for a discrepancynamely those taking multiple home medications.10
There are several limitations to our study. First, we did not have a control group that would allow for comparison of clinical outcomes between the intervention and standard practice. Second, only potential ADEs were avoided. We were not able to determine that an ADE would definitely have occurred if the reconciliation had not taken place. Third, this study was conducted in a single department at 1 institution. As such, the results may not be generalizable to services other than general medicine or to other hospitals. Fourth, we relied on cost data from 1 inpatient study that is more than a decade old to estimate the potential savings to the healthcare system.2 This demonstrates the need for new studies of the cost of ADEs in hospital and outpatient settings. Outpatient medication discrepancies may be more or less costly than their inpatient counterparts, which would impact the cost analysis of this study. Fifth, we did not rely on the brown bag method, asking the patient's family to bring in the medication bottles, for determining the HML. That would certainly have given us another method to confirm the HML. Moreover, the nurse did not confirm the HML with a second source if she felt that the list provided by the patient was accurate. Finally, while we can intervene on discharge discrepancies, we do not control what a patient chooses to do after discharge.27 Health literacy, financial issues, deficits in communication between patients' discharge providers and their primary care providers, and many other factors affect whether patients adhere to their discharge medication list.28
Since this is not a randomized controlled trial, this pilot study requires additional testing to determine if ADEs are actually avoided and costs saved. The HML protocol could be updated to include the brown bag method or other additional steps to verify the HML. Although not inexpensive, a home visit intervention could be tested as well.29, 30
In conclusion, potentially harmful unintended medication discrepancies occurred frequently at both hospital admission and discharge. A nurse‐pharmacist collaboration to monitor and intervene on these discrepancies allowed many to be reconciled before potentially causing harm to patients. The collaboration was relatively efficient and cost‐effective, and the process potentially improves patient safety.
- ,,,,.Adverse drug events in hospitalized patients. Excess length of stay, extra costs, and attributable mortality.JAMA.1997;277(4):301–306.
- ,,, et al.The costs of adverse drug events in hospitalized patients. Adverse Drug Events Prevention Study Group.JAMA.1997;277(4):307–311.
- ,,, et al.Incidence of adverse drug events and potential adverse drug events. Implications for prevention. ADE Prevention Study Group.JAMA.1995;274(1):29–34.
- ,Institute of Medicine (U.S.).Committee on Identifying and Preventing Medication Errors.Preventing Medication Errors.Washington, DC:National Academies Press;2007.
- ,,,,,.Frequency, type and clinical importance of medication history errors at admission to hospital: a systematic review.Can Med Assoc J.2005;173(5):510–515.
- ,,, et al.Unintended medication discrepancies at the time of hospital admission.Arch Intern Med.2005;165(4):424–429.
- ,,.Frequency and type of medication discrepancies in one tertiary care hospital.Healthc Q.2006;9(Spec No):119–123.
- ,,, et al.Medication reconciliation at hospital discharge: evaluating discrepancies.Ann Pharmacother.2008;42(10):1373–1379.
- ,,, et al.Classifying and predicting errors of inpatient medication reconciliation.J Gen Intern Med.2008;23(9):1414–1422.
- ,,, et al.Results of the Medications At Transitions and Clinical Handoffs (MATCH) study: an analysis of medication reconciliation errors and risk factors at hospital admission.J Gen Intern Med.25(5):441–447.
- .Reconciliation failures lead to medication errors.Jt Comm J Qual Patient Saf.2006;32(4):225–229.
- ,,,,.Relationship between medication errors and adverse drug events.J Gen Intern Med.1995;10(4):199–205.
- The Joint Commission.National Patient Safety Goals. 2006 Critical Access Hospital and Hospital National Patient Safety Goals.Oakbrook Terrace, IL:The Joint Commission;2006.
- The Joint Commission.Approved: modifications to National Patient Safety Goal on reconciling medication information.Jt Comm Perspect.2011;31(1):1,3–7.
- ,.Pharmacists' medication reconciliation‐related clinical interventions in a children's hospital.Jt Comm J Qual Patient Saf.2009;35(5):278–282.
- ,,,,,.Reconciliation of discrepancies in medication histories and admission orders of newly hospitalized patients.Am J Health Syst Pharm.2004;61(16):1689–1695.
- ,,, et al.Medication reconciliation: a practical tool to reduce the risk of medication errors.J Crit Care.2003;18(4):201–205.
- ,,,,,.Multidisciplinary approach to inpatient medication reconciliation in an academic setting.Am J Health Syst Pharm.2007;64(8):850–854.
- ,,.Reconcilable differences: correcting medication errors at hospital admission and discharge.Qual Saf Health Care.2006;15(2):122–126.
- ,,, et al.Effect of an electronic medication reconciliation application and process redesign on potential adverse drug events: a cluster‐randomized trial.Arch Intern Med.2009;169(8):771–780.
- ,,, et al.Pharmacist medication assessments in a surgical preadmission clinic.Arch Intern Med.2007;167(10):1034–1040.
- ,.Effectiveness of a pharmacist‐acquired medication history in promoting patient safety.Am J Health Syst Pharm.2002;59(22):2221–2225.
- ,,,,,.Inpatient medication reconciliation at admission and discharge: a retrospective cohort study of age and other risk factors for medication discrepancies.Am J Geriatr Pharmacother.2010;8(2):115–126.
- ,,, et al.Design and implementation of an application and associated services to support interdisciplinary medication reconciliation efforts at an integrated healthcare delivery network.J Am Med Inform Assoc.2006;13(6):581–592.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6(4):313–321.
- ,,, et al.Medication safety program reduces adverse drug events in a community hospital.Qual Saf Health Care.2005;14(3):169–174.
- ,,,.Posthospital medication discrepancies: prevalence and contributing factors.Arch Intern Med.2005;165(16):1842–1847.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297(8):831–841.
- ,,, et al.Adverse events among medical patients after discharge from hospital.Can Med Assoc J.2004;170(3):345–349.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138(3):161–167.
Adverse drug events (ADE), of which medication errors are one form, refer to harm caused by use of a drug. ADEs occur frequently and are associated with an increased length of stay, economic burden, and risk of death.1, 2 Classen et al and Bates et al estimate, respectively, that there are 1.2 to 1.8 preventable ADEs per 100 inpatient admissions.1, 3 Adjusting these data to current levels of yearly admissions, 380,000 to 400,000 preventable ADEs occur each year, and are projected to cost upwards of $3.5 billion annually in 2006 dollars.4
Medication reconciliation is an active process that occurs at transitions in care (admissions, transfers in level of care, and discharge) and is designed to prevent medication errors as the patient moves across the continuum of care. Medications used by the patient prior to hospitalization are considered when developing the inpatient therapeutic regimen.
Medications are ordered on admission based in part on what providers believe is the patient's home medication list (HML). A systematic review revealed that errors in medication history taking, including errors of omission and commission, are extremely common and clinically important.5 Such inaccuracies lead to unintended discrepancies between the hospital medication orders and the patient's true home medication regimen, and can result in patient harm.
Numerous studies have documented that inpatient discrepancies are common.610 From September 2004 to July 2005, data from the United States Pharmacopeia MEDMARX voluntary medication error reporting program revealed over 2000 medication errors associated with reconciliation failures: 22% occurred during admission and 12% occurred at time of discharge.11 A Canadian study demonstrated that 81 of 151 enrolled patients, who were prescribed 4 or more medications and were admitted to a medicine service, had at least 1 unintended discrepancy.6 Of those discrepancies, 38.6% were thought to have the potential to cause moderate or severe discomfort or clinical deterioration. Bates et al found that 0.9% of all inpatient medication errors lead to harm.12
The Joint Commission highlighted the importance of this problem by creating National Patient Safety Goal (NPSG) 8 in 2005, Accurately and completely reconcile medications across the continuum of care.13 This goal was modified and became effective on July 1, 2011.14 As a response, organizations have been developing physician‐led, nurse‐led, or pharmacist‐led medication reconciliation processes.8, 1522 Typically, these teams have time dedicated to producing the most accurate home list possible, a gold standard list. Examples of successful pharmacist‐led interventions to address this goal are described by investigators at Northwestern Memorial Hospital16 and Duke University Medical Center.23 Other interventions implemented to improve the reconciliation process include computerized provider order entry (CPOE) systems24 and combining information technology (IT) with process redesign involving physicians, pharmacists, and nurses.20 While the literature shows that there are multiple interventions that can reduce medication reconciliation errors, there is a dearth of evidence for interventions that are low‐cost and easily replicable.
Given that unintended medication discrepancies are common and harmful, we sought to develop a generalizable intervention. Our prospective pilot study explored whether an easily replicable nurse‐pharmacist led medication reconciliation process could efficiently and inexpensively identify unintended medication discrepancies, thereby preventing potential adverse drug events (PADEs).
METHODS
Patient Selection
The study was conducted at a 1000 bed urban, tertiary care hospital that serves a diverse patient population. We enrolled eligible patients over a 15‐month period, from January 2008 to March 2009, admitted to 2 resident‐covered general medicine teams. Each team is composed of an attending physician, 2 senior residents, 4 interns, a case manager, a pharmacist, and a social worker. Patients were excluded only if they did not consent or were discharged from the hospital in less than 24 hours. Patients were interviewed Monday through Friday, and those admitted over the weekend were interviewed the following Monday. The study was approved by The Johns Hopkins institutional review board (IRB).
Intervention Team
Baccalaureate‐prepared registered nurses (RNs) provided the primary intervention in this model. Both nurses had practiced as bedside clinicians at the hospital and had knowledge of hospital systems and structures. No additional training was provided.
The study pharmacist, who is board certified in pharmacotherapy, has a doctor of pharmacy degree and completed 1 year of Pharmacy Practice residency, as well as a 1‐year specialty residency in Internal Medicine. She spends the majority of her clinical time rounding with the inpatient medicine teams where she provides medication management recommendations.
Home Medication List Compilation
Informed consent was obtained by the study nurse 24‐48 hours after admission. The nurse completed an initial patient interview to determine the HML or preadmission medication list. The patient‐reported HML was compared to the history obtained by the physician. If both lists matched, the HML was considered complete. If a patient was not able to provide a written HML or recall medications, the nurses reviewed the electronic patient record (EPR), which documents previous discharge medication lists and Hopkins outpatient medication lists. If not convinced that the HML was accurate or complete, the nurses could use other sources of information, including patients' families, primary care physicians, and community pharmacies. Patients were then asked to verify the HML. At the start of the study, the nurses created a handwritten HML that they placed in the chart. As functionality of the CPOE improved during the study, the nurses entered the lists into the CPOE instead.
Reconciling the HML with the Admission Orders and Discharge Medication List
The nurses created the HML during the first 24‐48 hours of a patient's admission, so admission orders were entered before the resident physicians were aware of the nurse‐complied HML. By comparing the active medication orders to the HML, the nurses created a list of admission discrepancies. The nurse evaluated the discrepancies in the context of the treatment plan to determine if they appeared to be intended or unintended. The nurses consulted the study pharmacist if they were unsure if a discrepancy was intended. Questions about specific drug substitutions were clarified with the study pharmacist. For example, the nurses consulted the pharmacist about a patient who was taking carvedilol at home but was changed to metoprolol during the hospitalization.
After consultation with the pharmacist, the nurse reviewed all remaining potential unintended admission discrepancies with the physician team. A similar process was repeated on the day of discharge. For all patients transitioning out of the hospital, the nurses compared the active medication list and the HML to the discharge worksheet medication list and patient instructions. The nurses contacted the physician team when potential unintended discrepancies were identified. If unintended discrepancies were confirmed for a patient who had already been discharged earlier that day, a resolution plan was determined and the patient was contacted.
Intended Versus Unintended Discrepancies
After completing the nurse‐pharmacist review, the nurse presented the admission discrepancies that were thought to be unintended to the prescriber. If the medication order was not changed, the discrepancy was considered intended. If the prescriber changed the order, the discrepancy was considered unintended. Unintended discrepancies were identified within 48 hours of admission and also upon discharge. If an unintended discrepancy was identified during admission, it only counted as an admission discrepancy. However, if the same mistake occurred again during discharge, the unintended discrepancy was also counted as a discharge discrepancy. The nurses classified the discrepancy by type: medication omission, frequency error, route error, wrong dose, and/or wrong drug.
Rating Potential Harm From the Unintended Discrepancies
Adjudicators assessed the potential harm of unintended admission discrepancies that could occur during an average 4‐day hospitalization. Similarly, raters assessed the potential harm of unintended discharge medication discrepancies. Each of the 4 adjudicators, 2 physicians and 2 pharmacists, were blinded and independently adjudicated all unintended admission and discharge discrepancies, rating the potential harm from the discrepancy on a scale6 from 1 to 3:
Rank 1: unlikely to cause any harm or discomfort.
Rank 2: potential to cause moderate discomfort or clinical deterioration.
Rank 3: potential for severe discomfort or clinical deterioration.
To rate the potential harm of the discrepancies, the raters were provided with the patients' diagnoses, the medications in question, the types of discrepancies, and whether the discrepancy occurred at admission or discharge. The final potential harm level was determined by the majority rating unless a rating spread of Rank 1 to Rank 3 existed. In that case, consensus was reached by discussion. If the 4 raters were evenly split, the mean value was used.
Cost Analysis: Resources, Valuation, and Cost Savings
The time involved in implementing the protocol was recorded in minutes on an Excel spreadsheet. The time records included: participant interview, contact with secondary sources for medication history (a primary care physician or pharmacy), consultation with the study pharmacist, patient education, discharge activities, and consultation with the prescribers. The study pharmacist submitted additional time for follow‐up of issues after the initial consultation with the nurse.
The cost of an ADE in our study was estimated based on the Bates et al study, which reported an average cost of $5857.00 per inpatient ADE in 1996 dollars.2 Using an inflation adjustment ratio from 1996 to 2008 of 1.595, we calculated the cost of an ADE in 2008 dollars to be an average of $9344.12.
We compared the cost of the program with potential cost savings. We performed a threshold analysis to determine the minimum proportion of Rank 2‐3 potential ADEs that would actually need to result in harm in order for the intervention to yield a cost savings. We also estimated the costs of harm based on the Bates et al study which found that 0.9% of all inpatient medication errors results in harm.12
Statistical Analysis
We used logistic regression to test for associations between discrepancies and patient characteristics including age, race, length of stay, education, marital status, primary payor, severity of illness, and number of medications. The outcome measure was at least 1 unintended discrepancy on admission or discharge. A paired‐samples t test was calculated to compare the mean number of discrepancies on admission to the mean discrepancies at discharge.
RESULTS
We enrolled 563 patients who were admitted a total of 698 times. Only the first admission for each patient was analyzed. Patient demographics are presented in Table 1. Almost 70% of our enrolled patients were less than 65 years old, 65% of the patients were black, 58% lived within 5 miles of the Johns Hopkins Hospital, and a plurality of the patients were single and received Medicare. The mean number of medications was 7.8 (SD 4.9).
| Demographic Variable | Percentage |
|---|---|
| Demographic Variable | Mean SD |
| |
| Sex | |
| Male | 49.2 |
| Female | 50.8 |
| Age | |
| <65 | 69.5 |
| 65 to <85 | 27.5 |
| 85 | 3.0 |
| Marital status | |
| Single | 47.0 |
| Married | 30.0 |
| Divorced | 10.5 |
| Widowed | 10.7 |
| Payor | |
| Medicare | 42.3 |
| Medicaid | 25.0 |
| HMO | 8.0 |
| Self‐pay | 9.2 |
| Race | |
| White | 33.0 |
| Black | 65.1 |
| Other | 1.9 |
| APDRG complexity 1‐4 | |
| 1 | 5.2 |
| 2 | 28.5 |
| 3 | 47.4 |
| 4 | 18.9 |
| Education | |
| Less than eighth grade | 9.2 |
| Some high school | 21.7 |
| High school or GED | 28.9 |
| Some college | 15.1 |
| College degree or greater | 19.5 |
| Chose not to answer | 5.5 |
| ICD‐9 codes | |
| Diseases of the circulatory system | 25.5 |
| Diseases of the respiratory system | 11.1 |
| Diseases of the digestive system | 10.9 |
| Symptoms, signs, and ill‐defined conditions | 10.7 |
| Admission from the Emergency Room | 87 |
| Patient lives within 5 miles of the hospital | 58 |
| Age (years) | 55.4 16.6 |
| Number of medications per patient | 7.76 4.9 |
| Length of stay | 5.72 7.28 |
The most frequent source of the home medication list was from patient verbal recall (52%). Few patients had lists of their current medications when admitted. The second most commonly used source was the electronic patient record, 36.6%, which was used to verify and complete the home list. The patient's community pharmacist, 12.5%, was contacted when other sources did not result in a complete home list. The primary care site was contacted in 6.0% of the cases. Patients were then asked to verify the HML.
Of the 563 patients, 225 (40%; 95% confidence interval [CI], 36%‐44%) had at least 1 unintended discrepancy on admission or discharge. On admission and discharge, 28% (95% CI, 25%‐30%) and 25% (95% CI, 21%‐29%) of the patients, respectively, had an unintended discrepancy. Of those 225 patients who had an unintended discrepancy, 162 (72%) had a discrepancy ranked 2 or 3 on the potential harm scale.
Overall, there were more unintended discrepancies on admission (364) than at discharge (167) (Figure 1). The paired t test showed a significant decrease (t[562] = 2.066, P = 0.039) between the number of discrepancies on admission to discharge. However, the majority of these discrepancies on admission (55%) were Rank 1 on the potential harm scale, while the majority of the discharge discrepancies (85%) were likely to cause harm (Rank 2‐3). There were many more Rank 3 discrepancies upon discharge, 39, than on admission, 13. The percentage of Rank 2‐3 discrepancies on admission and discharge were 45% versus 85%, respectively. Interclass correlation of ratings before consensus was 0.58.

The most common unintended discrepancies were omissions of medications at admission, 74%, and discharge, 62%, followed by discrepancies in dosing (Table 2). The majority of omission discrepancies were categorized as Rank 1. Discrepancies in frequency and dosing were most likely to be adjudicated as Rank 2 or 3. Table 3 gives examples of how discrepancies were ranked.
| a. Type of Discrepancy on Admission | Total | Rank 1 | Rank 2 | Rank 3 |
|---|---|---|---|---|
| 364 (%) | 199 (%) | 152 (%) | 13 (%) | |
| ||||
| Omission | 270 (74) | 157 (79) | 102 (67) | 11 (85) |
| Frequency | 19 (5) | 7 (4) | 12 (8) | 0 |
| Route | 3 (1) | 1 (1) | 2 (1) | 0 |
| Dose | 54 (15) | 23 (12) | 29 (19) | 2 (15) |
| Drug | 18 (5) | 11 (5) | 7 (5) | 0 |
| b. Type of Discrepancy on Discharge | Total | Rank 1 | Rank 2 | Rank 3 |
| 167 (%) | 43 (%) | 85 (%) | 39 (%) | |
| Omission | 104 (62) | 37 (86) | 46 (54) | 21 (54) |
| Frequency | 15 (9) | 3 (7) | 10 (12) | 2 (5) |
| Route | 12 (7) | 2 (5) | 6 (7) | 4 (10) |
| Dose | 22 (13) | 0 | 14 (16) | 8 (21) |
| Drug | 14 (8) | 1 (2) | 9 (11) | 4 (10) |
| Rank | Time of Discrepancy | Clinical Information |
|---|---|---|
| ||
| 1 | Discharge | Elderly patient with sepsis from acute cystitis. Centrum Silver, part of the HML, was not on the discharge orders. |
| 2 | Discharge | Patient admitted with UTI. Metoprolol XL 100 mg was on the patient's HML but not on the discharge orders. |
| 3 | Admission | Patient admitted with hypertensive urgency. Clonidine 0.2 mg by mouth 3 times daily, which was on the patient's HML, was omitted. |
The only statistically significantly variable associated with the presence of discrepancies was the number of medications (odds ratio, 1.087; 95% CI, 1.044‐1.132). Each additional medication increased the odds of a discrepancy by 8.7%. Other variables, including age, race, length of stay, level of education, marital status, primary payor, and severity of illness, were not associated with prevalent discrepancies.
Cost Analysis: Resources, Utilization, and Cost Savings
On average, the nurses spent 11.2 minutes (SD 8.0 minutes) of their time conducting the admitting patient interview. The average total time for the protocol excluding the initial interview was 29.3 minutes (SD 30.2 minutes). The clinical pharmacist was consulted in 30% of the cases. The average consultation time was 7.5 minutes (SD 4.4). We determined the hospital's cost of the intervention by adding hourly wages plus benefits for the nurse, pharmacist, and physician multiplied by the time required of each team member. The intervention cost $31.82 per patient. Given (1) the total of 40.5 minutes per patient‐admission spent by the nurse for each of 563 patients admitted a total of 698 times over 15 months, (2) the assumption of 2000 hours of work in a 12‐month period, and (3) the assumption that these patients and all their admissions were representative of the 15‐month period, the estimated full‐time equivalents was 0.19.
Since Rank 1 discrepancies do not cause harm, we considered only Rank 2‐3 discrepancies. One hundred sixty‐two of the 563 (29%) patients had a discrepancy categorized as Rank 2‐3. Since the cost of the intervention per patient was $31.82, it cost $113.64 to find 1 discrepancy that could cause harm. If each ADE cost a hospital approximately $9344 in 2008, then preventing 1 discrepancy in every 290 patient encounters would offset the intervention costs. For every 290 patients, our data suggest that we would prevent 81 discrepancies. Every potential ADE does not result in an actual harm. Only 1.2% of the potential ADEs would have to result in harm for the cost of the intervention to be offset. Bates el al found that 0.9% of all inpatient medication errors lead to harm.12 Applying this rate to the total of 531 discrepancies found in the current study, 4.8 of them would have caused harm. Applying the inflation‐adjusted cost to these 4.8 harmful discrepancies, the total estimated cost averted would be $44,607; this compares favorably with the $17,915 cost of the nurse‐pharmacist intervention.
DISCUSSION
Inpatient medication reconciliation, an essential patient safety process, prevents potential ADEs and is mandated by The Joint Commission. Previous studies have shown that discrepancies are common occurrences for patients treated in tertiary centers,68 and those discrepancies can lead to patient discomfort or clinical deterioration.6, 8 Our current study supports this body of literature, as 40% of patients had at least 1 discrepancy on admission or discharge, and 29% of those discrepancies had the potential to result in moderate or severe discomfort or clinical deterioration. Although consistent with some findings,8 these numbers are generally lower than other studies,6, 25 where anywhere from 39% to 64% of the discrepancies were classified as Rank 2‐3.
Consistent with other studies, we found that omission was the most common type of discrepancy at admission as well as discharge.6, 8, 9, 21 In recent studies, omissions accounted for 46.5%10, 21 to 60%9 of the discrepancies. Further analysis in our study showed that the more medications a patient took, the higher the likelihood of discrepancya correlation also seen in other studies.9, 10 As the number of medications that a patient takes increases, the more difficult it becomes for all parties involved, including patients, families, and physicians, to keep an accurate recordleading to more opportunities for discrepancies.
Unintended medication discrepancies do not just occur on admission. While we identified many fewer discharge discrepancies, they were more likely to be categorized as Rank 2‐3. This is in contrast with other research that has found more discrepancies at discharge than admission.9, 11 In the current study, active medication reconciliation on admission likely led to a decrease in the number of discharge discrepancies. Even though there were fewer discharge discrepancies, the potential for harm was great and should not be underestimated.
Although many different types of interventions have been tried, this pilot study demonstrated a remarkably easy, generalizable, and inexpensive method. Other interventions have depended on wholesale reengineering of complicated processes,20, 26 pharmacists,10, 15, 16, 18, 19, 21, 22 or particular IT systems.20 Our intervention employed a nursing‐pharmacist model, which may either reduce the cost of healthcare, or at the very least, pay for itself. Each ADE is projected to cost $9300. The nurse‐pharmacist collaboration costs approximately $32 per patient. Thus, preventing only 1 ADE in 290 patient admissions would constitute a breakeven point for the interventiona goal that is likely achievable according to our study results. Even more cost‐effective would be to target only those patients at highest risk for a discrepancynamely those taking multiple home medications.10
There are several limitations to our study. First, we did not have a control group that would allow for comparison of clinical outcomes between the intervention and standard practice. Second, only potential ADEs were avoided. We were not able to determine that an ADE would definitely have occurred if the reconciliation had not taken place. Third, this study was conducted in a single department at 1 institution. As such, the results may not be generalizable to services other than general medicine or to other hospitals. Fourth, we relied on cost data from 1 inpatient study that is more than a decade old to estimate the potential savings to the healthcare system.2 This demonstrates the need for new studies of the cost of ADEs in hospital and outpatient settings. Outpatient medication discrepancies may be more or less costly than their inpatient counterparts, which would impact the cost analysis of this study. Fifth, we did not rely on the brown bag method, asking the patient's family to bring in the medication bottles, for determining the HML. That would certainly have given us another method to confirm the HML. Moreover, the nurse did not confirm the HML with a second source if she felt that the list provided by the patient was accurate. Finally, while we can intervene on discharge discrepancies, we do not control what a patient chooses to do after discharge.27 Health literacy, financial issues, deficits in communication between patients' discharge providers and their primary care providers, and many other factors affect whether patients adhere to their discharge medication list.28
Since this is not a randomized controlled trial, this pilot study requires additional testing to determine if ADEs are actually avoided and costs saved. The HML protocol could be updated to include the brown bag method or other additional steps to verify the HML. Although not inexpensive, a home visit intervention could be tested as well.29, 30
In conclusion, potentially harmful unintended medication discrepancies occurred frequently at both hospital admission and discharge. A nurse‐pharmacist collaboration to monitor and intervene on these discrepancies allowed many to be reconciled before potentially causing harm to patients. The collaboration was relatively efficient and cost‐effective, and the process potentially improves patient safety.
Adverse drug events (ADE), of which medication errors are one form, refer to harm caused by use of a drug. ADEs occur frequently and are associated with an increased length of stay, economic burden, and risk of death.1, 2 Classen et al and Bates et al estimate, respectively, that there are 1.2 to 1.8 preventable ADEs per 100 inpatient admissions.1, 3 Adjusting these data to current levels of yearly admissions, 380,000 to 400,000 preventable ADEs occur each year, and are projected to cost upwards of $3.5 billion annually in 2006 dollars.4
Medication reconciliation is an active process that occurs at transitions in care (admissions, transfers in level of care, and discharge) and is designed to prevent medication errors as the patient moves across the continuum of care. Medications used by the patient prior to hospitalization are considered when developing the inpatient therapeutic regimen.
Medications are ordered on admission based in part on what providers believe is the patient's home medication list (HML). A systematic review revealed that errors in medication history taking, including errors of omission and commission, are extremely common and clinically important.5 Such inaccuracies lead to unintended discrepancies between the hospital medication orders and the patient's true home medication regimen, and can result in patient harm.
Numerous studies have documented that inpatient discrepancies are common.610 From September 2004 to July 2005, data from the United States Pharmacopeia MEDMARX voluntary medication error reporting program revealed over 2000 medication errors associated with reconciliation failures: 22% occurred during admission and 12% occurred at time of discharge.11 A Canadian study demonstrated that 81 of 151 enrolled patients, who were prescribed 4 or more medications and were admitted to a medicine service, had at least 1 unintended discrepancy.6 Of those discrepancies, 38.6% were thought to have the potential to cause moderate or severe discomfort or clinical deterioration. Bates et al found that 0.9% of all inpatient medication errors lead to harm.12
The Joint Commission highlighted the importance of this problem by creating National Patient Safety Goal (NPSG) 8 in 2005, Accurately and completely reconcile medications across the continuum of care.13 This goal was modified and became effective on July 1, 2011.14 As a response, organizations have been developing physician‐led, nurse‐led, or pharmacist‐led medication reconciliation processes.8, 1522 Typically, these teams have time dedicated to producing the most accurate home list possible, a gold standard list. Examples of successful pharmacist‐led interventions to address this goal are described by investigators at Northwestern Memorial Hospital16 and Duke University Medical Center.23 Other interventions implemented to improve the reconciliation process include computerized provider order entry (CPOE) systems24 and combining information technology (IT) with process redesign involving physicians, pharmacists, and nurses.20 While the literature shows that there are multiple interventions that can reduce medication reconciliation errors, there is a dearth of evidence for interventions that are low‐cost and easily replicable.
Given that unintended medication discrepancies are common and harmful, we sought to develop a generalizable intervention. Our prospective pilot study explored whether an easily replicable nurse‐pharmacist led medication reconciliation process could efficiently and inexpensively identify unintended medication discrepancies, thereby preventing potential adverse drug events (PADEs).
METHODS
Patient Selection
The study was conducted at a 1000 bed urban, tertiary care hospital that serves a diverse patient population. We enrolled eligible patients over a 15‐month period, from January 2008 to March 2009, admitted to 2 resident‐covered general medicine teams. Each team is composed of an attending physician, 2 senior residents, 4 interns, a case manager, a pharmacist, and a social worker. Patients were excluded only if they did not consent or were discharged from the hospital in less than 24 hours. Patients were interviewed Monday through Friday, and those admitted over the weekend were interviewed the following Monday. The study was approved by The Johns Hopkins institutional review board (IRB).
Intervention Team
Baccalaureate‐prepared registered nurses (RNs) provided the primary intervention in this model. Both nurses had practiced as bedside clinicians at the hospital and had knowledge of hospital systems and structures. No additional training was provided.
The study pharmacist, who is board certified in pharmacotherapy, has a doctor of pharmacy degree and completed 1 year of Pharmacy Practice residency, as well as a 1‐year specialty residency in Internal Medicine. She spends the majority of her clinical time rounding with the inpatient medicine teams where she provides medication management recommendations.
Home Medication List Compilation
Informed consent was obtained by the study nurse 24‐48 hours after admission. The nurse completed an initial patient interview to determine the HML or preadmission medication list. The patient‐reported HML was compared to the history obtained by the physician. If both lists matched, the HML was considered complete. If a patient was not able to provide a written HML or recall medications, the nurses reviewed the electronic patient record (EPR), which documents previous discharge medication lists and Hopkins outpatient medication lists. If not convinced that the HML was accurate or complete, the nurses could use other sources of information, including patients' families, primary care physicians, and community pharmacies. Patients were then asked to verify the HML. At the start of the study, the nurses created a handwritten HML that they placed in the chart. As functionality of the CPOE improved during the study, the nurses entered the lists into the CPOE instead.
Reconciling the HML with the Admission Orders and Discharge Medication List
The nurses created the HML during the first 24‐48 hours of a patient's admission, so admission orders were entered before the resident physicians were aware of the nurse‐complied HML. By comparing the active medication orders to the HML, the nurses created a list of admission discrepancies. The nurse evaluated the discrepancies in the context of the treatment plan to determine if they appeared to be intended or unintended. The nurses consulted the study pharmacist if they were unsure if a discrepancy was intended. Questions about specific drug substitutions were clarified with the study pharmacist. For example, the nurses consulted the pharmacist about a patient who was taking carvedilol at home but was changed to metoprolol during the hospitalization.
After consultation with the pharmacist, the nurse reviewed all remaining potential unintended admission discrepancies with the physician team. A similar process was repeated on the day of discharge. For all patients transitioning out of the hospital, the nurses compared the active medication list and the HML to the discharge worksheet medication list and patient instructions. The nurses contacted the physician team when potential unintended discrepancies were identified. If unintended discrepancies were confirmed for a patient who had already been discharged earlier that day, a resolution plan was determined and the patient was contacted.
Intended Versus Unintended Discrepancies
After completing the nurse‐pharmacist review, the nurse presented the admission discrepancies that were thought to be unintended to the prescriber. If the medication order was not changed, the discrepancy was considered intended. If the prescriber changed the order, the discrepancy was considered unintended. Unintended discrepancies were identified within 48 hours of admission and also upon discharge. If an unintended discrepancy was identified during admission, it only counted as an admission discrepancy. However, if the same mistake occurred again during discharge, the unintended discrepancy was also counted as a discharge discrepancy. The nurses classified the discrepancy by type: medication omission, frequency error, route error, wrong dose, and/or wrong drug.
Rating Potential Harm From the Unintended Discrepancies
Adjudicators assessed the potential harm of unintended admission discrepancies that could occur during an average 4‐day hospitalization. Similarly, raters assessed the potential harm of unintended discharge medication discrepancies. Each of the 4 adjudicators, 2 physicians and 2 pharmacists, were blinded and independently adjudicated all unintended admission and discharge discrepancies, rating the potential harm from the discrepancy on a scale6 from 1 to 3:
Rank 1: unlikely to cause any harm or discomfort.
Rank 2: potential to cause moderate discomfort or clinical deterioration.
Rank 3: potential for severe discomfort or clinical deterioration.
To rate the potential harm of the discrepancies, the raters were provided with the patients' diagnoses, the medications in question, the types of discrepancies, and whether the discrepancy occurred at admission or discharge. The final potential harm level was determined by the majority rating unless a rating spread of Rank 1 to Rank 3 existed. In that case, consensus was reached by discussion. If the 4 raters were evenly split, the mean value was used.
Cost Analysis: Resources, Valuation, and Cost Savings
The time involved in implementing the protocol was recorded in minutes on an Excel spreadsheet. The time records included: participant interview, contact with secondary sources for medication history (a primary care physician or pharmacy), consultation with the study pharmacist, patient education, discharge activities, and consultation with the prescribers. The study pharmacist submitted additional time for follow‐up of issues after the initial consultation with the nurse.
The cost of an ADE in our study was estimated based on the Bates et al study, which reported an average cost of $5857.00 per inpatient ADE in 1996 dollars.2 Using an inflation adjustment ratio from 1996 to 2008 of 1.595, we calculated the cost of an ADE in 2008 dollars to be an average of $9344.12.
We compared the cost of the program with potential cost savings. We performed a threshold analysis to determine the minimum proportion of Rank 2‐3 potential ADEs that would actually need to result in harm in order for the intervention to yield a cost savings. We also estimated the costs of harm based on the Bates et al study which found that 0.9% of all inpatient medication errors results in harm.12
Statistical Analysis
We used logistic regression to test for associations between discrepancies and patient characteristics including age, race, length of stay, education, marital status, primary payor, severity of illness, and number of medications. The outcome measure was at least 1 unintended discrepancy on admission or discharge. A paired‐samples t test was calculated to compare the mean number of discrepancies on admission to the mean discrepancies at discharge.
RESULTS
We enrolled 563 patients who were admitted a total of 698 times. Only the first admission for each patient was analyzed. Patient demographics are presented in Table 1. Almost 70% of our enrolled patients were less than 65 years old, 65% of the patients were black, 58% lived within 5 miles of the Johns Hopkins Hospital, and a plurality of the patients were single and received Medicare. The mean number of medications was 7.8 (SD 4.9).
| Demographic Variable | Percentage |
|---|---|
| Demographic Variable | Mean SD |
| |
| Sex | |
| Male | 49.2 |
| Female | 50.8 |
| Age | |
| <65 | 69.5 |
| 65 to <85 | 27.5 |
| 85 | 3.0 |
| Marital status | |
| Single | 47.0 |
| Married | 30.0 |
| Divorced | 10.5 |
| Widowed | 10.7 |
| Payor | |
| Medicare | 42.3 |
| Medicaid | 25.0 |
| HMO | 8.0 |
| Self‐pay | 9.2 |
| Race | |
| White | 33.0 |
| Black | 65.1 |
| Other | 1.9 |
| APDRG complexity 1‐4 | |
| 1 | 5.2 |
| 2 | 28.5 |
| 3 | 47.4 |
| 4 | 18.9 |
| Education | |
| Less than eighth grade | 9.2 |
| Some high school | 21.7 |
| High school or GED | 28.9 |
| Some college | 15.1 |
| College degree or greater | 19.5 |
| Chose not to answer | 5.5 |
| ICD‐9 codes | |
| Diseases of the circulatory system | 25.5 |
| Diseases of the respiratory system | 11.1 |
| Diseases of the digestive system | 10.9 |
| Symptoms, signs, and ill‐defined conditions | 10.7 |
| Admission from the Emergency Room | 87 |
| Patient lives within 5 miles of the hospital | 58 |
| Age (years) | 55.4 16.6 |
| Number of medications per patient | 7.76 4.9 |
| Length of stay | 5.72 7.28 |
The most frequent source of the home medication list was from patient verbal recall (52%). Few patients had lists of their current medications when admitted. The second most commonly used source was the electronic patient record, 36.6%, which was used to verify and complete the home list. The patient's community pharmacist, 12.5%, was contacted when other sources did not result in a complete home list. The primary care site was contacted in 6.0% of the cases. Patients were then asked to verify the HML.
Of the 563 patients, 225 (40%; 95% confidence interval [CI], 36%‐44%) had at least 1 unintended discrepancy on admission or discharge. On admission and discharge, 28% (95% CI, 25%‐30%) and 25% (95% CI, 21%‐29%) of the patients, respectively, had an unintended discrepancy. Of those 225 patients who had an unintended discrepancy, 162 (72%) had a discrepancy ranked 2 or 3 on the potential harm scale.
Overall, there were more unintended discrepancies on admission (364) than at discharge (167) (Figure 1). The paired t test showed a significant decrease (t[562] = 2.066, P = 0.039) between the number of discrepancies on admission to discharge. However, the majority of these discrepancies on admission (55%) were Rank 1 on the potential harm scale, while the majority of the discharge discrepancies (85%) were likely to cause harm (Rank 2‐3). There were many more Rank 3 discrepancies upon discharge, 39, than on admission, 13. The percentage of Rank 2‐3 discrepancies on admission and discharge were 45% versus 85%, respectively. Interclass correlation of ratings before consensus was 0.58.

The most common unintended discrepancies were omissions of medications at admission, 74%, and discharge, 62%, followed by discrepancies in dosing (Table 2). The majority of omission discrepancies were categorized as Rank 1. Discrepancies in frequency and dosing were most likely to be adjudicated as Rank 2 or 3. Table 3 gives examples of how discrepancies were ranked.
| a. Type of Discrepancy on Admission | Total | Rank 1 | Rank 2 | Rank 3 |
|---|---|---|---|---|
| 364 (%) | 199 (%) | 152 (%) | 13 (%) | |
| ||||
| Omission | 270 (74) | 157 (79) | 102 (67) | 11 (85) |
| Frequency | 19 (5) | 7 (4) | 12 (8) | 0 |
| Route | 3 (1) | 1 (1) | 2 (1) | 0 |
| Dose | 54 (15) | 23 (12) | 29 (19) | 2 (15) |
| Drug | 18 (5) | 11 (5) | 7 (5) | 0 |
| b. Type of Discrepancy on Discharge | Total | Rank 1 | Rank 2 | Rank 3 |
| 167 (%) | 43 (%) | 85 (%) | 39 (%) | |
| Omission | 104 (62) | 37 (86) | 46 (54) | 21 (54) |
| Frequency | 15 (9) | 3 (7) | 10 (12) | 2 (5) |
| Route | 12 (7) | 2 (5) | 6 (7) | 4 (10) |
| Dose | 22 (13) | 0 | 14 (16) | 8 (21) |
| Drug | 14 (8) | 1 (2) | 9 (11) | 4 (10) |
| Rank | Time of Discrepancy | Clinical Information |
|---|---|---|
| ||
| 1 | Discharge | Elderly patient with sepsis from acute cystitis. Centrum Silver, part of the HML, was not on the discharge orders. |
| 2 | Discharge | Patient admitted with UTI. Metoprolol XL 100 mg was on the patient's HML but not on the discharge orders. |
| 3 | Admission | Patient admitted with hypertensive urgency. Clonidine 0.2 mg by mouth 3 times daily, which was on the patient's HML, was omitted. |
The only statistically significantly variable associated with the presence of discrepancies was the number of medications (odds ratio, 1.087; 95% CI, 1.044‐1.132). Each additional medication increased the odds of a discrepancy by 8.7%. Other variables, including age, race, length of stay, level of education, marital status, primary payor, and severity of illness, were not associated with prevalent discrepancies.
Cost Analysis: Resources, Utilization, and Cost Savings
On average, the nurses spent 11.2 minutes (SD 8.0 minutes) of their time conducting the admitting patient interview. The average total time for the protocol excluding the initial interview was 29.3 minutes (SD 30.2 minutes). The clinical pharmacist was consulted in 30% of the cases. The average consultation time was 7.5 minutes (SD 4.4). We determined the hospital's cost of the intervention by adding hourly wages plus benefits for the nurse, pharmacist, and physician multiplied by the time required of each team member. The intervention cost $31.82 per patient. Given (1) the total of 40.5 minutes per patient‐admission spent by the nurse for each of 563 patients admitted a total of 698 times over 15 months, (2) the assumption of 2000 hours of work in a 12‐month period, and (3) the assumption that these patients and all their admissions were representative of the 15‐month period, the estimated full‐time equivalents was 0.19.
Since Rank 1 discrepancies do not cause harm, we considered only Rank 2‐3 discrepancies. One hundred sixty‐two of the 563 (29%) patients had a discrepancy categorized as Rank 2‐3. Since the cost of the intervention per patient was $31.82, it cost $113.64 to find 1 discrepancy that could cause harm. If each ADE cost a hospital approximately $9344 in 2008, then preventing 1 discrepancy in every 290 patient encounters would offset the intervention costs. For every 290 patients, our data suggest that we would prevent 81 discrepancies. Every potential ADE does not result in an actual harm. Only 1.2% of the potential ADEs would have to result in harm for the cost of the intervention to be offset. Bates el al found that 0.9% of all inpatient medication errors lead to harm.12 Applying this rate to the total of 531 discrepancies found in the current study, 4.8 of them would have caused harm. Applying the inflation‐adjusted cost to these 4.8 harmful discrepancies, the total estimated cost averted would be $44,607; this compares favorably with the $17,915 cost of the nurse‐pharmacist intervention.
DISCUSSION
Inpatient medication reconciliation, an essential patient safety process, prevents potential ADEs and is mandated by The Joint Commission. Previous studies have shown that discrepancies are common occurrences for patients treated in tertiary centers,68 and those discrepancies can lead to patient discomfort or clinical deterioration.6, 8 Our current study supports this body of literature, as 40% of patients had at least 1 discrepancy on admission or discharge, and 29% of those discrepancies had the potential to result in moderate or severe discomfort or clinical deterioration. Although consistent with some findings,8 these numbers are generally lower than other studies,6, 25 where anywhere from 39% to 64% of the discrepancies were classified as Rank 2‐3.
Consistent with other studies, we found that omission was the most common type of discrepancy at admission as well as discharge.6, 8, 9, 21 In recent studies, omissions accounted for 46.5%10, 21 to 60%9 of the discrepancies. Further analysis in our study showed that the more medications a patient took, the higher the likelihood of discrepancya correlation also seen in other studies.9, 10 As the number of medications that a patient takes increases, the more difficult it becomes for all parties involved, including patients, families, and physicians, to keep an accurate recordleading to more opportunities for discrepancies.
Unintended medication discrepancies do not just occur on admission. While we identified many fewer discharge discrepancies, they were more likely to be categorized as Rank 2‐3. This is in contrast with other research that has found more discrepancies at discharge than admission.9, 11 In the current study, active medication reconciliation on admission likely led to a decrease in the number of discharge discrepancies. Even though there were fewer discharge discrepancies, the potential for harm was great and should not be underestimated.
Although many different types of interventions have been tried, this pilot study demonstrated a remarkably easy, generalizable, and inexpensive method. Other interventions have depended on wholesale reengineering of complicated processes,20, 26 pharmacists,10, 15, 16, 18, 19, 21, 22 or particular IT systems.20 Our intervention employed a nursing‐pharmacist model, which may either reduce the cost of healthcare, or at the very least, pay for itself. Each ADE is projected to cost $9300. The nurse‐pharmacist collaboration costs approximately $32 per patient. Thus, preventing only 1 ADE in 290 patient admissions would constitute a breakeven point for the interventiona goal that is likely achievable according to our study results. Even more cost‐effective would be to target only those patients at highest risk for a discrepancynamely those taking multiple home medications.10
There are several limitations to our study. First, we did not have a control group that would allow for comparison of clinical outcomes between the intervention and standard practice. Second, only potential ADEs were avoided. We were not able to determine that an ADE would definitely have occurred if the reconciliation had not taken place. Third, this study was conducted in a single department at 1 institution. As such, the results may not be generalizable to services other than general medicine or to other hospitals. Fourth, we relied on cost data from 1 inpatient study that is more than a decade old to estimate the potential savings to the healthcare system.2 This demonstrates the need for new studies of the cost of ADEs in hospital and outpatient settings. Outpatient medication discrepancies may be more or less costly than their inpatient counterparts, which would impact the cost analysis of this study. Fifth, we did not rely on the brown bag method, asking the patient's family to bring in the medication bottles, for determining the HML. That would certainly have given us another method to confirm the HML. Moreover, the nurse did not confirm the HML with a second source if she felt that the list provided by the patient was accurate. Finally, while we can intervene on discharge discrepancies, we do not control what a patient chooses to do after discharge.27 Health literacy, financial issues, deficits in communication between patients' discharge providers and their primary care providers, and many other factors affect whether patients adhere to their discharge medication list.28
Since this is not a randomized controlled trial, this pilot study requires additional testing to determine if ADEs are actually avoided and costs saved. The HML protocol could be updated to include the brown bag method or other additional steps to verify the HML. Although not inexpensive, a home visit intervention could be tested as well.29, 30
In conclusion, potentially harmful unintended medication discrepancies occurred frequently at both hospital admission and discharge. A nurse‐pharmacist collaboration to monitor and intervene on these discrepancies allowed many to be reconciled before potentially causing harm to patients. The collaboration was relatively efficient and cost‐effective, and the process potentially improves patient safety.
- ,,,,.Adverse drug events in hospitalized patients. Excess length of stay, extra costs, and attributable mortality.JAMA.1997;277(4):301–306.
- ,,, et al.The costs of adverse drug events in hospitalized patients. Adverse Drug Events Prevention Study Group.JAMA.1997;277(4):307–311.
- ,,, et al.Incidence of adverse drug events and potential adverse drug events. Implications for prevention. ADE Prevention Study Group.JAMA.1995;274(1):29–34.
- ,Institute of Medicine (U.S.).Committee on Identifying and Preventing Medication Errors.Preventing Medication Errors.Washington, DC:National Academies Press;2007.
- ,,,,,.Frequency, type and clinical importance of medication history errors at admission to hospital: a systematic review.Can Med Assoc J.2005;173(5):510–515.
- ,,, et al.Unintended medication discrepancies at the time of hospital admission.Arch Intern Med.2005;165(4):424–429.
- ,,.Frequency and type of medication discrepancies in one tertiary care hospital.Healthc Q.2006;9(Spec No):119–123.
- ,,, et al.Medication reconciliation at hospital discharge: evaluating discrepancies.Ann Pharmacother.2008;42(10):1373–1379.
- ,,, et al.Classifying and predicting errors of inpatient medication reconciliation.J Gen Intern Med.2008;23(9):1414–1422.
- ,,, et al.Results of the Medications At Transitions and Clinical Handoffs (MATCH) study: an analysis of medication reconciliation errors and risk factors at hospital admission.J Gen Intern Med.25(5):441–447.
- .Reconciliation failures lead to medication errors.Jt Comm J Qual Patient Saf.2006;32(4):225–229.
- ,,,,.Relationship between medication errors and adverse drug events.J Gen Intern Med.1995;10(4):199–205.
- The Joint Commission.National Patient Safety Goals. 2006 Critical Access Hospital and Hospital National Patient Safety Goals.Oakbrook Terrace, IL:The Joint Commission;2006.
- The Joint Commission.Approved: modifications to National Patient Safety Goal on reconciling medication information.Jt Comm Perspect.2011;31(1):1,3–7.
- ,.Pharmacists' medication reconciliation‐related clinical interventions in a children's hospital.Jt Comm J Qual Patient Saf.2009;35(5):278–282.
- ,,,,,.Reconciliation of discrepancies in medication histories and admission orders of newly hospitalized patients.Am J Health Syst Pharm.2004;61(16):1689–1695.
- ,,, et al.Medication reconciliation: a practical tool to reduce the risk of medication errors.J Crit Care.2003;18(4):201–205.
- ,,,,,.Multidisciplinary approach to inpatient medication reconciliation in an academic setting.Am J Health Syst Pharm.2007;64(8):850–854.
- ,,.Reconcilable differences: correcting medication errors at hospital admission and discharge.Qual Saf Health Care.2006;15(2):122–126.
- ,,, et al.Effect of an electronic medication reconciliation application and process redesign on potential adverse drug events: a cluster‐randomized trial.Arch Intern Med.2009;169(8):771–780.
- ,,, et al.Pharmacist medication assessments in a surgical preadmission clinic.Arch Intern Med.2007;167(10):1034–1040.
- ,.Effectiveness of a pharmacist‐acquired medication history in promoting patient safety.Am J Health Syst Pharm.2002;59(22):2221–2225.
- ,,,,,.Inpatient medication reconciliation at admission and discharge: a retrospective cohort study of age and other risk factors for medication discrepancies.Am J Geriatr Pharmacother.2010;8(2):115–126.
- ,,, et al.Design and implementation of an application and associated services to support interdisciplinary medication reconciliation efforts at an integrated healthcare delivery network.J Am Med Inform Assoc.2006;13(6):581–592.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6(4):313–321.
- ,,, et al.Medication safety program reduces adverse drug events in a community hospital.Qual Saf Health Care.2005;14(3):169–174.
- ,,,.Posthospital medication discrepancies: prevalence and contributing factors.Arch Intern Med.2005;165(16):1842–1847.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297(8):831–841.
- ,,, et al.Adverse events among medical patients after discharge from hospital.Can Med Assoc J.2004;170(3):345–349.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138(3):161–167.
- ,,,,.Adverse drug events in hospitalized patients. Excess length of stay, extra costs, and attributable mortality.JAMA.1997;277(4):301–306.
- ,,, et al.The costs of adverse drug events in hospitalized patients. Adverse Drug Events Prevention Study Group.JAMA.1997;277(4):307–311.
- ,,, et al.Incidence of adverse drug events and potential adverse drug events. Implications for prevention. ADE Prevention Study Group.JAMA.1995;274(1):29–34.
- ,Institute of Medicine (U.S.).Committee on Identifying and Preventing Medication Errors.Preventing Medication Errors.Washington, DC:National Academies Press;2007.
- ,,,,,.Frequency, type and clinical importance of medication history errors at admission to hospital: a systematic review.Can Med Assoc J.2005;173(5):510–515.
- ,,, et al.Unintended medication discrepancies at the time of hospital admission.Arch Intern Med.2005;165(4):424–429.
- ,,.Frequency and type of medication discrepancies in one tertiary care hospital.Healthc Q.2006;9(Spec No):119–123.
- ,,, et al.Medication reconciliation at hospital discharge: evaluating discrepancies.Ann Pharmacother.2008;42(10):1373–1379.
- ,,, et al.Classifying and predicting errors of inpatient medication reconciliation.J Gen Intern Med.2008;23(9):1414–1422.
- ,,, et al.Results of the Medications At Transitions and Clinical Handoffs (MATCH) study: an analysis of medication reconciliation errors and risk factors at hospital admission.J Gen Intern Med.25(5):441–447.
- .Reconciliation failures lead to medication errors.Jt Comm J Qual Patient Saf.2006;32(4):225–229.
- ,,,,.Relationship between medication errors and adverse drug events.J Gen Intern Med.1995;10(4):199–205.
- The Joint Commission.National Patient Safety Goals. 2006 Critical Access Hospital and Hospital National Patient Safety Goals.Oakbrook Terrace, IL:The Joint Commission;2006.
- The Joint Commission.Approved: modifications to National Patient Safety Goal on reconciling medication information.Jt Comm Perspect.2011;31(1):1,3–7.
- ,.Pharmacists' medication reconciliation‐related clinical interventions in a children's hospital.Jt Comm J Qual Patient Saf.2009;35(5):278–282.
- ,,,,,.Reconciliation of discrepancies in medication histories and admission orders of newly hospitalized patients.Am J Health Syst Pharm.2004;61(16):1689–1695.
- ,,, et al.Medication reconciliation: a practical tool to reduce the risk of medication errors.J Crit Care.2003;18(4):201–205.
- ,,,,,.Multidisciplinary approach to inpatient medication reconciliation in an academic setting.Am J Health Syst Pharm.2007;64(8):850–854.
- ,,.Reconcilable differences: correcting medication errors at hospital admission and discharge.Qual Saf Health Care.2006;15(2):122–126.
- ,,, et al.Effect of an electronic medication reconciliation application and process redesign on potential adverse drug events: a cluster‐randomized trial.Arch Intern Med.2009;169(8):771–780.
- ,,, et al.Pharmacist medication assessments in a surgical preadmission clinic.Arch Intern Med.2007;167(10):1034–1040.
- ,.Effectiveness of a pharmacist‐acquired medication history in promoting patient safety.Am J Health Syst Pharm.2002;59(22):2221–2225.
- ,,,,,.Inpatient medication reconciliation at admission and discharge: a retrospective cohort study of age and other risk factors for medication discrepancies.Am J Geriatr Pharmacother.2010;8(2):115–126.
- ,,, et al.Design and implementation of an application and associated services to support interdisciplinary medication reconciliation efforts at an integrated healthcare delivery network.J Am Med Inform Assoc.2006;13(6):581–592.
- ,,, et al.The impact of computerized physician order entry on medication error prevention.J Am Med Inform Assoc.1999;6(4):313–321.
- ,,, et al.Medication safety program reduces adverse drug events in a community hospital.Qual Saf Health Care.2005;14(3):169–174.
- ,,,.Posthospital medication discrepancies: prevalence and contributing factors.Arch Intern Med.2005;165(16):1842–1847.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297(8):831–841.
- ,,, et al.Adverse events among medical patients after discharge from hospital.Can Med Assoc J.2004;170(3):345–349.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138(3):161–167.
Copyright © 2012 Society of Hospital Medicine
CHF Management for Hospitalists
Caring for patients with acute decompensated heart failure (ADHF) is one of the core competencies of practice in hospitalist medicine. Congestive heart failure remains the most common discharge diagnosis as recorded in the National Hospital Discharge Survey, with over 1.1 million hospitalizations for heart failure in 2004.1 Furthermore, with the disproportionate growth in the population over age 65 that will occur over the next 20 years, heart failure prevalence will grow from its current value of 2.8% to 3.5% by 2030.2 This will result in an additional 3 million Americans with chronic heart failure, thereby sustaining ADHF as the most common reason for hospital admission. Despite an average hospital stay of 5 days, the readmission rate for heart failure was 26.9% at 30 days in a 2003‐2004 analysis of Medicare data.3 This high readmission rate is the target of reform as part of the recently passed Patient Protection and Accountability Act. Starting in fiscal year 2013, acute‐care hospitals with higher‐than‐expected readmission rates for heart failure will have a reduction in reimbursement for these admissions.4 Thus, there is substantial incentive for hospitalists to focus on providing the highest quality of care for patients with ADHF. Here we review the most recent evidence applicable to hospitalists for the diagnosis, risk stratification, and management of patients presenting with ADHF.
DIAGNOSIS
The hospitalist can establish the ADHF diagnosis efficiently by applying a structured approach based on the patient's symptoms, history, physical examination, and laboratory testing. The typical symptoms of ADHF include dyspnea, orthopnea, paroxysmal nocturnal dyspnea (PND), and lower extremity edema. In particular, patients complaining of PND and/or orthopnea are likely to have ADHF.5, 6 Patients may also report chest congestion or chest pain in an atypical pattern. A history of rapid weight gain suggests fluid overload, hence determination of the patient's dry weight is important to establish a target for congestive therapy. Patients with advanced systolic heart failure may also complain of nausea, abdominal pain, and abdominal fullness from ascites.7 In a patient with dyspnea, a history of heart failure, myocardial infarction, or coronary artery disease, all make the diagnosis of ADHF more likely.5
Performing a careful physical examination on a patient presenting with suspected ADHF will not only establish the diagnosis of heart failure, but also determine the hemodynamic profile. Patients presenting with ADHF can be separated into 4 hemodynamic profiles, based on vital sign and physical exam parameters: the presence or absence of congestion (wet or dry), and the presence or absence of adequate perfusion (warm or cold) (Figure 1).8 Parameters indicating the presence of congestion include: orthopnea, elevated jugular venous pulsation (JVP), lower extremity edema, hepatojugular reflux, ascites, and a loud P2 heart sound. Notably, rales are an uncommon physical finding in patients with ADHF, likely because pulmonary lymphatics compensate for chronically elevated filling pressures in such patients.9, 10 Parameters indicating inadequate perfusion include: hypotension (mean arterial pressure 60 mmHg), proportional pulse pressure 25%, cool extremities, altered mental status, and poor urine output (0.5 mL/kg/hr). We recommend assigning the patient to 1 of these 4 hemodynamic profiles, as the profile correlates with invasive hemodynamic measurements of pulmonary capillary wedge pressure and cardiac index, guides management, and predicts outcome.

Natriuretic peptide testing may help establish or exclude a diagnosis of ADHF. A recent expert consensus paper on natriuretic peptide testing recommends cutpoints for both B‐type natriuretic peptide (BNP) and N‐terminal proBNP (NT‐BNP) that indicate a very low (BNP 100 or NT‐BNP 300), intermediate (BNP 100‐400 or NT‐BNP 300‐1800), and high (BNP >400 or NT‐BNP >1800) probability of heart failure11 (Figure 2). However, 2 common conditions affect the utility of BNP testing. First, obese patients have lower levels, and thus a lower rule‐out cutpoint of 54 pg/mL is recommended when using BNP, whereas the cutpoint for NT‐BNP remains the same.12, 13 Second, in patients with renal dysfunction, levels are increased, and thus higher rule‐out cutpoints of 200 pg/mL (for BNP) and 1200 pg/mL (for NT‐BNP) are recommended for patients with a glomerular filtration rate 60 mL/min.14, 15 For patients with longstanding heart failure and chronically elevated levels of natriuretic peptides, there is a correlation between BNP levels and left ventricular filling pressure,16 but the change is more helpful than the absolute levels; a 50% increase over baseline, in conjunction with symptoms, usually reflects ADHF.11
Chest radiography will establish the presence or absence of pulmonary congestion. Classic teaching is that congestion starts with cephalization (pulmonary capillary wedge pressure 10‐15 mmHg), progresses to Kerley B lines (15‐20 mmHg), then to interstitial edema (20‐25 mmHg), and finally to alveolar edema (>25 mmHg).17 In patients presenting with dyspnea, any of these findings helps to establish the diagnosis of ADHF.5
MECHANISMS AND TERMINOLOGY
Data from ADHF registries show that hemodynamically stable patients presenting to the hospital with ADHF are an approximately equal mix of heart failure with reduced ejection fraction (HFrEF; ejection fraction 50%) and heart failure with preserved ejection fraction (HFpEF; ejection fraction 50%).18, 19 The important differences between these groups with regards to pathophysiology and etiology have been reviewed elsewhere.20 Establishing the heart failure mechanism (ie, reduced or preserved EF) is important because the medical management is distinct. Patients with HFrEF are more likely to be male, younger in age, to have ischemic heart disease, and to present with normal or low blood pressure. Patients with HFpEF are more likely to be female, older in age, to have hypertension or diabetes mellitus, and to present with elevated blood pressure.18, 19
The terminology used for inpatient heart failure coding has been the subject of renewed focus. For fiscal year 2008, the Centers for Medicare and Medicaid Services (CMS) overhauled its Diagnosis Related Group (DRG) system to better account for the severity of illness of hospitalized patients.21 In this revision, the existing DRG codes for heart failure were subdivided into 3 severity subclasses: major complication, complication, and non‐complication. Payment to hospitals for a heart failure DRG was changed to be proportional to the level of complication. Thus, for the first time, the clinicians' assessment of the acuity of heart failure determines the level of payment to the hospital. Not surprisingly, this has led to initiatives by hospitals to improve clinicians' coding of inpatients hospitalized with heart failure. A major impediment is that there are no established criteria for the application of each DRG code. Table 1 presents recommended clinical criteria for the application of these codes to patients with ADHF.
| ICD‐9 | DRG Code | Severity Subclass | Clinical Criteria | Hemodynamic Profile |
|---|---|---|---|---|
| ||||
| Acute decompensated heart failure | ||||
| 428.21 | Systolic, acute | MCC | New diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.23 | Systolic, acute on chronic | MCC | Established diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.41 | Combined systolic and diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.43 | Combined systolic and diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.31 | Diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| 428.33 | Diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| Chronic heart failure | ||||
| 428.22 | Systolic, chronic | CC | No previous symptoms, or history of clinical features of low‐output state but currently compensated, EF 50 | Dry‐warm |
| 428.40 | Combined systolic and diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| 428.32 | Diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| Other | ||||
| 428.1 | Left heart failure | CC | Clinical features of congestion, mechanism and EF is unknown | Wet‐warm or wet‐cold |
| 428.20 | Systolic heart failure, unspecified | CC | Clinical features of low‐output, acuity is unknown | Dry‐cold |
| 428.0 | Congestive heart failure, unspecified | NCC | Clinical features of right‐heart failure | Not applicable |
PRECIPITANTS AND ETIOLOGY
For patients presenting for the first time with a diagnosis of ADHF (de novo), a thorough evaluation should be performed to determine the mechanism and etiology of the patient's left ventricular dysfunction. After the initial history and physical exam, the American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend checking basic laboratory studies, an electrocardiogram, and an echocardiogram.22 The full assessment recommended by the ACC/AHA is detailed in Supporting Online Table 1 (in the online version of this article). Cardiac ischemia is the most common etiology of HFrEF, accounting for about 50% of cases. The common, non‐ischemic causes of systolic heart failure include atrial fibrillation, aortic stenosis, illicit cardiotoxic drugs (cocaine, methamphetamine), medical cardiotoxic drugs (adriamycin), as well as primary myocardial disorders such as myocarditis, idiopathic, or peripartum cardiomyopathy. HFpEF is most commonly associated with long‐standing hypertension and diabetes mellitus, but can also be caused by infiltrative, hypertrophic, and constrictive cardiomyopathies.
For patients with a history of heart failure, it is important to identify the precipitant for the decompensation, as it may be treated or avoided in the future. When no clear precipitant is identified, this is most concerning, as it indicates the patient's tenuous cardiac function. In the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF) registry, approximately 61% of patients were found to have at least 1 precipitating factor.23 The most common precipitants were respiratory process in 15.3%, acute coronary syndrome in 14.7%, arrhythmia in 13.5%, uncontrolled hypertension in 10.7%, medication non‐compliance in 8.9%, worsening renal function in 8.0%, and dietary non‐compliance in 5.2%.
RISK STRATIFICATION
Patients hospitalized with ADHF are at a significantly elevated risk for death, both during their hospitalization and after discharge. Numerous studies have shown that multiple clinical parameters assessed during the hospitalization, such as vital signs and laboratory values, predict outcome.6, 8, 24, 25 Some of the most elegant parameters are physical exam findings. As introduced above, the wet‐cold hemodynamic profile assessed at admission predicts increased mortality and urgent transplantation at 1 year.8 One of the most powerful risk stratification schemes for in‐hospital mortality is that developed from the Acute Decompensated Heart Failure (ADHERE) national registry. Three clinical parameters, blood urea nitrogen (BUN) >43 mg/dL, systolic blood pressure 115 mmHg, and serum creatinine >2.75 mg/dL, stratified patients into risk groups. Patients exhibiting all 3 parameters had a 22% in‐hospital mortality compared with 2% for patients with none of the 3 parameters.24
BNP and troponin also have a role in risk stratification of patients with ADHF. In the ADHERE registry, for every increase in the BNP of 400 pg/mL, the odds of risk‐adjusted mortality increased by 9%, in patients with both HFrEF and HFpEF.26 Similarly, an elevated admission troponin was associated with an in‐hospital mortality of 8.0%, versus 2.7% for troponin‐negative patients27; notably almost half of patients with a positive troponin had no history of ischemic heart disease. In the future, refinement and widespread application of these risk stratification methods should allow clinicians to triage patients to determine their location (eg, observation unit, inpatient, intensive care unit) and type of treatment (eg, oral or intravenous diuretic, vasodilator, inotrope).28
In the community, hospitalists care for many patients with ADHF without input from a cardiologist.29 However, there are several situations where the patient is at an increased risk of adverse outcomes, and therefore in which we recommend consulting a cardiologist (Table 2). Patients with hypotension, a cold hemodynamic profile, or worsening renal function due to poor cardiac function are at an especially elevated risk and should be considered for advanced therapies such as mechanical circulatory support or heart transplantation.
| Results of Evaluation | Indication for Referral | Purpose of Referral |
|---|---|---|
| ||
| Hypotension, cold hemodynamic profile | Inadequate perfusion | Pulmonary artery catheterization, inotropic therapy |
| Ischemic symptoms, positive troponin, abnormal ECG, echocardiogram with focal wall motion abnormalities | Cardiac ischemia | Coronary angiography and coronary intervention if indicated |
| Atrial fibrillation | Arrhythmia | Consideration of a rhythm control strategy |
| Ejection fraction 35% | Severe left ventricular systolic dysfunction | Implantable cardiac defibrillator and/or biventricular pacemaker |
| High diuretic dose requirements or decreasing urinary response to diuretics | Diuretic resistance | Consideration for vasodilator therapy or ultrafiltration |
| Increasing blood urea nitrogen and serum creatinine, decreasing urine output | Worsening renal function | Consideration of inotropic therapy |
CONGESTION AND DIURESIS
The syndrome of heart failure is due primarily to elevation of left ventricular filling pressures resulting in congestion, and therapies aimed at reducing congestion are of primary importance.30 For 50 years, treatment with diuretic medications has been the mainstay of therapy for patients admitted with ADHF. Vasodilator agents, specifically nitroglycerin and sodium nitroprusside, may also be beneficial in patients presenting with ADHF and hypertension.22 In 1 study, patients with acute pulmonary edema treated with high‐dose nitroglycerin experienced fewer adverse events as compared to those treated with high‐dose diuretics alone, suggesting that nitrates can more rapidly decrease congestion and thereby improve outcomes.31 Unfortunately vasodilators are underutilized, with only 5.8% of patients with elevated blood pressure (>160 mmHg) treated with nitrates in the OPTIMIZE‐HF registry.9
Diuretic choice, dosing, and administration method have traditionally been highly variable between practitioners. Oral diuretics are generally not preferred initially for patients with ADHF because of concerns of inadequate absorption from an edematous bowel and slow onset of action.32 For a patient who is not on diuretics as an outpatient, an initial dose of 40 mg intravenous furosemide is reasonable. For a patient with chronic heart failure on outpatient loop diuretic therapy, the Diuretic Optimization Strategies Evaluation (DOSE) study provides insight into diuretic dosing and administration. Patients were randomized to an administration route (bolus dosing every 12 hours or continuous infusion) and a dosing strategy (low‐dose or high‐dose).33 There were no differences in the primary endpoint of patient‐reported global assessment of symptoms, or the primary safety endpoint of change in serum creatinine from baseline to 72 hours between the bolus and continuous infusion groups or between the low‐dose and high‐dose groups. However, patients in the high‐dose group had decreased dyspnea at 72 hours, decreased body weight at 72 hours, increased fluid loss at 72 hours, and decreased NT‐BNP at 72 hours. These improvements came at the expense of a mild increase in creatinine. Therefore, in hospitalized patients with ADHF on outpatient furosemide, these data support initiation of high‐dose furosemide with a daily intravenous dose equal to 2.5 times their daily outpatient oral dose, using either bolus or continuous infusion.
All patients being treated with diuretic therapy should have close fluid intake and output monitoring, fluid restriction of 1500 to 2000 mL per day, a 2 gram sodium diet, and at least daily electrolyte monitoring. For patients with inadequate diuresis (generally less than 1 L per day in a patient with moderate volume overload), several options are available. If the urinary response to a furosemide dose is inadequate, the dose should be doubled and the urinary response followed. If there has been inadequate diuresis in a patient with a low serum albumin or significant proteinuria, furosemide should be switched to bumetanide, which is not protein‐bound and thus will achieve higher concentrations in the tubule.34
Longstanding treatment with loop diuretics leads to decreased renal responsiveness and an increased dose required to maintain euvolemia. Patients taking furosemide 80 mg daily or above (or an equivalent dose of other loop diuretics) are designated as diuretic‐resistant.35, 36 Diuretic resistance is associated with more severe heart failure, more advanced chronic kidney disease, and worsening renal function with the use of intravenous diuretics.35, 37, 38 There are no consensus recommendations available to guide the management of diuretic resistance, but several options exist. First, a thiazide diuretic, such as metolazone, can be given before the loop diuretic.39 This combination is frequently able to initiate a brisk diuresis, but patients require close monitoring for hypokalemia and worsening renal function. Recently, ultrafiltration has emerged as an option. In the Ultrafiltration versus Intravenous Diuretics for Patients Hospitalized for Acute Decompensated Congestive Heart Failure (UNLOAD) trial, patients with congestion treated with ultrafiltration had more weight and fluid loss at 48 hours compared to patients treated with intravenous furosemide, without any significant differences in renal function.40 For patients with oliguria and renal dysfunction, initiation of renal replacement therapy may be needed. We present an algorithm for the management of diuretic resistance in Figure 3.

The endpoints for discontinuation of diuretic therapy remain unclear. Traditionally, alleviation of the patient's congestive symptoms, edema, and attainment of the patient's self‐reported dry weight have served as endpoints for diuretic therapy. A more accurate approach may be daily assessments of the JVP, as normalization of the JVP may be a more accurate method to assess for euvolemia. When euvolemia has been achieved, patients should be switched to maintenance therapy at a diuretic dose of one‐fourth to one‐half the total daily dose used for diuresis. Patients should be observed for 24 hours on oral diuretic therapy to ensure that their fluid intake and output are balanced. Generally, we aim for slightly negative fluid balance (less than 500 mL) on an oral diuretic regimen prior to discharge, assuming some relaxation of the salt and fluid restriction once the patient is discharged home.
NEUROHORMONAL THERAPIES
Activation of neurohormonal systems, specifically the renin‐angiotensin‐aldosterone and beta‐adrenergic pathways, are the major mechanisms for disease progression in HFrEF, and agents which block these pathways improve functional status and survival in these patients. In the OPTIMIZE‐HF registry, patients treated with beta‐blockers on admission had a lower in‐hospital mortality.25 Although beta‐blockers are often discontinued in patients with ADHF, continuation of beta‐blocker treatment is associated with decreased mortality and rehospitalization at 60 to 90 days.41 While beta‐blocker initiation is often deferred to the outpatient setting, patients who receive a beta‐blocker at hospital discharge are 31 times more likely to be treated with a beta‐blocker at 60 to 90 day follow‐up.42 Only 3 agents, metoprolol succinate, carvedilol, and bisoprolol, have survival benefit in large clinical trials of systolic heart failure, and therefore are the only recommended agents.22 In the hospital, hypotension is a common reason for suspension or discontinuation of beta‐blocker therapy. However, in the absence of symptoms such as light‐headedness, patients with systolic blood pressure as low as 85 mmHg will benefit from beta‐blocker treatment.43 Thus, we recommend continuation or initiation of an evidence‐based beta‐blocker for all patients hospitalized with systolic heart failure in the absence of symptomatic hypotension, systolic blood pressure 85 mmHg, second or third degree heart block, or the need for intravenous inotropic therapy.
Inhibitors of the renin‐angiotensin‐aldosterone system also have an important role in patients with HFrEF. Patients treated with angiotensin converting enzyme inhibitors (ACEI) on admission have a lower in‐hospital mortality25 and a lower likelihood of readmission or death within 60 to 90 days.44 In practice, ACEI or angiotensin receptor blocker (ARB) treatment is frequently suspended or discontinued during treatment with diuretics out of concerns for worsening renal function, an association not borne out in trials.38, 45, 46 For patients that are not able to tolerate an indicated therapy, such as a beta‐blocker, ACEI, or ARB, the specific contraindication to treatment should be documented in the medical record.
For patients with HFpEF, no therapy has been shown to improve survival.19, 47 The mainstays of therapy are management of congestion, hypertension, and ventricular rate for patients with atrial fibrillation.22 Research into novel therapies for diastolic heart failure is ongoing.48
DISCHARGE
Patients hospitalized for ADHF are at an increased risk for adverse events following discharge. In an analysis of data from the Candesartan in Heart Failure Assessment of Reduction in Mortality and Morbidity (CHARM) trial, the risk of death was 6‐fold higher in the first month after discharge and remained elevated at 2‐fold higher at 2 years after hospitalization, as compared to persons never hospitalized.49 As yet, no model can accurately predict which ADHF patients will require readmission, though multiple clinical factors have been identified.50 In the OPTIMIZE‐HF registry, increasing admission serum creatinine, a history of chronic obstructive pulmonary disease or cerebrovascular disease, hospitalization for heart failure within the last 6 months, as well as treatment with nitrates, digoxin, diuretics, or mechanical ventilation, were all predictors of mortality and rehospitalization within 60 to 90 days after discharge.44 Furthermore, a BNP level of greater than 350 pg/mL or less than a 50% reduction in NT‐BNP during the hospital stay is also associated with an increased risk for rehospitalization or death.51, 52
Unfortunately, few interventions reduce heart failure readmission rates. In a recent analysis of Medicare claims data, hospitals with the highest rates of early follow‐up after discharge (defined as a clinic visit within 7 days of discharge) had decreased rates of readmission within 30 days.53 Thus, early follow‐up after discharge is essential. Not surprisingly, non‐compliance with weight self‐monitoring leads to increased readmission and mortality rates, and therefore patient education is essential.54 The benefit of home telemonitoring programs remains controversial and requires further study.55, 56 At our center, patients are required to follow up with their internist or cardiologist within 7 days of discharge, and the patient's discharge medication list, discharge weight, and laboratory studies on the day of discharge are faxed to the outpatient provider's office to ensure a seamless transition of care.
PERFORMANCE MEASURES AND GUIDELINES
Performance measures are being assessed with greater frequency in medicine to ensure that clinicians perform key assessments and provide treatments that can improve outcomes. Acute and chronic heart failure were 2 of the first areas to be assessed. In 1996, CMS developed a set of 4 measures for inpatient heart failure care (see Supporting Online Table 2 in the online version of this article).57 Each hospital's performance for these 4 measures is now published at the CMS website. The ACC, AHA, and the American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI) released a joint heart failure performance measurement set in 2011. This set removes 3 older recommendations (anticoagulation for patients with atrial fibrillation, discharge instructions, and smoking cessation counseling) and adds 2 new recommendations: prescription of an appropriate beta‐blocker at discharge and arrangement of a postdischarge follow‐up appointment.58, 59 The ACC will publish guidelines based on the ACC/AHA/AMA‐PCPI measure set in early 2012. Of the extant performance measures, both ACEI/ARB and beta‐blocker therapy at discharge are associated with improved outcomes.60, 61
CONCLUSION
With the aging of the population, hospitalizations for ADHF are projected to increase substantially, creating a greater necessity for hospitalists to diagnose, risk stratify, and manage inpatients with heart failure. Once the heart failure diagnosis has been established, determining the etiology of the decompensation and estimating the patient's risk for in‐hospital and postdischarge adverse events is essential. For patients with reduced systolic function, treatment with neurohormonal therapies, even while hospitalized, improves outcomes. Patients should be scheduled for follow‐up within 7 days after discharge to ensure clinical stability. Hospitalists should understand and adhere to the current performance measures for heart failure, as efforts tying payment to the quality of care are likely to evolve.
- ,,,.Heart failure‐related hospitalization in the U.S., 1979 to 2004.J Am Coll Cardiol.2008;52(6):428–434.
- ,,, et al.Forecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart Association.Circulation.2011;123(8):933–944.
- ,,.Rehospitalizations among patients in the Medicare Fee‐for‐Service Program.N Engl J Med.2009;360(14):1418–1428.
- 111th Congress: 2009–2010. H.R.3590: Patient Protection and Affordable Care Act. 2009. Available at: http://www.govtrack.us/congress/bill.xpd?bill=h111–3590294(15):1944–1956.
- ,,, et al.Value of clinician assessment of hemodynamics in advanced heart failure: the ESCAPE Trial.Circ: Heart Fail.2008;1(3):170–177.
- .Heart transplantation in patients with end‐stage heart failure and cardiac ascites.Circ J.2007;71(11):1744–1748.
- ,,, et al.Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure.J Am Coll Cardiol.2003;41(10):1797–1804.
- ,,, et al.Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure.JAMA.2006;296(18):2217–2226.
- ,,, et al.Hemodynamic factors associated with acute decompensated heart failure: part 1‐insights into pathophysiology.J Card Fail.2011;17(4):282–291.
- ,,, et al.State of the art: using natriuretic peptide levels in clinical practice.Eur J Heart Fail.2008;10(9):824–839.
- ,,, et al.How obesity affects the cut‐points for B‐type natriuretic peptide in the diagnosis of acute heart failure: results from the Breathing Not Properly Multinational Study.Am Heart J.2006;151(5):999–1005.
- ,,.Understanding amino‐terminal pro–B‐type natriuretic peptide in obesity.Am J Cardiol.2008;101(3):S89–S94.
- ,,, et al.B‐type natriuretic peptide and renal function in the diagnosis of heart failure: an analysis from the Breathing Not Properly Multinational Study.Am J Kidney Dis.2003;41(3):571–579.
- ,,, et al.Renal function, congestive heart failure, and amino‐terminal pro‐brain natriuretic peptide measurement.J Am Coll Cardiol.2006;47(1):91–97.
- ,,, et al.Optimal noninvasive assessment of left ventricular filling pressures: a comparison of tissue Doppler echocardiography and B‐type natriuretic peptide in patients with pulmonary artery catheters.Circulation.2004;109(20):2432–2439.
- ,,, et al.Clinical and radiologic features of pulmonary edema.Radiographics.1999;19(6):1507–1533.
- ,,,,.Clinical presentation, management, and in‐hospital outcomes of patients admitted with acute decompensated heart failure with preserved systolic function.J Am Coll Cardiol.2006;47(1):76–84.
- ,,, et al.Characteristics, treatments, and outcomes of patients with preserved systolic function hospitalized for heart failure.J Am Coll Cardiol.2007;50(8):768–777.
- ,.Systolic and diastolic heart failure: differences and similarities.J Card Fail.2007;13(7):569–576.
- Department of Health and Human Services. Medicare program; changes to the hospital inpatient prospective payment systems and fiscal year 2008 rates.Fed Reg.2007;72(162):47130–48175.
- ,,, et al.2009 focused update incorporated into the ACC/AHA 2005 guidelines for the diagnosis and management of heart failure in adults.J Am Coll Cardiol.2009;53(15):e1–e90.
- ,,, et al.Factors identified as precipitating hospital admissions for heart failure and clinical outcomes: findings from OPTIMIZE‐HF.Arch Intern Med.2008;168(8):847–854.
- ,,, et al.ADHERE Scientific Advisory Committee, Study Group, and Investigators. Risk stratification for in‐hospital mortality in acutely decompensated heart failure: classification and regression tree analysis.JAMA.2005;293(5):572–580.
- ,,, et al.Predictors of in‐hospital mortality in patients hospitalized for heart failure.J Am Coll Cardiol.2008;52(5):347–356.
- ,,,,.Admission B ‐type natriuretic peptide levels and in‐hospital mortality in acute decompensated heart failure.J Am Coll Cardiol.2007;49(19):1943–1950.
- ,,, et al.Cardiac troponin and outcome in acute heart failure.N Engl J Med.2008;358(20):2117–2126.
- ,,, et al.National Heart, Lung, and Blood Institute working group on emergency department management of acute heart failure: research challenges and opportunities.J Am Coll Cardiol.2010;56(5):343–351.
- ,,, et al.Association of consultation between generalists and cardiologists with quality and outcomes of heart failure care.Am Heart J.2003;145(6):1086–1093.
- ,,,.Congestion in acute heart failure syndromes: an essential target of evaluation and treatment.Am J Med.2006;119(12):S3–S10.
- ,,, et al.Randomised trial of high‐dose isosorbide dinitrate plus low‐dose furosemide versus high‐dose furosemide plus low‐dose isosorbide dinitrate in severe pulmonary oedema.Lancet.1998;351(9100):389–393.
- ,,,,.Furosemide absorption altered in decompensated congestive heart failure.Ann Intern Med.1985;102(3):314–318.
- ,,, et al.Diuretic strategies in patients with acute decompensated heart failure.N Engl J Med.2011;364(9):797–805.
- .Diuretic therapy.N Engl J Med.1998;339(6):387–395.
- .Diuretic resistance predicts mortality in patients with advanced heart failure.Am Heart J.2002;144(1):31–38.
- ,,,.Early ultrafiltration in patients with decompensated heart failure and diuretic resistance.J Am Coll Cardiol.2005;46(11):2047–2051.
- ,,.Relation of loop diuretic dose to mortality in advanced heart failure.Am J Cardiol.2006;97(12):1759–1764.
- ,,, et al.Worsening renal function in patients hospitalised for acute heart failure: clinical implications and prognostic significance.Eur J Heart Fail.2008;10(2):188–195.
- ,,.Combination of loop diuretics with thiazide‐type diuretics in heart failure.J Am Coll Cardiol.2010;56(19):1527–1534.
- ,,, et al.Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure.J Am Coll Cardiol.2007;49(6):675–683.
- ,,, et al.Influence of beta‐blocker continuation or withdrawal on outcomes in patients hospitalized with heart failure.J Am Coll Cardiol.2008;52(3):190–199.
- ,,, et al.Prospective evaluation of beta‐blocker use at the time of hospital discharge as a heart failure performance measure: results from OPTIMIZE‐HF.J Card Fail.2007;13(9):722–731.
- ,,, et al.Influence of pretreatment systolic blood pressure on the effect of carvedilol in patients with severe chronic heart failure.J Am Coll Cardiol.2004;43(8):1423–1429.
- ,,, et al.Predictors of mortality after discharge in patients hospitalized with heart failure: an analysis from the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF).Am Heart J.2008;156(4):662–673.
- ,,, et al.Relationship between heart failure treatment and development of worsening renal function among hospitalized patients.Am Heart J.2004;147(2):331–338.
- ,,, et al.Incidence, predictors at admission, and impact of worsening renal function among patients hospitalized with heart failure.J Am Coll Cardiol.2004;43(1):61–67.
- ,,, et al.Effects of candesartan in patients with chronic heart failure and preserved left‐ventricular ejection fraction: the CHARM‐Preserved Trial.Lancet.2003;362(9386):777–781.
- ,.Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment.Eur Heart J.2011;32(6):670–679.
- ,,, et al.Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure.Circulation.2007;116(13):1482–1487.
- ,,, et al.Statistical models and patient predictors of readmission for heart failure: a systematic review.Arch Intern Med.2008;168(13):1371–1386.
- ,,, et al.Predischarge B‐type natriuretic peptide assay for identifying patients at high risk of re‐admission after decompensated heart failure.J Am Coll Cardiol.2004;43(4):635–641.
- ,,, et al.Acute changes in N‐terminal pro‐B‐type natriuretic peptide during hospitalization and risk of readmission and mortality in patients with heart failure.Am J Cardiol.2011;107(8):1191–1195.
- ,,, et al.Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure.JAMA.2010;303(17):1716–1722.
- ,,,,.Compliance with non‐pharmacological recommendations and outcome in heart failure patients.Eur Heart J.2010;31(12):1486–1493.
- ,,, et al.Telemonitoring in patients with heart failure.N Engl J Med.2010;363(24):2301–2309.
- ,,, et al.Structured telephone support or telemonitoring programmes for patients with chronic heart failure.Cochrane Database Syst Rev.2010;(8):CD007228.
- ,.Heart failure performance measures and outcomes: real or illusory gains.JAMA.2009;302(7):792–794.
- American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI).Heart Failure Performance Measure Set. AMA‐PCPI; February 17,2011;1–85. http://www.ama‐assn.org/ama/pub/physician‐resources/physician‐consortium‐performance‐improvement.page. Accessed December 12, 2011.
- ,,, et al.ACC/AHA clinical performance measures for adults with chronic heart failure.J Am Coll Cardiol.2005;46(6):1144–1178.
- ,,, et al.Association between performance measures and clinical outcomes for patients hospitalized with heart failure.JAMA.2007;297(1):61–70.
- ,,, et al.Relationships between emerging measures of heart failure processes of care and clinical outcomes.Am Heart J.2010;159(3):406–413.
Caring for patients with acute decompensated heart failure (ADHF) is one of the core competencies of practice in hospitalist medicine. Congestive heart failure remains the most common discharge diagnosis as recorded in the National Hospital Discharge Survey, with over 1.1 million hospitalizations for heart failure in 2004.1 Furthermore, with the disproportionate growth in the population over age 65 that will occur over the next 20 years, heart failure prevalence will grow from its current value of 2.8% to 3.5% by 2030.2 This will result in an additional 3 million Americans with chronic heart failure, thereby sustaining ADHF as the most common reason for hospital admission. Despite an average hospital stay of 5 days, the readmission rate for heart failure was 26.9% at 30 days in a 2003‐2004 analysis of Medicare data.3 This high readmission rate is the target of reform as part of the recently passed Patient Protection and Accountability Act. Starting in fiscal year 2013, acute‐care hospitals with higher‐than‐expected readmission rates for heart failure will have a reduction in reimbursement for these admissions.4 Thus, there is substantial incentive for hospitalists to focus on providing the highest quality of care for patients with ADHF. Here we review the most recent evidence applicable to hospitalists for the diagnosis, risk stratification, and management of patients presenting with ADHF.
DIAGNOSIS
The hospitalist can establish the ADHF diagnosis efficiently by applying a structured approach based on the patient's symptoms, history, physical examination, and laboratory testing. The typical symptoms of ADHF include dyspnea, orthopnea, paroxysmal nocturnal dyspnea (PND), and lower extremity edema. In particular, patients complaining of PND and/or orthopnea are likely to have ADHF.5, 6 Patients may also report chest congestion or chest pain in an atypical pattern. A history of rapid weight gain suggests fluid overload, hence determination of the patient's dry weight is important to establish a target for congestive therapy. Patients with advanced systolic heart failure may also complain of nausea, abdominal pain, and abdominal fullness from ascites.7 In a patient with dyspnea, a history of heart failure, myocardial infarction, or coronary artery disease, all make the diagnosis of ADHF more likely.5
Performing a careful physical examination on a patient presenting with suspected ADHF will not only establish the diagnosis of heart failure, but also determine the hemodynamic profile. Patients presenting with ADHF can be separated into 4 hemodynamic profiles, based on vital sign and physical exam parameters: the presence or absence of congestion (wet or dry), and the presence or absence of adequate perfusion (warm or cold) (Figure 1).8 Parameters indicating the presence of congestion include: orthopnea, elevated jugular venous pulsation (JVP), lower extremity edema, hepatojugular reflux, ascites, and a loud P2 heart sound. Notably, rales are an uncommon physical finding in patients with ADHF, likely because pulmonary lymphatics compensate for chronically elevated filling pressures in such patients.9, 10 Parameters indicating inadequate perfusion include: hypotension (mean arterial pressure 60 mmHg), proportional pulse pressure 25%, cool extremities, altered mental status, and poor urine output (0.5 mL/kg/hr). We recommend assigning the patient to 1 of these 4 hemodynamic profiles, as the profile correlates with invasive hemodynamic measurements of pulmonary capillary wedge pressure and cardiac index, guides management, and predicts outcome.

Natriuretic peptide testing may help establish or exclude a diagnosis of ADHF. A recent expert consensus paper on natriuretic peptide testing recommends cutpoints for both B‐type natriuretic peptide (BNP) and N‐terminal proBNP (NT‐BNP) that indicate a very low (BNP 100 or NT‐BNP 300), intermediate (BNP 100‐400 or NT‐BNP 300‐1800), and high (BNP >400 or NT‐BNP >1800) probability of heart failure11 (Figure 2). However, 2 common conditions affect the utility of BNP testing. First, obese patients have lower levels, and thus a lower rule‐out cutpoint of 54 pg/mL is recommended when using BNP, whereas the cutpoint for NT‐BNP remains the same.12, 13 Second, in patients with renal dysfunction, levels are increased, and thus higher rule‐out cutpoints of 200 pg/mL (for BNP) and 1200 pg/mL (for NT‐BNP) are recommended for patients with a glomerular filtration rate 60 mL/min.14, 15 For patients with longstanding heart failure and chronically elevated levels of natriuretic peptides, there is a correlation between BNP levels and left ventricular filling pressure,16 but the change is more helpful than the absolute levels; a 50% increase over baseline, in conjunction with symptoms, usually reflects ADHF.11

Chest radiography will establish the presence or absence of pulmonary congestion. Classic teaching is that congestion starts with cephalization (pulmonary capillary wedge pressure 10‐15 mmHg), progresses to Kerley B lines (15‐20 mmHg), then to interstitial edema (20‐25 mmHg), and finally to alveolar edema (>25 mmHg).17 In patients presenting with dyspnea, any of these findings helps to establish the diagnosis of ADHF.5
MECHANISMS AND TERMINOLOGY
Data from ADHF registries show that hemodynamically stable patients presenting to the hospital with ADHF are an approximately equal mix of heart failure with reduced ejection fraction (HFrEF; ejection fraction 50%) and heart failure with preserved ejection fraction (HFpEF; ejection fraction 50%).18, 19 The important differences between these groups with regards to pathophysiology and etiology have been reviewed elsewhere.20 Establishing the heart failure mechanism (ie, reduced or preserved EF) is important because the medical management is distinct. Patients with HFrEF are more likely to be male, younger in age, to have ischemic heart disease, and to present with normal or low blood pressure. Patients with HFpEF are more likely to be female, older in age, to have hypertension or diabetes mellitus, and to present with elevated blood pressure.18, 19
The terminology used for inpatient heart failure coding has been the subject of renewed focus. For fiscal year 2008, the Centers for Medicare and Medicaid Services (CMS) overhauled its Diagnosis Related Group (DRG) system to better account for the severity of illness of hospitalized patients.21 In this revision, the existing DRG codes for heart failure were subdivided into 3 severity subclasses: major complication, complication, and non‐complication. Payment to hospitals for a heart failure DRG was changed to be proportional to the level of complication. Thus, for the first time, the clinicians' assessment of the acuity of heart failure determines the level of payment to the hospital. Not surprisingly, this has led to initiatives by hospitals to improve clinicians' coding of inpatients hospitalized with heart failure. A major impediment is that there are no established criteria for the application of each DRG code. Table 1 presents recommended clinical criteria for the application of these codes to patients with ADHF.
| ICD‐9 | DRG Code | Severity Subclass | Clinical Criteria | Hemodynamic Profile |
|---|---|---|---|---|
| ||||
| Acute decompensated heart failure | ||||
| 428.21 | Systolic, acute | MCC | New diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.23 | Systolic, acute on chronic | MCC | Established diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.41 | Combined systolic and diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.43 | Combined systolic and diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.31 | Diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| 428.33 | Diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| Chronic heart failure | ||||
| 428.22 | Systolic, chronic | CC | No previous symptoms, or history of clinical features of low‐output state but currently compensated, EF 50 | Dry‐warm |
| 428.40 | Combined systolic and diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| 428.32 | Diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| Other | ||||
| 428.1 | Left heart failure | CC | Clinical features of congestion, mechanism and EF is unknown | Wet‐warm or wet‐cold |
| 428.20 | Systolic heart failure, unspecified | CC | Clinical features of low‐output, acuity is unknown | Dry‐cold |
| 428.0 | Congestive heart failure, unspecified | NCC | Clinical features of right‐heart failure | Not applicable |
PRECIPITANTS AND ETIOLOGY
For patients presenting for the first time with a diagnosis of ADHF (de novo), a thorough evaluation should be performed to determine the mechanism and etiology of the patient's left ventricular dysfunction. After the initial history and physical exam, the American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend checking basic laboratory studies, an electrocardiogram, and an echocardiogram.22 The full assessment recommended by the ACC/AHA is detailed in Supporting Online Table 1 (in the online version of this article). Cardiac ischemia is the most common etiology of HFrEF, accounting for about 50% of cases. The common, non‐ischemic causes of systolic heart failure include atrial fibrillation, aortic stenosis, illicit cardiotoxic drugs (cocaine, methamphetamine), medical cardiotoxic drugs (adriamycin), as well as primary myocardial disorders such as myocarditis, idiopathic, or peripartum cardiomyopathy. HFpEF is most commonly associated with long‐standing hypertension and diabetes mellitus, but can also be caused by infiltrative, hypertrophic, and constrictive cardiomyopathies.
For patients with a history of heart failure, it is important to identify the precipitant for the decompensation, as it may be treated or avoided in the future. When no clear precipitant is identified, this is most concerning, as it indicates the patient's tenuous cardiac function. In the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF) registry, approximately 61% of patients were found to have at least 1 precipitating factor.23 The most common precipitants were respiratory process in 15.3%, acute coronary syndrome in 14.7%, arrhythmia in 13.5%, uncontrolled hypertension in 10.7%, medication non‐compliance in 8.9%, worsening renal function in 8.0%, and dietary non‐compliance in 5.2%.
RISK STRATIFICATION
Patients hospitalized with ADHF are at a significantly elevated risk for death, both during their hospitalization and after discharge. Numerous studies have shown that multiple clinical parameters assessed during the hospitalization, such as vital signs and laboratory values, predict outcome.6, 8, 24, 25 Some of the most elegant parameters are physical exam findings. As introduced above, the wet‐cold hemodynamic profile assessed at admission predicts increased mortality and urgent transplantation at 1 year.8 One of the most powerful risk stratification schemes for in‐hospital mortality is that developed from the Acute Decompensated Heart Failure (ADHERE) national registry. Three clinical parameters, blood urea nitrogen (BUN) >43 mg/dL, systolic blood pressure 115 mmHg, and serum creatinine >2.75 mg/dL, stratified patients into risk groups. Patients exhibiting all 3 parameters had a 22% in‐hospital mortality compared with 2% for patients with none of the 3 parameters.24
BNP and troponin also have a role in risk stratification of patients with ADHF. In the ADHERE registry, for every increase in the BNP of 400 pg/mL, the odds of risk‐adjusted mortality increased by 9%, in patients with both HFrEF and HFpEF.26 Similarly, an elevated admission troponin was associated with an in‐hospital mortality of 8.0%, versus 2.7% for troponin‐negative patients27; notably almost half of patients with a positive troponin had no history of ischemic heart disease. In the future, refinement and widespread application of these risk stratification methods should allow clinicians to triage patients to determine their location (eg, observation unit, inpatient, intensive care unit) and type of treatment (eg, oral or intravenous diuretic, vasodilator, inotrope).28
In the community, hospitalists care for many patients with ADHF without input from a cardiologist.29 However, there are several situations where the patient is at an increased risk of adverse outcomes, and therefore in which we recommend consulting a cardiologist (Table 2). Patients with hypotension, a cold hemodynamic profile, or worsening renal function due to poor cardiac function are at an especially elevated risk and should be considered for advanced therapies such as mechanical circulatory support or heart transplantation.
| Results of Evaluation | Indication for Referral | Purpose of Referral |
|---|---|---|
| ||
| Hypotension, cold hemodynamic profile | Inadequate perfusion | Pulmonary artery catheterization, inotropic therapy |
| Ischemic symptoms, positive troponin, abnormal ECG, echocardiogram with focal wall motion abnormalities | Cardiac ischemia | Coronary angiography and coronary intervention if indicated |
| Atrial fibrillation | Arrhythmia | Consideration of a rhythm control strategy |
| Ejection fraction 35% | Severe left ventricular systolic dysfunction | Implantable cardiac defibrillator and/or biventricular pacemaker |
| High diuretic dose requirements or decreasing urinary response to diuretics | Diuretic resistance | Consideration for vasodilator therapy or ultrafiltration |
| Increasing blood urea nitrogen and serum creatinine, decreasing urine output | Worsening renal function | Consideration of inotropic therapy |
CONGESTION AND DIURESIS
The syndrome of heart failure is due primarily to elevation of left ventricular filling pressures resulting in congestion, and therapies aimed at reducing congestion are of primary importance.30 For 50 years, treatment with diuretic medications has been the mainstay of therapy for patients admitted with ADHF. Vasodilator agents, specifically nitroglycerin and sodium nitroprusside, may also be beneficial in patients presenting with ADHF and hypertension.22 In 1 study, patients with acute pulmonary edema treated with high‐dose nitroglycerin experienced fewer adverse events as compared to those treated with high‐dose diuretics alone, suggesting that nitrates can more rapidly decrease congestion and thereby improve outcomes.31 Unfortunately vasodilators are underutilized, with only 5.8% of patients with elevated blood pressure (>160 mmHg) treated with nitrates in the OPTIMIZE‐HF registry.9
Diuretic choice, dosing, and administration method have traditionally been highly variable between practitioners. Oral diuretics are generally not preferred initially for patients with ADHF because of concerns of inadequate absorption from an edematous bowel and slow onset of action.32 For a patient who is not on diuretics as an outpatient, an initial dose of 40 mg intravenous furosemide is reasonable. For a patient with chronic heart failure on outpatient loop diuretic therapy, the Diuretic Optimization Strategies Evaluation (DOSE) study provides insight into diuretic dosing and administration. Patients were randomized to an administration route (bolus dosing every 12 hours or continuous infusion) and a dosing strategy (low‐dose or high‐dose).33 There were no differences in the primary endpoint of patient‐reported global assessment of symptoms, or the primary safety endpoint of change in serum creatinine from baseline to 72 hours between the bolus and continuous infusion groups or between the low‐dose and high‐dose groups. However, patients in the high‐dose group had decreased dyspnea at 72 hours, decreased body weight at 72 hours, increased fluid loss at 72 hours, and decreased NT‐BNP at 72 hours. These improvements came at the expense of a mild increase in creatinine. Therefore, in hospitalized patients with ADHF on outpatient furosemide, these data support initiation of high‐dose furosemide with a daily intravenous dose equal to 2.5 times their daily outpatient oral dose, using either bolus or continuous infusion.
All patients being treated with diuretic therapy should have close fluid intake and output monitoring, fluid restriction of 1500 to 2000 mL per day, a 2 gram sodium diet, and at least daily electrolyte monitoring. For patients with inadequate diuresis (generally less than 1 L per day in a patient with moderate volume overload), several options are available. If the urinary response to a furosemide dose is inadequate, the dose should be doubled and the urinary response followed. If there has been inadequate diuresis in a patient with a low serum albumin or significant proteinuria, furosemide should be switched to bumetanide, which is not protein‐bound and thus will achieve higher concentrations in the tubule.34
Longstanding treatment with loop diuretics leads to decreased renal responsiveness and an increased dose required to maintain euvolemia. Patients taking furosemide 80 mg daily or above (or an equivalent dose of other loop diuretics) are designated as diuretic‐resistant.35, 36 Diuretic resistance is associated with more severe heart failure, more advanced chronic kidney disease, and worsening renal function with the use of intravenous diuretics.35, 37, 38 There are no consensus recommendations available to guide the management of diuretic resistance, but several options exist. First, a thiazide diuretic, such as metolazone, can be given before the loop diuretic.39 This combination is frequently able to initiate a brisk diuresis, but patients require close monitoring for hypokalemia and worsening renal function. Recently, ultrafiltration has emerged as an option. In the Ultrafiltration versus Intravenous Diuretics for Patients Hospitalized for Acute Decompensated Congestive Heart Failure (UNLOAD) trial, patients with congestion treated with ultrafiltration had more weight and fluid loss at 48 hours compared to patients treated with intravenous furosemide, without any significant differences in renal function.40 For patients with oliguria and renal dysfunction, initiation of renal replacement therapy may be needed. We present an algorithm for the management of diuretic resistance in Figure 3.

The endpoints for discontinuation of diuretic therapy remain unclear. Traditionally, alleviation of the patient's congestive symptoms, edema, and attainment of the patient's self‐reported dry weight have served as endpoints for diuretic therapy. A more accurate approach may be daily assessments of the JVP, as normalization of the JVP may be a more accurate method to assess for euvolemia. When euvolemia has been achieved, patients should be switched to maintenance therapy at a diuretic dose of one‐fourth to one‐half the total daily dose used for diuresis. Patients should be observed for 24 hours on oral diuretic therapy to ensure that their fluid intake and output are balanced. Generally, we aim for slightly negative fluid balance (less than 500 mL) on an oral diuretic regimen prior to discharge, assuming some relaxation of the salt and fluid restriction once the patient is discharged home.
NEUROHORMONAL THERAPIES
Activation of neurohormonal systems, specifically the renin‐angiotensin‐aldosterone and beta‐adrenergic pathways, are the major mechanisms for disease progression in HFrEF, and agents which block these pathways improve functional status and survival in these patients. In the OPTIMIZE‐HF registry, patients treated with beta‐blockers on admission had a lower in‐hospital mortality.25 Although beta‐blockers are often discontinued in patients with ADHF, continuation of beta‐blocker treatment is associated with decreased mortality and rehospitalization at 60 to 90 days.41 While beta‐blocker initiation is often deferred to the outpatient setting, patients who receive a beta‐blocker at hospital discharge are 31 times more likely to be treated with a beta‐blocker at 60 to 90 day follow‐up.42 Only 3 agents, metoprolol succinate, carvedilol, and bisoprolol, have survival benefit in large clinical trials of systolic heart failure, and therefore are the only recommended agents.22 In the hospital, hypotension is a common reason for suspension or discontinuation of beta‐blocker therapy. However, in the absence of symptoms such as light‐headedness, patients with systolic blood pressure as low as 85 mmHg will benefit from beta‐blocker treatment.43 Thus, we recommend continuation or initiation of an evidence‐based beta‐blocker for all patients hospitalized with systolic heart failure in the absence of symptomatic hypotension, systolic blood pressure 85 mmHg, second or third degree heart block, or the need for intravenous inotropic therapy.
Inhibitors of the renin‐angiotensin‐aldosterone system also have an important role in patients with HFrEF. Patients treated with angiotensin converting enzyme inhibitors (ACEI) on admission have a lower in‐hospital mortality25 and a lower likelihood of readmission or death within 60 to 90 days.44 In practice, ACEI or angiotensin receptor blocker (ARB) treatment is frequently suspended or discontinued during treatment with diuretics out of concerns for worsening renal function, an association not borne out in trials.38, 45, 46 For patients that are not able to tolerate an indicated therapy, such as a beta‐blocker, ACEI, or ARB, the specific contraindication to treatment should be documented in the medical record.
For patients with HFpEF, no therapy has been shown to improve survival.19, 47 The mainstays of therapy are management of congestion, hypertension, and ventricular rate for patients with atrial fibrillation.22 Research into novel therapies for diastolic heart failure is ongoing.48
DISCHARGE
Patients hospitalized for ADHF are at an increased risk for adverse events following discharge. In an analysis of data from the Candesartan in Heart Failure Assessment of Reduction in Mortality and Morbidity (CHARM) trial, the risk of death was 6‐fold higher in the first month after discharge and remained elevated at 2‐fold higher at 2 years after hospitalization, as compared to persons never hospitalized.49 As yet, no model can accurately predict which ADHF patients will require readmission, though multiple clinical factors have been identified.50 In the OPTIMIZE‐HF registry, increasing admission serum creatinine, a history of chronic obstructive pulmonary disease or cerebrovascular disease, hospitalization for heart failure within the last 6 months, as well as treatment with nitrates, digoxin, diuretics, or mechanical ventilation, were all predictors of mortality and rehospitalization within 60 to 90 days after discharge.44 Furthermore, a BNP level of greater than 350 pg/mL or less than a 50% reduction in NT‐BNP during the hospital stay is also associated with an increased risk for rehospitalization or death.51, 52
Unfortunately, few interventions reduce heart failure readmission rates. In a recent analysis of Medicare claims data, hospitals with the highest rates of early follow‐up after discharge (defined as a clinic visit within 7 days of discharge) had decreased rates of readmission within 30 days.53 Thus, early follow‐up after discharge is essential. Not surprisingly, non‐compliance with weight self‐monitoring leads to increased readmission and mortality rates, and therefore patient education is essential.54 The benefit of home telemonitoring programs remains controversial and requires further study.55, 56 At our center, patients are required to follow up with their internist or cardiologist within 7 days of discharge, and the patient's discharge medication list, discharge weight, and laboratory studies on the day of discharge are faxed to the outpatient provider's office to ensure a seamless transition of care.
PERFORMANCE MEASURES AND GUIDELINES
Performance measures are being assessed with greater frequency in medicine to ensure that clinicians perform key assessments and provide treatments that can improve outcomes. Acute and chronic heart failure were 2 of the first areas to be assessed. In 1996, CMS developed a set of 4 measures for inpatient heart failure care (see Supporting Online Table 2 in the online version of this article).57 Each hospital's performance for these 4 measures is now published at the CMS website. The ACC, AHA, and the American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI) released a joint heart failure performance measurement set in 2011. This set removes 3 older recommendations (anticoagulation for patients with atrial fibrillation, discharge instructions, and smoking cessation counseling) and adds 2 new recommendations: prescription of an appropriate beta‐blocker at discharge and arrangement of a postdischarge follow‐up appointment.58, 59 The ACC will publish guidelines based on the ACC/AHA/AMA‐PCPI measure set in early 2012. Of the extant performance measures, both ACEI/ARB and beta‐blocker therapy at discharge are associated with improved outcomes.60, 61
CONCLUSION
With the aging of the population, hospitalizations for ADHF are projected to increase substantially, creating a greater necessity for hospitalists to diagnose, risk stratify, and manage inpatients with heart failure. Once the heart failure diagnosis has been established, determining the etiology of the decompensation and estimating the patient's risk for in‐hospital and postdischarge adverse events is essential. For patients with reduced systolic function, treatment with neurohormonal therapies, even while hospitalized, improves outcomes. Patients should be scheduled for follow‐up within 7 days after discharge to ensure clinical stability. Hospitalists should understand and adhere to the current performance measures for heart failure, as efforts tying payment to the quality of care are likely to evolve.
Caring for patients with acute decompensated heart failure (ADHF) is one of the core competencies of practice in hospitalist medicine. Congestive heart failure remains the most common discharge diagnosis as recorded in the National Hospital Discharge Survey, with over 1.1 million hospitalizations for heart failure in 2004.1 Furthermore, with the disproportionate growth in the population over age 65 that will occur over the next 20 years, heart failure prevalence will grow from its current value of 2.8% to 3.5% by 2030.2 This will result in an additional 3 million Americans with chronic heart failure, thereby sustaining ADHF as the most common reason for hospital admission. Despite an average hospital stay of 5 days, the readmission rate for heart failure was 26.9% at 30 days in a 2003‐2004 analysis of Medicare data.3 This high readmission rate is the target of reform as part of the recently passed Patient Protection and Accountability Act. Starting in fiscal year 2013, acute‐care hospitals with higher‐than‐expected readmission rates for heart failure will have a reduction in reimbursement for these admissions.4 Thus, there is substantial incentive for hospitalists to focus on providing the highest quality of care for patients with ADHF. Here we review the most recent evidence applicable to hospitalists for the diagnosis, risk stratification, and management of patients presenting with ADHF.
DIAGNOSIS
The hospitalist can establish the ADHF diagnosis efficiently by applying a structured approach based on the patient's symptoms, history, physical examination, and laboratory testing. The typical symptoms of ADHF include dyspnea, orthopnea, paroxysmal nocturnal dyspnea (PND), and lower extremity edema. In particular, patients complaining of PND and/or orthopnea are likely to have ADHF.5, 6 Patients may also report chest congestion or chest pain in an atypical pattern. A history of rapid weight gain suggests fluid overload, hence determination of the patient's dry weight is important to establish a target for congestive therapy. Patients with advanced systolic heart failure may also complain of nausea, abdominal pain, and abdominal fullness from ascites.7 In a patient with dyspnea, a history of heart failure, myocardial infarction, or coronary artery disease, all make the diagnosis of ADHF more likely.5
Performing a careful physical examination on a patient presenting with suspected ADHF will not only establish the diagnosis of heart failure, but also determine the hemodynamic profile. Patients presenting with ADHF can be separated into 4 hemodynamic profiles, based on vital sign and physical exam parameters: the presence or absence of congestion (wet or dry), and the presence or absence of adequate perfusion (warm or cold) (Figure 1).8 Parameters indicating the presence of congestion include: orthopnea, elevated jugular venous pulsation (JVP), lower extremity edema, hepatojugular reflux, ascites, and a loud P2 heart sound. Notably, rales are an uncommon physical finding in patients with ADHF, likely because pulmonary lymphatics compensate for chronically elevated filling pressures in such patients.9, 10 Parameters indicating inadequate perfusion include: hypotension (mean arterial pressure 60 mmHg), proportional pulse pressure 25%, cool extremities, altered mental status, and poor urine output (0.5 mL/kg/hr). We recommend assigning the patient to 1 of these 4 hemodynamic profiles, as the profile correlates with invasive hemodynamic measurements of pulmonary capillary wedge pressure and cardiac index, guides management, and predicts outcome.

Natriuretic peptide testing may help establish or exclude a diagnosis of ADHF. A recent expert consensus paper on natriuretic peptide testing recommends cutpoints for both B‐type natriuretic peptide (BNP) and N‐terminal proBNP (NT‐BNP) that indicate a very low (BNP 100 or NT‐BNP 300), intermediate (BNP 100‐400 or NT‐BNP 300‐1800), and high (BNP >400 or NT‐BNP >1800) probability of heart failure11 (Figure 2). However, 2 common conditions affect the utility of BNP testing. First, obese patients have lower levels, and thus a lower rule‐out cutpoint of 54 pg/mL is recommended when using BNP, whereas the cutpoint for NT‐BNP remains the same.12, 13 Second, in patients with renal dysfunction, levels are increased, and thus higher rule‐out cutpoints of 200 pg/mL (for BNP) and 1200 pg/mL (for NT‐BNP) are recommended for patients with a glomerular filtration rate 60 mL/min.14, 15 For patients with longstanding heart failure and chronically elevated levels of natriuretic peptides, there is a correlation between BNP levels and left ventricular filling pressure,16 but the change is more helpful than the absolute levels; a 50% increase over baseline, in conjunction with symptoms, usually reflects ADHF.11

Chest radiography will establish the presence or absence of pulmonary congestion. Classic teaching is that congestion starts with cephalization (pulmonary capillary wedge pressure 10‐15 mmHg), progresses to Kerley B lines (15‐20 mmHg), then to interstitial edema (20‐25 mmHg), and finally to alveolar edema (>25 mmHg).17 In patients presenting with dyspnea, any of these findings helps to establish the diagnosis of ADHF.5
MECHANISMS AND TERMINOLOGY
Data from ADHF registries show that hemodynamically stable patients presenting to the hospital with ADHF are an approximately equal mix of heart failure with reduced ejection fraction (HFrEF; ejection fraction 50%) and heart failure with preserved ejection fraction (HFpEF; ejection fraction 50%).18, 19 The important differences between these groups with regards to pathophysiology and etiology have been reviewed elsewhere.20 Establishing the heart failure mechanism (ie, reduced or preserved EF) is important because the medical management is distinct. Patients with HFrEF are more likely to be male, younger in age, to have ischemic heart disease, and to present with normal or low blood pressure. Patients with HFpEF are more likely to be female, older in age, to have hypertension or diabetes mellitus, and to present with elevated blood pressure.18, 19
The terminology used for inpatient heart failure coding has been the subject of renewed focus. For fiscal year 2008, the Centers for Medicare and Medicaid Services (CMS) overhauled its Diagnosis Related Group (DRG) system to better account for the severity of illness of hospitalized patients.21 In this revision, the existing DRG codes for heart failure were subdivided into 3 severity subclasses: major complication, complication, and non‐complication. Payment to hospitals for a heart failure DRG was changed to be proportional to the level of complication. Thus, for the first time, the clinicians' assessment of the acuity of heart failure determines the level of payment to the hospital. Not surprisingly, this has led to initiatives by hospitals to improve clinicians' coding of inpatients hospitalized with heart failure. A major impediment is that there are no established criteria for the application of each DRG code. Table 1 presents recommended clinical criteria for the application of these codes to patients with ADHF.
| ICD‐9 | DRG Code | Severity Subclass | Clinical Criteria | Hemodynamic Profile |
|---|---|---|---|---|
| ||||
| Acute decompensated heart failure | ||||
| 428.21 | Systolic, acute | MCC | New diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.23 | Systolic, acute on chronic | MCC | Established diagnosis, clinical features of low‐output or cold state, EF 30 | Dry‐cold |
| 428.41 | Combined systolic and diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.43 | Combined systolic and diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm or wet‐cold |
| 428.31 | Diastolic, acute | MCC | New diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| 428.33 | Diastolic, acute on chronic | MCC | Established diagnosis, clinical features of congestion, EF 50 | Wet‐warm |
| Chronic heart failure | ||||
| 428.22 | Systolic, chronic | CC | No previous symptoms, or history of clinical features of low‐output state but currently compensated, EF 50 | Dry‐warm |
| 428.40 | Combined systolic and diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| 428.32 | Diastolic, chronic | CC | History of clinical features of congestion but currently compensated, EF 50 | Dry‐warm |
| Other | ||||
| 428.1 | Left heart failure | CC | Clinical features of congestion, mechanism and EF is unknown | Wet‐warm or wet‐cold |
| 428.20 | Systolic heart failure, unspecified | CC | Clinical features of low‐output, acuity is unknown | Dry‐cold |
| 428.0 | Congestive heart failure, unspecified | NCC | Clinical features of right‐heart failure | Not applicable |
PRECIPITANTS AND ETIOLOGY
For patients presenting for the first time with a diagnosis of ADHF (de novo), a thorough evaluation should be performed to determine the mechanism and etiology of the patient's left ventricular dysfunction. After the initial history and physical exam, the American College of Cardiology/American Heart Association (ACC/AHA) guidelines recommend checking basic laboratory studies, an electrocardiogram, and an echocardiogram.22 The full assessment recommended by the ACC/AHA is detailed in Supporting Online Table 1 (in the online version of this article). Cardiac ischemia is the most common etiology of HFrEF, accounting for about 50% of cases. The common, non‐ischemic causes of systolic heart failure include atrial fibrillation, aortic stenosis, illicit cardiotoxic drugs (cocaine, methamphetamine), medical cardiotoxic drugs (adriamycin), as well as primary myocardial disorders such as myocarditis, idiopathic, or peripartum cardiomyopathy. HFpEF is most commonly associated with long‐standing hypertension and diabetes mellitus, but can also be caused by infiltrative, hypertrophic, and constrictive cardiomyopathies.
For patients with a history of heart failure, it is important to identify the precipitant for the decompensation, as it may be treated or avoided in the future. When no clear precipitant is identified, this is most concerning, as it indicates the patient's tenuous cardiac function. In the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF) registry, approximately 61% of patients were found to have at least 1 precipitating factor.23 The most common precipitants were respiratory process in 15.3%, acute coronary syndrome in 14.7%, arrhythmia in 13.5%, uncontrolled hypertension in 10.7%, medication non‐compliance in 8.9%, worsening renal function in 8.0%, and dietary non‐compliance in 5.2%.
RISK STRATIFICATION
Patients hospitalized with ADHF are at a significantly elevated risk for death, both during their hospitalization and after discharge. Numerous studies have shown that multiple clinical parameters assessed during the hospitalization, such as vital signs and laboratory values, predict outcome.6, 8, 24, 25 Some of the most elegant parameters are physical exam findings. As introduced above, the wet‐cold hemodynamic profile assessed at admission predicts increased mortality and urgent transplantation at 1 year.8 One of the most powerful risk stratification schemes for in‐hospital mortality is that developed from the Acute Decompensated Heart Failure (ADHERE) national registry. Three clinical parameters, blood urea nitrogen (BUN) >43 mg/dL, systolic blood pressure 115 mmHg, and serum creatinine >2.75 mg/dL, stratified patients into risk groups. Patients exhibiting all 3 parameters had a 22% in‐hospital mortality compared with 2% for patients with none of the 3 parameters.24
BNP and troponin also have a role in risk stratification of patients with ADHF. In the ADHERE registry, for every increase in the BNP of 400 pg/mL, the odds of risk‐adjusted mortality increased by 9%, in patients with both HFrEF and HFpEF.26 Similarly, an elevated admission troponin was associated with an in‐hospital mortality of 8.0%, versus 2.7% for troponin‐negative patients27; notably almost half of patients with a positive troponin had no history of ischemic heart disease. In the future, refinement and widespread application of these risk stratification methods should allow clinicians to triage patients to determine their location (eg, observation unit, inpatient, intensive care unit) and type of treatment (eg, oral or intravenous diuretic, vasodilator, inotrope).28
In the community, hospitalists care for many patients with ADHF without input from a cardiologist.29 However, there are several situations where the patient is at an increased risk of adverse outcomes, and therefore in which we recommend consulting a cardiologist (Table 2). Patients with hypotension, a cold hemodynamic profile, or worsening renal function due to poor cardiac function are at an especially elevated risk and should be considered for advanced therapies such as mechanical circulatory support or heart transplantation.
| Results of Evaluation | Indication for Referral | Purpose of Referral |
|---|---|---|
| ||
| Hypotension, cold hemodynamic profile | Inadequate perfusion | Pulmonary artery catheterization, inotropic therapy |
| Ischemic symptoms, positive troponin, abnormal ECG, echocardiogram with focal wall motion abnormalities | Cardiac ischemia | Coronary angiography and coronary intervention if indicated |
| Atrial fibrillation | Arrhythmia | Consideration of a rhythm control strategy |
| Ejection fraction 35% | Severe left ventricular systolic dysfunction | Implantable cardiac defibrillator and/or biventricular pacemaker |
| High diuretic dose requirements or decreasing urinary response to diuretics | Diuretic resistance | Consideration for vasodilator therapy or ultrafiltration |
| Increasing blood urea nitrogen and serum creatinine, decreasing urine output | Worsening renal function | Consideration of inotropic therapy |
CONGESTION AND DIURESIS
The syndrome of heart failure is due primarily to elevation of left ventricular filling pressures resulting in congestion, and therapies aimed at reducing congestion are of primary importance.30 For 50 years, treatment with diuretic medications has been the mainstay of therapy for patients admitted with ADHF. Vasodilator agents, specifically nitroglycerin and sodium nitroprusside, may also be beneficial in patients presenting with ADHF and hypertension.22 In 1 study, patients with acute pulmonary edema treated with high‐dose nitroglycerin experienced fewer adverse events as compared to those treated with high‐dose diuretics alone, suggesting that nitrates can more rapidly decrease congestion and thereby improve outcomes.31 Unfortunately vasodilators are underutilized, with only 5.8% of patients with elevated blood pressure (>160 mmHg) treated with nitrates in the OPTIMIZE‐HF registry.9
Diuretic choice, dosing, and administration method have traditionally been highly variable between practitioners. Oral diuretics are generally not preferred initially for patients with ADHF because of concerns of inadequate absorption from an edematous bowel and slow onset of action.32 For a patient who is not on diuretics as an outpatient, an initial dose of 40 mg intravenous furosemide is reasonable. For a patient with chronic heart failure on outpatient loop diuretic therapy, the Diuretic Optimization Strategies Evaluation (DOSE) study provides insight into diuretic dosing and administration. Patients were randomized to an administration route (bolus dosing every 12 hours or continuous infusion) and a dosing strategy (low‐dose or high‐dose).33 There were no differences in the primary endpoint of patient‐reported global assessment of symptoms, or the primary safety endpoint of change in serum creatinine from baseline to 72 hours between the bolus and continuous infusion groups or between the low‐dose and high‐dose groups. However, patients in the high‐dose group had decreased dyspnea at 72 hours, decreased body weight at 72 hours, increased fluid loss at 72 hours, and decreased NT‐BNP at 72 hours. These improvements came at the expense of a mild increase in creatinine. Therefore, in hospitalized patients with ADHF on outpatient furosemide, these data support initiation of high‐dose furosemide with a daily intravenous dose equal to 2.5 times their daily outpatient oral dose, using either bolus or continuous infusion.
All patients being treated with diuretic therapy should have close fluid intake and output monitoring, fluid restriction of 1500 to 2000 mL per day, a 2 gram sodium diet, and at least daily electrolyte monitoring. For patients with inadequate diuresis (generally less than 1 L per day in a patient with moderate volume overload), several options are available. If the urinary response to a furosemide dose is inadequate, the dose should be doubled and the urinary response followed. If there has been inadequate diuresis in a patient with a low serum albumin or significant proteinuria, furosemide should be switched to bumetanide, which is not protein‐bound and thus will achieve higher concentrations in the tubule.34
Longstanding treatment with loop diuretics leads to decreased renal responsiveness and an increased dose required to maintain euvolemia. Patients taking furosemide 80 mg daily or above (or an equivalent dose of other loop diuretics) are designated as diuretic‐resistant.35, 36 Diuretic resistance is associated with more severe heart failure, more advanced chronic kidney disease, and worsening renal function with the use of intravenous diuretics.35, 37, 38 There are no consensus recommendations available to guide the management of diuretic resistance, but several options exist. First, a thiazide diuretic, such as metolazone, can be given before the loop diuretic.39 This combination is frequently able to initiate a brisk diuresis, but patients require close monitoring for hypokalemia and worsening renal function. Recently, ultrafiltration has emerged as an option. In the Ultrafiltration versus Intravenous Diuretics for Patients Hospitalized for Acute Decompensated Congestive Heart Failure (UNLOAD) trial, patients with congestion treated with ultrafiltration had more weight and fluid loss at 48 hours compared to patients treated with intravenous furosemide, without any significant differences in renal function.40 For patients with oliguria and renal dysfunction, initiation of renal replacement therapy may be needed. We present an algorithm for the management of diuretic resistance in Figure 3.

The endpoints for discontinuation of diuretic therapy remain unclear. Traditionally, alleviation of the patient's congestive symptoms, edema, and attainment of the patient's self‐reported dry weight have served as endpoints for diuretic therapy. A more accurate approach may be daily assessments of the JVP, as normalization of the JVP may be a more accurate method to assess for euvolemia. When euvolemia has been achieved, patients should be switched to maintenance therapy at a diuretic dose of one‐fourth to one‐half the total daily dose used for diuresis. Patients should be observed for 24 hours on oral diuretic therapy to ensure that their fluid intake and output are balanced. Generally, we aim for slightly negative fluid balance (less than 500 mL) on an oral diuretic regimen prior to discharge, assuming some relaxation of the salt and fluid restriction once the patient is discharged home.
NEUROHORMONAL THERAPIES
Activation of neurohormonal systems, specifically the renin‐angiotensin‐aldosterone and beta‐adrenergic pathways, are the major mechanisms for disease progression in HFrEF, and agents which block these pathways improve functional status and survival in these patients. In the OPTIMIZE‐HF registry, patients treated with beta‐blockers on admission had a lower in‐hospital mortality.25 Although beta‐blockers are often discontinued in patients with ADHF, continuation of beta‐blocker treatment is associated with decreased mortality and rehospitalization at 60 to 90 days.41 While beta‐blocker initiation is often deferred to the outpatient setting, patients who receive a beta‐blocker at hospital discharge are 31 times more likely to be treated with a beta‐blocker at 60 to 90 day follow‐up.42 Only 3 agents, metoprolol succinate, carvedilol, and bisoprolol, have survival benefit in large clinical trials of systolic heart failure, and therefore are the only recommended agents.22 In the hospital, hypotension is a common reason for suspension or discontinuation of beta‐blocker therapy. However, in the absence of symptoms such as light‐headedness, patients with systolic blood pressure as low as 85 mmHg will benefit from beta‐blocker treatment.43 Thus, we recommend continuation or initiation of an evidence‐based beta‐blocker for all patients hospitalized with systolic heart failure in the absence of symptomatic hypotension, systolic blood pressure 85 mmHg, second or third degree heart block, or the need for intravenous inotropic therapy.
Inhibitors of the renin‐angiotensin‐aldosterone system also have an important role in patients with HFrEF. Patients treated with angiotensin converting enzyme inhibitors (ACEI) on admission have a lower in‐hospital mortality25 and a lower likelihood of readmission or death within 60 to 90 days.44 In practice, ACEI or angiotensin receptor blocker (ARB) treatment is frequently suspended or discontinued during treatment with diuretics out of concerns for worsening renal function, an association not borne out in trials.38, 45, 46 For patients that are not able to tolerate an indicated therapy, such as a beta‐blocker, ACEI, or ARB, the specific contraindication to treatment should be documented in the medical record.
For patients with HFpEF, no therapy has been shown to improve survival.19, 47 The mainstays of therapy are management of congestion, hypertension, and ventricular rate for patients with atrial fibrillation.22 Research into novel therapies for diastolic heart failure is ongoing.48
DISCHARGE
Patients hospitalized for ADHF are at an increased risk for adverse events following discharge. In an analysis of data from the Candesartan in Heart Failure Assessment of Reduction in Mortality and Morbidity (CHARM) trial, the risk of death was 6‐fold higher in the first month after discharge and remained elevated at 2‐fold higher at 2 years after hospitalization, as compared to persons never hospitalized.49 As yet, no model can accurately predict which ADHF patients will require readmission, though multiple clinical factors have been identified.50 In the OPTIMIZE‐HF registry, increasing admission serum creatinine, a history of chronic obstructive pulmonary disease or cerebrovascular disease, hospitalization for heart failure within the last 6 months, as well as treatment with nitrates, digoxin, diuretics, or mechanical ventilation, were all predictors of mortality and rehospitalization within 60 to 90 days after discharge.44 Furthermore, a BNP level of greater than 350 pg/mL or less than a 50% reduction in NT‐BNP during the hospital stay is also associated with an increased risk for rehospitalization or death.51, 52
Unfortunately, few interventions reduce heart failure readmission rates. In a recent analysis of Medicare claims data, hospitals with the highest rates of early follow‐up after discharge (defined as a clinic visit within 7 days of discharge) had decreased rates of readmission within 30 days.53 Thus, early follow‐up after discharge is essential. Not surprisingly, non‐compliance with weight self‐monitoring leads to increased readmission and mortality rates, and therefore patient education is essential.54 The benefit of home telemonitoring programs remains controversial and requires further study.55, 56 At our center, patients are required to follow up with their internist or cardiologist within 7 days of discharge, and the patient's discharge medication list, discharge weight, and laboratory studies on the day of discharge are faxed to the outpatient provider's office to ensure a seamless transition of care.
PERFORMANCE MEASURES AND GUIDELINES
Performance measures are being assessed with greater frequency in medicine to ensure that clinicians perform key assessments and provide treatments that can improve outcomes. Acute and chronic heart failure were 2 of the first areas to be assessed. In 1996, CMS developed a set of 4 measures for inpatient heart failure care (see Supporting Online Table 2 in the online version of this article).57 Each hospital's performance for these 4 measures is now published at the CMS website. The ACC, AHA, and the American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI) released a joint heart failure performance measurement set in 2011. This set removes 3 older recommendations (anticoagulation for patients with atrial fibrillation, discharge instructions, and smoking cessation counseling) and adds 2 new recommendations: prescription of an appropriate beta‐blocker at discharge and arrangement of a postdischarge follow‐up appointment.58, 59 The ACC will publish guidelines based on the ACC/AHA/AMA‐PCPI measure set in early 2012. Of the extant performance measures, both ACEI/ARB and beta‐blocker therapy at discharge are associated with improved outcomes.60, 61
CONCLUSION
With the aging of the population, hospitalizations for ADHF are projected to increase substantially, creating a greater necessity for hospitalists to diagnose, risk stratify, and manage inpatients with heart failure. Once the heart failure diagnosis has been established, determining the etiology of the decompensation and estimating the patient's risk for in‐hospital and postdischarge adverse events is essential. For patients with reduced systolic function, treatment with neurohormonal therapies, even while hospitalized, improves outcomes. Patients should be scheduled for follow‐up within 7 days after discharge to ensure clinical stability. Hospitalists should understand and adhere to the current performance measures for heart failure, as efforts tying payment to the quality of care are likely to evolve.
- ,,,.Heart failure‐related hospitalization in the U.S., 1979 to 2004.J Am Coll Cardiol.2008;52(6):428–434.
- ,,, et al.Forecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart Association.Circulation.2011;123(8):933–944.
- ,,.Rehospitalizations among patients in the Medicare Fee‐for‐Service Program.N Engl J Med.2009;360(14):1418–1428.
- 111th Congress: 2009–2010. H.R.3590: Patient Protection and Affordable Care Act. 2009. Available at: http://www.govtrack.us/congress/bill.xpd?bill=h111–3590294(15):1944–1956.
- ,,, et al.Value of clinician assessment of hemodynamics in advanced heart failure: the ESCAPE Trial.Circ: Heart Fail.2008;1(3):170–177.
- .Heart transplantation in patients with end‐stage heart failure and cardiac ascites.Circ J.2007;71(11):1744–1748.
- ,,, et al.Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure.J Am Coll Cardiol.2003;41(10):1797–1804.
- ,,, et al.Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure.JAMA.2006;296(18):2217–2226.
- ,,, et al.Hemodynamic factors associated with acute decompensated heart failure: part 1‐insights into pathophysiology.J Card Fail.2011;17(4):282–291.
- ,,, et al.State of the art: using natriuretic peptide levels in clinical practice.Eur J Heart Fail.2008;10(9):824–839.
- ,,, et al.How obesity affects the cut‐points for B‐type natriuretic peptide in the diagnosis of acute heart failure: results from the Breathing Not Properly Multinational Study.Am Heart J.2006;151(5):999–1005.
- ,,.Understanding amino‐terminal pro–B‐type natriuretic peptide in obesity.Am J Cardiol.2008;101(3):S89–S94.
- ,,, et al.B‐type natriuretic peptide and renal function in the diagnosis of heart failure: an analysis from the Breathing Not Properly Multinational Study.Am J Kidney Dis.2003;41(3):571–579.
- ,,, et al.Renal function, congestive heart failure, and amino‐terminal pro‐brain natriuretic peptide measurement.J Am Coll Cardiol.2006;47(1):91–97.
- ,,, et al.Optimal noninvasive assessment of left ventricular filling pressures: a comparison of tissue Doppler echocardiography and B‐type natriuretic peptide in patients with pulmonary artery catheters.Circulation.2004;109(20):2432–2439.
- ,,, et al.Clinical and radiologic features of pulmonary edema.Radiographics.1999;19(6):1507–1533.
- ,,,,.Clinical presentation, management, and in‐hospital outcomes of patients admitted with acute decompensated heart failure with preserved systolic function.J Am Coll Cardiol.2006;47(1):76–84.
- ,,, et al.Characteristics, treatments, and outcomes of patients with preserved systolic function hospitalized for heart failure.J Am Coll Cardiol.2007;50(8):768–777.
- ,.Systolic and diastolic heart failure: differences and similarities.J Card Fail.2007;13(7):569–576.
- Department of Health and Human Services. Medicare program; changes to the hospital inpatient prospective payment systems and fiscal year 2008 rates.Fed Reg.2007;72(162):47130–48175.
- ,,, et al.2009 focused update incorporated into the ACC/AHA 2005 guidelines for the diagnosis and management of heart failure in adults.J Am Coll Cardiol.2009;53(15):e1–e90.
- ,,, et al.Factors identified as precipitating hospital admissions for heart failure and clinical outcomes: findings from OPTIMIZE‐HF.Arch Intern Med.2008;168(8):847–854.
- ,,, et al.ADHERE Scientific Advisory Committee, Study Group, and Investigators. Risk stratification for in‐hospital mortality in acutely decompensated heart failure: classification and regression tree analysis.JAMA.2005;293(5):572–580.
- ,,, et al.Predictors of in‐hospital mortality in patients hospitalized for heart failure.J Am Coll Cardiol.2008;52(5):347–356.
- ,,,,.Admission B ‐type natriuretic peptide levels and in‐hospital mortality in acute decompensated heart failure.J Am Coll Cardiol.2007;49(19):1943–1950.
- ,,, et al.Cardiac troponin and outcome in acute heart failure.N Engl J Med.2008;358(20):2117–2126.
- ,,, et al.National Heart, Lung, and Blood Institute working group on emergency department management of acute heart failure: research challenges and opportunities.J Am Coll Cardiol.2010;56(5):343–351.
- ,,, et al.Association of consultation between generalists and cardiologists with quality and outcomes of heart failure care.Am Heart J.2003;145(6):1086–1093.
- ,,,.Congestion in acute heart failure syndromes: an essential target of evaluation and treatment.Am J Med.2006;119(12):S3–S10.
- ,,, et al.Randomised trial of high‐dose isosorbide dinitrate plus low‐dose furosemide versus high‐dose furosemide plus low‐dose isosorbide dinitrate in severe pulmonary oedema.Lancet.1998;351(9100):389–393.
- ,,,,.Furosemide absorption altered in decompensated congestive heart failure.Ann Intern Med.1985;102(3):314–318.
- ,,, et al.Diuretic strategies in patients with acute decompensated heart failure.N Engl J Med.2011;364(9):797–805.
- .Diuretic therapy.N Engl J Med.1998;339(6):387–395.
- .Diuretic resistance predicts mortality in patients with advanced heart failure.Am Heart J.2002;144(1):31–38.
- ,,,.Early ultrafiltration in patients with decompensated heart failure and diuretic resistance.J Am Coll Cardiol.2005;46(11):2047–2051.
- ,,.Relation of loop diuretic dose to mortality in advanced heart failure.Am J Cardiol.2006;97(12):1759–1764.
- ,,, et al.Worsening renal function in patients hospitalised for acute heart failure: clinical implications and prognostic significance.Eur J Heart Fail.2008;10(2):188–195.
- ,,.Combination of loop diuretics with thiazide‐type diuretics in heart failure.J Am Coll Cardiol.2010;56(19):1527–1534.
- ,,, et al.Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure.J Am Coll Cardiol.2007;49(6):675–683.
- ,,, et al.Influence of beta‐blocker continuation or withdrawal on outcomes in patients hospitalized with heart failure.J Am Coll Cardiol.2008;52(3):190–199.
- ,,, et al.Prospective evaluation of beta‐blocker use at the time of hospital discharge as a heart failure performance measure: results from OPTIMIZE‐HF.J Card Fail.2007;13(9):722–731.
- ,,, et al.Influence of pretreatment systolic blood pressure on the effect of carvedilol in patients with severe chronic heart failure.J Am Coll Cardiol.2004;43(8):1423–1429.
- ,,, et al.Predictors of mortality after discharge in patients hospitalized with heart failure: an analysis from the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF).Am Heart J.2008;156(4):662–673.
- ,,, et al.Relationship between heart failure treatment and development of worsening renal function among hospitalized patients.Am Heart J.2004;147(2):331–338.
- ,,, et al.Incidence, predictors at admission, and impact of worsening renal function among patients hospitalized with heart failure.J Am Coll Cardiol.2004;43(1):61–67.
- ,,, et al.Effects of candesartan in patients with chronic heart failure and preserved left‐ventricular ejection fraction: the CHARM‐Preserved Trial.Lancet.2003;362(9386):777–781.
- ,.Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment.Eur Heart J.2011;32(6):670–679.
- ,,, et al.Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure.Circulation.2007;116(13):1482–1487.
- ,,, et al.Statistical models and patient predictors of readmission for heart failure: a systematic review.Arch Intern Med.2008;168(13):1371–1386.
- ,,, et al.Predischarge B‐type natriuretic peptide assay for identifying patients at high risk of re‐admission after decompensated heart failure.J Am Coll Cardiol.2004;43(4):635–641.
- ,,, et al.Acute changes in N‐terminal pro‐B‐type natriuretic peptide during hospitalization and risk of readmission and mortality in patients with heart failure.Am J Cardiol.2011;107(8):1191–1195.
- ,,, et al.Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure.JAMA.2010;303(17):1716–1722.
- ,,,,.Compliance with non‐pharmacological recommendations and outcome in heart failure patients.Eur Heart J.2010;31(12):1486–1493.
- ,,, et al.Telemonitoring in patients with heart failure.N Engl J Med.2010;363(24):2301–2309.
- ,,, et al.Structured telephone support or telemonitoring programmes for patients with chronic heart failure.Cochrane Database Syst Rev.2010;(8):CD007228.
- ,.Heart failure performance measures and outcomes: real or illusory gains.JAMA.2009;302(7):792–794.
- American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI).Heart Failure Performance Measure Set. AMA‐PCPI; February 17,2011;1–85. http://www.ama‐assn.org/ama/pub/physician‐resources/physician‐consortium‐performance‐improvement.page. Accessed December 12, 2011.
- ,,, et al.ACC/AHA clinical performance measures for adults with chronic heart failure.J Am Coll Cardiol.2005;46(6):1144–1178.
- ,,, et al.Association between performance measures and clinical outcomes for patients hospitalized with heart failure.JAMA.2007;297(1):61–70.
- ,,, et al.Relationships between emerging measures of heart failure processes of care and clinical outcomes.Am Heart J.2010;159(3):406–413.
- ,,,.Heart failure‐related hospitalization in the U.S., 1979 to 2004.J Am Coll Cardiol.2008;52(6):428–434.
- ,,, et al.Forecasting the future of cardiovascular disease in the United States: a policy statement from the American Heart Association.Circulation.2011;123(8):933–944.
- ,,.Rehospitalizations among patients in the Medicare Fee‐for‐Service Program.N Engl J Med.2009;360(14):1418–1428.
- 111th Congress: 2009–2010. H.R.3590: Patient Protection and Affordable Care Act. 2009. Available at: http://www.govtrack.us/congress/bill.xpd?bill=h111–3590294(15):1944–1956.
- ,,, et al.Value of clinician assessment of hemodynamics in advanced heart failure: the ESCAPE Trial.Circ: Heart Fail.2008;1(3):170–177.
- .Heart transplantation in patients with end‐stage heart failure and cardiac ascites.Circ J.2007;71(11):1744–1748.
- ,,, et al.Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure.J Am Coll Cardiol.2003;41(10):1797–1804.
- ,,, et al.Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure.JAMA.2006;296(18):2217–2226.
- ,,, et al.Hemodynamic factors associated with acute decompensated heart failure: part 1‐insights into pathophysiology.J Card Fail.2011;17(4):282–291.
- ,,, et al.State of the art: using natriuretic peptide levels in clinical practice.Eur J Heart Fail.2008;10(9):824–839.
- ,,, et al.How obesity affects the cut‐points for B‐type natriuretic peptide in the diagnosis of acute heart failure: results from the Breathing Not Properly Multinational Study.Am Heart J.2006;151(5):999–1005.
- ,,.Understanding amino‐terminal pro–B‐type natriuretic peptide in obesity.Am J Cardiol.2008;101(3):S89–S94.
- ,,, et al.B‐type natriuretic peptide and renal function in the diagnosis of heart failure: an analysis from the Breathing Not Properly Multinational Study.Am J Kidney Dis.2003;41(3):571–579.
- ,,, et al.Renal function, congestive heart failure, and amino‐terminal pro‐brain natriuretic peptide measurement.J Am Coll Cardiol.2006;47(1):91–97.
- ,,, et al.Optimal noninvasive assessment of left ventricular filling pressures: a comparison of tissue Doppler echocardiography and B‐type natriuretic peptide in patients with pulmonary artery catheters.Circulation.2004;109(20):2432–2439.
- ,,, et al.Clinical and radiologic features of pulmonary edema.Radiographics.1999;19(6):1507–1533.
- ,,,,.Clinical presentation, management, and in‐hospital outcomes of patients admitted with acute decompensated heart failure with preserved systolic function.J Am Coll Cardiol.2006;47(1):76–84.
- ,,, et al.Characteristics, treatments, and outcomes of patients with preserved systolic function hospitalized for heart failure.J Am Coll Cardiol.2007;50(8):768–777.
- ,.Systolic and diastolic heart failure: differences and similarities.J Card Fail.2007;13(7):569–576.
- Department of Health and Human Services. Medicare program; changes to the hospital inpatient prospective payment systems and fiscal year 2008 rates.Fed Reg.2007;72(162):47130–48175.
- ,,, et al.2009 focused update incorporated into the ACC/AHA 2005 guidelines for the diagnosis and management of heart failure in adults.J Am Coll Cardiol.2009;53(15):e1–e90.
- ,,, et al.Factors identified as precipitating hospital admissions for heart failure and clinical outcomes: findings from OPTIMIZE‐HF.Arch Intern Med.2008;168(8):847–854.
- ,,, et al.ADHERE Scientific Advisory Committee, Study Group, and Investigators. Risk stratification for in‐hospital mortality in acutely decompensated heart failure: classification and regression tree analysis.JAMA.2005;293(5):572–580.
- ,,, et al.Predictors of in‐hospital mortality in patients hospitalized for heart failure.J Am Coll Cardiol.2008;52(5):347–356.
- ,,,,.Admission B ‐type natriuretic peptide levels and in‐hospital mortality in acute decompensated heart failure.J Am Coll Cardiol.2007;49(19):1943–1950.
- ,,, et al.Cardiac troponin and outcome in acute heart failure.N Engl J Med.2008;358(20):2117–2126.
- ,,, et al.National Heart, Lung, and Blood Institute working group on emergency department management of acute heart failure: research challenges and opportunities.J Am Coll Cardiol.2010;56(5):343–351.
- ,,, et al.Association of consultation between generalists and cardiologists with quality and outcomes of heart failure care.Am Heart J.2003;145(6):1086–1093.
- ,,,.Congestion in acute heart failure syndromes: an essential target of evaluation and treatment.Am J Med.2006;119(12):S3–S10.
- ,,, et al.Randomised trial of high‐dose isosorbide dinitrate plus low‐dose furosemide versus high‐dose furosemide plus low‐dose isosorbide dinitrate in severe pulmonary oedema.Lancet.1998;351(9100):389–393.
- ,,,,.Furosemide absorption altered in decompensated congestive heart failure.Ann Intern Med.1985;102(3):314–318.
- ,,, et al.Diuretic strategies in patients with acute decompensated heart failure.N Engl J Med.2011;364(9):797–805.
- .Diuretic therapy.N Engl J Med.1998;339(6):387–395.
- .Diuretic resistance predicts mortality in patients with advanced heart failure.Am Heart J.2002;144(1):31–38.
- ,,,.Early ultrafiltration in patients with decompensated heart failure and diuretic resistance.J Am Coll Cardiol.2005;46(11):2047–2051.
- ,,.Relation of loop diuretic dose to mortality in advanced heart failure.Am J Cardiol.2006;97(12):1759–1764.
- ,,, et al.Worsening renal function in patients hospitalised for acute heart failure: clinical implications and prognostic significance.Eur J Heart Fail.2008;10(2):188–195.
- ,,.Combination of loop diuretics with thiazide‐type diuretics in heart failure.J Am Coll Cardiol.2010;56(19):1527–1534.
- ,,, et al.Ultrafiltration versus intravenous diuretics for patients hospitalized for acute decompensated heart failure.J Am Coll Cardiol.2007;49(6):675–683.
- ,,, et al.Influence of beta‐blocker continuation or withdrawal on outcomes in patients hospitalized with heart failure.J Am Coll Cardiol.2008;52(3):190–199.
- ,,, et al.Prospective evaluation of beta‐blocker use at the time of hospital discharge as a heart failure performance measure: results from OPTIMIZE‐HF.J Card Fail.2007;13(9):722–731.
- ,,, et al.Influence of pretreatment systolic blood pressure on the effect of carvedilol in patients with severe chronic heart failure.J Am Coll Cardiol.2004;43(8):1423–1429.
- ,,, et al.Predictors of mortality after discharge in patients hospitalized with heart failure: an analysis from the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE‐HF).Am Heart J.2008;156(4):662–673.
- ,,, et al.Relationship between heart failure treatment and development of worsening renal function among hospitalized patients.Am Heart J.2004;147(2):331–338.
- ,,, et al.Incidence, predictors at admission, and impact of worsening renal function among patients hospitalized with heart failure.J Am Coll Cardiol.2004;43(1):61–67.
- ,,, et al.Effects of candesartan in patients with chronic heart failure and preserved left‐ventricular ejection fraction: the CHARM‐Preserved Trial.Lancet.2003;362(9386):777–781.
- ,.Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment.Eur Heart J.2011;32(6):670–679.
- ,,, et al.Influence of nonfatal hospitalization for heart failure on subsequent mortality in patients with chronic heart failure.Circulation.2007;116(13):1482–1487.
- ,,, et al.Statistical models and patient predictors of readmission for heart failure: a systematic review.Arch Intern Med.2008;168(13):1371–1386.
- ,,, et al.Predischarge B‐type natriuretic peptide assay for identifying patients at high risk of re‐admission after decompensated heart failure.J Am Coll Cardiol.2004;43(4):635–641.
- ,,, et al.Acute changes in N‐terminal pro‐B‐type natriuretic peptide during hospitalization and risk of readmission and mortality in patients with heart failure.Am J Cardiol.2011;107(8):1191–1195.
- ,,, et al.Relationship between early physician follow‐up and 30‐day readmission among Medicare beneficiaries hospitalized for heart failure.JAMA.2010;303(17):1716–1722.
- ,,,,.Compliance with non‐pharmacological recommendations and outcome in heart failure patients.Eur Heart J.2010;31(12):1486–1493.
- ,,, et al.Telemonitoring in patients with heart failure.N Engl J Med.2010;363(24):2301–2309.
- ,,, et al.Structured telephone support or telemonitoring programmes for patients with chronic heart failure.Cochrane Database Syst Rev.2010;(8):CD007228.
- ,.Heart failure performance measures and outcomes: real or illusory gains.JAMA.2009;302(7):792–794.
- American Medical Association's Physician Consortium for Performance Improvement (AMA‐PCPI).Heart Failure Performance Measure Set. AMA‐PCPI; February 17,2011;1–85. http://www.ama‐assn.org/ama/pub/physician‐resources/physician‐consortium‐performance‐improvement.page. Accessed December 12, 2011.
- ,,, et al.ACC/AHA clinical performance measures for adults with chronic heart failure.J Am Coll Cardiol.2005;46(6):1144–1178.
- ,,, et al.Association between performance measures and clinical outcomes for patients hospitalized with heart failure.JAMA.2007;297(1):61–70.
- ,,, et al.Relationships between emerging measures of heart failure processes of care and clinical outcomes.Am Heart J.2010;159(3):406–413.
Correction of CSF Protein
Traumatic lumbar puncture (LP) occurs when peripheral blood is introduced into the cerebrospinal fluid (CSF) as a result of needle trauma, which causes bleeding into the subarachnoid space. Traumatic LPs occur in up to 30% of LPs performed in children.1, 2 In addition to affecting the CSF white blood cell count, the presence of CSF red blood cells (RBCs) is associated with higher CSF protein concentrations due to the higher protein concentration in plasma compared with CSF and to the release of protein from lysed red blood cells. CSF protein concentration has been used in clinical decision rules for the prediction of bacterial meningitis in children.3 Elevated protein levels are difficult to interpret in cases of traumatic LP, and a diagnosis of bacterial meningitis may be more difficult to exclude on the basis of CSF test results.4
The interpretation of CSF protein levels is further complicated in the youngest infants due to both the changing composition of the CSF as well as the higher rates of traumatic LPs.5 Therefore, studies establishing a correction factor, adjusting observed CSF protein levels for the presence of CSF RBCs, that included predominantly older children may not be generalizable to neonates and young infants.6 We sought to determine the relationship between CSF RBC count and CSF protein in infants 56 days of age who underwent LP in the emergency department (ED).
METHODS
Study Design, Setting, and Participants
This cross‐sectional study was performed at The Children's Hospital of Philadelphia (Philadelphia, PA), an urban, tertiary care children's hospital. The Committees for the Protection of Human Subjects approved this study with a waiver of informed consent.
Infants 56 days of age and younger were eligible for inclusion if they had an LP performed as part of their ED evaluation between January 1, 2005 and July 31, 2009. At The Children's Hospital of Philadelphia, infants 56 days and younger routinely receive LPs for evaluation of fever.79 Patients undergoing LP in the ED were identified using computerized order entry records as previously described.5, 10
We excluded patients with conditions known to elevate CSF protein, including: serious bacterial infection (bacterial meningitis, urinary tract infection, bacteremia, pneumonia, septic arthritis, and bacterial gastroenteritis),11 presence of a ventricular shunt, aseptic meningitis (positive CSF enteroviral polymerase chain reaction or CSF herpes simplex virus polymerase chain reaction), congenital infections (eg, syphilis), seizure prior to presentation, and elevated bilirubin (if serum bilirubin was obtained). Due to the fact that grossly bloody CSF samples are difficult to interpret, we excluded those with a CSF RBC count >150,000 cells/mm3, a cutoff representing the 99th percentile of CSF RBC values in the cohort after applying other exclusion criteria.
Study Definitions
Bacterial meningitis was defined as either the isolation of a known bacterial pathogen from the CSF or, in patients who received antibiotics prior to evaluation, the combination of CSF pleocytosis and bacteria reported on CSF Gram stain. Bacteremia was defined as the isolation of a known bacterial pathogen from blood cultures excluding commensal skin flora. Urinary tract infection was defined as growth of a single known pathogen meeting 1 of 3 criteria: (1) 1000 colony‐forming units per mL for urine cultures obtained by suprapubic aspiration, (2) 50,000 colony‐forming units per mL from a catheterized specimen, or (3) 10,000 colony‐forming units per mL from a catheterized specimen in association with a positive urinalysis.1214
Statistical Analysis
Data analysis was performed using STATA version 12 (Stata Corp, College Station, TX). Linear regression was used to determine the association between CSF RBC and CSF protein. We analyzed the following groups of children: 1) all eligible patients; 2) children 28 days versus children >28 days; 3) vaginal versus cesarean delivery; and 4) patients without CSF pleocytosis. In the primary subanalysis, CSF pleocytosis was defined as CSF white blood cells (WBCs) >19 cells/mm3 for infants 28 days of age and CSF WBCs >9 cells/mm3 for infants 29 days of age, using reference values established by Kestenbaum et al.10 Alternate definitions of CSF pleocytosis were also examined using reference values proposed by Byington et al15 (age 28 days, >18 cells/mm3; age >29 days, >8.5 cells/mm3) and Chadwick et al16(age 0‐7 days, >26 cells/mm3; age 8‐28 days, >9 cells/mm3; age 29‐49 days, >8 cells/mm3; and age 50‐56 days, >7 cells/mm3). We did not correct CSF WBCs for the RBC count because prior studies suggest that such correction factors do not provide any advantage over uncorrected values.17 Finally, linear regression analysis was repeated while including subjects with >150,000 RBC/mm3 to determine the effect of including those patients on the association of CSF RBC count and protein concentrations. Subjects with grossly bloody CSF specimens, defined a priori as a CSF RBC >1,000,000/mm3, were excluded from this subanalysis.
RESULTS
There were 1986 infants, 56 days of age or younger, who underwent LP in the ED during the study period. Patients were excluded for the following reasons: missing medical record number (n = 16); missing CSF WBC, CSF RBC, or CSF protein values (n = 290); conditions known to elevate CSF protein concentrations (n = 426, as follows: presence of a ventricular shunt device [n = 48], serious bacterial infection [n = 149], congenital infection [n = 2], positive CSF polymerase chain reaction [PCR] test for either enterovirus or herpes simplex virus [n = 97], seizure prior to presentation [n = 98], or elevated serum bilirubin [n = 32]). An additional 13 patients with a CSF RBC count >150,000 cells/mm3 were also excluded.
For the remaining 1241 study infants, the median age was 34 days (interquartile range: 19 days‐46 days) and 554 patients (45%) were male. The median CSF RBC count was 40 cells/mm3 (interquartile range: 2‐1080 cells/mm3); 11.8% of patients had a CSF RBC count >10,000 cells/mm3.
CSF protein increased linearly with increasing CSF RBCs (Figure 1). The increase in the CSF protein concentration of 1.9 mg/dL per 1000 CSF RBCs for all patients was similar between different age groups and delivery types (Table 1). Restricting analysis to those patients without pleocytosis also yielded comparable results; applying 2 other definitions of pleocytosis did not change the magnitude of the association (Table 1).
| Patient Group | No. of Patients | Change in CSF protein (mg/dL) per 1000 RBCs (95% CI) |
|---|---|---|
| ||
| All eligible | 1241 | 1.9 (1.7‐2.1) |
| No CSF pleocytosis* | 1085 | 2.0 (1.7‐2.4) |
| Age | ||
| Age 28 days | 481 | 1.9 (1.5‐2.3) |
| Age >28 days | 760 | 1.9 (1.7‐2.1) |
| Mode of delivery | ||
| Vaginal | 741 | 1.9 (1.7‐2.2) |
| Cesarean | 366 | 1.7 (1.4‐2.0) |
In a subanalysis, we then included subjects with a CSF RBC count >150,000/mm3; one extreme outlier with a CSF RBC equal to 3,160,000/mm3 remained excluded. Inclusion of more traumatic samples lessened the overall correction factor. The CSF protein increased by 1.22 mg/dL (95% confidence interval: 1.14‐1.29 mg/dL) per 1000 RBC/mm3 increase in the CSF. In the subset without CSF pleocytosis, the CSF protein increased by 1.44 mg/dL (95% confidence interval: 1.33‐1.57 mg/dL) per 1000 RBC/mm3.
Three children had high CSF protein values (>500 mg/dL) despite the relative paucity of CSF RBCs. Two of these infants had respiratory syncytial virus bronchiolitis; neither infant had signs or symptoms of neurological illness. While details of the labor and delivery were not available, the CSF sample for one of these infants was reported to have xanthochromia, and the other infant was reported to have had a traumatic LP with a CSF sample that subsequently cleared. The third infant had fever without a specific source identified, but had a birth history of vaginal delivery and prolonged labor. The CSF sample from LP for this patient was reported as grossly bloody by the performing clinicians and by the Clinical Microbiology Laboratory, despite a CSF red blood cell count of only 5500 cells/mm3.
DISCUSSION
In a large cohort of infants 56 days of age, CSF protein increased by approximately 2 mg/dL for every 1000 cell/mm3 increase in CSF RBCs. This correction factor is higher than previously reported correction factors from studies including older infants and children.6, 18 Some of this difference may be explained by the presence of old blood related to the trauma of labor and delivery. Previous work has demonstrated that the presence of xanthochromia, another RBC breakdown product, in the CSF of young infants was associated with maternal labor and elevated CSF protein.19 Consistent with this hypothesis, the correction factor was nominally higher in those infants born by vaginal delivery compared with those born by cesarean section.
Several infants in our study had high CSF protein levels despite a paucity of CSF RBCs. By convention at our institution, the protein and glucose values are determined from the second tube, and the WBCs and RBCs are determined from the third tube. However, we could not determine the order in which the specimens for protein and RBCs were collected for individual specimens. Additionally, it is possible that delayed clearance of blood from a traumatic LP would cause the CSF protein level to be high, as measured in the second tube, but lead to few RBCs in the third tube. These circumstances could explain the discrepancy between CSF protein and CSF RBCs counts for some patients.
The CSF protein adjustment factor for infants 56 days of age in our study was almost twice the correction of 1.1 mg/dL for every 1000 RBC increase reported by Nigrovic et al among infants 90 days of age.6 There are differences in the design of the 2 studies. We excluded subjects with exceedingly large numbers of CSF RBCs and restricted inclusion to those 56 days of age or younger. When subjects with >150,000 RBCs/mm3 were included, the correction decreased to a value comparable to that reported by Nigrovic et al.6 Therefore, it is possible that inclusion of subjects with grossly bloody specimens in prior studies skewed the association between CSF protein and CSF RBCs. The number of subjects in our cohort with >150,000 CSF RBCs was too small to calculate a relevant correction factor for infants with exceedingly high CSF RBC counts.
The results of this study should be considered in the context of several limitations. Details regarding labor and delivery were not available. We suspect that old blood related to the trauma of birth provides partial explanation for the higher correction factor in neonates and young infants compared with older children. However, differences in CSF blood‐brain barrier permeability may also contribute to these differences, independent of the CSF RBC count. Additionally, though the study population included a large number of neonates and young infants, a relatively small proportion of subjects had high CSF RBC counts. Therefore, our results may not be generalizable to those with exceedingly high CSF RBCs. Finally, available clinical prediction rules to identify patients with CSF pleocytosis, who are at very low risk for bacterial meningitis, include CSF protein as a predictor.3, 20, 21 Although CSF protein in children with traumatic LPs may need adjustment prior to application of the clinical prediction rule, further study is needed before implementing this approach.
In conclusion, we found that CSF protein concentrations increased by approximately 2 mg/dL for every 1000 CSF RBCs. Correction of CSF protein for those with extremely high CSF RBCs may not be appropriate, as conventional linear models do not apply. These data may assist clinicians in interpreting CSF protein concentrations in infants 56 days of age and younger in the context of traumatic LPs.
- ,,,,,.Local anesthetic and stylet styles: factors associated with resident lumbar puncture success.Pediatrics.2006;117:876–881.
- ,,.Risk factors for traumatic or unsuccessful lumbar punctures in children.Ann Emerg Med.2007;49:762–771.
- ,,, et al.Clinical prediction rule for identifying children with cerebrospinal fluid pleocytosis at very low risk of bacterial meningitis.JAMA.2007;297:52–60.
- ,,.Interpretation of traumatic lumbar punctures: who can go home?Pediatrics.2003;111:525–528.
- ,,,,.Age‐specific reference values for cerebrospinal fluid protein concentration in neonates and young infants.J Hosp Med.2011;6:22–27.
- ,,.Correction of cerebrospinal fluid protein for the presence of red blood cells in children with a traumatic lumbar puncture.J Pediatr.2011;159:158–159.
- ,,.Failure of infant observation scales in detecting serious illness in febrile, 4‐ to 8‐week‐old infants.Pediatrics.1990;85:1040–1043.
- ,.Unpredictability of serious bacterial illness in febrile infants from birth to 1 month of age.Arch Pediatr Adolesc Med.1999;153:508–511.
- ,,.Outpatient management without antibiotics of fever in selected infants.N Engl J Med.1993;329:1437–1441.
- ,,,,.Defining cerebrospinal fluid white blood cell count reference values in neonates and young infants.Pediatrics.2010;125:257–264.
- ,,,,.Sterile cerebrospinal fluid pleocytosis in young infants with urinary tract infections.J Pediatr.2008;153:290–292.
- ,,, et al.Clinical and demographic factors associated with urinary tract infection in young febrile infants.Pediatrics.2005;116:644–648.
- ,,,,.Prevalence of urinary tract infection in febrile young children in the emergency department.Pediatrics.1998;102:e16.
- ,,,,,.Prevalence of urinary tract infection in febrile infants.J Pediatr.1993;123:17–23.
- ,,.Normative cerebrospinal fluid profiles in febrile infants.J Pediatr.2011;158:130–134.
- ,,,.Cerebrospinal fluid characteristics of infants who present to the emergency department with fever: establishing normal values by week of age.Pediatr Infect Dis J.2011;30:e63–e67.
- ,.Corrections for leukocytes and percent of neutrophils do not match observations in blood‐contaminated cerebrospinal fluid and have no value over uncorrected cells for diagnosis.Pediatr Infect Dis J.2006;25:8–11.
- ,,,,.Distinguishing cerebrospinal fluid abnormalities in children with bacterial meningitis and traumatic lumbar puncture.J Infect Dis.1990;162:251–254.
- ,,,.Cerebrospinal fluid xanthochromia in newborns is related to maternal labor before delivery.Pediatrics.2007;120:e1212–e1216.
- ,.Accuracy and test characteristics of ancillary tests of cerebrospinal fluid for predicting acute bacterial meningitis in children with low white blood cell counts in cerebrospinal fluid.Acad Emerg Med.2005;12:303–309.
- ,,,.A decision rule for predicting bacterial meningitis in children with cerebrospinal fluid pleocytosis when gram stain is negative or unavailable.Acad Emerg Med.2008;15:437–444.
Traumatic lumbar puncture (LP) occurs when peripheral blood is introduced into the cerebrospinal fluid (CSF) as a result of needle trauma, which causes bleeding into the subarachnoid space. Traumatic LPs occur in up to 30% of LPs performed in children.1, 2 In addition to affecting the CSF white blood cell count, the presence of CSF red blood cells (RBCs) is associated with higher CSF protein concentrations due to the higher protein concentration in plasma compared with CSF and to the release of protein from lysed red blood cells. CSF protein concentration has been used in clinical decision rules for the prediction of bacterial meningitis in children.3 Elevated protein levels are difficult to interpret in cases of traumatic LP, and a diagnosis of bacterial meningitis may be more difficult to exclude on the basis of CSF test results.4
The interpretation of CSF protein levels is further complicated in the youngest infants due to both the changing composition of the CSF as well as the higher rates of traumatic LPs.5 Therefore, studies establishing a correction factor, adjusting observed CSF protein levels for the presence of CSF RBCs, that included predominantly older children may not be generalizable to neonates and young infants.6 We sought to determine the relationship between CSF RBC count and CSF protein in infants 56 days of age who underwent LP in the emergency department (ED).
METHODS
Study Design, Setting, and Participants
This cross‐sectional study was performed at The Children's Hospital of Philadelphia (Philadelphia, PA), an urban, tertiary care children's hospital. The Committees for the Protection of Human Subjects approved this study with a waiver of informed consent.
Infants 56 days of age and younger were eligible for inclusion if they had an LP performed as part of their ED evaluation between January 1, 2005 and July 31, 2009. At The Children's Hospital of Philadelphia, infants 56 days and younger routinely receive LPs for evaluation of fever.79 Patients undergoing LP in the ED were identified using computerized order entry records as previously described.5, 10
We excluded patients with conditions known to elevate CSF protein, including: serious bacterial infection (bacterial meningitis, urinary tract infection, bacteremia, pneumonia, septic arthritis, and bacterial gastroenteritis),11 presence of a ventricular shunt, aseptic meningitis (positive CSF enteroviral polymerase chain reaction or CSF herpes simplex virus polymerase chain reaction), congenital infections (eg, syphilis), seizure prior to presentation, and elevated bilirubin (if serum bilirubin was obtained). Due to the fact that grossly bloody CSF samples are difficult to interpret, we excluded those with a CSF RBC count >150,000 cells/mm3, a cutoff representing the 99th percentile of CSF RBC values in the cohort after applying other exclusion criteria.
Study Definitions
Bacterial meningitis was defined as either the isolation of a known bacterial pathogen from the CSF or, in patients who received antibiotics prior to evaluation, the combination of CSF pleocytosis and bacteria reported on CSF Gram stain. Bacteremia was defined as the isolation of a known bacterial pathogen from blood cultures excluding commensal skin flora. Urinary tract infection was defined as growth of a single known pathogen meeting 1 of 3 criteria: (1) 1000 colony‐forming units per mL for urine cultures obtained by suprapubic aspiration, (2) 50,000 colony‐forming units per mL from a catheterized specimen, or (3) 10,000 colony‐forming units per mL from a catheterized specimen in association with a positive urinalysis.1214
Statistical Analysis
Data analysis was performed using STATA version 12 (Stata Corp, College Station, TX). Linear regression was used to determine the association between CSF RBC and CSF protein. We analyzed the following groups of children: 1) all eligible patients; 2) children 28 days versus children >28 days; 3) vaginal versus cesarean delivery; and 4) patients without CSF pleocytosis. In the primary subanalysis, CSF pleocytosis was defined as CSF white blood cells (WBCs) >19 cells/mm3 for infants 28 days of age and CSF WBCs >9 cells/mm3 for infants 29 days of age, using reference values established by Kestenbaum et al.10 Alternate definitions of CSF pleocytosis were also examined using reference values proposed by Byington et al15 (age 28 days, >18 cells/mm3; age >29 days, >8.5 cells/mm3) and Chadwick et al16(age 0‐7 days, >26 cells/mm3; age 8‐28 days, >9 cells/mm3; age 29‐49 days, >8 cells/mm3; and age 50‐56 days, >7 cells/mm3). We did not correct CSF WBCs for the RBC count because prior studies suggest that such correction factors do not provide any advantage over uncorrected values.17 Finally, linear regression analysis was repeated while including subjects with >150,000 RBC/mm3 to determine the effect of including those patients on the association of CSF RBC count and protein concentrations. Subjects with grossly bloody CSF specimens, defined a priori as a CSF RBC >1,000,000/mm3, were excluded from this subanalysis.
RESULTS
There were 1986 infants, 56 days of age or younger, who underwent LP in the ED during the study period. Patients were excluded for the following reasons: missing medical record number (n = 16); missing CSF WBC, CSF RBC, or CSF protein values (n = 290); conditions known to elevate CSF protein concentrations (n = 426, as follows: presence of a ventricular shunt device [n = 48], serious bacterial infection [n = 149], congenital infection [n = 2], positive CSF polymerase chain reaction [PCR] test for either enterovirus or herpes simplex virus [n = 97], seizure prior to presentation [n = 98], or elevated serum bilirubin [n = 32]). An additional 13 patients with a CSF RBC count >150,000 cells/mm3 were also excluded.
For the remaining 1241 study infants, the median age was 34 days (interquartile range: 19 days‐46 days) and 554 patients (45%) were male. The median CSF RBC count was 40 cells/mm3 (interquartile range: 2‐1080 cells/mm3); 11.8% of patients had a CSF RBC count >10,000 cells/mm3.
CSF protein increased linearly with increasing CSF RBCs (Figure 1). The increase in the CSF protein concentration of 1.9 mg/dL per 1000 CSF RBCs for all patients was similar between different age groups and delivery types (Table 1). Restricting analysis to those patients without pleocytosis also yielded comparable results; applying 2 other definitions of pleocytosis did not change the magnitude of the association (Table 1).

| Patient Group | No. of Patients | Change in CSF protein (mg/dL) per 1000 RBCs (95% CI) |
|---|---|---|
| ||
| All eligible | 1241 | 1.9 (1.7‐2.1) |
| No CSF pleocytosis* | 1085 | 2.0 (1.7‐2.4) |
| Age | ||
| Age 28 days | 481 | 1.9 (1.5‐2.3) |
| Age >28 days | 760 | 1.9 (1.7‐2.1) |
| Mode of delivery | ||
| Vaginal | 741 | 1.9 (1.7‐2.2) |
| Cesarean | 366 | 1.7 (1.4‐2.0) |
In a subanalysis, we then included subjects with a CSF RBC count >150,000/mm3; one extreme outlier with a CSF RBC equal to 3,160,000/mm3 remained excluded. Inclusion of more traumatic samples lessened the overall correction factor. The CSF protein increased by 1.22 mg/dL (95% confidence interval: 1.14‐1.29 mg/dL) per 1000 RBC/mm3 increase in the CSF. In the subset without CSF pleocytosis, the CSF protein increased by 1.44 mg/dL (95% confidence interval: 1.33‐1.57 mg/dL) per 1000 RBC/mm3.
Three children had high CSF protein values (>500 mg/dL) despite the relative paucity of CSF RBCs. Two of these infants had respiratory syncytial virus bronchiolitis; neither infant had signs or symptoms of neurological illness. While details of the labor and delivery were not available, the CSF sample for one of these infants was reported to have xanthochromia, and the other infant was reported to have had a traumatic LP with a CSF sample that subsequently cleared. The third infant had fever without a specific source identified, but had a birth history of vaginal delivery and prolonged labor. The CSF sample from LP for this patient was reported as grossly bloody by the performing clinicians and by the Clinical Microbiology Laboratory, despite a CSF red blood cell count of only 5500 cells/mm3.
DISCUSSION
In a large cohort of infants 56 days of age, CSF protein increased by approximately 2 mg/dL for every 1000 cell/mm3 increase in CSF RBCs. This correction factor is higher than previously reported correction factors from studies including older infants and children.6, 18 Some of this difference may be explained by the presence of old blood related to the trauma of labor and delivery. Previous work has demonstrated that the presence of xanthochromia, another RBC breakdown product, in the CSF of young infants was associated with maternal labor and elevated CSF protein.19 Consistent with this hypothesis, the correction factor was nominally higher in those infants born by vaginal delivery compared with those born by cesarean section.
Several infants in our study had high CSF protein levels despite a paucity of CSF RBCs. By convention at our institution, the protein and glucose values are determined from the second tube, and the WBCs and RBCs are determined from the third tube. However, we could not determine the order in which the specimens for protein and RBCs were collected for individual specimens. Additionally, it is possible that delayed clearance of blood from a traumatic LP would cause the CSF protein level to be high, as measured in the second tube, but lead to few RBCs in the third tube. These circumstances could explain the discrepancy between CSF protein and CSF RBCs counts for some patients.
The CSF protein adjustment factor for infants 56 days of age in our study was almost twice the correction of 1.1 mg/dL for every 1000 RBC increase reported by Nigrovic et al among infants 90 days of age.6 There are differences in the design of the 2 studies. We excluded subjects with exceedingly large numbers of CSF RBCs and restricted inclusion to those 56 days of age or younger. When subjects with >150,000 RBCs/mm3 were included, the correction decreased to a value comparable to that reported by Nigrovic et al.6 Therefore, it is possible that inclusion of subjects with grossly bloody specimens in prior studies skewed the association between CSF protein and CSF RBCs. The number of subjects in our cohort with >150,000 CSF RBCs was too small to calculate a relevant correction factor for infants with exceedingly high CSF RBC counts.
The results of this study should be considered in the context of several limitations. Details regarding labor and delivery were not available. We suspect that old blood related to the trauma of birth provides partial explanation for the higher correction factor in neonates and young infants compared with older children. However, differences in CSF blood‐brain barrier permeability may also contribute to these differences, independent of the CSF RBC count. Additionally, though the study population included a large number of neonates and young infants, a relatively small proportion of subjects had high CSF RBC counts. Therefore, our results may not be generalizable to those with exceedingly high CSF RBCs. Finally, available clinical prediction rules to identify patients with CSF pleocytosis, who are at very low risk for bacterial meningitis, include CSF protein as a predictor.3, 20, 21 Although CSF protein in children with traumatic LPs may need adjustment prior to application of the clinical prediction rule, further study is needed before implementing this approach.
In conclusion, we found that CSF protein concentrations increased by approximately 2 mg/dL for every 1000 CSF RBCs. Correction of CSF protein for those with extremely high CSF RBCs may not be appropriate, as conventional linear models do not apply. These data may assist clinicians in interpreting CSF protein concentrations in infants 56 days of age and younger in the context of traumatic LPs.
Traumatic lumbar puncture (LP) occurs when peripheral blood is introduced into the cerebrospinal fluid (CSF) as a result of needle trauma, which causes bleeding into the subarachnoid space. Traumatic LPs occur in up to 30% of LPs performed in children.1, 2 In addition to affecting the CSF white blood cell count, the presence of CSF red blood cells (RBCs) is associated with higher CSF protein concentrations due to the higher protein concentration in plasma compared with CSF and to the release of protein from lysed red blood cells. CSF protein concentration has been used in clinical decision rules for the prediction of bacterial meningitis in children.3 Elevated protein levels are difficult to interpret in cases of traumatic LP, and a diagnosis of bacterial meningitis may be more difficult to exclude on the basis of CSF test results.4
The interpretation of CSF protein levels is further complicated in the youngest infants due to both the changing composition of the CSF as well as the higher rates of traumatic LPs.5 Therefore, studies establishing a correction factor, adjusting observed CSF protein levels for the presence of CSF RBCs, that included predominantly older children may not be generalizable to neonates and young infants.6 We sought to determine the relationship between CSF RBC count and CSF protein in infants 56 days of age who underwent LP in the emergency department (ED).
METHODS
Study Design, Setting, and Participants
This cross‐sectional study was performed at The Children's Hospital of Philadelphia (Philadelphia, PA), an urban, tertiary care children's hospital. The Committees for the Protection of Human Subjects approved this study with a waiver of informed consent.
Infants 56 days of age and younger were eligible for inclusion if they had an LP performed as part of their ED evaluation between January 1, 2005 and July 31, 2009. At The Children's Hospital of Philadelphia, infants 56 days and younger routinely receive LPs for evaluation of fever.79 Patients undergoing LP in the ED were identified using computerized order entry records as previously described.5, 10
We excluded patients with conditions known to elevate CSF protein, including: serious bacterial infection (bacterial meningitis, urinary tract infection, bacteremia, pneumonia, septic arthritis, and bacterial gastroenteritis),11 presence of a ventricular shunt, aseptic meningitis (positive CSF enteroviral polymerase chain reaction or CSF herpes simplex virus polymerase chain reaction), congenital infections (eg, syphilis), seizure prior to presentation, and elevated bilirubin (if serum bilirubin was obtained). Due to the fact that grossly bloody CSF samples are difficult to interpret, we excluded those with a CSF RBC count >150,000 cells/mm3, a cutoff representing the 99th percentile of CSF RBC values in the cohort after applying other exclusion criteria.
Study Definitions
Bacterial meningitis was defined as either the isolation of a known bacterial pathogen from the CSF or, in patients who received antibiotics prior to evaluation, the combination of CSF pleocytosis and bacteria reported on CSF Gram stain. Bacteremia was defined as the isolation of a known bacterial pathogen from blood cultures excluding commensal skin flora. Urinary tract infection was defined as growth of a single known pathogen meeting 1 of 3 criteria: (1) 1000 colony‐forming units per mL for urine cultures obtained by suprapubic aspiration, (2) 50,000 colony‐forming units per mL from a catheterized specimen, or (3) 10,000 colony‐forming units per mL from a catheterized specimen in association with a positive urinalysis.1214
Statistical Analysis
Data analysis was performed using STATA version 12 (Stata Corp, College Station, TX). Linear regression was used to determine the association between CSF RBC and CSF protein. We analyzed the following groups of children: 1) all eligible patients; 2) children 28 days versus children >28 days; 3) vaginal versus cesarean delivery; and 4) patients without CSF pleocytosis. In the primary subanalysis, CSF pleocytosis was defined as CSF white blood cells (WBCs) >19 cells/mm3 for infants 28 days of age and CSF WBCs >9 cells/mm3 for infants 29 days of age, using reference values established by Kestenbaum et al.10 Alternate definitions of CSF pleocytosis were also examined using reference values proposed by Byington et al15 (age 28 days, >18 cells/mm3; age >29 days, >8.5 cells/mm3) and Chadwick et al16(age 0‐7 days, >26 cells/mm3; age 8‐28 days, >9 cells/mm3; age 29‐49 days, >8 cells/mm3; and age 50‐56 days, >7 cells/mm3). We did not correct CSF WBCs for the RBC count because prior studies suggest that such correction factors do not provide any advantage over uncorrected values.17 Finally, linear regression analysis was repeated while including subjects with >150,000 RBC/mm3 to determine the effect of including those patients on the association of CSF RBC count and protein concentrations. Subjects with grossly bloody CSF specimens, defined a priori as a CSF RBC >1,000,000/mm3, were excluded from this subanalysis.
RESULTS
There were 1986 infants, 56 days of age or younger, who underwent LP in the ED during the study period. Patients were excluded for the following reasons: missing medical record number (n = 16); missing CSF WBC, CSF RBC, or CSF protein values (n = 290); conditions known to elevate CSF protein concentrations (n = 426, as follows: presence of a ventricular shunt device [n = 48], serious bacterial infection [n = 149], congenital infection [n = 2], positive CSF polymerase chain reaction [PCR] test for either enterovirus or herpes simplex virus [n = 97], seizure prior to presentation [n = 98], or elevated serum bilirubin [n = 32]). An additional 13 patients with a CSF RBC count >150,000 cells/mm3 were also excluded.
For the remaining 1241 study infants, the median age was 34 days (interquartile range: 19 days‐46 days) and 554 patients (45%) were male. The median CSF RBC count was 40 cells/mm3 (interquartile range: 2‐1080 cells/mm3); 11.8% of patients had a CSF RBC count >10,000 cells/mm3.
CSF protein increased linearly with increasing CSF RBCs (Figure 1). The increase in the CSF protein concentration of 1.9 mg/dL per 1000 CSF RBCs for all patients was similar between different age groups and delivery types (Table 1). Restricting analysis to those patients without pleocytosis also yielded comparable results; applying 2 other definitions of pleocytosis did not change the magnitude of the association (Table 1).

| Patient Group | No. of Patients | Change in CSF protein (mg/dL) per 1000 RBCs (95% CI) |
|---|---|---|
| ||
| All eligible | 1241 | 1.9 (1.7‐2.1) |
| No CSF pleocytosis* | 1085 | 2.0 (1.7‐2.4) |
| Age | ||
| Age 28 days | 481 | 1.9 (1.5‐2.3) |
| Age >28 days | 760 | 1.9 (1.7‐2.1) |
| Mode of delivery | ||
| Vaginal | 741 | 1.9 (1.7‐2.2) |
| Cesarean | 366 | 1.7 (1.4‐2.0) |
In a subanalysis, we then included subjects with a CSF RBC count >150,000/mm3; one extreme outlier with a CSF RBC equal to 3,160,000/mm3 remained excluded. Inclusion of more traumatic samples lessened the overall correction factor. The CSF protein increased by 1.22 mg/dL (95% confidence interval: 1.14‐1.29 mg/dL) per 1000 RBC/mm3 increase in the CSF. In the subset without CSF pleocytosis, the CSF protein increased by 1.44 mg/dL (95% confidence interval: 1.33‐1.57 mg/dL) per 1000 RBC/mm3.
Three children had high CSF protein values (>500 mg/dL) despite the relative paucity of CSF RBCs. Two of these infants had respiratory syncytial virus bronchiolitis; neither infant had signs or symptoms of neurological illness. While details of the labor and delivery were not available, the CSF sample for one of these infants was reported to have xanthochromia, and the other infant was reported to have had a traumatic LP with a CSF sample that subsequently cleared. The third infant had fever without a specific source identified, but had a birth history of vaginal delivery and prolonged labor. The CSF sample from LP for this patient was reported as grossly bloody by the performing clinicians and by the Clinical Microbiology Laboratory, despite a CSF red blood cell count of only 5500 cells/mm3.
DISCUSSION
In a large cohort of infants 56 days of age, CSF protein increased by approximately 2 mg/dL for every 1000 cell/mm3 increase in CSF RBCs. This correction factor is higher than previously reported correction factors from studies including older infants and children.6, 18 Some of this difference may be explained by the presence of old blood related to the trauma of labor and delivery. Previous work has demonstrated that the presence of xanthochromia, another RBC breakdown product, in the CSF of young infants was associated with maternal labor and elevated CSF protein.19 Consistent with this hypothesis, the correction factor was nominally higher in those infants born by vaginal delivery compared with those born by cesarean section.
Several infants in our study had high CSF protein levels despite a paucity of CSF RBCs. By convention at our institution, the protein and glucose values are determined from the second tube, and the WBCs and RBCs are determined from the third tube. However, we could not determine the order in which the specimens for protein and RBCs were collected for individual specimens. Additionally, it is possible that delayed clearance of blood from a traumatic LP would cause the CSF protein level to be high, as measured in the second tube, but lead to few RBCs in the third tube. These circumstances could explain the discrepancy between CSF protein and CSF RBCs counts for some patients.
The CSF protein adjustment factor for infants 56 days of age in our study was almost twice the correction of 1.1 mg/dL for every 1000 RBC increase reported by Nigrovic et al among infants 90 days of age.6 There are differences in the design of the 2 studies. We excluded subjects with exceedingly large numbers of CSF RBCs and restricted inclusion to those 56 days of age or younger. When subjects with >150,000 RBCs/mm3 were included, the correction decreased to a value comparable to that reported by Nigrovic et al.6 Therefore, it is possible that inclusion of subjects with grossly bloody specimens in prior studies skewed the association between CSF protein and CSF RBCs. The number of subjects in our cohort with >150,000 CSF RBCs was too small to calculate a relevant correction factor for infants with exceedingly high CSF RBC counts.
The results of this study should be considered in the context of several limitations. Details regarding labor and delivery were not available. We suspect that old blood related to the trauma of birth provides partial explanation for the higher correction factor in neonates and young infants compared with older children. However, differences in CSF blood‐brain barrier permeability may also contribute to these differences, independent of the CSF RBC count. Additionally, though the study population included a large number of neonates and young infants, a relatively small proportion of subjects had high CSF RBC counts. Therefore, our results may not be generalizable to those with exceedingly high CSF RBCs. Finally, available clinical prediction rules to identify patients with CSF pleocytosis, who are at very low risk for bacterial meningitis, include CSF protein as a predictor.3, 20, 21 Although CSF protein in children with traumatic LPs may need adjustment prior to application of the clinical prediction rule, further study is needed before implementing this approach.
In conclusion, we found that CSF protein concentrations increased by approximately 2 mg/dL for every 1000 CSF RBCs. Correction of CSF protein for those with extremely high CSF RBCs may not be appropriate, as conventional linear models do not apply. These data may assist clinicians in interpreting CSF protein concentrations in infants 56 days of age and younger in the context of traumatic LPs.
- ,,,,,.Local anesthetic and stylet styles: factors associated with resident lumbar puncture success.Pediatrics.2006;117:876–881.
- ,,.Risk factors for traumatic or unsuccessful lumbar punctures in children.Ann Emerg Med.2007;49:762–771.
- ,,, et al.Clinical prediction rule for identifying children with cerebrospinal fluid pleocytosis at very low risk of bacterial meningitis.JAMA.2007;297:52–60.
- ,,.Interpretation of traumatic lumbar punctures: who can go home?Pediatrics.2003;111:525–528.
- ,,,,.Age‐specific reference values for cerebrospinal fluid protein concentration in neonates and young infants.J Hosp Med.2011;6:22–27.
- ,,.Correction of cerebrospinal fluid protein for the presence of red blood cells in children with a traumatic lumbar puncture.J Pediatr.2011;159:158–159.
- ,,.Failure of infant observation scales in detecting serious illness in febrile, 4‐ to 8‐week‐old infants.Pediatrics.1990;85:1040–1043.
- ,.Unpredictability of serious bacterial illness in febrile infants from birth to 1 month of age.Arch Pediatr Adolesc Med.1999;153:508–511.
- ,,.Outpatient management without antibiotics of fever in selected infants.N Engl J Med.1993;329:1437–1441.
- ,,,,.Defining cerebrospinal fluid white blood cell count reference values in neonates and young infants.Pediatrics.2010;125:257–264.
- ,,,,.Sterile cerebrospinal fluid pleocytosis in young infants with urinary tract infections.J Pediatr.2008;153:290–292.
- ,,, et al.Clinical and demographic factors associated with urinary tract infection in young febrile infants.Pediatrics.2005;116:644–648.
- ,,,,.Prevalence of urinary tract infection in febrile young children in the emergency department.Pediatrics.1998;102:e16.
- ,,,,,.Prevalence of urinary tract infection in febrile infants.J Pediatr.1993;123:17–23.
- ,,.Normative cerebrospinal fluid profiles in febrile infants.J Pediatr.2011;158:130–134.
- ,,,.Cerebrospinal fluid characteristics of infants who present to the emergency department with fever: establishing normal values by week of age.Pediatr Infect Dis J.2011;30:e63–e67.
- ,.Corrections for leukocytes and percent of neutrophils do not match observations in blood‐contaminated cerebrospinal fluid and have no value over uncorrected cells for diagnosis.Pediatr Infect Dis J.2006;25:8–11.
- ,,,,.Distinguishing cerebrospinal fluid abnormalities in children with bacterial meningitis and traumatic lumbar puncture.J Infect Dis.1990;162:251–254.
- ,,,.Cerebrospinal fluid xanthochromia in newborns is related to maternal labor before delivery.Pediatrics.2007;120:e1212–e1216.
- ,.Accuracy and test characteristics of ancillary tests of cerebrospinal fluid for predicting acute bacterial meningitis in children with low white blood cell counts in cerebrospinal fluid.Acad Emerg Med.2005;12:303–309.
- ,,,.A decision rule for predicting bacterial meningitis in children with cerebrospinal fluid pleocytosis when gram stain is negative or unavailable.Acad Emerg Med.2008;15:437–444.
- ,,,,,.Local anesthetic and stylet styles: factors associated with resident lumbar puncture success.Pediatrics.2006;117:876–881.
- ,,.Risk factors for traumatic or unsuccessful lumbar punctures in children.Ann Emerg Med.2007;49:762–771.
- ,,, et al.Clinical prediction rule for identifying children with cerebrospinal fluid pleocytosis at very low risk of bacterial meningitis.JAMA.2007;297:52–60.
- ,,.Interpretation of traumatic lumbar punctures: who can go home?Pediatrics.2003;111:525–528.
- ,,,,.Age‐specific reference values for cerebrospinal fluid protein concentration in neonates and young infants.J Hosp Med.2011;6:22–27.
- ,,.Correction of cerebrospinal fluid protein for the presence of red blood cells in children with a traumatic lumbar puncture.J Pediatr.2011;159:158–159.
- ,,.Failure of infant observation scales in detecting serious illness in febrile, 4‐ to 8‐week‐old infants.Pediatrics.1990;85:1040–1043.
- ,.Unpredictability of serious bacterial illness in febrile infants from birth to 1 month of age.Arch Pediatr Adolesc Med.1999;153:508–511.
- ,,.Outpatient management without antibiotics of fever in selected infants.N Engl J Med.1993;329:1437–1441.
- ,,,,.Defining cerebrospinal fluid white blood cell count reference values in neonates and young infants.Pediatrics.2010;125:257–264.
- ,,,,.Sterile cerebrospinal fluid pleocytosis in young infants with urinary tract infections.J Pediatr.2008;153:290–292.
- ,,, et al.Clinical and demographic factors associated with urinary tract infection in young febrile infants.Pediatrics.2005;116:644–648.
- ,,,,.Prevalence of urinary tract infection in febrile young children in the emergency department.Pediatrics.1998;102:e16.
- ,,,,,.Prevalence of urinary tract infection in febrile infants.J Pediatr.1993;123:17–23.
- ,,.Normative cerebrospinal fluid profiles in febrile infants.J Pediatr.2011;158:130–134.
- ,,,.Cerebrospinal fluid characteristics of infants who present to the emergency department with fever: establishing normal values by week of age.Pediatr Infect Dis J.2011;30:e63–e67.
- ,.Corrections for leukocytes and percent of neutrophils do not match observations in blood‐contaminated cerebrospinal fluid and have no value over uncorrected cells for diagnosis.Pediatr Infect Dis J.2006;25:8–11.
- ,,,,.Distinguishing cerebrospinal fluid abnormalities in children with bacterial meningitis and traumatic lumbar puncture.J Infect Dis.1990;162:251–254.
- ,,,.Cerebrospinal fluid xanthochromia in newborns is related to maternal labor before delivery.Pediatrics.2007;120:e1212–e1216.
- ,.Accuracy and test characteristics of ancillary tests of cerebrospinal fluid for predicting acute bacterial meningitis in children with low white blood cell counts in cerebrospinal fluid.Acad Emerg Med.2005;12:303–309.
- ,,,.A decision rule for predicting bacterial meningitis in children with cerebrospinal fluid pleocytosis when gram stain is negative or unavailable.Acad Emerg Med.2008;15:437–444.
Copyright © 2012 Society of Hospital Medicine
Family Support Groups: An Integral Part of Patient Care
When Ms. A. visited with her son’s psychiatrist, she broke into tears: "I don’t know what to do! My son hates us! He calls us ‘messengers of the devil!’ His sister is getting teased at school. Her classmates won’t talk to her, and they say her family is "really messed up." No one understands our problems, and I feel so alone. I am too ashamed to confide in any of our family friends. Our friends’ kids all seem to be doing so well. What should I do?"
Fortunately, the psychiatrist was aware of the NAMI Family-to-Family Education Program, which provided her with a free, 12-week course for family caregivers of individuals with severe mental illnesses. Family members who had been trained to run the program taught the course. It afforded Ms. A. the opportunity to learn current information about the biology of schizophrenia, medication, and strategies for medication adherence. She also got a chance to meet others who had experienced the maelstrom that mental illness can have on families. From another mother in the group, Ms. A. learned strategies aimed at managing her son when he became angry.
More importantly, however, she learned not to blame herself and to take time to look after her own health and well-being. She got advice about how to help her daughter manage the teasing at school. In addition, she took educational material into the school and gave it to her daughter’s homeroom teacher.
Families of individuals who suffer from mental illness experience a myriad of emotions: anger, frustration, hopelessness, sadness, fear, anxiety, shame, and loss. Psychiatrists must maintain a nonjudgmental and empathic stance with patients and their families.
In addition, we must be mindful of the profound influence mental illness has on the family system. Relatives might become psychologically distressed from the burden of caretaking and the social stigma of mental illness (J. Fam. Psychol. 2001;15:225-40). Recognizing that caregivers and families are often under stress and might have depression or anxiety must be a paramount goal of the clinician (J. Affect. Disord. 2010;121:10-21). Caregiver burden itself impairs the support that the caregiver can provide (Expressed Emotion in Families: Its Significance for Mental Illness, New York: The Guilford Press, 1985).
Psychiatrists are in the position to identify resources in the community that can help families build a knowledge base, which can be a tool for families to assist their loved ones and themselves. Support groups empower families to advocate for their loved ones, can alleviate the burdens of caregiving, and give family members a sense of community (Family Relations 1999;48:405-10).
The websites below contain helpful information, including support groups for families dealing with the mental illness of a loved one. We encourage clinicians to recommend these sites to families.
Many institutions provide patients and family education, so local or state resources can be of additional help. Please let us know about any websites that are not on our list that you would recommend to family members.
The following websites might help patients cope with family members who have mental illness:
P National Family Caregivers Association
P American Foundation for Suicide Prevention
P Families for Depression Awareness
P Depression and Bipolar Support Alliance
P The Children’s Society’s Include Project
P The National Alliance on Mental Illness Family-to-Family Education Program
P Support and Education Program for Families
P Children and Adults with Attention Deficit/Hyperactivity Disorder
P Substance Abuse and Mental Health Services Administration Resources for Military Families
P Compeer
P National PLAN (Planned Lifetime Assistance Network) Alliance
Dr. Heru is an associate professor of psychiatry at the University of Colorado at Denver, Aurora. She has been a member of the Association of Family Psychiatrists since 2002 and currently serves as the organization’s treasurer. In addition, she is the coauthor of two books on working with families and is the author of numerous articles on this topic. Dr. Ascher is a resident in psychiatry, department of psychiatry and behavioral sciences at Beth Israel Medical Center, New York. He is the Dear Abby Fellow, Group for the Advancement of Psychiatry (GAP) Family Committee. This commentary appears in Clinical Psychiatry News, a publication of Elsevier.
When Ms. A. visited with her son’s psychiatrist, she broke into tears: "I don’t know what to do! My son hates us! He calls us ‘messengers of the devil!’ His sister is getting teased at school. Her classmates won’t talk to her, and they say her family is "really messed up." No one understands our problems, and I feel so alone. I am too ashamed to confide in any of our family friends. Our friends’ kids all seem to be doing so well. What should I do?"
Fortunately, the psychiatrist was aware of the NAMI Family-to-Family Education Program, which provided her with a free, 12-week course for family caregivers of individuals with severe mental illnesses. Family members who had been trained to run the program taught the course. It afforded Ms. A. the opportunity to learn current information about the biology of schizophrenia, medication, and strategies for medication adherence. She also got a chance to meet others who had experienced the maelstrom that mental illness can have on families. From another mother in the group, Ms. A. learned strategies aimed at managing her son when he became angry.
More importantly, however, she learned not to blame herself and to take time to look after her own health and well-being. She got advice about how to help her daughter manage the teasing at school. In addition, she took educational material into the school and gave it to her daughter’s homeroom teacher.
Families of individuals who suffer from mental illness experience a myriad of emotions: anger, frustration, hopelessness, sadness, fear, anxiety, shame, and loss. Psychiatrists must maintain a nonjudgmental and empathic stance with patients and their families.
In addition, we must be mindful of the profound influence mental illness has on the family system. Relatives might become psychologically distressed from the burden of caretaking and the social stigma of mental illness (J. Fam. Psychol. 2001;15:225-40). Recognizing that caregivers and families are often under stress and might have depression or anxiety must be a paramount goal of the clinician (J. Affect. Disord. 2010;121:10-21). Caregiver burden itself impairs the support that the caregiver can provide (Expressed Emotion in Families: Its Significance for Mental Illness, New York: The Guilford Press, 1985).
Psychiatrists are in the position to identify resources in the community that can help families build a knowledge base, which can be a tool for families to assist their loved ones and themselves. Support groups empower families to advocate for their loved ones, can alleviate the burdens of caregiving, and give family members a sense of community (Family Relations 1999;48:405-10).
The websites below contain helpful information, including support groups for families dealing with the mental illness of a loved one. We encourage clinicians to recommend these sites to families.
Many institutions provide patients and family education, so local or state resources can be of additional help. Please let us know about any websites that are not on our list that you would recommend to family members.
The following websites might help patients cope with family members who have mental illness:
P National Family Caregivers Association
P American Foundation for Suicide Prevention
P Families for Depression Awareness
P Depression and Bipolar Support Alliance
P The Children’s Society’s Include Project
P The National Alliance on Mental Illness Family-to-Family Education Program
P Support and Education Program for Families
P Children and Adults with Attention Deficit/Hyperactivity Disorder
P Substance Abuse and Mental Health Services Administration Resources for Military Families
P Compeer
P National PLAN (Planned Lifetime Assistance Network) Alliance
Dr. Heru is an associate professor of psychiatry at the University of Colorado at Denver, Aurora. She has been a member of the Association of Family Psychiatrists since 2002 and currently serves as the organization’s treasurer. In addition, she is the coauthor of two books on working with families and is the author of numerous articles on this topic. Dr. Ascher is a resident in psychiatry, department of psychiatry and behavioral sciences at Beth Israel Medical Center, New York. He is the Dear Abby Fellow, Group for the Advancement of Psychiatry (GAP) Family Committee. This commentary appears in Clinical Psychiatry News, a publication of Elsevier.
When Ms. A. visited with her son’s psychiatrist, she broke into tears: "I don’t know what to do! My son hates us! He calls us ‘messengers of the devil!’ His sister is getting teased at school. Her classmates won’t talk to her, and they say her family is "really messed up." No one understands our problems, and I feel so alone. I am too ashamed to confide in any of our family friends. Our friends’ kids all seem to be doing so well. What should I do?"
Fortunately, the psychiatrist was aware of the NAMI Family-to-Family Education Program, which provided her with a free, 12-week course for family caregivers of individuals with severe mental illnesses. Family members who had been trained to run the program taught the course. It afforded Ms. A. the opportunity to learn current information about the biology of schizophrenia, medication, and strategies for medication adherence. She also got a chance to meet others who had experienced the maelstrom that mental illness can have on families. From another mother in the group, Ms. A. learned strategies aimed at managing her son when he became angry.
More importantly, however, she learned not to blame herself and to take time to look after her own health and well-being. She got advice about how to help her daughter manage the teasing at school. In addition, she took educational material into the school and gave it to her daughter’s homeroom teacher.
Families of individuals who suffer from mental illness experience a myriad of emotions: anger, frustration, hopelessness, sadness, fear, anxiety, shame, and loss. Psychiatrists must maintain a nonjudgmental and empathic stance with patients and their families.
In addition, we must be mindful of the profound influence mental illness has on the family system. Relatives might become psychologically distressed from the burden of caretaking and the social stigma of mental illness (J. Fam. Psychol. 2001;15:225-40). Recognizing that caregivers and families are often under stress and might have depression or anxiety must be a paramount goal of the clinician (J. Affect. Disord. 2010;121:10-21). Caregiver burden itself impairs the support that the caregiver can provide (Expressed Emotion in Families: Its Significance for Mental Illness, New York: The Guilford Press, 1985).
Psychiatrists are in the position to identify resources in the community that can help families build a knowledge base, which can be a tool for families to assist their loved ones and themselves. Support groups empower families to advocate for their loved ones, can alleviate the burdens of caregiving, and give family members a sense of community (Family Relations 1999;48:405-10).
The websites below contain helpful information, including support groups for families dealing with the mental illness of a loved one. We encourage clinicians to recommend these sites to families.
Many institutions provide patients and family education, so local or state resources can be of additional help. Please let us know about any websites that are not on our list that you would recommend to family members.
The following websites might help patients cope with family members who have mental illness:
P National Family Caregivers Association
P American Foundation for Suicide Prevention
P Families for Depression Awareness
P Depression and Bipolar Support Alliance
P The Children’s Society’s Include Project
P The National Alliance on Mental Illness Family-to-Family Education Program
P Support and Education Program for Families
P Children and Adults with Attention Deficit/Hyperactivity Disorder
P Substance Abuse and Mental Health Services Administration Resources for Military Families
P Compeer
P National PLAN (Planned Lifetime Assistance Network) Alliance
Dr. Heru is an associate professor of psychiatry at the University of Colorado at Denver, Aurora. She has been a member of the Association of Family Psychiatrists since 2002 and currently serves as the organization’s treasurer. In addition, she is the coauthor of two books on working with families and is the author of numerous articles on this topic. Dr. Ascher is a resident in psychiatry, department of psychiatry and behavioral sciences at Beth Israel Medical Center, New York. He is the Dear Abby Fellow, Group for the Advancement of Psychiatry (GAP) Family Committee. This commentary appears in Clinical Psychiatry News, a publication of Elsevier.
FDA Panel Endorses Obesity Drug Qnexa After All
SILVER SPRING, MD. – The combination formulation of phentermine and topiramate should be approved as a weight-loss treatment, with a risk-management plan that addresses the teratogenic effects of topiramate and a postmarketing study that evaluates cardiovascular outcomes associated with treatment, the majority of a Food and Drug Administration panel agreed at a meeting on Feb. 22.
The FDA’s Endocrinologic and Metabolic Drugs Advisory Committee voted 20 to 2 that the benefit-risk profile of the phentermine-topiramate combination supported its approval for the treatment of obesity in people with a body mass index (BMI) of at least 30 kg/m2, or those with a BMI of at least 27 kg/m2 who also have weight-related comorbidities. The manufacturer, Vivus, has proposed that the combination product – in three fixed-dose combinations – be approved for this population, in combination with diet and exercise.
If approved, this will be the first new obesity drug treatment approved in 13 years and will be marketed as Qnexa by Vivus. The combination product contains an immediate-release formulation of phentermine, which is a sympathomimetic amine approved for short-term weight loss, on the market in the United States since 1959; and a controlled-release formulation of topiramate, an antiepileptic drug approved for treating epilepsy in 1996, for migraine prophylaxis in 2004, and for pediatric epilepsy in 2011. Qnexa is manufactured in three fixed-dose combinations: the starting low dose of 3.75 mg of phentermine and 23 mg of topiramate, the recommended dose of 7.5 mg/46 mg, and the highest dose (15 mg/92 mg) for patients not reaching their weight-loss goal.
The two separate components are available at higher doses than those contained in the combination product.
At a meeting in July 2010, the same panel had agreed that the same product had been shown to be effective as a weight-loss agent, compared with placebo in two 1-year, pivotal studies in this patient population, but the majority voted against recommending approval of the agent because of concerns over the risk-benefit profile, particularly the potential for teratogenicity and increases in heart rate associated with treatment.
The FDA advised the company in October 2010 that the cardiovascular risks and teratogenic potential associated with treatment had not been adequately assessed, and requested that the company provide evidence that an increase in heart rate (a mean of 1.6 beats/minute at the highest dose) did not increase the risk for major adverse cardiovascular events, further evaluate the potential risk for oral clefts associated with prenatal exposure to the topiramate component, provide 2-year data, and develop a Risk Evaluation and Mitigation Strategy (REMS).
Components of the REMS as now planned include a patient medication guide explaining the risk of oral clefts (cleft lip with or without cleft palate) associated with first trimester exposure in studies and pregnancy registries, a certified pharmacy network that dispenses a month’s supply at a time via mail order, and a plan to educate prescribers about the teratogenic risk and train them to prescribe the drug appropriately. The product would be a category X drug, contraindicated during pregnancy, with the recommendation to immediately stop taking the drug if a woman becomes pregnant during treatment.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The combination formulation of phentermine and topiramate should be approved as a weight-loss treatment, with a risk-management plan that addresses the teratogenic effects of topiramate and a postmarketing study that evaluates cardiovascular outcomes associated with treatment, the majority of a Food and Drug Administration panel agreed at a meeting on Feb. 22.
The FDA’s Endocrinologic and Metabolic Drugs Advisory Committee voted 20 to 2 that the benefit-risk profile of the phentermine-topiramate combination supported its approval for the treatment of obesity in people with a body mass index (BMI) of at least 30 kg/m2, or those with a BMI of at least 27 kg/m2 who also have weight-related comorbidities. The manufacturer, Vivus, has proposed that the combination product – in three fixed-dose combinations – be approved for this population, in combination with diet and exercise.
If approved, this will be the first new obesity drug treatment approved in 13 years and will be marketed as Qnexa by Vivus. The combination product contains an immediate-release formulation of phentermine, which is a sympathomimetic amine approved for short-term weight loss, on the market in the United States since 1959; and a controlled-release formulation of topiramate, an antiepileptic drug approved for treating epilepsy in 1996, for migraine prophylaxis in 2004, and for pediatric epilepsy in 2011. Qnexa is manufactured in three fixed-dose combinations: the starting low dose of 3.75 mg of phentermine and 23 mg of topiramate, the recommended dose of 7.5 mg/46 mg, and the highest dose (15 mg/92 mg) for patients not reaching their weight-loss goal.
The two separate components are available at higher doses than those contained in the combination product.
At a meeting in July 2010, the same panel had agreed that the same product had been shown to be effective as a weight-loss agent, compared with placebo in two 1-year, pivotal studies in this patient population, but the majority voted against recommending approval of the agent because of concerns over the risk-benefit profile, particularly the potential for teratogenicity and increases in heart rate associated with treatment.
The FDA advised the company in October 2010 that the cardiovascular risks and teratogenic potential associated with treatment had not been adequately assessed, and requested that the company provide evidence that an increase in heart rate (a mean of 1.6 beats/minute at the highest dose) did not increase the risk for major adverse cardiovascular events, further evaluate the potential risk for oral clefts associated with prenatal exposure to the topiramate component, provide 2-year data, and develop a Risk Evaluation and Mitigation Strategy (REMS).
Components of the REMS as now planned include a patient medication guide explaining the risk of oral clefts (cleft lip with or without cleft palate) associated with first trimester exposure in studies and pregnancy registries, a certified pharmacy network that dispenses a month’s supply at a time via mail order, and a plan to educate prescribers about the teratogenic risk and train them to prescribe the drug appropriately. The product would be a category X drug, contraindicated during pregnancy, with the recommendation to immediately stop taking the drug if a woman becomes pregnant during treatment.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The combination formulation of phentermine and topiramate should be approved as a weight-loss treatment, with a risk-management plan that addresses the teratogenic effects of topiramate and a postmarketing study that evaluates cardiovascular outcomes associated with treatment, the majority of a Food and Drug Administration panel agreed at a meeting on Feb. 22.
The FDA’s Endocrinologic and Metabolic Drugs Advisory Committee voted 20 to 2 that the benefit-risk profile of the phentermine-topiramate combination supported its approval for the treatment of obesity in people with a body mass index (BMI) of at least 30 kg/m2, or those with a BMI of at least 27 kg/m2 who also have weight-related comorbidities. The manufacturer, Vivus, has proposed that the combination product – in three fixed-dose combinations – be approved for this population, in combination with diet and exercise.
If approved, this will be the first new obesity drug treatment approved in 13 years and will be marketed as Qnexa by Vivus. The combination product contains an immediate-release formulation of phentermine, which is a sympathomimetic amine approved for short-term weight loss, on the market in the United States since 1959; and a controlled-release formulation of topiramate, an antiepileptic drug approved for treating epilepsy in 1996, for migraine prophylaxis in 2004, and for pediatric epilepsy in 2011. Qnexa is manufactured in three fixed-dose combinations: the starting low dose of 3.75 mg of phentermine and 23 mg of topiramate, the recommended dose of 7.5 mg/46 mg, and the highest dose (15 mg/92 mg) for patients not reaching their weight-loss goal.
The two separate components are available at higher doses than those contained in the combination product.
At a meeting in July 2010, the same panel had agreed that the same product had been shown to be effective as a weight-loss agent, compared with placebo in two 1-year, pivotal studies in this patient population, but the majority voted against recommending approval of the agent because of concerns over the risk-benefit profile, particularly the potential for teratogenicity and increases in heart rate associated with treatment.
The FDA advised the company in October 2010 that the cardiovascular risks and teratogenic potential associated with treatment had not been adequately assessed, and requested that the company provide evidence that an increase in heart rate (a mean of 1.6 beats/minute at the highest dose) did not increase the risk for major adverse cardiovascular events, further evaluate the potential risk for oral clefts associated with prenatal exposure to the topiramate component, provide 2-year data, and develop a Risk Evaluation and Mitigation Strategy (REMS).
Components of the REMS as now planned include a patient medication guide explaining the risk of oral clefts (cleft lip with or without cleft palate) associated with first trimester exposure in studies and pregnancy registries, a certified pharmacy network that dispenses a month’s supply at a time via mail order, and a plan to educate prescribers about the teratogenic risk and train them to prescribe the drug appropriately. The product would be a category X drug, contraindicated during pregnancy, with the recommendation to immediately stop taking the drug if a woman becomes pregnant during treatment.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
FROM A MEETING OF THE FDA'S ENDOCRINOLOGIC AND METABOLIC DRUGS ADVISORY COMMITTEE
Her Chief Complaint Is ... And by the Way She’s Also Pregnant
We emergency physicians are generally a confident bunch. But in the time it takes to slip on a peel and hit the pavement (a bananosecond), some of us ratchet up adrenaline output when we pick up a chart and notice a history like 22 yo F, minor MVC, c/o headache and back pain, 32 weeks pregnant.
From whence comes this anxiety? A bit may stem from reading about those seven-figure lawsuit verdicts for pregnancy-related malpractice cases. However, tied to this are those questions and comments I often hear from residents seeking assurance, even when they know the answers.
Can I get this x-ray?
Is it OK to give her morphine IV? Should I start with 1 mg? (Sure, if it’s in the right acupuncture point.)
Wow, I’m so used to not treating asymptomatic elevated BP that I almost forgot to address it for this pregnant patient.
Getting answers from specialists can often be frustrating. The OB doc may be uncomfortable with the non-OB aspects of the case, while the other consulting specialists may be uncomfortable applying their expertise in the context of pregnancy.
I recall asking a surgeon to look at a third-trimester patient with likely appendicitis and an equivocal ultrasound. His plan related to me was, "We’ll sit on it overnight." After making some remark about his own application of procto-tocin, I suggested an MRI. He was a bit leery, but with some education and pressure on our radiologist to do our hospital’s first MRI to rule out appendicitis (accomplished without procedural sedation on that radiologist), we identified an acute appy.
As with many aspects of EM, it may be up to the EP to coordinate optimal care in these situations. In 1981, Dr. Arnold Greensher and I developed a system called Prenatal Care – A Systems Approach to help OBs and primary care physicians integrate prenatal care within a comprehensive risk management system. It includes frequently updated information on managing nonobstetric illness and injury in this population. The system’s development was coordinated with a panel of well-regarded academic specialists, including a group of perinatologists.
The track record for the system has been quite surprising to us, as well as to the medical malpractice insurers who purchased the system for their docs: There were more than 1.5 million deliveries during this time period with only 8 malpractice claims. The expected number of claims would be 400-700. For a large number of users, premium rates went down dramatically during a time when national rates were going in the opposite direction.
Over the past year, I’ve contributed two well-received articles for the Focus On series in ACEP News: Trauma in the Obstetric Patient in July 2010 and Perinatal Disaster Management in September 2011 (both can be found at www.acep.org/focuson). I was honored to be invited by the publication’s editorial panel to provide a quarterly column that focuses on unique aspects of emergency care of the pregnant patient. The goal of this column will be to provide practical recommendations for the EP on common presenting problems in this population. I will often have coauthors, including specialists in that topic, as well as perinatologist input. One of our residents will be an integral part of this group. Our column is not intended to be a standard of care, but rather a sound, easy-to-use package of recommendations that would be considered one avenue for providing optimal care.
Each article will have a clinical tool – a summary that can stand alone for easy reference. In fact, our Trauma Table is posted in a number of EDs that I have visited. As ACEP News technology progresses, we hope to have these as a library with the tables hyperlinked to the specific didactic parts of the articles.
In this issue, we debut our first article, Stroke in Pregnancy (pp. XX-XX). This will provide a nice supplement to any stroke protocols at your hospital. Later in 2012, we plan to have one on sepsis and another on cardiac emergencies, including acute coronary syndromes.
I look forward to sharing this column with you.
Dr. Roemer is an Associate Professor in the Department of Emergency Medicine, Oklahoma University School of Community Medicine, Tulsa.
We emergency physicians are generally a confident bunch. But in the time it takes to slip on a peel and hit the pavement (a bananosecond), some of us ratchet up adrenaline output when we pick up a chart and notice a history like 22 yo F, minor MVC, c/o headache and back pain, 32 weeks pregnant.
From whence comes this anxiety? A bit may stem from reading about those seven-figure lawsuit verdicts for pregnancy-related malpractice cases. However, tied to this are those questions and comments I often hear from residents seeking assurance, even when they know the answers.
Can I get this x-ray?
Is it OK to give her morphine IV? Should I start with 1 mg? (Sure, if it’s in the right acupuncture point.)
Wow, I’m so used to not treating asymptomatic elevated BP that I almost forgot to address it for this pregnant patient.
Getting answers from specialists can often be frustrating. The OB doc may be uncomfortable with the non-OB aspects of the case, while the other consulting specialists may be uncomfortable applying their expertise in the context of pregnancy.
I recall asking a surgeon to look at a third-trimester patient with likely appendicitis and an equivocal ultrasound. His plan related to me was, "We’ll sit on it overnight." After making some remark about his own application of procto-tocin, I suggested an MRI. He was a bit leery, but with some education and pressure on our radiologist to do our hospital’s first MRI to rule out appendicitis (accomplished without procedural sedation on that radiologist), we identified an acute appy.
As with many aspects of EM, it may be up to the EP to coordinate optimal care in these situations. In 1981, Dr. Arnold Greensher and I developed a system called Prenatal Care – A Systems Approach to help OBs and primary care physicians integrate prenatal care within a comprehensive risk management system. It includes frequently updated information on managing nonobstetric illness and injury in this population. The system’s development was coordinated with a panel of well-regarded academic specialists, including a group of perinatologists.
The track record for the system has been quite surprising to us, as well as to the medical malpractice insurers who purchased the system for their docs: There were more than 1.5 million deliveries during this time period with only 8 malpractice claims. The expected number of claims would be 400-700. For a large number of users, premium rates went down dramatically during a time when national rates were going in the opposite direction.
Over the past year, I’ve contributed two well-received articles for the Focus On series in ACEP News: Trauma in the Obstetric Patient in July 2010 and Perinatal Disaster Management in September 2011 (both can be found at www.acep.org/focuson). I was honored to be invited by the publication’s editorial panel to provide a quarterly column that focuses on unique aspects of emergency care of the pregnant patient. The goal of this column will be to provide practical recommendations for the EP on common presenting problems in this population. I will often have coauthors, including specialists in that topic, as well as perinatologist input. One of our residents will be an integral part of this group. Our column is not intended to be a standard of care, but rather a sound, easy-to-use package of recommendations that would be considered one avenue for providing optimal care.
Each article will have a clinical tool – a summary that can stand alone for easy reference. In fact, our Trauma Table is posted in a number of EDs that I have visited. As ACEP News technology progresses, we hope to have these as a library with the tables hyperlinked to the specific didactic parts of the articles.
In this issue, we debut our first article, Stroke in Pregnancy (pp. XX-XX). This will provide a nice supplement to any stroke protocols at your hospital. Later in 2012, we plan to have one on sepsis and another on cardiac emergencies, including acute coronary syndromes.
I look forward to sharing this column with you.
Dr. Roemer is an Associate Professor in the Department of Emergency Medicine, Oklahoma University School of Community Medicine, Tulsa.
We emergency physicians are generally a confident bunch. But in the time it takes to slip on a peel and hit the pavement (a bananosecond), some of us ratchet up adrenaline output when we pick up a chart and notice a history like 22 yo F, minor MVC, c/o headache and back pain, 32 weeks pregnant.
From whence comes this anxiety? A bit may stem from reading about those seven-figure lawsuit verdicts for pregnancy-related malpractice cases. However, tied to this are those questions and comments I often hear from residents seeking assurance, even when they know the answers.
Can I get this x-ray?
Is it OK to give her morphine IV? Should I start with 1 mg? (Sure, if it’s in the right acupuncture point.)
Wow, I’m so used to not treating asymptomatic elevated BP that I almost forgot to address it for this pregnant patient.
Getting answers from specialists can often be frustrating. The OB doc may be uncomfortable with the non-OB aspects of the case, while the other consulting specialists may be uncomfortable applying their expertise in the context of pregnancy.
I recall asking a surgeon to look at a third-trimester patient with likely appendicitis and an equivocal ultrasound. His plan related to me was, "We’ll sit on it overnight." After making some remark about his own application of procto-tocin, I suggested an MRI. He was a bit leery, but with some education and pressure on our radiologist to do our hospital’s first MRI to rule out appendicitis (accomplished without procedural sedation on that radiologist), we identified an acute appy.
As with many aspects of EM, it may be up to the EP to coordinate optimal care in these situations. In 1981, Dr. Arnold Greensher and I developed a system called Prenatal Care – A Systems Approach to help OBs and primary care physicians integrate prenatal care within a comprehensive risk management system. It includes frequently updated information on managing nonobstetric illness and injury in this population. The system’s development was coordinated with a panel of well-regarded academic specialists, including a group of perinatologists.
The track record for the system has been quite surprising to us, as well as to the medical malpractice insurers who purchased the system for their docs: There were more than 1.5 million deliveries during this time period with only 8 malpractice claims. The expected number of claims would be 400-700. For a large number of users, premium rates went down dramatically during a time when national rates were going in the opposite direction.
Over the past year, I’ve contributed two well-received articles for the Focus On series in ACEP News: Trauma in the Obstetric Patient in July 2010 and Perinatal Disaster Management in September 2011 (both can be found at www.acep.org/focuson). I was honored to be invited by the publication’s editorial panel to provide a quarterly column that focuses on unique aspects of emergency care of the pregnant patient. The goal of this column will be to provide practical recommendations for the EP on common presenting problems in this population. I will often have coauthors, including specialists in that topic, as well as perinatologist input. One of our residents will be an integral part of this group. Our column is not intended to be a standard of care, but rather a sound, easy-to-use package of recommendations that would be considered one avenue for providing optimal care.
Each article will have a clinical tool – a summary that can stand alone for easy reference. In fact, our Trauma Table is posted in a number of EDs that I have visited. As ACEP News technology progresses, we hope to have these as a library with the tables hyperlinked to the specific didactic parts of the articles.
In this issue, we debut our first article, Stroke in Pregnancy (pp. XX-XX). This will provide a nice supplement to any stroke protocols at your hospital. Later in 2012, we plan to have one on sepsis and another on cardiac emergencies, including acute coronary syndromes.
I look forward to sharing this column with you.
Dr. Roemer is an Associate Professor in the Department of Emergency Medicine, Oklahoma University School of Community Medicine, Tulsa.
EMA recommends conditional approval of lymphoma drug
The European Medicines Agency (EMA) has recommended that pixantrone dimaleate (Pixuvri) be granted conditional approval to treat non-Hodgkin B-cell lymphoma.
The approval is for pixantrone as single-agent therapy for patients with relapsed or refractory lymphoma.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended conditional approval of pixantrone because the data are not yet comprehensive. The CHMP has said more information is needed on the benefits of pixantrone in patients who received prior rituximab.
At the same time, the CHMP concluded that pixantrone satisfies an unmet medical need because there are no approved and standard treatments for this stage of the disease. Therefore, the benefits of making this medicine available on the market immediately outweigh the risks inherent in the fact that additional data are required.
The conditional approval of pixantrone will be renewed on a yearly basis until the obligation to provide additional data on rituximab-pretreated patients has been fulfilled. The applicant, CTI Life Sciences Ltd., has said it plans to provide the data by mid-2015.
The main study of pixantrone, the phase 3 EXTEND PIX301 trial, compared the drug to other chemotherapeutic agents in patients with relapsed or refractory non-Hodgkin lymphoma. The rate of response was 20% in the pixantrone arm and 6% in the comparator arm.
In addition, patients receiving pixantrone had longer progression-free survival than patients in the comparator group, with a median of 10.2 months and 7.6 months, respectively.
However, the CHMP noted that the benefit of pixantrone appeared to be lower in patients who had received prior rituximab. And a benefit was not established in patients who had not responded to their last treatment and received pixantrone as the fifth or later round of chemotherapy.
The most common side effects observed with pixantrone were neutropenia, leukopenia, anemia, thrombocytopenia, asthenia, pyrexia, cough, decreased ejection fraction, and nausea. The most common grade 3 and 4 adverse events were hematologic.
The CHMP’s recommendation for conditional approval has been sent to the European Commission for the adoption of a European Union-wide decision. For more information on pixantrone’s approval, visit the EMA website. ![]()
The European Medicines Agency (EMA) has recommended that pixantrone dimaleate (Pixuvri) be granted conditional approval to treat non-Hodgkin B-cell lymphoma.
The approval is for pixantrone as single-agent therapy for patients with relapsed or refractory lymphoma.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended conditional approval of pixantrone because the data are not yet comprehensive. The CHMP has said more information is needed on the benefits of pixantrone in patients who received prior rituximab.
At the same time, the CHMP concluded that pixantrone satisfies an unmet medical need because there are no approved and standard treatments for this stage of the disease. Therefore, the benefits of making this medicine available on the market immediately outweigh the risks inherent in the fact that additional data are required.
The conditional approval of pixantrone will be renewed on a yearly basis until the obligation to provide additional data on rituximab-pretreated patients has been fulfilled. The applicant, CTI Life Sciences Ltd., has said it plans to provide the data by mid-2015.
The main study of pixantrone, the phase 3 EXTEND PIX301 trial, compared the drug to other chemotherapeutic agents in patients with relapsed or refractory non-Hodgkin lymphoma. The rate of response was 20% in the pixantrone arm and 6% in the comparator arm.
In addition, patients receiving pixantrone had longer progression-free survival than patients in the comparator group, with a median of 10.2 months and 7.6 months, respectively.
However, the CHMP noted that the benefit of pixantrone appeared to be lower in patients who had received prior rituximab. And a benefit was not established in patients who had not responded to their last treatment and received pixantrone as the fifth or later round of chemotherapy.
The most common side effects observed with pixantrone were neutropenia, leukopenia, anemia, thrombocytopenia, asthenia, pyrexia, cough, decreased ejection fraction, and nausea. The most common grade 3 and 4 adverse events were hematologic.
The CHMP’s recommendation for conditional approval has been sent to the European Commission for the adoption of a European Union-wide decision. For more information on pixantrone’s approval, visit the EMA website. ![]()
The European Medicines Agency (EMA) has recommended that pixantrone dimaleate (Pixuvri) be granted conditional approval to treat non-Hodgkin B-cell lymphoma.
The approval is for pixantrone as single-agent therapy for patients with relapsed or refractory lymphoma.
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended conditional approval of pixantrone because the data are not yet comprehensive. The CHMP has said more information is needed on the benefits of pixantrone in patients who received prior rituximab.
At the same time, the CHMP concluded that pixantrone satisfies an unmet medical need because there are no approved and standard treatments for this stage of the disease. Therefore, the benefits of making this medicine available on the market immediately outweigh the risks inherent in the fact that additional data are required.
The conditional approval of pixantrone will be renewed on a yearly basis until the obligation to provide additional data on rituximab-pretreated patients has been fulfilled. The applicant, CTI Life Sciences Ltd., has said it plans to provide the data by mid-2015.
The main study of pixantrone, the phase 3 EXTEND PIX301 trial, compared the drug to other chemotherapeutic agents in patients with relapsed or refractory non-Hodgkin lymphoma. The rate of response was 20% in the pixantrone arm and 6% in the comparator arm.
In addition, patients receiving pixantrone had longer progression-free survival than patients in the comparator group, with a median of 10.2 months and 7.6 months, respectively.
However, the CHMP noted that the benefit of pixantrone appeared to be lower in patients who had received prior rituximab. And a benefit was not established in patients who had not responded to their last treatment and received pixantrone as the fifth or later round of chemotherapy.
The most common side effects observed with pixantrone were neutropenia, leukopenia, anemia, thrombocytopenia, asthenia, pyrexia, cough, decreased ejection fraction, and nausea. The most common grade 3 and 4 adverse events were hematologic.
The CHMP’s recommendation for conditional approval has been sent to the European Commission for the adoption of a European Union-wide decision. For more information on pixantrone’s approval, visit the EMA website. ![]()
HHS Pushes Back ICD-10 Deadline
Hospitalists keeping an eye on the planned implementation of the 10th revision of the International Statistical Classification of Diseases coding system (ICD-10) can breathe a temporary sigh of relief: The U.S. Department of Health and Human Services (HHS) announced last week that it would delay the October 2013 start date for using the new codes. No new date was given.
The decision came after the American Medical Association (AMA) launched a public campaign to persuade Congress and HHS to delay the transition to ICD-10. SHM's AMA delegate and public policy committee member Bradley Flansbaum, DO, MPH, SFHM, says SHM took no formal position on the start date but was watching the national discussion closely.
"This is a big jump," says Dr. Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York City. "As always with administrative procedures and the legacy systems of yesteryear, a lot of institutions are pushing back."
At issue, according to AMA leaders, is that physicians already are dealing with a litany of regulatory, technological, and coding changes tied to the national healthcare reform movement. The immediate implementation quintuples the number of billing codes to 68,000, an expansion that would be an "onslaught of overlapping regulatory mandates and reporting requirements," wrote James Madara, MD, AMA executive vice president and chief executive officer, in a letter this month to HHS Secretary Kathleen Sebilius (PDF).
"ICD-10 codes are important to many positive improvements in our healthcare system," Sebilius said in announcing the delay. "We have heard from many in the provider community who have concerns about the administrative burdens they face in the years ahead. We are committing to work with the provider community to re-examine the pace at which HHS and the nation implement these important improvements to our healthcare system."
Hospitalists keeping an eye on the planned implementation of the 10th revision of the International Statistical Classification of Diseases coding system (ICD-10) can breathe a temporary sigh of relief: The U.S. Department of Health and Human Services (HHS) announced last week that it would delay the October 2013 start date for using the new codes. No new date was given.
The decision came after the American Medical Association (AMA) launched a public campaign to persuade Congress and HHS to delay the transition to ICD-10. SHM's AMA delegate and public policy committee member Bradley Flansbaum, DO, MPH, SFHM, says SHM took no formal position on the start date but was watching the national discussion closely.
"This is a big jump," says Dr. Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York City. "As always with administrative procedures and the legacy systems of yesteryear, a lot of institutions are pushing back."
At issue, according to AMA leaders, is that physicians already are dealing with a litany of regulatory, technological, and coding changes tied to the national healthcare reform movement. The immediate implementation quintuples the number of billing codes to 68,000, an expansion that would be an "onslaught of overlapping regulatory mandates and reporting requirements," wrote James Madara, MD, AMA executive vice president and chief executive officer, in a letter this month to HHS Secretary Kathleen Sebilius (PDF).
"ICD-10 codes are important to many positive improvements in our healthcare system," Sebilius said in announcing the delay. "We have heard from many in the provider community who have concerns about the administrative burdens they face in the years ahead. We are committing to work with the provider community to re-examine the pace at which HHS and the nation implement these important improvements to our healthcare system."
Hospitalists keeping an eye on the planned implementation of the 10th revision of the International Statistical Classification of Diseases coding system (ICD-10) can breathe a temporary sigh of relief: The U.S. Department of Health and Human Services (HHS) announced last week that it would delay the October 2013 start date for using the new codes. No new date was given.
The decision came after the American Medical Association (AMA) launched a public campaign to persuade Congress and HHS to delay the transition to ICD-10. SHM's AMA delegate and public policy committee member Bradley Flansbaum, DO, MPH, SFHM, says SHM took no formal position on the start date but was watching the national discussion closely.
"This is a big jump," says Dr. Flansbaum, director of hospitalist services at Lenox Hill Hospital in New York City. "As always with administrative procedures and the legacy systems of yesteryear, a lot of institutions are pushing back."
At issue, according to AMA leaders, is that physicians already are dealing with a litany of regulatory, technological, and coding changes tied to the national healthcare reform movement. The immediate implementation quintuples the number of billing codes to 68,000, an expansion that would be an "onslaught of overlapping regulatory mandates and reporting requirements," wrote James Madara, MD, AMA executive vice president and chief executive officer, in a letter this month to HHS Secretary Kathleen Sebilius (PDF).
"ICD-10 codes are important to many positive improvements in our healthcare system," Sebilius said in announcing the delay. "We have heard from many in the provider community who have concerns about the administrative burdens they face in the years ahead. We are committing to work with the provider community to re-examine the pace at which HHS and the nation implement these important improvements to our healthcare system."
In the Literature: Research You Need to Know
Clinical question: In patients undergoing coronary and peripheral angiography, does acetylcysteine before and after the procedure protect the kidneys?
Background: Contrast-induced acute kidney injury is a serious complication of procedures that use iodinated contrast material and can lead to the need for dialysis, prolonged hospital stay, and increased cost and mortality. Acetylcysteine is thought to prevent this, but previous results from more than 40 trials conflict regarding its effectiveness.
Study design: Double-blinded randomized trial.
Setting: Forty-six centers in Brazil.
Synopsis: The study enrolled 2,308 patients with at least one risk factor for contrast-induced kidney injury and undergoing coronary or peripheral arterial diagnostic intravascular angiography or percutaneous intervention. Participants received two doses of acetylcysteine or placebo before and after contrast administration. End points included contrast-induced acute kidney injury, mortality, and the need for dialysis at 30 days.
Disappointingly, acetylcysteine did not significantly reduce the incidence of the end points in any patients, including the high-risk subgroups of those with diabetes mellitus and chronic renal failure, and those receiving the largest amounts of contrast. Limitations of the study include only a small number of events, as a larger number of events may help more accurately assess mortality and the need for dialysis. Additionally, creatinine may not be as good a marker for contrast-induced acute kidney injury as newer markers like cystatin C. The median volume of contrast used was low compared with previous studies, and cointerventions, such as hydration, were at the discretion of the attending physician.
Bottom line: Acetylcysteine use did not result in a lower incidence of contrast-induced acute kidney injury or other renal outcomes, and routine use prior to angiography is not recommended.
Citation: Berwange O, Cavalcanti AB, Sousa AG, et al. Acetylcysteine for prevention of renal outcomes in patients undergoing coronary and peripheral vascular angiography: main results from the randomized acetylcysteine for contrast-induced nephropathy trial (ACT). Circulation. 2011;124:1250-1259.
Clinical question: In patients undergoing coronary and peripheral angiography, does acetylcysteine before and after the procedure protect the kidneys?
Background: Contrast-induced acute kidney injury is a serious complication of procedures that use iodinated contrast material and can lead to the need for dialysis, prolonged hospital stay, and increased cost and mortality. Acetylcysteine is thought to prevent this, but previous results from more than 40 trials conflict regarding its effectiveness.
Study design: Double-blinded randomized trial.
Setting: Forty-six centers in Brazil.
Synopsis: The study enrolled 2,308 patients with at least one risk factor for contrast-induced kidney injury and undergoing coronary or peripheral arterial diagnostic intravascular angiography or percutaneous intervention. Participants received two doses of acetylcysteine or placebo before and after contrast administration. End points included contrast-induced acute kidney injury, mortality, and the need for dialysis at 30 days.
Disappointingly, acetylcysteine did not significantly reduce the incidence of the end points in any patients, including the high-risk subgroups of those with diabetes mellitus and chronic renal failure, and those receiving the largest amounts of contrast. Limitations of the study include only a small number of events, as a larger number of events may help more accurately assess mortality and the need for dialysis. Additionally, creatinine may not be as good a marker for contrast-induced acute kidney injury as newer markers like cystatin C. The median volume of contrast used was low compared with previous studies, and cointerventions, such as hydration, were at the discretion of the attending physician.
Bottom line: Acetylcysteine use did not result in a lower incidence of contrast-induced acute kidney injury or other renal outcomes, and routine use prior to angiography is not recommended.
Citation: Berwange O, Cavalcanti AB, Sousa AG, et al. Acetylcysteine for prevention of renal outcomes in patients undergoing coronary and peripheral vascular angiography: main results from the randomized acetylcysteine for contrast-induced nephropathy trial (ACT). Circulation. 2011;124:1250-1259.
Clinical question: In patients undergoing coronary and peripheral angiography, does acetylcysteine before and after the procedure protect the kidneys?
Background: Contrast-induced acute kidney injury is a serious complication of procedures that use iodinated contrast material and can lead to the need for dialysis, prolonged hospital stay, and increased cost and mortality. Acetylcysteine is thought to prevent this, but previous results from more than 40 trials conflict regarding its effectiveness.
Study design: Double-blinded randomized trial.
Setting: Forty-six centers in Brazil.
Synopsis: The study enrolled 2,308 patients with at least one risk factor for contrast-induced kidney injury and undergoing coronary or peripheral arterial diagnostic intravascular angiography or percutaneous intervention. Participants received two doses of acetylcysteine or placebo before and after contrast administration. End points included contrast-induced acute kidney injury, mortality, and the need for dialysis at 30 days.
Disappointingly, acetylcysteine did not significantly reduce the incidence of the end points in any patients, including the high-risk subgroups of those with diabetes mellitus and chronic renal failure, and those receiving the largest amounts of contrast. Limitations of the study include only a small number of events, as a larger number of events may help more accurately assess mortality and the need for dialysis. Additionally, creatinine may not be as good a marker for contrast-induced acute kidney injury as newer markers like cystatin C. The median volume of contrast used was low compared with previous studies, and cointerventions, such as hydration, were at the discretion of the attending physician.
Bottom line: Acetylcysteine use did not result in a lower incidence of contrast-induced acute kidney injury or other renal outcomes, and routine use prior to angiography is not recommended.
Citation: Berwange O, Cavalcanti AB, Sousa AG, et al. Acetylcysteine for prevention of renal outcomes in patients undergoing coronary and peripheral vascular angiography: main results from the randomized acetylcysteine for contrast-induced nephropathy trial (ACT). Circulation. 2011;124:1250-1259.