User login
Physician-Scientist Taps into Microbiome to Fight Cancer
The lowest point in the nascent career of Neelendu Dey, MD, helped seal his fate as a physician-scientist.
He had just started his first year as a resident at University of California, San Francisco. One of his patients was a 30-year-old woman who was dying of metastatic colorectal cancer. “I was in my mid-20s interacting with an individual just a few years older than I am, going through one of the most terrible health outcomes one could imagine,” Dr. Dey said.
He remembers asking the patient what he could do for her, how he could make her feel more comfortable. “That feeling of helplessness, particularly as we think about young people developing cancer, it really stuck with me through the years,” he said.
In an interview, he talked about his dual role as a physician and scientist, and how those two interests are guiding his research in precancerous conditions of the colon.
Cases like that of the young woman with colon cancer “really help drive the urgency of the work we do, and the research questions we ask, as we try to move the ball forward and help folks at earlier stages,” he said.
Q: Why did you choose GI?
When you think about what sorts of chronic diseases really impact your quality of life, gut health is one of the chief contributors among various aspects of health. And that really appealed to me — the ability to take someone who is essentially handicapped by a series of illnesses and symptoms that derive from the GI tract and enable them to return to the person they want to be, to be productive in the way that they want to be, and have a rewarding life.
As I thought about how I wanted to contribute to the future of medicine, one of the ways in which I’ve always thought that I would do that is through research. When I considered the fields that really appealed to me, both from that clinical standpoint and research standpoint, GI was one that really stood out. There has been a lot of exciting research going on in GI. My lab currently studies the microbiome, and I feel like this is an area in which we can contribute.
Q: What role does digestive health play in overall health?
Obviously, the direct answer is gut health is so critical in something like nutritional intake. Some GI symptoms, if your gut health has gone awry, can really be detrimental in terms of quality of life. But one less obvious role that digestive health plays is its long-term effects. We’re starting to appreciate that gut health, the gut microbiome, and gut immune education are probably long-term players. Some experiences in early life might shape our immunity in ways that have consequences for us much later in life. Whether we get early life antibiotics, for example, may potentially contribute to colorectal cancer down the line. Thinking about the long-term players is more challenging, but it’s also an appealing opportunity as we think about how we can shape medicine moving forward.
Q: What practice challenges have you faced in your career?
First, being a physician-scientist. It’s challenging to be either a physician alone or to be a researcher alone. And trying to do both includes the challenges of both individual worlds. It just takes more time to get all the prerequisite training. And second, there are just challenges with getting the opportunities to contribute in the ways that you want — to get the research funding, to get the papers out, things like that.
Q: Tell me about the work you’ve been doing in your lab to develop microbiome-based strategies for preventing and treating cancer.
The microbiome presents several opportunities when it comes to cancer prevention. One is identifying markers of cancer risk, or of general good health down the line. Some of those biomarkers could — potentially — feed directly into personalized risk assessment and maybe even inform a future screening strategy. The second opportunity the microbiome presents is if we identify a microbe that influences your cancer risk, can we then understand and exploit, or utilize, that mechanism to mitigate cancer risk in the future? Our lab has done work looking at subspecies levels of microbes that track with health or cancer. We’ve done some work to identify what these subspecies groupings are and have identified some links to certain precancerous changes in the colon. We think that there’s an opportunity here for future interventions.
Q: Have you published other papers?
We recently published another paper describing how some microbes can interact with a tumor suppressor gene and are influenced in a sex-biased manner to drive tumorigenesis in a mouse model. We think, based on what we’re seeing in human data, that there may be some relationships and we’re exploring that now as well.
Q: What is your vision for the future in GI, and in your career?
The vision that I have is to create clinical tools that can expand our reach and our effectiveness and cancer prevention. I think that there are opportunities for leveraging microbiome research to accomplish this. And one outcome I could imagine is leveraging some of these insights to expand noninvasive screening at even earlier ages than we do now. I mean, we just dialed back the recommended age for colonoscopy for average risk individuals to 45. But I could envision a future in which noninvasive screening starts earlier, in which the first stool-based tests that we deploy to assess personalized risk are used in the pediatric clinic.
Lightning Round
Texting or talking?
Talking
Favorite city in the United States besides the one you live in?
St. Louis
Cat or dog person?
Both
If you weren’t a GI, what would you be?
Musician
Best place you went on vacation?
Borneo
Favorite sport?
Soccer
Favorite ice cream?
Cashew-based salted caramel
What song do you have to sing along with when you hear it?
Sweet Child of Mine
Favorite movie or TV show?
25th Hour or Shawshank Redemption
Optimist or Pessimist?
Optimist
The lowest point in the nascent career of Neelendu Dey, MD, helped seal his fate as a physician-scientist.
He had just started his first year as a resident at University of California, San Francisco. One of his patients was a 30-year-old woman who was dying of metastatic colorectal cancer. “I was in my mid-20s interacting with an individual just a few years older than I am, going through one of the most terrible health outcomes one could imagine,” Dr. Dey said.
He remembers asking the patient what he could do for her, how he could make her feel more comfortable. “That feeling of helplessness, particularly as we think about young people developing cancer, it really stuck with me through the years,” he said.
In an interview, he talked about his dual role as a physician and scientist, and how those two interests are guiding his research in precancerous conditions of the colon.
Cases like that of the young woman with colon cancer “really help drive the urgency of the work we do, and the research questions we ask, as we try to move the ball forward and help folks at earlier stages,” he said.
Q: Why did you choose GI?
When you think about what sorts of chronic diseases really impact your quality of life, gut health is one of the chief contributors among various aspects of health. And that really appealed to me — the ability to take someone who is essentially handicapped by a series of illnesses and symptoms that derive from the GI tract and enable them to return to the person they want to be, to be productive in the way that they want to be, and have a rewarding life.
As I thought about how I wanted to contribute to the future of medicine, one of the ways in which I’ve always thought that I would do that is through research. When I considered the fields that really appealed to me, both from that clinical standpoint and research standpoint, GI was one that really stood out. There has been a lot of exciting research going on in GI. My lab currently studies the microbiome, and I feel like this is an area in which we can contribute.
Q: What role does digestive health play in overall health?
Obviously, the direct answer is gut health is so critical in something like nutritional intake. Some GI symptoms, if your gut health has gone awry, can really be detrimental in terms of quality of life. But one less obvious role that digestive health plays is its long-term effects. We’re starting to appreciate that gut health, the gut microbiome, and gut immune education are probably long-term players. Some experiences in early life might shape our immunity in ways that have consequences for us much later in life. Whether we get early life antibiotics, for example, may potentially contribute to colorectal cancer down the line. Thinking about the long-term players is more challenging, but it’s also an appealing opportunity as we think about how we can shape medicine moving forward.
Q: What practice challenges have you faced in your career?
First, being a physician-scientist. It’s challenging to be either a physician alone or to be a researcher alone. And trying to do both includes the challenges of both individual worlds. It just takes more time to get all the prerequisite training. And second, there are just challenges with getting the opportunities to contribute in the ways that you want — to get the research funding, to get the papers out, things like that.
Q: Tell me about the work you’ve been doing in your lab to develop microbiome-based strategies for preventing and treating cancer.
The microbiome presents several opportunities when it comes to cancer prevention. One is identifying markers of cancer risk, or of general good health down the line. Some of those biomarkers could — potentially — feed directly into personalized risk assessment and maybe even inform a future screening strategy. The second opportunity the microbiome presents is if we identify a microbe that influences your cancer risk, can we then understand and exploit, or utilize, that mechanism to mitigate cancer risk in the future? Our lab has done work looking at subspecies levels of microbes that track with health or cancer. We’ve done some work to identify what these subspecies groupings are and have identified some links to certain precancerous changes in the colon. We think that there’s an opportunity here for future interventions.
Q: Have you published other papers?
We recently published another paper describing how some microbes can interact with a tumor suppressor gene and are influenced in a sex-biased manner to drive tumorigenesis in a mouse model. We think, based on what we’re seeing in human data, that there may be some relationships and we’re exploring that now as well.
Q: What is your vision for the future in GI, and in your career?
The vision that I have is to create clinical tools that can expand our reach and our effectiveness and cancer prevention. I think that there are opportunities for leveraging microbiome research to accomplish this. And one outcome I could imagine is leveraging some of these insights to expand noninvasive screening at even earlier ages than we do now. I mean, we just dialed back the recommended age for colonoscopy for average risk individuals to 45. But I could envision a future in which noninvasive screening starts earlier, in which the first stool-based tests that we deploy to assess personalized risk are used in the pediatric clinic.
Lightning Round
Texting or talking?
Talking
Favorite city in the United States besides the one you live in?
St. Louis
Cat or dog person?
Both
If you weren’t a GI, what would you be?
Musician
Best place you went on vacation?
Borneo
Favorite sport?
Soccer
Favorite ice cream?
Cashew-based salted caramel
What song do you have to sing along with when you hear it?
Sweet Child of Mine
Favorite movie or TV show?
25th Hour or Shawshank Redemption
Optimist or Pessimist?
Optimist
The lowest point in the nascent career of Neelendu Dey, MD, helped seal his fate as a physician-scientist.
He had just started his first year as a resident at University of California, San Francisco. One of his patients was a 30-year-old woman who was dying of metastatic colorectal cancer. “I was in my mid-20s interacting with an individual just a few years older than I am, going through one of the most terrible health outcomes one could imagine,” Dr. Dey said.
He remembers asking the patient what he could do for her, how he could make her feel more comfortable. “That feeling of helplessness, particularly as we think about young people developing cancer, it really stuck with me through the years,” he said.
In an interview, he talked about his dual role as a physician and scientist, and how those two interests are guiding his research in precancerous conditions of the colon.
Cases like that of the young woman with colon cancer “really help drive the urgency of the work we do, and the research questions we ask, as we try to move the ball forward and help folks at earlier stages,” he said.
Q: Why did you choose GI?
When you think about what sorts of chronic diseases really impact your quality of life, gut health is one of the chief contributors among various aspects of health. And that really appealed to me — the ability to take someone who is essentially handicapped by a series of illnesses and symptoms that derive from the GI tract and enable them to return to the person they want to be, to be productive in the way that they want to be, and have a rewarding life.
As I thought about how I wanted to contribute to the future of medicine, one of the ways in which I’ve always thought that I would do that is through research. When I considered the fields that really appealed to me, both from that clinical standpoint and research standpoint, GI was one that really stood out. There has been a lot of exciting research going on in GI. My lab currently studies the microbiome, and I feel like this is an area in which we can contribute.
Q: What role does digestive health play in overall health?
Obviously, the direct answer is gut health is so critical in something like nutritional intake. Some GI symptoms, if your gut health has gone awry, can really be detrimental in terms of quality of life. But one less obvious role that digestive health plays is its long-term effects. We’re starting to appreciate that gut health, the gut microbiome, and gut immune education are probably long-term players. Some experiences in early life might shape our immunity in ways that have consequences for us much later in life. Whether we get early life antibiotics, for example, may potentially contribute to colorectal cancer down the line. Thinking about the long-term players is more challenging, but it’s also an appealing opportunity as we think about how we can shape medicine moving forward.
Q: What practice challenges have you faced in your career?
First, being a physician-scientist. It’s challenging to be either a physician alone or to be a researcher alone. And trying to do both includes the challenges of both individual worlds. It just takes more time to get all the prerequisite training. And second, there are just challenges with getting the opportunities to contribute in the ways that you want — to get the research funding, to get the papers out, things like that.
Q: Tell me about the work you’ve been doing in your lab to develop microbiome-based strategies for preventing and treating cancer.
The microbiome presents several opportunities when it comes to cancer prevention. One is identifying markers of cancer risk, or of general good health down the line. Some of those biomarkers could — potentially — feed directly into personalized risk assessment and maybe even inform a future screening strategy. The second opportunity the microbiome presents is if we identify a microbe that influences your cancer risk, can we then understand and exploit, or utilize, that mechanism to mitigate cancer risk in the future? Our lab has done work looking at subspecies levels of microbes that track with health or cancer. We’ve done some work to identify what these subspecies groupings are and have identified some links to certain precancerous changes in the colon. We think that there’s an opportunity here for future interventions.
Q: Have you published other papers?
We recently published another paper describing how some microbes can interact with a tumor suppressor gene and are influenced in a sex-biased manner to drive tumorigenesis in a mouse model. We think, based on what we’re seeing in human data, that there may be some relationships and we’re exploring that now as well.
Q: What is your vision for the future in GI, and in your career?
The vision that I have is to create clinical tools that can expand our reach and our effectiveness and cancer prevention. I think that there are opportunities for leveraging microbiome research to accomplish this. And one outcome I could imagine is leveraging some of these insights to expand noninvasive screening at even earlier ages than we do now. I mean, we just dialed back the recommended age for colonoscopy for average risk individuals to 45. But I could envision a future in which noninvasive screening starts earlier, in which the first stool-based tests that we deploy to assess personalized risk are used in the pediatric clinic.
Lightning Round
Texting or talking?
Talking
Favorite city in the United States besides the one you live in?
St. Louis
Cat or dog person?
Both
If you weren’t a GI, what would you be?
Musician
Best place you went on vacation?
Borneo
Favorite sport?
Soccer
Favorite ice cream?
Cashew-based salted caramel
What song do you have to sing along with when you hear it?
Sweet Child of Mine
Favorite movie or TV show?
25th Hour or Shawshank Redemption
Optimist or Pessimist?
Optimist
Statins: So Misunderstood
Recently, a patient of mine was hospitalized with chest pain. She was diagnosed with an acute coronary syndrome and started on a statin in addition to a beta-blocker, aspirin, and clopidogrel. After discharge, she had symptoms of dizziness and recurrent chest pain and her first thought was to stop the statin because she believed that her symptoms were statin-related side effects. I will cover a few areas where I think that there are some misunderstandings about statins.
Statins Are Not Bad For the Liver
When lovastatin first became available for prescription in the 1980s, frequent monitoring of transaminases was recommended. Patients and healthcare professionals became accustomed to frequent liver tests to monitor for statin toxicity, and to this day, some healthcare professionals still obtain liver function tests for this purpose.
But is there a reason to do this? Pfeffer and colleagues reported on the results of over 112,000 people enrolled in the West of Scotland Coronary Protection trial and found that the percentage of patients with any abnormal liver function test was similar (> 3 times the upper limit of normal for ALT) for patients taking pravastatin (1.4%) and for patients taking placebo (1.4%).1 A panel of liver experts concurred that statin-associated transaminase elevations were not indicative of liver damage or dysfunction.2 Furthermore, they noted that chronic liver disease and compensated cirrhosis were not contraindications to statin use.
In a small study, use of low-dose atorvastatin in patients with nonalcoholic steatohepatitis improved transaminase values in 75% of patients and liver steatosis and nonalcoholic fatty liver disease activity scores were significantly improved on biopsy in most of the patients.3 The US Food and Drug Administration (FDA) removed the recommendation for routine regular monitoring of liver function for patients on statins in 2012.4
Statins Do Not Cause Muscle Pain in Most Patients
Most muscle pain occurring in patients on statins is not due to the statin although patient concerns about muscle pain are common. In a meta-analysis of 19 large statin trials, 27.1% of participants treated with a statin reported at least one episode of muscle pain or weakness during a median of 4.3 years, compared with 26.6% of participants treated with placebo.5 Muscle pain for any reason is common, and patients on statins may stop therapy because of the symptoms.
Cohen and colleagues performed a survey of past and current statin users, asking about muscle symptoms.6 Muscle-related side effects were reported by 60% of former statin users and 25% of current users.
Herrett and colleagues performed an extensive series of n-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.7 Participants received either 2-month blocks of atorvastatin 20 mg or 2-month blocks of placebo, six times. They rated their muscle symptoms on a visual analogue scale at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further when they planned an n-of-1 trial that included statin, placebo, and no treatment.8 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on a random sequence and reported daily symptom scores. The mean symptom intensity score was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Statins Are Likely Helpful In the Very Elderly
Should we be using statins for primary prevention in our very old patients? For many years the answer was generally “no” on the basis of a lack of evidence. Patients in their 80s often were not included in clinical trials. The much used American Heart Association risk calculator stops at age 79. Given the prevalence of coronary artery disease in patients as they reach their 80s, wouldn’t primary prevention really be secondary prevention? Xu and colleagues in a recent study compared outcomes for patients who were treated with statins for primary prevention with a group who were not. In the patients aged 75-84 there was a risk reduction for major cardiovascular events of 1.2% over 5 years, and for those 85 and older the risk reduction was 4.4%. Importantly, there were no significantly increased risks for myopathies and liver dysfunction in either age group.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Pfeffer MA et al. Circulation. 2002;105(20):2341-6.
2. Cohen DE et al. Am J Cardiol. 2006;97(8A):77C-81C.
3. Hyogo H et al. Metabolism. 2008;57(12):1711-8.
4. FDA Drug Safety Communication: Important safety label changes to cholesterol-lowering statin drugs. 2012 Feb 28.
5. Cholesterol Treatment Trialists’ Collaboration. Lancet. 2022;400(10355):832-45.
6. Cohen JD et al. J Clin Lipidol. 2012;6(3):208-15.
7. Herrett E et al. BMJ. 2021 Feb 24;372:n1355.
8. Wood FA et al. N Engl J Med. 2020;383(22):2182-4.
9. Xu W et al. Ann Intern Med. 2024;177(6):701-10.
Recently, a patient of mine was hospitalized with chest pain. She was diagnosed with an acute coronary syndrome and started on a statin in addition to a beta-blocker, aspirin, and clopidogrel. After discharge, she had symptoms of dizziness and recurrent chest pain and her first thought was to stop the statin because she believed that her symptoms were statin-related side effects. I will cover a few areas where I think that there are some misunderstandings about statins.
Statins Are Not Bad For the Liver
When lovastatin first became available for prescription in the 1980s, frequent monitoring of transaminases was recommended. Patients and healthcare professionals became accustomed to frequent liver tests to monitor for statin toxicity, and to this day, some healthcare professionals still obtain liver function tests for this purpose.
But is there a reason to do this? Pfeffer and colleagues reported on the results of over 112,000 people enrolled in the West of Scotland Coronary Protection trial and found that the percentage of patients with any abnormal liver function test was similar (> 3 times the upper limit of normal for ALT) for patients taking pravastatin (1.4%) and for patients taking placebo (1.4%).1 A panel of liver experts concurred that statin-associated transaminase elevations were not indicative of liver damage or dysfunction.2 Furthermore, they noted that chronic liver disease and compensated cirrhosis were not contraindications to statin use.
In a small study, use of low-dose atorvastatin in patients with nonalcoholic steatohepatitis improved transaminase values in 75% of patients and liver steatosis and nonalcoholic fatty liver disease activity scores were significantly improved on biopsy in most of the patients.3 The US Food and Drug Administration (FDA) removed the recommendation for routine regular monitoring of liver function for patients on statins in 2012.4
Statins Do Not Cause Muscle Pain in Most Patients
Most muscle pain occurring in patients on statins is not due to the statin although patient concerns about muscle pain are common. In a meta-analysis of 19 large statin trials, 27.1% of participants treated with a statin reported at least one episode of muscle pain or weakness during a median of 4.3 years, compared with 26.6% of participants treated with placebo.5 Muscle pain for any reason is common, and patients on statins may stop therapy because of the symptoms.
Cohen and colleagues performed a survey of past and current statin users, asking about muscle symptoms.6 Muscle-related side effects were reported by 60% of former statin users and 25% of current users.
Herrett and colleagues performed an extensive series of n-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.7 Participants received either 2-month blocks of atorvastatin 20 mg or 2-month blocks of placebo, six times. They rated their muscle symptoms on a visual analogue scale at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further when they planned an n-of-1 trial that included statin, placebo, and no treatment.8 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on a random sequence and reported daily symptom scores. The mean symptom intensity score was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Statins Are Likely Helpful In the Very Elderly
Should we be using statins for primary prevention in our very old patients? For many years the answer was generally “no” on the basis of a lack of evidence. Patients in their 80s often were not included in clinical trials. The much used American Heart Association risk calculator stops at age 79. Given the prevalence of coronary artery disease in patients as they reach their 80s, wouldn’t primary prevention really be secondary prevention? Xu and colleagues in a recent study compared outcomes for patients who were treated with statins for primary prevention with a group who were not. In the patients aged 75-84 there was a risk reduction for major cardiovascular events of 1.2% over 5 years, and for those 85 and older the risk reduction was 4.4%. Importantly, there were no significantly increased risks for myopathies and liver dysfunction in either age group.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Pfeffer MA et al. Circulation. 2002;105(20):2341-6.
2. Cohen DE et al. Am J Cardiol. 2006;97(8A):77C-81C.
3. Hyogo H et al. Metabolism. 2008;57(12):1711-8.
4. FDA Drug Safety Communication: Important safety label changes to cholesterol-lowering statin drugs. 2012 Feb 28.
5. Cholesterol Treatment Trialists’ Collaboration. Lancet. 2022;400(10355):832-45.
6. Cohen JD et al. J Clin Lipidol. 2012;6(3):208-15.
7. Herrett E et al. BMJ. 2021 Feb 24;372:n1355.
8. Wood FA et al. N Engl J Med. 2020;383(22):2182-4.
9. Xu W et al. Ann Intern Med. 2024;177(6):701-10.
Recently, a patient of mine was hospitalized with chest pain. She was diagnosed with an acute coronary syndrome and started on a statin in addition to a beta-blocker, aspirin, and clopidogrel. After discharge, she had symptoms of dizziness and recurrent chest pain and her first thought was to stop the statin because she believed that her symptoms were statin-related side effects. I will cover a few areas where I think that there are some misunderstandings about statins.
Statins Are Not Bad For the Liver
When lovastatin first became available for prescription in the 1980s, frequent monitoring of transaminases was recommended. Patients and healthcare professionals became accustomed to frequent liver tests to monitor for statin toxicity, and to this day, some healthcare professionals still obtain liver function tests for this purpose.
But is there a reason to do this? Pfeffer and colleagues reported on the results of over 112,000 people enrolled in the West of Scotland Coronary Protection trial and found that the percentage of patients with any abnormal liver function test was similar (> 3 times the upper limit of normal for ALT) for patients taking pravastatin (1.4%) and for patients taking placebo (1.4%).1 A panel of liver experts concurred that statin-associated transaminase elevations were not indicative of liver damage or dysfunction.2 Furthermore, they noted that chronic liver disease and compensated cirrhosis were not contraindications to statin use.
In a small study, use of low-dose atorvastatin in patients with nonalcoholic steatohepatitis improved transaminase values in 75% of patients and liver steatosis and nonalcoholic fatty liver disease activity scores were significantly improved on biopsy in most of the patients.3 The US Food and Drug Administration (FDA) removed the recommendation for routine regular monitoring of liver function for patients on statins in 2012.4
Statins Do Not Cause Muscle Pain in Most Patients
Most muscle pain occurring in patients on statins is not due to the statin although patient concerns about muscle pain are common. In a meta-analysis of 19 large statin trials, 27.1% of participants treated with a statin reported at least one episode of muscle pain or weakness during a median of 4.3 years, compared with 26.6% of participants treated with placebo.5 Muscle pain for any reason is common, and patients on statins may stop therapy because of the symptoms.
Cohen and colleagues performed a survey of past and current statin users, asking about muscle symptoms.6 Muscle-related side effects were reported by 60% of former statin users and 25% of current users.
Herrett and colleagues performed an extensive series of n-of-1 trials involving 200 patients who had stopped or were considering stopping statins because of muscle symptoms.7 Participants received either 2-month blocks of atorvastatin 20 mg or 2-month blocks of placebo, six times. They rated their muscle symptoms on a visual analogue scale at the end of each block. There was no difference in muscle symptom scores between the statin and placebo periods.
Wood and colleagues took it a step further when they planned an n-of-1 trial that included statin, placebo, and no treatment.8 Each participant received four bottles of atorvastatin 20 mg, four bottles of placebo, and four empty bottles. Each month they used treatment from the bottles based on a random sequence and reported daily symptom scores. The mean symptom intensity score was 8.0 during no-tablet months, 15.4 during placebo months (P < .001, compared with no-tablet months), and 16.3 during statin months (P < .001, compared with no-tablet months; P = .39, compared with placebo).
Statins Are Likely Helpful In the Very Elderly
Should we be using statins for primary prevention in our very old patients? For many years the answer was generally “no” on the basis of a lack of evidence. Patients in their 80s often were not included in clinical trials. The much used American Heart Association risk calculator stops at age 79. Given the prevalence of coronary artery disease in patients as they reach their 80s, wouldn’t primary prevention really be secondary prevention? Xu and colleagues in a recent study compared outcomes for patients who were treated with statins for primary prevention with a group who were not. In the patients aged 75-84 there was a risk reduction for major cardiovascular events of 1.2% over 5 years, and for those 85 and older the risk reduction was 4.4%. Importantly, there were no significantly increased risks for myopathies and liver dysfunction in either age group.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Pfeffer MA et al. Circulation. 2002;105(20):2341-6.
2. Cohen DE et al. Am J Cardiol. 2006;97(8A):77C-81C.
3. Hyogo H et al. Metabolism. 2008;57(12):1711-8.
4. FDA Drug Safety Communication: Important safety label changes to cholesterol-lowering statin drugs. 2012 Feb 28.
5. Cholesterol Treatment Trialists’ Collaboration. Lancet. 2022;400(10355):832-45.
6. Cohen JD et al. J Clin Lipidol. 2012;6(3):208-15.
7. Herrett E et al. BMJ. 2021 Feb 24;372:n1355.
8. Wood FA et al. N Engl J Med. 2020;383(22):2182-4.
9. Xu W et al. Ann Intern Med. 2024;177(6):701-10.
Identifying, Treating Lyme Disease in Primary Care
Geographic spread of the ticks that most often cause Lyme disease in the United States and a rise in incidence of bites, resulting in 476,000 new US cases a year, have increased the chances that physicians who have never encountered a patient with Lyme disease will see their first cases.
“It’s increasing in areas where it was not seen before,” Steven E. Schutzer, MD, with the Department of Medicine, Rutgers New Jersey Medical School, Newark, said in an interview. Dr. Schutzer coauthored a report on diagnosing and treating Lyme disease with Patricia K. Coyle, MD, Department of Neurology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York.
The report, a Curbside Consult published in New England Journal of Medicine Evidence, comes amid high season for Lyme disease. Bites from an ixodid (hard shield) tick — almost always the source of the disease in the United States — are most common from April through October.
Identifying the Bite
About 70%-90% of the time, Lyme disease will be signaled by erythema migrans (EM) or lesion expanding from the tick bite site, the authors wrote. The “classic” presentation looks like a bullseye, but most of the time the skin will show a variation of that, the authors noted.
“The presence of EM is considered the best clinical diagnostic marker for Lyme disease,” they wrote.
Other dermatologic conditions, however, can complicate diagnosis: “EM mimickers include contact dermatitis, other arthropod bites, fixed drug eruptions, granuloma annulare, cellulitis, dermatophytosis, and systemic lupus erythematosus,” they wrote.
Testing Steps
“The current recommendation is to do two-step testing almost simultaneously,” Dr. Schutzer said in an interview. The first, he said, is an ELISA (enzyme-linked immunosorbent assay)-type test and the second one, used for years, has been a pictoral view of a Western immunoblot showing which antigens of the Lyme bacteria, Borrelia burgdorferi, the antibodies are reacting to.
However, the pictoral view is subjective and some of the antigens could be cross-reactive. So the U.S. Food and Drug Administration (FDA) “has been allowing newer substitutes like a second ELISA-like assay that often uses more recombinant, less cross-reactive antigen targets,” he said. The authors advised that, “The second-tier test should not be performed alone without the first tier.”
Dr. Schutzer advised physicians to check with the lab they plan to use before sending samples.
“If you’re a practicing physician and you know you’re using a particular laboratory, you should familiarize yourself with them, talking to one of the clinical pathologists involved in advance to know what the limitations are.” Take the time to talk with the person overseeing the test and get tips on how they want the sample transported and how the cases should be reported, he said.
If the patient has neurological symptoms, he said, before treating talk with a neurologist who can advise whether, for instance, a spinal tap is in order or whether an emergency department visit is appropriate.
“If you just start proceeding you may mess up the diagnostic signs that could show up in a lab test. Don’t be hesitant to ask for extra input from colleagues,” Dr. Schutzer said.
Suspicion in Endemic Areas
On Long Island, New York, where Lyme disease is endemic, internist Ian Storch, DO, said he sees “a few cases a season.
“We have a lot of people over the summer going to the Hamptons and areas out east for the weekend and tick bites are not uncommon,” he said. “People panic.”
He said one thing it’s important to tell patients is that the tick has to be on the skin for 48-72 hours to transmit the disease. If individuals were in a wooded area and were fine before they got there and the tick was attached for less than 2 days, “they’re usually fine.”
Another issue, Dr. Storch said, is patients sometimes want to get tested for Lyme disease immediately after a tick bite. But the antibody test doesn’t turn positive for weeks, he noted, and you can get a false-negative result. “If you’re worried and you really want to test, you need to wait 6 weeks to do the blood test.”
In his region, he said that although a tick bite is a red flag, he may also suspect Lyme disease when a patient presents with otherwise unexplained joint pain, weakness, lethargy, or fever. “In our area, those are things that would make you test for Lyme.”
He also urged consideration of Lyme in this new age of long COVID. Weakness, fatigue, and lethargy are also classic symptoms of long COVID, he noted. “Keep Lyme disease in your differential because there is a lot of overlap with chronic Lyme disease,” Dr. Storch said.
Discerning Lyme from Southern Tick–Associated Rash Illness
Bonnie M. Word, MD, director of the Houston Travel Medicine Clinic in Texas, where Lyme disease is not endemic, said Lyme disease “will not and should not be on the initial differential diagnosis for those residing in nonendemic areas unless a history of travel to an endemic area is obtained.”
She noted the typical EM rash may not be as distinct or easy to discern on black and brown skin. In addition, she said, EM may have many variations in presentation, such as a crusted center or faint borders, which could lead to a delay in diagnosis and treatment. She suggested consulting the CDC guidance on Lyme disease rashes.
Another challenge in diagnosis, she said, is the patient who presents with what appears to be a classic EM lesion but does not live in a Lyme-endemic area. In Texas, Southern Tick–Associated Rash Illness (STARI) may present with a similar lesion, she said.
“It is transmitted by the Lone Star Tick, which is found in the southeast and south-central US,” Dr. Word said. “However, its habitat is moving northward and westerly,” she said.
Adding Lyme disease to the differential diagnosis is reasonable, she said, if a patient presents with neurologic symptoms “such as a facial palsy, meningitis, radiculitis, and carditis if in addition to their symptoms there is evidence of an epidemiologic link to a Lyme-endemic region.”
She noted that a detailed travel history is important as “Lyme is also endemic in Eastern Canada, Europe, states of the former Soviet Union, China, Mongolia, and Japan.”
Primary care physicians play a critical role in evaluating, diagnosing, and treating most cases of early Lyme disease, thus limiting the number of people who will develop disseminated or late Lyme disease, she said. “The two latter manifestations are most often treated by infectious disease, neurology, or rheumatology specialists.”
Treatment*
Treatment is tailored to the clinical situation, Dr. Schutzer and Dr. Coyle write. A watch-and-wait approach may be appropriate in an asymptomatic but concerned person, even in an endemic area if the person has no known tick bite and no EM lesion.
If there is high risk of an infected ixodid tick bite in a high-incidence area and the tick was attached for at least 36 hours but less than 72 hours, one dose of doxycycline has been recommended as prophylaxis.
When a diagnosis of early nondisseminated Lyme disease is made after observation of an EM lesion, oral antibiotics are typically used to treat for 10 to 14 days. Suggested oral antibiotics and doses are 100 mg of doxycycline twice a day, 500 mg of amoxicillin three times a day, or 500 mg of cefuroxime twice a day, the authors write.
Dr. Schutzer said he hopes the paper serves as a refresher for those physicians who regularly see Lyme disease cases and also helps those newly included in the disease’s spreading regions.
“The earlier you diagnose it, the earlier you can treat it and the better the chance for a favorable outcome,” he said.
Dr. Schutzer, Dr. Coyle, Dr. Storch, and Dr. Word reported no relevant financial relationships.
*This story was updated on August, 2, 2024.
Geographic spread of the ticks that most often cause Lyme disease in the United States and a rise in incidence of bites, resulting in 476,000 new US cases a year, have increased the chances that physicians who have never encountered a patient with Lyme disease will see their first cases.
“It’s increasing in areas where it was not seen before,” Steven E. Schutzer, MD, with the Department of Medicine, Rutgers New Jersey Medical School, Newark, said in an interview. Dr. Schutzer coauthored a report on diagnosing and treating Lyme disease with Patricia K. Coyle, MD, Department of Neurology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York.
The report, a Curbside Consult published in New England Journal of Medicine Evidence, comes amid high season for Lyme disease. Bites from an ixodid (hard shield) tick — almost always the source of the disease in the United States — are most common from April through October.
Identifying the Bite
About 70%-90% of the time, Lyme disease will be signaled by erythema migrans (EM) or lesion expanding from the tick bite site, the authors wrote. The “classic” presentation looks like a bullseye, but most of the time the skin will show a variation of that, the authors noted.
“The presence of EM is considered the best clinical diagnostic marker for Lyme disease,” they wrote.
Other dermatologic conditions, however, can complicate diagnosis: “EM mimickers include contact dermatitis, other arthropod bites, fixed drug eruptions, granuloma annulare, cellulitis, dermatophytosis, and systemic lupus erythematosus,” they wrote.
Testing Steps
“The current recommendation is to do two-step testing almost simultaneously,” Dr. Schutzer said in an interview. The first, he said, is an ELISA (enzyme-linked immunosorbent assay)-type test and the second one, used for years, has been a pictoral view of a Western immunoblot showing which antigens of the Lyme bacteria, Borrelia burgdorferi, the antibodies are reacting to.
However, the pictoral view is subjective and some of the antigens could be cross-reactive. So the U.S. Food and Drug Administration (FDA) “has been allowing newer substitutes like a second ELISA-like assay that often uses more recombinant, less cross-reactive antigen targets,” he said. The authors advised that, “The second-tier test should not be performed alone without the first tier.”
Dr. Schutzer advised physicians to check with the lab they plan to use before sending samples.
“If you’re a practicing physician and you know you’re using a particular laboratory, you should familiarize yourself with them, talking to one of the clinical pathologists involved in advance to know what the limitations are.” Take the time to talk with the person overseeing the test and get tips on how they want the sample transported and how the cases should be reported, he said.
If the patient has neurological symptoms, he said, before treating talk with a neurologist who can advise whether, for instance, a spinal tap is in order or whether an emergency department visit is appropriate.
“If you just start proceeding you may mess up the diagnostic signs that could show up in a lab test. Don’t be hesitant to ask for extra input from colleagues,” Dr. Schutzer said.
Suspicion in Endemic Areas
On Long Island, New York, where Lyme disease is endemic, internist Ian Storch, DO, said he sees “a few cases a season.
“We have a lot of people over the summer going to the Hamptons and areas out east for the weekend and tick bites are not uncommon,” he said. “People panic.”
He said one thing it’s important to tell patients is that the tick has to be on the skin for 48-72 hours to transmit the disease. If individuals were in a wooded area and were fine before they got there and the tick was attached for less than 2 days, “they’re usually fine.”
Another issue, Dr. Storch said, is patients sometimes want to get tested for Lyme disease immediately after a tick bite. But the antibody test doesn’t turn positive for weeks, he noted, and you can get a false-negative result. “If you’re worried and you really want to test, you need to wait 6 weeks to do the blood test.”
In his region, he said that although a tick bite is a red flag, he may also suspect Lyme disease when a patient presents with otherwise unexplained joint pain, weakness, lethargy, or fever. “In our area, those are things that would make you test for Lyme.”
He also urged consideration of Lyme in this new age of long COVID. Weakness, fatigue, and lethargy are also classic symptoms of long COVID, he noted. “Keep Lyme disease in your differential because there is a lot of overlap with chronic Lyme disease,” Dr. Storch said.
Discerning Lyme from Southern Tick–Associated Rash Illness
Bonnie M. Word, MD, director of the Houston Travel Medicine Clinic in Texas, where Lyme disease is not endemic, said Lyme disease “will not and should not be on the initial differential diagnosis for those residing in nonendemic areas unless a history of travel to an endemic area is obtained.”
She noted the typical EM rash may not be as distinct or easy to discern on black and brown skin. In addition, she said, EM may have many variations in presentation, such as a crusted center or faint borders, which could lead to a delay in diagnosis and treatment. She suggested consulting the CDC guidance on Lyme disease rashes.
Another challenge in diagnosis, she said, is the patient who presents with what appears to be a classic EM lesion but does not live in a Lyme-endemic area. In Texas, Southern Tick–Associated Rash Illness (STARI) may present with a similar lesion, she said.
“It is transmitted by the Lone Star Tick, which is found in the southeast and south-central US,” Dr. Word said. “However, its habitat is moving northward and westerly,” she said.
Adding Lyme disease to the differential diagnosis is reasonable, she said, if a patient presents with neurologic symptoms “such as a facial palsy, meningitis, radiculitis, and carditis if in addition to their symptoms there is evidence of an epidemiologic link to a Lyme-endemic region.”
She noted that a detailed travel history is important as “Lyme is also endemic in Eastern Canada, Europe, states of the former Soviet Union, China, Mongolia, and Japan.”
Primary care physicians play a critical role in evaluating, diagnosing, and treating most cases of early Lyme disease, thus limiting the number of people who will develop disseminated or late Lyme disease, she said. “The two latter manifestations are most often treated by infectious disease, neurology, or rheumatology specialists.”
Treatment*
Treatment is tailored to the clinical situation, Dr. Schutzer and Dr. Coyle write. A watch-and-wait approach may be appropriate in an asymptomatic but concerned person, even in an endemic area if the person has no known tick bite and no EM lesion.
If there is high risk of an infected ixodid tick bite in a high-incidence area and the tick was attached for at least 36 hours but less than 72 hours, one dose of doxycycline has been recommended as prophylaxis.
When a diagnosis of early nondisseminated Lyme disease is made after observation of an EM lesion, oral antibiotics are typically used to treat for 10 to 14 days. Suggested oral antibiotics and doses are 100 mg of doxycycline twice a day, 500 mg of amoxicillin three times a day, or 500 mg of cefuroxime twice a day, the authors write.
Dr. Schutzer said he hopes the paper serves as a refresher for those physicians who regularly see Lyme disease cases and also helps those newly included in the disease’s spreading regions.
“The earlier you diagnose it, the earlier you can treat it and the better the chance for a favorable outcome,” he said.
Dr. Schutzer, Dr. Coyle, Dr. Storch, and Dr. Word reported no relevant financial relationships.
*This story was updated on August, 2, 2024.
Geographic spread of the ticks that most often cause Lyme disease in the United States and a rise in incidence of bites, resulting in 476,000 new US cases a year, have increased the chances that physicians who have never encountered a patient with Lyme disease will see their first cases.
“It’s increasing in areas where it was not seen before,” Steven E. Schutzer, MD, with the Department of Medicine, Rutgers New Jersey Medical School, Newark, said in an interview. Dr. Schutzer coauthored a report on diagnosing and treating Lyme disease with Patricia K. Coyle, MD, Department of Neurology, Renaissance School of Medicine at Stony Brook University, Stony Brook, New York.
The report, a Curbside Consult published in New England Journal of Medicine Evidence, comes amid high season for Lyme disease. Bites from an ixodid (hard shield) tick — almost always the source of the disease in the United States — are most common from April through October.
Identifying the Bite
About 70%-90% of the time, Lyme disease will be signaled by erythema migrans (EM) or lesion expanding from the tick bite site, the authors wrote. The “classic” presentation looks like a bullseye, but most of the time the skin will show a variation of that, the authors noted.
“The presence of EM is considered the best clinical diagnostic marker for Lyme disease,” they wrote.
Other dermatologic conditions, however, can complicate diagnosis: “EM mimickers include contact dermatitis, other arthropod bites, fixed drug eruptions, granuloma annulare, cellulitis, dermatophytosis, and systemic lupus erythematosus,” they wrote.
Testing Steps
“The current recommendation is to do two-step testing almost simultaneously,” Dr. Schutzer said in an interview. The first, he said, is an ELISA (enzyme-linked immunosorbent assay)-type test and the second one, used for years, has been a pictoral view of a Western immunoblot showing which antigens of the Lyme bacteria, Borrelia burgdorferi, the antibodies are reacting to.
However, the pictoral view is subjective and some of the antigens could be cross-reactive. So the U.S. Food and Drug Administration (FDA) “has been allowing newer substitutes like a second ELISA-like assay that often uses more recombinant, less cross-reactive antigen targets,” he said. The authors advised that, “The second-tier test should not be performed alone without the first tier.”
Dr. Schutzer advised physicians to check with the lab they plan to use before sending samples.
“If you’re a practicing physician and you know you’re using a particular laboratory, you should familiarize yourself with them, talking to one of the clinical pathologists involved in advance to know what the limitations are.” Take the time to talk with the person overseeing the test and get tips on how they want the sample transported and how the cases should be reported, he said.
If the patient has neurological symptoms, he said, before treating talk with a neurologist who can advise whether, for instance, a spinal tap is in order or whether an emergency department visit is appropriate.
“If you just start proceeding you may mess up the diagnostic signs that could show up in a lab test. Don’t be hesitant to ask for extra input from colleagues,” Dr. Schutzer said.
Suspicion in Endemic Areas
On Long Island, New York, where Lyme disease is endemic, internist Ian Storch, DO, said he sees “a few cases a season.
“We have a lot of people over the summer going to the Hamptons and areas out east for the weekend and tick bites are not uncommon,” he said. “People panic.”
He said one thing it’s important to tell patients is that the tick has to be on the skin for 48-72 hours to transmit the disease. If individuals were in a wooded area and were fine before they got there and the tick was attached for less than 2 days, “they’re usually fine.”
Another issue, Dr. Storch said, is patients sometimes want to get tested for Lyme disease immediately after a tick bite. But the antibody test doesn’t turn positive for weeks, he noted, and you can get a false-negative result. “If you’re worried and you really want to test, you need to wait 6 weeks to do the blood test.”
In his region, he said that although a tick bite is a red flag, he may also suspect Lyme disease when a patient presents with otherwise unexplained joint pain, weakness, lethargy, or fever. “In our area, those are things that would make you test for Lyme.”
He also urged consideration of Lyme in this new age of long COVID. Weakness, fatigue, and lethargy are also classic symptoms of long COVID, he noted. “Keep Lyme disease in your differential because there is a lot of overlap with chronic Lyme disease,” Dr. Storch said.
Discerning Lyme from Southern Tick–Associated Rash Illness
Bonnie M. Word, MD, director of the Houston Travel Medicine Clinic in Texas, where Lyme disease is not endemic, said Lyme disease “will not and should not be on the initial differential diagnosis for those residing in nonendemic areas unless a history of travel to an endemic area is obtained.”
She noted the typical EM rash may not be as distinct or easy to discern on black and brown skin. In addition, she said, EM may have many variations in presentation, such as a crusted center or faint borders, which could lead to a delay in diagnosis and treatment. She suggested consulting the CDC guidance on Lyme disease rashes.
Another challenge in diagnosis, she said, is the patient who presents with what appears to be a classic EM lesion but does not live in a Lyme-endemic area. In Texas, Southern Tick–Associated Rash Illness (STARI) may present with a similar lesion, she said.
“It is transmitted by the Lone Star Tick, which is found in the southeast and south-central US,” Dr. Word said. “However, its habitat is moving northward and westerly,” she said.
Adding Lyme disease to the differential diagnosis is reasonable, she said, if a patient presents with neurologic symptoms “such as a facial palsy, meningitis, radiculitis, and carditis if in addition to their symptoms there is evidence of an epidemiologic link to a Lyme-endemic region.”
She noted that a detailed travel history is important as “Lyme is also endemic in Eastern Canada, Europe, states of the former Soviet Union, China, Mongolia, and Japan.”
Primary care physicians play a critical role in evaluating, diagnosing, and treating most cases of early Lyme disease, thus limiting the number of people who will develop disseminated or late Lyme disease, she said. “The two latter manifestations are most often treated by infectious disease, neurology, or rheumatology specialists.”
Treatment*
Treatment is tailored to the clinical situation, Dr. Schutzer and Dr. Coyle write. A watch-and-wait approach may be appropriate in an asymptomatic but concerned person, even in an endemic area if the person has no known tick bite and no EM lesion.
If there is high risk of an infected ixodid tick bite in a high-incidence area and the tick was attached for at least 36 hours but less than 72 hours, one dose of doxycycline has been recommended as prophylaxis.
When a diagnosis of early nondisseminated Lyme disease is made after observation of an EM lesion, oral antibiotics are typically used to treat for 10 to 14 days. Suggested oral antibiotics and doses are 100 mg of doxycycline twice a day, 500 mg of amoxicillin three times a day, or 500 mg of cefuroxime twice a day, the authors write.
Dr. Schutzer said he hopes the paper serves as a refresher for those physicians who regularly see Lyme disease cases and also helps those newly included in the disease’s spreading regions.
“The earlier you diagnose it, the earlier you can treat it and the better the chance for a favorable outcome,” he said.
Dr. Schutzer, Dr. Coyle, Dr. Storch, and Dr. Word reported no relevant financial relationships.
*This story was updated on August, 2, 2024.
Maternity Care in Rural Areas Is in Crisis. Can More Doulas Help?
When Bristeria Clark went into labor with her son in 2015, her contractions were steady at first. Then, they stalled. Her cervix stopped dilating. After a few hours, doctors at Phoebe Putney Memorial Hospital in Albany, Georgia, prepped Ms. Clark for an emergency cesarean section.
It wasn’t the vaginal birth Ms. Clark had hoped for during her pregnancy.
“I was freaking out. That was my first child. Like, of course you don’t plan that,” she said. “I just remember the gas pulling up to my face and I ended up going to sleep.”
She remembered feeling a rush of relief when she woke to see that her baby boy was healthy.
Ms. Clark, a 33-year-old nursing student who also works full-time in county government, had another C-section when her second child was born in 2020. This time, the cesarean was planned.
Ms. Clark said she’s grateful the physicians and nurses who delivered both her babies were kind and caring during her labor and delivery. But looking back, she said, she wishes she had had a doula for one-on-one support through pregnancy, childbirth, and the postpartum period. Now she wants to give other women the option she didn’t have.
Ms. Clark is a member of Morehouse School of Medicine’s first class of rural doulas, called Perinatal Patient Navigators.
The program recently graduated a dozen participants, all Black women from southwestern Georgia. They have completed more than 5 months of training and are scheduled to begin working with pregnant and postpartum patients this year.
“We’re developing a workforce that’s going to be providing the support that Black women and birthing people need,” Natalie Hernandez-Green, an associate professor of obstetrics and gynecology at Morehouse School of Medicine, Atlanta, Georgia, said at the doula commencement ceremony in Albany, Georgia.
Albany is Morehouse School of Medicine’s second Perinatal Patient Navigator program site. The first has been up and running in Atlanta since training began in the fall of 2022.
Georgia has one of the highest rates of maternal mortality in the country, according to an analysis by KFF, a health information nonprofit that includes KFF Health News. And Black Georgians are more than twice as likely as White Georgians to die of causes related to pregnancy.
“It doesn’t matter whether you’re rich or poor. Black women are dying at [an] alarming rate from pregnancy-related complications,” said Dr. Hernandez-Green, who is also executive director of the Center for Maternal Health Equity at Morehouse School of Medicine. “And we’re about to change that, one person at a time.”
The presence of a doula, along with regular nursing care, is associated with improved labor and delivery outcomes, reduced stress, and higher rates of patient satisfaction, according to the American College of Obstetricians and Gynecologists.
Multiple studies also link doulas to fewer expensive childbirth interventions, including cesarean births.
Doulas are not medical professionals. They are trained to offer education about the pregnancy and postpartum periods, to guide patients through the healthcare system, and to provide emotional and physical support before, during, and after childbirth.
Morehouse School of Medicine’s program is among a growing number of similar efforts being introduced across the country as more communities look to doulas to help address maternal mortality and poor maternal health outcomes, particularly for Black women and other women of color.
Now that she has graduated, Ms. Clark said she’s looking forward to helping other women in her community as a doula. “To be that person that would be there for my clients, treat them like a sister or like a mother, in a sense of just treating them with utmost respect,” she said. “The ultimate goal is to make them feel comfortable and let them know ‘I’m here to support you.’ ” Her training has inspired her to become an advocate for maternal health issues in southwestern Georgia.
Grants fund Morehouse School of Medicine’s doula program, which costs $350,000 a year to operate. Graduates are given a $2,000 training stipend and the program places five graduates with healthcare providers in southwestern Georgia. Grant money also pays the doulas’ salaries for 1 year.
“It’s not sustainable if you’re chasing the next grant to fund it,” said Rachel Hardeman, a professor of health and racial equity at the University of Minnesota School of Public Health.
Thirteen states cover doulas through Medicaid, according to the Georgetown University Center for Children and Families.
Dr. Hardeman and others have found that when Medicaid programs cover doula care, states save millions of dollars in healthcare costs. “We were able to calculate the return on investment if Medicaid decided to reimburse doulas for pregnant people who are Medicaid beneficiaries,” she said.
That’s because doulas can help reduce the number of expensive medical interventions during and after birth, and improving delivery outcomes, including reduced cesarean sections.
Doulas can even reduce the likelihood of preterm birth.
“An infant that is born at a very, very early gestational age is going to require a great deal of resources and interventions to ensure that they survive and then continue to thrive,” Dr. Hardeman said.
There is growing demand for doula services in Georgia, said Fowzio Jama, director of research for Healthy Mothers, Healthy Babies Coalition of Georgia. Her group recently completed a pilot study that offered doula services to about 170 Georgians covered under Medicaid. “We had a wait-list of over 200 clients and we wanted to give them the support that they needed, but we just couldn’t with the given resources that we had,” Ms. Jama said.
Doula services can cost hundreds or thousands of dollars out-of-pocket, making it too expensive for many low-income people, rural communities, and communities of color, many of which suffer from shortages in maternity care, according to the March of Dimes.
The Healthy Mothers, Healthy Babies study found that matching high-risk patients with doulas — particularly doulas from similar racial and ethnic backgrounds — had a positive effect on patients.
“There was a reduced use of pitocin to induce labor. We saw fewer requests for pain medication. And with our infants, only 6% were low birth weight,” Ms. Jama said.
Still, she and others acknowledge that doulas alone can’t fix the problem of high maternal mortality and morbidity rates.
States, including Georgia, need to do more to bring comprehensive maternity care to communities that need more options, Dr. Hardeman said.
“I think it’s important to understand that doulas are not going to save us, and we should not put that expectation on them. Doulas are a tool,” she said. “They are a piece of the puzzle that is helping to impact a really, really complex issue.”
In the meantime, Joan Anderson, 55, said she’s excited to get to work supporting patients, especially from rural areas around Albany.
“I feel like I’m equipped to go out and be that voice, be that person that our community needs so bad,” said Ms. Anderson, a graduate of the Morehouse School of Medicine doula program. “I am encouraged to know that I will be joining in that mission, that fight for us, as far as maternal health is concerned.”
Ms. Anderson said that someday she wants to open a birthing center to provide maternity care. “We do not have one here in southwest Georgia at all,” Ms. Anderson said.
In addition to providing support during and after childbirth, Ms. Anderson and her fellow graduates are trained to assess their patients’ needs and connect them to services such as food assistance, mental health care, transportation to prenatal appointments, and breastfeeding assistance.
Their work is likely to have ripple effects across a largely rural corner of Georgia, said Sherrell Byrd, who co-founded and directs SOWEGA Rising, a nonprofit organization in southwestern Georgia.
“So many of the graduates are part of church networks, they are part of community organizations, some of them are our government workers. They’re very connected,” Ms. Byrd said. “And I think that connectedness is what’s going to help them be successful moving forward.”
This reporting is part of a fellowship with the Association of Health Care Journalists supported by The Commonwealth Fund. It comes from a partnership that includes WABE, NPR, and KFF Health News.
A version of this article first appeared on KFF Health News.
When Bristeria Clark went into labor with her son in 2015, her contractions were steady at first. Then, they stalled. Her cervix stopped dilating. After a few hours, doctors at Phoebe Putney Memorial Hospital in Albany, Georgia, prepped Ms. Clark for an emergency cesarean section.
It wasn’t the vaginal birth Ms. Clark had hoped for during her pregnancy.
“I was freaking out. That was my first child. Like, of course you don’t plan that,” she said. “I just remember the gas pulling up to my face and I ended up going to sleep.”
She remembered feeling a rush of relief when she woke to see that her baby boy was healthy.
Ms. Clark, a 33-year-old nursing student who also works full-time in county government, had another C-section when her second child was born in 2020. This time, the cesarean was planned.
Ms. Clark said she’s grateful the physicians and nurses who delivered both her babies were kind and caring during her labor and delivery. But looking back, she said, she wishes she had had a doula for one-on-one support through pregnancy, childbirth, and the postpartum period. Now she wants to give other women the option she didn’t have.
Ms. Clark is a member of Morehouse School of Medicine’s first class of rural doulas, called Perinatal Patient Navigators.
The program recently graduated a dozen participants, all Black women from southwestern Georgia. They have completed more than 5 months of training and are scheduled to begin working with pregnant and postpartum patients this year.
“We’re developing a workforce that’s going to be providing the support that Black women and birthing people need,” Natalie Hernandez-Green, an associate professor of obstetrics and gynecology at Morehouse School of Medicine, Atlanta, Georgia, said at the doula commencement ceremony in Albany, Georgia.
Albany is Morehouse School of Medicine’s second Perinatal Patient Navigator program site. The first has been up and running in Atlanta since training began in the fall of 2022.
Georgia has one of the highest rates of maternal mortality in the country, according to an analysis by KFF, a health information nonprofit that includes KFF Health News. And Black Georgians are more than twice as likely as White Georgians to die of causes related to pregnancy.
“It doesn’t matter whether you’re rich or poor. Black women are dying at [an] alarming rate from pregnancy-related complications,” said Dr. Hernandez-Green, who is also executive director of the Center for Maternal Health Equity at Morehouse School of Medicine. “And we’re about to change that, one person at a time.”
The presence of a doula, along with regular nursing care, is associated with improved labor and delivery outcomes, reduced stress, and higher rates of patient satisfaction, according to the American College of Obstetricians and Gynecologists.
Multiple studies also link doulas to fewer expensive childbirth interventions, including cesarean births.
Doulas are not medical professionals. They are trained to offer education about the pregnancy and postpartum periods, to guide patients through the healthcare system, and to provide emotional and physical support before, during, and after childbirth.
Morehouse School of Medicine’s program is among a growing number of similar efforts being introduced across the country as more communities look to doulas to help address maternal mortality and poor maternal health outcomes, particularly for Black women and other women of color.
Now that she has graduated, Ms. Clark said she’s looking forward to helping other women in her community as a doula. “To be that person that would be there for my clients, treat them like a sister or like a mother, in a sense of just treating them with utmost respect,” she said. “The ultimate goal is to make them feel comfortable and let them know ‘I’m here to support you.’ ” Her training has inspired her to become an advocate for maternal health issues in southwestern Georgia.
Grants fund Morehouse School of Medicine’s doula program, which costs $350,000 a year to operate. Graduates are given a $2,000 training stipend and the program places five graduates with healthcare providers in southwestern Georgia. Grant money also pays the doulas’ salaries for 1 year.
“It’s not sustainable if you’re chasing the next grant to fund it,” said Rachel Hardeman, a professor of health and racial equity at the University of Minnesota School of Public Health.
Thirteen states cover doulas through Medicaid, according to the Georgetown University Center for Children and Families.
Dr. Hardeman and others have found that when Medicaid programs cover doula care, states save millions of dollars in healthcare costs. “We were able to calculate the return on investment if Medicaid decided to reimburse doulas for pregnant people who are Medicaid beneficiaries,” she said.
That’s because doulas can help reduce the number of expensive medical interventions during and after birth, and improving delivery outcomes, including reduced cesarean sections.
Doulas can even reduce the likelihood of preterm birth.
“An infant that is born at a very, very early gestational age is going to require a great deal of resources and interventions to ensure that they survive and then continue to thrive,” Dr. Hardeman said.
There is growing demand for doula services in Georgia, said Fowzio Jama, director of research for Healthy Mothers, Healthy Babies Coalition of Georgia. Her group recently completed a pilot study that offered doula services to about 170 Georgians covered under Medicaid. “We had a wait-list of over 200 clients and we wanted to give them the support that they needed, but we just couldn’t with the given resources that we had,” Ms. Jama said.
Doula services can cost hundreds or thousands of dollars out-of-pocket, making it too expensive for many low-income people, rural communities, and communities of color, many of which suffer from shortages in maternity care, according to the March of Dimes.
The Healthy Mothers, Healthy Babies study found that matching high-risk patients with doulas — particularly doulas from similar racial and ethnic backgrounds — had a positive effect on patients.
“There was a reduced use of pitocin to induce labor. We saw fewer requests for pain medication. And with our infants, only 6% were low birth weight,” Ms. Jama said.
Still, she and others acknowledge that doulas alone can’t fix the problem of high maternal mortality and morbidity rates.
States, including Georgia, need to do more to bring comprehensive maternity care to communities that need more options, Dr. Hardeman said.
“I think it’s important to understand that doulas are not going to save us, and we should not put that expectation on them. Doulas are a tool,” she said. “They are a piece of the puzzle that is helping to impact a really, really complex issue.”
In the meantime, Joan Anderson, 55, said she’s excited to get to work supporting patients, especially from rural areas around Albany.
“I feel like I’m equipped to go out and be that voice, be that person that our community needs so bad,” said Ms. Anderson, a graduate of the Morehouse School of Medicine doula program. “I am encouraged to know that I will be joining in that mission, that fight for us, as far as maternal health is concerned.”
Ms. Anderson said that someday she wants to open a birthing center to provide maternity care. “We do not have one here in southwest Georgia at all,” Ms. Anderson said.
In addition to providing support during and after childbirth, Ms. Anderson and her fellow graduates are trained to assess their patients’ needs and connect them to services such as food assistance, mental health care, transportation to prenatal appointments, and breastfeeding assistance.
Their work is likely to have ripple effects across a largely rural corner of Georgia, said Sherrell Byrd, who co-founded and directs SOWEGA Rising, a nonprofit organization in southwestern Georgia.
“So many of the graduates are part of church networks, they are part of community organizations, some of them are our government workers. They’re very connected,” Ms. Byrd said. “And I think that connectedness is what’s going to help them be successful moving forward.”
This reporting is part of a fellowship with the Association of Health Care Journalists supported by The Commonwealth Fund. It comes from a partnership that includes WABE, NPR, and KFF Health News.
A version of this article first appeared on KFF Health News.
When Bristeria Clark went into labor with her son in 2015, her contractions were steady at first. Then, they stalled. Her cervix stopped dilating. After a few hours, doctors at Phoebe Putney Memorial Hospital in Albany, Georgia, prepped Ms. Clark for an emergency cesarean section.
It wasn’t the vaginal birth Ms. Clark had hoped for during her pregnancy.
“I was freaking out. That was my first child. Like, of course you don’t plan that,” she said. “I just remember the gas pulling up to my face and I ended up going to sleep.”
She remembered feeling a rush of relief when she woke to see that her baby boy was healthy.
Ms. Clark, a 33-year-old nursing student who also works full-time in county government, had another C-section when her second child was born in 2020. This time, the cesarean was planned.
Ms. Clark said she’s grateful the physicians and nurses who delivered both her babies were kind and caring during her labor and delivery. But looking back, she said, she wishes she had had a doula for one-on-one support through pregnancy, childbirth, and the postpartum period. Now she wants to give other women the option she didn’t have.
Ms. Clark is a member of Morehouse School of Medicine’s first class of rural doulas, called Perinatal Patient Navigators.
The program recently graduated a dozen participants, all Black women from southwestern Georgia. They have completed more than 5 months of training and are scheduled to begin working with pregnant and postpartum patients this year.
“We’re developing a workforce that’s going to be providing the support that Black women and birthing people need,” Natalie Hernandez-Green, an associate professor of obstetrics and gynecology at Morehouse School of Medicine, Atlanta, Georgia, said at the doula commencement ceremony in Albany, Georgia.
Albany is Morehouse School of Medicine’s second Perinatal Patient Navigator program site. The first has been up and running in Atlanta since training began in the fall of 2022.
Georgia has one of the highest rates of maternal mortality in the country, according to an analysis by KFF, a health information nonprofit that includes KFF Health News. And Black Georgians are more than twice as likely as White Georgians to die of causes related to pregnancy.
“It doesn’t matter whether you’re rich or poor. Black women are dying at [an] alarming rate from pregnancy-related complications,” said Dr. Hernandez-Green, who is also executive director of the Center for Maternal Health Equity at Morehouse School of Medicine. “And we’re about to change that, one person at a time.”
The presence of a doula, along with regular nursing care, is associated with improved labor and delivery outcomes, reduced stress, and higher rates of patient satisfaction, according to the American College of Obstetricians and Gynecologists.
Multiple studies also link doulas to fewer expensive childbirth interventions, including cesarean births.
Doulas are not medical professionals. They are trained to offer education about the pregnancy and postpartum periods, to guide patients through the healthcare system, and to provide emotional and physical support before, during, and after childbirth.
Morehouse School of Medicine’s program is among a growing number of similar efforts being introduced across the country as more communities look to doulas to help address maternal mortality and poor maternal health outcomes, particularly for Black women and other women of color.
Now that she has graduated, Ms. Clark said she’s looking forward to helping other women in her community as a doula. “To be that person that would be there for my clients, treat them like a sister or like a mother, in a sense of just treating them with utmost respect,” she said. “The ultimate goal is to make them feel comfortable and let them know ‘I’m here to support you.’ ” Her training has inspired her to become an advocate for maternal health issues in southwestern Georgia.
Grants fund Morehouse School of Medicine’s doula program, which costs $350,000 a year to operate. Graduates are given a $2,000 training stipend and the program places five graduates with healthcare providers in southwestern Georgia. Grant money also pays the doulas’ salaries for 1 year.
“It’s not sustainable if you’re chasing the next grant to fund it,” said Rachel Hardeman, a professor of health and racial equity at the University of Minnesota School of Public Health.
Thirteen states cover doulas through Medicaid, according to the Georgetown University Center for Children and Families.
Dr. Hardeman and others have found that when Medicaid programs cover doula care, states save millions of dollars in healthcare costs. “We were able to calculate the return on investment if Medicaid decided to reimburse doulas for pregnant people who are Medicaid beneficiaries,” she said.
That’s because doulas can help reduce the number of expensive medical interventions during and after birth, and improving delivery outcomes, including reduced cesarean sections.
Doulas can even reduce the likelihood of preterm birth.
“An infant that is born at a very, very early gestational age is going to require a great deal of resources and interventions to ensure that they survive and then continue to thrive,” Dr. Hardeman said.
There is growing demand for doula services in Georgia, said Fowzio Jama, director of research for Healthy Mothers, Healthy Babies Coalition of Georgia. Her group recently completed a pilot study that offered doula services to about 170 Georgians covered under Medicaid. “We had a wait-list of over 200 clients and we wanted to give them the support that they needed, but we just couldn’t with the given resources that we had,” Ms. Jama said.
Doula services can cost hundreds or thousands of dollars out-of-pocket, making it too expensive for many low-income people, rural communities, and communities of color, many of which suffer from shortages in maternity care, according to the March of Dimes.
The Healthy Mothers, Healthy Babies study found that matching high-risk patients with doulas — particularly doulas from similar racial and ethnic backgrounds — had a positive effect on patients.
“There was a reduced use of pitocin to induce labor. We saw fewer requests for pain medication. And with our infants, only 6% were low birth weight,” Ms. Jama said.
Still, she and others acknowledge that doulas alone can’t fix the problem of high maternal mortality and morbidity rates.
States, including Georgia, need to do more to bring comprehensive maternity care to communities that need more options, Dr. Hardeman said.
“I think it’s important to understand that doulas are not going to save us, and we should not put that expectation on them. Doulas are a tool,” she said. “They are a piece of the puzzle that is helping to impact a really, really complex issue.”
In the meantime, Joan Anderson, 55, said she’s excited to get to work supporting patients, especially from rural areas around Albany.
“I feel like I’m equipped to go out and be that voice, be that person that our community needs so bad,” said Ms. Anderson, a graduate of the Morehouse School of Medicine doula program. “I am encouraged to know that I will be joining in that mission, that fight for us, as far as maternal health is concerned.”
Ms. Anderson said that someday she wants to open a birthing center to provide maternity care. “We do not have one here in southwest Georgia at all,” Ms. Anderson said.
In addition to providing support during and after childbirth, Ms. Anderson and her fellow graduates are trained to assess their patients’ needs and connect them to services such as food assistance, mental health care, transportation to prenatal appointments, and breastfeeding assistance.
Their work is likely to have ripple effects across a largely rural corner of Georgia, said Sherrell Byrd, who co-founded and directs SOWEGA Rising, a nonprofit organization in southwestern Georgia.
“So many of the graduates are part of church networks, they are part of community organizations, some of them are our government workers. They’re very connected,” Ms. Byrd said. “And I think that connectedness is what’s going to help them be successful moving forward.”
This reporting is part of a fellowship with the Association of Health Care Journalists supported by The Commonwealth Fund. It comes from a partnership that includes WABE, NPR, and KFF Health News.
A version of this article first appeared on KFF Health News.
Study Quantifies Benefit of Newborn Screening for Vitamin B12 Deficiency
Newborn screening for neonatal vitamin B12 deficiency may lead to a fourfold reduction in chances of developing symptomatic vitamin B12 in the first year of life compared with infants without newborn screening, a hospital-based surveillance study in Germany indicates.
Vitamin B12 deficiency can impede development in infants, but the true impact of newborn screening versus no screening had not been known in Germany. Early treatment had been shown to be linked with normal development in infants who got newborn screening, but left unclear was how many who had newborn screening would have progressed to symptomatic vitamin B12 deficiency without treatment. Thus formal evidence for the benefit of the screening was lacking.
The nationwide surveillance study, led by Ulrike Mütze, MD, with the Heidelberg University Center for Child and Adolescent Medicine, was published online in Pediatrics. It used prospectively collected data from incident cases of infants under 12 months of age with vitamin B12 deficiency from 2021 to 2022.
The researchers analyzed 61 cases of vitamin B12 deficiency reported to the German Pediatric Surveillance Unit. They were either identified by newborn screening (n = 31) or diagnosed after the onset of suggestive symptoms (non-newborn screening; n = 30).
At a median 4 months of age, the great majority (90%) of the infants identified by newborn screening were still asymptomatic, while the non-newborn screening cohort presented with muscular hypotonia (68%), anemia (58%), developmental delay (44%), microcephalia (30%), and seizures (12%).
Symptomatically diagnosed vitamin B12 deficiency in the baby’s first year was reported four times more frequently in infants who did not receive newborn screening for neonatal vitamin B12 deficiency compared with those screened for vitamin B12 as newborns (Fisher’s Exact Test; odds ratio, 4.12 [95% confidence interval, 1.29-17.18], P = .008).
Clinical presentation of vitamin B12 deficiency in infants usually starts in the first months and reportedly includes, in addition to developmental delay, feeding difficulties, muscular hypotonia and weakness, severe failure to thrive, irritability, lethargy, and (as late symptoms) megaloblastic anemia and brain atrophy.
The current study confirmed these reports and highlighted that the most common presentations in symptomatic infantile vitamin B12 deficiency were muscular hypotonia, anemia, developmental delay, malnutrition or failure to thrive, and microcephalia, brain atrophy, or delayed myelination.
Stephen Walker, MD, a pediatric neurologist at University of Alabama, Birmingham, who was not involved with the study, said newborn screening for vitamin B12 deficiency is routine in the United States.
“In Alabama, we’re generally the last to adopt any of these newborn screenings ... and we’ve been doing it for several years,” he said. Vitamin B12 deficiency is one of 59 conditions included in the state’s newborn blood spot screening. In the United States, he added, when deficiencies are identified, cases are quickly referred to genetic or nutritional specialists.
In the Mütze et al. study, the authors conclude, “The incidence of symptomatic vitamin B12 deficiency accounts for about half of the estimated incidence of the vitamin B12 deficiency identified by newborn screening [NBS]. This supports the notion that not all newborns identified will develop a symptomatic infantile vitamin B12 deficiency but at the same time [this study demonstrates] the high beneficial potential of NBS for vitamin B12 deficiency.”
Dr. Mütze received a research grant from the Medical Faculty of Heidelberg University, Germany. Dr. Kölker, Dr. Hoffmann, and Dr. Mütze received research grants for their work on newborn screening from the Dietmar Hopp Foundation, St. Leon-Rot, Germany. Dr. Hoffmann, Dr. Janzen, and Dr. Röschinger are principal investigators for newborn screening pilot studies including neonatal vitamin B12 deficiency. The other authors have no relevant disclosures. Dr. Walker reports no relevant financial relationships.
Newborn screening for neonatal vitamin B12 deficiency may lead to a fourfold reduction in chances of developing symptomatic vitamin B12 in the first year of life compared with infants without newborn screening, a hospital-based surveillance study in Germany indicates.
Vitamin B12 deficiency can impede development in infants, but the true impact of newborn screening versus no screening had not been known in Germany. Early treatment had been shown to be linked with normal development in infants who got newborn screening, but left unclear was how many who had newborn screening would have progressed to symptomatic vitamin B12 deficiency without treatment. Thus formal evidence for the benefit of the screening was lacking.
The nationwide surveillance study, led by Ulrike Mütze, MD, with the Heidelberg University Center for Child and Adolescent Medicine, was published online in Pediatrics. It used prospectively collected data from incident cases of infants under 12 months of age with vitamin B12 deficiency from 2021 to 2022.
The researchers analyzed 61 cases of vitamin B12 deficiency reported to the German Pediatric Surveillance Unit. They were either identified by newborn screening (n = 31) or diagnosed after the onset of suggestive symptoms (non-newborn screening; n = 30).
At a median 4 months of age, the great majority (90%) of the infants identified by newborn screening were still asymptomatic, while the non-newborn screening cohort presented with muscular hypotonia (68%), anemia (58%), developmental delay (44%), microcephalia (30%), and seizures (12%).
Symptomatically diagnosed vitamin B12 deficiency in the baby’s first year was reported four times more frequently in infants who did not receive newborn screening for neonatal vitamin B12 deficiency compared with those screened for vitamin B12 as newborns (Fisher’s Exact Test; odds ratio, 4.12 [95% confidence interval, 1.29-17.18], P = .008).
Clinical presentation of vitamin B12 deficiency in infants usually starts in the first months and reportedly includes, in addition to developmental delay, feeding difficulties, muscular hypotonia and weakness, severe failure to thrive, irritability, lethargy, and (as late symptoms) megaloblastic anemia and brain atrophy.
The current study confirmed these reports and highlighted that the most common presentations in symptomatic infantile vitamin B12 deficiency were muscular hypotonia, anemia, developmental delay, malnutrition or failure to thrive, and microcephalia, brain atrophy, or delayed myelination.
Stephen Walker, MD, a pediatric neurologist at University of Alabama, Birmingham, who was not involved with the study, said newborn screening for vitamin B12 deficiency is routine in the United States.
“In Alabama, we’re generally the last to adopt any of these newborn screenings ... and we’ve been doing it for several years,” he said. Vitamin B12 deficiency is one of 59 conditions included in the state’s newborn blood spot screening. In the United States, he added, when deficiencies are identified, cases are quickly referred to genetic or nutritional specialists.
In the Mütze et al. study, the authors conclude, “The incidence of symptomatic vitamin B12 deficiency accounts for about half of the estimated incidence of the vitamin B12 deficiency identified by newborn screening [NBS]. This supports the notion that not all newborns identified will develop a symptomatic infantile vitamin B12 deficiency but at the same time [this study demonstrates] the high beneficial potential of NBS for vitamin B12 deficiency.”
Dr. Mütze received a research grant from the Medical Faculty of Heidelberg University, Germany. Dr. Kölker, Dr. Hoffmann, and Dr. Mütze received research grants for their work on newborn screening from the Dietmar Hopp Foundation, St. Leon-Rot, Germany. Dr. Hoffmann, Dr. Janzen, and Dr. Röschinger are principal investigators for newborn screening pilot studies including neonatal vitamin B12 deficiency. The other authors have no relevant disclosures. Dr. Walker reports no relevant financial relationships.
Newborn screening for neonatal vitamin B12 deficiency may lead to a fourfold reduction in chances of developing symptomatic vitamin B12 in the first year of life compared with infants without newborn screening, a hospital-based surveillance study in Germany indicates.
Vitamin B12 deficiency can impede development in infants, but the true impact of newborn screening versus no screening had not been known in Germany. Early treatment had been shown to be linked with normal development in infants who got newborn screening, but left unclear was how many who had newborn screening would have progressed to symptomatic vitamin B12 deficiency without treatment. Thus formal evidence for the benefit of the screening was lacking.
The nationwide surveillance study, led by Ulrike Mütze, MD, with the Heidelberg University Center for Child and Adolescent Medicine, was published online in Pediatrics. It used prospectively collected data from incident cases of infants under 12 months of age with vitamin B12 deficiency from 2021 to 2022.
The researchers analyzed 61 cases of vitamin B12 deficiency reported to the German Pediatric Surveillance Unit. They were either identified by newborn screening (n = 31) or diagnosed after the onset of suggestive symptoms (non-newborn screening; n = 30).
At a median 4 months of age, the great majority (90%) of the infants identified by newborn screening were still asymptomatic, while the non-newborn screening cohort presented with muscular hypotonia (68%), anemia (58%), developmental delay (44%), microcephalia (30%), and seizures (12%).
Symptomatically diagnosed vitamin B12 deficiency in the baby’s first year was reported four times more frequently in infants who did not receive newborn screening for neonatal vitamin B12 deficiency compared with those screened for vitamin B12 as newborns (Fisher’s Exact Test; odds ratio, 4.12 [95% confidence interval, 1.29-17.18], P = .008).
Clinical presentation of vitamin B12 deficiency in infants usually starts in the first months and reportedly includes, in addition to developmental delay, feeding difficulties, muscular hypotonia and weakness, severe failure to thrive, irritability, lethargy, and (as late symptoms) megaloblastic anemia and brain atrophy.
The current study confirmed these reports and highlighted that the most common presentations in symptomatic infantile vitamin B12 deficiency were muscular hypotonia, anemia, developmental delay, malnutrition or failure to thrive, and microcephalia, brain atrophy, or delayed myelination.
Stephen Walker, MD, a pediatric neurologist at University of Alabama, Birmingham, who was not involved with the study, said newborn screening for vitamin B12 deficiency is routine in the United States.
“In Alabama, we’re generally the last to adopt any of these newborn screenings ... and we’ve been doing it for several years,” he said. Vitamin B12 deficiency is one of 59 conditions included in the state’s newborn blood spot screening. In the United States, he added, when deficiencies are identified, cases are quickly referred to genetic or nutritional specialists.
In the Mütze et al. study, the authors conclude, “The incidence of symptomatic vitamin B12 deficiency accounts for about half of the estimated incidence of the vitamin B12 deficiency identified by newborn screening [NBS]. This supports the notion that not all newborns identified will develop a symptomatic infantile vitamin B12 deficiency but at the same time [this study demonstrates] the high beneficial potential of NBS for vitamin B12 deficiency.”
Dr. Mütze received a research grant from the Medical Faculty of Heidelberg University, Germany. Dr. Kölker, Dr. Hoffmann, and Dr. Mütze received research grants for their work on newborn screening from the Dietmar Hopp Foundation, St. Leon-Rot, Germany. Dr. Hoffmann, Dr. Janzen, and Dr. Röschinger are principal investigators for newborn screening pilot studies including neonatal vitamin B12 deficiency. The other authors have no relevant disclosures. Dr. Walker reports no relevant financial relationships.
FROM PEDIATRICS
Advanced Tissue Resection in Gastroenterology: Indications, Role, and Outcomes
Endoscopists are often faced with unique challenges in the management and resection of various gastrointestinal tract lesions. These challenges could be lesion-related, endoscopist-related, or practice-related (see Table 1). (ATR). Not only does this organ-sparing approach offer a less invasive alternative to surgery, but it has also proved to have outcomes comparable to those of surgical standard of practice in specific scenarios. 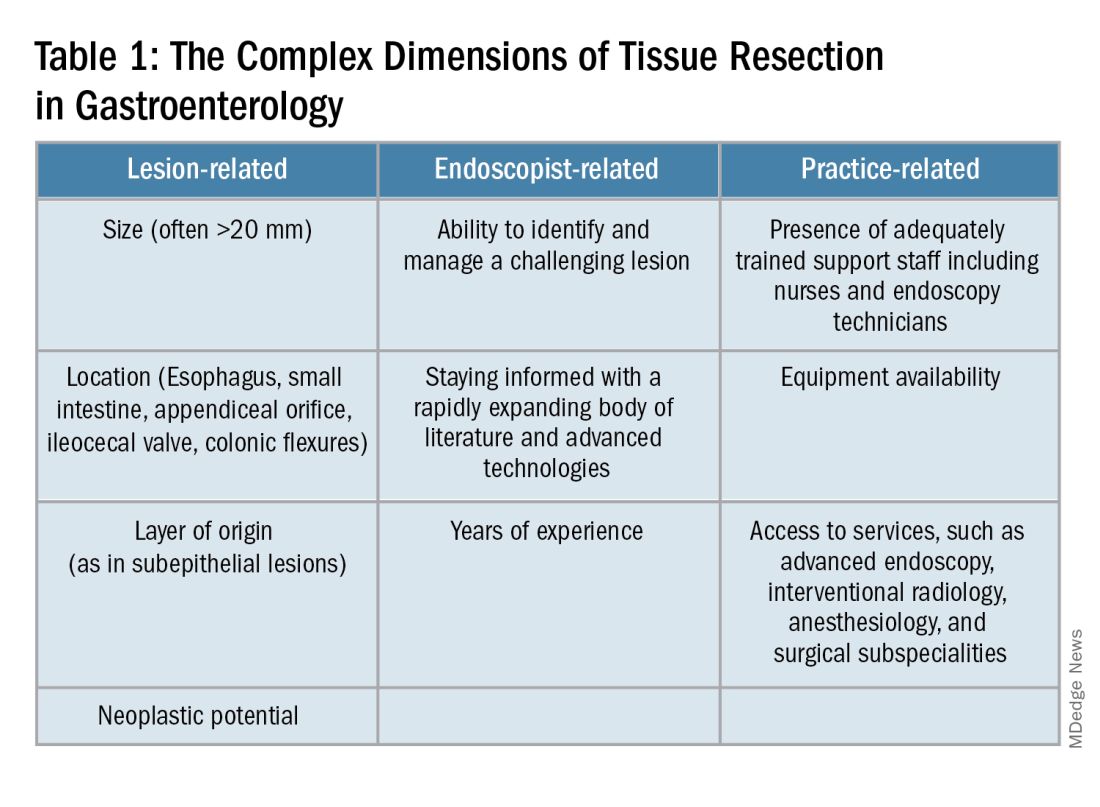
When Do You Refer to an Advanced Endoscopist?
One of the most critical steps in caring for patients with complex lesions is the ability to accurately determine whether a referral to an advanced endoscopist is warranted. The initial assessment of a lesion should always involve a careful assessment that risk stratifies the lesion depending on the location, size, neoplastic potential, and the feasibility of standard endoscopic resection compared to the need for surgical input.
A practical example in the case of colonic polyps is highlighted by the American Gastroenterology Association (AGA) guidelines recommending the referral of patients with polyps’ size ≥ 20 mm, challenging polypectomy location, or recurrent polyp at a prior polypectomy site to an endoscopic referral center.1 In the case of subepithelial lesions without endoscopic characteristics of benign etiology (i.e., lipomas, pancreatic rests, etc.), the threshold for referral to advanced endoscopists for further diagnostic testing by means of endoscopic ultrasonography or for therapeutic ATR should be lower.
Endoscopic tissue resection follows a spectrum, which often involves deeper layers of the gastrointestinal tract (GIT) as we progress along this spectrum (see Figure 1).
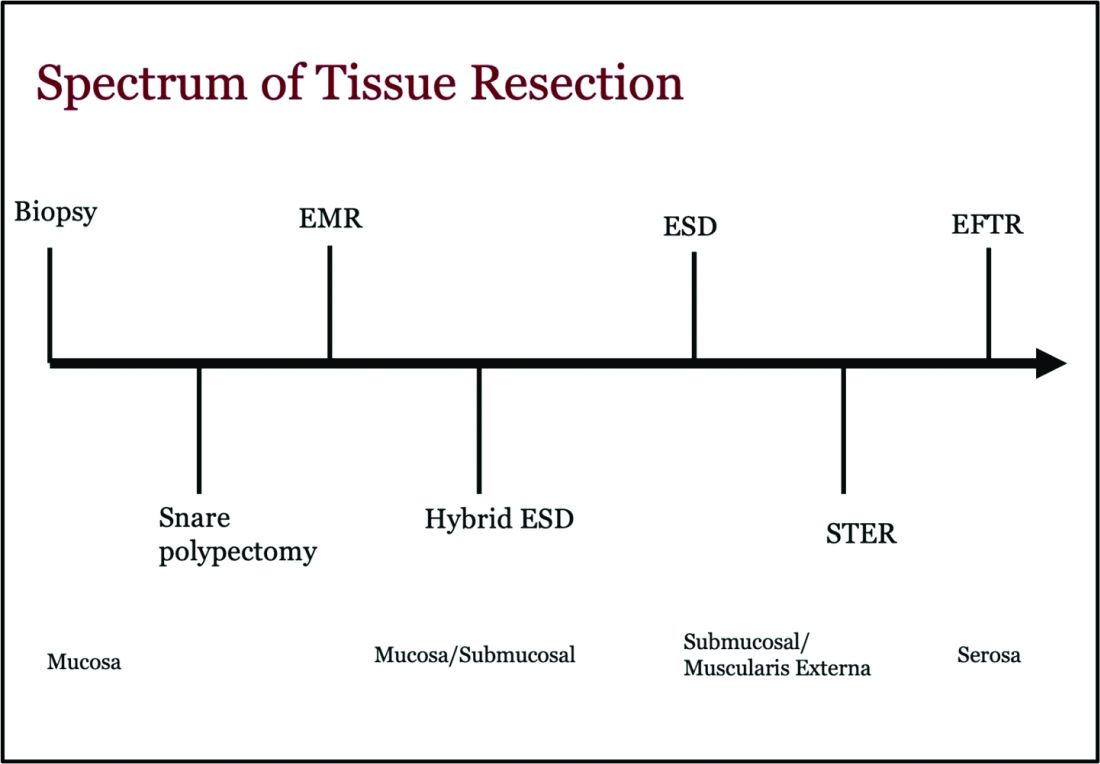
ATR, a term encompassing a variety of endoscopic techniques ranging from endoscopic mucosal resection to full thickness resection, has gained traction over the last years given the ability to effectively remove various lesions in a precise time and cost-effective manner while maintaining the integrity of the GIT and avoiding major surgery. The indications for ATR vary depending on the technique, but generally include the presence of large or poorly positioned lesions, particularly in high-risk areas of the GIT such as the esophagus and small intestine, lesions extending beyond the mucosal layer or originating from deeper layers, and when en bloc resection of select lesions is necessary.
For providers referring patients for ATR, we recommend a few important endoscopic pearls when caring for these patients.
1) Biopsy the lesion if there is concern for malignancy — While some studies have noted increased fibrosis during endoscopic submucosal dissection (ESD) and some guidelines recommend against biopsies pre ESD, we believe that when there is high pretest probability for malignancy, a biopsy should be obtained. This should involve the area that is most concerning for malignancy (at the margin or center).2
2) While marking a lesion with tattoo is helpful for surgical planning and for lesions difficult to locate endoscopically, we stress the importance of placing tattoos 3 to 5 centimeters distal to the lesion and avoiding tattooing the lesion itself, which has been shown to induce fibrosis and can make resection challenging. Based on an international Delphi consensus, expert recommendations on when and how to endoscopically tattoo a lesion can be instrumental in adequately localizing the lesion, allowing for endoscopic resection, and preventing unnecessary surgeries.3
3) If you encounter a lesion that you are not sure can be resected safely and efficaciously, we recommend against attempting resection that may result in partial resection. This can also induce fibrosis and scarring and limit future attempts at resection.
Endoscopic Mucosal Resection (EMR)
EMR is currently utilized for curative treatment of a wide array of GIT lesions limited to the mucosal layer, whether metaplastic, dysplastic, or even in cases with early mucosal cancer, where the risk of submucosal and lymphatic invasion is minimal.4 This makes EMR a versatile and proven therapy, often serving as the first-line treatment for many GIT lesions.
EMR has various techniques that could be categorized into suction or non-suction (lift and cut) techniques. In the suction technique, devices like multiband mucosectomy (MBM) are commonly used, especially in nodular Barrett’s dysplasia, forming a pseudopolyp for subsequent resection. The procedure is characterized by its safety, efficacy, and cost-effectiveness, contributing to its widespread adoption in clinical practice. In the lift and cut approach, a submucosal injection is utilized to separate the muscularis propria from the lesion, thereby reducing the risk of perforation. Different solutions, such as normal saline, hypertonic saline, 50% dextrose, or proprietary submucosal injection solutions, are employed for submucosal injection.5
The non-suction technique using a snare to resect polyps after injection is more often used in colonic and small intestinal EMR. Resection can be done via thermal energy in the form of cut or coagulation; however, there is rising data on the use of piecemeal cold snare resection for select flat polyps of the colon.6 There is also promising data on the role of underwater EMR, a common technique employed for colonic lesions, particularly if the lesion does not lift well with submucosal injection.7
Adverse events associated with EMR include bleeding (7%-8%) and perforation (0.9%-2%).8-9 Adequate submucosal fluid injection is crucial to prevent perforations. However, the main limitation of EMR is the piecemeal nature of resections for lesions larger than 20 mm, leading to compromised histopathologic evaluation for complete excision, especially in cases with superficial submucosal invasion (SMI). This can result in residual or recurrent tissue, reportedly 8% to 20%.10 Despite this limitation, EMR remains a reliable strategy, and recurrent lesions are generally manageable through repeat sessions. The importance of EMR as a therapeutic modality lies in its role in addressing lesions with favorable characteristics, where the risk of SMI is low.
Endoscopic Submucosal Dissection (ESD)
ESD is an evolving technique that can be utilized for submucosal lesions of the GIT, lesions not amenable to EMR due to submucosal fibrosis, when en bloc removal of a lesion is needed for accurate histopathological diagnosis, and when other techniques fail.11-12
ESD was only recently adopted in the United States, requires specialized training, and usually is a lengthier procedure than EMR.13 Compared to EMR, it has higher en bloc resection rates and lower recurrence rates, making it curative for lesions with superficial SMI and favorable histologic features.4,14 The safety profile of ESD appears favorable, with most of the adverse events managed successfully by endoscopic methods. Major complications include intraoperative and delayed perforation, intraoperative and delayed bleeding, aspiration pneumonia, thromboembolism, and stricture formation in the case of circumferential lesions.15
Despite being technically challenging, ESD may provide a cost-effective long-term solution by avoiding surgery, reducing the need for additional interventions by minimizing recurrence rates. Given the technical complexity of ESD, particularly the submucosal dissection portion, techniques such as hybrid ESD developed. Hybrid ESD combines snaring with circumferential mucosal incision and partial submucosal dissection. Although it promises shorter procedure times, reduced complication rates like perforation, and similar recurrence rates compared to traditional ESD, studies have shown lower success rates in en bloc resection.16-17
Both EMR and ESD are considered complementary strategies, and the choice between them should be dictated by lesion characteristics, patient preferences, and local expertise.
Submucosal Tunneling Endoscopic Resection (STER)
STER has emerged as a well-established technique for the endoscopic resection of GI subepithelial tumors (SETs) originating from the muscularis propria layer. The standard STER procedure involves a series of steps including submucosal elevation proximal to the SET, mucosotomy, creation of a submucosal tunnel, dissection of the SET within the tunnel, enucleation from the deep muscle layer, and subsequent specimen retrieval followed by mucosal closure.
This technique is typically recommended for SETs smaller than 3.5 cm, particularly those located in the mid or distal esophagus, cardia, or along the greater curvature of the gastric body.18 However, STER may pose technical challenges for larger SETs or lesions in anatomically difficult locations, where surgical resection is recommended instead.19 Notably, recent large-scale meta-analyses have showcased the favorable complete resection and en bloc resection rates of STER in treating GI SETs.20
Endoscopic Full Thickness Resection (EFTR)
EFTR has emerged as a valuable technique in the endoscopic management of gastrointestinal lesions, particularly SETs and lesions not amenable to EMR or ESD due to fibrosis. EFTR involves the resection of all layers of the GIT from mucosa to serosa, and therefore is well-suited for SETs arising from the muscularis propria (MP).20
EFTR entails two main concepts: tissue resection and complete defect closure. Conventional EFTR consists of several steps, which include mucosal and submucosal pre-cutting, circumferential incision, and dissection through the MP or serosa. This results in a full thickness defect, for which closure of the wall defect is achieved using standard endoscopic clips or a combination of clips and endoloops or endoscopic suturing.21 For lesions less than 2 cm, EFTR can be performed in a single step using a cap-mounted full thickness resection device (FTRD). This results in deployment of over-the-scope clip over the target lesion followed by snaring the lesions above the clip.21
Location of the SET generally dictates the specific modality of ATR. For example, esophageal SETs may be more amenable to STER given that the lesion typically runs parallel with the lumen of the tubular esophagus, which allows for easier dissection without the need of full or partial retroflexion. While gastric SETs can be resected with STER, it may be challenging and more effectively addressed with EFTR, particularly when the entire lesion can be grasped into the full-thickness resection device.22 Limited data exists for duodenal EFTR, and colorectal SETs closure is particularly challenging.
Conclusion
It is key to emphasize that ATR cannot be safely established in practice without the incorporation of a multidisciplinary team (surgeons, radiologists, etc.), specialized tools, and trained personnel. This requires dedicated endoscopic rooms, careful patient selection, and a comprehensive approach to patient care before, during, and after these procedures.
Moreover, it is important to note that some patients may require post-procedure hospitalization for observation to ensure no early complications are encountered. Optimal surveillance strategies after ATR rely heavily on the potential for residual or recurrent disease, underlying pathology, and the expertise of the advanced endoscopist. As the field continues to evolve, ongoing research and technological advances of devices will further enhance the efficacy and safety of ATR in gastroenterology.
Dr. Madi (@MahMadi90) is based in the Division of Gastroenterology and Hepatology, Saint Louis University School of Medicine, Saint Louis, Missouri. Dr. Rengarajan (@ArvindRenga) and Dr. Bazarbashi (@AhmadBazarbashi) are based in the Division of Gastroenterology, Washington University in St. Louis. The authors have no conflicts of interest to disclose, and no funding was required for this project.
References
1. Copland AP, et al. AGA Clinical Practice Update on appropriate and tailored polypectomy: Expert review. Clin Gastroenterol Hepatol. 2024 Mar. doi: 10.1016/j.cgh.2023.10.012.
2. Lee SP, et al. Effect of preceding biopsy on the results of endoscopic submucosal dissection for colorectal laterally spreading tumor. Dig Dis Sci. 2019 Oct. doi: 10.1007/s10620-019-05625-3.
3. Medina-Prado L, et al. When and how to use endoscopic tattooing in the colon: An international Delphi agreement. Clin Gastroenterol Hepatol. 2021 May. doi: 10.1016/j.cgh.2021.01.024.
4. Rashid MU, et al. EMR and ESD: Indications, techniques and results. Surg Oncol. 2022 Aug. doi: 10.1016/j.suronc.2022.101742.
5. Castro R, et al. Solutions for submucosal injection: What to choose and how to do it. World J Gastroenterol. 2019 Feb. doi: 10.3748/wjg.v25.i7.777.
6. Rex DK. Best practices for resection of diminutive and small polyps in the colorectum. Gastrointest Endosc Clin N Am. 2019 Oct. doi: 10.1016/j.giec.2019.06.004.
7. Lv XH, et al. Underwater EMR for nonpedunculated colorectal lesions. Gastrointest Endosc. 2023 Apr. doi: 10.1016/j.gie.2022.10.044.
8. Fujiya M, et al. Efficacy and adverse events of EMR and endoscopic submucosal dissection for the treatment of colon neoplasms: a meta-analysis of studies comparing EMR and endoscopic submucosal dissection. Gastrointest Endosc. 2015 Mar. doi: 10.1016/j.gie.2014.07.034.
9. Kandel P, Wallace MB. Colorectal endoscopic mucosal resection (EMR). Best Pract Res Clin Gastroenterol. 2017 Aug. doi: 10.1016/j.bpg.2017.05.006.
10. Kemper G, et al; ENDOCARE Study Group. Endoscopic techniques to reduce recurrence rates after colorectal EMR: systematic review and meta-analysis. Surg Endosc. 2021 Oct. doi: 10.1007/s00464-021-08574-z.
11. Goto O, et al. Expanding indications for ESD: submucosal disease (SMT/carcinoid tumors). Gastrointest Endosc Clin N Am. 2014 Apr. doi: 10.1016/j.giec.2013.11.006.
12. Wang K, et al. Endoscopic full-thickness resection, indication, methods and perspectives. Dig Endosc. 2023 Jan. doi: 10.1111/den.14474.
13. Herreros de Tejada A. ESD training: A challenging path to excellence. World J Gastrointest Endosc. 2014 Apr 16. doi: 10.4253/wjge.v6.i4.112.
14. Chiba H, et al. Safety and efficacy of simultaneous colorectal ESD for large synchronous colorectal lesions. Endosc Int Open. 2017 Jul. doi: 10.1055/s-0043-110567.
15. Mannath J, Ragunath K. Endoscopic mucosal resection: who and how? Therap Adv Gastroenterol. 2011 Sep. doi: 10.1177/1756283X10388683.
16. Wang XY, et al. Hybrid endoscopic submucosal dissection: An alternative resection modality for large laterally spreading tumors in the cecum? BMC Gastroenterol. 2021 May. doi: 10.1186/s12876-021-01766-w.
17. McCarty TR, et al. Hybrid endoscopic submucosal dissection (ESD) compared with conventional ESD for colorectal lesions: a systematic review and meta-analysis. Endoscopy. 2021 Oct. doi: 10.1055/a-1266-1855.
18. Jain D, et al. Submucosal tunneling endoscopic resection of upper gastrointestinal tract tumors arising from muscularis propria. Ann Gastroenterol. 2017 Feb. doi: 10.20524/aog.2017.0128.
19. Lv XH, et al. Efficacy and safety of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors: a systematic review and meta-analysis. Surg Endosc. 2017 Jan. doi: 10.1007/s00464-016-4978-7.
20. Cao B, et al. Efficacy and safety of submucosal tunneling endoscopic resection for gastric submucosal tumors: a systematic review and meta-analysis. Rev Esp Enferm Dig. 2021 Jan. doi: 10.17235/reed.2020.6989/2020.
21. Cai M, et al. Endoscopic full-thickness resection (EFTR) for gastrointestinal subepithelial tumors. Gastrointest Endosc Clin N Am. 2016 Apr. doi: 10.1016/j.giec.2015.12.013.
22. Brigic A, et al. A systematic review regarding the feasibility and safety of endoscopic full thickness resection (EFTR) for colonic lesions. Surg Endosc. 2013 Oct. doi: 10.1007/s00464-013-2946-z.
Endoscopists are often faced with unique challenges in the management and resection of various gastrointestinal tract lesions. These challenges could be lesion-related, endoscopist-related, or practice-related (see Table 1). (ATR). Not only does this organ-sparing approach offer a less invasive alternative to surgery, but it has also proved to have outcomes comparable to those of surgical standard of practice in specific scenarios. 
When Do You Refer to an Advanced Endoscopist?
One of the most critical steps in caring for patients with complex lesions is the ability to accurately determine whether a referral to an advanced endoscopist is warranted. The initial assessment of a lesion should always involve a careful assessment that risk stratifies the lesion depending on the location, size, neoplastic potential, and the feasibility of standard endoscopic resection compared to the need for surgical input.
A practical example in the case of colonic polyps is highlighted by the American Gastroenterology Association (AGA) guidelines recommending the referral of patients with polyps’ size ≥ 20 mm, challenging polypectomy location, or recurrent polyp at a prior polypectomy site to an endoscopic referral center.1 In the case of subepithelial lesions without endoscopic characteristics of benign etiology (i.e., lipomas, pancreatic rests, etc.), the threshold for referral to advanced endoscopists for further diagnostic testing by means of endoscopic ultrasonography or for therapeutic ATR should be lower.
Endoscopic tissue resection follows a spectrum, which often involves deeper layers of the gastrointestinal tract (GIT) as we progress along this spectrum (see Figure 1).

ATR, a term encompassing a variety of endoscopic techniques ranging from endoscopic mucosal resection to full thickness resection, has gained traction over the last years given the ability to effectively remove various lesions in a precise time and cost-effective manner while maintaining the integrity of the GIT and avoiding major surgery. The indications for ATR vary depending on the technique, but generally include the presence of large or poorly positioned lesions, particularly in high-risk areas of the GIT such as the esophagus and small intestine, lesions extending beyond the mucosal layer or originating from deeper layers, and when en bloc resection of select lesions is necessary.
For providers referring patients for ATR, we recommend a few important endoscopic pearls when caring for these patients.
1) Biopsy the lesion if there is concern for malignancy — While some studies have noted increased fibrosis during endoscopic submucosal dissection (ESD) and some guidelines recommend against biopsies pre ESD, we believe that when there is high pretest probability for malignancy, a biopsy should be obtained. This should involve the area that is most concerning for malignancy (at the margin or center).2
2) While marking a lesion with tattoo is helpful for surgical planning and for lesions difficult to locate endoscopically, we stress the importance of placing tattoos 3 to 5 centimeters distal to the lesion and avoiding tattooing the lesion itself, which has been shown to induce fibrosis and can make resection challenging. Based on an international Delphi consensus, expert recommendations on when and how to endoscopically tattoo a lesion can be instrumental in adequately localizing the lesion, allowing for endoscopic resection, and preventing unnecessary surgeries.3
3) If you encounter a lesion that you are not sure can be resected safely and efficaciously, we recommend against attempting resection that may result in partial resection. This can also induce fibrosis and scarring and limit future attempts at resection.
Endoscopic Mucosal Resection (EMR)
EMR is currently utilized for curative treatment of a wide array of GIT lesions limited to the mucosal layer, whether metaplastic, dysplastic, or even in cases with early mucosal cancer, where the risk of submucosal and lymphatic invasion is minimal.4 This makes EMR a versatile and proven therapy, often serving as the first-line treatment for many GIT lesions.
EMR has various techniques that could be categorized into suction or non-suction (lift and cut) techniques. In the suction technique, devices like multiband mucosectomy (MBM) are commonly used, especially in nodular Barrett’s dysplasia, forming a pseudopolyp for subsequent resection. The procedure is characterized by its safety, efficacy, and cost-effectiveness, contributing to its widespread adoption in clinical practice. In the lift and cut approach, a submucosal injection is utilized to separate the muscularis propria from the lesion, thereby reducing the risk of perforation. Different solutions, such as normal saline, hypertonic saline, 50% dextrose, or proprietary submucosal injection solutions, are employed for submucosal injection.5
The non-suction technique using a snare to resect polyps after injection is more often used in colonic and small intestinal EMR. Resection can be done via thermal energy in the form of cut or coagulation; however, there is rising data on the use of piecemeal cold snare resection for select flat polyps of the colon.6 There is also promising data on the role of underwater EMR, a common technique employed for colonic lesions, particularly if the lesion does not lift well with submucosal injection.7
Adverse events associated with EMR include bleeding (7%-8%) and perforation (0.9%-2%).8-9 Adequate submucosal fluid injection is crucial to prevent perforations. However, the main limitation of EMR is the piecemeal nature of resections for lesions larger than 20 mm, leading to compromised histopathologic evaluation for complete excision, especially in cases with superficial submucosal invasion (SMI). This can result in residual or recurrent tissue, reportedly 8% to 20%.10 Despite this limitation, EMR remains a reliable strategy, and recurrent lesions are generally manageable through repeat sessions. The importance of EMR as a therapeutic modality lies in its role in addressing lesions with favorable characteristics, where the risk of SMI is low.
Endoscopic Submucosal Dissection (ESD)
ESD is an evolving technique that can be utilized for submucosal lesions of the GIT, lesions not amenable to EMR due to submucosal fibrosis, when en bloc removal of a lesion is needed for accurate histopathological diagnosis, and when other techniques fail.11-12
ESD was only recently adopted in the United States, requires specialized training, and usually is a lengthier procedure than EMR.13 Compared to EMR, it has higher en bloc resection rates and lower recurrence rates, making it curative for lesions with superficial SMI and favorable histologic features.4,14 The safety profile of ESD appears favorable, with most of the adverse events managed successfully by endoscopic methods. Major complications include intraoperative and delayed perforation, intraoperative and delayed bleeding, aspiration pneumonia, thromboembolism, and stricture formation in the case of circumferential lesions.15
Despite being technically challenging, ESD may provide a cost-effective long-term solution by avoiding surgery, reducing the need for additional interventions by minimizing recurrence rates. Given the technical complexity of ESD, particularly the submucosal dissection portion, techniques such as hybrid ESD developed. Hybrid ESD combines snaring with circumferential mucosal incision and partial submucosal dissection. Although it promises shorter procedure times, reduced complication rates like perforation, and similar recurrence rates compared to traditional ESD, studies have shown lower success rates in en bloc resection.16-17
Both EMR and ESD are considered complementary strategies, and the choice between them should be dictated by lesion characteristics, patient preferences, and local expertise.
Submucosal Tunneling Endoscopic Resection (STER)
STER has emerged as a well-established technique for the endoscopic resection of GI subepithelial tumors (SETs) originating from the muscularis propria layer. The standard STER procedure involves a series of steps including submucosal elevation proximal to the SET, mucosotomy, creation of a submucosal tunnel, dissection of the SET within the tunnel, enucleation from the deep muscle layer, and subsequent specimen retrieval followed by mucosal closure.
This technique is typically recommended for SETs smaller than 3.5 cm, particularly those located in the mid or distal esophagus, cardia, or along the greater curvature of the gastric body.18 However, STER may pose technical challenges for larger SETs or lesions in anatomically difficult locations, where surgical resection is recommended instead.19 Notably, recent large-scale meta-analyses have showcased the favorable complete resection and en bloc resection rates of STER in treating GI SETs.20
Endoscopic Full Thickness Resection (EFTR)
EFTR has emerged as a valuable technique in the endoscopic management of gastrointestinal lesions, particularly SETs and lesions not amenable to EMR or ESD due to fibrosis. EFTR involves the resection of all layers of the GIT from mucosa to serosa, and therefore is well-suited for SETs arising from the muscularis propria (MP).20
EFTR entails two main concepts: tissue resection and complete defect closure. Conventional EFTR consists of several steps, which include mucosal and submucosal pre-cutting, circumferential incision, and dissection through the MP or serosa. This results in a full thickness defect, for which closure of the wall defect is achieved using standard endoscopic clips or a combination of clips and endoloops or endoscopic suturing.21 For lesions less than 2 cm, EFTR can be performed in a single step using a cap-mounted full thickness resection device (FTRD). This results in deployment of over-the-scope clip over the target lesion followed by snaring the lesions above the clip.21
Location of the SET generally dictates the specific modality of ATR. For example, esophageal SETs may be more amenable to STER given that the lesion typically runs parallel with the lumen of the tubular esophagus, which allows for easier dissection without the need of full or partial retroflexion. While gastric SETs can be resected with STER, it may be challenging and more effectively addressed with EFTR, particularly when the entire lesion can be grasped into the full-thickness resection device.22 Limited data exists for duodenal EFTR, and colorectal SETs closure is particularly challenging.
Conclusion
It is key to emphasize that ATR cannot be safely established in practice without the incorporation of a multidisciplinary team (surgeons, radiologists, etc.), specialized tools, and trained personnel. This requires dedicated endoscopic rooms, careful patient selection, and a comprehensive approach to patient care before, during, and after these procedures.
Moreover, it is important to note that some patients may require post-procedure hospitalization for observation to ensure no early complications are encountered. Optimal surveillance strategies after ATR rely heavily on the potential for residual or recurrent disease, underlying pathology, and the expertise of the advanced endoscopist. As the field continues to evolve, ongoing research and technological advances of devices will further enhance the efficacy and safety of ATR in gastroenterology.
Dr. Madi (@MahMadi90) is based in the Division of Gastroenterology and Hepatology, Saint Louis University School of Medicine, Saint Louis, Missouri. Dr. Rengarajan (@ArvindRenga) and Dr. Bazarbashi (@AhmadBazarbashi) are based in the Division of Gastroenterology, Washington University in St. Louis. The authors have no conflicts of interest to disclose, and no funding was required for this project.
References
1. Copland AP, et al. AGA Clinical Practice Update on appropriate and tailored polypectomy: Expert review. Clin Gastroenterol Hepatol. 2024 Mar. doi: 10.1016/j.cgh.2023.10.012.
2. Lee SP, et al. Effect of preceding biopsy on the results of endoscopic submucosal dissection for colorectal laterally spreading tumor. Dig Dis Sci. 2019 Oct. doi: 10.1007/s10620-019-05625-3.
3. Medina-Prado L, et al. When and how to use endoscopic tattooing in the colon: An international Delphi agreement. Clin Gastroenterol Hepatol. 2021 May. doi: 10.1016/j.cgh.2021.01.024.
4. Rashid MU, et al. EMR and ESD: Indications, techniques and results. Surg Oncol. 2022 Aug. doi: 10.1016/j.suronc.2022.101742.
5. Castro R, et al. Solutions for submucosal injection: What to choose and how to do it. World J Gastroenterol. 2019 Feb. doi: 10.3748/wjg.v25.i7.777.
6. Rex DK. Best practices for resection of diminutive and small polyps in the colorectum. Gastrointest Endosc Clin N Am. 2019 Oct. doi: 10.1016/j.giec.2019.06.004.
7. Lv XH, et al. Underwater EMR for nonpedunculated colorectal lesions. Gastrointest Endosc. 2023 Apr. doi: 10.1016/j.gie.2022.10.044.
8. Fujiya M, et al. Efficacy and adverse events of EMR and endoscopic submucosal dissection for the treatment of colon neoplasms: a meta-analysis of studies comparing EMR and endoscopic submucosal dissection. Gastrointest Endosc. 2015 Mar. doi: 10.1016/j.gie.2014.07.034.
9. Kandel P, Wallace MB. Colorectal endoscopic mucosal resection (EMR). Best Pract Res Clin Gastroenterol. 2017 Aug. doi: 10.1016/j.bpg.2017.05.006.
10. Kemper G, et al; ENDOCARE Study Group. Endoscopic techniques to reduce recurrence rates after colorectal EMR: systematic review and meta-analysis. Surg Endosc. 2021 Oct. doi: 10.1007/s00464-021-08574-z.
11. Goto O, et al. Expanding indications for ESD: submucosal disease (SMT/carcinoid tumors). Gastrointest Endosc Clin N Am. 2014 Apr. doi: 10.1016/j.giec.2013.11.006.
12. Wang K, et al. Endoscopic full-thickness resection, indication, methods and perspectives. Dig Endosc. 2023 Jan. doi: 10.1111/den.14474.
13. Herreros de Tejada A. ESD training: A challenging path to excellence. World J Gastrointest Endosc. 2014 Apr 16. doi: 10.4253/wjge.v6.i4.112.
14. Chiba H, et al. Safety and efficacy of simultaneous colorectal ESD for large synchronous colorectal lesions. Endosc Int Open. 2017 Jul. doi: 10.1055/s-0043-110567.
15. Mannath J, Ragunath K. Endoscopic mucosal resection: who and how? Therap Adv Gastroenterol. 2011 Sep. doi: 10.1177/1756283X10388683.
16. Wang XY, et al. Hybrid endoscopic submucosal dissection: An alternative resection modality for large laterally spreading tumors in the cecum? BMC Gastroenterol. 2021 May. doi: 10.1186/s12876-021-01766-w.
17. McCarty TR, et al. Hybrid endoscopic submucosal dissection (ESD) compared with conventional ESD for colorectal lesions: a systematic review and meta-analysis. Endoscopy. 2021 Oct. doi: 10.1055/a-1266-1855.
18. Jain D, et al. Submucosal tunneling endoscopic resection of upper gastrointestinal tract tumors arising from muscularis propria. Ann Gastroenterol. 2017 Feb. doi: 10.20524/aog.2017.0128.
19. Lv XH, et al. Efficacy and safety of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors: a systematic review and meta-analysis. Surg Endosc. 2017 Jan. doi: 10.1007/s00464-016-4978-7.
20. Cao B, et al. Efficacy and safety of submucosal tunneling endoscopic resection for gastric submucosal tumors: a systematic review and meta-analysis. Rev Esp Enferm Dig. 2021 Jan. doi: 10.17235/reed.2020.6989/2020.
21. Cai M, et al. Endoscopic full-thickness resection (EFTR) for gastrointestinal subepithelial tumors. Gastrointest Endosc Clin N Am. 2016 Apr. doi: 10.1016/j.giec.2015.12.013.
22. Brigic A, et al. A systematic review regarding the feasibility and safety of endoscopic full thickness resection (EFTR) for colonic lesions. Surg Endosc. 2013 Oct. doi: 10.1007/s00464-013-2946-z.
Endoscopists are often faced with unique challenges in the management and resection of various gastrointestinal tract lesions. These challenges could be lesion-related, endoscopist-related, or practice-related (see Table 1). (ATR). Not only does this organ-sparing approach offer a less invasive alternative to surgery, but it has also proved to have outcomes comparable to those of surgical standard of practice in specific scenarios. 
When Do You Refer to an Advanced Endoscopist?
One of the most critical steps in caring for patients with complex lesions is the ability to accurately determine whether a referral to an advanced endoscopist is warranted. The initial assessment of a lesion should always involve a careful assessment that risk stratifies the lesion depending on the location, size, neoplastic potential, and the feasibility of standard endoscopic resection compared to the need for surgical input.
A practical example in the case of colonic polyps is highlighted by the American Gastroenterology Association (AGA) guidelines recommending the referral of patients with polyps’ size ≥ 20 mm, challenging polypectomy location, or recurrent polyp at a prior polypectomy site to an endoscopic referral center.1 In the case of subepithelial lesions without endoscopic characteristics of benign etiology (i.e., lipomas, pancreatic rests, etc.), the threshold for referral to advanced endoscopists for further diagnostic testing by means of endoscopic ultrasonography or for therapeutic ATR should be lower.
Endoscopic tissue resection follows a spectrum, which often involves deeper layers of the gastrointestinal tract (GIT) as we progress along this spectrum (see Figure 1).

ATR, a term encompassing a variety of endoscopic techniques ranging from endoscopic mucosal resection to full thickness resection, has gained traction over the last years given the ability to effectively remove various lesions in a precise time and cost-effective manner while maintaining the integrity of the GIT and avoiding major surgery. The indications for ATR vary depending on the technique, but generally include the presence of large or poorly positioned lesions, particularly in high-risk areas of the GIT such as the esophagus and small intestine, lesions extending beyond the mucosal layer or originating from deeper layers, and when en bloc resection of select lesions is necessary.
For providers referring patients for ATR, we recommend a few important endoscopic pearls when caring for these patients.
1) Biopsy the lesion if there is concern for malignancy — While some studies have noted increased fibrosis during endoscopic submucosal dissection (ESD) and some guidelines recommend against biopsies pre ESD, we believe that when there is high pretest probability for malignancy, a biopsy should be obtained. This should involve the area that is most concerning for malignancy (at the margin or center).2
2) While marking a lesion with tattoo is helpful for surgical planning and for lesions difficult to locate endoscopically, we stress the importance of placing tattoos 3 to 5 centimeters distal to the lesion and avoiding tattooing the lesion itself, which has been shown to induce fibrosis and can make resection challenging. Based on an international Delphi consensus, expert recommendations on when and how to endoscopically tattoo a lesion can be instrumental in adequately localizing the lesion, allowing for endoscopic resection, and preventing unnecessary surgeries.3
3) If you encounter a lesion that you are not sure can be resected safely and efficaciously, we recommend against attempting resection that may result in partial resection. This can also induce fibrosis and scarring and limit future attempts at resection.
Endoscopic Mucosal Resection (EMR)
EMR is currently utilized for curative treatment of a wide array of GIT lesions limited to the mucosal layer, whether metaplastic, dysplastic, or even in cases with early mucosal cancer, where the risk of submucosal and lymphatic invasion is minimal.4 This makes EMR a versatile and proven therapy, often serving as the first-line treatment for many GIT lesions.
EMR has various techniques that could be categorized into suction or non-suction (lift and cut) techniques. In the suction technique, devices like multiband mucosectomy (MBM) are commonly used, especially in nodular Barrett’s dysplasia, forming a pseudopolyp for subsequent resection. The procedure is characterized by its safety, efficacy, and cost-effectiveness, contributing to its widespread adoption in clinical practice. In the lift and cut approach, a submucosal injection is utilized to separate the muscularis propria from the lesion, thereby reducing the risk of perforation. Different solutions, such as normal saline, hypertonic saline, 50% dextrose, or proprietary submucosal injection solutions, are employed for submucosal injection.5
The non-suction technique using a snare to resect polyps after injection is more often used in colonic and small intestinal EMR. Resection can be done via thermal energy in the form of cut or coagulation; however, there is rising data on the use of piecemeal cold snare resection for select flat polyps of the colon.6 There is also promising data on the role of underwater EMR, a common technique employed for colonic lesions, particularly if the lesion does not lift well with submucosal injection.7
Adverse events associated with EMR include bleeding (7%-8%) and perforation (0.9%-2%).8-9 Adequate submucosal fluid injection is crucial to prevent perforations. However, the main limitation of EMR is the piecemeal nature of resections for lesions larger than 20 mm, leading to compromised histopathologic evaluation for complete excision, especially in cases with superficial submucosal invasion (SMI). This can result in residual or recurrent tissue, reportedly 8% to 20%.10 Despite this limitation, EMR remains a reliable strategy, and recurrent lesions are generally manageable through repeat sessions. The importance of EMR as a therapeutic modality lies in its role in addressing lesions with favorable characteristics, where the risk of SMI is low.
Endoscopic Submucosal Dissection (ESD)
ESD is an evolving technique that can be utilized for submucosal lesions of the GIT, lesions not amenable to EMR due to submucosal fibrosis, when en bloc removal of a lesion is needed for accurate histopathological diagnosis, and when other techniques fail.11-12
ESD was only recently adopted in the United States, requires specialized training, and usually is a lengthier procedure than EMR.13 Compared to EMR, it has higher en bloc resection rates and lower recurrence rates, making it curative for lesions with superficial SMI and favorable histologic features.4,14 The safety profile of ESD appears favorable, with most of the adverse events managed successfully by endoscopic methods. Major complications include intraoperative and delayed perforation, intraoperative and delayed bleeding, aspiration pneumonia, thromboembolism, and stricture formation in the case of circumferential lesions.15
Despite being technically challenging, ESD may provide a cost-effective long-term solution by avoiding surgery, reducing the need for additional interventions by minimizing recurrence rates. Given the technical complexity of ESD, particularly the submucosal dissection portion, techniques such as hybrid ESD developed. Hybrid ESD combines snaring with circumferential mucosal incision and partial submucosal dissection. Although it promises shorter procedure times, reduced complication rates like perforation, and similar recurrence rates compared to traditional ESD, studies have shown lower success rates in en bloc resection.16-17
Both EMR and ESD are considered complementary strategies, and the choice between them should be dictated by lesion characteristics, patient preferences, and local expertise.
Submucosal Tunneling Endoscopic Resection (STER)
STER has emerged as a well-established technique for the endoscopic resection of GI subepithelial tumors (SETs) originating from the muscularis propria layer. The standard STER procedure involves a series of steps including submucosal elevation proximal to the SET, mucosotomy, creation of a submucosal tunnel, dissection of the SET within the tunnel, enucleation from the deep muscle layer, and subsequent specimen retrieval followed by mucosal closure.
This technique is typically recommended for SETs smaller than 3.5 cm, particularly those located in the mid or distal esophagus, cardia, or along the greater curvature of the gastric body.18 However, STER may pose technical challenges for larger SETs or lesions in anatomically difficult locations, where surgical resection is recommended instead.19 Notably, recent large-scale meta-analyses have showcased the favorable complete resection and en bloc resection rates of STER in treating GI SETs.20
Endoscopic Full Thickness Resection (EFTR)
EFTR has emerged as a valuable technique in the endoscopic management of gastrointestinal lesions, particularly SETs and lesions not amenable to EMR or ESD due to fibrosis. EFTR involves the resection of all layers of the GIT from mucosa to serosa, and therefore is well-suited for SETs arising from the muscularis propria (MP).20
EFTR entails two main concepts: tissue resection and complete defect closure. Conventional EFTR consists of several steps, which include mucosal and submucosal pre-cutting, circumferential incision, and dissection through the MP or serosa. This results in a full thickness defect, for which closure of the wall defect is achieved using standard endoscopic clips or a combination of clips and endoloops or endoscopic suturing.21 For lesions less than 2 cm, EFTR can be performed in a single step using a cap-mounted full thickness resection device (FTRD). This results in deployment of over-the-scope clip over the target lesion followed by snaring the lesions above the clip.21
Location of the SET generally dictates the specific modality of ATR. For example, esophageal SETs may be more amenable to STER given that the lesion typically runs parallel with the lumen of the tubular esophagus, which allows for easier dissection without the need of full or partial retroflexion. While gastric SETs can be resected with STER, it may be challenging and more effectively addressed with EFTR, particularly when the entire lesion can be grasped into the full-thickness resection device.22 Limited data exists for duodenal EFTR, and colorectal SETs closure is particularly challenging.
Conclusion
It is key to emphasize that ATR cannot be safely established in practice without the incorporation of a multidisciplinary team (surgeons, radiologists, etc.), specialized tools, and trained personnel. This requires dedicated endoscopic rooms, careful patient selection, and a comprehensive approach to patient care before, during, and after these procedures.
Moreover, it is important to note that some patients may require post-procedure hospitalization for observation to ensure no early complications are encountered. Optimal surveillance strategies after ATR rely heavily on the potential for residual or recurrent disease, underlying pathology, and the expertise of the advanced endoscopist. As the field continues to evolve, ongoing research and technological advances of devices will further enhance the efficacy and safety of ATR in gastroenterology.
Dr. Madi (@MahMadi90) is based in the Division of Gastroenterology and Hepatology, Saint Louis University School of Medicine, Saint Louis, Missouri. Dr. Rengarajan (@ArvindRenga) and Dr. Bazarbashi (@AhmadBazarbashi) are based in the Division of Gastroenterology, Washington University in St. Louis. The authors have no conflicts of interest to disclose, and no funding was required for this project.
References
1. Copland AP, et al. AGA Clinical Practice Update on appropriate and tailored polypectomy: Expert review. Clin Gastroenterol Hepatol. 2024 Mar. doi: 10.1016/j.cgh.2023.10.012.
2. Lee SP, et al. Effect of preceding biopsy on the results of endoscopic submucosal dissection for colorectal laterally spreading tumor. Dig Dis Sci. 2019 Oct. doi: 10.1007/s10620-019-05625-3.
3. Medina-Prado L, et al. When and how to use endoscopic tattooing in the colon: An international Delphi agreement. Clin Gastroenterol Hepatol. 2021 May. doi: 10.1016/j.cgh.2021.01.024.
4. Rashid MU, et al. EMR and ESD: Indications, techniques and results. Surg Oncol. 2022 Aug. doi: 10.1016/j.suronc.2022.101742.
5. Castro R, et al. Solutions for submucosal injection: What to choose and how to do it. World J Gastroenterol. 2019 Feb. doi: 10.3748/wjg.v25.i7.777.
6. Rex DK. Best practices for resection of diminutive and small polyps in the colorectum. Gastrointest Endosc Clin N Am. 2019 Oct. doi: 10.1016/j.giec.2019.06.004.
7. Lv XH, et al. Underwater EMR for nonpedunculated colorectal lesions. Gastrointest Endosc. 2023 Apr. doi: 10.1016/j.gie.2022.10.044.
8. Fujiya M, et al. Efficacy and adverse events of EMR and endoscopic submucosal dissection for the treatment of colon neoplasms: a meta-analysis of studies comparing EMR and endoscopic submucosal dissection. Gastrointest Endosc. 2015 Mar. doi: 10.1016/j.gie.2014.07.034.
9. Kandel P, Wallace MB. Colorectal endoscopic mucosal resection (EMR). Best Pract Res Clin Gastroenterol. 2017 Aug. doi: 10.1016/j.bpg.2017.05.006.
10. Kemper G, et al; ENDOCARE Study Group. Endoscopic techniques to reduce recurrence rates after colorectal EMR: systematic review and meta-analysis. Surg Endosc. 2021 Oct. doi: 10.1007/s00464-021-08574-z.
11. Goto O, et al. Expanding indications for ESD: submucosal disease (SMT/carcinoid tumors). Gastrointest Endosc Clin N Am. 2014 Apr. doi: 10.1016/j.giec.2013.11.006.
12. Wang K, et al. Endoscopic full-thickness resection, indication, methods and perspectives. Dig Endosc. 2023 Jan. doi: 10.1111/den.14474.
13. Herreros de Tejada A. ESD training: A challenging path to excellence. World J Gastrointest Endosc. 2014 Apr 16. doi: 10.4253/wjge.v6.i4.112.
14. Chiba H, et al. Safety and efficacy of simultaneous colorectal ESD for large synchronous colorectal lesions. Endosc Int Open. 2017 Jul. doi: 10.1055/s-0043-110567.
15. Mannath J, Ragunath K. Endoscopic mucosal resection: who and how? Therap Adv Gastroenterol. 2011 Sep. doi: 10.1177/1756283X10388683.
16. Wang XY, et al. Hybrid endoscopic submucosal dissection: An alternative resection modality for large laterally spreading tumors in the cecum? BMC Gastroenterol. 2021 May. doi: 10.1186/s12876-021-01766-w.
17. McCarty TR, et al. Hybrid endoscopic submucosal dissection (ESD) compared with conventional ESD for colorectal lesions: a systematic review and meta-analysis. Endoscopy. 2021 Oct. doi: 10.1055/a-1266-1855.
18. Jain D, et al. Submucosal tunneling endoscopic resection of upper gastrointestinal tract tumors arising from muscularis propria. Ann Gastroenterol. 2017 Feb. doi: 10.20524/aog.2017.0128.
19. Lv XH, et al. Efficacy and safety of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors: a systematic review and meta-analysis. Surg Endosc. 2017 Jan. doi: 10.1007/s00464-016-4978-7.
20. Cao B, et al. Efficacy and safety of submucosal tunneling endoscopic resection for gastric submucosal tumors: a systematic review and meta-analysis. Rev Esp Enferm Dig. 2021 Jan. doi: 10.17235/reed.2020.6989/2020.
21. Cai M, et al. Endoscopic full-thickness resection (EFTR) for gastrointestinal subepithelial tumors. Gastrointest Endosc Clin N Am. 2016 Apr. doi: 10.1016/j.giec.2015.12.013.
22. Brigic A, et al. A systematic review regarding the feasibility and safety of endoscopic full thickness resection (EFTR) for colonic lesions. Surg Endosc. 2013 Oct. doi: 10.1007/s00464-013-2946-z.
Promising New Data Support GLP-1s for Dementia Prevention
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM AAIC 2024
Red Meat Tied to Increased Dementia Risk
PHILADELPHIA – preliminary research shows.
Study participants who consumed 0.25 or more servings of processed meat per day, or roughly two servings per week, had a 15% higher risk for dementia, compared with those who consumed less than 0.10 serving per day, which is about three servings per month.
“Our study found a higher intake of red meat — particularly processed red meat — was associated with a higher risk of developing dementia, as well as worse cognition,” said study author Yuhan Li, MHS, research assistant, Channing Division of Network Medicine, Brigham and Women’s Hospital, Boston, Massachusetts.
However, the study also showed that replacing processed red meat with nuts and legumes could potentially lower this increased risk.
The findings were presented on at the 2024 Alzheimer’s Association International Conference (AAIC).
Inconsistent Research
Previous studies have shown an inconsistent association between red meat intake and cognitive health.
To assess the relationship between diet and dementia, the researchers used data from the Nurses’ Health Study, which began recruiting female registered nurses aged 30-55 years in 1976, and the Health Professionals Follow-Up Study, which began recruiting male health professionals aged 40-75 in 1986.
They assessed processed red meat intake by validated semi-quantitative food frequency questionnaires administered every 2-4 years. Participants were asked how often they consumed a serving of processed red meat.
Investigators also assessed intake of unprocessed red meat, including beef, pork, or lamb as a main dish, in a sandwich or hamburger, or in a mixed dish.
The investigators also looked at participants’ intake of nuts and legumes.
Dementia outcome was a composite endpoint of self-reported dementia and dementia-related death. “Specifically, participants reported a physician diagnosis of Alzheimer’s disease or other forms of dementia by questionnaire. Deaths were identified through state vital statistics records, the National Death Index, family reports, and the postal system,” said Ms. Li.
Three Cognitive Outcomes
Researchers examined three outcomes: dementia, subjective cognitive decline, and objective cognitive function. For dementia, they ascertained incident cases in 87,424 individuals in the UK’s National Health Service database without Parkinson’s disease or baseline dementia, stroke, or cancer.
They longitudinally collected information on subjective cognitive decline from 33,908 Nurses’ Health Study participants and 10,058 participants in the Health Professionals Follow-Up Study.
Cognitive function was assessed using the Telephone Interview for Cognitive Status (1995-2008) in a subset of 17,458 Nurses’ Health Study participants.
Over a follow-up of 38 years (1980-2018), there were 6856 dementia cases in the Nurses’ Health Study. Participants with processed red meat intake of 0.25 or more serving/day, compared with less than 0.10 serving/day, had 15% higher risk for dementia (hazard ratio [HR], 1.15; 95% CI, 1.08-1.23; P < .001).
In addition to an increased risk for dementia, intake of processed red meat was associated with accelerated cognitive aging in global cognition (1.61 years per 1–serving/day increment; 95% CI, 0.20, 3.03) and verbal memory (1.69 years per 1–serving/day increment; 95% CI, 0.13, 3.25; both P = .03).
Participants with processed red meat intake of 0.25 or more serving/day had a 14% higher likelihood of subjective cognitive decline, compared with those with intake less than 0.10 serving/day (odds ratio [OR], 1.14; 95% CI, 1.04-1.24; P = .004).
For unprocessed red meat, consuming 1.00 or more serving/day versus less than 0.50 serving/day was associated with a 16% higher likelihood of subjective cognitive decline (OR, 1.16; 95% CI, 1.04-1.30; P = .02).
Substitution Analysis
Researchers modeled the effects of replacing 1 serving/day of processed red meat with 1 serving/day of nuts and legumes on cognitive outcomes. They did this by treating food intakes as continuous variables and calculating the differences in coefficients of the two food items.
They found that substituting legumes and nuts was associated with a 23% lower risk for dementia (HR, 0.77; 95% CI, 0.69-0.86), 1.37 fewer years of cognitive aging (95% CI, –2.49 to –0.25), and 20% lower odds of subjective cognitive decline (OR, 0.80, 95% CI, 0.69-0.92).
The research cannot determine whether it’s the processing method itself or the type of red meat that affects cognition, Ms. Li cautioned.
“Our study is an epidemiologic study, not a biological mechanism study, but based on our findings, red meat may be related to worse cognition, and processed red meat may add additional risk,” she said.
She also noted that because the study focused solely on red meats, the study cannot determine the potential on the impact of other processed meats on cognition.
Although the study doesn’t address a possible mechanism linking processed red meat with cognition, Ms. Li said it’s possible such meats have high levels of relatively harmful substances, such as nitrites, N-nitroso compounds, and sodium, and that “these carry the additional risk to brain health.”
There are currently no specific guidelines regarding the “safe” amount of processed meat consumption specifically related to cognition, she said.
The study is important because of its large sample size, long follow-up period, and inclusion of repeated measurements of diet, the investigators noted. In addition, researchers assessed both processed and unprocessed red meat and evaluated multiple cognitive outcomes.
The investigators plan to assess the association between other modifiable factors and cognitive health.
Experts Weigh In
In a comment, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, agreed past studies on the topic have been “mixed,” with only some studies reporting links between cognition or dementia and processed red meat.
Another unique aspect of the study, said Dr. Sexton, was the replacement analysis showing the brain benefits of eating nuts and legumes in place of processed red meat. “So, it’s not just suggesting to people what not to do, but also what they can be doing instead.”
That’s why this large study with more than 130,000 adults that tracked individuals for close to 40 years in some cases “is so valuable,” she added.
In a release from the Science Media Centre in the United Kingdom, several other experts commented on the study. Among them, Kevin McConway, PhD, emeritus professor of applied statistics at the Open University, Milton Keynes, England, said that “it’s pretty well impossible to get a clear message from the information that is available so far about this research. It is a conference paper, and all we have seen so far is a press release, a brief summary of the research, and a diagram. There isn’t a detailed, peer-reviewed research report, not yet anyway. Putting out limited information like this isn’t the right way to report science.”
Dr. McConway also noted that the observational study recorded participants’ diets and dementia diagnoses over several years without assigning specific diets. Those who ate more red processed meat had higher rates of dementia and cognitive decline. However, it’s unclear if these differences are caused by red meat consumption or other factors, such as diet, age, ethnicity, or location.
Researchers typically adjust for these factors, but the available information doesn’t specify what adjustments were made or their impact, he noted, and without detailed data, it’s impossible to evaluate the study’s quality. Although eating more red processed meat might increase dementia risk, more research is needed to confirm this, Dr. McConway added.
Also commenting, Sebastian Walsh, a National Institute for Health and Care Research doctoral fellow who researches population-level approaches to dementia risk reduction at University of Cambridge, Cambridge, England, said that without seeing the full paper, it’s difficult to know exactly what to make of the study’s findings.
“On the surface, this is a large and long study. But it isn’t clear how the analysis was done — specifically what other factors were taken into account when looking at this apparent relationship between red meat and dementia.
“Despite a lot of research looking at specific foods and different diseases, the basic public health advice that eating a healthy, balanced diet is good for health is essentially unchanged. Most people know and accept this. What is most important is to find ways of supporting people, particularly those from poorer backgrounds, to follow this advice and address the obesity epidemic,” said Mr. Walsh.
The study was funded by a National Institutes of Health research grant. Ms. Li reports no relevant conflicts of interest. Dr. Sexton, Dr. McConway, and Mr. Walsh report no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – preliminary research shows.
Study participants who consumed 0.25 or more servings of processed meat per day, or roughly two servings per week, had a 15% higher risk for dementia, compared with those who consumed less than 0.10 serving per day, which is about three servings per month.
“Our study found a higher intake of red meat — particularly processed red meat — was associated with a higher risk of developing dementia, as well as worse cognition,” said study author Yuhan Li, MHS, research assistant, Channing Division of Network Medicine, Brigham and Women’s Hospital, Boston, Massachusetts.
However, the study also showed that replacing processed red meat with nuts and legumes could potentially lower this increased risk.
The findings were presented on at the 2024 Alzheimer’s Association International Conference (AAIC).
Inconsistent Research
Previous studies have shown an inconsistent association between red meat intake and cognitive health.
To assess the relationship between diet and dementia, the researchers used data from the Nurses’ Health Study, which began recruiting female registered nurses aged 30-55 years in 1976, and the Health Professionals Follow-Up Study, which began recruiting male health professionals aged 40-75 in 1986.
They assessed processed red meat intake by validated semi-quantitative food frequency questionnaires administered every 2-4 years. Participants were asked how often they consumed a serving of processed red meat.
Investigators also assessed intake of unprocessed red meat, including beef, pork, or lamb as a main dish, in a sandwich or hamburger, or in a mixed dish.
The investigators also looked at participants’ intake of nuts and legumes.
Dementia outcome was a composite endpoint of self-reported dementia and dementia-related death. “Specifically, participants reported a physician diagnosis of Alzheimer’s disease or other forms of dementia by questionnaire. Deaths were identified through state vital statistics records, the National Death Index, family reports, and the postal system,” said Ms. Li.
Three Cognitive Outcomes
Researchers examined three outcomes: dementia, subjective cognitive decline, and objective cognitive function. For dementia, they ascertained incident cases in 87,424 individuals in the UK’s National Health Service database without Parkinson’s disease or baseline dementia, stroke, or cancer.
They longitudinally collected information on subjective cognitive decline from 33,908 Nurses’ Health Study participants and 10,058 participants in the Health Professionals Follow-Up Study.
Cognitive function was assessed using the Telephone Interview for Cognitive Status (1995-2008) in a subset of 17,458 Nurses’ Health Study participants.
Over a follow-up of 38 years (1980-2018), there were 6856 dementia cases in the Nurses’ Health Study. Participants with processed red meat intake of 0.25 or more serving/day, compared with less than 0.10 serving/day, had 15% higher risk for dementia (hazard ratio [HR], 1.15; 95% CI, 1.08-1.23; P < .001).
In addition to an increased risk for dementia, intake of processed red meat was associated with accelerated cognitive aging in global cognition (1.61 years per 1–serving/day increment; 95% CI, 0.20, 3.03) and verbal memory (1.69 years per 1–serving/day increment; 95% CI, 0.13, 3.25; both P = .03).
Participants with processed red meat intake of 0.25 or more serving/day had a 14% higher likelihood of subjective cognitive decline, compared with those with intake less than 0.10 serving/day (odds ratio [OR], 1.14; 95% CI, 1.04-1.24; P = .004).
For unprocessed red meat, consuming 1.00 or more serving/day versus less than 0.50 serving/day was associated with a 16% higher likelihood of subjective cognitive decline (OR, 1.16; 95% CI, 1.04-1.30; P = .02).
Substitution Analysis
Researchers modeled the effects of replacing 1 serving/day of processed red meat with 1 serving/day of nuts and legumes on cognitive outcomes. They did this by treating food intakes as continuous variables and calculating the differences in coefficients of the two food items.
They found that substituting legumes and nuts was associated with a 23% lower risk for dementia (HR, 0.77; 95% CI, 0.69-0.86), 1.37 fewer years of cognitive aging (95% CI, –2.49 to –0.25), and 20% lower odds of subjective cognitive decline (OR, 0.80, 95% CI, 0.69-0.92).
The research cannot determine whether it’s the processing method itself or the type of red meat that affects cognition, Ms. Li cautioned.
“Our study is an epidemiologic study, not a biological mechanism study, but based on our findings, red meat may be related to worse cognition, and processed red meat may add additional risk,” she said.
She also noted that because the study focused solely on red meats, the study cannot determine the potential on the impact of other processed meats on cognition.
Although the study doesn’t address a possible mechanism linking processed red meat with cognition, Ms. Li said it’s possible such meats have high levels of relatively harmful substances, such as nitrites, N-nitroso compounds, and sodium, and that “these carry the additional risk to brain health.”
There are currently no specific guidelines regarding the “safe” amount of processed meat consumption specifically related to cognition, she said.
The study is important because of its large sample size, long follow-up period, and inclusion of repeated measurements of diet, the investigators noted. In addition, researchers assessed both processed and unprocessed red meat and evaluated multiple cognitive outcomes.
The investigators plan to assess the association between other modifiable factors and cognitive health.
Experts Weigh In
In a comment, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, agreed past studies on the topic have been “mixed,” with only some studies reporting links between cognition or dementia and processed red meat.
Another unique aspect of the study, said Dr. Sexton, was the replacement analysis showing the brain benefits of eating nuts and legumes in place of processed red meat. “So, it’s not just suggesting to people what not to do, but also what they can be doing instead.”
That’s why this large study with more than 130,000 adults that tracked individuals for close to 40 years in some cases “is so valuable,” she added.
In a release from the Science Media Centre in the United Kingdom, several other experts commented on the study. Among them, Kevin McConway, PhD, emeritus professor of applied statistics at the Open University, Milton Keynes, England, said that “it’s pretty well impossible to get a clear message from the information that is available so far about this research. It is a conference paper, and all we have seen so far is a press release, a brief summary of the research, and a diagram. There isn’t a detailed, peer-reviewed research report, not yet anyway. Putting out limited information like this isn’t the right way to report science.”
Dr. McConway also noted that the observational study recorded participants’ diets and dementia diagnoses over several years without assigning specific diets. Those who ate more red processed meat had higher rates of dementia and cognitive decline. However, it’s unclear if these differences are caused by red meat consumption or other factors, such as diet, age, ethnicity, or location.
Researchers typically adjust for these factors, but the available information doesn’t specify what adjustments were made or their impact, he noted, and without detailed data, it’s impossible to evaluate the study’s quality. Although eating more red processed meat might increase dementia risk, more research is needed to confirm this, Dr. McConway added.
Also commenting, Sebastian Walsh, a National Institute for Health and Care Research doctoral fellow who researches population-level approaches to dementia risk reduction at University of Cambridge, Cambridge, England, said that without seeing the full paper, it’s difficult to know exactly what to make of the study’s findings.
“On the surface, this is a large and long study. But it isn’t clear how the analysis was done — specifically what other factors were taken into account when looking at this apparent relationship between red meat and dementia.
“Despite a lot of research looking at specific foods and different diseases, the basic public health advice that eating a healthy, balanced diet is good for health is essentially unchanged. Most people know and accept this. What is most important is to find ways of supporting people, particularly those from poorer backgrounds, to follow this advice and address the obesity epidemic,” said Mr. Walsh.
The study was funded by a National Institutes of Health research grant. Ms. Li reports no relevant conflicts of interest. Dr. Sexton, Dr. McConway, and Mr. Walsh report no relevant disclosures.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – preliminary research shows.
Study participants who consumed 0.25 or more servings of processed meat per day, or roughly two servings per week, had a 15% higher risk for dementia, compared with those who consumed less than 0.10 serving per day, which is about three servings per month.
“Our study found a higher intake of red meat — particularly processed red meat — was associated with a higher risk of developing dementia, as well as worse cognition,” said study author Yuhan Li, MHS, research assistant, Channing Division of Network Medicine, Brigham and Women’s Hospital, Boston, Massachusetts.
However, the study also showed that replacing processed red meat with nuts and legumes could potentially lower this increased risk.
The findings were presented on at the 2024 Alzheimer’s Association International Conference (AAIC).
Inconsistent Research
Previous studies have shown an inconsistent association between red meat intake and cognitive health.
To assess the relationship between diet and dementia, the researchers used data from the Nurses’ Health Study, which began recruiting female registered nurses aged 30-55 years in 1976, and the Health Professionals Follow-Up Study, which began recruiting male health professionals aged 40-75 in 1986.
They assessed processed red meat intake by validated semi-quantitative food frequency questionnaires administered every 2-4 years. Participants were asked how often they consumed a serving of processed red meat.
Investigators also assessed intake of unprocessed red meat, including beef, pork, or lamb as a main dish, in a sandwich or hamburger, or in a mixed dish.
The investigators also looked at participants’ intake of nuts and legumes.
Dementia outcome was a composite endpoint of self-reported dementia and dementia-related death. “Specifically, participants reported a physician diagnosis of Alzheimer’s disease or other forms of dementia by questionnaire. Deaths were identified through state vital statistics records, the National Death Index, family reports, and the postal system,” said Ms. Li.
Three Cognitive Outcomes
Researchers examined three outcomes: dementia, subjective cognitive decline, and objective cognitive function. For dementia, they ascertained incident cases in 87,424 individuals in the UK’s National Health Service database without Parkinson’s disease or baseline dementia, stroke, or cancer.
They longitudinally collected information on subjective cognitive decline from 33,908 Nurses’ Health Study participants and 10,058 participants in the Health Professionals Follow-Up Study.
Cognitive function was assessed using the Telephone Interview for Cognitive Status (1995-2008) in a subset of 17,458 Nurses’ Health Study participants.
Over a follow-up of 38 years (1980-2018), there were 6856 dementia cases in the Nurses’ Health Study. Participants with processed red meat intake of 0.25 or more serving/day, compared with less than 0.10 serving/day, had 15% higher risk for dementia (hazard ratio [HR], 1.15; 95% CI, 1.08-1.23; P < .001).
In addition to an increased risk for dementia, intake of processed red meat was associated with accelerated cognitive aging in global cognition (1.61 years per 1–serving/day increment; 95% CI, 0.20, 3.03) and verbal memory (1.69 years per 1–serving/day increment; 95% CI, 0.13, 3.25; both P = .03).
Participants with processed red meat intake of 0.25 or more serving/day had a 14% higher likelihood of subjective cognitive decline, compared with those with intake less than 0.10 serving/day (odds ratio [OR], 1.14; 95% CI, 1.04-1.24; P = .004).
For unprocessed red meat, consuming 1.00 or more serving/day versus less than 0.50 serving/day was associated with a 16% higher likelihood of subjective cognitive decline (OR, 1.16; 95% CI, 1.04-1.30; P = .02).
Substitution Analysis
Researchers modeled the effects of replacing 1 serving/day of processed red meat with 1 serving/day of nuts and legumes on cognitive outcomes. They did this by treating food intakes as continuous variables and calculating the differences in coefficients of the two food items.
They found that substituting legumes and nuts was associated with a 23% lower risk for dementia (HR, 0.77; 95% CI, 0.69-0.86), 1.37 fewer years of cognitive aging (95% CI, –2.49 to –0.25), and 20% lower odds of subjective cognitive decline (OR, 0.80, 95% CI, 0.69-0.92).
The research cannot determine whether it’s the processing method itself or the type of red meat that affects cognition, Ms. Li cautioned.
“Our study is an epidemiologic study, not a biological mechanism study, but based on our findings, red meat may be related to worse cognition, and processed red meat may add additional risk,” she said.
She also noted that because the study focused solely on red meats, the study cannot determine the potential on the impact of other processed meats on cognition.
Although the study doesn’t address a possible mechanism linking processed red meat with cognition, Ms. Li said it’s possible such meats have high levels of relatively harmful substances, such as nitrites, N-nitroso compounds, and sodium, and that “these carry the additional risk to brain health.”
There are currently no specific guidelines regarding the “safe” amount of processed meat consumption specifically related to cognition, she said.
The study is important because of its large sample size, long follow-up period, and inclusion of repeated measurements of diet, the investigators noted. In addition, researchers assessed both processed and unprocessed red meat and evaluated multiple cognitive outcomes.
The investigators plan to assess the association between other modifiable factors and cognitive health.
Experts Weigh In
In a comment, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, agreed past studies on the topic have been “mixed,” with only some studies reporting links between cognition or dementia and processed red meat.
Another unique aspect of the study, said Dr. Sexton, was the replacement analysis showing the brain benefits of eating nuts and legumes in place of processed red meat. “So, it’s not just suggesting to people what not to do, but also what they can be doing instead.”
That’s why this large study with more than 130,000 adults that tracked individuals for close to 40 years in some cases “is so valuable,” she added.
In a release from the Science Media Centre in the United Kingdom, several other experts commented on the study. Among them, Kevin McConway, PhD, emeritus professor of applied statistics at the Open University, Milton Keynes, England, said that “it’s pretty well impossible to get a clear message from the information that is available so far about this research. It is a conference paper, and all we have seen so far is a press release, a brief summary of the research, and a diagram. There isn’t a detailed, peer-reviewed research report, not yet anyway. Putting out limited information like this isn’t the right way to report science.”
Dr. McConway also noted that the observational study recorded participants’ diets and dementia diagnoses over several years without assigning specific diets. Those who ate more red processed meat had higher rates of dementia and cognitive decline. However, it’s unclear if these differences are caused by red meat consumption or other factors, such as diet, age, ethnicity, or location.
Researchers typically adjust for these factors, but the available information doesn’t specify what adjustments were made or their impact, he noted, and without detailed data, it’s impossible to evaluate the study’s quality. Although eating more red processed meat might increase dementia risk, more research is needed to confirm this, Dr. McConway added.
Also commenting, Sebastian Walsh, a National Institute for Health and Care Research doctoral fellow who researches population-level approaches to dementia risk reduction at University of Cambridge, Cambridge, England, said that without seeing the full paper, it’s difficult to know exactly what to make of the study’s findings.
“On the surface, this is a large and long study. But it isn’t clear how the analysis was done — specifically what other factors were taken into account when looking at this apparent relationship between red meat and dementia.
“Despite a lot of research looking at specific foods and different diseases, the basic public health advice that eating a healthy, balanced diet is good for health is essentially unchanged. Most people know and accept this. What is most important is to find ways of supporting people, particularly those from poorer backgrounds, to follow this advice and address the obesity epidemic,” said Mr. Walsh.
The study was funded by a National Institutes of Health research grant. Ms. Li reports no relevant conflicts of interest. Dr. Sexton, Dr. McConway, and Mr. Walsh report no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM AAIC 2024
Will Hospital-at-Home Go Mainstream?
Jordan Stohler, a 42-year-old nurse in Knoxville, Tennessee, was readmitted to Fort Sanders Medical Center in June 2023 with sepsis after a double mastectomy.
She spent 5 days in the hospital after surgery to clear up the infection. Then she was offered a choice: She could either stay in the hospital while she received IV antibiotics, or she could go home and have the antibiotics given to her there under the Advanced Care at Home program of Covenant Health, the nine-hospital system to which Fort Sanders belongs.
She opted to go home, where she knew she’d be more comfortable and would be close to her beloved dog. In the end, she was very glad she did.
“I received great care in the hospital, but to be allowed to be in the comfort of your own home, to be around my dog, who I think is therapeutic, to be able to cook my own meals, and to have the same one-on-one nursing care that I would have gotten in the hospital was great,” Ms. Stohler said. “
Being cared for at home helped her heal, she said. “I probably would have gotten a little stir crazy if I’d stayed in the hospital any longer. I received excellent care at home.”
Currently, 322 hospitals in 37 states have Medicare waivers for these kinds of programs, although not all of them are currently functioning.
A recent survey published in JAMA found that nearly half of consumers would accept hospital-at-home, and more than a third were neutral on it. Only 17% said they’d rather be cared for in a brick-and-mortar hospital.
The findings of the JAMA survey confirm those of earlier studies, said Bruce Leff, MD, a professor at Johns Hopkins Medical School in Baltimore, who has researched hospital-at-home since the 1990s. Like the new study, those trials found that the results had no relationship to individual traits, such as socioeconomic status, medical conditions, age, gender, or race.
Whether a person felt comfortable with the idea of hospital-at-home boiled down “to a preference for receiving care at home or in the hospital,” he said. Some people distrust hospitals, and others feel insecure about receiving care at home, even if it is provided by qualified health care professionals.
How Patients Are Selected
While the details of hospital-at-home vary from program to program, the basic scenario is that patients who need certain kinds of acute care can be sent home from hospitals, emergency departments, or clinics to receive that care at home. Among the kinds of conditions that make stable patients eligible are heart failure, COPD, pneumonia, cellulitis, and COVID-19, said John Busigin, MD, a hospitalist and medical director of Covenant Advanced Care at Home.
When a patient is admitted to hospital-at-home, the hospital will send along whatever equipment and medications that person needs. In some cases, this may include a hospital bed, although Ms. Stohler used her own. An IV line was put into her arm, and the IV stand was placed next to the bed.
Ms. Stohler received a computer tablet that she used to communicate with doctors and nurses in Covenant’s “command center” in Knoxville. She also wore a watch with a button she could push in case of an emergency. And she had a telephone line that went directly to her medical team, in case she had an issue and the tablet didn’t work.
Twice a day, or as needed, specially trained paramedics came to Ms. Stohler’s home. They checked on the IV line, changed the IV bag, performed tests, and uploaded vital signs from monitoring equipment to Ms. Stohler’s tablet so it could be transmitted to the command center. A physician assistant came in on the second and fourth days of her weeklong stay in the program, and she saw a hospitalist remotely every day.
While some hospital-at-home programs have registered nurses visit patients at home, RNs are in short supply. To fill this gap, Covenant’s program uses community paramedics who have been in the field for at least 5 years, doing everything from intubating patients and placing them on ventilators to providing advanced cardiac life support, Dr. Busigin said. To get certified as community paramedics, they go through a 3-month training program.
Shortly after Ms. Stohler went into hospital-at-home, she had another crisis. Excess fluid had built up in her body because of all the IV fluids she’d received in the hospital while fighting the sepsis. As a result, she became short of breath. If she had been discharged to home rather than hospital-at-home, she said, she would have had to go to the emergency room. Instead, she sent out a distress call. One of the paramedics rushed to her house and gave her an IV diuretic medication, which helped her urinate to get rid of the excess fluid.
A small number of the estimated 300 people who have gone through the program had to be admitted to the hospital, Dr. Busigin said. Nationally, he said, about 5%-10% are admitted. But readmissions among the patients in the Covenant program have been 25% lower than for patients who received conventional hospital care and had the same conditions as those in hospital-at-home.
Studies have shown that these programs not only reduce readmissions, but also cost less, on average, and create a better patient experience than traditional hospital care does. And, according to the JAMA survey, most consumers like the idea. Fifty-six percent of people who took the survey agreed with the statement that people recover faster at home than in the hospital. Fifty-nine percent agreed they’d feel safe being treated at home, and 49% said they’d be more comfortable if treated at home.
The 1134 people who took the survey were also asked about their comfort level with providing various kinds of care to their loved ones during a hospital-at-home episode. The results varied with the type of task: For example, 82% of the respondents agreed or strongly agreed they could manage a patient’s medications, while just 41% said they’d be willing to change a feeding tube. Smaller percentages were willing to change an IV bag or a catheter or do wound care.
However, hospital-at-home programs don’t allow caregivers to take part in clinical care, which is prohibited by Medicare waivers and state licensing regulations. None of the 22 health systems that use the hospital-at-home services of Medically Home, including Covenant, ask caregivers to do anything along this line, said Pippa Shulman, DO, medical director of the company, which provides equipment, technology, and protocols for hospital programs
The only exception at Covenant, Dr. Busigin said, is that the hospital may train family members to do wound care when a patient is discharged from the hospital to Advanced Care at Home. They may also prepare meals for their loved ones, although the program provides balanced meals to patients if they want them. Ms. Stohler had some of these meals, which just had to be heated up, for the first few days of hospital-at-home, and later her relatives brought meals to her house.
Challenges for the Future
The number of Medicare hospital-at-home waivers has nearly doubled since 2021. A year earlier, when Medicare began reimbursing hospitals for acute care at home to help them cope with the overflow of COVID patients, there were only about 15-20 programs in the United States, said Dr. Leff of Johns Hopkins.
A big reason for the lack of use before the pandemic, Dr. Leff said, is that there was no payment system for hospitals that offered hospital-at-home. Now, they can get paid by Medicare and 10 state Medicaid programs, and a number of private payers are also coming on board. Ms. Stohler’s private insurer covered her hospital-at-home stay, and Dr. Busigin said several plans that contract with Covenant will pay for it.
Dr. Leff said he’s cautiously optimistic Congress will extend the Medicare waiver program, which is scheduled to end in December, for another 5 years. A couple of key House committees have signed off on a bill to do that, he said, and a Congressional Budget Office report found that the program did not cost Medicare more money.
But even if the waiver is renewed, some health systems may find it tough to deliver the service. The current version of this model depends a lot on technology, because telemedicine is used and reliable communication is needed for patients in hospital-at-home. That’s why many of the hospitals hire outside vendors like Medically Home to provide the infrastructure they need.
Medically Home manages the tablets given to patients and all connection and networking services, including internet and cellphone connections. It also provides technical services in the command centers that hospitals set up for the doctors and nurses who provide care remotely.
And the firm figures out how to deliver the standard care for each condition in each hospital-at-home. “We need to make sure that the patient is going to get what they need in the time frame it needs to be delivered in, and that it’s safe and effective for the patient,” Dr. Shulman said. “So we’ve developed logistical protocols for a multitude of disease states that allow us to provide high-acuity care in the home to a variety of complex patients.”
The health care workers use the hospital electronic health record for hospital-at-home patients, and vital signs uploaded from patient tablets flow directly into the electronic health record, she said.
Rural Areas Need Help
The use of hospital-at-home in rural areas holds a lot of promise, Dr. Leff said.
“A lot of rural hospitals have been closing, and hospital-at-home could be a mechanism to create hospital-level care where facilities have closed down. It’s easier to do this in urban areas, but it can be done in rural environments as well.”
Rami Karjian, CEO of Medically Home, agreed. The firm services hospital-at-home programs in rural areas of Oklahoma and California, using cellphones and paramedics in areas that lack broadband connections and nurses, he pointed out.
“Hospital-at-home can’t just be available to people who live in big cities,” he said. “The access problems in health care are pervasive, and this is part of how we solve access problems in rural areas.”
A version of this article first appeared on WebMD.com.
Jordan Stohler, a 42-year-old nurse in Knoxville, Tennessee, was readmitted to Fort Sanders Medical Center in June 2023 with sepsis after a double mastectomy.
She spent 5 days in the hospital after surgery to clear up the infection. Then she was offered a choice: She could either stay in the hospital while she received IV antibiotics, or she could go home and have the antibiotics given to her there under the Advanced Care at Home program of Covenant Health, the nine-hospital system to which Fort Sanders belongs.
She opted to go home, where she knew she’d be more comfortable and would be close to her beloved dog. In the end, she was very glad she did.
“I received great care in the hospital, but to be allowed to be in the comfort of your own home, to be around my dog, who I think is therapeutic, to be able to cook my own meals, and to have the same one-on-one nursing care that I would have gotten in the hospital was great,” Ms. Stohler said. “
Being cared for at home helped her heal, she said. “I probably would have gotten a little stir crazy if I’d stayed in the hospital any longer. I received excellent care at home.”
Currently, 322 hospitals in 37 states have Medicare waivers for these kinds of programs, although not all of them are currently functioning.
A recent survey published in JAMA found that nearly half of consumers would accept hospital-at-home, and more than a third were neutral on it. Only 17% said they’d rather be cared for in a brick-and-mortar hospital.
The findings of the JAMA survey confirm those of earlier studies, said Bruce Leff, MD, a professor at Johns Hopkins Medical School in Baltimore, who has researched hospital-at-home since the 1990s. Like the new study, those trials found that the results had no relationship to individual traits, such as socioeconomic status, medical conditions, age, gender, or race.
Whether a person felt comfortable with the idea of hospital-at-home boiled down “to a preference for receiving care at home or in the hospital,” he said. Some people distrust hospitals, and others feel insecure about receiving care at home, even if it is provided by qualified health care professionals.
How Patients Are Selected
While the details of hospital-at-home vary from program to program, the basic scenario is that patients who need certain kinds of acute care can be sent home from hospitals, emergency departments, or clinics to receive that care at home. Among the kinds of conditions that make stable patients eligible are heart failure, COPD, pneumonia, cellulitis, and COVID-19, said John Busigin, MD, a hospitalist and medical director of Covenant Advanced Care at Home.
When a patient is admitted to hospital-at-home, the hospital will send along whatever equipment and medications that person needs. In some cases, this may include a hospital bed, although Ms. Stohler used her own. An IV line was put into her arm, and the IV stand was placed next to the bed.
Ms. Stohler received a computer tablet that she used to communicate with doctors and nurses in Covenant’s “command center” in Knoxville. She also wore a watch with a button she could push in case of an emergency. And she had a telephone line that went directly to her medical team, in case she had an issue and the tablet didn’t work.
Twice a day, or as needed, specially trained paramedics came to Ms. Stohler’s home. They checked on the IV line, changed the IV bag, performed tests, and uploaded vital signs from monitoring equipment to Ms. Stohler’s tablet so it could be transmitted to the command center. A physician assistant came in on the second and fourth days of her weeklong stay in the program, and she saw a hospitalist remotely every day.
While some hospital-at-home programs have registered nurses visit patients at home, RNs are in short supply. To fill this gap, Covenant’s program uses community paramedics who have been in the field for at least 5 years, doing everything from intubating patients and placing them on ventilators to providing advanced cardiac life support, Dr. Busigin said. To get certified as community paramedics, they go through a 3-month training program.
Shortly after Ms. Stohler went into hospital-at-home, she had another crisis. Excess fluid had built up in her body because of all the IV fluids she’d received in the hospital while fighting the sepsis. As a result, she became short of breath. If she had been discharged to home rather than hospital-at-home, she said, she would have had to go to the emergency room. Instead, she sent out a distress call. One of the paramedics rushed to her house and gave her an IV diuretic medication, which helped her urinate to get rid of the excess fluid.
A small number of the estimated 300 people who have gone through the program had to be admitted to the hospital, Dr. Busigin said. Nationally, he said, about 5%-10% are admitted. But readmissions among the patients in the Covenant program have been 25% lower than for patients who received conventional hospital care and had the same conditions as those in hospital-at-home.
Studies have shown that these programs not only reduce readmissions, but also cost less, on average, and create a better patient experience than traditional hospital care does. And, according to the JAMA survey, most consumers like the idea. Fifty-six percent of people who took the survey agreed with the statement that people recover faster at home than in the hospital. Fifty-nine percent agreed they’d feel safe being treated at home, and 49% said they’d be more comfortable if treated at home.
The 1134 people who took the survey were also asked about their comfort level with providing various kinds of care to their loved ones during a hospital-at-home episode. The results varied with the type of task: For example, 82% of the respondents agreed or strongly agreed they could manage a patient’s medications, while just 41% said they’d be willing to change a feeding tube. Smaller percentages were willing to change an IV bag or a catheter or do wound care.
However, hospital-at-home programs don’t allow caregivers to take part in clinical care, which is prohibited by Medicare waivers and state licensing regulations. None of the 22 health systems that use the hospital-at-home services of Medically Home, including Covenant, ask caregivers to do anything along this line, said Pippa Shulman, DO, medical director of the company, which provides equipment, technology, and protocols for hospital programs
The only exception at Covenant, Dr. Busigin said, is that the hospital may train family members to do wound care when a patient is discharged from the hospital to Advanced Care at Home. They may also prepare meals for their loved ones, although the program provides balanced meals to patients if they want them. Ms. Stohler had some of these meals, which just had to be heated up, for the first few days of hospital-at-home, and later her relatives brought meals to her house.
Challenges for the Future
The number of Medicare hospital-at-home waivers has nearly doubled since 2021. A year earlier, when Medicare began reimbursing hospitals for acute care at home to help them cope with the overflow of COVID patients, there were only about 15-20 programs in the United States, said Dr. Leff of Johns Hopkins.
A big reason for the lack of use before the pandemic, Dr. Leff said, is that there was no payment system for hospitals that offered hospital-at-home. Now, they can get paid by Medicare and 10 state Medicaid programs, and a number of private payers are also coming on board. Ms. Stohler’s private insurer covered her hospital-at-home stay, and Dr. Busigin said several plans that contract with Covenant will pay for it.
Dr. Leff said he’s cautiously optimistic Congress will extend the Medicare waiver program, which is scheduled to end in December, for another 5 years. A couple of key House committees have signed off on a bill to do that, he said, and a Congressional Budget Office report found that the program did not cost Medicare more money.
But even if the waiver is renewed, some health systems may find it tough to deliver the service. The current version of this model depends a lot on technology, because telemedicine is used and reliable communication is needed for patients in hospital-at-home. That’s why many of the hospitals hire outside vendors like Medically Home to provide the infrastructure they need.
Medically Home manages the tablets given to patients and all connection and networking services, including internet and cellphone connections. It also provides technical services in the command centers that hospitals set up for the doctors and nurses who provide care remotely.
And the firm figures out how to deliver the standard care for each condition in each hospital-at-home. “We need to make sure that the patient is going to get what they need in the time frame it needs to be delivered in, and that it’s safe and effective for the patient,” Dr. Shulman said. “So we’ve developed logistical protocols for a multitude of disease states that allow us to provide high-acuity care in the home to a variety of complex patients.”
The health care workers use the hospital electronic health record for hospital-at-home patients, and vital signs uploaded from patient tablets flow directly into the electronic health record, she said.
Rural Areas Need Help
The use of hospital-at-home in rural areas holds a lot of promise, Dr. Leff said.
“A lot of rural hospitals have been closing, and hospital-at-home could be a mechanism to create hospital-level care where facilities have closed down. It’s easier to do this in urban areas, but it can be done in rural environments as well.”
Rami Karjian, CEO of Medically Home, agreed. The firm services hospital-at-home programs in rural areas of Oklahoma and California, using cellphones and paramedics in areas that lack broadband connections and nurses, he pointed out.
“Hospital-at-home can’t just be available to people who live in big cities,” he said. “The access problems in health care are pervasive, and this is part of how we solve access problems in rural areas.”
A version of this article first appeared on WebMD.com.
Jordan Stohler, a 42-year-old nurse in Knoxville, Tennessee, was readmitted to Fort Sanders Medical Center in June 2023 with sepsis after a double mastectomy.
She spent 5 days in the hospital after surgery to clear up the infection. Then she was offered a choice: She could either stay in the hospital while she received IV antibiotics, or she could go home and have the antibiotics given to her there under the Advanced Care at Home program of Covenant Health, the nine-hospital system to which Fort Sanders belongs.
She opted to go home, where she knew she’d be more comfortable and would be close to her beloved dog. In the end, she was very glad she did.
“I received great care in the hospital, but to be allowed to be in the comfort of your own home, to be around my dog, who I think is therapeutic, to be able to cook my own meals, and to have the same one-on-one nursing care that I would have gotten in the hospital was great,” Ms. Stohler said. “
Being cared for at home helped her heal, she said. “I probably would have gotten a little stir crazy if I’d stayed in the hospital any longer. I received excellent care at home.”
Currently, 322 hospitals in 37 states have Medicare waivers for these kinds of programs, although not all of them are currently functioning.
A recent survey published in JAMA found that nearly half of consumers would accept hospital-at-home, and more than a third were neutral on it. Only 17% said they’d rather be cared for in a brick-and-mortar hospital.
The findings of the JAMA survey confirm those of earlier studies, said Bruce Leff, MD, a professor at Johns Hopkins Medical School in Baltimore, who has researched hospital-at-home since the 1990s. Like the new study, those trials found that the results had no relationship to individual traits, such as socioeconomic status, medical conditions, age, gender, or race.
Whether a person felt comfortable with the idea of hospital-at-home boiled down “to a preference for receiving care at home or in the hospital,” he said. Some people distrust hospitals, and others feel insecure about receiving care at home, even if it is provided by qualified health care professionals.
How Patients Are Selected
While the details of hospital-at-home vary from program to program, the basic scenario is that patients who need certain kinds of acute care can be sent home from hospitals, emergency departments, or clinics to receive that care at home. Among the kinds of conditions that make stable patients eligible are heart failure, COPD, pneumonia, cellulitis, and COVID-19, said John Busigin, MD, a hospitalist and medical director of Covenant Advanced Care at Home.
When a patient is admitted to hospital-at-home, the hospital will send along whatever equipment and medications that person needs. In some cases, this may include a hospital bed, although Ms. Stohler used her own. An IV line was put into her arm, and the IV stand was placed next to the bed.
Ms. Stohler received a computer tablet that she used to communicate with doctors and nurses in Covenant’s “command center” in Knoxville. She also wore a watch with a button she could push in case of an emergency. And she had a telephone line that went directly to her medical team, in case she had an issue and the tablet didn’t work.
Twice a day, or as needed, specially trained paramedics came to Ms. Stohler’s home. They checked on the IV line, changed the IV bag, performed tests, and uploaded vital signs from monitoring equipment to Ms. Stohler’s tablet so it could be transmitted to the command center. A physician assistant came in on the second and fourth days of her weeklong stay in the program, and she saw a hospitalist remotely every day.
While some hospital-at-home programs have registered nurses visit patients at home, RNs are in short supply. To fill this gap, Covenant’s program uses community paramedics who have been in the field for at least 5 years, doing everything from intubating patients and placing them on ventilators to providing advanced cardiac life support, Dr. Busigin said. To get certified as community paramedics, they go through a 3-month training program.
Shortly after Ms. Stohler went into hospital-at-home, she had another crisis. Excess fluid had built up in her body because of all the IV fluids she’d received in the hospital while fighting the sepsis. As a result, she became short of breath. If she had been discharged to home rather than hospital-at-home, she said, she would have had to go to the emergency room. Instead, she sent out a distress call. One of the paramedics rushed to her house and gave her an IV diuretic medication, which helped her urinate to get rid of the excess fluid.
A small number of the estimated 300 people who have gone through the program had to be admitted to the hospital, Dr. Busigin said. Nationally, he said, about 5%-10% are admitted. But readmissions among the patients in the Covenant program have been 25% lower than for patients who received conventional hospital care and had the same conditions as those in hospital-at-home.
Studies have shown that these programs not only reduce readmissions, but also cost less, on average, and create a better patient experience than traditional hospital care does. And, according to the JAMA survey, most consumers like the idea. Fifty-six percent of people who took the survey agreed with the statement that people recover faster at home than in the hospital. Fifty-nine percent agreed they’d feel safe being treated at home, and 49% said they’d be more comfortable if treated at home.
The 1134 people who took the survey were also asked about their comfort level with providing various kinds of care to their loved ones during a hospital-at-home episode. The results varied with the type of task: For example, 82% of the respondents agreed or strongly agreed they could manage a patient’s medications, while just 41% said they’d be willing to change a feeding tube. Smaller percentages were willing to change an IV bag or a catheter or do wound care.
However, hospital-at-home programs don’t allow caregivers to take part in clinical care, which is prohibited by Medicare waivers and state licensing regulations. None of the 22 health systems that use the hospital-at-home services of Medically Home, including Covenant, ask caregivers to do anything along this line, said Pippa Shulman, DO, medical director of the company, which provides equipment, technology, and protocols for hospital programs
The only exception at Covenant, Dr. Busigin said, is that the hospital may train family members to do wound care when a patient is discharged from the hospital to Advanced Care at Home. They may also prepare meals for their loved ones, although the program provides balanced meals to patients if they want them. Ms. Stohler had some of these meals, which just had to be heated up, for the first few days of hospital-at-home, and later her relatives brought meals to her house.
Challenges for the Future
The number of Medicare hospital-at-home waivers has nearly doubled since 2021. A year earlier, when Medicare began reimbursing hospitals for acute care at home to help them cope with the overflow of COVID patients, there were only about 15-20 programs in the United States, said Dr. Leff of Johns Hopkins.
A big reason for the lack of use before the pandemic, Dr. Leff said, is that there was no payment system for hospitals that offered hospital-at-home. Now, they can get paid by Medicare and 10 state Medicaid programs, and a number of private payers are also coming on board. Ms. Stohler’s private insurer covered her hospital-at-home stay, and Dr. Busigin said several plans that contract with Covenant will pay for it.
Dr. Leff said he’s cautiously optimistic Congress will extend the Medicare waiver program, which is scheduled to end in December, for another 5 years. A couple of key House committees have signed off on a bill to do that, he said, and a Congressional Budget Office report found that the program did not cost Medicare more money.
But even if the waiver is renewed, some health systems may find it tough to deliver the service. The current version of this model depends a lot on technology, because telemedicine is used and reliable communication is needed for patients in hospital-at-home. That’s why many of the hospitals hire outside vendors like Medically Home to provide the infrastructure they need.
Medically Home manages the tablets given to patients and all connection and networking services, including internet and cellphone connections. It also provides technical services in the command centers that hospitals set up for the doctors and nurses who provide care remotely.
And the firm figures out how to deliver the standard care for each condition in each hospital-at-home. “We need to make sure that the patient is going to get what they need in the time frame it needs to be delivered in, and that it’s safe and effective for the patient,” Dr. Shulman said. “So we’ve developed logistical protocols for a multitude of disease states that allow us to provide high-acuity care in the home to a variety of complex patients.”
The health care workers use the hospital electronic health record for hospital-at-home patients, and vital signs uploaded from patient tablets flow directly into the electronic health record, she said.
Rural Areas Need Help
The use of hospital-at-home in rural areas holds a lot of promise, Dr. Leff said.
“A lot of rural hospitals have been closing, and hospital-at-home could be a mechanism to create hospital-level care where facilities have closed down. It’s easier to do this in urban areas, but it can be done in rural environments as well.”
Rami Karjian, CEO of Medically Home, agreed. The firm services hospital-at-home programs in rural areas of Oklahoma and California, using cellphones and paramedics in areas that lack broadband connections and nurses, he pointed out.
“Hospital-at-home can’t just be available to people who live in big cities,” he said. “The access problems in health care are pervasive, and this is part of how we solve access problems in rural areas.”
A version of this article first appeared on WebMD.com.
The Shield Sign of Cutaneous Metastases Is Associated With Carcinoma Hemorrhagiectoides
To the Editor:
We read with interest the Case Letter from Wang et al1 (Cutis. 2023;112:E13-E15) of a 60-year-old man whose metastatic salivary duct adenocarcinoma manifested with the shield sign as well as carcinoma hemorrhagiectoides. Cutaneous metastases have seldom been described in association with salivary duct carcinoma.2-7 In addition, carcinoma hemorrhagiectoides–associated shield sign has not been commonly reported.5,8-12
Salivary duct carcinoma—an uncommon head and neck malignancy characterized by androgen receptor expression—rarely is associated with cutaneous metastases. Based on a PubMed search of articles indexed for MEDLINE using the terms cutaneous, metastatic, salivary duct carcinoma, and/or skin, including the patient described by Wang et al,1 there have been 8 individuals with cutaneous metastases from this cancer. The morphology of the cutaneous metastases has varied from angiomatous to angiokeratomalike (black and keratotic) papules, bullae, macules (red), papules and nodules (erythematous and scaly), plaques (cellulitislike and confluent that were purpuric, hemorrhagic, and violaceous), pseudovesicles, purpuric papules, subcutaneous nodules, and an ulcer (superficial and mimicked a basal cell carcinoma).1-7 Remarkably, 4 of 8 patients (50%) with salivary duct carcinoma cutaneous metastases presented with a shield sign,5,7 including the case reported by Wang et al.1
The shield sign is a distinctive clinical manifestation of cutaneous metastasis.10 It was named to describe the skin metastases located predominantly on the chest area that would be covered by a medieval knight’s shield5,10,12; metastatic lesions also have been noted on the proximal arm and/or the upper back in a similar distribution.8,9 To date, based on a PubMed search of articles indexed for MEDLINE using the search terms breast cancer, carcinoma, hemorrhagiectoides, metastases, salivary duct carcinoma, shield, and/or sign, the shield sign has been described in 6 patients with cutaneous metastases either from salivary duct carcinoma (4 patients)1,5,7 or breast cancer (2 patients).8,9 The shield sign pathologically corresponds to carcinoma hemorrhagiectoides, an inflammatory pattern of cutaneous metastases.5,11
Inflammatory cutaneous metastatic carcinoma has 3 distinctive clinical and pathologic manifestations.11 Carcinoma erysipelatoides and carcinoma telangiectoides were the earlier described variants.11 In 2012, carcinoma hemorrhagiectoides was described as the third pattern of inflammatory cutaneous metastasis.5
Carcinoma erysipelatoides, which clinically mimics cutaneous streptococcal cellulitis, appears as a well-defined erythematous patch or plaque; the tumor cells can be found in the lymphatic vessels and either are absent or minimally present in the dermis. Carcinoma telangiectoides, which clinically mimics idiopathic telangiectases, appears as an erythematous patch with prominent telangiectases; the tumor cells can be found in the blood vessels and are either absent or minimally present in the dermis. Carcinoma hemorrhagiectoides appears as purpuric or violaceous indurated plaques; the tumor cells are not only found in the blood vessels, in the lymphatic vessels, or both, but also can be mildly to extensively present in the dermis.5,10,11
In conclusion, the shield sign is a unique presentation of inflammatory cutaneous metastatic carcinoma, which is associated with carcinoma hemorrhagiectoides. The clinical features of the infiltrated plaques correspond to the presence of tumor cells in the blood vessels, lymphatic vessels, and the dermis; in addition, the purpuric and violaceous appearance correlates with the presence of extravasated erythrocytes or hemorrhage in the dermis. To date, half of the patients with skin metastases from salivary duct carcinoma have presented with carcinoma hemorrhagiectoides–associated shield sign.
Authors’ Response
We appreciate and welcome the comments provided by the authors. Drawing attention to unusual pathologic manifestations of cutaneous metastatic salivary duct carcinoma manifesting with the shield sign, the authors present a comprehensive review of 3 distinctive presentations: carcinoma erysipelatoides, carcinoma telangiectoides, and carcinoma hemorrhagiectoides. The inclusion of these variants enriches the discussion and makes this letter a valuable addition to the literature on cutaneous metastatic carcinoma, particularly metastatic salivary duct carcinoma.
Xintong Wang, MD; William H. Westra, MD
From the Department of Pathology, Icahn School of Medicine at Mount Sinai, New York, New York.
The authors report no conflict of interest.
- Wang X, Vyas NS, Alghamdi AA, et al. Cutaneous presentation of metastatic salivary duct carcinoma. Cutis. 2023;112:E13-E15.
- Pollock JL, Catalano E. Metastatic ductal carcinoma of the parotid gland in a patient with sarcoidosis. Arch Dermatol. 1979;115:1098-1099.
- Pollock JL. Metastatic carcinoma of the parotid gland resembling carcinoma of the breast. J Am Acad Dermatol. 1996;34:1093.
- Aygit AC, Top H, Cakir B, et al. Salivary duct carcinoma of the parotid gland metastasizing to the skin: a case report and review of the literature. Am J Dermatopathol. 2005;27:48-50.
- Cohen PR, Prieto VG, Piha-Paul SA, et al. The “shield sign” in two men with metastatic salivary duct carcinoma to the skin: cutaneous metastases presenting as carcinoma hemorrhagiectoides. J Clin Aesthet Dermatol. 2012;5:27-36.
- Chakari W, Andersen L, Anderson JL. Cutaneous metastases from salivary duct carcinoma of the submandibular gland. Case Rep Dermatol. 2017;9:254-258.
- Shin JY, Eun DH, Lee JY, et al. A case of cutaneous metastases of salivary duct carcinoma mimicking radiation recall dermatitis. Ann Dermatol. 2020;32:436-438.
- Aravena RC, Aravena DC, Velasco MJ, et al. Carcinoma hemorrhagiectoides: case report of an uncommon presentation of cutaneous metastatic breast carcinoma. Dermatol Online J. 2017;23:13030/qt3hn3z850.
- Smith KA, Basko-Plluska J, Kothari AD, et al. Cutaneous metastatic breast adenocarcinoma. Cutis. 2020;105:E20-E22.
- Cohen PR, Kurzrock R. Cutaneous metastatic cancer: carcinoma hemorrhagiectoides presenting as the shield sign. Cureus. 2021;13:e12627.
- Cohen PR. Pleomorphic appearance of breast cancer cutaneous metastases. Cureus. 2021;13:e20301.
- Cohen PR, Prieto VG, Kurzrock R. Tumor lysis syndrome: introduction of a cutaneous variant and a new classification system. Cureus. 2021;13:e13816.
To the Editor:
We read with interest the Case Letter from Wang et al1 (Cutis. 2023;112:E13-E15) of a 60-year-old man whose metastatic salivary duct adenocarcinoma manifested with the shield sign as well as carcinoma hemorrhagiectoides. Cutaneous metastases have seldom been described in association with salivary duct carcinoma.2-7 In addition, carcinoma hemorrhagiectoides–associated shield sign has not been commonly reported.5,8-12
Salivary duct carcinoma—an uncommon head and neck malignancy characterized by androgen receptor expression—rarely is associated with cutaneous metastases. Based on a PubMed search of articles indexed for MEDLINE using the terms cutaneous, metastatic, salivary duct carcinoma, and/or skin, including the patient described by Wang et al,1 there have been 8 individuals with cutaneous metastases from this cancer. The morphology of the cutaneous metastases has varied from angiomatous to angiokeratomalike (black and keratotic) papules, bullae, macules (red), papules and nodules (erythematous and scaly), plaques (cellulitislike and confluent that were purpuric, hemorrhagic, and violaceous), pseudovesicles, purpuric papules, subcutaneous nodules, and an ulcer (superficial and mimicked a basal cell carcinoma).1-7 Remarkably, 4 of 8 patients (50%) with salivary duct carcinoma cutaneous metastases presented with a shield sign,5,7 including the case reported by Wang et al.1
The shield sign is a distinctive clinical manifestation of cutaneous metastasis.10 It was named to describe the skin metastases located predominantly on the chest area that would be covered by a medieval knight’s shield5,10,12; metastatic lesions also have been noted on the proximal arm and/or the upper back in a similar distribution.8,9 To date, based on a PubMed search of articles indexed for MEDLINE using the search terms breast cancer, carcinoma, hemorrhagiectoides, metastases, salivary duct carcinoma, shield, and/or sign, the shield sign has been described in 6 patients with cutaneous metastases either from salivary duct carcinoma (4 patients)1,5,7 or breast cancer (2 patients).8,9 The shield sign pathologically corresponds to carcinoma hemorrhagiectoides, an inflammatory pattern of cutaneous metastases.5,11
Inflammatory cutaneous metastatic carcinoma has 3 distinctive clinical and pathologic manifestations.11 Carcinoma erysipelatoides and carcinoma telangiectoides were the earlier described variants.11 In 2012, carcinoma hemorrhagiectoides was described as the third pattern of inflammatory cutaneous metastasis.5
Carcinoma erysipelatoides, which clinically mimics cutaneous streptococcal cellulitis, appears as a well-defined erythematous patch or plaque; the tumor cells can be found in the lymphatic vessels and either are absent or minimally present in the dermis. Carcinoma telangiectoides, which clinically mimics idiopathic telangiectases, appears as an erythematous patch with prominent telangiectases; the tumor cells can be found in the blood vessels and are either absent or minimally present in the dermis. Carcinoma hemorrhagiectoides appears as purpuric or violaceous indurated plaques; the tumor cells are not only found in the blood vessels, in the lymphatic vessels, or both, but also can be mildly to extensively present in the dermis.5,10,11
In conclusion, the shield sign is a unique presentation of inflammatory cutaneous metastatic carcinoma, which is associated with carcinoma hemorrhagiectoides. The clinical features of the infiltrated plaques correspond to the presence of tumor cells in the blood vessels, lymphatic vessels, and the dermis; in addition, the purpuric and violaceous appearance correlates with the presence of extravasated erythrocytes or hemorrhage in the dermis. To date, half of the patients with skin metastases from salivary duct carcinoma have presented with carcinoma hemorrhagiectoides–associated shield sign.
Authors’ Response
We appreciate and welcome the comments provided by the authors. Drawing attention to unusual pathologic manifestations of cutaneous metastatic salivary duct carcinoma manifesting with the shield sign, the authors present a comprehensive review of 3 distinctive presentations: carcinoma erysipelatoides, carcinoma telangiectoides, and carcinoma hemorrhagiectoides. The inclusion of these variants enriches the discussion and makes this letter a valuable addition to the literature on cutaneous metastatic carcinoma, particularly metastatic salivary duct carcinoma.
Xintong Wang, MD; William H. Westra, MD
From the Department of Pathology, Icahn School of Medicine at Mount Sinai, New York, New York.
The authors report no conflict of interest.
To the Editor:
We read with interest the Case Letter from Wang et al1 (Cutis. 2023;112:E13-E15) of a 60-year-old man whose metastatic salivary duct adenocarcinoma manifested with the shield sign as well as carcinoma hemorrhagiectoides. Cutaneous metastases have seldom been described in association with salivary duct carcinoma.2-7 In addition, carcinoma hemorrhagiectoides–associated shield sign has not been commonly reported.5,8-12
Salivary duct carcinoma—an uncommon head and neck malignancy characterized by androgen receptor expression—rarely is associated with cutaneous metastases. Based on a PubMed search of articles indexed for MEDLINE using the terms cutaneous, metastatic, salivary duct carcinoma, and/or skin, including the patient described by Wang et al,1 there have been 8 individuals with cutaneous metastases from this cancer. The morphology of the cutaneous metastases has varied from angiomatous to angiokeratomalike (black and keratotic) papules, bullae, macules (red), papules and nodules (erythematous and scaly), plaques (cellulitislike and confluent that were purpuric, hemorrhagic, and violaceous), pseudovesicles, purpuric papules, subcutaneous nodules, and an ulcer (superficial and mimicked a basal cell carcinoma).1-7 Remarkably, 4 of 8 patients (50%) with salivary duct carcinoma cutaneous metastases presented with a shield sign,5,7 including the case reported by Wang et al.1
The shield sign is a distinctive clinical manifestation of cutaneous metastasis.10 It was named to describe the skin metastases located predominantly on the chest area that would be covered by a medieval knight’s shield5,10,12; metastatic lesions also have been noted on the proximal arm and/or the upper back in a similar distribution.8,9 To date, based on a PubMed search of articles indexed for MEDLINE using the search terms breast cancer, carcinoma, hemorrhagiectoides, metastases, salivary duct carcinoma, shield, and/or sign, the shield sign has been described in 6 patients with cutaneous metastases either from salivary duct carcinoma (4 patients)1,5,7 or breast cancer (2 patients).8,9 The shield sign pathologically corresponds to carcinoma hemorrhagiectoides, an inflammatory pattern of cutaneous metastases.5,11
Inflammatory cutaneous metastatic carcinoma has 3 distinctive clinical and pathologic manifestations.11 Carcinoma erysipelatoides and carcinoma telangiectoides were the earlier described variants.11 In 2012, carcinoma hemorrhagiectoides was described as the third pattern of inflammatory cutaneous metastasis.5
Carcinoma erysipelatoides, which clinically mimics cutaneous streptococcal cellulitis, appears as a well-defined erythematous patch or plaque; the tumor cells can be found in the lymphatic vessels and either are absent or minimally present in the dermis. Carcinoma telangiectoides, which clinically mimics idiopathic telangiectases, appears as an erythematous patch with prominent telangiectases; the tumor cells can be found in the blood vessels and are either absent or minimally present in the dermis. Carcinoma hemorrhagiectoides appears as purpuric or violaceous indurated plaques; the tumor cells are not only found in the blood vessels, in the lymphatic vessels, or both, but also can be mildly to extensively present in the dermis.5,10,11
In conclusion, the shield sign is a unique presentation of inflammatory cutaneous metastatic carcinoma, which is associated with carcinoma hemorrhagiectoides. The clinical features of the infiltrated plaques correspond to the presence of tumor cells in the blood vessels, lymphatic vessels, and the dermis; in addition, the purpuric and violaceous appearance correlates with the presence of extravasated erythrocytes or hemorrhage in the dermis. To date, half of the patients with skin metastases from salivary duct carcinoma have presented with carcinoma hemorrhagiectoides–associated shield sign.
Authors’ Response
We appreciate and welcome the comments provided by the authors. Drawing attention to unusual pathologic manifestations of cutaneous metastatic salivary duct carcinoma manifesting with the shield sign, the authors present a comprehensive review of 3 distinctive presentations: carcinoma erysipelatoides, carcinoma telangiectoides, and carcinoma hemorrhagiectoides. The inclusion of these variants enriches the discussion and makes this letter a valuable addition to the literature on cutaneous metastatic carcinoma, particularly metastatic salivary duct carcinoma.
Xintong Wang, MD; William H. Westra, MD
From the Department of Pathology, Icahn School of Medicine at Mount Sinai, New York, New York.
The authors report no conflict of interest.
- Wang X, Vyas NS, Alghamdi AA, et al. Cutaneous presentation of metastatic salivary duct carcinoma. Cutis. 2023;112:E13-E15.
- Pollock JL, Catalano E. Metastatic ductal carcinoma of the parotid gland in a patient with sarcoidosis. Arch Dermatol. 1979;115:1098-1099.
- Pollock JL. Metastatic carcinoma of the parotid gland resembling carcinoma of the breast. J Am Acad Dermatol. 1996;34:1093.
- Aygit AC, Top H, Cakir B, et al. Salivary duct carcinoma of the parotid gland metastasizing to the skin: a case report and review of the literature. Am J Dermatopathol. 2005;27:48-50.
- Cohen PR, Prieto VG, Piha-Paul SA, et al. The “shield sign” in two men with metastatic salivary duct carcinoma to the skin: cutaneous metastases presenting as carcinoma hemorrhagiectoides. J Clin Aesthet Dermatol. 2012;5:27-36.
- Chakari W, Andersen L, Anderson JL. Cutaneous metastases from salivary duct carcinoma of the submandibular gland. Case Rep Dermatol. 2017;9:254-258.
- Shin JY, Eun DH, Lee JY, et al. A case of cutaneous metastases of salivary duct carcinoma mimicking radiation recall dermatitis. Ann Dermatol. 2020;32:436-438.
- Aravena RC, Aravena DC, Velasco MJ, et al. Carcinoma hemorrhagiectoides: case report of an uncommon presentation of cutaneous metastatic breast carcinoma. Dermatol Online J. 2017;23:13030/qt3hn3z850.
- Smith KA, Basko-Plluska J, Kothari AD, et al. Cutaneous metastatic breast adenocarcinoma. Cutis. 2020;105:E20-E22.
- Cohen PR, Kurzrock R. Cutaneous metastatic cancer: carcinoma hemorrhagiectoides presenting as the shield sign. Cureus. 2021;13:e12627.
- Cohen PR. Pleomorphic appearance of breast cancer cutaneous metastases. Cureus. 2021;13:e20301.
- Cohen PR, Prieto VG, Kurzrock R. Tumor lysis syndrome: introduction of a cutaneous variant and a new classification system. Cureus. 2021;13:e13816.
- Wang X, Vyas NS, Alghamdi AA, et al. Cutaneous presentation of metastatic salivary duct carcinoma. Cutis. 2023;112:E13-E15.
- Pollock JL, Catalano E. Metastatic ductal carcinoma of the parotid gland in a patient with sarcoidosis. Arch Dermatol. 1979;115:1098-1099.
- Pollock JL. Metastatic carcinoma of the parotid gland resembling carcinoma of the breast. J Am Acad Dermatol. 1996;34:1093.
- Aygit AC, Top H, Cakir B, et al. Salivary duct carcinoma of the parotid gland metastasizing to the skin: a case report and review of the literature. Am J Dermatopathol. 2005;27:48-50.
- Cohen PR, Prieto VG, Piha-Paul SA, et al. The “shield sign” in two men with metastatic salivary duct carcinoma to the skin: cutaneous metastases presenting as carcinoma hemorrhagiectoides. J Clin Aesthet Dermatol. 2012;5:27-36.
- Chakari W, Andersen L, Anderson JL. Cutaneous metastases from salivary duct carcinoma of the submandibular gland. Case Rep Dermatol. 2017;9:254-258.
- Shin JY, Eun DH, Lee JY, et al. A case of cutaneous metastases of salivary duct carcinoma mimicking radiation recall dermatitis. Ann Dermatol. 2020;32:436-438.
- Aravena RC, Aravena DC, Velasco MJ, et al. Carcinoma hemorrhagiectoides: case report of an uncommon presentation of cutaneous metastatic breast carcinoma. Dermatol Online J. 2017;23:13030/qt3hn3z850.
- Smith KA, Basko-Plluska J, Kothari AD, et al. Cutaneous metastatic breast adenocarcinoma. Cutis. 2020;105:E20-E22.
- Cohen PR, Kurzrock R. Cutaneous metastatic cancer: carcinoma hemorrhagiectoides presenting as the shield sign. Cureus. 2021;13:e12627.
- Cohen PR. Pleomorphic appearance of breast cancer cutaneous metastases. Cureus. 2021;13:e20301.
- Cohen PR, Prieto VG, Kurzrock R. Tumor lysis syndrome: introduction of a cutaneous variant and a new classification system. Cureus. 2021;13:e13816.






