User login
RA triple therapy wins cost-effectiveness comparison vs. etanercept plus methotrexate
SAN DIEGO – In patients with early rheumatoid arthritis, treatment with a combination of disease-modifying antirheumatic drugs is more cost effective than an immediate or step-up approach using etanercept and methotrexate, a new analysis demonstrated.
"The benefits from all strategies were comparable, but biologics strategies were almost twice as expensive [as] triple strategies, producing incremental cost-effectiveness ratios higher than what most health care settings would find acceptable," Kaleb Michaud, Ph.D., said at the annual meeting of the American College of Rheumatology.
Dr. Michaud of the division of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, and his associates analyzed data from 755 patients enrolled in the randomized, 2-year TEAR (Treatment of Early Aggressive RA) trial (Arthritis Rheum. 2012;64:2824-35), which compared the long-term effectiveness of using a triple therapy approach for treating RA with a strategy of using a etanercept (Enbrel) plus methotrexate. They studied four approaches: immediate triple therapy of methotrexate, sulfasalazine, and hydroxychloroquine; immediate treatment with etanercept and methotrexate; a step-up triple therapy; and a step-up etanercept therapy. The researchers incorporated estimates of annual discontinuation rates of triple therapy and etanercept of 22% and 10%, respectively, based on data from the National Data Bank for Rheumatic Diseases.
The analysis was limited to patients with RA duration of less than 3 years and those with active disease, defined as having four or more swollen joints with seropositivity and/or erosions. The mean disease duration was 3.6 months and the mean Disease Activity Score was 5.8. The researchers used a societal perspective and a Markov cohort model with 3% discounting to estimate quality-adjusted life year (QALY) measurements and the costs associated with therapy approaches in the TEAR trial.
Dr. Michaud, who is also codirector of the National Data Bank for Rheumatic Diseases, reported that the lifetime benefit of all four treatment approaches were comparable, within 0.06 QALYs of each other. However, the therapies that included etanercept were nearly twice as expensive because of the higher cost of the TNF inhibitor. Specifically, the costs were $152,400 in the immediate triple therapy group and $154,900 in the step-up triple therapy group, compared with $269,500 in the step-up etanercept group and $338,100 in the immediate etanercept group. The researchers determined that the lifetime incremental cost-effectiveness ratio of immediate etanercept to immediate triple therapy was $837,100/QALY.
"The immediate triple therapy dominated other arms at 1 and 2 years in this trial, and its results were most sensitive to the costs of etanercept and the reduction in triple therapy discontinuation rates," Dr. Michaud commented. "Our sensitivity analysis did not change our conclusions."
Dr. Michaud disclosed that the study was supported in part by a 2012 investigator award from the Rheumatology Research Foundation.
SAN DIEGO – In patients with early rheumatoid arthritis, treatment with a combination of disease-modifying antirheumatic drugs is more cost effective than an immediate or step-up approach using etanercept and methotrexate, a new analysis demonstrated.
"The benefits from all strategies were comparable, but biologics strategies were almost twice as expensive [as] triple strategies, producing incremental cost-effectiveness ratios higher than what most health care settings would find acceptable," Kaleb Michaud, Ph.D., said at the annual meeting of the American College of Rheumatology.
Dr. Michaud of the division of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, and his associates analyzed data from 755 patients enrolled in the randomized, 2-year TEAR (Treatment of Early Aggressive RA) trial (Arthritis Rheum. 2012;64:2824-35), which compared the long-term effectiveness of using a triple therapy approach for treating RA with a strategy of using a etanercept (Enbrel) plus methotrexate. They studied four approaches: immediate triple therapy of methotrexate, sulfasalazine, and hydroxychloroquine; immediate treatment with etanercept and methotrexate; a step-up triple therapy; and a step-up etanercept therapy. The researchers incorporated estimates of annual discontinuation rates of triple therapy and etanercept of 22% and 10%, respectively, based on data from the National Data Bank for Rheumatic Diseases.
The analysis was limited to patients with RA duration of less than 3 years and those with active disease, defined as having four or more swollen joints with seropositivity and/or erosions. The mean disease duration was 3.6 months and the mean Disease Activity Score was 5.8. The researchers used a societal perspective and a Markov cohort model with 3% discounting to estimate quality-adjusted life year (QALY) measurements and the costs associated with therapy approaches in the TEAR trial.
Dr. Michaud, who is also codirector of the National Data Bank for Rheumatic Diseases, reported that the lifetime benefit of all four treatment approaches were comparable, within 0.06 QALYs of each other. However, the therapies that included etanercept were nearly twice as expensive because of the higher cost of the TNF inhibitor. Specifically, the costs were $152,400 in the immediate triple therapy group and $154,900 in the step-up triple therapy group, compared with $269,500 in the step-up etanercept group and $338,100 in the immediate etanercept group. The researchers determined that the lifetime incremental cost-effectiveness ratio of immediate etanercept to immediate triple therapy was $837,100/QALY.
"The immediate triple therapy dominated other arms at 1 and 2 years in this trial, and its results were most sensitive to the costs of etanercept and the reduction in triple therapy discontinuation rates," Dr. Michaud commented. "Our sensitivity analysis did not change our conclusions."
Dr. Michaud disclosed that the study was supported in part by a 2012 investigator award from the Rheumatology Research Foundation.
SAN DIEGO – In patients with early rheumatoid arthritis, treatment with a combination of disease-modifying antirheumatic drugs is more cost effective than an immediate or step-up approach using etanercept and methotrexate, a new analysis demonstrated.
"The benefits from all strategies were comparable, but biologics strategies were almost twice as expensive [as] triple strategies, producing incremental cost-effectiveness ratios higher than what most health care settings would find acceptable," Kaleb Michaud, Ph.D., said at the annual meeting of the American College of Rheumatology.
Dr. Michaud of the division of rheumatology and immunology at the University of Nebraska Medical Center, Omaha, and his associates analyzed data from 755 patients enrolled in the randomized, 2-year TEAR (Treatment of Early Aggressive RA) trial (Arthritis Rheum. 2012;64:2824-35), which compared the long-term effectiveness of using a triple therapy approach for treating RA with a strategy of using a etanercept (Enbrel) plus methotrexate. They studied four approaches: immediate triple therapy of methotrexate, sulfasalazine, and hydroxychloroquine; immediate treatment with etanercept and methotrexate; a step-up triple therapy; and a step-up etanercept therapy. The researchers incorporated estimates of annual discontinuation rates of triple therapy and etanercept of 22% and 10%, respectively, based on data from the National Data Bank for Rheumatic Diseases.
The analysis was limited to patients with RA duration of less than 3 years and those with active disease, defined as having four or more swollen joints with seropositivity and/or erosions. The mean disease duration was 3.6 months and the mean Disease Activity Score was 5.8. The researchers used a societal perspective and a Markov cohort model with 3% discounting to estimate quality-adjusted life year (QALY) measurements and the costs associated with therapy approaches in the TEAR trial.
Dr. Michaud, who is also codirector of the National Data Bank for Rheumatic Diseases, reported that the lifetime benefit of all four treatment approaches were comparable, within 0.06 QALYs of each other. However, the therapies that included etanercept were nearly twice as expensive because of the higher cost of the TNF inhibitor. Specifically, the costs were $152,400 in the immediate triple therapy group and $154,900 in the step-up triple therapy group, compared with $269,500 in the step-up etanercept group and $338,100 in the immediate etanercept group. The researchers determined that the lifetime incremental cost-effectiveness ratio of immediate etanercept to immediate triple therapy was $837,100/QALY.
"The immediate triple therapy dominated other arms at 1 and 2 years in this trial, and its results were most sensitive to the costs of etanercept and the reduction in triple therapy discontinuation rates," Dr. Michaud commented. "Our sensitivity analysis did not change our conclusions."
Dr. Michaud disclosed that the study was supported in part by a 2012 investigator award from the Rheumatology Research Foundation.
AT THE ACR ANNUAL MEETING
Major finding: The incremental cost-effectiveness ratio of immediate etanercept to immediate triple therapy was $837,100/quality-adjusted life year.
Data source: An analysis of 755 patients enrolled in the randomized, 2-year Treatment of Early Aggressive RA (TEAR) trial and patient-level data from the National Data Bank for Rheumatic Diseases.
Disclosures: Dr. Michaud disclosed that the study was supported in part by a 2012 investigator award from the Rheumatology Research Foundation.
Delayed-release prednisone lessened rheumatoid arthritis symptoms
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
AT THE ACR ANNUAL MEETING
Major finding: Patients with moderate to severe active rheumatoid arthritis on stable disease-modifying antirheumatic drug therapy experienced a greater than 60-minute reduction in daily morning stiffness from a baseline of 143.5 minutes when they were switched from immediate- to delayed-release prednisone.
Data source: This was an analysis of the 207 RA patients who participated in the 9-month open-label phase of the prospective CAPRA-1 study.
Disclosures: The study was funded by Horizon Pharma, which markets immediate-release prednisone. Dr. Gibofsky is a consultant to the company.
Encouraging results for oral JAK 3 inhibitor in rheumatoid arthritis
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.
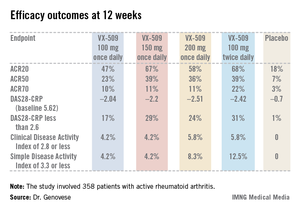
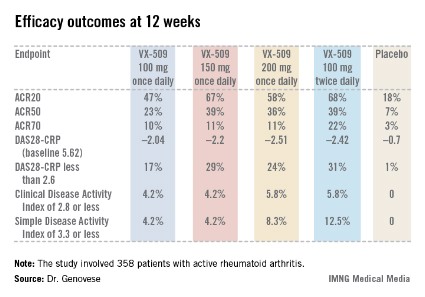
An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.

An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.

An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
AT THE ACR ANNUAL MEETING
Major finding: Rheumatoid arthritis patients had ACR20 responses of 47%-68% to various dosing regimens of the novel oral Janus kinase 3 inhibitor VX-509 at 12 weeks, compared with 18% in placebo-treated controls.
Data source: This was a phase IIb, double-blind, placebo-controlled, international study involving 358 patients with rheumatoid arthritis on background stable doses of methotrexate, continued during the trial.
Disclosures: The study was sponsored by Vertex Pharmaceuticals. The presenter disclosed receiving research grants from and serving as a consultant to Vertex.
Almost 23% of U.S. adults have arthritis
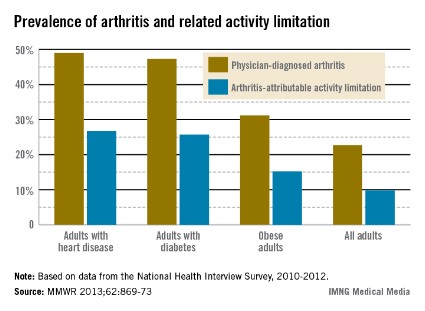
In 2010-2012, almost 23% of the adult population had physician-diagnosed arthritis and almost 10% had some form of activity limitation attributable to their arthritis, the Centers for Disease Control and Prevention reported.
The figures for adults aged 18 years and older with heart disease, diabetes, and obesity were even higher. Data from the National Health Interview Survey (NHIS) put arthritis prevalence at 49% for those with heart disease, 47% for those with diabetes, and 31% for those who are obese, according to the CDC (MMWR 2013;62:869-73).
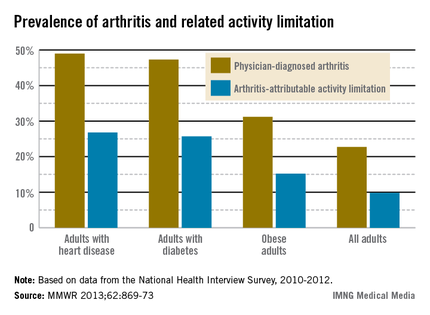
Among patients with heart disease, nearly 27% had arthritis-attributable activity limitation, as did almost 26% of those with diabetes and just over 15% of those who were obese, the CDC said.
The estimate of 52.5 million adults with arthritis "is consistent with an earlier projection and suggests that projections of 55.7 million adults with arthritis by 2015 and 67 million by 2030 are reasonable," the CDC noted, adding that the projected number of those with activity limitations (22.7 million), however, "exceeds the earlier projection of 22 million adults ... by 2020 and, therefore, might exceed the 25 million projected for 2030."
In 2010-2012, almost 23% of the adult population had physician-diagnosed arthritis and almost 10% had some form of activity limitation attributable to their arthritis, the Centers for Disease Control and Prevention reported.
The figures for adults aged 18 years and older with heart disease, diabetes, and obesity were even higher. Data from the National Health Interview Survey (NHIS) put arthritis prevalence at 49% for those with heart disease, 47% for those with diabetes, and 31% for those who are obese, according to the CDC (MMWR 2013;62:869-73).

Among patients with heart disease, nearly 27% had arthritis-attributable activity limitation, as did almost 26% of those with diabetes and just over 15% of those who were obese, the CDC said.
The estimate of 52.5 million adults with arthritis "is consistent with an earlier projection and suggests that projections of 55.7 million adults with arthritis by 2015 and 67 million by 2030 are reasonable," the CDC noted, adding that the projected number of those with activity limitations (22.7 million), however, "exceeds the earlier projection of 22 million adults ... by 2020 and, therefore, might exceed the 25 million projected for 2030."
In 2010-2012, almost 23% of the adult population had physician-diagnosed arthritis and almost 10% had some form of activity limitation attributable to their arthritis, the Centers for Disease Control and Prevention reported.
The figures for adults aged 18 years and older with heart disease, diabetes, and obesity were even higher. Data from the National Health Interview Survey (NHIS) put arthritis prevalence at 49% for those with heart disease, 47% for those with diabetes, and 31% for those who are obese, according to the CDC (MMWR 2013;62:869-73).

Among patients with heart disease, nearly 27% had arthritis-attributable activity limitation, as did almost 26% of those with diabetes and just over 15% of those who were obese, the CDC said.
The estimate of 52.5 million adults with arthritis "is consistent with an earlier projection and suggests that projections of 55.7 million adults with arthritis by 2015 and 67 million by 2030 are reasonable," the CDC noted, adding that the projected number of those with activity limitations (22.7 million), however, "exceeds the earlier projection of 22 million adults ... by 2020 and, therefore, might exceed the 25 million projected for 2030."
FROM MMWR
Soda pop boosts rheumatoid arthritis risk
SAN DIEGO – Women who regularly drink one or more sugar-sweetened soft drinks per day are at heightened risk of developing seropositive rheumatoid arthritis, according to a massive analysis of combined data from the prospective-cohort Nurses’ Health Study and Nurses’ Health Study II.
During 1.9 million person-years of prospective follow-up in the Nurses' Health Study (NHS) and 1.5 million person-years in NHS II, there were 563 confirmed cases of new-onset seropositive rheumatoid arthritis and 320 cases of seronegative rheumatoid arthritis. The two studies collectively enrolled 238,131 U.S. female registered nurses who periodically filled out validated food frequency questionnaires.
In a multivariate analysis adjusted for numerous potential confounding variables, women who reported drinking an average of one or more non-diet sodas per day had a 71% greater risk of developing seropositive rheumatoid arthritis during follow-up than did those who drank none or less than one per month (P = .005), Yang Hu reported at the annual meeting of the American College of Rheumatology.
It’s noteworthy that the increased risk was present at a cut point of one daily serving, a level of consumption many Americans wouldn’t view as excessive.
The mechanism for the observed association between frequent soft drink consumption and seropositive rheumatoid arthritis remains unclear. The investigators decided to look at sugar-containing soda consumption because it has been shown to be associated with overweight and obesity, type 2 diabetes, and cardiovascular disease. And since obesity has previously been shown to be associated with increased risk of rheumatoid arthritis, the thinking was that obesity might mediate the link between soda consumption and rheumatoid arthritis. Daily soda consumption, however, was independently associated with a 71% increase in risk even after controlling for body mass index in the multivariate analysis, noted Mr. Hu of Brigham and Women’s Hospital, Boston.
Other potential confounders that were adjusted for in the analysis included age, smoking, alcohol consumption, physical activity, reproductive factors, multivitamin use, income level, and diet quality.
The NHS and NHS II studies are funded by the National Institutes of Health. Mr. Hu reported having no financial conflicts of interest, but one of his associates disclosed relationships with several pharmaceutical companies.
*Correction, 11/15/2013: An earlier version of the article misstated the number of nurses involved in the analysis.
SAN DIEGO – Women who regularly drink one or more sugar-sweetened soft drinks per day are at heightened risk of developing seropositive rheumatoid arthritis, according to a massive analysis of combined data from the prospective-cohort Nurses’ Health Study and Nurses’ Health Study II.
During 1.9 million person-years of prospective follow-up in the Nurses' Health Study (NHS) and 1.5 million person-years in NHS II, there were 563 confirmed cases of new-onset seropositive rheumatoid arthritis and 320 cases of seronegative rheumatoid arthritis. The two studies collectively enrolled 238,131 U.S. female registered nurses who periodically filled out validated food frequency questionnaires.
In a multivariate analysis adjusted for numerous potential confounding variables, women who reported drinking an average of one or more non-diet sodas per day had a 71% greater risk of developing seropositive rheumatoid arthritis during follow-up than did those who drank none or less than one per month (P = .005), Yang Hu reported at the annual meeting of the American College of Rheumatology.
It’s noteworthy that the increased risk was present at a cut point of one daily serving, a level of consumption many Americans wouldn’t view as excessive.
The mechanism for the observed association between frequent soft drink consumption and seropositive rheumatoid arthritis remains unclear. The investigators decided to look at sugar-containing soda consumption because it has been shown to be associated with overweight and obesity, type 2 diabetes, and cardiovascular disease. And since obesity has previously been shown to be associated with increased risk of rheumatoid arthritis, the thinking was that obesity might mediate the link between soda consumption and rheumatoid arthritis. Daily soda consumption, however, was independently associated with a 71% increase in risk even after controlling for body mass index in the multivariate analysis, noted Mr. Hu of Brigham and Women’s Hospital, Boston.
Other potential confounders that were adjusted for in the analysis included age, smoking, alcohol consumption, physical activity, reproductive factors, multivitamin use, income level, and diet quality.
The NHS and NHS II studies are funded by the National Institutes of Health. Mr. Hu reported having no financial conflicts of interest, but one of his associates disclosed relationships with several pharmaceutical companies.
*Correction, 11/15/2013: An earlier version of the article misstated the number of nurses involved in the analysis.
SAN DIEGO – Women who regularly drink one or more sugar-sweetened soft drinks per day are at heightened risk of developing seropositive rheumatoid arthritis, according to a massive analysis of combined data from the prospective-cohort Nurses’ Health Study and Nurses’ Health Study II.
During 1.9 million person-years of prospective follow-up in the Nurses' Health Study (NHS) and 1.5 million person-years in NHS II, there were 563 confirmed cases of new-onset seropositive rheumatoid arthritis and 320 cases of seronegative rheumatoid arthritis. The two studies collectively enrolled 238,131 U.S. female registered nurses who periodically filled out validated food frequency questionnaires.
In a multivariate analysis adjusted for numerous potential confounding variables, women who reported drinking an average of one or more non-diet sodas per day had a 71% greater risk of developing seropositive rheumatoid arthritis during follow-up than did those who drank none or less than one per month (P = .005), Yang Hu reported at the annual meeting of the American College of Rheumatology.
It’s noteworthy that the increased risk was present at a cut point of one daily serving, a level of consumption many Americans wouldn’t view as excessive.
The mechanism for the observed association between frequent soft drink consumption and seropositive rheumatoid arthritis remains unclear. The investigators decided to look at sugar-containing soda consumption because it has been shown to be associated with overweight and obesity, type 2 diabetes, and cardiovascular disease. And since obesity has previously been shown to be associated with increased risk of rheumatoid arthritis, the thinking was that obesity might mediate the link between soda consumption and rheumatoid arthritis. Daily soda consumption, however, was independently associated with a 71% increase in risk even after controlling for body mass index in the multivariate analysis, noted Mr. Hu of Brigham and Women’s Hospital, Boston.
Other potential confounders that were adjusted for in the analysis included age, smoking, alcohol consumption, physical activity, reproductive factors, multivitamin use, income level, and diet quality.
The NHS and NHS II studies are funded by the National Institutes of Health. Mr. Hu reported having no financial conflicts of interest, but one of his associates disclosed relationships with several pharmaceutical companies.
*Correction, 11/15/2013: An earlier version of the article misstated the number of nurses involved in the analysis.
AT THE ACR ANNUAL MEETING
Major finding: Women who reported drinking an average of one or more sugar-containing soft drinks per day had a highly significant 71% increased risk of developing seropositive rheumatoid arthritis during prospective follow-up than those who drank none or less than one serving per month.
Data source: This was an analysis of 238,131 American female nurses* prospectively followed for 3.4 million person-years in the Nurses’ Health Study and Nurses’ Health Study II.
Disclosures: The studies were funded by the National Institutes of Health. Mr. Hu reported having no financial conflicts.
Orthopedic surgery among RA patients declining, study finds
SAN DIEGO – The overall proportion of rheumatoid arthritis patients undergoing orthopedic surgery is declining as intensive pharmacologic intervention with agents such as biologics play an increasingly prominent role in the treatment of the inflammatory disease, judging from results from a large, long-term study.
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat and his associates enrolled 2,342 men and women from RA patient registries in Sweden who filled out questionnaires including visual analog scales for general health and pain, and the Health Assessment Questionnaire (HAQ) in 1997, 2002, 2005, and 2009.
The response rate ranged from 62% to 74%, and the researchers linked responses to Swedish national health registry records to correlate the data with records of inpatient and outpatient surgeries, as well as the use of biologics. They went on to evaluate the incidence rate of orthopedic surgery in three time periods: from 1998-2001 (time period A), 2002-2006 (time period B), and 2007-2011 (time period C).
Dr. Hekmat, a rheumatologist and a PhD fellow at Malmö University, Sweden, reported that between 1998 and 2011 the incidence of all orthopedic procedures performed was 82.3 per 1,000 person-years. Significant declines were observed over the three time periods studied. For example, the incidence of any orthopedic surgery declined from 94.6 per 1,000 person-years in time period A to 82.6 per 1,000 person-years in time period B, and 71.8 per 1,000 person-years in time period C (P less than .001).
A similar association was observed for hip surgery alone (which fell from 27.8 per 1,000 person-years in time period A to 17.6 per 1,000 person-years in time period C; P less than .001). The incidence of orthopedic surgery on small joints such as hands, wrists, feet, and ankles fell from 43.8 per 1,000 person-years in time period A to 30.5 per 1,000 person-years in time period C (P less than .001).
No significant decline occurred in the incidence of knee surgeries performed during the study period. This actually rose slightly from 12.3 per 1,000 person-years in time period A to 12.9 per 1,000 person-years in time period C (P = .759).
Independent predictors of undergoing any orthopedic surgery included being female (hazard ratio 1.50) and having greater disability as measured by the HAQ (HR 1.37).
Dr. Hekmat speculated that the rate of orthopedic surgery was reduced by early intensive treatment in patients with RA, since the time period studied coincided with the increased use of pharmacologic interventions. "In this cohort we are targeting the patients more aggressively," he said. "It’s not wrong to guess that this decline may lead to better [disease] management."
Dr. Hekmat said that he had no relevant financial conflicts to disclose.
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat, Health Assessment Questionnaire, Malmö University, Sweden, orthopedic procedures,
SAN DIEGO – The overall proportion of rheumatoid arthritis patients undergoing orthopedic surgery is declining as intensive pharmacologic intervention with agents such as biologics play an increasingly prominent role in the treatment of the inflammatory disease, judging from results from a large, long-term study.
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat and his associates enrolled 2,342 men and women from RA patient registries in Sweden who filled out questionnaires including visual analog scales for general health and pain, and the Health Assessment Questionnaire (HAQ) in 1997, 2002, 2005, and 2009.
The response rate ranged from 62% to 74%, and the researchers linked responses to Swedish national health registry records to correlate the data with records of inpatient and outpatient surgeries, as well as the use of biologics. They went on to evaluate the incidence rate of orthopedic surgery in three time periods: from 1998-2001 (time period A), 2002-2006 (time period B), and 2007-2011 (time period C).
Dr. Hekmat, a rheumatologist and a PhD fellow at Malmö University, Sweden, reported that between 1998 and 2011 the incidence of all orthopedic procedures performed was 82.3 per 1,000 person-years. Significant declines were observed over the three time periods studied. For example, the incidence of any orthopedic surgery declined from 94.6 per 1,000 person-years in time period A to 82.6 per 1,000 person-years in time period B, and 71.8 per 1,000 person-years in time period C (P less than .001).
A similar association was observed for hip surgery alone (which fell from 27.8 per 1,000 person-years in time period A to 17.6 per 1,000 person-years in time period C; P less than .001). The incidence of orthopedic surgery on small joints such as hands, wrists, feet, and ankles fell from 43.8 per 1,000 person-years in time period A to 30.5 per 1,000 person-years in time period C (P less than .001).
No significant decline occurred in the incidence of knee surgeries performed during the study period. This actually rose slightly from 12.3 per 1,000 person-years in time period A to 12.9 per 1,000 person-years in time period C (P = .759).
Independent predictors of undergoing any orthopedic surgery included being female (hazard ratio 1.50) and having greater disability as measured by the HAQ (HR 1.37).
Dr. Hekmat speculated that the rate of orthopedic surgery was reduced by early intensive treatment in patients with RA, since the time period studied coincided with the increased use of pharmacologic interventions. "In this cohort we are targeting the patients more aggressively," he said. "It’s not wrong to guess that this decline may lead to better [disease] management."
Dr. Hekmat said that he had no relevant financial conflicts to disclose.
SAN DIEGO – The overall proportion of rheumatoid arthritis patients undergoing orthopedic surgery is declining as intensive pharmacologic intervention with agents such as biologics play an increasingly prominent role in the treatment of the inflammatory disease, judging from results from a large, long-term study.
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat and his associates enrolled 2,342 men and women from RA patient registries in Sweden who filled out questionnaires including visual analog scales for general health and pain, and the Health Assessment Questionnaire (HAQ) in 1997, 2002, 2005, and 2009.
The response rate ranged from 62% to 74%, and the researchers linked responses to Swedish national health registry records to correlate the data with records of inpatient and outpatient surgeries, as well as the use of biologics. They went on to evaluate the incidence rate of orthopedic surgery in three time periods: from 1998-2001 (time period A), 2002-2006 (time period B), and 2007-2011 (time period C).
Dr. Hekmat, a rheumatologist and a PhD fellow at Malmö University, Sweden, reported that between 1998 and 2011 the incidence of all orthopedic procedures performed was 82.3 per 1,000 person-years. Significant declines were observed over the three time periods studied. For example, the incidence of any orthopedic surgery declined from 94.6 per 1,000 person-years in time period A to 82.6 per 1,000 person-years in time period B, and 71.8 per 1,000 person-years in time period C (P less than .001).
A similar association was observed for hip surgery alone (which fell from 27.8 per 1,000 person-years in time period A to 17.6 per 1,000 person-years in time period C; P less than .001). The incidence of orthopedic surgery on small joints such as hands, wrists, feet, and ankles fell from 43.8 per 1,000 person-years in time period A to 30.5 per 1,000 person-years in time period C (P less than .001).
No significant decline occurred in the incidence of knee surgeries performed during the study period. This actually rose slightly from 12.3 per 1,000 person-years in time period A to 12.9 per 1,000 person-years in time period C (P = .759).
Independent predictors of undergoing any orthopedic surgery included being female (hazard ratio 1.50) and having greater disability as measured by the HAQ (HR 1.37).
Dr. Hekmat speculated that the rate of orthopedic surgery was reduced by early intensive treatment in patients with RA, since the time period studied coincided with the increased use of pharmacologic interventions. "In this cohort we are targeting the patients more aggressively," he said. "It’s not wrong to guess that this decline may lead to better [disease] management."
Dr. Hekmat said that he had no relevant financial conflicts to disclose.
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat, Health Assessment Questionnaire, Malmö University, Sweden, orthopedic procedures,
In an ongoing population-based study presented at the annual meeting of the American College of Rheumatology, Dr. Korosh Hekmat, Health Assessment Questionnaire, Malmö University, Sweden, orthopedic procedures,
AT THE ACR ANNUAL MEETING
Major finding: The incidence of any orthopedic surgery among rheumatoid arthritis patients declined from 94.6 per 1,000 person-years in 1998-2001 to 82.6 per 1,000 person-years in 2002-2016 and 71.8 per 1,000 person-years in 2007-2011 (P less than .001).
Data source: A study of 2,342 men and women from RA patient registries in Sweden who filled out questionnaires including visual analog scales for general health and pain, and the Health Assessment Questionnaire in 1997, 2002, 2005, and 2009.
Disclosures: Dr. Hekmat said that he had no relevant financial conflicts to disclose.
Antirheumatic drugs don’t boost surgical infection risk
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
SAN DIEGO – Rheumatoid arthritis patients undergoing surgery who stayed on their antirheumatic medication perioperatively didn’t have a higher risk of early postoperative infection compared with those who temporarily stopped treatment before surgery, according to findings from a large national Veterans Affairs study.
Rheumatologists are frequently consulted about this issue. Evidence to guide practice has been scarce, however, and until now many rheumatologists and surgeons have taken a conservative approach, reasoning that the immunosuppressive drugs employed in controlling inflammation in rheumatoid arthritis might also increase the risk of surgical wound infection.
A common practice has been to have RA patients stop their medication a month ahead of elective surgery, or at least two drug half-lives beforehand, then start treatment again roughly a month after the operation, or when the wound has healed. The new Veterans Affairs (VA) study findings suggest this practice may be unnecessary, Dr. Zaki Abou Zahr said at the annual meeting of the American College of Rheumatology.
Dr. Bernard Ng, his senior coinvestigator in the study, added that temporarily stopping antirheumatic agents before surgery may actually be harmful in that it increases the risk of a flare of the RA, which in turn would impede postoperative rehabilitation.
But there is a major caveat regarding the VA study: Participation was restricted to RA patients on only a single conventional disease-modifying antirheumatic drug (DMARD) or biologic agent leading up to surgery. This restriction, imposed to make for a more clear-cut analysis, means that the study results can’t be extrapolated to patients on multidrug therapy. And multidrug therapy is quite common. Indeed, slightly more than half of RA patients in the VA health care system are on combination therapy, most often methotrexate plus a biologic agent, noted Dr. Ng, chief of rheumatology at the VA Puget Sound Health Care System, Seattle.
Dr. Abou Zahr presented the retrospective cohort study involving 6,548 RA patients in VA administrative databases, all of whom were on antirheumatic drug monotherapy prior to surgery. The surgery was of all types, including cardiothoracic, gastrointestinal, vascular, and orthopedic, as well as emergent and elective.
The primary endpoints were the rate of wound infections, both superficial and deep, within 30 days post surgery, and the general infection rate – including pneumonia, sepsis, and urinary tract infections – during the same time frame.
Sixty-two percent of the 1,480 RA patients on a single biologic agent did not stop taking it preoperatively. One key study finding was that neither their postoperative wound infection rate nor their general infection rate differed significantly from rates in patients who temporarily halted their biologic agent. The same held true among the 70% of patients on a single conventional DMARD who did not stop taking their medication preoperatively, according to Dr. Abou Zahr of Baylor College of Medicine, Houston.
Dr. Ng said the investigators plan to extend their work to include RA patients on multiple antirheumatic drugs that they do or don’t temporarily stop when undergoing surgery within the VA system. The researchers also plan to take a close look at patients undergoing specific types of surgery to see if the postoperative infection risk in patients who remain on treatment varies according to their operation.
Dr. Fehmida Zahabi, a rheumatologist from Plano, Tex., who chaired a press conference highlighting the VA study findings, said that while she’d like to see a confirmatory study, "I think we’re getting to the point where we’re saying we should cautiously keep these patients on their medications. That’s what the data suggest."
She noted that before the VA study, the very limited evidence available to guide practice in this area centered on a 12-year-old British randomized trial involving RA patients on methotrexate undergoing elective orthopedic surgery. Those assigned to stop the drug from 2 weeks before surgery to 2 weeks post surgery had significantly more infections, surgical complications, and RA flares within 6 weeks after surgery (Ann. Rheum. Dis. 2001;60:214-7).
As for patients on multidrug therapy who are scheduled for surgery, her inclination until evidence becomes available for guidance is to pare down the regimen preoperatively, while keeping the patient on one or two drugs.
The VA study was funded by the Department of Veterans Affairs. Dr. Abou Zahr and Dr. Ng reported having no conflicts of interest.
AT THE ACR ANNUAL MEETING
Major finding: Rheumatoid arthritis patients who remained on their antirheumatic medication while they underwent various types of surgery did not have a significantly different 30-day wound infection rate than those who stopped treatment temporarily prior to surgery.
Data source: This was a retrospective observational cohort study involving 6,548 rheumatoid arthritis patients undergoing various types of surgery.
Disclosures: The study was funded by the Department of Veterans Affairs. The presenters reported having no financial conflicts.
Increasing proportion of musculoskeletal infections involve MRSA
ORLANDO – The proportion of pediatric musculoskeletal infections involving methicillin-resistant Staphylococcus aureus vs. methicillin-sensitive S. aureus has increased dramatically over the past decade, according to a retrospective review of cases.
Between 2001 and 2010, 148 patients at the Children’s Hospital of Philadelphia presented with acute musculoskeletal S. aureus infection, and overall, 37 of those involved methicillin-resistant S. aureus (MRSA), Eric Sarkissian reported at the annual meeting of the American Academy of Pediatrics conference.
The proportion of musculoskeletal cases involving MRSA increased from 12% in 2001-2002 to about 35% in 2010, said Mr. Sarkissian, a 4th-year medical student at Drexel University, Philadelphia.
"Furthermore, we found that MRSA infections were associated with a significantly more complicated hospital stay," Mr. Sarkissian said.
MRSA and methicillin-sensitive S. aureus (MSSA) patients were undergoing magnetic resonance imaging at similar rates, but the average duration of hospitalization was longer for the MRSA patients (13 vs. 8 days), and more MRSA patients required multiple surgical procedures (38% vs. 15%), he said.
In addition, mean presenting C-reactive protein levels were higher in the MRSA vs. MSSA patients (14.7 mg/L vs. 9.8 mg/L), as were infection-related complications, including deep vein thrombosis, septic emboli, septic shock, recurrent infection, and/or avascular necrosis (22% vs. 6%).
MRSA patients also were more often admitted to the ICU for further treatment.
Patients included in this series – the largest known case series of children and adolescents presenting with culture-positive S. aureus osteomyelitis and/or septic arthritis – were consecutive patients with positive cultures of blood, bone, or joint aspirate. Those with postoperative and chronic infections were excluded in an effort to minimize bias caused by nosocomial infections, Mr. Sarkissian noted.
The findings support the ongoing concern that MRSA is evolving and becoming more virulent and invasive over time, compared with MSSA infections. In addition, the findings underscore the need for prompt recognition and aggressive treatment of MRSA musculoskeletal infections, as early recognition is paramount for avoiding sequelae and improving patient outcomes.
"A high index of suspicion is still necessary in the setting of musculoskeletal infections in pediatric patients. Multidisciplinary care provides the most successful opportunity for having improved patient outcomes, and this requires employing broad spectrum antibiotic coverage, using aggressive surgical management, and having an awareness of potential complications," Mr. Sarkissian said.
While clinical prediction models have been developed to help in detecting MRSA infections, outcomes using the models have varied geographically, suggesting that there are unknown regional differences among MRSA cases. It remains to be seen whether available models will be useful for identifying MRSA at presentation, he said.
Mr. Sarkissian reported having no disclosures.
ORLANDO – The proportion of pediatric musculoskeletal infections involving methicillin-resistant Staphylococcus aureus vs. methicillin-sensitive S. aureus has increased dramatically over the past decade, according to a retrospective review of cases.
Between 2001 and 2010, 148 patients at the Children’s Hospital of Philadelphia presented with acute musculoskeletal S. aureus infection, and overall, 37 of those involved methicillin-resistant S. aureus (MRSA), Eric Sarkissian reported at the annual meeting of the American Academy of Pediatrics conference.
The proportion of musculoskeletal cases involving MRSA increased from 12% in 2001-2002 to about 35% in 2010, said Mr. Sarkissian, a 4th-year medical student at Drexel University, Philadelphia.
"Furthermore, we found that MRSA infections were associated with a significantly more complicated hospital stay," Mr. Sarkissian said.
MRSA and methicillin-sensitive S. aureus (MSSA) patients were undergoing magnetic resonance imaging at similar rates, but the average duration of hospitalization was longer for the MRSA patients (13 vs. 8 days), and more MRSA patients required multiple surgical procedures (38% vs. 15%), he said.
In addition, mean presenting C-reactive protein levels were higher in the MRSA vs. MSSA patients (14.7 mg/L vs. 9.8 mg/L), as were infection-related complications, including deep vein thrombosis, septic emboli, septic shock, recurrent infection, and/or avascular necrosis (22% vs. 6%).
MRSA patients also were more often admitted to the ICU for further treatment.
Patients included in this series – the largest known case series of children and adolescents presenting with culture-positive S. aureus osteomyelitis and/or septic arthritis – were consecutive patients with positive cultures of blood, bone, or joint aspirate. Those with postoperative and chronic infections were excluded in an effort to minimize bias caused by nosocomial infections, Mr. Sarkissian noted.
The findings support the ongoing concern that MRSA is evolving and becoming more virulent and invasive over time, compared with MSSA infections. In addition, the findings underscore the need for prompt recognition and aggressive treatment of MRSA musculoskeletal infections, as early recognition is paramount for avoiding sequelae and improving patient outcomes.
"A high index of suspicion is still necessary in the setting of musculoskeletal infections in pediatric patients. Multidisciplinary care provides the most successful opportunity for having improved patient outcomes, and this requires employing broad spectrum antibiotic coverage, using aggressive surgical management, and having an awareness of potential complications," Mr. Sarkissian said.
While clinical prediction models have been developed to help in detecting MRSA infections, outcomes using the models have varied geographically, suggesting that there are unknown regional differences among MRSA cases. It remains to be seen whether available models will be useful for identifying MRSA at presentation, he said.
Mr. Sarkissian reported having no disclosures.
ORLANDO – The proportion of pediatric musculoskeletal infections involving methicillin-resistant Staphylococcus aureus vs. methicillin-sensitive S. aureus has increased dramatically over the past decade, according to a retrospective review of cases.
Between 2001 and 2010, 148 patients at the Children’s Hospital of Philadelphia presented with acute musculoskeletal S. aureus infection, and overall, 37 of those involved methicillin-resistant S. aureus (MRSA), Eric Sarkissian reported at the annual meeting of the American Academy of Pediatrics conference.
The proportion of musculoskeletal cases involving MRSA increased from 12% in 2001-2002 to about 35% in 2010, said Mr. Sarkissian, a 4th-year medical student at Drexel University, Philadelphia.
"Furthermore, we found that MRSA infections were associated with a significantly more complicated hospital stay," Mr. Sarkissian said.
MRSA and methicillin-sensitive S. aureus (MSSA) patients were undergoing magnetic resonance imaging at similar rates, but the average duration of hospitalization was longer for the MRSA patients (13 vs. 8 days), and more MRSA patients required multiple surgical procedures (38% vs. 15%), he said.
In addition, mean presenting C-reactive protein levels were higher in the MRSA vs. MSSA patients (14.7 mg/L vs. 9.8 mg/L), as were infection-related complications, including deep vein thrombosis, septic emboli, septic shock, recurrent infection, and/or avascular necrosis (22% vs. 6%).
MRSA patients also were more often admitted to the ICU for further treatment.
Patients included in this series – the largest known case series of children and adolescents presenting with culture-positive S. aureus osteomyelitis and/or septic arthritis – were consecutive patients with positive cultures of blood, bone, or joint aspirate. Those with postoperative and chronic infections were excluded in an effort to minimize bias caused by nosocomial infections, Mr. Sarkissian noted.
The findings support the ongoing concern that MRSA is evolving and becoming more virulent and invasive over time, compared with MSSA infections. In addition, the findings underscore the need for prompt recognition and aggressive treatment of MRSA musculoskeletal infections, as early recognition is paramount for avoiding sequelae and improving patient outcomes.
"A high index of suspicion is still necessary in the setting of musculoskeletal infections in pediatric patients. Multidisciplinary care provides the most successful opportunity for having improved patient outcomes, and this requires employing broad spectrum antibiotic coverage, using aggressive surgical management, and having an awareness of potential complications," Mr. Sarkissian said.
While clinical prediction models have been developed to help in detecting MRSA infections, outcomes using the models have varied geographically, suggesting that there are unknown regional differences among MRSA cases. It remains to be seen whether available models will be useful for identifying MRSA at presentation, he said.
Mr. Sarkissian reported having no disclosures.
AT THE AAP NATIONAL CONFERENCE
Major finding: The proportion of musculoskeletal infection cases involving MRSA vs. MSSA increased from 12% to 35% between 2001 and 2010.
Data source: A retrospective study of 148 consecutive cases.
Disclosures: Mr. Sarkissian reported having no disclosures.
New drugs, strategies advance rheumatoid arthritis treatment
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
New biologics and more aggressive intervention strategies have advanced rheumatoid arthritis treatment over the last decade, Dr. Iain McInnes explains, but remission remains too infrequent. New drugs under development in the coming years offer promise for the future. For more on rheumatoid arthritis advances, visit http://www.rheumatologynews.com.
Online self-help tool benefits rheumatoid arthritis patients
Using an online self-help tool for 10 weeks improved rheumatoid arthritis patients’ quality of life up to 9 months after the intervention, according to a small, randomized trial.
However, patients didn’t see improvements in health status or pain and other symptoms beyond those of patients who were randomized to a waiting list and didn’t receive the intervention, according to Cheryl L. Shigaki, Ph.D., of the department of health psychology at the University of Missouri, Columbia, and her colleagues (Arthritis Care Res. 2013;65:1573-81).
The only previous study of an online self-management intervention involved patients with rheumatoid arthritis, osteoarthritis, or fibromyalgia, and those with RA showed sustained improvement in self-reported global health and activity limitation at 12 months after beginning the program (Arthritis Rheum. 2008;59:1009-17).
The self-management tool included content delivered via a visual slideshow, as well as access to the online site RAHelp.org, which provided peer support through social networking applications. Patients in the intervention group also had weekly phone calls with a counselor trained in cognitive-behavioral group interventions.
The mean age of the 106 patients who underwent the intervention (54 patients) or went on a wait list (52 patients) was 49 and 50 years, respectively, and 98 of the 106 patients were women. A total of 86% had attended graduate school or at least some college, and 94% of the 106 patients were white.
Immediately after the 10-week intervention period, participants in the treatment group had significantly better scores on the Arthritis Self-Efficacy Scale than did those in the wait-list group (83.9 vs. 68.5, respectively), and that difference was maintained 9 months after the intervention (84.1 vs. 68.6). The effect size associated with the intervention at both time points was large, at 0.92.
The intervention had significantly greater positive effect on quality of life than did being on the waiting list, Dr. Shigaki and her colleagues noted, and had a moderate effect size on Quality of Life Scale scores that was retained from immediately after the intervention (88.4 vs. 84.9) through 9 months later (88.0 vs. 83.1).
The investigators acknowledged that it’s difficult to determine how much of the intervention’s impact was attributable to the online materials and peer support, and how much was because of the phone calls with counselors. However, given the public’s increasing comfort with online communications since the trial began in 2003, they said, "we expect that a future version of RAHelp could be conducted safely and effectively without scheduled phone contacts, and that most, if not all, interactions could be conducted online."
The investigators cautioned that the participants were not blinded to the intervention and may have benefited from an attention effect. The participants also were early adopters of Internet tools and services, and they had a high level of education and income. Thus, their results may not be generalizable to a broader population. But they may represent people "who accept and are comfortable with, or even perhaps prefer, receiving health care services online."
The program could be scaled up by "significantly reducing the amount of clinician phone contact time without introducing untoward risk or consumer rejection," the researchers added.
Reimbursement could be a barrier to expanding similar programs, however. "At this time, there is no clear and consistent way to recoup costs associated with online self-management programming," Dr. Shigaki and her colleagues cautioned. But that could change as Medicare shifts its thinking on reimbursement for behavioral and psychosocial approaches that reduce the overall costs of chronic illness.
The study was funded by a grant from the National Institute on Disability and Rehabilitation Research of the Department of Education. One of the authors reported receiving honoraria from the Association of Rheumatology Health Professionals.
Using an online self-help tool for 10 weeks improved rheumatoid arthritis patients’ quality of life up to 9 months after the intervention, according to a small, randomized trial.
However, patients didn’t see improvements in health status or pain and other symptoms beyond those of patients who were randomized to a waiting list and didn’t receive the intervention, according to Cheryl L. Shigaki, Ph.D., of the department of health psychology at the University of Missouri, Columbia, and her colleagues (Arthritis Care Res. 2013;65:1573-81).
The only previous study of an online self-management intervention involved patients with rheumatoid arthritis, osteoarthritis, or fibromyalgia, and those with RA showed sustained improvement in self-reported global health and activity limitation at 12 months after beginning the program (Arthritis Rheum. 2008;59:1009-17).
The self-management tool included content delivered via a visual slideshow, as well as access to the online site RAHelp.org, which provided peer support through social networking applications. Patients in the intervention group also had weekly phone calls with a counselor trained in cognitive-behavioral group interventions.
The mean age of the 106 patients who underwent the intervention (54 patients) or went on a wait list (52 patients) was 49 and 50 years, respectively, and 98 of the 106 patients were women. A total of 86% had attended graduate school or at least some college, and 94% of the 106 patients were white.
Immediately after the 10-week intervention period, participants in the treatment group had significantly better scores on the Arthritis Self-Efficacy Scale than did those in the wait-list group (83.9 vs. 68.5, respectively), and that difference was maintained 9 months after the intervention (84.1 vs. 68.6). The effect size associated with the intervention at both time points was large, at 0.92.
The intervention had significantly greater positive effect on quality of life than did being on the waiting list, Dr. Shigaki and her colleagues noted, and had a moderate effect size on Quality of Life Scale scores that was retained from immediately after the intervention (88.4 vs. 84.9) through 9 months later (88.0 vs. 83.1).
The investigators acknowledged that it’s difficult to determine how much of the intervention’s impact was attributable to the online materials and peer support, and how much was because of the phone calls with counselors. However, given the public’s increasing comfort with online communications since the trial began in 2003, they said, "we expect that a future version of RAHelp could be conducted safely and effectively without scheduled phone contacts, and that most, if not all, interactions could be conducted online."
The investigators cautioned that the participants were not blinded to the intervention and may have benefited from an attention effect. The participants also were early adopters of Internet tools and services, and they had a high level of education and income. Thus, their results may not be generalizable to a broader population. But they may represent people "who accept and are comfortable with, or even perhaps prefer, receiving health care services online."
The program could be scaled up by "significantly reducing the amount of clinician phone contact time without introducing untoward risk or consumer rejection," the researchers added.
Reimbursement could be a barrier to expanding similar programs, however. "At this time, there is no clear and consistent way to recoup costs associated with online self-management programming," Dr. Shigaki and her colleagues cautioned. But that could change as Medicare shifts its thinking on reimbursement for behavioral and psychosocial approaches that reduce the overall costs of chronic illness.
The study was funded by a grant from the National Institute on Disability and Rehabilitation Research of the Department of Education. One of the authors reported receiving honoraria from the Association of Rheumatology Health Professionals.
Using an online self-help tool for 10 weeks improved rheumatoid arthritis patients’ quality of life up to 9 months after the intervention, according to a small, randomized trial.
However, patients didn’t see improvements in health status or pain and other symptoms beyond those of patients who were randomized to a waiting list and didn’t receive the intervention, according to Cheryl L. Shigaki, Ph.D., of the department of health psychology at the University of Missouri, Columbia, and her colleagues (Arthritis Care Res. 2013;65:1573-81).
The only previous study of an online self-management intervention involved patients with rheumatoid arthritis, osteoarthritis, or fibromyalgia, and those with RA showed sustained improvement in self-reported global health and activity limitation at 12 months after beginning the program (Arthritis Rheum. 2008;59:1009-17).
The self-management tool included content delivered via a visual slideshow, as well as access to the online site RAHelp.org, which provided peer support through social networking applications. Patients in the intervention group also had weekly phone calls with a counselor trained in cognitive-behavioral group interventions.
The mean age of the 106 patients who underwent the intervention (54 patients) or went on a wait list (52 patients) was 49 and 50 years, respectively, and 98 of the 106 patients were women. A total of 86% had attended graduate school or at least some college, and 94% of the 106 patients were white.
Immediately after the 10-week intervention period, participants in the treatment group had significantly better scores on the Arthritis Self-Efficacy Scale than did those in the wait-list group (83.9 vs. 68.5, respectively), and that difference was maintained 9 months after the intervention (84.1 vs. 68.6). The effect size associated with the intervention at both time points was large, at 0.92.
The intervention had significantly greater positive effect on quality of life than did being on the waiting list, Dr. Shigaki and her colleagues noted, and had a moderate effect size on Quality of Life Scale scores that was retained from immediately after the intervention (88.4 vs. 84.9) through 9 months later (88.0 vs. 83.1).
The investigators acknowledged that it’s difficult to determine how much of the intervention’s impact was attributable to the online materials and peer support, and how much was because of the phone calls with counselors. However, given the public’s increasing comfort with online communications since the trial began in 2003, they said, "we expect that a future version of RAHelp could be conducted safely and effectively without scheduled phone contacts, and that most, if not all, interactions could be conducted online."
The investigators cautioned that the participants were not blinded to the intervention and may have benefited from an attention effect. The participants also were early adopters of Internet tools and services, and they had a high level of education and income. Thus, their results may not be generalizable to a broader population. But they may represent people "who accept and are comfortable with, or even perhaps prefer, receiving health care services online."
The program could be scaled up by "significantly reducing the amount of clinician phone contact time without introducing untoward risk or consumer rejection," the researchers added.
Reimbursement could be a barrier to expanding similar programs, however. "At this time, there is no clear and consistent way to recoup costs associated with online self-management programming," Dr. Shigaki and her colleagues cautioned. But that could change as Medicare shifts its thinking on reimbursement for behavioral and psychosocial approaches that reduce the overall costs of chronic illness.
The study was funded by a grant from the National Institute on Disability and Rehabilitation Research of the Department of Education. One of the authors reported receiving honoraria from the Association of Rheumatology Health Professionals.
FROM ARTHRITIS CARE & RESEARCH
Major finding: Significant differences favoring the intervention group over the wait-list group were maintained from immediately after intervention through the 9-month follow-up on the Arthritis Self-Efficacy Scale (84 vs. 69, respectively) and the Quality of Life Scale (88 vs. about 84, respectively).
Data source: A 10-week, randomized trial involving a nationwide convenience sample of 106 adult participants with rheumatoid arthritis.
Disclosures: The study was funded by a grant from the National Institute on Disability and Rehabilitation Research of the Department of Education. One of the authors reported receiving honoraria from the Association of Rheumatology Health Professionals.