User login
Clinical prediction rule identifies risk for rheumatoid arthritis
A cut point of 8 or higher on the Leiden clinical prediction rule is linked to a good predictive value in determining which patients with undifferentiated arthritis are at risk for developing rheumatoid arthritis, according to a meta-analysis of data sets examined with the tool.
A number of research studies have indicated that early aggressive treatment of patients with the first signs of rheumatoid arthritis (RA) can delay or diminish joint damage and functional disability from disease progression. An estimated one-third of patients with undifferentiated arthritis (UA) will progress to RA, while approximately half will remit their disease; the remainder tend to develop other conditions, such as osteoarthritis, psoriatic arthritis, and reactive arthritis. Thus, it is important to identify which patients will progress to RA so they can benefit from early treatment, as well as prevent unnecessary treatment in those who won’t develop RA, the investigators said (Semin. Arthritis Rheum. 2013 Oct. 17 [doi:10.1016/j.semarthrit.2013.08.005]).
Emma McNally of the Royal College of Surgeons in Ireland, Dublin, and her associates performed this meta-analysis, using the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and the QUADAS (Quality Assessment of Diagnostic Accuracy Studies). Their search of the MEDLINE, Cochrane Library, EMBASE, Cinahl, and Google Scholar databases identified more than 5,000 papers; they also performed a hand search of references found in the retrieved articles. They winnowed the papers down to four articles containing six data sets. A total of 1,084 patients were included in the analysis of the Leiden clinical prediction rule (CPR), which showed that as the total Leiden CPR score increased, the sensitivity decreased and the specificity increased.
"The pooled data indicate that a cut-point of greater than or equal to 8 is better for ruling in RA, with a higher specificity (0.95) than sensitivity (0.49). However, the cut points of greater than or equal to 9 and greater than or equal to 10 were associated with higher specificities (0.99 [for both])," Ms. McNally and her associates wrote, and the latter "may offer a more optimal marker for ruling in RA and determining initiation of treatment."
The duration of follow-up in the studies in the meta-analysis varied from a minimum of 6 months to 30 months; three data sets followed patients for 1 year, as did the study from which the Leiden CPR was derived. The percentage of UA patients who developed RA ranged from 31% to 76% across the six data sets. Four data sets used a modified version of the Leiden CPR, replacing the variable "severity of morning stiffness" with "duration of morning stiffness."
In two studies, some patients started receiving disease-modifying antirheumatic drugs (DMARDs), and in one study, some patients also received glucocorticoids.
In addition to the fact that some patients were treated during the studies, which would likely increase the discriminative ability of the CPR, other considerations to make when evaluating this meta-analysis include the fact that revised classification criteria have been developed for the diagnosis of RA since the Leiden CPR was developed. The American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 criteria were developed to "facilitate the identification of RA at an earlier stage (prior to the development of bone damage), compared with the 1987 ACR criteria," the investigators explained. A review of the two criteria showed that the new 2010 criteria had a higher sensitivity but lower specificity, compared with the 1987 criteria. That and other factors led the authors to conclude that use of the 1987 criteria in the studies in this meta-analysis was appropriate.
Nonetheless, Ms. McNally and her associates said that no impact-analysis studies of the Leiden CPR have been performed, and assessing the clinical effect of a CPR is key. Such studies, usually randomized, controlled trials, would explore the effect of the rule on "patient outcomes such as progression to RA, prescription of DMARDs, prevention of joint destruction, and disease remission."
"Further studies also need to be conducted to determine the predictive value of the Leiden CPR in patients with UA, if the new ACR/EULAR 2010 criteria for the diagnosis of RA are implemented in clinical practice," they wrote. The results of one study so far suggest that in this situation, the rule might be more useful as a marker of disease persistence of UA, unless the CPR was modified to accommodate the new criteria.
This research was funded by the Health Research Board in Ireland. The authors listed no relevant financial disclosures.
A cut point of 8 or higher on the Leiden clinical prediction rule is linked to a good predictive value in determining which patients with undifferentiated arthritis are at risk for developing rheumatoid arthritis, according to a meta-analysis of data sets examined with the tool.
A number of research studies have indicated that early aggressive treatment of patients with the first signs of rheumatoid arthritis (RA) can delay or diminish joint damage and functional disability from disease progression. An estimated one-third of patients with undifferentiated arthritis (UA) will progress to RA, while approximately half will remit their disease; the remainder tend to develop other conditions, such as osteoarthritis, psoriatic arthritis, and reactive arthritis. Thus, it is important to identify which patients will progress to RA so they can benefit from early treatment, as well as prevent unnecessary treatment in those who won’t develop RA, the investigators said (Semin. Arthritis Rheum. 2013 Oct. 17 [doi:10.1016/j.semarthrit.2013.08.005]).
Emma McNally of the Royal College of Surgeons in Ireland, Dublin, and her associates performed this meta-analysis, using the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and the QUADAS (Quality Assessment of Diagnostic Accuracy Studies). Their search of the MEDLINE, Cochrane Library, EMBASE, Cinahl, and Google Scholar databases identified more than 5,000 papers; they also performed a hand search of references found in the retrieved articles. They winnowed the papers down to four articles containing six data sets. A total of 1,084 patients were included in the analysis of the Leiden clinical prediction rule (CPR), which showed that as the total Leiden CPR score increased, the sensitivity decreased and the specificity increased.
"The pooled data indicate that a cut-point of greater than or equal to 8 is better for ruling in RA, with a higher specificity (0.95) than sensitivity (0.49). However, the cut points of greater than or equal to 9 and greater than or equal to 10 were associated with higher specificities (0.99 [for both])," Ms. McNally and her associates wrote, and the latter "may offer a more optimal marker for ruling in RA and determining initiation of treatment."
The duration of follow-up in the studies in the meta-analysis varied from a minimum of 6 months to 30 months; three data sets followed patients for 1 year, as did the study from which the Leiden CPR was derived. The percentage of UA patients who developed RA ranged from 31% to 76% across the six data sets. Four data sets used a modified version of the Leiden CPR, replacing the variable "severity of morning stiffness" with "duration of morning stiffness."
In two studies, some patients started receiving disease-modifying antirheumatic drugs (DMARDs), and in one study, some patients also received glucocorticoids.
In addition to the fact that some patients were treated during the studies, which would likely increase the discriminative ability of the CPR, other considerations to make when evaluating this meta-analysis include the fact that revised classification criteria have been developed for the diagnosis of RA since the Leiden CPR was developed. The American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 criteria were developed to "facilitate the identification of RA at an earlier stage (prior to the development of bone damage), compared with the 1987 ACR criteria," the investigators explained. A review of the two criteria showed that the new 2010 criteria had a higher sensitivity but lower specificity, compared with the 1987 criteria. That and other factors led the authors to conclude that use of the 1987 criteria in the studies in this meta-analysis was appropriate.
Nonetheless, Ms. McNally and her associates said that no impact-analysis studies of the Leiden CPR have been performed, and assessing the clinical effect of a CPR is key. Such studies, usually randomized, controlled trials, would explore the effect of the rule on "patient outcomes such as progression to RA, prescription of DMARDs, prevention of joint destruction, and disease remission."
"Further studies also need to be conducted to determine the predictive value of the Leiden CPR in patients with UA, if the new ACR/EULAR 2010 criteria for the diagnosis of RA are implemented in clinical practice," they wrote. The results of one study so far suggest that in this situation, the rule might be more useful as a marker of disease persistence of UA, unless the CPR was modified to accommodate the new criteria.
This research was funded by the Health Research Board in Ireland. The authors listed no relevant financial disclosures.
A cut point of 8 or higher on the Leiden clinical prediction rule is linked to a good predictive value in determining which patients with undifferentiated arthritis are at risk for developing rheumatoid arthritis, according to a meta-analysis of data sets examined with the tool.
A number of research studies have indicated that early aggressive treatment of patients with the first signs of rheumatoid arthritis (RA) can delay or diminish joint damage and functional disability from disease progression. An estimated one-third of patients with undifferentiated arthritis (UA) will progress to RA, while approximately half will remit their disease; the remainder tend to develop other conditions, such as osteoarthritis, psoriatic arthritis, and reactive arthritis. Thus, it is important to identify which patients will progress to RA so they can benefit from early treatment, as well as prevent unnecessary treatment in those who won’t develop RA, the investigators said (Semin. Arthritis Rheum. 2013 Oct. 17 [doi:10.1016/j.semarthrit.2013.08.005]).
Emma McNally of the Royal College of Surgeons in Ireland, Dublin, and her associates performed this meta-analysis, using the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and the QUADAS (Quality Assessment of Diagnostic Accuracy Studies). Their search of the MEDLINE, Cochrane Library, EMBASE, Cinahl, and Google Scholar databases identified more than 5,000 papers; they also performed a hand search of references found in the retrieved articles. They winnowed the papers down to four articles containing six data sets. A total of 1,084 patients were included in the analysis of the Leiden clinical prediction rule (CPR), which showed that as the total Leiden CPR score increased, the sensitivity decreased and the specificity increased.
"The pooled data indicate that a cut-point of greater than or equal to 8 is better for ruling in RA, with a higher specificity (0.95) than sensitivity (0.49). However, the cut points of greater than or equal to 9 and greater than or equal to 10 were associated with higher specificities (0.99 [for both])," Ms. McNally and her associates wrote, and the latter "may offer a more optimal marker for ruling in RA and determining initiation of treatment."
The duration of follow-up in the studies in the meta-analysis varied from a minimum of 6 months to 30 months; three data sets followed patients for 1 year, as did the study from which the Leiden CPR was derived. The percentage of UA patients who developed RA ranged from 31% to 76% across the six data sets. Four data sets used a modified version of the Leiden CPR, replacing the variable "severity of morning stiffness" with "duration of morning stiffness."
In two studies, some patients started receiving disease-modifying antirheumatic drugs (DMARDs), and in one study, some patients also received glucocorticoids.
In addition to the fact that some patients were treated during the studies, which would likely increase the discriminative ability of the CPR, other considerations to make when evaluating this meta-analysis include the fact that revised classification criteria have been developed for the diagnosis of RA since the Leiden CPR was developed. The American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 criteria were developed to "facilitate the identification of RA at an earlier stage (prior to the development of bone damage), compared with the 1987 ACR criteria," the investigators explained. A review of the two criteria showed that the new 2010 criteria had a higher sensitivity but lower specificity, compared with the 1987 criteria. That and other factors led the authors to conclude that use of the 1987 criteria in the studies in this meta-analysis was appropriate.
Nonetheless, Ms. McNally and her associates said that no impact-analysis studies of the Leiden CPR have been performed, and assessing the clinical effect of a CPR is key. Such studies, usually randomized, controlled trials, would explore the effect of the rule on "patient outcomes such as progression to RA, prescription of DMARDs, prevention of joint destruction, and disease remission."
"Further studies also need to be conducted to determine the predictive value of the Leiden CPR in patients with UA, if the new ACR/EULAR 2010 criteria for the diagnosis of RA are implemented in clinical practice," they wrote. The results of one study so far suggest that in this situation, the rule might be more useful as a marker of disease persistence of UA, unless the CPR was modified to accommodate the new criteria.
This research was funded by the Health Research Board in Ireland. The authors listed no relevant financial disclosures.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
Major finding: A cut-point of 8 or higher is good for ruling in RA, with a higher specificity (0.95) than sensitivity (0.49).
Data source: Meta-analysis of four articles with six data sets including a total of 1,084 patients in the analysis of the Leiden clinical prediction rule.
Disclosures: This research was funded by the Health Research Board in Ireland. The authors listed no relevant financial disclosures.
Adjuvant fish oil may help recent-onset RA
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: The primary outcome measure, failure of triple-DMARD therapy, occurred in only 10.5% of study subjects receiving fish oil supplements, compared with 32.1% of control subjects.
Data Source: A double-blind, randomized controlled trial involving 139 consecutive patients with recently diagnosed RA who received standard triple-DMARD treatment plus either fish oil (86 patients) or a placebo (53 patients) and were followed for 1 year.
Disclosures: This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Most data reassure regarding TNF inhibitors and cancer
LAS VEGAS – Concerns about use of a tumor necrosis factor inhibitor in a rheumatoid arthritis patient with a history of cancer are common, but the data are largely reassuring, according to Dr. Iain McInnes.
He used as an example a case involving a 56-year-old woman who had erosive RA that was anti-citrullinated peptide antibody–positive (ACPA+). The woman failed triple therapy, responded well to etanercept, and achieved clinical remission after 9 months. The patient then developed breast cancer and her TNF inhibitor was stopped, as recommended by current American College of Rheumatology guidelines.
The patient returned a year later after undergoing "the whole gamut from the oncological armamentarium," including chemotherapy, radiation therapy, and surgery.
"So what do you do? She wants her TNF blocker back – she did very well on it," said Dr. McInnes, who is Muirhead Chair of Medicine and Director of Institute of Infection, Immunity, and Inflammation at the University of Glasgow, Scotland.
Prescribe a conventional disease-modifying antirheumatic drug (DMARD)? Try a different mode of action?
What if the patient is a 56-year-old female with ACPA+ erosive RA who failed triple therapy and leflunomide; is doing poorly; has a history of breast cancer treated successfully 3 years ago by lumpectomy, radiation therapy and chemotherapy; and asks for a change in therapy?
According to the ACR guidelines, any biologic agent can be used in a patient with solid malignancy or nonmelanoma skin cancer that was treated more than 5 years ago, while rituximab should be considered in those treated for a solid malignancy within the last 5 years, those treated with nonmelanoma skin cancer within the last 5 years, those treated for skin melanoma, and those treated for lymphoproliferative malignancy.
The British Society of Rheumatology guidelines are "broadly similar but less restrictive on the use of rituximab, and less restrictive of the time duration from solid malignancy to decision making," Dr. McInnes said at Perspectives in Rheumatic Diseases 2013.
A number of studies looking at whether TNF inhibitors are associated with increased cancer risk have been published and, basically, "the more one looks at the data, the less concerned one becomes," he said.
In a large meta-analysis that included 74 randomized controlled studies, more than 15,400 patients treated with TNF inhibitors, and nearly 7,500 controls, investigators found no evidence to support or refute a link between TNF inhibitors treatment and short-term risk of all cancers except nonmelanoma skin cancer. The risk of nonmelanoma skin cancer, however, was nearly more than doubled in the treatment group (Pharmacoedpidemiol. Drug Saf. 2011;20:119-30).
A 2012 study demonstrated a very low risk of malignancy among patients on TNF inhibitors across several registries (Rheum. Dis. Clin. North Am. 2012;38:761-70).
Also, a study published earlier this year showed that, in nearly 40,000 patients with various types of rheumatic diseases, treatment with anti-TNF drugs was not associated with a short-term increase in the risk of cancer, compared with commonly used therapies for immune-mediated chronic inflammatory diseases (Arthritis Rheum. 2013;65:48-58).
Another large population-based cohort study published this year (BMJ 2013;346:f1939), showed that RA patients treated with TNF inhibitors are not at increased overall risk for cancer, but they do have a 50% increased relative risk of invasive melanoma. The increase in absolute melanoma risk is small, however, and "may not markedly shift the overall risk-benefit balance of TNF inhibitors as used in clinical practice but might do so in patients at high risk of melanoma for other reasons."
Notably, another large study published in 2011 linked data from multiple Swedish clinical registries of RA with nationwide data on hospitalization and outpatient visits for RA, showing that cancers that occur in patients taking TNF inhibitors are not characterized by any distinction, such as a later stage at presentation or worse survival.
"This is true whether you look at overall cancer or lung, colorectal, or hematologic cancers. You can reassure your patient," Dr. McInnes said.
As for RA patients with a history of prior malignancy, findings from a 2010 analysis of data from the British Society for Rheumatology Biologics Register showed that, in 117 patients on a DMARD and 177 on an anti-TNF drug, the risk of cancer was actually greater among those on a DMARD, with a rate of incident malignancy per 1,000 person-years of 25.3 in the anti-TNF patients, compared with 38.3 in the DMARD patients (Arthritis Care Res. 2010;62:755-63).
"When you look at the numbers they are, by and large, reassuring, and this is the message you should give to patients, but make decisions on an individual-patient basis," Dr. McInnes said.
Dr. McInnes disclosed that he has been a speaker, adviser, and/or investigator for Janssen, Roche, Pfizer, BMS, and Novartis. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Concerns about use of a tumor necrosis factor inhibitor in a rheumatoid arthritis patient with a history of cancer are common, but the data are largely reassuring, according to Dr. Iain McInnes.
He used as an example a case involving a 56-year-old woman who had erosive RA that was anti-citrullinated peptide antibody–positive (ACPA+). The woman failed triple therapy, responded well to etanercept, and achieved clinical remission after 9 months. The patient then developed breast cancer and her TNF inhibitor was stopped, as recommended by current American College of Rheumatology guidelines.
The patient returned a year later after undergoing "the whole gamut from the oncological armamentarium," including chemotherapy, radiation therapy, and surgery.
"So what do you do? She wants her TNF blocker back – she did very well on it," said Dr. McInnes, who is Muirhead Chair of Medicine and Director of Institute of Infection, Immunity, and Inflammation at the University of Glasgow, Scotland.
Prescribe a conventional disease-modifying antirheumatic drug (DMARD)? Try a different mode of action?
What if the patient is a 56-year-old female with ACPA+ erosive RA who failed triple therapy and leflunomide; is doing poorly; has a history of breast cancer treated successfully 3 years ago by lumpectomy, radiation therapy and chemotherapy; and asks for a change in therapy?
According to the ACR guidelines, any biologic agent can be used in a patient with solid malignancy or nonmelanoma skin cancer that was treated more than 5 years ago, while rituximab should be considered in those treated for a solid malignancy within the last 5 years, those treated with nonmelanoma skin cancer within the last 5 years, those treated for skin melanoma, and those treated for lymphoproliferative malignancy.
The British Society of Rheumatology guidelines are "broadly similar but less restrictive on the use of rituximab, and less restrictive of the time duration from solid malignancy to decision making," Dr. McInnes said at Perspectives in Rheumatic Diseases 2013.
A number of studies looking at whether TNF inhibitors are associated with increased cancer risk have been published and, basically, "the more one looks at the data, the less concerned one becomes," he said.
In a large meta-analysis that included 74 randomized controlled studies, more than 15,400 patients treated with TNF inhibitors, and nearly 7,500 controls, investigators found no evidence to support or refute a link between TNF inhibitors treatment and short-term risk of all cancers except nonmelanoma skin cancer. The risk of nonmelanoma skin cancer, however, was nearly more than doubled in the treatment group (Pharmacoedpidemiol. Drug Saf. 2011;20:119-30).
A 2012 study demonstrated a very low risk of malignancy among patients on TNF inhibitors across several registries (Rheum. Dis. Clin. North Am. 2012;38:761-70).
Also, a study published earlier this year showed that, in nearly 40,000 patients with various types of rheumatic diseases, treatment with anti-TNF drugs was not associated with a short-term increase in the risk of cancer, compared with commonly used therapies for immune-mediated chronic inflammatory diseases (Arthritis Rheum. 2013;65:48-58).
Another large population-based cohort study published this year (BMJ 2013;346:f1939), showed that RA patients treated with TNF inhibitors are not at increased overall risk for cancer, but they do have a 50% increased relative risk of invasive melanoma. The increase in absolute melanoma risk is small, however, and "may not markedly shift the overall risk-benefit balance of TNF inhibitors as used in clinical practice but might do so in patients at high risk of melanoma for other reasons."
Notably, another large study published in 2011 linked data from multiple Swedish clinical registries of RA with nationwide data on hospitalization and outpatient visits for RA, showing that cancers that occur in patients taking TNF inhibitors are not characterized by any distinction, such as a later stage at presentation or worse survival.
"This is true whether you look at overall cancer or lung, colorectal, or hematologic cancers. You can reassure your patient," Dr. McInnes said.
As for RA patients with a history of prior malignancy, findings from a 2010 analysis of data from the British Society for Rheumatology Biologics Register showed that, in 117 patients on a DMARD and 177 on an anti-TNF drug, the risk of cancer was actually greater among those on a DMARD, with a rate of incident malignancy per 1,000 person-years of 25.3 in the anti-TNF patients, compared with 38.3 in the DMARD patients (Arthritis Care Res. 2010;62:755-63).
"When you look at the numbers they are, by and large, reassuring, and this is the message you should give to patients, but make decisions on an individual-patient basis," Dr. McInnes said.
Dr. McInnes disclosed that he has been a speaker, adviser, and/or investigator for Janssen, Roche, Pfizer, BMS, and Novartis. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Concerns about use of a tumor necrosis factor inhibitor in a rheumatoid arthritis patient with a history of cancer are common, but the data are largely reassuring, according to Dr. Iain McInnes.
He used as an example a case involving a 56-year-old woman who had erosive RA that was anti-citrullinated peptide antibody–positive (ACPA+). The woman failed triple therapy, responded well to etanercept, and achieved clinical remission after 9 months. The patient then developed breast cancer and her TNF inhibitor was stopped, as recommended by current American College of Rheumatology guidelines.
The patient returned a year later after undergoing "the whole gamut from the oncological armamentarium," including chemotherapy, radiation therapy, and surgery.
"So what do you do? She wants her TNF blocker back – she did very well on it," said Dr. McInnes, who is Muirhead Chair of Medicine and Director of Institute of Infection, Immunity, and Inflammation at the University of Glasgow, Scotland.
Prescribe a conventional disease-modifying antirheumatic drug (DMARD)? Try a different mode of action?
What if the patient is a 56-year-old female with ACPA+ erosive RA who failed triple therapy and leflunomide; is doing poorly; has a history of breast cancer treated successfully 3 years ago by lumpectomy, radiation therapy and chemotherapy; and asks for a change in therapy?
According to the ACR guidelines, any biologic agent can be used in a patient with solid malignancy or nonmelanoma skin cancer that was treated more than 5 years ago, while rituximab should be considered in those treated for a solid malignancy within the last 5 years, those treated with nonmelanoma skin cancer within the last 5 years, those treated for skin melanoma, and those treated for lymphoproliferative malignancy.
The British Society of Rheumatology guidelines are "broadly similar but less restrictive on the use of rituximab, and less restrictive of the time duration from solid malignancy to decision making," Dr. McInnes said at Perspectives in Rheumatic Diseases 2013.
A number of studies looking at whether TNF inhibitors are associated with increased cancer risk have been published and, basically, "the more one looks at the data, the less concerned one becomes," he said.
In a large meta-analysis that included 74 randomized controlled studies, more than 15,400 patients treated with TNF inhibitors, and nearly 7,500 controls, investigators found no evidence to support or refute a link between TNF inhibitors treatment and short-term risk of all cancers except nonmelanoma skin cancer. The risk of nonmelanoma skin cancer, however, was nearly more than doubled in the treatment group (Pharmacoedpidemiol. Drug Saf. 2011;20:119-30).
A 2012 study demonstrated a very low risk of malignancy among patients on TNF inhibitors across several registries (Rheum. Dis. Clin. North Am. 2012;38:761-70).
Also, a study published earlier this year showed that, in nearly 40,000 patients with various types of rheumatic diseases, treatment with anti-TNF drugs was not associated with a short-term increase in the risk of cancer, compared with commonly used therapies for immune-mediated chronic inflammatory diseases (Arthritis Rheum. 2013;65:48-58).
Another large population-based cohort study published this year (BMJ 2013;346:f1939), showed that RA patients treated with TNF inhibitors are not at increased overall risk for cancer, but they do have a 50% increased relative risk of invasive melanoma. The increase in absolute melanoma risk is small, however, and "may not markedly shift the overall risk-benefit balance of TNF inhibitors as used in clinical practice but might do so in patients at high risk of melanoma for other reasons."
Notably, another large study published in 2011 linked data from multiple Swedish clinical registries of RA with nationwide data on hospitalization and outpatient visits for RA, showing that cancers that occur in patients taking TNF inhibitors are not characterized by any distinction, such as a later stage at presentation or worse survival.
"This is true whether you look at overall cancer or lung, colorectal, or hematologic cancers. You can reassure your patient," Dr. McInnes said.
As for RA patients with a history of prior malignancy, findings from a 2010 analysis of data from the British Society for Rheumatology Biologics Register showed that, in 117 patients on a DMARD and 177 on an anti-TNF drug, the risk of cancer was actually greater among those on a DMARD, with a rate of incident malignancy per 1,000 person-years of 25.3 in the anti-TNF patients, compared with 38.3 in the DMARD patients (Arthritis Care Res. 2010;62:755-63).
"When you look at the numbers they are, by and large, reassuring, and this is the message you should give to patients, but make decisions on an individual-patient basis," Dr. McInnes said.
Dr. McInnes disclosed that he has been a speaker, adviser, and/or investigator for Janssen, Roche, Pfizer, BMS, and Novartis. The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
Data provide guidance on herpes zoster vaccination in rheumatoid arthritis
LAS VEGAS – Should live attenuated herpes zoster vaccine be administered to a 45-year-old patient with mild rheumatoid arthritis and a strong family history of autoimmune disease? Should it be administered to a patient with long-standing RA who is seropositive and taking methotrexate and prednisone?
Many physicians are justifiably concerned about whether vaccination could cause autoimmune disease, Dr. Daniel E. Furst said at the Perspectives in Rheumatic Diseases 2013 meeting.
"And there is a lot of rationale for this to happen," he said, explaining that vaccination theoretically has the potential to activate the innate immune system or cause an allergic reaction that may accelerate the immune response.
The available data "aren’t fantastic" and they tend to conflict, but they do provide some guidance, said Dr. Furst, the Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles.
A few cases of Guillain-Barré syndrome have occurred after administration of the H1N1 flu vaccine (about 1 in 100,000), and idiopathic thrombocytopenic purpura has occurred following measles, mumps, and rubella (MMR) vaccine (about 1 in 30,000), he said.
Reports regarding multiple sclerosis following hepatitis B vaccination have been conflicting.
For the first case – a 45-year-old man with a strong family history of autoimmune disease who had mild RA, with occasional wrist and knee pain but no swelling – Dr. Furst said he would tell the patient there is "very little going on there" in terms of concern about activating autoimmune disease, and he would likely provide the vaccine.
In the second case, however, it is important to consider the known increased risk of developing herpes zoster among patients with rheumatic diseases (about a twofold increased risk, largely due to prednisone use), and the risks associated with providing the vaccine – including the added risk that would come with adding a biologic to the treatment regimen.
The risk is further increased by the use of both anti–tumor necrosis factor (anti-TNF) and non–anti-TNF biologics, he said.
"It appears that anything you do to suppress the immune system increases the risk," he added.
The question is whether the benefits of vaccinating outweigh the risks. Herpes zoster vaccination is associated with about a 40% reduction in the risk of shingles in this patient population. In one retrospective study of more than 460,000 Medicare patients, about 18,000 developed herpes zoster. However, in 663 patients who were on anti-TNF therapy and who were vaccinated, none developed a disseminated case in the 42 days following vaccination.
"What this seems to suggest is that yes, you can [vaccinate]," he said. But because that evidence is from a retrospective study, he advised that if you choose to vaccinate these patients, "you better have your thoughts well organized and tell the patient there is some increased risk."
In his practice, he continues to vaccinate only before initiating biologic therapy or after a washout period.
"The evidence isn’t strong enough to change my practice," he said.
According to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, vaccination is okay in rheumatic disease patients who are on low-dose prednisone (less than 20 mg/day) and in those on low doses of methotrexate (less than 0.4 mg/kg per week), azathioprine (less than 3.0 mg/kg per day), or 6-mercaptopurine (1.5 mg/kg per day or less), but should be avoided in transplant patients, those who have active lymphomas or HIV, and those using high-dose steroids.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furst reported serving as a consultant or speaker for, and/or receiving research or grant support from, AbbVie, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Janssen, Genentech/Roche, Gilead, GlaxoSmithKline, Novartis, Pfizer, and UCB. He has received grant support from the National Institutes of Health.
LAS VEGAS – Should live attenuated herpes zoster vaccine be administered to a 45-year-old patient with mild rheumatoid arthritis and a strong family history of autoimmune disease? Should it be administered to a patient with long-standing RA who is seropositive and taking methotrexate and prednisone?
Many physicians are justifiably concerned about whether vaccination could cause autoimmune disease, Dr. Daniel E. Furst said at the Perspectives in Rheumatic Diseases 2013 meeting.
"And there is a lot of rationale for this to happen," he said, explaining that vaccination theoretically has the potential to activate the innate immune system or cause an allergic reaction that may accelerate the immune response.
The available data "aren’t fantastic" and they tend to conflict, but they do provide some guidance, said Dr. Furst, the Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles.
A few cases of Guillain-Barré syndrome have occurred after administration of the H1N1 flu vaccine (about 1 in 100,000), and idiopathic thrombocytopenic purpura has occurred following measles, mumps, and rubella (MMR) vaccine (about 1 in 30,000), he said.
Reports regarding multiple sclerosis following hepatitis B vaccination have been conflicting.
For the first case – a 45-year-old man with a strong family history of autoimmune disease who had mild RA, with occasional wrist and knee pain but no swelling – Dr. Furst said he would tell the patient there is "very little going on there" in terms of concern about activating autoimmune disease, and he would likely provide the vaccine.
In the second case, however, it is important to consider the known increased risk of developing herpes zoster among patients with rheumatic diseases (about a twofold increased risk, largely due to prednisone use), and the risks associated with providing the vaccine – including the added risk that would come with adding a biologic to the treatment regimen.
The risk is further increased by the use of both anti–tumor necrosis factor (anti-TNF) and non–anti-TNF biologics, he said.
"It appears that anything you do to suppress the immune system increases the risk," he added.
The question is whether the benefits of vaccinating outweigh the risks. Herpes zoster vaccination is associated with about a 40% reduction in the risk of shingles in this patient population. In one retrospective study of more than 460,000 Medicare patients, about 18,000 developed herpes zoster. However, in 663 patients who were on anti-TNF therapy and who were vaccinated, none developed a disseminated case in the 42 days following vaccination.
"What this seems to suggest is that yes, you can [vaccinate]," he said. But because that evidence is from a retrospective study, he advised that if you choose to vaccinate these patients, "you better have your thoughts well organized and tell the patient there is some increased risk."
In his practice, he continues to vaccinate only before initiating biologic therapy or after a washout period.
"The evidence isn’t strong enough to change my practice," he said.
According to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, vaccination is okay in rheumatic disease patients who are on low-dose prednisone (less than 20 mg/day) and in those on low doses of methotrexate (less than 0.4 mg/kg per week), azathioprine (less than 3.0 mg/kg per day), or 6-mercaptopurine (1.5 mg/kg per day or less), but should be avoided in transplant patients, those who have active lymphomas or HIV, and those using high-dose steroids.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furst reported serving as a consultant or speaker for, and/or receiving research or grant support from, AbbVie, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Janssen, Genentech/Roche, Gilead, GlaxoSmithKline, Novartis, Pfizer, and UCB. He has received grant support from the National Institutes of Health.
LAS VEGAS – Should live attenuated herpes zoster vaccine be administered to a 45-year-old patient with mild rheumatoid arthritis and a strong family history of autoimmune disease? Should it be administered to a patient with long-standing RA who is seropositive and taking methotrexate and prednisone?
Many physicians are justifiably concerned about whether vaccination could cause autoimmune disease, Dr. Daniel E. Furst said at the Perspectives in Rheumatic Diseases 2013 meeting.
"And there is a lot of rationale for this to happen," he said, explaining that vaccination theoretically has the potential to activate the innate immune system or cause an allergic reaction that may accelerate the immune response.
The available data "aren’t fantastic" and they tend to conflict, but they do provide some guidance, said Dr. Furst, the Carl M. Pearson Professor of Rheumatology at the University of California, Los Angeles.
A few cases of Guillain-Barré syndrome have occurred after administration of the H1N1 flu vaccine (about 1 in 100,000), and idiopathic thrombocytopenic purpura has occurred following measles, mumps, and rubella (MMR) vaccine (about 1 in 30,000), he said.
Reports regarding multiple sclerosis following hepatitis B vaccination have been conflicting.
For the first case – a 45-year-old man with a strong family history of autoimmune disease who had mild RA, with occasional wrist and knee pain but no swelling – Dr. Furst said he would tell the patient there is "very little going on there" in terms of concern about activating autoimmune disease, and he would likely provide the vaccine.
In the second case, however, it is important to consider the known increased risk of developing herpes zoster among patients with rheumatic diseases (about a twofold increased risk, largely due to prednisone use), and the risks associated with providing the vaccine – including the added risk that would come with adding a biologic to the treatment regimen.
The risk is further increased by the use of both anti–tumor necrosis factor (anti-TNF) and non–anti-TNF biologics, he said.
"It appears that anything you do to suppress the immune system increases the risk," he added.
The question is whether the benefits of vaccinating outweigh the risks. Herpes zoster vaccination is associated with about a 40% reduction in the risk of shingles in this patient population. In one retrospective study of more than 460,000 Medicare patients, about 18,000 developed herpes zoster. However, in 663 patients who were on anti-TNF therapy and who were vaccinated, none developed a disseminated case in the 42 days following vaccination.
"What this seems to suggest is that yes, you can [vaccinate]," he said. But because that evidence is from a retrospective study, he advised that if you choose to vaccinate these patients, "you better have your thoughts well organized and tell the patient there is some increased risk."
In his practice, he continues to vaccinate only before initiating biologic therapy or after a washout period.
"The evidence isn’t strong enough to change my practice," he said.
According to recommendations from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, vaccination is okay in rheumatic disease patients who are on low-dose prednisone (less than 20 mg/day) and in those on low doses of methotrexate (less than 0.4 mg/kg per week), azathioprine (less than 3.0 mg/kg per day), or 6-mercaptopurine (1.5 mg/kg per day or less), but should be avoided in transplant patients, those who have active lymphomas or HIV, and those using high-dose steroids.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furst reported serving as a consultant or speaker for, and/or receiving research or grant support from, AbbVie, Actelion, Amgen, Bristol-Myers Squibb, Celgene, Janssen, Genentech/Roche, Gilead, GlaxoSmithKline, Novartis, Pfizer, and UCB. He has received grant support from the National Institutes of Health.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
Palmar-plantar psoriasis? Anti-TNF therapy may be culprit
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
RA disease activity assessed differently by patients, physicians, and ultrasound
Rheumatoid arthritis patients were less likely than their physicians to score themselves as being in clinical remission on a widely used clinical composite index, based on a prospective, cross-sectional study.
Meanwhile, ultrasound assessments also showed variable correlation with both patient and physician assessments of tender and swollen joints in the study of 69 patients with RA who were judged to be in clinical remission by their rheumatologist, according to lead investigator Dr. Iustina Janta of the Hospital General Universitario Gregorio Marañón, Madrid, and her colleagues.
"Accurate assessment of RA activity in a nonactive state is of the utmost importance because therapeutic decisions should target sustained remission or, at a minimum, the lowest possible disease activity to improve RA outcomes," Dr. Janta and her coauthors wrote (Rheumatology 2013 Sept. 17 [doi:10.1093/rheumatology/ket297]).
The mean age of the cohort was 60 years, and most of the patients were women (77%). The patients’ mean disease duration was nearly 12 years, and overall, 70% were in treatment with synthetic disease-modifying antirheumatic drugs. Another 16% took biologics, and the remainder used both classes of drugs.
To assess concordance between patient and physician assessments of disease activity, patients first indicated to the investigators which joints on a mannequin corresponded to their own tender or swollen joints. They also ranked their global health assessment on a visual analogue scale from 0 to 10.
A physician blinded to each patient’s own assessment then recorded an assessment of each patient’s tender joint count and swollen joint count, as well as the physician’s perception of the patient’s global assessment of disease activity.
Dr. Janta and her associates found that the mean scores calculated on the Disease Activity Score 28 (DAS28) were significantly higher according to patients, at 3.34, compared with the physician tally, at 2.55 (P less than .0005).
Indeed, even though the attending rheumatologist had judged all 69 patients to be in clinical remission, DAS28 scores calculated from the patients’ assessments indicated that only 18 patients (26%) fit this bill. But according to the physician assessment, that number was 36 patients (52%) – another significant difference (P less than .0005).
Next, the researchers assessed concordance between doctors and patients on the simplified disease activity index (SDAI). Here, there was better agreement: The mean score assessment among patients was 12.33, compared with a physician mean assessment of 10.55 (P = .246). That translated to a statistically similar SDAI-based remission rate of 15% according to patients vs. 12% according to physicians (P = .172).
Finally, ultrasound examinations with B mode and power Doppler showed no significant correlations with patient- and physician-derived clinical assessments. The mean number of tender joints was 4.34 based on patient assessment, 3.12 by physician examination, and 4.09 based on B-mode ultrasound.
On the other hand, swollen joint counts (SJCs) were significantly lower in both patient (2.00) and physician (1.42) estimation, compared with B-mode ultrasound (4.09) (P less than .0005 for ultrasound vs. patient mean SJC; P = .033 for ultrasound vs. physician SJC).
The ultrasound results on power Doppler were reversed in comparison with B-mode assessment. Tender joint counts were much higher when estimated by patients (4.34) and physicians (3.12), compared with Doppler ultrasound (1.1, P less than .0005 for both).
But mean swollen joint counts on power Doppler ultrasound (1.1) correlated well with patient- and physician-assessed counts (2 and 1.42, respectively).
Dr. Janta and her associates wrote that in the future, given the current trend in patient involvement in disease activity assessments throughout the field, "longitudinal studies should investigate the predictive capability of patient self-assessment of RA activity in relation to relevant disease outcomes, which will address the real value of the discrepancies between patient-assessed and physician-assessed RA activity."
They also noted that "studies on the optimal weight of power Doppler findings in global RA activity assessment are needed."
The authors stated that they had no relevant financial conflicts. They did not report any outside funding for this study.
Rheumatoid arthritis patients were less likely than their physicians to score themselves as being in clinical remission on a widely used clinical composite index, based on a prospective, cross-sectional study.
Meanwhile, ultrasound assessments also showed variable correlation with both patient and physician assessments of tender and swollen joints in the study of 69 patients with RA who were judged to be in clinical remission by their rheumatologist, according to lead investigator Dr. Iustina Janta of the Hospital General Universitario Gregorio Marañón, Madrid, and her colleagues.
"Accurate assessment of RA activity in a nonactive state is of the utmost importance because therapeutic decisions should target sustained remission or, at a minimum, the lowest possible disease activity to improve RA outcomes," Dr. Janta and her coauthors wrote (Rheumatology 2013 Sept. 17 [doi:10.1093/rheumatology/ket297]).
The mean age of the cohort was 60 years, and most of the patients were women (77%). The patients’ mean disease duration was nearly 12 years, and overall, 70% were in treatment with synthetic disease-modifying antirheumatic drugs. Another 16% took biologics, and the remainder used both classes of drugs.
To assess concordance between patient and physician assessments of disease activity, patients first indicated to the investigators which joints on a mannequin corresponded to their own tender or swollen joints. They also ranked their global health assessment on a visual analogue scale from 0 to 10.
A physician blinded to each patient’s own assessment then recorded an assessment of each patient’s tender joint count and swollen joint count, as well as the physician’s perception of the patient’s global assessment of disease activity.
Dr. Janta and her associates found that the mean scores calculated on the Disease Activity Score 28 (DAS28) were significantly higher according to patients, at 3.34, compared with the physician tally, at 2.55 (P less than .0005).
Indeed, even though the attending rheumatologist had judged all 69 patients to be in clinical remission, DAS28 scores calculated from the patients’ assessments indicated that only 18 patients (26%) fit this bill. But according to the physician assessment, that number was 36 patients (52%) – another significant difference (P less than .0005).
Next, the researchers assessed concordance between doctors and patients on the simplified disease activity index (SDAI). Here, there was better agreement: The mean score assessment among patients was 12.33, compared with a physician mean assessment of 10.55 (P = .246). That translated to a statistically similar SDAI-based remission rate of 15% according to patients vs. 12% according to physicians (P = .172).
Finally, ultrasound examinations with B mode and power Doppler showed no significant correlations with patient- and physician-derived clinical assessments. The mean number of tender joints was 4.34 based on patient assessment, 3.12 by physician examination, and 4.09 based on B-mode ultrasound.
On the other hand, swollen joint counts (SJCs) were significantly lower in both patient (2.00) and physician (1.42) estimation, compared with B-mode ultrasound (4.09) (P less than .0005 for ultrasound vs. patient mean SJC; P = .033 for ultrasound vs. physician SJC).
The ultrasound results on power Doppler were reversed in comparison with B-mode assessment. Tender joint counts were much higher when estimated by patients (4.34) and physicians (3.12), compared with Doppler ultrasound (1.1, P less than .0005 for both).
But mean swollen joint counts on power Doppler ultrasound (1.1) correlated well with patient- and physician-assessed counts (2 and 1.42, respectively).
Dr. Janta and her associates wrote that in the future, given the current trend in patient involvement in disease activity assessments throughout the field, "longitudinal studies should investigate the predictive capability of patient self-assessment of RA activity in relation to relevant disease outcomes, which will address the real value of the discrepancies between patient-assessed and physician-assessed RA activity."
They also noted that "studies on the optimal weight of power Doppler findings in global RA activity assessment are needed."
The authors stated that they had no relevant financial conflicts. They did not report any outside funding for this study.
Rheumatoid arthritis patients were less likely than their physicians to score themselves as being in clinical remission on a widely used clinical composite index, based on a prospective, cross-sectional study.
Meanwhile, ultrasound assessments also showed variable correlation with both patient and physician assessments of tender and swollen joints in the study of 69 patients with RA who were judged to be in clinical remission by their rheumatologist, according to lead investigator Dr. Iustina Janta of the Hospital General Universitario Gregorio Marañón, Madrid, and her colleagues.
"Accurate assessment of RA activity in a nonactive state is of the utmost importance because therapeutic decisions should target sustained remission or, at a minimum, the lowest possible disease activity to improve RA outcomes," Dr. Janta and her coauthors wrote (Rheumatology 2013 Sept. 17 [doi:10.1093/rheumatology/ket297]).
The mean age of the cohort was 60 years, and most of the patients were women (77%). The patients’ mean disease duration was nearly 12 years, and overall, 70% were in treatment with synthetic disease-modifying antirheumatic drugs. Another 16% took biologics, and the remainder used both classes of drugs.
To assess concordance between patient and physician assessments of disease activity, patients first indicated to the investigators which joints on a mannequin corresponded to their own tender or swollen joints. They also ranked their global health assessment on a visual analogue scale from 0 to 10.
A physician blinded to each patient’s own assessment then recorded an assessment of each patient’s tender joint count and swollen joint count, as well as the physician’s perception of the patient’s global assessment of disease activity.
Dr. Janta and her associates found that the mean scores calculated on the Disease Activity Score 28 (DAS28) were significantly higher according to patients, at 3.34, compared with the physician tally, at 2.55 (P less than .0005).
Indeed, even though the attending rheumatologist had judged all 69 patients to be in clinical remission, DAS28 scores calculated from the patients’ assessments indicated that only 18 patients (26%) fit this bill. But according to the physician assessment, that number was 36 patients (52%) – another significant difference (P less than .0005).
Next, the researchers assessed concordance between doctors and patients on the simplified disease activity index (SDAI). Here, there was better agreement: The mean score assessment among patients was 12.33, compared with a physician mean assessment of 10.55 (P = .246). That translated to a statistically similar SDAI-based remission rate of 15% according to patients vs. 12% according to physicians (P = .172).
Finally, ultrasound examinations with B mode and power Doppler showed no significant correlations with patient- and physician-derived clinical assessments. The mean number of tender joints was 4.34 based on patient assessment, 3.12 by physician examination, and 4.09 based on B-mode ultrasound.
On the other hand, swollen joint counts (SJCs) were significantly lower in both patient (2.00) and physician (1.42) estimation, compared with B-mode ultrasound (4.09) (P less than .0005 for ultrasound vs. patient mean SJC; P = .033 for ultrasound vs. physician SJC).
The ultrasound results on power Doppler were reversed in comparison with B-mode assessment. Tender joint counts were much higher when estimated by patients (4.34) and physicians (3.12), compared with Doppler ultrasound (1.1, P less than .0005 for both).
But mean swollen joint counts on power Doppler ultrasound (1.1) correlated well with patient- and physician-assessed counts (2 and 1.42, respectively).
Dr. Janta and her associates wrote that in the future, given the current trend in patient involvement in disease activity assessments throughout the field, "longitudinal studies should investigate the predictive capability of patient self-assessment of RA activity in relation to relevant disease outcomes, which will address the real value of the discrepancies between patient-assessed and physician-assessed RA activity."
They also noted that "studies on the optimal weight of power Doppler findings in global RA activity assessment are needed."
The authors stated that they had no relevant financial conflicts. They did not report any outside funding for this study.
FROM RHEUMATOLOGY
Major finding: Disease Activity Score 28 ratings (DAS28) were significantly higher when patients assessed themselves (mean = 3.34) than when their physician did so (mean = 2.55; P less than .0005).
Data source: A prospective cross-sectional study of 69 rheumatoid arthritis patients judged to be in remission by their attending rheumatologist.
Disclosures: The authors stated that they had no relevant financial conflicts. They did not report any outside funding for this study.
Study sheds light on exacerbation risks with RA lung disease
An older age at the time of diagnosis and treatment with methotrexate were significantly associated with an increased risk of developing "acute exacerbation" of rheumatoid arthritis–associated interstitial lung disease in a retrospective cohort study of 51 patients.
The other risk factor associated with the development of acute exacerbation (AE) was a specific pattern on high-resolution CT scan, which was also associated with poorer survival, reported Dr. Hironao Hozumi, of Hamamatsu (Japan) University, and associates.
Overall, survival was also significantly lower among those who developed AE, according to the study. As far as the authors know, this is the first study to investigate the risk factors and prognosis associated with AE in patients who have been diagnosed with rheumatoid arthritis–associated interstitial lung disease (RA-ILD). Until this study, risk factors and prognosis for AEs in patients with RA-ILD have been unclear, they said.
AE is "a recently established and an increasingly recognized occurrence" in people with idiopathic pulmonary fibrosis, and it also affects people with other types of interstitial lung diseases, including those associated with collagen vascular disease, the authors noted. It was defined in the study as "acute deterioration in respiratory status, with newly developed bilateral ground-glass opacities and/or consolidations" visible on chest x-ray or CT scans.
The retrospective case-control study evaluated medical records and images of 51 patients consecutively diagnosed with RA-ILD at Hamamatsu University Hospital between 1995 and 2012. The median ages at the time of the RA and ILD diagnoses were 61 and 62 years, respectively. A majority (57%) of the patients were men. The patients had been followed for a median of 8.5 years (range, 1-17 years) before being diagnosed with AE (BMJ Open 2013;3:e003132).
During the observation period of 1-11 years, 11 of the 51 patients (22%) developed AE at a median age of 72 years (range, 60-86 years). In a univariate Cox hazard analysis, an older age at ILD diagnosis was associated with an 11% increased risk for AE occurrence.
Of the 11 patients who developed AE, 7 (64%) died of respiratory failure during the initial episode of AE, compared with 2 (5%) of the 40 patients who did not develop AE.
There was a usual interstitial pneumonia (UIP) pattern on high-resolution CT in 14 (27%) of the 51 patients. This pattern was found in 6 (55%) of the patients who had AE, compared with 8 (20%) of the 40 patients with no AE, a significant difference. This comparison gave a hazard ratio of 1.95 in a univariate Cox hazard analysis. For AE occurrence, a UIP pattern on high-resolution CT had a positive predictive value of 43% and a negative predictive value of 87%. The 1-year incidence of AE was 6.5% among those with a UIP pattern on high-resolution CT, compared with an overall rate of 2.8% in the study.
The overall 5-year survival was 90% for all the patients, but there was a significant difference among those with or without the UIP pattern (70% vs. 97%). Patients’ risk of death more than doubled with an AE occurrence (hazard ratio, 2.47).
The use of methotrexate was associated with a threefold increased risk of developing AE (HR, 3.04) in a univariate Cox hazard analysis. Of 10 patients treated with methotrexate, 6 (55%) in the AE group used the drug (including 5 who had been treated for at least 3 years and 1 treated for 1 year), compared with 4 (10%) in the non-AE group, a significant difference. Although methotrexate was discontinued, the respiratory condition of all six patients in the AE group continued to deteriorate and they had poor responses to corticosteroid therapy.
The authors found no associations with disease activity and the development of AE, so RA activity may not be related to the development of AE, the authors wrote. However, they noted that methotrexate "possibly accelerates the fibroproliferative process of RA-ILD."
There also were no significant differences in factors that included sex, age at the time of diagnosis of RA or RA-ILD, smoking habits, and other factors between those who did and did not develop an AE.
The investigators said that the limitations of the study included the retrospective design and the small sample size, and that "larger prospective studies investigating AEs in RA-ILD are indicated."
The authors had no disclosures to report. The study was partly funded by a grant from Japan’s Ministry of Health, Labor, and Welfare to the Diffuse Lung Diseases Research Group.
An older age at the time of diagnosis and treatment with methotrexate were significantly associated with an increased risk of developing "acute exacerbation" of rheumatoid arthritis–associated interstitial lung disease in a retrospective cohort study of 51 patients.
The other risk factor associated with the development of acute exacerbation (AE) was a specific pattern on high-resolution CT scan, which was also associated with poorer survival, reported Dr. Hironao Hozumi, of Hamamatsu (Japan) University, and associates.
Overall, survival was also significantly lower among those who developed AE, according to the study. As far as the authors know, this is the first study to investigate the risk factors and prognosis associated with AE in patients who have been diagnosed with rheumatoid arthritis–associated interstitial lung disease (RA-ILD). Until this study, risk factors and prognosis for AEs in patients with RA-ILD have been unclear, they said.
AE is "a recently established and an increasingly recognized occurrence" in people with idiopathic pulmonary fibrosis, and it also affects people with other types of interstitial lung diseases, including those associated with collagen vascular disease, the authors noted. It was defined in the study as "acute deterioration in respiratory status, with newly developed bilateral ground-glass opacities and/or consolidations" visible on chest x-ray or CT scans.
The retrospective case-control study evaluated medical records and images of 51 patients consecutively diagnosed with RA-ILD at Hamamatsu University Hospital between 1995 and 2012. The median ages at the time of the RA and ILD diagnoses were 61 and 62 years, respectively. A majority (57%) of the patients were men. The patients had been followed for a median of 8.5 years (range, 1-17 years) before being diagnosed with AE (BMJ Open 2013;3:e003132).
During the observation period of 1-11 years, 11 of the 51 patients (22%) developed AE at a median age of 72 years (range, 60-86 years). In a univariate Cox hazard analysis, an older age at ILD diagnosis was associated with an 11% increased risk for AE occurrence.
Of the 11 patients who developed AE, 7 (64%) died of respiratory failure during the initial episode of AE, compared with 2 (5%) of the 40 patients who did not develop AE.
There was a usual interstitial pneumonia (UIP) pattern on high-resolution CT in 14 (27%) of the 51 patients. This pattern was found in 6 (55%) of the patients who had AE, compared with 8 (20%) of the 40 patients with no AE, a significant difference. This comparison gave a hazard ratio of 1.95 in a univariate Cox hazard analysis. For AE occurrence, a UIP pattern on high-resolution CT had a positive predictive value of 43% and a negative predictive value of 87%. The 1-year incidence of AE was 6.5% among those with a UIP pattern on high-resolution CT, compared with an overall rate of 2.8% in the study.
The overall 5-year survival was 90% for all the patients, but there was a significant difference among those with or without the UIP pattern (70% vs. 97%). Patients’ risk of death more than doubled with an AE occurrence (hazard ratio, 2.47).
The use of methotrexate was associated with a threefold increased risk of developing AE (HR, 3.04) in a univariate Cox hazard analysis. Of 10 patients treated with methotrexate, 6 (55%) in the AE group used the drug (including 5 who had been treated for at least 3 years and 1 treated for 1 year), compared with 4 (10%) in the non-AE group, a significant difference. Although methotrexate was discontinued, the respiratory condition of all six patients in the AE group continued to deteriorate and they had poor responses to corticosteroid therapy.
The authors found no associations with disease activity and the development of AE, so RA activity may not be related to the development of AE, the authors wrote. However, they noted that methotrexate "possibly accelerates the fibroproliferative process of RA-ILD."
There also were no significant differences in factors that included sex, age at the time of diagnosis of RA or RA-ILD, smoking habits, and other factors between those who did and did not develop an AE.
The investigators said that the limitations of the study included the retrospective design and the small sample size, and that "larger prospective studies investigating AEs in RA-ILD are indicated."
The authors had no disclosures to report. The study was partly funded by a grant from Japan’s Ministry of Health, Labor, and Welfare to the Diffuse Lung Diseases Research Group.
An older age at the time of diagnosis and treatment with methotrexate were significantly associated with an increased risk of developing "acute exacerbation" of rheumatoid arthritis–associated interstitial lung disease in a retrospective cohort study of 51 patients.
The other risk factor associated with the development of acute exacerbation (AE) was a specific pattern on high-resolution CT scan, which was also associated with poorer survival, reported Dr. Hironao Hozumi, of Hamamatsu (Japan) University, and associates.
Overall, survival was also significantly lower among those who developed AE, according to the study. As far as the authors know, this is the first study to investigate the risk factors and prognosis associated with AE in patients who have been diagnosed with rheumatoid arthritis–associated interstitial lung disease (RA-ILD). Until this study, risk factors and prognosis for AEs in patients with RA-ILD have been unclear, they said.
AE is "a recently established and an increasingly recognized occurrence" in people with idiopathic pulmonary fibrosis, and it also affects people with other types of interstitial lung diseases, including those associated with collagen vascular disease, the authors noted. It was defined in the study as "acute deterioration in respiratory status, with newly developed bilateral ground-glass opacities and/or consolidations" visible on chest x-ray or CT scans.
The retrospective case-control study evaluated medical records and images of 51 patients consecutively diagnosed with RA-ILD at Hamamatsu University Hospital between 1995 and 2012. The median ages at the time of the RA and ILD diagnoses were 61 and 62 years, respectively. A majority (57%) of the patients were men. The patients had been followed for a median of 8.5 years (range, 1-17 years) before being diagnosed with AE (BMJ Open 2013;3:e003132).
During the observation period of 1-11 years, 11 of the 51 patients (22%) developed AE at a median age of 72 years (range, 60-86 years). In a univariate Cox hazard analysis, an older age at ILD diagnosis was associated with an 11% increased risk for AE occurrence.
Of the 11 patients who developed AE, 7 (64%) died of respiratory failure during the initial episode of AE, compared with 2 (5%) of the 40 patients who did not develop AE.
There was a usual interstitial pneumonia (UIP) pattern on high-resolution CT in 14 (27%) of the 51 patients. This pattern was found in 6 (55%) of the patients who had AE, compared with 8 (20%) of the 40 patients with no AE, a significant difference. This comparison gave a hazard ratio of 1.95 in a univariate Cox hazard analysis. For AE occurrence, a UIP pattern on high-resolution CT had a positive predictive value of 43% and a negative predictive value of 87%. The 1-year incidence of AE was 6.5% among those with a UIP pattern on high-resolution CT, compared with an overall rate of 2.8% in the study.
The overall 5-year survival was 90% for all the patients, but there was a significant difference among those with or without the UIP pattern (70% vs. 97%). Patients’ risk of death more than doubled with an AE occurrence (hazard ratio, 2.47).
The use of methotrexate was associated with a threefold increased risk of developing AE (HR, 3.04) in a univariate Cox hazard analysis. Of 10 patients treated with methotrexate, 6 (55%) in the AE group used the drug (including 5 who had been treated for at least 3 years and 1 treated for 1 year), compared with 4 (10%) in the non-AE group, a significant difference. Although methotrexate was discontinued, the respiratory condition of all six patients in the AE group continued to deteriorate and they had poor responses to corticosteroid therapy.
The authors found no associations with disease activity and the development of AE, so RA activity may not be related to the development of AE, the authors wrote. However, they noted that methotrexate "possibly accelerates the fibroproliferative process of RA-ILD."
There also were no significant differences in factors that included sex, age at the time of diagnosis of RA or RA-ILD, smoking habits, and other factors between those who did and did not develop an AE.
The investigators said that the limitations of the study included the retrospective design and the small sample size, and that "larger prospective studies investigating AEs in RA-ILD are indicated."
The authors had no disclosures to report. The study was partly funded by a grant from Japan’s Ministry of Health, Labor, and Welfare to the Diffuse Lung Diseases Research Group.
FROM BMJ OPEN
Major finding: The use of methotrexate was associated with a threefold increased risk of developing acute exacerbation of RA-ILD (HR, 3.04) in a univariate Cox hazard analysis.
Data source: A retrospective case-control study that looked at the medical records and images of 51 patients diagnosed with RA-ILD at an academic hospital in Japan between 1995 and 2012.
Disclosures: The authors had no disclosures to report. The study was partly funded by a grant from Japan’s Ministry of Health, Labor, and Welfare to the Diffuse Lung Diseases Research Group.
NSAIDs, methotrexate used most for rheumatoid arthritis
NSAIDs and methotrexate are the medications most often used by patients to treat rheumatoid arthritis, according to a survey conducted by rheumatoidarthritis.net.
Among those who answered the question on drug therapy, 86% had taken NSAIDs at some time and almost 54% were currently taking them, while 79% reported lifetime use of methotrexate and almost 43% were currently taking it. Oral steroids were third in lifetime use at 72%, but biologics were actually second in current use at 45%, the survey showed.
When looking at use of specific biologics, etanercept (Enbrel) led in lifetime use at 30% while adalimumab (Humira) was second at 25%. The top two spots were reversed for current use, with etanercept at 26% and adalimumab at 25%, rheumatoidarthritis.net reported. (The number of respondents varied for each question since not all patients were currently treating their RA with the options offered.) Infliximab (Remicade) was third in lifetime use (15%) and abatacept (Orencia) was third in current use (13%).
The survey also examined cost issues. Among respondents, more than 44% reported spending $1,000-$5,000 a year on RA treatment, 41% spent less than $1,000, 12% spent $5,000-$10,000, and 3% spent more than $10,000 a year. Almost 44% of RA patients said that they had avoided treatment at some time because of cost, and 32% reported using a coupon or free trial offer to help pay for treatment, according to the survey.
A total of 706 RA patients over age 18 completed the online "RA in America 2013" survey, which was conducted from June to August of 2013. The parent company of rheumatoidarthritis.net, Health Union LLC, reports that it "does not manufacture, sell nor market any product to diagnose, prevent or treat RA or any other disease."
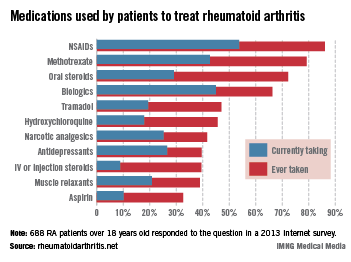
NSAIDs and methotrexate are the medications most often used by patients to treat rheumatoid arthritis, according to a survey conducted by rheumatoidarthritis.net.
Among those who answered the question on drug therapy, 86% had taken NSAIDs at some time and almost 54% were currently taking them, while 79% reported lifetime use of methotrexate and almost 43% were currently taking it. Oral steroids were third in lifetime use at 72%, but biologics were actually second in current use at 45%, the survey showed.
When looking at use of specific biologics, etanercept (Enbrel) led in lifetime use at 30% while adalimumab (Humira) was second at 25%. The top two spots were reversed for current use, with etanercept at 26% and adalimumab at 25%, rheumatoidarthritis.net reported. (The number of respondents varied for each question since not all patients were currently treating their RA with the options offered.) Infliximab (Remicade) was third in lifetime use (15%) and abatacept (Orencia) was third in current use (13%).
The survey also examined cost issues. Among respondents, more than 44% reported spending $1,000-$5,000 a year on RA treatment, 41% spent less than $1,000, 12% spent $5,000-$10,000, and 3% spent more than $10,000 a year. Almost 44% of RA patients said that they had avoided treatment at some time because of cost, and 32% reported using a coupon or free trial offer to help pay for treatment, according to the survey.
A total of 706 RA patients over age 18 completed the online "RA in America 2013" survey, which was conducted from June to August of 2013. The parent company of rheumatoidarthritis.net, Health Union LLC, reports that it "does not manufacture, sell nor market any product to diagnose, prevent or treat RA or any other disease."

NSAIDs and methotrexate are the medications most often used by patients to treat rheumatoid arthritis, according to a survey conducted by rheumatoidarthritis.net.
Among those who answered the question on drug therapy, 86% had taken NSAIDs at some time and almost 54% were currently taking them, while 79% reported lifetime use of methotrexate and almost 43% were currently taking it. Oral steroids were third in lifetime use at 72%, but biologics were actually second in current use at 45%, the survey showed.
When looking at use of specific biologics, etanercept (Enbrel) led in lifetime use at 30% while adalimumab (Humira) was second at 25%. The top two spots were reversed for current use, with etanercept at 26% and adalimumab at 25%, rheumatoidarthritis.net reported. (The number of respondents varied for each question since not all patients were currently treating their RA with the options offered.) Infliximab (Remicade) was third in lifetime use (15%) and abatacept (Orencia) was third in current use (13%).
The survey also examined cost issues. Among respondents, more than 44% reported spending $1,000-$5,000 a year on RA treatment, 41% spent less than $1,000, 12% spent $5,000-$10,000, and 3% spent more than $10,000 a year. Almost 44% of RA patients said that they had avoided treatment at some time because of cost, and 32% reported using a coupon or free trial offer to help pay for treatment, according to the survey.
A total of 706 RA patients over age 18 completed the online "RA in America 2013" survey, which was conducted from June to August of 2013. The parent company of rheumatoidarthritis.net, Health Union LLC, reports that it "does not manufacture, sell nor market any product to diagnose, prevent or treat RA or any other disease."

Patient global score predicts postbiological quality of life gain in RA
Danish researchers report that rheumatoid arthritis patients’ baseline patient global score component of the Disease-Activity Score–28 tool was most strongly correlated with patient-reported outcomes and was the best predictor of a gain in quality adjusted life years following 1 year of biological therapy in a prospective observational study.
An objective C-reactive protein (CRP) measure, however, had no predictive value, and no sharp demarcation between objective and subjective variables was apparent.
The finding that patient global score at baseline may drive a gain in quality adjusted life years (QALY) could improve understanding of which disease-related factors might affect the cost per QALY gains (an important measure for comparing the cost-effectiveness of treatments) and predict treatment response, according to Dr. Louise Linde of the DANBIO Registry, Glostrup, Denmark, and her colleagues (J. Rheumatol. 2013;40:1479-86).
Of 282 patients who completed the study, 68% gained QALYs following 1 year of biological therapy (mean of 0.14), 61% were European League Against Rheumatism (EULAR) criteria responders, and 62% achieved minimally important differences based on the European Quality of Life 5 Dimensions (EQ-5D) questionnaire.
The proportion of patients achieving both a EULAR and EQ-5D response was 55%. Cross-tabulation by response groups (EULAR response/EQ-5D response, EULAR no response/EQ-5D no response, EULAR response/EQ-5D no response, and EULAR no response/EQ-5D response) showed that the mean reduction in swollen joints across the groups ranged from 0 to 6 with clinically and statistically significant differences in three of six possible comparisons.
Similar patterns were seen for tender joints and patient global score, the investigators said.
"Thus, the mean reduction in tender joints ranged from –1 to 9 across the response groups. In addition, the EULAR no response/EQ-5D response group could be discriminated from the EULAR response/EQ-5D response group, leading to significant differences (3-9 tender joints) in four of six comparisons. Similarly, the mean reduction in patient global score ranged from –0.7 to 4.0 cm across the response groups, and highly significant differences of 2.0-4.7 cm were observed between the response groups in five of six comparisons," they said.
However, the mean reduction in CRP, which ranged from 6.0 to 16.5 mg/L across the response groups, distinguished only the EULAR response/EQ-5D response from the EULAR nonresponders.
All EULAR responders experienced significant reductions in swollen and tender joints, CRP, and patient global scores – regardless of EQ-5D status – and the EULAR nonresponders who had an EQ-5D response experienced similar improvements. However, those with no EQ-5D response experienced a worsening in patient global score of 0.7.
Patient global score was strongly correlated with responses on both the EULAR and European Quality of Life 5 Dimensions EQ-5D.
Regression analysis demonstrated that "the gain in QALY after 1 year of biological therapy increased with increasing baseline patient global score and number of swollen joints, and patients with two or more extraarticular manifestations gained more QALY compared with those without such manifestations," the investigators wrote.
"This finding may reflect the likely scenario that patients with very active or severe disease have more room for improvement. This does not imply, however that such patients will have a more favorable final status than those with less active or less severe disease at baseline, a conclusion not supported by our observational data," they noted.
The mean age of the patients in this study was 55 years. They were recruited from 17 routine care sites between November 2005 and July 2007. Clinical and patient-reported data were collected at baseline and after 3, 6, and 12 months of therapy. Clinical data were obtained via the Danish DANBIO registry, which includes 90% of Danish patients with RA who are receiving biological therapy. Collected clinical data included disease duration, 28 swollen and tender joint counts, CRP, patient global score on a visual analog scale, IgM-rheumatoid factor status, number of previous biological therapies, and concomitant use of methotrexate and glucocorticoids. Self-reported patient data included marital status, education, smoking behavior, body mass index, exercise habits, extra-articular features, joint surgery, and comorbidities.
In addition to the EQ-5D, participants also completed the validated Danish Health Assessment Questionnaire.
Though limited by the factors inherent in observational study design, the study is strengthened by the nationwide recruitment, the fact that is was carried out in a clinical setting, and the fact that the study population had similar baseline demographic, clinical, and patent-reported values as have been reported for other RA patients treated with biological therapies in routine care in several other registries.
"This result suggests that our findings may be generalizable to patients with RA treated in routine care with biologicals in countries with a public healthcare system similar to the Danish model," the investigators wrote.
An important – and perhaps critical – task for the rheumatology community is to document effective treatment, even though only a small proportion of patients achieve clinical remission, according to Dr. Theodore Pincus and his colleagues.
"This goal might be met more effectively by analyzing efforts to control the most significant individual core data set measures to achieve improved quality of life and reversal of premature mortality – patient global estimate and physical function on a patient questionnaire – rather than by focusing on indices or remission," they wrote in an editorial (J. Rheumatol. 2013;40:1469-74).

|
|
The findings by Dr. Linde and her colleagues document the value of a patient global estimate of status to explain improvement in quality of life associated with treatment, and suggest that control of patient global estimate could be more valuable than factors such as joint counts, radiographic scores, or laboratory tests for justifying expensive RA treatments.
The findings underscore the importance of assessing patient global estimate and physical function – another important factor for predicting outcomes. These can be assessed easily on the Health Assessment Questionnaire (HAQ) or the multidimensional HAQ, they noted.
"However, quantitative clinical data from a patient history in the form of a patient self-report questionnaire, and even from physical examination in the form of joint counts and physician global estimate, have not been incorporated by most clinical rheumatologists, at least in the United States," they wrote.
Completing these questionnaires creates no extra work or patient flow interference, as they could be done by patients in the waiting area, and could provide documentation of the value of expensive treatments.
"Routine collection of these quantitative data ... rather than simple notation of gestalt impressions by the physician, would appear to be an intellectual and ethical responsibility to our patients with RA," they concluded.
Dr. Pincus is with New York University and NYU Hospital for Joint Diseases, New York. No disclosures were reported.
An important – and perhaps critical – task for the rheumatology community is to document effective treatment, even though only a small proportion of patients achieve clinical remission, according to Dr. Theodore Pincus and his colleagues.
"This goal might be met more effectively by analyzing efforts to control the most significant individual core data set measures to achieve improved quality of life and reversal of premature mortality – patient global estimate and physical function on a patient questionnaire – rather than by focusing on indices or remission," they wrote in an editorial (J. Rheumatol. 2013;40:1469-74).

|
|
The findings by Dr. Linde and her colleagues document the value of a patient global estimate of status to explain improvement in quality of life associated with treatment, and suggest that control of patient global estimate could be more valuable than factors such as joint counts, radiographic scores, or laboratory tests for justifying expensive RA treatments.
The findings underscore the importance of assessing patient global estimate and physical function – another important factor for predicting outcomes. These can be assessed easily on the Health Assessment Questionnaire (HAQ) or the multidimensional HAQ, they noted.
"However, quantitative clinical data from a patient history in the form of a patient self-report questionnaire, and even from physical examination in the form of joint counts and physician global estimate, have not been incorporated by most clinical rheumatologists, at least in the United States," they wrote.
Completing these questionnaires creates no extra work or patient flow interference, as they could be done by patients in the waiting area, and could provide documentation of the value of expensive treatments.
"Routine collection of these quantitative data ... rather than simple notation of gestalt impressions by the physician, would appear to be an intellectual and ethical responsibility to our patients with RA," they concluded.
Dr. Pincus is with New York University and NYU Hospital for Joint Diseases, New York. No disclosures were reported.
An important – and perhaps critical – task for the rheumatology community is to document effective treatment, even though only a small proportion of patients achieve clinical remission, according to Dr. Theodore Pincus and his colleagues.
"This goal might be met more effectively by analyzing efforts to control the most significant individual core data set measures to achieve improved quality of life and reversal of premature mortality – patient global estimate and physical function on a patient questionnaire – rather than by focusing on indices or remission," they wrote in an editorial (J. Rheumatol. 2013;40:1469-74).

|
|
The findings by Dr. Linde and her colleagues document the value of a patient global estimate of status to explain improvement in quality of life associated with treatment, and suggest that control of patient global estimate could be more valuable than factors such as joint counts, radiographic scores, or laboratory tests for justifying expensive RA treatments.
The findings underscore the importance of assessing patient global estimate and physical function – another important factor for predicting outcomes. These can be assessed easily on the Health Assessment Questionnaire (HAQ) or the multidimensional HAQ, they noted.
"However, quantitative clinical data from a patient history in the form of a patient self-report questionnaire, and even from physical examination in the form of joint counts and physician global estimate, have not been incorporated by most clinical rheumatologists, at least in the United States," they wrote.
Completing these questionnaires creates no extra work or patient flow interference, as they could be done by patients in the waiting area, and could provide documentation of the value of expensive treatments.
"Routine collection of these quantitative data ... rather than simple notation of gestalt impressions by the physician, would appear to be an intellectual and ethical responsibility to our patients with RA," they concluded.
Dr. Pincus is with New York University and NYU Hospital for Joint Diseases, New York. No disclosures were reported.
Danish researchers report that rheumatoid arthritis patients’ baseline patient global score component of the Disease-Activity Score–28 tool was most strongly correlated with patient-reported outcomes and was the best predictor of a gain in quality adjusted life years following 1 year of biological therapy in a prospective observational study.
An objective C-reactive protein (CRP) measure, however, had no predictive value, and no sharp demarcation between objective and subjective variables was apparent.
The finding that patient global score at baseline may drive a gain in quality adjusted life years (QALY) could improve understanding of which disease-related factors might affect the cost per QALY gains (an important measure for comparing the cost-effectiveness of treatments) and predict treatment response, according to Dr. Louise Linde of the DANBIO Registry, Glostrup, Denmark, and her colleagues (J. Rheumatol. 2013;40:1479-86).
Of 282 patients who completed the study, 68% gained QALYs following 1 year of biological therapy (mean of 0.14), 61% were European League Against Rheumatism (EULAR) criteria responders, and 62% achieved minimally important differences based on the European Quality of Life 5 Dimensions (EQ-5D) questionnaire.
The proportion of patients achieving both a EULAR and EQ-5D response was 55%. Cross-tabulation by response groups (EULAR response/EQ-5D response, EULAR no response/EQ-5D no response, EULAR response/EQ-5D no response, and EULAR no response/EQ-5D response) showed that the mean reduction in swollen joints across the groups ranged from 0 to 6 with clinically and statistically significant differences in three of six possible comparisons.
Similar patterns were seen for tender joints and patient global score, the investigators said.
"Thus, the mean reduction in tender joints ranged from –1 to 9 across the response groups. In addition, the EULAR no response/EQ-5D response group could be discriminated from the EULAR response/EQ-5D response group, leading to significant differences (3-9 tender joints) in four of six comparisons. Similarly, the mean reduction in patient global score ranged from –0.7 to 4.0 cm across the response groups, and highly significant differences of 2.0-4.7 cm were observed between the response groups in five of six comparisons," they said.
However, the mean reduction in CRP, which ranged from 6.0 to 16.5 mg/L across the response groups, distinguished only the EULAR response/EQ-5D response from the EULAR nonresponders.
All EULAR responders experienced significant reductions in swollen and tender joints, CRP, and patient global scores – regardless of EQ-5D status – and the EULAR nonresponders who had an EQ-5D response experienced similar improvements. However, those with no EQ-5D response experienced a worsening in patient global score of 0.7.
Patient global score was strongly correlated with responses on both the EULAR and European Quality of Life 5 Dimensions EQ-5D.
Regression analysis demonstrated that "the gain in QALY after 1 year of biological therapy increased with increasing baseline patient global score and number of swollen joints, and patients with two or more extraarticular manifestations gained more QALY compared with those without such manifestations," the investigators wrote.
"This finding may reflect the likely scenario that patients with very active or severe disease have more room for improvement. This does not imply, however that such patients will have a more favorable final status than those with less active or less severe disease at baseline, a conclusion not supported by our observational data," they noted.
The mean age of the patients in this study was 55 years. They were recruited from 17 routine care sites between November 2005 and July 2007. Clinical and patient-reported data were collected at baseline and after 3, 6, and 12 months of therapy. Clinical data were obtained via the Danish DANBIO registry, which includes 90% of Danish patients with RA who are receiving biological therapy. Collected clinical data included disease duration, 28 swollen and tender joint counts, CRP, patient global score on a visual analog scale, IgM-rheumatoid factor status, number of previous biological therapies, and concomitant use of methotrexate and glucocorticoids. Self-reported patient data included marital status, education, smoking behavior, body mass index, exercise habits, extra-articular features, joint surgery, and comorbidities.
In addition to the EQ-5D, participants also completed the validated Danish Health Assessment Questionnaire.
Though limited by the factors inherent in observational study design, the study is strengthened by the nationwide recruitment, the fact that is was carried out in a clinical setting, and the fact that the study population had similar baseline demographic, clinical, and patent-reported values as have been reported for other RA patients treated with biological therapies in routine care in several other registries.
"This result suggests that our findings may be generalizable to patients with RA treated in routine care with biologicals in countries with a public healthcare system similar to the Danish model," the investigators wrote.
Danish researchers report that rheumatoid arthritis patients’ baseline patient global score component of the Disease-Activity Score–28 tool was most strongly correlated with patient-reported outcomes and was the best predictor of a gain in quality adjusted life years following 1 year of biological therapy in a prospective observational study.
An objective C-reactive protein (CRP) measure, however, had no predictive value, and no sharp demarcation between objective and subjective variables was apparent.
The finding that patient global score at baseline may drive a gain in quality adjusted life years (QALY) could improve understanding of which disease-related factors might affect the cost per QALY gains (an important measure for comparing the cost-effectiveness of treatments) and predict treatment response, according to Dr. Louise Linde of the DANBIO Registry, Glostrup, Denmark, and her colleagues (J. Rheumatol. 2013;40:1479-86).
Of 282 patients who completed the study, 68% gained QALYs following 1 year of biological therapy (mean of 0.14), 61% were European League Against Rheumatism (EULAR) criteria responders, and 62% achieved minimally important differences based on the European Quality of Life 5 Dimensions (EQ-5D) questionnaire.
The proportion of patients achieving both a EULAR and EQ-5D response was 55%. Cross-tabulation by response groups (EULAR response/EQ-5D response, EULAR no response/EQ-5D no response, EULAR response/EQ-5D no response, and EULAR no response/EQ-5D response) showed that the mean reduction in swollen joints across the groups ranged from 0 to 6 with clinically and statistically significant differences in three of six possible comparisons.
Similar patterns were seen for tender joints and patient global score, the investigators said.
"Thus, the mean reduction in tender joints ranged from –1 to 9 across the response groups. In addition, the EULAR no response/EQ-5D response group could be discriminated from the EULAR response/EQ-5D response group, leading to significant differences (3-9 tender joints) in four of six comparisons. Similarly, the mean reduction in patient global score ranged from –0.7 to 4.0 cm across the response groups, and highly significant differences of 2.0-4.7 cm were observed between the response groups in five of six comparisons," they said.
However, the mean reduction in CRP, which ranged from 6.0 to 16.5 mg/L across the response groups, distinguished only the EULAR response/EQ-5D response from the EULAR nonresponders.
All EULAR responders experienced significant reductions in swollen and tender joints, CRP, and patient global scores – regardless of EQ-5D status – and the EULAR nonresponders who had an EQ-5D response experienced similar improvements. However, those with no EQ-5D response experienced a worsening in patient global score of 0.7.
Patient global score was strongly correlated with responses on both the EULAR and European Quality of Life 5 Dimensions EQ-5D.
Regression analysis demonstrated that "the gain in QALY after 1 year of biological therapy increased with increasing baseline patient global score and number of swollen joints, and patients with two or more extraarticular manifestations gained more QALY compared with those without such manifestations," the investigators wrote.
"This finding may reflect the likely scenario that patients with very active or severe disease have more room for improvement. This does not imply, however that such patients will have a more favorable final status than those with less active or less severe disease at baseline, a conclusion not supported by our observational data," they noted.
The mean age of the patients in this study was 55 years. They were recruited from 17 routine care sites between November 2005 and July 2007. Clinical and patient-reported data were collected at baseline and after 3, 6, and 12 months of therapy. Clinical data were obtained via the Danish DANBIO registry, which includes 90% of Danish patients with RA who are receiving biological therapy. Collected clinical data included disease duration, 28 swollen and tender joint counts, CRP, patient global score on a visual analog scale, IgM-rheumatoid factor status, number of previous biological therapies, and concomitant use of methotrexate and glucocorticoids. Self-reported patient data included marital status, education, smoking behavior, body mass index, exercise habits, extra-articular features, joint surgery, and comorbidities.
In addition to the EQ-5D, participants also completed the validated Danish Health Assessment Questionnaire.
Though limited by the factors inherent in observational study design, the study is strengthened by the nationwide recruitment, the fact that is was carried out in a clinical setting, and the fact that the study population had similar baseline demographic, clinical, and patent-reported values as have been reported for other RA patients treated with biological therapies in routine care in several other registries.
"This result suggests that our findings may be generalizable to patients with RA treated in routine care with biologicals in countries with a public healthcare system similar to the Danish model," the investigators wrote.
FROM THE JOURNAL OF RHEUMATOLOGY
Major finding: Highly significant differences of 2.0-4.7 cm on the patient global score were observed between the response groups in five of six comparisons according to response or no response based on EULAR and EQ-5D criteria.
Data source: A prospective observational study of 282 RA patients.
Disclosures: No disclosures were reported.
Biosimilar matches up well against infliximab in RA trial
The investigational biosimilar drug CT-P13 proved to have equivalent efficacy and safety comparable to that of infliximab, the tumor necrosis factor–alpha inhibitor that it is manufactured to mimic.
The findings are based on data from a randomized, double-blind, multicenter trial of 606 patients with active rheumatoid arthritis who had inadequate response to methotrexate alone.
Dr. Dae Hyun Yoo of Hanyang University Hospital for Rheumatic Diseases in Seoul, South Korea, reported no difference in the percentage of patients who met American College of Rheumatology 20% improvement criteria (ACR20) at 30 weeks (60.9% for CT-P13 vs. 58.6% for infliximab), which was the primary outcome of the study. The 95% confidence intervals for CT-P13 (–6%-10%) and infliximab (–4%-12%) were within the prespecified range of plus or minus 15% for equivalency (Ann. Rheum. Dis. 2013;72:1613-20).
The overall number of patients who reported treatment-emergent adverse events was virtually the same in patients who took CT-P13 (181) or infliximab (183). The adverse events that were deemed to be related to the study medications also occurred in a similar number of CT-P13 (106) and infliximab (108) patients. Most of these were mild to moderate in intensity. The trial’s safety objective was to determine whether CT-P13 had safety that was comparable, not equivalent, to that of infliximab.
Biosimilar drugs are meant to be "highly similar, but not identical and not ‘bioidentical,’ to approved ‘reference agents,’ " the authors wrote. CT-P13 is an immunoglobulin G1 chimeric human-murine monoclonal antibody to tumor necrosis factor–alpha (TNF-alpha) that has an amino acid sequence identical to that of infliximab. It is produced in the same type of cell line as the one used to produce infliximab.
In this study, known as PLANETRA (Program evaLuating the Autoimmune Disease iNvEstigational Drug cT-p13 in RA Patients), the investigators enrolled patients according to the same criteria as those used in several other clinical trials with infliximab, including the ATTRACT study for which infliximab was approved for the treatment of RA. The results obtained with CT-P13 were similar to those of other RA trials with infliximab that had the same enrollment criteria. The trial was conducted at 100 centers in 19 countries in Europe, Asia, Latin America, and the Middle East.
Patients in the trial met the revised 1987 ACR classification criteria for at least 1 year prior to screening, and were required to have at least six swollen and at least six tender joints and at least two of the following: morning stiffness lasting 45 or more minutes; serum C-reactive protein (CRP) level greater than 2.0 mg/dL; and an erythrocyte sedimentation rate of greater than 28 mm/hour. They needed to meet those criteria despite taking methotrexate for at least 3 months on a stable dose of 12.5-25 mg/week for 4 or more weeks prior to screening. Patients were permitted to receive an equivalent of 10 mg prednisolone or less per day and NSAIDs, if they had received a stable dose for at least 4 weeks prior to screening.
Approximately 82% of the patients were female, and the median age of patients was 50 years at baseline. The patients received either study drug at a dose of 3 mg/kg infused over a 2-hour period at weeks 0, 2, and 6, and then every 8 weeks to week 30. The patients also received an antihistamine 30-60 minutes before the infusion, at the investigator’s discretion. Weekly methotrexate doses ranged from 12.5 mg to 25 mg. Patients also received 5 mg/week or more of folic acid.
Because the higher overall CRP level at baseline of the patients in the ATTRACT study could indicate that the PLANETRA cohort had less severe disease (3.1 mg/dL vs./ 1.9 mg/dL), Dr. Yoo and his associates conducted a post hoc sensitivity analysis of patients with baseline CRP greater than 2 mg/dL or 2 mg/dL or less. The results showed no difference in treatment response based on baseline CRP level.
The approximate 7.5% rate of infusion reactions noted in both in the CT-P13 and infliximab arms of the trial was lower than the 20% noted in the product label for infliximab, and antidrug antibody status did not appear to affect their incidence or response to the study drugs.
"Assessment of efficacy and safety of CT-P13 in patients with RA for up to 1 year is ongoing, and the positive results of this study provide a rationale for future studies of CT-P13 in the treatment of other TNF-mediated inflammatory diseases," the investigators wrote.
The study was sponsored by Celltrion. Many of the authors received research grants and/or consultancy fees and logistics support from the company. One author is a full-time employee of Celltrion, and another author declared receiving payment for lectures from the company.
The investigational biosimilar drug CT-P13 proved to have equivalent efficacy and safety comparable to that of infliximab, the tumor necrosis factor–alpha inhibitor that it is manufactured to mimic.
The findings are based on data from a randomized, double-blind, multicenter trial of 606 patients with active rheumatoid arthritis who had inadequate response to methotrexate alone.
Dr. Dae Hyun Yoo of Hanyang University Hospital for Rheumatic Diseases in Seoul, South Korea, reported no difference in the percentage of patients who met American College of Rheumatology 20% improvement criteria (ACR20) at 30 weeks (60.9% for CT-P13 vs. 58.6% for infliximab), which was the primary outcome of the study. The 95% confidence intervals for CT-P13 (–6%-10%) and infliximab (–4%-12%) were within the prespecified range of plus or minus 15% for equivalency (Ann. Rheum. Dis. 2013;72:1613-20).
The overall number of patients who reported treatment-emergent adverse events was virtually the same in patients who took CT-P13 (181) or infliximab (183). The adverse events that were deemed to be related to the study medications also occurred in a similar number of CT-P13 (106) and infliximab (108) patients. Most of these were mild to moderate in intensity. The trial’s safety objective was to determine whether CT-P13 had safety that was comparable, not equivalent, to that of infliximab.
Biosimilar drugs are meant to be "highly similar, but not identical and not ‘bioidentical,’ to approved ‘reference agents,’ " the authors wrote. CT-P13 is an immunoglobulin G1 chimeric human-murine monoclonal antibody to tumor necrosis factor–alpha (TNF-alpha) that has an amino acid sequence identical to that of infliximab. It is produced in the same type of cell line as the one used to produce infliximab.
In this study, known as PLANETRA (Program evaLuating the Autoimmune Disease iNvEstigational Drug cT-p13 in RA Patients), the investigators enrolled patients according to the same criteria as those used in several other clinical trials with infliximab, including the ATTRACT study for which infliximab was approved for the treatment of RA. The results obtained with CT-P13 were similar to those of other RA trials with infliximab that had the same enrollment criteria. The trial was conducted at 100 centers in 19 countries in Europe, Asia, Latin America, and the Middle East.
Patients in the trial met the revised 1987 ACR classification criteria for at least 1 year prior to screening, and were required to have at least six swollen and at least six tender joints and at least two of the following: morning stiffness lasting 45 or more minutes; serum C-reactive protein (CRP) level greater than 2.0 mg/dL; and an erythrocyte sedimentation rate of greater than 28 mm/hour. They needed to meet those criteria despite taking methotrexate for at least 3 months on a stable dose of 12.5-25 mg/week for 4 or more weeks prior to screening. Patients were permitted to receive an equivalent of 10 mg prednisolone or less per day and NSAIDs, if they had received a stable dose for at least 4 weeks prior to screening.
Approximately 82% of the patients were female, and the median age of patients was 50 years at baseline. The patients received either study drug at a dose of 3 mg/kg infused over a 2-hour period at weeks 0, 2, and 6, and then every 8 weeks to week 30. The patients also received an antihistamine 30-60 minutes before the infusion, at the investigator’s discretion. Weekly methotrexate doses ranged from 12.5 mg to 25 mg. Patients also received 5 mg/week or more of folic acid.
Because the higher overall CRP level at baseline of the patients in the ATTRACT study could indicate that the PLANETRA cohort had less severe disease (3.1 mg/dL vs./ 1.9 mg/dL), Dr. Yoo and his associates conducted a post hoc sensitivity analysis of patients with baseline CRP greater than 2 mg/dL or 2 mg/dL or less. The results showed no difference in treatment response based on baseline CRP level.
The approximate 7.5% rate of infusion reactions noted in both in the CT-P13 and infliximab arms of the trial was lower than the 20% noted in the product label for infliximab, and antidrug antibody status did not appear to affect their incidence or response to the study drugs.
"Assessment of efficacy and safety of CT-P13 in patients with RA for up to 1 year is ongoing, and the positive results of this study provide a rationale for future studies of CT-P13 in the treatment of other TNF-mediated inflammatory diseases," the investigators wrote.
The study was sponsored by Celltrion. Many of the authors received research grants and/or consultancy fees and logistics support from the company. One author is a full-time employee of Celltrion, and another author declared receiving payment for lectures from the company.
The investigational biosimilar drug CT-P13 proved to have equivalent efficacy and safety comparable to that of infliximab, the tumor necrosis factor–alpha inhibitor that it is manufactured to mimic.
The findings are based on data from a randomized, double-blind, multicenter trial of 606 patients with active rheumatoid arthritis who had inadequate response to methotrexate alone.
Dr. Dae Hyun Yoo of Hanyang University Hospital for Rheumatic Diseases in Seoul, South Korea, reported no difference in the percentage of patients who met American College of Rheumatology 20% improvement criteria (ACR20) at 30 weeks (60.9% for CT-P13 vs. 58.6% for infliximab), which was the primary outcome of the study. The 95% confidence intervals for CT-P13 (–6%-10%) and infliximab (–4%-12%) were within the prespecified range of plus or minus 15% for equivalency (Ann. Rheum. Dis. 2013;72:1613-20).
The overall number of patients who reported treatment-emergent adverse events was virtually the same in patients who took CT-P13 (181) or infliximab (183). The adverse events that were deemed to be related to the study medications also occurred in a similar number of CT-P13 (106) and infliximab (108) patients. Most of these were mild to moderate in intensity. The trial’s safety objective was to determine whether CT-P13 had safety that was comparable, not equivalent, to that of infliximab.
Biosimilar drugs are meant to be "highly similar, but not identical and not ‘bioidentical,’ to approved ‘reference agents,’ " the authors wrote. CT-P13 is an immunoglobulin G1 chimeric human-murine monoclonal antibody to tumor necrosis factor–alpha (TNF-alpha) that has an amino acid sequence identical to that of infliximab. It is produced in the same type of cell line as the one used to produce infliximab.
In this study, known as PLANETRA (Program evaLuating the Autoimmune Disease iNvEstigational Drug cT-p13 in RA Patients), the investigators enrolled patients according to the same criteria as those used in several other clinical trials with infliximab, including the ATTRACT study for which infliximab was approved for the treatment of RA. The results obtained with CT-P13 were similar to those of other RA trials with infliximab that had the same enrollment criteria. The trial was conducted at 100 centers in 19 countries in Europe, Asia, Latin America, and the Middle East.
Patients in the trial met the revised 1987 ACR classification criteria for at least 1 year prior to screening, and were required to have at least six swollen and at least six tender joints and at least two of the following: morning stiffness lasting 45 or more minutes; serum C-reactive protein (CRP) level greater than 2.0 mg/dL; and an erythrocyte sedimentation rate of greater than 28 mm/hour. They needed to meet those criteria despite taking methotrexate for at least 3 months on a stable dose of 12.5-25 mg/week for 4 or more weeks prior to screening. Patients were permitted to receive an equivalent of 10 mg prednisolone or less per day and NSAIDs, if they had received a stable dose for at least 4 weeks prior to screening.
Approximately 82% of the patients were female, and the median age of patients was 50 years at baseline. The patients received either study drug at a dose of 3 mg/kg infused over a 2-hour period at weeks 0, 2, and 6, and then every 8 weeks to week 30. The patients also received an antihistamine 30-60 minutes before the infusion, at the investigator’s discretion. Weekly methotrexate doses ranged from 12.5 mg to 25 mg. Patients also received 5 mg/week or more of folic acid.
Because the higher overall CRP level at baseline of the patients in the ATTRACT study could indicate that the PLANETRA cohort had less severe disease (3.1 mg/dL vs./ 1.9 mg/dL), Dr. Yoo and his associates conducted a post hoc sensitivity analysis of patients with baseline CRP greater than 2 mg/dL or 2 mg/dL or less. The results showed no difference in treatment response based on baseline CRP level.
The approximate 7.5% rate of infusion reactions noted in both in the CT-P13 and infliximab arms of the trial was lower than the 20% noted in the product label for infliximab, and antidrug antibody status did not appear to affect their incidence or response to the study drugs.
"Assessment of efficacy and safety of CT-P13 in patients with RA for up to 1 year is ongoing, and the positive results of this study provide a rationale for future studies of CT-P13 in the treatment of other TNF-mediated inflammatory diseases," the investigators wrote.
The study was sponsored by Celltrion. Many of the authors received research grants and/or consultancy fees and logistics support from the company. One author is a full-time employee of Celltrion, and another author declared receiving payment for lectures from the company.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major finding: A similar percentage of patients met the study’s primary outcome: the criteria necessary for an American College of Rheumatology 20 level response at 30 weeks (60.9% for CT-P13 vs. 58.6% for infliximab).
Data source: A randomized, double-blind, multicenter trial of CT-P13 at 3mg/kg plus methotrexate vs. infliximab at 3 mg/kg plus methotrexate in 606 patients with active RA.
Disclosures: The study was sponsored by Celltrion. Many of the authors received research grants and/or consultancy fees and logistics support from the company. One author is a full-time employee of Celltrion, and another author declared receiving payment for lectures from the company.