User login
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
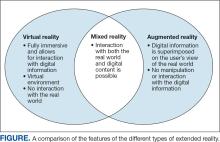
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30
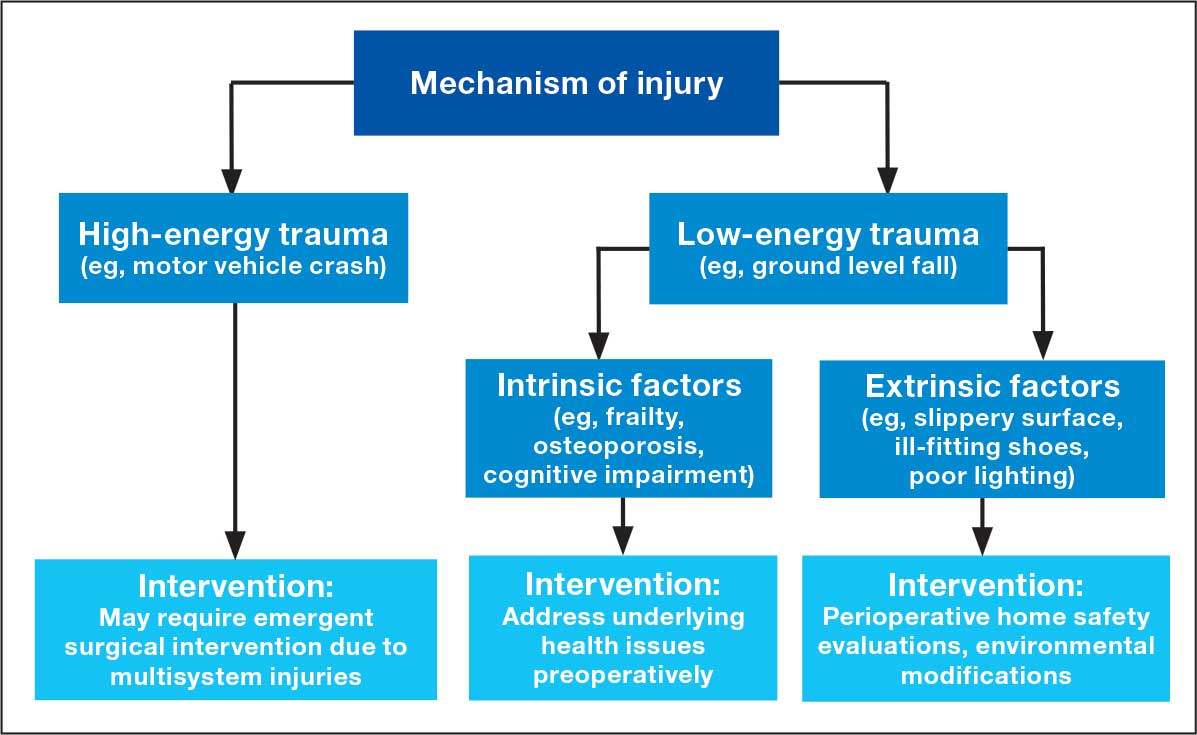
The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
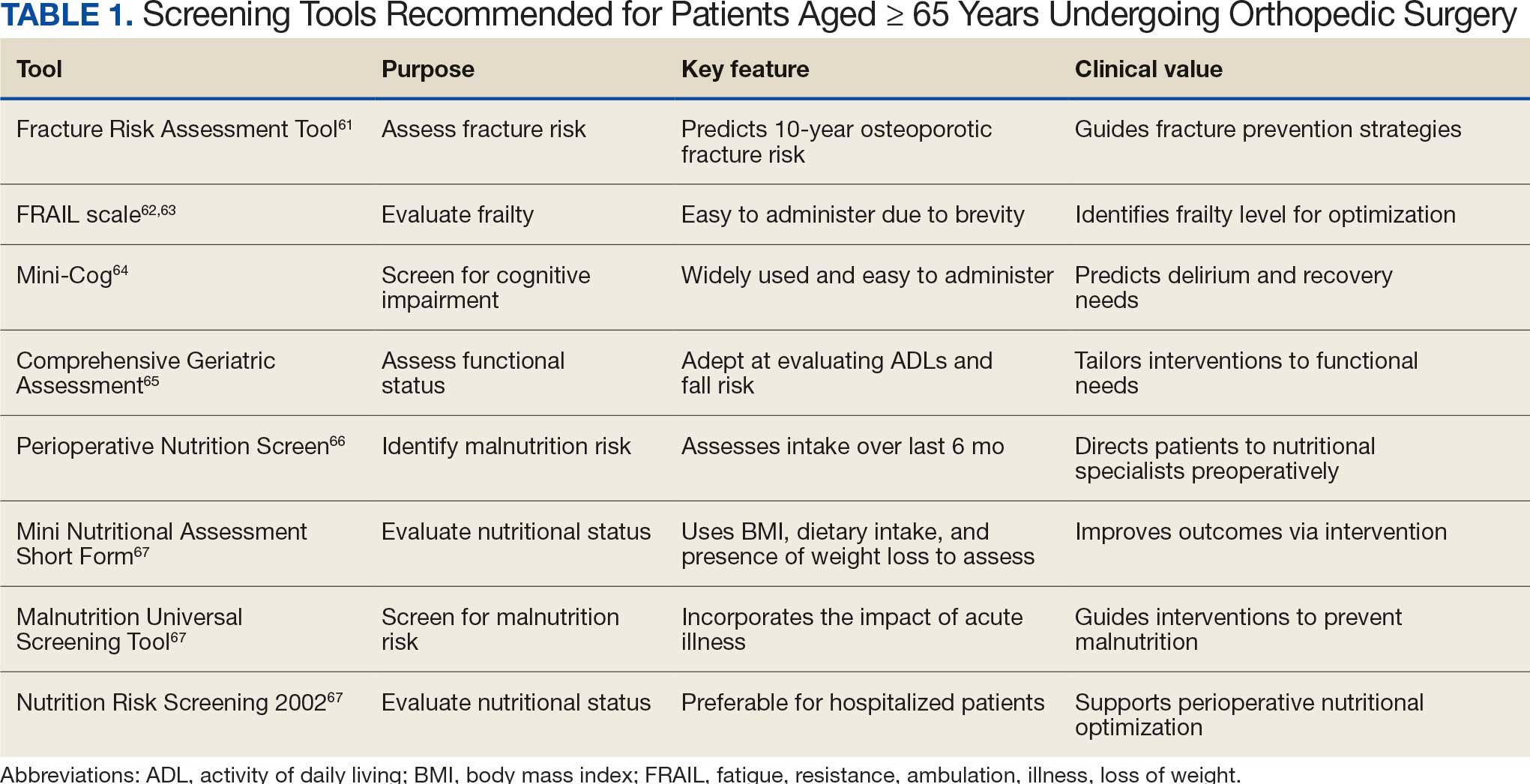
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.
Postoperative Complication Risks
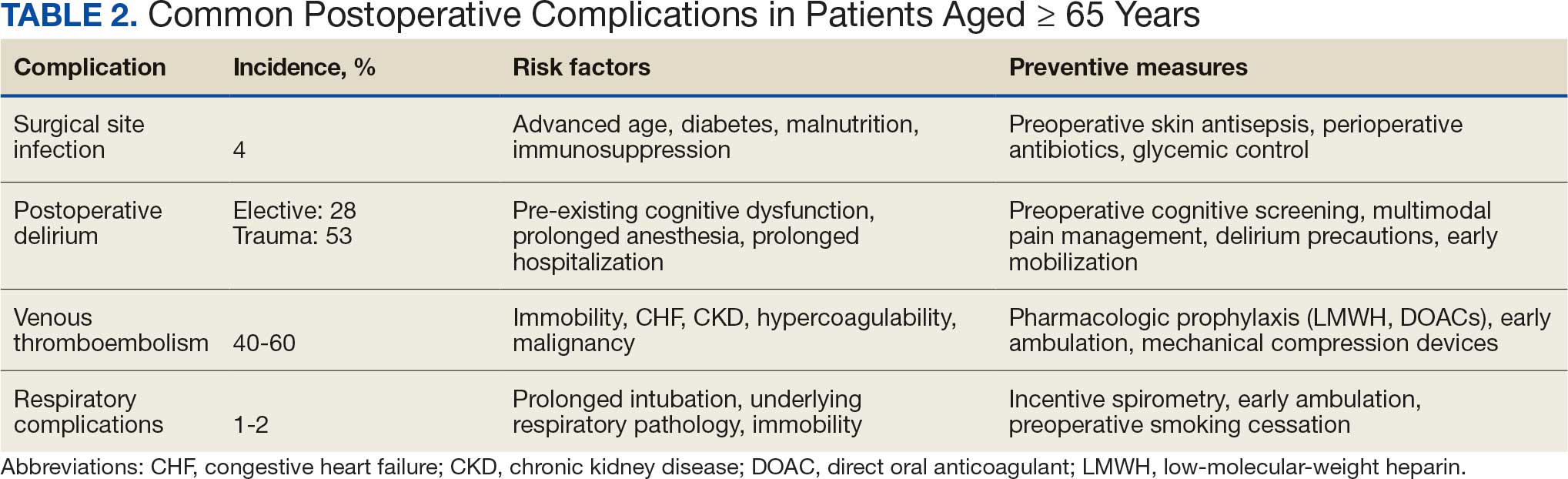
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Effects of Lumbar Fusion and Dual-Mobility Liners on Dislocation Rates Following Total Hip Arthroplasty in a Veteran Population
Effects of Lumbar Fusion and Dual-Mobility Liners on Dislocation Rates Following Total Hip Arthroplasty in a Veteran Population
Total hip arthroplasty (THA) is among the most common elective orthopedic procedures performed annually in the United States, with an estimated 635,000 to 909,000 THAs expected each year by 2030.1 Consequently, complication rates and revision surgeries related to THA have been increasing, along with the financial burden on the health care system.2-4 Optimizing outcomes for patients undergoing THA and identifying risk factors for treatment failure have become areas of focus.
Over the last decade, there has been a renewed interest in the effect of previous lumbar spine fusion (LSF) surgery on THA outcomes. Studies have explored the rates of complications, postoperative mobility, and THA implant impingement.5-8 However, the outcome receiving the most attention in recent literature is the rate and effect of dislocation in patients with lumbar fusion surgery. Large Medicare database analyses have discovered an association with increased rates of dislocations in patients with lumbar fusion surgeries compared with those without.9,10 Prosthetic hip dislocation is an expensive complication of THA and is projected to have greater impact through 2035 due to a growing number of THA procedures.11 Identifying risk factors associated with hip dislocation is paramount to mitigating its effect on patients who have undergone THA.
Recent research has found increased rates of THA dislocation and revision surgery in patients with LSF, with some studies showing previous LSF as the strongest independent predictor.6-16 However, controversy surrounds this relationship, including the sequence of procedures (LSF before or after THA), the time between procedures, and involvement of the sacrum in LSF. One study found that patients had a 106% increased risk of dislocation when LSF was performed before THA compared with patients who underwent LSF 5 years after undergoing THA, while another study showed no significant difference in dislocations pre- vs post-LSF.16,17 An additional study showed no significant difference in the rate of dislocation in patients without sacral involvement in the LSF, while also showing significantly higher rates of dislocation in LSF with sacral involvement.12 The researchers also found a trend toward more dislocations in longer lumbosacral fusions. Recent studies have also examined dislocation rates with lumbar fusion in patients treated with dual-mobility liners.18-20 The consensus from these studies is that dual-mobility liners significantly decrease the rate of dislocation in primary THAs with lumbar fusion.
The present study sought to determine the rates of hip dislocations in a US Department of Veterans Affairs (VA) hospital setting. To the authors’ knowledge, no retrospective study focusing on THAs in the veteran population has been performed. This study benefits from controlling for various surgeon techniques and surgical preferences when compared to large Medicare database studies because the orthopedic surgeon (ABK) only performed the posterior approach for all patients during the study period.
The primary objective of this study was to determine whether the rates of hip dislocation would, in fact, be higher in patients with lumbar fusion surgery, as recent database studies suggest. Secondary objectives included determining whether patient characteristics, comorbidities, number of levels fused, or inclusion of the sacrum in the fusion construct influenced dislocation rates. Furthermore, VA Dayton Healthcare System (VADHS) began routine use of dual-mobility liners for lumbar fusion patients in 2018, allowing for examination of these patients.
Methods
The Wright State University and VADHS Institutional Review Board approved this study design. A retrospective review of all primary THAs at VADHS was performed to investigate the relationship between previous lumbar spine fusion and the incidence of THA revision. Manual chart review was performed for patients who underwent primary THA between January 2003, and December 2022. One surgeon performed all surgeries using only the posterior approach. Patients were not excluded if they had bilateral procedures and all eligible hips were included. Patients with a concomitant diagnosis of fracture of the femoral head or femoral neck at the time of surgery were excluded. Additionally, only patients with ≥ 12 months of follow-up data were included.
The primary outcome was dislocation within 12 months of THA; the primary independent variable was LSF prior to THA. Covariates included patient demographics (age, sex, body mass index [BMI]) and Charlson Comorbidity Index (CCI) score, with additional data collected on the number of levels fused, sacral spine involvement, revision rates, and use of dual-mobility liners. Year of surgery was also included in analyses to account for any changes that may have occurred during the study period.
Statistical Analysis
Statistical analyses were performed in SAS 9.4. Patients were grouped into 2 cohorts, depending on whether they had received LSF prior to THA. Analyses were adjusted for repeated measures to account for the small percentage of patients with bilateral procedures.
Univariate comparisons between cohorts for covariates, as well as rates of dislocation and revision, were performed using the independent samples t test for continuous variables and the Fisher exact test for dichotomous categorical variables. Significant comorbidities, as well as age, sex, BMI, liner type, LSF cohort, and surgery year, were included in a logistic regression model to determine what effect, if any, they had on the likelihood of dislocation. Variables were removed using a backward stepwise approach, starting with the nonsignificant variable effect with the lowest χ2 value, and continuing until reaching a final model where all remaining variable effects were significant. For the variables retained in the final model, odds ratios (ORs) with 95% CIs were derived, with dislocation designated as the event. Individual comorbidity subcomponents of the CCI were also analyzed for their effects on dislocation using backward stepwise logistic regression. A secondary analysis among patients with LSF tested for the influence of the number of vertebral levels fused, the presence or absence of sacral involvement in the fusion, and the use of dual-mobility liners on the likelihood of hip dislocation.
Results
The LSF cohort included 39 patients with THA and prior LSF, 3 of whom had bilateral procedures, for a total of 42 hips. The non-LSF cohort included 813 patients with THA, 112 of whom had bilateral procedures, for a total of 925 hips. The LSF and non-LSF cohorts did not differ significantly in age, sex, BMI, CCI, or revision rates (Table). The LSF cohort included a significantly higher percentage of hips receiving dual-mobility liners than did the non-LSF cohort (23.8% vs 0.6%; P < .001) and had more than twice the rate of dislocation (4 of 42 hips [9.5%] vs 35 of 925 hips [3.8%]), although this difference was not statistically significant (P = .08).
The final logistic regression model with dislocation as the outcome was statistically significant (χ2, 17.47; P < .001) and retained 2 significant predictor variables: LSF cohort (χ2, 4.63; P = .03), and sex (χ2, 18.27; P < .001). Females were more likely than males to experience dislocation (OR, 5.84; 95% CI, 2.60-13.13; P < .001) as were patients who had LSF prior to THA (OR, 3.42; 95% CI, 1.12-10.47; P = .03) (Figure). None of the CCI subcomponent comorbidities significantly affected the probability of dislocation (myocardial infarction, P = .46; congestive heart failure, P = .47; peripheral vascular disease, P = .97; stroke, P = .51; dementia, P = .99; chronic obstructive pulmonary disease, P = .95; connective tissue disease, P = .25; peptic ulcer, P = .41; liver disease, P = .30; diabetes, P = .06; hemiplegia, P = .99; chronic kidney disease, P = .82; solid tumor, P = .90; leukemia, P = .99; lymphoma, P = .99; AIDS, P = .99). Within the LSF cohort, neither the number of levels fused (P = .83) nor sacral involvement (P = .42), significantly affected the probability of hip dislocation. None of the patients in either cohort who received dual-mobility liners subsequently dislocated their hips, nor did any of them require revision surgery.
Discussion
Spinopelvic biomechanics have been an area of increasing interest and research. Spinal fusion has been shown to alter the mobility of the pelvis and has been associated with decreased stability of THA implants.21 For example, in the setting of a fused spine, the lack of compensatory changes in pelvic tilt or acetabular anteversion when adjusting to a seated or standing position may predispose patients to impingement because the acetabular component is not properly positioned. Dual-mobility constructs mitigate this risk by providing an additional articulation, which increases jump distance and range of motion prior to impingement, thereby enhancing stability.
The use of dual-mobility liners in patients with LSF has also been examined.18-20 These studies demonstrate a reduced risk of postoperative THA dislocation in patients with previous LSF. The rate of postoperative complications and revisions for LSF patients with dual-mobility liners was also found to be similar to that of THAs without dual-mobility in patients without prior LSF. This study focused on a veteran population to demonstrate the efficacy of dual-mobility liners in patients with LSF. The results indicate that LSF prior to THA and female sex were predictors for prosthetic hip dislocations in the 12-month postoperative period in this patient population, which aligns with the current literature.
The dislocation rate in the LSF-THA group (9.5%) was higher than the dislocation rate in the control group (3.8%). Although not statistically significant in the univariate analysis, LSF was shown to be a significant risk factor after controlling for patient sex. Other studies have found the dislocation rate to be 3% to 7%, which is lower than the dislocation rate observed in this study.8,10,16
The reasons for this higher rate of dislocation are not entirely clear. A veteran population has poorer overall health than the general population, which may contribute to the higher than previously reported dislocation rates.22 These results can be applied to the management of veterans seeking THA.
There have been conflicting reports regarding the impact a patient’s sex has on THA outcomes in the general population.23-26 This study found that female patients had higher rates of dislocation within 1 year of THA than male patients. This difference, which could be due to differences in baseline anatomic hip morphology between the sexes; females tend to have smaller femoral head sizes and less offset compared with males.27,28 However, this finding could have been confounded by the small number of female veterans in the study cohort.
A type 2 diabetes mellitus (T2DM) diagnosis, which is a component of CCI, trended toward increased risk of prosthetic hip dislocation. Multiple studies have also discussed the increased risk of postoperative infections and revisions following THA in patients with T2DM.29-31 One study found T2DM to be an independent risk factor for immediate in-hospital postoperative complications following hip arthroplasty.32
Another factor that may influence postoperative dislocation risk is surgical approach. The posterior approach has historically been associated with higher rates of instability when compared to anterior or lateral THA.33 Researchers have also looked at the role that surgical approach plays in patients with prior LSF. Huebschmann et al confirmed that not only is LSF a significant risk factor for dislocation following THA, but anterior and laterally based surgical approaches may mitigate this risk.34
Limitations
As a retrospective cohort study, the reliability of the data hinges on complete documentation. Documentation of all encounters for dislocations was obtained from the VA Computerized Patient Record System, which may have led to some dislocation events being missed. However, as long as there was adequate postoperative follow-up, it was assumed all events outside the VA were included. Another limitation of this study was that male patients greatly outnumbered female patients, and this fact could limit the generalizability of findings to the population as a whole.
Conclusions
This study in a veteran population found that prior LSF and female sex were significant predictors for postoperative dislocation within 1 year of THA surgery. Additionally, the use of a dual-mobility liner was found to be protective against postoperative dislocation events. These data allow clinicians to better counsel veterans on the risk factors associated with postoperative dislocation and strategies to mitigate this risk.
- Sloan M, Premkumar A, Sheth NP. Projected volume of primary total joint arthroplasty in the U.S., 2014 to 2030. J Bone Joint Surg Am. 2018;100:1455-1460. doi:10.2106/JBJS.17.01617
- Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91:128-133. doi:10.2106/JBJS.H.00155
- Kurtz SM, Ong KL, Schmier J, et al. Future clinical and economic impact of revision total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89:144-151. doi:10.2106/JBJS.G.00587
- Kurtz SM, Ong KL, Schmier J, et al. Primary and revision arthroplasty surgery caseloads in the United States from 1990 to 2004. J Arthroplasty. 2009;24:195-203. doi:10.1016/j.arth.2007.11.015
- Yamato Y, Furuhashi H, Hasegawa T, et al. Simulation of implant impingement after spinal corrective fusion surgery in patients with previous total hip arthroplasty: a retrospective case series. Spine (Phila Pa 1976). 2021;46:512-519. doi:10.1097/BRS.0000000000003836
- Mudrick CA, Melvin JS, Springer BD. Late posterior hip instability after lumbar spinopelvic fusion. Arthroplast Today. 2015;1:25-29. doi:10.1016/j.artd.2015.05.002
- Diebo BG, Beyer GA, Grieco PW, et al. Complications in patients undergoing spinal fusion after THA. Clin Orthop Relat Res. 2018;476:412-417.doi:10.1007/s11999.0000000000000009 8.
- Sing DC, Barry JJ, Aguilar TU, et al. Prior lumbar spinal arthrodesis increases risk of prosthetic-related complication in total hip arthroplasty. J Arthroplasty. 2016;31:227-232.e1. doi:10.1016/j.arth.2016.02.069
- King CA, Landy DC, Martell JM, et al. Time to dislocation analysis of lumbar spine fusion following total hip arthroplasty: breaking up a happy home. J Arthroplasty. 2018;33:3768-3772. doi:10.1016/j.arth.2018.08.029
- Buckland AJ, Puvanesarajah V, Vigdorchik J, et al. Dislocation of a primary total hip arthroplasty is more common in patients with a lumbar spinal fusion. Bone Joint J. 2017;99-B:585-591.doi:10.1302/0301-620X.99B5.BJJ-2016-0657.R1
- Pirruccio K, Premkumar A, Sheth NP. The burden of prosthetic hip dislocations in the United States is projected to significantly increase by 2035. Hip Int. 2021;31:714-721. doi:10.1177/1120700020923619
- Salib CG, Reina N, Perry KI, et al. Lumbar fusion involving the sacrum increases dislocation risk in primary total hip arthroplasty. Bone Joint J. 2019;101-B:198-206. doi:10.1302/0301-620X.101B2.BJJ-2018-0754.R1
- An VVG, Phan K, Sivakumar BS, et al. Prior lumbar spinal fusion is associated with an increased risk of dislocation and revision in total hip arthroplasty: a meta-analysis. J Arthroplasty. 2018;33:297-300. doi:10.1016/j.arth.2017.08.040
- Klemt C, Padmanabha A, Tirumala V, et al. Lumbar spine fusion before revision total hip arthroplasty is associated with increased dislocation rates. J Am Acad Orthop Surg. 2021;29:e860-e868. doi:10.5435/JAAOS-D-20-00824
- Gausden EB, Parhar HS, Popper JE, et al. Risk factors for early dislocation following primary elective total hip arthroplasty. J Arthroplasty. 2018;33:1567-1571. doi:10.1016/j.arth.2017.12.034
- Malkani AL, Himschoot KJ, Ong KL, et al. Does timing of primary total hip arthroplasty prior to or after lumbar spine fusion have an effect on dislocation and revision rates?. J Arthroplasty. 2019;34:907-911. doi:10.1016/j.arth.2019.01.009
- Parilla FW, Shah RR, Gordon AC, et al. Does it matter: total hip arthroplasty or lumbar spinal fusion first? Preoperative sagittal spinopelvic measurements guide patient-specific surgical strategies in patients requiring both. J Arthroplasty. 2019;34:2652-2662. doi:10.1016/j.arth.2019.05.053
- Chalmers BP, Syku M, Sculco TP, et al. Dual-mobility constructs in primary total hip arthroplasty in high-risk patients with spinal fusions: our institutional experience. Arthroplast Today. 2020;6:749-754. doi:10.1016/j.artd.2020.07.024
- Nessler JM, Malkani AL, Sachdeva S, et al. Use of dual mobility cups in patients undergoing primary total hip arthroplasty with prior lumbar spine fusion. Int Orthop. 2020;44:857-862. doi:10.1007/s00264-020-04507-y
- Nessler JM, Malkani AL, Yep PJ, et al. Dislocation rates of primary total hip arthroplasty in patients with prior lumbar spine fusion and lumbar degenerative disk disease with and without utilization of dual mobility cups: an American Joint Replacement Registry study. J Am Acad Orthop Surg. 2023;31:e271-e277. doi:10.5435/JAAOS-D-22-00767
- Phan D, Bederman SS, Schwarzkopf R. The influence of sagittal spinal deformity on anteversion of the acetabular component in total hip arthroplasty. Bone Joint J. 2015;97-B:1017-1023. doi:10.1302/0301-620X.97B8.35700
- Agha Z, Lofgren RP, VanRuiswyk JV, et al. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160:3252-3257. doi:10.1001/archinte.160.21.325223.
- Basques BA, Bell JA, Fillingham YA, et al. Gender differences for hip and knee arthroplasty: complications and healthcare utilization. J Arthroplasty. 2019;34:1593-1597.e1. doi:10.1016/j.arth.2019.03.064
- Kim YH, Choi Y, Kim JS. Influence of patient-, design-, and surgery-related factors on rate of dislocation after primary cementless total hip arthroplasty. J Arthroplasty. 2009;24:1258-1263. doi:10.1016/j.arth.2009.03.017
- Chen A, Paxton L, Zheng X, et al. Association of sex with risk of 2-year revision among patients undergoing total hip arthroplasty. JAMA Netw Open. 2021;4:e2110687. doi:10.1001/jamanetworkopen.2021.10687
- Inacio MCS, Ake CF, Paxton EW, et al. Sex and risk of hip implant failure: assessing total hip arthroplasty outcomes in the United States. JAMA Intern Med. 2013;173:435-441. doi:10.1001/jamainternmed.2013.3271
- Karlson EW, Daltroy LH, Liang MH, et al. Gender differences in patient preferences may underlie differential utilization of elective surgery. Am J Med. 1997;102:524-530. doi:10.1016/s0002-9343(97)00050-8
- Kostamo T, Bourne RB, Whittaker JP, et al. No difference in gender-specific hip replacement outcomes. Clin Orthop Relat Res. 2009;467:135-140. doi:10.1007/s11999-008-0466-2
- Papagelopoulos PJ, Idusuyi OB, Wallrichs SL, et al. Long term outcome and survivorship analysis of primary total knee arthroplasty in patients with diabetes mellitus. Clin Orthop Relat Res. 1996;(330):124-132. doi:10.1097/00003086-199609000-00015
- Fitzgerald RH Jr, Nolan DR, Ilstrup DM, et al. Deep wound sepsis following total hip arthroplasty. J Bone Joint Surg Am. 1977;59:847-855.
- Blom AW, Brown J, Taylor AH, et al. Infection after total knee arthroplasty. J Bone Joint Surg Br. 2004;86:688-691. doi:10.1302/0301-620x.86b5.14887
- Jain NB, Guller U, Pietrobon R, et al. Comorbidities increase complication rates in patients having arthroplasty. Clin Orthop Relat Res. 2005;435:232-238. doi:10.1097/01.blo.0000156479.97488.a2
- Docter S, Philpott HT, Godkin L, et al. Comparison of intra and post-operative complication rates among surgical approaches in Total Hip Arthroplasty: A systematic review and meta-analysis. J Orthop. 2020;20:310-325. doi:10.1016/j.jor.2020.05.008
- Huebschmann NA, Lawrence KW, Robin JX, et al. Does surgical approach affect dislocation rate after total hip arthroplasty in patients who have prior lumbar spinal fusion? A retrospective analysis of 16,223 cases. J Arthroplasty. 2024;39:S306-S313. doi:10.1016/j.arth.2024.03.068
Total hip arthroplasty (THA) is among the most common elective orthopedic procedures performed annually in the United States, with an estimated 635,000 to 909,000 THAs expected each year by 2030.1 Consequently, complication rates and revision surgeries related to THA have been increasing, along with the financial burden on the health care system.2-4 Optimizing outcomes for patients undergoing THA and identifying risk factors for treatment failure have become areas of focus.
Over the last decade, there has been a renewed interest in the effect of previous lumbar spine fusion (LSF) surgery on THA outcomes. Studies have explored the rates of complications, postoperative mobility, and THA implant impingement.5-8 However, the outcome receiving the most attention in recent literature is the rate and effect of dislocation in patients with lumbar fusion surgery. Large Medicare database analyses have discovered an association with increased rates of dislocations in patients with lumbar fusion surgeries compared with those without.9,10 Prosthetic hip dislocation is an expensive complication of THA and is projected to have greater impact through 2035 due to a growing number of THA procedures.11 Identifying risk factors associated with hip dislocation is paramount to mitigating its effect on patients who have undergone THA.
Recent research has found increased rates of THA dislocation and revision surgery in patients with LSF, with some studies showing previous LSF as the strongest independent predictor.6-16 However, controversy surrounds this relationship, including the sequence of procedures (LSF before or after THA), the time between procedures, and involvement of the sacrum in LSF. One study found that patients had a 106% increased risk of dislocation when LSF was performed before THA compared with patients who underwent LSF 5 years after undergoing THA, while another study showed no significant difference in dislocations pre- vs post-LSF.16,17 An additional study showed no significant difference in the rate of dislocation in patients without sacral involvement in the LSF, while also showing significantly higher rates of dislocation in LSF with sacral involvement.12 The researchers also found a trend toward more dislocations in longer lumbosacral fusions. Recent studies have also examined dislocation rates with lumbar fusion in patients treated with dual-mobility liners.18-20 The consensus from these studies is that dual-mobility liners significantly decrease the rate of dislocation in primary THAs with lumbar fusion.
The present study sought to determine the rates of hip dislocations in a US Department of Veterans Affairs (VA) hospital setting. To the authors’ knowledge, no retrospective study focusing on THAs in the veteran population has been performed. This study benefits from controlling for various surgeon techniques and surgical preferences when compared to large Medicare database studies because the orthopedic surgeon (ABK) only performed the posterior approach for all patients during the study period.
The primary objective of this study was to determine whether the rates of hip dislocation would, in fact, be higher in patients with lumbar fusion surgery, as recent database studies suggest. Secondary objectives included determining whether patient characteristics, comorbidities, number of levels fused, or inclusion of the sacrum in the fusion construct influenced dislocation rates. Furthermore, VA Dayton Healthcare System (VADHS) began routine use of dual-mobility liners for lumbar fusion patients in 2018, allowing for examination of these patients.
Methods
The Wright State University and VADHS Institutional Review Board approved this study design. A retrospective review of all primary THAs at VADHS was performed to investigate the relationship between previous lumbar spine fusion and the incidence of THA revision. Manual chart review was performed for patients who underwent primary THA between January 2003, and December 2022. One surgeon performed all surgeries using only the posterior approach. Patients were not excluded if they had bilateral procedures and all eligible hips were included. Patients with a concomitant diagnosis of fracture of the femoral head or femoral neck at the time of surgery were excluded. Additionally, only patients with ≥ 12 months of follow-up data were included.
The primary outcome was dislocation within 12 months of THA; the primary independent variable was LSF prior to THA. Covariates included patient demographics (age, sex, body mass index [BMI]) and Charlson Comorbidity Index (CCI) score, with additional data collected on the number of levels fused, sacral spine involvement, revision rates, and use of dual-mobility liners. Year of surgery was also included in analyses to account for any changes that may have occurred during the study period.
Statistical Analysis
Statistical analyses were performed in SAS 9.4. Patients were grouped into 2 cohorts, depending on whether they had received LSF prior to THA. Analyses were adjusted for repeated measures to account for the small percentage of patients with bilateral procedures.
Univariate comparisons between cohorts for covariates, as well as rates of dislocation and revision, were performed using the independent samples t test for continuous variables and the Fisher exact test for dichotomous categorical variables. Significant comorbidities, as well as age, sex, BMI, liner type, LSF cohort, and surgery year, were included in a logistic regression model to determine what effect, if any, they had on the likelihood of dislocation. Variables were removed using a backward stepwise approach, starting with the nonsignificant variable effect with the lowest χ2 value, and continuing until reaching a final model where all remaining variable effects were significant. For the variables retained in the final model, odds ratios (ORs) with 95% CIs were derived, with dislocation designated as the event. Individual comorbidity subcomponents of the CCI were also analyzed for their effects on dislocation using backward stepwise logistic regression. A secondary analysis among patients with LSF tested for the influence of the number of vertebral levels fused, the presence or absence of sacral involvement in the fusion, and the use of dual-mobility liners on the likelihood of hip dislocation.
Results
The LSF cohort included 39 patients with THA and prior LSF, 3 of whom had bilateral procedures, for a total of 42 hips. The non-LSF cohort included 813 patients with THA, 112 of whom had bilateral procedures, for a total of 925 hips. The LSF and non-LSF cohorts did not differ significantly in age, sex, BMI, CCI, or revision rates (Table). The LSF cohort included a significantly higher percentage of hips receiving dual-mobility liners than did the non-LSF cohort (23.8% vs 0.6%; P < .001) and had more than twice the rate of dislocation (4 of 42 hips [9.5%] vs 35 of 925 hips [3.8%]), although this difference was not statistically significant (P = .08).

The final logistic regression model with dislocation as the outcome was statistically significant (χ2, 17.47; P < .001) and retained 2 significant predictor variables: LSF cohort (χ2, 4.63; P = .03), and sex (χ2, 18.27; P < .001). Females were more likely than males to experience dislocation (OR, 5.84; 95% CI, 2.60-13.13; P < .001) as were patients who had LSF prior to THA (OR, 3.42; 95% CI, 1.12-10.47; P = .03) (Figure). None of the CCI subcomponent comorbidities significantly affected the probability of dislocation (myocardial infarction, P = .46; congestive heart failure, P = .47; peripheral vascular disease, P = .97; stroke, P = .51; dementia, P = .99; chronic obstructive pulmonary disease, P = .95; connective tissue disease, P = .25; peptic ulcer, P = .41; liver disease, P = .30; diabetes, P = .06; hemiplegia, P = .99; chronic kidney disease, P = .82; solid tumor, P = .90; leukemia, P = .99; lymphoma, P = .99; AIDS, P = .99). Within the LSF cohort, neither the number of levels fused (P = .83) nor sacral involvement (P = .42), significantly affected the probability of hip dislocation. None of the patients in either cohort who received dual-mobility liners subsequently dislocated their hips, nor did any of them require revision surgery.

Discussion
Spinopelvic biomechanics have been an area of increasing interest and research. Spinal fusion has been shown to alter the mobility of the pelvis and has been associated with decreased stability of THA implants.21 For example, in the setting of a fused spine, the lack of compensatory changes in pelvic tilt or acetabular anteversion when adjusting to a seated or standing position may predispose patients to impingement because the acetabular component is not properly positioned. Dual-mobility constructs mitigate this risk by providing an additional articulation, which increases jump distance and range of motion prior to impingement, thereby enhancing stability.
The use of dual-mobility liners in patients with LSF has also been examined.18-20 These studies demonstrate a reduced risk of postoperative THA dislocation in patients with previous LSF. The rate of postoperative complications and revisions for LSF patients with dual-mobility liners was also found to be similar to that of THAs without dual-mobility in patients without prior LSF. This study focused on a veteran population to demonstrate the efficacy of dual-mobility liners in patients with LSF. The results indicate that LSF prior to THA and female sex were predictors for prosthetic hip dislocations in the 12-month postoperative period in this patient population, which aligns with the current literature.
The dislocation rate in the LSF-THA group (9.5%) was higher than the dislocation rate in the control group (3.8%). Although not statistically significant in the univariate analysis, LSF was shown to be a significant risk factor after controlling for patient sex. Other studies have found the dislocation rate to be 3% to 7%, which is lower than the dislocation rate observed in this study.8,10,16
The reasons for this higher rate of dislocation are not entirely clear. A veteran population has poorer overall health than the general population, which may contribute to the higher than previously reported dislocation rates.22 These results can be applied to the management of veterans seeking THA.
There have been conflicting reports regarding the impact a patient’s sex has on THA outcomes in the general population.23-26 This study found that female patients had higher rates of dislocation within 1 year of THA than male patients. This difference, which could be due to differences in baseline anatomic hip morphology between the sexes; females tend to have smaller femoral head sizes and less offset compared with males.27,28 However, this finding could have been confounded by the small number of female veterans in the study cohort.
A type 2 diabetes mellitus (T2DM) diagnosis, which is a component of CCI, trended toward increased risk of prosthetic hip dislocation. Multiple studies have also discussed the increased risk of postoperative infections and revisions following THA in patients with T2DM.29-31 One study found T2DM to be an independent risk factor for immediate in-hospital postoperative complications following hip arthroplasty.32
Another factor that may influence postoperative dislocation risk is surgical approach. The posterior approach has historically been associated with higher rates of instability when compared to anterior or lateral THA.33 Researchers have also looked at the role that surgical approach plays in patients with prior LSF. Huebschmann et al confirmed that not only is LSF a significant risk factor for dislocation following THA, but anterior and laterally based surgical approaches may mitigate this risk.34
Limitations
As a retrospective cohort study, the reliability of the data hinges on complete documentation. Documentation of all encounters for dislocations was obtained from the VA Computerized Patient Record System, which may have led to some dislocation events being missed. However, as long as there was adequate postoperative follow-up, it was assumed all events outside the VA were included. Another limitation of this study was that male patients greatly outnumbered female patients, and this fact could limit the generalizability of findings to the population as a whole.
Conclusions
This study in a veteran population found that prior LSF and female sex were significant predictors for postoperative dislocation within 1 year of THA surgery. Additionally, the use of a dual-mobility liner was found to be protective against postoperative dislocation events. These data allow clinicians to better counsel veterans on the risk factors associated with postoperative dislocation and strategies to mitigate this risk.
Total hip arthroplasty (THA) is among the most common elective orthopedic procedures performed annually in the United States, with an estimated 635,000 to 909,000 THAs expected each year by 2030.1 Consequently, complication rates and revision surgeries related to THA have been increasing, along with the financial burden on the health care system.2-4 Optimizing outcomes for patients undergoing THA and identifying risk factors for treatment failure have become areas of focus.
Over the last decade, there has been a renewed interest in the effect of previous lumbar spine fusion (LSF) surgery on THA outcomes. Studies have explored the rates of complications, postoperative mobility, and THA implant impingement.5-8 However, the outcome receiving the most attention in recent literature is the rate and effect of dislocation in patients with lumbar fusion surgery. Large Medicare database analyses have discovered an association with increased rates of dislocations in patients with lumbar fusion surgeries compared with those without.9,10 Prosthetic hip dislocation is an expensive complication of THA and is projected to have greater impact through 2035 due to a growing number of THA procedures.11 Identifying risk factors associated with hip dislocation is paramount to mitigating its effect on patients who have undergone THA.
Recent research has found increased rates of THA dislocation and revision surgery in patients with LSF, with some studies showing previous LSF as the strongest independent predictor.6-16 However, controversy surrounds this relationship, including the sequence of procedures (LSF before or after THA), the time between procedures, and involvement of the sacrum in LSF. One study found that patients had a 106% increased risk of dislocation when LSF was performed before THA compared with patients who underwent LSF 5 years after undergoing THA, while another study showed no significant difference in dislocations pre- vs post-LSF.16,17 An additional study showed no significant difference in the rate of dislocation in patients without sacral involvement in the LSF, while also showing significantly higher rates of dislocation in LSF with sacral involvement.12 The researchers also found a trend toward more dislocations in longer lumbosacral fusions. Recent studies have also examined dislocation rates with lumbar fusion in patients treated with dual-mobility liners.18-20 The consensus from these studies is that dual-mobility liners significantly decrease the rate of dislocation in primary THAs with lumbar fusion.
The present study sought to determine the rates of hip dislocations in a US Department of Veterans Affairs (VA) hospital setting. To the authors’ knowledge, no retrospective study focusing on THAs in the veteran population has been performed. This study benefits from controlling for various surgeon techniques and surgical preferences when compared to large Medicare database studies because the orthopedic surgeon (ABK) only performed the posterior approach for all patients during the study period.
The primary objective of this study was to determine whether the rates of hip dislocation would, in fact, be higher in patients with lumbar fusion surgery, as recent database studies suggest. Secondary objectives included determining whether patient characteristics, comorbidities, number of levels fused, or inclusion of the sacrum in the fusion construct influenced dislocation rates. Furthermore, VA Dayton Healthcare System (VADHS) began routine use of dual-mobility liners for lumbar fusion patients in 2018, allowing for examination of these patients.
Methods
The Wright State University and VADHS Institutional Review Board approved this study design. A retrospective review of all primary THAs at VADHS was performed to investigate the relationship between previous lumbar spine fusion and the incidence of THA revision. Manual chart review was performed for patients who underwent primary THA between January 2003, and December 2022. One surgeon performed all surgeries using only the posterior approach. Patients were not excluded if they had bilateral procedures and all eligible hips were included. Patients with a concomitant diagnosis of fracture of the femoral head or femoral neck at the time of surgery were excluded. Additionally, only patients with ≥ 12 months of follow-up data were included.
The primary outcome was dislocation within 12 months of THA; the primary independent variable was LSF prior to THA. Covariates included patient demographics (age, sex, body mass index [BMI]) and Charlson Comorbidity Index (CCI) score, with additional data collected on the number of levels fused, sacral spine involvement, revision rates, and use of dual-mobility liners. Year of surgery was also included in analyses to account for any changes that may have occurred during the study period.
Statistical Analysis
Statistical analyses were performed in SAS 9.4. Patients were grouped into 2 cohorts, depending on whether they had received LSF prior to THA. Analyses were adjusted for repeated measures to account for the small percentage of patients with bilateral procedures.
Univariate comparisons between cohorts for covariates, as well as rates of dislocation and revision, were performed using the independent samples t test for continuous variables and the Fisher exact test for dichotomous categorical variables. Significant comorbidities, as well as age, sex, BMI, liner type, LSF cohort, and surgery year, were included in a logistic regression model to determine what effect, if any, they had on the likelihood of dislocation. Variables were removed using a backward stepwise approach, starting with the nonsignificant variable effect with the lowest χ2 value, and continuing until reaching a final model where all remaining variable effects were significant. For the variables retained in the final model, odds ratios (ORs) with 95% CIs were derived, with dislocation designated as the event. Individual comorbidity subcomponents of the CCI were also analyzed for their effects on dislocation using backward stepwise logistic regression. A secondary analysis among patients with LSF tested for the influence of the number of vertebral levels fused, the presence or absence of sacral involvement in the fusion, and the use of dual-mobility liners on the likelihood of hip dislocation.
Results
The LSF cohort included 39 patients with THA and prior LSF, 3 of whom had bilateral procedures, for a total of 42 hips. The non-LSF cohort included 813 patients with THA, 112 of whom had bilateral procedures, for a total of 925 hips. The LSF and non-LSF cohorts did not differ significantly in age, sex, BMI, CCI, or revision rates (Table). The LSF cohort included a significantly higher percentage of hips receiving dual-mobility liners than did the non-LSF cohort (23.8% vs 0.6%; P < .001) and had more than twice the rate of dislocation (4 of 42 hips [9.5%] vs 35 of 925 hips [3.8%]), although this difference was not statistically significant (P = .08).

The final logistic regression model with dislocation as the outcome was statistically significant (χ2, 17.47; P < .001) and retained 2 significant predictor variables: LSF cohort (χ2, 4.63; P = .03), and sex (χ2, 18.27; P < .001). Females were more likely than males to experience dislocation (OR, 5.84; 95% CI, 2.60-13.13; P < .001) as were patients who had LSF prior to THA (OR, 3.42; 95% CI, 1.12-10.47; P = .03) (Figure). None of the CCI subcomponent comorbidities significantly affected the probability of dislocation (myocardial infarction, P = .46; congestive heart failure, P = .47; peripheral vascular disease, P = .97; stroke, P = .51; dementia, P = .99; chronic obstructive pulmonary disease, P = .95; connective tissue disease, P = .25; peptic ulcer, P = .41; liver disease, P = .30; diabetes, P = .06; hemiplegia, P = .99; chronic kidney disease, P = .82; solid tumor, P = .90; leukemia, P = .99; lymphoma, P = .99; AIDS, P = .99). Within the LSF cohort, neither the number of levels fused (P = .83) nor sacral involvement (P = .42), significantly affected the probability of hip dislocation. None of the patients in either cohort who received dual-mobility liners subsequently dislocated their hips, nor did any of them require revision surgery.

Discussion
Spinopelvic biomechanics have been an area of increasing interest and research. Spinal fusion has been shown to alter the mobility of the pelvis and has been associated with decreased stability of THA implants.21 For example, in the setting of a fused spine, the lack of compensatory changes in pelvic tilt or acetabular anteversion when adjusting to a seated or standing position may predispose patients to impingement because the acetabular component is not properly positioned. Dual-mobility constructs mitigate this risk by providing an additional articulation, which increases jump distance and range of motion prior to impingement, thereby enhancing stability.
The use of dual-mobility liners in patients with LSF has also been examined.18-20 These studies demonstrate a reduced risk of postoperative THA dislocation in patients with previous LSF. The rate of postoperative complications and revisions for LSF patients with dual-mobility liners was also found to be similar to that of THAs without dual-mobility in patients without prior LSF. This study focused on a veteran population to demonstrate the efficacy of dual-mobility liners in patients with LSF. The results indicate that LSF prior to THA and female sex were predictors for prosthetic hip dislocations in the 12-month postoperative period in this patient population, which aligns with the current literature.
The dislocation rate in the LSF-THA group (9.5%) was higher than the dislocation rate in the control group (3.8%). Although not statistically significant in the univariate analysis, LSF was shown to be a significant risk factor after controlling for patient sex. Other studies have found the dislocation rate to be 3% to 7%, which is lower than the dislocation rate observed in this study.8,10,16
The reasons for this higher rate of dislocation are not entirely clear. A veteran population has poorer overall health than the general population, which may contribute to the higher than previously reported dislocation rates.22 These results can be applied to the management of veterans seeking THA.
There have been conflicting reports regarding the impact a patient’s sex has on THA outcomes in the general population.23-26 This study found that female patients had higher rates of dislocation within 1 year of THA than male patients. This difference, which could be due to differences in baseline anatomic hip morphology between the sexes; females tend to have smaller femoral head sizes and less offset compared with males.27,28 However, this finding could have been confounded by the small number of female veterans in the study cohort.
A type 2 diabetes mellitus (T2DM) diagnosis, which is a component of CCI, trended toward increased risk of prosthetic hip dislocation. Multiple studies have also discussed the increased risk of postoperative infections and revisions following THA in patients with T2DM.29-31 One study found T2DM to be an independent risk factor for immediate in-hospital postoperative complications following hip arthroplasty.32
Another factor that may influence postoperative dislocation risk is surgical approach. The posterior approach has historically been associated with higher rates of instability when compared to anterior or lateral THA.33 Researchers have also looked at the role that surgical approach plays in patients with prior LSF. Huebschmann et al confirmed that not only is LSF a significant risk factor for dislocation following THA, but anterior and laterally based surgical approaches may mitigate this risk.34
Limitations
As a retrospective cohort study, the reliability of the data hinges on complete documentation. Documentation of all encounters for dislocations was obtained from the VA Computerized Patient Record System, which may have led to some dislocation events being missed. However, as long as there was adequate postoperative follow-up, it was assumed all events outside the VA were included. Another limitation of this study was that male patients greatly outnumbered female patients, and this fact could limit the generalizability of findings to the population as a whole.
Conclusions
This study in a veteran population found that prior LSF and female sex were significant predictors for postoperative dislocation within 1 year of THA surgery. Additionally, the use of a dual-mobility liner was found to be protective against postoperative dislocation events. These data allow clinicians to better counsel veterans on the risk factors associated with postoperative dislocation and strategies to mitigate this risk.
- Sloan M, Premkumar A, Sheth NP. Projected volume of primary total joint arthroplasty in the U.S., 2014 to 2030. J Bone Joint Surg Am. 2018;100:1455-1460. doi:10.2106/JBJS.17.01617
- Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91:128-133. doi:10.2106/JBJS.H.00155
- Kurtz SM, Ong KL, Schmier J, et al. Future clinical and economic impact of revision total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89:144-151. doi:10.2106/JBJS.G.00587
- Kurtz SM, Ong KL, Schmier J, et al. Primary and revision arthroplasty surgery caseloads in the United States from 1990 to 2004. J Arthroplasty. 2009;24:195-203. doi:10.1016/j.arth.2007.11.015
- Yamato Y, Furuhashi H, Hasegawa T, et al. Simulation of implant impingement after spinal corrective fusion surgery in patients with previous total hip arthroplasty: a retrospective case series. Spine (Phila Pa 1976). 2021;46:512-519. doi:10.1097/BRS.0000000000003836
- Mudrick CA, Melvin JS, Springer BD. Late posterior hip instability after lumbar spinopelvic fusion. Arthroplast Today. 2015;1:25-29. doi:10.1016/j.artd.2015.05.002
- Diebo BG, Beyer GA, Grieco PW, et al. Complications in patients undergoing spinal fusion after THA. Clin Orthop Relat Res. 2018;476:412-417.doi:10.1007/s11999.0000000000000009 8.
- Sing DC, Barry JJ, Aguilar TU, et al. Prior lumbar spinal arthrodesis increases risk of prosthetic-related complication in total hip arthroplasty. J Arthroplasty. 2016;31:227-232.e1. doi:10.1016/j.arth.2016.02.069
- King CA, Landy DC, Martell JM, et al. Time to dislocation analysis of lumbar spine fusion following total hip arthroplasty: breaking up a happy home. J Arthroplasty. 2018;33:3768-3772. doi:10.1016/j.arth.2018.08.029
- Buckland AJ, Puvanesarajah V, Vigdorchik J, et al. Dislocation of a primary total hip arthroplasty is more common in patients with a lumbar spinal fusion. Bone Joint J. 2017;99-B:585-591.doi:10.1302/0301-620X.99B5.BJJ-2016-0657.R1
- Pirruccio K, Premkumar A, Sheth NP. The burden of prosthetic hip dislocations in the United States is projected to significantly increase by 2035. Hip Int. 2021;31:714-721. doi:10.1177/1120700020923619
- Salib CG, Reina N, Perry KI, et al. Lumbar fusion involving the sacrum increases dislocation risk in primary total hip arthroplasty. Bone Joint J. 2019;101-B:198-206. doi:10.1302/0301-620X.101B2.BJJ-2018-0754.R1
- An VVG, Phan K, Sivakumar BS, et al. Prior lumbar spinal fusion is associated with an increased risk of dislocation and revision in total hip arthroplasty: a meta-analysis. J Arthroplasty. 2018;33:297-300. doi:10.1016/j.arth.2017.08.040
- Klemt C, Padmanabha A, Tirumala V, et al. Lumbar spine fusion before revision total hip arthroplasty is associated with increased dislocation rates. J Am Acad Orthop Surg. 2021;29:e860-e868. doi:10.5435/JAAOS-D-20-00824
- Gausden EB, Parhar HS, Popper JE, et al. Risk factors for early dislocation following primary elective total hip arthroplasty. J Arthroplasty. 2018;33:1567-1571. doi:10.1016/j.arth.2017.12.034
- Malkani AL, Himschoot KJ, Ong KL, et al. Does timing of primary total hip arthroplasty prior to or after lumbar spine fusion have an effect on dislocation and revision rates?. J Arthroplasty. 2019;34:907-911. doi:10.1016/j.arth.2019.01.009
- Parilla FW, Shah RR, Gordon AC, et al. Does it matter: total hip arthroplasty or lumbar spinal fusion first? Preoperative sagittal spinopelvic measurements guide patient-specific surgical strategies in patients requiring both. J Arthroplasty. 2019;34:2652-2662. doi:10.1016/j.arth.2019.05.053
- Chalmers BP, Syku M, Sculco TP, et al. Dual-mobility constructs in primary total hip arthroplasty in high-risk patients with spinal fusions: our institutional experience. Arthroplast Today. 2020;6:749-754. doi:10.1016/j.artd.2020.07.024
- Nessler JM, Malkani AL, Sachdeva S, et al. Use of dual mobility cups in patients undergoing primary total hip arthroplasty with prior lumbar spine fusion. Int Orthop. 2020;44:857-862. doi:10.1007/s00264-020-04507-y
- Nessler JM, Malkani AL, Yep PJ, et al. Dislocation rates of primary total hip arthroplasty in patients with prior lumbar spine fusion and lumbar degenerative disk disease with and without utilization of dual mobility cups: an American Joint Replacement Registry study. J Am Acad Orthop Surg. 2023;31:e271-e277. doi:10.5435/JAAOS-D-22-00767
- Phan D, Bederman SS, Schwarzkopf R. The influence of sagittal spinal deformity on anteversion of the acetabular component in total hip arthroplasty. Bone Joint J. 2015;97-B:1017-1023. doi:10.1302/0301-620X.97B8.35700
- Agha Z, Lofgren RP, VanRuiswyk JV, et al. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160:3252-3257. doi:10.1001/archinte.160.21.325223.
- Basques BA, Bell JA, Fillingham YA, et al. Gender differences for hip and knee arthroplasty: complications and healthcare utilization. J Arthroplasty. 2019;34:1593-1597.e1. doi:10.1016/j.arth.2019.03.064
- Kim YH, Choi Y, Kim JS. Influence of patient-, design-, and surgery-related factors on rate of dislocation after primary cementless total hip arthroplasty. J Arthroplasty. 2009;24:1258-1263. doi:10.1016/j.arth.2009.03.017
- Chen A, Paxton L, Zheng X, et al. Association of sex with risk of 2-year revision among patients undergoing total hip arthroplasty. JAMA Netw Open. 2021;4:e2110687. doi:10.1001/jamanetworkopen.2021.10687
- Inacio MCS, Ake CF, Paxton EW, et al. Sex and risk of hip implant failure: assessing total hip arthroplasty outcomes in the United States. JAMA Intern Med. 2013;173:435-441. doi:10.1001/jamainternmed.2013.3271
- Karlson EW, Daltroy LH, Liang MH, et al. Gender differences in patient preferences may underlie differential utilization of elective surgery. Am J Med. 1997;102:524-530. doi:10.1016/s0002-9343(97)00050-8
- Kostamo T, Bourne RB, Whittaker JP, et al. No difference in gender-specific hip replacement outcomes. Clin Orthop Relat Res. 2009;467:135-140. doi:10.1007/s11999-008-0466-2
- Papagelopoulos PJ, Idusuyi OB, Wallrichs SL, et al. Long term outcome and survivorship analysis of primary total knee arthroplasty in patients with diabetes mellitus. Clin Orthop Relat Res. 1996;(330):124-132. doi:10.1097/00003086-199609000-00015
- Fitzgerald RH Jr, Nolan DR, Ilstrup DM, et al. Deep wound sepsis following total hip arthroplasty. J Bone Joint Surg Am. 1977;59:847-855.
- Blom AW, Brown J, Taylor AH, et al. Infection after total knee arthroplasty. J Bone Joint Surg Br. 2004;86:688-691. doi:10.1302/0301-620x.86b5.14887
- Jain NB, Guller U, Pietrobon R, et al. Comorbidities increase complication rates in patients having arthroplasty. Clin Orthop Relat Res. 2005;435:232-238. doi:10.1097/01.blo.0000156479.97488.a2
- Docter S, Philpott HT, Godkin L, et al. Comparison of intra and post-operative complication rates among surgical approaches in Total Hip Arthroplasty: A systematic review and meta-analysis. J Orthop. 2020;20:310-325. doi:10.1016/j.jor.2020.05.008
- Huebschmann NA, Lawrence KW, Robin JX, et al. Does surgical approach affect dislocation rate after total hip arthroplasty in patients who have prior lumbar spinal fusion? A retrospective analysis of 16,223 cases. J Arthroplasty. 2024;39:S306-S313. doi:10.1016/j.arth.2024.03.068
- Sloan M, Premkumar A, Sheth NP. Projected volume of primary total joint arthroplasty in the U.S., 2014 to 2030. J Bone Joint Surg Am. 2018;100:1455-1460. doi:10.2106/JBJS.17.01617
- Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91:128-133. doi:10.2106/JBJS.H.00155
- Kurtz SM, Ong KL, Schmier J, et al. Future clinical and economic impact of revision total hip and knee arthroplasty. J Bone Joint Surg Am. 2007;89:144-151. doi:10.2106/JBJS.G.00587
- Kurtz SM, Ong KL, Schmier J, et al. Primary and revision arthroplasty surgery caseloads in the United States from 1990 to 2004. J Arthroplasty. 2009;24:195-203. doi:10.1016/j.arth.2007.11.015
- Yamato Y, Furuhashi H, Hasegawa T, et al. Simulation of implant impingement after spinal corrective fusion surgery in patients with previous total hip arthroplasty: a retrospective case series. Spine (Phila Pa 1976). 2021;46:512-519. doi:10.1097/BRS.0000000000003836
- Mudrick CA, Melvin JS, Springer BD. Late posterior hip instability after lumbar spinopelvic fusion. Arthroplast Today. 2015;1:25-29. doi:10.1016/j.artd.2015.05.002
- Diebo BG, Beyer GA, Grieco PW, et al. Complications in patients undergoing spinal fusion after THA. Clin Orthop Relat Res. 2018;476:412-417.doi:10.1007/s11999.0000000000000009 8.
- Sing DC, Barry JJ, Aguilar TU, et al. Prior lumbar spinal arthrodesis increases risk of prosthetic-related complication in total hip arthroplasty. J Arthroplasty. 2016;31:227-232.e1. doi:10.1016/j.arth.2016.02.069
- King CA, Landy DC, Martell JM, et al. Time to dislocation analysis of lumbar spine fusion following total hip arthroplasty: breaking up a happy home. J Arthroplasty. 2018;33:3768-3772. doi:10.1016/j.arth.2018.08.029
- Buckland AJ, Puvanesarajah V, Vigdorchik J, et al. Dislocation of a primary total hip arthroplasty is more common in patients with a lumbar spinal fusion. Bone Joint J. 2017;99-B:585-591.doi:10.1302/0301-620X.99B5.BJJ-2016-0657.R1
- Pirruccio K, Premkumar A, Sheth NP. The burden of prosthetic hip dislocations in the United States is projected to significantly increase by 2035. Hip Int. 2021;31:714-721. doi:10.1177/1120700020923619
- Salib CG, Reina N, Perry KI, et al. Lumbar fusion involving the sacrum increases dislocation risk in primary total hip arthroplasty. Bone Joint J. 2019;101-B:198-206. doi:10.1302/0301-620X.101B2.BJJ-2018-0754.R1
- An VVG, Phan K, Sivakumar BS, et al. Prior lumbar spinal fusion is associated with an increased risk of dislocation and revision in total hip arthroplasty: a meta-analysis. J Arthroplasty. 2018;33:297-300. doi:10.1016/j.arth.2017.08.040
- Klemt C, Padmanabha A, Tirumala V, et al. Lumbar spine fusion before revision total hip arthroplasty is associated with increased dislocation rates. J Am Acad Orthop Surg. 2021;29:e860-e868. doi:10.5435/JAAOS-D-20-00824
- Gausden EB, Parhar HS, Popper JE, et al. Risk factors for early dislocation following primary elective total hip arthroplasty. J Arthroplasty. 2018;33:1567-1571. doi:10.1016/j.arth.2017.12.034
- Malkani AL, Himschoot KJ, Ong KL, et al. Does timing of primary total hip arthroplasty prior to or after lumbar spine fusion have an effect on dislocation and revision rates?. J Arthroplasty. 2019;34:907-911. doi:10.1016/j.arth.2019.01.009
- Parilla FW, Shah RR, Gordon AC, et al. Does it matter: total hip arthroplasty or lumbar spinal fusion first? Preoperative sagittal spinopelvic measurements guide patient-specific surgical strategies in patients requiring both. J Arthroplasty. 2019;34:2652-2662. doi:10.1016/j.arth.2019.05.053
- Chalmers BP, Syku M, Sculco TP, et al. Dual-mobility constructs in primary total hip arthroplasty in high-risk patients with spinal fusions: our institutional experience. Arthroplast Today. 2020;6:749-754. doi:10.1016/j.artd.2020.07.024
- Nessler JM, Malkani AL, Sachdeva S, et al. Use of dual mobility cups in patients undergoing primary total hip arthroplasty with prior lumbar spine fusion. Int Orthop. 2020;44:857-862. doi:10.1007/s00264-020-04507-y
- Nessler JM, Malkani AL, Yep PJ, et al. Dislocation rates of primary total hip arthroplasty in patients with prior lumbar spine fusion and lumbar degenerative disk disease with and without utilization of dual mobility cups: an American Joint Replacement Registry study. J Am Acad Orthop Surg. 2023;31:e271-e277. doi:10.5435/JAAOS-D-22-00767
- Phan D, Bederman SS, Schwarzkopf R. The influence of sagittal spinal deformity on anteversion of the acetabular component in total hip arthroplasty. Bone Joint J. 2015;97-B:1017-1023. doi:10.1302/0301-620X.97B8.35700
- Agha Z, Lofgren RP, VanRuiswyk JV, et al. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160:3252-3257. doi:10.1001/archinte.160.21.325223.
- Basques BA, Bell JA, Fillingham YA, et al. Gender differences for hip and knee arthroplasty: complications and healthcare utilization. J Arthroplasty. 2019;34:1593-1597.e1. doi:10.1016/j.arth.2019.03.064
- Kim YH, Choi Y, Kim JS. Influence of patient-, design-, and surgery-related factors on rate of dislocation after primary cementless total hip arthroplasty. J Arthroplasty. 2009;24:1258-1263. doi:10.1016/j.arth.2009.03.017
- Chen A, Paxton L, Zheng X, et al. Association of sex with risk of 2-year revision among patients undergoing total hip arthroplasty. JAMA Netw Open. 2021;4:e2110687. doi:10.1001/jamanetworkopen.2021.10687
- Inacio MCS, Ake CF, Paxton EW, et al. Sex and risk of hip implant failure: assessing total hip arthroplasty outcomes in the United States. JAMA Intern Med. 2013;173:435-441. doi:10.1001/jamainternmed.2013.3271
- Karlson EW, Daltroy LH, Liang MH, et al. Gender differences in patient preferences may underlie differential utilization of elective surgery. Am J Med. 1997;102:524-530. doi:10.1016/s0002-9343(97)00050-8
- Kostamo T, Bourne RB, Whittaker JP, et al. No difference in gender-specific hip replacement outcomes. Clin Orthop Relat Res. 2009;467:135-140. doi:10.1007/s11999-008-0466-2
- Papagelopoulos PJ, Idusuyi OB, Wallrichs SL, et al. Long term outcome and survivorship analysis of primary total knee arthroplasty in patients with diabetes mellitus. Clin Orthop Relat Res. 1996;(330):124-132. doi:10.1097/00003086-199609000-00015
- Fitzgerald RH Jr, Nolan DR, Ilstrup DM, et al. Deep wound sepsis following total hip arthroplasty. J Bone Joint Surg Am. 1977;59:847-855.
- Blom AW, Brown J, Taylor AH, et al. Infection after total knee arthroplasty. J Bone Joint Surg Br. 2004;86:688-691. doi:10.1302/0301-620x.86b5.14887
- Jain NB, Guller U, Pietrobon R, et al. Comorbidities increase complication rates in patients having arthroplasty. Clin Orthop Relat Res. 2005;435:232-238. doi:10.1097/01.blo.0000156479.97488.a2
- Docter S, Philpott HT, Godkin L, et al. Comparison of intra and post-operative complication rates among surgical approaches in Total Hip Arthroplasty: A systematic review and meta-analysis. J Orthop. 2020;20:310-325. doi:10.1016/j.jor.2020.05.008
- Huebschmann NA, Lawrence KW, Robin JX, et al. Does surgical approach affect dislocation rate after total hip arthroplasty in patients who have prior lumbar spinal fusion? A retrospective analysis of 16,223 cases. J Arthroplasty. 2024;39:S306-S313. doi:10.1016/j.arth.2024.03.068
Effects of Lumbar Fusion and Dual-Mobility Liners on Dislocation Rates Following Total Hip Arthroplasty in a Veteran Population
Effects of Lumbar Fusion and Dual-Mobility Liners on Dislocation Rates Following Total Hip Arthroplasty in a Veteran Population
A Candida Glabrata-Associated Prosthetic Joint Infection: Case Report and Literature Review
A Candida Glabrata-Associated Prosthetic Joint Infection: Case Report and Literature Review
Prosthetic joint infection (PJI) occurs in about 1% to 2% of joint replacements. 1 Risk factors include immunosuppression, diabetes, chronic illnesses, and prolonged operative time.2 Bacterial infections constitute most of these infections, while fungal pathogens account for about 1%. Candida (C.) species, predominantly C. albicans, are responsible for most PJIs.1,3 In contrast, C. glabrata is a rare cause of fungal PJI, with only 18 PJI cases currently reported in the literature.4 C. glabrata PJI occurs more frequently among immunosuppressed patients and is associated with a higher treatment failure rate despite antifungal therapy.5 Treatment of fungal PJI is often complicated, involving multiple surgical debridements, prolonged antifungal therapy, and in some cases, prosthesis removal.6 However, given the rarity of C. glabrata as a PJI pathogen, no standardized treatment guidelines exist, leading to potential delays in diagnosis and tailored treatment.7,8
CASE PRESENTATION
A male Vietnam veteran aged 75 years presented to the emergency department in July 2023 with a fluid collection over his left hip surgical incision site. The patient had a complex medical history that included chronic kidney disease, well-controlled type 2 diabetes, hypertension, and osteoarthritis. His history was further complicated by nonalcoholic steatohepatitis with hepatocellular carcinoma that was treated with transarterial radioembolization and yttrium-90. The patient had undergone a left total hip arthroplasty in 1996 and subsequent open reduction and internal fixation about 9 months prior to his presentation. The patient reported the fluid had been present for about 6 weeks, while he received outpatient monitoring by the orthopedic surgery service. He sought emergency care after noting a moderate amount of purulent discharge on his clothing originating from his hip. In the week prior to admission, the patient observed progressive erythema, warmth, and tenderness over the incision site. Despite these symptoms, the patient remained ambulatory and able to walk long distances with the use of an assistive device.
Upon presentation, the patient was afebrile and normotensive. Laboratory testing revealed an elevated erythrocyte sedimentation rate of 77 mm/h (reference range, 0-20 mm/h) and a C-reactive protein of 9.8 mg/L (reference range, 0-2.5 mg/L), suggesting an underlying infectious process. A physical examination revealed a well-healed incision over the left hip with a poorly defined area of fluctuance and evidence of wound dehiscence. The left lower extremity was swollen with 2+ pitting edema, but tenderness was localized to the incision site. Magnetic resonance imaging of the left hip revealed a multiloculated fluid collection abutting the left greater trochanter with extension to the skin surface and inferior extension along the entire length of the surgical fixation hardware (Figure).
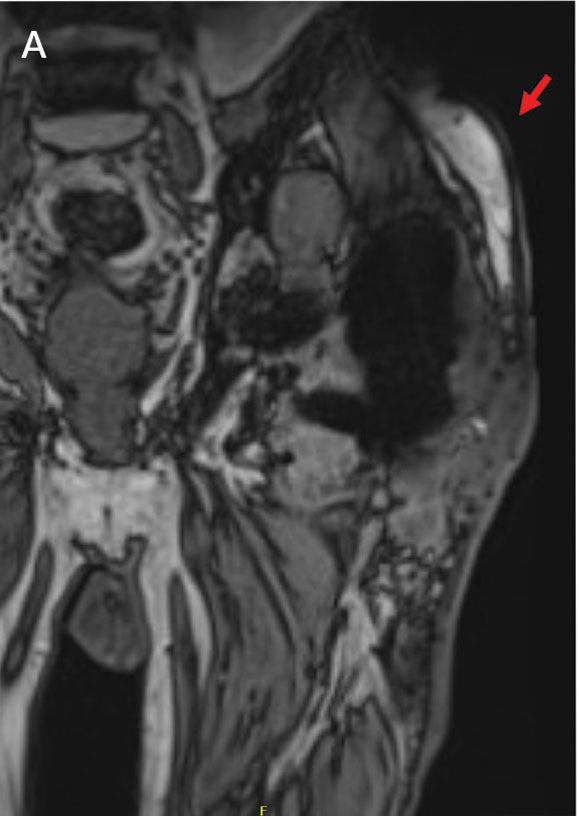
Upon admission, orthopedic surgery performed a bedside aspiration of the fluid collection. Samples were sent for analysis, including cell count and bacterial and fungal cultures. Initial blood cultures were sterile. Due to concerns for a bacterial infection, the patient was started on empiric intravenous (IV) ceftriaxone 2 g/day and IV vancomycin 1250 mg/day. Synovial fluid analysis revealed an elevated white blood cell count of 45,000/ìL, but bacterial cultures were negative. Five days after admission, the fungal culture from the left hip wound was notable for presence of C. glabrata, prompting an infectious diseases (ID) consultation. IV micafungin 100 mg/day was initiated as empiric antifungal therapy.
ID and orthopedic surgery teams determined that a combined medical and surgical approach would be best suited for infection control. They proposed 2 main approaches: complete hardware replacement with washout, which carried a higher morbidity risk but a better chance of infection resolution, or partial hardware replacement with washout, which was associated with a lower morbidity risk but a higher risk of infection persistence and recurrence. This decision was particularly challenging for the patient, who prioritized maintaining his functional status, including his ability to continue dancing for pleasure. The patient opted for a more conservative approach, electing to proceed with antifungal therapy and debridement while retaining the prosthetic joint.
After 11 days of hospitalization, the patient was discharged with a peripherally inserted central catheter for long-term antifungal infusions of micafungin 150 mg/day at home. Fungal sensitivity test results several days after discharge confirmed susceptibility to micafungin.
About 2 weeks after discharge, the patient underwent debridement and implant retention (DAIR). Wound cultures were positive for C. glabrata, Enterococcus faecalis, Staphylococcus epidermidis, and Corynebacterium tuberculostearicum. Based on susceptibilities, he completed a 2-month course of IV micafungin 150 mg daily and daptomycin 750 mg daily, followed by an oral suppressive regimen consisting of doxycycline 100 mg twice daily, amoxicillin-clavulanate 2 g twice daily, and fluconazole initially 800 mg daily adjusted to 400 mg daily. The patient continued wound management with twice-daily dressing changes.
Nine months after DAIR, the patient remained on suppressive antifungal and antibacterial therapy. He continued to experience serous drainage from the wound, which greatly affected his quality of life. After discussion with his family and the orthopedic surgery team, he agreed to proceed with a 2-staged revision arthroplasty involving prosthetic explant and antibiotic spacer placement. However, the surgery was postponed due to findings of anemia (hemoglobin, 8.9 g/dL) and thrombocytopenia (platelet count, 73 x 103/λL). At the time of this report, the patient was being monitored closely with his multidisciplinary care team for the planned orthopedic procedure.
DISCUSSION
PJI is the most common cause of primary hip arthroplasty failure; however, fungal species only make up about 1% of PJIs.3,9-11 Patients are typically immunocompromised, undergoing antineoplastic therapies for malignancy, or have other comorbid conditions such as diabetes.12,13 C. glabrata presents a unique diagnostic and therapeutic challenge as it is not only rare but also notorious for its resistance to common antifungal agents. C. glabrata is known to develop multidrug resistance through the rapid accumulation of genomic mutations.14 Its propensity towards forming protective biofilm also arms it with intrinsic resistance to agents like fluconazole.15 Furthermore, based on a review of the available reports in the literature, C. glabrata PJIs are often insidious and present with symptoms closely mimicking those of bacterial PJIs, as it did in the patient in this case.16
Synovial fluid analysis, fungal cultures, and sensitivity testing are paramount for ensuring proper diagnosis for fungal PJI. The patient in this case was empirically treated with micafungin based on recommendations from the ID team. When the sensitivities results were reviewed, the same antifungal therapy was continued. Echinocandins have a favorable toxicity profile in long-term use, as well as efficacy against biofilm-producing organisms like C. glabrata.17,18
While there are a few cases citing DAIR as a feasible surgical strategy for treating fungal PJI, more recent studies have reported greater success with a 2-staged revision arthroplasty involving some combination of debridement, placement of antibiotic-loaded bone cement spacers, and partial or total exchange of the infected prosthetic joint.4,19-23 In this case, complete hardware replacement would have offered the patient the most favorable outlook for eliminating this fungal infection. However, given the patient’s advanced age, significant underlying comorbidities, and functional status, medical management with antifungal therapy and DAIR was favored.
Based on the discussion from the 6-month follow-up visit, the patient was experiencing progressive and persistent wound drainage and frequent dressing changes, highlighting the limitations of medical management for PJI in the setting of retained prosthesis. If the patient ultimately proceeds with a more invasive surgical intervention, another important consideration will be the likelihood of fungal PJI recurrence. At present, fungal PJI recurrence rates following antifungal and surgical treatment have been reported to range between 0% to 50%, which is too imprecise to be considered clinically useful.22-24
Given the ambiguity surrounding management guidelines and limited treatment options, it is crucial to emphasize the timeline of this patient’s clinical presentation and subsequent course of treatment. Upon presentation to the ED in late July, fungal PJI was considered less likely. Initial blood cultures from presentation were negative, which is common with PJIs. It was not until 5 days later that the left hip wound culture showed moderate growth of C. glabrata. Identifying a PJI is clinically challenging due to the lack of standardized diagnostic criteria. However, timely identification and diagnosis of fungal PJI with appropriate antifungal therapy, in patients with limited curative options due to comorbidities, can significantly improve quality of life and overall outcomes.25 Routine fungal and mycobacterial cultures are not currently recommended in PJI guidelines, but this case illustrates it is imperative in immunocompromised hosts.26
This case and the current paucity of similar cases in the literature stress the importance of clinicians publishing their experience in the management of fungal PJI. We strongly recommend that clinicians approach each suspected PJI with careful consideration of the patient’s unique risk factors, comorbidities, and goals of care, when deciding on a curative vs suppressive approach to therapy.
CONCLUSIONS
This case report highlights the importance of considering fungal pathogens for PJIs, especially in high-risk patients, the value of obtaining fungal cultures, the necessity of a multidisciplinary approach, the role of antifungal susceptibility testing, and consideration for the feasibility of a surgical intervention. It underscores the challenges in diagnosis and treatment of C. glabrata-associated PJI, emphasizing the importance of clinician experience-sharing in developing evidence-based management strategies. As the understanding of fungal PJI evolves, continued research and clinical data collection remain crucial for improving patient outcomes in the management of these complex cases.
- Osmon DR, Berbari EF, Berendt AR, et al. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):1-10. doi:10.1093/cid/cis966
- Eka A, Chen AF. Patient-related medical risk factors for periprosthetic joint infection of the hip and knee. Ann Transl Med. 2015;3(16):233. doi:10.3978/j.issn.2305-5839.2015.09.26
- Darouiche RO, Hamill RJ, Musher DM, Young EJ, Harris RL. Periprosthetic candidal infections following arthroplasty. Rev Infect Dis. 1989;11(1):89-96. doi:10.1093/clinids/11.1.89
- Koutserimpas C, Zervakis SG, Maraki S, et al. Non-albicans Candida prosthetic joint infections: a systematic review of treatment. World J Clin Cases. 2019;7(12):1430- 1443. doi:10.12998/wjcc.v7.i12.1430
- Fidel PL Jr, Vazquez JA, Sobel JD. Candida glabrata: review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans. Clin Microbiol Rev. 1999;12(1):80-96. doi:10.1128/CMR.12.1.80
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Lee YR, Kim HJ, Lee EJ, Sohn JW, Kim MJ, Yoon YK. Prosthetic joint infections caused by candida species: a systematic review and a case series. Mycopathologia. 2019;184(1):23-33. doi:10.1007/s11046-018-0286-1
- Herndon CL, Rowe TM, Metcalf RW, et al. Treatment outcomes of fungal periprosthetic joint infection. J Arthroplasty. 2023;38(11):2436-2440.e1. doi:10.1016/j.arth.2023.05.009
- Delaunay C, Hamadouche M, Girard J, Duhamel A; SoFCOT. What are the causes for failures of primary hip arthroplasties in France? Clin Orthop Relat Res. 2013;471(12): 3863-3869. doi:10.1007/s11999-013-2935-5
- Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91(1): 128-133. doi:10.2106/JBJS.H.00155
- Furnes O, Lie SA, Espehaug B, Vollset SE, Engesaeter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586. doi:10.1302/0301-620x.83b4.11223
- Gonzalez MR, Bedi ADS, Karczewski D, Lozano-Calderon SA. Treatment and outcomes of fungal prosthetic joint infections: a systematic review of 225 cases. J Arthroplasty. 2023;38(11):2464-2471.e1. doi:10.1016/j.arth.2023.05.003
- Gonzalez MR, Pretell-Mazzini J, Lozano-Calderon SA. Risk factors and management of prosthetic joint infections in megaprostheses-a review of the literature. Antibiotics (Basel). 2023;13(1):25. doi:10.3390/antibiotics13010025
- Biswas C, Chen SC, Halliday C, et al. Identification of genetic markers of resistance to echinocandins, azoles and 5-fluorocytosine in Candida glabrata by next-generation sequencing: a feasibility study. Clin Microbiol Infect. 2017;23(9):676.e7-676.e10. doi:10.1016/j.cmi.2017.03.014
- Hassan Y, Chew SY, Than LTL. Candida glabrata: pathogenicity and resistance mechanisms for adaptation and survival. J Fungi (Basel). 2021;7(8):667. doi:10.3390/jof7080667
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Pierce CG, Uppuluri P, Tristan AR, et al. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nat Protoc. 2008;3(9):1494-1500. doi:10.1038/nport.2008.141
- Koutserimpas C, Samonis G, Velivassakis E, Iliopoulou- Kosmadaki S, Kontakis G, Kofteridis DP. Candida glabrata prosthetic joint infection, successfully treated with anidulafungin: a case report and review of the literature. Mycoses. 2018;61(4):266-269. doi:10.1111/myc.12736
- Brooks DH, Pupparo F. Successful salvage of a primary total knee arthroplasty infected with Candida parapsilosis. J Arthroplasty. 1998;13(6):707-712. doi:10.1016/s0883-5403(98)80017-x
- Merrer J, Dupont B, Nieszkowska A, De Jonghe B, Outin H. Candida albicans prosthetic arthritis treated with fluconazole alone. J Infect. 2001;42(3):208-209. doi:10.1053/jinf.2001.0819
- Koutserimpas C, Naoum S, Alpantaki K, et al. Fungal prosthetic joint infection in revised knee arthroplasty: an orthopaedic surgeon’s nightmare. Diagnostics (Basel). 2022;12(7):1606. doi:10.3390/diagnostics12071606
- Gao Z, Li X, Du Y, Peng Y, Wu W, Zhou Y. Success rate of fungal peri-prosthetic joint infection treated by 2-stage revision and potential risk factors of treatment failure: a retrospective study. Med Sci Monit. 2018;24:5549-5557. doi:10.12659/MSM.909168
- Hwang BH, Yoon JY, Nam CH, et al. Fungal periprosthetic joint infection after primary total knee replacement. J Bone Joint Surg Br. 2012;94(5):656-659. doi:10.1302/0301-620X.94B5.28125
- Ueng SW, Lee CY, Hu CC, Hsieh PH, Chang Y. What is the success of treatment of hip and knee candidal periprosthetic joint infection? Clin Orthop Relat Res. 2013;471(9):3002-3009. doi:10.1007/s11999-013-3007-6
- Nodzo, Scott R. MD; Bauer, Thomas MD, PhD; Pottinger, et al. Conventional diagnostic challenges in periprosthetic joint infection. J Am Acad Orthop Surg. 2015;23 Suppl:S18-S25. doi:10.5435/JAAOS-D-14-00385
- American Academy of Orthopaedic Surgeons. Diagnosis and prevention of periprosthetic joint infections. March 11, 2019. Accessed February 5, 2025. https://www.aaos.org/pjicpg
Prosthetic joint infection (PJI) occurs in about 1% to 2% of joint replacements. 1 Risk factors include immunosuppression, diabetes, chronic illnesses, and prolonged operative time.2 Bacterial infections constitute most of these infections, while fungal pathogens account for about 1%. Candida (C.) species, predominantly C. albicans, are responsible for most PJIs.1,3 In contrast, C. glabrata is a rare cause of fungal PJI, with only 18 PJI cases currently reported in the literature.4 C. glabrata PJI occurs more frequently among immunosuppressed patients and is associated with a higher treatment failure rate despite antifungal therapy.5 Treatment of fungal PJI is often complicated, involving multiple surgical debridements, prolonged antifungal therapy, and in some cases, prosthesis removal.6 However, given the rarity of C. glabrata as a PJI pathogen, no standardized treatment guidelines exist, leading to potential delays in diagnosis and tailored treatment.7,8
CASE PRESENTATION
A male Vietnam veteran aged 75 years presented to the emergency department in July 2023 with a fluid collection over his left hip surgical incision site. The patient had a complex medical history that included chronic kidney disease, well-controlled type 2 diabetes, hypertension, and osteoarthritis. His history was further complicated by nonalcoholic steatohepatitis with hepatocellular carcinoma that was treated with transarterial radioembolization and yttrium-90. The patient had undergone a left total hip arthroplasty in 1996 and subsequent open reduction and internal fixation about 9 months prior to his presentation. The patient reported the fluid had been present for about 6 weeks, while he received outpatient monitoring by the orthopedic surgery service. He sought emergency care after noting a moderate amount of purulent discharge on his clothing originating from his hip. In the week prior to admission, the patient observed progressive erythema, warmth, and tenderness over the incision site. Despite these symptoms, the patient remained ambulatory and able to walk long distances with the use of an assistive device.
Upon presentation, the patient was afebrile and normotensive. Laboratory testing revealed an elevated erythrocyte sedimentation rate of 77 mm/h (reference range, 0-20 mm/h) and a C-reactive protein of 9.8 mg/L (reference range, 0-2.5 mg/L), suggesting an underlying infectious process. A physical examination revealed a well-healed incision over the left hip with a poorly defined area of fluctuance and evidence of wound dehiscence. The left lower extremity was swollen with 2+ pitting edema, but tenderness was localized to the incision site. Magnetic resonance imaging of the left hip revealed a multiloculated fluid collection abutting the left greater trochanter with extension to the skin surface and inferior extension along the entire length of the surgical fixation hardware (Figure).


Upon admission, orthopedic surgery performed a bedside aspiration of the fluid collection. Samples were sent for analysis, including cell count and bacterial and fungal cultures. Initial blood cultures were sterile. Due to concerns for a bacterial infection, the patient was started on empiric intravenous (IV) ceftriaxone 2 g/day and IV vancomycin 1250 mg/day. Synovial fluid analysis revealed an elevated white blood cell count of 45,000/ìL, but bacterial cultures were negative. Five days after admission, the fungal culture from the left hip wound was notable for presence of C. glabrata, prompting an infectious diseases (ID) consultation. IV micafungin 100 mg/day was initiated as empiric antifungal therapy.
ID and orthopedic surgery teams determined that a combined medical and surgical approach would be best suited for infection control. They proposed 2 main approaches: complete hardware replacement with washout, which carried a higher morbidity risk but a better chance of infection resolution, or partial hardware replacement with washout, which was associated with a lower morbidity risk but a higher risk of infection persistence and recurrence. This decision was particularly challenging for the patient, who prioritized maintaining his functional status, including his ability to continue dancing for pleasure. The patient opted for a more conservative approach, electing to proceed with antifungal therapy and debridement while retaining the prosthetic joint.
After 11 days of hospitalization, the patient was discharged with a peripherally inserted central catheter for long-term antifungal infusions of micafungin 150 mg/day at home. Fungal sensitivity test results several days after discharge confirmed susceptibility to micafungin.
About 2 weeks after discharge, the patient underwent debridement and implant retention (DAIR). Wound cultures were positive for C. glabrata, Enterococcus faecalis, Staphylococcus epidermidis, and Corynebacterium tuberculostearicum. Based on susceptibilities, he completed a 2-month course of IV micafungin 150 mg daily and daptomycin 750 mg daily, followed by an oral suppressive regimen consisting of doxycycline 100 mg twice daily, amoxicillin-clavulanate 2 g twice daily, and fluconazole initially 800 mg daily adjusted to 400 mg daily. The patient continued wound management with twice-daily dressing changes.
Nine months after DAIR, the patient remained on suppressive antifungal and antibacterial therapy. He continued to experience serous drainage from the wound, which greatly affected his quality of life. After discussion with his family and the orthopedic surgery team, he agreed to proceed with a 2-staged revision arthroplasty involving prosthetic explant and antibiotic spacer placement. However, the surgery was postponed due to findings of anemia (hemoglobin, 8.9 g/dL) and thrombocytopenia (platelet count, 73 x 103/λL). At the time of this report, the patient was being monitored closely with his multidisciplinary care team for the planned orthopedic procedure.
DISCUSSION
PJI is the most common cause of primary hip arthroplasty failure; however, fungal species only make up about 1% of PJIs.3,9-11 Patients are typically immunocompromised, undergoing antineoplastic therapies for malignancy, or have other comorbid conditions such as diabetes.12,13 C. glabrata presents a unique diagnostic and therapeutic challenge as it is not only rare but also notorious for its resistance to common antifungal agents. C. glabrata is known to develop multidrug resistance through the rapid accumulation of genomic mutations.14 Its propensity towards forming protective biofilm also arms it with intrinsic resistance to agents like fluconazole.15 Furthermore, based on a review of the available reports in the literature, C. glabrata PJIs are often insidious and present with symptoms closely mimicking those of bacterial PJIs, as it did in the patient in this case.16
Synovial fluid analysis, fungal cultures, and sensitivity testing are paramount for ensuring proper diagnosis for fungal PJI. The patient in this case was empirically treated with micafungin based on recommendations from the ID team. When the sensitivities results were reviewed, the same antifungal therapy was continued. Echinocandins have a favorable toxicity profile in long-term use, as well as efficacy against biofilm-producing organisms like C. glabrata.17,18
While there are a few cases citing DAIR as a feasible surgical strategy for treating fungal PJI, more recent studies have reported greater success with a 2-staged revision arthroplasty involving some combination of debridement, placement of antibiotic-loaded bone cement spacers, and partial or total exchange of the infected prosthetic joint.4,19-23 In this case, complete hardware replacement would have offered the patient the most favorable outlook for eliminating this fungal infection. However, given the patient’s advanced age, significant underlying comorbidities, and functional status, medical management with antifungal therapy and DAIR was favored.
Based on the discussion from the 6-month follow-up visit, the patient was experiencing progressive and persistent wound drainage and frequent dressing changes, highlighting the limitations of medical management for PJI in the setting of retained prosthesis. If the patient ultimately proceeds with a more invasive surgical intervention, another important consideration will be the likelihood of fungal PJI recurrence. At present, fungal PJI recurrence rates following antifungal and surgical treatment have been reported to range between 0% to 50%, which is too imprecise to be considered clinically useful.22-24
Given the ambiguity surrounding management guidelines and limited treatment options, it is crucial to emphasize the timeline of this patient’s clinical presentation and subsequent course of treatment. Upon presentation to the ED in late July, fungal PJI was considered less likely. Initial blood cultures from presentation were negative, which is common with PJIs. It was not until 5 days later that the left hip wound culture showed moderate growth of C. glabrata. Identifying a PJI is clinically challenging due to the lack of standardized diagnostic criteria. However, timely identification and diagnosis of fungal PJI with appropriate antifungal therapy, in patients with limited curative options due to comorbidities, can significantly improve quality of life and overall outcomes.25 Routine fungal and mycobacterial cultures are not currently recommended in PJI guidelines, but this case illustrates it is imperative in immunocompromised hosts.26
This case and the current paucity of similar cases in the literature stress the importance of clinicians publishing their experience in the management of fungal PJI. We strongly recommend that clinicians approach each suspected PJI with careful consideration of the patient’s unique risk factors, comorbidities, and goals of care, when deciding on a curative vs suppressive approach to therapy.
CONCLUSIONS
This case report highlights the importance of considering fungal pathogens for PJIs, especially in high-risk patients, the value of obtaining fungal cultures, the necessity of a multidisciplinary approach, the role of antifungal susceptibility testing, and consideration for the feasibility of a surgical intervention. It underscores the challenges in diagnosis and treatment of C. glabrata-associated PJI, emphasizing the importance of clinician experience-sharing in developing evidence-based management strategies. As the understanding of fungal PJI evolves, continued research and clinical data collection remain crucial for improving patient outcomes in the management of these complex cases.
Prosthetic joint infection (PJI) occurs in about 1% to 2% of joint replacements. 1 Risk factors include immunosuppression, diabetes, chronic illnesses, and prolonged operative time.2 Bacterial infections constitute most of these infections, while fungal pathogens account for about 1%. Candida (C.) species, predominantly C. albicans, are responsible for most PJIs.1,3 In contrast, C. glabrata is a rare cause of fungal PJI, with only 18 PJI cases currently reported in the literature.4 C. glabrata PJI occurs more frequently among immunosuppressed patients and is associated with a higher treatment failure rate despite antifungal therapy.5 Treatment of fungal PJI is often complicated, involving multiple surgical debridements, prolonged antifungal therapy, and in some cases, prosthesis removal.6 However, given the rarity of C. glabrata as a PJI pathogen, no standardized treatment guidelines exist, leading to potential delays in diagnosis and tailored treatment.7,8
CASE PRESENTATION
A male Vietnam veteran aged 75 years presented to the emergency department in July 2023 with a fluid collection over his left hip surgical incision site. The patient had a complex medical history that included chronic kidney disease, well-controlled type 2 diabetes, hypertension, and osteoarthritis. His history was further complicated by nonalcoholic steatohepatitis with hepatocellular carcinoma that was treated with transarterial radioembolization and yttrium-90. The patient had undergone a left total hip arthroplasty in 1996 and subsequent open reduction and internal fixation about 9 months prior to his presentation. The patient reported the fluid had been present for about 6 weeks, while he received outpatient monitoring by the orthopedic surgery service. He sought emergency care after noting a moderate amount of purulent discharge on his clothing originating from his hip. In the week prior to admission, the patient observed progressive erythema, warmth, and tenderness over the incision site. Despite these symptoms, the patient remained ambulatory and able to walk long distances with the use of an assistive device.
Upon presentation, the patient was afebrile and normotensive. Laboratory testing revealed an elevated erythrocyte sedimentation rate of 77 mm/h (reference range, 0-20 mm/h) and a C-reactive protein of 9.8 mg/L (reference range, 0-2.5 mg/L), suggesting an underlying infectious process. A physical examination revealed a well-healed incision over the left hip with a poorly defined area of fluctuance and evidence of wound dehiscence. The left lower extremity was swollen with 2+ pitting edema, but tenderness was localized to the incision site. Magnetic resonance imaging of the left hip revealed a multiloculated fluid collection abutting the left greater trochanter with extension to the skin surface and inferior extension along the entire length of the surgical fixation hardware (Figure).


Upon admission, orthopedic surgery performed a bedside aspiration of the fluid collection. Samples were sent for analysis, including cell count and bacterial and fungal cultures. Initial blood cultures were sterile. Due to concerns for a bacterial infection, the patient was started on empiric intravenous (IV) ceftriaxone 2 g/day and IV vancomycin 1250 mg/day. Synovial fluid analysis revealed an elevated white blood cell count of 45,000/ìL, but bacterial cultures were negative. Five days after admission, the fungal culture from the left hip wound was notable for presence of C. glabrata, prompting an infectious diseases (ID) consultation. IV micafungin 100 mg/day was initiated as empiric antifungal therapy.
ID and orthopedic surgery teams determined that a combined medical and surgical approach would be best suited for infection control. They proposed 2 main approaches: complete hardware replacement with washout, which carried a higher morbidity risk but a better chance of infection resolution, or partial hardware replacement with washout, which was associated with a lower morbidity risk but a higher risk of infection persistence and recurrence. This decision was particularly challenging for the patient, who prioritized maintaining his functional status, including his ability to continue dancing for pleasure. The patient opted for a more conservative approach, electing to proceed with antifungal therapy and debridement while retaining the prosthetic joint.
After 11 days of hospitalization, the patient was discharged with a peripherally inserted central catheter for long-term antifungal infusions of micafungin 150 mg/day at home. Fungal sensitivity test results several days after discharge confirmed susceptibility to micafungin.
About 2 weeks after discharge, the patient underwent debridement and implant retention (DAIR). Wound cultures were positive for C. glabrata, Enterococcus faecalis, Staphylococcus epidermidis, and Corynebacterium tuberculostearicum. Based on susceptibilities, he completed a 2-month course of IV micafungin 150 mg daily and daptomycin 750 mg daily, followed by an oral suppressive regimen consisting of doxycycline 100 mg twice daily, amoxicillin-clavulanate 2 g twice daily, and fluconazole initially 800 mg daily adjusted to 400 mg daily. The patient continued wound management with twice-daily dressing changes.
Nine months after DAIR, the patient remained on suppressive antifungal and antibacterial therapy. He continued to experience serous drainage from the wound, which greatly affected his quality of life. After discussion with his family and the orthopedic surgery team, he agreed to proceed with a 2-staged revision arthroplasty involving prosthetic explant and antibiotic spacer placement. However, the surgery was postponed due to findings of anemia (hemoglobin, 8.9 g/dL) and thrombocytopenia (platelet count, 73 x 103/λL). At the time of this report, the patient was being monitored closely with his multidisciplinary care team for the planned orthopedic procedure.
DISCUSSION
PJI is the most common cause of primary hip arthroplasty failure; however, fungal species only make up about 1% of PJIs.3,9-11 Patients are typically immunocompromised, undergoing antineoplastic therapies for malignancy, or have other comorbid conditions such as diabetes.12,13 C. glabrata presents a unique diagnostic and therapeutic challenge as it is not only rare but also notorious for its resistance to common antifungal agents. C. glabrata is known to develop multidrug resistance through the rapid accumulation of genomic mutations.14 Its propensity towards forming protective biofilm also arms it with intrinsic resistance to agents like fluconazole.15 Furthermore, based on a review of the available reports in the literature, C. glabrata PJIs are often insidious and present with symptoms closely mimicking those of bacterial PJIs, as it did in the patient in this case.16
Synovial fluid analysis, fungal cultures, and sensitivity testing are paramount for ensuring proper diagnosis for fungal PJI. The patient in this case was empirically treated with micafungin based on recommendations from the ID team. When the sensitivities results were reviewed, the same antifungal therapy was continued. Echinocandins have a favorable toxicity profile in long-term use, as well as efficacy against biofilm-producing organisms like C. glabrata.17,18
While there are a few cases citing DAIR as a feasible surgical strategy for treating fungal PJI, more recent studies have reported greater success with a 2-staged revision arthroplasty involving some combination of debridement, placement of antibiotic-loaded bone cement spacers, and partial or total exchange of the infected prosthetic joint.4,19-23 In this case, complete hardware replacement would have offered the patient the most favorable outlook for eliminating this fungal infection. However, given the patient’s advanced age, significant underlying comorbidities, and functional status, medical management with antifungal therapy and DAIR was favored.
Based on the discussion from the 6-month follow-up visit, the patient was experiencing progressive and persistent wound drainage and frequent dressing changes, highlighting the limitations of medical management for PJI in the setting of retained prosthesis. If the patient ultimately proceeds with a more invasive surgical intervention, another important consideration will be the likelihood of fungal PJI recurrence. At present, fungal PJI recurrence rates following antifungal and surgical treatment have been reported to range between 0% to 50%, which is too imprecise to be considered clinically useful.22-24
Given the ambiguity surrounding management guidelines and limited treatment options, it is crucial to emphasize the timeline of this patient’s clinical presentation and subsequent course of treatment. Upon presentation to the ED in late July, fungal PJI was considered less likely. Initial blood cultures from presentation were negative, which is common with PJIs. It was not until 5 days later that the left hip wound culture showed moderate growth of C. glabrata. Identifying a PJI is clinically challenging due to the lack of standardized diagnostic criteria. However, timely identification and diagnosis of fungal PJI with appropriate antifungal therapy, in patients with limited curative options due to comorbidities, can significantly improve quality of life and overall outcomes.25 Routine fungal and mycobacterial cultures are not currently recommended in PJI guidelines, but this case illustrates it is imperative in immunocompromised hosts.26
This case and the current paucity of similar cases in the literature stress the importance of clinicians publishing their experience in the management of fungal PJI. We strongly recommend that clinicians approach each suspected PJI with careful consideration of the patient’s unique risk factors, comorbidities, and goals of care, when deciding on a curative vs suppressive approach to therapy.
CONCLUSIONS
This case report highlights the importance of considering fungal pathogens for PJIs, especially in high-risk patients, the value of obtaining fungal cultures, the necessity of a multidisciplinary approach, the role of antifungal susceptibility testing, and consideration for the feasibility of a surgical intervention. It underscores the challenges in diagnosis and treatment of C. glabrata-associated PJI, emphasizing the importance of clinician experience-sharing in developing evidence-based management strategies. As the understanding of fungal PJI evolves, continued research and clinical data collection remain crucial for improving patient outcomes in the management of these complex cases.
- Osmon DR, Berbari EF, Berendt AR, et al. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):1-10. doi:10.1093/cid/cis966
- Eka A, Chen AF. Patient-related medical risk factors for periprosthetic joint infection of the hip and knee. Ann Transl Med. 2015;3(16):233. doi:10.3978/j.issn.2305-5839.2015.09.26
- Darouiche RO, Hamill RJ, Musher DM, Young EJ, Harris RL. Periprosthetic candidal infections following arthroplasty. Rev Infect Dis. 1989;11(1):89-96. doi:10.1093/clinids/11.1.89
- Koutserimpas C, Zervakis SG, Maraki S, et al. Non-albicans Candida prosthetic joint infections: a systematic review of treatment. World J Clin Cases. 2019;7(12):1430- 1443. doi:10.12998/wjcc.v7.i12.1430
- Fidel PL Jr, Vazquez JA, Sobel JD. Candida glabrata: review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans. Clin Microbiol Rev. 1999;12(1):80-96. doi:10.1128/CMR.12.1.80
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Lee YR, Kim HJ, Lee EJ, Sohn JW, Kim MJ, Yoon YK. Prosthetic joint infections caused by candida species: a systematic review and a case series. Mycopathologia. 2019;184(1):23-33. doi:10.1007/s11046-018-0286-1
- Herndon CL, Rowe TM, Metcalf RW, et al. Treatment outcomes of fungal periprosthetic joint infection. J Arthroplasty. 2023;38(11):2436-2440.e1. doi:10.1016/j.arth.2023.05.009
- Delaunay C, Hamadouche M, Girard J, Duhamel A; SoFCOT. What are the causes for failures of primary hip arthroplasties in France? Clin Orthop Relat Res. 2013;471(12): 3863-3869. doi:10.1007/s11999-013-2935-5
- Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91(1): 128-133. doi:10.2106/JBJS.H.00155
- Furnes O, Lie SA, Espehaug B, Vollset SE, Engesaeter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586. doi:10.1302/0301-620x.83b4.11223
- Gonzalez MR, Bedi ADS, Karczewski D, Lozano-Calderon SA. Treatment and outcomes of fungal prosthetic joint infections: a systematic review of 225 cases. J Arthroplasty. 2023;38(11):2464-2471.e1. doi:10.1016/j.arth.2023.05.003
- Gonzalez MR, Pretell-Mazzini J, Lozano-Calderon SA. Risk factors and management of prosthetic joint infections in megaprostheses-a review of the literature. Antibiotics (Basel). 2023;13(1):25. doi:10.3390/antibiotics13010025
- Biswas C, Chen SC, Halliday C, et al. Identification of genetic markers of resistance to echinocandins, azoles and 5-fluorocytosine in Candida glabrata by next-generation sequencing: a feasibility study. Clin Microbiol Infect. 2017;23(9):676.e7-676.e10. doi:10.1016/j.cmi.2017.03.014
- Hassan Y, Chew SY, Than LTL. Candida glabrata: pathogenicity and resistance mechanisms for adaptation and survival. J Fungi (Basel). 2021;7(8):667. doi:10.3390/jof7080667
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Pierce CG, Uppuluri P, Tristan AR, et al. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nat Protoc. 2008;3(9):1494-1500. doi:10.1038/nport.2008.141
- Koutserimpas C, Samonis G, Velivassakis E, Iliopoulou- Kosmadaki S, Kontakis G, Kofteridis DP. Candida glabrata prosthetic joint infection, successfully treated with anidulafungin: a case report and review of the literature. Mycoses. 2018;61(4):266-269. doi:10.1111/myc.12736
- Brooks DH, Pupparo F. Successful salvage of a primary total knee arthroplasty infected with Candida parapsilosis. J Arthroplasty. 1998;13(6):707-712. doi:10.1016/s0883-5403(98)80017-x
- Merrer J, Dupont B, Nieszkowska A, De Jonghe B, Outin H. Candida albicans prosthetic arthritis treated with fluconazole alone. J Infect. 2001;42(3):208-209. doi:10.1053/jinf.2001.0819
- Koutserimpas C, Naoum S, Alpantaki K, et al. Fungal prosthetic joint infection in revised knee arthroplasty: an orthopaedic surgeon’s nightmare. Diagnostics (Basel). 2022;12(7):1606. doi:10.3390/diagnostics12071606
- Gao Z, Li X, Du Y, Peng Y, Wu W, Zhou Y. Success rate of fungal peri-prosthetic joint infection treated by 2-stage revision and potential risk factors of treatment failure: a retrospective study. Med Sci Monit. 2018;24:5549-5557. doi:10.12659/MSM.909168
- Hwang BH, Yoon JY, Nam CH, et al. Fungal periprosthetic joint infection after primary total knee replacement. J Bone Joint Surg Br. 2012;94(5):656-659. doi:10.1302/0301-620X.94B5.28125
- Ueng SW, Lee CY, Hu CC, Hsieh PH, Chang Y. What is the success of treatment of hip and knee candidal periprosthetic joint infection? Clin Orthop Relat Res. 2013;471(9):3002-3009. doi:10.1007/s11999-013-3007-6
- Nodzo, Scott R. MD; Bauer, Thomas MD, PhD; Pottinger, et al. Conventional diagnostic challenges in periprosthetic joint infection. J Am Acad Orthop Surg. 2015;23 Suppl:S18-S25. doi:10.5435/JAAOS-D-14-00385
- American Academy of Orthopaedic Surgeons. Diagnosis and prevention of periprosthetic joint infections. March 11, 2019. Accessed February 5, 2025. https://www.aaos.org/pjicpg
- Osmon DR, Berbari EF, Berendt AR, et al. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):1-10. doi:10.1093/cid/cis966
- Eka A, Chen AF. Patient-related medical risk factors for periprosthetic joint infection of the hip and knee. Ann Transl Med. 2015;3(16):233. doi:10.3978/j.issn.2305-5839.2015.09.26
- Darouiche RO, Hamill RJ, Musher DM, Young EJ, Harris RL. Periprosthetic candidal infections following arthroplasty. Rev Infect Dis. 1989;11(1):89-96. doi:10.1093/clinids/11.1.89
- Koutserimpas C, Zervakis SG, Maraki S, et al. Non-albicans Candida prosthetic joint infections: a systematic review of treatment. World J Clin Cases. 2019;7(12):1430- 1443. doi:10.12998/wjcc.v7.i12.1430
- Fidel PL Jr, Vazquez JA, Sobel JD. Candida glabrata: review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans. Clin Microbiol Rev. 1999;12(1):80-96. doi:10.1128/CMR.12.1.80
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Lee YR, Kim HJ, Lee EJ, Sohn JW, Kim MJ, Yoon YK. Prosthetic joint infections caused by candida species: a systematic review and a case series. Mycopathologia. 2019;184(1):23-33. doi:10.1007/s11046-018-0286-1
- Herndon CL, Rowe TM, Metcalf RW, et al. Treatment outcomes of fungal periprosthetic joint infection. J Arthroplasty. 2023;38(11):2436-2440.e1. doi:10.1016/j.arth.2023.05.009
- Delaunay C, Hamadouche M, Girard J, Duhamel A; SoFCOT. What are the causes for failures of primary hip arthroplasties in France? Clin Orthop Relat Res. 2013;471(12): 3863-3869. doi:10.1007/s11999-013-2935-5
- Bozic KJ, Kurtz SM, Lau E, Ong K, Vail TP, Berry DJ. The epidemiology of revision total hip arthroplasty in the United States. J Bone Joint Surg Am. 2009;91(1): 128-133. doi:10.2106/JBJS.H.00155
- Furnes O, Lie SA, Espehaug B, Vollset SE, Engesaeter LB, Havelin LI. Hip disease and the prognosis of total hip replacements. A review of 53,698 primary total hip replacements reported to the Norwegian Arthroplasty Register 1987-99. J Bone Joint Surg Br. 2001;83(4):579-586. doi:10.1302/0301-620x.83b4.11223
- Gonzalez MR, Bedi ADS, Karczewski D, Lozano-Calderon SA. Treatment and outcomes of fungal prosthetic joint infections: a systematic review of 225 cases. J Arthroplasty. 2023;38(11):2464-2471.e1. doi:10.1016/j.arth.2023.05.003
- Gonzalez MR, Pretell-Mazzini J, Lozano-Calderon SA. Risk factors and management of prosthetic joint infections in megaprostheses-a review of the literature. Antibiotics (Basel). 2023;13(1):25. doi:10.3390/antibiotics13010025
- Biswas C, Chen SC, Halliday C, et al. Identification of genetic markers of resistance to echinocandins, azoles and 5-fluorocytosine in Candida glabrata by next-generation sequencing: a feasibility study. Clin Microbiol Infect. 2017;23(9):676.e7-676.e10. doi:10.1016/j.cmi.2017.03.014
- Hassan Y, Chew SY, Than LTL. Candida glabrata: pathogenicity and resistance mechanisms for adaptation and survival. J Fungi (Basel). 2021;7(8):667. doi:10.3390/jof7080667
- Aboltins C, Daffy J, Choong P, Stanley P. Current concepts in the management of prosthetic joint infection. Intern Med J. 2014;44(9):834-840. doi:10.1111/imj.12510
- Pierce CG, Uppuluri P, Tristan AR, et al. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nat Protoc. 2008;3(9):1494-1500. doi:10.1038/nport.2008.141
- Koutserimpas C, Samonis G, Velivassakis E, Iliopoulou- Kosmadaki S, Kontakis G, Kofteridis DP. Candida glabrata prosthetic joint infection, successfully treated with anidulafungin: a case report and review of the literature. Mycoses. 2018;61(4):266-269. doi:10.1111/myc.12736
- Brooks DH, Pupparo F. Successful salvage of a primary total knee arthroplasty infected with Candida parapsilosis. J Arthroplasty. 1998;13(6):707-712. doi:10.1016/s0883-5403(98)80017-x
- Merrer J, Dupont B, Nieszkowska A, De Jonghe B, Outin H. Candida albicans prosthetic arthritis treated with fluconazole alone. J Infect. 2001;42(3):208-209. doi:10.1053/jinf.2001.0819
- Koutserimpas C, Naoum S, Alpantaki K, et al. Fungal prosthetic joint infection in revised knee arthroplasty: an orthopaedic surgeon’s nightmare. Diagnostics (Basel). 2022;12(7):1606. doi:10.3390/diagnostics12071606
- Gao Z, Li X, Du Y, Peng Y, Wu W, Zhou Y. Success rate of fungal peri-prosthetic joint infection treated by 2-stage revision and potential risk factors of treatment failure: a retrospective study. Med Sci Monit. 2018;24:5549-5557. doi:10.12659/MSM.909168
- Hwang BH, Yoon JY, Nam CH, et al. Fungal periprosthetic joint infection after primary total knee replacement. J Bone Joint Surg Br. 2012;94(5):656-659. doi:10.1302/0301-620X.94B5.28125
- Ueng SW, Lee CY, Hu CC, Hsieh PH, Chang Y. What is the success of treatment of hip and knee candidal periprosthetic joint infection? Clin Orthop Relat Res. 2013;471(9):3002-3009. doi:10.1007/s11999-013-3007-6
- Nodzo, Scott R. MD; Bauer, Thomas MD, PhD; Pottinger, et al. Conventional diagnostic challenges in periprosthetic joint infection. J Am Acad Orthop Surg. 2015;23 Suppl:S18-S25. doi:10.5435/JAAOS-D-14-00385
- American Academy of Orthopaedic Surgeons. Diagnosis and prevention of periprosthetic joint infections. March 11, 2019. Accessed February 5, 2025. https://www.aaos.org/pjicpg
A Candida Glabrata-Associated Prosthetic Joint Infection: Case Report and Literature Review
A Candida Glabrata-Associated Prosthetic Joint Infection: Case Report and Literature Review
Sports Injuries of the Hip in Primary Care
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.
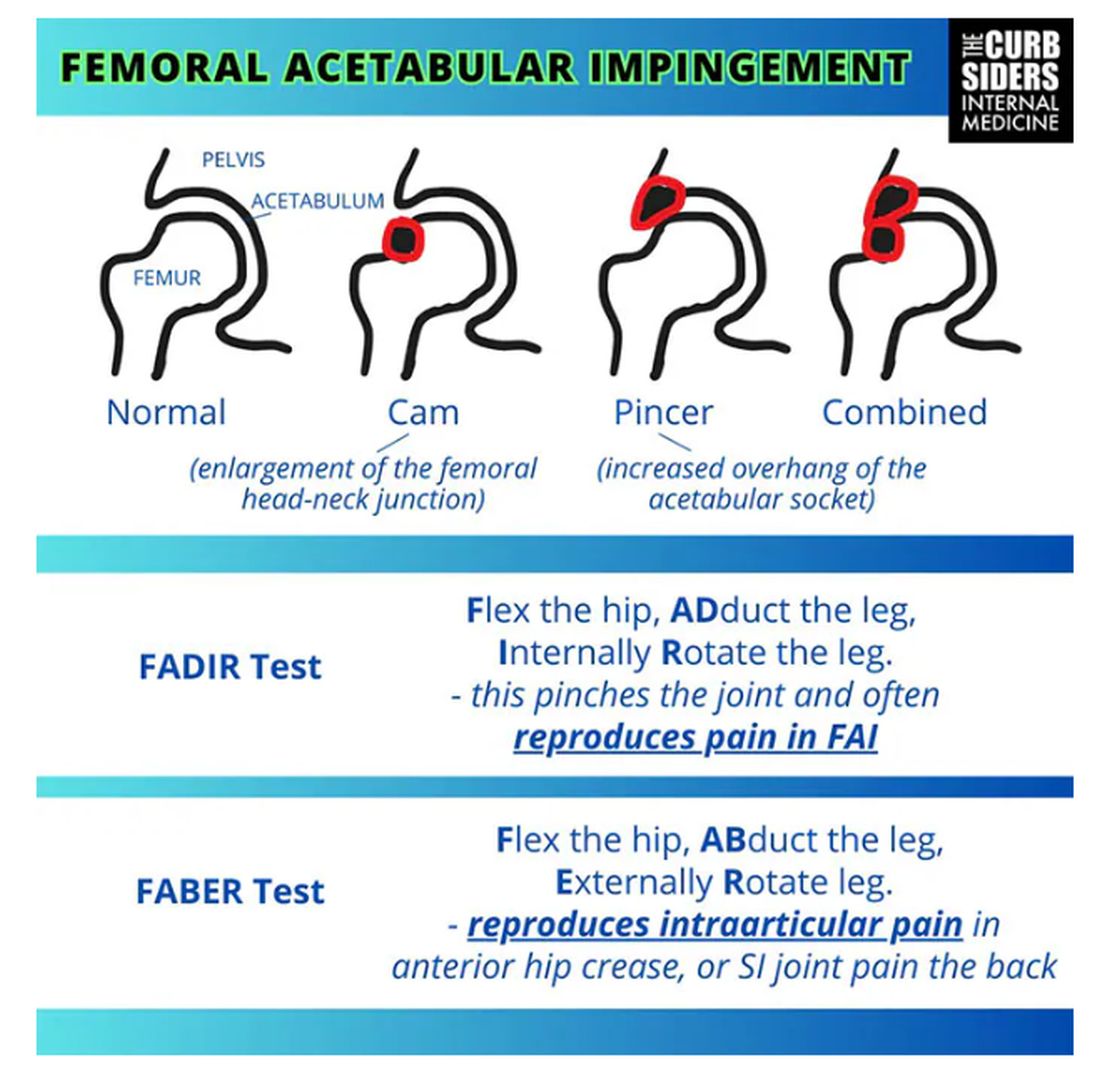
Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.

Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. I’m Dr Matthew Frank Watto, here with my great friend and America’s primary care physician, Dr Paul Nelson Williams. Paul, how are you feeling about sports injuries?
Paul N. Williams, MD: I’m feeling great, Matt.
Watto: You had a sports injury of the hip. Maybe that’s an overshare, Paul, but we talked about it on a podcast with Dr Carlin Senter (part 1 and part 2).
Williams: I think I’ve shared more than my hip injury, for sure.
Watto: Whenever a patient presented with hip pain, I used to pray it was trochanteric bursitis, which now I know is not really the right thing to think about. Intra-articular hip pain presents as anterior hip pain, usually in the crease of the hip. Depending on the patient’s age and history, the differential for that type of pain includes iliopsoas tendonitis, FAI syndrome, a labral tear, a bone stress injury of the femoral neck, or osteoarthritis.
So, what exactly is FAI and how might we diagnose it?
Williams: FAI is what the cool kids call femoral acetabular impingement, and it’s exactly what it sounds like.

Something is pinching or impinging upon the joint itself and preventing full range of motion. This is a ball-and-socket joint, so it should have tremendous range of motion, able to move in all planes. If it’s impinged, then pain will occur with certain movements. There’s a cam type, which is characterized by enlargement of the femoral head neck junction, or a pincer type, which has more to do with overhang of the acetabulum, and it can also be mixed. In any case, impingement upon the patient’s full range of motion results in pain.
You evaluate this with a couple of tests — the FABER and the FADIR.
The FABER is flexion, abduction, and external rotation, and the FADIR is flexion, adduction, and internal rotation. If you elicit anterior pain with either of those tests, it’s probably one of the intra-articular pathologies, although it is hard to know for sure which one it is because these tests are fairly sensitive but not very specific.
Watto: You can get x-rays to help with the diagnosis. You would order two views of the hip: an AP of the pelvis, which is just a straight-on shot to look for arthritis or fracture. Is there a healthy joint line there? The second is the Dunn view, in which the hip is flexed 90 degrees and abducted about 20 degrees. You are looking for fracture or impingement. You can diagnose FAI based on that view, and you might be able to diagnose a hip stress injury or osteoarthritis.
Unfortunately, you’re not going to see a labral tear, but Dr Senter said that both FAI and labral tears are treated the same way, with physical therapy. Patients with FAI who aren’t getting better might end up going for surgery, so at some point I would refer them to orthopedic surgery. But I feel much more comfortable now diagnosing these conditions with these tests.
Let’s talk a little bit about trochanteric pain syndrome. I used to think it was all bursitis. Why is that not correct?
Williams: It’s nice of you to feign ignorance for the purpose of education. It used to be thought of as bursitis, but these days we know it is probably more likely a tendinopathy.

Trochanteric pain syndrome was formerly known as trochanteric bursitis, but the bursa is not typically involved. Trochanteric pain syndrome is a tendinopathy of the surrounding structures: the gluteus medius, the iliotibial band, and the tensor fascia latae. The way these structures relate looks a bit like the face of a clock, as you can see on the infographic. In general, you manage this condition the same way you do with bursitis — physical therapy. You can also give corticosteroid injections. Physical therapy is probably more durable in terms of pain relief and functionality, but in the short term, corticosteroids might provide some degree of analgesia as well.
Watto: The last thing we wanted to mention is bone stress injury, which can occur in high-mileage runners (20 miles or more per week). Patients with bone stress injury need to rest, usually non‒weight bearing, for a period of time.

Treatment of a bone stress fracture depends on which side it’s on (top or bottom). If it’s on the top of the femoral neck (the tension side), it has to be fixed. If it’s on the compression side (the bottom side of the femoral neck), it might be able to be managed conservatively, but many patients are going to need surgery. This is a big deal. But it’s a spectrum; in some cases the bone is merely irritated and unhappy, without a break in the cortex. Those patients might not need surgery.
In patients with a fracture of the femoral neck — especially younger, healthier patients — you should think about getting a bone density test and screening for relative energy deficiency in sport. This used to be called the female athlete triad, which includes disrupted menstrual cycles, being underweight, and fracture. We should be screening patients, asking them in a nonjudgmental way about their relationship with food, to make sure they are getting an appropriate number of calories.
They are actually in an energy deficit. They’re not eating enough to maintain a healthy body with so much activity.
Williams: If you’re interested in this topic, you should refer to the full podcast with Dr Senter which is chock-full of helpful information.
Dr Watto, Clinical Assistant Professor, Department of Medicine, Perelman School of Medicine at University of Pennsylvania; Internist, Department of Medicine, Hospital Medicine Section, Pennsylvania Hospital, Philadelphia, Pennsylvania, has disclosed no relevant financial relationships. Dr Williams, Associate Professor of Clinical Medicine, Department of General Internal Medicine, Lewis Katz School of Medicine; Staff Physician, Department of General Internal Medicine, Temple Internal Medicine Associates, Philadelphia, Pennsylvania, disclosed ties with The Curbsiders.
A version of this article appeared on Medscape.com.
Using GLP-1s to Meet BMI Goal for Orthopedic Surgery
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
The woman, in severe pain from hip and knee osteoarthritis, was confined to a wheelchair and had been told that would likely be for life. To qualify for hip replacement surgery, she needed to lose 100 pounds, a seemingly impossible goal. But she wanted to try.
“We tried a couple of medicines — oral medicines off-label — topiramate, phentermine,” said Leslie Golden, MD, MPH, DABM, a family medicine physician and obesity medicine specialist in Watertown, Wisconsin, 42 miles northeast of Madison.
They weren’t enough. But then Golden turned to glucagon-like peptide 1 (GLP-1) receptor agonists, and they delivered.
“She did lose a significant amount of weight and was able to get the hip replacement,” said Golden.
It took a couple of years. However, seeing her walk into her office, rather than wheel in, “is still one of the joys of my practice,” Golden said. “She’s so grateful. She felt everyone else had written her off.”
As she told Golden: “If I fell and broke my leg today, they would take me to surgery without concern.”
Because her hip replacement was viewed as a nonemergency procedure, the accepted threshold for elective safe surgery was a body mass index (BMI) < 40. That BMI cutoff can vary from provider to provider and medical facility to medical facility but is often required for other surgeries as well, including kidney and lung transplants, gender-affirming surgery, bariatric surgery, hernia surgery, and in vitro fertilization procedures.
She worked with Rajit Chakravarty, MD, an adult reconstructive surgeon who practices in Watertown and nearby Madison, to oversee the weight loss.
High BMIs & Surgery Issues
High BMIs have long been linked with postsurgery complications, poor wound healing, and other issues, although some research now is questioning some of those associations. Even so, surgeons have long stressed weight loss for their patients with obesity before orthopedic and other procedures.
These days, surgeons are more likely to need to have that talk. In the last decade, the age-adjusted prevalence of severe obesity — a BMI of ≥ 40 — has increased from 7.7% to 9.7% of US adults. The number of joint replacements is also rising — more than 700,000 total knee arthroplasty (TKA) and more than 450,000 total hip arthroplasty (THA), according to the American Academy of Orthopaedic Surgeons. As the population ages, those numbers are expected to increase.
Making the GLP-1 Choice
GLP-1s aren’t the only choice, of course. But they’re often more effective, as Golden found, than other medications. And when his patients with obesity are offered bariatric surgery or GLP-1s, “people definitely want to avoid the bariatric surgery,” Chakravarty said.
With the Food and Drug Administration (FDA) approval of semaglutide (Wegovy) in June 2021 for chronic weight management and then tirzepatide (Zepbound) in November 2023, interest has boomed, he said, among his surgery candidates with a high BMI.
The FDA approved Wegovy based on clinical trials, including one in which participants lost an average of 12.4% of initial body weight compared with those on placebo. It approved Zepbound based on clinical trials, including one in which those on Zepbound lost an average of 18% of their body weight, compared with those on placebo.
The wheelchair-bound woman, now 65, began with a BMI of 63, Golden said. She negotiated a cutoff of 45 with the surgeon and got the go-ahead. Currently, her BMI is 36 as she stayed on the medications.
Beyond the benefit of GLP-1s helping patients meet the BMI cutoff, some research finds fewer postoperative infections and readmissions with their use. This study found the medications did lower both, and another found reduced readmissions and complications.
Growing Partnerships, Increasing Success
Helping patients lose weight isn’t just about lowering the BMI, Chakravarty pointed out. The aim is to improve nutritional health — to teach patients how to eat healthfully for their needs, in turn improving other health barometers. Referring them to an obesity medicine physician helps to meet those goals.
When Daniel Wiznia, MD, a Yale Medicine orthopedic surgeon and codirector of the Avascular Necrosis Program, has a patient who must delay a TKA or THA until they meet a BMI cutoff, he refers that patient to the Yale Medicine Center for Weight Management, New Haven, Connecticut, to learn about weight loss, including the options of anti-obesity medications or bariatric surgery.
Taking the GLP-1s can be a game changer, according to Wiznia and John Morton, MD, MPH, FACS, FASMBS, Yale’s medical director of Bariatric Surgery and professor and vice chair of surgery, who is a physician-director of the center. The program includes other options, such as bariatric surgery, and emphasizes diet and other lifestyle measures. GLP-1s give about a 15% weight loss, Morton said, compared with bariatric surgery providing up to 30%.
Sarah Stombaugh, MD, a family medicine and obesity medicine physician in Charlottesville, Virginia, often gets referrals from two orthopedic surgeons in her community. One recent patient in her early 60s had a BMI of 43.2, too high to qualify for the TKA she needed. On GLP-1s, the initial goal was to decrease a weight of 244 to 225, bringing the BMI to 39.9. The woman did that, then kept losing before her surgery was scheduled, getting to a weight of 210 or a BMI of 37 and staying there for 3 months before the surgery.
She had the TKA, and 5 months out, she is doing well, Stombaugh said. “We do medical weight loss primarily with the GLP-1s because they’re simply the best, the most effective,” Stombaugh said. She does occasionally use oral medications such as naltrexone/bupropion (Contrave).
Stombaugh sees the collaborating trend as still evolving. When she attends obesity medicine conferences, not all her colleagues report they are partnering with surgeons. But she predicts the practice will increase, saying the popularization of what she terms the more effective GLP-1 medications Wegovy and Zepbound is driving it. Partnering with the surgeon requires a conversation at the beginning, when the referral is made, about goals. After that, she sees her patient monthly and sends progress notes to the surgeon.
Golden collaborates with three orthopedic groups in her area, primarily for knee and hip surgeries, but has also helped patients meet the BMI cutoff before spine-related surgeries. She is helping a lung transplant patient now. She has seen several patients who must meet BMI requirements before starting in vitro fertilization, due to the need for conscious sedation for egg retrieval. She has had a few patients who had to meet a BMI cutoff for nonemergency hernia repair.
Insurance Issues
Insurance remains an issue for the pricey medications. “Only about a third of patients are routinely covered with insurance,” Morton said.
However, it’s improving, he said. Golden also finds about a third of private payers cover the medication but tries to use manufacturers’ coupons to help defray the costs (from about $1000 or $1400 to about $500 a month). She has sometimes gotten enough samples to get patients to their BMI goal
Morton consulted for Novo Nordisk, Eli Lilly, Olympus, Teleflex, and Johnson & Johnson.
A version of this article appeared on Medscape.com.
VA Awards Grants to Support Adaptive Sports
The US Department of Veterans Affairs (VA) is awarding $15.9 million in grants to fund adaptive sports, recreational activities, and equine therapy for > 15,000 veterans and service members living with disabilities.
Marine Corps veteran Jataya Taylor — who competed in wheelchair fencing at the 2024 Paralympics — experienced mental health symptoms until she began participating in adaptive sports through an organization supported by the VA Adaptive Sports Grant Program.
“Getting involved in adaptive sports was a saving grace for me,” Taylor said. “Participating in these programs got me on the bike to start with, then got me climbing, and eventually it became an important part of my mental health to participate. I found my people. I found my new network of friends.”
Adaptive sports, which are customized to fit the needs of veterans with disabilities, include paralympic sports, archery, cycling, skiing, hunting, rock climbing, and sky diving. Mike Gooler, another Marine Corps veteran, praised the Adaptive Sports Center’s facilities in Crested Butte, Colorado, calling it “nothing short of amazing.”
“[S]ki therapy has been instrumental in helping me navigate through my experiences and injuries,” Gooler said. “Skiing provides me with sense of freedom and empowerment … and having my family by my side, witnessing my progress and sharing the joy of skiing, was truly special.”
The grant program is facilitated and managed by the National Veterans Sports Programs and Special Events Office and will provide grants to 91 national, regional, and community-based programs for fiscal year 2024 across all 50 states, the District of Columbia, Guam, and Puerto Rico.
“These grants give veterans life-changing opportunities,” Secretary of VA Denis McDonough said. “We know adaptive sports and recreational activities can be transformational for veterans living with disabilities, improving their overall physical and mental health, and also giving them important community with fellow heroes who served.”
Information about the awardees and details of the program are available at www.va.gov/adaptivesports and on Facebook at Sports4Vets.
The US Department of Veterans Affairs (VA) is awarding $15.9 million in grants to fund adaptive sports, recreational activities, and equine therapy for > 15,000 veterans and service members living with disabilities.
Marine Corps veteran Jataya Taylor — who competed in wheelchair fencing at the 2024 Paralympics — experienced mental health symptoms until she began participating in adaptive sports through an organization supported by the VA Adaptive Sports Grant Program.
“Getting involved in adaptive sports was a saving grace for me,” Taylor said. “Participating in these programs got me on the bike to start with, then got me climbing, and eventually it became an important part of my mental health to participate. I found my people. I found my new network of friends.”
Adaptive sports, which are customized to fit the needs of veterans with disabilities, include paralympic sports, archery, cycling, skiing, hunting, rock climbing, and sky diving. Mike Gooler, another Marine Corps veteran, praised the Adaptive Sports Center’s facilities in Crested Butte, Colorado, calling it “nothing short of amazing.”
“[S]ki therapy has been instrumental in helping me navigate through my experiences and injuries,” Gooler said. “Skiing provides me with sense of freedom and empowerment … and having my family by my side, witnessing my progress and sharing the joy of skiing, was truly special.”
The grant program is facilitated and managed by the National Veterans Sports Programs and Special Events Office and will provide grants to 91 national, regional, and community-based programs for fiscal year 2024 across all 50 states, the District of Columbia, Guam, and Puerto Rico.
“These grants give veterans life-changing opportunities,” Secretary of VA Denis McDonough said. “We know adaptive sports and recreational activities can be transformational for veterans living with disabilities, improving their overall physical and mental health, and also giving them important community with fellow heroes who served.”
Information about the awardees and details of the program are available at www.va.gov/adaptivesports and on Facebook at Sports4Vets.
The US Department of Veterans Affairs (VA) is awarding $15.9 million in grants to fund adaptive sports, recreational activities, and equine therapy for > 15,000 veterans and service members living with disabilities.
Marine Corps veteran Jataya Taylor — who competed in wheelchair fencing at the 2024 Paralympics — experienced mental health symptoms until she began participating in adaptive sports through an organization supported by the VA Adaptive Sports Grant Program.
“Getting involved in adaptive sports was a saving grace for me,” Taylor said. “Participating in these programs got me on the bike to start with, then got me climbing, and eventually it became an important part of my mental health to participate. I found my people. I found my new network of friends.”
Adaptive sports, which are customized to fit the needs of veterans with disabilities, include paralympic sports, archery, cycling, skiing, hunting, rock climbing, and sky diving. Mike Gooler, another Marine Corps veteran, praised the Adaptive Sports Center’s facilities in Crested Butte, Colorado, calling it “nothing short of amazing.”
“[S]ki therapy has been instrumental in helping me navigate through my experiences and injuries,” Gooler said. “Skiing provides me with sense of freedom and empowerment … and having my family by my side, witnessing my progress and sharing the joy of skiing, was truly special.”
The grant program is facilitated and managed by the National Veterans Sports Programs and Special Events Office and will provide grants to 91 national, regional, and community-based programs for fiscal year 2024 across all 50 states, the District of Columbia, Guam, and Puerto Rico.
“These grants give veterans life-changing opportunities,” Secretary of VA Denis McDonough said. “We know adaptive sports and recreational activities can be transformational for veterans living with disabilities, improving their overall physical and mental health, and also giving them important community with fellow heroes who served.”
Information about the awardees and details of the program are available at www.va.gov/adaptivesports and on Facebook at Sports4Vets.
Total Hip Replacement Superior to Exercise Therapy for Improving Hip Osteoarthritis Pain and Function
For people with severe symptomatic hip osteoarthritis, total hip replacement (THR) alleviates hip pain and improves function much more effectively than a resistance training program supervised by a physiotherapist, according to the results of a randomized controlled clinical trial.
In the PROHIP study, the mean increases in Oxford Hip Scores from baseline to 6 months were 15.9 points for THR and 4.5 points for resistance training. The 11.4-point difference in scores was both statistically and clinically significant, the study’s investigators reported in The New England Journal of Medicine.
“Our results are clear: Surgery is superior to exercise in patients who have hip osteoarthritis and indication for surgery, and now we have finally proven that with the highest level of evidence,” corresponding author Thomas Frydendal, PT, PhD, MSc, told this news organization.
Frydendal, who was involved in the study while working on his PhD at University Hospital of Southern Denmark – Lillebaelt Hospital, Vejle, Denmark, the primary center for the trial, is now a postdoctoral researcher at the Department of Clinical Medicine, Aarhus University, and Department of Orthopedic Surgery, Aarhus University Hospital.
“We believe that our findings are pretty robust,” Frydendal added. “I think if someone in the world conducts a trial similar to ours, they will find fairly close or consistent findings, no matter what type of exercise they choose.”
The PROHIP Study
THR is routinely recommended for the management of severe hip osteoarthritis, but since there are no clinical trial data on the effectiveness of this procedure as compared with first-line treatment such as resistance training, the PROHIP study was conceived.
The trial was conducted at four Danish orthopedic centers and designed as a superiority study, the hypothesis being that THR would be better at alleviating self-reported hip pain and improving hip function than resistance training.
Of a possible 1474 individuals with a clinical suspicion of hip osteoarthritis, 791 were deemed eligible for inclusion in the trial. Inclusion criteria were being aged 50 years or older and having an indication for THR based on the presence of hip pain and clinical and radiographic findings.
However, the majority (86%) declined to enter the study, with almost half (43%) deciding to have a THR and enroll in a parallel observational cohort. This meant that only 110 (14%) individuals agreed to participate and underwent randomization, which does limit the study’s generalizability, the PROHIP investigators acknowledged.
Design and Study Population
The change in Oxford Hip Score from baseline to 6 months was selected as the primary outcome measure based on the findings of a prior qualitative study. This 12-item, patient-reported outcome measure gives a score ranging from 0 to 48, with higher scores indicating less hip pain and better hip function. The estimated minimal clinically important difference is a change of 5 points.
After a baseline assessment, 53 of 109 individuals were randomly assigned to undergo THR and 56 to participate in the resistance training program. Overall, the mean age of participants was 67.6 years, and half were women. The average duration of hip pain was a median of 1.7 years.
The median time to receipt of the allocated treatment was 2.8 months in the THR group and 0.5 months in the resistance training group.
Those allocated to the THR group also underwent a “fast track” program that involved patient education, pain management, and early mobilization.
The resistance training group received 12 weeks of exercise supervised by a physiotherapist and then offered 12 weeks of additional exercise conducted on their own. The physiotherapist-supervised exercise sessions were held twice weekly and lasted for 1 hour. These started with a 10-minute warm-up on a stationary bike, followed by a standard set of resistance-based exercises that included a leg press, hip extension, hip flexion, and hip abduction.
‘Reassuring’ Results
In a comment, consultant orthopedic surgeon Antony Palmer, MA, BMBCh, DPhil, said: “It’s reassuring that patients with advanced symptomatic osteoarthritis do well with hip replacements.”
THR does of course come with the potential risk for complications, but “the rate of these is what you’d expect for that procedure,” Palmer said, who works for the Nuffield Orthopaedic Centre, Oxford University Hospital NHS Foundation Trust, and is a senior clinical research fellow at Oxford University in England.
In the THR arm, there was one case of prosthetic joint infection, one hip dislocation, two revision surgeries, one instance of foot drop, and one case of gastroesophageal reflux. Meanwhile, in the resistance training group, there was one hip dislocation, one pelvic fracture, one case of atrial fibrillation, and one urinary tract and renal infection.
Overall, any serious adverse event was reported in six (12%) of 48 patients in the THR arm vs five (9%) of 55 participants in the resistance training group, of which only one, occurring in the resistance training group, resulted in discontinuation of the program.
Resistance Training Role
A notable finding was that, at 6 months, five (9%) people assigned to the THR arm had not undergone surgery, and 12 (21%) people in the resistance training group had undergone a THR.
This could suggest two things, Palmer suggested in the interview. The first is that there could be a small proportion of people assigned to THR who may not need the operation and do well with exercise therapy. And, conversely, there may be those who would do well having the surgery without first going through the intermediate stage of physical therapy.
It’s a suggestion that “maybe we’ve got to refine that a bit better and identify the patients that really do benefit from physiotherapy and who might not need hip replacement as a result,” Palmer said.
Or in other words, “should all patients undergo a program of physiotherapy before considering surgery?” he added.
Authors’ View
The PROHIP investigators conclude: “These results support current recommendations for the management of hip osteoarthritis and may be used to inform and guide shared decision making in clinical practice.”
Moreover, the results “do not oppose the use of resistance training as initial treatment,” says the authors.
Frydendal highlighted in his interview that nearly three out of four of the patients reported not to have undertaken any type of supervised exercise before entry into the study, which is a first-line, guideline-recommended option.
“If a patient tells me, ‘I haven’t done any exercise previously,’ I’d recommend starting with completing a 6- to 12-week exercise program that is tailored to your individual needs and evaluate your symptoms afterward,” he said.
“But we should refer the patient if our first-line treatment does not offer any improvements in the patient’s symptoms, as surgery with total hip replacement is clearly a really good treatment option,” Frydendal said.
The study was funded by the Danish Rheumatism Association, among other independent bodies. Frydendal and Palmer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For people with severe symptomatic hip osteoarthritis, total hip replacement (THR) alleviates hip pain and improves function much more effectively than a resistance training program supervised by a physiotherapist, according to the results of a randomized controlled clinical trial.
In the PROHIP study, the mean increases in Oxford Hip Scores from baseline to 6 months were 15.9 points for THR and 4.5 points for resistance training. The 11.4-point difference in scores was both statistically and clinically significant, the study’s investigators reported in The New England Journal of Medicine.
“Our results are clear: Surgery is superior to exercise in patients who have hip osteoarthritis and indication for surgery, and now we have finally proven that with the highest level of evidence,” corresponding author Thomas Frydendal, PT, PhD, MSc, told this news organization.
Frydendal, who was involved in the study while working on his PhD at University Hospital of Southern Denmark – Lillebaelt Hospital, Vejle, Denmark, the primary center for the trial, is now a postdoctoral researcher at the Department of Clinical Medicine, Aarhus University, and Department of Orthopedic Surgery, Aarhus University Hospital.
“We believe that our findings are pretty robust,” Frydendal added. “I think if someone in the world conducts a trial similar to ours, they will find fairly close or consistent findings, no matter what type of exercise they choose.”
The PROHIP Study
THR is routinely recommended for the management of severe hip osteoarthritis, but since there are no clinical trial data on the effectiveness of this procedure as compared with first-line treatment such as resistance training, the PROHIP study was conceived.
The trial was conducted at four Danish orthopedic centers and designed as a superiority study, the hypothesis being that THR would be better at alleviating self-reported hip pain and improving hip function than resistance training.
Of a possible 1474 individuals with a clinical suspicion of hip osteoarthritis, 791 were deemed eligible for inclusion in the trial. Inclusion criteria were being aged 50 years or older and having an indication for THR based on the presence of hip pain and clinical and radiographic findings.
However, the majority (86%) declined to enter the study, with almost half (43%) deciding to have a THR and enroll in a parallel observational cohort. This meant that only 110 (14%) individuals agreed to participate and underwent randomization, which does limit the study’s generalizability, the PROHIP investigators acknowledged.
Design and Study Population
The change in Oxford Hip Score from baseline to 6 months was selected as the primary outcome measure based on the findings of a prior qualitative study. This 12-item, patient-reported outcome measure gives a score ranging from 0 to 48, with higher scores indicating less hip pain and better hip function. The estimated minimal clinically important difference is a change of 5 points.
After a baseline assessment, 53 of 109 individuals were randomly assigned to undergo THR and 56 to participate in the resistance training program. Overall, the mean age of participants was 67.6 years, and half were women. The average duration of hip pain was a median of 1.7 years.
The median time to receipt of the allocated treatment was 2.8 months in the THR group and 0.5 months in the resistance training group.
Those allocated to the THR group also underwent a “fast track” program that involved patient education, pain management, and early mobilization.
The resistance training group received 12 weeks of exercise supervised by a physiotherapist and then offered 12 weeks of additional exercise conducted on their own. The physiotherapist-supervised exercise sessions were held twice weekly and lasted for 1 hour. These started with a 10-minute warm-up on a stationary bike, followed by a standard set of resistance-based exercises that included a leg press, hip extension, hip flexion, and hip abduction.
‘Reassuring’ Results
In a comment, consultant orthopedic surgeon Antony Palmer, MA, BMBCh, DPhil, said: “It’s reassuring that patients with advanced symptomatic osteoarthritis do well with hip replacements.”
THR does of course come with the potential risk for complications, but “the rate of these is what you’d expect for that procedure,” Palmer said, who works for the Nuffield Orthopaedic Centre, Oxford University Hospital NHS Foundation Trust, and is a senior clinical research fellow at Oxford University in England.
In the THR arm, there was one case of prosthetic joint infection, one hip dislocation, two revision surgeries, one instance of foot drop, and one case of gastroesophageal reflux. Meanwhile, in the resistance training group, there was one hip dislocation, one pelvic fracture, one case of atrial fibrillation, and one urinary tract and renal infection.
Overall, any serious adverse event was reported in six (12%) of 48 patients in the THR arm vs five (9%) of 55 participants in the resistance training group, of which only one, occurring in the resistance training group, resulted in discontinuation of the program.
Resistance Training Role
A notable finding was that, at 6 months, five (9%) people assigned to the THR arm had not undergone surgery, and 12 (21%) people in the resistance training group had undergone a THR.
This could suggest two things, Palmer suggested in the interview. The first is that there could be a small proportion of people assigned to THR who may not need the operation and do well with exercise therapy. And, conversely, there may be those who would do well having the surgery without first going through the intermediate stage of physical therapy.
It’s a suggestion that “maybe we’ve got to refine that a bit better and identify the patients that really do benefit from physiotherapy and who might not need hip replacement as a result,” Palmer said.
Or in other words, “should all patients undergo a program of physiotherapy before considering surgery?” he added.
Authors’ View
The PROHIP investigators conclude: “These results support current recommendations for the management of hip osteoarthritis and may be used to inform and guide shared decision making in clinical practice.”
Moreover, the results “do not oppose the use of resistance training as initial treatment,” says the authors.
Frydendal highlighted in his interview that nearly three out of four of the patients reported not to have undertaken any type of supervised exercise before entry into the study, which is a first-line, guideline-recommended option.
“If a patient tells me, ‘I haven’t done any exercise previously,’ I’d recommend starting with completing a 6- to 12-week exercise program that is tailored to your individual needs and evaluate your symptoms afterward,” he said.
“But we should refer the patient if our first-line treatment does not offer any improvements in the patient’s symptoms, as surgery with total hip replacement is clearly a really good treatment option,” Frydendal said.
The study was funded by the Danish Rheumatism Association, among other independent bodies. Frydendal and Palmer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
For people with severe symptomatic hip osteoarthritis, total hip replacement (THR) alleviates hip pain and improves function much more effectively than a resistance training program supervised by a physiotherapist, according to the results of a randomized controlled clinical trial.
In the PROHIP study, the mean increases in Oxford Hip Scores from baseline to 6 months were 15.9 points for THR and 4.5 points for resistance training. The 11.4-point difference in scores was both statistically and clinically significant, the study’s investigators reported in The New England Journal of Medicine.
“Our results are clear: Surgery is superior to exercise in patients who have hip osteoarthritis and indication for surgery, and now we have finally proven that with the highest level of evidence,” corresponding author Thomas Frydendal, PT, PhD, MSc, told this news organization.
Frydendal, who was involved in the study while working on his PhD at University Hospital of Southern Denmark – Lillebaelt Hospital, Vejle, Denmark, the primary center for the trial, is now a postdoctoral researcher at the Department of Clinical Medicine, Aarhus University, and Department of Orthopedic Surgery, Aarhus University Hospital.
“We believe that our findings are pretty robust,” Frydendal added. “I think if someone in the world conducts a trial similar to ours, they will find fairly close or consistent findings, no matter what type of exercise they choose.”
The PROHIP Study
THR is routinely recommended for the management of severe hip osteoarthritis, but since there are no clinical trial data on the effectiveness of this procedure as compared with first-line treatment such as resistance training, the PROHIP study was conceived.
The trial was conducted at four Danish orthopedic centers and designed as a superiority study, the hypothesis being that THR would be better at alleviating self-reported hip pain and improving hip function than resistance training.
Of a possible 1474 individuals with a clinical suspicion of hip osteoarthritis, 791 were deemed eligible for inclusion in the trial. Inclusion criteria were being aged 50 years or older and having an indication for THR based on the presence of hip pain and clinical and radiographic findings.
However, the majority (86%) declined to enter the study, with almost half (43%) deciding to have a THR and enroll in a parallel observational cohort. This meant that only 110 (14%) individuals agreed to participate and underwent randomization, which does limit the study’s generalizability, the PROHIP investigators acknowledged.
Design and Study Population
The change in Oxford Hip Score from baseline to 6 months was selected as the primary outcome measure based on the findings of a prior qualitative study. This 12-item, patient-reported outcome measure gives a score ranging from 0 to 48, with higher scores indicating less hip pain and better hip function. The estimated minimal clinically important difference is a change of 5 points.
After a baseline assessment, 53 of 109 individuals were randomly assigned to undergo THR and 56 to participate in the resistance training program. Overall, the mean age of participants was 67.6 years, and half were women. The average duration of hip pain was a median of 1.7 years.
The median time to receipt of the allocated treatment was 2.8 months in the THR group and 0.5 months in the resistance training group.
Those allocated to the THR group also underwent a “fast track” program that involved patient education, pain management, and early mobilization.
The resistance training group received 12 weeks of exercise supervised by a physiotherapist and then offered 12 weeks of additional exercise conducted on their own. The physiotherapist-supervised exercise sessions were held twice weekly and lasted for 1 hour. These started with a 10-minute warm-up on a stationary bike, followed by a standard set of resistance-based exercises that included a leg press, hip extension, hip flexion, and hip abduction.
‘Reassuring’ Results
In a comment, consultant orthopedic surgeon Antony Palmer, MA, BMBCh, DPhil, said: “It’s reassuring that patients with advanced symptomatic osteoarthritis do well with hip replacements.”
THR does of course come with the potential risk for complications, but “the rate of these is what you’d expect for that procedure,” Palmer said, who works for the Nuffield Orthopaedic Centre, Oxford University Hospital NHS Foundation Trust, and is a senior clinical research fellow at Oxford University in England.
In the THR arm, there was one case of prosthetic joint infection, one hip dislocation, two revision surgeries, one instance of foot drop, and one case of gastroesophageal reflux. Meanwhile, in the resistance training group, there was one hip dislocation, one pelvic fracture, one case of atrial fibrillation, and one urinary tract and renal infection.
Overall, any serious adverse event was reported in six (12%) of 48 patients in the THR arm vs five (9%) of 55 participants in the resistance training group, of which only one, occurring in the resistance training group, resulted in discontinuation of the program.
Resistance Training Role
A notable finding was that, at 6 months, five (9%) people assigned to the THR arm had not undergone surgery, and 12 (21%) people in the resistance training group had undergone a THR.
This could suggest two things, Palmer suggested in the interview. The first is that there could be a small proportion of people assigned to THR who may not need the operation and do well with exercise therapy. And, conversely, there may be those who would do well having the surgery without first going through the intermediate stage of physical therapy.
It’s a suggestion that “maybe we’ve got to refine that a bit better and identify the patients that really do benefit from physiotherapy and who might not need hip replacement as a result,” Palmer said.
Or in other words, “should all patients undergo a program of physiotherapy before considering surgery?” he added.
Authors’ View
The PROHIP investigators conclude: “These results support current recommendations for the management of hip osteoarthritis and may be used to inform and guide shared decision making in clinical practice.”
Moreover, the results “do not oppose the use of resistance training as initial treatment,” says the authors.
Frydendal highlighted in his interview that nearly three out of four of the patients reported not to have undertaken any type of supervised exercise before entry into the study, which is a first-line, guideline-recommended option.
“If a patient tells me, ‘I haven’t done any exercise previously,’ I’d recommend starting with completing a 6- to 12-week exercise program that is tailored to your individual needs and evaluate your symptoms afterward,” he said.
“But we should refer the patient if our first-line treatment does not offer any improvements in the patient’s symptoms, as surgery with total hip replacement is clearly a really good treatment option,” Frydendal said.
The study was funded by the Danish Rheumatism Association, among other independent bodies. Frydendal and Palmer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Disc Degeneration in Chronic Low Back Pain: Can Stem Cells Help?
TOPLINE:
Allogeneic bone marrow–derived mesenchymal stromal cells (BM-MSCs) are safe but do not show efficacy in treating intervertebral disc degeneration (IDD) in patients with chronic low back pain.
METHODOLOGY:
- The RESPINE trial assessed the efficacy and safety of a single intradiscal injection of allogeneic BM-MSCs in the treatment of chronic low back pain caused by single-level IDD.
- Overall, 114 patients (mean age, 40.9 years; 35% women) with IDD-associated chronic low back pain that was persistent for 3 months or more despite conventional medical therapy and without previous surgery, were recruited across four European countries from April 2018 to April 2021 and randomly assigned to receive either intradiscal injections of allogeneic BM-MSCs (n = 58) or sham injections (n = 56).
- The first co-primary endpoint was the rate of response to BM-MSC injections at 12 months after treatment, defined as improvement of at least 20% or 20 mm in the Visual Analog Scale for pain or improvement of at least 20% in the Oswestry Disability Index for functional status.
- The secondary co-primary endpoint was structural efficacy, based on disc fluid content measured by quantitative T2 MRI between baseline and month 12.
TAKEAWAY:
- At 12 months post-intervention, 74% of patients in the BM-MSC group were classified as responders compared with 68.8% in the placebo group. However, the difference between the groups was not statistically significant.
- The probability of being a responder was higher in the BM-MSC group than in the sham group; however, the findings did not reach statistical significance.
- The average change in disc fluid content, indicative of disc regeneration, from baseline to 12 months was 37.9% in the BM-MSC group and 41.7% in the placebo group, with no significant difference between the groups.
- The incidence of adverse events and serious adverse events was not significantly different between the treatment groups.
IN PRACTICE:
“BM-MSC represents a promising opportunity for the biological treatment of IDD, but only high-quality randomized controlled trials, comparing it to standard care, can determine whether it is a truly effective alternative to spine fusion or disc replacement,” the authors wrote.
SOURCE:
The study was led by Yves-Marie Pers, MD, PhD, Clinical Immunology and Osteoarticular Diseases Therapeutic Unit, CHRU Lapeyronie, Montpellier, France. It was published online on October 11, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
MRI results were collected from only 55 patients across both trial arms, which may have affected the statistical power of the findings. Although patients were monitored for up to 24 months, the long-term efficacy and safety of BM-MSC therapy for IDD may not have been fully captured. Selection bias could not be excluded because of the difficulty in accurately identifying patients with chronic low back pain caused by single-level IDD.
DISCLOSURES:
The study was funded by the European Union’s Horizon 2020 Research and Innovation Programme. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Allogeneic bone marrow–derived mesenchymal stromal cells (BM-MSCs) are safe but do not show efficacy in treating intervertebral disc degeneration (IDD) in patients with chronic low back pain.
METHODOLOGY:
- The RESPINE trial assessed the efficacy and safety of a single intradiscal injection of allogeneic BM-MSCs in the treatment of chronic low back pain caused by single-level IDD.
- Overall, 114 patients (mean age, 40.9 years; 35% women) with IDD-associated chronic low back pain that was persistent for 3 months or more despite conventional medical therapy and without previous surgery, were recruited across four European countries from April 2018 to April 2021 and randomly assigned to receive either intradiscal injections of allogeneic BM-MSCs (n = 58) or sham injections (n = 56).
- The first co-primary endpoint was the rate of response to BM-MSC injections at 12 months after treatment, defined as improvement of at least 20% or 20 mm in the Visual Analog Scale for pain or improvement of at least 20% in the Oswestry Disability Index for functional status.
- The secondary co-primary endpoint was structural efficacy, based on disc fluid content measured by quantitative T2 MRI between baseline and month 12.
TAKEAWAY:
- At 12 months post-intervention, 74% of patients in the BM-MSC group were classified as responders compared with 68.8% in the placebo group. However, the difference between the groups was not statistically significant.
- The probability of being a responder was higher in the BM-MSC group than in the sham group; however, the findings did not reach statistical significance.
- The average change in disc fluid content, indicative of disc regeneration, from baseline to 12 months was 37.9% in the BM-MSC group and 41.7% in the placebo group, with no significant difference between the groups.
- The incidence of adverse events and serious adverse events was not significantly different between the treatment groups.
IN PRACTICE:
“BM-MSC represents a promising opportunity for the biological treatment of IDD, but only high-quality randomized controlled trials, comparing it to standard care, can determine whether it is a truly effective alternative to spine fusion or disc replacement,” the authors wrote.
SOURCE:
The study was led by Yves-Marie Pers, MD, PhD, Clinical Immunology and Osteoarticular Diseases Therapeutic Unit, CHRU Lapeyronie, Montpellier, France. It was published online on October 11, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
MRI results were collected from only 55 patients across both trial arms, which may have affected the statistical power of the findings. Although patients were monitored for up to 24 months, the long-term efficacy and safety of BM-MSC therapy for IDD may not have been fully captured. Selection bias could not be excluded because of the difficulty in accurately identifying patients with chronic low back pain caused by single-level IDD.
DISCLOSURES:
The study was funded by the European Union’s Horizon 2020 Research and Innovation Programme. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Allogeneic bone marrow–derived mesenchymal stromal cells (BM-MSCs) are safe but do not show efficacy in treating intervertebral disc degeneration (IDD) in patients with chronic low back pain.
METHODOLOGY:
- The RESPINE trial assessed the efficacy and safety of a single intradiscal injection of allogeneic BM-MSCs in the treatment of chronic low back pain caused by single-level IDD.
- Overall, 114 patients (mean age, 40.9 years; 35% women) with IDD-associated chronic low back pain that was persistent for 3 months or more despite conventional medical therapy and without previous surgery, were recruited across four European countries from April 2018 to April 2021 and randomly assigned to receive either intradiscal injections of allogeneic BM-MSCs (n = 58) or sham injections (n = 56).
- The first co-primary endpoint was the rate of response to BM-MSC injections at 12 months after treatment, defined as improvement of at least 20% or 20 mm in the Visual Analog Scale for pain or improvement of at least 20% in the Oswestry Disability Index for functional status.
- The secondary co-primary endpoint was structural efficacy, based on disc fluid content measured by quantitative T2 MRI between baseline and month 12.
TAKEAWAY:
- At 12 months post-intervention, 74% of patients in the BM-MSC group were classified as responders compared with 68.8% in the placebo group. However, the difference between the groups was not statistically significant.
- The probability of being a responder was higher in the BM-MSC group than in the sham group; however, the findings did not reach statistical significance.
- The average change in disc fluid content, indicative of disc regeneration, from baseline to 12 months was 37.9% in the BM-MSC group and 41.7% in the placebo group, with no significant difference between the groups.
- The incidence of adverse events and serious adverse events was not significantly different between the treatment groups.
IN PRACTICE:
“BM-MSC represents a promising opportunity for the biological treatment of IDD, but only high-quality randomized controlled trials, comparing it to standard care, can determine whether it is a truly effective alternative to spine fusion or disc replacement,” the authors wrote.
SOURCE:
The study was led by Yves-Marie Pers, MD, PhD, Clinical Immunology and Osteoarticular Diseases Therapeutic Unit, CHRU Lapeyronie, Montpellier, France. It was published online on October 11, 2024, in Annals of the Rheumatic Diseases.
LIMITATIONS:
MRI results were collected from only 55 patients across both trial arms, which may have affected the statistical power of the findings. Although patients were monitored for up to 24 months, the long-term efficacy and safety of BM-MSC therapy for IDD may not have been fully captured. Selection bias could not be excluded because of the difficulty in accurately identifying patients with chronic low back pain caused by single-level IDD.
DISCLOSURES:
The study was funded by the European Union’s Horizon 2020 Research and Innovation Programme. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Arthroscopy Doesn’t Delay Total Knee Replacement in Knee Osteoarthritis
TOPLINE:
Adding arthroscopic surgery to nonoperative management neither delays nor accelerates the timing of total knee arthroplasty (TKA) in patients with knee osteoarthritis (OA).
METHODOLOGY:
- Some case series show that arthroscopic surgery for knee OA may delay more invasive procedures, such as TKA or osteotomy, while longitudinal cohort studies often contradict this. Current OA guidelines are yet to address this issue.
- This secondary analysis of a randomized trial compared the long-term incidence of TKA in 178 patients (mean age, 59 years; 64.3% women) with knee OA who were referred for potential arthroscopic surgery at a tertiary care center in Canada.
- The patients received nonoperative care with or without additional arthroscopic surgery.
- Patients in the arthroscopic surgery group had specific knee procedures (resection of degenerative knee tissues) along with nonoperative management (physical therapy plus medications as required), while the control group received nonoperative management alone.
- The primary outcome was TKA on the knee being studied, and the secondary outcome was TKA or osteotomy on either knee.
TAKEAWAY:
- During a median follow-up of 13.8 years, 37.6% of patients underwent TKA, with comparable proportions of patients in the arthroscopic surgery and control groups undergoing TKA (adjusted hazard ratio [aHR], 0.85; 95% CI, 0.52-1.40).
- The rates of TKA or osteotomy on either knee were similar in both groups (aHR, 0.91; 95% CI, 0.59-1.41).
- A time-stratified analysis done for 0-5 years, 5-10 years, and beyond 10 years of follow-up also showed a consistent interpretation.
- When patients with crossover to arthroscopic surgery during the follow-up were included, the results remained similar for both the primary (HR, 0.88; 95% CI, 0.53-1.44) and secondary (HR, 1.08; 95% CI, 0.69-1.68) outcomes.
IN PRACTICE:
“Our study findings do not support the use of arthroscopic surgery for OA of the knee.” “Arthroscopic surgery does not provide additional benefit to nonoperative management for improving pain, stiffness, and function and is likely not cost-effective at 2 years of follow-up,” the authors wrote.
SOURCE:
This study was led by Trevor B. Birmingham, PhD, Fowler Kennedy Sport Medicine Clinic, University of Western Ontario, London, Ontario, Canada. It was published online in JAMA Network Open
LIMITATIONS:
The study was designed to assess differences in 2-year patient-reported outcomes rather than long-term TKA incidence. Factors influencing decisions to undergo TKA or osteotomy were not considered. Moreover, the effects observed in this study should be evaluated considering the estimated confidence intervals.
DISCLOSURES:
This study was supported by the ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. Some authors declared consulting, performing contracted services, or receiving grant funding, royalties, and nonfinancial support from various sources.
A version of this article appeared on Medscape.com.
TOPLINE:
Adding arthroscopic surgery to nonoperative management neither delays nor accelerates the timing of total knee arthroplasty (TKA) in patients with knee osteoarthritis (OA).
METHODOLOGY:
- Some case series show that arthroscopic surgery for knee OA may delay more invasive procedures, such as TKA or osteotomy, while longitudinal cohort studies often contradict this. Current OA guidelines are yet to address this issue.
- This secondary analysis of a randomized trial compared the long-term incidence of TKA in 178 patients (mean age, 59 years; 64.3% women) with knee OA who were referred for potential arthroscopic surgery at a tertiary care center in Canada.
- The patients received nonoperative care with or without additional arthroscopic surgery.
- Patients in the arthroscopic surgery group had specific knee procedures (resection of degenerative knee tissues) along with nonoperative management (physical therapy plus medications as required), while the control group received nonoperative management alone.
- The primary outcome was TKA on the knee being studied, and the secondary outcome was TKA or osteotomy on either knee.
TAKEAWAY:
- During a median follow-up of 13.8 years, 37.6% of patients underwent TKA, with comparable proportions of patients in the arthroscopic surgery and control groups undergoing TKA (adjusted hazard ratio [aHR], 0.85; 95% CI, 0.52-1.40).
- The rates of TKA or osteotomy on either knee were similar in both groups (aHR, 0.91; 95% CI, 0.59-1.41).
- A time-stratified analysis done for 0-5 years, 5-10 years, and beyond 10 years of follow-up also showed a consistent interpretation.
- When patients with crossover to arthroscopic surgery during the follow-up were included, the results remained similar for both the primary (HR, 0.88; 95% CI, 0.53-1.44) and secondary (HR, 1.08; 95% CI, 0.69-1.68) outcomes.
IN PRACTICE:
“Our study findings do not support the use of arthroscopic surgery for OA of the knee.” “Arthroscopic surgery does not provide additional benefit to nonoperative management for improving pain, stiffness, and function and is likely not cost-effective at 2 years of follow-up,” the authors wrote.
SOURCE:
This study was led by Trevor B. Birmingham, PhD, Fowler Kennedy Sport Medicine Clinic, University of Western Ontario, London, Ontario, Canada. It was published online in JAMA Network Open
LIMITATIONS:
The study was designed to assess differences in 2-year patient-reported outcomes rather than long-term TKA incidence. Factors influencing decisions to undergo TKA or osteotomy were not considered. Moreover, the effects observed in this study should be evaluated considering the estimated confidence intervals.
DISCLOSURES:
This study was supported by the ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. Some authors declared consulting, performing contracted services, or receiving grant funding, royalties, and nonfinancial support from various sources.
A version of this article appeared on Medscape.com.
TOPLINE:
Adding arthroscopic surgery to nonoperative management neither delays nor accelerates the timing of total knee arthroplasty (TKA) in patients with knee osteoarthritis (OA).
METHODOLOGY:
- Some case series show that arthroscopic surgery for knee OA may delay more invasive procedures, such as TKA or osteotomy, while longitudinal cohort studies often contradict this. Current OA guidelines are yet to address this issue.
- This secondary analysis of a randomized trial compared the long-term incidence of TKA in 178 patients (mean age, 59 years; 64.3% women) with knee OA who were referred for potential arthroscopic surgery at a tertiary care center in Canada.
- The patients received nonoperative care with or without additional arthroscopic surgery.
- Patients in the arthroscopic surgery group had specific knee procedures (resection of degenerative knee tissues) along with nonoperative management (physical therapy plus medications as required), while the control group received nonoperative management alone.
- The primary outcome was TKA on the knee being studied, and the secondary outcome was TKA or osteotomy on either knee.
TAKEAWAY:
- During a median follow-up of 13.8 years, 37.6% of patients underwent TKA, with comparable proportions of patients in the arthroscopic surgery and control groups undergoing TKA (adjusted hazard ratio [aHR], 0.85; 95% CI, 0.52-1.40).
- The rates of TKA or osteotomy on either knee were similar in both groups (aHR, 0.91; 95% CI, 0.59-1.41).
- A time-stratified analysis done for 0-5 years, 5-10 years, and beyond 10 years of follow-up also showed a consistent interpretation.
- When patients with crossover to arthroscopic surgery during the follow-up were included, the results remained similar for both the primary (HR, 0.88; 95% CI, 0.53-1.44) and secondary (HR, 1.08; 95% CI, 0.69-1.68) outcomes.
IN PRACTICE:
“Our study findings do not support the use of arthroscopic surgery for OA of the knee.” “Arthroscopic surgery does not provide additional benefit to nonoperative management for improving pain, stiffness, and function and is likely not cost-effective at 2 years of follow-up,” the authors wrote.
SOURCE:
This study was led by Trevor B. Birmingham, PhD, Fowler Kennedy Sport Medicine Clinic, University of Western Ontario, London, Ontario, Canada. It was published online in JAMA Network Open
LIMITATIONS:
The study was designed to assess differences in 2-year patient-reported outcomes rather than long-term TKA incidence. Factors influencing decisions to undergo TKA or osteotomy were not considered. Moreover, the effects observed in this study should be evaluated considering the estimated confidence intervals.
DISCLOSURES:
This study was supported by the ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. Some authors declared consulting, performing contracted services, or receiving grant funding, royalties, and nonfinancial support from various sources.
A version of this article appeared on Medscape.com.