User login
Menopause accelerates RA functional decline
Rheumatoid arthritis gets worse after menopause, likely because of lower hormone levels, according to a review of 8,189 women in the National Data Bank for Rheumatic Diseases, published recently in Rheumatology.
The investigators compared scores on the Health Assessment Questionnaire (HAQ) between 2,005 premenopausal women with a mean age of 39.7 years; 611 women transitioning through menopause with a mean age of 50.7 years, and 5,573 postmenopausal women with a mean age of 62.3 years. As participants in the data bank, the women completed a questionnaire at regular intervals that included the HAQ, which is a 3-point measure of functional status, with 0 meaning no disability and 3 severe disability. They had all been diagnosed with rheumatoid arthritis prior to menopause.
“Women with RA have better functional status prior to menopause, even after controlling for covariates,” and after menopause, functional decline worsens and accelerates, said investigators led by Elizabeth Mollard, PhD, a nurse practitioner at the University of Nebraska Medical Center, Lincoln. The findings were “robust even after adjustment for other significant factors.”
The team also found that functional decline was less in women who had a longer reproductive life; had ever been pregnant; or had ever used hormone replacement therapy (HRT).
The findings support the notion that hormone exposure plays a role in RA severity, at least in women. It’s well known that RA activity trails off when women are pregnant, but increases after delivery, when hormone levels are returning to baseline. It’s also known that women who go through menopause early are at greater risk for developing RA. Longer reproductive life, pregnancy, and HRT use, meanwhile, all increase women’s hormonal exposure and were protective in the study.
“Women have changes in disease development and progression surrounding reproductive and hormonal events. ... Our results suggest further study on hormonal involvement in functional decline in women with RA,” the investigators said.
Menopausal stage was determined by survey response. Pregnant women and those with hysterectomies were excluded from the study, as were those who went through menopause before the age of 40 years, and those over the age of 55 who had not reported a menstruation cessation date.
There was no external funding for the work. Dr. Mollard had no disclosures.
SOURCE: Mollard E et. al. Rheumatology. 2018 Jan 29. doi: 10.1093/rheumatology/kex526
Rheumatoid arthritis gets worse after menopause, likely because of lower hormone levels, according to a review of 8,189 women in the National Data Bank for Rheumatic Diseases, published recently in Rheumatology.
The investigators compared scores on the Health Assessment Questionnaire (HAQ) between 2,005 premenopausal women with a mean age of 39.7 years; 611 women transitioning through menopause with a mean age of 50.7 years, and 5,573 postmenopausal women with a mean age of 62.3 years. As participants in the data bank, the women completed a questionnaire at regular intervals that included the HAQ, which is a 3-point measure of functional status, with 0 meaning no disability and 3 severe disability. They had all been diagnosed with rheumatoid arthritis prior to menopause.
“Women with RA have better functional status prior to menopause, even after controlling for covariates,” and after menopause, functional decline worsens and accelerates, said investigators led by Elizabeth Mollard, PhD, a nurse practitioner at the University of Nebraska Medical Center, Lincoln. The findings were “robust even after adjustment for other significant factors.”
The team also found that functional decline was less in women who had a longer reproductive life; had ever been pregnant; or had ever used hormone replacement therapy (HRT).
The findings support the notion that hormone exposure plays a role in RA severity, at least in women. It’s well known that RA activity trails off when women are pregnant, but increases after delivery, when hormone levels are returning to baseline. It’s also known that women who go through menopause early are at greater risk for developing RA. Longer reproductive life, pregnancy, and HRT use, meanwhile, all increase women’s hormonal exposure and were protective in the study.
“Women have changes in disease development and progression surrounding reproductive and hormonal events. ... Our results suggest further study on hormonal involvement in functional decline in women with RA,” the investigators said.
Menopausal stage was determined by survey response. Pregnant women and those with hysterectomies were excluded from the study, as were those who went through menopause before the age of 40 years, and those over the age of 55 who had not reported a menstruation cessation date.
There was no external funding for the work. Dr. Mollard had no disclosures.
SOURCE: Mollard E et. al. Rheumatology. 2018 Jan 29. doi: 10.1093/rheumatology/kex526
Rheumatoid arthritis gets worse after menopause, likely because of lower hormone levels, according to a review of 8,189 women in the National Data Bank for Rheumatic Diseases, published recently in Rheumatology.
The investigators compared scores on the Health Assessment Questionnaire (HAQ) between 2,005 premenopausal women with a mean age of 39.7 years; 611 women transitioning through menopause with a mean age of 50.7 years, and 5,573 postmenopausal women with a mean age of 62.3 years. As participants in the data bank, the women completed a questionnaire at regular intervals that included the HAQ, which is a 3-point measure of functional status, with 0 meaning no disability and 3 severe disability. They had all been diagnosed with rheumatoid arthritis prior to menopause.
“Women with RA have better functional status prior to menopause, even after controlling for covariates,” and after menopause, functional decline worsens and accelerates, said investigators led by Elizabeth Mollard, PhD, a nurse practitioner at the University of Nebraska Medical Center, Lincoln. The findings were “robust even after adjustment for other significant factors.”
The team also found that functional decline was less in women who had a longer reproductive life; had ever been pregnant; or had ever used hormone replacement therapy (HRT).
The findings support the notion that hormone exposure plays a role in RA severity, at least in women. It’s well known that RA activity trails off when women are pregnant, but increases after delivery, when hormone levels are returning to baseline. It’s also known that women who go through menopause early are at greater risk for developing RA. Longer reproductive life, pregnancy, and HRT use, meanwhile, all increase women’s hormonal exposure and were protective in the study.
“Women have changes in disease development and progression surrounding reproductive and hormonal events. ... Our results suggest further study on hormonal involvement in functional decline in women with RA,” the investigators said.
Menopausal stage was determined by survey response. Pregnant women and those with hysterectomies were excluded from the study, as were those who went through menopause before the age of 40 years, and those over the age of 55 who had not reported a menstruation cessation date.
There was no external funding for the work. Dr. Mollard had no disclosures.
SOURCE: Mollard E et. al. Rheumatology. 2018 Jan 29. doi: 10.1093/rheumatology/kex526
FROM RHEUMATOLOGY
Key clinical point:
Major finding: HAQ scores were 0.68 points higher in postmenopausal women, compared with premenopausal women of the same age.
Study details: Review of 8,189 women in the National Data Bank for Rheumatic Diseases.
Disclosures: There was no external funding for the work. The lead investigator had no disclosures.
Source: Mollard E et. al. Rheumatology. 2018 Jan 29. doi: 10.1093/rheumatology/kex526
JAK inhibitors look good for severe alopecia areata treatment
said Lucy Yichu Liu, MD, and Brett Andrew King, MD, of Yale University, New Haven, Conn.
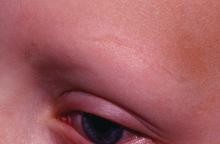
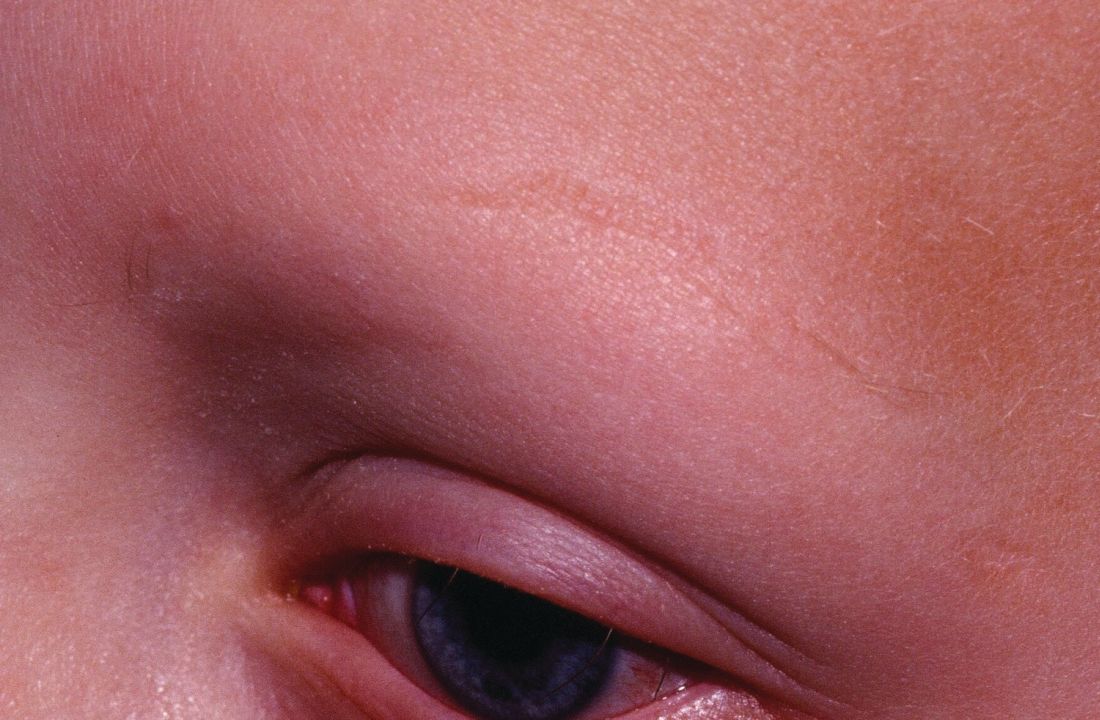
Standard medical therapies for alopecia areata – usually topical or injected corticosteroids and allergic contact sensitization – are not very effective for severe disease, particularly alopecia totalis and alopecia universalis. The Janus kinase (JAK) pathway recently has been suggested as a target for treatment.
Dr. Liu and Dr. King reviewed several studies, including a retrospective cohort study of 13 patients aged 12-17 years, in which 7 patients had 100% hair loss and 6 had 20%-70% scalp hair loss. The adolescents were treated with the JAK1/3 inhibitor tofacitinib citrate 5 mg twice daily for 2-16 months (median, 5 months). That led to 93% median improvement in Severity of Alopecia Tool (SALT) score (range, 1%-100%) from baseline. Nine patients experienced hair regrowth. There were mild adverse effects, such as upper respiratory infections and headaches.
In a retrospective cohort study of 90 adults taking tofacitinib at a dosage of 5-10 mg twice daily for 4 months or longer with or without prednisone (300 mg once monthly for three doses), patients were divided into those who were more or less likely to respond based on duration of disease. Of 65 patients with alopecia totalis, or alopecia universalis that had lasted 10 years or less, or alopecia areata, 77% had some hair regrowth; 58% had more than 50% improvement from baseline, and 20% achieved full regrowth of hair, Dr. Liu and Dr. King reported in the Journal of Investigative Dermatology Symposium Proceedings.
“Given the finding in adults that complete scalp hair loss for more than 10 years is less likely to respond to treatment, there may be merit to pursuing treatment, even if only intermittently, in adolescents or even younger patients with stable, severe alopecia areata, to prevent irreversible hair loss in the future,” they wrote.
A patient with alopecia universalis achieved partial scalp hair regrowth and complete eyebrow regrowth with compounded ruxolitinib, a topical JAK inhibitor, according to a 2016 case report. Dr. Liu and Dr. King reported that clinical trials with topical JAK inhibitors, including topical tofacitinib and topical ruxolitinib, currently are ongoing.
SOURCE: Liu LY et al. J Investig Dermatol Symp Proc. 2018 Jan. doi: 10.1016/j.jisp.2017.10.003.
said Lucy Yichu Liu, MD, and Brett Andrew King, MD, of Yale University, New Haven, Conn.
Standard medical therapies for alopecia areata – usually topical or injected corticosteroids and allergic contact sensitization – are not very effective for severe disease, particularly alopecia totalis and alopecia universalis. The Janus kinase (JAK) pathway recently has been suggested as a target for treatment.
Dr. Liu and Dr. King reviewed several studies, including a retrospective cohort study of 13 patients aged 12-17 years, in which 7 patients had 100% hair loss and 6 had 20%-70% scalp hair loss. The adolescents were treated with the JAK1/3 inhibitor tofacitinib citrate 5 mg twice daily for 2-16 months (median, 5 months). That led to 93% median improvement in Severity of Alopecia Tool (SALT) score (range, 1%-100%) from baseline. Nine patients experienced hair regrowth. There were mild adverse effects, such as upper respiratory infections and headaches.
In a retrospective cohort study of 90 adults taking tofacitinib at a dosage of 5-10 mg twice daily for 4 months or longer with or without prednisone (300 mg once monthly for three doses), patients were divided into those who were more or less likely to respond based on duration of disease. Of 65 patients with alopecia totalis, or alopecia universalis that had lasted 10 years or less, or alopecia areata, 77% had some hair regrowth; 58% had more than 50% improvement from baseline, and 20% achieved full regrowth of hair, Dr. Liu and Dr. King reported in the Journal of Investigative Dermatology Symposium Proceedings.
“Given the finding in adults that complete scalp hair loss for more than 10 years is less likely to respond to treatment, there may be merit to pursuing treatment, even if only intermittently, in adolescents or even younger patients with stable, severe alopecia areata, to prevent irreversible hair loss in the future,” they wrote.
A patient with alopecia universalis achieved partial scalp hair regrowth and complete eyebrow regrowth with compounded ruxolitinib, a topical JAK inhibitor, according to a 2016 case report. Dr. Liu and Dr. King reported that clinical trials with topical JAK inhibitors, including topical tofacitinib and topical ruxolitinib, currently are ongoing.
SOURCE: Liu LY et al. J Investig Dermatol Symp Proc. 2018 Jan. doi: 10.1016/j.jisp.2017.10.003.
said Lucy Yichu Liu, MD, and Brett Andrew King, MD, of Yale University, New Haven, Conn.
Standard medical therapies for alopecia areata – usually topical or injected corticosteroids and allergic contact sensitization – are not very effective for severe disease, particularly alopecia totalis and alopecia universalis. The Janus kinase (JAK) pathway recently has been suggested as a target for treatment.
Dr. Liu and Dr. King reviewed several studies, including a retrospective cohort study of 13 patients aged 12-17 years, in which 7 patients had 100% hair loss and 6 had 20%-70% scalp hair loss. The adolescents were treated with the JAK1/3 inhibitor tofacitinib citrate 5 mg twice daily for 2-16 months (median, 5 months). That led to 93% median improvement in Severity of Alopecia Tool (SALT) score (range, 1%-100%) from baseline. Nine patients experienced hair regrowth. There were mild adverse effects, such as upper respiratory infections and headaches.
In a retrospective cohort study of 90 adults taking tofacitinib at a dosage of 5-10 mg twice daily for 4 months or longer with or without prednisone (300 mg once monthly for three doses), patients were divided into those who were more or less likely to respond based on duration of disease. Of 65 patients with alopecia totalis, or alopecia universalis that had lasted 10 years or less, or alopecia areata, 77% had some hair regrowth; 58% had more than 50% improvement from baseline, and 20% achieved full regrowth of hair, Dr. Liu and Dr. King reported in the Journal of Investigative Dermatology Symposium Proceedings.
“Given the finding in adults that complete scalp hair loss for more than 10 years is less likely to respond to treatment, there may be merit to pursuing treatment, even if only intermittently, in adolescents or even younger patients with stable, severe alopecia areata, to prevent irreversible hair loss in the future,” they wrote.
A patient with alopecia universalis achieved partial scalp hair regrowth and complete eyebrow regrowth with compounded ruxolitinib, a topical JAK inhibitor, according to a 2016 case report. Dr. Liu and Dr. King reported that clinical trials with topical JAK inhibitors, including topical tofacitinib and topical ruxolitinib, currently are ongoing.
SOURCE: Liu LY et al. J Investig Dermatol Symp Proc. 2018 Jan. doi: 10.1016/j.jisp.2017.10.003.
FROM JOURNAL OF INVESTIGATIVE DERMATOLOGY SYMPOSIUM PROCEEDINGS
Checkpoint inhibitors look safe in rheumatology patients
People with rheumatologic diseases and cancer appear to be at no higher risk of having an adverse event or disease flare if they receive checkpoint inhibitor therapy, compared with the general population, experience from the Mayo Clinic suggests.
In a brief report published in Arthritis and Rheumatology, a team from the Mayo Clinic in Rochester, Minn., reported on 16 patients with rheumatologic diseases who received cancer immunotherapy. They found that only a minority experienced a flare of their disease or another immune-related event.
The rate of severe immune-related adverse effects (IRAEs) with a single immune checkpoint inhibitor (ICI) has been reported to be less than 2% among the average population. However, less is known about patients with underlying rheumatologic disease, largely because initial trials of ICIs had excluded patients with autoimmune diseases for fear the treatment would induce a disease flare, the researchers noted.
Small studies have suggested that people with inflammatory arthritis or connective tissue diseases have higher rates of IRAEs with immunotherapy, but it is unclear how often these events represented flares of their disease or new autoimmune events, and whether the events had any predictive significance for cancer survival.
In this study, researchers performed a retrospective review of medical records and identified 16 patients with rheumatologic diseases who had received checkpoint inhibitor therapy at the Mayo Clinic between 2011 and 2016.
The most common rheumatologic diseases among the 16 patients were rheumatoid arthritis, polymyalgia rheumatica, Sjögren’s syndrome, and systemic lupus erythematosus, and the most common cancers were malignant melanoma, pulmonary malignancies, and non-Hodgkin lymphoma. Seven of the patients were receiving immunosuppressive therapy or glucocorticoids for their rheumatologic disease upon initiation of a checkpoint inhibitor.
Ten patients had received a prior disease-modifying antirheumatic drug, but only two patients were still taking this at the time of ICI initiation.
Results showed that six of the patients (38%) had an IRAE or flare of their rheumatologic disease, two were graded as mild. All of the patients responded well to glucocorticoids and discontinuation of therapy. The most common event was colitis and just one patient had a flare of rheumatologic disease.
“This is consistent with what is currently known about the management of IRAEs,” the research team wrote. “This study adds further support to the emerging notion that the rate of IRAEs is not necessarily higher in this group compared to the general population.”
The type and severity of rheumatologic disease may play an important role in both the risk of disease flare and IRAEs, a factor that they were unable to assess in the current study, the researchers wrote.
“Further large, prospective studies are needed to address the link between the type, severity, and concurrent rheumatologic disease activity on the risk of flare and IRAE. It is possible that patients with more severe or active disease are at higher risk for these complications,” they wrote.
While patients in the study did not appear to have significantly increased incidence or severity of adverse effects, the research team advised that “treatment decisions must factor in clinical judgement.”
They noted that some studies had proposed predictive biomarkers, pretreatment workup, and monitoring, but this advice was based on a small body of evidence.
“Larger, prospective studies will be necessary to validate these findings and establish evidence-based guidelines for appropriate identification and rating of the rheumatologic IRAEs as well as their treatment, such that patients can continue to receive potentially life-saving cancer treatments,” they wrote.
One of the researchers reported advisory board membership with Bristol-Myers Squibb.
SOURCE: Richter M et al. Arthritis Rheumatol. 2018 Jan 24. doi: 10.1002/art.40397.
People with rheumatologic diseases and cancer appear to be at no higher risk of having an adverse event or disease flare if they receive checkpoint inhibitor therapy, compared with the general population, experience from the Mayo Clinic suggests.
In a brief report published in Arthritis and Rheumatology, a team from the Mayo Clinic in Rochester, Minn., reported on 16 patients with rheumatologic diseases who received cancer immunotherapy. They found that only a minority experienced a flare of their disease or another immune-related event.
The rate of severe immune-related adverse effects (IRAEs) with a single immune checkpoint inhibitor (ICI) has been reported to be less than 2% among the average population. However, less is known about patients with underlying rheumatologic disease, largely because initial trials of ICIs had excluded patients with autoimmune diseases for fear the treatment would induce a disease flare, the researchers noted.
Small studies have suggested that people with inflammatory arthritis or connective tissue diseases have higher rates of IRAEs with immunotherapy, but it is unclear how often these events represented flares of their disease or new autoimmune events, and whether the events had any predictive significance for cancer survival.
In this study, researchers performed a retrospective review of medical records and identified 16 patients with rheumatologic diseases who had received checkpoint inhibitor therapy at the Mayo Clinic between 2011 and 2016.
The most common rheumatologic diseases among the 16 patients were rheumatoid arthritis, polymyalgia rheumatica, Sjögren’s syndrome, and systemic lupus erythematosus, and the most common cancers were malignant melanoma, pulmonary malignancies, and non-Hodgkin lymphoma. Seven of the patients were receiving immunosuppressive therapy or glucocorticoids for their rheumatologic disease upon initiation of a checkpoint inhibitor.
Ten patients had received a prior disease-modifying antirheumatic drug, but only two patients were still taking this at the time of ICI initiation.
Results showed that six of the patients (38%) had an IRAE or flare of their rheumatologic disease, two were graded as mild. All of the patients responded well to glucocorticoids and discontinuation of therapy. The most common event was colitis and just one patient had a flare of rheumatologic disease.
“This is consistent with what is currently known about the management of IRAEs,” the research team wrote. “This study adds further support to the emerging notion that the rate of IRAEs is not necessarily higher in this group compared to the general population.”
The type and severity of rheumatologic disease may play an important role in both the risk of disease flare and IRAEs, a factor that they were unable to assess in the current study, the researchers wrote.
“Further large, prospective studies are needed to address the link between the type, severity, and concurrent rheumatologic disease activity on the risk of flare and IRAE. It is possible that patients with more severe or active disease are at higher risk for these complications,” they wrote.
While patients in the study did not appear to have significantly increased incidence or severity of adverse effects, the research team advised that “treatment decisions must factor in clinical judgement.”
They noted that some studies had proposed predictive biomarkers, pretreatment workup, and monitoring, but this advice was based on a small body of evidence.
“Larger, prospective studies will be necessary to validate these findings and establish evidence-based guidelines for appropriate identification and rating of the rheumatologic IRAEs as well as their treatment, such that patients can continue to receive potentially life-saving cancer treatments,” they wrote.
One of the researchers reported advisory board membership with Bristol-Myers Squibb.
SOURCE: Richter M et al. Arthritis Rheumatol. 2018 Jan 24. doi: 10.1002/art.40397.
People with rheumatologic diseases and cancer appear to be at no higher risk of having an adverse event or disease flare if they receive checkpoint inhibitor therapy, compared with the general population, experience from the Mayo Clinic suggests.
In a brief report published in Arthritis and Rheumatology, a team from the Mayo Clinic in Rochester, Minn., reported on 16 patients with rheumatologic diseases who received cancer immunotherapy. They found that only a minority experienced a flare of their disease or another immune-related event.
The rate of severe immune-related adverse effects (IRAEs) with a single immune checkpoint inhibitor (ICI) has been reported to be less than 2% among the average population. However, less is known about patients with underlying rheumatologic disease, largely because initial trials of ICIs had excluded patients with autoimmune diseases for fear the treatment would induce a disease flare, the researchers noted.
Small studies have suggested that people with inflammatory arthritis or connective tissue diseases have higher rates of IRAEs with immunotherapy, but it is unclear how often these events represented flares of their disease or new autoimmune events, and whether the events had any predictive significance for cancer survival.
In this study, researchers performed a retrospective review of medical records and identified 16 patients with rheumatologic diseases who had received checkpoint inhibitor therapy at the Mayo Clinic between 2011 and 2016.
The most common rheumatologic diseases among the 16 patients were rheumatoid arthritis, polymyalgia rheumatica, Sjögren’s syndrome, and systemic lupus erythematosus, and the most common cancers were malignant melanoma, pulmonary malignancies, and non-Hodgkin lymphoma. Seven of the patients were receiving immunosuppressive therapy or glucocorticoids for their rheumatologic disease upon initiation of a checkpoint inhibitor.
Ten patients had received a prior disease-modifying antirheumatic drug, but only two patients were still taking this at the time of ICI initiation.
Results showed that six of the patients (38%) had an IRAE or flare of their rheumatologic disease, two were graded as mild. All of the patients responded well to glucocorticoids and discontinuation of therapy. The most common event was colitis and just one patient had a flare of rheumatologic disease.
“This is consistent with what is currently known about the management of IRAEs,” the research team wrote. “This study adds further support to the emerging notion that the rate of IRAEs is not necessarily higher in this group compared to the general population.”
The type and severity of rheumatologic disease may play an important role in both the risk of disease flare and IRAEs, a factor that they were unable to assess in the current study, the researchers wrote.
“Further large, prospective studies are needed to address the link between the type, severity, and concurrent rheumatologic disease activity on the risk of flare and IRAE. It is possible that patients with more severe or active disease are at higher risk for these complications,” they wrote.
While patients in the study did not appear to have significantly increased incidence or severity of adverse effects, the research team advised that “treatment decisions must factor in clinical judgement.”
They noted that some studies had proposed predictive biomarkers, pretreatment workup, and monitoring, but this advice was based on a small body of evidence.
“Larger, prospective studies will be necessary to validate these findings and establish evidence-based guidelines for appropriate identification and rating of the rheumatologic IRAEs as well as their treatment, such that patients can continue to receive potentially life-saving cancer treatments,” they wrote.
One of the researchers reported advisory board membership with Bristol-Myers Squibb.
SOURCE: Richter M et al. Arthritis Rheumatol. 2018 Jan 24. doi: 10.1002/art.40397.
FROM ARTHRITIS AND RHEUMATOLOGY
Key clinical point:
Major finding: Six of 16 patients (38%) with rheumatologic disease and cancer had an IRAE or flare of their rheumatologic disease.
Study details: A single-center, retrospective records review to identify patients with rheumatologic diseases who had received checkpoint inhibitor therapy at Mayo Clinic between 2011 and 2016.
Disclosures: One of the authors reported advisory board membership with Bristol-Myers Squibb.
Source: Richter M et al. Arthritis Rheumatol. 2018 Jan 24. doi: 10.1002/art.40397.
Lower residual RA activity after initial ETN-MTX, better remission chance
There are four factors at baseline in adults with moderately active rheumatoid arthritis that predict those who are most likely to achieve remission with full-dose combo etanercept-methotrexate (ETN-MTX) induction treatment, said Josef S. Smolen, MD, of the University of Vienna, and his associates.
The original PRESERVE study found that after 604 adults with moderately active rheumatoid arthritis achieved low disease activity after 36 weeks of full-dose etanercept (50 mg once weekly) plus methotrexate, subsequent full-dose or reduced-dose (25 mg once weekly) ETN-MTX combos were better at maintaining remission than was methotrexate alone. At the time of the original study, a Disease Activity Score in 28 joints (DAS28) less than 2.6 was considered to denote remission in practice and clinical trials; now that the American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) Boolean definition of remission has been published, a DAS28 less than 2.6 no longer indicates remission according to ACR/EULAR criteria, but rather indicates minimal disease activity, the investigators explained.
This study aimed to determine predictors of remission, the researchers said in their article published in Arthritis Research & Therapy.
Those predictors at baseline are young age; body mass index less than 30 kg/m2; lower Health Assessment Questionnaire scores; and lower disease activity – as measured by DAS28, Simplified Disease Activity Index, and Clinical Disease Activity Index.
If the predictors are favorable and the patients have an “early, strong, and durable response to induction therapy, they are most likely to experience a sustained response after biologic tapering or withdrawal,” Dr. Smolen and his associates said.
In other words, the investigators found that, the lower the residual disease activity, the greater the chance of an enduring response.
“Our findings suggest that patients who did not achieve sustained remission in the open-label period and had higher DAS28 at week 36 were more likely to lose remission with maintenance therapy in the double-blind period. Not surprisingly, patients who achieved remission at only week 36, those who sustained remission at only weeks 28 and 36, and those who sustained remission at only weeks 20, 28, and 36, were at higher risk for loss of remission than patients who sustained remission from week 12 to week 36, indicating that depth of disease control is an important predictor of remission loss,” noted Dr. Smolen and his colleagues.
This study was sponsored by Pfizer. Dr. Smolen has received research grants and consulting fees from AbbVie, Pfizer, Roche, and other biopharmaceutical companies. Several of the investigators are employees of Pfizer and hold Pfizer stock. Another author is an employee of inVentiv Health and was contracted by Pfizer to provide statistical support.
SOURCE: Smolen JS et al. Arthritis Res Ther. doi: 10.1186/s13075-017-1484-9.
There are four factors at baseline in adults with moderately active rheumatoid arthritis that predict those who are most likely to achieve remission with full-dose combo etanercept-methotrexate (ETN-MTX) induction treatment, said Josef S. Smolen, MD, of the University of Vienna, and his associates.
The original PRESERVE study found that after 604 adults with moderately active rheumatoid arthritis achieved low disease activity after 36 weeks of full-dose etanercept (50 mg once weekly) plus methotrexate, subsequent full-dose or reduced-dose (25 mg once weekly) ETN-MTX combos were better at maintaining remission than was methotrexate alone. At the time of the original study, a Disease Activity Score in 28 joints (DAS28) less than 2.6 was considered to denote remission in practice and clinical trials; now that the American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) Boolean definition of remission has been published, a DAS28 less than 2.6 no longer indicates remission according to ACR/EULAR criteria, but rather indicates minimal disease activity, the investigators explained.
This study aimed to determine predictors of remission, the researchers said in their article published in Arthritis Research & Therapy.
Those predictors at baseline are young age; body mass index less than 30 kg/m2; lower Health Assessment Questionnaire scores; and lower disease activity – as measured by DAS28, Simplified Disease Activity Index, and Clinical Disease Activity Index.
If the predictors are favorable and the patients have an “early, strong, and durable response to induction therapy, they are most likely to experience a sustained response after biologic tapering or withdrawal,” Dr. Smolen and his associates said.
In other words, the investigators found that, the lower the residual disease activity, the greater the chance of an enduring response.
“Our findings suggest that patients who did not achieve sustained remission in the open-label period and had higher DAS28 at week 36 were more likely to lose remission with maintenance therapy in the double-blind period. Not surprisingly, patients who achieved remission at only week 36, those who sustained remission at only weeks 28 and 36, and those who sustained remission at only weeks 20, 28, and 36, were at higher risk for loss of remission than patients who sustained remission from week 12 to week 36, indicating that depth of disease control is an important predictor of remission loss,” noted Dr. Smolen and his colleagues.
This study was sponsored by Pfizer. Dr. Smolen has received research grants and consulting fees from AbbVie, Pfizer, Roche, and other biopharmaceutical companies. Several of the investigators are employees of Pfizer and hold Pfizer stock. Another author is an employee of inVentiv Health and was contracted by Pfizer to provide statistical support.
SOURCE: Smolen JS et al. Arthritis Res Ther. doi: 10.1186/s13075-017-1484-9.
There are four factors at baseline in adults with moderately active rheumatoid arthritis that predict those who are most likely to achieve remission with full-dose combo etanercept-methotrexate (ETN-MTX) induction treatment, said Josef S. Smolen, MD, of the University of Vienna, and his associates.
The original PRESERVE study found that after 604 adults with moderately active rheumatoid arthritis achieved low disease activity after 36 weeks of full-dose etanercept (50 mg once weekly) plus methotrexate, subsequent full-dose or reduced-dose (25 mg once weekly) ETN-MTX combos were better at maintaining remission than was methotrexate alone. At the time of the original study, a Disease Activity Score in 28 joints (DAS28) less than 2.6 was considered to denote remission in practice and clinical trials; now that the American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) Boolean definition of remission has been published, a DAS28 less than 2.6 no longer indicates remission according to ACR/EULAR criteria, but rather indicates minimal disease activity, the investigators explained.
This study aimed to determine predictors of remission, the researchers said in their article published in Arthritis Research & Therapy.
Those predictors at baseline are young age; body mass index less than 30 kg/m2; lower Health Assessment Questionnaire scores; and lower disease activity – as measured by DAS28, Simplified Disease Activity Index, and Clinical Disease Activity Index.
If the predictors are favorable and the patients have an “early, strong, and durable response to induction therapy, they are most likely to experience a sustained response after biologic tapering or withdrawal,” Dr. Smolen and his associates said.
In other words, the investigators found that, the lower the residual disease activity, the greater the chance of an enduring response.
“Our findings suggest that patients who did not achieve sustained remission in the open-label period and had higher DAS28 at week 36 were more likely to lose remission with maintenance therapy in the double-blind period. Not surprisingly, patients who achieved remission at only week 36, those who sustained remission at only weeks 28 and 36, and those who sustained remission at only weeks 20, 28, and 36, were at higher risk for loss of remission than patients who sustained remission from week 12 to week 36, indicating that depth of disease control is an important predictor of remission loss,” noted Dr. Smolen and his colleagues.
This study was sponsored by Pfizer. Dr. Smolen has received research grants and consulting fees from AbbVie, Pfizer, Roche, and other biopharmaceutical companies. Several of the investigators are employees of Pfizer and hold Pfizer stock. Another author is an employee of inVentiv Health and was contracted by Pfizer to provide statistical support.
SOURCE: Smolen JS et al. Arthritis Res Ther. doi: 10.1186/s13075-017-1484-9.
FROM ARTHRITIS RESEARCH & THERAPY
Key clinical point:
Major finding: Predictors of remission at baseline are young age; BMI less than 30 kg/m2; lower HAQ scores; and lower disease activity – as measured by DAS28, SDAI, and CDAI.
Study details: Post hoc analysis of 604 patients with moderately active rheumatoid arthritis from the PRESERVE trial.
Disclosures: This study was sponsored by Pfizer. Dr. Smolen has received research grants and consulting fees from AbbVie, Pfizer, Roche, and other biopharmaceutical companies. Several of the investigators are employees of Pfizer and hold Pfizer stock. Another author is an employee of inVentiv Health and was contracted by Pfizer to provide statistical support.
Source: Smolen JS et al. Arthritis Res Ther. doi: 10.1186/s13075-017-1484-9.
Low-dose rituximab cuts infection risk
Treating rheumatoid arthritis patients with lower doses of rituximab for long-term maintenance reduced the risk of serious infections and saved money, based on data from approximately 1,200 patients in a French registry.
In a study published in Rheumatology, Julien Henry, MD, of Institut pour la Sante et la Recherche Medicale, Paris, and colleagues reviewed data from 1,278 patients; 1,093 (85.5%) received a standard dose of rituximab, and 185 (14.5%) received a reduced dose for maintenance therapy. A standard dose was 1,000 mg per infusion given in two infusions 2 weeks apart), and a reduced dose was 500 mg per infusion given in two infusions 2 weeks apart.
After 5 years, maintenance was 55.5% in the standard group and 53.8% in the reduced group; with no significant difference (hazard ratio, 1.03). However, the cumulative dose for retreatment was 39% less in the reduced group (1.4 g per year vs. 2.3 g per year), “which is greatly cost effective,” the researchers wrote.
In addition, the rate of serious infections was significantly lower in the reduced-dose group, compared with the standard-dose group (2.2 per 100 patient-years vs. 4.1 per 100 patient-years; adjusted hazard ratio = 0.50).
“Of note, factors associated with risk of serious infection, such as baseline low gamma globulin or IgG levels, chronic lung or cardiac disease, and extra-articular involvement, did not differ between groups,” the researchers said.
The study findings were limited by several factors including the observational design and lack of data on certain RA outcome measures such as radiographic progression and function, the researchers noted. However, the results support data from similar studies and suggest that a lower dose of rituximab for retreatment of RA “did not alter the maintenance of the treatment at 5 years and is associated with a significant lower rate of serious infections,” they said.
Dr. Henry had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple drug companies, but the study received no specific funding from any of these companies.
SOURCE: Henry J et al. Rheumatology. 2017 Dec 15. doi: 10.1093/rheumatology/kex446.
Treating rheumatoid arthritis patients with lower doses of rituximab for long-term maintenance reduced the risk of serious infections and saved money, based on data from approximately 1,200 patients in a French registry.
In a study published in Rheumatology, Julien Henry, MD, of Institut pour la Sante et la Recherche Medicale, Paris, and colleagues reviewed data from 1,278 patients; 1,093 (85.5%) received a standard dose of rituximab, and 185 (14.5%) received a reduced dose for maintenance therapy. A standard dose was 1,000 mg per infusion given in two infusions 2 weeks apart), and a reduced dose was 500 mg per infusion given in two infusions 2 weeks apart.
After 5 years, maintenance was 55.5% in the standard group and 53.8% in the reduced group; with no significant difference (hazard ratio, 1.03). However, the cumulative dose for retreatment was 39% less in the reduced group (1.4 g per year vs. 2.3 g per year), “which is greatly cost effective,” the researchers wrote.
In addition, the rate of serious infections was significantly lower in the reduced-dose group, compared with the standard-dose group (2.2 per 100 patient-years vs. 4.1 per 100 patient-years; adjusted hazard ratio = 0.50).
“Of note, factors associated with risk of serious infection, such as baseline low gamma globulin or IgG levels, chronic lung or cardiac disease, and extra-articular involvement, did not differ between groups,” the researchers said.
The study findings were limited by several factors including the observational design and lack of data on certain RA outcome measures such as radiographic progression and function, the researchers noted. However, the results support data from similar studies and suggest that a lower dose of rituximab for retreatment of RA “did not alter the maintenance of the treatment at 5 years and is associated with a significant lower rate of serious infections,” they said.
Dr. Henry had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple drug companies, but the study received no specific funding from any of these companies.
SOURCE: Henry J et al. Rheumatology. 2017 Dec 15. doi: 10.1093/rheumatology/kex446.
Treating rheumatoid arthritis patients with lower doses of rituximab for long-term maintenance reduced the risk of serious infections and saved money, based on data from approximately 1,200 patients in a French registry.
In a study published in Rheumatology, Julien Henry, MD, of Institut pour la Sante et la Recherche Medicale, Paris, and colleagues reviewed data from 1,278 patients; 1,093 (85.5%) received a standard dose of rituximab, and 185 (14.5%) received a reduced dose for maintenance therapy. A standard dose was 1,000 mg per infusion given in two infusions 2 weeks apart), and a reduced dose was 500 mg per infusion given in two infusions 2 weeks apart.
After 5 years, maintenance was 55.5% in the standard group and 53.8% in the reduced group; with no significant difference (hazard ratio, 1.03). However, the cumulative dose for retreatment was 39% less in the reduced group (1.4 g per year vs. 2.3 g per year), “which is greatly cost effective,” the researchers wrote.
In addition, the rate of serious infections was significantly lower in the reduced-dose group, compared with the standard-dose group (2.2 per 100 patient-years vs. 4.1 per 100 patient-years; adjusted hazard ratio = 0.50).
“Of note, factors associated with risk of serious infection, such as baseline low gamma globulin or IgG levels, chronic lung or cardiac disease, and extra-articular involvement, did not differ between groups,” the researchers said.
The study findings were limited by several factors including the observational design and lack of data on certain RA outcome measures such as radiographic progression and function, the researchers noted. However, the results support data from similar studies and suggest that a lower dose of rituximab for retreatment of RA “did not alter the maintenance of the treatment at 5 years and is associated with a significant lower rate of serious infections,” they said.
Dr. Henry had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple drug companies, but the study received no specific funding from any of these companies.
SOURCE: Henry J et al. Rheumatology. 2017 Dec 15. doi: 10.1093/rheumatology/kex446.
FROM RHEUMATOLOGY
Key clinical point: A reduced dose of rituximab for RA maintenance resulted in fewer infections and lower cost.
Major finding: A long-term low dose of rituximab for RA patients led to a 39% total dose reduction.
Study details: An observational study of data from 1,278 adult patients in the Autoimmunity and Rituximab registry.
Disclosures: Dr. Henry had no financial conflicts to disclose. Several coauthors disclosed relationships with multiple drug companies, but the study received no specific funding from any of these companies.
Source: Henry J et al. Rheumatology. 2017 Dec 15. doi: 10.1093/rheumatology/kex446.
Ultrasound could have utility in predicting which RA patients stay in remission
Synovitis detected by ultrasound in patients with rheumatoid arthritis (RA) in clinical remission has a moderate but significant independent predictive value for the loss of remission, new research shows.
These findings, reported by Swiss investigators at multiple centers and with different sonographers and ultrasound (US) machines, adds to previous research that has shown that patients in clinical remission with US-detected residual synovitis had a tendency to flare more often and have a shorter duration of remission than did patients with sonographic remission.
“It is currently unknown whether the predictive value of US regarding flares and the duration of remission remains valid in the context of a large group of US assessors using different US machines, as encountered in a real-life setting,” wrote the research team, who conducted the research on behalf of the Swiss Sonography in Arthritis and Rheumatism group and the Swiss Clinical Quality Management in Rheumatic Diseases Foundation.
“Moreover, it remains unclear whether US should be recommended in everyday clinical practice to support therapeutic decisions and to monitor clinical remission,” they added.
In the current study, Dr. Zufferey and his associates analyzed how long 318 RA patients from the Swiss Clinical Quality Management in Rheumatic Diseases cohort remained in remission after they had their first US during remission. The investigators defined loss of remission in this study as either having a 28-joint Disease Activity Score greater than 2.6 or needing to step up treatment with disease-modifying antirheumatic drugs.
The researchers used a previously validated score developed by the Swiss Sonography in Arthritis and Rheumatism group for the definition of US-detected residual synovitis by using cutoffs for B-mode, Doppler, and combined modes that had been defined by previous studies. For example, the cutoffs for significant US-detected residual synovitis were: B-mode of 2 or more joints with synovitis grade of 2 or greater or a total B-mode score greater than 8 points (max score 66 points) and any Doppler activity inside the joint. A combined US synovitis score was defined as B-mode score greater than 8 and/or any Doppler-detected residual synovitis. Patients in clinical remission with a baseline US score above these cutoffs were considered to have significant US-detected residual synovitis (US+), compared with patients who did not have residual synovitis (US–).
The research team observed a loss of remission in 60% of 378 remission phases, with having a 28-joint Disease Activity Score greater than 2.6 deciding the loss of remission status in 66% of cases in both the US+ and the US– groups.
The time from the first US during remission to loss of remission was 2-5 months longer for US– patients than it was for US+ patients.
“After adjusting for potential confounders, [the hazard ratio] of loss of remission for all remission phases using the complete follow-up time was higher for patients with US-detected synovitis at baseline than for those without (combined US score hazard ratio, 1.4; 95% CI, 1.03-2.00 vs. HR, 1.5; 95% CI, 1.1-2.1 for left and right imputation, respectively),” the investigators wrote.
Hazard ratios for the loss of remission also showed a two- to threefold increase when US measurements were taken early in remission (that is, within 3-6 months), a finding that the authors wrote indicated “that the short-term predictive power of US-detected residual synovitis may be more significant than its long-term predictive power.”
The researchers concluded that their study showed that US could be useful in predicting how long patients were likely to remain in remission in “real-life conditions,” but said questions remained over whether it should be used at an individual level.
“According to our results, in particular regarding the moderate HR, a single US done in remission cannot be used as the unique predictor of flare,” they wrote.
Nevertheless, their results may have “some importance in promoting the widespread use of US in real-life conditions for the follow-up of RA, especially when in remission,” they said.
“The next step would be to evaluate whether previous US performed prior to reaching remission and repeated US performed while in remission, particularly after 3-6 months, could provide additional information that is useful to the clinician,” they added.
The Swiss Clinical Quality Management Foundation is supported by the Swiss Society of Rheumatology and nine pharmaceutical companies.
SOURCE: Zufferey P et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.161193
Synovitis detected by ultrasound in patients with rheumatoid arthritis (RA) in clinical remission has a moderate but significant independent predictive value for the loss of remission, new research shows.
These findings, reported by Swiss investigators at multiple centers and with different sonographers and ultrasound (US) machines, adds to previous research that has shown that patients in clinical remission with US-detected residual synovitis had a tendency to flare more often and have a shorter duration of remission than did patients with sonographic remission.
“It is currently unknown whether the predictive value of US regarding flares and the duration of remission remains valid in the context of a large group of US assessors using different US machines, as encountered in a real-life setting,” wrote the research team, who conducted the research on behalf of the Swiss Sonography in Arthritis and Rheumatism group and the Swiss Clinical Quality Management in Rheumatic Diseases Foundation.
“Moreover, it remains unclear whether US should be recommended in everyday clinical practice to support therapeutic decisions and to monitor clinical remission,” they added.
In the current study, Dr. Zufferey and his associates analyzed how long 318 RA patients from the Swiss Clinical Quality Management in Rheumatic Diseases cohort remained in remission after they had their first US during remission. The investigators defined loss of remission in this study as either having a 28-joint Disease Activity Score greater than 2.6 or needing to step up treatment with disease-modifying antirheumatic drugs.
The researchers used a previously validated score developed by the Swiss Sonography in Arthritis and Rheumatism group for the definition of US-detected residual synovitis by using cutoffs for B-mode, Doppler, and combined modes that had been defined by previous studies. For example, the cutoffs for significant US-detected residual synovitis were: B-mode of 2 or more joints with synovitis grade of 2 or greater or a total B-mode score greater than 8 points (max score 66 points) and any Doppler activity inside the joint. A combined US synovitis score was defined as B-mode score greater than 8 and/or any Doppler-detected residual synovitis. Patients in clinical remission with a baseline US score above these cutoffs were considered to have significant US-detected residual synovitis (US+), compared with patients who did not have residual synovitis (US–).
The research team observed a loss of remission in 60% of 378 remission phases, with having a 28-joint Disease Activity Score greater than 2.6 deciding the loss of remission status in 66% of cases in both the US+ and the US– groups.
The time from the first US during remission to loss of remission was 2-5 months longer for US– patients than it was for US+ patients.
“After adjusting for potential confounders, [the hazard ratio] of loss of remission for all remission phases using the complete follow-up time was higher for patients with US-detected synovitis at baseline than for those without (combined US score hazard ratio, 1.4; 95% CI, 1.03-2.00 vs. HR, 1.5; 95% CI, 1.1-2.1 for left and right imputation, respectively),” the investigators wrote.
Hazard ratios for the loss of remission also showed a two- to threefold increase when US measurements were taken early in remission (that is, within 3-6 months), a finding that the authors wrote indicated “that the short-term predictive power of US-detected residual synovitis may be more significant than its long-term predictive power.”
The researchers concluded that their study showed that US could be useful in predicting how long patients were likely to remain in remission in “real-life conditions,” but said questions remained over whether it should be used at an individual level.
“According to our results, in particular regarding the moderate HR, a single US done in remission cannot be used as the unique predictor of flare,” they wrote.
Nevertheless, their results may have “some importance in promoting the widespread use of US in real-life conditions for the follow-up of RA, especially when in remission,” they said.
“The next step would be to evaluate whether previous US performed prior to reaching remission and repeated US performed while in remission, particularly after 3-6 months, could provide additional information that is useful to the clinician,” they added.
The Swiss Clinical Quality Management Foundation is supported by the Swiss Society of Rheumatology and nine pharmaceutical companies.
SOURCE: Zufferey P et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.161193
Synovitis detected by ultrasound in patients with rheumatoid arthritis (RA) in clinical remission has a moderate but significant independent predictive value for the loss of remission, new research shows.
These findings, reported by Swiss investigators at multiple centers and with different sonographers and ultrasound (US) machines, adds to previous research that has shown that patients in clinical remission with US-detected residual synovitis had a tendency to flare more often and have a shorter duration of remission than did patients with sonographic remission.
“It is currently unknown whether the predictive value of US regarding flares and the duration of remission remains valid in the context of a large group of US assessors using different US machines, as encountered in a real-life setting,” wrote the research team, who conducted the research on behalf of the Swiss Sonography in Arthritis and Rheumatism group and the Swiss Clinical Quality Management in Rheumatic Diseases Foundation.
“Moreover, it remains unclear whether US should be recommended in everyday clinical practice to support therapeutic decisions and to monitor clinical remission,” they added.
In the current study, Dr. Zufferey and his associates analyzed how long 318 RA patients from the Swiss Clinical Quality Management in Rheumatic Diseases cohort remained in remission after they had their first US during remission. The investigators defined loss of remission in this study as either having a 28-joint Disease Activity Score greater than 2.6 or needing to step up treatment with disease-modifying antirheumatic drugs.
The researchers used a previously validated score developed by the Swiss Sonography in Arthritis and Rheumatism group for the definition of US-detected residual synovitis by using cutoffs for B-mode, Doppler, and combined modes that had been defined by previous studies. For example, the cutoffs for significant US-detected residual synovitis were: B-mode of 2 or more joints with synovitis grade of 2 or greater or a total B-mode score greater than 8 points (max score 66 points) and any Doppler activity inside the joint. A combined US synovitis score was defined as B-mode score greater than 8 and/or any Doppler-detected residual synovitis. Patients in clinical remission with a baseline US score above these cutoffs were considered to have significant US-detected residual synovitis (US+), compared with patients who did not have residual synovitis (US–).
The research team observed a loss of remission in 60% of 378 remission phases, with having a 28-joint Disease Activity Score greater than 2.6 deciding the loss of remission status in 66% of cases in both the US+ and the US– groups.
The time from the first US during remission to loss of remission was 2-5 months longer for US– patients than it was for US+ patients.
“After adjusting for potential confounders, [the hazard ratio] of loss of remission for all remission phases using the complete follow-up time was higher for patients with US-detected synovitis at baseline than for those without (combined US score hazard ratio, 1.4; 95% CI, 1.03-2.00 vs. HR, 1.5; 95% CI, 1.1-2.1 for left and right imputation, respectively),” the investigators wrote.
Hazard ratios for the loss of remission also showed a two- to threefold increase when US measurements were taken early in remission (that is, within 3-6 months), a finding that the authors wrote indicated “that the short-term predictive power of US-detected residual synovitis may be more significant than its long-term predictive power.”
The researchers concluded that their study showed that US could be useful in predicting how long patients were likely to remain in remission in “real-life conditions,” but said questions remained over whether it should be used at an individual level.
“According to our results, in particular regarding the moderate HR, a single US done in remission cannot be used as the unique predictor of flare,” they wrote.
Nevertheless, their results may have “some importance in promoting the widespread use of US in real-life conditions for the follow-up of RA, especially when in remission,” they said.
“The next step would be to evaluate whether previous US performed prior to reaching remission and repeated US performed while in remission, particularly after 3-6 months, could provide additional information that is useful to the clinician,” they added.
The Swiss Clinical Quality Management Foundation is supported by the Swiss Society of Rheumatology and nine pharmaceutical companies.
SOURCE: Zufferey P et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.161193
FROM JOURNAL OF RHEUMATOLOGY
Key clinical point: Ultrasound could be useful in predicting how long patients are likely to remain in remission in “real-life conditions.”
Major finding: Hazard ratios (HRs) for loss of remission were moderately but significantly higher for patients with ultrasound-detected synovitis at baseline than for those without (combined ultrasound score HR, 1.4; 95% CI, 1.03-2.00 vs. HR, 1.5; 95% CI, 1.1-2.1 for left and right imputation, respectively).
Study details: A multicenter cohort study of 318 RA patients from the Swiss Clinical Quality Management cohort.
Disclosures: The Swiss Clinical Quality Management Foundation is supported by the Swiss Society of Rheumatology and nine pharmaceutical companies.
Source: Zufferey P et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.161193
Recurrent serious infection risk captured in real world RA study
according to findings from a study of the British Society for Rheumatology Biologics Register–Rheumatoid Arthritis.
Sujith Subesinghe, MBBS, from the academic department of rheumatology at King’s College London and his coauthors identified 5,289 individuals from the registry who had experienced at least one episode of serious infection. All patients were also on either conventional synthetic disease-modifying antirheumatic drugs (DMARDs) or biologic DMARDs at the time of their index infection.
Serious infections were more likely to occur in individuals who were older, male, seropositive, steroid users, or who had higher baseline Health Assessment Questionnaire scores. The most common type of serious infection was respiratory, which accounted for 44% of all events.
Overall, recurrent infections tended to occur in the same organ as index infections. Patients who experienced sepsis as the index infection had the highest risk of recurrent serious infection of 19.7% per annum, representing a 32% increased risk for recurrent serious infection, compared with individuals whose index infection was in the respiratory tract. Patients whose initial infection was musculoskeletal or gastrointestinal had a recurrence rate below 10%.
“It has been shown previously that a history of SI [serious infection] is a strong predictor of subsequent SI, but what has not been shown before is that the organ class of the index event has a large impact on the likelihood of recurrent SI,” the authors wrote.
Each decade of increased age was associated with a 34% increase in risk of recurrent infection. Noting that multiple drug use was a surrogate measure of comorbidity at baseline, researchers found that individuals taking 6-10 drugs at baseline had a 26% increased risk for recurrent infection, while those treated with 11 or more drugs had a 74% increased risk.
The authors observed that baseline steroid use was higher among patients suffering sepsis, compared with other types of serious infection.
They observed that, although a strong association between serious infection and steroid use has been reported, no firm conclusions have been reached by systematic reviews or meta-analyses “Steroids are more likely to be prescribed to patients with more aggressive, recalcitrant disease; this group has a higher baseline infection risk, and therefore, there is potential for confounding by indication.”
However, baseline disease activity score, disease duration, seropositivity, and smoking did not significantly predict the likelihood of infection recurrence, despite the fact that these are traditional predictors of infection. The authors suggested this may be the result of a form of selection bias.
“Patients readmitted with SI were from an already at-risk group and therefore lack of association between traditional predictors of infection and recurrent SI may be spurious.”
Commenting on their findings, the authors observed that individuals with rheumatoid arthritis and a history of infection were a complex group to manage, and even small differences in relative infection risk with different biologics may become significant.
“Further research needs to be undertaken to understand the patterns of recurrent infection and to appreciate the nuances in differential infection profiles of immunosuppressive drugs, to promote safe therapeutic decisions and promote personalization of care.”
The study was supported by the British Society for Rheumatology. Two authors declared speaking fees or honoraria from the pharmaceutical industry.
SOURCE: Subesinghe S et al. Rheumatology [Oxford]. 2018 Jan 10. doi: 10.1093/rheumatology/kex469.
according to findings from a study of the British Society for Rheumatology Biologics Register–Rheumatoid Arthritis.
Sujith Subesinghe, MBBS, from the academic department of rheumatology at King’s College London and his coauthors identified 5,289 individuals from the registry who had experienced at least one episode of serious infection. All patients were also on either conventional synthetic disease-modifying antirheumatic drugs (DMARDs) or biologic DMARDs at the time of their index infection.
Serious infections were more likely to occur in individuals who were older, male, seropositive, steroid users, or who had higher baseline Health Assessment Questionnaire scores. The most common type of serious infection was respiratory, which accounted for 44% of all events.
Overall, recurrent infections tended to occur in the same organ as index infections. Patients who experienced sepsis as the index infection had the highest risk of recurrent serious infection of 19.7% per annum, representing a 32% increased risk for recurrent serious infection, compared with individuals whose index infection was in the respiratory tract. Patients whose initial infection was musculoskeletal or gastrointestinal had a recurrence rate below 10%.
“It has been shown previously that a history of SI [serious infection] is a strong predictor of subsequent SI, but what has not been shown before is that the organ class of the index event has a large impact on the likelihood of recurrent SI,” the authors wrote.
Each decade of increased age was associated with a 34% increase in risk of recurrent infection. Noting that multiple drug use was a surrogate measure of comorbidity at baseline, researchers found that individuals taking 6-10 drugs at baseline had a 26% increased risk for recurrent infection, while those treated with 11 or more drugs had a 74% increased risk.
The authors observed that baseline steroid use was higher among patients suffering sepsis, compared with other types of serious infection.
They observed that, although a strong association between serious infection and steroid use has been reported, no firm conclusions have been reached by systematic reviews or meta-analyses “Steroids are more likely to be prescribed to patients with more aggressive, recalcitrant disease; this group has a higher baseline infection risk, and therefore, there is potential for confounding by indication.”
However, baseline disease activity score, disease duration, seropositivity, and smoking did not significantly predict the likelihood of infection recurrence, despite the fact that these are traditional predictors of infection. The authors suggested this may be the result of a form of selection bias.
“Patients readmitted with SI were from an already at-risk group and therefore lack of association between traditional predictors of infection and recurrent SI may be spurious.”
Commenting on their findings, the authors observed that individuals with rheumatoid arthritis and a history of infection were a complex group to manage, and even small differences in relative infection risk with different biologics may become significant.
“Further research needs to be undertaken to understand the patterns of recurrent infection and to appreciate the nuances in differential infection profiles of immunosuppressive drugs, to promote safe therapeutic decisions and promote personalization of care.”
The study was supported by the British Society for Rheumatology. Two authors declared speaking fees or honoraria from the pharmaceutical industry.
SOURCE: Subesinghe S et al. Rheumatology [Oxford]. 2018 Jan 10. doi: 10.1093/rheumatology/kex469.
according to findings from a study of the British Society for Rheumatology Biologics Register–Rheumatoid Arthritis.
Sujith Subesinghe, MBBS, from the academic department of rheumatology at King’s College London and his coauthors identified 5,289 individuals from the registry who had experienced at least one episode of serious infection. All patients were also on either conventional synthetic disease-modifying antirheumatic drugs (DMARDs) or biologic DMARDs at the time of their index infection.
Serious infections were more likely to occur in individuals who were older, male, seropositive, steroid users, or who had higher baseline Health Assessment Questionnaire scores. The most common type of serious infection was respiratory, which accounted for 44% of all events.
Overall, recurrent infections tended to occur in the same organ as index infections. Patients who experienced sepsis as the index infection had the highest risk of recurrent serious infection of 19.7% per annum, representing a 32% increased risk for recurrent serious infection, compared with individuals whose index infection was in the respiratory tract. Patients whose initial infection was musculoskeletal or gastrointestinal had a recurrence rate below 10%.
“It has been shown previously that a history of SI [serious infection] is a strong predictor of subsequent SI, but what has not been shown before is that the organ class of the index event has a large impact on the likelihood of recurrent SI,” the authors wrote.
Each decade of increased age was associated with a 34% increase in risk of recurrent infection. Noting that multiple drug use was a surrogate measure of comorbidity at baseline, researchers found that individuals taking 6-10 drugs at baseline had a 26% increased risk for recurrent infection, while those treated with 11 or more drugs had a 74% increased risk.
The authors observed that baseline steroid use was higher among patients suffering sepsis, compared with other types of serious infection.
They observed that, although a strong association between serious infection and steroid use has been reported, no firm conclusions have been reached by systematic reviews or meta-analyses “Steroids are more likely to be prescribed to patients with more aggressive, recalcitrant disease; this group has a higher baseline infection risk, and therefore, there is potential for confounding by indication.”
However, baseline disease activity score, disease duration, seropositivity, and smoking did not significantly predict the likelihood of infection recurrence, despite the fact that these are traditional predictors of infection. The authors suggested this may be the result of a form of selection bias.
“Patients readmitted with SI were from an already at-risk group and therefore lack of association between traditional predictors of infection and recurrent SI may be spurious.”
Commenting on their findings, the authors observed that individuals with rheumatoid arthritis and a history of infection were a complex group to manage, and even small differences in relative infection risk with different biologics may become significant.
“Further research needs to be undertaken to understand the patterns of recurrent infection and to appreciate the nuances in differential infection profiles of immunosuppressive drugs, to promote safe therapeutic decisions and promote personalization of care.”
The study was supported by the British Society for Rheumatology. Two authors declared speaking fees or honoraria from the pharmaceutical industry.
SOURCE: Subesinghe S et al. Rheumatology [Oxford]. 2018 Jan 10. doi: 10.1093/rheumatology/kex469.
FROM RHEUMATOLOGY
Key clinical point: Recurrent serious infections are relatively common among individuals with RA, particularly in the first year after an initial serious infection.
Major finding: The baseline annual rate of serious infections in patients with RA increased to 14.1% after having infection.
Data source: Analysis of data from 5,289 individuals from the British Society for Rheumatology Biologics Register–Rheumatoid Arthritis cohort.
Disclosures: The study was supported by the British Society for Rheumatology. Two authors declared speaking fees or honoraria from the pharmaceutical industry.
Source: Subesinghe S et al. Rheumatology [Oxford]. 2018 Jan 10. doi: 10.1093/rheumatology/kex469.
Arthritis treatment costs per person held steady from 2008 to 2014
but the rise in proportion of the arthritis patients in the U.S. population led to billions more dollars being spent on this population, reported Amit D. Raval, PhD, and Ami Vyas, PhD, of the University of Rhode Island, Kingston.
Aggregate expenditures for patients with arthritis rose from $584.8 billion in 2008 to $645 billion in 2014, accounting for about 4% of the U.S. gross domestic product in these years, the investigators reported in the Journal of Rheumatology. “The increase in aggregate unadjusted total direct health care expenditures was mainly driven through an increase in weighted population of individuals with arthritis,” which rose from 56.1 million adults with arthritis in 2008 to 65.1 million in 2014. Nonetheless, there was a “slowdown in the intensity of adjusted incremental expenditures and [out of pocket expenditures] specifically from 2013, which led to a huge decline in the aggregate direct healthcare expenditures in 2014.”
During this time period, there was an increase in the percentage of Hispanics, poor, obese, and individuals with activity limitations and mental health disorders among both those with and without arthritis. This is consistent with the study findings that incremental health care expenditures in persons with arthritis were largely driven by difference in age, health status, and chronic conditions, such as hypertension, hyperlipidemia, and heart disease, among those with and without arthritis, Dr. Raval and Dr. Vyas said.
Annual total health care expenditures in persons with arthritis fell from $10,424 in 2008 to $9,910 in 2014. The average annual total out-of-pocket expenditures in 2008 was $1,493, which was 14% of total health care expenditures that year; in 2014, this fell to $1,099. There were similar trends in persons without arthritis, Dr. Raval and Dr. Vyas reported.
The top three expenditure categories in 2008 were outpatient (32.6%), inpatient (29.0%), and prescription drug costs (24.7%); in 2014, these changed to outpatient (33.8%), prescription drug (26.8%), and inpatient costs (26.4%). There are a number of ways to explain these trends, the researchers said. It is likely that outpatient services, such as outpatient orthopedic surgeries, are becoming “a common management option for arthritis.” Also, outpatient medication services now may include administration of injectables such as biologics. There also were “relatively stable” inpatient expenditures from 2008 to 2013 and a decline in 2014. “Our findings are consistent with studies assessing the effect of the Affordable Care Act, such as introduction of the Hospital Readmission Reduction Program,” said Dr. Raval and Dr. Vyas.
SOURCE: Amit R et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.170368.
but the rise in proportion of the arthritis patients in the U.S. population led to billions more dollars being spent on this population, reported Amit D. Raval, PhD, and Ami Vyas, PhD, of the University of Rhode Island, Kingston.
Aggregate expenditures for patients with arthritis rose from $584.8 billion in 2008 to $645 billion in 2014, accounting for about 4% of the U.S. gross domestic product in these years, the investigators reported in the Journal of Rheumatology. “The increase in aggregate unadjusted total direct health care expenditures was mainly driven through an increase in weighted population of individuals with arthritis,” which rose from 56.1 million adults with arthritis in 2008 to 65.1 million in 2014. Nonetheless, there was a “slowdown in the intensity of adjusted incremental expenditures and [out of pocket expenditures] specifically from 2013, which led to a huge decline in the aggregate direct healthcare expenditures in 2014.”
During this time period, there was an increase in the percentage of Hispanics, poor, obese, and individuals with activity limitations and mental health disorders among both those with and without arthritis. This is consistent with the study findings that incremental health care expenditures in persons with arthritis were largely driven by difference in age, health status, and chronic conditions, such as hypertension, hyperlipidemia, and heart disease, among those with and without arthritis, Dr. Raval and Dr. Vyas said.
Annual total health care expenditures in persons with arthritis fell from $10,424 in 2008 to $9,910 in 2014. The average annual total out-of-pocket expenditures in 2008 was $1,493, which was 14% of total health care expenditures that year; in 2014, this fell to $1,099. There were similar trends in persons without arthritis, Dr. Raval and Dr. Vyas reported.
The top three expenditure categories in 2008 were outpatient (32.6%), inpatient (29.0%), and prescription drug costs (24.7%); in 2014, these changed to outpatient (33.8%), prescription drug (26.8%), and inpatient costs (26.4%). There are a number of ways to explain these trends, the researchers said. It is likely that outpatient services, such as outpatient orthopedic surgeries, are becoming “a common management option for arthritis.” Also, outpatient medication services now may include administration of injectables such as biologics. There also were “relatively stable” inpatient expenditures from 2008 to 2013 and a decline in 2014. “Our findings are consistent with studies assessing the effect of the Affordable Care Act, such as introduction of the Hospital Readmission Reduction Program,” said Dr. Raval and Dr. Vyas.
SOURCE: Amit R et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.170368.
but the rise in proportion of the arthritis patients in the U.S. population led to billions more dollars being spent on this population, reported Amit D. Raval, PhD, and Ami Vyas, PhD, of the University of Rhode Island, Kingston.
Aggregate expenditures for patients with arthritis rose from $584.8 billion in 2008 to $645 billion in 2014, accounting for about 4% of the U.S. gross domestic product in these years, the investigators reported in the Journal of Rheumatology. “The increase in aggregate unadjusted total direct health care expenditures was mainly driven through an increase in weighted population of individuals with arthritis,” which rose from 56.1 million adults with arthritis in 2008 to 65.1 million in 2014. Nonetheless, there was a “slowdown in the intensity of adjusted incremental expenditures and [out of pocket expenditures] specifically from 2013, which led to a huge decline in the aggregate direct healthcare expenditures in 2014.”
During this time period, there was an increase in the percentage of Hispanics, poor, obese, and individuals with activity limitations and mental health disorders among both those with and without arthritis. This is consistent with the study findings that incremental health care expenditures in persons with arthritis were largely driven by difference in age, health status, and chronic conditions, such as hypertension, hyperlipidemia, and heart disease, among those with and without arthritis, Dr. Raval and Dr. Vyas said.
Annual total health care expenditures in persons with arthritis fell from $10,424 in 2008 to $9,910 in 2014. The average annual total out-of-pocket expenditures in 2008 was $1,493, which was 14% of total health care expenditures that year; in 2014, this fell to $1,099. There were similar trends in persons without arthritis, Dr. Raval and Dr. Vyas reported.
The top three expenditure categories in 2008 were outpatient (32.6%), inpatient (29.0%), and prescription drug costs (24.7%); in 2014, these changed to outpatient (33.8%), prescription drug (26.8%), and inpatient costs (26.4%). There are a number of ways to explain these trends, the researchers said. It is likely that outpatient services, such as outpatient orthopedic surgeries, are becoming “a common management option for arthritis.” Also, outpatient medication services now may include administration of injectables such as biologics. There also were “relatively stable” inpatient expenditures from 2008 to 2013 and a decline in 2014. “Our findings are consistent with studies assessing the effect of the Affordable Care Act, such as introduction of the Hospital Readmission Reduction Program,” said Dr. Raval and Dr. Vyas.
SOURCE: Amit R et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.170368.
FROM JOURNAL OF RHEUMATOLOGY
Key clinical point: The annual direct health care expenditures per person with arthritis remained relatively stable over the years 2008 to 2014.
Major finding: Aggregate expenditures for patients with arthritis rose from $584.8 billion in 2008 to $645 billion in 2014.
Study details: Medical Expenditure Panel Survey (MEPS) data from approximately 5,000-6,000 persons aged 18 years and older with arthritis each year during 2008-2014 and approximately 17,000-20,000 persons without arthritis each year during that period.
Disclosures: No information on relevant financial disclosures was evident.
Source: Amit R et al. J Rheumatol. 2018 Jan 15. doi: 10.3899/jrheum.170368.
Putting a number on biologic DMARD costs
Annual medical costs for patients with rheumatoid arthritis were almost three times higher for those who used biologic disease-modifying antirheumatic drugs (bDMARDs), compared with those who used any treatment regimen, according to meta-analysis of 12 studies conducted since bDMARDs were introduced in 1999.
RA patients who used bDMARDs had an average direct cost of $36,053 per year, which was 2.9 times higher than the $12,509 in annual direct medical costs for all RA patients on any treatment regimen. Proportionately, the difference was even greater for RA-specific care, with the annual cost of bDMARD care ($20,262) 5.4 times higher than that of all treatment regimens ($3,723), reported Andrew Hresko and his associates at Brigham and Women’s Hospital, Boston.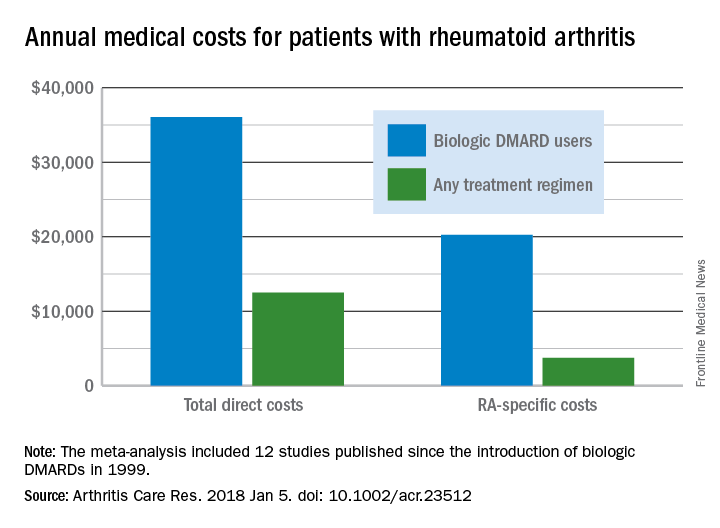
Funding for the study was supported through a grant from the National Institutes of Health. Mr. Hresko was supported by a fellowship from Tufts University. One of his associates receives research support from grants to his hospital from Amgen, Pfizer, Eli Lilly, AstraZeneca, Genentech, Bristol Myers Squibb, and Corrona. The third investigator is now an employee of Amgen but was not during the study.
SOURCE: Hresko A et al. Arthritis Care Res. 2018 Jan 5. doi: 10.1002/acr.23512.
Annual medical costs for patients with rheumatoid arthritis were almost three times higher for those who used biologic disease-modifying antirheumatic drugs (bDMARDs), compared with those who used any treatment regimen, according to meta-analysis of 12 studies conducted since bDMARDs were introduced in 1999.
RA patients who used bDMARDs had an average direct cost of $36,053 per year, which was 2.9 times higher than the $12,509 in annual direct medical costs for all RA patients on any treatment regimen. Proportionately, the difference was even greater for RA-specific care, with the annual cost of bDMARD care ($20,262) 5.4 times higher than that of all treatment regimens ($3,723), reported Andrew Hresko and his associates at Brigham and Women’s Hospital, Boston.
Funding for the study was supported through a grant from the National Institutes of Health. Mr. Hresko was supported by a fellowship from Tufts University. One of his associates receives research support from grants to his hospital from Amgen, Pfizer, Eli Lilly, AstraZeneca, Genentech, Bristol Myers Squibb, and Corrona. The third investigator is now an employee of Amgen but was not during the study.
SOURCE: Hresko A et al. Arthritis Care Res. 2018 Jan 5. doi: 10.1002/acr.23512.
Annual medical costs for patients with rheumatoid arthritis were almost three times higher for those who used biologic disease-modifying antirheumatic drugs (bDMARDs), compared with those who used any treatment regimen, according to meta-analysis of 12 studies conducted since bDMARDs were introduced in 1999.
RA patients who used bDMARDs had an average direct cost of $36,053 per year, which was 2.9 times higher than the $12,509 in annual direct medical costs for all RA patients on any treatment regimen. Proportionately, the difference was even greater for RA-specific care, with the annual cost of bDMARD care ($20,262) 5.4 times higher than that of all treatment regimens ($3,723), reported Andrew Hresko and his associates at Brigham and Women’s Hospital, Boston.
Funding for the study was supported through a grant from the National Institutes of Health. Mr. Hresko was supported by a fellowship from Tufts University. One of his associates receives research support from grants to his hospital from Amgen, Pfizer, Eli Lilly, AstraZeneca, Genentech, Bristol Myers Squibb, and Corrona. The third investigator is now an employee of Amgen but was not during the study.
SOURCE: Hresko A et al. Arthritis Care Res. 2018 Jan 5. doi: 10.1002/acr.23512.
FROM ARTHRITIS CARE & RESEARCH
Novel herpes zoster vaccine is more cost effective than old vaccine
The novel herpes zoster subunit vaccine (HZ/su) is more effective and less expensive than the currently used live attenuated virus (ZVL), according to a study from the Center for Value-Based Research.
Phuc Le, PhD, of the Cleveland Clinic and her colleague Michael Rothberg, MD, conducted an economic analysis of vaccine strategies from the societal perspective. This included the direct medical costs and productivity losses associated with HZ disease and complications.
The one-, two-, and three-way sensitivity analyses examined how different variables affected the cost-effectiveness of different vaccine strategies. The one-way analysis examined the association of input variables and cost-effectiveness. This included HZ/su prices, waning rate and initial efficacy of a dose of HZ/su, and the adherence rate. This analysis revealed that, compared with no vaccination, HZ/su would provide cost savings up to a price of $160, or $80 per dose.
Regardless of circumstance, HZ/su was always more effective than ZVL according to the two-way sensitivity analysis. This analysis took into account the joint effect of price, adherence to two doses of HZ/su, efficacy, and the waning rate of one dose and two doses of HZ/su and ZVL. Compared with ZVL, HZ/su would be less costly up to a price of $350 per series.
Adherence rates to vaccination schedules were important in determining the efficacy and waning rate which ultimately effected cost-effectiveness. The three-way sensitivity analysis found that, if HZ/su adherence to the second dose was greater than 56.8%, results were insensitive to the variation of single-dose efficacy and waning rate of HZ/su. But if adherence rates fell below 40%, combinations of waning rate and lower efficacy made HZ/su cost ineffective. Most importantly, ZVL was never cost effective for 60-year-old patients.
Despite a projected price of $280 per series, HZ/su is still more effective and less expensive than ZVL for adults 60 years or older. According to Dr. Le and Dr. Rothberg, the assumptions about the vaccine’s efficacy duration and price were reasonable. But, if the vaccine price were to rise in the future, or a single dose becomes much less effective than reported by GlaxoSmithKline, or if adherence to the second dose was remarkably low, the results of the study would be changed.
“An ACIP recommendation stating a preference for HZ/su over ZVL could lead to future price increases, which would render the vaccine no longer cost effective” wrote Dr. Le and Dr. Rothberg. “Therefore, a recommendation linked to periodic reassessment of cost-effectiveness based on the vaccine price might help to mitigate the effect of the recommendation on vaccine affordability.”
Dr. Le and Dr. Rothberg reported having no conflicts of interest.
The HZ/su vaccine is not yet approved by the U.S. Food and Drug Administration, but in 2017, the FDA Advisory Committee unanimously voted in favor of its use in adults age 50 years and older.
SOURCE: Phuc L et al. JAMA Intern Med. 2018 Jan 2. doi: 10.1001/jamainternmed.2017.7431. Najafzadeh M. JAMA Intern Med. 2018. doi: 10.1001/jamainternmed.2017.7442.
The herpes zoster (HZ) virus disproportionately affects elderly populations. As the U.S. population ages, tools and mechanisms to reduce the clinical and economic burden of HZ will be needed in the coming years.
The work of Dr. Le and Dr. Rothberg presents the results of an economic evaluation on randomized clinical trials of a yet-to-be-approved novel HZ subunit vaccine (HZ/su) to determine the economic and clinical benefit of the new vaccine, compared with the currently used vaccine. Although HZ/su is intended to be a two-dose vaccine, the study focused on a one-time vaccine strategy because booster vaccines are unpopular and not recommended by the Advisory Committee on Immunization Practices.
“If priced at $280 per two required doses, HZ/su appears to be a cost-saving option, compared with ZVL and a cost-effective option, compared with no-vaccine strategies,” wrote Dr. Najafzadeh. “However, the value of HZ/su vaccine would be even higher if it could be marketed at a price comparable to that of ZVL.” An added benefit of the HZ/su vaccine is that it can be used in immunocompromised patients.
Mehdi Najafzadeh, PhD , is an instructor in medicine at Harvard Medical School, Boston. He also serves as an associate statistician/epidemiologist in the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital, Boston.
The herpes zoster (HZ) virus disproportionately affects elderly populations. As the U.S. population ages, tools and mechanisms to reduce the clinical and economic burden of HZ will be needed in the coming years.
The work of Dr. Le and Dr. Rothberg presents the results of an economic evaluation on randomized clinical trials of a yet-to-be-approved novel HZ subunit vaccine (HZ/su) to determine the economic and clinical benefit of the new vaccine, compared with the currently used vaccine. Although HZ/su is intended to be a two-dose vaccine, the study focused on a one-time vaccine strategy because booster vaccines are unpopular and not recommended by the Advisory Committee on Immunization Practices.
“If priced at $280 per two required doses, HZ/su appears to be a cost-saving option, compared with ZVL and a cost-effective option, compared with no-vaccine strategies,” wrote Dr. Najafzadeh. “However, the value of HZ/su vaccine would be even higher if it could be marketed at a price comparable to that of ZVL.” An added benefit of the HZ/su vaccine is that it can be used in immunocompromised patients.
Mehdi Najafzadeh, PhD , is an instructor in medicine at Harvard Medical School, Boston. He also serves as an associate statistician/epidemiologist in the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital, Boston.
The herpes zoster (HZ) virus disproportionately affects elderly populations. As the U.S. population ages, tools and mechanisms to reduce the clinical and economic burden of HZ will be needed in the coming years.
The work of Dr. Le and Dr. Rothberg presents the results of an economic evaluation on randomized clinical trials of a yet-to-be-approved novel HZ subunit vaccine (HZ/su) to determine the economic and clinical benefit of the new vaccine, compared with the currently used vaccine. Although HZ/su is intended to be a two-dose vaccine, the study focused on a one-time vaccine strategy because booster vaccines are unpopular and not recommended by the Advisory Committee on Immunization Practices.
“If priced at $280 per two required doses, HZ/su appears to be a cost-saving option, compared with ZVL and a cost-effective option, compared with no-vaccine strategies,” wrote Dr. Najafzadeh. “However, the value of HZ/su vaccine would be even higher if it could be marketed at a price comparable to that of ZVL.” An added benefit of the HZ/su vaccine is that it can be used in immunocompromised patients.
Mehdi Najafzadeh, PhD , is an instructor in medicine at Harvard Medical School, Boston. He also serves as an associate statistician/epidemiologist in the division of pharmacoepidemiology and pharmacoeconomics at Brigham and Women’s Hospital, Boston.
The novel herpes zoster subunit vaccine (HZ/su) is more effective and less expensive than the currently used live attenuated virus (ZVL), according to a study from the Center for Value-Based Research.
Phuc Le, PhD, of the Cleveland Clinic and her colleague Michael Rothberg, MD, conducted an economic analysis of vaccine strategies from the societal perspective. This included the direct medical costs and productivity losses associated with HZ disease and complications.
The one-, two-, and three-way sensitivity analyses examined how different variables affected the cost-effectiveness of different vaccine strategies. The one-way analysis examined the association of input variables and cost-effectiveness. This included HZ/su prices, waning rate and initial efficacy of a dose of HZ/su, and the adherence rate. This analysis revealed that, compared with no vaccination, HZ/su would provide cost savings up to a price of $160, or $80 per dose.
Regardless of circumstance, HZ/su was always more effective than ZVL according to the two-way sensitivity analysis. This analysis took into account the joint effect of price, adherence to two doses of HZ/su, efficacy, and the waning rate of one dose and two doses of HZ/su and ZVL. Compared with ZVL, HZ/su would be less costly up to a price of $350 per series.
Adherence rates to vaccination schedules were important in determining the efficacy and waning rate which ultimately effected cost-effectiveness. The three-way sensitivity analysis found that, if HZ/su adherence to the second dose was greater than 56.8%, results were insensitive to the variation of single-dose efficacy and waning rate of HZ/su. But if adherence rates fell below 40%, combinations of waning rate and lower efficacy made HZ/su cost ineffective. Most importantly, ZVL was never cost effective for 60-year-old patients.
Despite a projected price of $280 per series, HZ/su is still more effective and less expensive than ZVL for adults 60 years or older. According to Dr. Le and Dr. Rothberg, the assumptions about the vaccine’s efficacy duration and price were reasonable. But, if the vaccine price were to rise in the future, or a single dose becomes much less effective than reported by GlaxoSmithKline, or if adherence to the second dose was remarkably low, the results of the study would be changed.
“An ACIP recommendation stating a preference for HZ/su over ZVL could lead to future price increases, which would render the vaccine no longer cost effective” wrote Dr. Le and Dr. Rothberg. “Therefore, a recommendation linked to periodic reassessment of cost-effectiveness based on the vaccine price might help to mitigate the effect of the recommendation on vaccine affordability.”
Dr. Le and Dr. Rothberg reported having no conflicts of interest.
The HZ/su vaccine is not yet approved by the U.S. Food and Drug Administration, but in 2017, the FDA Advisory Committee unanimously voted in favor of its use in adults age 50 years and older.
SOURCE: Phuc L et al. JAMA Intern Med. 2018 Jan 2. doi: 10.1001/jamainternmed.2017.7431. Najafzadeh M. JAMA Intern Med. 2018. doi: 10.1001/jamainternmed.2017.7442.
The novel herpes zoster subunit vaccine (HZ/su) is more effective and less expensive than the currently used live attenuated virus (ZVL), according to a study from the Center for Value-Based Research.
Phuc Le, PhD, of the Cleveland Clinic and her colleague Michael Rothberg, MD, conducted an economic analysis of vaccine strategies from the societal perspective. This included the direct medical costs and productivity losses associated with HZ disease and complications.
The one-, two-, and three-way sensitivity analyses examined how different variables affected the cost-effectiveness of different vaccine strategies. The one-way analysis examined the association of input variables and cost-effectiveness. This included HZ/su prices, waning rate and initial efficacy of a dose of HZ/su, and the adherence rate. This analysis revealed that, compared with no vaccination, HZ/su would provide cost savings up to a price of $160, or $80 per dose.
Regardless of circumstance, HZ/su was always more effective than ZVL according to the two-way sensitivity analysis. This analysis took into account the joint effect of price, adherence to two doses of HZ/su, efficacy, and the waning rate of one dose and two doses of HZ/su and ZVL. Compared with ZVL, HZ/su would be less costly up to a price of $350 per series.
Adherence rates to vaccination schedules were important in determining the efficacy and waning rate which ultimately effected cost-effectiveness. The three-way sensitivity analysis found that, if HZ/su adherence to the second dose was greater than 56.8%, results were insensitive to the variation of single-dose efficacy and waning rate of HZ/su. But if adherence rates fell below 40%, combinations of waning rate and lower efficacy made HZ/su cost ineffective. Most importantly, ZVL was never cost effective for 60-year-old patients.
Despite a projected price of $280 per series, HZ/su is still more effective and less expensive than ZVL for adults 60 years or older. According to Dr. Le and Dr. Rothberg, the assumptions about the vaccine’s efficacy duration and price were reasonable. But, if the vaccine price were to rise in the future, or a single dose becomes much less effective than reported by GlaxoSmithKline, or if adherence to the second dose was remarkably low, the results of the study would be changed.
“An ACIP recommendation stating a preference for HZ/su over ZVL could lead to future price increases, which would render the vaccine no longer cost effective” wrote Dr. Le and Dr. Rothberg. “Therefore, a recommendation linked to periodic reassessment of cost-effectiveness based on the vaccine price might help to mitigate the effect of the recommendation on vaccine affordability.”
Dr. Le and Dr. Rothberg reported having no conflicts of interest.
The HZ/su vaccine is not yet approved by the U.S. Food and Drug Administration, but in 2017, the FDA Advisory Committee unanimously voted in favor of its use in adults age 50 years and older.
SOURCE: Phuc L et al. JAMA Intern Med. 2018 Jan 2. doi: 10.1001/jamainternmed.2017.7431. Najafzadeh M. JAMA Intern Med. 2018. doi: 10.1001/jamainternmed.2017.7442.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Cost-effectiveness can be an important factor in considering patient treatment for herpes zoster.
Major finding:
Study details: The study was based on U.S. medical literature. Data were derived from adults 60 years or older in patient groups of 100-30,000 from July 1 to July 31, 2017.
Disclosures: None of the researchers had conflicts of interest to report.
Source: L. Phuc et al. JAMA Intern Med. 2018 Jan 2. doi: 10.1001/jamainternmed.2017.7431; Najafzadeh M. JAMA Intern Med. 2018. doi: 10.1001/jamainternmed.2017.7442.