User login
Researchers identify three distinct clinical-histologic-genetic subtypes in RA
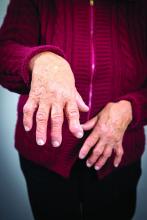
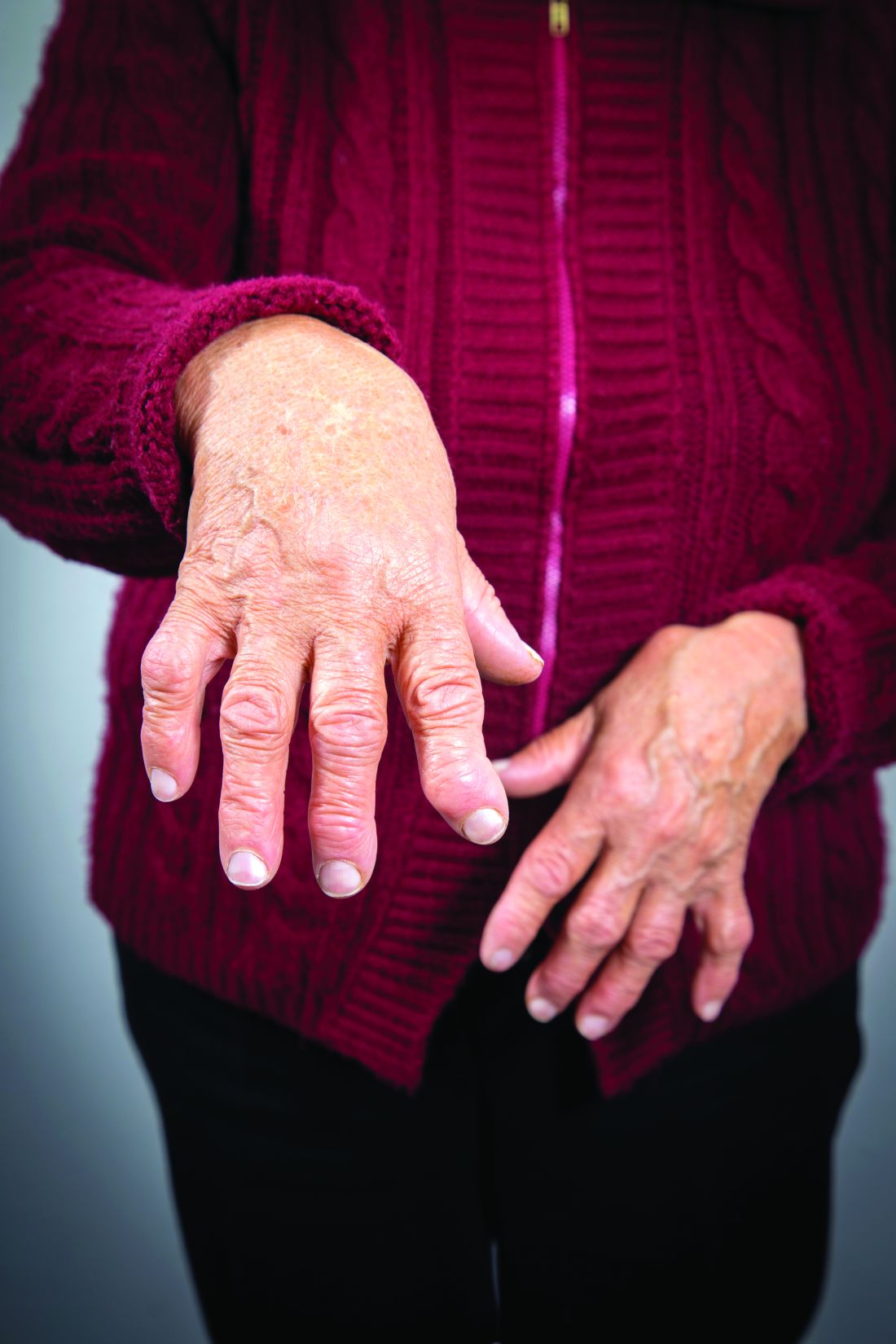
Researchers have identified three different synovial subtypes of rheumatoid arthritis that exhibit different mechanisms of pain and correlate with specific clinical phenotypes.
The findings could be clinically meaningful and may help guide optimal treatment strategies for patients, as well as provide a better understanding of the cause of pain in patients with high tender and swollen joint counts but little tissue inflammation, according to the research team led by Dana E. Orange, MD, of the Hospital for Special Surgery and Rockefeller University in New York.
The report was published in Arthritis & Rheumatology.
The assessment of the synovium in rheumatoid arthritis (RA) has the potential to provide guidance on optimal treatment strategies, they noted, but its classification has not yet factored into current diagnosis or treatment guidelines of RA.
In total, the research team analyzed 20 histologic features on 129 synovial tissue samples.
The researchers used machine learning integration to identify three distinct molecular subtypes of RA from a consensus clustering of the 500 most variable genes expressed in a subset of 45 synovial samples, including 39 from RA patients. The subtypes were high inflammatory, low inflammatory, and a mixed phenotype.
The researchers then took the histologic features that best corresponded to each subtype to develop a histology scoring algorithm that predicted the three gene expression subtypes (using only histology features), each of which were each associated with levels of erythrocyte sedimentation rate, C-reactive protein, and autoantibodies.
The histologic features that most strongly defined the high inflammatory subtype included three plasma cell features: binucleate plasma cells, plasma cell percentage, and Russell bodies. Patients with a high inflammatory synovial subtype also exhibited higher levels of markers of systemic inflammation and autoantibodies. For example, C-reactive protein was significantly correlated with pain in the high inflammatory group.
“This suggests that pain is associated with inflammation in patients with high inflammatory subtype and that pain may be driven by distinct mechanisms in the other patients,” the study authors wrote.
The low inflammatory subgroup was characterized by high neuronal and glycoprotein gene expression. But in this group, pain scores were not associated with elevated inflammatory markers.
“It is interesting that this subtype is characterized by a paucity of inflammatory infiltrates, yet maintains high pain scores and multiple tender/swollen joints – this too is consistent with other findings of patients with established RA,” the research team noted.
The mixed subtype shared features with both the high and low subtypes, the researchers said.
“Our work suggests that RA patients with longstanding disease and poor response to anti-inflammatory treatment may warrant synovial biopsy to determine their inflammatory subtype,” the researchers concluded.
Several research institutions and the Accelerating Medicines Partnership in Rheumatoid Arthritis and Lupus Network, a public-private partnership involving several pharmaceutical companies, patient advocacy groups, and the National Institutes of Health, funded the study.
SOURCE: Orange D et al. Arthritis Rheumatol. 2018 Feb 22. doi: 10.1002/art.40428.
Researchers have identified three different synovial subtypes of rheumatoid arthritis that exhibit different mechanisms of pain and correlate with specific clinical phenotypes.
The findings could be clinically meaningful and may help guide optimal treatment strategies for patients, as well as provide a better understanding of the cause of pain in patients with high tender and swollen joint counts but little tissue inflammation, according to the research team led by Dana E. Orange, MD, of the Hospital for Special Surgery and Rockefeller University in New York.
The report was published in Arthritis & Rheumatology.
The assessment of the synovium in rheumatoid arthritis (RA) has the potential to provide guidance on optimal treatment strategies, they noted, but its classification has not yet factored into current diagnosis or treatment guidelines of RA.
In total, the research team analyzed 20 histologic features on 129 synovial tissue samples.
The researchers used machine learning integration to identify three distinct molecular subtypes of RA from a consensus clustering of the 500 most variable genes expressed in a subset of 45 synovial samples, including 39 from RA patients. The subtypes were high inflammatory, low inflammatory, and a mixed phenotype.
The researchers then took the histologic features that best corresponded to each subtype to develop a histology scoring algorithm that predicted the three gene expression subtypes (using only histology features), each of which were each associated with levels of erythrocyte sedimentation rate, C-reactive protein, and autoantibodies.
The histologic features that most strongly defined the high inflammatory subtype included three plasma cell features: binucleate plasma cells, plasma cell percentage, and Russell bodies. Patients with a high inflammatory synovial subtype also exhibited higher levels of markers of systemic inflammation and autoantibodies. For example, C-reactive protein was significantly correlated with pain in the high inflammatory group.
“This suggests that pain is associated with inflammation in patients with high inflammatory subtype and that pain may be driven by distinct mechanisms in the other patients,” the study authors wrote.
The low inflammatory subgroup was characterized by high neuronal and glycoprotein gene expression. But in this group, pain scores were not associated with elevated inflammatory markers.
“It is interesting that this subtype is characterized by a paucity of inflammatory infiltrates, yet maintains high pain scores and multiple tender/swollen joints – this too is consistent with other findings of patients with established RA,” the research team noted.
The mixed subtype shared features with both the high and low subtypes, the researchers said.
“Our work suggests that RA patients with longstanding disease and poor response to anti-inflammatory treatment may warrant synovial biopsy to determine their inflammatory subtype,” the researchers concluded.
Several research institutions and the Accelerating Medicines Partnership in Rheumatoid Arthritis and Lupus Network, a public-private partnership involving several pharmaceutical companies, patient advocacy groups, and the National Institutes of Health, funded the study.
SOURCE: Orange D et al. Arthritis Rheumatol. 2018 Feb 22. doi: 10.1002/art.40428.
Researchers have identified three different synovial subtypes of rheumatoid arthritis that exhibit different mechanisms of pain and correlate with specific clinical phenotypes.
The findings could be clinically meaningful and may help guide optimal treatment strategies for patients, as well as provide a better understanding of the cause of pain in patients with high tender and swollen joint counts but little tissue inflammation, according to the research team led by Dana E. Orange, MD, of the Hospital for Special Surgery and Rockefeller University in New York.
The report was published in Arthritis & Rheumatology.
The assessment of the synovium in rheumatoid arthritis (RA) has the potential to provide guidance on optimal treatment strategies, they noted, but its classification has not yet factored into current diagnosis or treatment guidelines of RA.
In total, the research team analyzed 20 histologic features on 129 synovial tissue samples.
The researchers used machine learning integration to identify three distinct molecular subtypes of RA from a consensus clustering of the 500 most variable genes expressed in a subset of 45 synovial samples, including 39 from RA patients. The subtypes were high inflammatory, low inflammatory, and a mixed phenotype.
The researchers then took the histologic features that best corresponded to each subtype to develop a histology scoring algorithm that predicted the three gene expression subtypes (using only histology features), each of which were each associated with levels of erythrocyte sedimentation rate, C-reactive protein, and autoantibodies.
The histologic features that most strongly defined the high inflammatory subtype included three plasma cell features: binucleate plasma cells, plasma cell percentage, and Russell bodies. Patients with a high inflammatory synovial subtype also exhibited higher levels of markers of systemic inflammation and autoantibodies. For example, C-reactive protein was significantly correlated with pain in the high inflammatory group.
“This suggests that pain is associated with inflammation in patients with high inflammatory subtype and that pain may be driven by distinct mechanisms in the other patients,” the study authors wrote.
The low inflammatory subgroup was characterized by high neuronal and glycoprotein gene expression. But in this group, pain scores were not associated with elevated inflammatory markers.
“It is interesting that this subtype is characterized by a paucity of inflammatory infiltrates, yet maintains high pain scores and multiple tender/swollen joints – this too is consistent with other findings of patients with established RA,” the research team noted.
The mixed subtype shared features with both the high and low subtypes, the researchers said.
“Our work suggests that RA patients with longstanding disease and poor response to anti-inflammatory treatment may warrant synovial biopsy to determine their inflammatory subtype,” the researchers concluded.
Several research institutions and the Accelerating Medicines Partnership in Rheumatoid Arthritis and Lupus Network, a public-private partnership involving several pharmaceutical companies, patient advocacy groups, and the National Institutes of Health, funded the study.
SOURCE: Orange D et al. Arthritis Rheumatol. 2018 Feb 22. doi: 10.1002/art.40428.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Mechanisms of pain may differ in RA patients with different synovial subtypes.
Major findings:
Study details: Twenty histologic features were assessed on 129 synovial tissue samples from 123 RA patients and 6 OA patients.
Disclosures: Several research institutions and the Accelerating Medicines Partnership in Rheumatoid Arthritis and Lupus Network, a public-private partnership involving several pharmaceutical companies, patient advocacy groups, and the National Institutes of Health, funded the study.
Source: Orange D et al. Arthritis Rheumatol. 2018 Feb 22. doi: 10.1002/art.40428.
Badly behaved neutrophils are novel target in rheumatic diseases
MAUI, HAWAII – For most of her career as a pediatric rheumatologist and immunologist, Anne M. Stevens, MD, PhD, didn’t find neutrophils terribly interesting.
“I’m more of a T-cell person. T cells are very precise, they’re regulated, they’re very sophisticated cells. Neutrophils are like these bumbling idiots that just come in and hit everything in sight and cause all sorts of damage,” she observed at the 2018 Rheumatology Winter Clinical Symposium.
Recently, however, she’s changed her mind. Research in the past few years demonstrates that there’s a lot more to neutrophils than previously known. Neutrophils are not only the first-responder immune cells to acutely inflamed tissue, as has long been recognized, but they also play a huge role in initiating and perpetuating chronic inflammation. Indeed, neutrophils figure prominently in the pathogenesis of rheumatoid arthritis, systemic lupus erythematosus (SLE), antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, and perhaps in major nonautoimmune diseases as well, including atherosclerosis, type 1 diabetes, thrombosis, and some forms of kidney disease, explained Dr. Stevens, chief of pediatric rheumatology at the University of Washington, Seattle.
Indeed, it’s now clear that neutrophils can either die quietly or become activated in death by creating neutrophil extracellular traps, or NETs, in a process called NETosis. Even though the activated neutrophil is dead, its exteriorized NETs continue to function, grabbing and killing bacterial pathogens. But the NETs also attract immune cells. These NETs are long strands of sticky DNA containing chromatin, histones, elastase, myeloperoxidase, hypercitrullinated proteins, and other autoantigens. NETosis exposes these autoantigens to the immune system, with resultant generation of autoimmune responses in predisposed individuals.
Much detail has been learned about this process. For example, one type of neutrophil death results from lymphokine-activated killer cells releasing perforin, which pokes holes in the neutrophil cell membrane, allowing an influx of calcium. The inflow of calcium triggers activation of peptidyl arginine deiminases, and these enzymes in turn cause hypercitrullination of autoantigens, leading to formation of anti–citrullinated peptide autoantibodies (ACPAs). These ACPAs cause inflammation by inducing complement activation and binding to Fc gamma receptors on phagocytes.
Investigators at the Systemic Autoimmunity Branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other research centers have shown that mixing RA synovial fibroblasts with NETs induces production of interleukin-6 and ACPAs. In a mouse model of RA, this leads to cartilage loss in joints, providing a plausible mechanistic explanation for joint damage in RA (Sci Immunol. 2017 Apr;2[10]:eaag3358).
Potential therapeutic strategies targeting NETosis
The various treatment approaches under study aim to either inhibit NET release, curb recruitment of neutrophils for activation, promote migration of neutrophils away from sites of inflammation, or foster efferocytosis.
Among the therapeutic possibilities are calcineurin inhibitors as a means of preventing the influx of calcium into neutrophils, peptidyl arginine deiminase inhibitors such as Cl-amidine to prevent hypercitrullination, complement component 5a-receptor antagonists to decrease NET formation, the myeloperoxidase inhibitor known for now as PF-1355, and N-acetyl cysteine to scavenge proinflammatory reactive oxygen species and thereby reduce NET release.
The key will be to develop highly selective agents that encourage well-behaved neutrophils; across-the-board blockade of neutrophil activity would be terribly immunosuppressive and likely dangerous.
“What if we could block neutrophil recruitment just to the organ we’re worried about? If I’m worried about nephritis, let’s just block neutrophil infiltration into the kidneys. There are drugs being developed that will block the specific types of integrins involved,” Dr. Stevens said.
She reported having no financial conflicts regarding her presentation.
MAUI, HAWAII – For most of her career as a pediatric rheumatologist and immunologist, Anne M. Stevens, MD, PhD, didn’t find neutrophils terribly interesting.
“I’m more of a T-cell person. T cells are very precise, they’re regulated, they’re very sophisticated cells. Neutrophils are like these bumbling idiots that just come in and hit everything in sight and cause all sorts of damage,” she observed at the 2018 Rheumatology Winter Clinical Symposium.
Recently, however, she’s changed her mind. Research in the past few years demonstrates that there’s a lot more to neutrophils than previously known. Neutrophils are not only the first-responder immune cells to acutely inflamed tissue, as has long been recognized, but they also play a huge role in initiating and perpetuating chronic inflammation. Indeed, neutrophils figure prominently in the pathogenesis of rheumatoid arthritis, systemic lupus erythematosus (SLE), antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, and perhaps in major nonautoimmune diseases as well, including atherosclerosis, type 1 diabetes, thrombosis, and some forms of kidney disease, explained Dr. Stevens, chief of pediatric rheumatology at the University of Washington, Seattle.
Indeed, it’s now clear that neutrophils can either die quietly or become activated in death by creating neutrophil extracellular traps, or NETs, in a process called NETosis. Even though the activated neutrophil is dead, its exteriorized NETs continue to function, grabbing and killing bacterial pathogens. But the NETs also attract immune cells. These NETs are long strands of sticky DNA containing chromatin, histones, elastase, myeloperoxidase, hypercitrullinated proteins, and other autoantigens. NETosis exposes these autoantigens to the immune system, with resultant generation of autoimmune responses in predisposed individuals.
Much detail has been learned about this process. For example, one type of neutrophil death results from lymphokine-activated killer cells releasing perforin, which pokes holes in the neutrophil cell membrane, allowing an influx of calcium. The inflow of calcium triggers activation of peptidyl arginine deiminases, and these enzymes in turn cause hypercitrullination of autoantigens, leading to formation of anti–citrullinated peptide autoantibodies (ACPAs). These ACPAs cause inflammation by inducing complement activation and binding to Fc gamma receptors on phagocytes.
Investigators at the Systemic Autoimmunity Branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other research centers have shown that mixing RA synovial fibroblasts with NETs induces production of interleukin-6 and ACPAs. In a mouse model of RA, this leads to cartilage loss in joints, providing a plausible mechanistic explanation for joint damage in RA (Sci Immunol. 2017 Apr;2[10]:eaag3358).
Potential therapeutic strategies targeting NETosis
The various treatment approaches under study aim to either inhibit NET release, curb recruitment of neutrophils for activation, promote migration of neutrophils away from sites of inflammation, or foster efferocytosis.
Among the therapeutic possibilities are calcineurin inhibitors as a means of preventing the influx of calcium into neutrophils, peptidyl arginine deiminase inhibitors such as Cl-amidine to prevent hypercitrullination, complement component 5a-receptor antagonists to decrease NET formation, the myeloperoxidase inhibitor known for now as PF-1355, and N-acetyl cysteine to scavenge proinflammatory reactive oxygen species and thereby reduce NET release.
The key will be to develop highly selective agents that encourage well-behaved neutrophils; across-the-board blockade of neutrophil activity would be terribly immunosuppressive and likely dangerous.
“What if we could block neutrophil recruitment just to the organ we’re worried about? If I’m worried about nephritis, let’s just block neutrophil infiltration into the kidneys. There are drugs being developed that will block the specific types of integrins involved,” Dr. Stevens said.
She reported having no financial conflicts regarding her presentation.
MAUI, HAWAII – For most of her career as a pediatric rheumatologist and immunologist, Anne M. Stevens, MD, PhD, didn’t find neutrophils terribly interesting.
“I’m more of a T-cell person. T cells are very precise, they’re regulated, they’re very sophisticated cells. Neutrophils are like these bumbling idiots that just come in and hit everything in sight and cause all sorts of damage,” she observed at the 2018 Rheumatology Winter Clinical Symposium.
Recently, however, she’s changed her mind. Research in the past few years demonstrates that there’s a lot more to neutrophils than previously known. Neutrophils are not only the first-responder immune cells to acutely inflamed tissue, as has long been recognized, but they also play a huge role in initiating and perpetuating chronic inflammation. Indeed, neutrophils figure prominently in the pathogenesis of rheumatoid arthritis, systemic lupus erythematosus (SLE), antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis, and perhaps in major nonautoimmune diseases as well, including atherosclerosis, type 1 diabetes, thrombosis, and some forms of kidney disease, explained Dr. Stevens, chief of pediatric rheumatology at the University of Washington, Seattle.
Indeed, it’s now clear that neutrophils can either die quietly or become activated in death by creating neutrophil extracellular traps, or NETs, in a process called NETosis. Even though the activated neutrophil is dead, its exteriorized NETs continue to function, grabbing and killing bacterial pathogens. But the NETs also attract immune cells. These NETs are long strands of sticky DNA containing chromatin, histones, elastase, myeloperoxidase, hypercitrullinated proteins, and other autoantigens. NETosis exposes these autoantigens to the immune system, with resultant generation of autoimmune responses in predisposed individuals.
Much detail has been learned about this process. For example, one type of neutrophil death results from lymphokine-activated killer cells releasing perforin, which pokes holes in the neutrophil cell membrane, allowing an influx of calcium. The inflow of calcium triggers activation of peptidyl arginine deiminases, and these enzymes in turn cause hypercitrullination of autoantigens, leading to formation of anti–citrullinated peptide autoantibodies (ACPAs). These ACPAs cause inflammation by inducing complement activation and binding to Fc gamma receptors on phagocytes.
Investigators at the Systemic Autoimmunity Branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases and several other research centers have shown that mixing RA synovial fibroblasts with NETs induces production of interleukin-6 and ACPAs. In a mouse model of RA, this leads to cartilage loss in joints, providing a plausible mechanistic explanation for joint damage in RA (Sci Immunol. 2017 Apr;2[10]:eaag3358).
Potential therapeutic strategies targeting NETosis
The various treatment approaches under study aim to either inhibit NET release, curb recruitment of neutrophils for activation, promote migration of neutrophils away from sites of inflammation, or foster efferocytosis.
Among the therapeutic possibilities are calcineurin inhibitors as a means of preventing the influx of calcium into neutrophils, peptidyl arginine deiminase inhibitors such as Cl-amidine to prevent hypercitrullination, complement component 5a-receptor antagonists to decrease NET formation, the myeloperoxidase inhibitor known for now as PF-1355, and N-acetyl cysteine to scavenge proinflammatory reactive oxygen species and thereby reduce NET release.
The key will be to develop highly selective agents that encourage well-behaved neutrophils; across-the-board blockade of neutrophil activity would be terribly immunosuppressive and likely dangerous.
“What if we could block neutrophil recruitment just to the organ we’re worried about? If I’m worried about nephritis, let’s just block neutrophil infiltration into the kidneys. There are drugs being developed that will block the specific types of integrins involved,” Dr. Stevens said.
She reported having no financial conflicts regarding her presentation.
EXPERT ANALYSIS FROM RWCS 2018
Treatment priorities often differ between RA patients, clinicians
Patients with RA and their clinicians approach treatment goals differently based on knowledge, illness experience, and competing priorities, according to findings from a qualitative study.
The results of this study underscore “the tension between having an explicit shared goal between clinicians and patients, and experiencing inherently different – and at times opposing – conceptualization of how one formulates or achieves said goal,” wrote Jennifer L. Barton, MD, of the Department of Veteran Affairs Portland Health Care System and Oregon Health & Science University, Portland, and her coauthors.
Two domains – disease knowledge and psychosocial dynamics – emerged across all focus groups, Dr. Barton and her colleagues reported in Arthritis Care & Research.
Disease knowledge and education
In the knowledge domain, themes that emerged were informed choice, medication adherence and safety, and clinician assumption of patients’ inability to interpret information. Patients disclosed, for instance, a desire for more information on disease progression and medication side effects to inform future clinician visits, and often sought such information on the Internet.
“Patients highlighted the importance of RA knowledge to understanding what was happening to them physically and the impact of medication on their bodies, and their need to seek information outside of the clinical visit,” the authors noted.
Whereas patients discussed medications in the context of informed decision making, “many clinicians connected RA education to risk of nonadherence and medication safety,” the authors reported. Additionally, “patients did not discuss their adherence to clinician treatment recommendations, though patients expressed dissatisfaction with clinicians who they believed dismissed their medication concerns.”
Some clinicians expressed frustration with patients’ self-education efforts and attitudes toward alternative medicine, and patients reported feeling as though doctors had “diminished the importance of information” they shared.
“Several clinicians voiced perceived paradoxes in current expectations of their professional role as an expert who also defers to patient preferences,” the authors said. “Many clinicians voiced frustration with patients seeking knowledge from what they considered unreliable sources, which prompted varying levels of comfort among clinicians with some adopting a more paternalistic stance.”
Psychosocial dynamics of RA illness
The psychosocial dynamics domain focused on stress and found that patients’ experiences with RA informed their treatment preferences and affected patient-provider communication. For instance, patients who experienced inability to participate in activities because of pain or fatigue prioritized treatment goals aimed at pain reduction and increased energy, with minimal side effects.
“In contrast, clinicians talked about using objective clinical markers” to inform treatment strategy, the authors said. Although they acknowledged the psychosocial stress experienced by patients, clinicians cited limited time and resources as a main reason for their inability to adequately address these concerns.
Both patients and clinicians acknowledged the role of fear in the disease experience, with providers asserting that patients’ fear “disrupted effective communication and complicated patient willingness to follow treatment recommendations,” the authors said.
Lastly, both patients and clinicians described treatment decisions as a “negotiation” in which patients’ priorities of quality of life improvements, such as pain reduction, often were at odds with objective clinical goals of the provider, such as addressing underlying damage.
“Patients indicated that clinician goals focused on objective clinical markers and helping patients achieve remission; however, patients expressed a desire for clinicians to look beyond clinical markers and consider patients’ quality of life goals as well as being open to multiple treatment possibilities,” the authors said.
Areas for improvement
The results of the study highlight potential areas for improvement in patient-clinician communication, namely by balancing the imparting of clinician knowledge with sincere consideration of patient preferences and priorities. “The mismatch in attitudes towards the goal of knowledge between patients and clinicians may lead to suboptimal communication and lack of trust,” the authors wrote. “Patients’ desire for information on a range of RA topics is important, but the value attached to that knowledge is where patients and clinicians diverge.”
Resources aimed at facilitating a conversation around goals may lead to greater goal concordance, which could potentially result in more high value treatment, the authors concluded. “With tools and training to support patient goal-directed care in rheumatology, improved outcomes and reduced disparities may be achieved.”
The study was funded by a grant to Dr. Barton from the National Institutes of Health.
SOURCE: Barton JL et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23541.
Patients with RA and their clinicians approach treatment goals differently based on knowledge, illness experience, and competing priorities, according to findings from a qualitative study.
The results of this study underscore “the tension between having an explicit shared goal between clinicians and patients, and experiencing inherently different – and at times opposing – conceptualization of how one formulates or achieves said goal,” wrote Jennifer L. Barton, MD, of the Department of Veteran Affairs Portland Health Care System and Oregon Health & Science University, Portland, and her coauthors.
Two domains – disease knowledge and psychosocial dynamics – emerged across all focus groups, Dr. Barton and her colleagues reported in Arthritis Care & Research.
Disease knowledge and education
In the knowledge domain, themes that emerged were informed choice, medication adherence and safety, and clinician assumption of patients’ inability to interpret information. Patients disclosed, for instance, a desire for more information on disease progression and medication side effects to inform future clinician visits, and often sought such information on the Internet.
“Patients highlighted the importance of RA knowledge to understanding what was happening to them physically and the impact of medication on their bodies, and their need to seek information outside of the clinical visit,” the authors noted.
Whereas patients discussed medications in the context of informed decision making, “many clinicians connected RA education to risk of nonadherence and medication safety,” the authors reported. Additionally, “patients did not discuss their adherence to clinician treatment recommendations, though patients expressed dissatisfaction with clinicians who they believed dismissed their medication concerns.”
Some clinicians expressed frustration with patients’ self-education efforts and attitudes toward alternative medicine, and patients reported feeling as though doctors had “diminished the importance of information” they shared.
“Several clinicians voiced perceived paradoxes in current expectations of their professional role as an expert who also defers to patient preferences,” the authors said. “Many clinicians voiced frustration with patients seeking knowledge from what they considered unreliable sources, which prompted varying levels of comfort among clinicians with some adopting a more paternalistic stance.”
Psychosocial dynamics of RA illness
The psychosocial dynamics domain focused on stress and found that patients’ experiences with RA informed their treatment preferences and affected patient-provider communication. For instance, patients who experienced inability to participate in activities because of pain or fatigue prioritized treatment goals aimed at pain reduction and increased energy, with minimal side effects.
“In contrast, clinicians talked about using objective clinical markers” to inform treatment strategy, the authors said. Although they acknowledged the psychosocial stress experienced by patients, clinicians cited limited time and resources as a main reason for their inability to adequately address these concerns.
Both patients and clinicians acknowledged the role of fear in the disease experience, with providers asserting that patients’ fear “disrupted effective communication and complicated patient willingness to follow treatment recommendations,” the authors said.
Lastly, both patients and clinicians described treatment decisions as a “negotiation” in which patients’ priorities of quality of life improvements, such as pain reduction, often were at odds with objective clinical goals of the provider, such as addressing underlying damage.
“Patients indicated that clinician goals focused on objective clinical markers and helping patients achieve remission; however, patients expressed a desire for clinicians to look beyond clinical markers and consider patients’ quality of life goals as well as being open to multiple treatment possibilities,” the authors said.
Areas for improvement
The results of the study highlight potential areas for improvement in patient-clinician communication, namely by balancing the imparting of clinician knowledge with sincere consideration of patient preferences and priorities. “The mismatch in attitudes towards the goal of knowledge between patients and clinicians may lead to suboptimal communication and lack of trust,” the authors wrote. “Patients’ desire for information on a range of RA topics is important, but the value attached to that knowledge is where patients and clinicians diverge.”
Resources aimed at facilitating a conversation around goals may lead to greater goal concordance, which could potentially result in more high value treatment, the authors concluded. “With tools and training to support patient goal-directed care in rheumatology, improved outcomes and reduced disparities may be achieved.”
The study was funded by a grant to Dr. Barton from the National Institutes of Health.
SOURCE: Barton JL et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23541.
Patients with RA and their clinicians approach treatment goals differently based on knowledge, illness experience, and competing priorities, according to findings from a qualitative study.
The results of this study underscore “the tension between having an explicit shared goal between clinicians and patients, and experiencing inherently different – and at times opposing – conceptualization of how one formulates or achieves said goal,” wrote Jennifer L. Barton, MD, of the Department of Veteran Affairs Portland Health Care System and Oregon Health & Science University, Portland, and her coauthors.
Two domains – disease knowledge and psychosocial dynamics – emerged across all focus groups, Dr. Barton and her colleagues reported in Arthritis Care & Research.
Disease knowledge and education
In the knowledge domain, themes that emerged were informed choice, medication adherence and safety, and clinician assumption of patients’ inability to interpret information. Patients disclosed, for instance, a desire for more information on disease progression and medication side effects to inform future clinician visits, and often sought such information on the Internet.
“Patients highlighted the importance of RA knowledge to understanding what was happening to them physically and the impact of medication on their bodies, and their need to seek information outside of the clinical visit,” the authors noted.
Whereas patients discussed medications in the context of informed decision making, “many clinicians connected RA education to risk of nonadherence and medication safety,” the authors reported. Additionally, “patients did not discuss their adherence to clinician treatment recommendations, though patients expressed dissatisfaction with clinicians who they believed dismissed their medication concerns.”
Some clinicians expressed frustration with patients’ self-education efforts and attitudes toward alternative medicine, and patients reported feeling as though doctors had “diminished the importance of information” they shared.
“Several clinicians voiced perceived paradoxes in current expectations of their professional role as an expert who also defers to patient preferences,” the authors said. “Many clinicians voiced frustration with patients seeking knowledge from what they considered unreliable sources, which prompted varying levels of comfort among clinicians with some adopting a more paternalistic stance.”
Psychosocial dynamics of RA illness
The psychosocial dynamics domain focused on stress and found that patients’ experiences with RA informed their treatment preferences and affected patient-provider communication. For instance, patients who experienced inability to participate in activities because of pain or fatigue prioritized treatment goals aimed at pain reduction and increased energy, with minimal side effects.
“In contrast, clinicians talked about using objective clinical markers” to inform treatment strategy, the authors said. Although they acknowledged the psychosocial stress experienced by patients, clinicians cited limited time and resources as a main reason for their inability to adequately address these concerns.
Both patients and clinicians acknowledged the role of fear in the disease experience, with providers asserting that patients’ fear “disrupted effective communication and complicated patient willingness to follow treatment recommendations,” the authors said.
Lastly, both patients and clinicians described treatment decisions as a “negotiation” in which patients’ priorities of quality of life improvements, such as pain reduction, often were at odds with objective clinical goals of the provider, such as addressing underlying damage.
“Patients indicated that clinician goals focused on objective clinical markers and helping patients achieve remission; however, patients expressed a desire for clinicians to look beyond clinical markers and consider patients’ quality of life goals as well as being open to multiple treatment possibilities,” the authors said.
Areas for improvement
The results of the study highlight potential areas for improvement in patient-clinician communication, namely by balancing the imparting of clinician knowledge with sincere consideration of patient preferences and priorities. “The mismatch in attitudes towards the goal of knowledge between patients and clinicians may lead to suboptimal communication and lack of trust,” the authors wrote. “Patients’ desire for information on a range of RA topics is important, but the value attached to that knowledge is where patients and clinicians diverge.”
Resources aimed at facilitating a conversation around goals may lead to greater goal concordance, which could potentially result in more high value treatment, the authors concluded. “With tools and training to support patient goal-directed care in rheumatology, improved outcomes and reduced disparities may be achieved.”
The study was funded by a grant to Dr. Barton from the National Institutes of Health.
SOURCE: Barton JL et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23541.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: In the knowledge domain, recurrent issues were informed choice, medication adherence, and clinician assumption of patient inability to interpret information; the psychosocial dynamics domain found that patient illness experience affected treatment decisions and patient-provider communication.
Data source: A qualitative focus group study of 19 RA patients and 18 clinicians.
Disclosures: The study was funded by a grant to Dr. Barton from the National Institutes of Health.
Source: Barton JL et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23541.
RA associated with higher risk of psychiatric disorders
The incidence and prevalence of anxiety disorder, depression, and bipolar disorder are higher among patients with rheumatoid arthritis than individuals from the general population, according to findings from a Canadian retrospective matched cohort study.
In the study of 10,206 rheumatoid arthritis (RA) patients and 50,960 individuals matched from the general population of Manitoba between 1989 and 2012, depression incidence was higher in the RA group, compared with the matched group, when adjusted for factors including age, sex, year, region of residence, and socioeconomic status (incidence rate ratio = 1.46; 95% confidence interval, 1.35-1.58). Incidence of anxiety disorder (IRR = 1.24; 95% CI, 1.15-1.34) and bipolar disorder (IRR = 1.21; 95% CI, 1.00-1.47) were also higher in the RA group. The incidence of schizophrenia did not differ between groups (IRR = 0.96; 95% CI, 0.61-1.50), Ruth Ann Marrie, MD, PhD, of the University of Manitoba, Winnipeg, and her coauthors reported in Arthritis Care & Research.
To estimate psychiatric disorder incidence after RA diagnosis (or the index date in the matched population), the first claim had to occur after the index date, and had to be preceded by a 5-year period with no claims for that psychiatric disorder. To estimate lifetime prevalence, once a patient met the case definition for a disorder, he or she was considered affected in all subsequent years if alive and a Manitoba resident. To account for varying periods of remission, however, annual period prevalence was defined as a patient having one or more hospital claims or two or more physician claims for the disorder in that year, Dr. Marrie and her colleagues wrote.
The adjusted lifetime prevalence was also higher in the RA group for both depression (PR = 1.35; 95% CI, 1.26-1.45) and anxiety disorder (PR = 1.20; 95% CI, 1.13-1.27), as was the annual period prevalence of depression (PR = 1.36; 95% CI, 1.26-1.47) and anxiety disorder (PR = 1.30; 95% CI, 1.19-1.41). Neither lifetime prevalence of bipolar disorder (PR = 1.13; 95% CI, 0.95-1.36) and schizophrenia (PR = 1.02; 95% CI, 0.72-1.43) nor annual period prevalence of bipolar disorder (PR = 1.06; 95% CI, 0.86-1.31) and schizophrenia (PR = 0.68; 95% CI, 0.40-1.15) differed between the RA and matched cohorts, the authors reported.
Female sex was associated with risk of psychiatric disease, as was lower socioeconomic status and living in an urban area, the authors reported.
Although the study had strengths including a large study population and use of population-based data, it did not evaluate psychiatric multimorbidity, a “common and clinically relevant issue which may affect outcomes,” Dr. Marrie and her coauthors said in the report. Additionally, the use of administrative data makes it difficult to account for care provided by nonphysician providers, such as psychologists, and for conditions that cause symptoms but do not meet diagnostic criteria, the authors noted.
“Future studies should explore these issues in population-based clinical cohorts which comprehensively evaluate multiple psychiatric disorders,” they concluded.
The study was funded by the Canadian Institutes of Health Research and the Waugh Family Chair in Multiple Sclerosis. Dr. Marrie has conducted clinical trials for Sanofi Aventis. Two other authors disclosed financial ties to pharmaceutical companies.
SOURCE: Marrie R et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23539.
The incidence and prevalence of anxiety disorder, depression, and bipolar disorder are higher among patients with rheumatoid arthritis than individuals from the general population, according to findings from a Canadian retrospective matched cohort study.
In the study of 10,206 rheumatoid arthritis (RA) patients and 50,960 individuals matched from the general population of Manitoba between 1989 and 2012, depression incidence was higher in the RA group, compared with the matched group, when adjusted for factors including age, sex, year, region of residence, and socioeconomic status (incidence rate ratio = 1.46; 95% confidence interval, 1.35-1.58). Incidence of anxiety disorder (IRR = 1.24; 95% CI, 1.15-1.34) and bipolar disorder (IRR = 1.21; 95% CI, 1.00-1.47) were also higher in the RA group. The incidence of schizophrenia did not differ between groups (IRR = 0.96; 95% CI, 0.61-1.50), Ruth Ann Marrie, MD, PhD, of the University of Manitoba, Winnipeg, and her coauthors reported in Arthritis Care & Research.
To estimate psychiatric disorder incidence after RA diagnosis (or the index date in the matched population), the first claim had to occur after the index date, and had to be preceded by a 5-year period with no claims for that psychiatric disorder. To estimate lifetime prevalence, once a patient met the case definition for a disorder, he or she was considered affected in all subsequent years if alive and a Manitoba resident. To account for varying periods of remission, however, annual period prevalence was defined as a patient having one or more hospital claims or two or more physician claims for the disorder in that year, Dr. Marrie and her colleagues wrote.
The adjusted lifetime prevalence was also higher in the RA group for both depression (PR = 1.35; 95% CI, 1.26-1.45) and anxiety disorder (PR = 1.20; 95% CI, 1.13-1.27), as was the annual period prevalence of depression (PR = 1.36; 95% CI, 1.26-1.47) and anxiety disorder (PR = 1.30; 95% CI, 1.19-1.41). Neither lifetime prevalence of bipolar disorder (PR = 1.13; 95% CI, 0.95-1.36) and schizophrenia (PR = 1.02; 95% CI, 0.72-1.43) nor annual period prevalence of bipolar disorder (PR = 1.06; 95% CI, 0.86-1.31) and schizophrenia (PR = 0.68; 95% CI, 0.40-1.15) differed between the RA and matched cohorts, the authors reported.
Female sex was associated with risk of psychiatric disease, as was lower socioeconomic status and living in an urban area, the authors reported.
Although the study had strengths including a large study population and use of population-based data, it did not evaluate psychiatric multimorbidity, a “common and clinically relevant issue which may affect outcomes,” Dr. Marrie and her coauthors said in the report. Additionally, the use of administrative data makes it difficult to account for care provided by nonphysician providers, such as psychologists, and for conditions that cause symptoms but do not meet diagnostic criteria, the authors noted.
“Future studies should explore these issues in population-based clinical cohorts which comprehensively evaluate multiple psychiatric disorders,” they concluded.
The study was funded by the Canadian Institutes of Health Research and the Waugh Family Chair in Multiple Sclerosis. Dr. Marrie has conducted clinical trials for Sanofi Aventis. Two other authors disclosed financial ties to pharmaceutical companies.
SOURCE: Marrie R et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23539.
The incidence and prevalence of anxiety disorder, depression, and bipolar disorder are higher among patients with rheumatoid arthritis than individuals from the general population, according to findings from a Canadian retrospective matched cohort study.
In the study of 10,206 rheumatoid arthritis (RA) patients and 50,960 individuals matched from the general population of Manitoba between 1989 and 2012, depression incidence was higher in the RA group, compared with the matched group, when adjusted for factors including age, sex, year, region of residence, and socioeconomic status (incidence rate ratio = 1.46; 95% confidence interval, 1.35-1.58). Incidence of anxiety disorder (IRR = 1.24; 95% CI, 1.15-1.34) and bipolar disorder (IRR = 1.21; 95% CI, 1.00-1.47) were also higher in the RA group. The incidence of schizophrenia did not differ between groups (IRR = 0.96; 95% CI, 0.61-1.50), Ruth Ann Marrie, MD, PhD, of the University of Manitoba, Winnipeg, and her coauthors reported in Arthritis Care & Research.
To estimate psychiatric disorder incidence after RA diagnosis (or the index date in the matched population), the first claim had to occur after the index date, and had to be preceded by a 5-year period with no claims for that psychiatric disorder. To estimate lifetime prevalence, once a patient met the case definition for a disorder, he or she was considered affected in all subsequent years if alive and a Manitoba resident. To account for varying periods of remission, however, annual period prevalence was defined as a patient having one or more hospital claims or two or more physician claims for the disorder in that year, Dr. Marrie and her colleagues wrote.
The adjusted lifetime prevalence was also higher in the RA group for both depression (PR = 1.35; 95% CI, 1.26-1.45) and anxiety disorder (PR = 1.20; 95% CI, 1.13-1.27), as was the annual period prevalence of depression (PR = 1.36; 95% CI, 1.26-1.47) and anxiety disorder (PR = 1.30; 95% CI, 1.19-1.41). Neither lifetime prevalence of bipolar disorder (PR = 1.13; 95% CI, 0.95-1.36) and schizophrenia (PR = 1.02; 95% CI, 0.72-1.43) nor annual period prevalence of bipolar disorder (PR = 1.06; 95% CI, 0.86-1.31) and schizophrenia (PR = 0.68; 95% CI, 0.40-1.15) differed between the RA and matched cohorts, the authors reported.
Female sex was associated with risk of psychiatric disease, as was lower socioeconomic status and living in an urban area, the authors reported.
Although the study had strengths including a large study population and use of population-based data, it did not evaluate psychiatric multimorbidity, a “common and clinically relevant issue which may affect outcomes,” Dr. Marrie and her coauthors said in the report. Additionally, the use of administrative data makes it difficult to account for care provided by nonphysician providers, such as psychologists, and for conditions that cause symptoms but do not meet diagnostic criteria, the authors noted.
“Future studies should explore these issues in population-based clinical cohorts which comprehensively evaluate multiple psychiatric disorders,” they concluded.
The study was funded by the Canadian Institutes of Health Research and the Waugh Family Chair in Multiple Sclerosis. Dr. Marrie has conducted clinical trials for Sanofi Aventis. Two other authors disclosed financial ties to pharmaceutical companies.
SOURCE: Marrie R et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23539.
FROM ARTHRITIS CARE & RESEARCH
Key clinical point:
Major finding: Incidence of depression (IRR = 1.46; 95% CI, 1.35-1.58), anxiety disorder (IRR = 1.24; 95% CI, 1.15-1.34), and bipolar disorder (IRR = 1.21; 95% CI, 1.00-1.47) were higher in the RA group than in the matched group.
Data source: A retrospective matched cohort study of 10,206 RA patients and 50,960 matched individuals from the general population between 1989 and 2012.
Disclosures: The study was funded by the Canadian Institutes of Health Research and the Waugh Family Chair in Multiple Sclerosis. Dr. Marrie has conducted clinical trials for Sanofi Aventis. Two other authors disclosed financial ties to pharmaceutical companies.
Source: Marrie R et al. Arthritis Care Res. 2018 Feb 13. doi: 10.1002/acr.23539
RF-positive polyarticular JIA looks like adult RA
New evidence suggests the rheumatoid factor–positive polyarticular subtype of juvenile idiopathic arthritis bears a close genetic resemblance to adult rheumatoid arthritis, lending support to the growing suspicion that in arthritis, biological underpinnings are more important than age of onset when it comes to characterizing and, potentially, choosing treatments.
Previous work had shown that rheumatoid factor (RF)–negative patients have genetic risks similar to those of adults with RF-negative disease. “If the RF-negative patients in adult and childhood are similar, then maybe the RF-positive patients are similar in their genetic background as well. That’s what this study was testing,” study coauthor Anne M. Stevens, MD, PhD, division chief of rheumatology at the University of Washington, Seattle, said in an interview. The study was published online Feb. 9 in Arthritis & Rheumatology.
There are seven recognized categories of juvenile idiopathic arthritis (JIA), and all are believed to have genetic risk factors. Previously, the researchers used the Immunochip custom microarray to map 186 autoimmune disease-associated loci from 11 autoimmune phenotypes, including adult rheumatoid arthritis (RA). In the current work, the researchers analyzed 340 RF-positive polyarticular JIA cases (292 females) and 14,412 controls (8,002 females) from the United States, United Kingdom, Germany, Canada, and Norway. RF-positive polyarticular disease accounts for about 5% of JIA cases, and its symptoms and presentations resemble adult RA.
The researchers found associations in the human leukocyte antigen (HLA) region. The most significant was found at rs3129769, near HLA-DRB1 (P = 5.51 x 10-31). This single nucleotide polymorphism (SNP) was in strong linkage disequilibrium (LD, r2 = 0.88) with the rs660895 HLA-DRB1 SNP that has been reported in adult RA (P = 2.14 x 10-29).
The researchers examined links between RF-positive polyarticular JIA and the 27 SNPs that had been identified in the previous study of oligoarticular/RF-negative polyarticular JIA. Just 6 of those 27 SNPs were significantly associated with RF-positive polyarticular JIA (P less than .05). On the other hand, of 44 SNPs most strongly associated with RA, 19 were associated with RF-positive polyarticular JIA (P less than .05).
That suggests that RF-positive polyarticular JIA cases are different from other JIA cases. “They’re more like adult patients than they’re like child patients,” said Dr. Stevens.
The researchers also compared the weighted genetic risk scores (wGRS) produced from the top RA loci to wGRS produced from the top oligoarticular/RF-negative polyarticular JIA loci. The wGRS from the top RA loci was a better predictor of RF-positive polyarticular JIA cases (area under the curve [AUC] = 0.71 versus AUC = 0.58; P = 8.26 x 10-33).
The wGRS from RA had similar success in predicting RF-positive polyarticular JIA and early-onset RA cases (AUC = 0.75; P = .25), but it fared worse in predicting late-onset RA (at 70 years or older, AUC = 0.62), compared with the wGRS from RF-positive polyarticular JIA (P = 1.65 x 10-5).
Those results suggest that RF-positive polyarticular JIA more closely resembles younger RA cases than older RA cases.
“If you consider early-onset RA patients, less than 29 years old when they develop RA, they look like JIA patients. But older RA patients, who are over 70 when they develop RA, they look like they totally have a different genetic background,” Dr. Stevens said.
The study could have clinical implications. The lead author, Anne Hinks, PhD, is a research fellow at the University of Manchester (England) and has led the charge to characterize JIA. The wGRS score she developed has the potential to help physicians diagnose classify and treat JIA patients. Currently, they must rely on the International League of Associations for Rheumatology criteria, which can take months to work through and may lead to misclassification diagnoses.
And in any case, the emerging genetic research suggests that the underlying genetics of JIA may be a better way to classify patients. “There’s a lot of overlap and risk of misclassifying patients with the current system. This weighted genetic risk score that Dr. Hinks developed could be used to classify patients with one DNA sample. This is the kind of clinical test we need,” Dr. Stevens said.
The study received funding from a range of government and private sources. Dr. Stevens has a patent licensed to Quest Diagnostics, is conducting research collaborations with Seattle Genetics and Kineta, and has received fellowship support from Pfizer.
SOURCE: Hinks A et al. Arthritis Rheumatol. 2018 Feb 9. doi: 10.1002/art.40443
New evidence suggests the rheumatoid factor–positive polyarticular subtype of juvenile idiopathic arthritis bears a close genetic resemblance to adult rheumatoid arthritis, lending support to the growing suspicion that in arthritis, biological underpinnings are more important than age of onset when it comes to characterizing and, potentially, choosing treatments.
Previous work had shown that rheumatoid factor (RF)–negative patients have genetic risks similar to those of adults with RF-negative disease. “If the RF-negative patients in adult and childhood are similar, then maybe the RF-positive patients are similar in their genetic background as well. That’s what this study was testing,” study coauthor Anne M. Stevens, MD, PhD, division chief of rheumatology at the University of Washington, Seattle, said in an interview. The study was published online Feb. 9 in Arthritis & Rheumatology.
There are seven recognized categories of juvenile idiopathic arthritis (JIA), and all are believed to have genetic risk factors. Previously, the researchers used the Immunochip custom microarray to map 186 autoimmune disease-associated loci from 11 autoimmune phenotypes, including adult rheumatoid arthritis (RA). In the current work, the researchers analyzed 340 RF-positive polyarticular JIA cases (292 females) and 14,412 controls (8,002 females) from the United States, United Kingdom, Germany, Canada, and Norway. RF-positive polyarticular disease accounts for about 5% of JIA cases, and its symptoms and presentations resemble adult RA.
The researchers found associations in the human leukocyte antigen (HLA) region. The most significant was found at rs3129769, near HLA-DRB1 (P = 5.51 x 10-31). This single nucleotide polymorphism (SNP) was in strong linkage disequilibrium (LD, r2 = 0.88) with the rs660895 HLA-DRB1 SNP that has been reported in adult RA (P = 2.14 x 10-29).
The researchers examined links between RF-positive polyarticular JIA and the 27 SNPs that had been identified in the previous study of oligoarticular/RF-negative polyarticular JIA. Just 6 of those 27 SNPs were significantly associated with RF-positive polyarticular JIA (P less than .05). On the other hand, of 44 SNPs most strongly associated with RA, 19 were associated with RF-positive polyarticular JIA (P less than .05).
That suggests that RF-positive polyarticular JIA cases are different from other JIA cases. “They’re more like adult patients than they’re like child patients,” said Dr. Stevens.
The researchers also compared the weighted genetic risk scores (wGRS) produced from the top RA loci to wGRS produced from the top oligoarticular/RF-negative polyarticular JIA loci. The wGRS from the top RA loci was a better predictor of RF-positive polyarticular JIA cases (area under the curve [AUC] = 0.71 versus AUC = 0.58; P = 8.26 x 10-33).
The wGRS from RA had similar success in predicting RF-positive polyarticular JIA and early-onset RA cases (AUC = 0.75; P = .25), but it fared worse in predicting late-onset RA (at 70 years or older, AUC = 0.62), compared with the wGRS from RF-positive polyarticular JIA (P = 1.65 x 10-5).
Those results suggest that RF-positive polyarticular JIA more closely resembles younger RA cases than older RA cases.
“If you consider early-onset RA patients, less than 29 years old when they develop RA, they look like JIA patients. But older RA patients, who are over 70 when they develop RA, they look like they totally have a different genetic background,” Dr. Stevens said.
The study could have clinical implications. The lead author, Anne Hinks, PhD, is a research fellow at the University of Manchester (England) and has led the charge to characterize JIA. The wGRS score she developed has the potential to help physicians diagnose classify and treat JIA patients. Currently, they must rely on the International League of Associations for Rheumatology criteria, which can take months to work through and may lead to misclassification diagnoses.
And in any case, the emerging genetic research suggests that the underlying genetics of JIA may be a better way to classify patients. “There’s a lot of overlap and risk of misclassifying patients with the current system. This weighted genetic risk score that Dr. Hinks developed could be used to classify patients with one DNA sample. This is the kind of clinical test we need,” Dr. Stevens said.
The study received funding from a range of government and private sources. Dr. Stevens has a patent licensed to Quest Diagnostics, is conducting research collaborations with Seattle Genetics and Kineta, and has received fellowship support from Pfizer.
SOURCE: Hinks A et al. Arthritis Rheumatol. 2018 Feb 9. doi: 10.1002/art.40443
New evidence suggests the rheumatoid factor–positive polyarticular subtype of juvenile idiopathic arthritis bears a close genetic resemblance to adult rheumatoid arthritis, lending support to the growing suspicion that in arthritis, biological underpinnings are more important than age of onset when it comes to characterizing and, potentially, choosing treatments.
Previous work had shown that rheumatoid factor (RF)–negative patients have genetic risks similar to those of adults with RF-negative disease. “If the RF-negative patients in adult and childhood are similar, then maybe the RF-positive patients are similar in their genetic background as well. That’s what this study was testing,” study coauthor Anne M. Stevens, MD, PhD, division chief of rheumatology at the University of Washington, Seattle, said in an interview. The study was published online Feb. 9 in Arthritis & Rheumatology.
There are seven recognized categories of juvenile idiopathic arthritis (JIA), and all are believed to have genetic risk factors. Previously, the researchers used the Immunochip custom microarray to map 186 autoimmune disease-associated loci from 11 autoimmune phenotypes, including adult rheumatoid arthritis (RA). In the current work, the researchers analyzed 340 RF-positive polyarticular JIA cases (292 females) and 14,412 controls (8,002 females) from the United States, United Kingdom, Germany, Canada, and Norway. RF-positive polyarticular disease accounts for about 5% of JIA cases, and its symptoms and presentations resemble adult RA.
The researchers found associations in the human leukocyte antigen (HLA) region. The most significant was found at rs3129769, near HLA-DRB1 (P = 5.51 x 10-31). This single nucleotide polymorphism (SNP) was in strong linkage disequilibrium (LD, r2 = 0.88) with the rs660895 HLA-DRB1 SNP that has been reported in adult RA (P = 2.14 x 10-29).
The researchers examined links between RF-positive polyarticular JIA and the 27 SNPs that had been identified in the previous study of oligoarticular/RF-negative polyarticular JIA. Just 6 of those 27 SNPs were significantly associated with RF-positive polyarticular JIA (P less than .05). On the other hand, of 44 SNPs most strongly associated with RA, 19 were associated with RF-positive polyarticular JIA (P less than .05).
That suggests that RF-positive polyarticular JIA cases are different from other JIA cases. “They’re more like adult patients than they’re like child patients,” said Dr. Stevens.
The researchers also compared the weighted genetic risk scores (wGRS) produced from the top RA loci to wGRS produced from the top oligoarticular/RF-negative polyarticular JIA loci. The wGRS from the top RA loci was a better predictor of RF-positive polyarticular JIA cases (area under the curve [AUC] = 0.71 versus AUC = 0.58; P = 8.26 x 10-33).
The wGRS from RA had similar success in predicting RF-positive polyarticular JIA and early-onset RA cases (AUC = 0.75; P = .25), but it fared worse in predicting late-onset RA (at 70 years or older, AUC = 0.62), compared with the wGRS from RF-positive polyarticular JIA (P = 1.65 x 10-5).
Those results suggest that RF-positive polyarticular JIA more closely resembles younger RA cases than older RA cases.
“If you consider early-onset RA patients, less than 29 years old when they develop RA, they look like JIA patients. But older RA patients, who are over 70 when they develop RA, they look like they totally have a different genetic background,” Dr. Stevens said.
The study could have clinical implications. The lead author, Anne Hinks, PhD, is a research fellow at the University of Manchester (England) and has led the charge to characterize JIA. The wGRS score she developed has the potential to help physicians diagnose classify and treat JIA patients. Currently, they must rely on the International League of Associations for Rheumatology criteria, which can take months to work through and may lead to misclassification diagnoses.
And in any case, the emerging genetic research suggests that the underlying genetics of JIA may be a better way to classify patients. “There’s a lot of overlap and risk of misclassifying patients with the current system. This weighted genetic risk score that Dr. Hinks developed could be used to classify patients with one DNA sample. This is the kind of clinical test we need,” Dr. Stevens said.
The study received funding from a range of government and private sources. Dr. Stevens has a patent licensed to Quest Diagnostics, is conducting research collaborations with Seattle Genetics and Kineta, and has received fellowship support from Pfizer.
SOURCE: Hinks A et al. Arthritis Rheumatol. 2018 Feb 9. doi: 10.1002/art.40443
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Genetics underlying arthritis may be more important than age of onset.
Major finding: The weighted genetic risk scores produced from the top RA loci was a better predictor of RF-positive polyarticular JIA than that generated from the top oligoarticular/RF-negative polyarticular JIA loci.
Data source: Case-control analysis of 340 cases and 14,412 controls.
Disclosures: The study received funding from a range of government and private sources. Dr. Stevens has a patent licensed to Quest Diagnostics, is conducting research collaborations with Seattle Genetics and Kineta, and has received fellowship support from Pfizer.
Source: Hinks A et al. Arthritis Rheumatol. 2018 Feb 9. doi: 10.1002/art.40443.
Experts review the year in rheumatology ... and what lies ahead
MAUI, HAWAII – Arthur Kavanaugh, MD, program director for the Rheumatology Winter Clinical Symposium, likes to close out the meeting each year in high style by assembling selected conference faculty to offer their personal picks for the top developments in the field during the past year and make predictions about the year to come.
Here’s how they called it:
The top events in rheumatology during the last year
The rise of oral small molecules: The Janus kinase (JAK) inhibitors and other oral small molecules that have begun reaching the marketplace, with many more in development, will bring a paradigm shift in the treatment not only of rheumatic diseases, but in inflammatory bowel disease and skin diseases as well, predicted Alvin F. Wells, MD, PhD, a rheumatologist at Duke University in Durham, N.C., who is also director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
“The challenge is whether Medicare will cover the pills the way they cover the infusions and the other things we do,” according to Dr. Wells.
A bevy of new drugs for psoriatic arthritis and psoriasis: “I think the most important advance in the past year was the approval of a profusion of drugs for psoriatic arthritis and psoriasis. It’s really opened up the landscape for us in terms of treatment options. The downside is it’s going to take us a while to sort through which drugs fit where,” noted Eric J. Ruderman, MD, professor of medicine and associate chief of clinical affairs in the division of rheumatology at Northwestern University in Chicago.
“The drug I was most impressed with was tofacitinib [Xeljanz, an oral JAK inhibitor], not just by its effectiveness but by its potential to change the game, and particularly the data in tumor necrosis factor inhibitor inadequate responders. That was pretty solid data. It really opens the way to oral small molecules for joint diseases,” he added.
Interleukin-18 binding protein for monogenic inflammasome diseases: The biggest recent breakthrough in pediatric rheumatology was the Food and Drug Administration’s April 2017 designation of Breakthrough Therapy status for the recombinant human IL-18 binding protein known as tadekinig alfa for monogenic IL-18-associated autoinflammatory conditions, as well as the biologic’s Orphan Drug Designation for treatment of hemophagocytic lymphohistiocytosis, according to Anne M. Stevens, MD, PhD, professor of pediatrics at the University of Washington, Seattle, and chief of pediatric rheumatology at Seattle Children’s Hospital.
Novel treatment concept emerges in severe SLE: The study that knocked the socks off of Martin J. Bergman, MD, in 2017 was the Dutch SymBiose study, presented at both the European League Against Rheumatism and American College of Rheumatology annual meetings. It included just 14 patients with severe refractory SLE – including 10 with lupus nephritis – and tested a treatment strategy of rituximab (Rituxan) followed a few weeks later by a course of belimumab (Benlysta).
“The results were very dramatic, to say the least,” said Dr. Bergman of Drexel University in Philadelphia. Indeed, this one-two therapeutic punch resulted in sharply reduced levels of pathogenic autoantibodies and immune complex-mediated neutrophil extracellular traps while also knocking down very high baseline Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) scores to near zero, even while enabling patients to discontinue systemic corticosteroids and mycophenolate mofetil (CellCept). Several much larger clinical trials of this regimen and other similar ones are ongoing in an effort to duplicate the results.
Dr. Kavanaugh said the SymBiose study was one of his own top picks for study of the year as well.
Mainstream use of dupilumab (Dupixent) for moderate to severe atopic dermatitis: “This is a total game changer. It’s really changed a lot of people’s lives,” commented George M. Martin, MD, a dermatologist in private practice on Maui.
“Interestingly, historically drugs that started out in your realm later made their way to dermatology, but now we’re seeing the IL-23 inhibitors starting with us and then making their way into rheumatology and gastroenterology. The IL-23 inhibitors are very powerful drugs; when we’re seeing half of our psoriasis patients achieve PASI 100 responses, it’s very exciting. And these are durable responses,” he noted.
The opioid crisis: What’s the most important recent event in rheumatology?
“That’s easy: The biggest thing in all of medicine is the opioid crisis. Whether you recognize it or not, it’s gigantic. It’s $500 billion of the U.S. economy, every year. Forty percent of rheumatoid arthritis patients and 30% with ankylosing spondylitis are on opioids, and what goes along with that is a lot of ugly stuff,” said John J. Cush, MD, professor of medicine and rheumatology at Baylor University in Dallas.
Moreover, the FDA is now so leery of opioids that the agency has set the bar unrealistically high for approval of newer agents offering reduced abuse potential.
“I’ve been involved with or watched at least six FDA advisory panels looking at new, lower abuse-potential opioids in the last couple years. Only one agent got through,” according to Dr. Cush.
Dr. Troum commented, “I really think this whole opioid epidemic started with the campaign to make pain the fifth vital sign back in the 1990s. Some of the pharmaceutical companies took that concept and really ran with it.”
Rapamycin for inclusion body myositis: Dr. Kavanaugh’s pick for study of the year was what he described as “a brilliant presentation” of a French multicenter, placebo-controlled clinical trial of rapamycin for patients with inclusion body myositis at the 2017 ACR annual meeting.
“The French group considers IBM [inclusion body myositis] to be essentially Alzheimer’s disease of the muscle, marked by amyloid deposition. They chose to study rapamycin, which not only has immunosuppressive properties because it binds to mTOR [the mammalian target of rapamycin], but it also has the ability to inhibit amyloid protein deposition,” he explained.
The investigators reported improved 6-minute-walk distance and pulmonary function in the rapamycin group, whereas placebo-treated controls rapidly deteriorated.
“This is an approved drug for other indications, and we scratch our heads with IBM. It’s super nice to have something like that,” Dr. Kavanaugh observed.
A look at what’s in store
More tele-rheumatology: “I think the biggest thing is going to be more tele-rheumatology, more tele-ultrasound. Kaiser Permanente said 49% of their visits last year were virtual visits; that number is just going to grow,” predicted Dr. Wells.
Especially in medically underserved areas of the country, including large rural expanses, demand for remote tele-rheumatologic consults with high-quality imaging is going to increase, he added.
Here come cannabinoids for pain control: Dr. Troum predicted that in the depths of the national opioid epidemic, in a climate that discourages legitimate prescribing of traditional pain medications, rheumatologists can anticipate growing patient demand for cannabidiol and other cannabinoids for pain relief.
“I have patients coming in their 60s, 70s, and 80s – these are not young people – who are whispering to me, ‘Can I use this for my chronic pain?’ I think there’s going to be a big push for ways other than opioids to treat our patients’ pain,” according to Dr. Troum.
Tipping point nears for JAK inhibitors: In 2018, it will become clear just how seriously the Food and Drug Administration views the signal of possible increased venous thromboembolic risk associated with the oral JAK inhibitors for rheumatoid arthritis. The agency is expected to rule on Eli Lilly and Incyte’s resubmitted application for marketing approval for the JAK inhibitor baricitinib, which was tripped up earlier based in part upon VTE concerns.
“I think the big story in 2018 will be how the JAK story shakes out – whether this VTE thing has legs,” Dr. Ruderman predicted. “A sea change could be coming in our field, and it’s not coming next year or the year after, but 10 years from now: Are we going to move past the era of methotrexate and use generic small molecules instead? We’re going to find out within the next year whether that’s going to happen.”
Phase 3 results coming on tocilizumab for systemic sclerosis: “I think we’re going to see some really exciting systemic sclerosis data coming out this year,” Dr. Stevens said. Based upon the positive phase 2 results presented for tocilizumab (Actemra) last year, she’s optimistic that the ongoing phase 3 randomized trial will demonstrate a significant advantage over placebo in lung function. Also, ongoing separate clinical trials are evaluating an antifibrotic drug and rapamycin for systemic sclerosis.
Dr. Bergman, too, has high hopes for these studies: “I think we may finally be getting to a place where we can see effective drugs in systemic sclerosis.”
Amazon, Berkshire Hathaway, and JPMorgan Chase form a nonprofit to improve employee health care: In a recent press conference, the three CEOs weren’t specific about their plans, but Dr. Martin predicted the companies are likely to self-insure, bypassing Cigna and the other major health insurance companies and then contracting with physicians. He forecast that “probably within the next 5 years, what they do is going to affect everybody in this room.”
Rheumatologists will need to master a new mindset: Many rheumatologists have gotten comfortable with an all-tumor necrosis factor inhibitor treatment menu for their patients with moderate or severe rheumatoid arthritis. That’s got to change, according to Dr. Cush.
“We now have two IL-6 inhibitors, two IL-17 inhibitors, and we’ll soon have two JAK inhibitors. That’s going to be a direct threat to the not right- or left-brain, but the TNF-brain rheumatologist who now writes prescriptions for three TNF inhibitors in a row before questioning the efficacy. The idea is you will now be using drugs with other mechanisms of action first-line, or at the very least, second-line, and that’s going to be a paradigm shift for a lot of people,” he explained.
None of the speakers reported having financial conflicts regarding their comments.
MAUI, HAWAII – Arthur Kavanaugh, MD, program director for the Rheumatology Winter Clinical Symposium, likes to close out the meeting each year in high style by assembling selected conference faculty to offer their personal picks for the top developments in the field during the past year and make predictions about the year to come.
Here’s how they called it:
The top events in rheumatology during the last year
The rise of oral small molecules: The Janus kinase (JAK) inhibitors and other oral small molecules that have begun reaching the marketplace, with many more in development, will bring a paradigm shift in the treatment not only of rheumatic diseases, but in inflammatory bowel disease and skin diseases as well, predicted Alvin F. Wells, MD, PhD, a rheumatologist at Duke University in Durham, N.C., who is also director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
“The challenge is whether Medicare will cover the pills the way they cover the infusions and the other things we do,” according to Dr. Wells.
A bevy of new drugs for psoriatic arthritis and psoriasis: “I think the most important advance in the past year was the approval of a profusion of drugs for psoriatic arthritis and psoriasis. It’s really opened up the landscape for us in terms of treatment options. The downside is it’s going to take us a while to sort through which drugs fit where,” noted Eric J. Ruderman, MD, professor of medicine and associate chief of clinical affairs in the division of rheumatology at Northwestern University in Chicago.
“The drug I was most impressed with was tofacitinib [Xeljanz, an oral JAK inhibitor], not just by its effectiveness but by its potential to change the game, and particularly the data in tumor necrosis factor inhibitor inadequate responders. That was pretty solid data. It really opens the way to oral small molecules for joint diseases,” he added.
Interleukin-18 binding protein for monogenic inflammasome diseases: The biggest recent breakthrough in pediatric rheumatology was the Food and Drug Administration’s April 2017 designation of Breakthrough Therapy status for the recombinant human IL-18 binding protein known as tadekinig alfa for monogenic IL-18-associated autoinflammatory conditions, as well as the biologic’s Orphan Drug Designation for treatment of hemophagocytic lymphohistiocytosis, according to Anne M. Stevens, MD, PhD, professor of pediatrics at the University of Washington, Seattle, and chief of pediatric rheumatology at Seattle Children’s Hospital.
Novel treatment concept emerges in severe SLE: The study that knocked the socks off of Martin J. Bergman, MD, in 2017 was the Dutch SymBiose study, presented at both the European League Against Rheumatism and American College of Rheumatology annual meetings. It included just 14 patients with severe refractory SLE – including 10 with lupus nephritis – and tested a treatment strategy of rituximab (Rituxan) followed a few weeks later by a course of belimumab (Benlysta).
“The results were very dramatic, to say the least,” said Dr. Bergman of Drexel University in Philadelphia. Indeed, this one-two therapeutic punch resulted in sharply reduced levels of pathogenic autoantibodies and immune complex-mediated neutrophil extracellular traps while also knocking down very high baseline Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) scores to near zero, even while enabling patients to discontinue systemic corticosteroids and mycophenolate mofetil (CellCept). Several much larger clinical trials of this regimen and other similar ones are ongoing in an effort to duplicate the results.
Dr. Kavanaugh said the SymBiose study was one of his own top picks for study of the year as well.
Mainstream use of dupilumab (Dupixent) for moderate to severe atopic dermatitis: “This is a total game changer. It’s really changed a lot of people’s lives,” commented George M. Martin, MD, a dermatologist in private practice on Maui.
“Interestingly, historically drugs that started out in your realm later made their way to dermatology, but now we’re seeing the IL-23 inhibitors starting with us and then making their way into rheumatology and gastroenterology. The IL-23 inhibitors are very powerful drugs; when we’re seeing half of our psoriasis patients achieve PASI 100 responses, it’s very exciting. And these are durable responses,” he noted.
The opioid crisis: What’s the most important recent event in rheumatology?
“That’s easy: The biggest thing in all of medicine is the opioid crisis. Whether you recognize it or not, it’s gigantic. It’s $500 billion of the U.S. economy, every year. Forty percent of rheumatoid arthritis patients and 30% with ankylosing spondylitis are on opioids, and what goes along with that is a lot of ugly stuff,” said John J. Cush, MD, professor of medicine and rheumatology at Baylor University in Dallas.
Moreover, the FDA is now so leery of opioids that the agency has set the bar unrealistically high for approval of newer agents offering reduced abuse potential.
“I’ve been involved with or watched at least six FDA advisory panels looking at new, lower abuse-potential opioids in the last couple years. Only one agent got through,” according to Dr. Cush.
Dr. Troum commented, “I really think this whole opioid epidemic started with the campaign to make pain the fifth vital sign back in the 1990s. Some of the pharmaceutical companies took that concept and really ran with it.”
Rapamycin for inclusion body myositis: Dr. Kavanaugh’s pick for study of the year was what he described as “a brilliant presentation” of a French multicenter, placebo-controlled clinical trial of rapamycin for patients with inclusion body myositis at the 2017 ACR annual meeting.
“The French group considers IBM [inclusion body myositis] to be essentially Alzheimer’s disease of the muscle, marked by amyloid deposition. They chose to study rapamycin, which not only has immunosuppressive properties because it binds to mTOR [the mammalian target of rapamycin], but it also has the ability to inhibit amyloid protein deposition,” he explained.
The investigators reported improved 6-minute-walk distance and pulmonary function in the rapamycin group, whereas placebo-treated controls rapidly deteriorated.
“This is an approved drug for other indications, and we scratch our heads with IBM. It’s super nice to have something like that,” Dr. Kavanaugh observed.
A look at what’s in store
More tele-rheumatology: “I think the biggest thing is going to be more tele-rheumatology, more tele-ultrasound. Kaiser Permanente said 49% of their visits last year were virtual visits; that number is just going to grow,” predicted Dr. Wells.
Especially in medically underserved areas of the country, including large rural expanses, demand for remote tele-rheumatologic consults with high-quality imaging is going to increase, he added.
Here come cannabinoids for pain control: Dr. Troum predicted that in the depths of the national opioid epidemic, in a climate that discourages legitimate prescribing of traditional pain medications, rheumatologists can anticipate growing patient demand for cannabidiol and other cannabinoids for pain relief.
“I have patients coming in their 60s, 70s, and 80s – these are not young people – who are whispering to me, ‘Can I use this for my chronic pain?’ I think there’s going to be a big push for ways other than opioids to treat our patients’ pain,” according to Dr. Troum.
Tipping point nears for JAK inhibitors: In 2018, it will become clear just how seriously the Food and Drug Administration views the signal of possible increased venous thromboembolic risk associated with the oral JAK inhibitors for rheumatoid arthritis. The agency is expected to rule on Eli Lilly and Incyte’s resubmitted application for marketing approval for the JAK inhibitor baricitinib, which was tripped up earlier based in part upon VTE concerns.
“I think the big story in 2018 will be how the JAK story shakes out – whether this VTE thing has legs,” Dr. Ruderman predicted. “A sea change could be coming in our field, and it’s not coming next year or the year after, but 10 years from now: Are we going to move past the era of methotrexate and use generic small molecules instead? We’re going to find out within the next year whether that’s going to happen.”
Phase 3 results coming on tocilizumab for systemic sclerosis: “I think we’re going to see some really exciting systemic sclerosis data coming out this year,” Dr. Stevens said. Based upon the positive phase 2 results presented for tocilizumab (Actemra) last year, she’s optimistic that the ongoing phase 3 randomized trial will demonstrate a significant advantage over placebo in lung function. Also, ongoing separate clinical trials are evaluating an antifibrotic drug and rapamycin for systemic sclerosis.
Dr. Bergman, too, has high hopes for these studies: “I think we may finally be getting to a place where we can see effective drugs in systemic sclerosis.”
Amazon, Berkshire Hathaway, and JPMorgan Chase form a nonprofit to improve employee health care: In a recent press conference, the three CEOs weren’t specific about their plans, but Dr. Martin predicted the companies are likely to self-insure, bypassing Cigna and the other major health insurance companies and then contracting with physicians. He forecast that “probably within the next 5 years, what they do is going to affect everybody in this room.”
Rheumatologists will need to master a new mindset: Many rheumatologists have gotten comfortable with an all-tumor necrosis factor inhibitor treatment menu for their patients with moderate or severe rheumatoid arthritis. That’s got to change, according to Dr. Cush.
“We now have two IL-6 inhibitors, two IL-17 inhibitors, and we’ll soon have two JAK inhibitors. That’s going to be a direct threat to the not right- or left-brain, but the TNF-brain rheumatologist who now writes prescriptions for three TNF inhibitors in a row before questioning the efficacy. The idea is you will now be using drugs with other mechanisms of action first-line, or at the very least, second-line, and that’s going to be a paradigm shift for a lot of people,” he explained.
None of the speakers reported having financial conflicts regarding their comments.
MAUI, HAWAII – Arthur Kavanaugh, MD, program director for the Rheumatology Winter Clinical Symposium, likes to close out the meeting each year in high style by assembling selected conference faculty to offer their personal picks for the top developments in the field during the past year and make predictions about the year to come.
Here’s how they called it:
The top events in rheumatology during the last year
The rise of oral small molecules: The Janus kinase (JAK) inhibitors and other oral small molecules that have begun reaching the marketplace, with many more in development, will bring a paradigm shift in the treatment not only of rheumatic diseases, but in inflammatory bowel disease and skin diseases as well, predicted Alvin F. Wells, MD, PhD, a rheumatologist at Duke University in Durham, N.C., who is also director of the Rheumatology and Immunotherapy Center in Franklin, Wisc.
“The challenge is whether Medicare will cover the pills the way they cover the infusions and the other things we do,” according to Dr. Wells.
A bevy of new drugs for psoriatic arthritis and psoriasis: “I think the most important advance in the past year was the approval of a profusion of drugs for psoriatic arthritis and psoriasis. It’s really opened up the landscape for us in terms of treatment options. The downside is it’s going to take us a while to sort through which drugs fit where,” noted Eric J. Ruderman, MD, professor of medicine and associate chief of clinical affairs in the division of rheumatology at Northwestern University in Chicago.
“The drug I was most impressed with was tofacitinib [Xeljanz, an oral JAK inhibitor], not just by its effectiveness but by its potential to change the game, and particularly the data in tumor necrosis factor inhibitor inadequate responders. That was pretty solid data. It really opens the way to oral small molecules for joint diseases,” he added.
Interleukin-18 binding protein for monogenic inflammasome diseases: The biggest recent breakthrough in pediatric rheumatology was the Food and Drug Administration’s April 2017 designation of Breakthrough Therapy status for the recombinant human IL-18 binding protein known as tadekinig alfa for monogenic IL-18-associated autoinflammatory conditions, as well as the biologic’s Orphan Drug Designation for treatment of hemophagocytic lymphohistiocytosis, according to Anne M. Stevens, MD, PhD, professor of pediatrics at the University of Washington, Seattle, and chief of pediatric rheumatology at Seattle Children’s Hospital.
Novel treatment concept emerges in severe SLE: The study that knocked the socks off of Martin J. Bergman, MD, in 2017 was the Dutch SymBiose study, presented at both the European League Against Rheumatism and American College of Rheumatology annual meetings. It included just 14 patients with severe refractory SLE – including 10 with lupus nephritis – and tested a treatment strategy of rituximab (Rituxan) followed a few weeks later by a course of belimumab (Benlysta).
“The results were very dramatic, to say the least,” said Dr. Bergman of Drexel University in Philadelphia. Indeed, this one-two therapeutic punch resulted in sharply reduced levels of pathogenic autoantibodies and immune complex-mediated neutrophil extracellular traps while also knocking down very high baseline Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) scores to near zero, even while enabling patients to discontinue systemic corticosteroids and mycophenolate mofetil (CellCept). Several much larger clinical trials of this regimen and other similar ones are ongoing in an effort to duplicate the results.
Dr. Kavanaugh said the SymBiose study was one of his own top picks for study of the year as well.
Mainstream use of dupilumab (Dupixent) for moderate to severe atopic dermatitis: “This is a total game changer. It’s really changed a lot of people’s lives,” commented George M. Martin, MD, a dermatologist in private practice on Maui.
“Interestingly, historically drugs that started out in your realm later made their way to dermatology, but now we’re seeing the IL-23 inhibitors starting with us and then making their way into rheumatology and gastroenterology. The IL-23 inhibitors are very powerful drugs; when we’re seeing half of our psoriasis patients achieve PASI 100 responses, it’s very exciting. And these are durable responses,” he noted.
The opioid crisis: What’s the most important recent event in rheumatology?
“That’s easy: The biggest thing in all of medicine is the opioid crisis. Whether you recognize it or not, it’s gigantic. It’s $500 billion of the U.S. economy, every year. Forty percent of rheumatoid arthritis patients and 30% with ankylosing spondylitis are on opioids, and what goes along with that is a lot of ugly stuff,” said John J. Cush, MD, professor of medicine and rheumatology at Baylor University in Dallas.
Moreover, the FDA is now so leery of opioids that the agency has set the bar unrealistically high for approval of newer agents offering reduced abuse potential.
“I’ve been involved with or watched at least six FDA advisory panels looking at new, lower abuse-potential opioids in the last couple years. Only one agent got through,” according to Dr. Cush.
Dr. Troum commented, “I really think this whole opioid epidemic started with the campaign to make pain the fifth vital sign back in the 1990s. Some of the pharmaceutical companies took that concept and really ran with it.”
Rapamycin for inclusion body myositis: Dr. Kavanaugh’s pick for study of the year was what he described as “a brilliant presentation” of a French multicenter, placebo-controlled clinical trial of rapamycin for patients with inclusion body myositis at the 2017 ACR annual meeting.
“The French group considers IBM [inclusion body myositis] to be essentially Alzheimer’s disease of the muscle, marked by amyloid deposition. They chose to study rapamycin, which not only has immunosuppressive properties because it binds to mTOR [the mammalian target of rapamycin], but it also has the ability to inhibit amyloid protein deposition,” he explained.
The investigators reported improved 6-minute-walk distance and pulmonary function in the rapamycin group, whereas placebo-treated controls rapidly deteriorated.
“This is an approved drug for other indications, and we scratch our heads with IBM. It’s super nice to have something like that,” Dr. Kavanaugh observed.
A look at what’s in store
More tele-rheumatology: “I think the biggest thing is going to be more tele-rheumatology, more tele-ultrasound. Kaiser Permanente said 49% of their visits last year were virtual visits; that number is just going to grow,” predicted Dr. Wells.
Especially in medically underserved areas of the country, including large rural expanses, demand for remote tele-rheumatologic consults with high-quality imaging is going to increase, he added.
Here come cannabinoids for pain control: Dr. Troum predicted that in the depths of the national opioid epidemic, in a climate that discourages legitimate prescribing of traditional pain medications, rheumatologists can anticipate growing patient demand for cannabidiol and other cannabinoids for pain relief.
“I have patients coming in their 60s, 70s, and 80s – these are not young people – who are whispering to me, ‘Can I use this for my chronic pain?’ I think there’s going to be a big push for ways other than opioids to treat our patients’ pain,” according to Dr. Troum.
Tipping point nears for JAK inhibitors: In 2018, it will become clear just how seriously the Food and Drug Administration views the signal of possible increased venous thromboembolic risk associated with the oral JAK inhibitors for rheumatoid arthritis. The agency is expected to rule on Eli Lilly and Incyte’s resubmitted application for marketing approval for the JAK inhibitor baricitinib, which was tripped up earlier based in part upon VTE concerns.
“I think the big story in 2018 will be how the JAK story shakes out – whether this VTE thing has legs,” Dr. Ruderman predicted. “A sea change could be coming in our field, and it’s not coming next year or the year after, but 10 years from now: Are we going to move past the era of methotrexate and use generic small molecules instead? We’re going to find out within the next year whether that’s going to happen.”
Phase 3 results coming on tocilizumab for systemic sclerosis: “I think we’re going to see some really exciting systemic sclerosis data coming out this year,” Dr. Stevens said. Based upon the positive phase 2 results presented for tocilizumab (Actemra) last year, she’s optimistic that the ongoing phase 3 randomized trial will demonstrate a significant advantage over placebo in lung function. Also, ongoing separate clinical trials are evaluating an antifibrotic drug and rapamycin for systemic sclerosis.
Dr. Bergman, too, has high hopes for these studies: “I think we may finally be getting to a place where we can see effective drugs in systemic sclerosis.”
Amazon, Berkshire Hathaway, and JPMorgan Chase form a nonprofit to improve employee health care: In a recent press conference, the three CEOs weren’t specific about their plans, but Dr. Martin predicted the companies are likely to self-insure, bypassing Cigna and the other major health insurance companies and then contracting with physicians. He forecast that “probably within the next 5 years, what they do is going to affect everybody in this room.”
Rheumatologists will need to master a new mindset: Many rheumatologists have gotten comfortable with an all-tumor necrosis factor inhibitor treatment menu for their patients with moderate or severe rheumatoid arthritis. That’s got to change, according to Dr. Cush.
“We now have two IL-6 inhibitors, two IL-17 inhibitors, and we’ll soon have two JAK inhibitors. That’s going to be a direct threat to the not right- or left-brain, but the TNF-brain rheumatologist who now writes prescriptions for three TNF inhibitors in a row before questioning the efficacy. The idea is you will now be using drugs with other mechanisms of action first-line, or at the very least, second-line, and that’s going to be a paradigm shift for a lot of people,” he explained.
None of the speakers reported having financial conflicts regarding their comments.
EXPERT ANALYSIS FROM RWCS 2018
JAK inhibitors for RA: Is VTE risk overblown?
MAUI, HAWAII – Rheumatologists, regulatory agencies, and the pharmaceutical industry all have gone off the deep end in their fretting over what appears to be a low rate of venous thromboembolic events in the major randomized trials of the oral Janus kinase inhibitors for RA, Mark C. Genovese, MD, asserted at the 2018 Rheumatology Winter Clinical Symposium.
“The reality is all of our drugs pose potential risks. Unfortunately, I think that at least for the moment, the field has turned all attention in one direction: VTE [venous thromboembolic] events. I suspect there’s some truth [to the possible associated risk]. Certainly we are seeing these events. The question is, how overdone is this?” according to Dr. Genovese, professor of medicine and cochief of the division of immunology and rheumatology at Stanford (Calif.) University.
“I think the upadacitinib data has been entirely overshadowed by concerns about VTEs,” he said. “In the last year, we saw three significant phase 3 studies on upadacitinib arrive in the rheumatology community, and I think the only thing we talked about was VTEs.”
All parties interested in developing Janus kinase (JAK) inhibitors for the treatment of RA began to freak out about a possible increase in VTEs when in April 2017 the Food and Drug Administration turned down Eli Lilly and Incyte’s initial application for marketing approval of the JAK inhibitor baricitinib. Among the problems the agency cited was evidence of potential thrombotic risk.
The VTE rate in baricitinib clinical trials up to 48 weeks in duration was 0.53 events/100 patient-years, with no significant difference in risk between the2-mg and 4-mg doses. This appears to be a class effect for the oral JAK inhibitors, as low rates of VTE, albeit numerically higher than in placebo-treated controls, have also been recorded in the RA development programs for tofacitinib (Xeljanz) as well as the investigational agents filgotinib and upadacitinib, the rheumatologist noted.
This begs the question of whether these VTE rates are significantly higher than background rates in patients with RA or other rheumatologic diseases, which are known to be elevated relative to the general population. Indeed, a retrospective study of insurance claims data by investigators at Brigham and Women’s Hospital, Boston, concluded that the VTE rate in RA patients was 0.61 events/100 patient-years, 120% greater than in a matched patient population without RA. After fully adjusting for comorbid conditions and demographics, the relative risk increase associated with RA dropped to 40%, still significantly higher than in controls (Arthritis Care Res [Hoboken]. 2013 Oct;65[10]:1600-7).
Similarly, Canadian investigators conducted a meta-analysis of 25 studies with VTE data in patients with RA, systemic lupus erythematosus, Sjögren’s syndrome, systemic sclerosis, or inflammatory myositis. This meta-analysis included 10 studies of more than 5.2 million RA patients and nearly 900,000 controls. The conclusion: each of these rheumatic diseases was associated with a VTE rate more than three times higher than in the general population (Arthritis Res Ther. 2014 Sep 25;16[5]:435).
“Patients with RA are at higher risk for VTE than those without RA. It’s unfortunate, and it’s certainly something I don’t think many of us have thought much about before. It’s something we don’t often get to see and something we don’t like to think about,” the rheumatologist observed.
Dr. Genovese admitted to a degree of personal frustration with the current tunnel vision focus on VTEs in JAK inhibitor trials. At the 2017 annual meeting of the American College of Rheumatology he presented the results of the phase 3 SELECT-BEYOND study in which 499 RA patients who had previously failed to respond or were intolerant to biologic therapy were randomized to once daily upadacitinib at 15 or 30 mg or placebo on top of background methotrexate. At week 12, the ACR 20 response rate was 65% for upadacitinib at 15 mg, 56% at 30 mg, and 28% in placebo-treated controls.
“That’s almost a 40% placebo-adjusted response rate. In fact, it’s the highest response I’ve ever seen in a biologic inadequate-responder population. This really looked pretty good, but I don’t think anyone ever took notice. Why not? Because we were all worried about VTE,” he said.
There were in fact a handful of VTEs in upadacitinib-treated patients, Dr. Genovese was quick to note. But he was more impressed by the week 12 ACR 20 responses in patients who had previously failed on three or more biologics: 71% with upadacitinib at 15 mg and 50% at 30 mg, compared with 23% in controls. Moreover, among patients with a baseline history of failure to respond to anti–interleukin-6 therapy, the week 12 ACR 20 rate was 56% with upadacitinib at 15 mg and 58% at 30 mg, versus 20% in controls.
“This looks like a pretty effective drug for patients who’ve failed everything else in our practice,” he commented.
Dr. Genovese reminded his audience that the rheumatology community has a history of overreacting to safety signals in the early days after introduction of new therapies. Examples: tuberculosis with tumor necrosis factor inhibitors, lymphoma with abatacept (Orencia), lymphoma with anti–tumor necrosis factor agents, and cardiovascular events with anti–interleukin-6 inhibition.
“PML [progressive multifocal leukoencephalopathy] is a breathtaking side effect with rituximab [Rituxan], but we’ve gotten over that. We recognize that it’s a potential problem, but we still prescribe rituximab,” the rheumatologist noted. “We’re probably going to need to address the issue of which of our patients are potentially at higher risk for VTE, and maybe we avoid this class in those patients. Like we now do as we look at patients we think are at increased risk for infection, or multiple sclerosis, or TB, we may also need to think of VTE risk.”
But , he argued. There is a pressing unmet need for new therapies for RA with novel mechanisms of action. Only about one-half of patients on contemporary biologic therapies are still on that agent 5 years after initiating therapy.
“Virtually all our patients are partial responders. Everybody gets some benefit. But true remission is achieved by only a minority,” Dr. Genovese said. “The gap between where we are and where we want to be is actually much greater than we often perceive.”
He reported having financial relationships with AbbVie, which is developing upadacitinib, and more than a dozen other medical companies.
MAUI, HAWAII – Rheumatologists, regulatory agencies, and the pharmaceutical industry all have gone off the deep end in their fretting over what appears to be a low rate of venous thromboembolic events in the major randomized trials of the oral Janus kinase inhibitors for RA, Mark C. Genovese, MD, asserted at the 2018 Rheumatology Winter Clinical Symposium.
“The reality is all of our drugs pose potential risks. Unfortunately, I think that at least for the moment, the field has turned all attention in one direction: VTE [venous thromboembolic] events. I suspect there’s some truth [to the possible associated risk]. Certainly we are seeing these events. The question is, how overdone is this?” according to Dr. Genovese, professor of medicine and cochief of the division of immunology and rheumatology at Stanford (Calif.) University.
“I think the upadacitinib data has been entirely overshadowed by concerns about VTEs,” he said. “In the last year, we saw three significant phase 3 studies on upadacitinib arrive in the rheumatology community, and I think the only thing we talked about was VTEs.”
All parties interested in developing Janus kinase (JAK) inhibitors for the treatment of RA began to freak out about a possible increase in VTEs when in April 2017 the Food and Drug Administration turned down Eli Lilly and Incyte’s initial application for marketing approval of the JAK inhibitor baricitinib. Among the problems the agency cited was evidence of potential thrombotic risk.
The VTE rate in baricitinib clinical trials up to 48 weeks in duration was 0.53 events/100 patient-years, with no significant difference in risk between the2-mg and 4-mg doses. This appears to be a class effect for the oral JAK inhibitors, as low rates of VTE, albeit numerically higher than in placebo-treated controls, have also been recorded in the RA development programs for tofacitinib (Xeljanz) as well as the investigational agents filgotinib and upadacitinib, the rheumatologist noted.
This begs the question of whether these VTE rates are significantly higher than background rates in patients with RA or other rheumatologic diseases, which are known to be elevated relative to the general population. Indeed, a retrospective study of insurance claims data by investigators at Brigham and Women’s Hospital, Boston, concluded that the VTE rate in RA patients was 0.61 events/100 patient-years, 120% greater than in a matched patient population without RA. After fully adjusting for comorbid conditions and demographics, the relative risk increase associated with RA dropped to 40%, still significantly higher than in controls (Arthritis Care Res [Hoboken]. 2013 Oct;65[10]:1600-7).
Similarly, Canadian investigators conducted a meta-analysis of 25 studies with VTE data in patients with RA, systemic lupus erythematosus, Sjögren’s syndrome, systemic sclerosis, or inflammatory myositis. This meta-analysis included 10 studies of more than 5.2 million RA patients and nearly 900,000 controls. The conclusion: each of these rheumatic diseases was associated with a VTE rate more than three times higher than in the general population (Arthritis Res Ther. 2014 Sep 25;16[5]:435).
“Patients with RA are at higher risk for VTE than those without RA. It’s unfortunate, and it’s certainly something I don’t think many of us have thought much about before. It’s something we don’t often get to see and something we don’t like to think about,” the rheumatologist observed.
Dr. Genovese admitted to a degree of personal frustration with the current tunnel vision focus on VTEs in JAK inhibitor trials. At the 2017 annual meeting of the American College of Rheumatology he presented the results of the phase 3 SELECT-BEYOND study in which 499 RA patients who had previously failed to respond or were intolerant to biologic therapy were randomized to once daily upadacitinib at 15 or 30 mg or placebo on top of background methotrexate. At week 12, the ACR 20 response rate was 65% for upadacitinib at 15 mg, 56% at 30 mg, and 28% in placebo-treated controls.
“That’s almost a 40% placebo-adjusted response rate. In fact, it’s the highest response I’ve ever seen in a biologic inadequate-responder population. This really looked pretty good, but I don’t think anyone ever took notice. Why not? Because we were all worried about VTE,” he said.
There were in fact a handful of VTEs in upadacitinib-treated patients, Dr. Genovese was quick to note. But he was more impressed by the week 12 ACR 20 responses in patients who had previously failed on three or more biologics: 71% with upadacitinib at 15 mg and 50% at 30 mg, compared with 23% in controls. Moreover, among patients with a baseline history of failure to respond to anti–interleukin-6 therapy, the week 12 ACR 20 rate was 56% with upadacitinib at 15 mg and 58% at 30 mg, versus 20% in controls.
“This looks like a pretty effective drug for patients who’ve failed everything else in our practice,” he commented.
Dr. Genovese reminded his audience that the rheumatology community has a history of overreacting to safety signals in the early days after introduction of new therapies. Examples: tuberculosis with tumor necrosis factor inhibitors, lymphoma with abatacept (Orencia), lymphoma with anti–tumor necrosis factor agents, and cardiovascular events with anti–interleukin-6 inhibition.
“PML [progressive multifocal leukoencephalopathy] is a breathtaking side effect with rituximab [Rituxan], but we’ve gotten over that. We recognize that it’s a potential problem, but we still prescribe rituximab,” the rheumatologist noted. “We’re probably going to need to address the issue of which of our patients are potentially at higher risk for VTE, and maybe we avoid this class in those patients. Like we now do as we look at patients we think are at increased risk for infection, or multiple sclerosis, or TB, we may also need to think of VTE risk.”
But , he argued. There is a pressing unmet need for new therapies for RA with novel mechanisms of action. Only about one-half of patients on contemporary biologic therapies are still on that agent 5 years after initiating therapy.
“Virtually all our patients are partial responders. Everybody gets some benefit. But true remission is achieved by only a minority,” Dr. Genovese said. “The gap between where we are and where we want to be is actually much greater than we often perceive.”
He reported having financial relationships with AbbVie, which is developing upadacitinib, and more than a dozen other medical companies.
MAUI, HAWAII – Rheumatologists, regulatory agencies, and the pharmaceutical industry all have gone off the deep end in their fretting over what appears to be a low rate of venous thromboembolic events in the major randomized trials of the oral Janus kinase inhibitors for RA, Mark C. Genovese, MD, asserted at the 2018 Rheumatology Winter Clinical Symposium.
“The reality is all of our drugs pose potential risks. Unfortunately, I think that at least for the moment, the field has turned all attention in one direction: VTE [venous thromboembolic] events. I suspect there’s some truth [to the possible associated risk]. Certainly we are seeing these events. The question is, how overdone is this?” according to Dr. Genovese, professor of medicine and cochief of the division of immunology and rheumatology at Stanford (Calif.) University.
“I think the upadacitinib data has been entirely overshadowed by concerns about VTEs,” he said. “In the last year, we saw three significant phase 3 studies on upadacitinib arrive in the rheumatology community, and I think the only thing we talked about was VTEs.”
All parties interested in developing Janus kinase (JAK) inhibitors for the treatment of RA began to freak out about a possible increase in VTEs when in April 2017 the Food and Drug Administration turned down Eli Lilly and Incyte’s initial application for marketing approval of the JAK inhibitor baricitinib. Among the problems the agency cited was evidence of potential thrombotic risk.
The VTE rate in baricitinib clinical trials up to 48 weeks in duration was 0.53 events/100 patient-years, with no significant difference in risk between the2-mg and 4-mg doses. This appears to be a class effect for the oral JAK inhibitors, as low rates of VTE, albeit numerically higher than in placebo-treated controls, have also been recorded in the RA development programs for tofacitinib (Xeljanz) as well as the investigational agents filgotinib and upadacitinib, the rheumatologist noted.
This begs the question of whether these VTE rates are significantly higher than background rates in patients with RA or other rheumatologic diseases, which are known to be elevated relative to the general population. Indeed, a retrospective study of insurance claims data by investigators at Brigham and Women’s Hospital, Boston, concluded that the VTE rate in RA patients was 0.61 events/100 patient-years, 120% greater than in a matched patient population without RA. After fully adjusting for comorbid conditions and demographics, the relative risk increase associated with RA dropped to 40%, still significantly higher than in controls (Arthritis Care Res [Hoboken]. 2013 Oct;65[10]:1600-7).
Similarly, Canadian investigators conducted a meta-analysis of 25 studies with VTE data in patients with RA, systemic lupus erythematosus, Sjögren’s syndrome, systemic sclerosis, or inflammatory myositis. This meta-analysis included 10 studies of more than 5.2 million RA patients and nearly 900,000 controls. The conclusion: each of these rheumatic diseases was associated with a VTE rate more than three times higher than in the general population (Arthritis Res Ther. 2014 Sep 25;16[5]:435).
“Patients with RA are at higher risk for VTE than those without RA. It’s unfortunate, and it’s certainly something I don’t think many of us have thought much about before. It’s something we don’t often get to see and something we don’t like to think about,” the rheumatologist observed.
Dr. Genovese admitted to a degree of personal frustration with the current tunnel vision focus on VTEs in JAK inhibitor trials. At the 2017 annual meeting of the American College of Rheumatology he presented the results of the phase 3 SELECT-BEYOND study in which 499 RA patients who had previously failed to respond or were intolerant to biologic therapy were randomized to once daily upadacitinib at 15 or 30 mg or placebo on top of background methotrexate. At week 12, the ACR 20 response rate was 65% for upadacitinib at 15 mg, 56% at 30 mg, and 28% in placebo-treated controls.
“That’s almost a 40% placebo-adjusted response rate. In fact, it’s the highest response I’ve ever seen in a biologic inadequate-responder population. This really looked pretty good, but I don’t think anyone ever took notice. Why not? Because we were all worried about VTE,” he said.
There were in fact a handful of VTEs in upadacitinib-treated patients, Dr. Genovese was quick to note. But he was more impressed by the week 12 ACR 20 responses in patients who had previously failed on three or more biologics: 71% with upadacitinib at 15 mg and 50% at 30 mg, compared with 23% in controls. Moreover, among patients with a baseline history of failure to respond to anti–interleukin-6 therapy, the week 12 ACR 20 rate was 56% with upadacitinib at 15 mg and 58% at 30 mg, versus 20% in controls.
“This looks like a pretty effective drug for patients who’ve failed everything else in our practice,” he commented.
Dr. Genovese reminded his audience that the rheumatology community has a history of overreacting to safety signals in the early days after introduction of new therapies. Examples: tuberculosis with tumor necrosis factor inhibitors, lymphoma with abatacept (Orencia), lymphoma with anti–tumor necrosis factor agents, and cardiovascular events with anti–interleukin-6 inhibition.
“PML [progressive multifocal leukoencephalopathy] is a breathtaking side effect with rituximab [Rituxan], but we’ve gotten over that. We recognize that it’s a potential problem, but we still prescribe rituximab,” the rheumatologist noted. “We’re probably going to need to address the issue of which of our patients are potentially at higher risk for VTE, and maybe we avoid this class in those patients. Like we now do as we look at patients we think are at increased risk for infection, or multiple sclerosis, or TB, we may also need to think of VTE risk.”
But , he argued. There is a pressing unmet need for new therapies for RA with novel mechanisms of action. Only about one-half of patients on contemporary biologic therapies are still on that agent 5 years after initiating therapy.
“Virtually all our patients are partial responders. Everybody gets some benefit. But true remission is achieved by only a minority,” Dr. Genovese said. “The gap between where we are and where we want to be is actually much greater than we often perceive.”
He reported having financial relationships with AbbVie, which is developing upadacitinib, and more than a dozen other medical companies.
EXPERT ANALYSIS FROM RWCS 2018
‘Real-world’ study finds treat-to-target benefits out to 5 years
A treat-to-target (T2T) strategy in daily clinical practice for patients with early rheumatoid arthritis proved successful in maintaining good disease- and patient-related outcomes over a 5-year period at two rheumatology clinics in the Netherlands.
The observational study builds on previous research on the long-term results of continuous application of T2T strategies in rheumatoid arthritis, for which there have been few published studies. “Long-term data from more recent randomized controlled clinical trials, using a T2T approach and biologicals, have shown good clinical outcomes. However, the generalizability of these results is hampered by the selection of specific patient groups in clinical trials and strict exclusion criteria. Patients seen in real-life practice may differ substantially from those in randomized clinical trials,” first author Letty G.A. Versteeg of Medisch Spectrum Twente, Enschede, the Netherlands, and her colleagues wrote in Clinical Rheumatology.
The investigators examined outcomes for 229 patients with very early RA who enrolled in the Dutch Rheumatoid Arthritis Monitoring (DREAM) remission induction cohort during 2006-2009, which included 5 years of follow-up for 171 of the patients. These patients underwent a protocoled T2T strategy aimed at remission, defined as a 28-joint Disease Activity Score (DAS28) of less than 2.6.
“In previous publications on the [DREAM] remission induction cohort, successful implementation of T2T in daily clinical practice was demonstrated. Achieving remission within the first year of treatment was shown to be a realistic goal for an important proportion of patients,” the authors wrote.
All patients started methotrexate monotherapy at an initial dosage of 15 mg/week that could be increased to a maximum dosage of 25 mg/week in week 8. Patients took folic acid on the second day after methotrexate. By week 12, those with persistent disease activity added sulfasalazine, starting at 2,000 mg/day and increasing if necessary to a maximum of 3,000 mg/day at week 20. Patients whose DAS28 remained at 3.2 or greater at week 24 received a tumor necrosis factor inhibitor. Those who reached remission had no change in medication, and when remission lasted for at least 6 months, medication was gradually tapered and eventually discontinued. Patients who had flares in which disease activity increased to a DAS28 of 2.6 and higher restarted their last effective medication or dosage, which could subsequently be intensified if necessary. Patients with comorbidities and contraindications for medication were not excluded because deviations from the protocol were allowed. The protocol also allowed concomitant treatment with NSAIDs, prednisolone at a dosage of less than 10 mg/day, and intra-articular corticosteroid injections.
The rate of DAS28-defined remission rose to 63% (126 of 199 patients) by the end of the first year, and only 5% had high disease activity at 24 weeks. The rate of remission remained stable over the next 4 years. This rate of remission was reflected as a drop from an overall mean DAS28 of 4.93 at baseline to 2.49 at 5 years. The majority of the drop in DAS28 occurred during the first 3 months (–1.63 points), and by the end of the first year of treatment, mean disease activity stayed below 2.6 on the DAS28.
The investigators saw a sustained remission at least once in 144 of the 171 patients with 5-year outcome data available, including sustained remission for 1 year or longer in 115. Median time to the first sustained remission proved to be 50 weeks, and half had this last less than 97 weeks and half more than 97 weeks.
During the 5-year follow-up, 17% of patients received treatment with biologics, with a median start of their first biologic at about 54 weeks after baseline. This first biologic was used continuously for a median of 29 weeks, and close to one-third of patients who started a biologic switched to a second biologic after a median duration of 41 weeks on the first. About two-thirds did not need a second biologic. A total of 66% of patients who took a biologic had at least one period of sustained remission.
Functional disability improved overall at 5 years as determined by Health Assessment Questionnaire (HAQ) scores that were available for 107 patients. HAQ scores decreased from a median of 1.125 at baseline to 0.375 after 24 weeks (P less than .001), where they remained stable throughout the rest of follow-up. Overall, nearly 70% of the patients with available 5-year data had a change in their individual HAQ score that was clinically meaningful from baseline to 24 weeks.
“Our study describes long-term outcome of implementation and continuous application of T2T to RA patients in daily clinical practice. The outcomes are similar to or even better than the results of T2T randomized clinical trials, in which strict selection of patients and controlled conditions were followed. These ‘real-life data’ are of important additional value in the evidence for the effectiveness of a T2T approach in RA patients,” the investigators concluded.
They had no disclosures to report.
SOURCE: Versteeg G et al. Clin Rheumatol. 2018 Feb 1. doi: 10.1007/s10067-017-3962-5.
A treat-to-target (T2T) strategy in daily clinical practice for patients with early rheumatoid arthritis proved successful in maintaining good disease- and patient-related outcomes over a 5-year period at two rheumatology clinics in the Netherlands.
The observational study builds on previous research on the long-term results of continuous application of T2T strategies in rheumatoid arthritis, for which there have been few published studies. “Long-term data from more recent randomized controlled clinical trials, using a T2T approach and biologicals, have shown good clinical outcomes. However, the generalizability of these results is hampered by the selection of specific patient groups in clinical trials and strict exclusion criteria. Patients seen in real-life practice may differ substantially from those in randomized clinical trials,” first author Letty G.A. Versteeg of Medisch Spectrum Twente, Enschede, the Netherlands, and her colleagues wrote in Clinical Rheumatology.
The investigators examined outcomes for 229 patients with very early RA who enrolled in the Dutch Rheumatoid Arthritis Monitoring (DREAM) remission induction cohort during 2006-2009, which included 5 years of follow-up for 171 of the patients. These patients underwent a protocoled T2T strategy aimed at remission, defined as a 28-joint Disease Activity Score (DAS28) of less than 2.6.
“In previous publications on the [DREAM] remission induction cohort, successful implementation of T2T in daily clinical practice was demonstrated. Achieving remission within the first year of treatment was shown to be a realistic goal for an important proportion of patients,” the authors wrote.
All patients started methotrexate monotherapy at an initial dosage of 15 mg/week that could be increased to a maximum dosage of 25 mg/week in week 8. Patients took folic acid on the second day after methotrexate. By week 12, those with persistent disease activity added sulfasalazine, starting at 2,000 mg/day and increasing if necessary to a maximum of 3,000 mg/day at week 20. Patients whose DAS28 remained at 3.2 or greater at week 24 received a tumor necrosis factor inhibitor. Those who reached remission had no change in medication, and when remission lasted for at least 6 months, medication was gradually tapered and eventually discontinued. Patients who had flares in which disease activity increased to a DAS28 of 2.6 and higher restarted their last effective medication or dosage, which could subsequently be intensified if necessary. Patients with comorbidities and contraindications for medication were not excluded because deviations from the protocol were allowed. The protocol also allowed concomitant treatment with NSAIDs, prednisolone at a dosage of less than 10 mg/day, and intra-articular corticosteroid injections.
The rate of DAS28-defined remission rose to 63% (126 of 199 patients) by the end of the first year, and only 5% had high disease activity at 24 weeks. The rate of remission remained stable over the next 4 years. This rate of remission was reflected as a drop from an overall mean DAS28 of 4.93 at baseline to 2.49 at 5 years. The majority of the drop in DAS28 occurred during the first 3 months (–1.63 points), and by the end of the first year of treatment, mean disease activity stayed below 2.6 on the DAS28.
The investigators saw a sustained remission at least once in 144 of the 171 patients with 5-year outcome data available, including sustained remission for 1 year or longer in 115. Median time to the first sustained remission proved to be 50 weeks, and half had this last less than 97 weeks and half more than 97 weeks.
During the 5-year follow-up, 17% of patients received treatment with biologics, with a median start of their first biologic at about 54 weeks after baseline. This first biologic was used continuously for a median of 29 weeks, and close to one-third of patients who started a biologic switched to a second biologic after a median duration of 41 weeks on the first. About two-thirds did not need a second biologic. A total of 66% of patients who took a biologic had at least one period of sustained remission.
Functional disability improved overall at 5 years as determined by Health Assessment Questionnaire (HAQ) scores that were available for 107 patients. HAQ scores decreased from a median of 1.125 at baseline to 0.375 after 24 weeks (P less than .001), where they remained stable throughout the rest of follow-up. Overall, nearly 70% of the patients with available 5-year data had a change in their individual HAQ score that was clinically meaningful from baseline to 24 weeks.
“Our study describes long-term outcome of implementation and continuous application of T2T to RA patients in daily clinical practice. The outcomes are similar to or even better than the results of T2T randomized clinical trials, in which strict selection of patients and controlled conditions were followed. These ‘real-life data’ are of important additional value in the evidence for the effectiveness of a T2T approach in RA patients,” the investigators concluded.
They had no disclosures to report.
SOURCE: Versteeg G et al. Clin Rheumatol. 2018 Feb 1. doi: 10.1007/s10067-017-3962-5.
A treat-to-target (T2T) strategy in daily clinical practice for patients with early rheumatoid arthritis proved successful in maintaining good disease- and patient-related outcomes over a 5-year period at two rheumatology clinics in the Netherlands.
The observational study builds on previous research on the long-term results of continuous application of T2T strategies in rheumatoid arthritis, for which there have been few published studies. “Long-term data from more recent randomized controlled clinical trials, using a T2T approach and biologicals, have shown good clinical outcomes. However, the generalizability of these results is hampered by the selection of specific patient groups in clinical trials and strict exclusion criteria. Patients seen in real-life practice may differ substantially from those in randomized clinical trials,” first author Letty G.A. Versteeg of Medisch Spectrum Twente, Enschede, the Netherlands, and her colleagues wrote in Clinical Rheumatology.
The investigators examined outcomes for 229 patients with very early RA who enrolled in the Dutch Rheumatoid Arthritis Monitoring (DREAM) remission induction cohort during 2006-2009, which included 5 years of follow-up for 171 of the patients. These patients underwent a protocoled T2T strategy aimed at remission, defined as a 28-joint Disease Activity Score (DAS28) of less than 2.6.
“In previous publications on the [DREAM] remission induction cohort, successful implementation of T2T in daily clinical practice was demonstrated. Achieving remission within the first year of treatment was shown to be a realistic goal for an important proportion of patients,” the authors wrote.
All patients started methotrexate monotherapy at an initial dosage of 15 mg/week that could be increased to a maximum dosage of 25 mg/week in week 8. Patients took folic acid on the second day after methotrexate. By week 12, those with persistent disease activity added sulfasalazine, starting at 2,000 mg/day and increasing if necessary to a maximum of 3,000 mg/day at week 20. Patients whose DAS28 remained at 3.2 or greater at week 24 received a tumor necrosis factor inhibitor. Those who reached remission had no change in medication, and when remission lasted for at least 6 months, medication was gradually tapered and eventually discontinued. Patients who had flares in which disease activity increased to a DAS28 of 2.6 and higher restarted their last effective medication or dosage, which could subsequently be intensified if necessary. Patients with comorbidities and contraindications for medication were not excluded because deviations from the protocol were allowed. The protocol also allowed concomitant treatment with NSAIDs, prednisolone at a dosage of less than 10 mg/day, and intra-articular corticosteroid injections.
The rate of DAS28-defined remission rose to 63% (126 of 199 patients) by the end of the first year, and only 5% had high disease activity at 24 weeks. The rate of remission remained stable over the next 4 years. This rate of remission was reflected as a drop from an overall mean DAS28 of 4.93 at baseline to 2.49 at 5 years. The majority of the drop in DAS28 occurred during the first 3 months (–1.63 points), and by the end of the first year of treatment, mean disease activity stayed below 2.6 on the DAS28.
The investigators saw a sustained remission at least once in 144 of the 171 patients with 5-year outcome data available, including sustained remission for 1 year or longer in 115. Median time to the first sustained remission proved to be 50 weeks, and half had this last less than 97 weeks and half more than 97 weeks.
During the 5-year follow-up, 17% of patients received treatment with biologics, with a median start of their first biologic at about 54 weeks after baseline. This first biologic was used continuously for a median of 29 weeks, and close to one-third of patients who started a biologic switched to a second biologic after a median duration of 41 weeks on the first. About two-thirds did not need a second biologic. A total of 66% of patients who took a biologic had at least one period of sustained remission.
Functional disability improved overall at 5 years as determined by Health Assessment Questionnaire (HAQ) scores that were available for 107 patients. HAQ scores decreased from a median of 1.125 at baseline to 0.375 after 24 weeks (P less than .001), where they remained stable throughout the rest of follow-up. Overall, nearly 70% of the patients with available 5-year data had a change in their individual HAQ score that was clinically meaningful from baseline to 24 weeks.
“Our study describes long-term outcome of implementation and continuous application of T2T to RA patients in daily clinical practice. The outcomes are similar to or even better than the results of T2T randomized clinical trials, in which strict selection of patients and controlled conditions were followed. These ‘real-life data’ are of important additional value in the evidence for the effectiveness of a T2T approach in RA patients,” the investigators concluded.
They had no disclosures to report.
SOURCE: Versteeg G et al. Clin Rheumatol. 2018 Feb 1. doi: 10.1007/s10067-017-3962-5.
FROM CLINICAL RHEUMATOLOGY
Key clinical point:
Major finding: The rate of DAS28 remission rose to 63% by the end of the first year and remained stable over the next 4 years.
Study details: An observational cohort study of 171 patients with 5 years of follow-up data.
Disclosures: The investigators had no disclosures to report.
Source: Versteeg G et al. Clin Rheumatol. 2018 Feb 1. doi: 10.1007/s10067-017-3962-5.
ACR sounds more welcoming tone in new biosimilars position paper
The American College of Rheumatology has shifted from a more cautious stance toward the use of biosimilars in clinical practice to now recommend in a new position statement that health care providers incorporate biosimilars, where appropriate, into treatment regimens for their patients living with rheumatic diseases.
“Now that biosimilars have been used successfully in Europe, with rigorously acquired data supporting their broader use, and , S. Louis Bridges Jr., MD, PhD, chair of the ACR Committee on Research, wrote with seven other authors of the position statement in Arthritis & Rheumatology.
The ACR position statement addresses the issues of immunogenicity, extrapolation of indications, interchangeability, substitution, switching, and cost surrounding biosimilars.
The position statement’s authors said they expect switching and nonmedical substitution to become as common in the United States as it is in the rest of the world. They do not anticipate efficacy and safety issues for biosimilars based on available data regarding switching between reference products and biosimilars and their understanding of product drift.
“However, we encourage vigorous postmarketing surveillance of both biosimilars and their reference products as we enter the era where patients may undergo multiple switches as a result of insurance company and [pharmacy benefits manager] formulary preferences,” they said.
Immunogenicity
Concerns about immunogenicity for biosimilars approved in the United States have mostly been well addressed through studies showing similar frequencies of binding and neutralizing antidrug antibodies (ADA) in biosimilars and their reference products. Furthermore, no safety signals between biosimilars and their reference products have been observed that suggest a differential effect of ADA on efficacy, safety, or patient outcomes, the authors said. But they noted that “if immunogenicity findings are to be extrapolated from one disease to additional indications, the subjects being studied should be those most likely to develop ADA, such as subjects not receiving concomitant immunosuppressive medications.” The results of comparative immunogenicity studies carried out to date also indicate that “a patient who develops ADA to a reference drug with resultant loss of clinical response should not be switched to its biosimilar.”
Again, the authors said postmarketing pharmacovigilance using observational registry data would be critical to assessing the effect of switching on immunogenicity.
Extrapolation of indications
The extrapolation of biosimilars to reference product indications for which the biosimilar was not assessed in clinical trials continues to “be an area of uneasiness” among clinicians “who are surprised to find” that a biosimilar can be approved for inflammatory bowel disease in the absence of clinical trials in the relevant patient populations, the authors said. In geographic areas where it is not mandatory to use biosimilars, this lack of confidence in extrapolation of indications may limit their acceptance, the authors wrote, but data from studies such as NOR-SWITCH and DANBIO have provided reassuring evidence to support regulated extrapolation of indication for biosimilars.
However, since extrapolation of indications also applies to pediatric patients who often metabolize drugs faster than adults, the position paper says that “it may be important” to conduct pharmacodynamic and pharmacokinetic studies in children as well as postmarketing surveillance “since potential immunogenicity may be of particular importance in these younger patients with chronic diseases who might encounter several biological agents during their lifetime.”
Substitution, interchangeability, and switching
The “interchangeability” regulatory pathway in the United States that would allow substitution at the point of dispensing has not been finalized, but most states have enacted, or are in the process of enacting, legislation to regulate the practice, the statement says.
While substitution describes a change made by someone other than the prescriber, the authors note that switching defines the “intentional change initiated by a health care provider in partnership with the patient” for economic or medical reasons. Switching has been studied most often in open-label extension studies of biosimilar clinic trials and has shown no loss of efficacy or increase in adverse events.
Dr. Fleischmann contended that substitution, extrapolation, and interchangeability of biosimilars in clinical practice remain gray areas. For example, in a clinical trial, patients switching from a reference product to a biosimilar may show equivalency of clinical response and adverse events. “But as rheumatologists, we don’t treat groups of patients; we treat individual patients and here the results may be different,” he wrote in Arthritis & Rheumatology.
While the white paper appropriately points out that interchangeability among multiple biosimilars is a question that should be answered in postmarketing registries, Dr. Fleischmann noted that no interchangeability study has been reported, even though the FDA has issued guidance on how a study should be done.
“Although interchangeability may be safe and effective in many patients, until the results of such a study are available and properly analyzed, it is only conjecture that interchangeability is appropriate and safe,” he said.
Costs
The white paper acknowledged that the “only anticipated advantage” of a biosimilar over its reference product was lower cost, since both drugs should be therapeutically equivalent. “The degree to which the availability of biosimilars in the U.S. will drive down the cost of biologic therapy, and who will benefit from any cost reductions remains to be seen,” the authors wrote.
“To incentivize the use of biosimilars,” the authors suggested that “commercial and government insurance programs could harmonize drug prices with patients’ out-of-pocket costs and provider reimbursement. Currently, however, patients with commercial insurance are likely to have similar copayments for both biosimilars and originator biologics because of [pharmacy benefits manager]– or plan-mandated patient cost sharing. Also, patients’ out-of-pocket costs for biosimilars in the Medicare Part D (self-administered drug) program likely will be higher than for originator biologics because of a flaw that maintains, rather than reduces, biosimilar patient cost sharing in the coverage gap (also known as the “donut hole”) until 2020.”
It is not at all clear that a biosimilar would be cheaper for the individual patient, Dr. Fleischmann said. “It may be cheaper to the pharmacy benefit management firm, but this may not really help patient access to these medications. It is also not clear that nonmedical substitution will be effective in every patient nor has it been demonstrated that extrapolation is effective,” he wrote.
No disclosures were listed for the authors of the position statement. Dr. Fleischmann is a consultant for AbbVie, Amgen, Bristol-Myers Squibb, Celltrion, Eli Lilly, GlaxoSmithKline, Novartis, Pfizer, Sanofi Aventis, and UCB.
SOURCES: Bridges S et al. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40388; and Fleischmann R. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40402
The American College of Rheumatology has shifted from a more cautious stance toward the use of biosimilars in clinical practice to now recommend in a new position statement that health care providers incorporate biosimilars, where appropriate, into treatment regimens for their patients living with rheumatic diseases.
“Now that biosimilars have been used successfully in Europe, with rigorously acquired data supporting their broader use, and , S. Louis Bridges Jr., MD, PhD, chair of the ACR Committee on Research, wrote with seven other authors of the position statement in Arthritis & Rheumatology.
The ACR position statement addresses the issues of immunogenicity, extrapolation of indications, interchangeability, substitution, switching, and cost surrounding biosimilars.
The position statement’s authors said they expect switching and nonmedical substitution to become as common in the United States as it is in the rest of the world. They do not anticipate efficacy and safety issues for biosimilars based on available data regarding switching between reference products and biosimilars and their understanding of product drift.
“However, we encourage vigorous postmarketing surveillance of both biosimilars and their reference products as we enter the era where patients may undergo multiple switches as a result of insurance company and [pharmacy benefits manager] formulary preferences,” they said.
Immunogenicity
Concerns about immunogenicity for biosimilars approved in the United States have mostly been well addressed through studies showing similar frequencies of binding and neutralizing antidrug antibodies (ADA) in biosimilars and their reference products. Furthermore, no safety signals between biosimilars and their reference products have been observed that suggest a differential effect of ADA on efficacy, safety, or patient outcomes, the authors said. But they noted that “if immunogenicity findings are to be extrapolated from one disease to additional indications, the subjects being studied should be those most likely to develop ADA, such as subjects not receiving concomitant immunosuppressive medications.” The results of comparative immunogenicity studies carried out to date also indicate that “a patient who develops ADA to a reference drug with resultant loss of clinical response should not be switched to its biosimilar.”
Again, the authors said postmarketing pharmacovigilance using observational registry data would be critical to assessing the effect of switching on immunogenicity.
Extrapolation of indications
The extrapolation of biosimilars to reference product indications for which the biosimilar was not assessed in clinical trials continues to “be an area of uneasiness” among clinicians “who are surprised to find” that a biosimilar can be approved for inflammatory bowel disease in the absence of clinical trials in the relevant patient populations, the authors said. In geographic areas where it is not mandatory to use biosimilars, this lack of confidence in extrapolation of indications may limit their acceptance, the authors wrote, but data from studies such as NOR-SWITCH and DANBIO have provided reassuring evidence to support regulated extrapolation of indication for biosimilars.
However, since extrapolation of indications also applies to pediatric patients who often metabolize drugs faster than adults, the position paper says that “it may be important” to conduct pharmacodynamic and pharmacokinetic studies in children as well as postmarketing surveillance “since potential immunogenicity may be of particular importance in these younger patients with chronic diseases who might encounter several biological agents during their lifetime.”
Substitution, interchangeability, and switching
The “interchangeability” regulatory pathway in the United States that would allow substitution at the point of dispensing has not been finalized, but most states have enacted, or are in the process of enacting, legislation to regulate the practice, the statement says.
While substitution describes a change made by someone other than the prescriber, the authors note that switching defines the “intentional change initiated by a health care provider in partnership with the patient” for economic or medical reasons. Switching has been studied most often in open-label extension studies of biosimilar clinic trials and has shown no loss of efficacy or increase in adverse events.
Dr. Fleischmann contended that substitution, extrapolation, and interchangeability of biosimilars in clinical practice remain gray areas. For example, in a clinical trial, patients switching from a reference product to a biosimilar may show equivalency of clinical response and adverse events. “But as rheumatologists, we don’t treat groups of patients; we treat individual patients and here the results may be different,” he wrote in Arthritis & Rheumatology.
While the white paper appropriately points out that interchangeability among multiple biosimilars is a question that should be answered in postmarketing registries, Dr. Fleischmann noted that no interchangeability study has been reported, even though the FDA has issued guidance on how a study should be done.
“Although interchangeability may be safe and effective in many patients, until the results of such a study are available and properly analyzed, it is only conjecture that interchangeability is appropriate and safe,” he said.
Costs
The white paper acknowledged that the “only anticipated advantage” of a biosimilar over its reference product was lower cost, since both drugs should be therapeutically equivalent. “The degree to which the availability of biosimilars in the U.S. will drive down the cost of biologic therapy, and who will benefit from any cost reductions remains to be seen,” the authors wrote.
“To incentivize the use of biosimilars,” the authors suggested that “commercial and government insurance programs could harmonize drug prices with patients’ out-of-pocket costs and provider reimbursement. Currently, however, patients with commercial insurance are likely to have similar copayments for both biosimilars and originator biologics because of [pharmacy benefits manager]– or plan-mandated patient cost sharing. Also, patients’ out-of-pocket costs for biosimilars in the Medicare Part D (self-administered drug) program likely will be higher than for originator biologics because of a flaw that maintains, rather than reduces, biosimilar patient cost sharing in the coverage gap (also known as the “donut hole”) until 2020.”
It is not at all clear that a biosimilar would be cheaper for the individual patient, Dr. Fleischmann said. “It may be cheaper to the pharmacy benefit management firm, but this may not really help patient access to these medications. It is also not clear that nonmedical substitution will be effective in every patient nor has it been demonstrated that extrapolation is effective,” he wrote.
No disclosures were listed for the authors of the position statement. Dr. Fleischmann is a consultant for AbbVie, Amgen, Bristol-Myers Squibb, Celltrion, Eli Lilly, GlaxoSmithKline, Novartis, Pfizer, Sanofi Aventis, and UCB.
SOURCES: Bridges S et al. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40388; and Fleischmann R. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40402
The American College of Rheumatology has shifted from a more cautious stance toward the use of biosimilars in clinical practice to now recommend in a new position statement that health care providers incorporate biosimilars, where appropriate, into treatment regimens for their patients living with rheumatic diseases.
“Now that biosimilars have been used successfully in Europe, with rigorously acquired data supporting their broader use, and , S. Louis Bridges Jr., MD, PhD, chair of the ACR Committee on Research, wrote with seven other authors of the position statement in Arthritis & Rheumatology.
The ACR position statement addresses the issues of immunogenicity, extrapolation of indications, interchangeability, substitution, switching, and cost surrounding biosimilars.
The position statement’s authors said they expect switching and nonmedical substitution to become as common in the United States as it is in the rest of the world. They do not anticipate efficacy and safety issues for biosimilars based on available data regarding switching between reference products and biosimilars and their understanding of product drift.
“However, we encourage vigorous postmarketing surveillance of both biosimilars and their reference products as we enter the era where patients may undergo multiple switches as a result of insurance company and [pharmacy benefits manager] formulary preferences,” they said.
Immunogenicity
Concerns about immunogenicity for biosimilars approved in the United States have mostly been well addressed through studies showing similar frequencies of binding and neutralizing antidrug antibodies (ADA) in biosimilars and their reference products. Furthermore, no safety signals between biosimilars and their reference products have been observed that suggest a differential effect of ADA on efficacy, safety, or patient outcomes, the authors said. But they noted that “if immunogenicity findings are to be extrapolated from one disease to additional indications, the subjects being studied should be those most likely to develop ADA, such as subjects not receiving concomitant immunosuppressive medications.” The results of comparative immunogenicity studies carried out to date also indicate that “a patient who develops ADA to a reference drug with resultant loss of clinical response should not be switched to its biosimilar.”
Again, the authors said postmarketing pharmacovigilance using observational registry data would be critical to assessing the effect of switching on immunogenicity.
Extrapolation of indications
The extrapolation of biosimilars to reference product indications for which the biosimilar was not assessed in clinical trials continues to “be an area of uneasiness” among clinicians “who are surprised to find” that a biosimilar can be approved for inflammatory bowel disease in the absence of clinical trials in the relevant patient populations, the authors said. In geographic areas where it is not mandatory to use biosimilars, this lack of confidence in extrapolation of indications may limit their acceptance, the authors wrote, but data from studies such as NOR-SWITCH and DANBIO have provided reassuring evidence to support regulated extrapolation of indication for biosimilars.
However, since extrapolation of indications also applies to pediatric patients who often metabolize drugs faster than adults, the position paper says that “it may be important” to conduct pharmacodynamic and pharmacokinetic studies in children as well as postmarketing surveillance “since potential immunogenicity may be of particular importance in these younger patients with chronic diseases who might encounter several biological agents during their lifetime.”
Substitution, interchangeability, and switching
The “interchangeability” regulatory pathway in the United States that would allow substitution at the point of dispensing has not been finalized, but most states have enacted, or are in the process of enacting, legislation to regulate the practice, the statement says.
While substitution describes a change made by someone other than the prescriber, the authors note that switching defines the “intentional change initiated by a health care provider in partnership with the patient” for economic or medical reasons. Switching has been studied most often in open-label extension studies of biosimilar clinic trials and has shown no loss of efficacy or increase in adverse events.
Dr. Fleischmann contended that substitution, extrapolation, and interchangeability of biosimilars in clinical practice remain gray areas. For example, in a clinical trial, patients switching from a reference product to a biosimilar may show equivalency of clinical response and adverse events. “But as rheumatologists, we don’t treat groups of patients; we treat individual patients and here the results may be different,” he wrote in Arthritis & Rheumatology.
While the white paper appropriately points out that interchangeability among multiple biosimilars is a question that should be answered in postmarketing registries, Dr. Fleischmann noted that no interchangeability study has been reported, even though the FDA has issued guidance on how a study should be done.
“Although interchangeability may be safe and effective in many patients, until the results of such a study are available and properly analyzed, it is only conjecture that interchangeability is appropriate and safe,” he said.
Costs
The white paper acknowledged that the “only anticipated advantage” of a biosimilar over its reference product was lower cost, since both drugs should be therapeutically equivalent. “The degree to which the availability of biosimilars in the U.S. will drive down the cost of biologic therapy, and who will benefit from any cost reductions remains to be seen,” the authors wrote.
“To incentivize the use of biosimilars,” the authors suggested that “commercial and government insurance programs could harmonize drug prices with patients’ out-of-pocket costs and provider reimbursement. Currently, however, patients with commercial insurance are likely to have similar copayments for both biosimilars and originator biologics because of [pharmacy benefits manager]– or plan-mandated patient cost sharing. Also, patients’ out-of-pocket costs for biosimilars in the Medicare Part D (self-administered drug) program likely will be higher than for originator biologics because of a flaw that maintains, rather than reduces, biosimilar patient cost sharing in the coverage gap (also known as the “donut hole”) until 2020.”
It is not at all clear that a biosimilar would be cheaper for the individual patient, Dr. Fleischmann said. “It may be cheaper to the pharmacy benefit management firm, but this may not really help patient access to these medications. It is also not clear that nonmedical substitution will be effective in every patient nor has it been demonstrated that extrapolation is effective,” he wrote.
No disclosures were listed for the authors of the position statement. Dr. Fleischmann is a consultant for AbbVie, Amgen, Bristol-Myers Squibb, Celltrion, Eli Lilly, GlaxoSmithKline, Novartis, Pfizer, Sanofi Aventis, and UCB.
SOURCES: Bridges S et al. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40388; and Fleischmann R. Arthritis Rheumatol. 2018 Feb 7. doi: 10.1002/art.40402
FROM ARTHRITIS & RHEUMATOLOGY
Insights revealed into rheumatologist-patient discussions about biologics
A small study of the discussions of biologic treatment options between rheumatoid arthritis patients and their rheumatologists suggests they are limited in scope and may be influenced by clinicians’ preconceived notions about patient preferences.
In an analysis of video footage from office visits of 48 rheumatoid arthritis (RA) patients initiating biologics with 16 rheumatologists, only 5.6 minutes out of visits with an average duration of 15 minutes were spent discussing biologic therapy options. Postvisit interviews with patients highlighted problems in rheumatologists’ awareness of patients’ familiarity with biologic treatment options, particularly a lack of knowledge about intravenous (IV) options, according to findings from the ethnographic, observational study conducted by Nicholas Kottak, PhD, of Ethnographic Solutions LLC, and his colleagues and published in Arthritis Care & Research.
Of the 48 visits, the researchers found that subcutaneous (SC) therapy options were discussed in 45 visits and IV options were discussed in 35. In 27 of the 35 visits in which IV options were mentioned, the rheumatologists did not define or describe IV administration.
In postvisit interviews, all patients said they were previously aware that biologic therapy was available subcutaneously, but 22 entered their visit with no previous knowledge of IV options, 19 of whom remained unfamiliar after their visit.
According to the researchers, some patients said that they would prefer to receive IV biologic treatment and some would prefer SC delivery. However, all 16 participating rheumatologists thought that IV therapy would be less convenient for the patient than SC therapy, but only 22 of 48 patients said inconvenience was the main barrier to IV therapy. A total of 23 patients thought SC therapy would be easier, 17 thought IV therapy would be easier, 4 thought they would be the same, and 4 were unsure.
The way in which mode of biologic administration is discussed may also have financial implications, particularly for Medicare patients. “During 26 visits with Medicare patients, IV and SC therapies were mentioned during 22 and 23 visits, respectively. This is noteworthy as Medicare with supplemental insurance may preferably cover IV over SC therapy, since IV therapy qualifies as a medical benefit (Part B) and SC therapy falls under pharmacy benefits (Part D),” Dr. Kottak and his associates wrote.
• Preparing patients for biologic therapy at an earlier disease stage.
• Providing educational tools such as pamphlets and flyers.
• Increasing time spent with the patient and/or including nurse practitioners and physician assistants in the follow-up portion of the patient visit.
• Creating a treatment alliance with a patient to make the process more collaborative.
• Allowing patients to choose which biologic medication they start with while emphasizing the potential for positive outcomes and relative safety.
• Researching shared decision making between patients and physicians in a randomized, outcomes-based setting.
Janssen provided funding for the study as well as the writing of the report. Ethnographic Solutions received financial compensation from Janssen. Three of the six authors are employees of Janssen. The other researchers reported various financial disclosures, including, but not limited to, stock ownership, speaking fees, and advisory board positions.
SOURCE: Kottak N et al. Arthritis Care Res. 2018 Feb 5. doi: 10/1002/acr.23527
A small study of the discussions of biologic treatment options between rheumatoid arthritis patients and their rheumatologists suggests they are limited in scope and may be influenced by clinicians’ preconceived notions about patient preferences.
In an analysis of video footage from office visits of 48 rheumatoid arthritis (RA) patients initiating biologics with 16 rheumatologists, only 5.6 minutes out of visits with an average duration of 15 minutes were spent discussing biologic therapy options. Postvisit interviews with patients highlighted problems in rheumatologists’ awareness of patients’ familiarity with biologic treatment options, particularly a lack of knowledge about intravenous (IV) options, according to findings from the ethnographic, observational study conducted by Nicholas Kottak, PhD, of Ethnographic Solutions LLC, and his colleagues and published in Arthritis Care & Research.
Of the 48 visits, the researchers found that subcutaneous (SC) therapy options were discussed in 45 visits and IV options were discussed in 35. In 27 of the 35 visits in which IV options were mentioned, the rheumatologists did not define or describe IV administration.
In postvisit interviews, all patients said they were previously aware that biologic therapy was available subcutaneously, but 22 entered their visit with no previous knowledge of IV options, 19 of whom remained unfamiliar after their visit.
According to the researchers, some patients said that they would prefer to receive IV biologic treatment and some would prefer SC delivery. However, all 16 participating rheumatologists thought that IV therapy would be less convenient for the patient than SC therapy, but only 22 of 48 patients said inconvenience was the main barrier to IV therapy. A total of 23 patients thought SC therapy would be easier, 17 thought IV therapy would be easier, 4 thought they would be the same, and 4 were unsure.
The way in which mode of biologic administration is discussed may also have financial implications, particularly for Medicare patients. “During 26 visits with Medicare patients, IV and SC therapies were mentioned during 22 and 23 visits, respectively. This is noteworthy as Medicare with supplemental insurance may preferably cover IV over SC therapy, since IV therapy qualifies as a medical benefit (Part B) and SC therapy falls under pharmacy benefits (Part D),” Dr. Kottak and his associates wrote.
• Preparing patients for biologic therapy at an earlier disease stage.
• Providing educational tools such as pamphlets and flyers.
• Increasing time spent with the patient and/or including nurse practitioners and physician assistants in the follow-up portion of the patient visit.
• Creating a treatment alliance with a patient to make the process more collaborative.
• Allowing patients to choose which biologic medication they start with while emphasizing the potential for positive outcomes and relative safety.
• Researching shared decision making between patients and physicians in a randomized, outcomes-based setting.
Janssen provided funding for the study as well as the writing of the report. Ethnographic Solutions received financial compensation from Janssen. Three of the six authors are employees of Janssen. The other researchers reported various financial disclosures, including, but not limited to, stock ownership, speaking fees, and advisory board positions.
SOURCE: Kottak N et al. Arthritis Care Res. 2018 Feb 5. doi: 10/1002/acr.23527
A small study of the discussions of biologic treatment options between rheumatoid arthritis patients and their rheumatologists suggests they are limited in scope and may be influenced by clinicians’ preconceived notions about patient preferences.
In an analysis of video footage from office visits of 48 rheumatoid arthritis (RA) patients initiating biologics with 16 rheumatologists, only 5.6 minutes out of visits with an average duration of 15 minutes were spent discussing biologic therapy options. Postvisit interviews with patients highlighted problems in rheumatologists’ awareness of patients’ familiarity with biologic treatment options, particularly a lack of knowledge about intravenous (IV) options, according to findings from the ethnographic, observational study conducted by Nicholas Kottak, PhD, of Ethnographic Solutions LLC, and his colleagues and published in Arthritis Care & Research.
Of the 48 visits, the researchers found that subcutaneous (SC) therapy options were discussed in 45 visits and IV options were discussed in 35. In 27 of the 35 visits in which IV options were mentioned, the rheumatologists did not define or describe IV administration.
In postvisit interviews, all patients said they were previously aware that biologic therapy was available subcutaneously, but 22 entered their visit with no previous knowledge of IV options, 19 of whom remained unfamiliar after their visit.
According to the researchers, some patients said that they would prefer to receive IV biologic treatment and some would prefer SC delivery. However, all 16 participating rheumatologists thought that IV therapy would be less convenient for the patient than SC therapy, but only 22 of 48 patients said inconvenience was the main barrier to IV therapy. A total of 23 patients thought SC therapy would be easier, 17 thought IV therapy would be easier, 4 thought they would be the same, and 4 were unsure.
The way in which mode of biologic administration is discussed may also have financial implications, particularly for Medicare patients. “During 26 visits with Medicare patients, IV and SC therapies were mentioned during 22 and 23 visits, respectively. This is noteworthy as Medicare with supplemental insurance may preferably cover IV over SC therapy, since IV therapy qualifies as a medical benefit (Part B) and SC therapy falls under pharmacy benefits (Part D),” Dr. Kottak and his associates wrote.
• Preparing patients for biologic therapy at an earlier disease stage.
• Providing educational tools such as pamphlets and flyers.
• Increasing time spent with the patient and/or including nurse practitioners and physician assistants in the follow-up portion of the patient visit.
• Creating a treatment alliance with a patient to make the process more collaborative.
• Allowing patients to choose which biologic medication they start with while emphasizing the potential for positive outcomes and relative safety.
• Researching shared decision making between patients and physicians in a randomized, outcomes-based setting.
Janssen provided funding for the study as well as the writing of the report. Ethnographic Solutions received financial compensation from Janssen. Three of the six authors are employees of Janssen. The other researchers reported various financial disclosures, including, but not limited to, stock ownership, speaking fees, and advisory board positions.
SOURCE: Kottak N et al. Arthritis Care Res. 2018 Feb 5. doi: 10/1002/acr.23527
FROM ARTHRITIS CARE & RESEARCH
Key clinical point: Provide educational resources about biologics and don’t assume that subcutaneous therapy is more convenient or that patients understand IV therapy terminology and procedures.
Major finding: All 16 participating rheumatologists thought that intravenous therapy would be less convenient for the patient than subcutaneous therapy, but only 22 of 48 patients said inconvenience was the main barrier to IV therapy.
Study details: An ethnographic, observational study of 48 patients and 16 rheumatologists.
Disclosures: Janssen Pharmaceuticals funded the research. Dr. Kottak is president of Ethnographic Solutions LLC, which received compensation from Janssen Pharmaceuticals for this project.
Source: Kottak N et al. Arthritis Care Res. 2018 Feb 5. doi: 10/1002/acr.23527