User login
A Real Welcome Home: Permanent Housing for Homeless Veterans
Mr. G is a 67-year-old veteran. During the Vietnam War, he had the “most dangerous job”: helicopter door gunner and mechanic. He served in multiple combat missions and was under constant threat of attack. On returning to the U.S., he experienced anger outbursts, nightmares, hypervigilance, and urges to engage in dangerous behavior, such as driving a motorcycle more than 100 mph. Then he began abusing alcohol and drugs. Mr. G’s behavior and substance abuse eventually led to strained family relationships, termination from a high-paying job, and homelessness.
In 2001, the Washington DC VAMC homeless and substance abuse staff provided outreach and services that helped him secure permanent subsidized housing, achieve and maintain sobriety, get treatment for posttraumatic stress disorder (PTSD), and get full-time employment. Mr. G has maintained permanent supported housing status for 9 years, regained a sense of purpose, remained drug- and alcohol-free since 2001, attended a Vietnam combat PTSD support group weekly, and been an exemplary employee for 8 years.
In 2009, on any given night in the U.S., more than 75,000 veterans were without a permanent home and living on the streets, as Mr. G had been.1 Nearly 150,000 other veterans were spending the night in an emergency shelter or transitional housing. In early 2010, the U.S. Interagency Council on Homelessness (USICH) developed a strategic plan to align federal resources toward 4 key objectives, which included preventing and ending homelessness among veterans. Since then, the most dramatic reductions in homelessness have occurred among veterans, with an overall 33% decline in chronic homelessness nationwide.
According to the U.S. Department of Housing and Urban Development (HUD), permanent housing is defined as community-based housing without a designated length of stay in which formerly homeless individuals live as independently as possible.2 Under permanent housing, a program participant must be the tenant on a lease for at least 1 year, and the lease is renewable and terminable only for cause. The federal definition of the chronically homeless is a person who is homeless and lives in a place not meant for human habitation, in a safe haven, or in an emergency shelter continuously for at least 1 year; or on at least 4 separate occasions within the past 3 years; and who can be diagnosed with ≥ 1 of the following conditions: substance use disorder, serious mental illness, developmental disability as defined in section 102 of the Developmental Disabilities Assistance Bill of Rights Act of 2000 (42 U.S.C. 15002), PTSD, cognitive impairments caused by brain injury, and chronic physical illness or disability.3
Ending homelessness makes sense from a variety of perspectives. From a moral perspective, no one should experience homelessness, but this is especially true for the men and women who have served in the U.S. military. From a health care and resources perspective, homelessness is associated with poorer medical outcomes; higher medical costs for emergency department visits and hospital admissions; longer stays (often for conditions that could be treated in ambulatory settings); and increased mortality.4-7 From a societal perspective, homelessness is associated with costs for shelters and other forms of temporary housing and with higher justice system costs stemming from police, court, and jail involvement.8 The higher justice system costs are in part attributable to significantly longer incarcerations for homeless persons than for demographically similar inmates who have been similarly charged but have housing.9 According to recent studies, significant cost reductions have been achieved by addressing homelessness and providing permanent housing, particularly for the chronically homeless with mental illness.10-14
This article describes the efforts that have been made, through collaborations and coalitions of government and community partners, to identify and address risk factors for homelessness in Washington, DC—and ultimately to end veteran homelessness in the nation’s capitol.
Historic Perspective on Veteran Homelessness
Although the problem was first described after the Revolutionary War and again after the Civil War, homelessness among U.S. veterans has only recently been academically studied. During the colonial period, men often were promised pensions or land grants in exchange for military service. Several states failed to deliver on these promises, throwing veterans into dire financial circumstances and leaving them homeless. By the end of the Civil War, the size of the veteran population (almost 2 million, counting only those who fought for the Union), combined with an unemployment rate of 40% and economic downturns, led to thousands of veterans becoming homeless.15,16
Homelessness among veterans continued after World War I. In 1932, more than 15,000 homeless and disabled “Bonus Army,” World War I veterans, marched on Washington to demand payment of the financial benefits promised for their military service.
Although World War II and Korean War veterans did not experience homelessness as previous veterans had, the problem resurfaced after Vietnam—the combat veterans of that war were overrepresented among the homeless.17,18 Those at highest risk ranged from age 35 to 44 years in the early years of the all-volunteer military. It has been suggested that their increased risk may reflect social selection—volunteers for military service came from poor economic backgrounds with fewer social supports.19 A more recent study found that 3.8% of more than 300,000 Iraq and Afghanistan veterans who were followed for 5 years after military discharge experienced a homeless episode.20 Female veterans similarly are overrepresented among the homeless.21 Female veterans represent only 1% of the overall female population, yet 3% to 4% are homeless.
Homelessness has always been a social problem, but only during the 1970s and 1980s did homelessness increase in importance—the number of visibly homeless people rose during that period—and investigators began to study and address the issue. Experts have described several factors that contributed to the increase in homelessness during that time.22,23 First, as part of the deinstitutionalization initiative, thousands of mentally ill persons were released from state mental hospitals without a plan in place for affordable or supervised housing. Second, single-room-occupancy dwellings in poor areas, where transient single men lived, were demolished, and affordable housing options decreased. Third, economic and social changes were factors, such as a decreased need for seasonal and unskilled labor; reduced likelihood that relatives will take in homeless family members; and decriminalized public intoxication, loitering, and vagrancy. Out of these conditions came an interest in studying the causes of and risk factors for veteran homelessness and in developing a multipronged approach to end veteran homelessness.
Demographics
Nationally, veterans account for 9% of the homeless population.24 Predominantly, they are single men living in urban areas—only about 9% are women—and 43% served during the Vietnam era.24 Among homeless veterans, minorities are overrepresented—45% are African American or Hispanic, as contrasted with 10% and 3%, respectively, of the general population. More than two-thirds served in the military for more than 3 years, and 76% have a history of mental illness or substance abuse. Compared with the general homeless population, homeless veterans are older, better educated, more likely to have been married, and more likely to have health insurance, primarily through the VA.24,25
The Washington, DC, metropolitan area encompasses the District of Columbia, northern Virginia, the tricounty area of southern Maryland, and Montgomery and Prince George’s counties in Maryland. Demographics for veterans in this area vary somewhat from national figures. According to the 2015 Point-in-Time survey of the homeless, veterans accounted for 5% of the homeless population (less than the national percentage). Most homeless veterans were single men (11.6% were women) and African American (65% of single adults, 85% of adults in families). Forty-five percent reported being employed and 40% had a substance use disorder or a serious mental illness. A large proportion also had at least 1 physical disability, such as hypertension, hepatitis, arthritis, diabetes mellitus, or heart disease.26
Risk Factors
Multiple studies and multivariate analyses have determined that veteran status is associated with an increased risk for homelessness for both male and female veterans.27 Female veterans were 3 times and male veterans 2 times more likely than nonveterans to become homeless, even when poverty, age, race, and geographic variation were controlled. A recent systematic review of U.S. veterans found that the strongest and most consistent risk factors for homelessness were substance use disorders and mental illness, particularly psychotic disorders. Posttraumatic stress disorder was not more significant than other mental conditions but it was a risk factor. Low income, unemployment, and poor money management were also factors.
Social risk factors include lack of support from family and friends. Military service with multiple deployments, transfers, and on-base housing may contribute to interruption of social support and lead to social isolation, thus increasing veterans’ risk for homelessness. Some studies have found that veterans are more likely to report physical injury or medical problems as contributing to homelessness and more likely to have 2 or more chronic medical conditions. Last, history of incarceration and adverse childhood experiences (eg, behavioral problems, family instability, foster care, childhood abuse) also have been found to be risk factors for homelessness among veterans and nonveterans alike.
Understanding these risk factors is an important step in addressing homelessness. Homelessness prevention efforts can screen for these risk factors and then intervene as quickly as possible. Access to mental health and substance abuse services, employment assistance, disability benefits and other income supports, and social services may prevent initial and subsequent episodes of homelessness. The VA, as the largest integrated health system in the U.S., is a critical safety net for low-income and disabled veterans with complex psychosocial needs. One study found access to VA service-connected pensions was protective against homelessness.28
Addressing Homelessness
There are 10 USICH-recommended strategies for ending veteran homelessness (Table 1). These strategies cannot be achieved by any single federal agency or exclusively by government agencies—they require multipronged approaches and private and public partnerships. In early 2011, the staff of the Washington DC VAMC homeless program identified a single point of contact who would regularly meet with each Continuum of Care local planning body and each Public Housing Authority (PHA). This contact could identify homeless veterans regardless of the agency from which they were requesting assistance. The contact also facilitated identification of specific bottleneck issues contributing to delays in housing veterans. One such issue was that veterans were filling out application forms by themselves, and in some cases, their information was incomplete, or supporting documents (eg, government-issued IDs) were missing. The VA team adjusted the procedures to better meet veterans’ needs. Now veterans identified as meeting requirements for housing assistance are enrolled in classes in which a caseworker reviews their completed applications to ensure they are complete and supporting documents included. If an ID is needed, the caseworker facilitates the process and then submits the veteran’s application to the local PHA for processing. The result has been no returned applications.
Another issue was that in some cases a veteran spoke with the PHA and indicated interest in an apartment and only later found out that the apartment failed inspection. There would then be back-and-forth communications between PHA and landlord to have repairs made so the unit could pass inspection, which often resulted in long delays. The solution was to have a stock of preinspected housing options. The Washington DC VAMC homeless program now has a housing specialist who identifies preinspected units and can expedite the lease to a veteran. In addition, the homeless program in partnership with the PHA now sets up regular meet-and-greet events for landlords and veterans so veterans can preview available rentals.
Housing-First Model
The homeless program readily adopted the housing-first model. This model focuses on helping individuals and families access housing as quickly as possible and remain permanently housed; this assistance is not time-limited but ongoing. After housing placement, the client can choose from an array of both time-limited and long-term services, which are individualized to promote housing stability and individual well-being. Most important, housing is not contingent on compliance with services. Instead, participants must comply with a standard lease agreement and are provided the services and support that can help them do so successfully. Services are determined by completing needs assessments. In addition, as a veteran is applying for housing, a caseworker actively uses motivational interviewing techniques to enhance the likelihood that the veteran will accept services.
Providing Core Service to Veterans
In 2012, the Washington DC VAMC opened the Community Resource and Referral Center (CRRC) as a one-stop shop for homeless and at-risk veterans in need of basic, core, wraparound services through the VA. Basic services include showers, laundry facilities, and a chapel. The CRRC has onsite psychiatric services that can engage veterans in mental health and substance abuse treatment. There is also an onsite primary care team who specialize in working with the homeless and can address any acute or chronic medical problems. For veterans who want to return to employment, there is an onsite Compensated Work Therapy program, which has uniquely partnered with the VAMC as well as community partners (eg, National Archives, Smithsonian Institution, Quantico National Cemetery, Arlington National Cemetery) to offer therapeutic job experiences that often lead to gainful full-time employment.
Veterans can use an onsite computer lab to complete resumes and apply for employment online, with the assistance of veteran peer specialists and vocational rehabilitation specialists. For veterans who are unable to work, the homeless program team assists with disability applications. Social Security, Veterans Benefits Administration representatives, and Legal Aid have office hours at the CRRC to provide one-stop shopping for veterans. Additional community partnerships have led to classes to help veterans manage income effectively, and veterans are assisted with completing tax returns.
Representatives from the Veterans Justice Outreach (VJO) and Health Care for Re-entry Veterans (HCRV) programs are also onsite. The VJO program provides outreach, assessment, and case management for justice-involved veterans to prevent unnecessary criminalization of mental illness and extended incarceration. A VJO program specialist works with courts, legal representatives, and jails and acts as a liaison to engage veterans in treatment and prevent incarcerations through jail diversion strategies. The HCRV program assists incarcerated veterans with reentry into the community through outreach and prerelease assessments, and referrals and links to medical, psychiatric, and social services, including employment services on release. This program also can provide short-term case management assistance on release.
In 2013, to further the implementation of the USICH strategies in the Washington, DC, metropolitan area, 12 local government and nonprofit agencies entered into the Veterans NOW coalition (Table 2). This collaboration enabled development of a coordinated effort to identify all area veterans experiencing homelessness, regardless of which agency the veterans contacted. Further, this team set up processes to assess veterans’ housing and service needs and to match each veteran to the most appropriate housing resource. There was consensus regarding use of the Service Prioritization Decision Assistance Tool (SPDAT) as the evidence-informed approach for prioritizing client needs and identifying areas in which support is most likely needed to prevent housing instability.29 More than 50 staff members at 20 different agencies in the District of Columbia have now been trained and are using this tool.
Outcomes
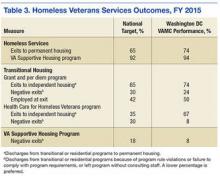
In 2010, the Point-in-Time count identified 718 homeless veterans in the Washington, DC, metropolitan area. By 2016, that number had dropped to 326 (55% reduction). During this same period, the number of veterans served by the Washington DC VAMC homeless program more than tripled, from 2,100 individuals in 2010 to nearly 6,400 in 2015. The coalition has housed or prevented homelessness for nearly 1,300 veterans during the past 2 years alone. Veterans were housed through multiple programs and efforts, including VA Supportive Housing (VASH), Supportive Services for Veteran Families, and Washington, DC Department of Human Services Permanent Supportive Housing. During the past year, > 60% of veterans were successfully placed in VASH housing within 90 days of application submission. Table 3 lists the national targets for assessing performance measurement and success. Not only were the various performance measure benchmarks exceeded, but more important, > 90% of veterans in VASH and Health Care for Homeless Veterans were able to keep their housing stabilized. Using SPDAT, the most chronically homeless and vulnerable were housed first, which accounts for the lower numbers of homeless Washington, DC, area veterans with substance abuse and mental health problems identified in the Point-in-Time survey.
Discussion
At the Washington DC VAMC, HCHV program staff members used evidence-based and evidence-informed tools, collaborated with community partners, and implemented recommended best practices to end veteran homelessness by 2015. Permanent supportive housing through VASH is crucial for helping veterans overcome their lack of income and in providing mechanisms for engaging in mental health and substance abuse services as well as primary care and therapeutic employment opportunities.
When former VA Secretary Eric K. Shinseki first announced the goal of ending veteran homelessness by 2015, many people questioned this goal’s attainability and feasibility. However, through the adoption of the strategies recommended by USICH, the establishment of government and community partnerships (including faith-based groups) and the implementation of programs addressing substance abuse issues, mental and physical health, income limitations, and employment, this goal now seems possible. Overall veteran homelessness decreased by 36% since 2010, and unsheltered homelessness decreased by nearly 50%.30 By the end of 2015, nearly 65,000 veterans are in permanent housing nationwide, and another 8,100 are in the process of obtaining permanent supportive housing. Also, 87% of unsheltered veterans were able to move to safe housing within 30 days of engagement. Last, Supportive Services for Veteran Families was able to assist more than 156,800 individuals (single veterans as well as their children and families).
Sustainability from a national perspective also depends on continued federal funding. Mr. G, described at the beginning of this article, served his country honorably but then experienced the factors that put him at risk for homelessness. Through a veteran-centric team approach, he was able to successfully address each of these factors. As there are another 500 homeless veterans in Washington, DC, much work still needs to be done. With important collaborations and partnerships now in place, the goal of ending veteran homelessness in the District of Columbia is within sight. When homelessness is a thing of the past, we will truly be able to Welcome Home each veteran.
1. U.S. Department of Housing and Urban Development, Office of Community Planning and Development; U.S. Department of Veterans Affairs, National Center on Homelessness Among Veterans Veteran Homelessness: A Supplemental Report to the 2009 Annual Homeless Assessment Report to Congress.https://www.hudexchange.info/resources/documents/2009AHARveterans Report.pdf. Accessed April 19, 2016.
2. HUD Exchange. Homeless emergency assistance and rapid transition to housing (HEARTH): defining “homeless” final rule. HUD Exchange website. https://www.hudexchange.info/resource/1928 /hearth-defining-homeless-final-rule. Published November 2011. Accessed April 5, 2016.
3. U.S. Department of Health and Human Services, Administration for Community Living (ACL), Administration on Intellectual and Developmental Disabilities. The Developmental Disabilities Assistance and Bill of Rights Act of 2000. http://www.acl.gov/Programs/AIDD/DDA_BOR _ACT_2000/docs/dd_act.pdf. Accessed April 5, 2016. 4. Hwang SW. Mortality among men using homeless shelters in Toronto, Ontario. JAMA. 2000;283(16):2152-2157.
5. O’Connell JJ. Premature Mortality in Homeless Populations: A Review of the Literature. Nashville, TN: National Health Care for the Homeless Council; 2005.
6. Salit SA, Kuhn EM, Hartz AJ, Vu JM, Mosso AL. Hopitalization costs associated with homelessness in New York City. N Engl J Med. 1998;338(24):1734-1740.
7. Kushel MB, Perry S, Bangsberg D, Clark R, Moss AR. Emergency department use among the homeless and marginally housed: results from a community-based study. Am J Public Health. 2002;92(5):778-784.
8. Larimer ME, Malone DK, Garner MD, et al. Healthcare and public service use and costs before and after provision of housing for chronically homeless persons with severe alcohol problems. JAMA. 2009;301(13):1349-1357.
9. McNeil DE, Binder RL, Robinson JC. Incarceration associated with homelessness, mental disorder, and co-occurring substance abuse. Psychiatr Serv. 2005;56(7):840-846.
10. Rosenheck R. Cost-effectiveness of services for mentally ill homeless people: the application of research to policy and practice. Am J Psychiatry. 2000;157(10):1563-1570.
11. Culhane DP, Metraux S, Hadley T. Public service reductions associated with placement of homeless people with severe mental illness in supportive housing. Housing Policy Debate. 2002;13(1):107-163.
12. Martinez TE, Burt MR. Impact of permanent supportive housing on the use of acute care health services by homeless adults. Psychiatr Serv. 2006;57(7):992-999.
13. Culhane DP, Parker WD, Poppe B, et al. Accountability, cost-effectiveness, and program performance: progress since 1998. In: Dennis D, Locke G, Khadduri J, eds. Toward Understanding Homelessness: The 2007 National Symposium on Homelessness Research. Washington, DC: Substance Abuse and Mental Health Services Administration, U.S. Dept of Health and Human Services; Office of Policy Development and Research, U.S. Dept of Housing and Urban Development; 2007.
14. Poulin SR, Maguire M, Metraux S, Culhane DP. Service use and costs for persons experiencing chronic homelessness in Philadelphia: a population-based study. Psychiatr Serv. 2010;61(11):1093-1098.
15. U.S. Department of Veterans Affairs. VA History in Brief. http://www.va.gov/opa/publications/archives /docs/history_in_brief.pdf. U.S. Department of Veteran Affairs website. Published December 27, 2013. Accessed April 19, 2016.
16. Kusmer KL. Down and Out, on the Road: The Homeless in American History. New York, NY: Oxford University Press; 2002.
17. Baumohl J, ed. Homelessness in America. Westport, CT: Oryx Press; 1996.
18. Fargo J, Metraux S, Byrne T, et al. Prevalence and risk of homelessness among U.S. veterans: a multisite investigation. http://www.va.gov/HOMELESS/docs/Center/Prevalence_Final.pdf. Published August 2011. Accessed April 19, 2016.
19. Tsai J, Mares AS, Rosenheck R, Gamache D. Do homeless veterans have the same needs and outcomes as non-veterans? Mil Med. 2012;177(1):27-31.
20. Metraux S, Clegg L, Daigh JD, Culhane DP, Kane V. Risk factors for becoming homeless among a cohort of veterans who served in the era of the Iraq and Afghanistan conflicts. Am J Public Health. 2013;103(suppl 2):S255-S261.
21. Perl L. Veterans and homelessness. Congressional research Service Report RL34024. https://www.fas .org/sgp/crs/misc/RL34024.pdf.Published November 6, 2015. Accessed April 7, 2016.
22. Rossi PH. Down and Out in America: The Origins of Homelessness. Chicago, IL: University of Chicago Press; 1989.
23. Burt M. Over the Edge: The Growth of Homelessness in the 1980s. New York, NY: Russell Sage Foundation; 1992.
24. National Coalition for the Homeless. Homeless Veterans [fact sheet]. http://www .nationalhomeless.org/factsheets/veterans.html. September 2009. Accessed April 19, 2016.
25. Tessler R, Rosenheck R, Gamache F. Comparison of homeless veterans with other homeless men in a large clinical outreach program. Psychiatr Q. 2002;73(2):109-119.
26. Chapman H. Homelessness in Metropolitan Washington: Results and Analysis From the 2015 Point-in-Time Count of Persons Experiencing Homelessness in the Metropolitan Washington Region. Metropolitan Washington Council of Governments website. https://www .mwcog.org/uploads/pub-documents/v15Wlk20150514094353.pdf. Published May 13, 2015. Accessed April 6, 2016.
27. Tsai J, Rosenheck R. Risk factors for homelessness among US veterans. Epidemiol Rev. 2015;37:177-195.
28. Edens EL, Kasprow W, Tsai J, Rosenheck R. Association of substance use and VA service-connected disability benefits with risk of homelessness among veterans. Am J Addict. 2011;20(5):412-419.
29. OrgCode Consulting. Service Prioritization Decision Assistance Tool (SPDAT). http://www.orgcode .com/product/spdat/. Accessed April 19, 2016.
30. Performance.gov. End veteran homelessness: progress update. Performance.gov website. https://www.performance.gov/content/end-veteran-homelessness#progressUpdate. Accessed April 6, 2016.
Mr. G is a 67-year-old veteran. During the Vietnam War, he had the “most dangerous job”: helicopter door gunner and mechanic. He served in multiple combat missions and was under constant threat of attack. On returning to the U.S., he experienced anger outbursts, nightmares, hypervigilance, and urges to engage in dangerous behavior, such as driving a motorcycle more than 100 mph. Then he began abusing alcohol and drugs. Mr. G’s behavior and substance abuse eventually led to strained family relationships, termination from a high-paying job, and homelessness.
In 2001, the Washington DC VAMC homeless and substance abuse staff provided outreach and services that helped him secure permanent subsidized housing, achieve and maintain sobriety, get treatment for posttraumatic stress disorder (PTSD), and get full-time employment. Mr. G has maintained permanent supported housing status for 9 years, regained a sense of purpose, remained drug- and alcohol-free since 2001, attended a Vietnam combat PTSD support group weekly, and been an exemplary employee for 8 years.
In 2009, on any given night in the U.S., more than 75,000 veterans were without a permanent home and living on the streets, as Mr. G had been.1 Nearly 150,000 other veterans were spending the night in an emergency shelter or transitional housing. In early 2010, the U.S. Interagency Council on Homelessness (USICH) developed a strategic plan to align federal resources toward 4 key objectives, which included preventing and ending homelessness among veterans. Since then, the most dramatic reductions in homelessness have occurred among veterans, with an overall 33% decline in chronic homelessness nationwide.
According to the U.S. Department of Housing and Urban Development (HUD), permanent housing is defined as community-based housing without a designated length of stay in which formerly homeless individuals live as independently as possible.2 Under permanent housing, a program participant must be the tenant on a lease for at least 1 year, and the lease is renewable and terminable only for cause. The federal definition of the chronically homeless is a person who is homeless and lives in a place not meant for human habitation, in a safe haven, or in an emergency shelter continuously for at least 1 year; or on at least 4 separate occasions within the past 3 years; and who can be diagnosed with ≥ 1 of the following conditions: substance use disorder, serious mental illness, developmental disability as defined in section 102 of the Developmental Disabilities Assistance Bill of Rights Act of 2000 (42 U.S.C. 15002), PTSD, cognitive impairments caused by brain injury, and chronic physical illness or disability.3
Ending homelessness makes sense from a variety of perspectives. From a moral perspective, no one should experience homelessness, but this is especially true for the men and women who have served in the U.S. military. From a health care and resources perspective, homelessness is associated with poorer medical outcomes; higher medical costs for emergency department visits and hospital admissions; longer stays (often for conditions that could be treated in ambulatory settings); and increased mortality.4-7 From a societal perspective, homelessness is associated with costs for shelters and other forms of temporary housing and with higher justice system costs stemming from police, court, and jail involvement.8 The higher justice system costs are in part attributable to significantly longer incarcerations for homeless persons than for demographically similar inmates who have been similarly charged but have housing.9 According to recent studies, significant cost reductions have been achieved by addressing homelessness and providing permanent housing, particularly for the chronically homeless with mental illness.10-14
This article describes the efforts that have been made, through collaborations and coalitions of government and community partners, to identify and address risk factors for homelessness in Washington, DC—and ultimately to end veteran homelessness in the nation’s capitol.
Historic Perspective on Veteran Homelessness
Although the problem was first described after the Revolutionary War and again after the Civil War, homelessness among U.S. veterans has only recently been academically studied. During the colonial period, men often were promised pensions or land grants in exchange for military service. Several states failed to deliver on these promises, throwing veterans into dire financial circumstances and leaving them homeless. By the end of the Civil War, the size of the veteran population (almost 2 million, counting only those who fought for the Union), combined with an unemployment rate of 40% and economic downturns, led to thousands of veterans becoming homeless.15,16
Homelessness among veterans continued after World War I. In 1932, more than 15,000 homeless and disabled “Bonus Army,” World War I veterans, marched on Washington to demand payment of the financial benefits promised for their military service.
Although World War II and Korean War veterans did not experience homelessness as previous veterans had, the problem resurfaced after Vietnam—the combat veterans of that war were overrepresented among the homeless.17,18 Those at highest risk ranged from age 35 to 44 years in the early years of the all-volunteer military. It has been suggested that their increased risk may reflect social selection—volunteers for military service came from poor economic backgrounds with fewer social supports.19 A more recent study found that 3.8% of more than 300,000 Iraq and Afghanistan veterans who were followed for 5 years after military discharge experienced a homeless episode.20 Female veterans similarly are overrepresented among the homeless.21 Female veterans represent only 1% of the overall female population, yet 3% to 4% are homeless.
Homelessness has always been a social problem, but only during the 1970s and 1980s did homelessness increase in importance—the number of visibly homeless people rose during that period—and investigators began to study and address the issue. Experts have described several factors that contributed to the increase in homelessness during that time.22,23 First, as part of the deinstitutionalization initiative, thousands of mentally ill persons were released from state mental hospitals without a plan in place for affordable or supervised housing. Second, single-room-occupancy dwellings in poor areas, where transient single men lived, were demolished, and affordable housing options decreased. Third, economic and social changes were factors, such as a decreased need for seasonal and unskilled labor; reduced likelihood that relatives will take in homeless family members; and decriminalized public intoxication, loitering, and vagrancy. Out of these conditions came an interest in studying the causes of and risk factors for veteran homelessness and in developing a multipronged approach to end veteran homelessness.
Demographics
Nationally, veterans account for 9% of the homeless population.24 Predominantly, they are single men living in urban areas—only about 9% are women—and 43% served during the Vietnam era.24 Among homeless veterans, minorities are overrepresented—45% are African American or Hispanic, as contrasted with 10% and 3%, respectively, of the general population. More than two-thirds served in the military for more than 3 years, and 76% have a history of mental illness or substance abuse. Compared with the general homeless population, homeless veterans are older, better educated, more likely to have been married, and more likely to have health insurance, primarily through the VA.24,25
The Washington, DC, metropolitan area encompasses the District of Columbia, northern Virginia, the tricounty area of southern Maryland, and Montgomery and Prince George’s counties in Maryland. Demographics for veterans in this area vary somewhat from national figures. According to the 2015 Point-in-Time survey of the homeless, veterans accounted for 5% of the homeless population (less than the national percentage). Most homeless veterans were single men (11.6% were women) and African American (65% of single adults, 85% of adults in families). Forty-five percent reported being employed and 40% had a substance use disorder or a serious mental illness. A large proportion also had at least 1 physical disability, such as hypertension, hepatitis, arthritis, diabetes mellitus, or heart disease.26
Risk Factors
Multiple studies and multivariate analyses have determined that veteran status is associated with an increased risk for homelessness for both male and female veterans.27 Female veterans were 3 times and male veterans 2 times more likely than nonveterans to become homeless, even when poverty, age, race, and geographic variation were controlled. A recent systematic review of U.S. veterans found that the strongest and most consistent risk factors for homelessness were substance use disorders and mental illness, particularly psychotic disorders. Posttraumatic stress disorder was not more significant than other mental conditions but it was a risk factor. Low income, unemployment, and poor money management were also factors.
Social risk factors include lack of support from family and friends. Military service with multiple deployments, transfers, and on-base housing may contribute to interruption of social support and lead to social isolation, thus increasing veterans’ risk for homelessness. Some studies have found that veterans are more likely to report physical injury or medical problems as contributing to homelessness and more likely to have 2 or more chronic medical conditions. Last, history of incarceration and adverse childhood experiences (eg, behavioral problems, family instability, foster care, childhood abuse) also have been found to be risk factors for homelessness among veterans and nonveterans alike.
Understanding these risk factors is an important step in addressing homelessness. Homelessness prevention efforts can screen for these risk factors and then intervene as quickly as possible. Access to mental health and substance abuse services, employment assistance, disability benefits and other income supports, and social services may prevent initial and subsequent episodes of homelessness. The VA, as the largest integrated health system in the U.S., is a critical safety net for low-income and disabled veterans with complex psychosocial needs. One study found access to VA service-connected pensions was protective against homelessness.28
Addressing Homelessness
There are 10 USICH-recommended strategies for ending veteran homelessness (Table 1). These strategies cannot be achieved by any single federal agency or exclusively by government agencies—they require multipronged approaches and private and public partnerships. In early 2011, the staff of the Washington DC VAMC homeless program identified a single point of contact who would regularly meet with each Continuum of Care local planning body and each Public Housing Authority (PHA). This contact could identify homeless veterans regardless of the agency from which they were requesting assistance. The contact also facilitated identification of specific bottleneck issues contributing to delays in housing veterans. One such issue was that veterans were filling out application forms by themselves, and in some cases, their information was incomplete, or supporting documents (eg, government-issued IDs) were missing. The VA team adjusted the procedures to better meet veterans’ needs. Now veterans identified as meeting requirements for housing assistance are enrolled in classes in which a caseworker reviews their completed applications to ensure they are complete and supporting documents included. If an ID is needed, the caseworker facilitates the process and then submits the veteran’s application to the local PHA for processing. The result has been no returned applications.
Another issue was that in some cases a veteran spoke with the PHA and indicated interest in an apartment and only later found out that the apartment failed inspection. There would then be back-and-forth communications between PHA and landlord to have repairs made so the unit could pass inspection, which often resulted in long delays. The solution was to have a stock of preinspected housing options. The Washington DC VAMC homeless program now has a housing specialist who identifies preinspected units and can expedite the lease to a veteran. In addition, the homeless program in partnership with the PHA now sets up regular meet-and-greet events for landlords and veterans so veterans can preview available rentals.
Housing-First Model
The homeless program readily adopted the housing-first model. This model focuses on helping individuals and families access housing as quickly as possible and remain permanently housed; this assistance is not time-limited but ongoing. After housing placement, the client can choose from an array of both time-limited and long-term services, which are individualized to promote housing stability and individual well-being. Most important, housing is not contingent on compliance with services. Instead, participants must comply with a standard lease agreement and are provided the services and support that can help them do so successfully. Services are determined by completing needs assessments. In addition, as a veteran is applying for housing, a caseworker actively uses motivational interviewing techniques to enhance the likelihood that the veteran will accept services.
Providing Core Service to Veterans
In 2012, the Washington DC VAMC opened the Community Resource and Referral Center (CRRC) as a one-stop shop for homeless and at-risk veterans in need of basic, core, wraparound services through the VA. Basic services include showers, laundry facilities, and a chapel. The CRRC has onsite psychiatric services that can engage veterans in mental health and substance abuse treatment. There is also an onsite primary care team who specialize in working with the homeless and can address any acute or chronic medical problems. For veterans who want to return to employment, there is an onsite Compensated Work Therapy program, which has uniquely partnered with the VAMC as well as community partners (eg, National Archives, Smithsonian Institution, Quantico National Cemetery, Arlington National Cemetery) to offer therapeutic job experiences that often lead to gainful full-time employment.
Veterans can use an onsite computer lab to complete resumes and apply for employment online, with the assistance of veteran peer specialists and vocational rehabilitation specialists. For veterans who are unable to work, the homeless program team assists with disability applications. Social Security, Veterans Benefits Administration representatives, and Legal Aid have office hours at the CRRC to provide one-stop shopping for veterans. Additional community partnerships have led to classes to help veterans manage income effectively, and veterans are assisted with completing tax returns.
Representatives from the Veterans Justice Outreach (VJO) and Health Care for Re-entry Veterans (HCRV) programs are also onsite. The VJO program provides outreach, assessment, and case management for justice-involved veterans to prevent unnecessary criminalization of mental illness and extended incarceration. A VJO program specialist works with courts, legal representatives, and jails and acts as a liaison to engage veterans in treatment and prevent incarcerations through jail diversion strategies. The HCRV program assists incarcerated veterans with reentry into the community through outreach and prerelease assessments, and referrals and links to medical, psychiatric, and social services, including employment services on release. This program also can provide short-term case management assistance on release.
In 2013, to further the implementation of the USICH strategies in the Washington, DC, metropolitan area, 12 local government and nonprofit agencies entered into the Veterans NOW coalition (Table 2). This collaboration enabled development of a coordinated effort to identify all area veterans experiencing homelessness, regardless of which agency the veterans contacted. Further, this team set up processes to assess veterans’ housing and service needs and to match each veteran to the most appropriate housing resource. There was consensus regarding use of the Service Prioritization Decision Assistance Tool (SPDAT) as the evidence-informed approach for prioritizing client needs and identifying areas in which support is most likely needed to prevent housing instability.29 More than 50 staff members at 20 different agencies in the District of Columbia have now been trained and are using this tool.
Outcomes
In 2010, the Point-in-Time count identified 718 homeless veterans in the Washington, DC, metropolitan area. By 2016, that number had dropped to 326 (55% reduction). During this same period, the number of veterans served by the Washington DC VAMC homeless program more than tripled, from 2,100 individuals in 2010 to nearly 6,400 in 2015. The coalition has housed or prevented homelessness for nearly 1,300 veterans during the past 2 years alone. Veterans were housed through multiple programs and efforts, including VA Supportive Housing (VASH), Supportive Services for Veteran Families, and Washington, DC Department of Human Services Permanent Supportive Housing. During the past year, > 60% of veterans were successfully placed in VASH housing within 90 days of application submission. Table 3 lists the national targets for assessing performance measurement and success. Not only were the various performance measure benchmarks exceeded, but more important, > 90% of veterans in VASH and Health Care for Homeless Veterans were able to keep their housing stabilized. Using SPDAT, the most chronically homeless and vulnerable were housed first, which accounts for the lower numbers of homeless Washington, DC, area veterans with substance abuse and mental health problems identified in the Point-in-Time survey.
Discussion
At the Washington DC VAMC, HCHV program staff members used evidence-based and evidence-informed tools, collaborated with community partners, and implemented recommended best practices to end veteran homelessness by 2015. Permanent supportive housing through VASH is crucial for helping veterans overcome their lack of income and in providing mechanisms for engaging in mental health and substance abuse services as well as primary care and therapeutic employment opportunities.
When former VA Secretary Eric K. Shinseki first announced the goal of ending veteran homelessness by 2015, many people questioned this goal’s attainability and feasibility. However, through the adoption of the strategies recommended by USICH, the establishment of government and community partnerships (including faith-based groups) and the implementation of programs addressing substance abuse issues, mental and physical health, income limitations, and employment, this goal now seems possible. Overall veteran homelessness decreased by 36% since 2010, and unsheltered homelessness decreased by nearly 50%.30 By the end of 2015, nearly 65,000 veterans are in permanent housing nationwide, and another 8,100 are in the process of obtaining permanent supportive housing. Also, 87% of unsheltered veterans were able to move to safe housing within 30 days of engagement. Last, Supportive Services for Veteran Families was able to assist more than 156,800 individuals (single veterans as well as their children and families).
Sustainability from a national perspective also depends on continued federal funding. Mr. G, described at the beginning of this article, served his country honorably but then experienced the factors that put him at risk for homelessness. Through a veteran-centric team approach, he was able to successfully address each of these factors. As there are another 500 homeless veterans in Washington, DC, much work still needs to be done. With important collaborations and partnerships now in place, the goal of ending veteran homelessness in the District of Columbia is within sight. When homelessness is a thing of the past, we will truly be able to Welcome Home each veteran.
Mr. G is a 67-year-old veteran. During the Vietnam War, he had the “most dangerous job”: helicopter door gunner and mechanic. He served in multiple combat missions and was under constant threat of attack. On returning to the U.S., he experienced anger outbursts, nightmares, hypervigilance, and urges to engage in dangerous behavior, such as driving a motorcycle more than 100 mph. Then he began abusing alcohol and drugs. Mr. G’s behavior and substance abuse eventually led to strained family relationships, termination from a high-paying job, and homelessness.
In 2001, the Washington DC VAMC homeless and substance abuse staff provided outreach and services that helped him secure permanent subsidized housing, achieve and maintain sobriety, get treatment for posttraumatic stress disorder (PTSD), and get full-time employment. Mr. G has maintained permanent supported housing status for 9 years, regained a sense of purpose, remained drug- and alcohol-free since 2001, attended a Vietnam combat PTSD support group weekly, and been an exemplary employee for 8 years.
In 2009, on any given night in the U.S., more than 75,000 veterans were without a permanent home and living on the streets, as Mr. G had been.1 Nearly 150,000 other veterans were spending the night in an emergency shelter or transitional housing. In early 2010, the U.S. Interagency Council on Homelessness (USICH) developed a strategic plan to align federal resources toward 4 key objectives, which included preventing and ending homelessness among veterans. Since then, the most dramatic reductions in homelessness have occurred among veterans, with an overall 33% decline in chronic homelessness nationwide.
According to the U.S. Department of Housing and Urban Development (HUD), permanent housing is defined as community-based housing without a designated length of stay in which formerly homeless individuals live as independently as possible.2 Under permanent housing, a program participant must be the tenant on a lease for at least 1 year, and the lease is renewable and terminable only for cause. The federal definition of the chronically homeless is a person who is homeless and lives in a place not meant for human habitation, in a safe haven, or in an emergency shelter continuously for at least 1 year; or on at least 4 separate occasions within the past 3 years; and who can be diagnosed with ≥ 1 of the following conditions: substance use disorder, serious mental illness, developmental disability as defined in section 102 of the Developmental Disabilities Assistance Bill of Rights Act of 2000 (42 U.S.C. 15002), PTSD, cognitive impairments caused by brain injury, and chronic physical illness or disability.3
Ending homelessness makes sense from a variety of perspectives. From a moral perspective, no one should experience homelessness, but this is especially true for the men and women who have served in the U.S. military. From a health care and resources perspective, homelessness is associated with poorer medical outcomes; higher medical costs for emergency department visits and hospital admissions; longer stays (often for conditions that could be treated in ambulatory settings); and increased mortality.4-7 From a societal perspective, homelessness is associated with costs for shelters and other forms of temporary housing and with higher justice system costs stemming from police, court, and jail involvement.8 The higher justice system costs are in part attributable to significantly longer incarcerations for homeless persons than for demographically similar inmates who have been similarly charged but have housing.9 According to recent studies, significant cost reductions have been achieved by addressing homelessness and providing permanent housing, particularly for the chronically homeless with mental illness.10-14
This article describes the efforts that have been made, through collaborations and coalitions of government and community partners, to identify and address risk factors for homelessness in Washington, DC—and ultimately to end veteran homelessness in the nation’s capitol.
Historic Perspective on Veteran Homelessness
Although the problem was first described after the Revolutionary War and again after the Civil War, homelessness among U.S. veterans has only recently been academically studied. During the colonial period, men often were promised pensions or land grants in exchange for military service. Several states failed to deliver on these promises, throwing veterans into dire financial circumstances and leaving them homeless. By the end of the Civil War, the size of the veteran population (almost 2 million, counting only those who fought for the Union), combined with an unemployment rate of 40% and economic downturns, led to thousands of veterans becoming homeless.15,16
Homelessness among veterans continued after World War I. In 1932, more than 15,000 homeless and disabled “Bonus Army,” World War I veterans, marched on Washington to demand payment of the financial benefits promised for their military service.
Although World War II and Korean War veterans did not experience homelessness as previous veterans had, the problem resurfaced after Vietnam—the combat veterans of that war were overrepresented among the homeless.17,18 Those at highest risk ranged from age 35 to 44 years in the early years of the all-volunteer military. It has been suggested that their increased risk may reflect social selection—volunteers for military service came from poor economic backgrounds with fewer social supports.19 A more recent study found that 3.8% of more than 300,000 Iraq and Afghanistan veterans who were followed for 5 years after military discharge experienced a homeless episode.20 Female veterans similarly are overrepresented among the homeless.21 Female veterans represent only 1% of the overall female population, yet 3% to 4% are homeless.
Homelessness has always been a social problem, but only during the 1970s and 1980s did homelessness increase in importance—the number of visibly homeless people rose during that period—and investigators began to study and address the issue. Experts have described several factors that contributed to the increase in homelessness during that time.22,23 First, as part of the deinstitutionalization initiative, thousands of mentally ill persons were released from state mental hospitals without a plan in place for affordable or supervised housing. Second, single-room-occupancy dwellings in poor areas, where transient single men lived, were demolished, and affordable housing options decreased. Third, economic and social changes were factors, such as a decreased need for seasonal and unskilled labor; reduced likelihood that relatives will take in homeless family members; and decriminalized public intoxication, loitering, and vagrancy. Out of these conditions came an interest in studying the causes of and risk factors for veteran homelessness and in developing a multipronged approach to end veteran homelessness.
Demographics
Nationally, veterans account for 9% of the homeless population.24 Predominantly, they are single men living in urban areas—only about 9% are women—and 43% served during the Vietnam era.24 Among homeless veterans, minorities are overrepresented—45% are African American or Hispanic, as contrasted with 10% and 3%, respectively, of the general population. More than two-thirds served in the military for more than 3 years, and 76% have a history of mental illness or substance abuse. Compared with the general homeless population, homeless veterans are older, better educated, more likely to have been married, and more likely to have health insurance, primarily through the VA.24,25
The Washington, DC, metropolitan area encompasses the District of Columbia, northern Virginia, the tricounty area of southern Maryland, and Montgomery and Prince George’s counties in Maryland. Demographics for veterans in this area vary somewhat from national figures. According to the 2015 Point-in-Time survey of the homeless, veterans accounted for 5% of the homeless population (less than the national percentage). Most homeless veterans were single men (11.6% were women) and African American (65% of single adults, 85% of adults in families). Forty-five percent reported being employed and 40% had a substance use disorder or a serious mental illness. A large proportion also had at least 1 physical disability, such as hypertension, hepatitis, arthritis, diabetes mellitus, or heart disease.26
Risk Factors
Multiple studies and multivariate analyses have determined that veteran status is associated with an increased risk for homelessness for both male and female veterans.27 Female veterans were 3 times and male veterans 2 times more likely than nonveterans to become homeless, even when poverty, age, race, and geographic variation were controlled. A recent systematic review of U.S. veterans found that the strongest and most consistent risk factors for homelessness were substance use disorders and mental illness, particularly psychotic disorders. Posttraumatic stress disorder was not more significant than other mental conditions but it was a risk factor. Low income, unemployment, and poor money management were also factors.
Social risk factors include lack of support from family and friends. Military service with multiple deployments, transfers, and on-base housing may contribute to interruption of social support and lead to social isolation, thus increasing veterans’ risk for homelessness. Some studies have found that veterans are more likely to report physical injury or medical problems as contributing to homelessness and more likely to have 2 or more chronic medical conditions. Last, history of incarceration and adverse childhood experiences (eg, behavioral problems, family instability, foster care, childhood abuse) also have been found to be risk factors for homelessness among veterans and nonveterans alike.
Understanding these risk factors is an important step in addressing homelessness. Homelessness prevention efforts can screen for these risk factors and then intervene as quickly as possible. Access to mental health and substance abuse services, employment assistance, disability benefits and other income supports, and social services may prevent initial and subsequent episodes of homelessness. The VA, as the largest integrated health system in the U.S., is a critical safety net for low-income and disabled veterans with complex psychosocial needs. One study found access to VA service-connected pensions was protective against homelessness.28
Addressing Homelessness
There are 10 USICH-recommended strategies for ending veteran homelessness (Table 1). These strategies cannot be achieved by any single federal agency or exclusively by government agencies—they require multipronged approaches and private and public partnerships. In early 2011, the staff of the Washington DC VAMC homeless program identified a single point of contact who would regularly meet with each Continuum of Care local planning body and each Public Housing Authority (PHA). This contact could identify homeless veterans regardless of the agency from which they were requesting assistance. The contact also facilitated identification of specific bottleneck issues contributing to delays in housing veterans. One such issue was that veterans were filling out application forms by themselves, and in some cases, their information was incomplete, or supporting documents (eg, government-issued IDs) were missing. The VA team adjusted the procedures to better meet veterans’ needs. Now veterans identified as meeting requirements for housing assistance are enrolled in classes in which a caseworker reviews their completed applications to ensure they are complete and supporting documents included. If an ID is needed, the caseworker facilitates the process and then submits the veteran’s application to the local PHA for processing. The result has been no returned applications.
Another issue was that in some cases a veteran spoke with the PHA and indicated interest in an apartment and only later found out that the apartment failed inspection. There would then be back-and-forth communications between PHA and landlord to have repairs made so the unit could pass inspection, which often resulted in long delays. The solution was to have a stock of preinspected housing options. The Washington DC VAMC homeless program now has a housing specialist who identifies preinspected units and can expedite the lease to a veteran. In addition, the homeless program in partnership with the PHA now sets up regular meet-and-greet events for landlords and veterans so veterans can preview available rentals.
Housing-First Model
The homeless program readily adopted the housing-first model. This model focuses on helping individuals and families access housing as quickly as possible and remain permanently housed; this assistance is not time-limited but ongoing. After housing placement, the client can choose from an array of both time-limited and long-term services, which are individualized to promote housing stability and individual well-being. Most important, housing is not contingent on compliance with services. Instead, participants must comply with a standard lease agreement and are provided the services and support that can help them do so successfully. Services are determined by completing needs assessments. In addition, as a veteran is applying for housing, a caseworker actively uses motivational interviewing techniques to enhance the likelihood that the veteran will accept services.
Providing Core Service to Veterans
In 2012, the Washington DC VAMC opened the Community Resource and Referral Center (CRRC) as a one-stop shop for homeless and at-risk veterans in need of basic, core, wraparound services through the VA. Basic services include showers, laundry facilities, and a chapel. The CRRC has onsite psychiatric services that can engage veterans in mental health and substance abuse treatment. There is also an onsite primary care team who specialize in working with the homeless and can address any acute or chronic medical problems. For veterans who want to return to employment, there is an onsite Compensated Work Therapy program, which has uniquely partnered with the VAMC as well as community partners (eg, National Archives, Smithsonian Institution, Quantico National Cemetery, Arlington National Cemetery) to offer therapeutic job experiences that often lead to gainful full-time employment.
Veterans can use an onsite computer lab to complete resumes and apply for employment online, with the assistance of veteran peer specialists and vocational rehabilitation specialists. For veterans who are unable to work, the homeless program team assists with disability applications. Social Security, Veterans Benefits Administration representatives, and Legal Aid have office hours at the CRRC to provide one-stop shopping for veterans. Additional community partnerships have led to classes to help veterans manage income effectively, and veterans are assisted with completing tax returns.
Representatives from the Veterans Justice Outreach (VJO) and Health Care for Re-entry Veterans (HCRV) programs are also onsite. The VJO program provides outreach, assessment, and case management for justice-involved veterans to prevent unnecessary criminalization of mental illness and extended incarceration. A VJO program specialist works with courts, legal representatives, and jails and acts as a liaison to engage veterans in treatment and prevent incarcerations through jail diversion strategies. The HCRV program assists incarcerated veterans with reentry into the community through outreach and prerelease assessments, and referrals and links to medical, psychiatric, and social services, including employment services on release. This program also can provide short-term case management assistance on release.
In 2013, to further the implementation of the USICH strategies in the Washington, DC, metropolitan area, 12 local government and nonprofit agencies entered into the Veterans NOW coalition (Table 2). This collaboration enabled development of a coordinated effort to identify all area veterans experiencing homelessness, regardless of which agency the veterans contacted. Further, this team set up processes to assess veterans’ housing and service needs and to match each veteran to the most appropriate housing resource. There was consensus regarding use of the Service Prioritization Decision Assistance Tool (SPDAT) as the evidence-informed approach for prioritizing client needs and identifying areas in which support is most likely needed to prevent housing instability.29 More than 50 staff members at 20 different agencies in the District of Columbia have now been trained and are using this tool.
Outcomes
In 2010, the Point-in-Time count identified 718 homeless veterans in the Washington, DC, metropolitan area. By 2016, that number had dropped to 326 (55% reduction). During this same period, the number of veterans served by the Washington DC VAMC homeless program more than tripled, from 2,100 individuals in 2010 to nearly 6,400 in 2015. The coalition has housed or prevented homelessness for nearly 1,300 veterans during the past 2 years alone. Veterans were housed through multiple programs and efforts, including VA Supportive Housing (VASH), Supportive Services for Veteran Families, and Washington, DC Department of Human Services Permanent Supportive Housing. During the past year, > 60% of veterans were successfully placed in VASH housing within 90 days of application submission. Table 3 lists the national targets for assessing performance measurement and success. Not only were the various performance measure benchmarks exceeded, but more important, > 90% of veterans in VASH and Health Care for Homeless Veterans were able to keep their housing stabilized. Using SPDAT, the most chronically homeless and vulnerable were housed first, which accounts for the lower numbers of homeless Washington, DC, area veterans with substance abuse and mental health problems identified in the Point-in-Time survey.
Discussion
At the Washington DC VAMC, HCHV program staff members used evidence-based and evidence-informed tools, collaborated with community partners, and implemented recommended best practices to end veteran homelessness by 2015. Permanent supportive housing through VASH is crucial for helping veterans overcome their lack of income and in providing mechanisms for engaging in mental health and substance abuse services as well as primary care and therapeutic employment opportunities.
When former VA Secretary Eric K. Shinseki first announced the goal of ending veteran homelessness by 2015, many people questioned this goal’s attainability and feasibility. However, through the adoption of the strategies recommended by USICH, the establishment of government and community partnerships (including faith-based groups) and the implementation of programs addressing substance abuse issues, mental and physical health, income limitations, and employment, this goal now seems possible. Overall veteran homelessness decreased by 36% since 2010, and unsheltered homelessness decreased by nearly 50%.30 By the end of 2015, nearly 65,000 veterans are in permanent housing nationwide, and another 8,100 are in the process of obtaining permanent supportive housing. Also, 87% of unsheltered veterans were able to move to safe housing within 30 days of engagement. Last, Supportive Services for Veteran Families was able to assist more than 156,800 individuals (single veterans as well as their children and families).
Sustainability from a national perspective also depends on continued federal funding. Mr. G, described at the beginning of this article, served his country honorably but then experienced the factors that put him at risk for homelessness. Through a veteran-centric team approach, he was able to successfully address each of these factors. As there are another 500 homeless veterans in Washington, DC, much work still needs to be done. With important collaborations and partnerships now in place, the goal of ending veteran homelessness in the District of Columbia is within sight. When homelessness is a thing of the past, we will truly be able to Welcome Home each veteran.
1. U.S. Department of Housing and Urban Development, Office of Community Planning and Development; U.S. Department of Veterans Affairs, National Center on Homelessness Among Veterans Veteran Homelessness: A Supplemental Report to the 2009 Annual Homeless Assessment Report to Congress.https://www.hudexchange.info/resources/documents/2009AHARveterans Report.pdf. Accessed April 19, 2016.
2. HUD Exchange. Homeless emergency assistance and rapid transition to housing (HEARTH): defining “homeless” final rule. HUD Exchange website. https://www.hudexchange.info/resource/1928 /hearth-defining-homeless-final-rule. Published November 2011. Accessed April 5, 2016.
3. U.S. Department of Health and Human Services, Administration for Community Living (ACL), Administration on Intellectual and Developmental Disabilities. The Developmental Disabilities Assistance and Bill of Rights Act of 2000. http://www.acl.gov/Programs/AIDD/DDA_BOR _ACT_2000/docs/dd_act.pdf. Accessed April 5, 2016. 4. Hwang SW. Mortality among men using homeless shelters in Toronto, Ontario. JAMA. 2000;283(16):2152-2157.
5. O’Connell JJ. Premature Mortality in Homeless Populations: A Review of the Literature. Nashville, TN: National Health Care for the Homeless Council; 2005.
6. Salit SA, Kuhn EM, Hartz AJ, Vu JM, Mosso AL. Hopitalization costs associated with homelessness in New York City. N Engl J Med. 1998;338(24):1734-1740.
7. Kushel MB, Perry S, Bangsberg D, Clark R, Moss AR. Emergency department use among the homeless and marginally housed: results from a community-based study. Am J Public Health. 2002;92(5):778-784.
8. Larimer ME, Malone DK, Garner MD, et al. Healthcare and public service use and costs before and after provision of housing for chronically homeless persons with severe alcohol problems. JAMA. 2009;301(13):1349-1357.
9. McNeil DE, Binder RL, Robinson JC. Incarceration associated with homelessness, mental disorder, and co-occurring substance abuse. Psychiatr Serv. 2005;56(7):840-846.
10. Rosenheck R. Cost-effectiveness of services for mentally ill homeless people: the application of research to policy and practice. Am J Psychiatry. 2000;157(10):1563-1570.
11. Culhane DP, Metraux S, Hadley T. Public service reductions associated with placement of homeless people with severe mental illness in supportive housing. Housing Policy Debate. 2002;13(1):107-163.
12. Martinez TE, Burt MR. Impact of permanent supportive housing on the use of acute care health services by homeless adults. Psychiatr Serv. 2006;57(7):992-999.
13. Culhane DP, Parker WD, Poppe B, et al. Accountability, cost-effectiveness, and program performance: progress since 1998. In: Dennis D, Locke G, Khadduri J, eds. Toward Understanding Homelessness: The 2007 National Symposium on Homelessness Research. Washington, DC: Substance Abuse and Mental Health Services Administration, U.S. Dept of Health and Human Services; Office of Policy Development and Research, U.S. Dept of Housing and Urban Development; 2007.
14. Poulin SR, Maguire M, Metraux S, Culhane DP. Service use and costs for persons experiencing chronic homelessness in Philadelphia: a population-based study. Psychiatr Serv. 2010;61(11):1093-1098.
15. U.S. Department of Veterans Affairs. VA History in Brief. http://www.va.gov/opa/publications/archives /docs/history_in_brief.pdf. U.S. Department of Veteran Affairs website. Published December 27, 2013. Accessed April 19, 2016.
16. Kusmer KL. Down and Out, on the Road: The Homeless in American History. New York, NY: Oxford University Press; 2002.
17. Baumohl J, ed. Homelessness in America. Westport, CT: Oryx Press; 1996.
18. Fargo J, Metraux S, Byrne T, et al. Prevalence and risk of homelessness among U.S. veterans: a multisite investigation. http://www.va.gov/HOMELESS/docs/Center/Prevalence_Final.pdf. Published August 2011. Accessed April 19, 2016.
19. Tsai J, Mares AS, Rosenheck R, Gamache D. Do homeless veterans have the same needs and outcomes as non-veterans? Mil Med. 2012;177(1):27-31.
20. Metraux S, Clegg L, Daigh JD, Culhane DP, Kane V. Risk factors for becoming homeless among a cohort of veterans who served in the era of the Iraq and Afghanistan conflicts. Am J Public Health. 2013;103(suppl 2):S255-S261.
21. Perl L. Veterans and homelessness. Congressional research Service Report RL34024. https://www.fas .org/sgp/crs/misc/RL34024.pdf.Published November 6, 2015. Accessed April 7, 2016.
22. Rossi PH. Down and Out in America: The Origins of Homelessness. Chicago, IL: University of Chicago Press; 1989.
23. Burt M. Over the Edge: The Growth of Homelessness in the 1980s. New York, NY: Russell Sage Foundation; 1992.
24. National Coalition for the Homeless. Homeless Veterans [fact sheet]. http://www .nationalhomeless.org/factsheets/veterans.html. September 2009. Accessed April 19, 2016.
25. Tessler R, Rosenheck R, Gamache F. Comparison of homeless veterans with other homeless men in a large clinical outreach program. Psychiatr Q. 2002;73(2):109-119.
26. Chapman H. Homelessness in Metropolitan Washington: Results and Analysis From the 2015 Point-in-Time Count of Persons Experiencing Homelessness in the Metropolitan Washington Region. Metropolitan Washington Council of Governments website. https://www .mwcog.org/uploads/pub-documents/v15Wlk20150514094353.pdf. Published May 13, 2015. Accessed April 6, 2016.
27. Tsai J, Rosenheck R. Risk factors for homelessness among US veterans. Epidemiol Rev. 2015;37:177-195.
28. Edens EL, Kasprow W, Tsai J, Rosenheck R. Association of substance use and VA service-connected disability benefits with risk of homelessness among veterans. Am J Addict. 2011;20(5):412-419.
29. OrgCode Consulting. Service Prioritization Decision Assistance Tool (SPDAT). http://www.orgcode .com/product/spdat/. Accessed April 19, 2016.
30. Performance.gov. End veteran homelessness: progress update. Performance.gov website. https://www.performance.gov/content/end-veteran-homelessness#progressUpdate. Accessed April 6, 2016.
1. U.S. Department of Housing and Urban Development, Office of Community Planning and Development; U.S. Department of Veterans Affairs, National Center on Homelessness Among Veterans Veteran Homelessness: A Supplemental Report to the 2009 Annual Homeless Assessment Report to Congress.https://www.hudexchange.info/resources/documents/2009AHARveterans Report.pdf. Accessed April 19, 2016.
2. HUD Exchange. Homeless emergency assistance and rapid transition to housing (HEARTH): defining “homeless” final rule. HUD Exchange website. https://www.hudexchange.info/resource/1928 /hearth-defining-homeless-final-rule. Published November 2011. Accessed April 5, 2016.
3. U.S. Department of Health and Human Services, Administration for Community Living (ACL), Administration on Intellectual and Developmental Disabilities. The Developmental Disabilities Assistance and Bill of Rights Act of 2000. http://www.acl.gov/Programs/AIDD/DDA_BOR _ACT_2000/docs/dd_act.pdf. Accessed April 5, 2016. 4. Hwang SW. Mortality among men using homeless shelters in Toronto, Ontario. JAMA. 2000;283(16):2152-2157.
5. O’Connell JJ. Premature Mortality in Homeless Populations: A Review of the Literature. Nashville, TN: National Health Care for the Homeless Council; 2005.
6. Salit SA, Kuhn EM, Hartz AJ, Vu JM, Mosso AL. Hopitalization costs associated with homelessness in New York City. N Engl J Med. 1998;338(24):1734-1740.
7. Kushel MB, Perry S, Bangsberg D, Clark R, Moss AR. Emergency department use among the homeless and marginally housed: results from a community-based study. Am J Public Health. 2002;92(5):778-784.
8. Larimer ME, Malone DK, Garner MD, et al. Healthcare and public service use and costs before and after provision of housing for chronically homeless persons with severe alcohol problems. JAMA. 2009;301(13):1349-1357.
9. McNeil DE, Binder RL, Robinson JC. Incarceration associated with homelessness, mental disorder, and co-occurring substance abuse. Psychiatr Serv. 2005;56(7):840-846.
10. Rosenheck R. Cost-effectiveness of services for mentally ill homeless people: the application of research to policy and practice. Am J Psychiatry. 2000;157(10):1563-1570.
11. Culhane DP, Metraux S, Hadley T. Public service reductions associated with placement of homeless people with severe mental illness in supportive housing. Housing Policy Debate. 2002;13(1):107-163.
12. Martinez TE, Burt MR. Impact of permanent supportive housing on the use of acute care health services by homeless adults. Psychiatr Serv. 2006;57(7):992-999.
13. Culhane DP, Parker WD, Poppe B, et al. Accountability, cost-effectiveness, and program performance: progress since 1998. In: Dennis D, Locke G, Khadduri J, eds. Toward Understanding Homelessness: The 2007 National Symposium on Homelessness Research. Washington, DC: Substance Abuse and Mental Health Services Administration, U.S. Dept of Health and Human Services; Office of Policy Development and Research, U.S. Dept of Housing and Urban Development; 2007.
14. Poulin SR, Maguire M, Metraux S, Culhane DP. Service use and costs for persons experiencing chronic homelessness in Philadelphia: a population-based study. Psychiatr Serv. 2010;61(11):1093-1098.
15. U.S. Department of Veterans Affairs. VA History in Brief. http://www.va.gov/opa/publications/archives /docs/history_in_brief.pdf. U.S. Department of Veteran Affairs website. Published December 27, 2013. Accessed April 19, 2016.
16. Kusmer KL. Down and Out, on the Road: The Homeless in American History. New York, NY: Oxford University Press; 2002.
17. Baumohl J, ed. Homelessness in America. Westport, CT: Oryx Press; 1996.
18. Fargo J, Metraux S, Byrne T, et al. Prevalence and risk of homelessness among U.S. veterans: a multisite investigation. http://www.va.gov/HOMELESS/docs/Center/Prevalence_Final.pdf. Published August 2011. Accessed April 19, 2016.
19. Tsai J, Mares AS, Rosenheck R, Gamache D. Do homeless veterans have the same needs and outcomes as non-veterans? Mil Med. 2012;177(1):27-31.
20. Metraux S, Clegg L, Daigh JD, Culhane DP, Kane V. Risk factors for becoming homeless among a cohort of veterans who served in the era of the Iraq and Afghanistan conflicts. Am J Public Health. 2013;103(suppl 2):S255-S261.
21. Perl L. Veterans and homelessness. Congressional research Service Report RL34024. https://www.fas .org/sgp/crs/misc/RL34024.pdf.Published November 6, 2015. Accessed April 7, 2016.
22. Rossi PH. Down and Out in America: The Origins of Homelessness. Chicago, IL: University of Chicago Press; 1989.
23. Burt M. Over the Edge: The Growth of Homelessness in the 1980s. New York, NY: Russell Sage Foundation; 1992.
24. National Coalition for the Homeless. Homeless Veterans [fact sheet]. http://www .nationalhomeless.org/factsheets/veterans.html. September 2009. Accessed April 19, 2016.
25. Tessler R, Rosenheck R, Gamache F. Comparison of homeless veterans with other homeless men in a large clinical outreach program. Psychiatr Q. 2002;73(2):109-119.
26. Chapman H. Homelessness in Metropolitan Washington: Results and Analysis From the 2015 Point-in-Time Count of Persons Experiencing Homelessness in the Metropolitan Washington Region. Metropolitan Washington Council of Governments website. https://www .mwcog.org/uploads/pub-documents/v15Wlk20150514094353.pdf. Published May 13, 2015. Accessed April 6, 2016.
27. Tsai J, Rosenheck R. Risk factors for homelessness among US veterans. Epidemiol Rev. 2015;37:177-195.
28. Edens EL, Kasprow W, Tsai J, Rosenheck R. Association of substance use and VA service-connected disability benefits with risk of homelessness among veterans. Am J Addict. 2011;20(5):412-419.
29. OrgCode Consulting. Service Prioritization Decision Assistance Tool (SPDAT). http://www.orgcode .com/product/spdat/. Accessed April 19, 2016.
30. Performance.gov. End veteran homelessness: progress update. Performance.gov website. https://www.performance.gov/content/end-veteran-homelessness#progressUpdate. Accessed April 6, 2016.
Military sexual trauma tied to homelessness risk among veterans
A positive screen for military sexual trauma among recently discharged male and female veterans may be a predictive factor for homelessness. In addition, the association between military sexual trauma and homelessness is stronger among male veterans, results of a retrospective study published April 20 show.
Emily Brignone, Dr. Adi V. Gundlapalli, and their associates examined health care data from the Veterans Health Administration’s 2011 Operation Enduring Freedom and Operation Iraqi Freedom official roster of veterans. All of the veterans separated from the military between fiscal years 2001 and 2011, and used Veterans Health Administration (VHA) services between fiscal years 2004 and 2013 (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0101).
The total study population included 601,892, 590,989, and 262,589 U.S. veterans who screened positive for military sexual trauma at 30 days, 1 year, and 5 years, respectively, after their initial VHA visit, reported Ms. Brignone and Dr. Gundlapalli, both of whom are affiliated with the Informatics, Decision Enhancement, and Analytic Sciences Center at the VA Salt Lake City Health Care System.
They found that the incidence of homelessness in this population was 1.6%, 4.4%, and 9.6% within 30 days, 1 year, and 5 years, respectively. The rates for male veterans were higher than for their female counterparts, as evidenced by 30-day, 1-year, and 5-year homelessness rates of 2.3%, 6.2%, and 11.8%, respectively, compared with 1.3%, 3.9%, and 8.9%. About 25% of female veterans report experiencing sexual trauma during their military service, compared with 1% of male veterans, research shows (Am J Public Health. 2007;97[12]:2160-6).
Meanwhile, the rates of positive military sexual trauma screens among homeless veterans were almost twice as high, compared with the rates of veterans who were not homeless (0.7%, 1.8%, and 4.3%, respectively).
Logistic regression analysis models adjusted for mental health and substance use diagnoses showed that military sexual trauma screen status remained significantly associated with homelessness, such that veterans with a positive military sexual trauma screen were roughly 1.5-fold more likely to be homelessness than those with a negative screen. The adjusted models also showed that the interaction between military sexual trauma status and sex remained significant for the 30-day and 1-year cohorts.
Ms. Brignone, Dr. Gundlapalli, and their associates cited several limitations. For example, a positive screen for military sexual trauma is a self-reported marker rather than a diagnosis. “Because a positive screen for [military sexual trauma] is associated with increased service use, there may be more opportunities to detect homelessness among veterans with a positive screen,” they wrote.
The investigators said further research is needed to understand the temporal associations between sexual trauma, mental health diagnoses, and sex-dependent differences. Understanding those associations might be useful in attempts at prevention and intervention of postdeployment homelessness, they wrote.
A U.S. Department of Veterans Affairs grant funded this project. The authors disclosed no conflicts of interest.
The results of the study by Emily Brignone, Dr. Adi V. Gundlapalli, and their associates should promote zero tolerance for the perpetration of sexual trauma in the military, Natalie Mota, Ph.D., and her associates wrote in an accompanying editorial. “Education and awareness about the widespread deleterious effects of [military sexual trauma] on mental and physical health as well as military cohesion and productivity could help to advance this aim,” they wrote.
Ms. Brignone’s study also should prompt efforts to identify veterans with military sexual trauma in order to facilitate timely connections to evidence-based interventions such as Housing First approaches. Also, continued research focused on personalizing screening and outreach efforts specifically targeted to this population will be required to identify veterans at increased risk for postdeployment homelessness, they wrote.
Other possible solutions to reducing postdeployment homelessness are facilitated, supported, and encouraged reporting of military sexual trauma, sensitive and effective assessment of military sexual trauma, and facilitated access to evidence-based interventions for military sexual trauma–related mental health problems across health care systems.
Dr. Mota is affiliated with the department of clinical health psychology at the University of Manitoba in Winnipeg (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0136).
The results of the study by Emily Brignone, Dr. Adi V. Gundlapalli, and their associates should promote zero tolerance for the perpetration of sexual trauma in the military, Natalie Mota, Ph.D., and her associates wrote in an accompanying editorial. “Education and awareness about the widespread deleterious effects of [military sexual trauma] on mental and physical health as well as military cohesion and productivity could help to advance this aim,” they wrote.
Ms. Brignone’s study also should prompt efforts to identify veterans with military sexual trauma in order to facilitate timely connections to evidence-based interventions such as Housing First approaches. Also, continued research focused on personalizing screening and outreach efforts specifically targeted to this population will be required to identify veterans at increased risk for postdeployment homelessness, they wrote.
Other possible solutions to reducing postdeployment homelessness are facilitated, supported, and encouraged reporting of military sexual trauma, sensitive and effective assessment of military sexual trauma, and facilitated access to evidence-based interventions for military sexual trauma–related mental health problems across health care systems.
Dr. Mota is affiliated with the department of clinical health psychology at the University of Manitoba in Winnipeg (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0136).
The results of the study by Emily Brignone, Dr. Adi V. Gundlapalli, and their associates should promote zero tolerance for the perpetration of sexual trauma in the military, Natalie Mota, Ph.D., and her associates wrote in an accompanying editorial. “Education and awareness about the widespread deleterious effects of [military sexual trauma] on mental and physical health as well as military cohesion and productivity could help to advance this aim,” they wrote.
Ms. Brignone’s study also should prompt efforts to identify veterans with military sexual trauma in order to facilitate timely connections to evidence-based interventions such as Housing First approaches. Also, continued research focused on personalizing screening and outreach efforts specifically targeted to this population will be required to identify veterans at increased risk for postdeployment homelessness, they wrote.
Other possible solutions to reducing postdeployment homelessness are facilitated, supported, and encouraged reporting of military sexual trauma, sensitive and effective assessment of military sexual trauma, and facilitated access to evidence-based interventions for military sexual trauma–related mental health problems across health care systems.
Dr. Mota is affiliated with the department of clinical health psychology at the University of Manitoba in Winnipeg (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0136).
A positive screen for military sexual trauma among recently discharged male and female veterans may be a predictive factor for homelessness. In addition, the association between military sexual trauma and homelessness is stronger among male veterans, results of a retrospective study published April 20 show.
Emily Brignone, Dr. Adi V. Gundlapalli, and their associates examined health care data from the Veterans Health Administration’s 2011 Operation Enduring Freedom and Operation Iraqi Freedom official roster of veterans. All of the veterans separated from the military between fiscal years 2001 and 2011, and used Veterans Health Administration (VHA) services between fiscal years 2004 and 2013 (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0101).
The total study population included 601,892, 590,989, and 262,589 U.S. veterans who screened positive for military sexual trauma at 30 days, 1 year, and 5 years, respectively, after their initial VHA visit, reported Ms. Brignone and Dr. Gundlapalli, both of whom are affiliated with the Informatics, Decision Enhancement, and Analytic Sciences Center at the VA Salt Lake City Health Care System.
They found that the incidence of homelessness in this population was 1.6%, 4.4%, and 9.6% within 30 days, 1 year, and 5 years, respectively. The rates for male veterans were higher than for their female counterparts, as evidenced by 30-day, 1-year, and 5-year homelessness rates of 2.3%, 6.2%, and 11.8%, respectively, compared with 1.3%, 3.9%, and 8.9%. About 25% of female veterans report experiencing sexual trauma during their military service, compared with 1% of male veterans, research shows (Am J Public Health. 2007;97[12]:2160-6).
Meanwhile, the rates of positive military sexual trauma screens among homeless veterans were almost twice as high, compared with the rates of veterans who were not homeless (0.7%, 1.8%, and 4.3%, respectively).
Logistic regression analysis models adjusted for mental health and substance use diagnoses showed that military sexual trauma screen status remained significantly associated with homelessness, such that veterans with a positive military sexual trauma screen were roughly 1.5-fold more likely to be homelessness than those with a negative screen. The adjusted models also showed that the interaction between military sexual trauma status and sex remained significant for the 30-day and 1-year cohorts.
Ms. Brignone, Dr. Gundlapalli, and their associates cited several limitations. For example, a positive screen for military sexual trauma is a self-reported marker rather than a diagnosis. “Because a positive screen for [military sexual trauma] is associated with increased service use, there may be more opportunities to detect homelessness among veterans with a positive screen,” they wrote.
The investigators said further research is needed to understand the temporal associations between sexual trauma, mental health diagnoses, and sex-dependent differences. Understanding those associations might be useful in attempts at prevention and intervention of postdeployment homelessness, they wrote.
A U.S. Department of Veterans Affairs grant funded this project. The authors disclosed no conflicts of interest.
A positive screen for military sexual trauma among recently discharged male and female veterans may be a predictive factor for homelessness. In addition, the association between military sexual trauma and homelessness is stronger among male veterans, results of a retrospective study published April 20 show.
Emily Brignone, Dr. Adi V. Gundlapalli, and their associates examined health care data from the Veterans Health Administration’s 2011 Operation Enduring Freedom and Operation Iraqi Freedom official roster of veterans. All of the veterans separated from the military between fiscal years 2001 and 2011, and used Veterans Health Administration (VHA) services between fiscal years 2004 and 2013 (JAMA Psychiatry. 2016 Apr 20. doi: 10.1001/jamapsychiatry.2016.0101).
The total study population included 601,892, 590,989, and 262,589 U.S. veterans who screened positive for military sexual trauma at 30 days, 1 year, and 5 years, respectively, after their initial VHA visit, reported Ms. Brignone and Dr. Gundlapalli, both of whom are affiliated with the Informatics, Decision Enhancement, and Analytic Sciences Center at the VA Salt Lake City Health Care System.
They found that the incidence of homelessness in this population was 1.6%, 4.4%, and 9.6% within 30 days, 1 year, and 5 years, respectively. The rates for male veterans were higher than for their female counterparts, as evidenced by 30-day, 1-year, and 5-year homelessness rates of 2.3%, 6.2%, and 11.8%, respectively, compared with 1.3%, 3.9%, and 8.9%. About 25% of female veterans report experiencing sexual trauma during their military service, compared with 1% of male veterans, research shows (Am J Public Health. 2007;97[12]:2160-6).
Meanwhile, the rates of positive military sexual trauma screens among homeless veterans were almost twice as high, compared with the rates of veterans who were not homeless (0.7%, 1.8%, and 4.3%, respectively).
Logistic regression analysis models adjusted for mental health and substance use diagnoses showed that military sexual trauma screen status remained significantly associated with homelessness, such that veterans with a positive military sexual trauma screen were roughly 1.5-fold more likely to be homelessness than those with a negative screen. The adjusted models also showed that the interaction between military sexual trauma status and sex remained significant for the 30-day and 1-year cohorts.
Ms. Brignone, Dr. Gundlapalli, and their associates cited several limitations. For example, a positive screen for military sexual trauma is a self-reported marker rather than a diagnosis. “Because a positive screen for [military sexual trauma] is associated with increased service use, there may be more opportunities to detect homelessness among veterans with a positive screen,” they wrote.
The investigators said further research is needed to understand the temporal associations between sexual trauma, mental health diagnoses, and sex-dependent differences. Understanding those associations might be useful in attempts at prevention and intervention of postdeployment homelessness, they wrote.
A U.S. Department of Veterans Affairs grant funded this project. The authors disclosed no conflicts of interest.
FROM JAMA PSYCHIATRY
Key clinical point: Male and female U.S. veterans who screen positive for military sexual trauma have an increased risk of homelessness postdeployment.
Major finding: The primary outcome – the incidence of homelessness among veterans with a positive screen for military sexual trauma – was 1.6% within 30 days, 4.4% within 1 year, and 9.6% within 5 years.
Data source: Health care data on three cohorts (30 days, 1 year, and 5 years) of veterans who separated from the military between fiscal years 2001 and 2011 from the Veterans Health Administration’s 2011 Operation Enduring Freedom and Operation Iraqi Freedom official roster.
Disclosures: A U.S. Department of Veterans Affairs grant funded this project. The authors disclosed no conflicts of interest.
PSYCHIATRY UPDATE 2016
View summaries from the event on the following pages.
Thursday, March 10, 2016
Make Way for Possibilities of an Adjunctive Treatment for Major Depressive Disorder
Roueen Rafeyan, MD, Feinberg School of Medicine at Northwestern University
Successful Aging
George T. Grossberg, MD, Saint Louis University
Psychopharmacology and Pregnancy: The New Labeling Changes and Implications for Clinical Practice
Marlene P. Freeman, MD, Massachusetts General Hospital
Anxiety Disorders in Women Across the Lifecycle
Marlene P. Freeman, MD, Massachusetts General Hospital
Mild Cognitive Impairment: “Senior Moments” and DSM-5
George T. Grossberg, MD, Saint Louis University
Assessing major depressive disorder and an option for treatment
Jay D. Fawver, MD, Indiana University School of Medicine
Innovative Treatments of Anxiety, Part 1 (Use of Benzodiazepines)
Mark H. Pollack, MD, Rush University Medical Center
Innovative Treatments of Anxiety, Part 2 (Other Standard and Novel Therapeutic Approaches)
Mark H. Pollack, MD, Rush University Medical Center
Treatment of Chronic Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital
Friday, March 11, 2016
Subtypes of Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Managing ADHD: What Matters Most When Selecting a Treatment Option
Michael Feld, MD, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Dr. Feld discussed the utility of the brand-name extended-release (ER) methylphenidate HCl (Aptensio) for its value in children—specifically, its ability to “extend the day” without additional dosing of a short-action medication. The design of Aptensio—a multilayered beaded delivery system in which every bead is both an immediate- and an extended-release vehicle—allows an early peak serum drug level and later peak level (at 8 hours). Aptensio is administered by sprinkling the contents of a capsule on applesauce; it is is safe practice, Dr. Feld explained, to augment the ER drug delivery with an immediate-release agent when deemed necessary, by observing how difficult it is for the patient to make it through the day at home, school, or work.
Overview of Autism Spectrum Disorder
Robert L. Hendren, DO, University of California, San Francisco
Comorbidity of Schizophrenia and Substance Abuse
Henry A. Nasrallah, MD, Saint Louis University
Overview of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Bipolar Depression: Presentation, Diagnosis, and Treatment in the Outpatient Psychiatry Practice Setting
Peter J. Weiden, MD, University of Illinois at Chicago
Neuroinflammation and Oxidative Stress in Schizophrenia and Mood Disorders: Biomarkers and Therapeutic Targets
Henry A. Nasrallah, MD, Saint Louis University
Clinical Management of Autism Spectrum Disorders: What Happens Over Time/Borderline Intellectual Functioning
Robert L. Hendren, DO, University of California, San Francisco
Management of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Saturday, March 12, 2015
Managing the Difficult Child
Anthony L. Rostain, MD, MA, University of Pennsylvania
Major Depression With Subsyndromal Mania/Hypomania: Implications for Diagnosis and Management
Trisha Suppes, MD, PhD, Stanford University School of Medicine, Roger S. McIntyre, MD, FRCPC, University of Toronto, and J. Craig Nelson, MD, University of California, San Francisco
General Overview of Sleep Disorders
Thomas Roth, PhD, Henry Ford Hospital
Comorbid ADHD with Substance Abuse
Anthony L. Rostain, MD, MA, University of Pennsylvania
How to Treat Patients with Insomnia
Thomas Roth, PhD, Henry Ford Hospital
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults
View summaries from the event on the following pages.
Thursday, March 10, 2016
Make Way for Possibilities of an Adjunctive Treatment for Major Depressive Disorder
Roueen Rafeyan, MD, Feinberg School of Medicine at Northwestern University
Successful Aging
George T. Grossberg, MD, Saint Louis University
Psychopharmacology and Pregnancy: The New Labeling Changes and Implications for Clinical Practice
Marlene P. Freeman, MD, Massachusetts General Hospital
Anxiety Disorders in Women Across the Lifecycle
Marlene P. Freeman, MD, Massachusetts General Hospital
Mild Cognitive Impairment: “Senior Moments” and DSM-5
George T. Grossberg, MD, Saint Louis University
Assessing major depressive disorder and an option for treatment
Jay D. Fawver, MD, Indiana University School of Medicine
Innovative Treatments of Anxiety, Part 1 (Use of Benzodiazepines)
Mark H. Pollack, MD, Rush University Medical Center
Innovative Treatments of Anxiety, Part 2 (Other Standard and Novel Therapeutic Approaches)
Mark H. Pollack, MD, Rush University Medical Center
Treatment of Chronic Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital
Friday, March 11, 2016
Subtypes of Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Managing ADHD: What Matters Most When Selecting a Treatment Option
Michael Feld, MD, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Dr. Feld discussed the utility of the brand-name extended-release (ER) methylphenidate HCl (Aptensio) for its value in children—specifically, its ability to “extend the day” without additional dosing of a short-action medication. The design of Aptensio—a multilayered beaded delivery system in which every bead is both an immediate- and an extended-release vehicle—allows an early peak serum drug level and later peak level (at 8 hours). Aptensio is administered by sprinkling the contents of a capsule on applesauce; it is is safe practice, Dr. Feld explained, to augment the ER drug delivery with an immediate-release agent when deemed necessary, by observing how difficult it is for the patient to make it through the day at home, school, or work.
Overview of Autism Spectrum Disorder
Robert L. Hendren, DO, University of California, San Francisco
Comorbidity of Schizophrenia and Substance Abuse
Henry A. Nasrallah, MD, Saint Louis University
Overview of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Bipolar Depression: Presentation, Diagnosis, and Treatment in the Outpatient Psychiatry Practice Setting
Peter J. Weiden, MD, University of Illinois at Chicago
Neuroinflammation and Oxidative Stress in Schizophrenia and Mood Disorders: Biomarkers and Therapeutic Targets
Henry A. Nasrallah, MD, Saint Louis University
Clinical Management of Autism Spectrum Disorders: What Happens Over Time/Borderline Intellectual Functioning
Robert L. Hendren, DO, University of California, San Francisco
Management of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Saturday, March 12, 2015
Managing the Difficult Child
Anthony L. Rostain, MD, MA, University of Pennsylvania
Major Depression With Subsyndromal Mania/Hypomania: Implications for Diagnosis and Management
Trisha Suppes, MD, PhD, Stanford University School of Medicine, Roger S. McIntyre, MD, FRCPC, University of Toronto, and J. Craig Nelson, MD, University of California, San Francisco
General Overview of Sleep Disorders
Thomas Roth, PhD, Henry Ford Hospital
Comorbid ADHD with Substance Abuse
Anthony L. Rostain, MD, MA, University of Pennsylvania
How to Treat Patients with Insomnia
Thomas Roth, PhD, Henry Ford Hospital
View summaries from the event on the following pages.
Thursday, March 10, 2016
Make Way for Possibilities of an Adjunctive Treatment for Major Depressive Disorder
Roueen Rafeyan, MD, Feinberg School of Medicine at Northwestern University
Successful Aging
George T. Grossberg, MD, Saint Louis University
Psychopharmacology and Pregnancy: The New Labeling Changes and Implications for Clinical Practice
Marlene P. Freeman, MD, Massachusetts General Hospital
Anxiety Disorders in Women Across the Lifecycle
Marlene P. Freeman, MD, Massachusetts General Hospital
Mild Cognitive Impairment: “Senior Moments” and DSM-5
George T. Grossberg, MD, Saint Louis University
Assessing major depressive disorder and an option for treatment
Jay D. Fawver, MD, Indiana University School of Medicine
Innovative Treatments of Anxiety, Part 1 (Use of Benzodiazepines)
Mark H. Pollack, MD, Rush University Medical Center
Innovative Treatments of Anxiety, Part 2 (Other Standard and Novel Therapeutic Approaches)
Mark H. Pollack, MD, Rush University Medical Center
Treatment of Chronic Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital
Friday, March 11, 2016
Subtypes of Depression
Andrew A. Nierenberg, MD, Massachusetts General Hospital, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Managing ADHD: What Matters Most When Selecting a Treatment Option
Michael Feld, MD, Alexian Brothers Behavioral Health Hospital for Violence Prevention Clinic/Program and ADHD Clinic
Dr. Feld discussed the utility of the brand-name extended-release (ER) methylphenidate HCl (Aptensio) for its value in children—specifically, its ability to “extend the day” without additional dosing of a short-action medication. The design of Aptensio—a multilayered beaded delivery system in which every bead is both an immediate- and an extended-release vehicle—allows an early peak serum drug level and later peak level (at 8 hours). Aptensio is administered by sprinkling the contents of a capsule on applesauce; it is is safe practice, Dr. Feld explained, to augment the ER drug delivery with an immediate-release agent when deemed necessary, by observing how difficult it is for the patient to make it through the day at home, school, or work.
Overview of Autism Spectrum Disorder
Robert L. Hendren, DO, University of California, San Francisco
Comorbidity of Schizophrenia and Substance Abuse
Henry A. Nasrallah, MD, Saint Louis University
Overview of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Bipolar Depression: Presentation, Diagnosis, and Treatment in the Outpatient Psychiatry Practice Setting
Peter J. Weiden, MD, University of Illinois at Chicago
Neuroinflammation and Oxidative Stress in Schizophrenia and Mood Disorders: Biomarkers and Therapeutic Targets
Henry A. Nasrallah, MD, Saint Louis University
Clinical Management of Autism Spectrum Disorders: What Happens Over Time/Borderline Intellectual Functioning
Robert L. Hendren, DO, University of California, San Francisco
Management of PTSD
Carol S. North, MD, MPE, DFAPA, University of Texas Southwestern Medical Center
Saturday, March 12, 2015
Managing the Difficult Child
Anthony L. Rostain, MD, MA, University of Pennsylvania
Major Depression With Subsyndromal Mania/Hypomania: Implications for Diagnosis and Management
Trisha Suppes, MD, PhD, Stanford University School of Medicine, Roger S. McIntyre, MD, FRCPC, University of Toronto, and J. Craig Nelson, MD, University of California, San Francisco
General Overview of Sleep Disorders
Thomas Roth, PhD, Henry Ford Hospital
Comorbid ADHD with Substance Abuse
Anthony L. Rostain, MD, MA, University of Pennsylvania
How to Treat Patients with Insomnia
Thomas Roth, PhD, Henry Ford Hospital
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults
personality disorder, DSM-5, adults with ADHD, residual depressive symptoms, treatment-resistant depression,antisocial personality disorder, bipolar disorder, schizophrenia, psychotic disorder, clozapine, bipolar disorder and substance abuse, mood disorders during pregnancy, premenstrual dysphoric disorder, depressive symptoms in perimenopause, smoking and the mentally ill, help patients with mental illness lose weight, substance abuse in older adults
Sleep apnea found in 57% of veterans with PTSD
Obstructive sleep apnea syndrome (OSAS) was diagnosed in more than half of 200 active duty service members with combat-related post-traumatic stress disorder (PTSD) who were studied at Walter Reed Army Medical Center in Washington.
Compared with age-matched peers with just one of these disorders, the service members with PTSD and OSAS had poorer somnolence and sleep-related quality of life and were less adherent and responsive to positive airway pressure therapy.
The findings “highlight the need for a high index of suspicion and a comprehensive approach to identifying and treating sleep-disordered breathing in these patients,” Dr. Christopher J. Lettieri of the Uniformed Services University in Bethesda, Md., and his associates wrote (Chest. 2016 Feb;149[2]:483-90). “Given the prevalence of OSAS in patients with PTSD and its adverse impact on symptoms and adherence, early identification may improve outcomes.”
In the observational cohort study, 200 consecutive active duty service members who were diagnosed with PTSD as part of post-deployment screening underwent sleep evaluations regardless of whether there was clinical suspicion of sleep-disordered breathing. More than half – about 57% – were diagnosed with OSAS. Almost 60% of the study group had mild traumatic brain injury, which has been connected in prior research to obstructive sleep apnea, and many had comorbid insomnia. Those who were diagnosed with OSAS were older and had higher BMIs than those not found to have OSAS.
All 200 patients were compared with 50 consecutive age-matched control patients who had OSAS but had not been deployed and did not have PTSD, as well as with 50 age-matched service members without prior deployment or either of the two disorders. All of the patients diagnosed with OSAS were prescribed positive airway pressure (PAP) therapy and evaluated after a month.
Sleep quality was poor in the majority of patients with PTSD, and OSAS and PTSD were both independently associated with increased daytime sleepiness and lower quality-of-life index scores. However, patients with both conditions fared significantly worse, particularly with respect to quality of life as measured by the Functional Outcomes of Sleep Questionnaire (FOSQ).
FOSQ scores were abnormal at baseline in 60% of those with PTSD and OSAS, 43% with PTSD alone, 24% with OSAS alone, and 7% of those with neither condition.
Service members with both conditions also were less likely to adhere to therapy; 30% regularly used continuous PAP therapy, compared with 55% of those who had OSAS alone.
And while continuous PAP therapy improved daytime sleepiness and quality of life in patients with both PTSD and OSAS, the degree of improvement was less than that experienced by those with OSAS alone. PTSD “represents an independent barrier to the effective treatment of OSAS and should prompt multipronged and individualized care,” they wrote.
The researchers reported having no financial disclosures.
Obstructive sleep apnea syndrome (OSAS) was diagnosed in more than half of 200 active duty service members with combat-related post-traumatic stress disorder (PTSD) who were studied at Walter Reed Army Medical Center in Washington.
Compared with age-matched peers with just one of these disorders, the service members with PTSD and OSAS had poorer somnolence and sleep-related quality of life and were less adherent and responsive to positive airway pressure therapy.
The findings “highlight the need for a high index of suspicion and a comprehensive approach to identifying and treating sleep-disordered breathing in these patients,” Dr. Christopher J. Lettieri of the Uniformed Services University in Bethesda, Md., and his associates wrote (Chest. 2016 Feb;149[2]:483-90). “Given the prevalence of OSAS in patients with PTSD and its adverse impact on symptoms and adherence, early identification may improve outcomes.”
In the observational cohort study, 200 consecutive active duty service members who were diagnosed with PTSD as part of post-deployment screening underwent sleep evaluations regardless of whether there was clinical suspicion of sleep-disordered breathing. More than half – about 57% – were diagnosed with OSAS. Almost 60% of the study group had mild traumatic brain injury, which has been connected in prior research to obstructive sleep apnea, and many had comorbid insomnia. Those who were diagnosed with OSAS were older and had higher BMIs than those not found to have OSAS.
All 200 patients were compared with 50 consecutive age-matched control patients who had OSAS but had not been deployed and did not have PTSD, as well as with 50 age-matched service members without prior deployment or either of the two disorders. All of the patients diagnosed with OSAS were prescribed positive airway pressure (PAP) therapy and evaluated after a month.
Sleep quality was poor in the majority of patients with PTSD, and OSAS and PTSD were both independently associated with increased daytime sleepiness and lower quality-of-life index scores. However, patients with both conditions fared significantly worse, particularly with respect to quality of life as measured by the Functional Outcomes of Sleep Questionnaire (FOSQ).
FOSQ scores were abnormal at baseline in 60% of those with PTSD and OSAS, 43% with PTSD alone, 24% with OSAS alone, and 7% of those with neither condition.
Service members with both conditions also were less likely to adhere to therapy; 30% regularly used continuous PAP therapy, compared with 55% of those who had OSAS alone.
And while continuous PAP therapy improved daytime sleepiness and quality of life in patients with both PTSD and OSAS, the degree of improvement was less than that experienced by those with OSAS alone. PTSD “represents an independent barrier to the effective treatment of OSAS and should prompt multipronged and individualized care,” they wrote.
The researchers reported having no financial disclosures.
Obstructive sleep apnea syndrome (OSAS) was diagnosed in more than half of 200 active duty service members with combat-related post-traumatic stress disorder (PTSD) who were studied at Walter Reed Army Medical Center in Washington.
Compared with age-matched peers with just one of these disorders, the service members with PTSD and OSAS had poorer somnolence and sleep-related quality of life and were less adherent and responsive to positive airway pressure therapy.
The findings “highlight the need for a high index of suspicion and a comprehensive approach to identifying and treating sleep-disordered breathing in these patients,” Dr. Christopher J. Lettieri of the Uniformed Services University in Bethesda, Md., and his associates wrote (Chest. 2016 Feb;149[2]:483-90). “Given the prevalence of OSAS in patients with PTSD and its adverse impact on symptoms and adherence, early identification may improve outcomes.”
In the observational cohort study, 200 consecutive active duty service members who were diagnosed with PTSD as part of post-deployment screening underwent sleep evaluations regardless of whether there was clinical suspicion of sleep-disordered breathing. More than half – about 57% – were diagnosed with OSAS. Almost 60% of the study group had mild traumatic brain injury, which has been connected in prior research to obstructive sleep apnea, and many had comorbid insomnia. Those who were diagnosed with OSAS were older and had higher BMIs than those not found to have OSAS.
All 200 patients were compared with 50 consecutive age-matched control patients who had OSAS but had not been deployed and did not have PTSD, as well as with 50 age-matched service members without prior deployment or either of the two disorders. All of the patients diagnosed with OSAS were prescribed positive airway pressure (PAP) therapy and evaluated after a month.
Sleep quality was poor in the majority of patients with PTSD, and OSAS and PTSD were both independently associated with increased daytime sleepiness and lower quality-of-life index scores. However, patients with both conditions fared significantly worse, particularly with respect to quality of life as measured by the Functional Outcomes of Sleep Questionnaire (FOSQ).
FOSQ scores were abnormal at baseline in 60% of those with PTSD and OSAS, 43% with PTSD alone, 24% with OSAS alone, and 7% of those with neither condition.
Service members with both conditions also were less likely to adhere to therapy; 30% regularly used continuous PAP therapy, compared with 55% of those who had OSAS alone.
And while continuous PAP therapy improved daytime sleepiness and quality of life in patients with both PTSD and OSAS, the degree of improvement was less than that experienced by those with OSAS alone. PTSD “represents an independent barrier to the effective treatment of OSAS and should prompt multipronged and individualized care,” they wrote.
The researchers reported having no financial disclosures.
FROM CHEST
Key clinical point: Obstructive sleep apnea is prevalent in service members with PTSD.
Major finding: More than 57% of active duty service members with combat-related PTSD were diagnosed with OSAS.
Data source: A case-controlled observational cohort study conducted at an academic military medical center and involving 200 consecutive patients with PTSD.
Disclosures: Dr. Lettieri and his colleagues did not report any conflicts of interest.
Sex assault by intimate partners as distressing as stranger attack
Sexual assault by an intimate partner was associated with a similar level of psychological distress and a greater likelihood of extragenital traumatic injuries, compared with sexual assaults by a stranger or an acquaintance.
Dr. Patrick Chariot of the department of forensic medicine at Hôpital Jean-Verdier in Bondy, France, and his colleagues conducted an observational and prospective study to compare the psychological and physical symptoms of women assaulted by intimate partners with the symptoms of women who were assaulted by an acquaintance or unknown person.
Participants included girls and women aged 15 years or older who were referred to a sexual assault center and received examination. Data was grouped according to the woman’s reported relationship to their assailant as a current or former intimate partner, stranger, or acquaintance (Obstet Gynecol. 2016 Feb;127:516–26).
A total of 767 patients were included in the study. Assault by an intimate partner was reported by 263 women, assault by an acquaintance by 229 women, and assault by a stranger by 275 women. A 1-month follow-up examination was performed in 38% of participants.
Nearly half of the study participants reported a previous physical or sexual assault. A history of previous assault was more often reported by women in the intimate partner group (71%), compared with those in the acquaintance group (49%) and those in the stranger group (28%).
Extragenital trauma was more common in women assaulted by intimate partners (52% versus 33% and 43%, in acquaintance and strangers, respectively).
The most common psychological symptoms reported at the time of examination included anxiety, fear, shame, and sadness with reports of fear being more common in those with an intimate partner assault (46% versus 30% and 25%). The most common symptoms reported at follow-up included sleep disorders, depression, fear, intrusive thoughts, social withdrawal, shame, and anxiety.
Most of the study participants had scores suggestive of a minor psychiatric disorder (89%) or post-traumatic stress disorder (79%), with similar results in all groups, according to the researchers.
“This study demonstrates that extragenital physical assaults coincident with the sexual assault are more commonly perpetrated by intimate assailants than either strangers or acquaintances,” the researchers wrote. “Additionally, there is no difference in the victim’s psychologic symptoms nor in the reaction to reports of the assault to family and friends.”
The researchers reported having no financial disclosures.
Sexual assault by an intimate partner was associated with a similar level of psychological distress and a greater likelihood of extragenital traumatic injuries, compared with sexual assaults by a stranger or an acquaintance.
Dr. Patrick Chariot of the department of forensic medicine at Hôpital Jean-Verdier in Bondy, France, and his colleagues conducted an observational and prospective study to compare the psychological and physical symptoms of women assaulted by intimate partners with the symptoms of women who were assaulted by an acquaintance or unknown person.
Participants included girls and women aged 15 years or older who were referred to a sexual assault center and received examination. Data was grouped according to the woman’s reported relationship to their assailant as a current or former intimate partner, stranger, or acquaintance (Obstet Gynecol. 2016 Feb;127:516–26).
A total of 767 patients were included in the study. Assault by an intimate partner was reported by 263 women, assault by an acquaintance by 229 women, and assault by a stranger by 275 women. A 1-month follow-up examination was performed in 38% of participants.
Nearly half of the study participants reported a previous physical or sexual assault. A history of previous assault was more often reported by women in the intimate partner group (71%), compared with those in the acquaintance group (49%) and those in the stranger group (28%).
Extragenital trauma was more common in women assaulted by intimate partners (52% versus 33% and 43%, in acquaintance and strangers, respectively).
The most common psychological symptoms reported at the time of examination included anxiety, fear, shame, and sadness with reports of fear being more common in those with an intimate partner assault (46% versus 30% and 25%). The most common symptoms reported at follow-up included sleep disorders, depression, fear, intrusive thoughts, social withdrawal, shame, and anxiety.
Most of the study participants had scores suggestive of a minor psychiatric disorder (89%) or post-traumatic stress disorder (79%), with similar results in all groups, according to the researchers.
“This study demonstrates that extragenital physical assaults coincident with the sexual assault are more commonly perpetrated by intimate assailants than either strangers or acquaintances,” the researchers wrote. “Additionally, there is no difference in the victim’s psychologic symptoms nor in the reaction to reports of the assault to family and friends.”
The researchers reported having no financial disclosures.
Sexual assault by an intimate partner was associated with a similar level of psychological distress and a greater likelihood of extragenital traumatic injuries, compared with sexual assaults by a stranger or an acquaintance.
Dr. Patrick Chariot of the department of forensic medicine at Hôpital Jean-Verdier in Bondy, France, and his colleagues conducted an observational and prospective study to compare the psychological and physical symptoms of women assaulted by intimate partners with the symptoms of women who were assaulted by an acquaintance or unknown person.
Participants included girls and women aged 15 years or older who were referred to a sexual assault center and received examination. Data was grouped according to the woman’s reported relationship to their assailant as a current or former intimate partner, stranger, or acquaintance (Obstet Gynecol. 2016 Feb;127:516–26).
A total of 767 patients were included in the study. Assault by an intimate partner was reported by 263 women, assault by an acquaintance by 229 women, and assault by a stranger by 275 women. A 1-month follow-up examination was performed in 38% of participants.
Nearly half of the study participants reported a previous physical or sexual assault. A history of previous assault was more often reported by women in the intimate partner group (71%), compared with those in the acquaintance group (49%) and those in the stranger group (28%).
Extragenital trauma was more common in women assaulted by intimate partners (52% versus 33% and 43%, in acquaintance and strangers, respectively).
The most common psychological symptoms reported at the time of examination included anxiety, fear, shame, and sadness with reports of fear being more common in those with an intimate partner assault (46% versus 30% and 25%). The most common symptoms reported at follow-up included sleep disorders, depression, fear, intrusive thoughts, social withdrawal, shame, and anxiety.
Most of the study participants had scores suggestive of a minor psychiatric disorder (89%) or post-traumatic stress disorder (79%), with similar results in all groups, according to the researchers.
“This study demonstrates that extragenital physical assaults coincident with the sexual assault are more commonly perpetrated by intimate assailants than either strangers or acquaintances,” the researchers wrote. “Additionally, there is no difference in the victim’s psychologic symptoms nor in the reaction to reports of the assault to family and friends.”
The researchers reported having no financial disclosures.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Intimate partner sexual assault had similar psychological distress and lack of support as those assaulted by a stranger or acquaintance.
Major finding: Extragenital trauma was more common in women assaulted by an intimate partner (52% vs. 33% and 43%, in acquaintance and strangers, respectively).
Data source: An observational and prospective study of 767 women aged 15 years or older who were referred to a sexual assault center.
Disclosures: The researchers reported having no financial disclosures.
Telehealth for Native Americans With PTSD
Native American veterans have the highest rate of posttraumatic stress disorder (PTSD) of any ethnic group and often face significant barriers to care, such as living in rural and remote areas. To help overcome those barriers, the Office of Rural Health (ORH) established its Native Domain, a national resource on health care issues for rural Native veterans.
The Native Domain’s Telemental Health Services provides ongoing mental health care—including medication management, case management, and individual, group, and family psychotherapy—via videoconferencing to veterans who live on or near rural American Indian reservations in Montana, Wyoming, and South Dakota.
This “unique service within the VA system” demonstrates an “innovative model,” according to the ORH. Studies have suggested that telemental health is as effective as face-to-face services. For example, a 2010 study of 125 veterans with PTSD found videoconferencing an “effective and feasible” way to increase access to evidence-based care.
Clinic staff include VA-employed Tribal Outreach Workers (TOWs), who are usually military veterans and members of the tribes they serve. Their varied duties range from helping Native veterans enroll in the clinic programs, showing patients how to use the videoconferencing equipment, troubleshooting clinic telecommunications equipment, to coordinating emergency crisis management. The TOWs also work closely with the remote clinicians and provide guidance on cultural and community issues that may be relevant to the patient’s care or treatment.
The American Indian Telemental Health video (www.ruralhealth.va.gov/media/american-indian-telemental-health.asx) is an overview of a series of clinics that use videoconferencing. More information on the telemental health clinics is available at www.ruralhealth.va.gov/native/programs/telemental-services.asp.
Native American veterans have the highest rate of posttraumatic stress disorder (PTSD) of any ethnic group and often face significant barriers to care, such as living in rural and remote areas. To help overcome those barriers, the Office of Rural Health (ORH) established its Native Domain, a national resource on health care issues for rural Native veterans.
The Native Domain’s Telemental Health Services provides ongoing mental health care—including medication management, case management, and individual, group, and family psychotherapy—via videoconferencing to veterans who live on or near rural American Indian reservations in Montana, Wyoming, and South Dakota.
This “unique service within the VA system” demonstrates an “innovative model,” according to the ORH. Studies have suggested that telemental health is as effective as face-to-face services. For example, a 2010 study of 125 veterans with PTSD found videoconferencing an “effective and feasible” way to increase access to evidence-based care.
Clinic staff include VA-employed Tribal Outreach Workers (TOWs), who are usually military veterans and members of the tribes they serve. Their varied duties range from helping Native veterans enroll in the clinic programs, showing patients how to use the videoconferencing equipment, troubleshooting clinic telecommunications equipment, to coordinating emergency crisis management. The TOWs also work closely with the remote clinicians and provide guidance on cultural and community issues that may be relevant to the patient’s care or treatment.
The American Indian Telemental Health video (www.ruralhealth.va.gov/media/american-indian-telemental-health.asx) is an overview of a series of clinics that use videoconferencing. More information on the telemental health clinics is available at www.ruralhealth.va.gov/native/programs/telemental-services.asp.
Native American veterans have the highest rate of posttraumatic stress disorder (PTSD) of any ethnic group and often face significant barriers to care, such as living in rural and remote areas. To help overcome those barriers, the Office of Rural Health (ORH) established its Native Domain, a national resource on health care issues for rural Native veterans.
The Native Domain’s Telemental Health Services provides ongoing mental health care—including medication management, case management, and individual, group, and family psychotherapy—via videoconferencing to veterans who live on or near rural American Indian reservations in Montana, Wyoming, and South Dakota.
This “unique service within the VA system” demonstrates an “innovative model,” according to the ORH. Studies have suggested that telemental health is as effective as face-to-face services. For example, a 2010 study of 125 veterans with PTSD found videoconferencing an “effective and feasible” way to increase access to evidence-based care.
Clinic staff include VA-employed Tribal Outreach Workers (TOWs), who are usually military veterans and members of the tribes they serve. Their varied duties range from helping Native veterans enroll in the clinic programs, showing patients how to use the videoconferencing equipment, troubleshooting clinic telecommunications equipment, to coordinating emergency crisis management. The TOWs also work closely with the remote clinicians and provide guidance on cultural and community issues that may be relevant to the patient’s care or treatment.
The American Indian Telemental Health video (www.ruralhealth.va.gov/media/american-indian-telemental-health.asx) is an overview of a series of clinics that use videoconferencing. More information on the telemental health clinics is available at www.ruralhealth.va.gov/native/programs/telemental-services.asp.
Think beyond prazosin when treating nightmares in PTSD
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
Evaluation of Clonidine and Prazosin for the Treatment of Nighttime Posttraumatic Stress Disorder Symptoms
Posttraumatic stress disorder (PTSD) remains a significant health concern in veterans and military personnel. Whereas the lifetime incidence of PTSD in the U.S. general population is about 7% to 8%, the estimated prevalence of PTSD in deployed U.S. military personnel is higher than the national average, ranging from 11% to 17%.1,2 These numbers may be even higher, depending on the branch of service, responsibilities within the military, and specific conflict in which the veteran served. For example, one study found that 31% of Vietnam veterans have PTSD, and another recent study has reported PTSD in 28.7% of veterans returning from military service in Iraq and Afghanistan.3,4
Posttraumatic stress disorder treatment guidelines from both the American Psychiatric Association and the VA and DoD recommend the use of selective serotonin reuptake inhibitors (SSRIs) or serotonin-norepinephrine reuptake inhibitors (SNRIs) as first-line pharmacotherapy for PTSD.5,6 However, SSRIs and SNRIs seem to be largely ineffective for the management of nighttime PTSD symptoms, such as insomnia and nightmares.7,8
Related: PTSD Increases Chance of Heart Failure
Researchers hypothesize that the sympathetic nervous system plays a significant role in the hyperarousal component of nighttime PTSD. The heightened responsiveness and disruption in restorative sleep seen in PTSD have been attributed to increased activity of norepinephrine in the central nervous system.9 Mechanistically, therapies that attenuate the increased noradrenergic signaling might be effective in the management of nighttime PTSD symptoms.
The body of evidence for the use of adrenergic agents for nighttime PTSD symptoms is growing. Prazosin, a peripherally acting α1-adrenergic receptor antagonist, has recently been demonstrated to be effective for nighttime PTSD symptoms in veterans in a series of small, randomized controlled trials.10-12 Data to support the use of clonidine, a centrally acting α2-adrenergic receptor agonist, are generally limited, with the most compelling data coming from a population of civilian Cambodian refugees.13,14 A 2007 article by Boehnlein and Kinzie includes a thorough review of the preclinical research, case reports, and early clinical studies that have led to the widespread use of these agents for PTSD despite the lack of FDA approval for this indication.13A previous retrospective review by Byers and colleagues compared the effectiveness and tolerability of prazosin and quetiapine for nighttime PTSD symptoms in veterans.15 The results of that review suggest that α1-adrenergic agents may be equally effective and better tolerated than alternative medication options (ie, atypical antipsychotics) for this purpose. The present study was adapted from this design to report concurrently on the real-world use of clonidine and prazosin for the treatment of nighttime PTSD symptoms.
Study Objectives
The primary objective of this retrospective chart review was to describe the experience of patients prescribed clonidine or prazosin for the treatment of nighttime PTSD symptoms, including initial effectiveness. The primary endpoint of initial drug effectiveness was documented improvement of nighttime PTSD symptoms in the patient’s chart within 6 months of the date of first prescription. Clonidine or prazosin was categorized as initially effective if a statement such as “frequency of nightmares decreased” or “patient’s nighttime PTSD symptoms have improved” was made within 6 months after initial prescription of the drug.
The secondary objectives of this study were to evaluate the long-term effectiveness and tolerability of prazosin. The endpoints used to assess these outcomes were the 2-year continuation rates of clonidine and prazosin (as a surrogate marker for long-term effectiveness) and the documented reasons for discontinuation of clonidine and prazosin for the treatment of nighttime PTSD symptoms (in order to assess tolerability).
Methods
An electronic database search was conducted to identify the VA Portland Health Care System (VAPHCS) patients with a diagnosis of PTSD who received a first prescription for clonidine or prazosin for nighttime PTSD symptoms from a VAPHCS mental health provider or primary care provider (PCP) from January 1, 2009, to December 31, 2011. Patients were excluded if they had any history of prior use of the drug being initiated, were co-initiated on both clonidine and prazosin (defined as starting the drugs within 30 days of each other), or had a concomitant diagnosis of schizophrenia, bipolar disorder, psychotic disorder, or cognitive disorder as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition. Patients with traumatic brain injury (TBI) were excluded only if it could be determined that the event had resulted in lasting cognitive impairment.
Study Population
All patients with a diagnosis of PTSD who received a first prescription for clonidine during the period specified were screened for inclusion; patients with PTSD who were first prescribed prazosin during the same period were randomly sampled to equalize patient populations. This was done to maximize the data set while examining groups of roughly equal size for each drug, as prazosin is used much more commonly than clonidine for nighttime PTSD symptoms at VAPHCS. The patients in each resulting group were screened to determine whether they met inclusion and exclusion criteria. All subjects included were followed for 2 years from the date of the initial prescription.
Study Design
Initial effectiveness of each agent was determined by reviewing subjects’ progress notes after the initial prescription of clonidine or prazosin for documentation of improvement in symptoms within 6 months of the prescription start date. A decrease in frequency or intensity of nighttime PTSD symptoms, nightmares, or insomnia, as documented in the patient chart, was interpreted as improvement of symptoms.
Long-term continuation was assessed by reviewing subjects’ prescription records, to determine whether prescription(s) for clonidine or prazosin continued for 2 years after the date of the initial prescription.
Any gap between medication fills that resulted in an anticipated period without medication of ≥ 6 months (eg, 9 months after receiving a 90-day supply) was considered discontinuation of therapy. Prescription refill history was also reviewed, and medication possession ratio (MPR) was calculated to assess whether patients were adherent to the study drug as prescribed. Adherence was defined as an MPR of ≥ 80%. Patients who left the VAPHCS service area but continued to receive care at another VA were assessed for continuation of therapy, but refill data and/or MPR were not assessed.
Tolerability was assessed by reviewing subjects’ medical records to determine whether therapy with clonidine or prazosin was discontinued due to documented adverse effects (AEs). The occurrence of AEs was determined by reviewing progress notes and other chart documentation surrounding the date of discontinuation. If the drug was discontinued but the reason was not explicitly documented or if the prescription expired without a documented reason for nonrenewing, the reason for discontinuation was coded as “not specified.” Discontinuation due to treatment failure, change in symptoms, nonadherence, or other causes was also recorded. If multiple reasons for discontinuation were cited for a single patient, all were included in the data. This project was approved by the institutional review board at the VAPHCS.
Related:Depression and Substance Abuse Intensify Suicide Risk
Statistical Considerations
Based on clinical experience, it was presumed that many of the patients who were prescribed clonidine would be receiving it as a second-line therapy after failing prazosin. Therefore, statistical analysis of the relative effectiveness and tolerability of clonidine and prazosin could not be performed. Neither power nor sample size needed to demonstrate any difference in effectiveness or tolerability between the groups was calculated. All results are expressed using descriptive statistics.
Results
An initial database search for patients with PTSD who received a first prescription for clonidine between January 1, 2009, and December 31, 2011, from a VAPHCS provider yielded a list of 149 patients. The same search criteria applied for prazosin yielded 1,116 patients, 149 of whom were randomly selected for screening. After screening, 42 patients on clonidine and 60 patients on prazosin were included in this analysis (Figure).
Patient Demographics
The average age of the clonidine patients was 38.5 years (range 21-65 years) (Table 1). The clonidine group was primarily male (90%) and white (83%). Eighteen of the 42 patients in the clonidine group had a baseline PTSD Checklist-Civilian version (PCL-C) score available within the 90 days before the first prescription of clonidine; the average baseline PCL-C score in this subgroup was 62 ± 12.0 (median 65.5, range 31-82). Most of the clonidine patients (71%) had a concomitant diagnosis of a depressive disorder. About one-quarter of the group (24%) had previously tried prazosin per prescription records. In 24 patients (57%), the first prescription for clonidine was written by a psychiatrist or psychiatric nurse practitioner; 18 patients (43%) were started on clonidine by PCPs.
The average age of the prazosin patients was 46.1 years (range 21-74 years). The prazosin group was also primarily male (93%) and white (88%). Twenty of the 60 patients in the prazosin group had a baseline PCL-C score available within the 90 days before the first prescription of prazosin; the average baseline PCL-C score in this subgroup was 55 ± 16.1 (median 64, range 30-72). Most of the prazosin patients (63%) had a concomitant diagnosis of a depressive disorder. Four patients (7%) had previously tried clonidine per prescription records. In 35 patients (58%), the first prescription for prazosin was written by a psychiatrist or psychiatric nurse practitioner; 25 patients (42%) were started on prazosin by PCPs.
Data pertaining to initial and long-term effectiveness, tolerability, and MPR for both clonidine and prazosin are presented in Table 2.
Clonidine
Of the 42 clonidine patients assessed, 24 (57%) had a positive response to the medication for nighttime PTSD symptoms documented in the Computerized Patient Record System (CPRS) within 6 months of starting therapy. Six months after starting clonidine, 23 patients (55%) continued to take clonidine. Two years after starting therapy, 8 of the original 42 patients continued on clonidine for an overall 2-year continuation rate of 19%.
Tolerability
Of the 34 patients who discontinued clonidine within 2 years, 13 patients (38%) cited ineffectiveness of therapy as a reason for discontinuation. Another 13 patients (38%) reported discontinuing therapy due to AEs. Sedation (4 patients, 12%), dizziness/hypotension (3 patients, 9%), and paradoxical worsening of PTSD symptoms (4 patients, 12%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were syncope (2 patients), erectile dysfunction (1 patient), rash (1 patient), myoclonus (1 patient), increased depression (1 patient), and fatigue (1 patient). One patient reported that he had discontinued clonidine due to symptom resolution/lack of need for treatment. In 8 of the 34 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 21 evaluable patients who continued to receive clonidine 6 months after initiation, 10 (48%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Six of the 21 patients (29%) had an MPR between 50% and 79%, and 5 patients (24%) had an MPR < 50%.
Of the 8 patients who continued on clonidine at the 2-year mark, 3 (38%) were adherent to therapy, with an MPR of ≥ 80%. Three more patients (38%) had a 2-year MPR between 50% and 80%, and 2 patients (25%) had an MPR < 50%.
Prazosin
Of the 60 prazosin patients assessed, 32 (53%) had a positive response to the medication for nighttime PTSD symptoms documented in the CPRS within 6 months of starting therapy. Six months after starting prazosin, 36 patients (60%) continued to take prazosin. Two years after starting therapy, 18 of the original 60 patients continued on prazosin for an overall 2-year continuation rate of 30%.
Tolerability
Of the 42 patients who discontinued prazosin within 2 years, six patients (14%) cited ineffectiveness of therapy as a reason for discontinuation. Thirteen patients (31%) reported discontinuing therapy due to AEs. Sedation (3 patients, 7%), dizziness/hypotension (3 patients, 7%), and paradoxical worsening of PTSD symptoms (6 patients, 14%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were headache (2 patients), altered mental status (1 patient), and fatigue (1 patient). Three patients reported that they had discontinued clonidine due to symptom resolution/lack of need for treatment. Other reasons for discontinuation not related to AEs included flight rules (1 patient), changes to antihypertensive regimen (1 patient), refill issues (1 patient), and cost (1 patient). In 15 of the 42 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 31 evaluable patients who continued to receive prazosin 6 months after initiation, 20 (65%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Five of the 31 patients (16%) had an MPR between 50% and 80%, and 6 patients (19%) had an MPR < 50%.
Of the 15 evaluable patients who continued on prazosin at the 2-year mark, 9 (60%) were adherent to therapy, with an MPR of ≥ 80%. Three patients (20%) had a 2-year MPR between 50% and 80%, and 3 patients (20%) had an MPR < 50%.
Discussion
Although prazosin has been shown to be effective for nighttime PTSD symptoms in both prospective and retrospective evaluations in veterans, this study provides the first evidence to support the use of clonidine in a veteran population.10-12,15
Interestingly, 42% of the patients assessed received their first prescription of an α2-adrenergic agent for nighttime PTSD symptoms from a PCP. Even with the recent increased focus on integrating mental health into primary care within the VA, this was a surprising finding. Primary care providers at VAPHCS may have a greater role in the outpatient management of PTSD than previously suspected. The information presented here may prove useful and applicable in both psychiatric and primary care treatment settings.
The study results indicated that a majority of subjects initially reported effectiveness with either clonidine or prazosin (53% and 57%, respectively). The initial effectiveness rate for prazosin is similar to those described in previous studies.10-13,15 The data also support a viable role for clonidine in the treatment of nighttime PTSD symptoms.
Regardless of initial improvement, the study results also suggest that the therapeutic benefit may not persist in the long term, as evidenced by a significant percentage of discontinuations attributed to ineffectiveness (38% for clonidine and 14% for prazosin) and a very low rate of long-term continuation (19% for clonidine and 30% for prazosin at 2 years). This latter observation contrasts with findings from previous studies; Byers and colleagues reported a 2-year prazosin continuation rate of 48.4% in a similar analysis, and Boehnlein and colleagues reported a sustained benefit of clonidine in responders over a 10-year period.14,15 The wide variety of reasons for discontinuation reported here may help providers who are considering clonidine or prazosin for their patients to anticipate barriers to long-term success.
Part of the discrepancy between these results and previously reported successes with clonidine and prazosin may be attributable to the classic issue of efficacy vs effectiveness. Many of the studies that have informed us on the efficacy and tolerability of prazosin for nighttime PTSD symptoms described outcomes of prospective clinical research. Furthermore, these prospective trials were limited to < 6 months in duration. To date, neither clonidine nor prazosin has been evaluated for long-term efficacy and effectiveness in well-designed, prospective trials. This retrospective analysis may help provide a realistic estimate of the long-term effectiveness of these therapies, especially within the veteran population.
Limitations
This was a single-center, retrospective study conducted primarily in white male patients. Although likely applicable to the U.S. veteran population at large, these data may be poorly generalizable to patient populations outside the VA health care system.
Aside from external validity, this study has several significant limitations. The primary limitation of this project is that it was not designed to allow for statistical comparison of clonidine and prazosin. Such an analysis would have better defined the role of clonidine in PTSD treatment, either by establishing similar effectiveness of clonidine and prazosin for nighttime symptoms or by providing evidence of the superiority of one over the other. In designing the project, investigators suspected based on experience that the majority of patients prescribed clonidine would receive the drug after having already failed first-line therapy with prazosin. Had this been the case, a direct comparison may have been biased in favor of prazosin. In retrospect, however, only 24% of the clonidine group had previously been prescribed prazosin, and only 7% of the prazosin group had been prescribed clonidine. This suggests that clonidine may be used first line more often than the investigators anticipated and that a future direct comparison would be worthwhile.
Second, the subjective data collected for this project required investigators to read and interpret chart notes, although the review of all records by a single investigator helped limit variability in interpretation. At times, information in the CPRS was incomplete in terms of determining continuation of therapy or cause for discontinuation.
Third, although it is implied that a significant number of veterans have combat-related PTSD, the nature of the traumatic event(s) leading to PTSD was not recorded in this study, and no subgroup analysis was done to compare the effect of α2-adrenergic agents between combat- and noncombat-related PTSD. Owing to their exclusion by design, it is also difficult to apply these results to veterans who have lasting cognitive impairment as a result of TBI, who are presumably among those most likely to have experienced traumas that could provoke PTSD.
The design of this project also did not include a subgroup analysis based on antidepressant type, and it is unclear whether the potential pharmacodynamic interaction between noradrenergic antidepressants (ie, SNRIs) and anti– α2-adrenergic agents had any impact on clinical outcomes. The use of complementary nonpharmacologic treatment modalities (ie, psychotherapy, eye movement desensitization and reprocessing) was also not evaluated.
Related: Female Service Members in the Long War
Finally, the primary outcome of patient-reported improvement in symptoms does not provide information on the magnitude or specific nature of benefits derived. Given the retrospective nature, data used in prospectively designed studies (eg, rating scales pertinent to PTSD), which might have helped to quantify the benefit of treatment, was not consistently available. Even a baseline PCL-C score, collected in order to describe the patient population, was available only in 37% of the patients assessed. Furthermore, nighttime PTSD symptoms vary among individuals, but the primary outcome of this study pools any benefits seen in areas such as nightmares, awakenings, night sweats, or sleep quality into a single outcome of symptom improvement.
Conclusions
This study indicates that both clonidine and prazosin may be effective for the treatment of nighttime PTSD symptoms in the veteran population but that their long-term utility may be limited by waning effectiveness, tolerability, and adherence issues. At this time, it is unclear whether either agent has an advantage over the other in terms of effectiveness or tolerability; further studies are needed to address that question.
Despite its limitations, the authors anticipate that this study will provide information regarding the effectiveness and tolerability of clonidine and prazosin to treat nighttime PTSD symptoms. Findings from this study may help clinicians to anticipate the needs and challenges of patients using β2-adrenergic agents for nighttime symptoms of PTSD.
Acknowledgements
The authors wish to acknowledge Brian Wilcox, PharmD, for his assistance in generating patient data reports, and Ronald Brown, RPh, MS, for his guidance regarding data analysis.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
2. Gates MA, Holowka DW, Vasterling JJ, Keane TM, Marx BP, Rosen RC. Posttraumatic stress disorder in veterans and military personnel: epidemiology, screening, and case recognition. Psychol Serv. 2012;9(4):361-382.
3. Kulka R, Schlenger WE, Fairbanks J, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. New York, NY: Brunnel/Mazel; 1990.
4. Barrera TL, Graham DP, Dunn NJ, Teng EJ. Influence of trauma history on panic and posttraumatic stress disorder in returning veterans. Psychol Serv. 2013;10(2):168-176.
5. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Acute Stress Disorder and Posttraumatic Stress Disorder. Arlington, VA: American Psychiatric Association; 2004.
6. U.S. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for management of post-traumatic stress. Version 2.0. U.S. Department of Veterans Affairs Website. http://www.healthquality.va.gov/guidelines/MH/ptsd/cpgPTSDFULL201011612c.pdf. Published October 2010. Accessed October 5, 2015.
7. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
8. Ravindran LN, Stein MB. Pharmacotherapy of post-traumatic stress disorder. In: Stein MB, Steckler T, eds. Behavioral Neurobiology of Anxiety and Its Treatment. Vol 2. Heidelberg, Germany: Springer; 2010:505-525.
9. Spoormaker VI, Montgomery P. Disturbed sleep in post-traumatic stress disorder: secondary symptom or core feature? Sleep Med Rev. 2008;12(3):169-184.
10. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo controlled study. Am J Psychiatry. 2003;160(2):371-373.
11. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
12. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170:1003-1010.
13. Boehnlein JK, Kinzie JD. Pharmacologic reduction of CNS noradrenergic activity in PTSD: the case for clonidine and prazosin. J Psychiatr Pract. 2007;13(2):72-78.
14. Boehnlein JK, Kinzie JD, Sekiya U, Riley C, Pou K, Rosborough B. A ten-year treatment outcome study of traumatized Cambodian refugees. J Nerve Ment Dis. 2004;192(10):658-663.
15. Byers MG, Allison KM, Wendel CS, Lee JK. Prazosin versus quetiapine for nighttime posttraumatic stress disorder symptoms in veterans: an assessment of long-term comparative effectiveness and safety. J Clin Psychopharmacol. 2010;30(3):225-229.
Posttraumatic stress disorder (PTSD) remains a significant health concern in veterans and military personnel. Whereas the lifetime incidence of PTSD in the U.S. general population is about 7% to 8%, the estimated prevalence of PTSD in deployed U.S. military personnel is higher than the national average, ranging from 11% to 17%.1,2 These numbers may be even higher, depending on the branch of service, responsibilities within the military, and specific conflict in which the veteran served. For example, one study found that 31% of Vietnam veterans have PTSD, and another recent study has reported PTSD in 28.7% of veterans returning from military service in Iraq and Afghanistan.3,4
Posttraumatic stress disorder treatment guidelines from both the American Psychiatric Association and the VA and DoD recommend the use of selective serotonin reuptake inhibitors (SSRIs) or serotonin-norepinephrine reuptake inhibitors (SNRIs) as first-line pharmacotherapy for PTSD.5,6 However, SSRIs and SNRIs seem to be largely ineffective for the management of nighttime PTSD symptoms, such as insomnia and nightmares.7,8
Related: PTSD Increases Chance of Heart Failure
Researchers hypothesize that the sympathetic nervous system plays a significant role in the hyperarousal component of nighttime PTSD. The heightened responsiveness and disruption in restorative sleep seen in PTSD have been attributed to increased activity of norepinephrine in the central nervous system.9 Mechanistically, therapies that attenuate the increased noradrenergic signaling might be effective in the management of nighttime PTSD symptoms.
The body of evidence for the use of adrenergic agents for nighttime PTSD symptoms is growing. Prazosin, a peripherally acting α1-adrenergic receptor antagonist, has recently been demonstrated to be effective for nighttime PTSD symptoms in veterans in a series of small, randomized controlled trials.10-12 Data to support the use of clonidine, a centrally acting α2-adrenergic receptor agonist, are generally limited, with the most compelling data coming from a population of civilian Cambodian refugees.13,14 A 2007 article by Boehnlein and Kinzie includes a thorough review of the preclinical research, case reports, and early clinical studies that have led to the widespread use of these agents for PTSD despite the lack of FDA approval for this indication.13A previous retrospective review by Byers and colleagues compared the effectiveness and tolerability of prazosin and quetiapine for nighttime PTSD symptoms in veterans.15 The results of that review suggest that α1-adrenergic agents may be equally effective and better tolerated than alternative medication options (ie, atypical antipsychotics) for this purpose. The present study was adapted from this design to report concurrently on the real-world use of clonidine and prazosin for the treatment of nighttime PTSD symptoms.
Study Objectives
The primary objective of this retrospective chart review was to describe the experience of patients prescribed clonidine or prazosin for the treatment of nighttime PTSD symptoms, including initial effectiveness. The primary endpoint of initial drug effectiveness was documented improvement of nighttime PTSD symptoms in the patient’s chart within 6 months of the date of first prescription. Clonidine or prazosin was categorized as initially effective if a statement such as “frequency of nightmares decreased” or “patient’s nighttime PTSD symptoms have improved” was made within 6 months after initial prescription of the drug.
The secondary objectives of this study were to evaluate the long-term effectiveness and tolerability of prazosin. The endpoints used to assess these outcomes were the 2-year continuation rates of clonidine and prazosin (as a surrogate marker for long-term effectiveness) and the documented reasons for discontinuation of clonidine and prazosin for the treatment of nighttime PTSD symptoms (in order to assess tolerability).
Methods
An electronic database search was conducted to identify the VA Portland Health Care System (VAPHCS) patients with a diagnosis of PTSD who received a first prescription for clonidine or prazosin for nighttime PTSD symptoms from a VAPHCS mental health provider or primary care provider (PCP) from January 1, 2009, to December 31, 2011. Patients were excluded if they had any history of prior use of the drug being initiated, were co-initiated on both clonidine and prazosin (defined as starting the drugs within 30 days of each other), or had a concomitant diagnosis of schizophrenia, bipolar disorder, psychotic disorder, or cognitive disorder as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition. Patients with traumatic brain injury (TBI) were excluded only if it could be determined that the event had resulted in lasting cognitive impairment.
Study Population
All patients with a diagnosis of PTSD who received a first prescription for clonidine during the period specified were screened for inclusion; patients with PTSD who were first prescribed prazosin during the same period were randomly sampled to equalize patient populations. This was done to maximize the data set while examining groups of roughly equal size for each drug, as prazosin is used much more commonly than clonidine for nighttime PTSD symptoms at VAPHCS. The patients in each resulting group were screened to determine whether they met inclusion and exclusion criteria. All subjects included were followed for 2 years from the date of the initial prescription.
Study Design
Initial effectiveness of each agent was determined by reviewing subjects’ progress notes after the initial prescription of clonidine or prazosin for documentation of improvement in symptoms within 6 months of the prescription start date. A decrease in frequency or intensity of nighttime PTSD symptoms, nightmares, or insomnia, as documented in the patient chart, was interpreted as improvement of symptoms.
Long-term continuation was assessed by reviewing subjects’ prescription records, to determine whether prescription(s) for clonidine or prazosin continued for 2 years after the date of the initial prescription.
Any gap between medication fills that resulted in an anticipated period without medication of ≥ 6 months (eg, 9 months after receiving a 90-day supply) was considered discontinuation of therapy. Prescription refill history was also reviewed, and medication possession ratio (MPR) was calculated to assess whether patients were adherent to the study drug as prescribed. Adherence was defined as an MPR of ≥ 80%. Patients who left the VAPHCS service area but continued to receive care at another VA were assessed for continuation of therapy, but refill data and/or MPR were not assessed.
Tolerability was assessed by reviewing subjects’ medical records to determine whether therapy with clonidine or prazosin was discontinued due to documented adverse effects (AEs). The occurrence of AEs was determined by reviewing progress notes and other chart documentation surrounding the date of discontinuation. If the drug was discontinued but the reason was not explicitly documented or if the prescription expired without a documented reason for nonrenewing, the reason for discontinuation was coded as “not specified.” Discontinuation due to treatment failure, change in symptoms, nonadherence, or other causes was also recorded. If multiple reasons for discontinuation were cited for a single patient, all were included in the data. This project was approved by the institutional review board at the VAPHCS.
Related:Depression and Substance Abuse Intensify Suicide Risk
Statistical Considerations
Based on clinical experience, it was presumed that many of the patients who were prescribed clonidine would be receiving it as a second-line therapy after failing prazosin. Therefore, statistical analysis of the relative effectiveness and tolerability of clonidine and prazosin could not be performed. Neither power nor sample size needed to demonstrate any difference in effectiveness or tolerability between the groups was calculated. All results are expressed using descriptive statistics.
Results
An initial database search for patients with PTSD who received a first prescription for clonidine between January 1, 2009, and December 31, 2011, from a VAPHCS provider yielded a list of 149 patients. The same search criteria applied for prazosin yielded 1,116 patients, 149 of whom were randomly selected for screening. After screening, 42 patients on clonidine and 60 patients on prazosin were included in this analysis (Figure).
Patient Demographics
The average age of the clonidine patients was 38.5 years (range 21-65 years) (Table 1). The clonidine group was primarily male (90%) and white (83%). Eighteen of the 42 patients in the clonidine group had a baseline PTSD Checklist-Civilian version (PCL-C) score available within the 90 days before the first prescription of clonidine; the average baseline PCL-C score in this subgroup was 62 ± 12.0 (median 65.5, range 31-82). Most of the clonidine patients (71%) had a concomitant diagnosis of a depressive disorder. About one-quarter of the group (24%) had previously tried prazosin per prescription records. In 24 patients (57%), the first prescription for clonidine was written by a psychiatrist or psychiatric nurse practitioner; 18 patients (43%) were started on clonidine by PCPs.
The average age of the prazosin patients was 46.1 years (range 21-74 years). The prazosin group was also primarily male (93%) and white (88%). Twenty of the 60 patients in the prazosin group had a baseline PCL-C score available within the 90 days before the first prescription of prazosin; the average baseline PCL-C score in this subgroup was 55 ± 16.1 (median 64, range 30-72). Most of the prazosin patients (63%) had a concomitant diagnosis of a depressive disorder. Four patients (7%) had previously tried clonidine per prescription records. In 35 patients (58%), the first prescription for prazosin was written by a psychiatrist or psychiatric nurse practitioner; 25 patients (42%) were started on prazosin by PCPs.
Data pertaining to initial and long-term effectiveness, tolerability, and MPR for both clonidine and prazosin are presented in Table 2.
Clonidine
Of the 42 clonidine patients assessed, 24 (57%) had a positive response to the medication for nighttime PTSD symptoms documented in the Computerized Patient Record System (CPRS) within 6 months of starting therapy. Six months after starting clonidine, 23 patients (55%) continued to take clonidine. Two years after starting therapy, 8 of the original 42 patients continued on clonidine for an overall 2-year continuation rate of 19%.
Tolerability
Of the 34 patients who discontinued clonidine within 2 years, 13 patients (38%) cited ineffectiveness of therapy as a reason for discontinuation. Another 13 patients (38%) reported discontinuing therapy due to AEs. Sedation (4 patients, 12%), dizziness/hypotension (3 patients, 9%), and paradoxical worsening of PTSD symptoms (4 patients, 12%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were syncope (2 patients), erectile dysfunction (1 patient), rash (1 patient), myoclonus (1 patient), increased depression (1 patient), and fatigue (1 patient). One patient reported that he had discontinued clonidine due to symptom resolution/lack of need for treatment. In 8 of the 34 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 21 evaluable patients who continued to receive clonidine 6 months after initiation, 10 (48%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Six of the 21 patients (29%) had an MPR between 50% and 79%, and 5 patients (24%) had an MPR < 50%.
Of the 8 patients who continued on clonidine at the 2-year mark, 3 (38%) were adherent to therapy, with an MPR of ≥ 80%. Three more patients (38%) had a 2-year MPR between 50% and 80%, and 2 patients (25%) had an MPR < 50%.
Prazosin
Of the 60 prazosin patients assessed, 32 (53%) had a positive response to the medication for nighttime PTSD symptoms documented in the CPRS within 6 months of starting therapy. Six months after starting prazosin, 36 patients (60%) continued to take prazosin. Two years after starting therapy, 18 of the original 60 patients continued on prazosin for an overall 2-year continuation rate of 30%.
Tolerability
Of the 42 patients who discontinued prazosin within 2 years, six patients (14%) cited ineffectiveness of therapy as a reason for discontinuation. Thirteen patients (31%) reported discontinuing therapy due to AEs. Sedation (3 patients, 7%), dizziness/hypotension (3 patients, 7%), and paradoxical worsening of PTSD symptoms (6 patients, 14%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were headache (2 patients), altered mental status (1 patient), and fatigue (1 patient). Three patients reported that they had discontinued clonidine due to symptom resolution/lack of need for treatment. Other reasons for discontinuation not related to AEs included flight rules (1 patient), changes to antihypertensive regimen (1 patient), refill issues (1 patient), and cost (1 patient). In 15 of the 42 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 31 evaluable patients who continued to receive prazosin 6 months after initiation, 20 (65%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Five of the 31 patients (16%) had an MPR between 50% and 80%, and 6 patients (19%) had an MPR < 50%.
Of the 15 evaluable patients who continued on prazosin at the 2-year mark, 9 (60%) were adherent to therapy, with an MPR of ≥ 80%. Three patients (20%) had a 2-year MPR between 50% and 80%, and 3 patients (20%) had an MPR < 50%.
Discussion
Although prazosin has been shown to be effective for nighttime PTSD symptoms in both prospective and retrospective evaluations in veterans, this study provides the first evidence to support the use of clonidine in a veteran population.10-12,15
Interestingly, 42% of the patients assessed received their first prescription of an α2-adrenergic agent for nighttime PTSD symptoms from a PCP. Even with the recent increased focus on integrating mental health into primary care within the VA, this was a surprising finding. Primary care providers at VAPHCS may have a greater role in the outpatient management of PTSD than previously suspected. The information presented here may prove useful and applicable in both psychiatric and primary care treatment settings.
The study results indicated that a majority of subjects initially reported effectiveness with either clonidine or prazosin (53% and 57%, respectively). The initial effectiveness rate for prazosin is similar to those described in previous studies.10-13,15 The data also support a viable role for clonidine in the treatment of nighttime PTSD symptoms.
Regardless of initial improvement, the study results also suggest that the therapeutic benefit may not persist in the long term, as evidenced by a significant percentage of discontinuations attributed to ineffectiveness (38% for clonidine and 14% for prazosin) and a very low rate of long-term continuation (19% for clonidine and 30% for prazosin at 2 years). This latter observation contrasts with findings from previous studies; Byers and colleagues reported a 2-year prazosin continuation rate of 48.4% in a similar analysis, and Boehnlein and colleagues reported a sustained benefit of clonidine in responders over a 10-year period.14,15 The wide variety of reasons for discontinuation reported here may help providers who are considering clonidine or prazosin for their patients to anticipate barriers to long-term success.
Part of the discrepancy between these results and previously reported successes with clonidine and prazosin may be attributable to the classic issue of efficacy vs effectiveness. Many of the studies that have informed us on the efficacy and tolerability of prazosin for nighttime PTSD symptoms described outcomes of prospective clinical research. Furthermore, these prospective trials were limited to < 6 months in duration. To date, neither clonidine nor prazosin has been evaluated for long-term efficacy and effectiveness in well-designed, prospective trials. This retrospective analysis may help provide a realistic estimate of the long-term effectiveness of these therapies, especially within the veteran population.
Limitations
This was a single-center, retrospective study conducted primarily in white male patients. Although likely applicable to the U.S. veteran population at large, these data may be poorly generalizable to patient populations outside the VA health care system.
Aside from external validity, this study has several significant limitations. The primary limitation of this project is that it was not designed to allow for statistical comparison of clonidine and prazosin. Such an analysis would have better defined the role of clonidine in PTSD treatment, either by establishing similar effectiveness of clonidine and prazosin for nighttime symptoms or by providing evidence of the superiority of one over the other. In designing the project, investigators suspected based on experience that the majority of patients prescribed clonidine would receive the drug after having already failed first-line therapy with prazosin. Had this been the case, a direct comparison may have been biased in favor of prazosin. In retrospect, however, only 24% of the clonidine group had previously been prescribed prazosin, and only 7% of the prazosin group had been prescribed clonidine. This suggests that clonidine may be used first line more often than the investigators anticipated and that a future direct comparison would be worthwhile.
Second, the subjective data collected for this project required investigators to read and interpret chart notes, although the review of all records by a single investigator helped limit variability in interpretation. At times, information in the CPRS was incomplete in terms of determining continuation of therapy or cause for discontinuation.
Third, although it is implied that a significant number of veterans have combat-related PTSD, the nature of the traumatic event(s) leading to PTSD was not recorded in this study, and no subgroup analysis was done to compare the effect of α2-adrenergic agents between combat- and noncombat-related PTSD. Owing to their exclusion by design, it is also difficult to apply these results to veterans who have lasting cognitive impairment as a result of TBI, who are presumably among those most likely to have experienced traumas that could provoke PTSD.
The design of this project also did not include a subgroup analysis based on antidepressant type, and it is unclear whether the potential pharmacodynamic interaction between noradrenergic antidepressants (ie, SNRIs) and anti– α2-adrenergic agents had any impact on clinical outcomes. The use of complementary nonpharmacologic treatment modalities (ie, psychotherapy, eye movement desensitization and reprocessing) was also not evaluated.
Related: Female Service Members in the Long War
Finally, the primary outcome of patient-reported improvement in symptoms does not provide information on the magnitude or specific nature of benefits derived. Given the retrospective nature, data used in prospectively designed studies (eg, rating scales pertinent to PTSD), which might have helped to quantify the benefit of treatment, was not consistently available. Even a baseline PCL-C score, collected in order to describe the patient population, was available only in 37% of the patients assessed. Furthermore, nighttime PTSD symptoms vary among individuals, but the primary outcome of this study pools any benefits seen in areas such as nightmares, awakenings, night sweats, or sleep quality into a single outcome of symptom improvement.
Conclusions
This study indicates that both clonidine and prazosin may be effective for the treatment of nighttime PTSD symptoms in the veteran population but that their long-term utility may be limited by waning effectiveness, tolerability, and adherence issues. At this time, it is unclear whether either agent has an advantage over the other in terms of effectiveness or tolerability; further studies are needed to address that question.
Despite its limitations, the authors anticipate that this study will provide information regarding the effectiveness and tolerability of clonidine and prazosin to treat nighttime PTSD symptoms. Findings from this study may help clinicians to anticipate the needs and challenges of patients using β2-adrenergic agents for nighttime symptoms of PTSD.
Acknowledgements
The authors wish to acknowledge Brian Wilcox, PharmD, for his assistance in generating patient data reports, and Ronald Brown, RPh, MS, for his guidance regarding data analysis.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Posttraumatic stress disorder (PTSD) remains a significant health concern in veterans and military personnel. Whereas the lifetime incidence of PTSD in the U.S. general population is about 7% to 8%, the estimated prevalence of PTSD in deployed U.S. military personnel is higher than the national average, ranging from 11% to 17%.1,2 These numbers may be even higher, depending on the branch of service, responsibilities within the military, and specific conflict in which the veteran served. For example, one study found that 31% of Vietnam veterans have PTSD, and another recent study has reported PTSD in 28.7% of veterans returning from military service in Iraq and Afghanistan.3,4
Posttraumatic stress disorder treatment guidelines from both the American Psychiatric Association and the VA and DoD recommend the use of selective serotonin reuptake inhibitors (SSRIs) or serotonin-norepinephrine reuptake inhibitors (SNRIs) as first-line pharmacotherapy for PTSD.5,6 However, SSRIs and SNRIs seem to be largely ineffective for the management of nighttime PTSD symptoms, such as insomnia and nightmares.7,8
Related: PTSD Increases Chance of Heart Failure
Researchers hypothesize that the sympathetic nervous system plays a significant role in the hyperarousal component of nighttime PTSD. The heightened responsiveness and disruption in restorative sleep seen in PTSD have been attributed to increased activity of norepinephrine in the central nervous system.9 Mechanistically, therapies that attenuate the increased noradrenergic signaling might be effective in the management of nighttime PTSD symptoms.
The body of evidence for the use of adrenergic agents for nighttime PTSD symptoms is growing. Prazosin, a peripherally acting α1-adrenergic receptor antagonist, has recently been demonstrated to be effective for nighttime PTSD symptoms in veterans in a series of small, randomized controlled trials.10-12 Data to support the use of clonidine, a centrally acting α2-adrenergic receptor agonist, are generally limited, with the most compelling data coming from a population of civilian Cambodian refugees.13,14 A 2007 article by Boehnlein and Kinzie includes a thorough review of the preclinical research, case reports, and early clinical studies that have led to the widespread use of these agents for PTSD despite the lack of FDA approval for this indication.13A previous retrospective review by Byers and colleagues compared the effectiveness and tolerability of prazosin and quetiapine for nighttime PTSD symptoms in veterans.15 The results of that review suggest that α1-adrenergic agents may be equally effective and better tolerated than alternative medication options (ie, atypical antipsychotics) for this purpose. The present study was adapted from this design to report concurrently on the real-world use of clonidine and prazosin for the treatment of nighttime PTSD symptoms.
Study Objectives
The primary objective of this retrospective chart review was to describe the experience of patients prescribed clonidine or prazosin for the treatment of nighttime PTSD symptoms, including initial effectiveness. The primary endpoint of initial drug effectiveness was documented improvement of nighttime PTSD symptoms in the patient’s chart within 6 months of the date of first prescription. Clonidine or prazosin was categorized as initially effective if a statement such as “frequency of nightmares decreased” or “patient’s nighttime PTSD symptoms have improved” was made within 6 months after initial prescription of the drug.
The secondary objectives of this study were to evaluate the long-term effectiveness and tolerability of prazosin. The endpoints used to assess these outcomes were the 2-year continuation rates of clonidine and prazosin (as a surrogate marker for long-term effectiveness) and the documented reasons for discontinuation of clonidine and prazosin for the treatment of nighttime PTSD symptoms (in order to assess tolerability).
Methods
An electronic database search was conducted to identify the VA Portland Health Care System (VAPHCS) patients with a diagnosis of PTSD who received a first prescription for clonidine or prazosin for nighttime PTSD symptoms from a VAPHCS mental health provider or primary care provider (PCP) from January 1, 2009, to December 31, 2011. Patients were excluded if they had any history of prior use of the drug being initiated, were co-initiated on both clonidine and prazosin (defined as starting the drugs within 30 days of each other), or had a concomitant diagnosis of schizophrenia, bipolar disorder, psychotic disorder, or cognitive disorder as defined in the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition. Patients with traumatic brain injury (TBI) were excluded only if it could be determined that the event had resulted in lasting cognitive impairment.
Study Population
All patients with a diagnosis of PTSD who received a first prescription for clonidine during the period specified were screened for inclusion; patients with PTSD who were first prescribed prazosin during the same period were randomly sampled to equalize patient populations. This was done to maximize the data set while examining groups of roughly equal size for each drug, as prazosin is used much more commonly than clonidine for nighttime PTSD symptoms at VAPHCS. The patients in each resulting group were screened to determine whether they met inclusion and exclusion criteria. All subjects included were followed for 2 years from the date of the initial prescription.
Study Design
Initial effectiveness of each agent was determined by reviewing subjects’ progress notes after the initial prescription of clonidine or prazosin for documentation of improvement in symptoms within 6 months of the prescription start date. A decrease in frequency or intensity of nighttime PTSD symptoms, nightmares, or insomnia, as documented in the patient chart, was interpreted as improvement of symptoms.
Long-term continuation was assessed by reviewing subjects’ prescription records, to determine whether prescription(s) for clonidine or prazosin continued for 2 years after the date of the initial prescription.
Any gap between medication fills that resulted in an anticipated period without medication of ≥ 6 months (eg, 9 months after receiving a 90-day supply) was considered discontinuation of therapy. Prescription refill history was also reviewed, and medication possession ratio (MPR) was calculated to assess whether patients were adherent to the study drug as prescribed. Adherence was defined as an MPR of ≥ 80%. Patients who left the VAPHCS service area but continued to receive care at another VA were assessed for continuation of therapy, but refill data and/or MPR were not assessed.
Tolerability was assessed by reviewing subjects’ medical records to determine whether therapy with clonidine or prazosin was discontinued due to documented adverse effects (AEs). The occurrence of AEs was determined by reviewing progress notes and other chart documentation surrounding the date of discontinuation. If the drug was discontinued but the reason was not explicitly documented or if the prescription expired without a documented reason for nonrenewing, the reason for discontinuation was coded as “not specified.” Discontinuation due to treatment failure, change in symptoms, nonadherence, or other causes was also recorded. If multiple reasons for discontinuation were cited for a single patient, all were included in the data. This project was approved by the institutional review board at the VAPHCS.
Related:Depression and Substance Abuse Intensify Suicide Risk
Statistical Considerations
Based on clinical experience, it was presumed that many of the patients who were prescribed clonidine would be receiving it as a second-line therapy after failing prazosin. Therefore, statistical analysis of the relative effectiveness and tolerability of clonidine and prazosin could not be performed. Neither power nor sample size needed to demonstrate any difference in effectiveness or tolerability between the groups was calculated. All results are expressed using descriptive statistics.
Results
An initial database search for patients with PTSD who received a first prescription for clonidine between January 1, 2009, and December 31, 2011, from a VAPHCS provider yielded a list of 149 patients. The same search criteria applied for prazosin yielded 1,116 patients, 149 of whom were randomly selected for screening. After screening, 42 patients on clonidine and 60 patients on prazosin were included in this analysis (Figure).
Patient Demographics
The average age of the clonidine patients was 38.5 years (range 21-65 years) (Table 1). The clonidine group was primarily male (90%) and white (83%). Eighteen of the 42 patients in the clonidine group had a baseline PTSD Checklist-Civilian version (PCL-C) score available within the 90 days before the first prescription of clonidine; the average baseline PCL-C score in this subgroup was 62 ± 12.0 (median 65.5, range 31-82). Most of the clonidine patients (71%) had a concomitant diagnosis of a depressive disorder. About one-quarter of the group (24%) had previously tried prazosin per prescription records. In 24 patients (57%), the first prescription for clonidine was written by a psychiatrist or psychiatric nurse practitioner; 18 patients (43%) were started on clonidine by PCPs.
The average age of the prazosin patients was 46.1 years (range 21-74 years). The prazosin group was also primarily male (93%) and white (88%). Twenty of the 60 patients in the prazosin group had a baseline PCL-C score available within the 90 days before the first prescription of prazosin; the average baseline PCL-C score in this subgroup was 55 ± 16.1 (median 64, range 30-72). Most of the prazosin patients (63%) had a concomitant diagnosis of a depressive disorder. Four patients (7%) had previously tried clonidine per prescription records. In 35 patients (58%), the first prescription for prazosin was written by a psychiatrist or psychiatric nurse practitioner; 25 patients (42%) were started on prazosin by PCPs.
Data pertaining to initial and long-term effectiveness, tolerability, and MPR for both clonidine and prazosin are presented in Table 2.
Clonidine
Of the 42 clonidine patients assessed, 24 (57%) had a positive response to the medication for nighttime PTSD symptoms documented in the Computerized Patient Record System (CPRS) within 6 months of starting therapy. Six months after starting clonidine, 23 patients (55%) continued to take clonidine. Two years after starting therapy, 8 of the original 42 patients continued on clonidine for an overall 2-year continuation rate of 19%.
Tolerability
Of the 34 patients who discontinued clonidine within 2 years, 13 patients (38%) cited ineffectiveness of therapy as a reason for discontinuation. Another 13 patients (38%) reported discontinuing therapy due to AEs. Sedation (4 patients, 12%), dizziness/hypotension (3 patients, 9%), and paradoxical worsening of PTSD symptoms (4 patients, 12%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were syncope (2 patients), erectile dysfunction (1 patient), rash (1 patient), myoclonus (1 patient), increased depression (1 patient), and fatigue (1 patient). One patient reported that he had discontinued clonidine due to symptom resolution/lack of need for treatment. In 8 of the 34 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 21 evaluable patients who continued to receive clonidine 6 months after initiation, 10 (48%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Six of the 21 patients (29%) had an MPR between 50% and 79%, and 5 patients (24%) had an MPR < 50%.
Of the 8 patients who continued on clonidine at the 2-year mark, 3 (38%) were adherent to therapy, with an MPR of ≥ 80%. Three more patients (38%) had a 2-year MPR between 50% and 80%, and 2 patients (25%) had an MPR < 50%.
Prazosin
Of the 60 prazosin patients assessed, 32 (53%) had a positive response to the medication for nighttime PTSD symptoms documented in the CPRS within 6 months of starting therapy. Six months after starting prazosin, 36 patients (60%) continued to take prazosin. Two years after starting therapy, 18 of the original 60 patients continued on prazosin for an overall 2-year continuation rate of 30%.
Tolerability
Of the 42 patients who discontinued prazosin within 2 years, six patients (14%) cited ineffectiveness of therapy as a reason for discontinuation. Thirteen patients (31%) reported discontinuing therapy due to AEs. Sedation (3 patients, 7%), dizziness/hypotension (3 patients, 7%), and paradoxical worsening of PTSD symptoms (6 patients, 14%) were the most common AEs leading to discontinuation. Other AEs cited as reasons for discontinuation were headache (2 patients), altered mental status (1 patient), and fatigue (1 patient). Three patients reported that they had discontinued clonidine due to symptom resolution/lack of need for treatment. Other reasons for discontinuation not related to AEs included flight rules (1 patient), changes to antihypertensive regimen (1 patient), refill issues (1 patient), and cost (1 patient). In 15 of the 42 patients, no reason for discontinuation was found in chart documentation.
Medication Possession Ratio
Among the 31 evaluable patients who continued to receive prazosin 6 months after initiation, 20 (65%) were determined to be highly adherent to therapy, with an MPR of ≥ 80%. Five of the 31 patients (16%) had an MPR between 50% and 80%, and 6 patients (19%) had an MPR < 50%.
Of the 15 evaluable patients who continued on prazosin at the 2-year mark, 9 (60%) were adherent to therapy, with an MPR of ≥ 80%. Three patients (20%) had a 2-year MPR between 50% and 80%, and 3 patients (20%) had an MPR < 50%.
Discussion
Although prazosin has been shown to be effective for nighttime PTSD symptoms in both prospective and retrospective evaluations in veterans, this study provides the first evidence to support the use of clonidine in a veteran population.10-12,15
Interestingly, 42% of the patients assessed received their first prescription of an α2-adrenergic agent for nighttime PTSD symptoms from a PCP. Even with the recent increased focus on integrating mental health into primary care within the VA, this was a surprising finding. Primary care providers at VAPHCS may have a greater role in the outpatient management of PTSD than previously suspected. The information presented here may prove useful and applicable in both psychiatric and primary care treatment settings.
The study results indicated that a majority of subjects initially reported effectiveness with either clonidine or prazosin (53% and 57%, respectively). The initial effectiveness rate for prazosin is similar to those described in previous studies.10-13,15 The data also support a viable role for clonidine in the treatment of nighttime PTSD symptoms.
Regardless of initial improvement, the study results also suggest that the therapeutic benefit may not persist in the long term, as evidenced by a significant percentage of discontinuations attributed to ineffectiveness (38% for clonidine and 14% for prazosin) and a very low rate of long-term continuation (19% for clonidine and 30% for prazosin at 2 years). This latter observation contrasts with findings from previous studies; Byers and colleagues reported a 2-year prazosin continuation rate of 48.4% in a similar analysis, and Boehnlein and colleagues reported a sustained benefit of clonidine in responders over a 10-year period.14,15 The wide variety of reasons for discontinuation reported here may help providers who are considering clonidine or prazosin for their patients to anticipate barriers to long-term success.
Part of the discrepancy between these results and previously reported successes with clonidine and prazosin may be attributable to the classic issue of efficacy vs effectiveness. Many of the studies that have informed us on the efficacy and tolerability of prazosin for nighttime PTSD symptoms described outcomes of prospective clinical research. Furthermore, these prospective trials were limited to < 6 months in duration. To date, neither clonidine nor prazosin has been evaluated for long-term efficacy and effectiveness in well-designed, prospective trials. This retrospective analysis may help provide a realistic estimate of the long-term effectiveness of these therapies, especially within the veteran population.
Limitations
This was a single-center, retrospective study conducted primarily in white male patients. Although likely applicable to the U.S. veteran population at large, these data may be poorly generalizable to patient populations outside the VA health care system.
Aside from external validity, this study has several significant limitations. The primary limitation of this project is that it was not designed to allow for statistical comparison of clonidine and prazosin. Such an analysis would have better defined the role of clonidine in PTSD treatment, either by establishing similar effectiveness of clonidine and prazosin for nighttime symptoms or by providing evidence of the superiority of one over the other. In designing the project, investigators suspected based on experience that the majority of patients prescribed clonidine would receive the drug after having already failed first-line therapy with prazosin. Had this been the case, a direct comparison may have been biased in favor of prazosin. In retrospect, however, only 24% of the clonidine group had previously been prescribed prazosin, and only 7% of the prazosin group had been prescribed clonidine. This suggests that clonidine may be used first line more often than the investigators anticipated and that a future direct comparison would be worthwhile.
Second, the subjective data collected for this project required investigators to read and interpret chart notes, although the review of all records by a single investigator helped limit variability in interpretation. At times, information in the CPRS was incomplete in terms of determining continuation of therapy or cause for discontinuation.
Third, although it is implied that a significant number of veterans have combat-related PTSD, the nature of the traumatic event(s) leading to PTSD was not recorded in this study, and no subgroup analysis was done to compare the effect of α2-adrenergic agents between combat- and noncombat-related PTSD. Owing to their exclusion by design, it is also difficult to apply these results to veterans who have lasting cognitive impairment as a result of TBI, who are presumably among those most likely to have experienced traumas that could provoke PTSD.
The design of this project also did not include a subgroup analysis based on antidepressant type, and it is unclear whether the potential pharmacodynamic interaction between noradrenergic antidepressants (ie, SNRIs) and anti– α2-adrenergic agents had any impact on clinical outcomes. The use of complementary nonpharmacologic treatment modalities (ie, psychotherapy, eye movement desensitization and reprocessing) was also not evaluated.
Related: Female Service Members in the Long War
Finally, the primary outcome of patient-reported improvement in symptoms does not provide information on the magnitude or specific nature of benefits derived. Given the retrospective nature, data used in prospectively designed studies (eg, rating scales pertinent to PTSD), which might have helped to quantify the benefit of treatment, was not consistently available. Even a baseline PCL-C score, collected in order to describe the patient population, was available only in 37% of the patients assessed. Furthermore, nighttime PTSD symptoms vary among individuals, but the primary outcome of this study pools any benefits seen in areas such as nightmares, awakenings, night sweats, or sleep quality into a single outcome of symptom improvement.
Conclusions
This study indicates that both clonidine and prazosin may be effective for the treatment of nighttime PTSD symptoms in the veteran population but that their long-term utility may be limited by waning effectiveness, tolerability, and adherence issues. At this time, it is unclear whether either agent has an advantage over the other in terms of effectiveness or tolerability; further studies are needed to address that question.
Despite its limitations, the authors anticipate that this study will provide information regarding the effectiveness and tolerability of clonidine and prazosin to treat nighttime PTSD symptoms. Findings from this study may help clinicians to anticipate the needs and challenges of patients using β2-adrenergic agents for nighttime symptoms of PTSD.
Acknowledgements
The authors wish to acknowledge Brian Wilcox, PharmD, for his assistance in generating patient data reports, and Ronald Brown, RPh, MS, for his guidance regarding data analysis.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
2. Gates MA, Holowka DW, Vasterling JJ, Keane TM, Marx BP, Rosen RC. Posttraumatic stress disorder in veterans and military personnel: epidemiology, screening, and case recognition. Psychol Serv. 2012;9(4):361-382.
3. Kulka R, Schlenger WE, Fairbanks J, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. New York, NY: Brunnel/Mazel; 1990.
4. Barrera TL, Graham DP, Dunn NJ, Teng EJ. Influence of trauma history on panic and posttraumatic stress disorder in returning veterans. Psychol Serv. 2013;10(2):168-176.
5. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Acute Stress Disorder and Posttraumatic Stress Disorder. Arlington, VA: American Psychiatric Association; 2004.
6. U.S. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for management of post-traumatic stress. Version 2.0. U.S. Department of Veterans Affairs Website. http://www.healthquality.va.gov/guidelines/MH/ptsd/cpgPTSDFULL201011612c.pdf. Published October 2010. Accessed October 5, 2015.
7. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
8. Ravindran LN, Stein MB. Pharmacotherapy of post-traumatic stress disorder. In: Stein MB, Steckler T, eds. Behavioral Neurobiology of Anxiety and Its Treatment. Vol 2. Heidelberg, Germany: Springer; 2010:505-525.
9. Spoormaker VI, Montgomery P. Disturbed sleep in post-traumatic stress disorder: secondary symptom or core feature? Sleep Med Rev. 2008;12(3):169-184.
10. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo controlled study. Am J Psychiatry. 2003;160(2):371-373.
11. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
12. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170:1003-1010.
13. Boehnlein JK, Kinzie JD. Pharmacologic reduction of CNS noradrenergic activity in PTSD: the case for clonidine and prazosin. J Psychiatr Pract. 2007;13(2):72-78.
14. Boehnlein JK, Kinzie JD, Sekiya U, Riley C, Pou K, Rosborough B. A ten-year treatment outcome study of traumatized Cambodian refugees. J Nerve Ment Dis. 2004;192(10):658-663.
15. Byers MG, Allison KM, Wendel CS, Lee JK. Prazosin versus quetiapine for nighttime posttraumatic stress disorder symptoms in veterans: an assessment of long-term comparative effectiveness and safety. J Clin Psychopharmacol. 2010;30(3):225-229.
1. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
2. Gates MA, Holowka DW, Vasterling JJ, Keane TM, Marx BP, Rosen RC. Posttraumatic stress disorder in veterans and military personnel: epidemiology, screening, and case recognition. Psychol Serv. 2012;9(4):361-382.
3. Kulka R, Schlenger WE, Fairbanks J, et al. Trauma and the Vietnam War Generation: Report of Findings From the National Vietnam Veterans Readjustment Study. New York, NY: Brunnel/Mazel; 1990.
4. Barrera TL, Graham DP, Dunn NJ, Teng EJ. Influence of trauma history on panic and posttraumatic stress disorder in returning veterans. Psychol Serv. 2013;10(2):168-176.
5. American Psychiatric Association. Practice Guideline for the Treatment of Patients With Acute Stress Disorder and Posttraumatic Stress Disorder. Arlington, VA: American Psychiatric Association; 2004.
6. U.S. Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for management of post-traumatic stress. Version 2.0. U.S. Department of Veterans Affairs Website. http://www.healthquality.va.gov/guidelines/MH/ptsd/cpgPTSDFULL201011612c.pdf. Published October 2010. Accessed October 5, 2015.
7. Berger W, Mendlowicz MV, Marques-Portella C, et al. Pharmacologic alternatives to antidepressants in posttraumatic stress disorder: a systematic review. Prog Neuropsychopharmacol Biol Psychiatry. 2009;33(2):169-180.
8. Ravindran LN, Stein MB. Pharmacotherapy of post-traumatic stress disorder. In: Stein MB, Steckler T, eds. Behavioral Neurobiology of Anxiety and Its Treatment. Vol 2. Heidelberg, Germany: Springer; 2010:505-525.
9. Spoormaker VI, Montgomery P. Disturbed sleep in post-traumatic stress disorder: secondary symptom or core feature? Sleep Med Rev. 2008;12(3):169-184.
10. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo controlled study. Am J Psychiatry. 2003;160(2):371-373.
11. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61(8):928-934.
12. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170:1003-1010.
13. Boehnlein JK, Kinzie JD. Pharmacologic reduction of CNS noradrenergic activity in PTSD: the case for clonidine and prazosin. J Psychiatr Pract. 2007;13(2):72-78.
14. Boehnlein JK, Kinzie JD, Sekiya U, Riley C, Pou K, Rosborough B. A ten-year treatment outcome study of traumatized Cambodian refugees. J Nerve Ment Dis. 2004;192(10):658-663.
15. Byers MG, Allison KM, Wendel CS, Lee JK. Prazosin versus quetiapine for nighttime posttraumatic stress disorder symptoms in veterans: an assessment of long-term comparative effectiveness and safety. J Clin Psychopharmacol. 2010;30(3):225-229.
AAP: Creating safe environment aids recovery from trauma disorders
WASHINGTON – Maintaining safety and helping to ensure caregiver well-being are critical interventions for children with posttraumatic stress disorder (PTSD) and other trauma syndromes, Dr. Mary Margaret Gleason said at the annual meeting of the American Academy of Pediatrics.
“Talk a lot [with caregivers] about safety, meaning the [importance of a] lack of violence in the home and the lack of corporal punishment,” said Dr. Gleason, a pediatrician and child and adolescent psychiatrist at Tulane University in New Orleans. “And equally important, make sure that parents are taking care of themselves and are getting treatment [if they’ve also experienced trauma].”
PTSD must be treated with evidence-based therapy or it will become more entrenched with time, she said. Both child-parent psychotherapy in toddlers and preschool-aged children, and cognitive behavioral therapy (CBT) in children of preschool age and up, lead to short-term and sustained reductions in children’s symptoms.
In communities without treatment specialists and resources, stressing safety becomes even more important. Help children with PTSD and other trauma disorders to learn to recognize and label their feelings, manage emotional dysregulation, learn the principles of CBT, and achieve deep relaxation. “You can put a pulse oximeter on a child and teach them how to do deep relaxation,” she said. “You can challenge them to bring down the numbers.”
Books on CBT for general anxiety – and, increasingly, online CBT tools – can be helpful for some families, as can the National Child Traumatic Stress Network’s online resources (www.nctsn.org).
Dr. Gleason advised looking for a broad range of disorders in children who have experienced trauma – whether the trauma is acute or chronic, and whether it involves personal or community events. “You need to be thinking about PTSD, certainly, but also attention-deficit/hyperactivity disorder (ADHD), disruptive behaviors, mood disorders, separation anxiety disorders, and sleep disorders,” she said. Conversely, “if a child develops another disorder after a traumatic event, look for signs of PTSD.”
A study of children living in and around New Orleans when Hurricane Katrina struck in 2005 found a wide range of disorders as well as high prevalence of PTSD. “There were [few] children who developed new diagnoses who didn’t also have some symptoms of PTSD,” Dr. Gleason said.
The Diagnostic and Statistical Manual of Mental Disorders–5 placed PTSD in the diagnostic category of trauma- and stressor-related disorders and included separate criteria for PTSD in preschool children. PTSD broadly involves symptoms of at least 1-month duration from four symptom clusters: Intrusive thoughts (e.g. distressing dreams), avoidance, negative alterations in cognition and mood, and changed arousal and reactivity.
Preschool PTSD applies to children ages 6 years and younger, and requires fewer symptoms to be present. The arousal/reactivity symptoms cluster includes irritability and extreme tantrums, she noted.
For children of any age, it is important to note that distressing dreams do not have to involve content specific to the traumatic event to meet diagnostic criteria, Dr. Gleason said.
Compared with PTSD, less is known about the presentation in school-age children and adolescents of reactive attachment disorder (RAD) and disinhibited social engagement disorder (DSED) – two other trauma-related disorders in DSM-5. Both are related to the attachment system and require a developmental age of at least 9 months, and both involve “pathogenic care,” which can mean emotional neglect and/or persistent disregard of the child’s basic physical needs.
The child with RAD “rarely seeks comfort” when distressed, has a limited response to comfort, and has limited positive affect, Dr. Gleason explained.
“When they fall and hurt their knee, or when they build a tower and it falls over, they don’t look for anyone to help them organize their feelings. And when someone tries to help them, this doesn’t reduce their distress,” she said.
There is some overlap with the clinical presentation of autism spectrum disorder, so it is important to rule out ASD in diagnosing RAD.
Treatment, she said, entails placement in an adequate caregiving environment. “What’s really important to know about RAD is that it’s really a reflection of the current caregiving environment,” Dr. Gleason said. “It’s not about the history. It’s about the [trauma] of what’s happening now.”
DSED is quite different in its clinical construct and course. “These children are indiscriminately social,” she said. “They’ll go off with a stranger … off into new situations without ever looking back to see if their caregiver is there.”
Unlike RAD, this disorder is not associated with current caregiving quality. “It’s important for caregivers to know that this syndrome is a reflection of what happened earlier and not what’s happening now,” Dr. Gleason said. “And it does respond to quality caregiving, but very, very slowly. It can take years.”
Dr. Gleason reported that she has no relevant financial disclosures.
WASHINGTON – Maintaining safety and helping to ensure caregiver well-being are critical interventions for children with posttraumatic stress disorder (PTSD) and other trauma syndromes, Dr. Mary Margaret Gleason said at the annual meeting of the American Academy of Pediatrics.
“Talk a lot [with caregivers] about safety, meaning the [importance of a] lack of violence in the home and the lack of corporal punishment,” said Dr. Gleason, a pediatrician and child and adolescent psychiatrist at Tulane University in New Orleans. “And equally important, make sure that parents are taking care of themselves and are getting treatment [if they’ve also experienced trauma].”
PTSD must be treated with evidence-based therapy or it will become more entrenched with time, she said. Both child-parent psychotherapy in toddlers and preschool-aged children, and cognitive behavioral therapy (CBT) in children of preschool age and up, lead to short-term and sustained reductions in children’s symptoms.
In communities without treatment specialists and resources, stressing safety becomes even more important. Help children with PTSD and other trauma disorders to learn to recognize and label their feelings, manage emotional dysregulation, learn the principles of CBT, and achieve deep relaxation. “You can put a pulse oximeter on a child and teach them how to do deep relaxation,” she said. “You can challenge them to bring down the numbers.”
Books on CBT for general anxiety – and, increasingly, online CBT tools – can be helpful for some families, as can the National Child Traumatic Stress Network’s online resources (www.nctsn.org).
Dr. Gleason advised looking for a broad range of disorders in children who have experienced trauma – whether the trauma is acute or chronic, and whether it involves personal or community events. “You need to be thinking about PTSD, certainly, but also attention-deficit/hyperactivity disorder (ADHD), disruptive behaviors, mood disorders, separation anxiety disorders, and sleep disorders,” she said. Conversely, “if a child develops another disorder after a traumatic event, look for signs of PTSD.”
A study of children living in and around New Orleans when Hurricane Katrina struck in 2005 found a wide range of disorders as well as high prevalence of PTSD. “There were [few] children who developed new diagnoses who didn’t also have some symptoms of PTSD,” Dr. Gleason said.
The Diagnostic and Statistical Manual of Mental Disorders–5 placed PTSD in the diagnostic category of trauma- and stressor-related disorders and included separate criteria for PTSD in preschool children. PTSD broadly involves symptoms of at least 1-month duration from four symptom clusters: Intrusive thoughts (e.g. distressing dreams), avoidance, negative alterations in cognition and mood, and changed arousal and reactivity.
Preschool PTSD applies to children ages 6 years and younger, and requires fewer symptoms to be present. The arousal/reactivity symptoms cluster includes irritability and extreme tantrums, she noted.
For children of any age, it is important to note that distressing dreams do not have to involve content specific to the traumatic event to meet diagnostic criteria, Dr. Gleason said.
Compared with PTSD, less is known about the presentation in school-age children and adolescents of reactive attachment disorder (RAD) and disinhibited social engagement disorder (DSED) – two other trauma-related disorders in DSM-5. Both are related to the attachment system and require a developmental age of at least 9 months, and both involve “pathogenic care,” which can mean emotional neglect and/or persistent disregard of the child’s basic physical needs.
The child with RAD “rarely seeks comfort” when distressed, has a limited response to comfort, and has limited positive affect, Dr. Gleason explained.
“When they fall and hurt their knee, or when they build a tower and it falls over, they don’t look for anyone to help them organize their feelings. And when someone tries to help them, this doesn’t reduce their distress,” she said.
There is some overlap with the clinical presentation of autism spectrum disorder, so it is important to rule out ASD in diagnosing RAD.
Treatment, she said, entails placement in an adequate caregiving environment. “What’s really important to know about RAD is that it’s really a reflection of the current caregiving environment,” Dr. Gleason said. “It’s not about the history. It’s about the [trauma] of what’s happening now.”
DSED is quite different in its clinical construct and course. “These children are indiscriminately social,” she said. “They’ll go off with a stranger … off into new situations without ever looking back to see if their caregiver is there.”
Unlike RAD, this disorder is not associated with current caregiving quality. “It’s important for caregivers to know that this syndrome is a reflection of what happened earlier and not what’s happening now,” Dr. Gleason said. “And it does respond to quality caregiving, but very, very slowly. It can take years.”
Dr. Gleason reported that she has no relevant financial disclosures.
WASHINGTON – Maintaining safety and helping to ensure caregiver well-being are critical interventions for children with posttraumatic stress disorder (PTSD) and other trauma syndromes, Dr. Mary Margaret Gleason said at the annual meeting of the American Academy of Pediatrics.
“Talk a lot [with caregivers] about safety, meaning the [importance of a] lack of violence in the home and the lack of corporal punishment,” said Dr. Gleason, a pediatrician and child and adolescent psychiatrist at Tulane University in New Orleans. “And equally important, make sure that parents are taking care of themselves and are getting treatment [if they’ve also experienced trauma].”
PTSD must be treated with evidence-based therapy or it will become more entrenched with time, she said. Both child-parent psychotherapy in toddlers and preschool-aged children, and cognitive behavioral therapy (CBT) in children of preschool age and up, lead to short-term and sustained reductions in children’s symptoms.
In communities without treatment specialists and resources, stressing safety becomes even more important. Help children with PTSD and other trauma disorders to learn to recognize and label their feelings, manage emotional dysregulation, learn the principles of CBT, and achieve deep relaxation. “You can put a pulse oximeter on a child and teach them how to do deep relaxation,” she said. “You can challenge them to bring down the numbers.”
Books on CBT for general anxiety – and, increasingly, online CBT tools – can be helpful for some families, as can the National Child Traumatic Stress Network’s online resources (www.nctsn.org).
Dr. Gleason advised looking for a broad range of disorders in children who have experienced trauma – whether the trauma is acute or chronic, and whether it involves personal or community events. “You need to be thinking about PTSD, certainly, but also attention-deficit/hyperactivity disorder (ADHD), disruptive behaviors, mood disorders, separation anxiety disorders, and sleep disorders,” she said. Conversely, “if a child develops another disorder after a traumatic event, look for signs of PTSD.”
A study of children living in and around New Orleans when Hurricane Katrina struck in 2005 found a wide range of disorders as well as high prevalence of PTSD. “There were [few] children who developed new diagnoses who didn’t also have some symptoms of PTSD,” Dr. Gleason said.
The Diagnostic and Statistical Manual of Mental Disorders–5 placed PTSD in the diagnostic category of trauma- and stressor-related disorders and included separate criteria for PTSD in preschool children. PTSD broadly involves symptoms of at least 1-month duration from four symptom clusters: Intrusive thoughts (e.g. distressing dreams), avoidance, negative alterations in cognition and mood, and changed arousal and reactivity.
Preschool PTSD applies to children ages 6 years and younger, and requires fewer symptoms to be present. The arousal/reactivity symptoms cluster includes irritability and extreme tantrums, she noted.
For children of any age, it is important to note that distressing dreams do not have to involve content specific to the traumatic event to meet diagnostic criteria, Dr. Gleason said.
Compared with PTSD, less is known about the presentation in school-age children and adolescents of reactive attachment disorder (RAD) and disinhibited social engagement disorder (DSED) – two other trauma-related disorders in DSM-5. Both are related to the attachment system and require a developmental age of at least 9 months, and both involve “pathogenic care,” which can mean emotional neglect and/or persistent disregard of the child’s basic physical needs.
The child with RAD “rarely seeks comfort” when distressed, has a limited response to comfort, and has limited positive affect, Dr. Gleason explained.
“When they fall and hurt their knee, or when they build a tower and it falls over, they don’t look for anyone to help them organize their feelings. And when someone tries to help them, this doesn’t reduce their distress,” she said.
There is some overlap with the clinical presentation of autism spectrum disorder, so it is important to rule out ASD in diagnosing RAD.
Treatment, she said, entails placement in an adequate caregiving environment. “What’s really important to know about RAD is that it’s really a reflection of the current caregiving environment,” Dr. Gleason said. “It’s not about the history. It’s about the [trauma] of what’s happening now.”
DSED is quite different in its clinical construct and course. “These children are indiscriminately social,” she said. “They’ll go off with a stranger … off into new situations without ever looking back to see if their caregiver is there.”
Unlike RAD, this disorder is not associated with current caregiving quality. “It’s important for caregivers to know that this syndrome is a reflection of what happened earlier and not what’s happening now,” Dr. Gleason said. “And it does respond to quality caregiving, but very, very slowly. It can take years.”
Dr. Gleason reported that she has no relevant financial disclosures.
EXPERT ANALYSIS FROM THE AAP NATIONAL CONFERENCE
PTSD in Women and Men
Beginning in September, Federal Practitioner will focus on female health and women service members, including interviews with authors of chapters from Women at War. For more, visit www.fedprac.com/articles/women-at-war.html.
In addition to coediting the Women at War volume, Dr. Ritchie authored the chapter, “Female Soldiers and Post-Traumatic Stress Disorder” (PTSD). In this interview, she discusses the evolving definition of PTSD, military and civilian research into the condition, its stressors, and how PTSD may differ in women and men.
For a limited time, a discount is being offered to Federal Practitioner readers. Click here and use the promo code AMPROMD9 at checkout.
Beginning in September, Federal Practitioner will focus on female health and women service members, including interviews with authors of chapters from Women at War. For more, visit www.fedprac.com/articles/women-at-war.html.
In addition to coediting the Women at War volume, Dr. Ritchie authored the chapter, “Female Soldiers and Post-Traumatic Stress Disorder” (PTSD). In this interview, she discusses the evolving definition of PTSD, military and civilian research into the condition, its stressors, and how PTSD may differ in women and men.
For a limited time, a discount is being offered to Federal Practitioner readers. Click here and use the promo code AMPROMD9 at checkout.
Beginning in September, Federal Practitioner will focus on female health and women service members, including interviews with authors of chapters from Women at War. For more, visit www.fedprac.com/articles/women-at-war.html.
In addition to coediting the Women at War volume, Dr. Ritchie authored the chapter, “Female Soldiers and Post-Traumatic Stress Disorder” (PTSD). In this interview, she discusses the evolving definition of PTSD, military and civilian research into the condition, its stressors, and how PTSD may differ in women and men.
For a limited time, a discount is being offered to Federal Practitioner readers. Click here and use the promo code AMPROMD9 at checkout.