User login
COVID-19 has brought more complex, longer office visits
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Evidence of this came from the latest Primary Care Collaborative (PCC) survey, which found that primary care clinicians are seeing more complex patients requiring longer appointments in the wake of COVID-19.
The PCC with the Larry A. Green Center regularly surveys primary care clinicians. This round of questions came August 14-17 and included 1,263 respondents from 49 states, the District of Columbia, and two territories.
More than 7 in 10 (71%) respondents said their patients are more complex and nearly the same percentage said appointments are taking more time.
Ann Greiner, president and CEO of the PCC, said in an interview that 55% of respondents reported that clinicians are struggling to keep up with pent-up demand after patients have delayed or canceled care. Sixty-five percent in the survey said they had seen a rise in children’s mental health issues, and 58% said they were unsure how to help their patients with long COVID.
In addition, primary care clinicians are having repeated conversations with patients on why they should get a vaccine and which one.
“I think that’s adding to the complexity. There is a lot going on here with patient trust,” Ms. Greiner said.
‘We’re going to be playing catch-up’
Jacqueline Fincher, MD, an internist in Thompson, Ga., said in an interview that appointments have gotten longer and more complex in the wake of the pandemic – “no question.”
The immediate past president of the American College of Physicians is seeing patients with chronic disease that has gone untreated for sometimes a year or more, she said.
“Their blood pressure was not under good control, they were under more stress, their sugars were up and weren’t being followed as closely for conditions such as congestive heart failure,” she said.
Dr. Fincher, who works in a rural practice 40 miles from Augusta, Ga., with her physician husband and two other physicians, said patients are ready to come back in, “but I don’t have enough slots for them.”
She said she prioritizes what to help patients with first and schedules the next tier for the next appointment, but added, “honestly, over the next 2 years we’re going to be playing catch-up.”
At the same time, the CDC has estimated that 45% of U.S. adults are at increased risk for complications from COVID-19 because of cardiovascular disease, diabetes, respiratory disease, hypertension, or cancer. Rates ranged from 19.8% for people 18-29 years old to 80.7% for people over 80 years of age.
Long COVID could overwhelm existing health care capacity
Primary care physicians are also having to diagnose sometimes “invisible” symptoms after people have recovered from acute COVID-19 infection. Diagnosing takes intent listening to patients who describe symptoms that tests can’t confirm.
As this news organization has previously reported, half of COVID-19 survivors report postacute sequelae of COVID-19 (PASC) lasting longer than 6 months.
“These long-term PASC effects occur on a scale that could overwhelm existing health care capacity, particularly in low- and middle-income countries,” the authors wrote.
Anxiety, depression ‘have gone off the charts’
Danielle Loeb, MD, MPH, associate professor of internal medicine at the University of Colorado in Denver, who studies complexity in primary care, said in the wake of COVID-19, more patients have developed “new, serious anxiety.”
“That got extremely exacerbated during the pandemic. Anxiety and depression have gone off the charts,” said Dr. Loeb, who prefers the pronoun “they.”
Dr. Loeb cares for a large number of transgender patients. As offices reopen, some patients are having trouble reintegrating into the workplace and resuming social contacts. The primary care doctor says appointments can get longer because of the need to complete tasks, such as filling out forms for Family Medical Leave Act for those not yet ready to return to work.
COVID-19–related fears are keeping many patients from coming into the office, Dr. Loeb said, either from fear of exposure or because they have mental health issues that keep them from feeling safe leaving the house.
“That really affects my ability to care for them,” they said.
Loss of employment in the pandemic or fear of job loss and subsequent changing of insurance has complicated primary care in terms of treatment and administrative tasks, according to Dr. Loeb.
To help treat patients with acute mental health issues and manage other patients, Dr. Loeb’s practice has brought in a social worker and a therapist.
Team-based care is key in the survival of primary care practices, though providing that is difficult in the smaller clinics because of the critical mass of patients needed to make it viable, they said.
“It’s the only answer. It’s the only way you don’t drown,” Dr. Loeb added. “I’m not drowning, and I credit that to my clinic having the help to support the mental health piece of things.”
Rethinking workflow
Tricia McGinnis, MPP, MPH, executive vice president of the nonprofit Center for Health Care Strategies (CHCS) says complexity has forced rethinking workflow.
“A lot of the trends we’re seeing in primary care were there pre-COVID, but COVID has exacerbated those trends,” she said in an interview.
“The good news ... is that it was already becoming clear that primary care needed to provide basic mental health services and integrate with behavioral health. It had also become clear that effective primary care needed to address social issues that keep patients from accessing health care,” she said.
Expanding care teams, as Dr. Loeb mentioned, is a key strategy, according to Ms. McGinnis. Potential teams would include the clinical staff, but also social workers and community health workers – people who come from the community primary care is serving who can help build trust with patients and connect the patient to the primary care team.
“There’s a lot that needs to happen that the clinician doesn’t need to do,” she said.
Telehealth can be a big factor in coordinating the team, Ms. McGinnis added.
“It’s thinking less about who’s doing the work, but more about the work that needs to be done to keep people healthy. Then let’s think about the type of workers best suited to perform those tasks,” she said.
As for reimbursing more complex care, population-based, up-front capitated payments linked to high-quality care and better outcomes will need to replace fee-for-service models, according to Ms. McGinnis.
That will provide reliable incomes for primary care offices, but also flexibility in how each patient with different levels of complexity is managed, she said.
Ms. Greiner, Dr. Fincher, Dr. Loeb, and Ms. McGinnis have no relevant financial relationships.
Time to retire race- and ethnicity-based carrier screening
The social reckoning of 2020 has led to many discussions and conversations around equity and disparities. With the COVID-19 pandemic, there has been a particular spotlight on health care disparities and race-based medicine. Racism in medicine is pervasive; little has been done over the years to dismantle and unlearn practices that continue to contribute to existing gaps and disparities. Race and ethnicity are both social constructs that have long been used within medical practice and in dictating the type of care an individual receives. Without a universal definition, race, ethnicity, and ancestry have long been used interchangeably within medicine and society. Appreciating that race and ethnicity-based constructs can have other social implications in health care, with their impact on structural racism beyond health care settings, these constructs may still be part of assessments and key modifiers to understanding health differences. It is imperative that medical providers examine the use of race and ethnicity within the care that they provide.
While racial determinants of health cannot be removed from historical access, utilization, and barriers related to reproductive care, guidelines structured around historical ethnicity and race further restrict universal access to carrier screening and informed reproductive testing decisions.
Carrier screening
The goal of preconception and prenatal carrier screening is to provide individuals and reproductive partners with information to optimize pregnancy outcomes based on personal values and preferences.1 The practice of carrier screening began almost half a century ago with screening for individual conditions seen more frequently in certain populations, such as Tay-Sachs disease in those of Ashkenazi Jewish descent and sickle cell disease in those of African descent. Cystic fibrosis carrier screening was first recommended for individuals of Northern European descent in 2001 before being recommended for pan ethnic screening a decade later. Other individual conditions are also recommended for screening based on race/ethnicity (eg, Canavan disease in the Ashkenazi Jewish population, Tay-Sachs disease in individuals of Cajun or French-Canadian descent).2-4 Practice guidelines from professional societies recommend offering carrier screening for individual conditions based on condition severity, race or ethnicity, prevalence, carrier frequency, detection rates, and residual risk.1 However, this process can be problematic, as the data frequently used in updating guidelines and recommendations come primarily from studies and databases where much of the cohort is White.5,6 Failing to identify genetic associations in diverse populations limits the ability to illuminate new discoveries that inform risk management and treatment, especially for populations that are disproportionately underserved in medicine.7
Need for expanded carrier screening
The evolution of genomics and technology within the realm of carrier screening has enabled the simultaneous screening for many serious Mendelian diseases, known as expanded carrier screening (ECS). A 2016 study illustrated that, in most racial/ethnic categories, the cumulative risk of severe and profound conditions found on ECS panels outside the guideline recommendations are greater than the risk identified by guideline-based panels.8 Additionally, a 2020 study showed that self-reported ethnicity was an imperfect indicator of genetic ancestry, with 9% of those in the cohort having a >50% genetic ancestry from a lineage inconsistent with their self-reported ethnicity.9 Data over the past decade have established the clinical utility,10 clinical validity,11 analytical validity,12 and cost-effectiveness13 of pan-ethnic ECS. In 2021, American College of Medical Genetics and Genomics (ACMG) recommended a panel of pan-ethnic conditions that should be offered to all patients due to smaller ethnicity-based panels failing to provide equitable evaluation of all racial and ethnic groups.14 The guidelines from the American College of Obstetricians and Gynecologists (ACOG) fall short of recommending that ECS be offered to all individuals in lieu of screening based on self-reported ethnicity.3,4
Phasing out ethnicity-based carrier screening
This begs the question: Do race, ethnicity, or ancestry have a role in carrier screening? While each may have had a role at the inception of offering carrier screening due to high costs of technology, recent studies have shown the limitations of using self-reported ethnicity in screening. Guideline-based carrier screenings miss a significant percentage of pregnancies (13% to 94%) affected by serious conditions on expanded carrier screening panels.8 Additionally, 40% of Americans cannot identify the ethnicity of all 4 grandparents.15
Founder mutations due to ancestry patterns are still present; however, stratification of care should only be pursued when the presence or absence of these markers would alter clinical management. While the reproductive risk an individual may receive varies based on their self-reported ethnicity, the clinically indicated follow-up testing is the same: offering carrier screening for the reproductive partner or gamete donor. With increased detection rates via sequencing for most autosomal recessive conditions, if the reproductive partner or gamete donor is not identified as a carrier, no further testing is generally indicated regardless of ancestry. Genotyping platforms should not be used for partner carrier screening as they primarily target common pathogenic variants based on dominant ancestry groups and do not provide the same risk reduction.
Continue to: Variant reporting...
Variant reporting
We have long known that databases and registries in the United States have an increased representation of individuals from European ancestries.5,6 However, there have been limited conversations about how the lack of representation within our databases and registries leads to inequities in guidelines and the care that we provide to patients. As a result, studies have shown higher rates of variants of uncertain significance (VUS) identified during genetic testing in non-White individuals than in Whites.16 When it comes to reporting of variants, carrier screening laboratories follow guidelines set forth by the ACMG, and most laboratories only report likely pathogenic or pathogenic variants.17 It is unknown how the higher rate of VUSs in the non-White population, and lack of data and representation in databases and software used to calculate predicted phenotype, impacts identification of at-risk carrier couples in these underrepresented populations. It is imperative that we increase knowledge and representation of variants across ethnicities to improve sensitivity and specificity across the population and not just for those of European descent.
Moving forward
Being aware of social- and race-based biases in carrier screening is important, but modifying structural systems to increase representation, access, and utility of carrier screening is a critical next step. Organizations like ACOG and ACMG have committed not only to understanding but also to addressing factors that have led to disparities and inequities in health care delivery and access.18,19 Actionable steps include offering a universal carrier screening program to all preconception and prenatal patients that addresses conditions with increased carrier frequency, in any population, defined as severe and moderate phenotype with established natural history.3,4 Educational materials should be provided to detail risks, benefits, and limitations of carrier screening, as well as shared decision making between patient and provider to align the patient’s wishes for the information provided by carrier screening.
A broader number of conditions offered through carrier screening will increase the likelihood of positive carrier results. The increase in carriers identified should be viewed as more accurate reproductive risk assessment in the context of equitable care, rather than justification for panels to be limited to specific ancestries. Simultaneous or tandem reproductive partner or donor testing can be considered to reduce clinical workload and time for results return.
In addition, increased representation of individuals who are from diverse ancestries in promotional and educational resources can reinforce that risk for Mendelian conditions is not specific to single ancestries or for targeted conditions. Future research should be conducted to examine the role of racial disparities related to carrier screening and greater inclusion and recruitment of diverse populations in data sets and research studies.
Learned biases toward race, religion, gender identity, sexual orientation, and economic status in the context of carrier screening should be examined and challenged to increase access for all patients who may benefit from this testing. For example, the use of gendered language within carrier screening guidelines and policies and how such screening is offered to patients should be examined. Guidelines do not specify what to do when someone is adopted, for instance, or does not know their ethnicity. It is important that, as genomic testing becomes more available, individuals and groups are not left behind and existing gaps are not further widened. Assessing for genetic variation that modifies for disease or treatment will be more powerful than stratifying based on race. Carrier screening panels should be comprehensive regardless of ancestry to ensure coverage for global genetic variation and to increase access for all patients to risk assessments that promote informed reproductive decision making.
Health equity requires unlearning certain behaviors
As clinicians we all have a commitment to educate and empower one another to offer care that helps promote health equity. Equitable care requires us to look at the current gaps and figure out what programs and initiatives need to be designed to address those gaps. Carrier screening is one such area in which we can work together to improve the overall care that our patients receive, but it is imperative that we examine our practices and unlearn behaviors that contribute to existing disparities. ●
- Edwards JG, Feldman G, Goldberg J, et al. Expanded carrier screening in reproductive medicine—points to consider: a joint statement of the American College of Medical Genetics and Genomics, American College of Obstetricians and Gynecologists, National Society of Genetic Counselors, Perinatal Quality Foundation, and Society for Maternal-Fetal Medicine. Obstet Gynecol. 2015;125:653-662. doi: 10.1097 /AOG.0000000000000666.
- Grody WW, Thompson BH, Gregg AR, et al. ACMG position statement on prenatal/preconception expanded carrier screening. Genet Med. 2013;15:482-483. doi: 10.1038/gim.2013.47.
- Committee Opinion No. 690. Summary: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129: 595-596. doi: 10.1097/AOG.0000000000001947.
- Committee Opinion No. 691. Carrier screening for genetic conditions. Obstet Gynecol. 2017;129:e41-e55. doi: 10.1097 /AOG.0000000000001952.
- Need AC, Goldstein DB. Next generation disparities in human genomics: concerns and remedies. Trends Genet. 2009;25:489-494. doi: 10.1016/j.tig.2009.09.012.
- Popejoy A, Fullerton S. Genomics is failing on diversity. Nature. 2016;538;161-164. doi: 10.1038/538161a.
- Ewing A. Reimagining health equity in genetic testing. Medpage Today. June 17, 2021. https://www.medpagetoday.com /opinion/second-opinions/93173. Accessed October 27, 2021.
- Haque IS, Lazarin GA, Kang HP, et al. Modeled fetal risk of genetic diseases identified by expanded carrier screening. JAMA. 2016;316:734-742. doi: 10.1001/jama.2016.11139.
- Kaseniit KE, Haque IS, Goldberg JD, et al. Genetic ancestry analysis on >93,000 individuals undergoing expanded carrier screening reveals limitations of ethnicity-based medical guidelines. Genet Med. 2020;22:1694-1702. doi: 10 .1038/s41436-020-0869-3.
- Johansen Taber KA, Beauchamp KA, Lazarin GA, et al. Clinical utility of expanded carrier screening: results-guided actionability and outcomes. Genet Med. 2019;21:1041-1048. doi: 10.1038/s41436-018-0321-0.
- Balzotti M, Meng L, Muzzey D, et al. Clinical validity of expanded carrier screening: Evaluating the gene-disease relationship in more than 200 conditions. Hum Mutat. 2020;41:1365-1371. doi: 10.1002/humu.24033.
- Hogan GJ, Vysotskaia VS, Beauchamp KA, et al. Validation of an expanded carrier screen that optimizes sensitivity via full-exon sequencing and panel-wide copy number variant identification. Clin Chem. 2018;64:1063-1073. doi: 10.1373 /clinchem.2018.286823.
- Beauchamp KA, Johansen Taber KA, Muzzey D. Clinical impact and cost-effectiveness of a 176-condition expanded carrier screen. Genet Med. 2019;21:1948-1957. doi: 10.1038/s41436-019-0455-8.
- Gregg AR, Aarabi M, Klugman S, et al. Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2021;23:1793-1806. doi: 10.1038/s41436-021-01203-z.
- Condit C, Templeton A, Bates BR, et al. Attitudinal barriers to delivery of race-targeted pharmacogenomics among informed lay persons. Genet Med. 2003;5:385-392. doi: 10 .1097/01.gim.0000087990.30961.72.
- Caswell-Jin J, Gupta T, Hall E, et al. Racial/ethnic differences in multiple-gene sequencing results for hereditary cancer risk. Genet Med. 2018;20:234-239.
- Richards S, Aziz N, Bale S, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405-424. doi:10.1038/gim.2015.30.
- Gregg AR. Message from ACMG President: overcoming disparities. Genet Med. 2020;22:1758.
The social reckoning of 2020 has led to many discussions and conversations around equity and disparities. With the COVID-19 pandemic, there has been a particular spotlight on health care disparities and race-based medicine. Racism in medicine is pervasive; little has been done over the years to dismantle and unlearn practices that continue to contribute to existing gaps and disparities. Race and ethnicity are both social constructs that have long been used within medical practice and in dictating the type of care an individual receives. Without a universal definition, race, ethnicity, and ancestry have long been used interchangeably within medicine and society. Appreciating that race and ethnicity-based constructs can have other social implications in health care, with their impact on structural racism beyond health care settings, these constructs may still be part of assessments and key modifiers to understanding health differences. It is imperative that medical providers examine the use of race and ethnicity within the care that they provide.
While racial determinants of health cannot be removed from historical access, utilization, and barriers related to reproductive care, guidelines structured around historical ethnicity and race further restrict universal access to carrier screening and informed reproductive testing decisions.
Carrier screening
The goal of preconception and prenatal carrier screening is to provide individuals and reproductive partners with information to optimize pregnancy outcomes based on personal values and preferences.1 The practice of carrier screening began almost half a century ago with screening for individual conditions seen more frequently in certain populations, such as Tay-Sachs disease in those of Ashkenazi Jewish descent and sickle cell disease in those of African descent. Cystic fibrosis carrier screening was first recommended for individuals of Northern European descent in 2001 before being recommended for pan ethnic screening a decade later. Other individual conditions are also recommended for screening based on race/ethnicity (eg, Canavan disease in the Ashkenazi Jewish population, Tay-Sachs disease in individuals of Cajun or French-Canadian descent).2-4 Practice guidelines from professional societies recommend offering carrier screening for individual conditions based on condition severity, race or ethnicity, prevalence, carrier frequency, detection rates, and residual risk.1 However, this process can be problematic, as the data frequently used in updating guidelines and recommendations come primarily from studies and databases where much of the cohort is White.5,6 Failing to identify genetic associations in diverse populations limits the ability to illuminate new discoveries that inform risk management and treatment, especially for populations that are disproportionately underserved in medicine.7
Need for expanded carrier screening
The evolution of genomics and technology within the realm of carrier screening has enabled the simultaneous screening for many serious Mendelian diseases, known as expanded carrier screening (ECS). A 2016 study illustrated that, in most racial/ethnic categories, the cumulative risk of severe and profound conditions found on ECS panels outside the guideline recommendations are greater than the risk identified by guideline-based panels.8 Additionally, a 2020 study showed that self-reported ethnicity was an imperfect indicator of genetic ancestry, with 9% of those in the cohort having a >50% genetic ancestry from a lineage inconsistent with their self-reported ethnicity.9 Data over the past decade have established the clinical utility,10 clinical validity,11 analytical validity,12 and cost-effectiveness13 of pan-ethnic ECS. In 2021, American College of Medical Genetics and Genomics (ACMG) recommended a panel of pan-ethnic conditions that should be offered to all patients due to smaller ethnicity-based panels failing to provide equitable evaluation of all racial and ethnic groups.14 The guidelines from the American College of Obstetricians and Gynecologists (ACOG) fall short of recommending that ECS be offered to all individuals in lieu of screening based on self-reported ethnicity.3,4
Phasing out ethnicity-based carrier screening
This begs the question: Do race, ethnicity, or ancestry have a role in carrier screening? While each may have had a role at the inception of offering carrier screening due to high costs of technology, recent studies have shown the limitations of using self-reported ethnicity in screening. Guideline-based carrier screenings miss a significant percentage of pregnancies (13% to 94%) affected by serious conditions on expanded carrier screening panels.8 Additionally, 40% of Americans cannot identify the ethnicity of all 4 grandparents.15
Founder mutations due to ancestry patterns are still present; however, stratification of care should only be pursued when the presence or absence of these markers would alter clinical management. While the reproductive risk an individual may receive varies based on their self-reported ethnicity, the clinically indicated follow-up testing is the same: offering carrier screening for the reproductive partner or gamete donor. With increased detection rates via sequencing for most autosomal recessive conditions, if the reproductive partner or gamete donor is not identified as a carrier, no further testing is generally indicated regardless of ancestry. Genotyping platforms should not be used for partner carrier screening as they primarily target common pathogenic variants based on dominant ancestry groups and do not provide the same risk reduction.

Continue to: Variant reporting...
Variant reporting
We have long known that databases and registries in the United States have an increased representation of individuals from European ancestries.5,6 However, there have been limited conversations about how the lack of representation within our databases and registries leads to inequities in guidelines and the care that we provide to patients. As a result, studies have shown higher rates of variants of uncertain significance (VUS) identified during genetic testing in non-White individuals than in Whites.16 When it comes to reporting of variants, carrier screening laboratories follow guidelines set forth by the ACMG, and most laboratories only report likely pathogenic or pathogenic variants.17 It is unknown how the higher rate of VUSs in the non-White population, and lack of data and representation in databases and software used to calculate predicted phenotype, impacts identification of at-risk carrier couples in these underrepresented populations. It is imperative that we increase knowledge and representation of variants across ethnicities to improve sensitivity and specificity across the population and not just for those of European descent.
Moving forward
Being aware of social- and race-based biases in carrier screening is important, but modifying structural systems to increase representation, access, and utility of carrier screening is a critical next step. Organizations like ACOG and ACMG have committed not only to understanding but also to addressing factors that have led to disparities and inequities in health care delivery and access.18,19 Actionable steps include offering a universal carrier screening program to all preconception and prenatal patients that addresses conditions with increased carrier frequency, in any population, defined as severe and moderate phenotype with established natural history.3,4 Educational materials should be provided to detail risks, benefits, and limitations of carrier screening, as well as shared decision making between patient and provider to align the patient’s wishes for the information provided by carrier screening.
A broader number of conditions offered through carrier screening will increase the likelihood of positive carrier results. The increase in carriers identified should be viewed as more accurate reproductive risk assessment in the context of equitable care, rather than justification for panels to be limited to specific ancestries. Simultaneous or tandem reproductive partner or donor testing can be considered to reduce clinical workload and time for results return.
In addition, increased representation of individuals who are from diverse ancestries in promotional and educational resources can reinforce that risk for Mendelian conditions is not specific to single ancestries or for targeted conditions. Future research should be conducted to examine the role of racial disparities related to carrier screening and greater inclusion and recruitment of diverse populations in data sets and research studies.
Learned biases toward race, religion, gender identity, sexual orientation, and economic status in the context of carrier screening should be examined and challenged to increase access for all patients who may benefit from this testing. For example, the use of gendered language within carrier screening guidelines and policies and how such screening is offered to patients should be examined. Guidelines do not specify what to do when someone is adopted, for instance, or does not know their ethnicity. It is important that, as genomic testing becomes more available, individuals and groups are not left behind and existing gaps are not further widened. Assessing for genetic variation that modifies for disease or treatment will be more powerful than stratifying based on race. Carrier screening panels should be comprehensive regardless of ancestry to ensure coverage for global genetic variation and to increase access for all patients to risk assessments that promote informed reproductive decision making.
Health equity requires unlearning certain behaviors
As clinicians we all have a commitment to educate and empower one another to offer care that helps promote health equity. Equitable care requires us to look at the current gaps and figure out what programs and initiatives need to be designed to address those gaps. Carrier screening is one such area in which we can work together to improve the overall care that our patients receive, but it is imperative that we examine our practices and unlearn behaviors that contribute to existing disparities. ●
The social reckoning of 2020 has led to many discussions and conversations around equity and disparities. With the COVID-19 pandemic, there has been a particular spotlight on health care disparities and race-based medicine. Racism in medicine is pervasive; little has been done over the years to dismantle and unlearn practices that continue to contribute to existing gaps and disparities. Race and ethnicity are both social constructs that have long been used within medical practice and in dictating the type of care an individual receives. Without a universal definition, race, ethnicity, and ancestry have long been used interchangeably within medicine and society. Appreciating that race and ethnicity-based constructs can have other social implications in health care, with their impact on structural racism beyond health care settings, these constructs may still be part of assessments and key modifiers to understanding health differences. It is imperative that medical providers examine the use of race and ethnicity within the care that they provide.
While racial determinants of health cannot be removed from historical access, utilization, and barriers related to reproductive care, guidelines structured around historical ethnicity and race further restrict universal access to carrier screening and informed reproductive testing decisions.
Carrier screening
The goal of preconception and prenatal carrier screening is to provide individuals and reproductive partners with information to optimize pregnancy outcomes based on personal values and preferences.1 The practice of carrier screening began almost half a century ago with screening for individual conditions seen more frequently in certain populations, such as Tay-Sachs disease in those of Ashkenazi Jewish descent and sickle cell disease in those of African descent. Cystic fibrosis carrier screening was first recommended for individuals of Northern European descent in 2001 before being recommended for pan ethnic screening a decade later. Other individual conditions are also recommended for screening based on race/ethnicity (eg, Canavan disease in the Ashkenazi Jewish population, Tay-Sachs disease in individuals of Cajun or French-Canadian descent).2-4 Practice guidelines from professional societies recommend offering carrier screening for individual conditions based on condition severity, race or ethnicity, prevalence, carrier frequency, detection rates, and residual risk.1 However, this process can be problematic, as the data frequently used in updating guidelines and recommendations come primarily from studies and databases where much of the cohort is White.5,6 Failing to identify genetic associations in diverse populations limits the ability to illuminate new discoveries that inform risk management and treatment, especially for populations that are disproportionately underserved in medicine.7
Need for expanded carrier screening
The evolution of genomics and technology within the realm of carrier screening has enabled the simultaneous screening for many serious Mendelian diseases, known as expanded carrier screening (ECS). A 2016 study illustrated that, in most racial/ethnic categories, the cumulative risk of severe and profound conditions found on ECS panels outside the guideline recommendations are greater than the risk identified by guideline-based panels.8 Additionally, a 2020 study showed that self-reported ethnicity was an imperfect indicator of genetic ancestry, with 9% of those in the cohort having a >50% genetic ancestry from a lineage inconsistent with their self-reported ethnicity.9 Data over the past decade have established the clinical utility,10 clinical validity,11 analytical validity,12 and cost-effectiveness13 of pan-ethnic ECS. In 2021, American College of Medical Genetics and Genomics (ACMG) recommended a panel of pan-ethnic conditions that should be offered to all patients due to smaller ethnicity-based panels failing to provide equitable evaluation of all racial and ethnic groups.14 The guidelines from the American College of Obstetricians and Gynecologists (ACOG) fall short of recommending that ECS be offered to all individuals in lieu of screening based on self-reported ethnicity.3,4
Phasing out ethnicity-based carrier screening
This begs the question: Do race, ethnicity, or ancestry have a role in carrier screening? While each may have had a role at the inception of offering carrier screening due to high costs of technology, recent studies have shown the limitations of using self-reported ethnicity in screening. Guideline-based carrier screenings miss a significant percentage of pregnancies (13% to 94%) affected by serious conditions on expanded carrier screening panels.8 Additionally, 40% of Americans cannot identify the ethnicity of all 4 grandparents.15
Founder mutations due to ancestry patterns are still present; however, stratification of care should only be pursued when the presence or absence of these markers would alter clinical management. While the reproductive risk an individual may receive varies based on their self-reported ethnicity, the clinically indicated follow-up testing is the same: offering carrier screening for the reproductive partner or gamete donor. With increased detection rates via sequencing for most autosomal recessive conditions, if the reproductive partner or gamete donor is not identified as a carrier, no further testing is generally indicated regardless of ancestry. Genotyping platforms should not be used for partner carrier screening as they primarily target common pathogenic variants based on dominant ancestry groups and do not provide the same risk reduction.

Continue to: Variant reporting...
Variant reporting
We have long known that databases and registries in the United States have an increased representation of individuals from European ancestries.5,6 However, there have been limited conversations about how the lack of representation within our databases and registries leads to inequities in guidelines and the care that we provide to patients. As a result, studies have shown higher rates of variants of uncertain significance (VUS) identified during genetic testing in non-White individuals than in Whites.16 When it comes to reporting of variants, carrier screening laboratories follow guidelines set forth by the ACMG, and most laboratories only report likely pathogenic or pathogenic variants.17 It is unknown how the higher rate of VUSs in the non-White population, and lack of data and representation in databases and software used to calculate predicted phenotype, impacts identification of at-risk carrier couples in these underrepresented populations. It is imperative that we increase knowledge and representation of variants across ethnicities to improve sensitivity and specificity across the population and not just for those of European descent.
Moving forward
Being aware of social- and race-based biases in carrier screening is important, but modifying structural systems to increase representation, access, and utility of carrier screening is a critical next step. Organizations like ACOG and ACMG have committed not only to understanding but also to addressing factors that have led to disparities and inequities in health care delivery and access.18,19 Actionable steps include offering a universal carrier screening program to all preconception and prenatal patients that addresses conditions with increased carrier frequency, in any population, defined as severe and moderate phenotype with established natural history.3,4 Educational materials should be provided to detail risks, benefits, and limitations of carrier screening, as well as shared decision making between patient and provider to align the patient’s wishes for the information provided by carrier screening.
A broader number of conditions offered through carrier screening will increase the likelihood of positive carrier results. The increase in carriers identified should be viewed as more accurate reproductive risk assessment in the context of equitable care, rather than justification for panels to be limited to specific ancestries. Simultaneous or tandem reproductive partner or donor testing can be considered to reduce clinical workload and time for results return.
In addition, increased representation of individuals who are from diverse ancestries in promotional and educational resources can reinforce that risk for Mendelian conditions is not specific to single ancestries or for targeted conditions. Future research should be conducted to examine the role of racial disparities related to carrier screening and greater inclusion and recruitment of diverse populations in data sets and research studies.
Learned biases toward race, religion, gender identity, sexual orientation, and economic status in the context of carrier screening should be examined and challenged to increase access for all patients who may benefit from this testing. For example, the use of gendered language within carrier screening guidelines and policies and how such screening is offered to patients should be examined. Guidelines do not specify what to do when someone is adopted, for instance, or does not know their ethnicity. It is important that, as genomic testing becomes more available, individuals and groups are not left behind and existing gaps are not further widened. Assessing for genetic variation that modifies for disease or treatment will be more powerful than stratifying based on race. Carrier screening panels should be comprehensive regardless of ancestry to ensure coverage for global genetic variation and to increase access for all patients to risk assessments that promote informed reproductive decision making.
Health equity requires unlearning certain behaviors
As clinicians we all have a commitment to educate and empower one another to offer care that helps promote health equity. Equitable care requires us to look at the current gaps and figure out what programs and initiatives need to be designed to address those gaps. Carrier screening is one such area in which we can work together to improve the overall care that our patients receive, but it is imperative that we examine our practices and unlearn behaviors that contribute to existing disparities. ●
- Edwards JG, Feldman G, Goldberg J, et al. Expanded carrier screening in reproductive medicine—points to consider: a joint statement of the American College of Medical Genetics and Genomics, American College of Obstetricians and Gynecologists, National Society of Genetic Counselors, Perinatal Quality Foundation, and Society for Maternal-Fetal Medicine. Obstet Gynecol. 2015;125:653-662. doi: 10.1097 /AOG.0000000000000666.
- Grody WW, Thompson BH, Gregg AR, et al. ACMG position statement on prenatal/preconception expanded carrier screening. Genet Med. 2013;15:482-483. doi: 10.1038/gim.2013.47.
- Committee Opinion No. 690. Summary: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129: 595-596. doi: 10.1097/AOG.0000000000001947.
- Committee Opinion No. 691. Carrier screening for genetic conditions. Obstet Gynecol. 2017;129:e41-e55. doi: 10.1097 /AOG.0000000000001952.
- Need AC, Goldstein DB. Next generation disparities in human genomics: concerns and remedies. Trends Genet. 2009;25:489-494. doi: 10.1016/j.tig.2009.09.012.
- Popejoy A, Fullerton S. Genomics is failing on diversity. Nature. 2016;538;161-164. doi: 10.1038/538161a.
- Ewing A. Reimagining health equity in genetic testing. Medpage Today. June 17, 2021. https://www.medpagetoday.com /opinion/second-opinions/93173. Accessed October 27, 2021.
- Haque IS, Lazarin GA, Kang HP, et al. Modeled fetal risk of genetic diseases identified by expanded carrier screening. JAMA. 2016;316:734-742. doi: 10.1001/jama.2016.11139.
- Kaseniit KE, Haque IS, Goldberg JD, et al. Genetic ancestry analysis on >93,000 individuals undergoing expanded carrier screening reveals limitations of ethnicity-based medical guidelines. Genet Med. 2020;22:1694-1702. doi: 10 .1038/s41436-020-0869-3.
- Johansen Taber KA, Beauchamp KA, Lazarin GA, et al. Clinical utility of expanded carrier screening: results-guided actionability and outcomes. Genet Med. 2019;21:1041-1048. doi: 10.1038/s41436-018-0321-0.
- Balzotti M, Meng L, Muzzey D, et al. Clinical validity of expanded carrier screening: Evaluating the gene-disease relationship in more than 200 conditions. Hum Mutat. 2020;41:1365-1371. doi: 10.1002/humu.24033.
- Hogan GJ, Vysotskaia VS, Beauchamp KA, et al. Validation of an expanded carrier screen that optimizes sensitivity via full-exon sequencing and panel-wide copy number variant identification. Clin Chem. 2018;64:1063-1073. doi: 10.1373 /clinchem.2018.286823.
- Beauchamp KA, Johansen Taber KA, Muzzey D. Clinical impact and cost-effectiveness of a 176-condition expanded carrier screen. Genet Med. 2019;21:1948-1957. doi: 10.1038/s41436-019-0455-8.
- Gregg AR, Aarabi M, Klugman S, et al. Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2021;23:1793-1806. doi: 10.1038/s41436-021-01203-z.
- Condit C, Templeton A, Bates BR, et al. Attitudinal barriers to delivery of race-targeted pharmacogenomics among informed lay persons. Genet Med. 2003;5:385-392. doi: 10 .1097/01.gim.0000087990.30961.72.
- Caswell-Jin J, Gupta T, Hall E, et al. Racial/ethnic differences in multiple-gene sequencing results for hereditary cancer risk. Genet Med. 2018;20:234-239.
- Richards S, Aziz N, Bale S, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405-424. doi:10.1038/gim.2015.30.
- Gregg AR. Message from ACMG President: overcoming disparities. Genet Med. 2020;22:1758.
- Edwards JG, Feldman G, Goldberg J, et al. Expanded carrier screening in reproductive medicine—points to consider: a joint statement of the American College of Medical Genetics and Genomics, American College of Obstetricians and Gynecologists, National Society of Genetic Counselors, Perinatal Quality Foundation, and Society for Maternal-Fetal Medicine. Obstet Gynecol. 2015;125:653-662. doi: 10.1097 /AOG.0000000000000666.
- Grody WW, Thompson BH, Gregg AR, et al. ACMG position statement on prenatal/preconception expanded carrier screening. Genet Med. 2013;15:482-483. doi: 10.1038/gim.2013.47.
- Committee Opinion No. 690. Summary: carrier screening in the age of genomic medicine. Obstet Gynecol. 2017;129: 595-596. doi: 10.1097/AOG.0000000000001947.
- Committee Opinion No. 691. Carrier screening for genetic conditions. Obstet Gynecol. 2017;129:e41-e55. doi: 10.1097 /AOG.0000000000001952.
- Need AC, Goldstein DB. Next generation disparities in human genomics: concerns and remedies. Trends Genet. 2009;25:489-494. doi: 10.1016/j.tig.2009.09.012.
- Popejoy A, Fullerton S. Genomics is failing on diversity. Nature. 2016;538;161-164. doi: 10.1038/538161a.
- Ewing A. Reimagining health equity in genetic testing. Medpage Today. June 17, 2021. https://www.medpagetoday.com /opinion/second-opinions/93173. Accessed October 27, 2021.
- Haque IS, Lazarin GA, Kang HP, et al. Modeled fetal risk of genetic diseases identified by expanded carrier screening. JAMA. 2016;316:734-742. doi: 10.1001/jama.2016.11139.
- Kaseniit KE, Haque IS, Goldberg JD, et al. Genetic ancestry analysis on >93,000 individuals undergoing expanded carrier screening reveals limitations of ethnicity-based medical guidelines. Genet Med. 2020;22:1694-1702. doi: 10 .1038/s41436-020-0869-3.
- Johansen Taber KA, Beauchamp KA, Lazarin GA, et al. Clinical utility of expanded carrier screening: results-guided actionability and outcomes. Genet Med. 2019;21:1041-1048. doi: 10.1038/s41436-018-0321-0.
- Balzotti M, Meng L, Muzzey D, et al. Clinical validity of expanded carrier screening: Evaluating the gene-disease relationship in more than 200 conditions. Hum Mutat. 2020;41:1365-1371. doi: 10.1002/humu.24033.
- Hogan GJ, Vysotskaia VS, Beauchamp KA, et al. Validation of an expanded carrier screen that optimizes sensitivity via full-exon sequencing and panel-wide copy number variant identification. Clin Chem. 2018;64:1063-1073. doi: 10.1373 /clinchem.2018.286823.
- Beauchamp KA, Johansen Taber KA, Muzzey D. Clinical impact and cost-effectiveness of a 176-condition expanded carrier screen. Genet Med. 2019;21:1948-1957. doi: 10.1038/s41436-019-0455-8.
- Gregg AR, Aarabi M, Klugman S, et al. Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2021;23:1793-1806. doi: 10.1038/s41436-021-01203-z.
- Condit C, Templeton A, Bates BR, et al. Attitudinal barriers to delivery of race-targeted pharmacogenomics among informed lay persons. Genet Med. 2003;5:385-392. doi: 10 .1097/01.gim.0000087990.30961.72.
- Caswell-Jin J, Gupta T, Hall E, et al. Racial/ethnic differences in multiple-gene sequencing results for hereditary cancer risk. Genet Med. 2018;20:234-239.
- Richards S, Aziz N, Bale S, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405-424. doi:10.1038/gim.2015.30.
- Gregg AR. Message from ACMG President: overcoming disparities. Genet Med. 2020;22:1758.
3D vs 2D mammography for detecting cancer in dense breasts
Text copyright DenseBreast-info.org.
Answer
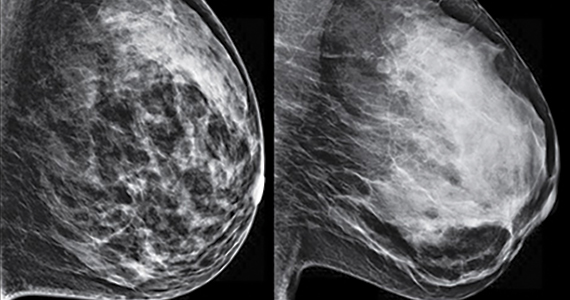
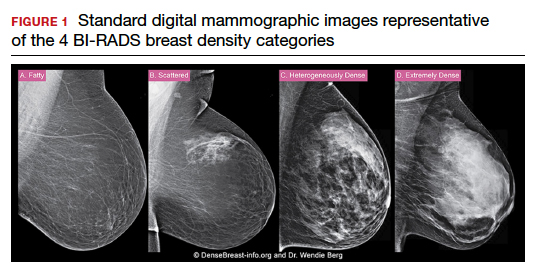
C. Overall, tomosynthesis depicts an additional 1 to 2 cancers per thousand women screened in the first round of screening when added to standard digital mammography;1-3 however, this improvement in cancer detection is only observed in women with fatty breasts (category A), scattered fibroglandular tissue (category B), and heterogeneously dense breasts (category C). Importantly, tomosynthesis does not significantly improve breast cancer detection in women with extremely dense breasts (category D).2,4
Digital breast tomosynthesis, also referred to as “3-dimensional mammography” (3D mammography) or tomosynthesis, uses a dedicated electronic detector system to obtain multiple projection images that are reconstructed by the computer to create thin slices or slabs of multiple slices of the breast. These slices can be individually “scrolled through” by the radiologist to reduce tissue overlap that may obscure breast cancers on a standard mammogram. While tomosynthesis improves breast cancer detection in women with fatty, scattered fibroglandular density, and heterogeneously dense breasts, there is very little soft tissue contrast in extremely dense breasts due to insufficient fat, and some cancers will remain hidden by dense tissue even on sliced images through the breast.
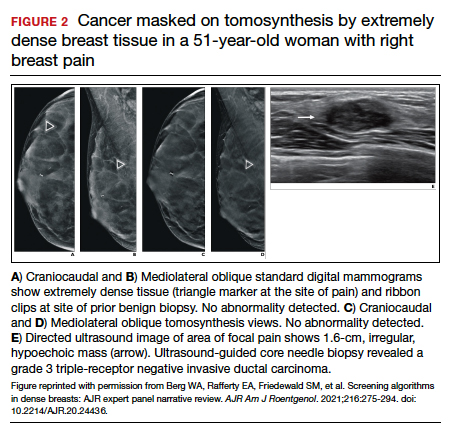
FIGURE 2 shows an example of cancer that was missed on tomosynthesis in a 51-year-old woman with extremely dense breasts and right breast pain. The cancer was masked by extremely dense tissue on standard digital mammography and tomosynthesis; no abnormalities were detected. Ultrasonography showed a 1.6-cm, irregular, hypoechoic mass at the site of pain, and biopsy revealed a grade 3 triple-receptor negative invasive ductal carcinoma.
In women with dense breasts, especially extremely dense breasts, supplemental screening beyond tomosynthesis should be considered. Although tomosynthesis doesn’t improve cancer detection in extremely dense breasts, it does reduce callbacks for additional testing in all breast densities compared with standard digital mammography. Callbacks are reduced from approximately 100‒120 per 1,000 women screened with standard digital mammography alone1,5 to an average of 80 per 1,000 women when tomosynthesis and standard mammography are interpreted together.1-3 ●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
- Conant EF, Zuckerman SP, McDonald ES, et al. Five consecutive years of screening with digital breast tomosynthesis: outcomes by screening year and round. Radiology. 2020;295:285-293.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786.
- Skaane P, Bandos AI, Niklason LT, et al. Digital mammography versus digital mammography plus tomosynthesis in breast cancer screening: the Oslo Tomosynthesis Screening Trial. Radiology. 2019;291:23-30.
- Lowry KP, Coley RY, Miglioretti DL, et al. Screening performance of digital breast tomosynthesis vs digital mammography in community practice by patient age, screening round, and breast density. JAMA Netw Open. 2020;3:e2011792.
- Lee CS, Sengupta D, Bhargavan-Chatfield M, et al. Association of patient age with outcomes of current-era, large-scale screening mammography: analysis of data from the National Mammography Database. JAMA Oncol. 2017;3:1134-1136.
Text copyright DenseBreast-info.org.
Answer
C. Overall, tomosynthesis depicts an additional 1 to 2 cancers per thousand women screened in the first round of screening when added to standard digital mammography;1-3 however, this improvement in cancer detection is only observed in women with fatty breasts (category A), scattered fibroglandular tissue (category B), and heterogeneously dense breasts (category C). Importantly, tomosynthesis does not significantly improve breast cancer detection in women with extremely dense breasts (category D).2,4
Digital breast tomosynthesis, also referred to as “3-dimensional mammography” (3D mammography) or tomosynthesis, uses a dedicated electronic detector system to obtain multiple projection images that are reconstructed by the computer to create thin slices or slabs of multiple slices of the breast. These slices can be individually “scrolled through” by the radiologist to reduce tissue overlap that may obscure breast cancers on a standard mammogram. While tomosynthesis improves breast cancer detection in women with fatty, scattered fibroglandular density, and heterogeneously dense breasts, there is very little soft tissue contrast in extremely dense breasts due to insufficient fat, and some cancers will remain hidden by dense tissue even on sliced images through the breast.
FIGURE 2 shows an example of cancer that was missed on tomosynthesis in a 51-year-old woman with extremely dense breasts and right breast pain. The cancer was masked by extremely dense tissue on standard digital mammography and tomosynthesis; no abnormalities were detected. Ultrasonography showed a 1.6-cm, irregular, hypoechoic mass at the site of pain, and biopsy revealed a grade 3 triple-receptor negative invasive ductal carcinoma.

In women with dense breasts, especially extremely dense breasts, supplemental screening beyond tomosynthesis should be considered. Although tomosynthesis doesn’t improve cancer detection in extremely dense breasts, it does reduce callbacks for additional testing in all breast densities compared with standard digital mammography. Callbacks are reduced from approximately 100‒120 per 1,000 women screened with standard digital mammography alone1,5 to an average of 80 per 1,000 women when tomosynthesis and standard mammography are interpreted together.1-3 ●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
Text copyright DenseBreast-info.org.
Answer
C. Overall, tomosynthesis depicts an additional 1 to 2 cancers per thousand women screened in the first round of screening when added to standard digital mammography;1-3 however, this improvement in cancer detection is only observed in women with fatty breasts (category A), scattered fibroglandular tissue (category B), and heterogeneously dense breasts (category C). Importantly, tomosynthesis does not significantly improve breast cancer detection in women with extremely dense breasts (category D).2,4
Digital breast tomosynthesis, also referred to as “3-dimensional mammography” (3D mammography) or tomosynthesis, uses a dedicated electronic detector system to obtain multiple projection images that are reconstructed by the computer to create thin slices or slabs of multiple slices of the breast. These slices can be individually “scrolled through” by the radiologist to reduce tissue overlap that may obscure breast cancers on a standard mammogram. While tomosynthesis improves breast cancer detection in women with fatty, scattered fibroglandular density, and heterogeneously dense breasts, there is very little soft tissue contrast in extremely dense breasts due to insufficient fat, and some cancers will remain hidden by dense tissue even on sliced images through the breast.
FIGURE 2 shows an example of cancer that was missed on tomosynthesis in a 51-year-old woman with extremely dense breasts and right breast pain. The cancer was masked by extremely dense tissue on standard digital mammography and tomosynthesis; no abnormalities were detected. Ultrasonography showed a 1.6-cm, irregular, hypoechoic mass at the site of pain, and biopsy revealed a grade 3 triple-receptor negative invasive ductal carcinoma.

In women with dense breasts, especially extremely dense breasts, supplemental screening beyond tomosynthesis should be considered. Although tomosynthesis doesn’t improve cancer detection in extremely dense breasts, it does reduce callbacks for additional testing in all breast densities compared with standard digital mammography. Callbacks are reduced from approximately 100‒120 per 1,000 women screened with standard digital mammography alone1,5 to an average of 80 per 1,000 women when tomosynthesis and standard mammography are interpreted together.1-3 ●
For more information, visit medically sourced DenseBreast-info.org. Comprehensive resources include a free CME opportunity, Dense Breasts and Supplemental Screening.
- Conant EF, Zuckerman SP, McDonald ES, et al. Five consecutive years of screening with digital breast tomosynthesis: outcomes by screening year and round. Radiology. 2020;295:285-293.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786.
- Skaane P, Bandos AI, Niklason LT, et al. Digital mammography versus digital mammography plus tomosynthesis in breast cancer screening: the Oslo Tomosynthesis Screening Trial. Radiology. 2019;291:23-30.
- Lowry KP, Coley RY, Miglioretti DL, et al. Screening performance of digital breast tomosynthesis vs digital mammography in community practice by patient age, screening round, and breast density. JAMA Netw Open. 2020;3:e2011792.
- Lee CS, Sengupta D, Bhargavan-Chatfield M, et al. Association of patient age with outcomes of current-era, large-scale screening mammography: analysis of data from the National Mammography Database. JAMA Oncol. 2017;3:1134-1136.
- Conant EF, Zuckerman SP, McDonald ES, et al. Five consecutive years of screening with digital breast tomosynthesis: outcomes by screening year and round. Radiology. 2020;295:285-293.
- Rafferty EA, Durand MA, Conant EF, et al. Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA. 2016;315:1784-1786.
- Skaane P, Bandos AI, Niklason LT, et al. Digital mammography versus digital mammography plus tomosynthesis in breast cancer screening: the Oslo Tomosynthesis Screening Trial. Radiology. 2019;291:23-30.
- Lowry KP, Coley RY, Miglioretti DL, et al. Screening performance of digital breast tomosynthesis vs digital mammography in community practice by patient age, screening round, and breast density. JAMA Netw Open. 2020;3:e2011792.
- Lee CS, Sengupta D, Bhargavan-Chatfield M, et al. Association of patient age with outcomes of current-era, large-scale screening mammography: analysis of data from the National Mammography Database. JAMA Oncol. 2017;3:1134-1136.
Quiz developed in collaboration with 
Boxed warnings: Legal risks that many physicians never see coming
Almost all physicians write prescriptions, and each prescription requires a physician to assess the risks and benefits of the drug. If an adverse drug reaction occurs, physicians may be called on to defend their risk-benefit assessment in court.
The assessment of risk is complicated when there is a boxed warning that describes potentially serious and life-threatening adverse reactions associated with a drug. Some of our most commonly prescribed drugs have boxed warnings, and drugs that were initially approved by the Food and Drug Administration without boxed warnings may have them added years later.
One serious problem with boxed warnings is that there are no reliable mechanisms for making sure that physicians are aware of them. The warnings are typically not seen by physicians as printed product labels, just as physicians often don’t see the pills and capsules that they prescribe. Pharmacists who receive packaged drugs from manufacturers may be the only ones to see an actual printed boxed warning, but even those pharmacists have little reason to read each label and note changes when handling many bulk packages.
This problem is aggravated by misperceptions that many physicians have about boxed warnings and the increasingly intense scrutiny given to them by mass media and the courts. Lawyers can use boxed warnings to make a drug look dangerous, even when it’s not, and to make physicians look reckless when prescribing it. Therefore, it is important for physicians to understand what boxed warnings are, what they are not, the problems they cause, and how to minimize these problems.
What is a ‘boxed warning’?
The marketing and sale of drugs in the United States requires approval by the FDA. Approval requires manufacturers to prepare a document containing “Full Prescribing Information” for the drug and to include a printed copy in every package of the drug that is sold. This document is commonly called a “package insert,” but the FDA designates this document as the manufacturer’s product “label.”
In 1979, the FDA began requiring some labels to appear within thick, black rectangular borders; these have come to be known as boxed warnings. Boxed warnings are usually placed at the beginning of a label. They may be added to the label of a previously approved drug already on the market or included in the product label when first approved and marketed.
The requirement for a boxed warning most often arises when a signal appears during review of postmarketing surveillance data suggesting a possible and plausible association between a drug and an adverse reaction. Warnings may also be initiated in response to petitions from public interest groups, or upon the discovery of serious toxicity in animals. Regardless of their origin, the intent of a boxed warning is to highlight information that may have important therapeutic consequences and warrants heightened awareness among physicians.
What a boxed warning is not
A boxed warning is not “issued” by the FDA; it is merely required by the FDA. Specific wording or a template may be suggested by the FDA, but product labels and boxed warnings are written and issued by the manufacturer. This distinction may seem minor, but extensive litigation has occurred over whether manufacturers have met their duty to warn consumers about possible risks when using their products, and this duty cannot be shifted to the FDA.
A boxed warning may not be added to a product label at the option of a manufacturer. The FDA allows a boxed warning only if it requires the warning, to preserve its impact. It should be noted that some medical information sources (e.g., PDR.net) may include a “BOXED WARNING” in their drug monographs, but monographs not written by a manufacturer are not regulated by the FDA, and the text of their boxed warnings do not always correspond to the boxed warning that was approved by the FDA.
A boxed warning is not an indication that revocation of FDA approval is being considered or that it is likely to be revoked. FDA approval is subject to ongoing review and may be revoked at any time, without a prior boxed warning.
A boxed warning is not the highest level of warning. The FDA may require a manufacturer to send out a “Dear Health Care Provider” (DHCP) letter when an even higher or more urgent level of warning is deemed necessary. DHCP letters are usually accompanied by revisions of the product label, but most label revisions – and even most boxed warnings – are not accompanied by DHCP letters.
A boxed warning is not a statement about causation. Most warnings describe an “association” between a drug and an adverse effect, or “increased risk,” or instances of a particular adverse effect that “have been reported” in persons taking a drug. The words in a boxed warning are carefully chosen and require careful reading; in most cases they refrain from stating that a drug actually causes an adverse effect. The postmarketing surveillance data on which most warnings are based generally cannot provide the kind of evidence required to establish causation, and an association may be nothing more than an uncommon manifestation of the disorder for which the drug has been prescribed.
A boxed warning is not a statement about the probability of an adverse reaction occurring. The requirement for a boxed warning correlates better to the new recognition of a possible association than to the probability of an association. For example, penicillin has long been known to cause fatal anaphylaxis in 1/100,000 first-time administrations, but it does not have a boxed warning. The adverse consequences described in boxed warnings are often far less frequent – so much so that most physicians will never see them.
A boxed warning does not define the standard of care. The warning is a requirement imposed on the manufacturer, not on the practice of medicine. For legal purposes, the “standard of care” for the practice of medicine is defined state by state and is typically cast in terms such as “what most physicians would do in similar circumstances.” Physicians often prescribe drugs in spite of boxed warnings, just as they often prescribe drugs for “off label” indications, always balancing risk versus benefit.
A boxed warning does not constitute a contraindication to the use of a medication. Some warnings state that a drug is contraindicated in some situations, but product labels have another mandated section for listing contraindications, and most boxed warnings have no corresponding entry in that section.
A boxed warning does not necessarily constitute current information, nor is it always updated when new or contrary information becomes available. Revisions to boxed warnings, and to product labels in general, are made only after detailed review at the FDA, and the process of deciding whether an existing boxed warning continues to be appropriate may divert limited regulatory resources from more urgent priorities. Consequently, revisions to a boxed warning may lag behind the data that justify a revision by months or years. Revisions may never occur if softening or eliminating a boxed warning is deemed to be not worth the cost by a manufacturer.
Boxed warning problems for physicians
There is no reliable mechanism for manufacturers or the FDA to communicate boxed warnings directly to physicians, so it’s not clear how physicians are expected to stay informed about the issuance or revision of boxed warnings. They may first learn about new or revised warnings in the mass media, which is paying ever-increasing attention to press releases from the FDA. However, it can be difficult for the media to accurately convey the subtle and complex nature of a boxed warning in nontechnical terms.
Many physicians subscribe to various medical news alerts and attend continuing medical education (CME) programs, which often do an excellent job of highlighting new warnings, while hospitals, clinics, and pharmacies may broadcast news about boxed warnings in newsletters or other notices. But these notifications are ephemeral and may be missed by physicians who are overwhelmed by email, notices, newsletters, and CME programs.
The warnings that pop up in electronic medical records systems are often so numerous that physicians become trained to ignore them. Printed advertisements in professional journals must include mandated boxed warnings, but their visibility is waning as physicians increasingly read journals online.
Another conundrum is how to inform the public about boxed warnings.
Manufacturers are prohibited from direct-to-consumer advertising of drugs with boxed warnings, although the warnings are easily found on the Internet. Some patients expect and welcome detailed information from their physicians, so it’s a good policy to always and repeatedly review this information with them, especially if they are members of an identified risk group. However, that policy may be counterproductive if it dissuades anxious patients from needed therapy despite risk-benefit considerations that strongly favor it. Boxed warnings are well known to have “spillover effects” in which the aspersions cast by a boxed warning for a relatively small subgroup of patients causes use of a drug to decline among all patients.
Compounding this conundrum is that physicians rarely have sufficient information to gauge the magnitude of a risk, given that boxed warnings are often based on information from surveillance systems that cannot accurately quantify the risk or even establish a causal relationship. The text of a boxed warning generally does not provide the information needed for evidence-based clinical practice such as a quantitative estimate of effect, information about source and trustworthiness of the evidence, and guidance on implementation. For these and other reasons, FDA policies about various boxed warnings have been the target of significant criticism.
Medication guides are one mechanism to address the challenge of informing patients about the risks of drugs they are taking. FDA-approved medication guides are available for most drugs dispensed as outpatient prescriptions, they’re written in plain language for the consumer, and they include paraphrased versions of any boxed warning. Ideally, patients review these guides with their physicians or pharmacists, but the guides may be lengthy and raise questions that may not be answerable (e.g., about incidence rates). Patients may decline to review this information when a drug is prescribed or dispensed, and they may discard printed copies given to them without reading.
What can physicians do to minimize boxed warning problems?
Physicians should periodically review the product labels for drugs they commonly prescribe, including drugs they’ve prescribed for a long time. Prescription renewal requests can be used as a prompt to check for changes in a patient’s condition or other medications that might place a patient in the target population of a boxed warning. Physicians can subscribe to newsletters that announce and discuss significant product label changes, including alerts directly from the FDA. Physicians may also enlist their office staff to find and review boxed warnings for drugs being prescribed, noting which ones should require a conversation with any patient who has been or will be receiving this drug. They may want to make explicit mention in their encounter record that a boxed warning, medication guide, or overall risk-benefit assessment has been discussed.
Summary
The nature of boxed warnings, the means by which they are disseminated, and their role in clinical practice are all in great need of improvement. Until that occurs, boxed warnings offer some, but only very limited, help to patients and physicians who struggle to understand the risks of medications.
Dr. Axelsen is professor in the departments of pharmacology, biochemistry, and biophysics, and of medicine, infectious diseases section, University of Pennsylvania, Philadelphia. He disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Almost all physicians write prescriptions, and each prescription requires a physician to assess the risks and benefits of the drug. If an adverse drug reaction occurs, physicians may be called on to defend their risk-benefit assessment in court.
The assessment of risk is complicated when there is a boxed warning that describes potentially serious and life-threatening adverse reactions associated with a drug. Some of our most commonly prescribed drugs have boxed warnings, and drugs that were initially approved by the Food and Drug Administration without boxed warnings may have them added years later.
One serious problem with boxed warnings is that there are no reliable mechanisms for making sure that physicians are aware of them. The warnings are typically not seen by physicians as printed product labels, just as physicians often don’t see the pills and capsules that they prescribe. Pharmacists who receive packaged drugs from manufacturers may be the only ones to see an actual printed boxed warning, but even those pharmacists have little reason to read each label and note changes when handling many bulk packages.
This problem is aggravated by misperceptions that many physicians have about boxed warnings and the increasingly intense scrutiny given to them by mass media and the courts. Lawyers can use boxed warnings to make a drug look dangerous, even when it’s not, and to make physicians look reckless when prescribing it. Therefore, it is important for physicians to understand what boxed warnings are, what they are not, the problems they cause, and how to minimize these problems.
What is a ‘boxed warning’?
The marketing and sale of drugs in the United States requires approval by the FDA. Approval requires manufacturers to prepare a document containing “Full Prescribing Information” for the drug and to include a printed copy in every package of the drug that is sold. This document is commonly called a “package insert,” but the FDA designates this document as the manufacturer’s product “label.”
In 1979, the FDA began requiring some labels to appear within thick, black rectangular borders; these have come to be known as boxed warnings. Boxed warnings are usually placed at the beginning of a label. They may be added to the label of a previously approved drug already on the market or included in the product label when first approved and marketed.
The requirement for a boxed warning most often arises when a signal appears during review of postmarketing surveillance data suggesting a possible and plausible association between a drug and an adverse reaction. Warnings may also be initiated in response to petitions from public interest groups, or upon the discovery of serious toxicity in animals. Regardless of their origin, the intent of a boxed warning is to highlight information that may have important therapeutic consequences and warrants heightened awareness among physicians.
What a boxed warning is not
A boxed warning is not “issued” by the FDA; it is merely required by the FDA. Specific wording or a template may be suggested by the FDA, but product labels and boxed warnings are written and issued by the manufacturer. This distinction may seem minor, but extensive litigation has occurred over whether manufacturers have met their duty to warn consumers about possible risks when using their products, and this duty cannot be shifted to the FDA.
A boxed warning may not be added to a product label at the option of a manufacturer. The FDA allows a boxed warning only if it requires the warning, to preserve its impact. It should be noted that some medical information sources (e.g., PDR.net) may include a “BOXED WARNING” in their drug monographs, but monographs not written by a manufacturer are not regulated by the FDA, and the text of their boxed warnings do not always correspond to the boxed warning that was approved by the FDA.
A boxed warning is not an indication that revocation of FDA approval is being considered or that it is likely to be revoked. FDA approval is subject to ongoing review and may be revoked at any time, without a prior boxed warning.
A boxed warning is not the highest level of warning. The FDA may require a manufacturer to send out a “Dear Health Care Provider” (DHCP) letter when an even higher or more urgent level of warning is deemed necessary. DHCP letters are usually accompanied by revisions of the product label, but most label revisions – and even most boxed warnings – are not accompanied by DHCP letters.
A boxed warning is not a statement about causation. Most warnings describe an “association” between a drug and an adverse effect, or “increased risk,” or instances of a particular adverse effect that “have been reported” in persons taking a drug. The words in a boxed warning are carefully chosen and require careful reading; in most cases they refrain from stating that a drug actually causes an adverse effect. The postmarketing surveillance data on which most warnings are based generally cannot provide the kind of evidence required to establish causation, and an association may be nothing more than an uncommon manifestation of the disorder for which the drug has been prescribed.
A boxed warning is not a statement about the probability of an adverse reaction occurring. The requirement for a boxed warning correlates better to the new recognition of a possible association than to the probability of an association. For example, penicillin has long been known to cause fatal anaphylaxis in 1/100,000 first-time administrations, but it does not have a boxed warning. The adverse consequences described in boxed warnings are often far less frequent – so much so that most physicians will never see them.
A boxed warning does not define the standard of care. The warning is a requirement imposed on the manufacturer, not on the practice of medicine. For legal purposes, the “standard of care” for the practice of medicine is defined state by state and is typically cast in terms such as “what most physicians would do in similar circumstances.” Physicians often prescribe drugs in spite of boxed warnings, just as they often prescribe drugs for “off label” indications, always balancing risk versus benefit.
A boxed warning does not constitute a contraindication to the use of a medication. Some warnings state that a drug is contraindicated in some situations, but product labels have another mandated section for listing contraindications, and most boxed warnings have no corresponding entry in that section.
A boxed warning does not necessarily constitute current information, nor is it always updated when new or contrary information becomes available. Revisions to boxed warnings, and to product labels in general, are made only after detailed review at the FDA, and the process of deciding whether an existing boxed warning continues to be appropriate may divert limited regulatory resources from more urgent priorities. Consequently, revisions to a boxed warning may lag behind the data that justify a revision by months or years. Revisions may never occur if softening or eliminating a boxed warning is deemed to be not worth the cost by a manufacturer.
Boxed warning problems for physicians
There is no reliable mechanism for manufacturers or the FDA to communicate boxed warnings directly to physicians, so it’s not clear how physicians are expected to stay informed about the issuance or revision of boxed warnings. They may first learn about new or revised warnings in the mass media, which is paying ever-increasing attention to press releases from the FDA. However, it can be difficult for the media to accurately convey the subtle and complex nature of a boxed warning in nontechnical terms.
Many physicians subscribe to various medical news alerts and attend continuing medical education (CME) programs, which often do an excellent job of highlighting new warnings, while hospitals, clinics, and pharmacies may broadcast news about boxed warnings in newsletters or other notices. But these notifications are ephemeral and may be missed by physicians who are overwhelmed by email, notices, newsletters, and CME programs.
The warnings that pop up in electronic medical records systems are often so numerous that physicians become trained to ignore them. Printed advertisements in professional journals must include mandated boxed warnings, but their visibility is waning as physicians increasingly read journals online.
Another conundrum is how to inform the public about boxed warnings.
Manufacturers are prohibited from direct-to-consumer advertising of drugs with boxed warnings, although the warnings are easily found on the Internet. Some patients expect and welcome detailed information from their physicians, so it’s a good policy to always and repeatedly review this information with them, especially if they are members of an identified risk group. However, that policy may be counterproductive if it dissuades anxious patients from needed therapy despite risk-benefit considerations that strongly favor it. Boxed warnings are well known to have “spillover effects” in which the aspersions cast by a boxed warning for a relatively small subgroup of patients causes use of a drug to decline among all patients.
Compounding this conundrum is that physicians rarely have sufficient information to gauge the magnitude of a risk, given that boxed warnings are often based on information from surveillance systems that cannot accurately quantify the risk or even establish a causal relationship. The text of a boxed warning generally does not provide the information needed for evidence-based clinical practice such as a quantitative estimate of effect, information about source and trustworthiness of the evidence, and guidance on implementation. For these and other reasons, FDA policies about various boxed warnings have been the target of significant criticism.
Medication guides are one mechanism to address the challenge of informing patients about the risks of drugs they are taking. FDA-approved medication guides are available for most drugs dispensed as outpatient prescriptions, they’re written in plain language for the consumer, and they include paraphrased versions of any boxed warning. Ideally, patients review these guides with their physicians or pharmacists, but the guides may be lengthy and raise questions that may not be answerable (e.g., about incidence rates). Patients may decline to review this information when a drug is prescribed or dispensed, and they may discard printed copies given to them without reading.
What can physicians do to minimize boxed warning problems?
Physicians should periodically review the product labels for drugs they commonly prescribe, including drugs they’ve prescribed for a long time. Prescription renewal requests can be used as a prompt to check for changes in a patient’s condition or other medications that might place a patient in the target population of a boxed warning. Physicians can subscribe to newsletters that announce and discuss significant product label changes, including alerts directly from the FDA. Physicians may also enlist their office staff to find and review boxed warnings for drugs being prescribed, noting which ones should require a conversation with any patient who has been or will be receiving this drug. They may want to make explicit mention in their encounter record that a boxed warning, medication guide, or overall risk-benefit assessment has been discussed.
Summary
The nature of boxed warnings, the means by which they are disseminated, and their role in clinical practice are all in great need of improvement. Until that occurs, boxed warnings offer some, but only very limited, help to patients and physicians who struggle to understand the risks of medications.
Dr. Axelsen is professor in the departments of pharmacology, biochemistry, and biophysics, and of medicine, infectious diseases section, University of Pennsylvania, Philadelphia. He disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Almost all physicians write prescriptions, and each prescription requires a physician to assess the risks and benefits of the drug. If an adverse drug reaction occurs, physicians may be called on to defend their risk-benefit assessment in court.
The assessment of risk is complicated when there is a boxed warning that describes potentially serious and life-threatening adverse reactions associated with a drug. Some of our most commonly prescribed drugs have boxed warnings, and drugs that were initially approved by the Food and Drug Administration without boxed warnings may have them added years later.
One serious problem with boxed warnings is that there are no reliable mechanisms for making sure that physicians are aware of them. The warnings are typically not seen by physicians as printed product labels, just as physicians often don’t see the pills and capsules that they prescribe. Pharmacists who receive packaged drugs from manufacturers may be the only ones to see an actual printed boxed warning, but even those pharmacists have little reason to read each label and note changes when handling many bulk packages.
This problem is aggravated by misperceptions that many physicians have about boxed warnings and the increasingly intense scrutiny given to them by mass media and the courts. Lawyers can use boxed warnings to make a drug look dangerous, even when it’s not, and to make physicians look reckless when prescribing it. Therefore, it is important for physicians to understand what boxed warnings are, what they are not, the problems they cause, and how to minimize these problems.
What is a ‘boxed warning’?
The marketing and sale of drugs in the United States requires approval by the FDA. Approval requires manufacturers to prepare a document containing “Full Prescribing Information” for the drug and to include a printed copy in every package of the drug that is sold. This document is commonly called a “package insert,” but the FDA designates this document as the manufacturer’s product “label.”
In 1979, the FDA began requiring some labels to appear within thick, black rectangular borders; these have come to be known as boxed warnings. Boxed warnings are usually placed at the beginning of a label. They may be added to the label of a previously approved drug already on the market or included in the product label when first approved and marketed.
The requirement for a boxed warning most often arises when a signal appears during review of postmarketing surveillance data suggesting a possible and plausible association between a drug and an adverse reaction. Warnings may also be initiated in response to petitions from public interest groups, or upon the discovery of serious toxicity in animals. Regardless of their origin, the intent of a boxed warning is to highlight information that may have important therapeutic consequences and warrants heightened awareness among physicians.
What a boxed warning is not
A boxed warning is not “issued” by the FDA; it is merely required by the FDA. Specific wording or a template may be suggested by the FDA, but product labels and boxed warnings are written and issued by the manufacturer. This distinction may seem minor, but extensive litigation has occurred over whether manufacturers have met their duty to warn consumers about possible risks when using their products, and this duty cannot be shifted to the FDA.
A boxed warning may not be added to a product label at the option of a manufacturer. The FDA allows a boxed warning only if it requires the warning, to preserve its impact. It should be noted that some medical information sources (e.g., PDR.net) may include a “BOXED WARNING” in their drug monographs, but monographs not written by a manufacturer are not regulated by the FDA, and the text of their boxed warnings do not always correspond to the boxed warning that was approved by the FDA.
A boxed warning is not an indication that revocation of FDA approval is being considered or that it is likely to be revoked. FDA approval is subject to ongoing review and may be revoked at any time, without a prior boxed warning.
A boxed warning is not the highest level of warning. The FDA may require a manufacturer to send out a “Dear Health Care Provider” (DHCP) letter when an even higher or more urgent level of warning is deemed necessary. DHCP letters are usually accompanied by revisions of the product label, but most label revisions – and even most boxed warnings – are not accompanied by DHCP letters.
A boxed warning is not a statement about causation. Most warnings describe an “association” between a drug and an adverse effect, or “increased risk,” or instances of a particular adverse effect that “have been reported” in persons taking a drug. The words in a boxed warning are carefully chosen and require careful reading; in most cases they refrain from stating that a drug actually causes an adverse effect. The postmarketing surveillance data on which most warnings are based generally cannot provide the kind of evidence required to establish causation, and an association may be nothing more than an uncommon manifestation of the disorder for which the drug has been prescribed.
A boxed warning is not a statement about the probability of an adverse reaction occurring. The requirement for a boxed warning correlates better to the new recognition of a possible association than to the probability of an association. For example, penicillin has long been known to cause fatal anaphylaxis in 1/100,000 first-time administrations, but it does not have a boxed warning. The adverse consequences described in boxed warnings are often far less frequent – so much so that most physicians will never see them.
A boxed warning does not define the standard of care. The warning is a requirement imposed on the manufacturer, not on the practice of medicine. For legal purposes, the “standard of care” for the practice of medicine is defined state by state and is typically cast in terms such as “what most physicians would do in similar circumstances.” Physicians often prescribe drugs in spite of boxed warnings, just as they often prescribe drugs for “off label” indications, always balancing risk versus benefit.
A boxed warning does not constitute a contraindication to the use of a medication. Some warnings state that a drug is contraindicated in some situations, but product labels have another mandated section for listing contraindications, and most boxed warnings have no corresponding entry in that section.
A boxed warning does not necessarily constitute current information, nor is it always updated when new or contrary information becomes available. Revisions to boxed warnings, and to product labels in general, are made only after detailed review at the FDA, and the process of deciding whether an existing boxed warning continues to be appropriate may divert limited regulatory resources from more urgent priorities. Consequently, revisions to a boxed warning may lag behind the data that justify a revision by months or years. Revisions may never occur if softening or eliminating a boxed warning is deemed to be not worth the cost by a manufacturer.
Boxed warning problems for physicians
There is no reliable mechanism for manufacturers or the FDA to communicate boxed warnings directly to physicians, so it’s not clear how physicians are expected to stay informed about the issuance or revision of boxed warnings. They may first learn about new or revised warnings in the mass media, which is paying ever-increasing attention to press releases from the FDA. However, it can be difficult for the media to accurately convey the subtle and complex nature of a boxed warning in nontechnical terms.
Many physicians subscribe to various medical news alerts and attend continuing medical education (CME) programs, which often do an excellent job of highlighting new warnings, while hospitals, clinics, and pharmacies may broadcast news about boxed warnings in newsletters or other notices. But these notifications are ephemeral and may be missed by physicians who are overwhelmed by email, notices, newsletters, and CME programs.
The warnings that pop up in electronic medical records systems are often so numerous that physicians become trained to ignore them. Printed advertisements in professional journals must include mandated boxed warnings, but their visibility is waning as physicians increasingly read journals online.
Another conundrum is how to inform the public about boxed warnings.
Manufacturers are prohibited from direct-to-consumer advertising of drugs with boxed warnings, although the warnings are easily found on the Internet. Some patients expect and welcome detailed information from their physicians, so it’s a good policy to always and repeatedly review this information with them, especially if they are members of an identified risk group. However, that policy may be counterproductive if it dissuades anxious patients from needed therapy despite risk-benefit considerations that strongly favor it. Boxed warnings are well known to have “spillover effects” in which the aspersions cast by a boxed warning for a relatively small subgroup of patients causes use of a drug to decline among all patients.
Compounding this conundrum is that physicians rarely have sufficient information to gauge the magnitude of a risk, given that boxed warnings are often based on information from surveillance systems that cannot accurately quantify the risk or even establish a causal relationship. The text of a boxed warning generally does not provide the information needed for evidence-based clinical practice such as a quantitative estimate of effect, information about source and trustworthiness of the evidence, and guidance on implementation. For these and other reasons, FDA policies about various boxed warnings have been the target of significant criticism.
Medication guides are one mechanism to address the challenge of informing patients about the risks of drugs they are taking. FDA-approved medication guides are available for most drugs dispensed as outpatient prescriptions, they’re written in plain language for the consumer, and they include paraphrased versions of any boxed warning. Ideally, patients review these guides with their physicians or pharmacists, but the guides may be lengthy and raise questions that may not be answerable (e.g., about incidence rates). Patients may decline to review this information when a drug is prescribed or dispensed, and they may discard printed copies given to them without reading.
What can physicians do to minimize boxed warning problems?
Physicians should periodically review the product labels for drugs they commonly prescribe, including drugs they’ve prescribed for a long time. Prescription renewal requests can be used as a prompt to check for changes in a patient’s condition or other medications that might place a patient in the target population of a boxed warning. Physicians can subscribe to newsletters that announce and discuss significant product label changes, including alerts directly from the FDA. Physicians may also enlist their office staff to find and review boxed warnings for drugs being prescribed, noting which ones should require a conversation with any patient who has been or will be receiving this drug. They may want to make explicit mention in their encounter record that a boxed warning, medication guide, or overall risk-benefit assessment has been discussed.
Summary
The nature of boxed warnings, the means by which they are disseminated, and their role in clinical practice are all in great need of improvement. Until that occurs, boxed warnings offer some, but only very limited, help to patients and physicians who struggle to understand the risks of medications.
Dr. Axelsen is professor in the departments of pharmacology, biochemistry, and biophysics, and of medicine, infectious diseases section, University of Pennsylvania, Philadelphia. He disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
Is telemedicine here to stay?
Dear colleagues and friends,
I am fortunate to receive the baton from Charles Kahi, MD, in facilitating the fascinating and timely debates that have characterized the AGA Perspective series. Favorable reimbursement changes and the need for social distancing fast-tracked telemedicine, a care delivery model that had been slowly evolving.
In this month’s Perspective column, Dr. Hernaez and Dr. Vaughn discuss the pros and cons of telemedicine in GI. Is it the new office visit? Or simply just good enough for when we really need it? I look forward to hearing your thoughts and experiences on the AGA Community forum as well as by email (ginews@gastro.org).
Gyanprakash A. Ketwaroo, MD, is assistant professor of medicine at Baylor College of Medicine, Houston. He is an associate editor for GI & Hepatology News.
It holds promise
BY RUBEN HERNAEZ, MD, MPH, PHD
It was around January 2020, when COVID-19 was something far, far away, and not particularly worrisome. I was performing a routine care visit of one of my out-of-state patients waitlisted for a liver transplant. All was going fine until he stated, “Hey doc, while I appreciate your time and visits, these travels to Houston are quite inconvenient for my family and me: It is a logistical ordeal, and my wife is always afraid of catching something in the airplane. Could you do the same remotely such as a videoconference?” And just like that, it sparked my interest in how to maintain his liver transplant care from a distance. The Federation of State Medical Boards defines telemedicine as “the practice of medicine using electronic communication, information technology, or other means between a physician in one location, and a patient in another location, with or without an intervening health care provider.1 What my patient was asking was to use a mode of telemedicine – a video visit – to receive the same quality of care. He brought up three critical points that I will discuss further: access to specialty care (such as transplant hepatology), reduction of costs (time and money), and improved patient satisfaction.
Arora and colleagues pioneered the Extension for Community Healthcare Outcomes (ECHO) project, providing complex specialty medical care to underserved populations through a model of team-based interdisciplinary development in hepatitis C infection treatment in underserved communities, with cure rates similar to the university settings.1 The University of Michigan–Veterans Affairs Medical Center used a similar approach, called Specialty Care Access Network–Extension of Community Healthcare Outcome (SCAN‐ECHO). They showed that telemedicine improved survival in 513 patients evaluated in this program compared to regular care (hazard ratio [HR]of 0.54, 95% confidence interval 0.36‐0.81, P = .003).2 So the evidence backs my patient’s request in providing advanced medical care using a telemedicine platform.
An extra benefit of telemedicine in the current climate crisis is reducing the carbon footprint: There’s no need to travel. Telemedicine has been shown to be cost effective: A study of claims data from Jefferson Health reported that patients who received care from an on-demand telemedicine program had net cost savings per telemedicine visit between $19 and $121 per visit compared with traditional in-person visits.3 Using telemonitoring, a form of telemedicine, Bloom et al. showed in 100 simulated patients with cirrhosis and ascites over a 6-month horizon that standard of care was $167,500 more expensive than telemonitoring. The net savings of telemonitoring was always superior in different clinical scenarios.4
Further, our patients significantly decrease travel time (almost instant), improve compliance with medical appointments (more flexibility), and no more headaches related to parking or getting lost around the medical campus. Not surprisingly, these perks from telemedicine are associated with patient-reported outcomes. Reed and colleagues reported patients’ experience with video telemedicine visits in Kaiser Permanente Northern California (n = 1,274) and showed that “67% generally needed to make one or more arrangements to attend an in-person office visit (55% time off from work, 29% coverage for another activity or responsibility, 15% child care or caregiving, and 10% another person to accompany them)”; in contrast, 87% reported a video visit as “more convenient for me,” and 93% stated that “my video visit adequately addressed my needs.”5 In liver transplantation, John et al. showed that, in the Veterans Health Administration, telehealth was associated with a significantly shorter time on the liver transplant waitlist (138.8 vs. 249 days), reduction in the time from referral to evaluation (HR, 0.15; 95% CI, 0.09-0.21; P < .01) and listing (HR, 0.26; 95% CI, 0.12-0.40; P < .01) in a study of 232 patients with advanced cirrhosis.6
So, should I change my approach to patients undergoing care for chronic liver/gastrointestinal diseases? I think so. Telemedicine and its tools provide clear benefits to our patients by increasing access to care, time and money savings, and satisfaction. I am fortunate to work within the largest healthcare network in the Nation – the Veterans Health Administration – and therefore, I can cross state lines to provide medical care/advice using the video-visit tool (VA VideoConnect). One could argue that some patients might find it challenging to access telemedicine appointments, but with adequate coaching or support from our teams, telemedicine visits are a click away.
Going back to my patient, I embraced his request and coached him on using VA VideoConnect. We can continue his waitlist medical care in the following months despite the COVID-19 pandemic using telemedicine. I can assess his asterixis and ascites via his cellphone; his primary care team fills in the vitals and labs to complete a virtual visit. There’s no question in my mind that telemedicine is here to stay and that we will continue to adapt e-health tools into video visits (for example, integrating vitals, measurement of frailty, and remote monitoring). The future of our specialty is here, and I envision we will eventually have home-based hospitalizations with daily virtual rounds.
Dr. Hernaez is with the section of gastroenterology and the Center for Innovations in Quality, Effectiveness, and Safety at the Michael E. DeBakey Veterans Affairs Medical Center and in the section of gastroenterology and hepatology in the department of medicine at Baylor College of Medicine, both in Houston. He has no relevant conflicts of interest.
References
1. Arora S et al. N Engl J Med. 2011 Jun 9;364(23):2199-207.
2. Su GL et al. Hepatology. 2018 Dec;68(6):2317-24.
3. Nord G et al. Am J Emerg Med. 2019 May;37(5):890-4.
4. Bloom PP et al. Dig Dis Sci. 2021. doi: 10.1007/s10620-021-07013-2.
5. Reed ME et al. Ann Intern Med. 2019 Aug;171:222-4.
6. John BV et al. Clin Gastroenterol Hepatol. 2020 Jul;18:1822-30.e4.
It has its limits
BY BYRON P. VAUGHN, MD, MS
The post-pandemic world will include telehealth. Technology disrupts business as usual and often brings positive change. But there are consequences. To employ telehealth into routine care equitably and effectively within a gastroenterology practice, we should consider two general questions: “Was the care I provided the same quality as if the patient was seen in person?” and more broadly, “am I satisfied with my practice’s implementation of telehealth?” This perspective will highlight several areas affecting gastroenterology care: lack of physical exam, disproportionate impact on certain populations, development of a patient-provider relationship, impact on physician well-being, and potential financial ramifications. We will all have to adapt to telemedicine to some extent. Understanding the trade-offs of this technology can help us position effectively in a gastroenterology practice.
Perhaps the most obvious limitation of telemedicine is the lack of vital signs and physical exam. Determining if a patient is “sick or not” is often one of the first lessons trainees learn overnight. Vital signs and physical exam are crucial in the complex triaging that occurs when evaluating for diagnoses with potentially urgent interventions. While most outpatient gastroenterology clinics are not evaluating an acute abdomen, in the correct context, the physical exam provides important nuance and often reassurance. My personal estimate is that 90% of a diagnosis is based on the history. But without physical contact, providers may increase costly downstream diagnostic testing or referrals to the emergency department. Increasing use of at-home or wearable health technology could help but requires system investments in infrastructure to implement.
Telemedicine requires a baseline level of equipment and knowledge to participate. A variety of populations will have either a knowledge gap or technology gap. Lack of rural high-speed Internet can lead to poor video quality, inhibiting effective communication and frustrating both provider and patient. In urban areas, there is a drastic wealth divide, and some groups may have difficulty obtaining sufficient equipment to complete a video visit. Even with adequate infrastructure and equipment, certain groups may be disadvantaged because of a lack of the technological savvy or literacy needed to navigate a virtual visit. The addition of interpreter services adds complexity to communication on top of the virtual interaction. These technology and knowledge gaps can produce confusion and potentially lead to worse care.1 Careful selection of appropriate patients for telemedicine is essential. Is the quality of care over a virtual visit the same for a business executive as that of a non-English speaking refugee?
The term “webside manner” precedes the pandemic but will be important in the lexicon of doctor-patient relationships moving forward. We routinely train physicians about the importance of small actions to improve our bedside manner, such as sitting down, reacting to body language, and making eye contact. First impressions matter in relationship building. For many of my established IBD patients, I can easily hop into a comfortable repertoire in person, virtually, or even on the phone. In addition, I know these people well enough to trust that I am providing the same level of care regardless of visit medium. However, a new patient virtual encounter requires nuance. I have met new patients while they are driving (I requested the patient park!), in public places, and at work. Despite instructions given to patients about the appropriate location for a virtual visit, the patient location is not in our control. For some patients this may increase the comfort of the visit. However, for others, it can lead to distractions or potentially limit the amount of information a patient is willing to share. Forming a patient-provider relationship virtually will require a new set of skills and specific training for many practitioners.
Telehealth can contribute to provider burnout. While a busy in-person clinic can be exhausting, I have found I can be more exhausted after a half-day of virtual clinic. There is an element of human connection that is difficult to replace online.2 On top of that loss, video visits are more psychologically demanding than in-person interactions. I also spend more time in a chair, have fewer coffee breaks, and have fewer professional interactions with the clinic staff and professional colleagues. Several other micro-stressors exist in virtual care that may make “Zoom fatigue” a real occupational hazard.3
Lastly, there are implications on reimbursement with telehealth. In Minnesota, a 2015 telemedicine law required private and state employee health plans to provide the same coverage for telemedicine as in-person visits, although patients had to drive to a clinic or facility to use secure telehealth equipment and have vital signs taken. With the pandemic, this stipulation is waived, and it seems likely to become permanent. However, reimbursement questions will arise, as there is a perception that a 30-minute telephone call should not cost as much as a 30-minute in-person visit, regardless of the content of the conversation.
We will have to learn to move forward with telehealth. The strength of telehealth is likely in patients with chronic, well controlled diseases, who have frequent interactions with health care. Examples of this include (although certainly are not limited to) established patients with well-controlled IBD, non-cirrhotic liver disease, and irritable bowel syndrome. Triaging patients who need in-person evaluation, ensuring patient and provider well-being, and creating a financially sustainable model of care are yet unresolved issues. Providers will likely vary in their personal acceptance of telehealth and will need to advocate within their own systems to obtain a hybrid model of telehealth that maximizes quality of care with job satisfaction.
Dr. Vaughn is associate professor of medicine and codirector of the inflammatory bowel disease program in the division of gastroenterology, hepatology, and nutrition at University of Minnesota, Minneapolis. He has received consulting fees from Prometheus and research support from Roche, Takeda, Celgene, Diasorin, and Crestovo.
References
1. George S et al. Stud Health Technol Inform. 2013;192:946.
2. Blank S. “What’s missing from Zoom reminds us what it means to be human,” 2020 Apr 27, Medium.
3. Williams N. Occup Med (Lond). 2021 Apr. doi: 10.1093/occmed/kqab041.
Dear colleagues and friends,
I am fortunate to receive the baton from Charles Kahi, MD, in facilitating the fascinating and timely debates that have characterized the AGA Perspective series. Favorable reimbursement changes and the need for social distancing fast-tracked telemedicine, a care delivery model that had been slowly evolving.
In this month’s Perspective column, Dr. Hernaez and Dr. Vaughn discuss the pros and cons of telemedicine in GI. Is it the new office visit? Or simply just good enough for when we really need it? I look forward to hearing your thoughts and experiences on the AGA Community forum as well as by email (ginews@gastro.org).
Gyanprakash A. Ketwaroo, MD, is assistant professor of medicine at Baylor College of Medicine, Houston. He is an associate editor for GI & Hepatology News.
It holds promise
BY RUBEN HERNAEZ, MD, MPH, PHD
It was around January 2020, when COVID-19 was something far, far away, and not particularly worrisome. I was performing a routine care visit of one of my out-of-state patients waitlisted for a liver transplant. All was going fine until he stated, “Hey doc, while I appreciate your time and visits, these travels to Houston are quite inconvenient for my family and me: It is a logistical ordeal, and my wife is always afraid of catching something in the airplane. Could you do the same remotely such as a videoconference?” And just like that, it sparked my interest in how to maintain his liver transplant care from a distance. The Federation of State Medical Boards defines telemedicine as “the practice of medicine using electronic communication, information technology, or other means between a physician in one location, and a patient in another location, with or without an intervening health care provider.1 What my patient was asking was to use a mode of telemedicine – a video visit – to receive the same quality of care. He brought up three critical points that I will discuss further: access to specialty care (such as transplant hepatology), reduction of costs (time and money), and improved patient satisfaction.
Arora and colleagues pioneered the Extension for Community Healthcare Outcomes (ECHO) project, providing complex specialty medical care to underserved populations through a model of team-based interdisciplinary development in hepatitis C infection treatment in underserved communities, with cure rates similar to the university settings.1 The University of Michigan–Veterans Affairs Medical Center used a similar approach, called Specialty Care Access Network–Extension of Community Healthcare Outcome (SCAN‐ECHO). They showed that telemedicine improved survival in 513 patients evaluated in this program compared to regular care (hazard ratio [HR]of 0.54, 95% confidence interval 0.36‐0.81, P = .003).2 So the evidence backs my patient’s request in providing advanced medical care using a telemedicine platform.
An extra benefit of telemedicine in the current climate crisis is reducing the carbon footprint: There’s no need to travel. Telemedicine has been shown to be cost effective: A study of claims data from Jefferson Health reported that patients who received care from an on-demand telemedicine program had net cost savings per telemedicine visit between $19 and $121 per visit compared with traditional in-person visits.3 Using telemonitoring, a form of telemedicine, Bloom et al. showed in 100 simulated patients with cirrhosis and ascites over a 6-month horizon that standard of care was $167,500 more expensive than telemonitoring. The net savings of telemonitoring was always superior in different clinical scenarios.4
Further, our patients significantly decrease travel time (almost instant), improve compliance with medical appointments (more flexibility), and no more headaches related to parking or getting lost around the medical campus. Not surprisingly, these perks from telemedicine are associated with patient-reported outcomes. Reed and colleagues reported patients’ experience with video telemedicine visits in Kaiser Permanente Northern California (n = 1,274) and showed that “67% generally needed to make one or more arrangements to attend an in-person office visit (55% time off from work, 29% coverage for another activity or responsibility, 15% child care or caregiving, and 10% another person to accompany them)”; in contrast, 87% reported a video visit as “more convenient for me,” and 93% stated that “my video visit adequately addressed my needs.”5 In liver transplantation, John et al. showed that, in the Veterans Health Administration, telehealth was associated with a significantly shorter time on the liver transplant waitlist (138.8 vs. 249 days), reduction in the time from referral to evaluation (HR, 0.15; 95% CI, 0.09-0.21; P < .01) and listing (HR, 0.26; 95% CI, 0.12-0.40; P < .01) in a study of 232 patients with advanced cirrhosis.6
So, should I change my approach to patients undergoing care for chronic liver/gastrointestinal diseases? I think so. Telemedicine and its tools provide clear benefits to our patients by increasing access to care, time and money savings, and satisfaction. I am fortunate to work within the largest healthcare network in the Nation – the Veterans Health Administration – and therefore, I can cross state lines to provide medical care/advice using the video-visit tool (VA VideoConnect). One could argue that some patients might find it challenging to access telemedicine appointments, but with adequate coaching or support from our teams, telemedicine visits are a click away.
Going back to my patient, I embraced his request and coached him on using VA VideoConnect. We can continue his waitlist medical care in the following months despite the COVID-19 pandemic using telemedicine. I can assess his asterixis and ascites via his cellphone; his primary care team fills in the vitals and labs to complete a virtual visit. There’s no question in my mind that telemedicine is here to stay and that we will continue to adapt e-health tools into video visits (for example, integrating vitals, measurement of frailty, and remote monitoring). The future of our specialty is here, and I envision we will eventually have home-based hospitalizations with daily virtual rounds.
Dr. Hernaez is with the section of gastroenterology and the Center for Innovations in Quality, Effectiveness, and Safety at the Michael E. DeBakey Veterans Affairs Medical Center and in the section of gastroenterology and hepatology in the department of medicine at Baylor College of Medicine, both in Houston. He has no relevant conflicts of interest.
References
1. Arora S et al. N Engl J Med. 2011 Jun 9;364(23):2199-207.
2. Su GL et al. Hepatology. 2018 Dec;68(6):2317-24.
3. Nord G et al. Am J Emerg Med. 2019 May;37(5):890-4.
4. Bloom PP et al. Dig Dis Sci. 2021. doi: 10.1007/s10620-021-07013-2.
5. Reed ME et al. Ann Intern Med. 2019 Aug;171:222-4.
6. John BV et al. Clin Gastroenterol Hepatol. 2020 Jul;18:1822-30.e4.
It has its limits
BY BYRON P. VAUGHN, MD, MS
The post-pandemic world will include telehealth. Technology disrupts business as usual and often brings positive change. But there are consequences. To employ telehealth into routine care equitably and effectively within a gastroenterology practice, we should consider two general questions: “Was the care I provided the same quality as if the patient was seen in person?” and more broadly, “am I satisfied with my practice’s implementation of telehealth?” This perspective will highlight several areas affecting gastroenterology care: lack of physical exam, disproportionate impact on certain populations, development of a patient-provider relationship, impact on physician well-being, and potential financial ramifications. We will all have to adapt to telemedicine to some extent. Understanding the trade-offs of this technology can help us position effectively in a gastroenterology practice.
Perhaps the most obvious limitation of telemedicine is the lack of vital signs and physical exam. Determining if a patient is “sick or not” is often one of the first lessons trainees learn overnight. Vital signs and physical exam are crucial in the complex triaging that occurs when evaluating for diagnoses with potentially urgent interventions. While most outpatient gastroenterology clinics are not evaluating an acute abdomen, in the correct context, the physical exam provides important nuance and often reassurance. My personal estimate is that 90% of a diagnosis is based on the history. But without physical contact, providers may increase costly downstream diagnostic testing or referrals to the emergency department. Increasing use of at-home or wearable health technology could help but requires system investments in infrastructure to implement.
Telemedicine requires a baseline level of equipment and knowledge to participate. A variety of populations will have either a knowledge gap or technology gap. Lack of rural high-speed Internet can lead to poor video quality, inhibiting effective communication and frustrating both provider and patient. In urban areas, there is a drastic wealth divide, and some groups may have difficulty obtaining sufficient equipment to complete a video visit. Even with adequate infrastructure and equipment, certain groups may be disadvantaged because of a lack of the technological savvy or literacy needed to navigate a virtual visit. The addition of interpreter services adds complexity to communication on top of the virtual interaction. These technology and knowledge gaps can produce confusion and potentially lead to worse care.1 Careful selection of appropriate patients for telemedicine is essential. Is the quality of care over a virtual visit the same for a business executive as that of a non-English speaking refugee?
The term “webside manner” precedes the pandemic but will be important in the lexicon of doctor-patient relationships moving forward. We routinely train physicians about the importance of small actions to improve our bedside manner, such as sitting down, reacting to body language, and making eye contact. First impressions matter in relationship building. For many of my established IBD patients, I can easily hop into a comfortable repertoire in person, virtually, or even on the phone. In addition, I know these people well enough to trust that I am providing the same level of care regardless of visit medium. However, a new patient virtual encounter requires nuance. I have met new patients while they are driving (I requested the patient park!), in public places, and at work. Despite instructions given to patients about the appropriate location for a virtual visit, the patient location is not in our control. For some patients this may increase the comfort of the visit. However, for others, it can lead to distractions or potentially limit the amount of information a patient is willing to share. Forming a patient-provider relationship virtually will require a new set of skills and specific training for many practitioners.
Telehealth can contribute to provider burnout. While a busy in-person clinic can be exhausting, I have found I can be more exhausted after a half-day of virtual clinic. There is an element of human connection that is difficult to replace online.2 On top of that loss, video visits are more psychologically demanding than in-person interactions. I also spend more time in a chair, have fewer coffee breaks, and have fewer professional interactions with the clinic staff and professional colleagues. Several other micro-stressors exist in virtual care that may make “Zoom fatigue” a real occupational hazard.3
Lastly, there are implications on reimbursement with telehealth. In Minnesota, a 2015 telemedicine law required private and state employee health plans to provide the same coverage for telemedicine as in-person visits, although patients had to drive to a clinic or facility to use secure telehealth equipment and have vital signs taken. With the pandemic, this stipulation is waived, and it seems likely to become permanent. However, reimbursement questions will arise, as there is a perception that a 30-minute telephone call should not cost as much as a 30-minute in-person visit, regardless of the content of the conversation.
We will have to learn to move forward with telehealth. The strength of telehealth is likely in patients with chronic, well controlled diseases, who have frequent interactions with health care. Examples of this include (although certainly are not limited to) established patients with well-controlled IBD, non-cirrhotic liver disease, and irritable bowel syndrome. Triaging patients who need in-person evaluation, ensuring patient and provider well-being, and creating a financially sustainable model of care are yet unresolved issues. Providers will likely vary in their personal acceptance of telehealth and will need to advocate within their own systems to obtain a hybrid model of telehealth that maximizes quality of care with job satisfaction.
Dr. Vaughn is associate professor of medicine and codirector of the inflammatory bowel disease program in the division of gastroenterology, hepatology, and nutrition at University of Minnesota, Minneapolis. He has received consulting fees from Prometheus and research support from Roche, Takeda, Celgene, Diasorin, and Crestovo.
References
1. George S et al. Stud Health Technol Inform. 2013;192:946.
2. Blank S. “What’s missing from Zoom reminds us what it means to be human,” 2020 Apr 27, Medium.
3. Williams N. Occup Med (Lond). 2021 Apr. doi: 10.1093/occmed/kqab041.
Dear colleagues and friends,
I am fortunate to receive the baton from Charles Kahi, MD, in facilitating the fascinating and timely debates that have characterized the AGA Perspective series. Favorable reimbursement changes and the need for social distancing fast-tracked telemedicine, a care delivery model that had been slowly evolving.
In this month’s Perspective column, Dr. Hernaez and Dr. Vaughn discuss the pros and cons of telemedicine in GI. Is it the new office visit? Or simply just good enough for when we really need it? I look forward to hearing your thoughts and experiences on the AGA Community forum as well as by email (ginews@gastro.org).
Gyanprakash A. Ketwaroo, MD, is assistant professor of medicine at Baylor College of Medicine, Houston. He is an associate editor for GI & Hepatology News.
It holds promise
BY RUBEN HERNAEZ, MD, MPH, PHD
It was around January 2020, when COVID-19 was something far, far away, and not particularly worrisome. I was performing a routine care visit of one of my out-of-state patients waitlisted for a liver transplant. All was going fine until he stated, “Hey doc, while I appreciate your time and visits, these travels to Houston are quite inconvenient for my family and me: It is a logistical ordeal, and my wife is always afraid of catching something in the airplane. Could you do the same remotely such as a videoconference?” And just like that, it sparked my interest in how to maintain his liver transplant care from a distance. The Federation of State Medical Boards defines telemedicine as “the practice of medicine using electronic communication, information technology, or other means between a physician in one location, and a patient in another location, with or without an intervening health care provider.1 What my patient was asking was to use a mode of telemedicine – a video visit – to receive the same quality of care. He brought up three critical points that I will discuss further: access to specialty care (such as transplant hepatology), reduction of costs (time and money), and improved patient satisfaction.
Arora and colleagues pioneered the Extension for Community Healthcare Outcomes (ECHO) project, providing complex specialty medical care to underserved populations through a model of team-based interdisciplinary development in hepatitis C infection treatment in underserved communities, with cure rates similar to the university settings.1 The University of Michigan–Veterans Affairs Medical Center used a similar approach, called Specialty Care Access Network–Extension of Community Healthcare Outcome (SCAN‐ECHO). They showed that telemedicine improved survival in 513 patients evaluated in this program compared to regular care (hazard ratio [HR]of 0.54, 95% confidence interval 0.36‐0.81, P = .003).2 So the evidence backs my patient’s request in providing advanced medical care using a telemedicine platform.
An extra benefit of telemedicine in the current climate crisis is reducing the carbon footprint: There’s no need to travel. Telemedicine has been shown to be cost effective: A study of claims data from Jefferson Health reported that patients who received care from an on-demand telemedicine program had net cost savings per telemedicine visit between $19 and $121 per visit compared with traditional in-person visits.3 Using telemonitoring, a form of telemedicine, Bloom et al. showed in 100 simulated patients with cirrhosis and ascites over a 6-month horizon that standard of care was $167,500 more expensive than telemonitoring. The net savings of telemonitoring was always superior in different clinical scenarios.4
Further, our patients significantly decrease travel time (almost instant), improve compliance with medical appointments (more flexibility), and no more headaches related to parking or getting lost around the medical campus. Not surprisingly, these perks from telemedicine are associated with patient-reported outcomes. Reed and colleagues reported patients’ experience with video telemedicine visits in Kaiser Permanente Northern California (n = 1,274) and showed that “67% generally needed to make one or more arrangements to attend an in-person office visit (55% time off from work, 29% coverage for another activity or responsibility, 15% child care or caregiving, and 10% another person to accompany them)”; in contrast, 87% reported a video visit as “more convenient for me,” and 93% stated that “my video visit adequately addressed my needs.”5 In liver transplantation, John et al. showed that, in the Veterans Health Administration, telehealth was associated with a significantly shorter time on the liver transplant waitlist (138.8 vs. 249 days), reduction in the time from referral to evaluation (HR, 0.15; 95% CI, 0.09-0.21; P < .01) and listing (HR, 0.26; 95% CI, 0.12-0.40; P < .01) in a study of 232 patients with advanced cirrhosis.6
So, should I change my approach to patients undergoing care for chronic liver/gastrointestinal diseases? I think so. Telemedicine and its tools provide clear benefits to our patients by increasing access to care, time and money savings, and satisfaction. I am fortunate to work within the largest healthcare network in the Nation – the Veterans Health Administration – and therefore, I can cross state lines to provide medical care/advice using the video-visit tool (VA VideoConnect). One could argue that some patients might find it challenging to access telemedicine appointments, but with adequate coaching or support from our teams, telemedicine visits are a click away.
Going back to my patient, I embraced his request and coached him on using VA VideoConnect. We can continue his waitlist medical care in the following months despite the COVID-19 pandemic using telemedicine. I can assess his asterixis and ascites via his cellphone; his primary care team fills in the vitals and labs to complete a virtual visit. There’s no question in my mind that telemedicine is here to stay and that we will continue to adapt e-health tools into video visits (for example, integrating vitals, measurement of frailty, and remote monitoring). The future of our specialty is here, and I envision we will eventually have home-based hospitalizations with daily virtual rounds.
Dr. Hernaez is with the section of gastroenterology and the Center for Innovations in Quality, Effectiveness, and Safety at the Michael E. DeBakey Veterans Affairs Medical Center and in the section of gastroenterology and hepatology in the department of medicine at Baylor College of Medicine, both in Houston. He has no relevant conflicts of interest.
References
1. Arora S et al. N Engl J Med. 2011 Jun 9;364(23):2199-207.
2. Su GL et al. Hepatology. 2018 Dec;68(6):2317-24.
3. Nord G et al. Am J Emerg Med. 2019 May;37(5):890-4.
4. Bloom PP et al. Dig Dis Sci. 2021. doi: 10.1007/s10620-021-07013-2.
5. Reed ME et al. Ann Intern Med. 2019 Aug;171:222-4.
6. John BV et al. Clin Gastroenterol Hepatol. 2020 Jul;18:1822-30.e4.
It has its limits
BY BYRON P. VAUGHN, MD, MS
The post-pandemic world will include telehealth. Technology disrupts business as usual and often brings positive change. But there are consequences. To employ telehealth into routine care equitably and effectively within a gastroenterology practice, we should consider two general questions: “Was the care I provided the same quality as if the patient was seen in person?” and more broadly, “am I satisfied with my practice’s implementation of telehealth?” This perspective will highlight several areas affecting gastroenterology care: lack of physical exam, disproportionate impact on certain populations, development of a patient-provider relationship, impact on physician well-being, and potential financial ramifications. We will all have to adapt to telemedicine to some extent. Understanding the trade-offs of this technology can help us position effectively in a gastroenterology practice.
Perhaps the most obvious limitation of telemedicine is the lack of vital signs and physical exam. Determining if a patient is “sick or not” is often one of the first lessons trainees learn overnight. Vital signs and physical exam are crucial in the complex triaging that occurs when evaluating for diagnoses with potentially urgent interventions. While most outpatient gastroenterology clinics are not evaluating an acute abdomen, in the correct context, the physical exam provides important nuance and often reassurance. My personal estimate is that 90% of a diagnosis is based on the history. But without physical contact, providers may increase costly downstream diagnostic testing or referrals to the emergency department. Increasing use of at-home or wearable health technology could help but requires system investments in infrastructure to implement.
Telemedicine requires a baseline level of equipment and knowledge to participate. A variety of populations will have either a knowledge gap or technology gap. Lack of rural high-speed Internet can lead to poor video quality, inhibiting effective communication and frustrating both provider and patient. In urban areas, there is a drastic wealth divide, and some groups may have difficulty obtaining sufficient equipment to complete a video visit. Even with adequate infrastructure and equipment, certain groups may be disadvantaged because of a lack of the technological savvy or literacy needed to navigate a virtual visit. The addition of interpreter services adds complexity to communication on top of the virtual interaction. These technology and knowledge gaps can produce confusion and potentially lead to worse care.1 Careful selection of appropriate patients for telemedicine is essential. Is the quality of care over a virtual visit the same for a business executive as that of a non-English speaking refugee?
The term “webside manner” precedes the pandemic but will be important in the lexicon of doctor-patient relationships moving forward. We routinely train physicians about the importance of small actions to improve our bedside manner, such as sitting down, reacting to body language, and making eye contact. First impressions matter in relationship building. For many of my established IBD patients, I can easily hop into a comfortable repertoire in person, virtually, or even on the phone. In addition, I know these people well enough to trust that I am providing the same level of care regardless of visit medium. However, a new patient virtual encounter requires nuance. I have met new patients while they are driving (I requested the patient park!), in public places, and at work. Despite instructions given to patients about the appropriate location for a virtual visit, the patient location is not in our control. For some patients this may increase the comfort of the visit. However, for others, it can lead to distractions or potentially limit the amount of information a patient is willing to share. Forming a patient-provider relationship virtually will require a new set of skills and specific training for many practitioners.
Telehealth can contribute to provider burnout. While a busy in-person clinic can be exhausting, I have found I can be more exhausted after a half-day of virtual clinic. There is an element of human connection that is difficult to replace online.2 On top of that loss, video visits are more psychologically demanding than in-person interactions. I also spend more time in a chair, have fewer coffee breaks, and have fewer professional interactions with the clinic staff and professional colleagues. Several other micro-stressors exist in virtual care that may make “Zoom fatigue” a real occupational hazard.3
Lastly, there are implications on reimbursement with telehealth. In Minnesota, a 2015 telemedicine law required private and state employee health plans to provide the same coverage for telemedicine as in-person visits, although patients had to drive to a clinic or facility to use secure telehealth equipment and have vital signs taken. With the pandemic, this stipulation is waived, and it seems likely to become permanent. However, reimbursement questions will arise, as there is a perception that a 30-minute telephone call should not cost as much as a 30-minute in-person visit, regardless of the content of the conversation.
We will have to learn to move forward with telehealth. The strength of telehealth is likely in patients with chronic, well controlled diseases, who have frequent interactions with health care. Examples of this include (although certainly are not limited to) established patients with well-controlled IBD, non-cirrhotic liver disease, and irritable bowel syndrome. Triaging patients who need in-person evaluation, ensuring patient and provider well-being, and creating a financially sustainable model of care are yet unresolved issues. Providers will likely vary in their personal acceptance of telehealth and will need to advocate within their own systems to obtain a hybrid model of telehealth that maximizes quality of care with job satisfaction.
Dr. Vaughn is associate professor of medicine and codirector of the inflammatory bowel disease program in the division of gastroenterology, hepatology, and nutrition at University of Minnesota, Minneapolis. He has received consulting fees from Prometheus and research support from Roche, Takeda, Celgene, Diasorin, and Crestovo.
References
1. George S et al. Stud Health Technol Inform. 2013;192:946.
2. Blank S. “What’s missing from Zoom reminds us what it means to be human,” 2020 Apr 27, Medium.
3. Williams N. Occup Med (Lond). 2021 Apr. doi: 10.1093/occmed/kqab041.
80% of Americans research recommendations post-visit
Confusion over health information and doctor advice is even higher among people who care for patients than among those who don’t provide care to their loved ones, the nationally representative survey from the AHIMA Foundation found.
The survey also shows that 80% of Americans – and an even higher portion of caregivers – are likely to research medical recommendations online after a doctor’s visit. But 1 in 4 people don’t know how to access their own medical records or find it difficult to do so.
The findings reflect the same low level of health literacy in the U.S. population that earlier surveys did. The results also indicate that little has changed since the Department of Health and Human Services released a National Action Plan to Improve Health Literacy in 2010.
That plan emphasized the need to develop and share accurate health information that helps people make decisions; to promote changes in the health care system that improve health information, communication, informed decision-making, and access to health services; and to increase the sharing and use of evidence-based health literacy practices.
According to the AHIMA Foundation report, 62% of Americans are not sure they understand their doctor’s advice and the health information discussed during a visit. Twenty-four percent say they don’t comprehend any of it, and 31% can’t remember what was said during the visit. Fifteen percent of those surveyed said they were more confused about their health than they were before the encounter with their doctor.
Caregivers have special issues
Forty-three percent of Americans are caregivers, the report notes, and 91% of those play an active role in managing someone else’s health. Millennials (65%) and Gen Xers (50%) are significantly more likely than Gen Zers (39%) and Boomers (20%) to be a caregiver.
Most caregivers have concerns about their loved ones’ ability to manage their own health. Most of them believe that doctors provide enough information, but 38% don’t believe a doctor can communicate effectively with the patient if the caregiver is not present.
Forty-three percent of caretakers don’t think their loved ones can understand medical information on their own. On the other hand, caregivers are more likely than people who don’t provide care to say the doctor confused them and to research the doctor’s advice after an appointment.
For many patients and caregivers, communications break down when they are with their health care provider. Twenty-two percent of Americans say they do not feel comfortable asking their doctor certain health questions. This inability to have a satisfactory dialogue with their doctor means that many patients leave their appointments without getting clear answers to their questions (24%) or without having an opportunity to ask any questions at all (17%).
This is not surprising, considering that a 2018 study found that doctors spend only 11 seconds, on average, listening to patients before interrupting them.
Depending on the internet
Overall, the AHIMA survey found, 42% of Americans research their doctor’s recommendations after an appointment. A higher percentage of caregivers than noncaregiver peers do so (47% vs. 38%). Eighty percent of respondents say they are “likely” to research their doctor’s advice online after a visit.
When they have a medical problem or a question about their condition, just as many Americans (59%) turn to the internet for an answer as contact their doctor directly, the survey found. Twenty-nine percent of the respondents consult friends, family, or colleagues; 23% look up medical records if they’re easily accessible; 19% ask pharmacists for advice; and 6% call an unspecified 800 number.
Americans feel secure in the health information they find on the internet. Among those who go online to look up information, 86% are confident that it is credible. And 42% report feeling relieved that they can find a lot of information about their health concerns. Respondents also say that the information they gather allows them to feel more confident in their doctor’s recommendations (35%) and that they feel better after having learned more on the internet than their doctor had told them (39%). Men are more likely than women to say that their confidence in their doctor’s recommendations increased after doing online research (40% vs. 30%).
Access to health records
Access to medical records would help people better understand their condition or diagnosis. But nearly half of Americans (48%) admit they don’t usually review their medical records until long after an appointment, and 52% say they rarely access their records at all.
One in four Americans say that they don’t know where to go to access their health information or that they didn’t find the process easy. More than half of those who have never had to find their records think the process would be difficult if they had to try.
Eighty-one percent of Americans use an online platform or portal to access their medical records or health information. Two-thirds of Americans who use an online portal trust that their medical information is kept safe and not shared with other people or organizations.
Four in five respondents agree that if they had access to all of their health information, including medical records, recommendations, conditions, and test results, they’d see an improvement in their health management. Fifty-nine percent of them believe they’d also be more confident about understanding their health, and 47% say they’d have greater trust in their doctor’s recommendations. Higher percentages of caregivers than noncaregivers say the same.
Younger people, those with a high school degree or less, and those who earn less than $50,000 are less likely than older, better educated, and more affluent people to understand their doctor’s health information and to ask questions of their providers.
People of color struggle with their relationships with doctors, are less satisfied than white people with the information they receive during visits, and are more likely than white peers to feel that if they had access to all their health information, they’d manage their health better and be more confident in their doctors’ recommendations, the survey found.
A version of this article first appeared on WebMD.com.
Confusion over health information and doctor advice is even higher among people who care for patients than among those who don’t provide care to their loved ones, the nationally representative survey from the AHIMA Foundation found.
The survey also shows that 80% of Americans – and an even higher portion of caregivers – are likely to research medical recommendations online after a doctor’s visit. But 1 in 4 people don’t know how to access their own medical records or find it difficult to do so.
The findings reflect the same low level of health literacy in the U.S. population that earlier surveys did. The results also indicate that little has changed since the Department of Health and Human Services released a National Action Plan to Improve Health Literacy in 2010.
That plan emphasized the need to develop and share accurate health information that helps people make decisions; to promote changes in the health care system that improve health information, communication, informed decision-making, and access to health services; and to increase the sharing and use of evidence-based health literacy practices.
According to the AHIMA Foundation report, 62% of Americans are not sure they understand their doctor’s advice and the health information discussed during a visit. Twenty-four percent say they don’t comprehend any of it, and 31% can’t remember what was said during the visit. Fifteen percent of those surveyed said they were more confused about their health than they were before the encounter with their doctor.
Caregivers have special issues
Forty-three percent of Americans are caregivers, the report notes, and 91% of those play an active role in managing someone else’s health. Millennials (65%) and Gen Xers (50%) are significantly more likely than Gen Zers (39%) and Boomers (20%) to be a caregiver.
Most caregivers have concerns about their loved ones’ ability to manage their own health. Most of them believe that doctors provide enough information, but 38% don’t believe a doctor can communicate effectively with the patient if the caregiver is not present.
Forty-three percent of caretakers don’t think their loved ones can understand medical information on their own. On the other hand, caregivers are more likely than people who don’t provide care to say the doctor confused them and to research the doctor’s advice after an appointment.
For many patients and caregivers, communications break down when they are with their health care provider. Twenty-two percent of Americans say they do not feel comfortable asking their doctor certain health questions. This inability to have a satisfactory dialogue with their doctor means that many patients leave their appointments without getting clear answers to their questions (24%) or without having an opportunity to ask any questions at all (17%).
This is not surprising, considering that a 2018 study found that doctors spend only 11 seconds, on average, listening to patients before interrupting them.
Depending on the internet
Overall, the AHIMA survey found, 42% of Americans research their doctor’s recommendations after an appointment. A higher percentage of caregivers than noncaregiver peers do so (47% vs. 38%). Eighty percent of respondents say they are “likely” to research their doctor’s advice online after a visit.
When they have a medical problem or a question about their condition, just as many Americans (59%) turn to the internet for an answer as contact their doctor directly, the survey found. Twenty-nine percent of the respondents consult friends, family, or colleagues; 23% look up medical records if they’re easily accessible; 19% ask pharmacists for advice; and 6% call an unspecified 800 number.
Americans feel secure in the health information they find on the internet. Among those who go online to look up information, 86% are confident that it is credible. And 42% report feeling relieved that they can find a lot of information about their health concerns. Respondents also say that the information they gather allows them to feel more confident in their doctor’s recommendations (35%) and that they feel better after having learned more on the internet than their doctor had told them (39%). Men are more likely than women to say that their confidence in their doctor’s recommendations increased after doing online research (40% vs. 30%).
Access to health records
Access to medical records would help people better understand their condition or diagnosis. But nearly half of Americans (48%) admit they don’t usually review their medical records until long after an appointment, and 52% say they rarely access their records at all.
One in four Americans say that they don’t know where to go to access their health information or that they didn’t find the process easy. More than half of those who have never had to find their records think the process would be difficult if they had to try.
Eighty-one percent of Americans use an online platform or portal to access their medical records or health information. Two-thirds of Americans who use an online portal trust that their medical information is kept safe and not shared with other people or organizations.
Four in five respondents agree that if they had access to all of their health information, including medical records, recommendations, conditions, and test results, they’d see an improvement in their health management. Fifty-nine percent of them believe they’d also be more confident about understanding their health, and 47% say they’d have greater trust in their doctor’s recommendations. Higher percentages of caregivers than noncaregivers say the same.
Younger people, those with a high school degree or less, and those who earn less than $50,000 are less likely than older, better educated, and more affluent people to understand their doctor’s health information and to ask questions of their providers.
People of color struggle with their relationships with doctors, are less satisfied than white people with the information they receive during visits, and are more likely than white peers to feel that if they had access to all their health information, they’d manage their health better and be more confident in their doctors’ recommendations, the survey found.
A version of this article first appeared on WebMD.com.
Confusion over health information and doctor advice is even higher among people who care for patients than among those who don’t provide care to their loved ones, the nationally representative survey from the AHIMA Foundation found.
The survey also shows that 80% of Americans – and an even higher portion of caregivers – are likely to research medical recommendations online after a doctor’s visit. But 1 in 4 people don’t know how to access their own medical records or find it difficult to do so.
The findings reflect the same low level of health literacy in the U.S. population that earlier surveys did. The results also indicate that little has changed since the Department of Health and Human Services released a National Action Plan to Improve Health Literacy in 2010.
That plan emphasized the need to develop and share accurate health information that helps people make decisions; to promote changes in the health care system that improve health information, communication, informed decision-making, and access to health services; and to increase the sharing and use of evidence-based health literacy practices.
According to the AHIMA Foundation report, 62% of Americans are not sure they understand their doctor’s advice and the health information discussed during a visit. Twenty-four percent say they don’t comprehend any of it, and 31% can’t remember what was said during the visit. Fifteen percent of those surveyed said they were more confused about their health than they were before the encounter with their doctor.
Caregivers have special issues
Forty-three percent of Americans are caregivers, the report notes, and 91% of those play an active role in managing someone else’s health. Millennials (65%) and Gen Xers (50%) are significantly more likely than Gen Zers (39%) and Boomers (20%) to be a caregiver.
Most caregivers have concerns about their loved ones’ ability to manage their own health. Most of them believe that doctors provide enough information, but 38% don’t believe a doctor can communicate effectively with the patient if the caregiver is not present.
Forty-three percent of caretakers don’t think their loved ones can understand medical information on their own. On the other hand, caregivers are more likely than people who don’t provide care to say the doctor confused them and to research the doctor’s advice after an appointment.
For many patients and caregivers, communications break down when they are with their health care provider. Twenty-two percent of Americans say they do not feel comfortable asking their doctor certain health questions. This inability to have a satisfactory dialogue with their doctor means that many patients leave their appointments without getting clear answers to their questions (24%) or without having an opportunity to ask any questions at all (17%).
This is not surprising, considering that a 2018 study found that doctors spend only 11 seconds, on average, listening to patients before interrupting them.
Depending on the internet
Overall, the AHIMA survey found, 42% of Americans research their doctor’s recommendations after an appointment. A higher percentage of caregivers than noncaregiver peers do so (47% vs. 38%). Eighty percent of respondents say they are “likely” to research their doctor’s advice online after a visit.
When they have a medical problem or a question about their condition, just as many Americans (59%) turn to the internet for an answer as contact their doctor directly, the survey found. Twenty-nine percent of the respondents consult friends, family, or colleagues; 23% look up medical records if they’re easily accessible; 19% ask pharmacists for advice; and 6% call an unspecified 800 number.
Americans feel secure in the health information they find on the internet. Among those who go online to look up information, 86% are confident that it is credible. And 42% report feeling relieved that they can find a lot of information about their health concerns. Respondents also say that the information they gather allows them to feel more confident in their doctor’s recommendations (35%) and that they feel better after having learned more on the internet than their doctor had told them (39%). Men are more likely than women to say that their confidence in their doctor’s recommendations increased after doing online research (40% vs. 30%).
Access to health records
Access to medical records would help people better understand their condition or diagnosis. But nearly half of Americans (48%) admit they don’t usually review their medical records until long after an appointment, and 52% say they rarely access their records at all.
One in four Americans say that they don’t know where to go to access their health information or that they didn’t find the process easy. More than half of those who have never had to find their records think the process would be difficult if they had to try.
Eighty-one percent of Americans use an online platform or portal to access their medical records or health information. Two-thirds of Americans who use an online portal trust that their medical information is kept safe and not shared with other people or organizations.
Four in five respondents agree that if they had access to all of their health information, including medical records, recommendations, conditions, and test results, they’d see an improvement in their health management. Fifty-nine percent of them believe they’d also be more confident about understanding their health, and 47% say they’d have greater trust in their doctor’s recommendations. Higher percentages of caregivers than noncaregivers say the same.
Younger people, those with a high school degree or less, and those who earn less than $50,000 are less likely than older, better educated, and more affluent people to understand their doctor’s health information and to ask questions of their providers.
People of color struggle with their relationships with doctors, are less satisfied than white people with the information they receive during visits, and are more likely than white peers to feel that if they had access to all their health information, they’d manage their health better and be more confident in their doctors’ recommendations, the survey found.
A version of this article first appeared on WebMD.com.
Now Takeda offers rebate if lung cancer drug fails to work
The rebate offer is for brigatinib (Alunbrig) which is approved for the treatment of adults with anaplastic lymphoma kinase positive (ALK+) metastatic non–small cell lung cancer (NSCLC) as detected by an FDA-approved test.
The move follows a rebate offer from Pfizer for crizotinib (Xalkori), which is also approved for ALK+ (as well as ROS1+) NSCLC, and also for ALK+ anaplastic large cell lymphoma
For its offer, Takeda has teamed up with Point32Health, the second-largest health plan in New England with about 2.3 million members. The new agreement will make brigatinib widely available to patients who may benefit from its use, say the companies.
If a patient is unable to remain on brigatinib for 3 months or longer because of effectiveness or tolerability, Takeda will refund a yet unspecified amount of money to Point32Health. Brigatinib’s list price is $17,000 for a month’s treatment.
“Given the importance of facilitating cutting-edge oncology treatment and also the reality that not all patients show a positive response, reimbursement for oncology treatments is an area that is prime for innovative financing approaches,” said Michael Sherman, MD, chief medical officer and executive vice president, Point32Health, in a statement. “Collaborating with Takeda to share risk makes this agreement a crucial milestone in bringing cost-effectiveness to cancer care.”
The Pfizer program for crizotinib is somewhat different. For one thing, Pfizer’s refund is offered to any patient who qualifies and not just those who are covered by a specific plan. Second, Takeda is thus far only refunding money to the insurer, whereas Pfizer will also reimburse patients for out-of-pocket expenses.
There is a similar approach that has been offered by Novartis for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
In addition, Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run health care system collected 200 million euros ($220 million) in refunds.
A version of this article first appeared on Medscape.com.
The rebate offer is for brigatinib (Alunbrig) which is approved for the treatment of adults with anaplastic lymphoma kinase positive (ALK+) metastatic non–small cell lung cancer (NSCLC) as detected by an FDA-approved test.
The move follows a rebate offer from Pfizer for crizotinib (Xalkori), which is also approved for ALK+ (as well as ROS1+) NSCLC, and also for ALK+ anaplastic large cell lymphoma
For its offer, Takeda has teamed up with Point32Health, the second-largest health plan in New England with about 2.3 million members. The new agreement will make brigatinib widely available to patients who may benefit from its use, say the companies.
If a patient is unable to remain on brigatinib for 3 months or longer because of effectiveness or tolerability, Takeda will refund a yet unspecified amount of money to Point32Health. Brigatinib’s list price is $17,000 for a month’s treatment.
“Given the importance of facilitating cutting-edge oncology treatment and also the reality that not all patients show a positive response, reimbursement for oncology treatments is an area that is prime for innovative financing approaches,” said Michael Sherman, MD, chief medical officer and executive vice president, Point32Health, in a statement. “Collaborating with Takeda to share risk makes this agreement a crucial milestone in bringing cost-effectiveness to cancer care.”
The Pfizer program for crizotinib is somewhat different. For one thing, Pfizer’s refund is offered to any patient who qualifies and not just those who are covered by a specific plan. Second, Takeda is thus far only refunding money to the insurer, whereas Pfizer will also reimburse patients for out-of-pocket expenses.
There is a similar approach that has been offered by Novartis for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
In addition, Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run health care system collected 200 million euros ($220 million) in refunds.
A version of this article first appeared on Medscape.com.
The rebate offer is for brigatinib (Alunbrig) which is approved for the treatment of adults with anaplastic lymphoma kinase positive (ALK+) metastatic non–small cell lung cancer (NSCLC) as detected by an FDA-approved test.
The move follows a rebate offer from Pfizer for crizotinib (Xalkori), which is also approved for ALK+ (as well as ROS1+) NSCLC, and also for ALK+ anaplastic large cell lymphoma
For its offer, Takeda has teamed up with Point32Health, the second-largest health plan in New England with about 2.3 million members. The new agreement will make brigatinib widely available to patients who may benefit from its use, say the companies.
If a patient is unable to remain on brigatinib for 3 months or longer because of effectiveness or tolerability, Takeda will refund a yet unspecified amount of money to Point32Health. Brigatinib’s list price is $17,000 for a month’s treatment.
“Given the importance of facilitating cutting-edge oncology treatment and also the reality that not all patients show a positive response, reimbursement for oncology treatments is an area that is prime for innovative financing approaches,” said Michael Sherman, MD, chief medical officer and executive vice president, Point32Health, in a statement. “Collaborating with Takeda to share risk makes this agreement a crucial milestone in bringing cost-effectiveness to cancer care.”
The Pfizer program for crizotinib is somewhat different. For one thing, Pfizer’s refund is offered to any patient who qualifies and not just those who are covered by a specific plan. Second, Takeda is thus far only refunding money to the insurer, whereas Pfizer will also reimburse patients for out-of-pocket expenses.
There is a similar approach that has been offered by Novartis for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
In addition, Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run health care system collected 200 million euros ($220 million) in refunds.
A version of this article first appeared on Medscape.com.
Which specialties get the biggest markups over Medicare rates?
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .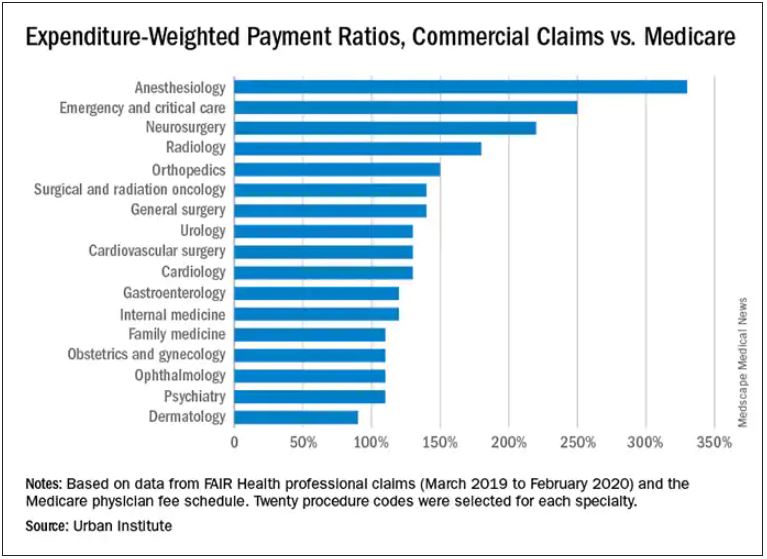
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
Anesthesiologists charge private insurers more than 300% above Medicare rates, a markup that is higher than that of 16 other specialties, according to a study released by the Urban Institute.
The Washington-based nonprofit institute found that the lowest markups were in psychiatry, ophthalmology, ob.gyn., family medicine, gastroenterology, and internal medicine, at 110%-120% of Medicare rates. .
In the middle are cardiology and cardiovascular surgery (130%), urology (130%), general surgery, surgical and radiation oncology (all at 140%), and orthopedics (150%).
At the top end were radiology (180%), neurosurgery (220%), emergency and critical care (250%), and anesthesiology (330%).
The wide variation in payments could be cited in support of the idea of applying Medicare rates across all physician specialties, say the study authors. Although lowering practitioner payments might lead to savings, it “will also create more pushback from providers, especially if these rates are introduced in the employer market,” write researchers Stacey McMorrow, PhD, Robert A. Berenson, MD, and John Holahan, PhD.
It is not known whether lowering commercial payment rates might decrease patient access, they write.
The authors also note that specialties in which the potential for a fee reduction was greatest were also the specialties for which baseline compensation was highest – from $350,000 annually for emergency physicians to $800,000 a year for neurosurgeons. Annual compensation for ob.gyns., dermatologists, and opthalmologists is about $350,000 a year, which suggests that “these specialties are similarly well compensated by both Medicare and commercial insurers,” the authors write.
The investigators assessed the top 20 procedure codes by expenditure in each of 17 physician specialties. They estimated the commercial-to-Medicare payment ratio for each service and constructed weighted averages across services for each specialty at the national level and for 12 states for which data for all the specialties and services were available.
The researchers analyzed claims from the FAIR Health database between March 2019 and March 2020. That database represents 60 insurers covering 150 million people.
Pediatric and geriatric specialties, nonphysician practitioners, out-of-network clinicians, and ambulatory surgery center claims were excluded. Codes with modifiers, J codes, and clinical laboratory services were also not included.
The charges used in the study were not the actual contracted rates. The authors instead used “imputed allowed amounts” for each claim line. That method was used to protect the confidentiality of the negotiated rates.
With regard to all specialties, the lowest compensated services were procedures, evaluation and management, and tests, which received 140%-150% of the Medicare rate. Treatments and imaging were marked up 160%. Anesthesia was reimbursed at a rate 330% higher than the rate Medicare would pay.
The authors also assessed geographic variation for the 12 states for which they had data.
Similar to findings in other studies, the researchers found that the markup was lowest in Pennsylvania (120%) and highest in Wisconsin (260%). The U.S. average was 160%. California and Missouri were at 150%; Michigan was right at the average.
For physicians in Illinois, Louisiana, Colorado, Texas, and New York, markups were 170%-180% over the Medicare rate. Markups for clinicians in New Jersey (190%) and Arizona (200%) were closest to the Wisconsin rate.
The authors note some study limitations, including the fact that they excluded out-of-network practitioners, “and such payments may disproportionately affect certain specialties.”
A version of this article first appeared on Medscape.com.
Pfizer offers refund if drug ‘doesn’t work’
The high cost of new cancer drugs has been the subject of many debates and discussions, but the issue remains largely unresolved.
Now, one pharmaceutical company is offering a refund if its drug “doesn’t work.”
For what it says is the first time in the industry,
“Through this pilot program, Pfizer will offer a warranty to patients and health plans -- Medicare Part D, commercial and those who pay cash -- who are prescribed Xalkori for an FDA [US Food and Drug Administration]–approved indication,” said a company spokesperson.
Although Pfizer claims that its pilot program is a first in the industry, there have been others that are similar.
In 2017, Novartis offered something similar for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run healthcare system collected €200 million ($220 million) in refunds.
Pfizer pledge
Crizotinib is a selective tyrosine kinase inhibitor used mainly in the treatment of metastatic non–small cell lung cancer for patients whose tumors are positive for ALK or ROS1, as detected by an FDA-approved test. This indication was approved a decade ago. Another indication, ALK-positive anaplastic large cell lymphoma, was added earlier this year.
Details of the Pfizer Pledge are posted on Pfizer’s website. Eligible patients are those for whom crizotinib is discontinued before the fourth 30-day supply is dispensed by the patient’s pharmacy.
“The warranty will reimburse an amount equal to the cost paid for the medicine,” the spokesperson added. “The insurance-backed warranty pilot program will be insured and managed by AIG.”
This program is only available for patients who reside in the United States.
If use of crizotinib is discontinued and documentation of ineffectiveness is provided, Pfizer will refund the out-of-pocket amount that was paid for up to the first three bottles (30-day supply) of crizotinib, up to a maximum of $19,144 for each month’s supply, or a total of $57,432. Pfizer will also refund the cost that was paid by Medicare or a commercial insurer.
“Also, we have made sure to develop a program that also allows for Medicare patients to be eligible, since they are exempt from copay cards and at risk for significant financial burden when starting an oncology treatment,” said the spokesperson.
The pilot program is available to patients who began taking crizotinib from June 1, 2021, through December 31, 2021.
So far, Pfizer is offering this warranty only for crizotinib, but that may change in the future.
“Once the pilot is complete, we will assess learnings and consider whether to build a more robust, scalable program capable of supporting multiple products,” the Pfizer spokesperson commented.
Previous scheme ended in court
Pfizer had previously tried a different approach to reducing drug costs: it had attempted to offer copay support programs to Medicare patients who were prescribed its cardiac drug tafamidis (Vyndaqe, Vyndamax).
Tafamidis, launched in 2019, is used for patients with transthyretin amyloid cardiomyopathy. For those patients, it has been shown to reduce all-cause mortality and cardiovascular hospitalizations. It costs about $225,000 a year and has been described as the most expensive cardiovascular drug in the United States.
Earlier this month, a court dismissed Pfizer’s challenge to an anti-kickback law that prevented the company from offering copay support programs to Medicare patients.
The judge ruled that Pfizer’s plan to offer direct payments to patients violated a federal ban on “knowingly or willfully” providing financial support to induce drug purchases, even in the absence of corrupt intent.
Pharmaceutical manufacturers are forbidden from subsidizing copayments for Medicare beneficiaries but are allowed to donate to independent nonprofit organizations that offer copay assistance. Pfizer sued the U.S. Department of Health and Human Services in June 2020 to get a court ruling that their proposed programs were legal.
The new pledge program for crizotinib operates from a different premise, the Pfizer spokesperson commented.
A version of this article first appeared on Medscape.com.
The high cost of new cancer drugs has been the subject of many debates and discussions, but the issue remains largely unresolved.
Now, one pharmaceutical company is offering a refund if its drug “doesn’t work.”
For what it says is the first time in the industry,
“Through this pilot program, Pfizer will offer a warranty to patients and health plans -- Medicare Part D, commercial and those who pay cash -- who are prescribed Xalkori for an FDA [US Food and Drug Administration]–approved indication,” said a company spokesperson.
Although Pfizer claims that its pilot program is a first in the industry, there have been others that are similar.
In 2017, Novartis offered something similar for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run healthcare system collected €200 million ($220 million) in refunds.
Pfizer pledge
Crizotinib is a selective tyrosine kinase inhibitor used mainly in the treatment of metastatic non–small cell lung cancer for patients whose tumors are positive for ALK or ROS1, as detected by an FDA-approved test. This indication was approved a decade ago. Another indication, ALK-positive anaplastic large cell lymphoma, was added earlier this year.
Details of the Pfizer Pledge are posted on Pfizer’s website. Eligible patients are those for whom crizotinib is discontinued before the fourth 30-day supply is dispensed by the patient’s pharmacy.
“The warranty will reimburse an amount equal to the cost paid for the medicine,” the spokesperson added. “The insurance-backed warranty pilot program will be insured and managed by AIG.”
This program is only available for patients who reside in the United States.
If use of crizotinib is discontinued and documentation of ineffectiveness is provided, Pfizer will refund the out-of-pocket amount that was paid for up to the first three bottles (30-day supply) of crizotinib, up to a maximum of $19,144 for each month’s supply, or a total of $57,432. Pfizer will also refund the cost that was paid by Medicare or a commercial insurer.
“Also, we have made sure to develop a program that also allows for Medicare patients to be eligible, since they are exempt from copay cards and at risk for significant financial burden when starting an oncology treatment,” said the spokesperson.
The pilot program is available to patients who began taking crizotinib from June 1, 2021, through December 31, 2021.
So far, Pfizer is offering this warranty only for crizotinib, but that may change in the future.
“Once the pilot is complete, we will assess learnings and consider whether to build a more robust, scalable program capable of supporting multiple products,” the Pfizer spokesperson commented.
Previous scheme ended in court
Pfizer had previously tried a different approach to reducing drug costs: it had attempted to offer copay support programs to Medicare patients who were prescribed its cardiac drug tafamidis (Vyndaqe, Vyndamax).
Tafamidis, launched in 2019, is used for patients with transthyretin amyloid cardiomyopathy. For those patients, it has been shown to reduce all-cause mortality and cardiovascular hospitalizations. It costs about $225,000 a year and has been described as the most expensive cardiovascular drug in the United States.
Earlier this month, a court dismissed Pfizer’s challenge to an anti-kickback law that prevented the company from offering copay support programs to Medicare patients.
The judge ruled that Pfizer’s plan to offer direct payments to patients violated a federal ban on “knowingly or willfully” providing financial support to induce drug purchases, even in the absence of corrupt intent.
Pharmaceutical manufacturers are forbidden from subsidizing copayments for Medicare beneficiaries but are allowed to donate to independent nonprofit organizations that offer copay assistance. Pfizer sued the U.S. Department of Health and Human Services in June 2020 to get a court ruling that their proposed programs were legal.
The new pledge program for crizotinib operates from a different premise, the Pfizer spokesperson commented.
A version of this article first appeared on Medscape.com.
The high cost of new cancer drugs has been the subject of many debates and discussions, but the issue remains largely unresolved.
Now, one pharmaceutical company is offering a refund if its drug “doesn’t work.”
For what it says is the first time in the industry,
“Through this pilot program, Pfizer will offer a warranty to patients and health plans -- Medicare Part D, commercial and those who pay cash -- who are prescribed Xalkori for an FDA [US Food and Drug Administration]–approved indication,” said a company spokesperson.
Although Pfizer claims that its pilot program is a first in the industry, there have been others that are similar.
In 2017, Novartis offered something similar for tisagenlecleucel (Kymriah), the CAR T-cell therapy that launched with a daunting price tag of $475,000. After receiving backlash over the cost, Novartis announced that if the drug does not work after the first month, patients pay nothing.
Italy has been using this system for several years. Pharmaceutical companies must refund money if the drug fails to work. In 2015, the state-run healthcare system collected €200 million ($220 million) in refunds.
Pfizer pledge
Crizotinib is a selective tyrosine kinase inhibitor used mainly in the treatment of metastatic non–small cell lung cancer for patients whose tumors are positive for ALK or ROS1, as detected by an FDA-approved test. This indication was approved a decade ago. Another indication, ALK-positive anaplastic large cell lymphoma, was added earlier this year.
Details of the Pfizer Pledge are posted on Pfizer’s website. Eligible patients are those for whom crizotinib is discontinued before the fourth 30-day supply is dispensed by the patient’s pharmacy.
“The warranty will reimburse an amount equal to the cost paid for the medicine,” the spokesperson added. “The insurance-backed warranty pilot program will be insured and managed by AIG.”
This program is only available for patients who reside in the United States.
If use of crizotinib is discontinued and documentation of ineffectiveness is provided, Pfizer will refund the out-of-pocket amount that was paid for up to the first three bottles (30-day supply) of crizotinib, up to a maximum of $19,144 for each month’s supply, or a total of $57,432. Pfizer will also refund the cost that was paid by Medicare or a commercial insurer.
“Also, we have made sure to develop a program that also allows for Medicare patients to be eligible, since they are exempt from copay cards and at risk for significant financial burden when starting an oncology treatment,” said the spokesperson.
The pilot program is available to patients who began taking crizotinib from June 1, 2021, through December 31, 2021.
So far, Pfizer is offering this warranty only for crizotinib, but that may change in the future.
“Once the pilot is complete, we will assess learnings and consider whether to build a more robust, scalable program capable of supporting multiple products,” the Pfizer spokesperson commented.
Previous scheme ended in court
Pfizer had previously tried a different approach to reducing drug costs: it had attempted to offer copay support programs to Medicare patients who were prescribed its cardiac drug tafamidis (Vyndaqe, Vyndamax).
Tafamidis, launched in 2019, is used for patients with transthyretin amyloid cardiomyopathy. For those patients, it has been shown to reduce all-cause mortality and cardiovascular hospitalizations. It costs about $225,000 a year and has been described as the most expensive cardiovascular drug in the United States.
Earlier this month, a court dismissed Pfizer’s challenge to an anti-kickback law that prevented the company from offering copay support programs to Medicare patients.
The judge ruled that Pfizer’s plan to offer direct payments to patients violated a federal ban on “knowingly or willfully” providing financial support to induce drug purchases, even in the absence of corrupt intent.
Pharmaceutical manufacturers are forbidden from subsidizing copayments for Medicare beneficiaries but are allowed to donate to independent nonprofit organizations that offer copay assistance. Pfizer sued the U.S. Department of Health and Human Services in June 2020 to get a court ruling that their proposed programs were legal.
The new pledge program for crizotinib operates from a different premise, the Pfizer spokesperson commented.
A version of this article first appeared on Medscape.com.
Patient loses prostate after biopsy slide switched; more
It’s difficult enough when a patient’s prostate is removed because of cancer. But it’s another thing altogether when the prostate is removed because of a medical error, as a report on 3 CBS Philly, among other news outlets, makes clear.
The patient, Eric Spangs, lives in southeastern Pennsylvania. Testing indicated an elevation in prostate-specific antigen (PSA) level. He subsequently underwent biopsy of the prostate, which appeared to indicate cancer. In time, though, Mr. Spangs learned there had been an error: the tissue section used in the microscopic diagnosis had come from the biopsy specimen of a different patient. Mr. Spangs himself didn’t actually have cancer.
Ordinarily, such news would be cause for celebration. But this was far from a normal situ ation: Following his initial cancer diagnosis, Mr. Spangs underwent a radical laparoscopic prostatectomy at a local hospital.
“It’s devastated me emotionally and physically,” Mr. Spangs said. It has also been emotionally devastating for his wife, Melissa. (The couple has five children.)
Their attorney, Aaron Freiwald, has filed a suit against the health system to which the local hospital belongs and the area’s largest urologic practice.
The Spangs wish to caution other patients not to make the same mistake they did: they failed to get a second opinion from an oncology specialist, as recommended by the American Cancer Society. (Eric Spangs did receive a second opinion from someone at the urologic practice, but that practice doesn’t specialize in oncology.)
The Spangs also worry about the patient who received the false-negative biopsy result. They have been assured, however, that that patient will be properly notified of his actual cancer status.
Fertility specialist uses own sperm to impregnate patients
A suit claims that a Rochester, N.Y., gynecologist and fertility specialist used his own sperm to inseminate multiple patients, according to a story reported by the Associated Press and other news outlets.
The suit was filed last month by the daughter — call her “Harriet Jones” — of one of the women who received fertility services from the doctor during the 1980s. Ms. Jones’s suit alleges that at the time, the doctor told her mother that the sperm donor would be a medical student at the University of Rochester. In fact, the donor was the doctor himself. He kept that fact a secret for years, even after Ms. Jones — his own daughter — sought him out for gynecologic services.
The secret gradually began to come to light in 2016, when Ms. Jones’s nonbiological father — the man who had helped to raise her — died. Curious about her biological father, Ms. Jones sought to learn his identity from the Rochester gynecologist who had treated her mother and was now her own gynecologist. The doctor said he couldn’t be of help; he claimed he hadn’t kept the relevant records.
Ms. Jones then submitted a blood sample to a direct-to-consumer genetic testing company. The results surprised her: Not only did she learn of her ethnicity, but she also discovered the existence of two half siblings, who were donor-conceived in 1984 and 1985, respectively, the very period when her own mother was undergoing insemination procedures. Ms. Jones subsequently discovered the existence of additional half siblings, all born in the first half of the 1980s.
Initially elated by the discoveries, Ms. Jones soon grew despondent and anxious. She suffered from migraine headaches, among other symptoms. Her biological father, it seemed, had been “a serial sperm donor.”
Still, she continued to go to her Rochester doctor for treatment, having no reason to suspect anything untoward about him. Her visits, including those for prolonged menstrual bleeding, involved routine breast and pelvic exams, transvaginal ultrasounds, and intrauterine contraceptive placements under sedation.
Over this period, her doctor was friendly, asking her a variety of questions about her personal life. During one especially strange visit, however, he began chuckling and said, “You’re a really good kid, such a good kid.” During this visit, he invited his wife into the exam room, presumably to meet Ms. Jones.
It was at this moment that Ms. Jones had a revelation: Could her gynecologist actually be her biological father?
In May 2021, Ms. Jones and a half brother with whom she had been in touch contacted the gynecologist’s daughter from his first marriage. All three underwent genetic testing. The results showed a 99.99% chance of a genetic link.
Ms. Jones has said in her suit that “no reasonable woman” would have submitted to pelvic examinations and other examinations by a doctor whom she knew to be her father.
Besides fraud, her suit alleges medical malpractice, battery, infliction of mental distress, and lack of informed consent. She is seeking compensation for all harm caused to her, including past and future economic damages, past unreimbursed medical expenses, and future expenses related to her mental health treatment and care.
The story included no further details about the civil litigation. As for criminal charges, it’s unlikely Ms. Jones’s biological father — her gynecologist — will face criminal charges for his alleged crimes because they fall outside of the state’s statute of limitations.
Parents say daughter’s stroke wasn’t identified
The Georgia parents of a young woman who died from a stroke following a series of alleged misdiagnoses are suing multiple practitioners, reports Legal Newswire and other news outlets
In June 2019, Michaela Smith was training for her job as a detention officer when she began experiencing a variety of symptoms, including headache, shortness of breath, throat swelling, and slurred speech. She was taken to the emergency department (ED) at Hamilton Medical Center, in Dalton, Ga.
There, she was examined by an attending ED doctor, who ordered a CT scan. The results were read by radiologist Michael J. Cooney, MD. In his reading, the Smith family’s lawsuit alleges, Dr. Cooney failed to identify the basilar artery sign, which is a key indicator of a vessel occlusion in stroke patients. Dr. Cooney concluded that Ms. Smith’s scan showed no acute intracranial abnormality. He sent her home without further discharge instructions.
At home, Ms. Smith fell asleep but awoke in an altered mental state, one of several classic stroke symptoms that she had been experiencing. She returned to the ED. This time, she was examined by David F. Hawkins, MD, an ED physician. Although his differential diagnosis identified Ms. Smith’s symptoms as most likely stroke related, Dr. Hawkins allegedly failed to immediately corroborate his findings with additional vascular imaging. Later in the day, Ms. Smith did undergo an MRI, which a second radiologist, Kevin F. Johnson, MD, misread as showing no signs of ischemia in her basilar artery, according to the lawsuit.
That same day, Dr. Hawkins conferred with a second neurologist, Jeffrey T. Glass, MD, who recommended that Ms. Smith be admitted to the hospital because of her deteriorating condition. The Smiths’ suit claims that Dr. Glass also failed to diagnosis their daughter’s underlying condition, although he did sign off on her transfer to Baroness Erlanger Hospital, in Chattanooga, Tenn.
There, Ms. Smith’s condition continued to worsen. She soon required mechanical ventilation and tube feeding. On July 3, 2019, she was pronounced dead.
“This is an egregious case of negligence,” said the attorney representing the Smiths, who are suing the physicians involved and their practices, as well as Hamilton Medical Center and several unnamed defendants.
“Although two radiology studies and her clinical presentation indicated that Michaela was having a catastrophic stroke, her doctors repeatedly misread the studies as normal, failed to diagnose the stroke, and failed to treat her deficits as a neurological emergency,” the family’s lawyer stated.
At press time, there had been no response from any of the defendants or their attorneys.
A version of this article first appeared on Medscape.com.
It’s difficult enough when a patient’s prostate is removed because of cancer. But it’s another thing altogether when the prostate is removed because of a medical error, as a report on 3 CBS Philly, among other news outlets, makes clear.
The patient, Eric Spangs, lives in southeastern Pennsylvania. Testing indicated an elevation in prostate-specific antigen (PSA) level. He subsequently underwent biopsy of the prostate, which appeared to indicate cancer. In time, though, Mr. Spangs learned there had been an error: the tissue section used in the microscopic diagnosis had come from the biopsy specimen of a different patient. Mr. Spangs himself didn’t actually have cancer.
Ordinarily, such news would be cause for celebration. But this was far from a normal situ ation: Following his initial cancer diagnosis, Mr. Spangs underwent a radical laparoscopic prostatectomy at a local hospital.
“It’s devastated me emotionally and physically,” Mr. Spangs said. It has also been emotionally devastating for his wife, Melissa. (The couple has five children.)
Their attorney, Aaron Freiwald, has filed a suit against the health system to which the local hospital belongs and the area’s largest urologic practice.
The Spangs wish to caution other patients not to make the same mistake they did: they failed to get a second opinion from an oncology specialist, as recommended by the American Cancer Society. (Eric Spangs did receive a second opinion from someone at the urologic practice, but that practice doesn’t specialize in oncology.)
The Spangs also worry about the patient who received the false-negative biopsy result. They have been assured, however, that that patient will be properly notified of his actual cancer status.
Fertility specialist uses own sperm to impregnate patients
A suit claims that a Rochester, N.Y., gynecologist and fertility specialist used his own sperm to inseminate multiple patients, according to a story reported by the Associated Press and other news outlets.
The suit was filed last month by the daughter — call her “Harriet Jones” — of one of the women who received fertility services from the doctor during the 1980s. Ms. Jones’s suit alleges that at the time, the doctor told her mother that the sperm donor would be a medical student at the University of Rochester. In fact, the donor was the doctor himself. He kept that fact a secret for years, even after Ms. Jones — his own daughter — sought him out for gynecologic services.
The secret gradually began to come to light in 2016, when Ms. Jones’s nonbiological father — the man who had helped to raise her — died. Curious about her biological father, Ms. Jones sought to learn his identity from the Rochester gynecologist who had treated her mother and was now her own gynecologist. The doctor said he couldn’t be of help; he claimed he hadn’t kept the relevant records.
Ms. Jones then submitted a blood sample to a direct-to-consumer genetic testing company. The results surprised her: Not only did she learn of her ethnicity, but she also discovered the existence of two half siblings, who were donor-conceived in 1984 and 1985, respectively, the very period when her own mother was undergoing insemination procedures. Ms. Jones subsequently discovered the existence of additional half siblings, all born in the first half of the 1980s.
Initially elated by the discoveries, Ms. Jones soon grew despondent and anxious. She suffered from migraine headaches, among other symptoms. Her biological father, it seemed, had been “a serial sperm donor.”
Still, she continued to go to her Rochester doctor for treatment, having no reason to suspect anything untoward about him. Her visits, including those for prolonged menstrual bleeding, involved routine breast and pelvic exams, transvaginal ultrasounds, and intrauterine contraceptive placements under sedation.
Over this period, her doctor was friendly, asking her a variety of questions about her personal life. During one especially strange visit, however, he began chuckling and said, “You’re a really good kid, such a good kid.” During this visit, he invited his wife into the exam room, presumably to meet Ms. Jones.
It was at this moment that Ms. Jones had a revelation: Could her gynecologist actually be her biological father?
In May 2021, Ms. Jones and a half brother with whom she had been in touch contacted the gynecologist’s daughter from his first marriage. All three underwent genetic testing. The results showed a 99.99% chance of a genetic link.
Ms. Jones has said in her suit that “no reasonable woman” would have submitted to pelvic examinations and other examinations by a doctor whom she knew to be her father.
Besides fraud, her suit alleges medical malpractice, battery, infliction of mental distress, and lack of informed consent. She is seeking compensation for all harm caused to her, including past and future economic damages, past unreimbursed medical expenses, and future expenses related to her mental health treatment and care.
The story included no further details about the civil litigation. As for criminal charges, it’s unlikely Ms. Jones’s biological father — her gynecologist — will face criminal charges for his alleged crimes because they fall outside of the state’s statute of limitations.
Parents say daughter’s stroke wasn’t identified
The Georgia parents of a young woman who died from a stroke following a series of alleged misdiagnoses are suing multiple practitioners, reports Legal Newswire and other news outlets
In June 2019, Michaela Smith was training for her job as a detention officer when she began experiencing a variety of symptoms, including headache, shortness of breath, throat swelling, and slurred speech. She was taken to the emergency department (ED) at Hamilton Medical Center, in Dalton, Ga.
There, she was examined by an attending ED doctor, who ordered a CT scan. The results were read by radiologist Michael J. Cooney, MD. In his reading, the Smith family’s lawsuit alleges, Dr. Cooney failed to identify the basilar artery sign, which is a key indicator of a vessel occlusion in stroke patients. Dr. Cooney concluded that Ms. Smith’s scan showed no acute intracranial abnormality. He sent her home without further discharge instructions.
At home, Ms. Smith fell asleep but awoke in an altered mental state, one of several classic stroke symptoms that she had been experiencing. She returned to the ED. This time, she was examined by David F. Hawkins, MD, an ED physician. Although his differential diagnosis identified Ms. Smith’s symptoms as most likely stroke related, Dr. Hawkins allegedly failed to immediately corroborate his findings with additional vascular imaging. Later in the day, Ms. Smith did undergo an MRI, which a second radiologist, Kevin F. Johnson, MD, misread as showing no signs of ischemia in her basilar artery, according to the lawsuit.
That same day, Dr. Hawkins conferred with a second neurologist, Jeffrey T. Glass, MD, who recommended that Ms. Smith be admitted to the hospital because of her deteriorating condition. The Smiths’ suit claims that Dr. Glass also failed to diagnosis their daughter’s underlying condition, although he did sign off on her transfer to Baroness Erlanger Hospital, in Chattanooga, Tenn.
There, Ms. Smith’s condition continued to worsen. She soon required mechanical ventilation and tube feeding. On July 3, 2019, she was pronounced dead.
“This is an egregious case of negligence,” said the attorney representing the Smiths, who are suing the physicians involved and their practices, as well as Hamilton Medical Center and several unnamed defendants.
“Although two radiology studies and her clinical presentation indicated that Michaela was having a catastrophic stroke, her doctors repeatedly misread the studies as normal, failed to diagnose the stroke, and failed to treat her deficits as a neurological emergency,” the family’s lawyer stated.
At press time, there had been no response from any of the defendants or their attorneys.
A version of this article first appeared on Medscape.com.
It’s difficult enough when a patient’s prostate is removed because of cancer. But it’s another thing altogether when the prostate is removed because of a medical error, as a report on 3 CBS Philly, among other news outlets, makes clear.
The patient, Eric Spangs, lives in southeastern Pennsylvania. Testing indicated an elevation in prostate-specific antigen (PSA) level. He subsequently underwent biopsy of the prostate, which appeared to indicate cancer. In time, though, Mr. Spangs learned there had been an error: the tissue section used in the microscopic diagnosis had come from the biopsy specimen of a different patient. Mr. Spangs himself didn’t actually have cancer.
Ordinarily, such news would be cause for celebration. But this was far from a normal situ ation: Following his initial cancer diagnosis, Mr. Spangs underwent a radical laparoscopic prostatectomy at a local hospital.
“It’s devastated me emotionally and physically,” Mr. Spangs said. It has also been emotionally devastating for his wife, Melissa. (The couple has five children.)
Their attorney, Aaron Freiwald, has filed a suit against the health system to which the local hospital belongs and the area’s largest urologic practice.
The Spangs wish to caution other patients not to make the same mistake they did: they failed to get a second opinion from an oncology specialist, as recommended by the American Cancer Society. (Eric Spangs did receive a second opinion from someone at the urologic practice, but that practice doesn’t specialize in oncology.)
The Spangs also worry about the patient who received the false-negative biopsy result. They have been assured, however, that that patient will be properly notified of his actual cancer status.
Fertility specialist uses own sperm to impregnate patients
A suit claims that a Rochester, N.Y., gynecologist and fertility specialist used his own sperm to inseminate multiple patients, according to a story reported by the Associated Press and other news outlets.
The suit was filed last month by the daughter — call her “Harriet Jones” — of one of the women who received fertility services from the doctor during the 1980s. Ms. Jones’s suit alleges that at the time, the doctor told her mother that the sperm donor would be a medical student at the University of Rochester. In fact, the donor was the doctor himself. He kept that fact a secret for years, even after Ms. Jones — his own daughter — sought him out for gynecologic services.
The secret gradually began to come to light in 2016, when Ms. Jones’s nonbiological father — the man who had helped to raise her — died. Curious about her biological father, Ms. Jones sought to learn his identity from the Rochester gynecologist who had treated her mother and was now her own gynecologist. The doctor said he couldn’t be of help; he claimed he hadn’t kept the relevant records.
Ms. Jones then submitted a blood sample to a direct-to-consumer genetic testing company. The results surprised her: Not only did she learn of her ethnicity, but she also discovered the existence of two half siblings, who were donor-conceived in 1984 and 1985, respectively, the very period when her own mother was undergoing insemination procedures. Ms. Jones subsequently discovered the existence of additional half siblings, all born in the first half of the 1980s.
Initially elated by the discoveries, Ms. Jones soon grew despondent and anxious. She suffered from migraine headaches, among other symptoms. Her biological father, it seemed, had been “a serial sperm donor.”
Still, she continued to go to her Rochester doctor for treatment, having no reason to suspect anything untoward about him. Her visits, including those for prolonged menstrual bleeding, involved routine breast and pelvic exams, transvaginal ultrasounds, and intrauterine contraceptive placements under sedation.
Over this period, her doctor was friendly, asking her a variety of questions about her personal life. During one especially strange visit, however, he began chuckling and said, “You’re a really good kid, such a good kid.” During this visit, he invited his wife into the exam room, presumably to meet Ms. Jones.
It was at this moment that Ms. Jones had a revelation: Could her gynecologist actually be her biological father?
In May 2021, Ms. Jones and a half brother with whom she had been in touch contacted the gynecologist’s daughter from his first marriage. All three underwent genetic testing. The results showed a 99.99% chance of a genetic link.
Ms. Jones has said in her suit that “no reasonable woman” would have submitted to pelvic examinations and other examinations by a doctor whom she knew to be her father.
Besides fraud, her suit alleges medical malpractice, battery, infliction of mental distress, and lack of informed consent. She is seeking compensation for all harm caused to her, including past and future economic damages, past unreimbursed medical expenses, and future expenses related to her mental health treatment and care.
The story included no further details about the civil litigation. As for criminal charges, it’s unlikely Ms. Jones’s biological father — her gynecologist — will face criminal charges for his alleged crimes because they fall outside of the state’s statute of limitations.
Parents say daughter’s stroke wasn’t identified
The Georgia parents of a young woman who died from a stroke following a series of alleged misdiagnoses are suing multiple practitioners, reports Legal Newswire and other news outlets
In June 2019, Michaela Smith was training for her job as a detention officer when she began experiencing a variety of symptoms, including headache, shortness of breath, throat swelling, and slurred speech. She was taken to the emergency department (ED) at Hamilton Medical Center, in Dalton, Ga.
There, she was examined by an attending ED doctor, who ordered a CT scan. The results were read by radiologist Michael J. Cooney, MD. In his reading, the Smith family’s lawsuit alleges, Dr. Cooney failed to identify the basilar artery sign, which is a key indicator of a vessel occlusion in stroke patients. Dr. Cooney concluded that Ms. Smith’s scan showed no acute intracranial abnormality. He sent her home without further discharge instructions.
At home, Ms. Smith fell asleep but awoke in an altered mental state, one of several classic stroke symptoms that she had been experiencing. She returned to the ED. This time, she was examined by David F. Hawkins, MD, an ED physician. Although his differential diagnosis identified Ms. Smith’s symptoms as most likely stroke related, Dr. Hawkins allegedly failed to immediately corroborate his findings with additional vascular imaging. Later in the day, Ms. Smith did undergo an MRI, which a second radiologist, Kevin F. Johnson, MD, misread as showing no signs of ischemia in her basilar artery, according to the lawsuit.
That same day, Dr. Hawkins conferred with a second neurologist, Jeffrey T. Glass, MD, who recommended that Ms. Smith be admitted to the hospital because of her deteriorating condition. The Smiths’ suit claims that Dr. Glass also failed to diagnosis their daughter’s underlying condition, although he did sign off on her transfer to Baroness Erlanger Hospital, in Chattanooga, Tenn.
There, Ms. Smith’s condition continued to worsen. She soon required mechanical ventilation and tube feeding. On July 3, 2019, she was pronounced dead.
“This is an egregious case of negligence,” said the attorney representing the Smiths, who are suing the physicians involved and their practices, as well as Hamilton Medical Center and several unnamed defendants.
“Although two radiology studies and her clinical presentation indicated that Michaela was having a catastrophic stroke, her doctors repeatedly misread the studies as normal, failed to diagnose the stroke, and failed to treat her deficits as a neurological emergency,” the family’s lawyer stated.
At press time, there had been no response from any of the defendants or their attorneys.
A version of this article first appeared on Medscape.com.