User login
COVID-19: We are in a war, without the most effective weapons to fight a novel viral pathogen
On June 17, 1775, American colonists, defending a forward redoubt on Breed’s Hill, ran out of gunpowder, and their position was overrun by British troops. The Battle of Bunker Hill resulted in the death of 140 colonists and 226 British soldiers, setting the stage for major combat throughout the colonies. American colonists lacked many necessary weapons. They had almost no gunpowder, few field cannons, and no warships. Yet, they fought on with the weapons at hand for 6 long years.
In the spring of 2020, American society has been shaken by the COVID-19 pandemic. Hospitals have been overrun with thousands of people infected with the disease. Some hospitals are breaking under the crush of intensely ill people filling up and spilling out of intensive care units. We are in a war, fighting a viral disease with a limited supply of weapons. We do not have access to the most powerful medical munitions: easily available rapid testing, proven antiviral medications, and an effective vaccine. Nevertheless, clinicians and patients are courageous, and we will continue the fight with the limited weapons we have until the pandemic is brought to an end.

The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19). The virus is aptly named because it is usually transmitted through close contact with respiratory droplets. The disease can progress acutely, and some people experience a remarkably severe respiratory syndrome, including tachypnea, hypoxia, and interstitial and alveolar opacities on chest x-ray, necessitating ventilatory support. The virus is an encapsulated single-stranded RNA virus. When viewed by electron microscopy, the virus appears to have a halo or crown, hence it is named “coronavirus.” Among infected individuals, the virus is present in the upper respiratory system and in feces but not in urine.1 The World Health Organization (WHO) believes that respiratory droplets and contaminated surfaces are the major routes of transmission.2 The highest risk of developing severe COVID-19 disease occurs in people with one or more of the following characteristics: age greater than 70 years, hypertension, diabetes, respiratory disease, heart disease, and immunosuppression.3,4 Pregnant women do not appear to be at increased risk for severe COVID-19 disease.4 The case fatality rate is highest in people 80 years of age or older.5
Who is infected with SARS-CoV-2?
Rapid high-fidelity testing for SARS-CoV-2 nucleic acid sequences would be the best approach to identifying people with COVID-19 disease. At the beginning of the pandemic, testing was strictly rationed because of lack of reagents and test swabs. Clinicians were permitted to test only a minority of people who had symptoms. Asymptomatic individuals were not eligible to be tested. This terribly flawed approach to screening permitted a vast army of SARS-CoV-2–positive asymptomatic and mildly symptomatic people to circulate unchecked in the general population, infecting dozens of other people, some of whom developed moderate or severe disease. The Centers for Disease Control and Prevention (CDC) has reported on 7 independent clusters of COVID-19 disease, each of which appear to have been caused by one asymptomatic infected individual.6 Another cluster of COVID-19 disease from China appears to have been caused by one asymptomatic infected individual.7 Based on limited data, it appears that there may be a 1- to 3-day window where an individual with COVID-19 may be asymptomatic and able to infect others. I suspect that we will soon discover, based on testing for the presence of high-titre anti SARS-CoV-2 antibodies, that many people with no history of illness and people with mild respiratory symptoms had an undiagnosed COVID-19 infection.
As testing capacity expands we likely will be testing all women, including asymptomatic women, before they arrive at the hospital for childbirth or gynecologic surgery, as well as all inpatients and women with respiratory symptoms having an ambulatory encounter.
With expanded testing capability, some pregnant women who were symptomatic and tested positive for SARS-CoV-2 have had sequential long-term follow-up testing. A frequent observation is that over one to two weeks the viral symptoms resolve and the nasopharyngeal test becomes negative for SARS-CoV-2 on multiple sequential tests, only to become positive at a later date. The cause of the positive-negative-negative-positive test results is unknown, but it raises the possibility that once a person tests positive for SARS-CoV-2, they may be able to transmit the infection over many weeks, even after viral symptoms resolve.
Continue to: COVID-19: Respiratory droplet or aerosol transmission?
COVID-19: Respiratory droplet or aerosol transmission?
Respiratory droplets are large particles (> 5 µm in diameter) that tend to be pulled to the ground or furniture surfaces by gravity. Respiratory droplets do not circulate in the air for an extended period of time. Droplet nuclei are small particles less than 5 µm in diameter. These small particles may become aerosolized and float through the air for an extended period of time. The CDC and WHO believe that under ordinary conditions, SARS-CoV-2 is transmitted through respiratory droplets and contact routes.2 In an analysis of more than 75,000 COVID-19 cases in China there were no reports of transmission by aerosolized airborne virus. Therefore, under ordinary conditions, surgical masks, face shields, gowns, and gloves provide a high level of protection from infection.8
In contrast to the WHO’s perspective, Dr. Harvey Fineberg, Chair of the National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, wrote a letter to the federal Office of Science and Technology Policy warning that normal breathing might generate aerosolization of the SARS-CoV-2 virus and result in airborne transmission.9 A report from the University of Nebraska Medical Center supports the concept of airborne transmission of SARS-CoV-2. In a study of 13 patients with COVID-19, room surfaces, toilet facilities, and air had evidence of viral contamination.10 The investigators concluded that disease spreads through respiratory droplets, person-to-person touch, contaminated surfaces, and airborne routes. Other investigators also have reported that aersolization of SARS-CoV-2 may occur.11 Professional societies recommend that all medical staff caring for potential or confirmed COVID-19 patients should use personal protective equipment (PPE), including respirators (N95 respirators) when available. Importantly, all medical staff should be trained in and adhere to proper donning and doffing of PPE. The controversy about the modes of transmission of SARS-CoV-2 will continue, but as clinicians we need to work within the constraints of the equipment we have.
Certain medical procedures and devices are known to generate aerosolization of respiratory secretions. These procedures and devices include: bronchoscopy, intubation, extubation, cardiopulmonary resuscitation, nebulization, high-flow oxygen masks, and continuous- and bilevel-positive airway pressure devices. When aerosols are generated during the care of a patient with COVID-19, surgical masks are not sufficient protection against infection. When an aerosol is generated maximal protection of health care workers from viral transmission requires use of a negative-pressure room and an N95 respirator or powered air-purifying respirator (PAPR) device. However, negative-pressure rooms, N95 masks, and PAPRs are in very short supply or are unavailable in some health systems. We are lucky at our hospital that all of the labor rooms can be configured to operate in a negative-pressure mode, limiting potential airborne spread of the virus on the unit. Many hospitals restrict the use of N95 masks to anesthesiologists, leaving nurses, ObGyns, and surgical technicians without the best protective equipment, risking their health. As one action to reduce aerosolization of virus, obstetricians can markedly reduce the use of oxygen masks and nasal cannulas by laboring women.
Universal use of surgical masks and mouth-nose coverings
During the entire COVID-19 pandemic, PPE has been in short supply, including severe shortages of N95 masks, PAPRs, and in some health systems, surgical masks, gowns, eye protection, and face shields. Given the severe shortages, some clinicians have needed to conserve PPE, using the same PPE across multiple patient encounters and across multiple work shifts.
Given that the virus is transmitted by respiratory droplets and contaminated surfaces, use of face coverings, including surgical masks, face shields, and gloves is critically important. Scrupulous hand hygiene is a simple approach to reducing infection risk. In my health system, all employees are required to wear a surgical mask, all day every day, requiring distribution of 35,000 masks daily.12 We also require every patient and visitor to our health care facilities to use a face mask. The purpose of the procedure or surgical mask is to prevent presymptomatic spread of COVID-19 from an asymptomatic health care worker to an uninfected patient or a colleague by reducing the transmission of respiratory droplets. Another benefit is to protect the uninfected health care worker from patients and colleagues who are infected and not yet diagnosed with COVID-19. The CDC now recommends that all people wear a mouth and nose covering when they are outside of their residence. America may become a nation where wearing masks in public becomes a routine practice. Since SARS-CoV-2 is transmitted by respiratory droplets, social distancing is an important preventive measure.
Continue to: Obstetric care...
Obstetric care
Can it be repeated too often? No. Containing COVID-19 disease requires social distancing, fastidious hand hygiene, and using a mask that covers the mouth and nose.
Pregnant women should be advised to assiduously practice social distancing and to wear a face covering or mask in public. Hand hygiene should be emphasized. Pregnant women with children should be advised to not allow their children to play with non‒cohabiting children because children may be asymptomatic vectors for COVID-19.
Pregnant health care workers should stop face-to-face contact with patients after 36 weeks’ gestation to avoid a late pregnancy infection that might cause the mother to be separated from her newborn. Based on data currently available, pregnancy in the absence of another risk factor is not a major risk factor for developing severe COVID-19 disease.13
Hyperthermia is a common feature of COVID-19. Acetaminophen is recommended treatment to suppress pyrexia during pregnancy.
The COVID-19 pandemic has transformed prenatal care from a series of face-to-face encounters at a health care facility to telemedicine either by telephone or a videoconferencing portal. Many factors contributed to the rapid switch to telemedicine, including orders by governors to restrict unnecessary travel, patients’ fear of contracting COVID-19 at their clinicians’ offices, clinicians’ fear of contracting COVID-19 from patients, and insurers’ rapid implementation of policies to pay for telemedicine visits. Most prenatal visits can be provided through telemedicine as long as the patient has a home blood pressure cuff and can reliably use the instrument. In-person visits may be required for blood testing, ultrasound assessment, anti-Rh immunoglobulin administration, and group B streptococcal infection screening. One regimen is to limit in-person prenatal visits to encounters at 12, 20, 28, and 36 weeks’ gestation when blood testing and ultrasound examinations are needed. The postpartum visit also may be conducted using telemedicine.
Pregnant women with COVID-19 and pneumonia are reported to have high rates of preterm birth less than 37 weeks (41%) and preterm prelabor rupture of membranes (19%).14
The rate of vertical transmission from mother to fetus is probably very low (<1%).15 However, based on serological studies, an occasional newborn has been reported to have IgM and IgG antibodies to the SARS-CoV-2 nucleoprotein at birth.16,17
Pregnant women should be consistently and regularly screened for symptoms of an upper respiratory infection, including: fever, new cough, new runny nose or nasal congestion, new sore throat, shortness of breath, muscle aches, and anosmia. A report of any of these symptoms should result in nucleic acid testing of a nasal swab for SARS-CoV-2 of all pregnant women. Given limited testing resources, however, symptomatic pregnant women with the following characteristics should be prioritized for testing: if the woman is more than 36 weeks pregnant, intrapartum, or in the hospital after delivery. Ambulatory pregnant women with symptoms who do not need medical care should quarantine themselves at home, if possible, or at another secure location away from their families. In some regions, testing of ambulatory patients with upper respiratory symptoms is limited.
All women scheduled for induction or cesarean delivery (CD) and their support person should have a symptom screen 24 to 48 hours before arrival to the hospital and should be rescreened prior to entry to labor and delivery. In this situation if the pregnant woman screens positive, she should be tested for SARS-CoV-2, and if the test result is positive, the scheduled induction and CD should be rescheduled, if possible. All hospitalized women and their support persons should be screened for symptoms daily. If the pregnant woman screens positive she should have a nucleic acid test for SARS-CoV-2. If the support person screens positive, he or she should be sent home.
Systemic glucocorticoids may worsen the course of COVID-19. For pregnant women with COVID-19 disease, betamethasone administration should be limited to women at high risk for preterm delivery within 7 days and only given to women between 23 weeks to 33 weeks 6 days of gestation. Women at risk for preterm delivery at 34 weeks to 36 weeks and 6 days of gestation should not be given betamethasone.
If cervical ripening is required, outpatient regimens should be prioritized.
One support person plays an important role in optimal labor outcome and should be permitted at the hospital. All support persons should wear a surgical or procedure mask.
Nitrous oxide for labor anesthesia should not be used during the pandemic because it might cause aerosolization of respiratory secretions, endangering health care workers. Neuraxial anesthesia is an optimal approach to labor anesthesia.
Labor management and timing of delivery does not need to be altered during the COVID-19 pandemic. However, pregnant women with moderate or severe COVID-19 disease who are not improving may have a modest improvement in respiratory function if they are delivered preterm.
At the beginning of the COVID pandemic, the CDC recommended separation of a COVID-positive mother and her newborn until the mother’s respiratory symptoms resolved. However, the CDC now recommends that, for a COVID-positive mother, joint decision-making should be used to decide whether to support the baby rooming-in with the mother or to practice separation of mother and baby at birth to reduce the risk for postnatal infection from mother to newborn. There is no evidence that breast milk contains virus that can cause an infection. One option is for the mother who recently tested positive for SARS-CoV-2 to provide newborn nutrition with expressed breast milk.
Pregnant women with COVID-19 may be at increased risk for venous thromboembolism. Some experts recommend that hospitalized pregnant women and postpartum women with COVID-19 receive thromboembolism prophylaxis.
The Chinese Centers for Disease Control and Prevention described a classification system for COVID-19 disease, including 3 categories18:
- mild: no dyspnea, no pneumonia, or mild pneumonia
- severe: dyspnea, respiratory frequency ≥ 30 breaths per minute, blood oxygen saturation ≤ 93%, lung infiltrates > 50% within 48 hours of onset of symptoms
- critical: respiratory failure, septic shock, or multiple organ dysfunction or failure.
Among 72,314 cases in China, 81% had mild disease, 14% had severe disease, and 5% had critical disease. In a report of 118 pregnant women in China, 92% of the women had mild disease; 8% had severe disease (hypoxemia), one of whom developed critical disease requiring mechanical ventilation.19 In this cohort, the most common presenting symptoms were fever (75%), cough (73%), chest tightness (18%), fatigue (17%), shortness of breath (7%), diarrhea (7%), and headache (6%). Lymphopenia was present in 44% of the women.
Severe and critical COVID-19 disease are associated with elevations in D-dimer, C-reactive protein, troponin, ferritin, and creatine phosphokinase levels. These markers return to the normal range with resolution of disease.
Continue to: Gynecologic care...
Gynecologic care
Gynecologists are highly impacted by the COVID-19 pandemic. Most state governments have requested that all elective surgery be suspended for the duration of the pandemic in order to redeploy health resources to the care of COVID-19 patients. Except for high-priority gynecologic surgery, including cancer surgery, treatment of heavy vaginal bleeding, and surgical care of ectopic pregnancy and miscarriage, most gynecologic surgery has ceased.
All office visits for routine gynecologic care have been suspended. Video and telephone visits can be used for contraceptive counseling and prescribing and for managing problems associated with the menopause, endometriosis, and vaginitis. Cervical cancer screening can be deferred for 3 to 6 months, depending on patient risk factors.
Medicines to treat COVID-19 infections
There are many highly effective medicines to manage HIV infection and medicines that cure hepatitis C. There is an urgent need to develop precision medicines to treat this disease. Early in the pandemic some experts thought that hydroxychloroquine might be helpful in the treatment of COVID-19 disease. But recent evidence suggests that hydroxychloroquine is probably not an effective treatment. As the pandemic has evolved, there is evidence that remdesivir may have modest efficacy in treating COVID-19 disease.20 Remdesivir has received emergency-use authorization by the FDA to treat COVID-19 infection.
Remdesivir
Based on expert opinion, in the absence of high-quality clinical trial evidence, our current practice is to offer pregnant women with severe or critical COVID-19 disease treatment with remdesivir.
Remdesivir (Gilead Sciences, Inc) is a nucleoside analog that inhibits RNA synthesis. A dose regimen for remdesivir is a 200-mg loading dose given intravenously, followed by 100 mg daily given intravenously for 5 to 10 days. Remdesivir may cause elevation of hepatic enzymes. Remdesivir has been administered to a few pregnant women to treat Ebola and Marburg virus disease.21
Experts in infectious disease are important resources for determining optimal medication regimens for the treatment of COVID-19 disease in pregnant women.
Continue to: Convalescent serum...
Convalescent serum
There are no high-quality studies demonstrating the efficacy of convalescent serum for treatment of COVID-19. A small case series suggests that there may be modest benefit to treatment of people with severe COVID-19 disease with convalescent serum.22
Testing for anti-SARS-CoV-2 IgM and IgG antibodies
We may have a serious problem in our current approach to detecting COVID-19 disease. Based on measurement of IgM and IgG antibodies to SARS-CoV-2 nucleocapsid protein, our current nucleic acid tests for SARS-CoV-2 may detect less than 80% of infections early in the course of disease. In two studies of IgM and IgG antibodies to the SARS-CoV-2 nucleocapsid protein, a single polymerase chain reaction test for SARS-CoV-2 had less than a 60% sensitivity for detecting the virus.23,24 During the second week of COVID-19 illness, IgM or IgG antibodies were detected in greater than 89% of infected patients.23 Severe disease resulted in high concentrations of antibody.
When testing for IgM and IgG antibodies is widely available, it may become an option to test all health care workers. This will permit the assignment of those health care workers with the highest levels of antibody to frontline duties with COVID-19 patients during the next disease outbreak, likely to occur at some point during the next 12 months.
A COVID-19 vaccine
Dozens of research teams, including pharmaceutical and biotechnology companies and many academic laboratories, are working on developing and testing vaccines to prevent COVID-19 disease. An effective vaccine would reduce the number of people who develop severe disease during the next outbreak, reducing deaths, avoiding a shutdown of the country, and allowing the health systems to function normally. A vaccine is unlikely to be widely available until sometime early in 2021.
Facing COVID-19 well-being and mental health
SARS-CoV-2, like all viral particles, is incredibly small. Remarkably, it has changed permanently life on earth. COVID-19 is affecting our physical health, psychological well-being, economics, and patterns of social interaction. As clinicians it is difficult to face a viral enemy that cannot be stopped from causing the death of more than 100,000 people, including some of our clinical colleagues, within a short period of time.
- F—focus on what is in your control
- A—acknowledge your thoughts and feelings
- C—come back to a focus on your body
- E—engage in what you are doing
- C—commit to acting effectively based on your core values
- O—opening up to difficult feelings and being kind to yourself and others
- V—values should guide your actions
- I—identify resources for help, assistance, support, and advice
- D—disinfect and practice social distancing.
This war will come to an end
During the American Revolution, colonists faced housing and food insecurity, epidemics of typhus and smallpox, traumatic injury including amputation of limbs, and a complete disruption of normal life activities. They persevered and, against the odds, successfully concluded the war. Unlike the colonists, who did not know if their conflict would end with success or failure, we clinicians know that the COVID-19 pandemic will end. We also know that eventually the global community of clinicians will develop and deploy the effective weapons we need to prevent a recurrence of this traumatic pandemic: population-wide testing for both the SARS-CoV-2 virus and serologic testing for IgG and IgM antibodies to the virus, effective antiviral medications, and a potent vaccine. ●
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA . doi: 10.1001/ jama . 2020 .3786.
- World Health Organization. Modes of transmission of virus causing COVID-19: implications for IPC precaution recommendations. March 29, 2020. https://www.who.int/publications-detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations. Accessed April 16, 2020.
- Arentz M, Yim E, Klaff L, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State [published online March 19, 2020]. JAMA . doi: 10.1001/ jama . 2020 .4326.
- Guan WJ, Liang WH, Zhao Y, et al; China Medical Treatment Expert Group for Covid-19. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis [published online March 26, 2020]. Eur Respir J . doi: 10.1183/13993003.00547- 2020 .
- Onder G, Rezza G, Brusaferro S. Case fatality rate and characteristics of patients dying in relation to COVID-19 in Italy [published online March 23, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4683.
- Wei WE, Li Z, Chiew CJ, et al. Presymptomatic transmission of SARS-CoV-2 - Singapore, January 23 to March 16, 2020. MMWR Morb Mortal Wkly Rep . 2020;69:411-415.
- Bai Y, Yao L, Wei T, et al. Presumed asymptomatic carrier transmission of COVID-19 [published online February 21, 2020]. JAMA. doi: 10.1001/ jama . 2020 .2565.
- Ong SW, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient [published online March 4, 2020]. JAMA . doi: 10.1001/ jama .2020.3227.
- Fineberg HV. Rapid expert consultation on the possibility of bioaerosol spread of SARS-CoV-2 for the COVID-19 pandemic. April 1, 2020. https://www.nap.edu/read/25769/chapter/1. Accessed April 16, 2020.
- Santarpia JL, River DN, Herrera V, et al. Transmission potential of SARS-CoV-2 in viral shedding observed at the University of Nebraska Medical Center. MedRxiv. March 26, 2020. doi.org10.1101/2020.03.23.20039466.
- Liu Y, Ning Z, Chen Y, et al. Aerodynamic characteristics and RNA concentration of SARS-CoV-2 aerosol in Wuhan Hospitals during COVID-19 outbreak. BioRxiv. March 10, 2020. doi.org/10.1101/2020.03.08.982637.
- Klompas M, Morris CA, Sinclair J, et al. Universal masking in hospitals in the COVID-19 era [published online April 1, 2020]. N Engl J Med. doi: 10.1056/NEJMp2006372.
- Liu D, Li L, Wu X, et al. Pregnancy and perinatal outcomes of women with coronavirus disease (COVID-19) pneumonia: a preliminary analysis. AJR Am J Roentgenol. 2020:1-6. doi: 10.2214/AJR.20.23072.
- Di Mascio D, Khalik A, Saccone G, et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol. doi:10.1016/j.ajogmf.2020.
100107. - Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA. doi: 10.1001/jama.2020.3786.
- Dong L, Tian J, He S, et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4621.
- Zeng H, Xu C, Fan J, et al. Antibodies in infants born to mothers with COVID-19 pneumonia [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4861.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the Coronavirus Diease 2019 (COVID-19) outbreak in China. Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention [published online February 24, 2020]. JAMA . doi: 10.1001/jama.2020.2648.
- Chen L, Li Q, Zheng D, et al. Clinical characteristics of pregnant women with COVID-19 in Wuhan, China [published online April 17, 2020]. N Engl J Med. doi 10.1056/NEJMc2009226.
- Chen Z, Hu J, Zhang Z, et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. MedRxiv. April 10, 2020. https://doi.org/10.1101/2020.03.22.20040758.
- Maulangu S, Dodd LE, Davey RT Jr, et al. A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med. 2019;381:2293-2303.
- Shen C, Wang Z, Zhao F, et al. Treatment of 5 critically ill patients with COVID-19 with convalescent plasma [published online March 27, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4783.
- Zhao J, Yuan Q, Wang H, et al. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019 [published online March 29, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa344.
- Guo L, Ren L, Yang S, et al. Profiling early humoral response to diagnose novel coronavirus disease (COVID-19) [published online March 21, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa310.
On June 17, 1775, American colonists, defending a forward redoubt on Breed’s Hill, ran out of gunpowder, and their position was overrun by British troops. The Battle of Bunker Hill resulted in the death of 140 colonists and 226 British soldiers, setting the stage for major combat throughout the colonies. American colonists lacked many necessary weapons. They had almost no gunpowder, few field cannons, and no warships. Yet, they fought on with the weapons at hand for 6 long years.
In the spring of 2020, American society has been shaken by the COVID-19 pandemic. Hospitals have been overrun with thousands of people infected with the disease. Some hospitals are breaking under the crush of intensely ill people filling up and spilling out of intensive care units. We are in a war, fighting a viral disease with a limited supply of weapons. We do not have access to the most powerful medical munitions: easily available rapid testing, proven antiviral medications, and an effective vaccine. Nevertheless, clinicians and patients are courageous, and we will continue the fight with the limited weapons we have until the pandemic is brought to an end.

The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19). The virus is aptly named because it is usually transmitted through close contact with respiratory droplets. The disease can progress acutely, and some people experience a remarkably severe respiratory syndrome, including tachypnea, hypoxia, and interstitial and alveolar opacities on chest x-ray, necessitating ventilatory support. The virus is an encapsulated single-stranded RNA virus. When viewed by electron microscopy, the virus appears to have a halo or crown, hence it is named “coronavirus.” Among infected individuals, the virus is present in the upper respiratory system and in feces but not in urine.1 The World Health Organization (WHO) believes that respiratory droplets and contaminated surfaces are the major routes of transmission.2 The highest risk of developing severe COVID-19 disease occurs in people with one or more of the following characteristics: age greater than 70 years, hypertension, diabetes, respiratory disease, heart disease, and immunosuppression.3,4 Pregnant women do not appear to be at increased risk for severe COVID-19 disease.4 The case fatality rate is highest in people 80 years of age or older.5
Who is infected with SARS-CoV-2?
Rapid high-fidelity testing for SARS-CoV-2 nucleic acid sequences would be the best approach to identifying people with COVID-19 disease. At the beginning of the pandemic, testing was strictly rationed because of lack of reagents and test swabs. Clinicians were permitted to test only a minority of people who had symptoms. Asymptomatic individuals were not eligible to be tested. This terribly flawed approach to screening permitted a vast army of SARS-CoV-2–positive asymptomatic and mildly symptomatic people to circulate unchecked in the general population, infecting dozens of other people, some of whom developed moderate or severe disease. The Centers for Disease Control and Prevention (CDC) has reported on 7 independent clusters of COVID-19 disease, each of which appear to have been caused by one asymptomatic infected individual.6 Another cluster of COVID-19 disease from China appears to have been caused by one asymptomatic infected individual.7 Based on limited data, it appears that there may be a 1- to 3-day window where an individual with COVID-19 may be asymptomatic and able to infect others. I suspect that we will soon discover, based on testing for the presence of high-titre anti SARS-CoV-2 antibodies, that many people with no history of illness and people with mild respiratory symptoms had an undiagnosed COVID-19 infection.
As testing capacity expands we likely will be testing all women, including asymptomatic women, before they arrive at the hospital for childbirth or gynecologic surgery, as well as all inpatients and women with respiratory symptoms having an ambulatory encounter.
With expanded testing capability, some pregnant women who were symptomatic and tested positive for SARS-CoV-2 have had sequential long-term follow-up testing. A frequent observation is that over one to two weeks the viral symptoms resolve and the nasopharyngeal test becomes negative for SARS-CoV-2 on multiple sequential tests, only to become positive at a later date. The cause of the positive-negative-negative-positive test results is unknown, but it raises the possibility that once a person tests positive for SARS-CoV-2, they may be able to transmit the infection over many weeks, even after viral symptoms resolve.
Continue to: COVID-19: Respiratory droplet or aerosol transmission?
COVID-19: Respiratory droplet or aerosol transmission?
Respiratory droplets are large particles (> 5 µm in diameter) that tend to be pulled to the ground or furniture surfaces by gravity. Respiratory droplets do not circulate in the air for an extended period of time. Droplet nuclei are small particles less than 5 µm in diameter. These small particles may become aerosolized and float through the air for an extended period of time. The CDC and WHO believe that under ordinary conditions, SARS-CoV-2 is transmitted through respiratory droplets and contact routes.2 In an analysis of more than 75,000 COVID-19 cases in China there were no reports of transmission by aerosolized airborne virus. Therefore, under ordinary conditions, surgical masks, face shields, gowns, and gloves provide a high level of protection from infection.8
In contrast to the WHO’s perspective, Dr. Harvey Fineberg, Chair of the National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, wrote a letter to the federal Office of Science and Technology Policy warning that normal breathing might generate aerosolization of the SARS-CoV-2 virus and result in airborne transmission.9 A report from the University of Nebraska Medical Center supports the concept of airborne transmission of SARS-CoV-2. In a study of 13 patients with COVID-19, room surfaces, toilet facilities, and air had evidence of viral contamination.10 The investigators concluded that disease spreads through respiratory droplets, person-to-person touch, contaminated surfaces, and airborne routes. Other investigators also have reported that aersolization of SARS-CoV-2 may occur.11 Professional societies recommend that all medical staff caring for potential or confirmed COVID-19 patients should use personal protective equipment (PPE), including respirators (N95 respirators) when available. Importantly, all medical staff should be trained in and adhere to proper donning and doffing of PPE. The controversy about the modes of transmission of SARS-CoV-2 will continue, but as clinicians we need to work within the constraints of the equipment we have.
Certain medical procedures and devices are known to generate aerosolization of respiratory secretions. These procedures and devices include: bronchoscopy, intubation, extubation, cardiopulmonary resuscitation, nebulization, high-flow oxygen masks, and continuous- and bilevel-positive airway pressure devices. When aerosols are generated during the care of a patient with COVID-19, surgical masks are not sufficient protection against infection. When an aerosol is generated maximal protection of health care workers from viral transmission requires use of a negative-pressure room and an N95 respirator or powered air-purifying respirator (PAPR) device. However, negative-pressure rooms, N95 masks, and PAPRs are in very short supply or are unavailable in some health systems. We are lucky at our hospital that all of the labor rooms can be configured to operate in a negative-pressure mode, limiting potential airborne spread of the virus on the unit. Many hospitals restrict the use of N95 masks to anesthesiologists, leaving nurses, ObGyns, and surgical technicians without the best protective equipment, risking their health. As one action to reduce aerosolization of virus, obstetricians can markedly reduce the use of oxygen masks and nasal cannulas by laboring women.
Universal use of surgical masks and mouth-nose coverings
During the entire COVID-19 pandemic, PPE has been in short supply, including severe shortages of N95 masks, PAPRs, and in some health systems, surgical masks, gowns, eye protection, and face shields. Given the severe shortages, some clinicians have needed to conserve PPE, using the same PPE across multiple patient encounters and across multiple work shifts.
Given that the virus is transmitted by respiratory droplets and contaminated surfaces, use of face coverings, including surgical masks, face shields, and gloves is critically important. Scrupulous hand hygiene is a simple approach to reducing infection risk. In my health system, all employees are required to wear a surgical mask, all day every day, requiring distribution of 35,000 masks daily.12 We also require every patient and visitor to our health care facilities to use a face mask. The purpose of the procedure or surgical mask is to prevent presymptomatic spread of COVID-19 from an asymptomatic health care worker to an uninfected patient or a colleague by reducing the transmission of respiratory droplets. Another benefit is to protect the uninfected health care worker from patients and colleagues who are infected and not yet diagnosed with COVID-19. The CDC now recommends that all people wear a mouth and nose covering when they are outside of their residence. America may become a nation where wearing masks in public becomes a routine practice. Since SARS-CoV-2 is transmitted by respiratory droplets, social distancing is an important preventive measure.
Continue to: Obstetric care...
Obstetric care
Can it be repeated too often? No. Containing COVID-19 disease requires social distancing, fastidious hand hygiene, and using a mask that covers the mouth and nose.
Pregnant women should be advised to assiduously practice social distancing and to wear a face covering or mask in public. Hand hygiene should be emphasized. Pregnant women with children should be advised to not allow their children to play with non‒cohabiting children because children may be asymptomatic vectors for COVID-19.
Pregnant health care workers should stop face-to-face contact with patients after 36 weeks’ gestation to avoid a late pregnancy infection that might cause the mother to be separated from her newborn. Based on data currently available, pregnancy in the absence of another risk factor is not a major risk factor for developing severe COVID-19 disease.13
Hyperthermia is a common feature of COVID-19. Acetaminophen is recommended treatment to suppress pyrexia during pregnancy.
The COVID-19 pandemic has transformed prenatal care from a series of face-to-face encounters at a health care facility to telemedicine either by telephone or a videoconferencing portal. Many factors contributed to the rapid switch to telemedicine, including orders by governors to restrict unnecessary travel, patients’ fear of contracting COVID-19 at their clinicians’ offices, clinicians’ fear of contracting COVID-19 from patients, and insurers’ rapid implementation of policies to pay for telemedicine visits. Most prenatal visits can be provided through telemedicine as long as the patient has a home blood pressure cuff and can reliably use the instrument. In-person visits may be required for blood testing, ultrasound assessment, anti-Rh immunoglobulin administration, and group B streptococcal infection screening. One regimen is to limit in-person prenatal visits to encounters at 12, 20, 28, and 36 weeks’ gestation when blood testing and ultrasound examinations are needed. The postpartum visit also may be conducted using telemedicine.
Pregnant women with COVID-19 and pneumonia are reported to have high rates of preterm birth less than 37 weeks (41%) and preterm prelabor rupture of membranes (19%).14
The rate of vertical transmission from mother to fetus is probably very low (<1%).15 However, based on serological studies, an occasional newborn has been reported to have IgM and IgG antibodies to the SARS-CoV-2 nucleoprotein at birth.16,17
Pregnant women should be consistently and regularly screened for symptoms of an upper respiratory infection, including: fever, new cough, new runny nose or nasal congestion, new sore throat, shortness of breath, muscle aches, and anosmia. A report of any of these symptoms should result in nucleic acid testing of a nasal swab for SARS-CoV-2 of all pregnant women. Given limited testing resources, however, symptomatic pregnant women with the following characteristics should be prioritized for testing: if the woman is more than 36 weeks pregnant, intrapartum, or in the hospital after delivery. Ambulatory pregnant women with symptoms who do not need medical care should quarantine themselves at home, if possible, or at another secure location away from their families. In some regions, testing of ambulatory patients with upper respiratory symptoms is limited.
All women scheduled for induction or cesarean delivery (CD) and their support person should have a symptom screen 24 to 48 hours before arrival to the hospital and should be rescreened prior to entry to labor and delivery. In this situation if the pregnant woman screens positive, she should be tested for SARS-CoV-2, and if the test result is positive, the scheduled induction and CD should be rescheduled, if possible. All hospitalized women and their support persons should be screened for symptoms daily. If the pregnant woman screens positive she should have a nucleic acid test for SARS-CoV-2. If the support person screens positive, he or she should be sent home.
Systemic glucocorticoids may worsen the course of COVID-19. For pregnant women with COVID-19 disease, betamethasone administration should be limited to women at high risk for preterm delivery within 7 days and only given to women between 23 weeks to 33 weeks 6 days of gestation. Women at risk for preterm delivery at 34 weeks to 36 weeks and 6 days of gestation should not be given betamethasone.
If cervical ripening is required, outpatient regimens should be prioritized.
One support person plays an important role in optimal labor outcome and should be permitted at the hospital. All support persons should wear a surgical or procedure mask.
Nitrous oxide for labor anesthesia should not be used during the pandemic because it might cause aerosolization of respiratory secretions, endangering health care workers. Neuraxial anesthesia is an optimal approach to labor anesthesia.
Labor management and timing of delivery does not need to be altered during the COVID-19 pandemic. However, pregnant women with moderate or severe COVID-19 disease who are not improving may have a modest improvement in respiratory function if they are delivered preterm.
At the beginning of the COVID pandemic, the CDC recommended separation of a COVID-positive mother and her newborn until the mother’s respiratory symptoms resolved. However, the CDC now recommends that, for a COVID-positive mother, joint decision-making should be used to decide whether to support the baby rooming-in with the mother or to practice separation of mother and baby at birth to reduce the risk for postnatal infection from mother to newborn. There is no evidence that breast milk contains virus that can cause an infection. One option is for the mother who recently tested positive for SARS-CoV-2 to provide newborn nutrition with expressed breast milk.
Pregnant women with COVID-19 may be at increased risk for venous thromboembolism. Some experts recommend that hospitalized pregnant women and postpartum women with COVID-19 receive thromboembolism prophylaxis.
The Chinese Centers for Disease Control and Prevention described a classification system for COVID-19 disease, including 3 categories18:
- mild: no dyspnea, no pneumonia, or mild pneumonia
- severe: dyspnea, respiratory frequency ≥ 30 breaths per minute, blood oxygen saturation ≤ 93%, lung infiltrates > 50% within 48 hours of onset of symptoms
- critical: respiratory failure, septic shock, or multiple organ dysfunction or failure.
Among 72,314 cases in China, 81% had mild disease, 14% had severe disease, and 5% had critical disease. In a report of 118 pregnant women in China, 92% of the women had mild disease; 8% had severe disease (hypoxemia), one of whom developed critical disease requiring mechanical ventilation.19 In this cohort, the most common presenting symptoms were fever (75%), cough (73%), chest tightness (18%), fatigue (17%), shortness of breath (7%), diarrhea (7%), and headache (6%). Lymphopenia was present in 44% of the women.
Severe and critical COVID-19 disease are associated with elevations in D-dimer, C-reactive protein, troponin, ferritin, and creatine phosphokinase levels. These markers return to the normal range with resolution of disease.
Continue to: Gynecologic care...
Gynecologic care
Gynecologists are highly impacted by the COVID-19 pandemic. Most state governments have requested that all elective surgery be suspended for the duration of the pandemic in order to redeploy health resources to the care of COVID-19 patients. Except for high-priority gynecologic surgery, including cancer surgery, treatment of heavy vaginal bleeding, and surgical care of ectopic pregnancy and miscarriage, most gynecologic surgery has ceased.
All office visits for routine gynecologic care have been suspended. Video and telephone visits can be used for contraceptive counseling and prescribing and for managing problems associated with the menopause, endometriosis, and vaginitis. Cervical cancer screening can be deferred for 3 to 6 months, depending on patient risk factors.
Medicines to treat COVID-19 infections
There are many highly effective medicines to manage HIV infection and medicines that cure hepatitis C. There is an urgent need to develop precision medicines to treat this disease. Early in the pandemic some experts thought that hydroxychloroquine might be helpful in the treatment of COVID-19 disease. But recent evidence suggests that hydroxychloroquine is probably not an effective treatment. As the pandemic has evolved, there is evidence that remdesivir may have modest efficacy in treating COVID-19 disease.20 Remdesivir has received emergency-use authorization by the FDA to treat COVID-19 infection.
Remdesivir
Based on expert opinion, in the absence of high-quality clinical trial evidence, our current practice is to offer pregnant women with severe or critical COVID-19 disease treatment with remdesivir.
Remdesivir (Gilead Sciences, Inc) is a nucleoside analog that inhibits RNA synthesis. A dose regimen for remdesivir is a 200-mg loading dose given intravenously, followed by 100 mg daily given intravenously for 5 to 10 days. Remdesivir may cause elevation of hepatic enzymes. Remdesivir has been administered to a few pregnant women to treat Ebola and Marburg virus disease.21
Experts in infectious disease are important resources for determining optimal medication regimens for the treatment of COVID-19 disease in pregnant women.
Continue to: Convalescent serum...
Convalescent serum
There are no high-quality studies demonstrating the efficacy of convalescent serum for treatment of COVID-19. A small case series suggests that there may be modest benefit to treatment of people with severe COVID-19 disease with convalescent serum.22
Testing for anti-SARS-CoV-2 IgM and IgG antibodies
We may have a serious problem in our current approach to detecting COVID-19 disease. Based on measurement of IgM and IgG antibodies to SARS-CoV-2 nucleocapsid protein, our current nucleic acid tests for SARS-CoV-2 may detect less than 80% of infections early in the course of disease. In two studies of IgM and IgG antibodies to the SARS-CoV-2 nucleocapsid protein, a single polymerase chain reaction test for SARS-CoV-2 had less than a 60% sensitivity for detecting the virus.23,24 During the second week of COVID-19 illness, IgM or IgG antibodies were detected in greater than 89% of infected patients.23 Severe disease resulted in high concentrations of antibody.
When testing for IgM and IgG antibodies is widely available, it may become an option to test all health care workers. This will permit the assignment of those health care workers with the highest levels of antibody to frontline duties with COVID-19 patients during the next disease outbreak, likely to occur at some point during the next 12 months.
A COVID-19 vaccine
Dozens of research teams, including pharmaceutical and biotechnology companies and many academic laboratories, are working on developing and testing vaccines to prevent COVID-19 disease. An effective vaccine would reduce the number of people who develop severe disease during the next outbreak, reducing deaths, avoiding a shutdown of the country, and allowing the health systems to function normally. A vaccine is unlikely to be widely available until sometime early in 2021.
Facing COVID-19 well-being and mental health
SARS-CoV-2, like all viral particles, is incredibly small. Remarkably, it has changed permanently life on earth. COVID-19 is affecting our physical health, psychological well-being, economics, and patterns of social interaction. As clinicians it is difficult to face a viral enemy that cannot be stopped from causing the death of more than 100,000 people, including some of our clinical colleagues, within a short period of time.
- F—focus on what is in your control
- A—acknowledge your thoughts and feelings
- C—come back to a focus on your body
- E—engage in what you are doing
- C—commit to acting effectively based on your core values
- O—opening up to difficult feelings and being kind to yourself and others
- V—values should guide your actions
- I—identify resources for help, assistance, support, and advice
- D—disinfect and practice social distancing.
This war will come to an end
During the American Revolution, colonists faced housing and food insecurity, epidemics of typhus and smallpox, traumatic injury including amputation of limbs, and a complete disruption of normal life activities. They persevered and, against the odds, successfully concluded the war. Unlike the colonists, who did not know if their conflict would end with success or failure, we clinicians know that the COVID-19 pandemic will end. We also know that eventually the global community of clinicians will develop and deploy the effective weapons we need to prevent a recurrence of this traumatic pandemic: population-wide testing for both the SARS-CoV-2 virus and serologic testing for IgG and IgM antibodies to the virus, effective antiviral medications, and a potent vaccine. ●
On June 17, 1775, American colonists, defending a forward redoubt on Breed’s Hill, ran out of gunpowder, and their position was overrun by British troops. The Battle of Bunker Hill resulted in the death of 140 colonists and 226 British soldiers, setting the stage for major combat throughout the colonies. American colonists lacked many necessary weapons. They had almost no gunpowder, few field cannons, and no warships. Yet, they fought on with the weapons at hand for 6 long years.
In the spring of 2020, American society has been shaken by the COVID-19 pandemic. Hospitals have been overrun with thousands of people infected with the disease. Some hospitals are breaking under the crush of intensely ill people filling up and spilling out of intensive care units. We are in a war, fighting a viral disease with a limited supply of weapons. We do not have access to the most powerful medical munitions: easily available rapid testing, proven antiviral medications, and an effective vaccine. Nevertheless, clinicians and patients are courageous, and we will continue the fight with the limited weapons we have until the pandemic is brought to an end.

The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19). The virus is aptly named because it is usually transmitted through close contact with respiratory droplets. The disease can progress acutely, and some people experience a remarkably severe respiratory syndrome, including tachypnea, hypoxia, and interstitial and alveolar opacities on chest x-ray, necessitating ventilatory support. The virus is an encapsulated single-stranded RNA virus. When viewed by electron microscopy, the virus appears to have a halo or crown, hence it is named “coronavirus.” Among infected individuals, the virus is present in the upper respiratory system and in feces but not in urine.1 The World Health Organization (WHO) believes that respiratory droplets and contaminated surfaces are the major routes of transmission.2 The highest risk of developing severe COVID-19 disease occurs in people with one or more of the following characteristics: age greater than 70 years, hypertension, diabetes, respiratory disease, heart disease, and immunosuppression.3,4 Pregnant women do not appear to be at increased risk for severe COVID-19 disease.4 The case fatality rate is highest in people 80 years of age or older.5
Who is infected with SARS-CoV-2?
Rapid high-fidelity testing for SARS-CoV-2 nucleic acid sequences would be the best approach to identifying people with COVID-19 disease. At the beginning of the pandemic, testing was strictly rationed because of lack of reagents and test swabs. Clinicians were permitted to test only a minority of people who had symptoms. Asymptomatic individuals were not eligible to be tested. This terribly flawed approach to screening permitted a vast army of SARS-CoV-2–positive asymptomatic and mildly symptomatic people to circulate unchecked in the general population, infecting dozens of other people, some of whom developed moderate or severe disease. The Centers for Disease Control and Prevention (CDC) has reported on 7 independent clusters of COVID-19 disease, each of which appear to have been caused by one asymptomatic infected individual.6 Another cluster of COVID-19 disease from China appears to have been caused by one asymptomatic infected individual.7 Based on limited data, it appears that there may be a 1- to 3-day window where an individual with COVID-19 may be asymptomatic and able to infect others. I suspect that we will soon discover, based on testing for the presence of high-titre anti SARS-CoV-2 antibodies, that many people with no history of illness and people with mild respiratory symptoms had an undiagnosed COVID-19 infection.
As testing capacity expands we likely will be testing all women, including asymptomatic women, before they arrive at the hospital for childbirth or gynecologic surgery, as well as all inpatients and women with respiratory symptoms having an ambulatory encounter.
With expanded testing capability, some pregnant women who were symptomatic and tested positive for SARS-CoV-2 have had sequential long-term follow-up testing. A frequent observation is that over one to two weeks the viral symptoms resolve and the nasopharyngeal test becomes negative for SARS-CoV-2 on multiple sequential tests, only to become positive at a later date. The cause of the positive-negative-negative-positive test results is unknown, but it raises the possibility that once a person tests positive for SARS-CoV-2, they may be able to transmit the infection over many weeks, even after viral symptoms resolve.
Continue to: COVID-19: Respiratory droplet or aerosol transmission?
COVID-19: Respiratory droplet or aerosol transmission?
Respiratory droplets are large particles (> 5 µm in diameter) that tend to be pulled to the ground or furniture surfaces by gravity. Respiratory droplets do not circulate in the air for an extended period of time. Droplet nuclei are small particles less than 5 µm in diameter. These small particles may become aerosolized and float through the air for an extended period of time. The CDC and WHO believe that under ordinary conditions, SARS-CoV-2 is transmitted through respiratory droplets and contact routes.2 In an analysis of more than 75,000 COVID-19 cases in China there were no reports of transmission by aerosolized airborne virus. Therefore, under ordinary conditions, surgical masks, face shields, gowns, and gloves provide a high level of protection from infection.8
In contrast to the WHO’s perspective, Dr. Harvey Fineberg, Chair of the National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats, wrote a letter to the federal Office of Science and Technology Policy warning that normal breathing might generate aerosolization of the SARS-CoV-2 virus and result in airborne transmission.9 A report from the University of Nebraska Medical Center supports the concept of airborne transmission of SARS-CoV-2. In a study of 13 patients with COVID-19, room surfaces, toilet facilities, and air had evidence of viral contamination.10 The investigators concluded that disease spreads through respiratory droplets, person-to-person touch, contaminated surfaces, and airborne routes. Other investigators also have reported that aersolization of SARS-CoV-2 may occur.11 Professional societies recommend that all medical staff caring for potential or confirmed COVID-19 patients should use personal protective equipment (PPE), including respirators (N95 respirators) when available. Importantly, all medical staff should be trained in and adhere to proper donning and doffing of PPE. The controversy about the modes of transmission of SARS-CoV-2 will continue, but as clinicians we need to work within the constraints of the equipment we have.
Certain medical procedures and devices are known to generate aerosolization of respiratory secretions. These procedures and devices include: bronchoscopy, intubation, extubation, cardiopulmonary resuscitation, nebulization, high-flow oxygen masks, and continuous- and bilevel-positive airway pressure devices. When aerosols are generated during the care of a patient with COVID-19, surgical masks are not sufficient protection against infection. When an aerosol is generated maximal protection of health care workers from viral transmission requires use of a negative-pressure room and an N95 respirator or powered air-purifying respirator (PAPR) device. However, negative-pressure rooms, N95 masks, and PAPRs are in very short supply or are unavailable in some health systems. We are lucky at our hospital that all of the labor rooms can be configured to operate in a negative-pressure mode, limiting potential airborne spread of the virus on the unit. Many hospitals restrict the use of N95 masks to anesthesiologists, leaving nurses, ObGyns, and surgical technicians without the best protective equipment, risking their health. As one action to reduce aerosolization of virus, obstetricians can markedly reduce the use of oxygen masks and nasal cannulas by laboring women.
Universal use of surgical masks and mouth-nose coverings
During the entire COVID-19 pandemic, PPE has been in short supply, including severe shortages of N95 masks, PAPRs, and in some health systems, surgical masks, gowns, eye protection, and face shields. Given the severe shortages, some clinicians have needed to conserve PPE, using the same PPE across multiple patient encounters and across multiple work shifts.
Given that the virus is transmitted by respiratory droplets and contaminated surfaces, use of face coverings, including surgical masks, face shields, and gloves is critically important. Scrupulous hand hygiene is a simple approach to reducing infection risk. In my health system, all employees are required to wear a surgical mask, all day every day, requiring distribution of 35,000 masks daily.12 We also require every patient and visitor to our health care facilities to use a face mask. The purpose of the procedure or surgical mask is to prevent presymptomatic spread of COVID-19 from an asymptomatic health care worker to an uninfected patient or a colleague by reducing the transmission of respiratory droplets. Another benefit is to protect the uninfected health care worker from patients and colleagues who are infected and not yet diagnosed with COVID-19. The CDC now recommends that all people wear a mouth and nose covering when they are outside of their residence. America may become a nation where wearing masks in public becomes a routine practice. Since SARS-CoV-2 is transmitted by respiratory droplets, social distancing is an important preventive measure.
Continue to: Obstetric care...
Obstetric care
Can it be repeated too often? No. Containing COVID-19 disease requires social distancing, fastidious hand hygiene, and using a mask that covers the mouth and nose.
Pregnant women should be advised to assiduously practice social distancing and to wear a face covering or mask in public. Hand hygiene should be emphasized. Pregnant women with children should be advised to not allow their children to play with non‒cohabiting children because children may be asymptomatic vectors for COVID-19.
Pregnant health care workers should stop face-to-face contact with patients after 36 weeks’ gestation to avoid a late pregnancy infection that might cause the mother to be separated from her newborn. Based on data currently available, pregnancy in the absence of another risk factor is not a major risk factor for developing severe COVID-19 disease.13
Hyperthermia is a common feature of COVID-19. Acetaminophen is recommended treatment to suppress pyrexia during pregnancy.
The COVID-19 pandemic has transformed prenatal care from a series of face-to-face encounters at a health care facility to telemedicine either by telephone or a videoconferencing portal. Many factors contributed to the rapid switch to telemedicine, including orders by governors to restrict unnecessary travel, patients’ fear of contracting COVID-19 at their clinicians’ offices, clinicians’ fear of contracting COVID-19 from patients, and insurers’ rapid implementation of policies to pay for telemedicine visits. Most prenatal visits can be provided through telemedicine as long as the patient has a home blood pressure cuff and can reliably use the instrument. In-person visits may be required for blood testing, ultrasound assessment, anti-Rh immunoglobulin administration, and group B streptococcal infection screening. One regimen is to limit in-person prenatal visits to encounters at 12, 20, 28, and 36 weeks’ gestation when blood testing and ultrasound examinations are needed. The postpartum visit also may be conducted using telemedicine.
Pregnant women with COVID-19 and pneumonia are reported to have high rates of preterm birth less than 37 weeks (41%) and preterm prelabor rupture of membranes (19%).14
The rate of vertical transmission from mother to fetus is probably very low (<1%).15 However, based on serological studies, an occasional newborn has been reported to have IgM and IgG antibodies to the SARS-CoV-2 nucleoprotein at birth.16,17
Pregnant women should be consistently and regularly screened for symptoms of an upper respiratory infection, including: fever, new cough, new runny nose or nasal congestion, new sore throat, shortness of breath, muscle aches, and anosmia. A report of any of these symptoms should result in nucleic acid testing of a nasal swab for SARS-CoV-2 of all pregnant women. Given limited testing resources, however, symptomatic pregnant women with the following characteristics should be prioritized for testing: if the woman is more than 36 weeks pregnant, intrapartum, or in the hospital after delivery. Ambulatory pregnant women with symptoms who do not need medical care should quarantine themselves at home, if possible, or at another secure location away from their families. In some regions, testing of ambulatory patients with upper respiratory symptoms is limited.
All women scheduled for induction or cesarean delivery (CD) and their support person should have a symptom screen 24 to 48 hours before arrival to the hospital and should be rescreened prior to entry to labor and delivery. In this situation if the pregnant woman screens positive, she should be tested for SARS-CoV-2, and if the test result is positive, the scheduled induction and CD should be rescheduled, if possible. All hospitalized women and their support persons should be screened for symptoms daily. If the pregnant woman screens positive she should have a nucleic acid test for SARS-CoV-2. If the support person screens positive, he or she should be sent home.
Systemic glucocorticoids may worsen the course of COVID-19. For pregnant women with COVID-19 disease, betamethasone administration should be limited to women at high risk for preterm delivery within 7 days and only given to women between 23 weeks to 33 weeks 6 days of gestation. Women at risk for preterm delivery at 34 weeks to 36 weeks and 6 days of gestation should not be given betamethasone.
If cervical ripening is required, outpatient regimens should be prioritized.
One support person plays an important role in optimal labor outcome and should be permitted at the hospital. All support persons should wear a surgical or procedure mask.
Nitrous oxide for labor anesthesia should not be used during the pandemic because it might cause aerosolization of respiratory secretions, endangering health care workers. Neuraxial anesthesia is an optimal approach to labor anesthesia.
Labor management and timing of delivery does not need to be altered during the COVID-19 pandemic. However, pregnant women with moderate or severe COVID-19 disease who are not improving may have a modest improvement in respiratory function if they are delivered preterm.
At the beginning of the COVID pandemic, the CDC recommended separation of a COVID-positive mother and her newborn until the mother’s respiratory symptoms resolved. However, the CDC now recommends that, for a COVID-positive mother, joint decision-making should be used to decide whether to support the baby rooming-in with the mother or to practice separation of mother and baby at birth to reduce the risk for postnatal infection from mother to newborn. There is no evidence that breast milk contains virus that can cause an infection. One option is for the mother who recently tested positive for SARS-CoV-2 to provide newborn nutrition with expressed breast milk.
Pregnant women with COVID-19 may be at increased risk for venous thromboembolism. Some experts recommend that hospitalized pregnant women and postpartum women with COVID-19 receive thromboembolism prophylaxis.
The Chinese Centers for Disease Control and Prevention described a classification system for COVID-19 disease, including 3 categories18:
- mild: no dyspnea, no pneumonia, or mild pneumonia
- severe: dyspnea, respiratory frequency ≥ 30 breaths per minute, blood oxygen saturation ≤ 93%, lung infiltrates > 50% within 48 hours of onset of symptoms
- critical: respiratory failure, septic shock, or multiple organ dysfunction or failure.
Among 72,314 cases in China, 81% had mild disease, 14% had severe disease, and 5% had critical disease. In a report of 118 pregnant women in China, 92% of the women had mild disease; 8% had severe disease (hypoxemia), one of whom developed critical disease requiring mechanical ventilation.19 In this cohort, the most common presenting symptoms were fever (75%), cough (73%), chest tightness (18%), fatigue (17%), shortness of breath (7%), diarrhea (7%), and headache (6%). Lymphopenia was present in 44% of the women.
Severe and critical COVID-19 disease are associated with elevations in D-dimer, C-reactive protein, troponin, ferritin, and creatine phosphokinase levels. These markers return to the normal range with resolution of disease.
Continue to: Gynecologic care...
Gynecologic care
Gynecologists are highly impacted by the COVID-19 pandemic. Most state governments have requested that all elective surgery be suspended for the duration of the pandemic in order to redeploy health resources to the care of COVID-19 patients. Except for high-priority gynecologic surgery, including cancer surgery, treatment of heavy vaginal bleeding, and surgical care of ectopic pregnancy and miscarriage, most gynecologic surgery has ceased.
All office visits for routine gynecologic care have been suspended. Video and telephone visits can be used for contraceptive counseling and prescribing and for managing problems associated with the menopause, endometriosis, and vaginitis. Cervical cancer screening can be deferred for 3 to 6 months, depending on patient risk factors.
Medicines to treat COVID-19 infections
There are many highly effective medicines to manage HIV infection and medicines that cure hepatitis C. There is an urgent need to develop precision medicines to treat this disease. Early in the pandemic some experts thought that hydroxychloroquine might be helpful in the treatment of COVID-19 disease. But recent evidence suggests that hydroxychloroquine is probably not an effective treatment. As the pandemic has evolved, there is evidence that remdesivir may have modest efficacy in treating COVID-19 disease.20 Remdesivir has received emergency-use authorization by the FDA to treat COVID-19 infection.
Remdesivir
Based on expert opinion, in the absence of high-quality clinical trial evidence, our current practice is to offer pregnant women with severe or critical COVID-19 disease treatment with remdesivir.
Remdesivir (Gilead Sciences, Inc) is a nucleoside analog that inhibits RNA synthesis. A dose regimen for remdesivir is a 200-mg loading dose given intravenously, followed by 100 mg daily given intravenously for 5 to 10 days. Remdesivir may cause elevation of hepatic enzymes. Remdesivir has been administered to a few pregnant women to treat Ebola and Marburg virus disease.21
Experts in infectious disease are important resources for determining optimal medication regimens for the treatment of COVID-19 disease in pregnant women.
Continue to: Convalescent serum...
Convalescent serum
There are no high-quality studies demonstrating the efficacy of convalescent serum for treatment of COVID-19. A small case series suggests that there may be modest benefit to treatment of people with severe COVID-19 disease with convalescent serum.22
Testing for anti-SARS-CoV-2 IgM and IgG antibodies
We may have a serious problem in our current approach to detecting COVID-19 disease. Based on measurement of IgM and IgG antibodies to SARS-CoV-2 nucleocapsid protein, our current nucleic acid tests for SARS-CoV-2 may detect less than 80% of infections early in the course of disease. In two studies of IgM and IgG antibodies to the SARS-CoV-2 nucleocapsid protein, a single polymerase chain reaction test for SARS-CoV-2 had less than a 60% sensitivity for detecting the virus.23,24 During the second week of COVID-19 illness, IgM or IgG antibodies were detected in greater than 89% of infected patients.23 Severe disease resulted in high concentrations of antibody.
When testing for IgM and IgG antibodies is widely available, it may become an option to test all health care workers. This will permit the assignment of those health care workers with the highest levels of antibody to frontline duties with COVID-19 patients during the next disease outbreak, likely to occur at some point during the next 12 months.
A COVID-19 vaccine
Dozens of research teams, including pharmaceutical and biotechnology companies and many academic laboratories, are working on developing and testing vaccines to prevent COVID-19 disease. An effective vaccine would reduce the number of people who develop severe disease during the next outbreak, reducing deaths, avoiding a shutdown of the country, and allowing the health systems to function normally. A vaccine is unlikely to be widely available until sometime early in 2021.
Facing COVID-19 well-being and mental health
SARS-CoV-2, like all viral particles, is incredibly small. Remarkably, it has changed permanently life on earth. COVID-19 is affecting our physical health, psychological well-being, economics, and patterns of social interaction. As clinicians it is difficult to face a viral enemy that cannot be stopped from causing the death of more than 100,000 people, including some of our clinical colleagues, within a short period of time.
- F—focus on what is in your control
- A—acknowledge your thoughts and feelings
- C—come back to a focus on your body
- E—engage in what you are doing
- C—commit to acting effectively based on your core values
- O—opening up to difficult feelings and being kind to yourself and others
- V—values should guide your actions
- I—identify resources for help, assistance, support, and advice
- D—disinfect and practice social distancing.
This war will come to an end
During the American Revolution, colonists faced housing and food insecurity, epidemics of typhus and smallpox, traumatic injury including amputation of limbs, and a complete disruption of normal life activities. They persevered and, against the odds, successfully concluded the war. Unlike the colonists, who did not know if their conflict would end with success or failure, we clinicians know that the COVID-19 pandemic will end. We also know that eventually the global community of clinicians will develop and deploy the effective weapons we need to prevent a recurrence of this traumatic pandemic: population-wide testing for both the SARS-CoV-2 virus and serologic testing for IgG and IgM antibodies to the virus, effective antiviral medications, and a potent vaccine. ●
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA . doi: 10.1001/ jama . 2020 .3786.
- World Health Organization. Modes of transmission of virus causing COVID-19: implications for IPC precaution recommendations. March 29, 2020. https://www.who.int/publications-detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations. Accessed April 16, 2020.
- Arentz M, Yim E, Klaff L, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State [published online March 19, 2020]. JAMA . doi: 10.1001/ jama . 2020 .4326.
- Guan WJ, Liang WH, Zhao Y, et al; China Medical Treatment Expert Group for Covid-19. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis [published online March 26, 2020]. Eur Respir J . doi: 10.1183/13993003.00547- 2020 .
- Onder G, Rezza G, Brusaferro S. Case fatality rate and characteristics of patients dying in relation to COVID-19 in Italy [published online March 23, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4683.
- Wei WE, Li Z, Chiew CJ, et al. Presymptomatic transmission of SARS-CoV-2 - Singapore, January 23 to March 16, 2020. MMWR Morb Mortal Wkly Rep . 2020;69:411-415.
- Bai Y, Yao L, Wei T, et al. Presumed asymptomatic carrier transmission of COVID-19 [published online February 21, 2020]. JAMA. doi: 10.1001/ jama . 2020 .2565.
- Ong SW, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient [published online March 4, 2020]. JAMA . doi: 10.1001/ jama .2020.3227.
- Fineberg HV. Rapid expert consultation on the possibility of bioaerosol spread of SARS-CoV-2 for the COVID-19 pandemic. April 1, 2020. https://www.nap.edu/read/25769/chapter/1. Accessed April 16, 2020.
- Santarpia JL, River DN, Herrera V, et al. Transmission potential of SARS-CoV-2 in viral shedding observed at the University of Nebraska Medical Center. MedRxiv. March 26, 2020. doi.org10.1101/2020.03.23.20039466.
- Liu Y, Ning Z, Chen Y, et al. Aerodynamic characteristics and RNA concentration of SARS-CoV-2 aerosol in Wuhan Hospitals during COVID-19 outbreak. BioRxiv. March 10, 2020. doi.org/10.1101/2020.03.08.982637.
- Klompas M, Morris CA, Sinclair J, et al. Universal masking in hospitals in the COVID-19 era [published online April 1, 2020]. N Engl J Med. doi: 10.1056/NEJMp2006372.
- Liu D, Li L, Wu X, et al. Pregnancy and perinatal outcomes of women with coronavirus disease (COVID-19) pneumonia: a preliminary analysis. AJR Am J Roentgenol. 2020:1-6. doi: 10.2214/AJR.20.23072.
- Di Mascio D, Khalik A, Saccone G, et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol. doi:10.1016/j.ajogmf.2020.
100107. - Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA. doi: 10.1001/jama.2020.3786.
- Dong L, Tian J, He S, et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4621.
- Zeng H, Xu C, Fan J, et al. Antibodies in infants born to mothers with COVID-19 pneumonia [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4861.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the Coronavirus Diease 2019 (COVID-19) outbreak in China. Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention [published online February 24, 2020]. JAMA . doi: 10.1001/jama.2020.2648.
- Chen L, Li Q, Zheng D, et al. Clinical characteristics of pregnant women with COVID-19 in Wuhan, China [published online April 17, 2020]. N Engl J Med. doi 10.1056/NEJMc2009226.
- Chen Z, Hu J, Zhang Z, et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. MedRxiv. April 10, 2020. https://doi.org/10.1101/2020.03.22.20040758.
- Maulangu S, Dodd LE, Davey RT Jr, et al. A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med. 2019;381:2293-2303.
- Shen C, Wang Z, Zhao F, et al. Treatment of 5 critically ill patients with COVID-19 with convalescent plasma [published online March 27, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4783.
- Zhao J, Yuan Q, Wang H, et al. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019 [published online March 29, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa344.
- Guo L, Ren L, Yang S, et al. Profiling early humoral response to diagnose novel coronavirus disease (COVID-19) [published online March 21, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa310.
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA . doi: 10.1001/ jama . 2020 .3786.
- World Health Organization. Modes of transmission of virus causing COVID-19: implications for IPC precaution recommendations. March 29, 2020. https://www.who.int/publications-detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations. Accessed April 16, 2020.
- Arentz M, Yim E, Klaff L, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State [published online March 19, 2020]. JAMA . doi: 10.1001/ jama . 2020 .4326.
- Guan WJ, Liang WH, Zhao Y, et al; China Medical Treatment Expert Group for Covid-19. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis [published online March 26, 2020]. Eur Respir J . doi: 10.1183/13993003.00547- 2020 .
- Onder G, Rezza G, Brusaferro S. Case fatality rate and characteristics of patients dying in relation to COVID-19 in Italy [published online March 23, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4683.
- Wei WE, Li Z, Chiew CJ, et al. Presymptomatic transmission of SARS-CoV-2 - Singapore, January 23 to March 16, 2020. MMWR Morb Mortal Wkly Rep . 2020;69:411-415.
- Bai Y, Yao L, Wei T, et al. Presumed asymptomatic carrier transmission of COVID-19 [published online February 21, 2020]. JAMA. doi: 10.1001/ jama . 2020 .2565.
- Ong SW, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient [published online March 4, 2020]. JAMA . doi: 10.1001/ jama .2020.3227.
- Fineberg HV. Rapid expert consultation on the possibility of bioaerosol spread of SARS-CoV-2 for the COVID-19 pandemic. April 1, 2020. https://www.nap.edu/read/25769/chapter/1. Accessed April 16, 2020.
- Santarpia JL, River DN, Herrera V, et al. Transmission potential of SARS-CoV-2 in viral shedding observed at the University of Nebraska Medical Center. MedRxiv. March 26, 2020. doi.org10.1101/2020.03.23.20039466.
- Liu Y, Ning Z, Chen Y, et al. Aerodynamic characteristics and RNA concentration of SARS-CoV-2 aerosol in Wuhan Hospitals during COVID-19 outbreak. BioRxiv. March 10, 2020. doi.org/10.1101/2020.03.08.982637.
- Klompas M, Morris CA, Sinclair J, et al. Universal masking in hospitals in the COVID-19 era [published online April 1, 2020]. N Engl J Med. doi: 10.1056/NEJMp2006372.
- Liu D, Li L, Wu X, et al. Pregnancy and perinatal outcomes of women with coronavirus disease (COVID-19) pneumonia: a preliminary analysis. AJR Am J Roentgenol. 2020:1-6. doi: 10.2214/AJR.20.23072.
- Di Mascio D, Khalik A, Saccone G, et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: a systematic review and meta-analysis. Am J Obstet Gynecol. doi:10.1016/j.ajogmf.2020.
100107. - Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens [published online March 11, 2020]. JAMA. doi: 10.1001/jama.2020.3786.
- Dong L, Tian J, He S, et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4621.
- Zeng H, Xu C, Fan J, et al. Antibodies in infants born to mothers with COVID-19 pneumonia [published online March 26, 2020]. JAMA. doi: 10.1001/ jama .2020.4861.
- Wu Z, McGoogan JM. Characteristics of and important lessons from the Coronavirus Diease 2019 (COVID-19) outbreak in China. Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention [published online February 24, 2020]. JAMA . doi: 10.1001/jama.2020.2648.
- Chen L, Li Q, Zheng D, et al. Clinical characteristics of pregnant women with COVID-19 in Wuhan, China [published online April 17, 2020]. N Engl J Med. doi 10.1056/NEJMc2009226.
- Chen Z, Hu J, Zhang Z, et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. MedRxiv. April 10, 2020. https://doi.org/10.1101/2020.03.22.20040758.
- Maulangu S, Dodd LE, Davey RT Jr, et al. A randomized, controlled trial of Ebola virus disease therapeutics. N Engl J Med. 2019;381:2293-2303.
- Shen C, Wang Z, Zhao F, et al. Treatment of 5 critically ill patients with COVID-19 with convalescent plasma [published online March 27, 2020]. JAMA. doi: 10.1001/ jama . 2020 .4783.
- Zhao J, Yuan Q, Wang H, et al. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019 [published online March 29, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa344.
- Guo L, Ren L, Yang S, et al. Profiling early humoral response to diagnose novel coronavirus disease (COVID-19) [published online March 21, 2020]. Clin Infect Dis. doi: 10.1093/cid/ciaa310.
Tip Sheet: Teledermatology 101



FDA grants EUA to muscle stimulator to reduce mechanical ventilator usage
The Food and Drug Administration has issued an Emergency Use Authorization (EUA) for the VentFree Respiratory Muscle Stimulator in order to potentially reduce the number of days adult patients, including those with COVID-19, require mechanical ventilation, according to a press release from Liberate Medical.
In comparison with mechanical ventilation, which is invasive and commonly weakens the breathing muscles, the VentFree system uses noninvasive neuromuscular electrical stimulation to contract the abdominal wall muscles in synchrony with exhalation during mechanical ventilation, according to the press release. This allows patients to begin treatment during the early stages of ventilation while they are sedated and to continue until they are weaned off of ventilation.
A pair of pilot randomized, controlled studies, completed in Europe and Australia, showed that VentFree helped to reduce ventilation duration and ICU length of stay, compared with placebo stimulation. The FDA granted VentFree Breakthrough Device status in 2019.
“We are grateful to the FDA for recognizing the potential of VentFree and feel privileged to have the opportunity to help patients on mechanical ventilation during the COVID-19 pandemic,” Angus McLachlan PhD, cofounder and CEO of Liberate Medical, said in the press release.
VentFree has been authorized for use only for the duration of the current COVID-19 emergency, as it has not yet been approved or cleared for usage by primary care providers.
The Food and Drug Administration has issued an Emergency Use Authorization (EUA) for the VentFree Respiratory Muscle Stimulator in order to potentially reduce the number of days adult patients, including those with COVID-19, require mechanical ventilation, according to a press release from Liberate Medical.
In comparison with mechanical ventilation, which is invasive and commonly weakens the breathing muscles, the VentFree system uses noninvasive neuromuscular electrical stimulation to contract the abdominal wall muscles in synchrony with exhalation during mechanical ventilation, according to the press release. This allows patients to begin treatment during the early stages of ventilation while they are sedated and to continue until they are weaned off of ventilation.
A pair of pilot randomized, controlled studies, completed in Europe and Australia, showed that VentFree helped to reduce ventilation duration and ICU length of stay, compared with placebo stimulation. The FDA granted VentFree Breakthrough Device status in 2019.
“We are grateful to the FDA for recognizing the potential of VentFree and feel privileged to have the opportunity to help patients on mechanical ventilation during the COVID-19 pandemic,” Angus McLachlan PhD, cofounder and CEO of Liberate Medical, said in the press release.
VentFree has been authorized for use only for the duration of the current COVID-19 emergency, as it has not yet been approved or cleared for usage by primary care providers.
The Food and Drug Administration has issued an Emergency Use Authorization (EUA) for the VentFree Respiratory Muscle Stimulator in order to potentially reduce the number of days adult patients, including those with COVID-19, require mechanical ventilation, according to a press release from Liberate Medical.
In comparison with mechanical ventilation, which is invasive and commonly weakens the breathing muscles, the VentFree system uses noninvasive neuromuscular electrical stimulation to contract the abdominal wall muscles in synchrony with exhalation during mechanical ventilation, according to the press release. This allows patients to begin treatment during the early stages of ventilation while they are sedated and to continue until they are weaned off of ventilation.
A pair of pilot randomized, controlled studies, completed in Europe and Australia, showed that VentFree helped to reduce ventilation duration and ICU length of stay, compared with placebo stimulation. The FDA granted VentFree Breakthrough Device status in 2019.
“We are grateful to the FDA for recognizing the potential of VentFree and feel privileged to have the opportunity to help patients on mechanical ventilation during the COVID-19 pandemic,” Angus McLachlan PhD, cofounder and CEO of Liberate Medical, said in the press release.
VentFree has been authorized for use only for the duration of the current COVID-19 emergency, as it has not yet been approved or cleared for usage by primary care providers.
COVID-19 death rate was twice as high in cancer patients in NYC study
COVID-19 patients with cancer had double the fatality rate of COVID-19 patients without cancer treated in an urban New York hospital system, according to data from a retrospective study.
with COVID-19 treated during the same time period in the same hospital system.
Vikas Mehta, MD, of Montefiore Medical Center, New York, and colleagues reported these results in Cancer Discovery.
“As New York has emerged as the current epicenter of the pandemic, we sought to investigate the risk posed by COVID-19 to our cancer population,” the authors wrote.
They identified 218 cancer patients treated for COVID-19 in the Montefiore Health System between March 18 and April 8, 2020. Three-quarters of patients had solid tumors, and 25% had hematologic malignancies. Most patients were adults (98.6%), their median age was 69 years (range, 10-92 years), and 58% were men.
In all, 28% of the cancer patients (61/218) died from COVID-19, including 25% (41/164) of those with solid tumors and 37% (20/54) of those with hematologic malignancies.
Deaths by cancer type
Among the 164 patients with solid tumors, case fatality rates were as follows:
- Pancreatic – 67% (2/3)
- Lung – 55% (6/11)
- Colorectal – 38% (8/21)
- Upper gastrointestinal – 38% (3/8)
- Gynecologic – 38% (5/13)
- Skin – 33% (1/3)
- Hepatobiliary – 29% (2/7)
- Bone/soft tissue – 20% (1/5)
- Genitourinary – 15% (7/46)
- Breast – 14% (4/28)
- Neurologic – 13% (1/8)
- Head and neck – 13% (1/8).
None of the three patients with neuroendocrine tumors died.
Among the 54 patients with hematologic malignancies, case fatality rates were as follows:
- Chronic myeloid leukemia – 100% (1/1)
- Hodgkin lymphoma – 60% (3/5)
- Myelodysplastic syndromes – 60% (3/5)
- Multiple myeloma – 38% (5/13)
- Non-Hodgkin lymphoma – 33% (5/15)
- Chronic lymphocytic leukemia – 33% (1/3)
- Myeloproliferative neoplasms – 29% (2/7).
None of the four patients with acute lymphoblastic leukemia died, and there was one patient with acute myeloid leukemia who did not die.
Factors associated with increased mortality
The researchers compared the 218 cancer patients with COVID-19 with 1,090 age- and sex-matched noncancer patients with COVID-19 treated in the Montefiore Health System between March 18 and April 8, 2020.
Case fatality rates in cancer patients with COVID-19 were significantly increased in all age groups, but older age was associated with higher mortality.
“We observed case fatality rates were elevated in all age cohorts in cancer patients and achieved statistical significance in the age groups 45-64 and in patients older than 75 years of age,” the authors reported.
Other factors significantly associated with higher mortality in a multivariable analysis included the presence of multiple comorbidities; the need for ICU support; and increased levels of d-dimer, lactate, and lactate dehydrogenase.
Additional factors, such as socioeconomic and health disparities, may also be significant predictors of mortality, according to the authors. They noted that this cohort largely consisted of patients from a socioeconomically underprivileged community where mortality because of COVID-19 is reportedly higher.
Proactive strategies moving forward
“We have been addressing the significant burden of the COVID-19 pandemic on our vulnerable cancer patients through a variety of ways,” said study author Balazs Halmos, MD, of Montefiore Medical Center.
The center set up a separate infusion unit exclusively for COVID-positive patients and established separate inpatient areas. Dr. Halmos and colleagues are also providing telemedicine, virtual supportive care services, telephonic counseling, and bilingual peer-support programs.
“Many questions remain as we continue to establish new practices for our cancer patients,” Dr. Halmos said. “We will find answers to these questions as we continue to focus on adaptation and not acceptance in response to the COVID crisis. Our patients deserve nothing less.”
The Albert Einstein Cancer Center supported this study. The authors reported having no conflicts of interest.
SOURCE: Mehta V et al. Cancer Discov. 2020 May 1. doi: 10.1158/2159-8290.CD-20-0516.
COVID-19 patients with cancer had double the fatality rate of COVID-19 patients without cancer treated in an urban New York hospital system, according to data from a retrospective study.
with COVID-19 treated during the same time period in the same hospital system.
Vikas Mehta, MD, of Montefiore Medical Center, New York, and colleagues reported these results in Cancer Discovery.
“As New York has emerged as the current epicenter of the pandemic, we sought to investigate the risk posed by COVID-19 to our cancer population,” the authors wrote.
They identified 218 cancer patients treated for COVID-19 in the Montefiore Health System between March 18 and April 8, 2020. Three-quarters of patients had solid tumors, and 25% had hematologic malignancies. Most patients were adults (98.6%), their median age was 69 years (range, 10-92 years), and 58% were men.
In all, 28% of the cancer patients (61/218) died from COVID-19, including 25% (41/164) of those with solid tumors and 37% (20/54) of those with hematologic malignancies.
Deaths by cancer type
Among the 164 patients with solid tumors, case fatality rates were as follows:
- Pancreatic – 67% (2/3)
- Lung – 55% (6/11)
- Colorectal – 38% (8/21)
- Upper gastrointestinal – 38% (3/8)
- Gynecologic – 38% (5/13)
- Skin – 33% (1/3)
- Hepatobiliary – 29% (2/7)
- Bone/soft tissue – 20% (1/5)
- Genitourinary – 15% (7/46)
- Breast – 14% (4/28)
- Neurologic – 13% (1/8)
- Head and neck – 13% (1/8).
None of the three patients with neuroendocrine tumors died.
Among the 54 patients with hematologic malignancies, case fatality rates were as follows:
- Chronic myeloid leukemia – 100% (1/1)
- Hodgkin lymphoma – 60% (3/5)
- Myelodysplastic syndromes – 60% (3/5)
- Multiple myeloma – 38% (5/13)
- Non-Hodgkin lymphoma – 33% (5/15)
- Chronic lymphocytic leukemia – 33% (1/3)
- Myeloproliferative neoplasms – 29% (2/7).
None of the four patients with acute lymphoblastic leukemia died, and there was one patient with acute myeloid leukemia who did not die.
Factors associated with increased mortality
The researchers compared the 218 cancer patients with COVID-19 with 1,090 age- and sex-matched noncancer patients with COVID-19 treated in the Montefiore Health System between March 18 and April 8, 2020.
Case fatality rates in cancer patients with COVID-19 were significantly increased in all age groups, but older age was associated with higher mortality.
“We observed case fatality rates were elevated in all age cohorts in cancer patients and achieved statistical significance in the age groups 45-64 and in patients older than 75 years of age,” the authors reported.
Other factors significantly associated with higher mortality in a multivariable analysis included the presence of multiple comorbidities; the need for ICU support; and increased levels of d-dimer, lactate, and lactate dehydrogenase.
Additional factors, such as socioeconomic and health disparities, may also be significant predictors of mortality, according to the authors. They noted that this cohort largely consisted of patients from a socioeconomically underprivileged community where mortality because of COVID-19 is reportedly higher.
Proactive strategies moving forward
“We have been addressing the significant burden of the COVID-19 pandemic on our vulnerable cancer patients through a variety of ways,” said study author Balazs Halmos, MD, of Montefiore Medical Center.
The center set up a separate infusion unit exclusively for COVID-positive patients and established separate inpatient areas. Dr. Halmos and colleagues are also providing telemedicine, virtual supportive care services, telephonic counseling, and bilingual peer-support programs.
“Many questions remain as we continue to establish new practices for our cancer patients,” Dr. Halmos said. “We will find answers to these questions as we continue to focus on adaptation and not acceptance in response to the COVID crisis. Our patients deserve nothing less.”
The Albert Einstein Cancer Center supported this study. The authors reported having no conflicts of interest.
SOURCE: Mehta V et al. Cancer Discov. 2020 May 1. doi: 10.1158/2159-8290.CD-20-0516.
COVID-19 patients with cancer had double the fatality rate of COVID-19 patients without cancer treated in an urban New York hospital system, according to data from a retrospective study.
with COVID-19 treated during the same time period in the same hospital system.
Vikas Mehta, MD, of Montefiore Medical Center, New York, and colleagues reported these results in Cancer Discovery.
“As New York has emerged as the current epicenter of the pandemic, we sought to investigate the risk posed by COVID-19 to our cancer population,” the authors wrote.
They identified 218 cancer patients treated for COVID-19 in the Montefiore Health System between March 18 and April 8, 2020. Three-quarters of patients had solid tumors, and 25% had hematologic malignancies. Most patients were adults (98.6%), their median age was 69 years (range, 10-92 years), and 58% were men.
In all, 28% of the cancer patients (61/218) died from COVID-19, including 25% (41/164) of those with solid tumors and 37% (20/54) of those with hematologic malignancies.
Deaths by cancer type
Among the 164 patients with solid tumors, case fatality rates were as follows:
- Pancreatic – 67% (2/3)
- Lung – 55% (6/11)
- Colorectal – 38% (8/21)
- Upper gastrointestinal – 38% (3/8)
- Gynecologic – 38% (5/13)
- Skin – 33% (1/3)
- Hepatobiliary – 29% (2/7)
- Bone/soft tissue – 20% (1/5)
- Genitourinary – 15% (7/46)
- Breast – 14% (4/28)
- Neurologic – 13% (1/8)
- Head and neck – 13% (1/8).
None of the three patients with neuroendocrine tumors died.
Among the 54 patients with hematologic malignancies, case fatality rates were as follows:
- Chronic myeloid leukemia – 100% (1/1)
- Hodgkin lymphoma – 60% (3/5)
- Myelodysplastic syndromes – 60% (3/5)
- Multiple myeloma – 38% (5/13)
- Non-Hodgkin lymphoma – 33% (5/15)
- Chronic lymphocytic leukemia – 33% (1/3)
- Myeloproliferative neoplasms – 29% (2/7).
None of the four patients with acute lymphoblastic leukemia died, and there was one patient with acute myeloid leukemia who did not die.
Factors associated with increased mortality
The researchers compared the 218 cancer patients with COVID-19 with 1,090 age- and sex-matched noncancer patients with COVID-19 treated in the Montefiore Health System between March 18 and April 8, 2020.
Case fatality rates in cancer patients with COVID-19 were significantly increased in all age groups, but older age was associated with higher mortality.
“We observed case fatality rates were elevated in all age cohorts in cancer patients and achieved statistical significance in the age groups 45-64 and in patients older than 75 years of age,” the authors reported.
Other factors significantly associated with higher mortality in a multivariable analysis included the presence of multiple comorbidities; the need for ICU support; and increased levels of d-dimer, lactate, and lactate dehydrogenase.
Additional factors, such as socioeconomic and health disparities, may also be significant predictors of mortality, according to the authors. They noted that this cohort largely consisted of patients from a socioeconomically underprivileged community where mortality because of COVID-19 is reportedly higher.
Proactive strategies moving forward
“We have been addressing the significant burden of the COVID-19 pandemic on our vulnerable cancer patients through a variety of ways,” said study author Balazs Halmos, MD, of Montefiore Medical Center.
The center set up a separate infusion unit exclusively for COVID-positive patients and established separate inpatient areas. Dr. Halmos and colleagues are also providing telemedicine, virtual supportive care services, telephonic counseling, and bilingual peer-support programs.
“Many questions remain as we continue to establish new practices for our cancer patients,” Dr. Halmos said. “We will find answers to these questions as we continue to focus on adaptation and not acceptance in response to the COVID crisis. Our patients deserve nothing less.”
The Albert Einstein Cancer Center supported this study. The authors reported having no conflicts of interest.
SOURCE: Mehta V et al. Cancer Discov. 2020 May 1. doi: 10.1158/2159-8290.CD-20-0516.
FROM CANCER DISCOVERY
Fountains of Wayne, and a hospitalist’s first day, remembered
Like many in the health care field, I have found it hard to watch the news over these past couple of months when it seems that almost every story is about COVID-19 or its repercussions. Luckily, I have two young daughters who “encourage” me to listen to the Frozen 2 soundtrack instead of putting on the evening news when I get home from work. Still, news manages to seep through my defenses. As I scrolled through some headlines recently, I learned of the death of musician Adam Schlesinger from COVID-19. He wasn’t a household name, but his death still hit me in unexpected ways.
I started internship in late June 2005, in a city (Portland, Ore.) about as different from my previous home (Dallas) as any two places can possibly be. I think the day before internship started still ranks as the most nervous of my life. I’m not sure how I slept at all that night, but somehow I did and arrived at the Portland Veterans Affairs Hospital the following morning to start my new career.
And then … nothing happened. Early on that first day, the electronic medical records crashed, and no patients were admitted during our time on “short call.” My upper level resident took care of the one or two established patients on the team (both discharged), so I ended the day with records that would not be broken during the remainder of my residency: 0 notes written, 0 patients seen. Perhaps the most successful first day that any intern, anywhere has ever had, although it prepared me quite poorly for all the subsequent days.
Since I had some time on my hands, I made the 20-minute walk to one of my new hometown’s record stores where Fountains of Wayne (FOW) was playing an acoustic in-store set. Their album from a few years prior, “Welcome Interstate Managers,” was in heavy rotation when I made the drive from Dallas to Portland. It was (and is) a great album for long drives – melodic, catchy, and (mostly) up-tempo. Adam and the band’s singer, Chris Collingwood, played several songs that night on the store’s stage. Then they headed out to the next city, and I headed back home and on to many far-busier days of residency.
We would cross paths again a decade later. I moved back to Texas and became a hospitalist. It turns out that, if you have enough hospitalists of a certain age and if enough of those hospitalists have unearned confidence in their musical ability, then a covers band will undoubtedly be formed. And so, it happened here in San Antonio. We were not selective in our song choices – we played songs from every decade of the last 50 years, bands as popular as the Beatles and as indie as the Rentals. And we played some FOW.
Our band (which will go nameless here so that our YouTube recordings are more difficult to find) played a grand total of one gig during our years of intermittent practicing. That one gig was my wedding rehearsal dinner and the penultimate song we played was “Stacy’s Mom,” which is notable for being both FOW’s biggest hit and a completely inappropriate song to play at a wedding rehearsal dinner. The crowd was probably around the same size as the one that had seen Adam and Chris play in Portland 10 years prior. I don’t think the applause we received was quite as genuine or deserved, though.
After Adam and Chris played their gig, there was an autograph session and I took home a signed poster. Last year, I decided to take it out of storage and hang it in my office. The date of the show and the first day of my physician career, a date now nearly 15 years ago, is written in psychedelic typography at the bottom. The store that I went to that day is no longer there, a victim of progress like so many other record stores across the country. Another location of the same store is still open in Portland. I hope that it and all the other small book and music stores across the country can survive this current crisis, but I know that many will not.
So, here’s to you Adam, and to all the others who have lost their lives to this terrible illness. As a small token of remembrance, I’ll be playing some Fountains of Wayne on the drive home tonight. It’s not quite the same as playing it on a cross-country drive, but hopefully, we will all be able to do that again soon.
Dr. Sehgal is a clinical associate professor of medicine in the division of general and hospital medicine at the South Texas Veterans Health Care System and UT-Health San Antonio. He is a member of the editorial advisory board for The Hospitalist.
Like many in the health care field, I have found it hard to watch the news over these past couple of months when it seems that almost every story is about COVID-19 or its repercussions. Luckily, I have two young daughters who “encourage” me to listen to the Frozen 2 soundtrack instead of putting on the evening news when I get home from work. Still, news manages to seep through my defenses. As I scrolled through some headlines recently, I learned of the death of musician Adam Schlesinger from COVID-19. He wasn’t a household name, but his death still hit me in unexpected ways.
I started internship in late June 2005, in a city (Portland, Ore.) about as different from my previous home (Dallas) as any two places can possibly be. I think the day before internship started still ranks as the most nervous of my life. I’m not sure how I slept at all that night, but somehow I did and arrived at the Portland Veterans Affairs Hospital the following morning to start my new career.
And then … nothing happened. Early on that first day, the electronic medical records crashed, and no patients were admitted during our time on “short call.” My upper level resident took care of the one or two established patients on the team (both discharged), so I ended the day with records that would not be broken during the remainder of my residency: 0 notes written, 0 patients seen. Perhaps the most successful first day that any intern, anywhere has ever had, although it prepared me quite poorly for all the subsequent days.
Since I had some time on my hands, I made the 20-minute walk to one of my new hometown’s record stores where Fountains of Wayne (FOW) was playing an acoustic in-store set. Their album from a few years prior, “Welcome Interstate Managers,” was in heavy rotation when I made the drive from Dallas to Portland. It was (and is) a great album for long drives – melodic, catchy, and (mostly) up-tempo. Adam and the band’s singer, Chris Collingwood, played several songs that night on the store’s stage. Then they headed out to the next city, and I headed back home and on to many far-busier days of residency.
We would cross paths again a decade later. I moved back to Texas and became a hospitalist. It turns out that, if you have enough hospitalists of a certain age and if enough of those hospitalists have unearned confidence in their musical ability, then a covers band will undoubtedly be formed. And so, it happened here in San Antonio. We were not selective in our song choices – we played songs from every decade of the last 50 years, bands as popular as the Beatles and as indie as the Rentals. And we played some FOW.
Our band (which will go nameless here so that our YouTube recordings are more difficult to find) played a grand total of one gig during our years of intermittent practicing. That one gig was my wedding rehearsal dinner and the penultimate song we played was “Stacy’s Mom,” which is notable for being both FOW’s biggest hit and a completely inappropriate song to play at a wedding rehearsal dinner. The crowd was probably around the same size as the one that had seen Adam and Chris play in Portland 10 years prior. I don’t think the applause we received was quite as genuine or deserved, though.
After Adam and Chris played their gig, there was an autograph session and I took home a signed poster. Last year, I decided to take it out of storage and hang it in my office. The date of the show and the first day of my physician career, a date now nearly 15 years ago, is written in psychedelic typography at the bottom. The store that I went to that day is no longer there, a victim of progress like so many other record stores across the country. Another location of the same store is still open in Portland. I hope that it and all the other small book and music stores across the country can survive this current crisis, but I know that many will not.
So, here’s to you Adam, and to all the others who have lost their lives to this terrible illness. As a small token of remembrance, I’ll be playing some Fountains of Wayne on the drive home tonight. It’s not quite the same as playing it on a cross-country drive, but hopefully, we will all be able to do that again soon.
Dr. Sehgal is a clinical associate professor of medicine in the division of general and hospital medicine at the South Texas Veterans Health Care System and UT-Health San Antonio. He is a member of the editorial advisory board for The Hospitalist.
Like many in the health care field, I have found it hard to watch the news over these past couple of months when it seems that almost every story is about COVID-19 or its repercussions. Luckily, I have two young daughters who “encourage” me to listen to the Frozen 2 soundtrack instead of putting on the evening news when I get home from work. Still, news manages to seep through my defenses. As I scrolled through some headlines recently, I learned of the death of musician Adam Schlesinger from COVID-19. He wasn’t a household name, but his death still hit me in unexpected ways.
I started internship in late June 2005, in a city (Portland, Ore.) about as different from my previous home (Dallas) as any two places can possibly be. I think the day before internship started still ranks as the most nervous of my life. I’m not sure how I slept at all that night, but somehow I did and arrived at the Portland Veterans Affairs Hospital the following morning to start my new career.
And then … nothing happened. Early on that first day, the electronic medical records crashed, and no patients were admitted during our time on “short call.” My upper level resident took care of the one or two established patients on the team (both discharged), so I ended the day with records that would not be broken during the remainder of my residency: 0 notes written, 0 patients seen. Perhaps the most successful first day that any intern, anywhere has ever had, although it prepared me quite poorly for all the subsequent days.
Since I had some time on my hands, I made the 20-minute walk to one of my new hometown’s record stores where Fountains of Wayne (FOW) was playing an acoustic in-store set. Their album from a few years prior, “Welcome Interstate Managers,” was in heavy rotation when I made the drive from Dallas to Portland. It was (and is) a great album for long drives – melodic, catchy, and (mostly) up-tempo. Adam and the band’s singer, Chris Collingwood, played several songs that night on the store’s stage. Then they headed out to the next city, and I headed back home and on to many far-busier days of residency.
We would cross paths again a decade later. I moved back to Texas and became a hospitalist. It turns out that, if you have enough hospitalists of a certain age and if enough of those hospitalists have unearned confidence in their musical ability, then a covers band will undoubtedly be formed. And so, it happened here in San Antonio. We were not selective in our song choices – we played songs from every decade of the last 50 years, bands as popular as the Beatles and as indie as the Rentals. And we played some FOW.
Our band (which will go nameless here so that our YouTube recordings are more difficult to find) played a grand total of one gig during our years of intermittent practicing. That one gig was my wedding rehearsal dinner and the penultimate song we played was “Stacy’s Mom,” which is notable for being both FOW’s biggest hit and a completely inappropriate song to play at a wedding rehearsal dinner. The crowd was probably around the same size as the one that had seen Adam and Chris play in Portland 10 years prior. I don’t think the applause we received was quite as genuine or deserved, though.
After Adam and Chris played their gig, there was an autograph session and I took home a signed poster. Last year, I decided to take it out of storage and hang it in my office. The date of the show and the first day of my physician career, a date now nearly 15 years ago, is written in psychedelic typography at the bottom. The store that I went to that day is no longer there, a victim of progress like so many other record stores across the country. Another location of the same store is still open in Portland. I hope that it and all the other small book and music stores across the country can survive this current crisis, but I know that many will not.
So, here’s to you Adam, and to all the others who have lost their lives to this terrible illness. As a small token of remembrance, I’ll be playing some Fountains of Wayne on the drive home tonight. It’s not quite the same as playing it on a cross-country drive, but hopefully, we will all be able to do that again soon.
Dr. Sehgal is a clinical associate professor of medicine in the division of general and hospital medicine at the South Texas Veterans Health Care System and UT-Health San Antonio. He is a member of the editorial advisory board for The Hospitalist.
Teledermatology Fast Facts
Due to the impact of the coronavirus disease 2019 (COVID-19) pandemic, many patients are working from home, which has led to a unique opportunity for dermatologists to step in and continue to care for their patients at home via telemedicine. With recent waivers and guidance from the Centers for Medicare & Medicaid Services (CMS), insurance coverage has been expanded for telehealth services, usually at the same level as an in-person visit. This editorial provides guidance for implementing telehealth services in your practice, and a tip sheet is available online for you to save and print. Please note that this information is changing on a day-to-day basis, so refer to the resources in the Table to get the latest updates.

Billing and Coding
The best reimbursements are for live telemedicine that emulates an outpatient visit and is billed using the same Current Procedural Terminology (CPT) codes (99201–99215). Previously, Medicare did not allow direct-to-patient visits to be billed, instead requiring a waiver for these services to be provided in underserved areas. During the COVID-19 pandemic, this requirement has been lifted, allowing all patients to be seen from any originating site (eg, the patient’s home).
Previously, the CMS had issued guidelines for telehealth visits that required that a physician-patient relationship be established in person prior to conducting telemedicine visits. These guidelines also have been waived for the duration of this public health emergency, allowing physicians to conduct new patient visits via telehealth and bill Medicare. Many commercial payors also are covering new patient visits via telehealth; however, it is best to check the patient’s plan first, as some plans may have different requirements or restrictions on allowable CPT codes and/or place of service. Prior requirements that physicians at a distant site (ie, the physician providing telemedicine services) be located at a site of clinical care also have been relaxed, thus allowing physicians to be located anywhere while providing services, even for those who are confined to their homes.
In general, commercial payors are covering telehealth visits at 100% of an in-person visit. Although COVID-19–related visits are covered by law, many payors including Aetna, Anthem, Blue Cross Blue Shield, Cigna, Emblem Health, Humana, and United Healthcare have indicated that they will waive all telehealth co-pays for a limited time, including visits not related to COVID-19. At the time of publication, only Aetna has issued a formal policy to this effect, so it is best to check with the insurer.1,2 However, it is important to note that regional and employer-specific plans may have different policies, so it is best to check with the insurance plans directly to confirm coverage and co-pay status.
Coding should be performed using the usual new/established patient visit codes for outpatients (99201–99215). A place of service (POS) code of 02 previously was used for all telehealth visits; however, the CMS is allowing offices to bill with their usual POS (generally POS 11) and modifier -95 in an updated rule that is active during this public health crisis. This change allows access to higher reimbursements, as POS 02 visits are paid at lower facility fee rates. Commercial insurers have varying policies on POS that are changing, so it is best to check with them individually.
In certain states, store-and-forward services may be billed using a GQ modifier for Medicaid; however, the remote check-in and telephone codes for Medicare do not reimburse well and generally are best avoided if a live telemedicine encounter is possible, as it provides better patient care and direct counseling capabilities, similar to an in-person visit. The CMS has indicated that it is now covering telephone visits (99441-99443) so that providers can contact patients through an audio-only device and bill for the encounter. Generally speaking, telephone visits reimburse the same or more than the virtual check-in codes (G2010/G2012) as long as the telephone encounter is more than 5-minutes long. Digital visits also are available (99421-99423), which include both store-and-forward photographs and a telephone call, but the reimbursements are similar to the telephone-only visit codes.3
Although the CMS has relaxed regulations for physicians to provide care across state lines, not all state licensing authorities have adopted similar measures, and the CMS waiver only applies to federally funded programs. It is important to check with state medical licensing authorities to see whether you are authorized to provide care if your patient is not located within the state where you hold your license at the time of the visit. Many states, but not all, have waived this requirement or have set up very expedient ways to apply for telemedicine licenses.
The CMS also released guidance that rules for documentation requirements have been temporarily relaxed,3 such that visits should be billed at a level of service consistent with either medical decision-making or total time spent by the provider, including face-to-face and non–face-to-face time spent on the patient. (Note: If billing by time, which usually is not advised, use the CMS definitions of time-based coding.) History and physical examination criteria do not have to be met.
Workflow
In general, it is best to maintain your current workflow as much as possible, with a live video encounter replacing only the patient interaction portion of the visit. You will need to maintain an infrastructure for scheduling visits, collecting co-pays (eg, over the telephone prior to the video visit), and documentation/billing.
It is best to have one device for conducting the actual video visit (eg, a laptop, tablet, or smartphone) and a separate device to use for documentation (eg, another device to access the electronic medical record). The CMS has advised that it will not enforce Health Insurance Portability and Accountability Act (HIPAA) rules,4 allowing physicians to use video conferencing and chat applications such as FaceTime, Skype, or Google Hangouts; however, patient safety is still an issue, and it is imperative to make sure you identify the patient correctly upon starting the visit. During the COVID-19 pandemic, numerous telehealth companies are offering temporary free video conferencing software that is HIPAA compliant, such as Doximity, VSee, Doxy.me, and Medweb. If you are able to go through one of these vendors, you will be able to continue conducting some telemedicine visits after the public health emergency, which may be helpful to your practice.
For some visits, such as acne patients on isotretinoin, you can write for a standing laboratory order that can be drawn at a laboratory center near your patient, and you can perform the counseling via telemedicine. For patients on isotretinoin, iPledge has issued a program update allowing the use of at-home pregnancy tests during the pandemic. The results must be communicated to the provider and documented with a time/date.5
Video Visit Tips and Pearls
Make sure to have well-defined parameters about what can be triaged via a single video visit. Suggestions include no total-body skin examinations and a limit of 1 rash or 2 lesions. Provide a disclaimer that it is not always possible to tell whether or not a lesion is concerning via a video visit, and the patient may have to come in for a biopsy at some point.
It is better to overcall via telemedicine than to undercall. Unless something is a very obvious seborrheic keratosis, skin tag, cherry angioma, or other benign lesion, it might be reasonable to tell a patient to come in for further evaluation of a worrisome lesion after things get back to normal. A static photograph from the patient can be helpful so it is clear what lesion is being examined during the current visit. If the patient has a skin cancer at a distant site in the future, there will be no doubt as to what lesion you examined. Having the capability to receive static images from the patient to serve as representative photographs of their chief concern is very helpful before the visit. Often, these images turn out to be better diagnostically than the live video itself, which can be compressed and show inaccurate colors. Some of the telemedicine vendors have this feature built-in, which is preferable. If you are asking patients to send you emails, it is better to have access to a HIPAA-compliant email inbox to avoid any potential issues down the line.
When scheduling a video visit, have your schedulers specifically tell patients that they should be on a high-speed Wi-Fi connection with good lighting in the room. You would be surprised that this is not intuitive for everyone!
Finally, most telemedicine visits are relatively short and to the point. In the beginning, start by scheduling patients every 15 to 20 minutes to allow for technical difficulties, but ultimately plan to be seeing patients at least every 10 minutes—it can be quite efficient!
- America’s Health Insurance Providers. Health insurance providers respond to coronavirus (COVID-19). https://www.ahip.org/health-insurance-providers-respond-to-coronavirus-covid-19/. Published April 22, 2020. Accessed April 23, 2020.
- Private payer coverage during COVID-19. American College of Physicians website. https://www.acponline.org/system/files/documents/clinical_information/resources/covid19/payer_chart_covid-19.pdf. Updated April 22, 2020. Accessed April 23, 2020.
- Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; policy and regulatory revisions in response to the COVID-19 public health emergency. https://www.cms.gov/files/document/covid-final-ifc.pdf. Published March 26, 2020. Accessed April 23, 2020.
- Notification of enforcement discretion for telehealth remote communications during the COVID-19 nationwide public health emergency. US Department of Health and Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/emergency-preparedness/notification-enforcement-discretion-telehealth/index.html. Updated March 30, 2020. Accessed April 23, 2020.
- Program update. iPledge website. https://www.ipledgeprogram.com/iPledgeUI/home.u. Accessed April 23, 2020.
Due to the impact of the coronavirus disease 2019 (COVID-19) pandemic, many patients are working from home, which has led to a unique opportunity for dermatologists to step in and continue to care for their patients at home via telemedicine. With recent waivers and guidance from the Centers for Medicare & Medicaid Services (CMS), insurance coverage has been expanded for telehealth services, usually at the same level as an in-person visit. This editorial provides guidance for implementing telehealth services in your practice, and a tip sheet is available online for you to save and print. Please note that this information is changing on a day-to-day basis, so refer to the resources in the Table to get the latest updates.

Billing and Coding
The best reimbursements are for live telemedicine that emulates an outpatient visit and is billed using the same Current Procedural Terminology (CPT) codes (99201–99215). Previously, Medicare did not allow direct-to-patient visits to be billed, instead requiring a waiver for these services to be provided in underserved areas. During the COVID-19 pandemic, this requirement has been lifted, allowing all patients to be seen from any originating site (eg, the patient’s home).
Previously, the CMS had issued guidelines for telehealth visits that required that a physician-patient relationship be established in person prior to conducting telemedicine visits. These guidelines also have been waived for the duration of this public health emergency, allowing physicians to conduct new patient visits via telehealth and bill Medicare. Many commercial payors also are covering new patient visits via telehealth; however, it is best to check the patient’s plan first, as some plans may have different requirements or restrictions on allowable CPT codes and/or place of service. Prior requirements that physicians at a distant site (ie, the physician providing telemedicine services) be located at a site of clinical care also have been relaxed, thus allowing physicians to be located anywhere while providing services, even for those who are confined to their homes.
In general, commercial payors are covering telehealth visits at 100% of an in-person visit. Although COVID-19–related visits are covered by law, many payors including Aetna, Anthem, Blue Cross Blue Shield, Cigna, Emblem Health, Humana, and United Healthcare have indicated that they will waive all telehealth co-pays for a limited time, including visits not related to COVID-19. At the time of publication, only Aetna has issued a formal policy to this effect, so it is best to check with the insurer.1,2 However, it is important to note that regional and employer-specific plans may have different policies, so it is best to check with the insurance plans directly to confirm coverage and co-pay status.
Coding should be performed using the usual new/established patient visit codes for outpatients (99201–99215). A place of service (POS) code of 02 previously was used for all telehealth visits; however, the CMS is allowing offices to bill with their usual POS (generally POS 11) and modifier -95 in an updated rule that is active during this public health crisis. This change allows access to higher reimbursements, as POS 02 visits are paid at lower facility fee rates. Commercial insurers have varying policies on POS that are changing, so it is best to check with them individually.
In certain states, store-and-forward services may be billed using a GQ modifier for Medicaid; however, the remote check-in and telephone codes for Medicare do not reimburse well and generally are best avoided if a live telemedicine encounter is possible, as it provides better patient care and direct counseling capabilities, similar to an in-person visit. The CMS has indicated that it is now covering telephone visits (99441-99443) so that providers can contact patients through an audio-only device and bill for the encounter. Generally speaking, telephone visits reimburse the same or more than the virtual check-in codes (G2010/G2012) as long as the telephone encounter is more than 5-minutes long. Digital visits also are available (99421-99423), which include both store-and-forward photographs and a telephone call, but the reimbursements are similar to the telephone-only visit codes.3
Although the CMS has relaxed regulations for physicians to provide care across state lines, not all state licensing authorities have adopted similar measures, and the CMS waiver only applies to federally funded programs. It is important to check with state medical licensing authorities to see whether you are authorized to provide care if your patient is not located within the state where you hold your license at the time of the visit. Many states, but not all, have waived this requirement or have set up very expedient ways to apply for telemedicine licenses.
The CMS also released guidance that rules for documentation requirements have been temporarily relaxed,3 such that visits should be billed at a level of service consistent with either medical decision-making or total time spent by the provider, including face-to-face and non–face-to-face time spent on the patient. (Note: If billing by time, which usually is not advised, use the CMS definitions of time-based coding.) History and physical examination criteria do not have to be met.
Workflow
In general, it is best to maintain your current workflow as much as possible, with a live video encounter replacing only the patient interaction portion of the visit. You will need to maintain an infrastructure for scheduling visits, collecting co-pays (eg, over the telephone prior to the video visit), and documentation/billing.
It is best to have one device for conducting the actual video visit (eg, a laptop, tablet, or smartphone) and a separate device to use for documentation (eg, another device to access the electronic medical record). The CMS has advised that it will not enforce Health Insurance Portability and Accountability Act (HIPAA) rules,4 allowing physicians to use video conferencing and chat applications such as FaceTime, Skype, or Google Hangouts; however, patient safety is still an issue, and it is imperative to make sure you identify the patient correctly upon starting the visit. During the COVID-19 pandemic, numerous telehealth companies are offering temporary free video conferencing software that is HIPAA compliant, such as Doximity, VSee, Doxy.me, and Medweb. If you are able to go through one of these vendors, you will be able to continue conducting some telemedicine visits after the public health emergency, which may be helpful to your practice.
For some visits, such as acne patients on isotretinoin, you can write for a standing laboratory order that can be drawn at a laboratory center near your patient, and you can perform the counseling via telemedicine. For patients on isotretinoin, iPledge has issued a program update allowing the use of at-home pregnancy tests during the pandemic. The results must be communicated to the provider and documented with a time/date.5
Video Visit Tips and Pearls
Make sure to have well-defined parameters about what can be triaged via a single video visit. Suggestions include no total-body skin examinations and a limit of 1 rash or 2 lesions. Provide a disclaimer that it is not always possible to tell whether or not a lesion is concerning via a video visit, and the patient may have to come in for a biopsy at some point.
It is better to overcall via telemedicine than to undercall. Unless something is a very obvious seborrheic keratosis, skin tag, cherry angioma, or other benign lesion, it might be reasonable to tell a patient to come in for further evaluation of a worrisome lesion after things get back to normal. A static photograph from the patient can be helpful so it is clear what lesion is being examined during the current visit. If the patient has a skin cancer at a distant site in the future, there will be no doubt as to what lesion you examined. Having the capability to receive static images from the patient to serve as representative photographs of their chief concern is very helpful before the visit. Often, these images turn out to be better diagnostically than the live video itself, which can be compressed and show inaccurate colors. Some of the telemedicine vendors have this feature built-in, which is preferable. If you are asking patients to send you emails, it is better to have access to a HIPAA-compliant email inbox to avoid any potential issues down the line.
When scheduling a video visit, have your schedulers specifically tell patients that they should be on a high-speed Wi-Fi connection with good lighting in the room. You would be surprised that this is not intuitive for everyone!
Finally, most telemedicine visits are relatively short and to the point. In the beginning, start by scheduling patients every 15 to 20 minutes to allow for technical difficulties, but ultimately plan to be seeing patients at least every 10 minutes—it can be quite efficient!
Due to the impact of the coronavirus disease 2019 (COVID-19) pandemic, many patients are working from home, which has led to a unique opportunity for dermatologists to step in and continue to care for their patients at home via telemedicine. With recent waivers and guidance from the Centers for Medicare & Medicaid Services (CMS), insurance coverage has been expanded for telehealth services, usually at the same level as an in-person visit. This editorial provides guidance for implementing telehealth services in your practice, and a tip sheet is available online for you to save and print. Please note that this information is changing on a day-to-day basis, so refer to the resources in the Table to get the latest updates.

Billing and Coding
The best reimbursements are for live telemedicine that emulates an outpatient visit and is billed using the same Current Procedural Terminology (CPT) codes (99201–99215). Previously, Medicare did not allow direct-to-patient visits to be billed, instead requiring a waiver for these services to be provided in underserved areas. During the COVID-19 pandemic, this requirement has been lifted, allowing all patients to be seen from any originating site (eg, the patient’s home).
Previously, the CMS had issued guidelines for telehealth visits that required that a physician-patient relationship be established in person prior to conducting telemedicine visits. These guidelines also have been waived for the duration of this public health emergency, allowing physicians to conduct new patient visits via telehealth and bill Medicare. Many commercial payors also are covering new patient visits via telehealth; however, it is best to check the patient’s plan first, as some plans may have different requirements or restrictions on allowable CPT codes and/or place of service. Prior requirements that physicians at a distant site (ie, the physician providing telemedicine services) be located at a site of clinical care also have been relaxed, thus allowing physicians to be located anywhere while providing services, even for those who are confined to their homes.
In general, commercial payors are covering telehealth visits at 100% of an in-person visit. Although COVID-19–related visits are covered by law, many payors including Aetna, Anthem, Blue Cross Blue Shield, Cigna, Emblem Health, Humana, and United Healthcare have indicated that they will waive all telehealth co-pays for a limited time, including visits not related to COVID-19. At the time of publication, only Aetna has issued a formal policy to this effect, so it is best to check with the insurer.1,2 However, it is important to note that regional and employer-specific plans may have different policies, so it is best to check with the insurance plans directly to confirm coverage and co-pay status.
Coding should be performed using the usual new/established patient visit codes for outpatients (99201–99215). A place of service (POS) code of 02 previously was used for all telehealth visits; however, the CMS is allowing offices to bill with their usual POS (generally POS 11) and modifier -95 in an updated rule that is active during this public health crisis. This change allows access to higher reimbursements, as POS 02 visits are paid at lower facility fee rates. Commercial insurers have varying policies on POS that are changing, so it is best to check with them individually.
In certain states, store-and-forward services may be billed using a GQ modifier for Medicaid; however, the remote check-in and telephone codes for Medicare do not reimburse well and generally are best avoided if a live telemedicine encounter is possible, as it provides better patient care and direct counseling capabilities, similar to an in-person visit. The CMS has indicated that it is now covering telephone visits (99441-99443) so that providers can contact patients through an audio-only device and bill for the encounter. Generally speaking, telephone visits reimburse the same or more than the virtual check-in codes (G2010/G2012) as long as the telephone encounter is more than 5-minutes long. Digital visits also are available (99421-99423), which include both store-and-forward photographs and a telephone call, but the reimbursements are similar to the telephone-only visit codes.3
Although the CMS has relaxed regulations for physicians to provide care across state lines, not all state licensing authorities have adopted similar measures, and the CMS waiver only applies to federally funded programs. It is important to check with state medical licensing authorities to see whether you are authorized to provide care if your patient is not located within the state where you hold your license at the time of the visit. Many states, but not all, have waived this requirement or have set up very expedient ways to apply for telemedicine licenses.
The CMS also released guidance that rules for documentation requirements have been temporarily relaxed,3 such that visits should be billed at a level of service consistent with either medical decision-making or total time spent by the provider, including face-to-face and non–face-to-face time spent on the patient. (Note: If billing by time, which usually is not advised, use the CMS definitions of time-based coding.) History and physical examination criteria do not have to be met.
Workflow
In general, it is best to maintain your current workflow as much as possible, with a live video encounter replacing only the patient interaction portion of the visit. You will need to maintain an infrastructure for scheduling visits, collecting co-pays (eg, over the telephone prior to the video visit), and documentation/billing.
It is best to have one device for conducting the actual video visit (eg, a laptop, tablet, or smartphone) and a separate device to use for documentation (eg, another device to access the electronic medical record). The CMS has advised that it will not enforce Health Insurance Portability and Accountability Act (HIPAA) rules,4 allowing physicians to use video conferencing and chat applications such as FaceTime, Skype, or Google Hangouts; however, patient safety is still an issue, and it is imperative to make sure you identify the patient correctly upon starting the visit. During the COVID-19 pandemic, numerous telehealth companies are offering temporary free video conferencing software that is HIPAA compliant, such as Doximity, VSee, Doxy.me, and Medweb. If you are able to go through one of these vendors, you will be able to continue conducting some telemedicine visits after the public health emergency, which may be helpful to your practice.
For some visits, such as acne patients on isotretinoin, you can write for a standing laboratory order that can be drawn at a laboratory center near your patient, and you can perform the counseling via telemedicine. For patients on isotretinoin, iPledge has issued a program update allowing the use of at-home pregnancy tests during the pandemic. The results must be communicated to the provider and documented with a time/date.5
Video Visit Tips and Pearls
Make sure to have well-defined parameters about what can be triaged via a single video visit. Suggestions include no total-body skin examinations and a limit of 1 rash or 2 lesions. Provide a disclaimer that it is not always possible to tell whether or not a lesion is concerning via a video visit, and the patient may have to come in for a biopsy at some point.
It is better to overcall via telemedicine than to undercall. Unless something is a very obvious seborrheic keratosis, skin tag, cherry angioma, or other benign lesion, it might be reasonable to tell a patient to come in for further evaluation of a worrisome lesion after things get back to normal. A static photograph from the patient can be helpful so it is clear what lesion is being examined during the current visit. If the patient has a skin cancer at a distant site in the future, there will be no doubt as to what lesion you examined. Having the capability to receive static images from the patient to serve as representative photographs of their chief concern is very helpful before the visit. Often, these images turn out to be better diagnostically than the live video itself, which can be compressed and show inaccurate colors. Some of the telemedicine vendors have this feature built-in, which is preferable. If you are asking patients to send you emails, it is better to have access to a HIPAA-compliant email inbox to avoid any potential issues down the line.
When scheduling a video visit, have your schedulers specifically tell patients that they should be on a high-speed Wi-Fi connection with good lighting in the room. You would be surprised that this is not intuitive for everyone!
Finally, most telemedicine visits are relatively short and to the point. In the beginning, start by scheduling patients every 15 to 20 minutes to allow for technical difficulties, but ultimately plan to be seeing patients at least every 10 minutes—it can be quite efficient!
- America’s Health Insurance Providers. Health insurance providers respond to coronavirus (COVID-19). https://www.ahip.org/health-insurance-providers-respond-to-coronavirus-covid-19/. Published April 22, 2020. Accessed April 23, 2020.
- Private payer coverage during COVID-19. American College of Physicians website. https://www.acponline.org/system/files/documents/clinical_information/resources/covid19/payer_chart_covid-19.pdf. Updated April 22, 2020. Accessed April 23, 2020.
- Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; policy and regulatory revisions in response to the COVID-19 public health emergency. https://www.cms.gov/files/document/covid-final-ifc.pdf. Published March 26, 2020. Accessed April 23, 2020.
- Notification of enforcement discretion for telehealth remote communications during the COVID-19 nationwide public health emergency. US Department of Health and Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/emergency-preparedness/notification-enforcement-discretion-telehealth/index.html. Updated March 30, 2020. Accessed April 23, 2020.
- Program update. iPledge website. https://www.ipledgeprogram.com/iPledgeUI/home.u. Accessed April 23, 2020.
- America’s Health Insurance Providers. Health insurance providers respond to coronavirus (COVID-19). https://www.ahip.org/health-insurance-providers-respond-to-coronavirus-covid-19/. Published April 22, 2020. Accessed April 23, 2020.
- Private payer coverage during COVID-19. American College of Physicians website. https://www.acponline.org/system/files/documents/clinical_information/resources/covid19/payer_chart_covid-19.pdf. Updated April 22, 2020. Accessed April 23, 2020.
- Centers for Medicare & Medicaid Services. Medicare and Medicaid programs; policy and regulatory revisions in response to the COVID-19 public health emergency. https://www.cms.gov/files/document/covid-final-ifc.pdf. Published March 26, 2020. Accessed April 23, 2020.
- Notification of enforcement discretion for telehealth remote communications during the COVID-19 nationwide public health emergency. US Department of Health and Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/emergency-preparedness/notification-enforcement-discretion-telehealth/index.html. Updated March 30, 2020. Accessed April 23, 2020.
- Program update. iPledge website. https://www.ipledgeprogram.com/iPledgeUI/home.u. Accessed April 23, 2020.
Doctor with a mask: Enhancing communication and empathy
Delivering a goodbye monologue to an elderly patient, I said: “Tomorrow, my colleague Dr. XYZ, who is an excellent physician, will be here in my place, and I will leave a detailed sign out for them.” I was on the last day of a 7-day-long block on hospital medicine service. Typically, when I say goodbye, some patients respond “thank you, enjoy your time,” some don’t care, and some show disappointment at the transition. This patient became uneasy, choking back tears, and said: “But, I don’t want a new doctor. You know me well. ... They don’t even allow my family in the hospital.”
That expression of anxiety, of having to build rapport with a new provider, concerns about continuity of care, and missing support of family members were not alien to me. As I instinctively took a step toward him to offer a comforting hug, an unsolicited voice in my head said, “social distancing.” I steered back, handing him a box of tissues. I continued: “You have come a long way, and things are looking good from here,” providing more details before I left the room. There was a change in my practice that week. I didn’t shake hands with my patients; I didn’t sit on any unassigned chair; I had no family members in the room asking me questions or supporting my patients. I was trying to show empathy or a smile behind a mask and protective eyewear. The business card with photograph had become more critical than ever for patients to “see” their doctor.
Moving from room to room and examining patients, it felt like the coronavirus was changing the practice of medicine beyond concerns of virus transmission, losing a patient, or putting in extra hours. I realized I was missing so-called “nonverbal communication” amid social distancing: facial expressions, social touch, and the support of family or friends to motivate or destress patients. With no visitors and curbed health care staff entries into patient’s rooms, social distancing was amounting to social isolation. My protective gear and social distancing seemed to be reducing my perceived empathy with patients, and the ability to build a good patient-physician relationship.
Amid alarms, beeps, and buzzes, patients were not only missing their families but also the familiar faces of their physicians. I needed to raise my game while embracing the “new normal” of health care. Cut to the next 13 patients: I paid more attention to voice, tone, and posture. I called patient families from the bedside instead of the office. I translated my emotions with words, loud and clear, replacing “your renal function looks better” (said without a smile) with “I am happy to see your renal function better.”
Through years of practice, I felt prepared to deal with feelings of denial, grief, anxiety, and much more, but the emotions arising as a result of this pandemic were unique. “I knew my mother was old, and this day would come,” said one of the inconsolable family members of a critically ill patient. “However, I wished to be at her side that day, not like this.” I spend my days listening to patient and family concerns about unemployment with quarantine, fears of spreading the disease to loved ones, and the possibility of medications not working.
After a long day, I went back to that first elderly patient to see if he was comfortable with the transition of care. I did a video conference with his daughter, and repeated my goodbyes. The patient smiled and said: “Doc, you deserve a break.” That day I learned about the challenges of good clinical rounding in coronavirus times, and how to overcome them. For “millennial” physicians, it is our first pandemic, and we are learning from it every day.
Driving home through empty streets, I concluded that my answers to the clinical questions asked by patients and families lean heavily on ever-changing data, and the treatments offered have yet to prove their mettle. As a result, I will continue to focus as much on the time-tested fundamentals of clinical practice: communication and empathy. I cannot allow the social distancing and the mask to hide my compassion, or take away from patient satisfaction. Shifting gears, I turned on my car radio, using music to reset my mind before attending to my now-homeschooling kids.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Wong CK et al. Effect of facemasks on empathy and relational continuity: A randomised controlled trial in primary care. BMC Fam Pract. 2013;14:200.
2. Little P et al. Randomised controlled trial of a brief intervention targeting predominantly nonverbal communication in general practice consultations. Br J Gen Pract. 2015;65(635):e351-6.
3. Varghese A. A doctor’s touch. TEDGlobal 2011. 2011 Jul. https://www.ted.com/talks/abraham_verghese_a_doctor_s_touch?language=en
Delivering a goodbye monologue to an elderly patient, I said: “Tomorrow, my colleague Dr. XYZ, who is an excellent physician, will be here in my place, and I will leave a detailed sign out for them.” I was on the last day of a 7-day-long block on hospital medicine service. Typically, when I say goodbye, some patients respond “thank you, enjoy your time,” some don’t care, and some show disappointment at the transition. This patient became uneasy, choking back tears, and said: “But, I don’t want a new doctor. You know me well. ... They don’t even allow my family in the hospital.”
That expression of anxiety, of having to build rapport with a new provider, concerns about continuity of care, and missing support of family members were not alien to me. As I instinctively took a step toward him to offer a comforting hug, an unsolicited voice in my head said, “social distancing.” I steered back, handing him a box of tissues. I continued: “You have come a long way, and things are looking good from here,” providing more details before I left the room. There was a change in my practice that week. I didn’t shake hands with my patients; I didn’t sit on any unassigned chair; I had no family members in the room asking me questions or supporting my patients. I was trying to show empathy or a smile behind a mask and protective eyewear. The business card with photograph had become more critical than ever for patients to “see” their doctor.
Moving from room to room and examining patients, it felt like the coronavirus was changing the practice of medicine beyond concerns of virus transmission, losing a patient, or putting in extra hours. I realized I was missing so-called “nonverbal communication” amid social distancing: facial expressions, social touch, and the support of family or friends to motivate or destress patients. With no visitors and curbed health care staff entries into patient’s rooms, social distancing was amounting to social isolation. My protective gear and social distancing seemed to be reducing my perceived empathy with patients, and the ability to build a good patient-physician relationship.
Amid alarms, beeps, and buzzes, patients were not only missing their families but also the familiar faces of their physicians. I needed to raise my game while embracing the “new normal” of health care. Cut to the next 13 patients: I paid more attention to voice, tone, and posture. I called patient families from the bedside instead of the office. I translated my emotions with words, loud and clear, replacing “your renal function looks better” (said without a smile) with “I am happy to see your renal function better.”
Through years of practice, I felt prepared to deal with feelings of denial, grief, anxiety, and much more, but the emotions arising as a result of this pandemic were unique. “I knew my mother was old, and this day would come,” said one of the inconsolable family members of a critically ill patient. “However, I wished to be at her side that day, not like this.” I spend my days listening to patient and family concerns about unemployment with quarantine, fears of spreading the disease to loved ones, and the possibility of medications not working.
After a long day, I went back to that first elderly patient to see if he was comfortable with the transition of care. I did a video conference with his daughter, and repeated my goodbyes. The patient smiled and said: “Doc, you deserve a break.” That day I learned about the challenges of good clinical rounding in coronavirus times, and how to overcome them. For “millennial” physicians, it is our first pandemic, and we are learning from it every day.
Driving home through empty streets, I concluded that my answers to the clinical questions asked by patients and families lean heavily on ever-changing data, and the treatments offered have yet to prove their mettle. As a result, I will continue to focus as much on the time-tested fundamentals of clinical practice: communication and empathy. I cannot allow the social distancing and the mask to hide my compassion, or take away from patient satisfaction. Shifting gears, I turned on my car radio, using music to reset my mind before attending to my now-homeschooling kids.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Wong CK et al. Effect of facemasks on empathy and relational continuity: A randomised controlled trial in primary care. BMC Fam Pract. 2013;14:200.
2. Little P et al. Randomised controlled trial of a brief intervention targeting predominantly nonverbal communication in general practice consultations. Br J Gen Pract. 2015;65(635):e351-6.
3. Varghese A. A doctor’s touch. TEDGlobal 2011. 2011 Jul. https://www.ted.com/talks/abraham_verghese_a_doctor_s_touch?language=en
Delivering a goodbye monologue to an elderly patient, I said: “Tomorrow, my colleague Dr. XYZ, who is an excellent physician, will be here in my place, and I will leave a detailed sign out for them.” I was on the last day of a 7-day-long block on hospital medicine service. Typically, when I say goodbye, some patients respond “thank you, enjoy your time,” some don’t care, and some show disappointment at the transition. This patient became uneasy, choking back tears, and said: “But, I don’t want a new doctor. You know me well. ... They don’t even allow my family in the hospital.”
That expression of anxiety, of having to build rapport with a new provider, concerns about continuity of care, and missing support of family members were not alien to me. As I instinctively took a step toward him to offer a comforting hug, an unsolicited voice in my head said, “social distancing.” I steered back, handing him a box of tissues. I continued: “You have come a long way, and things are looking good from here,” providing more details before I left the room. There was a change in my practice that week. I didn’t shake hands with my patients; I didn’t sit on any unassigned chair; I had no family members in the room asking me questions or supporting my patients. I was trying to show empathy or a smile behind a mask and protective eyewear. The business card with photograph had become more critical than ever for patients to “see” their doctor.
Moving from room to room and examining patients, it felt like the coronavirus was changing the practice of medicine beyond concerns of virus transmission, losing a patient, or putting in extra hours. I realized I was missing so-called “nonverbal communication” amid social distancing: facial expressions, social touch, and the support of family or friends to motivate or destress patients. With no visitors and curbed health care staff entries into patient’s rooms, social distancing was amounting to social isolation. My protective gear and social distancing seemed to be reducing my perceived empathy with patients, and the ability to build a good patient-physician relationship.
Amid alarms, beeps, and buzzes, patients were not only missing their families but also the familiar faces of their physicians. I needed to raise my game while embracing the “new normal” of health care. Cut to the next 13 patients: I paid more attention to voice, tone, and posture. I called patient families from the bedside instead of the office. I translated my emotions with words, loud and clear, replacing “your renal function looks better” (said without a smile) with “I am happy to see your renal function better.”
Through years of practice, I felt prepared to deal with feelings of denial, grief, anxiety, and much more, but the emotions arising as a result of this pandemic were unique. “I knew my mother was old, and this day would come,” said one of the inconsolable family members of a critically ill patient. “However, I wished to be at her side that day, not like this.” I spend my days listening to patient and family concerns about unemployment with quarantine, fears of spreading the disease to loved ones, and the possibility of medications not working.
After a long day, I went back to that first elderly patient to see if he was comfortable with the transition of care. I did a video conference with his daughter, and repeated my goodbyes. The patient smiled and said: “Doc, you deserve a break.” That day I learned about the challenges of good clinical rounding in coronavirus times, and how to overcome them. For “millennial” physicians, it is our first pandemic, and we are learning from it every day.
Driving home through empty streets, I concluded that my answers to the clinical questions asked by patients and families lean heavily on ever-changing data, and the treatments offered have yet to prove their mettle. As a result, I will continue to focus as much on the time-tested fundamentals of clinical practice: communication and empathy. I cannot allow the social distancing and the mask to hide my compassion, or take away from patient satisfaction. Shifting gears, I turned on my car radio, using music to reset my mind before attending to my now-homeschooling kids.
Dr. Saigal is a hospitalist and clinical assistant professor of medicine in the division of hospital medicine at the Ohio State University Wexner Medical Center, Columbus.
References
1. Wong CK et al. Effect of facemasks on empathy and relational continuity: A randomised controlled trial in primary care. BMC Fam Pract. 2013;14:200.
2. Little P et al. Randomised controlled trial of a brief intervention targeting predominantly nonverbal communication in general practice consultations. Br J Gen Pract. 2015;65(635):e351-6.
3. Varghese A. A doctor’s touch. TEDGlobal 2011. 2011 Jul. https://www.ted.com/talks/abraham_verghese_a_doctor_s_touch?language=en
Case reports illustrate heterogeneity of skin manifestations in COVID patients
Two infection.
It is not yet clear from these or other case reports which, if any, skin eruptions accompanying COVID-19 infections are caused by the virus, but the authors of the editorial, led by Lauren M. Madigan, MD, of the department of dermatology at the University of Utah, Salt Lake City, urged dermatologists to lead efforts to find out.
“To fully characterize skin manifestations, it may be necessary for dermatologists to evaluate these patients directly; comprehensive evaluation could reveal important morphologic clues, such as the subtle purpuric nature of skin lesions or the characteristic mucosal or ophthalmologic features of COVID-19,” the authors of the editorial stated.
So far, the patterns of skin symptoms, which have been identified in up to 20% of COVID-19–infected patients in some series, have been heterogeneous as demonstrated in the two published case reports.
In one case, a papulosquamous and erythematous periumbilical patch that appeared on the trunk in an elderly patient 1 day after hospital admission for acute respiratory distress rapidly evolved into a digitate papulosquamous eruption involving the upper arms, shoulder, and back. It was described as “clinically reminiscent” of pityriasis rosea by the authors, from the divisions of dermatology and venereology, pathology, intensive care, and the virology laboratory, of the Hôpital Cochin, Paris.
In the other, pruritic erythematous macules, papules, and petechiae affecting the buttocks, popliteal fossae, anterior thighs, and lower abdomen appeared 3 days after the onset of fever in a 48-year-old man hospitalized in Madrid. A biopsy demonstrated a superficial perivascular lymphocytic infiltrate with red cell extravasation and focal papillary edema, “along with focal parakeratosis and isolated dyskeratotic cells,” according to the authors of this report, from the department of dermatology at Ramon y Cajal University, Madrid.
It was unclear whether COVID-19 directly caused either skin eruption. In the patient with the digitate papulosquamous eruption, no virus could be isolated from the skin. Based on high levels of proinflammatory cytokines, it was hypothesized that the rash might have been secondary to an immune response. The rash resolved within a week, but the patient subsequently died of the infection.
In the second case, the petechial lesions, which developed before any treatment was initiated, were said to resemble those associated with other viruses, such as parvovirus B19. This led the investigators to speculate that SARS-CoV-2 “could affect the skin in a similar way,” even though other potential etiologies could not be excluded. Treated with a topical steroid and an oral antihistamine, the skin lesions resolved after 5 days. This patient was discharged after recovering from the respiratory illness after 12 days.
Like previously reported cutaneous eruptions associated with COVID-19 infection, these cases “raise more questions than they provide answers,” wrote the authors of the editorial, but the limited information currently available was the basis for encouraging dermatologists to get involved.
To participate, dermatologists need not necessarily be affiliated with an academic center, according to one of the editorial coauthors, Kanade Shinkai, MD, PhD, professor of dermatology at the University of California, San Francisco. She noted that any health professional is invited to submit cases of COVID-19–associated dermatoses to a registry set up by the American Academy of Dermatology.
It is hoped that cases captured in this registry will create sufficient data to allow clinically relevant patterns and etiologies to be characterized.
The need for data is clear to those on the front lines. Kirsten Lo Sicco, MD, associate director of the skin and cancer unit at New York University, reported that her center is already set up to collect data systematically. “At NYU, we are currently working on standardizing laboratory and histopathology work up for COVID-19 patients who present with various skin eruptions.”
The goal, she added, is “to better determine COVID-19 pathophysiology, systemic associations, patient outcomes, and potential therapeutics.”
“Presumably, many of the eruptions seen in the setting of COVID-19 infection are related,” Dr. Lo Sicco explained in an interview. However, skin complications of infection “may overlap with or be a result of other etiologies as well.”
While better testing for COVID-19 and more lesion biopsies will play a critical role in differentiating etiologies, “we must not overcall COVID-19–related skin eruptions and potentially overlook other diagnoses,” Dr. Lo Sicco said.
In recounting some challenges from the NYU experience so far, Dr. Lo Sicco described the difficulty of differentiating COVID-19–related skin eruptions from skin eruptions caused by treatments, such as antibiotics and antivirals, when the presentation is delayed.
“This is where collaboration with our dermatopathologists becomes important. Drug eruptions, viral exanthems, urticarial eruptions, vasculopathy, and vasculitis can all be differentiated on dermpath,” she said.
One early obstacle to the skin biopsies essential for these types of studies was the limited supply of personal protective equipment at many centers, including hospitals in New York. Biopsies could not be safely performed if supplies of masks and gowns were limited.
Recent evidence suggests that some of the more common morphologies, such as purpuric eruptions, livedo reticularis, and retiform purpura, are linked to the vasculopathy associated with COVID-19 infection, according to Dr. Lo Sicco, but this invites a new set of questions.
One is whether vasculopathies can be prevented with prophylactic anticoagulation. Many hospitalized COVID-19 patients are already receiving therapeutic anticoagulation, but Dr. Lo Sicco questioned whether prophylactic anticoagulation might improve prognosis for outpatients, such as those discharged or those never hospitalized. This is a strategy now being investigated.
Ultimately, she agreed with the thrust of the JAMA Dermatology editorial.
“Dermatologists are vital to determine if various morphologies, such as urticarial, vesicular, purpuric, or papulosquamous lesions, have any specific systemic implications or relate to differences in patient outcomes,” she said.
These are exactly the types of issues being actively investigated at her center.
Neither the authors of the case reports nor of the editorial reported any conflicts of interest.
SOURCEs: Madigan LM et al. JAMA Dermatol. 2020 Apr 30. doi:10.1001/jamadermatol.2020.1438; Diaz-Guimaraens B et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1741; Sanchez A et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1704.
Two infection.
It is not yet clear from these or other case reports which, if any, skin eruptions accompanying COVID-19 infections are caused by the virus, but the authors of the editorial, led by Lauren M. Madigan, MD, of the department of dermatology at the University of Utah, Salt Lake City, urged dermatologists to lead efforts to find out.
“To fully characterize skin manifestations, it may be necessary for dermatologists to evaluate these patients directly; comprehensive evaluation could reveal important morphologic clues, such as the subtle purpuric nature of skin lesions or the characteristic mucosal or ophthalmologic features of COVID-19,” the authors of the editorial stated.
So far, the patterns of skin symptoms, which have been identified in up to 20% of COVID-19–infected patients in some series, have been heterogeneous as demonstrated in the two published case reports.
In one case, a papulosquamous and erythematous periumbilical patch that appeared on the trunk in an elderly patient 1 day after hospital admission for acute respiratory distress rapidly evolved into a digitate papulosquamous eruption involving the upper arms, shoulder, and back. It was described as “clinically reminiscent” of pityriasis rosea by the authors, from the divisions of dermatology and venereology, pathology, intensive care, and the virology laboratory, of the Hôpital Cochin, Paris.
In the other, pruritic erythematous macules, papules, and petechiae affecting the buttocks, popliteal fossae, anterior thighs, and lower abdomen appeared 3 days after the onset of fever in a 48-year-old man hospitalized in Madrid. A biopsy demonstrated a superficial perivascular lymphocytic infiltrate with red cell extravasation and focal papillary edema, “along with focal parakeratosis and isolated dyskeratotic cells,” according to the authors of this report, from the department of dermatology at Ramon y Cajal University, Madrid.
It was unclear whether COVID-19 directly caused either skin eruption. In the patient with the digitate papulosquamous eruption, no virus could be isolated from the skin. Based on high levels of proinflammatory cytokines, it was hypothesized that the rash might have been secondary to an immune response. The rash resolved within a week, but the patient subsequently died of the infection.
In the second case, the petechial lesions, which developed before any treatment was initiated, were said to resemble those associated with other viruses, such as parvovirus B19. This led the investigators to speculate that SARS-CoV-2 “could affect the skin in a similar way,” even though other potential etiologies could not be excluded. Treated with a topical steroid and an oral antihistamine, the skin lesions resolved after 5 days. This patient was discharged after recovering from the respiratory illness after 12 days.
Like previously reported cutaneous eruptions associated with COVID-19 infection, these cases “raise more questions than they provide answers,” wrote the authors of the editorial, but the limited information currently available was the basis for encouraging dermatologists to get involved.
To participate, dermatologists need not necessarily be affiliated with an academic center, according to one of the editorial coauthors, Kanade Shinkai, MD, PhD, professor of dermatology at the University of California, San Francisco. She noted that any health professional is invited to submit cases of COVID-19–associated dermatoses to a registry set up by the American Academy of Dermatology.
It is hoped that cases captured in this registry will create sufficient data to allow clinically relevant patterns and etiologies to be characterized.
The need for data is clear to those on the front lines. Kirsten Lo Sicco, MD, associate director of the skin and cancer unit at New York University, reported that her center is already set up to collect data systematically. “At NYU, we are currently working on standardizing laboratory and histopathology work up for COVID-19 patients who present with various skin eruptions.”
The goal, she added, is “to better determine COVID-19 pathophysiology, systemic associations, patient outcomes, and potential therapeutics.”
“Presumably, many of the eruptions seen in the setting of COVID-19 infection are related,” Dr. Lo Sicco explained in an interview. However, skin complications of infection “may overlap with or be a result of other etiologies as well.”
While better testing for COVID-19 and more lesion biopsies will play a critical role in differentiating etiologies, “we must not overcall COVID-19–related skin eruptions and potentially overlook other diagnoses,” Dr. Lo Sicco said.
In recounting some challenges from the NYU experience so far, Dr. Lo Sicco described the difficulty of differentiating COVID-19–related skin eruptions from skin eruptions caused by treatments, such as antibiotics and antivirals, when the presentation is delayed.
“This is where collaboration with our dermatopathologists becomes important. Drug eruptions, viral exanthems, urticarial eruptions, vasculopathy, and vasculitis can all be differentiated on dermpath,” she said.
One early obstacle to the skin biopsies essential for these types of studies was the limited supply of personal protective equipment at many centers, including hospitals in New York. Biopsies could not be safely performed if supplies of masks and gowns were limited.
Recent evidence suggests that some of the more common morphologies, such as purpuric eruptions, livedo reticularis, and retiform purpura, are linked to the vasculopathy associated with COVID-19 infection, according to Dr. Lo Sicco, but this invites a new set of questions.
One is whether vasculopathies can be prevented with prophylactic anticoagulation. Many hospitalized COVID-19 patients are already receiving therapeutic anticoagulation, but Dr. Lo Sicco questioned whether prophylactic anticoagulation might improve prognosis for outpatients, such as those discharged or those never hospitalized. This is a strategy now being investigated.
Ultimately, she agreed with the thrust of the JAMA Dermatology editorial.
“Dermatologists are vital to determine if various morphologies, such as urticarial, vesicular, purpuric, or papulosquamous lesions, have any specific systemic implications or relate to differences in patient outcomes,” she said.
These are exactly the types of issues being actively investigated at her center.
Neither the authors of the case reports nor of the editorial reported any conflicts of interest.
SOURCEs: Madigan LM et al. JAMA Dermatol. 2020 Apr 30. doi:10.1001/jamadermatol.2020.1438; Diaz-Guimaraens B et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1741; Sanchez A et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1704.
Two infection.
It is not yet clear from these or other case reports which, if any, skin eruptions accompanying COVID-19 infections are caused by the virus, but the authors of the editorial, led by Lauren M. Madigan, MD, of the department of dermatology at the University of Utah, Salt Lake City, urged dermatologists to lead efforts to find out.
“To fully characterize skin manifestations, it may be necessary for dermatologists to evaluate these patients directly; comprehensive evaluation could reveal important morphologic clues, such as the subtle purpuric nature of skin lesions or the characteristic mucosal or ophthalmologic features of COVID-19,” the authors of the editorial stated.
So far, the patterns of skin symptoms, which have been identified in up to 20% of COVID-19–infected patients in some series, have been heterogeneous as demonstrated in the two published case reports.
In one case, a papulosquamous and erythematous periumbilical patch that appeared on the trunk in an elderly patient 1 day after hospital admission for acute respiratory distress rapidly evolved into a digitate papulosquamous eruption involving the upper arms, shoulder, and back. It was described as “clinically reminiscent” of pityriasis rosea by the authors, from the divisions of dermatology and venereology, pathology, intensive care, and the virology laboratory, of the Hôpital Cochin, Paris.
In the other, pruritic erythematous macules, papules, and petechiae affecting the buttocks, popliteal fossae, anterior thighs, and lower abdomen appeared 3 days after the onset of fever in a 48-year-old man hospitalized in Madrid. A biopsy demonstrated a superficial perivascular lymphocytic infiltrate with red cell extravasation and focal papillary edema, “along with focal parakeratosis and isolated dyskeratotic cells,” according to the authors of this report, from the department of dermatology at Ramon y Cajal University, Madrid.
It was unclear whether COVID-19 directly caused either skin eruption. In the patient with the digitate papulosquamous eruption, no virus could be isolated from the skin. Based on high levels of proinflammatory cytokines, it was hypothesized that the rash might have been secondary to an immune response. The rash resolved within a week, but the patient subsequently died of the infection.
In the second case, the petechial lesions, which developed before any treatment was initiated, were said to resemble those associated with other viruses, such as parvovirus B19. This led the investigators to speculate that SARS-CoV-2 “could affect the skin in a similar way,” even though other potential etiologies could not be excluded. Treated with a topical steroid and an oral antihistamine, the skin lesions resolved after 5 days. This patient was discharged after recovering from the respiratory illness after 12 days.
Like previously reported cutaneous eruptions associated with COVID-19 infection, these cases “raise more questions than they provide answers,” wrote the authors of the editorial, but the limited information currently available was the basis for encouraging dermatologists to get involved.
To participate, dermatologists need not necessarily be affiliated with an academic center, according to one of the editorial coauthors, Kanade Shinkai, MD, PhD, professor of dermatology at the University of California, San Francisco. She noted that any health professional is invited to submit cases of COVID-19–associated dermatoses to a registry set up by the American Academy of Dermatology.
It is hoped that cases captured in this registry will create sufficient data to allow clinically relevant patterns and etiologies to be characterized.
The need for data is clear to those on the front lines. Kirsten Lo Sicco, MD, associate director of the skin and cancer unit at New York University, reported that her center is already set up to collect data systematically. “At NYU, we are currently working on standardizing laboratory and histopathology work up for COVID-19 patients who present with various skin eruptions.”
The goal, she added, is “to better determine COVID-19 pathophysiology, systemic associations, patient outcomes, and potential therapeutics.”
“Presumably, many of the eruptions seen in the setting of COVID-19 infection are related,” Dr. Lo Sicco explained in an interview. However, skin complications of infection “may overlap with or be a result of other etiologies as well.”
While better testing for COVID-19 and more lesion biopsies will play a critical role in differentiating etiologies, “we must not overcall COVID-19–related skin eruptions and potentially overlook other diagnoses,” Dr. Lo Sicco said.
In recounting some challenges from the NYU experience so far, Dr. Lo Sicco described the difficulty of differentiating COVID-19–related skin eruptions from skin eruptions caused by treatments, such as antibiotics and antivirals, when the presentation is delayed.
“This is where collaboration with our dermatopathologists becomes important. Drug eruptions, viral exanthems, urticarial eruptions, vasculopathy, and vasculitis can all be differentiated on dermpath,” she said.
One early obstacle to the skin biopsies essential for these types of studies was the limited supply of personal protective equipment at many centers, including hospitals in New York. Biopsies could not be safely performed if supplies of masks and gowns were limited.
Recent evidence suggests that some of the more common morphologies, such as purpuric eruptions, livedo reticularis, and retiform purpura, are linked to the vasculopathy associated with COVID-19 infection, according to Dr. Lo Sicco, but this invites a new set of questions.
One is whether vasculopathies can be prevented with prophylactic anticoagulation. Many hospitalized COVID-19 patients are already receiving therapeutic anticoagulation, but Dr. Lo Sicco questioned whether prophylactic anticoagulation might improve prognosis for outpatients, such as those discharged or those never hospitalized. This is a strategy now being investigated.
Ultimately, she agreed with the thrust of the JAMA Dermatology editorial.
“Dermatologists are vital to determine if various morphologies, such as urticarial, vesicular, purpuric, or papulosquamous lesions, have any specific systemic implications or relate to differences in patient outcomes,” she said.
These are exactly the types of issues being actively investigated at her center.
Neither the authors of the case reports nor of the editorial reported any conflicts of interest.
SOURCEs: Madigan LM et al. JAMA Dermatol. 2020 Apr 30. doi:10.1001/jamadermatol.2020.1438; Diaz-Guimaraens B et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1741; Sanchez A et al. JAMA Dermatol. 2020 Apr 30. doi: 10.1001/jamadermatol.2020.1704.
Gastroenterology groups map a return to elective endoscopy
Gastroenterologists can safely return to elective procedures when adequate personal protective equipment (PPE) is available, professional societies say.
Noting that some states have already lifted restrictions imposed to guard against COVID-19, the American Gastroenterological Association (AGA) and the Digestive Health Physicians Association (DHPA) on April 27 announced guidelines for resuming procedures delayed by the pandemic.
“Gastroenterologists are looking for some framework, however fluid it might be, to guide them in the next 2 to 4 weeks,” Paul Berggreen, MD, secretary of the DHPA, told Medscape Medical News.
The AGA and DHPA guidelines envision a return to elective procedures in areas where COVID-19 cases have been declining for at least 2 weeks and where they are permitted by government directives.
Decisions hinge on the availability of testing, Berggreen said. The guidelines recommend polymerase chain reaction (PCR) tests for COVID-19 infections prior to elective endoscopy. When these tests are not available, a daily temperature log for 10 days prior to the procedure may substitute, they say.
However, if no PCR test is done, the guidelines call on all procedure room personnel to use N95 masks or the equivalent. If these masks aren’t available, “consider delaying resumption of endoscopic procedures,” the guidelines say. The procedure should also be postponed or moved to an inpatient setting in the event of a positive test, according to the guidelines.
Only if the patient has a negative test result should the procedure go forward with the use of standard surgical masks rather than N95 masks or the equivalent, the guidelines say.
The mask recommendations differ slightly from a decision tree put forward during an American College of Gastroenterology (ACG) webinar on April 27, ACG President Mark Pochapin, MD, told Medscape Medical News.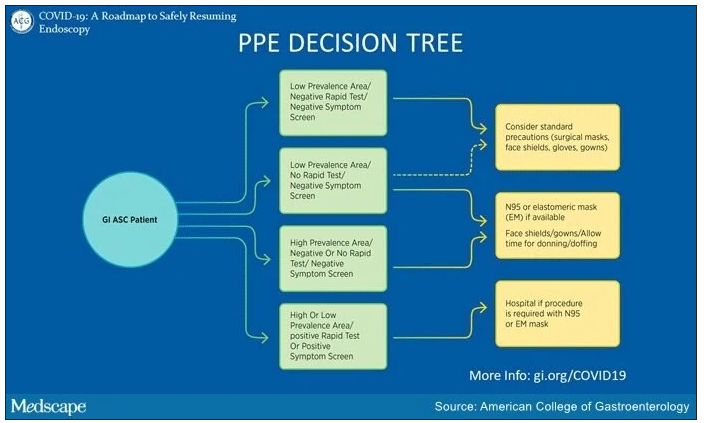
The ACG decision tree takes into consideration local prevalence of COVID-19. “In a low prevalence area if you have a negative test you can wear a regular surgical mask if the patient wears a surgical mask,” Pochapin said.
The ACG decision tree also envisions the possibility of endoscopy with surgical masks along with face shields in areas with low prevalence of COVID-19, even in the absence of testing, if a patient doesn’t have symptoms.
In contrast, the ACG decision tree calls for N95 or elastomeric masks in areas of high prevalence even with a negative test and a negative symptom screen. And it calls for a hospital procedure with an N95 or elastomeric mask in patients with either a positive symptom screen or a positive test.
In addition to masks, the AGA and DHPA guidelines recommend use of other PPE, such as water-resistant gowns, shoe covers, scrubs, double-gloving, and surgical head coverings.
They recommend daily screening of endoscopy center staff with temperature checks and surveys of COVID-19 symptoms and exposure.
Moreover, they call for social distancing of patients, visitors, and staff, and high-level disinfection of endoscopes. They recommend against endotracheal intubation of patients undergoing elective upper endoscopy.
The number of states allowing elective procedures is changing by the day, Berggreen said. In Arizona, where he practices, Gov. Doug Ducey removed all restrictions on elective procedures starting May 1. But other states, where caseloads have been higher, including New York and Massachusetts, have yet to follow suit.
“We are still in kind of a holding pattern,” Richard Hodin, MD, AGAF, chief of gastrointestinal surgery at Massachusetts General Hospital in Boston, told Medscape Medical News. Currently his facility is only doing procedures when a patient’s life is in danger.
The availability of testing also ranges widely from one practice to another. New York University, where Pochapin is director of gastroenterology, is able to do its own tests. Berggreen’s practice, Arizona Digestive Health in Phoenix, has assigned staff to swab patients in its parking lot and then send the samples to a lab by courier for analysis.
Berggreen said his practice has been essentially closed for the past month. In May, he expects his team will do about 30% of its normal volume. They will not start with purely elective procedures, such as following up on a polyp, but with semi-urgent cases, said Berggreen.
“Elective procedures can still be delayed a little bit longer,” he said. “But we’re trying to take care of our patients that are not purely elective: somebody with abdominal pain that you think is very likely a stomach ulcer, somebody with rectal bleeding or persistent diarrhea that›s really impacting their life and you›re thinking this could be an inflammatory condition of the colon.»
Berggreen said he is reassured by a recent survey of 968 healthcare workers in Northern Italy who conducted gastrointestinal endoscopy there during the COVID-19 outbreak. Only 4.3% of respondents tested positive for COVID-19, and 85.7% of these infections occurred before the introduction of PPE and measures to reduce cases. Results were similarly encouraging for patients.
Providing more endoscopy will relieve many patients, Berggreen said. “We all understand the need to limit the spread of the coronavirus but we also have patients who are going to start to have more struggles and potentially worse outcomes by sitting on a condition that requires endoscopy to diagnose and appropriately manage.”
Neither Berggreen, Pochapin, nor Hodin reported any relevant financial interests. The authors of the survey did not report a source of funding or any relevant financial interests.
This article first appeared on Medscape.com.
Gastroenterologists can safely return to elective procedures when adequate personal protective equipment (PPE) is available, professional societies say.
Noting that some states have already lifted restrictions imposed to guard against COVID-19, the American Gastroenterological Association (AGA) and the Digestive Health Physicians Association (DHPA) on April 27 announced guidelines for resuming procedures delayed by the pandemic.
“Gastroenterologists are looking for some framework, however fluid it might be, to guide them in the next 2 to 4 weeks,” Paul Berggreen, MD, secretary of the DHPA, told Medscape Medical News.
The AGA and DHPA guidelines envision a return to elective procedures in areas where COVID-19 cases have been declining for at least 2 weeks and where they are permitted by government directives.
Decisions hinge on the availability of testing, Berggreen said. The guidelines recommend polymerase chain reaction (PCR) tests for COVID-19 infections prior to elective endoscopy. When these tests are not available, a daily temperature log for 10 days prior to the procedure may substitute, they say.
However, if no PCR test is done, the guidelines call on all procedure room personnel to use N95 masks or the equivalent. If these masks aren’t available, “consider delaying resumption of endoscopic procedures,” the guidelines say. The procedure should also be postponed or moved to an inpatient setting in the event of a positive test, according to the guidelines.
Only if the patient has a negative test result should the procedure go forward with the use of standard surgical masks rather than N95 masks or the equivalent, the guidelines say.
The mask recommendations differ slightly from a decision tree put forward during an American College of Gastroenterology (ACG) webinar on April 27, ACG President Mark Pochapin, MD, told Medscape Medical News.
The ACG decision tree takes into consideration local prevalence of COVID-19. “In a low prevalence area if you have a negative test you can wear a regular surgical mask if the patient wears a surgical mask,” Pochapin said.
The ACG decision tree also envisions the possibility of endoscopy with surgical masks along with face shields in areas with low prevalence of COVID-19, even in the absence of testing, if a patient doesn’t have symptoms.
In contrast, the ACG decision tree calls for N95 or elastomeric masks in areas of high prevalence even with a negative test and a negative symptom screen. And it calls for a hospital procedure with an N95 or elastomeric mask in patients with either a positive symptom screen or a positive test.
In addition to masks, the AGA and DHPA guidelines recommend use of other PPE, such as water-resistant gowns, shoe covers, scrubs, double-gloving, and surgical head coverings.
They recommend daily screening of endoscopy center staff with temperature checks and surveys of COVID-19 symptoms and exposure.
Moreover, they call for social distancing of patients, visitors, and staff, and high-level disinfection of endoscopes. They recommend against endotracheal intubation of patients undergoing elective upper endoscopy.
The number of states allowing elective procedures is changing by the day, Berggreen said. In Arizona, where he practices, Gov. Doug Ducey removed all restrictions on elective procedures starting May 1. But other states, where caseloads have been higher, including New York and Massachusetts, have yet to follow suit.
“We are still in kind of a holding pattern,” Richard Hodin, MD, AGAF, chief of gastrointestinal surgery at Massachusetts General Hospital in Boston, told Medscape Medical News. Currently his facility is only doing procedures when a patient’s life is in danger.
The availability of testing also ranges widely from one practice to another. New York University, where Pochapin is director of gastroenterology, is able to do its own tests. Berggreen’s practice, Arizona Digestive Health in Phoenix, has assigned staff to swab patients in its parking lot and then send the samples to a lab by courier for analysis.
Berggreen said his practice has been essentially closed for the past month. In May, he expects his team will do about 30% of its normal volume. They will not start with purely elective procedures, such as following up on a polyp, but with semi-urgent cases, said Berggreen.
“Elective procedures can still be delayed a little bit longer,” he said. “But we’re trying to take care of our patients that are not purely elective: somebody with abdominal pain that you think is very likely a stomach ulcer, somebody with rectal bleeding or persistent diarrhea that›s really impacting their life and you›re thinking this could be an inflammatory condition of the colon.»
Berggreen said he is reassured by a recent survey of 968 healthcare workers in Northern Italy who conducted gastrointestinal endoscopy there during the COVID-19 outbreak. Only 4.3% of respondents tested positive for COVID-19, and 85.7% of these infections occurred before the introduction of PPE and measures to reduce cases. Results were similarly encouraging for patients.
Providing more endoscopy will relieve many patients, Berggreen said. “We all understand the need to limit the spread of the coronavirus but we also have patients who are going to start to have more struggles and potentially worse outcomes by sitting on a condition that requires endoscopy to diagnose and appropriately manage.”
Neither Berggreen, Pochapin, nor Hodin reported any relevant financial interests. The authors of the survey did not report a source of funding or any relevant financial interests.
This article first appeared on Medscape.com.
Gastroenterologists can safely return to elective procedures when adequate personal protective equipment (PPE) is available, professional societies say.
Noting that some states have already lifted restrictions imposed to guard against COVID-19, the American Gastroenterological Association (AGA) and the Digestive Health Physicians Association (DHPA) on April 27 announced guidelines for resuming procedures delayed by the pandemic.
“Gastroenterologists are looking for some framework, however fluid it might be, to guide them in the next 2 to 4 weeks,” Paul Berggreen, MD, secretary of the DHPA, told Medscape Medical News.
The AGA and DHPA guidelines envision a return to elective procedures in areas where COVID-19 cases have been declining for at least 2 weeks and where they are permitted by government directives.
Decisions hinge on the availability of testing, Berggreen said. The guidelines recommend polymerase chain reaction (PCR) tests for COVID-19 infections prior to elective endoscopy. When these tests are not available, a daily temperature log for 10 days prior to the procedure may substitute, they say.
However, if no PCR test is done, the guidelines call on all procedure room personnel to use N95 masks or the equivalent. If these masks aren’t available, “consider delaying resumption of endoscopic procedures,” the guidelines say. The procedure should also be postponed or moved to an inpatient setting in the event of a positive test, according to the guidelines.
Only if the patient has a negative test result should the procedure go forward with the use of standard surgical masks rather than N95 masks or the equivalent, the guidelines say.
The mask recommendations differ slightly from a decision tree put forward during an American College of Gastroenterology (ACG) webinar on April 27, ACG President Mark Pochapin, MD, told Medscape Medical News.
The ACG decision tree takes into consideration local prevalence of COVID-19. “In a low prevalence area if you have a negative test you can wear a regular surgical mask if the patient wears a surgical mask,” Pochapin said.
The ACG decision tree also envisions the possibility of endoscopy with surgical masks along with face shields in areas with low prevalence of COVID-19, even in the absence of testing, if a patient doesn’t have symptoms.
In contrast, the ACG decision tree calls for N95 or elastomeric masks in areas of high prevalence even with a negative test and a negative symptom screen. And it calls for a hospital procedure with an N95 or elastomeric mask in patients with either a positive symptom screen or a positive test.
In addition to masks, the AGA and DHPA guidelines recommend use of other PPE, such as water-resistant gowns, shoe covers, scrubs, double-gloving, and surgical head coverings.
They recommend daily screening of endoscopy center staff with temperature checks and surveys of COVID-19 symptoms and exposure.
Moreover, they call for social distancing of patients, visitors, and staff, and high-level disinfection of endoscopes. They recommend against endotracheal intubation of patients undergoing elective upper endoscopy.
The number of states allowing elective procedures is changing by the day, Berggreen said. In Arizona, where he practices, Gov. Doug Ducey removed all restrictions on elective procedures starting May 1. But other states, where caseloads have been higher, including New York and Massachusetts, have yet to follow suit.
“We are still in kind of a holding pattern,” Richard Hodin, MD, AGAF, chief of gastrointestinal surgery at Massachusetts General Hospital in Boston, told Medscape Medical News. Currently his facility is only doing procedures when a patient’s life is in danger.
The availability of testing also ranges widely from one practice to another. New York University, where Pochapin is director of gastroenterology, is able to do its own tests. Berggreen’s practice, Arizona Digestive Health in Phoenix, has assigned staff to swab patients in its parking lot and then send the samples to a lab by courier for analysis.
Berggreen said his practice has been essentially closed for the past month. In May, he expects his team will do about 30% of its normal volume. They will not start with purely elective procedures, such as following up on a polyp, but with semi-urgent cases, said Berggreen.
“Elective procedures can still be delayed a little bit longer,” he said. “But we’re trying to take care of our patients that are not purely elective: somebody with abdominal pain that you think is very likely a stomach ulcer, somebody with rectal bleeding or persistent diarrhea that›s really impacting their life and you›re thinking this could be an inflammatory condition of the colon.»
Berggreen said he is reassured by a recent survey of 968 healthcare workers in Northern Italy who conducted gastrointestinal endoscopy there during the COVID-19 outbreak. Only 4.3% of respondents tested positive for COVID-19, and 85.7% of these infections occurred before the introduction of PPE and measures to reduce cases. Results were similarly encouraging for patients.
Providing more endoscopy will relieve many patients, Berggreen said. “We all understand the need to limit the spread of the coronavirus but we also have patients who are going to start to have more struggles and potentially worse outcomes by sitting on a condition that requires endoscopy to diagnose and appropriately manage.”
Neither Berggreen, Pochapin, nor Hodin reported any relevant financial interests. The authors of the survey did not report a source of funding or any relevant financial interests.
This article first appeared on Medscape.com.
Reporting for duty at the front lines of COVID-19: From your editor in chief
I am writing this in mid-April, in time for our May issue. These are unusual times. During unusual times, people rise up and do unusual and exemplary things. I firmly believe in the ability of humans to rise to the occasion and step out of their boundaries and boxes when needed. And the current COVID-19 pandemic is no exception. Our patients need us. The medical community needs us, and hematologists around the world have stepped up to help in any ways they can.
Since the beginning of the year with rumblings of the emergence of a novel SARs-CoV-2 virus in patients with influenza-like illness, the hematology community has banded together to figure out what this will mean for our patients battling malignant and nonmalignant blood disorders.
With very little published literature to go on, we have had to glean from our experience with the H1N1 influenza pandemic to develop a strategy to support hematology patients who may develop COVID-19 infection. With more questions than answers, institutions around the country and globally began to collaborate and communicate furiously with each other to learn from those who had experienced the effects of the virus before we had. We have been learning to anticipate blood supply changes, treatment modifications, and therapeutic needs for those who will inevitably get the virus.
Concern rose not just for the patients but also for the providers and clinical team who care for the hematology patients. How do we preserve and protect our workforce? A pandemic does not prevent new-diagnosis leukemia or myelodysplastic syndrome blast crisis from presenting as usual at 3 p.m. on a Friday afternoon. Who is at highest risk among the staff? If we practice social distancing what does that look like in a hematology clinic, in an infusion room? Or on the stem cell transplant in-patient unit? So many questions with minimal scientific evidence to guide our decisions.
As a sickle cell disease (SCD) specialist, I had some unique concerns. Roughly 10%-15% of the sickle cell population in the United States are supported by monthly red blood cell exchange transfusions, a lifesaving therapy to prevent recurrent stroke and to manage severe end organ damage. The vast majority of patients are on some disease-modifying therapy that requires ongoing lifelong monitoring of hematologic parameters, as well as renal and hepatic function. Most SCD patients also are members of racial minorities, live in densely populated parts of the city, and have significant social determinants of health that make adherence to social distancing mandates near impossible.
Frequent exposure to acute care for painful exacerbations of their disease, preexisting comorbidities involving the lung, heart, and kidney, and their underlying cellular and humoral immune dysfunction also put our patients at heightened risk of contracting COVID-19 infection.
So how have we handled the COVID-19 pandemic thus far? We have engaged various partners, collaborators, and colleagues to figure things out. Our institutions have established incident command operations to supervise and guide bed management, staff deployment/redeployment, and the supply chain, particularly as regards personal protective equipment; and to support physician and staff wellness. Our administrative leaders have partnered seamlessly with clinical leaders to rapidly roll out robust telemedicine strategies so that we can continue to provide ongoing medical care as best we can.
We have worked tirelessly across disciplines to develop guidance documents that are specialty specific with ways to support disease populations working with the hospitalist and acute care units to define testing, treatment, and admission and discharge criteria. We have engaged communications teams that have developed health-literate public messaging for the patients and the community about coronavirus as well as the rapidly changing public health guidelines to help #flattenthecurve.
As providers, we have reviewed our patient panels to determine who can tolerate appointment delays and who has to come in to be seen with minimal impact to health outcomes. We have read more articles in the past month than perhaps the past year; listened to more podcasts, webinars, and virtual lectures on COVID-19 and strategies to halt spread of the virus – just trying to learn more. We have engaged in social media – following COVID-19 public and private groups – to get and to offer support, as well as keep a finger on the pulse of the health community around this pandemic.
For my SCD population, I have had to decide who can tolerate simple rather than exchange transfusions for the next 3-6 months and what is the minimum number of red cell units we can safely use per red cell exchange procedure as we prepare for a possible blood supply shortage. The hematology community has worked tirelessly with national societies and numerous stakeholder groups to develop a comprehensive toolkit with regularly updated information about COVID-19 relevant to the hematology community (hematology.org/covid-19).
At a practice level, we are proactively reaching out to our hematology patients and their families to reassure them and connect them with resources and support while ensuring that they have adequate supply of their daily hematology medications with tips like using the pharmacy drive-through or home medication delivery options. The past 2 weeks in Charlotte, N.C., have been hectic with preparation. My days are long; a mixture of telemedicine visits, strategic meetings, and meetings to cascade the newest plan to the staff so that they know and are comfortable with it for the patients they take care of.
When the adrenaline from each day begins to wane, we think of our individual families; we worry about relatives far and near. We mourn the loss of loved ones or other hematologists or providers who have succumbed to the COVID-19 virus. We take a minute to think about ourselves and how this pandemic affects us individually and personally. I think about my older sister who runs a smaller hospital in the Bronx, N.Y. She is at the epicenter of the pandemic and is short-staffed in the ICU and medicine floors. Because she is an ob.gyn, she has called me for guidance on a pregnant woman with anemia and sickle cell trait. I hadn’t heard from her in 24 hours and I began to wonder – is she okay? Why didn’t she answer my call this morning? Is she sick? Did she get the information I sent to her linking her with our virtual ICU experts so she can implement a similar program for her hospital?
Next, I think of my younger sister in Long Island, N.Y., who was covering shifts as a hospitalist. She had asked me to send her some hematology tips on managing disseminated intravascular coagulopathy in COVID-19 patients as she has limited access to consultants. She sees an average of seven to nine COVID-19–positive patients and several persons under investigation per shift.
I also think of my 76-year-old mom who is upset that she cannot go to the adult center because of social distancing. So, even though I am weary, I do a FaceTime call with mom. I try to explain why it’s important for her to stay indoors. It’s only temporary, I reassure her; but I cannot say how long “temporary” is.
I pack up my bags, change out of my scrubs to head to my car thinking of my daughter who just turned 21 years old and was so excited about her college graduation in May. She had a meltdown yesterday because her university announced there will be no in-person gradation. I wonder how I can help her see the big picture and yet, why should she? She’s only 21.
Then I get a page – it’s a patient with sickle cell disease – my first COVID-19–positive patient. As I take the history and turn my computer back on to do this consult, I realize that this is what all the preparation was for. The sickle cell guidance document we had worked on over the past weeks to outline a step-by-step approach to managing a SCD patient with COVID-19 that is intentionally aligned with our institutions COVID-19 treatment protocol with specific nuances relevant to patients with red blood cell disorders, was now being put to use. I felt glad for my patient that we were prepared and had a semblance of a plan on how to approach his care.
The battle is far from over. Actually, as of my writing this, it’s just starting in my part of the country. The days will continue to be long. I continue to appreciate the beauty of the human spirit among the people we work with, the hematology community we belong to, and the patients that we serve. I am committed (as are all of you) to staying “on-duty” for as long as I can, and I’d like to take this opportunity to say to all the hematologists out there – “Thank you for your service and for reporting for duty to the front lines.”
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.
I am writing this in mid-April, in time for our May issue. These are unusual times. During unusual times, people rise up and do unusual and exemplary things. I firmly believe in the ability of humans to rise to the occasion and step out of their boundaries and boxes when needed. And the current COVID-19 pandemic is no exception. Our patients need us. The medical community needs us, and hematologists around the world have stepped up to help in any ways they can.
Since the beginning of the year with rumblings of the emergence of a novel SARs-CoV-2 virus in patients with influenza-like illness, the hematology community has banded together to figure out what this will mean for our patients battling malignant and nonmalignant blood disorders.
With very little published literature to go on, we have had to glean from our experience with the H1N1 influenza pandemic to develop a strategy to support hematology patients who may develop COVID-19 infection. With more questions than answers, institutions around the country and globally began to collaborate and communicate furiously with each other to learn from those who had experienced the effects of the virus before we had. We have been learning to anticipate blood supply changes, treatment modifications, and therapeutic needs for those who will inevitably get the virus.
Concern rose not just for the patients but also for the providers and clinical team who care for the hematology patients. How do we preserve and protect our workforce? A pandemic does not prevent new-diagnosis leukemia or myelodysplastic syndrome blast crisis from presenting as usual at 3 p.m. on a Friday afternoon. Who is at highest risk among the staff? If we practice social distancing what does that look like in a hematology clinic, in an infusion room? Or on the stem cell transplant in-patient unit? So many questions with minimal scientific evidence to guide our decisions.
As a sickle cell disease (SCD) specialist, I had some unique concerns. Roughly 10%-15% of the sickle cell population in the United States are supported by monthly red blood cell exchange transfusions, a lifesaving therapy to prevent recurrent stroke and to manage severe end organ damage. The vast majority of patients are on some disease-modifying therapy that requires ongoing lifelong monitoring of hematologic parameters, as well as renal and hepatic function. Most SCD patients also are members of racial minorities, live in densely populated parts of the city, and have significant social determinants of health that make adherence to social distancing mandates near impossible.
Frequent exposure to acute care for painful exacerbations of their disease, preexisting comorbidities involving the lung, heart, and kidney, and their underlying cellular and humoral immune dysfunction also put our patients at heightened risk of contracting COVID-19 infection.
So how have we handled the COVID-19 pandemic thus far? We have engaged various partners, collaborators, and colleagues to figure things out. Our institutions have established incident command operations to supervise and guide bed management, staff deployment/redeployment, and the supply chain, particularly as regards personal protective equipment; and to support physician and staff wellness. Our administrative leaders have partnered seamlessly with clinical leaders to rapidly roll out robust telemedicine strategies so that we can continue to provide ongoing medical care as best we can.
We have worked tirelessly across disciplines to develop guidance documents that are specialty specific with ways to support disease populations working with the hospitalist and acute care units to define testing, treatment, and admission and discharge criteria. We have engaged communications teams that have developed health-literate public messaging for the patients and the community about coronavirus as well as the rapidly changing public health guidelines to help #flattenthecurve.
As providers, we have reviewed our patient panels to determine who can tolerate appointment delays and who has to come in to be seen with minimal impact to health outcomes. We have read more articles in the past month than perhaps the past year; listened to more podcasts, webinars, and virtual lectures on COVID-19 and strategies to halt spread of the virus – just trying to learn more. We have engaged in social media – following COVID-19 public and private groups – to get and to offer support, as well as keep a finger on the pulse of the health community around this pandemic.
For my SCD population, I have had to decide who can tolerate simple rather than exchange transfusions for the next 3-6 months and what is the minimum number of red cell units we can safely use per red cell exchange procedure as we prepare for a possible blood supply shortage. The hematology community has worked tirelessly with national societies and numerous stakeholder groups to develop a comprehensive toolkit with regularly updated information about COVID-19 relevant to the hematology community (hematology.org/covid-19).
At a practice level, we are proactively reaching out to our hematology patients and their families to reassure them and connect them with resources and support while ensuring that they have adequate supply of their daily hematology medications with tips like using the pharmacy drive-through or home medication delivery options. The past 2 weeks in Charlotte, N.C., have been hectic with preparation. My days are long; a mixture of telemedicine visits, strategic meetings, and meetings to cascade the newest plan to the staff so that they know and are comfortable with it for the patients they take care of.
When the adrenaline from each day begins to wane, we think of our individual families; we worry about relatives far and near. We mourn the loss of loved ones or other hematologists or providers who have succumbed to the COVID-19 virus. We take a minute to think about ourselves and how this pandemic affects us individually and personally. I think about my older sister who runs a smaller hospital in the Bronx, N.Y. She is at the epicenter of the pandemic and is short-staffed in the ICU and medicine floors. Because she is an ob.gyn, she has called me for guidance on a pregnant woman with anemia and sickle cell trait. I hadn’t heard from her in 24 hours and I began to wonder – is she okay? Why didn’t she answer my call this morning? Is she sick? Did she get the information I sent to her linking her with our virtual ICU experts so she can implement a similar program for her hospital?
Next, I think of my younger sister in Long Island, N.Y., who was covering shifts as a hospitalist. She had asked me to send her some hematology tips on managing disseminated intravascular coagulopathy in COVID-19 patients as she has limited access to consultants. She sees an average of seven to nine COVID-19–positive patients and several persons under investigation per shift.
I also think of my 76-year-old mom who is upset that she cannot go to the adult center because of social distancing. So, even though I am weary, I do a FaceTime call with mom. I try to explain why it’s important for her to stay indoors. It’s only temporary, I reassure her; but I cannot say how long “temporary” is.
I pack up my bags, change out of my scrubs to head to my car thinking of my daughter who just turned 21 years old and was so excited about her college graduation in May. She had a meltdown yesterday because her university announced there will be no in-person gradation. I wonder how I can help her see the big picture and yet, why should she? She’s only 21.
Then I get a page – it’s a patient with sickle cell disease – my first COVID-19–positive patient. As I take the history and turn my computer back on to do this consult, I realize that this is what all the preparation was for. The sickle cell guidance document we had worked on over the past weeks to outline a step-by-step approach to managing a SCD patient with COVID-19 that is intentionally aligned with our institutions COVID-19 treatment protocol with specific nuances relevant to patients with red blood cell disorders, was now being put to use. I felt glad for my patient that we were prepared and had a semblance of a plan on how to approach his care.
The battle is far from over. Actually, as of my writing this, it’s just starting in my part of the country. The days will continue to be long. I continue to appreciate the beauty of the human spirit among the people we work with, the hematology community we belong to, and the patients that we serve. I am committed (as are all of you) to staying “on-duty” for as long as I can, and I’d like to take this opportunity to say to all the hematologists out there – “Thank you for your service and for reporting for duty to the front lines.”
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.
I am writing this in mid-April, in time for our May issue. These are unusual times. During unusual times, people rise up and do unusual and exemplary things. I firmly believe in the ability of humans to rise to the occasion and step out of their boundaries and boxes when needed. And the current COVID-19 pandemic is no exception. Our patients need us. The medical community needs us, and hematologists around the world have stepped up to help in any ways they can.
Since the beginning of the year with rumblings of the emergence of a novel SARs-CoV-2 virus in patients with influenza-like illness, the hematology community has banded together to figure out what this will mean for our patients battling malignant and nonmalignant blood disorders.
With very little published literature to go on, we have had to glean from our experience with the H1N1 influenza pandemic to develop a strategy to support hematology patients who may develop COVID-19 infection. With more questions than answers, institutions around the country and globally began to collaborate and communicate furiously with each other to learn from those who had experienced the effects of the virus before we had. We have been learning to anticipate blood supply changes, treatment modifications, and therapeutic needs for those who will inevitably get the virus.
Concern rose not just for the patients but also for the providers and clinical team who care for the hematology patients. How do we preserve and protect our workforce? A pandemic does not prevent new-diagnosis leukemia or myelodysplastic syndrome blast crisis from presenting as usual at 3 p.m. on a Friday afternoon. Who is at highest risk among the staff? If we practice social distancing what does that look like in a hematology clinic, in an infusion room? Or on the stem cell transplant in-patient unit? So many questions with minimal scientific evidence to guide our decisions.
As a sickle cell disease (SCD) specialist, I had some unique concerns. Roughly 10%-15% of the sickle cell population in the United States are supported by monthly red blood cell exchange transfusions, a lifesaving therapy to prevent recurrent stroke and to manage severe end organ damage. The vast majority of patients are on some disease-modifying therapy that requires ongoing lifelong monitoring of hematologic parameters, as well as renal and hepatic function. Most SCD patients also are members of racial minorities, live in densely populated parts of the city, and have significant social determinants of health that make adherence to social distancing mandates near impossible.
Frequent exposure to acute care for painful exacerbations of their disease, preexisting comorbidities involving the lung, heart, and kidney, and their underlying cellular and humoral immune dysfunction also put our patients at heightened risk of contracting COVID-19 infection.
So how have we handled the COVID-19 pandemic thus far? We have engaged various partners, collaborators, and colleagues to figure things out. Our institutions have established incident command operations to supervise and guide bed management, staff deployment/redeployment, and the supply chain, particularly as regards personal protective equipment; and to support physician and staff wellness. Our administrative leaders have partnered seamlessly with clinical leaders to rapidly roll out robust telemedicine strategies so that we can continue to provide ongoing medical care as best we can.
We have worked tirelessly across disciplines to develop guidance documents that are specialty specific with ways to support disease populations working with the hospitalist and acute care units to define testing, treatment, and admission and discharge criteria. We have engaged communications teams that have developed health-literate public messaging for the patients and the community about coronavirus as well as the rapidly changing public health guidelines to help #flattenthecurve.
As providers, we have reviewed our patient panels to determine who can tolerate appointment delays and who has to come in to be seen with minimal impact to health outcomes. We have read more articles in the past month than perhaps the past year; listened to more podcasts, webinars, and virtual lectures on COVID-19 and strategies to halt spread of the virus – just trying to learn more. We have engaged in social media – following COVID-19 public and private groups – to get and to offer support, as well as keep a finger on the pulse of the health community around this pandemic.
For my SCD population, I have had to decide who can tolerate simple rather than exchange transfusions for the next 3-6 months and what is the minimum number of red cell units we can safely use per red cell exchange procedure as we prepare for a possible blood supply shortage. The hematology community has worked tirelessly with national societies and numerous stakeholder groups to develop a comprehensive toolkit with regularly updated information about COVID-19 relevant to the hematology community (hematology.org/covid-19).
At a practice level, we are proactively reaching out to our hematology patients and their families to reassure them and connect them with resources and support while ensuring that they have adequate supply of their daily hematology medications with tips like using the pharmacy drive-through or home medication delivery options. The past 2 weeks in Charlotte, N.C., have been hectic with preparation. My days are long; a mixture of telemedicine visits, strategic meetings, and meetings to cascade the newest plan to the staff so that they know and are comfortable with it for the patients they take care of.
When the adrenaline from each day begins to wane, we think of our individual families; we worry about relatives far and near. We mourn the loss of loved ones or other hematologists or providers who have succumbed to the COVID-19 virus. We take a minute to think about ourselves and how this pandemic affects us individually and personally. I think about my older sister who runs a smaller hospital in the Bronx, N.Y. She is at the epicenter of the pandemic and is short-staffed in the ICU and medicine floors. Because she is an ob.gyn, she has called me for guidance on a pregnant woman with anemia and sickle cell trait. I hadn’t heard from her in 24 hours and I began to wonder – is she okay? Why didn’t she answer my call this morning? Is she sick? Did she get the information I sent to her linking her with our virtual ICU experts so she can implement a similar program for her hospital?
Next, I think of my younger sister in Long Island, N.Y., who was covering shifts as a hospitalist. She had asked me to send her some hematology tips on managing disseminated intravascular coagulopathy in COVID-19 patients as she has limited access to consultants. She sees an average of seven to nine COVID-19–positive patients and several persons under investigation per shift.
I also think of my 76-year-old mom who is upset that she cannot go to the adult center because of social distancing. So, even though I am weary, I do a FaceTime call with mom. I try to explain why it’s important for her to stay indoors. It’s only temporary, I reassure her; but I cannot say how long “temporary” is.
I pack up my bags, change out of my scrubs to head to my car thinking of my daughter who just turned 21 years old and was so excited about her college graduation in May. She had a meltdown yesterday because her university announced there will be no in-person gradation. I wonder how I can help her see the big picture and yet, why should she? She’s only 21.
Then I get a page – it’s a patient with sickle cell disease – my first COVID-19–positive patient. As I take the history and turn my computer back on to do this consult, I realize that this is what all the preparation was for. The sickle cell guidance document we had worked on over the past weeks to outline a step-by-step approach to managing a SCD patient with COVID-19 that is intentionally aligned with our institutions COVID-19 treatment protocol with specific nuances relevant to patients with red blood cell disorders, was now being put to use. I felt glad for my patient that we were prepared and had a semblance of a plan on how to approach his care.
The battle is far from over. Actually, as of my writing this, it’s just starting in my part of the country. The days will continue to be long. I continue to appreciate the beauty of the human spirit among the people we work with, the hematology community we belong to, and the patients that we serve. I am committed (as are all of you) to staying “on-duty” for as long as I can, and I’d like to take this opportunity to say to all the hematologists out there – “Thank you for your service and for reporting for duty to the front lines.”
Ifeyinwa (Ify) Osunkwo, MD, MPH, is a professor of medicine and the director of the Sickle Cell Disease Enterprise at the Levine Cancer Institute, Atrium Health, Charlotte, N.C. She is the editor in chief of Hematology News.











