User login
FDA panel recommends CBD for pediatric seizure disorders
Advisors to the Food and Drug Administration have found a generally favorable benefit-risk profile for cannabidiol oil solution in the treatment of two forms of severe pediatric seizure disorders. The drug is under expedited review by the agency.
If the FDA supports this recommendation, cannabidiol oral solution would be the first cannabis-based medication approved in the United States.
“At this point, this is a spectacular advance,” said committee member John Mendelson, MD, chief medical officer of Ria Health, San Francisco.
Fellow committee member Mark Green, MD, concurred. “It is clearly an honor to be making a decision based on science and public interest, rather than political discussion,” said Dr. Green, professor of neurology, anesthesiology, and rehabilitation medicine at Icahn School of Medicine, Mt Sinai, New York.
Patients with LGS taking cannabidiol oral solution at 20 mg/kg/day in two clinical trials saw a 42%-44% reduction in drop seizure frequency over a 14-week treatment period (P = .0047 and P = .0135, compared to placebo). A 50% reduction of drop seizure frequency was seen in 40% and 44% of LGS patients in the two clinical trials.
For patients with DS, 20 mg/kg/day of cannabidiol oral solution resulted in a 39% decrease in convulsive seizure frequency during a 14-week treatment period (P less than .05). There was a numeric, but not statistically significant, increase in the number of DS patients who saw a 50% reduction in convulsive seizure frequency on this dose.
Sustained efficacy for both seizure disorders has been seen during an extended open-label follow-on study.
The potential for abuse of the non-psychoactive substance derived from cannabis plants was judged very low. Though animal studies didn’t yield a significant signal for abuse potential with cannabidiol oral solution, the fact that cannabis is currently a schedule I drug prompted the FDA to recommend a human abuse potential study.
From the totality of the studies, “We see little evidence that cannabidiol has meaningful abuse potential, even at supratherapeutic doses in adults,” said Katherine Bonson, PhD, a pharmacologist with the FDA’s Controlled Substance Staff, in the Office of the Center Director of the Center for Drug Evaluation and Research (CDER).
The committee also agreed with the FDA staff that the mild to moderate elevations in liver enzymes seen with cannabidiol oral solution administration can be managed with package labeling and patient monitoring.
Both the FDA and GW Pharmaceuticals, which seeks to market cannabidiol oral solution as Epidiolex, acknowledged that the cannabidiol oral solution was associated with a significant elevation in liver transaminases in some patients. In pooled data, 16.3% of patients taking the drug at the higher dose of 20 mg/kg/day experienced transaminase elevation greater than three times the upper limit of normal, compared with 0.9% of those taking placebo.
Concomitant use of valproic acid was associated with greater likelihood of transaminase elevation. In patients taking both valproic acid and cannabidiol oral solution at the higher dose, 13% experienced transaminase elevations over five times the upper limit of normal.
However, many patients had mild transaminase elevations at baseline, and most cases of transaminase elevation did not require discontinuation of cannabidiol oral solution. Both the sponsor and the FDA agreed that no cases of severe liver injury meeting Hy’s law criteria were seen during the clinical trials; the two cases of “hepatic failure” reported were not associated with elevated bilirubin or international normalized ratio (INR) levels.
The FDA staff clinical reviewer who presented the agency’s overview of liver safety did note one still-unknown factor.
“There are not enough patients exposed to this drug to know whether some might have a smoldering inflammatory response that might potentially – and I can only say potentially – cause a problem down the line,” said Lara Dimick-Santos, MD, of the Division of Gastroenterology and Inborn Errors Products of the Office of Drug Evaluation III, Office of New Drugs.
Advisory committee member Dr. Mendelson asked for long-term monitoring, noting that “we need to watch for long-term safety data because this is a novel drug.”
The FDA’s re-analysis of data from 3 randomized, double-blind, placebo-controlled trials agreed with the efficacy findings reported by GW Pharmaceuticals. Cannabidiol oral solution met its primary endpoint of a reduction in frequency of seizures in LGS and DS patients in all 3 pivotal clinical trials, showing a significant improvement in seizure control when added to standard of care antiepileptic drug therapy for patients with drug-resistant LGS and DS.
The safety evaluation was based on a total of 1,756 patients who were exposed to cannabidiol oral solution, “adequate exposure to allow for assessment of safety,” said Natalie Getzoff, MD, a clinical review in the CDER’s Division of Neurology Products. Though 20 deaths were seen in the study population, “overall, the causes of death were varied and not unexpected for the patient population, and not clearly linked to the drug,” she said. “At this point in our review, we have not identified any obstacles to approval.”
To be included in the clinical trials, LGS patients had to be 2-55 years old, using at least one antiepileptic drug and still having at least 8 drop seizures every 4 weeks and at least two drop seizures weekly. DS patients were aged 2-18 years old, also using at least one antiepileptic drug at baseline and having at least 4 convulsive seizures in a 4 week period.
A total of 235 patients with LGS and 88 patients with DS were enrolled in the clinical trials, and an additional 157 patients with LGS and 209 patients with DS were enrolled in the open-label extension study.
An expanded access program for individuals with refractory epilepsy is ongoing, and 684 patients have been enrolled to date, including 97 LGS patients and 64 DS patients.
The proposed indications for cannabidiol oral solution are for the adjunctive treatment of seizures associated with LGS and DS in patients 2 years of age and older. Initial dosing recommendations are to titrate to a dose of 10 mg/kg/day, with dose adjustments permissible up to 20 mg/kg/day depending on clinical response and tolerability. Cannabidiol received fast track designation in 2014, and rare pediatric designation for LGS and DS in 2017.
The FDA is not obligated to support the recommendations of its advisory committees, though it often does. The agency is slated to take action on cannabidiol oral solution by June.
This article was updated 4/19/18.
Advisors to the Food and Drug Administration have found a generally favorable benefit-risk profile for cannabidiol oil solution in the treatment of two forms of severe pediatric seizure disorders. The drug is under expedited review by the agency.
If the FDA supports this recommendation, cannabidiol oral solution would be the first cannabis-based medication approved in the United States.
“At this point, this is a spectacular advance,” said committee member John Mendelson, MD, chief medical officer of Ria Health, San Francisco.
Fellow committee member Mark Green, MD, concurred. “It is clearly an honor to be making a decision based on science and public interest, rather than political discussion,” said Dr. Green, professor of neurology, anesthesiology, and rehabilitation medicine at Icahn School of Medicine, Mt Sinai, New York.
Patients with LGS taking cannabidiol oral solution at 20 mg/kg/day in two clinical trials saw a 42%-44% reduction in drop seizure frequency over a 14-week treatment period (P = .0047 and P = .0135, compared to placebo). A 50% reduction of drop seizure frequency was seen in 40% and 44% of LGS patients in the two clinical trials.
For patients with DS, 20 mg/kg/day of cannabidiol oral solution resulted in a 39% decrease in convulsive seizure frequency during a 14-week treatment period (P less than .05). There was a numeric, but not statistically significant, increase in the number of DS patients who saw a 50% reduction in convulsive seizure frequency on this dose.
Sustained efficacy for both seizure disorders has been seen during an extended open-label follow-on study.
The potential for abuse of the non-psychoactive substance derived from cannabis plants was judged very low. Though animal studies didn’t yield a significant signal for abuse potential with cannabidiol oral solution, the fact that cannabis is currently a schedule I drug prompted the FDA to recommend a human abuse potential study.
From the totality of the studies, “We see little evidence that cannabidiol has meaningful abuse potential, even at supratherapeutic doses in adults,” said Katherine Bonson, PhD, a pharmacologist with the FDA’s Controlled Substance Staff, in the Office of the Center Director of the Center for Drug Evaluation and Research (CDER).
The committee also agreed with the FDA staff that the mild to moderate elevations in liver enzymes seen with cannabidiol oral solution administration can be managed with package labeling and patient monitoring.
Both the FDA and GW Pharmaceuticals, which seeks to market cannabidiol oral solution as Epidiolex, acknowledged that the cannabidiol oral solution was associated with a significant elevation in liver transaminases in some patients. In pooled data, 16.3% of patients taking the drug at the higher dose of 20 mg/kg/day experienced transaminase elevation greater than three times the upper limit of normal, compared with 0.9% of those taking placebo.
Concomitant use of valproic acid was associated with greater likelihood of transaminase elevation. In patients taking both valproic acid and cannabidiol oral solution at the higher dose, 13% experienced transaminase elevations over five times the upper limit of normal.
However, many patients had mild transaminase elevations at baseline, and most cases of transaminase elevation did not require discontinuation of cannabidiol oral solution. Both the sponsor and the FDA agreed that no cases of severe liver injury meeting Hy’s law criteria were seen during the clinical trials; the two cases of “hepatic failure” reported were not associated with elevated bilirubin or international normalized ratio (INR) levels.
The FDA staff clinical reviewer who presented the agency’s overview of liver safety did note one still-unknown factor.
“There are not enough patients exposed to this drug to know whether some might have a smoldering inflammatory response that might potentially – and I can only say potentially – cause a problem down the line,” said Lara Dimick-Santos, MD, of the Division of Gastroenterology and Inborn Errors Products of the Office of Drug Evaluation III, Office of New Drugs.
Advisory committee member Dr. Mendelson asked for long-term monitoring, noting that “we need to watch for long-term safety data because this is a novel drug.”
The FDA’s re-analysis of data from 3 randomized, double-blind, placebo-controlled trials agreed with the efficacy findings reported by GW Pharmaceuticals. Cannabidiol oral solution met its primary endpoint of a reduction in frequency of seizures in LGS and DS patients in all 3 pivotal clinical trials, showing a significant improvement in seizure control when added to standard of care antiepileptic drug therapy for patients with drug-resistant LGS and DS.
The safety evaluation was based on a total of 1,756 patients who were exposed to cannabidiol oral solution, “adequate exposure to allow for assessment of safety,” said Natalie Getzoff, MD, a clinical review in the CDER’s Division of Neurology Products. Though 20 deaths were seen in the study population, “overall, the causes of death were varied and not unexpected for the patient population, and not clearly linked to the drug,” she said. “At this point in our review, we have not identified any obstacles to approval.”
To be included in the clinical trials, LGS patients had to be 2-55 years old, using at least one antiepileptic drug and still having at least 8 drop seizures every 4 weeks and at least two drop seizures weekly. DS patients were aged 2-18 years old, also using at least one antiepileptic drug at baseline and having at least 4 convulsive seizures in a 4 week period.
A total of 235 patients with LGS and 88 patients with DS were enrolled in the clinical trials, and an additional 157 patients with LGS and 209 patients with DS were enrolled in the open-label extension study.
An expanded access program for individuals with refractory epilepsy is ongoing, and 684 patients have been enrolled to date, including 97 LGS patients and 64 DS patients.
The proposed indications for cannabidiol oral solution are for the adjunctive treatment of seizures associated with LGS and DS in patients 2 years of age and older. Initial dosing recommendations are to titrate to a dose of 10 mg/kg/day, with dose adjustments permissible up to 20 mg/kg/day depending on clinical response and tolerability. Cannabidiol received fast track designation in 2014, and rare pediatric designation for LGS and DS in 2017.
The FDA is not obligated to support the recommendations of its advisory committees, though it often does. The agency is slated to take action on cannabidiol oral solution by June.
This article was updated 4/19/18.
Advisors to the Food and Drug Administration have found a generally favorable benefit-risk profile for cannabidiol oil solution in the treatment of two forms of severe pediatric seizure disorders. The drug is under expedited review by the agency.
If the FDA supports this recommendation, cannabidiol oral solution would be the first cannabis-based medication approved in the United States.
“At this point, this is a spectacular advance,” said committee member John Mendelson, MD, chief medical officer of Ria Health, San Francisco.
Fellow committee member Mark Green, MD, concurred. “It is clearly an honor to be making a decision based on science and public interest, rather than political discussion,” said Dr. Green, professor of neurology, anesthesiology, and rehabilitation medicine at Icahn School of Medicine, Mt Sinai, New York.
Patients with LGS taking cannabidiol oral solution at 20 mg/kg/day in two clinical trials saw a 42%-44% reduction in drop seizure frequency over a 14-week treatment period (P = .0047 and P = .0135, compared to placebo). A 50% reduction of drop seizure frequency was seen in 40% and 44% of LGS patients in the two clinical trials.
For patients with DS, 20 mg/kg/day of cannabidiol oral solution resulted in a 39% decrease in convulsive seizure frequency during a 14-week treatment period (P less than .05). There was a numeric, but not statistically significant, increase in the number of DS patients who saw a 50% reduction in convulsive seizure frequency on this dose.
Sustained efficacy for both seizure disorders has been seen during an extended open-label follow-on study.
The potential for abuse of the non-psychoactive substance derived from cannabis plants was judged very low. Though animal studies didn’t yield a significant signal for abuse potential with cannabidiol oral solution, the fact that cannabis is currently a schedule I drug prompted the FDA to recommend a human abuse potential study.
From the totality of the studies, “We see little evidence that cannabidiol has meaningful abuse potential, even at supratherapeutic doses in adults,” said Katherine Bonson, PhD, a pharmacologist with the FDA’s Controlled Substance Staff, in the Office of the Center Director of the Center for Drug Evaluation and Research (CDER).
The committee also agreed with the FDA staff that the mild to moderate elevations in liver enzymes seen with cannabidiol oral solution administration can be managed with package labeling and patient monitoring.
Both the FDA and GW Pharmaceuticals, which seeks to market cannabidiol oral solution as Epidiolex, acknowledged that the cannabidiol oral solution was associated with a significant elevation in liver transaminases in some patients. In pooled data, 16.3% of patients taking the drug at the higher dose of 20 mg/kg/day experienced transaminase elevation greater than three times the upper limit of normal, compared with 0.9% of those taking placebo.
Concomitant use of valproic acid was associated with greater likelihood of transaminase elevation. In patients taking both valproic acid and cannabidiol oral solution at the higher dose, 13% experienced transaminase elevations over five times the upper limit of normal.
However, many patients had mild transaminase elevations at baseline, and most cases of transaminase elevation did not require discontinuation of cannabidiol oral solution. Both the sponsor and the FDA agreed that no cases of severe liver injury meeting Hy’s law criteria were seen during the clinical trials; the two cases of “hepatic failure” reported were not associated with elevated bilirubin or international normalized ratio (INR) levels.
The FDA staff clinical reviewer who presented the agency’s overview of liver safety did note one still-unknown factor.
“There are not enough patients exposed to this drug to know whether some might have a smoldering inflammatory response that might potentially – and I can only say potentially – cause a problem down the line,” said Lara Dimick-Santos, MD, of the Division of Gastroenterology and Inborn Errors Products of the Office of Drug Evaluation III, Office of New Drugs.
Advisory committee member Dr. Mendelson asked for long-term monitoring, noting that “we need to watch for long-term safety data because this is a novel drug.”
The FDA’s re-analysis of data from 3 randomized, double-blind, placebo-controlled trials agreed with the efficacy findings reported by GW Pharmaceuticals. Cannabidiol oral solution met its primary endpoint of a reduction in frequency of seizures in LGS and DS patients in all 3 pivotal clinical trials, showing a significant improvement in seizure control when added to standard of care antiepileptic drug therapy for patients with drug-resistant LGS and DS.
The safety evaluation was based on a total of 1,756 patients who were exposed to cannabidiol oral solution, “adequate exposure to allow for assessment of safety,” said Natalie Getzoff, MD, a clinical review in the CDER’s Division of Neurology Products. Though 20 deaths were seen in the study population, “overall, the causes of death were varied and not unexpected for the patient population, and not clearly linked to the drug,” she said. “At this point in our review, we have not identified any obstacles to approval.”
To be included in the clinical trials, LGS patients had to be 2-55 years old, using at least one antiepileptic drug and still having at least 8 drop seizures every 4 weeks and at least two drop seizures weekly. DS patients were aged 2-18 years old, also using at least one antiepileptic drug at baseline and having at least 4 convulsive seizures in a 4 week period.
A total of 235 patients with LGS and 88 patients with DS were enrolled in the clinical trials, and an additional 157 patients with LGS and 209 patients with DS were enrolled in the open-label extension study.
An expanded access program for individuals with refractory epilepsy is ongoing, and 684 patients have been enrolled to date, including 97 LGS patients and 64 DS patients.
The proposed indications for cannabidiol oral solution are for the adjunctive treatment of seizures associated with LGS and DS in patients 2 years of age and older. Initial dosing recommendations are to titrate to a dose of 10 mg/kg/day, with dose adjustments permissible up to 20 mg/kg/day depending on clinical response and tolerability. Cannabidiol received fast track designation in 2014, and rare pediatric designation for LGS and DS in 2017.
The FDA is not obligated to support the recommendations of its advisory committees, though it often does. The agency is slated to take action on cannabidiol oral solution by June.
This article was updated 4/19/18.
REPORTING FROM AN FDA ADVISORY COMMITTEE MEETING
FDA approves burosumab to treat X-linked hypophosphatemia
The Food and Drug Administration has approved burosumab to treat X-linked hypophosphatemia, a rare, heritable form of rickets, in adults and children at least 1 year of age. Burosumab received expedited review by the FDA as a breakthrough therapy, and it also has been designated an orphan drug.
X-linked hypophosphatemia (XLH) “differs from other forms of rickets in that vitamin D therapy is not effective. This is the first FDA-approved medication for the treatment of XLH and a real breakthrough for those living with this serious disease,” said Julie Beitz, MD, in an FDA press release announcing the approval of the fully human monoclonal antibody. Dr. Beitz is director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research.
Four clinical trials formed the basis for the FDA’s approval of burosumab. A pivotal randomized, placebo-controlled, double-blind study of adults with XLH that randomized 134 patients 1:1 to subcutaneous burosumab or placebo found that normal phosphorus levels were achieved in 94% of adults receiving burosumab, compared with 8% of those who received placebo.
For children, biweekly burosumab resulted in normal phosphorus levels for 94%-100% of participants. Significant skeletal radiographic improvement also was seen for adults and children taking burosumab.
In both adults and children, burosumab had a good safety profile, with no significant changes in serum or urine levels of calcium or in serum levels of intact parathyroid hormone.
X-linked hypophosphatemia is variable in severity; many patients with XLH will have bone pain, bowed legs, and a waddling gait; enthesopathy with associated joint pain; and dental problems and pain. Many also have short stature and, unlike rickets caused by dietary deficiency and some malabsorption problems, XLH is not improved by conventional vitamin D therapy.
The rare disorder, affecting about 3,000 children and 12,000 adults in the United States, is caused by a mutation in the PHEX gene, with resultant overactivity of fibroblast growth factor 23 (FGF23). Burosumab binds FGF23, increasing renal tubular reabsorption of phosphorus and 1,25-dihydroxycholecalciferol, also called calcitriol.
Previously, XLH had been treated by frequent oral supplementation with phosphate and calcitriol. Burosumab will be marketed by Ultragenyx Pharmaceutical as Crysvita. It is given as a once-monthly or biweekly subcutaneous injection.
The Food and Drug Administration has approved burosumab to treat X-linked hypophosphatemia, a rare, heritable form of rickets, in adults and children at least 1 year of age. Burosumab received expedited review by the FDA as a breakthrough therapy, and it also has been designated an orphan drug.
X-linked hypophosphatemia (XLH) “differs from other forms of rickets in that vitamin D therapy is not effective. This is the first FDA-approved medication for the treatment of XLH and a real breakthrough for those living with this serious disease,” said Julie Beitz, MD, in an FDA press release announcing the approval of the fully human monoclonal antibody. Dr. Beitz is director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research.
Four clinical trials formed the basis for the FDA’s approval of burosumab. A pivotal randomized, placebo-controlled, double-blind study of adults with XLH that randomized 134 patients 1:1 to subcutaneous burosumab or placebo found that normal phosphorus levels were achieved in 94% of adults receiving burosumab, compared with 8% of those who received placebo.
For children, biweekly burosumab resulted in normal phosphorus levels for 94%-100% of participants. Significant skeletal radiographic improvement also was seen for adults and children taking burosumab.
In both adults and children, burosumab had a good safety profile, with no significant changes in serum or urine levels of calcium or in serum levels of intact parathyroid hormone.
X-linked hypophosphatemia is variable in severity; many patients with XLH will have bone pain, bowed legs, and a waddling gait; enthesopathy with associated joint pain; and dental problems and pain. Many also have short stature and, unlike rickets caused by dietary deficiency and some malabsorption problems, XLH is not improved by conventional vitamin D therapy.
The rare disorder, affecting about 3,000 children and 12,000 adults in the United States, is caused by a mutation in the PHEX gene, with resultant overactivity of fibroblast growth factor 23 (FGF23). Burosumab binds FGF23, increasing renal tubular reabsorption of phosphorus and 1,25-dihydroxycholecalciferol, also called calcitriol.
Previously, XLH had been treated by frequent oral supplementation with phosphate and calcitriol. Burosumab will be marketed by Ultragenyx Pharmaceutical as Crysvita. It is given as a once-monthly or biweekly subcutaneous injection.
The Food and Drug Administration has approved burosumab to treat X-linked hypophosphatemia, a rare, heritable form of rickets, in adults and children at least 1 year of age. Burosumab received expedited review by the FDA as a breakthrough therapy, and it also has been designated an orphan drug.
X-linked hypophosphatemia (XLH) “differs from other forms of rickets in that vitamin D therapy is not effective. This is the first FDA-approved medication for the treatment of XLH and a real breakthrough for those living with this serious disease,” said Julie Beitz, MD, in an FDA press release announcing the approval of the fully human monoclonal antibody. Dr. Beitz is director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research.
Four clinical trials formed the basis for the FDA’s approval of burosumab. A pivotal randomized, placebo-controlled, double-blind study of adults with XLH that randomized 134 patients 1:1 to subcutaneous burosumab or placebo found that normal phosphorus levels were achieved in 94% of adults receiving burosumab, compared with 8% of those who received placebo.
For children, biweekly burosumab resulted in normal phosphorus levels for 94%-100% of participants. Significant skeletal radiographic improvement also was seen for adults and children taking burosumab.
In both adults and children, burosumab had a good safety profile, with no significant changes in serum or urine levels of calcium or in serum levels of intact parathyroid hormone.
X-linked hypophosphatemia is variable in severity; many patients with XLH will have bone pain, bowed legs, and a waddling gait; enthesopathy with associated joint pain; and dental problems and pain. Many also have short stature and, unlike rickets caused by dietary deficiency and some malabsorption problems, XLH is not improved by conventional vitamin D therapy.
The rare disorder, affecting about 3,000 children and 12,000 adults in the United States, is caused by a mutation in the PHEX gene, with resultant overactivity of fibroblast growth factor 23 (FGF23). Burosumab binds FGF23, increasing renal tubular reabsorption of phosphorus and 1,25-dihydroxycholecalciferol, also called calcitriol.
Previously, XLH had been treated by frequent oral supplementation with phosphate and calcitriol. Burosumab will be marketed by Ultragenyx Pharmaceutical as Crysvita. It is given as a once-monthly or biweekly subcutaneous injection.
FDA approves immunotherapy combo for advanced RCC
The Food and Drug Administration has granted approvals to
The approvals were based on statistically significant improvements in overall survival (OS) and objective response rate (ORR) for patients receiving the combination of nivolumab and ipilimumab (n = 425), compared with those receiving sunitinib (n = 422) in CheckMate 214, the FDA said in a press statement.
Median OS was not yet reached in the combination arm at follow-up of 32 months, compared with 25.9 months in the sunitinib arm (hazard ratio, 0.63; 95% confidence interval, 0.44-0.89; P less than .0001). The ORR was 41.6% (95% CI, 36.9-46.5) for the combination versus 26.5% (95% CI, 22.4-31) in the sunitinib arm (P less than .0001).
Efficacy of the combination was not established for patients with favorable-risk disease.
The most common adverse reactions were fatigue, rash, diarrhea, musculoskeletal pain, pruritus, nausea, cough, pyrexia, arthralgia, and decreased appetite.
The recommended schedule and dose is 3 mg/kg nivolumab, followed by 1 mg/kg ipilimumab, on the same day every 3 weeks for four doses, then 240 mg nivolumab every 2 weeks or 480 mg every 4 weeks, the FDA said.
Nivolumab is marketed as Opdivo and ipilimumab as Yervoy by Bristol-Myers Squibb.
The Food and Drug Administration has granted approvals to
The approvals were based on statistically significant improvements in overall survival (OS) and objective response rate (ORR) for patients receiving the combination of nivolumab and ipilimumab (n = 425), compared with those receiving sunitinib (n = 422) in CheckMate 214, the FDA said in a press statement.
Median OS was not yet reached in the combination arm at follow-up of 32 months, compared with 25.9 months in the sunitinib arm (hazard ratio, 0.63; 95% confidence interval, 0.44-0.89; P less than .0001). The ORR was 41.6% (95% CI, 36.9-46.5) for the combination versus 26.5% (95% CI, 22.4-31) in the sunitinib arm (P less than .0001).
Efficacy of the combination was not established for patients with favorable-risk disease.
The most common adverse reactions were fatigue, rash, diarrhea, musculoskeletal pain, pruritus, nausea, cough, pyrexia, arthralgia, and decreased appetite.
The recommended schedule and dose is 3 mg/kg nivolumab, followed by 1 mg/kg ipilimumab, on the same day every 3 weeks for four doses, then 240 mg nivolumab every 2 weeks or 480 mg every 4 weeks, the FDA said.
Nivolumab is marketed as Opdivo and ipilimumab as Yervoy by Bristol-Myers Squibb.
The Food and Drug Administration has granted approvals to
The approvals were based on statistically significant improvements in overall survival (OS) and objective response rate (ORR) for patients receiving the combination of nivolumab and ipilimumab (n = 425), compared with those receiving sunitinib (n = 422) in CheckMate 214, the FDA said in a press statement.
Median OS was not yet reached in the combination arm at follow-up of 32 months, compared with 25.9 months in the sunitinib arm (hazard ratio, 0.63; 95% confidence interval, 0.44-0.89; P less than .0001). The ORR was 41.6% (95% CI, 36.9-46.5) for the combination versus 26.5% (95% CI, 22.4-31) in the sunitinib arm (P less than .0001).
Efficacy of the combination was not established for patients with favorable-risk disease.
The most common adverse reactions were fatigue, rash, diarrhea, musculoskeletal pain, pruritus, nausea, cough, pyrexia, arthralgia, and decreased appetite.
The recommended schedule and dose is 3 mg/kg nivolumab, followed by 1 mg/kg ipilimumab, on the same day every 3 weeks for four doses, then 240 mg nivolumab every 2 weeks or 480 mg every 4 weeks, the FDA said.
Nivolumab is marketed as Opdivo and ipilimumab as Yervoy by Bristol-Myers Squibb.
CDC: Marijuana use may spur industries to rethink current policies
according to a report from the Centers for Disease Control and Prevention.
As legal recreational marijuana use continues to expand across the United States, marijuana has been shown to inhibit certain motor skills, which has made it crucial for employers to have a better understanding how best to approach safety training, according to the study published in the Morbidity and Mortality Weekly Report.
“We have been looking at some of the behavioral risk factors associated with marijuana legalization and were interested in the data broken down by industry and occupation, which could help employers make decisions on any kind of safety and drug use policies in the workplace,” lead author Roberta Smith, RN, occupational health program manager at the Colorado Department of Public Health and Environment, said in a interview. “This doesn’t necessarily imply any impairment on the job, but these data will reinforce current policies and encourage employers to go back and see how their work places operate and make sure their employees are good to staff.”
To examine current marijuana use by working adults and the industries and occupations in which they are employed, the Colorado Department of Public Health and Environment analyzed data from the state’s Behavioral Risk Factor Surveillance System regarding current marijuana use (at least 1 day during the preceding 30 days) among 10,169 persons who had responded to the current marijuana use question.
Participants were over the age of 21 years old and were either employed at the time of the survey or had been unemployed for less than a year.
In the overall population, 14.6% reported using marijuana, with higher prevalence in men (17.2%) and those 18-25 years old (29.6%).
By industry, accommodation and food service workers reported the highest rate of use at 30.1%, followed by those in the arts, entertainment, and recreation industry with 28.3%.
While the highest percentage of reported users came from food services and entertainment, safety-sensitive jobs like construction saw rates as high as 20% when not adjusted for age, according to investigators,
Ms. Smith and her colleagues found use varied across safety-sensitive industries, with high rates in construction (19.7%), waste management (18.8%), and manufacturing (16.3%) that were above the total population prevalence. Meanwhile, mining, health care, and transportation were all 10% or lower, which may be because of more regular drug testing.
“It might be reassuring that our health care professionals are on the lower end of use,” said Ms. Smith. “Having worked in a medical facility, I know drug policies for workers are clear and employees are aware of drug testing and when it will occur.”
While the health care industry reported low usage, 15.8% of health care support management workers, such as x-ray technicians, reported marijuana use.
When adjusted for age, the prevalence among workers in certain industries – such as food services, arts, and construction industry – saw significant decreases, which lead investigators to conclude younger employees would be a key target for more marijuana-related drug-use policies.
Ms. Smith and her colleagues recognized the population used may not be a full representation of all Colorado employees and that missing data regarding how often individuals used marijuana within 30 days could offer different considerations for workplace impairment.
Investigators also noted the data may have been influenced by self-reported bias or recording errors by survey takers.
Moving forward, Ms. Smith and her colleagues are interested in how this data might shift as more states conduct their own research and as marijuana policy changes.
This report was funded by the CDC, and investigators report no relevant financial disclosures.
SOURCE: Smith R et al. MMWR. 2018 Apr 13;67(14):409-13.
according to a report from the Centers for Disease Control and Prevention.
As legal recreational marijuana use continues to expand across the United States, marijuana has been shown to inhibit certain motor skills, which has made it crucial for employers to have a better understanding how best to approach safety training, according to the study published in the Morbidity and Mortality Weekly Report.
“We have been looking at some of the behavioral risk factors associated with marijuana legalization and were interested in the data broken down by industry and occupation, which could help employers make decisions on any kind of safety and drug use policies in the workplace,” lead author Roberta Smith, RN, occupational health program manager at the Colorado Department of Public Health and Environment, said in a interview. “This doesn’t necessarily imply any impairment on the job, but these data will reinforce current policies and encourage employers to go back and see how their work places operate and make sure their employees are good to staff.”
To examine current marijuana use by working adults and the industries and occupations in which they are employed, the Colorado Department of Public Health and Environment analyzed data from the state’s Behavioral Risk Factor Surveillance System regarding current marijuana use (at least 1 day during the preceding 30 days) among 10,169 persons who had responded to the current marijuana use question.
Participants were over the age of 21 years old and were either employed at the time of the survey or had been unemployed for less than a year.
In the overall population, 14.6% reported using marijuana, with higher prevalence in men (17.2%) and those 18-25 years old (29.6%).
By industry, accommodation and food service workers reported the highest rate of use at 30.1%, followed by those in the arts, entertainment, and recreation industry with 28.3%.
While the highest percentage of reported users came from food services and entertainment, safety-sensitive jobs like construction saw rates as high as 20% when not adjusted for age, according to investigators,
Ms. Smith and her colleagues found use varied across safety-sensitive industries, with high rates in construction (19.7%), waste management (18.8%), and manufacturing (16.3%) that were above the total population prevalence. Meanwhile, mining, health care, and transportation were all 10% or lower, which may be because of more regular drug testing.
“It might be reassuring that our health care professionals are on the lower end of use,” said Ms. Smith. “Having worked in a medical facility, I know drug policies for workers are clear and employees are aware of drug testing and when it will occur.”
While the health care industry reported low usage, 15.8% of health care support management workers, such as x-ray technicians, reported marijuana use.
When adjusted for age, the prevalence among workers in certain industries – such as food services, arts, and construction industry – saw significant decreases, which lead investigators to conclude younger employees would be a key target for more marijuana-related drug-use policies.
Ms. Smith and her colleagues recognized the population used may not be a full representation of all Colorado employees and that missing data regarding how often individuals used marijuana within 30 days could offer different considerations for workplace impairment.
Investigators also noted the data may have been influenced by self-reported bias or recording errors by survey takers.
Moving forward, Ms. Smith and her colleagues are interested in how this data might shift as more states conduct their own research and as marijuana policy changes.
This report was funded by the CDC, and investigators report no relevant financial disclosures.
SOURCE: Smith R et al. MMWR. 2018 Apr 13;67(14):409-13.
according to a report from the Centers for Disease Control and Prevention.
As legal recreational marijuana use continues to expand across the United States, marijuana has been shown to inhibit certain motor skills, which has made it crucial for employers to have a better understanding how best to approach safety training, according to the study published in the Morbidity and Mortality Weekly Report.
“We have been looking at some of the behavioral risk factors associated with marijuana legalization and were interested in the data broken down by industry and occupation, which could help employers make decisions on any kind of safety and drug use policies in the workplace,” lead author Roberta Smith, RN, occupational health program manager at the Colorado Department of Public Health and Environment, said in a interview. “This doesn’t necessarily imply any impairment on the job, but these data will reinforce current policies and encourage employers to go back and see how their work places operate and make sure their employees are good to staff.”
To examine current marijuana use by working adults and the industries and occupations in which they are employed, the Colorado Department of Public Health and Environment analyzed data from the state’s Behavioral Risk Factor Surveillance System regarding current marijuana use (at least 1 day during the preceding 30 days) among 10,169 persons who had responded to the current marijuana use question.
Participants were over the age of 21 years old and were either employed at the time of the survey or had been unemployed for less than a year.
In the overall population, 14.6% reported using marijuana, with higher prevalence in men (17.2%) and those 18-25 years old (29.6%).
By industry, accommodation and food service workers reported the highest rate of use at 30.1%, followed by those in the arts, entertainment, and recreation industry with 28.3%.
While the highest percentage of reported users came from food services and entertainment, safety-sensitive jobs like construction saw rates as high as 20% when not adjusted for age, according to investigators,
Ms. Smith and her colleagues found use varied across safety-sensitive industries, with high rates in construction (19.7%), waste management (18.8%), and manufacturing (16.3%) that were above the total population prevalence. Meanwhile, mining, health care, and transportation were all 10% or lower, which may be because of more regular drug testing.
“It might be reassuring that our health care professionals are on the lower end of use,” said Ms. Smith. “Having worked in a medical facility, I know drug policies for workers are clear and employees are aware of drug testing and when it will occur.”
While the health care industry reported low usage, 15.8% of health care support management workers, such as x-ray technicians, reported marijuana use.
When adjusted for age, the prevalence among workers in certain industries – such as food services, arts, and construction industry – saw significant decreases, which lead investigators to conclude younger employees would be a key target for more marijuana-related drug-use policies.
Ms. Smith and her colleagues recognized the population used may not be a full representation of all Colorado employees and that missing data regarding how often individuals used marijuana within 30 days could offer different considerations for workplace impairment.
Investigators also noted the data may have been influenced by self-reported bias or recording errors by survey takers.
Moving forward, Ms. Smith and her colleagues are interested in how this data might shift as more states conduct their own research and as marijuana policy changes.
This report was funded by the CDC, and investigators report no relevant financial disclosures.
SOURCE: Smith R et al. MMWR. 2018 Apr 13;67(14):409-13.
FROM MMWR
Complication rates rise after decline in uterine fibroid morcellation
The rate of major and minor 30-day complications from the treatment of uterine fibroids has increased significantly since the Food and Drug Administration’s black-box warning against the use of power morcellation, data suggest.
Researchers examined the incidence of 30-day posthysterectomy complications in 75,487 women who underwent treatment for benign gynecologic indications before and after November 2014, when the FDA’s edict was issued over concerns about the risk of disseminating benign or malignant disease. Of these women, 25,571 had uterine fibroids as the indication for hysterectomy.
The retrospective cohort study, published online April 11 in JAMA Surgery, showed that while the overall rate of complications in the cohort was relatively unchanged before and after the FDA’s warning, complication rates increased significantly in women undergoing treatment for uterine fibroids.
Before the FDA’s announcement, the 30-day major complication rate in women undergoing hysterectomy for uterine fibroids was 1.9%, which increased to 2.4% after the FDA’s warning (odds ratio, 1.23; 95% confidence interval, 1.04-1.47; P = .02). Similarly, the rate of minor 30-day complications increased from 2.7% before the warning to 3.3% after the warning (OR, 1.21; 95% CI, 1.04-1.40; P = .01), after adjustment for factors such as age, body mass index, comorbidities, and other associated procedures.
“This 20% increase in the odds of major and minor complications could translate into a large number of additional complications among the 200,000 hysterectomies performed annually for uterine fibroids in the United States,” wrote Francesco Multinu, MD, of the department of obstetrics and gynecology at the Mayo Clinic, Rochester, Minn., and his coauthors.
Overall, the researchers saw a much higher rate of major complications in women undergoing open abdominal surgery, compared with women who underwent minimally invasive surgery or vaginal hysterectomy (3.5% vs. 1.7% vs. 1.7%). A similar pattern was seen in the subgroup of women who underwent hysterectomy for uterine fibroids (2.8% vs. 1.8% vs. 1.8%).
However, minor 30-day complication rates were higher in women who underwent vaginal hysterectomy (4.5%), compared with open abdominal surgery (4.1%) and minimally invasive surgery (3.2%). In women with uterine fibroids, the minor complication rates were slightly higher in those who underwent open hysterectomy or vaginal hysterectomy than in those who had minimally invasive surgery, but this was not statistically significant.
The type of surgery in women with uterine fibroids changed significantly after the FDA announcement. Before the black-box warning against power morcellation was issued, 37.2% of hysterectomy procedures for uterine fibroids were open, 56.1% were minimally invasive, and 6.7% were vaginal. After the announcement, the percentage that were open procedures increased to 43%, minimally invasive procedures decreased to 49.7%, and vaginal hysterectomies increased to 7.3% (P less than .001).
A similar but less pronounced trend was seen across all hysterectomies for benign gynecological indications.
The authors noted that the study analyzed data on 30-day complications, so they weren’t able to draw conclusions about longer-term outcomes.
“Although caution is required to avoid morcellation of unexpected uterine malignant neoplasms, our results should be considered by women and clinicians during the process of shared decision making and by medical societies and regulatory bodies when issuing safety communications,” the authors wrote.
The study was supported by a grant from the National Center for Advancing Translational Sciences, and one author was supported by the University of Insubria, and by Fondo Miglierina, Varese, Italy. No conflicts of interest were declared.
SOURCE: Multinu F et al. JAMA Surg. 2018 Apr 11. doi: 10.1001/jamasurg.2018.0141.
The rate of major and minor 30-day complications from the treatment of uterine fibroids has increased significantly since the Food and Drug Administration’s black-box warning against the use of power morcellation, data suggest.
Researchers examined the incidence of 30-day posthysterectomy complications in 75,487 women who underwent treatment for benign gynecologic indications before and after November 2014, when the FDA’s edict was issued over concerns about the risk of disseminating benign or malignant disease. Of these women, 25,571 had uterine fibroids as the indication for hysterectomy.
The retrospective cohort study, published online April 11 in JAMA Surgery, showed that while the overall rate of complications in the cohort was relatively unchanged before and after the FDA’s warning, complication rates increased significantly in women undergoing treatment for uterine fibroids.
Before the FDA’s announcement, the 30-day major complication rate in women undergoing hysterectomy for uterine fibroids was 1.9%, which increased to 2.4% after the FDA’s warning (odds ratio, 1.23; 95% confidence interval, 1.04-1.47; P = .02). Similarly, the rate of minor 30-day complications increased from 2.7% before the warning to 3.3% after the warning (OR, 1.21; 95% CI, 1.04-1.40; P = .01), after adjustment for factors such as age, body mass index, comorbidities, and other associated procedures.
“This 20% increase in the odds of major and minor complications could translate into a large number of additional complications among the 200,000 hysterectomies performed annually for uterine fibroids in the United States,” wrote Francesco Multinu, MD, of the department of obstetrics and gynecology at the Mayo Clinic, Rochester, Minn., and his coauthors.
Overall, the researchers saw a much higher rate of major complications in women undergoing open abdominal surgery, compared with women who underwent minimally invasive surgery or vaginal hysterectomy (3.5% vs. 1.7% vs. 1.7%). A similar pattern was seen in the subgroup of women who underwent hysterectomy for uterine fibroids (2.8% vs. 1.8% vs. 1.8%).
However, minor 30-day complication rates were higher in women who underwent vaginal hysterectomy (4.5%), compared with open abdominal surgery (4.1%) and minimally invasive surgery (3.2%). In women with uterine fibroids, the minor complication rates were slightly higher in those who underwent open hysterectomy or vaginal hysterectomy than in those who had minimally invasive surgery, but this was not statistically significant.
The type of surgery in women with uterine fibroids changed significantly after the FDA announcement. Before the black-box warning against power morcellation was issued, 37.2% of hysterectomy procedures for uterine fibroids were open, 56.1% were minimally invasive, and 6.7% were vaginal. After the announcement, the percentage that were open procedures increased to 43%, minimally invasive procedures decreased to 49.7%, and vaginal hysterectomies increased to 7.3% (P less than .001).
A similar but less pronounced trend was seen across all hysterectomies for benign gynecological indications.
The authors noted that the study analyzed data on 30-day complications, so they weren’t able to draw conclusions about longer-term outcomes.
“Although caution is required to avoid morcellation of unexpected uterine malignant neoplasms, our results should be considered by women and clinicians during the process of shared decision making and by medical societies and regulatory bodies when issuing safety communications,” the authors wrote.
The study was supported by a grant from the National Center for Advancing Translational Sciences, and one author was supported by the University of Insubria, and by Fondo Miglierina, Varese, Italy. No conflicts of interest were declared.
SOURCE: Multinu F et al. JAMA Surg. 2018 Apr 11. doi: 10.1001/jamasurg.2018.0141.
The rate of major and minor 30-day complications from the treatment of uterine fibroids has increased significantly since the Food and Drug Administration’s black-box warning against the use of power morcellation, data suggest.
Researchers examined the incidence of 30-day posthysterectomy complications in 75,487 women who underwent treatment for benign gynecologic indications before and after November 2014, when the FDA’s edict was issued over concerns about the risk of disseminating benign or malignant disease. Of these women, 25,571 had uterine fibroids as the indication for hysterectomy.
The retrospective cohort study, published online April 11 in JAMA Surgery, showed that while the overall rate of complications in the cohort was relatively unchanged before and after the FDA’s warning, complication rates increased significantly in women undergoing treatment for uterine fibroids.
Before the FDA’s announcement, the 30-day major complication rate in women undergoing hysterectomy for uterine fibroids was 1.9%, which increased to 2.4% after the FDA’s warning (odds ratio, 1.23; 95% confidence interval, 1.04-1.47; P = .02). Similarly, the rate of minor 30-day complications increased from 2.7% before the warning to 3.3% after the warning (OR, 1.21; 95% CI, 1.04-1.40; P = .01), after adjustment for factors such as age, body mass index, comorbidities, and other associated procedures.
“This 20% increase in the odds of major and minor complications could translate into a large number of additional complications among the 200,000 hysterectomies performed annually for uterine fibroids in the United States,” wrote Francesco Multinu, MD, of the department of obstetrics and gynecology at the Mayo Clinic, Rochester, Minn., and his coauthors.
Overall, the researchers saw a much higher rate of major complications in women undergoing open abdominal surgery, compared with women who underwent minimally invasive surgery or vaginal hysterectomy (3.5% vs. 1.7% vs. 1.7%). A similar pattern was seen in the subgroup of women who underwent hysterectomy for uterine fibroids (2.8% vs. 1.8% vs. 1.8%).
However, minor 30-day complication rates were higher in women who underwent vaginal hysterectomy (4.5%), compared with open abdominal surgery (4.1%) and minimally invasive surgery (3.2%). In women with uterine fibroids, the minor complication rates were slightly higher in those who underwent open hysterectomy or vaginal hysterectomy than in those who had minimally invasive surgery, but this was not statistically significant.
The type of surgery in women with uterine fibroids changed significantly after the FDA announcement. Before the black-box warning against power morcellation was issued, 37.2% of hysterectomy procedures for uterine fibroids were open, 56.1% were minimally invasive, and 6.7% were vaginal. After the announcement, the percentage that were open procedures increased to 43%, minimally invasive procedures decreased to 49.7%, and vaginal hysterectomies increased to 7.3% (P less than .001).
A similar but less pronounced trend was seen across all hysterectomies for benign gynecological indications.
The authors noted that the study analyzed data on 30-day complications, so they weren’t able to draw conclusions about longer-term outcomes.
“Although caution is required to avoid morcellation of unexpected uterine malignant neoplasms, our results should be considered by women and clinicians during the process of shared decision making and by medical societies and regulatory bodies when issuing safety communications,” the authors wrote.
The study was supported by a grant from the National Center for Advancing Translational Sciences, and one author was supported by the University of Insubria, and by Fondo Miglierina, Varese, Italy. No conflicts of interest were declared.
SOURCE: Multinu F et al. JAMA Surg. 2018 Apr 11. doi: 10.1001/jamasurg.2018.0141.
FROM JAMA SURGERY
Key clinical point: Major complications from uterine fibroid hysterectomies have increased with more open surgeries.
Major finding: The rate of 30-day complications after uterine fibroid hysterectomy has increased by 20%.
Study details: A retrospective cohort study of 75,487 women.
Disclosures: The study was supported by a grant from the National Center for Advancing Translational Sciences, a component of the National Institutes of Health, and one author was supported by the University of Insubria, and by Fondo Miglierina, Varese, Italy. No conflicts of interest were declared.
Source: Multinu F et al. JAMA Surg. 2018 Apr 11. doi: 10.1001/jamasurg.2018.0141.
FDA approves marketing for retinal imaging device that uses artificial intelligence
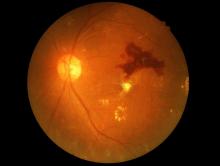
The Food and Drug Administration has permitted the marketing of IDx-DR, a retinal imaging device that uses artificial intelligence (AI) to detect greater than a mild level of diabetic retinopathy in adult patients with diabetes. It also is the first authorized device that provides a screening tool without the need of an eye care specialist, making it ideal for health care providers who do not specialize in eye care.
“Early detection of retinopathy is an important part of managing care for the millions of people with diabetes, yet many patients with diabetes are not adequately screened for diabetic retinopathy.” About 50% of people with diabetes do not have the recommended annual retinopathy screening exam, Malvina Eydelman, MD, director of the division of ophthalmic, and ear, nose, and throat devices at the FDA’s Center for Devices and Radiological Health noted in a press release.
The software behind IDx-DR utilizes an artificial intelligence program to analyze retinal images captured by a retinal camera, the Topcon NW400. After one or more images are taken, they are uploaded to a Cloud server, on which IDx-DR software is installed, where they can be analyzed using an algorithm. In cases in which the images are of sufficient quality, the software provides the doctor with one of two results: “More than mild diabetic retinopathy detected: refer to an eye care professional” or “Negative for more than mild diabetic retinopathy; rescreen in 12 months.”
If a positive result is detected, patients should see an eye care specialist for further diagnostic evaluation and treatment as soon as possible.
The FDA reviewed data obtained from a clinical study prior to approving IDx-DR for marketing. The study looked at retinal images from 900 patients at 10 primary care sites. The aim of the study was to determine how often IDx-DR was correct in identifying mild diabetic retinopathy. The device was accurate nearly 90% of the time, correctly identifying mild diabetic retinopathy 87.4% of the time. It was also able to identify correctly patients who did not have mild diabetic retinopathy 89.5% of the time.
IDx-DR was approved via the FDA’s De Novo premarket review pathway, which offers a way to approve novel, low to moderate risk devices for which there are no similar devices previously approved. It also was granted a Breakthrough Device designation, because of its effectiveness in treating or diagnosing a irreversibly debilitating disease or condition.
Michael Abramoff, MD, PhD, and founder and president of IDx, said in an interview: “The FDA’s authorization to market IDx-DR is a historic moment that will launch a transformation in the way U.S. health care is delivered. Autonomous AI systems have massive potential to improve health care productivity, lower health care costs, and improve accessibility and quality. As the first of its kind to be authorized for commercialization, IDx-DR provides a roadmap for the safe and responsible use of AI in medicine.”
IDx-DR is intended to identify mild diabetic retinopathy and should not be used to detect rapidly progressive cases of diabetic retinopathy, particularly in pregnant women in which the disease can progress quickly. Patients with a history of laser treatment, surgery, or injections in the eye and other types of retinopathy, including radiation retinopathy, should not use IDx-DR.
For more information on other uses of AI in the treatment of diabetic disorders, click here.
The Food and Drug Administration has permitted the marketing of IDx-DR, a retinal imaging device that uses artificial intelligence (AI) to detect greater than a mild level of diabetic retinopathy in adult patients with diabetes. It also is the first authorized device that provides a screening tool without the need of an eye care specialist, making it ideal for health care providers who do not specialize in eye care.
“Early detection of retinopathy is an important part of managing care for the millions of people with diabetes, yet many patients with diabetes are not adequately screened for diabetic retinopathy.” About 50% of people with diabetes do not have the recommended annual retinopathy screening exam, Malvina Eydelman, MD, director of the division of ophthalmic, and ear, nose, and throat devices at the FDA’s Center for Devices and Radiological Health noted in a press release.
The software behind IDx-DR utilizes an artificial intelligence program to analyze retinal images captured by a retinal camera, the Topcon NW400. After one or more images are taken, they are uploaded to a Cloud server, on which IDx-DR software is installed, where they can be analyzed using an algorithm. In cases in which the images are of sufficient quality, the software provides the doctor with one of two results: “More than mild diabetic retinopathy detected: refer to an eye care professional” or “Negative for more than mild diabetic retinopathy; rescreen in 12 months.”
If a positive result is detected, patients should see an eye care specialist for further diagnostic evaluation and treatment as soon as possible.
The FDA reviewed data obtained from a clinical study prior to approving IDx-DR for marketing. The study looked at retinal images from 900 patients at 10 primary care sites. The aim of the study was to determine how often IDx-DR was correct in identifying mild diabetic retinopathy. The device was accurate nearly 90% of the time, correctly identifying mild diabetic retinopathy 87.4% of the time. It was also able to identify correctly patients who did not have mild diabetic retinopathy 89.5% of the time.
IDx-DR was approved via the FDA’s De Novo premarket review pathway, which offers a way to approve novel, low to moderate risk devices for which there are no similar devices previously approved. It also was granted a Breakthrough Device designation, because of its effectiveness in treating or diagnosing a irreversibly debilitating disease or condition.
Michael Abramoff, MD, PhD, and founder and president of IDx, said in an interview: “The FDA’s authorization to market IDx-DR is a historic moment that will launch a transformation in the way U.S. health care is delivered. Autonomous AI systems have massive potential to improve health care productivity, lower health care costs, and improve accessibility and quality. As the first of its kind to be authorized for commercialization, IDx-DR provides a roadmap for the safe and responsible use of AI in medicine.”
IDx-DR is intended to identify mild diabetic retinopathy and should not be used to detect rapidly progressive cases of diabetic retinopathy, particularly in pregnant women in which the disease can progress quickly. Patients with a history of laser treatment, surgery, or injections in the eye and other types of retinopathy, including radiation retinopathy, should not use IDx-DR.
For more information on other uses of AI in the treatment of diabetic disorders, click here.
The Food and Drug Administration has permitted the marketing of IDx-DR, a retinal imaging device that uses artificial intelligence (AI) to detect greater than a mild level of diabetic retinopathy in adult patients with diabetes. It also is the first authorized device that provides a screening tool without the need of an eye care specialist, making it ideal for health care providers who do not specialize in eye care.
“Early detection of retinopathy is an important part of managing care for the millions of people with diabetes, yet many patients with diabetes are not adequately screened for diabetic retinopathy.” About 50% of people with diabetes do not have the recommended annual retinopathy screening exam, Malvina Eydelman, MD, director of the division of ophthalmic, and ear, nose, and throat devices at the FDA’s Center for Devices and Radiological Health noted in a press release.
The software behind IDx-DR utilizes an artificial intelligence program to analyze retinal images captured by a retinal camera, the Topcon NW400. After one or more images are taken, they are uploaded to a Cloud server, on which IDx-DR software is installed, where they can be analyzed using an algorithm. In cases in which the images are of sufficient quality, the software provides the doctor with one of two results: “More than mild diabetic retinopathy detected: refer to an eye care professional” or “Negative for more than mild diabetic retinopathy; rescreen in 12 months.”
If a positive result is detected, patients should see an eye care specialist for further diagnostic evaluation and treatment as soon as possible.
The FDA reviewed data obtained from a clinical study prior to approving IDx-DR for marketing. The study looked at retinal images from 900 patients at 10 primary care sites. The aim of the study was to determine how often IDx-DR was correct in identifying mild diabetic retinopathy. The device was accurate nearly 90% of the time, correctly identifying mild diabetic retinopathy 87.4% of the time. It was also able to identify correctly patients who did not have mild diabetic retinopathy 89.5% of the time.
IDx-DR was approved via the FDA’s De Novo premarket review pathway, which offers a way to approve novel, low to moderate risk devices for which there are no similar devices previously approved. It also was granted a Breakthrough Device designation, because of its effectiveness in treating or diagnosing a irreversibly debilitating disease or condition.
Michael Abramoff, MD, PhD, and founder and president of IDx, said in an interview: “The FDA’s authorization to market IDx-DR is a historic moment that will launch a transformation in the way U.S. health care is delivered. Autonomous AI systems have massive potential to improve health care productivity, lower health care costs, and improve accessibility and quality. As the first of its kind to be authorized for commercialization, IDx-DR provides a roadmap for the safe and responsible use of AI in medicine.”
IDx-DR is intended to identify mild diabetic retinopathy and should not be used to detect rapidly progressive cases of diabetic retinopathy, particularly in pregnant women in which the disease can progress quickly. Patients with a history of laser treatment, surgery, or injections in the eye and other types of retinopathy, including radiation retinopathy, should not use IDx-DR.
For more information on other uses of AI in the treatment of diabetic disorders, click here.
FDA orders new restrictions on sale, distribution of Essure
The Food and Drug Administration has restricted the sale and distribution of Bayer’s Essure permanent contraception device to only health care providers and facilities that provide patients with information about the risks and benefits of the device.
In an order issued April 9, the FDA’s Center for Devices and Radiological Health informed Bayer of the restrictions, citing a need to “provide reasonable assurance of the safety and effectiveness of the device.”
Bayer must implement the restrictions immediately and must ensure health care provider compliance.
“The FDA is taking this step after becoming aware that some women were not being adequately informed of Essure’s risks before getting the device implanted, despite previous significant efforts to educate patients and doctors about the risks associated with this device,” the agency said in a press release, adding that it “plans to enforce these requirements and will take appropriate action for a failure to comply, including applicable criminal and civil penalties.”
Essure, which is the only permanently implanted birth control device for women on the market that does not require a surgical incision, was approved in 2002 and has been associated with adverse events including perforation of the uterus and/or fallopian tubes, device migration into the abdomen or pelvis, persistent pain, and suspected allergic reactions or hypersensitivity. Some women have also reported headache, fatigue, weight changes, hair loss, and mood changes.
In 2016, based on product safety monitoring by the FDA, a postmarketing study was ordered, as was a boxed warning and a more comprehensive patient decision checklist to inform patients about the risk of adverse events.
“We’ve been closely evaluating new information on the use of Essure, and based on our review of a growing body of evidence, we believe this product requires additional, meaningful safeguards to ensure women are able to make informed decisions about risk when considering this option,” FDA commissioner Scott Gottlieb, MD, said in the press release, adding that it is unacceptable that some women were not being adequately informed.
“Every single woman receiving this device should fully understand the associated risks,” he said.
Terri Cornelison, MD, assistant director for the health of women in the Center for Devices and Radiological Health added that ensuring informed decision making is just one important step in ongoing efforts to monitor the Essure device.
“We remain committed to carefully and throughly considering all new data and evidence and will continue to work with patients affected by this device as part of our process,” she said. “While some women may continue to choose Essure as their birth control option based on current information, as new information becomes available, the FDA will continue to keep the public informed of the agency’s evaluation and findings and consider regulatory options that appropriately balance benefits and risks for Essure.”
The Food and Drug Administration has restricted the sale and distribution of Bayer’s Essure permanent contraception device to only health care providers and facilities that provide patients with information about the risks and benefits of the device.
In an order issued April 9, the FDA’s Center for Devices and Radiological Health informed Bayer of the restrictions, citing a need to “provide reasonable assurance of the safety and effectiveness of the device.”
Bayer must implement the restrictions immediately and must ensure health care provider compliance.
“The FDA is taking this step after becoming aware that some women were not being adequately informed of Essure’s risks before getting the device implanted, despite previous significant efforts to educate patients and doctors about the risks associated with this device,” the agency said in a press release, adding that it “plans to enforce these requirements and will take appropriate action for a failure to comply, including applicable criminal and civil penalties.”
Essure, which is the only permanently implanted birth control device for women on the market that does not require a surgical incision, was approved in 2002 and has been associated with adverse events including perforation of the uterus and/or fallopian tubes, device migration into the abdomen or pelvis, persistent pain, and suspected allergic reactions or hypersensitivity. Some women have also reported headache, fatigue, weight changes, hair loss, and mood changes.
In 2016, based on product safety monitoring by the FDA, a postmarketing study was ordered, as was a boxed warning and a more comprehensive patient decision checklist to inform patients about the risk of adverse events.
“We’ve been closely evaluating new information on the use of Essure, and based on our review of a growing body of evidence, we believe this product requires additional, meaningful safeguards to ensure women are able to make informed decisions about risk when considering this option,” FDA commissioner Scott Gottlieb, MD, said in the press release, adding that it is unacceptable that some women were not being adequately informed.
“Every single woman receiving this device should fully understand the associated risks,” he said.
Terri Cornelison, MD, assistant director for the health of women in the Center for Devices and Radiological Health added that ensuring informed decision making is just one important step in ongoing efforts to monitor the Essure device.
“We remain committed to carefully and throughly considering all new data and evidence and will continue to work with patients affected by this device as part of our process,” she said. “While some women may continue to choose Essure as their birth control option based on current information, as new information becomes available, the FDA will continue to keep the public informed of the agency’s evaluation and findings and consider regulatory options that appropriately balance benefits and risks for Essure.”
The Food and Drug Administration has restricted the sale and distribution of Bayer’s Essure permanent contraception device to only health care providers and facilities that provide patients with information about the risks and benefits of the device.
In an order issued April 9, the FDA’s Center for Devices and Radiological Health informed Bayer of the restrictions, citing a need to “provide reasonable assurance of the safety and effectiveness of the device.”
Bayer must implement the restrictions immediately and must ensure health care provider compliance.
“The FDA is taking this step after becoming aware that some women were not being adequately informed of Essure’s risks before getting the device implanted, despite previous significant efforts to educate patients and doctors about the risks associated with this device,” the agency said in a press release, adding that it “plans to enforce these requirements and will take appropriate action for a failure to comply, including applicable criminal and civil penalties.”
Essure, which is the only permanently implanted birth control device for women on the market that does not require a surgical incision, was approved in 2002 and has been associated with adverse events including perforation of the uterus and/or fallopian tubes, device migration into the abdomen or pelvis, persistent pain, and suspected allergic reactions or hypersensitivity. Some women have also reported headache, fatigue, weight changes, hair loss, and mood changes.
In 2016, based on product safety monitoring by the FDA, a postmarketing study was ordered, as was a boxed warning and a more comprehensive patient decision checklist to inform patients about the risk of adverse events.
“We’ve been closely evaluating new information on the use of Essure, and based on our review of a growing body of evidence, we believe this product requires additional, meaningful safeguards to ensure women are able to make informed decisions about risk when considering this option,” FDA commissioner Scott Gottlieb, MD, said in the press release, adding that it is unacceptable that some women were not being adequately informed.
“Every single woman receiving this device should fully understand the associated risks,” he said.
Terri Cornelison, MD, assistant director for the health of women in the Center for Devices and Radiological Health added that ensuring informed decision making is just one important step in ongoing efforts to monitor the Essure device.
“We remain committed to carefully and throughly considering all new data and evidence and will continue to work with patients affected by this device as part of our process,” she said. “While some women may continue to choose Essure as their birth control option based on current information, as new information becomes available, the FDA will continue to keep the public informed of the agency’s evaluation and findings and consider regulatory options that appropriately balance benefits and risks for Essure.”
Pregnant women in clinical trials: FDA questions how to include them
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Unusual antibiotic resistance found in more than 200 bacteria
The Centers for Disease Control and Prevention’s Antibiotic Resistance (AR) Lab Network has detected 221 instances of bacteria with especially rare resistance genes in the United States, according to a Vital Signs report published online and expanded upon in CDC’s MMWR Weekly.
The MMWR Weekly report, “Containment of Novel Multidrug-Resistant Organisms and Resistance Mechanisms,” which goes deeper into the science behind the issue, shows that in 9 months, in all states and Puerto Rico, health department workers in the AR lab network tested 5,776 samples of highly resistant bacteria, according to Anne Schuchat, MD, principal deputy director of the CDC. These bacteria were immediately tested for unusual resistance – “those genes that were highly resistant, or rare, with special resistance that could spread,” she said.
“Of the 5,776, about 1 in 4 of the bacteria had a gene that helped it spread its resistance. And there were 221 instances of an especially rare resistance gene,” she added. This prompted intense screening, revealing “that 1 in 10 tests were also positive. Meaning the unusual resistance may have spread to other patients. And could have continued spreading if left undetected.”
The report looked at carbapenem-resistant Enterobacteriaceae (CRE) and Enterobacteriaceae with extended-spectrum beta-lactamases (ESBL) infection data from the National Healthcare Safety Network from 2006-2015 to calculate changes in the year over year proportions of these infections and how an enhanced detection and control strategy curbs carbapenem resistance.
This strategy includes components such as timely implementation of appropriate infection control measures, conducting a health care and contact investigation with follow-up, and implementing a system to ensure adherence to infection control measures.
“With independent, or single facility approaches to control spread, a dangerous type of unusual resistance in Enterobacteriaceae [ESBL phenotype] decreased by about 2% per year [(risk ratio [RR] = 0.98, P less than .001)].” With a more aggressive approach, using guidance such as CDC’s CRE toolkit, released in 2009, another type of unusual resistance [CRE] in the same bacteria (Enterobacteriaceae) decreased by nearly 15% per year (RR = 0.85, P less than .01).
The difference may be due in part to the more directed response utilized to slow the spread of the “nightmare bacteria,” once it was identified, said Dr. Schuchat.
These results show massive promise even if only partially effective, specifically for CRE.
“CDC estimates show that if only 20% effective, the containment strategy can reduce the number of nightmare bacteria [CRE] cases by 76% over 3 years in one area.”
Due to the nature of antibiotic resistance and its ability to spread, this poses a significant public health threat. Antibiotics are not simply used to treat infections but are a safety net that is used in cancer treatment, surgery, and ICU care, Dr. Schuchat pointed out. The rise of antibiotic resistance is a threat to that safety net and accounts for nearly 2,000,000 antibiotic resistant infections and approximately 23,000 deaths per year.
But aggressive responses to these infections can control their spread. Dr. Schuchat used the analogy of controlling a fire to illustrate the concept.
“Much like a fire, finding and stopping unusual resistance early when it’s just a spark protects people.”
Dr. Schuchat reiterated that simply identifying the issue is only part of the equation.
“Detection is not enough on its own. When there is a fire, somebody needs to put it out. CDC supports more than 500 local staff across the country to combat antibiotic resistance wherever it emerges.”
While the report highlights the strides that have been made in combating antibiotic resistance, Paul Auwaerter, MD, president of the Infectious Diseases Society of America, released a statement highlighting the need to further fund these efforts.
“The report spells out the need to accelerate efforts to curb resistance or face an increasing burden including novel resistance mutations that threaten health,” stated Dr. Auwaerter. “The efforts detailed in the Vital Signs report were made possible through new congressional funding in 2016 to combat antibiotic resistance. We urge Congress to sustain and to grow that investment so that further progress will prepare us to meet the future challenges of antibiotic resistance from a position of strength,” he added.
A fact sheet with a brief summation of the vital signs report is available here.
The Centers for Disease Control and Prevention’s Antibiotic Resistance (AR) Lab Network has detected 221 instances of bacteria with especially rare resistance genes in the United States, according to a Vital Signs report published online and expanded upon in CDC’s MMWR Weekly.
The MMWR Weekly report, “Containment of Novel Multidrug-Resistant Organisms and Resistance Mechanisms,” which goes deeper into the science behind the issue, shows that in 9 months, in all states and Puerto Rico, health department workers in the AR lab network tested 5,776 samples of highly resistant bacteria, according to Anne Schuchat, MD, principal deputy director of the CDC. These bacteria were immediately tested for unusual resistance – “those genes that were highly resistant, or rare, with special resistance that could spread,” she said.
“Of the 5,776, about 1 in 4 of the bacteria had a gene that helped it spread its resistance. And there were 221 instances of an especially rare resistance gene,” she added. This prompted intense screening, revealing “that 1 in 10 tests were also positive. Meaning the unusual resistance may have spread to other patients. And could have continued spreading if left undetected.”
The report looked at carbapenem-resistant Enterobacteriaceae (CRE) and Enterobacteriaceae with extended-spectrum beta-lactamases (ESBL) infection data from the National Healthcare Safety Network from 2006-2015 to calculate changes in the year over year proportions of these infections and how an enhanced detection and control strategy curbs carbapenem resistance.
This strategy includes components such as timely implementation of appropriate infection control measures, conducting a health care and contact investigation with follow-up, and implementing a system to ensure adherence to infection control measures.
“With independent, or single facility approaches to control spread, a dangerous type of unusual resistance in Enterobacteriaceae [ESBL phenotype] decreased by about 2% per year [(risk ratio [RR] = 0.98, P less than .001)].” With a more aggressive approach, using guidance such as CDC’s CRE toolkit, released in 2009, another type of unusual resistance [CRE] in the same bacteria (Enterobacteriaceae) decreased by nearly 15% per year (RR = 0.85, P less than .01).
The difference may be due in part to the more directed response utilized to slow the spread of the “nightmare bacteria,” once it was identified, said Dr. Schuchat.
These results show massive promise even if only partially effective, specifically for CRE.
“CDC estimates show that if only 20% effective, the containment strategy can reduce the number of nightmare bacteria [CRE] cases by 76% over 3 years in one area.”
Due to the nature of antibiotic resistance and its ability to spread, this poses a significant public health threat. Antibiotics are not simply used to treat infections but are a safety net that is used in cancer treatment, surgery, and ICU care, Dr. Schuchat pointed out. The rise of antibiotic resistance is a threat to that safety net and accounts for nearly 2,000,000 antibiotic resistant infections and approximately 23,000 deaths per year.
But aggressive responses to these infections can control their spread. Dr. Schuchat used the analogy of controlling a fire to illustrate the concept.
“Much like a fire, finding and stopping unusual resistance early when it’s just a spark protects people.”
Dr. Schuchat reiterated that simply identifying the issue is only part of the equation.
“Detection is not enough on its own. When there is a fire, somebody needs to put it out. CDC supports more than 500 local staff across the country to combat antibiotic resistance wherever it emerges.”
While the report highlights the strides that have been made in combating antibiotic resistance, Paul Auwaerter, MD, president of the Infectious Diseases Society of America, released a statement highlighting the need to further fund these efforts.
“The report spells out the need to accelerate efforts to curb resistance or face an increasing burden including novel resistance mutations that threaten health,” stated Dr. Auwaerter. “The efforts detailed in the Vital Signs report were made possible through new congressional funding in 2016 to combat antibiotic resistance. We urge Congress to sustain and to grow that investment so that further progress will prepare us to meet the future challenges of antibiotic resistance from a position of strength,” he added.
A fact sheet with a brief summation of the vital signs report is available here.
The Centers for Disease Control and Prevention’s Antibiotic Resistance (AR) Lab Network has detected 221 instances of bacteria with especially rare resistance genes in the United States, according to a Vital Signs report published online and expanded upon in CDC’s MMWR Weekly.
The MMWR Weekly report, “Containment of Novel Multidrug-Resistant Organisms and Resistance Mechanisms,” which goes deeper into the science behind the issue, shows that in 9 months, in all states and Puerto Rico, health department workers in the AR lab network tested 5,776 samples of highly resistant bacteria, according to Anne Schuchat, MD, principal deputy director of the CDC. These bacteria were immediately tested for unusual resistance – “those genes that were highly resistant, or rare, with special resistance that could spread,” she said.
“Of the 5,776, about 1 in 4 of the bacteria had a gene that helped it spread its resistance. And there were 221 instances of an especially rare resistance gene,” she added. This prompted intense screening, revealing “that 1 in 10 tests were also positive. Meaning the unusual resistance may have spread to other patients. And could have continued spreading if left undetected.”
The report looked at carbapenem-resistant Enterobacteriaceae (CRE) and Enterobacteriaceae with extended-spectrum beta-lactamases (ESBL) infection data from the National Healthcare Safety Network from 2006-2015 to calculate changes in the year over year proportions of these infections and how an enhanced detection and control strategy curbs carbapenem resistance.
This strategy includes components such as timely implementation of appropriate infection control measures, conducting a health care and contact investigation with follow-up, and implementing a system to ensure adherence to infection control measures.
“With independent, or single facility approaches to control spread, a dangerous type of unusual resistance in Enterobacteriaceae [ESBL phenotype] decreased by about 2% per year [(risk ratio [RR] = 0.98, P less than .001)].” With a more aggressive approach, using guidance such as CDC’s CRE toolkit, released in 2009, another type of unusual resistance [CRE] in the same bacteria (Enterobacteriaceae) decreased by nearly 15% per year (RR = 0.85, P less than .01).
The difference may be due in part to the more directed response utilized to slow the spread of the “nightmare bacteria,” once it was identified, said Dr. Schuchat.
These results show massive promise even if only partially effective, specifically for CRE.
“CDC estimates show that if only 20% effective, the containment strategy can reduce the number of nightmare bacteria [CRE] cases by 76% over 3 years in one area.”
Due to the nature of antibiotic resistance and its ability to spread, this poses a significant public health threat. Antibiotics are not simply used to treat infections but are a safety net that is used in cancer treatment, surgery, and ICU care, Dr. Schuchat pointed out. The rise of antibiotic resistance is a threat to that safety net and accounts for nearly 2,000,000 antibiotic resistant infections and approximately 23,000 deaths per year.
But aggressive responses to these infections can control their spread. Dr. Schuchat used the analogy of controlling a fire to illustrate the concept.
“Much like a fire, finding and stopping unusual resistance early when it’s just a spark protects people.”
Dr. Schuchat reiterated that simply identifying the issue is only part of the equation.
“Detection is not enough on its own. When there is a fire, somebody needs to put it out. CDC supports more than 500 local staff across the country to combat antibiotic resistance wherever it emerges.”
While the report highlights the strides that have been made in combating antibiotic resistance, Paul Auwaerter, MD, president of the Infectious Diseases Society of America, released a statement highlighting the need to further fund these efforts.
“The report spells out the need to accelerate efforts to curb resistance or face an increasing burden including novel resistance mutations that threaten health,” stated Dr. Auwaerter. “The efforts detailed in the Vital Signs report were made possible through new congressional funding in 2016 to combat antibiotic resistance. We urge Congress to sustain and to grow that investment so that further progress will prepare us to meet the future challenges of antibiotic resistance from a position of strength,” he added.
A fact sheet with a brief summation of the vital signs report is available here.
FROM A CDC TELEBRIEFING
FDA recalls kratom products for salmonella contamination
The Food and Drug Administration on April 3 recalled all products containing kratom manufactured by Triangle Pharmanaturals LLC, after a number of supplements tested positive for salmonella.
The FDA advises consumers to get rid of products including Raw Form Organics Maeng Da Kratom Emerald Green, Raw Form Organics Maeng Da Kratom Ivory White, and Raw Form Organics Maeng Da Kratom Ruby Red.
Evidence of the contamination was found after two samples were collected from a retail store in Oregon by the Oregon Public Health Division and tested positive for salmonella.
The recall was ordered after Triangle Pharmanaturals did not comply with a March 30 formal request from the FDA to voluntarily recall their products.
“This action is based on the imminent health risk posed by the contamination of this product with salmonella, and the refusal of this company to voluntarily act to protect its customers and issue a recall, despite our repeated requests and actions,” said FDA Commissioner Scott Gottlieb, M.D., said in a statement. “The action today is based on the risks posed by the contamination of this particular product with a potentially dangerous pathogen.”
At press time, Triangle Pharmanaturals did not respond to a request for comment.
This is the most recent in a list recalls of kratom products as part of an ongoing investigation of a salmonella outbreak by the FDA; however Triangle Pharmanaturals’ noncompliance is unique to the agency, according to an FDA representative.
“This is the first time the agency has issued a mandatory recall order to protect Americans from contaminated food products,” Michael Felberbaum, an FDA press officer, said in an interview. “This is the third time the FDA has invoked its mandatory recall authority, but the first time the agency ordered a mandatory recall because a company has opted not to voluntarily recall after the FDA’s notification of an opportunity to initiate a voluntary recall.”
Earlier in March, the CDC reported 87 people in 35 states infected with either Salmonella Javiana, Salmonela Okatie, or Salmonella Thompson, which have been associated with the outbreak.
While salmonella was identified in Triangle Pharmanaturals’ products, the strains identified are not currently linked to the outbreak.
Kratom, a plant that commonly grows in South East Asian countries like Thailand, Malaysia, Indonesia, and Papua New Guinea, has recently been used to produce food supplements and marketed as an alternative to addictive pain medication like opioids, as well as used to help treat opioid withdrawal symptoms.
Use of the food supplement has fired debate among physicians, patients, and public officials as all sides continue to determine its efficacy and how, or whether, it should be given a drug classification.
The Food and Drug Administration on April 3 recalled all products containing kratom manufactured by Triangle Pharmanaturals LLC, after a number of supplements tested positive for salmonella.
The FDA advises consumers to get rid of products including Raw Form Organics Maeng Da Kratom Emerald Green, Raw Form Organics Maeng Da Kratom Ivory White, and Raw Form Organics Maeng Da Kratom Ruby Red.
Evidence of the contamination was found after two samples were collected from a retail store in Oregon by the Oregon Public Health Division and tested positive for salmonella.
The recall was ordered after Triangle Pharmanaturals did not comply with a March 30 formal request from the FDA to voluntarily recall their products.
“This action is based on the imminent health risk posed by the contamination of this product with salmonella, and the refusal of this company to voluntarily act to protect its customers and issue a recall, despite our repeated requests and actions,” said FDA Commissioner Scott Gottlieb, M.D., said in a statement. “The action today is based on the risks posed by the contamination of this particular product with a potentially dangerous pathogen.”
At press time, Triangle Pharmanaturals did not respond to a request for comment.
This is the most recent in a list recalls of kratom products as part of an ongoing investigation of a salmonella outbreak by the FDA; however Triangle Pharmanaturals’ noncompliance is unique to the agency, according to an FDA representative.
“This is the first time the agency has issued a mandatory recall order to protect Americans from contaminated food products,” Michael Felberbaum, an FDA press officer, said in an interview. “This is the third time the FDA has invoked its mandatory recall authority, but the first time the agency ordered a mandatory recall because a company has opted not to voluntarily recall after the FDA’s notification of an opportunity to initiate a voluntary recall.”
Earlier in March, the CDC reported 87 people in 35 states infected with either Salmonella Javiana, Salmonela Okatie, or Salmonella Thompson, which have been associated with the outbreak.
While salmonella was identified in Triangle Pharmanaturals’ products, the strains identified are not currently linked to the outbreak.
Kratom, a plant that commonly grows in South East Asian countries like Thailand, Malaysia, Indonesia, and Papua New Guinea, has recently been used to produce food supplements and marketed as an alternative to addictive pain medication like opioids, as well as used to help treat opioid withdrawal symptoms.
Use of the food supplement has fired debate among physicians, patients, and public officials as all sides continue to determine its efficacy and how, or whether, it should be given a drug classification.
The Food and Drug Administration on April 3 recalled all products containing kratom manufactured by Triangle Pharmanaturals LLC, after a number of supplements tested positive for salmonella.
The FDA advises consumers to get rid of products including Raw Form Organics Maeng Da Kratom Emerald Green, Raw Form Organics Maeng Da Kratom Ivory White, and Raw Form Organics Maeng Da Kratom Ruby Red.
Evidence of the contamination was found after two samples were collected from a retail store in Oregon by the Oregon Public Health Division and tested positive for salmonella.
The recall was ordered after Triangle Pharmanaturals did not comply with a March 30 formal request from the FDA to voluntarily recall their products.
“This action is based on the imminent health risk posed by the contamination of this product with salmonella, and the refusal of this company to voluntarily act to protect its customers and issue a recall, despite our repeated requests and actions,” said FDA Commissioner Scott Gottlieb, M.D., said in a statement. “The action today is based on the risks posed by the contamination of this particular product with a potentially dangerous pathogen.”
At press time, Triangle Pharmanaturals did not respond to a request for comment.
This is the most recent in a list recalls of kratom products as part of an ongoing investigation of a salmonella outbreak by the FDA; however Triangle Pharmanaturals’ noncompliance is unique to the agency, according to an FDA representative.
“This is the first time the agency has issued a mandatory recall order to protect Americans from contaminated food products,” Michael Felberbaum, an FDA press officer, said in an interview. “This is the third time the FDA has invoked its mandatory recall authority, but the first time the agency ordered a mandatory recall because a company has opted not to voluntarily recall after the FDA’s notification of an opportunity to initiate a voluntary recall.”
Earlier in March, the CDC reported 87 people in 35 states infected with either Salmonella Javiana, Salmonela Okatie, or Salmonella Thompson, which have been associated with the outbreak.
While salmonella was identified in Triangle Pharmanaturals’ products, the strains identified are not currently linked to the outbreak.
Kratom, a plant that commonly grows in South East Asian countries like Thailand, Malaysia, Indonesia, and Papua New Guinea, has recently been used to produce food supplements and marketed as an alternative to addictive pain medication like opioids, as well as used to help treat opioid withdrawal symptoms.
Use of the food supplement has fired debate among physicians, patients, and public officials as all sides continue to determine its efficacy and how, or whether, it should be given a drug classification.