User login
British Society of Rheumatology issues first U.K. lupus guideline
From diagnosing, assessing, and managing common manifestations of nonrenal lupus, such as skin rashes and arthritis, through dealing with less common but potentially more serious problems such as kidney disease, the guideline aims to help everyone involved in the management of patients with SLE to give the best, evidenced-based care.
U.K.-specific guidance
The British Society for Rheumatology’s (BSR) guideline is the first to specifically cover lupus management in the United Kingdom, and it builds on existing European League Against Rheumatism (EULAR) guidance published almost a decade ago (Ann Rheum Dis. 2008;67:195-205), and more recent EULAR/European Renal Association–European Dialysis and Transplant Association (Ann Rheum Dis. 2012;71:1771-82) and American College of Rheumatology (Arthritis Care Res. 2012;64:797-808) recommendations on managing lupus nephritis (LN).
The BSR’s full guideline provides a summary of the EULAR/ERA-EDTA’s LN guidelines, and the degree to which their new guidelines concur.
As with all BSR guidelines, the recommendations have been developed by a multidisciplinary team. This included academic and consultant rheumatologists and nephrologists, rheumatology trainees, a primary care physician, a clinical nurse specialist, a patient representative, and a lay member. This should make the guideline relevant to anyone who may come across someone with SLE, including primary care physicians, dermatologists, and emergency medicine practitioners.
“These recommendations are based on the literature review covering the diagnosis, assessment, monitoring, and treatment of mild, moderate, and severe lupus, including neuropsychiatric disease,” the guideline authors stated. They noted that the reason they looked only at nonrenal disease was because the EULAR/ERA-EDTA recommendations for LN have been published close to the time that work was started on the guideline. Each of the recommendations the multidisciplinary team devised was carefully graded and the degree to which members of the team agreed with each recommendation was calculated.
Diagnosis recommendations
One of the key recommendations regarding the diagnosis of SLE is that a combination of clinical features and at least one relevant immunologic irregularity needs to be present. Blood tests, including serologic marker tests, should be performed if there is clinical suspicion of lupus.
Another recommendation on diagnosis is that if antinuclear antibodies are absent, then it is unlikely that the patient has lupus. This is because around 95% of SLE patients will test positive for antinuclear antibodies. Antiphospholipid antibodies should be tested in all patients with lupus at baseline, according to the guideline.
Monitoring recommendations
“Patients with SLE should be monitored on a regular basis for disease manifestations, drug toxicities, and comorbidities,” Dr. Gordon and her associates advised in one of the recommendations on monitoring patients. In another, they wrote that those with active disease need reviewing at least every 1-3 months, which should include evaluation of patients’ blood pressure, urine, renal function, anti-dsDNA antibodies, complement, a full blood count, and liver function tests.
It is also important to monitor patients for the presence of antiphospholipid antibodies, which are associated with thrombotic events, and it is always important to be on the lookout for comorbidities such as atherosclerotic disease and manage modifiable risk factors such as hypertension. The guideline does not go into detail about managing all of the potential complications of lupus, however, as these are covered by other national guidelines.
Treatment recommendations
Guidance on treatment is separated into how to treat patients with mild, moderate, and severe SLE. The guideline does not cover topical or systemic treatment for isolated cutaneous lupus, nor does it look at how to manage pediatric patients, the authors noted. General guidance is given on how to treat patients, and specific dosing regimens are beyond the scope of the guidelines.
The recommendations encourage the use of a variety of treatments to try to ensure less reliance on the use of steroids to control symptoms. The guideline authors noted that only hydroxychloroquine, corticosteroids, and belimumab are currently licensed treatments for lupus in the United Kingdom.
For mild disease, the disease-modifying antirheumatic drugs hydroxychloroquine and methotrexate are suggested, as are nonsteroidal anti-inflammatory drugs. If prednisolone is used, then it should be in low doses (7.5 mg or less per day). Patients should be encouraged to use sunscreen and sun avoidance to protect them against ultraviolet-induced skin lesions.
For moderate disease, higher steroid doses may be needed and immunosuppressives might be warranted, and in refractory cases, monoclonal antibody treatment may be necessary.
For severe disease, thorough investigation is essential to exclude other possible causes of any renal or neuropsychiatric manifestations. Immunosuppressive treatment is recommended, with biologic therapies considered on a case-by-case basis. Intravenous immunoglobulin and plasmapheresis may also be an option in certain patients.
Key standards of care
As general standards of care, Dr. Gordon and her coauthors wrote that “lupus patients should be referred to a physician with experience in managing lupus who can confirm the diagnosis, assess the level of disease activity, and provide advice on treatment and monitoring of the disease, its complications, and side effects of therapy.”
Furthermore, a multidisciplinary team approach is needed, and if a treatment is not helping get patients into a state of low disease activity within an expected time frame, “it is important to consider adherence to treatment and adjusting the therapy to reduce the accumulation of chronic damage.” It is also recommended that patients receive personalized advice, written information, and education about the disease and its drug treatment.
No specific funding was received from any organizations in the public, commercial, or not-for-profit sectors to carry out the work described in the guideline.
Dr. Gordon has undertaken consultancies and received honoraria from Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, MedImmune, Merck Serono, Parexel, Roche, and UCB. She has been a member of the speakers bureau for GlaxoSmithKline, UCB, and Eli Lilly. She also has received research grant support from Aspreva/Vifor Pharma in the past and currently receives it from UCB.
From diagnosing, assessing, and managing common manifestations of nonrenal lupus, such as skin rashes and arthritis, through dealing with less common but potentially more serious problems such as kidney disease, the guideline aims to help everyone involved in the management of patients with SLE to give the best, evidenced-based care.
U.K.-specific guidance
The British Society for Rheumatology’s (BSR) guideline is the first to specifically cover lupus management in the United Kingdom, and it builds on existing European League Against Rheumatism (EULAR) guidance published almost a decade ago (Ann Rheum Dis. 2008;67:195-205), and more recent EULAR/European Renal Association–European Dialysis and Transplant Association (Ann Rheum Dis. 2012;71:1771-82) and American College of Rheumatology (Arthritis Care Res. 2012;64:797-808) recommendations on managing lupus nephritis (LN).
The BSR’s full guideline provides a summary of the EULAR/ERA-EDTA’s LN guidelines, and the degree to which their new guidelines concur.
As with all BSR guidelines, the recommendations have been developed by a multidisciplinary team. This included academic and consultant rheumatologists and nephrologists, rheumatology trainees, a primary care physician, a clinical nurse specialist, a patient representative, and a lay member. This should make the guideline relevant to anyone who may come across someone with SLE, including primary care physicians, dermatologists, and emergency medicine practitioners.
“These recommendations are based on the literature review covering the diagnosis, assessment, monitoring, and treatment of mild, moderate, and severe lupus, including neuropsychiatric disease,” the guideline authors stated. They noted that the reason they looked only at nonrenal disease was because the EULAR/ERA-EDTA recommendations for LN have been published close to the time that work was started on the guideline. Each of the recommendations the multidisciplinary team devised was carefully graded and the degree to which members of the team agreed with each recommendation was calculated.
Diagnosis recommendations
One of the key recommendations regarding the diagnosis of SLE is that a combination of clinical features and at least one relevant immunologic irregularity needs to be present. Blood tests, including serologic marker tests, should be performed if there is clinical suspicion of lupus.
Another recommendation on diagnosis is that if antinuclear antibodies are absent, then it is unlikely that the patient has lupus. This is because around 95% of SLE patients will test positive for antinuclear antibodies. Antiphospholipid antibodies should be tested in all patients with lupus at baseline, according to the guideline.
Monitoring recommendations
“Patients with SLE should be monitored on a regular basis for disease manifestations, drug toxicities, and comorbidities,” Dr. Gordon and her associates advised in one of the recommendations on monitoring patients. In another, they wrote that those with active disease need reviewing at least every 1-3 months, which should include evaluation of patients’ blood pressure, urine, renal function, anti-dsDNA antibodies, complement, a full blood count, and liver function tests.
It is also important to monitor patients for the presence of antiphospholipid antibodies, which are associated with thrombotic events, and it is always important to be on the lookout for comorbidities such as atherosclerotic disease and manage modifiable risk factors such as hypertension. The guideline does not go into detail about managing all of the potential complications of lupus, however, as these are covered by other national guidelines.
Treatment recommendations
Guidance on treatment is separated into how to treat patients with mild, moderate, and severe SLE. The guideline does not cover topical or systemic treatment for isolated cutaneous lupus, nor does it look at how to manage pediatric patients, the authors noted. General guidance is given on how to treat patients, and specific dosing regimens are beyond the scope of the guidelines.
The recommendations encourage the use of a variety of treatments to try to ensure less reliance on the use of steroids to control symptoms. The guideline authors noted that only hydroxychloroquine, corticosteroids, and belimumab are currently licensed treatments for lupus in the United Kingdom.
For mild disease, the disease-modifying antirheumatic drugs hydroxychloroquine and methotrexate are suggested, as are nonsteroidal anti-inflammatory drugs. If prednisolone is used, then it should be in low doses (7.5 mg or less per day). Patients should be encouraged to use sunscreen and sun avoidance to protect them against ultraviolet-induced skin lesions.
For moderate disease, higher steroid doses may be needed and immunosuppressives might be warranted, and in refractory cases, monoclonal antibody treatment may be necessary.
For severe disease, thorough investigation is essential to exclude other possible causes of any renal or neuropsychiatric manifestations. Immunosuppressive treatment is recommended, with biologic therapies considered on a case-by-case basis. Intravenous immunoglobulin and plasmapheresis may also be an option in certain patients.
Key standards of care
As general standards of care, Dr. Gordon and her coauthors wrote that “lupus patients should be referred to a physician with experience in managing lupus who can confirm the diagnosis, assess the level of disease activity, and provide advice on treatment and monitoring of the disease, its complications, and side effects of therapy.”
Furthermore, a multidisciplinary team approach is needed, and if a treatment is not helping get patients into a state of low disease activity within an expected time frame, “it is important to consider adherence to treatment and adjusting the therapy to reduce the accumulation of chronic damage.” It is also recommended that patients receive personalized advice, written information, and education about the disease and its drug treatment.
No specific funding was received from any organizations in the public, commercial, or not-for-profit sectors to carry out the work described in the guideline.
Dr. Gordon has undertaken consultancies and received honoraria from Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, MedImmune, Merck Serono, Parexel, Roche, and UCB. She has been a member of the speakers bureau for GlaxoSmithKline, UCB, and Eli Lilly. She also has received research grant support from Aspreva/Vifor Pharma in the past and currently receives it from UCB.
From diagnosing, assessing, and managing common manifestations of nonrenal lupus, such as skin rashes and arthritis, through dealing with less common but potentially more serious problems such as kidney disease, the guideline aims to help everyone involved in the management of patients with SLE to give the best, evidenced-based care.
U.K.-specific guidance
The British Society for Rheumatology’s (BSR) guideline is the first to specifically cover lupus management in the United Kingdom, and it builds on existing European League Against Rheumatism (EULAR) guidance published almost a decade ago (Ann Rheum Dis. 2008;67:195-205), and more recent EULAR/European Renal Association–European Dialysis and Transplant Association (Ann Rheum Dis. 2012;71:1771-82) and American College of Rheumatology (Arthritis Care Res. 2012;64:797-808) recommendations on managing lupus nephritis (LN).
The BSR’s full guideline provides a summary of the EULAR/ERA-EDTA’s LN guidelines, and the degree to which their new guidelines concur.
As with all BSR guidelines, the recommendations have been developed by a multidisciplinary team. This included academic and consultant rheumatologists and nephrologists, rheumatology trainees, a primary care physician, a clinical nurse specialist, a patient representative, and a lay member. This should make the guideline relevant to anyone who may come across someone with SLE, including primary care physicians, dermatologists, and emergency medicine practitioners.
“These recommendations are based on the literature review covering the diagnosis, assessment, monitoring, and treatment of mild, moderate, and severe lupus, including neuropsychiatric disease,” the guideline authors stated. They noted that the reason they looked only at nonrenal disease was because the EULAR/ERA-EDTA recommendations for LN have been published close to the time that work was started on the guideline. Each of the recommendations the multidisciplinary team devised was carefully graded and the degree to which members of the team agreed with each recommendation was calculated.
Diagnosis recommendations
One of the key recommendations regarding the diagnosis of SLE is that a combination of clinical features and at least one relevant immunologic irregularity needs to be present. Blood tests, including serologic marker tests, should be performed if there is clinical suspicion of lupus.
Another recommendation on diagnosis is that if antinuclear antibodies are absent, then it is unlikely that the patient has lupus. This is because around 95% of SLE patients will test positive for antinuclear antibodies. Antiphospholipid antibodies should be tested in all patients with lupus at baseline, according to the guideline.
Monitoring recommendations
“Patients with SLE should be monitored on a regular basis for disease manifestations, drug toxicities, and comorbidities,” Dr. Gordon and her associates advised in one of the recommendations on monitoring patients. In another, they wrote that those with active disease need reviewing at least every 1-3 months, which should include evaluation of patients’ blood pressure, urine, renal function, anti-dsDNA antibodies, complement, a full blood count, and liver function tests.
It is also important to monitor patients for the presence of antiphospholipid antibodies, which are associated with thrombotic events, and it is always important to be on the lookout for comorbidities such as atherosclerotic disease and manage modifiable risk factors such as hypertension. The guideline does not go into detail about managing all of the potential complications of lupus, however, as these are covered by other national guidelines.
Treatment recommendations
Guidance on treatment is separated into how to treat patients with mild, moderate, and severe SLE. The guideline does not cover topical or systemic treatment for isolated cutaneous lupus, nor does it look at how to manage pediatric patients, the authors noted. General guidance is given on how to treat patients, and specific dosing regimens are beyond the scope of the guidelines.
The recommendations encourage the use of a variety of treatments to try to ensure less reliance on the use of steroids to control symptoms. The guideline authors noted that only hydroxychloroquine, corticosteroids, and belimumab are currently licensed treatments for lupus in the United Kingdom.
For mild disease, the disease-modifying antirheumatic drugs hydroxychloroquine and methotrexate are suggested, as are nonsteroidal anti-inflammatory drugs. If prednisolone is used, then it should be in low doses (7.5 mg or less per day). Patients should be encouraged to use sunscreen and sun avoidance to protect them against ultraviolet-induced skin lesions.
For moderate disease, higher steroid doses may be needed and immunosuppressives might be warranted, and in refractory cases, monoclonal antibody treatment may be necessary.
For severe disease, thorough investigation is essential to exclude other possible causes of any renal or neuropsychiatric manifestations. Immunosuppressive treatment is recommended, with biologic therapies considered on a case-by-case basis. Intravenous immunoglobulin and plasmapheresis may also be an option in certain patients.
Key standards of care
As general standards of care, Dr. Gordon and her coauthors wrote that “lupus patients should be referred to a physician with experience in managing lupus who can confirm the diagnosis, assess the level of disease activity, and provide advice on treatment and monitoring of the disease, its complications, and side effects of therapy.”
Furthermore, a multidisciplinary team approach is needed, and if a treatment is not helping get patients into a state of low disease activity within an expected time frame, “it is important to consider adherence to treatment and adjusting the therapy to reduce the accumulation of chronic damage.” It is also recommended that patients receive personalized advice, written information, and education about the disease and its drug treatment.
No specific funding was received from any organizations in the public, commercial, or not-for-profit sectors to carry out the work described in the guideline.
Dr. Gordon has undertaken consultancies and received honoraria from Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, MedImmune, Merck Serono, Parexel, Roche, and UCB. She has been a member of the speakers bureau for GlaxoSmithKline, UCB, and Eli Lilly. She also has received research grant support from Aspreva/Vifor Pharma in the past and currently receives it from UCB.
FROM RHEUMATOLOGY
Vaccination program cut hospital-treated RV gastroenteritis in young children
under 5 years, and it more than adequately pays for itself in secondary health care costs, said Tuija Leino of the National Institute for Health and Welfare, Helsinki, and associates.
Since 2009, all Finnish children younger than 5 years have been offered an RV vaccine.
The investigators conducted a register-based study comparing the RV disease burden before and after introduction of RV vaccination, with the years 1999-2005 as the prevaccine period and the years 2010-2014 as the vaccination period. The study population was all children younger than 5 years living in Finland during the two study periods.
The relative reduction in inpatient RVGE incidence ranged from 86% in the 4-year-old children to 94% in the 0-year-old children, the researchers reported. That amounted to 843 prevented inpatient cases in children under 5 years.
The highest incidence of RVGE, which is rarely treated in hospital outpatients, was 0.4/1,000 person-years in children aged 1 year or younger. The relative reduction in outpatient RVGE incidence was 86% in the 0-year-olds and 100% in the 3- and 4-year-olds. The RV vaccination program prevented only 64 hospital outpatient cases in children younger than 5 years of age in 2014.
RV vaccination also resulted in a reduction of unspecified viral gastroenteritis (UVGE) incidence by 84%. Because the incidence of UVGE in 1-year-olds during the prevaccine era was more than twice the incidence of RVGE, the absolute reduction from 10.7 to 1.7 per 1,000 person-years “reflects a removal of a much larger disease burden than the reduction in the most specific outcome of inpatient RVGE,” the researchers said.
The annually prevented inpatient UVGE cases in children up to 4 years was 1,522, almost twice as many as the prevented inpatient RVGE cases. In the prevaccine era, the UVGE reduction was greatest among 1-year-olds, at 71%. In children younger than 5 years of age, 1,313 UVGE hospital outpatient cases were prevented in 2014.
“Considering secondary health care, the program annually pays for itself almost two times over in Finland,” the investigators concluded.
Read more in Vaccine (2017 Oct 9;35[42]:5611-7).
The researchers had no conflicts of interest.
cnellist@frontlinmedcom.com
under 5 years, and it more than adequately pays for itself in secondary health care costs, said Tuija Leino of the National Institute for Health and Welfare, Helsinki, and associates.
Since 2009, all Finnish children younger than 5 years have been offered an RV vaccine.
The investigators conducted a register-based study comparing the RV disease burden before and after introduction of RV vaccination, with the years 1999-2005 as the prevaccine period and the years 2010-2014 as the vaccination period. The study population was all children younger than 5 years living in Finland during the two study periods.
The relative reduction in inpatient RVGE incidence ranged from 86% in the 4-year-old children to 94% in the 0-year-old children, the researchers reported. That amounted to 843 prevented inpatient cases in children under 5 years.
The highest incidence of RVGE, which is rarely treated in hospital outpatients, was 0.4/1,000 person-years in children aged 1 year or younger. The relative reduction in outpatient RVGE incidence was 86% in the 0-year-olds and 100% in the 3- and 4-year-olds. The RV vaccination program prevented only 64 hospital outpatient cases in children younger than 5 years of age in 2014.
RV vaccination also resulted in a reduction of unspecified viral gastroenteritis (UVGE) incidence by 84%. Because the incidence of UVGE in 1-year-olds during the prevaccine era was more than twice the incidence of RVGE, the absolute reduction from 10.7 to 1.7 per 1,000 person-years “reflects a removal of a much larger disease burden than the reduction in the most specific outcome of inpatient RVGE,” the researchers said.
The annually prevented inpatient UVGE cases in children up to 4 years was 1,522, almost twice as many as the prevented inpatient RVGE cases. In the prevaccine era, the UVGE reduction was greatest among 1-year-olds, at 71%. In children younger than 5 years of age, 1,313 UVGE hospital outpatient cases were prevented in 2014.
“Considering secondary health care, the program annually pays for itself almost two times over in Finland,” the investigators concluded.
Read more in Vaccine (2017 Oct 9;35[42]:5611-7).
The researchers had no conflicts of interest.
cnellist@frontlinmedcom.com
under 5 years, and it more than adequately pays for itself in secondary health care costs, said Tuija Leino of the National Institute for Health and Welfare, Helsinki, and associates.
Since 2009, all Finnish children younger than 5 years have been offered an RV vaccine.
The investigators conducted a register-based study comparing the RV disease burden before and after introduction of RV vaccination, with the years 1999-2005 as the prevaccine period and the years 2010-2014 as the vaccination period. The study population was all children younger than 5 years living in Finland during the two study periods.
The relative reduction in inpatient RVGE incidence ranged from 86% in the 4-year-old children to 94% in the 0-year-old children, the researchers reported. That amounted to 843 prevented inpatient cases in children under 5 years.
The highest incidence of RVGE, which is rarely treated in hospital outpatients, was 0.4/1,000 person-years in children aged 1 year or younger. The relative reduction in outpatient RVGE incidence was 86% in the 0-year-olds and 100% in the 3- and 4-year-olds. The RV vaccination program prevented only 64 hospital outpatient cases in children younger than 5 years of age in 2014.
RV vaccination also resulted in a reduction of unspecified viral gastroenteritis (UVGE) incidence by 84%. Because the incidence of UVGE in 1-year-olds during the prevaccine era was more than twice the incidence of RVGE, the absolute reduction from 10.7 to 1.7 per 1,000 person-years “reflects a removal of a much larger disease burden than the reduction in the most specific outcome of inpatient RVGE,” the researchers said.
The annually prevented inpatient UVGE cases in children up to 4 years was 1,522, almost twice as many as the prevented inpatient RVGE cases. In the prevaccine era, the UVGE reduction was greatest among 1-year-olds, at 71%. In children younger than 5 years of age, 1,313 UVGE hospital outpatient cases were prevented in 2014.
“Considering secondary health care, the program annually pays for itself almost two times over in Finland,” the investigators concluded.
Read more in Vaccine (2017 Oct 9;35[42]:5611-7).
The researchers had no conflicts of interest.
cnellist@frontlinmedcom.com
from vaccine
Delivering Palliative Care in a Community Hospital: Experiences and Lessons Learned from the Front Lines
From the Division of Palliative Care, Butler Health System, Butler, PA (Drs. Stein, Reefer, Selvaggi, Ms. Doverspike); the University of Pittsburgh Medical Center, Pittsburgh, PA (Dr. Rajagopal); and the Duke Cancer Institute and Duke Fuqua School of Business, Durham, NC (Dr. Kamal).
Abstract
- Objective: To describe an approach to develop a community-centric palliative care program in a rural community health system and to review data collected over the program’s first year.
- Methods: We describe the underlying foundations of our program development including the health system’s prioritization of a palliative care program, funding opportunities, collaboration with community supports, and the importance of building a team and program that reflects a community’s needs. Data were collected through a program-maintained spreadsheet and a data monitoring system available through the Global Palliative Care Quality Alliance.
- Results: 516 new inpatient consultations were seen during the first year, for a penetration of 3.7%. The demographics of the patients who received consultation reflect that of the surrounding community. Over 50% of patients seen within the first year died, and hospice utilization at home and within facilities and inpatient hospice units increased. In addition, 79% of the patients seen by the palliative care team had a confirmed code status of do not resuscitate and do not intubate.
- Conclusions: Butler Health System’s approach to development of a palliative care program has resulted in increasing utilization of palliative care services in the hospital. Having hospital administration support, community support, and understanding the individualized needs of a community has been essential for the program’s expansion.
Key words: palliative care; program development; community hospital; rural.
Since its inception, palliative care has been committed to providing specialty-level consultation services to individuals with serious illness and their loved ones. The field has focused heavily on growth and acceptance, consistently moving upstream with regards to illness trajectory, across diseases, and across demographic variables such as age (eg, pediatric quality of life programs) and race (eg, community outreach programs addressing racial disparities in hospice use). An important frontier that remains challenging for much of the field is expansion into the community setting, where resources, implicit acceptance, and patient populations may vary.
As health system leaders appreciate the positive impacts palliative medicine on patient care and care quality, barriers to implementing palliative care programs in community hospitals must be addressed in ways tailored to the unique needs of smaller organizations and their communities. The goal of this paper is to outline the approach taken to develop Butler Health System’s community-centric palliative care program, describe our program’s underlying foundation rooted in community supports, and recount steps we have taken thus far to impact patient care in our hospital, health system, and community through the program’s first year.
Community Hospital Palliative Care—The Necessity and the Challenges
Palliative care has made strides in its growth and acceptance in the last decade; yet, the distribution of that growth has been skewed. Although 67% of hospitals now report access to specialist palliative care programs, most of the 148% growth over the last decade has been actualized in larger hospitals. Ninety percent of hospitals with greater than 300 beds report palliative care service availability whereas only 56% of small hospitals were identified to have this specialty care [1].
The inequity of access is also seen in other countries. A recent Canadian study retrospectively examined access to care of 23,860 deceased patients in Nova Scotia. Although they found 40.9% of study subjects were enrolled in a palliative care program at urban, academic centers, patients in a rural setting were only a third as likely to be enrolled in a palliative care program [2]. This access gap has important effects on patient-level outcomes, as evidence has consistently demonstrated that patients in rural settings who receive palliative care have decreased unnecessary hospitalizations and less in-hospital deaths [3].
While evidence of improved outcomes is strong, important barriers stand in the way. In a 2013 study, 374 health care providers at 236 rural hospitals in 7 states were interviewed to determine barriers to providing palliative care in rural settings. Barriers identified include a lack of administrative support, access to basic palliative care training for primary care physicians, and limited relationships to hospices [4]. Additional challenges include lack of access to tertiary-level specialty clinicians, access to and misconceptions about prescription medications, transportation for patients and providers, and incorporating a patient’s community supports [5–7].
Proposed Solutions
Techniques to improve palliative care access for rural and community centers that have been previously reviewed in the literature include videoconferencing with tertiary care experts in palliative care and education through small community-level lectures [8–10]. Goals of rural and suburban palliative care programs are broadly similar to programs at academic medical centers; however, few studies have identified impact of palliative medicine on patient care in community settings. In one suburban practice, a study found that patients were more likely to die at home if they had multiple caregivers, increased length of time under palliative care, and older age upon referral [11].
The United States has few large-scale pilot programs attempting to address the palliative needs of a more suburban or rural population. Of these, the Minnesota Rural Palliative Care Initiative developed by Stratis Health is perhaps the best publicized. Stratis Health developed and led an 18-month learning collaborative from October 2008 to April 2010 through which community teams developed or improved palliative care services. Through this initiative, a community-based health care practice model was developed that took advantage of the strong interrelationships within rural communities. After 18 months, 6 out of 10 rural Minnesota communities had formal palliative care programs, and 8 to 9 out of 10 had capabilities to at least address advance directives as well as provider and community education [12]. In another initiative, the NIH established a new suburban clinic with tertiary providers specifically for resource intensive, underserved patients [13]. The clinic was established by partnering with a service that was already in place in the community. Twenty-seven patients were seen within 7 months. The most common consults were patients with numerous comorbidities and chronic pain rather than terminal diagnoses. Given the intensive need of these patients, the authors felt that a consultation service and an interdisciplinary team that included psychosocial/spiritual/social work providers offered the most efficient method of delivering advanced palliative care needs.
The research regarding both solutions to challenges and novel methods of addressing the care gap remains sparse as evidenced by the conclusions of multiple systematic reviews and meta-analyses and the inability of the Cochrane review to find papers meeting inclusion criteria regarding techniques of community support in palliative care [14,15]. There remains a need to identify practical techniques of implementing palliative care in rural and suburban settings.
The Butler Health System Experience
In August 2015, we set out to start the first hospital-based palliative care consultation service in the Butler Health System. The health system is a nonprofit, single-hospital system anchored by Butler Memorial Hospital, a 294-bed community hospital located within a rural Pennsylvania county of 186,000 residents, 35 miles north of Pittsburgh. Butler County consists of a predominantly white, non-Hispanic population with over 15% of the residents being older than 65 years of age. The median household income is $61,000 earned primarily through blue collar occupations [16]. Driven by 53 employed primary care physicians, the health system provides services for 75,000 patients at sites covering an area of 4000 square miles. The hospital provides general medical, critical, surgical and subspecialty care and behavior health services as a regional referral center for 4 surrounding rural counties, accepting 12,500 inpatient admissions annually. A hospitalist service admits the majority of oncology patients, and the intensive care unit (ICU) is an open unit, where patients are admitted to the hospitalist, primary care, or surgical service.
While no formal needs assessment was performed prior to program development, perceptions of inadequate pain control, overuse of naloxone, underutilization of hospice services, and lack of consistent quality in end-of-life care were identified. These concerns were voiced at the levels of direct patient care on the floors, and by nursing and physician hospital leadership. Prior to our program, the chief medical officer attended the national Center to Advance Palliative Care conference to better understand the field of palliative care and its impact on improving quality of care. Concurrently, our health system was expanding its inpatient capabilities (eg, advanced neurologic and cardiac services), resulting in admissions with increased disease severity and illness complexity. With the vision of improved patient care, prioritizing quality end-of-life care and symptom management, the hospital board and administration overwhelmingly supported the development of the palliative care program, philosophically and financially.
Laying a Foundation—Funding, Collaboration, and Team Building
Funding and staffing are 2 important factors when building any program. Sources of funding for palliative care programs may include hospital support, billing revenue, grants, and philanthropy. Program development was a priority for the hospital and community. To help offset costs, efforts to raise financial support focused on utilizing the health system’s annual fundraising events. Through the generosity of individuals in the community, the hospital’s annual gala event, and donations from the hospital’s auxiliary, a total of $230,000 was raised prior to program initiation. Funds budgeted through direct hospital support and fundraising were allocated towards hiring palliative care team members and community marketing projects.
The hospital’s surrounding community is fortunate to have 2 local inpatient hospice facilities, and these relationships were imperative to providing quality end-of-life care preceding our palliative care program. A formal partnership was previously established with one while the other remains an important referral facility due to its proximity to the hospital. These hospice services are encouraged to participate in our weekly palliative care interdisciplinary team meetings. Their incorporation has improved coordination, continuity, and translation of care upon patient discharge from the acute hospital setting. Additionally, the relationships have been beneficial in tracking patients’ outcomes and data collection.
The standard structure of a palliative care team described by the Joint Commission and National Consensus Panel for Palliative Care consists of a physician, registered nurse or advanced practice provider, chaplaincy, and social work. Despite this recommendation, less than 40% of surveyed hospitals met the criteria, and less than 25% have dedicated funding to cover these positions [17]. Upon inception of our palliative care program, 2.6 funded full-time equivalents (FTEs) were allocated. These positions included a physician (1.0 FTE), a physician assistant (1.0 FTE), and a part time palliative care social worker (0.6 FTE). The 2015 National Palliative Care Registry found that 3.2 funded FTEs per 10,000 admissions is the average for hospitals with 150 to 299 beds [17]. The uncertainty of the utilization and consult volume, and the limited amount of qualified palliative care trained practitioners, resulted in the palliative program starting below this mean at 2.1 funded FTEs per 10,000 admissions. All the funded positions were located on site at the hospital. The pre-existing volunteer hospital chaplain service was identified as the pastoral care component for the program.
Increased FTEs have been associated with increased palliative care service penetration and ultimately in decreased time to consult [18]. In response to increasing consult volumes, concerns for delays in time to consult, and in preparation for expansion to an outpatient service, the palliative care department acquired an additional funded physician FTE (1.0). Ultimately the service reached a total of 3.6 FTE for inpatient services during its first 12 months; proportionately this resulted in an increase to 2.9 FTE per 10,000 admissions based on the yearly admission rate of 12,500 patients.
Educational Outreach
The success of a palliative care program depends on other clinicians’ acceptance and referral to the clinical program. We took a 2-pronged approach, focusing on both hospital-based and community-based education. The hospital-based nursing education included 30-minute presentations on general overviews of palliative care, differences between palliative care and hospice, and acute symptom management at the end of life. The palliative care team presented to all medical, surgical, and intensive care units and encompassed all shifts of nursing staff. These lectures included pre- and post-tests to assess for impact and feedback. Similar educational presentations, as well as an hour-long presentation on opioids and palliative care, were available for physicians for CME opportunities. We also distributed concise palliative care referral packets to outpatient primary care offices through the health system’s marketing team. The referral packets included examples of diagnoses, clinical scenarios, and symptoms to assist in the physicians’ understanding of palliative care services. The palliative care team also met with clinic office managers to discuss the program and answer questions.
There were also educational opportunities for patients and families in our community. Taking advantage of previously developed partnerships between the hospital system and local media outlets, the palliative care team performed local radio spots to educate the community on topics including an overview of palliative care, how to request palliative care, and the difference between palliative care and hospice care. We partnered with a local hospice agency and developed a well-received bereavement seminar for patients, family members, and employees and included the topic of advanced care planning.
Data Collection
We collect data using 2 different tools: a self-maintained spreadsheet shared between our palliative care clinicians, and a collective data tool (QDACT) included in our membership with and maintained by the Global Palliative Care Quality Alliance. Data collected and tracked in our spreadsheet includes date of consult, patient age, primary and secondary diagnoses, disposition, goals of care discussions, date of death, and 30-day readmissions. Through the QDACT data monitoring program, we are tracking and analyzing quality measures including symptom assessment and management and code status conversion. The QDACT database also provides financial data specific to our institution such as cost savings based on our billing, readmission rates, and length of stay.
Results
Projections, Volumes, and Penetration
Prior to the start of our program, our chief medical office used Center to Advance Palliative Care tools to project inpatient consultation volumes at our institution. Variables that are recommended by this center to guide projections include number of hospital admissions per year, hospital occupancy, disposition to hospice, as well as generalized estimations of inpatient mortality rates. Based on our data, it was expected that our program would receive 204 new inpatient consults in our first year, and 774 follow-up visits. Our actual new inpatient consults totaled 516, with 919 follow-up visits. Palliative care penetration (percentage of annual hospital admissions seen by the palliative care team) our first year was 3.7% (Table 1).
Consultation Demographics
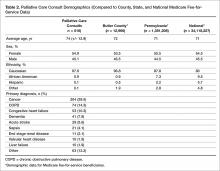
The demographics of the patients seen by the palliative care team reflect that of Butler County’s Medicare fee-for-service (FFS) population (Table 2); however, differences were seen at the state and national level with regard to ethnicity (Table 2).
Almost half of consultations (49%) were placed by the hospitalist service. Since the ICU is an open unit, critical care consults are not adequately reflected by analysis of the ordering physician alone. Analysis of consultation location revealed that 27% of inpatient consults were located within the ICU.
Patient Outcomes and Disposition
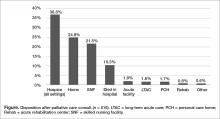
Outcomes and discharge data from the first year were collected and reviewed. Ten percent of the patients seen by palliative care died in the hospital, and 51% of patients that were seen by palliative care died within the program’s first year. Thirty-seven percent of patients discharged from the hospital utilized hospice services at home, in residential nursing facility, or at an inpatient hospice unit. The remaining 53% were discharged without hospice services to home or facility (Figure).
Hospice utilization by the health system increased during our first year. Compared to the 2014 calendar year, there were a total of 263 referrals for hospice services. During the first year of the palliative care program, which started August 2015, there were a total of 293 referrals. Of the 293 total hospice referrals, 190 (64.8%) of these referrals were for patients seen by the palliative care team.
Change of Code Status
Code status and changes in codes status data were collected. Of 462 individual patients prior to or at the time of palliative care consults, 43% were full code, 4% limited code, 8% unknown status, and 45% Do Not Resuscitate. After palliative care consult, 61% of the patients who were previously full/limited/unknown converted to do not resuscitate and do not intubate status. In total, 79% of patients seen by palliative care had a confirmed code status of Do Not Resuscitate and Do Not Intubate status after consult.
Discussion
In our first year, our palliative care program exceeded the expected number of inpatient consults, corresponding with a penetration of 3.7%. With the increase of funded FTEs, preliminary data shows that the department’s penetration continues to rise remaining consistent with the data and expectations [18]. During the second year, it is anticipated that over 600 inpatient palliative care consultations will be performed with an estimated penetration of 4%. This increasing penetration reflects the rising utilization of palliative care within our hospital. Since inception of the program, the service has expanded into an outpatient clinic 2 days per week. The palliative care clinic is staffed by a registered nurse (funded 0.6 FTE) and covered by the same physicians and physician assistant providing the inpatient services. The department acquired an unfunded but designated chaplaincy volunteer to assist with patients’ spiritual needs. We believe that the success of our program during the first year was related to multiple factors: a focus of integration and education by the palliative care department, health system administration buy-in, and identification of surrounding community needs.
In addition to patient care, our palliative care department also prioritizes “tangible” impacts to better establish our contributions to the health system. We have done this through participation on hospital committees, hospital policy revision teams, and by developing innovative solutions such as a terminal extubation protocol and order set for our ICU. The health system and its administration have recognized the importance of educating nursing and physician staff on palliative care services, and have supported these continued efforts alongside our clinical obligations.
Concurrent with administration buy-in, financial supports for our palliative care services were initially supplemented by the health system. Our department understands the importance of recognizing limitations of resources in communities and their hospitals. In efforts to minimize the department’s impact on our own health system’s financial resources, we have strived to offset our costs. We helped the hospital system meet pay-for-performance palliative care metrics set by the large local insurers resulting in financial hospital reimbursement valued at $600,000 in 2016.
The question of how the program may translate into other communities raises a major limitation: the homogeneity our population. The community surrounding the hospital is primarily Caucasian, with minimal representation of minority populations. While the patient population seen by our palliative care team is reflective of our surrounding county, it does not represent Medicare FFS beneficiaries on a national level or many other types of community hospitals across the country. Variations of ethnicity, age, diagnoses, and faith are fundamental, which highlights the importance of understanding the community in which a program is developed.
The rising trajectory of our palliative care service utilization has prompted a discussion of future endeavors for our program. Expectations for a continued shortage of hospice and palliative care physicians [19] and concerns for practitioner burnout [20] underlie our thoughtful approach to expansion of inpatient and outpatient services. At this time, potential projects include a consultation trigger system and incorporation of palliative care providers in ICU rounding, as well as possible expansion of outpatient services through implantation of an advanced practitioner into surrounding nursing homes and primary care offices.
We have found a growing utilization of our program at Butler Health System. Our first year experience has highlighted the importance of identifying community and hospital administrative champions as a foundation. Additionally, understanding the specific characteristics of one’s surrounding community may allow for improved integration and acceptance of palliative care in a community setting. Our program continues to work with the health system, community, and philanthropic organizations to expand the ever-growing need for palliative care services.
1. Dumanovsky T, Augustin R, Rogers M, et al. The growth of palliative care in U.S. hospitals: a status report. J Palliat Med 2016;19:8–15.
2. Lavergne MR, Lethbridge L, Johnston G, et al. Examining palliative care program use and place of death in rural and urban contexts: a Canadian population-based study using linked data. Rural Remote Health 2015;15:3134.
3. Seow H, Brazil K, Sussman J, et al. Impact of community based, specialist palliative care teams on hospitalisations and emergency department visits late in life and hospital deaths: a pooled analysis. BMJ 2014;348:g3496.
4. Fink RM, Oman KS, Youngwerth J, et al. A palliative care needs assessment of rural hospitals. J Palliat Med 2013;16:638–44.
5. Dumont S, Jacobs P, Turcotte V, et al. Palliative care costs in Canada: A descriptive comparison of studies of urban and rural patients near end of life. J Palliat Med 2015;29:908–17.
6. Kaasalainen S, Brazil K, Williams A, et al. Nurses' experiences providing palliative care to individuals living in rural communities: aspects of the physical residential setting. Rural Remote Health 2014;14:2728.
7. Ahmed N, Bestall JC, Ahmedzai SH, et al. Systematic review of the problems and issues of accessing specialist palliative care by patients, carers and health and social care professionals. J Palliat Med 2004;18:525–42.
8. Ray RA, Fried O, Lindsay D. Palliative care professional education via video conference builds confidence to deliver palliative care in rural and remote locations. BMC Health Serv Res 2014;14:272.
9. Bakitas MA, Elk R, Astin M, et al. Systematic review of palliative care in the rural setting. Cancer Control 2015;22:450–64.
10. Akiyama M, Hirai K, Takebayashi T, et al. The effects of community-wide dissemination of information on perceptions of palliative care, knowledge about opioids, and sense of security among cancer patients, their families, and the general public. Support Care Cancer 2016;24: 347–56.
11. Maida V. Factors that promote success in home palliative care: a study of a large suburban palliative care practice. J Palliat Care 2002;18:282–6.
12. Ceronsky L, Shearer J, Weng K, et al. Minnesota Rural Palliative Care Initiative: building palliative care capacity in rural Minnesota. J Palliat Med 2013;16:310–3.
13. Aggarwal SK, Ghosh A, Cheng MJ, et al. Initiating pain and palliative care outpatient services for the suburban underserved in Montgomery County, Maryland: Lessons learned at the NIH Clinical Center and MobileMed. Palliat Support Care 2015;16:1–6.
14. Rainsford S, MacLeod RD, Glasgow NJ. Place of death in rural palliative care: A systematic review. J Palliat Med 2016;30:745–63.
15. Horey D, Street AF, O'Connor M, et al. Training and supportive programs for palliative care volunteers in community settings. Cochrane Database Syst Rev 2015 Jul 20;(7):CD009500.
16. The United States Census Bureau: QuickFacts: Butler County, Pennsylvania. Accessed 10 Mar 2017 at www.census.gov/quickfacts/table/PST045216/42019,00.
17. Spetz J, Dudley N, Trupin L, et al. Few hospital palliative care programs meet national staffing recommendations. Health Aff 2016;35:1690–7.
18. Dumanovsky T, Rogers M, Spragens LH, et al. Impact of Staffing on Access to Palliative Care in U.S. Hospitals. J Palliat Med. 2015 Dec; 18(12). Pages 998-999.
19. Lupu D, American Academy of Hospice and Palliative Medicine Workforce Task Force. Estimate of current hospice and palliative medicine physician workforce shortage. J Pain Symptom Manage 2010;40:899–911.
20. Kamal AH, Bull JK, Wolf SP, et al. Prevalence and predictors of burnout among hospice and palliative care clinicians in the U.S. J Pain Symptom Manage 2016;51:690–6.
From the Division of Palliative Care, Butler Health System, Butler, PA (Drs. Stein, Reefer, Selvaggi, Ms. Doverspike); the University of Pittsburgh Medical Center, Pittsburgh, PA (Dr. Rajagopal); and the Duke Cancer Institute and Duke Fuqua School of Business, Durham, NC (Dr. Kamal).
Abstract
- Objective: To describe an approach to develop a community-centric palliative care program in a rural community health system and to review data collected over the program’s first year.
- Methods: We describe the underlying foundations of our program development including the health system’s prioritization of a palliative care program, funding opportunities, collaboration with community supports, and the importance of building a team and program that reflects a community’s needs. Data were collected through a program-maintained spreadsheet and a data monitoring system available through the Global Palliative Care Quality Alliance.
- Results: 516 new inpatient consultations were seen during the first year, for a penetration of 3.7%. The demographics of the patients who received consultation reflect that of the surrounding community. Over 50% of patients seen within the first year died, and hospice utilization at home and within facilities and inpatient hospice units increased. In addition, 79% of the patients seen by the palliative care team had a confirmed code status of do not resuscitate and do not intubate.
- Conclusions: Butler Health System’s approach to development of a palliative care program has resulted in increasing utilization of palliative care services in the hospital. Having hospital administration support, community support, and understanding the individualized needs of a community has been essential for the program’s expansion.
Key words: palliative care; program development; community hospital; rural.
Since its inception, palliative care has been committed to providing specialty-level consultation services to individuals with serious illness and their loved ones. The field has focused heavily on growth and acceptance, consistently moving upstream with regards to illness trajectory, across diseases, and across demographic variables such as age (eg, pediatric quality of life programs) and race (eg, community outreach programs addressing racial disparities in hospice use). An important frontier that remains challenging for much of the field is expansion into the community setting, where resources, implicit acceptance, and patient populations may vary.
As health system leaders appreciate the positive impacts palliative medicine on patient care and care quality, barriers to implementing palliative care programs in community hospitals must be addressed in ways tailored to the unique needs of smaller organizations and their communities. The goal of this paper is to outline the approach taken to develop Butler Health System’s community-centric palliative care program, describe our program’s underlying foundation rooted in community supports, and recount steps we have taken thus far to impact patient care in our hospital, health system, and community through the program’s first year.
Community Hospital Palliative Care—The Necessity and the Challenges
Palliative care has made strides in its growth and acceptance in the last decade; yet, the distribution of that growth has been skewed. Although 67% of hospitals now report access to specialist palliative care programs, most of the 148% growth over the last decade has been actualized in larger hospitals. Ninety percent of hospitals with greater than 300 beds report palliative care service availability whereas only 56% of small hospitals were identified to have this specialty care [1].
The inequity of access is also seen in other countries. A recent Canadian study retrospectively examined access to care of 23,860 deceased patients in Nova Scotia. Although they found 40.9% of study subjects were enrolled in a palliative care program at urban, academic centers, patients in a rural setting were only a third as likely to be enrolled in a palliative care program [2]. This access gap has important effects on patient-level outcomes, as evidence has consistently demonstrated that patients in rural settings who receive palliative care have decreased unnecessary hospitalizations and less in-hospital deaths [3].
While evidence of improved outcomes is strong, important barriers stand in the way. In a 2013 study, 374 health care providers at 236 rural hospitals in 7 states were interviewed to determine barriers to providing palliative care in rural settings. Barriers identified include a lack of administrative support, access to basic palliative care training for primary care physicians, and limited relationships to hospices [4]. Additional challenges include lack of access to tertiary-level specialty clinicians, access to and misconceptions about prescription medications, transportation for patients and providers, and incorporating a patient’s community supports [5–7].
Proposed Solutions
Techniques to improve palliative care access for rural and community centers that have been previously reviewed in the literature include videoconferencing with tertiary care experts in palliative care and education through small community-level lectures [8–10]. Goals of rural and suburban palliative care programs are broadly similar to programs at academic medical centers; however, few studies have identified impact of palliative medicine on patient care in community settings. In one suburban practice, a study found that patients were more likely to die at home if they had multiple caregivers, increased length of time under palliative care, and older age upon referral [11].
The United States has few large-scale pilot programs attempting to address the palliative needs of a more suburban or rural population. Of these, the Minnesota Rural Palliative Care Initiative developed by Stratis Health is perhaps the best publicized. Stratis Health developed and led an 18-month learning collaborative from October 2008 to April 2010 through which community teams developed or improved palliative care services. Through this initiative, a community-based health care practice model was developed that took advantage of the strong interrelationships within rural communities. After 18 months, 6 out of 10 rural Minnesota communities had formal palliative care programs, and 8 to 9 out of 10 had capabilities to at least address advance directives as well as provider and community education [12]. In another initiative, the NIH established a new suburban clinic with tertiary providers specifically for resource intensive, underserved patients [13]. The clinic was established by partnering with a service that was already in place in the community. Twenty-seven patients were seen within 7 months. The most common consults were patients with numerous comorbidities and chronic pain rather than terminal diagnoses. Given the intensive need of these patients, the authors felt that a consultation service and an interdisciplinary team that included psychosocial/spiritual/social work providers offered the most efficient method of delivering advanced palliative care needs.
The research regarding both solutions to challenges and novel methods of addressing the care gap remains sparse as evidenced by the conclusions of multiple systematic reviews and meta-analyses and the inability of the Cochrane review to find papers meeting inclusion criteria regarding techniques of community support in palliative care [14,15]. There remains a need to identify practical techniques of implementing palliative care in rural and suburban settings.
The Butler Health System Experience
In August 2015, we set out to start the first hospital-based palliative care consultation service in the Butler Health System. The health system is a nonprofit, single-hospital system anchored by Butler Memorial Hospital, a 294-bed community hospital located within a rural Pennsylvania county of 186,000 residents, 35 miles north of Pittsburgh. Butler County consists of a predominantly white, non-Hispanic population with over 15% of the residents being older than 65 years of age. The median household income is $61,000 earned primarily through blue collar occupations [16]. Driven by 53 employed primary care physicians, the health system provides services for 75,000 patients at sites covering an area of 4000 square miles. The hospital provides general medical, critical, surgical and subspecialty care and behavior health services as a regional referral center for 4 surrounding rural counties, accepting 12,500 inpatient admissions annually. A hospitalist service admits the majority of oncology patients, and the intensive care unit (ICU) is an open unit, where patients are admitted to the hospitalist, primary care, or surgical service.
While no formal needs assessment was performed prior to program development, perceptions of inadequate pain control, overuse of naloxone, underutilization of hospice services, and lack of consistent quality in end-of-life care were identified. These concerns were voiced at the levels of direct patient care on the floors, and by nursing and physician hospital leadership. Prior to our program, the chief medical officer attended the national Center to Advance Palliative Care conference to better understand the field of palliative care and its impact on improving quality of care. Concurrently, our health system was expanding its inpatient capabilities (eg, advanced neurologic and cardiac services), resulting in admissions with increased disease severity and illness complexity. With the vision of improved patient care, prioritizing quality end-of-life care and symptom management, the hospital board and administration overwhelmingly supported the development of the palliative care program, philosophically and financially.
Laying a Foundation—Funding, Collaboration, and Team Building
Funding and staffing are 2 important factors when building any program. Sources of funding for palliative care programs may include hospital support, billing revenue, grants, and philanthropy. Program development was a priority for the hospital and community. To help offset costs, efforts to raise financial support focused on utilizing the health system’s annual fundraising events. Through the generosity of individuals in the community, the hospital’s annual gala event, and donations from the hospital’s auxiliary, a total of $230,000 was raised prior to program initiation. Funds budgeted through direct hospital support and fundraising were allocated towards hiring palliative care team members and community marketing projects.
The hospital’s surrounding community is fortunate to have 2 local inpatient hospice facilities, and these relationships were imperative to providing quality end-of-life care preceding our palliative care program. A formal partnership was previously established with one while the other remains an important referral facility due to its proximity to the hospital. These hospice services are encouraged to participate in our weekly palliative care interdisciplinary team meetings. Their incorporation has improved coordination, continuity, and translation of care upon patient discharge from the acute hospital setting. Additionally, the relationships have been beneficial in tracking patients’ outcomes and data collection.
The standard structure of a palliative care team described by the Joint Commission and National Consensus Panel for Palliative Care consists of a physician, registered nurse or advanced practice provider, chaplaincy, and social work. Despite this recommendation, less than 40% of surveyed hospitals met the criteria, and less than 25% have dedicated funding to cover these positions [17]. Upon inception of our palliative care program, 2.6 funded full-time equivalents (FTEs) were allocated. These positions included a physician (1.0 FTE), a physician assistant (1.0 FTE), and a part time palliative care social worker (0.6 FTE). The 2015 National Palliative Care Registry found that 3.2 funded FTEs per 10,000 admissions is the average for hospitals with 150 to 299 beds [17]. The uncertainty of the utilization and consult volume, and the limited amount of qualified palliative care trained practitioners, resulted in the palliative program starting below this mean at 2.1 funded FTEs per 10,000 admissions. All the funded positions were located on site at the hospital. The pre-existing volunteer hospital chaplain service was identified as the pastoral care component for the program.
Increased FTEs have been associated with increased palliative care service penetration and ultimately in decreased time to consult [18]. In response to increasing consult volumes, concerns for delays in time to consult, and in preparation for expansion to an outpatient service, the palliative care department acquired an additional funded physician FTE (1.0). Ultimately the service reached a total of 3.6 FTE for inpatient services during its first 12 months; proportionately this resulted in an increase to 2.9 FTE per 10,000 admissions based on the yearly admission rate of 12,500 patients.
Educational Outreach
The success of a palliative care program depends on other clinicians’ acceptance and referral to the clinical program. We took a 2-pronged approach, focusing on both hospital-based and community-based education. The hospital-based nursing education included 30-minute presentations on general overviews of palliative care, differences between palliative care and hospice, and acute symptom management at the end of life. The palliative care team presented to all medical, surgical, and intensive care units and encompassed all shifts of nursing staff. These lectures included pre- and post-tests to assess for impact and feedback. Similar educational presentations, as well as an hour-long presentation on opioids and palliative care, were available for physicians for CME opportunities. We also distributed concise palliative care referral packets to outpatient primary care offices through the health system’s marketing team. The referral packets included examples of diagnoses, clinical scenarios, and symptoms to assist in the physicians’ understanding of palliative care services. The palliative care team also met with clinic office managers to discuss the program and answer questions.
There were also educational opportunities for patients and families in our community. Taking advantage of previously developed partnerships between the hospital system and local media outlets, the palliative care team performed local radio spots to educate the community on topics including an overview of palliative care, how to request palliative care, and the difference between palliative care and hospice care. We partnered with a local hospice agency and developed a well-received bereavement seminar for patients, family members, and employees and included the topic of advanced care planning.
Data Collection
We collect data using 2 different tools: a self-maintained spreadsheet shared between our palliative care clinicians, and a collective data tool (QDACT) included in our membership with and maintained by the Global Palliative Care Quality Alliance. Data collected and tracked in our spreadsheet includes date of consult, patient age, primary and secondary diagnoses, disposition, goals of care discussions, date of death, and 30-day readmissions. Through the QDACT data monitoring program, we are tracking and analyzing quality measures including symptom assessment and management and code status conversion. The QDACT database also provides financial data specific to our institution such as cost savings based on our billing, readmission rates, and length of stay.
Results
Projections, Volumes, and Penetration
Prior to the start of our program, our chief medical office used Center to Advance Palliative Care tools to project inpatient consultation volumes at our institution. Variables that are recommended by this center to guide projections include number of hospital admissions per year, hospital occupancy, disposition to hospice, as well as generalized estimations of inpatient mortality rates. Based on our data, it was expected that our program would receive 204 new inpatient consults in our first year, and 774 follow-up visits. Our actual new inpatient consults totaled 516, with 919 follow-up visits. Palliative care penetration (percentage of annual hospital admissions seen by the palliative care team) our first year was 3.7% (Table 1).
Consultation Demographics
The demographics of the patients seen by the palliative care team reflect that of Butler County’s Medicare fee-for-service (FFS) population (Table 2); however, differences were seen at the state and national level with regard to ethnicity (Table 2).
Almost half of consultations (49%) were placed by the hospitalist service. Since the ICU is an open unit, critical care consults are not adequately reflected by analysis of the ordering physician alone. Analysis of consultation location revealed that 27% of inpatient consults were located within the ICU.
Patient Outcomes and Disposition
Outcomes and discharge data from the first year were collected and reviewed. Ten percent of the patients seen by palliative care died in the hospital, and 51% of patients that were seen by palliative care died within the program’s first year. Thirty-seven percent of patients discharged from the hospital utilized hospice services at home, in residential nursing facility, or at an inpatient hospice unit. The remaining 53% were discharged without hospice services to home or facility (Figure).
Hospice utilization by the health system increased during our first year. Compared to the 2014 calendar year, there were a total of 263 referrals for hospice services. During the first year of the palliative care program, which started August 2015, there were a total of 293 referrals. Of the 293 total hospice referrals, 190 (64.8%) of these referrals were for patients seen by the palliative care team.
Change of Code Status
Code status and changes in codes status data were collected. Of 462 individual patients prior to or at the time of palliative care consults, 43% were full code, 4% limited code, 8% unknown status, and 45% Do Not Resuscitate. After palliative care consult, 61% of the patients who were previously full/limited/unknown converted to do not resuscitate and do not intubate status. In total, 79% of patients seen by palliative care had a confirmed code status of Do Not Resuscitate and Do Not Intubate status after consult.
Discussion
In our first year, our palliative care program exceeded the expected number of inpatient consults, corresponding with a penetration of 3.7%. With the increase of funded FTEs, preliminary data shows that the department’s penetration continues to rise remaining consistent with the data and expectations [18]. During the second year, it is anticipated that over 600 inpatient palliative care consultations will be performed with an estimated penetration of 4%. This increasing penetration reflects the rising utilization of palliative care within our hospital. Since inception of the program, the service has expanded into an outpatient clinic 2 days per week. The palliative care clinic is staffed by a registered nurse (funded 0.6 FTE) and covered by the same physicians and physician assistant providing the inpatient services. The department acquired an unfunded but designated chaplaincy volunteer to assist with patients’ spiritual needs. We believe that the success of our program during the first year was related to multiple factors: a focus of integration and education by the palliative care department, health system administration buy-in, and identification of surrounding community needs.
In addition to patient care, our palliative care department also prioritizes “tangible” impacts to better establish our contributions to the health system. We have done this through participation on hospital committees, hospital policy revision teams, and by developing innovative solutions such as a terminal extubation protocol and order set for our ICU. The health system and its administration have recognized the importance of educating nursing and physician staff on palliative care services, and have supported these continued efforts alongside our clinical obligations.
Concurrent with administration buy-in, financial supports for our palliative care services were initially supplemented by the health system. Our department understands the importance of recognizing limitations of resources in communities and their hospitals. In efforts to minimize the department’s impact on our own health system’s financial resources, we have strived to offset our costs. We helped the hospital system meet pay-for-performance palliative care metrics set by the large local insurers resulting in financial hospital reimbursement valued at $600,000 in 2016.
The question of how the program may translate into other communities raises a major limitation: the homogeneity our population. The community surrounding the hospital is primarily Caucasian, with minimal representation of minority populations. While the patient population seen by our palliative care team is reflective of our surrounding county, it does not represent Medicare FFS beneficiaries on a national level or many other types of community hospitals across the country. Variations of ethnicity, age, diagnoses, and faith are fundamental, which highlights the importance of understanding the community in which a program is developed.
The rising trajectory of our palliative care service utilization has prompted a discussion of future endeavors for our program. Expectations for a continued shortage of hospice and palliative care physicians [19] and concerns for practitioner burnout [20] underlie our thoughtful approach to expansion of inpatient and outpatient services. At this time, potential projects include a consultation trigger system and incorporation of palliative care providers in ICU rounding, as well as possible expansion of outpatient services through implantation of an advanced practitioner into surrounding nursing homes and primary care offices.
We have found a growing utilization of our program at Butler Health System. Our first year experience has highlighted the importance of identifying community and hospital administrative champions as a foundation. Additionally, understanding the specific characteristics of one’s surrounding community may allow for improved integration and acceptance of palliative care in a community setting. Our program continues to work with the health system, community, and philanthropic organizations to expand the ever-growing need for palliative care services.
From the Division of Palliative Care, Butler Health System, Butler, PA (Drs. Stein, Reefer, Selvaggi, Ms. Doverspike); the University of Pittsburgh Medical Center, Pittsburgh, PA (Dr. Rajagopal); and the Duke Cancer Institute and Duke Fuqua School of Business, Durham, NC (Dr. Kamal).
Abstract
- Objective: To describe an approach to develop a community-centric palliative care program in a rural community health system and to review data collected over the program’s first year.
- Methods: We describe the underlying foundations of our program development including the health system’s prioritization of a palliative care program, funding opportunities, collaboration with community supports, and the importance of building a team and program that reflects a community’s needs. Data were collected through a program-maintained spreadsheet and a data monitoring system available through the Global Palliative Care Quality Alliance.
- Results: 516 new inpatient consultations were seen during the first year, for a penetration of 3.7%. The demographics of the patients who received consultation reflect that of the surrounding community. Over 50% of patients seen within the first year died, and hospice utilization at home and within facilities and inpatient hospice units increased. In addition, 79% of the patients seen by the palliative care team had a confirmed code status of do not resuscitate and do not intubate.
- Conclusions: Butler Health System’s approach to development of a palliative care program has resulted in increasing utilization of palliative care services in the hospital. Having hospital administration support, community support, and understanding the individualized needs of a community has been essential for the program’s expansion.
Key words: palliative care; program development; community hospital; rural.
Since its inception, palliative care has been committed to providing specialty-level consultation services to individuals with serious illness and their loved ones. The field has focused heavily on growth and acceptance, consistently moving upstream with regards to illness trajectory, across diseases, and across demographic variables such as age (eg, pediatric quality of life programs) and race (eg, community outreach programs addressing racial disparities in hospice use). An important frontier that remains challenging for much of the field is expansion into the community setting, where resources, implicit acceptance, and patient populations may vary.
As health system leaders appreciate the positive impacts palliative medicine on patient care and care quality, barriers to implementing palliative care programs in community hospitals must be addressed in ways tailored to the unique needs of smaller organizations and their communities. The goal of this paper is to outline the approach taken to develop Butler Health System’s community-centric palliative care program, describe our program’s underlying foundation rooted in community supports, and recount steps we have taken thus far to impact patient care in our hospital, health system, and community through the program’s first year.
Community Hospital Palliative Care—The Necessity and the Challenges
Palliative care has made strides in its growth and acceptance in the last decade; yet, the distribution of that growth has been skewed. Although 67% of hospitals now report access to specialist palliative care programs, most of the 148% growth over the last decade has been actualized in larger hospitals. Ninety percent of hospitals with greater than 300 beds report palliative care service availability whereas only 56% of small hospitals were identified to have this specialty care [1].
The inequity of access is also seen in other countries. A recent Canadian study retrospectively examined access to care of 23,860 deceased patients in Nova Scotia. Although they found 40.9% of study subjects were enrolled in a palliative care program at urban, academic centers, patients in a rural setting were only a third as likely to be enrolled in a palliative care program [2]. This access gap has important effects on patient-level outcomes, as evidence has consistently demonstrated that patients in rural settings who receive palliative care have decreased unnecessary hospitalizations and less in-hospital deaths [3].
While evidence of improved outcomes is strong, important barriers stand in the way. In a 2013 study, 374 health care providers at 236 rural hospitals in 7 states were interviewed to determine barriers to providing palliative care in rural settings. Barriers identified include a lack of administrative support, access to basic palliative care training for primary care physicians, and limited relationships to hospices [4]. Additional challenges include lack of access to tertiary-level specialty clinicians, access to and misconceptions about prescription medications, transportation for patients and providers, and incorporating a patient’s community supports [5–7].
Proposed Solutions
Techniques to improve palliative care access for rural and community centers that have been previously reviewed in the literature include videoconferencing with tertiary care experts in palliative care and education through small community-level lectures [8–10]. Goals of rural and suburban palliative care programs are broadly similar to programs at academic medical centers; however, few studies have identified impact of palliative medicine on patient care in community settings. In one suburban practice, a study found that patients were more likely to die at home if they had multiple caregivers, increased length of time under palliative care, and older age upon referral [11].
The United States has few large-scale pilot programs attempting to address the palliative needs of a more suburban or rural population. Of these, the Minnesota Rural Palliative Care Initiative developed by Stratis Health is perhaps the best publicized. Stratis Health developed and led an 18-month learning collaborative from October 2008 to April 2010 through which community teams developed or improved palliative care services. Through this initiative, a community-based health care practice model was developed that took advantage of the strong interrelationships within rural communities. After 18 months, 6 out of 10 rural Minnesota communities had formal palliative care programs, and 8 to 9 out of 10 had capabilities to at least address advance directives as well as provider and community education [12]. In another initiative, the NIH established a new suburban clinic with tertiary providers specifically for resource intensive, underserved patients [13]. The clinic was established by partnering with a service that was already in place in the community. Twenty-seven patients were seen within 7 months. The most common consults were patients with numerous comorbidities and chronic pain rather than terminal diagnoses. Given the intensive need of these patients, the authors felt that a consultation service and an interdisciplinary team that included psychosocial/spiritual/social work providers offered the most efficient method of delivering advanced palliative care needs.
The research regarding both solutions to challenges and novel methods of addressing the care gap remains sparse as evidenced by the conclusions of multiple systematic reviews and meta-analyses and the inability of the Cochrane review to find papers meeting inclusion criteria regarding techniques of community support in palliative care [14,15]. There remains a need to identify practical techniques of implementing palliative care in rural and suburban settings.
The Butler Health System Experience
In August 2015, we set out to start the first hospital-based palliative care consultation service in the Butler Health System. The health system is a nonprofit, single-hospital system anchored by Butler Memorial Hospital, a 294-bed community hospital located within a rural Pennsylvania county of 186,000 residents, 35 miles north of Pittsburgh. Butler County consists of a predominantly white, non-Hispanic population with over 15% of the residents being older than 65 years of age. The median household income is $61,000 earned primarily through blue collar occupations [16]. Driven by 53 employed primary care physicians, the health system provides services for 75,000 patients at sites covering an area of 4000 square miles. The hospital provides general medical, critical, surgical and subspecialty care and behavior health services as a regional referral center for 4 surrounding rural counties, accepting 12,500 inpatient admissions annually. A hospitalist service admits the majority of oncology patients, and the intensive care unit (ICU) is an open unit, where patients are admitted to the hospitalist, primary care, or surgical service.
While no formal needs assessment was performed prior to program development, perceptions of inadequate pain control, overuse of naloxone, underutilization of hospice services, and lack of consistent quality in end-of-life care were identified. These concerns were voiced at the levels of direct patient care on the floors, and by nursing and physician hospital leadership. Prior to our program, the chief medical officer attended the national Center to Advance Palliative Care conference to better understand the field of palliative care and its impact on improving quality of care. Concurrently, our health system was expanding its inpatient capabilities (eg, advanced neurologic and cardiac services), resulting in admissions with increased disease severity and illness complexity. With the vision of improved patient care, prioritizing quality end-of-life care and symptom management, the hospital board and administration overwhelmingly supported the development of the palliative care program, philosophically and financially.
Laying a Foundation—Funding, Collaboration, and Team Building
Funding and staffing are 2 important factors when building any program. Sources of funding for palliative care programs may include hospital support, billing revenue, grants, and philanthropy. Program development was a priority for the hospital and community. To help offset costs, efforts to raise financial support focused on utilizing the health system’s annual fundraising events. Through the generosity of individuals in the community, the hospital’s annual gala event, and donations from the hospital’s auxiliary, a total of $230,000 was raised prior to program initiation. Funds budgeted through direct hospital support and fundraising were allocated towards hiring palliative care team members and community marketing projects.
The hospital’s surrounding community is fortunate to have 2 local inpatient hospice facilities, and these relationships were imperative to providing quality end-of-life care preceding our palliative care program. A formal partnership was previously established with one while the other remains an important referral facility due to its proximity to the hospital. These hospice services are encouraged to participate in our weekly palliative care interdisciplinary team meetings. Their incorporation has improved coordination, continuity, and translation of care upon patient discharge from the acute hospital setting. Additionally, the relationships have been beneficial in tracking patients’ outcomes and data collection.
The standard structure of a palliative care team described by the Joint Commission and National Consensus Panel for Palliative Care consists of a physician, registered nurse or advanced practice provider, chaplaincy, and social work. Despite this recommendation, less than 40% of surveyed hospitals met the criteria, and less than 25% have dedicated funding to cover these positions [17]. Upon inception of our palliative care program, 2.6 funded full-time equivalents (FTEs) were allocated. These positions included a physician (1.0 FTE), a physician assistant (1.0 FTE), and a part time palliative care social worker (0.6 FTE). The 2015 National Palliative Care Registry found that 3.2 funded FTEs per 10,000 admissions is the average for hospitals with 150 to 299 beds [17]. The uncertainty of the utilization and consult volume, and the limited amount of qualified palliative care trained practitioners, resulted in the palliative program starting below this mean at 2.1 funded FTEs per 10,000 admissions. All the funded positions were located on site at the hospital. The pre-existing volunteer hospital chaplain service was identified as the pastoral care component for the program.
Increased FTEs have been associated with increased palliative care service penetration and ultimately in decreased time to consult [18]. In response to increasing consult volumes, concerns for delays in time to consult, and in preparation for expansion to an outpatient service, the palliative care department acquired an additional funded physician FTE (1.0). Ultimately the service reached a total of 3.6 FTE for inpatient services during its first 12 months; proportionately this resulted in an increase to 2.9 FTE per 10,000 admissions based on the yearly admission rate of 12,500 patients.
Educational Outreach
The success of a palliative care program depends on other clinicians’ acceptance and referral to the clinical program. We took a 2-pronged approach, focusing on both hospital-based and community-based education. The hospital-based nursing education included 30-minute presentations on general overviews of palliative care, differences between palliative care and hospice, and acute symptom management at the end of life. The palliative care team presented to all medical, surgical, and intensive care units and encompassed all shifts of nursing staff. These lectures included pre- and post-tests to assess for impact and feedback. Similar educational presentations, as well as an hour-long presentation on opioids and palliative care, were available for physicians for CME opportunities. We also distributed concise palliative care referral packets to outpatient primary care offices through the health system’s marketing team. The referral packets included examples of diagnoses, clinical scenarios, and symptoms to assist in the physicians’ understanding of palliative care services. The palliative care team also met with clinic office managers to discuss the program and answer questions.
There were also educational opportunities for patients and families in our community. Taking advantage of previously developed partnerships between the hospital system and local media outlets, the palliative care team performed local radio spots to educate the community on topics including an overview of palliative care, how to request palliative care, and the difference between palliative care and hospice care. We partnered with a local hospice agency and developed a well-received bereavement seminar for patients, family members, and employees and included the topic of advanced care planning.
Data Collection
We collect data using 2 different tools: a self-maintained spreadsheet shared between our palliative care clinicians, and a collective data tool (QDACT) included in our membership with and maintained by the Global Palliative Care Quality Alliance. Data collected and tracked in our spreadsheet includes date of consult, patient age, primary and secondary diagnoses, disposition, goals of care discussions, date of death, and 30-day readmissions. Through the QDACT data monitoring program, we are tracking and analyzing quality measures including symptom assessment and management and code status conversion. The QDACT database also provides financial data specific to our institution such as cost savings based on our billing, readmission rates, and length of stay.
Results
Projections, Volumes, and Penetration
Prior to the start of our program, our chief medical office used Center to Advance Palliative Care tools to project inpatient consultation volumes at our institution. Variables that are recommended by this center to guide projections include number of hospital admissions per year, hospital occupancy, disposition to hospice, as well as generalized estimations of inpatient mortality rates. Based on our data, it was expected that our program would receive 204 new inpatient consults in our first year, and 774 follow-up visits. Our actual new inpatient consults totaled 516, with 919 follow-up visits. Palliative care penetration (percentage of annual hospital admissions seen by the palliative care team) our first year was 3.7% (Table 1).
Consultation Demographics
The demographics of the patients seen by the palliative care team reflect that of Butler County’s Medicare fee-for-service (FFS) population (Table 2); however, differences were seen at the state and national level with regard to ethnicity (Table 2).
Almost half of consultations (49%) were placed by the hospitalist service. Since the ICU is an open unit, critical care consults are not adequately reflected by analysis of the ordering physician alone. Analysis of consultation location revealed that 27% of inpatient consults were located within the ICU.
Patient Outcomes and Disposition
Outcomes and discharge data from the first year were collected and reviewed. Ten percent of the patients seen by palliative care died in the hospital, and 51% of patients that were seen by palliative care died within the program’s first year. Thirty-seven percent of patients discharged from the hospital utilized hospice services at home, in residential nursing facility, or at an inpatient hospice unit. The remaining 53% were discharged without hospice services to home or facility (Figure).
Hospice utilization by the health system increased during our first year. Compared to the 2014 calendar year, there were a total of 263 referrals for hospice services. During the first year of the palliative care program, which started August 2015, there were a total of 293 referrals. Of the 293 total hospice referrals, 190 (64.8%) of these referrals were for patients seen by the palliative care team.
Change of Code Status
Code status and changes in codes status data were collected. Of 462 individual patients prior to or at the time of palliative care consults, 43% were full code, 4% limited code, 8% unknown status, and 45% Do Not Resuscitate. After palliative care consult, 61% of the patients who were previously full/limited/unknown converted to do not resuscitate and do not intubate status. In total, 79% of patients seen by palliative care had a confirmed code status of Do Not Resuscitate and Do Not Intubate status after consult.
Discussion
In our first year, our palliative care program exceeded the expected number of inpatient consults, corresponding with a penetration of 3.7%. With the increase of funded FTEs, preliminary data shows that the department’s penetration continues to rise remaining consistent with the data and expectations [18]. During the second year, it is anticipated that over 600 inpatient palliative care consultations will be performed with an estimated penetration of 4%. This increasing penetration reflects the rising utilization of palliative care within our hospital. Since inception of the program, the service has expanded into an outpatient clinic 2 days per week. The palliative care clinic is staffed by a registered nurse (funded 0.6 FTE) and covered by the same physicians and physician assistant providing the inpatient services. The department acquired an unfunded but designated chaplaincy volunteer to assist with patients’ spiritual needs. We believe that the success of our program during the first year was related to multiple factors: a focus of integration and education by the palliative care department, health system administration buy-in, and identification of surrounding community needs.
In addition to patient care, our palliative care department also prioritizes “tangible” impacts to better establish our contributions to the health system. We have done this through participation on hospital committees, hospital policy revision teams, and by developing innovative solutions such as a terminal extubation protocol and order set for our ICU. The health system and its administration have recognized the importance of educating nursing and physician staff on palliative care services, and have supported these continued efforts alongside our clinical obligations.
Concurrent with administration buy-in, financial supports for our palliative care services were initially supplemented by the health system. Our department understands the importance of recognizing limitations of resources in communities and their hospitals. In efforts to minimize the department’s impact on our own health system’s financial resources, we have strived to offset our costs. We helped the hospital system meet pay-for-performance palliative care metrics set by the large local insurers resulting in financial hospital reimbursement valued at $600,000 in 2016.
The question of how the program may translate into other communities raises a major limitation: the homogeneity our population. The community surrounding the hospital is primarily Caucasian, with minimal representation of minority populations. While the patient population seen by our palliative care team is reflective of our surrounding county, it does not represent Medicare FFS beneficiaries on a national level or many other types of community hospitals across the country. Variations of ethnicity, age, diagnoses, and faith are fundamental, which highlights the importance of understanding the community in which a program is developed.
The rising trajectory of our palliative care service utilization has prompted a discussion of future endeavors for our program. Expectations for a continued shortage of hospice and palliative care physicians [19] and concerns for practitioner burnout [20] underlie our thoughtful approach to expansion of inpatient and outpatient services. At this time, potential projects include a consultation trigger system and incorporation of palliative care providers in ICU rounding, as well as possible expansion of outpatient services through implantation of an advanced practitioner into surrounding nursing homes and primary care offices.
We have found a growing utilization of our program at Butler Health System. Our first year experience has highlighted the importance of identifying community and hospital administrative champions as a foundation. Additionally, understanding the specific characteristics of one’s surrounding community may allow for improved integration and acceptance of palliative care in a community setting. Our program continues to work with the health system, community, and philanthropic organizations to expand the ever-growing need for palliative care services.
1. Dumanovsky T, Augustin R, Rogers M, et al. The growth of palliative care in U.S. hospitals: a status report. J Palliat Med 2016;19:8–15.
2. Lavergne MR, Lethbridge L, Johnston G, et al. Examining palliative care program use and place of death in rural and urban contexts: a Canadian population-based study using linked data. Rural Remote Health 2015;15:3134.
3. Seow H, Brazil K, Sussman J, et al. Impact of community based, specialist palliative care teams on hospitalisations and emergency department visits late in life and hospital deaths: a pooled analysis. BMJ 2014;348:g3496.
4. Fink RM, Oman KS, Youngwerth J, et al. A palliative care needs assessment of rural hospitals. J Palliat Med 2013;16:638–44.
5. Dumont S, Jacobs P, Turcotte V, et al. Palliative care costs in Canada: A descriptive comparison of studies of urban and rural patients near end of life. J Palliat Med 2015;29:908–17.
6. Kaasalainen S, Brazil K, Williams A, et al. Nurses' experiences providing palliative care to individuals living in rural communities: aspects of the physical residential setting. Rural Remote Health 2014;14:2728.
7. Ahmed N, Bestall JC, Ahmedzai SH, et al. Systematic review of the problems and issues of accessing specialist palliative care by patients, carers and health and social care professionals. J Palliat Med 2004;18:525–42.
8. Ray RA, Fried O, Lindsay D. Palliative care professional education via video conference builds confidence to deliver palliative care in rural and remote locations. BMC Health Serv Res 2014;14:272.
9. Bakitas MA, Elk R, Astin M, et al. Systematic review of palliative care in the rural setting. Cancer Control 2015;22:450–64.
10. Akiyama M, Hirai K, Takebayashi T, et al. The effects of community-wide dissemination of information on perceptions of palliative care, knowledge about opioids, and sense of security among cancer patients, their families, and the general public. Support Care Cancer 2016;24: 347–56.
11. Maida V. Factors that promote success in home palliative care: a study of a large suburban palliative care practice. J Palliat Care 2002;18:282–6.
12. Ceronsky L, Shearer J, Weng K, et al. Minnesota Rural Palliative Care Initiative: building palliative care capacity in rural Minnesota. J Palliat Med 2013;16:310–3.
13. Aggarwal SK, Ghosh A, Cheng MJ, et al. Initiating pain and palliative care outpatient services for the suburban underserved in Montgomery County, Maryland: Lessons learned at the NIH Clinical Center and MobileMed. Palliat Support Care 2015;16:1–6.
14. Rainsford S, MacLeod RD, Glasgow NJ. Place of death in rural palliative care: A systematic review. J Palliat Med 2016;30:745–63.
15. Horey D, Street AF, O'Connor M, et al. Training and supportive programs for palliative care volunteers in community settings. Cochrane Database Syst Rev 2015 Jul 20;(7):CD009500.
16. The United States Census Bureau: QuickFacts: Butler County, Pennsylvania. Accessed 10 Mar 2017 at www.census.gov/quickfacts/table/PST045216/42019,00.
17. Spetz J, Dudley N, Trupin L, et al. Few hospital palliative care programs meet national staffing recommendations. Health Aff 2016;35:1690–7.
18. Dumanovsky T, Rogers M, Spragens LH, et al. Impact of Staffing on Access to Palliative Care in U.S. Hospitals. J Palliat Med. 2015 Dec; 18(12). Pages 998-999.
19. Lupu D, American Academy of Hospice and Palliative Medicine Workforce Task Force. Estimate of current hospice and palliative medicine physician workforce shortage. J Pain Symptom Manage 2010;40:899–911.
20. Kamal AH, Bull JK, Wolf SP, et al. Prevalence and predictors of burnout among hospice and palliative care clinicians in the U.S. J Pain Symptom Manage 2016;51:690–6.
1. Dumanovsky T, Augustin R, Rogers M, et al. The growth of palliative care in U.S. hospitals: a status report. J Palliat Med 2016;19:8–15.
2. Lavergne MR, Lethbridge L, Johnston G, et al. Examining palliative care program use and place of death in rural and urban contexts: a Canadian population-based study using linked data. Rural Remote Health 2015;15:3134.
3. Seow H, Brazil K, Sussman J, et al. Impact of community based, specialist palliative care teams on hospitalisations and emergency department visits late in life and hospital deaths: a pooled analysis. BMJ 2014;348:g3496.
4. Fink RM, Oman KS, Youngwerth J, et al. A palliative care needs assessment of rural hospitals. J Palliat Med 2013;16:638–44.
5. Dumont S, Jacobs P, Turcotte V, et al. Palliative care costs in Canada: A descriptive comparison of studies of urban and rural patients near end of life. J Palliat Med 2015;29:908–17.
6. Kaasalainen S, Brazil K, Williams A, et al. Nurses' experiences providing palliative care to individuals living in rural communities: aspects of the physical residential setting. Rural Remote Health 2014;14:2728.
7. Ahmed N, Bestall JC, Ahmedzai SH, et al. Systematic review of the problems and issues of accessing specialist palliative care by patients, carers and health and social care professionals. J Palliat Med 2004;18:525–42.
8. Ray RA, Fried O, Lindsay D. Palliative care professional education via video conference builds confidence to deliver palliative care in rural and remote locations. BMC Health Serv Res 2014;14:272.
9. Bakitas MA, Elk R, Astin M, et al. Systematic review of palliative care in the rural setting. Cancer Control 2015;22:450–64.
10. Akiyama M, Hirai K, Takebayashi T, et al. The effects of community-wide dissemination of information on perceptions of palliative care, knowledge about opioids, and sense of security among cancer patients, their families, and the general public. Support Care Cancer 2016;24: 347–56.
11. Maida V. Factors that promote success in home palliative care: a study of a large suburban palliative care practice. J Palliat Care 2002;18:282–6.
12. Ceronsky L, Shearer J, Weng K, et al. Minnesota Rural Palliative Care Initiative: building palliative care capacity in rural Minnesota. J Palliat Med 2013;16:310–3.
13. Aggarwal SK, Ghosh A, Cheng MJ, et al. Initiating pain and palliative care outpatient services for the suburban underserved in Montgomery County, Maryland: Lessons learned at the NIH Clinical Center and MobileMed. Palliat Support Care 2015;16:1–6.
14. Rainsford S, MacLeod RD, Glasgow NJ. Place of death in rural palliative care: A systematic review. J Palliat Med 2016;30:745–63.
15. Horey D, Street AF, O'Connor M, et al. Training and supportive programs for palliative care volunteers in community settings. Cochrane Database Syst Rev 2015 Jul 20;(7):CD009500.
16. The United States Census Bureau: QuickFacts: Butler County, Pennsylvania. Accessed 10 Mar 2017 at www.census.gov/quickfacts/table/PST045216/42019,00.
17. Spetz J, Dudley N, Trupin L, et al. Few hospital palliative care programs meet national staffing recommendations. Health Aff 2016;35:1690–7.
18. Dumanovsky T, Rogers M, Spragens LH, et al. Impact of Staffing on Access to Palliative Care in U.S. Hospitals. J Palliat Med. 2015 Dec; 18(12). Pages 998-999.
19. Lupu D, American Academy of Hospice and Palliative Medicine Workforce Task Force. Estimate of current hospice and palliative medicine physician workforce shortage. J Pain Symptom Manage 2010;40:899–911.
20. Kamal AH, Bull JK, Wolf SP, et al. Prevalence and predictors of burnout among hospice and palliative care clinicians in the U.S. J Pain Symptom Manage 2016;51:690–6.
Does newly discovered vasoactive peptide ELABELA reveal essential mechanisms for preeclampsia development?
EXPERT COMMENTARY
Preeclampsia is a disorder of impaired placentation. ELABELA (ELA) encodes an endogenous ligand for the apelin receptor and is detected in preimplantation human blastocysts and in 2 organs in adults: the placenta and kidney. Recently, an international team of researchers described their study findings on ELA and its role in placental vascular development and preeclampsia in mice.
Details of the study
To delineate the contribution of ELA to mammalian development, Ho and colleagues generated Ela knockout mice. The investigators showed that during development, the ELA protein is first detected in the early placenta and becomes abundant later in placenta formation. They also demonstrated that ELA is a pregnancy hormone that circulates in the blood of pregnant, but not nonpregnant, mice.
Placental structure. The Ela knockout mice had placentas that demonstrated thin labyrinths, with poor vascularization, increased apoptosis, and reduced proliferation. Further, RNA analysis of ELA-lacking placentas revealed a gene expression profile indicative of hypoxia, including the upregulation of certain genes involved in blood vessel building. Placental vessels showed overall stunted architecture characterized by little or no extension and branching of angiogenic sprouts and impaired formation of the adequate labyrinth network required for proper perfusion in the placenta.
Given these gene expression findings and the fact that placental and vascular abnormalities have long been suspected to underlie preeclampsia, the investigators sought to determine if ELA-lacking mice exhibit preeclampsia. Evidence indicated they do.
Indicators of preeclampsia. Pregnant ELA-lacking mice had significantly higher levels of proteinuria and significantly higher blood pressure than either pregnant wild-type mice or nonpregnant ELA-lacking mice. Further, at the end of pregnancy, histology and transmission electron microscopy of kidney glomerular sections from ELA-lacking pregnant mice revealed signs of endotheliosis, a unique renal pathology that is also observed in women with preeclampsia. Pups of ELA-lacking mothers tended to weigh less than those of wild-type mothers, a situation that may be similar to the fetal intrauterine growth restriction commonly seen in women with preeclampsia.
Angiogenic factors. The authors then looked at levels of angiogenic proteins implicated in the pathogenesis of preeclampsia to determine if ELA is upstream. They found that ELA-lacking mice placentas had increased levels of sFlt1, Vegfa, and Plgf mRNA; these transcriptional changes, however, did not translate into significantly elevated plasma levels of the respective proteins. Thus, these findings indicate that ELA acts independently of, and possibly earlier than, angiogenic factors in the pathogenesis of preeclampsia.
Experimental treatment. The authors further showed that infusing recombinant ELA protein could alleviate symptoms of preeclampsia in mice. Injection of ELA protein in ELA-lacking mice led to reduction of blood pressure, reversal of glomerular endotheliosis, and rescue of fetal growth restriction.
Study strengths and weaknesses
This animal study contributes compelling molecular evidence of ELA’s role in mammalian placental development and angiogenesis, revealing that ELA deficiency leads to preeclampsia and placental abnormalities in pregnant mice. How ELA acts in humans and human pregnancy, however, has yet to be explored.
-- Sarosh Rana, MD, MPH
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
EXPERT COMMENTARY
Preeclampsia is a disorder of impaired placentation. ELABELA (ELA) encodes an endogenous ligand for the apelin receptor and is detected in preimplantation human blastocysts and in 2 organs in adults: the placenta and kidney. Recently, an international team of researchers described their study findings on ELA and its role in placental vascular development and preeclampsia in mice.
Details of the study
To delineate the contribution of ELA to mammalian development, Ho and colleagues generated Ela knockout mice. The investigators showed that during development, the ELA protein is first detected in the early placenta and becomes abundant later in placenta formation. They also demonstrated that ELA is a pregnancy hormone that circulates in the blood of pregnant, but not nonpregnant, mice.
Placental structure. The Ela knockout mice had placentas that demonstrated thin labyrinths, with poor vascularization, increased apoptosis, and reduced proliferation. Further, RNA analysis of ELA-lacking placentas revealed a gene expression profile indicative of hypoxia, including the upregulation of certain genes involved in blood vessel building. Placental vessels showed overall stunted architecture characterized by little or no extension and branching of angiogenic sprouts and impaired formation of the adequate labyrinth network required for proper perfusion in the placenta.
Given these gene expression findings and the fact that placental and vascular abnormalities have long been suspected to underlie preeclampsia, the investigators sought to determine if ELA-lacking mice exhibit preeclampsia. Evidence indicated they do.
Indicators of preeclampsia. Pregnant ELA-lacking mice had significantly higher levels of proteinuria and significantly higher blood pressure than either pregnant wild-type mice or nonpregnant ELA-lacking mice. Further, at the end of pregnancy, histology and transmission electron microscopy of kidney glomerular sections from ELA-lacking pregnant mice revealed signs of endotheliosis, a unique renal pathology that is also observed in women with preeclampsia. Pups of ELA-lacking mothers tended to weigh less than those of wild-type mothers, a situation that may be similar to the fetal intrauterine growth restriction commonly seen in women with preeclampsia.
Angiogenic factors. The authors then looked at levels of angiogenic proteins implicated in the pathogenesis of preeclampsia to determine if ELA is upstream. They found that ELA-lacking mice placentas had increased levels of sFlt1, Vegfa, and Plgf mRNA; these transcriptional changes, however, did not translate into significantly elevated plasma levels of the respective proteins. Thus, these findings indicate that ELA acts independently of, and possibly earlier than, angiogenic factors in the pathogenesis of preeclampsia.
Experimental treatment. The authors further showed that infusing recombinant ELA protein could alleviate symptoms of preeclampsia in mice. Injection of ELA protein in ELA-lacking mice led to reduction of blood pressure, reversal of glomerular endotheliosis, and rescue of fetal growth restriction.
Study strengths and weaknesses
This animal study contributes compelling molecular evidence of ELA’s role in mammalian placental development and angiogenesis, revealing that ELA deficiency leads to preeclampsia and placental abnormalities in pregnant mice. How ELA acts in humans and human pregnancy, however, has yet to be explored.
-- Sarosh Rana, MD, MPH
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
EXPERT COMMENTARY
Preeclampsia is a disorder of impaired placentation. ELABELA (ELA) encodes an endogenous ligand for the apelin receptor and is detected in preimplantation human blastocysts and in 2 organs in adults: the placenta and kidney. Recently, an international team of researchers described their study findings on ELA and its role in placental vascular development and preeclampsia in mice.
Details of the study
To delineate the contribution of ELA to mammalian development, Ho and colleagues generated Ela knockout mice. The investigators showed that during development, the ELA protein is first detected in the early placenta and becomes abundant later in placenta formation. They also demonstrated that ELA is a pregnancy hormone that circulates in the blood of pregnant, but not nonpregnant, mice.
Placental structure. The Ela knockout mice had placentas that demonstrated thin labyrinths, with poor vascularization, increased apoptosis, and reduced proliferation. Further, RNA analysis of ELA-lacking placentas revealed a gene expression profile indicative of hypoxia, including the upregulation of certain genes involved in blood vessel building. Placental vessels showed overall stunted architecture characterized by little or no extension and branching of angiogenic sprouts and impaired formation of the adequate labyrinth network required for proper perfusion in the placenta.
Given these gene expression findings and the fact that placental and vascular abnormalities have long been suspected to underlie preeclampsia, the investigators sought to determine if ELA-lacking mice exhibit preeclampsia. Evidence indicated they do.
Indicators of preeclampsia. Pregnant ELA-lacking mice had significantly higher levels of proteinuria and significantly higher blood pressure than either pregnant wild-type mice or nonpregnant ELA-lacking mice. Further, at the end of pregnancy, histology and transmission electron microscopy of kidney glomerular sections from ELA-lacking pregnant mice revealed signs of endotheliosis, a unique renal pathology that is also observed in women with preeclampsia. Pups of ELA-lacking mothers tended to weigh less than those of wild-type mothers, a situation that may be similar to the fetal intrauterine growth restriction commonly seen in women with preeclampsia.
Angiogenic factors. The authors then looked at levels of angiogenic proteins implicated in the pathogenesis of preeclampsia to determine if ELA is upstream. They found that ELA-lacking mice placentas had increased levels of sFlt1, Vegfa, and Plgf mRNA; these transcriptional changes, however, did not translate into significantly elevated plasma levels of the respective proteins. Thus, these findings indicate that ELA acts independently of, and possibly earlier than, angiogenic factors in the pathogenesis of preeclampsia.
Experimental treatment. The authors further showed that infusing recombinant ELA protein could alleviate symptoms of preeclampsia in mice. Injection of ELA protein in ELA-lacking mice led to reduction of blood pressure, reversal of glomerular endotheliosis, and rescue of fetal growth restriction.
Study strengths and weaknesses
This animal study contributes compelling molecular evidence of ELA’s role in mammalian placental development and angiogenesis, revealing that ELA deficiency leads to preeclampsia and placental abnormalities in pregnant mice. How ELA acts in humans and human pregnancy, however, has yet to be explored.
-- Sarosh Rana, MD, MPH
Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
Debunking Actinic Keratosis Myths: Are Patients With Darker Skin At Risk for Actinic Keratoses?
Myth: Actinic keratoses are only seen in patients with lighter skin
Actinic keratoses (AKs) are precancerous lesions that may turn into squamous cell carcinoma if left untreated. UV rays cause AKs, either from outdoor sun exposure or tanning beds. According to the American Academy of Dermatology, AKs are more likely to develop in patients 40 years or older with fair skin; hair color that is naturally blonde or red; eye color that is naturally blue, green, or hazel; skin that freckles or burns when in the sun; a weakened immune system; and occupations involving substances that contain polycyclic aromatic hydrocarbons such as coal or tar.
A 2007 study compared the most common diagnoses among patients of different racial and ethnic groups in New York City. Alexis et al found that AK was in the top 10 diagnoses in white patients but not for black patients. They postulated that photoprotective factors in darkly pigmented skin such as larger and more numerous melanosomes that contain more melanin and are more dispersed throughout the epidermis result in a lower incidence of skin cancers in the skin of color (SOC) population.
RELATED ARTICLE: Common Dermatologic Disorders in Skin of Color: A Comparative Practice Survey
However, a recent skin cancer awareness study in Cutis reported that even though SOC populations have lower incidences of skin cancer such as melanoma, basal cell carcinoma, and squamous cell carcinoma, they exhibit higher death rates. Furthermore, black individuals are more likely to present with advanced-stage melanoma and acral lentiginous melanomas compared to white individuals. Kailas et al stated, “Overall, SOC patients have the poorest skin cancer prognosis, and the data suggest that the reason for this paradox is delayed diagnosis.” They evaluated several knowledge-based interventions for increasing skin cancer awareness, knowledge, and protective behaviors in SOC populations, including the use of visuals such as photographs to allow SOC patients to visualize different skin tones, educational interventions in another language, and pamphlets.
Dermatologists should be aware that education of SOC patients is important to eradicate the common misconception that these patients do not have to worry about AKs and other skin cancers. Remind these patients that they need to protect their skin from the sun, just as patients with fair skin do. Further research in the dermatology community should focus on educational interventions that will help increase knowledge regarding skin cancer in SOC populations.
Expert Commentary
Although more common in patients with lighter skin, actinic keratosis and skin cancer can be seen in patients of all skin types. Many patients are unaware of this risk and do not use sunscreen and other sun-protective measures. We, as a specialty, have to educate our patients and the public of the risk for actinic keratosis and skin cancer in all skin types.
—Gary Goldenberg, MD (New York, New York)
Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
American Academy of Dermatology. Actinic keratosis. https://www.aad.org/public/diseases/scaly-skin/actinic-keratosis. Accessed October 17, 2017.
Kailas A, Botwin AL, Pritchett EN, et al. Assessing the effectiveness of knowledge-based interventions in increasing skin cancer awareness, knowledge, and protective behaviors in skin of color populations. Cutis. 2017;100:235-240.
Myth: Actinic keratoses are only seen in patients with lighter skin
Actinic keratoses (AKs) are precancerous lesions that may turn into squamous cell carcinoma if left untreated. UV rays cause AKs, either from outdoor sun exposure or tanning beds. According to the American Academy of Dermatology, AKs are more likely to develop in patients 40 years or older with fair skin; hair color that is naturally blonde or red; eye color that is naturally blue, green, or hazel; skin that freckles or burns when in the sun; a weakened immune system; and occupations involving substances that contain polycyclic aromatic hydrocarbons such as coal or tar.
A 2007 study compared the most common diagnoses among patients of different racial and ethnic groups in New York City. Alexis et al found that AK was in the top 10 diagnoses in white patients but not for black patients. They postulated that photoprotective factors in darkly pigmented skin such as larger and more numerous melanosomes that contain more melanin and are more dispersed throughout the epidermis result in a lower incidence of skin cancers in the skin of color (SOC) population.
RELATED ARTICLE: Common Dermatologic Disorders in Skin of Color: A Comparative Practice Survey
However, a recent skin cancer awareness study in Cutis reported that even though SOC populations have lower incidences of skin cancer such as melanoma, basal cell carcinoma, and squamous cell carcinoma, they exhibit higher death rates. Furthermore, black individuals are more likely to present with advanced-stage melanoma and acral lentiginous melanomas compared to white individuals. Kailas et al stated, “Overall, SOC patients have the poorest skin cancer prognosis, and the data suggest that the reason for this paradox is delayed diagnosis.” They evaluated several knowledge-based interventions for increasing skin cancer awareness, knowledge, and protective behaviors in SOC populations, including the use of visuals such as photographs to allow SOC patients to visualize different skin tones, educational interventions in another language, and pamphlets.
Dermatologists should be aware that education of SOC patients is important to eradicate the common misconception that these patients do not have to worry about AKs and other skin cancers. Remind these patients that they need to protect their skin from the sun, just as patients with fair skin do. Further research in the dermatology community should focus on educational interventions that will help increase knowledge regarding skin cancer in SOC populations.
Expert Commentary
Although more common in patients with lighter skin, actinic keratosis and skin cancer can be seen in patients of all skin types. Many patients are unaware of this risk and do not use sunscreen and other sun-protective measures. We, as a specialty, have to educate our patients and the public of the risk for actinic keratosis and skin cancer in all skin types.
—Gary Goldenberg, MD (New York, New York)
Myth: Actinic keratoses are only seen in patients with lighter skin
Actinic keratoses (AKs) are precancerous lesions that may turn into squamous cell carcinoma if left untreated. UV rays cause AKs, either from outdoor sun exposure or tanning beds. According to the American Academy of Dermatology, AKs are more likely to develop in patients 40 years or older with fair skin; hair color that is naturally blonde or red; eye color that is naturally blue, green, or hazel; skin that freckles or burns when in the sun; a weakened immune system; and occupations involving substances that contain polycyclic aromatic hydrocarbons such as coal or tar.
A 2007 study compared the most common diagnoses among patients of different racial and ethnic groups in New York City. Alexis et al found that AK was in the top 10 diagnoses in white patients but not for black patients. They postulated that photoprotective factors in darkly pigmented skin such as larger and more numerous melanosomes that contain more melanin and are more dispersed throughout the epidermis result in a lower incidence of skin cancers in the skin of color (SOC) population.
RELATED ARTICLE: Common Dermatologic Disorders in Skin of Color: A Comparative Practice Survey
However, a recent skin cancer awareness study in Cutis reported that even though SOC populations have lower incidences of skin cancer such as melanoma, basal cell carcinoma, and squamous cell carcinoma, they exhibit higher death rates. Furthermore, black individuals are more likely to present with advanced-stage melanoma and acral lentiginous melanomas compared to white individuals. Kailas et al stated, “Overall, SOC patients have the poorest skin cancer prognosis, and the data suggest that the reason for this paradox is delayed diagnosis.” They evaluated several knowledge-based interventions for increasing skin cancer awareness, knowledge, and protective behaviors in SOC populations, including the use of visuals such as photographs to allow SOC patients to visualize different skin tones, educational interventions in another language, and pamphlets.
Dermatologists should be aware that education of SOC patients is important to eradicate the common misconception that these patients do not have to worry about AKs and other skin cancers. Remind these patients that they need to protect their skin from the sun, just as patients with fair skin do. Further research in the dermatology community should focus on educational interventions that will help increase knowledge regarding skin cancer in SOC populations.
Expert Commentary
Although more common in patients with lighter skin, actinic keratosis and skin cancer can be seen in patients of all skin types. Many patients are unaware of this risk and do not use sunscreen and other sun-protective measures. We, as a specialty, have to educate our patients and the public of the risk for actinic keratosis and skin cancer in all skin types.
—Gary Goldenberg, MD (New York, New York)
Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
American Academy of Dermatology. Actinic keratosis. https://www.aad.org/public/diseases/scaly-skin/actinic-keratosis. Accessed October 17, 2017.
Kailas A, Botwin AL, Pritchett EN, et al. Assessing the effectiveness of knowledge-based interventions in increasing skin cancer awareness, knowledge, and protective behaviors in skin of color populations. Cutis. 2017;100:235-240.
Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
American Academy of Dermatology. Actinic keratosis. https://www.aad.org/public/diseases/scaly-skin/actinic-keratosis. Accessed October 17, 2017.
Kailas A, Botwin AL, Pritchett EN, et al. Assessing the effectiveness of knowledge-based interventions in increasing skin cancer awareness, knowledge, and protective behaviors in skin of color populations. Cutis. 2017;100:235-240.
Maternally derived pneumococcal, meningococcal antibodies may affect vaccine effectiveness
, according to a new study.
That information may be useful in deciding the impact of vaccination programs that use a combination of maternal and infant vaccines and consider schedules with a delayed start, said Merryn Voysey of the University of Oxford (England) and her associates.
In this study, 5,097 children in 16 cohorts from 13 countries had pneumococcal antibody concentrations assessed from blood samples taken before their first dose of vaccine, and 2,925 infants from 5 cohorts in 4 countries had meningococcal antibody concentrations available.
At the time of their first vaccination, the children were ages 5-23 weeks and were from countries in Europe, Africa, Latin America, and South and East Asia. These populations have no routine programs of immunization in pregnancy, the researchers said. So, the maternal antibodies are passively acquired, and the decay rates may differ from those induced by maternal vaccinations.
The seroprevalence of maternal antibodies in infants was 92% for pneumococcal serotype 14 and 80% for serotype 19F; it was 30% for serotype 4 and 34% for serotype 1. Thirteen percent of infants had detectable levels of group C meningococcal antibodies prior to vaccination, and 43% had group A antibodies.
For the pneumococcal antibodies, “there was statistically significant variation in half-life estimates between country cohorts and between serotypes (both P less than .0001),” the researchers said. The half-life estimate was lowest – at 39 days – for serotype 6B, and highest – at 48 days – for serotype 5. The overall estimate across serotypes was 43 days.
“The age of the child was not significantly associated with decay rates (P = .103), confirming the assumption of exponential decay,” they said.
For the meningococcal antibodies, the half-lives were 43 days for group A and 40 days for group C.
“Substantial proportions of infants have antibodies to many vaccine serotypes of pneumococcus at the age when a vaccine program might normally commence,” the investigators noted. “Conversely, antibodies against capsular groups A and C meningococcal polysaccharides were less common, particularly for group C, which was only present in 13% of infants in the four countries contained in this analysis.
“Higher levels of group A meningococcal antibodies than group C have also been seen in unvaccinated adults of childbearing age in the Netherlands, and in mothers in the United Kingdom,” the researchers added. “Passively acquired maternal antibody has been shown to adversely affect the magnitude of the immune response to vaccination with pneumococcal conjugate vaccine, and increase the occurrence of otitis media in infants under 6 months of age.”
The proportion of infants who had maternal antipneumococcal antibodies differed between serotypes, the authors noted. Almost all infants had serotype 14 pneumococcal antibodies, and very high proportions of infants had serotype 19F antibodies.
“We have previously shown that the antibody response to vaccination with pneumococcal conjugate vaccine is adversely affected by the presence of maternal antibody,” the investigators said. “This inhibitory effect is greatest for serotype 14, with children seropositive from maternal antibodies having a response to vaccination that is only three-quarters the magnitude of those with no maternal antibody.”
Read more in Vaccine (2017 Oct 13;35[43]:5850-7).
, according to a new study.
That information may be useful in deciding the impact of vaccination programs that use a combination of maternal and infant vaccines and consider schedules with a delayed start, said Merryn Voysey of the University of Oxford (England) and her associates.
In this study, 5,097 children in 16 cohorts from 13 countries had pneumococcal antibody concentrations assessed from blood samples taken before their first dose of vaccine, and 2,925 infants from 5 cohorts in 4 countries had meningococcal antibody concentrations available.
At the time of their first vaccination, the children were ages 5-23 weeks and were from countries in Europe, Africa, Latin America, and South and East Asia. These populations have no routine programs of immunization in pregnancy, the researchers said. So, the maternal antibodies are passively acquired, and the decay rates may differ from those induced by maternal vaccinations.
The seroprevalence of maternal antibodies in infants was 92% for pneumococcal serotype 14 and 80% for serotype 19F; it was 30% for serotype 4 and 34% for serotype 1. Thirteen percent of infants had detectable levels of group C meningococcal antibodies prior to vaccination, and 43% had group A antibodies.
For the pneumococcal antibodies, “there was statistically significant variation in half-life estimates between country cohorts and between serotypes (both P less than .0001),” the researchers said. The half-life estimate was lowest – at 39 days – for serotype 6B, and highest – at 48 days – for serotype 5. The overall estimate across serotypes was 43 days.
“The age of the child was not significantly associated with decay rates (P = .103), confirming the assumption of exponential decay,” they said.
For the meningococcal antibodies, the half-lives were 43 days for group A and 40 days for group C.
“Substantial proportions of infants have antibodies to many vaccine serotypes of pneumococcus at the age when a vaccine program might normally commence,” the investigators noted. “Conversely, antibodies against capsular groups A and C meningococcal polysaccharides were less common, particularly for group C, which was only present in 13% of infants in the four countries contained in this analysis.
“Higher levels of group A meningococcal antibodies than group C have also been seen in unvaccinated adults of childbearing age in the Netherlands, and in mothers in the United Kingdom,” the researchers added. “Passively acquired maternal antibody has been shown to adversely affect the magnitude of the immune response to vaccination with pneumococcal conjugate vaccine, and increase the occurrence of otitis media in infants under 6 months of age.”
The proportion of infants who had maternal antipneumococcal antibodies differed between serotypes, the authors noted. Almost all infants had serotype 14 pneumococcal antibodies, and very high proportions of infants had serotype 19F antibodies.
“We have previously shown that the antibody response to vaccination with pneumococcal conjugate vaccine is adversely affected by the presence of maternal antibody,” the investigators said. “This inhibitory effect is greatest for serotype 14, with children seropositive from maternal antibodies having a response to vaccination that is only three-quarters the magnitude of those with no maternal antibody.”
Read more in Vaccine (2017 Oct 13;35[43]:5850-7).
, according to a new study.
That information may be useful in deciding the impact of vaccination programs that use a combination of maternal and infant vaccines and consider schedules with a delayed start, said Merryn Voysey of the University of Oxford (England) and her associates.
In this study, 5,097 children in 16 cohorts from 13 countries had pneumococcal antibody concentrations assessed from blood samples taken before their first dose of vaccine, and 2,925 infants from 5 cohorts in 4 countries had meningococcal antibody concentrations available.
At the time of their first vaccination, the children were ages 5-23 weeks and were from countries in Europe, Africa, Latin America, and South and East Asia. These populations have no routine programs of immunization in pregnancy, the researchers said. So, the maternal antibodies are passively acquired, and the decay rates may differ from those induced by maternal vaccinations.
The seroprevalence of maternal antibodies in infants was 92% for pneumococcal serotype 14 and 80% for serotype 19F; it was 30% for serotype 4 and 34% for serotype 1. Thirteen percent of infants had detectable levels of group C meningococcal antibodies prior to vaccination, and 43% had group A antibodies.
For the pneumococcal antibodies, “there was statistically significant variation in half-life estimates between country cohorts and between serotypes (both P less than .0001),” the researchers said. The half-life estimate was lowest – at 39 days – for serotype 6B, and highest – at 48 days – for serotype 5. The overall estimate across serotypes was 43 days.
“The age of the child was not significantly associated with decay rates (P = .103), confirming the assumption of exponential decay,” they said.
For the meningococcal antibodies, the half-lives were 43 days for group A and 40 days for group C.
“Substantial proportions of infants have antibodies to many vaccine serotypes of pneumococcus at the age when a vaccine program might normally commence,” the investigators noted. “Conversely, antibodies against capsular groups A and C meningococcal polysaccharides were less common, particularly for group C, which was only present in 13% of infants in the four countries contained in this analysis.
“Higher levels of group A meningococcal antibodies than group C have also been seen in unvaccinated adults of childbearing age in the Netherlands, and in mothers in the United Kingdom,” the researchers added. “Passively acquired maternal antibody has been shown to adversely affect the magnitude of the immune response to vaccination with pneumococcal conjugate vaccine, and increase the occurrence of otitis media in infants under 6 months of age.”
The proportion of infants who had maternal antipneumococcal antibodies differed between serotypes, the authors noted. Almost all infants had serotype 14 pneumococcal antibodies, and very high proportions of infants had serotype 19F antibodies.
“We have previously shown that the antibody response to vaccination with pneumococcal conjugate vaccine is adversely affected by the presence of maternal antibody,” the investigators said. “This inhibitory effect is greatest for serotype 14, with children seropositive from maternal antibodies having a response to vaccination that is only three-quarters the magnitude of those with no maternal antibody.”
Read more in Vaccine (2017 Oct 13;35[43]:5850-7).
FROM VACCINE
Vitality predicts T2DM major cardiovascular event risk
LISBON – according to the results of a primary care study.
Seldom feeling “full of pep” or not having “a lot of energy” was associated with an increased risk of a major cardiovascular event (MACE) in middle-aged (55-66 years) adults with T2DM, Marta Vergara, MD, reported at the annual meeting of the European Association for the Study of Diabetes.
“It’s well known that patients with type 2 diabetes have a high risk for developing cardiovascular disease, and it is the main cause of death,” said Dr. Vergara of Linköping (Sweden) University.
While several risk factors for cardiovascular disease are known and widely monitored for in clinical practice worldwide, including psychological aspects such as mental stress, ways to identify patients earlier are needed.
“We need more clinically useful and easy-to-manage measurement instruments,” Dr. Vergara said.
Using data from the ongoing Cardiovascular Risk Factors in Patients With Diabetes – a Prospective Study in Primary Care (CARDIPP), Dr. Vergara and colleagues identified two questions used in the 36-Item Short Form (SF-36) that might fit the bill:
- ”How much time during the past 4 weeks did you feel full of pep?”
- “How much time during the past 4 weeks did you have a lot of energy?”
Patients had answered these questions using a 6-point scale ranging from 1, all of the time, to 6 none of the time.
In addition to completing the full SF-36 at recruitment, all patients enrolled in CARDIPP underwent thorough assessment of baseline cardiovascular risk. This included asking about their duration of diabetes and smoking history, taking glycemic and anthropometric readings, and recording their systolic blood pressure. Pulse wave velocity was also used to assess patients’ carotid-femoral artery stiffness.
Over a follow-up period of 7 years, 59 (7.8%) of 761 men and women aged 55-66 years developed ischemic heart disease or had a stroke and needed hospital treatment or died.
“Our data support the use of questions about sense of vitality in order to add prognostic information about subsequent risk of MACE independently of traditional risk markers, such as diabetes duration, age, HbA1c, gender, smoking, systolic blood pressure, and carotid-femoral pulse wave velocity and sagittal abdominal diameter,” Dr. Vergara said.
“We suggest that not feeling full of pep and not having a lot of energy could be used as risk markers for MACE in patients with type 2 diabetes.” These are two easy questions that could be asked as part of the risk assessment process in primary care, she suggested. “Other questions from the SF-36, such as feeling tired or worn out, were not independent markers of MACE,” Dr. Vergara said.
She said she had no relevant financial disclosures.
LISBON – according to the results of a primary care study.
Seldom feeling “full of pep” or not having “a lot of energy” was associated with an increased risk of a major cardiovascular event (MACE) in middle-aged (55-66 years) adults with T2DM, Marta Vergara, MD, reported at the annual meeting of the European Association for the Study of Diabetes.
“It’s well known that patients with type 2 diabetes have a high risk for developing cardiovascular disease, and it is the main cause of death,” said Dr. Vergara of Linköping (Sweden) University.
While several risk factors for cardiovascular disease are known and widely monitored for in clinical practice worldwide, including psychological aspects such as mental stress, ways to identify patients earlier are needed.
“We need more clinically useful and easy-to-manage measurement instruments,” Dr. Vergara said.
Using data from the ongoing Cardiovascular Risk Factors in Patients With Diabetes – a Prospective Study in Primary Care (CARDIPP), Dr. Vergara and colleagues identified two questions used in the 36-Item Short Form (SF-36) that might fit the bill:
- ”How much time during the past 4 weeks did you feel full of pep?”
- “How much time during the past 4 weeks did you have a lot of energy?”
Patients had answered these questions using a 6-point scale ranging from 1, all of the time, to 6 none of the time.
In addition to completing the full SF-36 at recruitment, all patients enrolled in CARDIPP underwent thorough assessment of baseline cardiovascular risk. This included asking about their duration of diabetes and smoking history, taking glycemic and anthropometric readings, and recording their systolic blood pressure. Pulse wave velocity was also used to assess patients’ carotid-femoral artery stiffness.
Over a follow-up period of 7 years, 59 (7.8%) of 761 men and women aged 55-66 years developed ischemic heart disease or had a stroke and needed hospital treatment or died.
“Our data support the use of questions about sense of vitality in order to add prognostic information about subsequent risk of MACE independently of traditional risk markers, such as diabetes duration, age, HbA1c, gender, smoking, systolic blood pressure, and carotid-femoral pulse wave velocity and sagittal abdominal diameter,” Dr. Vergara said.
“We suggest that not feeling full of pep and not having a lot of energy could be used as risk markers for MACE in patients with type 2 diabetes.” These are two easy questions that could be asked as part of the risk assessment process in primary care, she suggested. “Other questions from the SF-36, such as feeling tired or worn out, were not independent markers of MACE,” Dr. Vergara said.
She said she had no relevant financial disclosures.
LISBON – according to the results of a primary care study.
Seldom feeling “full of pep” or not having “a lot of energy” was associated with an increased risk of a major cardiovascular event (MACE) in middle-aged (55-66 years) adults with T2DM, Marta Vergara, MD, reported at the annual meeting of the European Association for the Study of Diabetes.
“It’s well known that patients with type 2 diabetes have a high risk for developing cardiovascular disease, and it is the main cause of death,” said Dr. Vergara of Linköping (Sweden) University.
While several risk factors for cardiovascular disease are known and widely monitored for in clinical practice worldwide, including psychological aspects such as mental stress, ways to identify patients earlier are needed.
“We need more clinically useful and easy-to-manage measurement instruments,” Dr. Vergara said.
Using data from the ongoing Cardiovascular Risk Factors in Patients With Diabetes – a Prospective Study in Primary Care (CARDIPP), Dr. Vergara and colleagues identified two questions used in the 36-Item Short Form (SF-36) that might fit the bill:
- ”How much time during the past 4 weeks did you feel full of pep?”
- “How much time during the past 4 weeks did you have a lot of energy?”
Patients had answered these questions using a 6-point scale ranging from 1, all of the time, to 6 none of the time.
In addition to completing the full SF-36 at recruitment, all patients enrolled in CARDIPP underwent thorough assessment of baseline cardiovascular risk. This included asking about their duration of diabetes and smoking history, taking glycemic and anthropometric readings, and recording their systolic blood pressure. Pulse wave velocity was also used to assess patients’ carotid-femoral artery stiffness.
Over a follow-up period of 7 years, 59 (7.8%) of 761 men and women aged 55-66 years developed ischemic heart disease or had a stroke and needed hospital treatment or died.
“Our data support the use of questions about sense of vitality in order to add prognostic information about subsequent risk of MACE independently of traditional risk markers, such as diabetes duration, age, HbA1c, gender, smoking, systolic blood pressure, and carotid-femoral pulse wave velocity and sagittal abdominal diameter,” Dr. Vergara said.
“We suggest that not feeling full of pep and not having a lot of energy could be used as risk markers for MACE in patients with type 2 diabetes.” These are two easy questions that could be asked as part of the risk assessment process in primary care, she suggested. “Other questions from the SF-36, such as feeling tired or worn out, were not independent markers of MACE,” Dr. Vergara said.
She said she had no relevant financial disclosures.
AT EASD 2017
Key clinical point: Two simple questions about vitality could help assess the risk of major cardiovascular events in patients with type 2 diabetes mellitus.
Major finding: The hazard ratios for seldom feeling “full of pep” and seldom having “lots of energy” and MACE were a respective 1.31 (P = .003) and 1.44 (P less than .0001).
Data source: A prospective, observational primary care study of 761 patients with type 2 diabetes mellitus.
Disclosures: The presenting author had no relevant financial disclosures.
We’ll be there, covering the news
GI & Hepatology News reporters are geared up to cover the Liver Meeting® at the Walter E. Washington Convention Center, in Washington, starting this weekend. The annual meeting of the American Association for the Study of Liver Diseases is a worldwide meeting of liver specialists that will include presentations of new information on every level of knowledge about the liver from the hepatocyte to organ transplantation.
Onsite reporters will cover new biomarkers for nonalcoholic fatty liver disease, surveillance and treatments for hepatocellular carcinoma, and pre- and posttransplant factors that affect morbidity and mortality.
Highly anticipated presentations include:
- Early liver transplant good for patients with severe alcoholic hepatitis.
- Asians have highest rate of herbal dietary supplement DILI liver transplantations.
- Bilirubin levels associated with transplant-free survival in PBS patients.
GI & Hepatology News reporters are geared up to cover the Liver Meeting® at the Walter E. Washington Convention Center, in Washington, starting this weekend. The annual meeting of the American Association for the Study of Liver Diseases is a worldwide meeting of liver specialists that will include presentations of new information on every level of knowledge about the liver from the hepatocyte to organ transplantation.
Onsite reporters will cover new biomarkers for nonalcoholic fatty liver disease, surveillance and treatments for hepatocellular carcinoma, and pre- and posttransplant factors that affect morbidity and mortality.
Highly anticipated presentations include:
- Early liver transplant good for patients with severe alcoholic hepatitis.
- Asians have highest rate of herbal dietary supplement DILI liver transplantations.
- Bilirubin levels associated with transplant-free survival in PBS patients.
GI & Hepatology News reporters are geared up to cover the Liver Meeting® at the Walter E. Washington Convention Center, in Washington, starting this weekend. The annual meeting of the American Association for the Study of Liver Diseases is a worldwide meeting of liver specialists that will include presentations of new information on every level of knowledge about the liver from the hepatocyte to organ transplantation.
Onsite reporters will cover new biomarkers for nonalcoholic fatty liver disease, surveillance and treatments for hepatocellular carcinoma, and pre- and posttransplant factors that affect morbidity and mortality.
Highly anticipated presentations include:
- Early liver transplant good for patients with severe alcoholic hepatitis.
- Asians have highest rate of herbal dietary supplement DILI liver transplantations.
- Bilirubin levels associated with transplant-free survival in PBS patients.
Societies unite to develop cardiology subspecialty MOC products
Four medical societies are banding together to help cardiology subspecialists get through the maintenance of certification process.
The American College of Cardiology, Heart Failure Society of America, Heart Rhythm Society, and Society for Cardiovascular Angiography and Interventions are working together to develop new modules to help subspecialists meet the American Board of Internal Medicine’s current 10-year maintenance of certification examination.
The groups first must reach an agreement with ABIM; they can then collaborate to enhance the existing ACC self-assessment program (SAP) line with CathSAP, EPSAP, and Heart Failure SAP products to help fulfill the MOC needs of interventionalists, electrophysiologists, and heart failure specialists, respectively.
The societies hope to make the SAPs available in time for the ABIM rollout of the 2-year Knowledge Check-in assessment option. The current plan is for the rollout of a general cardiology product in 2019; electrophysiologists, heart failure, and interventionalists in 2020; and adult congenital in 2023.
“It is the shared goal of ACC, HFSA, HRS, and SCAI to help our collective members ensure their patients are receiving the highest-quality, evidence-based care,” ACC President Mary Norine Walsh, MD, said in a statement. “In offering additional pathways for cardiologists who wish to maintain their professional certification, we can more effectively and efficiently help busy clinicians keep up with current knowledge in their specific areas of practice.”
Four medical societies are banding together to help cardiology subspecialists get through the maintenance of certification process.
The American College of Cardiology, Heart Failure Society of America, Heart Rhythm Society, and Society for Cardiovascular Angiography and Interventions are working together to develop new modules to help subspecialists meet the American Board of Internal Medicine’s current 10-year maintenance of certification examination.
The groups first must reach an agreement with ABIM; they can then collaborate to enhance the existing ACC self-assessment program (SAP) line with CathSAP, EPSAP, and Heart Failure SAP products to help fulfill the MOC needs of interventionalists, electrophysiologists, and heart failure specialists, respectively.
The societies hope to make the SAPs available in time for the ABIM rollout of the 2-year Knowledge Check-in assessment option. The current plan is for the rollout of a general cardiology product in 2019; electrophysiologists, heart failure, and interventionalists in 2020; and adult congenital in 2023.
“It is the shared goal of ACC, HFSA, HRS, and SCAI to help our collective members ensure their patients are receiving the highest-quality, evidence-based care,” ACC President Mary Norine Walsh, MD, said in a statement. “In offering additional pathways for cardiologists who wish to maintain their professional certification, we can more effectively and efficiently help busy clinicians keep up with current knowledge in their specific areas of practice.”
Four medical societies are banding together to help cardiology subspecialists get through the maintenance of certification process.
The American College of Cardiology, Heart Failure Society of America, Heart Rhythm Society, and Society for Cardiovascular Angiography and Interventions are working together to develop new modules to help subspecialists meet the American Board of Internal Medicine’s current 10-year maintenance of certification examination.
The groups first must reach an agreement with ABIM; they can then collaborate to enhance the existing ACC self-assessment program (SAP) line with CathSAP, EPSAP, and Heart Failure SAP products to help fulfill the MOC needs of interventionalists, electrophysiologists, and heart failure specialists, respectively.
The societies hope to make the SAPs available in time for the ABIM rollout of the 2-year Knowledge Check-in assessment option. The current plan is for the rollout of a general cardiology product in 2019; electrophysiologists, heart failure, and interventionalists in 2020; and adult congenital in 2023.
“It is the shared goal of ACC, HFSA, HRS, and SCAI to help our collective members ensure their patients are receiving the highest-quality, evidence-based care,” ACC President Mary Norine Walsh, MD, said in a statement. “In offering additional pathways for cardiologists who wish to maintain their professional certification, we can more effectively and efficiently help busy clinicians keep up with current knowledge in their specific areas of practice.”
Guidelines cut acute chest syndrome hospital returns in pediatric sickle cell
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
FROM JAMA PEDIATRICS
Key clinical point: Treatment with the recommended antibiotics was effective in reducing hospital readmissions for acute chest syndrome in children and young adults up to age 22 years with sickle cell disease.
Major finding: Hospital readmission for 30-day acute chest syndrome and all-cause readmission were significantly lower (odds ratio, 0.71 and 0.50, respectively) among children with sickle cell disease who received antibiotics (macrolides and cephalosporins) according to current guidelines, compared with those who did not.
Data source: A retrospective, multicenter study of 14,480 hospitalizations at 41 locations involving 7,178 children and young adults aged 0-22 years.
Disclosures: The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.