User login
Cross-contamination of Pathology Specimens: A Cautionary Tale
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
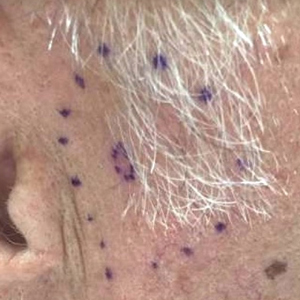
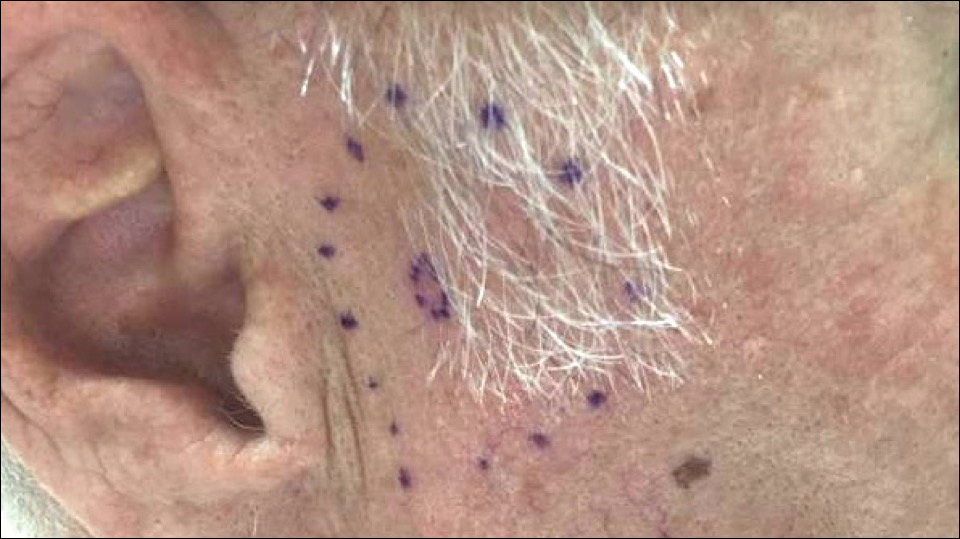
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.
The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
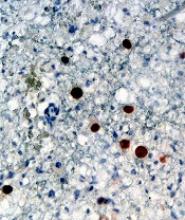
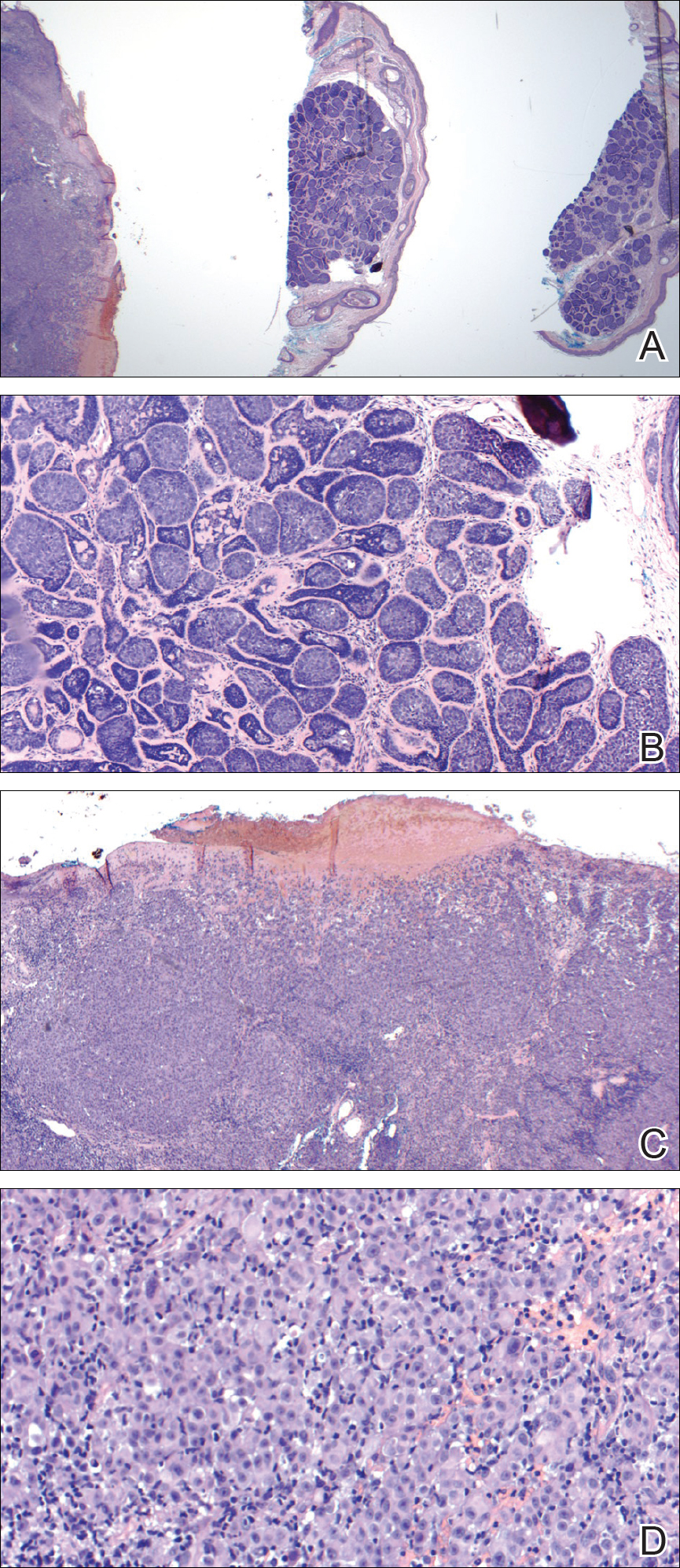
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.
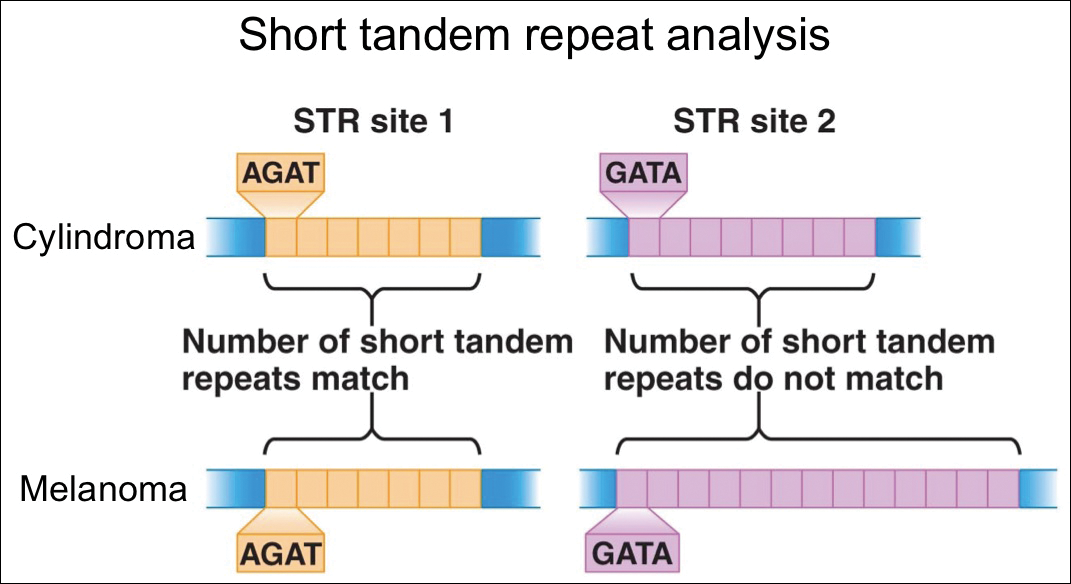
For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.
Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Resident Pearl
- Although cross-contamination of pathology specimens is rare, it does occur and can impact diagnosis and management if detected early.
Ultrasound screening improves ovarian cancer survival?
Also today, fat attenuation index boosts coronary CT prognostication, peripheral arterial disease diagnostic tests vary wildly in patients with diabetes, and big drinkers face a new massive health burden.
Amazon Alexa
Apple Podcasts
Spotify
Also today, fat attenuation index boosts coronary CT prognostication, peripheral arterial disease diagnostic tests vary wildly in patients with diabetes, and big drinkers face a new massive health burden.
Amazon Alexa
Apple Podcasts
Spotify
Also today, fat attenuation index boosts coronary CT prognostication, peripheral arterial disease diagnostic tests vary wildly in patients with diabetes, and big drinkers face a new massive health burden.
Amazon Alexa
Apple Podcasts
Spotify
Study supports sequencing in kids with cancer
SAN DIEGO—Comprehensive next-generation sequencing is both feasible and clinically useful in pediatric cancer patients, a new study suggests.
Researchers sequenced samples from 253 pediatric cancer patients and found that, in 79% of cases, there was at least one finding that could help guide care.
Scott Newman, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, presented these findings at the American Society of Human Genetics (ASHG) 2018 Annual Meeting (abstract 52).
The researchers conducted whole-genome, exome, and transcriptome sequencing of the patients’ tumors, as well as sequencing non-cancerous tissues from the same patients.
Of the 253 patients studied, 123 had hematologic malignancies.
The researchers found a mean of four pathogenic or likely pathogenic variants per patient (range, 0-18). This included prognostic (21.8%) and diagnostic (15.1%) variants as well as variants that could be targeted therapeutically (6.8%).
In all, 79% of the patients had at least one variant that was targetable, diagnostic, or prognostic. And test results were available within about 40 days, quickly enough that they could be used to guide care.
“With results available in a clinically relevant time frame, and pricing becoming increasingly comparable to the radiology and pathology tests, WGS [whole-genome sequencing] is becoming more accessible to pediatric oncology patients,” Dr. Newman said.
This work was part of the Genomes for Kids study (G4K), an effort to understand how best to use genetic data for pediatric cancer diagnosis and treatment. St. Jude has compiled the information from G4K into a publicly accessible online database.
The researchers have continued to perform sequencing on current patients, and, since the original study ended, have successfully used this method on roughly 300 additional patients. The team plans to continue studying sequencing methods in hopes of producing clinically applicable data more quickly.
G4K was sponsored by St. Jude.
SAN DIEGO—Comprehensive next-generation sequencing is both feasible and clinically useful in pediatric cancer patients, a new study suggests.
Researchers sequenced samples from 253 pediatric cancer patients and found that, in 79% of cases, there was at least one finding that could help guide care.
Scott Newman, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, presented these findings at the American Society of Human Genetics (ASHG) 2018 Annual Meeting (abstract 52).
The researchers conducted whole-genome, exome, and transcriptome sequencing of the patients’ tumors, as well as sequencing non-cancerous tissues from the same patients.
Of the 253 patients studied, 123 had hematologic malignancies.
The researchers found a mean of four pathogenic or likely pathogenic variants per patient (range, 0-18). This included prognostic (21.8%) and diagnostic (15.1%) variants as well as variants that could be targeted therapeutically (6.8%).
In all, 79% of the patients had at least one variant that was targetable, diagnostic, or prognostic. And test results were available within about 40 days, quickly enough that they could be used to guide care.
“With results available in a clinically relevant time frame, and pricing becoming increasingly comparable to the radiology and pathology tests, WGS [whole-genome sequencing] is becoming more accessible to pediatric oncology patients,” Dr. Newman said.
This work was part of the Genomes for Kids study (G4K), an effort to understand how best to use genetic data for pediatric cancer diagnosis and treatment. St. Jude has compiled the information from G4K into a publicly accessible online database.
The researchers have continued to perform sequencing on current patients, and, since the original study ended, have successfully used this method on roughly 300 additional patients. The team plans to continue studying sequencing methods in hopes of producing clinically applicable data more quickly.
G4K was sponsored by St. Jude.
SAN DIEGO—Comprehensive next-generation sequencing is both feasible and clinically useful in pediatric cancer patients, a new study suggests.
Researchers sequenced samples from 253 pediatric cancer patients and found that, in 79% of cases, there was at least one finding that could help guide care.
Scott Newman, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, presented these findings at the American Society of Human Genetics (ASHG) 2018 Annual Meeting (abstract 52).
The researchers conducted whole-genome, exome, and transcriptome sequencing of the patients’ tumors, as well as sequencing non-cancerous tissues from the same patients.
Of the 253 patients studied, 123 had hematologic malignancies.
The researchers found a mean of four pathogenic or likely pathogenic variants per patient (range, 0-18). This included prognostic (21.8%) and diagnostic (15.1%) variants as well as variants that could be targeted therapeutically (6.8%).
In all, 79% of the patients had at least one variant that was targetable, diagnostic, or prognostic. And test results were available within about 40 days, quickly enough that they could be used to guide care.
“With results available in a clinically relevant time frame, and pricing becoming increasingly comparable to the radiology and pathology tests, WGS [whole-genome sequencing] is becoming more accessible to pediatric oncology patients,” Dr. Newman said.
This work was part of the Genomes for Kids study (G4K), an effort to understand how best to use genetic data for pediatric cancer diagnosis and treatment. St. Jude has compiled the information from G4K into a publicly accessible online database.
The researchers have continued to perform sequencing on current patients, and, since the original study ended, have successfully used this method on roughly 300 additional patients. The team plans to continue studying sequencing methods in hopes of producing clinically applicable data more quickly.
G4K was sponsored by St. Jude.
‘Intense’ end-of-life care may be common in HSCT recipients
Patients who die within a year of allogeneic hematopoietic stem cell transplant (HSCT) tend to receive “medically intense” end-of-life care, an analysis suggests.
Researchers studied more than 2,000 patients who died within a year of allogeneic HSCT and found that a majority of the patients died in the hospital, and about half of them were admitted to the intensive care unit (ICU).
However, patient age, underlying diagnosis, and other factors influenced the likelihood of receiving intense end-of-life care.
For example, patients diagnosed with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS) were less likely than patients with acute lymphoblastic leukemia (ALL) to receive medically intense care.
Emily Johnston, MD, of the University of Alabama at Birmingham, and her colleagues reported these findings in the Journal of Clinical Oncology.
The researchers studied 2,135 patients in California who underwent inpatient HSCT and died within a year of the transplant (not as a result of peripartum events or trauma) between 2000 and 2013.
Fifty-three percent of the patients received some type of medically intense intervention, and 57% had at least two types of intense interventions.
Eighty-three percent of patients died in hospital, and 43% spent all of their last 30 days in the hospital.
Forty-nine percent of patients were admitted to the ICU, 45% were intubated, 22% underwent hemodialysis, and 8% received cardiopulmonary resuscitation.
Factors associated with intense care
The researchers said receipt of a medically intense intervention varied by age at death, underlying diagnosis, year of HSCT, location of care, and comorbidities. However, use of intense interventions did not vary according to sex, race/ethnicity, insurance type, or income.
Compared to patients age 60 and older, patients in the following age groups were more likely to receive medically intense interventions:
- Ages 15 to 21—odds ratio (OR)=2.6 (P<0.001)
- Ages 30 to 39—OR=1.8 (P<0.01)
- Ages 40 to 49—OR=1.4 (P<0.05).
Patients with comorbidities were more likely to receive intense interventions as well. The OR was 1.6 (P<0.01) for patients with one comorbidity and 2.5 (P<0.001) for patients with two or more comorbidities.
Patients with AML or MDS were less likely than patients with ALL to receive a medically intense intervention—OR=0.7 (P<0.05).
Patients who were transplanted between 2000 and 2004 were less likely to receive an intense intervention than patients transplanted between 2010 and 2013—OR=0.7 (P<0.01).
Patients who changed hospitals between HSCT and death were less likely to receive an intense intervention than patients who stayed at the same hospital. The OR was 0.3 if they transferred to a community hospital and 0.4 if they transferred to a specialty hospital (P<0.001 for both).
Patients living in rural areas were less likely than urban patients to receive a medically intense intervention—OR=0.6 (P<0.05).
“From our data, we understand there is a correlation with high-intensity end-of-life care in patients who die within one year after receiving a stem cell transplant, but we are still unsure if that was the care they wanted,” Dr. Johnston said.
“The findings suggest that, as oncologists, we need to start having end-of-life care conversations earlier with patients to determine if a high-intensity treatment plan is consistent with their goals or if a lower-intensity treatment plan is best. It’s not a one-size-fits-all approach in end-of-life care.”
This research was supported by Stanford University. One study author reported relationships with Corvus Pharmaceuticals, Shire Pharmaceuticals, and Adaptive Biotechnologies. All other authors reported no conflicts.
Patients who die within a year of allogeneic hematopoietic stem cell transplant (HSCT) tend to receive “medically intense” end-of-life care, an analysis suggests.
Researchers studied more than 2,000 patients who died within a year of allogeneic HSCT and found that a majority of the patients died in the hospital, and about half of them were admitted to the intensive care unit (ICU).
However, patient age, underlying diagnosis, and other factors influenced the likelihood of receiving intense end-of-life care.
For example, patients diagnosed with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS) were less likely than patients with acute lymphoblastic leukemia (ALL) to receive medically intense care.
Emily Johnston, MD, of the University of Alabama at Birmingham, and her colleagues reported these findings in the Journal of Clinical Oncology.
The researchers studied 2,135 patients in California who underwent inpatient HSCT and died within a year of the transplant (not as a result of peripartum events or trauma) between 2000 and 2013.
Fifty-three percent of the patients received some type of medically intense intervention, and 57% had at least two types of intense interventions.
Eighty-three percent of patients died in hospital, and 43% spent all of their last 30 days in the hospital.
Forty-nine percent of patients were admitted to the ICU, 45% were intubated, 22% underwent hemodialysis, and 8% received cardiopulmonary resuscitation.
Factors associated with intense care
The researchers said receipt of a medically intense intervention varied by age at death, underlying diagnosis, year of HSCT, location of care, and comorbidities. However, use of intense interventions did not vary according to sex, race/ethnicity, insurance type, or income.
Compared to patients age 60 and older, patients in the following age groups were more likely to receive medically intense interventions:
- Ages 15 to 21—odds ratio (OR)=2.6 (P<0.001)
- Ages 30 to 39—OR=1.8 (P<0.01)
- Ages 40 to 49—OR=1.4 (P<0.05).
Patients with comorbidities were more likely to receive intense interventions as well. The OR was 1.6 (P<0.01) for patients with one comorbidity and 2.5 (P<0.001) for patients with two or more comorbidities.
Patients with AML or MDS were less likely than patients with ALL to receive a medically intense intervention—OR=0.7 (P<0.05).
Patients who were transplanted between 2000 and 2004 were less likely to receive an intense intervention than patients transplanted between 2010 and 2013—OR=0.7 (P<0.01).
Patients who changed hospitals between HSCT and death were less likely to receive an intense intervention than patients who stayed at the same hospital. The OR was 0.3 if they transferred to a community hospital and 0.4 if they transferred to a specialty hospital (P<0.001 for both).
Patients living in rural areas were less likely than urban patients to receive a medically intense intervention—OR=0.6 (P<0.05).
“From our data, we understand there is a correlation with high-intensity end-of-life care in patients who die within one year after receiving a stem cell transplant, but we are still unsure if that was the care they wanted,” Dr. Johnston said.
“The findings suggest that, as oncologists, we need to start having end-of-life care conversations earlier with patients to determine if a high-intensity treatment plan is consistent with their goals or if a lower-intensity treatment plan is best. It’s not a one-size-fits-all approach in end-of-life care.”
This research was supported by Stanford University. One study author reported relationships with Corvus Pharmaceuticals, Shire Pharmaceuticals, and Adaptive Biotechnologies. All other authors reported no conflicts.
Patients who die within a year of allogeneic hematopoietic stem cell transplant (HSCT) tend to receive “medically intense” end-of-life care, an analysis suggests.
Researchers studied more than 2,000 patients who died within a year of allogeneic HSCT and found that a majority of the patients died in the hospital, and about half of them were admitted to the intensive care unit (ICU).
However, patient age, underlying diagnosis, and other factors influenced the likelihood of receiving intense end-of-life care.
For example, patients diagnosed with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS) were less likely than patients with acute lymphoblastic leukemia (ALL) to receive medically intense care.
Emily Johnston, MD, of the University of Alabama at Birmingham, and her colleagues reported these findings in the Journal of Clinical Oncology.
The researchers studied 2,135 patients in California who underwent inpatient HSCT and died within a year of the transplant (not as a result of peripartum events or trauma) between 2000 and 2013.
Fifty-three percent of the patients received some type of medically intense intervention, and 57% had at least two types of intense interventions.
Eighty-three percent of patients died in hospital, and 43% spent all of their last 30 days in the hospital.
Forty-nine percent of patients were admitted to the ICU, 45% were intubated, 22% underwent hemodialysis, and 8% received cardiopulmonary resuscitation.
Factors associated with intense care
The researchers said receipt of a medically intense intervention varied by age at death, underlying diagnosis, year of HSCT, location of care, and comorbidities. However, use of intense interventions did not vary according to sex, race/ethnicity, insurance type, or income.
Compared to patients age 60 and older, patients in the following age groups were more likely to receive medically intense interventions:
- Ages 15 to 21—odds ratio (OR)=2.6 (P<0.001)
- Ages 30 to 39—OR=1.8 (P<0.01)
- Ages 40 to 49—OR=1.4 (P<0.05).
Patients with comorbidities were more likely to receive intense interventions as well. The OR was 1.6 (P<0.01) for patients with one comorbidity and 2.5 (P<0.001) for patients with two or more comorbidities.
Patients with AML or MDS were less likely than patients with ALL to receive a medically intense intervention—OR=0.7 (P<0.05).
Patients who were transplanted between 2000 and 2004 were less likely to receive an intense intervention than patients transplanted between 2010 and 2013—OR=0.7 (P<0.01).
Patients who changed hospitals between HSCT and death were less likely to receive an intense intervention than patients who stayed at the same hospital. The OR was 0.3 if they transferred to a community hospital and 0.4 if they transferred to a specialty hospital (P<0.001 for both).
Patients living in rural areas were less likely than urban patients to receive a medically intense intervention—OR=0.6 (P<0.05).
“From our data, we understand there is a correlation with high-intensity end-of-life care in patients who die within one year after receiving a stem cell transplant, but we are still unsure if that was the care they wanted,” Dr. Johnston said.
“The findings suggest that, as oncologists, we need to start having end-of-life care conversations earlier with patients to determine if a high-intensity treatment plan is consistent with their goals or if a lower-intensity treatment plan is best. It’s not a one-size-fits-all approach in end-of-life care.”
This research was supported by Stanford University. One study author reported relationships with Corvus Pharmaceuticals, Shire Pharmaceuticals, and Adaptive Biotechnologies. All other authors reported no conflicts.
Adoptive T-cell therapy treats PML
Adoptive T-cell therapy has proven effective for treating progressive multifocal leukoencephalopathy (PML), according to research published in The New England Journal of Medicine.
Researchers observed substantial improvements in three PML patients infused with donor T cells targeting the BK virus.
Although one patient ultimately died, two had complete clearance of the JC virus and no clinical signs of PML after treatment.
“The JC and BK viruses are genetically similar and share proteins that can be targeted by the immune system,” said study author Katy Rezvani, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Because of these similarities, we hypothesized that T cells developed against BK virus may also be effective against JC virus infection.”
Dr. Rezvani’s team developed a novel approach for the generation of BK virus-specific T cells from healthy donors and established a bank of viral-specific T cells for immediate clinical use.
The researchers treated three patients with third-party, partially human leukocyte antigen (HLA)-matched, BK virus-specific T cells.
Patient 1 was a 32-year-old female with acute myeloid leukemia (AML) who previously received a double cord blood transplant.
Patient 2 was a 73-year-old female with JAK2-positive polycythemia rubra vera (PV) who had been treated with ruxolitinib.
Patient 3 was a 35-year-old man with AIDS who had discontinued highly active antiretroviral therapy due to side effects and who was no longer able to walk.
Following the first infusion, all three patients had a reduction in JC viral load in their cerebrospinal fluid. Viral loads dropped from:
- 700 to 78 copies in the AML patient
- 230,000 to 5,200 in the PV patient
- 4,300 to 1,300 in the AIDS patient.
“After infusion of viral-specific T cells, patients 1 and 3 had clinical improvement with significant reduction in JC virus in their cerebrospinal fluid,” Dr. Rezvani said.
“Both patients responded despite persistent T-cell immunodeficiency, supporting the concept that the response was mediated by the adoptively infused viral-specific T cells, and there were no infusion-related reactions.”
The AML patient received two additional infusions, which resulted in clearance of the virus in the cerebrospinal fluid and no signs of PML 27 months after the first infusion.
The PV patient received a second infusion that further reduced JC viral load, but no additional improvement was seen. The patient died 8 months after the first infusion.
The AIDS patient received additional infusions, resulting in complete clearance of the JC virus. This patient has regained mobility, and, 9 months after the first infusion, he is able to walk with a cane.
“We are encouraged that off-the-shelf, third-party, partially HLA-matched BK viral-specific T cells may provide a therapy for PML,” Dr. Rezvani said. “Further study in a larger group of patients is required to determine the success rate, durability, and longer-term adverse events with this treatment.”
This study was supported with funding from the Myelodysplastic Syndromes and Acute Myeloid Leukemia Moon Shot, part of MD Anderson’s Moon Shots Program, as well as the National Institutes of Health.
Adoptive T-cell therapy has proven effective for treating progressive multifocal leukoencephalopathy (PML), according to research published in The New England Journal of Medicine.
Researchers observed substantial improvements in three PML patients infused with donor T cells targeting the BK virus.
Although one patient ultimately died, two had complete clearance of the JC virus and no clinical signs of PML after treatment.
“The JC and BK viruses are genetically similar and share proteins that can be targeted by the immune system,” said study author Katy Rezvani, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Because of these similarities, we hypothesized that T cells developed against BK virus may also be effective against JC virus infection.”
Dr. Rezvani’s team developed a novel approach for the generation of BK virus-specific T cells from healthy donors and established a bank of viral-specific T cells for immediate clinical use.
The researchers treated three patients with third-party, partially human leukocyte antigen (HLA)-matched, BK virus-specific T cells.
Patient 1 was a 32-year-old female with acute myeloid leukemia (AML) who previously received a double cord blood transplant.
Patient 2 was a 73-year-old female with JAK2-positive polycythemia rubra vera (PV) who had been treated with ruxolitinib.
Patient 3 was a 35-year-old man with AIDS who had discontinued highly active antiretroviral therapy due to side effects and who was no longer able to walk.
Following the first infusion, all three patients had a reduction in JC viral load in their cerebrospinal fluid. Viral loads dropped from:
- 700 to 78 copies in the AML patient
- 230,000 to 5,200 in the PV patient
- 4,300 to 1,300 in the AIDS patient.
“After infusion of viral-specific T cells, patients 1 and 3 had clinical improvement with significant reduction in JC virus in their cerebrospinal fluid,” Dr. Rezvani said.
“Both patients responded despite persistent T-cell immunodeficiency, supporting the concept that the response was mediated by the adoptively infused viral-specific T cells, and there were no infusion-related reactions.”
The AML patient received two additional infusions, which resulted in clearance of the virus in the cerebrospinal fluid and no signs of PML 27 months after the first infusion.
The PV patient received a second infusion that further reduced JC viral load, but no additional improvement was seen. The patient died 8 months after the first infusion.
The AIDS patient received additional infusions, resulting in complete clearance of the JC virus. This patient has regained mobility, and, 9 months after the first infusion, he is able to walk with a cane.
“We are encouraged that off-the-shelf, third-party, partially HLA-matched BK viral-specific T cells may provide a therapy for PML,” Dr. Rezvani said. “Further study in a larger group of patients is required to determine the success rate, durability, and longer-term adverse events with this treatment.”
This study was supported with funding from the Myelodysplastic Syndromes and Acute Myeloid Leukemia Moon Shot, part of MD Anderson’s Moon Shots Program, as well as the National Institutes of Health.
Adoptive T-cell therapy has proven effective for treating progressive multifocal leukoencephalopathy (PML), according to research published in The New England Journal of Medicine.
Researchers observed substantial improvements in three PML patients infused with donor T cells targeting the BK virus.
Although one patient ultimately died, two had complete clearance of the JC virus and no clinical signs of PML after treatment.
“The JC and BK viruses are genetically similar and share proteins that can be targeted by the immune system,” said study author Katy Rezvani, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Because of these similarities, we hypothesized that T cells developed against BK virus may also be effective against JC virus infection.”
Dr. Rezvani’s team developed a novel approach for the generation of BK virus-specific T cells from healthy donors and established a bank of viral-specific T cells for immediate clinical use.
The researchers treated three patients with third-party, partially human leukocyte antigen (HLA)-matched, BK virus-specific T cells.
Patient 1 was a 32-year-old female with acute myeloid leukemia (AML) who previously received a double cord blood transplant.
Patient 2 was a 73-year-old female with JAK2-positive polycythemia rubra vera (PV) who had been treated with ruxolitinib.
Patient 3 was a 35-year-old man with AIDS who had discontinued highly active antiretroviral therapy due to side effects and who was no longer able to walk.
Following the first infusion, all three patients had a reduction in JC viral load in their cerebrospinal fluid. Viral loads dropped from:
- 700 to 78 copies in the AML patient
- 230,000 to 5,200 in the PV patient
- 4,300 to 1,300 in the AIDS patient.
“After infusion of viral-specific T cells, patients 1 and 3 had clinical improvement with significant reduction in JC virus in their cerebrospinal fluid,” Dr. Rezvani said.
“Both patients responded despite persistent T-cell immunodeficiency, supporting the concept that the response was mediated by the adoptively infused viral-specific T cells, and there were no infusion-related reactions.”
The AML patient received two additional infusions, which resulted in clearance of the virus in the cerebrospinal fluid and no signs of PML 27 months after the first infusion.
The PV patient received a second infusion that further reduced JC viral load, but no additional improvement was seen. The patient died 8 months after the first infusion.
The AIDS patient received additional infusions, resulting in complete clearance of the JC virus. This patient has regained mobility, and, 9 months after the first infusion, he is able to walk with a cane.
“We are encouraged that off-the-shelf, third-party, partially HLA-matched BK viral-specific T cells may provide a therapy for PML,” Dr. Rezvani said. “Further study in a larger group of patients is required to determine the success rate, durability, and longer-term adverse events with this treatment.”
This study was supported with funding from the Myelodysplastic Syndromes and Acute Myeloid Leukemia Moon Shot, part of MD Anderson’s Moon Shots Program, as well as the National Institutes of Health.
XR-naltrexone beats oral medication for opioid use disorder
Monthly injection is tied to twice rate of treatment retention
Opioid addiction patients given extended-release naltrexone had about twice the rate of treatment retention at 6 months as those given oral naltrexone, a study of 60 adults shows.
“These study findings have immediate clinical relevance for treatment of opioid use disorder, at a time when an opioid epidemic continues unabated in the United States,” wrote Maria A. Sullivan, MD, PhD, of Columbia University, New York, and her colleagues.
Naltrexone remains an alternative to agonist treatment for opioid use disorder, but adherence to a daily pill regimen is often poor. “Extended-release (XR) parenteral formulations of naltrexone, as monthly injection or implants, circumvent the daily pill requirement and have shown promising effectiveness,” the researchers said.
In a study published in the American Journal of Psychiatry, 60 opioid-dependent adults who had completed an inpatient detoxification program were randomized to 50 mg/day of oral naltrexone (or 100 mg on Mondays and Wednesdays and 150 mg on Fridays for those living alone) plus behavior therapy or a 380-mg intramuscular injection of XR-naltrexone monthly plus behavior therapy.
After 6 months, 57% of patients in the XR-naltrexone group were retained in treatment, compared with 28% of the oral group (hazard ratio, 2.18), reported Dr. Sullivan and her colleagues.
A total of 40 adverse events were reported. Insomnia was the most common and occurred in 52% of the patients; it was more common in the oral group, compared with the extended-release group (70% vs. 35%, respectively). A total of nine serious adverse events were reported, and five participants discontinued the study as a result. However, only one event, a case of hives determined to be an allergic reaction, was attributed to the study drug.
The findings were limited by several factors, including the small size, open-label design, and lack of urine data for patients after they discontinued the study, the researchers noted. However, the results “support the use of XR-naltrexone combined with behavioral therapy as an effective treatment for patients seeking opioid withdrawal and nonagonist treatment for preventing relapse to opioid use disorder,” they said. In addition, given the difference in effectiveness and the high risk of a failed treatment with oral naltrexone,” they said.
The study was supported in part by grants from the National Institute on Drug Abuse. Dr. Sullivan had no financial conflicts to disclose.
SOURCE: Sullivan MA et al. Am J Psychiatry. 2018. doi: 10.1176/appi.ajp.2018.17070732.
Monthly injection is tied to twice rate of treatment retention
Monthly injection is tied to twice rate of treatment retention
Opioid addiction patients given extended-release naltrexone had about twice the rate of treatment retention at 6 months as those given oral naltrexone, a study of 60 adults shows.
“These study findings have immediate clinical relevance for treatment of opioid use disorder, at a time when an opioid epidemic continues unabated in the United States,” wrote Maria A. Sullivan, MD, PhD, of Columbia University, New York, and her colleagues.
Naltrexone remains an alternative to agonist treatment for opioid use disorder, but adherence to a daily pill regimen is often poor. “Extended-release (XR) parenteral formulations of naltrexone, as monthly injection or implants, circumvent the daily pill requirement and have shown promising effectiveness,” the researchers said.
In a study published in the American Journal of Psychiatry, 60 opioid-dependent adults who had completed an inpatient detoxification program were randomized to 50 mg/day of oral naltrexone (or 100 mg on Mondays and Wednesdays and 150 mg on Fridays for those living alone) plus behavior therapy or a 380-mg intramuscular injection of XR-naltrexone monthly plus behavior therapy.
After 6 months, 57% of patients in the XR-naltrexone group were retained in treatment, compared with 28% of the oral group (hazard ratio, 2.18), reported Dr. Sullivan and her colleagues.
A total of 40 adverse events were reported. Insomnia was the most common and occurred in 52% of the patients; it was more common in the oral group, compared with the extended-release group (70% vs. 35%, respectively). A total of nine serious adverse events were reported, and five participants discontinued the study as a result. However, only one event, a case of hives determined to be an allergic reaction, was attributed to the study drug.
The findings were limited by several factors, including the small size, open-label design, and lack of urine data for patients after they discontinued the study, the researchers noted. However, the results “support the use of XR-naltrexone combined with behavioral therapy as an effective treatment for patients seeking opioid withdrawal and nonagonist treatment for preventing relapse to opioid use disorder,” they said. In addition, given the difference in effectiveness and the high risk of a failed treatment with oral naltrexone,” they said.
The study was supported in part by grants from the National Institute on Drug Abuse. Dr. Sullivan had no financial conflicts to disclose.
SOURCE: Sullivan MA et al. Am J Psychiatry. 2018. doi: 10.1176/appi.ajp.2018.17070732.
Opioid addiction patients given extended-release naltrexone had about twice the rate of treatment retention at 6 months as those given oral naltrexone, a study of 60 adults shows.
“These study findings have immediate clinical relevance for treatment of opioid use disorder, at a time when an opioid epidemic continues unabated in the United States,” wrote Maria A. Sullivan, MD, PhD, of Columbia University, New York, and her colleagues.
Naltrexone remains an alternative to agonist treatment for opioid use disorder, but adherence to a daily pill regimen is often poor. “Extended-release (XR) parenteral formulations of naltrexone, as monthly injection or implants, circumvent the daily pill requirement and have shown promising effectiveness,” the researchers said.
In a study published in the American Journal of Psychiatry, 60 opioid-dependent adults who had completed an inpatient detoxification program were randomized to 50 mg/day of oral naltrexone (or 100 mg on Mondays and Wednesdays and 150 mg on Fridays for those living alone) plus behavior therapy or a 380-mg intramuscular injection of XR-naltrexone monthly plus behavior therapy.
After 6 months, 57% of patients in the XR-naltrexone group were retained in treatment, compared with 28% of the oral group (hazard ratio, 2.18), reported Dr. Sullivan and her colleagues.
A total of 40 adverse events were reported. Insomnia was the most common and occurred in 52% of the patients; it was more common in the oral group, compared with the extended-release group (70% vs. 35%, respectively). A total of nine serious adverse events were reported, and five participants discontinued the study as a result. However, only one event, a case of hives determined to be an allergic reaction, was attributed to the study drug.
The findings were limited by several factors, including the small size, open-label design, and lack of urine data for patients after they discontinued the study, the researchers noted. However, the results “support the use of XR-naltrexone combined with behavioral therapy as an effective treatment for patients seeking opioid withdrawal and nonagonist treatment for preventing relapse to opioid use disorder,” they said. In addition, given the difference in effectiveness and the high risk of a failed treatment with oral naltrexone,” they said.
The study was supported in part by grants from the National Institute on Drug Abuse. Dr. Sullivan had no financial conflicts to disclose.
SOURCE: Sullivan MA et al. Am J Psychiatry. 2018. doi: 10.1176/appi.ajp.2018.17070732.
FROM THE AMERICAN JOURNAL OF PSYCHIATRY
Key clinical point: Extended-release injected naltrexone retains about twice as many patients in treatment for opioid abuse as oral naltrexone.
Major finding: Significantly more extended-release naltrexone patients remained in treatment, compared with oral naltrexone patients (57% vs. 21%).
Study details: The data come from 60 adults with opioid use disorder who transitioned to outpatient treatment.
Disclosures: The study was supported in part by grants from the National Institute on Drug Abuse. Dr. Sullivan had no financial conflicts to disclose.
Source: Sullivan MA et al. Am J Psychiatry. 2018. doi: 10.1176/appi.ajp.2018.17070732.
Pay attention to kidney disease risk in people living with HIV
The prevalence of chronic kidney disease (CKD) in people living with HIV varied widely, depending on population and criteria, according to a systematic literature review of the PubMed and PsycInfo databases for articles published from January 2000 through August 2016.
The review included all studies that involved adults older than 21 years of age, investigated people living with HIV with CKD, reported prevalence of CKD, and were published in a peer-reviewed journal, according to Jungmin Park, PhD, RN, of CHA University, Pocheon-Si, South Korea, and her colleague.
Out of an initial search yielding 1,960 citations in PubMed and 5,356 citations in PsycInfo, the results were pared down to 21 articles, which met all of the inclusion/exclusion criteria and were used for the final analysis.
The risk factors for CKD in people living with HIV cited most often in the studies consisted of medications, hypertension, older age, diabetes mellitus, hepatitis coinfection (with hepatitis C virus more prominent than hepatitis B virus), low CD4+ T-cell count, and race, Dr. Park and her colleague reported.
Of the various risk factors, the only ones unique to HIV were viral load and CD4+ T-cell count. One study reporting on 5,538 treatment-naive patients in mainland China suggested that HIV viral replication in renal cells may be the cause of renal damage in patients with high viral loads, meaning that viral suppression would improve renal function. However, all of these risk factors are intrinsically linked, according to Dr. Park and her colleague. They added that managing viral load alone would be ineffective in preventing CKD: “Therefore [people living with HIV] will need to effectively manage every aspect of their health, including metabolic and cardiovascular systems.”
Of the 43,114 people living with HIV across the 21 studies, 3,218 (7.3%) had CKD. The reported prevalence of CKD ranged from 2.3% to 53.3%, with the African population having the highest prevalence. Some of the wide variation was possibly attributable to differences in the definitions of CKD used across the various studies.
“The risk of under-diagnosis of CKD can lead to long-term health complications. Health care providers must monitor kidney function and treatment for renal damage carefully, especially for people living with HIV with additional diagnoses of diabetes and/or hypertension, and for those who are aging,” Dr. Park and her colleague concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Park, J et al. J Assoc Nurses AIDS Care. 2018;29:655-66.
The prevalence of chronic kidney disease (CKD) in people living with HIV varied widely, depending on population and criteria, according to a systematic literature review of the PubMed and PsycInfo databases for articles published from January 2000 through August 2016.
The review included all studies that involved adults older than 21 years of age, investigated people living with HIV with CKD, reported prevalence of CKD, and were published in a peer-reviewed journal, according to Jungmin Park, PhD, RN, of CHA University, Pocheon-Si, South Korea, and her colleague.
Out of an initial search yielding 1,960 citations in PubMed and 5,356 citations in PsycInfo, the results were pared down to 21 articles, which met all of the inclusion/exclusion criteria and were used for the final analysis.
The risk factors for CKD in people living with HIV cited most often in the studies consisted of medications, hypertension, older age, diabetes mellitus, hepatitis coinfection (with hepatitis C virus more prominent than hepatitis B virus), low CD4+ T-cell count, and race, Dr. Park and her colleague reported.
Of the various risk factors, the only ones unique to HIV were viral load and CD4+ T-cell count. One study reporting on 5,538 treatment-naive patients in mainland China suggested that HIV viral replication in renal cells may be the cause of renal damage in patients with high viral loads, meaning that viral suppression would improve renal function. However, all of these risk factors are intrinsically linked, according to Dr. Park and her colleague. They added that managing viral load alone would be ineffective in preventing CKD: “Therefore [people living with HIV] will need to effectively manage every aspect of their health, including metabolic and cardiovascular systems.”
Of the 43,114 people living with HIV across the 21 studies, 3,218 (7.3%) had CKD. The reported prevalence of CKD ranged from 2.3% to 53.3%, with the African population having the highest prevalence. Some of the wide variation was possibly attributable to differences in the definitions of CKD used across the various studies.
“The risk of under-diagnosis of CKD can lead to long-term health complications. Health care providers must monitor kidney function and treatment for renal damage carefully, especially for people living with HIV with additional diagnoses of diabetes and/or hypertension, and for those who are aging,” Dr. Park and her colleague concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Park, J et al. J Assoc Nurses AIDS Care. 2018;29:655-66.
The prevalence of chronic kidney disease (CKD) in people living with HIV varied widely, depending on population and criteria, according to a systematic literature review of the PubMed and PsycInfo databases for articles published from January 2000 through August 2016.
The review included all studies that involved adults older than 21 years of age, investigated people living with HIV with CKD, reported prevalence of CKD, and were published in a peer-reviewed journal, according to Jungmin Park, PhD, RN, of CHA University, Pocheon-Si, South Korea, and her colleague.
Out of an initial search yielding 1,960 citations in PubMed and 5,356 citations in PsycInfo, the results were pared down to 21 articles, which met all of the inclusion/exclusion criteria and were used for the final analysis.
The risk factors for CKD in people living with HIV cited most often in the studies consisted of medications, hypertension, older age, diabetes mellitus, hepatitis coinfection (with hepatitis C virus more prominent than hepatitis B virus), low CD4+ T-cell count, and race, Dr. Park and her colleague reported.
Of the various risk factors, the only ones unique to HIV were viral load and CD4+ T-cell count. One study reporting on 5,538 treatment-naive patients in mainland China suggested that HIV viral replication in renal cells may be the cause of renal damage in patients with high viral loads, meaning that viral suppression would improve renal function. However, all of these risk factors are intrinsically linked, according to Dr. Park and her colleague. They added that managing viral load alone would be ineffective in preventing CKD: “Therefore [people living with HIV] will need to effectively manage every aspect of their health, including metabolic and cardiovascular systems.”
Of the 43,114 people living with HIV across the 21 studies, 3,218 (7.3%) had CKD. The reported prevalence of CKD ranged from 2.3% to 53.3%, with the African population having the highest prevalence. Some of the wide variation was possibly attributable to differences in the definitions of CKD used across the various studies.
“The risk of under-diagnosis of CKD can lead to long-term health complications. Health care providers must monitor kidney function and treatment for renal damage carefully, especially for people living with HIV with additional diagnoses of diabetes and/or hypertension, and for those who are aging,” Dr. Park and her colleague concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Park, J et al. J Assoc Nurses AIDS Care. 2018;29:655-66.
FROM THE JOURNAL OF THE ASSOCIATION OF NURSES IN AIDS CARE
Key clinical point: Chronic kidney disease in people living with HIV varies widely across geographic regions.
Major finding: The reported prevalence of CKD in PLWH ranged from 2.3% to 53.3%, with the African population having the highest prevalence.
Study details: Systematic literature review of the PubMed and PsycInfo databases for articles published from January 2000 through August 2016.
Disclosures: The authors reported that they had no conflicts of interest.
Source: J Assoc Nurses AIDS Care. 2018;29:655-66).
HIV testing low in U.S. women engaged in risky behavior
HIV testing rates were low among women whose sexual behaviors increased their risk of HIV infection, and they were especially low among women who reported having anal sex, according to a report published in the American Journal of Obstetrics & Gynecology.
Data from the 2011-2015 National Survey of Family Growth were analyzed to estimate the proportion of sexually active, nonpregnant U.S. women aged 15-44 years who had had an HIV test within the past year. The data was stratified by those who reported anal sex and other risk factors, including having more than two sexual partners, condomless sex with a new partner or multiple partners, gonorrhea in the past year, or any history of syphilis, according to Mary Evans, MD, of the National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention at the Centers for Disease Control and Prevention and her colleagues.
Among the 42.4 million sexually active women assessed, 9.0 million (20%) reported they had had anal sex in the past year. Of these, 19% reported that their providers asked about their types of intercourse, and 20% reported an HIV test within the past year. Overall, HIV testing was higher among women who reported anal sex and whose providers asked about types of sex engaged in than it was among those women whose provider did not ask (38% vs. 16%, respectively; P less than .001). However, HIV testing in the past year was higher for women with other forms of risky behaviors as compared with anal sex, ranging from 35.8% to 47.2%.
“Women who report sexual behaviors such as anal sex would benefit from an HIV test and an assessment for [prevention with preexposure prophylaxis] eligibility. Women’s health care providers are uniquely poised to provide HIV prevention for women who tend to have frequent encounters with the health care system,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Evans ME et al. Am J Obstet Gynecol. 2018 Oct;219(4):383.e1-7.
HIV testing rates were low among women whose sexual behaviors increased their risk of HIV infection, and they were especially low among women who reported having anal sex, according to a report published in the American Journal of Obstetrics & Gynecology.
Data from the 2011-2015 National Survey of Family Growth were analyzed to estimate the proportion of sexually active, nonpregnant U.S. women aged 15-44 years who had had an HIV test within the past year. The data was stratified by those who reported anal sex and other risk factors, including having more than two sexual partners, condomless sex with a new partner or multiple partners, gonorrhea in the past year, or any history of syphilis, according to Mary Evans, MD, of the National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention at the Centers for Disease Control and Prevention and her colleagues.
Among the 42.4 million sexually active women assessed, 9.0 million (20%) reported they had had anal sex in the past year. Of these, 19% reported that their providers asked about their types of intercourse, and 20% reported an HIV test within the past year. Overall, HIV testing was higher among women who reported anal sex and whose providers asked about types of sex engaged in than it was among those women whose provider did not ask (38% vs. 16%, respectively; P less than .001). However, HIV testing in the past year was higher for women with other forms of risky behaviors as compared with anal sex, ranging from 35.8% to 47.2%.
“Women who report sexual behaviors such as anal sex would benefit from an HIV test and an assessment for [prevention with preexposure prophylaxis] eligibility. Women’s health care providers are uniquely poised to provide HIV prevention for women who tend to have frequent encounters with the health care system,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Evans ME et al. Am J Obstet Gynecol. 2018 Oct;219(4):383.e1-7.
HIV testing rates were low among women whose sexual behaviors increased their risk of HIV infection, and they were especially low among women who reported having anal sex, according to a report published in the American Journal of Obstetrics & Gynecology.
Data from the 2011-2015 National Survey of Family Growth were analyzed to estimate the proportion of sexually active, nonpregnant U.S. women aged 15-44 years who had had an HIV test within the past year. The data was stratified by those who reported anal sex and other risk factors, including having more than two sexual partners, condomless sex with a new partner or multiple partners, gonorrhea in the past year, or any history of syphilis, according to Mary Evans, MD, of the National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention at the Centers for Disease Control and Prevention and her colleagues.
Among the 42.4 million sexually active women assessed, 9.0 million (20%) reported they had had anal sex in the past year. Of these, 19% reported that their providers asked about their types of intercourse, and 20% reported an HIV test within the past year. Overall, HIV testing was higher among women who reported anal sex and whose providers asked about types of sex engaged in than it was among those women whose provider did not ask (38% vs. 16%, respectively; P less than .001). However, HIV testing in the past year was higher for women with other forms of risky behaviors as compared with anal sex, ranging from 35.8% to 47.2%.
“Women who report sexual behaviors such as anal sex would benefit from an HIV test and an assessment for [prevention with preexposure prophylaxis] eligibility. Women’s health care providers are uniquely poised to provide HIV prevention for women who tend to have frequent encounters with the health care system,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Evans ME et al. Am J Obstet Gynecol. 2018 Oct;219(4):383.e1-7.
FROM THE AMERICAN JOURNAL OF OBSTETRICS & GYNECOLOGY
Key clinical point: Health care providers don’t ask sexually active women about risky behavior that would raise their risk of HIV infection.
Major finding: Of women who reported having anal sex, 19% reported that their providers asked about their types of intercourse.
Study details: Data from the 2011-2015 National Survey of Family Growth.
Disclosures: The authors reported that they had no conflicts of interest.
Source: Evans ME et al. Am J Obstet Gynecol. 2018 Oct;219(4):383.e1-7.
Bipolar patients’ relatives face increased cardiovascular risk
BARCELONA – Young patients recently diagnosed with bipolar disorder are at double the 30-year risk of cardiovascular disease, compared with the general population, and their unaffected first-degree relatives are nearly as high risk, Klara Coello, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
The clinical implication of this finding is that unaffected first-degree relatives of patients with bipolar disorder – an affective disorder typically diagnosed at age 15-24 – should be targeted for intensified primary cardiovascular prevention, with a focus on smoking and dyslipidemia, both of which were more prevalent in these patients and their unaffected relatives than in the general population in her study, noted Dr. Coello, a doctoral candidate with the Copenhagen Affective Disorders Research Center at the University of Copenhagen.
She and her coinvestigators presented a cross-sectional study in which they calculated the 30-year Framingham Risk Scores for 221 patients recently diagnosed bipolar disorder – 95% of whom had been diagnosed within the past 2 years – along with 50 unaffected first-degree relatives and 119 age- and sex-matched controls. The investigators used the Framingham Risk Score because the widely used American Heart Association/American College of Cardiology Atherosclerotic Cardiovascular Disease Risk Estimator applies only to individuals aged 40 and up.
The key findings: The 30-year risk of cardiovascular disease for patients with bipolar was 98.5% greater than that of controls, and the calculated risk of the unaffected first-degree relatives was increased by 85.4%, compared with that of controls.
The Framingham Risk Score is determined on the basis of old-school cardiovascular risk factors, including age, gender, lipids, systolic blood pressure, diabetes, and smoking. 45% of the bipolar patients were smokers, as were 20% of their first-degree relatives and 13% of controls.
The Danish finding of increased cardiovascular risk in young adults with bipolar disorder recapitulates an American Heart Association Scientific Statement, which was published in Circulation (2015 Sep 8;132[10]:965-86). The statement was intended to alert clinicians that these affective disorders constitute “moderate-risk” conditions for arterial dysfunction prior to age 30 and for premature cardiovascular disease (CVD). The statement declared that this risk is likely mediated not only by the classic cardiovascular risk factors but also by disease-related inflammation, oxidative stress, sleep disruption, and the adverse metabolic effects of many psychotropic medications.
“The magnitude of increased risk for CVD in adulthood is substantial,” according to the AHA expert panel’s scientific statement.
Dr. Coello’s study only took into account levels of the traditional cardiovascular risk factors. Where the study broke new ground that hadn’t been explored in the AHA scientific statement, however, was in identifying unaffected first-degree relatives as an additional at-risk group.
She reported having no financial conflicts regarding her study, which constitutes her PhD thesis.
BARCELONA – Young patients recently diagnosed with bipolar disorder are at double the 30-year risk of cardiovascular disease, compared with the general population, and their unaffected first-degree relatives are nearly as high risk, Klara Coello, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
The clinical implication of this finding is that unaffected first-degree relatives of patients with bipolar disorder – an affective disorder typically diagnosed at age 15-24 – should be targeted for intensified primary cardiovascular prevention, with a focus on smoking and dyslipidemia, both of which were more prevalent in these patients and their unaffected relatives than in the general population in her study, noted Dr. Coello, a doctoral candidate with the Copenhagen Affective Disorders Research Center at the University of Copenhagen.
She and her coinvestigators presented a cross-sectional study in which they calculated the 30-year Framingham Risk Scores for 221 patients recently diagnosed bipolar disorder – 95% of whom had been diagnosed within the past 2 years – along with 50 unaffected first-degree relatives and 119 age- and sex-matched controls. The investigators used the Framingham Risk Score because the widely used American Heart Association/American College of Cardiology Atherosclerotic Cardiovascular Disease Risk Estimator applies only to individuals aged 40 and up.
The key findings: The 30-year risk of cardiovascular disease for patients with bipolar was 98.5% greater than that of controls, and the calculated risk of the unaffected first-degree relatives was increased by 85.4%, compared with that of controls.
The Framingham Risk Score is determined on the basis of old-school cardiovascular risk factors, including age, gender, lipids, systolic blood pressure, diabetes, and smoking. 45% of the bipolar patients were smokers, as were 20% of their first-degree relatives and 13% of controls.
The Danish finding of increased cardiovascular risk in young adults with bipolar disorder recapitulates an American Heart Association Scientific Statement, which was published in Circulation (2015 Sep 8;132[10]:965-86). The statement was intended to alert clinicians that these affective disorders constitute “moderate-risk” conditions for arterial dysfunction prior to age 30 and for premature cardiovascular disease (CVD). The statement declared that this risk is likely mediated not only by the classic cardiovascular risk factors but also by disease-related inflammation, oxidative stress, sleep disruption, and the adverse metabolic effects of many psychotropic medications.
“The magnitude of increased risk for CVD in adulthood is substantial,” according to the AHA expert panel’s scientific statement.
Dr. Coello’s study only took into account levels of the traditional cardiovascular risk factors. Where the study broke new ground that hadn’t been explored in the AHA scientific statement, however, was in identifying unaffected first-degree relatives as an additional at-risk group.
She reported having no financial conflicts regarding her study, which constitutes her PhD thesis.
BARCELONA – Young patients recently diagnosed with bipolar disorder are at double the 30-year risk of cardiovascular disease, compared with the general population, and their unaffected first-degree relatives are nearly as high risk, Klara Coello, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
The clinical implication of this finding is that unaffected first-degree relatives of patients with bipolar disorder – an affective disorder typically diagnosed at age 15-24 – should be targeted for intensified primary cardiovascular prevention, with a focus on smoking and dyslipidemia, both of which were more prevalent in these patients and their unaffected relatives than in the general population in her study, noted Dr. Coello, a doctoral candidate with the Copenhagen Affective Disorders Research Center at the University of Copenhagen.
She and her coinvestigators presented a cross-sectional study in which they calculated the 30-year Framingham Risk Scores for 221 patients recently diagnosed bipolar disorder – 95% of whom had been diagnosed within the past 2 years – along with 50 unaffected first-degree relatives and 119 age- and sex-matched controls. The investigators used the Framingham Risk Score because the widely used American Heart Association/American College of Cardiology Atherosclerotic Cardiovascular Disease Risk Estimator applies only to individuals aged 40 and up.
The key findings: The 30-year risk of cardiovascular disease for patients with bipolar was 98.5% greater than that of controls, and the calculated risk of the unaffected first-degree relatives was increased by 85.4%, compared with that of controls.
The Framingham Risk Score is determined on the basis of old-school cardiovascular risk factors, including age, gender, lipids, systolic blood pressure, diabetes, and smoking. 45% of the bipolar patients were smokers, as were 20% of their first-degree relatives and 13% of controls.
The Danish finding of increased cardiovascular risk in young adults with bipolar disorder recapitulates an American Heart Association Scientific Statement, which was published in Circulation (2015 Sep 8;132[10]:965-86). The statement was intended to alert clinicians that these affective disorders constitute “moderate-risk” conditions for arterial dysfunction prior to age 30 and for premature cardiovascular disease (CVD). The statement declared that this risk is likely mediated not only by the classic cardiovascular risk factors but also by disease-related inflammation, oxidative stress, sleep disruption, and the adverse metabolic effects of many psychotropic medications.
“The magnitude of increased risk for CVD in adulthood is substantial,” according to the AHA expert panel’s scientific statement.
Dr. Coello’s study only took into account levels of the traditional cardiovascular risk factors. Where the study broke new ground that hadn’t been explored in the AHA scientific statement, however, was in identifying unaffected first-degree relatives as an additional at-risk group.
She reported having no financial conflicts regarding her study, which constitutes her PhD thesis.
REPORTING FROM THE ECNP CONGRESS
Key clinical point: The first-degree relatives of patients with bipolar disorder should be targeted for intensified primary cardiovascular prevention.
Major finding: Thirty-year cardiovascular risk was increased by 98.5% in recently diagnosed bipolar patients and by 85.4% in their unaffected first-degree relatives, compared with the general population.
Study details: This cross-sectional study involved calculation of 30-year Framingham Risk Scores for 221 patients recently diagnosed with bipolar disorder, 50 unaffected first-degree relatives, and 119 age- and sex-matched controls.
Disclosures: The study presenter reported having no financial conflicts of interest.
Don’t let the mortgage preapproval process give you a stomachache
You are trying to buy your first home. Maybe you have heard stories from family, friends, and colleagues about nightmare scenarios when purchasing a home. There are many facets to the home-financing process, and a little bit of planning can reduce a significant amount of time and stress. Where do you begin? What do lenders look for when preapproving a borrower? What steps do I take to get preapproved for a mortgage loan? This article will help guide you through these initial stages to ultimately guide you to settlement on your new home.
Where to begin?
- Start by drafting a budget. How much of a monthly housing payment can you afford? Planning a budget is an extremely valuable exercise at any point in life, not just when buying a home. Often, borrowers will ask the question “How much can I afford?” The better question to ask is “Can I qualify for a home that meets the maximum monthly payment I have budgeted for?”
- What funds would I use for purchasing a home? Down payments and closing costs can add up quickly. Do you have funds readily available in an account you hold? Will you be obtaining a gift from a family member? Generally, funds for down payment are not allowed to be borrowed, unless the money is coming from an account secured by your own assets (for instance, borrowing from your own retirement account). Don’t think you necessarily need to put 20% down. Some loan programs offer little or no down payment options, while other programs may offer down payment assistance options.
- If you are not aware of your credit standing, run a free credit report to verify accurate information. Federal law allows consumers to access one free credit report annually with each of the three credit bureaus (Equifax, Experian, TransUnion). Knowing your credit history and data on your credit report is very important. If there are known or unknown issues on your credit report, it’s always best to at least be informed. You can access your free report at www.annualcreditreport.com.
- Start planning ahead with some of the documentation you will need for a loan approval. Lenders will request items such as tax returns and W-2s from the past 2 years, your recent pay stubs covering a 30-day period, most recent 2 months asset account statements (bank accounts, investment accounts, retirement accounts, etc.), as well as other documentation, depending on your specific scenario.
What are lenders looking at when preapproving an applicant?
Many people will often start to search for homes without having prepared for the preapproval process. This is not necessarily an issue and it doesn’t mean you will not be preapproved. Planning ahead could help you avoid any unforeseen problems and avoid rushing into the mortgage application process when trying to place an offer on a home.
In addition to supplying information on residence and employment/student history for the past 2 years, there are three primary components to a borrower’s credit portfolio:
1) Debt-to-income ratio: What monthly expenses will show on your credit report (car loans/leases, student loans, credit card payments, personal loans/lines of credit, and mortgages for other properties owned)? Do you own any other real estate? Do you have other required obligations, such as alimony or child support payments? To calculate, first combine these liabilities on a monthly expense basis along with the new proposed monthly housing payment. Take these monthly liabilities and divide by monthly income. Gross income (pretax) for employees of a company they do not own is typically utilized (bonus or commission income can have some alternate rules to be allowed as qualifying income); for self-employed borrowers, tax returns will be required to be reviewed; tax write offs could reduce qualifying income. Self-employed individuals will typically need to show a 2-year income history via personal tax returns (as well as business tax returns if applicable). See Figure 1 for an example of a debt-to-income ratio calculation. Many loan programs will require a debt-to-income ratio of 45% or less. There are various loan programs that will be more or less restrictive than this percentage. A lender will be able to guide you to the proper program for your scenario.
2) Liquid assets: Lenders will review the amount of liquid funds you have available for down payment, closing costs, and any necessary reserves. These may include, but are not limited to, checking/savings/money market accounts, investment accounts (stocks, bonds, mutual funds), and retirement funds. Are there enough allowable funds available for the down payment and closing costs, as well as any required reserves needed for qualification? Large non–payroll deposits can be required to be sourced to make sure the funds are from an allowable source.
3) Credit history/scores: Buying a home will be one of the largest purchases you will make in a lifetime. Credit scores have a major impact on the cost of credit (the interest rate you will obtain). Having higher scores could result in a lower interest rate, as well as open up certain loan programs that may be more advantageous for you. Oftentimes, lenders will take the middle of the three scores as your mortgage score (one score from each of the three credit bureaus). In most cases, if applying jointly, the lowest of the middle scores for all borrowers is the score that is used as the score for the applicants. In general, a 740 middle credit score is considered to be excellent for mortgage financing but is not a requirement for all programs.
**You may have heard about specific mortgage programs for physicians. These programs are intended for use for lesser down payments, and/or not calculating student loan payments when qualifying for home financing. As future income potential is typically not considered when determining debt-to-income ratios, not counting these liabilities potentially increases borrowing power.
You are now ready to be preapproved for mortgage financing. What should you do next?
- Talk to a trusted lender. Ask your real estate agent, family, friends, or colleagues for local lender recommendations. Real estate agents will want to make sure you have spoken with a lender and completed a preapproval application to ensure that you can be preapproved for financing before showing you homes. If you need a loan to purchase a home, a preapproval letter will be required to submit with an offer letter. The application contains questions such as your address and employment history for the past 2 years, income and asset information, as well as a series of other financial information. A hard credit inquiry will need to be performed in order for the lender to issue a preapproval. What should you expect from a lender in addition to competitive rates and an array of programs? Some people prefer more of a hands-on approach. Working with a lender who provides regular status updates and makes him/herself easily accessible for all of your questions can certainly be an attractive feature. Working with a local lender also may be reassuring, as he or she should have plenty of experience with the market in which you are purchasing.
- Search for homes. Upon being given the green light for your preapproval and a price range within your comfort zone, connect with your local real estate professional to search for homes. Plan to spend time with your agent discussing all your needs for your new home.
- Submit an offer. Your lender will be able to provide an estimate of closing costs and monthly payments for homes that you are considering buying before you make an offer. You will want to be sure you are comfortable with the financial obligation prior to making your offer. With your offer, an initial good faith deposit (earnest money deposit) will be required. Your real estate agent will guide you on the proper amount of the deposit.
Conclusion
Once you and the seller have come to terms, you will look to discuss with your lender the rate and program options to secure (locking in an interest rate and program), as well as to complete the formal mortgage application. The lender will request additional documentation, if you have not already provided documents, in order for you to obtain a conditional mortgage commitment. The lender also will order an appraisal to ensure the property value supports the price you have agreed to pay for it. Your real estate agent will guide you through the various deadlines and requirements in the contract for items like home inspections, ordering a title search to obtain title insurance, and other nonfinancing contingencies. Some areas may require attorneys for contract review and closing, which your agent will discuss with you. As you can see, buying a home is not an instant process. Taking the appropriate steps to prepare for your mortgage preapproval could save you a lot of time and stress.
Mr. Wishnick is a 15-year mortgage industry veteran, vice president of mortgage lending with Guaranteed Rate (NMLS #2611) and was ranked as a Top 1% mortgage originator by Mortgage Executive Magazine. He can be reached at rob.wishnick@rate.com.
All information provided in this publication is for informational and educational purposes only, and in no way is any of the content contained herein to be construed as financial, investment, or legal advice or instruction. Guaranteed Rate does not guarantee the quality, accuracy, completeness or timelines of the information in this publication.
You are trying to buy your first home. Maybe you have heard stories from family, friends, and colleagues about nightmare scenarios when purchasing a home. There are many facets to the home-financing process, and a little bit of planning can reduce a significant amount of time and stress. Where do you begin? What do lenders look for when preapproving a borrower? What steps do I take to get preapproved for a mortgage loan? This article will help guide you through these initial stages to ultimately guide you to settlement on your new home.
Where to begin?
- Start by drafting a budget. How much of a monthly housing payment can you afford? Planning a budget is an extremely valuable exercise at any point in life, not just when buying a home. Often, borrowers will ask the question “How much can I afford?” The better question to ask is “Can I qualify for a home that meets the maximum monthly payment I have budgeted for?”
- What funds would I use for purchasing a home? Down payments and closing costs can add up quickly. Do you have funds readily available in an account you hold? Will you be obtaining a gift from a family member? Generally, funds for down payment are not allowed to be borrowed, unless the money is coming from an account secured by your own assets (for instance, borrowing from your own retirement account). Don’t think you necessarily need to put 20% down. Some loan programs offer little or no down payment options, while other programs may offer down payment assistance options.
- If you are not aware of your credit standing, run a free credit report to verify accurate information. Federal law allows consumers to access one free credit report annually with each of the three credit bureaus (Equifax, Experian, TransUnion). Knowing your credit history and data on your credit report is very important. If there are known or unknown issues on your credit report, it’s always best to at least be informed. You can access your free report at www.annualcreditreport.com.
- Start planning ahead with some of the documentation you will need for a loan approval. Lenders will request items such as tax returns and W-2s from the past 2 years, your recent pay stubs covering a 30-day period, most recent 2 months asset account statements (bank accounts, investment accounts, retirement accounts, etc.), as well as other documentation, depending on your specific scenario.
What are lenders looking at when preapproving an applicant?
Many people will often start to search for homes without having prepared for the preapproval process. This is not necessarily an issue and it doesn’t mean you will not be preapproved. Planning ahead could help you avoid any unforeseen problems and avoid rushing into the mortgage application process when trying to place an offer on a home.
In addition to supplying information on residence and employment/student history for the past 2 years, there are three primary components to a borrower’s credit portfolio:
1) Debt-to-income ratio: What monthly expenses will show on your credit report (car loans/leases, student loans, credit card payments, personal loans/lines of credit, and mortgages for other properties owned)? Do you own any other real estate? Do you have other required obligations, such as alimony or child support payments? To calculate, first combine these liabilities on a monthly expense basis along with the new proposed monthly housing payment. Take these monthly liabilities and divide by monthly income. Gross income (pretax) for employees of a company they do not own is typically utilized (bonus or commission income can have some alternate rules to be allowed as qualifying income); for self-employed borrowers, tax returns will be required to be reviewed; tax write offs could reduce qualifying income. Self-employed individuals will typically need to show a 2-year income history via personal tax returns (as well as business tax returns if applicable). See Figure 1 for an example of a debt-to-income ratio calculation. Many loan programs will require a debt-to-income ratio of 45% or less. There are various loan programs that will be more or less restrictive than this percentage. A lender will be able to guide you to the proper program for your scenario.
2) Liquid assets: Lenders will review the amount of liquid funds you have available for down payment, closing costs, and any necessary reserves. These may include, but are not limited to, checking/savings/money market accounts, investment accounts (stocks, bonds, mutual funds), and retirement funds. Are there enough allowable funds available for the down payment and closing costs, as well as any required reserves needed for qualification? Large non–payroll deposits can be required to be sourced to make sure the funds are from an allowable source.
3) Credit history/scores: Buying a home will be one of the largest purchases you will make in a lifetime. Credit scores have a major impact on the cost of credit (the interest rate you will obtain). Having higher scores could result in a lower interest rate, as well as open up certain loan programs that may be more advantageous for you. Oftentimes, lenders will take the middle of the three scores as your mortgage score (one score from each of the three credit bureaus). In most cases, if applying jointly, the lowest of the middle scores for all borrowers is the score that is used as the score for the applicants. In general, a 740 middle credit score is considered to be excellent for mortgage financing but is not a requirement for all programs.
**You may have heard about specific mortgage programs for physicians. These programs are intended for use for lesser down payments, and/or not calculating student loan payments when qualifying for home financing. As future income potential is typically not considered when determining debt-to-income ratios, not counting these liabilities potentially increases borrowing power.
You are now ready to be preapproved for mortgage financing. What should you do next?
- Talk to a trusted lender. Ask your real estate agent, family, friends, or colleagues for local lender recommendations. Real estate agents will want to make sure you have spoken with a lender and completed a preapproval application to ensure that you can be preapproved for financing before showing you homes. If you need a loan to purchase a home, a preapproval letter will be required to submit with an offer letter. The application contains questions such as your address and employment history for the past 2 years, income and asset information, as well as a series of other financial information. A hard credit inquiry will need to be performed in order for the lender to issue a preapproval. What should you expect from a lender in addition to competitive rates and an array of programs? Some people prefer more of a hands-on approach. Working with a lender who provides regular status updates and makes him/herself easily accessible for all of your questions can certainly be an attractive feature. Working with a local lender also may be reassuring, as he or she should have plenty of experience with the market in which you are purchasing.
- Search for homes. Upon being given the green light for your preapproval and a price range within your comfort zone, connect with your local real estate professional to search for homes. Plan to spend time with your agent discussing all your needs for your new home.
- Submit an offer. Your lender will be able to provide an estimate of closing costs and monthly payments for homes that you are considering buying before you make an offer. You will want to be sure you are comfortable with the financial obligation prior to making your offer. With your offer, an initial good faith deposit (earnest money deposit) will be required. Your real estate agent will guide you on the proper amount of the deposit.
Conclusion
Once you and the seller have come to terms, you will look to discuss with your lender the rate and program options to secure (locking in an interest rate and program), as well as to complete the formal mortgage application. The lender will request additional documentation, if you have not already provided documents, in order for you to obtain a conditional mortgage commitment. The lender also will order an appraisal to ensure the property value supports the price you have agreed to pay for it. Your real estate agent will guide you through the various deadlines and requirements in the contract for items like home inspections, ordering a title search to obtain title insurance, and other nonfinancing contingencies. Some areas may require attorneys for contract review and closing, which your agent will discuss with you. As you can see, buying a home is not an instant process. Taking the appropriate steps to prepare for your mortgage preapproval could save you a lot of time and stress.
Mr. Wishnick is a 15-year mortgage industry veteran, vice president of mortgage lending with Guaranteed Rate (NMLS #2611) and was ranked as a Top 1% mortgage originator by Mortgage Executive Magazine. He can be reached at rob.wishnick@rate.com.
All information provided in this publication is for informational and educational purposes only, and in no way is any of the content contained herein to be construed as financial, investment, or legal advice or instruction. Guaranteed Rate does not guarantee the quality, accuracy, completeness or timelines of the information in this publication.
You are trying to buy your first home. Maybe you have heard stories from family, friends, and colleagues about nightmare scenarios when purchasing a home. There are many facets to the home-financing process, and a little bit of planning can reduce a significant amount of time and stress. Where do you begin? What do lenders look for when preapproving a borrower? What steps do I take to get preapproved for a mortgage loan? This article will help guide you through these initial stages to ultimately guide you to settlement on your new home.
Where to begin?
- Start by drafting a budget. How much of a monthly housing payment can you afford? Planning a budget is an extremely valuable exercise at any point in life, not just when buying a home. Often, borrowers will ask the question “How much can I afford?” The better question to ask is “Can I qualify for a home that meets the maximum monthly payment I have budgeted for?”
- What funds would I use for purchasing a home? Down payments and closing costs can add up quickly. Do you have funds readily available in an account you hold? Will you be obtaining a gift from a family member? Generally, funds for down payment are not allowed to be borrowed, unless the money is coming from an account secured by your own assets (for instance, borrowing from your own retirement account). Don’t think you necessarily need to put 20% down. Some loan programs offer little or no down payment options, while other programs may offer down payment assistance options.
- If you are not aware of your credit standing, run a free credit report to verify accurate information. Federal law allows consumers to access one free credit report annually with each of the three credit bureaus (Equifax, Experian, TransUnion). Knowing your credit history and data on your credit report is very important. If there are known or unknown issues on your credit report, it’s always best to at least be informed. You can access your free report at www.annualcreditreport.com.
- Start planning ahead with some of the documentation you will need for a loan approval. Lenders will request items such as tax returns and W-2s from the past 2 years, your recent pay stubs covering a 30-day period, most recent 2 months asset account statements (bank accounts, investment accounts, retirement accounts, etc.), as well as other documentation, depending on your specific scenario.
What are lenders looking at when preapproving an applicant?
Many people will often start to search for homes without having prepared for the preapproval process. This is not necessarily an issue and it doesn’t mean you will not be preapproved. Planning ahead could help you avoid any unforeseen problems and avoid rushing into the mortgage application process when trying to place an offer on a home.
In addition to supplying information on residence and employment/student history for the past 2 years, there are three primary components to a borrower’s credit portfolio:
1) Debt-to-income ratio: What monthly expenses will show on your credit report (car loans/leases, student loans, credit card payments, personal loans/lines of credit, and mortgages for other properties owned)? Do you own any other real estate? Do you have other required obligations, such as alimony or child support payments? To calculate, first combine these liabilities on a monthly expense basis along with the new proposed monthly housing payment. Take these monthly liabilities and divide by monthly income. Gross income (pretax) for employees of a company they do not own is typically utilized (bonus or commission income can have some alternate rules to be allowed as qualifying income); for self-employed borrowers, tax returns will be required to be reviewed; tax write offs could reduce qualifying income. Self-employed individuals will typically need to show a 2-year income history via personal tax returns (as well as business tax returns if applicable). See Figure 1 for an example of a debt-to-income ratio calculation. Many loan programs will require a debt-to-income ratio of 45% or less. There are various loan programs that will be more or less restrictive than this percentage. A lender will be able to guide you to the proper program for your scenario.
2) Liquid assets: Lenders will review the amount of liquid funds you have available for down payment, closing costs, and any necessary reserves. These may include, but are not limited to, checking/savings/money market accounts, investment accounts (stocks, bonds, mutual funds), and retirement funds. Are there enough allowable funds available for the down payment and closing costs, as well as any required reserves needed for qualification? Large non–payroll deposits can be required to be sourced to make sure the funds are from an allowable source.
3) Credit history/scores: Buying a home will be one of the largest purchases you will make in a lifetime. Credit scores have a major impact on the cost of credit (the interest rate you will obtain). Having higher scores could result in a lower interest rate, as well as open up certain loan programs that may be more advantageous for you. Oftentimes, lenders will take the middle of the three scores as your mortgage score (one score from each of the three credit bureaus). In most cases, if applying jointly, the lowest of the middle scores for all borrowers is the score that is used as the score for the applicants. In general, a 740 middle credit score is considered to be excellent for mortgage financing but is not a requirement for all programs.
**You may have heard about specific mortgage programs for physicians. These programs are intended for use for lesser down payments, and/or not calculating student loan payments when qualifying for home financing. As future income potential is typically not considered when determining debt-to-income ratios, not counting these liabilities potentially increases borrowing power.
You are now ready to be preapproved for mortgage financing. What should you do next?
- Talk to a trusted lender. Ask your real estate agent, family, friends, or colleagues for local lender recommendations. Real estate agents will want to make sure you have spoken with a lender and completed a preapproval application to ensure that you can be preapproved for financing before showing you homes. If you need a loan to purchase a home, a preapproval letter will be required to submit with an offer letter. The application contains questions such as your address and employment history for the past 2 years, income and asset information, as well as a series of other financial information. A hard credit inquiry will need to be performed in order for the lender to issue a preapproval. What should you expect from a lender in addition to competitive rates and an array of programs? Some people prefer more of a hands-on approach. Working with a lender who provides regular status updates and makes him/herself easily accessible for all of your questions can certainly be an attractive feature. Working with a local lender also may be reassuring, as he or she should have plenty of experience with the market in which you are purchasing.
- Search for homes. Upon being given the green light for your preapproval and a price range within your comfort zone, connect with your local real estate professional to search for homes. Plan to spend time with your agent discussing all your needs for your new home.
- Submit an offer. Your lender will be able to provide an estimate of closing costs and monthly payments for homes that you are considering buying before you make an offer. You will want to be sure you are comfortable with the financial obligation prior to making your offer. With your offer, an initial good faith deposit (earnest money deposit) will be required. Your real estate agent will guide you on the proper amount of the deposit.
Conclusion
Once you and the seller have come to terms, you will look to discuss with your lender the rate and program options to secure (locking in an interest rate and program), as well as to complete the formal mortgage application. The lender will request additional documentation, if you have not already provided documents, in order for you to obtain a conditional mortgage commitment. The lender also will order an appraisal to ensure the property value supports the price you have agreed to pay for it. Your real estate agent will guide you through the various deadlines and requirements in the contract for items like home inspections, ordering a title search to obtain title insurance, and other nonfinancing contingencies. Some areas may require attorneys for contract review and closing, which your agent will discuss with you. As you can see, buying a home is not an instant process. Taking the appropriate steps to prepare for your mortgage preapproval could save you a lot of time and stress.
Mr. Wishnick is a 15-year mortgage industry veteran, vice president of mortgage lending with Guaranteed Rate (NMLS #2611) and was ranked as a Top 1% mortgage originator by Mortgage Executive Magazine. He can be reached at rob.wishnick@rate.com.
All information provided in this publication is for informational and educational purposes only, and in no way is any of the content contained herein to be construed as financial, investment, or legal advice or instruction. Guaranteed Rate does not guarantee the quality, accuracy, completeness or timelines of the information in this publication.