User login
Neurology Reviews covers innovative and emerging news in neurology and neuroscience every month, with a focus on practical approaches to treating Parkinson's disease, epilepsy, headache, stroke, multiple sclerosis, Alzheimer's disease, and other neurologic disorders.
PML
Progressive multifocal leukoencephalopathy
Rituxan
The leading independent newspaper covering neurology news and commentary.
Mississippi–Ohio River valley linked to higher risk of Parkinson’s disease
according to findings from a study that was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
The association was attributed to concentrations of particulate matter (PM) 2.5 in the Mississippi–Ohio River valley, which was on average higher than in other areas, but that didn’t entirely explain the increase in Parkinson’s disease in that region, Brittany Krzyzanowski, PhD, a postdoctoral research fellow in the neuroepidemiology research program of the department of neurology at Barrow Neurological Institute, Dignity Health St. Joseph’s Hospital and Medical Center, Phoenix, said in an interview.
“This study revealed Parkinson’s disease hot spots in the Mississippi–Ohio River valley, a region that has some of the highest levels of air pollution in the nation,” she said, “but we also still find a relationship between air pollution and Parkinson’s risk in the regions in the western half of the United States where Parkinson’s disease and air pollution levels are relatively low.”
Dr. Krzyzanowski and colleagues evaluated 22,546,965 Medicare beneficiaries in 2009, using a multimethod approach that included geospatial analytical techniques to categorize their exposure to PM2.5 based on age, sex, race, smoking status, and health care usage. The researchers also performed individual-level case-control analysis to assess PM2.5 results at the county level. The Medicare beneficiaries were grouped according to average exposure, with the lowest group having an average annual exposure of 5 mcg/m3 and the group with the highest exposure having an average annual exposure of 19 mcg/m3.
In total, researchers identified 83,674 Medicare beneficiaries with incident Parkinson’s disease, with 434 new cases per 100,000 people in the highest exposure group, compared with 359 new cases per 100,000 people in the lowest-exposure group. The relative risk for Parkinson’s disease increased in the highest quartile of PM2.5 by 25%, compared with the lowest quartile after adjusting for factors such as age, smoking status, and health care usage (95% confidence interval, 20%–29%).
The results showed the nationwide average annual PM2.5 was associated with incident Parkinson’s disease, and the Rocky Mountain region carried a strong association between PM2.5 and Parkson’s disease with a 16% increase in risk per level of exposure to PM2.5. While the Mississippi-Ohio River valley was also associated with Parkinson’s disease, there was a weaker association between PM2.5 and Parkinson’s disease, which the researchers attributed to a “ceiling effect” of PM2.5 between approximately 12-19 mcg/m3.
Dr. Krzyzanowski said that use of a large-population-based dataset and high-resolution location data were major strengths of the study. “Having this level of information leaves less room for uncertainty in our measures and analyses,” she said. “Our study also leveraged innovative geographic information systems which allowed us to refine local patterns of disease by using population behavior and demographic information (such as smoking and age) to ensure that we could provide the most accurate map representation available to date.”
A focus on air pollution
Existing research in examining the etiology of Parkinson’s mainly focused on exposure to pesticides,* Dr. Krzyzanowski explained, and “consists of studies using relatively small populations and low-resolution air pollution data.” Genetics is another possible cause, she noted, but only explains some Parkinson’s disease cases.
“Our work suggests that we should also be looking at air pollution as a contributor in the development of Parkinson’s disease,” she said.
Ray Dorsey, MD, professor of neurology at the University of Rochester (N.Y.), who was not involved with the study, said that evidence is mounting that “air pollution may be an important causal factor in Parkinson’s and especially Alzheimer’s disease.”
“This study by a well-regarded group of researchers adds epidemiological evidence for that association,” he said. Another strength is that the study was conducted in the United States, as many epidemiological studies evaluating air pollution and Parkinson’s disease have been performed outside the country because of “a dearth of reliable data sources.”
“This study, along with others, suggest that some of the important environmental toxicants tied to brain disease may be inhaled,” Dr. Dorsey said. “The nose may be the front door to the brain.”
Dr. Krzyzanowski said the next step in their research is further examination of different types of air pollution. “Air pollution contains a variety of toxic components which vary from region to region. Understanding the different components in air pollution and how they interact with climate, temperature, and topography could help explain the regional differences we observed.”
One potential limitation in the study is a lag between air pollution exposure and development of Parkinson’s disease, Dr. Dorsey noted.
“Here, it looks like (but I am not certain) that the investigators looked at current air pollution levels and new cases of Parkinson’s. Ideally, for incident cases of Parkinson’s disease, we would want to know historical data on exposure to air pollution,” he said.
Future studies should include prospective evaluation of adults as well as babies and children who have been exposed to both high and low levels of air pollution. That kind of study “would be incredibly valuable for determining the role of an important environmental toxicant in many brain diseases, including stroke, Alzheimer’s, and Parkinson’s,” he said.
Dr. Krzyzanowski and Dr. Dorsey reported no relevant financial disclosures. This study was supported by grants from the Department of Defense, the National Institute of Environmental Health Sciences, and The Michael J. Fox Foundation for Parkinson’s Research.
*Correction, 4/14/23: An earlier version of this article mischaracterized the disease that was the subject of this research.
according to findings from a study that was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
The association was attributed to concentrations of particulate matter (PM) 2.5 in the Mississippi–Ohio River valley, which was on average higher than in other areas, but that didn’t entirely explain the increase in Parkinson’s disease in that region, Brittany Krzyzanowski, PhD, a postdoctoral research fellow in the neuroepidemiology research program of the department of neurology at Barrow Neurological Institute, Dignity Health St. Joseph’s Hospital and Medical Center, Phoenix, said in an interview.
“This study revealed Parkinson’s disease hot spots in the Mississippi–Ohio River valley, a region that has some of the highest levels of air pollution in the nation,” she said, “but we also still find a relationship between air pollution and Parkinson’s risk in the regions in the western half of the United States where Parkinson’s disease and air pollution levels are relatively low.”
Dr. Krzyzanowski and colleagues evaluated 22,546,965 Medicare beneficiaries in 2009, using a multimethod approach that included geospatial analytical techniques to categorize their exposure to PM2.5 based on age, sex, race, smoking status, and health care usage. The researchers also performed individual-level case-control analysis to assess PM2.5 results at the county level. The Medicare beneficiaries were grouped according to average exposure, with the lowest group having an average annual exposure of 5 mcg/m3 and the group with the highest exposure having an average annual exposure of 19 mcg/m3.
In total, researchers identified 83,674 Medicare beneficiaries with incident Parkinson’s disease, with 434 new cases per 100,000 people in the highest exposure group, compared with 359 new cases per 100,000 people in the lowest-exposure group. The relative risk for Parkinson’s disease increased in the highest quartile of PM2.5 by 25%, compared with the lowest quartile after adjusting for factors such as age, smoking status, and health care usage (95% confidence interval, 20%–29%).
The results showed the nationwide average annual PM2.5 was associated with incident Parkinson’s disease, and the Rocky Mountain region carried a strong association between PM2.5 and Parkson’s disease with a 16% increase in risk per level of exposure to PM2.5. While the Mississippi-Ohio River valley was also associated with Parkinson’s disease, there was a weaker association between PM2.5 and Parkinson’s disease, which the researchers attributed to a “ceiling effect” of PM2.5 between approximately 12-19 mcg/m3.
Dr. Krzyzanowski said that use of a large-population-based dataset and high-resolution location data were major strengths of the study. “Having this level of information leaves less room for uncertainty in our measures and analyses,” she said. “Our study also leveraged innovative geographic information systems which allowed us to refine local patterns of disease by using population behavior and demographic information (such as smoking and age) to ensure that we could provide the most accurate map representation available to date.”
A focus on air pollution
Existing research in examining the etiology of Parkinson’s mainly focused on exposure to pesticides,* Dr. Krzyzanowski explained, and “consists of studies using relatively small populations and low-resolution air pollution data.” Genetics is another possible cause, she noted, but only explains some Parkinson’s disease cases.
“Our work suggests that we should also be looking at air pollution as a contributor in the development of Parkinson’s disease,” she said.
Ray Dorsey, MD, professor of neurology at the University of Rochester (N.Y.), who was not involved with the study, said that evidence is mounting that “air pollution may be an important causal factor in Parkinson’s and especially Alzheimer’s disease.”
“This study by a well-regarded group of researchers adds epidemiological evidence for that association,” he said. Another strength is that the study was conducted in the United States, as many epidemiological studies evaluating air pollution and Parkinson’s disease have been performed outside the country because of “a dearth of reliable data sources.”
“This study, along with others, suggest that some of the important environmental toxicants tied to brain disease may be inhaled,” Dr. Dorsey said. “The nose may be the front door to the brain.”
Dr. Krzyzanowski said the next step in their research is further examination of different types of air pollution. “Air pollution contains a variety of toxic components which vary from region to region. Understanding the different components in air pollution and how they interact with climate, temperature, and topography could help explain the regional differences we observed.”
One potential limitation in the study is a lag between air pollution exposure and development of Parkinson’s disease, Dr. Dorsey noted.
“Here, it looks like (but I am not certain) that the investigators looked at current air pollution levels and new cases of Parkinson’s. Ideally, for incident cases of Parkinson’s disease, we would want to know historical data on exposure to air pollution,” he said.
Future studies should include prospective evaluation of adults as well as babies and children who have been exposed to both high and low levels of air pollution. That kind of study “would be incredibly valuable for determining the role of an important environmental toxicant in many brain diseases, including stroke, Alzheimer’s, and Parkinson’s,” he said.
Dr. Krzyzanowski and Dr. Dorsey reported no relevant financial disclosures. This study was supported by grants from the Department of Defense, the National Institute of Environmental Health Sciences, and The Michael J. Fox Foundation for Parkinson’s Research.
*Correction, 4/14/23: An earlier version of this article mischaracterized the disease that was the subject of this research.
according to findings from a study that was released ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
The association was attributed to concentrations of particulate matter (PM) 2.5 in the Mississippi–Ohio River valley, which was on average higher than in other areas, but that didn’t entirely explain the increase in Parkinson’s disease in that region, Brittany Krzyzanowski, PhD, a postdoctoral research fellow in the neuroepidemiology research program of the department of neurology at Barrow Neurological Institute, Dignity Health St. Joseph’s Hospital and Medical Center, Phoenix, said in an interview.
“This study revealed Parkinson’s disease hot spots in the Mississippi–Ohio River valley, a region that has some of the highest levels of air pollution in the nation,” she said, “but we also still find a relationship between air pollution and Parkinson’s risk in the regions in the western half of the United States where Parkinson’s disease and air pollution levels are relatively low.”
Dr. Krzyzanowski and colleagues evaluated 22,546,965 Medicare beneficiaries in 2009, using a multimethod approach that included geospatial analytical techniques to categorize their exposure to PM2.5 based on age, sex, race, smoking status, and health care usage. The researchers also performed individual-level case-control analysis to assess PM2.5 results at the county level. The Medicare beneficiaries were grouped according to average exposure, with the lowest group having an average annual exposure of 5 mcg/m3 and the group with the highest exposure having an average annual exposure of 19 mcg/m3.
In total, researchers identified 83,674 Medicare beneficiaries with incident Parkinson’s disease, with 434 new cases per 100,000 people in the highest exposure group, compared with 359 new cases per 100,000 people in the lowest-exposure group. The relative risk for Parkinson’s disease increased in the highest quartile of PM2.5 by 25%, compared with the lowest quartile after adjusting for factors such as age, smoking status, and health care usage (95% confidence interval, 20%–29%).
The results showed the nationwide average annual PM2.5 was associated with incident Parkinson’s disease, and the Rocky Mountain region carried a strong association between PM2.5 and Parkson’s disease with a 16% increase in risk per level of exposure to PM2.5. While the Mississippi-Ohio River valley was also associated with Parkinson’s disease, there was a weaker association between PM2.5 and Parkinson’s disease, which the researchers attributed to a “ceiling effect” of PM2.5 between approximately 12-19 mcg/m3.
Dr. Krzyzanowski said that use of a large-population-based dataset and high-resolution location data were major strengths of the study. “Having this level of information leaves less room for uncertainty in our measures and analyses,” she said. “Our study also leveraged innovative geographic information systems which allowed us to refine local patterns of disease by using population behavior and demographic information (such as smoking and age) to ensure that we could provide the most accurate map representation available to date.”
A focus on air pollution
Existing research in examining the etiology of Parkinson’s mainly focused on exposure to pesticides,* Dr. Krzyzanowski explained, and “consists of studies using relatively small populations and low-resolution air pollution data.” Genetics is another possible cause, she noted, but only explains some Parkinson’s disease cases.
“Our work suggests that we should also be looking at air pollution as a contributor in the development of Parkinson’s disease,” she said.
Ray Dorsey, MD, professor of neurology at the University of Rochester (N.Y.), who was not involved with the study, said that evidence is mounting that “air pollution may be an important causal factor in Parkinson’s and especially Alzheimer’s disease.”
“This study by a well-regarded group of researchers adds epidemiological evidence for that association,” he said. Another strength is that the study was conducted in the United States, as many epidemiological studies evaluating air pollution and Parkinson’s disease have been performed outside the country because of “a dearth of reliable data sources.”
“This study, along with others, suggest that some of the important environmental toxicants tied to brain disease may be inhaled,” Dr. Dorsey said. “The nose may be the front door to the brain.”
Dr. Krzyzanowski said the next step in their research is further examination of different types of air pollution. “Air pollution contains a variety of toxic components which vary from region to region. Understanding the different components in air pollution and how they interact with climate, temperature, and topography could help explain the regional differences we observed.”
One potential limitation in the study is a lag between air pollution exposure and development of Parkinson’s disease, Dr. Dorsey noted.
“Here, it looks like (but I am not certain) that the investigators looked at current air pollution levels and new cases of Parkinson’s. Ideally, for incident cases of Parkinson’s disease, we would want to know historical data on exposure to air pollution,” he said.
Future studies should include prospective evaluation of adults as well as babies and children who have been exposed to both high and low levels of air pollution. That kind of study “would be incredibly valuable for determining the role of an important environmental toxicant in many brain diseases, including stroke, Alzheimer’s, and Parkinson’s,” he said.
Dr. Krzyzanowski and Dr. Dorsey reported no relevant financial disclosures. This study was supported by grants from the Department of Defense, the National Institute of Environmental Health Sciences, and The Michael J. Fox Foundation for Parkinson’s Research.
*Correction, 4/14/23: An earlier version of this article mischaracterized the disease that was the subject of this research.
FROM AAN 2023
Seven ‘simple’ cardiovascular health measures linked to reduced dementia risk in women
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
according to results of a study that was released early, ahead of its scheduled presentation at the annual meeting of the American Academy of Neurology.
Epidemiologist Pamela M. Rist, ScD, assistant professor of medicine at Harvard Medical School and associate epidemiologist at Brigham and Women’s Hospital, both in Boston, and colleagues, used data from 13,720 women whose mean age was 54 when they enrolled in the Harvard-based Women’s Health Study between 1992 and 1995. Subjects in that study were followed up in 2004.
Putting ‘Life’s Simple 7’ to the test
Dr. Rist and colleagues used the Harvard data to discern how well closely women conformed, during the initial study period and at 10-year follow up, to what the American Heart Association describes as “Life’s Simple 7,” a list of behavioral and biometric measures that indicate and predict cardiovascular health. The measures include four modifiable behaviors – not smoking, healthy weight, a healthy diet, and being physically active – along with three biometric measures of blood pressure, cholesterol, and blood sugar (AHA has since added a sleep component).
Researchers assigned women one point for each desirable habit or measure on the list, with subjects’ average Simple 7 score at baseline 4.3, and 4.2 at 10 years’ follow-up.
The investigators then looked at Medicare data for the study subjects from 2011 to 2018 – approximately 20 years after their enrollment in the Women’s Health Study – seeking dementia diagnoses. Some 13% of the study cohort (n = 1,771) had gone on to develop dementia.
Each point on the Simple 7 score at baseline corresponded with a 6% reduction in later dementia risk, Dr. Rist and her colleagues found after adjusting for variables including age and education (odds ratio per one unit change in score, 0.94; 95% CI, 0.90-0.98). This effect was similar for Simple 7 scores measured at 10 years of follow-up (OR, 0.95; 95% CI, 0.91-1.00).
“It can be empowering for people to know that by taking steps such as exercising for a half an hour a day or keeping their blood pressure under control, they can reduce their risk of dementia,” Dr. Rist said in a statement on the findings.
‘A simple take-home message’
Reached for comment, Andrew E. Budson, MD, chief of cognitive-behavioral neurology at the VA Boston Healthcare System, praised Dr. Rist and colleagues’ study as one that “builds on existing knowledge to provide a simple take-home message that empowers women to take control of their dementia risk.”
Each of the seven known risk factors – being active, eating better, maintaining a healthy weight, not smoking, maintaining a healthy blood pressure, controlling cholesterol, and having low blood sugar – “was associated with a 6% reduced risk of dementia,” Dr. Budson continued. “So, women who work to address all seven risk factors can reduce their risk of developing dementia by 42%: a huge amount. Moreover, although this study only looked at women, I am confident that if men follow this same advice they will also be able to reduce their risk of dementia, although we don’t know if the size of the effect will be the same.”
Dr. Rist and colleagues’ study was supported by the National Institutes of Health. None of the study authors reported conflicts of interest. Dr. Budson has reported receiving past compensation as a speaker for Eli Lilly.
FROM AAN 2023
Helping a patient buck the odds
I’m not going to get rich off Mike.
Of course, I’m not going to get rich off anyone, nor do I want to. I’m not here to rip anyone off.
Mike goes back with me, roughly 23 years.
He was born with cerebral palsy and refractory seizures. His birth mother gave him up quickly, and he was adopted by a couple who knew what they were getting into (to me that constitutes sainthood).
Over the years Mike has done his best to buck the odds. He’s tried to stay employed, in spite of his physical limitations, working variously as a janitor, grocery courtesy clerk, and store greeter. He tells me that he can still work and wants to, even with having to rely on public transportation.
By the time he came to me he’d been through several neurologists and even more failed epilepsy drugs. His brain MRI and EEGs showed multifocal seizures from numerous inoperable cortical heterotopias.
I dabbled with a few newer drugs at the time for him, without success. Finally, I reached for the neurological equivalent of unstable dynamite – Felbatol (felbamate).
As it often does, it worked. One of my attendings in training (you, Bob) told me it was the home-run drug. When nothing else worked, it might – but you had to handle it carefully.
Fortunately, after 23 years, that hasn’t happened. Mike’s labs have looked good. His seizures have dropped from several a week to a few per year.
Ten years ago Mike had to change insurance to one I don’t take, and had me forward his records to another neurologist. That office told him they don’t handle Felbatol. As did another. And another.
Mike, understandably, doesn’t want to change meds. This is the only drug that’s given him a decent quality of life, and let him have a job. That’s pretty important to him.
So, I see him for free now, once or twice a year. Sometimes he offers me a token payment of $5-$10, but I turn it down. He needs it more than I do, for bus fair to my office if nothing else.
I’m sure some would be critical of me, saying that I should be more open to new drugs and treatments. I am, believe me. But Mike can’t afford many of them, or the loss of work they’d entail if his seizures worsen. He doesn’t want to take that chance, and I don’t blame him.
Of course, none of us can see everyone for free. In fact, he’s the only one I do. I’m not greedy, but I also have to pay my rent, staff, and mortgage.
But taking money from Mike, who’s come up on the short end of the stick in so many ways, doesn’t seem right. I can’t do it, and really don’t want to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m not going to get rich off Mike.
Of course, I’m not going to get rich off anyone, nor do I want to. I’m not here to rip anyone off.
Mike goes back with me, roughly 23 years.
He was born with cerebral palsy and refractory seizures. His birth mother gave him up quickly, and he was adopted by a couple who knew what they were getting into (to me that constitutes sainthood).
Over the years Mike has done his best to buck the odds. He’s tried to stay employed, in spite of his physical limitations, working variously as a janitor, grocery courtesy clerk, and store greeter. He tells me that he can still work and wants to, even with having to rely on public transportation.
By the time he came to me he’d been through several neurologists and even more failed epilepsy drugs. His brain MRI and EEGs showed multifocal seizures from numerous inoperable cortical heterotopias.
I dabbled with a few newer drugs at the time for him, without success. Finally, I reached for the neurological equivalent of unstable dynamite – Felbatol (felbamate).
As it often does, it worked. One of my attendings in training (you, Bob) told me it was the home-run drug. When nothing else worked, it might – but you had to handle it carefully.
Fortunately, after 23 years, that hasn’t happened. Mike’s labs have looked good. His seizures have dropped from several a week to a few per year.
Ten years ago Mike had to change insurance to one I don’t take, and had me forward his records to another neurologist. That office told him they don’t handle Felbatol. As did another. And another.
Mike, understandably, doesn’t want to change meds. This is the only drug that’s given him a decent quality of life, and let him have a job. That’s pretty important to him.
So, I see him for free now, once or twice a year. Sometimes he offers me a token payment of $5-$10, but I turn it down. He needs it more than I do, for bus fair to my office if nothing else.
I’m sure some would be critical of me, saying that I should be more open to new drugs and treatments. I am, believe me. But Mike can’t afford many of them, or the loss of work they’d entail if his seizures worsen. He doesn’t want to take that chance, and I don’t blame him.
Of course, none of us can see everyone for free. In fact, he’s the only one I do. I’m not greedy, but I also have to pay my rent, staff, and mortgage.
But taking money from Mike, who’s come up on the short end of the stick in so many ways, doesn’t seem right. I can’t do it, and really don’t want to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
I’m not going to get rich off Mike.
Of course, I’m not going to get rich off anyone, nor do I want to. I’m not here to rip anyone off.
Mike goes back with me, roughly 23 years.
He was born with cerebral palsy and refractory seizures. His birth mother gave him up quickly, and he was adopted by a couple who knew what they were getting into (to me that constitutes sainthood).
Over the years Mike has done his best to buck the odds. He’s tried to stay employed, in spite of his physical limitations, working variously as a janitor, grocery courtesy clerk, and store greeter. He tells me that he can still work and wants to, even with having to rely on public transportation.
By the time he came to me he’d been through several neurologists and even more failed epilepsy drugs. His brain MRI and EEGs showed multifocal seizures from numerous inoperable cortical heterotopias.
I dabbled with a few newer drugs at the time for him, without success. Finally, I reached for the neurological equivalent of unstable dynamite – Felbatol (felbamate).
As it often does, it worked. One of my attendings in training (you, Bob) told me it was the home-run drug. When nothing else worked, it might – but you had to handle it carefully.
Fortunately, after 23 years, that hasn’t happened. Mike’s labs have looked good. His seizures have dropped from several a week to a few per year.
Ten years ago Mike had to change insurance to one I don’t take, and had me forward his records to another neurologist. That office told him they don’t handle Felbatol. As did another. And another.
Mike, understandably, doesn’t want to change meds. This is the only drug that’s given him a decent quality of life, and let him have a job. That’s pretty important to him.
So, I see him for free now, once or twice a year. Sometimes he offers me a token payment of $5-$10, but I turn it down. He needs it more than I do, for bus fair to my office if nothing else.
I’m sure some would be critical of me, saying that I should be more open to new drugs and treatments. I am, believe me. But Mike can’t afford many of them, or the loss of work they’d entail if his seizures worsen. He doesn’t want to take that chance, and I don’t blame him.
Of course, none of us can see everyone for free. In fact, he’s the only one I do. I’m not greedy, but I also have to pay my rent, staff, and mortgage.
But taking money from Mike, who’s come up on the short end of the stick in so many ways, doesn’t seem right. I can’t do it, and really don’t want to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Study gives new insight into timing of combo treatment in metastatic NSCLC
However, patients still fared poorly on average since overall survival remained low and didn’t change significantly.
While not conclusive, the new research – released at European Lung Cancer Congress 2023 – offers early insight into the best timing for the experimental combination treatment, study coauthor Yanyan Lou, MD, PhD, an oncologist at Mayo Clinic in Jacksonville, Fla., said in an interview.
The wide availability of radiation therapy could also allow the therapy to be administered even in regions with poor access to sophisticated medical care, she said. “Radiation is a very feasible approach that pretty much everybody in your community can get.”
Radiotherapy is typically not added to immunotherapy in patients with non–small cell lung cancer. But “there has been recent interest in the combination: Would tumor necrosis from radiation enhance the immunogenicity of the tumor and thus enhance the effect of immunotherapy?” oncologist Toby Campbell, MD, of University of Wisconsin–Madison, said in an interview.
Research has indeed suggested that the treatments may have a synergistic effect, he said, and it’s clear that “strategies to try and increase immunogenicity are an important area to investigate.”
But he cautioned that “we have a long way to go to understanding how immunogenicity works and how the gut microbiome, tumor, immunotherapy, and the immune system interact with one another.”
For the new study, researchers retrospectively analyzed cases of 225 patients with metastatic non–small cell lung cancer (male = 56%, median age = 68, 79% adenocarcinoma) who were treated with immunotherapy at Mayo Clinic–Jacksonville from 2011 to 2022. The study excluded those who received targeted therapy or prior concurrent chemoradiotherapy and durvalumab.
The most common metastases were bone and central nervous system types (41% and 25%, respectively). Fifty-six percent of patients received radiotherapy before or during immunotherapy. Another 27% never received radiotherapy, and 17% received it after immunotherapy was discontinued.
Common types of immunotherapy included pembrolizumab (78%), nivolumab (14%), and atezolizumab (12%).
Overall, the researchers found no statistically significant differences in various outcomes between patients who received radiotherapy before or during immunotherapy compared with those who didn’t get radiotherapy (progression-free survival: 5.9 vs. 5.5 months, P = .66; overall survival: 16.9 vs. 13.1 months, P = .84; immune-related adverse events: 26.2% vs. 34.4%, P = .24).
However, the researchers found that progression-free survival was significantly higher in one group: Those who received radiotherapy 1-12 months before immunotherapy vs. those who received it less than 1 month before (12.6 vs. 4.2 months, hazard ratio [HR], 0.46, 95% confidence interval [CI], 0.26-0.83, P = .005,) and those who never received radiotherapy (12.6 vs. 5.5 months, HR, 0.56, 95% CI, 0.36-0.89, P = .0197).
There wasn’t a statistically significant difference in overall survival.
The small number of subjects and the variation in treatment protocols may have prevented the study from revealing a survival benefit, Dr. Lou said.
As for adverse effects, she said a preliminary analysis didn’t turn up any.
It’s not clear why a 1- to 12-month gap between radiotherapy and immunotherapy may be most effective, she said. Moving forward, “we need validate this in a large cohort,” she noted.
In regard to cost, immunotherapy is notoriously expensive. Pembrolizumab, for example, has a list price of $10,897 per 200-mg dose given every 3 weeks, and patients may take the drug for a year or two.
Dr. Campbell, who didn’t take part in the new study, said it suggests that research into radiation-immunotherapy combination treatment may be worthwhile.
No funding was reported. The study authors and Dr. Campbell reported no disclosures.
However, patients still fared poorly on average since overall survival remained low and didn’t change significantly.
While not conclusive, the new research – released at European Lung Cancer Congress 2023 – offers early insight into the best timing for the experimental combination treatment, study coauthor Yanyan Lou, MD, PhD, an oncologist at Mayo Clinic in Jacksonville, Fla., said in an interview.
The wide availability of radiation therapy could also allow the therapy to be administered even in regions with poor access to sophisticated medical care, she said. “Radiation is a very feasible approach that pretty much everybody in your community can get.”
Radiotherapy is typically not added to immunotherapy in patients with non–small cell lung cancer. But “there has been recent interest in the combination: Would tumor necrosis from radiation enhance the immunogenicity of the tumor and thus enhance the effect of immunotherapy?” oncologist Toby Campbell, MD, of University of Wisconsin–Madison, said in an interview.
Research has indeed suggested that the treatments may have a synergistic effect, he said, and it’s clear that “strategies to try and increase immunogenicity are an important area to investigate.”
But he cautioned that “we have a long way to go to understanding how immunogenicity works and how the gut microbiome, tumor, immunotherapy, and the immune system interact with one another.”
For the new study, researchers retrospectively analyzed cases of 225 patients with metastatic non–small cell lung cancer (male = 56%, median age = 68, 79% adenocarcinoma) who were treated with immunotherapy at Mayo Clinic–Jacksonville from 2011 to 2022. The study excluded those who received targeted therapy or prior concurrent chemoradiotherapy and durvalumab.
The most common metastases were bone and central nervous system types (41% and 25%, respectively). Fifty-six percent of patients received radiotherapy before or during immunotherapy. Another 27% never received radiotherapy, and 17% received it after immunotherapy was discontinued.
Common types of immunotherapy included pembrolizumab (78%), nivolumab (14%), and atezolizumab (12%).
Overall, the researchers found no statistically significant differences in various outcomes between patients who received radiotherapy before or during immunotherapy compared with those who didn’t get radiotherapy (progression-free survival: 5.9 vs. 5.5 months, P = .66; overall survival: 16.9 vs. 13.1 months, P = .84; immune-related adverse events: 26.2% vs. 34.4%, P = .24).
However, the researchers found that progression-free survival was significantly higher in one group: Those who received radiotherapy 1-12 months before immunotherapy vs. those who received it less than 1 month before (12.6 vs. 4.2 months, hazard ratio [HR], 0.46, 95% confidence interval [CI], 0.26-0.83, P = .005,) and those who never received radiotherapy (12.6 vs. 5.5 months, HR, 0.56, 95% CI, 0.36-0.89, P = .0197).
There wasn’t a statistically significant difference in overall survival.
The small number of subjects and the variation in treatment protocols may have prevented the study from revealing a survival benefit, Dr. Lou said.
As for adverse effects, she said a preliminary analysis didn’t turn up any.
It’s not clear why a 1- to 12-month gap between radiotherapy and immunotherapy may be most effective, she said. Moving forward, “we need validate this in a large cohort,” she noted.
In regard to cost, immunotherapy is notoriously expensive. Pembrolizumab, for example, has a list price of $10,897 per 200-mg dose given every 3 weeks, and patients may take the drug for a year or two.
Dr. Campbell, who didn’t take part in the new study, said it suggests that research into radiation-immunotherapy combination treatment may be worthwhile.
No funding was reported. The study authors and Dr. Campbell reported no disclosures.
However, patients still fared poorly on average since overall survival remained low and didn’t change significantly.
While not conclusive, the new research – released at European Lung Cancer Congress 2023 – offers early insight into the best timing for the experimental combination treatment, study coauthor Yanyan Lou, MD, PhD, an oncologist at Mayo Clinic in Jacksonville, Fla., said in an interview.
The wide availability of radiation therapy could also allow the therapy to be administered even in regions with poor access to sophisticated medical care, she said. “Radiation is a very feasible approach that pretty much everybody in your community can get.”
Radiotherapy is typically not added to immunotherapy in patients with non–small cell lung cancer. But “there has been recent interest in the combination: Would tumor necrosis from radiation enhance the immunogenicity of the tumor and thus enhance the effect of immunotherapy?” oncologist Toby Campbell, MD, of University of Wisconsin–Madison, said in an interview.
Research has indeed suggested that the treatments may have a synergistic effect, he said, and it’s clear that “strategies to try and increase immunogenicity are an important area to investigate.”
But he cautioned that “we have a long way to go to understanding how immunogenicity works and how the gut microbiome, tumor, immunotherapy, and the immune system interact with one another.”
For the new study, researchers retrospectively analyzed cases of 225 patients with metastatic non–small cell lung cancer (male = 56%, median age = 68, 79% adenocarcinoma) who were treated with immunotherapy at Mayo Clinic–Jacksonville from 2011 to 2022. The study excluded those who received targeted therapy or prior concurrent chemoradiotherapy and durvalumab.
The most common metastases were bone and central nervous system types (41% and 25%, respectively). Fifty-six percent of patients received radiotherapy before or during immunotherapy. Another 27% never received radiotherapy, and 17% received it after immunotherapy was discontinued.
Common types of immunotherapy included pembrolizumab (78%), nivolumab (14%), and atezolizumab (12%).
Overall, the researchers found no statistically significant differences in various outcomes between patients who received radiotherapy before or during immunotherapy compared with those who didn’t get radiotherapy (progression-free survival: 5.9 vs. 5.5 months, P = .66; overall survival: 16.9 vs. 13.1 months, P = .84; immune-related adverse events: 26.2% vs. 34.4%, P = .24).
However, the researchers found that progression-free survival was significantly higher in one group: Those who received radiotherapy 1-12 months before immunotherapy vs. those who received it less than 1 month before (12.6 vs. 4.2 months, hazard ratio [HR], 0.46, 95% confidence interval [CI], 0.26-0.83, P = .005,) and those who never received radiotherapy (12.6 vs. 5.5 months, HR, 0.56, 95% CI, 0.36-0.89, P = .0197).
There wasn’t a statistically significant difference in overall survival.
The small number of subjects and the variation in treatment protocols may have prevented the study from revealing a survival benefit, Dr. Lou said.
As for adverse effects, she said a preliminary analysis didn’t turn up any.
It’s not clear why a 1- to 12-month gap between radiotherapy and immunotherapy may be most effective, she said. Moving forward, “we need validate this in a large cohort,” she noted.
In regard to cost, immunotherapy is notoriously expensive. Pembrolizumab, for example, has a list price of $10,897 per 200-mg dose given every 3 weeks, and patients may take the drug for a year or two.
Dr. Campbell, who didn’t take part in the new study, said it suggests that research into radiation-immunotherapy combination treatment may be worthwhile.
No funding was reported. The study authors and Dr. Campbell reported no disclosures.
FROM ELCC 2023
First target doesn’t affect survival in NSCLC with brain metastases
“The findings of our study highlight the importance of adopting a personalized, case-based approach when treating each patient” instead of always treating the brain or lung first, lead author Arvind Kumar, a medical student at Icahn School of Medicine at Mount Sinai, New York, said in an interview.
The study was released at European Lung Cancer Congress 2023.
According to the author, current guidelines recommend treating the brain first in patients with non–small cell lung cancer and a tumor that has spread to the brain.
“Determining whether the brain or body gets treated first depends on where the symptoms are coming from, how severe the symptoms are, how bulky the disease is, and how long the treatment to each is expected to take,” radiation oncologist Henry S. Park, MD, MPH, chief of the thoracic radiotherapy program at Yale University, New Haven, Conn., said in an interview. “Often the brain is treated first since surgery is used for both diagnosis of metastatic disease as well as removal of the brain metastasis, especially if it is causing symptoms. The radiosurgery that follows tends to occur within a day or a few days.”
However, he said, “if the brain disease is small and not causing symptoms, and the lung disease is more problematic, then we will often treat the body first and fit in the brain treatment later.”
For the new study, researchers identified 1,044 patients in the National Cancer Database with non–small cell lung cancer and brain metastases who received systemic therapy plus surgery, brain stereotactic radiosurgery, or lung radiation. All were treated from 2010 to 2019; 79.0% received brain treatment first, and the other 21.0% received lung treatment first.
There was no statistically significant difference in overall survival between those whose brains were treated first and those whose lungs were treated first (hazard ratio, 1.24, 95% confidence interval [CI], 0.91-1.70, P = .17). A propensity score–matched analysis turned up no difference in 5-year survival (38.2% of those whose brains were treated first, 95% CI, 27.5-34.4, vs. 38.0% of those whose lungs were treated first, 95% CI, 29.9-44.7, P = .32.)
“These results were consistent regardless of which combination of treatment modalities the patient received – neurosurgery versus brain stereotactic radiosurgery, thoracic surgery versus thoracic radiation,” the author said.
He cautioned that “our study only included patients who were considered candidates for either surgery or radiation to both the brain and lung. The results of our study should therefore be cautiously interpreted for patients who may have contraindications to such treatment.”
Dr. Park, who didn’t take part in the study, said “the results are consistent with what I would generally expect.”
He added: “The take-home message for clinicians should be that there is no one correct answer in how to manage non–small cell lung cancer with synchronous limited metastatic disease in only the brain. If the brain disease is bulky and/or causes symptoms while the body disease isn’t – or if a biopsy or surgery is required to prove that the patient in fact has metastatic disease – then the brain disease should be treated first. On the other hand, if the body disease is bulky and/or causing symptoms while the brain disease isn’t – and there is no need for surgery but rather only a biopsy of the brain – then the body disease can be treated first.”
No funding was reported. The study authors and Dr. Park reported no financial conflicts or other disclosures.
“The findings of our study highlight the importance of adopting a personalized, case-based approach when treating each patient” instead of always treating the brain or lung first, lead author Arvind Kumar, a medical student at Icahn School of Medicine at Mount Sinai, New York, said in an interview.
The study was released at European Lung Cancer Congress 2023.
According to the author, current guidelines recommend treating the brain first in patients with non–small cell lung cancer and a tumor that has spread to the brain.
“Determining whether the brain or body gets treated first depends on where the symptoms are coming from, how severe the symptoms are, how bulky the disease is, and how long the treatment to each is expected to take,” radiation oncologist Henry S. Park, MD, MPH, chief of the thoracic radiotherapy program at Yale University, New Haven, Conn., said in an interview. “Often the brain is treated first since surgery is used for both diagnosis of metastatic disease as well as removal of the brain metastasis, especially if it is causing symptoms. The radiosurgery that follows tends to occur within a day or a few days.”
However, he said, “if the brain disease is small and not causing symptoms, and the lung disease is more problematic, then we will often treat the body first and fit in the brain treatment later.”
For the new study, researchers identified 1,044 patients in the National Cancer Database with non–small cell lung cancer and brain metastases who received systemic therapy plus surgery, brain stereotactic radiosurgery, or lung radiation. All were treated from 2010 to 2019; 79.0% received brain treatment first, and the other 21.0% received lung treatment first.
There was no statistically significant difference in overall survival between those whose brains were treated first and those whose lungs were treated first (hazard ratio, 1.24, 95% confidence interval [CI], 0.91-1.70, P = .17). A propensity score–matched analysis turned up no difference in 5-year survival (38.2% of those whose brains were treated first, 95% CI, 27.5-34.4, vs. 38.0% of those whose lungs were treated first, 95% CI, 29.9-44.7, P = .32.)
“These results were consistent regardless of which combination of treatment modalities the patient received – neurosurgery versus brain stereotactic radiosurgery, thoracic surgery versus thoracic radiation,” the author said.
He cautioned that “our study only included patients who were considered candidates for either surgery or radiation to both the brain and lung. The results of our study should therefore be cautiously interpreted for patients who may have contraindications to such treatment.”
Dr. Park, who didn’t take part in the study, said “the results are consistent with what I would generally expect.”
He added: “The take-home message for clinicians should be that there is no one correct answer in how to manage non–small cell lung cancer with synchronous limited metastatic disease in only the brain. If the brain disease is bulky and/or causes symptoms while the body disease isn’t – or if a biopsy or surgery is required to prove that the patient in fact has metastatic disease – then the brain disease should be treated first. On the other hand, if the body disease is bulky and/or causing symptoms while the brain disease isn’t – and there is no need for surgery but rather only a biopsy of the brain – then the body disease can be treated first.”
No funding was reported. The study authors and Dr. Park reported no financial conflicts or other disclosures.
“The findings of our study highlight the importance of adopting a personalized, case-based approach when treating each patient” instead of always treating the brain or lung first, lead author Arvind Kumar, a medical student at Icahn School of Medicine at Mount Sinai, New York, said in an interview.
The study was released at European Lung Cancer Congress 2023.
According to the author, current guidelines recommend treating the brain first in patients with non–small cell lung cancer and a tumor that has spread to the brain.
“Determining whether the brain or body gets treated first depends on where the symptoms are coming from, how severe the symptoms are, how bulky the disease is, and how long the treatment to each is expected to take,” radiation oncologist Henry S. Park, MD, MPH, chief of the thoracic radiotherapy program at Yale University, New Haven, Conn., said in an interview. “Often the brain is treated first since surgery is used for both diagnosis of metastatic disease as well as removal of the brain metastasis, especially if it is causing symptoms. The radiosurgery that follows tends to occur within a day or a few days.”
However, he said, “if the brain disease is small and not causing symptoms, and the lung disease is more problematic, then we will often treat the body first and fit in the brain treatment later.”
For the new study, researchers identified 1,044 patients in the National Cancer Database with non–small cell lung cancer and brain metastases who received systemic therapy plus surgery, brain stereotactic radiosurgery, or lung radiation. All were treated from 2010 to 2019; 79.0% received brain treatment first, and the other 21.0% received lung treatment first.
There was no statistically significant difference in overall survival between those whose brains were treated first and those whose lungs were treated first (hazard ratio, 1.24, 95% confidence interval [CI], 0.91-1.70, P = .17). A propensity score–matched analysis turned up no difference in 5-year survival (38.2% of those whose brains were treated first, 95% CI, 27.5-34.4, vs. 38.0% of those whose lungs were treated first, 95% CI, 29.9-44.7, P = .32.)
“These results were consistent regardless of which combination of treatment modalities the patient received – neurosurgery versus brain stereotactic radiosurgery, thoracic surgery versus thoracic radiation,” the author said.
He cautioned that “our study only included patients who were considered candidates for either surgery or radiation to both the brain and lung. The results of our study should therefore be cautiously interpreted for patients who may have contraindications to such treatment.”
Dr. Park, who didn’t take part in the study, said “the results are consistent with what I would generally expect.”
He added: “The take-home message for clinicians should be that there is no one correct answer in how to manage non–small cell lung cancer with synchronous limited metastatic disease in only the brain. If the brain disease is bulky and/or causes symptoms while the body disease isn’t – or if a biopsy or surgery is required to prove that the patient in fact has metastatic disease – then the brain disease should be treated first. On the other hand, if the body disease is bulky and/or causing symptoms while the brain disease isn’t – and there is no need for surgery but rather only a biopsy of the brain – then the body disease can be treated first.”
No funding was reported. The study authors and Dr. Park reported no financial conflicts or other disclosures.
FROM ELCC 2023
New guidelines for cannabis in chronic pain management released
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New clinical practice guidelines for cannabis in chronic pain management have been released.
Developed by a group of Canadian researchers, clinicians, and patients, the guidelines note that cannabinoid-based medicines (CBM) may help clinicians offer an effective, less addictive, alternative to opioids in patients with chronic noncancer pain and comorbid conditions.
“We don’t recommend using CBM first line for anything pretty much because there are other alternatives that may be more effective and also offer fewer side effects,” lead guideline author Alan Bell, MD, assistant professor of family and community medicine at the University of Toronto, told this news organization.
“But I would strongly argue that I would use cannabis-based medicine over opioids every time. Why would you use a high potency-high toxicity agent when there’s a low potency-low toxicity alternative?” he said.
The guidelines were published online in the journal Cannabis and Cannabinoid Research.
Examining the evidence
A consistent criticism of CBM has been the lack of quality research supporting its therapeutic utility. To develop the current recommendations, the task force reviewed 47 pain management studies enrolling more than 11,000 patients. Almost half of the studies (n = 22) were randomized controlled trials (RCTs) and 12 of the 19 included systematic reviews focused solely on RCTs.
Overall, 38 of the 47 included studies demonstrated that CBM provided at least moderate benefits for chronic pain, resulting in a “strong” recommendation – mostly as an adjunct or replacement treatment in individuals living with chronic pain.
Overall, the guidelines place a high value on improving chronic pain and functionality, and addressing co-occurring conditions such as insomnia, anxiety and depression, mobility, and inflammation. They also provide practical dosing and formulation tips to support the use of CBM in the clinical setting.
When it comes to chronic pain, CBM is not a panacea. However, prior research suggests cannabinoids and opioids share several pharmacologic properties, including independent but possibly related mechanisms for antinociception, making them an intriguing combination.
In the current guidelines, all of the four studies specifically addressing combined opioids and vaporized cannabis flower demonstrated further pain reduction, reinforcing the conclusion that the benefits of CBM for improving pain control in patients taking opioids outweigh the risk of nonserious adverse events (AEs), such as dry mouth, dizziness, increased appetite, sedation, and concentration difficulties.
The recommendations also highlighted evidence demonstrating that a majority of participants were able to reduce use of routine pain medications with concomitant CBM/opioid administration, while simultaneously offering secondary benefits such as improved sleep, anxiety, and mood, as well as prevention of opioid tolerance and dose escalation.
Importantly, the guidelines offer an evidence-based algorithm with a clear framework for tapering patients off opioids, especially those who are on > 50 mg MED, which places them with a twofold greater risk for fatal overdose.
An effective alternative
Commenting on the new guidelines, Mark Wallace, MD, who has extensive experience researching and treating pain patients with medical cannabis, said the genesis of his interest in medical cannabis mirrors the guidelines’ focus.
“What got me interested in medical cannabis was trying to get patients off of opioids,” said Dr. Wallace, professor of anesthesiology and chief of the division of pain medicine in the department of anesthesiology at the University of California, San Diego. Dr. Wallace, who was not involved in the guidelines’ development study, said that he’s “titrated hundreds of patients off of opioids using cannabis.”
Dr. Wallace said he found the guidelines’ dosing recommendations helpful.
“If you stay within the 1- to 5-mg dosing range, the risks are so incredibly low, you’re not going to harm the patient.”
While there are patients who abuse cannabis and CBMs, Dr. Wallace noted that he has seen only one patient in the past 20 years who was overusing the medical cannabis. He added that his patient population does not use medical cannabis to get high and, in fact, wants to avoid doses that produce that effect at all costs.
Also commenting on the guidelines, Christopher Gilligan, MD, MBA, associate chief medical officer and a pain medicine physician at Brigham and Women’s Hospital in Boston, who was not involved in the guidelines’ development, points to the risks.
“When we have an opportunity to use cannabinoids in place of opioids for our patients, I think that that’s a positive thing ... and a wise choice in terms of risk benefit,” Dr. Gilligan said.
On the other hand, he cautioned that “freely prescribing” cannabinoids for chronic pain in patients who aren’t on opioids is not good practice.
“We have to take seriously the potential adverse effects of [cannabis], including marijuana use disorder, interference with learning, memory impairment, and psychotic breakthroughs,” said Dr. Gilligan.
Given the current climate, it would appear that CBM is a long way from being endorsed by the Food and Drug Administration, but for clinicians interested in trying CBM for chronic pain patients, the guidelines may offer a roadmap for initiation and an alternative to prescribing opioids.
Dr. Bell, Dr. Gilligan, and Dr. Wallace report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CANNABIS AND CANNABINOID RESEARCH
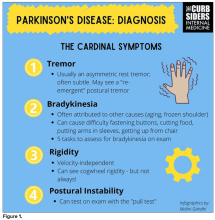
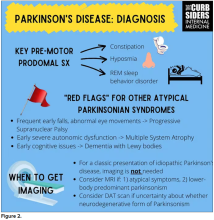
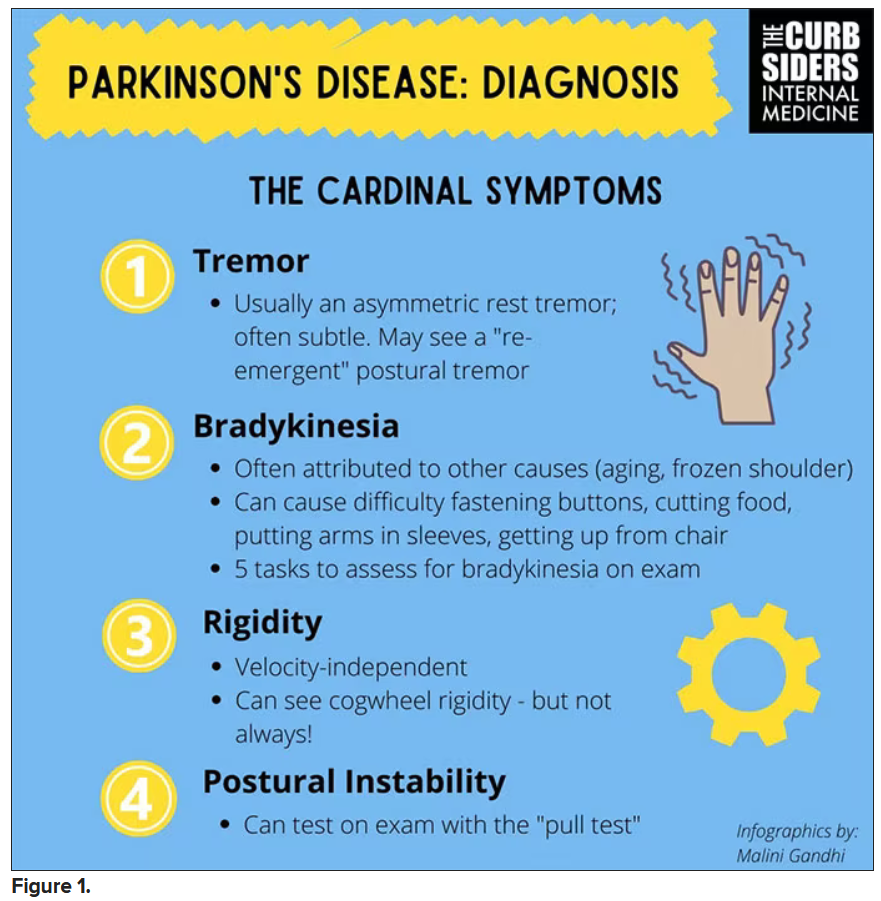
Picking up the premotor symptoms of Parkinson’s
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. We had a great discussion on Parkinson’s Disease for Primary Care with Dr. Albert Hung. Paul, this was something that really made me nervous. I didn’t have a lot of comfort with it. But he taught us a lot of tips about how to recognize Parkinson’s.
I hadn’t been as aware of the premotor symptoms: constipation, hyposmia (loss of sense of smell), and rapid eye movement sleep behavior disorder. If patients have those early on and they aren’t explained by other things (especially the REM sleep behavior disorder), you should really key in because those patients are at risk of developing Parkinson’s years down the line. Those symptoms could present first, which just kind of blew my mind.
What tips do you have about how to recognize Parkinson’s? Do you want to talk about the physical exam?
Paul N. Williams, MD: You know I love the physical exam stuff, so I’m happy to talk about that.
You were deeply upset that cogwheel rigidity was not pathognomonic for Parkinson’s, but you made the point – and our guest agreed – that asymmetry tends to be the key here. And I really appreciated the point about reemergent tremor. This is this idea of a resting tremor. If someone has more parkinsonian features, you might see an intention tremor with essential tremor. If they reach out, it might seem steady at first, but if they hold long enough, then the tremor may kind of reemerge. I thought that was a neat distinction.
And this idea of cogwheel rigidity is a combination of some of the cardinal features of Parkinson’s – it’s a little bit of tremor and a little bit of rigidity too. There’s a baseline increase in tone, and then the tremor is superimposed on top of that. When you’re feeling cogwheeling, that’s actually what you’re feeling on examination. Parkinson’s, with all of its physical exam findings has always fascinated me.
Dr. Watto: He also told us about some red flags.
With classic idiopathic parkinsonism, there’s asymmetric involvement of the tremor. So red flags include a symmetric tremor, which might be something other than idiopathic parkinsonism. He also mentioned that one of the reasons you may want to get imaging (which is not always necessary if someone has a classic presentation), is if you see lower body–predominant symptoms of parkinsonism. These patients have rigidity or slowness of movement in their legs, but their upper bodies are not affected. They don’t have masked facies or the tremor in their hands. You might get an MRI in that case because that could be presentation of vascular dementia or vascular disease in the brain or even normal pressure hydrocephalus, which is a treatable condition. That would be one reason to get imaging.
What if the patient was exposed to a drug like a dopamine antagonist? They will get better in a couple of days, right?
Dr. Williams: This was a really fascinating point because we typically think if a patient’s symptoms are related to a drug exposure – in this case, drug-induced parkinsonism – we can just stop the medication and the symptoms will disappear in a couple of days as the drug leaves the system. But as it turns out, it might take much longer. A mistake that Dr Hung often sees is that the clinician stops the possibly offending agent, but when they don’t see an immediate relief of symptoms, they assume the drug wasn’t causing them. You really have to give the patient a fair shot off the medication to experience recovery because those symptoms can last weeks or even months after the drug is discontinued.
Dr. Watto: Dr Hung looks at the patient’s problem list and asks whether is there any reason this patient might have been exposed to one of these medications?
We’re not going to get too much into specific Parkinson’s treatment, but I was glad to hear that exercise actually improves mobility and may even have some neuroprotective effects. He mentioned ongoing trials looking at that. We always love an excuse to tell patients that they should be moving around more and being physically active.
Dr. Williams: That was one of the more shocking things I learned, that exercise might actually be good for you. That will deeply inform my practice. Many of the treatments that we use for Parkinson’s only address symptoms. They don’t address progression or fix anything, but exercise can help with that.
Dr. Watto: Paul, the last question I wanted to ask you is about our role in primary care. Patients with Parkinson’s have autonomic symptoms. They have neurocognitive symptoms. What is our role in that as primary care physicians?
Dr. Williams: Myriad symptoms can accompany Parkinson’s, and we have experience with most of them. We should all feel fairly comfortable dealing with constipation, which can be a very bothersome symptom. And we can use our full arsenal for symptoms such as depression, anxiety, and even apathy – the anhedonia, which apparently can be the predominant feature. We do have the tools to address these problems.
This might be a situation where we might reach for bupropion or a tricyclic antidepressant, which might not be your initial choice for a patient with a possibly annoying mood disorder. But for someone with Parkinson’s disease, this actually may be very helpful. We know how to manage a lot of the symptoms that come along with Parkinson’s that are not just the motor symptoms, and we should take ownership of those things.
Dr. Watto: You can hear the rest of this podcast here. This has been another episode of The Curbsiders bringing you a little knowledge food for your brain hole. Until next time, I’ve been Dr Matthew Frank Watto.
Dr. Williams: And I’m Dr Paul Nelson Williams.
Dr. Watto is a clinical assistant professor, department of medicine, at the University of Pennsylvania, Philadelphia. Dr. Williams is Associate Professor of Clinical Medicine, Department of General Internal Medicine, at Temple University, Philadelphia. Neither Dr. Watto nor Dr. Williams reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. We had a great discussion on Parkinson’s Disease for Primary Care with Dr. Albert Hung. Paul, this was something that really made me nervous. I didn’t have a lot of comfort with it. But he taught us a lot of tips about how to recognize Parkinson’s.
I hadn’t been as aware of the premotor symptoms: constipation, hyposmia (loss of sense of smell), and rapid eye movement sleep behavior disorder. If patients have those early on and they aren’t explained by other things (especially the REM sleep behavior disorder), you should really key in because those patients are at risk of developing Parkinson’s years down the line. Those symptoms could present first, which just kind of blew my mind.
What tips do you have about how to recognize Parkinson’s? Do you want to talk about the physical exam?
Paul N. Williams, MD: You know I love the physical exam stuff, so I’m happy to talk about that.
You were deeply upset that cogwheel rigidity was not pathognomonic for Parkinson’s, but you made the point – and our guest agreed – that asymmetry tends to be the key here. And I really appreciated the point about reemergent tremor. This is this idea of a resting tremor. If someone has more parkinsonian features, you might see an intention tremor with essential tremor. If they reach out, it might seem steady at first, but if they hold long enough, then the tremor may kind of reemerge. I thought that was a neat distinction.
And this idea of cogwheel rigidity is a combination of some of the cardinal features of Parkinson’s – it’s a little bit of tremor and a little bit of rigidity too. There’s a baseline increase in tone, and then the tremor is superimposed on top of that. When you’re feeling cogwheeling, that’s actually what you’re feeling on examination. Parkinson’s, with all of its physical exam findings has always fascinated me.
Dr. Watto: He also told us about some red flags.
With classic idiopathic parkinsonism, there’s asymmetric involvement of the tremor. So red flags include a symmetric tremor, which might be something other than idiopathic parkinsonism. He also mentioned that one of the reasons you may want to get imaging (which is not always necessary if someone has a classic presentation), is if you see lower body–predominant symptoms of parkinsonism. These patients have rigidity or slowness of movement in their legs, but their upper bodies are not affected. They don’t have masked facies or the tremor in their hands. You might get an MRI in that case because that could be presentation of vascular dementia or vascular disease in the brain or even normal pressure hydrocephalus, which is a treatable condition. That would be one reason to get imaging.
What if the patient was exposed to a drug like a dopamine antagonist? They will get better in a couple of days, right?
Dr. Williams: This was a really fascinating point because we typically think if a patient’s symptoms are related to a drug exposure – in this case, drug-induced parkinsonism – we can just stop the medication and the symptoms will disappear in a couple of days as the drug leaves the system. But as it turns out, it might take much longer. A mistake that Dr Hung often sees is that the clinician stops the possibly offending agent, but when they don’t see an immediate relief of symptoms, they assume the drug wasn’t causing them. You really have to give the patient a fair shot off the medication to experience recovery because those symptoms can last weeks or even months after the drug is discontinued.
Dr. Watto: Dr Hung looks at the patient’s problem list and asks whether is there any reason this patient might have been exposed to one of these medications?
We’re not going to get too much into specific Parkinson’s treatment, but I was glad to hear that exercise actually improves mobility and may even have some neuroprotective effects. He mentioned ongoing trials looking at that. We always love an excuse to tell patients that they should be moving around more and being physically active.
Dr. Williams: That was one of the more shocking things I learned, that exercise might actually be good for you. That will deeply inform my practice. Many of the treatments that we use for Parkinson’s only address symptoms. They don’t address progression or fix anything, but exercise can help with that.
Dr. Watto: Paul, the last question I wanted to ask you is about our role in primary care. Patients with Parkinson’s have autonomic symptoms. They have neurocognitive symptoms. What is our role in that as primary care physicians?
Dr. Williams: Myriad symptoms can accompany Parkinson’s, and we have experience with most of them. We should all feel fairly comfortable dealing with constipation, which can be a very bothersome symptom. And we can use our full arsenal for symptoms such as depression, anxiety, and even apathy – the anhedonia, which apparently can be the predominant feature. We do have the tools to address these problems.
This might be a situation where we might reach for bupropion or a tricyclic antidepressant, which might not be your initial choice for a patient with a possibly annoying mood disorder. But for someone with Parkinson’s disease, this actually may be very helpful. We know how to manage a lot of the symptoms that come along with Parkinson’s that are not just the motor symptoms, and we should take ownership of those things.
Dr. Watto: You can hear the rest of this podcast here. This has been another episode of The Curbsiders bringing you a little knowledge food for your brain hole. Until next time, I’ve been Dr Matthew Frank Watto.
Dr. Williams: And I’m Dr Paul Nelson Williams.
Dr. Watto is a clinical assistant professor, department of medicine, at the University of Pennsylvania, Philadelphia. Dr. Williams is Associate Professor of Clinical Medicine, Department of General Internal Medicine, at Temple University, Philadelphia. Neither Dr. Watto nor Dr. Williams reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Matthew F. Watto, MD: Welcome back to The Curbsiders. We had a great discussion on Parkinson’s Disease for Primary Care with Dr. Albert Hung. Paul, this was something that really made me nervous. I didn’t have a lot of comfort with it. But he taught us a lot of tips about how to recognize Parkinson’s.
I hadn’t been as aware of the premotor symptoms: constipation, hyposmia (loss of sense of smell), and rapid eye movement sleep behavior disorder. If patients have those early on and they aren’t explained by other things (especially the REM sleep behavior disorder), you should really key in because those patients are at risk of developing Parkinson’s years down the line. Those symptoms could present first, which just kind of blew my mind.
What tips do you have about how to recognize Parkinson’s? Do you want to talk about the physical exam?
Paul N. Williams, MD: You know I love the physical exam stuff, so I’m happy to talk about that.
You were deeply upset that cogwheel rigidity was not pathognomonic for Parkinson’s, but you made the point – and our guest agreed – that asymmetry tends to be the key here. And I really appreciated the point about reemergent tremor. This is this idea of a resting tremor. If someone has more parkinsonian features, you might see an intention tremor with essential tremor. If they reach out, it might seem steady at first, but if they hold long enough, then the tremor may kind of reemerge. I thought that was a neat distinction.
And this idea of cogwheel rigidity is a combination of some of the cardinal features of Parkinson’s – it’s a little bit of tremor and a little bit of rigidity too. There’s a baseline increase in tone, and then the tremor is superimposed on top of that. When you’re feeling cogwheeling, that’s actually what you’re feeling on examination. Parkinson’s, with all of its physical exam findings has always fascinated me.
Dr. Watto: He also told us about some red flags.
With classic idiopathic parkinsonism, there’s asymmetric involvement of the tremor. So red flags include a symmetric tremor, which might be something other than idiopathic parkinsonism. He also mentioned that one of the reasons you may want to get imaging (which is not always necessary if someone has a classic presentation), is if you see lower body–predominant symptoms of parkinsonism. These patients have rigidity or slowness of movement in their legs, but their upper bodies are not affected. They don’t have masked facies or the tremor in their hands. You might get an MRI in that case because that could be presentation of vascular dementia or vascular disease in the brain or even normal pressure hydrocephalus, which is a treatable condition. That would be one reason to get imaging.
What if the patient was exposed to a drug like a dopamine antagonist? They will get better in a couple of days, right?
Dr. Williams: This was a really fascinating point because we typically think if a patient’s symptoms are related to a drug exposure – in this case, drug-induced parkinsonism – we can just stop the medication and the symptoms will disappear in a couple of days as the drug leaves the system. But as it turns out, it might take much longer. A mistake that Dr Hung often sees is that the clinician stops the possibly offending agent, but when they don’t see an immediate relief of symptoms, they assume the drug wasn’t causing them. You really have to give the patient a fair shot off the medication to experience recovery because those symptoms can last weeks or even months after the drug is discontinued.
Dr. Watto: Dr Hung looks at the patient’s problem list and asks whether is there any reason this patient might have been exposed to one of these medications?
We’re not going to get too much into specific Parkinson’s treatment, but I was glad to hear that exercise actually improves mobility and may even have some neuroprotective effects. He mentioned ongoing trials looking at that. We always love an excuse to tell patients that they should be moving around more and being physically active.
Dr. Williams: That was one of the more shocking things I learned, that exercise might actually be good for you. That will deeply inform my practice. Many of the treatments that we use for Parkinson’s only address symptoms. They don’t address progression or fix anything, but exercise can help with that.
Dr. Watto: Paul, the last question I wanted to ask you is about our role in primary care. Patients with Parkinson’s have autonomic symptoms. They have neurocognitive symptoms. What is our role in that as primary care physicians?
Dr. Williams: Myriad symptoms can accompany Parkinson’s, and we have experience with most of them. We should all feel fairly comfortable dealing with constipation, which can be a very bothersome symptom. And we can use our full arsenal for symptoms such as depression, anxiety, and even apathy – the anhedonia, which apparently can be the predominant feature. We do have the tools to address these problems.
This might be a situation where we might reach for bupropion or a tricyclic antidepressant, which might not be your initial choice for a patient with a possibly annoying mood disorder. But for someone with Parkinson’s disease, this actually may be very helpful. We know how to manage a lot of the symptoms that come along with Parkinson’s that are not just the motor symptoms, and we should take ownership of those things.
Dr. Watto: You can hear the rest of this podcast here. This has been another episode of The Curbsiders bringing you a little knowledge food for your brain hole. Until next time, I’ve been Dr Matthew Frank Watto.
Dr. Williams: And I’m Dr Paul Nelson Williams.
Dr. Watto is a clinical assistant professor, department of medicine, at the University of Pennsylvania, Philadelphia. Dr. Williams is Associate Professor of Clinical Medicine, Department of General Internal Medicine, at Temple University, Philadelphia. Neither Dr. Watto nor Dr. Williams reported any relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Antiamyloids linked to accelerated brain atrophy
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
a comprehensive meta-analysis of MRI data from clinical trials suggests.
Depending on the anti–amyloid-beta drug class, these agents can accelerate loss of whole brain and hippocampal volume and increase ventricular volume. This has been shown for some of the beta-secretase inhibitors and with several of the antiamyloid monoclonal antibodies, researchers noted.
“These data warrant concern, but we can’t make any firm conclusions yet. It is possible that the finding is not detrimental, but the usual interpretation of this finding is that volume changes are a surrogate for disease progression,” study investigator Scott Ayton, PhD, of the Florey Institute of Neuroscience and Mental Health, University of Melbourne, said in an interview.
“These data should be factored into the decisions by clinicians when they consider prescribing antiamyloid therapies. Like any side effect, clinicians should inform patients regarding the risk of brain atrophy. Patients should be actively monitored for this side effect,” Dr. Ayton said.
The study was published online in Neurology.
Earlier progression from MCI to AD?
Dr. Ayton and colleagues evaluated brain volume changes in 31 clinical trials of anti–amyloid-beta drugs that demonstrated a favorable change in at least one biomarker of pathological amyloid-beta and included detailed MRI data sufficient to assess the volumetric changes in at least one brain region.
A meta-analysis on the highest dose in each trial on the hippocampus, ventricles, and whole brain showed drug-induced acceleration of volume changes that varied by anti–amyloid-beta drug class.
Secretase inhibitors accelerated atrophy in the hippocampus (mean difference –37.1 mcL; –19.6% relative to change in placebo) and whole brain (mean difference –3.3 mL; –21.8% relative to change in placebo), but not ventricles.
Conversely, monoclonal antibodies caused accelerated ventricular enlargement (mean difference +1.3 mL; +23.8% relative to change in placebo), which was driven by the subset of monoclonal antibodies that induce amyloid-related imaging abnormalities (ARIA) (+2.1 mL; +38.7% relative to change in placebo). There was a “striking correlation between ventricular volume and ARIA frequency,” the investigators reported.
The effect of ARIA-inducing monoclonal antibodies on whole brain volume varied, with accelerated whole brain volume loss caused by donanemab (mean difference –4.6 mL; +23% relative to change in placebo) and lecanemab (–5.2 mL; +36.4% relative to change in placebo). This was not observed with aducanumab and bapineuzumab.
Monoclonal antibodies did not cause accelerated volume loss to the hippocampus regardless of whether they caused ARIA.
The researchers also modeled the effect of anti–amyloid-beta drugs on brain volume changes. In this analysis, participants with mild cognitive impairment (MCI) treated with anti–amyloid-beta drugs were projected to have a “material regression” toward brain volumes typical of AD roughly 8 months earlier than untreated peers.
The data, they note, “permit robust conclusions regarding the effect of [anti–amyloid-beta] drug classes on different brain structures, but the lack of individual patient data (which has yet to be released) limits the interpretations of our findings.”
“Questions like which brain regions are impacted by [anti–amyloid-beta] drugs and whether the volume changes are related to ARIA, plaque loss, cognitive/noncognitive outcomes, or clinical factors such as age, sex, and apoE4 genotype can and should be addressed with available data,” said Dr. Ayton.
Dr. Ayton and colleagues called on data safety monitoring boards (DSMBs) for current clinical trials of anti–amyloid-beta drugs to review volumetric data to determine if patient safety is at risk, particularly in patients who develop ARIA.
In addition, they noted ethics boards that approve trials for anti–amyloid-beta drugs “should request that volume changes be actively monitored. Long-term follow-up of brain volumes should be factored into the trial designs to determine if brain atrophy is progressive, particularly in patients who develop ARIA.”
Finally, they added that drug companies that have conducted trials of anti–amyloid-beta drugs should interrogate prior data on brain volume, report the findings, and release the data for researchers to investigate.
“I have been banging on about this for years,” said Dr. Ayton. “Unfortunately, my raising of this issue has not led to any response. The data are not available, and the basic questions haven’t been asked (publicly).”
Commendable research
In an accompanying editorial, Frederik Barkhof, MD, PhD, with Amsterdam University Medical Centers, and David Knopman, MD, with Mayo Clinic Alzheimer’s Disease Research Center, Rochester, Minn., wrote that the investigators should be “commended” for their analysis.
“The reality in 2023 is that the relevance of brain volume reductions in this therapeutic context remains uncertain,” they wrote.
“Longer periods of observation will be needed to know whether the brain volume losses continue at an accelerated rate or if they attenuate or disappear. Ultimately, it’s the clinical outcomes that matter, regardless of the MRI changes,” Barkhof and Knopman concluded.
The research was supported by funds from the Australian National Health & Medical Research Council. Dr. Ayton reported being a consultant for Eisai in the past 3 years. Dr. Barkhof reported serving on the data and safety monitoring board for Prothena and the A45-AHEAD studies; being a steering committee member for Merck, Bayer, and Biogen; and being a consultant for IXICO, Roche, Celltrion, Rewind Therapeutics, and Combinostics. Dr. Knopman reported serving on the DSMB for the Dominantly Inherited Alzheimer Network Treatment Unit study; serving on a DSMB for a tau therapeutic for Biogen; being an investigator for clinical trials sponsored by Biogen, Lilly Pharmaceuticals, and the University of Southern California. He reported consulting with Roche, Samus Therapeutics, Magellan Health, BioVie, and Alzeca Biosciences.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
Malpractice risks for docs who oversee NPs or PAs
Even in states that have abolished requirements that NPs be physician-supervised, physicians may still be liable by virtue of employing the NP, according to William P. Sullivan, DO, an attorney and emergency physician in Frankfort, Ill.
Indeed, the vast majority of lawsuits against NPs and PAs name the supervising physician. According to a study of claims against NPs from 2011 to 2016, 82% of the cases also named the supervising physician.
Employed or contracted physicians assigned to supervise NPs or PAs are also affected, Dr. Sullivan said. “The employed physicians’ contract with a hospital or staffing company may require them to assist in the selection, supervision, and/or training of NPs or PAs,” he said. He added that supervisory duties may also be assigned through hospital bylaws.
“The physician is usually not paid anything extra for this work and may not be given extra time to perform it,” Dr. Sullivan said. But still, he said, that physician could be named in a lawsuit and wind up bearing some responsibility for an NP’s or PA’s mistake.
In addition to facing medical malpractice suits, Dr. Sullivan said, doctors are often sanctioned by state licensure boards for improperly supervising NPs and PAs. Licensure boards often require extensive protocols for supervision of NPs and PAs.
Yet more states are removing supervision requirements
With the addition of Kansas and New York in 2022 and California in 2023, 27 states no longer require supervision for all or most NPs. Sixteen of those states, including New York and California, have instituted progressive practice authority that requires temporary supervision of new NPs but then removes supervision after a period of 6 months to 4 years, depending on the state, for the rest of their career.
“When it comes to NP independence, the horse is already out of the barn,” Dr. Sullivan said. “It’s unlikely that states will repeal laws granting NPs independence, and in fact, more states are likely to pass them.”
*PAs, in contrast, are well behind NPs in achieving independence, but the American Academy of Physician Associates (AAPA) is calling to eliminate a mandated relationship with a specific physician. So far, Utah, North Dakota and Wyoming have ended physician supervision of PAs, while California and Hawaii have eliminated mandated chart review. Other states are considering eliminating physician supervision of PAs, according to the AAPA.
In states that have abolished oversight requirements for NPs, “liability can then shift to the NP when the NP is fully independent,” Cathy Klein, an advanced practice registered nurse who helped found the NP profession 50 years ago, told this news organization. “More NPs are starting their own practices, and in many cases, patients actually prefer to see an NP.”
As more NPs became more autonomous, the average payment that NPs incurred in professional liability lawsuits rose by 10.5% from 2017 to 2022, to $332,187, according to the Nurses Service Organization (NSO), a nursing malpractice insurer.
The number of malpractice judgments against autonomous NPs alone has also been rising. From 2012 to 2017, autonomous NPs’ share of all NP cases rose from 7% to 16.4%, the NSO reported.
The good news for physicians is that states’ removal of restrictions on NPs has reduced physicians’ liability to some extent. A 2017 study found that enacting less restrictive scope-of-practice laws for NPs decreased the number of payments made by physicians in NP cases by as much as 31%.
However, the top location for NP payouts remains the physician’s office, not the autonomous NP’s practice, according to the latter NSO report. Plaintiffs sue NPs’ and PAs’ supervising physicians on the basis of legal concepts, such as vicarious liability and respondeat superior. Even if the physician-employer never saw the patient, he or she can be held liable.
Court cases in which supervising physician was found liable
There are plenty of judgments against supervising or collaborating physicians when the NP or PA made the error. Typically, the doctor was faulted for paying little attention to the NP or PA he or she was supposed to supervise.
Dr. Sullivan points to a 2016 case in which a New York jury held a physician 40% liable for a $7 million judgment in a malpractice case involving a PA’s care of a patient in the emergency department. The case is Shajan v. South Nassau Community Hospital in New York.
“The patient presented with nontraumatic leg pain to his lower leg, was diagnosed by the PA with a muscle strain, and discharged without a physician evaluation,” Dr. Sullivan said. The next day, the patient visited an orthopedist who immediately diagnosed compartment syndrome, an emergent condition in which pressure builds up in an affected extremity, damaging the muscles and nerves. “The patient developed irreversible nerve damage and chronic regional pain syndrome,” he said.
A malpractice lawsuit named the PA and the emergency physician he was supposed to be reporting to. Even though the physician had never seen the patient, he had signed off on the PA’s note from a patient’s ED visit. “Testimony during the trial focused on hospital protocols that the supervising physician was supposed to take,” Dr. Sullivan said.
When doctors share fault, they frequently failed to follow the collaborative agreement with the NP or PA. In Collip v. Ratts, a 2015 Indiana case in which the patient died from a drug interaction, the doctor’s certified public accountant stated that the doctor was required to review at least 5% of the NP’s charts every week to evaluate her prescriptive practices.
The doctor admitted that he never reviewed the NP’s charts on a weekly basis. He did conduct some cursory reviews of some of the NP’s notes, and in them he noted concerns for her prescribing practices and suggested she attend a narcotics-prescribing seminar, but he did not follow up to make sure she had done this.
Sometimes the NP or PA who made the mistake may actually be dropped from the lawsuit, leaving the supervising physician fully liable. In these cases, courts reason that a fully engaged supervisor could have prevented the error. In the 2006 case of Husak v. Siegal, the Florida Supreme Court dropped the NP from the case, ruling that the NP had provided the supervising doctor all the information he needed in order to tell her what to do for the patient.
The court noted the physician had failed to look at the chart, even though he was required to do so under his supervisory agreement with the NP. The doctor “could have made the correct diagnosis or referral had he been attentive,” the court said. Therefore, there was “no evidence of independent negligence” by the NP, even though she was the one who had made the incorrect diagnosis that harmed the patient.
When states require an autonomous NP to have a supervisory relationship with a doctor, the supervisor may be unavailable and may fail to designate a substitute. In Texas in January 2019, a 7-year-old girl died of pneumonia after being treated by an NP in an urgent care clinic. The NP had told the parents that the child could safely go home and only needed ibuprofen. The parents brought the girl back home, and she died 15 hours later. The Wattenbargers sued the NP, and the doctor’s supervision was a topic in the trial.
The supervising physician for the NP was out of the country at the time. He said that he had found a substitute, but the substitute doctor testified she had no idea she was designated to be the substitute, according to Niran Al-Agba, MD, a family physician in Silverdale, Wash., who has written on the Texas case. Dr. Al-Agba told this news organization the case appears to have been settled confidentially.
Different standards for expert witnesses
In many states, courts do not allow physicians to testify as expert witnesses in malpractice cases against NPs, arguing that nurses have a different set of standards than doctors have, Dr. Sullivan reported.
These states include Arkansas, Illinois, North Carolina, and New York, according to a report by SEAK Inc., an expert witness training program. The report said most other states allow physician experts in these cases, but they may still require that they have experience with the nursing standard of care.
Dr. Sullivan said some courts are whittling away at the ban on physician experts, and the ban may eventually disappear. He reported that in Oklahoma, which normally upholds the ban, a judge recently allowed a physician-expert to testify in a case involving the death of a 19-year-old woman, Alexus Ochoa, in an ED staffed by an NP. The judge reasoned that Ms. Ochoa’s parents assumed the ED was staffed by physicians and would adhere to medical standards.
Supervision pointers from a physician
Physicians who supervise NPs or PAs say it is important to keep track of their skills and help them sharpen their expertise. Their scope of practice and physicians’ supervisory responsibilities are included in the collaborative agreement.
Arthur Apolinario, MD, a family physician in Clinton, N.C., says his 10-physician practice, which employs six NPs and one PA, works under a collaborative agreement. “The agreement defines each person’s scope of practice. They can’t do certain procedures, such as surgery, and they need extra training before doing certain tasks alone, such as joint injection.
“You have to always figure that if there is a lawsuit against one of them, you as the supervising physician would be named,” said Dr. Apolinario, who is also president of the North Carolina Medical Society. “We try to avert mistakes by meeting regularly with our NPs and PAs and making sure they keep up to date.”
Collaborating with autonomous NPs
Even when NPs operate independently in states that have abolished supervision, physicians may still have some liability if they give NPs advice, Dr. Al-Agba said.
At her Washington state practice, Dr. Al-Agba shares an office with an autonomous NP. “We share overhead and a front desk, but we have separate patients,” Dr. Al-Agba said. “This arrangement works very well for both of us.”
The NP sometimes asks her for advice. When this occurs, Dr. Al-Agba said she always makes sure to see the patient first. “If you don’t actually see the patient, there could be a misunderstanding that could lead to an error,” she said.
Conclusion
Even though NPs now have autonomy in most states, supervising physicians may still be liable for NP malpractice by virtue of being their employers, and physicians in the remaining states are liable for NPs through state law and for PAs in virtually all the states. To determine the supervising physician’s fault, courts often study whether the physician has met the terms of the collaborative agreement.
Physicians can reduce collaborating NPs’ and PAs’ liability by properly training them, by verifying their scope of practice, by making themselves easily available for consultation, and by occasionally seeing their patients. If their NPs and PAs do commit malpractice, supervising physicians may be able to protect themselves from liability by adhering to all requirements of the collaborative agreement.
*Correction, 4/19/2023: An earlier version of this story misstated the name of the AAPA and the states that have ended physician supervision of PAs.
A version of this article first appeared on Medscape.com.
Even in states that have abolished requirements that NPs be physician-supervised, physicians may still be liable by virtue of employing the NP, according to William P. Sullivan, DO, an attorney and emergency physician in Frankfort, Ill.
Indeed, the vast majority of lawsuits against NPs and PAs name the supervising physician. According to a study of claims against NPs from 2011 to 2016, 82% of the cases also named the supervising physician.
Employed or contracted physicians assigned to supervise NPs or PAs are also affected, Dr. Sullivan said. “The employed physicians’ contract with a hospital or staffing company may require them to assist in the selection, supervision, and/or training of NPs or PAs,” he said. He added that supervisory duties may also be assigned through hospital bylaws.
“The physician is usually not paid anything extra for this work and may not be given extra time to perform it,” Dr. Sullivan said. But still, he said, that physician could be named in a lawsuit and wind up bearing some responsibility for an NP’s or PA’s mistake.
In addition to facing medical malpractice suits, Dr. Sullivan said, doctors are often sanctioned by state licensure boards for improperly supervising NPs and PAs. Licensure boards often require extensive protocols for supervision of NPs and PAs.
Yet more states are removing supervision requirements
With the addition of Kansas and New York in 2022 and California in 2023, 27 states no longer require supervision for all or most NPs. Sixteen of those states, including New York and California, have instituted progressive practice authority that requires temporary supervision of new NPs but then removes supervision after a period of 6 months to 4 years, depending on the state, for the rest of their career.
“When it comes to NP independence, the horse is already out of the barn,” Dr. Sullivan said. “It’s unlikely that states will repeal laws granting NPs independence, and in fact, more states are likely to pass them.”
*PAs, in contrast, are well behind NPs in achieving independence, but the American Academy of Physician Associates (AAPA) is calling to eliminate a mandated relationship with a specific physician. So far, Utah, North Dakota and Wyoming have ended physician supervision of PAs, while California and Hawaii have eliminated mandated chart review. Other states are considering eliminating physician supervision of PAs, according to the AAPA.
In states that have abolished oversight requirements for NPs, “liability can then shift to the NP when the NP is fully independent,” Cathy Klein, an advanced practice registered nurse who helped found the NP profession 50 years ago, told this news organization. “More NPs are starting their own practices, and in many cases, patients actually prefer to see an NP.”
As more NPs became more autonomous, the average payment that NPs incurred in professional liability lawsuits rose by 10.5% from 2017 to 2022, to $332,187, according to the Nurses Service Organization (NSO), a nursing malpractice insurer.
The number of malpractice judgments against autonomous NPs alone has also been rising. From 2012 to 2017, autonomous NPs’ share of all NP cases rose from 7% to 16.4%, the NSO reported.
The good news for physicians is that states’ removal of restrictions on NPs has reduced physicians’ liability to some extent. A 2017 study found that enacting less restrictive scope-of-practice laws for NPs decreased the number of payments made by physicians in NP cases by as much as 31%.
However, the top location for NP payouts remains the physician’s office, not the autonomous NP’s practice, according to the latter NSO report. Plaintiffs sue NPs’ and PAs’ supervising physicians on the basis of legal concepts, such as vicarious liability and respondeat superior. Even if the physician-employer never saw the patient, he or she can be held liable.
Court cases in which supervising physician was found liable
There are plenty of judgments against supervising or collaborating physicians when the NP or PA made the error. Typically, the doctor was faulted for paying little attention to the NP or PA he or she was supposed to supervise.
Dr. Sullivan points to a 2016 case in which a New York jury held a physician 40% liable for a $7 million judgment in a malpractice case involving a PA’s care of a patient in the emergency department. The case is Shajan v. South Nassau Community Hospital in New York.
“The patient presented with nontraumatic leg pain to his lower leg, was diagnosed by the PA with a muscle strain, and discharged without a physician evaluation,” Dr. Sullivan said. The next day, the patient visited an orthopedist who immediately diagnosed compartment syndrome, an emergent condition in which pressure builds up in an affected extremity, damaging the muscles and nerves. “The patient developed irreversible nerve damage and chronic regional pain syndrome,” he said.
A malpractice lawsuit named the PA and the emergency physician he was supposed to be reporting to. Even though the physician had never seen the patient, he had signed off on the PA’s note from a patient’s ED visit. “Testimony during the trial focused on hospital protocols that the supervising physician was supposed to take,” Dr. Sullivan said.
When doctors share fault, they frequently failed to follow the collaborative agreement with the NP or PA. In Collip v. Ratts, a 2015 Indiana case in which the patient died from a drug interaction, the doctor’s certified public accountant stated that the doctor was required to review at least 5% of the NP’s charts every week to evaluate her prescriptive practices.
The doctor admitted that he never reviewed the NP’s charts on a weekly basis. He did conduct some cursory reviews of some of the NP’s notes, and in them he noted concerns for her prescribing practices and suggested she attend a narcotics-prescribing seminar, but he did not follow up to make sure she had done this.
Sometimes the NP or PA who made the mistake may actually be dropped from the lawsuit, leaving the supervising physician fully liable. In these cases, courts reason that a fully engaged supervisor could have prevented the error. In the 2006 case of Husak v. Siegal, the Florida Supreme Court dropped the NP from the case, ruling that the NP had provided the supervising doctor all the information he needed in order to tell her what to do for the patient.
The court noted the physician had failed to look at the chart, even though he was required to do so under his supervisory agreement with the NP. The doctor “could have made the correct diagnosis or referral had he been attentive,” the court said. Therefore, there was “no evidence of independent negligence” by the NP, even though she was the one who had made the incorrect diagnosis that harmed the patient.
When states require an autonomous NP to have a supervisory relationship with a doctor, the supervisor may be unavailable and may fail to designate a substitute. In Texas in January 2019, a 7-year-old girl died of pneumonia after being treated by an NP in an urgent care clinic. The NP had told the parents that the child could safely go home and only needed ibuprofen. The parents brought the girl back home, and she died 15 hours later. The Wattenbargers sued the NP, and the doctor’s supervision was a topic in the trial.
The supervising physician for the NP was out of the country at the time. He said that he had found a substitute, but the substitute doctor testified she had no idea she was designated to be the substitute, according to Niran Al-Agba, MD, a family physician in Silverdale, Wash., who has written on the Texas case. Dr. Al-Agba told this news organization the case appears to have been settled confidentially.
Different standards for expert witnesses
In many states, courts do not allow physicians to testify as expert witnesses in malpractice cases against NPs, arguing that nurses have a different set of standards than doctors have, Dr. Sullivan reported.
These states include Arkansas, Illinois, North Carolina, and New York, according to a report by SEAK Inc., an expert witness training program. The report said most other states allow physician experts in these cases, but they may still require that they have experience with the nursing standard of care.
Dr. Sullivan said some courts are whittling away at the ban on physician experts, and the ban may eventually disappear. He reported that in Oklahoma, which normally upholds the ban, a judge recently allowed a physician-expert to testify in a case involving the death of a 19-year-old woman, Alexus Ochoa, in an ED staffed by an NP. The judge reasoned that Ms. Ochoa’s parents assumed the ED was staffed by physicians and would adhere to medical standards.
Supervision pointers from a physician
Physicians who supervise NPs or PAs say it is important to keep track of their skills and help them sharpen their expertise. Their scope of practice and physicians’ supervisory responsibilities are included in the collaborative agreement.
Arthur Apolinario, MD, a family physician in Clinton, N.C., says his 10-physician practice, which employs six NPs and one PA, works under a collaborative agreement. “The agreement defines each person’s scope of practice. They can’t do certain procedures, such as surgery, and they need extra training before doing certain tasks alone, such as joint injection.
“You have to always figure that if there is a lawsuit against one of them, you as the supervising physician would be named,” said Dr. Apolinario, who is also president of the North Carolina Medical Society. “We try to avert mistakes by meeting regularly with our NPs and PAs and making sure they keep up to date.”
Collaborating with autonomous NPs
Even when NPs operate independently in states that have abolished supervision, physicians may still have some liability if they give NPs advice, Dr. Al-Agba said.
At her Washington state practice, Dr. Al-Agba shares an office with an autonomous NP. “We share overhead and a front desk, but we have separate patients,” Dr. Al-Agba said. “This arrangement works very well for both of us.”
The NP sometimes asks her for advice. When this occurs, Dr. Al-Agba said she always makes sure to see the patient first. “If you don’t actually see the patient, there could be a misunderstanding that could lead to an error,” she said.
Conclusion
Even though NPs now have autonomy in most states, supervising physicians may still be liable for NP malpractice by virtue of being their employers, and physicians in the remaining states are liable for NPs through state law and for PAs in virtually all the states. To determine the supervising physician’s fault, courts often study whether the physician has met the terms of the collaborative agreement.
Physicians can reduce collaborating NPs’ and PAs’ liability by properly training them, by verifying their scope of practice, by making themselves easily available for consultation, and by occasionally seeing their patients. If their NPs and PAs do commit malpractice, supervising physicians may be able to protect themselves from liability by adhering to all requirements of the collaborative agreement.
*Correction, 4/19/2023: An earlier version of this story misstated the name of the AAPA and the states that have ended physician supervision of PAs.
A version of this article first appeared on Medscape.com.
Even in states that have abolished requirements that NPs be physician-supervised, physicians may still be liable by virtue of employing the NP, according to William P. Sullivan, DO, an attorney and emergency physician in Frankfort, Ill.
Indeed, the vast majority of lawsuits against NPs and PAs name the supervising physician. According to a study of claims against NPs from 2011 to 2016, 82% of the cases also named the supervising physician.
Employed or contracted physicians assigned to supervise NPs or PAs are also affected, Dr. Sullivan said. “The employed physicians’ contract with a hospital or staffing company may require them to assist in the selection, supervision, and/or training of NPs or PAs,” he said. He added that supervisory duties may also be assigned through hospital bylaws.
“The physician is usually not paid anything extra for this work and may not be given extra time to perform it,” Dr. Sullivan said. But still, he said, that physician could be named in a lawsuit and wind up bearing some responsibility for an NP’s or PA’s mistake.
In addition to facing medical malpractice suits, Dr. Sullivan said, doctors are often sanctioned by state licensure boards for improperly supervising NPs and PAs. Licensure boards often require extensive protocols for supervision of NPs and PAs.
Yet more states are removing supervision requirements
With the addition of Kansas and New York in 2022 and California in 2023, 27 states no longer require supervision for all or most NPs. Sixteen of those states, including New York and California, have instituted progressive practice authority that requires temporary supervision of new NPs but then removes supervision after a period of 6 months to 4 years, depending on the state, for the rest of their career.
“When it comes to NP independence, the horse is already out of the barn,” Dr. Sullivan said. “It’s unlikely that states will repeal laws granting NPs independence, and in fact, more states are likely to pass them.”
*PAs, in contrast, are well behind NPs in achieving independence, but the American Academy of Physician Associates (AAPA) is calling to eliminate a mandated relationship with a specific physician. So far, Utah, North Dakota and Wyoming have ended physician supervision of PAs, while California and Hawaii have eliminated mandated chart review. Other states are considering eliminating physician supervision of PAs, according to the AAPA.
In states that have abolished oversight requirements for NPs, “liability can then shift to the NP when the NP is fully independent,” Cathy Klein, an advanced practice registered nurse who helped found the NP profession 50 years ago, told this news organization. “More NPs are starting their own practices, and in many cases, patients actually prefer to see an NP.”
As more NPs became more autonomous, the average payment that NPs incurred in professional liability lawsuits rose by 10.5% from 2017 to 2022, to $332,187, according to the Nurses Service Organization (NSO), a nursing malpractice insurer.
The number of malpractice judgments against autonomous NPs alone has also been rising. From 2012 to 2017, autonomous NPs’ share of all NP cases rose from 7% to 16.4%, the NSO reported.
The good news for physicians is that states’ removal of restrictions on NPs has reduced physicians’ liability to some extent. A 2017 study found that enacting less restrictive scope-of-practice laws for NPs decreased the number of payments made by physicians in NP cases by as much as 31%.
However, the top location for NP payouts remains the physician’s office, not the autonomous NP’s practice, according to the latter NSO report. Plaintiffs sue NPs’ and PAs’ supervising physicians on the basis of legal concepts, such as vicarious liability and respondeat superior. Even if the physician-employer never saw the patient, he or she can be held liable.
Court cases in which supervising physician was found liable
There are plenty of judgments against supervising or collaborating physicians when the NP or PA made the error. Typically, the doctor was faulted for paying little attention to the NP or PA he or she was supposed to supervise.
Dr. Sullivan points to a 2016 case in which a New York jury held a physician 40% liable for a $7 million judgment in a malpractice case involving a PA’s care of a patient in the emergency department. The case is Shajan v. South Nassau Community Hospital in New York.
“The patient presented with nontraumatic leg pain to his lower leg, was diagnosed by the PA with a muscle strain, and discharged without a physician evaluation,” Dr. Sullivan said. The next day, the patient visited an orthopedist who immediately diagnosed compartment syndrome, an emergent condition in which pressure builds up in an affected extremity, damaging the muscles and nerves. “The patient developed irreversible nerve damage and chronic regional pain syndrome,” he said.
A malpractice lawsuit named the PA and the emergency physician he was supposed to be reporting to. Even though the physician had never seen the patient, he had signed off on the PA’s note from a patient’s ED visit. “Testimony during the trial focused on hospital protocols that the supervising physician was supposed to take,” Dr. Sullivan said.
When doctors share fault, they frequently failed to follow the collaborative agreement with the NP or PA. In Collip v. Ratts, a 2015 Indiana case in which the patient died from a drug interaction, the doctor’s certified public accountant stated that the doctor was required to review at least 5% of the NP’s charts every week to evaluate her prescriptive practices.
The doctor admitted that he never reviewed the NP’s charts on a weekly basis. He did conduct some cursory reviews of some of the NP’s notes, and in them he noted concerns for her prescribing practices and suggested she attend a narcotics-prescribing seminar, but he did not follow up to make sure she had done this.
Sometimes the NP or PA who made the mistake may actually be dropped from the lawsuit, leaving the supervising physician fully liable. In these cases, courts reason that a fully engaged supervisor could have prevented the error. In the 2006 case of Husak v. Siegal, the Florida Supreme Court dropped the NP from the case, ruling that the NP had provided the supervising doctor all the information he needed in order to tell her what to do for the patient.
The court noted the physician had failed to look at the chart, even though he was required to do so under his supervisory agreement with the NP. The doctor “could have made the correct diagnosis or referral had he been attentive,” the court said. Therefore, there was “no evidence of independent negligence” by the NP, even though she was the one who had made the incorrect diagnosis that harmed the patient.
When states require an autonomous NP to have a supervisory relationship with a doctor, the supervisor may be unavailable and may fail to designate a substitute. In Texas in January 2019, a 7-year-old girl died of pneumonia after being treated by an NP in an urgent care clinic. The NP had told the parents that the child could safely go home and only needed ibuprofen. The parents brought the girl back home, and she died 15 hours later. The Wattenbargers sued the NP, and the doctor’s supervision was a topic in the trial.
The supervising physician for the NP was out of the country at the time. He said that he had found a substitute, but the substitute doctor testified she had no idea she was designated to be the substitute, according to Niran Al-Agba, MD, a family physician in Silverdale, Wash., who has written on the Texas case. Dr. Al-Agba told this news organization the case appears to have been settled confidentially.
Different standards for expert witnesses
In many states, courts do not allow physicians to testify as expert witnesses in malpractice cases against NPs, arguing that nurses have a different set of standards than doctors have, Dr. Sullivan reported.
These states include Arkansas, Illinois, North Carolina, and New York, according to a report by SEAK Inc., an expert witness training program. The report said most other states allow physician experts in these cases, but they may still require that they have experience with the nursing standard of care.
Dr. Sullivan said some courts are whittling away at the ban on physician experts, and the ban may eventually disappear. He reported that in Oklahoma, which normally upholds the ban, a judge recently allowed a physician-expert to testify in a case involving the death of a 19-year-old woman, Alexus Ochoa, in an ED staffed by an NP. The judge reasoned that Ms. Ochoa’s parents assumed the ED was staffed by physicians and would adhere to medical standards.
Supervision pointers from a physician
Physicians who supervise NPs or PAs say it is important to keep track of their skills and help them sharpen their expertise. Their scope of practice and physicians’ supervisory responsibilities are included in the collaborative agreement.
Arthur Apolinario, MD, a family physician in Clinton, N.C., says his 10-physician practice, which employs six NPs and one PA, works under a collaborative agreement. “The agreement defines each person’s scope of practice. They can’t do certain procedures, such as surgery, and they need extra training before doing certain tasks alone, such as joint injection.
“You have to always figure that if there is a lawsuit against one of them, you as the supervising physician would be named,” said Dr. Apolinario, who is also president of the North Carolina Medical Society. “We try to avert mistakes by meeting regularly with our NPs and PAs and making sure they keep up to date.”
Collaborating with autonomous NPs
Even when NPs operate independently in states that have abolished supervision, physicians may still have some liability if they give NPs advice, Dr. Al-Agba said.
At her Washington state practice, Dr. Al-Agba shares an office with an autonomous NP. “We share overhead and a front desk, but we have separate patients,” Dr. Al-Agba said. “This arrangement works very well for both of us.”
The NP sometimes asks her for advice. When this occurs, Dr. Al-Agba said she always makes sure to see the patient first. “If you don’t actually see the patient, there could be a misunderstanding that could lead to an error,” she said.
Conclusion
Even though NPs now have autonomy in most states, supervising physicians may still be liable for NP malpractice by virtue of being their employers, and physicians in the remaining states are liable for NPs through state law and for PAs in virtually all the states. To determine the supervising physician’s fault, courts often study whether the physician has met the terms of the collaborative agreement.
Physicians can reduce collaborating NPs’ and PAs’ liability by properly training them, by verifying their scope of practice, by making themselves easily available for consultation, and by occasionally seeing their patients. If their NPs and PAs do commit malpractice, supervising physicians may be able to protect themselves from liability by adhering to all requirements of the collaborative agreement.
*Correction, 4/19/2023: An earlier version of this story misstated the name of the AAPA and the states that have ended physician supervision of PAs.
A version of this article first appeared on Medscape.com.
Noisy incubators could stunt infant hearing
Incubators save the lives of many babies, but new data suggest that the ambient noise associated with the incubator experience could put babies’ hearing and language development skills at risk.
Previous studies have shown that the neonatal intensive care unit is a noisy environment, but specific data on levels of sound inside and outside incubators are limited, wrote Christoph Reuter, MA, a musicology professor at the University of Vienna, and colleagues.
“By the age of 3 years, deficits in language acquisition are detectable in nearly 50% of very preterm infants,” and high levels of NICU noise have been cited as possible contributors to this increased risk, the researchers say.
In a study published in Frontiers in Pediatrics, the researchers aimed to compare real-life NICU noise with previously reported levels to describe the sound characteristics and to identify resonance characteristics inside an incubator.
The study was conducted at the Pediatric Simulation Center at the Medical University of Vienna. The researchers placed a simulation mannequin with an ear microphone inside an incubator. They also placed microphones outside the incubator to collect measures of outside noise and activity involved in NICU care.
Data regarding sound were collected for 11 environmental noises and 12 incubator handlings using weighted and unweighted decibel levels. Specific environmental noises included starting the incubator engine; environmental noise with incubator off; environmental noise with incubator on; normal conversation; light conversation; laughter; telephone sounds; the infusion pump alarm; the monitor alarm (anomaly); the monitor alarm (emergency); and blood pressure measurement.
The 12 incubator handling noises included those associated with water flap, water pouring into the incubator, incubator doors opening properly, incubators doors closing properly, incubator doors closing improperly, hatch closing, hatch opening, incubator drawer, neighbor incubator doors closing (1.82 m distance), taking a stethoscope from the incubator wall, putting a stethoscope on the incubator, and suctioning tube. Noise from six levels of respiratory support was also measured.
The researchers reported that the incubator tended to dampen most sounds but also that some sounds resonated inside the incubator, which raised the interior noise level by as much as 28 decibels.
Most of the measures using both A-weighted decibels (dBA) and sound pressure level decibels (dBSPL) were above the 45-decibel level for neonatal sound exposure recommended by the American Academy of Pediatrics. The measurements (dBA) versus unweighted (dBSPL) are limited in that they are designed to measure low levels of sound and therefore might underestimate proportions of high and low frequencies at stronger levels, the researchers acknowledge.
Overall, most measures were clustered in the 55-75 decibel range, although some sound levels for incubator handling, while below levels previously reported in the literature, reached approximately 100 decibels.
The noise involved inside the incubator was not perceived as loud by those working with the incubator, the researchers note.
As for resonance inside the incubator, the researchers measured a low-frequency main resonance of 97 Hz, but they write that this resonance can be hard to capture in weighted measurements. However, the resonance means that “noises from the outside sound more tonal inside the incubator, booming and muffled as well as less rough or noisy,” and sounds inside the incubator are similarly affected, the researchers say.
“Most of the noise situations described in this manuscript far exceed not only the recommendation of the AAP but also international guidelines provided by the World Health Organization and the U.S. Environmental Protection Agency,” which recommend, respectively, maximum dBA levels of 35 dBA and 45 dBA for daytime and 30 dBA and 35 dBA for night, the researchers indicate.
Potential long-term implications are that babies who spend time in the NICU are at risk for hearing impairment, which could lead to delays in language acquisition, they say.
The findings were limited by several factors, including the variance among the incubators, which prevents generalizability, the researchers note. Other limitations include the use of a simulation room rather than everyday conditions, in which the environmental sounds would likely be even louder.
However, the results provide insights into the specifics of incubator and NICU noise and suggest that sound be a consideration in the development and promotion of incubators to help protect the hearing of the infants inside them, the researchers conclude.
A generalist’s take
“This is an interesting study looking at the level and character of the sound experienced by preterm infants inside an incubator and how it may compare to sounds experienced within the mother’s womb,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
In society at large, “there has been more focus lately on the general environment and its effect on health, and this study is a unique take on this concept,” he said. “Although in general the incubators work to dampen external sounds, low-frequency sounds may actually resonate more inside the incubators, and taps on the outside or inside of the incubator itself are amplified within the incubator,” he noted. “It is sad but not surprising that the decibel levels experienced by the infants in the incubators exceed the recommended levels recommended by AAP.”
As for additional research, “it would be interesting to see the results of trials looking at various short- or long-term outcomes experienced by infants exposed to a lower-level noise compared to the current levels,” Dr. Joos told this news organization.
A neonatologist’s perspective
“As the field of neonatology advances, we are caring for an ever-growing number of extremely preterm infants,” said Caitlin M. Drumm, MD, of Walter Reed National Military Medical Center, Bethesda, Md., in an interview.
“These infants will spend the first few months of their lives within an incubator in the neonatal intensive care unit, so it is important to understand the potential long-term implications of environmental effects on these vulnerable patients,” she said.
“As in prior studies, it was not surprising that essentially every environmental, handling, or respiratory intervention led to noise levels higher than the limit recommended by the American Academy of Pediatrics,” Dr. Drumm said. “What was surprising was just how high above the 45-dB recommended noise limit many environmental stimuli are. For example, the authors cite respiratory flow rates of 8 L/min or higher as risky for hearing health at 84.72 dBSPL, “ she said.
The key message for clinicians is to be aware of noise levels in the NICU, Dr. Drumm said. “Environmental stimuli as simple as putting a stethoscope on the incubator lead to noise levels well above the limit recommended by the American Academy of Pediatrics. The entire NICU care team has a role to play in minimizing environmental sound hazards for our most critically ill patients.”
Looking ahead, “future research should focus on providing more information correlating neonatal environmental sound exposure to long-term hearing and neurodevelopmental outcomes,” she said.
The study received no outside funding. The researchers report no relevant financial relationships. Dr. Joos serves on the editorial advisory board of Pediatric News. Dr. Drumm has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Incubators save the lives of many babies, but new data suggest that the ambient noise associated with the incubator experience could put babies’ hearing and language development skills at risk.
Previous studies have shown that the neonatal intensive care unit is a noisy environment, but specific data on levels of sound inside and outside incubators are limited, wrote Christoph Reuter, MA, a musicology professor at the University of Vienna, and colleagues.
“By the age of 3 years, deficits in language acquisition are detectable in nearly 50% of very preterm infants,” and high levels of NICU noise have been cited as possible contributors to this increased risk, the researchers say.
In a study published in Frontiers in Pediatrics, the researchers aimed to compare real-life NICU noise with previously reported levels to describe the sound characteristics and to identify resonance characteristics inside an incubator.
The study was conducted at the Pediatric Simulation Center at the Medical University of Vienna. The researchers placed a simulation mannequin with an ear microphone inside an incubator. They also placed microphones outside the incubator to collect measures of outside noise and activity involved in NICU care.
Data regarding sound were collected for 11 environmental noises and 12 incubator handlings using weighted and unweighted decibel levels. Specific environmental noises included starting the incubator engine; environmental noise with incubator off; environmental noise with incubator on; normal conversation; light conversation; laughter; telephone sounds; the infusion pump alarm; the monitor alarm (anomaly); the monitor alarm (emergency); and blood pressure measurement.
The 12 incubator handling noises included those associated with water flap, water pouring into the incubator, incubator doors opening properly, incubators doors closing properly, incubator doors closing improperly, hatch closing, hatch opening, incubator drawer, neighbor incubator doors closing (1.82 m distance), taking a stethoscope from the incubator wall, putting a stethoscope on the incubator, and suctioning tube. Noise from six levels of respiratory support was also measured.
The researchers reported that the incubator tended to dampen most sounds but also that some sounds resonated inside the incubator, which raised the interior noise level by as much as 28 decibels.
Most of the measures using both A-weighted decibels (dBA) and sound pressure level decibels (dBSPL) were above the 45-decibel level for neonatal sound exposure recommended by the American Academy of Pediatrics. The measurements (dBA) versus unweighted (dBSPL) are limited in that they are designed to measure low levels of sound and therefore might underestimate proportions of high and low frequencies at stronger levels, the researchers acknowledge.
Overall, most measures were clustered in the 55-75 decibel range, although some sound levels for incubator handling, while below levels previously reported in the literature, reached approximately 100 decibels.
The noise involved inside the incubator was not perceived as loud by those working with the incubator, the researchers note.
As for resonance inside the incubator, the researchers measured a low-frequency main resonance of 97 Hz, but they write that this resonance can be hard to capture in weighted measurements. However, the resonance means that “noises from the outside sound more tonal inside the incubator, booming and muffled as well as less rough or noisy,” and sounds inside the incubator are similarly affected, the researchers say.
“Most of the noise situations described in this manuscript far exceed not only the recommendation of the AAP but also international guidelines provided by the World Health Organization and the U.S. Environmental Protection Agency,” which recommend, respectively, maximum dBA levels of 35 dBA and 45 dBA for daytime and 30 dBA and 35 dBA for night, the researchers indicate.
Potential long-term implications are that babies who spend time in the NICU are at risk for hearing impairment, which could lead to delays in language acquisition, they say.
The findings were limited by several factors, including the variance among the incubators, which prevents generalizability, the researchers note. Other limitations include the use of a simulation room rather than everyday conditions, in which the environmental sounds would likely be even louder.
However, the results provide insights into the specifics of incubator and NICU noise and suggest that sound be a consideration in the development and promotion of incubators to help protect the hearing of the infants inside them, the researchers conclude.
A generalist’s take
“This is an interesting study looking at the level and character of the sound experienced by preterm infants inside an incubator and how it may compare to sounds experienced within the mother’s womb,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
In society at large, “there has been more focus lately on the general environment and its effect on health, and this study is a unique take on this concept,” he said. “Although in general the incubators work to dampen external sounds, low-frequency sounds may actually resonate more inside the incubators, and taps on the outside or inside of the incubator itself are amplified within the incubator,” he noted. “It is sad but not surprising that the decibel levels experienced by the infants in the incubators exceed the recommended levels recommended by AAP.”
As for additional research, “it would be interesting to see the results of trials looking at various short- or long-term outcomes experienced by infants exposed to a lower-level noise compared to the current levels,” Dr. Joos told this news organization.
A neonatologist’s perspective
“As the field of neonatology advances, we are caring for an ever-growing number of extremely preterm infants,” said Caitlin M. Drumm, MD, of Walter Reed National Military Medical Center, Bethesda, Md., in an interview.
“These infants will spend the first few months of their lives within an incubator in the neonatal intensive care unit, so it is important to understand the potential long-term implications of environmental effects on these vulnerable patients,” she said.
“As in prior studies, it was not surprising that essentially every environmental, handling, or respiratory intervention led to noise levels higher than the limit recommended by the American Academy of Pediatrics,” Dr. Drumm said. “What was surprising was just how high above the 45-dB recommended noise limit many environmental stimuli are. For example, the authors cite respiratory flow rates of 8 L/min or higher as risky for hearing health at 84.72 dBSPL, “ she said.
The key message for clinicians is to be aware of noise levels in the NICU, Dr. Drumm said. “Environmental stimuli as simple as putting a stethoscope on the incubator lead to noise levels well above the limit recommended by the American Academy of Pediatrics. The entire NICU care team has a role to play in minimizing environmental sound hazards for our most critically ill patients.”
Looking ahead, “future research should focus on providing more information correlating neonatal environmental sound exposure to long-term hearing and neurodevelopmental outcomes,” she said.
The study received no outside funding. The researchers report no relevant financial relationships. Dr. Joos serves on the editorial advisory board of Pediatric News. Dr. Drumm has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Incubators save the lives of many babies, but new data suggest that the ambient noise associated with the incubator experience could put babies’ hearing and language development skills at risk.
Previous studies have shown that the neonatal intensive care unit is a noisy environment, but specific data on levels of sound inside and outside incubators are limited, wrote Christoph Reuter, MA, a musicology professor at the University of Vienna, and colleagues.
“By the age of 3 years, deficits in language acquisition are detectable in nearly 50% of very preterm infants,” and high levels of NICU noise have been cited as possible contributors to this increased risk, the researchers say.
In a study published in Frontiers in Pediatrics, the researchers aimed to compare real-life NICU noise with previously reported levels to describe the sound characteristics and to identify resonance characteristics inside an incubator.
The study was conducted at the Pediatric Simulation Center at the Medical University of Vienna. The researchers placed a simulation mannequin with an ear microphone inside an incubator. They also placed microphones outside the incubator to collect measures of outside noise and activity involved in NICU care.
Data regarding sound were collected for 11 environmental noises and 12 incubator handlings using weighted and unweighted decibel levels. Specific environmental noises included starting the incubator engine; environmental noise with incubator off; environmental noise with incubator on; normal conversation; light conversation; laughter; telephone sounds; the infusion pump alarm; the monitor alarm (anomaly); the monitor alarm (emergency); and blood pressure measurement.
The 12 incubator handling noises included those associated with water flap, water pouring into the incubator, incubator doors opening properly, incubators doors closing properly, incubator doors closing improperly, hatch closing, hatch opening, incubator drawer, neighbor incubator doors closing (1.82 m distance), taking a stethoscope from the incubator wall, putting a stethoscope on the incubator, and suctioning tube. Noise from six levels of respiratory support was also measured.
The researchers reported that the incubator tended to dampen most sounds but also that some sounds resonated inside the incubator, which raised the interior noise level by as much as 28 decibels.
Most of the measures using both A-weighted decibels (dBA) and sound pressure level decibels (dBSPL) were above the 45-decibel level for neonatal sound exposure recommended by the American Academy of Pediatrics. The measurements (dBA) versus unweighted (dBSPL) are limited in that they are designed to measure low levels of sound and therefore might underestimate proportions of high and low frequencies at stronger levels, the researchers acknowledge.
Overall, most measures were clustered in the 55-75 decibel range, although some sound levels for incubator handling, while below levels previously reported in the literature, reached approximately 100 decibels.
The noise involved inside the incubator was not perceived as loud by those working with the incubator, the researchers note.
As for resonance inside the incubator, the researchers measured a low-frequency main resonance of 97 Hz, but they write that this resonance can be hard to capture in weighted measurements. However, the resonance means that “noises from the outside sound more tonal inside the incubator, booming and muffled as well as less rough or noisy,” and sounds inside the incubator are similarly affected, the researchers say.
“Most of the noise situations described in this manuscript far exceed not only the recommendation of the AAP but also international guidelines provided by the World Health Organization and the U.S. Environmental Protection Agency,” which recommend, respectively, maximum dBA levels of 35 dBA and 45 dBA for daytime and 30 dBA and 35 dBA for night, the researchers indicate.
Potential long-term implications are that babies who spend time in the NICU are at risk for hearing impairment, which could lead to delays in language acquisition, they say.
The findings were limited by several factors, including the variance among the incubators, which prevents generalizability, the researchers note. Other limitations include the use of a simulation room rather than everyday conditions, in which the environmental sounds would likely be even louder.
However, the results provide insights into the specifics of incubator and NICU noise and suggest that sound be a consideration in the development and promotion of incubators to help protect the hearing of the infants inside them, the researchers conclude.
A generalist’s take
“This is an interesting study looking at the level and character of the sound experienced by preterm infants inside an incubator and how it may compare to sounds experienced within the mother’s womb,” said Tim Joos, MD, a Seattle-based clinician with a combination internal medicine/pediatrics practice, in an interview.
In society at large, “there has been more focus lately on the general environment and its effect on health, and this study is a unique take on this concept,” he said. “Although in general the incubators work to dampen external sounds, low-frequency sounds may actually resonate more inside the incubators, and taps on the outside or inside of the incubator itself are amplified within the incubator,” he noted. “It is sad but not surprising that the decibel levels experienced by the infants in the incubators exceed the recommended levels recommended by AAP.”
As for additional research, “it would be interesting to see the results of trials looking at various short- or long-term outcomes experienced by infants exposed to a lower-level noise compared to the current levels,” Dr. Joos told this news organization.
A neonatologist’s perspective
“As the field of neonatology advances, we are caring for an ever-growing number of extremely preterm infants,” said Caitlin M. Drumm, MD, of Walter Reed National Military Medical Center, Bethesda, Md., in an interview.
“These infants will spend the first few months of their lives within an incubator in the neonatal intensive care unit, so it is important to understand the potential long-term implications of environmental effects on these vulnerable patients,” she said.
“As in prior studies, it was not surprising that essentially every environmental, handling, or respiratory intervention led to noise levels higher than the limit recommended by the American Academy of Pediatrics,” Dr. Drumm said. “What was surprising was just how high above the 45-dB recommended noise limit many environmental stimuli are. For example, the authors cite respiratory flow rates of 8 L/min or higher as risky for hearing health at 84.72 dBSPL, “ she said.
The key message for clinicians is to be aware of noise levels in the NICU, Dr. Drumm said. “Environmental stimuli as simple as putting a stethoscope on the incubator lead to noise levels well above the limit recommended by the American Academy of Pediatrics. The entire NICU care team has a role to play in minimizing environmental sound hazards for our most critically ill patients.”
Looking ahead, “future research should focus on providing more information correlating neonatal environmental sound exposure to long-term hearing and neurodevelopmental outcomes,” she said.
The study received no outside funding. The researchers report no relevant financial relationships. Dr. Joos serves on the editorial advisory board of Pediatric News. Dr. Drumm has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.