User login
ID Practitioner is an independent news source that provides infectious disease specialists with timely and relevant news and commentary about clinical developments and the impact of health care policy on the infectious disease specialist’s practice. Specialty focus topics include antimicrobial resistance, emerging infections, global ID, hepatitis, HIV, hospital-acquired infections, immunizations and vaccines, influenza, mycoses, pediatric infections, and STIs. Infectious Diseases News is owned by Frontline Medical Communications.
sofosbuvir
ritonavir with dasabuvir
discount
support path
program
ritonavir
greedy
ledipasvir
assistance
viekira pak
vpak
advocacy
needy
protest
abbvie
paritaprevir
ombitasvir
direct-acting antivirals
dasabuvir
gilead
fake-ovir
support
v pak
oasis
harvoni
section[contains(@class, 'footer-nav-section-wrapper')]
div[contains(@class, 'pane-pub-article-idp')]
div[contains(@class, 'pane-medstat-latest-articles-articles-section')]
div[contains(@class, 'pane-pub-home-idp')]
div[contains(@class, 'pane-pub-topic-idp')]
When you see something ...
Over the last several decades science has fallen off this country’s radar screen. Yes, STEM (science, technology, engineering, and mathematics) has recently had a brief moment in the spotlight as a buzzword de jour. But the critical importance of careful and systematic investigation into the world around us using observation and trial and error is a tough sell to a large segment of our population.
The COVID-19 pandemic is providing an excellent opportunity for science and medicine to showcase their star qualities. Of course some people in leadership positions persist in disregarding the value of scientific investigation. But I get the feeling that the fear generated by the pandemic is creating some converts among many previous science skeptics. This gathering enthusiasm among the general population is a predictably slow process because that’s the way science works. It often doesn’t provide quick answers. And it is difficult for the nonscientist to see the beauty in the reality that the things we thought were true 2 months ago are likely to be proven wrong today as more observations accumulate.
A recent New York Times article examines the career of one such unscrupulous physician/scientist whose recent exploits threaten to undo much of the positive image the pandemic has cast on science (“The Doctor Behind the Disputed Covid Data,” by Ellen Gabler and Roni Caryn Rabin, The New York Times, July 27, 2020). The subject of the article is the physician who was responsible for providing some of the large data sets on which several papers were published about the apparent ineffectiveness and danger of using hydroxychloroquine in COVID-19 patients. The authenticity of the data sets recently has been seriously questioned, and the articles have been retracted by the journals in which they had appeared.
Based on numerous interviews with coworkers, the Times reporters present a strong case that this individual’s long history of unreliability make his association with allegedly fraudulent data set not surprising but maybe even predictable. At one point in his training, there appears to have been serious questions about advancing the physician to the next level. Despite these concerns, he was allowed to continue and complete his specialty training. It is of note that in his last year of clinical practice, the physician became the subject of three serious malpractice claims that question his competence.
I suspect that some of you have crossed paths with physicians whose competence and/or moral character you found concerning. Were they peers? Were you the individual’s supervisor or was he or she your mentor? How did you respond? Did anyone respond at all?
There has been a lot written and said in recent months about how and when to respond to respond to sexual harassment in the workplace. But I don’t recall reading any articles that discuss how one should respond to incompetence. Of course competency can be a relative term, but in most cases significant incompetence is hard to miss because it tends to be repeated.
It is easy for the airports and subway systems to post signs that say “If you see something say something.” It’s a different story for hospitals and medical schools that may have systems in place for reporting and following up on poor practice. But my sense is that there are too many cases that slip through the cracks.
This is another example of a problem for which I don’t have a solution. However, if this column prompts just one of you who sees something to say something then I have had a good day.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
Over the last several decades science has fallen off this country’s radar screen. Yes, STEM (science, technology, engineering, and mathematics) has recently had a brief moment in the spotlight as a buzzword de jour. But the critical importance of careful and systematic investigation into the world around us using observation and trial and error is a tough sell to a large segment of our population.
The COVID-19 pandemic is providing an excellent opportunity for science and medicine to showcase their star qualities. Of course some people in leadership positions persist in disregarding the value of scientific investigation. But I get the feeling that the fear generated by the pandemic is creating some converts among many previous science skeptics. This gathering enthusiasm among the general population is a predictably slow process because that’s the way science works. It often doesn’t provide quick answers. And it is difficult for the nonscientist to see the beauty in the reality that the things we thought were true 2 months ago are likely to be proven wrong today as more observations accumulate.
A recent New York Times article examines the career of one such unscrupulous physician/scientist whose recent exploits threaten to undo much of the positive image the pandemic has cast on science (“The Doctor Behind the Disputed Covid Data,” by Ellen Gabler and Roni Caryn Rabin, The New York Times, July 27, 2020). The subject of the article is the physician who was responsible for providing some of the large data sets on which several papers were published about the apparent ineffectiveness and danger of using hydroxychloroquine in COVID-19 patients. The authenticity of the data sets recently has been seriously questioned, and the articles have been retracted by the journals in which they had appeared.
Based on numerous interviews with coworkers, the Times reporters present a strong case that this individual’s long history of unreliability make his association with allegedly fraudulent data set not surprising but maybe even predictable. At one point in his training, there appears to have been serious questions about advancing the physician to the next level. Despite these concerns, he was allowed to continue and complete his specialty training. It is of note that in his last year of clinical practice, the physician became the subject of three serious malpractice claims that question his competence.
I suspect that some of you have crossed paths with physicians whose competence and/or moral character you found concerning. Were they peers? Were you the individual’s supervisor or was he or she your mentor? How did you respond? Did anyone respond at all?
There has been a lot written and said in recent months about how and when to respond to respond to sexual harassment in the workplace. But I don’t recall reading any articles that discuss how one should respond to incompetence. Of course competency can be a relative term, but in most cases significant incompetence is hard to miss because it tends to be repeated.
It is easy for the airports and subway systems to post signs that say “If you see something say something.” It’s a different story for hospitals and medical schools that may have systems in place for reporting and following up on poor practice. But my sense is that there are too many cases that slip through the cracks.
This is another example of a problem for which I don’t have a solution. However, if this column prompts just one of you who sees something to say something then I have had a good day.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
Over the last several decades science has fallen off this country’s radar screen. Yes, STEM (science, technology, engineering, and mathematics) has recently had a brief moment in the spotlight as a buzzword de jour. But the critical importance of careful and systematic investigation into the world around us using observation and trial and error is a tough sell to a large segment of our population.
The COVID-19 pandemic is providing an excellent opportunity for science and medicine to showcase their star qualities. Of course some people in leadership positions persist in disregarding the value of scientific investigation. But I get the feeling that the fear generated by the pandemic is creating some converts among many previous science skeptics. This gathering enthusiasm among the general population is a predictably slow process because that’s the way science works. It often doesn’t provide quick answers. And it is difficult for the nonscientist to see the beauty in the reality that the things we thought were true 2 months ago are likely to be proven wrong today as more observations accumulate.
A recent New York Times article examines the career of one such unscrupulous physician/scientist whose recent exploits threaten to undo much of the positive image the pandemic has cast on science (“The Doctor Behind the Disputed Covid Data,” by Ellen Gabler and Roni Caryn Rabin, The New York Times, July 27, 2020). The subject of the article is the physician who was responsible for providing some of the large data sets on which several papers were published about the apparent ineffectiveness and danger of using hydroxychloroquine in COVID-19 patients. The authenticity of the data sets recently has been seriously questioned, and the articles have been retracted by the journals in which they had appeared.
Based on numerous interviews with coworkers, the Times reporters present a strong case that this individual’s long history of unreliability make his association with allegedly fraudulent data set not surprising but maybe even predictable. At one point in his training, there appears to have been serious questions about advancing the physician to the next level. Despite these concerns, he was allowed to continue and complete his specialty training. It is of note that in his last year of clinical practice, the physician became the subject of three serious malpractice claims that question his competence.
I suspect that some of you have crossed paths with physicians whose competence and/or moral character you found concerning. Were they peers? Were you the individual’s supervisor or was he or she your mentor? How did you respond? Did anyone respond at all?
There has been a lot written and said in recent months about how and when to respond to respond to sexual harassment in the workplace. But I don’t recall reading any articles that discuss how one should respond to incompetence. Of course competency can be a relative term, but in most cases significant incompetence is hard to miss because it tends to be repeated.
It is easy for the airports and subway systems to post signs that say “If you see something say something.” It’s a different story for hospitals and medical schools that may have systems in place for reporting and following up on poor practice. But my sense is that there are too many cases that slip through the cracks.
This is another example of a problem for which I don’t have a solution. However, if this column prompts just one of you who sees something to say something then I have had a good day.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
Septicemia first among hospital inpatient costs
according to a recent analysis from the Agency for Healthcare Research and Quality.
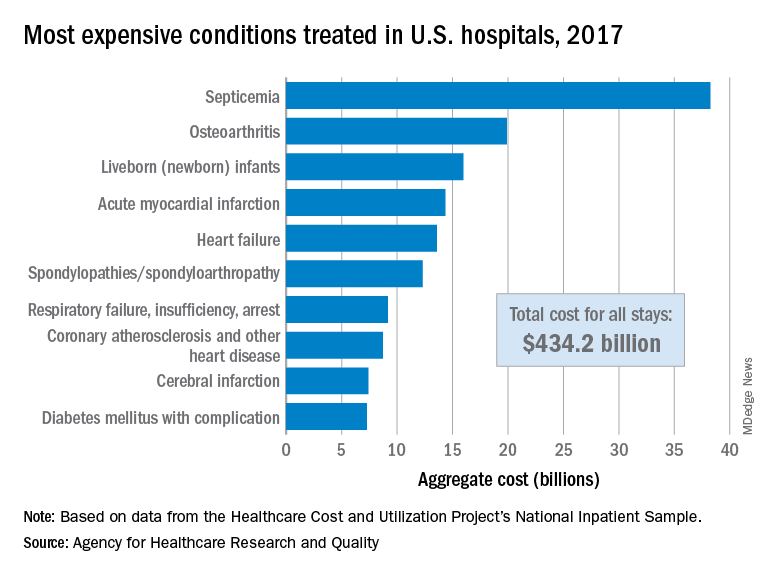
The single most expensive inpatient condition that year, representing about 8.8% of all hospital costs, was septicemia at $38.2 billion, nearly double the $19.9 billion spent on the next most expensive condition, osteoarthritis, Lan Liang, PhD, of the AHRQ, and associates said in a statistical brief.
These figures “represent the hospital’s costs to produce the services – not the amount paid for services by payers – and they do not include separately billed physician fees associated with the hospitalization,” they noted.
Third in overall cost for 2017 but first in total number of stays were live-born infants, with 3.7 million admissions costing just under $16 billion. Hospital costs for acute myocardial infarction ($14.3 billion) made it the fourth most expensive condition, with heart failure fifth at $13.6 billion, based on data from the Healthcare Cost and Utilization Project’s National Inpatient Sample.
The 20 most expensive conditions, which also included coronary atherosclerosis, pneumonia, renal failure, and lower-limb fracture, accounted for close to 47% of all hospital costs and over 43% of all stays in 2017. The total amount spent by hospitals that year, $1.1 trillion, constituted nearly a third of all health care expenditures and was 4.7% higher than in 2016, Dr. Liang and associates reported.
“Although this growth represented deceleration, compared with the 5.8% increase between 2014 and 2015, the consistent year-to-year rise in hospital-related expenses remains a central concern among policymakers,” they wrote.
according to a recent analysis from the Agency for Healthcare Research and Quality.

The single most expensive inpatient condition that year, representing about 8.8% of all hospital costs, was septicemia at $38.2 billion, nearly double the $19.9 billion spent on the next most expensive condition, osteoarthritis, Lan Liang, PhD, of the AHRQ, and associates said in a statistical brief.
These figures “represent the hospital’s costs to produce the services – not the amount paid for services by payers – and they do not include separately billed physician fees associated with the hospitalization,” they noted.
Third in overall cost for 2017 but first in total number of stays were live-born infants, with 3.7 million admissions costing just under $16 billion. Hospital costs for acute myocardial infarction ($14.3 billion) made it the fourth most expensive condition, with heart failure fifth at $13.6 billion, based on data from the Healthcare Cost and Utilization Project’s National Inpatient Sample.
The 20 most expensive conditions, which also included coronary atherosclerosis, pneumonia, renal failure, and lower-limb fracture, accounted for close to 47% of all hospital costs and over 43% of all stays in 2017. The total amount spent by hospitals that year, $1.1 trillion, constituted nearly a third of all health care expenditures and was 4.7% higher than in 2016, Dr. Liang and associates reported.
“Although this growth represented deceleration, compared with the 5.8% increase between 2014 and 2015, the consistent year-to-year rise in hospital-related expenses remains a central concern among policymakers,” they wrote.
according to a recent analysis from the Agency for Healthcare Research and Quality.

The single most expensive inpatient condition that year, representing about 8.8% of all hospital costs, was septicemia at $38.2 billion, nearly double the $19.9 billion spent on the next most expensive condition, osteoarthritis, Lan Liang, PhD, of the AHRQ, and associates said in a statistical brief.
These figures “represent the hospital’s costs to produce the services – not the amount paid for services by payers – and they do not include separately billed physician fees associated with the hospitalization,” they noted.
Third in overall cost for 2017 but first in total number of stays were live-born infants, with 3.7 million admissions costing just under $16 billion. Hospital costs for acute myocardial infarction ($14.3 billion) made it the fourth most expensive condition, with heart failure fifth at $13.6 billion, based on data from the Healthcare Cost and Utilization Project’s National Inpatient Sample.
The 20 most expensive conditions, which also included coronary atherosclerosis, pneumonia, renal failure, and lower-limb fracture, accounted for close to 47% of all hospital costs and over 43% of all stays in 2017. The total amount spent by hospitals that year, $1.1 trillion, constituted nearly a third of all health care expenditures and was 4.7% higher than in 2016, Dr. Liang and associates reported.
“Although this growth represented deceleration, compared with the 5.8% increase between 2014 and 2015, the consistent year-to-year rise in hospital-related expenses remains a central concern among policymakers,” they wrote.
Hepatitis screening now for all patients with cancer on therapy
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
Weight gain persists as HIV-treatment issue
People living with HIV who put on extra pounds and develop metabolic syndrome or related disorders linked in part to certain antiretroviral agents remain a concern today, even as the drugs used to suppress HIV infection have evolved over the decades.
Linkage of HIV treatment with lipodystrophy and insulin resistance or diabetes began in the 1990s with protease inhibitors (Clin Infect Dis. 2000 Jun;30[suppl 2]:s135-42). Several reports over the years also tied any form of effective antiretroviral therapy to weight gain in HIV patients (Antivir Ther. 2012;17[7]:1281-9). More recently, reports have rattled the HIV-treatment community by associating alarmingly high levels of weight gain with a useful and relatively new drug, tenofovir alafenamide fumarate (TAF) – a nucleoside reverse transcriptase inhibitor (NRTI) approved for use in the United States in late 2016, as well as certain agents from an entirely different antiretroviral therapy (ART) class, the integrase strand transfer inhibitors (INSTIs). Both TAF and the INSTIs have come to play major roles in the HIV-treatment landscape, despite relevant and concerning recent weight gain observations with these drugs, such as in a 2019 meta-analysis of eight trials with 5,680 treatment-naive patients who started ART during 2003-2015 (Clin Infect Dis. 2019 Oct 14;doi: 10.1093/cid/ciz999).
“Weight gain is clearly seen in studies of dolutegravir [DTG] or bictegravir [BTG] with TAF,” wrote W.D. Francois Venter, PhD and Andrew Hill, PhD in a recent published commentary on the topic (Lancet HIV. 2020 Jun 1;7[6]:e389-400). Both DTG and BTG are INSTI class members.
“Excessive weight gain, defined as more than 10% over baseline, has recently been observed among people with HIV initiating or switching to regimens incorporating TAF, an INSTI, or both, particularly DTG,” wrote Jordan E. Lake, MD, an HIV specialist at the University of Texas Health Science Center at Houston, in a recent commentary posted online. Women and Black patients “are at even greater risk for excessive weight gain,” Dr. Lake added.
“In recent times, it has emerged that weight gain is more pronounced with the integrase inhibitor class of agents, especially dolutegravir and bictegravir, the so-called second-generation” INSTIs, said Anna Maria Geretti, MD, a professor of clinical infection, microbiology, and immunology at the University of Liverpool, England. ”The effect is more pronounced in women and people of non-White ethnicity, and is of concern because of the associated potential risk of metabolic syndrome, cardiovascular disease, etc.,” Dr. Geretti said in an interview.
The unprecedented susceptibility to weight gain seen recently in non-White women may in part have resulted from the tendency of many earlier treatment trials to have cohorts comprised predominantly of White men, Dr. Venter noted in an interview.
Alarming weight gains reported
Perhaps the most eye-popping example of the potential for weight gain with the combination of TAF with an INSTI came in a recent report from the ADVANCE trial, a randomized, head-to-head comparison of three regimens in 1,053 HIV patients in South Africa. After 144 weeks on a regimen of TAF (Vemlidy), DTG (Tivicay), and FTC (emtricitabine, Emtriva), another NRTI, women gained an averaged of more than 12 kg, compared with their baseline weight, significantly more than in two comparator groups, Simiso Sokhela, MB, reported at the virtual meeting of the International AIDS conference. The women in ADVANCE on the TAF-DTG-FTC regimen also had an 11% rate of incident metabolic syndrome during their first 96 weeks on treatment, compared with rates of 8% among patients on a different form of tenofovir, tenofovir disoproxil fumarate (TDF), along with DTG-FTC, and 5% among those on TDF–EFV (efavirenz, Sustiva)–FTC said Dr. Sokhela, an HIV researcher at Ezintsha, a division of the University of the Witwatersrand in Johannesburg, South Africa.
“We believe that these results support the World Health Organization guidelines that reserve TAF for only patients with osteoporosis or impaired renal function,” Dr. Sokhela said during a press briefing at the conference. The WHO guidelines list the first-line regimen as TDF-DTG-3TC (lamivudine; Epivir) or FTC. “The risk for becoming obese continued to increase after 96 weeks” of chronic use of these drugs, she added.
“All regimens are now brilliant at viral control. Finding the ones that don’t make patients obese or have other long-term side effects is now the priority,” noted Dr. Venter, a professor and HIV researcher at University of the Witwatersrand, head of Ezintsha, and lead investigator of ADVANCE. Clinicians and researchers have recently thought that combining TAF and an INSTI plus FTC or a similar NRTI “would be the ultimate regimen to replace the nonnucleoside reverse transcriptase inhibitors (NNRTIs)” such as EFV, “but now we have a major headache” with unexpectedly high weight gains in some patients, Dr. Venter said.
Weight gains “over 10 kg are unlikely to be acceptable in any circumstances, especially when starting body mass index is already borderline overweight,” wrote Dr. Venter along with Dr. Hill in their commentary. Until recently, many clinicians chalked up weight gain on newly begun ART as a manifestation of the patient’s “return-to-health,” but this interpretation “gives a positive spin to a potentially serious and common side effect,” they added.
More from ADVANCE
The primary efficacy endpoint of ADVANCE was suppression of viral load to less than 50 RNA copies/mL after 48 weeks on treatment, and the result showed that the TAF-DTG-FTC regimen and the TDF-DTG-FTC regimen were each noninferior to the control regimen of TDF-EFV-FTC (New Engl J Med. 2019 Aug 29;381[9]:803-15). Virtually all of the enrolled patients were Black, and 59% were women. Planned follow-up of all patients ran for 96 weeks. After 48 weeks, weight gain among the women averaged 6.4 kg, 3.2 kg, and 1.7 kg in the TAF-DTG, TDF-DTG, and TDF-EFV arms respectively. After 96 weeks, the average weight gains among women were 8.2 kg, 4.6 kg, and 3.2 kg, respectively, in new results reported by Dr. Sokhela at the IAC. Follow-up to 144 weeks was partial and included about a quarter of the enrolled women, with gains averaging 12.3 kg, 7.4 kg, and 5.5 kg respectively. The pattern of weight gain among men tracked the pattern in women, but the magnitude of gain was less. Among men followed for 144 weeks, average gain among those on TAF-DTG-FTC was 7.2 kg, the largest gain seen among men on any regimen and at any follow-up time in the study.
Dr. Sokhela also reported data on body composition analyses, which showed that the weight gains were largely in fat rather than lean tissue, fat accumulation was significantly greater in women than men, and that in both sexes fat accumulated roughly equally in the trunk and on limbs.
An additional analysis looked at the incidence of new-onset obesity among the women who had a normal body mass index at baseline. After 96 weeks, incident obesity occurred in 14% of women on the TAG-DTG-FTC regimen, 8% on TDF-DTG-FTC, and in 2% of women maintained on TDF-EFV-FTC, said Dr. Hill in a separate report at the conference.
Weight starts to weigh in
“I am very mindful of weight gain potential, and I talk to patients about it. It doesn’t determine what regimen I choose for a patient” right now, “but it’s only a matter of time before it starts influencing what we do, particularly if we can achieve efficacy with fewer drugs,” commented Babafemi O. Taiwo, MD, professor of medicine and chief of infectious diseases at Northwestern University in Chicago. “I’ve had some patients show up with a weight gain of 20 kg, and that shouldn’t happen,” he said during a recent online educational session. Dr. Taiwo said his recent practice has been to warn patients about possible weight gain and to urge them to get back in touch with him quickly if it happens.
“Virologic suppression is the most important goal with ART, and the U.S. Department of Health and Human Services currently recommends INSTI-based ART for most PWH [people with HIV],” wrote Dr. Lake in April 2020. “I counsel all PWH initiating ART about the potential for weight gain, and I discuss their current diet and healthy lifestyle habits. I explain to patients that we will monitor their weight, and if weight gain seems more than either of us are comfortable with then we will reassess. Only a small percentage of patients experience excessive weight gain after starting ART.” Dr. Lake also stressed that she had not yet begun to change the regimen a patient is on solely because of weight gain. “We do not know whether this weight gain is reversible,” she noted.
“I do not anticipate that a risk of weight gain at present will dictate a change in guidelines,” said Dr. Geretti. “Drugs such as dolutegravir and bictegravir are very effective, and they are unlikely to cause drug resistance. Further data on the mechanism of weight gain and the reversibility after a change of treatment will help refine drug selection in the near future,” she predicted.
“I consider weight gain when prescribing because my patients hear about this. It’s a side effect that my patients really care about, and I don’t blame them,” said Lisa Hightow-Weidman, MD, a professor and HIV specialist at the University of North Carolina at Chapel Hill, during an on-line educational session. “If you don’t discuss it with a patient and then weight gain happens and the patient finds out [the known risk from their treatment] they may have an issue,” she noted. But weight gain is not a reason to avoid these drugs. “They are great medications in many ways, with once-daily regimens and few side effects.”
Weight gain during pregnancy a special concern
An additional analysis of data from ADVANCE presented at the conference highlighted what the observed weight gain on ART could mean for women who become pregnant while on treatment. Based on a systematic literature review, the ADVANCE investigators calculated the relative risk for six obesity-related pregnancy complications, compared with nonobese women: preterm delivery, gestational diabetes, gestational hypertension, preeclampsia, postpartum hemorrhage, and caesarean delivery. Based on the obesity changes among women on their assigned ART in ADVANCE, the researchers calculated the predicted incidence of these six complications. The analysis showed that for every 1,000 women, those on TAG-DTG-FTC would have an excess of 53 obesity-related pregnancy complications, those on TDF-DTG-FTC would develop 28 excess pregnancy complications, and those on TDG-EFV-FTC would have four excess complications, reported Dr. Hill at the International AIDS conference.
The researchers also ran a similar simulation for the incidence of neonatal complications that could result when mothers are obese because of their ART. The six neonatal complications included in this analysis were small for gestational age, large for gestational age, macrosomia, neonatal death, stillbirth, and neural tube defects. Based on the excess rate of incident obesity, they calculated that for every 1,000 pregnancies women on TAD-DTG-FTC would have 24 additional infants born with one of these complications, women on TDF-DTG-FTC would have an excess of 13 of these events, and women on TDG-EFV-FTC would have an excess of three such obesity-related neonatal complications, Dr. Hill said.
Sorting out the drugs
Results from several additional studies reported at the conference have started trying to discern exactly which ART drugs and regimens pose the greatest weight gain risk and which have the least risk while retaining high efficacy and resistance barriers.
Further evidence implicating any type of ART as a driver of increased weight came from a review of 8,256 adults infected with HIV and members of the Kaiser Permanente health system in three U.S. regions during 2000-2016. Researchers matched these cases using several demographic factors with just under 130,000 members without HIV. Those infected by HIV had half the prevalence of obesity as the matched controls at baseline. During 12 years of follow-up, those infected with HIV had a threefold higher rate of weight gain than those who were uninfected. Annual weight gain averaged 0.06 kg/year among the uninfected people and 0.22 kg/year among those infected with HIV, a statistically significant difference that was consistent regardless of whether people started the study at a normal body mass index, overweight, or obese, reported Michael J. Silverberg, PhD, an epidemiologist with Kaiser Permanente in Oakland, Calif.
Another study tried to focus on the weight gain impact when patients on three-drug ART regimens changed from taking TDF to TAF. This analysis used data collected in the OPERA (Observational Pharmaco-Epidemiology Research & Analysis) longitudinal cohort of about 115,000 U.S. PWH. The observational cohort included nearly 7,000 patients who made a TDF-to-TAF switch, including 3,288 patients who maintained treatment during this switch with an INSTI, 1,454 who maintained a background regimen based on a NNRTI, 1,430 patients who also switched from an INSTI to a different drug, and 747 patients maintained on a boosted dose of a protease inhibitor. All patients were well controlled on their baseline regimen, with at least two consecutive measures showing undetectable viral load.
Patients who maintained their background regimens while changing from TDF to TAF had a 2.0-2.6 kg increase in weight during the 9 months immediately following their switch to TAF, reported Patrick Mallon, MB, a professor of microbial diseases at University College Dublin. Among the patients who both switched to TAF and also switched to treatment with an INSTI, weight gain during the 9 months after the switch averaged 2.6-4.5 kg, depending on which INSTI was started. Patients who switched to treatment with elvitegravir/cobicistat (an INSTI plus a boosting agent) averaged a gain of 2.6 kg during 9 months, those who switched to DTG averaged a 3.1-kg gain, and those who switched to BTG averaged a 4.6-kg increase, Dr. Mallon reported at the conference.
These findings “give us a good sense that the weight gain is real. This is not just overeating or not exercising, but weight changes coincidental with a change in HIV treatment,” commented David Wohl, MD, professor of medicine and site leader of the HIV Prevention and Treatment Clinical Trials Unit at the University of North Carolina at Chapel Hill, during an online educational session.
Contrary to this evidence suggesting a consistent uptick in weight when patients start TAF treatment was a recent report on 629 HIV patients randomized to treatment with TAF-BTG-FTC or abacavir (an NRTI, Ziagen)–DTG-3TC, which found similar weight gains between these two regimens after 144 weeks on treatment (Lancet HIV. 2020 Jun;7[6]:e389-400). This finding had the effect of “strengthening the argument that TAF is simply an innocent bystander” and does not play a central role in weight gain, and supporting the notion that the alternative tenofovir formulation, TDF, differs from TAF by promoting weight loss, Dr. Venter and Dr. Hill suggested in their commentary that accompanied this report.
The new findings from Dr. Mallon raise “serious questions about the way we have moved to TAF as a replacement for TDF, especially because the benefits [from TAF] are for a small subgroup – patients with renal disease or osteoporosis,” Dr. Venter said in an interview. “The question is, will we see weight gain like this” if TAF was combined with a non-INSTI drug? he wondered.
While some study results have suggested a mitigating effect from TDF on weight gain, that wasn’t the case in the AFRICOS (African Cohort Study) study of 1,954 PWH who started treatment with TDF-DTG-FTC (742 patients) or a different three-drug regimen. After a median of 225 days on treatment, those who started on TDF-DTG-FTC had an adjusted, 85% higher rate of developing a high body mass index, compared with patients on a different ART regimen, Julie Ake, MD, reported in a talk at the conference. Her conclusion focused on the possible involvement of DTG: “Consistent with previous reports, dolutegravir was significantly associated with an increased risk of developing high body mass index,” said Dr. Ake, director of the U.S. Military HIV Research Program in Bethesda, Md. and leader of AFRICOS.
A potential workaround to some drugs that cause excessive the weight gain is to just not use them. That was part of the rationale for the TANGO study, which took 741 HIV-infected patients with successful viral suppression on a regimen of TAF-FTC plus one or two additional agents and switched half of them to a TAF-less, two-drug regimen of DTG-FTC. This open-label study’s primary endpoint was noninferiority for viral suppression of the DTG-FTC regimen, compared with patients who stayed on their starting regimen, and the results proved that DTG-FTC was just as effective over 48 weeks for this outcome (Clin Infect Dis. 2020 Jan 6. doi: 10.1093/cid/ciz1243).
At the conference, TANGO’s lead investigator, Jean van Wyk, MD, reported the weight and metabolic effects of the switch. The results showed a similar and small weight gain (on average less than 1 kg) during 48 week follow-up regardless of whether patients remained on their baseline, TAF-containing regimen or switched to DTG-FTC, said Dr. van Wyk, global medical lead for HIV treatment at Viiv Healthcare, the company that markets DTG. About three-quarters of patients in both arms received “boosted” dosages of their drugs, and in this subgroup, patients on DTG-FTC showed statistically significant benefits in several lipid levels, fasting glucose level, and in their degree of insulin resistance. Dr. van Wyk said. These between-group differences were not statistically significant among the “unboosted” patients, and the results failed to show a significant between-group difference in the incidence of metabolic syndrome.
Dr. Venter called these results “exciting,” and noted that he already uses the DTG-FTC two-drug combination “a lot” to treat PWH and renal disease.
A second alternative regimen showcased in a talk at the conference used the three-drug regimen of TDF-FTC plus the NNRTI, DOR (doravirine, Pifeltro). The DRIVE-SHIFT trial enrolled 670 HIV patients with successfully suppressed viral load on conventional regimens who were either switched to TDF-DOR-FTC or maintained on their baseline treatment. After 48 weeks, results confirmed the primary efficacy endpoint of noninferiority for maintenance of suppression with the investigational regimen (J Acquir Immune Defic Syndr. 2019 Aug;81[4]:463-72).
A post-hoc analysis looked at weight changes among these patients after as much as 144 weeks of follow-up. The results showed that patients switched to TDF-DOR-FTC had an average weight increase of 1.2-1.4 kg after more than 2 years on the new regimen, with fewer than 10% of patients having a 10% or greater weight gain with DOR, a “next-generation” NNRTI, reported Princy N. Kumar, MD, professor at Georgetown University and chief of infectious diseases at MedStar Georgetown University Hospital in Washington. “Weight gain was minimal, even over the long term,” she noted.
The tested DOR-based regimen also looks “very exciting,” but the populations it’s been tested have also been largely limited to White men, and limited data exist about the regimen’s performance in pregnant women, commented Dr. Venter. The DRIVE-SHIRT patient cohort was about 85% men, and about three-quarters White.
More weight data needed
HIV-treatment researchers and clinicians seem agreed that weight gain and other metabolic effects from HIV treatment need more assessment and evidence because current data, while suggestive, is also inconclusive.
“Clinical trials are desperately needed to understand the mechanisms of and potential therapeutic options for excessive weight gain on ART,” wrote Dr. Lake in her commentary in April. “While more research is needed,” the new data reported at the virtual International AIDS conference “get us closer to understanding the effects of integrase inhibitors and TAF on weight and the potential metabolic consequences,” she commented as chair of the conference session where these reports occurred.
“Further data on the mechanism of weight gain and its reversibility after a change of treatment will help refine drug selection in the near future,” predicted Dr. Geretti.
“It’s hard to understand physiologically how drugs from such different classes all seem to have weight effects; it’s maddening,” said Dr. Venter. “We need decent studies in all patient populations. That will now be the priority,” he declared. “Patients shouldn’t have to choose” between drugs that most effectively control their HIV infection and drugs that don’t pose a risk for weight gain or metabolic derangements. PWH “should not have to face obesity as their new epidemic,” he wrote with Dr. Hill.
ADVANCE was funded in part by Viiv, the company that markets dolutegravir (Tivicay), and received drugs supplied by Gilead and Viiv. TANGO was sponsored by Viiv. DRIVE-SHIFT was funded by Merck, the company that markets doravirine (Pifeltro). Dr. Lake, Dr. Sokhela, Dr. Ake, and Dr. Kumar had no disclosures, Dr. Venter has received personal fees from Adcock Ingraham, Aspen Healthcare, Johnson and Johnson, Merck, Mylan, Roche, and Viiv. Dr. Hill has received payments from Merck. Dr. Geretti has received honoraria and research funding from Gilead, Jansse, Roche, and Viiv. Dr. Taiwo has had financial relationships with Gilead, Janssen, and Viiv. Dr. Hightow-Weidman has received honoraria from Gilead and Jansse. Dr. Wohl has been a consultant to Gilead, Johnson and Johnson, and Merck. Dr. Silverberg received research funding from Gilead. Dr. Mallon has been an advisor to and speaker on behalf of Bristol-Myers Squibb, Cilag, Gilead, Jansse, Merck Sharp & Dohme, and Viiv. Dr. van Wyk is a Viiv employee.
People living with HIV who put on extra pounds and develop metabolic syndrome or related disorders linked in part to certain antiretroviral agents remain a concern today, even as the drugs used to suppress HIV infection have evolved over the decades.
Linkage of HIV treatment with lipodystrophy and insulin resistance or diabetes began in the 1990s with protease inhibitors (Clin Infect Dis. 2000 Jun;30[suppl 2]:s135-42). Several reports over the years also tied any form of effective antiretroviral therapy to weight gain in HIV patients (Antivir Ther. 2012;17[7]:1281-9). More recently, reports have rattled the HIV-treatment community by associating alarmingly high levels of weight gain with a useful and relatively new drug, tenofovir alafenamide fumarate (TAF) – a nucleoside reverse transcriptase inhibitor (NRTI) approved for use in the United States in late 2016, as well as certain agents from an entirely different antiretroviral therapy (ART) class, the integrase strand transfer inhibitors (INSTIs). Both TAF and the INSTIs have come to play major roles in the HIV-treatment landscape, despite relevant and concerning recent weight gain observations with these drugs, such as in a 2019 meta-analysis of eight trials with 5,680 treatment-naive patients who started ART during 2003-2015 (Clin Infect Dis. 2019 Oct 14;doi: 10.1093/cid/ciz999).
“Weight gain is clearly seen in studies of dolutegravir [DTG] or bictegravir [BTG] with TAF,” wrote W.D. Francois Venter, PhD and Andrew Hill, PhD in a recent published commentary on the topic (Lancet HIV. 2020 Jun 1;7[6]:e389-400). Both DTG and BTG are INSTI class members.
“Excessive weight gain, defined as more than 10% over baseline, has recently been observed among people with HIV initiating or switching to regimens incorporating TAF, an INSTI, or both, particularly DTG,” wrote Jordan E. Lake, MD, an HIV specialist at the University of Texas Health Science Center at Houston, in a recent commentary posted online. Women and Black patients “are at even greater risk for excessive weight gain,” Dr. Lake added.
“In recent times, it has emerged that weight gain is more pronounced with the integrase inhibitor class of agents, especially dolutegravir and bictegravir, the so-called second-generation” INSTIs, said Anna Maria Geretti, MD, a professor of clinical infection, microbiology, and immunology at the University of Liverpool, England. ”The effect is more pronounced in women and people of non-White ethnicity, and is of concern because of the associated potential risk of metabolic syndrome, cardiovascular disease, etc.,” Dr. Geretti said in an interview.
The unprecedented susceptibility to weight gain seen recently in non-White women may in part have resulted from the tendency of many earlier treatment trials to have cohorts comprised predominantly of White men, Dr. Venter noted in an interview.
Alarming weight gains reported
Perhaps the most eye-popping example of the potential for weight gain with the combination of TAF with an INSTI came in a recent report from the ADVANCE trial, a randomized, head-to-head comparison of three regimens in 1,053 HIV patients in South Africa. After 144 weeks on a regimen of TAF (Vemlidy), DTG (Tivicay), and FTC (emtricitabine, Emtriva), another NRTI, women gained an averaged of more than 12 kg, compared with their baseline weight, significantly more than in two comparator groups, Simiso Sokhela, MB, reported at the virtual meeting of the International AIDS conference. The women in ADVANCE on the TAF-DTG-FTC regimen also had an 11% rate of incident metabolic syndrome during their first 96 weeks on treatment, compared with rates of 8% among patients on a different form of tenofovir, tenofovir disoproxil fumarate (TDF), along with DTG-FTC, and 5% among those on TDF–EFV (efavirenz, Sustiva)–FTC said Dr. Sokhela, an HIV researcher at Ezintsha, a division of the University of the Witwatersrand in Johannesburg, South Africa.
“We believe that these results support the World Health Organization guidelines that reserve TAF for only patients with osteoporosis or impaired renal function,” Dr. Sokhela said during a press briefing at the conference. The WHO guidelines list the first-line regimen as TDF-DTG-3TC (lamivudine; Epivir) or FTC. “The risk for becoming obese continued to increase after 96 weeks” of chronic use of these drugs, she added.
“All regimens are now brilliant at viral control. Finding the ones that don’t make patients obese or have other long-term side effects is now the priority,” noted Dr. Venter, a professor and HIV researcher at University of the Witwatersrand, head of Ezintsha, and lead investigator of ADVANCE. Clinicians and researchers have recently thought that combining TAF and an INSTI plus FTC or a similar NRTI “would be the ultimate regimen to replace the nonnucleoside reverse transcriptase inhibitors (NNRTIs)” such as EFV, “but now we have a major headache” with unexpectedly high weight gains in some patients, Dr. Venter said.
Weight gains “over 10 kg are unlikely to be acceptable in any circumstances, especially when starting body mass index is already borderline overweight,” wrote Dr. Venter along with Dr. Hill in their commentary. Until recently, many clinicians chalked up weight gain on newly begun ART as a manifestation of the patient’s “return-to-health,” but this interpretation “gives a positive spin to a potentially serious and common side effect,” they added.
More from ADVANCE
The primary efficacy endpoint of ADVANCE was suppression of viral load to less than 50 RNA copies/mL after 48 weeks on treatment, and the result showed that the TAF-DTG-FTC regimen and the TDF-DTG-FTC regimen were each noninferior to the control regimen of TDF-EFV-FTC (New Engl J Med. 2019 Aug 29;381[9]:803-15). Virtually all of the enrolled patients were Black, and 59% were women. Planned follow-up of all patients ran for 96 weeks. After 48 weeks, weight gain among the women averaged 6.4 kg, 3.2 kg, and 1.7 kg in the TAF-DTG, TDF-DTG, and TDF-EFV arms respectively. After 96 weeks, the average weight gains among women were 8.2 kg, 4.6 kg, and 3.2 kg, respectively, in new results reported by Dr. Sokhela at the IAC. Follow-up to 144 weeks was partial and included about a quarter of the enrolled women, with gains averaging 12.3 kg, 7.4 kg, and 5.5 kg respectively. The pattern of weight gain among men tracked the pattern in women, but the magnitude of gain was less. Among men followed for 144 weeks, average gain among those on TAF-DTG-FTC was 7.2 kg, the largest gain seen among men on any regimen and at any follow-up time in the study.
Dr. Sokhela also reported data on body composition analyses, which showed that the weight gains were largely in fat rather than lean tissue, fat accumulation was significantly greater in women than men, and that in both sexes fat accumulated roughly equally in the trunk and on limbs.
An additional analysis looked at the incidence of new-onset obesity among the women who had a normal body mass index at baseline. After 96 weeks, incident obesity occurred in 14% of women on the TAG-DTG-FTC regimen, 8% on TDF-DTG-FTC, and in 2% of women maintained on TDF-EFV-FTC, said Dr. Hill in a separate report at the conference.
Weight starts to weigh in
“I am very mindful of weight gain potential, and I talk to patients about it. It doesn’t determine what regimen I choose for a patient” right now, “but it’s only a matter of time before it starts influencing what we do, particularly if we can achieve efficacy with fewer drugs,” commented Babafemi O. Taiwo, MD, professor of medicine and chief of infectious diseases at Northwestern University in Chicago. “I’ve had some patients show up with a weight gain of 20 kg, and that shouldn’t happen,” he said during a recent online educational session. Dr. Taiwo said his recent practice has been to warn patients about possible weight gain and to urge them to get back in touch with him quickly if it happens.
“Virologic suppression is the most important goal with ART, and the U.S. Department of Health and Human Services currently recommends INSTI-based ART for most PWH [people with HIV],” wrote Dr. Lake in April 2020. “I counsel all PWH initiating ART about the potential for weight gain, and I discuss their current diet and healthy lifestyle habits. I explain to patients that we will monitor their weight, and if weight gain seems more than either of us are comfortable with then we will reassess. Only a small percentage of patients experience excessive weight gain after starting ART.” Dr. Lake also stressed that she had not yet begun to change the regimen a patient is on solely because of weight gain. “We do not know whether this weight gain is reversible,” she noted.
“I do not anticipate that a risk of weight gain at present will dictate a change in guidelines,” said Dr. Geretti. “Drugs such as dolutegravir and bictegravir are very effective, and they are unlikely to cause drug resistance. Further data on the mechanism of weight gain and the reversibility after a change of treatment will help refine drug selection in the near future,” she predicted.
“I consider weight gain when prescribing because my patients hear about this. It’s a side effect that my patients really care about, and I don’t blame them,” said Lisa Hightow-Weidman, MD, a professor and HIV specialist at the University of North Carolina at Chapel Hill, during an on-line educational session. “If you don’t discuss it with a patient and then weight gain happens and the patient finds out [the known risk from their treatment] they may have an issue,” she noted. But weight gain is not a reason to avoid these drugs. “They are great medications in many ways, with once-daily regimens and few side effects.”
Weight gain during pregnancy a special concern
An additional analysis of data from ADVANCE presented at the conference highlighted what the observed weight gain on ART could mean for women who become pregnant while on treatment. Based on a systematic literature review, the ADVANCE investigators calculated the relative risk for six obesity-related pregnancy complications, compared with nonobese women: preterm delivery, gestational diabetes, gestational hypertension, preeclampsia, postpartum hemorrhage, and caesarean delivery. Based on the obesity changes among women on their assigned ART in ADVANCE, the researchers calculated the predicted incidence of these six complications. The analysis showed that for every 1,000 women, those on TAG-DTG-FTC would have an excess of 53 obesity-related pregnancy complications, those on TDF-DTG-FTC would develop 28 excess pregnancy complications, and those on TDG-EFV-FTC would have four excess complications, reported Dr. Hill at the International AIDS conference.
The researchers also ran a similar simulation for the incidence of neonatal complications that could result when mothers are obese because of their ART. The six neonatal complications included in this analysis were small for gestational age, large for gestational age, macrosomia, neonatal death, stillbirth, and neural tube defects. Based on the excess rate of incident obesity, they calculated that for every 1,000 pregnancies women on TAD-DTG-FTC would have 24 additional infants born with one of these complications, women on TDF-DTG-FTC would have an excess of 13 of these events, and women on TDG-EFV-FTC would have an excess of three such obesity-related neonatal complications, Dr. Hill said.
Sorting out the drugs
Results from several additional studies reported at the conference have started trying to discern exactly which ART drugs and regimens pose the greatest weight gain risk and which have the least risk while retaining high efficacy and resistance barriers.
Further evidence implicating any type of ART as a driver of increased weight came from a review of 8,256 adults infected with HIV and members of the Kaiser Permanente health system in three U.S. regions during 2000-2016. Researchers matched these cases using several demographic factors with just under 130,000 members without HIV. Those infected by HIV had half the prevalence of obesity as the matched controls at baseline. During 12 years of follow-up, those infected with HIV had a threefold higher rate of weight gain than those who were uninfected. Annual weight gain averaged 0.06 kg/year among the uninfected people and 0.22 kg/year among those infected with HIV, a statistically significant difference that was consistent regardless of whether people started the study at a normal body mass index, overweight, or obese, reported Michael J. Silverberg, PhD, an epidemiologist with Kaiser Permanente in Oakland, Calif.
Another study tried to focus on the weight gain impact when patients on three-drug ART regimens changed from taking TDF to TAF. This analysis used data collected in the OPERA (Observational Pharmaco-Epidemiology Research & Analysis) longitudinal cohort of about 115,000 U.S. PWH. The observational cohort included nearly 7,000 patients who made a TDF-to-TAF switch, including 3,288 patients who maintained treatment during this switch with an INSTI, 1,454 who maintained a background regimen based on a NNRTI, 1,430 patients who also switched from an INSTI to a different drug, and 747 patients maintained on a boosted dose of a protease inhibitor. All patients were well controlled on their baseline regimen, with at least two consecutive measures showing undetectable viral load.
Patients who maintained their background regimens while changing from TDF to TAF had a 2.0-2.6 kg increase in weight during the 9 months immediately following their switch to TAF, reported Patrick Mallon, MB, a professor of microbial diseases at University College Dublin. Among the patients who both switched to TAF and also switched to treatment with an INSTI, weight gain during the 9 months after the switch averaged 2.6-4.5 kg, depending on which INSTI was started. Patients who switched to treatment with elvitegravir/cobicistat (an INSTI plus a boosting agent) averaged a gain of 2.6 kg during 9 months, those who switched to DTG averaged a 3.1-kg gain, and those who switched to BTG averaged a 4.6-kg increase, Dr. Mallon reported at the conference.
These findings “give us a good sense that the weight gain is real. This is not just overeating or not exercising, but weight changes coincidental with a change in HIV treatment,” commented David Wohl, MD, professor of medicine and site leader of the HIV Prevention and Treatment Clinical Trials Unit at the University of North Carolina at Chapel Hill, during an online educational session.
Contrary to this evidence suggesting a consistent uptick in weight when patients start TAF treatment was a recent report on 629 HIV patients randomized to treatment with TAF-BTG-FTC or abacavir (an NRTI, Ziagen)–DTG-3TC, which found similar weight gains between these two regimens after 144 weeks on treatment (Lancet HIV. 2020 Jun;7[6]:e389-400). This finding had the effect of “strengthening the argument that TAF is simply an innocent bystander” and does not play a central role in weight gain, and supporting the notion that the alternative tenofovir formulation, TDF, differs from TAF by promoting weight loss, Dr. Venter and Dr. Hill suggested in their commentary that accompanied this report.
The new findings from Dr. Mallon raise “serious questions about the way we have moved to TAF as a replacement for TDF, especially because the benefits [from TAF] are for a small subgroup – patients with renal disease or osteoporosis,” Dr. Venter said in an interview. “The question is, will we see weight gain like this” if TAF was combined with a non-INSTI drug? he wondered.
While some study results have suggested a mitigating effect from TDF on weight gain, that wasn’t the case in the AFRICOS (African Cohort Study) study of 1,954 PWH who started treatment with TDF-DTG-FTC (742 patients) or a different three-drug regimen. After a median of 225 days on treatment, those who started on TDF-DTG-FTC had an adjusted, 85% higher rate of developing a high body mass index, compared with patients on a different ART regimen, Julie Ake, MD, reported in a talk at the conference. Her conclusion focused on the possible involvement of DTG: “Consistent with previous reports, dolutegravir was significantly associated with an increased risk of developing high body mass index,” said Dr. Ake, director of the U.S. Military HIV Research Program in Bethesda, Md. and leader of AFRICOS.
A potential workaround to some drugs that cause excessive the weight gain is to just not use them. That was part of the rationale for the TANGO study, which took 741 HIV-infected patients with successful viral suppression on a regimen of TAF-FTC plus one or two additional agents and switched half of them to a TAF-less, two-drug regimen of DTG-FTC. This open-label study’s primary endpoint was noninferiority for viral suppression of the DTG-FTC regimen, compared with patients who stayed on their starting regimen, and the results proved that DTG-FTC was just as effective over 48 weeks for this outcome (Clin Infect Dis. 2020 Jan 6. doi: 10.1093/cid/ciz1243).
At the conference, TANGO’s lead investigator, Jean van Wyk, MD, reported the weight and metabolic effects of the switch. The results showed a similar and small weight gain (on average less than 1 kg) during 48 week follow-up regardless of whether patients remained on their baseline, TAF-containing regimen or switched to DTG-FTC, said Dr. van Wyk, global medical lead for HIV treatment at Viiv Healthcare, the company that markets DTG. About three-quarters of patients in both arms received “boosted” dosages of their drugs, and in this subgroup, patients on DTG-FTC showed statistically significant benefits in several lipid levels, fasting glucose level, and in their degree of insulin resistance. Dr. van Wyk said. These between-group differences were not statistically significant among the “unboosted” patients, and the results failed to show a significant between-group difference in the incidence of metabolic syndrome.
Dr. Venter called these results “exciting,” and noted that he already uses the DTG-FTC two-drug combination “a lot” to treat PWH and renal disease.
A second alternative regimen showcased in a talk at the conference used the three-drug regimen of TDF-FTC plus the NNRTI, DOR (doravirine, Pifeltro). The DRIVE-SHIFT trial enrolled 670 HIV patients with successfully suppressed viral load on conventional regimens who were either switched to TDF-DOR-FTC or maintained on their baseline treatment. After 48 weeks, results confirmed the primary efficacy endpoint of noninferiority for maintenance of suppression with the investigational regimen (J Acquir Immune Defic Syndr. 2019 Aug;81[4]:463-72).
A post-hoc analysis looked at weight changes among these patients after as much as 144 weeks of follow-up. The results showed that patients switched to TDF-DOR-FTC had an average weight increase of 1.2-1.4 kg after more than 2 years on the new regimen, with fewer than 10% of patients having a 10% or greater weight gain with DOR, a “next-generation” NNRTI, reported Princy N. Kumar, MD, professor at Georgetown University and chief of infectious diseases at MedStar Georgetown University Hospital in Washington. “Weight gain was minimal, even over the long term,” she noted.
The tested DOR-based regimen also looks “very exciting,” but the populations it’s been tested have also been largely limited to White men, and limited data exist about the regimen’s performance in pregnant women, commented Dr. Venter. The DRIVE-SHIRT patient cohort was about 85% men, and about three-quarters White.
More weight data needed
HIV-treatment researchers and clinicians seem agreed that weight gain and other metabolic effects from HIV treatment need more assessment and evidence because current data, while suggestive, is also inconclusive.
“Clinical trials are desperately needed to understand the mechanisms of and potential therapeutic options for excessive weight gain on ART,” wrote Dr. Lake in her commentary in April. “While more research is needed,” the new data reported at the virtual International AIDS conference “get us closer to understanding the effects of integrase inhibitors and TAF on weight and the potential metabolic consequences,” she commented as chair of the conference session where these reports occurred.
“Further data on the mechanism of weight gain and its reversibility after a change of treatment will help refine drug selection in the near future,” predicted Dr. Geretti.
“It’s hard to understand physiologically how drugs from such different classes all seem to have weight effects; it’s maddening,” said Dr. Venter. “We need decent studies in all patient populations. That will now be the priority,” he declared. “Patients shouldn’t have to choose” between drugs that most effectively control their HIV infection and drugs that don’t pose a risk for weight gain or metabolic derangements. PWH “should not have to face obesity as their new epidemic,” he wrote with Dr. Hill.
ADVANCE was funded in part by Viiv, the company that markets dolutegravir (Tivicay), and received drugs supplied by Gilead and Viiv. TANGO was sponsored by Viiv. DRIVE-SHIFT was funded by Merck, the company that markets doravirine (Pifeltro). Dr. Lake, Dr. Sokhela, Dr. Ake, and Dr. Kumar had no disclosures, Dr. Venter has received personal fees from Adcock Ingraham, Aspen Healthcare, Johnson and Johnson, Merck, Mylan, Roche, and Viiv. Dr. Hill has received payments from Merck. Dr. Geretti has received honoraria and research funding from Gilead, Jansse, Roche, and Viiv. Dr. Taiwo has had financial relationships with Gilead, Janssen, and Viiv. Dr. Hightow-Weidman has received honoraria from Gilead and Jansse. Dr. Wohl has been a consultant to Gilead, Johnson and Johnson, and Merck. Dr. Silverberg received research funding from Gilead. Dr. Mallon has been an advisor to and speaker on behalf of Bristol-Myers Squibb, Cilag, Gilead, Jansse, Merck Sharp & Dohme, and Viiv. Dr. van Wyk is a Viiv employee.
People living with HIV who put on extra pounds and develop metabolic syndrome or related disorders linked in part to certain antiretroviral agents remain a concern today, even as the drugs used to suppress HIV infection have evolved over the decades.
Linkage of HIV treatment with lipodystrophy and insulin resistance or diabetes began in the 1990s with protease inhibitors (Clin Infect Dis. 2000 Jun;30[suppl 2]:s135-42). Several reports over the years also tied any form of effective antiretroviral therapy to weight gain in HIV patients (Antivir Ther. 2012;17[7]:1281-9). More recently, reports have rattled the HIV-treatment community by associating alarmingly high levels of weight gain with a useful and relatively new drug, tenofovir alafenamide fumarate (TAF) – a nucleoside reverse transcriptase inhibitor (NRTI) approved for use in the United States in late 2016, as well as certain agents from an entirely different antiretroviral therapy (ART) class, the integrase strand transfer inhibitors (INSTIs). Both TAF and the INSTIs have come to play major roles in the HIV-treatment landscape, despite relevant and concerning recent weight gain observations with these drugs, such as in a 2019 meta-analysis of eight trials with 5,680 treatment-naive patients who started ART during 2003-2015 (Clin Infect Dis. 2019 Oct 14;doi: 10.1093/cid/ciz999).
“Weight gain is clearly seen in studies of dolutegravir [DTG] or bictegravir [BTG] with TAF,” wrote W.D. Francois Venter, PhD and Andrew Hill, PhD in a recent published commentary on the topic (Lancet HIV. 2020 Jun 1;7[6]:e389-400). Both DTG and BTG are INSTI class members.
“Excessive weight gain, defined as more than 10% over baseline, has recently been observed among people with HIV initiating or switching to regimens incorporating TAF, an INSTI, or both, particularly DTG,” wrote Jordan E. Lake, MD, an HIV specialist at the University of Texas Health Science Center at Houston, in a recent commentary posted online. Women and Black patients “are at even greater risk for excessive weight gain,” Dr. Lake added.
“In recent times, it has emerged that weight gain is more pronounced with the integrase inhibitor class of agents, especially dolutegravir and bictegravir, the so-called second-generation” INSTIs, said Anna Maria Geretti, MD, a professor of clinical infection, microbiology, and immunology at the University of Liverpool, England. ”The effect is more pronounced in women and people of non-White ethnicity, and is of concern because of the associated potential risk of metabolic syndrome, cardiovascular disease, etc.,” Dr. Geretti said in an interview.
The unprecedented susceptibility to weight gain seen recently in non-White women may in part have resulted from the tendency of many earlier treatment trials to have cohorts comprised predominantly of White men, Dr. Venter noted in an interview.
Alarming weight gains reported
Perhaps the most eye-popping example of the potential for weight gain with the combination of TAF with an INSTI came in a recent report from the ADVANCE trial, a randomized, head-to-head comparison of three regimens in 1,053 HIV patients in South Africa. After 144 weeks on a regimen of TAF (Vemlidy), DTG (Tivicay), and FTC (emtricitabine, Emtriva), another NRTI, women gained an averaged of more than 12 kg, compared with their baseline weight, significantly more than in two comparator groups, Simiso Sokhela, MB, reported at the virtual meeting of the International AIDS conference. The women in ADVANCE on the TAF-DTG-FTC regimen also had an 11% rate of incident metabolic syndrome during their first 96 weeks on treatment, compared with rates of 8% among patients on a different form of tenofovir, tenofovir disoproxil fumarate (TDF), along with DTG-FTC, and 5% among those on TDF–EFV (efavirenz, Sustiva)–FTC said Dr. Sokhela, an HIV researcher at Ezintsha, a division of the University of the Witwatersrand in Johannesburg, South Africa.
“We believe that these results support the World Health Organization guidelines that reserve TAF for only patients with osteoporosis or impaired renal function,” Dr. Sokhela said during a press briefing at the conference. The WHO guidelines list the first-line regimen as TDF-DTG-3TC (lamivudine; Epivir) or FTC. “The risk for becoming obese continued to increase after 96 weeks” of chronic use of these drugs, she added.
“All regimens are now brilliant at viral control. Finding the ones that don’t make patients obese or have other long-term side effects is now the priority,” noted Dr. Venter, a professor and HIV researcher at University of the Witwatersrand, head of Ezintsha, and lead investigator of ADVANCE. Clinicians and researchers have recently thought that combining TAF and an INSTI plus FTC or a similar NRTI “would be the ultimate regimen to replace the nonnucleoside reverse transcriptase inhibitors (NNRTIs)” such as EFV, “but now we have a major headache” with unexpectedly high weight gains in some patients, Dr. Venter said.
Weight gains “over 10 kg are unlikely to be acceptable in any circumstances, especially when starting body mass index is already borderline overweight,” wrote Dr. Venter along with Dr. Hill in their commentary. Until recently, many clinicians chalked up weight gain on newly begun ART as a manifestation of the patient’s “return-to-health,” but this interpretation “gives a positive spin to a potentially serious and common side effect,” they added.
More from ADVANCE
The primary efficacy endpoint of ADVANCE was suppression of viral load to less than 50 RNA copies/mL after 48 weeks on treatment, and the result showed that the TAF-DTG-FTC regimen and the TDF-DTG-FTC regimen were each noninferior to the control regimen of TDF-EFV-FTC (New Engl J Med. 2019 Aug 29;381[9]:803-15). Virtually all of the enrolled patients were Black, and 59% were women. Planned follow-up of all patients ran for 96 weeks. After 48 weeks, weight gain among the women averaged 6.4 kg, 3.2 kg, and 1.7 kg in the TAF-DTG, TDF-DTG, and TDF-EFV arms respectively. After 96 weeks, the average weight gains among women were 8.2 kg, 4.6 kg, and 3.2 kg, respectively, in new results reported by Dr. Sokhela at the IAC. Follow-up to 144 weeks was partial and included about a quarter of the enrolled women, with gains averaging 12.3 kg, 7.4 kg, and 5.5 kg respectively. The pattern of weight gain among men tracked the pattern in women, but the magnitude of gain was less. Among men followed for 144 weeks, average gain among those on TAF-DTG-FTC was 7.2 kg, the largest gain seen among men on any regimen and at any follow-up time in the study.
Dr. Sokhela also reported data on body composition analyses, which showed that the weight gains were largely in fat rather than lean tissue, fat accumulation was significantly greater in women than men, and that in both sexes fat accumulated roughly equally in the trunk and on limbs.
An additional analysis looked at the incidence of new-onset obesity among the women who had a normal body mass index at baseline. After 96 weeks, incident obesity occurred in 14% of women on the TAG-DTG-FTC regimen, 8% on TDF-DTG-FTC, and in 2% of women maintained on TDF-EFV-FTC, said Dr. Hill in a separate report at the conference.
Weight starts to weigh in
“I am very mindful of weight gain potential, and I talk to patients about it. It doesn’t determine what regimen I choose for a patient” right now, “but it’s only a matter of time before it starts influencing what we do, particularly if we can achieve efficacy with fewer drugs,” commented Babafemi O. Taiwo, MD, professor of medicine and chief of infectious diseases at Northwestern University in Chicago. “I’ve had some patients show up with a weight gain of 20 kg, and that shouldn’t happen,” he said during a recent online educational session. Dr. Taiwo said his recent practice has been to warn patients about possible weight gain and to urge them to get back in touch with him quickly if it happens.
“Virologic suppression is the most important goal with ART, and the U.S. Department of Health and Human Services currently recommends INSTI-based ART for most PWH [people with HIV],” wrote Dr. Lake in April 2020. “I counsel all PWH initiating ART about the potential for weight gain, and I discuss their current diet and healthy lifestyle habits. I explain to patients that we will monitor their weight, and if weight gain seems more than either of us are comfortable with then we will reassess. Only a small percentage of patients experience excessive weight gain after starting ART.” Dr. Lake also stressed that she had not yet begun to change the regimen a patient is on solely because of weight gain. “We do not know whether this weight gain is reversible,” she noted.
“I do not anticipate that a risk of weight gain at present will dictate a change in guidelines,” said Dr. Geretti. “Drugs such as dolutegravir and bictegravir are very effective, and they are unlikely to cause drug resistance. Further data on the mechanism of weight gain and the reversibility after a change of treatment will help refine drug selection in the near future,” she predicted.
“I consider weight gain when prescribing because my patients hear about this. It’s a side effect that my patients really care about, and I don’t blame them,” said Lisa Hightow-Weidman, MD, a professor and HIV specialist at the University of North Carolina at Chapel Hill, during an on-line educational session. “If you don’t discuss it with a patient and then weight gain happens and the patient finds out [the known risk from their treatment] they may have an issue,” she noted. But weight gain is not a reason to avoid these drugs. “They are great medications in many ways, with once-daily regimens and few side effects.”
Weight gain during pregnancy a special concern
An additional analysis of data from ADVANCE presented at the conference highlighted what the observed weight gain on ART could mean for women who become pregnant while on treatment. Based on a systematic literature review, the ADVANCE investigators calculated the relative risk for six obesity-related pregnancy complications, compared with nonobese women: preterm delivery, gestational diabetes, gestational hypertension, preeclampsia, postpartum hemorrhage, and caesarean delivery. Based on the obesity changes among women on their assigned ART in ADVANCE, the researchers calculated the predicted incidence of these six complications. The analysis showed that for every 1,000 women, those on TAG-DTG-FTC would have an excess of 53 obesity-related pregnancy complications, those on TDF-DTG-FTC would develop 28 excess pregnancy complications, and those on TDG-EFV-FTC would have four excess complications, reported Dr. Hill at the International AIDS conference.
The researchers also ran a similar simulation for the incidence of neonatal complications that could result when mothers are obese because of their ART. The six neonatal complications included in this analysis were small for gestational age, large for gestational age, macrosomia, neonatal death, stillbirth, and neural tube defects. Based on the excess rate of incident obesity, they calculated that for every 1,000 pregnancies women on TAD-DTG-FTC would have 24 additional infants born with one of these complications, women on TDF-DTG-FTC would have an excess of 13 of these events, and women on TDG-EFV-FTC would have an excess of three such obesity-related neonatal complications, Dr. Hill said.
Sorting out the drugs
Results from several additional studies reported at the conference have started trying to discern exactly which ART drugs and regimens pose the greatest weight gain risk and which have the least risk while retaining high efficacy and resistance barriers.
Further evidence implicating any type of ART as a driver of increased weight came from a review of 8,256 adults infected with HIV and members of the Kaiser Permanente health system in three U.S. regions during 2000-2016. Researchers matched these cases using several demographic factors with just under 130,000 members without HIV. Those infected by HIV had half the prevalence of obesity as the matched controls at baseline. During 12 years of follow-up, those infected with HIV had a threefold higher rate of weight gain than those who were uninfected. Annual weight gain averaged 0.06 kg/year among the uninfected people and 0.22 kg/year among those infected with HIV, a statistically significant difference that was consistent regardless of whether people started the study at a normal body mass index, overweight, or obese, reported Michael J. Silverberg, PhD, an epidemiologist with Kaiser Permanente in Oakland, Calif.
Another study tried to focus on the weight gain impact when patients on three-drug ART regimens changed from taking TDF to TAF. This analysis used data collected in the OPERA (Observational Pharmaco-Epidemiology Research & Analysis) longitudinal cohort of about 115,000 U.S. PWH. The observational cohort included nearly 7,000 patients who made a TDF-to-TAF switch, including 3,288 patients who maintained treatment during this switch with an INSTI, 1,454 who maintained a background regimen based on a NNRTI, 1,430 patients who also switched from an INSTI to a different drug, and 747 patients maintained on a boosted dose of a protease inhibitor. All patients were well controlled on their baseline regimen, with at least two consecutive measures showing undetectable viral load.
Patients who maintained their background regimens while changing from TDF to TAF had a 2.0-2.6 kg increase in weight during the 9 months immediately following their switch to TAF, reported Patrick Mallon, MB, a professor of microbial diseases at University College Dublin. Among the patients who both switched to TAF and also switched to treatment with an INSTI, weight gain during the 9 months after the switch averaged 2.6-4.5 kg, depending on which INSTI was started. Patients who switched to treatment with elvitegravir/cobicistat (an INSTI plus a boosting agent) averaged a gain of 2.6 kg during 9 months, those who switched to DTG averaged a 3.1-kg gain, and those who switched to BTG averaged a 4.6-kg increase, Dr. Mallon reported at the conference.
These findings “give us a good sense that the weight gain is real. This is not just overeating or not exercising, but weight changes coincidental with a change in HIV treatment,” commented David Wohl, MD, professor of medicine and site leader of the HIV Prevention and Treatment Clinical Trials Unit at the University of North Carolina at Chapel Hill, during an online educational session.
Contrary to this evidence suggesting a consistent uptick in weight when patients start TAF treatment was a recent report on 629 HIV patients randomized to treatment with TAF-BTG-FTC or abacavir (an NRTI, Ziagen)–DTG-3TC, which found similar weight gains between these two regimens after 144 weeks on treatment (Lancet HIV. 2020 Jun;7[6]:e389-400). This finding had the effect of “strengthening the argument that TAF is simply an innocent bystander” and does not play a central role in weight gain, and supporting the notion that the alternative tenofovir formulation, TDF, differs from TAF by promoting weight loss, Dr. Venter and Dr. Hill suggested in their commentary that accompanied this report.
The new findings from Dr. Mallon raise “serious questions about the way we have moved to TAF as a replacement for TDF, especially because the benefits [from TAF] are for a small subgroup – patients with renal disease or osteoporosis,” Dr. Venter said in an interview. “The question is, will we see weight gain like this” if TAF was combined with a non-INSTI drug? he wondered.
While some study results have suggested a mitigating effect from TDF on weight gain, that wasn’t the case in the AFRICOS (African Cohort Study) study of 1,954 PWH who started treatment with TDF-DTG-FTC (742 patients) or a different three-drug regimen. After a median of 225 days on treatment, those who started on TDF-DTG-FTC had an adjusted, 85% higher rate of developing a high body mass index, compared with patients on a different ART regimen, Julie Ake, MD, reported in a talk at the conference. Her conclusion focused on the possible involvement of DTG: “Consistent with previous reports, dolutegravir was significantly associated with an increased risk of developing high body mass index,” said Dr. Ake, director of the U.S. Military HIV Research Program in Bethesda, Md. and leader of AFRICOS.
A potential workaround to some drugs that cause excessive the weight gain is to just not use them. That was part of the rationale for the TANGO study, which took 741 HIV-infected patients with successful viral suppression on a regimen of TAF-FTC plus one or two additional agents and switched half of them to a TAF-less, two-drug regimen of DTG-FTC. This open-label study’s primary endpoint was noninferiority for viral suppression of the DTG-FTC regimen, compared with patients who stayed on their starting regimen, and the results proved that DTG-FTC was just as effective over 48 weeks for this outcome (Clin Infect Dis. 2020 Jan 6. doi: 10.1093/cid/ciz1243).
At the conference, TANGO’s lead investigator, Jean van Wyk, MD, reported the weight and metabolic effects of the switch. The results showed a similar and small weight gain (on average less than 1 kg) during 48 week follow-up regardless of whether patients remained on their baseline, TAF-containing regimen or switched to DTG-FTC, said Dr. van Wyk, global medical lead for HIV treatment at Viiv Healthcare, the company that markets DTG. About three-quarters of patients in both arms received “boosted” dosages of their drugs, and in this subgroup, patients on DTG-FTC showed statistically significant benefits in several lipid levels, fasting glucose level, and in their degree of insulin resistance. Dr. van Wyk said. These between-group differences were not statistically significant among the “unboosted” patients, and the results failed to show a significant between-group difference in the incidence of metabolic syndrome.
Dr. Venter called these results “exciting,” and noted that he already uses the DTG-FTC two-drug combination “a lot” to treat PWH and renal disease.
A second alternative regimen showcased in a talk at the conference used the three-drug regimen of TDF-FTC plus the NNRTI, DOR (doravirine, Pifeltro). The DRIVE-SHIFT trial enrolled 670 HIV patients with successfully suppressed viral load on conventional regimens who were either switched to TDF-DOR-FTC or maintained on their baseline treatment. After 48 weeks, results confirmed the primary efficacy endpoint of noninferiority for maintenance of suppression with the investigational regimen (J Acquir Immune Defic Syndr. 2019 Aug;81[4]:463-72).
A post-hoc analysis looked at weight changes among these patients after as much as 144 weeks of follow-up. The results showed that patients switched to TDF-DOR-FTC had an average weight increase of 1.2-1.4 kg after more than 2 years on the new regimen, with fewer than 10% of patients having a 10% or greater weight gain with DOR, a “next-generation” NNRTI, reported Princy N. Kumar, MD, professor at Georgetown University and chief of infectious diseases at MedStar Georgetown University Hospital in Washington. “Weight gain was minimal, even over the long term,” she noted.
The tested DOR-based regimen also looks “very exciting,” but the populations it’s been tested have also been largely limited to White men, and limited data exist about the regimen’s performance in pregnant women, commented Dr. Venter. The DRIVE-SHIRT patient cohort was about 85% men, and about three-quarters White.
More weight data needed
HIV-treatment researchers and clinicians seem agreed that weight gain and other metabolic effects from HIV treatment need more assessment and evidence because current data, while suggestive, is also inconclusive.
“Clinical trials are desperately needed to understand the mechanisms of and potential therapeutic options for excessive weight gain on ART,” wrote Dr. Lake in her commentary in April. “While more research is needed,” the new data reported at the virtual International AIDS conference “get us closer to understanding the effects of integrase inhibitors and TAF on weight and the potential metabolic consequences,” she commented as chair of the conference session where these reports occurred.
“Further data on the mechanism of weight gain and its reversibility after a change of treatment will help refine drug selection in the near future,” predicted Dr. Geretti.
“It’s hard to understand physiologically how drugs from such different classes all seem to have weight effects; it’s maddening,” said Dr. Venter. “We need decent studies in all patient populations. That will now be the priority,” he declared. “Patients shouldn’t have to choose” between drugs that most effectively control their HIV infection and drugs that don’t pose a risk for weight gain or metabolic derangements. PWH “should not have to face obesity as their new epidemic,” he wrote with Dr. Hill.
ADVANCE was funded in part by Viiv, the company that markets dolutegravir (Tivicay), and received drugs supplied by Gilead and Viiv. TANGO was sponsored by Viiv. DRIVE-SHIFT was funded by Merck, the company that markets doravirine (Pifeltro). Dr. Lake, Dr. Sokhela, Dr. Ake, and Dr. Kumar had no disclosures, Dr. Venter has received personal fees from Adcock Ingraham, Aspen Healthcare, Johnson and Johnson, Merck, Mylan, Roche, and Viiv. Dr. Hill has received payments from Merck. Dr. Geretti has received honoraria and research funding from Gilead, Jansse, Roche, and Viiv. Dr. Taiwo has had financial relationships with Gilead, Janssen, and Viiv. Dr. Hightow-Weidman has received honoraria from Gilead and Jansse. Dr. Wohl has been a consultant to Gilead, Johnson and Johnson, and Merck. Dr. Silverberg received research funding from Gilead. Dr. Mallon has been an advisor to and speaker on behalf of Bristol-Myers Squibb, Cilag, Gilead, Jansse, Merck Sharp & Dohme, and Viiv. Dr. van Wyk is a Viiv employee.
FROM AIDS 2020
Value of palliative care shines clearly in a crisis
Hospitalists have played a key role
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
Hospitalists have played a key role
Hospitalists have played a key role
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
For some palliative care professionals, the COVID-19 pandemic, particularly in viral hot spots like New York City, represents a “moment” that could lead to greater awareness of what this service offers to seriously ill patients in a crisis.
They say it has provided an opportunity to show what palliative care teams can contribute to the difficult circumstances of patients with severe symptoms, isolated and alone in quarantined hospitals, with poor survival rates, perhaps sedated for extended stays on scarce ventilators – and for their family members, who are able to visit them only virtually via telephone or tablet.
But it has also highlighted gaps – including insufficient staffing for some palliative care teams. Hospitalists and other clinicians in the hospital need to learn the basics of primary palliative care, such as how to communicate bad news, initiate goals of care conversations, and address common symptoms of serious illness, such as pain. That way, they could shoulder more of the demand for this kind of care when palliative care specialists are in short supply.
Hospitalists, some of whom also have pursued a specialization in palliative care, have played key roles in clarifying and redefining the new role for palliative care, whom it is meant for, and who should provide it. Central to this new role is the greater use of telemedicine – for talking to hospitalized patients without increasing viral exposure, for linking up with family members who can’t visit their loved ones in the hospital, and for helping frontline hospital staff who need a palliative care consultation – or just a chance to debrief on what they are seeing.
A pandemic wake-up call
Elizabeth Gundersen, MD, FHM, FAAHPM, director of the hospice and palliative medicine fellowship program at the Charles E. Schmidt College of Medicine at Florida Atlantic University (FAU) in Boca Raton, practiced hospital medicine for 10 years before pursuing a fellowship in hospice and palliative medicine and working as an academic palliative medicine physician. She calls the pandemic a wake-up call for gaps in care and all the things that weren’t working well in the health care system.
“Now we are seeing more clearly what’s lacking – or broken – and what we will carry forward from this experience into the post-COVID world,” she said. Some hospitalists do palliative care very well, and others don’t feel as comfortable in having these difficult conversations with patients. But in the uncertain course of the virus they get thrust into it.
Although FAU’s associated hospitals were not as inundated with COVID-19 patients in the early weeks of the pandemic as were other regions, the volume of other patients plummeted, Dr. Gundersen said, adding that “there’s still been incredible intensity and worry about the virus. For me, the basic role of palliative care hasn’t changed, and the phrase I have always used when introducing myself – ‘we’re an extra layer of support for the patient and family’ – still holds true,” she said.
“I try to make it clear to people that palliative care is not synonymous with end-of-life care. We don’t want people to think that a palliative care referral implies imminent death. The goal is not to get more people to have a do not attempt resuscitation (DNAR) order, but to determine the patient and family’s treatment goals and whether a DNAR order fits those goals.”
The tough conversations
Dr. Gundersen is cochair of SHM’s Palliative Care Special Interest Group, along with Rab Razzak, MD, clinical director of palliative medicine at University Hospitals Cleveland Medical Center, one of the hospitals affiliated with Case Western University in Cleveland. (Connect with them on Twitter: @Top_Gundersen and @rabrazzak.)
Dr. Razzak also transitioned from hospital medicine to palliative medicine 10 years ago. “As a hospitalist, I enjoyed the tough conversations and bringing the human element into my health care interactions,” he explained. “To me, palliative care is a philosophy of care that puts the person we call the patient at the center of the interaction, while we try to figure out how to best care for them as a person.”
When the pandemic hit, University Hospitals made 20 ICU beds available for COVID-19 patients, Dr. Razzak said. This unit has since been full but not overflowing, while overall hospital census went down. The palliative care team at the hospital includes four inpatient doctors, nurse practitioners, and a chaplain, as well as an outpatient team primarily focused on oncology.
“In some settings, palliative care has been at the forefront of difficult conversations, when things aren’t going well for the patient and there’s much uncertainty,” Dr. Razzak said. The interface between hospital medicine and palliative care can be complementary, he added. “We talk about primary palliative care, which we want every discipline to be able to do – lead meaningful conversations, help manage symptoms.”
The take-home message for hospitalists, he said, is to get training in how to have these discussions, using such resources as VitalTalk (https://www.vitaltalk.org/), a nonprofit organization that disseminates education in communication skills for difficult conversations, and the Center to Advance Palliative Care (www.capc.org) at Icahn School of Medicine at Mount Sinai in New York City. “Once you’ve mastered the conversation, it will get easier. But ask for help when you need it, and learn how to know when you need it.”
Dr. Gundersen added that hospital medicine groups and palliative care teams could reach out to each other and talk about what they did in the crisis and how they can work together in the future. She recommends frequent ongoing support and collaboration that could range from formal conferences or training sessions to informal team interactions, perhaps with sandwiches in the doctor’s lounge – provided that there’s room for social distancing. She has recently started giving talks in the community and grand rounds presentations in hospitals about palliative care.
Other approaches and applications
In New York City, the initial epicenter for the pandemic in the United States, the adult palliative care service of Columbia University Medical Center (CUMC) experienced a sevenfold increase in consultation requests at the apex of the crisis, said its director, Craig Blinderman, MD. That demand was impossible to meet with existing staff. So Dr. Blinderman and colleagues established a virtual consultation model, recruiting and deploying volunteer out-of-state palliative care specialists to staff it.
An eight-bed palliative care unit was opened at CUMC for COVID-19 patients whose surrogates had opted not to initiate or continue intubation or life-sustaining treatments. This helped to relieve some of the pressures on the ICUs while making it possible for in-person visits to the hospice unit by families – in full PPE. Palliative care staff were embedded in various units in the hospital.
A palliative care response team composed of a hospice and palliative medicine fellow and four psychiatry residents or fellows, based in the emergency department and with supervision from the palliative care team, provided time-critical goals of care conversations with families using telemedicine – and a forum for listening to their suffering. Dr. Blinderman and colleagues also have found time to write up their experience for medical journals.1,2
There’s no reason to think that hospitalists, with a little basic training, couldn’t be having these same goals of care conversations, Dr. Blinderman said. “But the fact that hospitalists, at the pandemic’s peak, along with ICU doctors, were seeing an unprecedented magnitude of dying on a daily basis generated a lot of moral distress for them.”
Palliative care professionals, because they engage with these issues in a different way, may be somewhat better equipped to deal with the sheer emotional demands when so many are dying, as at the peak of the surge in New York. “We don’t see dying as a failure on our part but an opportunity to relieve suffering,” Dr. Blinderman said. And the palliative care field also emphasizes the importance of self-care for its practitioners.
“How do we meet the incredible palliative care needs in the epicenter of a pandemic? That question also applies to other kinds of crises we could imagine, for example, climate-related disasters,” Dr. Blinderman said. “What lessons have we learned about the value of palliative care and how to start incorporating it more integrally into the delivery of hospital care? Here we showed that we could work collaboratively with our colleagues at other major medical centers, bringing together their expertise to help us when we didn’t have the bandwidth to meet the demand,” he said.
Scripts can help
“Also, it won’t make sense to just go back to normal (after the crisis fades),” Dr. Blinderman said. “We need to take a close look at how our society is functioning in the wake of the pandemic and the ways the health care system has failed us. We have learned that we’re all interconnected and we need to work together to serve our communities – locally and nationally – applying basic distributive justice.”
Could there be, for example, a national infrastructure for mobilizing and deploying palliative care resources to areas of greatest need, similar to what was done in New York?
At Northwestern Medicine in Chicago, a number of palliative care clinicians at the system’s hospitals worked together to develop scripts designed to help other clinicians start goals of care conversations with patients and families, for use in the hospital as well as in outpatient primary care and other settings, with results integrated into the system’s electronic health record.
Front-line clinicians may not have the time to ask for formal consults from palliative care because of high volume and rapidly changing patient status, explained Eytan Szmuilowicz, MD, director of the section of palliative medicine at Northwestern Memorial Hospital. Or they may not have access to specialty-level palliative care in their settings.
The scripts are aimed at primary care, emergency physicians, and hospitalists needing to consider critical care placement or attempted resuscitation and to ICU clinicians helping families make decisions about life-sustaining treatments. They also can help facilitate advance care planning discussions. An example is “CALMER,” a six-step mnemonic guide to promote goals of care discussions with hospitalized patients. For more information on these scripts, contact Dr. Szmuilowicz: Eytan.Szmuilowicz@nm.org.
Eerily quiet
The COVID-19 crisis has been quite a whirlwind for hospital medicine, said Jeanie Youngwerth, MD, a hospitalist and program director of the palliative care service at the University of Colorado in Denver, which was a significant viral hotspot early on.
“When it first started, things seemed to change almost overnight – starting on Friday, March 13. People had to take action right away to develop work flows and the technology to allow us to see as many patients as possible,” she said. By the time Monday came, it was a whole new ballgame.
Dr. Youngwerth and two colleagues worked quickly to develop inpatient telemedicine capacity where none existed. “We knew we would not be going into patients’ rooms, but most of our team showed up in the hospital to work with the primary care teams. Our job was to see what we could do that actually made a difference,” she said.
“The hospital became a very strange place. You’d walk down the hallway and it was eerily quiet. Everybody you came across was being so nice to each other.” Televisits became a powerful way to bring the human connection back to medical care.
“What we learned from families was that they were thirsting to have some kind of connection with their loved one, and to be able to talk about their loved one and who they were as a person,” she said. “We’d contact the family through video visits and then, when the family meeting ended, the nurse would bring an iPad into the patient’s room so the family could see their loved one on a ventilator. They would immediately start communicating with their loved one, praying aloud, singing, playing music. It would make a huge difference for the family – and for the staff.”
References
1. Nakagawa S et al. Pandemic palliative care consultations spanning state and institutional borders. J Am Geriatr Soc. 2020 May 22. doi: 10.1111/jgs.16643.
2. Lee J Abrukin L, Flores S. Early intervention of palliative care in the emergency department during the COVID-19 pandemic. JAMA Intern Med. 2020 Jun 5. doi: 10.1001/jamainternmed.2020.2713.
The best and worst states for health care in 2020
according to the personal finance website WalletHub.
The Bay State finds itself at the top of the company’s annual ranking of state health care systems this year after finishing second in 2019 to Minnesota, which is now ranked second. Rhode Island is third this year, followed by Washington, D.C., and North Dakota, WalletHub reported Aug. 3.
The inclusion of Washington, D.C., allowed Georgia to finish 51st out of 50 states, just below the quartet of Louisiana (50th), Alabama (49th), North Carolina (48th), and Mississippi (47th). Alaska, which occupied the bottom spot in 2019, moved up to 42nd this year, the analysis showed.
The rankings are based on 44 (up from 43 last year) metrics that are grouped into three broad categories: cost (6 metrics), access (24 metrics), and outcomes (14 metrics). The one new measure added for 2020? That would be health infrastructure for coronavirus, which is itself based on a different WalletHub ranking.
Massachusetts’ top finish this year was driven by strong showings in such metrics as average monthly insurance premium (first), physicians per capita (second), insured children (first) and adults (first), and infant mortality rate (fourth). The state was 1st overall in outcomes and 4th in access but only 20th in cost, the company said.
Positive signs among the lowest-ranked states include Louisiana’s 18th-place finish in access, ahead of such top 10 states as Iowa and Hawaii, and Mississippi’s 17th in cost, which is higher than four of the states in the top 10, including Massachusetts, WalletHub said in the report.
Data for the analysis came from 22 different sources, including the Institute for Health Metrics and Evaluation, Centers for Medicare & Medicaid Services, Association of American Medical Colleges, and the American Telemedicine Association.
according to the personal finance website WalletHub.

The Bay State finds itself at the top of the company’s annual ranking of state health care systems this year after finishing second in 2019 to Minnesota, which is now ranked second. Rhode Island is third this year, followed by Washington, D.C., and North Dakota, WalletHub reported Aug. 3.
The inclusion of Washington, D.C., allowed Georgia to finish 51st out of 50 states, just below the quartet of Louisiana (50th), Alabama (49th), North Carolina (48th), and Mississippi (47th). Alaska, which occupied the bottom spot in 2019, moved up to 42nd this year, the analysis showed.
The rankings are based on 44 (up from 43 last year) metrics that are grouped into three broad categories: cost (6 metrics), access (24 metrics), and outcomes (14 metrics). The one new measure added for 2020? That would be health infrastructure for coronavirus, which is itself based on a different WalletHub ranking.
Massachusetts’ top finish this year was driven by strong showings in such metrics as average monthly insurance premium (first), physicians per capita (second), insured children (first) and adults (first), and infant mortality rate (fourth). The state was 1st overall in outcomes and 4th in access but only 20th in cost, the company said.
Positive signs among the lowest-ranked states include Louisiana’s 18th-place finish in access, ahead of such top 10 states as Iowa and Hawaii, and Mississippi’s 17th in cost, which is higher than four of the states in the top 10, including Massachusetts, WalletHub said in the report.
Data for the analysis came from 22 different sources, including the Institute for Health Metrics and Evaluation, Centers for Medicare & Medicaid Services, Association of American Medical Colleges, and the American Telemedicine Association.
according to the personal finance website WalletHub.

The Bay State finds itself at the top of the company’s annual ranking of state health care systems this year after finishing second in 2019 to Minnesota, which is now ranked second. Rhode Island is third this year, followed by Washington, D.C., and North Dakota, WalletHub reported Aug. 3.
The inclusion of Washington, D.C., allowed Georgia to finish 51st out of 50 states, just below the quartet of Louisiana (50th), Alabama (49th), North Carolina (48th), and Mississippi (47th). Alaska, which occupied the bottom spot in 2019, moved up to 42nd this year, the analysis showed.
The rankings are based on 44 (up from 43 last year) metrics that are grouped into three broad categories: cost (6 metrics), access (24 metrics), and outcomes (14 metrics). The one new measure added for 2020? That would be health infrastructure for coronavirus, which is itself based on a different WalletHub ranking.
Massachusetts’ top finish this year was driven by strong showings in such metrics as average monthly insurance premium (first), physicians per capita (second), insured children (first) and adults (first), and infant mortality rate (fourth). The state was 1st overall in outcomes and 4th in access but only 20th in cost, the company said.
Positive signs among the lowest-ranked states include Louisiana’s 18th-place finish in access, ahead of such top 10 states as Iowa and Hawaii, and Mississippi’s 17th in cost, which is higher than four of the states in the top 10, including Massachusetts, WalletHub said in the report.
Data for the analysis came from 22 different sources, including the Institute for Health Metrics and Evaluation, Centers for Medicare & Medicaid Services, Association of American Medical Colleges, and the American Telemedicine Association.
Global study to track COVID-19’s impact on the brain
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
At its annual meeting, the Alzheimer’s Association announced the launch of a global study to examine the impact of COVID-19 on the brain, as well as policy recommendations to better address the COVID-19 crisis in long-term care facilities. The study will be led by researchers at the Alzheimer’s Association and the University of Texas Health, San Antonio, with participation from more than 30 countries and technical guidance from the World Health Organization.
The target sample size is 20,000-40,000 total participants.
Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, announced the study’s launch during a COVID-19–focused panel discussion at the virtual annual meeting of the Alzheimer’s Association International Conference 2020.
“To build a strong foundation for this research, we will align with existing studies, such as the Framingham Heart Study, and clinicians from around the world on how the data are going to be collected, obtained, and shared. We are going to have cross-study collaborations to understand the impact of the virus on the brain directly,” said Dr. Carrillo. “We will have some very good data to present next year at AAIC.”
‘Frightening’ headlines
As previously reported, mounting evidence suggests that SARS-CoV-2 invades the central nervous system, causing a wide range of neurologic and neuropsychiatric complications, including stroke, psychosis, altered mental state, and dementia-like syndrome. It’s likely that “dementia does not increase the risk for COVID-19, just like dementia does not increase risk for the flu. But increased age, being in a long-term care setting, and common health conditions that often accompany dementia may increase the risk,” Dr. Carrillo said.
Panel member Beth Kallmyer, MSW, vice president of care and support at the Alzheimer’s Association, spoke about the ongoing challenges long-term care facilities are facing during the pandemic. “You’ve all seen the headlines, and they’re frightening, frankly,” she said. An estimated 59,000 residents and employees of long-term care have died as a result of COVID-19, which is 42% of all U.S. deaths.
The long-term care community is being impacted at “significantly greater rates than the rest of society and yet we don’t have things in place to protect them. We also know that individuals living with dementia make up a large percentage of those that are living in long-term care,” Ms. Kallmyer said.
She noted that infection control is always a challenge in long-term care settings, but infection control during a pandemic “takes it to a whole other level.” Quarantining is hard for anyone, “but when you layer dementia on top of that we have a real challenge.” One long-term care provider told Ms. Kallmyer that “we might be saving them from COVID, but we’re losing them to social isolation and cognitive decline.”
New recommendations
Ms. Kallmyer outlined new policy recommendations from the Alzheimer’s Association to address the COVID-19 crisis in long-term and community-based care settings. They include:
- Testing every resident, employee, and visitor each time they leave and come back, so residents would not need to be confined to their own rooms
- Having a single portal that is easy and efficient for reporting cases
- Developing “surge activation” protocols to respond to hot spots, including the possibility of “strike teams” that go in and help during an outbreak
- Making sure all long-term care providers have full access to all needed personal protective equipment (PPE)
“Five months in and long-term care providers still don’t have adequate PPE. This is unacceptable,” said Ms. Kallmyer. “We have to be able to provide them with PPE.”
Panel member Gregory A. Jicha, MD, PhD, Sanders-Brown Center on Aging, University of Kentucky, Lexington, spoke about the critical need to continue Alzheimer’s disease research during the pandemic, noting that the number of promising targets for Alzheimer’s disease and related dementias has “never been higher or more comprehensive.”
Measures to ensure safety of researchers and participants include screening for symptoms (50% effective), social distancing (93% effective), minimizing exposure time (50% effective), limiting staff to 50% (50% effective), cloth/paper masks (80% effective), and testing (99.25% effective), Dr. Jicha noted.
With no safety measures in place, the risk of getting COVID-19 from a research visit is 1 in 20; when all these safety measures are combined, the risk is 1 in over 1.5 million, so “we can essentially eradicate or minimize the risks for COVID to less that of a lightning strike,” he said.
Dr. Carrillo, Ms. Kallmyer, and Dr. Jicha disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AAIC 2020
COVID-19 taking financial toll on people in U.S. with diabetes
The COVID-19 pandemic is taking a particularly severe financial toll on people with diabetes, new research from the United States suggests.
Results from a national online survey of 5,000 people with diabetes conducted between June 26 and July 1, 2020, were posted July 29 on the American Diabetes Association website.
The survey, conducted by the diabetes research company dQ&A in association with the ADA, revealed that Americans with diabetes are experiencing extreme financial pressures, leading to medication and supply rationing.
A high proportion of respondents had either lost income or are working in jobs that place them at risk for catching the novel coronavirus.
“These new numbers show the urgency needed to adopt measures to protect and assist the millions of people with diabetes who are suffering through this pandemic,” Tracey D. Brown, CEO of the ADA, said in a statement.
She called for states to extend health care coverage to people who have lost their jobs, for the eradication of insulin copays during the pandemic, and for increased COVID-19 testing capacity in high-risk communities.
“If these actions aren’t taken immediately, we will continue to see devastating impacts and outcomes for millions of vulnerable Americans,” Ms. Brown stressed.
COVID-19 has worsened financial pressures for people with diabetes
In the survey, 24% of respondents reported having used savings, loans, or stimulus check money to pay for diabetes care in the past 3 months. Among those who have lost income, half are using savings or stimulus money.
A quarter of respondents said they have been self-rationing supplies to cut costs.
Extrapolating to the entire U.S. population with diabetes, dQ&A estimated that roughly 650,000 are skipping insulin doses or taking less than prescribed, and 3 million are skipping blood glucose tests.
In June, the unemployment rate for people with diabetes was 18%, higher than the national rate of 12%.
Also higher is the proportion of those working prior to the pandemic who have since lost income: 33%, compared with 29% for the general population.
Among those who are self-employed, 7 in 10 of those with diabetes have lost some or all of their income.
Many with diabetes who are employed are vulnerable to exposure
Of those who remain employed, half said they can’t work from home.
Of those, 60% work in essential industries, with 22% in health care. A large majority, 90%, reported lack of social distancing at work and nearly a third work in places that don’t require masks.
“People with diabetes are helping to provide the services we all depend on during this pandemic, even as it puts their own well-being at risk,” the report said.
It concluded that “these numbers represent a conservative estimate of the pandemic’s impact. They are generated from an ongoing online study of the diabetes population amongst people who have opted in to participate.”
A version of this article originally appeared on Medscape.com.
The COVID-19 pandemic is taking a particularly severe financial toll on people with diabetes, new research from the United States suggests.
Results from a national online survey of 5,000 people with diabetes conducted between June 26 and July 1, 2020, were posted July 29 on the American Diabetes Association website.
The survey, conducted by the diabetes research company dQ&A in association with the ADA, revealed that Americans with diabetes are experiencing extreme financial pressures, leading to medication and supply rationing.
A high proportion of respondents had either lost income or are working in jobs that place them at risk for catching the novel coronavirus.
“These new numbers show the urgency needed to adopt measures to protect and assist the millions of people with diabetes who are suffering through this pandemic,” Tracey D. Brown, CEO of the ADA, said in a statement.
She called for states to extend health care coverage to people who have lost their jobs, for the eradication of insulin copays during the pandemic, and for increased COVID-19 testing capacity in high-risk communities.
“If these actions aren’t taken immediately, we will continue to see devastating impacts and outcomes for millions of vulnerable Americans,” Ms. Brown stressed.
COVID-19 has worsened financial pressures for people with diabetes
In the survey, 24% of respondents reported having used savings, loans, or stimulus check money to pay for diabetes care in the past 3 months. Among those who have lost income, half are using savings or stimulus money.
A quarter of respondents said they have been self-rationing supplies to cut costs.
Extrapolating to the entire U.S. population with diabetes, dQ&A estimated that roughly 650,000 are skipping insulin doses or taking less than prescribed, and 3 million are skipping blood glucose tests.
In June, the unemployment rate for people with diabetes was 18%, higher than the national rate of 12%.
Also higher is the proportion of those working prior to the pandemic who have since lost income: 33%, compared with 29% for the general population.
Among those who are self-employed, 7 in 10 of those with diabetes have lost some or all of their income.
Many with diabetes who are employed are vulnerable to exposure
Of those who remain employed, half said they can’t work from home.
Of those, 60% work in essential industries, with 22% in health care. A large majority, 90%, reported lack of social distancing at work and nearly a third work in places that don’t require masks.
“People with diabetes are helping to provide the services we all depend on during this pandemic, even as it puts their own well-being at risk,” the report said.
It concluded that “these numbers represent a conservative estimate of the pandemic’s impact. They are generated from an ongoing online study of the diabetes population amongst people who have opted in to participate.”
A version of this article originally appeared on Medscape.com.
The COVID-19 pandemic is taking a particularly severe financial toll on people with diabetes, new research from the United States suggests.
Results from a national online survey of 5,000 people with diabetes conducted between June 26 and July 1, 2020, were posted July 29 on the American Diabetes Association website.
The survey, conducted by the diabetes research company dQ&A in association with the ADA, revealed that Americans with diabetes are experiencing extreme financial pressures, leading to medication and supply rationing.
A high proportion of respondents had either lost income or are working in jobs that place them at risk for catching the novel coronavirus.
“These new numbers show the urgency needed to adopt measures to protect and assist the millions of people with diabetes who are suffering through this pandemic,” Tracey D. Brown, CEO of the ADA, said in a statement.
She called for states to extend health care coverage to people who have lost their jobs, for the eradication of insulin copays during the pandemic, and for increased COVID-19 testing capacity in high-risk communities.
“If these actions aren’t taken immediately, we will continue to see devastating impacts and outcomes for millions of vulnerable Americans,” Ms. Brown stressed.
COVID-19 has worsened financial pressures for people with diabetes
In the survey, 24% of respondents reported having used savings, loans, or stimulus check money to pay for diabetes care in the past 3 months. Among those who have lost income, half are using savings or stimulus money.
A quarter of respondents said they have been self-rationing supplies to cut costs.
Extrapolating to the entire U.S. population with diabetes, dQ&A estimated that roughly 650,000 are skipping insulin doses or taking less than prescribed, and 3 million are skipping blood glucose tests.
In June, the unemployment rate for people with diabetes was 18%, higher than the national rate of 12%.
Also higher is the proportion of those working prior to the pandemic who have since lost income: 33%, compared with 29% for the general population.
Among those who are self-employed, 7 in 10 of those with diabetes have lost some or all of their income.
Many with diabetes who are employed are vulnerable to exposure
Of those who remain employed, half said they can’t work from home.
Of those, 60% work in essential industries, with 22% in health care. A large majority, 90%, reported lack of social distancing at work and nearly a third work in places that don’t require masks.
“People with diabetes are helping to provide the services we all depend on during this pandemic, even as it puts their own well-being at risk,” the report said.
It concluded that “these numbers represent a conservative estimate of the pandemic’s impact. They are generated from an ongoing online study of the diabetes population amongst people who have opted in to participate.”
A version of this article originally appeared on Medscape.com.
ACS disagrees with CDC on HPV vaccination in adults
The ACS has endorsed two recommendations made by the CDC’s Advisory Committee on Immunization Practices, but the ACS does not agree with a third recommendation for older adults.
The ACIP recommends shared clinical decision-making regarding human papillomavirus (HPV) vaccination in some adults aged 27-45 years who are not adequately vaccinated. The ACS does not endorse this recommendation “because of the low effectiveness and low cancer prevention potential of vaccination in this age group, the burden of decision-making on patients and clinicians, and the lack of sufficient guidance on the selection of individuals who might benefit,” wrote Debbie Saslow, PhD, of the ACS’s section on human papillomavirus and gynecologic cancers, and colleagues.
Dr. Saslow and colleagues detailed the ACS recommendations in CA: A Cancer Journal for Clinicians.
The HPV vaccine protects against the virus that can cause cervical, oropharyngeal, anal, vaginal, vulvar, and penile cancers. For younger people, the ACIP recommends routine HPV vaccination of boys and girls aged 9-12 years and catch-up vaccination in everyone up to age 26 who has not been fully immunized against HPV.
The ACS endorses both of these recommendations. It also advises clinicians to tell patients aged 22-26 years who haven’t received the HPV vaccine or completed the series that the vaccine is less effective at reducing the risk of cancer at older ages.
After the Food and Drug Administration approved the HPV vaccine for adults aged 27-45 years, the ACIP updated its recommendations to state that routine catch-up vaccination is not recommended for anyone aged over 26 years. However, the ACIP recommended that these older adults talk with their providers about the risks and benefits of the vaccine to determine whether to get it.
The ACS subsequently conducted a methodological review of the ACIP’s recommendations and published its own adapted guidance, stating that the ACS does not endorse the shared decision-making. Administering the HPV vaccine to adults aged over 26 years would only prevent an estimated 0.5% of additional cancer cases, 0.4% additional cases of cervical precancer, and 0.3% additional cases of genital warts over the next 100 years, compared with vaccination under age 26.
“In addition to the low effectiveness and low cancer prevention potential of vaccination in this age group, other considerations included the burden of decision-making on patients and clinicians and the lack of sufficient guidance on the selection of individuals who might benefit,” according to the guidance. The ACS also expressed concern that these provider-patient discussions could interfere with the public health goal of increasing HPV vaccination in younger people.
HPV vaccination rates have lagged substantially behind other routinely recommended childhood vaccinations. Just over half (51%) of U.S. teens aged 13-17 years were up to date with HPV vaccination, and 68% had received one dose of the vaccine in 2018, according to the National Immunization Survey.
It’s very uncommon for a professional medical organization to not endorse recommendations from the CDC, particularly with vaccines, according to Robert A. Bednarczyk, PhD, an assistant professor of public health at Emory University, Atlanta, who specializes in HPV vaccination research but was not involved with the ACS statement or the ACIP recommendations.
“Often, for vaccination recommendations, there is a harmonization between health care provider organizations, such as the American Academy of Pediatrics, American Academy of Family Physicians, etc., when new vaccination schedules are released,” Dr. Bednarczyk said.
He acknowledged the ACS’s reasons for not endorsing the ACIP’s HPV recommendations in older adults: the burden of shared decision-making given the communication issues, the vaccine’s lower effectiveness in this population, and the ongoing HPV vaccine shortage.
But Dr. Bednarczyk also pointed out that the ACIP’s recommendation opens the door to these discussions when they may actually be needed, such as in adults at greater risk for HPV. He cited data suggesting that, in 2015, divorces occurred in 24 out of 1,000 married people aged 25-39 years and 21 out of 1,000 people aged 40-49.
“When you consider these marriages that end, in addition to marriages that end when one spouse dies, there is a potential for individuals who previously had a low risk of HPV acquisition now entering into new potential sexual relationships,” Dr. Bednarczyk said. “Additionally, it has been estimated that approximately 4% of the U.S. population are in open or consensually nonmonogamous relationships, where exposure to more sexual partners may increase their risk for HPV. These are just some examples of where conversations with health care providers, and shared clinical decision-making, can help with a targeted reduction of HPV risk.”
The ACIP recommendation regarding adults aged 27-45 years also provides people in this age group with insurance coverage for the HPV vaccine if they choose to get it, Dr. Bednarczyk pointed out. Insurance companies may not be required to cover HPV vaccination in people aged over 26 years without the CDC’s recommendation, even if it’s not for routine immunization.
Dr. Bednarczyk agreed, however, with how the ACS adapted the CDC’s recommendation for routine vaccination in youth. The CDC’s routine recommendation is at ages 11-12 but can begin at 9 years, according to the ACIP. The ACS guidance qualifies this statement to place more emphasis on encouraging the vaccine earlier.
“Routine HPV vaccination between ages 9-12 is expected to achieve higher on-time vaccination rates, resulting in increased numbers of cancers prevented,” according to the ACS. “Health care providers are encouraged to start offering the HPV vaccine at age 9 or 10.”
Dr. Bednarczyk pointed to some of his past research finding low proportions of teens fully vaccinated against HPV by age 13 years (J Infect Dis. 2019 Jul 31;220[5]:730-4). Therefore, “any efforts to encourage vaccination, including starting the series at ages 9-10 years may help,” he said.
He also agreed that there may be diminished effectiveness with vaccinating adults aged 22-26, “but this should also be considered relative to an individual’s risk of acquiring HPV.”
While an HPV vaccine shortage is a major concern and HPV vaccination efforts should remain most focused on young teens, adults should not necessarily be neglected, Dr. Bednarczyk noted.
“Given how common HPV infection is in the population, open discussion between patients and health care providers can help identify those adults for whom HPV vaccination can be effective,” he said.
The development of the ACS guideline was supported by ACS operational funds. The ACS has received an independent educational grant from Merck Sharp & Dohme for a project intended to increase HPV vaccination rates. Dr. Saslow is the principal investigator for a cooperative agreement between the ACS and the CDC to support the National HPV Vaccination Roundtable and is coprincipal investigator of a cooperative agreement between the ACS and CDC to support initiatives to increase HPV vaccination. The remaining authors and Dr. Bednarczyk reported no relevant disclosures.
SOURCE: Saslow D et al. CA Cancer J Clin. 2020 Jul 8. doi: 10.3322/caac.21616.
The ACS has endorsed two recommendations made by the CDC’s Advisory Committee on Immunization Practices, but the ACS does not agree with a third recommendation for older adults.
The ACIP recommends shared clinical decision-making regarding human papillomavirus (HPV) vaccination in some adults aged 27-45 years who are not adequately vaccinated. The ACS does not endorse this recommendation “because of the low effectiveness and low cancer prevention potential of vaccination in this age group, the burden of decision-making on patients and clinicians, and the lack of sufficient guidance on the selection of individuals who might benefit,” wrote Debbie Saslow, PhD, of the ACS’s section on human papillomavirus and gynecologic cancers, and colleagues.
Dr. Saslow and colleagues detailed the ACS recommendations in CA: A Cancer Journal for Clinicians.
The HPV vaccine protects against the virus that can cause cervical, oropharyngeal, anal, vaginal, vulvar, and penile cancers. For younger people, the ACIP recommends routine HPV vaccination of boys and girls aged 9-12 years and catch-up vaccination in everyone up to age 26 who has not been fully immunized against HPV.
The ACS endorses both of these recommendations. It also advises clinicians to tell patients aged 22-26 years who haven’t received the HPV vaccine or completed the series that the vaccine is less effective at reducing the risk of cancer at older ages.
After the Food and Drug Administration approved the HPV vaccine for adults aged 27-45 years, the ACIP updated its recommendations to state that routine catch-up vaccination is not recommended for anyone aged over 26 years. However, the ACIP recommended that these older adults talk with their providers about the risks and benefits of the vaccine to determine whether to get it.
The ACS subsequently conducted a methodological review of the ACIP’s recommendations and published its own adapted guidance, stating that the ACS does not endorse the shared decision-making. Administering the HPV vaccine to adults aged over 26 years would only prevent an estimated 0.5% of additional cancer cases, 0.4% additional cases of cervical precancer, and 0.3% additional cases of genital warts over the next 100 years, compared with vaccination under age 26.
“In addition to the low effectiveness and low cancer prevention potential of vaccination in this age group, other considerations included the burden of decision-making on patients and clinicians and the lack of sufficient guidance on the selection of individuals who might benefit,” according to the guidance. The ACS also expressed concern that these provider-patient discussions could interfere with the public health goal of increasing HPV vaccination in younger people.
HPV vaccination rates have lagged substantially behind other routinely recommended childhood vaccinations. Just over half (51%) of U.S. teens aged 13-17 years were up to date with HPV vaccination, and 68% had received one dose of the vaccine in 2018, according to the National Immunization Survey.
It’s very uncommon for a professional medical organization to not endorse recommendations from the CDC, particularly with vaccines, according to Robert A. Bednarczyk, PhD, an assistant professor of public health at Emory University, Atlanta, who specializes in HPV vaccination research but was not involved with the ACS statement or the ACIP recommendations.
“Often, for vaccination recommendations, there is a harmonization between health care provider organizations, such as the American Academy of Pediatrics, American Academy of Family Physicians, etc., when new vaccination schedules are released,” Dr. Bednarczyk said.
He acknowledged the ACS’s reasons for not endorsing the ACIP’s HPV recommendations in older adults: the burden of shared decision-making given the communication issues, the vaccine’s lower effectiveness in this population, and the ongoing HPV vaccine shortage.
But Dr. Bednarczyk also pointed out that the ACIP’s recommendation opens the door to these discussions when they may actually be needed, such as in adults at greater risk for HPV. He cited data suggesting that, in 2015, divorces occurred in 24 out of 1,000 married people aged 25-39 years and 21 out of 1,000 people aged 40-49.
“When you consider these marriages that end, in addition to marriages that end when one spouse dies, there is a potential for individuals who previously had a low risk of HPV acquisition now entering into new potential sexual relationships,” Dr. Bednarczyk said. “Additionally, it has been estimated that approximately 4% of the U.S. population are in open or consensually nonmonogamous relationships, where exposure to more sexual partners may increase their risk for HPV. These are just some examples of where conversations with health care providers, and shared clinical decision-making, can help with a targeted reduction of HPV risk.”
The ACIP recommendation regarding adults aged 27-45 years also provides people in this age group with insurance coverage for the HPV vaccine if they choose to get it, Dr. Bednarczyk pointed out. Insurance companies may not be required to cover HPV vaccination in people aged over 26 years without the CDC’s recommendation, even if it’s not for routine immunization.
Dr. Bednarczyk agreed, however, with how the ACS adapted the CDC’s recommendation for routine vaccination in youth. The CDC’s routine recommendation is at ages 11-12 but can begin at 9 years, according to the ACIP. The ACS guidance qualifies this statement to place more emphasis on encouraging the vaccine earlier.
“Routine HPV vaccination between ages 9-12 is expected to achieve higher on-time vaccination rates, resulting in increased numbers of cancers prevented,” according to the ACS. “Health care providers are encouraged to start offering the HPV vaccine at age 9 or 10.”
Dr. Bednarczyk pointed to some of his past research finding low proportions of teens fully vaccinated against HPV by age 13 years (J Infect Dis. 2019 Jul 31;220[5]:730-4). Therefore, “any efforts to encourage vaccination, including starting the series at ages 9-10 years may help,” he said.
He also agreed that there may be diminished effectiveness with vaccinating adults aged 22-26, “but this should also be considered relative to an individual’s risk of acquiring HPV.”
While an HPV vaccine shortage is a major concern and HPV vaccination efforts should remain most focused on young teens, adults should not necessarily be neglected, Dr. Bednarczyk noted.
“Given how common HPV infection is in the population, open discussion between patients and health care providers can help identify those adults for whom HPV vaccination can be effective,” he said.
The development of the ACS guideline was supported by ACS operational funds. The ACS has received an independent educational grant from Merck Sharp & Dohme for a project intended to increase HPV vaccination rates. Dr. Saslow is the principal investigator for a cooperative agreement between the ACS and the CDC to support the National HPV Vaccination Roundtable and is coprincipal investigator of a cooperative agreement between the ACS and CDC to support initiatives to increase HPV vaccination. The remaining authors and Dr. Bednarczyk reported no relevant disclosures.
SOURCE: Saslow D et al. CA Cancer J Clin. 2020 Jul 8. doi: 10.3322/caac.21616.
The ACS has endorsed two recommendations made by the CDC’s Advisory Committee on Immunization Practices, but the ACS does not agree with a third recommendation for older adults.
The ACIP recommends shared clinical decision-making regarding human papillomavirus (HPV) vaccination in some adults aged 27-45 years who are not adequately vaccinated. The ACS does not endorse this recommendation “because of the low effectiveness and low cancer prevention potential of vaccination in this age group, the burden of decision-making on patients and clinicians, and the lack of sufficient guidance on the selection of individuals who might benefit,” wrote Debbie Saslow, PhD, of the ACS’s section on human papillomavirus and gynecologic cancers, and colleagues.
Dr. Saslow and colleagues detailed the ACS recommendations in CA: A Cancer Journal for Clinicians.
The HPV vaccine protects against the virus that can cause cervical, oropharyngeal, anal, vaginal, vulvar, and penile cancers. For younger people, the ACIP recommends routine HPV vaccination of boys and girls aged 9-12 years and catch-up vaccination in everyone up to age 26 who has not been fully immunized against HPV.
The ACS endorses both of these recommendations. It also advises clinicians to tell patients aged 22-26 years who haven’t received the HPV vaccine or completed the series that the vaccine is less effective at reducing the risk of cancer at older ages.
After the Food and Drug Administration approved the HPV vaccine for adults aged 27-45 years, the ACIP updated its recommendations to state that routine catch-up vaccination is not recommended for anyone aged over 26 years. However, the ACIP recommended that these older adults talk with their providers about the risks and benefits of the vaccine to determine whether to get it.
The ACS subsequently conducted a methodological review of the ACIP’s recommendations and published its own adapted guidance, stating that the ACS does not endorse the shared decision-making. Administering the HPV vaccine to adults aged over 26 years would only prevent an estimated 0.5% of additional cancer cases, 0.4% additional cases of cervical precancer, and 0.3% additional cases of genital warts over the next 100 years, compared with vaccination under age 26.
“In addition to the low effectiveness and low cancer prevention potential of vaccination in this age group, other considerations included the burden of decision-making on patients and clinicians and the lack of sufficient guidance on the selection of individuals who might benefit,” according to the guidance. The ACS also expressed concern that these provider-patient discussions could interfere with the public health goal of increasing HPV vaccination in younger people.
HPV vaccination rates have lagged substantially behind other routinely recommended childhood vaccinations. Just over half (51%) of U.S. teens aged 13-17 years were up to date with HPV vaccination, and 68% had received one dose of the vaccine in 2018, according to the National Immunization Survey.
It’s very uncommon for a professional medical organization to not endorse recommendations from the CDC, particularly with vaccines, according to Robert A. Bednarczyk, PhD, an assistant professor of public health at Emory University, Atlanta, who specializes in HPV vaccination research but was not involved with the ACS statement or the ACIP recommendations.
“Often, for vaccination recommendations, there is a harmonization between health care provider organizations, such as the American Academy of Pediatrics, American Academy of Family Physicians, etc., when new vaccination schedules are released,” Dr. Bednarczyk said.
He acknowledged the ACS’s reasons for not endorsing the ACIP’s HPV recommendations in older adults: the burden of shared decision-making given the communication issues, the vaccine’s lower effectiveness in this population, and the ongoing HPV vaccine shortage.
But Dr. Bednarczyk also pointed out that the ACIP’s recommendation opens the door to these discussions when they may actually be needed, such as in adults at greater risk for HPV. He cited data suggesting that, in 2015, divorces occurred in 24 out of 1,000 married people aged 25-39 years and 21 out of 1,000 people aged 40-49.
“When you consider these marriages that end, in addition to marriages that end when one spouse dies, there is a potential for individuals who previously had a low risk of HPV acquisition now entering into new potential sexual relationships,” Dr. Bednarczyk said. “Additionally, it has been estimated that approximately 4% of the U.S. population are in open or consensually nonmonogamous relationships, where exposure to more sexual partners may increase their risk for HPV. These are just some examples of where conversations with health care providers, and shared clinical decision-making, can help with a targeted reduction of HPV risk.”
The ACIP recommendation regarding adults aged 27-45 years also provides people in this age group with insurance coverage for the HPV vaccine if they choose to get it, Dr. Bednarczyk pointed out. Insurance companies may not be required to cover HPV vaccination in people aged over 26 years without the CDC’s recommendation, even if it’s not for routine immunization.
Dr. Bednarczyk agreed, however, with how the ACS adapted the CDC’s recommendation for routine vaccination in youth. The CDC’s routine recommendation is at ages 11-12 but can begin at 9 years, according to the ACIP. The ACS guidance qualifies this statement to place more emphasis on encouraging the vaccine earlier.
“Routine HPV vaccination between ages 9-12 is expected to achieve higher on-time vaccination rates, resulting in increased numbers of cancers prevented,” according to the ACS. “Health care providers are encouraged to start offering the HPV vaccine at age 9 or 10.”
Dr. Bednarczyk pointed to some of his past research finding low proportions of teens fully vaccinated against HPV by age 13 years (J Infect Dis. 2019 Jul 31;220[5]:730-4). Therefore, “any efforts to encourage vaccination, including starting the series at ages 9-10 years may help,” he said.
He also agreed that there may be diminished effectiveness with vaccinating adults aged 22-26, “but this should also be considered relative to an individual’s risk of acquiring HPV.”
While an HPV vaccine shortage is a major concern and HPV vaccination efforts should remain most focused on young teens, adults should not necessarily be neglected, Dr. Bednarczyk noted.
“Given how common HPV infection is in the population, open discussion between patients and health care providers can help identify those adults for whom HPV vaccination can be effective,” he said.
The development of the ACS guideline was supported by ACS operational funds. The ACS has received an independent educational grant from Merck Sharp & Dohme for a project intended to increase HPV vaccination rates. Dr. Saslow is the principal investigator for a cooperative agreement between the ACS and the CDC to support the National HPV Vaccination Roundtable and is coprincipal investigator of a cooperative agreement between the ACS and CDC to support initiatives to increase HPV vaccination. The remaining authors and Dr. Bednarczyk reported no relevant disclosures.
SOURCE: Saslow D et al. CA Cancer J Clin. 2020 Jul 8. doi: 10.3322/caac.21616.
FROM CA: A CANCER JOURNAL FOR CLINICIANS
COVID-19–related skin changes: The hidden racism in documentation
Belatedly, the disproportionate impact of COVID-19 on patients of color is getting attention. By now, we’ve read the headlines. Black people in the United States make up about 13% of the population but account for almost three times (34%) as many deaths. This story repeats – in other countries and in other minority communities.
Early detection is critical both to initiate supportive care and to isolate affected individuals and limit spread. Skin manifestations of COVID-19, especially those that occur early in the disease (eg, vesicular eruptions) or have prognostic significance (livedo, retiform purpura, necrosis), are critical to this goal of early recognition.
In this context, a recent systematic literature review looked at all articles describing skin manifestations associated with COVID-19. The investigators identified 46 articles published between March and May 2020 which included a total of 130 clinical images.
The following findings from this study are striking:
- 92% of the published images of COVID-associated skin manifestations were in I-III.
- Only 6% of COVID skin lesions included in the articles were in patients with skin type IV.
- None showed COVID skin lesions in skin types V or VI.
- Only six of the articles reported race and ethnicity demographics. In those, 91% of the patients were White and 9% were Hispanic.
These results reveal a critical lack of representative clinical images of COVID-associated skin manifestations in patients of color. This deficiency is made all the more egregious given the fact that patients of color, including those who are Black, Latinx, and Native American, have been especially hard hit by the COVID-19 pandemic and suffer disproportionate disease-related morbidity and mortality.
As the study authors point out, skin manifestations in people of color often differ significantly from findings in White skin (for example, look at the figure depicting the rash typical of Kawasaki disease in a dark-skinned child compared with a light-skinned child). It is not a stretch to suggest that skin manifestations associated with COVID-19 may look very different in darker skin.
This isn’t a new phenomenon. Almost half of dermatologists feel that they’ve had insufficient exposure to skin disease in darker skin types. Skin of color remains underrepresented in medical journals.
Like other forms of passive, institutional racism, this deficiency will only be improved if dermatologists and dermatology publications actively seek out COVID-associated skin manifestations in patients of color and prioritize sharing these images. A medical student in the United Kingdom has gotten the ball rolling, compiling a handbook of clinical signs in darker skin types as part of a student-staff partnership at St. George’s Hospital and the University of London. At this time, Mind the Gap is looking for a publisher.
Dr. Lipper is an assistant clinical professor at the University of Vermont, Burlington, and a staff physician in the department of dermatology at Danbury (Conn.) Hospital. He has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Belatedly, the disproportionate impact of COVID-19 on patients of color is getting attention. By now, we’ve read the headlines. Black people in the United States make up about 13% of the population but account for almost three times (34%) as many deaths. This story repeats – in other countries and in other minority communities.
Early detection is critical both to initiate supportive care and to isolate affected individuals and limit spread. Skin manifestations of COVID-19, especially those that occur early in the disease (eg, vesicular eruptions) or have prognostic significance (livedo, retiform purpura, necrosis), are critical to this goal of early recognition.
In this context, a recent systematic literature review looked at all articles describing skin manifestations associated with COVID-19. The investigators identified 46 articles published between March and May 2020 which included a total of 130 clinical images.
The following findings from this study are striking:
- 92% of the published images of COVID-associated skin manifestations were in I-III.
- Only 6% of COVID skin lesions included in the articles were in patients with skin type IV.
- None showed COVID skin lesions in skin types V or VI.
- Only six of the articles reported race and ethnicity demographics. In those, 91% of the patients were White and 9% were Hispanic.
These results reveal a critical lack of representative clinical images of COVID-associated skin manifestations in patients of color. This deficiency is made all the more egregious given the fact that patients of color, including those who are Black, Latinx, and Native American, have been especially hard hit by the COVID-19 pandemic and suffer disproportionate disease-related morbidity and mortality.
As the study authors point out, skin manifestations in people of color often differ significantly from findings in White skin (for example, look at the figure depicting the rash typical of Kawasaki disease in a dark-skinned child compared with a light-skinned child). It is not a stretch to suggest that skin manifestations associated with COVID-19 may look very different in darker skin.
This isn’t a new phenomenon. Almost half of dermatologists feel that they’ve had insufficient exposure to skin disease in darker skin types. Skin of color remains underrepresented in medical journals.
Like other forms of passive, institutional racism, this deficiency will only be improved if dermatologists and dermatology publications actively seek out COVID-associated skin manifestations in patients of color and prioritize sharing these images. A medical student in the United Kingdom has gotten the ball rolling, compiling a handbook of clinical signs in darker skin types as part of a student-staff partnership at St. George’s Hospital and the University of London. At this time, Mind the Gap is looking for a publisher.
Dr. Lipper is an assistant clinical professor at the University of Vermont, Burlington, and a staff physician in the department of dermatology at Danbury (Conn.) Hospital. He has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Belatedly, the disproportionate impact of COVID-19 on patients of color is getting attention. By now, we’ve read the headlines. Black people in the United States make up about 13% of the population but account for almost three times (34%) as many deaths. This story repeats – in other countries and in other minority communities.
Early detection is critical both to initiate supportive care and to isolate affected individuals and limit spread. Skin manifestations of COVID-19, especially those that occur early in the disease (eg, vesicular eruptions) or have prognostic significance (livedo, retiform purpura, necrosis), are critical to this goal of early recognition.
In this context, a recent systematic literature review looked at all articles describing skin manifestations associated with COVID-19. The investigators identified 46 articles published between March and May 2020 which included a total of 130 clinical images.
The following findings from this study are striking:
- 92% of the published images of COVID-associated skin manifestations were in I-III.
- Only 6% of COVID skin lesions included in the articles were in patients with skin type IV.
- None showed COVID skin lesions in skin types V or VI.
- Only six of the articles reported race and ethnicity demographics. In those, 91% of the patients were White and 9% were Hispanic.
These results reveal a critical lack of representative clinical images of COVID-associated skin manifestations in patients of color. This deficiency is made all the more egregious given the fact that patients of color, including those who are Black, Latinx, and Native American, have been especially hard hit by the COVID-19 pandemic and suffer disproportionate disease-related morbidity and mortality.
As the study authors point out, skin manifestations in people of color often differ significantly from findings in White skin (for example, look at the figure depicting the rash typical of Kawasaki disease in a dark-skinned child compared with a light-skinned child). It is not a stretch to suggest that skin manifestations associated with COVID-19 may look very different in darker skin.
This isn’t a new phenomenon. Almost half of dermatologists feel that they’ve had insufficient exposure to skin disease in darker skin types. Skin of color remains underrepresented in medical journals.
Like other forms of passive, institutional racism, this deficiency will only be improved if dermatologists and dermatology publications actively seek out COVID-associated skin manifestations in patients of color and prioritize sharing these images. A medical student in the United Kingdom has gotten the ball rolling, compiling a handbook of clinical signs in darker skin types as part of a student-staff partnership at St. George’s Hospital and the University of London. At this time, Mind the Gap is looking for a publisher.
Dr. Lipper is an assistant clinical professor at the University of Vermont, Burlington, and a staff physician in the department of dermatology at Danbury (Conn.) Hospital. He has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.







