User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
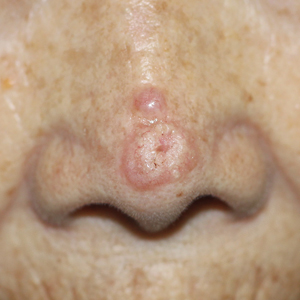
Solitary Papule on the Upper Back
Solitary Papule on the Upper Back
THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
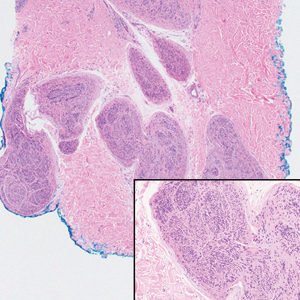
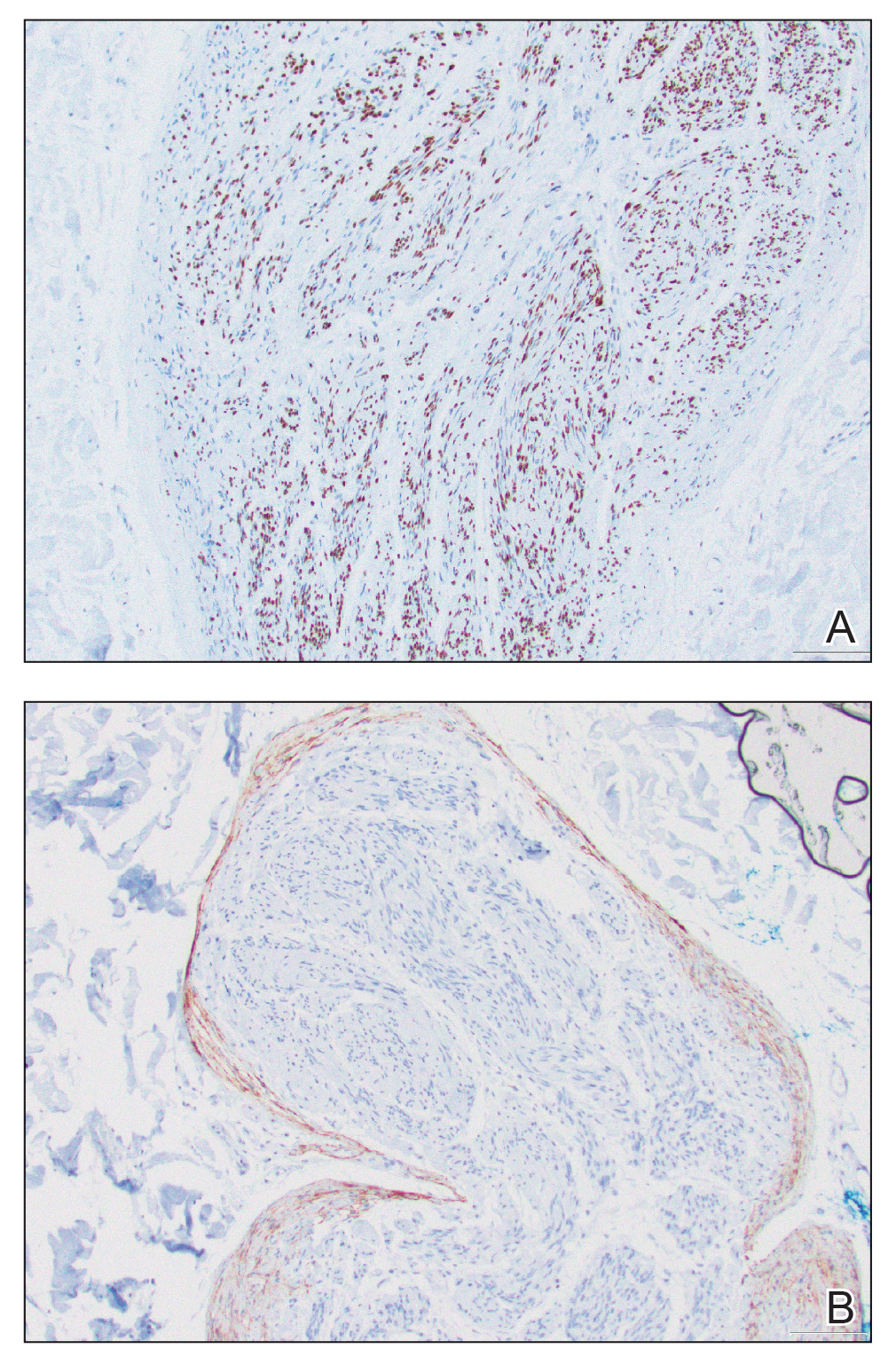
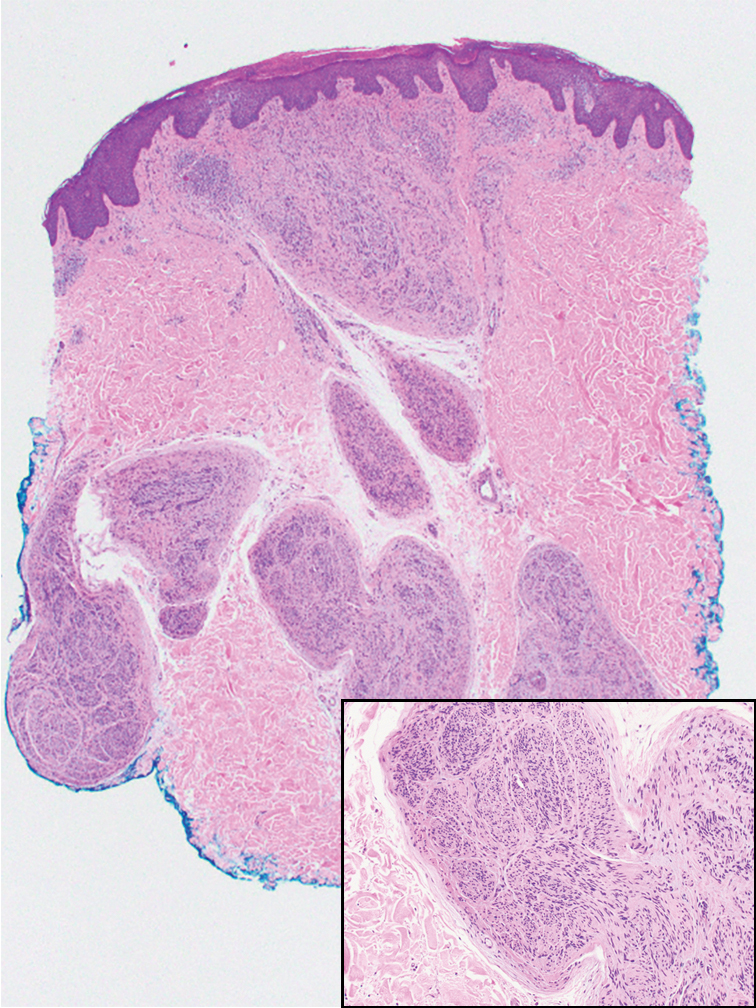
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.
Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
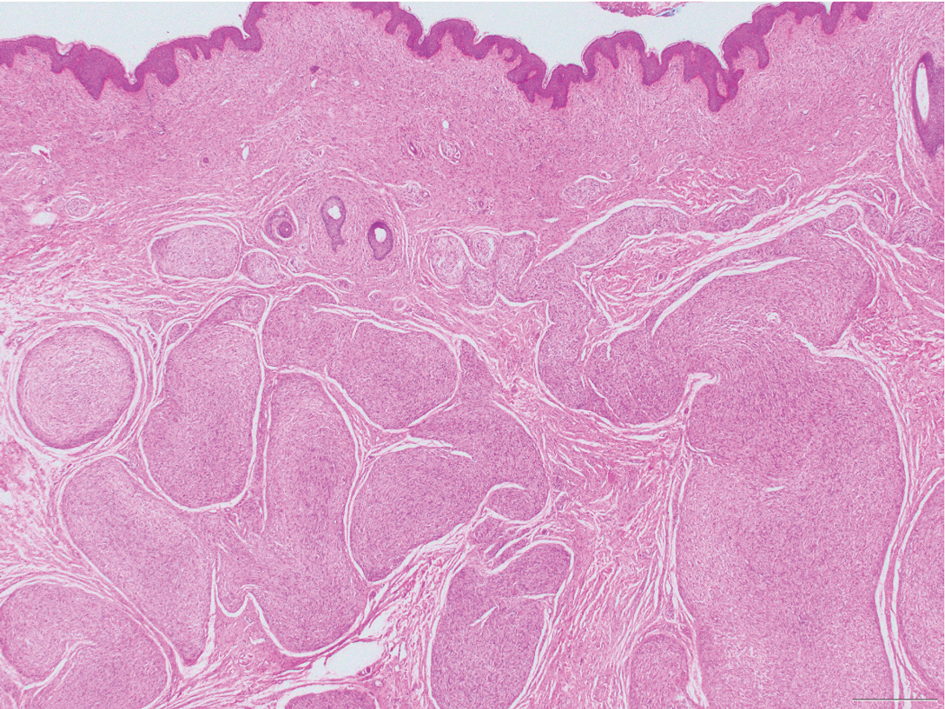
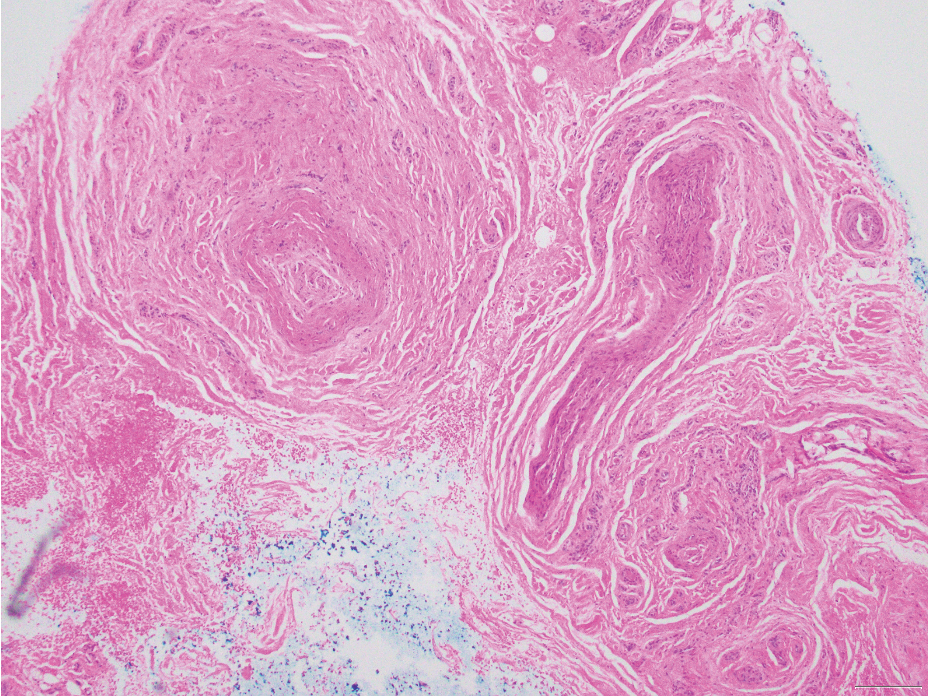
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.
Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12
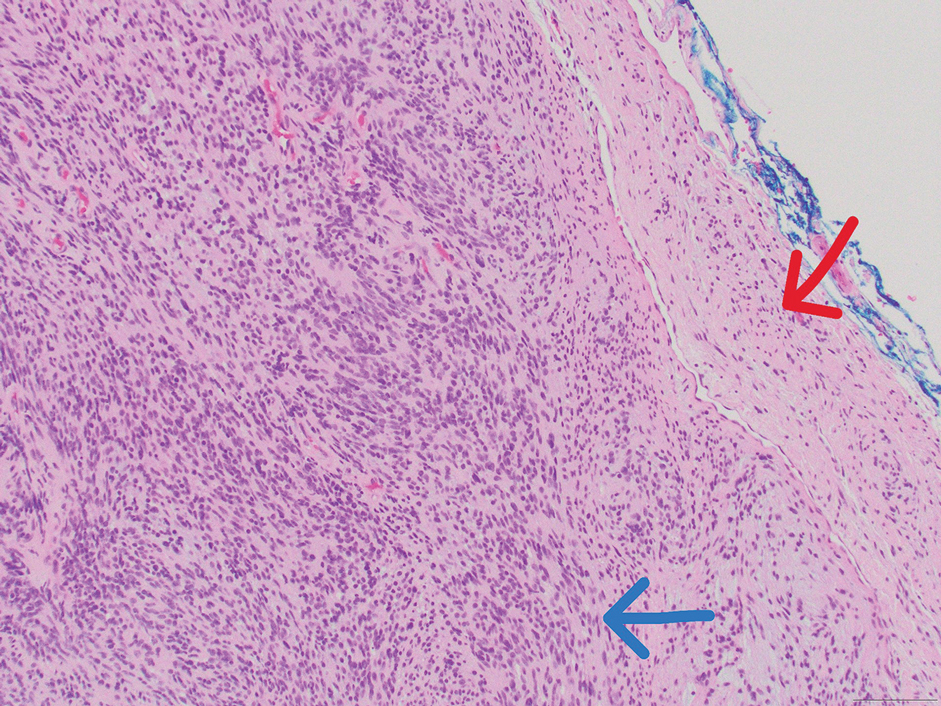
Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.
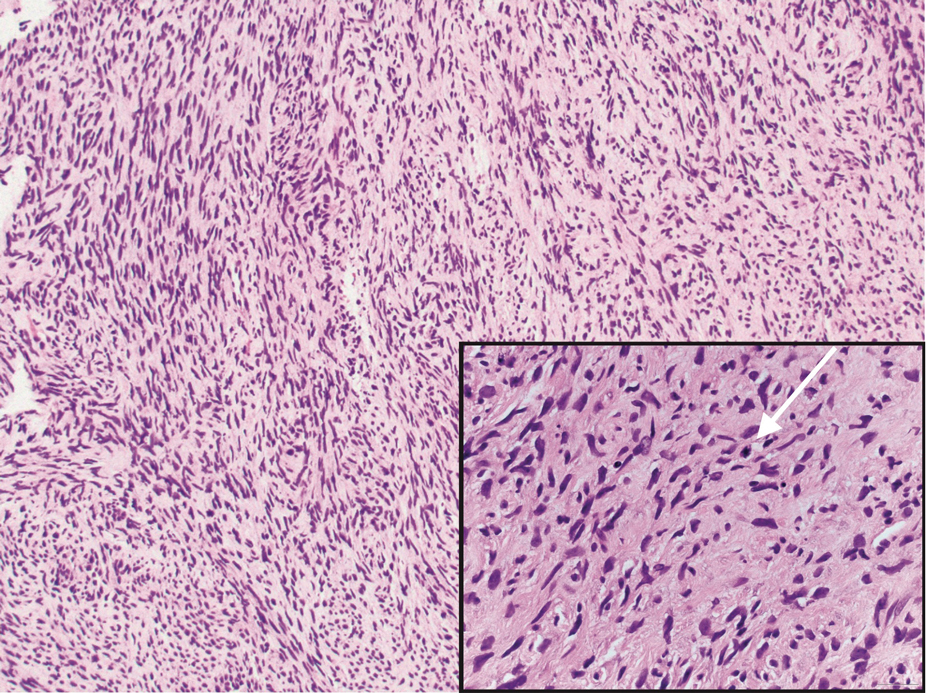
Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.
- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.

Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.

Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12

Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.

Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.

THE DIAGNOSIS: Plexiform Palisaded Encapsulated Neuroma
Microscopically, there was a superficial to deep dermal proliferation of tapered spindle cells in fascicles that were well circumscribed in nodules throughout the dermis with pale background stroma, mild mucin, and a thin capsule. The tapered spindle cells stained positive for SOX-10 and negative for Melan-A (Figure 1A). Staining for epithelial membrane antigen highlighted delicate cells around the periphery of the nodules, consistent with perineurium (Figure 1B). A diagnosis of plexiform palisaded encapsulated neuroma was made. No additional treatment was pursued due to the benign nature of the condition.

Palisaded encapsulated neuroma (PEN), also referred to as solitary circumscribed neuroma,1 is a benign, generally solitary neurogenic tumor that manifests predominantly on the skin, particularly in areas of frequent outside trauma such as the face. Lesions also may occur on mucosal and acral sites.2 First described by Reed et al3 in 1972, PEN characteristically manifests as a well-circumscribed, dermal nodule with a distinctive palisading pattern of Schwann cells and axons within a delicate perineurial capsule, the latter of which may be incomplete.3 Palisaded encapsulated neuroma frequently exhibits clefting between the tumor and the surrounding dermis. While PEN generally is sporadic, rare cases have been reported in association with Cowden syndrome and neurofibromatosis type 2.4,5
While the nodular growth pattern is most common, PEN also may present in epithelioid, plexiform, multinodular, or fungating subtypes.6 The plexiform subtype of PEN is rare. It has a complex growth pattern and a tendency to involve multiple adjacent nerve bundles in a plexiform arrangement.6,7 In two independent reviews characterizing the predominant growth patterns of PEN, nonnodular growth patterns were observed in a minority of the 85 cases: 18.8% (16/85) were plexiform, 7.1% (6/85) were multinodular or multilobular, and 7.1% (6/85) were fungating.6,7
The clinical presentation of plexiform PEN often includes a painless, slow-growing mass, and it predominantly occurs in middle-aged adults.2 Immunohistochemical staining reveals diffuse positivity for SOX-10 and S-100, which highlights the neural origin of the tumor.6 This variant, like conventional PEN, lacks notable atypia or mitotic activity.
Palisaded encapsulated neuroma, regardless of subtype, has an excellent prognosis, with no known cases of malignant transformation, and surgical excision with clear margins is curative.8
The differential diagnosis for plexiform PEN includes plexiform variants of neurofibroma and schwannoma, traumatic neuroma, and malignant peripheral nerve sheath tumor.
Neurofibromas are nonencapsulated lesions composed of spindle cells with wavy nuclei dispersed in a myxoid background.8 Neurofibromas can manifest in various locations throughout the body, including the skin, subcutaneous tissues, and internal organs. They are slow-growing tumors but may accelerate during periods of hormonal changes, such as pregnancy and puberty, or in cases of malignant transformation.8 Although plexiform neurofibromas are benign, malignant transformation can occur, particularly in patients with neurofibromatosis type 1 (NF1).8,9 Neurofibromas may assume one of 3 growth patterns: localized, diffuse, or plexiform.8 Plexiform neurofibromas exhibit a multinodular, ropelike growth pattern with a mix of Schwann cells and fibroblasts (Figure 2).8,9 These lesions are pathognomonic for NF1 and can infiltrate the surrounding tissue. They may involve large nerve trunks, leading to a more complex growth pattern compared to solitary neurofibromas.8,9 The plexiform variants of both neurofibromas and PEN demonstrate a multinodular growth pattern; however, plexiform neurofibromas are nonencapsulated and show a more diffuse infiltrative nature, whereas plexiform PEN remains well circumscribed. Additionally, plexiform neurofibromas are associated with NF1, while plexiform PEN lacks this genetic association.

Schwannomas are encapsulated tumors that originate from the outer sheath of peripheral nerves, usually positioned eccentrically to the nerve fibers. Schwannomas are characterized by Antoni A and Antoni B areas, which usually are absent in PEN. Antoni A areas are composed of compact spindle cells arranged in palisades with Verocay bodies, while Antoni B areas are more loosely arranged and have a myxoid background (Figure 3).8,9 Schwannomas stain positive for S-100 and often show degenerative changes such as cystic degeneration or calcification, particularly in larger lesions.8,9 Plexiform schwannoma is a rare variant of schwannoma, and while it carries a substantial risk for local recurrence with rates as high as 50%, it has not been shown to possess malignant or metastatic potential.10 Unlike PEN, schwannomas have a consistent capsule but share S-100 positivity with PEN. Verocay bodies occasionally can be observed in PENs, with studies reporting their presence in 20% to 36% of cases.7,11,12 Additionally, some schwannomas may exhibit few Verocay bodies or poorly developed forms, which can make histopathologic distinction more challenging.7,11,12

Traumatic neuromas result from nerve regeneration following any type of outside trauma that is deep enough to cause nerve injury. The lesion often is painful and associated with prior trauma or surgery. Under optimal conditions, the severed ends of a nerve reconnect through the orderly growth of axons from the proximal stump to the distal stump, guided by tubes formed by proliferating Schwann cells. If the nerve ends are not properly aligned or if the distal stump is absent, the axons may proliferate in a disorganized manner at the proximal stump, resulting in the formation of a traumatic neuroma.8 Histologically, these lesions exhibit disorganized, proliferating nerve fibers intermixed with fibrous stroma.8,13 The nerve fibers are not encapsulated, and there is an irregular arrangement of axons and Schwann cells (Figure 4).8,13 Unlike PEN, which usually is encapsulated and well organized with fascicular architecture, traumatic neuromas exhibit a disorganized, haphazard arrangement of neural elements and lack a capsule.8 Clinically, traumatic neuromas also are more likely to be painful.

Malignant peripheral nerve sheath tumors are aggressive malignant spindle-cell tumors that may be associated with NF1 or occur sporadically.9,14 The spindle cells are arranged in fascicles, and these tumors can have areas of necrosis, hemorrhage, and high mitotic activity.9,15 The spindle cells may be arranged in a herringbone pattern, and alternating areas of hypocellularity and hypercellularity impart a marbled appearance (Figure 5).16 Malignant peripheral nerve sheath tumors frequently exhibit inactivation of the SWI/SNF-related, matrix- associated, actin-dependent regulator of chromatin subfamily B member 1 gene and loss of integrase interactor 1 protein. Transformation from plexiform neurofibroma to malignant peripheral nerve sheath tumor frequently is accompanied by progressive genomic changes.17 Malignant peripheral nerve sheath tumors differ substantially from PEN in their aggressive histologic features, including nuclear atypia and mitotic figures, which are absent in PEN.

- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
- Fletcher CD. Solitary circumscribed neuroma of the skin (so-called palisaded, encapsulated neuroma). a clinicopathologic and immunohistochemical study. Am J Surg Pathol. 1989;13:574-580. doi:10.1097/00000478-198907000-00005
- Jokinen CH, Ragsdale BD, Argenyi ZB. Expanding the clinicopathologic spectrum of palisaded encapsulated neuroma. J Cutan Pathol. 2010;37:43-48. doi:10.1111/j.1600-0560.2009.01380.x
- Reed RJ, Fine RM, Meltzer HD. Palisaded encapsulated neuromas of the skin. Arch Dermatol. 1972;106:865-870.
- Harris E, Mir A. Acral plexiform palisaded encapsulated neuromas as the initial cutaneous manifestation of Cowden syndrome. Pediatr Dermatol. 2017;34:E219-E220. doi:10.1111/pde.13161
- Arole V, Shaker N, Kim LR, et al. Multiple cutaneous solitary circumscribed neuroma in a patient with neurofibromatosis type 2: an “incidentaloma” or new association?. Int J Surg Pathol. 2023;31:734-737. doi:10.1177/10668969221120782
- Argenyi ZB, Cooper PH, Cruz DS. Plexiform and other unusual variants of palisaded encapsulated neuroma. J Cutan Pathol. 1993;20:34-39. doi:10.1111/j.1600-0560.1993.tb01246.x
- Leblebici C, Savli TC, Yeni B, et al. Palisaded encapsulated (solitary circumscribed) neuroma: a review of 30 cases. Int J Surg Pathol. 2019;27:506-514. doi:10.1177/1066896919833172
- Weiss SW, Goldblum JR. Enzinger and Weiss’s Soft Tissue Tumors. 6th ed. Elsevier Saunders; 2014.
- Rodriguez FJ, Folpe AL, Giannini C, et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol. 2012;123:295-319. doi:10.1007 /s00401-012-0954-z
- Berg JC, Scheithauer BW, Spinner RJ, et al. Plexiform schwannoma: a clinicopathologic overview with emphasis on the head and neck region. Hum Pathol. 2008;39:633-640. doi:10.1016 /j.humpath.2007.10.029
- Koutlas IG, Scheithauer BW. Palisaded encapsulated (“solitary circumscribed”) neuroma of the oral cavity: a review of 55 cases. Head Neck Pathol. 2010;4:15-26. doi:10.1007/s12105-010-0162-x
- Kossard S, Kumar A, Wilkinson B. Neural spectrum: palisaded encapsulated neuroma and verocay body poor dermal schwannoma. J Cutan Pathol. 1999;26:31-36. doi:10.1111/j.1600-0560.1999 .tb01787.x
- Yang H, Dong Y, Wang Z, et al. Traumatic neuromas of peripheral nerves: diagnosis, management and future perspectives. Front Neurol. 2023;13:1039529. doi:10.3389/fneur.2022.1039529
- Knight SWE, Knight TE, Santiago T, et al. Malignant peripheral nerve sheath tumors-a comprehensive review of pathophysiology, diagnosis, and multidisciplinary management. Children (Basel). 2022;9:38. doi:10.3390/children9010038
- Perry A, Gutmann DH. Malignant peripheral nerve sheath tumors: clinical and genetic aspects of pathogenesis. Clin Neuropathol. 2000;19:105-114.
- Lindberg G, Lucas D, Cassarino D, et al, eds. Diagnostic Pathology: Soft Tissue Tumors. 3rd ed. Elsevier; 2023.
- Miettinen MM, Antonescu CR, Fletcher CDM, et al. Histopathologic evaluation of atypical neurofibromatous tumors and their transformation into malignant peripheral nerve sheath tumor in patients with neurofibromatosis 1-a consensus overview. Hum Pathol. 2017;67:1-10. doi:10.1016/j.humpath.2017.05.010
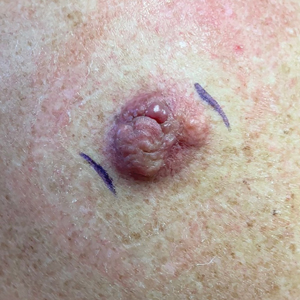
Solitary Papule on the Upper Back
Solitary Papule on the Upper Back
An 88-year-old woman presented to the dermatology clinic with an asymptomatic papule on the left upper back of unknown duration. The patient reported that her medical history was negative for eczematous dermatitis, hypertension, and osteoarthritis. Physical examination revealed a firm, well-circumscribed, flesh-colored, 6-mm papule with no overlying scale or ulceration. No other concerning lesions were noted on full skin examination. A punch biopsy of the papule was performed.
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
Bullous pemphigoid (BP) is a chronic autoimmune skin condition characterized by large, fluid-filled blisters (bullae) on the skin. There usually is severe itching, which may antedate the bullae by several weeks. Bullous pemphigoid most commonly affects adults aged 60 years and older with comorbidities such as metabolic syndrome or other autoimmune diseases. The typical blistering is subepidermal, and its occurrence seems to be driven by autoantibodies targeting the epidermal basement membrane zone.
Conventional treatment for BP has long included systemic corticosteroids and immunosuppressants; this combination of drugs effectively controls inflammation but also is associated with considerable risks such as infection, hyperglycemia with worsening diabetes, hypertension, osteoporosis and fracture risk, and delirium, particularly in older patients. Recent developments in biologic therapy have supported the utilization of potentially safer alternatives. Specifically, dupilumab has demonstrated efficacy in the treatment of atopic dermatitis. Case reports and small research studies included in a recent review have suggested that dupilumab could be effective in managing BP, potentially offering a safer treatment option.1 Importantly, dupilumab recently was approved for BP by the US Food and Drug Administration, making it the first biologic approved for treating this condition.2 In this article, we discuss dupilumab as an option for clinicians to prescribe as an initial therapy for BP.
Pathophysiology of Bullous Pemphigoid and Dupilumab
Bullous pemphigoid pathophysiology is driven by 2 main components: immunologic and inflammatory processes. The immune-mediated attack consists of autoantibodies against the hemidesmosomal proteins BP180 and BP230 in the basement membrane zone, which anchor the epidermis to the dermis.1 When these proteins are targeted, a cascade of responses is triggered, including the release of inflammatory cells, particularly eosinophils and neutrophils, into the skin. These cells release inflammatory mediators that break down the basement membrane, resulting in the separation of the epidermis from the dermis and leading to subepidermal blister formation.
The inflammation associated with BP largely is driven by a T helper 2–dominant immune response, with increased levels of cytokines such as interleukin (IL) 4 and IL-13.1 The chronic inflammation contributes to the characteristic blisters and intense pruritus seen in patients with BP. Traditional treatments such as corticosteroids work broadly to suppress this inflammation by reducing the production of all proinflammatory cytokines; however, they lack specificity, which dampens the targeted immune response against BP and broadly suppresses the immune system, increasing the risk for infection and other adverse effects. For older patients with multiple health conditions, these adverse effects can impact quality of life and pose health risks.
Interleukin 4 and IL-13 contribute to eosinophilia by upregulating the expression of eosinophil chemoattractant cytokines that promote eosinophil migration and adhesion.1 This is where dupilumab presents a promising alternative. Dupilumab is a monoclonal antibody that selectively targets the key cytokines implicated in BP. It binds to the IL-4 receptor alpha subunit, which IL-4 and IL-13 share, thereby inhibiting the signaling of both cytokines.1
Dupilumab Reports and Case Series
Current evidence supporting the use of dupilumab for BP consists primarily of case series and uncontrolled clinical observations, both of which are useful to formulate but not to test hypotheses.3 One case series of 13 patients diagnosed with BP reported that 92.3% (12/13) of patients achieved disease clearance or satisfactory control, with 53.8% (7/13) achieving total lesion clearance and no adverse events observed.4 In a larger case series, 87.0% of 146 patients achieved disease control with an initial dose of dupilumab 600 mg followed by 300 mg every 2 weeks for 4 weeks, though the study also looked at secondary outcomes for up to 64 weeks.5 Both these findings are compatible with the idea that dupilumab has efficacy in BP, but they cannot be used to test hypotheses.3,4
In a single case report of refractory BP, the combination of dupilumab and omalizumab—a monoclonal antibody that blocks the binding of IgE to mast cells, basophils, and dendritic cells—has shown efficacy, particularly when BP is unresponsive to standard therapies.6 The case involved a 72-year-old woman with unresponsive BP who was treated with a combination of dupilumab and omalizumab. She experienced marked improvement in pruritus and bullae accompanied by normalization of anti-BP180 autoantibody levels. This result points to the possibility that IL-4 and IL-13 blockade could influence autoantibody production. The authors attributed the reduction in pruritus to dupilumab, although the drug was administered in combination with omalizumab.6
Small Randomized Trial of Dupilumab
In addition to these uncontrolled clinical observations, dupilumab recently demonstrated positive results in a small randomized trial.7 Patients who were assigned to receive dupilumab were 5 times more likely to achieve sustained remission compared to those assigned to a placebo. All randomized patients also were treated with a standard-of-care oral corticosteroid regimen, which complicated the ability to isolate the specific effects of dupilumab.7 Finally, a completed totality of evidence on the prescription of dupilumab for refractory BP should include more reliable results from larger randomized trials.8
Final Thoughts
Dupilumab is now approved by the US Food and Drug Administration for the treatment of BP, marking an important milestone as the first targeted biologic therapy for this condition. While the totality of evidence is still evolving, the available data indicate both efficacy and a favorable safety profile when compared to traditional corticosteroids and immunosuppressants. Based on the available data, we suggest that health care providers consider dupilumab as a first-line monotherapy in appropriate patients with BP without specific contraindications, particularly as a practical and safe alternative to corticosteroids and immunosuppressive therapies in older adults with comorbidities. If the patient demonstrates clinical improvement in symptom relief, the therapy can be continued. This strategy minimizes initial exposure to more potent biologics or therapies with higher toxicity profiles. For patients without adequate disease control or with severe initial presentations requiring rapid improvement, clinicians may consider escalation to adjunctive or alternative therapies, such as rituximab, omalizumab, or a short course of systemic corticosteroids for acute management.
Emerging data suggest that dupilumab is a promising therapy for BP. We suggest that it may be a safer targeted alternative to traditional corticosteroids and immunosuppressive therapies. The ability of dupilumab to mitigate inflammation without broadly suppressing the immune system suggests that it may turn out to be an especially valuable option for older patients and those with comorbid conditions.
Acknowledgement—The authors thank Jodi Fiedler, MD (Boca Raton, FL), for her expert advice and assistance.
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
- Zhang L, Chen Z, Wang L, et al. Bullous pemphigoid: the role of type 2 inflammation in its pathogenesis and the prospect of targeted therapy. Front Immunol. 2023;14:1115083. doi:10.3389/fimmu.2023.1115083
- Sanofi. Dupixent approved in the US as the only targeted medicine to treat patients with bullous pemphigoid. News release. June 20, 2025. Accessed September 8, 2025. https://www.sanofi.com/en/media-room/press-releases/2025/2025-06-20-05-00-00-3102518
- Hennekens CH, DeMets D. Statistical association and causation: contributions of different types of evidence. JAMA. 2011;306:1134-1136. doi:10.1001/jama.2011.322
- Abdat R, Waldman RA, de Bedout V, et al. Dupilumab as a novel therapy for bullous pemphigoid: a multicenter case series. J Am Acad Dermatol. 2020;83:46-52. doi:10.1016/j.jaad.2020.01.089
- Zhao L, Wang Q, Liang G, et al. Evaluation of dupilumab in patients with bullous pemphigoid. JAMA Dermatol. 2023;159:953-960. doi:10.1001/jamadermatol.2023.2428
- Seyed Jafari SM, Feldmeyer L, Bossart S, et al. Case report: combination of omalizumab and dupilumab for recalcitrant bullous pemphigoid. Front Immunol. 2021;11:611549. doi:10.3389/fimmu.2020.611549
- US National Library of Medicine. A multicenter, randomized, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of dupilumab in adult patients with bullous pemphigoid. ClinicalTrials.gov. Published 2019. Accessed March 10, 2026. https://clinicaltrials.gov/ct2/show/NCT04206553
- Hennekens CH, DeMets D. The need for large scale randomized evidence without undue emphasis on small trials, meta-analyses or subgroup analyses. JAMA. 2009;302:2361-2362. doi:10.1001/jama.2009.1829
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
Dupilumab for Bullous Pemphigoid: To Treat or Not to Treat?
PRACTICE POINTS
- Bullous pemphigoid (BP) is a pruritic autoimmune blistering disease in older adults that often requires systemic therapy with associated risks.
- Dupilumab targets interleukin (IL) 4/IL-13–driven inflammation and shows promising efficacy in BP.
- As the first approved biologic for BP, dupilumab may offer a safer alternative to corticosteroids in select patients.
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.

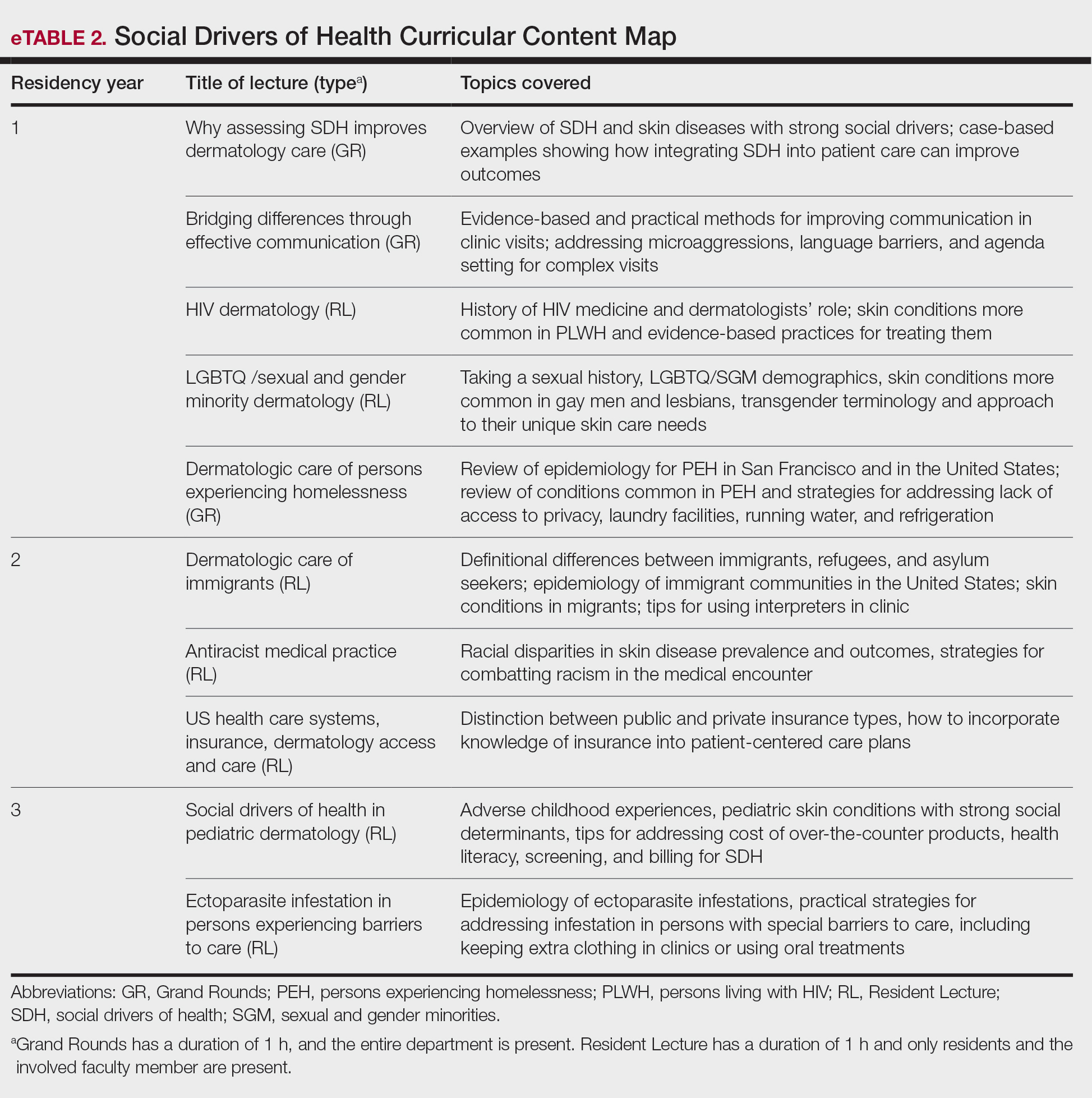
To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Practice Points
- Integrating a formal curriculum on social drivers of health, including didactics on structural racism, cultural humility, communication, and practical strategies, can help residents learn to routinely assess social needs and develop feasible patient-centered care plans.
- Classroom learning paired with experiential rotations and community engagement in safety-net hospitals can help to build empathy, practical skills, and comfort managing real-world social barriers.
- Creation of trainee leadership roles and dedicated program support (eg, departmental funding) can sustain curriculum improvements, foster advocacy skills, and diversify the workforce pipeline.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing an electronic medical record (EMR) is one of the most important clinical and financial decisions a dermatology practice will make. An effective system can help streamline workflows, support high-quality patient care, and protect revenue, while the wrong choice can slow clinicians down and add to the administrative burden.
Dermatology workflows involve unique documentation, imaging, and billing needs that are not always well served by generic EMR platforms. To help guide the selection of an EMR, the following framework outlines key features and practice considerations specific to dermatology practices.
While many general EMRs offer customization, dermatology practices benefit greatly from ready-built, specialty-specific documentation tools. Key elements to evaluate include the following:
- Preconfigured dermatology templates for common conditions and procedures (eg, acne, psoriasis, melanoma, biopsies, cosmetic treatments)
- Smart-phrase libraries tailored to dermatologic language and examinations
- Ability to create, modify, and share custom templates across providers
Why It Matters—Efficient charting reduces documentation time, improves consistency, and supports accurate coding.
Dermatology is a highly visual specialty, making clinical photography and image management essential. Important capabilities of an EMR include the following:
- Easy capture, annotation, and longitudinal tracking of clinical images
- Seamless embedding of photographs directly into the patient chart
- Side-by-side comparison of current and prior images
- Secure image storage and camera integration
- Body-mapping tools to mark and track lesion locations visually
Why It Matters—A high-quality image workflow supports diagnosis, treatment planning, patient education, and medicolegal documentation.
For insurance-based practices, robust billing and revenue cycle management (RCM) tools are critical. For direct-care models, some of these items may be prioritized lower. Key features to compare include the following:
- Support for International Classification of Diseases, 10th Revision, Clinical Modification; Current Procedural Terminology; and dermatology-specific code sets
- Automated coding suggestions tied to clinical documentation
- Reviewing claims for errors and inconsistencies prior to submitting to payers’ insurance eligibility verification
- Electronic Remittance Advice/Explanation of Benefits posting and denial management workflows
- Support for cosmetic and self-pay billing
- Ability to generate superbills (itemized receipts for medical services that include International Classification of Diseases Tenth Revision and Current Procedural Terminology codes; patients can submit these directly to their insurance company for reimbursement) for direct-pay practices
Why It Matters—Strong RCM functionality protects revenue, reduces denials, and minimizes staff workload.
The most effective EMRs tightly integrate clinical charting with daily practice operations. Features to evaluate include the following:
- Integrated scheduling with color-coded calendars
- Appointment-type templates and block scheduling
- Automated patient reminders via text or email
- Support for multiprovider and multilocation practices
- Integration with outside pathology or lab services
Why It Matters—Clear and templated scheduling and practice integration help practices run more smoothly by reducing administrative workload and errors and coordinating communication between providers and even ancillary services.
Patient communication and virtual care are increasingly important in dermatology. When evaluating EMRs, compare the following:
- Built-in telehealth functionality vs third-party integrations
- Automated appointment reminders
- Patient portal features (forms, messaging, results)
- Online booking capabilities
Why It Matters—Integrated telehealth and patient communication tools improve access to care, enhance patient engagement, and streamline scheduling, messaging, and virtual visits within dermatology workflows.
Reporting capabilities support clinical quality, compliance, and business decision-making. Key reporting areas include the following:
- Clinical reports (outcomes, lesion tracking, disease management)
- Financial reports (revenue per provider, payer mix, visit types)
- Customizable or exportable reporting tools
Why It Matters—Robust reporting and analytics help dermatology practices track clinical outcomes, monitor financial performance, and make data-driven decisions to improve both patient care and practice management.
The user experience after implementation of the EMR is just as important as the software itself. Evaluate the following after the EMR is implemented:
- Initial training and onboarding resources
- Availability of dermatology-specific support teams
- Ongoing education, help centers, or user communities
- Access to dedicated implementation or success managers
Why It Matters—Strong training and support resources help ensure a smoother EMR implementation, faster staff adoption, and ongoing optimization of the system for dermatology workflows.
Finally, look beyond the sticker price. The total cost of ownership includes far more than monthly fees. Compare the following:
- Upfront costs (implementation, data migration, training)
- Subscription pricing (per provider or per user)
- Billing or RCM fees (including percentages of collections if applicable) and payment processing fees
- Costs for add-on modules (telehealth, imaging, analytics)
- Contract length and termination terms
Why It Matters—Understanding the full cost of ownership helps dermatology practices choose an EMR that fits their budget long-term while avoiding unexpected fees and contractual limitations.
There is no single “best” EMR for every dermatology practice. The right choice depends on your practice model, payer mix, clinical focus, and growth plans. By evaluating EMRs through a dermatology-specific lens and asking the right questions, you can choose a system that supports both excellent patient care and long-term practice success.
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
Choosing the Right Electronic Medical Record: Key Features and Considerations for Dermatology Practices
PRACTICE POINTS
- Choosing an electronic medical record (EMR) built for dermatology workflow is a critical part of practice management.
- Features of an EMR that should be evaluated include support for clinical documentation, scheduling and billing, and customer support.
- The proper EMR can reduce administrative tasks and protect practice revenue, but there is no one-size-fits-all option.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
Melasma is a chronic hyperpigmentation disorder with a high recurrence rate. Advances in pathogenesis, diagnosis, and treatment have shifted dermatologic approaches for both initial management and long-term care. These advances highlight the need to move beyond conventional paradigms for melasma to adopt a more comprehensive and holistic approach to evaluation and management.
Diagnosis: Beyond the Wood Lamp
Diagnosis of melasma is primarily clinical, based on visual examination, with tools such as a Wood lamp (320-400 nm) aiding in classification as epidermal, dermal, or mixed types.1 Dermoscopy is another useful tool that commonly demonstrates a brown reticular pseudonetwork, vascular patterns, and brown clods.2 Reflectance confocal microscopy allows detailed assessment of pigment distribution across skin layers and may serve as a useful diagnostic adjunct.3 Two-photon excitation microscopy also has shown concordance with reflectance confocal microscopy in identifying key pathologic features of melasma and allows quantitative assessment of pigment burden.4,5 Biopsy remains warranted in cases when the diagnosis is unclear.6,7 These advanced tools provide additional options for noninvasive imaging of melasma, which may be useful during both diagnosis and treatment.
Pathogenesis: Beyond the Melanocyte
Recent insights into pathogenesis have shifted the view of melasma from a purely melanocyte-driven disorder to one involving complex epidermal-dermal interactions influenced by chronic UV and visible light exposure and sustained cutaneous inflammation. Evidence suggests involvement of additional cell types and structural components, including epidermal barrier dysfunction, basement membrane alterations, senescent fibroblasts, mast cell activity, vascular remodeling, and dermal solar elastosis. Senescent fibroblasts secrete melanogenic and angiogenic mediators (including stem cell factor, vascular endothelial growth factor, endothelin 1, and matrix metalloproteinases) while mast cell degranulation releases histamine, tryptase, and other proteases that drive melanocyte activation, extracellular matrix degradation, and persistent hyperpigmentation.8 New research highlights the importance of epidermal-dermal communication in melasma disease activity.8,9 Exposure to UV and visible light leads to keratinocyte injury and fibroblast dysfunction, promoting the release of inflammatory and melanogenic mediators that stimulate melanocytes and weaken the basement membrane.8 Mast cell activation and vascular signaling also have been implicated, reinforcing the contribution of inflammation and dermal remodeling to ongoing pigmentation.9 This broader understanding helps explain the difficulty of sustaining long-term remission.8,9 The multifaceted and evolving understanding of melasma’s complex pathogenesis highlights areas for future therapeutic targets beyond melanin, which may help lead to greater clearance and remission periods.
Treatment: Beyond Hydroquinone
Melasma treatment has shifted toward a longitudinal approach reflecting its chronic relapsing nature. Effective management should include combination therapy of broad-spectrum photoprotection, topical therapies, systemic therapies (when appropriate), and adjunctive procedural modalities, while emphasizing the importance of maintenance therapy and relapse prevention.10,11 Growing evidence demonstrates that UV and visible light contribute to melanogenesis and prolonged pigmentation, particularly in individuals with skin of color.12,13 Visible light, particularly within the high-energy blue wavelength spectrum (400-500 nm), induces reactive oxygen species and stimulates melanogenesis, resulting in persistent hyperpigmentation and exacerbation of melasma in darker skin types.13 Iron oxide, a mineral-based physical filter commonly used in tinted mineral sunscreens, enhances protection from visible light, thus improving melasma outcomes.12,13 In a randomized double-blind placebo-controlled trial, oral polypodium leucotomos extract, an antioxidant with photoprotective effects, improved Melasma Area and Severity Index (MASI)/modified MASI (mMASI) scores and melasma-rated quality of life measures when used as an adjunct to sunscreen with or without topical hydroquinone.14
Topical therapy serves as the mainstay of melasma treatment. Hydroquinone- and retinoid-based regimens including triple-combination therapy remain an effective gold standard for active disease; however, there still are concerns about irritant dermatitis with both as well as ochronosis with long-term use of hydroquinone.15 Recent studies highlight the efficacy of nonhydroquinone topical therapies such as azelaic acid, tranexamic acid (TXA), thiamidol, cysteamine, metformin, malassezin, 2-mercaptonicotinoyl glycine, niacinamide, kojic acid, ascorbic acid, and botanical-derived compounds, many of which demonstrate reductions in disease severity and more favorable tolerability profiles.16-19 Combinations of these ingredients can allow for addressing the various pathogenic factors of melasma beyond pigmentation, including vascularity, inflammation, and solar damage.
Systemic therapy, particularly oral TXA, has emerged as an option for patients with moderate to severe or refractory melasma. Clinical trials and observational studies demonstrate improvement with oral TXA; however, relapse after discontinuation is common, emphasizing the importance of careful patient selection, established treatment duration, and concurrent maintenance therapy.16,20 Although it is well tolerated, adverse effects include gastrointestinal discomfort, menstrual irregularities, and headache. Additionally, prescreening for contraindications and clotting risk factors is needed to avoid thromboembolic events.21
Procedural interventions are best used as adjunctive therapies rather than primary treatment. Superficial and medium peels can be used depending on baseline skin type.22 Platelet-rich plasma is a promising adjunctive therapy for melasma, demonstrating reductions in MASI scores following intradermal or microneedling-assisted delivery, likely mediated through growth factor–driven inhibition of melanogenesis and modulation of tyrosinase activity. Platelet-rich plasma promotes dermal remodeling through angiogenesis, collagen synthesis, and extracellular matrix production improving skin texture and pigmentation.23 Botulinum toxin A may improve pigmentation by modulating melanogenesis as well as inflammatory and vascular pathways.24
Radiofrequency microneedling provides a dermal-targeting therapeutic approach, with some evidence showing that monthly treatments can help maintain improvements achieved with conventional therapy by modulating photoaged dermal structures impacted by melanogenic signaling.25 Histopathologic correlation studies suggest that microneedling used alone or as a delivery platform alongside topical agents may be beneficial in dermal-predominant disease, while laser-based therapies should be reserved for recalcitrant cases due to the risk for postinflammatory hyperpigmentation and melasma recurrence, particularly in those with darker skin types.26 Lasers such as Q-switched 1064-nm Nd:YAG as well as nonablative lasers used in conservative settings are options for melasma treatment, including in patients with skin of color.26,27 Laser toning with serial low-fluence (<3 J/cm2), large-spot 1064-nm Nd:YAG has been used to reduce inflammation and risk for postinflammatory hyperpigmentation.28 Additionally, randomized split-face data demonstrate that picosecond 1064-nm devices achieve comparable reductions in mMASI scores to traditional nanosecond devices, and combination strategies incorporating intense pulsed light (IPL) have shown greater MASI improvement compared with IPL alone.28,29 However, IPL generally is limited to lighter skin phototypes due to the risk for postinflammatory hyperpigmentation in darker skin.30
Lastly, long-term management requires patient education on the importance of year-round maintenance therapy, consistency, and compliance. Reinforcing adherence to photoprotection, supporting barrier repair, and setting expectations regarding prognosis are essential components to optimizing melasma treatment outcomes.12
Future Insights: Beyond the Current State of Melasma Research and Treatment
Future advances in melasma management likely will focus on improving disease assessment and long-term treatment durability. Tools such as the MASI and mMASI are widely used but are limited by interobserver variability and reduced sensitivity in darker skin types, highlighting the need to incorporate objective measures such as colorimetry.10,26 There also is a need for studies that relate clinical improvement to underlying pathology. Histopathologic correlation remains limited across most therapeutic trials, and more studies should assess long-term outcomes and quality-of-life measures given the relapsing nature of melasma.26
Future interventions will continue to move past the treatment of pigment alone. Increasing recognition of vascular, inflammatory, and barrier-related contributions to melasma, as well as targeting of other cell types affected by melasma, will be important considerations in future treatment innovations.8,11 In summary, improved inclusion of patients with skin of color in clinical trials, expansion of clinical outcomes measures and objective parameters of disease evaluation, novel approaches to treatment and relapse prevention, and greater attention to patient adherence and education will be essential to achieving advancements in melasma care.12
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
- Sanchez NP, Pathak MA, Sato S, et al. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981;4:698-710.
- Bostan E, Cakir A. The dermoscopic characteristics of melasma in relation to different skin phototypes, distribution patterns and wood lamp findings: a cross-sectional study of 236 melasma lesions. Arch Dermatol Res. 2023;315:1927-1938.
- Guida S, Longo C, Ronga R, et al. Melasma and reflectance confocal microscopy: from baseline to treatment monitoring. Int J Dermatol. 2024;63:1007-1012.
- Ning X, Yang J, Ouyang H, et al. In vivo evaluation of melasma pathologic features and treatment response by 2-photon microscopy. JAMA Dermatol. 2025;161:1039-1047.
- Chen K-J, Han Y, Wang Z-Y, et al. Submicron resolution techniques: multiphoton microscopy in skin disease. Exp Dermatol. 2023;32:1613-1623.
- Philipp-Dormston WG. Melasma: a step-by-step approach towards a multimodal combination therapy. Clin Cosmet Investig Dermatol. 2024;17:1203-1216.
- Doolan B, Gupta M. Melasma. Aust J Gen Pract. 2021;50:880-885.
- Ali L, Al Niaimi F. Pathogenesis of melasma explained. Int J Dermatol. 2025;64:1201-1212.
- Artzi O, Horovitz T, Bar-Ilan E, et al. The pathogenesis of melasma and implications for treatment. J Cosmet Dermatol. 2021;20:3432-3445.
- Gan C, Rodrigues M. An update on new and existing treatments for the management of melasma. Am J Clin Dermatol. 2024;25:717-733.
- Sarkar R, Desai SR, Sinha S, et al. Delphi consensus on melasma management by international experts and pigmentary disorders society. J Eur Acad Dermatol Venereol. Published online September 25, 2025.
- Desai SR, Alexis AF, Elbuluk N, et al. Best practices in the treatment of melasma with a focus on patients with skin of color. J Am Acad Dermatol. 2024;90:269-279.
- Lim HW, Kohli I, Ruvolo E, et al. Impact of visible light on skin health: the role of antioxidants and free radical quenchers in skin protection. J Am Acad Deramtol. 2022;86(3 suppl):S27-S37.
- Goh CL, Chuah SY, Tien S, et al. Double-blind, placebo-controlled trial to evaluate the effectiveness of polypodium leucotomos extract in the treatment of melasma in Asian skin: a pilot study. J Clin Aesthet Dermatol. 2018;11:14-19.
- Mahajan VK, Patil A, Blicharz L, et al. Medical therapies for melasma. J Cosmet Dermatol. 2022;21:3707-3728.
- Suliman RS, Alhuwayshil J, Almuflehi AA, et al. Emerging topical therapies for melasma: a comparative analysis of efficacy and safety. J Dermatolog Treat. 2025;36:2591502.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18:S1545961619P1156X.
- Grimes PE, Dias S, Oparaugo NC, et al. A randomized, controlled, split-face, double-blind study comparing topical malassezin to hydroquinone 4% for melasma. J Drugs Dermatol. 2026;25:25-29.
- Passeron T, Kerob D, Le Dantec G, et al. Efficacy and tolerability of a new facial 2-mercaptonicotinoyl glycine-containing depigmenting serum versus hydroquinone 4% over 3-month treatment of facial melasma. Dermatol Ther (Heidelb). 2025;15:2379-2390.
- McKesey J, Tovar-Garza A, Pandya AG. Melasma treatment: an evidence-based review. Am J Clin Dermatol. 2020;21:173-225.
- Bala HR, Lee S, Wong C, et al. Oral tranexamic acid for the treatment of melasma: a review. Dermatol Surg. 2018;44:814-825.
- Sarkar R, Katoch S. Chemical peels in treatment of melasma. Dermatol Clin. 2024;42:21-32.
- Sarkar R, Gupta M. Platelet-rich plasma in melasma—a systematic review. Dermatol Surg. 2022;48:131-134.
- Pongklaokam J, Manuskiatti W, Wanitphakdeedecha R, et al. Efficacy and safety of letibotulinum toxin A for the treatment of melasma in two different dilutions: a randomized double-blind split-face study. Toxins (Basel). 2025;17:349.
- Han HJ, Kim JC, Park YJ, et al. Targeting the dermis for melasma maintenance treatment. Sci Rep. 2024;14:949.
- Zhang AD, Lazar M, Akhundova E, et al. A scoping review on melasma treatments and their histopathologic correlates. Dermatopathology (Basel). 2025;12:13.
- Goldman MP, Wu DC, Khetarpal S, et al. Dual 1550-nm erbium glass fiber and 1927-nm thulium fiber non-ablative fractional laser system treatment in patients with skin of color: a review of clinical studies and unmet needs. Lasers Surg Med. Published online February 10, 2026.
- Chen J, Yu N, Peng L, et al. Efficacy of low-fluence 1064 nm Q-switched Nd: YAG laser for the treatment of melasma: a meta-analysis and systematic review. J Cosmet Dermatol. 2022;21.
- Feng J, Huang L. Comparison of picosecond and nanosecond Nd:YAG 1064-nm lasers in the treatment of melasma: a split-face randomized clinical trial. Plast Reconstr Surg. 2023;151:772-777.
- Passeron T, Genedy R, Salah L, et al., Laser treatment of hyperpigmented lesions: position statement of the European Society of Laser in Dermatology. J Eur Acad Dermatol Venereol. 2019;33:987-1005.
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Beyond Conventional Paradigms: Rethinking the Evaluation and Management of Melasma
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1
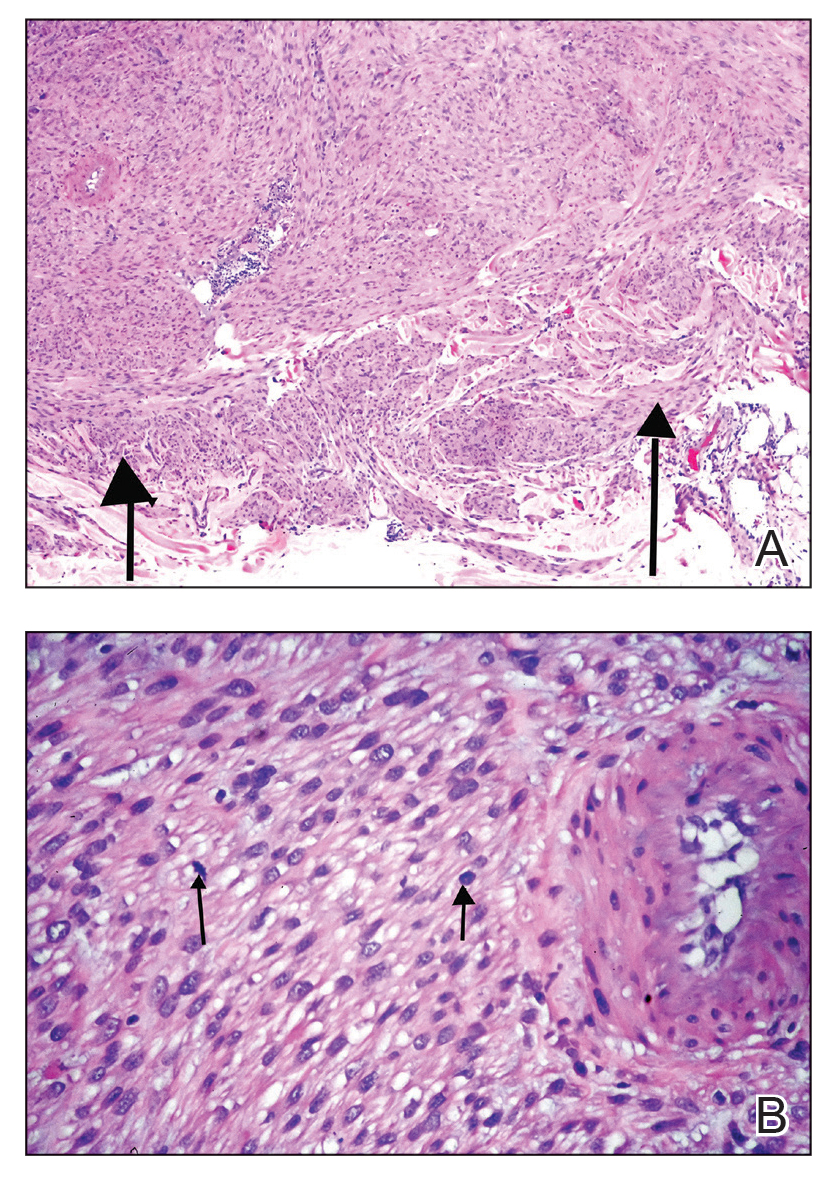
Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
THE DIAGNOSIS: Cutaneous Leiomyosarcoma
Based on the clinical and histopathologic findings, our patient was diagnosed with primary cutaneous leiomyosarcoma (LMS), a rare soft-tissue neoplasm that arises from smooth muscle and typically manifests as a firm pink nodule.1 The neoplasm may occur in the area of a prior traumatic injury or develop spontaneously without an identifiable cause.1-3 Cutaneous LMS represents 2% to 3% of all soft-tissue sarcomas worldwide, with an estimated incidence of 1 in 500,000 annually.1,4 Men who are in their fifth to seventh decades of life are at the highest risk for LMS.1
Histologically, cutaneous LMS can be subclassified as dermal, which has a low metastatic risk and excellent prognosis, or subcutaneous, which is associated with poorer outcomes and vascular muscle origin.1 In our case, hematoxylin and eosin staining revealed fascicles of smooth muscle fibers with hypercellularity, atypia, and mitotic figures (Figure). The neoplasm stained positive for desmin, vimentin, and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.1

Standard treatment for LMS is surgical excision.5 Poor prognostic factors include lesions with a diameter of 5 cm or larger, deep subcutaneous tumor invasion, and distant metastases.2,5
The differential diagnosis may include dermatofibrosarcoma protuberans, which can have a similar pink nodular appearance and also may manifest after injury6; however, this lesion would stain positive for CD34 on histopathology.1 Nodular melanoma also can manifest as a solitary red, raised lesion, but it would stain positive for SOX10, PRAME, and Melan-A on histopathology.7 Basal cell carcinoma, which also may have a similar clinical appearance, is associated with nests of basaloid cells and palisading nuclei histologically.8 Lastly, atypical fibroxanthoma also manifests as a red nodule or plaque and is associated with atypical mitotic figures on histology; however, it notably stains negative for desmin.9
In summary, cutaneous LMS should be included in the differential diagnosis for raised, pink nodules. Given its nonspecific clinical presentation, this rare and malignant neoplasm requires biopsy and immunohistochemical staining for accurate diagnosis.
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
- Soares Queirós C, Filipe P, Soares de Almeida L. Cutaneous leiomyosarcoma: a 20-year retrospective study and review of the literature. Anais Brasileiros de Dermatologia. 2021;96:278-283. doi:10.1016/j.abd.2020.10.003
- Kim NG, Kim JO, Park YJ, et al. Cutaneous leiomyosarcoma of the face. Arch Craniofac Surg. 2017;18:145-148. doi:10.7181/acfs.2017.18.2.145
- Litaiem N, Tabka M, Nefiss M, et al. Cutaneous leiomyosarcoma mimicking arteriovenous malformation. Clin Case Rep. 2020;8:2538-2540. doi:10.1002/ccr3.3222
- Hmida L, Letaief F, Doghri R, et al. Cutaneous leiomyosarcoma on the trunk: an unusual presentation with an aggressive course - case report and review of literature. Pan Afr Med J. 2018;31:190. doi:10.11604/pamj.2018.31.190.16682
- Cazzato G, Sergi MC, Sablone S, et al. Advanced cutaneous leiomyosarcoma of the forearm. Dermatopathology (Basel). 2021;8:40-44. doi:10.3390/dermatopathology8010008
- Li Y, Wang C, Xiang B, et al. Clinical features, pathological findings and treatment of recurrent dermatofibrosarcoma protuberans. J Cancer. 2017;8:1319-1323. doi:10.7150/jca.17988
- Hernandez LE, Frech FS, Mohsin N, et al. Nodular melanoma: a review of pathogenesis, presentation, diagnosis and treatment. J Dermatol & Skin Sci. 2021;3:25-30. doi:10.29245/2767-5092/2021/3.1144
- Steele RB, Puckette Y. Basal cell carcinoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated November 7, 2025. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK482439/
- Kolb L, Schmieder GJ. Atypical fibroxanthoma. StatPearls [Internet]. StatPearls Publishing; 2026. Updated September 18, 2022. Accessed March 3, 2026. http://www.ncbi.nlm.nih.gov/books/NBK459342/
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
Painful, Purpuric, Nodular Lesion With an Irregular Surface on the Shoulder
A 53-year-old man presented to the dermatology clinic for evaluation of a painful, purpuric, nodular lesion on the left shoulder of 3 months’ duration. The lesion had an irregular surface that was surrounded by an erythematous ring. Biopsy revealed fascicles of eosinophilic cells within the dermis. The nuclei were heterogeneous in size and shape and had blunted ends. Frequent atypia and mitotic figures were observed, and the lesion extended into the subcutis. Immunostaining was positive for desmin and smooth muscle actin and negative for SOX10, Melan-A, PRAME (preferentially expressed antigen in melanoma), CD34, and Factor XIIIa.
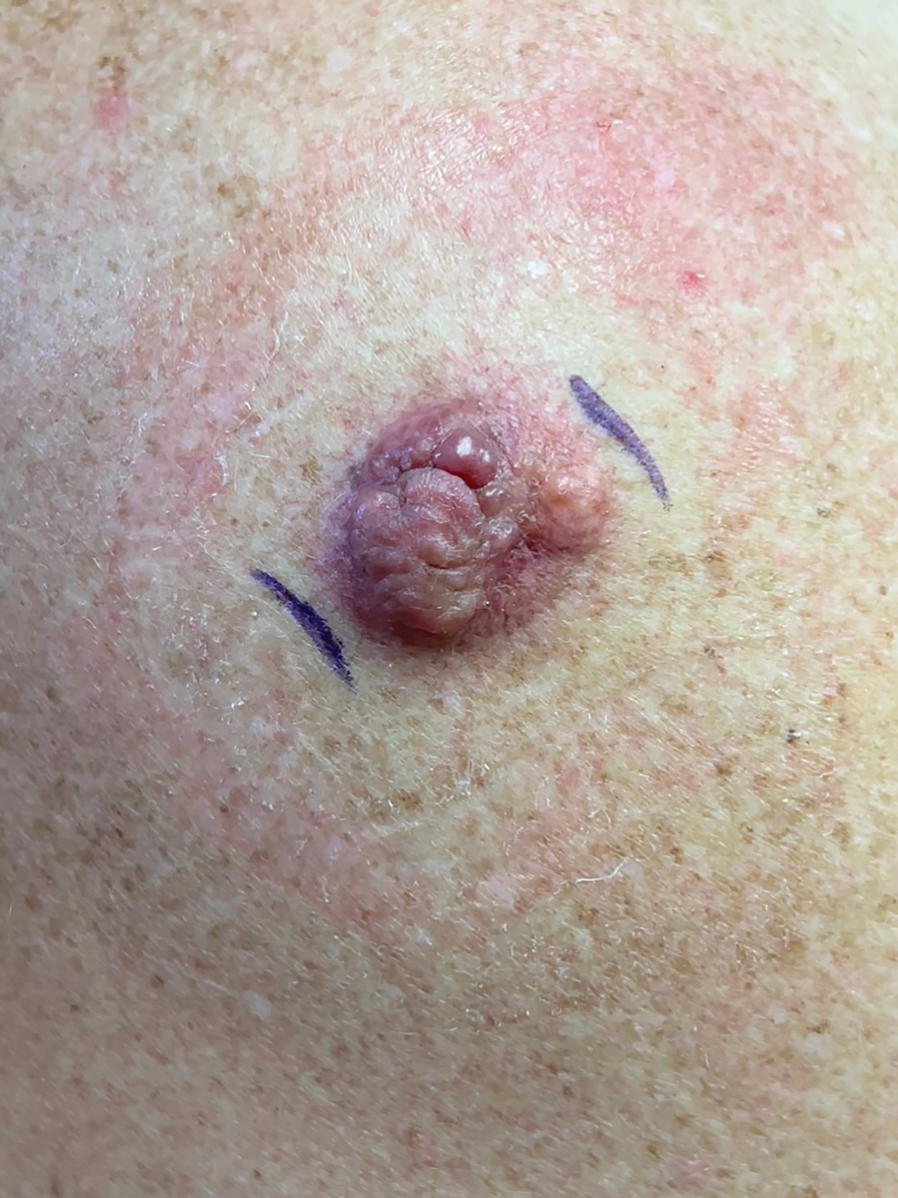
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.
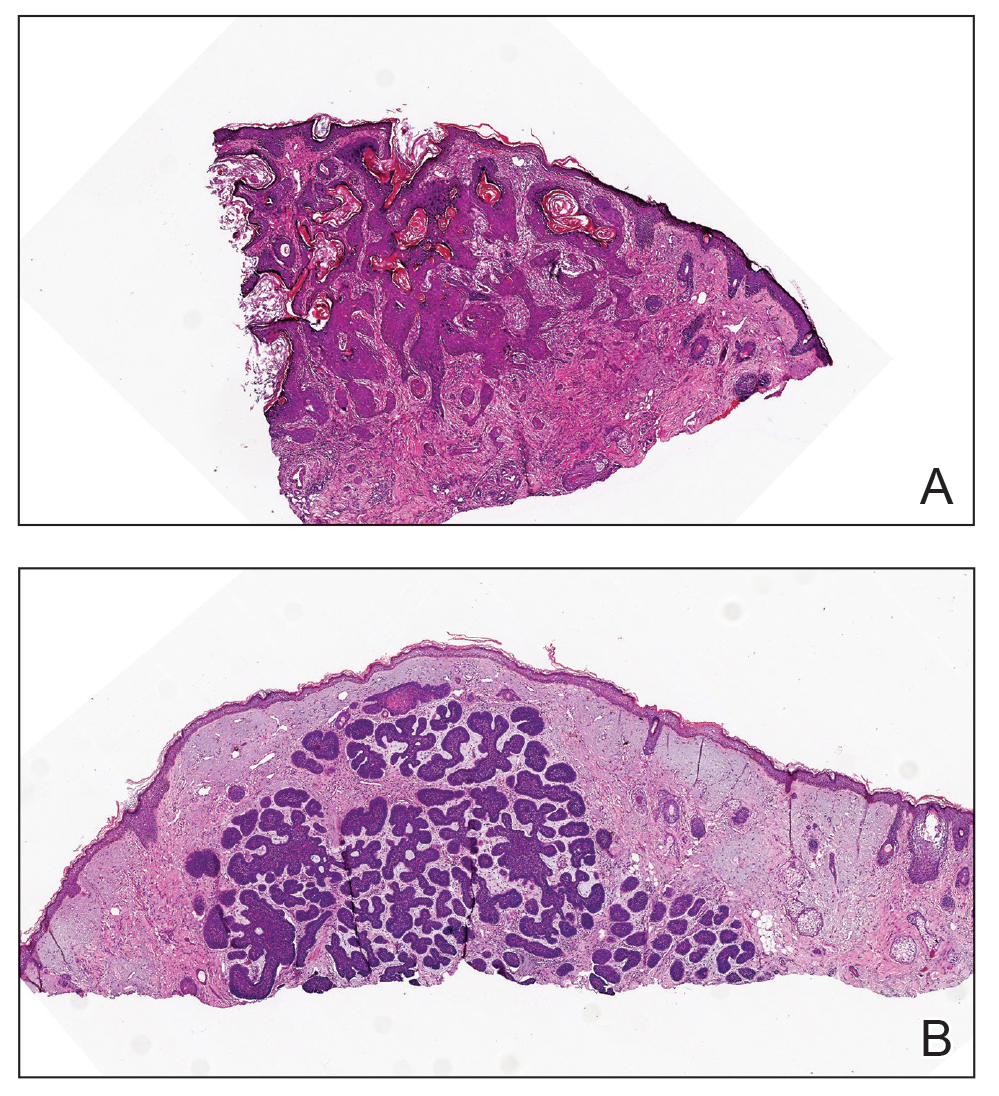
Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
THE DIAGNOSIS: Coexisting Squamous Cell Carcinoma and Basal Cell Carcinoma
Dermoscopy of the plaque showed central ulceration with blood spots surrounded by branched linear vessels, which was suggestive of squamous cell carcinoma (SCC)(Figure 1A). The nodule showed shiny, white-red, structureless areas with small gray spots, bright white crystalline streaks, and short fine telangiectasias suggestive of basal cell carcinoma (BCC)(Figure 1B). Histopathology showed that the plaque had irregular nests, cords, and sheets of neoplastic keratinocytes invading the dermis (Figure 2A) and the nodule had discrete nests of basaloid cells with peripheral palisading in the dermis (Figure 2B), which confirmed the diagnosis of coexisting SCC and BCC. The patient underwent surgical excision of the lesions, which achieved clear margins. At the 2-year follow-up, there was no sign of recurrence.


Squamous cell carcinoma is the second most frequent cancer in humans. Older patients are more susceptible due to chronic UV exposure.1 Basal cell carcinoma is the most common human cancer worldwide.2 These skin cancers have different clinical manifestations, pathologic features, treatment methods, and prognoses. The coexistence of 2 types of skin cancer presents a diagnostic challenge. Possible causes of this phenomenon are not clear. It may simply be a coincidence since the lesions typically occur in sun-exposed areas such as the nose, which may be affected by photodamage.3 According to the field cancerization theory, chronically sun-exposed areas are at higher risk for development of coexisting skin cancers.4 A more interesting explanation is the interaction theory, which suggests that one tumor produces epidermal or stromal changes that induce the formation of a second independent tumor via the paracrine effect (ie, growth mediators from nearby cells).4
Dermoscopy is an important noninvasive diagnostic tool for the evaluation of skin cancer, particularly early detection. Dermoscopic findings of blood vessels, ulcers, the fiber sign, blood spots, white structureless areas, keratin, and centered vessels indicate a diagnosis of SCC.5 In contrast, common dermoscopic findings for BCC include arborizing vessels, ulceration, shiny white structures, and blue-gray ovoid nests or globules.6
Irritated seborrheic keratosis is an inflammatory variant of seborrheic keratosis, which often is challenging to identify clinically due to its similar features with SCC; however, SCC is more likely to demonstrate dotted or branched vessels, white structureless areas, white circles around follicles, irregular or peripheral vessel patterns, and central scales on dermoscopy. In contrast, irritated seborrheic keratosis is more likely to have hairpin vessels, regular vessel patterns, and white halos around vessels, which may aid in the differentiation between the two entities.6
Due to the higher sensitivity of dermoscopy for detecting pigmented BCC compared to nonpigmented BCC, it holds substantial diagnostic value in Asian populations, in whom pigmented BCC is the most common subtype.6,8 However, the lack of pigmentation in the nodule in our case posed a diagnostic challenge, as the diagnosis of BCC had to rely on subtle vascular and shiny white structures rather than more obvious pigment clues. This absence of pigment, however, also helped rule out pigmented BCC as a diagnosis for the nodule. Short fine telangiectasias is the second most common vascular pattern in BCC, and bright white structures are highly suggestive of nonpigmented BCC.6 Therefore, dermoscopic findings of bright-white structures with fine telangiectasias should be alerted to the possibility of nonpigmented BCC.
Basosquamous carcinoma has clinical and dermoscopic features between SCC and BCC, and the presence of dermatoscopic features from both BCC and SCC should raise suspicion, but the diagnosis is particularly challenging because its presentation is nonspecific.9 We need to be vigilant about the possibility of coexistence of 2 types of skin cancer, and that regular physical examination and dermatoscopy are very important for early detection and diagnosis.
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
- Corchado-Cobos R, García-Sancha N, González-Sarmiento R, et al. Cutaneous squamous cell carcinoma: from biology to therapy. Int J Mol Sci. 2020;21:2956. doi:10.3390/ijms21082956
- Cameron MC, Lee E, Hibler BP, et al. Basal cell carcinoma: epidemiology; pathophysiology; clinical and histological subtypes; and disease associations. J Am Acad Dermatol. 2019;80:303-317. doi:10.1016/j.jaad.2018.03.060
- Kraemer KH, Lee MM, Scotto J. Xeroderma pigmentosum. Cutaneous, ocular, and neurologic abnormalities in 830 published cases. Arch Dermatol. 1987;123:241-250. doi:10.1001/archderm.123.2.241
- Cornejo KM, Deng AC. Malignant melanoma within squamous cell carcinoma and basal cell carcinoma: is it a combined or collision tumor? a case report and review of the literature. Am J Dermatopathol. 2013;35:226-34. doi:10.1097/DAD.0b013e3182545e27
- Ertop Dog˘an P, Akay BN, Okçu Heper A, et al. Dermatoscopic findings and dermatopathological correlates in clinical variants of actinic keratosis, Bowen’s disease, keratoacanthoma, and squamous cell carcinoma. Dermatol Ther. 2021;34:E14877. doi:10.1111/dth.14877.
- Álvarez-Salafranca M, Ara M, Zaballos P. Dermoscopy in basal cell carcinoma: an updated review. Actas Dermosifiliogr (Engl Ed). 2021;112:330-338. doi:10.1016/j.ad.2020.11.011
- Papageorgiou C, Spyridis I, Manoli SM, et al. Accuracy of dermoscopic criteria for the differential diagnosis between irritated seborrheic keratosis and squamous cell carcinoma. J Am Acad Dermatol. 2021;85:1143-1150. doi:10.1016/j.jaad.2020.02.019
- Cheng SY, Luk NM, Chong LY. Special features of non-melanoma skin cancer in Hong Kong Chinese patients: 10-year retrospective study. Hong Kong Med J. 2001;7:22-28.
- Murgia G, Denaro N, Boggio F, et al. Basosquamous carcinoma: comprehensive clinical and histopathological aspects, novel imaging tools, and therapeutic approaches. Cells. 2023;23:2737. doi:10.3390/cells12232737
Asymptomatic Plaque and Nodule on the Nose
Asymptomatic Plaque and Nodule on the Nose
An 80-year-old Asian woman presented to the hospital dermatology clinic for evaluation of 2 lesions on the nose of 2 years’ duration. The patient reported that the lesions had initially manifested as an asymptomatic red rash, but within the past month, the affected area had grown in diameter with a rough surface and occasional bleeding. Additionally, a smaller new rash appeared around the original plaque. She had no personal or family history of skin cancer. Physical examination revealed a 1.5-cm reddish plaque on the tip of the nose with a central ulcer filled with viscous exudate. Another 0.5-cm reddish nodule with a smooth surface also was noted adjacent to the plaque. Dermoscopy and a skin biopsy of both lesions were performed.
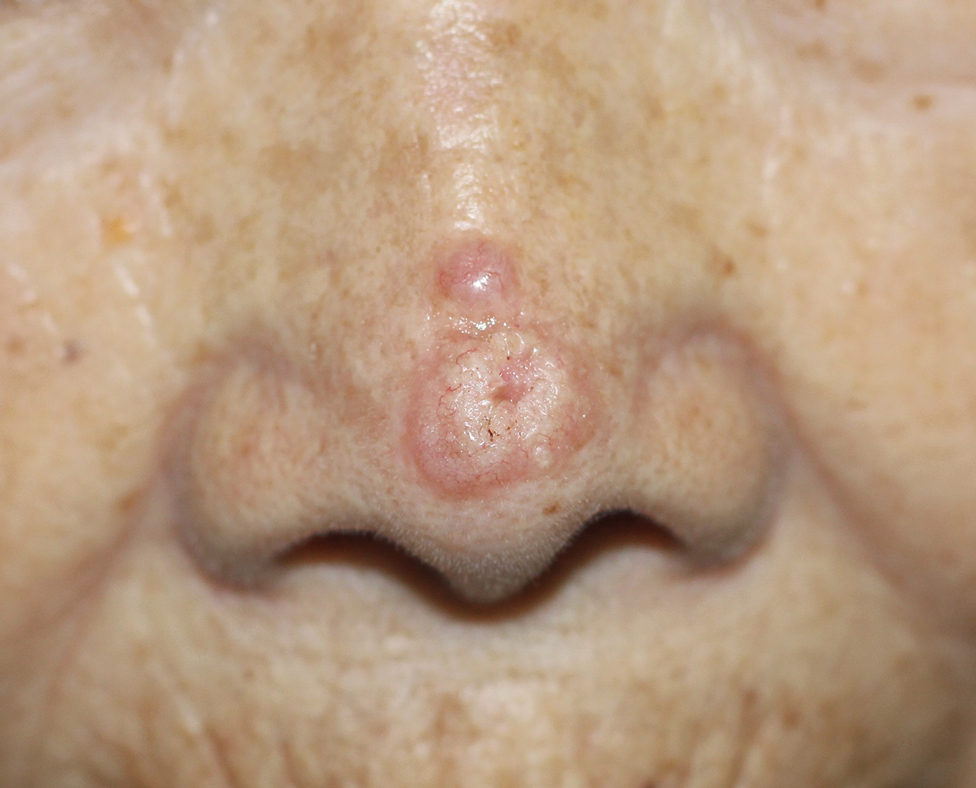
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
THE DIAGNOSIS: Pseudopsoriatic Nails With Pterygium Inversum Unguis
Based on the clinical findings and the patient’s history of gel manicures, a diagnosis of pseudopsoriatic nails with pterygium inversum unguis (PIU) was made. The patient was advised to avoid gel manicures and any other chemical or mechanical trauma to the nails. No other treatment was administered. Improvements including healthy nail growth and disappearing color and structure changes within the nail plates were noted at 2 months’ follow-up.
The durability and availability of gel manicures has been increasingly popular due to their ideal cosmetic results. A gel manicure involves applying a gel nail polish (GNP) containing acrylate or methacrylate monomers that harden after exposure to UV light through a photopolymerization reaction. Acrylate polymers including ethylene glycol dimethacrylate, 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, 2-hydroxypropyl methacrylate, methyl methacrylate, and tetrahydrofurfuryl methacrylate are known to cause allergic contact dermatitis in patients who wear acrylate-based GNP.1Hydroxyethyl methacrylate is the most common sensitizer among these acrylates. Fingertip dry dermatitis, fissured painful pulpitis of the fingers, and periungual erythema are the most common manifestations of methacrylate allergy; however, there also are reports of onycholysis and onychodystrophy in patients with severe allergic contact dermatitis caused by acrylates.2,3
In contrast to common public misconception that GNP may strengthen the nails, scientific evidence has shown otherwise. Besides allergic contact dermatitis, mechanical damage and UV-induced skin manifestations have been reported in association with GNP.1,3,4 Pseudopsoriatic nails are characterized by onycholysis accompanied by subungual hyperkeratosis, closely resembling the nail findings seen in psoriasis. This condition may occur due to mechanical damage and acrylate sensitization.2,4 Pterygium inversum unguis, also known as ventral pterygium, occurs as a result of hyponychium trauma due to either application or removal processes of GNP and/ or exposure to chemical ingredients and is one of the most striking clinical manifestations of GNP use.5 In our patient, all fingernails were affected by PIU.
Patients presenting with pseudopsoriatic nail changes and/or PIU should be questioned about potential exposure to GNP and/or sculpted nails, also known as custom artificial nails or nail prostheses. Diagnosis primarily is made clinically, but microbial cultures or skin biopsy may be required to exclude psoriasis and fungal infections in some patients. Patch testing with acrylate series in particular also is necessary in patients presenting with hand dermatitis. As it is the most common contact sensitizer in the acrylic material of the GNPs, screening for 2-hydroxyethyl methacrylate allergy is recommended in these patients.1 Almost all adverse effects related to use of GNP may be reversible upon discontinuation of exposure.
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
- Litaiem N, Baklouti M, Zeglaoui F. Side effects of gel nail polish: a systematic review. Clin Dermatol. 2022;40:706-715. doi:10.1016 /j.clindermatol.2022.07.008
- Engelina S, Shim TN. Atypical cases of pseudo-psoriatic nails associated with acrylate contact allergy. Contact Dermatitis. 2021; 84:342-344. doi:10.1111/cod.13741
- Draelos ZD. Nail cosmetics and adornment. Dermatol Clinics. 2021;39:351-359. doi:10.1016/j.det.2021.01.001
- Rieder EA, Tosti A. Cosmetically induced disorders of the nail with update on contemporary nail manicures. J Clin Aesthet Dermatol. 2016;9:39-44.
- Cervantes J, Sanchez M, Eber AE, et al. Pterygium inversum unguis secondary to gel polish. J Eur Acad Dermatol Venereol. 2018;32:160-163. doi:10.1111/jdv.14603
Pseudoleukonychia of the Distal Fingernails
Pseudoleukonychia of the Distal Fingernails
An otherwise healthy 36-year-old woman presented to the dermatology department for evaluation of disfiguring nail changes and subungual verrucous skin lesions of 3 weeks’ duration. A review of systems and the patient’s personal and family history were unremarkable. She denied any recent trauma or chemical exposure but noted that she had regularly been patronizing a beauty salon for gel manicures over the past year; her most recent visit was 6 weeks prior to the current presentation. She previously was treated at another dermatology clinic with local corticosteroid creams without any improvement. Dermatologic examination revealed pseudoleukonychia of the distal fingernails surrounded by an erythematous and/or haemorrhagic border. Overgrowth and adherence of the hyponychium to the nail plate also was noted in almost all the fingernails. A prior complete blood cell count and biochemistry panel were within reference range.
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).
Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
Have you ever heard of the American Dermatological Association (ADA)? While many residents may not yet be familiar with this group, its members are among the most respected leaders in dermatology. They serve as current and past presidents of influential organizations including the American Academy of Dermatology (Susan C. Taylor, MD [Philadelphia, Pennsylvania]), the American Society for Dermatologic Surgery (M. Laurin Council, MD, MBA [Creve Coeur, Missouri]), and the Association of Professors of Dermatology (Sewon Kang, MD [Baltimore, Maryland]). Others lead certification boards or serve as editors of key journals like the Journal of the American Academy of Dermatology (Dirk Elston, MD [Charleston, South Carolina]), JAMA Dermatology (Kanade Shinkai, MD [San Francisco, California], and Cutis (Vincent A. DeLeo, MD [Los Angeles, California]).
The ADA is celebrating its 150th anniversary in 2026. What makes the organization so enduring is not just its history, but its culture. The members of the ADA foster deep, long-lasting relationships, and its meetings are purposefully designed to balance structured scientific sessions with unscheduled time for reflection, conversation, and connection. That intentional design cultivates learning, innovation, and wellness.
Steven Covey’s The 7 Habits of Highly Effective People1 highlights the importance of renewal and relationship building, as does the Harvard Study of Adult Development, one of the longest-running research projects on well-being.2-4 The key conclusion? Relationships are the strongest predictors of long, healthy, and fulfilling lives, not wealth or achievement. Medical training is intense, and the emphasis often falls squarely on achievement. But the friendships you form in medical school, residency, and early career are just as formative. Membership with the ADA continues this spirit of connection throughout one’s professional life, with meetings that welcome spouses and partners and encourage engagement across generations.
A hallmark of ADA culture is its commitment to mentoring and mutual support. Need advice about transitioning from private practice to academia? Navigating department leadership? Applying for a grant? Considering industry, editorial, or global health roles? Within the ADA, there’s someone who has done it and is eager to help. Recent meetings have addressed future-facing topics such as artificial intelligence, bedside diagnostics, workforce advocacy, and global health while also carving out time for rejuvenating activities: book clubs with best-selling authors, sessions on the arts, storytelling, wellness, and travel. This holistic programming reflects the ADA’s belief in supporting the whole physician.3 Members understand the value of relationships and appreciate these opportunities to learn about the passions and interests of their colleagues (Table).

Candidates are nominated by current members and must be board certified and at least 10 years beyond completion of their training. Members vote upon candidates in a rank voting system each year. If someone is nominated and not selected, they did not fail—they may be nominated again. The idea behind this membership process is to keep the organization small enough that members can get to know one another—there are currently 552 active members. Importantly, the ADA has embraced diversity and inclusion. While historically male- and White-dominated, recent inductee classes now reflect gender parity and a broader range of backgrounds, enriching the organization with fresh perspectives.5-8
For residents and fellows, the lesson is clear: friendships, mentorship, and time for reflection are not luxuries—they are essential. Burnout stems from relentless output in isolation; however, in cultures that prioritize renewal, authenticity, and community, physicians can flourish.9 Membership in small professional organizations is an important step towards avoiding isolation. We encourage you to be active in your local, state, and national organizations.
The ADA stands as a powerful example of how professional societies can help you build the kind of life and career you want, not just a résumé. From informal beachside conversations to high-level scientific discussions, its enduring strength is this: leaders helping others lead.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
- Covey SR. The 7 Habits of Highly Effective People: Powerful Lessons in Personal Change. Simon & Schuster; 1989.
- Waldinger R, Schulz M. The Good Life: Lessons From the World’s Longest Scientific Study of Happiness. Simon & Schuster; 2023.
- Malone JC, Liu SR, Vaillant GE, et al. Midlife Eriksonian psychosocial development: setting the stage for late-life cognitive and emotional health. Dev Psychol. 2016;52:496-508. doi:10.1037/a0039875
- Vaillant GE, Milofsky E. Natural history of male psychological health: IX. Empirical evidence for Erikson’s model of the life cycle. Am J Psychiatry. 1980;137:1348-59. doi:10.1176/ajp.137.11.1348
- American Dermatological Association. Accessed August 8, 2025. https://www.ada1.org
- Pariser DM. Illustrated History of the American Dermatological Association, 1876–2020. American Dermatological Association; 2020.
- Smith JG Jr, Johnson ML. 125th anniversary of the American Dermatological Association. Arch Dermatol. 2001;137:1520. doi:10.1001/archderm.137.11.1520
- Rodriguez R, Anderson L, Woolhiser E, et al. Diversity among American Dermatological Association members by sex and geographic region. JMIR Dermatol. 2024;7:E47802. doi:10.2196/47802
- Grant A. Think Again: The Power of Knowing What You Don’t Know. Viking; 2021.
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Leaders Helping Leaders: The American Dermatological Association as a Model of Lifelong Professional Community
Practice Points
- Professional relationships are a critical determinant of career longevity and personal well-being. Structured opportunities for mentorship, reflection, and intergenerational dialogue within professional societies can help mitigate burnout and foster sustained leadership development.
- Medical societies serve as leadership incubators. Through selective membership, cross-organizational representation, and sustained engagement, the American Dermatological Association models how professional community strengthens the specialty beyond scientific exchange alone.
- Physicians benefit from intentional engagement in organized dermatology. Participation in local, state, and national societies cultivates mentorship networks, broadens perspective, and lays the foundation for future leadership opportunities.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6
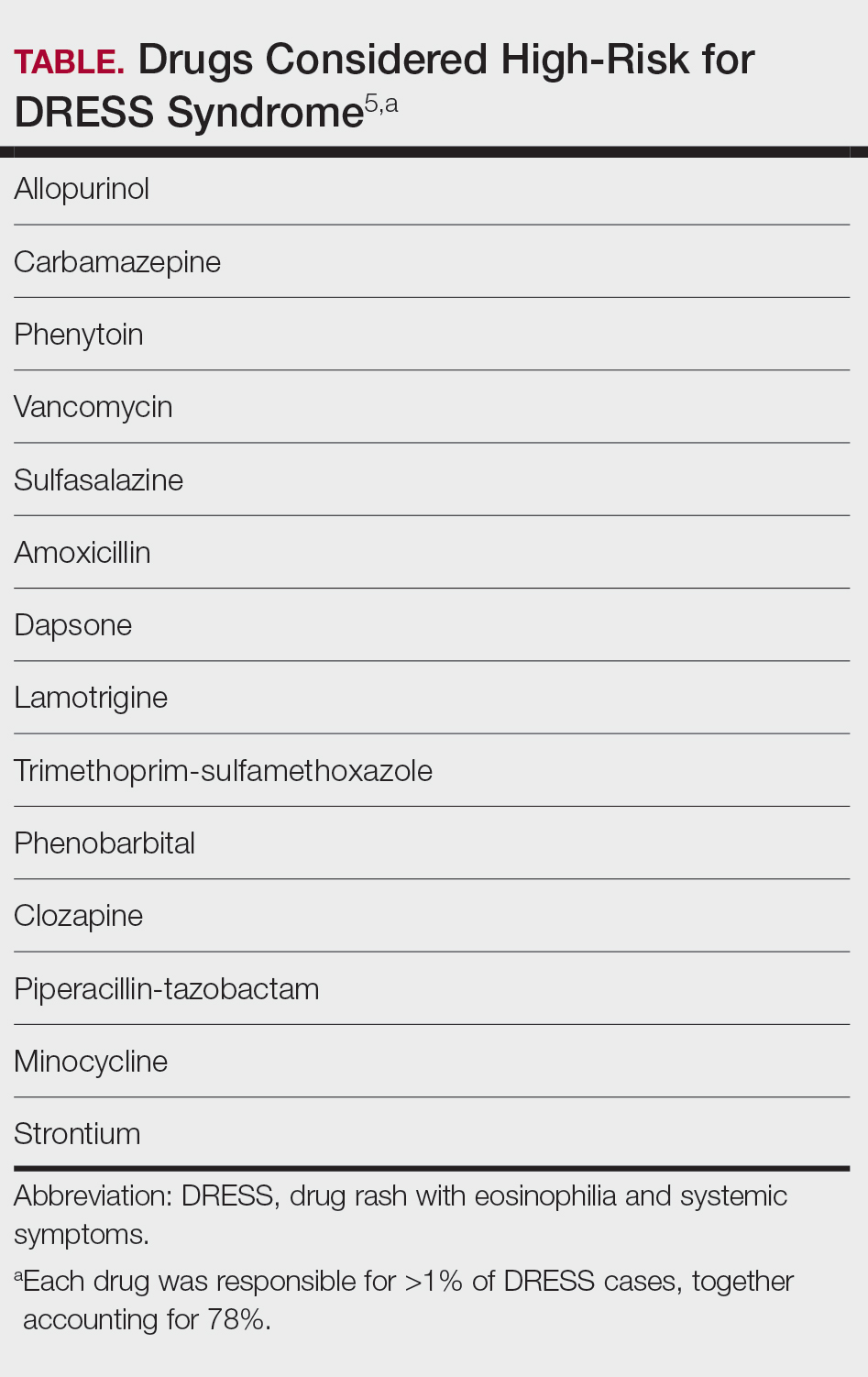
A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
Dermatologists play a central role in the care of hospitalized patients with skin disease. This review summarizes research from January 2024 to December 2025 on severe cutaneous adverse drug reactions, emerging infectious diseases, hidradenitis suppurativa (HS), and inpatient dermatology workforce issues. Key developments include improved recognition and management of drug reactions; updated diagnostic and prognostic tools for Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN); and guidance for emerging infections such as measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Evidence-based strategies for HS aim to reduce unnecessary admissions and optimize care. Workforce challenges, including limited access, high call burden, and potential for artificial intelligence (AI)–assisted diagnosis, are also highlighted. These findings emphasize the critical contributions of dermatologists to hospital-based care and provide emerging evidence to guide clinical practice.
Dermatologists play a critical role in the care of hospitalized patients. Herein, we review the research developments between January 2024 and December 2025 most relevant to the care of hospitalized patients with skin disease, including severe cutaneous adverse reactions (SCARs), emerging and re-emerging infectious diseases, hidradenitis suppurativa (HS), and access to inpatient dermatology services.
Severe Cutaneous Adverse Drug Reactions
Severe cutaneous adverse drug reactions are among the most frequent reasons for inpatient dermatology consultation. A National Inpatient Sample study identified more than 160,000 cases of drug rash with eosinophilia and systemic symptoms (DRESS syndrome) between January 2016 and December 2020.1 The overall mortality rate was 2.0%, substantially lower than the rates of up to 10% reported in earlier studies.2 Case burden and mortality peaked during the fall months, possibly due to either increased use of antibiotics or increased viral infection or reactivation during these months.1
A retrospective cohort study of patients with probable or definite DRESS syndrome showed that, among 93 patients with at least 1 viral marker tested, human herpesvirus (HHV) reactivation was found in 42% (39/93), including HHV-6 (28%)(24/85), Epstein-Barr virus (17%)(15/87), and cytomegalovirus (20%)(18/89); furthermore, viral reactivation was associated with higher 1-year mortality (odds ratio, 3.9), dialysis initiation, flares of disease, and longer hospital stay (all P<.05).1 Multiple reactivations were associated with higher inpatient mortality and 1-year mortality; however, despite apparent prognostic importance, the role of screening for viral reactivation in DRESS syndrome is undefined.
A systematic review of 610 studies including 2122 patients with DRESS syndrome demonstrated that, among 193 causal agents identified, 14 drugs accounted for more than 1% of cases each and therefore were considered high risk. Seventy-eight percent of cases were attributed to these 14 drugs (Table).5 A TriNetX Query study analyzed antibiotic exposures across SCARs and reported that sulfonamides (hazard ratio [HR], 7.5), aminoglycosides (HR, 3.7), and tetracyclines (HR, 1.7) were associated with an elevated risk for SCARs. Sulfonamides had the highest absolute incidence of SCARs, followed by cephalosporins and penicillins.6

A multicenter randomized clinical trial7 compared high-potency topical corticosteroids (clobetasol 30 g/d) to systemic corticosteroids (prednisone 0.5 mg/kg/d) for treatment of moderate DRESS syndrome. On day 30, 53.8% (14/26) of patients in the topical group had achieved remission of visceral involvement, compared to 72.0% (18/25) in the systemic group. Before day 30, 23.1% (6/26) of patients in the topical group worsened, necessitating transition to high-dose systemic steroids. When inpatient monitoring is available, low-dose systemic corticosteroids or high-potency topical steroids may be reasonable management strategies for moderate DRESS syndrome7; however, the frequent need for treatment intensification suggests limitations to this strategy.
Since prolonged courses of systemic steroids generally are necessary for management of DRESS syndrome, steroid-sparing options are needed. A retrospective case series examined interleukin 5 inhibition in patients with possible DRESS syndrome (Registry of Severe Cutaneous Adverse Reactions score ≥3). All patients demonstrated rapid eosinophil reduction within 1 to 3 days (mean [SD] time to resolution, 1.4 [0.9] days) after treatment with mepolizumab or benralizumab, with clinical improvement occurring at a mean (SD) of 16 (3.7) days (range, 13-21 days).8
A French cohort study of 1221 adult patients with Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) reported in-hospital mortality of 19% and a total mortality of 34% at 1 year.9 Risk factors contributing to in-hospital mortality included age, history of/current diagnosis of cancer, dementia, and liver disease, while postdischarge mortality was associated with acute kidney injury and sepsis. Long-term complications included ophthalmologic and mood disorders.9
A new set of diagnostic criteria for SJS/TEN, known as the Niigata criteria,10 includes 3 main items: severe mucosal lesions in cutaneous-mucosal transition zones (eg, eyes, lips, vulva) or generalized erythema with necrotic lesions; fever of 38.5 °C or higher; and necrosis of the epidermis seen on histopathology. Because epidermal detachment involving 10% of the body surface area (BSA) is an important mortality risk predicter, SJS is defined as less than 10% BSA involvement, and TEN has been redefined as 10% or more BSA involvement (not ≥30%). A new prognostic score—clinical risk score for TEN (CRISTEN)—can be tabulated at the point of care without laboratory values. It was developed based on the 10 most important risk factors for death in a retrospective study of 382 patients, which included age 65 years or older; epidermal detachment involving 10% BSA or higher; an antibiotic as causative agent; systemic corticosteroid therapy before the onset of SJS/TEN; involvement of all 3 mucosal surfaces; and medical comorbidities such as renal impairment, diabetes, cardiac disease, active cancer, and bacterial infection.11
New potential therapeutic targets for SJS/TEN include PC111 (monoclonal antibody to Fas ligand), formyl peptide receptor 1 antagonists (which inhibit necroptosis induced by formyl peptide receptor 1–annexin A1 interaction), daratumumab (which depletes cytotoxic CD8-positive and CD38-positive T cells), and Janus kinase (JAK) inhibitors.10 Spatial proteomics showed marked enrichment of type I and type II interferon signatures as well as activation of signal transducer and activator of transcription 1. In vitro, tofacitinib reduced keratinocyte-directed cytotoxicity, and in vivo JAK inhibitors ameliorated disease severity in 2 TEN mouse models. Patients with TEN that was refractory to corticosteroid therapy received rescue treatment with JAK inhibitors and had re-epithelization within several days with marked reduction in levels of phosphorylated signal transducer and activator of transcription 1.12 Controlled studies are needed to assess the potential role of JAK inhibitors for SJS/TEN.
Emerging and Re-emerging Infectious Diseases
Dermatologists may encounter emerging or re-emerging infections, performing an essential public health role in the process. In 2025, a total of 2281 confirmed cases of measles had been reported across 45 of the United States.13 During the COVID-19 pandemic, measles vaccine coverage in the United States dropped to 93%—down from 95% to 97% prepandemic. Worldwide, 2022 saw an increase of 1.4 million measles cases (18% increase) and 41,200 excess deaths (43% increase) compared to the previous year. Complications of measles include pneumonia, blindness, otitis media, and encephalitis, with 1 in 5 (20%) unvaccinated people with measles in the United States requiring hospitalization.14 A vaccine coverage rate higher than 95% is needed to prevent community spread of disease. Since efforts to detect and rapidly isolate cases of measles are critical, dermatologists should consider measles in the differential of morbilliform eruptions with viral symptoms and ask about vaccination status.
Since 2023, dengue infection rates have tripled in the Americas, representing the highest levels recorded since tracking began in 1980. In 2024, there were more than 12 million cases, with approximately 8000 deaths reported. Ninety percent of cases occur in Argentina, Brazil, Colombia, and Mexico, but local transmission has been reported in Arizona, California, Florida, Hawaii, and Texas.15 The characteristic exanthem of dengue is diffuse erythema with islands of sparing.<
Unlike during the 2022 outbreak of mpox clade II, which predominantly impacted men who have sex with men, there now is an ongoing outbreak of mpox clades 1a and 1b in the Democratic Republic of the Congo and surrounding countries that more commonly affects children and heterosexual adults. It is also more transmissible and virulent. Cases of mpox clade I have been reported in several European countries and across the United States, mostly among travelers from areas of active transmission. Vaccination of at-risk individuals is considered effective; however, tecovirimat is not.16
Outbreaks of 2 emerging zoonotic orthopoxviruses recently have been reported. Buffalopox virus (BPXV) is transmitted via direct contact with the skin of infected cattle and buffalo as well as fomites and has been responsible for human cases in South Asia. Characteristics of BPXV include macules, umbilicated papules, vesicles, pustules, and eschars that evolve over several weeks, with a predilection for the hands and face. It can manifest with prodromal symptoms of fever, malaise, and lymphadenopathy.17 Borealpox virus (formerly known as Alaskapox) has similar manifestations. Its reservoir includes small mammals such as voles and shrews, but it also has been found in cats and dogs and has been responsible for at least one human fatality. Cidofovir may be an effective therapy for both BPXV and borealpox virus, and prior smallpox vaccination may provide protection.18 These outbreaks demonstrate the continued importance of research for more effective vaccines and therapies against smallpox and other orthopoxviruses.19 A recent review provided a detailed overview of the epidemiology, transmission, dermatologic findings, and management strategies associated with smallpox and other bioweapons.20
In 2023, a case was reported of a patient in a New York City hospital with tinea that was refractory to multiple rounds of topical antifungals, which called attention to the presence of Trichophyton indotineae in the United States.21 Since then, additional reports and case series have characterized the clinical presentation of T indotineae as widespread and atypical, refractory to traditional therapies, and most often encountered in travelers returning from Bangladesh or elsewhere in South Asia.22 The diagnosis should be confirmed via DNA testing of fungal culture. Itraconazole 100 to 200 mg/d is the antifungal therapy of choice.23
Other series have reported cases of tinea genitalis caused by Trichophyton mentagrophytes type VII seen predominately in sex workers and others engaging in high-risk sexual contact, highlighting the spread of dermatophytes through sexual activity.24-26 Lastly, it is important to culture pustules and consider atypical pathogens in patients with chronic folliculitis not responding to typical therapies such as tetracycline antibiotics. A case series reported the presence of pustules in the beard area of 7 men who have sex with men, with culture data showing Klebsiella aerogenes. Prolonged courses of fluoroquinolones were necessary for clearance.27
Reducing HS Admissions Through Evidence-Based Management
Hidradenitis suppurativa is a frequent cause of emergency department visits and hospital admissions. In an analysis of the Nationwide Readmissions Database, 17.8% (392/2204) of patients admitted to the hospital with HS were readmitted within 30 days, a number comparable to that of heart failure.28
Flaring HS can produce symptoms that mimic sepsis. A retrospective cohort study examining sepsislike features in HS showed that more than 50% (30/58) of those admitted to the hospital with an HS flare were misdiagnosed with sepsis, and more than 80% (53/64) of those patients received intravenous antibiotics.29 A National Inpatient Sample (January 2016-December 2018) study demonstrated minimal rates of true infection in patients admitted with HS flares,30 while patients with HS diagnosed as sepsis do not sustain the mortality expected from true sepsis. Improving recognition of HS and differentiation of the disease from true sepsis could decrease unnecessary antibiotic use, hospital admissions, and cost, underscoring the need for a framework to reliably and reproducibly distinguish sepsis from HS flare.31
While severe HS is difficult to manage, there may be a window of opportunity in which appropriate treatment of early disease may prevent progression and decrease inpatient utilization. A prospective cohort study of 335 biologic-naïve patients with mild to moderate HS (Hurley stages I and II) followed over a median of 2 years showed that active smoking, body mass index higher than 25, and the presence of disease in 2 or more anatomic areas were factors predictive of progression to severe disease.32
Despite high utilization of emergency and inpatient care, there has been no consensus on inpatient management of HS. A Delphi consensus exercise including 26 expert dermatologists reached consensus on 40 statements.33 Specific recommendations involve multidisciplinary care, including from a dermatologist; consideration of comorbid medical conditions; supportive care measures (wound care, pain control); evidence-based medical management, including initiation or adjustment of biologic therapies; targeted surgical intervention; nutritional support and maintenance of glycemic control; and attention to transitional care at discharge, including home health services, verification of insurance status, and timely outpatient dermatology follow-up.34 A retrospective review of 98 patients treated with intravenous ertapenem for a mean duration of 13 weeks demonstrated improvement in clinical and inflammatory markers.35 Patients with severe or treatment-refractory HS, including those admitted to the hospital, may benefit from initiation of this therapy in select circumstances.
Hospital Dermatology Workforce
Inpatient dermatology consultations are extremely valuable for improving diagnostic accuracy, reducing admissions for pseudocellulitis and inflammatory skin conditions, and keeping cancer patients on needed therapies.36-38 Despite this clear value added, a cross-sectional analysis of inpatient Medicare claims data from January 2013 to December 2019 found that the number of dermatologists performing more than 10 inpatient consults per year decreased from 356 to 281.39 Additionally, medical centers in which dermatology encounters occurred decreased from 239 to 157 during the same period. Ninety-eight percent of inpatient dermatologists were in metropolitan areas, with large regions lacking access to inpatient dermatology consultation altogether.39
A survey of Society for Pediatric Dermatology members similarly characterized the state of the pediatric dermatology workforce performing hospital consultation.40 Seventy-five percent reported a high call burden, defined as more than 11 days or nights per month, more than 1 weekend per month, and/or more than 5 hours per week seeing patients. Ninety-one percent of consultation services are based within academic institutions, reflecting disparities in access.40 A prospective cohort study of academic pediatric dermatologists reported that 310 curbside consultations were performed over 24 weeks; of these calls, 17% occurred during weeknights and 23% on weekends. None of these curbside interactions was reimbursed.41 These findings underscore the burden of uncompensated time a subset of pediatric dermatologists dedicates to inpatient consultations, highlighting the need for improved financial and administrative support and an increased number of physicians performing this role.
A survey study42 suggested that unfamiliarity with the inpatient setting, rather than medical knowledge, is the most important barrier to inpatient work among clinical dermatologists. Proposed interventions include resource guides (eg, hospital maps, pager numbers for key individuals, and protocols for urgent specimens). Reference guides and refresher courses may decrease gaps in knowledge or awareness among dermatologists in ambulatory practice.42 Another way to bolster the inpatient dermatology workforce may be to provide more guidance to qualified advanced practice providers to triage and address dermatologic emergencies.43
Artificial intelligence (AI) also has been explored as a tool for diagnosing complex dermatologic conditions. One study presented 15 published inpatient dermatology cases to 7 dermatologists. Participants were asked to formulate their top 3 differential diagnoses and were then shown AI-generated differentials and asked to submit a revised differential. Participants showed a diagnostic accuracy of 69% before seeing the AI-generated differential diagnosis and 79% after; however, in cases in which the AI differential was incorrect, diagnostic accuracy of the dermatologists decreased after being shown the AI model.44
Final Thoughts
This January 2024 to December 2025 review of research relevant to hospital dermatology highlights important developments and ongoing challenges in SCARs, emerging and re-emerging infectious diseases, HS, and the inpatient dermatology workforce. Dermatologists continue to play a critical role in the care of hospitalized patients with skin disease.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
- Desai AD, Thomas C. Seasonal trends in drug reaction with eosinophilia and systemic symptoms. J Am Acad Dermatol. 2025;92:183-185.
- Wei BM, Fox LP, Kaffenberger BH, et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. 2024;90:885-908. doi:10.1016/j.jaad.2023.02.072
- Chan LCE, Sultana R, Choo KJL, et al. Viral reactivation and clinical outcomes in drug reaction with eosinophilia and systemic symptoms (DRESS). Sci Rep. 2024;14:28492.
- Brüggen MC, Walsh S, Ameri MM, et al. Management of adult patients with drug reaction with eosinophilia and systemic symptoms: a Delphi-based international consensus. JAMA Dermatol. 2024;160:37-44
- Hansen E, Gallardo M, Yan A, et al. Risk assessment of drugs associated with DRESS syndrome based on publication frequency: a systematic review. J Am Acad Dermatol. 2024;91:962-966.
- Neubauer ZJK, Chan R, Singal A, et al. SCAR-ed by antibiotics: a retrospective cohort study of severe cutaneous adverse reactions (SCAR) relative risk. J Am Acad Dermatol. 2025;92:1143-1145.
- Ingen-Housz-Oro S, Guichard E, Milpied B, et al. Topical versus oral corticosteroids in moderate drug reaction with eosinophilia and systemic symptoms: a multicenter randomized clinical trial. J Am Acad Dermatol. 2024;91:544-547.
- Hijaz B, Nambudiri VE, Imadojemu S. IL-5 inhibitor treatment in drug reaction with eosinophilia and systemic symptoms. JAMA Dermatol. 2025;161:661-663.
- Bettuzzi T, Lebrun-Vignes B, Ingen-Housz-Oro S, et al. Incidence, in-hospital and long-term mortality, and sequelae of epidermal necrolysis in adults. JAMA Dermatol. 2024;160:1288-1296.
- Hama N, Aoki S, Chen CB, et al. Recent progress in Stevens-Johnson syndrome/toxic epidermal necrolysis: diagnostic criteria, pathogenesis and treatment. Br J Dermatol. 2024;192:9-18.
- Hama N, Sunaga Y, Ochiai H, et al. Development and validation of a novel score to predict mortality in Stevens-Johnson syndrome and toxic epidermal necrolysis: CRISTEN. J Allergy Clin Immunol Pract. 2023;11:3161-3168.e2.
- Nordmann TM, Anderton H, Hasegawa A, et al. Spatial proteomics identifies JAKi as treatment for a lethal skin disease. Nature. 2024;635:1001-1009.
- Centers for Disease Control and Prevention. Measles cases and outbreaks. Updated January 7, 2026. Accessed January 12, 2026. https://www.cdc.gov/measles/data-research/
- Rubin R. Despite safe and effective vaccine, measles cases and deaths increased worldwide from 2021 to 2022. JAMA. 2024;331:188-189.
- Orrall A. Dengue cases in the Americas highest recorded. JAMA. 2025;333:452.
- Harris E. As mpox cases surge in Africa, WHO declares a global emergency-here’s what to know. JAMA. 2024;332:862-864.
- Burningham KM, Hinojosa T, Cavazos A, et al. Buffalopox: an emerging cutaneous disease in humans. J Eur Acad Dermatol Venereol. 2025;39:404-406.
- Parker ER. Emergence of Alaskapox infection: what dermatologists need to know. J Am Acad Dermatol. 2024;91:397-399.
- Gostin LO, Singaravelu S, Hynes N. Smallpox readiness: modern strategies against an ancient disease. JAMA. 2024;332:873-874.
- Osborne S, Kam O, Thacker S, et al. Review of category A bioweapons with cutaneous features: epidemiology, clinical presentation, and contemporary management strategies. J Am Acad Dermatol. 2025;93:165-175.
- Caplan AS, Chaturvedi S, Zhu Y, et al. Notes from the field: first reported U.S. cases of tinea caused by Trichophyton indotineae - New York City, December 2021-March 2023. MMWR Morb Mortal Wkly Rep. 2023;72:536-537.
- McKenna M. Why the rise of this drug-resistant fungus is raising international concern. JAMA. 2024;332:859-861.
- Caplan AS, Todd GC, Zhu Y, et al. Clinical course, antifungal susceptibility, and genomic sequencing of Trichophyton indotineae. JAMA Dermatol. 2024;160:701-709.
- Jabet A, Bérot V, Chiarabini T, et al. Trichophyton mentagrophytes ITS genotype VII infections among men who have sex with men in France: an ongoing phenomenon. J Eur Acad Dermatol Venereol. 2025;39:407-415.
- Luchsinger I, Bosshard PP, Kasper RS, et al. Tinea genitalis: a new entity of sexually transmitted infection? Case series and review of the literature. Sex Transm Infect. 2015;91:493-496.
- Khurana A, Sharath S, Sardana K, et al. Therapeutic updates on the management of tinea corporis or cruris in the era of Trichophyton indotineae: separating evidence from hype-a narrative review. Indian J Dermatol. 2023;68:525-540.
- Bérot V, Monsel G, Dauendorffer JN, et al; Groupe Infectiologie Dermatologique et Infections Sexuellement Transmissibles (GrIDIST) de la Société Française de Dermatologie. Klebsiella aerogenes-related facial folliculitis in men having sex with men: a hypothetical new STI?J Eur Acad Dermatol Venereol. 2025;39:E10-E12.
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the Nationwide Readmissions Database. J Am Acad Dermatol. 2022;87:188-192.
- AbdelHameid D, Wang L, Mauskar MM, et al. Sepsis-like features in hidradenitis suppurativa flares requiring admission: a retrospective cohort study. J Am Acad Dermatol. 2024;90:1291-1294.
- Ehizogie E, Maghari I, Lo S, et al. Hidradenitis suppurativa, systemic inflammatory response syndrome and sepsis: a database study. Br J Dermatol. 2024;191:451-453.
- Maghari I, Abiad H, Griffin T, et al. Hidradenitis suppurativa (HS), systemic inflammatory response syndrome and sepsis, sepsis caused by HS: an empty systematic review. Br J Dermatol. 2024;191:449-450.
- Kjærsgaard Andersen R, Pedersen O, Eidsmo L, et al. Initial steps towards developing a predictive algorithm of disease progression for hidradenitis suppurativa (HS): results from a Cox proportional hazard regression analysis on disease progression among a cohort of 335 Danish patients with HS. Br J Dermatol. 2024;190:904-914.
- Needham M, Pichardo R, Alavi A, et al. Inpatient management of hidradenitis suppurativa: a Delphi consensus study. Cutis. 2024;113:251-254.
- Maskan Bermudez N, Elman SA, Kirsner RS, et al. Management of hidradenitis suppurativa in the inpatient setting: a clinical guide. Arch Dermatol Res. 2025;317:202.
- Nosrati A, Ch’en PY, Torpey ME, et al. Efficacy and durability of intravenous ertapenem therapy for recalcitrant hidradenitis suppurativa. JAMA Dermatol. 2024;160:312-318.
- Tracey EH, Forrestel A, Rosenbach M, et al. Inpatient dermatology consultation in patients with hematologic malignancies. J Am Acad Dermatol. 2016;75:835-836.
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543.
- Jacoby TV, Shah N, Asdourian MS, et al. Dermatology evaluation for cutaneous immune-related adverse events is associated with improved survival in cancer patients treated with checkpoint inhibition. J Am Acad Dermatol. 2023;88:711-714.
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts-a cross-sectional analysis. Arch Dermatol Res. 2024;316:103.
- Pineider JL, Rangu SA, Shaw KS, et al. Pediatric consultative dermatology: a survey of the Society for Pediatric Dermatology workforce reveals shortcomings in existing practice models of pediatric dermatology consult services in the United States. Pediatr Dermatol. 2024;41:270-274.
- Puar NK, Canty KM, Newell BD, et al. An evaluation of pediatric dermatology curbside consultations in an academic center: a prospective cohort study. J Am Acad Dermatol. 2024;90:1258-1260.
- Lau CB, Smith GP. Strategies for improving dermatologist comfort and quality of patient care in inpatient settings: a cross-sectional survey study. Arch Dermatol Res. 2024;316:575.
- Hazim AH. Empowering advanced clinical practitioners in managing acute dermatological emergencies. Br J Nurs. 2024;33:448-455.
- Macklis P, Kaffenberger B, Kirven R, et al. Dermatology diagnostic accuracy is improved by artificial intelligence-generated differential diagnoses. Int J Dermatol. 2025;64:960-962.
Hospital Dermatology: Review of Research in 2024-2025
Hospital Dermatology: Review of Research in 2024-2025
Practice Points
- In suspected drug reaction with eosinophilia and systemic symptoms, discontinue the offending drug; test for human herpesvirus 6, Epstein-Barr virus, and cytomegalovirus when available; and treat moderate cases with low-dose corticosteroids. Reserve interleukin 5 inhibitors for refractory disease.
- For Stevens-Johnson syndrome and toxic epidermal necrolysis (TEN), apply Niigata diagnostic criteria and clinical risk score for TEN, refer patients with 10% or more body surface area detachment to higher-level or burn care, and consider targeted therapies for refractory cases.
- When assessing infectious rashes, consider measles, dengue, mpox, orthopoxviruses, and resistant dermatophytes. Review the patient’s vaccination and travel history, isolate suspected measles cases, and confirm atypical tinea with culture or DNA testing.
- To reduce unnecessary admissions and optimize care for hidradenitis suppurativa, avoid misdiagnosing flares as sepsis, implement multidisciplinary protocols, consider selective intravenous antibiotics, and support expanded inpatient dermatology coverage.