User login
Reflectance confocal microscopy: The future looks bright
CHICAGO – The future looks bright for to rule out malignancy, Ann M. John, MD, asserted at the annual meeting of the American College of Mohs Surgery.
“With the advent of dermoscopy, dermatologists were able to elucidate both benign and malignant patterns to help further guide their decision to biopsy or not. This increased diagnostic accuracy of suspicious lesions by 30%, while reducing the benign to malignant ratio of biopsies performed from 18:1 to 4:1. However, there are still lesions that are equivocal on dermoscopy, as we all know, and for this, there’s reflectance confocal microscopy,” observed Dr. John, of Robert Wood Johnson Medical School, New Brunswick, N.J.
RCM is a device technology that’s been cleared by the Food and Drug Administration since 2008 for the imaging of clinically suspicious lesions. It employs laser scanning to assess the light-scattering properties of cells in the epidermis and dermis, generating images with resolution comparable to histology.
RCM took a back seat initially while American dermatologists were gradually coming to embrace dermoscopy, which their European colleagues had done years earlier. Now, with the availability of handheld RCM for use in the dermatology clinic, expect RCM to assume a growing role in daily practice.
To illustrate the power of RCM as a diagnostic aid, she presented a single-center retrospective study of 1,189 clinically suspicious skin lesions that were equivocal on dermoscopy and then assessed using RCM with 1 year of subsequent patient follow-up. Overall, 155 lesions were deemed positive for cancer or atypia by RCM, while 1,034 were determined to be benign. Of those 155, 46 lesions were considered false positives because of their benign appearance on histologic inspection of the biopsy sample. Only 2 of the 1,034 lesions identified as negative by RCM proved to be false negatives on the basis of clinical changes within 1 year.
The overall sensitivity and specificity of RCM was 98.2% and 99.8%, respectively, with a positive predictive value of 70.3% and a negative predictive value of 99.8%.
The entire RCM procedure takes a skilled technician 15-20 minutes per lesion. As a practical matter, other investigators have estimated that RCM results in a cost savings of about $308,000 per million health plan members per year by reducing the need for biopsies (Dermatol Clin. 2016 Oct;34[4]:367-75).
In addition to evaluating clinically suspicious lesions, other situations in which RCM offers practical value include its use directly before the first cut during Mohs surgery in order to determine the margins of atypia; ex vivo imaging of Mohs margins, which has been shown to be comparable with frozen sections in accuracy but takes only one-third of the time; and imaging of biopsied lesions in order to determine the diagnosis relatively quickly, Dr. John noted.
She reported having no financial conflicts regarding her study.
CHICAGO – The future looks bright for to rule out malignancy, Ann M. John, MD, asserted at the annual meeting of the American College of Mohs Surgery.
“With the advent of dermoscopy, dermatologists were able to elucidate both benign and malignant patterns to help further guide their decision to biopsy or not. This increased diagnostic accuracy of suspicious lesions by 30%, while reducing the benign to malignant ratio of biopsies performed from 18:1 to 4:1. However, there are still lesions that are equivocal on dermoscopy, as we all know, and for this, there’s reflectance confocal microscopy,” observed Dr. John, of Robert Wood Johnson Medical School, New Brunswick, N.J.
RCM is a device technology that’s been cleared by the Food and Drug Administration since 2008 for the imaging of clinically suspicious lesions. It employs laser scanning to assess the light-scattering properties of cells in the epidermis and dermis, generating images with resolution comparable to histology.
RCM took a back seat initially while American dermatologists were gradually coming to embrace dermoscopy, which their European colleagues had done years earlier. Now, with the availability of handheld RCM for use in the dermatology clinic, expect RCM to assume a growing role in daily practice.
To illustrate the power of RCM as a diagnostic aid, she presented a single-center retrospective study of 1,189 clinically suspicious skin lesions that were equivocal on dermoscopy and then assessed using RCM with 1 year of subsequent patient follow-up. Overall, 155 lesions were deemed positive for cancer or atypia by RCM, while 1,034 were determined to be benign. Of those 155, 46 lesions were considered false positives because of their benign appearance on histologic inspection of the biopsy sample. Only 2 of the 1,034 lesions identified as negative by RCM proved to be false negatives on the basis of clinical changes within 1 year.
The overall sensitivity and specificity of RCM was 98.2% and 99.8%, respectively, with a positive predictive value of 70.3% and a negative predictive value of 99.8%.
The entire RCM procedure takes a skilled technician 15-20 minutes per lesion. As a practical matter, other investigators have estimated that RCM results in a cost savings of about $308,000 per million health plan members per year by reducing the need for biopsies (Dermatol Clin. 2016 Oct;34[4]:367-75).
In addition to evaluating clinically suspicious lesions, other situations in which RCM offers practical value include its use directly before the first cut during Mohs surgery in order to determine the margins of atypia; ex vivo imaging of Mohs margins, which has been shown to be comparable with frozen sections in accuracy but takes only one-third of the time; and imaging of biopsied lesions in order to determine the diagnosis relatively quickly, Dr. John noted.
She reported having no financial conflicts regarding her study.
CHICAGO – The future looks bright for to rule out malignancy, Ann M. John, MD, asserted at the annual meeting of the American College of Mohs Surgery.
“With the advent of dermoscopy, dermatologists were able to elucidate both benign and malignant patterns to help further guide their decision to biopsy or not. This increased diagnostic accuracy of suspicious lesions by 30%, while reducing the benign to malignant ratio of biopsies performed from 18:1 to 4:1. However, there are still lesions that are equivocal on dermoscopy, as we all know, and for this, there’s reflectance confocal microscopy,” observed Dr. John, of Robert Wood Johnson Medical School, New Brunswick, N.J.
RCM is a device technology that’s been cleared by the Food and Drug Administration since 2008 for the imaging of clinically suspicious lesions. It employs laser scanning to assess the light-scattering properties of cells in the epidermis and dermis, generating images with resolution comparable to histology.
RCM took a back seat initially while American dermatologists were gradually coming to embrace dermoscopy, which their European colleagues had done years earlier. Now, with the availability of handheld RCM for use in the dermatology clinic, expect RCM to assume a growing role in daily practice.
To illustrate the power of RCM as a diagnostic aid, she presented a single-center retrospective study of 1,189 clinically suspicious skin lesions that were equivocal on dermoscopy and then assessed using RCM with 1 year of subsequent patient follow-up. Overall, 155 lesions were deemed positive for cancer or atypia by RCM, while 1,034 were determined to be benign. Of those 155, 46 lesions were considered false positives because of their benign appearance on histologic inspection of the biopsy sample. Only 2 of the 1,034 lesions identified as negative by RCM proved to be false negatives on the basis of clinical changes within 1 year.
The overall sensitivity and specificity of RCM was 98.2% and 99.8%, respectively, with a positive predictive value of 70.3% and a negative predictive value of 99.8%.
The entire RCM procedure takes a skilled technician 15-20 minutes per lesion. As a practical matter, other investigators have estimated that RCM results in a cost savings of about $308,000 per million health plan members per year by reducing the need for biopsies (Dermatol Clin. 2016 Oct;34[4]:367-75).
In addition to evaluating clinically suspicious lesions, other situations in which RCM offers practical value include its use directly before the first cut during Mohs surgery in order to determine the margins of atypia; ex vivo imaging of Mohs margins, which has been shown to be comparable with frozen sections in accuracy but takes only one-third of the time; and imaging of biopsied lesions in order to determine the diagnosis relatively quickly, Dr. John noted.
She reported having no financial conflicts regarding her study.
REPORTING FROM THE ACMS ANNUAL MEETING
Key clinical point: The future looks bright for reflectance confocal microscopy in dermatology.
Major finding: The sensitivity and specificity of reflectance confocal microscopy for diagnosis of skin cancer in patients with equivocal dermoscopic findings was 98.2% and 99.8%, respectively.
Study details: This retrospective single center study included 1,189 clinically suspicious skin lesions with equivocal dermoscopy findings, which were then evaluated using reflectance confocal microscopy.
Disclosures: The presenter reported having no financial conflicts regarding her study.
Mohs underutilized for melanoma of head and neck
CHICAGO – Contemporary national guidelines undervalue the benefits of Mohs micrographic surgery for patients with melanoma of the head and neck, William C. Fix asserted at the annual meeting of the American College of Mohs Surgery.

Mr. Fix, a medical student at the University of Pennsylvania, Philadelphia, presented a single-center retrospective study of 13,644 cases of head and neck skin cancer treated with Mohs micrographic surgery (MMS) for margin control. The cohort included 1,065 melanomas in situ, 410 invasive melanomas, more than 8,700 basal cell carcinomas, and 3,343 squamous cell carcinomas.
Mr. Fix and his coinvestigators undertook this observational study because they identified a gap in current guidelines for treatment of skin cancers of the head and neck. For example, the National Comprehensive Cancer Network recommends margin control at the time of primary surgery for BCCs and SCCs deemed at high risk for local recurrence and defines what those high-risk features are. For melanomas, however, the guidelines recommend wide local excision, even though that approach has roughly a 10% recurrence rate, compared with less than 1% for MMS.
Moreover, the 2012 appropriate use criteria for MMS put forth by the American Academy of Dermatology in concert with several other medical societies are unclear about invasive melanoma. As a result of this lack of guidance, the use of margin control in primary surgery for melanoma is applied in less than 4% of cases, according to Mr. Fix.
The University of Pennsylvania data he presented showed that melanomas of the head and neck were significantly more likely to be large in size, to be poorly defined, and to have other high-risk features for local recurrence than were the BCCs and SCCs. In a multivariate logistic regression analysis controlling for high-risk characteristics, melanomas were independently associated with a twofold increased likelihood of requiring flap reconstruction compared with BCCs and SCCs of the head and neck.
“We’ve shown that melanomas have high-risk features for local recurrence, possibly to a greater extent than BCCs and SCCs. These features help us triage resource use for BCC and SCC. Could these same features help us make decisions for melanomas?” he asked rhetorically.
Mr. Fix reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
CHICAGO – Contemporary national guidelines undervalue the benefits of Mohs micrographic surgery for patients with melanoma of the head and neck, William C. Fix asserted at the annual meeting of the American College of Mohs Surgery.

Mr. Fix, a medical student at the University of Pennsylvania, Philadelphia, presented a single-center retrospective study of 13,644 cases of head and neck skin cancer treated with Mohs micrographic surgery (MMS) for margin control. The cohort included 1,065 melanomas in situ, 410 invasive melanomas, more than 8,700 basal cell carcinomas, and 3,343 squamous cell carcinomas.
Mr. Fix and his coinvestigators undertook this observational study because they identified a gap in current guidelines for treatment of skin cancers of the head and neck. For example, the National Comprehensive Cancer Network recommends margin control at the time of primary surgery for BCCs and SCCs deemed at high risk for local recurrence and defines what those high-risk features are. For melanomas, however, the guidelines recommend wide local excision, even though that approach has roughly a 10% recurrence rate, compared with less than 1% for MMS.
Moreover, the 2012 appropriate use criteria for MMS put forth by the American Academy of Dermatology in concert with several other medical societies are unclear about invasive melanoma. As a result of this lack of guidance, the use of margin control in primary surgery for melanoma is applied in less than 4% of cases, according to Mr. Fix.
The University of Pennsylvania data he presented showed that melanomas of the head and neck were significantly more likely to be large in size, to be poorly defined, and to have other high-risk features for local recurrence than were the BCCs and SCCs. In a multivariate logistic regression analysis controlling for high-risk characteristics, melanomas were independently associated with a twofold increased likelihood of requiring flap reconstruction compared with BCCs and SCCs of the head and neck.
“We’ve shown that melanomas have high-risk features for local recurrence, possibly to a greater extent than BCCs and SCCs. These features help us triage resource use for BCC and SCC. Could these same features help us make decisions for melanomas?” he asked rhetorically.
Mr. Fix reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
CHICAGO – Contemporary national guidelines undervalue the benefits of Mohs micrographic surgery for patients with melanoma of the head and neck, William C. Fix asserted at the annual meeting of the American College of Mohs Surgery.

Mr. Fix, a medical student at the University of Pennsylvania, Philadelphia, presented a single-center retrospective study of 13,644 cases of head and neck skin cancer treated with Mohs micrographic surgery (MMS) for margin control. The cohort included 1,065 melanomas in situ, 410 invasive melanomas, more than 8,700 basal cell carcinomas, and 3,343 squamous cell carcinomas.
Mr. Fix and his coinvestigators undertook this observational study because they identified a gap in current guidelines for treatment of skin cancers of the head and neck. For example, the National Comprehensive Cancer Network recommends margin control at the time of primary surgery for BCCs and SCCs deemed at high risk for local recurrence and defines what those high-risk features are. For melanomas, however, the guidelines recommend wide local excision, even though that approach has roughly a 10% recurrence rate, compared with less than 1% for MMS.
Moreover, the 2012 appropriate use criteria for MMS put forth by the American Academy of Dermatology in concert with several other medical societies are unclear about invasive melanoma. As a result of this lack of guidance, the use of margin control in primary surgery for melanoma is applied in less than 4% of cases, according to Mr. Fix.
The University of Pennsylvania data he presented showed that melanomas of the head and neck were significantly more likely to be large in size, to be poorly defined, and to have other high-risk features for local recurrence than were the BCCs and SCCs. In a multivariate logistic regression analysis controlling for high-risk characteristics, melanomas were independently associated with a twofold increased likelihood of requiring flap reconstruction compared with BCCs and SCCs of the head and neck.
“We’ve shown that melanomas have high-risk features for local recurrence, possibly to a greater extent than BCCs and SCCs. These features help us triage resource use for BCC and SCC. Could these same features help us make decisions for melanomas?” he asked rhetorically.
Mr. Fix reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
REPORTING FROM THE ACMS ANNUAL MEETING
Key clinical point: Margin control at the time of primary surgery for melanoma of the head and neck makes sense.
Major finding: Patients with a melanoma of the head and neck were twice as likely to require secondary flap reconstruction compared with patients with a basal cell carcinoma or squamous cell carcinoma of the head and neck.
Study details: A retrospective single-center study of 13,644 cases of skin cancer of the head and neck treated with Mohs surgery.
Disclosures: The presenter reported having no financial conflicts regarding the study, which was conducted free of commercial support.
ESMO scale offers guidance on cancer targets
The European Society for Medical Oncology (ESMO) has published a proposed scale that would rank molecular targets for various cancers by how well they can be treated with new or emerging drugs.
The ESMO Scale of Clinical Actionability for Molecular Targets is designed to “harmonize and standardize the reporting and interpretation of clinically relevant genomics data,” according to Joaquin Mateo, MD, PhD, from the Vall d’Hebron Institute of Oncology in Barcelona, Spain, and his fellow members of the ESMO Translational Research and Precision Medicine Working Group.
“A major challenge for oncologists in the clinic is to distinguish between findings that represent proven clinical value or potential value based on preliminary clinical or preclinical evidence from hypothetical gene-drug matches and findings that are currently irrelevant for clinical practice,” they wrote in Annals of Oncology.
The scale groups targets into one of six tiers based on levels of evidence ranging from the gold standard of prospective, randomized clinical trials to targets for which there are no evidence and only hypothetical actionability. The primary goal is to help oncologists assign priority to potential targets when they review results of gene-sequencing panels for individual patients, according to the developers.
Briefly, the six tiers are:
Tier I includes targets that are agreed to be suitable for routine use and a recommended specific drug when a specific molecular alteration is detected. Examples include trastuzumab for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, and inhibitors of epidermal growth factor receptor (EGFR) in patients with non–small cell lung cancer positive for EGFR mutations.
Tier II includes “investigational targets that likely define a patient population that benefits from a targeted drug but additional data are needed.” This tier includes agents that work in the phosphatidylinostiol 3-kinase pathway.
Tier III is similar to Tier II, in that it includes investigational targets that define a patient population with proven benefit from a targeted therapy, but in this case the target is detected in a different tumor type that has not previously been studied. For example, the targeted agent vemurafenib (Zelboraf), which extends survival of patients with metastatic melanomas carrying the BRAF V600E mutation, has only limited activity against BRAF-mutated colorectal cancers.
Tier IV includes targets with preclinical evidence of actionability.
Tier V includes targets with “evidence of relevant antitumor activity, not resulting in clinical meaningful benefit as single treatment but supporting development of cotargeting approaches.” The authors cite the example of PIK3CA inhibitors in patients with estrogen receptor–positive, HER2-negative breast cancers who also have PIK3CA activating mutations. In clinical trials, this strategy led to objective responses but not change outcomes.
The final tier is not Tier VI, as might be expected, but Tier X, with the X in this case being the unknown – that is, alterations/mutations for which there is neither preclinical nor clinical evidence to support their hypothetical use as a drug target.
“This clinical benefit–centered classification system offers a common language for all the actors involved in clinical cancer drug development. Its implementation in sequencing reports, tumor boards, and scientific communication can enable precise treatment decisions and facilitate discussions with patients about novel therapeutic options,” Dr. Mateo and his associates wrote in their conclusion.
The development process was supported by ESMO. Multiple coauthors reported financial relationships with various companies as well as grants/support from other foundations or charities.
SOURCE: Mateo J et al. Ann Oncol. 2018 Aug 21. doi: 10.1093/annonc/mdy263.
The European Society for Medical Oncology (ESMO) has published a proposed scale that would rank molecular targets for various cancers by how well they can be treated with new or emerging drugs.
The ESMO Scale of Clinical Actionability for Molecular Targets is designed to “harmonize and standardize the reporting and interpretation of clinically relevant genomics data,” according to Joaquin Mateo, MD, PhD, from the Vall d’Hebron Institute of Oncology in Barcelona, Spain, and his fellow members of the ESMO Translational Research and Precision Medicine Working Group.
“A major challenge for oncologists in the clinic is to distinguish between findings that represent proven clinical value or potential value based on preliminary clinical or preclinical evidence from hypothetical gene-drug matches and findings that are currently irrelevant for clinical practice,” they wrote in Annals of Oncology.
The scale groups targets into one of six tiers based on levels of evidence ranging from the gold standard of prospective, randomized clinical trials to targets for which there are no evidence and only hypothetical actionability. The primary goal is to help oncologists assign priority to potential targets when they review results of gene-sequencing panels for individual patients, according to the developers.
Briefly, the six tiers are:
Tier I includes targets that are agreed to be suitable for routine use and a recommended specific drug when a specific molecular alteration is detected. Examples include trastuzumab for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, and inhibitors of epidermal growth factor receptor (EGFR) in patients with non–small cell lung cancer positive for EGFR mutations.
Tier II includes “investigational targets that likely define a patient population that benefits from a targeted drug but additional data are needed.” This tier includes agents that work in the phosphatidylinostiol 3-kinase pathway.
Tier III is similar to Tier II, in that it includes investigational targets that define a patient population with proven benefit from a targeted therapy, but in this case the target is detected in a different tumor type that has not previously been studied. For example, the targeted agent vemurafenib (Zelboraf), which extends survival of patients with metastatic melanomas carrying the BRAF V600E mutation, has only limited activity against BRAF-mutated colorectal cancers.
Tier IV includes targets with preclinical evidence of actionability.
Tier V includes targets with “evidence of relevant antitumor activity, not resulting in clinical meaningful benefit as single treatment but supporting development of cotargeting approaches.” The authors cite the example of PIK3CA inhibitors in patients with estrogen receptor–positive, HER2-negative breast cancers who also have PIK3CA activating mutations. In clinical trials, this strategy led to objective responses but not change outcomes.
The final tier is not Tier VI, as might be expected, but Tier X, with the X in this case being the unknown – that is, alterations/mutations for which there is neither preclinical nor clinical evidence to support their hypothetical use as a drug target.
“This clinical benefit–centered classification system offers a common language for all the actors involved in clinical cancer drug development. Its implementation in sequencing reports, tumor boards, and scientific communication can enable precise treatment decisions and facilitate discussions with patients about novel therapeutic options,” Dr. Mateo and his associates wrote in their conclusion.
The development process was supported by ESMO. Multiple coauthors reported financial relationships with various companies as well as grants/support from other foundations or charities.
SOURCE: Mateo J et al. Ann Oncol. 2018 Aug 21. doi: 10.1093/annonc/mdy263.
The European Society for Medical Oncology (ESMO) has published a proposed scale that would rank molecular targets for various cancers by how well they can be treated with new or emerging drugs.
The ESMO Scale of Clinical Actionability for Molecular Targets is designed to “harmonize and standardize the reporting and interpretation of clinically relevant genomics data,” according to Joaquin Mateo, MD, PhD, from the Vall d’Hebron Institute of Oncology in Barcelona, Spain, and his fellow members of the ESMO Translational Research and Precision Medicine Working Group.
“A major challenge for oncologists in the clinic is to distinguish between findings that represent proven clinical value or potential value based on preliminary clinical or preclinical evidence from hypothetical gene-drug matches and findings that are currently irrelevant for clinical practice,” they wrote in Annals of Oncology.
The scale groups targets into one of six tiers based on levels of evidence ranging from the gold standard of prospective, randomized clinical trials to targets for which there are no evidence and only hypothetical actionability. The primary goal is to help oncologists assign priority to potential targets when they review results of gene-sequencing panels for individual patients, according to the developers.
Briefly, the six tiers are:
Tier I includes targets that are agreed to be suitable for routine use and a recommended specific drug when a specific molecular alteration is detected. Examples include trastuzumab for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, and inhibitors of epidermal growth factor receptor (EGFR) in patients with non–small cell lung cancer positive for EGFR mutations.
Tier II includes “investigational targets that likely define a patient population that benefits from a targeted drug but additional data are needed.” This tier includes agents that work in the phosphatidylinostiol 3-kinase pathway.
Tier III is similar to Tier II, in that it includes investigational targets that define a patient population with proven benefit from a targeted therapy, but in this case the target is detected in a different tumor type that has not previously been studied. For example, the targeted agent vemurafenib (Zelboraf), which extends survival of patients with metastatic melanomas carrying the BRAF V600E mutation, has only limited activity against BRAF-mutated colorectal cancers.
Tier IV includes targets with preclinical evidence of actionability.
Tier V includes targets with “evidence of relevant antitumor activity, not resulting in clinical meaningful benefit as single treatment but supporting development of cotargeting approaches.” The authors cite the example of PIK3CA inhibitors in patients with estrogen receptor–positive, HER2-negative breast cancers who also have PIK3CA activating mutations. In clinical trials, this strategy led to objective responses but not change outcomes.
The final tier is not Tier VI, as might be expected, but Tier X, with the X in this case being the unknown – that is, alterations/mutations for which there is neither preclinical nor clinical evidence to support their hypothetical use as a drug target.
“This clinical benefit–centered classification system offers a common language for all the actors involved in clinical cancer drug development. Its implementation in sequencing reports, tumor boards, and scientific communication can enable precise treatment decisions and facilitate discussions with patients about novel therapeutic options,” Dr. Mateo and his associates wrote in their conclusion.
The development process was supported by ESMO. Multiple coauthors reported financial relationships with various companies as well as grants/support from other foundations or charities.
SOURCE: Mateo J et al. Ann Oncol. 2018 Aug 21. doi: 10.1093/annonc/mdy263.
FROM ANNALS OF ONCOLOGY
Key clinical point: The scale is intended to standardize reporting and interpretation of cancer gene panel results to help oncologists plan treatment.
Major finding: The scale divides current and future therapeutic targets into tiers based on levels of clinical and preclinical evidence.
Study details: Proposed guiding principles for a classification system developed by the Translational Research and Precision Medicine Working Group of the European Society of Medical Oncology.
Disclosures: The development process was supported by ESMO. Multiple coauthors reported financial relationships with various companies as well as grants/support from other foundations or charities.
Source: Mateo J et al. Ann Oncol. 2018 Aug 21. doi: 10.1093/annonc/mdy263.
PD1 mRNA may predict response to anti-PD1 monotherapy across cancer types
Expression of programmed death-1 (PD1) mRNA may predict outcomes after anti-PD1 therapy across cancer types, according to investigators.
High levels of PD1 mRNA were significantly associated with response to anti-PD1 monotherapy, investigators found in an analysis of tumor samples from 117 patients with advanced cancers who had received either nivolumab or pembrolizumab.
Further validation of PD1 mRNA is warranted to help select patients who might benefit from an anti-PD1 treatment strategy, wrote investigator Aleix Prat, MD, PhD, of Hospital Clínic of Barcelona, and his coinvestigators.
“Identification of reproducible biomarkers that can be applied to predict benefit of anti-PD1 monotherapy might be of clinical value,” Dr. Prat and his coinvestigators note. The report is in Annals of Oncology.
Previous studies support use of PDL1 expression by immunohistochemistry as a biomarker for pembrolizumab in non–small-cell lung cancer; however, that biomarker has some technical limitations, and has not been predictive in other cancer types and with other anti-PD1 drugs including nivolumab, Dr. Prat and his coinvestigators said.
The 117 tumor samples evaluated for PD1 mRNA expression comprised 59 advanced melanomas, 32 non–small-cell lung cancers, 14 renal cell cancers, and 12 other tumors, according to the report. Sixty-two of the patients had been treated with pembrolizumab, and 55 received nivolumab.
About one-quarter of the samples (28.2%) were classified as “PD1-high” with a preestablished cutoff value developed by Dr. Prat and his coinvestigators.
The overall response rate was 51.5% for the patients who had PD1-high tumors, versus 23.8% for the remaining tumors (P less than .001). Those non-PD1-high tumors, when grouped as PD1-intermediate and PD1-low, had overall response rates of 26.6% and 15.0%, respectively.
Median progression-free survival was 8.17 months for PD1-high tumors and 3.18 months for the rest of the tumors (P = .011), the report shows. Similarly, overall survival was a median of 23.4 months for PD1-high tumors and 14.9 months for the rest (P = .330).
Dr. Prat and his colleagues detailed earlier investigations validating PD1 mRNA as a biomarker, including an analysis of PD1 and immune-related gene expression in 10,078 samples from 34 cancer types in The Cancer Genome Atlas.
In that analysis, PD1 was strongly correlated with a group of 30 genes that were “significantly enriched” in biological processes including CD8-T-cell activation, the investigators said.
Moreover, high levels of PD1 mRNA expression were strongly correlated with overall response rates reported in the literature for anti-PD1 monotherapy, they added.
They also reported results of an analysis they used to develop the PD1-high cutoff value. That analysis was based on PD1 mRNA expression in 773 tumor samples across 17 tumor types.
“Our results are consistent with the hypothesis that identification of a preexisting and stable adaptive immune response using PD1 mRNA expression predicts outcome across cancer-types following anti-PD1 monotherapy,” the researchers wrote.
The work was partially sponsored by Instituto de Salud Carlos III, Spanish Society of Medical Oncology, Banco Bilbao Vizcaya Argentaria Foundation, Pas a Pas, Save the Mama, and the Breast Cancer Research Foundation. Dr. Prat disclosed an advisory role with Nanostring Technologies.
SOURCE: Paré L et al. Ann Oncol. 2018 Aug 27. doi: 10.1093/annonc/mdy335.
Expression of programmed death-1 (PD1) mRNA may predict outcomes after anti-PD1 therapy across cancer types, according to investigators.
High levels of PD1 mRNA were significantly associated with response to anti-PD1 monotherapy, investigators found in an analysis of tumor samples from 117 patients with advanced cancers who had received either nivolumab or pembrolizumab.
Further validation of PD1 mRNA is warranted to help select patients who might benefit from an anti-PD1 treatment strategy, wrote investigator Aleix Prat, MD, PhD, of Hospital Clínic of Barcelona, and his coinvestigators.
“Identification of reproducible biomarkers that can be applied to predict benefit of anti-PD1 monotherapy might be of clinical value,” Dr. Prat and his coinvestigators note. The report is in Annals of Oncology.
Previous studies support use of PDL1 expression by immunohistochemistry as a biomarker for pembrolizumab in non–small-cell lung cancer; however, that biomarker has some technical limitations, and has not been predictive in other cancer types and with other anti-PD1 drugs including nivolumab, Dr. Prat and his coinvestigators said.
The 117 tumor samples evaluated for PD1 mRNA expression comprised 59 advanced melanomas, 32 non–small-cell lung cancers, 14 renal cell cancers, and 12 other tumors, according to the report. Sixty-two of the patients had been treated with pembrolizumab, and 55 received nivolumab.
About one-quarter of the samples (28.2%) were classified as “PD1-high” with a preestablished cutoff value developed by Dr. Prat and his coinvestigators.
The overall response rate was 51.5% for the patients who had PD1-high tumors, versus 23.8% for the remaining tumors (P less than .001). Those non-PD1-high tumors, when grouped as PD1-intermediate and PD1-low, had overall response rates of 26.6% and 15.0%, respectively.
Median progression-free survival was 8.17 months for PD1-high tumors and 3.18 months for the rest of the tumors (P = .011), the report shows. Similarly, overall survival was a median of 23.4 months for PD1-high tumors and 14.9 months for the rest (P = .330).
Dr. Prat and his colleagues detailed earlier investigations validating PD1 mRNA as a biomarker, including an analysis of PD1 and immune-related gene expression in 10,078 samples from 34 cancer types in The Cancer Genome Atlas.
In that analysis, PD1 was strongly correlated with a group of 30 genes that were “significantly enriched” in biological processes including CD8-T-cell activation, the investigators said.
Moreover, high levels of PD1 mRNA expression were strongly correlated with overall response rates reported in the literature for anti-PD1 monotherapy, they added.
They also reported results of an analysis they used to develop the PD1-high cutoff value. That analysis was based on PD1 mRNA expression in 773 tumor samples across 17 tumor types.
“Our results are consistent with the hypothesis that identification of a preexisting and stable adaptive immune response using PD1 mRNA expression predicts outcome across cancer-types following anti-PD1 monotherapy,” the researchers wrote.
The work was partially sponsored by Instituto de Salud Carlos III, Spanish Society of Medical Oncology, Banco Bilbao Vizcaya Argentaria Foundation, Pas a Pas, Save the Mama, and the Breast Cancer Research Foundation. Dr. Prat disclosed an advisory role with Nanostring Technologies.
SOURCE: Paré L et al. Ann Oncol. 2018 Aug 27. doi: 10.1093/annonc/mdy335.
Expression of programmed death-1 (PD1) mRNA may predict outcomes after anti-PD1 therapy across cancer types, according to investigators.
High levels of PD1 mRNA were significantly associated with response to anti-PD1 monotherapy, investigators found in an analysis of tumor samples from 117 patients with advanced cancers who had received either nivolumab or pembrolizumab.
Further validation of PD1 mRNA is warranted to help select patients who might benefit from an anti-PD1 treatment strategy, wrote investigator Aleix Prat, MD, PhD, of Hospital Clínic of Barcelona, and his coinvestigators.
“Identification of reproducible biomarkers that can be applied to predict benefit of anti-PD1 monotherapy might be of clinical value,” Dr. Prat and his coinvestigators note. The report is in Annals of Oncology.
Previous studies support use of PDL1 expression by immunohistochemistry as a biomarker for pembrolizumab in non–small-cell lung cancer; however, that biomarker has some technical limitations, and has not been predictive in other cancer types and with other anti-PD1 drugs including nivolumab, Dr. Prat and his coinvestigators said.
The 117 tumor samples evaluated for PD1 mRNA expression comprised 59 advanced melanomas, 32 non–small-cell lung cancers, 14 renal cell cancers, and 12 other tumors, according to the report. Sixty-two of the patients had been treated with pembrolizumab, and 55 received nivolumab.
About one-quarter of the samples (28.2%) were classified as “PD1-high” with a preestablished cutoff value developed by Dr. Prat and his coinvestigators.
The overall response rate was 51.5% for the patients who had PD1-high tumors, versus 23.8% for the remaining tumors (P less than .001). Those non-PD1-high tumors, when grouped as PD1-intermediate and PD1-low, had overall response rates of 26.6% and 15.0%, respectively.
Median progression-free survival was 8.17 months for PD1-high tumors and 3.18 months for the rest of the tumors (P = .011), the report shows. Similarly, overall survival was a median of 23.4 months for PD1-high tumors and 14.9 months for the rest (P = .330).
Dr. Prat and his colleagues detailed earlier investigations validating PD1 mRNA as a biomarker, including an analysis of PD1 and immune-related gene expression in 10,078 samples from 34 cancer types in The Cancer Genome Atlas.
In that analysis, PD1 was strongly correlated with a group of 30 genes that were “significantly enriched” in biological processes including CD8-T-cell activation, the investigators said.
Moreover, high levels of PD1 mRNA expression were strongly correlated with overall response rates reported in the literature for anti-PD1 monotherapy, they added.
They also reported results of an analysis they used to develop the PD1-high cutoff value. That analysis was based on PD1 mRNA expression in 773 tumor samples across 17 tumor types.
“Our results are consistent with the hypothesis that identification of a preexisting and stable adaptive immune response using PD1 mRNA expression predicts outcome across cancer-types following anti-PD1 monotherapy,” the researchers wrote.
The work was partially sponsored by Instituto de Salud Carlos III, Spanish Society of Medical Oncology, Banco Bilbao Vizcaya Argentaria Foundation, Pas a Pas, Save the Mama, and the Breast Cancer Research Foundation. Dr. Prat disclosed an advisory role with Nanostring Technologies.
SOURCE: Paré L et al. Ann Oncol. 2018 Aug 27. doi: 10.1093/annonc/mdy335.
FROM ANNALS OF ONCOLOGY
Key clinical point: Expression of programmed death-1 (PD1) mRNA may predict outcomes after anti-PD1 therapy across cancer types.
Major finding: The overall response rate was 51.5% for the patients who had PD1-high tumors, versus 23.8% for the remaining tumors (P less than .001).
Study details: Evaluation of PD1 mRNA expression in tumor samples from 117 patients treated with either nivolumab or pembrolizumab monotherapy.
Disclosures: The work was partially sponsored by the Instituto de Salud Carlos III, Spanish Society of Medical Oncology, Banco Bilbao Vizcaya Argentaria Foundation, Pas a Pas, Save the Mama, and the Breast Cancer Research Foundation. One author disclosed an advisory role with Nanostring Technologies.
Source: Paré L et al. Ann Oncol. 2018 Aug 27. doi: 10.1093/annonc/mdy335.
Nivolumab plus ipilimumab effective in melanoma brain metastases
Treatment with nivolumab plus ipilimumab resulted in clinically meaningful efficacy for melanoma patients with asymptomatic, previously untreated brain metastases, results of an open-label, multicenter, phase 2 study have shown.
The combination of these two immune checkpoint inhibitors produced intracranial responses in more than half of the patients treated, and perhaps more importantly, according to the study investigators, the combination treatment prevented intracranial progression for more than 6 months in 64% of the study population.
“These results are relevant in a population in whom progression can quickly result in substantial neurologic symptoms, functional impairment, and the need for glucocorticoid therapy,” the study investigators wrote in the New England Journal of Medicine.
The investigators, led by Hussein A. Tawbi, MD, PhD, of the University of Texas MD Anderson Cancer, Houston, initially enrolled 101 patients with histologically confirmed melanoma and metastases to the brain that were asymptomatic. All patients had an Eastern Cooperative Oncology Group performance status of 0-1 and had not received systemic glucocorticoid therapy within 10 days of study treatment.
The primary endpoint of the study was the rate of intracranial benefit, defined as the percentage of patients with complete response, partial response, or stable disease for at least 6 months after starting treatment.
For 94 patients with at least 6 months of follow-up at the time of analysis (median follow-up, 14 months), the rate of intracranial benefit was 57%, including complete responses in 26%, partial responses in 30%, and stable disease in 2%, the investigators reported. The rate of extracranial benefit was similar, at 56%.
The 6-month rate of progression-free survival was 64.2% for intracranial assessments, while the 6-month overall survival rate was 92.3%, according to results of an initial assessment.
Grade 3 or 4 adverse events thought to be related to treatment occurred in 55% of patients and led to treatment discontinuation in 20%; the most common were increased levels of ALT and AST.
Dr. Tawbi and his colleagues said that, while cross-trial comparisons have inherent limitations, the rate of intracranial response seen in this trial is similar to what was seen in the COMBI-MB study of dabrafenib plus trametinib in patients with BRAF-mutant melanoma and brain metastases. However, in that study, published in 2017 in the Lancet, the combination of a BRAF inhibitor and MEK inhibitor had rates of intracranial response and progression-free survival that were “substantially shorter” than the rates of extracranial response and progression-free survival.
“In our study, the use of immunotherapy seemed capable of inducing intracranial responses that were very similar to extracranial responses in character, depth, and duration,” they wrote.
Dr. Tawbi and his coinvestigators enrolled an additional 20 symptomatic patients with brain metastases following a study protocol amendment; however, results from that cohort are not being reported yet because of inadequate follow-up length, they said.
The study was supported by Bristol-Myers Squibb and a grant from the National Cancer Institute. Dr. Tawbi reported disclosures related to Bristol-Myers Squibb, Merck, Genentech, and Novartis. His coauthors reported additional disclosures related to MedImmune, AstraZeneca, Dynavax Technologies, Genoptix, Exelixis, Acceleron Pharma, and Eisai, among others.
SOURCE: Tawbi HA et al. N Engl J Med. 2018 Aug 23. doi: 10.1056/NEJMoa1805453.
These data show that checkpoint inhibitors can be similarly effective in CNS metastases as they can be in extracranial metastases related to melanoma, according to Samra Turajlic, MD, PhD, and James Larkin, FRCP, PhD, of the Renal and Skin Units at the Royal Marsden National Health Service Foundation Trust in London.
Based on the study results, larger trials are warranted, including patients with CNS metastases from melanoma, kidney, lung, and other cancers where checkpoint inhibitors have demonstrated efficacy, Dr. Turajlic, who is also with the Translational Cancer Therapeutics Laboratory at the Francis Crick Institute in London, and Dr. Larkin wrote in an editorial.
“Such patients should no longer generally be excluded from clinical trials,” they wrote.
While the study by Dr. Tawbi and his colleagues was small, they added, its results are relevant to clinical practice because of the high rate of response, rapid response time, and side effect profile, which was manageable.
In fact, the nivolumab plus ipilimumab regimen described in this study should be considered first-line therapy for all patients who meet the study’s inclusion criteria, they asserted.
However, the results should “absolutely not” be extrapolated to higher-risk patients, such as those with leptomeningeal disease or with low performance status, which investigators excluded from the present study.
“There are good data showing that patients with cerebral metastases can be stratified into groups that have very different survival and morbidity,” Dr. Turajlic and Dr. Larkin wrote. “Caution is necessary until we have data across all the groups.”
These comment are based on an editorial in the New England Journal of Medicine (doi: 10.1056/NEJMe1807752) . Dr. Turajlic reported patents pending for an indel biomarker (PCT/GB2018/051893) and an indel therapeutic (PCT/GB2018/051892). Dr. Larkin reported disclosures related to Bristol-Myers Squibb, Novartis, Genentech, Pierre-Fabre, Incyte, and AstraZeneca.
These data show that checkpoint inhibitors can be similarly effective in CNS metastases as they can be in extracranial metastases related to melanoma, according to Samra Turajlic, MD, PhD, and James Larkin, FRCP, PhD, of the Renal and Skin Units at the Royal Marsden National Health Service Foundation Trust in London.
Based on the study results, larger trials are warranted, including patients with CNS metastases from melanoma, kidney, lung, and other cancers where checkpoint inhibitors have demonstrated efficacy, Dr. Turajlic, who is also with the Translational Cancer Therapeutics Laboratory at the Francis Crick Institute in London, and Dr. Larkin wrote in an editorial.
“Such patients should no longer generally be excluded from clinical trials,” they wrote.
While the study by Dr. Tawbi and his colleagues was small, they added, its results are relevant to clinical practice because of the high rate of response, rapid response time, and side effect profile, which was manageable.
In fact, the nivolumab plus ipilimumab regimen described in this study should be considered first-line therapy for all patients who meet the study’s inclusion criteria, they asserted.
However, the results should “absolutely not” be extrapolated to higher-risk patients, such as those with leptomeningeal disease or with low performance status, which investigators excluded from the present study.
“There are good data showing that patients with cerebral metastases can be stratified into groups that have very different survival and morbidity,” Dr. Turajlic and Dr. Larkin wrote. “Caution is necessary until we have data across all the groups.”
These comment are based on an editorial in the New England Journal of Medicine (doi: 10.1056/NEJMe1807752) . Dr. Turajlic reported patents pending for an indel biomarker (PCT/GB2018/051893) and an indel therapeutic (PCT/GB2018/051892). Dr. Larkin reported disclosures related to Bristol-Myers Squibb, Novartis, Genentech, Pierre-Fabre, Incyte, and AstraZeneca.
These data show that checkpoint inhibitors can be similarly effective in CNS metastases as they can be in extracranial metastases related to melanoma, according to Samra Turajlic, MD, PhD, and James Larkin, FRCP, PhD, of the Renal and Skin Units at the Royal Marsden National Health Service Foundation Trust in London.
Based on the study results, larger trials are warranted, including patients with CNS metastases from melanoma, kidney, lung, and other cancers where checkpoint inhibitors have demonstrated efficacy, Dr. Turajlic, who is also with the Translational Cancer Therapeutics Laboratory at the Francis Crick Institute in London, and Dr. Larkin wrote in an editorial.
“Such patients should no longer generally be excluded from clinical trials,” they wrote.
While the study by Dr. Tawbi and his colleagues was small, they added, its results are relevant to clinical practice because of the high rate of response, rapid response time, and side effect profile, which was manageable.
In fact, the nivolumab plus ipilimumab regimen described in this study should be considered first-line therapy for all patients who meet the study’s inclusion criteria, they asserted.
However, the results should “absolutely not” be extrapolated to higher-risk patients, such as those with leptomeningeal disease or with low performance status, which investigators excluded from the present study.
“There are good data showing that patients with cerebral metastases can be stratified into groups that have very different survival and morbidity,” Dr. Turajlic and Dr. Larkin wrote. “Caution is necessary until we have data across all the groups.”
These comment are based on an editorial in the New England Journal of Medicine (doi: 10.1056/NEJMe1807752) . Dr. Turajlic reported patents pending for an indel biomarker (PCT/GB2018/051893) and an indel therapeutic (PCT/GB2018/051892). Dr. Larkin reported disclosures related to Bristol-Myers Squibb, Novartis, Genentech, Pierre-Fabre, Incyte, and AstraZeneca.
Treatment with nivolumab plus ipilimumab resulted in clinically meaningful efficacy for melanoma patients with asymptomatic, previously untreated brain metastases, results of an open-label, multicenter, phase 2 study have shown.
The combination of these two immune checkpoint inhibitors produced intracranial responses in more than half of the patients treated, and perhaps more importantly, according to the study investigators, the combination treatment prevented intracranial progression for more than 6 months in 64% of the study population.
“These results are relevant in a population in whom progression can quickly result in substantial neurologic symptoms, functional impairment, and the need for glucocorticoid therapy,” the study investigators wrote in the New England Journal of Medicine.
The investigators, led by Hussein A. Tawbi, MD, PhD, of the University of Texas MD Anderson Cancer, Houston, initially enrolled 101 patients with histologically confirmed melanoma and metastases to the brain that were asymptomatic. All patients had an Eastern Cooperative Oncology Group performance status of 0-1 and had not received systemic glucocorticoid therapy within 10 days of study treatment.
The primary endpoint of the study was the rate of intracranial benefit, defined as the percentage of patients with complete response, partial response, or stable disease for at least 6 months after starting treatment.
For 94 patients with at least 6 months of follow-up at the time of analysis (median follow-up, 14 months), the rate of intracranial benefit was 57%, including complete responses in 26%, partial responses in 30%, and stable disease in 2%, the investigators reported. The rate of extracranial benefit was similar, at 56%.
The 6-month rate of progression-free survival was 64.2% for intracranial assessments, while the 6-month overall survival rate was 92.3%, according to results of an initial assessment.
Grade 3 or 4 adverse events thought to be related to treatment occurred in 55% of patients and led to treatment discontinuation in 20%; the most common were increased levels of ALT and AST.
Dr. Tawbi and his colleagues said that, while cross-trial comparisons have inherent limitations, the rate of intracranial response seen in this trial is similar to what was seen in the COMBI-MB study of dabrafenib plus trametinib in patients with BRAF-mutant melanoma and brain metastases. However, in that study, published in 2017 in the Lancet, the combination of a BRAF inhibitor and MEK inhibitor had rates of intracranial response and progression-free survival that were “substantially shorter” than the rates of extracranial response and progression-free survival.
“In our study, the use of immunotherapy seemed capable of inducing intracranial responses that were very similar to extracranial responses in character, depth, and duration,” they wrote.
Dr. Tawbi and his coinvestigators enrolled an additional 20 symptomatic patients with brain metastases following a study protocol amendment; however, results from that cohort are not being reported yet because of inadequate follow-up length, they said.
The study was supported by Bristol-Myers Squibb and a grant from the National Cancer Institute. Dr. Tawbi reported disclosures related to Bristol-Myers Squibb, Merck, Genentech, and Novartis. His coauthors reported additional disclosures related to MedImmune, AstraZeneca, Dynavax Technologies, Genoptix, Exelixis, Acceleron Pharma, and Eisai, among others.
SOURCE: Tawbi HA et al. N Engl J Med. 2018 Aug 23. doi: 10.1056/NEJMoa1805453.
Treatment with nivolumab plus ipilimumab resulted in clinically meaningful efficacy for melanoma patients with asymptomatic, previously untreated brain metastases, results of an open-label, multicenter, phase 2 study have shown.
The combination of these two immune checkpoint inhibitors produced intracranial responses in more than half of the patients treated, and perhaps more importantly, according to the study investigators, the combination treatment prevented intracranial progression for more than 6 months in 64% of the study population.
“These results are relevant in a population in whom progression can quickly result in substantial neurologic symptoms, functional impairment, and the need for glucocorticoid therapy,” the study investigators wrote in the New England Journal of Medicine.
The investigators, led by Hussein A. Tawbi, MD, PhD, of the University of Texas MD Anderson Cancer, Houston, initially enrolled 101 patients with histologically confirmed melanoma and metastases to the brain that were asymptomatic. All patients had an Eastern Cooperative Oncology Group performance status of 0-1 and had not received systemic glucocorticoid therapy within 10 days of study treatment.
The primary endpoint of the study was the rate of intracranial benefit, defined as the percentage of patients with complete response, partial response, or stable disease for at least 6 months after starting treatment.
For 94 patients with at least 6 months of follow-up at the time of analysis (median follow-up, 14 months), the rate of intracranial benefit was 57%, including complete responses in 26%, partial responses in 30%, and stable disease in 2%, the investigators reported. The rate of extracranial benefit was similar, at 56%.
The 6-month rate of progression-free survival was 64.2% for intracranial assessments, while the 6-month overall survival rate was 92.3%, according to results of an initial assessment.
Grade 3 or 4 adverse events thought to be related to treatment occurred in 55% of patients and led to treatment discontinuation in 20%; the most common were increased levels of ALT and AST.
Dr. Tawbi and his colleagues said that, while cross-trial comparisons have inherent limitations, the rate of intracranial response seen in this trial is similar to what was seen in the COMBI-MB study of dabrafenib plus trametinib in patients with BRAF-mutant melanoma and brain metastases. However, in that study, published in 2017 in the Lancet, the combination of a BRAF inhibitor and MEK inhibitor had rates of intracranial response and progression-free survival that were “substantially shorter” than the rates of extracranial response and progression-free survival.
“In our study, the use of immunotherapy seemed capable of inducing intracranial responses that were very similar to extracranial responses in character, depth, and duration,” they wrote.
Dr. Tawbi and his coinvestigators enrolled an additional 20 symptomatic patients with brain metastases following a study protocol amendment; however, results from that cohort are not being reported yet because of inadequate follow-up length, they said.
The study was supported by Bristol-Myers Squibb and a grant from the National Cancer Institute. Dr. Tawbi reported disclosures related to Bristol-Myers Squibb, Merck, Genentech, and Novartis. His coauthors reported additional disclosures related to MedImmune, AstraZeneca, Dynavax Technologies, Genoptix, Exelixis, Acceleron Pharma, and Eisai, among others.
SOURCE: Tawbi HA et al. N Engl J Med. 2018 Aug 23. doi: 10.1056/NEJMoa1805453.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Nivolumab plus ipilimumab resulted in clinically meaningful responses and progression-free survival for melanoma patients with asymptomatic, previously untreated brain metastases.
Major finding: The reported rate of intracranial benefit was 57% of patients, including complete responses in 26%, partial responses in 30%, and stable disease for at least 6 months in 2%.
Study details: An open-label, multicenter, phase 2 study initially enrolling 101 patients with histologically confirmed melanoma and metastases to the brain.
Disclosures: The study was supported by Bristol-Myers Squibb and a grant from the National Cancer Institute. The study authors reported disclosures related to Bristol-Myers Squibb, Merck, Genentech, Novartis, MedImmune, AstraZeneca, Dynavax Technologies, Genoptix, Exelixis, Acceleron Pharma, Eisai, and others.
Source: Tawbi HA et al. N Engl J Med. 2018 Aug 23. doi: 10.1056/NEJMoa1805453.
Adding checkpoint inhibitors to radiotherapy requires particular caution in this one scenario
Among scenarios where immune checkpoint inhibitors (ICIs) might be combined, particular caution is needed in the setting of brain metastases, according to authors of a recent clinical review.
While evidence to date is mixed, some studies do suggest that adding ICIs to high-dose stereotactic intracranial radiotherapy for brain metastases might increase the risk of treatment-related brain necrosis, the authors said.
By contrast, the balance of evidence suggests ICIs can be safely combined with palliative radiotherapy without site-specific increases in adverse events, they added.
Likewise, in patients with non–small-cell lung cancer, ICIs do not appear to increase incidence of grade 3 or greater pneumonitis when given after definitive chemoradiotherapy, in both retrospective and prospective investigations.
Nevertheless, the addition of ICIs to radiotherapy requires careful further study because of the potential for increased type or severity of toxicities, including the immune-related adverse events associated with ICIs, wrote corresponding author Jay S. Loeffler, MD, of Massachusetts General Hospital, Boston, and his colleagues.
“Caution is warranted when combining radiotherapy and ICI, especially with intracranial radiotherapy,” the researchers wrote. Their report is in Nature Reviews Clinical Oncology.
Some studies have indicated a higher rate of treatment-associated brain necrosis when ICIs are combined with intracranial radiotherapy, while others have shown no such trend, the authors said.
In one single-institution experience involving 180 patients with brain metastases undergoing stereotactic radiotherapy, incidence of treatment-associated brain necrosis was significantly higher in patients receiving an ICI, with an odds ratio of 2.4 (95% confidence interval, 1.06-5.44; P = .03).
Similarly, a retrospective single institution 480-patient study showed an incidence of treatment-associated brain necrosis of 20% for ICIs plus stereotactic radiotherapy versus 7% for radiotherapy alone (P less than .001), but substantial differences in baseline characteristics between groups limited the strength of the study’s conclusions, according to the researchers.
Increased risk is primarily in the form of asymptomatic or minimally symptomatic episodes in some series, the authors noted. A retrospective, 54-patient report showed a rate of treatment-associated brain necrosis of 30% when ICIs were combined with stereotactic radiotherapy, versus 21% for radiotherapy alone (P = .08), but the incidence of symptomatic cases was 15% in both groups, they noted.
“Intriguingly, the findings of several studies have demonstrated an association between [treatment-associated brain necrosis] and improved survival outcomes in patients with melanoma brain metastases that is similar to the independent observations of an analogous relationship between risk of [immune-related adverse events] in general and responsiveness to ICI,” the researchers wrote.
Most of the Food and Drug Administration–approved indications for ICIs are in the metastatic setting, where palliative radiotherapy is frequently important, the authors noted.
In two retrospective studies of patients with metastatic cancers receiving palliative radiotherapy with ICIs, there was a lack of clear association between the irradiated site and specific immune-related adverse events; that lack of association suggests that any toxicities arising from interactions between palliative radiotherapy and ICIs are mainly systemic, rather than local, the authors wrote.
Several retrospective series in advanced-stage melanoma patients have suggested that palliative radiotherapy plus ICIs is safe and does not significantly increase incidence of immune-related adverse events. However, findings from one series showed a correlation between both the ICI and radiotherapy dose given and the incidence of immune-related adverse events.
Prospective studies will be essential to optimize the balance between disease control and risk of morbidity associated with ICIs and radiotherapy combinations, the authors concluded.
The researchers declared no competing interests related to their review article.
SOURCE: Hwang WL, et al. Nat Rev Clin Oncol. 2018 Aug;15(8):477-494.
Among scenarios where immune checkpoint inhibitors (ICIs) might be combined, particular caution is needed in the setting of brain metastases, according to authors of a recent clinical review.
While evidence to date is mixed, some studies do suggest that adding ICIs to high-dose stereotactic intracranial radiotherapy for brain metastases might increase the risk of treatment-related brain necrosis, the authors said.
By contrast, the balance of evidence suggests ICIs can be safely combined with palliative radiotherapy without site-specific increases in adverse events, they added.
Likewise, in patients with non–small-cell lung cancer, ICIs do not appear to increase incidence of grade 3 or greater pneumonitis when given after definitive chemoradiotherapy, in both retrospective and prospective investigations.
Nevertheless, the addition of ICIs to radiotherapy requires careful further study because of the potential for increased type or severity of toxicities, including the immune-related adverse events associated with ICIs, wrote corresponding author Jay S. Loeffler, MD, of Massachusetts General Hospital, Boston, and his colleagues.
“Caution is warranted when combining radiotherapy and ICI, especially with intracranial radiotherapy,” the researchers wrote. Their report is in Nature Reviews Clinical Oncology.
Some studies have indicated a higher rate of treatment-associated brain necrosis when ICIs are combined with intracranial radiotherapy, while others have shown no such trend, the authors said.
In one single-institution experience involving 180 patients with brain metastases undergoing stereotactic radiotherapy, incidence of treatment-associated brain necrosis was significantly higher in patients receiving an ICI, with an odds ratio of 2.4 (95% confidence interval, 1.06-5.44; P = .03).
Similarly, a retrospective single institution 480-patient study showed an incidence of treatment-associated brain necrosis of 20% for ICIs plus stereotactic radiotherapy versus 7% for radiotherapy alone (P less than .001), but substantial differences in baseline characteristics between groups limited the strength of the study’s conclusions, according to the researchers.
Increased risk is primarily in the form of asymptomatic or minimally symptomatic episodes in some series, the authors noted. A retrospective, 54-patient report showed a rate of treatment-associated brain necrosis of 30% when ICIs were combined with stereotactic radiotherapy, versus 21% for radiotherapy alone (P = .08), but the incidence of symptomatic cases was 15% in both groups, they noted.
“Intriguingly, the findings of several studies have demonstrated an association between [treatment-associated brain necrosis] and improved survival outcomes in patients with melanoma brain metastases that is similar to the independent observations of an analogous relationship between risk of [immune-related adverse events] in general and responsiveness to ICI,” the researchers wrote.
Most of the Food and Drug Administration–approved indications for ICIs are in the metastatic setting, where palliative radiotherapy is frequently important, the authors noted.
In two retrospective studies of patients with metastatic cancers receiving palliative radiotherapy with ICIs, there was a lack of clear association between the irradiated site and specific immune-related adverse events; that lack of association suggests that any toxicities arising from interactions between palliative radiotherapy and ICIs are mainly systemic, rather than local, the authors wrote.
Several retrospective series in advanced-stage melanoma patients have suggested that palliative radiotherapy plus ICIs is safe and does not significantly increase incidence of immune-related adverse events. However, findings from one series showed a correlation between both the ICI and radiotherapy dose given and the incidence of immune-related adverse events.
Prospective studies will be essential to optimize the balance between disease control and risk of morbidity associated with ICIs and radiotherapy combinations, the authors concluded.
The researchers declared no competing interests related to their review article.
SOURCE: Hwang WL, et al. Nat Rev Clin Oncol. 2018 Aug;15(8):477-494.
Among scenarios where immune checkpoint inhibitors (ICIs) might be combined, particular caution is needed in the setting of brain metastases, according to authors of a recent clinical review.
While evidence to date is mixed, some studies do suggest that adding ICIs to high-dose stereotactic intracranial radiotherapy for brain metastases might increase the risk of treatment-related brain necrosis, the authors said.
By contrast, the balance of evidence suggests ICIs can be safely combined with palliative radiotherapy without site-specific increases in adverse events, they added.
Likewise, in patients with non–small-cell lung cancer, ICIs do not appear to increase incidence of grade 3 or greater pneumonitis when given after definitive chemoradiotherapy, in both retrospective and prospective investigations.
Nevertheless, the addition of ICIs to radiotherapy requires careful further study because of the potential for increased type or severity of toxicities, including the immune-related adverse events associated with ICIs, wrote corresponding author Jay S. Loeffler, MD, of Massachusetts General Hospital, Boston, and his colleagues.
“Caution is warranted when combining radiotherapy and ICI, especially with intracranial radiotherapy,” the researchers wrote. Their report is in Nature Reviews Clinical Oncology.
Some studies have indicated a higher rate of treatment-associated brain necrosis when ICIs are combined with intracranial radiotherapy, while others have shown no such trend, the authors said.
In one single-institution experience involving 180 patients with brain metastases undergoing stereotactic radiotherapy, incidence of treatment-associated brain necrosis was significantly higher in patients receiving an ICI, with an odds ratio of 2.4 (95% confidence interval, 1.06-5.44; P = .03).
Similarly, a retrospective single institution 480-patient study showed an incidence of treatment-associated brain necrosis of 20% for ICIs plus stereotactic radiotherapy versus 7% for radiotherapy alone (P less than .001), but substantial differences in baseline characteristics between groups limited the strength of the study’s conclusions, according to the researchers.
Increased risk is primarily in the form of asymptomatic or minimally symptomatic episodes in some series, the authors noted. A retrospective, 54-patient report showed a rate of treatment-associated brain necrosis of 30% when ICIs were combined with stereotactic radiotherapy, versus 21% for radiotherapy alone (P = .08), but the incidence of symptomatic cases was 15% in both groups, they noted.
“Intriguingly, the findings of several studies have demonstrated an association between [treatment-associated brain necrosis] and improved survival outcomes in patients with melanoma brain metastases that is similar to the independent observations of an analogous relationship between risk of [immune-related adverse events] in general and responsiveness to ICI,” the researchers wrote.
Most of the Food and Drug Administration–approved indications for ICIs are in the metastatic setting, where palliative radiotherapy is frequently important, the authors noted.
In two retrospective studies of patients with metastatic cancers receiving palliative radiotherapy with ICIs, there was a lack of clear association between the irradiated site and specific immune-related adverse events; that lack of association suggests that any toxicities arising from interactions between palliative radiotherapy and ICIs are mainly systemic, rather than local, the authors wrote.
Several retrospective series in advanced-stage melanoma patients have suggested that palliative radiotherapy plus ICIs is safe and does not significantly increase incidence of immune-related adverse events. However, findings from one series showed a correlation between both the ICI and radiotherapy dose given and the incidence of immune-related adverse events.
Prospective studies will be essential to optimize the balance between disease control and risk of morbidity associated with ICIs and radiotherapy combinations, the authors concluded.
The researchers declared no competing interests related to their review article.
SOURCE: Hwang WL, et al. Nat Rev Clin Oncol. 2018 Aug;15(8):477-494.
FROM NATURE REVIEWS CLINICAL ONCOLOGY
Key clinical point: Some studies suggest that adding ICIs to high-dose stereotactic intracranial radiotherapy for brain metastases might increase the risk of treatment-related brain necrosis.
Major finding: The balance of evidence suggests ICIs can be safely combined with palliative radiotherapy.
Study details: A literature review.
Disclosures: The researchers declared no competing interests related to their review article.
Source: Hwang WL et al. Nat Rev Clin Oncol. 2018 Aug;15(8):477-94.
Meeting the potential of immunotherapy: new targets provide rational combinations
The relationship between the immune system and tumors is complex and dynamic, and for immunotherapy to reach its full potential it will likely need to attack on multiple fronts. Here, we discuss some of the latest and most promising developments in the immuno-oncology field designed to build on the successes and address limitations.
The anti-tumor immune response
Cancer is a disease of genomic instability, whereby genetic alterations ranging from a single nucleotide to the whole chromosome level frequently occur. Although cancers derive from a patient’s own tissues, these genetic differences can mark the cancer cell as non-self, triggering an immune response to eliminate these cells.
The first hints of this anti-tumor immunity date back more than a century and a half and sparked the concept of mobilizing the immune system to treat patients.1-3 Although early pioneers achieved little progress in this regard, their efforts provided invaluable insights into the complex and dynamic relationship between a tumor and the immune system that are now translating into real clinical successes.
We now understand that the immune system has a dual role in both restraining and promoting cancer development and have translated this understanding into the theory of cancer immunoediting. Immunoediting has three stages: elimination, wherein the tumor is seemingly destroyed by the innate and adaptive immune response; equilibrium, in which cancer cells that were able to escape elimination are selected for growth; and escape, whereby these resistant cancer cells overwhelm the immune system and develop into a symptomatic lesion.4,5
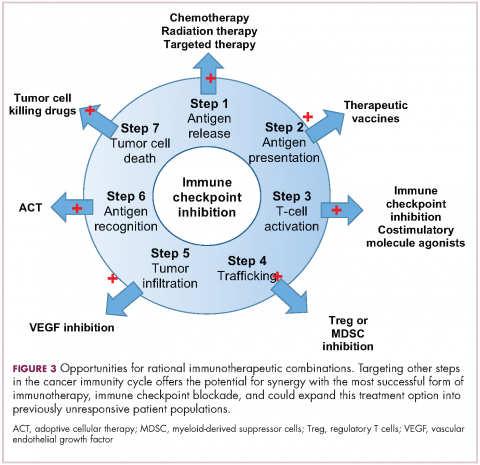
Immuno-oncologists have also described the cancer immunity cycle to capture the steps that are required for an effective anti-tumor immune response and defects in this cycle form the basis of the most common mechanisms used by cancer cells to subvert the anti-tumor immune response. Much like the cancer hallmarks did for molecularly targeted cancer drugs, the cancer immunity cycle serves as the intellectual framework for cancer immunotherapy.6,7
Exploiting nature’s weapon of mass destruction
Initially, attempts at immunotherapy focused on boosting the immune response using adjuvants and cytokines. The characterization of subtle differences between tumor cells and normal cells led to the development of vaccines and cell-based therapies that exploited these tumor-associated antigens (TAAs).1-6
Despite the approval of a therapeutic vaccine, sipuleucel-T, in 2010 for the treatment of metastatic prostate cancer, in general the success of vaccines has been limited. Marketing authorization for sipuleucel-T was recently withdrawn in Europe, and although it is still available in the United States, it is not widely used because of issues with production and administration. Other vaccines, such as GVAX, which looked particularly promising in early-stage clinical trials, failed to show clinical efficacy in subsequent testing.8,9
Cell-based therapies, such as adoptive cellular therapy (ACT), in which immune cells are removed from the host, primed to attack cancer cells, and then reinfused back into the patient, have focused on T cells because they are the major effectors of the adaptive immune response. Clinical success with the most common approach, tumor-infiltrating lymphocyte (TIL)
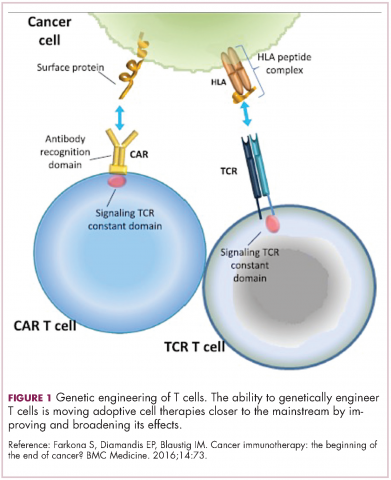
Two key techniques have been developed (Figure 1). T-cell receptor (TCR) therapy involves genetically modifying the receptor on the surface of T cells that is responsible for recognizing antigens bound to major histocompatibility complex (MHC) molecules on the surface of antigen-presenting cells (APCs). The TCR can be altered to recognize a specific TAA or modified to improve its antigen recognition and binding capabilities. This type of therapy is limited by the fact that the TCRs need to be genetically matched to the patient’s immune type.
Releasing the brakes
To ensure that it is only activated at the appropriate time and not in response to the antigens expressed on the surface of the host’s own tissues or harmless materials, the immune system has developed numerous mechanisms for immunological tolerance. Cancer cells are able to exploit these mechanisms to allow them to evade the anti-tumor immune response. One of the main ways in which they do this is by manipulating the signaling pathways involved in T-cell activation, which play a vital role in tolerance.12
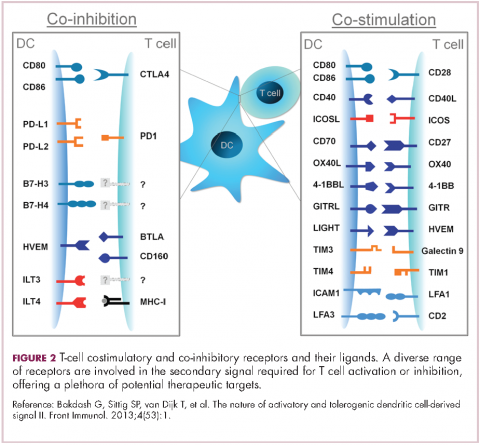
To become fully activated, T cells require a primary signal generated by an interaction between the TCR and the antigen-MHC complex on the surface of an APC, followed by secondary costimulatory signals generated by a range of different receptors present on the T-cell surface binding to their ligands on the APC.
If the second signal is inhibitory rather than stimulatory, then the T cell is deactivated instead of becoming activated. Two key coinhibitory receptors are programmed cell death 1 (PD-1) and cytotoxic T-lymphocyte antigen 4 (CTLA-4) and tumor cells are able to overcome the anti-tumor immune response in part by expressing the ligands that bind these receptors to dampen the activity of tumor-infiltrating T cells and induce tolerance.13
The development of inhibitors of CTLA-4 and PD-1 and their respective ligands has driven some of the most dramatic successes with cancer immunotherapy, particularly with PD-1-targeting drugs which have fewer side effects. Targeting of this pathway has resulted in durable responses, revolutionizing the treatment of metastatic melanoma, with recently published long-term survival data for pembrolizumab showing that 40% of patients were alive 3 years after initiating treatment and, in a separate study, 34% of nivolumab-treated patients were still alive after 5 years.14,15 More recently, PD-1 inhibitors have been slowly expanding into a range of other cancer types and 4 immune checkpoint inhibitors are now approved by the United States Food and Drug Administration (FDA): ipilimumab (Yervoy), nivolumab (Opdivo), pembrolizumab (Keytruda) and atezolizumab (Tecentriq).
Six years on from the first approval in this drug class and an extensive network of coinhibitory receptors has been uncovered – so-called immune checkpoints – many of which are now also serving as therapeutic targets (Table, Figure 2).16 Lymphocyte activation gene 3 (LAG-3) is a member of the immunoglobulin superfamily of receptors that is expressed on a number of different types of immune cell. In addition to negatively regulating cytotoxic T-cell activation like PD-1 and CTLA-4, it is also thought to regulate the immunosuppressive functions of regulatory T cells and the maturation and activation of dendritic cells. T-cell immunoglobulin and mucin domain-containing 3 (TIM-3) is found on the surface of helper and cytotoxic T cells and regulates T-cell inhibition as well as macrophage activation. Inhibitors of both proteins have been developed that are being evaluated in phase 1 or 2 clinical trials in a variety of tumor types.17
Indeed, although T cells have commanded the most attention, there is growing appreciation of the potential for targeting other types of immune cell that play a role in the anti-tumor immune response or in fostering an immunosuppressive microenvironment. NK cells have been a particular focus, since they represent the body’s first line of immune defense and they appear to have analogous inhibitory and activating receptors expressed on their surface that regulate their cytotoxic activity.
The best-defined NK cell receptors are the killer cell immunoglobulin-like receptors (KIRs) that bind to the MHC class I proteins found on the surface of all cells that distinguish them as ‘self’ or ‘non-self’. KIRs can be either activating or inhibitory, depending upon their structure and the ligands to which they bind.19 To date, 2 antibodies targeting inhibitory KIRs have been developed. Though there has been some disappointment with these drugs, most recently a phase 2 trial of lirilumab in elderly patients with acute myeloid leukemia, which missed its primary endpoint, they continue to be evaluated in clinical trials.20
The inhibitory immune checkpoint field has also expanded to include molecules that regulate T-cell activity in other ways. Most prominently, this includes enzymes like indoleamine-2,3 dioxygenase (IDO), which is involved in the metabolism of the essential amino acid tryptophan. IDO-induced depletion of tryptophan and generation of tryptophan metabolites is toxic to cytotoxic T cells, and IDO is also thought to directly activate regulatory T cells, thus the net effect of IDO is immunosuppression. Two IDO inhibitors are currently being developed.21
Stepping on the gas
Despite their unprecedented success, immune checkpoint inhibitors are not effective in all patients or in all tumor types. Their efficacy is limited in large part by the requirement for a pre-existing anti-tumor immune response. If there are no T cells within the tumor microenvironment then releasing the brakes on the immune system won’t help.
More recently, researchers have returned to the idea of stimulating an anti-tumor immune response, this time by targeting the other side of the immune checkpoint coin, the costimulatory molecules. These drugs could prove more effective as they aren’t reliant on a pre-existing anti-tumor immune response. A number of agonist antibodies designed to target these receptors have now been developed and are undergoing clinical evaluation.22
Furthest along in development are those targeting OX40, a costimulatory molecule that is upregulated on the surface of T cells once they have been fully activated by the TCR signal and an initial costimulatory signal. OX40 is thought to be involved in a more long-term immune response and in the formation of a memory response. A mouse monoclonal antibody had a potent immune-stimulating effect accompanied by the regression of at least 1 metastatic lesion in 30% of patients treated in a phase 1 clinical trial, but was limited by the generation of anti-mouse antibodies. 7 OX40 agonists are now in clinical development, 6 fully human monoclonal antibodies and 1 OX40 ligand-Fc fusion protein, MEDI-6383.23
Combinations are key
Many researchers are now reaching the conclusion that combination therapy is likely to be key in expanding the scope of immunotherapy into currently unresponsive patient populations. Investigating rational combinations is already becoming a burgeoning area of the immuno-oncology field, with a variety of different strategies being tested.
Now the question becomes what are the optimal combinations and the timing and sequencing of combination therapy is likely to be a paramount consideration. Developing combinations that have distinct mechanisms of action or target multiple steps in the cancer immunity cycle offers the greatest potential for therapeutic synergy since this is most likely to address potential mechanisms of resistance by blocking other paths to immune evasion for cancer cells (Figure 3).
Given the expanding network of immune-checkpoint inhibitors and agonists, the focal point of combination therapy has been combining immune checkpoint-targeting drugs with different mechanisms of action, including those that would simultaneously release the brakes and step on the gas pedal. The vast majority of ongoing clinical trials of approved checkpoint inhibitors and the drugs in development listed in the table are combination trials.
These efforts yielded the first FDA-approved combination immunotherapy regimen in 2015; nivolumab and ipilimumab for the treatment of metastatic melanoma. Approval was based on the demonstration of improved ORR, prolonged response duration, and improved progression-free survival among 142 patients treated with the combination, compared to either drug alone.24
The results of a phase 1/2 trial evaluating the combination of a 4-1BB receptor agonist urelumab with nivolumab in hematologic malignancies and solid tumors found the combination to be safe and particularly effective in patients with advanced/metastatic melanoma, with an ORR of 50%.25 Nivolumab was also combined with the CD27 agonist varlilumab in a phase 1/2 clinical trial of patients with solid tumors, for which data was also recently released. Among 46 patients enrolled, primarily those with colorectal and ovarian cancer the combination had an acceptable safety profile and favorable changes in intratumoral immune biomarkers were observed. The phase 2 portion of the trial is ongoing.26
Meanwhile, Incyte’s IDO inhibitor epacadostat has recently been making waves in combination with pembrolizumab in patients with advanced solid tumors. It demonstrated particularly promising clinical activity in patients with metastatic melanoma, with an overall response rate (ORR) of 57%, including 2 complete responses (CRs), prompting initiation of a phase 3 trial of this combination (NCT02752074).27
1. Adams JL, Smothers J, Srinivasan R, et al. Big opportunities for small molecules in immuno-oncology. Nat Rev Drug Disc. 2015;14:603-622.
2. D’Errico G, Machado HL, Sainz Jr B. A current perspective on cancer immune therapy: step-by-step approach to constructing the magic bullet. Clin Trans Med. 2017;6:3.
3. Farkona S, Diamandis EP, Blaustig IM. Cancer immunotherapy: the beginning of the end of cancer? BMC Med. 2016;14:73.
4. Meiliana A, Dewi NM, Wijaya A. Cancer immunotherapy: a review. Indones Biomed J. 2016;8(1):1-20.
5. Smyth MJ, Ngiow SF, Ribas A, et al. Combination cancer immunotherapies tailored to the tumor microenvironment. Nat Rev Clin Oncol. 2016;13:143-158.
6. de Charette M, Marabelle A, Houot R. Turning tumor cells into antigen presenting cells: The next step to improve cancer immunotherapy? Eur J Cancer 2016;68:134-147.
7. Chen DS and Mellman I. Oncology Meets Immunology: The Cancer-Immunity Cycle. Immunity 2013;39:1-10.
8. Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature 2011;480:480-489.
9. Le DT, Wang-Gillam A, Picozzi V Jr, et al. A phase 2, randomized trial of GVAX Pancreas and CRS-207 immunotherapy versus GVAX alone in patients with metastatic pancreatic adenocarcinoma: Updated results. Presented at: the ASCO Gastrointestinal Cancers Symposium; January 16-18, 2014; San Francisco, CA. Abstract 177.
10. Sharpe M and Mount N. Genetically modified T cells in cancer therapy: opportunities and challenges. Dis Model Mech. 2015;8(4):337-350.
11. Perica K, Varela JC, Oelke M, et al. Adoptive T Cell Immunotherapy for Cancer. Ram Mai Med J. 2015;6(1):e0004.
12. Xing Y and Hogquist KA. T-Cell Tolerance: Central and Peripheral. Cold Spring Harb Perspect Biol. 2012;4:a006957.
13. Buchbinder EI and Desai A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016;39(1):98-106.
14. Robert C, Ribas A, Hamid O, et al. 3-year overall survival for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. J Clin Oncol. 2016(suppl;abstr 9503).
15. Hodi SF, Kluger HM, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Presented at the 2016 AACR Annual Meeting; April 16-20; New Orleans, LA. Abstract CT001.
16. Bakdash G, Sittig SP, van Dijk T, et al. The nature of activatory and tolerogenic dendritic cell-derived signal II. Front Immunol. 2013;4(53):1-18.
17. Sheridan C. Immuno-oncology moves beyond PD-1. Nat Biotechnol. 2015;33(7):673-675.
18. Blake SJ, Dougall WC, Miles JJ, et al. Molecular pathways: targeting CD96 and TIGIT for cancer immunotherapy. Clin Cancer Res. 2016;22(21):5183-5188.
19. Carotta S. Targeting NK cells for anticancer immunotherapy: clinical and preclinical approaches. Front Immunol. 2016;7:152.
20. Innate Pharma Web site. Innate Pharma Announces Top-Line Results from EFFIKIR Trial Evaluating the Efficacy of Lirilumab as a Single Agent in Elderly Patients with Acute Myeloid Leukemia. http://www.innate-pharma.com/en/news-events/press-releases/innate-pharma-announces-top-line-results-effikir-trial-evaluating-efficacy-lirilumab-single-agent-elderly-patients-acute-myeloid-leukemia. Last updated February 6, 2017. Accessed online February 22, 2017.
21. Sheridan C. IDO inhibitors move center stage in immuno-oncology. Nat Biotechnol. 2015;33(4):321-322.
22. Sanmamed MF, Pastor F, Rodriguez A, et al. Agonists of co-stimulation in cancer immunotherapy directed against CD137, OX40, GITR, CD27, CD28, and ICOS. Semin Oncol. 2015;42(4):640-655.
23. Linch SN, McNamara MJ, Redmond WL. OX40 agonists and combination immunotherapy: putting the pedal to the metal. Front Oncol. 2015;5:34.
24. U.S. Food and Drug Administration Web site. Nivolumab in combination with ipilimumab. https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm465274.htm. Last updated October 1, 2015. Accessed online February 22, 2017.
25. Massarelli E. Clinical safety and efficacy assessment of the CD137 agonist urelumab alone and in combination with nivolumab in patients with hematologic and solid tumor malignancies. Presented at the 31st Annual Meeting of the Society for the Immunotherapy of Cancer; November 9-13, 2016; National Harbor, MD. Abstract 239.
26. Sanborn RE, Pishvain MJ, Callahan MK, et al. Phase I results from the combination of an immune-activating anti-CD27 antibody (varlilumab) in combination with PD-1 blockade (nivolumab): activation across multiple immune pathways without untoward immune-related adverse events. Clin Cancer Res. 2016;76(14):suppl. Abstract CT023.
27. Gangadhar T, Hamid O, Smith D.C, et al. Epacadostat plus pembrolizumab in patients with advanced melanoma and select solid tumors: updated phase 1 results from ECHO-202/KEYNOTE-037. Ann Oncol. 2016;27(6):379-400.
The relationship between the immune system and tumors is complex and dynamic, and for immunotherapy to reach its full potential it will likely need to attack on multiple fronts. Here, we discuss some of the latest and most promising developments in the immuno-oncology field designed to build on the successes and address limitations.
The anti-tumor immune response
Cancer is a disease of genomic instability, whereby genetic alterations ranging from a single nucleotide to the whole chromosome level frequently occur. Although cancers derive from a patient’s own tissues, these genetic differences can mark the cancer cell as non-self, triggering an immune response to eliminate these cells.
The first hints of this anti-tumor immunity date back more than a century and a half and sparked the concept of mobilizing the immune system to treat patients.1-3 Although early pioneers achieved little progress in this regard, their efforts provided invaluable insights into the complex and dynamic relationship between a tumor and the immune system that are now translating into real clinical successes.
We now understand that the immune system has a dual role in both restraining and promoting cancer development and have translated this understanding into the theory of cancer immunoediting. Immunoediting has three stages: elimination, wherein the tumor is seemingly destroyed by the innate and adaptive immune response; equilibrium, in which cancer cells that were able to escape elimination are selected for growth; and escape, whereby these resistant cancer cells overwhelm the immune system and develop into a symptomatic lesion.4,5
Immuno-oncologists have also described the cancer immunity cycle to capture the steps that are required for an effective anti-tumor immune response and defects in this cycle form the basis of the most common mechanisms used by cancer cells to subvert the anti-tumor immune response. Much like the cancer hallmarks did for molecularly targeted cancer drugs, the cancer immunity cycle serves as the intellectual framework for cancer immunotherapy.6,7
Exploiting nature’s weapon of mass destruction
Initially, attempts at immunotherapy focused on boosting the immune response using adjuvants and cytokines. The characterization of subtle differences between tumor cells and normal cells led to the development of vaccines and cell-based therapies that exploited these tumor-associated antigens (TAAs).1-6
Despite the approval of a therapeutic vaccine, sipuleucel-T, in 2010 for the treatment of metastatic prostate cancer, in general the success of vaccines has been limited. Marketing authorization for sipuleucel-T was recently withdrawn in Europe, and although it is still available in the United States, it is not widely used because of issues with production and administration. Other vaccines, such as GVAX, which looked particularly promising in early-stage clinical trials, failed to show clinical efficacy in subsequent testing.8,9
Cell-based therapies, such as adoptive cellular therapy (ACT), in which immune cells are removed from the host, primed to attack cancer cells, and then reinfused back into the patient, have focused on T cells because they are the major effectors of the adaptive immune response. Clinical success with the most common approach, tumor-infiltrating lymphocyte (TIL)
Two key techniques have been developed (Figure 1). T-cell receptor (TCR) therapy involves genetically modifying the receptor on the surface of T cells that is responsible for recognizing antigens bound to major histocompatibility complex (MHC) molecules on the surface of antigen-presenting cells (APCs). The TCR can be altered to recognize a specific TAA or modified to improve its antigen recognition and binding capabilities. This type of therapy is limited by the fact that the TCRs need to be genetically matched to the patient’s immune type.
Releasing the brakes
To ensure that it is only activated at the appropriate time and not in response to the antigens expressed on the surface of the host’s own tissues or harmless materials, the immune system has developed numerous mechanisms for immunological tolerance. Cancer cells are able to exploit these mechanisms to allow them to evade the anti-tumor immune response. One of the main ways in which they do this is by manipulating the signaling pathways involved in T-cell activation, which play a vital role in tolerance.12
To become fully activated, T cells require a primary signal generated by an interaction between the TCR and the antigen-MHC complex on the surface of an APC, followed by secondary costimulatory signals generated by a range of different receptors present on the T-cell surface binding to their ligands on the APC.
If the second signal is inhibitory rather than stimulatory, then the T cell is deactivated instead of becoming activated. Two key coinhibitory receptors are programmed cell death 1 (PD-1) and cytotoxic T-lymphocyte antigen 4 (CTLA-4) and tumor cells are able to overcome the anti-tumor immune response in part by expressing the ligands that bind these receptors to dampen the activity of tumor-infiltrating T cells and induce tolerance.13
The development of inhibitors of CTLA-4 and PD-1 and their respective ligands has driven some of the most dramatic successes with cancer immunotherapy, particularly with PD-1-targeting drugs which have fewer side effects. Targeting of this pathway has resulted in durable responses, revolutionizing the treatment of metastatic melanoma, with recently published long-term survival data for pembrolizumab showing that 40% of patients were alive 3 years after initiating treatment and, in a separate study, 34% of nivolumab-treated patients were still alive after 5 years.14,15 More recently, PD-1 inhibitors have been slowly expanding into a range of other cancer types and 4 immune checkpoint inhibitors are now approved by the United States Food and Drug Administration (FDA): ipilimumab (Yervoy), nivolumab (Opdivo), pembrolizumab (Keytruda) and atezolizumab (Tecentriq).
Six years on from the first approval in this drug class and an extensive network of coinhibitory receptors has been uncovered – so-called immune checkpoints – many of which are now also serving as therapeutic targets (Table, Figure 2).16 Lymphocyte activation gene 3 (LAG-3) is a member of the immunoglobulin superfamily of receptors that is expressed on a number of different types of immune cell. In addition to negatively regulating cytotoxic T-cell activation like PD-1 and CTLA-4, it is also thought to regulate the immunosuppressive functions of regulatory T cells and the maturation and activation of dendritic cells. T-cell immunoglobulin and mucin domain-containing 3 (TIM-3) is found on the surface of helper and cytotoxic T cells and regulates T-cell inhibition as well as macrophage activation. Inhibitors of both proteins have been developed that are being evaluated in phase 1 or 2 clinical trials in a variety of tumor types.17
Indeed, although T cells have commanded the most attention, there is growing appreciation of the potential for targeting other types of immune cell that play a role in the anti-tumor immune response or in fostering an immunosuppressive microenvironment. NK cells have been a particular focus, since they represent the body’s first line of immune defense and they appear to have analogous inhibitory and activating receptors expressed on their surface that regulate their cytotoxic activity.
The best-defined NK cell receptors are the killer cell immunoglobulin-like receptors (KIRs) that bind to the MHC class I proteins found on the surface of all cells that distinguish them as ‘self’ or ‘non-self’. KIRs can be either activating or inhibitory, depending upon their structure and the ligands to which they bind.19 To date, 2 antibodies targeting inhibitory KIRs have been developed. Though there has been some disappointment with these drugs, most recently a phase 2 trial of lirilumab in elderly patients with acute myeloid leukemia, which missed its primary endpoint, they continue to be evaluated in clinical trials.20
The inhibitory immune checkpoint field has also expanded to include molecules that regulate T-cell activity in other ways. Most prominently, this includes enzymes like indoleamine-2,3 dioxygenase (IDO), which is involved in the metabolism of the essential amino acid tryptophan. IDO-induced depletion of tryptophan and generation of tryptophan metabolites is toxic to cytotoxic T cells, and IDO is also thought to directly activate regulatory T cells, thus the net effect of IDO is immunosuppression. Two IDO inhibitors are currently being developed.21
Stepping on the gas
Despite their unprecedented success, immune checkpoint inhibitors are not effective in all patients or in all tumor types. Their efficacy is limited in large part by the requirement for a pre-existing anti-tumor immune response. If there are no T cells within the tumor microenvironment then releasing the brakes on the immune system won’t help.
More recently, researchers have returned to the idea of stimulating an anti-tumor immune response, this time by targeting the other side of the immune checkpoint coin, the costimulatory molecules. These drugs could prove more effective as they aren’t reliant on a pre-existing anti-tumor immune response. A number of agonist antibodies designed to target these receptors have now been developed and are undergoing clinical evaluation.22
Furthest along in development are those targeting OX40, a costimulatory molecule that is upregulated on the surface of T cells once they have been fully activated by the TCR signal and an initial costimulatory signal. OX40 is thought to be involved in a more long-term immune response and in the formation of a memory response. A mouse monoclonal antibody had a potent immune-stimulating effect accompanied by the regression of at least 1 metastatic lesion in 30% of patients treated in a phase 1 clinical trial, but was limited by the generation of anti-mouse antibodies. 7 OX40 agonists are now in clinical development, 6 fully human monoclonal antibodies and 1 OX40 ligand-Fc fusion protein, MEDI-6383.23
Combinations are key
Many researchers are now reaching the conclusion that combination therapy is likely to be key in expanding the scope of immunotherapy into currently unresponsive patient populations. Investigating rational combinations is already becoming a burgeoning area of the immuno-oncology field, with a variety of different strategies being tested.
Now the question becomes what are the optimal combinations and the timing and sequencing of combination therapy is likely to be a paramount consideration. Developing combinations that have distinct mechanisms of action or target multiple steps in the cancer immunity cycle offers the greatest potential for therapeutic synergy since this is most likely to address potential mechanisms of resistance by blocking other paths to immune evasion for cancer cells (Figure 3).
Given the expanding network of immune-checkpoint inhibitors and agonists, the focal point of combination therapy has been combining immune checkpoint-targeting drugs with different mechanisms of action, including those that would simultaneously release the brakes and step on the gas pedal. The vast majority of ongoing clinical trials of approved checkpoint inhibitors and the drugs in development listed in the table are combination trials.
These efforts yielded the first FDA-approved combination immunotherapy regimen in 2015; nivolumab and ipilimumab for the treatment of metastatic melanoma. Approval was based on the demonstration of improved ORR, prolonged response duration, and improved progression-free survival among 142 patients treated with the combination, compared to either drug alone.24
The results of a phase 1/2 trial evaluating the combination of a 4-1BB receptor agonist urelumab with nivolumab in hematologic malignancies and solid tumors found the combination to be safe and particularly effective in patients with advanced/metastatic melanoma, with an ORR of 50%.25 Nivolumab was also combined with the CD27 agonist varlilumab in a phase 1/2 clinical trial of patients with solid tumors, for which data was also recently released. Among 46 patients enrolled, primarily those with colorectal and ovarian cancer the combination had an acceptable safety profile and favorable changes in intratumoral immune biomarkers were observed. The phase 2 portion of the trial is ongoing.26
Meanwhile, Incyte’s IDO inhibitor epacadostat has recently been making waves in combination with pembrolizumab in patients with advanced solid tumors. It demonstrated particularly promising clinical activity in patients with metastatic melanoma, with an overall response rate (ORR) of 57%, including 2 complete responses (CRs), prompting initiation of a phase 3 trial of this combination (NCT02752074).27
The relationship between the immune system and tumors is complex and dynamic, and for immunotherapy to reach its full potential it will likely need to attack on multiple fronts. Here, we discuss some of the latest and most promising developments in the immuno-oncology field designed to build on the successes and address limitations.
The anti-tumor immune response
Cancer is a disease of genomic instability, whereby genetic alterations ranging from a single nucleotide to the whole chromosome level frequently occur. Although cancers derive from a patient’s own tissues, these genetic differences can mark the cancer cell as non-self, triggering an immune response to eliminate these cells.
The first hints of this anti-tumor immunity date back more than a century and a half and sparked the concept of mobilizing the immune system to treat patients.1-3 Although early pioneers achieved little progress in this regard, their efforts provided invaluable insights into the complex and dynamic relationship between a tumor and the immune system that are now translating into real clinical successes.
We now understand that the immune system has a dual role in both restraining and promoting cancer development and have translated this understanding into the theory of cancer immunoediting. Immunoediting has three stages: elimination, wherein the tumor is seemingly destroyed by the innate and adaptive immune response; equilibrium, in which cancer cells that were able to escape elimination are selected for growth; and escape, whereby these resistant cancer cells overwhelm the immune system and develop into a symptomatic lesion.4,5
Immuno-oncologists have also described the cancer immunity cycle to capture the steps that are required for an effective anti-tumor immune response and defects in this cycle form the basis of the most common mechanisms used by cancer cells to subvert the anti-tumor immune response. Much like the cancer hallmarks did for molecularly targeted cancer drugs, the cancer immunity cycle serves as the intellectual framework for cancer immunotherapy.6,7
Exploiting nature’s weapon of mass destruction
Initially, attempts at immunotherapy focused on boosting the immune response using adjuvants and cytokines. The characterization of subtle differences between tumor cells and normal cells led to the development of vaccines and cell-based therapies that exploited these tumor-associated antigens (TAAs).1-6
Despite the approval of a therapeutic vaccine, sipuleucel-T, in 2010 for the treatment of metastatic prostate cancer, in general the success of vaccines has been limited. Marketing authorization for sipuleucel-T was recently withdrawn in Europe, and although it is still available in the United States, it is not widely used because of issues with production and administration. Other vaccines, such as GVAX, which looked particularly promising in early-stage clinical trials, failed to show clinical efficacy in subsequent testing.8,9
Cell-based therapies, such as adoptive cellular therapy (ACT), in which immune cells are removed from the host, primed to attack cancer cells, and then reinfused back into the patient, have focused on T cells because they are the major effectors of the adaptive immune response. Clinical success with the most common approach, tumor-infiltrating lymphocyte (TIL)
Two key techniques have been developed (Figure 1). T-cell receptor (TCR) therapy involves genetically modifying the receptor on the surface of T cells that is responsible for recognizing antigens bound to major histocompatibility complex (MHC) molecules on the surface of antigen-presenting cells (APCs). The TCR can be altered to recognize a specific TAA or modified to improve its antigen recognition and binding capabilities. This type of therapy is limited by the fact that the TCRs need to be genetically matched to the patient’s immune type.
Releasing the brakes
To ensure that it is only activated at the appropriate time and not in response to the antigens expressed on the surface of the host’s own tissues or harmless materials, the immune system has developed numerous mechanisms for immunological tolerance. Cancer cells are able to exploit these mechanisms to allow them to evade the anti-tumor immune response. One of the main ways in which they do this is by manipulating the signaling pathways involved in T-cell activation, which play a vital role in tolerance.12
To become fully activated, T cells require a primary signal generated by an interaction between the TCR and the antigen-MHC complex on the surface of an APC, followed by secondary costimulatory signals generated by a range of different receptors present on the T-cell surface binding to their ligands on the APC.
If the second signal is inhibitory rather than stimulatory, then the T cell is deactivated instead of becoming activated. Two key coinhibitory receptors are programmed cell death 1 (PD-1) and cytotoxic T-lymphocyte antigen 4 (CTLA-4) and tumor cells are able to overcome the anti-tumor immune response in part by expressing the ligands that bind these receptors to dampen the activity of tumor-infiltrating T cells and induce tolerance.13
The development of inhibitors of CTLA-4 and PD-1 and their respective ligands has driven some of the most dramatic successes with cancer immunotherapy, particularly with PD-1-targeting drugs which have fewer side effects. Targeting of this pathway has resulted in durable responses, revolutionizing the treatment of metastatic melanoma, with recently published long-term survival data for pembrolizumab showing that 40% of patients were alive 3 years after initiating treatment and, in a separate study, 34% of nivolumab-treated patients were still alive after 5 years.14,15 More recently, PD-1 inhibitors have been slowly expanding into a range of other cancer types and 4 immune checkpoint inhibitors are now approved by the United States Food and Drug Administration (FDA): ipilimumab (Yervoy), nivolumab (Opdivo), pembrolizumab (Keytruda) and atezolizumab (Tecentriq).
Six years on from the first approval in this drug class and an extensive network of coinhibitory receptors has been uncovered – so-called immune checkpoints – many of which are now also serving as therapeutic targets (Table, Figure 2).16 Lymphocyte activation gene 3 (LAG-3) is a member of the immunoglobulin superfamily of receptors that is expressed on a number of different types of immune cell. In addition to negatively regulating cytotoxic T-cell activation like PD-1 and CTLA-4, it is also thought to regulate the immunosuppressive functions of regulatory T cells and the maturation and activation of dendritic cells. T-cell immunoglobulin and mucin domain-containing 3 (TIM-3) is found on the surface of helper and cytotoxic T cells and regulates T-cell inhibition as well as macrophage activation. Inhibitors of both proteins have been developed that are being evaluated in phase 1 or 2 clinical trials in a variety of tumor types.17
Indeed, although T cells have commanded the most attention, there is growing appreciation of the potential for targeting other types of immune cell that play a role in the anti-tumor immune response or in fostering an immunosuppressive microenvironment. NK cells have been a particular focus, since they represent the body’s first line of immune defense and they appear to have analogous inhibitory and activating receptors expressed on their surface that regulate their cytotoxic activity.
The best-defined NK cell receptors are the killer cell immunoglobulin-like receptors (KIRs) that bind to the MHC class I proteins found on the surface of all cells that distinguish them as ‘self’ or ‘non-self’. KIRs can be either activating or inhibitory, depending upon their structure and the ligands to which they bind.19 To date, 2 antibodies targeting inhibitory KIRs have been developed. Though there has been some disappointment with these drugs, most recently a phase 2 trial of lirilumab in elderly patients with acute myeloid leukemia, which missed its primary endpoint, they continue to be evaluated in clinical trials.20
The inhibitory immune checkpoint field has also expanded to include molecules that regulate T-cell activity in other ways. Most prominently, this includes enzymes like indoleamine-2,3 dioxygenase (IDO), which is involved in the metabolism of the essential amino acid tryptophan. IDO-induced depletion of tryptophan and generation of tryptophan metabolites is toxic to cytotoxic T cells, and IDO is also thought to directly activate regulatory T cells, thus the net effect of IDO is immunosuppression. Two IDO inhibitors are currently being developed.21
Stepping on the gas
Despite their unprecedented success, immune checkpoint inhibitors are not effective in all patients or in all tumor types. Their efficacy is limited in large part by the requirement for a pre-existing anti-tumor immune response. If there are no T cells within the tumor microenvironment then releasing the brakes on the immune system won’t help.
More recently, researchers have returned to the idea of stimulating an anti-tumor immune response, this time by targeting the other side of the immune checkpoint coin, the costimulatory molecules. These drugs could prove more effective as they aren’t reliant on a pre-existing anti-tumor immune response. A number of agonist antibodies designed to target these receptors have now been developed and are undergoing clinical evaluation.22
Furthest along in development are those targeting OX40, a costimulatory molecule that is upregulated on the surface of T cells once they have been fully activated by the TCR signal and an initial costimulatory signal. OX40 is thought to be involved in a more long-term immune response and in the formation of a memory response. A mouse monoclonal antibody had a potent immune-stimulating effect accompanied by the regression of at least 1 metastatic lesion in 30% of patients treated in a phase 1 clinical trial, but was limited by the generation of anti-mouse antibodies. 7 OX40 agonists are now in clinical development, 6 fully human monoclonal antibodies and 1 OX40 ligand-Fc fusion protein, MEDI-6383.23
Combinations are key
Many researchers are now reaching the conclusion that combination therapy is likely to be key in expanding the scope of immunotherapy into currently unresponsive patient populations. Investigating rational combinations is already becoming a burgeoning area of the immuno-oncology field, with a variety of different strategies being tested.
Now the question becomes what are the optimal combinations and the timing and sequencing of combination therapy is likely to be a paramount consideration. Developing combinations that have distinct mechanisms of action or target multiple steps in the cancer immunity cycle offers the greatest potential for therapeutic synergy since this is most likely to address potential mechanisms of resistance by blocking other paths to immune evasion for cancer cells (Figure 3).
Given the expanding network of immune-checkpoint inhibitors and agonists, the focal point of combination therapy has been combining immune checkpoint-targeting drugs with different mechanisms of action, including those that would simultaneously release the brakes and step on the gas pedal. The vast majority of ongoing clinical trials of approved checkpoint inhibitors and the drugs in development listed in the table are combination trials.
These efforts yielded the first FDA-approved combination immunotherapy regimen in 2015; nivolumab and ipilimumab for the treatment of metastatic melanoma. Approval was based on the demonstration of improved ORR, prolonged response duration, and improved progression-free survival among 142 patients treated with the combination, compared to either drug alone.24
The results of a phase 1/2 trial evaluating the combination of a 4-1BB receptor agonist urelumab with nivolumab in hematologic malignancies and solid tumors found the combination to be safe and particularly effective in patients with advanced/metastatic melanoma, with an ORR of 50%.25 Nivolumab was also combined with the CD27 agonist varlilumab in a phase 1/2 clinical trial of patients with solid tumors, for which data was also recently released. Among 46 patients enrolled, primarily those with colorectal and ovarian cancer the combination had an acceptable safety profile and favorable changes in intratumoral immune biomarkers were observed. The phase 2 portion of the trial is ongoing.26
Meanwhile, Incyte’s IDO inhibitor epacadostat has recently been making waves in combination with pembrolizumab in patients with advanced solid tumors. It demonstrated particularly promising clinical activity in patients with metastatic melanoma, with an overall response rate (ORR) of 57%, including 2 complete responses (CRs), prompting initiation of a phase 3 trial of this combination (NCT02752074).27
1. Adams JL, Smothers J, Srinivasan R, et al. Big opportunities for small molecules in immuno-oncology. Nat Rev Drug Disc. 2015;14:603-622.
2. D’Errico G, Machado HL, Sainz Jr B. A current perspective on cancer immune therapy: step-by-step approach to constructing the magic bullet. Clin Trans Med. 2017;6:3.
3. Farkona S, Diamandis EP, Blaustig IM. Cancer immunotherapy: the beginning of the end of cancer? BMC Med. 2016;14:73.
4. Meiliana A, Dewi NM, Wijaya A. Cancer immunotherapy: a review. Indones Biomed J. 2016;8(1):1-20.
5. Smyth MJ, Ngiow SF, Ribas A, et al. Combination cancer immunotherapies tailored to the tumor microenvironment. Nat Rev Clin Oncol. 2016;13:143-158.
6. de Charette M, Marabelle A, Houot R. Turning tumor cells into antigen presenting cells: The next step to improve cancer immunotherapy? Eur J Cancer 2016;68:134-147.
7. Chen DS and Mellman I. Oncology Meets Immunology: The Cancer-Immunity Cycle. Immunity 2013;39:1-10.
8. Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature 2011;480:480-489.
9. Le DT, Wang-Gillam A, Picozzi V Jr, et al. A phase 2, randomized trial of GVAX Pancreas and CRS-207 immunotherapy versus GVAX alone in patients with metastatic pancreatic adenocarcinoma: Updated results. Presented at: the ASCO Gastrointestinal Cancers Symposium; January 16-18, 2014; San Francisco, CA. Abstract 177.
10. Sharpe M and Mount N. Genetically modified T cells in cancer therapy: opportunities and challenges. Dis Model Mech. 2015;8(4):337-350.
11. Perica K, Varela JC, Oelke M, et al. Adoptive T Cell Immunotherapy for Cancer. Ram Mai Med J. 2015;6(1):e0004.
12. Xing Y and Hogquist KA. T-Cell Tolerance: Central and Peripheral. Cold Spring Harb Perspect Biol. 2012;4:a006957.
13. Buchbinder EI and Desai A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016;39(1):98-106.
14. Robert C, Ribas A, Hamid O, et al. 3-year overall survival for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. J Clin Oncol. 2016(suppl;abstr 9503).
15. Hodi SF, Kluger HM, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Presented at the 2016 AACR Annual Meeting; April 16-20; New Orleans, LA. Abstract CT001.
16. Bakdash G, Sittig SP, van Dijk T, et al. The nature of activatory and tolerogenic dendritic cell-derived signal II. Front Immunol. 2013;4(53):1-18.
17. Sheridan C. Immuno-oncology moves beyond PD-1. Nat Biotechnol. 2015;33(7):673-675.
18. Blake SJ, Dougall WC, Miles JJ, et al. Molecular pathways: targeting CD96 and TIGIT for cancer immunotherapy. Clin Cancer Res. 2016;22(21):5183-5188.
19. Carotta S. Targeting NK cells for anticancer immunotherapy: clinical and preclinical approaches. Front Immunol. 2016;7:152.
20. Innate Pharma Web site. Innate Pharma Announces Top-Line Results from EFFIKIR Trial Evaluating the Efficacy of Lirilumab as a Single Agent in Elderly Patients with Acute Myeloid Leukemia. http://www.innate-pharma.com/en/news-events/press-releases/innate-pharma-announces-top-line-results-effikir-trial-evaluating-efficacy-lirilumab-single-agent-elderly-patients-acute-myeloid-leukemia. Last updated February 6, 2017. Accessed online February 22, 2017.
21. Sheridan C. IDO inhibitors move center stage in immuno-oncology. Nat Biotechnol. 2015;33(4):321-322.
22. Sanmamed MF, Pastor F, Rodriguez A, et al. Agonists of co-stimulation in cancer immunotherapy directed against CD137, OX40, GITR, CD27, CD28, and ICOS. Semin Oncol. 2015;42(4):640-655.
23. Linch SN, McNamara MJ, Redmond WL. OX40 agonists and combination immunotherapy: putting the pedal to the metal. Front Oncol. 2015;5:34.
24. U.S. Food and Drug Administration Web site. Nivolumab in combination with ipilimumab. https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm465274.htm. Last updated October 1, 2015. Accessed online February 22, 2017.
25. Massarelli E. Clinical safety and efficacy assessment of the CD137 agonist urelumab alone and in combination with nivolumab in patients with hematologic and solid tumor malignancies. Presented at the 31st Annual Meeting of the Society for the Immunotherapy of Cancer; November 9-13, 2016; National Harbor, MD. Abstract 239.
26. Sanborn RE, Pishvain MJ, Callahan MK, et al. Phase I results from the combination of an immune-activating anti-CD27 antibody (varlilumab) in combination with PD-1 blockade (nivolumab): activation across multiple immune pathways without untoward immune-related adverse events. Clin Cancer Res. 2016;76(14):suppl. Abstract CT023.
27. Gangadhar T, Hamid O, Smith D.C, et al. Epacadostat plus pembrolizumab in patients with advanced melanoma and select solid tumors: updated phase 1 results from ECHO-202/KEYNOTE-037. Ann Oncol. 2016;27(6):379-400.
1. Adams JL, Smothers J, Srinivasan R, et al. Big opportunities for small molecules in immuno-oncology. Nat Rev Drug Disc. 2015;14:603-622.
2. D’Errico G, Machado HL, Sainz Jr B. A current perspective on cancer immune therapy: step-by-step approach to constructing the magic bullet. Clin Trans Med. 2017;6:3.
3. Farkona S, Diamandis EP, Blaustig IM. Cancer immunotherapy: the beginning of the end of cancer? BMC Med. 2016;14:73.
4. Meiliana A, Dewi NM, Wijaya A. Cancer immunotherapy: a review. Indones Biomed J. 2016;8(1):1-20.
5. Smyth MJ, Ngiow SF, Ribas A, et al. Combination cancer immunotherapies tailored to the tumor microenvironment. Nat Rev Clin Oncol. 2016;13:143-158.
6. de Charette M, Marabelle A, Houot R. Turning tumor cells into antigen presenting cells: The next step to improve cancer immunotherapy? Eur J Cancer 2016;68:134-147.
7. Chen DS and Mellman I. Oncology Meets Immunology: The Cancer-Immunity Cycle. Immunity 2013;39:1-10.
8. Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature 2011;480:480-489.
9. Le DT, Wang-Gillam A, Picozzi V Jr, et al. A phase 2, randomized trial of GVAX Pancreas and CRS-207 immunotherapy versus GVAX alone in patients with metastatic pancreatic adenocarcinoma: Updated results. Presented at: the ASCO Gastrointestinal Cancers Symposium; January 16-18, 2014; San Francisco, CA. Abstract 177.
10. Sharpe M and Mount N. Genetically modified T cells in cancer therapy: opportunities and challenges. Dis Model Mech. 2015;8(4):337-350.
11. Perica K, Varela JC, Oelke M, et al. Adoptive T Cell Immunotherapy for Cancer. Ram Mai Med J. 2015;6(1):e0004.
12. Xing Y and Hogquist KA. T-Cell Tolerance: Central and Peripheral. Cold Spring Harb Perspect Biol. 2012;4:a006957.
13. Buchbinder EI and Desai A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016;39(1):98-106.
14. Robert C, Ribas A, Hamid O, et al. 3-year overall survival for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. J Clin Oncol. 2016(suppl;abstr 9503).
15. Hodi SF, Kluger HM, Sznol M, et al. Durable, long-term survival in previously treated patients with advanced melanoma who received nivolumab monotherapy in a phase I trial. Presented at the 2016 AACR Annual Meeting; April 16-20; New Orleans, LA. Abstract CT001.
16. Bakdash G, Sittig SP, van Dijk T, et al. The nature of activatory and tolerogenic dendritic cell-derived signal II. Front Immunol. 2013;4(53):1-18.
17. Sheridan C. Immuno-oncology moves beyond PD-1. Nat Biotechnol. 2015;33(7):673-675.
18. Blake SJ, Dougall WC, Miles JJ, et al. Molecular pathways: targeting CD96 and TIGIT for cancer immunotherapy. Clin Cancer Res. 2016;22(21):5183-5188.
19. Carotta S. Targeting NK cells for anticancer immunotherapy: clinical and preclinical approaches. Front Immunol. 2016;7:152.
20. Innate Pharma Web site. Innate Pharma Announces Top-Line Results from EFFIKIR Trial Evaluating the Efficacy of Lirilumab as a Single Agent in Elderly Patients with Acute Myeloid Leukemia. http://www.innate-pharma.com/en/news-events/press-releases/innate-pharma-announces-top-line-results-effikir-trial-evaluating-efficacy-lirilumab-single-agent-elderly-patients-acute-myeloid-leukemia. Last updated February 6, 2017. Accessed online February 22, 2017.
21. Sheridan C. IDO inhibitors move center stage in immuno-oncology. Nat Biotechnol. 2015;33(4):321-322.
22. Sanmamed MF, Pastor F, Rodriguez A, et al. Agonists of co-stimulation in cancer immunotherapy directed against CD137, OX40, GITR, CD27, CD28, and ICOS. Semin Oncol. 2015;42(4):640-655.
23. Linch SN, McNamara MJ, Redmond WL. OX40 agonists and combination immunotherapy: putting the pedal to the metal. Front Oncol. 2015;5:34.
24. U.S. Food and Drug Administration Web site. Nivolumab in combination with ipilimumab. https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm465274.htm. Last updated October 1, 2015. Accessed online February 22, 2017.
25. Massarelli E. Clinical safety and efficacy assessment of the CD137 agonist urelumab alone and in combination with nivolumab in patients with hematologic and solid tumor malignancies. Presented at the 31st Annual Meeting of the Society for the Immunotherapy of Cancer; November 9-13, 2016; National Harbor, MD. Abstract 239.
26. Sanborn RE, Pishvain MJ, Callahan MK, et al. Phase I results from the combination of an immune-activating anti-CD27 antibody (varlilumab) in combination with PD-1 blockade (nivolumab): activation across multiple immune pathways without untoward immune-related adverse events. Clin Cancer Res. 2016;76(14):suppl. Abstract CT023.
27. Gangadhar T, Hamid O, Smith D.C, et al. Epacadostat plus pembrolizumab in patients with advanced melanoma and select solid tumors: updated phase 1 results from ECHO-202/KEYNOTE-037. Ann Oncol. 2016;27(6):379-400.
Melanoma diagnosis does not deter pregnancy
Women in the United States do not appear to be delaying pregnancy after a diagnosis of melanoma, despite general recommendations to wait at least 2 years to attempt pregnancy because it might increase the risk of recurrence or exacerbate disease, investigators reported in the Journal of Surgical Research.
A review of records from a large national health care database showed that women aged 18-40 years with melanoma who were not pregnant on the index date had a significantly higher rate of pregnancy within 2 years, compared with matched controls, reported Julie A. DiSano, MD, from Penn State University, Hershey.
“These results suggest that a diagnosis of melanoma may serve as an impetus for some families to begin childbearing or have additional children sooner than they otherwise would have,” they wrote.
The investigators also found, reassuringly, that women who became pregnant after a melanoma diagnosis were not at increased risk for requiring additional therapy for the malignancy, at least in the short term.
Although earlier studies suggested that women who were pregnant at the time of a melanoma diagnosis had worse prognoses when compared with women who were not pregnant at the time of diagnosis, more recent studies have indicated women who are pregnant when diagnosed have similar outcomes as nonpregnant women with the same disease stage, the investigators noted.
“What is unclear and difficult to study is the relationship between melanoma and subsequent pregnancy rates, and pregnancy on melanoma outcomes. Very little data exist to guide women and physicians as to the safety of pregnancy after a diagnosis of melanoma. As a result, there are no formal guidelines for physicians who wish to counsel their patients regarding pregnancy after melanoma, and it is unknown whether women receive any counseling at all,” they wrote.
To get a clearer picture of the link between melanoma and subsequent pregnancy, the investigators scanned the Truven Health MarketScan database and identified 11,801 women from 18-40 years with melanoma who were not pregnant on the index date, determined by the earliest claim for melanoma diagnosis or therapy.
Each patient was matched on a 1:1 basis with women who did not have a melanoma claim at any time; cases were matched with controls on the basis of year of index date, age at index date, state of residence, and pregnancy status in the 90 days before the index date.
The authors found that the rate of pregnancy within 2 years of the index date was 15.8% for cases, compared with 13.6% for controls (P less than .001).
They also found, however, that women who required postsurgical therapy, suggesting more advanced disease stage or early recurrence, had a significantly lower probability of becoming pregnant within the first 9 months after the index date (hazard ratio, 0.26; P = .003).
There were no significant differences in the rate of postsurgical treatment by pregnancy status at either 3, 6, 9, or 12 months after surgery (P less than .05 for each), or in a Cox regression model for all time points (HR, 0.68, P = .23).
The authors offered several possible explanations for the higher pregnancy rates among women with melanoma, including the possibility that a cancer diagnosis could bring some couples closer together and “reorder” their priorities about starting a family.
“Another hypothesis is that families facing a melanoma diagnosis may decide to complete childbearing sooner in case the cancer recurs and subsequent treatment compromises fertility. Either way, the increased likelihood of pregnancy after melanoma diagnosis suggests an optimism about their future among families in the current childbearing generation in the United States,” they wrote.
The authors cautioned that the database does not include information about disease stage, and that “more detailed stage information is needed before revisiting recommendations.”
The study was supported by a Barsumian Trust grant; the authors reported having no conflicts of interest.
SOURCE: DiSano JA et al. J Surg Res. 2018 Jun 16. doi: 10.1016/j.jss.2018.05.026.
Women in the United States do not appear to be delaying pregnancy after a diagnosis of melanoma, despite general recommendations to wait at least 2 years to attempt pregnancy because it might increase the risk of recurrence or exacerbate disease, investigators reported in the Journal of Surgical Research.
A review of records from a large national health care database showed that women aged 18-40 years with melanoma who were not pregnant on the index date had a significantly higher rate of pregnancy within 2 years, compared with matched controls, reported Julie A. DiSano, MD, from Penn State University, Hershey.
“These results suggest that a diagnosis of melanoma may serve as an impetus for some families to begin childbearing or have additional children sooner than they otherwise would have,” they wrote.
The investigators also found, reassuringly, that women who became pregnant after a melanoma diagnosis were not at increased risk for requiring additional therapy for the malignancy, at least in the short term.
Although earlier studies suggested that women who were pregnant at the time of a melanoma diagnosis had worse prognoses when compared with women who were not pregnant at the time of diagnosis, more recent studies have indicated women who are pregnant when diagnosed have similar outcomes as nonpregnant women with the same disease stage, the investigators noted.
“What is unclear and difficult to study is the relationship between melanoma and subsequent pregnancy rates, and pregnancy on melanoma outcomes. Very little data exist to guide women and physicians as to the safety of pregnancy after a diagnosis of melanoma. As a result, there are no formal guidelines for physicians who wish to counsel their patients regarding pregnancy after melanoma, and it is unknown whether women receive any counseling at all,” they wrote.
To get a clearer picture of the link between melanoma and subsequent pregnancy, the investigators scanned the Truven Health MarketScan database and identified 11,801 women from 18-40 years with melanoma who were not pregnant on the index date, determined by the earliest claim for melanoma diagnosis or therapy.
Each patient was matched on a 1:1 basis with women who did not have a melanoma claim at any time; cases were matched with controls on the basis of year of index date, age at index date, state of residence, and pregnancy status in the 90 days before the index date.
The authors found that the rate of pregnancy within 2 years of the index date was 15.8% for cases, compared with 13.6% for controls (P less than .001).
They also found, however, that women who required postsurgical therapy, suggesting more advanced disease stage or early recurrence, had a significantly lower probability of becoming pregnant within the first 9 months after the index date (hazard ratio, 0.26; P = .003).
There were no significant differences in the rate of postsurgical treatment by pregnancy status at either 3, 6, 9, or 12 months after surgery (P less than .05 for each), or in a Cox regression model for all time points (HR, 0.68, P = .23).
The authors offered several possible explanations for the higher pregnancy rates among women with melanoma, including the possibility that a cancer diagnosis could bring some couples closer together and “reorder” their priorities about starting a family.
“Another hypothesis is that families facing a melanoma diagnosis may decide to complete childbearing sooner in case the cancer recurs and subsequent treatment compromises fertility. Either way, the increased likelihood of pregnancy after melanoma diagnosis suggests an optimism about their future among families in the current childbearing generation in the United States,” they wrote.
The authors cautioned that the database does not include information about disease stage, and that “more detailed stage information is needed before revisiting recommendations.”
The study was supported by a Barsumian Trust grant; the authors reported having no conflicts of interest.
SOURCE: DiSano JA et al. J Surg Res. 2018 Jun 16. doi: 10.1016/j.jss.2018.05.026.
Women in the United States do not appear to be delaying pregnancy after a diagnosis of melanoma, despite general recommendations to wait at least 2 years to attempt pregnancy because it might increase the risk of recurrence or exacerbate disease, investigators reported in the Journal of Surgical Research.
A review of records from a large national health care database showed that women aged 18-40 years with melanoma who were not pregnant on the index date had a significantly higher rate of pregnancy within 2 years, compared with matched controls, reported Julie A. DiSano, MD, from Penn State University, Hershey.
“These results suggest that a diagnosis of melanoma may serve as an impetus for some families to begin childbearing or have additional children sooner than they otherwise would have,” they wrote.
The investigators also found, reassuringly, that women who became pregnant after a melanoma diagnosis were not at increased risk for requiring additional therapy for the malignancy, at least in the short term.
Although earlier studies suggested that women who were pregnant at the time of a melanoma diagnosis had worse prognoses when compared with women who were not pregnant at the time of diagnosis, more recent studies have indicated women who are pregnant when diagnosed have similar outcomes as nonpregnant women with the same disease stage, the investigators noted.
“What is unclear and difficult to study is the relationship between melanoma and subsequent pregnancy rates, and pregnancy on melanoma outcomes. Very little data exist to guide women and physicians as to the safety of pregnancy after a diagnosis of melanoma. As a result, there are no formal guidelines for physicians who wish to counsel their patients regarding pregnancy after melanoma, and it is unknown whether women receive any counseling at all,” they wrote.
To get a clearer picture of the link between melanoma and subsequent pregnancy, the investigators scanned the Truven Health MarketScan database and identified 11,801 women from 18-40 years with melanoma who were not pregnant on the index date, determined by the earliest claim for melanoma diagnosis or therapy.
Each patient was matched on a 1:1 basis with women who did not have a melanoma claim at any time; cases were matched with controls on the basis of year of index date, age at index date, state of residence, and pregnancy status in the 90 days before the index date.
The authors found that the rate of pregnancy within 2 years of the index date was 15.8% for cases, compared with 13.6% for controls (P less than .001).
They also found, however, that women who required postsurgical therapy, suggesting more advanced disease stage or early recurrence, had a significantly lower probability of becoming pregnant within the first 9 months after the index date (hazard ratio, 0.26; P = .003).
There were no significant differences in the rate of postsurgical treatment by pregnancy status at either 3, 6, 9, or 12 months after surgery (P less than .05 for each), or in a Cox regression model for all time points (HR, 0.68, P = .23).
The authors offered several possible explanations for the higher pregnancy rates among women with melanoma, including the possibility that a cancer diagnosis could bring some couples closer together and “reorder” their priorities about starting a family.
“Another hypothesis is that families facing a melanoma diagnosis may decide to complete childbearing sooner in case the cancer recurs and subsequent treatment compromises fertility. Either way, the increased likelihood of pregnancy after melanoma diagnosis suggests an optimism about their future among families in the current childbearing generation in the United States,” they wrote.
The authors cautioned that the database does not include information about disease stage, and that “more detailed stage information is needed before revisiting recommendations.”
The study was supported by a Barsumian Trust grant; the authors reported having no conflicts of interest.
SOURCE: DiSano JA et al. J Surg Res. 2018 Jun 16. doi: 10.1016/j.jss.2018.05.026.
FROM THE JOURNAL OF SURGICAL RESEARCH
Key clinical point: Pregnancy after melanoma does not appear to increase risk for melanoma recurrence.
Major finding: The rate of pregnancy for women with melanoma was 15.8%, compared with 13.6% for controls (P less than .001).
Study details: A retrospective study of claims database records on 11,801 women with melanoma and an equal number of matched controls.
Disclosures: The study was supported by a Barsumian Trust grant; the authors reported having no conflicts of interest.
Source: DiSano JA et al. J Surg Res. 2018 Jun 16. doi: 10.1016/j.jss.2018.05.026.
Going Digital With Dermoscopy
Dermoscopic examination has been proven to increase diagnostic accuracy and decrease unnecessary biopsies of both melanoma and nonmelanoma skin cancers.1,2 Digital dermoscopy refers to acquiring and storing digital dermoscopic photographs via digital camera, smart image capture devices such as smartphones and tablets, or any other devices used for image acquisition. The stored images may then be used in a variety of ways, including sequential digital monitoring, teledermoscopy, and machine learning.
Sequential Digital Monitoring
Sequential digital dermoscopy imaging (SDDI) is the capture and storage of dermoscopic images of suspicious lesions that are then monitored over time for changes. Studies have shown that SDDI allows for early detection of melanomas and leads to a decrease in the number of unnecessary excisions.3,4 A meta-analysis of SDDI found that the chance of detecting melanoma increased with the length of monitoring, which suggests that continued follow-up, especially in high-risk groups, is crucial.4
Teledermoscopy
Teledermatology (telederm) is on the rise in the United States, with the number of programs and consultations increasing yearly. One study showed a 48% increase in telederm programs in the last 5 years.5 Studies have shown the addition of digital dermoscopic images improved the diagnostic accuracy in telederm skin cancer screenings versus clinical images alone.6,7
Telederm currently is practiced in 2 main models: live-interactive video consultation and storage of images for future consultation (store and forward). Medicare currently only reimburses live-interactive telederm for patients in nonmetropolitan areas and store-and-forward telederm pilot programs in Alaska and Hawaii; however, Medicaid does reimburse for store and forward in a handful of states.8 Similar to dermatoscope use during clinical examination, there currently is no additional reimbursement for teledermoscopy. Of note, a willingness-to-pay survey of 214 students from a southwestern university health center showed that participants were willing to pay an average (SD) of $55.27 ($39.11) out of pocket for a teledermoscopy/telederm evaluation, citing factors such as convenience.9
Direct-to-consumer telederm offers a new way for patients to receive care.10 Some dermatoscopes (eg, DermLite HÜD [3Gen], Molescope/Molescope II [Metaoptima Technology Inc]) currently are marketed directly to consumers along with telederm services to facilitate direct-to-patient teledermoscopy.11,12
Machine Learning
Big data and machine learning has been hailed as the future of medicine and dermatology alike.13 Machine learning is a type of artificial intelligence that uses computational algorithms (eg, neural networks) that allow computer programs to automatically improve their accuracy (learn) by analyzing large data sets. In dermatology, machine learning has been most notably used to train computers to identify images of skin cancer by way of large image databases.14-17 One algorithm, a convolutional neural network (CNN), made headlines in 2017 when it was able to identify dermoscopic and clinical images of skin cancer with comparable accuracy to a group of 21 dermatologists.14 In 2018, the International Skin Imaging Collaboration (ISIC) published results of a study of the diagnostic accuracy of 25 computer algorithms compared to 8 dermatologists using a set of 100 dermoscopic images of melanoma and benign nevi.15 Using the average sensitivity of the dermatologists (82%), the top fusion algorithm in the study had a sensitivity of 76% versus 59% for the dermatologists (P=.02). These results compared the mean sensitivity of the dermatologists, as some individual dermatologists outperformed the algorithm.15 More recently, another CNN was compared to 58 international dermatologists in the classification of a set of 100 dermoscopic images (20 melanoma and 80 melanocytic nevi).16 Using the mean sensitivity of the dermatologists (86.6%), the CNN had a specificity of 92.5% versus 71.3% for dermatologists (P<.01). In the second part of the study, the dermatologists were given some clinical information and close-up photographs of the lesions, which improved their average (SD) sensitivity and specificity to 88.9% (9.6%)(P=.19) and 75.7% (11.7%)(P<.05), respectively. When compared to the CNN at this higher sensitivity, the CNN still had a higher specificity than the dermatologists (82.5% vs 75.7% [P<.01]).16 However, in real-life clinical practice dermatologists perform better, not only because they can collect more in-person clinical information but also because humans gather more information during live examination than when they are interpreting close-up clinical and/or dermoscopic images. In a sense, we currently are limited to comparing data that is incommensurable.
Machine learning studies have other notable limitations, such as data sets that do not contain a full spectrum of skin lesions or less common lesions (eg, pigmented seborrheic keratoses, amelanotic melanomas) and variation in image databases used.15,16 For machine algorithms to improve, they require access to high-quality and ideally standardized digital dermoscopic image databases. The ISIC and other organizations currently have databases specifically for this purpose, but more images are needed.18 As additional practitioners incorporate digital dermoscopy in their clinical practice, the potential for larger databases and more accurate algorithms becomes a possibility.
Image Acquisition
Many devices are available for digital dermoscopic image acquisition, including dermatoscopes that attach to smartphones and/or digital cameras and all-in-one systems (eTable). The exact system employed will depend on the practitioner's requirements for price, portability, speed, image quality, and software. Digital single-lens reflex (DSLR) cameras boast the highest image quality, while video dermoscopy traditionally yields stored images with poor resolution.19 Macroscopic images obtained by other imaging devices, including spectral imaging devices and reflectance confocal microscopy, usually are yielded via video dermoscopy or a video camera to capture images; thus, stored images generally are not as high quality.

Smartphones are increasingly used for clinical imaging in dermatology.20 Although DSLR cameras still take the highest-quality images, current smartphone image quality is comparable to digital cameras.21,22 Computational photography uses computer processing power to enhance image quality and may bring smartphone image quality closer to DSLR cameras.22,23 Smartphones with newer dual-lens cameras have been reported to further improve image quality.21 Current smartphones have the option of enabling high-dynamic-range imaging, which combines multiple images taken with different exposures to create a single image with improved dynamic range of luminosity. It has been reported that high-dynamic-range imaging may even enhance dermoscopic features of more challenging hypopigmented skin cancers.24
Standardizing Imaging
There has been a concerted effort to standardize digital dermatologic image acquisition.25,26 Standardization promises to facilitate data analysis, improve collaboration, protect patient privacy, and improve patient care.13,26,27 At the forefront of image standardization is the ISIC organization, which recently published its Delphi consensus guidelines on standards for lesion imaging, including dermoscopy.26
The true holy grail of image standardization is the Digital Imaging and Communications in Medicine (DICOM) standard.26-28 The DICOM is a comprehensive imaging standard for storage, annotation, transfer, and display of images, and it is most notable for its use in radiology. The DICOM also could be applied to new imaging modalities in dermatology (eg, optical coherence tomography, reflectance confocal microscopy). Past efforts to develop a DICOM standard for dermatology were undertaken by a working group that has since disbanded.27 Work by the ISIC and many others will hopefully lead to adoption of the DICOM standard by dermatology at some point in the future.
Protected Health Information
The Health Insurance Portability and Accountability Act (HIPAA) requires protected health information (PHI) to be stored in a secure manner with limited access that sufficiently protects identifiable patient information. Although dermoscopic images generally are deidentified, they often are stored alongside clinical photographs and data that contains PHI in clinical practice.
Image storage can take 2 forms: (1) physical local storage on internal and external hard drives or (2) remote storage (eg, cloud-based storage). Encryption is essential regardless of the method of storage. It is required by law that loss of nonencrypted PHI be reported to all potentially affected patients, the US Department of Health & Human Services, and local/state media depending on the number of patients affected. Loss of PHI can result in fines of up to $1.5 million.29 On the contrary, loss of properly encrypted data would not be required to be reported.30
As smart image acquisition devices begin to dominate the clinical setting, practitioners need to be vigilant in securing patient PHI. There are multiple applications (apps) that allow for secure encrypted digital dermoscopic image acquisition and storage on smartphones. Additionally, it is important to secure smartphones with complex passcodes (eg, a mix of special characters, numbers, uppercase and lowercase letters). Most dermatoscope manufacturers have apps for image acquisition and storage that can be tied into other platforms or storage systems (eg, DermLite app [3Gen], Handyscope [FotoFinder Systems GmbH], VEOS app [Canfield Scientific, Inc]).28 Other options include syncing images with current electronic medical record technologies, transferring photographs to HIPAA-compliant cloud storage, or transferring photographs to an encrypted computer and/or external hard drive. Some tips for securing data based on HIPAA and other guidelines are listed in the Table.30,31

Conclusion
The expansion of teledermoscopy alongside direct-to-patient services may create additional incentives for clinicians to incorporate digital dermoscopy into their practice. As more practitioners adopt digital dermoscopy, machine learning driven by technological advancements and larger image data sets could influence the future practice of dermatology. With the rise in digital dermoscopy by way of smartphones, additional steps must be taken to ensure patients' PHI is safeguarded. Digital dermoscopy is a dynamic field that will likely see continued growth in the coming years.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Rosendahl C, Tschandl P, Cameron A, et al. Diagnostic accuracy of dermatoscopy for melanocytic and nonmelanocytic pigmented lesions. J Am Acad Dermatol. 2011;64:1068-1073.
- Salerni G, Lovatto L, Carrera C, et al. Melanomas detected in a follow-up program compared with melanomas referred to a melanoma unit. Arch Dermatol. 2011;147:549-555.
- Salerni G, Terán T, Puig S, et al. Meta-analysis of digital dermoscopy follow-up of melanocytic skin lesions: a study on behalf of the International Dermoscopy Society. J Eur Acad Dermatol Venereol. 2013;27:805-814.
- Yim KM, Armstrong AW, Oh DH, et al. Teledermatology in the United States: an update in a dynamic era [published online January 22, 2018]. Telemed J E Health. doi:10.1089/tmj.2017.0253.
- Ferrándiz L, Ojeda-Vila T, Corrales A, et al. Internet-based skin cancer screening using clinical images alone or in conjunction with dermoscopic images: a randomized teledermoscopy trial. J Am Acad Dermatol. 2017;76:676-682.
- Şenel E, Baba M, Durdu M. The contribution of teledermatoscopy to the diagnosis and management of non-melanocytic skin tumours. J Telemed Telecare. 2013;19:60-63.
- State telehealth laws and Medicaid program policies: a comprehensive scan of the 50 states and District of Columbia. Public Health Institute Center for Connected Health Policy website. http://www.cchpca.org/sites/default/files/resources/
50%20State%20FINAL%20April%202016.pdf. Published March 2016. Accessed July 2, 2018. - Raghu TS, Yiannias J, Sharma N, et al. Willingness to pay for teledermoscopy services at a university health center. J Patient Exp. 2018. doi:10.11772374373517748657.
- Fogel AL, Sarin KY. A survey of direct-to-consumer teledermatology services available to US patients: explosive growth, opportunities and controversy. J Telemed Telecare. 2017;23:19-25.
- MoleScope. MetaOptima Technology Inc website. https://molescope.com/product/. Accessed July 2, 2018.
- DermLite HÜD. 3Gen website. https://dermlite.com/products/dermlite-hud. Accessed July 2, 2018.
- Park AJ, Ko JM, Swerlick RA. Crowdsourcing dermatology: DataDerm, big data analytics, and machine learning technology. J Am Acad Dermatol. 2018;78:643-644.
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118.
- Marchetti MA, Codella NCF, Dusza SW, et al; International Skin Imaging Collaboration. results of the 2016 International Skin Imaging Collaboration International Symposium on Biomedical Imaging challenge: comparison of the accuracy of computer algorithms to dermatologists for the diagnosis of melanoma from dermoscopic images. J Am Acad Dermatol. 2018;78:270-277.
- Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists [published online May 28, 2018]. doi:10.1093/annonc/mdy166.
- Prado G, Kovarik C. Cutting edge technology in dermatology: virtual reality and artificial intelligence. Cutis. 2018;101:236-237.
- Sultana NN, Puhan NB. Recent deep learning methods for melanoma detection: a review. In: Ghosh D, Giri D, Mohapatra R, et al, eds. Mathematics and Computing. Singapore: Springer Nature; 2018:118-132.
- Lake A, Jones B. Dermoscopy: to cross-polarize, or not to cross-polarize, that is the question. J Vis Commun Med. 2015;38:36-50.
- Abbott LM, Magnusson RS, Gibbs E, et al. Smartphone use in dermatology for clinical photography and consultation: current practice and the law [published online February 28, 2017]. Australas J Dermatol. 2018;59:101-107.
- Hauser W, Neveu B, Jourdain JB, et al. Image quality benchmark of computational bokeh. Electron Imaging. 2018;2018:1-10.
- Ignatov A, Kobyshev N, Timofte R, et al. DSLR-quality photos on mobile devices with deep convolutional networks. 2017 IEEE International Conference on Computer Vision (ICCV). Venice, Italy: IEEE; 2017:3297-3305.
- Greengard S. Computational photography comes into focus. Commun ACM. 2014;57:19-21.
- Braun RP, Marghoob A. High-dynamic-range dermoscopy imaging and diagnosis of hypopigmented skin cancers. JAMA Dermatol. 2015;151:456-457.
- Quigley EA, Tokay BA, Jewell ST, et al. Technology and technique standards for camera-acquired digital dermatologic images: a systematic review. JAMA Dermatol. 2015;151:883-890.
- Katragadda C, Finnane A, Soyer HP, et al. Technique standards for skin lesion imaging a delphi consensus statement. JAMA Dermatol. 2017;153:207-213.
- Caffery LJ, Clunie D, Curiel-Lewandrowski C, et al. Transforming dermatologic imaging for the digital era: metadata and standards [published online January 17, 2018]. J Digit Imaging. doi:10.1007/s10278-017-0045-8.
- Pagliarello C, Stanganelli I, Fabrizi G, et al. Digital dermoscopy monitoring: is it time to define a quality standard? Acta Derm Venereol. 2017;97:864-865.
- HITECH Act Enforcement Interim Final Rule. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/hitech-act-enforcement-interim-final-rule/index.html. Updated June 16, 2017. Accessed July 2, 2018.
- Guidance to render unsecured protected health information unusable, unreadable, or indecipherable to unauthorized individuals. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/breach-notification/guidance/index.html. Updated July 26, 2013. Accessed July 2, 2018.
- Scarfone K, Souppaya M, Sexton M. Guide to Storage Encryption Technologies for End User Devices. Gaithersburg, MD: US Department of Commerce; 2007. NIST Special Publication 800-111.
Dermoscopic examination has been proven to increase diagnostic accuracy and decrease unnecessary biopsies of both melanoma and nonmelanoma skin cancers.1,2 Digital dermoscopy refers to acquiring and storing digital dermoscopic photographs via digital camera, smart image capture devices such as smartphones and tablets, or any other devices used for image acquisition. The stored images may then be used in a variety of ways, including sequential digital monitoring, teledermoscopy, and machine learning.
Sequential Digital Monitoring
Sequential digital dermoscopy imaging (SDDI) is the capture and storage of dermoscopic images of suspicious lesions that are then monitored over time for changes. Studies have shown that SDDI allows for early detection of melanomas and leads to a decrease in the number of unnecessary excisions.3,4 A meta-analysis of SDDI found that the chance of detecting melanoma increased with the length of monitoring, which suggests that continued follow-up, especially in high-risk groups, is crucial.4
Teledermoscopy
Teledermatology (telederm) is on the rise in the United States, with the number of programs and consultations increasing yearly. One study showed a 48% increase in telederm programs in the last 5 years.5 Studies have shown the addition of digital dermoscopic images improved the diagnostic accuracy in telederm skin cancer screenings versus clinical images alone.6,7
Telederm currently is practiced in 2 main models: live-interactive video consultation and storage of images for future consultation (store and forward). Medicare currently only reimburses live-interactive telederm for patients in nonmetropolitan areas and store-and-forward telederm pilot programs in Alaska and Hawaii; however, Medicaid does reimburse for store and forward in a handful of states.8 Similar to dermatoscope use during clinical examination, there currently is no additional reimbursement for teledermoscopy. Of note, a willingness-to-pay survey of 214 students from a southwestern university health center showed that participants were willing to pay an average (SD) of $55.27 ($39.11) out of pocket for a teledermoscopy/telederm evaluation, citing factors such as convenience.9
Direct-to-consumer telederm offers a new way for patients to receive care.10 Some dermatoscopes (eg, DermLite HÜD [3Gen], Molescope/Molescope II [Metaoptima Technology Inc]) currently are marketed directly to consumers along with telederm services to facilitate direct-to-patient teledermoscopy.11,12
Machine Learning
Big data and machine learning has been hailed as the future of medicine and dermatology alike.13 Machine learning is a type of artificial intelligence that uses computational algorithms (eg, neural networks) that allow computer programs to automatically improve their accuracy (learn) by analyzing large data sets. In dermatology, machine learning has been most notably used to train computers to identify images of skin cancer by way of large image databases.14-17 One algorithm, a convolutional neural network (CNN), made headlines in 2017 when it was able to identify dermoscopic and clinical images of skin cancer with comparable accuracy to a group of 21 dermatologists.14 In 2018, the International Skin Imaging Collaboration (ISIC) published results of a study of the diagnostic accuracy of 25 computer algorithms compared to 8 dermatologists using a set of 100 dermoscopic images of melanoma and benign nevi.15 Using the average sensitivity of the dermatologists (82%), the top fusion algorithm in the study had a sensitivity of 76% versus 59% for the dermatologists (P=.02). These results compared the mean sensitivity of the dermatologists, as some individual dermatologists outperformed the algorithm.15 More recently, another CNN was compared to 58 international dermatologists in the classification of a set of 100 dermoscopic images (20 melanoma and 80 melanocytic nevi).16 Using the mean sensitivity of the dermatologists (86.6%), the CNN had a specificity of 92.5% versus 71.3% for dermatologists (P<.01). In the second part of the study, the dermatologists were given some clinical information and close-up photographs of the lesions, which improved their average (SD) sensitivity and specificity to 88.9% (9.6%)(P=.19) and 75.7% (11.7%)(P<.05), respectively. When compared to the CNN at this higher sensitivity, the CNN still had a higher specificity than the dermatologists (82.5% vs 75.7% [P<.01]).16 However, in real-life clinical practice dermatologists perform better, not only because they can collect more in-person clinical information but also because humans gather more information during live examination than when they are interpreting close-up clinical and/or dermoscopic images. In a sense, we currently are limited to comparing data that is incommensurable.
Machine learning studies have other notable limitations, such as data sets that do not contain a full spectrum of skin lesions or less common lesions (eg, pigmented seborrheic keratoses, amelanotic melanomas) and variation in image databases used.15,16 For machine algorithms to improve, they require access to high-quality and ideally standardized digital dermoscopic image databases. The ISIC and other organizations currently have databases specifically for this purpose, but more images are needed.18 As additional practitioners incorporate digital dermoscopy in their clinical practice, the potential for larger databases and more accurate algorithms becomes a possibility.
Image Acquisition
Many devices are available for digital dermoscopic image acquisition, including dermatoscopes that attach to smartphones and/or digital cameras and all-in-one systems (eTable). The exact system employed will depend on the practitioner's requirements for price, portability, speed, image quality, and software. Digital single-lens reflex (DSLR) cameras boast the highest image quality, while video dermoscopy traditionally yields stored images with poor resolution.19 Macroscopic images obtained by other imaging devices, including spectral imaging devices and reflectance confocal microscopy, usually are yielded via video dermoscopy or a video camera to capture images; thus, stored images generally are not as high quality.

Smartphones are increasingly used for clinical imaging in dermatology.20 Although DSLR cameras still take the highest-quality images, current smartphone image quality is comparable to digital cameras.21,22 Computational photography uses computer processing power to enhance image quality and may bring smartphone image quality closer to DSLR cameras.22,23 Smartphones with newer dual-lens cameras have been reported to further improve image quality.21 Current smartphones have the option of enabling high-dynamic-range imaging, which combines multiple images taken with different exposures to create a single image with improved dynamic range of luminosity. It has been reported that high-dynamic-range imaging may even enhance dermoscopic features of more challenging hypopigmented skin cancers.24
Standardizing Imaging
There has been a concerted effort to standardize digital dermatologic image acquisition.25,26 Standardization promises to facilitate data analysis, improve collaboration, protect patient privacy, and improve patient care.13,26,27 At the forefront of image standardization is the ISIC organization, which recently published its Delphi consensus guidelines on standards for lesion imaging, including dermoscopy.26
The true holy grail of image standardization is the Digital Imaging and Communications in Medicine (DICOM) standard.26-28 The DICOM is a comprehensive imaging standard for storage, annotation, transfer, and display of images, and it is most notable for its use in radiology. The DICOM also could be applied to new imaging modalities in dermatology (eg, optical coherence tomography, reflectance confocal microscopy). Past efforts to develop a DICOM standard for dermatology were undertaken by a working group that has since disbanded.27 Work by the ISIC and many others will hopefully lead to adoption of the DICOM standard by dermatology at some point in the future.
Protected Health Information
The Health Insurance Portability and Accountability Act (HIPAA) requires protected health information (PHI) to be stored in a secure manner with limited access that sufficiently protects identifiable patient information. Although dermoscopic images generally are deidentified, they often are stored alongside clinical photographs and data that contains PHI in clinical practice.
Image storage can take 2 forms: (1) physical local storage on internal and external hard drives or (2) remote storage (eg, cloud-based storage). Encryption is essential regardless of the method of storage. It is required by law that loss of nonencrypted PHI be reported to all potentially affected patients, the US Department of Health & Human Services, and local/state media depending on the number of patients affected. Loss of PHI can result in fines of up to $1.5 million.29 On the contrary, loss of properly encrypted data would not be required to be reported.30
As smart image acquisition devices begin to dominate the clinical setting, practitioners need to be vigilant in securing patient PHI. There are multiple applications (apps) that allow for secure encrypted digital dermoscopic image acquisition and storage on smartphones. Additionally, it is important to secure smartphones with complex passcodes (eg, a mix of special characters, numbers, uppercase and lowercase letters). Most dermatoscope manufacturers have apps for image acquisition and storage that can be tied into other platforms or storage systems (eg, DermLite app [3Gen], Handyscope [FotoFinder Systems GmbH], VEOS app [Canfield Scientific, Inc]).28 Other options include syncing images with current electronic medical record technologies, transferring photographs to HIPAA-compliant cloud storage, or transferring photographs to an encrypted computer and/or external hard drive. Some tips for securing data based on HIPAA and other guidelines are listed in the Table.30,31

Conclusion
The expansion of teledermoscopy alongside direct-to-patient services may create additional incentives for clinicians to incorporate digital dermoscopy into their practice. As more practitioners adopt digital dermoscopy, machine learning driven by technological advancements and larger image data sets could influence the future practice of dermatology. With the rise in digital dermoscopy by way of smartphones, additional steps must be taken to ensure patients' PHI is safeguarded. Digital dermoscopy is a dynamic field that will likely see continued growth in the coming years.
Dermoscopic examination has been proven to increase diagnostic accuracy and decrease unnecessary biopsies of both melanoma and nonmelanoma skin cancers.1,2 Digital dermoscopy refers to acquiring and storing digital dermoscopic photographs via digital camera, smart image capture devices such as smartphones and tablets, or any other devices used for image acquisition. The stored images may then be used in a variety of ways, including sequential digital monitoring, teledermoscopy, and machine learning.
Sequential Digital Monitoring
Sequential digital dermoscopy imaging (SDDI) is the capture and storage of dermoscopic images of suspicious lesions that are then monitored over time for changes. Studies have shown that SDDI allows for early detection of melanomas and leads to a decrease in the number of unnecessary excisions.3,4 A meta-analysis of SDDI found that the chance of detecting melanoma increased with the length of monitoring, which suggests that continued follow-up, especially in high-risk groups, is crucial.4
Teledermoscopy
Teledermatology (telederm) is on the rise in the United States, with the number of programs and consultations increasing yearly. One study showed a 48% increase in telederm programs in the last 5 years.5 Studies have shown the addition of digital dermoscopic images improved the diagnostic accuracy in telederm skin cancer screenings versus clinical images alone.6,7
Telederm currently is practiced in 2 main models: live-interactive video consultation and storage of images for future consultation (store and forward). Medicare currently only reimburses live-interactive telederm for patients in nonmetropolitan areas and store-and-forward telederm pilot programs in Alaska and Hawaii; however, Medicaid does reimburse for store and forward in a handful of states.8 Similar to dermatoscope use during clinical examination, there currently is no additional reimbursement for teledermoscopy. Of note, a willingness-to-pay survey of 214 students from a southwestern university health center showed that participants were willing to pay an average (SD) of $55.27 ($39.11) out of pocket for a teledermoscopy/telederm evaluation, citing factors such as convenience.9
Direct-to-consumer telederm offers a new way for patients to receive care.10 Some dermatoscopes (eg, DermLite HÜD [3Gen], Molescope/Molescope II [Metaoptima Technology Inc]) currently are marketed directly to consumers along with telederm services to facilitate direct-to-patient teledermoscopy.11,12
Machine Learning
Big data and machine learning has been hailed as the future of medicine and dermatology alike.13 Machine learning is a type of artificial intelligence that uses computational algorithms (eg, neural networks) that allow computer programs to automatically improve their accuracy (learn) by analyzing large data sets. In dermatology, machine learning has been most notably used to train computers to identify images of skin cancer by way of large image databases.14-17 One algorithm, a convolutional neural network (CNN), made headlines in 2017 when it was able to identify dermoscopic and clinical images of skin cancer with comparable accuracy to a group of 21 dermatologists.14 In 2018, the International Skin Imaging Collaboration (ISIC) published results of a study of the diagnostic accuracy of 25 computer algorithms compared to 8 dermatologists using a set of 100 dermoscopic images of melanoma and benign nevi.15 Using the average sensitivity of the dermatologists (82%), the top fusion algorithm in the study had a sensitivity of 76% versus 59% for the dermatologists (P=.02). These results compared the mean sensitivity of the dermatologists, as some individual dermatologists outperformed the algorithm.15 More recently, another CNN was compared to 58 international dermatologists in the classification of a set of 100 dermoscopic images (20 melanoma and 80 melanocytic nevi).16 Using the mean sensitivity of the dermatologists (86.6%), the CNN had a specificity of 92.5% versus 71.3% for dermatologists (P<.01). In the second part of the study, the dermatologists were given some clinical information and close-up photographs of the lesions, which improved their average (SD) sensitivity and specificity to 88.9% (9.6%)(P=.19) and 75.7% (11.7%)(P<.05), respectively. When compared to the CNN at this higher sensitivity, the CNN still had a higher specificity than the dermatologists (82.5% vs 75.7% [P<.01]).16 However, in real-life clinical practice dermatologists perform better, not only because they can collect more in-person clinical information but also because humans gather more information during live examination than when they are interpreting close-up clinical and/or dermoscopic images. In a sense, we currently are limited to comparing data that is incommensurable.
Machine learning studies have other notable limitations, such as data sets that do not contain a full spectrum of skin lesions or less common lesions (eg, pigmented seborrheic keratoses, amelanotic melanomas) and variation in image databases used.15,16 For machine algorithms to improve, they require access to high-quality and ideally standardized digital dermoscopic image databases. The ISIC and other organizations currently have databases specifically for this purpose, but more images are needed.18 As additional practitioners incorporate digital dermoscopy in their clinical practice, the potential for larger databases and more accurate algorithms becomes a possibility.
Image Acquisition
Many devices are available for digital dermoscopic image acquisition, including dermatoscopes that attach to smartphones and/or digital cameras and all-in-one systems (eTable). The exact system employed will depend on the practitioner's requirements for price, portability, speed, image quality, and software. Digital single-lens reflex (DSLR) cameras boast the highest image quality, while video dermoscopy traditionally yields stored images with poor resolution.19 Macroscopic images obtained by other imaging devices, including spectral imaging devices and reflectance confocal microscopy, usually are yielded via video dermoscopy or a video camera to capture images; thus, stored images generally are not as high quality.

Smartphones are increasingly used for clinical imaging in dermatology.20 Although DSLR cameras still take the highest-quality images, current smartphone image quality is comparable to digital cameras.21,22 Computational photography uses computer processing power to enhance image quality and may bring smartphone image quality closer to DSLR cameras.22,23 Smartphones with newer dual-lens cameras have been reported to further improve image quality.21 Current smartphones have the option of enabling high-dynamic-range imaging, which combines multiple images taken with different exposures to create a single image with improved dynamic range of luminosity. It has been reported that high-dynamic-range imaging may even enhance dermoscopic features of more challenging hypopigmented skin cancers.24
Standardizing Imaging
There has been a concerted effort to standardize digital dermatologic image acquisition.25,26 Standardization promises to facilitate data analysis, improve collaboration, protect patient privacy, and improve patient care.13,26,27 At the forefront of image standardization is the ISIC organization, which recently published its Delphi consensus guidelines on standards for lesion imaging, including dermoscopy.26
The true holy grail of image standardization is the Digital Imaging and Communications in Medicine (DICOM) standard.26-28 The DICOM is a comprehensive imaging standard for storage, annotation, transfer, and display of images, and it is most notable for its use in radiology. The DICOM also could be applied to new imaging modalities in dermatology (eg, optical coherence tomography, reflectance confocal microscopy). Past efforts to develop a DICOM standard for dermatology were undertaken by a working group that has since disbanded.27 Work by the ISIC and many others will hopefully lead to adoption of the DICOM standard by dermatology at some point in the future.
Protected Health Information
The Health Insurance Portability and Accountability Act (HIPAA) requires protected health information (PHI) to be stored in a secure manner with limited access that sufficiently protects identifiable patient information. Although dermoscopic images generally are deidentified, they often are stored alongside clinical photographs and data that contains PHI in clinical practice.
Image storage can take 2 forms: (1) physical local storage on internal and external hard drives or (2) remote storage (eg, cloud-based storage). Encryption is essential regardless of the method of storage. It is required by law that loss of nonencrypted PHI be reported to all potentially affected patients, the US Department of Health & Human Services, and local/state media depending on the number of patients affected. Loss of PHI can result in fines of up to $1.5 million.29 On the contrary, loss of properly encrypted data would not be required to be reported.30
As smart image acquisition devices begin to dominate the clinical setting, practitioners need to be vigilant in securing patient PHI. There are multiple applications (apps) that allow for secure encrypted digital dermoscopic image acquisition and storage on smartphones. Additionally, it is important to secure smartphones with complex passcodes (eg, a mix of special characters, numbers, uppercase and lowercase letters). Most dermatoscope manufacturers have apps for image acquisition and storage that can be tied into other platforms or storage systems (eg, DermLite app [3Gen], Handyscope [FotoFinder Systems GmbH], VEOS app [Canfield Scientific, Inc]).28 Other options include syncing images with current electronic medical record technologies, transferring photographs to HIPAA-compliant cloud storage, or transferring photographs to an encrypted computer and/or external hard drive. Some tips for securing data based on HIPAA and other guidelines are listed in the Table.30,31

Conclusion
The expansion of teledermoscopy alongside direct-to-patient services may create additional incentives for clinicians to incorporate digital dermoscopy into their practice. As more practitioners adopt digital dermoscopy, machine learning driven by technological advancements and larger image data sets could influence the future practice of dermatology. With the rise in digital dermoscopy by way of smartphones, additional steps must be taken to ensure patients' PHI is safeguarded. Digital dermoscopy is a dynamic field that will likely see continued growth in the coming years.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Rosendahl C, Tschandl P, Cameron A, et al. Diagnostic accuracy of dermatoscopy for melanocytic and nonmelanocytic pigmented lesions. J Am Acad Dermatol. 2011;64:1068-1073.
- Salerni G, Lovatto L, Carrera C, et al. Melanomas detected in a follow-up program compared with melanomas referred to a melanoma unit. Arch Dermatol. 2011;147:549-555.
- Salerni G, Terán T, Puig S, et al. Meta-analysis of digital dermoscopy follow-up of melanocytic skin lesions: a study on behalf of the International Dermoscopy Society. J Eur Acad Dermatol Venereol. 2013;27:805-814.
- Yim KM, Armstrong AW, Oh DH, et al. Teledermatology in the United States: an update in a dynamic era [published online January 22, 2018]. Telemed J E Health. doi:10.1089/tmj.2017.0253.
- Ferrándiz L, Ojeda-Vila T, Corrales A, et al. Internet-based skin cancer screening using clinical images alone or in conjunction with dermoscopic images: a randomized teledermoscopy trial. J Am Acad Dermatol. 2017;76:676-682.
- Şenel E, Baba M, Durdu M. The contribution of teledermatoscopy to the diagnosis and management of non-melanocytic skin tumours. J Telemed Telecare. 2013;19:60-63.
- State telehealth laws and Medicaid program policies: a comprehensive scan of the 50 states and District of Columbia. Public Health Institute Center for Connected Health Policy website. http://www.cchpca.org/sites/default/files/resources/
50%20State%20FINAL%20April%202016.pdf. Published March 2016. Accessed July 2, 2018. - Raghu TS, Yiannias J, Sharma N, et al. Willingness to pay for teledermoscopy services at a university health center. J Patient Exp. 2018. doi:10.11772374373517748657.
- Fogel AL, Sarin KY. A survey of direct-to-consumer teledermatology services available to US patients: explosive growth, opportunities and controversy. J Telemed Telecare. 2017;23:19-25.
- MoleScope. MetaOptima Technology Inc website. https://molescope.com/product/. Accessed July 2, 2018.
- DermLite HÜD. 3Gen website. https://dermlite.com/products/dermlite-hud. Accessed July 2, 2018.
- Park AJ, Ko JM, Swerlick RA. Crowdsourcing dermatology: DataDerm, big data analytics, and machine learning technology. J Am Acad Dermatol. 2018;78:643-644.
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118.
- Marchetti MA, Codella NCF, Dusza SW, et al; International Skin Imaging Collaboration. results of the 2016 International Skin Imaging Collaboration International Symposium on Biomedical Imaging challenge: comparison of the accuracy of computer algorithms to dermatologists for the diagnosis of melanoma from dermoscopic images. J Am Acad Dermatol. 2018;78:270-277.
- Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists [published online May 28, 2018]. doi:10.1093/annonc/mdy166.
- Prado G, Kovarik C. Cutting edge technology in dermatology: virtual reality and artificial intelligence. Cutis. 2018;101:236-237.
- Sultana NN, Puhan NB. Recent deep learning methods for melanoma detection: a review. In: Ghosh D, Giri D, Mohapatra R, et al, eds. Mathematics and Computing. Singapore: Springer Nature; 2018:118-132.
- Lake A, Jones B. Dermoscopy: to cross-polarize, or not to cross-polarize, that is the question. J Vis Commun Med. 2015;38:36-50.
- Abbott LM, Magnusson RS, Gibbs E, et al. Smartphone use in dermatology for clinical photography and consultation: current practice and the law [published online February 28, 2017]. Australas J Dermatol. 2018;59:101-107.
- Hauser W, Neveu B, Jourdain JB, et al. Image quality benchmark of computational bokeh. Electron Imaging. 2018;2018:1-10.
- Ignatov A, Kobyshev N, Timofte R, et al. DSLR-quality photos on mobile devices with deep convolutional networks. 2017 IEEE International Conference on Computer Vision (ICCV). Venice, Italy: IEEE; 2017:3297-3305.
- Greengard S. Computational photography comes into focus. Commun ACM. 2014;57:19-21.
- Braun RP, Marghoob A. High-dynamic-range dermoscopy imaging and diagnosis of hypopigmented skin cancers. JAMA Dermatol. 2015;151:456-457.
- Quigley EA, Tokay BA, Jewell ST, et al. Technology and technique standards for camera-acquired digital dermatologic images: a systematic review. JAMA Dermatol. 2015;151:883-890.
- Katragadda C, Finnane A, Soyer HP, et al. Technique standards for skin lesion imaging a delphi consensus statement. JAMA Dermatol. 2017;153:207-213.
- Caffery LJ, Clunie D, Curiel-Lewandrowski C, et al. Transforming dermatologic imaging for the digital era: metadata and standards [published online January 17, 2018]. J Digit Imaging. doi:10.1007/s10278-017-0045-8.
- Pagliarello C, Stanganelli I, Fabrizi G, et al. Digital dermoscopy monitoring: is it time to define a quality standard? Acta Derm Venereol. 2017;97:864-865.
- HITECH Act Enforcement Interim Final Rule. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/hitech-act-enforcement-interim-final-rule/index.html. Updated June 16, 2017. Accessed July 2, 2018.
- Guidance to render unsecured protected health information unusable, unreadable, or indecipherable to unauthorized individuals. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/breach-notification/guidance/index.html. Updated July 26, 2013. Accessed July 2, 2018.
- Scarfone K, Souppaya M, Sexton M. Guide to Storage Encryption Technologies for End User Devices. Gaithersburg, MD: US Department of Commerce; 2007. NIST Special Publication 800-111.
- Vestergaard ME, Macaskill P, Holt PE, et al. Dermoscopy compared with naked eye examination for the diagnosis of primary melanoma: a meta-analysis of studies performed in a clinical setting. Br J Dermatol. 2008;159:669-676.
- Rosendahl C, Tschandl P, Cameron A, et al. Diagnostic accuracy of dermatoscopy for melanocytic and nonmelanocytic pigmented lesions. J Am Acad Dermatol. 2011;64:1068-1073.
- Salerni G, Lovatto L, Carrera C, et al. Melanomas detected in a follow-up program compared with melanomas referred to a melanoma unit. Arch Dermatol. 2011;147:549-555.
- Salerni G, Terán T, Puig S, et al. Meta-analysis of digital dermoscopy follow-up of melanocytic skin lesions: a study on behalf of the International Dermoscopy Society. J Eur Acad Dermatol Venereol. 2013;27:805-814.
- Yim KM, Armstrong AW, Oh DH, et al. Teledermatology in the United States: an update in a dynamic era [published online January 22, 2018]. Telemed J E Health. doi:10.1089/tmj.2017.0253.
- Ferrándiz L, Ojeda-Vila T, Corrales A, et al. Internet-based skin cancer screening using clinical images alone or in conjunction with dermoscopic images: a randomized teledermoscopy trial. J Am Acad Dermatol. 2017;76:676-682.
- Şenel E, Baba M, Durdu M. The contribution of teledermatoscopy to the diagnosis and management of non-melanocytic skin tumours. J Telemed Telecare. 2013;19:60-63.
- State telehealth laws and Medicaid program policies: a comprehensive scan of the 50 states and District of Columbia. Public Health Institute Center for Connected Health Policy website. http://www.cchpca.org/sites/default/files/resources/
50%20State%20FINAL%20April%202016.pdf. Published March 2016. Accessed July 2, 2018. - Raghu TS, Yiannias J, Sharma N, et al. Willingness to pay for teledermoscopy services at a university health center. J Patient Exp. 2018. doi:10.11772374373517748657.
- Fogel AL, Sarin KY. A survey of direct-to-consumer teledermatology services available to US patients: explosive growth, opportunities and controversy. J Telemed Telecare. 2017;23:19-25.
- MoleScope. MetaOptima Technology Inc website. https://molescope.com/product/. Accessed July 2, 2018.
- DermLite HÜD. 3Gen website. https://dermlite.com/products/dermlite-hud. Accessed July 2, 2018.
- Park AJ, Ko JM, Swerlick RA. Crowdsourcing dermatology: DataDerm, big data analytics, and machine learning technology. J Am Acad Dermatol. 2018;78:643-644.
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118.
- Marchetti MA, Codella NCF, Dusza SW, et al; International Skin Imaging Collaboration. results of the 2016 International Skin Imaging Collaboration International Symposium on Biomedical Imaging challenge: comparison of the accuracy of computer algorithms to dermatologists for the diagnosis of melanoma from dermoscopic images. J Am Acad Dermatol. 2018;78:270-277.
- Haenssle HA, Fink C, Schneiderbauer R, et al. Man against machine: diagnostic performance of a deep learning convolutional neural network for dermoscopic melanoma recognition in comparison to 58 dermatologists [published online May 28, 2018]. doi:10.1093/annonc/mdy166.
- Prado G, Kovarik C. Cutting edge technology in dermatology: virtual reality and artificial intelligence. Cutis. 2018;101:236-237.
- Sultana NN, Puhan NB. Recent deep learning methods for melanoma detection: a review. In: Ghosh D, Giri D, Mohapatra R, et al, eds. Mathematics and Computing. Singapore: Springer Nature; 2018:118-132.
- Lake A, Jones B. Dermoscopy: to cross-polarize, or not to cross-polarize, that is the question. J Vis Commun Med. 2015;38:36-50.
- Abbott LM, Magnusson RS, Gibbs E, et al. Smartphone use in dermatology for clinical photography and consultation: current practice and the law [published online February 28, 2017]. Australas J Dermatol. 2018;59:101-107.
- Hauser W, Neveu B, Jourdain JB, et al. Image quality benchmark of computational bokeh. Electron Imaging. 2018;2018:1-10.
- Ignatov A, Kobyshev N, Timofte R, et al. DSLR-quality photos on mobile devices with deep convolutional networks. 2017 IEEE International Conference on Computer Vision (ICCV). Venice, Italy: IEEE; 2017:3297-3305.
- Greengard S. Computational photography comes into focus. Commun ACM. 2014;57:19-21.
- Braun RP, Marghoob A. High-dynamic-range dermoscopy imaging and diagnosis of hypopigmented skin cancers. JAMA Dermatol. 2015;151:456-457.
- Quigley EA, Tokay BA, Jewell ST, et al. Technology and technique standards for camera-acquired digital dermatologic images: a systematic review. JAMA Dermatol. 2015;151:883-890.
- Katragadda C, Finnane A, Soyer HP, et al. Technique standards for skin lesion imaging a delphi consensus statement. JAMA Dermatol. 2017;153:207-213.
- Caffery LJ, Clunie D, Curiel-Lewandrowski C, et al. Transforming dermatologic imaging for the digital era: metadata and standards [published online January 17, 2018]. J Digit Imaging. doi:10.1007/s10278-017-0045-8.
- Pagliarello C, Stanganelli I, Fabrizi G, et al. Digital dermoscopy monitoring: is it time to define a quality standard? Acta Derm Venereol. 2017;97:864-865.
- HITECH Act Enforcement Interim Final Rule. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/special-topics/hitech-act-enforcement-interim-final-rule/index.html. Updated June 16, 2017. Accessed July 2, 2018.
- Guidance to render unsecured protected health information unusable, unreadable, or indecipherable to unauthorized individuals. US Department of Health & Human Services website. https://www.hhs.gov/hipaa/for-professionals/breach-notification/guidance/index.html. Updated July 26, 2013. Accessed July 2, 2018.
- Scarfone K, Souppaya M, Sexton M. Guide to Storage Encryption Technologies for End User Devices. Gaithersburg, MD: US Department of Commerce; 2007. NIST Special Publication 800-111.
SPOTme addresses unmet need for skin cancer screening
Almost half of the individuals diagnosed with melanoma in a free skin cancer screening program otherwise would not have gone to a doctor to have their skin examined, according to an analysis of the American Academy of Dermatology’s national skin cancer screening program, during 1986-2014.
The SPOTme program, a national skin cancer screening and education program conducted by volunteer dermatologists, was launched in 1985. More than 2 million free screenings have been provided by the program in a “predominantly high-risk population, rendering important clinical diagnoses for hundreds of thousands of participants,” according to first authors Jean-Phillip Okhovat, MD, of Beth Israel Deaconess Medical Center, and Derek Beaulieu, MD, of Tufts University, both in Boston, and their colleagues.
The analysis was published online in the Journal of the American Academy of Dermatology on July 26.
Their study analyzed data on almost 2 million people screened through the program from 1986-2014. About 62% were women; 90% were white, about 2% were black, and almost 4% were Hispanic. Almost 80% had no regular dermatologist, almost 73% had not been screened previously, almost 45% had never had a skin cancer check, and 9% were uninsured. Almost 31% reported a mole that had recently change in size, color, or shape; almost 34% said they had a family history of skin cancer, and about 14% said they had a personal history of skin cancer.
Participants were asked about demographics and risk factors, although some questions changed from year to year (for example, in 2009 and 2010, participants were asked about melanoma risk factors, and from 1992 through 2010, participants were asked about their access to dermatologic care).
During 1991-2014 (which did not include data for 1995, 1996, and 2000, which were not available), the screening program resulted in 20,628 clinical melanoma diagnoses, 156,087 clinical dysplastic nevi diagnoses, 32,893 clinical squamous cell carcinoma diagnoses, and 129,848 clinical basal cell carcinoma diagnoses.
Of those clinically diagnosed with melanoma during 1992-2010, 83% said they did not have a regular dermatologist, 77% said they had not been screened previously, and 47% said they would not have seen a doctor for a skin exam if the SPOTme program had not been available.
Of those screened in 2009 and 2010 , 72% were considered at high risk for melanoma (older than age 65 years, having a history of sunburns, a family history of skin cancer, and/or more than 50 moles or unusual moles).
Among the other findings was that from 1992 to 2010, about 12% of those with a clinical melanoma diagnosis were not insured, which increased over time, from almost 11% during 1992-1999 to almost 16% during 2007-2010.
The “consistently high rates” of multiple skin cancer risk factors among those newly screened in the study are consistent with previously reported data, “suggesting that there is an untapped pool of at-risk Americans who have yet to be screened for skin cancer,” the authors wrote. “While the SPOTme program cannot be expected to meet the demands of this larger at-risk population, increased publicity and educational campaigns led by the AAD and assistance to primary care physicians in triaging of patients who should be seen by dermatologists could decrease the number of Americans who need to be screened,” they added.
Limitations of the study included the inability to confirm the clinical diagnoses with histopathology, and no data from the providers were available.
The authors had no disclosures. SPOTme, part of the AAD’s SPOT Skin Cancer initiative, is supported by a grant from Bristol-Myers Squibb.
SOURCE: Okhovat JP et al. J Am Acad Dermatol. https://doi./org/10.1016.j.jaad.2018.05.1242.
Almost half of the individuals diagnosed with melanoma in a free skin cancer screening program otherwise would not have gone to a doctor to have their skin examined, according to an analysis of the American Academy of Dermatology’s national skin cancer screening program, during 1986-2014.
The SPOTme program, a national skin cancer screening and education program conducted by volunteer dermatologists, was launched in 1985. More than 2 million free screenings have been provided by the program in a “predominantly high-risk population, rendering important clinical diagnoses for hundreds of thousands of participants,” according to first authors Jean-Phillip Okhovat, MD, of Beth Israel Deaconess Medical Center, and Derek Beaulieu, MD, of Tufts University, both in Boston, and their colleagues.
The analysis was published online in the Journal of the American Academy of Dermatology on July 26.
Their study analyzed data on almost 2 million people screened through the program from 1986-2014. About 62% were women; 90% were white, about 2% were black, and almost 4% were Hispanic. Almost 80% had no regular dermatologist, almost 73% had not been screened previously, almost 45% had never had a skin cancer check, and 9% were uninsured. Almost 31% reported a mole that had recently change in size, color, or shape; almost 34% said they had a family history of skin cancer, and about 14% said they had a personal history of skin cancer.
Participants were asked about demographics and risk factors, although some questions changed from year to year (for example, in 2009 and 2010, participants were asked about melanoma risk factors, and from 1992 through 2010, participants were asked about their access to dermatologic care).
During 1991-2014 (which did not include data for 1995, 1996, and 2000, which were not available), the screening program resulted in 20,628 clinical melanoma diagnoses, 156,087 clinical dysplastic nevi diagnoses, 32,893 clinical squamous cell carcinoma diagnoses, and 129,848 clinical basal cell carcinoma diagnoses.
Of those clinically diagnosed with melanoma during 1992-2010, 83% said they did not have a regular dermatologist, 77% said they had not been screened previously, and 47% said they would not have seen a doctor for a skin exam if the SPOTme program had not been available.
Of those screened in 2009 and 2010 , 72% were considered at high risk for melanoma (older than age 65 years, having a history of sunburns, a family history of skin cancer, and/or more than 50 moles or unusual moles).
Among the other findings was that from 1992 to 2010, about 12% of those with a clinical melanoma diagnosis were not insured, which increased over time, from almost 11% during 1992-1999 to almost 16% during 2007-2010.
The “consistently high rates” of multiple skin cancer risk factors among those newly screened in the study are consistent with previously reported data, “suggesting that there is an untapped pool of at-risk Americans who have yet to be screened for skin cancer,” the authors wrote. “While the SPOTme program cannot be expected to meet the demands of this larger at-risk population, increased publicity and educational campaigns led by the AAD and assistance to primary care physicians in triaging of patients who should be seen by dermatologists could decrease the number of Americans who need to be screened,” they added.
Limitations of the study included the inability to confirm the clinical diagnoses with histopathology, and no data from the providers were available.
The authors had no disclosures. SPOTme, part of the AAD’s SPOT Skin Cancer initiative, is supported by a grant from Bristol-Myers Squibb.
SOURCE: Okhovat JP et al. J Am Acad Dermatol. https://doi./org/10.1016.j.jaad.2018.05.1242.
Almost half of the individuals diagnosed with melanoma in a free skin cancer screening program otherwise would not have gone to a doctor to have their skin examined, according to an analysis of the American Academy of Dermatology’s national skin cancer screening program, during 1986-2014.
The SPOTme program, a national skin cancer screening and education program conducted by volunteer dermatologists, was launched in 1985. More than 2 million free screenings have been provided by the program in a “predominantly high-risk population, rendering important clinical diagnoses for hundreds of thousands of participants,” according to first authors Jean-Phillip Okhovat, MD, of Beth Israel Deaconess Medical Center, and Derek Beaulieu, MD, of Tufts University, both in Boston, and their colleagues.
The analysis was published online in the Journal of the American Academy of Dermatology on July 26.
Their study analyzed data on almost 2 million people screened through the program from 1986-2014. About 62% were women; 90% were white, about 2% were black, and almost 4% were Hispanic. Almost 80% had no regular dermatologist, almost 73% had not been screened previously, almost 45% had never had a skin cancer check, and 9% were uninsured. Almost 31% reported a mole that had recently change in size, color, or shape; almost 34% said they had a family history of skin cancer, and about 14% said they had a personal history of skin cancer.
Participants were asked about demographics and risk factors, although some questions changed from year to year (for example, in 2009 and 2010, participants were asked about melanoma risk factors, and from 1992 through 2010, participants were asked about their access to dermatologic care).
During 1991-2014 (which did not include data for 1995, 1996, and 2000, which were not available), the screening program resulted in 20,628 clinical melanoma diagnoses, 156,087 clinical dysplastic nevi diagnoses, 32,893 clinical squamous cell carcinoma diagnoses, and 129,848 clinical basal cell carcinoma diagnoses.
Of those clinically diagnosed with melanoma during 1992-2010, 83% said they did not have a regular dermatologist, 77% said they had not been screened previously, and 47% said they would not have seen a doctor for a skin exam if the SPOTme program had not been available.
Of those screened in 2009 and 2010 , 72% were considered at high risk for melanoma (older than age 65 years, having a history of sunburns, a family history of skin cancer, and/or more than 50 moles or unusual moles).
Among the other findings was that from 1992 to 2010, about 12% of those with a clinical melanoma diagnosis were not insured, which increased over time, from almost 11% during 1992-1999 to almost 16% during 2007-2010.
The “consistently high rates” of multiple skin cancer risk factors among those newly screened in the study are consistent with previously reported data, “suggesting that there is an untapped pool of at-risk Americans who have yet to be screened for skin cancer,” the authors wrote. “While the SPOTme program cannot be expected to meet the demands of this larger at-risk population, increased publicity and educational campaigns led by the AAD and assistance to primary care physicians in triaging of patients who should be seen by dermatologists could decrease the number of Americans who need to be screened,” they added.
Limitations of the study included the inability to confirm the clinical diagnoses with histopathology, and no data from the providers were available.
The authors had no disclosures. SPOTme, part of the AAD’s SPOT Skin Cancer initiative, is supported by a grant from Bristol-Myers Squibb.
SOURCE: Okhovat JP et al. J Am Acad Dermatol. https://doi./org/10.1016.j.jaad.2018.05.1242.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Free skin cancer screening programs help meet an unmet need for people at high risk for skin cancer.
Major finding: Of those who received a clinical diagnosis of melanoma during 1992-2010, 47% said they would not have seen a doctor for a skin exam if the free program had not been available.
Study details: The study analyzed data on almost 2 million people screened through the free SPOTme skin cancer screening program during 1986-2014.
Disclosures: The authors had no disclosures. SPOTme, part of the AAD’s SPOT Skin Cancer initiative, is supported by a grant from Bristol-Myers Squibb.
Source: Okhovat JP et al. J Am Acad Dermatol. https://doi./org/10.1016.j.jaad.2018.05.1242.