User login
Melanoma survival shorter in those given high dose glucocorticoids for ipilimumab-induced hypophysitis
than did patients taking low-dose steroids for the adverse event, according to a new retrospective analysis in Cancer.
“Treatment with high-dose glucocorticoids does not appear to confer any obvious advantage to patients with IH (ipilimumab-induced hypophysitis) and may negatively affect tumor response to CPI (checkpoint-inhibitor therapy),” wrote Alexander Faje, MD, a neuroendocrinologist at Massachusetts General Hospital, Boston. “We recommend against the routine use of higher doses in these patients and that such treatment should be reserved for clinical indications like visual compromise or perhaps for intractable headache.”
Hypophysitis after treatment with a CTLA-4 inhibitor, such as ipilimumab, can approach 12%, though it is much less common with the checkpoint inhibitors that target PD-1 and PD-L1. Past studies examining the effects of glucocorticoids for immune-related adverse events have compared patients with severe events to those with minimal or no events. Since emergence of hypophysitis correlates with better overall survival, this is a flawed approach, the researchers said.
For their study, the researchers compared groups of patients with the same immune-related adverse events who received treatment with varying amounts of glucocorticoids.
They reviewed outcomes for 64 melanoma patients who had received ipilimumab monotherapy and were diagnosed with ipilimumab-induced hypophysitis treated in the Partners Healthcare system. Fourteen patients had received low-dose glucocorticoids, defined as a maximum average daily dose of 7.5 mg of prednisone or the equivalent. Fifty patients received high-dose glucocorticoids, defined as anything above that amount.
Overall survival and time to treatment failure were significantly higher in the low-dose group than the high-dose group (P = .002 for OS and P = .001 for TTF). Median overall survival was 23.3 months and time to treatment failure was 11.4 months in those given high-dose steroids. Median overall survival had not been reached in those given low-dose steroids.
While the findings are preliminary, the authors noted they may have implications for managing other immune-related adverse events. “Although the use of lower doses of immunosuppressive medications may be less of an option in many circumstances for other (immune-related adverse events), therapeutic parsimony would seem desirable with more tailored regimens as the biologic mechanisms underpinning these processes are further elucidated.”
SOURCE: Faje AT et al. Cancer. 2018 Jul 5.
The study results provide further evidence that hypophysitis appears to be an adverse effect of ipilimumab therapy that is linked with improved outcomes in melanoma. As hypophysitis tends to be self-limited, it can be treated safely with replacement therapy rather than high-dose steroid therapy, which was associated with reduced overall survival.
But the low number of patients on low-dose glucocorticoids – just 14 – is a limitation of the study and the group’s favorable outcomes could have been due to chance alone. Further, the 7.5-mg cut-off for high- vs. low-dose steroid therapy is somewhat arbitrary.
In support of the study’s overall conclusions, however, exploratory analyses have produced similar findings at somewhat higher cut-offs.
As the mechanism of hypophysitis is somewhat distinct compared with other immune-checkpoint toxicities, it might not be appropriate to generalize these findings to other toxic responses to these drugs.
The mechanisms of toxicities related to immune-checkpoint inhibitors are still not well defined. Unraveling those mechanisms may identify patients at high risk and hold the potential for aiding in the design of novel therapeutics that unleash antitumor immunity. Glucocorticoids are a fairly effective treatment for immune-related toxicities but remain a blunt, nonspecific way to suppress aberrant immunity. Designing inhibitors of culprit cellular populations or cytokines may combat toxicity without compromising the efficacy of immune therapy or promoting systemic immunosuppression.
Douglas B. Johnson, MD, is with Vanderbilt University Medical Center and Vanderbilt Ingram Cancer Center, Nashville, Tenn. He made his remarks in an editorial (Cancer 2018 Jul 5. doi: 10.1002/cncr.31627.
The study results provide further evidence that hypophysitis appears to be an adverse effect of ipilimumab therapy that is linked with improved outcomes in melanoma. As hypophysitis tends to be self-limited, it can be treated safely with replacement therapy rather than high-dose steroid therapy, which was associated with reduced overall survival.
But the low number of patients on low-dose glucocorticoids – just 14 – is a limitation of the study and the group’s favorable outcomes could have been due to chance alone. Further, the 7.5-mg cut-off for high- vs. low-dose steroid therapy is somewhat arbitrary.
In support of the study’s overall conclusions, however, exploratory analyses have produced similar findings at somewhat higher cut-offs.
As the mechanism of hypophysitis is somewhat distinct compared with other immune-checkpoint toxicities, it might not be appropriate to generalize these findings to other toxic responses to these drugs.
The mechanisms of toxicities related to immune-checkpoint inhibitors are still not well defined. Unraveling those mechanisms may identify patients at high risk and hold the potential for aiding in the design of novel therapeutics that unleash antitumor immunity. Glucocorticoids are a fairly effective treatment for immune-related toxicities but remain a blunt, nonspecific way to suppress aberrant immunity. Designing inhibitors of culprit cellular populations or cytokines may combat toxicity without compromising the efficacy of immune therapy or promoting systemic immunosuppression.
Douglas B. Johnson, MD, is with Vanderbilt University Medical Center and Vanderbilt Ingram Cancer Center, Nashville, Tenn. He made his remarks in an editorial (Cancer 2018 Jul 5. doi: 10.1002/cncr.31627.
The study results provide further evidence that hypophysitis appears to be an adverse effect of ipilimumab therapy that is linked with improved outcomes in melanoma. As hypophysitis tends to be self-limited, it can be treated safely with replacement therapy rather than high-dose steroid therapy, which was associated with reduced overall survival.
But the low number of patients on low-dose glucocorticoids – just 14 – is a limitation of the study and the group’s favorable outcomes could have been due to chance alone. Further, the 7.5-mg cut-off for high- vs. low-dose steroid therapy is somewhat arbitrary.
In support of the study’s overall conclusions, however, exploratory analyses have produced similar findings at somewhat higher cut-offs.
As the mechanism of hypophysitis is somewhat distinct compared with other immune-checkpoint toxicities, it might not be appropriate to generalize these findings to other toxic responses to these drugs.
The mechanisms of toxicities related to immune-checkpoint inhibitors are still not well defined. Unraveling those mechanisms may identify patients at high risk and hold the potential for aiding in the design of novel therapeutics that unleash antitumor immunity. Glucocorticoids are a fairly effective treatment for immune-related toxicities but remain a blunt, nonspecific way to suppress aberrant immunity. Designing inhibitors of culprit cellular populations or cytokines may combat toxicity without compromising the efficacy of immune therapy or promoting systemic immunosuppression.
Douglas B. Johnson, MD, is with Vanderbilt University Medical Center and Vanderbilt Ingram Cancer Center, Nashville, Tenn. He made his remarks in an editorial (Cancer 2018 Jul 5. doi: 10.1002/cncr.31627.
than did patients taking low-dose steroids for the adverse event, according to a new retrospective analysis in Cancer.
“Treatment with high-dose glucocorticoids does not appear to confer any obvious advantage to patients with IH (ipilimumab-induced hypophysitis) and may negatively affect tumor response to CPI (checkpoint-inhibitor therapy),” wrote Alexander Faje, MD, a neuroendocrinologist at Massachusetts General Hospital, Boston. “We recommend against the routine use of higher doses in these patients and that such treatment should be reserved for clinical indications like visual compromise or perhaps for intractable headache.”
Hypophysitis after treatment with a CTLA-4 inhibitor, such as ipilimumab, can approach 12%, though it is much less common with the checkpoint inhibitors that target PD-1 and PD-L1. Past studies examining the effects of glucocorticoids for immune-related adverse events have compared patients with severe events to those with minimal or no events. Since emergence of hypophysitis correlates with better overall survival, this is a flawed approach, the researchers said.
For their study, the researchers compared groups of patients with the same immune-related adverse events who received treatment with varying amounts of glucocorticoids.
They reviewed outcomes for 64 melanoma patients who had received ipilimumab monotherapy and were diagnosed with ipilimumab-induced hypophysitis treated in the Partners Healthcare system. Fourteen patients had received low-dose glucocorticoids, defined as a maximum average daily dose of 7.5 mg of prednisone or the equivalent. Fifty patients received high-dose glucocorticoids, defined as anything above that amount.
Overall survival and time to treatment failure were significantly higher in the low-dose group than the high-dose group (P = .002 for OS and P = .001 for TTF). Median overall survival was 23.3 months and time to treatment failure was 11.4 months in those given high-dose steroids. Median overall survival had not been reached in those given low-dose steroids.
While the findings are preliminary, the authors noted they may have implications for managing other immune-related adverse events. “Although the use of lower doses of immunosuppressive medications may be less of an option in many circumstances for other (immune-related adverse events), therapeutic parsimony would seem desirable with more tailored regimens as the biologic mechanisms underpinning these processes are further elucidated.”
SOURCE: Faje AT et al. Cancer. 2018 Jul 5.
than did patients taking low-dose steroids for the adverse event, according to a new retrospective analysis in Cancer.
“Treatment with high-dose glucocorticoids does not appear to confer any obvious advantage to patients with IH (ipilimumab-induced hypophysitis) and may negatively affect tumor response to CPI (checkpoint-inhibitor therapy),” wrote Alexander Faje, MD, a neuroendocrinologist at Massachusetts General Hospital, Boston. “We recommend against the routine use of higher doses in these patients and that such treatment should be reserved for clinical indications like visual compromise or perhaps for intractable headache.”
Hypophysitis after treatment with a CTLA-4 inhibitor, such as ipilimumab, can approach 12%, though it is much less common with the checkpoint inhibitors that target PD-1 and PD-L1. Past studies examining the effects of glucocorticoids for immune-related adverse events have compared patients with severe events to those with minimal or no events. Since emergence of hypophysitis correlates with better overall survival, this is a flawed approach, the researchers said.
For their study, the researchers compared groups of patients with the same immune-related adverse events who received treatment with varying amounts of glucocorticoids.
They reviewed outcomes for 64 melanoma patients who had received ipilimumab monotherapy and were diagnosed with ipilimumab-induced hypophysitis treated in the Partners Healthcare system. Fourteen patients had received low-dose glucocorticoids, defined as a maximum average daily dose of 7.5 mg of prednisone or the equivalent. Fifty patients received high-dose glucocorticoids, defined as anything above that amount.
Overall survival and time to treatment failure were significantly higher in the low-dose group than the high-dose group (P = .002 for OS and P = .001 for TTF). Median overall survival was 23.3 months and time to treatment failure was 11.4 months in those given high-dose steroids. Median overall survival had not been reached in those given low-dose steroids.
While the findings are preliminary, the authors noted they may have implications for managing other immune-related adverse events. “Although the use of lower doses of immunosuppressive medications may be less of an option in many circumstances for other (immune-related adverse events), therapeutic parsimony would seem desirable with more tailored regimens as the biologic mechanisms underpinning these processes are further elucidated.”
SOURCE: Faje AT et al. Cancer. 2018 Jul 5.
FROM CANCER
Key clinical point: Significantly lower overall survival and shorter time to treatment failure was seen in melanoma patients taking higher doses of glucocorticoids for ipilimumab-induced hypophysitis than in those taking lower doses.
Major finding: Median overall survival was 23.3 months and time to treatment failure was 11.4 months in those given high-dose steroids. Median overall survival had not been reached in those given low-dose steroids.
Study details: A retrospective review of 64 melanoma patients on single-agent ipilimumab therapy who were given glucocorticoids for ipilimumab-induced hypophysitis.
Disclosures: No funding source disclosed. The authors made no disclosures related to the submitted work.
Source: Faje AT et al. Cancer 2018 Jul 5.
Immunotherapies extend survival for melanoma patients with brain metastases
Since the Food and Drug Administration first approved checkpoint blockade immunotherapy (CBI) and BRAFV600-targeted therapy in 2011, survival times for patients with melanoma brain metastases (MBMs) have significantly improved, with a 91% increase in 4-year overall survival (OS) from 7.4% to 14.1%.
“The management of advanced melanoma has traditionally been tempered by limited responses to conventional therapies, resulting in a median overall survival (OS) of less than 1 year,” wrote J. Bryan Iorgulescu, MD, of Brigham and Women’s Hospital in Boston, and his colleagues. The report was published in Cancer Immunology Research. “The landscape of advanced melanoma treatment was revolutionized” by the approval of immunotherapy agents, beginning in 2011.
The current, retrospective study involved 2,753 patients with stage IV melanoma. Patient data were drawn from the National Cancer Database, with diagnoses made between 2010 and 2015. Patient management, overall survival, and disease characteristics were evaluated.
During initial review, the researchers found that 35.8% of patients with stage IV melanoma had brain involvement. These patients were further categorized by those with MBM only (39.7%) versus those with extracranial metastatic disease (60.3%), which included involvement of lung (82.9%), liver (8.1%), bone (6.0%), and lymph nodes or distant subcutaneous skin (3%). MBM-only disease was independently predicted by both younger age and geographic location.
Patients receiving first-line CBI therapy demonstrated improved 4-year OS (28.1% vs. 11.1%; P less than.001) and median OS (12.4 months vs. 5.2 months; P less than .001).
Improvements with CBI were most dramatic in patients with MBM-only disease. In these cases, 4-year OS improved from 16.9% to 51.5% (P less than .001), while median OS jumped from 7.7 months to 56.4 months (P less than .001).
Improved OS was also associated with fewer comorbidities, younger age, management at an academic cancer center, single-fraction stereotactic radiosurgery, and resection of the MBM.
“Our findings help bridge the gaps in early clinical trials of CBIs that largely excluded stage IV melanoma patients with MBMs, with checkpoint immunotherapy demonstrating a more than doubling of the median and 4-year OS of MBMs,” the authors concluded.
SOURCE: Iorgulescu et al. Cancer Immunol Res. 2018 July 12 doi: 10.1158/2326-6066.CIR-18-0067.
Since the Food and Drug Administration first approved checkpoint blockade immunotherapy (CBI) and BRAFV600-targeted therapy in 2011, survival times for patients with melanoma brain metastases (MBMs) have significantly improved, with a 91% increase in 4-year overall survival (OS) from 7.4% to 14.1%.
“The management of advanced melanoma has traditionally been tempered by limited responses to conventional therapies, resulting in a median overall survival (OS) of less than 1 year,” wrote J. Bryan Iorgulescu, MD, of Brigham and Women’s Hospital in Boston, and his colleagues. The report was published in Cancer Immunology Research. “The landscape of advanced melanoma treatment was revolutionized” by the approval of immunotherapy agents, beginning in 2011.
The current, retrospective study involved 2,753 patients with stage IV melanoma. Patient data were drawn from the National Cancer Database, with diagnoses made between 2010 and 2015. Patient management, overall survival, and disease characteristics were evaluated.
During initial review, the researchers found that 35.8% of patients with stage IV melanoma had brain involvement. These patients were further categorized by those with MBM only (39.7%) versus those with extracranial metastatic disease (60.3%), which included involvement of lung (82.9%), liver (8.1%), bone (6.0%), and lymph nodes or distant subcutaneous skin (3%). MBM-only disease was independently predicted by both younger age and geographic location.
Patients receiving first-line CBI therapy demonstrated improved 4-year OS (28.1% vs. 11.1%; P less than.001) and median OS (12.4 months vs. 5.2 months; P less than .001).
Improvements with CBI were most dramatic in patients with MBM-only disease. In these cases, 4-year OS improved from 16.9% to 51.5% (P less than .001), while median OS jumped from 7.7 months to 56.4 months (P less than .001).
Improved OS was also associated with fewer comorbidities, younger age, management at an academic cancer center, single-fraction stereotactic radiosurgery, and resection of the MBM.
“Our findings help bridge the gaps in early clinical trials of CBIs that largely excluded stage IV melanoma patients with MBMs, with checkpoint immunotherapy demonstrating a more than doubling of the median and 4-year OS of MBMs,” the authors concluded.
SOURCE: Iorgulescu et al. Cancer Immunol Res. 2018 July 12 doi: 10.1158/2326-6066.CIR-18-0067.
Since the Food and Drug Administration first approved checkpoint blockade immunotherapy (CBI) and BRAFV600-targeted therapy in 2011, survival times for patients with melanoma brain metastases (MBMs) have significantly improved, with a 91% increase in 4-year overall survival (OS) from 7.4% to 14.1%.
“The management of advanced melanoma has traditionally been tempered by limited responses to conventional therapies, resulting in a median overall survival (OS) of less than 1 year,” wrote J. Bryan Iorgulescu, MD, of Brigham and Women’s Hospital in Boston, and his colleagues. The report was published in Cancer Immunology Research. “The landscape of advanced melanoma treatment was revolutionized” by the approval of immunotherapy agents, beginning in 2011.
The current, retrospective study involved 2,753 patients with stage IV melanoma. Patient data were drawn from the National Cancer Database, with diagnoses made between 2010 and 2015. Patient management, overall survival, and disease characteristics were evaluated.
During initial review, the researchers found that 35.8% of patients with stage IV melanoma had brain involvement. These patients were further categorized by those with MBM only (39.7%) versus those with extracranial metastatic disease (60.3%), which included involvement of lung (82.9%), liver (8.1%), bone (6.0%), and lymph nodes or distant subcutaneous skin (3%). MBM-only disease was independently predicted by both younger age and geographic location.
Patients receiving first-line CBI therapy demonstrated improved 4-year OS (28.1% vs. 11.1%; P less than.001) and median OS (12.4 months vs. 5.2 months; P less than .001).
Improvements with CBI were most dramatic in patients with MBM-only disease. In these cases, 4-year OS improved from 16.9% to 51.5% (P less than .001), while median OS jumped from 7.7 months to 56.4 months (P less than .001).
Improved OS was also associated with fewer comorbidities, younger age, management at an academic cancer center, single-fraction stereotactic radiosurgery, and resection of the MBM.
“Our findings help bridge the gaps in early clinical trials of CBIs that largely excluded stage IV melanoma patients with MBMs, with checkpoint immunotherapy demonstrating a more than doubling of the median and 4-year OS of MBMs,” the authors concluded.
SOURCE: Iorgulescu et al. Cancer Immunol Res. 2018 July 12 doi: 10.1158/2326-6066.CIR-18-0067.
FROM CANCER IMMUNOLOGY RESEARCH
Key clinical point: Checkpoint blockade immunotherapy and BRAFV600-targeted therapy improve survival for patients with melanoma brain metastases.
Major finding: Patients with melanoma brain metastases receiving first-line checkpoint blockade immunotherapy had an improved 4-year overall survival (28.1% vs. 11.1%; P less than .001) and median overall survival (12.4 months vs. 5.2 months; P less than .001).
Study details: A retrospective study of 2,753 patients with stage IV melanoma and brain metastases, from the National Cancer Database, between 2010 and 2015.
Disclosures: The study was supported by the National Institute of Health, Abbvie, Bristol-Myers Squibb, Merck, and others. No conflicts of interest were reported.
Source: Iorgulescu et al. Cancer Immunol Res. 2018 July 12. doi: 10.1158/2326-6066.CIR-18-0067.
Sunscreen use in grade schoolers: Wide racial, ethnic disparities seen
and the figures were much lower for non-Hispanic black children.
Just 23% of fifth graders almost always used sunscreen, according to data drawn from the Healthy Passages study, which surveyed the parents or caregivers of 5,119 fifth graders. That figure was similar in the 1,802 Hispanic respondents, but fell to just 6% of the 1,748 non-Hispanic black respondents.
Some other factors that were associated with less chance of adherence to sunscreen use included being male and having lower socioeconomic status, wrote Christina M. Correnti, MD, and her study coauthors. The report was published in in Pediatric Dermatology. Perhaps surprisingly, they said, “School-based sun-safety education and involvement in team sports were not significant factors.”
Healthy Passages is a prospective multisite cohort study of child and adolescent health. Dr. Correnti, a dermatology resident at the University of Maryland, Baltimore, and her colleagues used baseline Healthy Passages data collected from the period of 2004-2006. Children enrolled in fifth grade at public schools in Birmingham, Ala., Houston, and Los Angeles, together with their caregivers, participated in the survey. Deidentified demographic data were collected, and participants were asked about four preventive health behaviors in addition to sunscreen use and flossing teeth: brushing teeth, helmet use, seatbelt use, and well-child examinations.
Dr. Correnti and her colleagues used multivariable analysis to calculate odds ratios for the association between the various demographic factors and other preventive behaviors and sunscreen use. They found that sunscreen adherence was correlated with all other preventive behaviors (P less than .001), but that the interrelationship with helmet use was confounded by racial and ethnic variables. Seatbelt use was not significantly correlated with sunscreen use for non-Hispanic black or Hispanic respondents.
“Children from more-educated and affluent households were more likely to use sun protection. Perhaps they had greater parental awareness and practice of sun safe habits,” wrote Dr. Correnti and her colleagues, noting that other work has shown that even low-income parents generally don’t see the cost of sunscreen as a barrier to use.
Although overall use of sunscreen among non-Hispanic black children was low, both non-Hispanic black and Hispanic children were more likely to use sunscreen if they had three or more sunburns within the prior 12 months. “Although darker skin tones may afford some sun protection, melanoma incidence is growing in Hispanic populations,” the researchers wrote.
To address these overall low rates of sunscreen use, the investigators discussed the utility of a variety of education options. The well-child visit affords an opportunity to reinforce the importance of preventive behaviors, but physicians may run into a time crunch and forgo thorough sun safety education, they said. Written materials can be a useful adjunct for clinicians in this setting.
“Health care practitioners may use absence of other preventive behaviors as potential markers for inadequate sunscreen use, prompting a point-of-care sun-safety intervention,” they suggested.
A school-based public health approach offers another route for education. “School sun-safety programs may alleviate the primary care burden,” wrote Dr. Correnti and her coinvestigators. The opportunity to deliver repeated, age-tailored messages as children progress through school may be effective in promoting healthy sun behaviors. Messaging that focuses on the negative effects of sun exposure on appearance such as age spots and wrinkles have been more effective than those warning of the risk of skin cancer for teens; investigating appearance-based content for this age group might be a good idea, the authors said.
The fact that the survey sites were in southern cities may mean that national rates of consistent sunscreen use for elementary schoolers may be even lower, said Dr. Correnti and her coauthors. Many other real-world factors, such as frequency and amount of sunscreen applied and the use of sun-protective clothing, couldn’t be captured by the survey, they acknowledged.
“Even in the most adherent group, non-Hispanic whites, only 44.8% always used sunscreen,” the researchers wrote. The study’s findings leave plenty of room for implementation of broad-based programs, especially in low-resource communities.
The National Institutes of Health funded the research. Dr. Correnti was supported by NIH awards.
SOURCE: Correnti CM et al. Pediatr Dermatol. 2018. doi: 10.1111/pde.13550.
and the figures were much lower for non-Hispanic black children.
Just 23% of fifth graders almost always used sunscreen, according to data drawn from the Healthy Passages study, which surveyed the parents or caregivers of 5,119 fifth graders. That figure was similar in the 1,802 Hispanic respondents, but fell to just 6% of the 1,748 non-Hispanic black respondents.
Some other factors that were associated with less chance of adherence to sunscreen use included being male and having lower socioeconomic status, wrote Christina M. Correnti, MD, and her study coauthors. The report was published in in Pediatric Dermatology. Perhaps surprisingly, they said, “School-based sun-safety education and involvement in team sports were not significant factors.”
Healthy Passages is a prospective multisite cohort study of child and adolescent health. Dr. Correnti, a dermatology resident at the University of Maryland, Baltimore, and her colleagues used baseline Healthy Passages data collected from the period of 2004-2006. Children enrolled in fifth grade at public schools in Birmingham, Ala., Houston, and Los Angeles, together with their caregivers, participated in the survey. Deidentified demographic data were collected, and participants were asked about four preventive health behaviors in addition to sunscreen use and flossing teeth: brushing teeth, helmet use, seatbelt use, and well-child examinations.
Dr. Correnti and her colleagues used multivariable analysis to calculate odds ratios for the association between the various demographic factors and other preventive behaviors and sunscreen use. They found that sunscreen adherence was correlated with all other preventive behaviors (P less than .001), but that the interrelationship with helmet use was confounded by racial and ethnic variables. Seatbelt use was not significantly correlated with sunscreen use for non-Hispanic black or Hispanic respondents.
“Children from more-educated and affluent households were more likely to use sun protection. Perhaps they had greater parental awareness and practice of sun safe habits,” wrote Dr. Correnti and her colleagues, noting that other work has shown that even low-income parents generally don’t see the cost of sunscreen as a barrier to use.
Although overall use of sunscreen among non-Hispanic black children was low, both non-Hispanic black and Hispanic children were more likely to use sunscreen if they had three or more sunburns within the prior 12 months. “Although darker skin tones may afford some sun protection, melanoma incidence is growing in Hispanic populations,” the researchers wrote.
To address these overall low rates of sunscreen use, the investigators discussed the utility of a variety of education options. The well-child visit affords an opportunity to reinforce the importance of preventive behaviors, but physicians may run into a time crunch and forgo thorough sun safety education, they said. Written materials can be a useful adjunct for clinicians in this setting.
“Health care practitioners may use absence of other preventive behaviors as potential markers for inadequate sunscreen use, prompting a point-of-care sun-safety intervention,” they suggested.
A school-based public health approach offers another route for education. “School sun-safety programs may alleviate the primary care burden,” wrote Dr. Correnti and her coinvestigators. The opportunity to deliver repeated, age-tailored messages as children progress through school may be effective in promoting healthy sun behaviors. Messaging that focuses on the negative effects of sun exposure on appearance such as age spots and wrinkles have been more effective than those warning of the risk of skin cancer for teens; investigating appearance-based content for this age group might be a good idea, the authors said.
The fact that the survey sites were in southern cities may mean that national rates of consistent sunscreen use for elementary schoolers may be even lower, said Dr. Correnti and her coauthors. Many other real-world factors, such as frequency and amount of sunscreen applied and the use of sun-protective clothing, couldn’t be captured by the survey, they acknowledged.
“Even in the most adherent group, non-Hispanic whites, only 44.8% always used sunscreen,” the researchers wrote. The study’s findings leave plenty of room for implementation of broad-based programs, especially in low-resource communities.
The National Institutes of Health funded the research. Dr. Correnti was supported by NIH awards.
SOURCE: Correnti CM et al. Pediatr Dermatol. 2018. doi: 10.1111/pde.13550.
and the figures were much lower for non-Hispanic black children.
Just 23% of fifth graders almost always used sunscreen, according to data drawn from the Healthy Passages study, which surveyed the parents or caregivers of 5,119 fifth graders. That figure was similar in the 1,802 Hispanic respondents, but fell to just 6% of the 1,748 non-Hispanic black respondents.
Some other factors that were associated with less chance of adherence to sunscreen use included being male and having lower socioeconomic status, wrote Christina M. Correnti, MD, and her study coauthors. The report was published in in Pediatric Dermatology. Perhaps surprisingly, they said, “School-based sun-safety education and involvement in team sports were not significant factors.”
Healthy Passages is a prospective multisite cohort study of child and adolescent health. Dr. Correnti, a dermatology resident at the University of Maryland, Baltimore, and her colleagues used baseline Healthy Passages data collected from the period of 2004-2006. Children enrolled in fifth grade at public schools in Birmingham, Ala., Houston, and Los Angeles, together with their caregivers, participated in the survey. Deidentified demographic data were collected, and participants were asked about four preventive health behaviors in addition to sunscreen use and flossing teeth: brushing teeth, helmet use, seatbelt use, and well-child examinations.
Dr. Correnti and her colleagues used multivariable analysis to calculate odds ratios for the association between the various demographic factors and other preventive behaviors and sunscreen use. They found that sunscreen adherence was correlated with all other preventive behaviors (P less than .001), but that the interrelationship with helmet use was confounded by racial and ethnic variables. Seatbelt use was not significantly correlated with sunscreen use for non-Hispanic black or Hispanic respondents.
“Children from more-educated and affluent households were more likely to use sun protection. Perhaps they had greater parental awareness and practice of sun safe habits,” wrote Dr. Correnti and her colleagues, noting that other work has shown that even low-income parents generally don’t see the cost of sunscreen as a barrier to use.
Although overall use of sunscreen among non-Hispanic black children was low, both non-Hispanic black and Hispanic children were more likely to use sunscreen if they had three or more sunburns within the prior 12 months. “Although darker skin tones may afford some sun protection, melanoma incidence is growing in Hispanic populations,” the researchers wrote.
To address these overall low rates of sunscreen use, the investigators discussed the utility of a variety of education options. The well-child visit affords an opportunity to reinforce the importance of preventive behaviors, but physicians may run into a time crunch and forgo thorough sun safety education, they said. Written materials can be a useful adjunct for clinicians in this setting.
“Health care practitioners may use absence of other preventive behaviors as potential markers for inadequate sunscreen use, prompting a point-of-care sun-safety intervention,” they suggested.
A school-based public health approach offers another route for education. “School sun-safety programs may alleviate the primary care burden,” wrote Dr. Correnti and her coinvestigators. The opportunity to deliver repeated, age-tailored messages as children progress through school may be effective in promoting healthy sun behaviors. Messaging that focuses on the negative effects of sun exposure on appearance such as age spots and wrinkles have been more effective than those warning of the risk of skin cancer for teens; investigating appearance-based content for this age group might be a good idea, the authors said.
The fact that the survey sites were in southern cities may mean that national rates of consistent sunscreen use for elementary schoolers may be even lower, said Dr. Correnti and her coauthors. Many other real-world factors, such as frequency and amount of sunscreen applied and the use of sun-protective clothing, couldn’t be captured by the survey, they acknowledged.
“Even in the most adherent group, non-Hispanic whites, only 44.8% always used sunscreen,” the researchers wrote. The study’s findings leave plenty of room for implementation of broad-based programs, especially in low-resource communities.
The National Institutes of Health funded the research. Dr. Correnti was supported by NIH awards.
SOURCE: Correnti CM et al. Pediatr Dermatol. 2018. doi: 10.1111/pde.13550.
FROM PEDIATRIC DERMATOLOGY
Key clinical point: Most parents surveyed said their children didn’t use sunscreen consistently.
Major finding: Of non-Hispanic black children, 6% almost always used sunscreen.
Study details: Data drawn from Healthy Passages, a prospective cohort study of 5,119 fifth-graders and their parents or caregivers.
Disclosures: The National Institutes of Health funded the research. Dr. Correnti was supported by NIH awards.
Source: Correnti C et al. Pediatr Dermatol. 2018. doi: 10.1111/pde.13550.
Clinical Pearl: Mohs Cantaloupe Analogy for the Dermatology Resident
Practice Gap
Mohs micrographic surgery (MMS) is a highly curative tissue-sparing skin cancer treatment1 and is a required component of dermatology residency training. According to the Accreditation Council for Graduate Medical Education, residents must have exposure “either through direct observation or as an assistant in Mohs micrographic surgery, and reconstruction of these defects, to include flaps and grafts.”2 The MMS technique allows for complete circumferential peripheral and deep margin assessment of excised specimens; however, the conformation of a 3-dimensional gross tissue specimen into a 2-dimensional specimen as represented on a microscope slide is challenging to conceptualize.
Behavioral science research has shown that analogies and metaphors help integrate topics into a memorable format and produce deeper comprehension.3 As such, analogies can aid in the visualization of these complex spatial concepts. The MMS tissue-processing technique has been compared to flattening a pie pan.4 More recently, a peanut butter cup analogy was described as a visualization tool for explaining the various steps of MMS to patients.5 Although these analogies may help elucidate certain aspects of the MMS technique, none adequately account for the multilayered anatomy of the skin.
The Technique
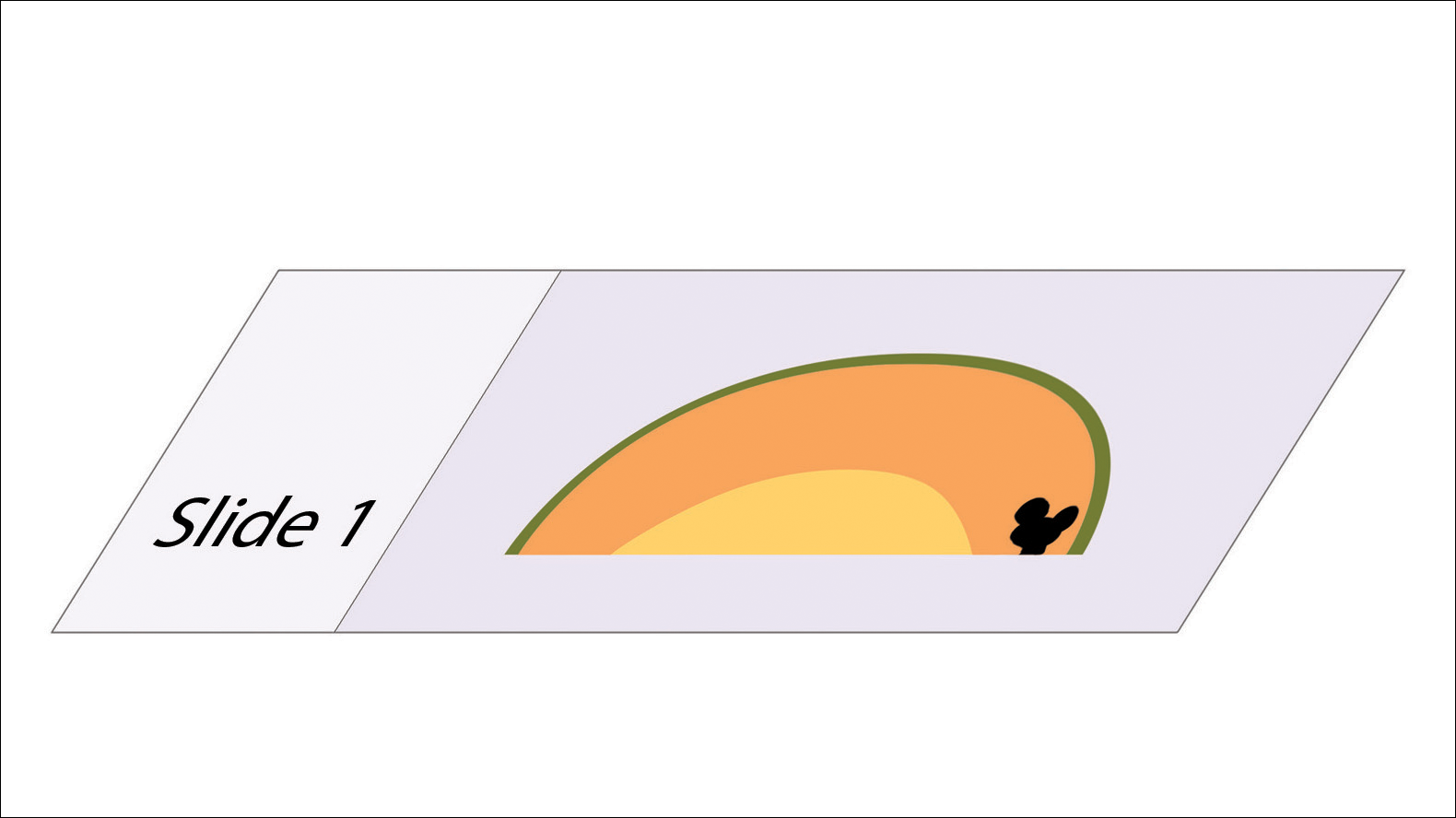
To address this need, we developed the cantaloupe analogy, which provides visual representation of the 3 basic skin layers: (1) the rind represents the epidermis; (2) the flesh represents the dermis, and (3) the seed cavity represents the subcutaneous layer (Figures 1 and 2).


In MMS tissue processing, the peripheral margin of the ovoid excised skin specimen is pressed down into the same plane as the deepest layer through a process called relaxation.4 The cantaloupe represents the dome shape of the relaxed tissue, which is then serially sectioned in horizontal layers from deep to superficial (Figure 2). The first slice represents the deepest subcutaneous layer and most peripheral dermal and epidermal layers of the specimen (Figure 3). Using the cantaloupe analogy, subsequent stages (if warranted) would be guided by the location of the residual skin cancer. If the skin cancer is in the epidermis (rind) or dermis (flesh), then a skin specimen from the perimeter of the defect would be indicated. Residual skin cancer extending into the subcutaneous layer (seed cavity) would require a deeper resection.
Practice Implications
The cantaloupe provides a simple analogy to conceptualize the transition from the multilayered 3-dimensional skin tissue specimen to the 2-dimensional histologic slide specimen. Use of this cantaloupe analogy will aid dermatology residents and others interested in gaining a clearer understanding of MMS.
- Semkova K, Mallipeddi R, Robson A, et al. Mohs micrographic surgery concordance between Mohs surgeons and dermatopathologists. Dermatol Surg. 2013;39:1648-1652.
- ACGME program requirements for graduate medical education in dermatology. Accreditation Council for Graduate Medical Education website. https://www.acgme.org/Portals/0/PFAssets/ProgramRequirements/080_dermatology_2017-07-01.pdf. Updated July 1, 2017. Accessed June 6, 2018.
- Wolfe CR. Plant a tree in cyberspace: metaphor and analogy as design elements in Web-based learning environments. CyberPsychol Behav. 2001;4:67-76.
- Beck B, Peters SR. Frozen section techniques used in Mohs micrographic surgery. In: Peters SR, ed. A Practical Guide to Frozen Section Technique. New York, NY: Springer; 2010:151-170.
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744.
Practice Gap
Mohs micrographic surgery (MMS) is a highly curative tissue-sparing skin cancer treatment1 and is a required component of dermatology residency training. According to the Accreditation Council for Graduate Medical Education, residents must have exposure “either through direct observation or as an assistant in Mohs micrographic surgery, and reconstruction of these defects, to include flaps and grafts.”2 The MMS technique allows for complete circumferential peripheral and deep margin assessment of excised specimens; however, the conformation of a 3-dimensional gross tissue specimen into a 2-dimensional specimen as represented on a microscope slide is challenging to conceptualize.
Behavioral science research has shown that analogies and metaphors help integrate topics into a memorable format and produce deeper comprehension.3 As such, analogies can aid in the visualization of these complex spatial concepts. The MMS tissue-processing technique has been compared to flattening a pie pan.4 More recently, a peanut butter cup analogy was described as a visualization tool for explaining the various steps of MMS to patients.5 Although these analogies may help elucidate certain aspects of the MMS technique, none adequately account for the multilayered anatomy of the skin.
The Technique
To address this need, we developed the cantaloupe analogy, which provides visual representation of the 3 basic skin layers: (1) the rind represents the epidermis; (2) the flesh represents the dermis, and (3) the seed cavity represents the subcutaneous layer (Figures 1 and 2).


In MMS tissue processing, the peripheral margin of the ovoid excised skin specimen is pressed down into the same plane as the deepest layer through a process called relaxation.4 The cantaloupe represents the dome shape of the relaxed tissue, which is then serially sectioned in horizontal layers from deep to superficial (Figure 2). The first slice represents the deepest subcutaneous layer and most peripheral dermal and epidermal layers of the specimen (Figure 3). Using the cantaloupe analogy, subsequent stages (if warranted) would be guided by the location of the residual skin cancer. If the skin cancer is in the epidermis (rind) or dermis (flesh), then a skin specimen from the perimeter of the defect would be indicated. Residual skin cancer extending into the subcutaneous layer (seed cavity) would require a deeper resection.

Practice Implications
The cantaloupe provides a simple analogy to conceptualize the transition from the multilayered 3-dimensional skin tissue specimen to the 2-dimensional histologic slide specimen. Use of this cantaloupe analogy will aid dermatology residents and others interested in gaining a clearer understanding of MMS.
Practice Gap
Mohs micrographic surgery (MMS) is a highly curative tissue-sparing skin cancer treatment1 and is a required component of dermatology residency training. According to the Accreditation Council for Graduate Medical Education, residents must have exposure “either through direct observation or as an assistant in Mohs micrographic surgery, and reconstruction of these defects, to include flaps and grafts.”2 The MMS technique allows for complete circumferential peripheral and deep margin assessment of excised specimens; however, the conformation of a 3-dimensional gross tissue specimen into a 2-dimensional specimen as represented on a microscope slide is challenging to conceptualize.
Behavioral science research has shown that analogies and metaphors help integrate topics into a memorable format and produce deeper comprehension.3 As such, analogies can aid in the visualization of these complex spatial concepts. The MMS tissue-processing technique has been compared to flattening a pie pan.4 More recently, a peanut butter cup analogy was described as a visualization tool for explaining the various steps of MMS to patients.5 Although these analogies may help elucidate certain aspects of the MMS technique, none adequately account for the multilayered anatomy of the skin.
The Technique
To address this need, we developed the cantaloupe analogy, which provides visual representation of the 3 basic skin layers: (1) the rind represents the epidermis; (2) the flesh represents the dermis, and (3) the seed cavity represents the subcutaneous layer (Figures 1 and 2).


In MMS tissue processing, the peripheral margin of the ovoid excised skin specimen is pressed down into the same plane as the deepest layer through a process called relaxation.4 The cantaloupe represents the dome shape of the relaxed tissue, which is then serially sectioned in horizontal layers from deep to superficial (Figure 2). The first slice represents the deepest subcutaneous layer and most peripheral dermal and epidermal layers of the specimen (Figure 3). Using the cantaloupe analogy, subsequent stages (if warranted) would be guided by the location of the residual skin cancer. If the skin cancer is in the epidermis (rind) or dermis (flesh), then a skin specimen from the perimeter of the defect would be indicated. Residual skin cancer extending into the subcutaneous layer (seed cavity) would require a deeper resection.

Practice Implications
The cantaloupe provides a simple analogy to conceptualize the transition from the multilayered 3-dimensional skin tissue specimen to the 2-dimensional histologic slide specimen. Use of this cantaloupe analogy will aid dermatology residents and others interested in gaining a clearer understanding of MMS.
- Semkova K, Mallipeddi R, Robson A, et al. Mohs micrographic surgery concordance between Mohs surgeons and dermatopathologists. Dermatol Surg. 2013;39:1648-1652.
- ACGME program requirements for graduate medical education in dermatology. Accreditation Council for Graduate Medical Education website. https://www.acgme.org/Portals/0/PFAssets/ProgramRequirements/080_dermatology_2017-07-01.pdf. Updated July 1, 2017. Accessed June 6, 2018.
- Wolfe CR. Plant a tree in cyberspace: metaphor and analogy as design elements in Web-based learning environments. CyberPsychol Behav. 2001;4:67-76.
- Beck B, Peters SR. Frozen section techniques used in Mohs micrographic surgery. In: Peters SR, ed. A Practical Guide to Frozen Section Technique. New York, NY: Springer; 2010:151-170.
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744.
- Semkova K, Mallipeddi R, Robson A, et al. Mohs micrographic surgery concordance between Mohs surgeons and dermatopathologists. Dermatol Surg. 2013;39:1648-1652.
- ACGME program requirements for graduate medical education in dermatology. Accreditation Council for Graduate Medical Education website. https://www.acgme.org/Portals/0/PFAssets/ProgramRequirements/080_dermatology_2017-07-01.pdf. Updated July 1, 2017. Accessed June 6, 2018.
- Wolfe CR. Plant a tree in cyberspace: metaphor and analogy as design elements in Web-based learning environments. CyberPsychol Behav. 2001;4:67-76.
- Beck B, Peters SR. Frozen section techniques used in Mohs micrographic surgery. In: Peters SR, ed. A Practical Guide to Frozen Section Technique. New York, NY: Springer; 2010:151-170.
- Lee E, Wolverton JE, Somani AK. A simple, effective analogy to elucidate the Mohs micrographic surgery procedure—the peanut butter cup. JAMA Dermatol. 2017;153:743-744.
FDA approves encorafenib/binimetinib for advanced melanoma with BRAF mutations
The Food and Drug Administration has approved combination therapy of encorafenib (Braftovi) and binimetinib (Mektovi) for the treatment of unresectable or metastatic melanoma with BRAF V600E or BRAF V600K mutations; the FDA also has approved the THxID BRAF Kit as a companion diagnostic for this combination therapy.
The approval was based on results from the randomized, active-controlled, open-label, multicenter COLUMBUS trial, which included 517 patients. Progression-free survival, according to RECIST 1.1 criteria, was the major efficacy measure; the median progression-free survival was 14.9 months in the encorafenib/binimetinib combination arm versus 7.3 months in the vemurafenib (Zelboraf) monotherapy arm (hazard ratio, 0.54; 95% confidence interval, 0.41-0.71; P less than .0001).
Fatigue, nausea, diarrhea, vomiting, abdominal pain, and arthralgia were the most common adverse reactions. Discontinuation of therapy from adverse reactions occurred in 5% of patients receiving the combination, the FDA said in a press statement.
The full prescribing information for encorafenib and binimetinib can be found on the FDA website.
The Food and Drug Administration has approved combination therapy of encorafenib (Braftovi) and binimetinib (Mektovi) for the treatment of unresectable or metastatic melanoma with BRAF V600E or BRAF V600K mutations; the FDA also has approved the THxID BRAF Kit as a companion diagnostic for this combination therapy.
The approval was based on results from the randomized, active-controlled, open-label, multicenter COLUMBUS trial, which included 517 patients. Progression-free survival, according to RECIST 1.1 criteria, was the major efficacy measure; the median progression-free survival was 14.9 months in the encorafenib/binimetinib combination arm versus 7.3 months in the vemurafenib (Zelboraf) monotherapy arm (hazard ratio, 0.54; 95% confidence interval, 0.41-0.71; P less than .0001).
Fatigue, nausea, diarrhea, vomiting, abdominal pain, and arthralgia were the most common adverse reactions. Discontinuation of therapy from adverse reactions occurred in 5% of patients receiving the combination, the FDA said in a press statement.
The full prescribing information for encorafenib and binimetinib can be found on the FDA website.
The Food and Drug Administration has approved combination therapy of encorafenib (Braftovi) and binimetinib (Mektovi) for the treatment of unresectable or metastatic melanoma with BRAF V600E or BRAF V600K mutations; the FDA also has approved the THxID BRAF Kit as a companion diagnostic for this combination therapy.
The approval was based on results from the randomized, active-controlled, open-label, multicenter COLUMBUS trial, which included 517 patients. Progression-free survival, according to RECIST 1.1 criteria, was the major efficacy measure; the median progression-free survival was 14.9 months in the encorafenib/binimetinib combination arm versus 7.3 months in the vemurafenib (Zelboraf) monotherapy arm (hazard ratio, 0.54; 95% confidence interval, 0.41-0.71; P less than .0001).
Fatigue, nausea, diarrhea, vomiting, abdominal pain, and arthralgia were the most common adverse reactions. Discontinuation of therapy from adverse reactions occurred in 5% of patients receiving the combination, the FDA said in a press statement.
The full prescribing information for encorafenib and binimetinib can be found on the FDA website.
Registry data provide evidence that Mohs surgery remains underutilized
CHICAGO – Analysis of U.S. national cancer registry data shows that, contrary to expectation, the , Sean Condon, MD, reported at the annual meeting of the American College of Mohs Surgery.
Ditto for the use of Mohs in patients with the rare cutaneous malignancies for which published evidence clearly demonstrates Mohs outperforms wide local excision, which is employed seven times more frequently than Mohs in such situations.
“Mohs utilization did not increase after the Affordable Care Act [ACA], despite new health insurance coverage for 20 million previously uninsured adults,” Dr. Condon said. “Surprisingly, after the ACA we actually saw a decrease in Mohs use for melanoma in situ.”
Indeed, his retrospective study of more than 25,000 patients in the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registries showed that the proportion of patients with melanoma in situ treated with Mohs declined from 13.9% during 2008-2009 – prior to ACA implementation – to 12.3% in 2011-2013, after the ACA took effect. That’s a statistically significant 13% drop, even though numerous published studies have shown outcomes in melanoma in situ are better with Mohs, said the dermatologist, who conducted the study while completing a Mohs surgery fellowship at the Cleveland Clinic. He is now in private practice in Thousand Oaks, Calif.
His analysis included 19,013 patients treated in 2008-2014 for melanoma in situ and 6,309 others treated for rare cutaneous malignancies deemed appropriate for Mohs according to the criteria formally developed jointly by the American Academy of Dermatology, the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery (J Am Acad Dermatol. 2012 Oct;67[4]:531-50). These rare malignancies include adnexal carcinoma, Merkel cell carcinoma, dermatofibrosarcoma, extramammary Paget disease, sebaceous adenocarcinoma, and leiomyosarcoma.
“These rare cutaneous malignancies were historically treated with wide local excision. However, numerous studies have lately shown that lower recurrence rates were found with Mohs compared with wide local excision,” Dr. Condon noted.
Nonetheless, the proportion of the rare cutaneous malignancies treated using Mohs was unaffected by implementation of the ACA. Nor was it influenced one way or the other by publication of the joint Mohs appropriate use criteria in 2012: The Mohs-treated proportion of such cases was 15.25% in 2010-2011 and 14.6% in 2013-2014.
Similarly, even though the appropriate use criteria identified melanoma in situ as Mohs appropriate, the proportion of those malignancies treated via Mohs was the same before and after the 2012 release of the criteria.
“It’s commonly thought that Mohs is overused. However, our study and our data clearly identify that Mohs is being underutilized for melanoma in situ and for rare cutaneous malignancies. This represents a knowledge gap for other specialties regarding best-practice therapy,” Dr. Condon said.
He and his coinvestigators searched for socioeconomic predictors of Mohs utilization by matching the nationally representative SEER data with U.S. census data. They examined the impact of three metrics: insurance status, income, and poverty. They found that low-income patients and those in the highest quartile of poverty were significantly less likely to have Mohs surgery for their melanoma in situ and rare cutaneous malignancies throughout the study years. Lack of health insurance had no impact on Mohs utilization for melanoma in situ but was independently associated with decreased likelihood of Mohs for the rare cutaneous malignancies. White patients were 2-fold to 2.4-fold more likely to have Mohs surgery for their rare cutaneous malignancies than were black patients.
“One can conclude that Mohs micrographic surgery may be skewed toward more affluent patients, and lower socioeconomic status areas have less Mohs access. So our data from this study support a role for targeted education and improved patient access to Mohs,” Dr. Condon said.
He noted that because the SEER registries don’t track squamous or basal cell carcinomas, it’s unknown whether Mohs is also underutilized for the higher-risk forms of these most common of all skin cancers.
Dr. Condon reported having no financial conflicts regarding his study, conducted free of commercial support.
CHICAGO – Analysis of U.S. national cancer registry data shows that, contrary to expectation, the , Sean Condon, MD, reported at the annual meeting of the American College of Mohs Surgery.
Ditto for the use of Mohs in patients with the rare cutaneous malignancies for which published evidence clearly demonstrates Mohs outperforms wide local excision, which is employed seven times more frequently than Mohs in such situations.
“Mohs utilization did not increase after the Affordable Care Act [ACA], despite new health insurance coverage for 20 million previously uninsured adults,” Dr. Condon said. “Surprisingly, after the ACA we actually saw a decrease in Mohs use for melanoma in situ.”
Indeed, his retrospective study of more than 25,000 patients in the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registries showed that the proportion of patients with melanoma in situ treated with Mohs declined from 13.9% during 2008-2009 – prior to ACA implementation – to 12.3% in 2011-2013, after the ACA took effect. That’s a statistically significant 13% drop, even though numerous published studies have shown outcomes in melanoma in situ are better with Mohs, said the dermatologist, who conducted the study while completing a Mohs surgery fellowship at the Cleveland Clinic. He is now in private practice in Thousand Oaks, Calif.
His analysis included 19,013 patients treated in 2008-2014 for melanoma in situ and 6,309 others treated for rare cutaneous malignancies deemed appropriate for Mohs according to the criteria formally developed jointly by the American Academy of Dermatology, the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery (J Am Acad Dermatol. 2012 Oct;67[4]:531-50). These rare malignancies include adnexal carcinoma, Merkel cell carcinoma, dermatofibrosarcoma, extramammary Paget disease, sebaceous adenocarcinoma, and leiomyosarcoma.
“These rare cutaneous malignancies were historically treated with wide local excision. However, numerous studies have lately shown that lower recurrence rates were found with Mohs compared with wide local excision,” Dr. Condon noted.
Nonetheless, the proportion of the rare cutaneous malignancies treated using Mohs was unaffected by implementation of the ACA. Nor was it influenced one way or the other by publication of the joint Mohs appropriate use criteria in 2012: The Mohs-treated proportion of such cases was 15.25% in 2010-2011 and 14.6% in 2013-2014.
Similarly, even though the appropriate use criteria identified melanoma in situ as Mohs appropriate, the proportion of those malignancies treated via Mohs was the same before and after the 2012 release of the criteria.
“It’s commonly thought that Mohs is overused. However, our study and our data clearly identify that Mohs is being underutilized for melanoma in situ and for rare cutaneous malignancies. This represents a knowledge gap for other specialties regarding best-practice therapy,” Dr. Condon said.
He and his coinvestigators searched for socioeconomic predictors of Mohs utilization by matching the nationally representative SEER data with U.S. census data. They examined the impact of three metrics: insurance status, income, and poverty. They found that low-income patients and those in the highest quartile of poverty were significantly less likely to have Mohs surgery for their melanoma in situ and rare cutaneous malignancies throughout the study years. Lack of health insurance had no impact on Mohs utilization for melanoma in situ but was independently associated with decreased likelihood of Mohs for the rare cutaneous malignancies. White patients were 2-fold to 2.4-fold more likely to have Mohs surgery for their rare cutaneous malignancies than were black patients.
“One can conclude that Mohs micrographic surgery may be skewed toward more affluent patients, and lower socioeconomic status areas have less Mohs access. So our data from this study support a role for targeted education and improved patient access to Mohs,” Dr. Condon said.
He noted that because the SEER registries don’t track squamous or basal cell carcinomas, it’s unknown whether Mohs is also underutilized for the higher-risk forms of these most common of all skin cancers.
Dr. Condon reported having no financial conflicts regarding his study, conducted free of commercial support.
CHICAGO – Analysis of U.S. national cancer registry data shows that, contrary to expectation, the , Sean Condon, MD, reported at the annual meeting of the American College of Mohs Surgery.
Ditto for the use of Mohs in patients with the rare cutaneous malignancies for which published evidence clearly demonstrates Mohs outperforms wide local excision, which is employed seven times more frequently than Mohs in such situations.
“Mohs utilization did not increase after the Affordable Care Act [ACA], despite new health insurance coverage for 20 million previously uninsured adults,” Dr. Condon said. “Surprisingly, after the ACA we actually saw a decrease in Mohs use for melanoma in situ.”
Indeed, his retrospective study of more than 25,000 patients in the National Cancer Institute’s SEER (Surveillance, Epidemiology, and End Results) registries showed that the proportion of patients with melanoma in situ treated with Mohs declined from 13.9% during 2008-2009 – prior to ACA implementation – to 12.3% in 2011-2013, after the ACA took effect. That’s a statistically significant 13% drop, even though numerous published studies have shown outcomes in melanoma in situ are better with Mohs, said the dermatologist, who conducted the study while completing a Mohs surgery fellowship at the Cleveland Clinic. He is now in private practice in Thousand Oaks, Calif.
His analysis included 19,013 patients treated in 2008-2014 for melanoma in situ and 6,309 others treated for rare cutaneous malignancies deemed appropriate for Mohs according to the criteria formally developed jointly by the American Academy of Dermatology, the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery (J Am Acad Dermatol. 2012 Oct;67[4]:531-50). These rare malignancies include adnexal carcinoma, Merkel cell carcinoma, dermatofibrosarcoma, extramammary Paget disease, sebaceous adenocarcinoma, and leiomyosarcoma.
“These rare cutaneous malignancies were historically treated with wide local excision. However, numerous studies have lately shown that lower recurrence rates were found with Mohs compared with wide local excision,” Dr. Condon noted.
Nonetheless, the proportion of the rare cutaneous malignancies treated using Mohs was unaffected by implementation of the ACA. Nor was it influenced one way or the other by publication of the joint Mohs appropriate use criteria in 2012: The Mohs-treated proportion of such cases was 15.25% in 2010-2011 and 14.6% in 2013-2014.
Similarly, even though the appropriate use criteria identified melanoma in situ as Mohs appropriate, the proportion of those malignancies treated via Mohs was the same before and after the 2012 release of the criteria.
“It’s commonly thought that Mohs is overused. However, our study and our data clearly identify that Mohs is being underutilized for melanoma in situ and for rare cutaneous malignancies. This represents a knowledge gap for other specialties regarding best-practice therapy,” Dr. Condon said.
He and his coinvestigators searched for socioeconomic predictors of Mohs utilization by matching the nationally representative SEER data with U.S. census data. They examined the impact of three metrics: insurance status, income, and poverty. They found that low-income patients and those in the highest quartile of poverty were significantly less likely to have Mohs surgery for their melanoma in situ and rare cutaneous malignancies throughout the study years. Lack of health insurance had no impact on Mohs utilization for melanoma in situ but was independently associated with decreased likelihood of Mohs for the rare cutaneous malignancies. White patients were 2-fold to 2.4-fold more likely to have Mohs surgery for their rare cutaneous malignancies than were black patients.
“One can conclude that Mohs micrographic surgery may be skewed toward more affluent patients, and lower socioeconomic status areas have less Mohs access. So our data from this study support a role for targeted education and improved patient access to Mohs,” Dr. Condon said.
He noted that because the SEER registries don’t track squamous or basal cell carcinomas, it’s unknown whether Mohs is also underutilized for the higher-risk forms of these most common of all skin cancers.
Dr. Condon reported having no financial conflicts regarding his study, conducted free of commercial support.
REPORTING FROM THE ACMS 50TH ANNUAL MEETING
Key clinical point: Mohs micrographic surgery remains seriously underutilized for the skin cancers for which it is most advantageous.
Major finding: The use of Mohs micrographic surgery to treat melanoma in situ declined significantly after passage of the Affordable Care Act.
Study details: This was a retrospective study of national SEER data on more than 25,000 patients treated for melanoma in situ or rare cutaneous malignancies during 2008-2014.
Disclosures: The presenter reported having no financial conflicts regarding his study, conducted free of commercial support.
New Guidelines for Nonmelanoma Skin Cancer: What You Need to Know
Polypoid Melanoma: An Aggressive Variant of Nodular Melanoma
To the Editor:
An 81-year-old man presented with a nodular polypoid lesion that developed on a flat lesion on the back of 2 years’ duration. The lesion grew progressively over the course of 3 months prior to presentation. The patient had a history of melanoma in situ on the forehead that was treated with conventional surgery with clear surgical margins 6 years prior to the current presentation.
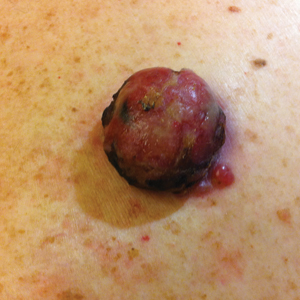
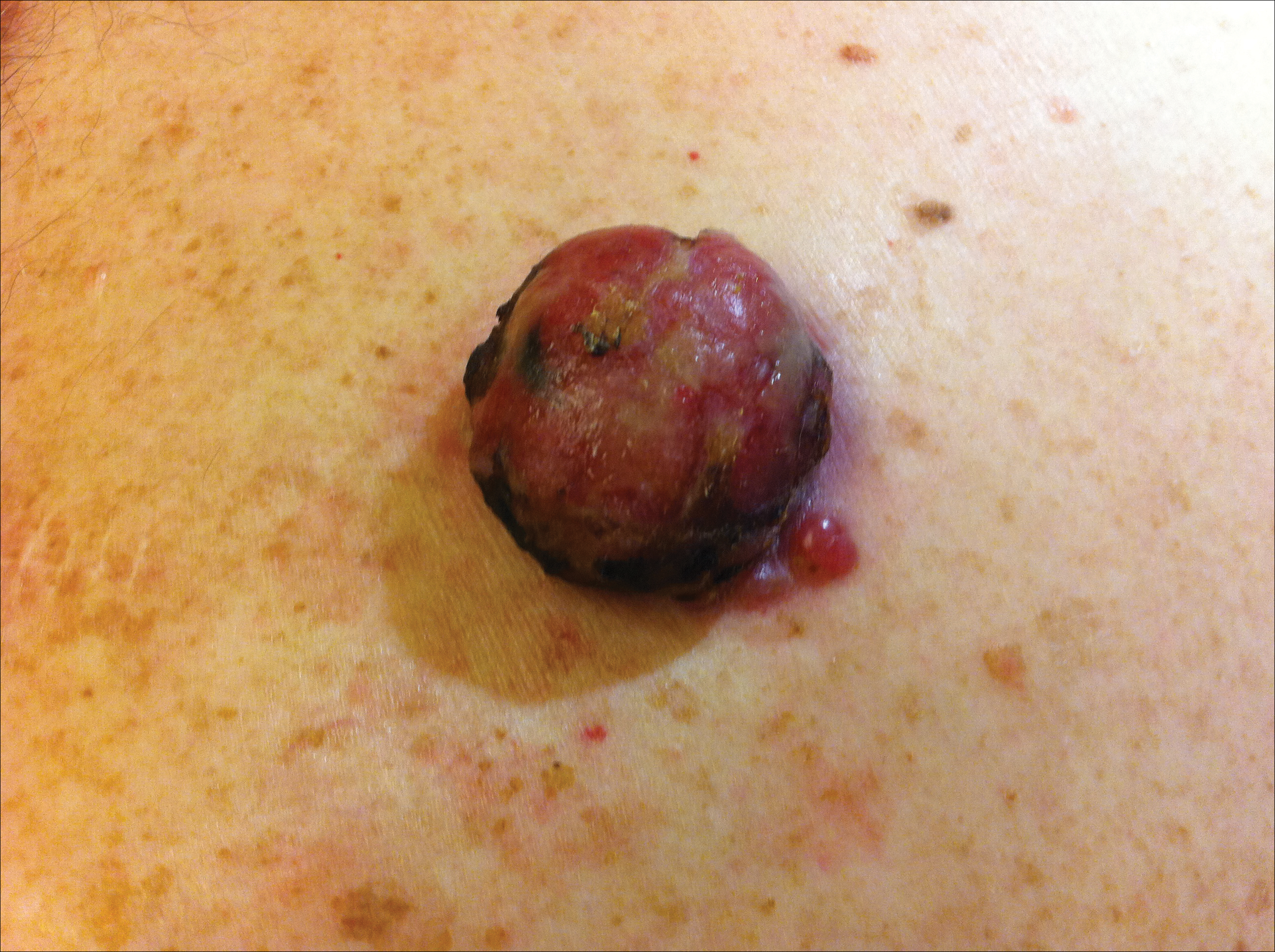
On physical examination the patient had a 4×2-cm ulcerated polypoid lesion on the back. The lesion was pink with a pigmented base. Additionally, 2 pink papules with superficial telangiectases were observed around the main lesion (Figure 1).
The gross section showed an exophytic tumor largely growing above the skin surface (Figure 2). Histopathologic analysis revealed an ulcerated lesion consisting of confluent nest and sheets of epithelioid and spindle atypical cells with numerous mitotic figures and necrotic foci (Figure 3). The thickness of the lesion was 2200 µm, and the mitotic count was 28 mitoses/mm2. There also was peritumoral vascular invasion and satellite metastasis within the perilesional hypodermis measuring 0.4 mm. Immunohistochemistry staining for S-100, human melanoma black 45 (HMB-45)(Figure 4), and Melan-A was positive in neoplastic cells.
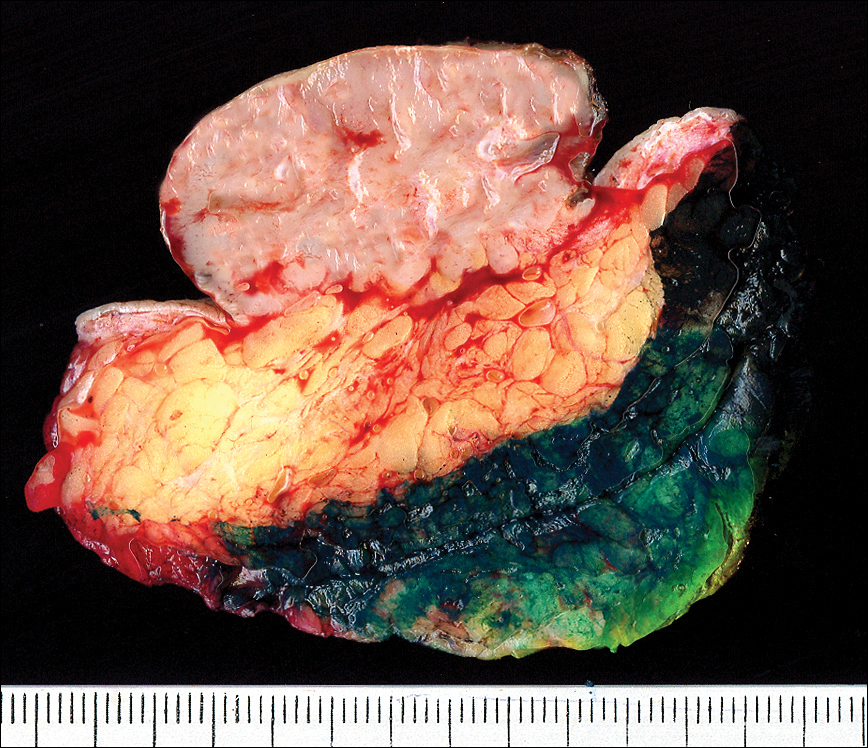
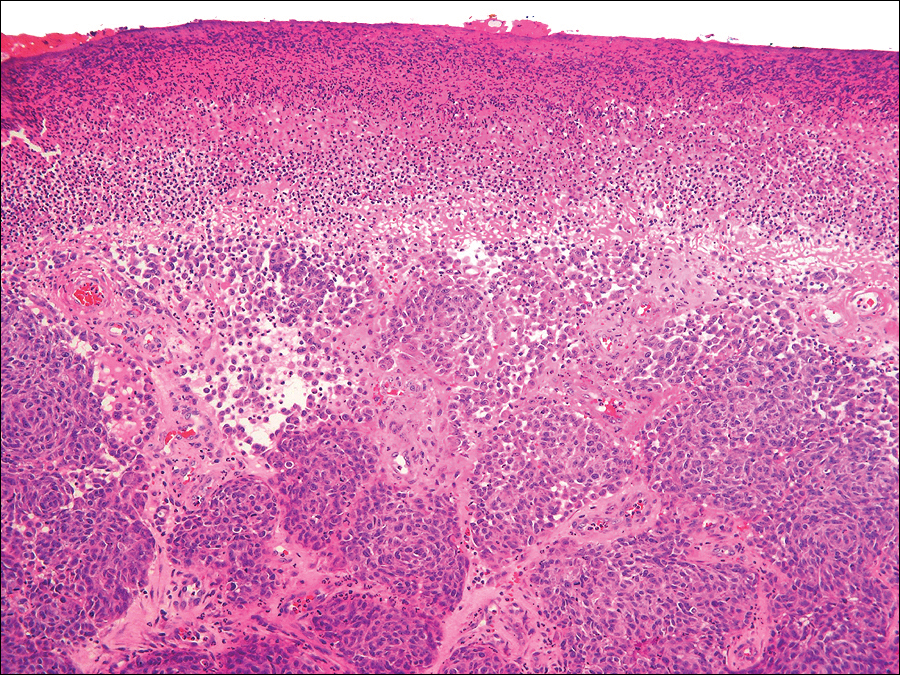

The dissemination study revealed multiple mediastinal and axillary lymphadenopathies and lesions with metastatic appearance in the brain, liver, pancreas, and muscle, together with peritoneal carcinomatosis. The patient was lost to follow-up and did not follow coadjuvant therapy with interferon alfa.
Polypoid melanoma initially was described as a type of melanoma characterized by an exophytic growth in which most of the tumor is located on the cutaneous surface, together with ulceration.1 It usually occurs in patients aged 20 to 39 years,2 and the reported incidence ranges from 1.9% to 43.3%.1 It more commonly affects mucosae, including the upper respiratory tract, esophagus, and vagina. Polypoid melanoma has a rapid progression and a poor prognosis.3 Polypoid melanoma involving the skin primarily affects the back and has a 5-year survival rate of 32% to 42%.4 Poor prognosis has been attributed to the high risk for vascular embolization under the lesion.5 Histologically, there is marked cell atypia with nuclear and cellular pleomorphism and a high mitotic count. The tumor rarely involves the reticular dermis.1,2
Polypoid melanomas are rare; however, reported frequency rates cover a wide range. These frequency rates may be due to the definition of polypoid melanoma used by the pathologist issuing the report. One of the most accepted definitions at present is a pigmented macule that progresses in months with a rapid vertical growth, invading the epidermis and the papillary dermis.2 The differential diagnosis includes pyogenic granuloma, squamous cell carcinoma, basal cell carcinoma, soft tissue sarcomas, and hemangioma.
Although our patient had a history of melanoma and the polypoid lesion developed from a flat lesion, he was late to seek medical care. The diagnosis of melanoma is made on increasingly smaller lesions with better prognosis, but there still are reports of larger melanomas. This case highlights the role dermatologists serve in the education of patients on their diagnoses and risk factors so that we may be able to diagnose non–life-threatening small lesions. It is important to remember this morphologic variety of melanoma and highlight its rapid progression and poor prognosis.
- Knezević F, Duancić V, Sitić S, et al. Histological types of polypoid cutaneous melanoma II. Coll Antropol. 2007;31:1049-1053.
- Dini M, Quercioli F, Caldarella V, et al. Head and neck polypoid melanoma. J Craniofac Surg. 2012;23:E23-E25.
- Plotnick H, Rachmaninoff N, VandenBerg HJ Jr. Polypoid melanoma: a virulent variant of nodular melanoma. report of three cases and literature review. J Am Acad Dermatol. 1990;23(5, pt 1):880-884.
- Manci EA, Balch CM, Murad TM, et al. Polypoid melanoma, a virulent variant of the nodular growth pattern. Am J Clin Pathol. 1981;75:810-815.
- De Giorgi V, Massi D, Gerlini G, et al. Immediate local and regional recurrence after the excision of a polypoid melanoma: tumor dormancy or tumor activation? Dermatol Surg. 2003;29:664-667.
To the Editor:
An 81-year-old man presented with a nodular polypoid lesion that developed on a flat lesion on the back of 2 years’ duration. The lesion grew progressively over the course of 3 months prior to presentation. The patient had a history of melanoma in situ on the forehead that was treated with conventional surgery with clear surgical margins 6 years prior to the current presentation.
On physical examination the patient had a 4×2-cm ulcerated polypoid lesion on the back. The lesion was pink with a pigmented base. Additionally, 2 pink papules with superficial telangiectases were observed around the main lesion (Figure 1).
The gross section showed an exophytic tumor largely growing above the skin surface (Figure 2). Histopathologic analysis revealed an ulcerated lesion consisting of confluent nest and sheets of epithelioid and spindle atypical cells with numerous mitotic figures and necrotic foci (Figure 3). The thickness of the lesion was 2200 µm, and the mitotic count was 28 mitoses/mm2. There also was peritumoral vascular invasion and satellite metastasis within the perilesional hypodermis measuring 0.4 mm. Immunohistochemistry staining for S-100, human melanoma black 45 (HMB-45)(Figure 4), and Melan-A was positive in neoplastic cells.




The dissemination study revealed multiple mediastinal and axillary lymphadenopathies and lesions with metastatic appearance in the brain, liver, pancreas, and muscle, together with peritoneal carcinomatosis. The patient was lost to follow-up and did not follow coadjuvant therapy with interferon alfa.
Polypoid melanoma initially was described as a type of melanoma characterized by an exophytic growth in which most of the tumor is located on the cutaneous surface, together with ulceration.1 It usually occurs in patients aged 20 to 39 years,2 and the reported incidence ranges from 1.9% to 43.3%.1 It more commonly affects mucosae, including the upper respiratory tract, esophagus, and vagina. Polypoid melanoma has a rapid progression and a poor prognosis.3 Polypoid melanoma involving the skin primarily affects the back and has a 5-year survival rate of 32% to 42%.4 Poor prognosis has been attributed to the high risk for vascular embolization under the lesion.5 Histologically, there is marked cell atypia with nuclear and cellular pleomorphism and a high mitotic count. The tumor rarely involves the reticular dermis.1,2
Polypoid melanomas are rare; however, reported frequency rates cover a wide range. These frequency rates may be due to the definition of polypoid melanoma used by the pathologist issuing the report. One of the most accepted definitions at present is a pigmented macule that progresses in months with a rapid vertical growth, invading the epidermis and the papillary dermis.2 The differential diagnosis includes pyogenic granuloma, squamous cell carcinoma, basal cell carcinoma, soft tissue sarcomas, and hemangioma.
Although our patient had a history of melanoma and the polypoid lesion developed from a flat lesion, he was late to seek medical care. The diagnosis of melanoma is made on increasingly smaller lesions with better prognosis, but there still are reports of larger melanomas. This case highlights the role dermatologists serve in the education of patients on their diagnoses and risk factors so that we may be able to diagnose non–life-threatening small lesions. It is important to remember this morphologic variety of melanoma and highlight its rapid progression and poor prognosis.
To the Editor:
An 81-year-old man presented with a nodular polypoid lesion that developed on a flat lesion on the back of 2 years’ duration. The lesion grew progressively over the course of 3 months prior to presentation. The patient had a history of melanoma in situ on the forehead that was treated with conventional surgery with clear surgical margins 6 years prior to the current presentation.
On physical examination the patient had a 4×2-cm ulcerated polypoid lesion on the back. The lesion was pink with a pigmented base. Additionally, 2 pink papules with superficial telangiectases were observed around the main lesion (Figure 1).
The gross section showed an exophytic tumor largely growing above the skin surface (Figure 2). Histopathologic analysis revealed an ulcerated lesion consisting of confluent nest and sheets of epithelioid and spindle atypical cells with numerous mitotic figures and necrotic foci (Figure 3). The thickness of the lesion was 2200 µm, and the mitotic count was 28 mitoses/mm2. There also was peritumoral vascular invasion and satellite metastasis within the perilesional hypodermis measuring 0.4 mm. Immunohistochemistry staining for S-100, human melanoma black 45 (HMB-45)(Figure 4), and Melan-A was positive in neoplastic cells.




The dissemination study revealed multiple mediastinal and axillary lymphadenopathies and lesions with metastatic appearance in the brain, liver, pancreas, and muscle, together with peritoneal carcinomatosis. The patient was lost to follow-up and did not follow coadjuvant therapy with interferon alfa.
Polypoid melanoma initially was described as a type of melanoma characterized by an exophytic growth in which most of the tumor is located on the cutaneous surface, together with ulceration.1 It usually occurs in patients aged 20 to 39 years,2 and the reported incidence ranges from 1.9% to 43.3%.1 It more commonly affects mucosae, including the upper respiratory tract, esophagus, and vagina. Polypoid melanoma has a rapid progression and a poor prognosis.3 Polypoid melanoma involving the skin primarily affects the back and has a 5-year survival rate of 32% to 42%.4 Poor prognosis has been attributed to the high risk for vascular embolization under the lesion.5 Histologically, there is marked cell atypia with nuclear and cellular pleomorphism and a high mitotic count. The tumor rarely involves the reticular dermis.1,2
Polypoid melanomas are rare; however, reported frequency rates cover a wide range. These frequency rates may be due to the definition of polypoid melanoma used by the pathologist issuing the report. One of the most accepted definitions at present is a pigmented macule that progresses in months with a rapid vertical growth, invading the epidermis and the papillary dermis.2 The differential diagnosis includes pyogenic granuloma, squamous cell carcinoma, basal cell carcinoma, soft tissue sarcomas, and hemangioma.
Although our patient had a history of melanoma and the polypoid lesion developed from a flat lesion, he was late to seek medical care. The diagnosis of melanoma is made on increasingly smaller lesions with better prognosis, but there still are reports of larger melanomas. This case highlights the role dermatologists serve in the education of patients on their diagnoses and risk factors so that we may be able to diagnose non–life-threatening small lesions. It is important to remember this morphologic variety of melanoma and highlight its rapid progression and poor prognosis.
- Knezević F, Duancić V, Sitić S, et al. Histological types of polypoid cutaneous melanoma II. Coll Antropol. 2007;31:1049-1053.
- Dini M, Quercioli F, Caldarella V, et al. Head and neck polypoid melanoma. J Craniofac Surg. 2012;23:E23-E25.
- Plotnick H, Rachmaninoff N, VandenBerg HJ Jr. Polypoid melanoma: a virulent variant of nodular melanoma. report of three cases and literature review. J Am Acad Dermatol. 1990;23(5, pt 1):880-884.
- Manci EA, Balch CM, Murad TM, et al. Polypoid melanoma, a virulent variant of the nodular growth pattern. Am J Clin Pathol. 1981;75:810-815.
- De Giorgi V, Massi D, Gerlini G, et al. Immediate local and regional recurrence after the excision of a polypoid melanoma: tumor dormancy or tumor activation? Dermatol Surg. 2003;29:664-667.
- Knezević F, Duancić V, Sitić S, et al. Histological types of polypoid cutaneous melanoma II. Coll Antropol. 2007;31:1049-1053.
- Dini M, Quercioli F, Caldarella V, et al. Head and neck polypoid melanoma. J Craniofac Surg. 2012;23:E23-E25.
- Plotnick H, Rachmaninoff N, VandenBerg HJ Jr. Polypoid melanoma: a virulent variant of nodular melanoma. report of three cases and literature review. J Am Acad Dermatol. 1990;23(5, pt 1):880-884.
- Manci EA, Balch CM, Murad TM, et al. Polypoid melanoma, a virulent variant of the nodular growth pattern. Am J Clin Pathol. 1981;75:810-815.
- De Giorgi V, Massi D, Gerlini G, et al. Immediate local and regional recurrence after the excision of a polypoid melanoma: tumor dormancy or tumor activation? Dermatol Surg. 2003;29:664-667.
Practice Points
- The differential diagnosis of polypoid melanoma includes pyogenic granuloma and squamous cell carcinoma.
- Polypoid melanoma has a poor prognosis because of its thickness and ulceration at the time of diagnosis and the risk of vascular embolization.
IMPACT study: Matched targeted therapy improves survival in advanced cancer
CHICAGO – according to findings from a retrospective analysis of molecularly profiled patients.
Of 3,743 patients tested as part of IMPACT (Initiative for Molecular Profiling and Advanced Cancer Therapy), 1,307 (34.9%) had at least one targetable molecular alteration. Of those, 711 (54.4%) received either matched targeted therapy that was being tested in a clinical trial or – in a small number of cases – therapy with an approved treatment used off label, and 596 (45.6%) received nonmatched therapy, Apostolia-Maria Tsimberidou, MD, reported during a press briefing at the annual meeting of the American Society of Clinical Oncology.
The objective response rates in 697 evaluable matched therapy patients was 16.2% versus 5.4% in 571 evaluable nonmatched patients, and stable disease for at least 6 months occurred in 18.7% and 14.7% of patients, respectively, for an overall disease control rate of 34.9% versus 20.1%, said Dr. Tsimberidou, a professor at the University of Texas MD Anderson Cancer Center, Houston.
Median progression-free survival in those who received matched versus nonmatched therapy was 4.0 months and 2.8 months, respectively (hazard ratio, 0.67), and median overall survival was 9.3 and 7.3 months, respectively (HR, 0.72), she said.
The 3-year overall survival rate was 15% versus 7%, respectively, and 10-year survival was 6% and 1%, respectively.
Patients included in IMPACT had a mean age of 57 years, and 39% were men. They were heavily pretreated (mean number of prior therapies was 4); only 2.8% of patients had no prior treatment. Cancers included gastrointestinal (24.2%), gynecologic (19.4%), breast (13.5%), melanoma (11.9%) and lung (8.7%).
In this video interview, Dr. Tsimberidou describes the rationale, methodology, and findings of IMPACT, including the use of a prognostic scoring system developed as part of the study to predict overall survival based on baseline characteristics, such as baseline p13K/AKT/mTOR pathway molecular alterations, which were shown on multivariate analysis in IMPACT to predict shorter overall survival versus other alterations. Other predictors of shorter survival included liver metastases, elevated lactate dehydrogenase levels, poor functional status, low albumin levels, elevated platelet counts, and age of 60 years or older.
“We [also] wanted to see if adding the intervention ... would hold significance in this multivariate model, and we found that ... nonmatched therapy was associated with adverse survival; it was an independent factor associated with worse survival,” she said. “Therefore, matched targeted therapy is associated with longer survival.”
In the randomized, phase 2 trial IMPACT 2, progression-free survival will be compared in patients with and without matched targeted therapy, and the prognostic scoring system developed as part of IMPACT to predict overall survival based on baseline characteristics will be further evaluated, she said.
During a discussion of the findings during the press briefing, ASCO Expert Catherine M. Diefenbach, MD, said the type of precision medicine studied in IMPACT is “the wave of the future.
“Large scale efforts such as ASCO’s TAPUR or the NCI-MATCH trial will bring these efforts to many, many more patients, and hopefully usher in a new way of treating advanced cancer patients that will improve overall survival for many more patients,” said Dr. Diefenbach, of New York University.
Dr. Tsimberidou reported a consulting or advisory role with Roche, as well as research funding to her institution from EMD Serono, Baxter, Foundation Medicine, ONYX, Bayer, Boston Biomedical, Placon, IMMATICS, Karus Therapeutics, and StemCells.
SOURCE: Tsimberidou AM et al. ASCO 2018, Abstract LBA 2553.
CHICAGO – according to findings from a retrospective analysis of molecularly profiled patients.
Of 3,743 patients tested as part of IMPACT (Initiative for Molecular Profiling and Advanced Cancer Therapy), 1,307 (34.9%) had at least one targetable molecular alteration. Of those, 711 (54.4%) received either matched targeted therapy that was being tested in a clinical trial or – in a small number of cases – therapy with an approved treatment used off label, and 596 (45.6%) received nonmatched therapy, Apostolia-Maria Tsimberidou, MD, reported during a press briefing at the annual meeting of the American Society of Clinical Oncology.
The objective response rates in 697 evaluable matched therapy patients was 16.2% versus 5.4% in 571 evaluable nonmatched patients, and stable disease for at least 6 months occurred in 18.7% and 14.7% of patients, respectively, for an overall disease control rate of 34.9% versus 20.1%, said Dr. Tsimberidou, a professor at the University of Texas MD Anderson Cancer Center, Houston.
Median progression-free survival in those who received matched versus nonmatched therapy was 4.0 months and 2.8 months, respectively (hazard ratio, 0.67), and median overall survival was 9.3 and 7.3 months, respectively (HR, 0.72), she said.
The 3-year overall survival rate was 15% versus 7%, respectively, and 10-year survival was 6% and 1%, respectively.
Patients included in IMPACT had a mean age of 57 years, and 39% were men. They were heavily pretreated (mean number of prior therapies was 4); only 2.8% of patients had no prior treatment. Cancers included gastrointestinal (24.2%), gynecologic (19.4%), breast (13.5%), melanoma (11.9%) and lung (8.7%).
In this video interview, Dr. Tsimberidou describes the rationale, methodology, and findings of IMPACT, including the use of a prognostic scoring system developed as part of the study to predict overall survival based on baseline characteristics, such as baseline p13K/AKT/mTOR pathway molecular alterations, which were shown on multivariate analysis in IMPACT to predict shorter overall survival versus other alterations. Other predictors of shorter survival included liver metastases, elevated lactate dehydrogenase levels, poor functional status, low albumin levels, elevated platelet counts, and age of 60 years or older.
“We [also] wanted to see if adding the intervention ... would hold significance in this multivariate model, and we found that ... nonmatched therapy was associated with adverse survival; it was an independent factor associated with worse survival,” she said. “Therefore, matched targeted therapy is associated with longer survival.”
In the randomized, phase 2 trial IMPACT 2, progression-free survival will be compared in patients with and without matched targeted therapy, and the prognostic scoring system developed as part of IMPACT to predict overall survival based on baseline characteristics will be further evaluated, she said.
During a discussion of the findings during the press briefing, ASCO Expert Catherine M. Diefenbach, MD, said the type of precision medicine studied in IMPACT is “the wave of the future.
“Large scale efforts such as ASCO’s TAPUR or the NCI-MATCH trial will bring these efforts to many, many more patients, and hopefully usher in a new way of treating advanced cancer patients that will improve overall survival for many more patients,” said Dr. Diefenbach, of New York University.
Dr. Tsimberidou reported a consulting or advisory role with Roche, as well as research funding to her institution from EMD Serono, Baxter, Foundation Medicine, ONYX, Bayer, Boston Biomedical, Placon, IMMATICS, Karus Therapeutics, and StemCells.
SOURCE: Tsimberidou AM et al. ASCO 2018, Abstract LBA 2553.
CHICAGO – according to findings from a retrospective analysis of molecularly profiled patients.
Of 3,743 patients tested as part of IMPACT (Initiative for Molecular Profiling and Advanced Cancer Therapy), 1,307 (34.9%) had at least one targetable molecular alteration. Of those, 711 (54.4%) received either matched targeted therapy that was being tested in a clinical trial or – in a small number of cases – therapy with an approved treatment used off label, and 596 (45.6%) received nonmatched therapy, Apostolia-Maria Tsimberidou, MD, reported during a press briefing at the annual meeting of the American Society of Clinical Oncology.
The objective response rates in 697 evaluable matched therapy patients was 16.2% versus 5.4% in 571 evaluable nonmatched patients, and stable disease for at least 6 months occurred in 18.7% and 14.7% of patients, respectively, for an overall disease control rate of 34.9% versus 20.1%, said Dr. Tsimberidou, a professor at the University of Texas MD Anderson Cancer Center, Houston.
Median progression-free survival in those who received matched versus nonmatched therapy was 4.0 months and 2.8 months, respectively (hazard ratio, 0.67), and median overall survival was 9.3 and 7.3 months, respectively (HR, 0.72), she said.
The 3-year overall survival rate was 15% versus 7%, respectively, and 10-year survival was 6% and 1%, respectively.
Patients included in IMPACT had a mean age of 57 years, and 39% were men. They were heavily pretreated (mean number of prior therapies was 4); only 2.8% of patients had no prior treatment. Cancers included gastrointestinal (24.2%), gynecologic (19.4%), breast (13.5%), melanoma (11.9%) and lung (8.7%).
In this video interview, Dr. Tsimberidou describes the rationale, methodology, and findings of IMPACT, including the use of a prognostic scoring system developed as part of the study to predict overall survival based on baseline characteristics, such as baseline p13K/AKT/mTOR pathway molecular alterations, which were shown on multivariate analysis in IMPACT to predict shorter overall survival versus other alterations. Other predictors of shorter survival included liver metastases, elevated lactate dehydrogenase levels, poor functional status, low albumin levels, elevated platelet counts, and age of 60 years or older.
“We [also] wanted to see if adding the intervention ... would hold significance in this multivariate model, and we found that ... nonmatched therapy was associated with adverse survival; it was an independent factor associated with worse survival,” she said. “Therefore, matched targeted therapy is associated with longer survival.”
In the randomized, phase 2 trial IMPACT 2, progression-free survival will be compared in patients with and without matched targeted therapy, and the prognostic scoring system developed as part of IMPACT to predict overall survival based on baseline characteristics will be further evaluated, she said.
During a discussion of the findings during the press briefing, ASCO Expert Catherine M. Diefenbach, MD, said the type of precision medicine studied in IMPACT is “the wave of the future.
“Large scale efforts such as ASCO’s TAPUR or the NCI-MATCH trial will bring these efforts to many, many more patients, and hopefully usher in a new way of treating advanced cancer patients that will improve overall survival for many more patients,” said Dr. Diefenbach, of New York University.
Dr. Tsimberidou reported a consulting or advisory role with Roche, as well as research funding to her institution from EMD Serono, Baxter, Foundation Medicine, ONYX, Bayer, Boston Biomedical, Placon, IMMATICS, Karus Therapeutics, and StemCells.
SOURCE: Tsimberidou AM et al. ASCO 2018, Abstract LBA 2553.
REPORTING FROM ASCO 2018
Key clinical point: Matched targeted therapy improved survival in patients with advanced cancer.
Major finding: The 3-yearoverall survival rate with matched versus nonmatched therapy was 15% and 7%, respectively.
Study details: A retrospective analysis (IMPACT) of 3,743 molecularly profiled advanced cancer patients.
Disclosures: Dr. Tsimberidou reported a consulting or advisory role with Roche, as well as research funding to her institution from EMD Serono, Baxter, Foundation Medicine, ONYX Medical, Bayer, Boston Biomedical, Placon, IMMATICS, Karus Therapeutics, and StemCells.
Source: Tsimberidou AM et al. ASCO 2018, Abstract LBA 2553.
New criteria slightly improved melanoma staging
Use of the recently updated AJCC Cancer Staging Manual, 8th edition, produced about 10% better accuracy when interpreting T1a invasive melanomas in a national study.
For stage T1b or greater cases, using the AJCC 8 instead of the AJCC 7 yielded about 6% higher concordance between interpretations and consensus reference diagnoses, said Joann G. Elmore, MD, MPH, of the David Geffen School of Medicine at the University of California, Los Angeles, and her associates. “Intraobserver reproducibility of diagnoses also improved,” but “concordance and reproducibility remain low,” the researchers wrote. The report was published in JAMA Open Network.
For the study, 187 pathologists each interpreted one of five glass slide sets of melanocytic skin biopsies. At least 8 months later, they interpreted the same set again, both times recording their interpretations on the online Pathology Assessment Tool and Hierarchy for Diagnosis (MPATH-Dx). Researchers then used AJCC 7 and AJCC 8 criteria to group these interpretations into one of five MPATH-Dx classes. The final analysis included only the 116 cases of invasive melanoma, because changes in the AJCC 8 criteria affected only MPATH-Dx classes IV (T1a) and V (T1b and higher), the investigators explained.
For T1a invasive melanomas, overall concordance between interpretations and consensus diagnoses rose from 44% (95% confidence interval, 41%-48%) under the AJCC 7 criteria to 54% (95% CI, 51%-57%) under the AJCC 8 criteria. For stage T1b or higher lesions, concordance rose from 72% to 78%. Intrapathologist reproducibility improved from 59% to 64% for T1a cases and from 74% to 77% for T1b or higher cases. Thus, the AJCC 8 produced “modest” improvements in concordance and reproducibility over the AJCC 7, the researchers said.
One of the most likely explanations is that the AJCC 8 eliminated dermal mitotic activity as a criterion for T1b melanomas, which is “potentially unreliable” in thin lesions, they added. Persistently low concordance and reproducibility “suggest that conventional histopathology has been parsed to a degree that falls below the limits of reliability,” they concluded.
The National Cancer Institute provided funding. Dr. Elmore disclosed ties to Myriad Genetics, SciBase, and the National Institutes of Health.
SOURCE: Elmore JG et al. JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0083.
Compared with the seventh edition, use of the eighth AJCC Cancer Staging Manual (AJCC 8) produced “modest but significant improvements” in reproducibility and concordance when microstaging cutaneous melanoma, Peter M. Ferguson, MBChB, PhD, FRCPA, Jeffrey E. Gershenwald, MD, and Richard A. Scolyer, MD, FRCPA, wrote in an editorial accompanying the study.
But the study revealed “significant” shortcomings in reproducibility, they added. Future studies should explore the reproducibility of individual AJCC parameters and how experience and access to relevant case data affects reproducibility, they wrote.
The study used the Melanocytic Pathology Assessment Tool and Hierarchy for Diagnosis system instead of classifying cases by AJCC tumor categories. Consequently, pathologists collapsed several AJCC tumor subcategories into the Melanoma Pathology Study class V, which limited interpretability of results for T1b melanomas, an important threshold for recommending sentinel node biopsy. “[We] have published higher rates of concordance using AJCC 7 criteria, albeit with access to clinical information and reporting by a small number of pathologists with expertise reporting melanomas at a high-volume referral center,” the editorialists emphasized.
Dr. Ferguson and Dr. Scolyer are with the University of Sydney. Dr. Gershenwald is with the University of Texas MD Anderson Cancer Center, Houston. The editorialists disclosed support from the Robert and Lynne Grossman Family Foundation, the Michael and Patricia Booker Melanoma Research Endowment, Melanoma Institute Australia, the Deborah and John McMurtrie Melanoma Institute Australia Pathology Fellowship, and the Australian National Health and Medical Research Council Fellowship. Dr. Gershenwald disclosed ties to Merck, Syndax, Castle Biosciences, and the AJCC (JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0086 ).
Compared with the seventh edition, use of the eighth AJCC Cancer Staging Manual (AJCC 8) produced “modest but significant improvements” in reproducibility and concordance when microstaging cutaneous melanoma, Peter M. Ferguson, MBChB, PhD, FRCPA, Jeffrey E. Gershenwald, MD, and Richard A. Scolyer, MD, FRCPA, wrote in an editorial accompanying the study.
But the study revealed “significant” shortcomings in reproducibility, they added. Future studies should explore the reproducibility of individual AJCC parameters and how experience and access to relevant case data affects reproducibility, they wrote.
The study used the Melanocytic Pathology Assessment Tool and Hierarchy for Diagnosis system instead of classifying cases by AJCC tumor categories. Consequently, pathologists collapsed several AJCC tumor subcategories into the Melanoma Pathology Study class V, which limited interpretability of results for T1b melanomas, an important threshold for recommending sentinel node biopsy. “[We] have published higher rates of concordance using AJCC 7 criteria, albeit with access to clinical information and reporting by a small number of pathologists with expertise reporting melanomas at a high-volume referral center,” the editorialists emphasized.
Dr. Ferguson and Dr. Scolyer are with the University of Sydney. Dr. Gershenwald is with the University of Texas MD Anderson Cancer Center, Houston. The editorialists disclosed support from the Robert and Lynne Grossman Family Foundation, the Michael and Patricia Booker Melanoma Research Endowment, Melanoma Institute Australia, the Deborah and John McMurtrie Melanoma Institute Australia Pathology Fellowship, and the Australian National Health and Medical Research Council Fellowship. Dr. Gershenwald disclosed ties to Merck, Syndax, Castle Biosciences, and the AJCC (JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0086 ).
Compared with the seventh edition, use of the eighth AJCC Cancer Staging Manual (AJCC 8) produced “modest but significant improvements” in reproducibility and concordance when microstaging cutaneous melanoma, Peter M. Ferguson, MBChB, PhD, FRCPA, Jeffrey E. Gershenwald, MD, and Richard A. Scolyer, MD, FRCPA, wrote in an editorial accompanying the study.
But the study revealed “significant” shortcomings in reproducibility, they added. Future studies should explore the reproducibility of individual AJCC parameters and how experience and access to relevant case data affects reproducibility, they wrote.
The study used the Melanocytic Pathology Assessment Tool and Hierarchy for Diagnosis system instead of classifying cases by AJCC tumor categories. Consequently, pathologists collapsed several AJCC tumor subcategories into the Melanoma Pathology Study class V, which limited interpretability of results for T1b melanomas, an important threshold for recommending sentinel node biopsy. “[We] have published higher rates of concordance using AJCC 7 criteria, albeit with access to clinical information and reporting by a small number of pathologists with expertise reporting melanomas at a high-volume referral center,” the editorialists emphasized.
Dr. Ferguson and Dr. Scolyer are with the University of Sydney. Dr. Gershenwald is with the University of Texas MD Anderson Cancer Center, Houston. The editorialists disclosed support from the Robert and Lynne Grossman Family Foundation, the Michael and Patricia Booker Melanoma Research Endowment, Melanoma Institute Australia, the Deborah and John McMurtrie Melanoma Institute Australia Pathology Fellowship, and the Australian National Health and Medical Research Council Fellowship. Dr. Gershenwald disclosed ties to Merck, Syndax, Castle Biosciences, and the AJCC (JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0086 ).
Use of the recently updated AJCC Cancer Staging Manual, 8th edition, produced about 10% better accuracy when interpreting T1a invasive melanomas in a national study.
For stage T1b or greater cases, using the AJCC 8 instead of the AJCC 7 yielded about 6% higher concordance between interpretations and consensus reference diagnoses, said Joann G. Elmore, MD, MPH, of the David Geffen School of Medicine at the University of California, Los Angeles, and her associates. “Intraobserver reproducibility of diagnoses also improved,” but “concordance and reproducibility remain low,” the researchers wrote. The report was published in JAMA Open Network.
For the study, 187 pathologists each interpreted one of five glass slide sets of melanocytic skin biopsies. At least 8 months later, they interpreted the same set again, both times recording their interpretations on the online Pathology Assessment Tool and Hierarchy for Diagnosis (MPATH-Dx). Researchers then used AJCC 7 and AJCC 8 criteria to group these interpretations into one of five MPATH-Dx classes. The final analysis included only the 116 cases of invasive melanoma, because changes in the AJCC 8 criteria affected only MPATH-Dx classes IV (T1a) and V (T1b and higher), the investigators explained.
For T1a invasive melanomas, overall concordance between interpretations and consensus diagnoses rose from 44% (95% confidence interval, 41%-48%) under the AJCC 7 criteria to 54% (95% CI, 51%-57%) under the AJCC 8 criteria. For stage T1b or higher lesions, concordance rose from 72% to 78%. Intrapathologist reproducibility improved from 59% to 64% for T1a cases and from 74% to 77% for T1b or higher cases. Thus, the AJCC 8 produced “modest” improvements in concordance and reproducibility over the AJCC 7, the researchers said.
One of the most likely explanations is that the AJCC 8 eliminated dermal mitotic activity as a criterion for T1b melanomas, which is “potentially unreliable” in thin lesions, they added. Persistently low concordance and reproducibility “suggest that conventional histopathology has been parsed to a degree that falls below the limits of reliability,” they concluded.
The National Cancer Institute provided funding. Dr. Elmore disclosed ties to Myriad Genetics, SciBase, and the National Institutes of Health.
SOURCE: Elmore JG et al. JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0083.
Use of the recently updated AJCC Cancer Staging Manual, 8th edition, produced about 10% better accuracy when interpreting T1a invasive melanomas in a national study.
For stage T1b or greater cases, using the AJCC 8 instead of the AJCC 7 yielded about 6% higher concordance between interpretations and consensus reference diagnoses, said Joann G. Elmore, MD, MPH, of the David Geffen School of Medicine at the University of California, Los Angeles, and her associates. “Intraobserver reproducibility of diagnoses also improved,” but “concordance and reproducibility remain low,” the researchers wrote. The report was published in JAMA Open Network.
For the study, 187 pathologists each interpreted one of five glass slide sets of melanocytic skin biopsies. At least 8 months later, they interpreted the same set again, both times recording their interpretations on the online Pathology Assessment Tool and Hierarchy for Diagnosis (MPATH-Dx). Researchers then used AJCC 7 and AJCC 8 criteria to group these interpretations into one of five MPATH-Dx classes. The final analysis included only the 116 cases of invasive melanoma, because changes in the AJCC 8 criteria affected only MPATH-Dx classes IV (T1a) and V (T1b and higher), the investigators explained.
For T1a invasive melanomas, overall concordance between interpretations and consensus diagnoses rose from 44% (95% confidence interval, 41%-48%) under the AJCC 7 criteria to 54% (95% CI, 51%-57%) under the AJCC 8 criteria. For stage T1b or higher lesions, concordance rose from 72% to 78%. Intrapathologist reproducibility improved from 59% to 64% for T1a cases and from 74% to 77% for T1b or higher cases. Thus, the AJCC 8 produced “modest” improvements in concordance and reproducibility over the AJCC 7, the researchers said.
One of the most likely explanations is that the AJCC 8 eliminated dermal mitotic activity as a criterion for T1b melanomas, which is “potentially unreliable” in thin lesions, they added. Persistently low concordance and reproducibility “suggest that conventional histopathology has been parsed to a degree that falls below the limits of reliability,” they concluded.
The National Cancer Institute provided funding. Dr. Elmore disclosed ties to Myriad Genetics, SciBase, and the National Institutes of Health.
SOURCE: Elmore JG et al. JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0083.
FROM JAMA NETWORK OPEN
Key clinical point: Use of the recently updated AJCC Cancer Staging Manual, 8th edition, somewhat improved concordance and reproducibility for invasive melanoma, compared with the previous edition.
Major finding: For diagnoses of T1a invasive melanoma, concordance with consensus diagnoses rose from 44% with the AJCC 7 to 54% with the AJCC 8. For T1b, concordance rose from 72% to 78%.
Study details: Interpretations of 116 invasive melanomas by 187 pathologists.
Disclosures: The National Cancer Institute provided funding. Dr. Elmore disclosed ties to Myriad Genetics, SciBase, and the National Institutes of Health.
Source: Elmore JG et al. JAMA Network Open. 2018 May 18. doi: 10.1001/jamanetworkopen.2018.0083.