User login
Ropivacaine called top anesthesia for nail surgery
CHICAGO – Ropivacaine has a fast onset of action, longer duration than either lidocaine or bupivacaine, and it’s the only one of the three that’s inherently vasoconstrictive. For Brienne Cressey, MD, those features make ropivacaine the local anesthetic of choice in performing nail surgery.
“Local anesthesia is really key for nail surgery. If you don’t have good anesthesia it’s not a good experience for either the surgeon or the patient,” she observed at the annual meeting of the American College of Mohs Surgery.
Lidocaine has a fast onset – less than 1 minute – but a problematic short duration of 30-120 minutes. Bupivacaine has the disadvantage of a slow onset of up to 5 minutes, albeit with a longer duration of anesthesia at 2-4 hours. Ropivacaine has a fast onset, plus a duration of up to 8 hours. And unlike lidocaine and bupivacaine, which are vasodilatory, ropivacaine is vasoconstrictive.
“With lidocaine, you get a lot of blood right after you take off your tourniquet. With ropivacaine, you get really nice reperfusion, but it’s not too much. You take off the tourniquet, check to see you’ve got reperfusion, then you add a little ropivacaine – about 0.5 mL – on either side of the base of the distal phalanx. It stops the bleeding immediately and you can easily put on a pressure dressing. It’s a nice way to get the patient over the hump of those first hours of pain and lets them drive home in comfort,” explained Dr. Cressey, a dermatologist working in a group practice at Dermatology Professionals in East Greenwich, R.I.
Ropivacaine is less cardiotoxic than bupivacaine. And ropivacaine offers an additional advantage: Its pH is such that no buffering is necessary. “Ropivacaine doesn’t require any compounding. You can just use it at 1% straight out of the bottle. That’s what we do in our office, and we’ve had very good experience with it,” according to the dermatologist.
Achieving smooth sailing with local anesthesia
Dr. Cressey delivers ropivacaine slowly through a 30-gauge needle, which makes for a smaller, less painful puncture. She utilizes a topical cold spray, and places a vibrating machine as a distractant proximal to where she is injecting. She keeps the anesthetic at room temperature or warms it to body temperature in a water bath as another means of reducing the pain of injection.
The distal digital block
This is a cross between a traditional proximal digital block and a wing block. It works well for the second, third, and fourth digits, which are mostly volar dominant. The block bathes the volar nerve branch in anesthesia at the midline of the finger or toe.
Dr. Cressey begins by injecting ropivacaine proximal and lateral to the junction of the proximal nail fold and lateral nail fold. After creating a dermal wheal, she directs her needle perpendicularly downward toward the finger or toe pad, injecting 1-4 mL of anesthesia, depending upon digit size. Visible blanching will progress digitally. If resistance is encountered, it suggests the needle has penetrated a ligament or other fibrous tissue. Simply withdraw the needle and continue injecting.
“What’s nice about the distal digital block is you get an immediate effect, and there’s good hemostasis during the procedure as well,” she said.
Dr. Cressey reported no financial conflicts regarding her presentation.
CHICAGO – Ropivacaine has a fast onset of action, longer duration than either lidocaine or bupivacaine, and it’s the only one of the three that’s inherently vasoconstrictive. For Brienne Cressey, MD, those features make ropivacaine the local anesthetic of choice in performing nail surgery.
“Local anesthesia is really key for nail surgery. If you don’t have good anesthesia it’s not a good experience for either the surgeon or the patient,” she observed at the annual meeting of the American College of Mohs Surgery.
Lidocaine has a fast onset – less than 1 minute – but a problematic short duration of 30-120 minutes. Bupivacaine has the disadvantage of a slow onset of up to 5 minutes, albeit with a longer duration of anesthesia at 2-4 hours. Ropivacaine has a fast onset, plus a duration of up to 8 hours. And unlike lidocaine and bupivacaine, which are vasodilatory, ropivacaine is vasoconstrictive.
“With lidocaine, you get a lot of blood right after you take off your tourniquet. With ropivacaine, you get really nice reperfusion, but it’s not too much. You take off the tourniquet, check to see you’ve got reperfusion, then you add a little ropivacaine – about 0.5 mL – on either side of the base of the distal phalanx. It stops the bleeding immediately and you can easily put on a pressure dressing. It’s a nice way to get the patient over the hump of those first hours of pain and lets them drive home in comfort,” explained Dr. Cressey, a dermatologist working in a group practice at Dermatology Professionals in East Greenwich, R.I.
Ropivacaine is less cardiotoxic than bupivacaine. And ropivacaine offers an additional advantage: Its pH is such that no buffering is necessary. “Ropivacaine doesn’t require any compounding. You can just use it at 1% straight out of the bottle. That’s what we do in our office, and we’ve had very good experience with it,” according to the dermatologist.
Achieving smooth sailing with local anesthesia
Dr. Cressey delivers ropivacaine slowly through a 30-gauge needle, which makes for a smaller, less painful puncture. She utilizes a topical cold spray, and places a vibrating machine as a distractant proximal to where she is injecting. She keeps the anesthetic at room temperature or warms it to body temperature in a water bath as another means of reducing the pain of injection.
The distal digital block
This is a cross between a traditional proximal digital block and a wing block. It works well for the second, third, and fourth digits, which are mostly volar dominant. The block bathes the volar nerve branch in anesthesia at the midline of the finger or toe.
Dr. Cressey begins by injecting ropivacaine proximal and lateral to the junction of the proximal nail fold and lateral nail fold. After creating a dermal wheal, she directs her needle perpendicularly downward toward the finger or toe pad, injecting 1-4 mL of anesthesia, depending upon digit size. Visible blanching will progress digitally. If resistance is encountered, it suggests the needle has penetrated a ligament or other fibrous tissue. Simply withdraw the needle and continue injecting.
“What’s nice about the distal digital block is you get an immediate effect, and there’s good hemostasis during the procedure as well,” she said.
Dr. Cressey reported no financial conflicts regarding her presentation.
CHICAGO – Ropivacaine has a fast onset of action, longer duration than either lidocaine or bupivacaine, and it’s the only one of the three that’s inherently vasoconstrictive. For Brienne Cressey, MD, those features make ropivacaine the local anesthetic of choice in performing nail surgery.
“Local anesthesia is really key for nail surgery. If you don’t have good anesthesia it’s not a good experience for either the surgeon or the patient,” she observed at the annual meeting of the American College of Mohs Surgery.
Lidocaine has a fast onset – less than 1 minute – but a problematic short duration of 30-120 minutes. Bupivacaine has the disadvantage of a slow onset of up to 5 minutes, albeit with a longer duration of anesthesia at 2-4 hours. Ropivacaine has a fast onset, plus a duration of up to 8 hours. And unlike lidocaine and bupivacaine, which are vasodilatory, ropivacaine is vasoconstrictive.
“With lidocaine, you get a lot of blood right after you take off your tourniquet. With ropivacaine, you get really nice reperfusion, but it’s not too much. You take off the tourniquet, check to see you’ve got reperfusion, then you add a little ropivacaine – about 0.5 mL – on either side of the base of the distal phalanx. It stops the bleeding immediately and you can easily put on a pressure dressing. It’s a nice way to get the patient over the hump of those first hours of pain and lets them drive home in comfort,” explained Dr. Cressey, a dermatologist working in a group practice at Dermatology Professionals in East Greenwich, R.I.
Ropivacaine is less cardiotoxic than bupivacaine. And ropivacaine offers an additional advantage: Its pH is such that no buffering is necessary. “Ropivacaine doesn’t require any compounding. You can just use it at 1% straight out of the bottle. That’s what we do in our office, and we’ve had very good experience with it,” according to the dermatologist.
Achieving smooth sailing with local anesthesia
Dr. Cressey delivers ropivacaine slowly through a 30-gauge needle, which makes for a smaller, less painful puncture. She utilizes a topical cold spray, and places a vibrating machine as a distractant proximal to where she is injecting. She keeps the anesthetic at room temperature or warms it to body temperature in a water bath as another means of reducing the pain of injection.
The distal digital block
This is a cross between a traditional proximal digital block and a wing block. It works well for the second, third, and fourth digits, which are mostly volar dominant. The block bathes the volar nerve branch in anesthesia at the midline of the finger or toe.
Dr. Cressey begins by injecting ropivacaine proximal and lateral to the junction of the proximal nail fold and lateral nail fold. After creating a dermal wheal, she directs her needle perpendicularly downward toward the finger or toe pad, injecting 1-4 mL of anesthesia, depending upon digit size. Visible blanching will progress digitally. If resistance is encountered, it suggests the needle has penetrated a ligament or other fibrous tissue. Simply withdraw the needle and continue injecting.
“What’s nice about the distal digital block is you get an immediate effect, and there’s good hemostasis during the procedure as well,” she said.
Dr. Cressey reported no financial conflicts regarding her presentation.
EXPERT ANALYSIS FROM THE ACMS ANNUAL MEETING
Using Dermoscopy to Identify Melanoma and Improve Diagnostic Discrimination (FULL)
From 1982 to 2011, the melanoma incidence rate doubled in the US.1 In 2018, an estimated 87,290 cases of melanoma in situ and 91,270 cases of invasive melanoma will be diagnosed in the US, and 9,320 deaths will be attributable to melanoma.2 Early detection of melanoma is critically important to reduce melanoma-related mortality, with 5-year survival rates as high as 97% at stage 1A vs a 20% 5-year survival when there is distant metastasis.2,3 Melanoma is particularly relevant for medical providers working with veterans because melanoma disproportionately affects service members with an incidence rate ratio of 1.62 (95% confidence interval [CI], 1.40-1.86) compared with that of the general population.4
Biopsy is the definitive diagnostic tool for melanoma. Histologic analysis differentiates melanoma from seborrheic keratoses, pigmented nevi, dermatofibromas, and other pigmented lesions that can resemble melanoma on clinical examination. However, biopsy must be used judiciously as unnecessary biopsies contribute to health care costs and leave scars, which can have psychosocial implications. With benign nevi outnumbering melanoma about 2 million to 1, biopsy is indicated once a threshold of suspicion is obtained.5
Dermoscopic Tool
Dermoscopy is a microscopy-based tool to improve noninvasive diagnostic discrimination of skin lesions based on color and structure analysis. Coloration provides an indication of the composition of elements present in the skin with keratin appearing yellow, blood appearing red, and collagen appearing white. Coloration also suggests pigment depth as melanin appears black when located in the stratum corneum, brown when located deeper in the epidermis, and blue when located in the dermis.6 Finally, characteristic histopathologic alterations in the dermoepidermal junction, rete ridges, pigment-containing cells, and/or melanocyte granules that occur in melanoma are recognizable with dermoscopy.6
In 2001, Bafounta and colleagues performed the first meta-analysis on the efficacy of dermoscopy compared with that of clinical evaluation, finding that dermoscopy performed specifically by dermatology-trained clinicians improved the accuracy of identifying melanoma from an odds ratio of 16 (95% CI, 9-31) with naked eye examination to 76 (95% CI, 25-223) with dermoscopy.7
More recently, Terushkin and colleagues demonstrated that diagnosis specificity improves when a general dermatologist is trained in dermoscopic pattern recognition. Naked eye examination produced a benign to malignant ratio (BMR) of 18.4:1, indicating that about 18 of 19 biopsies considered suspicious for melanoma ultimately yielded benign melanocytic lesions. Although the BMR for the general dermatologist experienced an increase after dermoscopy training, the ratio eventually decreased to 7.9:1.8
Dermoscopic Analysis
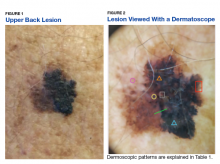
Some of the common patterns recognized in melanoma are demonstrated in Figures 1 and 2. Figure 1 is a close-up of a patient’s upper back showing a solitary asymmetric melanocytic lesion containing multiple red, brown, black, and blue hues.
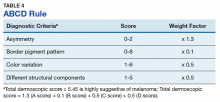
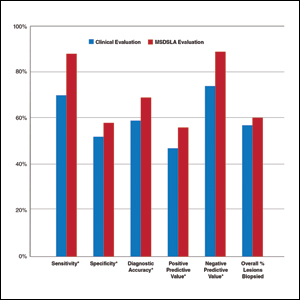
Pattern analysis, the dermoscopic interpretation method preferred by pigmented lesion specialists, requires simultaneously assessing numerous lesion patterns that vary depending on body site.10 Alternative dermoscopic algorithms that focus on the most common features of melanoma have been developed to aid practitioners with the interpretation of dermoscopy findings: the 7-point checklist, the Menzies method, the ABCD rule, and the CASH algorithm (Tables 2, 3, 4, and 5).
Argenziano and colleagues developed the 7-point checklist in 1998. Two points are assigned to the lesion for each of the major criteria and 1 point for each minor criteria.
The Menzies method was developed by Menzies and colleagues in 1996. To be classified as melanoma, the pigmented lesion must show an absence of pattern symmetry and color uniformity while simultaneously exhibiting at least one of the following: blue-white veil, multiple brown dots, pseudopods, radial streaming, scarlike depigmentation, peripheral block dots/globules, 5 to 6 colors, multiple blue/gray dots, and a broadened network.12
The ABCD rule is a more technical dermoscopic evaluation algorithm developed in 1994 by Stolz and colleagues. This method yields a numeric value called the total dermoscopic score (TDS) based on Asymmetry, Border pigment pattern, Color variation, and 5 Different structural components.
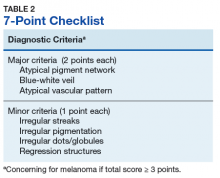
Henning and colleagues developed the CASH algorithm in 2006 with the intention of assisting less experienced dermoscopy users with lesion evaluation.14 This algorithm tallies points for Color, Architectural disorder, Symmetry, and Homogeneity. One point is attributed to a lesion for each light brown, dark brown, black, red, white, and/or blue region present. Architectural disorder is assigned a point value between 0 and 2, with 0 indicating the absence of or minimal architectural disorder, 1 indicating moderate disorder, and 2 indicating marked disorder. Symmetry is assigned a point value between 0 and 2, with 0 points assigned to a lesion that exhibits biaxial symmetry, 1 point assigned to a lesion that exhibits monoaxial symmetry, and 2 points assigned to a lesion that exhibits biaxial asymmetry. Finally, 1 point is attributed to a lesion for evidence of each of the following: atypical network, dots/globules, streaks/pseudopods, blue-white veil, regression structures, blotches > 10% of the overall lesion size, and polymorphous blood vessels. The lesion in Figure 2 scores 16 points out of the maximum total CASH score of 17. Any lesion scoring 8 or more is suggestive of malignant melanoma.14
Finally, the TADA method was developed by Rogers and colleagues in 2016.15 This method uses sequential questions to evaluate lesions. First, “Does the lesion exhibit clear dermoscopic evidence of an angioma, dermatofibroma, or seborrheic keratosis?” If “yes,” then no additional dermoscopic evaluation is necessary, and it is recommended to monitor the lesion. If the answer to the first question is “no,” then ask, “Does the lesion exhibit architectural disorder?” The presence of architectural disorder is based on an overall impression of the lesion, which includes symmetry with regard to structures and colors. Any lesion deemed to exhibit architectural disorder should be biopsied. If the lesion has no architectural disorder, the third question is, “Does the lesion contain any starburst patterns, blue-black or gray coloration, shiny white structures, negative networks, ulcers or erosions, and/or vessels?” If “yes,” then the lesion should be biopsied. Since the lesion in Figure 2 exhibits marked architectural disorder in terms of symmetry and color, analysis of the lesion with the TADA method would yield a recommendation for biopsy.15
Dermoscopy in Practice
A. Bernard Ackerman, MD, a key figure in the modern era of dermatopathology, wrote an editorial for the Journal of the American Academy of Dermatology in 1985 titled “No one should die of malignant melanoma.” The editorial highlighted that the visual changes associated with melanoma often manifest years prior to malignant invasion and advocated that all physicians should have competence in melanoma detection, specifically mentioning the importance of training primary care physicians (PCPs), dermatologists, and pathologists in this regard.16 This sentiment is equally as true now as it was in 1985.
Naked eye examination paired with an evaluation of patient risk factors for melanoma, including fair skin types, significant sun exposure history, history of sunburn, geographic location, and personal and family history of melanoma, are the foundation of melanoma detection efforts.17 Studies suggest that the triage skills of PCPs could be improved in regard to the evaluation of pigmented lesions. Primary care residents, for instance, did not accurately diagnose 40% of malignant melanoma cases.18,19 Additionally, a meta-analysis demonstrated that PCP accuracy when diagnosing malignant melanoma ranged between 49% and 80%, significantly less than the 85% to 89% exhibited by practicing dermatologists.19 Dermoscopy could be incorporated as an element of the skin examination to enhance lesion discrimination among PCPs, as it has proven use in dermatologic practice.
Dermoscopy is not readily used by PCPs. A survey study of 705 family practitioners in the US performed by Morris and colleagues demonstrated that only 8.3% of participants currently use a dermatoscope to evaluate pigmented lesions.20 The most common barriers to dermoscopy use cited by PCPs in the US include the cost of the dermatoscope, the time required to acquire proficiency, and the lack of financial reimbursement.16 True utilization of dermoscopy among PCPs is higher than this figure suggests due to the number of PCPs who access dermoscopic evaluations via teledermatology. All 21 Veterans Integrated Services Networks of the Veterans Health Administration (VHA) system, for instance, participate in teledermatology and jointly employ more than 1,150 trained telehealth clinical technicians who created a collective 107,000 teledermatology encounters with and without dermoscopy for evaluation by dermatologists in the most recent fiscal year(Martin Weinstock, written communication, October 2017). Nonetheless, it is necessary to determine the contribution that wider utilization of dermoscopy among PCPs would have on melanoma surveillance.
Studies show that dermoscopic algorithms improve the sensitivity while slightly decreasing the specificity of PCPs to detect melanoma compared with that of the naked eye examination. Dolianitis and colleagues demonstrated that a baseline sensitivity of 60.9% for melanoma detection improved to 85.4% with the 7-point checklist, 85.4% with Menzies method, and 77.5% with the ABCD rule, while the baseline specificity of 85.4% moderated to 73.0%, 77.7%, and 80.4%, respectively, among 61 medical practitioners after studying dermoscopy techniques from 2 CDs.21 Westerhoff and colleagues performed a randomized controlled trial with 74 PCPs to determine the effect of a minimal intervention on melanoma diagnostic accuracy. The intervention consisted of providing participants in the experimental group with an atlas of microscopic features common to melanoma to be read at the participants’ leisure, a 1-hour presentation on microscopy, and a 25-questionpractice quiz. Participants randomized to the intervention group improved their diagnostic accuracy from 57.8% to 75.9% with the use of dermoscopy. This group also experiencedimproved accuracy in its clinical diagnosis of melanoma from 54.6% to 62.7%.22
Argenziano and colleagues demonstrated similar results after PCPs attended a 4-hour workshop on dermoscopy. The 73 PCPs in this study evaluated 2,522 lesions randomized to naked eye examination or dermoscopy. The BMR, calculated from the data provided, improved from 12.6:1 to 10.5:1, respectively, when dermoscopy was incorporated into lesion analysis, while the sensitivity increased from 54.1% to 79.2% and the negative predictive value increased from 95.8% to 98.1%. It is important to note that the BMR and negative predictive value improved in tandem, indicating that PCPs were more discriminatory with their referrals for evaluation by dermatology while capturing a greater percentage of melanomas.23
These studies are not without limitations that preclude broad generalizations. For example, Dolianitis and colleagues and Westerhoff and colleagues provided participants with dermoscopic images of the lesions to be evaluated instead of requiring personal use of a dermatoscope, whereas the study by Argenziano and colleagues incorporated only 6 histopathologically proven malignant melanomas into each of the naked eye examination and the experimental dermoscopy groups.21-23 Yet these studies suggest that broader use of dermoscopy among PCPs could improve the accuracy of melanoma detection given clinically relevant training.
Several additional studies identify positive correlations associated with dermoscopy use among PCPs. A recent survey of 425 French general practitioners found that 8% of the study participants acknowledged owning a dermatoscope. Dermatoscope owners spent a statistically significant longer time analyzing each pigmented skin lesions, exhibited greater confidence in their analysis of pigmented lesions, and issued fewer overall referrals to dermatologists.24
Similarly, Rosendahl and colleagues evaluated the number needed to treat (NNT) (equivalent to the BMR) among 193 Australian PCPs and found that the NNT was inversely correlated to the frequency with which the physicians used dermoscopy. However, it was difficult to determine the definitive cause of the reduced NNT in this study because a similar effect was observed when NNT was evaluated based on general practitioner subspecialization.25 Again, despite limitations, these studies suggest that increased dermoscopy use among PCPs could reduce the morbidity of lifelong scarring as well as the short-term anxiety associated with a possible melanoma diagnosis.
Limitations
Even in the hands of a trained dermatologist, dermoscopy has limitations. Featureless melanoma is a term applied to melanoma lesions bereft of classical findings on both naked eye examination and dermoscopy. Menzies, a dermatologic pioneer in dermoscopy, acknowledged this limitation in 1996 while showing that 8% of melanomas evaded dermoscopic detection. He proceeded to discuss the importance of clinical history in melanoma detection because all of the featureless melanomas exhibited recent changes in size, shape, and/or color.26 More recently, sequential dermoscopy (mole mapping) imaging has been implemented to successfully identify these lesions.27 Thus, dermoscopy cannot replace dermatologists trained in the art of visual assessment with honed clinical diagnostic acumen. Rather, dermoscopy is a tool to enhance the assessment of clinically suspicious lesions and aid diagnostic discrimination of uncertain pigmented lesions.
Conclusion
Primary care physicians are on the frontline of medicine and often the first to have the opportunity to detect the presence of melanoma. Notably, 52.2% of the 884.7 million medical office visits performed annually in the US are with PCPs.28 Despite the benefits, dermoscopy is not uniformly used by dermatologists in the US. Of dermatologists practicing for more than 20 years, 76.2% use dermoscopy compared with 97.8% of dermatologists in practice for less than 5 years. This illustrates an increased use in tandem with dermatology residencies integrating dermoscopy training as a component of the curriculum, showing the importance of incorporating dermoscopy into medical school and residency training for PCPs..29-31 Guidelines regarding dermoscopy training and dermoscopic evaluation algorithms should be established, routinely taught in medical education, and actively incorporated into training curriculum for PCPs in order to improve patient care and realize the potential health care savings associated with the early diagnosis and treatment of melanoma. Dermoscopic-teledermatology consultations present a viable opportunity within the VHA to expedite access to care for veterans and simultaneously offer evaluative feedback on lesions to referring PCPs, as skilled, dermoscopy-trained dermatologists render the diagnoses. Given the devastating mortality rate of melanoma, continued multidisciplinary education on identifying melanoma is of the utmost importance for patient care. Widespread implementation of dermoscopy and dermoscopic-teledermatology consultations could save lives and slow the ever-increasing economic burden associated with melanoma treatment, costing $1.467 billion in 2016.32
1. Guy GP Jr, Thomas CC, Thompson T, Watson M, Massetti GM, Richardson LC. Vital signs: melanoma incidence and mortality trends and projections-United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64(21):591-596.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7-30.
3. American Cancer Society. Cancer facts & figures 2017. Atlanta: American Cancer Society; 2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed April 19, 2018.
4. Lea CS, Efird JT, Toland AE, Lewis DR, Phillips CJ. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179(3):247-253.
5. Thomas L, Puig S. Dermoscopy, digital dermoscopy and other diagnostic tools in the early detection of melanoma and follow-up of high-risk skin cancer patients. Acta Derm Venereol. 2017;97(218):14-21.
6. Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88(7):441-450.
7. Bafounta ML, Beauchet A, Aegerter P, Saiag P. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma? Results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137(10):1343-1350.
8. Terushkin V, Warycha M, Levy M, Kopf AW, Cohen DE, Polsky D. Analysis of the benign to malignant ratio of lesions biopsied by a general dermatologist before and after the adoption of dermoscopy. Arch Dermatol. 2010;146(3):343-344.
9. Wolner ZJ, Yélamos O, Liopyris K, Rogers T, Marchetti MA, Marghoob AA. Enhancing skin cancer diagnosis with dermoscopy. Dermatol Clin. 2017;35(4):417-437.
10. Carli P, Quercioli E, Sestini S, et al. Pattern analysis, not simplified algorithms, is the most reliable method for teaching dermoscopy for melanoma diagnosis to residents in dermatology. Br J Dermatol. 2003;148(5):981-984.
11. Argenziano G, Fabbrocini G, Carli P, De Giorgi V, Sammarco E, Delfino M. Epiluminescence microscopy for the diagnosis of doubtful melanocytic skin lesions. Comparison of the ABCD rule of dermatoscopy and a new 7-point checklist based on pattern analysis. Arch Dermatol. 1998;134(12):1563-1570.
12. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
13. Nachbar F, Stolz W, Merkle T, et al. The ABCD rule of dermatoscopy. High prospective value in the diagnosis of doubtful melanocytic skin lesions. J Am Acad Dermatol. 1994;30(4):551-559.
14. Henning JS, Dusza SW, Wang SQ, et al. The CASH (color, architecture, symmetry, and homogeneity) algorithm for dermoscopy. J Am Acad Dermatol. 2007;56(1):45-52.
15. Rogers T, Marino M, Dusza SW, Bajaj S, Marchetti MA, Marghoob A. Triage amalgamated dermoscopic algorithm (TADA) for skin cancer screening. Dermatol Pract Concept. 2017;7(2):39-46.
16. Ackerman AB. No one should die of malignant melanoma. J Am Acad Dermatol. 1985;12(1):115-116.
17. Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II: sun exposure. Eur J Cancer. 2005;41(1):45-60.
18. Gerbert B, Maurer T, Berger T, et al. Primary care physicians as gatekeepers in managed care. Primary care physicians’ and dermatologists’ skills at secondary prevention of skin cancer. Arch Dermatol. 1996;132(9):1030-1038.
19. Corbo MD, Wismer J. Agreement between dermatologists and primary care practitioners in the diagnosis of malignant melanoma: review of the literature. J Cutan Med Surg. 2012;16(5):306-310.
20. Morris JB, Alfonso SV, Hernandez N, Fernández MI. Examining the factors associated with past and present dermoscopy use among family physicians. Dermatol Pract Concept. 2017;7(4):63-70.
21. Dolianitis C, Kelly J, Wolfe R, Simpson P. Comparative performance of 4 dermoscopic algorithms by nonexperts for the diagnosis of melanocytic lesions. Arch Dermatol. 2005;141(8):1008-1014.
22. Westerhoff K, Mccarthy WH, Menzies SW. Increase in the sensitivity for melanoma diagnosis by primary care physicians using skin surface microscopy. Br J Dermatol. 2000;143(5):1016-1020.
23. Argenziano G, Puig S, Zalaudek I, et al. Dermoscopy improves accuracy of primary care physicians to triage lesions suggestive of skin cancer. J Clin Oncol. 2006;24(12):1877-1882.
24. Chappuis P, Duru G, Marchal O, Girier P, Dalle S, Thomas L. Dermoscopy, a useful tool for general practitioners in melanoma screening: a nationwide survey. Br J Dermatol. 2016;175(4):744-750.
25. Rosendahl C, Williams G, Eley D, et al. The impact of subspecialization and dermatoscopy use on accuracy of melanoma diagnosis among primary care doctors in Australia. J Am Acad Dermatol. 2012;67(5):846-852.
26. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
27. Kittler H, Guitera P, Riedl E, et al. Identification of clinically featureless incipient melanoma using sequential dermoscopy imaging. Arch Dermatol. 2006;142(9):1113-1119.
28. Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. https://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated May 3, 2017. Accessed April 10, 2018.
29. Murzaku EC, Hayan S, Rao BK. Methods and rates of dermoscopy usage: a cross-sectional survey of US dermatologists stratified by years in practice. J Am Acad Dermatol. 2014;71(2):393-395.
30. Nehal KS, Oliveria SA, Marghoob AA, et al. Use of and beliefs about dermoscopy in the management of patients with pigmented lesions: a survey of dermatology residency programmes in the United States. Melanoma Res. 2002;12(6):601-605.
31. Wu TP, Newlove T, Smith L, Vuong CH, Stein JA, Polsky D. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68(6):1000-1005.
32. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76(5):958-972
From 1982 to 2011, the melanoma incidence rate doubled in the US.1 In 2018, an estimated 87,290 cases of melanoma in situ and 91,270 cases of invasive melanoma will be diagnosed in the US, and 9,320 deaths will be attributable to melanoma.2 Early detection of melanoma is critically important to reduce melanoma-related mortality, with 5-year survival rates as high as 97% at stage 1A vs a 20% 5-year survival when there is distant metastasis.2,3 Melanoma is particularly relevant for medical providers working with veterans because melanoma disproportionately affects service members with an incidence rate ratio of 1.62 (95% confidence interval [CI], 1.40-1.86) compared with that of the general population.4
Biopsy is the definitive diagnostic tool for melanoma. Histologic analysis differentiates melanoma from seborrheic keratoses, pigmented nevi, dermatofibromas, and other pigmented lesions that can resemble melanoma on clinical examination. However, biopsy must be used judiciously as unnecessary biopsies contribute to health care costs and leave scars, which can have psychosocial implications. With benign nevi outnumbering melanoma about 2 million to 1, biopsy is indicated once a threshold of suspicion is obtained.5
Dermoscopic Tool
Dermoscopy is a microscopy-based tool to improve noninvasive diagnostic discrimination of skin lesions based on color and structure analysis. Coloration provides an indication of the composition of elements present in the skin with keratin appearing yellow, blood appearing red, and collagen appearing white. Coloration also suggests pigment depth as melanin appears black when located in the stratum corneum, brown when located deeper in the epidermis, and blue when located in the dermis.6 Finally, characteristic histopathologic alterations in the dermoepidermal junction, rete ridges, pigment-containing cells, and/or melanocyte granules that occur in melanoma are recognizable with dermoscopy.6
In 2001, Bafounta and colleagues performed the first meta-analysis on the efficacy of dermoscopy compared with that of clinical evaluation, finding that dermoscopy performed specifically by dermatology-trained clinicians improved the accuracy of identifying melanoma from an odds ratio of 16 (95% CI, 9-31) with naked eye examination to 76 (95% CI, 25-223) with dermoscopy.7
More recently, Terushkin and colleagues demonstrated that diagnosis specificity improves when a general dermatologist is trained in dermoscopic pattern recognition. Naked eye examination produced a benign to malignant ratio (BMR) of 18.4:1, indicating that about 18 of 19 biopsies considered suspicious for melanoma ultimately yielded benign melanocytic lesions. Although the BMR for the general dermatologist experienced an increase after dermoscopy training, the ratio eventually decreased to 7.9:1.8
Dermoscopic Analysis
Some of the common patterns recognized in melanoma are demonstrated in Figures 1 and 2. Figure 1 is a close-up of a patient’s upper back showing a solitary asymmetric melanocytic lesion containing multiple red, brown, black, and blue hues.
Pattern analysis, the dermoscopic interpretation method preferred by pigmented lesion specialists, requires simultaneously assessing numerous lesion patterns that vary depending on body site.10 Alternative dermoscopic algorithms that focus on the most common features of melanoma have been developed to aid practitioners with the interpretation of dermoscopy findings: the 7-point checklist, the Menzies method, the ABCD rule, and the CASH algorithm (Tables 2, 3, 4, and 5).
Argenziano and colleagues developed the 7-point checklist in 1998. Two points are assigned to the lesion for each of the major criteria and 1 point for each minor criteria.
The Menzies method was developed by Menzies and colleagues in 1996. To be classified as melanoma, the pigmented lesion must show an absence of pattern symmetry and color uniformity while simultaneously exhibiting at least one of the following: blue-white veil, multiple brown dots, pseudopods, radial streaming, scarlike depigmentation, peripheral block dots/globules, 5 to 6 colors, multiple blue/gray dots, and a broadened network.12
The ABCD rule is a more technical dermoscopic evaluation algorithm developed in 1994 by Stolz and colleagues. This method yields a numeric value called the total dermoscopic score (TDS) based on Asymmetry, Border pigment pattern, Color variation, and 5 Different structural components.
Henning and colleagues developed the CASH algorithm in 2006 with the intention of assisting less experienced dermoscopy users with lesion evaluation.14 This algorithm tallies points for Color, Architectural disorder, Symmetry, and Homogeneity. One point is attributed to a lesion for each light brown, dark brown, black, red, white, and/or blue region present. Architectural disorder is assigned a point value between 0 and 2, with 0 indicating the absence of or minimal architectural disorder, 1 indicating moderate disorder, and 2 indicating marked disorder. Symmetry is assigned a point value between 0 and 2, with 0 points assigned to a lesion that exhibits biaxial symmetry, 1 point assigned to a lesion that exhibits monoaxial symmetry, and 2 points assigned to a lesion that exhibits biaxial asymmetry. Finally, 1 point is attributed to a lesion for evidence of each of the following: atypical network, dots/globules, streaks/pseudopods, blue-white veil, regression structures, blotches > 10% of the overall lesion size, and polymorphous blood vessels. The lesion in Figure 2 scores 16 points out of the maximum total CASH score of 17. Any lesion scoring 8 or more is suggestive of malignant melanoma.14
Finally, the TADA method was developed by Rogers and colleagues in 2016.15 This method uses sequential questions to evaluate lesions. First, “Does the lesion exhibit clear dermoscopic evidence of an angioma, dermatofibroma, or seborrheic keratosis?” If “yes,” then no additional dermoscopic evaluation is necessary, and it is recommended to monitor the lesion. If the answer to the first question is “no,” then ask, “Does the lesion exhibit architectural disorder?” The presence of architectural disorder is based on an overall impression of the lesion, which includes symmetry with regard to structures and colors. Any lesion deemed to exhibit architectural disorder should be biopsied. If the lesion has no architectural disorder, the third question is, “Does the lesion contain any starburst patterns, blue-black or gray coloration, shiny white structures, negative networks, ulcers or erosions, and/or vessels?” If “yes,” then the lesion should be biopsied. Since the lesion in Figure 2 exhibits marked architectural disorder in terms of symmetry and color, analysis of the lesion with the TADA method would yield a recommendation for biopsy.15
Dermoscopy in Practice
A. Bernard Ackerman, MD, a key figure in the modern era of dermatopathology, wrote an editorial for the Journal of the American Academy of Dermatology in 1985 titled “No one should die of malignant melanoma.” The editorial highlighted that the visual changes associated with melanoma often manifest years prior to malignant invasion and advocated that all physicians should have competence in melanoma detection, specifically mentioning the importance of training primary care physicians (PCPs), dermatologists, and pathologists in this regard.16 This sentiment is equally as true now as it was in 1985.
Naked eye examination paired with an evaluation of patient risk factors for melanoma, including fair skin types, significant sun exposure history, history of sunburn, geographic location, and personal and family history of melanoma, are the foundation of melanoma detection efforts.17 Studies suggest that the triage skills of PCPs could be improved in regard to the evaluation of pigmented lesions. Primary care residents, for instance, did not accurately diagnose 40% of malignant melanoma cases.18,19 Additionally, a meta-analysis demonstrated that PCP accuracy when diagnosing malignant melanoma ranged between 49% and 80%, significantly less than the 85% to 89% exhibited by practicing dermatologists.19 Dermoscopy could be incorporated as an element of the skin examination to enhance lesion discrimination among PCPs, as it has proven use in dermatologic practice.
Dermoscopy is not readily used by PCPs. A survey study of 705 family practitioners in the US performed by Morris and colleagues demonstrated that only 8.3% of participants currently use a dermatoscope to evaluate pigmented lesions.20 The most common barriers to dermoscopy use cited by PCPs in the US include the cost of the dermatoscope, the time required to acquire proficiency, and the lack of financial reimbursement.16 True utilization of dermoscopy among PCPs is higher than this figure suggests due to the number of PCPs who access dermoscopic evaluations via teledermatology. All 21 Veterans Integrated Services Networks of the Veterans Health Administration (VHA) system, for instance, participate in teledermatology and jointly employ more than 1,150 trained telehealth clinical technicians who created a collective 107,000 teledermatology encounters with and without dermoscopy for evaluation by dermatologists in the most recent fiscal year(Martin Weinstock, written communication, October 2017). Nonetheless, it is necessary to determine the contribution that wider utilization of dermoscopy among PCPs would have on melanoma surveillance.
Studies show that dermoscopic algorithms improve the sensitivity while slightly decreasing the specificity of PCPs to detect melanoma compared with that of the naked eye examination. Dolianitis and colleagues demonstrated that a baseline sensitivity of 60.9% for melanoma detection improved to 85.4% with the 7-point checklist, 85.4% with Menzies method, and 77.5% with the ABCD rule, while the baseline specificity of 85.4% moderated to 73.0%, 77.7%, and 80.4%, respectively, among 61 medical practitioners after studying dermoscopy techniques from 2 CDs.21 Westerhoff and colleagues performed a randomized controlled trial with 74 PCPs to determine the effect of a minimal intervention on melanoma diagnostic accuracy. The intervention consisted of providing participants in the experimental group with an atlas of microscopic features common to melanoma to be read at the participants’ leisure, a 1-hour presentation on microscopy, and a 25-questionpractice quiz. Participants randomized to the intervention group improved their diagnostic accuracy from 57.8% to 75.9% with the use of dermoscopy. This group also experiencedimproved accuracy in its clinical diagnosis of melanoma from 54.6% to 62.7%.22
Argenziano and colleagues demonstrated similar results after PCPs attended a 4-hour workshop on dermoscopy. The 73 PCPs in this study evaluated 2,522 lesions randomized to naked eye examination or dermoscopy. The BMR, calculated from the data provided, improved from 12.6:1 to 10.5:1, respectively, when dermoscopy was incorporated into lesion analysis, while the sensitivity increased from 54.1% to 79.2% and the negative predictive value increased from 95.8% to 98.1%. It is important to note that the BMR and negative predictive value improved in tandem, indicating that PCPs were more discriminatory with their referrals for evaluation by dermatology while capturing a greater percentage of melanomas.23
These studies are not without limitations that preclude broad generalizations. For example, Dolianitis and colleagues and Westerhoff and colleagues provided participants with dermoscopic images of the lesions to be evaluated instead of requiring personal use of a dermatoscope, whereas the study by Argenziano and colleagues incorporated only 6 histopathologically proven malignant melanomas into each of the naked eye examination and the experimental dermoscopy groups.21-23 Yet these studies suggest that broader use of dermoscopy among PCPs could improve the accuracy of melanoma detection given clinically relevant training.
Several additional studies identify positive correlations associated with dermoscopy use among PCPs. A recent survey of 425 French general practitioners found that 8% of the study participants acknowledged owning a dermatoscope. Dermatoscope owners spent a statistically significant longer time analyzing each pigmented skin lesions, exhibited greater confidence in their analysis of pigmented lesions, and issued fewer overall referrals to dermatologists.24
Similarly, Rosendahl and colleagues evaluated the number needed to treat (NNT) (equivalent to the BMR) among 193 Australian PCPs and found that the NNT was inversely correlated to the frequency with which the physicians used dermoscopy. However, it was difficult to determine the definitive cause of the reduced NNT in this study because a similar effect was observed when NNT was evaluated based on general practitioner subspecialization.25 Again, despite limitations, these studies suggest that increased dermoscopy use among PCPs could reduce the morbidity of lifelong scarring as well as the short-term anxiety associated with a possible melanoma diagnosis.
Limitations
Even in the hands of a trained dermatologist, dermoscopy has limitations. Featureless melanoma is a term applied to melanoma lesions bereft of classical findings on both naked eye examination and dermoscopy. Menzies, a dermatologic pioneer in dermoscopy, acknowledged this limitation in 1996 while showing that 8% of melanomas evaded dermoscopic detection. He proceeded to discuss the importance of clinical history in melanoma detection because all of the featureless melanomas exhibited recent changes in size, shape, and/or color.26 More recently, sequential dermoscopy (mole mapping) imaging has been implemented to successfully identify these lesions.27 Thus, dermoscopy cannot replace dermatologists trained in the art of visual assessment with honed clinical diagnostic acumen. Rather, dermoscopy is a tool to enhance the assessment of clinically suspicious lesions and aid diagnostic discrimination of uncertain pigmented lesions.
Conclusion
Primary care physicians are on the frontline of medicine and often the first to have the opportunity to detect the presence of melanoma. Notably, 52.2% of the 884.7 million medical office visits performed annually in the US are with PCPs.28 Despite the benefits, dermoscopy is not uniformly used by dermatologists in the US. Of dermatologists practicing for more than 20 years, 76.2% use dermoscopy compared with 97.8% of dermatologists in practice for less than 5 years. This illustrates an increased use in tandem with dermatology residencies integrating dermoscopy training as a component of the curriculum, showing the importance of incorporating dermoscopy into medical school and residency training for PCPs..29-31 Guidelines regarding dermoscopy training and dermoscopic evaluation algorithms should be established, routinely taught in medical education, and actively incorporated into training curriculum for PCPs in order to improve patient care and realize the potential health care savings associated with the early diagnosis and treatment of melanoma. Dermoscopic-teledermatology consultations present a viable opportunity within the VHA to expedite access to care for veterans and simultaneously offer evaluative feedback on lesions to referring PCPs, as skilled, dermoscopy-trained dermatologists render the diagnoses. Given the devastating mortality rate of melanoma, continued multidisciplinary education on identifying melanoma is of the utmost importance for patient care. Widespread implementation of dermoscopy and dermoscopic-teledermatology consultations could save lives and slow the ever-increasing economic burden associated with melanoma treatment, costing $1.467 billion in 2016.32
From 1982 to 2011, the melanoma incidence rate doubled in the US.1 In 2018, an estimated 87,290 cases of melanoma in situ and 91,270 cases of invasive melanoma will be diagnosed in the US, and 9,320 deaths will be attributable to melanoma.2 Early detection of melanoma is critically important to reduce melanoma-related mortality, with 5-year survival rates as high as 97% at stage 1A vs a 20% 5-year survival when there is distant metastasis.2,3 Melanoma is particularly relevant for medical providers working with veterans because melanoma disproportionately affects service members with an incidence rate ratio of 1.62 (95% confidence interval [CI], 1.40-1.86) compared with that of the general population.4
Biopsy is the definitive diagnostic tool for melanoma. Histologic analysis differentiates melanoma from seborrheic keratoses, pigmented nevi, dermatofibromas, and other pigmented lesions that can resemble melanoma on clinical examination. However, biopsy must be used judiciously as unnecessary biopsies contribute to health care costs and leave scars, which can have psychosocial implications. With benign nevi outnumbering melanoma about 2 million to 1, biopsy is indicated once a threshold of suspicion is obtained.5
Dermoscopic Tool
Dermoscopy is a microscopy-based tool to improve noninvasive diagnostic discrimination of skin lesions based on color and structure analysis. Coloration provides an indication of the composition of elements present in the skin with keratin appearing yellow, blood appearing red, and collagen appearing white. Coloration also suggests pigment depth as melanin appears black when located in the stratum corneum, brown when located deeper in the epidermis, and blue when located in the dermis.6 Finally, characteristic histopathologic alterations in the dermoepidermal junction, rete ridges, pigment-containing cells, and/or melanocyte granules that occur in melanoma are recognizable with dermoscopy.6
In 2001, Bafounta and colleagues performed the first meta-analysis on the efficacy of dermoscopy compared with that of clinical evaluation, finding that dermoscopy performed specifically by dermatology-trained clinicians improved the accuracy of identifying melanoma from an odds ratio of 16 (95% CI, 9-31) with naked eye examination to 76 (95% CI, 25-223) with dermoscopy.7
More recently, Terushkin and colleagues demonstrated that diagnosis specificity improves when a general dermatologist is trained in dermoscopic pattern recognition. Naked eye examination produced a benign to malignant ratio (BMR) of 18.4:1, indicating that about 18 of 19 biopsies considered suspicious for melanoma ultimately yielded benign melanocytic lesions. Although the BMR for the general dermatologist experienced an increase after dermoscopy training, the ratio eventually decreased to 7.9:1.8
Dermoscopic Analysis
Some of the common patterns recognized in melanoma are demonstrated in Figures 1 and 2. Figure 1 is a close-up of a patient’s upper back showing a solitary asymmetric melanocytic lesion containing multiple red, brown, black, and blue hues.
Pattern analysis, the dermoscopic interpretation method preferred by pigmented lesion specialists, requires simultaneously assessing numerous lesion patterns that vary depending on body site.10 Alternative dermoscopic algorithms that focus on the most common features of melanoma have been developed to aid practitioners with the interpretation of dermoscopy findings: the 7-point checklist, the Menzies method, the ABCD rule, and the CASH algorithm (Tables 2, 3, 4, and 5).
Argenziano and colleagues developed the 7-point checklist in 1998. Two points are assigned to the lesion for each of the major criteria and 1 point for each minor criteria.
The Menzies method was developed by Menzies and colleagues in 1996. To be classified as melanoma, the pigmented lesion must show an absence of pattern symmetry and color uniformity while simultaneously exhibiting at least one of the following: blue-white veil, multiple brown dots, pseudopods, radial streaming, scarlike depigmentation, peripheral block dots/globules, 5 to 6 colors, multiple blue/gray dots, and a broadened network.12
The ABCD rule is a more technical dermoscopic evaluation algorithm developed in 1994 by Stolz and colleagues. This method yields a numeric value called the total dermoscopic score (TDS) based on Asymmetry, Border pigment pattern, Color variation, and 5 Different structural components.
Henning and colleagues developed the CASH algorithm in 2006 with the intention of assisting less experienced dermoscopy users with lesion evaluation.14 This algorithm tallies points for Color, Architectural disorder, Symmetry, and Homogeneity. One point is attributed to a lesion for each light brown, dark brown, black, red, white, and/or blue region present. Architectural disorder is assigned a point value between 0 and 2, with 0 indicating the absence of or minimal architectural disorder, 1 indicating moderate disorder, and 2 indicating marked disorder. Symmetry is assigned a point value between 0 and 2, with 0 points assigned to a lesion that exhibits biaxial symmetry, 1 point assigned to a lesion that exhibits monoaxial symmetry, and 2 points assigned to a lesion that exhibits biaxial asymmetry. Finally, 1 point is attributed to a lesion for evidence of each of the following: atypical network, dots/globules, streaks/pseudopods, blue-white veil, regression structures, blotches > 10% of the overall lesion size, and polymorphous blood vessels. The lesion in Figure 2 scores 16 points out of the maximum total CASH score of 17. Any lesion scoring 8 or more is suggestive of malignant melanoma.14
Finally, the TADA method was developed by Rogers and colleagues in 2016.15 This method uses sequential questions to evaluate lesions. First, “Does the lesion exhibit clear dermoscopic evidence of an angioma, dermatofibroma, or seborrheic keratosis?” If “yes,” then no additional dermoscopic evaluation is necessary, and it is recommended to monitor the lesion. If the answer to the first question is “no,” then ask, “Does the lesion exhibit architectural disorder?” The presence of architectural disorder is based on an overall impression of the lesion, which includes symmetry with regard to structures and colors. Any lesion deemed to exhibit architectural disorder should be biopsied. If the lesion has no architectural disorder, the third question is, “Does the lesion contain any starburst patterns, blue-black or gray coloration, shiny white structures, negative networks, ulcers or erosions, and/or vessels?” If “yes,” then the lesion should be biopsied. Since the lesion in Figure 2 exhibits marked architectural disorder in terms of symmetry and color, analysis of the lesion with the TADA method would yield a recommendation for biopsy.15
Dermoscopy in Practice
A. Bernard Ackerman, MD, a key figure in the modern era of dermatopathology, wrote an editorial for the Journal of the American Academy of Dermatology in 1985 titled “No one should die of malignant melanoma.” The editorial highlighted that the visual changes associated with melanoma often manifest years prior to malignant invasion and advocated that all physicians should have competence in melanoma detection, specifically mentioning the importance of training primary care physicians (PCPs), dermatologists, and pathologists in this regard.16 This sentiment is equally as true now as it was in 1985.
Naked eye examination paired with an evaluation of patient risk factors for melanoma, including fair skin types, significant sun exposure history, history of sunburn, geographic location, and personal and family history of melanoma, are the foundation of melanoma detection efforts.17 Studies suggest that the triage skills of PCPs could be improved in regard to the evaluation of pigmented lesions. Primary care residents, for instance, did not accurately diagnose 40% of malignant melanoma cases.18,19 Additionally, a meta-analysis demonstrated that PCP accuracy when diagnosing malignant melanoma ranged between 49% and 80%, significantly less than the 85% to 89% exhibited by practicing dermatologists.19 Dermoscopy could be incorporated as an element of the skin examination to enhance lesion discrimination among PCPs, as it has proven use in dermatologic practice.
Dermoscopy is not readily used by PCPs. A survey study of 705 family practitioners in the US performed by Morris and colleagues demonstrated that only 8.3% of participants currently use a dermatoscope to evaluate pigmented lesions.20 The most common barriers to dermoscopy use cited by PCPs in the US include the cost of the dermatoscope, the time required to acquire proficiency, and the lack of financial reimbursement.16 True utilization of dermoscopy among PCPs is higher than this figure suggests due to the number of PCPs who access dermoscopic evaluations via teledermatology. All 21 Veterans Integrated Services Networks of the Veterans Health Administration (VHA) system, for instance, participate in teledermatology and jointly employ more than 1,150 trained telehealth clinical technicians who created a collective 107,000 teledermatology encounters with and without dermoscopy for evaluation by dermatologists in the most recent fiscal year(Martin Weinstock, written communication, October 2017). Nonetheless, it is necessary to determine the contribution that wider utilization of dermoscopy among PCPs would have on melanoma surveillance.
Studies show that dermoscopic algorithms improve the sensitivity while slightly decreasing the specificity of PCPs to detect melanoma compared with that of the naked eye examination. Dolianitis and colleagues demonstrated that a baseline sensitivity of 60.9% for melanoma detection improved to 85.4% with the 7-point checklist, 85.4% with Menzies method, and 77.5% with the ABCD rule, while the baseline specificity of 85.4% moderated to 73.0%, 77.7%, and 80.4%, respectively, among 61 medical practitioners after studying dermoscopy techniques from 2 CDs.21 Westerhoff and colleagues performed a randomized controlled trial with 74 PCPs to determine the effect of a minimal intervention on melanoma diagnostic accuracy. The intervention consisted of providing participants in the experimental group with an atlas of microscopic features common to melanoma to be read at the participants’ leisure, a 1-hour presentation on microscopy, and a 25-questionpractice quiz. Participants randomized to the intervention group improved their diagnostic accuracy from 57.8% to 75.9% with the use of dermoscopy. This group also experiencedimproved accuracy in its clinical diagnosis of melanoma from 54.6% to 62.7%.22
Argenziano and colleagues demonstrated similar results after PCPs attended a 4-hour workshop on dermoscopy. The 73 PCPs in this study evaluated 2,522 lesions randomized to naked eye examination or dermoscopy. The BMR, calculated from the data provided, improved from 12.6:1 to 10.5:1, respectively, when dermoscopy was incorporated into lesion analysis, while the sensitivity increased from 54.1% to 79.2% and the negative predictive value increased from 95.8% to 98.1%. It is important to note that the BMR and negative predictive value improved in tandem, indicating that PCPs were more discriminatory with their referrals for evaluation by dermatology while capturing a greater percentage of melanomas.23
These studies are not without limitations that preclude broad generalizations. For example, Dolianitis and colleagues and Westerhoff and colleagues provided participants with dermoscopic images of the lesions to be evaluated instead of requiring personal use of a dermatoscope, whereas the study by Argenziano and colleagues incorporated only 6 histopathologically proven malignant melanomas into each of the naked eye examination and the experimental dermoscopy groups.21-23 Yet these studies suggest that broader use of dermoscopy among PCPs could improve the accuracy of melanoma detection given clinically relevant training.
Several additional studies identify positive correlations associated with dermoscopy use among PCPs. A recent survey of 425 French general practitioners found that 8% of the study participants acknowledged owning a dermatoscope. Dermatoscope owners spent a statistically significant longer time analyzing each pigmented skin lesions, exhibited greater confidence in their analysis of pigmented lesions, and issued fewer overall referrals to dermatologists.24
Similarly, Rosendahl and colleagues evaluated the number needed to treat (NNT) (equivalent to the BMR) among 193 Australian PCPs and found that the NNT was inversely correlated to the frequency with which the physicians used dermoscopy. However, it was difficult to determine the definitive cause of the reduced NNT in this study because a similar effect was observed when NNT was evaluated based on general practitioner subspecialization.25 Again, despite limitations, these studies suggest that increased dermoscopy use among PCPs could reduce the morbidity of lifelong scarring as well as the short-term anxiety associated with a possible melanoma diagnosis.
Limitations
Even in the hands of a trained dermatologist, dermoscopy has limitations. Featureless melanoma is a term applied to melanoma lesions bereft of classical findings on both naked eye examination and dermoscopy. Menzies, a dermatologic pioneer in dermoscopy, acknowledged this limitation in 1996 while showing that 8% of melanomas evaded dermoscopic detection. He proceeded to discuss the importance of clinical history in melanoma detection because all of the featureless melanomas exhibited recent changes in size, shape, and/or color.26 More recently, sequential dermoscopy (mole mapping) imaging has been implemented to successfully identify these lesions.27 Thus, dermoscopy cannot replace dermatologists trained in the art of visual assessment with honed clinical diagnostic acumen. Rather, dermoscopy is a tool to enhance the assessment of clinically suspicious lesions and aid diagnostic discrimination of uncertain pigmented lesions.
Conclusion
Primary care physicians are on the frontline of medicine and often the first to have the opportunity to detect the presence of melanoma. Notably, 52.2% of the 884.7 million medical office visits performed annually in the US are with PCPs.28 Despite the benefits, dermoscopy is not uniformly used by dermatologists in the US. Of dermatologists practicing for more than 20 years, 76.2% use dermoscopy compared with 97.8% of dermatologists in practice for less than 5 years. This illustrates an increased use in tandem with dermatology residencies integrating dermoscopy training as a component of the curriculum, showing the importance of incorporating dermoscopy into medical school and residency training for PCPs..29-31 Guidelines regarding dermoscopy training and dermoscopic evaluation algorithms should be established, routinely taught in medical education, and actively incorporated into training curriculum for PCPs in order to improve patient care and realize the potential health care savings associated with the early diagnosis and treatment of melanoma. Dermoscopic-teledermatology consultations present a viable opportunity within the VHA to expedite access to care for veterans and simultaneously offer evaluative feedback on lesions to referring PCPs, as skilled, dermoscopy-trained dermatologists render the diagnoses. Given the devastating mortality rate of melanoma, continued multidisciplinary education on identifying melanoma is of the utmost importance for patient care. Widespread implementation of dermoscopy and dermoscopic-teledermatology consultations could save lives and slow the ever-increasing economic burden associated with melanoma treatment, costing $1.467 billion in 2016.32
1. Guy GP Jr, Thomas CC, Thompson T, Watson M, Massetti GM, Richardson LC. Vital signs: melanoma incidence and mortality trends and projections-United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64(21):591-596.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7-30.
3. American Cancer Society. Cancer facts & figures 2017. Atlanta: American Cancer Society; 2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed April 19, 2018.
4. Lea CS, Efird JT, Toland AE, Lewis DR, Phillips CJ. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179(3):247-253.
5. Thomas L, Puig S. Dermoscopy, digital dermoscopy and other diagnostic tools in the early detection of melanoma and follow-up of high-risk skin cancer patients. Acta Derm Venereol. 2017;97(218):14-21.
6. Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88(7):441-450.
7. Bafounta ML, Beauchet A, Aegerter P, Saiag P. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma? Results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137(10):1343-1350.
8. Terushkin V, Warycha M, Levy M, Kopf AW, Cohen DE, Polsky D. Analysis of the benign to malignant ratio of lesions biopsied by a general dermatologist before and after the adoption of dermoscopy. Arch Dermatol. 2010;146(3):343-344.
9. Wolner ZJ, Yélamos O, Liopyris K, Rogers T, Marchetti MA, Marghoob AA. Enhancing skin cancer diagnosis with dermoscopy. Dermatol Clin. 2017;35(4):417-437.
10. Carli P, Quercioli E, Sestini S, et al. Pattern analysis, not simplified algorithms, is the most reliable method for teaching dermoscopy for melanoma diagnosis to residents in dermatology. Br J Dermatol. 2003;148(5):981-984.
11. Argenziano G, Fabbrocini G, Carli P, De Giorgi V, Sammarco E, Delfino M. Epiluminescence microscopy for the diagnosis of doubtful melanocytic skin lesions. Comparison of the ABCD rule of dermatoscopy and a new 7-point checklist based on pattern analysis. Arch Dermatol. 1998;134(12):1563-1570.
12. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
13. Nachbar F, Stolz W, Merkle T, et al. The ABCD rule of dermatoscopy. High prospective value in the diagnosis of doubtful melanocytic skin lesions. J Am Acad Dermatol. 1994;30(4):551-559.
14. Henning JS, Dusza SW, Wang SQ, et al. The CASH (color, architecture, symmetry, and homogeneity) algorithm for dermoscopy. J Am Acad Dermatol. 2007;56(1):45-52.
15. Rogers T, Marino M, Dusza SW, Bajaj S, Marchetti MA, Marghoob A. Triage amalgamated dermoscopic algorithm (TADA) for skin cancer screening. Dermatol Pract Concept. 2017;7(2):39-46.
16. Ackerman AB. No one should die of malignant melanoma. J Am Acad Dermatol. 1985;12(1):115-116.
17. Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II: sun exposure. Eur J Cancer. 2005;41(1):45-60.
18. Gerbert B, Maurer T, Berger T, et al. Primary care physicians as gatekeepers in managed care. Primary care physicians’ and dermatologists’ skills at secondary prevention of skin cancer. Arch Dermatol. 1996;132(9):1030-1038.
19. Corbo MD, Wismer J. Agreement between dermatologists and primary care practitioners in the diagnosis of malignant melanoma: review of the literature. J Cutan Med Surg. 2012;16(5):306-310.
20. Morris JB, Alfonso SV, Hernandez N, Fernández MI. Examining the factors associated with past and present dermoscopy use among family physicians. Dermatol Pract Concept. 2017;7(4):63-70.
21. Dolianitis C, Kelly J, Wolfe R, Simpson P. Comparative performance of 4 dermoscopic algorithms by nonexperts for the diagnosis of melanocytic lesions. Arch Dermatol. 2005;141(8):1008-1014.
22. Westerhoff K, Mccarthy WH, Menzies SW. Increase in the sensitivity for melanoma diagnosis by primary care physicians using skin surface microscopy. Br J Dermatol. 2000;143(5):1016-1020.
23. Argenziano G, Puig S, Zalaudek I, et al. Dermoscopy improves accuracy of primary care physicians to triage lesions suggestive of skin cancer. J Clin Oncol. 2006;24(12):1877-1882.
24. Chappuis P, Duru G, Marchal O, Girier P, Dalle S, Thomas L. Dermoscopy, a useful tool for general practitioners in melanoma screening: a nationwide survey. Br J Dermatol. 2016;175(4):744-750.
25. Rosendahl C, Williams G, Eley D, et al. The impact of subspecialization and dermatoscopy use on accuracy of melanoma diagnosis among primary care doctors in Australia. J Am Acad Dermatol. 2012;67(5):846-852.
26. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
27. Kittler H, Guitera P, Riedl E, et al. Identification of clinically featureless incipient melanoma using sequential dermoscopy imaging. Arch Dermatol. 2006;142(9):1113-1119.
28. Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. https://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated May 3, 2017. Accessed April 10, 2018.
29. Murzaku EC, Hayan S, Rao BK. Methods and rates of dermoscopy usage: a cross-sectional survey of US dermatologists stratified by years in practice. J Am Acad Dermatol. 2014;71(2):393-395.
30. Nehal KS, Oliveria SA, Marghoob AA, et al. Use of and beliefs about dermoscopy in the management of patients with pigmented lesions: a survey of dermatology residency programmes in the United States. Melanoma Res. 2002;12(6):601-605.
31. Wu TP, Newlove T, Smith L, Vuong CH, Stein JA, Polsky D. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68(6):1000-1005.
32. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76(5):958-972
1. Guy GP Jr, Thomas CC, Thompson T, Watson M, Massetti GM, Richardson LC. Vital signs: melanoma incidence and mortality trends and projections-United States, 1982-2030. MMWR Morb Mortal Wkly Rep. 2015;64(21):591-596.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7-30.
3. American Cancer Society. Cancer facts & figures 2017. Atlanta: American Cancer Society; 2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf. Accessed April 19, 2018.
4. Lea CS, Efird JT, Toland AE, Lewis DR, Phillips CJ. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179(3):247-253.
5. Thomas L, Puig S. Dermoscopy, digital dermoscopy and other diagnostic tools in the early detection of melanoma and follow-up of high-risk skin cancer patients. Acta Derm Venereol. 2017;97(218):14-21.
6. Marghoob AA, Usatine RP, Jaimes N. Dermoscopy for the family physician. Am Fam Physician. 2013;88(7):441-450.
7. Bafounta ML, Beauchet A, Aegerter P, Saiag P. Is dermoscopy (epiluminescence microscopy) useful for the diagnosis of melanoma? Results of a meta-analysis using techniques adapted to the evaluation of diagnostic tests. Arch Dermatol. 2001;137(10):1343-1350.
8. Terushkin V, Warycha M, Levy M, Kopf AW, Cohen DE, Polsky D. Analysis of the benign to malignant ratio of lesions biopsied by a general dermatologist before and after the adoption of dermoscopy. Arch Dermatol. 2010;146(3):343-344.
9. Wolner ZJ, Yélamos O, Liopyris K, Rogers T, Marchetti MA, Marghoob AA. Enhancing skin cancer diagnosis with dermoscopy. Dermatol Clin. 2017;35(4):417-437.
10. Carli P, Quercioli E, Sestini S, et al. Pattern analysis, not simplified algorithms, is the most reliable method for teaching dermoscopy for melanoma diagnosis to residents in dermatology. Br J Dermatol. 2003;148(5):981-984.
11. Argenziano G, Fabbrocini G, Carli P, De Giorgi V, Sammarco E, Delfino M. Epiluminescence microscopy for the diagnosis of doubtful melanocytic skin lesions. Comparison of the ABCD rule of dermatoscopy and a new 7-point checklist based on pattern analysis. Arch Dermatol. 1998;134(12):1563-1570.
12. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
13. Nachbar F, Stolz W, Merkle T, et al. The ABCD rule of dermatoscopy. High prospective value in the diagnosis of doubtful melanocytic skin lesions. J Am Acad Dermatol. 1994;30(4):551-559.
14. Henning JS, Dusza SW, Wang SQ, et al. The CASH (color, architecture, symmetry, and homogeneity) algorithm for dermoscopy. J Am Acad Dermatol. 2007;56(1):45-52.
15. Rogers T, Marino M, Dusza SW, Bajaj S, Marchetti MA, Marghoob A. Triage amalgamated dermoscopic algorithm (TADA) for skin cancer screening. Dermatol Pract Concept. 2017;7(2):39-46.
16. Ackerman AB. No one should die of malignant melanoma. J Am Acad Dermatol. 1985;12(1):115-116.
17. Gandini S, Sera F, Cattaruzza MS, et al. Meta-analysis of risk factors for cutaneous melanoma: II: sun exposure. Eur J Cancer. 2005;41(1):45-60.
18. Gerbert B, Maurer T, Berger T, et al. Primary care physicians as gatekeepers in managed care. Primary care physicians’ and dermatologists’ skills at secondary prevention of skin cancer. Arch Dermatol. 1996;132(9):1030-1038.
19. Corbo MD, Wismer J. Agreement between dermatologists and primary care practitioners in the diagnosis of malignant melanoma: review of the literature. J Cutan Med Surg. 2012;16(5):306-310.
20. Morris JB, Alfonso SV, Hernandez N, Fernández MI. Examining the factors associated with past and present dermoscopy use among family physicians. Dermatol Pract Concept. 2017;7(4):63-70.
21. Dolianitis C, Kelly J, Wolfe R, Simpson P. Comparative performance of 4 dermoscopic algorithms by nonexperts for the diagnosis of melanocytic lesions. Arch Dermatol. 2005;141(8):1008-1014.
22. Westerhoff K, Mccarthy WH, Menzies SW. Increase in the sensitivity for melanoma diagnosis by primary care physicians using skin surface microscopy. Br J Dermatol. 2000;143(5):1016-1020.
23. Argenziano G, Puig S, Zalaudek I, et al. Dermoscopy improves accuracy of primary care physicians to triage lesions suggestive of skin cancer. J Clin Oncol. 2006;24(12):1877-1882.
24. Chappuis P, Duru G, Marchal O, Girier P, Dalle S, Thomas L. Dermoscopy, a useful tool for general practitioners in melanoma screening: a nationwide survey. Br J Dermatol. 2016;175(4):744-750.
25. Rosendahl C, Williams G, Eley D, et al. The impact of subspecialization and dermatoscopy use on accuracy of melanoma diagnosis among primary care doctors in Australia. J Am Acad Dermatol. 2012;67(5):846-852.
26. Menzies SW, Ingvar C, Crotty KA, McCarthy WH. Frequency and morphologic characteristics of invasive melanomas lacking specific surface microscopic features. Arch Dermatol. 1996;132(10):1178-1182.
27. Kittler H, Guitera P, Riedl E, et al. Identification of clinically featureless incipient melanoma using sequential dermoscopy imaging. Arch Dermatol. 2006;142(9):1113-1119.
28. Centers for Disease Control and Prevention. Ambulatory care use and physician office visits. https://www.cdc.gov/nchs/fastats/physician-visits.htm. Updated May 3, 2017. Accessed April 10, 2018.
29. Murzaku EC, Hayan S, Rao BK. Methods and rates of dermoscopy usage: a cross-sectional survey of US dermatologists stratified by years in practice. J Am Acad Dermatol. 2014;71(2):393-395.
30. Nehal KS, Oliveria SA, Marghoob AA, et al. Use of and beliefs about dermoscopy in the management of patients with pigmented lesions: a survey of dermatology residency programmes in the United States. Melanoma Res. 2002;12(6):601-605.
31. Wu TP, Newlove T, Smith L, Vuong CH, Stein JA, Polsky D. The importance of dedicated dermoscopy training during residency: a survey of US dermatology chief residents. J Am Acad Dermatol. 2013;68(6):1000-1005.
32. Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States. J Am Acad Dermatol. 2017;76(5):958-972
Pediatric cancers are on the rise
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
REPORTING FROM ASPHO 2018
Key clinical point: Major finding: From 2001 to 2014, there were 196,200 incident cases of pediatric cancer for an overall cancer incidence rate of 173 per 1 million.
Study details: A review of data from the United States Cancer Statistics for children under age 20.
Disclosures: The CDC supported the study. Dr. Siegel and his coauthors are CDC employees. They reported having no conflicts of interest.
Source: Siegel DA et al. ASPHO 2018, Abstract 605.
Is cancer immunotherapy more effective in men than women?
Cancer immunotherapy using checkpoint inhibitors may achieve greater mortality reductions in men than they do in women, new research has suggested.
In a meta-analysis and systematic review published in Lancet Oncology, researchers analyzed 20 randomized, controlled trials of immune checkpoint inhibitors that included detail on overall survival and patients’ sex; altogether, these studies involved 11,351 patients with advanced or metastatic cancers.
They found that while men treated with checkpoint inhibitors had a significant 28% reduced risk of death, compared with male controls, the survival benefit in women was smaller (14% reduced risk of death, compared with female controls).
Fabio Conforti, MD, from the European Institute of Oncology, Milan, and coauthors commented that the magnitude of the difference between the effect seen men and that in women was clinically significant.
“The pooled reduction of risk of death was double the size for male patients than for female patients – a difference that is similar to the size of the difference in survival benefit observed between patients with non–small cell lung cancer with PD-L1 positive (greater than 1%) tumors versus negative tumors, who were treated with anti-PD-1,” they wrote.
This difference between the benefit seen men and that in women was evident across all the subgroups in the study, which included subgroups based on cancer histotype, line of treatment, drugs used, and type of control.
However there was greater heterogeneity in the magnitude of the effect of checkpoint inhibitors on mortality in men than there was in women. The authors suggested this could be explained by the fact that the drugs have lower efficacy in women and this may therefore reduce the variability of results when compared with those in men.
The authors also looked at whether the studies that compared immunotherapies with nonimmunological therapies might show a different effect, but they still found a significantly higher benefit in men, compared with women.
The overall study population was two-thirds male and one-third female. The checkpoint inhibitors used were ipilimumab, tremelimumab, nivolumab, and pembrolizumab, and the trials were conducted in patients with melanoma, non–small cell lung cancer, head and neck cancer, renal cell carcinoma, urothelial tumors, gastric tumors, and mesothelioma.
Men have almost double the risk of mortality from cancer than do women, the authors said, with the greatest differences seen in melanoma, lung cancer, larynx cancer, esophagus cancer, and bladder cancer.
“This male-biased mortality is hypothesized to reflect differences not only in behavioral and biological factors, including causes of cancer and hormonal regulation, but also in the immune system.”
Despite this, sex is rarely taken into account when new therapeutic approaches are tested, the authors said.
They also commented on the fact that there was a relatively low number of women included in each trial, an issue that was recognized as far back as the 1990s as a major problem in medical trials.
“Our results further highlight this problem, showing clinically relevant differences in the efficacy of two important classes of immunological drugs, namely anti–CTLA-4 and anti–PD-1 antibodies, when compared with controls in male and female patients with advanced solid tumors,” they wrote.
They noted that they couldn’t exclude the possibility that the effect may be the result of other variables that were distributed differently between the sexes. However, they also qualified this by saying that variables known to affect the efficacy of immune checkpoint inhibitors, such as PD-L1 expression and mutation status, were not likely to explain the results.
Given their findings, the authors said a patient’s sex should be taken into account when weighing the risks and benefits of checkpoint inhibitors given the magnitude of benefit was sex-dependent. They also called for future immunotherapy studies to include more women.
No funding or conflicts of interest were declared.
SOURCE: Conforti F et al. Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045(18)30261-4.
While cancer immunotherapy represents one of the most significant clinical advances in cancer treatment in the past decade, the basic but important clinical question about different effects between men and woman has not been addressed until now. The authors of this study are to be congratulated on such a comprehensive and well-conducted analysis, but the data does not completely support their final conclusion that checkpoint inhibitors benefit men more than women.
There are a large number of baseline characteristics of solid tumors that might differ between men and women and that have also been reported to impact the outcomes of patients treated with checkpoint inhibitors. Some of these may be lifestyle or behavioral characteristics – such as different smoking habits between men and women with non–small cell lung cancer – or differences in the distribution of oncogenic driver mutations between men and women.
We should therefore be cautious in jumping to conclusions and changing the current standard of care with respect to checkpoint inhibitors. In particular, we should not be denying treatment to women who are otherwise indicated for checkpoint inhibitors, based on these findings.
Omar Abdel-Rahman, MD, is from the clinical oncology department of the faculty of medicine at Ain Shams University in Cairo and from the Tom Baker Cancer Centre in Calgary. These comments are taken from an accompanying editorial (Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045[18]30270-5.) No conflicts of interest were declared.
While cancer immunotherapy represents one of the most significant clinical advances in cancer treatment in the past decade, the basic but important clinical question about different effects between men and woman has not been addressed until now. The authors of this study are to be congratulated on such a comprehensive and well-conducted analysis, but the data does not completely support their final conclusion that checkpoint inhibitors benefit men more than women.
There are a large number of baseline characteristics of solid tumors that might differ between men and women and that have also been reported to impact the outcomes of patients treated with checkpoint inhibitors. Some of these may be lifestyle or behavioral characteristics – such as different smoking habits between men and women with non–small cell lung cancer – or differences in the distribution of oncogenic driver mutations between men and women.
We should therefore be cautious in jumping to conclusions and changing the current standard of care with respect to checkpoint inhibitors. In particular, we should not be denying treatment to women who are otherwise indicated for checkpoint inhibitors, based on these findings.
Omar Abdel-Rahman, MD, is from the clinical oncology department of the faculty of medicine at Ain Shams University in Cairo and from the Tom Baker Cancer Centre in Calgary. These comments are taken from an accompanying editorial (Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045[18]30270-5.) No conflicts of interest were declared.
While cancer immunotherapy represents one of the most significant clinical advances in cancer treatment in the past decade, the basic but important clinical question about different effects between men and woman has not been addressed until now. The authors of this study are to be congratulated on such a comprehensive and well-conducted analysis, but the data does not completely support their final conclusion that checkpoint inhibitors benefit men more than women.
There are a large number of baseline characteristics of solid tumors that might differ between men and women and that have also been reported to impact the outcomes of patients treated with checkpoint inhibitors. Some of these may be lifestyle or behavioral characteristics – such as different smoking habits between men and women with non–small cell lung cancer – or differences in the distribution of oncogenic driver mutations between men and women.
We should therefore be cautious in jumping to conclusions and changing the current standard of care with respect to checkpoint inhibitors. In particular, we should not be denying treatment to women who are otherwise indicated for checkpoint inhibitors, based on these findings.
Omar Abdel-Rahman, MD, is from the clinical oncology department of the faculty of medicine at Ain Shams University in Cairo and from the Tom Baker Cancer Centre in Calgary. These comments are taken from an accompanying editorial (Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045[18]30270-5.) No conflicts of interest were declared.
Cancer immunotherapy using checkpoint inhibitors may achieve greater mortality reductions in men than they do in women, new research has suggested.
In a meta-analysis and systematic review published in Lancet Oncology, researchers analyzed 20 randomized, controlled trials of immune checkpoint inhibitors that included detail on overall survival and patients’ sex; altogether, these studies involved 11,351 patients with advanced or metastatic cancers.
They found that while men treated with checkpoint inhibitors had a significant 28% reduced risk of death, compared with male controls, the survival benefit in women was smaller (14% reduced risk of death, compared with female controls).
Fabio Conforti, MD, from the European Institute of Oncology, Milan, and coauthors commented that the magnitude of the difference between the effect seen men and that in women was clinically significant.
“The pooled reduction of risk of death was double the size for male patients than for female patients – a difference that is similar to the size of the difference in survival benefit observed between patients with non–small cell lung cancer with PD-L1 positive (greater than 1%) tumors versus negative tumors, who were treated with anti-PD-1,” they wrote.
This difference between the benefit seen men and that in women was evident across all the subgroups in the study, which included subgroups based on cancer histotype, line of treatment, drugs used, and type of control.
However there was greater heterogeneity in the magnitude of the effect of checkpoint inhibitors on mortality in men than there was in women. The authors suggested this could be explained by the fact that the drugs have lower efficacy in women and this may therefore reduce the variability of results when compared with those in men.
The authors also looked at whether the studies that compared immunotherapies with nonimmunological therapies might show a different effect, but they still found a significantly higher benefit in men, compared with women.
The overall study population was two-thirds male and one-third female. The checkpoint inhibitors used were ipilimumab, tremelimumab, nivolumab, and pembrolizumab, and the trials were conducted in patients with melanoma, non–small cell lung cancer, head and neck cancer, renal cell carcinoma, urothelial tumors, gastric tumors, and mesothelioma.
Men have almost double the risk of mortality from cancer than do women, the authors said, with the greatest differences seen in melanoma, lung cancer, larynx cancer, esophagus cancer, and bladder cancer.
“This male-biased mortality is hypothesized to reflect differences not only in behavioral and biological factors, including causes of cancer and hormonal regulation, but also in the immune system.”
Despite this, sex is rarely taken into account when new therapeutic approaches are tested, the authors said.
They also commented on the fact that there was a relatively low number of women included in each trial, an issue that was recognized as far back as the 1990s as a major problem in medical trials.
“Our results further highlight this problem, showing clinically relevant differences in the efficacy of two important classes of immunological drugs, namely anti–CTLA-4 and anti–PD-1 antibodies, when compared with controls in male and female patients with advanced solid tumors,” they wrote.
They noted that they couldn’t exclude the possibility that the effect may be the result of other variables that were distributed differently between the sexes. However, they also qualified this by saying that variables known to affect the efficacy of immune checkpoint inhibitors, such as PD-L1 expression and mutation status, were not likely to explain the results.
Given their findings, the authors said a patient’s sex should be taken into account when weighing the risks and benefits of checkpoint inhibitors given the magnitude of benefit was sex-dependent. They also called for future immunotherapy studies to include more women.
No funding or conflicts of interest were declared.
SOURCE: Conforti F et al. Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045(18)30261-4.
Cancer immunotherapy using checkpoint inhibitors may achieve greater mortality reductions in men than they do in women, new research has suggested.
In a meta-analysis and systematic review published in Lancet Oncology, researchers analyzed 20 randomized, controlled trials of immune checkpoint inhibitors that included detail on overall survival and patients’ sex; altogether, these studies involved 11,351 patients with advanced or metastatic cancers.
They found that while men treated with checkpoint inhibitors had a significant 28% reduced risk of death, compared with male controls, the survival benefit in women was smaller (14% reduced risk of death, compared with female controls).
Fabio Conforti, MD, from the European Institute of Oncology, Milan, and coauthors commented that the magnitude of the difference between the effect seen men and that in women was clinically significant.
“The pooled reduction of risk of death was double the size for male patients than for female patients – a difference that is similar to the size of the difference in survival benefit observed between patients with non–small cell lung cancer with PD-L1 positive (greater than 1%) tumors versus negative tumors, who were treated with anti-PD-1,” they wrote.
This difference between the benefit seen men and that in women was evident across all the subgroups in the study, which included subgroups based on cancer histotype, line of treatment, drugs used, and type of control.
However there was greater heterogeneity in the magnitude of the effect of checkpoint inhibitors on mortality in men than there was in women. The authors suggested this could be explained by the fact that the drugs have lower efficacy in women and this may therefore reduce the variability of results when compared with those in men.
The authors also looked at whether the studies that compared immunotherapies with nonimmunological therapies might show a different effect, but they still found a significantly higher benefit in men, compared with women.
The overall study population was two-thirds male and one-third female. The checkpoint inhibitors used were ipilimumab, tremelimumab, nivolumab, and pembrolizumab, and the trials were conducted in patients with melanoma, non–small cell lung cancer, head and neck cancer, renal cell carcinoma, urothelial tumors, gastric tumors, and mesothelioma.
Men have almost double the risk of mortality from cancer than do women, the authors said, with the greatest differences seen in melanoma, lung cancer, larynx cancer, esophagus cancer, and bladder cancer.
“This male-biased mortality is hypothesized to reflect differences not only in behavioral and biological factors, including causes of cancer and hormonal regulation, but also in the immune system.”
Despite this, sex is rarely taken into account when new therapeutic approaches are tested, the authors said.
They also commented on the fact that there was a relatively low number of women included in each trial, an issue that was recognized as far back as the 1990s as a major problem in medical trials.
“Our results further highlight this problem, showing clinically relevant differences in the efficacy of two important classes of immunological drugs, namely anti–CTLA-4 and anti–PD-1 antibodies, when compared with controls in male and female patients with advanced solid tumors,” they wrote.
They noted that they couldn’t exclude the possibility that the effect may be the result of other variables that were distributed differently between the sexes. However, they also qualified this by saying that variables known to affect the efficacy of immune checkpoint inhibitors, such as PD-L1 expression and mutation status, were not likely to explain the results.
Given their findings, the authors said a patient’s sex should be taken into account when weighing the risks and benefits of checkpoint inhibitors given the magnitude of benefit was sex-dependent. They also called for future immunotherapy studies to include more women.
No funding or conflicts of interest were declared.
SOURCE: Conforti F et al. Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045(18)30261-4.
FROM LANCET ONCOLOGY
Key clinical point: Checkpoint inhibitors are linked with greater mortality reductions in men than in women.
Major finding: Checkpoint inhibitors are associated with a 28% reduction in cancer mortality in men and 14% in women.
Study details: Systematic review and meta-analysis of 20 randomized, controlled trials involving 11,351 patients.
Disclosures: No funding or conflicts of interest were declared.
Source: Conforti F et al. Lancet Oncol. 2018 May 16. doi: 10.1016/S1470-2045(18)30261-4.
Mask provides effective, cheap protection from hazardous electrocautery plumes
CHICAGO – Routine use of an N95 mask during electrocautery is an effective and inexpensive way for dermatologic surgeons to protect themselves from toxic, airborne particulate matter in the smoke generated during the procedure, Emily de Golian, MD, said at the annual meeting of the American College of Mohs Surgery.
“Our data suggest clear as well as superiority to the laser masks that are used in hair removal procedures and ablative procedures in cosmetic clinics,” commented Dr. de Golian, a Mohs micrographic surgery fellow at the University of California, San Diego.
This matter of self-protection from the effects of electrocautery smoke plumes deserves greater attention from the dermatologic community, according to Dr. de Golian. There is solid evidence that these plumes contain high concentrations of known carcinogens, including benzene, acetonitrile, and butadiene – indeed, concentrations far in excess of what’s found in second-hand cigarette smoke. Moreover, many of these airborne carcinogens and other toxins have been linked to leukemia, neurologic disorders, lung cancer, thrombotic disorders, lung disease, and infectious disease transmission, albeit not convincingly so to date in dermatologic surgeons. But why wait for definitive evidence to accrue?
“In light of these hazards – and according to governmental guidelines – dermatologic surgeons would be wise to adopt protective measures during surgical procedures,” Dr. de Golian said.
But they haven’t. She cited a national survey conducted several years ago by a colleague in which 79% of the 316 responding dermatologic surgeons indicated they use no smoke management whatsoever, neither masks nor a local exhaust evacuation system. Only 10% employed smoke management 25%-50% of the time during electrocautery, and a scant 11% of dermatologic surgeons did so at least 75% of the time (Dermatol Surg. 2014 Dec;40[12]:1373-7).
Given the far more substantial expense of installing an office smoke evacuation system, mask filtration becomes an attractive alternative. But the relative efficacy of the various types of masks in blocking fine and ultrafine particulate matter contained in electrocautery plumes hadn’t previously been systematically studied. This created the impetus for Dr. de Golian’s study.
The N95 masks were the clear winner, particularly when it came to filtering the ultrafine particles, which are of greatest concern because they remain suspended in air longer and penetrate deeper into the respiratory tract than larger particles. The N95 masks proved superior to procedural masks, which in turn were significantly more effective than the laser masks. The differences between mask performance for larger particle filtration were smaller, although the N95 remained number one. She noted that the study results probably underestimate the true filtration efficacy of N95 masks, since they form a tighter seal with the face in clinical practice than with the other two mask types.
Mask self-protection “is easily applicable in your own practice, and it meets NIOSH-recommended [National Institute for Occupational Safety and Health] standards for safety in the workplace,” the dermatologist noted.
In the next phase of her research, she plans to evaluate the optimal technology and techniques of smoke evacuation in the surgical suite. That’s an attractive method because it protects everyone in the room, not just the surgeon. And while the practitioner survey indicates this technology isn’t widely used by dermatologic surgeons on a routine basis at present, that could change, particularly in the current era in which patient-reported outcomes and satisfaction surveys have taken on added weight.
“Patients prefer not smelling their own tissue burning,” Dr. de Golian said.
She reported no financial conflicts regarding her study, which was conducted free of commercial support.
CHICAGO – Routine use of an N95 mask during electrocautery is an effective and inexpensive way for dermatologic surgeons to protect themselves from toxic, airborne particulate matter in the smoke generated during the procedure, Emily de Golian, MD, said at the annual meeting of the American College of Mohs Surgery.
“Our data suggest clear as well as superiority to the laser masks that are used in hair removal procedures and ablative procedures in cosmetic clinics,” commented Dr. de Golian, a Mohs micrographic surgery fellow at the University of California, San Diego.
This matter of self-protection from the effects of electrocautery smoke plumes deserves greater attention from the dermatologic community, according to Dr. de Golian. There is solid evidence that these plumes contain high concentrations of known carcinogens, including benzene, acetonitrile, and butadiene – indeed, concentrations far in excess of what’s found in second-hand cigarette smoke. Moreover, many of these airborne carcinogens and other toxins have been linked to leukemia, neurologic disorders, lung cancer, thrombotic disorders, lung disease, and infectious disease transmission, albeit not convincingly so to date in dermatologic surgeons. But why wait for definitive evidence to accrue?
“In light of these hazards – and according to governmental guidelines – dermatologic surgeons would be wise to adopt protective measures during surgical procedures,” Dr. de Golian said.
But they haven’t. She cited a national survey conducted several years ago by a colleague in which 79% of the 316 responding dermatologic surgeons indicated they use no smoke management whatsoever, neither masks nor a local exhaust evacuation system. Only 10% employed smoke management 25%-50% of the time during electrocautery, and a scant 11% of dermatologic surgeons did so at least 75% of the time (Dermatol Surg. 2014 Dec;40[12]:1373-7).
Given the far more substantial expense of installing an office smoke evacuation system, mask filtration becomes an attractive alternative. But the relative efficacy of the various types of masks in blocking fine and ultrafine particulate matter contained in electrocautery plumes hadn’t previously been systematically studied. This created the impetus for Dr. de Golian’s study.
The N95 masks were the clear winner, particularly when it came to filtering the ultrafine particles, which are of greatest concern because they remain suspended in air longer and penetrate deeper into the respiratory tract than larger particles. The N95 masks proved superior to procedural masks, which in turn were significantly more effective than the laser masks. The differences between mask performance for larger particle filtration were smaller, although the N95 remained number one. She noted that the study results probably underestimate the true filtration efficacy of N95 masks, since they form a tighter seal with the face in clinical practice than with the other two mask types.
Mask self-protection “is easily applicable in your own practice, and it meets NIOSH-recommended [National Institute for Occupational Safety and Health] standards for safety in the workplace,” the dermatologist noted.
In the next phase of her research, she plans to evaluate the optimal technology and techniques of smoke evacuation in the surgical suite. That’s an attractive method because it protects everyone in the room, not just the surgeon. And while the practitioner survey indicates this technology isn’t widely used by dermatologic surgeons on a routine basis at present, that could change, particularly in the current era in which patient-reported outcomes and satisfaction surveys have taken on added weight.
“Patients prefer not smelling their own tissue burning,” Dr. de Golian said.
She reported no financial conflicts regarding her study, which was conducted free of commercial support.
CHICAGO – Routine use of an N95 mask during electrocautery is an effective and inexpensive way for dermatologic surgeons to protect themselves from toxic, airborne particulate matter in the smoke generated during the procedure, Emily de Golian, MD, said at the annual meeting of the American College of Mohs Surgery.
“Our data suggest clear as well as superiority to the laser masks that are used in hair removal procedures and ablative procedures in cosmetic clinics,” commented Dr. de Golian, a Mohs micrographic surgery fellow at the University of California, San Diego.
This matter of self-protection from the effects of electrocautery smoke plumes deserves greater attention from the dermatologic community, according to Dr. de Golian. There is solid evidence that these plumes contain high concentrations of known carcinogens, including benzene, acetonitrile, and butadiene – indeed, concentrations far in excess of what’s found in second-hand cigarette smoke. Moreover, many of these airborne carcinogens and other toxins have been linked to leukemia, neurologic disorders, lung cancer, thrombotic disorders, lung disease, and infectious disease transmission, albeit not convincingly so to date in dermatologic surgeons. But why wait for definitive evidence to accrue?
“In light of these hazards – and according to governmental guidelines – dermatologic surgeons would be wise to adopt protective measures during surgical procedures,” Dr. de Golian said.
But they haven’t. She cited a national survey conducted several years ago by a colleague in which 79% of the 316 responding dermatologic surgeons indicated they use no smoke management whatsoever, neither masks nor a local exhaust evacuation system. Only 10% employed smoke management 25%-50% of the time during electrocautery, and a scant 11% of dermatologic surgeons did so at least 75% of the time (Dermatol Surg. 2014 Dec;40[12]:1373-7).
Given the far more substantial expense of installing an office smoke evacuation system, mask filtration becomes an attractive alternative. But the relative efficacy of the various types of masks in blocking fine and ultrafine particulate matter contained in electrocautery plumes hadn’t previously been systematically studied. This created the impetus for Dr. de Golian’s study.
The N95 masks were the clear winner, particularly when it came to filtering the ultrafine particles, which are of greatest concern because they remain suspended in air longer and penetrate deeper into the respiratory tract than larger particles. The N95 masks proved superior to procedural masks, which in turn were significantly more effective than the laser masks. The differences between mask performance for larger particle filtration were smaller, although the N95 remained number one. She noted that the study results probably underestimate the true filtration efficacy of N95 masks, since they form a tighter seal with the face in clinical practice than with the other two mask types.
Mask self-protection “is easily applicable in your own practice, and it meets NIOSH-recommended [National Institute for Occupational Safety and Health] standards for safety in the workplace,” the dermatologist noted.
In the next phase of her research, she plans to evaluate the optimal technology and techniques of smoke evacuation in the surgical suite. That’s an attractive method because it protects everyone in the room, not just the surgeon. And while the practitioner survey indicates this technology isn’t widely used by dermatologic surgeons on a routine basis at present, that could change, particularly in the current era in which patient-reported outcomes and satisfaction surveys have taken on added weight.
“Patients prefer not smelling their own tissue burning,” Dr. de Golian said.
She reported no financial conflicts regarding her study, which was conducted free of commercial support.
REPORTING FROM THE ACMS ANNUAL MEETING
Key clinical point: Electrocautery smoke is bad news, and wearing an N95 mask affords protection.
Major finding: The N95 mask was significantly more effective than basic procedural or laser masks at filtering particulate matter less than 1 mcm in size contained in electrocautery smoke.
Study details: This study utilized highly sensitive airborne particle counting devices to assess the relative protective filtration afforded by three types of masks.
Disclosures: The presenter reported no financial conflicts regarding this study, which was conducted free of commercial support.
Checkpoint inhibitors get to patients quickly
Checkpoint inhibitors got to melanoma, non–small cell lung cancer, and renal cell carcinoma patients quickly in clinical practice after Food and Drug Administration approval – usually within 4 months – but the patients treated in clinical settings tended to be older than those treated in trials, which has caused concern about whether real-world efficacy will prove to be the same, according to a study in JAMA Oncology.
“Such rapid adoption stands in contrast to older estimates that suggest it takes years or even decades for new treatments to be adopted,” wrote lead author Cary Gross, MD, professor of medicine at Yale University, New Haven, and his coauthors. “We found significant differences in age between patients treated in practice and those treated in trials, which highlights the need to clarify the risks and benefits of checkpoint inhibitors in general populations of patients.”
Researchers drew data on nivolumab and pembrolizumab use from the Flatiron Health longitudinal EHR database, which included 233 academic and community oncology practices. In each of the three disease cohorts, adoption was seen within 4 months for at least 60% of patients. Uptake was quickest for the melanoma patients, 76% of whom received a checkpoint inhibitor within 4 months, investigators wrote. Factors for the fast adoption could include high disease severity, a preference for novelty, perceived gains over existing treatments, and promotional activities, such as media reports and advertising directly to consumers, they wrote.
More patients in real-world practice were aged 65 years or older, ranging from as little as 61% at the lowest end of the range at one center to as much as 64% at the highest end at another. In the clinical trials, the percentage of patients aged 65 years or older ranged from 32% in one trial to 41% in another. Researchers wrote that these higher real-world rates are concerning because there are still questions regarding whether differences in immune responses will cause differences in efficacy between older and younger patients, as well as safety considerations among different age groups.
“Although data suggest that outcomes are similar between older and younger patients receiving anti–PD-1 agents for melanoma, there is little evidence to guide anti–PD-1 treatment of older patients with NSCLC [non–small cell lung cancer],” Dr. Gross and his coinvestigators wrote.
Investigators wrote that the findings are cause for caution.
“As FDA officials develop more flexible standards for approval, which the 21st Century Cures Act requires them to do, it is possible that many patients will receive drugs before much is known about clinical outcomes,” Dr. Gross said. “Further integrations of real-world evidence might allow the FDA to better assess the drugs that they approve on the basis of nonrepresentative trial participants.”
SOURCE: Gross C et al. JAMA Oncol. 2018 May 10. doi: 10.1001/jamaoncol.2018.0798.
Checkpoint inhibitors got to melanoma, non–small cell lung cancer, and renal cell carcinoma patients quickly in clinical practice after Food and Drug Administration approval – usually within 4 months – but the patients treated in clinical settings tended to be older than those treated in trials, which has caused concern about whether real-world efficacy will prove to be the same, according to a study in JAMA Oncology.
“Such rapid adoption stands in contrast to older estimates that suggest it takes years or even decades for new treatments to be adopted,” wrote lead author Cary Gross, MD, professor of medicine at Yale University, New Haven, and his coauthors. “We found significant differences in age between patients treated in practice and those treated in trials, which highlights the need to clarify the risks and benefits of checkpoint inhibitors in general populations of patients.”
Researchers drew data on nivolumab and pembrolizumab use from the Flatiron Health longitudinal EHR database, which included 233 academic and community oncology practices. In each of the three disease cohorts, adoption was seen within 4 months for at least 60% of patients. Uptake was quickest for the melanoma patients, 76% of whom received a checkpoint inhibitor within 4 months, investigators wrote. Factors for the fast adoption could include high disease severity, a preference for novelty, perceived gains over existing treatments, and promotional activities, such as media reports and advertising directly to consumers, they wrote.
More patients in real-world practice were aged 65 years or older, ranging from as little as 61% at the lowest end of the range at one center to as much as 64% at the highest end at another. In the clinical trials, the percentage of patients aged 65 years or older ranged from 32% in one trial to 41% in another. Researchers wrote that these higher real-world rates are concerning because there are still questions regarding whether differences in immune responses will cause differences in efficacy between older and younger patients, as well as safety considerations among different age groups.
“Although data suggest that outcomes are similar between older and younger patients receiving anti–PD-1 agents for melanoma, there is little evidence to guide anti–PD-1 treatment of older patients with NSCLC [non–small cell lung cancer],” Dr. Gross and his coinvestigators wrote.
Investigators wrote that the findings are cause for caution.
“As FDA officials develop more flexible standards for approval, which the 21st Century Cures Act requires them to do, it is possible that many patients will receive drugs before much is known about clinical outcomes,” Dr. Gross said. “Further integrations of real-world evidence might allow the FDA to better assess the drugs that they approve on the basis of nonrepresentative trial participants.”
SOURCE: Gross C et al. JAMA Oncol. 2018 May 10. doi: 10.1001/jamaoncol.2018.0798.
Checkpoint inhibitors got to melanoma, non–small cell lung cancer, and renal cell carcinoma patients quickly in clinical practice after Food and Drug Administration approval – usually within 4 months – but the patients treated in clinical settings tended to be older than those treated in trials, which has caused concern about whether real-world efficacy will prove to be the same, according to a study in JAMA Oncology.
“Such rapid adoption stands in contrast to older estimates that suggest it takes years or even decades for new treatments to be adopted,” wrote lead author Cary Gross, MD, professor of medicine at Yale University, New Haven, and his coauthors. “We found significant differences in age between patients treated in practice and those treated in trials, which highlights the need to clarify the risks and benefits of checkpoint inhibitors in general populations of patients.”
Researchers drew data on nivolumab and pembrolizumab use from the Flatiron Health longitudinal EHR database, which included 233 academic and community oncology practices. In each of the three disease cohorts, adoption was seen within 4 months for at least 60% of patients. Uptake was quickest for the melanoma patients, 76% of whom received a checkpoint inhibitor within 4 months, investigators wrote. Factors for the fast adoption could include high disease severity, a preference for novelty, perceived gains over existing treatments, and promotional activities, such as media reports and advertising directly to consumers, they wrote.
More patients in real-world practice were aged 65 years or older, ranging from as little as 61% at the lowest end of the range at one center to as much as 64% at the highest end at another. In the clinical trials, the percentage of patients aged 65 years or older ranged from 32% in one trial to 41% in another. Researchers wrote that these higher real-world rates are concerning because there are still questions regarding whether differences in immune responses will cause differences in efficacy between older and younger patients, as well as safety considerations among different age groups.
“Although data suggest that outcomes are similar between older and younger patients receiving anti–PD-1 agents for melanoma, there is little evidence to guide anti–PD-1 treatment of older patients with NSCLC [non–small cell lung cancer],” Dr. Gross and his coinvestigators wrote.
Investigators wrote that the findings are cause for caution.
“As FDA officials develop more flexible standards for approval, which the 21st Century Cures Act requires them to do, it is possible that many patients will receive drugs before much is known about clinical outcomes,” Dr. Gross said. “Further integrations of real-world evidence might allow the FDA to better assess the drugs that they approve on the basis of nonrepresentative trial participants.”
SOURCE: Gross C et al. JAMA Oncol. 2018 May 10. doi: 10.1001/jamaoncol.2018.0798.
FROM JAMA ONCOLOGY
Key clinical point: Fast adoption but older patient age is seen in clinical use of checkpoint inhibitors.
Major finding: About 60% of patients were aged 65 years and older in clinical settings, compared with about 30%-40% in trials.
Study details: A retrospective study of data from the Flatiron Health longitudinal EHR database, which includes 233 academic and community practices.
Disclosures: The study was sponsored by the National Institutes of Health. Investigators reported receiving financial support from Bristol-Myers Squibb, Eli Lilly, Johnson & Johnson, and other companies.
Source: Gross C et al. JAMA Oncology. 2018 May 10. doi: 10.1001/jamaoncol.2018.0798.
Enhanced Melanoma Diagnosis With Multispectral Digital Skin Lesion Analysis
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods
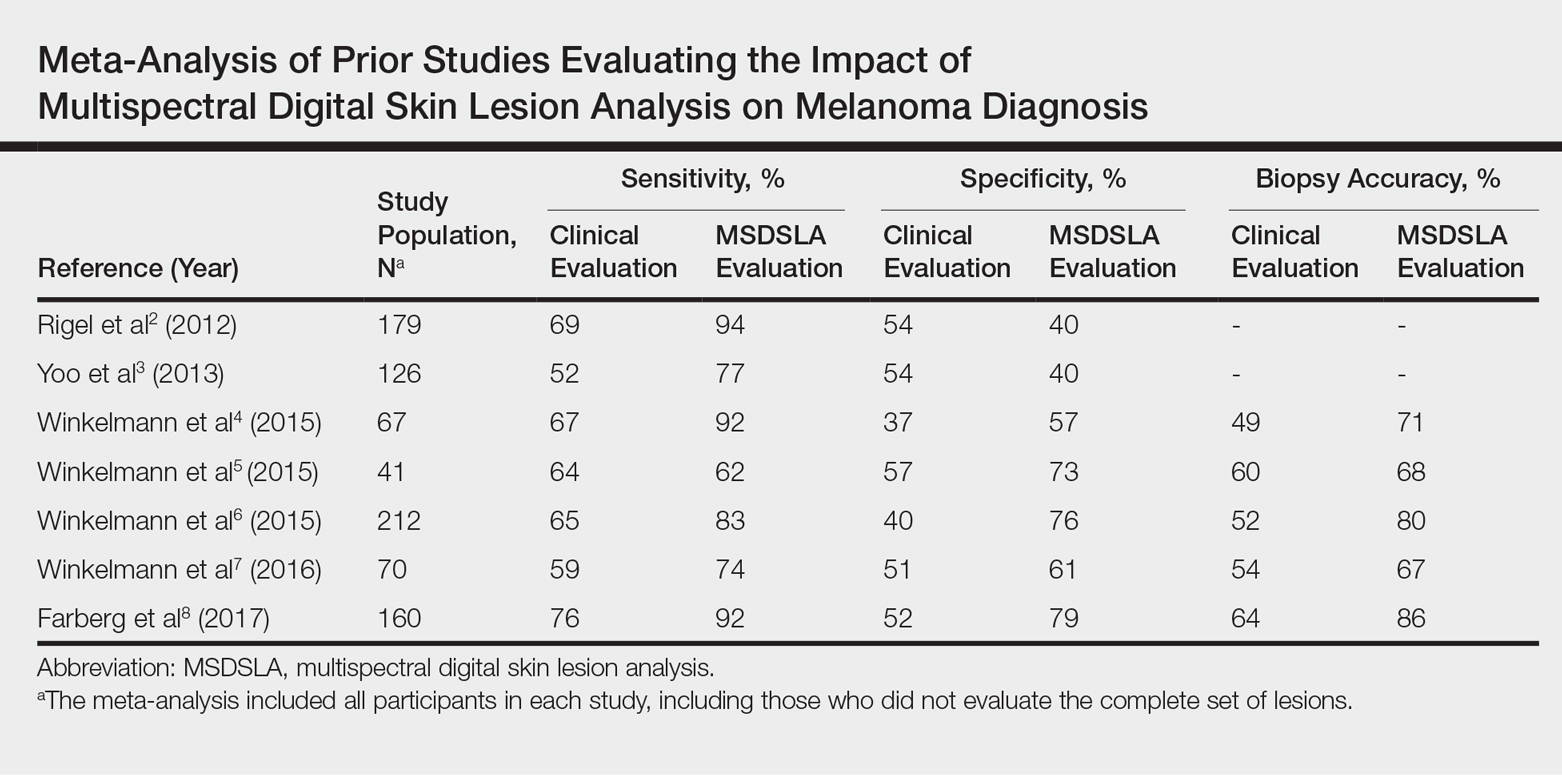
Results
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.
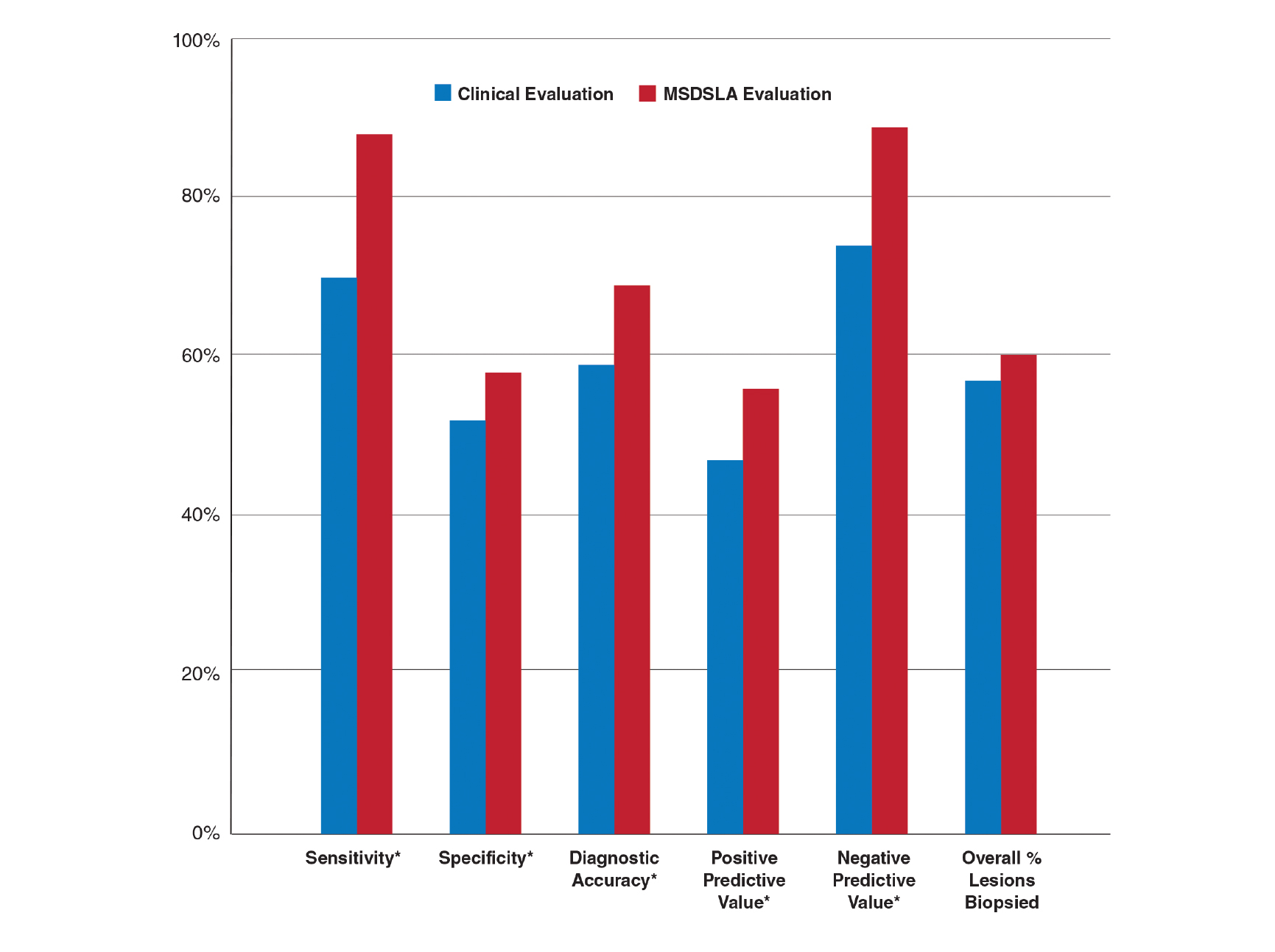
Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods

Results
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.

Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
Early detection of melanoma, which is known to improve survival rates, remains a challenge for dermatologists. Suspicious pigmented lesions typically are evaluated via clinical examination and dermoscopy; however, new technologies are being developed to provide additional objective information for clinicians to incorporate into their biopsy decisions.
Multispectral digital skin lesion analysis (MSDSLA) uses 10 bands of visible and near-infrared light (430–950 nm) to image and analyze pigmented skin lesions (PSLs) down to 2.5 mm below the skin surface and measures the distribution of melanin using 75 unique algorithms to determine the degree of the morphologic disorder. Using a logical regression model previously validated on a set of 1632 PSLs, the probability of melanoma and probability of being a melanoma/PSL of high-risk malignant potential are then provided to the clinician.1
In this study, we analyzed aggregate data from 7 prior studies2-8 to better determine how MSDSLA impacts the biopsy decisions of dermatologists and nondermatologists following clinical examination and dermoscopic evaluation of PSLs.
Methods

Results
Overall sensitivity for the detection of melanoma or other high-grade PSLs improved from 70% on clinical and dermoscopic evaluation to 88% after MSDSLA information was provided (P<.0001), and specificity increased from 52% to 58% (P<.001). Diagnostic accuracy also improved from 59% on clinical evaluation to 69% after review of MSDSLA findings (P<.0001). The positive predictive value of biopsy decisions was 47% following clinical evaluation, which improved to 56% after evaluation of MSDSLA findings (P<.001), and the negative predictive value increased from 74% to 89% (P<.0001). The overall percentage of lesions selected for biopsy did not significantly change following MSDSLA data integration (57% vs 60%)(Figure). Given that similar numbers of lesions were biopsied with improved sensitivity and specificity, the integration of MSDSLA data into the biopsy decision led to an improved biopsy ratio (ratio of melanomas biopsied to total biopsies) and fewer unnecessary biopsies.

Comment
Our broad analysis further supported the findings of prior studies that decisions to biopsy clinically suspicious PSLs are more sensitive, specific, and accurate when practitioners are provided MSDSLA information following clinical examination.2-8
Given the evolution in health care economics, it is clear that greater emphasis will continue to be placed on superior, evidence-based, effective care. The reported diagnostic sensitivities and specificities of clinical evaluation and dermoscopy for melanoma detection vary widely throughout the literature, with sensitivities ranging from 58% to over 90% and specificities ranging from 77% to 99%.9-11
Our study had several limitations. For this analysis to be more representative of lesion biopsy selection in the clinical setting, biopsy sensitivity (correctly identifying lesions appropriate for biopsy) vs melanoma sensitivity (identifying a lesion as melanoma) was used.13 The overall sensitivity found was within the range of prior studies,2-8 but this approach may have potentially led to a lower specificity due to an increased number of lesions biopsied. Additionally, the melanomas selected for these studies were early (malignant melanoma in situ or mean thickness of invasive malignant melanoma of 0.3 mm), and the nonmelanomas (including low-grade dysplastic nevi) were not necessarily diagnostically straightforward. This may have led to the clinical and dermoscopic sensitivity and specificity noted being lower than in some prior studies.9-11
The risk of missing a melanoma with MSDSLA devices has led manufacturers to strive for a high sensitivity for their devices, leading to lower specificity as a consequence. For this reason and other ambiguous practical considerations (eg, device and patient costs, difficulty with insurance reimbursement), the adoption of this technology into routine clinical practice has remained relatively static; however, using enhanced diagnostic technologies such as MSDSLA may help with more accurate identification of high-risk PSLs, thereby leading to earlier detection and overall less expensive, more cost-effective treatment of melanoma.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
- Monheit G, Cognetta AB, Ferris L, et al. The performance of MelaFind: a prospective multicenter study. Arch Dermatol. 2011;147:188-194.
- Rigel DS, Roy M, Yoo J, et al. Impact of guidance from a computer-aided multispectral digital skin lesion analysis device on decision to biopsy lesions clinically suggestive of melanoma. Arch Dermatol. 2012;148:541-543.
- Yoo J, Rigel DS, Roy M, et al. Impact of guidance from a multispectral digital skin lesion analysis device on dermatology residents decisions to biopsy lesions clinically suggestive of melanoma. J Am Acad Dermatol. 2013;68:AB152.
- Winkelmann RR, Yoo J, Tucker N, et al. Impact of guidance provided by a multispectral digital skin lesion analysis device following dermoscopy on decisions to biopsy atypical melanocytic lesions. J Clin Aesthet Dermatol. 2015;8:21-24.
- Winkelmann RR, Hauschild A, Tucker N, et al. The impact of multispectral digital skin lesion analysis on German dermatologist decisions to biopsy atypical pigmented lesions with clinical characteristics of melanoma. J Clin Aesthet Dermatol. 2015;8:27-29.
- Winkelmann RR, Tucker N, White R, et al. Pigmented skin lesion biopsies after computer-aided multispectral digital skin lesion analysis. J Am Osteopath Assoc. 2015;115:666-669.
- Winkelmann RR, Farberg AS, Tucker N, et al. Enhancement of international dermatologists’ pigmented skin lesion biopsy decisions following dermoscopy with subsequent integration of multispectral digital skin lesion analysis [published online July 1, 2016]. J Clin Aesthet Dermatol. 2016;9:53-55.
- Farberg AS, Winkelmann RR, Tucker N, et al. The impact of quantitative data provided by a multi-spectral digital skin lesion analysis device on dermatologists’ decisions to biopsy pigmented lesions [published online September 1, 2017]. J Clin Aesthet Dermatol. 2017;10:24-26.
- Wolf IH, Smolle J, Soyer HP, et al. Sensitivity in the clinical diagnosis of malignant melanoma. Melanoma Res. 1998;8:425-429.
- Kittler H, Pehamberger H, Wolff K, et al. Diagnostic accuracy of dermoscopy. Lancet Oncol. 2002;3:159-165.
- Ascierto PA, Palmieri G, Celentano E, et al. Sensitivity and specificity of epiluminescence microscopy: evaluation on a sample of 2731 excised cutaneous pigmented lesions: the Melanoma Cooperative Study. Br J Dermatol. 2000;142:893-898.
- Carli P, Nardini P, Crocetti E, et al. Frequency and characteristics of melanomas missed at a pigmented lesion clinic: a registry-based study. Melanoma Res. 2004;14:403-407.
- Friedman RJ, Gutkowicz-Krusin D, Farber MJ, et al. The diagnostic performance of expert dermoscopists vs a computer-vision system on small-diameter melanomas. Arch Dermatol. 2008;144:476-482.
Practice Points
- Multispectral digital skin lesion analysis (MSDSLA) can be a valuable tool in the evaluation of pigmented skin lesions (PSLs).
- MSDSLA may help to better identify high-risk PSLs and improve cost of care.
Mohs Micrographic Surgery for Digital Melanoma and Nonmelanoma Skin Cancers
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.
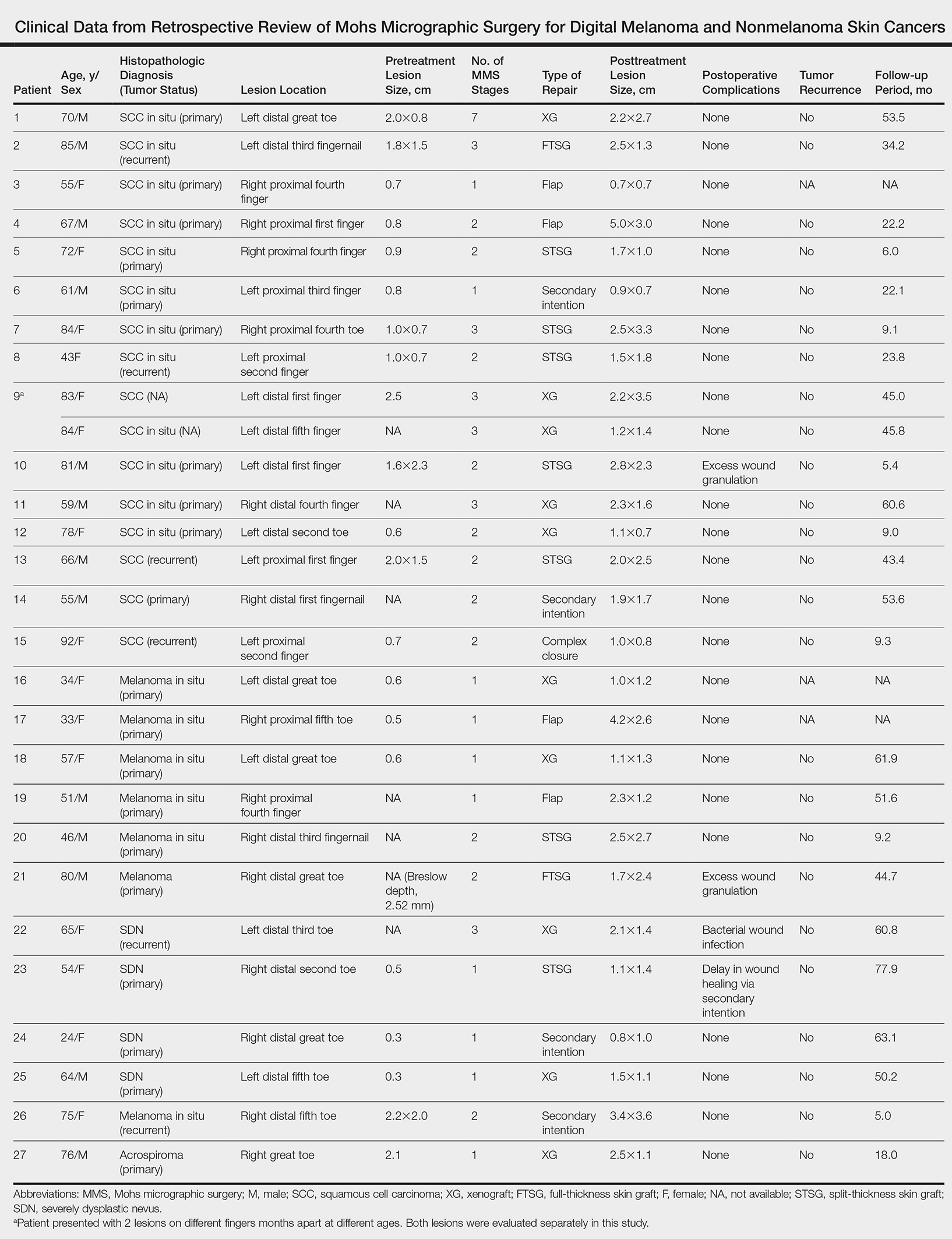
Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.
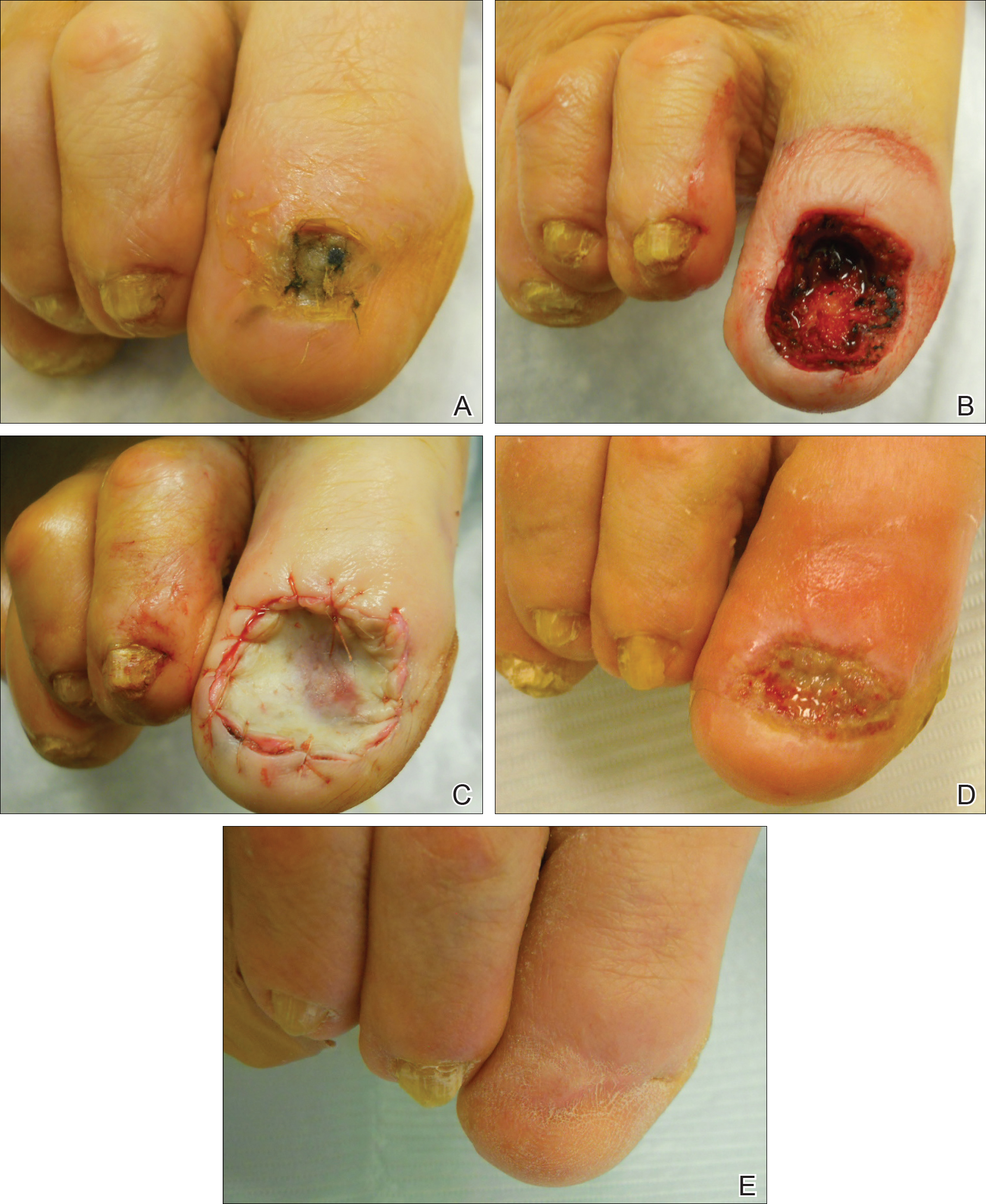
Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.

Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.

Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
Mohs micrographic surgery (MMS) is a specialized surgical technique for the treatment of melanoma and nonmelanoma skin cancers (NMSCs).1-3 The procedure involves surgical excision, histopathologic examination, precise mapping of malignant tissue, and wound management. Indications for MMS in skin cancer patients include recurring lesions, lesions in high-risk anatomic locations, aggressive histologic subtypes (ie, morpheaform, micronodular, infiltrative, high-grade, poorly differentiated), perineural invasion, large lesion size (>2 cm in diameter), poorly defined lateral or vertical clinical borders, rapid growth of the lesion, immunocompromised status, and sites of positive margins on prior excision. The therapeutic advantages of MMS include tissue conservation and optimal margin control in cosmetically or functionally sensitive areas, such as acral sites (eg, hands, feet, digits).1,3
The intricacies of the nail apparatus complicate diagnostic biopsy and precise delineation of peripheral margins in digital skin cancers; thus, early diagnosis and intraoperative histologic examination of the margins are essential. Traditionally, the surgical approach to subungual cutaneous tumors such as melanoma has included digital amputation4; however, a study of the treatment of subungual melanoma revealed no difference in survival based on the level of amputation, therefore advocating for less radical treatment.4
Interestingly, MMS for cutaneous tumors localized to the digits is not frequently reviewed in the dermatologic literature. We present a retrospective case series evaluating the clinical outcomes of digital melanoma and NMSCs treated with MMS.
Methods
A retrospective chart review was performed at a private dermatology practice to identify patients who underwent MMS for melanoma or NMSC localized to the digits from January 2009 to December 2014. All patients were treated in the office by 1 Mohs surgeon (A.H.) and were evaluated before and after MMS. Data were collected from the electronic medical record of the practice, including patient demographics, histopathologic diagnosis, tumor status (primary or recurrent lesion), anatomic site of the tumor, preoperative and postoperative size of the lesion, number of MMS stages, surgical repair technique, postoperative complications, and follow-up period.
Results
Twenty-seven patients (13 male, 14 female) with a total of 28 lesions (malignant melanoma or NMSC) localized to the digits were identified (Table). The mean age at the time of MMS was 64.07 years.

Surgical techniques used for repair following MMS included xenograft (10/28 [35.71%]); split-thickness skin graft (7/28 [25.0%]); secondary intention (4/28 [14.29%]); flap (4/28 [14.29%]); full-thickness skin graft (2/28 [7.14%]); and complex closure (1/28 [3.57%]). Clinical preoperative, operative, and postoperative photos from Patient 21 in this series are shown here (Figure). Two patients required bony phalanx resection due to invasion of the tumor into the periosteum: 1 had a malignant melanoma (Breslow depth, 2.52 mm); the other had an SCC. In addition, following removal of a severely dysplastic nevus, debulked tissue revealed melanoma in 1 patient.

Postoperative complications were noted in 4 (14.29%) of 28 MMS procedures, including bacterial wound infection (3.57%), excess granulation tissue that required wound debridement (7.14%), and delay in wound healing (3.57%). Follow-up data were available for 25 of the 28 MMS procedures (mean follow-up, 35.4 months), during which no recurrences were observed.
Comment
Mohs micrographic surgery is a specialized technique used in the treatment of cutaneous tumors, including basal cell carcinoma, SCC, melanoma in situ, atypical fibroxanthoma, dermatofibrosarcoma protuberans, sebaceous carcinoma, microcystic adnexal carcinoma, and Merkel cell carcinoma, among other cutaneous tumors.1-3 Mohs micrographic surgery provides the advantage of tissue conservation as well as optimal margin control in cosmetically or functionally sensitive areas while providing a higher cure rate than surgical excision. During the procedure, the surgical margin is examined histologically, thus ensuring definitive removal of the tumor but minimal loss of surrounding normal tissue.1-3 Mohs micrographic surgery is particularly useful for treating lesions on acral sites (eg, hands, feet, and digits).3-5
The treatment of digital skin cancers has evolved over the past 50 years with advancements resulting in more precise, tissue-sparing methods, in contrast to previous treatments such as amputation and wide local excision.6 More specifically, traditional digital amputation for the treatment of subungual melanoma has been reevaluated in multiple studies, which did not demonstrate a statistically significant difference in survival based on the level of amputation, thereby favoring less radical treatment.4,6 Moehrle et al7 found no statistical difference in recurrence rate when comparing patients with digital melanomas treated with partial amputation and those treated with digit-sparing surgery with limited excision and histologic evaluation of margins. Additionally, in a study conducted by Lazar et al,8 no recurrence of 13 subungual malignancies treated with MMS that utilized a full-thickness graft was reported at 4-year follow-up. In a large retrospective series of digital melanomas treated with MMS, Terushkin et al5 reported that 96.5% (55/57) of patients with primary melanomas that were treated with MMS avoided amputation, and the 5- and 10-year melanoma-specific survival rates for all patients treated with MMS were 95.0% and 82.6%, respectively.
In our study, cutaneous malignancies were located most often on the fingers, and the most common skin cancer identified was SCC in situ. The literature has shown that SCC in situ and SCC are the most common cutaneous neoplasms of the digits and nail unit.9 The most common specific anatomic site of cutaneous malignancy in our study was the great toe, followed by the fourth finger. A study conducted by Tan et al9 revealed that the great toe was the most common location of melanoma of the nail bed and subungual region, followed by the thumb. In contrast, primary subungual SCCs occur most frequently on the finger, with rare cases involving the toes.10
The etiology of digital SCC may involve extensive sun exposure, chronic trauma and wounds, and viral infection.9,11 More specifically, the dermatologic literature provides evidence of human papillomavirus (HPV) type 16 involvement in the pathogenesis of digital and periungual SCC. A genital-digital mechanism of spread has been implicated.11,12 An increased recurrence rate of HPV-associated digital SCCs has been reported following MMS, likely secondary to residual postsurgical HPV infection.11,12
Maintaining function and cosmesis of the hands, feet, and digits following MMS can be challenging, sometimes requiring skin grafts and flaps to close the defect. In the 28 MMS procedures evaluated in our study, 19 (67.9%) surgical defects were repaired with a graft (ie, split-thickness skin graft, full-thickness skin graft, xenograft), 4 (14.3%) with a flap (advancement and rotation), 4 (14.3%) by secondary intention, and 1 (3.6%) with primary complex closure.
Surgical grafts can be categorized based on the origin of the graft.2,13 Autografts, derived from the patient’s skin, are the most frequently used dermatologic graft and can be further categorized as full-thickness skin grafts, which include the epidermis and the entire dermis, thus preserving adnexal structures, and split-thickness skin grafts, which include the epidermis and partial dermis.2,13
A cross-sectional survey of fellowship-trained Mohs surgeons revealed that more than two-thirds of repairs for cutaneous acral cancers were performed using a primary closure technique, and one-fourth of closures were performed using secondary intention.15 Of the less frequently utilized skin-graft repairs, more were for acral lesions on the legs than on the arms.14 The type of procedure and graft used is dependent on multiple variables, including the anatomic location of the lesion and final size of the defect following MMS.2 Similarly, the use of specific types of sutures depends on the anatomic location of the lesion, relative thickness of the skin, degree of tension, and desired cosmetic result.15 The expertise of a hand surgeon may be required, particularly in cases in which the extensor tendon of the distal interphalangeal joint is compromised, manifested by a droopy fingertip when the hand is held horizontally. Additionally, special attention should be paid to removing the entire nail matrix before skin grafting for subungual tumors to avoid nail growth under the skin graft.
Evaluation of debulked tissue from digital skin cancers proved to be important in our study. In Patient 21, debulked tissue revealed melanoma following removal of a severely dysplastic nevus. This finding emphasizes the importance of complete excision of such lesions, as remaining underlying portions of the lesion can reveal residual tumor of the same or different histopathology.
In a prospective study, MMS was shown to have a low rate (0.91%; 95% confidence interval, 0.38%-1.45%) of surgical site infection in the absence of prophylactic antibiotics.16 The highest rates of surgical site infection were closely associated with flap closure. In our study, most patients had an uncomplicated and successful postoperative recovery. Only 1 (3.57%) of the 28 MMS procedures (Patient 22) was complicated by a bacterial wound infection postoperatively. The lesion removed in this case was a severely dysplastic melanocytic nevus on the toe. Infection resolved after a course of oral antibiotics, but the underlying cause of the wound infection in the patient was unclear. Other postoperative complications in our study included delayed wound healing and excess granulation tissue requiring wound debridement.
There are limited data in the dermatologic literature regarding outcomes following MMS for the treatment of cutaneous malignancies localized to the digits.
Additional limitations of this case review include its single-center and retrospective design, the small sample size, and 1 Mohs surgeon having performed all surgeries.
Conclusion
This study provides further evidence of the benefit of MMS for the treatment of malignant melanoma and NMSCs of the digits. This procedure provides margin-controlled excision of these malignant neoplasms while preserving maximal normal tissue, thereby providing patients with improved postoperative function and cosmesis. Long-term follow-up data demonstrating a lack of tumor recurrence underscores the assertion that MMS is safe and effective for the treatment of skin cancer of the digits.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
- Dim-Jamora KC, Perone JB. Management of cutaneous tumors with mohs micrographic surgery. Semin Plast Surg. 2008;22:247-256.
- McLeod MP, Choudhary S, Alqubaisy YA, et al. Indications for Mohs micrographic surgery. In: Nouri K, ed. Mohs Micrographic Surgery. New York, NY: Springer; 2012:5-13.
- Loosemore MP, Morales-Burgos A, Goldberg LH. Acral lentiginous melanoma of the toe treated using Mohs surgery with sparing of the digit and subsequent reconstruction using split-thickness skin graft. Dermatol Surg. 2013;39:136-138.
- Rayatt SS, Dancey AL, Davison PM. Thumb subungual melanoma: is amputation necessary? J Plast Reconstr Aesthet Surg. 2007;60:635-638.
- Terushkin V, Brodland DG, Sharon DJ, et al. Digit-sparing Mohs surgery for melanoma. Dermatol Surg. 2016;42:83-93.
- Viola KV, Jhaveri MB, Soulos PR, et al. Mohs micrographic surgery and surgical excision for nonmelanoma skin cancer treatment in the Medicare population. Arch Dermatol. 2012;148:473-477.
- Moehrle M, Metzger S, Schippert W. “Functional” surgery in subungual melanoma. Dermatol Surg. 2003;29:366-374.
- Lazar A, Abimelec P, Dumontier C, et al. Full thickness skin graft from nail unit reconstruction. J Hand Surg Br. 2005;30:194-198.
- Tan KB, Moncrieff M, Thompson JF, et al. Subungual melanoma: a study of 124 cases highlighting features of early lesions, potential for histologic reports. Am J Surg Pathol. 2007;31:1902-1912.
- Nasca MR, Innocenzi D, Micali G. Subungual squamous cell carcinoma of the toe: report on three cases. Dermatol Surg. 2004;30:345-348.
- Dika E, Piraccini BM, Balestri RR, et al. Mohs surgery for squamous cell carcinoma of the nail: report of 15 cases. our experience and a long-term follow-up. Br J Dermatol. 2012;167:1310-1314.
- Alam M, Caldwell JB, Eliezri YD. Human papillomavirus-associated digital squamous cell carcinoma: literature review and report of 21 new cases. J Am Acad Dermatol. 2003;48:385-393.
- Filho L, Anselmo J, Dadalti P, et al. Skin grafts in cutaneous oncology. Braz Ann Dermatol. 2006;81:465-472.
- Raimer DW, Group AR, Petitt MS, et al. Porcine xenograft biosynthetic wound dressings for the management of postoperative Mohs wounds. Dermatol Online J. 2011;17:1.
- Alam M, Helenowksi IB, Cohen JL, et al. Association between type of reconstruction after Mohs micrographic surgery and surgeon-, patient-, and tumor-specific features: a cross-sectional study. Dermatol Surg. 2013;39:51-55.
- Rogers HD, Desciak EB, Marcus RP, et al. Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol. 2010;63:842-851.
Practice Points
- Melanoma and nonmelanoma skin cancers of the digits traditionally have been treated with wide local surgical excision and even amputation.
- Conservative tissue sparing techniques such as Mohs micrographic surgery can be used to treat digital skin cancers with high cure rates and improved functional and cosmetic results.
Penile Squamous Cell Carcinoma With Urethral Extension Treated With Mohs Micrographic Surgery
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.
Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.


Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).
Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.

Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.



Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).

Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
Penile squamous cell carcinoma (SCC) with considerable urethral extension is uncommon and difficult to manage. It often is resistant to less invasive and nonsurgical treatments and frequently results in partial or total penectomy, which can lead to cosmetic disfigurement, functional issues, and psychological distress. We report a case of penile SCC in situ with considerable urethral extension with a focus of cells suspicious for moderately well-differentiated and invasive SCC that was treated with
Mohs micrographic surgery with distal urethrectomy and reconstruction is a valuable treatment technique for cases of SCC involving the glans penis and distal urethra. It offers equivalent or better overall cure rates compared to more radical interventions. Additionally, preservation of the penis with MMS spares patients from considerable physical and psychosocial morbidity. Our case, along with growing body of literature,1-4 calls on dermatologists and urologists to consider MMS as a treatment for penile SCC with or without urethral involvement.
Case Report
A 61-year-old man presented to the dermatology department with a pruritic lesion on the penis that had been present for 6 years. Shave biopsy demonstrated SCC in situ with a focus of cells suspicious for moderately well-differentiated and invasive SCC. Physical examination revealed an ill-defined, 2.2×1.9-cm, pink, eroded plaque involving the tip of the penis and surrounding the external urinary meatus (Figure 1). There was no palpable inguinal lymphadenopathy.

Distal penectomy and lymph node biopsy was recommended following evaluation by the urologic oncology department, but the patient declined these interventions and presented to our dermatology department (A.H.) for a second opinion. The tumor, including the invasive perineural portion, was removed using MMS several weeks after initially presenting to urologic oncology. Ventral meatotomy allowed access to the SCC in situ portion extending proximally up the pendulous urethra (Figure 2). Clear margins were obtained after the eighth stage of MMS, which required removal of 4 to 5 cm of the distal urethra (Figure 3). Reconstruction of the wound required urethral advancement, urethrostomy, and meatoplasty. A positive outcome was achieved with preservation of the length and shape of the penis as well as the cosmetic appearance of the glans penis (Figure 4). The patient was satisfied with the outcome. At 49 months’ follow-up, no evidence of local recurrence or disease progression was noted, and the distal urethrostomy remained intact and functional.



Comment
Penile SCC is a rare malignancy that represents between 0.4% and 0.6% of all malignant tumors in the United States and occurs most commonly in men aged 50 to 70 years.4 The incidence is higher in developing countries, approaching 10% of malignancies in men. It occurs most commonly on the glans penis, prepuce, and coronal sulcus, and has multiple possible appearances, including erythematous and indurated, warty and exophytic, or flat and ulcerated lesions.5 Some reports indicate that more than 40% of penile SCCs are attributable to human papilloma virus,6 while lack of circumcision, chronic inflammation, poor hygiene, balanitis xerotica obliterans, penile trauma, human immunodeficiency virus, UVA treatment of penile psoriasis, and tobacco use are known risk factors.5
Invasive penile SCC generally is treated with penectomy (partial or total), radiation therapy, or MMS; SCC in situ can be treated with topical chemotherapy, laser therapy, and wide local excision (2-cm margins) including circumcision, complete glansectomy, or MMS.5 Squamous cell carcinoma in situ with urethral involvement treated with nonsurgical therapies is associated with higher recurrence rates, ultimately necessitating more aggressive treatments, most commonly partial penectomy.7 The high local recurrence rate of SCC in situ with urethral involvement treated with nonsurgical therapies reflects the fact that determining the presence of urethral extension is difficult and, if present, is inherently inaccessible to these local therapies because the urethra is not an outward-facing tissue surface; MMS represents one possible solution to these issues.
Across all treatment modalities, the most prognostic factor of cancer-specific survival in patients with penile SCC is pelvic lymph node involvement. Some reports cite 5-year survival rates as low as 0% in the setting of pelvic lymph node involvement,5 whereas others had cited rates of 29% to 40%4; 5-year survival rates of higher than 85% have been reported in node-negative patients.4 Recurrence rates vary widely by treatment modality, ranging from less than 10% with partial penectomy and long-term follow-up8 and up to 50% within 2 years with penile-preserving approaches (eg, topical chemotherapy, laser therapy, radiotherapy).5 Multiple case series of penile cancer (the most common of which was SCC/SCC in situ) treated with MMS report comparable and at times superior survival and recurrence data (Table).1-4 Slightly higher recurrences of penile SCC treated with MMS compared to penectomy have been reported, along with considerably higher recurrence rates compared to nonpenile cutaneous SCC treated with MMS (reported to be less than 3%).4 The elastic and expansile nature of penile tissue may lead to distortion from swelling/local anesthesia when taking individual Mohs layers. Additionally, as a large percentage of penile SCCs are attributable to human papillomavirus, difficulty in detecting human papilloma virus–infected cells (which may have oncogenic potential) with the naked eye or histologically with typical staining techniques may help explain the higher recurrence rate of penile SCC treated with MMS compared to penectomy. Despite the higher recurrence rates, survival is comparable or higher in cases treated with MMS (Table).

Partial penectomy also has a negative impact on health-related quality of life. Kieffer et al9 compared the impact of penile-sparing surgery (PSS)(including MMS) versus partial or total penectomy on sexual function and health-related quality of life in 90 patients with penile cancer. Although the association between the extent of surgery (partial penectomy/total penectomy/PSS) surgery type and extent and most outcome measures was not statistically significant, partial penectomy was associated with significantly more problems with orgasm (P=.031), concerns about appearance (P=.008), interference in daily life (P=.032), and urinary function (P<.0001) when compared to patients treated with PSS.9 Although this study included only laser/local excision with or without circumcision or glans penis amputation with or without reconstruction as PSSs and did not explicitly include MMS, MMS is clearly a tissue-sparing technique and the study results are generaliz
Conclusion
Penile SCC with considerable urethral extension is uncommon, difficult to manage, and often is resistant to less invasive and nonsurgical treatments. As a result, partial or total penectomy is sometimes necessary. Such cases benefit from MMS with distal urethrectomy and reconstruction because MMS provides equivalent or better overall cure rates compared to more radical interventions.1-4 Importantly, preservation of the penis with MMS can spare patients considerable physical and psychosocial morbidity. Partial penectomy is associated with more health-related quality-of-life problems with orgasm, concerns about appearance, interference in daily life, and urinary function compared to PSSs such as MMS.9 This case, and a growing body of literature, are a call to dermatologists and urologists to consider MMS as a treatment for penile SCC, even with involvement of the urethra.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
- Brown MD, Zachary CB, Grekin RC, et al. Penile tumors: their management by Mohs micrographic surgery. J Dermatol Surg Oncol. 1987;13:1163-1167.
- Mohs FE, Snow SN, Larson PO. Mohs micrographic surgery for penile tumors. Urol Clin North Am. 1992;19:291-304.
- Shindel AW, Mann MW, Lev RY, et al. Mohs micrographic surgery for penile cancer: management and long-term followup. J Urol. 2007;178:1980-1985.
- Machan M, Brodland D, Zitelli J. Penile squamous cell carcinoma: penis-preserving treatment with Mohs micrographic surgery. Dermatol Surg. 2016;42:936-944.
- Spiess PE, Horenblas S, Pagliaro LC, et al. Current concepts in penile cancer. J Natl Compr Canc Netw. 2013;11:617-624.
- Hernandez BY, Barnholtz-Sloan J, German RR, et al. Burden of invasive squamous cell carcinoma of the penis in the United States, 1998-2003. Cancer. 2008;113(10 suppl):2883-2891.
- Nash PA, Bihrle R, Gleason PE, et al. Mohs micrographic surgery and distal urethrectomy with immediate urethral reconstruction for glanular carcinoma in situ with significant urethral extension. Urology. 1996;47:108-110.
- Djordjevic ML, Palminteri E, Martins F. Male genital reconstruction for the penile cancer survivor. Curr Opin Urol. 2014;24:427-433.
- Kieffer JM, Djajadiningrat RS, van Muilekom EA, et al. Quality of life for patients treated for penile cancer. J Urol. 2014;192:1105-1110.
Resident Pearl
- Penile squamous cell carcinoma (SCC) often is treated with partial or total penectomy, especially when there is urethral extension. Mohs micrographic surgery for penile SCC results in equivalent or better overall cure rates and decreases morbidity.
Circulating biomarkers predicted melanoma survival
Among patients with metastatic melanoma, detectable circulating levels of BRAFV600 mutated circulating tumor DNA and elevated levels of circulating hepatocyte growth factor separately predicted significantly worse survival, regardless of treatment.
After adjusting for lactate dehydrogenase, ECOG (Eastern Cooperative Oncology Group) status, disease stage, and treatment, hazard ratios for overall survival were 1.75 (95% confidence interval, 1.35-2.28) for detectable versus undetectable circulating tumor DNA (ctDNA) and 1.24 (95% CI, 1.00-1.53) for high versus low circulating hepatocyte growth factor (cHGF), reported William Lu, PhD, of Genentech in South San Francisco, with his associates. Their retrospective, exploratory analysis of the phase 3 BRIM-3 trial was published in JCO Precision Oncology.
BRIM-3 was a multicenter, open-label trial of 675 patients with previously untreated unresectable stage IIIC or stage IV melanoma that tested positive for the BRAFV600 mutation. Patients received either vemurafenib or dacarbazine therapy.
Baseline ctDNA, baseline cHGF, and ECOG performance status most strongly predicted overall survival, said the investigators. Patients whose mutant ctDNA fraction exceeded 0.039 had an 11.5-month shorter median overall survival on vemurafenib (P less than .001) and a 13.9-month shorter median overall survival on dacarbazine (P less than .001) than patients whose mutant ctDNA fraction was less than 0.039, they explained. Similarly, median overall survival times were 5.4 months shorter for vemurafenib (P = .002) and 12.3 months shorter for dacarbazine (P less than .001) when patients had high (more than 438 pg/mL) versus low circulating cHGF.
Median overall survival for vemurafenib was shortest (7.3 months) in the subgroup of 64 patients with detectable BRAFV600 ctDNA and elevated cHGF, according to the researchers. In contrast, median overall survival for vemurafenib was longest (22.0 months) when patients had undetectable ctDNA and an ECOG status of 0.
“Although similar studies have been performed, to our knowledge, our study is unique in its size and in its design as a randomized trial,” Dr. Lu and associates concluded. “The biomarkers in this study can be readily acquired and measured, and require only small volumes of plasma.” They suggested validating the findings in a separate dataset.
F. Hoffman-La Roche provided funding. Roche Molecular Systems makes the Cobas 4800 BRAFV600 Mutation Test used in the study. Dr. Lu disclosed employment and research funding from Genentech/Roche.
SOURCE: Lu W et al. JCO Precis Oncol. 2018 Apr 25. doi: 10.1200/PO.17.00168.
Among patients with metastatic melanoma, detectable circulating levels of BRAFV600 mutated circulating tumor DNA and elevated levels of circulating hepatocyte growth factor separately predicted significantly worse survival, regardless of treatment.
After adjusting for lactate dehydrogenase, ECOG (Eastern Cooperative Oncology Group) status, disease stage, and treatment, hazard ratios for overall survival were 1.75 (95% confidence interval, 1.35-2.28) for detectable versus undetectable circulating tumor DNA (ctDNA) and 1.24 (95% CI, 1.00-1.53) for high versus low circulating hepatocyte growth factor (cHGF), reported William Lu, PhD, of Genentech in South San Francisco, with his associates. Their retrospective, exploratory analysis of the phase 3 BRIM-3 trial was published in JCO Precision Oncology.
BRIM-3 was a multicenter, open-label trial of 675 patients with previously untreated unresectable stage IIIC or stage IV melanoma that tested positive for the BRAFV600 mutation. Patients received either vemurafenib or dacarbazine therapy.
Baseline ctDNA, baseline cHGF, and ECOG performance status most strongly predicted overall survival, said the investigators. Patients whose mutant ctDNA fraction exceeded 0.039 had an 11.5-month shorter median overall survival on vemurafenib (P less than .001) and a 13.9-month shorter median overall survival on dacarbazine (P less than .001) than patients whose mutant ctDNA fraction was less than 0.039, they explained. Similarly, median overall survival times were 5.4 months shorter for vemurafenib (P = .002) and 12.3 months shorter for dacarbazine (P less than .001) when patients had high (more than 438 pg/mL) versus low circulating cHGF.
Median overall survival for vemurafenib was shortest (7.3 months) in the subgroup of 64 patients with detectable BRAFV600 ctDNA and elevated cHGF, according to the researchers. In contrast, median overall survival for vemurafenib was longest (22.0 months) when patients had undetectable ctDNA and an ECOG status of 0.
“Although similar studies have been performed, to our knowledge, our study is unique in its size and in its design as a randomized trial,” Dr. Lu and associates concluded. “The biomarkers in this study can be readily acquired and measured, and require only small volumes of plasma.” They suggested validating the findings in a separate dataset.
F. Hoffman-La Roche provided funding. Roche Molecular Systems makes the Cobas 4800 BRAFV600 Mutation Test used in the study. Dr. Lu disclosed employment and research funding from Genentech/Roche.
SOURCE: Lu W et al. JCO Precis Oncol. 2018 Apr 25. doi: 10.1200/PO.17.00168.
Among patients with metastatic melanoma, detectable circulating levels of BRAFV600 mutated circulating tumor DNA and elevated levels of circulating hepatocyte growth factor separately predicted significantly worse survival, regardless of treatment.
After adjusting for lactate dehydrogenase, ECOG (Eastern Cooperative Oncology Group) status, disease stage, and treatment, hazard ratios for overall survival were 1.75 (95% confidence interval, 1.35-2.28) for detectable versus undetectable circulating tumor DNA (ctDNA) and 1.24 (95% CI, 1.00-1.53) for high versus low circulating hepatocyte growth factor (cHGF), reported William Lu, PhD, of Genentech in South San Francisco, with his associates. Their retrospective, exploratory analysis of the phase 3 BRIM-3 trial was published in JCO Precision Oncology.
BRIM-3 was a multicenter, open-label trial of 675 patients with previously untreated unresectable stage IIIC or stage IV melanoma that tested positive for the BRAFV600 mutation. Patients received either vemurafenib or dacarbazine therapy.
Baseline ctDNA, baseline cHGF, and ECOG performance status most strongly predicted overall survival, said the investigators. Patients whose mutant ctDNA fraction exceeded 0.039 had an 11.5-month shorter median overall survival on vemurafenib (P less than .001) and a 13.9-month shorter median overall survival on dacarbazine (P less than .001) than patients whose mutant ctDNA fraction was less than 0.039, they explained. Similarly, median overall survival times were 5.4 months shorter for vemurafenib (P = .002) and 12.3 months shorter for dacarbazine (P less than .001) when patients had high (more than 438 pg/mL) versus low circulating cHGF.
Median overall survival for vemurafenib was shortest (7.3 months) in the subgroup of 64 patients with detectable BRAFV600 ctDNA and elevated cHGF, according to the researchers. In contrast, median overall survival for vemurafenib was longest (22.0 months) when patients had undetectable ctDNA and an ECOG status of 0.
“Although similar studies have been performed, to our knowledge, our study is unique in its size and in its design as a randomized trial,” Dr. Lu and associates concluded. “The biomarkers in this study can be readily acquired and measured, and require only small volumes of plasma.” They suggested validating the findings in a separate dataset.
F. Hoffman-La Roche provided funding. Roche Molecular Systems makes the Cobas 4800 BRAFV600 Mutation Test used in the study. Dr. Lu disclosed employment and research funding from Genentech/Roche.
SOURCE: Lu W et al. JCO Precis Oncol. 2018 Apr 25. doi: 10.1200/PO.17.00168.
FROM JCO PRECISION ONCOLOGY
Key clinical point: Among patients with metastatic melanoma, high circulating levels of BRAFV600 mutated circulating tumor DNA and circulating hepatocyte growth factor predicted significantly worse overall survival, irrespective of treatment.
Major finding: Adjusted hazard ratios for overall survival were 1.75 for high versus undetectable circulating tumor DNA and 1.24 for high versus low circulating hepatocyte growth factor.
Study details: A retrospective, exploratory analysis of 675 patients with metastatic BRAFV600 mutated advanced melanoma.
Disclosures: F. Hoffman-La Roche provided funding. Roche Molecular Systems makes the Cobas 4800 BRAFV600 Mutation Test used in the study. Dr. Lu disclosed employment and research funding from Genentech/Roche.
Source: Lu W et al. JCO Precis Oncol. 2018 Apr 25. doi: 10.1200/PO.17.00168.