User login
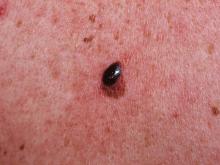
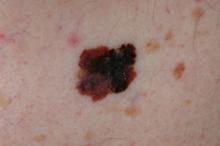
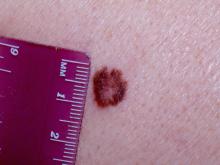
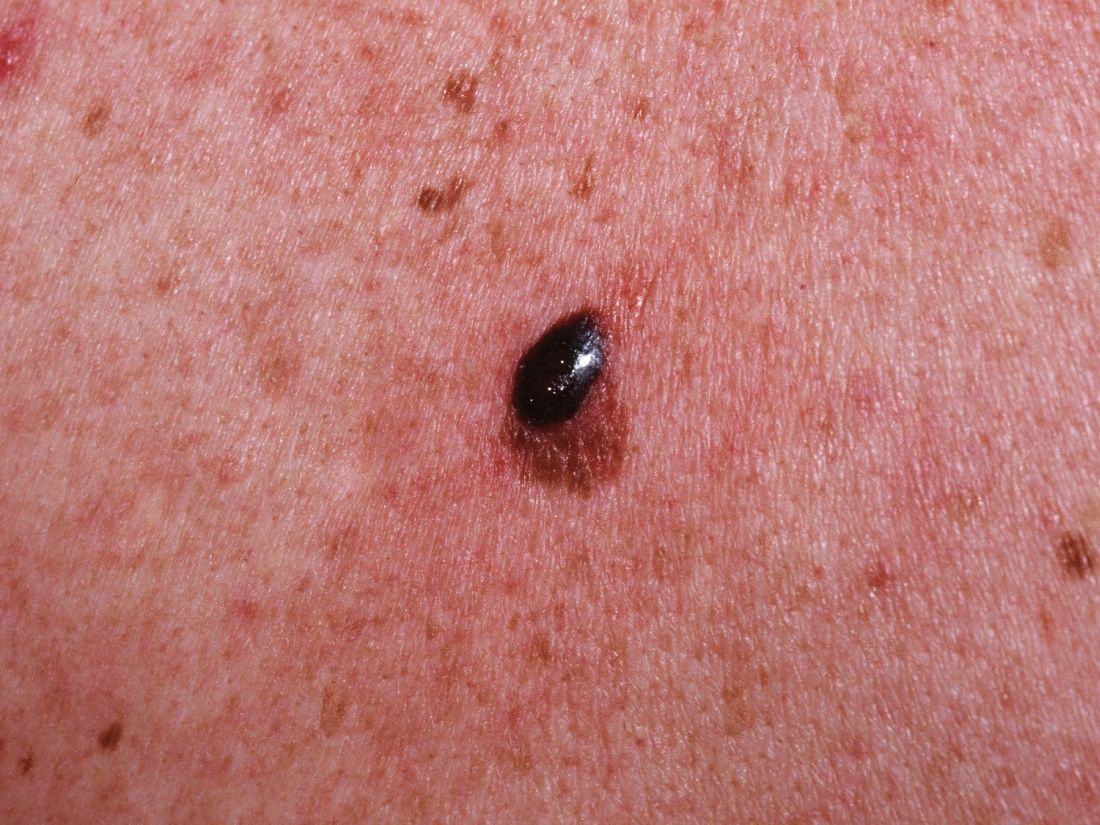
World Trade Center responders face greater cancer burden, including greater risk of multiple myeloma
Rescue and recovery workers who were involved in the aftermath of the World Trade Center disaster may face a greater cancer burden than the general population, according to two studies published in JAMA Oncology.
In particular, they may be at risk of developing multiple myeloma at an earlier age.
The first study was a closed-cohort study of 14,474 employees of the Fire Department of the City of New York (FDNY) who were exposed to the World Trade Center disaster but were cancer-free as of Jan. 1, 2012. The aim was to project cancer incidence from 2012 through 2031, based on data from the FDNY World Trade Center Health Program, and compare those rates with age-, race-, and sex-specific New York cancer rates from the general population.
The modeling projected a “modestly” higher number of cancer cases in the white male subgroup of rescue and recovery workers exposed to the World Trade Center (2,714 vs. 2,596 for the general population of New York; P less than .001). Specifically, the investigators projected significantly higher case counts of prostate cancer (1,437 vs. 863), thyroid cancer (73 vs. 57), and melanoma (201 vs. 131), compared with the general population in New York, but fewer lung (237 vs. 373), colorectal (172 vs. 267), and kidney cancers (66 vs. 132) (P less than .001 for all).
“Our findings suggest that the FDNY WTC-exposed cohort may experience a greater burden of cancer than would be expected from a population with similar demographic characteristics,” wrote Rachel Zeig-Owens, DrPH, from the Montefiore Medical Center and Albert Einstein College of Medicine, both in New York, and coauthors, highlighting prostate cancer as a particular concern.
However, they also acknowledged that the elevated rates observed in people exposed to the World Trade Center disaster could be a result of increased surveillance, even though they did attempt to correct for that, and that firefighters in general might face higher risks.
“It is possible that firefighters have a higher risk of cancer than the general population owing to exposures associated with the occupation,” they wrote. However occupation could also have the opposite effect, as rescue and recovery workers tend to have lower smoking rates, which may explain the relatively low rates of certain cancers such as lung cancer, they said.
A second study examined the effect of the World Trade Center disaster on the risk of multiple myeloma and monoclonal gammopathies in exposed firefighters.
The seroprevalence study of monoclonal gammopathies of undetermined significance (MGUS) in 781 exposed firefighters revealed that the age-standardized prevalence of these was 76% higher in this population than it was in a white male reference population living in Minnesota.
In particular, the age-standardized prevalence of light-chain MGUS was more than threefold higher in exposed firefighters, compared with the reference population.
Researchers also analyzed a case series of 16 exposed white male firefighters who received a diagnosis of multiple myeloma after Sept. 11, 2001. Of the 14 patients for whom data on the monoclonal protein isotype was available, half had light-chain multiple myeloma.
“These findings are of interest due to previously observed associations between light-chain multiple myeloma and light-chain MGUS and exposure to toxins, and chronic immune stimulation,” wrote Ola Landgren, MD, PhD, from the Memorial Sloan Kettering Cancer Center and his coauthors.
Seven patients were also assessed for CD20 expression – a marker of poorer prognosis – and 71% were found to be CD20-positive, a prevalence around 3.5-fold higher than that seen in the general population.
The cohort with multiple myeloma was diagnosed on average 12 years younger than those in the general population. The authors commented that this was unlikely to be caused by lead-time bias because the time from first symptoms to clinical manifestation of the disease is usually around 1 year.
“Taken together, our results show that environmental exposure due to the WTC attacks is associated with myeloma precursor disease (MGUS and light-chain MGUS) and may be a risk factor for the development of multiple myeloma at an earlier age, particularly the light-chain subtype,” the authors wrote.
The first study was supported by the National Institute of Occupational Safety and Health; no conflicts of interest were declared.
The second study was supported by the V Foundation for Cancer Research, the Byrne Fund for the benefit of Memorial Sloan-Kettering Cancer Center, the National Cancer Institute, the Albert Einstein Cancer Center, and the National Institute for Occupational Safety and Health; no conflicts of interest were declared.
SOURCE: Zeig-Owens R et al. JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0504. Landgren O et al. JAMA Oncology. 2018 April 16. doi: 10.1001/jamaoncol.2018.0509.
When the heroes of the World Trade Center are diagnosed with even a common cancer, there is a natural tendency to assume that the diagnosis is the result of their service during the disaster. However, it is important to appreciate that the firefighting profession is known to be associated with higher risks of monoclonal gammopathy of undetermined significance and multiple myelomas, compared with the general population.
Given that, it would have been preferable to compare the World Trade Center–exposed populations with an equally intensively screened, age-matched cohort of firefighters from another major city.
If we apply Sir Richard Doll’s rule that a single epidemiologic study cannot be persuasive until the lower bound of the 95% confidence interval is greater than three, the relative risks in the study by Landgren and colleagues are too small to be persuasive.
The predicted increases in cancers of the prostate, thyroid, and myeloma are interesting, but these have also been previously reported in firefighters from other cities.
Despite this, we owe it to these men and women to find the truth and determine the illnesses that are associated with their service.
Otis W. Brawley, MD, is chief medical and scientific officer and executive vice president of the American Cancer Society and a professor at Emory University, Atlanta. These comments are taken from an accompanying editorial (JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0498.) No conflicts of interest were declared.
When the heroes of the World Trade Center are diagnosed with even a common cancer, there is a natural tendency to assume that the diagnosis is the result of their service during the disaster. However, it is important to appreciate that the firefighting profession is known to be associated with higher risks of monoclonal gammopathy of undetermined significance and multiple myelomas, compared with the general population.
Given that, it would have been preferable to compare the World Trade Center–exposed populations with an equally intensively screened, age-matched cohort of firefighters from another major city.
If we apply Sir Richard Doll’s rule that a single epidemiologic study cannot be persuasive until the lower bound of the 95% confidence interval is greater than three, the relative risks in the study by Landgren and colleagues are too small to be persuasive.
The predicted increases in cancers of the prostate, thyroid, and myeloma are interesting, but these have also been previously reported in firefighters from other cities.
Despite this, we owe it to these men and women to find the truth and determine the illnesses that are associated with their service.
Otis W. Brawley, MD, is chief medical and scientific officer and executive vice president of the American Cancer Society and a professor at Emory University, Atlanta. These comments are taken from an accompanying editorial (JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0498.) No conflicts of interest were declared.
When the heroes of the World Trade Center are diagnosed with even a common cancer, there is a natural tendency to assume that the diagnosis is the result of their service during the disaster. However, it is important to appreciate that the firefighting profession is known to be associated with higher risks of monoclonal gammopathy of undetermined significance and multiple myelomas, compared with the general population.
Given that, it would have been preferable to compare the World Trade Center–exposed populations with an equally intensively screened, age-matched cohort of firefighters from another major city.
If we apply Sir Richard Doll’s rule that a single epidemiologic study cannot be persuasive until the lower bound of the 95% confidence interval is greater than three, the relative risks in the study by Landgren and colleagues are too small to be persuasive.
The predicted increases in cancers of the prostate, thyroid, and myeloma are interesting, but these have also been previously reported in firefighters from other cities.
Despite this, we owe it to these men and women to find the truth and determine the illnesses that are associated with their service.
Otis W. Brawley, MD, is chief medical and scientific officer and executive vice president of the American Cancer Society and a professor at Emory University, Atlanta. These comments are taken from an accompanying editorial (JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0498.) No conflicts of interest were declared.
Rescue and recovery workers who were involved in the aftermath of the World Trade Center disaster may face a greater cancer burden than the general population, according to two studies published in JAMA Oncology.
In particular, they may be at risk of developing multiple myeloma at an earlier age.
The first study was a closed-cohort study of 14,474 employees of the Fire Department of the City of New York (FDNY) who were exposed to the World Trade Center disaster but were cancer-free as of Jan. 1, 2012. The aim was to project cancer incidence from 2012 through 2031, based on data from the FDNY World Trade Center Health Program, and compare those rates with age-, race-, and sex-specific New York cancer rates from the general population.
The modeling projected a “modestly” higher number of cancer cases in the white male subgroup of rescue and recovery workers exposed to the World Trade Center (2,714 vs. 2,596 for the general population of New York; P less than .001). Specifically, the investigators projected significantly higher case counts of prostate cancer (1,437 vs. 863), thyroid cancer (73 vs. 57), and melanoma (201 vs. 131), compared with the general population in New York, but fewer lung (237 vs. 373), colorectal (172 vs. 267), and kidney cancers (66 vs. 132) (P less than .001 for all).
“Our findings suggest that the FDNY WTC-exposed cohort may experience a greater burden of cancer than would be expected from a population with similar demographic characteristics,” wrote Rachel Zeig-Owens, DrPH, from the Montefiore Medical Center and Albert Einstein College of Medicine, both in New York, and coauthors, highlighting prostate cancer as a particular concern.
However, they also acknowledged that the elevated rates observed in people exposed to the World Trade Center disaster could be a result of increased surveillance, even though they did attempt to correct for that, and that firefighters in general might face higher risks.
“It is possible that firefighters have a higher risk of cancer than the general population owing to exposures associated with the occupation,” they wrote. However occupation could also have the opposite effect, as rescue and recovery workers tend to have lower smoking rates, which may explain the relatively low rates of certain cancers such as lung cancer, they said.
A second study examined the effect of the World Trade Center disaster on the risk of multiple myeloma and monoclonal gammopathies in exposed firefighters.
The seroprevalence study of monoclonal gammopathies of undetermined significance (MGUS) in 781 exposed firefighters revealed that the age-standardized prevalence of these was 76% higher in this population than it was in a white male reference population living in Minnesota.
In particular, the age-standardized prevalence of light-chain MGUS was more than threefold higher in exposed firefighters, compared with the reference population.
Researchers also analyzed a case series of 16 exposed white male firefighters who received a diagnosis of multiple myeloma after Sept. 11, 2001. Of the 14 patients for whom data on the monoclonal protein isotype was available, half had light-chain multiple myeloma.
“These findings are of interest due to previously observed associations between light-chain multiple myeloma and light-chain MGUS and exposure to toxins, and chronic immune stimulation,” wrote Ola Landgren, MD, PhD, from the Memorial Sloan Kettering Cancer Center and his coauthors.
Seven patients were also assessed for CD20 expression – a marker of poorer prognosis – and 71% were found to be CD20-positive, a prevalence around 3.5-fold higher than that seen in the general population.
The cohort with multiple myeloma was diagnosed on average 12 years younger than those in the general population. The authors commented that this was unlikely to be caused by lead-time bias because the time from first symptoms to clinical manifestation of the disease is usually around 1 year.
“Taken together, our results show that environmental exposure due to the WTC attacks is associated with myeloma precursor disease (MGUS and light-chain MGUS) and may be a risk factor for the development of multiple myeloma at an earlier age, particularly the light-chain subtype,” the authors wrote.
The first study was supported by the National Institute of Occupational Safety and Health; no conflicts of interest were declared.
The second study was supported by the V Foundation for Cancer Research, the Byrne Fund for the benefit of Memorial Sloan-Kettering Cancer Center, the National Cancer Institute, the Albert Einstein Cancer Center, and the National Institute for Occupational Safety and Health; no conflicts of interest were declared.
SOURCE: Zeig-Owens R et al. JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0504. Landgren O et al. JAMA Oncology. 2018 April 16. doi: 10.1001/jamaoncol.2018.0509.
Rescue and recovery workers who were involved in the aftermath of the World Trade Center disaster may face a greater cancer burden than the general population, according to two studies published in JAMA Oncology.
In particular, they may be at risk of developing multiple myeloma at an earlier age.
The first study was a closed-cohort study of 14,474 employees of the Fire Department of the City of New York (FDNY) who were exposed to the World Trade Center disaster but were cancer-free as of Jan. 1, 2012. The aim was to project cancer incidence from 2012 through 2031, based on data from the FDNY World Trade Center Health Program, and compare those rates with age-, race-, and sex-specific New York cancer rates from the general population.
The modeling projected a “modestly” higher number of cancer cases in the white male subgroup of rescue and recovery workers exposed to the World Trade Center (2,714 vs. 2,596 for the general population of New York; P less than .001). Specifically, the investigators projected significantly higher case counts of prostate cancer (1,437 vs. 863), thyroid cancer (73 vs. 57), and melanoma (201 vs. 131), compared with the general population in New York, but fewer lung (237 vs. 373), colorectal (172 vs. 267), and kidney cancers (66 vs. 132) (P less than .001 for all).
“Our findings suggest that the FDNY WTC-exposed cohort may experience a greater burden of cancer than would be expected from a population with similar demographic characteristics,” wrote Rachel Zeig-Owens, DrPH, from the Montefiore Medical Center and Albert Einstein College of Medicine, both in New York, and coauthors, highlighting prostate cancer as a particular concern.
However, they also acknowledged that the elevated rates observed in people exposed to the World Trade Center disaster could be a result of increased surveillance, even though they did attempt to correct for that, and that firefighters in general might face higher risks.
“It is possible that firefighters have a higher risk of cancer than the general population owing to exposures associated with the occupation,” they wrote. However occupation could also have the opposite effect, as rescue and recovery workers tend to have lower smoking rates, which may explain the relatively low rates of certain cancers such as lung cancer, they said.
A second study examined the effect of the World Trade Center disaster on the risk of multiple myeloma and monoclonal gammopathies in exposed firefighters.
The seroprevalence study of monoclonal gammopathies of undetermined significance (MGUS) in 781 exposed firefighters revealed that the age-standardized prevalence of these was 76% higher in this population than it was in a white male reference population living in Minnesota.
In particular, the age-standardized prevalence of light-chain MGUS was more than threefold higher in exposed firefighters, compared with the reference population.
Researchers also analyzed a case series of 16 exposed white male firefighters who received a diagnosis of multiple myeloma after Sept. 11, 2001. Of the 14 patients for whom data on the monoclonal protein isotype was available, half had light-chain multiple myeloma.
“These findings are of interest due to previously observed associations between light-chain multiple myeloma and light-chain MGUS and exposure to toxins, and chronic immune stimulation,” wrote Ola Landgren, MD, PhD, from the Memorial Sloan Kettering Cancer Center and his coauthors.
Seven patients were also assessed for CD20 expression – a marker of poorer prognosis – and 71% were found to be CD20-positive, a prevalence around 3.5-fold higher than that seen in the general population.
The cohort with multiple myeloma was diagnosed on average 12 years younger than those in the general population. The authors commented that this was unlikely to be caused by lead-time bias because the time from first symptoms to clinical manifestation of the disease is usually around 1 year.
“Taken together, our results show that environmental exposure due to the WTC attacks is associated with myeloma precursor disease (MGUS and light-chain MGUS) and may be a risk factor for the development of multiple myeloma at an earlier age, particularly the light-chain subtype,” the authors wrote.
The first study was supported by the National Institute of Occupational Safety and Health; no conflicts of interest were declared.
The second study was supported by the V Foundation for Cancer Research, the Byrne Fund for the benefit of Memorial Sloan-Kettering Cancer Center, the National Cancer Institute, the Albert Einstein Cancer Center, and the National Institute for Occupational Safety and Health; no conflicts of interest were declared.
SOURCE: Zeig-Owens R et al. JAMA Oncology. 2018 April 26. doi: 10.1001/jamaoncol.2018.0504. Landgren O et al. JAMA Oncology. 2018 April 16. doi: 10.1001/jamaoncol.2018.0509.
FROM JAMA ONCOLOGY
Key clinical point: Monoclonal gammopathies and multiple myeloma may occur more often and earlier in World Trade Center rescue workers.
Major finding: Prevalence of light-chain monoclonal gammopathies is threefold higher in exposed firefighters than in a reference population of white males.
Study details: A cohort study in 14,474 employees of the Fire Department of the City of New York exposed to the Sept. 11, 2001, World Trade Center disaster, a case series of 16 exposed white male firefighters diagnosed with multiple myeloma, and a seroprevalence study of monoclonal gammopathies of undetermined significance in 781 exposed firefighters.
Disclosures: The first study was supported by the National Institute of Occupational Safety and Health; no conflicts of interest were declared. The second study was supported by the V Foundation for Cancer Research, the Byrne Fund for the benefit of Memorial Sloan-Kettering Cancer Center, the National Cancer Institute, the Albert Einstein Cancer Center, and the National Institute for Occupational Safety and Health; no conflicts of interest were declared.
Source: Zeig-Owens R et al. JAMA Oncology 2018, Apr 26. doi: 10.1001/jamaoncol.2018.0504. Landgren O et al. JAMA Oncology 2018, Apr 26. doi: 10.1001/jamaoncol.2018.0509.
Ideal sun protection practices by parents low in Toronto
according to Marcus G. Tan, MD, of the University of Toronto, and his associates.
Too much sun exposure as a child is a known risk factor for skin cancer, and “it has been estimated that approximately half of cumulative ultraviolet exposure by age 60 occurs before age 20.” So it is important that children be protected from excessive sun exposure, the investigators wrote, underscoring the reason for their study of parental sun protection by Canadian parents in Toronto, which has a multiracial population.
Parental sun protection efforts were considered ideal if they used sun protection every day; on sunny days all year round; or during the summer, using at least SPF 30, and reapplying sunscreen at least every 2 hours. Parental efforts would be considered nonideal if they did not meet any of these three criteria.
Of 183 parents, 17% used ideal sun protection for their children; 28% were in the lighter-skinned group (Fitzpatrick phototype I-III) and 5% in the darker-skinned group (Fitzpatrick phototype IV-V), The greater likelihood of using ideal sun protection among those in the lighter-skinned group was statistically significant (odds ratio, 7.4; P less than .001). As each child grew 1 year older, parents were 31% less likely to use ideal sun protection (OR, 0.69; P = .007).
Parents whose children were in the lighter-skinned group were “more likely to believe that sun exposure was harmful” for their children (OR, 17.2), and “to perceive value in sun protection,” (OR, 11.4) the researchers said. The parents whose children were in the darker-skinned group were more likely to consider sun protection as having drawbacks (OR, 9.2), and to believe that darker skin tone provides more sun protection (OR, 12.4). Neither of the two groups was more likely to consider that sun exposure provided health benefits or that it improved appearance.
Parents whose children were in the lighter-skinned group reported significantly more frequent use of sunscreen (OR, 3.6), sunglasses (OR, 2.3), and sun suits (OR, 2.7). The parents of the children in this group reported similar or higher rates of use of hats, long-sleeved clothing, and umbrellas; seeking shade; and staying indoors between 12 pm and 2 pm, but these rates were not statistically significantly different from those reported by the parents of children in the darker-skinned group, the investigators said.
“Our study suggests that more targeted education guidelines are necessary to ensure sun safety for all children, especially in a multiracial population,” said Dr. Tan and his colleagues. “Dermatologists and primary care physicians are in ideal positions to initiate personalized efforts to improve overall rates of sun protection.”
Dr. Tan reported no relevant financial disclosures. Dr. Miriam Weinstein has worked as an advisory board member for Johnson & Johnson, which manufactures sunscreen; and Dr. Shudeshna Nag consults for Valeant, which manufactures sunscreen.
SOURCE: Tan MG et al. Pediatr Dermatol. 2018 Feb 13. doi: 10.1111/pde.13433.
according to Marcus G. Tan, MD, of the University of Toronto, and his associates.
Too much sun exposure as a child is a known risk factor for skin cancer, and “it has been estimated that approximately half of cumulative ultraviolet exposure by age 60 occurs before age 20.” So it is important that children be protected from excessive sun exposure, the investigators wrote, underscoring the reason for their study of parental sun protection by Canadian parents in Toronto, which has a multiracial population.
Parental sun protection efforts were considered ideal if they used sun protection every day; on sunny days all year round; or during the summer, using at least SPF 30, and reapplying sunscreen at least every 2 hours. Parental efforts would be considered nonideal if they did not meet any of these three criteria.
Of 183 parents, 17% used ideal sun protection for their children; 28% were in the lighter-skinned group (Fitzpatrick phototype I-III) and 5% in the darker-skinned group (Fitzpatrick phototype IV-V), The greater likelihood of using ideal sun protection among those in the lighter-skinned group was statistically significant (odds ratio, 7.4; P less than .001). As each child grew 1 year older, parents were 31% less likely to use ideal sun protection (OR, 0.69; P = .007).
Parents whose children were in the lighter-skinned group were “more likely to believe that sun exposure was harmful” for their children (OR, 17.2), and “to perceive value in sun protection,” (OR, 11.4) the researchers said. The parents whose children were in the darker-skinned group were more likely to consider sun protection as having drawbacks (OR, 9.2), and to believe that darker skin tone provides more sun protection (OR, 12.4). Neither of the two groups was more likely to consider that sun exposure provided health benefits or that it improved appearance.
Parents whose children were in the lighter-skinned group reported significantly more frequent use of sunscreen (OR, 3.6), sunglasses (OR, 2.3), and sun suits (OR, 2.7). The parents of the children in this group reported similar or higher rates of use of hats, long-sleeved clothing, and umbrellas; seeking shade; and staying indoors between 12 pm and 2 pm, but these rates were not statistically significantly different from those reported by the parents of children in the darker-skinned group, the investigators said.
“Our study suggests that more targeted education guidelines are necessary to ensure sun safety for all children, especially in a multiracial population,” said Dr. Tan and his colleagues. “Dermatologists and primary care physicians are in ideal positions to initiate personalized efforts to improve overall rates of sun protection.”
Dr. Tan reported no relevant financial disclosures. Dr. Miriam Weinstein has worked as an advisory board member for Johnson & Johnson, which manufactures sunscreen; and Dr. Shudeshna Nag consults for Valeant, which manufactures sunscreen.
SOURCE: Tan MG et al. Pediatr Dermatol. 2018 Feb 13. doi: 10.1111/pde.13433.
according to Marcus G. Tan, MD, of the University of Toronto, and his associates.
Too much sun exposure as a child is a known risk factor for skin cancer, and “it has been estimated that approximately half of cumulative ultraviolet exposure by age 60 occurs before age 20.” So it is important that children be protected from excessive sun exposure, the investigators wrote, underscoring the reason for their study of parental sun protection by Canadian parents in Toronto, which has a multiracial population.
Parental sun protection efforts were considered ideal if they used sun protection every day; on sunny days all year round; or during the summer, using at least SPF 30, and reapplying sunscreen at least every 2 hours. Parental efforts would be considered nonideal if they did not meet any of these three criteria.
Of 183 parents, 17% used ideal sun protection for their children; 28% were in the lighter-skinned group (Fitzpatrick phototype I-III) and 5% in the darker-skinned group (Fitzpatrick phototype IV-V), The greater likelihood of using ideal sun protection among those in the lighter-skinned group was statistically significant (odds ratio, 7.4; P less than .001). As each child grew 1 year older, parents were 31% less likely to use ideal sun protection (OR, 0.69; P = .007).
Parents whose children were in the lighter-skinned group were “more likely to believe that sun exposure was harmful” for their children (OR, 17.2), and “to perceive value in sun protection,” (OR, 11.4) the researchers said. The parents whose children were in the darker-skinned group were more likely to consider sun protection as having drawbacks (OR, 9.2), and to believe that darker skin tone provides more sun protection (OR, 12.4). Neither of the two groups was more likely to consider that sun exposure provided health benefits or that it improved appearance.
Parents whose children were in the lighter-skinned group reported significantly more frequent use of sunscreen (OR, 3.6), sunglasses (OR, 2.3), and sun suits (OR, 2.7). The parents of the children in this group reported similar or higher rates of use of hats, long-sleeved clothing, and umbrellas; seeking shade; and staying indoors between 12 pm and 2 pm, but these rates were not statistically significantly different from those reported by the parents of children in the darker-skinned group, the investigators said.
“Our study suggests that more targeted education guidelines are necessary to ensure sun safety for all children, especially in a multiracial population,” said Dr. Tan and his colleagues. “Dermatologists and primary care physicians are in ideal positions to initiate personalized efforts to improve overall rates of sun protection.”
Dr. Tan reported no relevant financial disclosures. Dr. Miriam Weinstein has worked as an advisory board member for Johnson & Johnson, which manufactures sunscreen; and Dr. Shudeshna Nag consults for Valeant, which manufactures sunscreen.
SOURCE: Tan MG et al. Pediatr Dermatol. 2018 Feb 13. doi: 10.1111/pde.13433.
FROM PEDIATRIC DERMATOLOGY
Key clinical point: More targeted education of parents regarding sun protection practices may be needed.
Major finding: Of 183 parents, 17% used ideal sun protection; 28% were in the lighter-skinned group and 5% in the darker-skinned group (odds ratio, 7.4; P less than .001).
Study details: The parents of 183 children aged from 6 months to 6 years in Toronto completed a questionnaire about their sun protection practices.
Disclosures: Dr. Marcus G. Tan reported no relevant financial disclosures. Dr.Miriam Weinstein has worked as an advisory board member for Johnson & Johnson, which manufactures sunscreen; and Dr. Shudeshna Nag consults for Valeant, which manufactures sunscreen.
Source: Tan MG et al. Pediatr Dermatol. 2018 Feb 13. doi: 10.1111/pde.13433.
Early results favorable for combo TLR9 agonist + pembro in advanced melanoma
CHICAGO – The intratumoral Toll-Like Receptor 9 (TLR-9) agonist, CMP-001, in combination with pembrolizumab in advanced melanoma patients, was well tolerated with a durable systemic clinical response, according to early results from an ongoing phase 1 trial.
Objective response rates on weekly (n = 56) and every 3 weeks schedules (n = 13) were 23% (13%-36%) and 15% (2%-45%) respectively, reported Mohammed M. Milhem, MBBS, of the University of Iowa, Iowa City.
For those dosed weekly at low dose (less than 5 mL) and high dose (5 mL or more), the ORR was 19% (n = 43, 95% confidence interval, 8%-33%) and 27% (n = 26, 95% CI, 12%-48%), respectively. Activity was demonstrated in subjects regardless of tumor burden, Dr. Milhem said at the annual meeting of the American Association for Cancer Research.
In this phase 1b study with a 3+3 design of dose escalation and expansion, the researchers enrolled patients with advanced melanoma who did not respond or had progressed resistant on prior anti-PD-1 monotherapy or in combination. CMP-001 was injected intratumorally in combination with pembrolizumab as per label intravenously.
The study drug CMP-001 has two components, a 30-mer CpG-A DNA oligonucleotide and a nonvirulent virus-like particle (VLP). The CpG-A DNA is packaged within the VLP that protects it from degradation and also allows TLR9 receptor uptake. CpG-A DNA acts as a TLR9 agonist by binding to it, thereby activating plasmacytoid dendritic cells (pDCs) within the tumor microenvironment. The activation results in secretion of large amounts of type 1 interferon and Th1 chemokines, changing the microenvironment from a “cold/desert-like” immune suppressed state to a “hot” antitumor inflamed state, Dr. Milhem said.
“The T cells thus generated can mediate tumor rejection both in the injected and noninjected tumor,” he said. Two CMP-001 schedules were evaluated, weekly for 7 weeks or weekly for 2 weeks, followed thereafter by every 3 weeks until discontinuation (due to progression, toxicity, investigator decision, or withdrawal of consent). Scans were done every 12 weeks and tumor response was assessed by RECIST v1.1.
The CMP-001 dose escalation scheme ranged from 1 mg to 10 mg. The maximum tolerated dose was not reached and the dose of 5 mg/weekly plus pembrolizumab was used for the dose expansion phase. It was up to the investigator to increase the dose to 10 mg since maximum tolerated dose was not reached. The key inclusion criteria were metastatic or unresectable melanoma; in the dose escalation phase prior best response to anti-PD1-based therapy was disease progression or stable disease. In the dose expansion phase, patients who had progressed on anti-PD1 based therapy were allowed regardless of best response. There was no restriction on the number of prior lines of therapy.
A total of 69 subjects were treated, 44 subjects from dose escalation and 25 in the expansion phase (ongoing). Two subjects discontinued because of treatment-related adverse events. The rest of the patients had a manageable toxicity profile consisting predominantly of fever, nausea/vomiting, hypotension and rigors. Severe grade 3/4 treatment-related adverse events were reported in more than 1 subject, with hypotension (n = 9, 13%) being the most prominent AE, followed by anemia (n = 2, 3%), chills (n = 2, 3%), and hypertension (n = 2, 3%). Hypotension was manageable by responsive fluid resuscitation and in some patients required stress dose steroids. Most of these side effects occurred 1-4 hours after the CMP-001 injection.
Of the 18 responders, 1 progressed, 2 withdrew consent, and 13 remain on study with 2 subjects maintaining their response though week 72. The median duration of response was not reached. Regression of noninjected tumors occurred in cutaneous, nodal, hepatic, and splenic metastases.
“CMP-001 plus pembrolizumab induced systemic antitumor activity, and not just local efficacy since both injected and noninjected target lesions changed from baseline per RECIST,” Dr. Milhem said. Not only did the responders show a rapid reduction in target lesions from baseline, but also a durable tumor regression as usually seen with other immunotherapeutics.
Immunohistochemical analysis of tumor biopsies demonstrated increase in CD8 (greater than fivefold) and PD-L1 expression, 5 weeks after therapy in a subset of patients with pre- and posttreatment biopsies. Transcriptional analysis by RNA-seq revealed induction of T cell inflamed gene signature, notably significant upregulation of TLR, and IFN-responsive genes.
It would be interesting to further investigate how this combination therapy compares with other strategies in a similar clinical scenario, such as oncolytic virus, other TLR ligands or means of APC activation, discussant Jedd Wolchok, MD, PhD, pointed out. Understanding resistance mechanisms at an individual patient level and optimal patient selection for this combination therapy remains a challenge, he said.
Dr. Milhem had no financial relationships to disclose.
SOURCE: Milhem MD et al. AACR Annual Meeting Abstract CT144.
CHICAGO – The intratumoral Toll-Like Receptor 9 (TLR-9) agonist, CMP-001, in combination with pembrolizumab in advanced melanoma patients, was well tolerated with a durable systemic clinical response, according to early results from an ongoing phase 1 trial.
Objective response rates on weekly (n = 56) and every 3 weeks schedules (n = 13) were 23% (13%-36%) and 15% (2%-45%) respectively, reported Mohammed M. Milhem, MBBS, of the University of Iowa, Iowa City.
For those dosed weekly at low dose (less than 5 mL) and high dose (5 mL or more), the ORR was 19% (n = 43, 95% confidence interval, 8%-33%) and 27% (n = 26, 95% CI, 12%-48%), respectively. Activity was demonstrated in subjects regardless of tumor burden, Dr. Milhem said at the annual meeting of the American Association for Cancer Research.
In this phase 1b study with a 3+3 design of dose escalation and expansion, the researchers enrolled patients with advanced melanoma who did not respond or had progressed resistant on prior anti-PD-1 monotherapy or in combination. CMP-001 was injected intratumorally in combination with pembrolizumab as per label intravenously.
The study drug CMP-001 has two components, a 30-mer CpG-A DNA oligonucleotide and a nonvirulent virus-like particle (VLP). The CpG-A DNA is packaged within the VLP that protects it from degradation and also allows TLR9 receptor uptake. CpG-A DNA acts as a TLR9 agonist by binding to it, thereby activating plasmacytoid dendritic cells (pDCs) within the tumor microenvironment. The activation results in secretion of large amounts of type 1 interferon and Th1 chemokines, changing the microenvironment from a “cold/desert-like” immune suppressed state to a “hot” antitumor inflamed state, Dr. Milhem said.
“The T cells thus generated can mediate tumor rejection both in the injected and noninjected tumor,” he said. Two CMP-001 schedules were evaluated, weekly for 7 weeks or weekly for 2 weeks, followed thereafter by every 3 weeks until discontinuation (due to progression, toxicity, investigator decision, or withdrawal of consent). Scans were done every 12 weeks and tumor response was assessed by RECIST v1.1.
The CMP-001 dose escalation scheme ranged from 1 mg to 10 mg. The maximum tolerated dose was not reached and the dose of 5 mg/weekly plus pembrolizumab was used for the dose expansion phase. It was up to the investigator to increase the dose to 10 mg since maximum tolerated dose was not reached. The key inclusion criteria were metastatic or unresectable melanoma; in the dose escalation phase prior best response to anti-PD1-based therapy was disease progression or stable disease. In the dose expansion phase, patients who had progressed on anti-PD1 based therapy were allowed regardless of best response. There was no restriction on the number of prior lines of therapy.
A total of 69 subjects were treated, 44 subjects from dose escalation and 25 in the expansion phase (ongoing). Two subjects discontinued because of treatment-related adverse events. The rest of the patients had a manageable toxicity profile consisting predominantly of fever, nausea/vomiting, hypotension and rigors. Severe grade 3/4 treatment-related adverse events were reported in more than 1 subject, with hypotension (n = 9, 13%) being the most prominent AE, followed by anemia (n = 2, 3%), chills (n = 2, 3%), and hypertension (n = 2, 3%). Hypotension was manageable by responsive fluid resuscitation and in some patients required stress dose steroids. Most of these side effects occurred 1-4 hours after the CMP-001 injection.
Of the 18 responders, 1 progressed, 2 withdrew consent, and 13 remain on study with 2 subjects maintaining their response though week 72. The median duration of response was not reached. Regression of noninjected tumors occurred in cutaneous, nodal, hepatic, and splenic metastases.
“CMP-001 plus pembrolizumab induced systemic antitumor activity, and not just local efficacy since both injected and noninjected target lesions changed from baseline per RECIST,” Dr. Milhem said. Not only did the responders show a rapid reduction in target lesions from baseline, but also a durable tumor regression as usually seen with other immunotherapeutics.
Immunohistochemical analysis of tumor biopsies demonstrated increase in CD8 (greater than fivefold) and PD-L1 expression, 5 weeks after therapy in a subset of patients with pre- and posttreatment biopsies. Transcriptional analysis by RNA-seq revealed induction of T cell inflamed gene signature, notably significant upregulation of TLR, and IFN-responsive genes.
It would be interesting to further investigate how this combination therapy compares with other strategies in a similar clinical scenario, such as oncolytic virus, other TLR ligands or means of APC activation, discussant Jedd Wolchok, MD, PhD, pointed out. Understanding resistance mechanisms at an individual patient level and optimal patient selection for this combination therapy remains a challenge, he said.
Dr. Milhem had no financial relationships to disclose.
SOURCE: Milhem MD et al. AACR Annual Meeting Abstract CT144.
CHICAGO – The intratumoral Toll-Like Receptor 9 (TLR-9) agonist, CMP-001, in combination with pembrolizumab in advanced melanoma patients, was well tolerated with a durable systemic clinical response, according to early results from an ongoing phase 1 trial.
Objective response rates on weekly (n = 56) and every 3 weeks schedules (n = 13) were 23% (13%-36%) and 15% (2%-45%) respectively, reported Mohammed M. Milhem, MBBS, of the University of Iowa, Iowa City.
For those dosed weekly at low dose (less than 5 mL) and high dose (5 mL or more), the ORR was 19% (n = 43, 95% confidence interval, 8%-33%) and 27% (n = 26, 95% CI, 12%-48%), respectively. Activity was demonstrated in subjects regardless of tumor burden, Dr. Milhem said at the annual meeting of the American Association for Cancer Research.
In this phase 1b study with a 3+3 design of dose escalation and expansion, the researchers enrolled patients with advanced melanoma who did not respond or had progressed resistant on prior anti-PD-1 monotherapy or in combination. CMP-001 was injected intratumorally in combination with pembrolizumab as per label intravenously.
The study drug CMP-001 has two components, a 30-mer CpG-A DNA oligonucleotide and a nonvirulent virus-like particle (VLP). The CpG-A DNA is packaged within the VLP that protects it from degradation and also allows TLR9 receptor uptake. CpG-A DNA acts as a TLR9 agonist by binding to it, thereby activating plasmacytoid dendritic cells (pDCs) within the tumor microenvironment. The activation results in secretion of large amounts of type 1 interferon and Th1 chemokines, changing the microenvironment from a “cold/desert-like” immune suppressed state to a “hot” antitumor inflamed state, Dr. Milhem said.
“The T cells thus generated can mediate tumor rejection both in the injected and noninjected tumor,” he said. Two CMP-001 schedules were evaluated, weekly for 7 weeks or weekly for 2 weeks, followed thereafter by every 3 weeks until discontinuation (due to progression, toxicity, investigator decision, or withdrawal of consent). Scans were done every 12 weeks and tumor response was assessed by RECIST v1.1.
The CMP-001 dose escalation scheme ranged from 1 mg to 10 mg. The maximum tolerated dose was not reached and the dose of 5 mg/weekly plus pembrolizumab was used for the dose expansion phase. It was up to the investigator to increase the dose to 10 mg since maximum tolerated dose was not reached. The key inclusion criteria were metastatic or unresectable melanoma; in the dose escalation phase prior best response to anti-PD1-based therapy was disease progression or stable disease. In the dose expansion phase, patients who had progressed on anti-PD1 based therapy were allowed regardless of best response. There was no restriction on the number of prior lines of therapy.
A total of 69 subjects were treated, 44 subjects from dose escalation and 25 in the expansion phase (ongoing). Two subjects discontinued because of treatment-related adverse events. The rest of the patients had a manageable toxicity profile consisting predominantly of fever, nausea/vomiting, hypotension and rigors. Severe grade 3/4 treatment-related adverse events were reported in more than 1 subject, with hypotension (n = 9, 13%) being the most prominent AE, followed by anemia (n = 2, 3%), chills (n = 2, 3%), and hypertension (n = 2, 3%). Hypotension was manageable by responsive fluid resuscitation and in some patients required stress dose steroids. Most of these side effects occurred 1-4 hours after the CMP-001 injection.
Of the 18 responders, 1 progressed, 2 withdrew consent, and 13 remain on study with 2 subjects maintaining their response though week 72. The median duration of response was not reached. Regression of noninjected tumors occurred in cutaneous, nodal, hepatic, and splenic metastases.
“CMP-001 plus pembrolizumab induced systemic antitumor activity, and not just local efficacy since both injected and noninjected target lesions changed from baseline per RECIST,” Dr. Milhem said. Not only did the responders show a rapid reduction in target lesions from baseline, but also a durable tumor regression as usually seen with other immunotherapeutics.
Immunohistochemical analysis of tumor biopsies demonstrated increase in CD8 (greater than fivefold) and PD-L1 expression, 5 weeks after therapy in a subset of patients with pre- and posttreatment biopsies. Transcriptional analysis by RNA-seq revealed induction of T cell inflamed gene signature, notably significant upregulation of TLR, and IFN-responsive genes.
It would be interesting to further investigate how this combination therapy compares with other strategies in a similar clinical scenario, such as oncolytic virus, other TLR ligands or means of APC activation, discussant Jedd Wolchok, MD, PhD, pointed out. Understanding resistance mechanisms at an individual patient level and optimal patient selection for this combination therapy remains a challenge, he said.
Dr. Milhem had no financial relationships to disclose.
SOURCE: Milhem MD et al. AACR Annual Meeting Abstract CT144.
REPORTING FROM THE AACR ANNUAL MEETING
Key clinical point: The combination demonstrated a manageable toxicity profile with ORR of 22%.
Major finding: Objective response rates on weekly (n = 56) and every 3 weeks schedules (n = 13) were 23% (13%-36%) and 15% (2%-45%) respectively.
Study details: This phase 1b study comprised 69 patients (44 in escalation and 25 in expansion).
Disclosures: Dr. Milhem had no financial relationships to disclose.
Source: Milhem MD et al. AACR Annual Meeting. Abstract CT144.
KEYNOTE-054: Adjuvant pembrolizumab beat placebo in high-risk resected melanoma
CHICAGO – Adjuvant pembrolizumab for resected high-risk melanoma slowed the rate of recurrence or death by 43% compared with placebo in a phase 3 trial of 1,519 patients.
After 15 months of follow-up, 12-month rates of recurrence-free survival (RFS) were 75% for pembrolizumab and 61% for placebo (hazard ratio, 0.57; P less than .001), Alexander M.M. Eggermont, MD, PhD, reported at the annual meeting of the American Association for Cancer Research.
By 18 months, the RFS difference between the arms had widened even more (71% versus 53%), Dr. Eggermont and his associates said at the meeting. The report was published simultaneously in the New England Journal of Medicine.
Adjuvant pembrolizumab was effective irrespective of PD-L1 tumor expression status. In a subgroup of more than 800 patients with PD-L1-positive tumors, 12-month RFS rates were 77% for pembrolizumab and 63% for placebo (HR, 0.54; 95% CI, 0.42 to 0.69; P less than .001). Among 116 patients who were PD-L1-negative, these rates were 72% and 52%, respectively (HR, 0.47; P = .01).
Treatment produced no new safety signals, said Dr. Eggermont of Gustave Roussy Cancer Campus Grand Paris and University Paris-Saclay, Villejuif, France. Grade 3 or higher toxicities affected 15% of pembrolizumab patients. Myositis caused one pembrolizumab-related death.
The findings bolster data suggesting that adjuvant therapy can stop or delay recurrence in resected high-risk melanoma. Previously, adjuvant ipilimumab was approved after significantly extending RFS and overall survival in the placebo-controlled European Organization for Research and Treatment of Cancer 18071 trial. More recently, adjuvant dabrafenib plus trametinib reduced the risk of recurrence compared with placebo in completely resected stage III melanoma with BRAF mutations (COMBI-AD), and adjuvant nivolumab significantly improved RFS and was less toxic than was ipilimumab in patients with advanced resected BRAF-mutated and BRAF-wild-type melanomas (CheckMate 238).
Like the EORTC 18071 trial, KEYNOTE-054 (EORTC 1325) enrolled adults with completely resected stage III cutaneous melanoma. Patients with stage IIIa disease were high-risk, with sentinel node tumors exceeding 1-mm diameter per Rotterdam criteria. Stage IIIB or IIIC patients had no in-transit metastases. In all, 1,015 patients received up to 18 doses of pembrolizumab (200 mg infused every 3 weeks) or placebo for approximately 1 year. Relapsers could either repeat pembrolizumab or cross over to the pembrolizumab arm.
Treatment-related adverse events occurred in 78% of pembrolizumab patients and 66% of placebo recipients. As in prior studies, the most frequent adverse effects of pembrolizumab included fatigue or asthenia (37%), skin reactions (28%), diarrhea (19%), arthralgia (12%), nausea (11%), and dyspnea (6%). Rates of immune-related adverse events of any grade were 37% versus 9%. The most common immune-related adverse event was endocrinopathy (23%), specifically hypothyroidism (14%) and hyperthyroidism (10%). Grade 3 or higher toxicities affected 15% of pembrolizumab recipients and most often consisted of colitis (2%), endocrine disorders (1.8%), or hepatobiliary disorders (1.4%). Myositis caused the only pembrolizumab-related death.
Patients and clinicians await KEYNOTE-054 readouts on distant metastasis-free survival and overall survival. In past trials of adjuvant interferon alfa or ipilimumab for high-risk melanoma, RFS and overall survival closely correlated, Dr. Eggermont noted. KEYNOTE-54 can be expected to produce similar findings unless post-relapse therapy – including crossover to the pembrolizumab arm – narrows the survival advantage of adjuvant treatment, he added.
Merck makes pembrolizumab and funded the trial. Dr. Eggermont disclosed ties to Actelion, Agenus, Bayer, BMS, Incyte, ISA Pharmaceuticals, HalioDX, Merck-Serono, MSD, Nektar, Novartis, Pfizer, and Sanofi outside the submitted work.
SOURCE: Eggermont AMM et al. AACR Annual Meeting Abstract CT001.
CHICAGO – Adjuvant pembrolizumab for resected high-risk melanoma slowed the rate of recurrence or death by 43% compared with placebo in a phase 3 trial of 1,519 patients.
After 15 months of follow-up, 12-month rates of recurrence-free survival (RFS) were 75% for pembrolizumab and 61% for placebo (hazard ratio, 0.57; P less than .001), Alexander M.M. Eggermont, MD, PhD, reported at the annual meeting of the American Association for Cancer Research.
By 18 months, the RFS difference between the arms had widened even more (71% versus 53%), Dr. Eggermont and his associates said at the meeting. The report was published simultaneously in the New England Journal of Medicine.
Adjuvant pembrolizumab was effective irrespective of PD-L1 tumor expression status. In a subgroup of more than 800 patients with PD-L1-positive tumors, 12-month RFS rates were 77% for pembrolizumab and 63% for placebo (HR, 0.54; 95% CI, 0.42 to 0.69; P less than .001). Among 116 patients who were PD-L1-negative, these rates were 72% and 52%, respectively (HR, 0.47; P = .01).
Treatment produced no new safety signals, said Dr. Eggermont of Gustave Roussy Cancer Campus Grand Paris and University Paris-Saclay, Villejuif, France. Grade 3 or higher toxicities affected 15% of pembrolizumab patients. Myositis caused one pembrolizumab-related death.
The findings bolster data suggesting that adjuvant therapy can stop or delay recurrence in resected high-risk melanoma. Previously, adjuvant ipilimumab was approved after significantly extending RFS and overall survival in the placebo-controlled European Organization for Research and Treatment of Cancer 18071 trial. More recently, adjuvant dabrafenib plus trametinib reduced the risk of recurrence compared with placebo in completely resected stage III melanoma with BRAF mutations (COMBI-AD), and adjuvant nivolumab significantly improved RFS and was less toxic than was ipilimumab in patients with advanced resected BRAF-mutated and BRAF-wild-type melanomas (CheckMate 238).
Like the EORTC 18071 trial, KEYNOTE-054 (EORTC 1325) enrolled adults with completely resected stage III cutaneous melanoma. Patients with stage IIIa disease were high-risk, with sentinel node tumors exceeding 1-mm diameter per Rotterdam criteria. Stage IIIB or IIIC patients had no in-transit metastases. In all, 1,015 patients received up to 18 doses of pembrolizumab (200 mg infused every 3 weeks) or placebo for approximately 1 year. Relapsers could either repeat pembrolizumab or cross over to the pembrolizumab arm.
Treatment-related adverse events occurred in 78% of pembrolizumab patients and 66% of placebo recipients. As in prior studies, the most frequent adverse effects of pembrolizumab included fatigue or asthenia (37%), skin reactions (28%), diarrhea (19%), arthralgia (12%), nausea (11%), and dyspnea (6%). Rates of immune-related adverse events of any grade were 37% versus 9%. The most common immune-related adverse event was endocrinopathy (23%), specifically hypothyroidism (14%) and hyperthyroidism (10%). Grade 3 or higher toxicities affected 15% of pembrolizumab recipients and most often consisted of colitis (2%), endocrine disorders (1.8%), or hepatobiliary disorders (1.4%). Myositis caused the only pembrolizumab-related death.
Patients and clinicians await KEYNOTE-054 readouts on distant metastasis-free survival and overall survival. In past trials of adjuvant interferon alfa or ipilimumab for high-risk melanoma, RFS and overall survival closely correlated, Dr. Eggermont noted. KEYNOTE-54 can be expected to produce similar findings unless post-relapse therapy – including crossover to the pembrolizumab arm – narrows the survival advantage of adjuvant treatment, he added.
Merck makes pembrolizumab and funded the trial. Dr. Eggermont disclosed ties to Actelion, Agenus, Bayer, BMS, Incyte, ISA Pharmaceuticals, HalioDX, Merck-Serono, MSD, Nektar, Novartis, Pfizer, and Sanofi outside the submitted work.
SOURCE: Eggermont AMM et al. AACR Annual Meeting Abstract CT001.
CHICAGO – Adjuvant pembrolizumab for resected high-risk melanoma slowed the rate of recurrence or death by 43% compared with placebo in a phase 3 trial of 1,519 patients.
After 15 months of follow-up, 12-month rates of recurrence-free survival (RFS) were 75% for pembrolizumab and 61% for placebo (hazard ratio, 0.57; P less than .001), Alexander M.M. Eggermont, MD, PhD, reported at the annual meeting of the American Association for Cancer Research.
By 18 months, the RFS difference between the arms had widened even more (71% versus 53%), Dr. Eggermont and his associates said at the meeting. The report was published simultaneously in the New England Journal of Medicine.
Adjuvant pembrolizumab was effective irrespective of PD-L1 tumor expression status. In a subgroup of more than 800 patients with PD-L1-positive tumors, 12-month RFS rates were 77% for pembrolizumab and 63% for placebo (HR, 0.54; 95% CI, 0.42 to 0.69; P less than .001). Among 116 patients who were PD-L1-negative, these rates were 72% and 52%, respectively (HR, 0.47; P = .01).
Treatment produced no new safety signals, said Dr. Eggermont of Gustave Roussy Cancer Campus Grand Paris and University Paris-Saclay, Villejuif, France. Grade 3 or higher toxicities affected 15% of pembrolizumab patients. Myositis caused one pembrolizumab-related death.
The findings bolster data suggesting that adjuvant therapy can stop or delay recurrence in resected high-risk melanoma. Previously, adjuvant ipilimumab was approved after significantly extending RFS and overall survival in the placebo-controlled European Organization for Research and Treatment of Cancer 18071 trial. More recently, adjuvant dabrafenib plus trametinib reduced the risk of recurrence compared with placebo in completely resected stage III melanoma with BRAF mutations (COMBI-AD), and adjuvant nivolumab significantly improved RFS and was less toxic than was ipilimumab in patients with advanced resected BRAF-mutated and BRAF-wild-type melanomas (CheckMate 238).
Like the EORTC 18071 trial, KEYNOTE-054 (EORTC 1325) enrolled adults with completely resected stage III cutaneous melanoma. Patients with stage IIIa disease were high-risk, with sentinel node tumors exceeding 1-mm diameter per Rotterdam criteria. Stage IIIB or IIIC patients had no in-transit metastases. In all, 1,015 patients received up to 18 doses of pembrolizumab (200 mg infused every 3 weeks) or placebo for approximately 1 year. Relapsers could either repeat pembrolizumab or cross over to the pembrolizumab arm.
Treatment-related adverse events occurred in 78% of pembrolizumab patients and 66% of placebo recipients. As in prior studies, the most frequent adverse effects of pembrolizumab included fatigue or asthenia (37%), skin reactions (28%), diarrhea (19%), arthralgia (12%), nausea (11%), and dyspnea (6%). Rates of immune-related adverse events of any grade were 37% versus 9%. The most common immune-related adverse event was endocrinopathy (23%), specifically hypothyroidism (14%) and hyperthyroidism (10%). Grade 3 or higher toxicities affected 15% of pembrolizumab recipients and most often consisted of colitis (2%), endocrine disorders (1.8%), or hepatobiliary disorders (1.4%). Myositis caused the only pembrolizumab-related death.
Patients and clinicians await KEYNOTE-054 readouts on distant metastasis-free survival and overall survival. In past trials of adjuvant interferon alfa or ipilimumab for high-risk melanoma, RFS and overall survival closely correlated, Dr. Eggermont noted. KEYNOTE-54 can be expected to produce similar findings unless post-relapse therapy – including crossover to the pembrolizumab arm – narrows the survival advantage of adjuvant treatment, he added.
Merck makes pembrolizumab and funded the trial. Dr. Eggermont disclosed ties to Actelion, Agenus, Bayer, BMS, Incyte, ISA Pharmaceuticals, HalioDX, Merck-Serono, MSD, Nektar, Novartis, Pfizer, and Sanofi outside the submitted work.
SOURCE: Eggermont AMM et al. AACR Annual Meeting Abstract CT001.
REPORTING FROM THE AACR ANNUAL MEETING
Key clinical point: Adjuvant pembrolizumab (200 mg every 3 weeks) significantly extended recurrence-free survival in adults with high-risk, completely resected stage III melanoma.
Major finding: After 15 months of median follow-up, 12-month rates of recurrence-free survival were 75% for pembrolizumab and 61% for placebo (hazard ratio, 0.57; P less than .001). There was one treatment-related death in the pembrolizumab group.
Study details: KEYNOTE-054, a randomized, double-blind, phase 3 trial of 1,019 patients.
Disclosures: Merck makes pembrolizumab and funded the trial.
Source: Eggermont AMM et al. AACR Annual Meeting. Abstract CT001.
Gene strong predictor of metastasis in melanoma
CHICAGO – Investigators have identified four genes that are overexpressed in primary melanoma, including one, CXCL1, that holds promise as a strong predictor of future metastatic disease, according to study results presented at the Society of Surgical Oncology Annual Cancer Symposium.
The study implicated four genes strongly expressed in primary melanoma tumors of patients who develop distant metastases – CXCL1, CXCL2, CBL, and CD276 – said Jennifer Erdrich, MD, MPH, of Cedars Sinai Medical Center, Los Angeles. However, CXCL1 stood out. “CXCL1 overexpression is an independent predictor of developing metastatic disease. Patients with CXCL1 overexpression in the primary tumor in our study had decreased overall 5-year survival.” CXCL1 may be a useful predictive marker in primary melanoma and a potential target for immunotherapy, she said.
The rationale for analyzing the 79 genes implicated in cancer only rather than the entire array of 22,000 genes was to reduce the odds of a high false-discovery rate from 5% to 0.007%. “This is what strengthens our findings in a cohort of 37 patients,” Dr. Erdrich said.
The study analyzed pathological characteristics of the metastatic and nonmetastatic groups. Most characteristics were similar between the two groups, including location of the primary tumor in the trunk and extremities of 67% and 71%, respectively, and age of 60 years and older. The analysis noted two deviations: primary tumor size was thicker in the metastatic group (2.1 mm vs. 1.05 mm; P = .6), although Dr. Erdrich noted this was “not significantly different”; and a higher rate of ulceration in the metastatic group (50% vs. 13%; P = .05).
The genes CXCL1 and CXCL2 are both chemokines involved in growth and inflammation. “CXCL1 expression was 2.51 times greater in the metastatic group,” Dr. Erdrich said (P less than .001). Overexpression in the other three genes of interest was: CXCL2, 1.68 times greater (P less than .01); CD276, which is involved in T-cell immunity, 1.16 times greater (P = .04); and C-CBL, which is a photo-oncogene involved in the ubiquitin pathway, 1.15 times greater (P = .01). “The overexpression of all four of these was statistically significant,” she said.
Univariate analysis found ulceration of the primary along with overexpression of
the four genes to be significant predictors of metastasis. “However, in our multivariate model, three of the genes dropped out but CXCL1 remained robust,” she said.
Dr. Erdrich noted that CXCL1 is a cytokine located on chromosome 4, is secreted by macrophages, exerts its signal through CXCR2, and is one of five cytokines upregulated in lesions that respond to immunotherapy (Br J Dermatol. 2016;175:966-78).
CXCL1 compares favorably with S100, the existing blood-based biomarker for predicting recurrence in high-risk melanoma, as a predictor of metastases, Dr. Erdrich said, with an area under the curve of 0.80 versus 0.66; sensitivity of 67% versus 77%; specificity of 97% versus 61%; positive predictive value of 80% versus 40%; and negative predictive value of 94% versus 88% (Anticancer Res. 1999;19:2685-90; Cancer. 2003;97:1737-45).
The study also looked at overall survival in patients with low and high expression of CXCL1. “The patients with high expression had 5-year survival of only 50% compared to those of low expression, whose 5-year survival was 97%,” Dr. Erdrich said.
Dr. Erdrich and her coauthors reported having no financial relationships.
SOURCE: Erdrich J et al. SSO 2018, Abstract 82.
CHICAGO – Investigators have identified four genes that are overexpressed in primary melanoma, including one, CXCL1, that holds promise as a strong predictor of future metastatic disease, according to study results presented at the Society of Surgical Oncology Annual Cancer Symposium.
The study implicated four genes strongly expressed in primary melanoma tumors of patients who develop distant metastases – CXCL1, CXCL2, CBL, and CD276 – said Jennifer Erdrich, MD, MPH, of Cedars Sinai Medical Center, Los Angeles. However, CXCL1 stood out. “CXCL1 overexpression is an independent predictor of developing metastatic disease. Patients with CXCL1 overexpression in the primary tumor in our study had decreased overall 5-year survival.” CXCL1 may be a useful predictive marker in primary melanoma and a potential target for immunotherapy, she said.
The rationale for analyzing the 79 genes implicated in cancer only rather than the entire array of 22,000 genes was to reduce the odds of a high false-discovery rate from 5% to 0.007%. “This is what strengthens our findings in a cohort of 37 patients,” Dr. Erdrich said.
The study analyzed pathological characteristics of the metastatic and nonmetastatic groups. Most characteristics were similar between the two groups, including location of the primary tumor in the trunk and extremities of 67% and 71%, respectively, and age of 60 years and older. The analysis noted two deviations: primary tumor size was thicker in the metastatic group (2.1 mm vs. 1.05 mm; P = .6), although Dr. Erdrich noted this was “not significantly different”; and a higher rate of ulceration in the metastatic group (50% vs. 13%; P = .05).
The genes CXCL1 and CXCL2 are both chemokines involved in growth and inflammation. “CXCL1 expression was 2.51 times greater in the metastatic group,” Dr. Erdrich said (P less than .001). Overexpression in the other three genes of interest was: CXCL2, 1.68 times greater (P less than .01); CD276, which is involved in T-cell immunity, 1.16 times greater (P = .04); and C-CBL, which is a photo-oncogene involved in the ubiquitin pathway, 1.15 times greater (P = .01). “The overexpression of all four of these was statistically significant,” she said.
Univariate analysis found ulceration of the primary along with overexpression of
the four genes to be significant predictors of metastasis. “However, in our multivariate model, three of the genes dropped out but CXCL1 remained robust,” she said.
Dr. Erdrich noted that CXCL1 is a cytokine located on chromosome 4, is secreted by macrophages, exerts its signal through CXCR2, and is one of five cytokines upregulated in lesions that respond to immunotherapy (Br J Dermatol. 2016;175:966-78).
CXCL1 compares favorably with S100, the existing blood-based biomarker for predicting recurrence in high-risk melanoma, as a predictor of metastases, Dr. Erdrich said, with an area under the curve of 0.80 versus 0.66; sensitivity of 67% versus 77%; specificity of 97% versus 61%; positive predictive value of 80% versus 40%; and negative predictive value of 94% versus 88% (Anticancer Res. 1999;19:2685-90; Cancer. 2003;97:1737-45).
The study also looked at overall survival in patients with low and high expression of CXCL1. “The patients with high expression had 5-year survival of only 50% compared to those of low expression, whose 5-year survival was 97%,” Dr. Erdrich said.
Dr. Erdrich and her coauthors reported having no financial relationships.
SOURCE: Erdrich J et al. SSO 2018, Abstract 82.
CHICAGO – Investigators have identified four genes that are overexpressed in primary melanoma, including one, CXCL1, that holds promise as a strong predictor of future metastatic disease, according to study results presented at the Society of Surgical Oncology Annual Cancer Symposium.
The study implicated four genes strongly expressed in primary melanoma tumors of patients who develop distant metastases – CXCL1, CXCL2, CBL, and CD276 – said Jennifer Erdrich, MD, MPH, of Cedars Sinai Medical Center, Los Angeles. However, CXCL1 stood out. “CXCL1 overexpression is an independent predictor of developing metastatic disease. Patients with CXCL1 overexpression in the primary tumor in our study had decreased overall 5-year survival.” CXCL1 may be a useful predictive marker in primary melanoma and a potential target for immunotherapy, she said.
The rationale for analyzing the 79 genes implicated in cancer only rather than the entire array of 22,000 genes was to reduce the odds of a high false-discovery rate from 5% to 0.007%. “This is what strengthens our findings in a cohort of 37 patients,” Dr. Erdrich said.
The study analyzed pathological characteristics of the metastatic and nonmetastatic groups. Most characteristics were similar between the two groups, including location of the primary tumor in the trunk and extremities of 67% and 71%, respectively, and age of 60 years and older. The analysis noted two deviations: primary tumor size was thicker in the metastatic group (2.1 mm vs. 1.05 mm; P = .6), although Dr. Erdrich noted this was “not significantly different”; and a higher rate of ulceration in the metastatic group (50% vs. 13%; P = .05).
The genes CXCL1 and CXCL2 are both chemokines involved in growth and inflammation. “CXCL1 expression was 2.51 times greater in the metastatic group,” Dr. Erdrich said (P less than .001). Overexpression in the other three genes of interest was: CXCL2, 1.68 times greater (P less than .01); CD276, which is involved in T-cell immunity, 1.16 times greater (P = .04); and C-CBL, which is a photo-oncogene involved in the ubiquitin pathway, 1.15 times greater (P = .01). “The overexpression of all four of these was statistically significant,” she said.
Univariate analysis found ulceration of the primary along with overexpression of
the four genes to be significant predictors of metastasis. “However, in our multivariate model, three of the genes dropped out but CXCL1 remained robust,” she said.
Dr. Erdrich noted that CXCL1 is a cytokine located on chromosome 4, is secreted by macrophages, exerts its signal through CXCR2, and is one of five cytokines upregulated in lesions that respond to immunotherapy (Br J Dermatol. 2016;175:966-78).
CXCL1 compares favorably with S100, the existing blood-based biomarker for predicting recurrence in high-risk melanoma, as a predictor of metastases, Dr. Erdrich said, with an area under the curve of 0.80 versus 0.66; sensitivity of 67% versus 77%; specificity of 97% versus 61%; positive predictive value of 80% versus 40%; and negative predictive value of 94% versus 88% (Anticancer Res. 1999;19:2685-90; Cancer. 2003;97:1737-45).
The study also looked at overall survival in patients with low and high expression of CXCL1. “The patients with high expression had 5-year survival of only 50% compared to those of low expression, whose 5-year survival was 97%,” Dr. Erdrich said.
Dr. Erdrich and her coauthors reported having no financial relationships.
SOURCE: Erdrich J et al. SSO 2018, Abstract 82.
REPORTING FROM SSO 2018
Key clinical point: The CXCL1 gene may predict metastatic risk in primary melanoma.
Major findings: CXCL1 overexpression yielded 50% 5-year survival, almost half that of underexpression.
Study details: Gene analysis of samples from 37 patients with nonmetastatic primary melanoma who had surgical removal of primary lesion with median follow-up of 38 months.
Disclosures: Dr. Erdrich and her coauthors reported having no financial disclosures.
Source: Erdrich J et al. SSO 2018, Abstract 82.
Extracapsular spread predicts survival in SLN+ melanoma
CHICAGO – Extracapsular extension, or extracapsular spread (ECS), has been recognized as a risk factor in melanoma patients with macrometastatic (N+) disease, but a study from the United Kingdom has found it may also be an important indicator of progression-free and overall survival in patients who have sentinel node positive (SLN+) micrometastatic disease, a researcher reported at the Society of Surgical Oncology Annual Cancer Symposium.
“There is limited published data on ECS in micrometastatic disease, although there is progression-free survival data published in the literature,” Michelle Lo, MBCHB, MRCS, of Norfolk and Norwich University Hospitals, in Norwich, England, said in presenting the results. “The goal of the study was to determine the incidence of ECS in the micrometastatic group and to determine the prognostic significance of this.”
The study found that the incidence of ECS in the N+ group was significantly higher than the SLN+ group, 52.4% vs. 16.2% (P less than .0001). ECS proved to be a significant prognostic indicator of disease-specific survival and overall survival for both N+ and SLN+ disease. “There was no statistical difference in Breslow thickness between the two groups regardless of ECS,” she said.
Both the N+ and SLN+ groups with ECS had more lymph nodes than the ECS-absent subgroups, Dr. Lo said. However, in the ECS-absent subgroups, N+ patients had twice the number of lymph nodes than SLN+ patients. “This would suggest that ECS is a high-risk phenotype from the outset of metastases rather than something that’s developed over time,” she said. “Our data is in line with international staging data.”
The ECS-absent SLN– disease group had the most favorable survival outcomes, while those with ECS-present N+ disease had the worst outcomes. The prognosis of ECS-present, SLN+ patients was statistically similar to the ECS-absent, N+ group, she said.
In patients with SLN+ disease, Breslow thickness and N-stage were independent prognostic indicators for progression-free survival (hazard ratio 2.4, P less than .0001) and disease-free survival (HR 2.3, P less than .0001), Dr. Lo noted that median progression-free survival in SLN+ and N+ disease was 20 and 10 months, respectively. “Within our cohort of patients with ECS present in the micrometastatic group, their disease progressed within 3 years,” she said.
A multivariate analysis showed the survival data from this study was consistent with American Joint Committee on Cancer staging criteria, Dr. Lo said. “ECS is well recognized in the macrometastatic group, but we demonstrated from our data that the incidence of ECS in the micrometastatic group is one in six. It’s an independent risk factor for disease progression and an independent risk factor for worst disease-specific and overall survival, and it upstages micrometastatic disease.” ECS upstages stage III disease in a fashion similar to that of ulceration in primary melanoma, she said.
“In the absence of data to suggest otherwise, we would still recommend completion lymph node dissection in our micrometastatic group where ECS is present, and we would advocate that ECS should be included as an independent staging variable in the future,” Dr. Lo said.
Dr. Lo and her coauthors reported having no financial disclosures.
SOURCE: Lo M, et al. SSO 2018 Abstract 70.
CHICAGO – Extracapsular extension, or extracapsular spread (ECS), has been recognized as a risk factor in melanoma patients with macrometastatic (N+) disease, but a study from the United Kingdom has found it may also be an important indicator of progression-free and overall survival in patients who have sentinel node positive (SLN+) micrometastatic disease, a researcher reported at the Society of Surgical Oncology Annual Cancer Symposium.
“There is limited published data on ECS in micrometastatic disease, although there is progression-free survival data published in the literature,” Michelle Lo, MBCHB, MRCS, of Norfolk and Norwich University Hospitals, in Norwich, England, said in presenting the results. “The goal of the study was to determine the incidence of ECS in the micrometastatic group and to determine the prognostic significance of this.”
The study found that the incidence of ECS in the N+ group was significantly higher than the SLN+ group, 52.4% vs. 16.2% (P less than .0001). ECS proved to be a significant prognostic indicator of disease-specific survival and overall survival for both N+ and SLN+ disease. “There was no statistical difference in Breslow thickness between the two groups regardless of ECS,” she said.
Both the N+ and SLN+ groups with ECS had more lymph nodes than the ECS-absent subgroups, Dr. Lo said. However, in the ECS-absent subgroups, N+ patients had twice the number of lymph nodes than SLN+ patients. “This would suggest that ECS is a high-risk phenotype from the outset of metastases rather than something that’s developed over time,” she said. “Our data is in line with international staging data.”
The ECS-absent SLN– disease group had the most favorable survival outcomes, while those with ECS-present N+ disease had the worst outcomes. The prognosis of ECS-present, SLN+ patients was statistically similar to the ECS-absent, N+ group, she said.
In patients with SLN+ disease, Breslow thickness and N-stage were independent prognostic indicators for progression-free survival (hazard ratio 2.4, P less than .0001) and disease-free survival (HR 2.3, P less than .0001), Dr. Lo noted that median progression-free survival in SLN+ and N+ disease was 20 and 10 months, respectively. “Within our cohort of patients with ECS present in the micrometastatic group, their disease progressed within 3 years,” she said.
A multivariate analysis showed the survival data from this study was consistent with American Joint Committee on Cancer staging criteria, Dr. Lo said. “ECS is well recognized in the macrometastatic group, but we demonstrated from our data that the incidence of ECS in the micrometastatic group is one in six. It’s an independent risk factor for disease progression and an independent risk factor for worst disease-specific and overall survival, and it upstages micrometastatic disease.” ECS upstages stage III disease in a fashion similar to that of ulceration in primary melanoma, she said.
“In the absence of data to suggest otherwise, we would still recommend completion lymph node dissection in our micrometastatic group where ECS is present, and we would advocate that ECS should be included as an independent staging variable in the future,” Dr. Lo said.
Dr. Lo and her coauthors reported having no financial disclosures.
SOURCE: Lo M, et al. SSO 2018 Abstract 70.
CHICAGO – Extracapsular extension, or extracapsular spread (ECS), has been recognized as a risk factor in melanoma patients with macrometastatic (N+) disease, but a study from the United Kingdom has found it may also be an important indicator of progression-free and overall survival in patients who have sentinel node positive (SLN+) micrometastatic disease, a researcher reported at the Society of Surgical Oncology Annual Cancer Symposium.
“There is limited published data on ECS in micrometastatic disease, although there is progression-free survival data published in the literature,” Michelle Lo, MBCHB, MRCS, of Norfolk and Norwich University Hospitals, in Norwich, England, said in presenting the results. “The goal of the study was to determine the incidence of ECS in the micrometastatic group and to determine the prognostic significance of this.”
The study found that the incidence of ECS in the N+ group was significantly higher than the SLN+ group, 52.4% vs. 16.2% (P less than .0001). ECS proved to be a significant prognostic indicator of disease-specific survival and overall survival for both N+ and SLN+ disease. “There was no statistical difference in Breslow thickness between the two groups regardless of ECS,” she said.
Both the N+ and SLN+ groups with ECS had more lymph nodes than the ECS-absent subgroups, Dr. Lo said. However, in the ECS-absent subgroups, N+ patients had twice the number of lymph nodes than SLN+ patients. “This would suggest that ECS is a high-risk phenotype from the outset of metastases rather than something that’s developed over time,” she said. “Our data is in line with international staging data.”
The ECS-absent SLN– disease group had the most favorable survival outcomes, while those with ECS-present N+ disease had the worst outcomes. The prognosis of ECS-present, SLN+ patients was statistically similar to the ECS-absent, N+ group, she said.
In patients with SLN+ disease, Breslow thickness and N-stage were independent prognostic indicators for progression-free survival (hazard ratio 2.4, P less than .0001) and disease-free survival (HR 2.3, P less than .0001), Dr. Lo noted that median progression-free survival in SLN+ and N+ disease was 20 and 10 months, respectively. “Within our cohort of patients with ECS present in the micrometastatic group, their disease progressed within 3 years,” she said.
A multivariate analysis showed the survival data from this study was consistent with American Joint Committee on Cancer staging criteria, Dr. Lo said. “ECS is well recognized in the macrometastatic group, but we demonstrated from our data that the incidence of ECS in the micrometastatic group is one in six. It’s an independent risk factor for disease progression and an independent risk factor for worst disease-specific and overall survival, and it upstages micrometastatic disease.” ECS upstages stage III disease in a fashion similar to that of ulceration in primary melanoma, she said.
“In the absence of data to suggest otherwise, we would still recommend completion lymph node dissection in our micrometastatic group where ECS is present, and we would advocate that ECS should be included as an independent staging variable in the future,” Dr. Lo said.
Dr. Lo and her coauthors reported having no financial disclosures.
SOURCE: Lo M, et al. SSO 2018 Abstract 70.
REPORTING FROM SSO 2018
Key clinical point: Extracapsular spread (ECS) may be a reliable biomarker of survival in stage III melanoma.
Major finding: ECS status was an indicator of progression-free survival (hazard ratio 2.4; P less than .0001) in micrometastatic disease.
Study details: Retrospective cohort study of 515 patients who had micro- or macrometastatic lymphadenopathy at two U.K. centers from 2000 to 2017.
Disclosures: Dr. Lo and coauthors reported having no financial disclosures.
Source: Lo M et al. SSO 2018 Abstract 70.
What’s new in the latest melanoma guidelines
KAUAI, HAWAII – Melanoma , resulting in an evidence-based improved prognosis for many of them, Laura Korb Ferris, MD, PhD, said at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
Dr. Ferris, of the department of dermatology, University of Pittsburgh, highlighted some of the key changes in the eighth edition of the AJCC staging manual, which is now in effect. She also described the clinical implications of important updates introduced in the 2018 National Comprehensive Cancer Network (NCCN) guidelines for the diagnosis and management of melanoma.
The AJCC eighth edition
The eighth edition is built upon an AJCC database of more than 46,000 patients with stage I-III melanoma diagnosed since 1998 at 10 academic medical centers. The AJCC panel made no changes in stage IV melanoma guidance because the newer targeted therapies have rapidly changed treatment outcomes in that setting and longer follow-up is needed to assess the full impact.
The current edition of the AJCC melanoma staging manual creates a new subcategory within pathologic stage III. In the melanoma staging world, that’s exciting news, especially because this change has important implications for prognosis.
This fourth subcategory, stage IIID, is for melanomas, which in the Tumor, Nodes, Metastasis (TNM) classification scheme, are primary tumor stage T4b, meaning greater than 4.0 mm in thickness and with ulceration; regional lymph node N3a, b, or c, based upon the number of metastatic nodes involved and whether they were clinically occult nodal metastases detected by sentinel lymph node biopsy (SLNB) or clinically detected; and M0, meaning no distant metastatic disease. In the 8th edition, the AJCC staging system can be applied in patients with T2 through T4 primary melanoma only if they have undergone SLNB.
This new approach to stage III disease makes for more homogeneous patient subgroups, which in turn provides much better stratification of prognosis than was possible in the seventh edition of the AJCC staging manual, which dates back to 2010. Most strikingly, the 5-year melanoma-specific survival rate for patients with stage IIIA disease was 78% in the seventh edition of AJCC, but it climbs to 93% in the eighth edition. For patients with stage IIIB melanoma, 5-year melanoma-specific survival improved from 59% in the seventh edition to 83% in the current iteration, while in stage IIIC, the jump is from 40% to 69%. All this is made possible because the eighth edition separates out patients with the new stage IIID, whose 5-year melanoma-specific survival is only 32%, Dr. Ferris explained.
Among the other key points to remember about the eighth edition of AJCC:
- Tumor thickness is now measured to the nearest 0.1 mm rather than to the nearest 0.01 mm, as previously. Thus, a 0.75-mm-thick melanoma is now rounded up to 0.8 mm, while a 0.74-mm melanoma becomes a 0.7-mm tumor.
- Based upon recent evidence, tumors that are 0.8-1.0 mm thick, with or without ulceration, are now classified at T1b. So are ulcerated lesions that are less than 0.8 mm.
- Dermal mitotic rate is no longer used in staging T1 tumors, although it’s still supposed to be included in pathology reports.
- The T category definitions of primary tumors have been clarified in the eighth edition. A tumor is now classified as T0 only if there is no evidence of a primary tumor. Tx is employed when the primary tumor thickness can’t be determined, as for example when the biopsy specimen was obtained by curettage. Tis is utilized for melanoma in situ.
- The N subcategory definitions of regional nodal status have been revised. Microsatellites, clinical satellites, and in-transit metastases are now categorized as N1c, N2c, or N3c based upon the number of tumor-involved regional lymph nodes. These features are no longer defined by their size or distance from the primary tumor.
2018 NCCN melanoma guidelines
The guidelines have been revised to recommend against SLNB if a patient’s pretest probability of finding a positive SLN is less than 5%. This includes patients who have a clinical stage IA/T1a melanoma with a Breslow thickness of less than 0.8 mm without ulceration.
There is to be no SLNB in patients with microsatellites, clinical satellites, or in-transit metastases because SLN status has no prognostic significance in this situation.
Routine ordering of prognostic genetic tests for BRAF or the multigene test panels that are now commercially available is not recommended except to guide systemic therapy or to determine if a patient is a candidate for a specific clinical trial. “Basically, there is not a place to use this information in the NCCN guidelines,” according to the dermatologist.
What about completion lymphadenectomy in the SLN-positive melanoma patient?
Completion lymph node dissection looks increasingly like a procedure in search of an indication. Results of the National Cancer Institute–sponsored Multicenter Selective Lymphadenectomy Trial–II (MSLT-II) demonstrated not even a hint of a difference in 3-year melanoma-specific survival in 1,934 melanoma patients with sentinel lymph node metastases regardless of whether they were randomized to immediate completion lymph node dissection or ultrasound-based nodal monitoring. Moreover, completion lymphadenectomy was associated with significant morbidity: a 24.1% incidence of lymphedema, compared with a 6.3% rate in the observation group (N Engl J Med. 2017 Jun 8;376[23]:2211-22).
On the other hand, Dr. Ferris noted that many newer drugs are being approved for the treatment of stage III melanoma, and in all the pivotal clinical trials, patients had to have undergone completion lymph node dissection as a condition of participation. So the surgery becomes a consideration if physicians want to use the newer agents the way they were used successfully in the trials.
The full eighth edition of the AJCC cancer staging manual is available for purchase. For physicians with a specific interest in melanoma, Dr. Ferris recommended as an extremely useful alternative the AJCC expert writing panel’s free downloadable summary of the evidence-based changes made in melanoma staging (CA Cancer J Clin. 2017 Nov;67[6]:472-92). The 2018 NCCN guidelines (Melanoma. Version 1.2018 Oct. 11, 2017) are available for free (www.NCCN.org).
Dr. Ferris reported serving as a consultant to DermTech.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Melanoma , resulting in an evidence-based improved prognosis for many of them, Laura Korb Ferris, MD, PhD, said at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
Dr. Ferris, of the department of dermatology, University of Pittsburgh, highlighted some of the key changes in the eighth edition of the AJCC staging manual, which is now in effect. She also described the clinical implications of important updates introduced in the 2018 National Comprehensive Cancer Network (NCCN) guidelines for the diagnosis and management of melanoma.
The AJCC eighth edition
The eighth edition is built upon an AJCC database of more than 46,000 patients with stage I-III melanoma diagnosed since 1998 at 10 academic medical centers. The AJCC panel made no changes in stage IV melanoma guidance because the newer targeted therapies have rapidly changed treatment outcomes in that setting and longer follow-up is needed to assess the full impact.
The current edition of the AJCC melanoma staging manual creates a new subcategory within pathologic stage III. In the melanoma staging world, that’s exciting news, especially because this change has important implications for prognosis.
This fourth subcategory, stage IIID, is for melanomas, which in the Tumor, Nodes, Metastasis (TNM) classification scheme, are primary tumor stage T4b, meaning greater than 4.0 mm in thickness and with ulceration; regional lymph node N3a, b, or c, based upon the number of metastatic nodes involved and whether they were clinically occult nodal metastases detected by sentinel lymph node biopsy (SLNB) or clinically detected; and M0, meaning no distant metastatic disease. In the 8th edition, the AJCC staging system can be applied in patients with T2 through T4 primary melanoma only if they have undergone SLNB.
This new approach to stage III disease makes for more homogeneous patient subgroups, which in turn provides much better stratification of prognosis than was possible in the seventh edition of the AJCC staging manual, which dates back to 2010. Most strikingly, the 5-year melanoma-specific survival rate for patients with stage IIIA disease was 78% in the seventh edition of AJCC, but it climbs to 93% in the eighth edition. For patients with stage IIIB melanoma, 5-year melanoma-specific survival improved from 59% in the seventh edition to 83% in the current iteration, while in stage IIIC, the jump is from 40% to 69%. All this is made possible because the eighth edition separates out patients with the new stage IIID, whose 5-year melanoma-specific survival is only 32%, Dr. Ferris explained.
Among the other key points to remember about the eighth edition of AJCC:
- Tumor thickness is now measured to the nearest 0.1 mm rather than to the nearest 0.01 mm, as previously. Thus, a 0.75-mm-thick melanoma is now rounded up to 0.8 mm, while a 0.74-mm melanoma becomes a 0.7-mm tumor.
- Based upon recent evidence, tumors that are 0.8-1.0 mm thick, with or without ulceration, are now classified at T1b. So are ulcerated lesions that are less than 0.8 mm.
- Dermal mitotic rate is no longer used in staging T1 tumors, although it’s still supposed to be included in pathology reports.
- The T category definitions of primary tumors have been clarified in the eighth edition. A tumor is now classified as T0 only if there is no evidence of a primary tumor. Tx is employed when the primary tumor thickness can’t be determined, as for example when the biopsy specimen was obtained by curettage. Tis is utilized for melanoma in situ.
- The N subcategory definitions of regional nodal status have been revised. Microsatellites, clinical satellites, and in-transit metastases are now categorized as N1c, N2c, or N3c based upon the number of tumor-involved regional lymph nodes. These features are no longer defined by their size or distance from the primary tumor.
2018 NCCN melanoma guidelines
The guidelines have been revised to recommend against SLNB if a patient’s pretest probability of finding a positive SLN is less than 5%. This includes patients who have a clinical stage IA/T1a melanoma with a Breslow thickness of less than 0.8 mm without ulceration.
There is to be no SLNB in patients with microsatellites, clinical satellites, or in-transit metastases because SLN status has no prognostic significance in this situation.
Routine ordering of prognostic genetic tests for BRAF or the multigene test panels that are now commercially available is not recommended except to guide systemic therapy or to determine if a patient is a candidate for a specific clinical trial. “Basically, there is not a place to use this information in the NCCN guidelines,” according to the dermatologist.
What about completion lymphadenectomy in the SLN-positive melanoma patient?
Completion lymph node dissection looks increasingly like a procedure in search of an indication. Results of the National Cancer Institute–sponsored Multicenter Selective Lymphadenectomy Trial–II (MSLT-II) demonstrated not even a hint of a difference in 3-year melanoma-specific survival in 1,934 melanoma patients with sentinel lymph node metastases regardless of whether they were randomized to immediate completion lymph node dissection or ultrasound-based nodal monitoring. Moreover, completion lymphadenectomy was associated with significant morbidity: a 24.1% incidence of lymphedema, compared with a 6.3% rate in the observation group (N Engl J Med. 2017 Jun 8;376[23]:2211-22).
On the other hand, Dr. Ferris noted that many newer drugs are being approved for the treatment of stage III melanoma, and in all the pivotal clinical trials, patients had to have undergone completion lymph node dissection as a condition of participation. So the surgery becomes a consideration if physicians want to use the newer agents the way they were used successfully in the trials.
The full eighth edition of the AJCC cancer staging manual is available for purchase. For physicians with a specific interest in melanoma, Dr. Ferris recommended as an extremely useful alternative the AJCC expert writing panel’s free downloadable summary of the evidence-based changes made in melanoma staging (CA Cancer J Clin. 2017 Nov;67[6]:472-92). The 2018 NCCN guidelines (Melanoma. Version 1.2018 Oct. 11, 2017) are available for free (www.NCCN.org).
Dr. Ferris reported serving as a consultant to DermTech.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
KAUAI, HAWAII – Melanoma , resulting in an evidence-based improved prognosis for many of them, Laura Korb Ferris, MD, PhD, said at the Hawaii Dermatology Seminar provided by Skin Disease Education Foundation/Global Academy for Medical Education.
Dr. Ferris, of the department of dermatology, University of Pittsburgh, highlighted some of the key changes in the eighth edition of the AJCC staging manual, which is now in effect. She also described the clinical implications of important updates introduced in the 2018 National Comprehensive Cancer Network (NCCN) guidelines for the diagnosis and management of melanoma.
The AJCC eighth edition
The eighth edition is built upon an AJCC database of more than 46,000 patients with stage I-III melanoma diagnosed since 1998 at 10 academic medical centers. The AJCC panel made no changes in stage IV melanoma guidance because the newer targeted therapies have rapidly changed treatment outcomes in that setting and longer follow-up is needed to assess the full impact.
The current edition of the AJCC melanoma staging manual creates a new subcategory within pathologic stage III. In the melanoma staging world, that’s exciting news, especially because this change has important implications for prognosis.
This fourth subcategory, stage IIID, is for melanomas, which in the Tumor, Nodes, Metastasis (TNM) classification scheme, are primary tumor stage T4b, meaning greater than 4.0 mm in thickness and with ulceration; regional lymph node N3a, b, or c, based upon the number of metastatic nodes involved and whether they were clinically occult nodal metastases detected by sentinel lymph node biopsy (SLNB) or clinically detected; and M0, meaning no distant metastatic disease. In the 8th edition, the AJCC staging system can be applied in patients with T2 through T4 primary melanoma only if they have undergone SLNB.
This new approach to stage III disease makes for more homogeneous patient subgroups, which in turn provides much better stratification of prognosis than was possible in the seventh edition of the AJCC staging manual, which dates back to 2010. Most strikingly, the 5-year melanoma-specific survival rate for patients with stage IIIA disease was 78% in the seventh edition of AJCC, but it climbs to 93% in the eighth edition. For patients with stage IIIB melanoma, 5-year melanoma-specific survival improved from 59% in the seventh edition to 83% in the current iteration, while in stage IIIC, the jump is from 40% to 69%. All this is made possible because the eighth edition separates out patients with the new stage IIID, whose 5-year melanoma-specific survival is only 32%, Dr. Ferris explained.
Among the other key points to remember about the eighth edition of AJCC:
- Tumor thickness is now measured to the nearest 0.1 mm rather than to the nearest 0.01 mm, as previously. Thus, a 0.75-mm-thick melanoma is now rounded up to 0.8 mm, while a 0.74-mm melanoma becomes a 0.7-mm tumor.
- Based upon recent evidence, tumors that are 0.8-1.0 mm thick, with or without ulceration, are now classified at T1b. So are ulcerated lesions that are less than 0.8 mm.
- Dermal mitotic rate is no longer used in staging T1 tumors, although it’s still supposed to be included in pathology reports.
- The T category definitions of primary tumors have been clarified in the eighth edition. A tumor is now classified as T0 only if there is no evidence of a primary tumor. Tx is employed when the primary tumor thickness can’t be determined, as for example when the biopsy specimen was obtained by curettage. Tis is utilized for melanoma in situ.
- The N subcategory definitions of regional nodal status have been revised. Microsatellites, clinical satellites, and in-transit metastases are now categorized as N1c, N2c, or N3c based upon the number of tumor-involved regional lymph nodes. These features are no longer defined by their size or distance from the primary tumor.
2018 NCCN melanoma guidelines
The guidelines have been revised to recommend against SLNB if a patient’s pretest probability of finding a positive SLN is less than 5%. This includes patients who have a clinical stage IA/T1a melanoma with a Breslow thickness of less than 0.8 mm without ulceration.
There is to be no SLNB in patients with microsatellites, clinical satellites, or in-transit metastases because SLN status has no prognostic significance in this situation.
Routine ordering of prognostic genetic tests for BRAF or the multigene test panels that are now commercially available is not recommended except to guide systemic therapy or to determine if a patient is a candidate for a specific clinical trial. “Basically, there is not a place to use this information in the NCCN guidelines,” according to the dermatologist.
What about completion lymphadenectomy in the SLN-positive melanoma patient?
Completion lymph node dissection looks increasingly like a procedure in search of an indication. Results of the National Cancer Institute–sponsored Multicenter Selective Lymphadenectomy Trial–II (MSLT-II) demonstrated not even a hint of a difference in 3-year melanoma-specific survival in 1,934 melanoma patients with sentinel lymph node metastases regardless of whether they were randomized to immediate completion lymph node dissection or ultrasound-based nodal monitoring. Moreover, completion lymphadenectomy was associated with significant morbidity: a 24.1% incidence of lymphedema, compared with a 6.3% rate in the observation group (N Engl J Med. 2017 Jun 8;376[23]:2211-22).
On the other hand, Dr. Ferris noted that many newer drugs are being approved for the treatment of stage III melanoma, and in all the pivotal clinical trials, patients had to have undergone completion lymph node dissection as a condition of participation. So the surgery becomes a consideration if physicians want to use the newer agents the way they were used successfully in the trials.
The full eighth edition of the AJCC cancer staging manual is available for purchase. For physicians with a specific interest in melanoma, Dr. Ferris recommended as an extremely useful alternative the AJCC expert writing panel’s free downloadable summary of the evidence-based changes made in melanoma staging (CA Cancer J Clin. 2017 Nov;67[6]:472-92). The 2018 NCCN guidelines (Melanoma. Version 1.2018 Oct. 11, 2017) are available for free (www.NCCN.org).
Dr. Ferris reported serving as a consultant to DermTech.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Surgery after immunotherapy effective in advanced melanoma
CHICAGO – Surgical resection is an effective treatment in selected patients with advanced melanoma treated with checkpoint blockade immunotherapy, according to a study of an institutional database at Memorial Sloan Kettering Cancer Center in New York presented at the Society of Surgical Oncology Annual Cancer Symposium.
“In the era of improved systemic therapy, checkpoint blockade for metastatic melanoma and the ability to surgically resect all disease after treatment is associated with an estimated survival of 75%, better than what’s been previously reported,” said Danielle M. Bello, MD, of Memorial Sloan Kettering.
The study analyzed a cohort of 237 patients who had unresectable stage III and IV melanoma and were treated with checkpoint blockade, including CTLA-4, programmed cell death protein 1 (PD-1), and programmed death-ligand 1 inhibitors, and then had surgical resection during 2003-2017.
Dr. Bello noted two previous studies that had reported encouraging outcomes in advanced melanoma. The first highlighted the role for surgery in stage IV melanoma. In that phase 3 clinical trial, patients had resection of up to five sites of metastatic disease and were then randomized to one of two treatment arms: bacillus Calmette-Guérin and allogeneic whole-cell vaccine (Canvaxin) or bacillus Calmette-Guérin and placebo. While this trial found no difference in overall survival between groups, it did report a 5-year overall survival exceeding 40% in both treatment arms, which highlighted that Stage IV patients who underwent resection of all their disease had survival outcomes superior to outcomes previously reported (Ann Surg Onc. 2017 Dec;24[13]:3991-4000). The second trial, the recent Checkmate 067 trial, emphasized the role of effective systemic checkpoint blockade in advanced stage III and IV melanoma. It reported that patients treated with combined nivolumab/ipilimumab therapy had not reached median overall survival at minimum 36 months of follow-up (N Engl J Med. 2017 Oct 5;377[14]:1345-56).
“We know that checkpoint inhibitor therapy has revolutionized the landscape of unresectable stage III and IV melanoma,” Dr. Bello said. However, despite encouraging trial readouts of overall survival, progression-free survival is a different story. “We know that the median progression-free survival even in our best combination therapy is 11.5 months, meaning that 50% of patients will go on to progress in a year and many will go on to surgical resection of their disease and do quite well,” she said.
Dr. Bello and her coauthors set out to describe outcomes of a “highly selective group” of patients who had surgical resection after checkpoint inhibitor therapy. “The majority of patients in our study had a cutaneous primary melanoma,” she said. Median age was 63 years, and 88% had stage IV disease. Regarding checkpoint blockade regimen, 62% received anti–CTLA-4, and 29% received combination anti–PD-1 and anti–CLTA-4 either sequentially or concomitantly prior to resection.
The median time from the start of immunotherapy to the first operation was 7 months. Forty-six percent had no further postoperative treatment after resection. In those, who did require further treatment, the majority received anti–PD-1 followed by targeted BRAF/MEK therapy, she said.
The analysis stratified patients into the following three categories based on radiological response to immunotherapy:
- Overall response to checkpoint blockade and the index lesion was either smaller since initiation of therapy or stabilized (12; 5.1%). Half of this group had a pathological complete response.
- Isolated site of progressive disease with residual stable disease elsewhere or as the only site of progressive disease (106; 44.7%).
- Multiple sites of progressive and palliative operations (119; 50.2%).
Median overall survival was 21 months in the entire study cohort with a median follow-up of 23 months, Dr. Bello said. “Those resected to no evidence of disease (NED) – 87 patients – had an estimated 5-year overall survival of 75%.” The NED group did not reach median OS.
The analysis also stratified overall survival by response to immunotherapy. “Patients with responding or stable disease had an estimated 90% 5-year overall survival,” Dr. Bellow said. “Those with one isolated progressive lesion that was resected had a 60% 5-year overall survival.” A more detailed analysis of the latter group found that those who had a resection to NED had an improved overall survival of 75% at 5 years. Resected patients who had residual remaining disease had a 30% 5-year overall survival.
“Further follow-up is needed to assess the durability and contributions of surgery, and further studies are underway to identify biomarkers associated with improved survival after immunotherapy and surgery,” Dr. Bello said.
SOURCE: Bello DM et al. SSO 2018, Abstract 5.
CHICAGO – Surgical resection is an effective treatment in selected patients with advanced melanoma treated with checkpoint blockade immunotherapy, according to a study of an institutional database at Memorial Sloan Kettering Cancer Center in New York presented at the Society of Surgical Oncology Annual Cancer Symposium.
“In the era of improved systemic therapy, checkpoint blockade for metastatic melanoma and the ability to surgically resect all disease after treatment is associated with an estimated survival of 75%, better than what’s been previously reported,” said Danielle M. Bello, MD, of Memorial Sloan Kettering.
The study analyzed a cohort of 237 patients who had unresectable stage III and IV melanoma and were treated with checkpoint blockade, including CTLA-4, programmed cell death protein 1 (PD-1), and programmed death-ligand 1 inhibitors, and then had surgical resection during 2003-2017.
Dr. Bello noted two previous studies that had reported encouraging outcomes in advanced melanoma. The first highlighted the role for surgery in stage IV melanoma. In that phase 3 clinical trial, patients had resection of up to five sites of metastatic disease and were then randomized to one of two treatment arms: bacillus Calmette-Guérin and allogeneic whole-cell vaccine (Canvaxin) or bacillus Calmette-Guérin and placebo. While this trial found no difference in overall survival between groups, it did report a 5-year overall survival exceeding 40% in both treatment arms, which highlighted that Stage IV patients who underwent resection of all their disease had survival outcomes superior to outcomes previously reported (Ann Surg Onc. 2017 Dec;24[13]:3991-4000). The second trial, the recent Checkmate 067 trial, emphasized the role of effective systemic checkpoint blockade in advanced stage III and IV melanoma. It reported that patients treated with combined nivolumab/ipilimumab therapy had not reached median overall survival at minimum 36 months of follow-up (N Engl J Med. 2017 Oct 5;377[14]:1345-56).
“We know that checkpoint inhibitor therapy has revolutionized the landscape of unresectable stage III and IV melanoma,” Dr. Bello said. However, despite encouraging trial readouts of overall survival, progression-free survival is a different story. “We know that the median progression-free survival even in our best combination therapy is 11.5 months, meaning that 50% of patients will go on to progress in a year and many will go on to surgical resection of their disease and do quite well,” she said.
Dr. Bello and her coauthors set out to describe outcomes of a “highly selective group” of patients who had surgical resection after checkpoint inhibitor therapy. “The majority of patients in our study had a cutaneous primary melanoma,” she said. Median age was 63 years, and 88% had stage IV disease. Regarding checkpoint blockade regimen, 62% received anti–CTLA-4, and 29% received combination anti–PD-1 and anti–CLTA-4 either sequentially or concomitantly prior to resection.
The median time from the start of immunotherapy to the first operation was 7 months. Forty-six percent had no further postoperative treatment after resection. In those, who did require further treatment, the majority received anti–PD-1 followed by targeted BRAF/MEK therapy, she said.
The analysis stratified patients into the following three categories based on radiological response to immunotherapy:
- Overall response to checkpoint blockade and the index lesion was either smaller since initiation of therapy or stabilized (12; 5.1%). Half of this group had a pathological complete response.
- Isolated site of progressive disease with residual stable disease elsewhere or as the only site of progressive disease (106; 44.7%).
- Multiple sites of progressive and palliative operations (119; 50.2%).
Median overall survival was 21 months in the entire study cohort with a median follow-up of 23 months, Dr. Bello said. “Those resected to no evidence of disease (NED) – 87 patients – had an estimated 5-year overall survival of 75%.” The NED group did not reach median OS.
The analysis also stratified overall survival by response to immunotherapy. “Patients with responding or stable disease had an estimated 90% 5-year overall survival,” Dr. Bellow said. “Those with one isolated progressive lesion that was resected had a 60% 5-year overall survival.” A more detailed analysis of the latter group found that those who had a resection to NED had an improved overall survival of 75% at 5 years. Resected patients who had residual remaining disease had a 30% 5-year overall survival.
“Further follow-up is needed to assess the durability and contributions of surgery, and further studies are underway to identify biomarkers associated with improved survival after immunotherapy and surgery,” Dr. Bello said.
SOURCE: Bello DM et al. SSO 2018, Abstract 5.
CHICAGO – Surgical resection is an effective treatment in selected patients with advanced melanoma treated with checkpoint blockade immunotherapy, according to a study of an institutional database at Memorial Sloan Kettering Cancer Center in New York presented at the Society of Surgical Oncology Annual Cancer Symposium.
“In the era of improved systemic therapy, checkpoint blockade for metastatic melanoma and the ability to surgically resect all disease after treatment is associated with an estimated survival of 75%, better than what’s been previously reported,” said Danielle M. Bello, MD, of Memorial Sloan Kettering.
The study analyzed a cohort of 237 patients who had unresectable stage III and IV melanoma and were treated with checkpoint blockade, including CTLA-4, programmed cell death protein 1 (PD-1), and programmed death-ligand 1 inhibitors, and then had surgical resection during 2003-2017.
Dr. Bello noted two previous studies that had reported encouraging outcomes in advanced melanoma. The first highlighted the role for surgery in stage IV melanoma. In that phase 3 clinical trial, patients had resection of up to five sites of metastatic disease and were then randomized to one of two treatment arms: bacillus Calmette-Guérin and allogeneic whole-cell vaccine (Canvaxin) or bacillus Calmette-Guérin and placebo. While this trial found no difference in overall survival between groups, it did report a 5-year overall survival exceeding 40% in both treatment arms, which highlighted that Stage IV patients who underwent resection of all their disease had survival outcomes superior to outcomes previously reported (Ann Surg Onc. 2017 Dec;24[13]:3991-4000). The second trial, the recent Checkmate 067 trial, emphasized the role of effective systemic checkpoint blockade in advanced stage III and IV melanoma. It reported that patients treated with combined nivolumab/ipilimumab therapy had not reached median overall survival at minimum 36 months of follow-up (N Engl J Med. 2017 Oct 5;377[14]:1345-56).
“We know that checkpoint inhibitor therapy has revolutionized the landscape of unresectable stage III and IV melanoma,” Dr. Bello said. However, despite encouraging trial readouts of overall survival, progression-free survival is a different story. “We know that the median progression-free survival even in our best combination therapy is 11.5 months, meaning that 50% of patients will go on to progress in a year and many will go on to surgical resection of their disease and do quite well,” she said.
Dr. Bello and her coauthors set out to describe outcomes of a “highly selective group” of patients who had surgical resection after checkpoint inhibitor therapy. “The majority of patients in our study had a cutaneous primary melanoma,” she said. Median age was 63 years, and 88% had stage IV disease. Regarding checkpoint blockade regimen, 62% received anti–CTLA-4, and 29% received combination anti–PD-1 and anti–CLTA-4 either sequentially or concomitantly prior to resection.
The median time from the start of immunotherapy to the first operation was 7 months. Forty-six percent had no further postoperative treatment after resection. In those, who did require further treatment, the majority received anti–PD-1 followed by targeted BRAF/MEK therapy, she said.
The analysis stratified patients into the following three categories based on radiological response to immunotherapy:
- Overall response to checkpoint blockade and the index lesion was either smaller since initiation of therapy or stabilized (12; 5.1%). Half of this group had a pathological complete response.
- Isolated site of progressive disease with residual stable disease elsewhere or as the only site of progressive disease (106; 44.7%).
- Multiple sites of progressive and palliative operations (119; 50.2%).
Median overall survival was 21 months in the entire study cohort with a median follow-up of 23 months, Dr. Bello said. “Those resected to no evidence of disease (NED) – 87 patients – had an estimated 5-year overall survival of 75%.” The NED group did not reach median OS.
The analysis also stratified overall survival by response to immunotherapy. “Patients with responding or stable disease had an estimated 90% 5-year overall survival,” Dr. Bellow said. “Those with one isolated progressive lesion that was resected had a 60% 5-year overall survival.” A more detailed analysis of the latter group found that those who had a resection to NED had an improved overall survival of 75% at 5 years. Resected patients who had residual remaining disease had a 30% 5-year overall survival.
“Further follow-up is needed to assess the durability and contributions of surgery, and further studies are underway to identify biomarkers associated with improved survival after immunotherapy and surgery,” Dr. Bello said.
SOURCE: Bello DM et al. SSO 2018, Abstract 5.
REPORTING FROM SSO 2018
Key clinical point: Surgery after immunotherapy can achieve good outcomes in advanced melanoma.
Major findings: Complete resection achieved an estimated 5-year overall survival of 75%.
Study details: Analysis of a cohort of 237 patients from a prospectively maintained institutional melanoma database who had surgery after immunotherapy for unresectable stage III and IV melanoma during 2003-2017.
Disclosures: Dr. Bello reported having no financial disclosures. Some coauthors reported financial relationships with various pharmaceutical companies.
Source: Bello DM et al. SSO 2018, Abstract 5.
Tanning is the new tobacco
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@mdedge.com.
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@mdedge.com.
I was driving to work the other day, perched up in my pickup truck (somehow you knew that) and noticed a fancy race car in front of me with a vanity tag. It read HRTATTK 4. Well, I thought after four heart attacks maybe I would splurge on a special car too (more likely a newer truck). Then I noticed smoke coming out of the driver’s window, and I could see this guy in his side view mirror, presumably Mr. “Heart Attack 4,” puffing away on a cigarette. Wow.
Then I got to work and saw my secretary, who works with her oxygen on, out back puffing a cigarette. Wow.
It turns out that cigarette smoke contains substances that act as a monoamine oxidase (MAO) A inhibitor, prolonging the dopamine high in the brain (Proc Natl Acad Sci U S A. 1996 Nov 26;93[24]:14065-9). Makes sense and may explain the above smoking behavior. I truly believe cigarettes are as or more addictive than any other dopamine enhancing drug.
More than 50 years ago, a national campaign against smoking was launched after the 1964 Surgeon General’s report concluded that smoking was a major health hazard. (Looking back, one of the few losses of not having to pull journal articles from the stacks in the library, is that medical students and residents can’t shake their heads in wonder at the cigarette ads in old medical journals.) The impact of the national antismoking campaign has been dramatic, but smoking remains the leading preventable cause of death in the United States and globally, according to the Centers for Disease Control and Prevention.
in 2006, from 1992 (Arch Dermatol. 2010;146[3]:283-7), dermatologists had good footing on which to start a major prevention campaign. The American Cancer Society got on board, and in 2014, acting surgeon general Boris Lushniak, MD, issued a call to action to prevent skin cancer along with Howard Koh, MD, the assistant secretary of health, in “The Surgeon General’s Call to Action to Prevent Skin Cancer” in 2014, and the campaign was on.
Well, I am delighted to pass on a report from Leonard Lichtenfeld, MD, deputy chief medical officer for the American Cancer Society, who recently described in his March 2018 blog what may the first signs of the effectiveness of efforts to promote protection from ultraviolet ray exposure (JAMA Dermatol. 2018;154[3]:361-2). He writes: “In young white women ages 15 to 24, the incidence of melanoma has declined an average of 5.5% per year from January 2005 through December 2014. Not 5.5% over those ten years but 5.5 % PER YEAR. That’s remarkable, to say the least.”
As for the reasons behind these trends, he says, “no one can say for certain,” but he refers to national data indicating that indoor tanning has decreased in the past few years, especially among adolescents and young adults.
Dr. Coldiron is in private practice but maintains a clinical assistant professorship at the University of Cincinnati. He cares for patients, teaches medical students and residents, and has several active clinical research projects. Dr. Coldiron is the author of more than 80 scientific letters, papers, and several book chapters, and he speaks frequently on a variety of topics. He is a past president of the American Academy of Dermatology. Write to him at dermnews@mdedge.com.
Diabetes from checkpoint inhibitors probably means lifelong insulin
CHICAGO – , according to Priyanka Iyer, MD, an endocrinology fellow at MD Anderson Cancer Center, Houston.
“As long as we get glycemic control, they can continue,” she said at the annual meeting of the Endocrine Society.
Diabetes is a known side effect of immune checkpoint inhibitors (ICIs) but it’s rare, occurring in maybe 0.17% of patients, and its natural history and risk factors are unknown.
ICIs are fairly new agents, and as their use expands beyond clinical trials, “we anticipate seeing larger numbers of cases. Patients should be educated about the symptoms of uncontrolled blood sugars while on ICIs,” and endocrinologists “have to get involved and recognize this entity sooner,” Dr. Iyer said.
In short, her team found that ICI-mediated diabetes can occur in patients with or without preexisting diabetes, and that most patients have evidence of beta-cell failure, likely T-cell mediated destruction due to immune activation. In all but one case, patients remained on insulin at a median follow-up of 44 weeks, even after stopping ICIs. For most, ICI-mediated diabetes likely means lifelong insulin.
They were all on the programmed cell death protein (PD-1) inhibitors nivolumab (Opdivo) or pembrolizumab (Keytruda), or the PD-1 ligand (PD-L) inhibitor durvalumab (Imfinzi). The agents are used for a range of cancers, including renal cell, melanoma, and Hodgkin lymphoma. There were no diabetes cases in patients on single-agent ipilimumab (Yervoy) or tremelimumab, which target cytotoxic T-lymphocyte associated antigen-4 and are used for melanoma and mesothelioma.
Median time to diabetes presentation after the start of ICI treatment was 12.3 weeks but ranged from 1 to 67.2 weeks. Half of the cases presented in diabetic ketoacidosis (DKA). Patients had upward trending hyperglycemia and most had diabetes symptoms for a while before diagnosis. They presented with a blood glucose above 250 mg/dL, and more than half above 500 mg/dL. Median hemoglobin A1c at presentation was 8%, but ranged up to 12.5%.
Every patient required insulin, including the six that discontinued ICIs after developing diabetes. Diabetes resolved in just one patient at 10.2 months; she presented with DKA.
There were no obvious predisposing factors. None of the patients had histories of type 1 diabetes or other autoimmune disease. Five patients had well-controlled type 2 diabetes prior to ICI initiation; four had prediabetes. Some had family members with type 2 diabetes, but not type 1. Four had prior ICI exposure. Just three patients were on concomitant steroids.
A few patients also developed thyroid or pituitary dysfunction, which are more common side effects of ICIs.
The median age at diabetes presentation was 61 years and ranged from 32 to 82 years. The majority of patients were men, which reflects MD Anderson demographics, not a predisposing risk factor, Dr. Iyer said.
Melanoma was the most common cancer, followed by renal cell and prostate; patients had stage 2-4 disease. About half the subjects were on single agent anti-PD-1 treatment, about a third on anti-PD-1 combination treatment, and the rest on anti-PD-L1 combination therapy. C-peptide levels were below 0.9 ng/mL at diabetes diagnosis in most of the patients. Eleven of the 20 tested (55%) were positive for the pancreatic islet cell antibody GAD65.
The investigators had no disclosures. A funding source was not reported.
SOURCE: Iyer PC et al. Abstract OR05-5.
CHICAGO – , according to Priyanka Iyer, MD, an endocrinology fellow at MD Anderson Cancer Center, Houston.
“As long as we get glycemic control, they can continue,” she said at the annual meeting of the Endocrine Society.
Diabetes is a known side effect of immune checkpoint inhibitors (ICIs) but it’s rare, occurring in maybe 0.17% of patients, and its natural history and risk factors are unknown.
ICIs are fairly new agents, and as their use expands beyond clinical trials, “we anticipate seeing larger numbers of cases. Patients should be educated about the symptoms of uncontrolled blood sugars while on ICIs,” and endocrinologists “have to get involved and recognize this entity sooner,” Dr. Iyer said.
In short, her team found that ICI-mediated diabetes can occur in patients with or without preexisting diabetes, and that most patients have evidence of beta-cell failure, likely T-cell mediated destruction due to immune activation. In all but one case, patients remained on insulin at a median follow-up of 44 weeks, even after stopping ICIs. For most, ICI-mediated diabetes likely means lifelong insulin.
They were all on the programmed cell death protein (PD-1) inhibitors nivolumab (Opdivo) or pembrolizumab (Keytruda), or the PD-1 ligand (PD-L) inhibitor durvalumab (Imfinzi). The agents are used for a range of cancers, including renal cell, melanoma, and Hodgkin lymphoma. There were no diabetes cases in patients on single-agent ipilimumab (Yervoy) or tremelimumab, which target cytotoxic T-lymphocyte associated antigen-4 and are used for melanoma and mesothelioma.
Median time to diabetes presentation after the start of ICI treatment was 12.3 weeks but ranged from 1 to 67.2 weeks. Half of the cases presented in diabetic ketoacidosis (DKA). Patients had upward trending hyperglycemia and most had diabetes symptoms for a while before diagnosis. They presented with a blood glucose above 250 mg/dL, and more than half above 500 mg/dL. Median hemoglobin A1c at presentation was 8%, but ranged up to 12.5%.
Every patient required insulin, including the six that discontinued ICIs after developing diabetes. Diabetes resolved in just one patient at 10.2 months; she presented with DKA.
There were no obvious predisposing factors. None of the patients had histories of type 1 diabetes or other autoimmune disease. Five patients had well-controlled type 2 diabetes prior to ICI initiation; four had prediabetes. Some had family members with type 2 diabetes, but not type 1. Four had prior ICI exposure. Just three patients were on concomitant steroids.
A few patients also developed thyroid or pituitary dysfunction, which are more common side effects of ICIs.
The median age at diabetes presentation was 61 years and ranged from 32 to 82 years. The majority of patients were men, which reflects MD Anderson demographics, not a predisposing risk factor, Dr. Iyer said.
Melanoma was the most common cancer, followed by renal cell and prostate; patients had stage 2-4 disease. About half the subjects were on single agent anti-PD-1 treatment, about a third on anti-PD-1 combination treatment, and the rest on anti-PD-L1 combination therapy. C-peptide levels were below 0.9 ng/mL at diabetes diagnosis in most of the patients. Eleven of the 20 tested (55%) were positive for the pancreatic islet cell antibody GAD65.
The investigators had no disclosures. A funding source was not reported.
SOURCE: Iyer PC et al. Abstract OR05-5.
CHICAGO – , according to Priyanka Iyer, MD, an endocrinology fellow at MD Anderson Cancer Center, Houston.
“As long as we get glycemic control, they can continue,” she said at the annual meeting of the Endocrine Society.
Diabetes is a known side effect of immune checkpoint inhibitors (ICIs) but it’s rare, occurring in maybe 0.17% of patients, and its natural history and risk factors are unknown.
ICIs are fairly new agents, and as their use expands beyond clinical trials, “we anticipate seeing larger numbers of cases. Patients should be educated about the symptoms of uncontrolled blood sugars while on ICIs,” and endocrinologists “have to get involved and recognize this entity sooner,” Dr. Iyer said.
In short, her team found that ICI-mediated diabetes can occur in patients with or without preexisting diabetes, and that most patients have evidence of beta-cell failure, likely T-cell mediated destruction due to immune activation. In all but one case, patients remained on insulin at a median follow-up of 44 weeks, even after stopping ICIs. For most, ICI-mediated diabetes likely means lifelong insulin.
They were all on the programmed cell death protein (PD-1) inhibitors nivolumab (Opdivo) or pembrolizumab (Keytruda), or the PD-1 ligand (PD-L) inhibitor durvalumab (Imfinzi). The agents are used for a range of cancers, including renal cell, melanoma, and Hodgkin lymphoma. There were no diabetes cases in patients on single-agent ipilimumab (Yervoy) or tremelimumab, which target cytotoxic T-lymphocyte associated antigen-4 and are used for melanoma and mesothelioma.
Median time to diabetes presentation after the start of ICI treatment was 12.3 weeks but ranged from 1 to 67.2 weeks. Half of the cases presented in diabetic ketoacidosis (DKA). Patients had upward trending hyperglycemia and most had diabetes symptoms for a while before diagnosis. They presented with a blood glucose above 250 mg/dL, and more than half above 500 mg/dL. Median hemoglobin A1c at presentation was 8%, but ranged up to 12.5%.
Every patient required insulin, including the six that discontinued ICIs after developing diabetes. Diabetes resolved in just one patient at 10.2 months; she presented with DKA.
There were no obvious predisposing factors. None of the patients had histories of type 1 diabetes or other autoimmune disease. Five patients had well-controlled type 2 diabetes prior to ICI initiation; four had prediabetes. Some had family members with type 2 diabetes, but not type 1. Four had prior ICI exposure. Just three patients were on concomitant steroids.
A few patients also developed thyroid or pituitary dysfunction, which are more common side effects of ICIs.
The median age at diabetes presentation was 61 years and ranged from 32 to 82 years. The majority of patients were men, which reflects MD Anderson demographics, not a predisposing risk factor, Dr. Iyer said.
Melanoma was the most common cancer, followed by renal cell and prostate; patients had stage 2-4 disease. About half the subjects were on single agent anti-PD-1 treatment, about a third on anti-PD-1 combination treatment, and the rest on anti-PD-L1 combination therapy. C-peptide levels were below 0.9 ng/mL at diabetes diagnosis in most of the patients. Eleven of the 20 tested (55%) were positive for the pancreatic islet cell antibody GAD65.
The investigators had no disclosures. A funding source was not reported.
SOURCE: Iyer PC et al. Abstract OR05-5.
REPORTING FROM ENDO 2018
Key clinical point: Be on the lookout for new-onset diabetes when patients start immune checkpoint inhibitors.
Major finding: In all but one case, patients remained on insulin at a median follow-up of 44 weeks, even after stopping ICIs.
Study details: Review of 24 cases.
Disclosures: The investigators had no disclosures. A funding source was not reported.
Source: Iyer PC et al. Abstract OR05-5.