User login
Novel cell therapy beats immunotherapy in melanoma
PARIS – Cell therapies have already had a huge impact on the treatment of blood cancers, but progress in solid tumors has proved more difficult. Now, in a first multicenter randomized trial to compare the two,
The cell therapy used in this trial was composed of adoptive tumor infiltrating lymphocytes (TIL), which were made individually for each patient, just as chimeric antigen receptor T cells (CAR T cells) are for patients with blood cancers. However, the process involved is somewhat different, as TILs are made from lymphocytes that have infiltrated the patient’s tumor and are obtained by surgery in the tumor, whereas CAR T cells are made from circulating blood cells.
The phase 3 trial involved 168 patients with unresectable stage IIIC-4 melanoma and showed that patients who were treated with TILs achieved a significantly improved progression-free survival (PFS) when compared with standard immunotherapy with ipilimumab (Yervoy).
The median PFS was more than doubled to 7.2 months with TILs versus 3.1 months with ipilimumab (hazard ratio, 0.50; P < .001).
“We do think that TIL could possibly become a new treatment option for patients with advanced stage melanoma,” commented lead author John Haanen, MD, PhD, research group leader at the Netherlands Cancer Institute in Amsterdam and a professor in translational immunotherapy of cancer at Leiden (the Netherlands) University Medical Center.
He presented the findings at a presidential symposium during the European Society for Medical Oncology Annual Congress, Paris.
“The results of this trial may fuel further research of TIL in other cancer types, potentially demonstrating benefit in many other solid tumors and expanding available treatments for patients,” said Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was approached for comment by this news organization and was not involved in the research.
Immune checkpoint inhibitors and targeted therapies have become the standard of care for advanced melanoma and greatly improved patient outcomes, she said. But as about half of patients treated with these agents will not achieve a durable benefit, there remains a need for new treatment options.
“Although immunotherapy can yield impressive long-term responses, a substantial percentage of patients will have no response, or no durable response, to checkpoint inhibitors,” said Dr. Dimitrova. “TIL therapy has proven effectiveness in melanoma. However, no phase III trials have been done to date to compare its effectiveness to a standard of care regimen.”
She noted that these results are consistent with past reports of an approximately 50% response rate with an impressive 20% complete response rate in the TIL group. Data from a phase 2 trial reported last year, for example, showed an objective response rate of 36.4%.
“It will be important to determine the persistence of antitumor activity and whether there are biomarkers that could help with patient selection given the resource intensity of the therapy,” Dr. Dimitrova said. “TIL therapy will likely become a new standard of care in metastatic melanoma refractory to immune checkpoint inhibitors.”
Superior to immunotherapy
In the current study, Dr. Haanen and colleagues randomly assigned 168 patients to TIL or ipilimumab (3 mg/kg every 3 weeks, maximum 4 doses). Patients were stratified for BRAFV600 mutation status, treatment line and center, and the majority (86%) were refractory to anti–PD-1 treatment.
Patients in the TIL group underwent resection of a melanoma lesion (2-3 cm) for the ex vivo outgrowth and expansion of tumor-resident T cells. Before the cultured TILs were infused back into the patients from which they were made, the patient underwent nonmyeloablative, lymphodepleting chemotherapy with cyclophosphamide plus fludarabine that was followed by high-dose interleukin-2.
The study’s primary endpoint was progression-free survival, and secondary endpoints included overall and complete response rate, overall survival, and safety.
At a median follow-up of 33 months, TIL significantly improved progression-free survival, compared with ipilimumab. The overall response rate also favored TIL, compared with ipilimumab (49% vs. 21%), with 20% versus 7% complete responses, respectively.
The median overall survival was 25.8 months for TIL and 18.9 months for ipilimumab (HR, 0.83; P = 0.39).
Grade 3 or higher treatment-related adverse events occurred in all TIL and 57% of ipilimumab patients, although Dr. Haanen noted they were manageable and, in most cases, resolved by the time patients were discharged from the hospital.
“There were no new safety concerns with TIL,” said Dr. Haanen, “And these toxicities are driven by the chemotherapy and interleukin-2 that are part of the TIL regimen. There were no long-term sequelae in patients treated with TIL, and health-related quality of life was higher in patients treated with TIL.”
Ultra-personalized
Also commenting on the study, Anthony J. Olszanski, MD, RPh, associate professor and vice chair of clinical research, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, agreed that the treatment of patients with melanoma who do not respond to or progress after receiving treatment with immunotherapy is “challenging and represents an unmet need.”
“TIL therapy is, in some ways, ultra-personalized therapy, because we harvest immune cells from the patient’s tumor, expand them outside of the body, and then re-infuse them,” he said. “This trial, which randomized patients between TIL versus the CTLA-4 inhibitor, ipilimumab, has shown an impressive progression-free survival and overall response rate benefit and will help establish TIL therapy as a viable treatment strategy for some patients.”
The study was supported by the Dutch Cancer Society, the Netherlands Organization for Health Research and Development, the Dutch Ministry of Health, Stichting Avento, Copenhagen University Hospital, Herlev, the Danish Cancer Society, and Capital Region of Denmark Research Foundation.
Dr. Haanen and several of the co-authors have declared multiple relationships with industry as noted in the abstract. Dr. Olszanski reports participation in advisory boards for BMS, Merck, and Instil Bio, and he reports running trials for them.
A version of this article first appeared on Medscape.com.
PARIS – Cell therapies have already had a huge impact on the treatment of blood cancers, but progress in solid tumors has proved more difficult. Now, in a first multicenter randomized trial to compare the two,
The cell therapy used in this trial was composed of adoptive tumor infiltrating lymphocytes (TIL), which were made individually for each patient, just as chimeric antigen receptor T cells (CAR T cells) are for patients with blood cancers. However, the process involved is somewhat different, as TILs are made from lymphocytes that have infiltrated the patient’s tumor and are obtained by surgery in the tumor, whereas CAR T cells are made from circulating blood cells.
The phase 3 trial involved 168 patients with unresectable stage IIIC-4 melanoma and showed that patients who were treated with TILs achieved a significantly improved progression-free survival (PFS) when compared with standard immunotherapy with ipilimumab (Yervoy).
The median PFS was more than doubled to 7.2 months with TILs versus 3.1 months with ipilimumab (hazard ratio, 0.50; P < .001).
“We do think that TIL could possibly become a new treatment option for patients with advanced stage melanoma,” commented lead author John Haanen, MD, PhD, research group leader at the Netherlands Cancer Institute in Amsterdam and a professor in translational immunotherapy of cancer at Leiden (the Netherlands) University Medical Center.
He presented the findings at a presidential symposium during the European Society for Medical Oncology Annual Congress, Paris.
“The results of this trial may fuel further research of TIL in other cancer types, potentially demonstrating benefit in many other solid tumors and expanding available treatments for patients,” said Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was approached for comment by this news organization and was not involved in the research.
Immune checkpoint inhibitors and targeted therapies have become the standard of care for advanced melanoma and greatly improved patient outcomes, she said. But as about half of patients treated with these agents will not achieve a durable benefit, there remains a need for new treatment options.
“Although immunotherapy can yield impressive long-term responses, a substantial percentage of patients will have no response, or no durable response, to checkpoint inhibitors,” said Dr. Dimitrova. “TIL therapy has proven effectiveness in melanoma. However, no phase III trials have been done to date to compare its effectiveness to a standard of care regimen.”
She noted that these results are consistent with past reports of an approximately 50% response rate with an impressive 20% complete response rate in the TIL group. Data from a phase 2 trial reported last year, for example, showed an objective response rate of 36.4%.
“It will be important to determine the persistence of antitumor activity and whether there are biomarkers that could help with patient selection given the resource intensity of the therapy,” Dr. Dimitrova said. “TIL therapy will likely become a new standard of care in metastatic melanoma refractory to immune checkpoint inhibitors.”
Superior to immunotherapy
In the current study, Dr. Haanen and colleagues randomly assigned 168 patients to TIL or ipilimumab (3 mg/kg every 3 weeks, maximum 4 doses). Patients were stratified for BRAFV600 mutation status, treatment line and center, and the majority (86%) were refractory to anti–PD-1 treatment.
Patients in the TIL group underwent resection of a melanoma lesion (2-3 cm) for the ex vivo outgrowth and expansion of tumor-resident T cells. Before the cultured TILs were infused back into the patients from which they were made, the patient underwent nonmyeloablative, lymphodepleting chemotherapy with cyclophosphamide plus fludarabine that was followed by high-dose interleukin-2.
The study’s primary endpoint was progression-free survival, and secondary endpoints included overall and complete response rate, overall survival, and safety.
At a median follow-up of 33 months, TIL significantly improved progression-free survival, compared with ipilimumab. The overall response rate also favored TIL, compared with ipilimumab (49% vs. 21%), with 20% versus 7% complete responses, respectively.
The median overall survival was 25.8 months for TIL and 18.9 months for ipilimumab (HR, 0.83; P = 0.39).
Grade 3 or higher treatment-related adverse events occurred in all TIL and 57% of ipilimumab patients, although Dr. Haanen noted they were manageable and, in most cases, resolved by the time patients were discharged from the hospital.
“There were no new safety concerns with TIL,” said Dr. Haanen, “And these toxicities are driven by the chemotherapy and interleukin-2 that are part of the TIL regimen. There were no long-term sequelae in patients treated with TIL, and health-related quality of life was higher in patients treated with TIL.”
Ultra-personalized
Also commenting on the study, Anthony J. Olszanski, MD, RPh, associate professor and vice chair of clinical research, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, agreed that the treatment of patients with melanoma who do not respond to or progress after receiving treatment with immunotherapy is “challenging and represents an unmet need.”
“TIL therapy is, in some ways, ultra-personalized therapy, because we harvest immune cells from the patient’s tumor, expand them outside of the body, and then re-infuse them,” he said. “This trial, which randomized patients between TIL versus the CTLA-4 inhibitor, ipilimumab, has shown an impressive progression-free survival and overall response rate benefit and will help establish TIL therapy as a viable treatment strategy for some patients.”
The study was supported by the Dutch Cancer Society, the Netherlands Organization for Health Research and Development, the Dutch Ministry of Health, Stichting Avento, Copenhagen University Hospital, Herlev, the Danish Cancer Society, and Capital Region of Denmark Research Foundation.
Dr. Haanen and several of the co-authors have declared multiple relationships with industry as noted in the abstract. Dr. Olszanski reports participation in advisory boards for BMS, Merck, and Instil Bio, and he reports running trials for them.
A version of this article first appeared on Medscape.com.
PARIS – Cell therapies have already had a huge impact on the treatment of blood cancers, but progress in solid tumors has proved more difficult. Now, in a first multicenter randomized trial to compare the two,
The cell therapy used in this trial was composed of adoptive tumor infiltrating lymphocytes (TIL), which were made individually for each patient, just as chimeric antigen receptor T cells (CAR T cells) are for patients with blood cancers. However, the process involved is somewhat different, as TILs are made from lymphocytes that have infiltrated the patient’s tumor and are obtained by surgery in the tumor, whereas CAR T cells are made from circulating blood cells.
The phase 3 trial involved 168 patients with unresectable stage IIIC-4 melanoma and showed that patients who were treated with TILs achieved a significantly improved progression-free survival (PFS) when compared with standard immunotherapy with ipilimumab (Yervoy).
The median PFS was more than doubled to 7.2 months with TILs versus 3.1 months with ipilimumab (hazard ratio, 0.50; P < .001).
“We do think that TIL could possibly become a new treatment option for patients with advanced stage melanoma,” commented lead author John Haanen, MD, PhD, research group leader at the Netherlands Cancer Institute in Amsterdam and a professor in translational immunotherapy of cancer at Leiden (the Netherlands) University Medical Center.
He presented the findings at a presidential symposium during the European Society for Medical Oncology Annual Congress, Paris.
“The results of this trial may fuel further research of TIL in other cancer types, potentially demonstrating benefit in many other solid tumors and expanding available treatments for patients,” said Maya Dimitrova, MD, medical oncologist at NYU Langone Perlmutter Cancer Center. She was approached for comment by this news organization and was not involved in the research.
Immune checkpoint inhibitors and targeted therapies have become the standard of care for advanced melanoma and greatly improved patient outcomes, she said. But as about half of patients treated with these agents will not achieve a durable benefit, there remains a need for new treatment options.
“Although immunotherapy can yield impressive long-term responses, a substantial percentage of patients will have no response, or no durable response, to checkpoint inhibitors,” said Dr. Dimitrova. “TIL therapy has proven effectiveness in melanoma. However, no phase III trials have been done to date to compare its effectiveness to a standard of care regimen.”
She noted that these results are consistent with past reports of an approximately 50% response rate with an impressive 20% complete response rate in the TIL group. Data from a phase 2 trial reported last year, for example, showed an objective response rate of 36.4%.
“It will be important to determine the persistence of antitumor activity and whether there are biomarkers that could help with patient selection given the resource intensity of the therapy,” Dr. Dimitrova said. “TIL therapy will likely become a new standard of care in metastatic melanoma refractory to immune checkpoint inhibitors.”
Superior to immunotherapy
In the current study, Dr. Haanen and colleagues randomly assigned 168 patients to TIL or ipilimumab (3 mg/kg every 3 weeks, maximum 4 doses). Patients were stratified for BRAFV600 mutation status, treatment line and center, and the majority (86%) were refractory to anti–PD-1 treatment.
Patients in the TIL group underwent resection of a melanoma lesion (2-3 cm) for the ex vivo outgrowth and expansion of tumor-resident T cells. Before the cultured TILs were infused back into the patients from which they were made, the patient underwent nonmyeloablative, lymphodepleting chemotherapy with cyclophosphamide plus fludarabine that was followed by high-dose interleukin-2.
The study’s primary endpoint was progression-free survival, and secondary endpoints included overall and complete response rate, overall survival, and safety.
At a median follow-up of 33 months, TIL significantly improved progression-free survival, compared with ipilimumab. The overall response rate also favored TIL, compared with ipilimumab (49% vs. 21%), with 20% versus 7% complete responses, respectively.
The median overall survival was 25.8 months for TIL and 18.9 months for ipilimumab (HR, 0.83; P = 0.39).
Grade 3 or higher treatment-related adverse events occurred in all TIL and 57% of ipilimumab patients, although Dr. Haanen noted they were manageable and, in most cases, resolved by the time patients were discharged from the hospital.
“There were no new safety concerns with TIL,” said Dr. Haanen, “And these toxicities are driven by the chemotherapy and interleukin-2 that are part of the TIL regimen. There were no long-term sequelae in patients treated with TIL, and health-related quality of life was higher in patients treated with TIL.”
Ultra-personalized
Also commenting on the study, Anthony J. Olszanski, MD, RPh, associate professor and vice chair of clinical research, department of hematology/oncology at Fox Chase Cancer Center, Philadelphia, agreed that the treatment of patients with melanoma who do not respond to or progress after receiving treatment with immunotherapy is “challenging and represents an unmet need.”
“TIL therapy is, in some ways, ultra-personalized therapy, because we harvest immune cells from the patient’s tumor, expand them outside of the body, and then re-infuse them,” he said. “This trial, which randomized patients between TIL versus the CTLA-4 inhibitor, ipilimumab, has shown an impressive progression-free survival and overall response rate benefit and will help establish TIL therapy as a viable treatment strategy for some patients.”
The study was supported by the Dutch Cancer Society, the Netherlands Organization for Health Research and Development, the Dutch Ministry of Health, Stichting Avento, Copenhagen University Hospital, Herlev, the Danish Cancer Society, and Capital Region of Denmark Research Foundation.
Dr. Haanen and several of the co-authors have declared multiple relationships with industry as noted in the abstract. Dr. Olszanski reports participation in advisory boards for BMS, Merck, and Instil Bio, and he reports running trials for them.
A version of this article first appeared on Medscape.com.
‘Smoking gun–level’ evidence found linking air pollution with lung cancer
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
PARIS – Air pollution has been recognized as a risk factor for lung cancer for about 2 decades, and already present in normal lung cells to cause cancer.
Think of it as “smoking gun–level” evidence that may explain why many nonsmokers still develop non–small cell lung cancer, said Charles Swanton, PhD, from the Francis Crick Institute and Cancer Research UK Chief Clinician, London.
“What this work shows is that air pollution is directly causing lung cancer but through a slightly unexpected pathway,” he said at a briefing prior to his presentation of the data in a presidential symposium held earlier this month in Paris at the European Society for Medical Oncology Congress 2022.
Importantly, he and his team also propose a mechanism for blocking the effects of air pollution with monoclonal antibodies directed against the inflammatory cytokine interleukein-1 beta.
Carcinogenesis explored
Lung cancer in never-smokers has a low mutational burden, with about 5- to 10-fold fewer mutations in a nonsmoker, compared with an ever smoker or current smoker, Dr. Swanton noted.
“The other thing to say about never-smokers is that they don’t have a clear environmental carcinogenic signature. So how do you square the circle? You’ve got the problem that you know that air pollution is associated with lung cancer – we don’t know if it causes it – but we also see that we’ve got no DNA mutations due to an environmental carcinogen,” he said during his symposium presentation.
The traditional model proposed to explain how carcinogens cause cancer holds that exposure to a carcinogen causes DNA mutations that lead to clonal expansion and tumor growth.
“But there are some major problems with this model,” Dr. Swanton said.
For example, normal skin contains a “patchwork of mutant clones,” but skin cancer is still uncommon, he said, and in studies in mice, 17 of 20 environmental carcinogens did not induce DNA mutations. He also noted that a common melanoma driver mutation, BRAF V600E, is not induced by exposure to a ultraviolet light.
“Any explanation for never-smoking lung cancer would have to fulfill three criteria: one, you have to explain why geographic variation exists; two, you have to prove causation; and three, you have to explain how cancers can be initiated without directly causing DNA mutations,” he said.
Normal lung tissues in nonsmoking adults can harbor pre-existing mutations, with the number of mutations increasing likely as a consequence of aging. In fact, more than 50% of normal lung biopsy tissues have been shown to harbor driver KRAS and/or EGFR mutations, Dr. Swanton said.
“In our research, these mutations alone only weakly potentiated cancer in laboratory models. However, when lung cells with these mutations were exposed to air pollutants, we saw more cancers and these occurred more quickly than when lung cells with these mutations were not exposed to pollutants, suggesting that air pollution promotes the initiation of lung cancer in cells harboring driver gene mutations. The next step is to discover why some lung cells with mutations become cancerous when exposed to pollutants while others don’t,” he said.
Geographical exposures
Looking at data on 447,932 participants in the UK Biobank, the investigators found that increasing exposure to ambient air particles smaller than 2.5 mcm (PM2.5) was significantly associated with seven cancer types, including lung cancer. They also saw an association between PM2.5 exposure levels and EGFR-mutated lung cancer incidence in the United Kingdom, South Korea, and Taiwan.
And crucially, as Dr. Swanton and associates showed in mouse models, exposure of lung cells bearing somatic EGFR and KRAS mutations to PM2.5 causes recruitment of macrophages that in turn secrete IL-1B, resulting in a transdifferentiation of EGFR-mutated cells into a cancer stem cell state, and tumor formation.
Importantly, pollution-induced tumor formation can be blocked by antibodies directed against IL-1B, Dr. Swanton said.
He pointed to a 2017 study in The Lancet suggesting that anti-inflammatory therapy with the anti–IL-1 antibody canakinumab (Ilaris) could reduce incident lung cancer and lung cancer deaths.
‘Elegant first demonstration’
“This is a very meaningful demonstration, from epidemiological data to preclinical models of the role of PM2.5 air pollutants in the promotion of lung cancer, and it provides us with very important insights into the mechanism through which nonsmokers can get lung cancer,” commented Suzette Delaloge, MD, from the cancer interception program at Institut Goustave Roussy in Villejuif, France, the invited discussant.
“But beyond that, it also has a great impact on our vision of carcinogenesis, with this very elegant first demonstration of the alternative nonmutagenic, carcinogenetic promotion hypothesis for fine particulate matter,” she said.
Questions still to be answered include whether PM2.5 pollutants could also be mutagenic, is the oncogenic pathway ubiquitous in tissue, which components of PM2.5 might drive the effect, how long of an exposure is required to promote lung cancer, and why and how persons without cancer develop specific driver mutations such as EGFR, she said.
“This research is intriguing and exciting as it means that we can ask whether, in the future, it will be possible to use lung scans to look for precancerous lesions in the lungs and try to reverse them with medicines such as interleukin-1B inhibitors,” said Tony Mok, MD, a lung cancer specialist at the Chinese University of Hong Kong, who was not involved in the study.
“We don’t yet know whether it will be possible to use highly sensitive EGFR profiling on blood or other samples to find nonsmokers who are predisposed to lung cancer and may benefit from lung scanning, so discussions are still very speculative,” he said in a statement.
The study was supported by Cancer Research UK, the Lung Cancer Research Foundations, Rosetrees Trust, the Mark Foundation for Cancer Research and the Ruth Strauss Foundation. Dr. Swanton disclosed grants/research support, honoraria, and stock ownership with multiple entities. Dr. Delaloge disclosed institutional financing and research funding from multiple companies. Dr. Mok disclosed stock ownership and honoraria with multiple companies.
AT ESMO CONGRESS 2022
Overall survival dips with vitamin D deficiency in melanoma
, according to research presented at the annual congress of the European Academy of Dermatology and Venereology.
Whereas the 5-year overall survival was 90% when vitamin D serum levels were above a 10 ng/mL threshold, it was 84% when levels fell below it. Notably, the gap in overall survival between those above and below the threshold appeared to widen as time went on.
The research adds to existing evidence that “vitamin D levels can play an important and independent role in patients’ survival outcomes,” study investigator Inés Gracia-Darder, MD, told this news organization. “The important application in clinical practice would be to know if vitamin D supplementation influences the survival of melanoma patients,” said Dr. Gracia-Darder, a clinical specialist in dermatology at the Hospital Universitari Son Espases, Mallorca, Spain.
Known association, but not much data
“It is not a new finding,” but there are limited data, especially in melanoma, said Julie De Smedt, MD, of KU Leuven, Belgium, who was asked to comment on the results. Other groups have shown, certainly for cancer in general, that vitamin D can have an effect on overall survival.
“Low levels of vitamin D are associated with the pathological parameters of the melanoma, such as the thickness of the tumor,” Dr. De Smedt said in an interview, indicating that it’s not just overall survival that might be affected.
“So we assume that also has an effect on melanoma-specific survival,” she added.
That assumption, however, is not supported by the data Dr. Gracia-Darder presented, as there was no difference in melanoma-specific survival among the two groups of patients that had been studied.
Retrospective cohort analysis
Vitamin D levels had been studied in 264 patients who were included in the retrospective cohort analysis. All had invasive melanomas, and all had been seen at the Hospital Clinic of Barcelona between January 1998 and June 2021. Their mean age was 57 years, and the median follow-up was 6.7 years.
For inclusion, all patients had to have had their vitamin D levels measured after being diagnosed with melanoma; those with a 25-hydroxyvitamin D3 serum level of less than 10 ng/mL were deemed to be vitamin D deficient, whereas those with levels of 10 ng/mL and above were deemed normal or insufficient.
A measurement less than 10 ng/mL is considered vitamin D deficiency, Dr. De Smedt said. “But there is a difference between countries, and there’s also a difference between societies,” noting the cut-off used in the lab where she works is 20 ng/mL. This makes it difficult to compare studies, she said.
Independent association with overall survival
Seasonal variation in vitamin D levels were considered as a possible confounding factor, but Dr. Gracia-Darder noted that there was a similar distribution of measurements taken between October to March and April to September.
Univariate and multivariate analyses established vitamin D deficiency as being independently associated with overall survival with hazard ratios of 2.34 and 2.45, respectively.
Other predictive factors were having a higher Breslow index, as well as older age and gender.
Time to recommend vitamin D supplementation?
So should patients with melanoma have their vitamin D levels routinely checked? And what about advising them to take vitamin D supplements?
“In our practice, we analyze the vitamin D levels of our patients,” Dr. Gracia-Darder said. Patients are told to limit their exposure to the sun because of their skin cancer, so they are very likely to become vitamin D deficient.
While dietary changes or supplements might be suggested, there’s no real evidence to support upping vitamin D levels to date, so “future prospective studies are needed,” Dr. Gracia-Darder added.
Such studies have already started, including one in Italy, one in Australia, and another study that Dr. De Smedt has been involved with for the past few years.
Called the ViDMe study, it’s a multicenter, randomized, double-blind trial in which patients are being given a high-dose oral vitamin D supplement or placebo once a month for at least 1 year. About 430 patients with a first cutaneous malignant melanoma have been included in the trial, which started in December 2012.
It is hoped that the results will show that the supplementation will have had a protective effect on the risk of relapse and that there will be a correlation between vitamin D levels in the blood and vitamin D receptor immunoreactivity in the tumor.
“The study is still blinded,” Dr. De Smedt said. “We will unblind in the coming months and then at the end of the year, maybe next year, we will have the results.”
The study reported by Dr. Gracia-Darder did not receive any specific funding. Dr. Gracia-Darder disclosed that the melanoma unit where the study was performed receives many grants and funds to carry out research. She reported no other relevant financial relationships. Dr. De Smedt had no relevant financial relationships. The ViDMe study is sponsored by the Universitaire Ziekenhuizen Leuven.
A version of this article first appeared on Medscape.com.
, according to research presented at the annual congress of the European Academy of Dermatology and Venereology.
Whereas the 5-year overall survival was 90% when vitamin D serum levels were above a 10 ng/mL threshold, it was 84% when levels fell below it. Notably, the gap in overall survival between those above and below the threshold appeared to widen as time went on.
The research adds to existing evidence that “vitamin D levels can play an important and independent role in patients’ survival outcomes,” study investigator Inés Gracia-Darder, MD, told this news organization. “The important application in clinical practice would be to know if vitamin D supplementation influences the survival of melanoma patients,” said Dr. Gracia-Darder, a clinical specialist in dermatology at the Hospital Universitari Son Espases, Mallorca, Spain.
Known association, but not much data
“It is not a new finding,” but there are limited data, especially in melanoma, said Julie De Smedt, MD, of KU Leuven, Belgium, who was asked to comment on the results. Other groups have shown, certainly for cancer in general, that vitamin D can have an effect on overall survival.
“Low levels of vitamin D are associated with the pathological parameters of the melanoma, such as the thickness of the tumor,” Dr. De Smedt said in an interview, indicating that it’s not just overall survival that might be affected.
“So we assume that also has an effect on melanoma-specific survival,” she added.
That assumption, however, is not supported by the data Dr. Gracia-Darder presented, as there was no difference in melanoma-specific survival among the two groups of patients that had been studied.
Retrospective cohort analysis
Vitamin D levels had been studied in 264 patients who were included in the retrospective cohort analysis. All had invasive melanomas, and all had been seen at the Hospital Clinic of Barcelona between January 1998 and June 2021. Their mean age was 57 years, and the median follow-up was 6.7 years.
For inclusion, all patients had to have had their vitamin D levels measured after being diagnosed with melanoma; those with a 25-hydroxyvitamin D3 serum level of less than 10 ng/mL were deemed to be vitamin D deficient, whereas those with levels of 10 ng/mL and above were deemed normal or insufficient.
A measurement less than 10 ng/mL is considered vitamin D deficiency, Dr. De Smedt said. “But there is a difference between countries, and there’s also a difference between societies,” noting the cut-off used in the lab where she works is 20 ng/mL. This makes it difficult to compare studies, she said.
Independent association with overall survival
Seasonal variation in vitamin D levels were considered as a possible confounding factor, but Dr. Gracia-Darder noted that there was a similar distribution of measurements taken between October to March and April to September.
Univariate and multivariate analyses established vitamin D deficiency as being independently associated with overall survival with hazard ratios of 2.34 and 2.45, respectively.
Other predictive factors were having a higher Breslow index, as well as older age and gender.
Time to recommend vitamin D supplementation?
So should patients with melanoma have their vitamin D levels routinely checked? And what about advising them to take vitamin D supplements?
“In our practice, we analyze the vitamin D levels of our patients,” Dr. Gracia-Darder said. Patients are told to limit their exposure to the sun because of their skin cancer, so they are very likely to become vitamin D deficient.
While dietary changes or supplements might be suggested, there’s no real evidence to support upping vitamin D levels to date, so “future prospective studies are needed,” Dr. Gracia-Darder added.
Such studies have already started, including one in Italy, one in Australia, and another study that Dr. De Smedt has been involved with for the past few years.
Called the ViDMe study, it’s a multicenter, randomized, double-blind trial in which patients are being given a high-dose oral vitamin D supplement or placebo once a month for at least 1 year. About 430 patients with a first cutaneous malignant melanoma have been included in the trial, which started in December 2012.
It is hoped that the results will show that the supplementation will have had a protective effect on the risk of relapse and that there will be a correlation between vitamin D levels in the blood and vitamin D receptor immunoreactivity in the tumor.
“The study is still blinded,” Dr. De Smedt said. “We will unblind in the coming months and then at the end of the year, maybe next year, we will have the results.”
The study reported by Dr. Gracia-Darder did not receive any specific funding. Dr. Gracia-Darder disclosed that the melanoma unit where the study was performed receives many grants and funds to carry out research. She reported no other relevant financial relationships. Dr. De Smedt had no relevant financial relationships. The ViDMe study is sponsored by the Universitaire Ziekenhuizen Leuven.
A version of this article first appeared on Medscape.com.
, according to research presented at the annual congress of the European Academy of Dermatology and Venereology.
Whereas the 5-year overall survival was 90% when vitamin D serum levels were above a 10 ng/mL threshold, it was 84% when levels fell below it. Notably, the gap in overall survival between those above and below the threshold appeared to widen as time went on.
The research adds to existing evidence that “vitamin D levels can play an important and independent role in patients’ survival outcomes,” study investigator Inés Gracia-Darder, MD, told this news organization. “The important application in clinical practice would be to know if vitamin D supplementation influences the survival of melanoma patients,” said Dr. Gracia-Darder, a clinical specialist in dermatology at the Hospital Universitari Son Espases, Mallorca, Spain.
Known association, but not much data
“It is not a new finding,” but there are limited data, especially in melanoma, said Julie De Smedt, MD, of KU Leuven, Belgium, who was asked to comment on the results. Other groups have shown, certainly for cancer in general, that vitamin D can have an effect on overall survival.
“Low levels of vitamin D are associated with the pathological parameters of the melanoma, such as the thickness of the tumor,” Dr. De Smedt said in an interview, indicating that it’s not just overall survival that might be affected.
“So we assume that also has an effect on melanoma-specific survival,” she added.
That assumption, however, is not supported by the data Dr. Gracia-Darder presented, as there was no difference in melanoma-specific survival among the two groups of patients that had been studied.
Retrospective cohort analysis
Vitamin D levels had been studied in 264 patients who were included in the retrospective cohort analysis. All had invasive melanomas, and all had been seen at the Hospital Clinic of Barcelona between January 1998 and June 2021. Their mean age was 57 years, and the median follow-up was 6.7 years.
For inclusion, all patients had to have had their vitamin D levels measured after being diagnosed with melanoma; those with a 25-hydroxyvitamin D3 serum level of less than 10 ng/mL were deemed to be vitamin D deficient, whereas those with levels of 10 ng/mL and above were deemed normal or insufficient.
A measurement less than 10 ng/mL is considered vitamin D deficiency, Dr. De Smedt said. “But there is a difference between countries, and there’s also a difference between societies,” noting the cut-off used in the lab where she works is 20 ng/mL. This makes it difficult to compare studies, she said.
Independent association with overall survival
Seasonal variation in vitamin D levels were considered as a possible confounding factor, but Dr. Gracia-Darder noted that there was a similar distribution of measurements taken between October to March and April to September.
Univariate and multivariate analyses established vitamin D deficiency as being independently associated with overall survival with hazard ratios of 2.34 and 2.45, respectively.
Other predictive factors were having a higher Breslow index, as well as older age and gender.
Time to recommend vitamin D supplementation?
So should patients with melanoma have their vitamin D levels routinely checked? And what about advising them to take vitamin D supplements?
“In our practice, we analyze the vitamin D levels of our patients,” Dr. Gracia-Darder said. Patients are told to limit their exposure to the sun because of their skin cancer, so they are very likely to become vitamin D deficient.
While dietary changes or supplements might be suggested, there’s no real evidence to support upping vitamin D levels to date, so “future prospective studies are needed,” Dr. Gracia-Darder added.
Such studies have already started, including one in Italy, one in Australia, and another study that Dr. De Smedt has been involved with for the past few years.
Called the ViDMe study, it’s a multicenter, randomized, double-blind trial in which patients are being given a high-dose oral vitamin D supplement or placebo once a month for at least 1 year. About 430 patients with a first cutaneous malignant melanoma have been included in the trial, which started in December 2012.
It is hoped that the results will show that the supplementation will have had a protective effect on the risk of relapse and that there will be a correlation between vitamin D levels in the blood and vitamin D receptor immunoreactivity in the tumor.
“The study is still blinded,” Dr. De Smedt said. “We will unblind in the coming months and then at the end of the year, maybe next year, we will have the results.”
The study reported by Dr. Gracia-Darder did not receive any specific funding. Dr. Gracia-Darder disclosed that the melanoma unit where the study was performed receives many grants and funds to carry out research. She reported no other relevant financial relationships. Dr. De Smedt had no relevant financial relationships. The ViDMe study is sponsored by the Universitaire Ziekenhuizen Leuven.
A version of this article first appeared on Medscape.com.
FROM THE EADV CONGRESS
Ultra-Late Cutaneous Melanoma Recurrence Following 49 Years of Quiescence
To the Editor:
Ultra-late melanoma recurrence represents a minority of cases in which the quiescent period lasts longer than 15 years, and epidemiologic studies have reported recurrence rates of 6% to 10% during the ultra-late period.1 Even more uncommon are cases that span many decades (eg, >30 years), but all are useful in understanding the cellular behavior leading to the reactivation of fully excised melanomas. Few cases have been reported in which recurrence occurs more than 35 years after the original diagnosis of melanoma. Unfortunately, mechanisms underlying this long stable quiescence and subsequent reactivation are poorly understood, which is why it is important to identify and document cases. We present a case of local recurrence of cutaneous melanoma on the patient’s lower back after a 49-year disease-free period.
A 78-year-old White woman presented to a private dermatology office for a full-body skin examination. She had a medical history of a cutaneous melanoma that had been removed on the lower back 49 years prior; Parkinson disease of 10 years’ duration; and an enlarged thyroid nodule with decreased thyrotropin and hyperthyroidism, atrial fibrillation, mitral valve prolapse, osteoarthritis in the knees, and actinic keratoses, all of which were chronic conditions lasting years to decades. She was taking several medications for these medical conditions. Her surgical history included a hysterectomy, hip replacement, hernia repair, cardioversion, and tonsillectomy in childhood. Her family medical history included breast cancer in her paternal grandmother and aunt; hypertension in her father; and sarcoma in her mother at 78 years of age, which initially was identified in the sacrum and metastasized to the lungs causing death. No family history of melanoma or other skin cancers was reported. Prior to the original diagnosis of melanoma at 29 years of age, she had no history of skin cancer or any other medical condition other than acne. The patient did report spending a great deal of time in the sun during high school.
The patient reported developing the original cutaneous melanoma during her second pregnancy at 29 years of age and recalled that it was excised with wide margins. There had been a mole on her back that was present for years but changed in size during pregnancy, prompting the original visit to the primary care physician for evaluation. Remarkably, the original pathology report was obtained from the patient and revealed a specimen consisting of a 3.7×1.7-cm skin
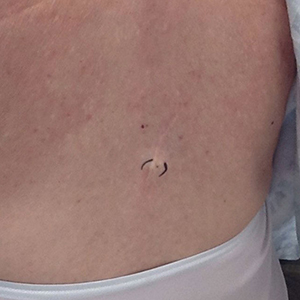
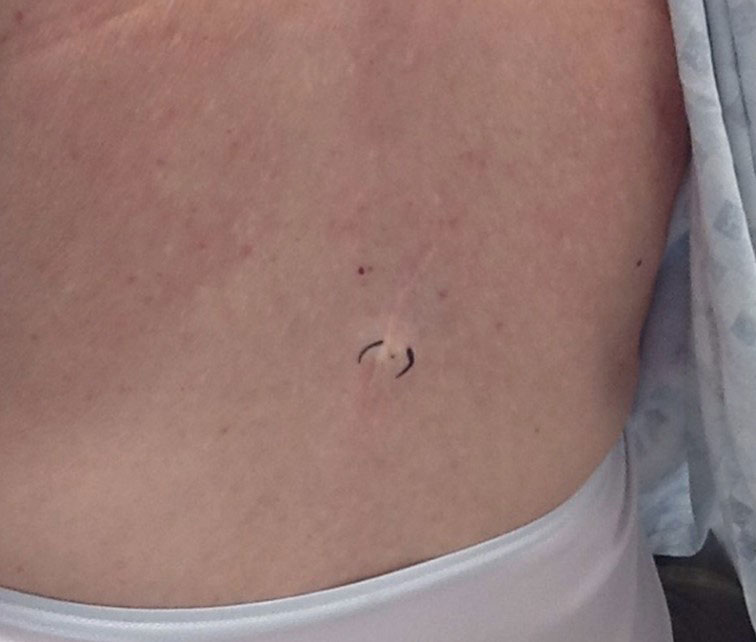
Physical examination at the current presentation 49 years later revealed an even-bordered 2-mm black macule that was located approximately 1 cm from the original melanoma excision scar line (Figure). A biopsy was performed and sent to a dermatopathologist. Microscopic evaluation revealed nests, islands, and sheets of atypical epithelioid melanocytes extending through the dermis between collagen bundles. The melanocytes varied in size and shape with moderate nuclear pleomorphism present. Scattered mitotic figures and necrotic melanocytes were present, which most likely represented cutaneous satellite metastases of melanoma. Subsequent chest radiography, full-body positron emission tomography, and standard laboratory blood tests were unremarkable except for an enlarged right thyroid gland and moderate cardiomegaly. The patient was sent to a surgical oncologist for excision with wide surgical margins, and she elected not to have a sentinel lymph node biopsy. At follow-up 3, 6, 12, and 24 months later, there were no signs of recurrence based on direct clinical examination. The patient subsequently was lost to follow-up.
Recurrence rates of melanoma vary by stage and age at diagnosis, but prior studies have reported a recurrence rate of approximately 6% after 10 or more years following the initial diagnosis.2 Ultra-late recurrences of approximately 4 decades or more are extremely rare. A PubMed search of articles indexed for MEDLINE using the terms melanoma and ultra-late recurrence revealed 4 reported cases with a quiescent period of 38 or more years.3-6 All cases were metastatic melanomas in women; spanned 38, 40, 41, and 45 years from the initial melanoma diagnosis to recurrence; and all of the recurrences except one were regional or distal metastatic lesions (eg, lymph node, brain). In one case, both the original and recurrent lesions occurred on the left elbow.6 The original lesions occurred on the legs, elbow, and back of the neck, and there were no notable concomitant medical conditions. The patients were aged 72, 73, 73, and 84 years at recurrence.3-6 However, generalizations from these cases are limited given the potential for selection bias (eg, men may be less likely to visit a clinic for follow-up and nevi examination) and the likelihood that many cases of ultra-late melanoma recurrence are unrecognized or unreported.
More recently, genomic analyses on melanoma lesions occurring 30 years apart confirmed that the second lesion was indeed a recurrence, although with numerous additional mutations.7 The specific mechanisms underlying the dormancy and subsequent reemergence of metastatic lesions are unclear, but
It also is worth highlighting the concomitant diagnosis of Parkinson disease in our patient. In recent years, Parkinson disease has been linked to melanoma in both epidemiologic and genetic studies. For example, one large-scale study found a 50% increased risk for developing Parkinson disease in patients with melanoma (and vice versa), and this finding has been replicated in other studies.10 Moreover, patients with Parkinson disease have a 2-fold increase in their risk for developing melanoma, demonstrating that it is a bidirectional pathway. Not surprisingly, associations between melanin and neuromelanin pathways have been identified as a potential link between these diseases, and scientists are in the process of understanding the genetic components of both.10 It is unknown if specific genetic mutations contributed to both diseases in our case, but follow-up genetic testing on the recurrent melanoma specimen currently is being pursued.
The 49-year quiescent period in our case of recurrent cutaneous malignant melanoma potentially represents the longest ultra-late recurrence of melanoma in the literature to date based on a review of indexed publications. Moreover, it is relatively unique compared to other similar cases in that the recurrence was within a centimeter of the original excisional scar. Most metastases occur in locoregional lymph nodes or the lungs3; therefore, it is unusual to find one so close to the original lesion, especially one that occurred decades later. Factors associated with ultra-late recurrences are unknown, primarily because of the rarity of these cases as well as the biases and other factors that limit existing studies. However, genetic sequencing may provide information regarding these factors and related processes. Genetic sequencing specifically points to a small cell group remaining after excision of the primary tumor, which mutates while proliferating. Low antigenicity and tolerance to immunity during the quiescent period may explain the long duration of dormancy.6 More recently, there have been efforts to identify immunohistochemical signatures that may predict late recurrences, though the data are preliminary in nature.11
Given the latency period and location of the recurrence, our case demonstrates that even fully excised melanomas may recur locally many decades later, hence patients should be aware of the importance of a lifetime of vigilance after being diagnosed with melanoma.
- Tsao H, Cosimi AB, Sober AJ. Ultra-late recurrence (15 years or longer) of cutaneous melanoma. Cancer. 1997;79:2361-2370.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Mansour D, Kejariwal D. It is never too late: ultra-late recurrence of melanoma with distant metastases [published online March 8, 2012]. BMJ Case Rep. 2012:bcr0120125474. doi:10.1136/bcr.01.2012.5474
- Saleh D, Peach AHS. Ultra-late recurrence of malignant melanoma after 40 years of quiescent disease. J Surg Oncol. 2011;103:290-291.
- Goodenough J, Cozon CL, Liew SH. An incidental finding of a nodal recurrence of cutaneous malignant melanoma after a 45-year disease-free period [published online June 4, 2014]. BMJ Case Rep. 2014:bcr2014204289. doi:10.1136/bcr-2014-204289
- Nakamura M, Obayashi M, Yoshimitsu M, et al. Comparative whole-exome sequencing of an ultra-late recurrent malignant melanoma. Br J Dermatol. 2021;184:762-763.
- Miller JJ, Lofgren KA, Hughes SR, et al. Genomic analysis of melanoma evolution following a 30-year disease-free interval. J Cutan Pathol. 2017;44:805-808.
- North JP, Kageshita T, Pinkel D, et al. Distribution and significance of occult intraepidermal tumor cells surrounding primary melanoma. J Invest Dermatol. 2008;128:2024-2030.
- Massi G, LeBoit PE. Recurrent and persistent melanoma. In: Massi G, LeBoit PE, eds. Histological Diagnosis of Nevi and Melanoma. 2nd ed. Springer-Verlag; 2014:689-698.
- Bose A, Petsko GA, Eliezer D. Parkinson’s disease and melanoma: co-occurrence and mechanisms. J Parkinsons Dis. 2018;8:385-398.
- Reschke R, Dumann K, Ziemer M. Risk stratification and clinical characteristics of patients with late recurrence of melanoma (>10 years).J Clin Med. 2022;11:2026.
To the Editor:
Ultra-late melanoma recurrence represents a minority of cases in which the quiescent period lasts longer than 15 years, and epidemiologic studies have reported recurrence rates of 6% to 10% during the ultra-late period.1 Even more uncommon are cases that span many decades (eg, >30 years), but all are useful in understanding the cellular behavior leading to the reactivation of fully excised melanomas. Few cases have been reported in which recurrence occurs more than 35 years after the original diagnosis of melanoma. Unfortunately, mechanisms underlying this long stable quiescence and subsequent reactivation are poorly understood, which is why it is important to identify and document cases. We present a case of local recurrence of cutaneous melanoma on the patient’s lower back after a 49-year disease-free period.
A 78-year-old White woman presented to a private dermatology office for a full-body skin examination. She had a medical history of a cutaneous melanoma that had been removed on the lower back 49 years prior; Parkinson disease of 10 years’ duration; and an enlarged thyroid nodule with decreased thyrotropin and hyperthyroidism, atrial fibrillation, mitral valve prolapse, osteoarthritis in the knees, and actinic keratoses, all of which were chronic conditions lasting years to decades. She was taking several medications for these medical conditions. Her surgical history included a hysterectomy, hip replacement, hernia repair, cardioversion, and tonsillectomy in childhood. Her family medical history included breast cancer in her paternal grandmother and aunt; hypertension in her father; and sarcoma in her mother at 78 years of age, which initially was identified in the sacrum and metastasized to the lungs causing death. No family history of melanoma or other skin cancers was reported. Prior to the original diagnosis of melanoma at 29 years of age, she had no history of skin cancer or any other medical condition other than acne. The patient did report spending a great deal of time in the sun during high school.
The patient reported developing the original cutaneous melanoma during her second pregnancy at 29 years of age and recalled that it was excised with wide margins. There had been a mole on her back that was present for years but changed in size during pregnancy, prompting the original visit to the primary care physician for evaluation. Remarkably, the original pathology report was obtained from the patient and revealed a specimen consisting of a 3.7×1.7-cm skin
Physical examination at the current presentation 49 years later revealed an even-bordered 2-mm black macule that was located approximately 1 cm from the original melanoma excision scar line (Figure). A biopsy was performed and sent to a dermatopathologist. Microscopic evaluation revealed nests, islands, and sheets of atypical epithelioid melanocytes extending through the dermis between collagen bundles. The melanocytes varied in size and shape with moderate nuclear pleomorphism present. Scattered mitotic figures and necrotic melanocytes were present, which most likely represented cutaneous satellite metastases of melanoma. Subsequent chest radiography, full-body positron emission tomography, and standard laboratory blood tests were unremarkable except for an enlarged right thyroid gland and moderate cardiomegaly. The patient was sent to a surgical oncologist for excision with wide surgical margins, and she elected not to have a sentinel lymph node biopsy. At follow-up 3, 6, 12, and 24 months later, there were no signs of recurrence based on direct clinical examination. The patient subsequently was lost to follow-up.

Recurrence rates of melanoma vary by stage and age at diagnosis, but prior studies have reported a recurrence rate of approximately 6% after 10 or more years following the initial diagnosis.2 Ultra-late recurrences of approximately 4 decades or more are extremely rare. A PubMed search of articles indexed for MEDLINE using the terms melanoma and ultra-late recurrence revealed 4 reported cases with a quiescent period of 38 or more years.3-6 All cases were metastatic melanomas in women; spanned 38, 40, 41, and 45 years from the initial melanoma diagnosis to recurrence; and all of the recurrences except one were regional or distal metastatic lesions (eg, lymph node, brain). In one case, both the original and recurrent lesions occurred on the left elbow.6 The original lesions occurred on the legs, elbow, and back of the neck, and there were no notable concomitant medical conditions. The patients were aged 72, 73, 73, and 84 years at recurrence.3-6 However, generalizations from these cases are limited given the potential for selection bias (eg, men may be less likely to visit a clinic for follow-up and nevi examination) and the likelihood that many cases of ultra-late melanoma recurrence are unrecognized or unreported.
More recently, genomic analyses on melanoma lesions occurring 30 years apart confirmed that the second lesion was indeed a recurrence, although with numerous additional mutations.7 The specific mechanisms underlying the dormancy and subsequent reemergence of metastatic lesions are unclear, but
It also is worth highlighting the concomitant diagnosis of Parkinson disease in our patient. In recent years, Parkinson disease has been linked to melanoma in both epidemiologic and genetic studies. For example, one large-scale study found a 50% increased risk for developing Parkinson disease in patients with melanoma (and vice versa), and this finding has been replicated in other studies.10 Moreover, patients with Parkinson disease have a 2-fold increase in their risk for developing melanoma, demonstrating that it is a bidirectional pathway. Not surprisingly, associations between melanin and neuromelanin pathways have been identified as a potential link between these diseases, and scientists are in the process of understanding the genetic components of both.10 It is unknown if specific genetic mutations contributed to both diseases in our case, but follow-up genetic testing on the recurrent melanoma specimen currently is being pursued.
The 49-year quiescent period in our case of recurrent cutaneous malignant melanoma potentially represents the longest ultra-late recurrence of melanoma in the literature to date based on a review of indexed publications. Moreover, it is relatively unique compared to other similar cases in that the recurrence was within a centimeter of the original excisional scar. Most metastases occur in locoregional lymph nodes or the lungs3; therefore, it is unusual to find one so close to the original lesion, especially one that occurred decades later. Factors associated with ultra-late recurrences are unknown, primarily because of the rarity of these cases as well as the biases and other factors that limit existing studies. However, genetic sequencing may provide information regarding these factors and related processes. Genetic sequencing specifically points to a small cell group remaining after excision of the primary tumor, which mutates while proliferating. Low antigenicity and tolerance to immunity during the quiescent period may explain the long duration of dormancy.6 More recently, there have been efforts to identify immunohistochemical signatures that may predict late recurrences, though the data are preliminary in nature.11
Given the latency period and location of the recurrence, our case demonstrates that even fully excised melanomas may recur locally many decades later, hence patients should be aware of the importance of a lifetime of vigilance after being diagnosed with melanoma.
To the Editor:
Ultra-late melanoma recurrence represents a minority of cases in which the quiescent period lasts longer than 15 years, and epidemiologic studies have reported recurrence rates of 6% to 10% during the ultra-late period.1 Even more uncommon are cases that span many decades (eg, >30 years), but all are useful in understanding the cellular behavior leading to the reactivation of fully excised melanomas. Few cases have been reported in which recurrence occurs more than 35 years after the original diagnosis of melanoma. Unfortunately, mechanisms underlying this long stable quiescence and subsequent reactivation are poorly understood, which is why it is important to identify and document cases. We present a case of local recurrence of cutaneous melanoma on the patient’s lower back after a 49-year disease-free period.
A 78-year-old White woman presented to a private dermatology office for a full-body skin examination. She had a medical history of a cutaneous melanoma that had been removed on the lower back 49 years prior; Parkinson disease of 10 years’ duration; and an enlarged thyroid nodule with decreased thyrotropin and hyperthyroidism, atrial fibrillation, mitral valve prolapse, osteoarthritis in the knees, and actinic keratoses, all of which were chronic conditions lasting years to decades. She was taking several medications for these medical conditions. Her surgical history included a hysterectomy, hip replacement, hernia repair, cardioversion, and tonsillectomy in childhood. Her family medical history included breast cancer in her paternal grandmother and aunt; hypertension in her father; and sarcoma in her mother at 78 years of age, which initially was identified in the sacrum and metastasized to the lungs causing death. No family history of melanoma or other skin cancers was reported. Prior to the original diagnosis of melanoma at 29 years of age, she had no history of skin cancer or any other medical condition other than acne. The patient did report spending a great deal of time in the sun during high school.
The patient reported developing the original cutaneous melanoma during her second pregnancy at 29 years of age and recalled that it was excised with wide margins. There had been a mole on her back that was present for years but changed in size during pregnancy, prompting the original visit to the primary care physician for evaluation. Remarkably, the original pathology report was obtained from the patient and revealed a specimen consisting of a 3.7×1.7-cm skin
Physical examination at the current presentation 49 years later revealed an even-bordered 2-mm black macule that was located approximately 1 cm from the original melanoma excision scar line (Figure). A biopsy was performed and sent to a dermatopathologist. Microscopic evaluation revealed nests, islands, and sheets of atypical epithelioid melanocytes extending through the dermis between collagen bundles. The melanocytes varied in size and shape with moderate nuclear pleomorphism present. Scattered mitotic figures and necrotic melanocytes were present, which most likely represented cutaneous satellite metastases of melanoma. Subsequent chest radiography, full-body positron emission tomography, and standard laboratory blood tests were unremarkable except for an enlarged right thyroid gland and moderate cardiomegaly. The patient was sent to a surgical oncologist for excision with wide surgical margins, and she elected not to have a sentinel lymph node biopsy. At follow-up 3, 6, 12, and 24 months later, there were no signs of recurrence based on direct clinical examination. The patient subsequently was lost to follow-up.

Recurrence rates of melanoma vary by stage and age at diagnosis, but prior studies have reported a recurrence rate of approximately 6% after 10 or more years following the initial diagnosis.2 Ultra-late recurrences of approximately 4 decades or more are extremely rare. A PubMed search of articles indexed for MEDLINE using the terms melanoma and ultra-late recurrence revealed 4 reported cases with a quiescent period of 38 or more years.3-6 All cases were metastatic melanomas in women; spanned 38, 40, 41, and 45 years from the initial melanoma diagnosis to recurrence; and all of the recurrences except one were regional or distal metastatic lesions (eg, lymph node, brain). In one case, both the original and recurrent lesions occurred on the left elbow.6 The original lesions occurred on the legs, elbow, and back of the neck, and there were no notable concomitant medical conditions. The patients were aged 72, 73, 73, and 84 years at recurrence.3-6 However, generalizations from these cases are limited given the potential for selection bias (eg, men may be less likely to visit a clinic for follow-up and nevi examination) and the likelihood that many cases of ultra-late melanoma recurrence are unrecognized or unreported.
More recently, genomic analyses on melanoma lesions occurring 30 years apart confirmed that the second lesion was indeed a recurrence, although with numerous additional mutations.7 The specific mechanisms underlying the dormancy and subsequent reemergence of metastatic lesions are unclear, but
It also is worth highlighting the concomitant diagnosis of Parkinson disease in our patient. In recent years, Parkinson disease has been linked to melanoma in both epidemiologic and genetic studies. For example, one large-scale study found a 50% increased risk for developing Parkinson disease in patients with melanoma (and vice versa), and this finding has been replicated in other studies.10 Moreover, patients with Parkinson disease have a 2-fold increase in their risk for developing melanoma, demonstrating that it is a bidirectional pathway. Not surprisingly, associations between melanin and neuromelanin pathways have been identified as a potential link between these diseases, and scientists are in the process of understanding the genetic components of both.10 It is unknown if specific genetic mutations contributed to both diseases in our case, but follow-up genetic testing on the recurrent melanoma specimen currently is being pursued.
The 49-year quiescent period in our case of recurrent cutaneous malignant melanoma potentially represents the longest ultra-late recurrence of melanoma in the literature to date based on a review of indexed publications. Moreover, it is relatively unique compared to other similar cases in that the recurrence was within a centimeter of the original excisional scar. Most metastases occur in locoregional lymph nodes or the lungs3; therefore, it is unusual to find one so close to the original lesion, especially one that occurred decades later. Factors associated with ultra-late recurrences are unknown, primarily because of the rarity of these cases as well as the biases and other factors that limit existing studies. However, genetic sequencing may provide information regarding these factors and related processes. Genetic sequencing specifically points to a small cell group remaining after excision of the primary tumor, which mutates while proliferating. Low antigenicity and tolerance to immunity during the quiescent period may explain the long duration of dormancy.6 More recently, there have been efforts to identify immunohistochemical signatures that may predict late recurrences, though the data are preliminary in nature.11
Given the latency period and location of the recurrence, our case demonstrates that even fully excised melanomas may recur locally many decades later, hence patients should be aware of the importance of a lifetime of vigilance after being diagnosed with melanoma.
- Tsao H, Cosimi AB, Sober AJ. Ultra-late recurrence (15 years or longer) of cutaneous melanoma. Cancer. 1997;79:2361-2370.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Mansour D, Kejariwal D. It is never too late: ultra-late recurrence of melanoma with distant metastases [published online March 8, 2012]. BMJ Case Rep. 2012:bcr0120125474. doi:10.1136/bcr.01.2012.5474
- Saleh D, Peach AHS. Ultra-late recurrence of malignant melanoma after 40 years of quiescent disease. J Surg Oncol. 2011;103:290-291.
- Goodenough J, Cozon CL, Liew SH. An incidental finding of a nodal recurrence of cutaneous malignant melanoma after a 45-year disease-free period [published online June 4, 2014]. BMJ Case Rep. 2014:bcr2014204289. doi:10.1136/bcr-2014-204289
- Nakamura M, Obayashi M, Yoshimitsu M, et al. Comparative whole-exome sequencing of an ultra-late recurrent malignant melanoma. Br J Dermatol. 2021;184:762-763.
- Miller JJ, Lofgren KA, Hughes SR, et al. Genomic analysis of melanoma evolution following a 30-year disease-free interval. J Cutan Pathol. 2017;44:805-808.
- North JP, Kageshita T, Pinkel D, et al. Distribution and significance of occult intraepidermal tumor cells surrounding primary melanoma. J Invest Dermatol. 2008;128:2024-2030.
- Massi G, LeBoit PE. Recurrent and persistent melanoma. In: Massi G, LeBoit PE, eds. Histological Diagnosis of Nevi and Melanoma. 2nd ed. Springer-Verlag; 2014:689-698.
- Bose A, Petsko GA, Eliezer D. Parkinson’s disease and melanoma: co-occurrence and mechanisms. J Parkinsons Dis. 2018;8:385-398.
- Reschke R, Dumann K, Ziemer M. Risk stratification and clinical characteristics of patients with late recurrence of melanoma (>10 years).J Clin Med. 2022;11:2026.
- Tsao H, Cosimi AB, Sober AJ. Ultra-late recurrence (15 years or longer) of cutaneous melanoma. Cancer. 1997;79:2361-2370.
- Faries MB, Steen S, Ye X, et al. Late recurrence in melanoma: clinical implications of lost dormancy. J Am Coll Surg. 2013;217:27-34.
- Mansour D, Kejariwal D. It is never too late: ultra-late recurrence of melanoma with distant metastases [published online March 8, 2012]. BMJ Case Rep. 2012:bcr0120125474. doi:10.1136/bcr.01.2012.5474
- Saleh D, Peach AHS. Ultra-late recurrence of malignant melanoma after 40 years of quiescent disease. J Surg Oncol. 2011;103:290-291.
- Goodenough J, Cozon CL, Liew SH. An incidental finding of a nodal recurrence of cutaneous malignant melanoma after a 45-year disease-free period [published online June 4, 2014]. BMJ Case Rep. 2014:bcr2014204289. doi:10.1136/bcr-2014-204289
- Nakamura M, Obayashi M, Yoshimitsu M, et al. Comparative whole-exome sequencing of an ultra-late recurrent malignant melanoma. Br J Dermatol. 2021;184:762-763.
- Miller JJ, Lofgren KA, Hughes SR, et al. Genomic analysis of melanoma evolution following a 30-year disease-free interval. J Cutan Pathol. 2017;44:805-808.
- North JP, Kageshita T, Pinkel D, et al. Distribution and significance of occult intraepidermal tumor cells surrounding primary melanoma. J Invest Dermatol. 2008;128:2024-2030.
- Massi G, LeBoit PE. Recurrent and persistent melanoma. In: Massi G, LeBoit PE, eds. Histological Diagnosis of Nevi and Melanoma. 2nd ed. Springer-Verlag; 2014:689-698.
- Bose A, Petsko GA, Eliezer D. Parkinson’s disease and melanoma: co-occurrence and mechanisms. J Parkinsons Dis. 2018;8:385-398.
- Reschke R, Dumann K, Ziemer M. Risk stratification and clinical characteristics of patients with late recurrence of melanoma (>10 years).J Clin Med. 2022;11:2026.
Practice Points
- In some cases of ultra-late malignant melanoma recurrence, the quiescent period can last more than 30 years.
- There does not appear to be specificity with location since ultra-late melanoma recurrences can occur locally, regionally, and distally, and original lesions appear to be randomly distributed in these cases.
- Mechanisms for ultra-late melanoma recurrence are poorly understood; histologically, unrecognizable aberrations in the skin beyond the histopathologic margins may represent an early phase of disease that lies dormant for many years before reemerging in response to external or immunologic changes.
- Patients with malignant melanoma are at a higher risk for developing Parkinson disease (and vice versa) given the link between melanin and neuromelanin pathways.
Disparities of Cutaneous Malignancies in the US Military
Occupational sun exposure is a well-known risk factor for the development of melanoma and nonmelanoma skin cancer (NMSC). In addition to sun exposure, US military personnel may face other risk factors such as lack of access to adequate sun protection, work in equatorial latitudes, and increased exposure to carcinogens. In one study, fewer than 30% of surveyed soldiers reported regular sunscreen use during deployment and reported the face, neck, and upper extremities were unprotected at least 70% of the time.1 Skin cancer risk factors that are more common in military service members include inadequate sunscreen access, insufficient sun protection, harsh weather conditions, more immediate safety concerns than sun protection, and male gender. A higher incidence of melanoma and NMSC has been correlated with the more common demographics of US veterans such as male sex, older age, and White race.2
Although not uncommon in both civilian and military populations, we present the case of a military service member who developed skin cancer at an early age potentially due to occupational sun exposure. We also provide a review of the literature to examine the risk factors and incidence of melanoma and NMSC in US military personnel and veterans and provide recommendations for skin cancer prevention, screening, and intervention in the military population.
Case Report
A 37-year-old White active-duty male service member in the US Navy (USN) presented with a nonhealing lesion on the nose of 2 years’ duration that had been gradually growing and bleeding for several weeks. He participated in several sea deployments while onboard a naval destroyer over his 10-year military career. He did not routinely use sunscreen during his deployments. His personal and family medical history lacked risk factors for skin cancer other than his skin tone and frequent sun exposure.
Physical examination revealed a 1-cm ulcerated plaque with rolled borders and prominent telangiectases on the mid nasal dorsum. A shave biopsy was performed to confirm the diagnosis of nodular basal cell carcinoma (BCC). The patient underwent Mohs micrographic surgery, which required repair with an advancement flap. He currently continues his active-duty service and is preparing for his next overseas deployment.
Literature Review
We conducted a review of PubMed articles indexed for MEDLINE using the search terms skin cancer, melanoma, nonmelanoma skin cancer, basal cell carcinoma, squamous cell carcinoma, keratoacanthoma, Merkel cell carcinoma, dermatofibrosarcoma protuberans, or sebaceous carcinoma along with military, Army, Navy, Air Force, or veterans. Studies from January 1984 to April 2020 were included in our qualitative review. All articles were reviewed, and those that did not examine skin cancer and the military population in the United States were excluded. Relevant data, such as results of skin cancer incidence or risk factors or insights about developing skin cancer in this affected population, were extracted from the selected publications.
Several studies showed overall increased age-adjusted incidence rates of melanoma and NMSC among military service personnel compared to age-matched controls in the general population.2 A survey of draft-age men during World War II found a slightly higher percentage of respondents with history of melanoma compared to the control group (83% [74/89] vs 76% [49/65]). Of those who had a history of melanoma, 34% (30/89) served in the tropics compared to 6% (4/65) in the control group.3 A tumor registry review found the age-adjusted melanoma incidence rates per 100,000 person-years for White individuals in the military vs the general population was 33.6 vs 27.5 among those aged 45 to 49 years, 49.8 vs 32.2 among those aged 50 to 54 years, and 178.5 vs 39.2 among those aged 55 to 59 years.4 Among published literature reviews, members of the US Air Force (USAF) had the highest rates of melanoma compared to other military branches, with an incidence rate of 7.6 vs 6.3 among USAF males vs Army males and 9.0 vs 5.5 among USAF females vs Army females.4 These findings were further supported by another study showing a higher incidence rate of melanoma in USAF members compared to Army personnel (17.8 vs 9.5) and a 62% greater melanoma incidence in active-duty military personnel compared to the general population when adjusted for age, race, sex, and year of diagnosis.5 Additionally, a meta-analysis reported a standardized incidence ratio of 1.4 (95% CI, 1.1-1.9) for malignant melanoma and 1.8 (95% CI, 1.3-2.8) for NMSC among military pilots compared to the general population.6 It is important to note that these data are limited to published peer-reviewed studies within PubMed and may not reflect the true skin cancer incidence.
More comprehensive studies are needed to compare NMSC incidence rates in nonpilot military populations compared to the general population. From 2005 to 2014, the average annual NMSC incidence rate in the USAF was 64.4 per 100,000 person-years, with the highest rate at 97.4 per 100,000 person-years in 2007.7 However, this study did not directly compare military service members to the general population. Service in tropical environments among World War II veterans was associated with an increased risk for NMSC. Sixty-six percent of patients with BCC (n=197) and 68% with squamous cell carcinoma (SCC)(n=41) were stationed in the Pacific, despite the number and demographics of soldiers deployed to the Pacific and Europe being approximately equal.8 During a 6-month period in 2008, a Combat Dermatology Clinic in Iraq showed 5% (n=129) of visits were for treatment of actinic keratoses (AKs), while 8% of visits (n=205) were related to skin cancer, including BCC, SCC, mycosis fungoides, and melanoma.9 Overall, these studies confirm a higher rate of melanoma in military service members vs the general population and indicate USAF members may be at the greatest risk for developing melanoma and NMSC among the service branches. Further studies are needed to elucidate why this might be the case and should concentrate on demographics, service locations, uniform wear and personal protective equipment standards, and use of sun-protective measures across each service branch.
Our search yielded no aggregate studies to determine if there is an increased rate of other types of skin cancer in military service members such as Merkel cell carcinoma, dermatofibrosarcoma protuberans, and microcystic adnexal carcinoma (MAC). Gerall et al10 described a case of MAC in a 43-year-old USAF U-2 pilot with a 15-year history of a slow-growing soft-tissue nodule on the cheek. The patient’s young age differed from the typical age of MAC occurrence (ie, 60–70 years), which led to the possibility that his profession contributed to the development of MAC and the relatively young age of onset.10
Etiology of Disease
The results of our literature review indicated that skin cancers are more prevalent among active-duty military personnel and veterans than in the general population; they also suggest that frequent sun exposure and lack of sun protection may be key etiologic factors. In 2015, only 23% of veterans (n=49) reported receiving skin cancer awareness education from the US Military.1 Among soldiers returning from Iraq and Afghanistan (n=212), only 13% reported routine sunscreen use, and
Exposure to UV radiation at higher altitudes (with corresponding higher UV energy) and altered sleep-wake cycles (with resulting altered immune defenses) may contribute to higher rates of melanoma and NMSC among USAF pilots.11 During a 57-minute flight at 30,000-ft altitude, a pilot is exposed to a UVA dose equivalent to 20 minutes inside a tanning booth.12 Although UVB transmission through plastic and glass windshields was reported to be less than 1%, UVA transmission ranged from 0.4% to 53.5%. The UVA dose for a pilot flying a light aircraft in Las Vegas, Nevada, was reported to be 127 μW/cm2 at ground level vs 242 μW/cm2 at a 30,000-ft altitude.12 Therefore, cosmic radiation exposure for military pilots is higher than for commercial pilots, as they fly at higher altitudes. U-2 pilots are exposed to 20 times the cosmic radiation dose at sea level and 10 times the exposure of commercial pilots.10
It currently is unknown why service in the USAF would increase skin cancer risk compared to service in other branches; however, there are some differences between military branches that require further research, including ethnic demographics, uniform wear and personal protective equipment standards, duty assignment locations, and the hours the military members are asked to work outside with direct sunlight exposure for each branch of service. Environmental exposures may differ based on the military branch gear requirements; for example, when on the flight line or flight deck, USN aircrews are required to wear cranials (helmets), eyewear (visor or goggles), and long-sleeved shirts. When at sea, USN flight crews wear gloves, headgear, goggles, pants, and long-sleeved shirts to identify their duty onboard. All of these measures offer good sun protection and are carried over to the land-based flight lines in the USN and Marine Corps. Neither the Army nor the USAF commonly utilize these practices. Conversely, the USAF does not allow flight line workers including fuelers, maintainers, and aircrew to wear coveralls due to the risk of being blown off, becoming foreign object debris, and being sucked into jet engines. However, in-flight protective gear such as goggles, gloves, and coveralls are worn.12 Perhaps the USAF may attract, recruit, or commission people with inherently more risk for skin cancer (eg, White individuals). How racial and ethnic factors may affect skin cancer incidence in military branches is an area for future research efforts.
Recommendations
Given the considerable increase in risk factors, efforts are needed to reduce the disparity in skin cancer rates between US military personnel and their civilian counterparts through appropriate prevention, screening, and intervention programs.
Prevention—In wartime settings as well as in training and other peacetime activities, active-duty military members cannot avoid harmful midday sun exposure. Additionally, application and reapplication of sunscreen can be challenging. Sunscreen, broad-spectrum lip balm, and wide-brimmed “boonie” hats can be ordered by supply personnel.13 We recommend that a standard sunscreen supply be available to all active-duty military service members. The long-sleeved, tightly woven fabric of military uniforms also can provide protection from the sun but can be difficult to tolerate for extended periods of time in warm climates. Breathable, lightweight, sun-protective clothing is commercially available and could be incorporated into military uniforms.
All service members should be educated about skin cancer risks while addressing common myths and inaccuracies. Fifty percent (n=50) of surveyed veterans thought discussions of skin cancer prevention and safety during basic training could help prevent skin cancer in service members.14 Suggestions from respondents included education about sun exposure consequences, use of graphic images of skin cancer in teaching, providing protective clothing and sunscreen to active-duty military service members, and discussion about sun protection with physicians during annual physicals. When veterans with a history of skin cancer were surveyed about their personal risk for skin cancer, most believed they were at little risk (average perceived risk response score, 2.2 out of 5 [1=no risk; 5=high risk]).14 The majority explained that they did not seek sun protection after warnings of skin cancer risk because they did not think skin cancer would happen to them,14 though the incidence of NMSC in the United States at the time of these surveys was estimated to be 3.5 million per year.14,15 Another study found that only 13% of veterans knew the back is the most common site of melanoma in men.1 The Army Public Health Center has informational fact sheets available online or in dermatologists’ offices that detail correct sunscreen application techniques and how to reduce sun exposure.16,17 However, military service members reported that they prefer physicians to communicate with them directly about skin cancer risks vs reading brochures in physician offices or gaining information from television, radio, military training, or the Internet (4.4 out 5 rating for communication methods of risks associated with skin cancer [1=ineffective; 5=very effective]).14 However, only 27% of nondermatologist physicians counseled or screened their patients on skin cancer or sunscreen yearly, 49% even less frequently, with 24% never counseling or screening at all. Because not all service members may be able to regularly see a dermatologist, efforts should be focused on increasing primary care physician awareness on counseling and screening.18
Early Detection—Military service members should be educated on how to perform skin self-examinations to alert their providers earlier to concerning lesions. The American Academy of Dermatology publishes infographics regarding the ABCDEs of melanoma and how to perform skin self-examinations.19,20 Although the US Preventive Services Task Force concluded there was insufficient evidence to recommend skin self-examination for all adults, the increased risk that military service members and veterans have requires further studies to examine the utility of self-screening in this population.20 Given the evidence of a higher incidence of melanoma in military service members vs the general population after 45 years of age,4 we recommend starting yearly in-person screenings performed by primary care physicians or dermatologists at this age. Ensuring every service member has routine in-office skin examinations can be difficult given the limited number of active-duty military dermatologists. Civilian dermatologists also could be helpful in this respect.
Teleconsultation, teledermoscopy, or store-and-forward imaging services for concerning lesions could be utilized when in-person consultations with a dermatologist are not feasible or cannot be performed in a timely manner. From 2004 to 2012, 40% of 10,817 teleconsultations were dermatology consultations from deployed or remote environments.21 Teleconsultation can be performed via email through the global military teleconsultation portal.22 These methods can lead to earlier detection of skin cancer rather than delaying evaluation for an in-person consultation.23
Intervention—High-risk patients who have been diagnosed with NMSC or many AKs should consider oral, procedural, or topical chemoprevention to reduce the risk for additional skin cancers as both primary and secondary prevention. In a double-blind, randomized, controlled trial of 386 individuals with a history of 2 or more NMSCs, participants were randomly assigned to receive either 500 mg of nicotinamide twice daily or placebo for 12 months. Compared to the placebo group, the nicotinamide group had a 23% lower rate of new NMSCs and an 11% lower rate of new AKs at 12 months.24 The use of acitretin also has been studied in transplant recipients for the chemoprevention of NMSC. In a double-blind, randomized, controlled trial of renal transplant recipients with more than 10 AKs randomized to receive either 30 mg/d of acitretin or placebo for 6 months, 11% of the acitretin group reported a new NMSC compared to 47% in the placebo group.25 An open-label study of 27 renal transplant recipients treated with methyl-esterified aminolevulinic acid–photodynamic therapy and red light demonstrated an increased mean time to occurrence of an AK, SCC, BCC, keratoacanthoma, or wart from 6.8 months in untreated areas compared to 9.6 months in treated areas.25 In active-duty locations where access to red and blue light sources is unavailable, the use of daylight photodynamic therapy can be considered, as it does not require any special equipment. Topical treatments such as 5-fluorouracil and imiquimod can be used for treatment and chemoprevention of NMSC. In a follow-up study from the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial, patients who applied 5-fluorouracil cream 5% twice daily to the face and ears for 4 weeks had a 75% risk reduction in developing SCC requiring surgery compared to the control group for the first year after treatment.26,27
Final Thoughts
Focusing on the efforts we propose can help the US Military expand their prevention, screening, and intervention programs for skin cancer in service members. Further research can then be performed to determine which programs have the greatest impact on rates of skin cancer among military and veteran personnel. Given these higher incidences and risk of exposure for skin cancer among service members, the various services may consider mandating sunscreen use as part of the uniform to prevent skin cancer. To maximize effectiveness, these efforts to prevent the development of skin cancer among military and veteran personnel should be adopted nationally.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192.
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol Biomarkers Prev. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179:247-253.
- Sanlorenzo M, Vujic I, Posch C, et al. The risk of melanoma in pilots and cabin crew: UV measurements in flying airplanes. JAMA Dermatol. 2015;151:450-452.
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737.
- Henning JS, Firoz, BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Gerall CD, Sippel MR, Yracheta JL, et al. Microcystic adnexal carcinoma: a rare, commonly misdiagnosed malignancy. Mil Med. 2019;184:948-950.
- Wilkison B, Wong E. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Proctor SP, Heaton KJ, Smith KW, et al. The Occupational JP8 Neuroepidemiology Study (OJENES): repeated workday exposure and central nervous system functioning among US Air Force personnel. Neurotoxicology. 2011;32:799-808.
- Soldiers protect themselves from skin cancer. US Army website. Published February 28, 2019. Accessed August 21, 2022. https://www.army.mil/article/17601/soldiers_protect_themselves_from_skin_cancer
- Fisher V, Lee D, McGrath J, et al. Veterans speak up: current warnings on skin cancer miss the target, suggestions for improvement. Mil Med. 2015;180:892-897.
- Rogers HW, Weinstick MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Sun safety. Army Public Health Center website. Updated June 6, 2019. Accessed August 21, 2022. https://phc.amedd.army.mil/topics/discond/hipss/Pages/Sun-Safety.aspx
- Outdoor ultraviolet radiation hazards and protection. Army Public Health Center website. Accessed August 21, 2022. https://phc.amedd.army.mil/PHC%20Resource%20Library/OutdoorUltravioletRadiationHazardsandProtection_FS_24-017-1115.pdf
- Saraiya M, Frank E, Elon L, et al. Personal and clinical skin cancer prevention practices of US women physicians. Arch Dermatol. 2000;136:633-642.
- What to look for: ABCDEs of melanoma. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/at-risk/abcdes
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/check-skin
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the US military in a deployed setting. Mil Med. 2014;179:1347-1353.
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:2034-2039.
- Day WG, Shirvastava V, Roman JW. Synchronous teledermoscopy in military treatment facilities. Mil Med. 2020;185:1334-1337.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- Wulf HC, Pavel S, Stender I, et al. Topical photodynamic therapy for prevention of new skin lesions in renal transplant recipients. Acta Derm Venereol. 2006;86:25-28.
- Weinstock MA, Thwin SS, Siegel JA, et al; Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCC) Group. Chemoprevention of basal and squamous cell carcinoma with a single course of fluorouracil, 5%, cream: a randomized clinical trial. JAMA Dermatol. 2018;154:167-174.
Occupational sun exposure is a well-known risk factor for the development of melanoma and nonmelanoma skin cancer (NMSC). In addition to sun exposure, US military personnel may face other risk factors such as lack of access to adequate sun protection, work in equatorial latitudes, and increased exposure to carcinogens. In one study, fewer than 30% of surveyed soldiers reported regular sunscreen use during deployment and reported the face, neck, and upper extremities were unprotected at least 70% of the time.1 Skin cancer risk factors that are more common in military service members include inadequate sunscreen access, insufficient sun protection, harsh weather conditions, more immediate safety concerns than sun protection, and male gender. A higher incidence of melanoma and NMSC has been correlated with the more common demographics of US veterans such as male sex, older age, and White race.2
Although not uncommon in both civilian and military populations, we present the case of a military service member who developed skin cancer at an early age potentially due to occupational sun exposure. We also provide a review of the literature to examine the risk factors and incidence of melanoma and NMSC in US military personnel and veterans and provide recommendations for skin cancer prevention, screening, and intervention in the military population.
Case Report
A 37-year-old White active-duty male service member in the US Navy (USN) presented with a nonhealing lesion on the nose of 2 years’ duration that had been gradually growing and bleeding for several weeks. He participated in several sea deployments while onboard a naval destroyer over his 10-year military career. He did not routinely use sunscreen during his deployments. His personal and family medical history lacked risk factors for skin cancer other than his skin tone and frequent sun exposure.
Physical examination revealed a 1-cm ulcerated plaque with rolled borders and prominent telangiectases on the mid nasal dorsum. A shave biopsy was performed to confirm the diagnosis of nodular basal cell carcinoma (BCC). The patient underwent Mohs micrographic surgery, which required repair with an advancement flap. He currently continues his active-duty service and is preparing for his next overseas deployment.
Literature Review
We conducted a review of PubMed articles indexed for MEDLINE using the search terms skin cancer, melanoma, nonmelanoma skin cancer, basal cell carcinoma, squamous cell carcinoma, keratoacanthoma, Merkel cell carcinoma, dermatofibrosarcoma protuberans, or sebaceous carcinoma along with military, Army, Navy, Air Force, or veterans. Studies from January 1984 to April 2020 were included in our qualitative review. All articles were reviewed, and those that did not examine skin cancer and the military population in the United States were excluded. Relevant data, such as results of skin cancer incidence or risk factors or insights about developing skin cancer in this affected population, were extracted from the selected publications.
Several studies showed overall increased age-adjusted incidence rates of melanoma and NMSC among military service personnel compared to age-matched controls in the general population.2 A survey of draft-age men during World War II found a slightly higher percentage of respondents with history of melanoma compared to the control group (83% [74/89] vs 76% [49/65]). Of those who had a history of melanoma, 34% (30/89) served in the tropics compared to 6% (4/65) in the control group.3 A tumor registry review found the age-adjusted melanoma incidence rates per 100,000 person-years for White individuals in the military vs the general population was 33.6 vs 27.5 among those aged 45 to 49 years, 49.8 vs 32.2 among those aged 50 to 54 years, and 178.5 vs 39.2 among those aged 55 to 59 years.4 Among published literature reviews, members of the US Air Force (USAF) had the highest rates of melanoma compared to other military branches, with an incidence rate of 7.6 vs 6.3 among USAF males vs Army males and 9.0 vs 5.5 among USAF females vs Army females.4 These findings were further supported by another study showing a higher incidence rate of melanoma in USAF members compared to Army personnel (17.8 vs 9.5) and a 62% greater melanoma incidence in active-duty military personnel compared to the general population when adjusted for age, race, sex, and year of diagnosis.5 Additionally, a meta-analysis reported a standardized incidence ratio of 1.4 (95% CI, 1.1-1.9) for malignant melanoma and 1.8 (95% CI, 1.3-2.8) for NMSC among military pilots compared to the general population.6 It is important to note that these data are limited to published peer-reviewed studies within PubMed and may not reflect the true skin cancer incidence.
More comprehensive studies are needed to compare NMSC incidence rates in nonpilot military populations compared to the general population. From 2005 to 2014, the average annual NMSC incidence rate in the USAF was 64.4 per 100,000 person-years, with the highest rate at 97.4 per 100,000 person-years in 2007.7 However, this study did not directly compare military service members to the general population. Service in tropical environments among World War II veterans was associated with an increased risk for NMSC. Sixty-six percent of patients with BCC (n=197) and 68% with squamous cell carcinoma (SCC)(n=41) were stationed in the Pacific, despite the number and demographics of soldiers deployed to the Pacific and Europe being approximately equal.8 During a 6-month period in 2008, a Combat Dermatology Clinic in Iraq showed 5% (n=129) of visits were for treatment of actinic keratoses (AKs), while 8% of visits (n=205) were related to skin cancer, including BCC, SCC, mycosis fungoides, and melanoma.9 Overall, these studies confirm a higher rate of melanoma in military service members vs the general population and indicate USAF members may be at the greatest risk for developing melanoma and NMSC among the service branches. Further studies are needed to elucidate why this might be the case and should concentrate on demographics, service locations, uniform wear and personal protective equipment standards, and use of sun-protective measures across each service branch.
Our search yielded no aggregate studies to determine if there is an increased rate of other types of skin cancer in military service members such as Merkel cell carcinoma, dermatofibrosarcoma protuberans, and microcystic adnexal carcinoma (MAC). Gerall et al10 described a case of MAC in a 43-year-old USAF U-2 pilot with a 15-year history of a slow-growing soft-tissue nodule on the cheek. The patient’s young age differed from the typical age of MAC occurrence (ie, 60–70 years), which led to the possibility that his profession contributed to the development of MAC and the relatively young age of onset.10
Etiology of Disease
The results of our literature review indicated that skin cancers are more prevalent among active-duty military personnel and veterans than in the general population; they also suggest that frequent sun exposure and lack of sun protection may be key etiologic factors. In 2015, only 23% of veterans (n=49) reported receiving skin cancer awareness education from the US Military.1 Among soldiers returning from Iraq and Afghanistan (n=212), only 13% reported routine sunscreen use, and
Exposure to UV radiation at higher altitudes (with corresponding higher UV energy) and altered sleep-wake cycles (with resulting altered immune defenses) may contribute to higher rates of melanoma and NMSC among USAF pilots.11 During a 57-minute flight at 30,000-ft altitude, a pilot is exposed to a UVA dose equivalent to 20 minutes inside a tanning booth.12 Although UVB transmission through plastic and glass windshields was reported to be less than 1%, UVA transmission ranged from 0.4% to 53.5%. The UVA dose for a pilot flying a light aircraft in Las Vegas, Nevada, was reported to be 127 μW/cm2 at ground level vs 242 μW/cm2 at a 30,000-ft altitude.12 Therefore, cosmic radiation exposure for military pilots is higher than for commercial pilots, as they fly at higher altitudes. U-2 pilots are exposed to 20 times the cosmic radiation dose at sea level and 10 times the exposure of commercial pilots.10
It currently is unknown why service in the USAF would increase skin cancer risk compared to service in other branches; however, there are some differences between military branches that require further research, including ethnic demographics, uniform wear and personal protective equipment standards, duty assignment locations, and the hours the military members are asked to work outside with direct sunlight exposure for each branch of service. Environmental exposures may differ based on the military branch gear requirements; for example, when on the flight line or flight deck, USN aircrews are required to wear cranials (helmets), eyewear (visor or goggles), and long-sleeved shirts. When at sea, USN flight crews wear gloves, headgear, goggles, pants, and long-sleeved shirts to identify their duty onboard. All of these measures offer good sun protection and are carried over to the land-based flight lines in the USN and Marine Corps. Neither the Army nor the USAF commonly utilize these practices. Conversely, the USAF does not allow flight line workers including fuelers, maintainers, and aircrew to wear coveralls due to the risk of being blown off, becoming foreign object debris, and being sucked into jet engines. However, in-flight protective gear such as goggles, gloves, and coveralls are worn.12 Perhaps the USAF may attract, recruit, or commission people with inherently more risk for skin cancer (eg, White individuals). How racial and ethnic factors may affect skin cancer incidence in military branches is an area for future research efforts.
Recommendations
Given the considerable increase in risk factors, efforts are needed to reduce the disparity in skin cancer rates between US military personnel and their civilian counterparts through appropriate prevention, screening, and intervention programs.
Prevention—In wartime settings as well as in training and other peacetime activities, active-duty military members cannot avoid harmful midday sun exposure. Additionally, application and reapplication of sunscreen can be challenging. Sunscreen, broad-spectrum lip balm, and wide-brimmed “boonie” hats can be ordered by supply personnel.13 We recommend that a standard sunscreen supply be available to all active-duty military service members. The long-sleeved, tightly woven fabric of military uniforms also can provide protection from the sun but can be difficult to tolerate for extended periods of time in warm climates. Breathable, lightweight, sun-protective clothing is commercially available and could be incorporated into military uniforms.
All service members should be educated about skin cancer risks while addressing common myths and inaccuracies. Fifty percent (n=50) of surveyed veterans thought discussions of skin cancer prevention and safety during basic training could help prevent skin cancer in service members.14 Suggestions from respondents included education about sun exposure consequences, use of graphic images of skin cancer in teaching, providing protective clothing and sunscreen to active-duty military service members, and discussion about sun protection with physicians during annual physicals. When veterans with a history of skin cancer were surveyed about their personal risk for skin cancer, most believed they were at little risk (average perceived risk response score, 2.2 out of 5 [1=no risk; 5=high risk]).14 The majority explained that they did not seek sun protection after warnings of skin cancer risk because they did not think skin cancer would happen to them,14 though the incidence of NMSC in the United States at the time of these surveys was estimated to be 3.5 million per year.14,15 Another study found that only 13% of veterans knew the back is the most common site of melanoma in men.1 The Army Public Health Center has informational fact sheets available online or in dermatologists’ offices that detail correct sunscreen application techniques and how to reduce sun exposure.16,17 However, military service members reported that they prefer physicians to communicate with them directly about skin cancer risks vs reading brochures in physician offices or gaining information from television, radio, military training, or the Internet (4.4 out 5 rating for communication methods of risks associated with skin cancer [1=ineffective; 5=very effective]).14 However, only 27% of nondermatologist physicians counseled or screened their patients on skin cancer or sunscreen yearly, 49% even less frequently, with 24% never counseling or screening at all. Because not all service members may be able to regularly see a dermatologist, efforts should be focused on increasing primary care physician awareness on counseling and screening.18
Early Detection—Military service members should be educated on how to perform skin self-examinations to alert their providers earlier to concerning lesions. The American Academy of Dermatology publishes infographics regarding the ABCDEs of melanoma and how to perform skin self-examinations.19,20 Although the US Preventive Services Task Force concluded there was insufficient evidence to recommend skin self-examination for all adults, the increased risk that military service members and veterans have requires further studies to examine the utility of self-screening in this population.20 Given the evidence of a higher incidence of melanoma in military service members vs the general population after 45 years of age,4 we recommend starting yearly in-person screenings performed by primary care physicians or dermatologists at this age. Ensuring every service member has routine in-office skin examinations can be difficult given the limited number of active-duty military dermatologists. Civilian dermatologists also could be helpful in this respect.
Teleconsultation, teledermoscopy, or store-and-forward imaging services for concerning lesions could be utilized when in-person consultations with a dermatologist are not feasible or cannot be performed in a timely manner. From 2004 to 2012, 40% of 10,817 teleconsultations were dermatology consultations from deployed or remote environments.21 Teleconsultation can be performed via email through the global military teleconsultation portal.22 These methods can lead to earlier detection of skin cancer rather than delaying evaluation for an in-person consultation.23
Intervention—High-risk patients who have been diagnosed with NMSC or many AKs should consider oral, procedural, or topical chemoprevention to reduce the risk for additional skin cancers as both primary and secondary prevention. In a double-blind, randomized, controlled trial of 386 individuals with a history of 2 or more NMSCs, participants were randomly assigned to receive either 500 mg of nicotinamide twice daily or placebo for 12 months. Compared to the placebo group, the nicotinamide group had a 23% lower rate of new NMSCs and an 11% lower rate of new AKs at 12 months.24 The use of acitretin also has been studied in transplant recipients for the chemoprevention of NMSC. In a double-blind, randomized, controlled trial of renal transplant recipients with more than 10 AKs randomized to receive either 30 mg/d of acitretin or placebo for 6 months, 11% of the acitretin group reported a new NMSC compared to 47% in the placebo group.25 An open-label study of 27 renal transplant recipients treated with methyl-esterified aminolevulinic acid–photodynamic therapy and red light demonstrated an increased mean time to occurrence of an AK, SCC, BCC, keratoacanthoma, or wart from 6.8 months in untreated areas compared to 9.6 months in treated areas.25 In active-duty locations where access to red and blue light sources is unavailable, the use of daylight photodynamic therapy can be considered, as it does not require any special equipment. Topical treatments such as 5-fluorouracil and imiquimod can be used for treatment and chemoprevention of NMSC. In a follow-up study from the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial, patients who applied 5-fluorouracil cream 5% twice daily to the face and ears for 4 weeks had a 75% risk reduction in developing SCC requiring surgery compared to the control group for the first year after treatment.26,27
Final Thoughts
Focusing on the efforts we propose can help the US Military expand their prevention, screening, and intervention programs for skin cancer in service members. Further research can then be performed to determine which programs have the greatest impact on rates of skin cancer among military and veteran personnel. Given these higher incidences and risk of exposure for skin cancer among service members, the various services may consider mandating sunscreen use as part of the uniform to prevent skin cancer. To maximize effectiveness, these efforts to prevent the development of skin cancer among military and veteran personnel should be adopted nationally.
Occupational sun exposure is a well-known risk factor for the development of melanoma and nonmelanoma skin cancer (NMSC). In addition to sun exposure, US military personnel may face other risk factors such as lack of access to adequate sun protection, work in equatorial latitudes, and increased exposure to carcinogens. In one study, fewer than 30% of surveyed soldiers reported regular sunscreen use during deployment and reported the face, neck, and upper extremities were unprotected at least 70% of the time.1 Skin cancer risk factors that are more common in military service members include inadequate sunscreen access, insufficient sun protection, harsh weather conditions, more immediate safety concerns than sun protection, and male gender. A higher incidence of melanoma and NMSC has been correlated with the more common demographics of US veterans such as male sex, older age, and White race.2
Although not uncommon in both civilian and military populations, we present the case of a military service member who developed skin cancer at an early age potentially due to occupational sun exposure. We also provide a review of the literature to examine the risk factors and incidence of melanoma and NMSC in US military personnel and veterans and provide recommendations for skin cancer prevention, screening, and intervention in the military population.
Case Report
A 37-year-old White active-duty male service member in the US Navy (USN) presented with a nonhealing lesion on the nose of 2 years’ duration that had been gradually growing and bleeding for several weeks. He participated in several sea deployments while onboard a naval destroyer over his 10-year military career. He did not routinely use sunscreen during his deployments. His personal and family medical history lacked risk factors for skin cancer other than his skin tone and frequent sun exposure.
Physical examination revealed a 1-cm ulcerated plaque with rolled borders and prominent telangiectases on the mid nasal dorsum. A shave biopsy was performed to confirm the diagnosis of nodular basal cell carcinoma (BCC). The patient underwent Mohs micrographic surgery, which required repair with an advancement flap. He currently continues his active-duty service and is preparing for his next overseas deployment.
Literature Review
We conducted a review of PubMed articles indexed for MEDLINE using the search terms skin cancer, melanoma, nonmelanoma skin cancer, basal cell carcinoma, squamous cell carcinoma, keratoacanthoma, Merkel cell carcinoma, dermatofibrosarcoma protuberans, or sebaceous carcinoma along with military, Army, Navy, Air Force, or veterans. Studies from January 1984 to April 2020 were included in our qualitative review. All articles were reviewed, and those that did not examine skin cancer and the military population in the United States were excluded. Relevant data, such as results of skin cancer incidence or risk factors or insights about developing skin cancer in this affected population, were extracted from the selected publications.
Several studies showed overall increased age-adjusted incidence rates of melanoma and NMSC among military service personnel compared to age-matched controls in the general population.2 A survey of draft-age men during World War II found a slightly higher percentage of respondents with history of melanoma compared to the control group (83% [74/89] vs 76% [49/65]). Of those who had a history of melanoma, 34% (30/89) served in the tropics compared to 6% (4/65) in the control group.3 A tumor registry review found the age-adjusted melanoma incidence rates per 100,000 person-years for White individuals in the military vs the general population was 33.6 vs 27.5 among those aged 45 to 49 years, 49.8 vs 32.2 among those aged 50 to 54 years, and 178.5 vs 39.2 among those aged 55 to 59 years.4 Among published literature reviews, members of the US Air Force (USAF) had the highest rates of melanoma compared to other military branches, with an incidence rate of 7.6 vs 6.3 among USAF males vs Army males and 9.0 vs 5.5 among USAF females vs Army females.4 These findings were further supported by another study showing a higher incidence rate of melanoma in USAF members compared to Army personnel (17.8 vs 9.5) and a 62% greater melanoma incidence in active-duty military personnel compared to the general population when adjusted for age, race, sex, and year of diagnosis.5 Additionally, a meta-analysis reported a standardized incidence ratio of 1.4 (95% CI, 1.1-1.9) for malignant melanoma and 1.8 (95% CI, 1.3-2.8) for NMSC among military pilots compared to the general population.6 It is important to note that these data are limited to published peer-reviewed studies within PubMed and may not reflect the true skin cancer incidence.
More comprehensive studies are needed to compare NMSC incidence rates in nonpilot military populations compared to the general population. From 2005 to 2014, the average annual NMSC incidence rate in the USAF was 64.4 per 100,000 person-years, with the highest rate at 97.4 per 100,000 person-years in 2007.7 However, this study did not directly compare military service members to the general population. Service in tropical environments among World War II veterans was associated with an increased risk for NMSC. Sixty-six percent of patients with BCC (n=197) and 68% with squamous cell carcinoma (SCC)(n=41) were stationed in the Pacific, despite the number and demographics of soldiers deployed to the Pacific and Europe being approximately equal.8 During a 6-month period in 2008, a Combat Dermatology Clinic in Iraq showed 5% (n=129) of visits were for treatment of actinic keratoses (AKs), while 8% of visits (n=205) were related to skin cancer, including BCC, SCC, mycosis fungoides, and melanoma.9 Overall, these studies confirm a higher rate of melanoma in military service members vs the general population and indicate USAF members may be at the greatest risk for developing melanoma and NMSC among the service branches. Further studies are needed to elucidate why this might be the case and should concentrate on demographics, service locations, uniform wear and personal protective equipment standards, and use of sun-protective measures across each service branch.
Our search yielded no aggregate studies to determine if there is an increased rate of other types of skin cancer in military service members such as Merkel cell carcinoma, dermatofibrosarcoma protuberans, and microcystic adnexal carcinoma (MAC). Gerall et al10 described a case of MAC in a 43-year-old USAF U-2 pilot with a 15-year history of a slow-growing soft-tissue nodule on the cheek. The patient’s young age differed from the typical age of MAC occurrence (ie, 60–70 years), which led to the possibility that his profession contributed to the development of MAC and the relatively young age of onset.10
Etiology of Disease
The results of our literature review indicated that skin cancers are more prevalent among active-duty military personnel and veterans than in the general population; they also suggest that frequent sun exposure and lack of sun protection may be key etiologic factors. In 2015, only 23% of veterans (n=49) reported receiving skin cancer awareness education from the US Military.1 Among soldiers returning from Iraq and Afghanistan (n=212), only 13% reported routine sunscreen use, and
Exposure to UV radiation at higher altitudes (with corresponding higher UV energy) and altered sleep-wake cycles (with resulting altered immune defenses) may contribute to higher rates of melanoma and NMSC among USAF pilots.11 During a 57-minute flight at 30,000-ft altitude, a pilot is exposed to a UVA dose equivalent to 20 minutes inside a tanning booth.12 Although UVB transmission through plastic and glass windshields was reported to be less than 1%, UVA transmission ranged from 0.4% to 53.5%. The UVA dose for a pilot flying a light aircraft in Las Vegas, Nevada, was reported to be 127 μW/cm2 at ground level vs 242 μW/cm2 at a 30,000-ft altitude.12 Therefore, cosmic radiation exposure for military pilots is higher than for commercial pilots, as they fly at higher altitudes. U-2 pilots are exposed to 20 times the cosmic radiation dose at sea level and 10 times the exposure of commercial pilots.10
It currently is unknown why service in the USAF would increase skin cancer risk compared to service in other branches; however, there are some differences between military branches that require further research, including ethnic demographics, uniform wear and personal protective equipment standards, duty assignment locations, and the hours the military members are asked to work outside with direct sunlight exposure for each branch of service. Environmental exposures may differ based on the military branch gear requirements; for example, when on the flight line or flight deck, USN aircrews are required to wear cranials (helmets), eyewear (visor or goggles), and long-sleeved shirts. When at sea, USN flight crews wear gloves, headgear, goggles, pants, and long-sleeved shirts to identify their duty onboard. All of these measures offer good sun protection and are carried over to the land-based flight lines in the USN and Marine Corps. Neither the Army nor the USAF commonly utilize these practices. Conversely, the USAF does not allow flight line workers including fuelers, maintainers, and aircrew to wear coveralls due to the risk of being blown off, becoming foreign object debris, and being sucked into jet engines. However, in-flight protective gear such as goggles, gloves, and coveralls are worn.12 Perhaps the USAF may attract, recruit, or commission people with inherently more risk for skin cancer (eg, White individuals). How racial and ethnic factors may affect skin cancer incidence in military branches is an area for future research efforts.
Recommendations
Given the considerable increase in risk factors, efforts are needed to reduce the disparity in skin cancer rates between US military personnel and their civilian counterparts through appropriate prevention, screening, and intervention programs.
Prevention—In wartime settings as well as in training and other peacetime activities, active-duty military members cannot avoid harmful midday sun exposure. Additionally, application and reapplication of sunscreen can be challenging. Sunscreen, broad-spectrum lip balm, and wide-brimmed “boonie” hats can be ordered by supply personnel.13 We recommend that a standard sunscreen supply be available to all active-duty military service members. The long-sleeved, tightly woven fabric of military uniforms also can provide protection from the sun but can be difficult to tolerate for extended periods of time in warm climates. Breathable, lightweight, sun-protective clothing is commercially available and could be incorporated into military uniforms.
All service members should be educated about skin cancer risks while addressing common myths and inaccuracies. Fifty percent (n=50) of surveyed veterans thought discussions of skin cancer prevention and safety during basic training could help prevent skin cancer in service members.14 Suggestions from respondents included education about sun exposure consequences, use of graphic images of skin cancer in teaching, providing protective clothing and sunscreen to active-duty military service members, and discussion about sun protection with physicians during annual physicals. When veterans with a history of skin cancer were surveyed about their personal risk for skin cancer, most believed they were at little risk (average perceived risk response score, 2.2 out of 5 [1=no risk; 5=high risk]).14 The majority explained that they did not seek sun protection after warnings of skin cancer risk because they did not think skin cancer would happen to them,14 though the incidence of NMSC in the United States at the time of these surveys was estimated to be 3.5 million per year.14,15 Another study found that only 13% of veterans knew the back is the most common site of melanoma in men.1 The Army Public Health Center has informational fact sheets available online or in dermatologists’ offices that detail correct sunscreen application techniques and how to reduce sun exposure.16,17 However, military service members reported that they prefer physicians to communicate with them directly about skin cancer risks vs reading brochures in physician offices or gaining information from television, radio, military training, or the Internet (4.4 out 5 rating for communication methods of risks associated with skin cancer [1=ineffective; 5=very effective]).14 However, only 27% of nondermatologist physicians counseled or screened their patients on skin cancer or sunscreen yearly, 49% even less frequently, with 24% never counseling or screening at all. Because not all service members may be able to regularly see a dermatologist, efforts should be focused on increasing primary care physician awareness on counseling and screening.18
Early Detection—Military service members should be educated on how to perform skin self-examinations to alert their providers earlier to concerning lesions. The American Academy of Dermatology publishes infographics regarding the ABCDEs of melanoma and how to perform skin self-examinations.19,20 Although the US Preventive Services Task Force concluded there was insufficient evidence to recommend skin self-examination for all adults, the increased risk that military service members and veterans have requires further studies to examine the utility of self-screening in this population.20 Given the evidence of a higher incidence of melanoma in military service members vs the general population after 45 years of age,4 we recommend starting yearly in-person screenings performed by primary care physicians or dermatologists at this age. Ensuring every service member has routine in-office skin examinations can be difficult given the limited number of active-duty military dermatologists. Civilian dermatologists also could be helpful in this respect.
Teleconsultation, teledermoscopy, or store-and-forward imaging services for concerning lesions could be utilized when in-person consultations with a dermatologist are not feasible or cannot be performed in a timely manner. From 2004 to 2012, 40% of 10,817 teleconsultations were dermatology consultations from deployed or remote environments.21 Teleconsultation can be performed via email through the global military teleconsultation portal.22 These methods can lead to earlier detection of skin cancer rather than delaying evaluation for an in-person consultation.23
Intervention—High-risk patients who have been diagnosed with NMSC or many AKs should consider oral, procedural, or topical chemoprevention to reduce the risk for additional skin cancers as both primary and secondary prevention. In a double-blind, randomized, controlled trial of 386 individuals with a history of 2 or more NMSCs, participants were randomly assigned to receive either 500 mg of nicotinamide twice daily or placebo for 12 months. Compared to the placebo group, the nicotinamide group had a 23% lower rate of new NMSCs and an 11% lower rate of new AKs at 12 months.24 The use of acitretin also has been studied in transplant recipients for the chemoprevention of NMSC. In a double-blind, randomized, controlled trial of renal transplant recipients with more than 10 AKs randomized to receive either 30 mg/d of acitretin or placebo for 6 months, 11% of the acitretin group reported a new NMSC compared to 47% in the placebo group.25 An open-label study of 27 renal transplant recipients treated with methyl-esterified aminolevulinic acid–photodynamic therapy and red light demonstrated an increased mean time to occurrence of an AK, SCC, BCC, keratoacanthoma, or wart from 6.8 months in untreated areas compared to 9.6 months in treated areas.25 In active-duty locations where access to red and blue light sources is unavailable, the use of daylight photodynamic therapy can be considered, as it does not require any special equipment. Topical treatments such as 5-fluorouracil and imiquimod can be used for treatment and chemoprevention of NMSC. In a follow-up study from the Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial, patients who applied 5-fluorouracil cream 5% twice daily to the face and ears for 4 weeks had a 75% risk reduction in developing SCC requiring surgery compared to the control group for the first year after treatment.26,27
Final Thoughts
Focusing on the efforts we propose can help the US Military expand their prevention, screening, and intervention programs for skin cancer in service members. Further research can then be performed to determine which programs have the greatest impact on rates of skin cancer among military and veteran personnel. Given these higher incidences and risk of exposure for skin cancer among service members, the various services may consider mandating sunscreen use as part of the uniform to prevent skin cancer. To maximize effectiveness, these efforts to prevent the development of skin cancer among military and veteran personnel should be adopted nationally.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192.
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol Biomarkers Prev. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179:247-253.
- Sanlorenzo M, Vujic I, Posch C, et al. The risk of melanoma in pilots and cabin crew: UV measurements in flying airplanes. JAMA Dermatol. 2015;151:450-452.
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737.
- Henning JS, Firoz, BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Gerall CD, Sippel MR, Yracheta JL, et al. Microcystic adnexal carcinoma: a rare, commonly misdiagnosed malignancy. Mil Med. 2019;184:948-950.
- Wilkison B, Wong E. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Proctor SP, Heaton KJ, Smith KW, et al. The Occupational JP8 Neuroepidemiology Study (OJENES): repeated workday exposure and central nervous system functioning among US Air Force personnel. Neurotoxicology. 2011;32:799-808.
- Soldiers protect themselves from skin cancer. US Army website. Published February 28, 2019. Accessed August 21, 2022. https://www.army.mil/article/17601/soldiers_protect_themselves_from_skin_cancer
- Fisher V, Lee D, McGrath J, et al. Veterans speak up: current warnings on skin cancer miss the target, suggestions for improvement. Mil Med. 2015;180:892-897.
- Rogers HW, Weinstick MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Sun safety. Army Public Health Center website. Updated June 6, 2019. Accessed August 21, 2022. https://phc.amedd.army.mil/topics/discond/hipss/Pages/Sun-Safety.aspx
- Outdoor ultraviolet radiation hazards and protection. Army Public Health Center website. Accessed August 21, 2022. https://phc.amedd.army.mil/PHC%20Resource%20Library/OutdoorUltravioletRadiationHazardsandProtection_FS_24-017-1115.pdf
- Saraiya M, Frank E, Elon L, et al. Personal and clinical skin cancer prevention practices of US women physicians. Arch Dermatol. 2000;136:633-642.
- What to look for: ABCDEs of melanoma. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/at-risk/abcdes
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/check-skin
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the US military in a deployed setting. Mil Med. 2014;179:1347-1353.
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:2034-2039.
- Day WG, Shirvastava V, Roman JW. Synchronous teledermoscopy in military treatment facilities. Mil Med. 2020;185:1334-1337.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- Wulf HC, Pavel S, Stender I, et al. Topical photodynamic therapy for prevention of new skin lesions in renal transplant recipients. Acta Derm Venereol. 2006;86:25-28.
- Weinstock MA, Thwin SS, Siegel JA, et al; Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCC) Group. Chemoprevention of basal and squamous cell carcinoma with a single course of fluorouracil, 5%, cream: a randomized clinical trial. JAMA Dermatol. 2018;154:167-174.
- Powers JG, Patel NA, Powers EM, et al. Skin cancer risk factors and preventative behaviors among United States military veterans deployed to Iraq and Afghanistan. J Invest Dermatol. 2015;135:2871-2873.
- Riemenschneider K, Liu J, Powers JG. Skin cancer in the military: a systematic review of melanoma and nonmelanoma skin cancer incidence, prevention, and screening among active duty and veteran personnel. J Am Acad Dermatol. 2018;78:1185-1192.
- Brown J, Kopf AW, Rigel DS, et al. Malignant melanoma in World War II veterans. Int J Dermatol. 1984;23:661-663.
- Zhou J, Enewold L, Zahm SH, et al. Melanoma incidence rates among whites in the U.S. Military. Cancer Epidemiol Biomarkers Prev. 2011;20:318-323.
- Lea CS, Efird JT, Toland AE, et al. Melanoma incidence rates in active duty military personnel compared with a population-based registry in the United States, 2000-2007. Mil Med. 2014;179:247-253.
- Sanlorenzo M, Vujic I, Posch C, et al. The risk of melanoma in pilots and cabin crew: UV measurements in flying airplanes. JAMA Dermatol. 2015;151:450-452.
- Lee T, Taubman SB, Williams VF. Incident diagnoses of non-melanoma skin cancer, active component, U.S. Armed Forces, 2005-2014. MSMR. 2016;23:2-6.
- Ramani ML, Bennett RG. High prevalence of skin cancer in World War II servicemen stationed in the Pacific theater. J Am Acad Dermatol. 1993;28:733-737.
- Henning JS, Firoz, BF. Combat dermatology: the prevalence of skin disease in a deployed dermatology clinic in Iraq. J Drugs Dermatol. 2010;9:210-214.
- Gerall CD, Sippel MR, Yracheta JL, et al. Microcystic adnexal carcinoma: a rare, commonly misdiagnosed malignancy. Mil Med. 2019;184:948-950.
- Wilkison B, Wong E. Skin cancer in military pilots: a special population with special risk factors. Cutis. 2017;100:218-220.
- Proctor SP, Heaton KJ, Smith KW, et al. The Occupational JP8 Neuroepidemiology Study (OJENES): repeated workday exposure and central nervous system functioning among US Air Force personnel. Neurotoxicology. 2011;32:799-808.
- Soldiers protect themselves from skin cancer. US Army website. Published February 28, 2019. Accessed August 21, 2022. https://www.army.mil/article/17601/soldiers_protect_themselves_from_skin_cancer
- Fisher V, Lee D, McGrath J, et al. Veterans speak up: current warnings on skin cancer miss the target, suggestions for improvement. Mil Med. 2015;180:892-897.
- Rogers HW, Weinstick MA, Harris AR, et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146:283-287.
- Sun safety. Army Public Health Center website. Updated June 6, 2019. Accessed August 21, 2022. https://phc.amedd.army.mil/topics/discond/hipss/Pages/Sun-Safety.aspx
- Outdoor ultraviolet radiation hazards and protection. Army Public Health Center website. Accessed August 21, 2022. https://phc.amedd.army.mil/PHC%20Resource%20Library/OutdoorUltravioletRadiationHazardsandProtection_FS_24-017-1115.pdf
- Saraiya M, Frank E, Elon L, et al. Personal and clinical skin cancer prevention practices of US women physicians. Arch Dermatol. 2000;136:633-642.
- What to look for: ABCDEs of melanoma. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/at-risk/abcdes
- Detect skin cancer: how to perform a skin self-exam. American Academy of Dermatology website. Accessed August 21, 2022. https://www.aad.org/public/diseases/skin-cancer/find/check-skin
- Hwang JS, Lappan CM, Sperling LC, et al. Utilization of telemedicine in the US military in a deployed setting. Mil Med. 2014;179:1347-1353.
- Bartling SJ, Rivard SC, Meyerle JH. Melanoma in an active duty marine. Mil Med. 2017;182:2034-2039.
- Day WG, Shirvastava V, Roman JW. Synchronous teledermoscopy in military treatment facilities. Mil Med. 2020;185:1334-1337.
- Chen AC, Martin AJ, Choy B, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373:1618-1626.
- Bavinck JN, Tieben LM, Van der Woude FJ, et al. Prevention of skin cancer and reduction of keratotic skin lesions during acitretin therapy in renal transplant recipients: a double-blind, placebo-controlled study. J Clin Oncol. 1995;13:1933-1938.
- Wulf HC, Pavel S, Stender I, et al. Topical photodynamic therapy for prevention of new skin lesions in renal transplant recipients. Acta Derm Venereol. 2006;86:25-28.
- Weinstock MA, Thwin SS, Siegel JA, et al; Veterans Affairs Keratinocyte Carcinoma Chemoprevention Trial (VAKCC) Group. Chemoprevention of basal and squamous cell carcinoma with a single course of fluorouracil, 5%, cream: a randomized clinical trial. JAMA Dermatol. 2018;154:167-174.
Practice Points
- Skin cancer is more prevalent among military personnel and veterans, especially those in the US Air Force. Frequent and/or prolonged sun exposure and lack of sun protection may be key factors.
- Future research should compare the prevalence of skin cancer in nonpilot military populations to the general US population; explore racial and ethnic differences by military branch and their influence on skin cancers; analyze each branch’s sun-protective measures, uniform wear and personal protective equipment standards, duty assignment locations, and the hours the military members are asked to work outside with direct sunlight exposure; and explore the effects of appropriate military skin cancer intervention and screening programs.
Association of BRAF V600E Status of Incident Melanoma and Risk for a Second Primary Malignancy: A Population-Based Study
The incidence of cutaneous melanoma in the United States has increased in the last 30 years, with the American Cancer Society estimating that 99,780 new melanomas will be diagnosed and 7650 melanoma-related deaths will occur in 2022.1 Patients with melanoma have an increased risk for developing a second primary melanoma or other malignancy, such as salivary gland, small intestine, breast, prostate, renal, or thyroid cancer, but most commonly nonmelanoma skin cancer.2,3 The incidence rate of melanoma among residents of Olmsted County, Minnesota, from 1970 through 2009 has already been described for various age groups4-7; however, the incidence of a second primary malignancy, including melanoma, within these incident cohorts remains unknown.
Mutations in the BRAF oncogene occur in approximately 50% of melanomas.8,9
Although the BRAF mutation event in melanoma is sporadic and should not necessarily affect the development of an unrelated malignancy, we hypothesized that the exposures that may have predisposed a particular individual to a BRAF-mutated melanoma also may have a higher chance of predisposing that individual to the development of another primary malignancy. In this population-based study, we aimed to determine whether the specific melanoma feature of mutant BRAF V600E expression was associated with the development of a second primary malignancy.
Methods
This study was approved by the institutional review boards of the Mayo Clinic and Olmsted Medical Center (both in Rochester, Minnesota). The reporting of this study is compliant with the Strengthening the Reporting of Observational Studies in Epidemiology statement.15
Patient Selection and BRAF Assessment—The Rochester Epidemiology Project (REP) links comprehensive health care records for virtually all residents of Olmsted County, Minnesota, across different medical providers. The REP provides an index of diagnostic and therapeutic procedures, tracks timelines and outcomes of individuals and their medical conditions, and is ideal for population-based studies.
We obtained a list of all residents of Olmsted County aged 18 to 60 years who had a melanoma diagnosed according to the International Classification of Diseases, Ninth Revision, from January 1, 1970, through December 30, 2009; these cohorts have been analyzed previously.4-7 Of the 638 individuals identified, 380 had a melanoma tissue block on file at Mayo Clinic with enough tumor present in available tissue blocks for BRAF assessment. All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) to confirm the diagnosis of melanoma. Tissue blocks were recut, and formalin-fixed, paraffin-embedded tissue sections were stained for BRAF V600E (Spring Bioscience Corporation). BRAF-stained specimens and the associated hematoxylin and eosin−stained slides were reviewed. Melanocyte cytoplasmic staining for BRAF was graded as negative if no staining was evident. BRAF was graded as positive if focal or partial staining was observed (<50% of tumor or low BRAF expression) or if diffuse staining was evident (>50% of tumor or high BRAF expression).
Using resources of the REP, we confirmed patients’ residency status in Olmsted County at the time of diagnosis of the incident melanoma. Patients who denied access to their medical records for research purposes were excluded. We used the complete record of each patient to confirm the date of diagnosis of the incident melanoma. Baseline characteristics of patients and their incident melanomas (eg, anatomic site and pathologic stage according to the American Joint Committee on Cancer classification) were obtained. When only the Clark level was included in the dermatopathology report, the corresponding Breslow thickness was extrapolated from the Clark level,18 and the pathologic stage according to the American Joint Committee on Cancer classification (7th edition) was determined.
For our study, specific diagnostic codes—International Classification of Diseases, Ninth and Tenth Revisions; Hospital International Classification of Diseases Adaptation19; and Berkson16—were applied across individual records to identify all second primary malignancies using the resources of the REP. The diagnosis date, morphology, and anatomic location of second primary malignancies were confirmed from examination of the clinical records.
Statistical Analysis—Baseline characteristics were compared by BRAF V600E expression using Wilcoxon rank sum and χ2 tests. The rate of developing a second primary malignancy at 5, 10, 15, and 20 years after the incident malignant melanoma was estimated with the Kaplan-Meier method. The duration of follow-up was calculated from the incident melanoma date to the second primary malignancy date or the last follow-up date. Patients with a history of the malignancy of interest, except skin cancers, before the incident melanoma date were excluded because it was not possible to distinguish between recurrence of a prior malignancy and a second primary malignancy. Associations of BRAF V600E expression with the development of a second primary malignancy were evaluated with Cox proportional hazards regression models and summarized with hazard ratios (HRs) and 95% CIs; all associations were adjusted for potential confounders such as age at the incident melanoma, year of the incident melanoma, and sex.
Results

Cumulative Incidence of Second Primary Melanoma—Of 133 patients with positive BRAF V600E expression, we identified 14 (10.5%), 1 (0.8%), and 1 (0.8%) who had 1, 2, and 4 subsequent melanomas, respectively. Of the 247 patients with negative BRAF V600E expression, we identified 15 (6%), 4 (1.6%), 2 (0.8%), and 1 (0.4%) patients who had 1, 2, 3, and 4 subsequent melanomas, respectively; BRAF V600E expression was not associated with the number of subsequent melanomas (P=.37; Wilcoxon rank sum test). The cumulative incidences of developing a second primary melanoma (n=38 among the 380 patients studied) at 5, 10, 15, and 20 years after the incident melanoma were 5.3%, 7.6%, 8.1%, and 14.6%, respectively.
Cumulative Incidence of All Second Primary Malignancies—Of the 380 patients studied, 60 (16%) had at least 1 malignancy diagnosed before the incident melanoma. Of the remaining 320 patients, 104 later had at least 1 malignancy develop, including a second primary melanoma, at a median (IQR) of 8.0 (2.7–16.2) years after the incident melanoma; the 104 patients with at least 1 subsequent malignancy included 40 with BRAF-positive and 64 with BRAF-negative melanomas. The cumulative incidences of developing at least 1 malignancy of any kind at 5, 10, 15, and 20 years after the incident melanoma were 15.0%, 20.5%, 31.2%, and 47.0%, respectively. Table 2 shows the number of patients with at least 1 second primary malignancy after the incident melanoma stratified by BRAF status.
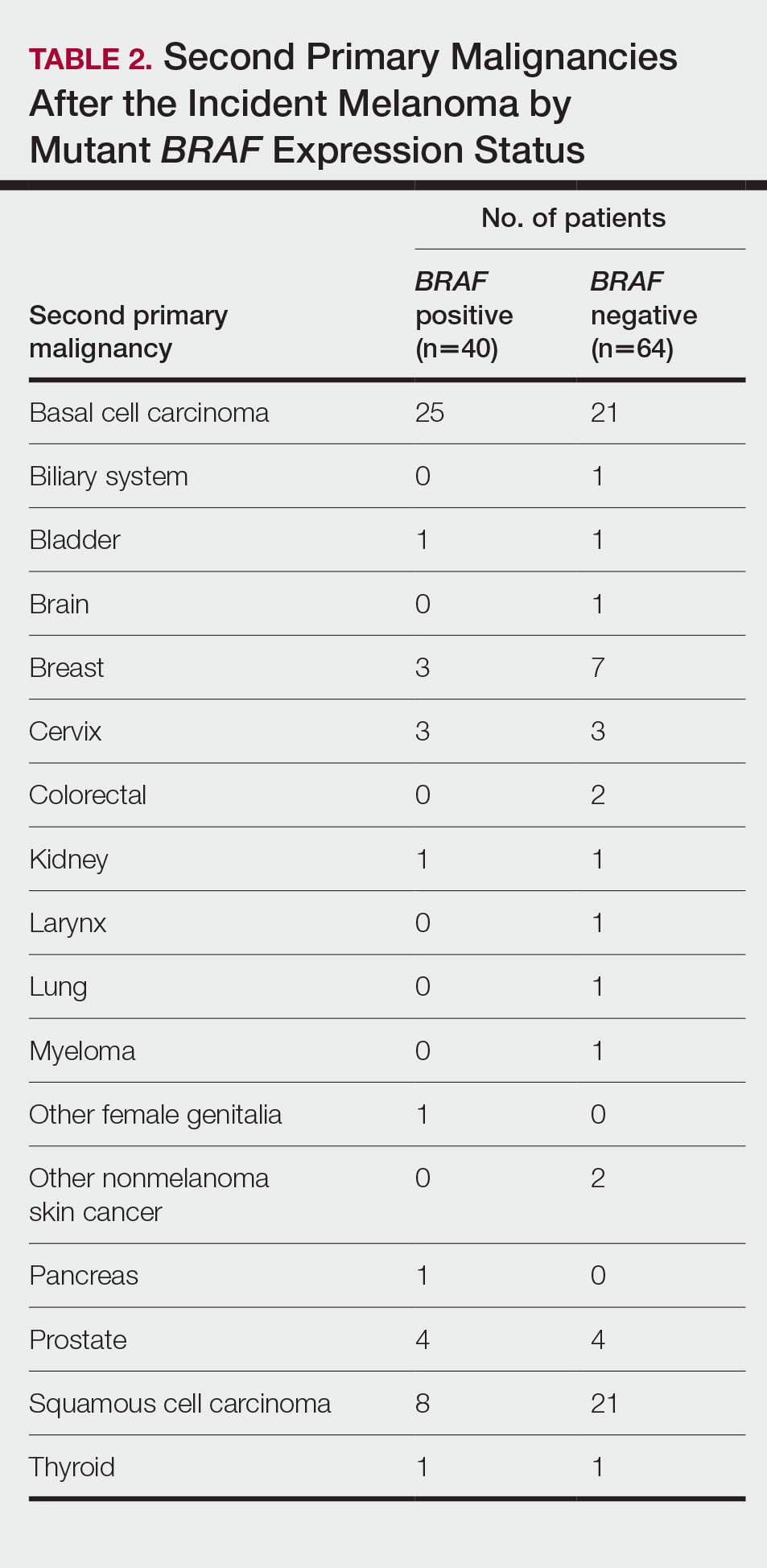
BRAF V600E Expression and Association With Second Primary Malignancy—The eTable shows the associations of mutant BRAF V600E expression status with the development of a new primary malignancy. Malignancies affecting fewer than 10 patients were excluded from the analysis because there were too few events to support the Cox model. Positive BRAF V600E expression was associated with subsequent development of BCCs (HR, 2.32; 95% CI, 1.35-3.99; P=.002) and the development of all combined second primary malignancies excluding melanoma (HR, 1.65; 95% CI, 1.06-2.56; P=.03). However, BRAF V600E status was no longer a significant factor when all second primary malignancies, including second melanomas, were considered (P=.06). Table 3 shows the 5-, 10-, 15-, and 20-year cumulative incidences of all second primary malignancies according to mutant BRAF status.
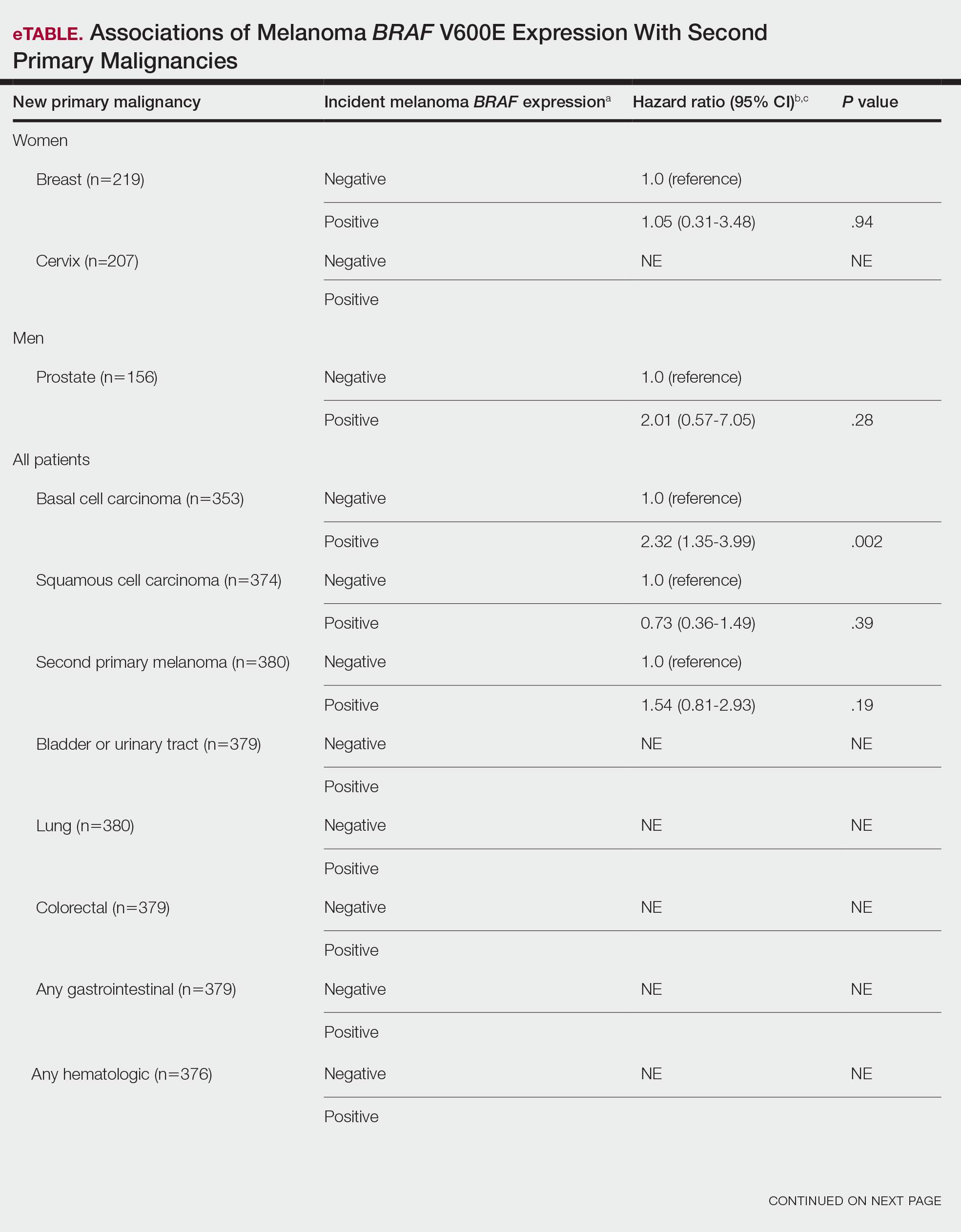
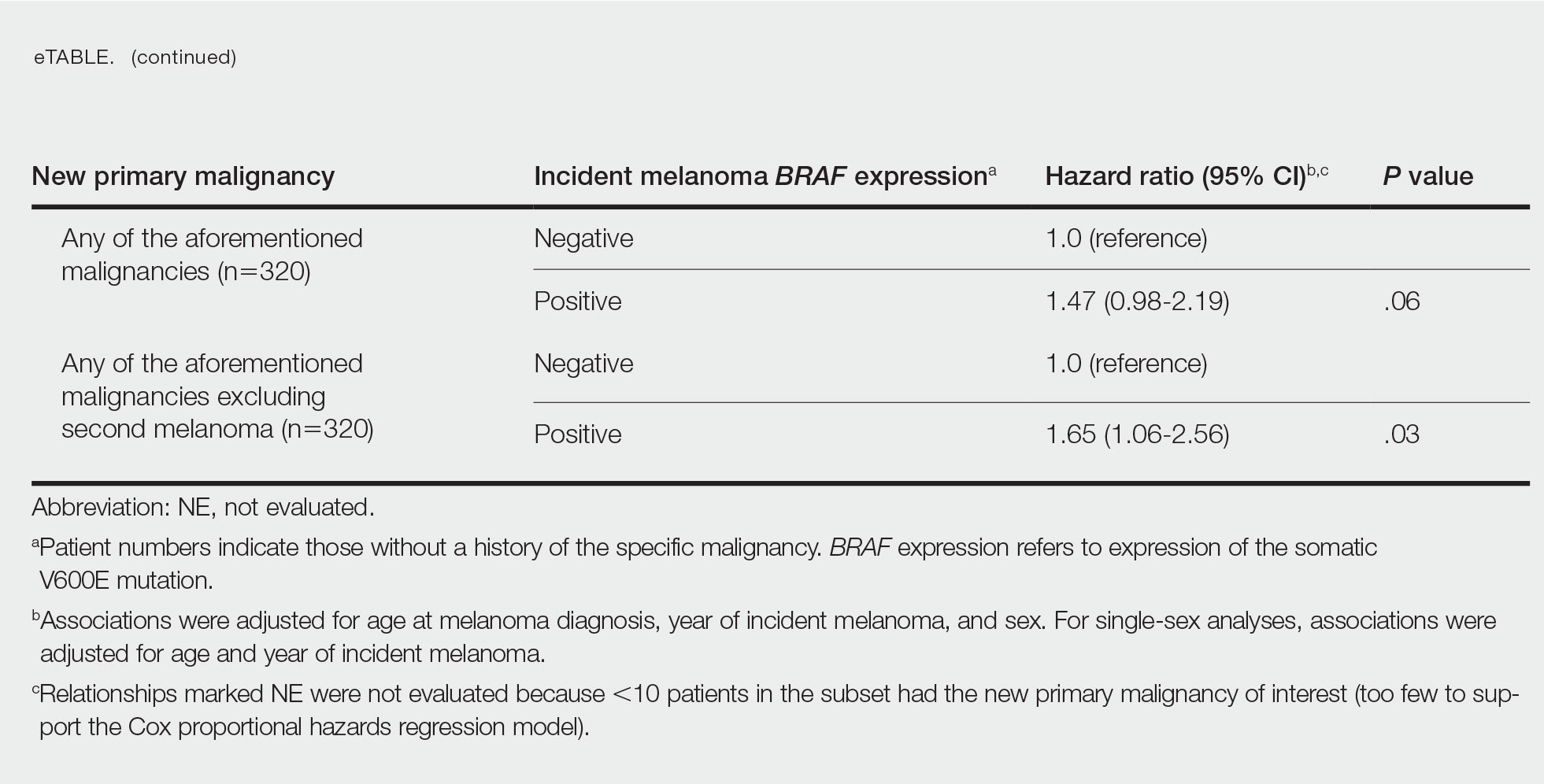
Comment
Association of BRAF V600E Expression With Second Primary Malignancies—BRAF V600E expression of an incident melanoma was associated with the development of all combined second primary malignancies excluding melanoma; however, this association was not statistically significant when second primary melanomas were included. A possible explanation is that individuals with more than 1 primary melanoma possess additional genetic risk—CDKN2A or CDKN4 gene mutations or MC1R variation—that outweighed the effect of BRAF expression in the statistical analysis.
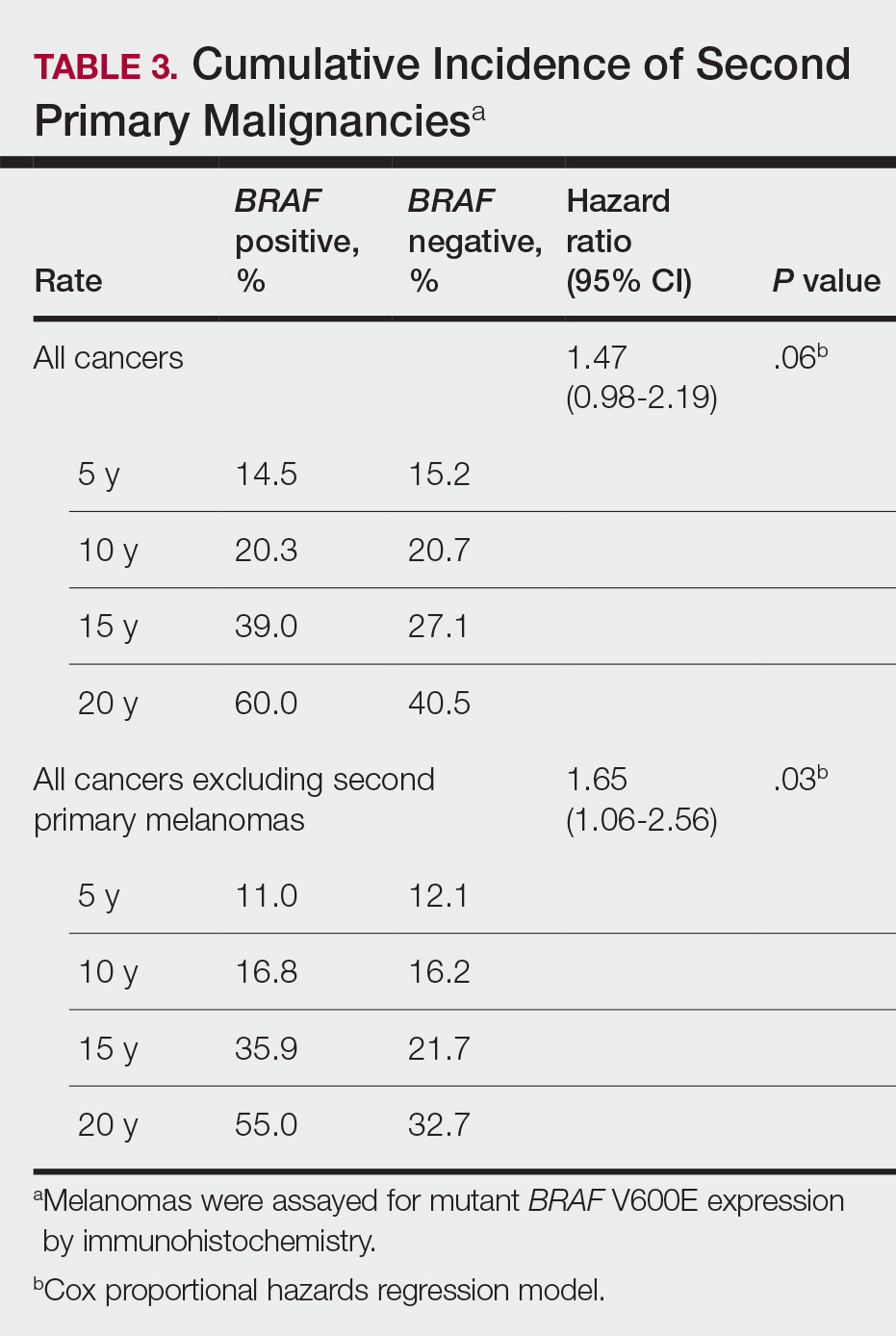
The 5- and 10-year cumulative incidences of all second primary malignancies excluding second primary melanoma were similar between BRAF-positive and BRAF-negative melanoma, but the 15- and 20-year cumulative incidences were greater for the BRAF-positive cohort. This could reflect the association of BRAF expression with BCCs and the increased likelihood of their occurrence with cumulative sun exposure and advancing age. BRAF expression was associated with the development of BCCs, but the reason for this association was unclear. BRAF-mutated melanoma occurs more frequently on sun-protected sites,20 whereas sporadic BCC generally occurs on sun-exposed sites. However, BRAF-mutated melanoma is associated with high levels of ambient UV exposure early in life, particularly birth through 20 years of age,21 and we speculate that such early UV exposure influences the later development of BCCs.
Development of BRAF-Mutated Cancers—It currently is not understood why the same somatic mutation can cause different types of cancer. A recent translational research study showed that in mice models, precursor cells of the pancreas and bile duct responded differently when exposed to PIK3CA and KRAS oncogenes, and tumorigenesis is influenced by specific cooperating genetic events in the tissue microenvironment. Future research investigating these molecular interactions may lead to better understanding of cancer pathogenesis and direct the design of new targeted therapies.22,23
Regarding environmental influences on the development of BRAF-mutated cancers, we found 1 population-based study that identified an association between high iodine content of drinking water and the prevalence of T1799A BRAF papillary thyroid carcinoma in 5 regions in China.24 Another study identified an increased risk for colorectal cancer and nonmelanoma skin cancer in the first-degree relatives of index patients with BRAF V600E colorectal cancer.25 Two studies by institutions in China and Sweden reported the frequency of BRAF mutations in cohorts of patients with melanoma.26,27
Additional studies investigating a possible association between BRAF-mutated melanoma and other cancers with larger numbers of participants than in our study may become more feasible in the future with increased routine genetic testing of biopsied cancers.
Study Limitations—Limitations of this retrospective epidemiologic study include the possibility of ascertainment bias during data collection. We did not account for known risk factors for cancer (eg, excessive sun exposure, smoking).
The main clinical implications from this study are that we do not have enough evidence to recommend BRAF testing for all incident melanomas, and BRAF-mutated melanomas cannot be associated with increased risk for developing other forms of cancer, with the possible exception of BCCs
Conclusion
Physicians should be aware of the risk for a second primary malignancy after an incident melanoma, and we emphasize the importance of long-term cancer surveillance.
Acknowledgment—We thank Ms. Jayne H. Feind (Rochester, Minnesota) for assistance with study coordination.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 12, 2022. Accessed August 15, 2022.https://www.cancer.org/cancer/melanoma-skin-cancer/about/key-statistics.html
- American Cancer Society. Second Cancers After Melanoma Skin Cancer. Accessed August 19, 2022. https://www.cancer.org/cancer/melanoma-skin-cancer/after-treatment/second-cancers.html
- Spanogle JP, Clarke CA, Aroner S, et al. Risk of second primary malignancies following cutaneous melanoma diagnosis: a population-based study. J Am Acad Dermatol. 2010;62:757-767.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Lowe GC, Brewer JD, Peters MS, et al. Incidence of melanoma in the pediatric population: a population-based study in Olmsted County, Minnesota. Pediatr Derm. 2015;32:618-620.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma [editorial]. J Transl Med. 2012;10:85.
- Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-954.
- Miller AJ, Mihm MC Jr. Melanoma. N Engl J Med. 2006;355:51-65.
- Tiacci E, Trifonov V, Schiavoni G, et al. BRAF mutations in hairy-cell leukemia. N Engl J Med. 2011;364:2305-2315.
- Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer. 2005;12:245-262.
- Moreau S, Saiag P, Aegerter P, et al. Prognostic value of BRAF(V600) mutations in melanoma patients after resection of metastatic lymph nodes. Ann Surg Oncol. 2012;19:4314-4321.
- Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61:344-349.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- St. Sauver JL, Grossardt BR, Yawn BP, et al. Data resource profile: the Rochester Epidemiology Project (REP) medical records-linkage system. Int J Epidemiol. 2012;41:1614-1624.
- National Cancer Institute. Staging: melanoma of the skin, vulva, penis and scrotum staging. Accessed August 15, 2022. https://training.seer.cancer.gov/melanoma/abstract-code-stage/staging.html
- Pakhomov SV, Buntrock JD, Chute CG. Automating the assignment of diagnosis codes to patient encounters using example-based and machine learning techniques. J Am Med Inform Assoc. 2006;13:516-525.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- German Cancer Research Center. Why identical mutations cause different types of cancer. July 19, 2021. Accessed August 15, 2022. https://www.dkfz.de/en/presse/pressemitteilungen/2021/dkfz-pm-21-41-Why-identical-mutations-cause-different-types-of-cancer.php
- Falcomatà C, Bärthel S, Ulrich A, et al. Genetic screens identify a context-specific PI3K/p27Kip1 node driving extrahepatic biliary cancer. Cancer Discov. 2021;11:3158-3177.
- Guan H, Ji M, Bao R, et al. Association of high iodine intake with the T1799A BRAF mutation in papillary thyroid cancer. J Clin Endocrinol Metab. 2009;94:1612-1617.
- Wish TA, Hyde AJ, Parfrey PS, et al. Increased cancer predisposition in family members of colorectal cancer patients harboring the p.V600E BRAF mutation: a population-based study. Cancer Epidemiol Biomarkers Prev. 2010;19:1831-1839.
- Zebary A, Omholt K, Vassilaki I, et al. KIT, NRAS, BRAF and PTEN mutations in a sample of Swedish patients with acral lentiginous melanoma. J Dermatol Sci. 2013;72:284-289.
- Si L, Kong Y, Xu X, et al. Prevalence of BRAF V600E mutation in Chinese melanoma patients: large scale analysis of BRAF and NRAS mutations in a 432-case cohort. Eur J Cancer. 2012;48:94-100.
- Safaee Ardekani G, Jafarnejad SM, Khosravi S, et al. Disease progression and patient survival are significantly influenced by BRAF protein expression in primary melanoma. Br J Dermatol. 2013;169:320-328.
The incidence of cutaneous melanoma in the United States has increased in the last 30 years, with the American Cancer Society estimating that 99,780 new melanomas will be diagnosed and 7650 melanoma-related deaths will occur in 2022.1 Patients with melanoma have an increased risk for developing a second primary melanoma or other malignancy, such as salivary gland, small intestine, breast, prostate, renal, or thyroid cancer, but most commonly nonmelanoma skin cancer.2,3 The incidence rate of melanoma among residents of Olmsted County, Minnesota, from 1970 through 2009 has already been described for various age groups4-7; however, the incidence of a second primary malignancy, including melanoma, within these incident cohorts remains unknown.
Mutations in the BRAF oncogene occur in approximately 50% of melanomas.8,9
Although the BRAF mutation event in melanoma is sporadic and should not necessarily affect the development of an unrelated malignancy, we hypothesized that the exposures that may have predisposed a particular individual to a BRAF-mutated melanoma also may have a higher chance of predisposing that individual to the development of another primary malignancy. In this population-based study, we aimed to determine whether the specific melanoma feature of mutant BRAF V600E expression was associated with the development of a second primary malignancy.
Methods
This study was approved by the institutional review boards of the Mayo Clinic and Olmsted Medical Center (both in Rochester, Minnesota). The reporting of this study is compliant with the Strengthening the Reporting of Observational Studies in Epidemiology statement.15
Patient Selection and BRAF Assessment—The Rochester Epidemiology Project (REP) links comprehensive health care records for virtually all residents of Olmsted County, Minnesota, across different medical providers. The REP provides an index of diagnostic and therapeutic procedures, tracks timelines and outcomes of individuals and their medical conditions, and is ideal for population-based studies.
We obtained a list of all residents of Olmsted County aged 18 to 60 years who had a melanoma diagnosed according to the International Classification of Diseases, Ninth Revision, from January 1, 1970, through December 30, 2009; these cohorts have been analyzed previously.4-7 Of the 638 individuals identified, 380 had a melanoma tissue block on file at Mayo Clinic with enough tumor present in available tissue blocks for BRAF assessment. All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) to confirm the diagnosis of melanoma. Tissue blocks were recut, and formalin-fixed, paraffin-embedded tissue sections were stained for BRAF V600E (Spring Bioscience Corporation). BRAF-stained specimens and the associated hematoxylin and eosin−stained slides were reviewed. Melanocyte cytoplasmic staining for BRAF was graded as negative if no staining was evident. BRAF was graded as positive if focal or partial staining was observed (<50% of tumor or low BRAF expression) or if diffuse staining was evident (>50% of tumor or high BRAF expression).
Using resources of the REP, we confirmed patients’ residency status in Olmsted County at the time of diagnosis of the incident melanoma. Patients who denied access to their medical records for research purposes were excluded. We used the complete record of each patient to confirm the date of diagnosis of the incident melanoma. Baseline characteristics of patients and their incident melanomas (eg, anatomic site and pathologic stage according to the American Joint Committee on Cancer classification) were obtained. When only the Clark level was included in the dermatopathology report, the corresponding Breslow thickness was extrapolated from the Clark level,18 and the pathologic stage according to the American Joint Committee on Cancer classification (7th edition) was determined.
For our study, specific diagnostic codes—International Classification of Diseases, Ninth and Tenth Revisions; Hospital International Classification of Diseases Adaptation19; and Berkson16—were applied across individual records to identify all second primary malignancies using the resources of the REP. The diagnosis date, morphology, and anatomic location of second primary malignancies were confirmed from examination of the clinical records.
Statistical Analysis—Baseline characteristics were compared by BRAF V600E expression using Wilcoxon rank sum and χ2 tests. The rate of developing a second primary malignancy at 5, 10, 15, and 20 years after the incident malignant melanoma was estimated with the Kaplan-Meier method. The duration of follow-up was calculated from the incident melanoma date to the second primary malignancy date or the last follow-up date. Patients with a history of the malignancy of interest, except skin cancers, before the incident melanoma date were excluded because it was not possible to distinguish between recurrence of a prior malignancy and a second primary malignancy. Associations of BRAF V600E expression with the development of a second primary malignancy were evaluated with Cox proportional hazards regression models and summarized with hazard ratios (HRs) and 95% CIs; all associations were adjusted for potential confounders such as age at the incident melanoma, year of the incident melanoma, and sex.
Results

Cumulative Incidence of Second Primary Melanoma—Of 133 patients with positive BRAF V600E expression, we identified 14 (10.5%), 1 (0.8%), and 1 (0.8%) who had 1, 2, and 4 subsequent melanomas, respectively. Of the 247 patients with negative BRAF V600E expression, we identified 15 (6%), 4 (1.6%), 2 (0.8%), and 1 (0.4%) patients who had 1, 2, 3, and 4 subsequent melanomas, respectively; BRAF V600E expression was not associated with the number of subsequent melanomas (P=.37; Wilcoxon rank sum test). The cumulative incidences of developing a second primary melanoma (n=38 among the 380 patients studied) at 5, 10, 15, and 20 years after the incident melanoma were 5.3%, 7.6%, 8.1%, and 14.6%, respectively.
Cumulative Incidence of All Second Primary Malignancies—Of the 380 patients studied, 60 (16%) had at least 1 malignancy diagnosed before the incident melanoma. Of the remaining 320 patients, 104 later had at least 1 malignancy develop, including a second primary melanoma, at a median (IQR) of 8.0 (2.7–16.2) years after the incident melanoma; the 104 patients with at least 1 subsequent malignancy included 40 with BRAF-positive and 64 with BRAF-negative melanomas. The cumulative incidences of developing at least 1 malignancy of any kind at 5, 10, 15, and 20 years after the incident melanoma were 15.0%, 20.5%, 31.2%, and 47.0%, respectively. Table 2 shows the number of patients with at least 1 second primary malignancy after the incident melanoma stratified by BRAF status.

BRAF V600E Expression and Association With Second Primary Malignancy—The eTable shows the associations of mutant BRAF V600E expression status with the development of a new primary malignancy. Malignancies affecting fewer than 10 patients were excluded from the analysis because there were too few events to support the Cox model. Positive BRAF V600E expression was associated with subsequent development of BCCs (HR, 2.32; 95% CI, 1.35-3.99; P=.002) and the development of all combined second primary malignancies excluding melanoma (HR, 1.65; 95% CI, 1.06-2.56; P=.03). However, BRAF V600E status was no longer a significant factor when all second primary malignancies, including second melanomas, were considered (P=.06). Table 3 shows the 5-, 10-, 15-, and 20-year cumulative incidences of all second primary malignancies according to mutant BRAF status.


Comment
Association of BRAF V600E Expression With Second Primary Malignancies—BRAF V600E expression of an incident melanoma was associated with the development of all combined second primary malignancies excluding melanoma; however, this association was not statistically significant when second primary melanomas were included. A possible explanation is that individuals with more than 1 primary melanoma possess additional genetic risk—CDKN2A or CDKN4 gene mutations or MC1R variation—that outweighed the effect of BRAF expression in the statistical analysis.

The 5- and 10-year cumulative incidences of all second primary malignancies excluding second primary melanoma were similar between BRAF-positive and BRAF-negative melanoma, but the 15- and 20-year cumulative incidences were greater for the BRAF-positive cohort. This could reflect the association of BRAF expression with BCCs and the increased likelihood of their occurrence with cumulative sun exposure and advancing age. BRAF expression was associated with the development of BCCs, but the reason for this association was unclear. BRAF-mutated melanoma occurs more frequently on sun-protected sites,20 whereas sporadic BCC generally occurs on sun-exposed sites. However, BRAF-mutated melanoma is associated with high levels of ambient UV exposure early in life, particularly birth through 20 years of age,21 and we speculate that such early UV exposure influences the later development of BCCs.
Development of BRAF-Mutated Cancers—It currently is not understood why the same somatic mutation can cause different types of cancer. A recent translational research study showed that in mice models, precursor cells of the pancreas and bile duct responded differently when exposed to PIK3CA and KRAS oncogenes, and tumorigenesis is influenced by specific cooperating genetic events in the tissue microenvironment. Future research investigating these molecular interactions may lead to better understanding of cancer pathogenesis and direct the design of new targeted therapies.22,23
Regarding environmental influences on the development of BRAF-mutated cancers, we found 1 population-based study that identified an association between high iodine content of drinking water and the prevalence of T1799A BRAF papillary thyroid carcinoma in 5 regions in China.24 Another study identified an increased risk for colorectal cancer and nonmelanoma skin cancer in the first-degree relatives of index patients with BRAF V600E colorectal cancer.25 Two studies by institutions in China and Sweden reported the frequency of BRAF mutations in cohorts of patients with melanoma.26,27
Additional studies investigating a possible association between BRAF-mutated melanoma and other cancers with larger numbers of participants than in our study may become more feasible in the future with increased routine genetic testing of biopsied cancers.
Study Limitations—Limitations of this retrospective epidemiologic study include the possibility of ascertainment bias during data collection. We did not account for known risk factors for cancer (eg, excessive sun exposure, smoking).
The main clinical implications from this study are that we do not have enough evidence to recommend BRAF testing for all incident melanomas, and BRAF-mutated melanomas cannot be associated with increased risk for developing other forms of cancer, with the possible exception of BCCs
Conclusion
Physicians should be aware of the risk for a second primary malignancy after an incident melanoma, and we emphasize the importance of long-term cancer surveillance.
Acknowledgment—We thank Ms. Jayne H. Feind (Rochester, Minnesota) for assistance with study coordination.
The incidence of cutaneous melanoma in the United States has increased in the last 30 years, with the American Cancer Society estimating that 99,780 new melanomas will be diagnosed and 7650 melanoma-related deaths will occur in 2022.1 Patients with melanoma have an increased risk for developing a second primary melanoma or other malignancy, such as salivary gland, small intestine, breast, prostate, renal, or thyroid cancer, but most commonly nonmelanoma skin cancer.2,3 The incidence rate of melanoma among residents of Olmsted County, Minnesota, from 1970 through 2009 has already been described for various age groups4-7; however, the incidence of a second primary malignancy, including melanoma, within these incident cohorts remains unknown.
Mutations in the BRAF oncogene occur in approximately 50% of melanomas.8,9
Although the BRAF mutation event in melanoma is sporadic and should not necessarily affect the development of an unrelated malignancy, we hypothesized that the exposures that may have predisposed a particular individual to a BRAF-mutated melanoma also may have a higher chance of predisposing that individual to the development of another primary malignancy. In this population-based study, we aimed to determine whether the specific melanoma feature of mutant BRAF V600E expression was associated with the development of a second primary malignancy.
Methods
This study was approved by the institutional review boards of the Mayo Clinic and Olmsted Medical Center (both in Rochester, Minnesota). The reporting of this study is compliant with the Strengthening the Reporting of Observational Studies in Epidemiology statement.15
Patient Selection and BRAF Assessment—The Rochester Epidemiology Project (REP) links comprehensive health care records for virtually all residents of Olmsted County, Minnesota, across different medical providers. The REP provides an index of diagnostic and therapeutic procedures, tracks timelines and outcomes of individuals and their medical conditions, and is ideal for population-based studies.
We obtained a list of all residents of Olmsted County aged 18 to 60 years who had a melanoma diagnosed according to the International Classification of Diseases, Ninth Revision, from January 1, 1970, through December 30, 2009; these cohorts have been analyzed previously.4-7 Of the 638 individuals identified, 380 had a melanoma tissue block on file at Mayo Clinic with enough tumor present in available tissue blocks for BRAF assessment. All specimens were reviewed by a board-certified dermatopathologist (J.S.L.) to confirm the diagnosis of melanoma. Tissue blocks were recut, and formalin-fixed, paraffin-embedded tissue sections were stained for BRAF V600E (Spring Bioscience Corporation). BRAF-stained specimens and the associated hematoxylin and eosin−stained slides were reviewed. Melanocyte cytoplasmic staining for BRAF was graded as negative if no staining was evident. BRAF was graded as positive if focal or partial staining was observed (<50% of tumor or low BRAF expression) or if diffuse staining was evident (>50% of tumor or high BRAF expression).
Using resources of the REP, we confirmed patients’ residency status in Olmsted County at the time of diagnosis of the incident melanoma. Patients who denied access to their medical records for research purposes were excluded. We used the complete record of each patient to confirm the date of diagnosis of the incident melanoma. Baseline characteristics of patients and their incident melanomas (eg, anatomic site and pathologic stage according to the American Joint Committee on Cancer classification) were obtained. When only the Clark level was included in the dermatopathology report, the corresponding Breslow thickness was extrapolated from the Clark level,18 and the pathologic stage according to the American Joint Committee on Cancer classification (7th edition) was determined.
For our study, specific diagnostic codes—International Classification of Diseases, Ninth and Tenth Revisions; Hospital International Classification of Diseases Adaptation19; and Berkson16—were applied across individual records to identify all second primary malignancies using the resources of the REP. The diagnosis date, morphology, and anatomic location of second primary malignancies were confirmed from examination of the clinical records.
Statistical Analysis—Baseline characteristics were compared by BRAF V600E expression using Wilcoxon rank sum and χ2 tests. The rate of developing a second primary malignancy at 5, 10, 15, and 20 years after the incident malignant melanoma was estimated with the Kaplan-Meier method. The duration of follow-up was calculated from the incident melanoma date to the second primary malignancy date or the last follow-up date. Patients with a history of the malignancy of interest, except skin cancers, before the incident melanoma date were excluded because it was not possible to distinguish between recurrence of a prior malignancy and a second primary malignancy. Associations of BRAF V600E expression with the development of a second primary malignancy were evaluated with Cox proportional hazards regression models and summarized with hazard ratios (HRs) and 95% CIs; all associations were adjusted for potential confounders such as age at the incident melanoma, year of the incident melanoma, and sex.
Results

Cumulative Incidence of Second Primary Melanoma—Of 133 patients with positive BRAF V600E expression, we identified 14 (10.5%), 1 (0.8%), and 1 (0.8%) who had 1, 2, and 4 subsequent melanomas, respectively. Of the 247 patients with negative BRAF V600E expression, we identified 15 (6%), 4 (1.6%), 2 (0.8%), and 1 (0.4%) patients who had 1, 2, 3, and 4 subsequent melanomas, respectively; BRAF V600E expression was not associated with the number of subsequent melanomas (P=.37; Wilcoxon rank sum test). The cumulative incidences of developing a second primary melanoma (n=38 among the 380 patients studied) at 5, 10, 15, and 20 years after the incident melanoma were 5.3%, 7.6%, 8.1%, and 14.6%, respectively.
Cumulative Incidence of All Second Primary Malignancies—Of the 380 patients studied, 60 (16%) had at least 1 malignancy diagnosed before the incident melanoma. Of the remaining 320 patients, 104 later had at least 1 malignancy develop, including a second primary melanoma, at a median (IQR) of 8.0 (2.7–16.2) years after the incident melanoma; the 104 patients with at least 1 subsequent malignancy included 40 with BRAF-positive and 64 with BRAF-negative melanomas. The cumulative incidences of developing at least 1 malignancy of any kind at 5, 10, 15, and 20 years after the incident melanoma were 15.0%, 20.5%, 31.2%, and 47.0%, respectively. Table 2 shows the number of patients with at least 1 second primary malignancy after the incident melanoma stratified by BRAF status.

BRAF V600E Expression and Association With Second Primary Malignancy—The eTable shows the associations of mutant BRAF V600E expression status with the development of a new primary malignancy. Malignancies affecting fewer than 10 patients were excluded from the analysis because there were too few events to support the Cox model. Positive BRAF V600E expression was associated with subsequent development of BCCs (HR, 2.32; 95% CI, 1.35-3.99; P=.002) and the development of all combined second primary malignancies excluding melanoma (HR, 1.65; 95% CI, 1.06-2.56; P=.03). However, BRAF V600E status was no longer a significant factor when all second primary malignancies, including second melanomas, were considered (P=.06). Table 3 shows the 5-, 10-, 15-, and 20-year cumulative incidences of all second primary malignancies according to mutant BRAF status.


Comment
Association of BRAF V600E Expression With Second Primary Malignancies—BRAF V600E expression of an incident melanoma was associated with the development of all combined second primary malignancies excluding melanoma; however, this association was not statistically significant when second primary melanomas were included. A possible explanation is that individuals with more than 1 primary melanoma possess additional genetic risk—CDKN2A or CDKN4 gene mutations or MC1R variation—that outweighed the effect of BRAF expression in the statistical analysis.

The 5- and 10-year cumulative incidences of all second primary malignancies excluding second primary melanoma were similar between BRAF-positive and BRAF-negative melanoma, but the 15- and 20-year cumulative incidences were greater for the BRAF-positive cohort. This could reflect the association of BRAF expression with BCCs and the increased likelihood of their occurrence with cumulative sun exposure and advancing age. BRAF expression was associated with the development of BCCs, but the reason for this association was unclear. BRAF-mutated melanoma occurs more frequently on sun-protected sites,20 whereas sporadic BCC generally occurs on sun-exposed sites. However, BRAF-mutated melanoma is associated with high levels of ambient UV exposure early in life, particularly birth through 20 years of age,21 and we speculate that such early UV exposure influences the later development of BCCs.
Development of BRAF-Mutated Cancers—It currently is not understood why the same somatic mutation can cause different types of cancer. A recent translational research study showed that in mice models, precursor cells of the pancreas and bile duct responded differently when exposed to PIK3CA and KRAS oncogenes, and tumorigenesis is influenced by specific cooperating genetic events in the tissue microenvironment. Future research investigating these molecular interactions may lead to better understanding of cancer pathogenesis and direct the design of new targeted therapies.22,23
Regarding environmental influences on the development of BRAF-mutated cancers, we found 1 population-based study that identified an association between high iodine content of drinking water and the prevalence of T1799A BRAF papillary thyroid carcinoma in 5 regions in China.24 Another study identified an increased risk for colorectal cancer and nonmelanoma skin cancer in the first-degree relatives of index patients with BRAF V600E colorectal cancer.25 Two studies by institutions in China and Sweden reported the frequency of BRAF mutations in cohorts of patients with melanoma.26,27
Additional studies investigating a possible association between BRAF-mutated melanoma and other cancers with larger numbers of participants than in our study may become more feasible in the future with increased routine genetic testing of biopsied cancers.
Study Limitations—Limitations of this retrospective epidemiologic study include the possibility of ascertainment bias during data collection. We did not account for known risk factors for cancer (eg, excessive sun exposure, smoking).
The main clinical implications from this study are that we do not have enough evidence to recommend BRAF testing for all incident melanomas, and BRAF-mutated melanomas cannot be associated with increased risk for developing other forms of cancer, with the possible exception of BCCs
Conclusion
Physicians should be aware of the risk for a second primary malignancy after an incident melanoma, and we emphasize the importance of long-term cancer surveillance.
Acknowledgment—We thank Ms. Jayne H. Feind (Rochester, Minnesota) for assistance with study coordination.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 12, 2022. Accessed August 15, 2022.https://www.cancer.org/cancer/melanoma-skin-cancer/about/key-statistics.html
- American Cancer Society. Second Cancers After Melanoma Skin Cancer. Accessed August 19, 2022. https://www.cancer.org/cancer/melanoma-skin-cancer/after-treatment/second-cancers.html
- Spanogle JP, Clarke CA, Aroner S, et al. Risk of second primary malignancies following cutaneous melanoma diagnosis: a population-based study. J Am Acad Dermatol. 2010;62:757-767.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Lowe GC, Brewer JD, Peters MS, et al. Incidence of melanoma in the pediatric population: a population-based study in Olmsted County, Minnesota. Pediatr Derm. 2015;32:618-620.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma [editorial]. J Transl Med. 2012;10:85.
- Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-954.
- Miller AJ, Mihm MC Jr. Melanoma. N Engl J Med. 2006;355:51-65.
- Tiacci E, Trifonov V, Schiavoni G, et al. BRAF mutations in hairy-cell leukemia. N Engl J Med. 2011;364:2305-2315.
- Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer. 2005;12:245-262.
- Moreau S, Saiag P, Aegerter P, et al. Prognostic value of BRAF(V600) mutations in melanoma patients after resection of metastatic lymph nodes. Ann Surg Oncol. 2012;19:4314-4321.
- Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61:344-349.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- St. Sauver JL, Grossardt BR, Yawn BP, et al. Data resource profile: the Rochester Epidemiology Project (REP) medical records-linkage system. Int J Epidemiol. 2012;41:1614-1624.
- National Cancer Institute. Staging: melanoma of the skin, vulva, penis and scrotum staging. Accessed August 15, 2022. https://training.seer.cancer.gov/melanoma/abstract-code-stage/staging.html
- Pakhomov SV, Buntrock JD, Chute CG. Automating the assignment of diagnosis codes to patient encounters using example-based and machine learning techniques. J Am Med Inform Assoc. 2006;13:516-525.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- German Cancer Research Center. Why identical mutations cause different types of cancer. July 19, 2021. Accessed August 15, 2022. https://www.dkfz.de/en/presse/pressemitteilungen/2021/dkfz-pm-21-41-Why-identical-mutations-cause-different-types-of-cancer.php
- Falcomatà C, Bärthel S, Ulrich A, et al. Genetic screens identify a context-specific PI3K/p27Kip1 node driving extrahepatic biliary cancer. Cancer Discov. 2021;11:3158-3177.
- Guan H, Ji M, Bao R, et al. Association of high iodine intake with the T1799A BRAF mutation in papillary thyroid cancer. J Clin Endocrinol Metab. 2009;94:1612-1617.
- Wish TA, Hyde AJ, Parfrey PS, et al. Increased cancer predisposition in family members of colorectal cancer patients harboring the p.V600E BRAF mutation: a population-based study. Cancer Epidemiol Biomarkers Prev. 2010;19:1831-1839.
- Zebary A, Omholt K, Vassilaki I, et al. KIT, NRAS, BRAF and PTEN mutations in a sample of Swedish patients with acral lentiginous melanoma. J Dermatol Sci. 2013;72:284-289.
- Si L, Kong Y, Xu X, et al. Prevalence of BRAF V600E mutation in Chinese melanoma patients: large scale analysis of BRAF and NRAS mutations in a 432-case cohort. Eur J Cancer. 2012;48:94-100.
- Safaee Ardekani G, Jafarnejad SM, Khosravi S, et al. Disease progression and patient survival are significantly influenced by BRAF protein expression in primary melanoma. Br J Dermatol. 2013;169:320-328.
- American Cancer Society. Key statistics for melanoma skin cancer. Updated January 12, 2022. Accessed August 15, 2022.https://www.cancer.org/cancer/melanoma-skin-cancer/about/key-statistics.html
- American Cancer Society. Second Cancers After Melanoma Skin Cancer. Accessed August 19, 2022. https://www.cancer.org/cancer/melanoma-skin-cancer/after-treatment/second-cancers.html
- Spanogle JP, Clarke CA, Aroner S, et al. Risk of second primary malignancies following cutaneous melanoma diagnosis: a population-based study. J Am Acad Dermatol. 2010;62:757-767.
- Olazagasti Lourido JM, Ma JE, Lohse CM, et al. Increasing incidence of melanoma in the elderly: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2016;91:1555-1562.
- Reed KB, Brewer JD, Lohse CM, et al. Increasing incidence of melanoma among young adults: an epidemiological study in Olmsted County, Minnesota. Mayo Clin Proc. 2012;87:328-334.
- Lowe GC, Brewer JD, Peters MS, et al. Incidence of melanoma in the pediatric population: a population-based study in Olmsted County, Minnesota. Pediatr Derm. 2015;32:618-620.
- Lowe GC, Saavedra A, Reed KB, et al. Increasing incidence of melanoma among middle-aged adults: an epidemiologic study in Olmsted County, Minnesota. Mayo Clin Proc. 2014;89:52-59.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma [editorial]. J Transl Med. 2012;10:85.
- Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-954.
- Miller AJ, Mihm MC Jr. Melanoma. N Engl J Med. 2006;355:51-65.
- Tiacci E, Trifonov V, Schiavoni G, et al. BRAF mutations in hairy-cell leukemia. N Engl J Med. 2011;364:2305-2315.
- Xing M. BRAF mutation in thyroid cancer. Endocr Relat Cancer. 2005;12:245-262.
- Moreau S, Saiag P, Aegerter P, et al. Prognostic value of BRAF(V600) mutations in melanoma patients after resection of metastatic lymph nodes. Ann Surg Oncol. 2012;19:4314-4321.
- Flaherty KT, Robert C, Hersey P, et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med. 2012;367:107-114.
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61:344-349.
- Rocca WA, Yawn BP, St Sauver JL, et al. History of the Rochester Epidemiology Project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87:1202-1213.
- St. Sauver JL, Grossardt BR, Yawn BP, et al. Data resource profile: the Rochester Epidemiology Project (REP) medical records-linkage system. Int J Epidemiol. 2012;41:1614-1624.
- National Cancer Institute. Staging: melanoma of the skin, vulva, penis and scrotum staging. Accessed August 15, 2022. https://training.seer.cancer.gov/melanoma/abstract-code-stage/staging.html
- Pakhomov SV, Buntrock JD, Chute CG. Automating the assignment of diagnosis codes to patient encounters using example-based and machine learning techniques. J Am Med Inform Assoc. 2006;13:516-525.
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147.
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991-997.
- German Cancer Research Center. Why identical mutations cause different types of cancer. July 19, 2021. Accessed August 15, 2022. https://www.dkfz.de/en/presse/pressemitteilungen/2021/dkfz-pm-21-41-Why-identical-mutations-cause-different-types-of-cancer.php
- Falcomatà C, Bärthel S, Ulrich A, et al. Genetic screens identify a context-specific PI3K/p27Kip1 node driving extrahepatic biliary cancer. Cancer Discov. 2021;11:3158-3177.
- Guan H, Ji M, Bao R, et al. Association of high iodine intake with the T1799A BRAF mutation in papillary thyroid cancer. J Clin Endocrinol Metab. 2009;94:1612-1617.
- Wish TA, Hyde AJ, Parfrey PS, et al. Increased cancer predisposition in family members of colorectal cancer patients harboring the p.V600E BRAF mutation: a population-based study. Cancer Epidemiol Biomarkers Prev. 2010;19:1831-1839.
- Zebary A, Omholt K, Vassilaki I, et al. KIT, NRAS, BRAF and PTEN mutations in a sample of Swedish patients with acral lentiginous melanoma. J Dermatol Sci. 2013;72:284-289.
- Si L, Kong Y, Xu X, et al. Prevalence of BRAF V600E mutation in Chinese melanoma patients: large scale analysis of BRAF and NRAS mutations in a 432-case cohort. Eur J Cancer. 2012;48:94-100.
- Safaee Ardekani G, Jafarnejad SM, Khosravi S, et al. Disease progression and patient survival are significantly influenced by BRAF protein expression in primary melanoma. Br J Dermatol. 2013;169:320-328.
Practice Points
- Dermatologists should be aware of the long-term risk of second primary malignancies after an incident melanoma.
- BRAF mutations occur in melanomas and several other cancers. Our study found that melanoma BRAF V600E expression is associated with an increased risk for basal cell carcinomas.
Hydroquinone, found in skin-lightening agents worldwide, linked with increased skin cancer risk
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
FROM SID 2022
Consider the ‘long game’ in tumor management following Mohs surgery
PORTLAND, ORE. – In his nearly 2 decades of dermatology practice, Keith L. Duffy, MD, has seen his share of cases where Mohs surgery was misused or misappropriated.
, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “I want to protect our specialty. I see patients who have dozens of skin cancers. I want to emphasize the long game of management in those patients. You have to think about the tumors in terms of decades.”
In 2012, an ad hoc task force from the American Academy of Dermatology (AAD), the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery developed appropriate use criteria (AUC) for 270 scenarios for which Mohs micrographic surgery (MMS) is frequently considered. The task force used a 9-point scale to rate each indication, as follows:
- A score of 7 to 9: The use of MMS is appropriate for the specific indication and is generally considered acceptable.
- A score of 4 to 6: The use of MMS is uncertain for the specific indication, although its use may be appropriate and acceptable.
- A score of 1 to 3: The use of MMS is inappropriate for the specific indication and is generally not considered acceptable.
These ratings were translated into a free Mohs Surgery Appropriate Use Criteria App developed by the AAD.
Subsequently, Dr. Duffy and colleagues retrospectively examined the University of Utah’s adherence to the Mohs AUC over the course of 3 months. Their analysis, published in 2015, included 1,026 nonmelanoma skin cancers in 724 patients. Of the 1,026 cancers, 350 (34.1%) were treated with MMS. Of these, 339 (96.9%) were deemed appropriate based on the AUC guidelines, 4 (1.1%) were deemed uncertain, and 7 (2%) were deemed inappropriate.
There were also 611 skin cancers that were not treated with Mohs but met criteria for treatment with Mohs. “Most of these were AUC 7 tumors,” Dr. Duffy said. “When I see an AUC 7 tumor, I give high consideration for certain anatomic locations, especially the lower leg, scalp, eyelid, genitalia, ear, hands, and feet. I also think about the patient’s age, the number of skin cancers, and histological characteristics. Consider the long game in management and remember that skin cancer patients can make a near infinite amount of skin cancers, so be conservative when excising skin cancers to preserve precious skin.”
In his opinion, full thickness wounds requiring sutures should be avoided on the scalp and lower leg, if possible. “Most carcinomas in these locations are superficial and not aggressive in immunocompetent patients,” said Dr. Duffy, who said he has had one patient in 12 years who was not a transplant patient who had a metastatic squamous cell carcinoma on the lower leg. “Postop complications can be totally avoided. I don’t worry about these patients bleeding or [about] dehiscence. They can go back and play golf the next day, so you save valuable skin where the real estate is precious. This underscores a practice pearl: Incorporate the Mohs AUC and consideration of anatomic location when considering the most appropriate treatment of skin cancers.”
He also advises dermatologists to consider the histopathologic characteristics of the tumor when treating skin cancers to reduce complications and save tissue, so that patients can resume their lifestyle. “When you read the pathology report, really think about what the dermatopathologist saw under the microscope,” said Dr. Duffy, who is an investigator at the University of Utah’s Huntsman Cancer Institute. He said that he is able to review the slides for 90% of his own cases before surgery. “I’m lucky that way, but if you have any questions, your dermatopathologist should be on speed dial.”
Ultimately, he concluded, proper selection of a treatment modality for a specific tumor and patient rules the day. “Tumors should be thought about in the context of the patient and not as a single or isolated cancer,” he said.
Dr. Duffy reported having no relevant disclosures.
PORTLAND, ORE. – In his nearly 2 decades of dermatology practice, Keith L. Duffy, MD, has seen his share of cases where Mohs surgery was misused or misappropriated.
, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “I want to protect our specialty. I see patients who have dozens of skin cancers. I want to emphasize the long game of management in those patients. You have to think about the tumors in terms of decades.”
In 2012, an ad hoc task force from the American Academy of Dermatology (AAD), the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery developed appropriate use criteria (AUC) for 270 scenarios for which Mohs micrographic surgery (MMS) is frequently considered. The task force used a 9-point scale to rate each indication, as follows:
- A score of 7 to 9: The use of MMS is appropriate for the specific indication and is generally considered acceptable.
- A score of 4 to 6: The use of MMS is uncertain for the specific indication, although its use may be appropriate and acceptable.
- A score of 1 to 3: The use of MMS is inappropriate for the specific indication and is generally not considered acceptable.
These ratings were translated into a free Mohs Surgery Appropriate Use Criteria App developed by the AAD.
Subsequently, Dr. Duffy and colleagues retrospectively examined the University of Utah’s adherence to the Mohs AUC over the course of 3 months. Their analysis, published in 2015, included 1,026 nonmelanoma skin cancers in 724 patients. Of the 1,026 cancers, 350 (34.1%) were treated with MMS. Of these, 339 (96.9%) were deemed appropriate based on the AUC guidelines, 4 (1.1%) were deemed uncertain, and 7 (2%) were deemed inappropriate.
There were also 611 skin cancers that were not treated with Mohs but met criteria for treatment with Mohs. “Most of these were AUC 7 tumors,” Dr. Duffy said. “When I see an AUC 7 tumor, I give high consideration for certain anatomic locations, especially the lower leg, scalp, eyelid, genitalia, ear, hands, and feet. I also think about the patient’s age, the number of skin cancers, and histological characteristics. Consider the long game in management and remember that skin cancer patients can make a near infinite amount of skin cancers, so be conservative when excising skin cancers to preserve precious skin.”
In his opinion, full thickness wounds requiring sutures should be avoided on the scalp and lower leg, if possible. “Most carcinomas in these locations are superficial and not aggressive in immunocompetent patients,” said Dr. Duffy, who said he has had one patient in 12 years who was not a transplant patient who had a metastatic squamous cell carcinoma on the lower leg. “Postop complications can be totally avoided. I don’t worry about these patients bleeding or [about] dehiscence. They can go back and play golf the next day, so you save valuable skin where the real estate is precious. This underscores a practice pearl: Incorporate the Mohs AUC and consideration of anatomic location when considering the most appropriate treatment of skin cancers.”
He also advises dermatologists to consider the histopathologic characteristics of the tumor when treating skin cancers to reduce complications and save tissue, so that patients can resume their lifestyle. “When you read the pathology report, really think about what the dermatopathologist saw under the microscope,” said Dr. Duffy, who is an investigator at the University of Utah’s Huntsman Cancer Institute. He said that he is able to review the slides for 90% of his own cases before surgery. “I’m lucky that way, but if you have any questions, your dermatopathologist should be on speed dial.”
Ultimately, he concluded, proper selection of a treatment modality for a specific tumor and patient rules the day. “Tumors should be thought about in the context of the patient and not as a single or isolated cancer,” he said.
Dr. Duffy reported having no relevant disclosures.
PORTLAND, ORE. – In his nearly 2 decades of dermatology practice, Keith L. Duffy, MD, has seen his share of cases where Mohs surgery was misused or misappropriated.
, Salt Lake City, said at the annual meeting of the Pacific Dermatologic Association. “I want to protect our specialty. I see patients who have dozens of skin cancers. I want to emphasize the long game of management in those patients. You have to think about the tumors in terms of decades.”
In 2012, an ad hoc task force from the American Academy of Dermatology (AAD), the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery developed appropriate use criteria (AUC) for 270 scenarios for which Mohs micrographic surgery (MMS) is frequently considered. The task force used a 9-point scale to rate each indication, as follows:
- A score of 7 to 9: The use of MMS is appropriate for the specific indication and is generally considered acceptable.
- A score of 4 to 6: The use of MMS is uncertain for the specific indication, although its use may be appropriate and acceptable.
- A score of 1 to 3: The use of MMS is inappropriate for the specific indication and is generally not considered acceptable.
These ratings were translated into a free Mohs Surgery Appropriate Use Criteria App developed by the AAD.
Subsequently, Dr. Duffy and colleagues retrospectively examined the University of Utah’s adherence to the Mohs AUC over the course of 3 months. Their analysis, published in 2015, included 1,026 nonmelanoma skin cancers in 724 patients. Of the 1,026 cancers, 350 (34.1%) were treated with MMS. Of these, 339 (96.9%) were deemed appropriate based on the AUC guidelines, 4 (1.1%) were deemed uncertain, and 7 (2%) were deemed inappropriate.
There were also 611 skin cancers that were not treated with Mohs but met criteria for treatment with Mohs. “Most of these were AUC 7 tumors,” Dr. Duffy said. “When I see an AUC 7 tumor, I give high consideration for certain anatomic locations, especially the lower leg, scalp, eyelid, genitalia, ear, hands, and feet. I also think about the patient’s age, the number of skin cancers, and histological characteristics. Consider the long game in management and remember that skin cancer patients can make a near infinite amount of skin cancers, so be conservative when excising skin cancers to preserve precious skin.”
In his opinion, full thickness wounds requiring sutures should be avoided on the scalp and lower leg, if possible. “Most carcinomas in these locations are superficial and not aggressive in immunocompetent patients,” said Dr. Duffy, who said he has had one patient in 12 years who was not a transplant patient who had a metastatic squamous cell carcinoma on the lower leg. “Postop complications can be totally avoided. I don’t worry about these patients bleeding or [about] dehiscence. They can go back and play golf the next day, so you save valuable skin where the real estate is precious. This underscores a practice pearl: Incorporate the Mohs AUC and consideration of anatomic location when considering the most appropriate treatment of skin cancers.”
He also advises dermatologists to consider the histopathologic characteristics of the tumor when treating skin cancers to reduce complications and save tissue, so that patients can resume their lifestyle. “When you read the pathology report, really think about what the dermatopathologist saw under the microscope,” said Dr. Duffy, who is an investigator at the University of Utah’s Huntsman Cancer Institute. He said that he is able to review the slides for 90% of his own cases before surgery. “I’m lucky that way, but if you have any questions, your dermatopathologist should be on speed dial.”
Ultimately, he concluded, proper selection of a treatment modality for a specific tumor and patient rules the day. “Tumors should be thought about in the context of the patient and not as a single or isolated cancer,” he said.
Dr. Duffy reported having no relevant disclosures.
AT PDA 2022
Second opinions on melanocytic lesions swayed when first opinion is known
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Getting cancer research on track again may require a ‘behemoth’ effort
In 2016, as vice president, Joe Biden launched the Cancer Moonshot program just 1 year after his son Beau died from glioblastoma multiforme. His objective, he said, was to “cure” cancer, but to get close to that goal, to get cancer research just back up to pre-COVID-19 pandemic levels.
There has been a significant decrease in the launch of new clinical trials for cancer and biologic therapies since 2020. “That can affect every aspect of our research operation. It really affected our capacity to continue to move forward at a fast pace. It will require a behemoth effort to get back to pre-COVID times,” said Tanios S. Bekaii-Saab, MD, leader of the gastrointestinal cancer program at Mayo Clinic in Phoenix.
Congress passed the 21st Century Cures Act in 2016 authorizing $1.8 billion for Cancer Moonshot over 7 years. More recently, the program received $194 million from the $6.9 billion National Cancer Institute budget in FY 2022.
Joseph Alvarnas, MD, a hematologist oncologist and vice president of government affairs at City of Hope, Duarte, Calif., sees the Moonshot budget as a potential shortcoming.
“The priorities are well founded and based on what we would think are the most important things to cover, but, if we’re going to achieve these extraordinarily ambitious goals of halving cancer mortality and serving communities more equitably, it’s going to need more funding positioned at making these things real,” he said.
Moonshot is being positioned as an opportunity to double down on efforts started in 2016, but treating cancer is complex and goes well beyond funding new research.
“We know that we have amazing research and progress around innovations that will drive us toward the goal of reducing the death rate from cancer. But we also know that we have tools that aren’t reaching all parts of the country, so we have a great opportunity to make sure that we’re doing all we can to prevent, detect and treat cancer,” Dr. Carnival said.
Can cancer be cured?
The Biden administration relaunched Moonshot in 2022 with newly defined goals: Cut the rate of cancer-related deaths in half within 25 years; improve the experience of people with cancer, cancer survivors, and their families; and “end cancer as we know it,” President Biden said in a press conference in February.
Cancer is the second leading cause of death in the United States after heart disease, but it may indeed be possible to cut the total number of cancer-related deaths in half over the next 25 years.
“As a hematologist who’s been involved in both research and clinical care, I think it’s important to realize this is actually doable. Between 1990 and 2020 cancer mortality rates decreased by 31%, and in the last American Cancer Society’s annual report, mortality rates dropped by the largest percentages for 2 consecutive years in a row. The question shifts now from ‘Is this possible? to ‘How do we ensure that it’s possible?’ The spirit of Cancer Moonshot 2.0 is identifying the multiple paths to move this effort forward,” Dr. Alvarnas said.
But without a significant infusion of cash for research, it’s doubtful cancer-related deaths will drop by 50% over the next 25 years.
“There are a lot of big and lofty goals in Cancer Moonshot, and the words ‘ending cancer,’ well those are big words,” Dr. Bekaii-Saab said. “The reality is how do we measure in 25 years the impact of this today? I think it will require significantly more funding over the next few years to achieve the goals set by the Moonshot. Otherwise it will be a 7-year done deal that will accrue a lot of great numbers but won’t make a dent in those goals for the next 25 years. To stop it at some point and not invest more into it, we will probably lose most of the benefit.”
Closing the loop on data sharing
Moonshot has been instrumental in fostering research collaborations by encouraging data sharing among scientists.
“It also brought together a new way for the National Cancer Institute and Department of Energy to drive progress on some of the big data initiatives. The initial Cancer Moonshot infused a sense of urgency and hope into this effort,” said Danielle Carnival, PhD, coordinator of Cancer Moonshot.
Between 2017 and 2022, Cancer Moonshot created more than 70 consortiums or programs, and funded about 240 research projects. Its fundamental goals of improving data sharing and encouraging collaboration are very important, Dr. Bekaii-Saab said.
“Because, historically, what happens with cancer is that researchers compete for resources...and they become very protective of their data. Sharing gets more difficult, collaborations become more onerous, and it becomes counterproductive,” he said.
Dr. Bekaii-Saab highlighted two networks created specifically for data sharing. They include the Human Tumor Atlas for cellular, morphological, and molecular tumor data, and PDXNet, a patient derived xenograft research network.
A shift in funding priorities?
Cancer funding has been stagnant for years. When adjusted for growth, it hasn’t had a significant infusion of funding since at least 2003—at least in relative terms, Dr. Bekaii-Saab said. “This affects a lot of the things we do, including NCI-funded clinical trials. It pushes us to work with the private sector, which is not necessarily a detriment, but it doesn’t advance the academic mission at the same level. So, overall, I wouldn’t call it tragic, but I do think we’re falling behind,” he said.
“I think when we do the process for the budget for FY24 and after we’ve had time to really explore the best ideas and build the foundation for some of these new aspects of the Cancer Moonshot, we hope to have something more concrete going toward these efforts,” Dr. Carnival said.
But in addition to funding, Dr. Alvarnas says, it is equally important to address gaps in care. Not all patients have access to existing cancer treatments.
“The great challenge to us in the 2020s is not only about developing new and more effective technologies, but also in doing a better job of getting existing life-saving treatments into the hands of underserved populations. One of the really positive challenges set forth by the Biden administration is the idea that financing care equity is as important, if not more so, than advancing technologies. If there’s been stagnation, it’s because from a government and resourcing point of view, that priority has been ineffectively supported financially.”
The pandemic stymies cancer research
The pandemic has had a significant impact on cancer research. As in other fields, it disrupted ongoing research, but it may have also contributed to the loss of employees who resigned in what’s been called the “Great Resignation.” “A lot of employees just decided to change jobs in the middle of the pandemic, which led to a cancer research staffing crisis,” Dr. Bekaii-Saab said.
“We all recognized that turning so much of the attention of the entire biomedical research engine and health system to the COVID-19 pandemic would have an impact across cancer research, screenings and care,” Dr. Carnival said. “There is work to do to get us back to whole, but from a research perspective, we’ve seen a reorientation of the trial networks we were using for COVID-19 research, back to their initial purpose. Some of those are cancer and oncology networks, so we’re excited about that and fully believe that we can catch up.”
But then there’s also the impact the pandemic has had on cancer patients who delayed their care at the primary level. This, Dr. Bekaii-Saab fears, will lead to more patients presenting with more advanced disease in years to come. “One of the biggest problems was that a lot of patients delayed their care at the primary level. My biggest concern is that in the years to come we will see a lot more patients presenting with more advanced cancer.”
In 2016, as vice president, Joe Biden launched the Cancer Moonshot program just 1 year after his son Beau died from glioblastoma multiforme. His objective, he said, was to “cure” cancer, but to get close to that goal, to get cancer research just back up to pre-COVID-19 pandemic levels.
There has been a significant decrease in the launch of new clinical trials for cancer and biologic therapies since 2020. “That can affect every aspect of our research operation. It really affected our capacity to continue to move forward at a fast pace. It will require a behemoth effort to get back to pre-COVID times,” said Tanios S. Bekaii-Saab, MD, leader of the gastrointestinal cancer program at Mayo Clinic in Phoenix.
Congress passed the 21st Century Cures Act in 2016 authorizing $1.8 billion for Cancer Moonshot over 7 years. More recently, the program received $194 million from the $6.9 billion National Cancer Institute budget in FY 2022.
Joseph Alvarnas, MD, a hematologist oncologist and vice president of government affairs at City of Hope, Duarte, Calif., sees the Moonshot budget as a potential shortcoming.
“The priorities are well founded and based on what we would think are the most important things to cover, but, if we’re going to achieve these extraordinarily ambitious goals of halving cancer mortality and serving communities more equitably, it’s going to need more funding positioned at making these things real,” he said.
Moonshot is being positioned as an opportunity to double down on efforts started in 2016, but treating cancer is complex and goes well beyond funding new research.
“We know that we have amazing research and progress around innovations that will drive us toward the goal of reducing the death rate from cancer. But we also know that we have tools that aren’t reaching all parts of the country, so we have a great opportunity to make sure that we’re doing all we can to prevent, detect and treat cancer,” Dr. Carnival said.
Can cancer be cured?
The Biden administration relaunched Moonshot in 2022 with newly defined goals: Cut the rate of cancer-related deaths in half within 25 years; improve the experience of people with cancer, cancer survivors, and their families; and “end cancer as we know it,” President Biden said in a press conference in February.
Cancer is the second leading cause of death in the United States after heart disease, but it may indeed be possible to cut the total number of cancer-related deaths in half over the next 25 years.
“As a hematologist who’s been involved in both research and clinical care, I think it’s important to realize this is actually doable. Between 1990 and 2020 cancer mortality rates decreased by 31%, and in the last American Cancer Society’s annual report, mortality rates dropped by the largest percentages for 2 consecutive years in a row. The question shifts now from ‘Is this possible? to ‘How do we ensure that it’s possible?’ The spirit of Cancer Moonshot 2.0 is identifying the multiple paths to move this effort forward,” Dr. Alvarnas said.
But without a significant infusion of cash for research, it’s doubtful cancer-related deaths will drop by 50% over the next 25 years.
“There are a lot of big and lofty goals in Cancer Moonshot, and the words ‘ending cancer,’ well those are big words,” Dr. Bekaii-Saab said. “The reality is how do we measure in 25 years the impact of this today? I think it will require significantly more funding over the next few years to achieve the goals set by the Moonshot. Otherwise it will be a 7-year done deal that will accrue a lot of great numbers but won’t make a dent in those goals for the next 25 years. To stop it at some point and not invest more into it, we will probably lose most of the benefit.”
Closing the loop on data sharing
Moonshot has been instrumental in fostering research collaborations by encouraging data sharing among scientists.
“It also brought together a new way for the National Cancer Institute and Department of Energy to drive progress on some of the big data initiatives. The initial Cancer Moonshot infused a sense of urgency and hope into this effort,” said Danielle Carnival, PhD, coordinator of Cancer Moonshot.
Between 2017 and 2022, Cancer Moonshot created more than 70 consortiums or programs, and funded about 240 research projects. Its fundamental goals of improving data sharing and encouraging collaboration are very important, Dr. Bekaii-Saab said.
“Because, historically, what happens with cancer is that researchers compete for resources...and they become very protective of their data. Sharing gets more difficult, collaborations become more onerous, and it becomes counterproductive,” he said.
Dr. Bekaii-Saab highlighted two networks created specifically for data sharing. They include the Human Tumor Atlas for cellular, morphological, and molecular tumor data, and PDXNet, a patient derived xenograft research network.
A shift in funding priorities?
Cancer funding has been stagnant for years. When adjusted for growth, it hasn’t had a significant infusion of funding since at least 2003—at least in relative terms, Dr. Bekaii-Saab said. “This affects a lot of the things we do, including NCI-funded clinical trials. It pushes us to work with the private sector, which is not necessarily a detriment, but it doesn’t advance the academic mission at the same level. So, overall, I wouldn’t call it tragic, but I do think we’re falling behind,” he said.
“I think when we do the process for the budget for FY24 and after we’ve had time to really explore the best ideas and build the foundation for some of these new aspects of the Cancer Moonshot, we hope to have something more concrete going toward these efforts,” Dr. Carnival said.
But in addition to funding, Dr. Alvarnas says, it is equally important to address gaps in care. Not all patients have access to existing cancer treatments.
“The great challenge to us in the 2020s is not only about developing new and more effective technologies, but also in doing a better job of getting existing life-saving treatments into the hands of underserved populations. One of the really positive challenges set forth by the Biden administration is the idea that financing care equity is as important, if not more so, than advancing technologies. If there’s been stagnation, it’s because from a government and resourcing point of view, that priority has been ineffectively supported financially.”
The pandemic stymies cancer research
The pandemic has had a significant impact on cancer research. As in other fields, it disrupted ongoing research, but it may have also contributed to the loss of employees who resigned in what’s been called the “Great Resignation.” “A lot of employees just decided to change jobs in the middle of the pandemic, which led to a cancer research staffing crisis,” Dr. Bekaii-Saab said.
“We all recognized that turning so much of the attention of the entire biomedical research engine and health system to the COVID-19 pandemic would have an impact across cancer research, screenings and care,” Dr. Carnival said. “There is work to do to get us back to whole, but from a research perspective, we’ve seen a reorientation of the trial networks we were using for COVID-19 research, back to their initial purpose. Some of those are cancer and oncology networks, so we’re excited about that and fully believe that we can catch up.”
But then there’s also the impact the pandemic has had on cancer patients who delayed their care at the primary level. This, Dr. Bekaii-Saab fears, will lead to more patients presenting with more advanced disease in years to come. “One of the biggest problems was that a lot of patients delayed their care at the primary level. My biggest concern is that in the years to come we will see a lot more patients presenting with more advanced cancer.”
In 2016, as vice president, Joe Biden launched the Cancer Moonshot program just 1 year after his son Beau died from glioblastoma multiforme. His objective, he said, was to “cure” cancer, but to get close to that goal, to get cancer research just back up to pre-COVID-19 pandemic levels.
There has been a significant decrease in the launch of new clinical trials for cancer and biologic therapies since 2020. “That can affect every aspect of our research operation. It really affected our capacity to continue to move forward at a fast pace. It will require a behemoth effort to get back to pre-COVID times,” said Tanios S. Bekaii-Saab, MD, leader of the gastrointestinal cancer program at Mayo Clinic in Phoenix.
Congress passed the 21st Century Cures Act in 2016 authorizing $1.8 billion for Cancer Moonshot over 7 years. More recently, the program received $194 million from the $6.9 billion National Cancer Institute budget in FY 2022.
Joseph Alvarnas, MD, a hematologist oncologist and vice president of government affairs at City of Hope, Duarte, Calif., sees the Moonshot budget as a potential shortcoming.
“The priorities are well founded and based on what we would think are the most important things to cover, but, if we’re going to achieve these extraordinarily ambitious goals of halving cancer mortality and serving communities more equitably, it’s going to need more funding positioned at making these things real,” he said.
Moonshot is being positioned as an opportunity to double down on efforts started in 2016, but treating cancer is complex and goes well beyond funding new research.
“We know that we have amazing research and progress around innovations that will drive us toward the goal of reducing the death rate from cancer. But we also know that we have tools that aren’t reaching all parts of the country, so we have a great opportunity to make sure that we’re doing all we can to prevent, detect and treat cancer,” Dr. Carnival said.
Can cancer be cured?
The Biden administration relaunched Moonshot in 2022 with newly defined goals: Cut the rate of cancer-related deaths in half within 25 years; improve the experience of people with cancer, cancer survivors, and their families; and “end cancer as we know it,” President Biden said in a press conference in February.
Cancer is the second leading cause of death in the United States after heart disease, but it may indeed be possible to cut the total number of cancer-related deaths in half over the next 25 years.
“As a hematologist who’s been involved in both research and clinical care, I think it’s important to realize this is actually doable. Between 1990 and 2020 cancer mortality rates decreased by 31%, and in the last American Cancer Society’s annual report, mortality rates dropped by the largest percentages for 2 consecutive years in a row. The question shifts now from ‘Is this possible? to ‘How do we ensure that it’s possible?’ The spirit of Cancer Moonshot 2.0 is identifying the multiple paths to move this effort forward,” Dr. Alvarnas said.
But without a significant infusion of cash for research, it’s doubtful cancer-related deaths will drop by 50% over the next 25 years.
“There are a lot of big and lofty goals in Cancer Moonshot, and the words ‘ending cancer,’ well those are big words,” Dr. Bekaii-Saab said. “The reality is how do we measure in 25 years the impact of this today? I think it will require significantly more funding over the next few years to achieve the goals set by the Moonshot. Otherwise it will be a 7-year done deal that will accrue a lot of great numbers but won’t make a dent in those goals for the next 25 years. To stop it at some point and not invest more into it, we will probably lose most of the benefit.”
Closing the loop on data sharing
Moonshot has been instrumental in fostering research collaborations by encouraging data sharing among scientists.
“It also brought together a new way for the National Cancer Institute and Department of Energy to drive progress on some of the big data initiatives. The initial Cancer Moonshot infused a sense of urgency and hope into this effort,” said Danielle Carnival, PhD, coordinator of Cancer Moonshot.
Between 2017 and 2022, Cancer Moonshot created more than 70 consortiums or programs, and funded about 240 research projects. Its fundamental goals of improving data sharing and encouraging collaboration are very important, Dr. Bekaii-Saab said.
“Because, historically, what happens with cancer is that researchers compete for resources...and they become very protective of their data. Sharing gets more difficult, collaborations become more onerous, and it becomes counterproductive,” he said.
Dr. Bekaii-Saab highlighted two networks created specifically for data sharing. They include the Human Tumor Atlas for cellular, morphological, and molecular tumor data, and PDXNet, a patient derived xenograft research network.
A shift in funding priorities?
Cancer funding has been stagnant for years. When adjusted for growth, it hasn’t had a significant infusion of funding since at least 2003—at least in relative terms, Dr. Bekaii-Saab said. “This affects a lot of the things we do, including NCI-funded clinical trials. It pushes us to work with the private sector, which is not necessarily a detriment, but it doesn’t advance the academic mission at the same level. So, overall, I wouldn’t call it tragic, but I do think we’re falling behind,” he said.
“I think when we do the process for the budget for FY24 and after we’ve had time to really explore the best ideas and build the foundation for some of these new aspects of the Cancer Moonshot, we hope to have something more concrete going toward these efforts,” Dr. Carnival said.
But in addition to funding, Dr. Alvarnas says, it is equally important to address gaps in care. Not all patients have access to existing cancer treatments.
“The great challenge to us in the 2020s is not only about developing new and more effective technologies, but also in doing a better job of getting existing life-saving treatments into the hands of underserved populations. One of the really positive challenges set forth by the Biden administration is the idea that financing care equity is as important, if not more so, than advancing technologies. If there’s been stagnation, it’s because from a government and resourcing point of view, that priority has been ineffectively supported financially.”
The pandemic stymies cancer research
The pandemic has had a significant impact on cancer research. As in other fields, it disrupted ongoing research, but it may have also contributed to the loss of employees who resigned in what’s been called the “Great Resignation.” “A lot of employees just decided to change jobs in the middle of the pandemic, which led to a cancer research staffing crisis,” Dr. Bekaii-Saab said.
“We all recognized that turning so much of the attention of the entire biomedical research engine and health system to the COVID-19 pandemic would have an impact across cancer research, screenings and care,” Dr. Carnival said. “There is work to do to get us back to whole, but from a research perspective, we’ve seen a reorientation of the trial networks we were using for COVID-19 research, back to their initial purpose. Some of those are cancer and oncology networks, so we’re excited about that and fully believe that we can catch up.”
But then there’s also the impact the pandemic has had on cancer patients who delayed their care at the primary level. This, Dr. Bekaii-Saab fears, will lead to more patients presenting with more advanced disease in years to come. “One of the biggest problems was that a lot of patients delayed their care at the primary level. My biggest concern is that in the years to come we will see a lot more patients presenting with more advanced cancer.”