User login
Is Lp(a) a marker for aortic calcium onset?
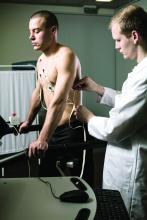
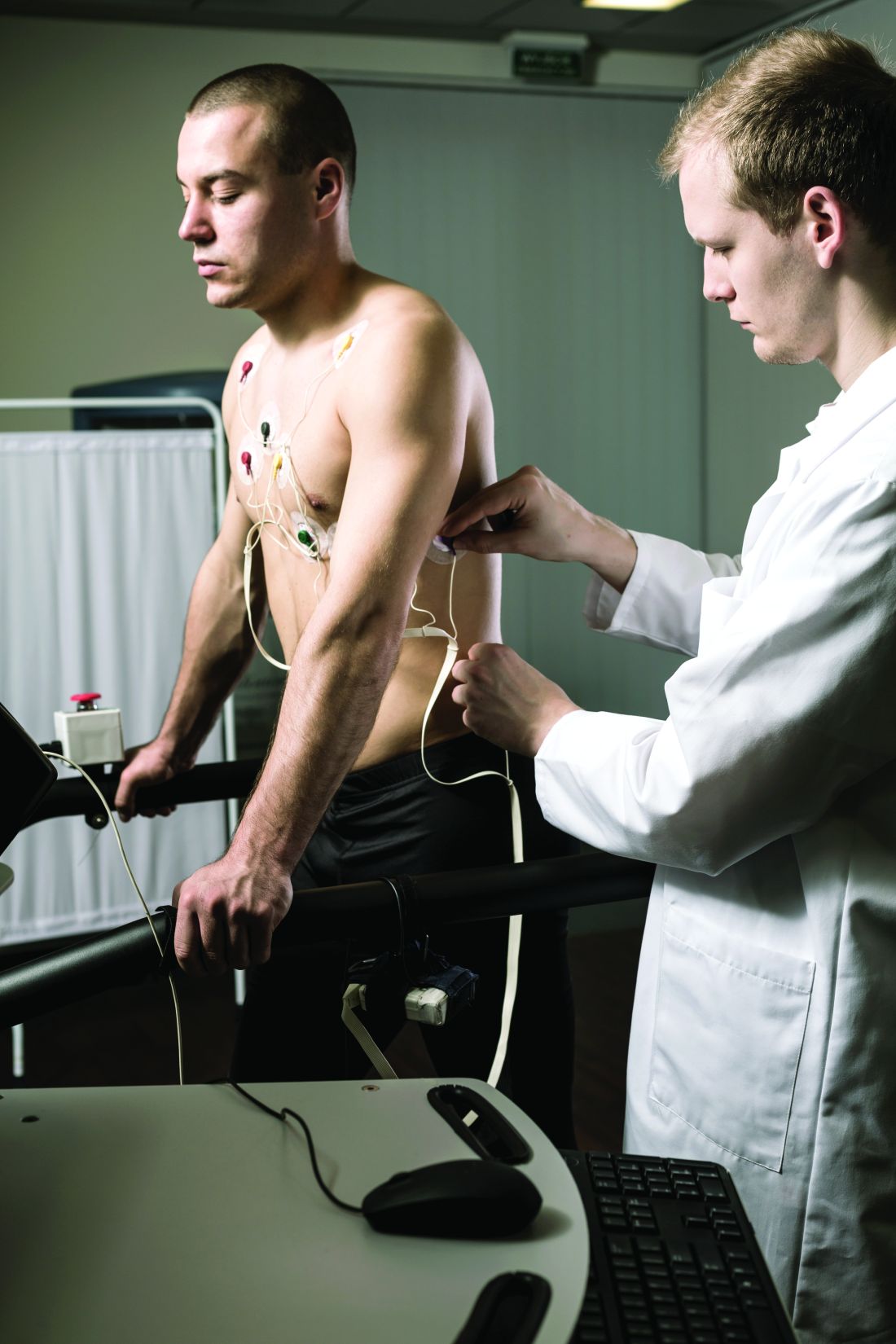
Lipoprotein(a) has long been thought to be a potential marker of aortic valve disease, and the population-based Rotterdam Study in the Netherlands has reported that Lp(a) has a strong association with new-onset aortic valve calcium (AVC), but not necessarily with progression of aortic valve disease.
Reporting in the European Heart Journal, the study authors analyzed data on 922 participants in the Rotterdam Study whose Lp(a) was measured along with a computed tomography scan upon enrollment, followed by CT scan 14 years later. At baseline, 702 participants didn’t have AVC, but the follow-up scan identified new-onset AVC in 415 (59.1%).
The investigators found an association between Lp(a) concentration and baseline AVC, with an odds ratio of 1.43 for each 50 mg/dL higher Lp(a) (95% confidence interval, 1.15-1.79), as well as new-onset AVC, with an OR of 1.30 for each 50 mg/dL increase in Lp(a) (95% CI, 1.02-1.65). However, the study found no association between rising Lp(a) levels and AVC progression; it found only an association between baseline AVC score and progression (P < .001).
‘Trigger’ for calcification but not progression
“This suggests that Lp(a) is an important trigger in the initiation of aortic valve calcification, but once the valve is calcified, disease progression may be primarily driven by other factors such as the baseline calcium burden of the valve and likely other unknown factors,” senior study author Daniel Bos, MD, PhD, said in e-mailed comments.
Dr. Bos and coauthors claim this is the first study to show that even minor AVC progresses independently of Lp(a).
“There are previous studies that showed a possible relationship between Lp(a) [and] progression of aortic valve calcium,” he said. “Our study suggests that the most meaningful benefit of Lp(a) lowering may actually be prior to the onset of aortic valve calcification.”
While no treatments have been approved for lowering Lp(a), the study findings could be meaningful if trials, including the ongoing phase 3 Lp(a) HORIZON trial of the investigational antisense agent pelacarsen (NCT04023552), show promising results, Dr. Bos said. Citing Lp(a) HORIZON, he said, “If the study shows Lp(a) lowering leads to a reduction in incident cardiovascular disease, similar strategies may be applied to prevent, rather than slow down, progression of aortic valve calcification.”
Dr. Bos called the Rotterdam Study results “an important first pointer into that direction.” He added, “We will need randomized trials to provide a definitive answer to the question whether Lp(a) lowering may prevent aortic valve calcium.”
Focus on AVC is study ‘weakness’
The study findings raise a key question for clinical trials of investigative Lp(a)-lowering therapies as well as how to use those therapies to treat aortic valve disease, said Christie Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston.
The findings could be “problematic” for these clinical trials, he said. “This study is just looking at calcium progression,” Dr. Ballantyne noted. “What we want to know about clinically is the progression to aortic stenosis, and then in particular to progression from mild disease to moderate or severe disease, because once you get into more severe disease, one has to do an intervention with either surgery or TAVR [transcatheter aortic valve replacement].”
He considered the study’s focus on AVC rather than aortic valve function a weakness and noted that only 14 study participants had TAVR. “We’re going to need much bigger numbers to look into this question of progression, including progression to severe diseases,” he said.
However, the Rotterdam Study showed the importance of CT in evaluating AVC, which can easily be done in other trials to further explore the association between Lp(a) and AVC, Dr. Ballantyne said.
Dr. Bos has no relevant disclosures. Study coauthors disclosed relationships with Amgen, Sanofi, Reservlogix, Athera, Experio, Novartis and Ionis Pharmaceuticals. Dr. Ballantyne disclosed relationships with Amgen and Novartis.
Lipoprotein(a) has long been thought to be a potential marker of aortic valve disease, and the population-based Rotterdam Study in the Netherlands has reported that Lp(a) has a strong association with new-onset aortic valve calcium (AVC), but not necessarily with progression of aortic valve disease.
Reporting in the European Heart Journal, the study authors analyzed data on 922 participants in the Rotterdam Study whose Lp(a) was measured along with a computed tomography scan upon enrollment, followed by CT scan 14 years later. At baseline, 702 participants didn’t have AVC, but the follow-up scan identified new-onset AVC in 415 (59.1%).
The investigators found an association between Lp(a) concentration and baseline AVC, with an odds ratio of 1.43 for each 50 mg/dL higher Lp(a) (95% confidence interval, 1.15-1.79), as well as new-onset AVC, with an OR of 1.30 for each 50 mg/dL increase in Lp(a) (95% CI, 1.02-1.65). However, the study found no association between rising Lp(a) levels and AVC progression; it found only an association between baseline AVC score and progression (P < .001).
‘Trigger’ for calcification but not progression
“This suggests that Lp(a) is an important trigger in the initiation of aortic valve calcification, but once the valve is calcified, disease progression may be primarily driven by other factors such as the baseline calcium burden of the valve and likely other unknown factors,” senior study author Daniel Bos, MD, PhD, said in e-mailed comments.
Dr. Bos and coauthors claim this is the first study to show that even minor AVC progresses independently of Lp(a).
“There are previous studies that showed a possible relationship between Lp(a) [and] progression of aortic valve calcium,” he said. “Our study suggests that the most meaningful benefit of Lp(a) lowering may actually be prior to the onset of aortic valve calcification.”
While no treatments have been approved for lowering Lp(a), the study findings could be meaningful if trials, including the ongoing phase 3 Lp(a) HORIZON trial of the investigational antisense agent pelacarsen (NCT04023552), show promising results, Dr. Bos said. Citing Lp(a) HORIZON, he said, “If the study shows Lp(a) lowering leads to a reduction in incident cardiovascular disease, similar strategies may be applied to prevent, rather than slow down, progression of aortic valve calcification.”
Dr. Bos called the Rotterdam Study results “an important first pointer into that direction.” He added, “We will need randomized trials to provide a definitive answer to the question whether Lp(a) lowering may prevent aortic valve calcium.”
Focus on AVC is study ‘weakness’
The study findings raise a key question for clinical trials of investigative Lp(a)-lowering therapies as well as how to use those therapies to treat aortic valve disease, said Christie Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston.
The findings could be “problematic” for these clinical trials, he said. “This study is just looking at calcium progression,” Dr. Ballantyne noted. “What we want to know about clinically is the progression to aortic stenosis, and then in particular to progression from mild disease to moderate or severe disease, because once you get into more severe disease, one has to do an intervention with either surgery or TAVR [transcatheter aortic valve replacement].”
He considered the study’s focus on AVC rather than aortic valve function a weakness and noted that only 14 study participants had TAVR. “We’re going to need much bigger numbers to look into this question of progression, including progression to severe diseases,” he said.
However, the Rotterdam Study showed the importance of CT in evaluating AVC, which can easily be done in other trials to further explore the association between Lp(a) and AVC, Dr. Ballantyne said.
Dr. Bos has no relevant disclosures. Study coauthors disclosed relationships with Amgen, Sanofi, Reservlogix, Athera, Experio, Novartis and Ionis Pharmaceuticals. Dr. Ballantyne disclosed relationships with Amgen and Novartis.
Lipoprotein(a) has long been thought to be a potential marker of aortic valve disease, and the population-based Rotterdam Study in the Netherlands has reported that Lp(a) has a strong association with new-onset aortic valve calcium (AVC), but not necessarily with progression of aortic valve disease.
Reporting in the European Heart Journal, the study authors analyzed data on 922 participants in the Rotterdam Study whose Lp(a) was measured along with a computed tomography scan upon enrollment, followed by CT scan 14 years later. At baseline, 702 participants didn’t have AVC, but the follow-up scan identified new-onset AVC in 415 (59.1%).
The investigators found an association between Lp(a) concentration and baseline AVC, with an odds ratio of 1.43 for each 50 mg/dL higher Lp(a) (95% confidence interval, 1.15-1.79), as well as new-onset AVC, with an OR of 1.30 for each 50 mg/dL increase in Lp(a) (95% CI, 1.02-1.65). However, the study found no association between rising Lp(a) levels and AVC progression; it found only an association between baseline AVC score and progression (P < .001).
‘Trigger’ for calcification but not progression
“This suggests that Lp(a) is an important trigger in the initiation of aortic valve calcification, but once the valve is calcified, disease progression may be primarily driven by other factors such as the baseline calcium burden of the valve and likely other unknown factors,” senior study author Daniel Bos, MD, PhD, said in e-mailed comments.
Dr. Bos and coauthors claim this is the first study to show that even minor AVC progresses independently of Lp(a).
“There are previous studies that showed a possible relationship between Lp(a) [and] progression of aortic valve calcium,” he said. “Our study suggests that the most meaningful benefit of Lp(a) lowering may actually be prior to the onset of aortic valve calcification.”
While no treatments have been approved for lowering Lp(a), the study findings could be meaningful if trials, including the ongoing phase 3 Lp(a) HORIZON trial of the investigational antisense agent pelacarsen (NCT04023552), show promising results, Dr. Bos said. Citing Lp(a) HORIZON, he said, “If the study shows Lp(a) lowering leads to a reduction in incident cardiovascular disease, similar strategies may be applied to prevent, rather than slow down, progression of aortic valve calcification.”
Dr. Bos called the Rotterdam Study results “an important first pointer into that direction.” He added, “We will need randomized trials to provide a definitive answer to the question whether Lp(a) lowering may prevent aortic valve calcium.”
Focus on AVC is study ‘weakness’
The study findings raise a key question for clinical trials of investigative Lp(a)-lowering therapies as well as how to use those therapies to treat aortic valve disease, said Christie Ballantyne, MD, chief of cardiology at Baylor College of Medicine in Houston.
The findings could be “problematic” for these clinical trials, he said. “This study is just looking at calcium progression,” Dr. Ballantyne noted. “What we want to know about clinically is the progression to aortic stenosis, and then in particular to progression from mild disease to moderate or severe disease, because once you get into more severe disease, one has to do an intervention with either surgery or TAVR [transcatheter aortic valve replacement].”
He considered the study’s focus on AVC rather than aortic valve function a weakness and noted that only 14 study participants had TAVR. “We’re going to need much bigger numbers to look into this question of progression, including progression to severe diseases,” he said.
However, the Rotterdam Study showed the importance of CT in evaluating AVC, which can easily be done in other trials to further explore the association between Lp(a) and AVC, Dr. Ballantyne said.
Dr. Bos has no relevant disclosures. Study coauthors disclosed relationships with Amgen, Sanofi, Reservlogix, Athera, Experio, Novartis and Ionis Pharmaceuticals. Dr. Ballantyne disclosed relationships with Amgen and Novartis.
FROM THE EUROPEAN HEART JOURNAL
RADIANCE II: Positive signal for ultrasound renal denervation
Top-line results released on July 26 from the RADIANCE II trial show the Paradise ultrasound renal denervation system significantly reduces daytime ambulatory systolic blood pressure, compared with a sham procedure at 2 months in patients with mild to moderate uncontrolled hypertension.
The trial was conducted in 224 patients who were previously treated with up to two medications and were randomized while off medication at more than 60 centers in 8 countries. No further details or results were provided.
The pivotal RADIANCE II trial, required for FDA approval, is the third and largest randomized, sham-controlled study following positive results reported by RADIANCE-HTN SOLO and RADIANCE-HTN TRIO, ReCor Medical and its subsidiary Otsuka Medical Devices noted in the announcement.
The field of renal denervation fell out of favor after the largest trial in 535 patients, SYMPLICITY HTN-3, failed to show a significant reduction in systolic blood pressure at 6 months, compared with sham control in resistant hypertension.
A version of this article first appeared on Medscape.com.
Top-line results released on July 26 from the RADIANCE II trial show the Paradise ultrasound renal denervation system significantly reduces daytime ambulatory systolic blood pressure, compared with a sham procedure at 2 months in patients with mild to moderate uncontrolled hypertension.
The trial was conducted in 224 patients who were previously treated with up to two medications and were randomized while off medication at more than 60 centers in 8 countries. No further details or results were provided.
The pivotal RADIANCE II trial, required for FDA approval, is the third and largest randomized, sham-controlled study following positive results reported by RADIANCE-HTN SOLO and RADIANCE-HTN TRIO, ReCor Medical and its subsidiary Otsuka Medical Devices noted in the announcement.
The field of renal denervation fell out of favor after the largest trial in 535 patients, SYMPLICITY HTN-3, failed to show a significant reduction in systolic blood pressure at 6 months, compared with sham control in resistant hypertension.
A version of this article first appeared on Medscape.com.
Top-line results released on July 26 from the RADIANCE II trial show the Paradise ultrasound renal denervation system significantly reduces daytime ambulatory systolic blood pressure, compared with a sham procedure at 2 months in patients with mild to moderate uncontrolled hypertension.
The trial was conducted in 224 patients who were previously treated with up to two medications and were randomized while off medication at more than 60 centers in 8 countries. No further details or results were provided.
The pivotal RADIANCE II trial, required for FDA approval, is the third and largest randomized, sham-controlled study following positive results reported by RADIANCE-HTN SOLO and RADIANCE-HTN TRIO, ReCor Medical and its subsidiary Otsuka Medical Devices noted in the announcement.
The field of renal denervation fell out of favor after the largest trial in 535 patients, SYMPLICITY HTN-3, failed to show a significant reduction in systolic blood pressure at 6 months, compared with sham control in resistant hypertension.
A version of this article first appeared on Medscape.com.
‘Stunning variation’ in CV test, procedure costs revealed at top U.S. hospitals
Wide variation in the cost of common cardiovascular (CV) tests and procedures, from stress tests to coronary interventions, was revealed in a cross-sectional analysis based on publicly available data from 20 top-ranked hospitals in the United States.
The analysis also suggested a low level of compliance with the 2021 Hospital Price Transparency Final Rule among the 20 centers.
“The variation we found in payer-negotiated prices for identical cardiovascular tests and procedures was stunning,” Rishi K. Wadhera, MD, MPP, MPhil, Beth Israel Deaconess Medical Center, Boston, told this news organization.
“For example, there was a 10-fold difference in the median price of an echocardiogram, and these differences were even larger for common procedures” such as percutaneous coronary intervention (PCI) and pacemaker implantation, he said. “It’s hard to argue that this variation reflects quality of care, given that we looked at a top group of highly ranked hospitals.”
“Even more striking was how the price of a cardiovascular test within the very same hospital could differ across commercial insurance companies,” he said. “For example, the price of a stress test varied 5-fold in one hospital, and in another hospital, more than 4-fold for a coronary angiogram.”
Dr. Wadhera is senior author on the study published online as a research letter in JAMA Internal Medicine, with lead author Andrew S. Oseran, MD, MBA, also from Beth Israel Deaconess Medical Center.
Difficulties with data, interpretation
The researchers looked at payer and self-pay cash prices for noninvasive and invasive CV tests and procedures at the U.S. News & World Report 2021 top 20–ranked U.S. hospitals, based in part on Current Procedural Terminology codes.
Price differences among the hospitals were derived from median negotiated prices for each test and procedure at the centers across all payers. The interquartile ratio (IQR) of prices for each test or procedure across payers was used to evaluate within-hospital price variation.
“Only 80% of the hospitals reported prices for some cardiovascular tests and procedures,” Dr. Wadhera said. “For the most part, even among the hospitals that did report this information, it was extremely challenging to navigate and interpret the data provided.”
Further, the team found that only 7 of the 20 hospitals reported prices for all CV tests and procedures. Centers that did not post prices for some tests or procedures are named in the report’s Figure 1 and Figure 2.
The number of insurance plans listed for each test or procedure ranged from 1 to 432 in the analysis. Median prices ranged from $204 to $2,588 for an echocardiogram, $463 to $3,230 for a stress test, $2,821 to $9,382 for right heart catheterization, $2,868 to $9,203 for a coronary angiogram, $657 to $25,521 for a PCI, and $506 to $20,002 for pacemaker implantation, the report states.
A similar pattern was seen for self-pay cash prices.
Within-hospital variation also ranged broadly. For example, the widest IQR ranges were $3,143-$12,926 for a right heart catheterization, $4,011-$14,486 for a coronary angiogram, $11,325-$23,392 for a PCI, and $8,474-$22,694 for pacemaker implantation.
The report cites a number of limitations to the analysis, among those, the need to rely on the hospitals themselves for data quality and accuracy.
‘More needed besides transparency’
“As a means to better understand health care costs, many opined that full price transparency would leverage market dynamics and result in lower costs,” observed Clyde W. Yancy, MD, MSc, professor of medicine and chief of cardiology at Northwestern Medicine, Chicago. The findings “by an expert group of outcomes scientists make clear that more is needed besides price transparency to lower cost,” he said in an interview.
That said, he added, “there are sufficient variations and allowances made for data collection that it is preferable to hold the current findings circumspect at best. Importantly, the voice of the hospitals does not appear.”
Although “price variation among the top 20 hospitals is substantial,” he observed, “without a better assessment of root cause, actual charge capture, prevailing market dynamics – especially nursing and ancillary staff costs – and the general influence of inflation, it is too difficult to emerge with a precise interpretation.”
Across the 20 hospitals, “there are likely to be 20 different business models,” he added, with negotiated prices reflecting “at least regional, if not institutional, variations.”
“These are complex issues. The several-fold price differences in standard procedures are a concern and an area worth further study with the intention of lowering health care costs,” Dr. Yancy said. “But clearly our next efforts should not address lowering prices per se but understanding how prices are set [and] the connection with reimbursement and actual payments.”
Dr. Wadhera discloses receiving personal fees from Abbott and CVS Health unrelated to the current study; disclosures for the other authors are in the report. Dr. Yancy is deputy editor of JAMA Cardiology.
A version of this article first appeared on Medscape.com.
Wide variation in the cost of common cardiovascular (CV) tests and procedures, from stress tests to coronary interventions, was revealed in a cross-sectional analysis based on publicly available data from 20 top-ranked hospitals in the United States.
The analysis also suggested a low level of compliance with the 2021 Hospital Price Transparency Final Rule among the 20 centers.
“The variation we found in payer-negotiated prices for identical cardiovascular tests and procedures was stunning,” Rishi K. Wadhera, MD, MPP, MPhil, Beth Israel Deaconess Medical Center, Boston, told this news organization.
“For example, there was a 10-fold difference in the median price of an echocardiogram, and these differences were even larger for common procedures” such as percutaneous coronary intervention (PCI) and pacemaker implantation, he said. “It’s hard to argue that this variation reflects quality of care, given that we looked at a top group of highly ranked hospitals.”
“Even more striking was how the price of a cardiovascular test within the very same hospital could differ across commercial insurance companies,” he said. “For example, the price of a stress test varied 5-fold in one hospital, and in another hospital, more than 4-fold for a coronary angiogram.”
Dr. Wadhera is senior author on the study published online as a research letter in JAMA Internal Medicine, with lead author Andrew S. Oseran, MD, MBA, also from Beth Israel Deaconess Medical Center.
Difficulties with data, interpretation
The researchers looked at payer and self-pay cash prices for noninvasive and invasive CV tests and procedures at the U.S. News & World Report 2021 top 20–ranked U.S. hospitals, based in part on Current Procedural Terminology codes.
Price differences among the hospitals were derived from median negotiated prices for each test and procedure at the centers across all payers. The interquartile ratio (IQR) of prices for each test or procedure across payers was used to evaluate within-hospital price variation.
“Only 80% of the hospitals reported prices for some cardiovascular tests and procedures,” Dr. Wadhera said. “For the most part, even among the hospitals that did report this information, it was extremely challenging to navigate and interpret the data provided.”
Further, the team found that only 7 of the 20 hospitals reported prices for all CV tests and procedures. Centers that did not post prices for some tests or procedures are named in the report’s Figure 1 and Figure 2.
The number of insurance plans listed for each test or procedure ranged from 1 to 432 in the analysis. Median prices ranged from $204 to $2,588 for an echocardiogram, $463 to $3,230 for a stress test, $2,821 to $9,382 for right heart catheterization, $2,868 to $9,203 for a coronary angiogram, $657 to $25,521 for a PCI, and $506 to $20,002 for pacemaker implantation, the report states.
A similar pattern was seen for self-pay cash prices.
Within-hospital variation also ranged broadly. For example, the widest IQR ranges were $3,143-$12,926 for a right heart catheterization, $4,011-$14,486 for a coronary angiogram, $11,325-$23,392 for a PCI, and $8,474-$22,694 for pacemaker implantation.
The report cites a number of limitations to the analysis, among those, the need to rely on the hospitals themselves for data quality and accuracy.
‘More needed besides transparency’
“As a means to better understand health care costs, many opined that full price transparency would leverage market dynamics and result in lower costs,” observed Clyde W. Yancy, MD, MSc, professor of medicine and chief of cardiology at Northwestern Medicine, Chicago. The findings “by an expert group of outcomes scientists make clear that more is needed besides price transparency to lower cost,” he said in an interview.
That said, he added, “there are sufficient variations and allowances made for data collection that it is preferable to hold the current findings circumspect at best. Importantly, the voice of the hospitals does not appear.”
Although “price variation among the top 20 hospitals is substantial,” he observed, “without a better assessment of root cause, actual charge capture, prevailing market dynamics – especially nursing and ancillary staff costs – and the general influence of inflation, it is too difficult to emerge with a precise interpretation.”
Across the 20 hospitals, “there are likely to be 20 different business models,” he added, with negotiated prices reflecting “at least regional, if not institutional, variations.”
“These are complex issues. The several-fold price differences in standard procedures are a concern and an area worth further study with the intention of lowering health care costs,” Dr. Yancy said. “But clearly our next efforts should not address lowering prices per se but understanding how prices are set [and] the connection with reimbursement and actual payments.”
Dr. Wadhera discloses receiving personal fees from Abbott and CVS Health unrelated to the current study; disclosures for the other authors are in the report. Dr. Yancy is deputy editor of JAMA Cardiology.
A version of this article first appeared on Medscape.com.
Wide variation in the cost of common cardiovascular (CV) tests and procedures, from stress tests to coronary interventions, was revealed in a cross-sectional analysis based on publicly available data from 20 top-ranked hospitals in the United States.
The analysis also suggested a low level of compliance with the 2021 Hospital Price Transparency Final Rule among the 20 centers.
“The variation we found in payer-negotiated prices for identical cardiovascular tests and procedures was stunning,” Rishi K. Wadhera, MD, MPP, MPhil, Beth Israel Deaconess Medical Center, Boston, told this news organization.
“For example, there was a 10-fold difference in the median price of an echocardiogram, and these differences were even larger for common procedures” such as percutaneous coronary intervention (PCI) and pacemaker implantation, he said. “It’s hard to argue that this variation reflects quality of care, given that we looked at a top group of highly ranked hospitals.”
“Even more striking was how the price of a cardiovascular test within the very same hospital could differ across commercial insurance companies,” he said. “For example, the price of a stress test varied 5-fold in one hospital, and in another hospital, more than 4-fold for a coronary angiogram.”
Dr. Wadhera is senior author on the study published online as a research letter in JAMA Internal Medicine, with lead author Andrew S. Oseran, MD, MBA, also from Beth Israel Deaconess Medical Center.
Difficulties with data, interpretation
The researchers looked at payer and self-pay cash prices for noninvasive and invasive CV tests and procedures at the U.S. News & World Report 2021 top 20–ranked U.S. hospitals, based in part on Current Procedural Terminology codes.
Price differences among the hospitals were derived from median negotiated prices for each test and procedure at the centers across all payers. The interquartile ratio (IQR) of prices for each test or procedure across payers was used to evaluate within-hospital price variation.
“Only 80% of the hospitals reported prices for some cardiovascular tests and procedures,” Dr. Wadhera said. “For the most part, even among the hospitals that did report this information, it was extremely challenging to navigate and interpret the data provided.”
Further, the team found that only 7 of the 20 hospitals reported prices for all CV tests and procedures. Centers that did not post prices for some tests or procedures are named in the report’s Figure 1 and Figure 2.
The number of insurance plans listed for each test or procedure ranged from 1 to 432 in the analysis. Median prices ranged from $204 to $2,588 for an echocardiogram, $463 to $3,230 for a stress test, $2,821 to $9,382 for right heart catheterization, $2,868 to $9,203 for a coronary angiogram, $657 to $25,521 for a PCI, and $506 to $20,002 for pacemaker implantation, the report states.
A similar pattern was seen for self-pay cash prices.
Within-hospital variation also ranged broadly. For example, the widest IQR ranges were $3,143-$12,926 for a right heart catheterization, $4,011-$14,486 for a coronary angiogram, $11,325-$23,392 for a PCI, and $8,474-$22,694 for pacemaker implantation.
The report cites a number of limitations to the analysis, among those, the need to rely on the hospitals themselves for data quality and accuracy.
‘More needed besides transparency’
“As a means to better understand health care costs, many opined that full price transparency would leverage market dynamics and result in lower costs,” observed Clyde W. Yancy, MD, MSc, professor of medicine and chief of cardiology at Northwestern Medicine, Chicago. The findings “by an expert group of outcomes scientists make clear that more is needed besides price transparency to lower cost,” he said in an interview.
That said, he added, “there are sufficient variations and allowances made for data collection that it is preferable to hold the current findings circumspect at best. Importantly, the voice of the hospitals does not appear.”
Although “price variation among the top 20 hospitals is substantial,” he observed, “without a better assessment of root cause, actual charge capture, prevailing market dynamics – especially nursing and ancillary staff costs – and the general influence of inflation, it is too difficult to emerge with a precise interpretation.”
Across the 20 hospitals, “there are likely to be 20 different business models,” he added, with negotiated prices reflecting “at least regional, if not institutional, variations.”
“These are complex issues. The several-fold price differences in standard procedures are a concern and an area worth further study with the intention of lowering health care costs,” Dr. Yancy said. “But clearly our next efforts should not address lowering prices per se but understanding how prices are set [and] the connection with reimbursement and actual payments.”
Dr. Wadhera discloses receiving personal fees from Abbott and CVS Health unrelated to the current study; disclosures for the other authors are in the report. Dr. Yancy is deputy editor of JAMA Cardiology.
A version of this article first appeared on Medscape.com.
Interventional imagers take on central role and more radiation
Interventional echocardiographers have become an increasingly critical part of the structural heart team but may be paying the price in terms of radiation exposure, a new study suggests.
Results showed that interventional echocardiographers receive threefold higher head-level radiation doses than interventional cardiologists during left atrial appendage occlusion (LAAO) closures and 11-fold higher doses during mitral valve transcatheter edge-to-edge repair (TEER).
“Over the last 5-10 years there’s been exponential growth in these two procedures, TEER and LAAO, and while that’s been very exciting, I think there hasn’t been as much research into how to protect these individuals,” lead author David A. McNamara, MD, MPH, Spectrum Health, Grand Rapids, Mich., told this news organization.
The study was published in JAMA Network Open.
Previous studies have focused largely on radiation exposure and mitigation efforts during coronary interventions, but the room set-up for LAAO and TEER and shielding techniques to mitigate radiation exposure are vastly different, he noted.
A 2017 study reported that radiation exposure was significantly higher for imaging specialists than structural heart specialists and varied by procedure type.
For the current study, Dr. McNamara, an echocardiographer by training, and colleagues collected data from 30 consecutive LAAO and 30 consecutive TEER procedures performed at their institution between July 2016 and January 2018.
Interventional imagers, interventional cardiologists, and sonographers all wore a lead skirt, apron, and thyroid collar, as well as a dosimeter to collect radiation data.
Interventional cardiologists stood immediately adjacent to the procedure table and used a ceiling-mounted, upper-body lead shield and a lower-body shield extending from the table to the floor. The echocardiographer stood at the patient’s head and used a mobile accessory shield raised to a height that allowed the imager to extend their arms over the shield to manipulate a transesophageal echocardiogram probe throughout the case.
The median fluoroscopy time was 9.2 minutes for LAAO and 20.9 minutes for TEER. The median air kerma was 164 mGy and 109 mGy, respectively.
Interventional echocardiographers received a median per case radiation dose of 10.6 µSv, compared with 2.1 µSv for interventional cardiologists. The result was similar for TEER (10.5 vs. 0.9 µSv) and LAAO (10.6 vs. 3.5 µSv; P < .001 for all).
The odds of interventional echocardiographers having a radiation dose greater than 20 µSV were 7.5 times greater than for interventional cardiologists (P < .001).
“It’s not the direction of the association, but really the magnitude is what surprised us,” observed Dr. McNamara.
The team was pleasantly surprised, he said, that sonographers, a “vastly understudied group,” received significantly lower median radiation doses than interventional imagers during LAAO (0.2 µSV) and TEER procedures (0.0 µSv; P < .001 for both).
The average distances from the radiation source were 26 cm (10.2 inches) for the echocardiographer, 36 cm (14.2 inches) for the interventional cardiologist, and 250 cm (8.2 feet) for the sonographer.
“These folks [sonographers] were much further away than both the physicians performing these cases, and that is what we hypothesize drove their very low rates, but that should also help inform our mitigation techniques for physicians and for all other cath lab members in the room,” Dr. McNamara said.
He noted that Spectrum Health has been at the forefront in terms of research into radiation exposure and mitigation, has good institutional radiation safety education, and used dose-lowering fluoroscopy systems (AlluraClarity, Philips) with real-time image noise reduction technology and a frame rate of 15 frames per second for the study. “So we’re hopeful that this actually represents a somewhat best-case scenario for what is being done at multiple institutions throughout the nation.”
Nevertheless, there is a huge amount of variability in radiation exposure, Dr. McNamara observed. “First and foremost, we really just have to identify our problem and highlight that this is something that needs some advocacy from our [professional] groups.”
Sunil Rao, MD, the newly minted president of the Society of Cardiovascular Angiography and Interventions (SCAI), said, “This is a really important study, because it expands the potential occupational hazards outside of what we traditionally think of as the team that does interventional procedures ... we have to recognize that the procedures we’re doing in the cath lab have changed.”
“Showing that our colleagues are getting 3-10 times radiation exposure is a really important piece of information to have out there. I think it’s really sort of a call to action,” Dr. Rao, professor of medicine at Duke University, Durham, N.C., told this news organization.
Nevertheless, he observed that practices have shifted somewhat since the study and that interventional cardiologists working with imaging physicians are more cognizant of radiation exposure issues.
“When I talk with our folks here that are doing structural heart procedures, they’re making sure that they’re not stepping on the fluoro pedal while the echocardiographer is manipulating the TE probe,” Dr. Rao said. “The echocardiographer is oftentimes using a much bigger shield than what was described in the study, and remember there’s an exponential decrease in the radiation exposure by distance, so they’re stepping back during the fluoroscopy time.”
Although the volume of TEER and LAAO procedures, as well as tricuspid interventions, will continue to climb, Dr. Rao said he expects radiation exposure to the imaging cardiologist will fall thanks to greater use of newer-generation imaging systems with dose-reduction features and better shielding strategies.
He noted that several of SCAI’s “best practices” documents call attention to radiation safety and that SCAI is creating a pathway where imaging cardiologists can become fellows of the society, which was traditionally reserved for interventionalists.
Still, imaging and cardiovascular societies have yet to endorse standardized safety procedures for interventional imagers, nor is information routinely collected on radiation exposure in national registries.
“We just don’t have the budgets or the interest nationally to do that kind of thing, so it has to be done locally,” Dr. Rao said. “And the person who I think is responsible for that is really the cath lab director and the cath lab nurse manager, who really should work hand-in-glove to make sure that radiation safety is at the top of the priority list.”
The study was funded by the Frederik Meijer Heart & Vascular Institute, Spectrum Health, and by Corindus. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, approval of the manuscript; and the decision to submit the manuscript for publication. Senior author Ryan Madder, MD, reports receiving research support, speaker honoraria, and grants, and serving on the advisory board of Corindus. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
Interventional echocardiographers have become an increasingly critical part of the structural heart team but may be paying the price in terms of radiation exposure, a new study suggests.
Results showed that interventional echocardiographers receive threefold higher head-level radiation doses than interventional cardiologists during left atrial appendage occlusion (LAAO) closures and 11-fold higher doses during mitral valve transcatheter edge-to-edge repair (TEER).
“Over the last 5-10 years there’s been exponential growth in these two procedures, TEER and LAAO, and while that’s been very exciting, I think there hasn’t been as much research into how to protect these individuals,” lead author David A. McNamara, MD, MPH, Spectrum Health, Grand Rapids, Mich., told this news organization.
The study was published in JAMA Network Open.
Previous studies have focused largely on radiation exposure and mitigation efforts during coronary interventions, but the room set-up for LAAO and TEER and shielding techniques to mitigate radiation exposure are vastly different, he noted.
A 2017 study reported that radiation exposure was significantly higher for imaging specialists than structural heart specialists and varied by procedure type.
For the current study, Dr. McNamara, an echocardiographer by training, and colleagues collected data from 30 consecutive LAAO and 30 consecutive TEER procedures performed at their institution between July 2016 and January 2018.
Interventional imagers, interventional cardiologists, and sonographers all wore a lead skirt, apron, and thyroid collar, as well as a dosimeter to collect radiation data.
Interventional cardiologists stood immediately adjacent to the procedure table and used a ceiling-mounted, upper-body lead shield and a lower-body shield extending from the table to the floor. The echocardiographer stood at the patient’s head and used a mobile accessory shield raised to a height that allowed the imager to extend their arms over the shield to manipulate a transesophageal echocardiogram probe throughout the case.
The median fluoroscopy time was 9.2 minutes for LAAO and 20.9 minutes for TEER. The median air kerma was 164 mGy and 109 mGy, respectively.
Interventional echocardiographers received a median per case radiation dose of 10.6 µSv, compared with 2.1 µSv for interventional cardiologists. The result was similar for TEER (10.5 vs. 0.9 µSv) and LAAO (10.6 vs. 3.5 µSv; P < .001 for all).
The odds of interventional echocardiographers having a radiation dose greater than 20 µSV were 7.5 times greater than for interventional cardiologists (P < .001).
“It’s not the direction of the association, but really the magnitude is what surprised us,” observed Dr. McNamara.
The team was pleasantly surprised, he said, that sonographers, a “vastly understudied group,” received significantly lower median radiation doses than interventional imagers during LAAO (0.2 µSV) and TEER procedures (0.0 µSv; P < .001 for both).
The average distances from the radiation source were 26 cm (10.2 inches) for the echocardiographer, 36 cm (14.2 inches) for the interventional cardiologist, and 250 cm (8.2 feet) for the sonographer.
“These folks [sonographers] were much further away than both the physicians performing these cases, and that is what we hypothesize drove their very low rates, but that should also help inform our mitigation techniques for physicians and for all other cath lab members in the room,” Dr. McNamara said.
He noted that Spectrum Health has been at the forefront in terms of research into radiation exposure and mitigation, has good institutional radiation safety education, and used dose-lowering fluoroscopy systems (AlluraClarity, Philips) with real-time image noise reduction technology and a frame rate of 15 frames per second for the study. “So we’re hopeful that this actually represents a somewhat best-case scenario for what is being done at multiple institutions throughout the nation.”
Nevertheless, there is a huge amount of variability in radiation exposure, Dr. McNamara observed. “First and foremost, we really just have to identify our problem and highlight that this is something that needs some advocacy from our [professional] groups.”
Sunil Rao, MD, the newly minted president of the Society of Cardiovascular Angiography and Interventions (SCAI), said, “This is a really important study, because it expands the potential occupational hazards outside of what we traditionally think of as the team that does interventional procedures ... we have to recognize that the procedures we’re doing in the cath lab have changed.”
“Showing that our colleagues are getting 3-10 times radiation exposure is a really important piece of information to have out there. I think it’s really sort of a call to action,” Dr. Rao, professor of medicine at Duke University, Durham, N.C., told this news organization.
Nevertheless, he observed that practices have shifted somewhat since the study and that interventional cardiologists working with imaging physicians are more cognizant of radiation exposure issues.
“When I talk with our folks here that are doing structural heart procedures, they’re making sure that they’re not stepping on the fluoro pedal while the echocardiographer is manipulating the TE probe,” Dr. Rao said. “The echocardiographer is oftentimes using a much bigger shield than what was described in the study, and remember there’s an exponential decrease in the radiation exposure by distance, so they’re stepping back during the fluoroscopy time.”
Although the volume of TEER and LAAO procedures, as well as tricuspid interventions, will continue to climb, Dr. Rao said he expects radiation exposure to the imaging cardiologist will fall thanks to greater use of newer-generation imaging systems with dose-reduction features and better shielding strategies.
He noted that several of SCAI’s “best practices” documents call attention to radiation safety and that SCAI is creating a pathway where imaging cardiologists can become fellows of the society, which was traditionally reserved for interventionalists.
Still, imaging and cardiovascular societies have yet to endorse standardized safety procedures for interventional imagers, nor is information routinely collected on radiation exposure in national registries.
“We just don’t have the budgets or the interest nationally to do that kind of thing, so it has to be done locally,” Dr. Rao said. “And the person who I think is responsible for that is really the cath lab director and the cath lab nurse manager, who really should work hand-in-glove to make sure that radiation safety is at the top of the priority list.”
The study was funded by the Frederik Meijer Heart & Vascular Institute, Spectrum Health, and by Corindus. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, approval of the manuscript; and the decision to submit the manuscript for publication. Senior author Ryan Madder, MD, reports receiving research support, speaker honoraria, and grants, and serving on the advisory board of Corindus. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
Interventional echocardiographers have become an increasingly critical part of the structural heart team but may be paying the price in terms of radiation exposure, a new study suggests.
Results showed that interventional echocardiographers receive threefold higher head-level radiation doses than interventional cardiologists during left atrial appendage occlusion (LAAO) closures and 11-fold higher doses during mitral valve transcatheter edge-to-edge repair (TEER).
“Over the last 5-10 years there’s been exponential growth in these two procedures, TEER and LAAO, and while that’s been very exciting, I think there hasn’t been as much research into how to protect these individuals,” lead author David A. McNamara, MD, MPH, Spectrum Health, Grand Rapids, Mich., told this news organization.
The study was published in JAMA Network Open.
Previous studies have focused largely on radiation exposure and mitigation efforts during coronary interventions, but the room set-up for LAAO and TEER and shielding techniques to mitigate radiation exposure are vastly different, he noted.
A 2017 study reported that radiation exposure was significantly higher for imaging specialists than structural heart specialists and varied by procedure type.
For the current study, Dr. McNamara, an echocardiographer by training, and colleagues collected data from 30 consecutive LAAO and 30 consecutive TEER procedures performed at their institution between July 2016 and January 2018.
Interventional imagers, interventional cardiologists, and sonographers all wore a lead skirt, apron, and thyroid collar, as well as a dosimeter to collect radiation data.
Interventional cardiologists stood immediately adjacent to the procedure table and used a ceiling-mounted, upper-body lead shield and a lower-body shield extending from the table to the floor. The echocardiographer stood at the patient’s head and used a mobile accessory shield raised to a height that allowed the imager to extend their arms over the shield to manipulate a transesophageal echocardiogram probe throughout the case.
The median fluoroscopy time was 9.2 minutes for LAAO and 20.9 minutes for TEER. The median air kerma was 164 mGy and 109 mGy, respectively.
Interventional echocardiographers received a median per case radiation dose of 10.6 µSv, compared with 2.1 µSv for interventional cardiologists. The result was similar for TEER (10.5 vs. 0.9 µSv) and LAAO (10.6 vs. 3.5 µSv; P < .001 for all).
The odds of interventional echocardiographers having a radiation dose greater than 20 µSV were 7.5 times greater than for interventional cardiologists (P < .001).
“It’s not the direction of the association, but really the magnitude is what surprised us,” observed Dr. McNamara.
The team was pleasantly surprised, he said, that sonographers, a “vastly understudied group,” received significantly lower median radiation doses than interventional imagers during LAAO (0.2 µSV) and TEER procedures (0.0 µSv; P < .001 for both).
The average distances from the radiation source were 26 cm (10.2 inches) for the echocardiographer, 36 cm (14.2 inches) for the interventional cardiologist, and 250 cm (8.2 feet) for the sonographer.
“These folks [sonographers] were much further away than both the physicians performing these cases, and that is what we hypothesize drove their very low rates, but that should also help inform our mitigation techniques for physicians and for all other cath lab members in the room,” Dr. McNamara said.
He noted that Spectrum Health has been at the forefront in terms of research into radiation exposure and mitigation, has good institutional radiation safety education, and used dose-lowering fluoroscopy systems (AlluraClarity, Philips) with real-time image noise reduction technology and a frame rate of 15 frames per second for the study. “So we’re hopeful that this actually represents a somewhat best-case scenario for what is being done at multiple institutions throughout the nation.”
Nevertheless, there is a huge amount of variability in radiation exposure, Dr. McNamara observed. “First and foremost, we really just have to identify our problem and highlight that this is something that needs some advocacy from our [professional] groups.”
Sunil Rao, MD, the newly minted president of the Society of Cardiovascular Angiography and Interventions (SCAI), said, “This is a really important study, because it expands the potential occupational hazards outside of what we traditionally think of as the team that does interventional procedures ... we have to recognize that the procedures we’re doing in the cath lab have changed.”
“Showing that our colleagues are getting 3-10 times radiation exposure is a really important piece of information to have out there. I think it’s really sort of a call to action,” Dr. Rao, professor of medicine at Duke University, Durham, N.C., told this news organization.
Nevertheless, he observed that practices have shifted somewhat since the study and that interventional cardiologists working with imaging physicians are more cognizant of radiation exposure issues.
“When I talk with our folks here that are doing structural heart procedures, they’re making sure that they’re not stepping on the fluoro pedal while the echocardiographer is manipulating the TE probe,” Dr. Rao said. “The echocardiographer is oftentimes using a much bigger shield than what was described in the study, and remember there’s an exponential decrease in the radiation exposure by distance, so they’re stepping back during the fluoroscopy time.”
Although the volume of TEER and LAAO procedures, as well as tricuspid interventions, will continue to climb, Dr. Rao said he expects radiation exposure to the imaging cardiologist will fall thanks to greater use of newer-generation imaging systems with dose-reduction features and better shielding strategies.
He noted that several of SCAI’s “best practices” documents call attention to radiation safety and that SCAI is creating a pathway where imaging cardiologists can become fellows of the society, which was traditionally reserved for interventionalists.
Still, imaging and cardiovascular societies have yet to endorse standardized safety procedures for interventional imagers, nor is information routinely collected on radiation exposure in national registries.
“We just don’t have the budgets or the interest nationally to do that kind of thing, so it has to be done locally,” Dr. Rao said. “And the person who I think is responsible for that is really the cath lab director and the cath lab nurse manager, who really should work hand-in-glove to make sure that radiation safety is at the top of the priority list.”
The study was funded by the Frederik Meijer Heart & Vascular Institute, Spectrum Health, and by Corindus. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, approval of the manuscript; and the decision to submit the manuscript for publication. Senior author Ryan Madder, MD, reports receiving research support, speaker honoraria, and grants, and serving on the advisory board of Corindus. No other disclosures were reported.
A version of this article first appeared on Medscape.com.
Access to certified stroke centers divided by race, income
Hospitals in low-income and rural areas of the United States are much less likely to adopt stroke certification than hospitals in high-income and urban communities, a new study shows.
Further, other results showed that, after adjustment for population and hospital size, access to stroke-certified hospitals is significantly lower in Black, racially segregated communities.
The study was published online in JAMA Neurology.
Noting that stroke-certified hospitals provide higher-quality stroke care, the authors, led by Yu-Chu Shen, PhD, Naval Postgraduate School, Monterey, Calif., conclude that: “Our findings suggest that structural inequities in stroke care may be an important consideration in eliminating stroke disparities for vulnerable populations.”
In an audio interview on the JAMA Neurology website, senior author Renee Y. Hsia, MD, University of California, San Francisco, said: “Our findings show there are clear disparities in which communities are getting access to stroke certified hospitals.”
She called for more help for hospitals in underserved areas to obtain stroke certification.
Dr. Hsia explained that hospitals can seek certification at their own expense and that although stroke care is expensive, it is also lucrative in terms of reimbursement. So it tends to be the private for-profit hospitals that seek these certifications. “If you are a county hospital on a really tight budget, you’re not going to have the extra cash on hand to be applying for stroke certification,” she commented.
This can result in an increase in hospitals with stroke certification – but not in the areas that need it the most.
Dr. Hsia points out that this has happened in cardiac care. One study showed a 44% increase in hospitals providing percutaneous coronary intervention over a 10-year period, but the percentage of the population that had better access increased by less than 1%.
“In general, in the United States we have a mentality that ‘more is better,’ and because there is no government regulation in health care, any time a hospital applies for these specialized services we just generally think that’s a good thing. But this might not always be the case,” Dr. Hsia noted. “We have a very market-based approach, and this doesn’t lead to equity. It leads to profit maximization, and that is not synonymous with what’s good for patients or populations.”
She suggested that in future the process of certification should include some consideration of how it will affect population-based equity.
“Rather than rubber stamping an application just because hospitals have certain resources, we need to ask what the benefit is of providing this service,” Dr. Hsia said. “Does this community really need it? If not, maybe we should invest these resources into helping a hospital in a community that needs it more.”
Dr. Hsia explained that she and her colleagues conducted their study to investigate whether there were structural issues that might be contributing to disparities in stroke care.
“We like to think emergency stroke care is equitable. Anyone can call 911 or go the emergency room. But, actually, there is a big disparity on who receives what type of care,” she said. “We know Black patients are less likely to receive thrombolytics and mechanical thrombectomy compared to White patents. And wealthy patients are more likely to receive thrombectomy compared to patients from the poorest zip codes.”
She said there is a tendency to think this is a result of some sort of bias on the part of health care professionals. “We wanted to look deep down in the system and whether the built environment of health care supply and geographic distribution of services contributed to access and treatment inequities.”
The study combined a dataset of hospital stroke certification from all general acute nonfederal hospitals in the continental United States from January 2009 to December 2019. National, hospital, and census data were used to identify historically underserved communities by racial and ethnic composition, income distribution, and rurality.
A total of 4,984 hospitals were assessed. Results showed that over the 11-year study period, the number of hospitals with stroke certification grew from 961 (19%) to 1,763 (36%).
Without controlling for population and hospital size, hospitals in predominantly Black, racially segregated areas were 1.67-fold more likely to adopt stroke care of any level than those in predominantly non-Black, racially segregated areas (hazard ratio, 1.67; 95% confidence interval, 1.41-1.97).
However, after adjustment for population and hospital size, the likelihood of adopting stroke care among hospitals serving Black, racially segregated communities was significantly lower than among those serving non-Black, racially segregated communities (HR, 0.74; 95% CI, 0.62-0.89).
“In other words, on a per-capita basis, a hospital serving a predominantly Black, racially segregated community was 26% less likely to adopt stroke certification of any level than a hospital in a predominantly non-Black, racially segregated community,” the authors state.
In terms of socioeconomic factors, hospitals serving low-income, economically integrated (HR, 0.23) and low-income, economically segregated (HR, 0.29) areas were far less likely to adopt any level of stroke care certification than hospitals serving high-income areas, regardless of income segregation.
Rural hospitals were also much less likely to adopt any level of stroke care than urban hospitals (HR, 0.10).
“Our results suggest that it might be necessary to incentivize hospitals operating in underserved communities to seek stroke certification or to entice hospitals with higher propensity to adopt stroke care to operate in such communities so access at the per-patient level becomes more equitable,” the authors say.
This project was supported by the Pilot Project Award from the National Bureau of Economic Research Center for Aging and Health Research, funded by the National Institute on Aging and by the National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Shen and Dr. Hsia have received grants from the National Institute of Aging and the National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
Hospitals in low-income and rural areas of the United States are much less likely to adopt stroke certification than hospitals in high-income and urban communities, a new study shows.
Further, other results showed that, after adjustment for population and hospital size, access to stroke-certified hospitals is significantly lower in Black, racially segregated communities.
The study was published online in JAMA Neurology.
Noting that stroke-certified hospitals provide higher-quality stroke care, the authors, led by Yu-Chu Shen, PhD, Naval Postgraduate School, Monterey, Calif., conclude that: “Our findings suggest that structural inequities in stroke care may be an important consideration in eliminating stroke disparities for vulnerable populations.”
In an audio interview on the JAMA Neurology website, senior author Renee Y. Hsia, MD, University of California, San Francisco, said: “Our findings show there are clear disparities in which communities are getting access to stroke certified hospitals.”
She called for more help for hospitals in underserved areas to obtain stroke certification.
Dr. Hsia explained that hospitals can seek certification at their own expense and that although stroke care is expensive, it is also lucrative in terms of reimbursement. So it tends to be the private for-profit hospitals that seek these certifications. “If you are a county hospital on a really tight budget, you’re not going to have the extra cash on hand to be applying for stroke certification,” she commented.
This can result in an increase in hospitals with stroke certification – but not in the areas that need it the most.
Dr. Hsia points out that this has happened in cardiac care. One study showed a 44% increase in hospitals providing percutaneous coronary intervention over a 10-year period, but the percentage of the population that had better access increased by less than 1%.
“In general, in the United States we have a mentality that ‘more is better,’ and because there is no government regulation in health care, any time a hospital applies for these specialized services we just generally think that’s a good thing. But this might not always be the case,” Dr. Hsia noted. “We have a very market-based approach, and this doesn’t lead to equity. It leads to profit maximization, and that is not synonymous with what’s good for patients or populations.”
She suggested that in future the process of certification should include some consideration of how it will affect population-based equity.
“Rather than rubber stamping an application just because hospitals have certain resources, we need to ask what the benefit is of providing this service,” Dr. Hsia said. “Does this community really need it? If not, maybe we should invest these resources into helping a hospital in a community that needs it more.”
Dr. Hsia explained that she and her colleagues conducted their study to investigate whether there were structural issues that might be contributing to disparities in stroke care.
“We like to think emergency stroke care is equitable. Anyone can call 911 or go the emergency room. But, actually, there is a big disparity on who receives what type of care,” she said. “We know Black patients are less likely to receive thrombolytics and mechanical thrombectomy compared to White patents. And wealthy patients are more likely to receive thrombectomy compared to patients from the poorest zip codes.”
She said there is a tendency to think this is a result of some sort of bias on the part of health care professionals. “We wanted to look deep down in the system and whether the built environment of health care supply and geographic distribution of services contributed to access and treatment inequities.”
The study combined a dataset of hospital stroke certification from all general acute nonfederal hospitals in the continental United States from January 2009 to December 2019. National, hospital, and census data were used to identify historically underserved communities by racial and ethnic composition, income distribution, and rurality.
A total of 4,984 hospitals were assessed. Results showed that over the 11-year study period, the number of hospitals with stroke certification grew from 961 (19%) to 1,763 (36%).
Without controlling for population and hospital size, hospitals in predominantly Black, racially segregated areas were 1.67-fold more likely to adopt stroke care of any level than those in predominantly non-Black, racially segregated areas (hazard ratio, 1.67; 95% confidence interval, 1.41-1.97).
However, after adjustment for population and hospital size, the likelihood of adopting stroke care among hospitals serving Black, racially segregated communities was significantly lower than among those serving non-Black, racially segregated communities (HR, 0.74; 95% CI, 0.62-0.89).
“In other words, on a per-capita basis, a hospital serving a predominantly Black, racially segregated community was 26% less likely to adopt stroke certification of any level than a hospital in a predominantly non-Black, racially segregated community,” the authors state.
In terms of socioeconomic factors, hospitals serving low-income, economically integrated (HR, 0.23) and low-income, economically segregated (HR, 0.29) areas were far less likely to adopt any level of stroke care certification than hospitals serving high-income areas, regardless of income segregation.
Rural hospitals were also much less likely to adopt any level of stroke care than urban hospitals (HR, 0.10).
“Our results suggest that it might be necessary to incentivize hospitals operating in underserved communities to seek stroke certification or to entice hospitals with higher propensity to adopt stroke care to operate in such communities so access at the per-patient level becomes more equitable,” the authors say.
This project was supported by the Pilot Project Award from the National Bureau of Economic Research Center for Aging and Health Research, funded by the National Institute on Aging and by the National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Shen and Dr. Hsia have received grants from the National Institute of Aging and the National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
Hospitals in low-income and rural areas of the United States are much less likely to adopt stroke certification than hospitals in high-income and urban communities, a new study shows.
Further, other results showed that, after adjustment for population and hospital size, access to stroke-certified hospitals is significantly lower in Black, racially segregated communities.
The study was published online in JAMA Neurology.
Noting that stroke-certified hospitals provide higher-quality stroke care, the authors, led by Yu-Chu Shen, PhD, Naval Postgraduate School, Monterey, Calif., conclude that: “Our findings suggest that structural inequities in stroke care may be an important consideration in eliminating stroke disparities for vulnerable populations.”
In an audio interview on the JAMA Neurology website, senior author Renee Y. Hsia, MD, University of California, San Francisco, said: “Our findings show there are clear disparities in which communities are getting access to stroke certified hospitals.”
She called for more help for hospitals in underserved areas to obtain stroke certification.
Dr. Hsia explained that hospitals can seek certification at their own expense and that although stroke care is expensive, it is also lucrative in terms of reimbursement. So it tends to be the private for-profit hospitals that seek these certifications. “If you are a county hospital on a really tight budget, you’re not going to have the extra cash on hand to be applying for stroke certification,” she commented.
This can result in an increase in hospitals with stroke certification – but not in the areas that need it the most.
Dr. Hsia points out that this has happened in cardiac care. One study showed a 44% increase in hospitals providing percutaneous coronary intervention over a 10-year period, but the percentage of the population that had better access increased by less than 1%.
“In general, in the United States we have a mentality that ‘more is better,’ and because there is no government regulation in health care, any time a hospital applies for these specialized services we just generally think that’s a good thing. But this might not always be the case,” Dr. Hsia noted. “We have a very market-based approach, and this doesn’t lead to equity. It leads to profit maximization, and that is not synonymous with what’s good for patients or populations.”
She suggested that in future the process of certification should include some consideration of how it will affect population-based equity.
“Rather than rubber stamping an application just because hospitals have certain resources, we need to ask what the benefit is of providing this service,” Dr. Hsia said. “Does this community really need it? If not, maybe we should invest these resources into helping a hospital in a community that needs it more.”
Dr. Hsia explained that she and her colleagues conducted their study to investigate whether there were structural issues that might be contributing to disparities in stroke care.
“We like to think emergency stroke care is equitable. Anyone can call 911 or go the emergency room. But, actually, there is a big disparity on who receives what type of care,” she said. “We know Black patients are less likely to receive thrombolytics and mechanical thrombectomy compared to White patents. And wealthy patients are more likely to receive thrombectomy compared to patients from the poorest zip codes.”
She said there is a tendency to think this is a result of some sort of bias on the part of health care professionals. “We wanted to look deep down in the system and whether the built environment of health care supply and geographic distribution of services contributed to access and treatment inequities.”
The study combined a dataset of hospital stroke certification from all general acute nonfederal hospitals in the continental United States from January 2009 to December 2019. National, hospital, and census data were used to identify historically underserved communities by racial and ethnic composition, income distribution, and rurality.
A total of 4,984 hospitals were assessed. Results showed that over the 11-year study period, the number of hospitals with stroke certification grew from 961 (19%) to 1,763 (36%).
Without controlling for population and hospital size, hospitals in predominantly Black, racially segregated areas were 1.67-fold more likely to adopt stroke care of any level than those in predominantly non-Black, racially segregated areas (hazard ratio, 1.67; 95% confidence interval, 1.41-1.97).
However, after adjustment for population and hospital size, the likelihood of adopting stroke care among hospitals serving Black, racially segregated communities was significantly lower than among those serving non-Black, racially segregated communities (HR, 0.74; 95% CI, 0.62-0.89).
“In other words, on a per-capita basis, a hospital serving a predominantly Black, racially segregated community was 26% less likely to adopt stroke certification of any level than a hospital in a predominantly non-Black, racially segregated community,” the authors state.
In terms of socioeconomic factors, hospitals serving low-income, economically integrated (HR, 0.23) and low-income, economically segregated (HR, 0.29) areas were far less likely to adopt any level of stroke care certification than hospitals serving high-income areas, regardless of income segregation.
Rural hospitals were also much less likely to adopt any level of stroke care than urban hospitals (HR, 0.10).
“Our results suggest that it might be necessary to incentivize hospitals operating in underserved communities to seek stroke certification or to entice hospitals with higher propensity to adopt stroke care to operate in such communities so access at the per-patient level becomes more equitable,” the authors say.
This project was supported by the Pilot Project Award from the National Bureau of Economic Research Center for Aging and Health Research, funded by the National Institute on Aging and by the National Center for Advancing Translational Sciences, National Institutes of Health. Dr. Shen and Dr. Hsia have received grants from the National Institute of Aging and the National Heart, Lung, and Blood Institute.
A version of this article first appeared on Medscape.com.
Anorexia linked to notable shrinkage of key brain structures
, a new brain imaging study shows.
The reductions of cortical thickness, subcortical volumes, and cortical surface area were “very pronounced in acutely underweight anorexia,” Stefan Ehrlich, MD, PhD, head of the Eating Disorder Treatment and Research Center, Technical University, Dresden, Germany, told this news organization.
Yet even a “partial weight gain brings some normalization of these shrinkages. From this it can be deduced that a fast/early normalization of weight is also very important for brain health,” said Dr. Ehrlich.
The study was published online in Biological Psychiatry.
‘A wake-up call’
Researchers with the international ENIGMA Eating Disorders Working Group analyzed T1-weighted structural magnetic resonance imaging scans for nearly 2,000 people with AN (including those in recovery) and healthy controls across 22 sites worldwide.
In the AN sample, reductions in cortical thickness, subcortical volumes, and, to a lesser extent, cortical surface area, were “sizable (Cohen’s d up to 0.95), widespread, and co-localized with hub regions,” they report.
These reductions were two and four times larger than the abnormalities in brain size and shape seen in patients with other mental illnesses, the researchers note.
Noting the harmful impact of anorexia-related undernutrition on the brain, these deficits were associated with lower body mass index in the AN sample and were less severe in partially weight-restored patients – implying that, with appropriate early treatment and support, the brain might be able to repair itself, the investigators note.
“This really is a wake-up call, showing the need for early interventions for people with eating disorders,” Paul Thompson, PhD, author and lead scientist for the ENIGMA Consortium, said in a news release.
“The international scale of this work is extraordinary. Scientists from 22 centers worldwide pooled their brain scans to create the most detailed picture to date of how anorexia affects the brain,” Dr. Thompson added.
“The brain changes in anorexia were more severe than in other any psychiatric condition we have studied. Effects of treatments and interventions can now be evaluated, using these new brain maps as a reference,” he noted.
Immediate clinical implications
Reached for comment, Allison Eliscu, MD, chief of the division of adolescent medicine, department of pediatrics, at Stony Brook (N.Y.) University, said the findings have immediate implications for clinical care.
“When we talk to our patients and the parents, a lot of them focus on things that they can see, such as the way they look. It adds a lot to the conversation to be able to say: You’re obviously not seeing these changes in the brain, but they’re happening and could be potentially long term if you don’t start weight restoring, or if you weight restore and then continue to drop again,” Dr. Eliscu said in an interview.
The findings, she said, really do highlight what anorexia can do to the brain.
“Adolescents need to know, anorexia can absolutely decrease the size of your brain in different areas; you’re not just losing weight in your belly and your thighs, you’re losing weight in the brain as well and that’s really concerning,” said Dr. Eliscu.
The study had no commercial funding. The authors and Dr. Eliscu report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
, a new brain imaging study shows.
The reductions of cortical thickness, subcortical volumes, and cortical surface area were “very pronounced in acutely underweight anorexia,” Stefan Ehrlich, MD, PhD, head of the Eating Disorder Treatment and Research Center, Technical University, Dresden, Germany, told this news organization.
Yet even a “partial weight gain brings some normalization of these shrinkages. From this it can be deduced that a fast/early normalization of weight is also very important for brain health,” said Dr. Ehrlich.
The study was published online in Biological Psychiatry.
‘A wake-up call’
Researchers with the international ENIGMA Eating Disorders Working Group analyzed T1-weighted structural magnetic resonance imaging scans for nearly 2,000 people with AN (including those in recovery) and healthy controls across 22 sites worldwide.
In the AN sample, reductions in cortical thickness, subcortical volumes, and, to a lesser extent, cortical surface area, were “sizable (Cohen’s d up to 0.95), widespread, and co-localized with hub regions,” they report.
These reductions were two and four times larger than the abnormalities in brain size and shape seen in patients with other mental illnesses, the researchers note.
Noting the harmful impact of anorexia-related undernutrition on the brain, these deficits were associated with lower body mass index in the AN sample and were less severe in partially weight-restored patients – implying that, with appropriate early treatment and support, the brain might be able to repair itself, the investigators note.
“This really is a wake-up call, showing the need for early interventions for people with eating disorders,” Paul Thompson, PhD, author and lead scientist for the ENIGMA Consortium, said in a news release.
“The international scale of this work is extraordinary. Scientists from 22 centers worldwide pooled their brain scans to create the most detailed picture to date of how anorexia affects the brain,” Dr. Thompson added.
“The brain changes in anorexia were more severe than in other any psychiatric condition we have studied. Effects of treatments and interventions can now be evaluated, using these new brain maps as a reference,” he noted.
Immediate clinical implications
Reached for comment, Allison Eliscu, MD, chief of the division of adolescent medicine, department of pediatrics, at Stony Brook (N.Y.) University, said the findings have immediate implications for clinical care.
“When we talk to our patients and the parents, a lot of them focus on things that they can see, such as the way they look. It adds a lot to the conversation to be able to say: You’re obviously not seeing these changes in the brain, but they’re happening and could be potentially long term if you don’t start weight restoring, or if you weight restore and then continue to drop again,” Dr. Eliscu said in an interview.
The findings, she said, really do highlight what anorexia can do to the brain.
“Adolescents need to know, anorexia can absolutely decrease the size of your brain in different areas; you’re not just losing weight in your belly and your thighs, you’re losing weight in the brain as well and that’s really concerning,” said Dr. Eliscu.
The study had no commercial funding. The authors and Dr. Eliscu report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
, a new brain imaging study shows.
The reductions of cortical thickness, subcortical volumes, and cortical surface area were “very pronounced in acutely underweight anorexia,” Stefan Ehrlich, MD, PhD, head of the Eating Disorder Treatment and Research Center, Technical University, Dresden, Germany, told this news organization.
Yet even a “partial weight gain brings some normalization of these shrinkages. From this it can be deduced that a fast/early normalization of weight is also very important for brain health,” said Dr. Ehrlich.
The study was published online in Biological Psychiatry.
‘A wake-up call’
Researchers with the international ENIGMA Eating Disorders Working Group analyzed T1-weighted structural magnetic resonance imaging scans for nearly 2,000 people with AN (including those in recovery) and healthy controls across 22 sites worldwide.
In the AN sample, reductions in cortical thickness, subcortical volumes, and, to a lesser extent, cortical surface area, were “sizable (Cohen’s d up to 0.95), widespread, and co-localized with hub regions,” they report.
These reductions were two and four times larger than the abnormalities in brain size and shape seen in patients with other mental illnesses, the researchers note.
Noting the harmful impact of anorexia-related undernutrition on the brain, these deficits were associated with lower body mass index in the AN sample and were less severe in partially weight-restored patients – implying that, with appropriate early treatment and support, the brain might be able to repair itself, the investigators note.
“This really is a wake-up call, showing the need for early interventions for people with eating disorders,” Paul Thompson, PhD, author and lead scientist for the ENIGMA Consortium, said in a news release.
“The international scale of this work is extraordinary. Scientists from 22 centers worldwide pooled their brain scans to create the most detailed picture to date of how anorexia affects the brain,” Dr. Thompson added.
“The brain changes in anorexia were more severe than in other any psychiatric condition we have studied. Effects of treatments and interventions can now be evaluated, using these new brain maps as a reference,” he noted.
Immediate clinical implications
Reached for comment, Allison Eliscu, MD, chief of the division of adolescent medicine, department of pediatrics, at Stony Brook (N.Y.) University, said the findings have immediate implications for clinical care.
“When we talk to our patients and the parents, a lot of them focus on things that they can see, such as the way they look. It adds a lot to the conversation to be able to say: You’re obviously not seeing these changes in the brain, but they’re happening and could be potentially long term if you don’t start weight restoring, or if you weight restore and then continue to drop again,” Dr. Eliscu said in an interview.
The findings, she said, really do highlight what anorexia can do to the brain.
“Adolescents need to know, anorexia can absolutely decrease the size of your brain in different areas; you’re not just losing weight in your belly and your thighs, you’re losing weight in the brain as well and that’s really concerning,” said Dr. Eliscu.
The study had no commercial funding. The authors and Dr. Eliscu report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM BIOLOGICAL PSYCHIATRY
Women benefit but lag behind in intracoronary imaging in PCI
A real-world analysis reveals that women are consistently less likely to undergo intracoronary imaging as part of percutaneous coronary intervention (PCI), even though it benefits both sexes equally.
Results from nearly all PCIs performed in England and Wales between 2006 and 2019 showed the absolute rate of intracoronary imaging with either intravascular ultrasound (IVUS) or optical coherence tomography (OCT) was 5% lower in the later study years among women at 14.5%, compared with 19.6% in men (P < .001).
After adjustment, female sex was an independent predictor of lower intracoronary imaging use (odds ratio, 0.93; 95% confidence interval, 0.91-0.96), according to the study, published in JACC: Cardiovascular Interventions.
“One of the thoughts I had when we were running this analysis was, well, maybe the indications for that imaging, as recommended by guidelines, are less common in women,” Mamas Mamas, MD, told this news organization. “So what we did was to look at just cases where imaging is recommended by the EAPCI [European Association of Percutaneous Coronary Intervention].”
Again, the use of intracoronary imaging was consistently lower among women than among men for all of the following EAPCI-recommended indications:
- Acute coronary syndrome: 11.6% vs. 12.3% (P < .01).
- Stent thrombosis: 30.9% vs. 34.9% (P < .01).
- Long lesions: 13.1% vs. 16.3% (P < .01).
- Chronic total occlusions: 16.2% vs. 18.3% (P < .01).
- Left main stem PCI: 55.1% vs. 57.5% (P < .01).
- In-stent restenosis: 28.0% vs. 30.7%.
- Calcified lesions: 36.6% vs. 40.1% (P < .01).
- Renal disease: 17.4% vs. 19.5% (P < .01).
As to what might be driving the lower use, Dr. Mamas dismissed the argument that women undergo much simpler PCI, which wouldn’t benefit from imaging. Women do have smaller coronary arteries, however, and there is a belief that it’s easier to eyeball the size of vessels that are smaller rather than larger.
“I’m not convinced that’s entirely true,” he said. “I don’t have a good answer for you, I’m afraid. I don’t really know why we’re seeing it. I just think it’s one of those disparities that is important to highlight.”
Central to this belief is that the benefits of intracoronary imaging were found to be similar in men and women. Intracoronary imaging was associated with lower adjusted odds of in-hospital mortality (OR, 0.56; 95% CI, 0.48-0.64) and major adverse cardiac and cerebrovascular events (OR, 0.83; 95% CI, 0.76-0.91) in women and men (OR, 0.48; 95% CI, 0.44-0.53 and OR, 0.75; 95% CI, 0.71-0.80, respectively), compared with nonimaging groups.
“This really should be a call to arms, particularly given that we show this disparity persists, even in guideline-recommended cases where we should be using it,” said Dr. Mamas, from the Keele (England) Cardiovascular Research Group, Keele University, and Royal Stoke University Hospital, Stoke-on-Trent, England.
“Actually, I would argue that we should be using more imaging in women than men anyway because many of the presentations for acute coronary syndromes in women, like spontaneous coronary artery dissection or MINOCA [MI with nonobstructive coronary arteries], you often need intracoronary imaging to make that kind of diagnosis,” he observed.
Getting worse, not better
Previous studies have shown that women are less likely than men in acute coronary syndromes to receive the transradial approach and P2Y12 inhibitors, but none have specifically looked at intracoronary imaging, Dr. Mamas said.
To fill the gap, the researchers drew on data from 994,478 patients in the British Cardiovascular Intervention Society registry, of whom, 8.4% of 738,616 men and 7.9% of 255,862 women received intracoronary imaging.
Women in the imaging group were older, more likely to be an ethnic minority, and more likely to undergo PCI for non–ST-segment elevation MI than their male counterparts.
One of the more surprising findings was that rates of IVUS and OCT were superimposable between the sexes at the start of the study but quickly diverged starting in around 2012, when the technology took off, Dr. Mamas said. In the most recent data, use was about 3% lower in women overall and rising to 6% in those with stable angina.
“Whilst the disparities between men and women are significant, the bigger question is why are we using so little imaging in guideline-recommended cases where there is a benefit?” he said.
Possible actionable items, he suggested, include providing older physicians who didn’t have access to intracoronary imaging during their training with opportunities in their cath lab or with industry sponsors to increase their skills and confidence. Intracoronary imaging use could also be routinely captured in U.S. and European PCI registries and used as a quality metric.
“In left main, you see a massive difference between centers, and that’s the kind of data that drives discussion,” Dr. Mamas said. “If we start reporting quality metrics, such as radial use, intracoronary imaging, P2Y12 inhibitors by center, then you’ve got something to benchmark centers against.”
Nathaniel Smilowitz, MD, an interventional cardiologist at New York Langone Health, who was not associated with the study, said that it’s troubling to see that the utilization intravascular imaging is so low, despite randomized trials and large meta-analyses showing a mortality benefit associated with its use in PCI.
“Even among men, only 19.6% in the later years were getting intravascular imaging performed to guide their coronary intervention, so one out of five,” he said. “There are opportunities to improve.”
Dr. Smilowitz said he’s also perplexed as to why adoption would be lower in women but that the findings echo those in other domains where women receive less intensive cardiovascular therapy.
“There’s no biological, really plausible, mechanism as to why the need for intravascular imaging would be lower and, particularly, because they showed in stent thrombosis, for example, where intravascular imaging is tremendously important, there were still sex differences,” he said. “So even with clear indications for imaging, women just received the optimal therapy less often than men. It’s disappointing.”
Dr. Smilowitz agreed that there may be a need to incorporate intravascular imaging into metrics, which are reported back to physicians, potentially even for comparisons with peers or regional rates to incentivize physicians to improve uptake.
“As a society, we’ve been quite slow to integrate intravascular imaging to guide PCI and we can do better,” he said.
A version of this article first appeared on Medscape.com.
A real-world analysis reveals that women are consistently less likely to undergo intracoronary imaging as part of percutaneous coronary intervention (PCI), even though it benefits both sexes equally.
Results from nearly all PCIs performed in England and Wales between 2006 and 2019 showed the absolute rate of intracoronary imaging with either intravascular ultrasound (IVUS) or optical coherence tomography (OCT) was 5% lower in the later study years among women at 14.5%, compared with 19.6% in men (P < .001).
After adjustment, female sex was an independent predictor of lower intracoronary imaging use (odds ratio, 0.93; 95% confidence interval, 0.91-0.96), according to the study, published in JACC: Cardiovascular Interventions.
“One of the thoughts I had when we were running this analysis was, well, maybe the indications for that imaging, as recommended by guidelines, are less common in women,” Mamas Mamas, MD, told this news organization. “So what we did was to look at just cases where imaging is recommended by the EAPCI [European Association of Percutaneous Coronary Intervention].”
Again, the use of intracoronary imaging was consistently lower among women than among men for all of the following EAPCI-recommended indications:
- Acute coronary syndrome: 11.6% vs. 12.3% (P < .01).
- Stent thrombosis: 30.9% vs. 34.9% (P < .01).
- Long lesions: 13.1% vs. 16.3% (P < .01).
- Chronic total occlusions: 16.2% vs. 18.3% (P < .01).
- Left main stem PCI: 55.1% vs. 57.5% (P < .01).
- In-stent restenosis: 28.0% vs. 30.7%.
- Calcified lesions: 36.6% vs. 40.1% (P < .01).
- Renal disease: 17.4% vs. 19.5% (P < .01).
As to what might be driving the lower use, Dr. Mamas dismissed the argument that women undergo much simpler PCI, which wouldn’t benefit from imaging. Women do have smaller coronary arteries, however, and there is a belief that it’s easier to eyeball the size of vessels that are smaller rather than larger.
“I’m not convinced that’s entirely true,” he said. “I don’t have a good answer for you, I’m afraid. I don’t really know why we’re seeing it. I just think it’s one of those disparities that is important to highlight.”
Central to this belief is that the benefits of intracoronary imaging were found to be similar in men and women. Intracoronary imaging was associated with lower adjusted odds of in-hospital mortality (OR, 0.56; 95% CI, 0.48-0.64) and major adverse cardiac and cerebrovascular events (OR, 0.83; 95% CI, 0.76-0.91) in women and men (OR, 0.48; 95% CI, 0.44-0.53 and OR, 0.75; 95% CI, 0.71-0.80, respectively), compared with nonimaging groups.
“This really should be a call to arms, particularly given that we show this disparity persists, even in guideline-recommended cases where we should be using it,” said Dr. Mamas, from the Keele (England) Cardiovascular Research Group, Keele University, and Royal Stoke University Hospital, Stoke-on-Trent, England.
“Actually, I would argue that we should be using more imaging in women than men anyway because many of the presentations for acute coronary syndromes in women, like spontaneous coronary artery dissection or MINOCA [MI with nonobstructive coronary arteries], you often need intracoronary imaging to make that kind of diagnosis,” he observed.
Getting worse, not better
Previous studies have shown that women are less likely than men in acute coronary syndromes to receive the transradial approach and P2Y12 inhibitors, but none have specifically looked at intracoronary imaging, Dr. Mamas said.
To fill the gap, the researchers drew on data from 994,478 patients in the British Cardiovascular Intervention Society registry, of whom, 8.4% of 738,616 men and 7.9% of 255,862 women received intracoronary imaging.
Women in the imaging group were older, more likely to be an ethnic minority, and more likely to undergo PCI for non–ST-segment elevation MI than their male counterparts.
One of the more surprising findings was that rates of IVUS and OCT were superimposable between the sexes at the start of the study but quickly diverged starting in around 2012, when the technology took off, Dr. Mamas said. In the most recent data, use was about 3% lower in women overall and rising to 6% in those with stable angina.
“Whilst the disparities between men and women are significant, the bigger question is why are we using so little imaging in guideline-recommended cases where there is a benefit?” he said.
Possible actionable items, he suggested, include providing older physicians who didn’t have access to intracoronary imaging during their training with opportunities in their cath lab or with industry sponsors to increase their skills and confidence. Intracoronary imaging use could also be routinely captured in U.S. and European PCI registries and used as a quality metric.
“In left main, you see a massive difference between centers, and that’s the kind of data that drives discussion,” Dr. Mamas said. “If we start reporting quality metrics, such as radial use, intracoronary imaging, P2Y12 inhibitors by center, then you’ve got something to benchmark centers against.”
Nathaniel Smilowitz, MD, an interventional cardiologist at New York Langone Health, who was not associated with the study, said that it’s troubling to see that the utilization intravascular imaging is so low, despite randomized trials and large meta-analyses showing a mortality benefit associated with its use in PCI.
“Even among men, only 19.6% in the later years were getting intravascular imaging performed to guide their coronary intervention, so one out of five,” he said. “There are opportunities to improve.”
Dr. Smilowitz said he’s also perplexed as to why adoption would be lower in women but that the findings echo those in other domains where women receive less intensive cardiovascular therapy.
“There’s no biological, really plausible, mechanism as to why the need for intravascular imaging would be lower and, particularly, because they showed in stent thrombosis, for example, where intravascular imaging is tremendously important, there were still sex differences,” he said. “So even with clear indications for imaging, women just received the optimal therapy less often than men. It’s disappointing.”
Dr. Smilowitz agreed that there may be a need to incorporate intravascular imaging into metrics, which are reported back to physicians, potentially even for comparisons with peers or regional rates to incentivize physicians to improve uptake.
“As a society, we’ve been quite slow to integrate intravascular imaging to guide PCI and we can do better,” he said.
A version of this article first appeared on Medscape.com.
A real-world analysis reveals that women are consistently less likely to undergo intracoronary imaging as part of percutaneous coronary intervention (PCI), even though it benefits both sexes equally.
Results from nearly all PCIs performed in England and Wales between 2006 and 2019 showed the absolute rate of intracoronary imaging with either intravascular ultrasound (IVUS) or optical coherence tomography (OCT) was 5% lower in the later study years among women at 14.5%, compared with 19.6% in men (P < .001).
After adjustment, female sex was an independent predictor of lower intracoronary imaging use (odds ratio, 0.93; 95% confidence interval, 0.91-0.96), according to the study, published in JACC: Cardiovascular Interventions.
“One of the thoughts I had when we were running this analysis was, well, maybe the indications for that imaging, as recommended by guidelines, are less common in women,” Mamas Mamas, MD, told this news organization. “So what we did was to look at just cases where imaging is recommended by the EAPCI [European Association of Percutaneous Coronary Intervention].”
Again, the use of intracoronary imaging was consistently lower among women than among men for all of the following EAPCI-recommended indications:
- Acute coronary syndrome: 11.6% vs. 12.3% (P < .01).
- Stent thrombosis: 30.9% vs. 34.9% (P < .01).
- Long lesions: 13.1% vs. 16.3% (P < .01).
- Chronic total occlusions: 16.2% vs. 18.3% (P < .01).
- Left main stem PCI: 55.1% vs. 57.5% (P < .01).
- In-stent restenosis: 28.0% vs. 30.7%.
- Calcified lesions: 36.6% vs. 40.1% (P < .01).
- Renal disease: 17.4% vs. 19.5% (P < .01).
As to what might be driving the lower use, Dr. Mamas dismissed the argument that women undergo much simpler PCI, which wouldn’t benefit from imaging. Women do have smaller coronary arteries, however, and there is a belief that it’s easier to eyeball the size of vessels that are smaller rather than larger.
“I’m not convinced that’s entirely true,” he said. “I don’t have a good answer for you, I’m afraid. I don’t really know why we’re seeing it. I just think it’s one of those disparities that is important to highlight.”
Central to this belief is that the benefits of intracoronary imaging were found to be similar in men and women. Intracoronary imaging was associated with lower adjusted odds of in-hospital mortality (OR, 0.56; 95% CI, 0.48-0.64) and major adverse cardiac and cerebrovascular events (OR, 0.83; 95% CI, 0.76-0.91) in women and men (OR, 0.48; 95% CI, 0.44-0.53 and OR, 0.75; 95% CI, 0.71-0.80, respectively), compared with nonimaging groups.
“This really should be a call to arms, particularly given that we show this disparity persists, even in guideline-recommended cases where we should be using it,” said Dr. Mamas, from the Keele (England) Cardiovascular Research Group, Keele University, and Royal Stoke University Hospital, Stoke-on-Trent, England.
“Actually, I would argue that we should be using more imaging in women than men anyway because many of the presentations for acute coronary syndromes in women, like spontaneous coronary artery dissection or MINOCA [MI with nonobstructive coronary arteries], you often need intracoronary imaging to make that kind of diagnosis,” he observed.
Getting worse, not better
Previous studies have shown that women are less likely than men in acute coronary syndromes to receive the transradial approach and P2Y12 inhibitors, but none have specifically looked at intracoronary imaging, Dr. Mamas said.
To fill the gap, the researchers drew on data from 994,478 patients in the British Cardiovascular Intervention Society registry, of whom, 8.4% of 738,616 men and 7.9% of 255,862 women received intracoronary imaging.
Women in the imaging group were older, more likely to be an ethnic minority, and more likely to undergo PCI for non–ST-segment elevation MI than their male counterparts.
One of the more surprising findings was that rates of IVUS and OCT were superimposable between the sexes at the start of the study but quickly diverged starting in around 2012, when the technology took off, Dr. Mamas said. In the most recent data, use was about 3% lower in women overall and rising to 6% in those with stable angina.
“Whilst the disparities between men and women are significant, the bigger question is why are we using so little imaging in guideline-recommended cases where there is a benefit?” he said.
Possible actionable items, he suggested, include providing older physicians who didn’t have access to intracoronary imaging during their training with opportunities in their cath lab or with industry sponsors to increase their skills and confidence. Intracoronary imaging use could also be routinely captured in U.S. and European PCI registries and used as a quality metric.
“In left main, you see a massive difference between centers, and that’s the kind of data that drives discussion,” Dr. Mamas said. “If we start reporting quality metrics, such as radial use, intracoronary imaging, P2Y12 inhibitors by center, then you’ve got something to benchmark centers against.”
Nathaniel Smilowitz, MD, an interventional cardiologist at New York Langone Health, who was not associated with the study, said that it’s troubling to see that the utilization intravascular imaging is so low, despite randomized trials and large meta-analyses showing a mortality benefit associated with its use in PCI.
“Even among men, only 19.6% in the later years were getting intravascular imaging performed to guide their coronary intervention, so one out of five,” he said. “There are opportunities to improve.”
Dr. Smilowitz said he’s also perplexed as to why adoption would be lower in women but that the findings echo those in other domains where women receive less intensive cardiovascular therapy.
“There’s no biological, really plausible, mechanism as to why the need for intravascular imaging would be lower and, particularly, because they showed in stent thrombosis, for example, where intravascular imaging is tremendously important, there were still sex differences,” he said. “So even with clear indications for imaging, women just received the optimal therapy less often than men. It’s disappointing.”
Dr. Smilowitz agreed that there may be a need to incorporate intravascular imaging into metrics, which are reported back to physicians, potentially even for comparisons with peers or regional rates to incentivize physicians to improve uptake.
“As a society, we’ve been quite slow to integrate intravascular imaging to guide PCI and we can do better,” he said.
A version of this article first appeared on Medscape.com.
FROM JACC: CARDIOVASCULAR INTERVENTIONS
White children more likely to get imaging in EDs: Study
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
Non-Hispanic White children were more likely to receive diagnostic imaging at children’s hospitals’ emergency departments across the United States than were Hispanic children and non-Hispanic Black children, according to a large study published in JAMA Network Open.
Researchers found that, the more the percentage of children from minority groups cared for by a hospital increased, the wider the imaging gap between those children and non-Hispanic White children.
The cross-sectional study, led by Margaret E. Samuels-Kalow, MD, MPhil, MSHP, with the department of emergency medicine, Massachusetts General Hospital and Harvard Medical School in Boston, included 38 children’s hospitals and more than 12 million ED visits.
“These findings emphasize the urgent need for interventions at the hospital level to improve equity in imaging in pediatric emergency medicine,” the authors write.
Patients included in the study were younger than 18 and visited an ED from January 2016 through December 2019. Data were pulled from the Pediatric Health Information System.
Of the more than 12 million visits in this study, 3.5 million (28.7%) involved at least one diagnostic imaging test.
Diagnostic imaging was performed in 1.5 million visits (34.2%) for non-Hispanic White children; 790,961 (24.6%) for non-Hispanic Black children; and 907,222 (26.1%) for Hispanic children (P < .001).
Non-Hispanic Black children were consistently less likely to get diagnostic imaging than non-Hispanic White counterparts at every hospital in the study, no matter the imaging modality: radiography, ultrasonography, computed tomography, or magnetic resonance imaging.
Hispanic patients were generally less likely to get imaging than non-Hispanic White patients, though results were less consistent for ultrasound and MRI.
In a sensitivity analysis, when looking at imaging from patients’ first visit across the study cohort, non-Hispanic Black children were significantly less likely to get imaging than non-Hispanic White children (adjusted odds ratio, 0.77; 95% confidence interval, 0.74-0.79).
“This remained significant even after adjustment for a priori specified confounders including hospital propensity to image,” the authors write.
Authors acknowledge that it is possible that some of the differences may be attributable to the patient mix regarding severity of cases or indications for imaging by hospital, but they note that all models were adjusted for diagnosis-related group and other potential confounders.
This study did not assess whether one group is being overtested. Researchers also note that higher rates of imaging do not necessarily indicate higher quality of care.
However, the authors note, previous research has suggested overtesting of non-Hispanic White patients for head CT and chest pain, as well as patterns of overtreatment of non-Hispanic White patients who have bronchiolitis or viral upper respiratory tract infections.
Medell Briggs-Malonson, MD, MPH, chief of health equity, diversity and inclusion for the University of California, Los Angeles, Hospital and Clinic System, who was not part of the study, said in an interview “this all rings true.”
“This is not the first study we have had in either the pediatric or adult populations that shows disparate levels of care as well as health outcomes. Now we are starting to be able to measure it,” she said.
This study is further evidence of medical racism, she says, and highlights that it’s not the hospital choice or the insurance type affecting the numbers, she said.
“When you control for those factors, it looks to be it’s only due to race and that’s because of the very deep levels of implicit bias as well as explicit bias that we still have in our health systems and even in our providers,” said Dr. Briggs-Malonson, who is also an associate professor of emergency medicine at UCLA. “It’s incredibly important to identify and immediately address.”
What can be done?
Changing these patterns starts with knowing the numbers, the authors write.
“Hospitals should measure their own differences in imaging rates and increase awareness of existing areas of differential treatment as a starting point for improvement,” Dr. Samuels-Kalow and coauthors say.
Dr. Briggs-Malonson added that guidelines are very clear about when children should get imaging. Adhering to evidence-based guidelines can help avoid variations in care from external factors.
“If children are not receiving the absolute best comprehensive evaluation in the emergency department that they deserve, we can miss many different illnesses, which can lead to worse outcomes,” she noted.
As for what might motivate lack of imaging, Dr. Briggs-Malonson pointed to longstanding trends of providers thinking complaints raised by minority patients may not be as severe as they report. Conversely, in caring for White patients there may be a feeling that more tests and imaging may be better out of more fear of missing something, she said.
At UCLA, she says, dashboards have been developed to track statistics on care by age, race, ethnicity, language, insurance type, etc., though not specifically in pediatric imaging, to assess and address any care inequities.
Summer L. Kaplan, MD, MS, director of emergency radiology at Children’s Hospital of Philadelphia, who also was not part of the study, said the finding of racial disparities in pediatric ED imaging provides evidence that gaps still exist in providing the best care to all children and families seeking emergency care.
“However, it is important to recognize that more imaging does not equal better care,” she said. “More imaging may be associated with unnecessary, low-value tests that may add radiation and other risks but do not improve care.”
She said higher rates of imaging may occur when patients present early in the course of a disease, when the differential diagnosis remains broad.
If families have delayed seeking care because of time constraints, transportation problems, cost of care, or mistrust of the health system, children may present later in the course of a disease and require less imaging for a diagnosis, she explained.
“This paper offers a valuable look at the inequities that exist in pediatric emergency imaging use, and further research will be essential to understand and address the causes of these differences,” Dr. Kaplan said.
A coauthor reported compensation as a member of a Medical Review Committee for Highmark. Other coauthors reported grants from the U.S. Agency for Healthcare Research and Quality outside the submitted work. Dr. Briggs-Malonson and Dr. Kaplan reported no relevant financial relationships.
FROM JAMA NETWORK OPEN
Coronary CT Angiography Compared to Coronary Angiography or Standard of Care in Patients With Intermediate-Risk Stable Chest Pain
Study 1 Overview (SCOT-HEART Investigators)
Objective: To assess cardiovascular mortality and nonfatal myocardial infarction at 5 years in patients with stable chest pain referred to cardiology clinic for management with either standard care plus computed tomography angiography (CTA) or standard care alone.
Design: Multicenter, randomized, open-label prospective study.
Setting and participants: A total of 4146 patients with stable chest pain were randomized to standard care or standard care plus CTA at 12 centers across Scotland and were followed for 5 years.
Main outcome measures: The primary end point was a composite of death from coronary heart disease or nonfatal myocardial infarction. Main secondary end points were nonfatal myocardial infarction, nonfatal stroke, and frequency of invasive coronary angiography (ICA) and coronary revascularization with percutaneous coronary intervention or coronary artery bypass grafting.
Main results: The primary outcome including the composite of cardiovascular death or nonfatal myocardial infarction was lower in the CTA group than in the standard-care group at 2.3% (48 of 2073 patients) vs 3.9% (81 of 2073 patients), respectively (hazard ratio, 0.59; 95% CI, 0.41-0.84; P = .004). Although there was a higher rate of ICA and coronary revascularization in the CTA group than in the standard-care group in the first few months of follow-up, the overall rates were similar at 5 years, with ICA performed in 491 patients and 502 patients in the CTA vs standard-care groups, respectively (hazard ratio, 1.00; 95% CI, 0.88-1.13). Similarly, coronary revascularization was performed in 279 patients in the CTA group and in 267 patients in the standard-care group (hazard ratio, 1.07; 95% CI, 0.91-1.27). There were, however, more preventive therapies initiated in patients in the CTA group than in the standard-care group (odds ratio, 1.40; 95% CI, 1.19-1.65).
Conclusion: In patients with stable chest pain, the use of CTA in addition to standard care resulted in a significantly lower rate of death from coronary heart disease or nonfatal myocardial infarction at 5 years; the main contributor to this outcome was a reduced nonfatal myocardial infarction rate. There was no difference in the rate of coronary angiography or coronary revascularization between the 2 groups at 5 years.
Study 2 Overview (DISCHARGE Trial Group)
Objective: To compare the effectiveness of computed tomography (CT) with ICA as a diagnostic tool in patients with stable chest pain and intermediate pretest probability of coronary artery disease (CAD).
Design: Multicenter, randomized, assessor-blinded pragmatic prospective study.
Setting and participants: A total of 3667 patients with stable chest pain and intermediate pretest probability of CAD were enrolled at 26 centers and randomized into CT or ICA groups. Only 3561 patients were included in the modified intention-to-treat analysis, with 1808 patients and 1753 patients in the CT and ICA groups, respectively.
Main outcome measures: The primary outcome was a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke over 3.5 years. The main secondary outcomes were major procedure-related complications and patient-reported angina pectoris during the last 4 weeks of follow up.
Main results: The primary outcome occurred in 38 of 1808 patients (2.1%) in the CT group and in 52 of 1753 patients (3.0%) in the ICA group (hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). The secondary outcomes showed that major procedure-related complications occurred in 9 patients (0.5%) in the CT group and in 33 patients (1.9%) in the ICA group (hazard ratio, 0.26; 95% CI, 0.13-0.55). Rates of patient-reported angina in the final 4 weeks of follow-up were 8.8% in the CT group and 7.5% in the ICA group (odds ratio, 1.17; 95% CI, 0.92-1.48).
Conclusion: Risk of major adverse cardiovascular events from the primary outcome were similar in both the CT and ICA groups among patients with stable chest pain and intermediate pretest probability of CAD. Patients referred for CT had a lower rate of coronary angiography leading to fewer major procedure-related complications in these patients than in those referred for ICA.
Commentary
Evaluation and treatment of obstructive atherosclerosis is an important part of clinical care in patients presenting with angina symptoms.1 Thus, the initial investigation for patients with suspected obstructive CAD includes ruling out acute coronary syndrome and assessing quality of life.1 The diagnostic test should be tailored to the pretest probability for the diagnosis of obstructive CAD.2
In the United States, stress testing traditionally has been used for the initial assessment in patients with suspected CAD,3 but recently CTA has been utilized more frequently for this purpose. Compared to a stress test, which often helps identify and assess ischemia, CTA can provide anatomical assessment, with higher sensitivity to identify CAD.4 Furthermore, it can distinguish nonobstructive plaques that can be challenging to identify with stress test alone.
Whether CTA is superior to stress testing as the initial assessment for CAD has been debated. The randomized PROMISE trial compared patients with stable angina who underwent functional stress testing or CTA as an initial strategy.5 They reported a similar outcome between the 2 groups at a median follow-up of 2 years. However, in the original SCOT-HEART trial (CT coronary angiography in patients with suspected angina due to coronary heart disease), which was published in the same year as the PROMISE trial, the patients who underwent initial assessment with CTA had a numerically lower composite end point of cardiac death and myocardial infarction at a median follow-up of 1.7 years (1.3% vs 2.0%, P = .053).6
Given this result, the SCOT-HEART investigators extended the follow-up to evaluate the composite end point of death from coronary heart disease or nonfatal myocardial infarction at 5 years.7 This trial enrolled patients who were initially referred to a cardiology clinic for evaluation of chest pain, and they were randomized to standard care plus CTA or standard care alone. At a median duration of 4.8 years, the primary outcome was lower in the CTA group (2.3%, 48 patients) than in the standard-care group (3.9%, 81 patients) (hazard ratio, 0.58; 95% CI, 0.41-0.84; P = .004). Both groups had similar rates of invasive coronary angiography and had similar coronary revascularization rates.
It is hypothesized that this lower rate of nonfatal myocardial infarction in patients with CTA plus standard care is associated with a higher rate of preventive therapies initiated in patients in the CTA-plus-standard-care group compared to standard care alone. However, the difference in the standard-care group should be noted when compared to the PROMISE trial. In the PROMISE trial, the comparator group had predominantly stress imaging (either nuclear stress test or echocardiography), while in the SCOT-HEART trial, the group had predominantly stress electrocardiogram (ECG), and only 10% of the patients underwent stress imaging. It is possible the difference seen in the rate of nonfatal myocardial infarction was due to suboptimal diagnosis of CAD with stress ECG, which has lower sensitivity compared to stress imaging.
The DISCHARGE trial investigated the effectiveness of CTA vs ICA as the initial diagnostic test in the management of patients with stable chest pain and an intermediate pretest probability of obstructive CAD.8 At 3.5 years of follow-up, the primary composite of cardiovascular death, myocardial infarction, or stroke was similar in both groups (2.1% vs 3.0; hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). Importantly, as fewer patients underwent ICA, the risk of procedure-related complication was lower in the CTA group than in the ICA group. However, it is important to note that only 25% of the patients diagnosed with obstructive CAD had greater than 50% vessel stenosis, which raises the question of whether an initial invasive strategy is appropriate for this population.
The strengths of these 2 studies include the large number of patients enrolled along with adequate follow-up, 5 years in the SCOT-HEART trial and 3.5 years in the DISCHARGE trial. The 2 studies overall suggest the usefulness of CTA for assessment of CAD. However, the control groups were very different in these 2 trials. In the SCOT-HEART study, the comparator group was primarily assessed by stress ECG, while in the DISCHARGE study, the comparator group was primary assessed by ICA. In the PROMISE trial, the composite end point of death, myocardial infarction, hospitalization for unstable angina, or major procedural complication was similar when the strategy of initial CTA was compared to functional testing with imaging (exercise ECG, nuclear stress testing, or echocardiography).5 Thus, clinical assessment is still needed when clinicians are selecting the appropriate diagnostic test for patients with suspected CAD. The most recent guidelines give similar recommendations for CTA compared to stress imaging.9 Whether further improvement in CTA acquisition or the addition of CT fractional flow reserve can further improve outcomes requires additional study.
Applications for Clinical Practice and System Implementation
In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful in diagnosis compared to stress ECG and in reducing utilization of low-yield ICA. Whether CTA is more useful compared to the other noninvasive stress imaging modalities in this population requires further study.
Practice Points
- In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful compared to stress ECG.
- Use of CTA can potentially reduce the use of low-yield coronary angiography.
–Thai Nguyen, MD, Albert Chan, MD, Taishi Hirai, MD
University of Missouri, Columbia, MO
1. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41(3):407-477. doi:10.1093/eurheartj/ehz425
2. Nakano S, Kohsaka S, Chikamori T et al. JCS 2022 guideline focused update on diagnosis and treatment in patients with stable coronary artery disease. Circ J. 2022;86(5):882-915. doi:10.1253/circj.CJ-21-1041.
3. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012;60(24):e44-e164. doi:10.1016/j.jacc.2012.07.013
4. Arbab-Zadeh A, Di Carli MF, Cerci R, et al. Accuracy of computed tomographic angiography and single-photon emission computed tomography-acquired myocardial perfusion imaging for the diagnosis of coronary artery disease. Circ Cardiovasc Imaging. 2015;8(10):e003533. doi:10.1161/CIRCIMAGING
5. Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015;372(14):1291-300. doi:10.1056/NEJMoa1415516
6. SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet. 2015;385:2383-2391. doi:10.1016/S0140-6736(15)60291-4
7. SCOT-HEART Investigators, Newby DE, Adamson PD, et al. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med. 2018;379(10):924-933. doi:10.1056/NEJMoa1805971
8. DISCHARGE Trial Group, Maurovich-Horvat P, Bosserdt M, et al. CT or invasive coronary angiography in stable chest pain. N Engl J Med. 2022;386(17):1591-1602. doi:10.1056/NEJMoa2200963
9. Writing Committee Members, Lawton JS, Tamis-Holland JE, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
Study 1 Overview (SCOT-HEART Investigators)
Objective: To assess cardiovascular mortality and nonfatal myocardial infarction at 5 years in patients with stable chest pain referred to cardiology clinic for management with either standard care plus computed tomography angiography (CTA) or standard care alone.
Design: Multicenter, randomized, open-label prospective study.
Setting and participants: A total of 4146 patients with stable chest pain were randomized to standard care or standard care plus CTA at 12 centers across Scotland and were followed for 5 years.
Main outcome measures: The primary end point was a composite of death from coronary heart disease or nonfatal myocardial infarction. Main secondary end points were nonfatal myocardial infarction, nonfatal stroke, and frequency of invasive coronary angiography (ICA) and coronary revascularization with percutaneous coronary intervention or coronary artery bypass grafting.
Main results: The primary outcome including the composite of cardiovascular death or nonfatal myocardial infarction was lower in the CTA group than in the standard-care group at 2.3% (48 of 2073 patients) vs 3.9% (81 of 2073 patients), respectively (hazard ratio, 0.59; 95% CI, 0.41-0.84; P = .004). Although there was a higher rate of ICA and coronary revascularization in the CTA group than in the standard-care group in the first few months of follow-up, the overall rates were similar at 5 years, with ICA performed in 491 patients and 502 patients in the CTA vs standard-care groups, respectively (hazard ratio, 1.00; 95% CI, 0.88-1.13). Similarly, coronary revascularization was performed in 279 patients in the CTA group and in 267 patients in the standard-care group (hazard ratio, 1.07; 95% CI, 0.91-1.27). There were, however, more preventive therapies initiated in patients in the CTA group than in the standard-care group (odds ratio, 1.40; 95% CI, 1.19-1.65).
Conclusion: In patients with stable chest pain, the use of CTA in addition to standard care resulted in a significantly lower rate of death from coronary heart disease or nonfatal myocardial infarction at 5 years; the main contributor to this outcome was a reduced nonfatal myocardial infarction rate. There was no difference in the rate of coronary angiography or coronary revascularization between the 2 groups at 5 years.
Study 2 Overview (DISCHARGE Trial Group)
Objective: To compare the effectiveness of computed tomography (CT) with ICA as a diagnostic tool in patients with stable chest pain and intermediate pretest probability of coronary artery disease (CAD).
Design: Multicenter, randomized, assessor-blinded pragmatic prospective study.
Setting and participants: A total of 3667 patients with stable chest pain and intermediate pretest probability of CAD were enrolled at 26 centers and randomized into CT or ICA groups. Only 3561 patients were included in the modified intention-to-treat analysis, with 1808 patients and 1753 patients in the CT and ICA groups, respectively.
Main outcome measures: The primary outcome was a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke over 3.5 years. The main secondary outcomes were major procedure-related complications and patient-reported angina pectoris during the last 4 weeks of follow up.
Main results: The primary outcome occurred in 38 of 1808 patients (2.1%) in the CT group and in 52 of 1753 patients (3.0%) in the ICA group (hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). The secondary outcomes showed that major procedure-related complications occurred in 9 patients (0.5%) in the CT group and in 33 patients (1.9%) in the ICA group (hazard ratio, 0.26; 95% CI, 0.13-0.55). Rates of patient-reported angina in the final 4 weeks of follow-up were 8.8% in the CT group and 7.5% in the ICA group (odds ratio, 1.17; 95% CI, 0.92-1.48).
Conclusion: Risk of major adverse cardiovascular events from the primary outcome were similar in both the CT and ICA groups among patients with stable chest pain and intermediate pretest probability of CAD. Patients referred for CT had a lower rate of coronary angiography leading to fewer major procedure-related complications in these patients than in those referred for ICA.
Commentary
Evaluation and treatment of obstructive atherosclerosis is an important part of clinical care in patients presenting with angina symptoms.1 Thus, the initial investigation for patients with suspected obstructive CAD includes ruling out acute coronary syndrome and assessing quality of life.1 The diagnostic test should be tailored to the pretest probability for the diagnosis of obstructive CAD.2
In the United States, stress testing traditionally has been used for the initial assessment in patients with suspected CAD,3 but recently CTA has been utilized more frequently for this purpose. Compared to a stress test, which often helps identify and assess ischemia, CTA can provide anatomical assessment, with higher sensitivity to identify CAD.4 Furthermore, it can distinguish nonobstructive plaques that can be challenging to identify with stress test alone.
Whether CTA is superior to stress testing as the initial assessment for CAD has been debated. The randomized PROMISE trial compared patients with stable angina who underwent functional stress testing or CTA as an initial strategy.5 They reported a similar outcome between the 2 groups at a median follow-up of 2 years. However, in the original SCOT-HEART trial (CT coronary angiography in patients with suspected angina due to coronary heart disease), which was published in the same year as the PROMISE trial, the patients who underwent initial assessment with CTA had a numerically lower composite end point of cardiac death and myocardial infarction at a median follow-up of 1.7 years (1.3% vs 2.0%, P = .053).6
Given this result, the SCOT-HEART investigators extended the follow-up to evaluate the composite end point of death from coronary heart disease or nonfatal myocardial infarction at 5 years.7 This trial enrolled patients who were initially referred to a cardiology clinic for evaluation of chest pain, and they were randomized to standard care plus CTA or standard care alone. At a median duration of 4.8 years, the primary outcome was lower in the CTA group (2.3%, 48 patients) than in the standard-care group (3.9%, 81 patients) (hazard ratio, 0.58; 95% CI, 0.41-0.84; P = .004). Both groups had similar rates of invasive coronary angiography and had similar coronary revascularization rates.
It is hypothesized that this lower rate of nonfatal myocardial infarction in patients with CTA plus standard care is associated with a higher rate of preventive therapies initiated in patients in the CTA-plus-standard-care group compared to standard care alone. However, the difference in the standard-care group should be noted when compared to the PROMISE trial. In the PROMISE trial, the comparator group had predominantly stress imaging (either nuclear stress test or echocardiography), while in the SCOT-HEART trial, the group had predominantly stress electrocardiogram (ECG), and only 10% of the patients underwent stress imaging. It is possible the difference seen in the rate of nonfatal myocardial infarction was due to suboptimal diagnosis of CAD with stress ECG, which has lower sensitivity compared to stress imaging.
The DISCHARGE trial investigated the effectiveness of CTA vs ICA as the initial diagnostic test in the management of patients with stable chest pain and an intermediate pretest probability of obstructive CAD.8 At 3.5 years of follow-up, the primary composite of cardiovascular death, myocardial infarction, or stroke was similar in both groups (2.1% vs 3.0; hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). Importantly, as fewer patients underwent ICA, the risk of procedure-related complication was lower in the CTA group than in the ICA group. However, it is important to note that only 25% of the patients diagnosed with obstructive CAD had greater than 50% vessel stenosis, which raises the question of whether an initial invasive strategy is appropriate for this population.
The strengths of these 2 studies include the large number of patients enrolled along with adequate follow-up, 5 years in the SCOT-HEART trial and 3.5 years in the DISCHARGE trial. The 2 studies overall suggest the usefulness of CTA for assessment of CAD. However, the control groups were very different in these 2 trials. In the SCOT-HEART study, the comparator group was primarily assessed by stress ECG, while in the DISCHARGE study, the comparator group was primary assessed by ICA. In the PROMISE trial, the composite end point of death, myocardial infarction, hospitalization for unstable angina, or major procedural complication was similar when the strategy of initial CTA was compared to functional testing with imaging (exercise ECG, nuclear stress testing, or echocardiography).5 Thus, clinical assessment is still needed when clinicians are selecting the appropriate diagnostic test for patients with suspected CAD. The most recent guidelines give similar recommendations for CTA compared to stress imaging.9 Whether further improvement in CTA acquisition or the addition of CT fractional flow reserve can further improve outcomes requires additional study.
Applications for Clinical Practice and System Implementation
In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful in diagnosis compared to stress ECG and in reducing utilization of low-yield ICA. Whether CTA is more useful compared to the other noninvasive stress imaging modalities in this population requires further study.
Practice Points
- In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful compared to stress ECG.
- Use of CTA can potentially reduce the use of low-yield coronary angiography.
–Thai Nguyen, MD, Albert Chan, MD, Taishi Hirai, MD
University of Missouri, Columbia, MO
Study 1 Overview (SCOT-HEART Investigators)
Objective: To assess cardiovascular mortality and nonfatal myocardial infarction at 5 years in patients with stable chest pain referred to cardiology clinic for management with either standard care plus computed tomography angiography (CTA) or standard care alone.
Design: Multicenter, randomized, open-label prospective study.
Setting and participants: A total of 4146 patients with stable chest pain were randomized to standard care or standard care plus CTA at 12 centers across Scotland and were followed for 5 years.
Main outcome measures: The primary end point was a composite of death from coronary heart disease or nonfatal myocardial infarction. Main secondary end points were nonfatal myocardial infarction, nonfatal stroke, and frequency of invasive coronary angiography (ICA) and coronary revascularization with percutaneous coronary intervention or coronary artery bypass grafting.
Main results: The primary outcome including the composite of cardiovascular death or nonfatal myocardial infarction was lower in the CTA group than in the standard-care group at 2.3% (48 of 2073 patients) vs 3.9% (81 of 2073 patients), respectively (hazard ratio, 0.59; 95% CI, 0.41-0.84; P = .004). Although there was a higher rate of ICA and coronary revascularization in the CTA group than in the standard-care group in the first few months of follow-up, the overall rates were similar at 5 years, with ICA performed in 491 patients and 502 patients in the CTA vs standard-care groups, respectively (hazard ratio, 1.00; 95% CI, 0.88-1.13). Similarly, coronary revascularization was performed in 279 patients in the CTA group and in 267 patients in the standard-care group (hazard ratio, 1.07; 95% CI, 0.91-1.27). There were, however, more preventive therapies initiated in patients in the CTA group than in the standard-care group (odds ratio, 1.40; 95% CI, 1.19-1.65).
Conclusion: In patients with stable chest pain, the use of CTA in addition to standard care resulted in a significantly lower rate of death from coronary heart disease or nonfatal myocardial infarction at 5 years; the main contributor to this outcome was a reduced nonfatal myocardial infarction rate. There was no difference in the rate of coronary angiography or coronary revascularization between the 2 groups at 5 years.
Study 2 Overview (DISCHARGE Trial Group)
Objective: To compare the effectiveness of computed tomography (CT) with ICA as a diagnostic tool in patients with stable chest pain and intermediate pretest probability of coronary artery disease (CAD).
Design: Multicenter, randomized, assessor-blinded pragmatic prospective study.
Setting and participants: A total of 3667 patients with stable chest pain and intermediate pretest probability of CAD were enrolled at 26 centers and randomized into CT or ICA groups. Only 3561 patients were included in the modified intention-to-treat analysis, with 1808 patients and 1753 patients in the CT and ICA groups, respectively.
Main outcome measures: The primary outcome was a composite of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke over 3.5 years. The main secondary outcomes were major procedure-related complications and patient-reported angina pectoris during the last 4 weeks of follow up.
Main results: The primary outcome occurred in 38 of 1808 patients (2.1%) in the CT group and in 52 of 1753 patients (3.0%) in the ICA group (hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). The secondary outcomes showed that major procedure-related complications occurred in 9 patients (0.5%) in the CT group and in 33 patients (1.9%) in the ICA group (hazard ratio, 0.26; 95% CI, 0.13-0.55). Rates of patient-reported angina in the final 4 weeks of follow-up were 8.8% in the CT group and 7.5% in the ICA group (odds ratio, 1.17; 95% CI, 0.92-1.48).
Conclusion: Risk of major adverse cardiovascular events from the primary outcome were similar in both the CT and ICA groups among patients with stable chest pain and intermediate pretest probability of CAD. Patients referred for CT had a lower rate of coronary angiography leading to fewer major procedure-related complications in these patients than in those referred for ICA.
Commentary
Evaluation and treatment of obstructive atherosclerosis is an important part of clinical care in patients presenting with angina symptoms.1 Thus, the initial investigation for patients with suspected obstructive CAD includes ruling out acute coronary syndrome and assessing quality of life.1 The diagnostic test should be tailored to the pretest probability for the diagnosis of obstructive CAD.2
In the United States, stress testing traditionally has been used for the initial assessment in patients with suspected CAD,3 but recently CTA has been utilized more frequently for this purpose. Compared to a stress test, which often helps identify and assess ischemia, CTA can provide anatomical assessment, with higher sensitivity to identify CAD.4 Furthermore, it can distinguish nonobstructive plaques that can be challenging to identify with stress test alone.
Whether CTA is superior to stress testing as the initial assessment for CAD has been debated. The randomized PROMISE trial compared patients with stable angina who underwent functional stress testing or CTA as an initial strategy.5 They reported a similar outcome between the 2 groups at a median follow-up of 2 years. However, in the original SCOT-HEART trial (CT coronary angiography in patients with suspected angina due to coronary heart disease), which was published in the same year as the PROMISE trial, the patients who underwent initial assessment with CTA had a numerically lower composite end point of cardiac death and myocardial infarction at a median follow-up of 1.7 years (1.3% vs 2.0%, P = .053).6
Given this result, the SCOT-HEART investigators extended the follow-up to evaluate the composite end point of death from coronary heart disease or nonfatal myocardial infarction at 5 years.7 This trial enrolled patients who were initially referred to a cardiology clinic for evaluation of chest pain, and they were randomized to standard care plus CTA or standard care alone. At a median duration of 4.8 years, the primary outcome was lower in the CTA group (2.3%, 48 patients) than in the standard-care group (3.9%, 81 patients) (hazard ratio, 0.58; 95% CI, 0.41-0.84; P = .004). Both groups had similar rates of invasive coronary angiography and had similar coronary revascularization rates.
It is hypothesized that this lower rate of nonfatal myocardial infarction in patients with CTA plus standard care is associated with a higher rate of preventive therapies initiated in patients in the CTA-plus-standard-care group compared to standard care alone. However, the difference in the standard-care group should be noted when compared to the PROMISE trial. In the PROMISE trial, the comparator group had predominantly stress imaging (either nuclear stress test or echocardiography), while in the SCOT-HEART trial, the group had predominantly stress electrocardiogram (ECG), and only 10% of the patients underwent stress imaging. It is possible the difference seen in the rate of nonfatal myocardial infarction was due to suboptimal diagnosis of CAD with stress ECG, which has lower sensitivity compared to stress imaging.
The DISCHARGE trial investigated the effectiveness of CTA vs ICA as the initial diagnostic test in the management of patients with stable chest pain and an intermediate pretest probability of obstructive CAD.8 At 3.5 years of follow-up, the primary composite of cardiovascular death, myocardial infarction, or stroke was similar in both groups (2.1% vs 3.0; hazard ratio, 0.70; 95% CI, 0.46-1.07; P = .10). Importantly, as fewer patients underwent ICA, the risk of procedure-related complication was lower in the CTA group than in the ICA group. However, it is important to note that only 25% of the patients diagnosed with obstructive CAD had greater than 50% vessel stenosis, which raises the question of whether an initial invasive strategy is appropriate for this population.
The strengths of these 2 studies include the large number of patients enrolled along with adequate follow-up, 5 years in the SCOT-HEART trial and 3.5 years in the DISCHARGE trial. The 2 studies overall suggest the usefulness of CTA for assessment of CAD. However, the control groups were very different in these 2 trials. In the SCOT-HEART study, the comparator group was primarily assessed by stress ECG, while in the DISCHARGE study, the comparator group was primary assessed by ICA. In the PROMISE trial, the composite end point of death, myocardial infarction, hospitalization for unstable angina, or major procedural complication was similar when the strategy of initial CTA was compared to functional testing with imaging (exercise ECG, nuclear stress testing, or echocardiography).5 Thus, clinical assessment is still needed when clinicians are selecting the appropriate diagnostic test for patients with suspected CAD. The most recent guidelines give similar recommendations for CTA compared to stress imaging.9 Whether further improvement in CTA acquisition or the addition of CT fractional flow reserve can further improve outcomes requires additional study.
Applications for Clinical Practice and System Implementation
In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful in diagnosis compared to stress ECG and in reducing utilization of low-yield ICA. Whether CTA is more useful compared to the other noninvasive stress imaging modalities in this population requires further study.
Practice Points
- In patients with stable chest pain and intermediate pretest probability of CAD, CTA is useful compared to stress ECG.
- Use of CTA can potentially reduce the use of low-yield coronary angiography.
–Thai Nguyen, MD, Albert Chan, MD, Taishi Hirai, MD
University of Missouri, Columbia, MO
1. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41(3):407-477. doi:10.1093/eurheartj/ehz425
2. Nakano S, Kohsaka S, Chikamori T et al. JCS 2022 guideline focused update on diagnosis and treatment in patients with stable coronary artery disease. Circ J. 2022;86(5):882-915. doi:10.1253/circj.CJ-21-1041.
3. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012;60(24):e44-e164. doi:10.1016/j.jacc.2012.07.013
4. Arbab-Zadeh A, Di Carli MF, Cerci R, et al. Accuracy of computed tomographic angiography and single-photon emission computed tomography-acquired myocardial perfusion imaging for the diagnosis of coronary artery disease. Circ Cardiovasc Imaging. 2015;8(10):e003533. doi:10.1161/CIRCIMAGING
5. Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015;372(14):1291-300. doi:10.1056/NEJMoa1415516
6. SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet. 2015;385:2383-2391. doi:10.1016/S0140-6736(15)60291-4
7. SCOT-HEART Investigators, Newby DE, Adamson PD, et al. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med. 2018;379(10):924-933. doi:10.1056/NEJMoa1805971
8. DISCHARGE Trial Group, Maurovich-Horvat P, Bosserdt M, et al. CT or invasive coronary angiography in stable chest pain. N Engl J Med. 2022;386(17):1591-1602. doi:10.1056/NEJMoa2200963
9. Writing Committee Members, Lawton JS, Tamis-Holland JE, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
1. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41(3):407-477. doi:10.1093/eurheartj/ehz425
2. Nakano S, Kohsaka S, Chikamori T et al. JCS 2022 guideline focused update on diagnosis and treatment in patients with stable coronary artery disease. Circ J. 2022;86(5):882-915. doi:10.1253/circj.CJ-21-1041.
3. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012;60(24):e44-e164. doi:10.1016/j.jacc.2012.07.013
4. Arbab-Zadeh A, Di Carli MF, Cerci R, et al. Accuracy of computed tomographic angiography and single-photon emission computed tomography-acquired myocardial perfusion imaging for the diagnosis of coronary artery disease. Circ Cardiovasc Imaging. 2015;8(10):e003533. doi:10.1161/CIRCIMAGING
5. Douglas PS, Hoffmann U, Patel MR, et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med. 2015;372(14):1291-300. doi:10.1056/NEJMoa1415516
6. SCOT-HEART investigators. CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet. 2015;385:2383-2391. doi:10.1016/S0140-6736(15)60291-4
7. SCOT-HEART Investigators, Newby DE, Adamson PD, et al. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med. 2018;379(10):924-933. doi:10.1056/NEJMoa1805971
8. DISCHARGE Trial Group, Maurovich-Horvat P, Bosserdt M, et al. CT or invasive coronary angiography in stable chest pain. N Engl J Med. 2022;386(17):1591-1602. doi:10.1056/NEJMoa2200963
9. Writing Committee Members, Lawton JS, Tamis-Holland JE, et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2022;79(2):e21-e129. doi:10.1016/j.jacc.2021.09.006
‘Double-edged’ impact of sparring on the brains of MMA fighters
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
, early research suggests.
Investigators found sparring, defined as strategically hitting opponents with kicks, punches, and other strikes during practice sessions, is linked to increased white matter hyperintensities in the brain, pointing to possible vascular damage from repeated head trauma. However, the study results also show sparring was associated with a larger bilateral caudate which, in theory, is neuroprotective.
“From our preliminary study, sparring practice in MMA fighters may have a ‘double-edged sword’ effect on the brain,” study investigator Aaron Esagoff, a second-year medical student at Johns Hopkins University School of Medicine, Baltimore, told this news organization.
“The combination of complex movements along with constant strategy and anticipation of your opponent’s next move may provide a neuroprotective effect on the caudate,” Mr. Esagoff said. However, he added, more research is needed into understanding this particular finding.
The study results were presented at the American Psychiatric Association (APA) 2022 Annual Meeting.
Growing popularity
MMA is a full-contact combat sport that has become increasingly popular over the past 15 years. It combines techniques from boxing, wrestling, karate, judo, and jujitsu.
To prepare for fights, MMA practitioners incorporate sparring and grappling, which use techniques such as chokes and locks to submit an opponent. Head protection is sometimes incorporated during practice, but is not the norm during a fight, said Mr. Esagoff.
The study investigated sparring during practice rather than fights because, he said, MMA competitors only fight a few times a year but spend hundreds of hours training. “So the health effects of training are going to be really important,” he said.
As with other combat sports, MMA involves hits to the head. Previous research has shown repetitive head trauma can lead to neurodegenerative diseases, including chronic traumatic encephalopathy (CTE) and Alzheimer’s disease, Mr. Esagoff noted.
Previous studies have also linked more professional fights and years of fighting to a decrease in brain volume among MMA fighters, he added.
The new analysis was conducted as part of the Professional Fighters Brain Health Study, a longitudinal cohort study of MMA professional fighters. It included 92 fighters with data available on MRI and habits regarding practicing. The mean age of the participants was 30 years, 62% were White, and 85% were men.
The study examined sparring but did not include grappling because of “several challenges” with the current data analysis, Mr. Esagoff said. Researchers adjusted for age, sex, education, race, number of fights, total intracranial volume, and type of MRI scanner used.
A ‘highly strategic’ sport
Results showed a strong association between the number of sparring rounds per week and increased white matter hyperintensity volume (mcL) on MRI (P = .039).
This suggests white matter damage, possibly a result of direct neuronal injury, vascular damage, or immune modulation, said Mr. Esagoff. However, another mechanism may be involved, he added.
There was also a significant association between sparring and increased size of the caudate nucleus, an area of the brain involved in movement, learning, and memory (P = .014 for right caudate volume, P = .012 for left caudate volume).
There are some theories that might explain this finding, said Mr. Esagoff. For example, individuals who spar more may get better at avoiding impacts and injuries during a fight, which might in turn affect the size of the caudate.
The controlled movements and techniques used during sparring could also affect the caudate. “Some research has shown that behavior, learning, and/or exercise may increase the size of certain brain regions,” Mr. Esagoff said.
He noted the “highly strategic” nature of combat sports – and used the example of Brazilian jiu-jitsu. That sport “is known as human chess because it takes a thoughtful approach to defeat a larger opponent with base, leverage, and technique,” he said.
However, Mr. Esagoff stressed that while it is possible movements involved in MMA increase caudate size, this is just a theory at this point.
A study limitation was that fighters volunteered to participate and may not represent all fighters. As well, the study was cross-sectional and looked at only one point in time, so it cannot infer causation.
Overall, the new findings should help inform fighters, governing bodies, and the public about the potential risks and benefits of different styles of MMA fighting and practice, although more research is needed, said Mr. Esagoff.
He and his team now plan to conduct a longer-term study and investigate effects of grappling on brain structure and function in addition to sparring.
Jury still out
Commenting on the study, Howard Liu, MD, chair of the University of Nebraska Medical Center department of psychiatry and incoming chair of the APA’s Council on Communications, said the jury “is clearly still out” when it comes to the investigation of brain impacts.
“We don’t know quite what these changes fully correlate to,” said Dr. Liu, who moderated a press briefing highlighting the study.
He underlined the importance of protecting athletes vulnerable to head trauma, be they professionals or those involved at the youth sports level.
Dr. Liu also noted the “extreme popularity” and rapid growth of MMA around the world, which he said provides an opportunity for researchers to study these professional fighters.
“This is a unique population that signed up in the midst of hundreds of hours of sparring to advance neuroscience, and that’s quite amazing,” he said.
A version of this article first appeared on Medscape.com.
FROM APA 2022