User login
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
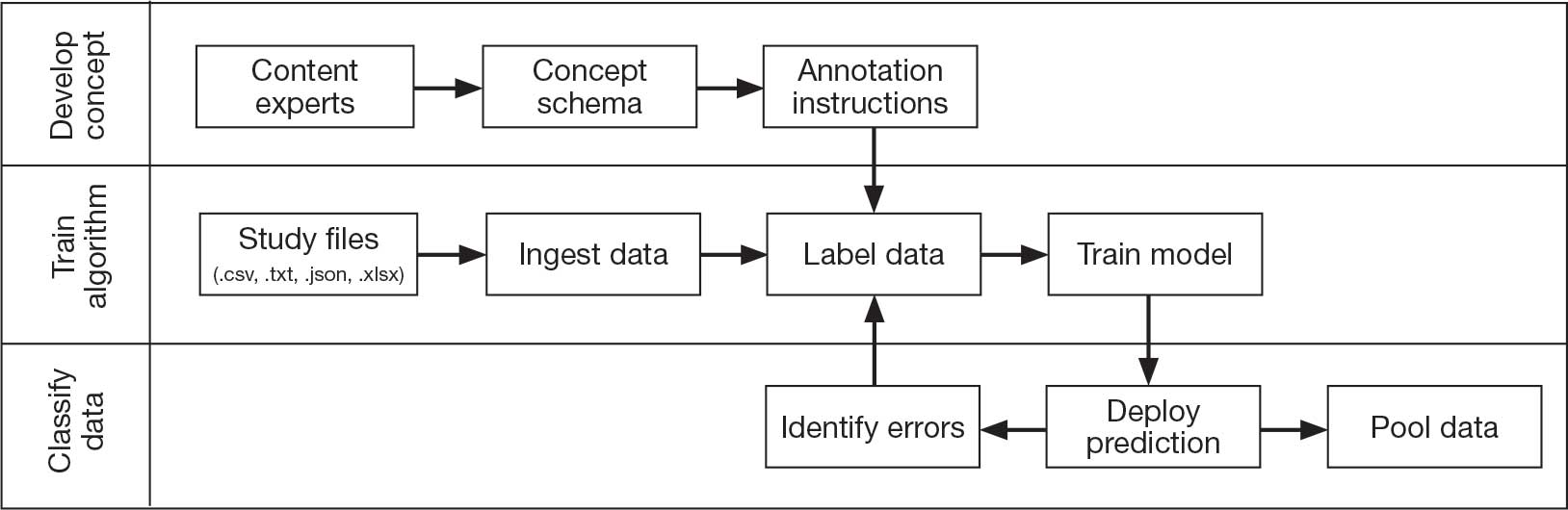
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).
Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
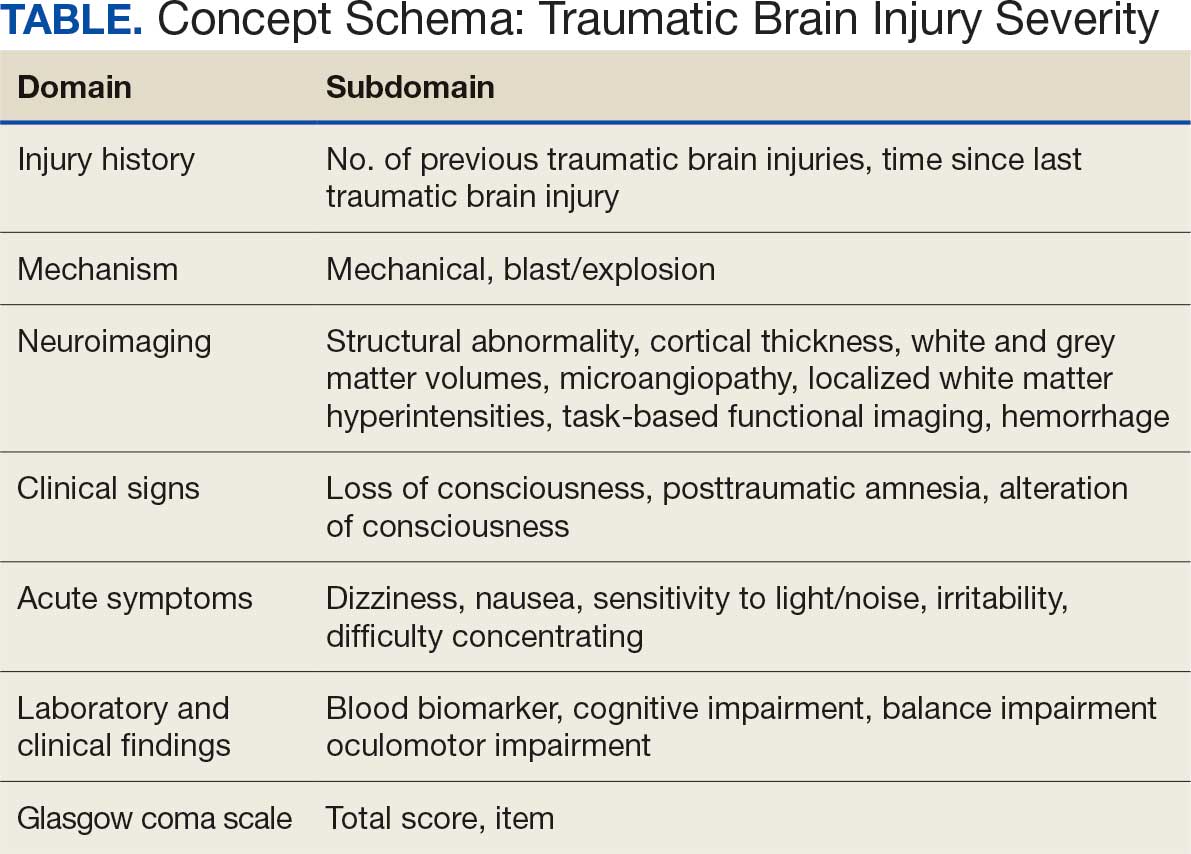
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.
Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
In leveraging existing, readily available evidence-based health care information (eg, systematic reviews, clinical practice guidelines), clinicians have historically made recommendations based on treatment responses of the average patient.1 Recently, this approach has been expanded into data-driven, evidence-based precision medical care for individuals across a wide range of disciplines and care settings. These precision medicine approaches use information related to an individual’s genes, environment, and lifestyle to tailor recommendations regarding prevention, diagnosis, and treatment.
Applying precision medicine approaches to the unique exposures and experiences of service members and veterans—particularly those who served in combat environments—through the incorporation of biopsychosocial factors into medical decision-making may be even more pertinent. This sentiment is reflected in Section 305 of the Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, which outlines the Precision Medicine Initiative of the US Department of Veterans Affairs (VA) to identify and validate brain and mental health biomarkers.2 Despite widespread consensus regarding the promise of precision medicine, large, rich datasets with elements pertaining to common military exposures such as traumatic brain injury (TBI) and posttraumatic stress disorder (PTSD) are limited.
Existing datasets, most of which are relatively small or focus on specific cohorts (eg, older veterans, transitioning veterans), continue to create barriers to advancing precision medicine. For example, in classically designed clinical trials, analyses are generally conducted in a manner that may obfuscate efficacy among subcohorts of individuals, thereby underscoring the need to explore alternative strategies to unify existing datasets capable of revealing such heterogeneity.3 The evidence base for precision medical care is limited, drawing from published trials with relatively small sample sizes and even larger cohort studies have limited biomarker data. Additionally, these models are often exploratory during development, and to avoid statistical overfitting of an exploratory model, validation in similar datasets is needed—an added burden when data sources are small or underpowered to begin with.
A promising approach is to combine and harmonize the largest, most deeply characterized data sources from similar samples. Although combining such datasets may appear to require minimal time and effort, harmonizing similar variables in an evidence-based and replicable manner requires time and expertise, even when participant characteristics and outcomes are similar.4-7
Challenges related to harmonization are related to the wide range of strategies (eg, self-report questionnaires, clinical interviews, electronic health record review) used to measure common brain and mental health constructs, such as depression. Even when similar methods (eg, self-report measures) are implemented, challenges persist. For example, if a study used a depression measure that focused primarily on cognitive symptoms (eg, pessimism, self-dislike, suicidal ideation) and another study used a depression measure composed of items more heavily weighted towards somatic symptoms (eg, insomnia, loss of appetite, weight loss, decreased libido), combining their data could be challenging, particularly if researchers, clinicians, or administrators are interested in more than dichotomous outcomes (eg, depression vs no depression).8,9
To address this knowledge gap and harmonize multimodal data from varied sources, well-planned and reproducible curation is needed. Longitudinal cohort studies of service members and veterans with military combat and training exposure histories provide researchers and other stakeholders access to extant biopsychosocial data shown to affect risk for adverse health outcomes; however, efforts to facilitate individually tailored treatment or other precision medicine approaches would benefit from the synthesis of such datasets.10
Members of the VA Total Brain Diagnostics (TBD) team are engaged in harmonizing variables from the Long-Term Impact of Military-Relevant Brain Injury Consortium–Chronic Effects of Neurotrauma Consortium (LIMBIC-CENC)11 and the Translational Research Center for TBI and Stress Disorders (TRACTS).12-21 While there is overlap across LIMBIC-CENC and TRACTS with respect to data domains, considerable data harmonization is needed to allow for future valid and meaningful analyses, particularly those involving multivariable predictors.
Data Sources
Both data sources for the TBD harmonization project, LIMBIC-CENC and TRACTS, include extensive, longitudinal data collected from relatively large cohorts of veterans and service members with combat exposure. Both studies collect detailed data related to potential brain injury history and include participants with and without a history of TBI. Similarly, both include extensive collection of fluid biomarkers and imaging data, as well as measures of biopsychosocial functioning.
Data collection sites for LIMBIC-CENC include 16 recruitment sites, 9 at VA medical centers (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego) and 7 at military treatment sites (Alexandria, San Diego, Tampa, Tacoma, Columbia, Coronado, Hinesville), in addition to 11 assessment sites (Richmond, Houston, Tampa, San Antonio, Portland, Minneapolis, Boston, Salisbury, San Diego, Alexandria, Augusta). Data for TRACTS are collected at sites in Boston and Houston.
LIMBIC-CENC is a 12-year, 17-site cohort of service members and veteran participants with combat exposure who are well characterized at baseline and undergo annual reassessments. As of December 2025, > 3100 participants have been recruited, and nearly 90% remain in follow-up. Data collection includes > 6200 annual follow-up evaluations and > 1550 5-year re-evaluations, with 400 enrolled participants followed up annually.
TRACTS is a 16-year, 2-site cohort of veterans with combat exposure who complete comprehensive assessments at enrollment, undergo annual reassessments, and complete comprehensive reassessment every 5 years thereafter. As of December 2025, > 1075 participants have completed baseline (Time 1) assessments, > 600 have completed the 2-year re-evaluation (Time 2), > 175 have completed the 5-year re-evaluation (Time 3), and > 35 have completed 10-year evaluations (Time 4), with about 50 new participants added and 100 enrolled participants followed up annually. More data on participant characteristics are available for both LIMBIC-CENC and TRACTS in previous publications.11,22These 2 ongoing, prospective, longitudinal cohorts of service members and veterans offer access to a wide range of potential risk factors that can affect response to care and outcomes, including demographics (eg, age, sex), injury characteristics (eg, pre-exposure factors, exposure factors), biomarkers (eg, serum, saliva, brain imaging, evoked potentials), and functional measures (eg, computerized posturography, computerized eye tracking, sensory testing, clinical examination, neuropsychological assessments, symptom questionnaires).
Harmonization Strategy
Pooling and harmonizing data from large studies evaluating similar participant cohorts and conditions involves numerous steps to appropriately handle a variety of measurements and disparate variable names. The TBD team adapted a model data harmonization system developed by O’Neil et al through initial work harmonizing the Federal Interagency Traumatic Brain Injury Research Informatics System (FITBIR).4-7 This process was expanded and generalized by the research team to combine data from LIMBIC-CENC and TRACTS to create a single pooled dataset for analysis (Figure).

Injury Research database.
This approach was selected because it accommodates heterogeneous study designs (eg, cross-sectional, longitudinal, case-control), data collection methods (eg, clinical assessment, self-reported, objective blood, and imaging biomarkers), and various assessments of the same construct (ie, different measures of brain injury). While exact matches for data collection methods and measures may be easily harmonized, the timing of assessment, number of assessments, assessment tool version, and other factors must be considered. The goal was to harmonize data from LIMBIC-CENC and TRACTS to allow additional data sources to be harmonized and incorporated in the future.
Original data files from each study were reshaped to represent participant-level observations with 1 unique measurement per row. The measurement represents what information was collected and the value recorded represents the unique observation. These data are linked to metadata from the original study, which includes the study’s definition of each measurement, how it was collected, and any available information regarding when it was collected in reference to study enrollment or injury. Additional information on the file source, row, and column position of each data point was added to enable recreation of the original data as needed.
The resulting dataset was used to harmonize measurements from LIMBIC-CENC and TRACTS into a priori-defined schemas for brain- and mental health-relevant concepts, including TBI severity, PTSD, substance use, depression, suicidal ideation, and functioning (including cognitive, physical, and social functioning). This process was facilitated using natural language processing (NLP). Each study uniquely defines all measurements and provides written definitions with the data. Measurement definitions serve as records describing what was collected, how it was collected, and how the study may have uniquely defined information for its purposes. For example, definitions of exposure to brain injury and severity of brain injury may differ between studies, and the study-provided definition defines these differences.
Definitions were converted into numeric vectors through sentence embedding, a process that preserves the semantic meaning of the definition.23 Cosine similarity was used as the primary metric to compare the semantic textual similarity between pairs of measurement definitions. Cosine similarity ranges from 0 to 1, where 0 indicates no meaningful similarity and 1 indicates they have identical meanings.24 This approach leverages the relationship between the definitions of each measurement provided by a study and enables quick comparison of all pairwise combinations of measurement definitions between studies.
Subsets of similar measurements across studies were organized into a priori-defined schema. Clinical experts then reviewed each schema and further refined them into domains, (eg, mechanism of injury, clinical signs, acute symptoms) and subdomains (children), such as loss of consciousness, amnesia, and alteration of consciousness. This approach allows efficient handling of 2 specific cases that commonly occur when pooling and harmonizing datasets: (1) identifying the same measurement with differing names; and (2) identifying different measurements with definitions that each relate to the same domain.
The Table provides a general example of the schema for TBI severity. This was an iterative process in which clinical experts reviewed study-defined measurement definitions to develop general harmonized domains, and NLP techniques facilitated and accelerated identification and organization of measurements within these domains.

Expected Impact
Harmonization combining LIMBIC-CENC and TRACTS datasets is ongoing. Preliminary descriptive analyses of baseline cohort data indicate that harmonization across data sources is appropriate, given the lack of significant heterogeneity across sites and studies for most domains. Work by members of the TBD team is expected to lay the foundation for the use of existing and ongoing prospective, longitudinal datasets (eg, LIMBIC-CENC, TRACTS) and linked large datasets (eg, VA Informatics and Computing Infrastructure including electronic health records, VA Million Veteran Program, DaVINCI [US Department of Defense and VA Infrastructure for Clinical Intelligence]) to generate generalizable, clinically relevant information to advance precision brain and mental health care among service members and veterans.
By enhancing existing practice, this synthesized dataset has the potential to inform tailored and personalized medicine approaches designed to meet the needs of veterans and service members. These data will serve as the starting point for multivariable models examining the intersection of physiologic, behavioral, and environmental factors. The goal of this data harmonization effort is to better elucidate how clinicians and researchers can select optimal approaches for veterans and service members with TBI histories by accounting for a comprehensive set of physiologic, behavioral, and environmental factors in an individually tailored manner. These data may further extend existing clinical practice guideline approaches, inform shared decision-making, and enhance functional outcomes beyond those currently available.
Conclusions
Individuals who have served in the military have unique biopsychosocial exposures that are associated with brain and mental health disorders. To address these needs, the nationwide TBD team has initiated the creation of a unified, longitudinal dataset that includes harmonized measures from existing LIMBIC-CENC and TRACTS protocols. Initial data harmonization efforts are required to facilitate precision prognostics, diagnostics, and tailored interventions, with the goal of improving veterans’ brain and mental health and psychosocial functioning and enabling tailored and evidence-informed, individualized clinical care.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
- The Promise of Precision Medicine. National Institutes of Health (NIH). Updated January 21, 2025. Accessed January 5, 2026. https://www.nih.gov/about-nih/nih-turning-discovery-into-health/promise-precision-medicine.
- Commander John Scott Hannon Veterans Mental Health Care Improvement Act of 2019, S 785, 116th Cong (2019-2020) Accessed January 5, 2026. https://www.congress.gov/bill/116th-congress/senate-bill/785
- Cheng C, Messerschmidt L, Bravo I, et al. A general primer for data harmonization. Sci Data. 2024;11:152. doi:10.1038/s41597-024-02956-3
- Neil M, Cameron D, Clauss K, et al. A proof-of-concept study demonstrating how FITBIR datasets can be harmonized to examine posttraumatic stress disorder-traumatic brain injury associations. J Behav Data Sci. 2024;4:45-62. doi:10.35566/jbds/oneil
- O’Neil ME, Cameron D, Krushnic D, et al. Using harmonized FITBIR datasets to examine associations between TBI history and cognitive functioning. Appl Neuropsychol Adult. doi:10.1080/23279095.2024.2401974
- O’Neil ME, Krushnic D, Clauss K, et al. Harmonizing federal interagency traumatic brain injury research data to examine depression and suicide-related outcomes. Rehabil Psychol. 2024;69:159-170. doi:10.1037/rep0000547
- O’Neil ME, Krushnic D, Walker WC, et al. Increased risk for clinically significant sleep disturbances in mild traumatic brain injury: an approach to leveraging the federal interagency traumatic brain injury research database. Brain Sci. 2024;14:921. doi:10.3390/brainsci14090921
- Uher R, Perlis RH, Placentino A, et al. Self-report and clinician-rated measures of depression severity: can one replace the other? Depress Anxiety. 2012;29:1043-1049. doi:10.1002/da.21993
- Hung CI, Weng LJ, Su YJ, et al. Depression and somatic symptoms scale: a new scale with both depression and somatic symptoms emphasized. Psychiatry Clin Neurosci. 2006;60:700-708. doi:10.1111/j.1440-1819.2006.01585.x
- Stewart IJ, Howard JT, Amuan ME, et al. Traumatic brain injury is associated with the subsequent risk of atrial fibrillation or atrial flutter. Heart Rhythm. 2025;22:661-667. doi:10.1016/j.hrthm.2024.09.019
- Cifu DX. Clinical research findings from the long-term impact of military-relevant brain injury consortium-chronic effects of neurotrauma consortium (LIMBIC-CENC) 2013-2021. Brain Inj. 2022;36:587-597.doi:10.1080/02699052.2022.2033843
- Fonda JR, Fredman L, Brogly SB, et al. Traumatic brain injury and attempted suicide among veterans of the wars in Iraq and Afghanistan. Am J Epidemiol. 2017;186:220-226. doi:10.1093/aje/kwx044
- Fortier CB, Amick MM, Kenna A, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime (BAT-L) clinical interview and the VA TBI screen. J Head Trauma Rehabil. 2015;30:E1-7. doi:10.1097/htr.0000000000000008
- Grande LJ, Robinson ME, Radigan LJ, et al. Verbal memory deficits in OEF/OIF/OND veterans exposed to blasts at close range. J Int Neuropsychol Soc. 2018;24:466-475. doi:10.1017/S1355617717001242
- Hayes JP, Logue MW, Sadeh N, et al. Mild traumatic brain injury is associated with reduced cortical thickness in those at risk for Alzheimer’s disease. Brain. 2017;140:813-825. doi:10.1093/brain/aww344
- Lippa SM, Fonda JR, Fortier CB, et al. Deployment-related psychiatric and behavioral conditions and their association with functional disability in OEF/OIF/OND veterans. J Trauma Stress. 2015;28:25-33. doi:10.1002/jts.21979
- McGlinchey RE, Milberg WP, Fonda JR, et al. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudinal prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Radigan LJ, McGlinchey RE, Milberg WP, et al. Correspondence of the Boston Assessment of Traumatic Brain Injury-Lifetime and the VA Comprehensive TBI Evaluation. J Head Trauma Rehabil. 2018;33:E51-E55. doi:10.1097/htr.0000000000000361
- Sydnor VJ, Bouix S, Pasternak O, et al. Mild traumatic brain injury impacts associations between limbic system microstructure and post-traumatic stress disorder symptomatology. Neuroimage Clin. 2020;26:102190. doi:10.1016/j.nicl.2020.102190
- Van Etten EJ, Knight AR, Colaizzi TA, et al. Peritraumatic context and long-term outcomes of concussion. JAMA Netw Open. 2025;8:e2455622. doi:10.1001/jamanetworkopen.2024.55622
- Andrews RJ, Fonda JR, Levin LK, et al. Comprehensive analysis of the predictors of neurobehavioral symptom reporting in veterans. Neurology. 2018;91:e732-e745. doi:10.1212/wnl.0000000000006034
- McGlinchey RE, Milberg WP, Fonda JR, Fortier CB. A methodology for assessing deployment trauma and its consequences in OEF/OIF/OND veterans: the TRACTS longitudional prospective cohort study. Int J Methods Psychiatr Res. 2017;26:e1556. doi:10.1002/mpr.1556
- Reimers N, Gurevych I. Sentence-BERT: Sentence embeddings using Siamese BERT-Networks. 2019. Conference on Empirical Methods in Natural Language Processing.
- Singhal A. Modern information retrieval: a brief overview. IEEE Data Eng Bull. 2001;24:34-43.
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Total Brain Diagnostics: Advancing Precision Brain and Mental Health at the Department of Veterans Affairs
Impact of Blast Exposures
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
Blast exposure has been associated with a wide range of negative outcomes, including alterations in brain structure and function, poorer cognitive functioning, and increased severity of psychiatric and health symptoms. Long-term effects also include chronic secondary downstream effects, such as neuroinflammation, neurotoxicity, cellular senescence, and neurodegeneration.
Now, a recent US Department of Veterans Affairs (VA) study of 114 post-9/11 combat veterans suggests that lifetime blast exposure severity is independently associated with accelerated epigenetic aging, even after accounting for PTSD and TBI. The field of epigenetics refers to how environment influences genes by changing the chemicals attached to them.
This cross-sectional study analyzed participants enrolled in 2 coordinated VA research protocols: the Chronic Effects of Neurotrauma Consortium Study 34 and the Post-Deployment Mental Health Study. Researchers measured biological aging using DunedinPACE, an epigenetic biomarker derived from whole-blood DNA methylation data.
Greater blast exposure severity was significantly associated with faster DunedinPACE. Mild TBI history was also independently associated with faster aging, whereas PTSD diagnosis was not. No significant interaction effects were observed. Exploratory analyses suggested that higher-intensity and more frequent blast exposures contributed to more accelerated aging.
The researchers said their findings suggest that accelerated biological aging may represent a pathway linking blast exposure to increased vulnerability for age-related disease and could inform early identification of at-risk veterans.
Preclinical work has “undeniably demonstrated that primary blast forces can directly induce neurotrauma with associated, ongoing symptoms,” according to the authors of a 2024 study. “[H]owever, these findings have not translated into clinical work.” Most human studies of blast exposure use data obtained from assessments of TBI. That approach is limited, they said, because blast exposure does not always result in symptoms of concussion or TBI, and clinical symptoms of TBI are not necessary for blast-induced neurotrauma to occur.
Moreover, understanding how and why blast exposure often results in negative consequences is still lagging, and interventions and treatments have lagged comparatively, the researchers noted. In large part, they added, this is because there is no broadly endorsed definition of blast exposure. They illustrated their point with examples of terms used in earlier research: blast TBI, primary blast TBI, pressure severity, distance from the blast, and frequency of exposure. The lack of standardized language, they suggested, “prevents synthesis of existing literature into a cohesive understanding of the field.”
Those researchers called for concerted and collaborative efforts to advance the study of blast exposure, including developing a standardized definition of blast exposure and curating an empirical literature base allowing clear comparisons of results across studies. They also urged raising awareness about blast-related negative outcomes with education at all levels: continuing education opportunities, round tables at annual conference meetings, grand rounds in hospital or academic medical center settings, and journal clubs.
The BEACON Act: Partnership, Privatization, or Both?
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Seeking to modernize treatment for traumatic brain injury (TBI), Reps. Jack Bergman (R-MI) and Sarah Elfreth (D-MD) introduced the bipartisan BEACON Act to Congress on January 9. The legislation aims to expand access to innovative, evidence-based, nonpharmacological therapies to treat TBI beyond medication-centered approaches that do not always address the long-term and individualized needs of these veterans. These current methods leave “gaps in recovery, wellness, and post-service outcomes,” Bergman and Elfreth argued.
During a March 5 House Committee on Veterans’ Affairs Subcommittee on Health hearing, discussion centered on the proposed BEACON Act, as well as the additional challenges Neurology Centers of Excellence (CoEs) face to address TBI in veterans.
The act proposes awarding $60 million in grants over 3 years to private entities for TBI treatment and research and establishing 2 US Department of Veterans Affairs (VA) grant programs. The TBI Innovation Grant Program would support clinical studies and partnerships between community health care institutions, academic institutions, and the VA. The Independent Research Grant Program would advance third-party research and “implementation of proven alternative treatments,” with oversight by an independent entity modeled after the VA National Center for PTSD.
The proposed legislation has drawn criticism. “I do not disagree that veterans may need support from several different avenues to support their recovery journeys and I don't discount the role that nonprofits and academic affiliates play in facilitating and supporting that care,” said Ranking Member Rep. Julia Brownley (D-CA) said. “However, I need to draw the line at legislation that will take money from existing VA programs and redirect it to outside organizations and providers to do essentially the very same thing VA is already doing.”
Russell Gore, MD, a neurologist and chief medical officer of Avalon Action Alliance, called VA TBI care fragmented and said the BEACON Act offers an opportunity to enhance it.
“This legislation is designed to evaluate effective treatments and leverage civilian and academic TBI expertise that is aligned with the VA’s mission,” he said. “This is not an attempt to privatize care, but to complement VA research and clinical capacity… With smart, coordinated partnerships and targeted investment, we can reach more veterans earlier, treat them more effectively.”
The VA has 5 polytrauma rehabilitation centers, 23 polytrauma network sites, and numerous clinics supporting > 110 TBI teams. It also has 42 CoEs related to neurology.
In a prepared statement, Glenn Graham, MD, PhD, retired Executive Director of the VA’s Neurology Clinical Programs representing the Association of VA Neurology Services cited the CoEs’ contribution to standardization of care. “Without systemwide coordination, practice patterns can vary. A veteran in a rural facility should receive the same standard of neurological assessment and management as a veteran treated in one of our flagship medical centers,” he said, before highlighting the capabilities of tele-neurology, electronic consultation, and remote interpretation of diagnostic studies to reduce travel burdens and promote equity in access.
Graham cautioned, though, that the CoEs face challenges with budgeting and recent VA reductions in force. The proposed legislation, Graham said, would use VA appropriations to fund extramural research and “could drain vital resources from ongoing research, training and clinical programs, diverting funds to institutions with uncertain track records and limited experience working with the veteran population.”
Several people highlighted the world-renowned research coming out of the VA, efforts that both veterans and the general public endorse.
Russell Lemle, former chief psychologist for the San Francisco VA Healthcare System and a senior policy analyst at the Veterans Healthcare Policy Institute, wrote with Jasper Craven: “The private sector has nothing commensurate with this level of care. And yet this bill would push TBI treatment out to private grantees, part of the accelerating movement to privatize the entire VA—even its signature, best-in-class programs.
“The act aims to divert resources from the VA’s world-class TBI and PTSD programs by creating a parallel treatment framework.”
Gore, however, said the Avalon Action Alliance supports a “fill-the-void” approach of “capacity augmentation, not privatization.”
“The intent is to complement VA by partnering with high-performing programs capable of delivering comprehensive assessment, interdisciplinary treatment, and structured follow-up for veterans who are not effectively reached (or not successfully retained) within traditional pathways,” he said.
Traumatic Brain Injury May Reactivate Herpes Virus Leading to Neurodegeneration
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
a new study suggested.
Using a three-dimensional (3D) human brain tissue model, researchers observed that quiescent HSV-1 can be reactivated by a mechanical jolt mimicking concussion, leading to signature markers of Alzheimer’s disease, including neuroinflammation and production of amyloid beta and phosphorylated tau (p-tau) and gliosis — a phenotype made worse by repeated head injury.
“This opens the question as to whether antiviral drugs or anti-inflammatory agents might be useful as early preventive treatments after head trauma to stop HSV-1 activation in its tracks and lower the risk of Alzheimer’s disease,” lead investigator Dana Cairns, PhD, with the Department of Biomedical Engineering at Tufts University, Medford, Massachusetts, said in a statement.
But outside experts urged caution in drawing any firm conclusions, pending further study.
The study was published online in the journal Science Signaling.
HSV-1: A Major Alzheimer’s Disease Risk Factor?
TBI is a major risk factor for Alzheimer’s disease and dementia, but the pathways in the brain leading from TBI to dementia are unknown.
HSV-1 is found in over 80% of people; varicella zoster virus (VZV) is found in about 95%. Both viruses are known to enter the brain and lay dormant in neurons and glial cells. Prior evidence indicates that HSV-1 in the brain of APOE-ε4 carriers confers a strong risk for Alzheimer’s disease.
A number of years ago, the team created a 3D model of human brain tissue to study the link between TBI, the viruses, and dementia. The model is 6 mm wide, shaped like a donut, and made of a spongy material of silk protein and collagen saturated with neural stem cells. The cells mature into neurons, communicate with each other, and form a network that mimics the brain environment.
In an earlier study using the model quiescently infected with HSV-1, Cairns and colleagues found that subsequent exposure to VZV created the inflammatory conditions that led to reactivation of HSV-1.
This led them to wonder what would happen if they subjected the brain tissue model to a physical disruption akin to a concussion. Would HSV-1 wake up and start the process of neurodegeneration?
To investigate, they examined the effects of one or more controlled blows to the 3D human brain tissue model in the absence or presence of quiescent HSV-1 infection.
After repeated, mild controlled blows, researchers found that the latently infected 3D brain tissue displayed reactivated HSV-1 and the production and accumulation of amyloid beta and p-tau — which promotes neurodegeneration. The blows also activated gliosis, which is associated with destructive neuroinflammation.
These effects are collectively associated with Alzheimer’s disease, dementia, and chronic traumatic encephalopathy, they pointed out, and were increased with additional injury but were absent in tissue not infected with HSV-1.
“These data suggest that HSV-1 in the brain is pivotal in increasing the risk of Alzheimer’s disease, as other recent studies using cerebral organoids have suggested,” the researchers wrote.
They propose that following brain injury, “whether by infection or mechanical damage, the resulting inflammation induces HSV-1 reactivation in the brain leading to the development of Alzheimer’s disease/dementia and that HSV-1 is a major cause of the disease, especially in APOE4 carriers.”
Future studies should investigate “possible ways of mitigating or stopping the damage caused by head injury, thereby reducing subsequent development of Alzheimer’s disease by implementing efforts to prevent the reactivation of virus in brain such as anti-inflammatory and/or antiviral treatment post-injury,” researchers suggested.
Outside Experts Weigh in
Several outside experts offered perspective on the study in a statement from the UK nonprofit Science Media Centre.
Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, London, England, said that, while the study is interesting, there are limitations.
“The increase in Alzheimer’s-like brain changes in these latent virus-containing cells subjected to injury does not resemble the pathology that is found in the brain of people with Alzheimer’s disease,” Spires-Jones noted.
“These experiments were also in cells grown in artificial conditions without important Alzheimer’s-related factors such as age and blood vessel changes. Finally, these experiments were repeated in a small number of experimental replicates (three times per experiment), so these results will need to be confirmed in more relevant biological systems with larger studies to be sure there is a biological link between latent herpes simplex virus type 1, brain injury, and Alzheimer’s pathology,” Spires-Jones cautioned.
Robert Howard, MD, MRCPsych, University College London (UCL) Division of Psychiatry, said the study suggests a possible mechanism for the association between HSV-1, brain injury, and Alzheimer’s disease.
“However, as so often in science, it is very important to bear in mind that association does not mean causation. Much more research will be needed before this can be seriously considered a plausible mechanism for the development of dementia,” Howard cautioned.
“Avoidance of brain injuries, such as those encountered in some contact sports, is already known to be an important way to prevent dementia, and I’m unconvinced that this reflects anything more complicated than mechanical damage causing death of brain cells,” he added.
Jennifer Pocock, PhD, with UCL Queen Square Institute of Neurology, noted the role of microglia, which are activated by mild and repetitive TBI, isn’t addressed in the study.
“This paper seems to suggest that only astrocytes contribute to the reported neuroinflammation in brain tissue. Also, the inclusion of APOE3/4 is not clearly defined. Because of this, the findings are likely to represent an over interpretation for the ‘real world’ as the inclusion of microglia may negate or accentuate them, depending on the severity of the TBI,” Pocock said.
The study was funded by the US Army Research Office and Department of Defense. The authors have declared no relevant conflicts of interest. Spires-Jones and Howard had no relevant disclosures related to this study. Pocock has received research funding from AstraZeneca and Daiichi Sankyo.
A version of this article appeared on Medscape.com.
FROM SCIENCE SIGNALING
Donepezil Shows Promise in TBI Recovery
TOPLINE:
Donepezil was associated with improved verbal memory and enhanced recall and processing speed, compared with placebo, in patients with severe traumatic brain injury (TBI), with a favorable safety profile despite mild to moderate gastrointestinal side effects.
METHODOLOGY:
- A four-site, randomized, parallel-group, double-blind, placebo-controlled, 10-week clinical trial (MEMRI-TBI-D) was conducted between 2013 and 2019 to evaluate the efficacy of donepezil for verbal memory impairments following severe TBI.
- 75 adults (75% men; mean age, 37 years) with complicated mild, moderate, or severe nonpenetrating TBI at least 6 months prior to study participation were included and randomly assigned to receive donepezil (n = 37) or placebo (n = 38).
- Participants received 5 mg donepezil daily or matching placebo for 2 weeks, then donepezil at 10 mg daily or matching placebo for 8 weeks; treatment was discontinued at 10 weeks, with an additional 4-week observation period.
- Verbal memory was assessed using the Hopkins Verbal Learning Test–Revised (HVLT-R). The primary outcome measure was verbal learning, evaluated through the HVLT-R total recall (ie, Total Trials 1-3) score.
TAKEAWAY:
- Compared with placebo, donepezil was associated with significantly greater improvements in verbal learning in both modified intent-to-treat and per-protocol analyses (P = .034 and .036, respectively).
- Treatment-responder rates were significantly higher in the donepezil group than in the placebo group (42 vs 18%; P = .03), with donepezil responders showing significant improvements in delayed recall and processing speed.
- Although there were no serious adverse events in either group, treatment-emergent adverse events were significantly more common in the donepezil group vs placebo (46% vs 8%; P < .001). No serious adverse events occurred in either group.
- Diarrhea and nausea were significantly more common in the donepezil group than in the placebo group (Fisher’s exact test: diarrhea, P = .03; nausea, P = .01).
IN PRACTICE:
“This study demonstrates the efficacy of donepezil on severe, persistent verbal memory impairments after predominantly severe TBI, with significant benefit for a subset of persons with such injuries, as well as a relatively favorable safety and tolerability profile,” the investigators wrote.
SOURCE:
The study was led by David B. Arciniegas, MD, University of Colorado School of Medicine, Aurora. It was published online in The Journal of Neuropsychiatry and Clinical Neurosciences.
LIMITATIONS:
The study included a relatively small sample with predominantly severe TBI requiring hospitalization and inpatient rehabilitation. The sample characteristics limit the generalizability of the findings to persons with other severities of TBI, other types of memory impairments, or more complex neuropsychiatric presentations. The study population had an average of 14 years of education, making generalizability to individuals with lower education levels uncertain. Additionally, while measures of information processing speed and immediate auditory attention were included, specific measures of sustained or selective attention were not, making it difficult to rule out improvements in higher-level attention as potential contributors to the observed verbal memory performance improvements.
DISCLOSURES:
The study was funded by the National Institute on Disability, Independent Living, and Rehabilitation Research, with in-kind support from TIRR Memorial Hermann. Four authors disclosed various financial and professional affiliations, including advisory roles with pharmaceutical and diagnostic companies, support from institutional awards, and involvement in programs funded by external organizations. One author served as the editor of The Journal of Neuropsychiatry and Clinical Neurosciences, with an independent editor overseeing the review and publication process for this article.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Donepezil was associated with improved verbal memory and enhanced recall and processing speed, compared with placebo, in patients with severe traumatic brain injury (TBI), with a favorable safety profile despite mild to moderate gastrointestinal side effects.
METHODOLOGY:
- A four-site, randomized, parallel-group, double-blind, placebo-controlled, 10-week clinical trial (MEMRI-TBI-D) was conducted between 2013 and 2019 to evaluate the efficacy of donepezil for verbal memory impairments following severe TBI.
- 75 adults (75% men; mean age, 37 years) with complicated mild, moderate, or severe nonpenetrating TBI at least 6 months prior to study participation were included and randomly assigned to receive donepezil (n = 37) or placebo (n = 38).
- Participants received 5 mg donepezil daily or matching placebo for 2 weeks, then donepezil at 10 mg daily or matching placebo for 8 weeks; treatment was discontinued at 10 weeks, with an additional 4-week observation period.
- Verbal memory was assessed using the Hopkins Verbal Learning Test–Revised (HVLT-R). The primary outcome measure was verbal learning, evaluated through the HVLT-R total recall (ie, Total Trials 1-3) score.
TAKEAWAY:
- Compared with placebo, donepezil was associated with significantly greater improvements in verbal learning in both modified intent-to-treat and per-protocol analyses (P = .034 and .036, respectively).
- Treatment-responder rates were significantly higher in the donepezil group than in the placebo group (42 vs 18%; P = .03), with donepezil responders showing significant improvements in delayed recall and processing speed.
- Although there were no serious adverse events in either group, treatment-emergent adverse events were significantly more common in the donepezil group vs placebo (46% vs 8%; P < .001). No serious adverse events occurred in either group.
- Diarrhea and nausea were significantly more common in the donepezil group than in the placebo group (Fisher’s exact test: diarrhea, P = .03; nausea, P = .01).
IN PRACTICE:
“This study demonstrates the efficacy of donepezil on severe, persistent verbal memory impairments after predominantly severe TBI, with significant benefit for a subset of persons with such injuries, as well as a relatively favorable safety and tolerability profile,” the investigators wrote.
SOURCE:
The study was led by David B. Arciniegas, MD, University of Colorado School of Medicine, Aurora. It was published online in The Journal of Neuropsychiatry and Clinical Neurosciences.
LIMITATIONS:
The study included a relatively small sample with predominantly severe TBI requiring hospitalization and inpatient rehabilitation. The sample characteristics limit the generalizability of the findings to persons with other severities of TBI, other types of memory impairments, or more complex neuropsychiatric presentations. The study population had an average of 14 years of education, making generalizability to individuals with lower education levels uncertain. Additionally, while measures of information processing speed and immediate auditory attention were included, specific measures of sustained or selective attention were not, making it difficult to rule out improvements in higher-level attention as potential contributors to the observed verbal memory performance improvements.
DISCLOSURES:
The study was funded by the National Institute on Disability, Independent Living, and Rehabilitation Research, with in-kind support from TIRR Memorial Hermann. Four authors disclosed various financial and professional affiliations, including advisory roles with pharmaceutical and diagnostic companies, support from institutional awards, and involvement in programs funded by external organizations. One author served as the editor of The Journal of Neuropsychiatry and Clinical Neurosciences, with an independent editor overseeing the review and publication process for this article.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
Donepezil was associated with improved verbal memory and enhanced recall and processing speed, compared with placebo, in patients with severe traumatic brain injury (TBI), with a favorable safety profile despite mild to moderate gastrointestinal side effects.
METHODOLOGY:
- A four-site, randomized, parallel-group, double-blind, placebo-controlled, 10-week clinical trial (MEMRI-TBI-D) was conducted between 2013 and 2019 to evaluate the efficacy of donepezil for verbal memory impairments following severe TBI.
- 75 adults (75% men; mean age, 37 years) with complicated mild, moderate, or severe nonpenetrating TBI at least 6 months prior to study participation were included and randomly assigned to receive donepezil (n = 37) or placebo (n = 38).
- Participants received 5 mg donepezil daily or matching placebo for 2 weeks, then donepezil at 10 mg daily or matching placebo for 8 weeks; treatment was discontinued at 10 weeks, with an additional 4-week observation period.
- Verbal memory was assessed using the Hopkins Verbal Learning Test–Revised (HVLT-R). The primary outcome measure was verbal learning, evaluated through the HVLT-R total recall (ie, Total Trials 1-3) score.
TAKEAWAY:
- Compared with placebo, donepezil was associated with significantly greater improvements in verbal learning in both modified intent-to-treat and per-protocol analyses (P = .034 and .036, respectively).
- Treatment-responder rates were significantly higher in the donepezil group than in the placebo group (42 vs 18%; P = .03), with donepezil responders showing significant improvements in delayed recall and processing speed.
- Although there were no serious adverse events in either group, treatment-emergent adverse events were significantly more common in the donepezil group vs placebo (46% vs 8%; P < .001). No serious adverse events occurred in either group.
- Diarrhea and nausea were significantly more common in the donepezil group than in the placebo group (Fisher’s exact test: diarrhea, P = .03; nausea, P = .01).
IN PRACTICE:
“This study demonstrates the efficacy of donepezil on severe, persistent verbal memory impairments after predominantly severe TBI, with significant benefit for a subset of persons with such injuries, as well as a relatively favorable safety and tolerability profile,” the investigators wrote.
SOURCE:
The study was led by David B. Arciniegas, MD, University of Colorado School of Medicine, Aurora. It was published online in The Journal of Neuropsychiatry and Clinical Neurosciences.
LIMITATIONS:
The study included a relatively small sample with predominantly severe TBI requiring hospitalization and inpatient rehabilitation. The sample characteristics limit the generalizability of the findings to persons with other severities of TBI, other types of memory impairments, or more complex neuropsychiatric presentations. The study population had an average of 14 years of education, making generalizability to individuals with lower education levels uncertain. Additionally, while measures of information processing speed and immediate auditory attention were included, specific measures of sustained or selective attention were not, making it difficult to rule out improvements in higher-level attention as potential contributors to the observed verbal memory performance improvements.
DISCLOSURES:
The study was funded by the National Institute on Disability, Independent Living, and Rehabilitation Research, with in-kind support from TIRR Memorial Hermann. Four authors disclosed various financial and professional affiliations, including advisory roles with pharmaceutical and diagnostic companies, support from institutional awards, and involvement in programs funded by external organizations. One author served as the editor of The Journal of Neuropsychiatry and Clinical Neurosciences, with an independent editor overseeing the review and publication process for this article.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
A History of Concussion Linked to Maternal Mental Illness
A history of concussion can have serious long-term mental health implications for women, even years after giving birth, according to a new study.
Researchers looked at all people who delivered babies in Ontario, Canada, and found that those with a predelivery history of concussion were 25% more likely to have a serious mental illness up to 14 years after giving birth than those with no history of concussion.
The findings indicate the need for early identification and screening of women with a history of concussion, as well as ongoing, long-term supports to prevent adverse psychiatric outcomes, wrote the authors.
“I played a lot of sports growing up, and I definitely would not have thought about how a concussion could affect childbearing or parenting,” author Samantha Krueger, RM, MSc, told this news organization. She completed the research as part of her studies at the University of Toronto, Ontario.
The data were published on November 4 in The Journal of Clinical Psychiatry.
Implications for Prevention
“Birthing people, and women in general, are an often-overlooked population in the scientific literature on traumatic brain injury, including concussion. There is a potential interplay between concussion history and the challenges of being a new parent (such as labor and birth, lack of sleep, and increased noise) that make this an important population to study,” said Krueger.
The researchers conducted a population-based cohort study of all women who gave birth in Ontario between 2007 and 2017. Follow-up continued until 2021. The primary outcome was severe maternal mental illness, which was defined as a psychiatric emergency department visit, psychiatric hospital admission, or self-harm or suicide in the 14 years after delivery.
The researchers identified 18,064 women with a predelivery history of concussion and 736,689 women without a history of concussion during the study period. Women with a predelivery history of concussion were more likely than those without such a history to live in a rural area and have a history of assault or mental illness.
Overall, 11.3% (n = 2033) of the women with a predelivery history of concussion developed severe maternal mental illness (14.7 per 1000 person-years), compared with 6.8% (n = 49,928) of the women without a predelivery history of concussion (7.9 per 1000 person-years).
The adjusted hazard ratio (aHR) was 1.25. The association was strongest in women who had a predelivery history of concussion but no history of mental illness (aHR, 1.33).
“We hope to increase awareness of the seriousness of having a concussion, even when it is considered a mild head injury,” Krueger said. “The results have important implications for concussion prevention measures for young people and for the provision of postpartum supports (such as mental health and other social supports like sleep relief) to mitigate the risk of serious mental illness outcomes in birthing people with a history of concussion.”
Healthcare providers, including maternity care providers, should be asking about concussion history and providing mental health screening and supports to clients and their families to detect mental illness before a serious outcome occurs, Krueger added.
“Maternity care providers can help birthing people and their families set up supports for after the baby is born and teach families about mental health symptoms to look out for. It’s also important that providers be certain that their care is trauma informed to avoid triggering a trauma response when providing care,” she said.
Area of Concern
“This research is novel and highlights an area of major concern,” Simon Sherry, PhD, professor of psychology and neuroscience at Dalhousie University in Halifax, Nova Scotia, Canada, told this news organization. Sherry did not participate in the study.
“Postpartum depression occurs in approximately 10%-25% of mothers, but it is likely that many more cases go undiagnosed. It is attributed to hormonal changes, genetic predisposition, and environmental factors, and while previous depression or mental illness is frequently considered a risk factor, traumatic brain injuries or concussions usually are not,” Sherry said.
“Mothers are already an at-risk population for mental illness, as illustrated by the high rates of postpartum depression, and so are people with a history of concussion or traumatic brain injury. What sets this study apart is that it shows the heightened risk for women with the combination of those two distinct risk factors. Identifying these risk factors is essential to providing preventive care. If care providers know a patient is at increased risk when starting a pregnancy, then they will likely catch warning signs earlier,” he said.
“Additionally, as the article suggests, maternal mental health often is not studied beyond the first postpartum year,” Sherry said.
“Mental health struggles during the first postpartum year have largely been normalized as part of the transition into parenthood, but mental health issues among parents later in life are less accepted. After birth, so much emphasis is moved from the parent to the child. Parents rightly prioritize their children, but our job as care providers is to ensure we are also prioritizing them. The prolonged period of this study helps illustrate how important the practice of prioritizing mothers’ mental health is,” he added.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. The Canadian Institutes of Health Research also supported the study. Krueger is supported by a Canadian Institutes of Health Research Canada Graduate Scholarship Masters Award. Sherry reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A history of concussion can have serious long-term mental health implications for women, even years after giving birth, according to a new study.
Researchers looked at all people who delivered babies in Ontario, Canada, and found that those with a predelivery history of concussion were 25% more likely to have a serious mental illness up to 14 years after giving birth than those with no history of concussion.
The findings indicate the need for early identification and screening of women with a history of concussion, as well as ongoing, long-term supports to prevent adverse psychiatric outcomes, wrote the authors.
“I played a lot of sports growing up, and I definitely would not have thought about how a concussion could affect childbearing or parenting,” author Samantha Krueger, RM, MSc, told this news organization. She completed the research as part of her studies at the University of Toronto, Ontario.
The data were published on November 4 in The Journal of Clinical Psychiatry.
Implications for Prevention
“Birthing people, and women in general, are an often-overlooked population in the scientific literature on traumatic brain injury, including concussion. There is a potential interplay between concussion history and the challenges of being a new parent (such as labor and birth, lack of sleep, and increased noise) that make this an important population to study,” said Krueger.
The researchers conducted a population-based cohort study of all women who gave birth in Ontario between 2007 and 2017. Follow-up continued until 2021. The primary outcome was severe maternal mental illness, which was defined as a psychiatric emergency department visit, psychiatric hospital admission, or self-harm or suicide in the 14 years after delivery.
The researchers identified 18,064 women with a predelivery history of concussion and 736,689 women without a history of concussion during the study period. Women with a predelivery history of concussion were more likely than those without such a history to live in a rural area and have a history of assault or mental illness.
Overall, 11.3% (n = 2033) of the women with a predelivery history of concussion developed severe maternal mental illness (14.7 per 1000 person-years), compared with 6.8% (n = 49,928) of the women without a predelivery history of concussion (7.9 per 1000 person-years).
The adjusted hazard ratio (aHR) was 1.25. The association was strongest in women who had a predelivery history of concussion but no history of mental illness (aHR, 1.33).
“We hope to increase awareness of the seriousness of having a concussion, even when it is considered a mild head injury,” Krueger said. “The results have important implications for concussion prevention measures for young people and for the provision of postpartum supports (such as mental health and other social supports like sleep relief) to mitigate the risk of serious mental illness outcomes in birthing people with a history of concussion.”
Healthcare providers, including maternity care providers, should be asking about concussion history and providing mental health screening and supports to clients and their families to detect mental illness before a serious outcome occurs, Krueger added.
“Maternity care providers can help birthing people and their families set up supports for after the baby is born and teach families about mental health symptoms to look out for. It’s also important that providers be certain that their care is trauma informed to avoid triggering a trauma response when providing care,” she said.
Area of Concern
“This research is novel and highlights an area of major concern,” Simon Sherry, PhD, professor of psychology and neuroscience at Dalhousie University in Halifax, Nova Scotia, Canada, told this news organization. Sherry did not participate in the study.
“Postpartum depression occurs in approximately 10%-25% of mothers, but it is likely that many more cases go undiagnosed. It is attributed to hormonal changes, genetic predisposition, and environmental factors, and while previous depression or mental illness is frequently considered a risk factor, traumatic brain injuries or concussions usually are not,” Sherry said.
“Mothers are already an at-risk population for mental illness, as illustrated by the high rates of postpartum depression, and so are people with a history of concussion or traumatic brain injury. What sets this study apart is that it shows the heightened risk for women with the combination of those two distinct risk factors. Identifying these risk factors is essential to providing preventive care. If care providers know a patient is at increased risk when starting a pregnancy, then they will likely catch warning signs earlier,” he said.
“Additionally, as the article suggests, maternal mental health often is not studied beyond the first postpartum year,” Sherry said.
“Mental health struggles during the first postpartum year have largely been normalized as part of the transition into parenthood, but mental health issues among parents later in life are less accepted. After birth, so much emphasis is moved from the parent to the child. Parents rightly prioritize their children, but our job as care providers is to ensure we are also prioritizing them. The prolonged period of this study helps illustrate how important the practice of prioritizing mothers’ mental health is,” he added.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. The Canadian Institutes of Health Research also supported the study. Krueger is supported by a Canadian Institutes of Health Research Canada Graduate Scholarship Masters Award. Sherry reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A history of concussion can have serious long-term mental health implications for women, even years after giving birth, according to a new study.
Researchers looked at all people who delivered babies in Ontario, Canada, and found that those with a predelivery history of concussion were 25% more likely to have a serious mental illness up to 14 years after giving birth than those with no history of concussion.
The findings indicate the need for early identification and screening of women with a history of concussion, as well as ongoing, long-term supports to prevent adverse psychiatric outcomes, wrote the authors.
“I played a lot of sports growing up, and I definitely would not have thought about how a concussion could affect childbearing or parenting,” author Samantha Krueger, RM, MSc, told this news organization. She completed the research as part of her studies at the University of Toronto, Ontario.
The data were published on November 4 in The Journal of Clinical Psychiatry.
Implications for Prevention
“Birthing people, and women in general, are an often-overlooked population in the scientific literature on traumatic brain injury, including concussion. There is a potential interplay between concussion history and the challenges of being a new parent (such as labor and birth, lack of sleep, and increased noise) that make this an important population to study,” said Krueger.
The researchers conducted a population-based cohort study of all women who gave birth in Ontario between 2007 and 2017. Follow-up continued until 2021. The primary outcome was severe maternal mental illness, which was defined as a psychiatric emergency department visit, psychiatric hospital admission, or self-harm or suicide in the 14 years after delivery.
The researchers identified 18,064 women with a predelivery history of concussion and 736,689 women without a history of concussion during the study period. Women with a predelivery history of concussion were more likely than those without such a history to live in a rural area and have a history of assault or mental illness.
Overall, 11.3% (n = 2033) of the women with a predelivery history of concussion developed severe maternal mental illness (14.7 per 1000 person-years), compared with 6.8% (n = 49,928) of the women without a predelivery history of concussion (7.9 per 1000 person-years).
The adjusted hazard ratio (aHR) was 1.25. The association was strongest in women who had a predelivery history of concussion but no history of mental illness (aHR, 1.33).
“We hope to increase awareness of the seriousness of having a concussion, even when it is considered a mild head injury,” Krueger said. “The results have important implications for concussion prevention measures for young people and for the provision of postpartum supports (such as mental health and other social supports like sleep relief) to mitigate the risk of serious mental illness outcomes in birthing people with a history of concussion.”
Healthcare providers, including maternity care providers, should be asking about concussion history and providing mental health screening and supports to clients and their families to detect mental illness before a serious outcome occurs, Krueger added.
“Maternity care providers can help birthing people and their families set up supports for after the baby is born and teach families about mental health symptoms to look out for. It’s also important that providers be certain that their care is trauma informed to avoid triggering a trauma response when providing care,” she said.
Area of Concern
“This research is novel and highlights an area of major concern,” Simon Sherry, PhD, professor of psychology and neuroscience at Dalhousie University in Halifax, Nova Scotia, Canada, told this news organization. Sherry did not participate in the study.
“Postpartum depression occurs in approximately 10%-25% of mothers, but it is likely that many more cases go undiagnosed. It is attributed to hormonal changes, genetic predisposition, and environmental factors, and while previous depression or mental illness is frequently considered a risk factor, traumatic brain injuries or concussions usually are not,” Sherry said.
“Mothers are already an at-risk population for mental illness, as illustrated by the high rates of postpartum depression, and so are people with a history of concussion or traumatic brain injury. What sets this study apart is that it shows the heightened risk for women with the combination of those two distinct risk factors. Identifying these risk factors is essential to providing preventive care. If care providers know a patient is at increased risk when starting a pregnancy, then they will likely catch warning signs earlier,” he said.
“Additionally, as the article suggests, maternal mental health often is not studied beyond the first postpartum year,” Sherry said.
“Mental health struggles during the first postpartum year have largely been normalized as part of the transition into parenthood, but mental health issues among parents later in life are less accepted. After birth, so much emphasis is moved from the parent to the child. Parents rightly prioritize their children, but our job as care providers is to ensure we are also prioritizing them. The prolonged period of this study helps illustrate how important the practice of prioritizing mothers’ mental health is,” he added.
The study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. The Canadian Institutes of Health Research also supported the study. Krueger is supported by a Canadian Institutes of Health Research Canada Graduate Scholarship Masters Award. Sherry reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
A New, Easily Identifiable Sign of Concussion?
Spontaneous Headshake After a Kinematic Event (SHAAKE) refers to the rapid, back-and-forth head movement athletes exhibit following a blow to the head. This voluntary motion typically occurs within seconds to minutes after impact and is a familiar response in athletes.
In a recent survey, 7 out of 10 adult athletes recalled making this movement after a collision, and three out of four times they attributed this back-and-forth head movement to a concussion. The association was strongest among football players, who reported that over 90% of SHAAKE episodes were associated with a concussion.
The results were published online in Diagnostics.
Call to Action
“Everyone” — including sports and medical organizations — “should be adding this to their list of potential concussion signs and their protocol immediately,” study investigator Chris Nowinski, PhD, CEO and co-founder of the Concussion Legacy Foundation, told this news organization.
Nowinski said it’s “fascinating” that this concussion sign hasn’t been formally studied or added to formal concussion screening metrics before now, given that it’s been depicted in movies, television, and cartoons for decades.
Coaches, medical professionals, and concussion spotters should be trained to recognize when a SHAAKE happens, he said.
“The interesting thing is, I don’t think coaches or parents need much training other than to officially tie this to suspicion of a concussion,” Nowinski added.
The Case of Miami Dolphins QB Tua Tagovailoa
Nowinski said he was tipped off to SHAAKE as a concussion sign after Miami Dolphins quarterback Tua Tagovailoa’s controversial undiagnosed concussion during a National Football League (NFL) game in 2022.
After Tagovailoa’s head hit the ground, he rapidly shook his head side to side, indicating displaying SHAAKE, before stumbling and collapsing. At the time, a sideline doctor attributed his collapse to a prior back injury.
If Tagovailoa had been diagnosed with a concussion, he likely would not have been playing in a game just 4 days later, where he lost consciousness after suffering a suspected second concussion and was removed from the field on a stretcher.
For the survey, Nowinski and colleagues showed 347 current and former athletes, including 109 football players, video examples of SHAAKE and them asked about their experiences with this potential indicator of concussion.
Nearly 69% of athletes reported exhibiting a SHAAKE during their career, and 93% of those reported a SHAAKE in association with concussion at least once. Athletes reported SHAAKE a median of five times in their lives.
Of the athletes who reported SHAAKE, 85% linked this head-shaking movement to concussion symptoms such as disorientation (71%) and dizziness (54%).
Across all sports, SHAAKE showed a sensitivity of 49.6% and a positive predictive value (PPV) of 72.4% for diagnosing concussions.
Among football players, sensitivity improved to 52.3%, with an estimated specificity of 99.9%, a PPV of 91.9%, and an estimated negative predictive value of 99.5%.
The main limitation of the survey was the potential for recall bias due to survey participants self-reporting prior concussions. The researchers called for future prospective studies to validate SHAAKE as a sign of concussion.
Instant Replay for Brain Injury?
Experts echoed the need for validation. SHAAKE represents a “promising advance” in objective TBI assessment, particularly for sideline evaluation, said Shaheen Lakhan, MD, PhD, neurologist, and researcher based in Miami, Florida, who wasn’t involved in the research.
The potential value of SHAAKE is “particularly notable given the well-documented tendency for athletes to minimize or conceal symptoms to maintain play eligibility, a limitation that has historically challenged our reliance on subjective reporting and observational assessments,” Lakhan said.
“Moving forward, validation through prospective studies incorporating real-time video analysis, helmet sensor data, and clinician-confirmed TBI diagnoses will be essential. With appropriate validation, SHAAKE could emerge as a valuable component of our sideline assessment arsenal, complementing rather than replacing existing diagnostic approaches,” Lakhan said.
“SHAAKE could be the ‘instant replay’ for brain injuries that sports medicine has been waiting for — but like any new technology, we need to make sure it works for every player, not just some,” Lakhan added.
Also weighing in, Richard Figler, MD, director of the Concussion Center, Cleveland Clinic Sports Medicine Center, Cleveland, cautioned that the survey participants were recruited from a concussion registry and self-reported an average of 23 concussions — more than one third of which happened 5-10 years prior — which begs the question, “How much are they actually remembering?”
“Our goal is to make sure that the athletes are safe and that we’re not missing concussions, and we don’t have great tools to start off with. This study opens up the door for some prospective studies [of SHAAKE] moving forward. I think we need more data before this should be listed as a definitive marker,” said Figler, who also wasn’t involved in the study.
In any case, he said, when it comes to suspected concussion in sports, “when in doubt, you sit them out,” Figler said.
This research received no external funding. Nowinski has received travel reimbursement from the NFL Players Association (NFLPA), NFL, World Rugby, WWE, and All Elite Wrestling; served as an expert witness in cases related to concussion and chronic traumatic encephalopathy; and is compensated for speaking appearances and serving on the NFL Concussion Settlement Player Advocacy Committee. Daniel H. Daneshvar served as an expert witness in legal cases involving brain injury and concussion and received funding from the Football Players Health Study at Harvard University, which is funded by the NFLPA and evaluates patients for the MGH Brain and Body TRUST Center, sponsored in part by the NFLPA. Lakhan and Figler had no relevant disclosures.
A version of this article appeared on Medscape.com.
Spontaneous Headshake After a Kinematic Event (SHAAKE) refers to the rapid, back-and-forth head movement athletes exhibit following a blow to the head. This voluntary motion typically occurs within seconds to minutes after impact and is a familiar response in athletes.
In a recent survey, 7 out of 10 adult athletes recalled making this movement after a collision, and three out of four times they attributed this back-and-forth head movement to a concussion. The association was strongest among football players, who reported that over 90% of SHAAKE episodes were associated with a concussion.
The results were published online in Diagnostics.
Call to Action
“Everyone” — including sports and medical organizations — “should be adding this to their list of potential concussion signs and their protocol immediately,” study investigator Chris Nowinski, PhD, CEO and co-founder of the Concussion Legacy Foundation, told this news organization.
Nowinski said it’s “fascinating” that this concussion sign hasn’t been formally studied or added to formal concussion screening metrics before now, given that it’s been depicted in movies, television, and cartoons for decades.
Coaches, medical professionals, and concussion spotters should be trained to recognize when a SHAAKE happens, he said.
“The interesting thing is, I don’t think coaches or parents need much training other than to officially tie this to suspicion of a concussion,” Nowinski added.
The Case of Miami Dolphins QB Tua Tagovailoa
Nowinski said he was tipped off to SHAAKE as a concussion sign after Miami Dolphins quarterback Tua Tagovailoa’s controversial undiagnosed concussion during a National Football League (NFL) game in 2022.
After Tagovailoa’s head hit the ground, he rapidly shook his head side to side, indicating displaying SHAAKE, before stumbling and collapsing. At the time, a sideline doctor attributed his collapse to a prior back injury.
If Tagovailoa had been diagnosed with a concussion, he likely would not have been playing in a game just 4 days later, where he lost consciousness after suffering a suspected second concussion and was removed from the field on a stretcher.
For the survey, Nowinski and colleagues showed 347 current and former athletes, including 109 football players, video examples of SHAAKE and them asked about their experiences with this potential indicator of concussion.
Nearly 69% of athletes reported exhibiting a SHAAKE during their career, and 93% of those reported a SHAAKE in association with concussion at least once. Athletes reported SHAAKE a median of five times in their lives.
Of the athletes who reported SHAAKE, 85% linked this head-shaking movement to concussion symptoms such as disorientation (71%) and dizziness (54%).
Across all sports, SHAAKE showed a sensitivity of 49.6% and a positive predictive value (PPV) of 72.4% for diagnosing concussions.
Among football players, sensitivity improved to 52.3%, with an estimated specificity of 99.9%, a PPV of 91.9%, and an estimated negative predictive value of 99.5%.
The main limitation of the survey was the potential for recall bias due to survey participants self-reporting prior concussions. The researchers called for future prospective studies to validate SHAAKE as a sign of concussion.
Instant Replay for Brain Injury?
Experts echoed the need for validation. SHAAKE represents a “promising advance” in objective TBI assessment, particularly for sideline evaluation, said Shaheen Lakhan, MD, PhD, neurologist, and researcher based in Miami, Florida, who wasn’t involved in the research.
The potential value of SHAAKE is “particularly notable given the well-documented tendency for athletes to minimize or conceal symptoms to maintain play eligibility, a limitation that has historically challenged our reliance on subjective reporting and observational assessments,” Lakhan said.
“Moving forward, validation through prospective studies incorporating real-time video analysis, helmet sensor data, and clinician-confirmed TBI diagnoses will be essential. With appropriate validation, SHAAKE could emerge as a valuable component of our sideline assessment arsenal, complementing rather than replacing existing diagnostic approaches,” Lakhan said.
“SHAAKE could be the ‘instant replay’ for brain injuries that sports medicine has been waiting for — but like any new technology, we need to make sure it works for every player, not just some,” Lakhan added.
Also weighing in, Richard Figler, MD, director of the Concussion Center, Cleveland Clinic Sports Medicine Center, Cleveland, cautioned that the survey participants were recruited from a concussion registry and self-reported an average of 23 concussions — more than one third of which happened 5-10 years prior — which begs the question, “How much are they actually remembering?”
“Our goal is to make sure that the athletes are safe and that we’re not missing concussions, and we don’t have great tools to start off with. This study opens up the door for some prospective studies [of SHAAKE] moving forward. I think we need more data before this should be listed as a definitive marker,” said Figler, who also wasn’t involved in the study.
In any case, he said, when it comes to suspected concussion in sports, “when in doubt, you sit them out,” Figler said.
This research received no external funding. Nowinski has received travel reimbursement from the NFL Players Association (NFLPA), NFL, World Rugby, WWE, and All Elite Wrestling; served as an expert witness in cases related to concussion and chronic traumatic encephalopathy; and is compensated for speaking appearances and serving on the NFL Concussion Settlement Player Advocacy Committee. Daniel H. Daneshvar served as an expert witness in legal cases involving brain injury and concussion and received funding from the Football Players Health Study at Harvard University, which is funded by the NFLPA and evaluates patients for the MGH Brain and Body TRUST Center, sponsored in part by the NFLPA. Lakhan and Figler had no relevant disclosures.
A version of this article appeared on Medscape.com.
Spontaneous Headshake After a Kinematic Event (SHAAKE) refers to the rapid, back-and-forth head movement athletes exhibit following a blow to the head. This voluntary motion typically occurs within seconds to minutes after impact and is a familiar response in athletes.
In a recent survey, 7 out of 10 adult athletes recalled making this movement after a collision, and three out of four times they attributed this back-and-forth head movement to a concussion. The association was strongest among football players, who reported that over 90% of SHAAKE episodes were associated with a concussion.
The results were published online in Diagnostics.
Call to Action
“Everyone” — including sports and medical organizations — “should be adding this to their list of potential concussion signs and their protocol immediately,” study investigator Chris Nowinski, PhD, CEO and co-founder of the Concussion Legacy Foundation, told this news organization.
Nowinski said it’s “fascinating” that this concussion sign hasn’t been formally studied or added to formal concussion screening metrics before now, given that it’s been depicted in movies, television, and cartoons for decades.
Coaches, medical professionals, and concussion spotters should be trained to recognize when a SHAAKE happens, he said.
“The interesting thing is, I don’t think coaches or parents need much training other than to officially tie this to suspicion of a concussion,” Nowinski added.
The Case of Miami Dolphins QB Tua Tagovailoa
Nowinski said he was tipped off to SHAAKE as a concussion sign after Miami Dolphins quarterback Tua Tagovailoa’s controversial undiagnosed concussion during a National Football League (NFL) game in 2022.
After Tagovailoa’s head hit the ground, he rapidly shook his head side to side, indicating displaying SHAAKE, before stumbling and collapsing. At the time, a sideline doctor attributed his collapse to a prior back injury.
If Tagovailoa had been diagnosed with a concussion, he likely would not have been playing in a game just 4 days later, where he lost consciousness after suffering a suspected second concussion and was removed from the field on a stretcher.
For the survey, Nowinski and colleagues showed 347 current and former athletes, including 109 football players, video examples of SHAAKE and them asked about their experiences with this potential indicator of concussion.
Nearly 69% of athletes reported exhibiting a SHAAKE during their career, and 93% of those reported a SHAAKE in association with concussion at least once. Athletes reported SHAAKE a median of five times in their lives.
Of the athletes who reported SHAAKE, 85% linked this head-shaking movement to concussion symptoms such as disorientation (71%) and dizziness (54%).
Across all sports, SHAAKE showed a sensitivity of 49.6% and a positive predictive value (PPV) of 72.4% for diagnosing concussions.
Among football players, sensitivity improved to 52.3%, with an estimated specificity of 99.9%, a PPV of 91.9%, and an estimated negative predictive value of 99.5%.
The main limitation of the survey was the potential for recall bias due to survey participants self-reporting prior concussions. The researchers called for future prospective studies to validate SHAAKE as a sign of concussion.
Instant Replay for Brain Injury?
Experts echoed the need for validation. SHAAKE represents a “promising advance” in objective TBI assessment, particularly for sideline evaluation, said Shaheen Lakhan, MD, PhD, neurologist, and researcher based in Miami, Florida, who wasn’t involved in the research.
The potential value of SHAAKE is “particularly notable given the well-documented tendency for athletes to minimize or conceal symptoms to maintain play eligibility, a limitation that has historically challenged our reliance on subjective reporting and observational assessments,” Lakhan said.
“Moving forward, validation through prospective studies incorporating real-time video analysis, helmet sensor data, and clinician-confirmed TBI diagnoses will be essential. With appropriate validation, SHAAKE could emerge as a valuable component of our sideline assessment arsenal, complementing rather than replacing existing diagnostic approaches,” Lakhan said.
“SHAAKE could be the ‘instant replay’ for brain injuries that sports medicine has been waiting for — but like any new technology, we need to make sure it works for every player, not just some,” Lakhan added.
Also weighing in, Richard Figler, MD, director of the Concussion Center, Cleveland Clinic Sports Medicine Center, Cleveland, cautioned that the survey participants were recruited from a concussion registry and self-reported an average of 23 concussions — more than one third of which happened 5-10 years prior — which begs the question, “How much are they actually remembering?”
“Our goal is to make sure that the athletes are safe and that we’re not missing concussions, and we don’t have great tools to start off with. This study opens up the door for some prospective studies [of SHAAKE] moving forward. I think we need more data before this should be listed as a definitive marker,” said Figler, who also wasn’t involved in the study.
In any case, he said, when it comes to suspected concussion in sports, “when in doubt, you sit them out,” Figler said.
This research received no external funding. Nowinski has received travel reimbursement from the NFL Players Association (NFLPA), NFL, World Rugby, WWE, and All Elite Wrestling; served as an expert witness in cases related to concussion and chronic traumatic encephalopathy; and is compensated for speaking appearances and serving on the NFL Concussion Settlement Player Advocacy Committee. Daniel H. Daneshvar served as an expert witness in legal cases involving brain injury and concussion and received funding from the Football Players Health Study at Harvard University, which is funded by the NFLPA and evaluates patients for the MGH Brain and Body TRUST Center, sponsored in part by the NFLPA. Lakhan and Figler had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM DIAGNOSTICS
MDMA Is Off the Table, So What’s Next for PTSD?
It has been 24 years since a pharmaceutical was last approved for posttraumatic stress disorder (PTSD). The condition is notoriously difficult to treat, with up to 40% patients finding no relief from symptoms through psychotherapy or current medications.
Many clinicians, advocates, and patients had pinned their hopes on the psychedelic drug midomafetamine with assisted therapy (MDMA-AT). However, in August, the US Food and Drug Administration (FDA) rejected it. At this point, it’s unclear when the therapy will be available, if ever.
“Not getting the FDA approval of any drug at this point is a setback for the field,” Lori Davis, MD, a senior research psychiatrist at the Birmingham Veterans Affairs (VA) Health Care System in Birmingham, Alabama, told Medscape Medical News.
Having an FDA-approved product would have helped increase public awareness of PTSD and driven interest in developing new therapies, said Davis, who is also adjunct professor of psychiatry at the Heersink School of Medicine, University of Alabama at Birmingham.
A Treatable Condition
So with MDMA-AT off the table, where does the field go next?
A public meeting in September hosted by the Reagan-Udall Foundation for the FDA in sought to answer that question. Agency officials joined representatives from the Department of Defense (DoD) and VA, patients, advocates, and industry representatives to discuss the current treatment landscape and what can be done to accelerate development of PTSD treatment.
Despite the common belief that PTSD is intractable, it “is a treatable condition,” Paula P. Schnurr, PhD, executive director of the VA National Center for PTSD, said at the meeting.
“There are effective treatments that work well for a lot of people, although not everyone has a satisfactory response,” she added.
The most effective psychotherapies are “trauma-focused,” and include cognitive processing therapy, eye movement desensitization and reprocessing, and prolonged exposure, according to the VA National Center for PTSD.
Three drugs have been approved by the FDA for PTSD: Venlafaxine (Effexor) in 1993, sertraline (Zoloft) in 1999, and paroxetine (Paxil) in 2000.
However, as the September meeting demonstrated, more therapies are needed.
“It’s clear to FDA and the federal government at large that there is an unmet need for safe and effective therapies to treat PTSD,” Bernard Fischer, MD, deputy director of the Division of Psychiatry in the Office of New Drugs at FDA’s Center for Drug Evaluation and Research, said at the meeting.
There is no shortage of research, Fischer added. Nearly 500 trials focused on PTSD are listed on clinicaltrials.gov are recruiting participants now or plan to soon.
Unsurprisingly, one of the primary drivers of PTSD therapeutics research is the VA. About 14% of the 5.7 million veterans who received care through the VA in 2023 had a diagnosis of PTSD.
“The US military is currently losing thousands of service members each year to PTSD- related disability discharges,” US Army Maj. Aaron Wolfgang, MD, a psychiatrist at the Walter Reed National Military Medical Center, said at the meeting. Only about 12%-20% of patients achieve remission with conventional therapies, added Wolfgang, who also is an assistant professor at the Uniformed Services University.
“For these reasons, establishing better treatments for PTSD is not only a matter of humanitarianism but also a pressing matter of national security,” he said.
The VA has committed at least $230 million to more than 140 active research projects in PTSD, Miriam J. Smyth, PhD, acting director of the clinical science, research and development service at the VA, said at the Reagan-Udall meeting.
One of the VA projects is the PTSD psychopharmacology initiative, which began in 2017 and now has 14 active clinical trials, said Smyth, who is also acting director for brain behavior and mental health at the VA. The first study should be finished by 2025.
The Million Veteran Program, with more than 1 million enrollees, has led to the discovery of genes related to re-experiencing traumatic memories and has confirmed that both PTSD and traumatic brain injury are risk factors for dementia, Smyth said.
The DoD has created a novel platform that establishes a common infrastructure for testing multiple drugs, called M-PACT. The platform allows sharing of placebo data across treatment arms. Drugs cycle off the platform if evidence indicates probability of success or failure.
Four trials are actively recruiting veterans and current service members. One is looking at vilazodone, approved in 2011 for major depressive disorder. It is being compared with placebo and fluoxetine in a trial that is currently recruiting.
Another trial will study daridorexant (sold as Quviviq), an orexin receptor antagonist, against placebo. The FDA approved daridorexant in 2022 as an insomnia treatment. A core issue in PTSD is sleep disruption, noted Davis.
New Therapies on the Way
Separately, Davis and colleagues are also studying methylphenidate, the stimulant used for attention-deficit/hyperactivity disorder. It may help with neurocognitive complaints and reduce PTSD symptoms, said Davis.
Because it is generic, few pharmaceutical manufacturers are likely to test it for PTSD, she said. But eventually, their work may lead a company to test newer stimulants for PTSD, she said.
Another potential therapeutic, BNC210, received Fast Track designation for PTSD from the FDA in 2019. Bionomics Limited in Australia will soon launch phase 3 trials of the investigational oral drug, which is a negative allosteric modulator of the alpha-7 nicotinic acetylcholine receptor. In late July, the company announced “ favorable feedback” from the agency on its phase 2 study, which led to the decision to move forward with larger trials.
Researchers at Brigham and Women’s Hospital have just reported that they may have found a target within the brain that will allow for transcranial magnetic stimulation (TMS) to ameliorate PTSD symptoms. They published results of a mapping effort in Nature Neuroscience and reported on one patient who had improved symptoms after receiving TMS for severe PTSD.
But perhaps one of the most promising treatments is a combination of sertraline and the new psychiatric medication brexpiprazole.
Brexpiprazole was developed by Otsuka Pharmaceutical and approved in the United States in 2015 as an adjunctive therapy to antidepressants for major depressive disorder and as a treatment for schizophrenia. In 2023, the FDA approved it for Alzheimer’s-related agitation. However, according to Otsuka, its mechanism of action is unknown.
Its efficacy may be mediated through a combination of partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, antagonist activity at serotonin 5-HT2A receptors, as well as antagonism of alpha-1B/2C receptors, said the company.
“It is the combination, rather than either alone, that’s going to have that broad synergistic pharmacology that is obviously potent for ameliorating the symptoms of PTSD,” said Davis, who has received consulting fees from Otsuka. “That’s an exciting development.”
Otsuka and partner Lundbeck Pharmaceuticals reported results in May from the companies’ phase 2 and 3 randomized clinical trials. The therapy achieved a statistically significant reduction (P <.05) in PTSD symptoms compared with sertraline plus placebo. This was without any supplemental psychotherapy.
The FDA accepted the companies’ new drug application in June and is expected to make a decision on approval in February 2025.
The Potential of Psychedelics
Though Lykos Therapeutics may have to go back to the drawing board on its MDMA-AT, psychedelics still have potential as PTSD therapies, Smyth said, who added that the VA is continuing to encourage study of MDMA and other psychedelic agents.
The VA issued a call for proposals for research on psychedelics in January, focused on MDMA or psilocybin in combination with psychotherapy. The administration received the first wave of applications early in the summer.
Scientific peer review panels made up of research experts from within and outside the VA have reviewed the applications and funding announcements are expected this fall, Smyth said.
Wolfgang, the Army psychiatrist, said, “Under the psychedelic treatment research clinical trial award, we welcome investigators to apply to what we anticipate will usher in a new era of innovation and hope for service members and their families who need it the most.”
Psychedelic studies are also proceeding without VA funding, as they have for years, when most of the trials were backed by universities or foundations or other private money. Johns Hopkins University is recruiting for a study in which patients would receive psilocybin along with trauma-focused psychotherapy, as is Ohio State University.
London-based Compass Pathways said in May that it successfully completed a phase 2 trial of Comp360, its synthetic psilocybin, in PTSD. The company has started a phase 3 study in treatment-resistant depression but has not given any further updates on PTSD.
Davis said that she believes that the FDA’s rejection of Lykos won’t lead to a shutdown of exploration of psychedelics.
“I think it informs these designs going forward, but it doesn’t eliminate that whole field of research,” she said.
Davis reported receiving consulting fees from Boehringer Ingelheim and Otsuka and research funding from Alkermes, the Patient-Centered Outcomes Research Institute, and the VA. Schnurr, Fischer, Smyth, and Wolfgang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
It has been 24 years since a pharmaceutical was last approved for posttraumatic stress disorder (PTSD). The condition is notoriously difficult to treat, with up to 40% patients finding no relief from symptoms through psychotherapy or current medications.
Many clinicians, advocates, and patients had pinned their hopes on the psychedelic drug midomafetamine with assisted therapy (MDMA-AT). However, in August, the US Food and Drug Administration (FDA) rejected it. At this point, it’s unclear when the therapy will be available, if ever.
“Not getting the FDA approval of any drug at this point is a setback for the field,” Lori Davis, MD, a senior research psychiatrist at the Birmingham Veterans Affairs (VA) Health Care System in Birmingham, Alabama, told Medscape Medical News.
Having an FDA-approved product would have helped increase public awareness of PTSD and driven interest in developing new therapies, said Davis, who is also adjunct professor of psychiatry at the Heersink School of Medicine, University of Alabama at Birmingham.
A Treatable Condition
So with MDMA-AT off the table, where does the field go next?
A public meeting in September hosted by the Reagan-Udall Foundation for the FDA in sought to answer that question. Agency officials joined representatives from the Department of Defense (DoD) and VA, patients, advocates, and industry representatives to discuss the current treatment landscape and what can be done to accelerate development of PTSD treatment.
Despite the common belief that PTSD is intractable, it “is a treatable condition,” Paula P. Schnurr, PhD, executive director of the VA National Center for PTSD, said at the meeting.
“There are effective treatments that work well for a lot of people, although not everyone has a satisfactory response,” she added.
The most effective psychotherapies are “trauma-focused,” and include cognitive processing therapy, eye movement desensitization and reprocessing, and prolonged exposure, according to the VA National Center for PTSD.
Three drugs have been approved by the FDA for PTSD: Venlafaxine (Effexor) in 1993, sertraline (Zoloft) in 1999, and paroxetine (Paxil) in 2000.
However, as the September meeting demonstrated, more therapies are needed.
“It’s clear to FDA and the federal government at large that there is an unmet need for safe and effective therapies to treat PTSD,” Bernard Fischer, MD, deputy director of the Division of Psychiatry in the Office of New Drugs at FDA’s Center for Drug Evaluation and Research, said at the meeting.
There is no shortage of research, Fischer added. Nearly 500 trials focused on PTSD are listed on clinicaltrials.gov are recruiting participants now or plan to soon.
Unsurprisingly, one of the primary drivers of PTSD therapeutics research is the VA. About 14% of the 5.7 million veterans who received care through the VA in 2023 had a diagnosis of PTSD.
“The US military is currently losing thousands of service members each year to PTSD- related disability discharges,” US Army Maj. Aaron Wolfgang, MD, a psychiatrist at the Walter Reed National Military Medical Center, said at the meeting. Only about 12%-20% of patients achieve remission with conventional therapies, added Wolfgang, who also is an assistant professor at the Uniformed Services University.
“For these reasons, establishing better treatments for PTSD is not only a matter of humanitarianism but also a pressing matter of national security,” he said.
The VA has committed at least $230 million to more than 140 active research projects in PTSD, Miriam J. Smyth, PhD, acting director of the clinical science, research and development service at the VA, said at the Reagan-Udall meeting.
One of the VA projects is the PTSD psychopharmacology initiative, which began in 2017 and now has 14 active clinical trials, said Smyth, who is also acting director for brain behavior and mental health at the VA. The first study should be finished by 2025.
The Million Veteran Program, with more than 1 million enrollees, has led to the discovery of genes related to re-experiencing traumatic memories and has confirmed that both PTSD and traumatic brain injury are risk factors for dementia, Smyth said.
The DoD has created a novel platform that establishes a common infrastructure for testing multiple drugs, called M-PACT. The platform allows sharing of placebo data across treatment arms. Drugs cycle off the platform if evidence indicates probability of success or failure.
Four trials are actively recruiting veterans and current service members. One is looking at vilazodone, approved in 2011 for major depressive disorder. It is being compared with placebo and fluoxetine in a trial that is currently recruiting.
Another trial will study daridorexant (sold as Quviviq), an orexin receptor antagonist, against placebo. The FDA approved daridorexant in 2022 as an insomnia treatment. A core issue in PTSD is sleep disruption, noted Davis.
New Therapies on the Way
Separately, Davis and colleagues are also studying methylphenidate, the stimulant used for attention-deficit/hyperactivity disorder. It may help with neurocognitive complaints and reduce PTSD symptoms, said Davis.
Because it is generic, few pharmaceutical manufacturers are likely to test it for PTSD, she said. But eventually, their work may lead a company to test newer stimulants for PTSD, she said.
Another potential therapeutic, BNC210, received Fast Track designation for PTSD from the FDA in 2019. Bionomics Limited in Australia will soon launch phase 3 trials of the investigational oral drug, which is a negative allosteric modulator of the alpha-7 nicotinic acetylcholine receptor. In late July, the company announced “ favorable feedback” from the agency on its phase 2 study, which led to the decision to move forward with larger trials.
Researchers at Brigham and Women’s Hospital have just reported that they may have found a target within the brain that will allow for transcranial magnetic stimulation (TMS) to ameliorate PTSD symptoms. They published results of a mapping effort in Nature Neuroscience and reported on one patient who had improved symptoms after receiving TMS for severe PTSD.
But perhaps one of the most promising treatments is a combination of sertraline and the new psychiatric medication brexpiprazole.
Brexpiprazole was developed by Otsuka Pharmaceutical and approved in the United States in 2015 as an adjunctive therapy to antidepressants for major depressive disorder and as a treatment for schizophrenia. In 2023, the FDA approved it for Alzheimer’s-related agitation. However, according to Otsuka, its mechanism of action is unknown.
Its efficacy may be mediated through a combination of partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, antagonist activity at serotonin 5-HT2A receptors, as well as antagonism of alpha-1B/2C receptors, said the company.
“It is the combination, rather than either alone, that’s going to have that broad synergistic pharmacology that is obviously potent for ameliorating the symptoms of PTSD,” said Davis, who has received consulting fees from Otsuka. “That’s an exciting development.”
Otsuka and partner Lundbeck Pharmaceuticals reported results in May from the companies’ phase 2 and 3 randomized clinical trials. The therapy achieved a statistically significant reduction (P <.05) in PTSD symptoms compared with sertraline plus placebo. This was without any supplemental psychotherapy.
The FDA accepted the companies’ new drug application in June and is expected to make a decision on approval in February 2025.
The Potential of Psychedelics
Though Lykos Therapeutics may have to go back to the drawing board on its MDMA-AT, psychedelics still have potential as PTSD therapies, Smyth said, who added that the VA is continuing to encourage study of MDMA and other psychedelic agents.
The VA issued a call for proposals for research on psychedelics in January, focused on MDMA or psilocybin in combination with psychotherapy. The administration received the first wave of applications early in the summer.
Scientific peer review panels made up of research experts from within and outside the VA have reviewed the applications and funding announcements are expected this fall, Smyth said.
Wolfgang, the Army psychiatrist, said, “Under the psychedelic treatment research clinical trial award, we welcome investigators to apply to what we anticipate will usher in a new era of innovation and hope for service members and their families who need it the most.”
Psychedelic studies are also proceeding without VA funding, as they have for years, when most of the trials were backed by universities or foundations or other private money. Johns Hopkins University is recruiting for a study in which patients would receive psilocybin along with trauma-focused psychotherapy, as is Ohio State University.
London-based Compass Pathways said in May that it successfully completed a phase 2 trial of Comp360, its synthetic psilocybin, in PTSD. The company has started a phase 3 study in treatment-resistant depression but has not given any further updates on PTSD.
Davis said that she believes that the FDA’s rejection of Lykos won’t lead to a shutdown of exploration of psychedelics.
“I think it informs these designs going forward, but it doesn’t eliminate that whole field of research,” she said.
Davis reported receiving consulting fees from Boehringer Ingelheim and Otsuka and research funding from Alkermes, the Patient-Centered Outcomes Research Institute, and the VA. Schnurr, Fischer, Smyth, and Wolfgang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
It has been 24 years since a pharmaceutical was last approved for posttraumatic stress disorder (PTSD). The condition is notoriously difficult to treat, with up to 40% patients finding no relief from symptoms through psychotherapy or current medications.
Many clinicians, advocates, and patients had pinned their hopes on the psychedelic drug midomafetamine with assisted therapy (MDMA-AT). However, in August, the US Food and Drug Administration (FDA) rejected it. At this point, it’s unclear when the therapy will be available, if ever.
“Not getting the FDA approval of any drug at this point is a setback for the field,” Lori Davis, MD, a senior research psychiatrist at the Birmingham Veterans Affairs (VA) Health Care System in Birmingham, Alabama, told Medscape Medical News.
Having an FDA-approved product would have helped increase public awareness of PTSD and driven interest in developing new therapies, said Davis, who is also adjunct professor of psychiatry at the Heersink School of Medicine, University of Alabama at Birmingham.
A Treatable Condition
So with MDMA-AT off the table, where does the field go next?
A public meeting in September hosted by the Reagan-Udall Foundation for the FDA in sought to answer that question. Agency officials joined representatives from the Department of Defense (DoD) and VA, patients, advocates, and industry representatives to discuss the current treatment landscape and what can be done to accelerate development of PTSD treatment.
Despite the common belief that PTSD is intractable, it “is a treatable condition,” Paula P. Schnurr, PhD, executive director of the VA National Center for PTSD, said at the meeting.
“There are effective treatments that work well for a lot of people, although not everyone has a satisfactory response,” she added.
The most effective psychotherapies are “trauma-focused,” and include cognitive processing therapy, eye movement desensitization and reprocessing, and prolonged exposure, according to the VA National Center for PTSD.
Three drugs have been approved by the FDA for PTSD: Venlafaxine (Effexor) in 1993, sertraline (Zoloft) in 1999, and paroxetine (Paxil) in 2000.
However, as the September meeting demonstrated, more therapies are needed.
“It’s clear to FDA and the federal government at large that there is an unmet need for safe and effective therapies to treat PTSD,” Bernard Fischer, MD, deputy director of the Division of Psychiatry in the Office of New Drugs at FDA’s Center for Drug Evaluation and Research, said at the meeting.
There is no shortage of research, Fischer added. Nearly 500 trials focused on PTSD are listed on clinicaltrials.gov are recruiting participants now or plan to soon.
Unsurprisingly, one of the primary drivers of PTSD therapeutics research is the VA. About 14% of the 5.7 million veterans who received care through the VA in 2023 had a diagnosis of PTSD.
“The US military is currently losing thousands of service members each year to PTSD- related disability discharges,” US Army Maj. Aaron Wolfgang, MD, a psychiatrist at the Walter Reed National Military Medical Center, said at the meeting. Only about 12%-20% of patients achieve remission with conventional therapies, added Wolfgang, who also is an assistant professor at the Uniformed Services University.
“For these reasons, establishing better treatments for PTSD is not only a matter of humanitarianism but also a pressing matter of national security,” he said.
The VA has committed at least $230 million to more than 140 active research projects in PTSD, Miriam J. Smyth, PhD, acting director of the clinical science, research and development service at the VA, said at the Reagan-Udall meeting.
One of the VA projects is the PTSD psychopharmacology initiative, which began in 2017 and now has 14 active clinical trials, said Smyth, who is also acting director for brain behavior and mental health at the VA. The first study should be finished by 2025.
The Million Veteran Program, with more than 1 million enrollees, has led to the discovery of genes related to re-experiencing traumatic memories and has confirmed that both PTSD and traumatic brain injury are risk factors for dementia, Smyth said.
The DoD has created a novel platform that establishes a common infrastructure for testing multiple drugs, called M-PACT. The platform allows sharing of placebo data across treatment arms. Drugs cycle off the platform if evidence indicates probability of success or failure.
Four trials are actively recruiting veterans and current service members. One is looking at vilazodone, approved in 2011 for major depressive disorder. It is being compared with placebo and fluoxetine in a trial that is currently recruiting.
Another trial will study daridorexant (sold as Quviviq), an orexin receptor antagonist, against placebo. The FDA approved daridorexant in 2022 as an insomnia treatment. A core issue in PTSD is sleep disruption, noted Davis.
New Therapies on the Way
Separately, Davis and colleagues are also studying methylphenidate, the stimulant used for attention-deficit/hyperactivity disorder. It may help with neurocognitive complaints and reduce PTSD symptoms, said Davis.
Because it is generic, few pharmaceutical manufacturers are likely to test it for PTSD, she said. But eventually, their work may lead a company to test newer stimulants for PTSD, she said.
Another potential therapeutic, BNC210, received Fast Track designation for PTSD from the FDA in 2019. Bionomics Limited in Australia will soon launch phase 3 trials of the investigational oral drug, which is a negative allosteric modulator of the alpha-7 nicotinic acetylcholine receptor. In late July, the company announced “ favorable feedback” from the agency on its phase 2 study, which led to the decision to move forward with larger trials.
Researchers at Brigham and Women’s Hospital have just reported that they may have found a target within the brain that will allow for transcranial magnetic stimulation (TMS) to ameliorate PTSD symptoms. They published results of a mapping effort in Nature Neuroscience and reported on one patient who had improved symptoms after receiving TMS for severe PTSD.
But perhaps one of the most promising treatments is a combination of sertraline and the new psychiatric medication brexpiprazole.
Brexpiprazole was developed by Otsuka Pharmaceutical and approved in the United States in 2015 as an adjunctive therapy to antidepressants for major depressive disorder and as a treatment for schizophrenia. In 2023, the FDA approved it for Alzheimer’s-related agitation. However, according to Otsuka, its mechanism of action is unknown.
Its efficacy may be mediated through a combination of partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, antagonist activity at serotonin 5-HT2A receptors, as well as antagonism of alpha-1B/2C receptors, said the company.
“It is the combination, rather than either alone, that’s going to have that broad synergistic pharmacology that is obviously potent for ameliorating the symptoms of PTSD,” said Davis, who has received consulting fees from Otsuka. “That’s an exciting development.”
Otsuka and partner Lundbeck Pharmaceuticals reported results in May from the companies’ phase 2 and 3 randomized clinical trials. The therapy achieved a statistically significant reduction (P <.05) in PTSD symptoms compared with sertraline plus placebo. This was without any supplemental psychotherapy.
The FDA accepted the companies’ new drug application in June and is expected to make a decision on approval in February 2025.
The Potential of Psychedelics
Though Lykos Therapeutics may have to go back to the drawing board on its MDMA-AT, psychedelics still have potential as PTSD therapies, Smyth said, who added that the VA is continuing to encourage study of MDMA and other psychedelic agents.
The VA issued a call for proposals for research on psychedelics in January, focused on MDMA or psilocybin in combination with psychotherapy. The administration received the first wave of applications early in the summer.
Scientific peer review panels made up of research experts from within and outside the VA have reviewed the applications and funding announcements are expected this fall, Smyth said.
Wolfgang, the Army psychiatrist, said, “Under the psychedelic treatment research clinical trial award, we welcome investigators to apply to what we anticipate will usher in a new era of innovation and hope for service members and their families who need it the most.”
Psychedelic studies are also proceeding without VA funding, as they have for years, when most of the trials were backed by universities or foundations or other private money. Johns Hopkins University is recruiting for a study in which patients would receive psilocybin along with trauma-focused psychotherapy, as is Ohio State University.
London-based Compass Pathways said in May that it successfully completed a phase 2 trial of Comp360, its synthetic psilocybin, in PTSD. The company has started a phase 3 study in treatment-resistant depression but has not given any further updates on PTSD.
Davis said that she believes that the FDA’s rejection of Lykos won’t lead to a shutdown of exploration of psychedelics.
“I think it informs these designs going forward, but it doesn’t eliminate that whole field of research,” she said.
Davis reported receiving consulting fees from Boehringer Ingelheim and Otsuka and research funding from Alkermes, the Patient-Centered Outcomes Research Institute, and the VA. Schnurr, Fischer, Smyth, and Wolfgang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
1 in 4 Unresponsive Coma Patients May Retain Some Awareness
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
“We found that at least 1 in 4 patients who are unresponsive to commands might actually be quite present and highly cognitive,” said study investigator Nicholas D. Schiff, MD, Feil Family Brain & Mind Research Institute and Department of Neurology, Weill Cornell Medicine, Rockefeller University Hospital, New York.
“In other words, if you go to the bedside and carefully examine someone with a severe brain injury and find no evidence of responsiveness, no one has been able to give you an a priori number to say how likely you are to be wrong in thinking this person is actually unaware, not processing language, and not capable of high-level cognitive work. And the answer to that now is at least 1 in 4 times.”
The findings were published online in The New England Journal of Medicine.
Clinical Implications?
Cognitive motor dissociation (CMD) is a condition whereby patients with a severe brain injury who are unresponsive to commands at the bedside show brain activity on functional MRI (fMRI) or electroencephalography (EEG) when presented with selective motor imagery commands, such as “imagine playing tennis,” or “ imagine opening and closing your hand.”
Previous research shows that CMD is present in 10%-20% of people with a disorder of consciousness, a rate similar to that in patients with acute or chronic brain injury.
Understanding that a patient who appears unconscious has signs of cognitive processing could change the way clinicians and family interact with such individuals. Unresponsive patients who are aware may eventually be able to harness emerging communication technologies such as brain-computer interfaces.
In addition, knowing an individual’s CMD status could aid in prognosis. “We know from one study that there’s a four times increased likelihood that patients will be independent in a year in their function if they have cognitive motor dissociation,” said Dr. Schiff.
Unlike most previous studies of CMD, which were conducted at single sites and had relatively small cohorts, this new study included 353 adults with a disorder of consciousness (mean age, 37.9 years; 64% male) at six multinational sites.
Participants were recruited using a variety of methods, including consecutive enrollment of critically ill patients in the intensive care unit and enrollment of those with chronic illness or injury who were in the postacute phase of brain injury.
Response to Commands
Study participants were at different stages of recovery from an acute brain injury that had occurred an average of 8 months before the study started.
To determine the presence or absence of an observable response to commands among participants, trained staff used the Coma Recovery Scale–Revised (CRS-R); scores on this instrument range from 0 to 23, and higher scores indicate better neurobehavioral function.
About 40% of individuals were diagnosed with coma or vegetative state, 29% with minimally conscious state–minus, and 22% with minimally conscious state–plus. In all, 10% had emerged from a minimally conscious state.
Researchers assessed response to timed and repeated commands using fMRI or EEG in participants without an observable response to verbal commands, including those with a behavioral diagnosis of coma, vegetative state, or minimally conscious state–minus, and in participants with an observable response to verbal commands.
Of the 353 study participants, 61% underwent at least one fMRI assessment and 74% at least one EEG assessment. Both fMRI and EEG were performed in 35% of participants.
Dr. Schiff explained the two assessment types provide slightly different information, in that they measuring different types of brain signals. He also noted that although “every medical center in the world” has EEG, many do not have fMRI.
The brain imaging assessments captured brain activity within the motor area of the frontal cortex when tasked with motor imagery.
Of the 241 participants deemed to be in a coma or vegetative state or minimally conscious state–minus on the basis of CRS-R score, 60 (25%) had a response to commands on task-based fMRI, task-based EEG, or both.
The percentage of participants with CMD varied across study sites, from 2% to 45%, but Dr. Schiff said the reason for this is unclear.
The proportion of participants with CMD may have been even higher if all individuals had been assessed with both imaging techniques, he said.
Higher Rate of Awareness Than in Previous Research
The investigators noted that the percentage of participants with CMD in their study was up to 10 percentage points higher than in previous studies. This may be due to the multimodal approach that classified participants undergoing assessment with both fMRI and EEG on the basis of responses on either technique, they said.
The median age was lower among participants with CMD than those without CMD (30.5 years vs 45.3 years).
Compared with participants without CMD, a higher percentage of those with such dissociation had brain trauma as an etiologic factor (65% vs 38%) and a diagnosis of minimally conscious state–minus on the CRS-R (53% vs 38%).
Among people with CMD, 18% were assessed with fMRI only, 22% with EEG only, and 60% with both fMRI and EEG.
Dr. Schiff noted that the use of both fMRI and EEG appears to be more sensitive in detecting brain activity during tasks compared with use of one of these techniques alone.
Of the 112 participants with a diagnosis of minimally conscious state–plus or who had emerged from the minimally conscious state, 38% had a response to commands on task-based fMRI, task-based EEG, or both. Among these participants, 23% were assessed with fMRI only, 19% with EEG only, and 58% with both fMRI and EEG.
Research shows “it’s very clear that people with severe brain injury continue to get better over time,” noted Dr. Schiff. “Every month and week matters, and so it probably is the case that a lot of these patients are picking up the level of recovery, and the later we go out to measure them, the more likely we are to find people who are CMD than not.”
These new results should prompt further study to explore whether detection of CMD can lead to improved outcomes, the investigators noted. “In addition, the standardization, validation, and simplification of task-based fMRI and EEG methods that are used to detect cognitive motor dissociation are needed to prompt widespread clinical integration of these techniques and investigation of the bioethical implications of the findings.”
All study participants with chronic brain injury had survived their initial illness or injury and had access to a research facility with advanced fMRI and EEG capabilities. “This survival bias may reflect greater cognitive reserve and resilience over time among the participants. As such, the results of our study may not be generalizable to the overall population of patients with cognitive motor dissociation,” the investigators wrote.
Another study limitation was that participating sites used heterogeneous strategies to acquire, analyze, and interpret data, which led to differences in the number, type, and ordering of the cognitive tasks assessed on fMRI and EEG.
“These differences, along with variations in recruitment strategies and participant characteristics, may have contributed to the unequal percentage of participants with cognitive motor dissociation observed at each site. Our findings may therefore not be generalizable across all centers,” the researchers wrote.
Only a few academic medical centers have the specially trained personnel and techniques needed to assess patients for CMD — which, the researchers noted, limits the feasibility of performing these assessments in general practice.
Challenging Research
Commenting on the research, Aarti Sarwal, MD, professor of neurology and section chief, Neurocritical Care, Virginia Commonwealth University, Richmond, Virginia, noted that this was a “very challenging” study to perform, given that only a few academic centers are equipped to perform both fMRI and quantitative EEG analysis.
“In general, finding patients this far out, who have access to clinical, radiological, and electrophysiological testing and were provided good care enough to receive these, is a mammoth task in itself.”
Dr. Sarwal said the study builds on efforts of the Curing Coma campaign , a clinical, scientific, and public health effort of the Neurocritical Care Society to tackle the concept of coma as a treatable medical entity.
“It continues to highlight the challenges of prognostication in acute brain injured patients by showing a higher presence of cognitive function than previously perceived,” she said.
Dr. Sarwal believes that the study’s largest impact is underscoring the need for more research into understanding the degree and quality of cognitive processing in patients with a disorder of consciousness. But it also underlines the need for a “healthy debate” on the cost/benefit analysis of pursuing such research, given the limited number of patients with access to resources.
“This debate needs to include the caregivers and families outside the traditional realms of stakeholders overseeing the science.”
Although communication with comatose patients is still “a ways away,” this research is “a step in the right direction,” said Dr. Sarwal.
The study was funded by the James S. McDonnell Foundation and others. Dr. Schiff and Dr. Sarwal report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Federal Health Care Data Trends 2024
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue:
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue:
Federal Health Care Data Trends is a special supplement to Federal Practitioner, showcasing the latest research in health care for veterans and active-duty military members via compelling infographics. Click below to view highlights from the issue: