User login
Could EHR Pharmacy Errors Put Veterans at Risk?
Will the new US Department of Veterans Affairs (VA) pharmacy software be safe and effective? That was the topic when David Case, the VA Deputy Inspector General, spoke in the US House of Representatives Veterans Affairs Committee technology modernization subcommittee hearing on February 15.
Questions like that have dogged the project since 2018, when the VA began rolling out the Oracle Cerner electronic health record (EHR) system as the successor to ViSTA.
The Oracle system has been beset by one glitch after another since its arrival. And in that time, Case said, the VA Office of Inspector General (OIG) has been engaging with VA employees at sites in Washington, Oregon, Ohio, Illinois, and other locations where the modernization program has been piloted.
The most recent OIG investigation of pharmacy-related patient safety issues began with a review of an allegation of a prescription backlog at Columbus, Ohio, where the system went live on April 30, 2022. The OIG found that facility leaders took “timely and sustainable steps” to manage that issue. However, other unresolved patient safety issues came to light, such as medication inaccuracies, inaccurate medication data, and insufficient staffing. The OIG also found staff were creating “numerous work arounds” to provide patient care, and that the volume of staff educational materials for pharmacy-related functions was “overwhelming.”
Those problems were just the latest in a long queue. In May 2021, after the first VA deployment of the new EHR at the Mann-Grandstaff VA Medical Center in Spokane, Washington, a pharmacy patient safety team under the VA National Center for Patient Safety (NCPS) also had identified patient safety issues and “multiple” concerns regarding the system’s usability. For example, updates to a patient’s active medication list were not routinely reflected at the patient’s next appointment. Despite knowing about such challenges, Case noted in his report, VA leaders deployed the new EHR at 4 more VA medical centers.
Cerner/ViSTA Communication
One major cause of the current problems is the way the systems “talk” to each other. EHR information is communicated between VHA facilities through channels that include the Joint Longitudinal Viewer (JLV) and the Health Data Repository, which stores patient-specific clinical information from both the legacy and the new EHR systems. The JLV application allows clinicians to access a read only version of a patient’s EHR from both systems.
Every medication used in VHA has a VA Unique Identifier (VUID). When a patient is prescribed a medication at a new EHR site, that medication’s VUID is sent to the Health Data Repository. If that patient seeks care from a legacy health care practitioner (HCP), and that HCP enters a medication order, a software interface accesses the VUID from the Health Data Repository to verify that the medication being prescribed is safe and compatible with the medications and allergies previously documented in the patient’s record.
However, on March 31, 2023, staff from a ViSTA site found an incorrect medication order when prescribing a new medication to a patient who had received care and medications at a new EHR site. This in turn led to the discovery that an error in Oracle software coding had resulted in the “widespread transmission” of incorrect VUIDs from new EHR sites to legacy EHR sites, the OIG found. VA leaders and HCPs were notified of the potential clinical impact and were given specific instructions on how to mitigate the issue. They were asked to “please share widely.”
On top of that, days later, patient safety managers across the Veterans Health Administration (VHA) were told that drug-to-drug interactions, duplicate medication orders, and allergy checks were not functioning as expected, and they too were provided with remedial actions.
Oracle applied a successful software patch on in April 2023, to ensure accurate VUIDs were attached to all mail order pharmacy–processed prescriptions from that date forward. However, the OIG learned the incorrect VUIDs sent from new EHR sites and stored in the Health Data Repository from as far back as October 2020 had not been corrected. Case told the subcommittee that on November 29, 2023, the VHA Pharmacy Council reported withdrawing a request for Oracle to send corrected medication VUID data to the Health Data Repository, on the presumption that remaining inaccurate VUIDs would expire in early April 2024, and the data would be corrected at that time.
The OIG is concerned, Case said, that patient medication data remains inaccurate almost a year after VA learned of the issue. The mail order pharmacy-related data generated from approximately 120,000 patients served by new EHR sites are still incorrect. These patients face an ongoing risk of an adverse medication-related event if they receive care and medications from a VA medical center using the legacy EHR system.
The OIG also learned of other problems associated with transmission of medication and allergy information, which could have consequences such as:
- Patient medications being discontinued or stopped by new HCPs using Cerner that appear in ViSTA as active and current prescriptions;
- Allergy-warning messages not appearing when intended or inappropriately appearing for the wrong medication;
- Duplicate medication order checks not appearing when intended or inappropriately appearing for the wrong drug;
- Patient active medication lists having incomplete or inaccurate information, such as missing prescriptions, duplicate prescriptions, or incorrect medication order statuses.
The OIG warned VHA employees about the risks, although it wasn’t possible to determine who might actually be at risk. A VHA leader told the OIG that all patients who have been prescribed any medications or have medication allergies documented at a at a Cerner site are at risk. That could mean as many as 250,000 patients: As of September 2023, approximately 190,000 patients had a medication prescribed and 126,000 had an allergy documented at a new EHR site.
Case Example
Not surprisingly, “the OIG is not confident in [EHRM-Integration Office] leaders’ oversight and control of the new systems’ Health Data Repository interface programming,” Case said. He cited the case of a patient with posttraumatic stress disorder and traumatic brain injury with adrenal insufficiency. Four days prior to admission, a ViSTA site pharmacist used the EHR to perform a medication reconciliation for the patient. The data available did not include the patient’s most recent prednisone prescription, which had been ordered by an HCP at a facility using Cerner.
A nurse practitioner performed another reconciliation when the patient was admitted to the residential program, but the patient was unsure of all their medications. Because the most recent prednisone prescription was not visible in ViSTA, the prednisone appeared to have been completed at least 3 months prior to admission and was therefore not prescribed in the admission medication orders.
Five days into the residential program, the patient began exhibiting unusual behaviors associated with the lack of prednisone. The patient realized they needed more prednisone, but the nurse explained there was no prednisone on the patient’s medication list. Eventually, the patient found the active prednisone order on their personal cell phone and was transferred to a local emergency department for care.
Work Arounds
The VHA’s efforts to forestall or mitigate system errors have in some cases had a cascade effect. For example, HCPs must essentially back up what the automated software is intended to do, with “complex, time-consuming” multistep manual safety checks when prescribing new medications for patients previously cared for at a Cerner site. The OIG is concerned that this increased vigilance is “unsustainable” by pharmacists and frontline staff and could lead to burnout and medication-related patient safety events. After the new EHR launched, the OIG found, burnout symptoms for pharmacy staff increased. Nonetheless, Case told the committee, OIG staff “have observed [employees’] unwavering commitment to prioritizing the care of patients while mitigating implementation challenges.”
EHR-related workload burdens have necessitated other adjustments. Columbus, for instance, hired 9 full-time clinical pharmacists—a 62% staffing increase—to help reduce the backlog. Pharmacy leaders created approximately 29 additional work-arounds to support pharmacy staff and prevent delays. Facility pharmacy leaders also developed approximately 25 educational materials, such as tip sheets, reference guides, and job aids. The OIG’s concern—apart from the overwhelming amount of information for staff to implement—is that such prophylactic measures may in fact give rise to inconsistent practices, which increase risks to patient safety.
Committed to Working With the VA
Mike Sicilia, executive vice president of Oracle Corporation, told lawmakers in the hearing, “After the initial deployments, it became clear that the pharmacy system needed to be enhanced to better meet VA’s needs. To that end, in August 2022, shortly after Oracle completed its acquisition of Cerner, VA contracted with us for seven enhancements that overall would adapt the pharmacy system to a more bidirectional system between VA providers placing prescription orders and VA pharmacists fulfilling and dispensing them.” Those enhancements are all live for VA providers and pharmacists to use now, he said, except for one that is undergoing additional testing.
He added, “As with any healthcare technology system, there is a need for continuous improvements but that does not mean the system is not safe and effective in its current state. Oracle is committed to working with VA … throughout the reset period to identify workflows and other items that can be simplified or streamlined to improve the overall user and pharmacy experience.”
Standardizing workflows and ensuring training and communications to pharmacists about the latest updates will discourage use of work-arounds, Sicilia said, and “help with improving morale and satisfaction with the system.” During a visit in early February by VA and the Oracle team to the Lovell Federal Health Care Center in North Chicago, “feedback from pharmacists was positive about the training and readiness for using the new pharmacy system.”
The backlog, at least, may be resolved. Sicilia said on average more than 215,000 outpatient prescriptions are being filled each month. “The current live sites do not have a backlog in filling prescriptions. Recent data from this month show that three of the five live sites have zero prescriptions waiting to be processed that are older than seven days. The two other live sites have an average of two prescriptions older than seven days,” he said.
Although Oracle Health has since resolved some of the identified issues, the OIG is concerned that the new EHR will continue to be deployed at medical facilities despite “myriad” as-yet unresolved issues related to inaccurate medication ordering, reconciliation, and dispensing. The VHA has paused Cerner deployments multiple times.
“It is unclear whether identified problems are being adequately resolved before additional deployments,” Case said. “There is also the question of whether there is sufficient transparency and communication among EHRM-IO, VHA and facility leaders, VA leaders, and Oracle Health needed for quality control and critical coordination. Trust in VA is also dependent on patients being fully and quickly advised when issues affecting them are identified and addressed. As VA moves toward its deployment next month at a complex facility jointly operated with the Department of Defense, transparency, communication, and program management will be essential to getting it right. Failures in these areas risk cascading problems.”
Will the new US Department of Veterans Affairs (VA) pharmacy software be safe and effective? That was the topic when David Case, the VA Deputy Inspector General, spoke in the US House of Representatives Veterans Affairs Committee technology modernization subcommittee hearing on February 15.
Questions like that have dogged the project since 2018, when the VA began rolling out the Oracle Cerner electronic health record (EHR) system as the successor to ViSTA.
The Oracle system has been beset by one glitch after another since its arrival. And in that time, Case said, the VA Office of Inspector General (OIG) has been engaging with VA employees at sites in Washington, Oregon, Ohio, Illinois, and other locations where the modernization program has been piloted.
The most recent OIG investigation of pharmacy-related patient safety issues began with a review of an allegation of a prescription backlog at Columbus, Ohio, where the system went live on April 30, 2022. The OIG found that facility leaders took “timely and sustainable steps” to manage that issue. However, other unresolved patient safety issues came to light, such as medication inaccuracies, inaccurate medication data, and insufficient staffing. The OIG also found staff were creating “numerous work arounds” to provide patient care, and that the volume of staff educational materials for pharmacy-related functions was “overwhelming.”
Those problems were just the latest in a long queue. In May 2021, after the first VA deployment of the new EHR at the Mann-Grandstaff VA Medical Center in Spokane, Washington, a pharmacy patient safety team under the VA National Center for Patient Safety (NCPS) also had identified patient safety issues and “multiple” concerns regarding the system’s usability. For example, updates to a patient’s active medication list were not routinely reflected at the patient’s next appointment. Despite knowing about such challenges, Case noted in his report, VA leaders deployed the new EHR at 4 more VA medical centers.
Cerner/ViSTA Communication
One major cause of the current problems is the way the systems “talk” to each other. EHR information is communicated between VHA facilities through channels that include the Joint Longitudinal Viewer (JLV) and the Health Data Repository, which stores patient-specific clinical information from both the legacy and the new EHR systems. The JLV application allows clinicians to access a read only version of a patient’s EHR from both systems.
Every medication used in VHA has a VA Unique Identifier (VUID). When a patient is prescribed a medication at a new EHR site, that medication’s VUID is sent to the Health Data Repository. If that patient seeks care from a legacy health care practitioner (HCP), and that HCP enters a medication order, a software interface accesses the VUID from the Health Data Repository to verify that the medication being prescribed is safe and compatible with the medications and allergies previously documented in the patient’s record.
However, on March 31, 2023, staff from a ViSTA site found an incorrect medication order when prescribing a new medication to a patient who had received care and medications at a new EHR site. This in turn led to the discovery that an error in Oracle software coding had resulted in the “widespread transmission” of incorrect VUIDs from new EHR sites to legacy EHR sites, the OIG found. VA leaders and HCPs were notified of the potential clinical impact and were given specific instructions on how to mitigate the issue. They were asked to “please share widely.”
On top of that, days later, patient safety managers across the Veterans Health Administration (VHA) were told that drug-to-drug interactions, duplicate medication orders, and allergy checks were not functioning as expected, and they too were provided with remedial actions.
Oracle applied a successful software patch on in April 2023, to ensure accurate VUIDs were attached to all mail order pharmacy–processed prescriptions from that date forward. However, the OIG learned the incorrect VUIDs sent from new EHR sites and stored in the Health Data Repository from as far back as October 2020 had not been corrected. Case told the subcommittee that on November 29, 2023, the VHA Pharmacy Council reported withdrawing a request for Oracle to send corrected medication VUID data to the Health Data Repository, on the presumption that remaining inaccurate VUIDs would expire in early April 2024, and the data would be corrected at that time.
The OIG is concerned, Case said, that patient medication data remains inaccurate almost a year after VA learned of the issue. The mail order pharmacy-related data generated from approximately 120,000 patients served by new EHR sites are still incorrect. These patients face an ongoing risk of an adverse medication-related event if they receive care and medications from a VA medical center using the legacy EHR system.
The OIG also learned of other problems associated with transmission of medication and allergy information, which could have consequences such as:
- Patient medications being discontinued or stopped by new HCPs using Cerner that appear in ViSTA as active and current prescriptions;
- Allergy-warning messages not appearing when intended or inappropriately appearing for the wrong medication;
- Duplicate medication order checks not appearing when intended or inappropriately appearing for the wrong drug;
- Patient active medication lists having incomplete or inaccurate information, such as missing prescriptions, duplicate prescriptions, or incorrect medication order statuses.
The OIG warned VHA employees about the risks, although it wasn’t possible to determine who might actually be at risk. A VHA leader told the OIG that all patients who have been prescribed any medications or have medication allergies documented at a at a Cerner site are at risk. That could mean as many as 250,000 patients: As of September 2023, approximately 190,000 patients had a medication prescribed and 126,000 had an allergy documented at a new EHR site.
Case Example
Not surprisingly, “the OIG is not confident in [EHRM-Integration Office] leaders’ oversight and control of the new systems’ Health Data Repository interface programming,” Case said. He cited the case of a patient with posttraumatic stress disorder and traumatic brain injury with adrenal insufficiency. Four days prior to admission, a ViSTA site pharmacist used the EHR to perform a medication reconciliation for the patient. The data available did not include the patient’s most recent prednisone prescription, which had been ordered by an HCP at a facility using Cerner.
A nurse practitioner performed another reconciliation when the patient was admitted to the residential program, but the patient was unsure of all their medications. Because the most recent prednisone prescription was not visible in ViSTA, the prednisone appeared to have been completed at least 3 months prior to admission and was therefore not prescribed in the admission medication orders.
Five days into the residential program, the patient began exhibiting unusual behaviors associated with the lack of prednisone. The patient realized they needed more prednisone, but the nurse explained there was no prednisone on the patient’s medication list. Eventually, the patient found the active prednisone order on their personal cell phone and was transferred to a local emergency department for care.
Work Arounds
The VHA’s efforts to forestall or mitigate system errors have in some cases had a cascade effect. For example, HCPs must essentially back up what the automated software is intended to do, with “complex, time-consuming” multistep manual safety checks when prescribing new medications for patients previously cared for at a Cerner site. The OIG is concerned that this increased vigilance is “unsustainable” by pharmacists and frontline staff and could lead to burnout and medication-related patient safety events. After the new EHR launched, the OIG found, burnout symptoms for pharmacy staff increased. Nonetheless, Case told the committee, OIG staff “have observed [employees’] unwavering commitment to prioritizing the care of patients while mitigating implementation challenges.”
EHR-related workload burdens have necessitated other adjustments. Columbus, for instance, hired 9 full-time clinical pharmacists—a 62% staffing increase—to help reduce the backlog. Pharmacy leaders created approximately 29 additional work-arounds to support pharmacy staff and prevent delays. Facility pharmacy leaders also developed approximately 25 educational materials, such as tip sheets, reference guides, and job aids. The OIG’s concern—apart from the overwhelming amount of information for staff to implement—is that such prophylactic measures may in fact give rise to inconsistent practices, which increase risks to patient safety.
Committed to Working With the VA
Mike Sicilia, executive vice president of Oracle Corporation, told lawmakers in the hearing, “After the initial deployments, it became clear that the pharmacy system needed to be enhanced to better meet VA’s needs. To that end, in August 2022, shortly after Oracle completed its acquisition of Cerner, VA contracted with us for seven enhancements that overall would adapt the pharmacy system to a more bidirectional system between VA providers placing prescription orders and VA pharmacists fulfilling and dispensing them.” Those enhancements are all live for VA providers and pharmacists to use now, he said, except for one that is undergoing additional testing.
He added, “As with any healthcare technology system, there is a need for continuous improvements but that does not mean the system is not safe and effective in its current state. Oracle is committed to working with VA … throughout the reset period to identify workflows and other items that can be simplified or streamlined to improve the overall user and pharmacy experience.”
Standardizing workflows and ensuring training and communications to pharmacists about the latest updates will discourage use of work-arounds, Sicilia said, and “help with improving morale and satisfaction with the system.” During a visit in early February by VA and the Oracle team to the Lovell Federal Health Care Center in North Chicago, “feedback from pharmacists was positive about the training and readiness for using the new pharmacy system.”
The backlog, at least, may be resolved. Sicilia said on average more than 215,000 outpatient prescriptions are being filled each month. “The current live sites do not have a backlog in filling prescriptions. Recent data from this month show that three of the five live sites have zero prescriptions waiting to be processed that are older than seven days. The two other live sites have an average of two prescriptions older than seven days,” he said.
Although Oracle Health has since resolved some of the identified issues, the OIG is concerned that the new EHR will continue to be deployed at medical facilities despite “myriad” as-yet unresolved issues related to inaccurate medication ordering, reconciliation, and dispensing. The VHA has paused Cerner deployments multiple times.
“It is unclear whether identified problems are being adequately resolved before additional deployments,” Case said. “There is also the question of whether there is sufficient transparency and communication among EHRM-IO, VHA and facility leaders, VA leaders, and Oracle Health needed for quality control and critical coordination. Trust in VA is also dependent on patients being fully and quickly advised when issues affecting them are identified and addressed. As VA moves toward its deployment next month at a complex facility jointly operated with the Department of Defense, transparency, communication, and program management will be essential to getting it right. Failures in these areas risk cascading problems.”
Will the new US Department of Veterans Affairs (VA) pharmacy software be safe and effective? That was the topic when David Case, the VA Deputy Inspector General, spoke in the US House of Representatives Veterans Affairs Committee technology modernization subcommittee hearing on February 15.
Questions like that have dogged the project since 2018, when the VA began rolling out the Oracle Cerner electronic health record (EHR) system as the successor to ViSTA.
The Oracle system has been beset by one glitch after another since its arrival. And in that time, Case said, the VA Office of Inspector General (OIG) has been engaging with VA employees at sites in Washington, Oregon, Ohio, Illinois, and other locations where the modernization program has been piloted.
The most recent OIG investigation of pharmacy-related patient safety issues began with a review of an allegation of a prescription backlog at Columbus, Ohio, where the system went live on April 30, 2022. The OIG found that facility leaders took “timely and sustainable steps” to manage that issue. However, other unresolved patient safety issues came to light, such as medication inaccuracies, inaccurate medication data, and insufficient staffing. The OIG also found staff were creating “numerous work arounds” to provide patient care, and that the volume of staff educational materials for pharmacy-related functions was “overwhelming.”
Those problems were just the latest in a long queue. In May 2021, after the first VA deployment of the new EHR at the Mann-Grandstaff VA Medical Center in Spokane, Washington, a pharmacy patient safety team under the VA National Center for Patient Safety (NCPS) also had identified patient safety issues and “multiple” concerns regarding the system’s usability. For example, updates to a patient’s active medication list were not routinely reflected at the patient’s next appointment. Despite knowing about such challenges, Case noted in his report, VA leaders deployed the new EHR at 4 more VA medical centers.
Cerner/ViSTA Communication
One major cause of the current problems is the way the systems “talk” to each other. EHR information is communicated between VHA facilities through channels that include the Joint Longitudinal Viewer (JLV) and the Health Data Repository, which stores patient-specific clinical information from both the legacy and the new EHR systems. The JLV application allows clinicians to access a read only version of a patient’s EHR from both systems.
Every medication used in VHA has a VA Unique Identifier (VUID). When a patient is prescribed a medication at a new EHR site, that medication’s VUID is sent to the Health Data Repository. If that patient seeks care from a legacy health care practitioner (HCP), and that HCP enters a medication order, a software interface accesses the VUID from the Health Data Repository to verify that the medication being prescribed is safe and compatible with the medications and allergies previously documented in the patient’s record.
However, on March 31, 2023, staff from a ViSTA site found an incorrect medication order when prescribing a new medication to a patient who had received care and medications at a new EHR site. This in turn led to the discovery that an error in Oracle software coding had resulted in the “widespread transmission” of incorrect VUIDs from new EHR sites to legacy EHR sites, the OIG found. VA leaders and HCPs were notified of the potential clinical impact and were given specific instructions on how to mitigate the issue. They were asked to “please share widely.”
On top of that, days later, patient safety managers across the Veterans Health Administration (VHA) were told that drug-to-drug interactions, duplicate medication orders, and allergy checks were not functioning as expected, and they too were provided with remedial actions.
Oracle applied a successful software patch on in April 2023, to ensure accurate VUIDs were attached to all mail order pharmacy–processed prescriptions from that date forward. However, the OIG learned the incorrect VUIDs sent from new EHR sites and stored in the Health Data Repository from as far back as October 2020 had not been corrected. Case told the subcommittee that on November 29, 2023, the VHA Pharmacy Council reported withdrawing a request for Oracle to send corrected medication VUID data to the Health Data Repository, on the presumption that remaining inaccurate VUIDs would expire in early April 2024, and the data would be corrected at that time.
The OIG is concerned, Case said, that patient medication data remains inaccurate almost a year after VA learned of the issue. The mail order pharmacy-related data generated from approximately 120,000 patients served by new EHR sites are still incorrect. These patients face an ongoing risk of an adverse medication-related event if they receive care and medications from a VA medical center using the legacy EHR system.
The OIG also learned of other problems associated with transmission of medication and allergy information, which could have consequences such as:
- Patient medications being discontinued or stopped by new HCPs using Cerner that appear in ViSTA as active and current prescriptions;
- Allergy-warning messages not appearing when intended or inappropriately appearing for the wrong medication;
- Duplicate medication order checks not appearing when intended or inappropriately appearing for the wrong drug;
- Patient active medication lists having incomplete or inaccurate information, such as missing prescriptions, duplicate prescriptions, or incorrect medication order statuses.
The OIG warned VHA employees about the risks, although it wasn’t possible to determine who might actually be at risk. A VHA leader told the OIG that all patients who have been prescribed any medications or have medication allergies documented at a at a Cerner site are at risk. That could mean as many as 250,000 patients: As of September 2023, approximately 190,000 patients had a medication prescribed and 126,000 had an allergy documented at a new EHR site.
Case Example
Not surprisingly, “the OIG is not confident in [EHRM-Integration Office] leaders’ oversight and control of the new systems’ Health Data Repository interface programming,” Case said. He cited the case of a patient with posttraumatic stress disorder and traumatic brain injury with adrenal insufficiency. Four days prior to admission, a ViSTA site pharmacist used the EHR to perform a medication reconciliation for the patient. The data available did not include the patient’s most recent prednisone prescription, which had been ordered by an HCP at a facility using Cerner.
A nurse practitioner performed another reconciliation when the patient was admitted to the residential program, but the patient was unsure of all their medications. Because the most recent prednisone prescription was not visible in ViSTA, the prednisone appeared to have been completed at least 3 months prior to admission and was therefore not prescribed in the admission medication orders.
Five days into the residential program, the patient began exhibiting unusual behaviors associated with the lack of prednisone. The patient realized they needed more prednisone, but the nurse explained there was no prednisone on the patient’s medication list. Eventually, the patient found the active prednisone order on their personal cell phone and was transferred to a local emergency department for care.
Work Arounds
The VHA’s efforts to forestall or mitigate system errors have in some cases had a cascade effect. For example, HCPs must essentially back up what the automated software is intended to do, with “complex, time-consuming” multistep manual safety checks when prescribing new medications for patients previously cared for at a Cerner site. The OIG is concerned that this increased vigilance is “unsustainable” by pharmacists and frontline staff and could lead to burnout and medication-related patient safety events. After the new EHR launched, the OIG found, burnout symptoms for pharmacy staff increased. Nonetheless, Case told the committee, OIG staff “have observed [employees’] unwavering commitment to prioritizing the care of patients while mitigating implementation challenges.”
EHR-related workload burdens have necessitated other adjustments. Columbus, for instance, hired 9 full-time clinical pharmacists—a 62% staffing increase—to help reduce the backlog. Pharmacy leaders created approximately 29 additional work-arounds to support pharmacy staff and prevent delays. Facility pharmacy leaders also developed approximately 25 educational materials, such as tip sheets, reference guides, and job aids. The OIG’s concern—apart from the overwhelming amount of information for staff to implement—is that such prophylactic measures may in fact give rise to inconsistent practices, which increase risks to patient safety.
Committed to Working With the VA
Mike Sicilia, executive vice president of Oracle Corporation, told lawmakers in the hearing, “After the initial deployments, it became clear that the pharmacy system needed to be enhanced to better meet VA’s needs. To that end, in August 2022, shortly after Oracle completed its acquisition of Cerner, VA contracted with us for seven enhancements that overall would adapt the pharmacy system to a more bidirectional system between VA providers placing prescription orders and VA pharmacists fulfilling and dispensing them.” Those enhancements are all live for VA providers and pharmacists to use now, he said, except for one that is undergoing additional testing.
He added, “As with any healthcare technology system, there is a need for continuous improvements but that does not mean the system is not safe and effective in its current state. Oracle is committed to working with VA … throughout the reset period to identify workflows and other items that can be simplified or streamlined to improve the overall user and pharmacy experience.”
Standardizing workflows and ensuring training and communications to pharmacists about the latest updates will discourage use of work-arounds, Sicilia said, and “help with improving morale and satisfaction with the system.” During a visit in early February by VA and the Oracle team to the Lovell Federal Health Care Center in North Chicago, “feedback from pharmacists was positive about the training and readiness for using the new pharmacy system.”
The backlog, at least, may be resolved. Sicilia said on average more than 215,000 outpatient prescriptions are being filled each month. “The current live sites do not have a backlog in filling prescriptions. Recent data from this month show that three of the five live sites have zero prescriptions waiting to be processed that are older than seven days. The two other live sites have an average of two prescriptions older than seven days,” he said.
Although Oracle Health has since resolved some of the identified issues, the OIG is concerned that the new EHR will continue to be deployed at medical facilities despite “myriad” as-yet unresolved issues related to inaccurate medication ordering, reconciliation, and dispensing. The VHA has paused Cerner deployments multiple times.
“It is unclear whether identified problems are being adequately resolved before additional deployments,” Case said. “There is also the question of whether there is sufficient transparency and communication among EHRM-IO, VHA and facility leaders, VA leaders, and Oracle Health needed for quality control and critical coordination. Trust in VA is also dependent on patients being fully and quickly advised when issues affecting them are identified and addressed. As VA moves toward its deployment next month at a complex facility jointly operated with the Department of Defense, transparency, communication, and program management will be essential to getting it right. Failures in these areas risk cascading problems.”
Implementing Trustworthy AI in VA High Reliability Health Care Organizations
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
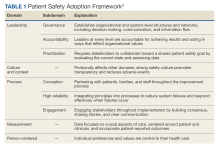
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
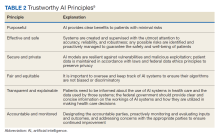
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
Artificial intelligence (AI) has lagged in health care but has considerable potential to improve quality, safety, clinician experience, and access to care. It is being tested in areas like billing, hospital operations, and preventing adverse events (eg, sepsis mortality) with some early success. However, there are still many barriers preventing the widespread use of AI, such as data problems, mismatched rewards, and workplace obstacles. Innovative projects, partnerships, better rewards, and more investment could remove barriers. Implemented reliably and safely, AI can add to what clinicians know, help them work faster, cut costs, and, most importantly, improve patient care.1
AI can potentially bring several clinical benefits, such as reducing the administrative strain on clinicians and granting them more time for direct patient care. It can also improve diagnostic accuracy by analyzing patient data and diagnostic images, providing differential diagnoses, and increasing access to care by providing medical information and essential online services to patients.2
High Reliability Organizations
High reliability health care organizations have considerable experience safely launching new programs. For example, the Patient Safety Adoption Framework gives practical tips for smoothly rolling out safety initiatives (Table 1). Developed with experts and diverse views, this framework has 5 key areas: leadership, culture and context, process, measurement, and person-centeredness. These address adoption problems, guide leaders step-by-step, and focus on leadership buy-in, safety culture, cooperation, and local customization. Checklists and tools make it systematic to go from ideas to action on patient safety.3
Leadership involves establishing organizational commitment behind new safety programs. This visible commitment signals importance and priorities to others. Leaders model desired behaviors and language around safety, allocate resources, remove obstacles, and keep initiatives energized over time through consistent messaging.4 Culture and context recognizes that safety culture differs across units and facilities. Local input tailors programs to fit and examines strengths to build on, like psychological safety. Surveys gauge the existing culture and its need for change. Process details how to plan, design, test, implement, and improve new safety practices and provides a phased roadmap from idea to results. Measurement collects data to drive improvement and show impact. Metrics track progress and allow benchmarking. Person-centeredness puts patients first in safety efforts through participation, education, and transparency.
The Veterans Health Administration piloted a comprehensive high reliability hospital (HRH) model. Over 3 years, the Veterans Health Administration focused on leadership, culture, and process improvement at a hospital. After initiating the model, the pilot hospital improved its safety culture, reported more minor safety issues, and reduced deaths and complications better than other hospitals. The high-reliability approach successfully instilled principles and improved culture and outcomes. The HRH model is set to be expanded to 18 more US Department of Veterans Affairs (VA) sites for further evaluation across diverse settings.5
Trustworthy AI Framework
AI systems are growing more powerful and widespread, including in health care. Unfortunately, irresponsible AI can introduce new harm. ChatGPT and other large language models, for example, sometimes are known to provide erroneous information in a compelling way. Clinicians and patients who use such programs can act on such information, which would lead to unforeseen negative consequences. Several frameworks on ethical AI have come from governmental groups.6-9 In 2023, the VA National AI Institute suggested a Trustworthy AI Framework based on core principles tailored for federal health care. The framework has 6 key principles: purposeful, effective and safe, secure and private, fair and equitable, transparent and explainable, and accountable and monitored (Table 2).10
First, AI must clearly help veterans while minimizing risks. To ensure purpose, the VA will assess patient and clinician needs and design AI that targets meaningful problems to avoid scope creep or feature bloat. For example, adding new features to the AI software after release can clutter and complicate the interface, making it difficult to use. Rigorous testing will confirm that AI meets intent prior to deployment. Second, AI is designed and checked for effectiveness, safety, and reliability. The VA pledges to monitor AI’s impact to ensure it performs as expected without unintended consequences. Algorithms will be stress tested across representative datasets and approval processes will screen for safety issues. Third, AI models are secured from vulnerabilities and misuse. Technical controls will prevent unauthorized access or changes to AI systems. Audits will check for appropriate internal usage per policies. Continual patches and upgrades will maintain security. Fourth, the VA manages AI for fairness, avoiding bias. They will proactively assess datasets and algorithms for potential biases based on protected attributes like race, gender, or age. Biased outputs will be addressed through techniques such as data augmentation, reweighting, and algorithm tweaks. Fifth, transparency explains AI’s role in care. Documentation will detail an AI system’s data sources, methodology, testing, limitations, and integration with clinical workflows. Clinicians and patients will receive education on interpreting AI outputs. Finally, the VA pledges to closely monitor AI systems to sustain trust. The VA will establish oversight processes to quickly identify any declines in reliability or unfair impacts on subgroups. AI models will be retrained as needed based on incoming data patterns.
Each Trustworthy AI Framework principle connects to others in existing frameworks. The purpose principle aligns with human-centric AI focused on benefits. Effectiveness and safety link to technical robustness and risk management principles. Security maps to privacy protection principles. Fairness connects to principles of avoiding bias and discrimination. Transparency corresponds with accountable and explainable AI. Monitoring and accountability tie back to governance principles. Overall, the VA framework aims to guide ethical AI based on context. It offers a model for managing risks and building trust in health care AI.
Combining VA principles with high-reliability safety principles can ensure that AI benefits veterans. The leadership and culture aspects will drive commitment to trustworthy AI practices. Leaders will communicate the importance of responsible AI through words and actions. Culture surveys can assess baseline awareness of AI ethics issues to target education. AI security and fairness will be emphasized as safety critical. The process aspect will institute policies and procedures to uphold AI principles through the project lifecycle. For example, structured testing processes will validate safety. Measurement will collect data on principles like transparency and fairness. Dashboards can track metrics like explainability and biases. A patient-centered approach will incorporate veteran perspectives on AI through participatory design and advisory councils. They can give input on AI explainability and potential biases based on their diverse backgrounds.
Conclusions
Joint principles will lead to successful AI that improves care while proactively managing risks. Involve leaders to stress the necessity of eliminating biases. Build security into the AI development process. Co-design AI transparency features with end users. Closely monitor the impact of AI across safety, fairness, and other principles. Adhering to both Trustworthy AI and high reliability organizations principles will earn veterans’ confidence. Health care organizations like the VA can integrate ethical AI safely via established frameworks. With responsible design and implementation, AI’s potential to enhance care quality, safety, and access can be realized.
Acknowledgments
We would like to acknowledge Joshua Mueller, Theo Tiffney, John Zachary, and Gil Alterovitz for their excellent work creating the VA Trustworthy Principles. This material is the result of work supported by resources and the use of facilities at the James A. Haley Veterans’ Hospital.
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
1. Sahni NR, Carrus B. Artificial intelligence in U.S. health care delivery. N Engl J Med. 2023;389(4):348-358. doi:10.1056/NEJMra2204673
2. Borkowski AA, Jakey CE, Mastorides SM, et al. Applications of ChatGPT and large language models in medicine and health care: benefits and pitfalls. Fed Pract. 2023;40(6):170-173. doi:10.12788/fp.0386
3. Moyal-Smith R, Margo J, Maloney FL, et al. The patient safety adoption framework: a practical framework to bridge the know-do gap. J Patient Saf. 2023;19(4):243-248. doi:10.1097/PTS.0000000000001118
4. Isaacks DB, Anderson TM, Moore SC, Patterson W, Govindan S. High reliability organization principles improve VA workplace burnout: the Truman THRIVE2 model. Am J Med Qual. 2021;36(6):422-428. doi:10.1097/01.JMQ.0000735516.35323.97
5. Sculli GL, Pendley-Louis R, Neily J, et al. A high-reliability organization framework for health care: a multiyear implementation strategy and associated outcomes. J Patient Saf. 2022;18(1):64-70. doi:10.1097/PTS.0000000000000788
6. National Institute of Standards and Technology. AI risk management framework. Accessed January 2, 2024. https://www.nist.gov/itl/ai-risk-management-framework
7. Executive Office of the President, Office of Science and Technology Policy. Blueprint for an AI Bill of Rights. Accessed January 11, 2024. https://www.whitehouse.gov/ostp/ai-bill-of-rights
8. Executive Office of the President. Executive Order 13960: promoting the use of trustworthy artificial intelligence in the federal government. Fed Regist. 2020;89(236):78939-78943.
9. Biden JR. Executive Order on the safe, secure, and trustworthy development and use of artificial intelligence. Published October 30, 2023. Accessed January 11, 2024. https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/
10. US Department of Veterans Affairs. Trustworthy AI. Accessed January 11, 2024. https://department.va.gov/ai/trustworthy/
Age-Friendly Health Systems and Meeting the Principles of High Reliability Organizations in the VHA
The Veterans Health Administration (VHA) is the largest integrated health care system in the US, providing care to more than 9 million enrolled veterans at 1298 facilities.1 In February 2019, the VHA identified key action steps to become a high reliability organization (HRO), transforming how employees think about patient safety and care quality.2 The VHA is also working toward becoming the largest age-friendly health system in the US to be recognized by the Institute for Healthcare Improvement (IHI) for its commitment to providing care guided by the 4Ms (what matters, medication, mentation, and mobility), causing no harm, and aligning care with what matters to older veterans.3 In this article, we describe how the Age-Friendly Health Systems (AFHS) movement supports the culture shift observed in HROs.
Age-Friendly Veteran Care
By 2060, the US population of adults aged ≥ 65 years is projected to increase to about 95 million.3 In the VHA, nearly half of veteran enrollees are aged ≥ 65 years, necessitating evidence-based models of care, such as the 4Ms, to meet their complex care needs.3 Historically, the VHA has been a leader in caring for older adults, recognizing the value of age-friendly care for veterans.4 In 1975, the VHA established the Geriatric Research, Education, and Clinical Centers (GRECCs) to serve as catalysts for developing, implementing, and refining enduring models of geriatric care.4 For 5 decades, GRECCs have driven innovations related to the 4Ms.
The VHA is well positioned to be a leader in the AFHS movement, building on decades of GRECC innovations and geriatric programs that align with the 4Ms and providing specialized geriatric training for health care professionals to expand age-friendly care to new settings and health systems.4 The AFHS movement organizes the 4Ms into a simple framework for frontline staff, and the VHA has recently begun tracking 4Ms care in the electronic health record (EHR) to facilitate evaluation and continuous improvement.
AFHS use the 4Ms as a framework to be implemented in every care setting, from the emergency department to inpatient units, outpatient settings, and postacute and long-term care. By assessing and acting on each M and practicing the 4Ms collectively, all members of the care team work to improve health outcomes and prevent avoidable harm.5
The 4Ms
What matters, is the driver of this person-centered approach. Any member of the care team may initiate a what matters conversation with the older adult to understand their personal values, health goals, and care preferences. When compared with usual care, care aligned with the older adult’s health priorities has been shown to decrease the use of high-risk medications and reduce treatment burden.6 The VHA has adopted Whole Health principles of care and the Patient Priorities Care approach to identify and support what matters to veterans.7,8
Addressing polypharmacy and identifying and deprescribing potentially inappropriate medications are essential in preventing adverse drug events, drug-drug interactions, and medication nonadherence.9 In the VHA, VIONE (Vital, Important, Optional, Not indicated, Every medication has an indication) is a rapidly expanding medication deprescribing program that exemplifies HRO principles.9 VIONE provides medication management that supports shared decision making, reducing risk and improving patient safety and quality of life.9 As of June 2023, > 600,000 unique veterans have benefited from VIONE, with an average of 2.2 medications deprescribed per patient with an annual cost avoidance of > $100 million.10
Assessing and acting on mentation includes preventing, identifying, and managing depression and dementia in outpatient settings and delirium in hospital and long-term care settings.5 There are many tools and clinical reminders available in the EHR so that interdisciplinary teams can document changes to mentation and identify opportunities for continuous improvement.
Closely aligned with mentation is mobility, with evidence suggesting that regular physical activity reduces the risk of falls (preventing associated complications), maintains physical functioning, and lowers the risk of cognitive impairment and depression.5 Ensuring early, frequent, and safe mobility helps patients achieve better health outcomes and prevent injury.5 Mobility programs within the VHA include the STRIDE program for the inpatient setting and Gerofit for outpatient settings.11,12
HRO Principles
An HRO is a complex environment of care that experiences fewer than anticipated accidents or adverse events by (1) establishing trust among leaders and staff by balancing individual accountability with systems thinking; (2) empowering staff to lead continuous process improvements; and (3) creating an environment where employees feel safe to report harm or near misses, focusing on the reasons errors occur.13 The work of AFHS incorporates HRO principles with an emphasis on 3 elements. First, it involves interactive systems and processes needed to support 4Ms care across care settings. Second, AFHS acknowledge the complexity of age-friendly work and deference to the expertise of interdisciplinary team members. Finally, AFHS are committed to resilience by overcoming failures and challenges to implementation and long-term sustainment as a standard of practice.
Case study
The names and details in this case have been modified to protect patient privacy. It is representative of many Community Living Centers (CLCs) involved in AFHS that work to create a safe, person-centered environment for veterans.
In a CLC team workroom, 2 nurses were discussing a long-term care resident. The nurses approached the attending physician and explained that they were worried about Sgt Johnson, who seemed depressed and sometimes combative. They had noticed a change in his behavior when they helped him clean up after an episode of incontinence and were concerned that he would try to get out of bed on his own and fall. The attending physician thanked them for sharing their concerns. Sgt Johnson was a retired Army veteran who had a long, decorated military career. His chronic health conditions had led to muscle weakness, and he fell and broke a hip before this admission. He had an uneventful hip replacement but was showing signs of depression due to his limited mobility, loss of independence, and inability to live at home without additional support.
The attending physician knocked on the door of his room, sat down next to the bed, and asked, “How are you feeling today?” Sgt Johnson tersely replied, “About the same.” The physician asked, “Sgt Johnson, what matters most to you related to your recovery? What is important to you?” Sgt Johnson responded, “Feeling like a man!” The doctor replied, “So what makes you feel ‘not like a man’?” The Sgt replied, “Having to be cleaned up by the nurses and not being able to use the toilet on my own.” The physician surmised that his decline in physical functioning had a connection to his worsening depression and combativeness and said to the Sgt, “Let’s get the team together and work out a plan to get you strong enough to use a bedside commode by yourself. Let’s make that the first goal in our plan to get you back to using the toilet independently. Can you work with us on that?” He smiled and said, “Sir, yes Sir!”
At the weekly interdisciplinary team meeting, the team discussed Sgt Johnson’s wishes and the nurses’ safety concerns. The physician reported to the team what mattered to the veteran. The nurses arranged for a bedside commode and supplies to be placed in his room, encouraged and assisted him, and provided a privacy screen. The physical therapist continued to support his mobility needs, concentrating on transfers, small steps like standing and turning with a walker to get in position to use the bedside commode, and later the bathroom toilet. The psychologist addressed what matters to Sgt Johnson and his mentation, health goals, and coping strategies. The social worker provided support and counseling for the veteran and his family. The pharmacist checked his medications to be sure that none were affecting his gastrointestinal tract and his ability to move safely and do what matters to him. Knowing what mattered to Sgt Johnson was the driver of the interdisciplinary care plan to provide 4Ms care.
The team worked collaboratively with the veteran to develop and set attainable goals around toileting and regaining his dignity. This improved his overall recovery. As Sgt Johnson became more independent, his mood gradually improved and he began to participate in other activities and interact with other residents on the unit, and he did not experience any falls. By addressing the 4Ms, the interdisciplinary team coordinated efforts to provide high-quality, person-centered care. They built trust with the veteran, shared accountability, and followed HRO principles to keep the veteran safe.
Becoming an Age-Friendly HRO
Becoming an HRO is a dynamic, ever-changing process to maintain high standards, improve care quality, and cause no harm. There are 3 pillars and 5 principles that guide an HRO. The pillars are critical areas of focus and include leadership commitment, culture of safety, and continuous process improvement.14 The first of 5 HRO principles is sensitivity to operations. This is defined as an awareness of how processes and systems impact the entire organization, the downstream impact.15 Focusing on the 4Ms helps develop the capability of frontline staff to provide high-quality care for older adults while ensuring that processes are in place to support the work. The 4Ms provide an efficient way to organize interdisciplinary team meetings, provide warm handoffs using Situation-Background-Assessment-Recommendation, and standardize documentation. Involvement in the AFHS movement improves communication, care quality, and patient and staff satisfaction to meet this HRO principle.15
The second HRO principle, reluctance to simplify, ensures that direct care staff and leaders delve further into issues to find solutions.15 AFHS use the Plan-Do-Study-Act cycle to put the 4Ms into practice; this cycle helps teams test small increments of change, study their performance, and act to ensure that all 4Ms are being practiced as a set. AFHS teams are encouraged to review at least 3 months of data after implementation of the 4Ms, working to find solutions if there are gaps or issues identified.
The third principle, preoccupation with failure, refers to shared attentiveness—being prepared for the unexpected and learning from mistakes.15 The entire AFHS team shares responsibility for providing 4Ms care, where staff are empowered to report any safety concerns or close calls. The fourth principle of deference to expertise includes listening to staff who have the most knowledge for the task at hand, which aligns with the collaborative interdisciplinary teamwork of age-friendly teams.15
The final HRO principle, commitment to resilience, includes continuous learning, interdisciplinary team training, and sharing of lessons learned.15 Although IHI offers 2 levels of AFHS recognition, teams are continuously learning to improve and sustain care beyond level 2, Committed to Care Excellence recognition.16
The Table shows the VHA’s AFHS implementation strategies and the HRO principles adapted from the Joint Commission’s High Reliability Health Care Maturity Model and the IHI’s Framework for Safe, Reliable, and Effective Care. The VHA is developing a national dashboard to capture age-friendly processes and health outcome measures that address patient safety and care quality.
Conclusions
AFHS empowers VHA teams to honor veterans’ care preferences and values, supporting their independence, dignity, and quality of life across care settings. The adoption of AFHS brings evidence-based practices to the point of care by addressing common pitfalls in the care of older adults, drawing attention to, and calling for action on inappropriate medication use, physical inactivity, and assessment of the vulnerable brain. The 4Ms also serve as a framework to continuously improve care and cause zero harm, reinforcing HRO pillars and principles across the VHA, and ensuring that older adults reliably receive the evidence-based, high-quality care they deserve.
1. Veterans Health Administration. Providing healthcare for veterans. Updated June 20, 2023. Accessed June 26, 2023. https://www.va.gov/health
2. Veazie S, Peterson K, Bourne D. Evidence brief: implementation of high reliability organization principles. Washington, DC: Evidence Synthesis Program, Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs. VA ESP Project #09-199; 2019. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/esp/high-reliability-org.cfm
3. Church K, Munro S, Shaughnessy M, Clancy C. Age-Friendly Health Systems: improving care for older adults in the Veterans Health Administration. Health Serv Res. 2023;58(suppl 1):5-8. doi:10.1111/1475-6773.14110
4. Farrell TW, Volden TA, Butler JM, et al. Age-friendly care in the Veterans Health Administration: past, present, and future. J Am Geriatr Soc. 2023;71(1):18-25. doi:10.1111/jgs.18070
5. Mate K, Fulmer T, Pelton L, et al. Evidence for the 4Ms: interactions and outcomes across the care continuum. J Aging Health. 2021;33(7-8):469-481. doi:10.1177/0898264321991658
6. Tinetti ME, Naik AD, Dindo L, et al. Association of patient priorities-aligned decision-making with patient outcomes and ambulatory health care burden among older adults with multiple chronic conditions: A nonrandomized clinical trial. JAMA Intern Med. 2019;179(12):1688-1697. doi:10.1001/jamainternmed.2019.4235
7. US Department of Veterans Affairs. What is whole health? Updated: October 31, 2023. November 30, 2023. https://www.va.gov/wholehealth
8. Patient Priorities Care. Updated 2019. Accessed November 30, 2023. https://patientprioritiescare.org
9. Battar S, Watson Dickerson KR, Sedgwick C, Cmelik T. Understanding principles of high reliability organizations through the eyes of VIONE: a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
10. VA Diffusion Marketplace. VIONE- medication optimization and polypharmacy reduction initiative. Accessed November 30, 2023. https://marketplace.va.gov/innovations/vione
11. US Department of Veterans Affairs, Office of Research and Development. STRIDE program to keep hospitalized veterans mobile. Updated November 6, 2018. Accessed November 30, 2023. https://www.research.va.gov/research_in_action/STRIDE-program-to-keep-hospitalized-Veterans-mobile.cfm
12. US Department of Veterans Affairs, VA Geriatrics and Extended Care. Gerofit: a program promoting exercise and health for older veterans. Updated August 2, 2023. Accessed November 30, 2023. https://www.va.gov/GERIATRICS/pages/gerofit_Home.asp
13. US Department of Veterans Affairs, Health Services Research and Development. VHA’s vision for a high reliability organization. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-1
14. US Department of Veterans Affairs, Health Services Research and Development. Three HRO evaluation priorities. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-2
15. Oster CA, Deakins S. Practical application of high-reliability principles in healthcare to optimize quality and safety outcomes. J Nurs Adm. 2018;48(1):50-55. doi:10.1097/NNA.0000000000000570
16. Institute for Healthcare Improvement. Age-Friendly Health Systems recognitions. Accessed November 30, 2023. https://www.ihi.org/Engage/Initiatives/Age-Friendly-Health-Systems/Pages/Recognition.aspx
The Veterans Health Administration (VHA) is the largest integrated health care system in the US, providing care to more than 9 million enrolled veterans at 1298 facilities.1 In February 2019, the VHA identified key action steps to become a high reliability organization (HRO), transforming how employees think about patient safety and care quality.2 The VHA is also working toward becoming the largest age-friendly health system in the US to be recognized by the Institute for Healthcare Improvement (IHI) for its commitment to providing care guided by the 4Ms (what matters, medication, mentation, and mobility), causing no harm, and aligning care with what matters to older veterans.3 In this article, we describe how the Age-Friendly Health Systems (AFHS) movement supports the culture shift observed in HROs.
Age-Friendly Veteran Care
By 2060, the US population of adults aged ≥ 65 years is projected to increase to about 95 million.3 In the VHA, nearly half of veteran enrollees are aged ≥ 65 years, necessitating evidence-based models of care, such as the 4Ms, to meet their complex care needs.3 Historically, the VHA has been a leader in caring for older adults, recognizing the value of age-friendly care for veterans.4 In 1975, the VHA established the Geriatric Research, Education, and Clinical Centers (GRECCs) to serve as catalysts for developing, implementing, and refining enduring models of geriatric care.4 For 5 decades, GRECCs have driven innovations related to the 4Ms.
The VHA is well positioned to be a leader in the AFHS movement, building on decades of GRECC innovations and geriatric programs that align with the 4Ms and providing specialized geriatric training for health care professionals to expand age-friendly care to new settings and health systems.4 The AFHS movement organizes the 4Ms into a simple framework for frontline staff, and the VHA has recently begun tracking 4Ms care in the electronic health record (EHR) to facilitate evaluation and continuous improvement.
AFHS use the 4Ms as a framework to be implemented in every care setting, from the emergency department to inpatient units, outpatient settings, and postacute and long-term care. By assessing and acting on each M and practicing the 4Ms collectively, all members of the care team work to improve health outcomes and prevent avoidable harm.5
The 4Ms
What matters, is the driver of this person-centered approach. Any member of the care team may initiate a what matters conversation with the older adult to understand their personal values, health goals, and care preferences. When compared with usual care, care aligned with the older adult’s health priorities has been shown to decrease the use of high-risk medications and reduce treatment burden.6 The VHA has adopted Whole Health principles of care and the Patient Priorities Care approach to identify and support what matters to veterans.7,8
Addressing polypharmacy and identifying and deprescribing potentially inappropriate medications are essential in preventing adverse drug events, drug-drug interactions, and medication nonadherence.9 In the VHA, VIONE (Vital, Important, Optional, Not indicated, Every medication has an indication) is a rapidly expanding medication deprescribing program that exemplifies HRO principles.9 VIONE provides medication management that supports shared decision making, reducing risk and improving patient safety and quality of life.9 As of June 2023, > 600,000 unique veterans have benefited from VIONE, with an average of 2.2 medications deprescribed per patient with an annual cost avoidance of > $100 million.10
Assessing and acting on mentation includes preventing, identifying, and managing depression and dementia in outpatient settings and delirium in hospital and long-term care settings.5 There are many tools and clinical reminders available in the EHR so that interdisciplinary teams can document changes to mentation and identify opportunities for continuous improvement.
Closely aligned with mentation is mobility, with evidence suggesting that regular physical activity reduces the risk of falls (preventing associated complications), maintains physical functioning, and lowers the risk of cognitive impairment and depression.5 Ensuring early, frequent, and safe mobility helps patients achieve better health outcomes and prevent injury.5 Mobility programs within the VHA include the STRIDE program for the inpatient setting and Gerofit for outpatient settings.11,12
HRO Principles
An HRO is a complex environment of care that experiences fewer than anticipated accidents or adverse events by (1) establishing trust among leaders and staff by balancing individual accountability with systems thinking; (2) empowering staff to lead continuous process improvements; and (3) creating an environment where employees feel safe to report harm or near misses, focusing on the reasons errors occur.13 The work of AFHS incorporates HRO principles with an emphasis on 3 elements. First, it involves interactive systems and processes needed to support 4Ms care across care settings. Second, AFHS acknowledge the complexity of age-friendly work and deference to the expertise of interdisciplinary team members. Finally, AFHS are committed to resilience by overcoming failures and challenges to implementation and long-term sustainment as a standard of practice.
Case study
The names and details in this case have been modified to protect patient privacy. It is representative of many Community Living Centers (CLCs) involved in AFHS that work to create a safe, person-centered environment for veterans.
In a CLC team workroom, 2 nurses were discussing a long-term care resident. The nurses approached the attending physician and explained that they were worried about Sgt Johnson, who seemed depressed and sometimes combative. They had noticed a change in his behavior when they helped him clean up after an episode of incontinence and were concerned that he would try to get out of bed on his own and fall. The attending physician thanked them for sharing their concerns. Sgt Johnson was a retired Army veteran who had a long, decorated military career. His chronic health conditions had led to muscle weakness, and he fell and broke a hip before this admission. He had an uneventful hip replacement but was showing signs of depression due to his limited mobility, loss of independence, and inability to live at home without additional support.
The attending physician knocked on the door of his room, sat down next to the bed, and asked, “How are you feeling today?” Sgt Johnson tersely replied, “About the same.” The physician asked, “Sgt Johnson, what matters most to you related to your recovery? What is important to you?” Sgt Johnson responded, “Feeling like a man!” The doctor replied, “So what makes you feel ‘not like a man’?” The Sgt replied, “Having to be cleaned up by the nurses and not being able to use the toilet on my own.” The physician surmised that his decline in physical functioning had a connection to his worsening depression and combativeness and said to the Sgt, “Let’s get the team together and work out a plan to get you strong enough to use a bedside commode by yourself. Let’s make that the first goal in our plan to get you back to using the toilet independently. Can you work with us on that?” He smiled and said, “Sir, yes Sir!”
At the weekly interdisciplinary team meeting, the team discussed Sgt Johnson’s wishes and the nurses’ safety concerns. The physician reported to the team what mattered to the veteran. The nurses arranged for a bedside commode and supplies to be placed in his room, encouraged and assisted him, and provided a privacy screen. The physical therapist continued to support his mobility needs, concentrating on transfers, small steps like standing and turning with a walker to get in position to use the bedside commode, and later the bathroom toilet. The psychologist addressed what matters to Sgt Johnson and his mentation, health goals, and coping strategies. The social worker provided support and counseling for the veteran and his family. The pharmacist checked his medications to be sure that none were affecting his gastrointestinal tract and his ability to move safely and do what matters to him. Knowing what mattered to Sgt Johnson was the driver of the interdisciplinary care plan to provide 4Ms care.
The team worked collaboratively with the veteran to develop and set attainable goals around toileting and regaining his dignity. This improved his overall recovery. As Sgt Johnson became more independent, his mood gradually improved and he began to participate in other activities and interact with other residents on the unit, and he did not experience any falls. By addressing the 4Ms, the interdisciplinary team coordinated efforts to provide high-quality, person-centered care. They built trust with the veteran, shared accountability, and followed HRO principles to keep the veteran safe.
Becoming an Age-Friendly HRO
Becoming an HRO is a dynamic, ever-changing process to maintain high standards, improve care quality, and cause no harm. There are 3 pillars and 5 principles that guide an HRO. The pillars are critical areas of focus and include leadership commitment, culture of safety, and continuous process improvement.14 The first of 5 HRO principles is sensitivity to operations. This is defined as an awareness of how processes and systems impact the entire organization, the downstream impact.15 Focusing on the 4Ms helps develop the capability of frontline staff to provide high-quality care for older adults while ensuring that processes are in place to support the work. The 4Ms provide an efficient way to organize interdisciplinary team meetings, provide warm handoffs using Situation-Background-Assessment-Recommendation, and standardize documentation. Involvement in the AFHS movement improves communication, care quality, and patient and staff satisfaction to meet this HRO principle.15
The second HRO principle, reluctance to simplify, ensures that direct care staff and leaders delve further into issues to find solutions.15 AFHS use the Plan-Do-Study-Act cycle to put the 4Ms into practice; this cycle helps teams test small increments of change, study their performance, and act to ensure that all 4Ms are being practiced as a set. AFHS teams are encouraged to review at least 3 months of data after implementation of the 4Ms, working to find solutions if there are gaps or issues identified.
The third principle, preoccupation with failure, refers to shared attentiveness—being prepared for the unexpected and learning from mistakes.15 The entire AFHS team shares responsibility for providing 4Ms care, where staff are empowered to report any safety concerns or close calls. The fourth principle of deference to expertise includes listening to staff who have the most knowledge for the task at hand, which aligns with the collaborative interdisciplinary teamwork of age-friendly teams.15
The final HRO principle, commitment to resilience, includes continuous learning, interdisciplinary team training, and sharing of lessons learned.15 Although IHI offers 2 levels of AFHS recognition, teams are continuously learning to improve and sustain care beyond level 2, Committed to Care Excellence recognition.16
The Table shows the VHA’s AFHS implementation strategies and the HRO principles adapted from the Joint Commission’s High Reliability Health Care Maturity Model and the IHI’s Framework for Safe, Reliable, and Effective Care. The VHA is developing a national dashboard to capture age-friendly processes and health outcome measures that address patient safety and care quality.
Conclusions
AFHS empowers VHA teams to honor veterans’ care preferences and values, supporting their independence, dignity, and quality of life across care settings. The adoption of AFHS brings evidence-based practices to the point of care by addressing common pitfalls in the care of older adults, drawing attention to, and calling for action on inappropriate medication use, physical inactivity, and assessment of the vulnerable brain. The 4Ms also serve as a framework to continuously improve care and cause zero harm, reinforcing HRO pillars and principles across the VHA, and ensuring that older adults reliably receive the evidence-based, high-quality care they deserve.
The Veterans Health Administration (VHA) is the largest integrated health care system in the US, providing care to more than 9 million enrolled veterans at 1298 facilities.1 In February 2019, the VHA identified key action steps to become a high reliability organization (HRO), transforming how employees think about patient safety and care quality.2 The VHA is also working toward becoming the largest age-friendly health system in the US to be recognized by the Institute for Healthcare Improvement (IHI) for its commitment to providing care guided by the 4Ms (what matters, medication, mentation, and mobility), causing no harm, and aligning care with what matters to older veterans.3 In this article, we describe how the Age-Friendly Health Systems (AFHS) movement supports the culture shift observed in HROs.
Age-Friendly Veteran Care
By 2060, the US population of adults aged ≥ 65 years is projected to increase to about 95 million.3 In the VHA, nearly half of veteran enrollees are aged ≥ 65 years, necessitating evidence-based models of care, such as the 4Ms, to meet their complex care needs.3 Historically, the VHA has been a leader in caring for older adults, recognizing the value of age-friendly care for veterans.4 In 1975, the VHA established the Geriatric Research, Education, and Clinical Centers (GRECCs) to serve as catalysts for developing, implementing, and refining enduring models of geriatric care.4 For 5 decades, GRECCs have driven innovations related to the 4Ms.
The VHA is well positioned to be a leader in the AFHS movement, building on decades of GRECC innovations and geriatric programs that align with the 4Ms and providing specialized geriatric training for health care professionals to expand age-friendly care to new settings and health systems.4 The AFHS movement organizes the 4Ms into a simple framework for frontline staff, and the VHA has recently begun tracking 4Ms care in the electronic health record (EHR) to facilitate evaluation and continuous improvement.
AFHS use the 4Ms as a framework to be implemented in every care setting, from the emergency department to inpatient units, outpatient settings, and postacute and long-term care. By assessing and acting on each M and practicing the 4Ms collectively, all members of the care team work to improve health outcomes and prevent avoidable harm.5
The 4Ms
What matters, is the driver of this person-centered approach. Any member of the care team may initiate a what matters conversation with the older adult to understand their personal values, health goals, and care preferences. When compared with usual care, care aligned with the older adult’s health priorities has been shown to decrease the use of high-risk medications and reduce treatment burden.6 The VHA has adopted Whole Health principles of care and the Patient Priorities Care approach to identify and support what matters to veterans.7,8
Addressing polypharmacy and identifying and deprescribing potentially inappropriate medications are essential in preventing adverse drug events, drug-drug interactions, and medication nonadherence.9 In the VHA, VIONE (Vital, Important, Optional, Not indicated, Every medication has an indication) is a rapidly expanding medication deprescribing program that exemplifies HRO principles.9 VIONE provides medication management that supports shared decision making, reducing risk and improving patient safety and quality of life.9 As of June 2023, > 600,000 unique veterans have benefited from VIONE, with an average of 2.2 medications deprescribed per patient with an annual cost avoidance of > $100 million.10
Assessing and acting on mentation includes preventing, identifying, and managing depression and dementia in outpatient settings and delirium in hospital and long-term care settings.5 There are many tools and clinical reminders available in the EHR so that interdisciplinary teams can document changes to mentation and identify opportunities for continuous improvement.
Closely aligned with mentation is mobility, with evidence suggesting that regular physical activity reduces the risk of falls (preventing associated complications), maintains physical functioning, and lowers the risk of cognitive impairment and depression.5 Ensuring early, frequent, and safe mobility helps patients achieve better health outcomes and prevent injury.5 Mobility programs within the VHA include the STRIDE program for the inpatient setting and Gerofit for outpatient settings.11,12
HRO Principles
An HRO is a complex environment of care that experiences fewer than anticipated accidents or adverse events by (1) establishing trust among leaders and staff by balancing individual accountability with systems thinking; (2) empowering staff to lead continuous process improvements; and (3) creating an environment where employees feel safe to report harm or near misses, focusing on the reasons errors occur.13 The work of AFHS incorporates HRO principles with an emphasis on 3 elements. First, it involves interactive systems and processes needed to support 4Ms care across care settings. Second, AFHS acknowledge the complexity of age-friendly work and deference to the expertise of interdisciplinary team members. Finally, AFHS are committed to resilience by overcoming failures and challenges to implementation and long-term sustainment as a standard of practice.
Case study
The names and details in this case have been modified to protect patient privacy. It is representative of many Community Living Centers (CLCs) involved in AFHS that work to create a safe, person-centered environment for veterans.
In a CLC team workroom, 2 nurses were discussing a long-term care resident. The nurses approached the attending physician and explained that they were worried about Sgt Johnson, who seemed depressed and sometimes combative. They had noticed a change in his behavior when they helped him clean up after an episode of incontinence and were concerned that he would try to get out of bed on his own and fall. The attending physician thanked them for sharing their concerns. Sgt Johnson was a retired Army veteran who had a long, decorated military career. His chronic health conditions had led to muscle weakness, and he fell and broke a hip before this admission. He had an uneventful hip replacement but was showing signs of depression due to his limited mobility, loss of independence, and inability to live at home without additional support.
The attending physician knocked on the door of his room, sat down next to the bed, and asked, “How are you feeling today?” Sgt Johnson tersely replied, “About the same.” The physician asked, “Sgt Johnson, what matters most to you related to your recovery? What is important to you?” Sgt Johnson responded, “Feeling like a man!” The doctor replied, “So what makes you feel ‘not like a man’?” The Sgt replied, “Having to be cleaned up by the nurses and not being able to use the toilet on my own.” The physician surmised that his decline in physical functioning had a connection to his worsening depression and combativeness and said to the Sgt, “Let’s get the team together and work out a plan to get you strong enough to use a bedside commode by yourself. Let’s make that the first goal in our plan to get you back to using the toilet independently. Can you work with us on that?” He smiled and said, “Sir, yes Sir!”
At the weekly interdisciplinary team meeting, the team discussed Sgt Johnson’s wishes and the nurses’ safety concerns. The physician reported to the team what mattered to the veteran. The nurses arranged for a bedside commode and supplies to be placed in his room, encouraged and assisted him, and provided a privacy screen. The physical therapist continued to support his mobility needs, concentrating on transfers, small steps like standing and turning with a walker to get in position to use the bedside commode, and later the bathroom toilet. The psychologist addressed what matters to Sgt Johnson and his mentation, health goals, and coping strategies. The social worker provided support and counseling for the veteran and his family. The pharmacist checked his medications to be sure that none were affecting his gastrointestinal tract and his ability to move safely and do what matters to him. Knowing what mattered to Sgt Johnson was the driver of the interdisciplinary care plan to provide 4Ms care.
The team worked collaboratively with the veteran to develop and set attainable goals around toileting and regaining his dignity. This improved his overall recovery. As Sgt Johnson became more independent, his mood gradually improved and he began to participate in other activities and interact with other residents on the unit, and he did not experience any falls. By addressing the 4Ms, the interdisciplinary team coordinated efforts to provide high-quality, person-centered care. They built trust with the veteran, shared accountability, and followed HRO principles to keep the veteran safe.
Becoming an Age-Friendly HRO
Becoming an HRO is a dynamic, ever-changing process to maintain high standards, improve care quality, and cause no harm. There are 3 pillars and 5 principles that guide an HRO. The pillars are critical areas of focus and include leadership commitment, culture of safety, and continuous process improvement.14 The first of 5 HRO principles is sensitivity to operations. This is defined as an awareness of how processes and systems impact the entire organization, the downstream impact.15 Focusing on the 4Ms helps develop the capability of frontline staff to provide high-quality care for older adults while ensuring that processes are in place to support the work. The 4Ms provide an efficient way to organize interdisciplinary team meetings, provide warm handoffs using Situation-Background-Assessment-Recommendation, and standardize documentation. Involvement in the AFHS movement improves communication, care quality, and patient and staff satisfaction to meet this HRO principle.15
The second HRO principle, reluctance to simplify, ensures that direct care staff and leaders delve further into issues to find solutions.15 AFHS use the Plan-Do-Study-Act cycle to put the 4Ms into practice; this cycle helps teams test small increments of change, study their performance, and act to ensure that all 4Ms are being practiced as a set. AFHS teams are encouraged to review at least 3 months of data after implementation of the 4Ms, working to find solutions if there are gaps or issues identified.
The third principle, preoccupation with failure, refers to shared attentiveness—being prepared for the unexpected and learning from mistakes.15 The entire AFHS team shares responsibility for providing 4Ms care, where staff are empowered to report any safety concerns or close calls. The fourth principle of deference to expertise includes listening to staff who have the most knowledge for the task at hand, which aligns with the collaborative interdisciplinary teamwork of age-friendly teams.15
The final HRO principle, commitment to resilience, includes continuous learning, interdisciplinary team training, and sharing of lessons learned.15 Although IHI offers 2 levels of AFHS recognition, teams are continuously learning to improve and sustain care beyond level 2, Committed to Care Excellence recognition.16
The Table shows the VHA’s AFHS implementation strategies and the HRO principles adapted from the Joint Commission’s High Reliability Health Care Maturity Model and the IHI’s Framework for Safe, Reliable, and Effective Care. The VHA is developing a national dashboard to capture age-friendly processes and health outcome measures that address patient safety and care quality.
Conclusions
AFHS empowers VHA teams to honor veterans’ care preferences and values, supporting their independence, dignity, and quality of life across care settings. The adoption of AFHS brings evidence-based practices to the point of care by addressing common pitfalls in the care of older adults, drawing attention to, and calling for action on inappropriate medication use, physical inactivity, and assessment of the vulnerable brain. The 4Ms also serve as a framework to continuously improve care and cause zero harm, reinforcing HRO pillars and principles across the VHA, and ensuring that older adults reliably receive the evidence-based, high-quality care they deserve.
1. Veterans Health Administration. Providing healthcare for veterans. Updated June 20, 2023. Accessed June 26, 2023. https://www.va.gov/health
2. Veazie S, Peterson K, Bourne D. Evidence brief: implementation of high reliability organization principles. Washington, DC: Evidence Synthesis Program, Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs. VA ESP Project #09-199; 2019. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/esp/high-reliability-org.cfm
3. Church K, Munro S, Shaughnessy M, Clancy C. Age-Friendly Health Systems: improving care for older adults in the Veterans Health Administration. Health Serv Res. 2023;58(suppl 1):5-8. doi:10.1111/1475-6773.14110
4. Farrell TW, Volden TA, Butler JM, et al. Age-friendly care in the Veterans Health Administration: past, present, and future. J Am Geriatr Soc. 2023;71(1):18-25. doi:10.1111/jgs.18070
5. Mate K, Fulmer T, Pelton L, et al. Evidence for the 4Ms: interactions and outcomes across the care continuum. J Aging Health. 2021;33(7-8):469-481. doi:10.1177/0898264321991658
6. Tinetti ME, Naik AD, Dindo L, et al. Association of patient priorities-aligned decision-making with patient outcomes and ambulatory health care burden among older adults with multiple chronic conditions: A nonrandomized clinical trial. JAMA Intern Med. 2019;179(12):1688-1697. doi:10.1001/jamainternmed.2019.4235
7. US Department of Veterans Affairs. What is whole health? Updated: October 31, 2023. November 30, 2023. https://www.va.gov/wholehealth
8. Patient Priorities Care. Updated 2019. Accessed November 30, 2023. https://patientprioritiescare.org
9. Battar S, Watson Dickerson KR, Sedgwick C, Cmelik T. Understanding principles of high reliability organizations through the eyes of VIONE: a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
10. VA Diffusion Marketplace. VIONE- medication optimization and polypharmacy reduction initiative. Accessed November 30, 2023. https://marketplace.va.gov/innovations/vione
11. US Department of Veterans Affairs, Office of Research and Development. STRIDE program to keep hospitalized veterans mobile. Updated November 6, 2018. Accessed November 30, 2023. https://www.research.va.gov/research_in_action/STRIDE-program-to-keep-hospitalized-Veterans-mobile.cfm
12. US Department of Veterans Affairs, VA Geriatrics and Extended Care. Gerofit: a program promoting exercise and health for older veterans. Updated August 2, 2023. Accessed November 30, 2023. https://www.va.gov/GERIATRICS/pages/gerofit_Home.asp
13. US Department of Veterans Affairs, Health Services Research and Development. VHA’s vision for a high reliability organization. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-1
14. US Department of Veterans Affairs, Health Services Research and Development. Three HRO evaluation priorities. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-2
15. Oster CA, Deakins S. Practical application of high-reliability principles in healthcare to optimize quality and safety outcomes. J Nurs Adm. 2018;48(1):50-55. doi:10.1097/NNA.0000000000000570
16. Institute for Healthcare Improvement. Age-Friendly Health Systems recognitions. Accessed November 30, 2023. https://www.ihi.org/Engage/Initiatives/Age-Friendly-Health-Systems/Pages/Recognition.aspx
1. Veterans Health Administration. Providing healthcare for veterans. Updated June 20, 2023. Accessed June 26, 2023. https://www.va.gov/health
2. Veazie S, Peterson K, Bourne D. Evidence brief: implementation of high reliability organization principles. Washington, DC: Evidence Synthesis Program, Health Services Research and Development Service, Office of Research and Development, Department of Veterans Affairs. VA ESP Project #09-199; 2019. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/esp/high-reliability-org.cfm
3. Church K, Munro S, Shaughnessy M, Clancy C. Age-Friendly Health Systems: improving care for older adults in the Veterans Health Administration. Health Serv Res. 2023;58(suppl 1):5-8. doi:10.1111/1475-6773.14110
4. Farrell TW, Volden TA, Butler JM, et al. Age-friendly care in the Veterans Health Administration: past, present, and future. J Am Geriatr Soc. 2023;71(1):18-25. doi:10.1111/jgs.18070
5. Mate K, Fulmer T, Pelton L, et al. Evidence for the 4Ms: interactions and outcomes across the care continuum. J Aging Health. 2021;33(7-8):469-481. doi:10.1177/0898264321991658
6. Tinetti ME, Naik AD, Dindo L, et al. Association of patient priorities-aligned decision-making with patient outcomes and ambulatory health care burden among older adults with multiple chronic conditions: A nonrandomized clinical trial. JAMA Intern Med. 2019;179(12):1688-1697. doi:10.1001/jamainternmed.2019.4235
7. US Department of Veterans Affairs. What is whole health? Updated: October 31, 2023. November 30, 2023. https://www.va.gov/wholehealth
8. Patient Priorities Care. Updated 2019. Accessed November 30, 2023. https://patientprioritiescare.org
9. Battar S, Watson Dickerson KR, Sedgwick C, Cmelik T. Understanding principles of high reliability organizations through the eyes of VIONE: a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
10. VA Diffusion Marketplace. VIONE- medication optimization and polypharmacy reduction initiative. Accessed November 30, 2023. https://marketplace.va.gov/innovations/vione
11. US Department of Veterans Affairs, Office of Research and Development. STRIDE program to keep hospitalized veterans mobile. Updated November 6, 2018. Accessed November 30, 2023. https://www.research.va.gov/research_in_action/STRIDE-program-to-keep-hospitalized-Veterans-mobile.cfm
12. US Department of Veterans Affairs, VA Geriatrics and Extended Care. Gerofit: a program promoting exercise and health for older veterans. Updated August 2, 2023. Accessed November 30, 2023. https://www.va.gov/GERIATRICS/pages/gerofit_Home.asp
13. US Department of Veterans Affairs, Health Services Research and Development. VHA’s vision for a high reliability organization. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-1
14. US Department of Veterans Affairs, Health Services Research and Development. Three HRO evaluation priorities. Updated August 14, 2020. Accessed November 30, 2023. https://www.hsrd.research.va.gov/publications/forum/summer20/default.cfm?ForumMenu=summer20-2
15. Oster CA, Deakins S. Practical application of high-reliability principles in healthcare to optimize quality and safety outcomes. J Nurs Adm. 2018;48(1):50-55. doi:10.1097/NNA.0000000000000570
16. Institute for Healthcare Improvement. Age-Friendly Health Systems recognitions. Accessed November 30, 2023. https://www.ihi.org/Engage/Initiatives/Age-Friendly-Health-Systems/Pages/Recognition.aspx
Fellowships in Complex Medical Dermatology
Complex medical dermatology has become an emerging field in dermatology. Although a rather protean and broad term, complex medical dermatology encompasses patients with autoimmune conditions, bullous disease, connective tissue disease, vasculitis, severe dermatoses requiring immunomodulation, and inpatient consultations. Importantly, dermatology inpatient consultations aid in lowering health care costs due to accurate diagnoses, correct treatment, and decreased hospital stays.1 A fellowship is not required for holding an inpatient role in the hospital system as a dermatologist but can be beneficial. There are combined internal medicine–dermatology programs available for medical students applying to dermatology residency, but a complex medical dermatology fellowship is an option after residency for those who are interested. I believe that a focused complex medical dermatology fellowship differs from the training offered in combined internal medicine–dermatology residency. My fellow colleagues in combined internal medicine–dermatology programs are exposed to systemic manifestations of cutaneous disease and are experts in the interplay between the skin and other organ systems. However, the focus of their programs is with the intention of becoming double boarded in internal medicine and dermatology with comprehensive exposure to both fields. In my fellowship, I am able to tailor my schedule to focus on any dermatologic disease such as connective tissue disease, pruritus, graft vs host disease, and Merkel cell carcinoma. I ultimately can determine a niche in dermatology and hone my skills for a year under supervision.
Available Fellowships
Fellowship Locations—Importantly, the complex medical dermatology fellowship is not accredited by the Accreditation Council for Graduate Medical Education, which can make it difficult to identify and apply to programs. The complex medical dermatology fellowship is different than a rheumatology-dermatology fellowship, cutaneous oncology fellowship, pediatric dermatology fellowship, or other subspecialty fellowships such as those in itch or autoimmune blistering diseases. The fellowship often encompasses gaining clinical expertise in many of these conditions. I performed a thorough search online and spoke with complex medical dermatologists to compile a list of programs that offer a complex medical dermatology fellowship: Brigham and Women’s Hospital (Boston, Massachusetts); University of California San Francisco (San Francisco, California); University of Pennsylvania (Philadelphia, Pennsylvania); Cleveland Clinic (Cleveland, Ohio); and New York University (New York, New York)(Table). Only 1 spot is offered at each of these programs.
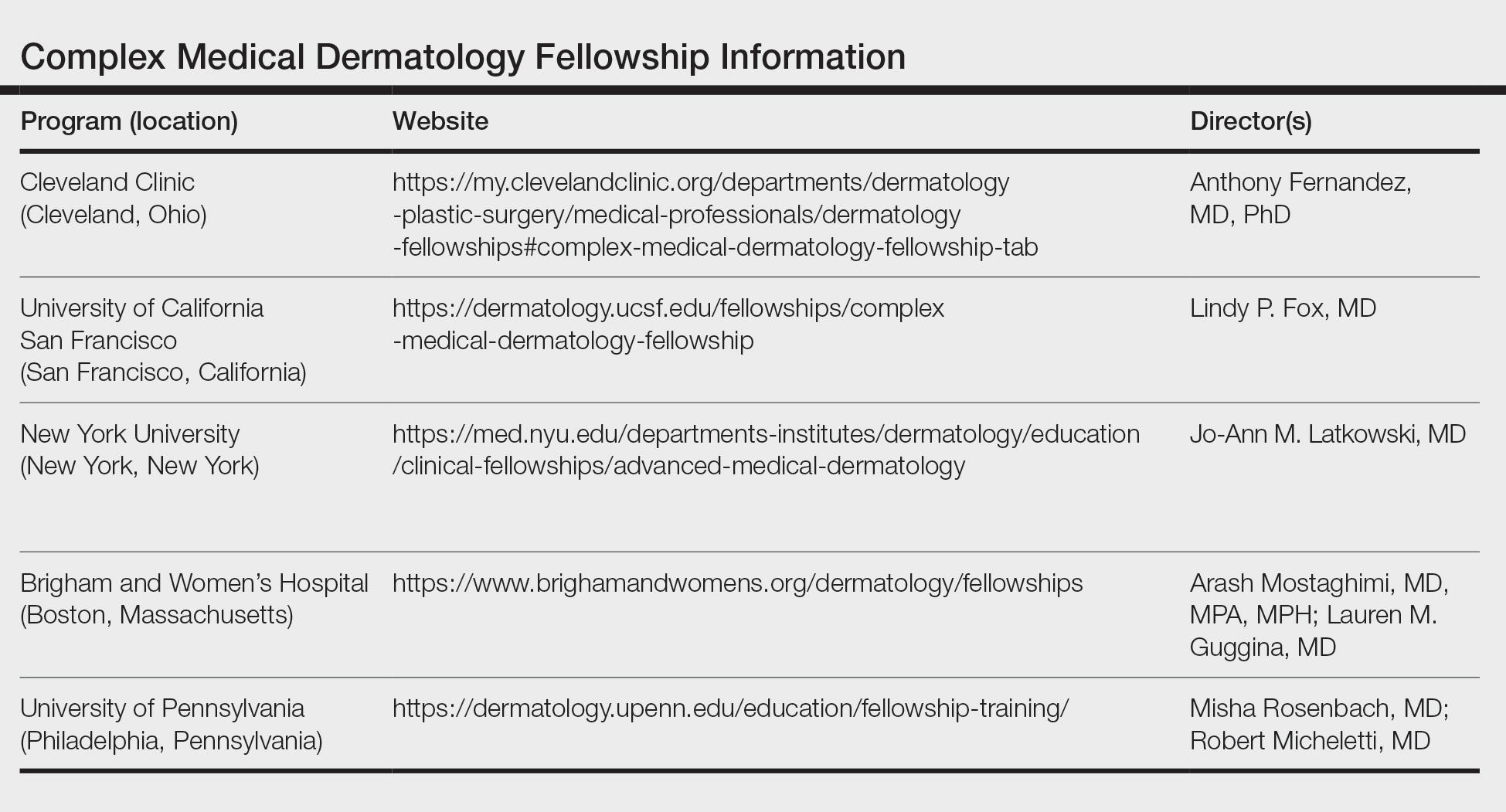
Reason to Pursue the Fellowship—There are many reasons to pursue a fellowship in complex medical dermatology such as a desire to enhance exposure to the field, to practice in an academic center and develop a niche within dermatology, to practice dermatology in an inpatient setting, to improve delivery of health care to medically challenging populations in a community setting, and to become an expert on cutaneous manifestations of internal and systemic disease.
Application—There is no standardized application or deadline for this fellowship; however, there is a concerted attempt from some of the programs to offer interviews and decisions at a similar time. Deadlines and contact information are listed on the program websites, along with more details (Table).
Recommendations—I would recommend reaching out at the beginning of postgraduate year (PGY) 4 to these programs and voicing your interest in the fellowship. It is possible to set up an away rotation at some of the programs, and if your program offers elective time, pursuing an away rotation during PGY-3 or early in PGY-4 can prove to be advantageous. Furthermore, during my application cycle I toured the University of California San Francisco, University of Pennsylvania, and Brigham and Women’s Hospital to gain further insight into each program.
Brigham and Women’s Complex Medical Dermatology Fellowship
I am currently the complex medical dermatology fellow at Brigham and Women’s Hospital, and it has been an outstanding experience thus far. The program offers numerous subspecialty clinics focusing solely on cutaneous-oncodermatology, psoriasis, rheumatology-dermatology, skin of color, mole mapping backed by artificial intelligence, cosmetics, high-risk skin cancer, neutrophilic dermatoses, patch testing, phototherapy, psychodermatology, and transplant dermatology. In addition to a wide variety of subspecialty clinics, fellows have the opportunity to participate in inpatient dermatology rounds and act as a junior attending. I appreciate the flexibility of this program combined with the ability to work alongside worldwide experts. There are numerous teaching opportunities, and all of the faculty are amiable and intelligent and emphasize wellness, education, and autonomy. Overall, my experience and decision to pursue a complex medical dermatology fellowship has been extremely rewarding and invaluable. I am gaining additional skills to aid medically challenging patients while pursuing my true passion in dermatology.
1. Sahni DR. Inpatient dermatology consultation services in hospital institutions. Cutis. 2023;111:E11-E12. doi:10.12788/cutis.0776.
Complex medical dermatology has become an emerging field in dermatology. Although a rather protean and broad term, complex medical dermatology encompasses patients with autoimmune conditions, bullous disease, connective tissue disease, vasculitis, severe dermatoses requiring immunomodulation, and inpatient consultations. Importantly, dermatology inpatient consultations aid in lowering health care costs due to accurate diagnoses, correct treatment, and decreased hospital stays.1 A fellowship is not required for holding an inpatient role in the hospital system as a dermatologist but can be beneficial. There are combined internal medicine–dermatology programs available for medical students applying to dermatology residency, but a complex medical dermatology fellowship is an option after residency for those who are interested. I believe that a focused complex medical dermatology fellowship differs from the training offered in combined internal medicine–dermatology residency. My fellow colleagues in combined internal medicine–dermatology programs are exposed to systemic manifestations of cutaneous disease and are experts in the interplay between the skin and other organ systems. However, the focus of their programs is with the intention of becoming double boarded in internal medicine and dermatology with comprehensive exposure to both fields. In my fellowship, I am able to tailor my schedule to focus on any dermatologic disease such as connective tissue disease, pruritus, graft vs host disease, and Merkel cell carcinoma. I ultimately can determine a niche in dermatology and hone my skills for a year under supervision.
Available Fellowships
Fellowship Locations—Importantly, the complex medical dermatology fellowship is not accredited by the Accreditation Council for Graduate Medical Education, which can make it difficult to identify and apply to programs. The complex medical dermatology fellowship is different than a rheumatology-dermatology fellowship, cutaneous oncology fellowship, pediatric dermatology fellowship, or other subspecialty fellowships such as those in itch or autoimmune blistering diseases. The fellowship often encompasses gaining clinical expertise in many of these conditions. I performed a thorough search online and spoke with complex medical dermatologists to compile a list of programs that offer a complex medical dermatology fellowship: Brigham and Women’s Hospital (Boston, Massachusetts); University of California San Francisco (San Francisco, California); University of Pennsylvania (Philadelphia, Pennsylvania); Cleveland Clinic (Cleveland, Ohio); and New York University (New York, New York)(Table). Only 1 spot is offered at each of these programs.

Reason to Pursue the Fellowship—There are many reasons to pursue a fellowship in complex medical dermatology such as a desire to enhance exposure to the field, to practice in an academic center and develop a niche within dermatology, to practice dermatology in an inpatient setting, to improve delivery of health care to medically challenging populations in a community setting, and to become an expert on cutaneous manifestations of internal and systemic disease.
Application—There is no standardized application or deadline for this fellowship; however, there is a concerted attempt from some of the programs to offer interviews and decisions at a similar time. Deadlines and contact information are listed on the program websites, along with more details (Table).
Recommendations—I would recommend reaching out at the beginning of postgraduate year (PGY) 4 to these programs and voicing your interest in the fellowship. It is possible to set up an away rotation at some of the programs, and if your program offers elective time, pursuing an away rotation during PGY-3 or early in PGY-4 can prove to be advantageous. Furthermore, during my application cycle I toured the University of California San Francisco, University of Pennsylvania, and Brigham and Women’s Hospital to gain further insight into each program.
Brigham and Women’s Complex Medical Dermatology Fellowship
I am currently the complex medical dermatology fellow at Brigham and Women’s Hospital, and it has been an outstanding experience thus far. The program offers numerous subspecialty clinics focusing solely on cutaneous-oncodermatology, psoriasis, rheumatology-dermatology, skin of color, mole mapping backed by artificial intelligence, cosmetics, high-risk skin cancer, neutrophilic dermatoses, patch testing, phototherapy, psychodermatology, and transplant dermatology. In addition to a wide variety of subspecialty clinics, fellows have the opportunity to participate in inpatient dermatology rounds and act as a junior attending. I appreciate the flexibility of this program combined with the ability to work alongside worldwide experts. There are numerous teaching opportunities, and all of the faculty are amiable and intelligent and emphasize wellness, education, and autonomy. Overall, my experience and decision to pursue a complex medical dermatology fellowship has been extremely rewarding and invaluable. I am gaining additional skills to aid medically challenging patients while pursuing my true passion in dermatology.
Complex medical dermatology has become an emerging field in dermatology. Although a rather protean and broad term, complex medical dermatology encompasses patients with autoimmune conditions, bullous disease, connective tissue disease, vasculitis, severe dermatoses requiring immunomodulation, and inpatient consultations. Importantly, dermatology inpatient consultations aid in lowering health care costs due to accurate diagnoses, correct treatment, and decreased hospital stays.1 A fellowship is not required for holding an inpatient role in the hospital system as a dermatologist but can be beneficial. There are combined internal medicine–dermatology programs available for medical students applying to dermatology residency, but a complex medical dermatology fellowship is an option after residency for those who are interested. I believe that a focused complex medical dermatology fellowship differs from the training offered in combined internal medicine–dermatology residency. My fellow colleagues in combined internal medicine–dermatology programs are exposed to systemic manifestations of cutaneous disease and are experts in the interplay between the skin and other organ systems. However, the focus of their programs is with the intention of becoming double boarded in internal medicine and dermatology with comprehensive exposure to both fields. In my fellowship, I am able to tailor my schedule to focus on any dermatologic disease such as connective tissue disease, pruritus, graft vs host disease, and Merkel cell carcinoma. I ultimately can determine a niche in dermatology and hone my skills for a year under supervision.
Available Fellowships
Fellowship Locations—Importantly, the complex medical dermatology fellowship is not accredited by the Accreditation Council for Graduate Medical Education, which can make it difficult to identify and apply to programs. The complex medical dermatology fellowship is different than a rheumatology-dermatology fellowship, cutaneous oncology fellowship, pediatric dermatology fellowship, or other subspecialty fellowships such as those in itch or autoimmune blistering diseases. The fellowship often encompasses gaining clinical expertise in many of these conditions. I performed a thorough search online and spoke with complex medical dermatologists to compile a list of programs that offer a complex medical dermatology fellowship: Brigham and Women’s Hospital (Boston, Massachusetts); University of California San Francisco (San Francisco, California); University of Pennsylvania (Philadelphia, Pennsylvania); Cleveland Clinic (Cleveland, Ohio); and New York University (New York, New York)(Table). Only 1 spot is offered at each of these programs.

Reason to Pursue the Fellowship—There are many reasons to pursue a fellowship in complex medical dermatology such as a desire to enhance exposure to the field, to practice in an academic center and develop a niche within dermatology, to practice dermatology in an inpatient setting, to improve delivery of health care to medically challenging populations in a community setting, and to become an expert on cutaneous manifestations of internal and systemic disease.
Application—There is no standardized application or deadline for this fellowship; however, there is a concerted attempt from some of the programs to offer interviews and decisions at a similar time. Deadlines and contact information are listed on the program websites, along with more details (Table).
Recommendations—I would recommend reaching out at the beginning of postgraduate year (PGY) 4 to these programs and voicing your interest in the fellowship. It is possible to set up an away rotation at some of the programs, and if your program offers elective time, pursuing an away rotation during PGY-3 or early in PGY-4 can prove to be advantageous. Furthermore, during my application cycle I toured the University of California San Francisco, University of Pennsylvania, and Brigham and Women’s Hospital to gain further insight into each program.
Brigham and Women’s Complex Medical Dermatology Fellowship
I am currently the complex medical dermatology fellow at Brigham and Women’s Hospital, and it has been an outstanding experience thus far. The program offers numerous subspecialty clinics focusing solely on cutaneous-oncodermatology, psoriasis, rheumatology-dermatology, skin of color, mole mapping backed by artificial intelligence, cosmetics, high-risk skin cancer, neutrophilic dermatoses, patch testing, phototherapy, psychodermatology, and transplant dermatology. In addition to a wide variety of subspecialty clinics, fellows have the opportunity to participate in inpatient dermatology rounds and act as a junior attending. I appreciate the flexibility of this program combined with the ability to work alongside worldwide experts. There are numerous teaching opportunities, and all of the faculty are amiable and intelligent and emphasize wellness, education, and autonomy. Overall, my experience and decision to pursue a complex medical dermatology fellowship has been extremely rewarding and invaluable. I am gaining additional skills to aid medically challenging patients while pursuing my true passion in dermatology.
1. Sahni DR. Inpatient dermatology consultation services in hospital institutions. Cutis. 2023;111:E11-E12. doi:10.12788/cutis.0776.
1. Sahni DR. Inpatient dermatology consultation services in hospital institutions. Cutis. 2023;111:E11-E12. doi:10.12788/cutis.0776.
RESIDENT PEARL
- Complex medical dermatology is a rewarding and fascinating subspecialty of dermatology, and additional training can be accomplished through a fellowship at a variety of prestigious institutions.
Camp Lejeune Family Members Now Eligible for Health Care Reimbursement Related to Parkinson Disease
Family members of veterans exposed to contaminated drinking water at Marine Corps Base Camp Lejeune, Jacksonville, North Carolina, from August 1, 1953, to December 31, 1987, are now eligible for reimbursement of health care costs associated with Parkinson disease (PD) under the Camp Lejeune Family Member Program, the US Department of Veterans Affairs (VA) has announced.
That brings the number of illnesses or conditions those family members can be reimbursed for to 16: esophageal, lung, breast, bladder, and kidney cancer, leukemia, multiple myeloma, renal toxicity, miscarriage, hepatic steatosis, female infertility, myelodysplastic syndromes, scleroderma, neurobehavioral effects, non-Hodgkin lymphoma, and Parkinson disease.
A recent JAMA study of 340,489 service members found that the risk of PD is 70% higher for veterans stationed at Camp Lejeune (n = 279) compared with veterans stationed at Camp Pendleton, California (n = 151).
The researchers say water supplies at Camp Lejeune were contaminated with several volatile organic compounds. They suggest that the risk of PD may be related to trichloroethylene exposure (TCE), a volatile organic compound widely used as a cleaning agent, in the manufacturing of some refrigerants, and found in paints and other products. In January, the US Environmental Protection Agency issued a revised risk determination saying that TCE presents an unreasonable risk to the health of workers, occupational nonusers (workers nearby but not in direct contact with this chemical), consumers, and bystanders.
Levels at Camp Lejeune were highest for TCE, with monthly median values greater than 70-fold the permissible amount.
Camp Lejeune veterans also had a significantly increased risk of prodromal PD diagnoses, including tremor, anxiety, and erectile dysfunction, and higher cumulative prodromal risk scores. No excess risk was found for other forms of neurodegenerative parkinsonism.
The PACT Act allows veterans and their families to file lawsuits for harm caused by exposure to contaminated water at Camp Lejeune. “Veterans and their families deserve no-cost health care for the conditions they developed due to the contaminated water at Camp Lejeune,” said VA’s Under Secretary for Health, Dr. Shereef Elnahal, MD. “We’re proud to add Parkinson disease to the list of conditions that are covered for veteran family members, and we implore anyone who may be living with this disease—or any of the other conditions covered by VA’s Camp Lejeune Family Member Program—to apply for assistance today.”
Family members of veterans exposed to contaminated drinking water at Marine Corps Base Camp Lejeune, Jacksonville, North Carolina, from August 1, 1953, to December 31, 1987, are now eligible for reimbursement of health care costs associated with Parkinson disease (PD) under the Camp Lejeune Family Member Program, the US Department of Veterans Affairs (VA) has announced.
That brings the number of illnesses or conditions those family members can be reimbursed for to 16: esophageal, lung, breast, bladder, and kidney cancer, leukemia, multiple myeloma, renal toxicity, miscarriage, hepatic steatosis, female infertility, myelodysplastic syndromes, scleroderma, neurobehavioral effects, non-Hodgkin lymphoma, and Parkinson disease.
A recent JAMA study of 340,489 service members found that the risk of PD is 70% higher for veterans stationed at Camp Lejeune (n = 279) compared with veterans stationed at Camp Pendleton, California (n = 151).
The researchers say water supplies at Camp Lejeune were contaminated with several volatile organic compounds. They suggest that the risk of PD may be related to trichloroethylene exposure (TCE), a volatile organic compound widely used as a cleaning agent, in the manufacturing of some refrigerants, and found in paints and other products. In January, the US Environmental Protection Agency issued a revised risk determination saying that TCE presents an unreasonable risk to the health of workers, occupational nonusers (workers nearby but not in direct contact with this chemical), consumers, and bystanders.
Levels at Camp Lejeune were highest for TCE, with monthly median values greater than 70-fold the permissible amount.
Camp Lejeune veterans also had a significantly increased risk of prodromal PD diagnoses, including tremor, anxiety, and erectile dysfunction, and higher cumulative prodromal risk scores. No excess risk was found for other forms of neurodegenerative parkinsonism.
The PACT Act allows veterans and their families to file lawsuits for harm caused by exposure to contaminated water at Camp Lejeune. “Veterans and their families deserve no-cost health care for the conditions they developed due to the contaminated water at Camp Lejeune,” said VA’s Under Secretary for Health, Dr. Shereef Elnahal, MD. “We’re proud to add Parkinson disease to the list of conditions that are covered for veteran family members, and we implore anyone who may be living with this disease—or any of the other conditions covered by VA’s Camp Lejeune Family Member Program—to apply for assistance today.”
Family members of veterans exposed to contaminated drinking water at Marine Corps Base Camp Lejeune, Jacksonville, North Carolina, from August 1, 1953, to December 31, 1987, are now eligible for reimbursement of health care costs associated with Parkinson disease (PD) under the Camp Lejeune Family Member Program, the US Department of Veterans Affairs (VA) has announced.
That brings the number of illnesses or conditions those family members can be reimbursed for to 16: esophageal, lung, breast, bladder, and kidney cancer, leukemia, multiple myeloma, renal toxicity, miscarriage, hepatic steatosis, female infertility, myelodysplastic syndromes, scleroderma, neurobehavioral effects, non-Hodgkin lymphoma, and Parkinson disease.
A recent JAMA study of 340,489 service members found that the risk of PD is 70% higher for veterans stationed at Camp Lejeune (n = 279) compared with veterans stationed at Camp Pendleton, California (n = 151).
The researchers say water supplies at Camp Lejeune were contaminated with several volatile organic compounds. They suggest that the risk of PD may be related to trichloroethylene exposure (TCE), a volatile organic compound widely used as a cleaning agent, in the manufacturing of some refrigerants, and found in paints and other products. In January, the US Environmental Protection Agency issued a revised risk determination saying that TCE presents an unreasonable risk to the health of workers, occupational nonusers (workers nearby but not in direct contact with this chemical), consumers, and bystanders.
Levels at Camp Lejeune were highest for TCE, with monthly median values greater than 70-fold the permissible amount.
Camp Lejeune veterans also had a significantly increased risk of prodromal PD diagnoses, including tremor, anxiety, and erectile dysfunction, and higher cumulative prodromal risk scores. No excess risk was found for other forms of neurodegenerative parkinsonism.
The PACT Act allows veterans and their families to file lawsuits for harm caused by exposure to contaminated water at Camp Lejeune. “Veterans and their families deserve no-cost health care for the conditions they developed due to the contaminated water at Camp Lejeune,” said VA’s Under Secretary for Health, Dr. Shereef Elnahal, MD. “We’re proud to add Parkinson disease to the list of conditions that are covered for veteran family members, and we implore anyone who may be living with this disease—or any of the other conditions covered by VA’s Camp Lejeune Family Member Program—to apply for assistance today.”
VA Partners to Open Clinics, Build Facilities that Increase Veteran Access to Health Care
The US Department of Veterans Affairs (VA) has been establishing partnerships right, left, and center to improve and expand care for veterans. Instead of going it alone, VA is partnering with academic affiliates, Native American tribes, and the military to take advantage of state and federal funds.
In California, the VA Palo Alto Health Care System and Stanford Medicine announced a deal to plan, build, and operate a National Cancer Institute–designated joint cancer care and research center on the VA Palo Alto campus. The partnership is another offshoot of the PACT Act, in part because of the number of veterans who need cancer treatment related to, for instance, airborne toxins. The influx of veterans via the PACT Act could represent “the largest expansion of veterans’ benefits in history,” VA Under Secretary for Health Shereef Elnahal, MD, MBA, said at a press event about the collaboration. “This will allow us to partner with every powerhouse academic center in the country if we do this right. For research, training, and care delivery, it’s all one bucket of cancer care that veterans deserve.”
A separate partnership between the Cherokee Nation and Eastern Oklahoma VA Healthcare System will establish a VA clinic inside the Cherokee Nation’s Vinita Health Center, an hour northeast of Tulsa. The clinic, expected to open early next year, will serve any veteran. “Cherokees and other Native Americans serve in the US military at a higher rate than any other group, and veterans hold a special place in our hearts,” Cherokee Nation Principal Chief Chuck Hoskin Jr. said in a statement. “I am honored to do my part in covering veterans’ long-term health needs.”
The VA serves about 53,000 veterans living in eastern Oklahoma. Officials predict that partnership could serve as a roadmap for how rural America can work with tribes to increase care for veterans. “As we look ahead, this partnership with the VA can be a model for other tribes and communities across the nation,” Hoskin said.
Another collaborative plan, this one by the VA and US Department of Defense (DoD), will give about 37,000 Gulf Coast–area veterans access to care at a new Naval Hospital Pensacola clinic. Local veterans who previously received care from community clinicians or traveled to the Biloxi VA Medical Center in Mississippi will now be able to receive same-day, outpatient surgical care. “This partnership will help VA provide more care, more quickly, to more Gulf Coast veterans—as close to their homes as possible,” said Elnahal.
An agreement with the University of Pennsylvania Health System (UPHS) will improve infrastructure at the Coatesville VA Medical Center by repurposing a recently closed hospital nearby for outpatient, acute mental health, and long-term care services. “The PACT Act allows for great synergy between Penn Medicine and the VA, and we hope to leverage this new model to set the standard for how our nation approaches military medicine,” UPHS CEO Kevin B. Mahoney said.
An Eastern Oklahoma VA Health Care System hospital scheduled to open in 2025 in Tulsa was partially funded through the Communities Helping Invest through Property and Improvements Needed (CHIP-IN) program, the state of Oklahoma, the city of Tulsa, and the nonprofit team of Oklahoma State University Medical and the Anne and Henry Zarrow Foundation. When completed, the 58-bed hospital will serve approximately 38,000 veterans.
The US Department of Veterans Affairs (VA) has been establishing partnerships right, left, and center to improve and expand care for veterans. Instead of going it alone, VA is partnering with academic affiliates, Native American tribes, and the military to take advantage of state and federal funds.
In California, the VA Palo Alto Health Care System and Stanford Medicine announced a deal to plan, build, and operate a National Cancer Institute–designated joint cancer care and research center on the VA Palo Alto campus. The partnership is another offshoot of the PACT Act, in part because of the number of veterans who need cancer treatment related to, for instance, airborne toxins. The influx of veterans via the PACT Act could represent “the largest expansion of veterans’ benefits in history,” VA Under Secretary for Health Shereef Elnahal, MD, MBA, said at a press event about the collaboration. “This will allow us to partner with every powerhouse academic center in the country if we do this right. For research, training, and care delivery, it’s all one bucket of cancer care that veterans deserve.”
A separate partnership between the Cherokee Nation and Eastern Oklahoma VA Healthcare System will establish a VA clinic inside the Cherokee Nation’s Vinita Health Center, an hour northeast of Tulsa. The clinic, expected to open early next year, will serve any veteran. “Cherokees and other Native Americans serve in the US military at a higher rate than any other group, and veterans hold a special place in our hearts,” Cherokee Nation Principal Chief Chuck Hoskin Jr. said in a statement. “I am honored to do my part in covering veterans’ long-term health needs.”
The VA serves about 53,000 veterans living in eastern Oklahoma. Officials predict that partnership could serve as a roadmap for how rural America can work with tribes to increase care for veterans. “As we look ahead, this partnership with the VA can be a model for other tribes and communities across the nation,” Hoskin said.
Another collaborative plan, this one by the VA and US Department of Defense (DoD), will give about 37,000 Gulf Coast–area veterans access to care at a new Naval Hospital Pensacola clinic. Local veterans who previously received care from community clinicians or traveled to the Biloxi VA Medical Center in Mississippi will now be able to receive same-day, outpatient surgical care. “This partnership will help VA provide more care, more quickly, to more Gulf Coast veterans—as close to their homes as possible,” said Elnahal.
An agreement with the University of Pennsylvania Health System (UPHS) will improve infrastructure at the Coatesville VA Medical Center by repurposing a recently closed hospital nearby for outpatient, acute mental health, and long-term care services. “The PACT Act allows for great synergy between Penn Medicine and the VA, and we hope to leverage this new model to set the standard for how our nation approaches military medicine,” UPHS CEO Kevin B. Mahoney said.
An Eastern Oklahoma VA Health Care System hospital scheduled to open in 2025 in Tulsa was partially funded through the Communities Helping Invest through Property and Improvements Needed (CHIP-IN) program, the state of Oklahoma, the city of Tulsa, and the nonprofit team of Oklahoma State University Medical and the Anne and Henry Zarrow Foundation. When completed, the 58-bed hospital will serve approximately 38,000 veterans.
The US Department of Veterans Affairs (VA) has been establishing partnerships right, left, and center to improve and expand care for veterans. Instead of going it alone, VA is partnering with academic affiliates, Native American tribes, and the military to take advantage of state and federal funds.
In California, the VA Palo Alto Health Care System and Stanford Medicine announced a deal to plan, build, and operate a National Cancer Institute–designated joint cancer care and research center on the VA Palo Alto campus. The partnership is another offshoot of the PACT Act, in part because of the number of veterans who need cancer treatment related to, for instance, airborne toxins. The influx of veterans via the PACT Act could represent “the largest expansion of veterans’ benefits in history,” VA Under Secretary for Health Shereef Elnahal, MD, MBA, said at a press event about the collaboration. “This will allow us to partner with every powerhouse academic center in the country if we do this right. For research, training, and care delivery, it’s all one bucket of cancer care that veterans deserve.”
A separate partnership between the Cherokee Nation and Eastern Oklahoma VA Healthcare System will establish a VA clinic inside the Cherokee Nation’s Vinita Health Center, an hour northeast of Tulsa. The clinic, expected to open early next year, will serve any veteran. “Cherokees and other Native Americans serve in the US military at a higher rate than any other group, and veterans hold a special place in our hearts,” Cherokee Nation Principal Chief Chuck Hoskin Jr. said in a statement. “I am honored to do my part in covering veterans’ long-term health needs.”
The VA serves about 53,000 veterans living in eastern Oklahoma. Officials predict that partnership could serve as a roadmap for how rural America can work with tribes to increase care for veterans. “As we look ahead, this partnership with the VA can be a model for other tribes and communities across the nation,” Hoskin said.
Another collaborative plan, this one by the VA and US Department of Defense (DoD), will give about 37,000 Gulf Coast–area veterans access to care at a new Naval Hospital Pensacola clinic. Local veterans who previously received care from community clinicians or traveled to the Biloxi VA Medical Center in Mississippi will now be able to receive same-day, outpatient surgical care. “This partnership will help VA provide more care, more quickly, to more Gulf Coast veterans—as close to their homes as possible,” said Elnahal.
An agreement with the University of Pennsylvania Health System (UPHS) will improve infrastructure at the Coatesville VA Medical Center by repurposing a recently closed hospital nearby for outpatient, acute mental health, and long-term care services. “The PACT Act allows for great synergy between Penn Medicine and the VA, and we hope to leverage this new model to set the standard for how our nation approaches military medicine,” UPHS CEO Kevin B. Mahoney said.
An Eastern Oklahoma VA Health Care System hospital scheduled to open in 2025 in Tulsa was partially funded through the Communities Helping Invest through Property and Improvements Needed (CHIP-IN) program, the state of Oklahoma, the city of Tulsa, and the nonprofit team of Oklahoma State University Medical and the Anne and Henry Zarrow Foundation. When completed, the 58-bed hospital will serve approximately 38,000 veterans.
Who Gets to Determine Whether Home Is “Unsafe” at the End of Life?
Sometimes a patient at the end of life (EOL) just wants to go home. We recently treated such a patient, “Joe,” a 66-year-old veteran with end-stage chronic obstructive pulmonary disorder (COPD), severe hearing loss, and heavy alcohol use. A neighbor brought Joe to the hospital when he developed a urinary tract infection. Before hospitalization, Joe spent his days in bed. His neighbor was his designated health care agent (HCA) and caregiver, dropping off meals and bringing Joe to medical appointments. Joe had no other social support. In the hospital, Joe could not participate in physical therapy (PT) evaluations due to severe dyspnea on exertion. He was recommended for home PT, a home health aide, and home nursing, but Joe declined these services out of concern for encroachment on his independence. Given his heavy alcohol use, limited support, and functional limitations, the hospitalist team felt that Joe would be best served in a skilled nursing facility. As the palliative care team, we were consulted and felt that he was eligible for hospice. Joe simply wanted to go home.
Many patients like Joe experience functional decline at EOL, leading to increased care needs and transitions between sites of care.1 Some hospitalized patients at EOL want to transition directly to home, but due to their limited functioning and social support, discharge home may be deemed unsafe by health care professionals (HCPs). Clinicians then face the difficult balancing act of honoring patient wishes and avoiding a bad outcome. For patients at EOL, issues of capacity and risk become particularly salient. Furthermore, the unique structure of the US Department of Veterans Affairs (VA) health system and the psychosocial needs of some veterans add additional considerations for complex EOL discharges.2
End-of-life Decision Making
While patients may express strong preferences regarding their health care, their decision-making ability may worsen as they approach EOL. Contributing factors include older age, effects of hospitalization, treatment adverse effects, and comorbidities, including cognitive impairment. Studies of terminally ill patients show high rates of impaired decisional capacity.3,4 It is critical to assess capacity as part of discharge planning. Even when patients have the capacity, families and caregivers have an important voice, since they are often instrumental in maintaining patients at home.
Defining Risk
Determining whether a discharge is risky or unsafe is highly subjective, with differing opinions among clinicians and between patients and clinicians.5-7 In a qualitative study by Coombs and colleagues, HCPs tended toward a risk-averse approach to discharge decisions, sometimes favoring discharge to care facilities despite patient preferences.6 This approach also reflects pressures from the health care system to decrease the length of stay and reduce readmissions, important metrics for patient care and cost containment. However, keeping patients hospitalized or in nursing facilities does not completely mitigate risks (eg, falls) and carries other hazards (eg, nosocomial infections), as highlighted during the COVID-19 pandemic.7,8 The prospect of malpractice lawsuits and HCP moral distress about perceived risky home situations can also understandably affect decision making.
At the same time, risk calculation changes depending on the patient’s clinical status and priorities. Coombs and colleagues found that in contrast to clinicians, patients nearing EOL are willing to accept increasing risks and suboptimal living conditions to remain at home.6 What may be intolerable for a younger, healthier patient with a long life expectancy may be acceptable for someone who is approaching EOL. In our framework, a risky home discharge at EOL is considered one in which other adverse events, such as falls or inadequate symptom management, are likely.
Ethical Considerations
Unsafe discharges are challenging in part because some of the pillars of medical ethics can conflict. Prior articles have analyzed the ethical concerns of unsafe discharges in detail.9-11 Briefly, when patients wish to return home against initial medical recommendations, treatment teams may focus on the principles of beneficence and nonmaleficence, as exemplified by the desire to minimize harm, and justice, in which clinicians consider resource allocation and risks that a home discharge poses to family members, caregivers, and home health professionals. However, autonomy is important to consider as well. The concept of dignity of risk highlights the imperative to respect others’ decisions even when they increase the chance of harm, particularly given the overall shift in medicine from paternalism to shared decision making.12 Accommodating patient choice in how and where health care is received allows patients to regain some control over their lives, thereby enhancing their quality of life and promoting patient dignity, especially in their remaining days.13
Discharge Risk Framework
Our risk assessment framework helps clinicians more objectively identify factors that increase or decrease risk, inform discharge planning, partner with patients and families, give patients a prominent role in EOL decisions, and mitigate the risk of a bad outcome. This concept has been used in psychiatry, in which formal suicide assessment includes identifying risk factors and protective factors to estimate suicide risk and determine interventions.14 Similar to suicide risk estimation, this framework is based on clinical judgment rather than a specific calculation.
While this framework serves as a guide for determining and mitigating risk, we encourage teams to consider legal or ethical consultations in challenging cases, such as those in which patients lack both capacity and an involved HCA.
Step 1: Determine the patient’s capacity regarding disposition planning. Patients at EOL are at a higher risk of impaired decision-making capabilities; therefore, capacity evaluation is a critical step.
Step 2: Identify risk factors and protective factors for discharge home. Risk factors are intrinsic and extrinsic factors that increase risk such as functional or sensory impairments. Protective factors are intrinsic and extrinsic factors that decrease risk, including a good understanding of illness and consistent connection with the health care system (Table 1).
Step 3: Determine discharge to home risk level based on identified risk factors and protective factors. Patients may be at low, moderate, or high risk of having an adverse event, such as a fall or inadequate symptom control (Table 2).
Step 4: Identify risk mitigation strategies. These should be tailored to the patient based on the factors identified in Step 2. Examples include home nursing and therapy, mental health treatment, a medical alert system, and frequent contact between the patient and health care team.
Step 5: Meet with inpatient and outpatient HCP teams. Meetings should include the primary care professional (PCP) or relevant subspecialist, such as an oncologist for patients with cancer. For veterans receiving care solely at a local VA medical center, this can be easier to facilitate, but for veterans who receive care through both VA and non-VA systems, this step may require additional coordination. We also recommend including interdisciplinary team members, such as social workers, case managers, and the relevant home care or hospice agency. Certain agencies may decline admission if they perceive increased risk, such as no 24-hour care, perceived self-neglect, and limited instrumental support. During this meeting, HCPs discuss risk mitigation strategies identified in Step 4 and create a plan to propose to patients and families.
Step 6: Meet with patient, HCA, and family members. In addition to sharing information about prognosis, assessing caregiver capabilities and burden can guide conversations about discharge. The discharge plan should be determined through shared decision making.11 If the patient lacks capacity regarding disposition planning, this should be shared with the HCA. However, even when patients lack capacity, it is important to continue to engage them to understand their goals and preferences.
Step 7: Maximize risk mitigation strategies. If a moderate- or high-risk discharge is requested, the health care team should maximize risk mitigation strategies. For low-risk discharges, risk mitigation strategies can still promote safety, especially since risk increases as patients progress toward EOL. In some instances, patients, their HCAs, or caregivers may decline all risk mitigation strategies despite best efforts to communicate and negotiate options. In such circumstances, we recommend discussing the case with the outpatient team for a warm handoff. HCPs should also document all efforts (helpful from a legal standpoint as well as for the patient’s future treatment teams) and respect the decision to discharge home.
Applying the Framework
Our patient Joe provides a good illustration of how to implement this EOL framework. He was deemed to have the capacity to make decisions regarding discharge (Step 1). We determined his risk factors and protective factors for discharge (Step 2). His poor functional status, limited instrumental support, heavy alcohol use, rejection of home services, and communication barriers due to severe hearing impairment all increased his risk. Protective factors included an appreciation of functional limitations, intact cognition, and an involved HCA. Based on his limited instrumental support and poor function but good insight into limitations, discharge home was deemed to be of moderate risk (Step 3). Although risk factors such as alcohol use and severe hearing impairment could have raised his level to high risk, we felt that his involved HCA maintained him in the moderate-risk category.
We worked with the hospitalist team, PT, and audiology to identify multiple risk mitigation strategies: frequent phone calls between the HCA and outpatient palliative care team, home PT to improve transfers from bed to bedside commode, home nursing services either through a routine agency or hospice, and hearing aids for better communication (Steps 4 and 5). We then proposed these strategies to Joe and his HCA (Step 6). Due to concerns about infringement on his independence, Joe declined all home services but agreed to twice-daily check-ins by his HCA, frequent communication between his HCA and our team, and new hearing aids.
Joe returned home with the agreed-upon risk mitigation strategies in place (Step 7). Despite clinicians’ original reservations about sending Joe home without formal services, his HCA maintained close contact with our team, noting that Joe remained stable and happy to be at home in the months following discharge.
Conclusions
Fortunately, VA HCPs operate in an integrated health care system with access to psychological, social, and at-home medical support that can help mitigate risks. Still, we have benefitted from having a tool to help us evaluate risk systematically. Even if patients, families, and HCPs disagree on ideal discharge plans, this tool helps clinicians approach discharges methodically while maintaining open communication and partnership with patients. In doing so, our framework reflects the shift in medical culture from a patriarchal approach to shared decision-making practices regarding all aspects of medical care. Furthermore, we hope that this can help reduce clinician moral distress stemming from these challenging cases.
Future research on best practices for discharge risk assessment and optimizing home safety are needed. We also hope to evaluate the impact and effectiveness of our framework through interviews with key stakeholders. For Joe and other veterans like him, where to spend their final days may be the last important decision they make in life, and our framework allows for their voices to be better heard throughout the decision-making process.
Acknowledgments
We thank Brooke Lifland, MD, for her theoretical contributions to the concept behind this paper.
1. Committee on Approaching Death: Addressing Key End of Life Issues; Institute of Medicine. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington (DC): National Academies Press (US); March 19, 2015.
2. Casarett D, Pickard A, Amos Bailey F, et al. Important aspects of end-of-life care among veterans: implications for measurement and quality improvement. J Pain Symptom Manage. 2008;35(2):115-125. doi:10.1016/j.jpainsymman.2007.03.008
3. Kolva E, Rosenfeld B, Brescia R, Comfort C. Assessing decision-making capacity at end of life. Gen Hosp Psychiatry. 2014;36(4):392-397. doi:10.1016/j.genhosppsych.2014.02.013
4. Kolva E, Rosenfeld B, Saracino R. Assessing the decision-making capacity of terminally ill patients with cancer. Am J Geriatr Psychiatry. 2018;26(5):523-531. doi:10.1016/j.jagp.2017.11.012
5. Macmillan MS. Hospital staff’s perceptions of risk associated with the discharge of elderly people from acute hospital care. J Adv Nurs. 1994;19(2):249-256. doi:10.1111/j.1365-2648.1994.tb01078.x
6. Coombs MA, Parker R, de Vries K. Managing risk during care transitions when approaching end of life: A qualitative study of patients’ and health care professionals’ decision making. Palliat Med. 2017;31(7):617-624. doi:10.1177/0269216316673476
7. Hyslop B. ‘Not safe for discharge’? Words, values, and person-centred care. Age Ageing. 2020;49(3):334-336. doi:10.1093/ageing/afz170
8. Goodacre S. Safe discharge: an irrational, unhelpful and unachievable concept. Emerg Med J. 2006;23(10):753-755. doi:10.1136/emj.2006.037903
9. Swidler RN, Seastrum T, Shelton W. Difficult hospital inpatient discharge decisions: ethical, legal and clinical practice issues. Am J Bioeth. 2007;7(3):23-28. doi:10.1080/15265160601171739
10. Hill J, Filer W. Safety and ethical considerations in discharging patients to suboptimal living situations. AMA J Ethics. 2015;17(6):506-510. Published 2015 Jun 1. doi:10.1001/journalofethics.2015.17.6.ecas2-1506
11. West JC. What is an ethically informed approach to managing patient safety risk during discharge planning?. AMA J Ethics. 2020;22(11):E919-E923. Published 2020 Nov 1. doi:10.1001/amajethics.2020.919
12. Mukherjee D. Discharge decisions and the dignity of risk. Hastings Cent Rep. 2015;45(3):7-8. doi:10.1002/hast.441
13. Wheatley VJ, Baker JI. “Please, I want to go home”: ethical issues raised when considering choice of place of care in palliative care. Postgrad Med J. 2007;83(984):643-648. doi:10.1136/pgmj.2007.058487
14. Work Group on Suicidal Behaviors. Practice guideline for the assessment and treatment of patients with suicidal behaviors. Am J Psychiatry. 2003;160(suppl 11):1-60.
Sometimes a patient at the end of life (EOL) just wants to go home. We recently treated such a patient, “Joe,” a 66-year-old veteran with end-stage chronic obstructive pulmonary disorder (COPD), severe hearing loss, and heavy alcohol use. A neighbor brought Joe to the hospital when he developed a urinary tract infection. Before hospitalization, Joe spent his days in bed. His neighbor was his designated health care agent (HCA) and caregiver, dropping off meals and bringing Joe to medical appointments. Joe had no other social support. In the hospital, Joe could not participate in physical therapy (PT) evaluations due to severe dyspnea on exertion. He was recommended for home PT, a home health aide, and home nursing, but Joe declined these services out of concern for encroachment on his independence. Given his heavy alcohol use, limited support, and functional limitations, the hospitalist team felt that Joe would be best served in a skilled nursing facility. As the palliative care team, we were consulted and felt that he was eligible for hospice. Joe simply wanted to go home.
Many patients like Joe experience functional decline at EOL, leading to increased care needs and transitions between sites of care.1 Some hospitalized patients at EOL want to transition directly to home, but due to their limited functioning and social support, discharge home may be deemed unsafe by health care professionals (HCPs). Clinicians then face the difficult balancing act of honoring patient wishes and avoiding a bad outcome. For patients at EOL, issues of capacity and risk become particularly salient. Furthermore, the unique structure of the US Department of Veterans Affairs (VA) health system and the psychosocial needs of some veterans add additional considerations for complex EOL discharges.2
End-of-life Decision Making
While patients may express strong preferences regarding their health care, their decision-making ability may worsen as they approach EOL. Contributing factors include older age, effects of hospitalization, treatment adverse effects, and comorbidities, including cognitive impairment. Studies of terminally ill patients show high rates of impaired decisional capacity.3,4 It is critical to assess capacity as part of discharge planning. Even when patients have the capacity, families and caregivers have an important voice, since they are often instrumental in maintaining patients at home.
Defining Risk
Determining whether a discharge is risky or unsafe is highly subjective, with differing opinions among clinicians and between patients and clinicians.5-7 In a qualitative study by Coombs and colleagues, HCPs tended toward a risk-averse approach to discharge decisions, sometimes favoring discharge to care facilities despite patient preferences.6 This approach also reflects pressures from the health care system to decrease the length of stay and reduce readmissions, important metrics for patient care and cost containment. However, keeping patients hospitalized or in nursing facilities does not completely mitigate risks (eg, falls) and carries other hazards (eg, nosocomial infections), as highlighted during the COVID-19 pandemic.7,8 The prospect of malpractice lawsuits and HCP moral distress about perceived risky home situations can also understandably affect decision making.
At the same time, risk calculation changes depending on the patient’s clinical status and priorities. Coombs and colleagues found that in contrast to clinicians, patients nearing EOL are willing to accept increasing risks and suboptimal living conditions to remain at home.6 What may be intolerable for a younger, healthier patient with a long life expectancy may be acceptable for someone who is approaching EOL. In our framework, a risky home discharge at EOL is considered one in which other adverse events, such as falls or inadequate symptom management, are likely.
Ethical Considerations
Unsafe discharges are challenging in part because some of the pillars of medical ethics can conflict. Prior articles have analyzed the ethical concerns of unsafe discharges in detail.9-11 Briefly, when patients wish to return home against initial medical recommendations, treatment teams may focus on the principles of beneficence and nonmaleficence, as exemplified by the desire to minimize harm, and justice, in which clinicians consider resource allocation and risks that a home discharge poses to family members, caregivers, and home health professionals. However, autonomy is important to consider as well. The concept of dignity of risk highlights the imperative to respect others’ decisions even when they increase the chance of harm, particularly given the overall shift in medicine from paternalism to shared decision making.12 Accommodating patient choice in how and where health care is received allows patients to regain some control over their lives, thereby enhancing their quality of life and promoting patient dignity, especially in their remaining days.13
Discharge Risk Framework
Our risk assessment framework helps clinicians more objectively identify factors that increase or decrease risk, inform discharge planning, partner with patients and families, give patients a prominent role in EOL decisions, and mitigate the risk of a bad outcome. This concept has been used in psychiatry, in which formal suicide assessment includes identifying risk factors and protective factors to estimate suicide risk and determine interventions.14 Similar to suicide risk estimation, this framework is based on clinical judgment rather than a specific calculation.
While this framework serves as a guide for determining and mitigating risk, we encourage teams to consider legal or ethical consultations in challenging cases, such as those in which patients lack both capacity and an involved HCA.
Step 1: Determine the patient’s capacity regarding disposition planning. Patients at EOL are at a higher risk of impaired decision-making capabilities; therefore, capacity evaluation is a critical step.
Step 2: Identify risk factors and protective factors for discharge home. Risk factors are intrinsic and extrinsic factors that increase risk such as functional or sensory impairments. Protective factors are intrinsic and extrinsic factors that decrease risk, including a good understanding of illness and consistent connection with the health care system (Table 1).
Step 3: Determine discharge to home risk level based on identified risk factors and protective factors. Patients may be at low, moderate, or high risk of having an adverse event, such as a fall or inadequate symptom control (Table 2).
Step 4: Identify risk mitigation strategies. These should be tailored to the patient based on the factors identified in Step 2. Examples include home nursing and therapy, mental health treatment, a medical alert system, and frequent contact between the patient and health care team.
Step 5: Meet with inpatient and outpatient HCP teams. Meetings should include the primary care professional (PCP) or relevant subspecialist, such as an oncologist for patients with cancer. For veterans receiving care solely at a local VA medical center, this can be easier to facilitate, but for veterans who receive care through both VA and non-VA systems, this step may require additional coordination. We also recommend including interdisciplinary team members, such as social workers, case managers, and the relevant home care or hospice agency. Certain agencies may decline admission if they perceive increased risk, such as no 24-hour care, perceived self-neglect, and limited instrumental support. During this meeting, HCPs discuss risk mitigation strategies identified in Step 4 and create a plan to propose to patients and families.
Step 6: Meet with patient, HCA, and family members. In addition to sharing information about prognosis, assessing caregiver capabilities and burden can guide conversations about discharge. The discharge plan should be determined through shared decision making.11 If the patient lacks capacity regarding disposition planning, this should be shared with the HCA. However, even when patients lack capacity, it is important to continue to engage them to understand their goals and preferences.
Step 7: Maximize risk mitigation strategies. If a moderate- or high-risk discharge is requested, the health care team should maximize risk mitigation strategies. For low-risk discharges, risk mitigation strategies can still promote safety, especially since risk increases as patients progress toward EOL. In some instances, patients, their HCAs, or caregivers may decline all risk mitigation strategies despite best efforts to communicate and negotiate options. In such circumstances, we recommend discussing the case with the outpatient team for a warm handoff. HCPs should also document all efforts (helpful from a legal standpoint as well as for the patient’s future treatment teams) and respect the decision to discharge home.
Applying the Framework
Our patient Joe provides a good illustration of how to implement this EOL framework. He was deemed to have the capacity to make decisions regarding discharge (Step 1). We determined his risk factors and protective factors for discharge (Step 2). His poor functional status, limited instrumental support, heavy alcohol use, rejection of home services, and communication barriers due to severe hearing impairment all increased his risk. Protective factors included an appreciation of functional limitations, intact cognition, and an involved HCA. Based on his limited instrumental support and poor function but good insight into limitations, discharge home was deemed to be of moderate risk (Step 3). Although risk factors such as alcohol use and severe hearing impairment could have raised his level to high risk, we felt that his involved HCA maintained him in the moderate-risk category.
We worked with the hospitalist team, PT, and audiology to identify multiple risk mitigation strategies: frequent phone calls between the HCA and outpatient palliative care team, home PT to improve transfers from bed to bedside commode, home nursing services either through a routine agency or hospice, and hearing aids for better communication (Steps 4 and 5). We then proposed these strategies to Joe and his HCA (Step 6). Due to concerns about infringement on his independence, Joe declined all home services but agreed to twice-daily check-ins by his HCA, frequent communication between his HCA and our team, and new hearing aids.
Joe returned home with the agreed-upon risk mitigation strategies in place (Step 7). Despite clinicians’ original reservations about sending Joe home without formal services, his HCA maintained close contact with our team, noting that Joe remained stable and happy to be at home in the months following discharge.
Conclusions
Fortunately, VA HCPs operate in an integrated health care system with access to psychological, social, and at-home medical support that can help mitigate risks. Still, we have benefitted from having a tool to help us evaluate risk systematically. Even if patients, families, and HCPs disagree on ideal discharge plans, this tool helps clinicians approach discharges methodically while maintaining open communication and partnership with patients. In doing so, our framework reflects the shift in medical culture from a patriarchal approach to shared decision-making practices regarding all aspects of medical care. Furthermore, we hope that this can help reduce clinician moral distress stemming from these challenging cases.
Future research on best practices for discharge risk assessment and optimizing home safety are needed. We also hope to evaluate the impact and effectiveness of our framework through interviews with key stakeholders. For Joe and other veterans like him, where to spend their final days may be the last important decision they make in life, and our framework allows for their voices to be better heard throughout the decision-making process.
Acknowledgments
We thank Brooke Lifland, MD, for her theoretical contributions to the concept behind this paper.
Sometimes a patient at the end of life (EOL) just wants to go home. We recently treated such a patient, “Joe,” a 66-year-old veteran with end-stage chronic obstructive pulmonary disorder (COPD), severe hearing loss, and heavy alcohol use. A neighbor brought Joe to the hospital when he developed a urinary tract infection. Before hospitalization, Joe spent his days in bed. His neighbor was his designated health care agent (HCA) and caregiver, dropping off meals and bringing Joe to medical appointments. Joe had no other social support. In the hospital, Joe could not participate in physical therapy (PT) evaluations due to severe dyspnea on exertion. He was recommended for home PT, a home health aide, and home nursing, but Joe declined these services out of concern for encroachment on his independence. Given his heavy alcohol use, limited support, and functional limitations, the hospitalist team felt that Joe would be best served in a skilled nursing facility. As the palliative care team, we were consulted and felt that he was eligible for hospice. Joe simply wanted to go home.
Many patients like Joe experience functional decline at EOL, leading to increased care needs and transitions between sites of care.1 Some hospitalized patients at EOL want to transition directly to home, but due to their limited functioning and social support, discharge home may be deemed unsafe by health care professionals (HCPs). Clinicians then face the difficult balancing act of honoring patient wishes and avoiding a bad outcome. For patients at EOL, issues of capacity and risk become particularly salient. Furthermore, the unique structure of the US Department of Veterans Affairs (VA) health system and the psychosocial needs of some veterans add additional considerations for complex EOL discharges.2
End-of-life Decision Making
While patients may express strong preferences regarding their health care, their decision-making ability may worsen as they approach EOL. Contributing factors include older age, effects of hospitalization, treatment adverse effects, and comorbidities, including cognitive impairment. Studies of terminally ill patients show high rates of impaired decisional capacity.3,4 It is critical to assess capacity as part of discharge planning. Even when patients have the capacity, families and caregivers have an important voice, since they are often instrumental in maintaining patients at home.
Defining Risk
Determining whether a discharge is risky or unsafe is highly subjective, with differing opinions among clinicians and between patients and clinicians.5-7 In a qualitative study by Coombs and colleagues, HCPs tended toward a risk-averse approach to discharge decisions, sometimes favoring discharge to care facilities despite patient preferences.6 This approach also reflects pressures from the health care system to decrease the length of stay and reduce readmissions, important metrics for patient care and cost containment. However, keeping patients hospitalized or in nursing facilities does not completely mitigate risks (eg, falls) and carries other hazards (eg, nosocomial infections), as highlighted during the COVID-19 pandemic.7,8 The prospect of malpractice lawsuits and HCP moral distress about perceived risky home situations can also understandably affect decision making.
At the same time, risk calculation changes depending on the patient’s clinical status and priorities. Coombs and colleagues found that in contrast to clinicians, patients nearing EOL are willing to accept increasing risks and suboptimal living conditions to remain at home.6 What may be intolerable for a younger, healthier patient with a long life expectancy may be acceptable for someone who is approaching EOL. In our framework, a risky home discharge at EOL is considered one in which other adverse events, such as falls or inadequate symptom management, are likely.
Ethical Considerations
Unsafe discharges are challenging in part because some of the pillars of medical ethics can conflict. Prior articles have analyzed the ethical concerns of unsafe discharges in detail.9-11 Briefly, when patients wish to return home against initial medical recommendations, treatment teams may focus on the principles of beneficence and nonmaleficence, as exemplified by the desire to minimize harm, and justice, in which clinicians consider resource allocation and risks that a home discharge poses to family members, caregivers, and home health professionals. However, autonomy is important to consider as well. The concept of dignity of risk highlights the imperative to respect others’ decisions even when they increase the chance of harm, particularly given the overall shift in medicine from paternalism to shared decision making.12 Accommodating patient choice in how and where health care is received allows patients to regain some control over their lives, thereby enhancing their quality of life and promoting patient dignity, especially in their remaining days.13
Discharge Risk Framework
Our risk assessment framework helps clinicians more objectively identify factors that increase or decrease risk, inform discharge planning, partner with patients and families, give patients a prominent role in EOL decisions, and mitigate the risk of a bad outcome. This concept has been used in psychiatry, in which formal suicide assessment includes identifying risk factors and protective factors to estimate suicide risk and determine interventions.14 Similar to suicide risk estimation, this framework is based on clinical judgment rather than a specific calculation.
While this framework serves as a guide for determining and mitigating risk, we encourage teams to consider legal or ethical consultations in challenging cases, such as those in which patients lack both capacity and an involved HCA.
Step 1: Determine the patient’s capacity regarding disposition planning. Patients at EOL are at a higher risk of impaired decision-making capabilities; therefore, capacity evaluation is a critical step.
Step 2: Identify risk factors and protective factors for discharge home. Risk factors are intrinsic and extrinsic factors that increase risk such as functional or sensory impairments. Protective factors are intrinsic and extrinsic factors that decrease risk, including a good understanding of illness and consistent connection with the health care system (Table 1).
Step 3: Determine discharge to home risk level based on identified risk factors and protective factors. Patients may be at low, moderate, or high risk of having an adverse event, such as a fall or inadequate symptom control (Table 2).
Step 4: Identify risk mitigation strategies. These should be tailored to the patient based on the factors identified in Step 2. Examples include home nursing and therapy, mental health treatment, a medical alert system, and frequent contact between the patient and health care team.
Step 5: Meet with inpatient and outpatient HCP teams. Meetings should include the primary care professional (PCP) or relevant subspecialist, such as an oncologist for patients with cancer. For veterans receiving care solely at a local VA medical center, this can be easier to facilitate, but for veterans who receive care through both VA and non-VA systems, this step may require additional coordination. We also recommend including interdisciplinary team members, such as social workers, case managers, and the relevant home care or hospice agency. Certain agencies may decline admission if they perceive increased risk, such as no 24-hour care, perceived self-neglect, and limited instrumental support. During this meeting, HCPs discuss risk mitigation strategies identified in Step 4 and create a plan to propose to patients and families.
Step 6: Meet with patient, HCA, and family members. In addition to sharing information about prognosis, assessing caregiver capabilities and burden can guide conversations about discharge. The discharge plan should be determined through shared decision making.11 If the patient lacks capacity regarding disposition planning, this should be shared with the HCA. However, even when patients lack capacity, it is important to continue to engage them to understand their goals and preferences.
Step 7: Maximize risk mitigation strategies. If a moderate- or high-risk discharge is requested, the health care team should maximize risk mitigation strategies. For low-risk discharges, risk mitigation strategies can still promote safety, especially since risk increases as patients progress toward EOL. In some instances, patients, their HCAs, or caregivers may decline all risk mitigation strategies despite best efforts to communicate and negotiate options. In such circumstances, we recommend discussing the case with the outpatient team for a warm handoff. HCPs should also document all efforts (helpful from a legal standpoint as well as for the patient’s future treatment teams) and respect the decision to discharge home.
Applying the Framework
Our patient Joe provides a good illustration of how to implement this EOL framework. He was deemed to have the capacity to make decisions regarding discharge (Step 1). We determined his risk factors and protective factors for discharge (Step 2). His poor functional status, limited instrumental support, heavy alcohol use, rejection of home services, and communication barriers due to severe hearing impairment all increased his risk. Protective factors included an appreciation of functional limitations, intact cognition, and an involved HCA. Based on his limited instrumental support and poor function but good insight into limitations, discharge home was deemed to be of moderate risk (Step 3). Although risk factors such as alcohol use and severe hearing impairment could have raised his level to high risk, we felt that his involved HCA maintained him in the moderate-risk category.
We worked with the hospitalist team, PT, and audiology to identify multiple risk mitigation strategies: frequent phone calls between the HCA and outpatient palliative care team, home PT to improve transfers from bed to bedside commode, home nursing services either through a routine agency or hospice, and hearing aids for better communication (Steps 4 and 5). We then proposed these strategies to Joe and his HCA (Step 6). Due to concerns about infringement on his independence, Joe declined all home services but agreed to twice-daily check-ins by his HCA, frequent communication between his HCA and our team, and new hearing aids.
Joe returned home with the agreed-upon risk mitigation strategies in place (Step 7). Despite clinicians’ original reservations about sending Joe home without formal services, his HCA maintained close contact with our team, noting that Joe remained stable and happy to be at home in the months following discharge.
Conclusions
Fortunately, VA HCPs operate in an integrated health care system with access to psychological, social, and at-home medical support that can help mitigate risks. Still, we have benefitted from having a tool to help us evaluate risk systematically. Even if patients, families, and HCPs disagree on ideal discharge plans, this tool helps clinicians approach discharges methodically while maintaining open communication and partnership with patients. In doing so, our framework reflects the shift in medical culture from a patriarchal approach to shared decision-making practices regarding all aspects of medical care. Furthermore, we hope that this can help reduce clinician moral distress stemming from these challenging cases.
Future research on best practices for discharge risk assessment and optimizing home safety are needed. We also hope to evaluate the impact and effectiveness of our framework through interviews with key stakeholders. For Joe and other veterans like him, where to spend their final days may be the last important decision they make in life, and our framework allows for their voices to be better heard throughout the decision-making process.
Acknowledgments
We thank Brooke Lifland, MD, for her theoretical contributions to the concept behind this paper.
1. Committee on Approaching Death: Addressing Key End of Life Issues; Institute of Medicine. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington (DC): National Academies Press (US); March 19, 2015.
2. Casarett D, Pickard A, Amos Bailey F, et al. Important aspects of end-of-life care among veterans: implications for measurement and quality improvement. J Pain Symptom Manage. 2008;35(2):115-125. doi:10.1016/j.jpainsymman.2007.03.008
3. Kolva E, Rosenfeld B, Brescia R, Comfort C. Assessing decision-making capacity at end of life. Gen Hosp Psychiatry. 2014;36(4):392-397. doi:10.1016/j.genhosppsych.2014.02.013
4. Kolva E, Rosenfeld B, Saracino R. Assessing the decision-making capacity of terminally ill patients with cancer. Am J Geriatr Psychiatry. 2018;26(5):523-531. doi:10.1016/j.jagp.2017.11.012
5. Macmillan MS. Hospital staff’s perceptions of risk associated with the discharge of elderly people from acute hospital care. J Adv Nurs. 1994;19(2):249-256. doi:10.1111/j.1365-2648.1994.tb01078.x
6. Coombs MA, Parker R, de Vries K. Managing risk during care transitions when approaching end of life: A qualitative study of patients’ and health care professionals’ decision making. Palliat Med. 2017;31(7):617-624. doi:10.1177/0269216316673476
7. Hyslop B. ‘Not safe for discharge’? Words, values, and person-centred care. Age Ageing. 2020;49(3):334-336. doi:10.1093/ageing/afz170
8. Goodacre S. Safe discharge: an irrational, unhelpful and unachievable concept. Emerg Med J. 2006;23(10):753-755. doi:10.1136/emj.2006.037903
9. Swidler RN, Seastrum T, Shelton W. Difficult hospital inpatient discharge decisions: ethical, legal and clinical practice issues. Am J Bioeth. 2007;7(3):23-28. doi:10.1080/15265160601171739
10. Hill J, Filer W. Safety and ethical considerations in discharging patients to suboptimal living situations. AMA J Ethics. 2015;17(6):506-510. Published 2015 Jun 1. doi:10.1001/journalofethics.2015.17.6.ecas2-1506
11. West JC. What is an ethically informed approach to managing patient safety risk during discharge planning?. AMA J Ethics. 2020;22(11):E919-E923. Published 2020 Nov 1. doi:10.1001/amajethics.2020.919
12. Mukherjee D. Discharge decisions and the dignity of risk. Hastings Cent Rep. 2015;45(3):7-8. doi:10.1002/hast.441
13. Wheatley VJ, Baker JI. “Please, I want to go home”: ethical issues raised when considering choice of place of care in palliative care. Postgrad Med J. 2007;83(984):643-648. doi:10.1136/pgmj.2007.058487
14. Work Group on Suicidal Behaviors. Practice guideline for the assessment and treatment of patients with suicidal behaviors. Am J Psychiatry. 2003;160(suppl 11):1-60.
1. Committee on Approaching Death: Addressing Key End of Life Issues; Institute of Medicine. Dying in America: Improving Quality and Honoring Individual Preferences Near the End of Life. Washington (DC): National Academies Press (US); March 19, 2015.
2. Casarett D, Pickard A, Amos Bailey F, et al. Important aspects of end-of-life care among veterans: implications for measurement and quality improvement. J Pain Symptom Manage. 2008;35(2):115-125. doi:10.1016/j.jpainsymman.2007.03.008
3. Kolva E, Rosenfeld B, Brescia R, Comfort C. Assessing decision-making capacity at end of life. Gen Hosp Psychiatry. 2014;36(4):392-397. doi:10.1016/j.genhosppsych.2014.02.013
4. Kolva E, Rosenfeld B, Saracino R. Assessing the decision-making capacity of terminally ill patients with cancer. Am J Geriatr Psychiatry. 2018;26(5):523-531. doi:10.1016/j.jagp.2017.11.012
5. Macmillan MS. Hospital staff’s perceptions of risk associated with the discharge of elderly people from acute hospital care. J Adv Nurs. 1994;19(2):249-256. doi:10.1111/j.1365-2648.1994.tb01078.x
6. Coombs MA, Parker R, de Vries K. Managing risk during care transitions when approaching end of life: A qualitative study of patients’ and health care professionals’ decision making. Palliat Med. 2017;31(7):617-624. doi:10.1177/0269216316673476
7. Hyslop B. ‘Not safe for discharge’? Words, values, and person-centred care. Age Ageing. 2020;49(3):334-336. doi:10.1093/ageing/afz170
8. Goodacre S. Safe discharge: an irrational, unhelpful and unachievable concept. Emerg Med J. 2006;23(10):753-755. doi:10.1136/emj.2006.037903
9. Swidler RN, Seastrum T, Shelton W. Difficult hospital inpatient discharge decisions: ethical, legal and clinical practice issues. Am J Bioeth. 2007;7(3):23-28. doi:10.1080/15265160601171739
10. Hill J, Filer W. Safety and ethical considerations in discharging patients to suboptimal living situations. AMA J Ethics. 2015;17(6):506-510. Published 2015 Jun 1. doi:10.1001/journalofethics.2015.17.6.ecas2-1506
11. West JC. What is an ethically informed approach to managing patient safety risk during discharge planning?. AMA J Ethics. 2020;22(11):E919-E923. Published 2020 Nov 1. doi:10.1001/amajethics.2020.919
12. Mukherjee D. Discharge decisions and the dignity of risk. Hastings Cent Rep. 2015;45(3):7-8. doi:10.1002/hast.441
13. Wheatley VJ, Baker JI. “Please, I want to go home”: ethical issues raised when considering choice of place of care in palliative care. Postgrad Med J. 2007;83(984):643-648. doi:10.1136/pgmj.2007.058487
14. Work Group on Suicidal Behaviors. Practice guideline for the assessment and treatment of patients with suicidal behaviors. Am J Psychiatry. 2003;160(suppl 11):1-60.
Where Have All the Future Veterans Gone?
Word to the Nation: Guard zealously your right to serve in the Armed Forces, for without them, there will be no other rights to guard.
John F. Kennedy 1
The title of this Veterans Day editorial is a paraphrase of the legendary folk artist Pete Seeger’s protest song popularized during the Vietnam War. On January 27, 1973, in the wake of the widespread antiwar movement, Secretary of Defense Melvin Laird announced an end to the dreaded draft.2
For nearly 50 years, the all-volunteer military was celebrated as an outstanding achievement that professionalized the armed services and arguably made the US military among the most highly trained and effective fighting forces in the world. That was until an ongoing recruitment crisis threatened to write a different and far more disturbing conclusion to what the government had heralded as a “success story.”3
The recruiting crisis is a complicated problem with many facets that have received increasing attention from journalists, the media, experts, think tanks, and the government. Given this complexity, this will be a 2-part editorial: This column examines the scope of the crisis and the putative causes of the problem with recruiting Americans to serve in uniform. The next column will examine the potential impact of the shortage of service members on federal health care practice.
The Recruiting Crisis
Over the past several years, nearly every branch of the armed forces has struggled with recruitment, especially the Army. In April of this year, the US Department of Defense (DoD) reported that the Army, Navy, and Air Force would all fail to meet recruitment goals; only the Marines and Space Forces were expected to reach their targets.4 At the end of its fiscal year (October 1), the Army acknowledged that its 55,000 recruits were 10,000 fewer soldiers than it had aimed to enlist.5 But this was still more people joining the ranks than in 2022 when the Army was 15,000 recruits below the mark.6
Challenging Trends
There are many putative causes and proposed solutions for the recruitment crisis. Among the most serious is a marked drop in the American public’s confidence in the military. A June 2023 Gallup poll found that only 60% of citizens expressed “a great deal” or “quite a lot” of confidence in the military. This was the nadir of a 5-year decline that this year reached the lowest point since 1997/1998.7 For many Americans in and out of uniform, the ignoble end to the long war in Afghanistan leaving behind friends and allies contrary to the military ethos is cited as a significant contributor to both the loss of confidence in the military and the recruiting crisis.8
These cultural developments reinforce each other. Now, many veterans do not want their relatives and friends to follow them into the armed services. A 2021 survey by the Military Family Advisory Network found that slightly more than 60% of veterans and active-duty service members would recommend a military career to a potential recruit. This was down from 75% in 2019.9 Veterans cite a variety of reasons for discouraging their fellow citizens from serving, including low pay compared with civilian employment, especially in a labor-hungry job market; and the military failure to fulfill health care promises, housing, and other social services, especially for the growing number experiencing mental health disorders related to their service.10
Two facts about recruitment heighten the negative impact of some veterans’ change of attitude toward joining the services. First, since the end of the draft, military life in the US has become a family tradition. Published in 2011, a Pew Research Center study found that even then, a decreasing number of Americans had a family connection to the military. More respondents aged ≥ 50 years had a parent, child, spouse, or sibling who had served compared with those aged 30 to 49 years and those aged 18 to 29 (77%, 57%, and 33%, respectively).11 Second, since the end of the draft, far fewer Americans have had military experience. Only 1% of the nation is currently in military service, and the veteran population is steadily declining. In 1980, 18% of adult Americans were veterans; 20 years later, that number is only 7%.12 This makes it less likely that a high school or college student will have a personal or even a passing relationship with a teacher, coach, or other mentoring adult who is or has been a military member. This demographic discrepancy has generated what sociologists call the military-civilian gap.10 That division has been manipulated in the increasingly vehement culture wars and generational struggles that are splitting the country.12
This relatively recent sociological trend is reflected in a growing lack of interest among many young Americans in armed forces service. A DoD survey of participants aged 16 to 24 years regarding their intention to serve in the military found that 89% were probably not going to pursue a career in uniform. More than 65% of respondents indicated that the possibility of physical injury, death, or psychological trauma was the primary deterrent for considering enlisting.13 The latter barrier is directly related to our work as practitioners caring for service members and veterans, and through our compassion and competence, we may help bridge the widening divide between the military and civilian spheres. These numbers speak to the unwilling; there is also a significant group of Americans who want to serve yet are unable to due to their history, diagnoses, or condition.14 Their motivation to be military members in the face of the recruitment challenges highlighted here present federal practitioners with ethical questions that will be the subject of the next column.
Armed Forces and Veterans Day
This column’s epigraph is from President John F. Kennedy, a decorated World War II Navy combat veteran who decreed Armed Forces Day an official holiday a decade before conscription ended.1 The commemoration was to thank and honor all individuals currently serving in the military for their patriotism and sacrifice. President Kennedy’s Word to the Nation could not be timelier on Veterans Day 2023. The data reviewed here raise profound questions as to where tomorrow’s service members and the veterans of the future will come from, and how we will persuade them that though there are real risks to military service, the rewards are both tangible and transcendent.
1. US Department of Defense. Armed Forces Day. Accessed October 17, 2023. https://afd.defense.gov/History
2. Zipkin A. The military draft ended 50 years ago, dividing a generation. The Washington Post. January 27, 2023. Accessed October 17, 2023. https://www.washingtonpost.com/history/2023/01/27/draft-end-conscription-1973
3. Lopez TC. All-volunteer force proves successful for U.S. military. March 2, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3316678/all-volunteer-force-proves-successful-for-us-military
4. Garamone J. Vice-chiefs talk recruiting shortfalls, readiness issues. April 20, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3369472/vice-chiefs-talk-recruiting-shortfalls-readiness-issues
5. Winkie D. Army recruiters at two-thirds of contract goals as the fiscal year closes. Military Times. September 7, 2023. Accessed October 17, 2023. https://www.armytimes.com/news/recruiting/2023/09/07/army-recruiters-at-two-thirds-of-contract-goals-as-fiscal-year-closes
6. Baldor LC. Army misses recruiting goal by 15,000 soldiers. Accessed October 17, 2023. https://www.armytimes.com/news/your-army/2022/10/02/army-misses-recruiting-goal-by-15000-soldiers
7. Younis M. Confidence in U.S. military lowest in over two decades. Accessed October 17, 2023. https://news.gallup.com/poll/509189/confidence-military-lowest-two-decades.aspx
8. Rogin A, Corkery A. Why recruiting and confidence in America’s armed forces is so low right now? Accessed October 17, 2023. https://www.pbs.org/newshour/show/why-recruiting-and-confidence-in-americas-armed-forces-is-so-low-right-now
9. Military Family Advisory Network. 2021 military family support programming survey. Accessed October 17, 2023. https://www.mfan.org/wp-content/uploads/2022/07/Executive-Summary-MFAN-Programming-Survey-Results-2021.pdf
10. Kesling B. The military recruiting crisis: even veterans don’t want their family to join. Wall Street Journal. 30 June 2023. Accessed October 17, 2023. https://www.wsj.com/articles/military-recruiting-crisis-veterans-dont-want-their-children-to-join-510e1a25
11. Pew Research Center. The military-civilian gap: fewer family connections. Accessed October 17, 2023. https://www.pewresearch.org/social-trends/2011/11/23/the-military-civilian-gap-fewer-family-connections
12. Myers M. Is the military too ‘woke’ to recruit? Accessed October 17, 2023. https://www.militarytimes.com/news/your-military/2022/10/13/is-the-military-too-woke-to-recruit
13. Schaeffer K. The changing face of America’s veteran population. Accessed October 17, 2023. https://www.pewresearch.org/short-reads/2021/04/05/the-changing-face-of-americas-veteran-population
14. Phillips D. With few able and fewer willing, U.S. military can’t find recruits. New York Times. July 14, 2023. Accessed October 17, 2023. https://www.nytimes.com/2022/07/14/us/us-military-recruiting-enlistment.html
Word to the Nation: Guard zealously your right to serve in the Armed Forces, for without them, there will be no other rights to guard.
John F. Kennedy 1
The title of this Veterans Day editorial is a paraphrase of the legendary folk artist Pete Seeger’s protest song popularized during the Vietnam War. On January 27, 1973, in the wake of the widespread antiwar movement, Secretary of Defense Melvin Laird announced an end to the dreaded draft.2
For nearly 50 years, the all-volunteer military was celebrated as an outstanding achievement that professionalized the armed services and arguably made the US military among the most highly trained and effective fighting forces in the world. That was until an ongoing recruitment crisis threatened to write a different and far more disturbing conclusion to what the government had heralded as a “success story.”3
The recruiting crisis is a complicated problem with many facets that have received increasing attention from journalists, the media, experts, think tanks, and the government. Given this complexity, this will be a 2-part editorial: This column examines the scope of the crisis and the putative causes of the problem with recruiting Americans to serve in uniform. The next column will examine the potential impact of the shortage of service members on federal health care practice.
The Recruiting Crisis
Over the past several years, nearly every branch of the armed forces has struggled with recruitment, especially the Army. In April of this year, the US Department of Defense (DoD) reported that the Army, Navy, and Air Force would all fail to meet recruitment goals; only the Marines and Space Forces were expected to reach their targets.4 At the end of its fiscal year (October 1), the Army acknowledged that its 55,000 recruits were 10,000 fewer soldiers than it had aimed to enlist.5 But this was still more people joining the ranks than in 2022 when the Army was 15,000 recruits below the mark.6
Challenging Trends
There are many putative causes and proposed solutions for the recruitment crisis. Among the most serious is a marked drop in the American public’s confidence in the military. A June 2023 Gallup poll found that only 60% of citizens expressed “a great deal” or “quite a lot” of confidence in the military. This was the nadir of a 5-year decline that this year reached the lowest point since 1997/1998.7 For many Americans in and out of uniform, the ignoble end to the long war in Afghanistan leaving behind friends and allies contrary to the military ethos is cited as a significant contributor to both the loss of confidence in the military and the recruiting crisis.8
These cultural developments reinforce each other. Now, many veterans do not want their relatives and friends to follow them into the armed services. A 2021 survey by the Military Family Advisory Network found that slightly more than 60% of veterans and active-duty service members would recommend a military career to a potential recruit. This was down from 75% in 2019.9 Veterans cite a variety of reasons for discouraging their fellow citizens from serving, including low pay compared with civilian employment, especially in a labor-hungry job market; and the military failure to fulfill health care promises, housing, and other social services, especially for the growing number experiencing mental health disorders related to their service.10
Two facts about recruitment heighten the negative impact of some veterans’ change of attitude toward joining the services. First, since the end of the draft, military life in the US has become a family tradition. Published in 2011, a Pew Research Center study found that even then, a decreasing number of Americans had a family connection to the military. More respondents aged ≥ 50 years had a parent, child, spouse, or sibling who had served compared with those aged 30 to 49 years and those aged 18 to 29 (77%, 57%, and 33%, respectively).11 Second, since the end of the draft, far fewer Americans have had military experience. Only 1% of the nation is currently in military service, and the veteran population is steadily declining. In 1980, 18% of adult Americans were veterans; 20 years later, that number is only 7%.12 This makes it less likely that a high school or college student will have a personal or even a passing relationship with a teacher, coach, or other mentoring adult who is or has been a military member. This demographic discrepancy has generated what sociologists call the military-civilian gap.10 That division has been manipulated in the increasingly vehement culture wars and generational struggles that are splitting the country.12
This relatively recent sociological trend is reflected in a growing lack of interest among many young Americans in armed forces service. A DoD survey of participants aged 16 to 24 years regarding their intention to serve in the military found that 89% were probably not going to pursue a career in uniform. More than 65% of respondents indicated that the possibility of physical injury, death, or psychological trauma was the primary deterrent for considering enlisting.13 The latter barrier is directly related to our work as practitioners caring for service members and veterans, and through our compassion and competence, we may help bridge the widening divide between the military and civilian spheres. These numbers speak to the unwilling; there is also a significant group of Americans who want to serve yet are unable to due to their history, diagnoses, or condition.14 Their motivation to be military members in the face of the recruitment challenges highlighted here present federal practitioners with ethical questions that will be the subject of the next column.
Armed Forces and Veterans Day
This column’s epigraph is from President John F. Kennedy, a decorated World War II Navy combat veteran who decreed Armed Forces Day an official holiday a decade before conscription ended.1 The commemoration was to thank and honor all individuals currently serving in the military for their patriotism and sacrifice. President Kennedy’s Word to the Nation could not be timelier on Veterans Day 2023. The data reviewed here raise profound questions as to where tomorrow’s service members and the veterans of the future will come from, and how we will persuade them that though there are real risks to military service, the rewards are both tangible and transcendent.
Word to the Nation: Guard zealously your right to serve in the Armed Forces, for without them, there will be no other rights to guard.
John F. Kennedy 1
The title of this Veterans Day editorial is a paraphrase of the legendary folk artist Pete Seeger’s protest song popularized during the Vietnam War. On January 27, 1973, in the wake of the widespread antiwar movement, Secretary of Defense Melvin Laird announced an end to the dreaded draft.2
For nearly 50 years, the all-volunteer military was celebrated as an outstanding achievement that professionalized the armed services and arguably made the US military among the most highly trained and effective fighting forces in the world. That was until an ongoing recruitment crisis threatened to write a different and far more disturbing conclusion to what the government had heralded as a “success story.”3
The recruiting crisis is a complicated problem with many facets that have received increasing attention from journalists, the media, experts, think tanks, and the government. Given this complexity, this will be a 2-part editorial: This column examines the scope of the crisis and the putative causes of the problem with recruiting Americans to serve in uniform. The next column will examine the potential impact of the shortage of service members on federal health care practice.
The Recruiting Crisis
Over the past several years, nearly every branch of the armed forces has struggled with recruitment, especially the Army. In April of this year, the US Department of Defense (DoD) reported that the Army, Navy, and Air Force would all fail to meet recruitment goals; only the Marines and Space Forces were expected to reach their targets.4 At the end of its fiscal year (October 1), the Army acknowledged that its 55,000 recruits were 10,000 fewer soldiers than it had aimed to enlist.5 But this was still more people joining the ranks than in 2022 when the Army was 15,000 recruits below the mark.6
Challenging Trends
There are many putative causes and proposed solutions for the recruitment crisis. Among the most serious is a marked drop in the American public’s confidence in the military. A June 2023 Gallup poll found that only 60% of citizens expressed “a great deal” or “quite a lot” of confidence in the military. This was the nadir of a 5-year decline that this year reached the lowest point since 1997/1998.7 For many Americans in and out of uniform, the ignoble end to the long war in Afghanistan leaving behind friends and allies contrary to the military ethos is cited as a significant contributor to both the loss of confidence in the military and the recruiting crisis.8
These cultural developments reinforce each other. Now, many veterans do not want their relatives and friends to follow them into the armed services. A 2021 survey by the Military Family Advisory Network found that slightly more than 60% of veterans and active-duty service members would recommend a military career to a potential recruit. This was down from 75% in 2019.9 Veterans cite a variety of reasons for discouraging their fellow citizens from serving, including low pay compared with civilian employment, especially in a labor-hungry job market; and the military failure to fulfill health care promises, housing, and other social services, especially for the growing number experiencing mental health disorders related to their service.10
Two facts about recruitment heighten the negative impact of some veterans’ change of attitude toward joining the services. First, since the end of the draft, military life in the US has become a family tradition. Published in 2011, a Pew Research Center study found that even then, a decreasing number of Americans had a family connection to the military. More respondents aged ≥ 50 years had a parent, child, spouse, or sibling who had served compared with those aged 30 to 49 years and those aged 18 to 29 (77%, 57%, and 33%, respectively).11 Second, since the end of the draft, far fewer Americans have had military experience. Only 1% of the nation is currently in military service, and the veteran population is steadily declining. In 1980, 18% of adult Americans were veterans; 20 years later, that number is only 7%.12 This makes it less likely that a high school or college student will have a personal or even a passing relationship with a teacher, coach, or other mentoring adult who is or has been a military member. This demographic discrepancy has generated what sociologists call the military-civilian gap.10 That division has been manipulated in the increasingly vehement culture wars and generational struggles that are splitting the country.12
This relatively recent sociological trend is reflected in a growing lack of interest among many young Americans in armed forces service. A DoD survey of participants aged 16 to 24 years regarding their intention to serve in the military found that 89% were probably not going to pursue a career in uniform. More than 65% of respondents indicated that the possibility of physical injury, death, or psychological trauma was the primary deterrent for considering enlisting.13 The latter barrier is directly related to our work as practitioners caring for service members and veterans, and through our compassion and competence, we may help bridge the widening divide between the military and civilian spheres. These numbers speak to the unwilling; there is also a significant group of Americans who want to serve yet are unable to due to their history, diagnoses, or condition.14 Their motivation to be military members in the face of the recruitment challenges highlighted here present federal practitioners with ethical questions that will be the subject of the next column.
Armed Forces and Veterans Day
This column’s epigraph is from President John F. Kennedy, a decorated World War II Navy combat veteran who decreed Armed Forces Day an official holiday a decade before conscription ended.1 The commemoration was to thank and honor all individuals currently serving in the military for their patriotism and sacrifice. President Kennedy’s Word to the Nation could not be timelier on Veterans Day 2023. The data reviewed here raise profound questions as to where tomorrow’s service members and the veterans of the future will come from, and how we will persuade them that though there are real risks to military service, the rewards are both tangible and transcendent.
1. US Department of Defense. Armed Forces Day. Accessed October 17, 2023. https://afd.defense.gov/History
2. Zipkin A. The military draft ended 50 years ago, dividing a generation. The Washington Post. January 27, 2023. Accessed October 17, 2023. https://www.washingtonpost.com/history/2023/01/27/draft-end-conscription-1973
3. Lopez TC. All-volunteer force proves successful for U.S. military. March 2, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3316678/all-volunteer-force-proves-successful-for-us-military
4. Garamone J. Vice-chiefs talk recruiting shortfalls, readiness issues. April 20, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3369472/vice-chiefs-talk-recruiting-shortfalls-readiness-issues
5. Winkie D. Army recruiters at two-thirds of contract goals as the fiscal year closes. Military Times. September 7, 2023. Accessed October 17, 2023. https://www.armytimes.com/news/recruiting/2023/09/07/army-recruiters-at-two-thirds-of-contract-goals-as-fiscal-year-closes
6. Baldor LC. Army misses recruiting goal by 15,000 soldiers. Accessed October 17, 2023. https://www.armytimes.com/news/your-army/2022/10/02/army-misses-recruiting-goal-by-15000-soldiers
7. Younis M. Confidence in U.S. military lowest in over two decades. Accessed October 17, 2023. https://news.gallup.com/poll/509189/confidence-military-lowest-two-decades.aspx
8. Rogin A, Corkery A. Why recruiting and confidence in America’s armed forces is so low right now? Accessed October 17, 2023. https://www.pbs.org/newshour/show/why-recruiting-and-confidence-in-americas-armed-forces-is-so-low-right-now
9. Military Family Advisory Network. 2021 military family support programming survey. Accessed October 17, 2023. https://www.mfan.org/wp-content/uploads/2022/07/Executive-Summary-MFAN-Programming-Survey-Results-2021.pdf
10. Kesling B. The military recruiting crisis: even veterans don’t want their family to join. Wall Street Journal. 30 June 2023. Accessed October 17, 2023. https://www.wsj.com/articles/military-recruiting-crisis-veterans-dont-want-their-children-to-join-510e1a25
11. Pew Research Center. The military-civilian gap: fewer family connections. Accessed October 17, 2023. https://www.pewresearch.org/social-trends/2011/11/23/the-military-civilian-gap-fewer-family-connections
12. Myers M. Is the military too ‘woke’ to recruit? Accessed October 17, 2023. https://www.militarytimes.com/news/your-military/2022/10/13/is-the-military-too-woke-to-recruit
13. Schaeffer K. The changing face of America’s veteran population. Accessed October 17, 2023. https://www.pewresearch.org/short-reads/2021/04/05/the-changing-face-of-americas-veteran-population
14. Phillips D. With few able and fewer willing, U.S. military can’t find recruits. New York Times. July 14, 2023. Accessed October 17, 2023. https://www.nytimes.com/2022/07/14/us/us-military-recruiting-enlistment.html
1. US Department of Defense. Armed Forces Day. Accessed October 17, 2023. https://afd.defense.gov/History
2. Zipkin A. The military draft ended 50 years ago, dividing a generation. The Washington Post. January 27, 2023. Accessed October 17, 2023. https://www.washingtonpost.com/history/2023/01/27/draft-end-conscription-1973
3. Lopez TC. All-volunteer force proves successful for U.S. military. March 2, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3316678/all-volunteer-force-proves-successful-for-us-military
4. Garamone J. Vice-chiefs talk recruiting shortfalls, readiness issues. April 20, 2023. Accessed October 17, 2023. https://www.defense.gov/News/News-Stories/Article/Article/3369472/vice-chiefs-talk-recruiting-shortfalls-readiness-issues
5. Winkie D. Army recruiters at two-thirds of contract goals as the fiscal year closes. Military Times. September 7, 2023. Accessed October 17, 2023. https://www.armytimes.com/news/recruiting/2023/09/07/army-recruiters-at-two-thirds-of-contract-goals-as-fiscal-year-closes
6. Baldor LC. Army misses recruiting goal by 15,000 soldiers. Accessed October 17, 2023. https://www.armytimes.com/news/your-army/2022/10/02/army-misses-recruiting-goal-by-15000-soldiers
7. Younis M. Confidence in U.S. military lowest in over two decades. Accessed October 17, 2023. https://news.gallup.com/poll/509189/confidence-military-lowest-two-decades.aspx
8. Rogin A, Corkery A. Why recruiting and confidence in America’s armed forces is so low right now? Accessed October 17, 2023. https://www.pbs.org/newshour/show/why-recruiting-and-confidence-in-americas-armed-forces-is-so-low-right-now
9. Military Family Advisory Network. 2021 military family support programming survey. Accessed October 17, 2023. https://www.mfan.org/wp-content/uploads/2022/07/Executive-Summary-MFAN-Programming-Survey-Results-2021.pdf
10. Kesling B. The military recruiting crisis: even veterans don’t want their family to join. Wall Street Journal. 30 June 2023. Accessed October 17, 2023. https://www.wsj.com/articles/military-recruiting-crisis-veterans-dont-want-their-children-to-join-510e1a25
11. Pew Research Center. The military-civilian gap: fewer family connections. Accessed October 17, 2023. https://www.pewresearch.org/social-trends/2011/11/23/the-military-civilian-gap-fewer-family-connections
12. Myers M. Is the military too ‘woke’ to recruit? Accessed October 17, 2023. https://www.militarytimes.com/news/your-military/2022/10/13/is-the-military-too-woke-to-recruit
13. Schaeffer K. The changing face of America’s veteran population. Accessed October 17, 2023. https://www.pewresearch.org/short-reads/2021/04/05/the-changing-face-of-americas-veteran-population
14. Phillips D. With few able and fewer willing, U.S. military can’t find recruits. New York Times. July 14, 2023. Accessed October 17, 2023. https://www.nytimes.com/2022/07/14/us/us-military-recruiting-enlistment.html
How VA Innovative Partnerships and Health Care Systems Can Respond to National Needs: NOSE Trial Example
Traditional manufacturing concentrates capacity into a few discrete locations while applying lean and just-in-time philosophies to maximize profit during times of somewhat predictable supply and demand. This approach exposed nationwide vulnerabilities even during local crises, such as the United States saline shortages following closure of a single plant in Puerto Rico following Hurricane Maria in 2017.1 Interruptions to the supply chain due to pandemic plant closure, weather, politics, or surge demand can cause immediate and lasting shortages. Nasal swabs were a clear example.
At the onset of COVID-19, 2 companies—Puritan in Guilford, Maine, and Copan in Italy—manufactured nearly all of the highly specialized nasopharyngeal (NP) swabs singled out by the Centers for Disease Control and Prevention (CDC) and the US Food and Drug Administration (FDA) to test patients for COVID-19. Demand for swabs skyrocketed as the virus spread, and they became unattainable. The lack of swabs meant patients went undiagnosed. Without knowing who was positive, people with symptoms and known contacts were presumed positive and quarantined, impacting isolated patients, the health care professionals treating them, and the entire US economy.
3-Dimensional Printing Solutions
Manufacturing NP swabs is not trivial. Their simple shape conceals complexity and requires highly specialized equipment. The lead time for one non-US machine manufacturer was > 6 months at the start of the pandemic.
Digital manufacturing/3-dimensional (3D) printing represented a potential solution to the supply chain crisis.2 Designers created digital blueprints for 3D-printed goods, face masks, face shields, and ventilator splitters were rapidly created and shared.3,4 Scrambling to fill the critical need for NP swabs, many hospitals, businesses, and academic centers began 3D printing swabs. This effort was spearheaded by University of South Florida (USF) and Northwell Health researchers and clinicians, who designed and tested a 3D-printed NP swab from photocurable resin that was printable on 2 models of Formlabs printers.5 Several other 3D-printed NP swab designs soon followed. This innovation and problem-solving renaissance faced several challenges well known to traditional manufacturers of regulated products but novel to newcomers.
The first challlenge was that these NP swabs predate FDA oversight of medical device development and manufacturing and no testing standards existed. Designers began casting prototypes out without guidance about the critical features and clinical functions required. Many of these designs did not have a clinical evaluation pathway to test safety and efficacy.
The second challlenge was that these swabs were being produced by facilities not registered with the FDA. This raised concerns about the quality of unlisted medical products developed and manufactured at novel facilities.
The third challenge was that small-scale novel approaches may offset local shortages but could not address national needs. The self-organized infrastructure for this crisis was ad hoc, local, and lacked coordinated federal support. This led to rolling shortages of these materials for years.
Two studies were performed early in the pandemic. The first study evaluated 4 prototypes of different manufacturer designs, finding excellent concordance among them and their control swab.6 A second study demonstrated the USF swab to be noninferior to the standard of care.7 Both studies acknowledged and addressed the first challenge for their designs.
COLLABORATIONS
Interagency
Before the pandemic, the US Department of Veterans Affairs (VA) had been coordinating with the FDA, the National Institutes of Health (NIH), and the nonprofit America Makes to bring medical product development and manufacturing closer to the point of care.
At the outset of the COVID-19 pandemic, the collaboration was formalized to address new challenges.8 The objectives of this collaboration were the following: (1) host a digital repository for 3D-printed digital designs for personal protectice equipment and other medical supplies in or at risk of shortage; (2) provide scientifically based ratings for designs according to clinical and field testing; and (3) offer education to health care workers and the public about the digital manufacturing of medical goods and devices.4,9
A key output of this collaboration was the COVID 3D Trusted Repository For Users And Suppliers Through Testing (COVID 3D TRUST), a curated archive of designs. In most cases, existing FDA standards and guidance formed the basis of testing strategies with deviations due to limited access to traditional testing facilities and reagents.
To address novel NP swabs, working with its COVID 3D TRUST partners, the VA gathered a combined list of clinical- and engineering-informed customer requirements and performed a hazard analysis. The result was a list of design inputs for NP swabs and 8 standard test protocols to evaluate key functions (Table).10 These protocols are meant to benchmark novel 3D-printed swabs against the key functions of established, traditionally manufactured swabs, which have a long record of safety and efficacy. The protocols, developed by the VA and undergoing validation by the US Army, empower and inform consumers and provide performance metrics to swab designers and manufacturers. The testing protocols and preliminary test results developed by the VA are publicly available at the NIH.11
Intra-agency
The use of the inputs and verification tests noted in the Table may reduce the risk of poor design but were inadequate to evaluate the clinical safety and efficacy of novel swabs. Recognizing this, the VA Office of Healthcare Innovation and Learning (OHIL) and the Office of Research and Development (ORD) launched the Nasal Swab Objective and Statistical Evaluation (NOSE) study to formally evaluate the safety and efficacy of 3D-printed swabs in the field. This multisite clinical study was a close collaboration between the OHIL and ORD. The OHIL provided the quality system and manufacturing oversight and delivery of the swabs, and the ORD provided scientific review, research infrastructure, human subjects oversight, administrative support, and funding and fiscal oversight. The OHIL/ORD collaboration resulted in the successful completion of the NOSE study.
This study (manuscript under preparation) yielded two 3D-printing production processes and swab designs that had comparable performance to the standard of care, were manufacturable compliant with FDA guidelines, and could be produced at scale in a distributed manner. This approach directly addressed the 3 challenges described earlier.
LESSONS LEARNED
Swabs were an example of supply challenges in the pandemic, but advanced manufacturing (notably, digital designs leading to 3D-printed solutions) also served as a temporary solution to device and product shortages during the COVID-19 pandemic. Digital designs and 3D printing as manufacturing techniques have the following key advantages: (1) they are distributed in nature, both in the breadth of locations that have access to these manufacturing platforms and in the depth of material choice that can be used to fabricate products, which alleviates the threat of a disaster impacting manufacturing capacity or a material stream; (2) they do not require retooling of machinery so new products can deploy rapidly and on demand; and (3) the speed of digital iteration, printing, and revision allows for rapid product development and production.
There also are notable disadvantages to these techniques. First, because 3D printing is a newer technology, there is less general depth of knowledge regarding design and material choice for additive manufacturing. Second, the flexibility of 3D printing means that operators must increase awareness of the factors that might cause the fabrication of a part to fail in either printing or postprocessing. Third, there are significant gaps in understanding how materials and manufacturing processes will perform in high-stakes settings such as health care, where performance and biocompatibility may be critical to support life-sustaining functions. Fourth, digital files are vulnerable to intentional or unintentional alteration. These alterations might weaken design integrity and be imperceptible to the manufacturer or end user. This is a prevalent challenge in all open-source designs.
The pandemic materialized quickly and created vast supply chain challenges. To address this crisis, it was clear that the average 17-year interval between research and translation in the US was unacceptable. The VA was able to accelerate swiftly many existing processes to meet this need, build new capabilities, and establish new practices for the rapid evaluation and deployment of health care products and guidance. This agile and innovative cooperation was critical in the success of the VA’s national support for pandemic solutions.
Finally, although COVID 3D TRUST was able to provide testing of submitted designs, this collaboration was not a substitute for the “peacetime” process of manufacturing site registration with the FDA and product listing. COVID 3D TRUST could evaluate designs only, not the production process, safety, and efficacy.
CALLS TO ACTION
The pandemic's impact on medical supply chain security persists, as does the need for greater foresight and crisis preparation. We must act now to avoid experiencing again the magnitude of fatalities (civilian and veteran) and the devastation to the US economy and livelihoods that occurred during this single biological event. To this end, creating a digital stockpile of federally curated, crisis-ready designs for as-needed distribution across our US industrial base would offer a second line of defense against life-threatening supply chain interruptions. The realization of such a digital stockpile requires calls to action among multiple contributors.
Collaborations
The VA’s Fourth Mission is to improve the nation’s preparedness for response to war, terrorism, national emergencies, and natural disasters. The VA does this by developing plans and taking actions to ensure continued service to veterans, as well as to support national, state, and local emergency management, public health, safety, and homeland security efforts.
The VA partnership with the FDA and NIH during the pandemic enabled successful coordination among federal agencies. Numerous other agencies, including the US Department of Defense (DoD), the Biomedical Advanced Research and Development Authority (BARDA), and the Defense Advanced Research Projects Agency (DARPA), also developed and executed successful initiatives.12-14 The joint awareness and management of these efforts, however, could be strengthened through more formal agreements and processes in peacetime. The VA/FDA/NIH Memorandum of Understanding is a prototype example of each agency lending its subject matter expertise to address a host of pandemic challenges collectively, cooperatively, and efficiently.8
Public-private partnerships (eg, VA/FDA/NIH and America Makes) led to coordinated responses for crisis readiness. The Advanced Manufacturing Crisis Product Response Program, a multipartner collaboration that included VA, addressed 7 crisis scenarios, 3 of which were specifically related to COVID-19.15 In addition, both BARDA and DARPA had successful public-private collaborations, and the DoD supported national logistics and other efforts.12-14 Clearly, industry and government both recognize complementary synergies: (1) the depth of resources of US industry; and (2) the national resources, coordination, and clinical insight available through federal agencies that can address the challenges of future crises quickly and efficiently.
When traditional supply chains and manufacturing processes failed during the pandemic, new techniques were exploited to fill the unmet material needs. Novel techniques and product pathways, however, are untested or undeveloped. The collaboration between the ORD and OHIL in support of NP swab testing and production is an example of bringing research insight, regulated product development, and manufacturing together to support a complete product life cycle.
Joint Awareness and Management
The VA continues to refine the joint awareness and management (JAM) process of products from ideation to translation, to shorten the time from research to product delivery. JAM is a VA collaborative committee of partners from ORD research offices and technology transfer program, and the OHIL Office of Advanced Manufacturing, which seeks additional support and guidance from VHA clinical service lines, VA Office of General Council, and VA Office of Acquisitions, Logistics, and Construction.
This team enables the rapid identification of unmet veteran health care product needs. In addition, JAM leverages the resources of each group to support products from problem identification to solution ideation, regulated development, production, and delivery into clinical service lines. While the concept of JAM arose to meet the crisis needs of the pandemic, it persists in delivering advanced health care solutions to veterans.
A Proposed Plan
The next national crisis is likely to involve and threaten national health care security. We propose that federal agencies be brought together to form a federally supported digital stockpile. This digital stockpile must encompass, at minimum, the following features: (1) preservation of novel, scalable medical supplies and products generated during the COVID-19 pandemic, to avoid the loss of this work; (2) clinical maturation of those existing supplies and products to refine their features and functions under the guidance of clinical, regulatory, and manufacturing experts—and validate those outputs with clinical evidence; (3) manufacturing maturation of those existing supplies and products, such that complete design and production processes are developed with the intent to distribute to multiple public manufacturers during the next crisis; (4) a call for new designs/intake portal for new designs to be matured and curated as vulnerabilities are identified; (5) supply chain crisis drills executed to test public-private preparedness to ensure design transfer is turnkey and can be engaged quickly during the next crisis; and (6) public-private engagement to develop strategy, scenarios, and policy to ensure that when supply chains next fail, additional surge capacity can be quickly added to protect American lives and health care, and that when supply chains resume, surge capacity can be redirected or stood down to protect the competitive markets.
This digital stockpile can complement and be part of the Strategic National Stockpile. Whereas the Strategic National Stockpile is a reserve of physical products that may offset product shortages, the digital stockpile is a reserve of turnkey, transferable designs that may offset supply chain disruptions and production-capacity shortages.
CONCLUSIONS
The success of 3D-printed NP swabs is a specific example of the importance of collaborations across industry, government, innovators, and researchers. More important than a sole product, however, these collaborations demonstrated the potential for game-changing approaches to how public-private partnerships support the continuity of health care operations nationally and prevent the potential for unnecessary loss of life due to capacity and supply chain disruptions.
As the largest health care system in the US, the VA has a unique capability to lead in the assessment of other novel 3D-printed medical devices in partnership with the FDA. The VA has a unique patient-centered perspective on medical device efficacy, and as a government institution, it is a trusted independent source for medical device evaluation. The VA’s role in the evaluation of 3D-printed medical devices will benefit veterans and their families, clinicians, hospitals, and the broader public by providing a gold-standard evaluation for the growing medical 3D-printing industry to follow. By creating new pathways and expectations for how federal agencies maintain crisis preparedness—such as establishing a digital stockpile—we can be equipped to serve the US health care system and minimize the effects of supply chain disruptions.
1. Sacks CA, Kesselheim AS, Fralick M. The shortage of normal saline in the wake of Hurricane Maria. JAMA Intern Med. 2018;178(7):885–886. doi:10.1001/jamainternmed.2018.1936
2. Bauchner H, Fontanarosa PB, Livingston EH. Conserving supply of personal protective equipment–a call for ideas. JAMA. 2020;323(19):1911. doi:10.1001/jama.2020.4770
3. Sinha MS, Bourgeois FT, Sorger PK. Personal protective equipment for COVID-19: distributed fabrication and additive manufacturing. Am J Public Health. 2020;110(8):1162-1164. doi:10.2105/AJPH.2020.305753
4. McCarthy MC, Di Prima M, Cruz P, et al. Trust in the time of Covid-19: 3D printing and additive manufacturing (3DP/AM) as a solution to supply chain gaps. NEJM Catalyst. 2021;2(6). doi:10.1056/CAT.21.0321
5. Ford J, Goldstein T, Trahan S, Neuwirth A, Tatoris K, Decker S. A 3D-printed nasopharyngeal swab for COVID-19 diagnostic testing. 3D Print Med. 2020;6(1):21. Published 2020 Aug 15. doi:10.1186/s41205-020-00076-3
6. Callahan CJ, Lee R, Zulauf K, et al. Open development and clinical validation of multiple 3D-printed sample-collection swabs: rapid resolution of a critical COVID-19 testing bottleneck. Preprint. medRxiv. 2020;2020.04.14.20065094. Published 2020 Apr 17. doi:10.1101/2020.04.14.20065094
7. Decker SJ, Goldstein TA, Ford JM, et al. 3-dimensional printed alternative to the standard synthetic flocked nasopharyngeal swabs used for coronavirus disease 2019 testing. Clin Infect Dis. 2021;73(9):e3027-e3032. doi:10.1093/cid/ciaa1366
8. US Food and Drug Administration. Memorandum of understanding: rapid response to Covid-19 using 3d printing between National Institutes of Health within U.S. Department of Health and Human Services and Food and Drug Administration, U.S. Department of Health and Human Services and Veterans Health Administration within the U.S. Department of Veterans Affairs. March 26, 2020. Accessed August 31, 2023. https://www.fda.gov/about-fda/domestic-mous/mou-225-20-008
9. National Institutes of Health, NIH 3D Print Exchange. Covid 3D trust: trusted repository for users and suppliers through testing. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=search
10. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - assessment criteria. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabassessment
11. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - general information. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabinfo
12. US Department of Defense. Coronavirus: DOD response. December 20, 2022. Accessed August 31, 2023. https://www.defense.gov/Spotlights/Coronavirus-DoD-Response
13. US Department of Health and Human Services, Biomedical Advanced Research and Development Authority. BARDA COVID-19 response. Updated May 25, 2023. Accessed August 31, 2023. https://www.medicalcountermeasures.gov/barda/barda-covid-19-response
14. Green S. Pandemic prevention platform (P3). Accessed August 31, 2023. https://www.darpa.mil/program/pandemic-prevention-platform
15. America Makes. America makes completes successful scenario testing for crisis response program [press release]. May 25, 2021. Accessed August 31, 2023. https://www.americamakes.us/america-makes-completes-successful-scenario-testing-for-crisis-response-program
Traditional manufacturing concentrates capacity into a few discrete locations while applying lean and just-in-time philosophies to maximize profit during times of somewhat predictable supply and demand. This approach exposed nationwide vulnerabilities even during local crises, such as the United States saline shortages following closure of a single plant in Puerto Rico following Hurricane Maria in 2017.1 Interruptions to the supply chain due to pandemic plant closure, weather, politics, or surge demand can cause immediate and lasting shortages. Nasal swabs were a clear example.
At the onset of COVID-19, 2 companies—Puritan in Guilford, Maine, and Copan in Italy—manufactured nearly all of the highly specialized nasopharyngeal (NP) swabs singled out by the Centers for Disease Control and Prevention (CDC) and the US Food and Drug Administration (FDA) to test patients for COVID-19. Demand for swabs skyrocketed as the virus spread, and they became unattainable. The lack of swabs meant patients went undiagnosed. Without knowing who was positive, people with symptoms and known contacts were presumed positive and quarantined, impacting isolated patients, the health care professionals treating them, and the entire US economy.
3-Dimensional Printing Solutions
Manufacturing NP swabs is not trivial. Their simple shape conceals complexity and requires highly specialized equipment. The lead time for one non-US machine manufacturer was > 6 months at the start of the pandemic.
Digital manufacturing/3-dimensional (3D) printing represented a potential solution to the supply chain crisis.2 Designers created digital blueprints for 3D-printed goods, face masks, face shields, and ventilator splitters were rapidly created and shared.3,4 Scrambling to fill the critical need for NP swabs, many hospitals, businesses, and academic centers began 3D printing swabs. This effort was spearheaded by University of South Florida (USF) and Northwell Health researchers and clinicians, who designed and tested a 3D-printed NP swab from photocurable resin that was printable on 2 models of Formlabs printers.5 Several other 3D-printed NP swab designs soon followed. This innovation and problem-solving renaissance faced several challenges well known to traditional manufacturers of regulated products but novel to newcomers.
The first challlenge was that these NP swabs predate FDA oversight of medical device development and manufacturing and no testing standards existed. Designers began casting prototypes out without guidance about the critical features and clinical functions required. Many of these designs did not have a clinical evaluation pathway to test safety and efficacy.
The second challlenge was that these swabs were being produced by facilities not registered with the FDA. This raised concerns about the quality of unlisted medical products developed and manufactured at novel facilities.
The third challenge was that small-scale novel approaches may offset local shortages but could not address national needs. The self-organized infrastructure for this crisis was ad hoc, local, and lacked coordinated federal support. This led to rolling shortages of these materials for years.
Two studies were performed early in the pandemic. The first study evaluated 4 prototypes of different manufacturer designs, finding excellent concordance among them and their control swab.6 A second study demonstrated the USF swab to be noninferior to the standard of care.7 Both studies acknowledged and addressed the first challenge for their designs.
COLLABORATIONS
Interagency
Before the pandemic, the US Department of Veterans Affairs (VA) had been coordinating with the FDA, the National Institutes of Health (NIH), and the nonprofit America Makes to bring medical product development and manufacturing closer to the point of care.
At the outset of the COVID-19 pandemic, the collaboration was formalized to address new challenges.8 The objectives of this collaboration were the following: (1) host a digital repository for 3D-printed digital designs for personal protectice equipment and other medical supplies in or at risk of shortage; (2) provide scientifically based ratings for designs according to clinical and field testing; and (3) offer education to health care workers and the public about the digital manufacturing of medical goods and devices.4,9
A key output of this collaboration was the COVID 3D Trusted Repository For Users And Suppliers Through Testing (COVID 3D TRUST), a curated archive of designs. In most cases, existing FDA standards and guidance formed the basis of testing strategies with deviations due to limited access to traditional testing facilities and reagents.
To address novel NP swabs, working with its COVID 3D TRUST partners, the VA gathered a combined list of clinical- and engineering-informed customer requirements and performed a hazard analysis. The result was a list of design inputs for NP swabs and 8 standard test protocols to evaluate key functions (Table).10 These protocols are meant to benchmark novel 3D-printed swabs against the key functions of established, traditionally manufactured swabs, which have a long record of safety and efficacy. The protocols, developed by the VA and undergoing validation by the US Army, empower and inform consumers and provide performance metrics to swab designers and manufacturers. The testing protocols and preliminary test results developed by the VA are publicly available at the NIH.11
Intra-agency
The use of the inputs and verification tests noted in the Table may reduce the risk of poor design but were inadequate to evaluate the clinical safety and efficacy of novel swabs. Recognizing this, the VA Office of Healthcare Innovation and Learning (OHIL) and the Office of Research and Development (ORD) launched the Nasal Swab Objective and Statistical Evaluation (NOSE) study to formally evaluate the safety and efficacy of 3D-printed swabs in the field. This multisite clinical study was a close collaboration between the OHIL and ORD. The OHIL provided the quality system and manufacturing oversight and delivery of the swabs, and the ORD provided scientific review, research infrastructure, human subjects oversight, administrative support, and funding and fiscal oversight. The OHIL/ORD collaboration resulted in the successful completion of the NOSE study.
This study (manuscript under preparation) yielded two 3D-printing production processes and swab designs that had comparable performance to the standard of care, were manufacturable compliant with FDA guidelines, and could be produced at scale in a distributed manner. This approach directly addressed the 3 challenges described earlier.
LESSONS LEARNED
Swabs were an example of supply challenges in the pandemic, but advanced manufacturing (notably, digital designs leading to 3D-printed solutions) also served as a temporary solution to device and product shortages during the COVID-19 pandemic. Digital designs and 3D printing as manufacturing techniques have the following key advantages: (1) they are distributed in nature, both in the breadth of locations that have access to these manufacturing platforms and in the depth of material choice that can be used to fabricate products, which alleviates the threat of a disaster impacting manufacturing capacity or a material stream; (2) they do not require retooling of machinery so new products can deploy rapidly and on demand; and (3) the speed of digital iteration, printing, and revision allows for rapid product development and production.
There also are notable disadvantages to these techniques. First, because 3D printing is a newer technology, there is less general depth of knowledge regarding design and material choice for additive manufacturing. Second, the flexibility of 3D printing means that operators must increase awareness of the factors that might cause the fabrication of a part to fail in either printing or postprocessing. Third, there are significant gaps in understanding how materials and manufacturing processes will perform in high-stakes settings such as health care, where performance and biocompatibility may be critical to support life-sustaining functions. Fourth, digital files are vulnerable to intentional or unintentional alteration. These alterations might weaken design integrity and be imperceptible to the manufacturer or end user. This is a prevalent challenge in all open-source designs.
The pandemic materialized quickly and created vast supply chain challenges. To address this crisis, it was clear that the average 17-year interval between research and translation in the US was unacceptable. The VA was able to accelerate swiftly many existing processes to meet this need, build new capabilities, and establish new practices for the rapid evaluation and deployment of health care products and guidance. This agile and innovative cooperation was critical in the success of the VA’s national support for pandemic solutions.
Finally, although COVID 3D TRUST was able to provide testing of submitted designs, this collaboration was not a substitute for the “peacetime” process of manufacturing site registration with the FDA and product listing. COVID 3D TRUST could evaluate designs only, not the production process, safety, and efficacy.
CALLS TO ACTION
The pandemic's impact on medical supply chain security persists, as does the need for greater foresight and crisis preparation. We must act now to avoid experiencing again the magnitude of fatalities (civilian and veteran) and the devastation to the US economy and livelihoods that occurred during this single biological event. To this end, creating a digital stockpile of federally curated, crisis-ready designs for as-needed distribution across our US industrial base would offer a second line of defense against life-threatening supply chain interruptions. The realization of such a digital stockpile requires calls to action among multiple contributors.
Collaborations
The VA’s Fourth Mission is to improve the nation’s preparedness for response to war, terrorism, national emergencies, and natural disasters. The VA does this by developing plans and taking actions to ensure continued service to veterans, as well as to support national, state, and local emergency management, public health, safety, and homeland security efforts.
The VA partnership with the FDA and NIH during the pandemic enabled successful coordination among federal agencies. Numerous other agencies, including the US Department of Defense (DoD), the Biomedical Advanced Research and Development Authority (BARDA), and the Defense Advanced Research Projects Agency (DARPA), also developed and executed successful initiatives.12-14 The joint awareness and management of these efforts, however, could be strengthened through more formal agreements and processes in peacetime. The VA/FDA/NIH Memorandum of Understanding is a prototype example of each agency lending its subject matter expertise to address a host of pandemic challenges collectively, cooperatively, and efficiently.8
Public-private partnerships (eg, VA/FDA/NIH and America Makes) led to coordinated responses for crisis readiness. The Advanced Manufacturing Crisis Product Response Program, a multipartner collaboration that included VA, addressed 7 crisis scenarios, 3 of which were specifically related to COVID-19.15 In addition, both BARDA and DARPA had successful public-private collaborations, and the DoD supported national logistics and other efforts.12-14 Clearly, industry and government both recognize complementary synergies: (1) the depth of resources of US industry; and (2) the national resources, coordination, and clinical insight available through federal agencies that can address the challenges of future crises quickly and efficiently.
When traditional supply chains and manufacturing processes failed during the pandemic, new techniques were exploited to fill the unmet material needs. Novel techniques and product pathways, however, are untested or undeveloped. The collaboration between the ORD and OHIL in support of NP swab testing and production is an example of bringing research insight, regulated product development, and manufacturing together to support a complete product life cycle.
Joint Awareness and Management
The VA continues to refine the joint awareness and management (JAM) process of products from ideation to translation, to shorten the time from research to product delivery. JAM is a VA collaborative committee of partners from ORD research offices and technology transfer program, and the OHIL Office of Advanced Manufacturing, which seeks additional support and guidance from VHA clinical service lines, VA Office of General Council, and VA Office of Acquisitions, Logistics, and Construction.
This team enables the rapid identification of unmet veteran health care product needs. In addition, JAM leverages the resources of each group to support products from problem identification to solution ideation, regulated development, production, and delivery into clinical service lines. While the concept of JAM arose to meet the crisis needs of the pandemic, it persists in delivering advanced health care solutions to veterans.
A Proposed Plan
The next national crisis is likely to involve and threaten national health care security. We propose that federal agencies be brought together to form a federally supported digital stockpile. This digital stockpile must encompass, at minimum, the following features: (1) preservation of novel, scalable medical supplies and products generated during the COVID-19 pandemic, to avoid the loss of this work; (2) clinical maturation of those existing supplies and products to refine their features and functions under the guidance of clinical, regulatory, and manufacturing experts—and validate those outputs with clinical evidence; (3) manufacturing maturation of those existing supplies and products, such that complete design and production processes are developed with the intent to distribute to multiple public manufacturers during the next crisis; (4) a call for new designs/intake portal for new designs to be matured and curated as vulnerabilities are identified; (5) supply chain crisis drills executed to test public-private preparedness to ensure design transfer is turnkey and can be engaged quickly during the next crisis; and (6) public-private engagement to develop strategy, scenarios, and policy to ensure that when supply chains next fail, additional surge capacity can be quickly added to protect American lives and health care, and that when supply chains resume, surge capacity can be redirected or stood down to protect the competitive markets.
This digital stockpile can complement and be part of the Strategic National Stockpile. Whereas the Strategic National Stockpile is a reserve of physical products that may offset product shortages, the digital stockpile is a reserve of turnkey, transferable designs that may offset supply chain disruptions and production-capacity shortages.
CONCLUSIONS
The success of 3D-printed NP swabs is a specific example of the importance of collaborations across industry, government, innovators, and researchers. More important than a sole product, however, these collaborations demonstrated the potential for game-changing approaches to how public-private partnerships support the continuity of health care operations nationally and prevent the potential for unnecessary loss of life due to capacity and supply chain disruptions.
As the largest health care system in the US, the VA has a unique capability to lead in the assessment of other novel 3D-printed medical devices in partnership with the FDA. The VA has a unique patient-centered perspective on medical device efficacy, and as a government institution, it is a trusted independent source for medical device evaluation. The VA’s role in the evaluation of 3D-printed medical devices will benefit veterans and their families, clinicians, hospitals, and the broader public by providing a gold-standard evaluation for the growing medical 3D-printing industry to follow. By creating new pathways and expectations for how federal agencies maintain crisis preparedness—such as establishing a digital stockpile—we can be equipped to serve the US health care system and minimize the effects of supply chain disruptions.
Traditional manufacturing concentrates capacity into a few discrete locations while applying lean and just-in-time philosophies to maximize profit during times of somewhat predictable supply and demand. This approach exposed nationwide vulnerabilities even during local crises, such as the United States saline shortages following closure of a single plant in Puerto Rico following Hurricane Maria in 2017.1 Interruptions to the supply chain due to pandemic plant closure, weather, politics, or surge demand can cause immediate and lasting shortages. Nasal swabs were a clear example.
At the onset of COVID-19, 2 companies—Puritan in Guilford, Maine, and Copan in Italy—manufactured nearly all of the highly specialized nasopharyngeal (NP) swabs singled out by the Centers for Disease Control and Prevention (CDC) and the US Food and Drug Administration (FDA) to test patients for COVID-19. Demand for swabs skyrocketed as the virus spread, and they became unattainable. The lack of swabs meant patients went undiagnosed. Without knowing who was positive, people with symptoms and known contacts were presumed positive and quarantined, impacting isolated patients, the health care professionals treating them, and the entire US economy.
3-Dimensional Printing Solutions
Manufacturing NP swabs is not trivial. Their simple shape conceals complexity and requires highly specialized equipment. The lead time for one non-US machine manufacturer was > 6 months at the start of the pandemic.
Digital manufacturing/3-dimensional (3D) printing represented a potential solution to the supply chain crisis.2 Designers created digital blueprints for 3D-printed goods, face masks, face shields, and ventilator splitters were rapidly created and shared.3,4 Scrambling to fill the critical need for NP swabs, many hospitals, businesses, and academic centers began 3D printing swabs. This effort was spearheaded by University of South Florida (USF) and Northwell Health researchers and clinicians, who designed and tested a 3D-printed NP swab from photocurable resin that was printable on 2 models of Formlabs printers.5 Several other 3D-printed NP swab designs soon followed. This innovation and problem-solving renaissance faced several challenges well known to traditional manufacturers of regulated products but novel to newcomers.
The first challlenge was that these NP swabs predate FDA oversight of medical device development and manufacturing and no testing standards existed. Designers began casting prototypes out without guidance about the critical features and clinical functions required. Many of these designs did not have a clinical evaluation pathway to test safety and efficacy.
The second challlenge was that these swabs were being produced by facilities not registered with the FDA. This raised concerns about the quality of unlisted medical products developed and manufactured at novel facilities.
The third challenge was that small-scale novel approaches may offset local shortages but could not address national needs. The self-organized infrastructure for this crisis was ad hoc, local, and lacked coordinated federal support. This led to rolling shortages of these materials for years.
Two studies were performed early in the pandemic. The first study evaluated 4 prototypes of different manufacturer designs, finding excellent concordance among them and their control swab.6 A second study demonstrated the USF swab to be noninferior to the standard of care.7 Both studies acknowledged and addressed the first challenge for their designs.
COLLABORATIONS
Interagency
Before the pandemic, the US Department of Veterans Affairs (VA) had been coordinating with the FDA, the National Institutes of Health (NIH), and the nonprofit America Makes to bring medical product development and manufacturing closer to the point of care.
At the outset of the COVID-19 pandemic, the collaboration was formalized to address new challenges.8 The objectives of this collaboration were the following: (1) host a digital repository for 3D-printed digital designs for personal protectice equipment and other medical supplies in or at risk of shortage; (2) provide scientifically based ratings for designs according to clinical and field testing; and (3) offer education to health care workers and the public about the digital manufacturing of medical goods and devices.4,9
A key output of this collaboration was the COVID 3D Trusted Repository For Users And Suppliers Through Testing (COVID 3D TRUST), a curated archive of designs. In most cases, existing FDA standards and guidance formed the basis of testing strategies with deviations due to limited access to traditional testing facilities and reagents.
To address novel NP swabs, working with its COVID 3D TRUST partners, the VA gathered a combined list of clinical- and engineering-informed customer requirements and performed a hazard analysis. The result was a list of design inputs for NP swabs and 8 standard test protocols to evaluate key functions (Table).10 These protocols are meant to benchmark novel 3D-printed swabs against the key functions of established, traditionally manufactured swabs, which have a long record of safety and efficacy. The protocols, developed by the VA and undergoing validation by the US Army, empower and inform consumers and provide performance metrics to swab designers and manufacturers. The testing protocols and preliminary test results developed by the VA are publicly available at the NIH.11
Intra-agency
The use of the inputs and verification tests noted in the Table may reduce the risk of poor design but were inadequate to evaluate the clinical safety and efficacy of novel swabs. Recognizing this, the VA Office of Healthcare Innovation and Learning (OHIL) and the Office of Research and Development (ORD) launched the Nasal Swab Objective and Statistical Evaluation (NOSE) study to formally evaluate the safety and efficacy of 3D-printed swabs in the field. This multisite clinical study was a close collaboration between the OHIL and ORD. The OHIL provided the quality system and manufacturing oversight and delivery of the swabs, and the ORD provided scientific review, research infrastructure, human subjects oversight, administrative support, and funding and fiscal oversight. The OHIL/ORD collaboration resulted in the successful completion of the NOSE study.
This study (manuscript under preparation) yielded two 3D-printing production processes and swab designs that had comparable performance to the standard of care, were manufacturable compliant with FDA guidelines, and could be produced at scale in a distributed manner. This approach directly addressed the 3 challenges described earlier.
LESSONS LEARNED
Swabs were an example of supply challenges in the pandemic, but advanced manufacturing (notably, digital designs leading to 3D-printed solutions) also served as a temporary solution to device and product shortages during the COVID-19 pandemic. Digital designs and 3D printing as manufacturing techniques have the following key advantages: (1) they are distributed in nature, both in the breadth of locations that have access to these manufacturing platforms and in the depth of material choice that can be used to fabricate products, which alleviates the threat of a disaster impacting manufacturing capacity or a material stream; (2) they do not require retooling of machinery so new products can deploy rapidly and on demand; and (3) the speed of digital iteration, printing, and revision allows for rapid product development and production.
There also are notable disadvantages to these techniques. First, because 3D printing is a newer technology, there is less general depth of knowledge regarding design and material choice for additive manufacturing. Second, the flexibility of 3D printing means that operators must increase awareness of the factors that might cause the fabrication of a part to fail in either printing or postprocessing. Third, there are significant gaps in understanding how materials and manufacturing processes will perform in high-stakes settings such as health care, where performance and biocompatibility may be critical to support life-sustaining functions. Fourth, digital files are vulnerable to intentional or unintentional alteration. These alterations might weaken design integrity and be imperceptible to the manufacturer or end user. This is a prevalent challenge in all open-source designs.
The pandemic materialized quickly and created vast supply chain challenges. To address this crisis, it was clear that the average 17-year interval between research and translation in the US was unacceptable. The VA was able to accelerate swiftly many existing processes to meet this need, build new capabilities, and establish new practices for the rapid evaluation and deployment of health care products and guidance. This agile and innovative cooperation was critical in the success of the VA’s national support for pandemic solutions.
Finally, although COVID 3D TRUST was able to provide testing of submitted designs, this collaboration was not a substitute for the “peacetime” process of manufacturing site registration with the FDA and product listing. COVID 3D TRUST could evaluate designs only, not the production process, safety, and efficacy.
CALLS TO ACTION
The pandemic's impact on medical supply chain security persists, as does the need for greater foresight and crisis preparation. We must act now to avoid experiencing again the magnitude of fatalities (civilian and veteran) and the devastation to the US economy and livelihoods that occurred during this single biological event. To this end, creating a digital stockpile of federally curated, crisis-ready designs for as-needed distribution across our US industrial base would offer a second line of defense against life-threatening supply chain interruptions. The realization of such a digital stockpile requires calls to action among multiple contributors.
Collaborations
The VA’s Fourth Mission is to improve the nation’s preparedness for response to war, terrorism, national emergencies, and natural disasters. The VA does this by developing plans and taking actions to ensure continued service to veterans, as well as to support national, state, and local emergency management, public health, safety, and homeland security efforts.
The VA partnership with the FDA and NIH during the pandemic enabled successful coordination among federal agencies. Numerous other agencies, including the US Department of Defense (DoD), the Biomedical Advanced Research and Development Authority (BARDA), and the Defense Advanced Research Projects Agency (DARPA), also developed and executed successful initiatives.12-14 The joint awareness and management of these efforts, however, could be strengthened through more formal agreements and processes in peacetime. The VA/FDA/NIH Memorandum of Understanding is a prototype example of each agency lending its subject matter expertise to address a host of pandemic challenges collectively, cooperatively, and efficiently.8
Public-private partnerships (eg, VA/FDA/NIH and America Makes) led to coordinated responses for crisis readiness. The Advanced Manufacturing Crisis Product Response Program, a multipartner collaboration that included VA, addressed 7 crisis scenarios, 3 of which were specifically related to COVID-19.15 In addition, both BARDA and DARPA had successful public-private collaborations, and the DoD supported national logistics and other efforts.12-14 Clearly, industry and government both recognize complementary synergies: (1) the depth of resources of US industry; and (2) the national resources, coordination, and clinical insight available through federal agencies that can address the challenges of future crises quickly and efficiently.
When traditional supply chains and manufacturing processes failed during the pandemic, new techniques were exploited to fill the unmet material needs. Novel techniques and product pathways, however, are untested or undeveloped. The collaboration between the ORD and OHIL in support of NP swab testing and production is an example of bringing research insight, regulated product development, and manufacturing together to support a complete product life cycle.
Joint Awareness and Management
The VA continues to refine the joint awareness and management (JAM) process of products from ideation to translation, to shorten the time from research to product delivery. JAM is a VA collaborative committee of partners from ORD research offices and technology transfer program, and the OHIL Office of Advanced Manufacturing, which seeks additional support and guidance from VHA clinical service lines, VA Office of General Council, and VA Office of Acquisitions, Logistics, and Construction.
This team enables the rapid identification of unmet veteran health care product needs. In addition, JAM leverages the resources of each group to support products from problem identification to solution ideation, regulated development, production, and delivery into clinical service lines. While the concept of JAM arose to meet the crisis needs of the pandemic, it persists in delivering advanced health care solutions to veterans.
A Proposed Plan
The next national crisis is likely to involve and threaten national health care security. We propose that federal agencies be brought together to form a federally supported digital stockpile. This digital stockpile must encompass, at minimum, the following features: (1) preservation of novel, scalable medical supplies and products generated during the COVID-19 pandemic, to avoid the loss of this work; (2) clinical maturation of those existing supplies and products to refine their features and functions under the guidance of clinical, regulatory, and manufacturing experts—and validate those outputs with clinical evidence; (3) manufacturing maturation of those existing supplies and products, such that complete design and production processes are developed with the intent to distribute to multiple public manufacturers during the next crisis; (4) a call for new designs/intake portal for new designs to be matured and curated as vulnerabilities are identified; (5) supply chain crisis drills executed to test public-private preparedness to ensure design transfer is turnkey and can be engaged quickly during the next crisis; and (6) public-private engagement to develop strategy, scenarios, and policy to ensure that when supply chains next fail, additional surge capacity can be quickly added to protect American lives and health care, and that when supply chains resume, surge capacity can be redirected or stood down to protect the competitive markets.
This digital stockpile can complement and be part of the Strategic National Stockpile. Whereas the Strategic National Stockpile is a reserve of physical products that may offset product shortages, the digital stockpile is a reserve of turnkey, transferable designs that may offset supply chain disruptions and production-capacity shortages.
CONCLUSIONS
The success of 3D-printed NP swabs is a specific example of the importance of collaborations across industry, government, innovators, and researchers. More important than a sole product, however, these collaborations demonstrated the potential for game-changing approaches to how public-private partnerships support the continuity of health care operations nationally and prevent the potential for unnecessary loss of life due to capacity and supply chain disruptions.
As the largest health care system in the US, the VA has a unique capability to lead in the assessment of other novel 3D-printed medical devices in partnership with the FDA. The VA has a unique patient-centered perspective on medical device efficacy, and as a government institution, it is a trusted independent source for medical device evaluation. The VA’s role in the evaluation of 3D-printed medical devices will benefit veterans and their families, clinicians, hospitals, and the broader public by providing a gold-standard evaluation for the growing medical 3D-printing industry to follow. By creating new pathways and expectations for how federal agencies maintain crisis preparedness—such as establishing a digital stockpile—we can be equipped to serve the US health care system and minimize the effects of supply chain disruptions.
1. Sacks CA, Kesselheim AS, Fralick M. The shortage of normal saline in the wake of Hurricane Maria. JAMA Intern Med. 2018;178(7):885–886. doi:10.1001/jamainternmed.2018.1936
2. Bauchner H, Fontanarosa PB, Livingston EH. Conserving supply of personal protective equipment–a call for ideas. JAMA. 2020;323(19):1911. doi:10.1001/jama.2020.4770
3. Sinha MS, Bourgeois FT, Sorger PK. Personal protective equipment for COVID-19: distributed fabrication and additive manufacturing. Am J Public Health. 2020;110(8):1162-1164. doi:10.2105/AJPH.2020.305753
4. McCarthy MC, Di Prima M, Cruz P, et al. Trust in the time of Covid-19: 3D printing and additive manufacturing (3DP/AM) as a solution to supply chain gaps. NEJM Catalyst. 2021;2(6). doi:10.1056/CAT.21.0321
5. Ford J, Goldstein T, Trahan S, Neuwirth A, Tatoris K, Decker S. A 3D-printed nasopharyngeal swab for COVID-19 diagnostic testing. 3D Print Med. 2020;6(1):21. Published 2020 Aug 15. doi:10.1186/s41205-020-00076-3
6. Callahan CJ, Lee R, Zulauf K, et al. Open development and clinical validation of multiple 3D-printed sample-collection swabs: rapid resolution of a critical COVID-19 testing bottleneck. Preprint. medRxiv. 2020;2020.04.14.20065094. Published 2020 Apr 17. doi:10.1101/2020.04.14.20065094
7. Decker SJ, Goldstein TA, Ford JM, et al. 3-dimensional printed alternative to the standard synthetic flocked nasopharyngeal swabs used for coronavirus disease 2019 testing. Clin Infect Dis. 2021;73(9):e3027-e3032. doi:10.1093/cid/ciaa1366
8. US Food and Drug Administration. Memorandum of understanding: rapid response to Covid-19 using 3d printing between National Institutes of Health within U.S. Department of Health and Human Services and Food and Drug Administration, U.S. Department of Health and Human Services and Veterans Health Administration within the U.S. Department of Veterans Affairs. March 26, 2020. Accessed August 31, 2023. https://www.fda.gov/about-fda/domestic-mous/mou-225-20-008
9. National Institutes of Health, NIH 3D Print Exchange. Covid 3D trust: trusted repository for users and suppliers through testing. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=search
10. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - assessment criteria. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabassessment
11. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - general information. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabinfo
12. US Department of Defense. Coronavirus: DOD response. December 20, 2022. Accessed August 31, 2023. https://www.defense.gov/Spotlights/Coronavirus-DoD-Response
13. US Department of Health and Human Services, Biomedical Advanced Research and Development Authority. BARDA COVID-19 response. Updated May 25, 2023. Accessed August 31, 2023. https://www.medicalcountermeasures.gov/barda/barda-covid-19-response
14. Green S. Pandemic prevention platform (P3). Accessed August 31, 2023. https://www.darpa.mil/program/pandemic-prevention-platform
15. America Makes. America makes completes successful scenario testing for crisis response program [press release]. May 25, 2021. Accessed August 31, 2023. https://www.americamakes.us/america-makes-completes-successful-scenario-testing-for-crisis-response-program
1. Sacks CA, Kesselheim AS, Fralick M. The shortage of normal saline in the wake of Hurricane Maria. JAMA Intern Med. 2018;178(7):885–886. doi:10.1001/jamainternmed.2018.1936
2. Bauchner H, Fontanarosa PB, Livingston EH. Conserving supply of personal protective equipment–a call for ideas. JAMA. 2020;323(19):1911. doi:10.1001/jama.2020.4770
3. Sinha MS, Bourgeois FT, Sorger PK. Personal protective equipment for COVID-19: distributed fabrication and additive manufacturing. Am J Public Health. 2020;110(8):1162-1164. doi:10.2105/AJPH.2020.305753
4. McCarthy MC, Di Prima M, Cruz P, et al. Trust in the time of Covid-19: 3D printing and additive manufacturing (3DP/AM) as a solution to supply chain gaps. NEJM Catalyst. 2021;2(6). doi:10.1056/CAT.21.0321
5. Ford J, Goldstein T, Trahan S, Neuwirth A, Tatoris K, Decker S. A 3D-printed nasopharyngeal swab for COVID-19 diagnostic testing. 3D Print Med. 2020;6(1):21. Published 2020 Aug 15. doi:10.1186/s41205-020-00076-3
6. Callahan CJ, Lee R, Zulauf K, et al. Open development and clinical validation of multiple 3D-printed sample-collection swabs: rapid resolution of a critical COVID-19 testing bottleneck. Preprint. medRxiv. 2020;2020.04.14.20065094. Published 2020 Apr 17. doi:10.1101/2020.04.14.20065094
7. Decker SJ, Goldstein TA, Ford JM, et al. 3-dimensional printed alternative to the standard synthetic flocked nasopharyngeal swabs used for coronavirus disease 2019 testing. Clin Infect Dis. 2021;73(9):e3027-e3032. doi:10.1093/cid/ciaa1366
8. US Food and Drug Administration. Memorandum of understanding: rapid response to Covid-19 using 3d printing between National Institutes of Health within U.S. Department of Health and Human Services and Food and Drug Administration, U.S. Department of Health and Human Services and Veterans Health Administration within the U.S. Department of Veterans Affairs. March 26, 2020. Accessed August 31, 2023. https://www.fda.gov/about-fda/domestic-mous/mou-225-20-008
9. National Institutes of Health, NIH 3D Print Exchange. Covid 3D trust: trusted repository for users and suppliers through testing. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=search
10. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - assessment criteria. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabassessment
11. National Institutes of Health, NIH 3D Print Exchange. 3D printed nasal swabs - general information. August 17, 2020. Accessed August 31, 2023. https://3d.nih.gov/collections/covid-19-response?tab=swabinfo
12. US Department of Defense. Coronavirus: DOD response. December 20, 2022. Accessed August 31, 2023. https://www.defense.gov/Spotlights/Coronavirus-DoD-Response
13. US Department of Health and Human Services, Biomedical Advanced Research and Development Authority. BARDA COVID-19 response. Updated May 25, 2023. Accessed August 31, 2023. https://www.medicalcountermeasures.gov/barda/barda-covid-19-response
14. Green S. Pandemic prevention platform (P3). Accessed August 31, 2023. https://www.darpa.mil/program/pandemic-prevention-platform
15. America Makes. America makes completes successful scenario testing for crisis response program [press release]. May 25, 2021. Accessed August 31, 2023. https://www.americamakes.us/america-makes-completes-successful-scenario-testing-for-crisis-response-program
VA SHIELD: A Biorepository for Veterans and the Nation
The Veterans Health Administration (VHA) clinicians, clinician-investigators, and investigators perform basic and translational research for the benefit of our nation and are widely recognized for treating patients and discovering cures.1,2 In May 2020, the US Department of Veterans Affairs (VA) launched the VA Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD). The goal of this novel enterprise was to assemble a comprehensive specimen and data repository for emerging life-threatening diseases and to address future challenges. VA SHIELD was specifically charged with creating a biorepository to advance research, improve diagnostic and therapeutic capabilities, and develop strategies for immediate deployment to VA clinical environments. One main objective of VA SHIELD is to harness the clinical and scientific strengths of the VA in order to create a more cohesive collaboration between preexisting clinical research efforts within the VA.
ANATOMY OF VA SHIELD
The charge and scope of VA SHIELD is unique.3 As an entity, this program leverages the strengths of the diverse VHA network, has a broad potential impact on national health care, is positioned to respond rapidly to national and international health-related events, and substantially contributes to clinical research and development. In addition, VA SHIELD upholds VA’s Fourth Mission, which is to contribute to national emergencies and support emergency management, public health, safety, and homeland security efforts.
VA SHIELD is part of the VA Office of Research and Development (ORD). The coordinating center (CC), headquartered in Cleveland, Ohio, is the central operational partner, leading VA SHIELD and interacting with other important VA programs, including laboratory, clinical science, rehabilitation, and health services. The VA SHIELD CC oversees all aspects of operations, including biospecimen collection, creating and enforcing of standard operating procedures, ensuring the quality of the samples, processing research applications, distribution of samples, financing, and progress reports. The CC also initiates and maintains interagency collaborations, convenes stakeholders, and develops strategic plans to address emerging diseases.
The VA SHIELD Executive Steering Committee (ESC) is composed of infectious disease, biorepository, and public health specialists. The ESC provides scientific and programmatic direction to the CC, including operational activities and guidance regarding biorepository priorities and scientific agenda, and measuring and reporting on VA SHIELD accomplishments.
The primary function of the Programmatic and Scientific Review Board (PSRB) is to evaluate incoming research proposals for specimen and data use for feasibility and make recommendations to the VA SHIELD CC. The PSRB evaluates and ensures that data and specimen use align with VA SHIELD ethical, clinical, and scientific objectives.
VA SHIELD IN PRACTICE
VA SHIELD consisted of 11 specimen collection sites (Atlanta, GA; Boise, ID; Bronx, NY; Cincinnati, OH; Cleveland, OH; Durham, NC; Houston, TX; Los Angeles, CA; Mountain Home, TN; Palo Alto, CA; and Tucson, AZ), a data processing center in Boston, MA, and 2 central biorepositories in Palo Alto, CA, and Tucson, AZ. Information flow is a coordinated process among specimen collection sites, data processing centers, and the biorepositories. Initially, each local collection site identifies residual specimens that would have been discarded after clinical laboratory testing. These samples currently account for the majority of biological material within VA SHIELD via a novel collection protocol known as “Sweep,” which allows residual clinical discarded samples as well as samples from patients with new emerging infectious and noninfectious diseases of concern to be collected at the time of first emergence and submitted to VA SHIELD during the course of routine veteran health care.3 These clinical discarded samples are de-identified and transferred from the clinical laboratory to VA SHIELD. The VA Central Institutional Review Board (cIRB) has approved the use of these samples as nonhuman subject research. Biological samples are collected, processed, aliquoted, shipped to, and stored at the central biorepository sites.
The Umbrella amendment to Sweep that has been approved also by the VA cIRB, will allow VA SHIELD sites to prospectively consent veterans and collect biospecimens and additional clinical and self-reported information. The implementation of Umbrella could significantly enhance collection and research. Although Sweep is a onetime collection of samples, the Umbrella protocol will allow the longitudinal collection of samples from the same patient. Additionally, the Umbrella amendment will allow VA SHIELD to accept samples from other preexisting biorepositories or specimen collections.
Central Biorepositories
VA SHIELD has a federated organization with 2 central specimen biorepositories (Palo Alto, CA and Tucson, AZ), and an enterprise data processing center (Boston, MA). The specimen biorepositories receive de-identified specimens that are stored until distribution to approved research projects. The samples and data are linked using an electronic honest broker system to protect privacy, which integrates de-identified specimens with requested clinical and demographic data as needed for approved projects. The honest broker system is operated by independent personnel and does not have vested interest in any studies being performed under VA SHIELD. The integration of sample and associated data is done only as needed when characterization of the donor/participant is necessary byresearch aims or project outcomes. The process is facilitated by a nationally supported laboratory information management system (LIMS), managed by the VA SHIELD data center, that assists with all data requests. The clinical and demographic data are collected from VA electronic health record (EHR), available through VA Corporate Data Warehouse (CDW) and VA Informatics and Computing Infrastructure (VINCI) as needed and integrated with the biorepository samples information for approved VA SHIELD studies. The CDW is the largest longitudinal EHR data collection in the US and has the ability to provide access to national clinical and demographic data.
VA SHIELD interacts with multiple VA programs and other entities (Figure). For example, Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) is a network of 5 VA medical centers supported by the Centers for Disease Control and Prevention.4 Its initial goal was to perform surveillance for acute gastroenteritis. In 2020, SUPERNOVA shifted to conduct surveillance for COVID-19 variants among veterans.5 VA SHIELD also interacts with VHA genomic surveillance and sequencing programs: the VA Sequencing Collaborations United for Research and Epidemiology (SeqCURE) and VA Sequencing for Research Clinical and Epidemiology (SeqFORCE), described by Krishnan and colleagues.6
Working Groups
To encourage research projects that use biospecimens, VA SHIELD developed content-oriented research working groups. The goal is to inspire collaborations between VA scientists and prevent redundant or overlapping projects. Currently working groups are focused on long COVID, and COVID-19 neurology, pathogen host response, epidemiology and sequencing, cancer and cancer biomarkers, antimicrobial resistance, and vector-borne diseases. Working groups meet regularly to discuss projects and report progress. Working groups also may consider samples that might benefit VA health research and identify potential veteran populations for future research. Working groups connect VA SHIELD and investigators and guide the collection and use of resources.
Ethical Considerations
We recognize the significant ethical concerns for biobanking of specimens. However, there is no general consensus or guideline that addresses all of the complex ethical issues regarding biobanking.7 To address these ethical concerns, we applied the VA Ethical Framework Principles for Access to and Use of Veteran Data principles to VA SHIELD, including all parties who oversee the access to, sharing of, or the use of data, or who access or use its data.8
Conclusions
The VA has assembled a scientific enterprise dedicated to combating emerging infectious diseases and other threats to our patients. This enterprise has been modeled in its structure and oversight to support VA SHIELD. The establishment of a real-time biorepository and data procurement system linked to clinical samples is a bold step forward to address current and future challenges. Similarly, the integration and cooperation of multiple arms within the VA that transcend disciplines and boundaries promise to shepherd a new era of system-wide investigation. In the future, VA SHIELD will integrate with other existing government agencies to advance mutual scientific agendas. VA SHIELD has established the data and biorepository infrastructure to develop innovative and novel technologies to address future challenges. The alignment of basic science, clinical, and translational research goals under one governance is a significant advancement compared with previous models of research coordination.
VA SHIELD was developed to meet an immediate need; it was also framed to be a research enterprise that harnesses the robust clinical and research environment in VHA. The VA SHIELD infrastructure was conceptualized to harmonize specimen and data collection across the VA, allowing researchers to leverage broader collection efforts. Building a biorepository and data collection system within the largest integrated health care system has the potential to provide a lasting impact on VHA and on our nation’s health.
Acknowledgments
The authors wish to acknowledge Ms. Daphne Swancutt for her contribution as copywriter for this manuscript. The authors wish to acknowledge the VA SHIELD investigators: Mary Cloud Ammons, David Beenhouwer, Sheldon T. Brown, Victoria Davey, Abhinav Diwan, John B. Harley, Mark Holodniy, Vincent C. Marconi, Jonathan Moorman, Emerson B. Padiernos, Ian F. Robey, Maria Rodriguez-Barradas, Jason Wertheim, Christopher W. Woods.
1. Lipshy KA, Itani K, Chu D, et al. Sentinel contributions of US Department of Veterans Affairs surgeons in shaping the face of health care. JAMA Surg. 2021;156(4):380-386. doi:10.1001/jamasurg.2020.6372
2. Zucker S, Crabbe JC, Cooper G 4th, et al. Veterans Administration support for medical research: opinions of the endangered species of physician-scientists. FASEB J. 2004;18(13):1481-1486. doi:10.1096/fj.04-1573lfe
3. Harley JB, Pyarajan S, Partan ES, et al. The US Department of Veterans Affairs Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD): a biorepository addressing national health threats. Open Forum Infect Dis. 2022;9(12):ofac641. doi:10.1093/ofid/ofac641
4. Meites E, Bajema KL, Kambhampati A, et al; SUPERNOVA COVID-19 Surveillance Group. Adapting the Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) for COVID-19 among hospitalized adults: surveillance protocol. Front Public Health. 2021;9:739076. doi:10.3389/fpubh.2021.739076
5. Bajema KL, Dahl RM, Evener SL, et al; SUPERNOVA COVID-19 Surveillance Group; Surveillance Platform for Enteric and Respiratory Infectious Organisms at the VA (SUPERNOVA) COVID-19 Surveillance Group. Comparative effectiveness and antibody responses to Moderna and Pfizer-BioNTech COVID-19 vaccines among hospitalized veterans–five Veterans Affairs Medical Centers, United States, February 1-September 30, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(49):1700-1705. doi:10.15585/mmwr.mm7049a2external icon
6. Krishnan J, Woods C, Holodniy M, et al. Nationwide genomic surveillance and response to coronavirus disease 2019 (COVID-19): SeqCURE and SeqFORCE consortiums. Fed Pract. 2023;40(suppl 5):S44-S47. doi:10.12788/fp.0417
7. Ashcroft JW, Macpherson CC. The complex ethical landscape of biobanking. Lancet Public Health. 2019;(6):e274-e275. doi:10.1016/S2468-2667(19)30081-7
8. Principle-Based Ethics Framework for Access to and Use of Veteran Data. Fed Regist. 2022;87(129):40451-40452.
The Veterans Health Administration (VHA) clinicians, clinician-investigators, and investigators perform basic and translational research for the benefit of our nation and are widely recognized for treating patients and discovering cures.1,2 In May 2020, the US Department of Veterans Affairs (VA) launched the VA Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD). The goal of this novel enterprise was to assemble a comprehensive specimen and data repository for emerging life-threatening diseases and to address future challenges. VA SHIELD was specifically charged with creating a biorepository to advance research, improve diagnostic and therapeutic capabilities, and develop strategies for immediate deployment to VA clinical environments. One main objective of VA SHIELD is to harness the clinical and scientific strengths of the VA in order to create a more cohesive collaboration between preexisting clinical research efforts within the VA.
ANATOMY OF VA SHIELD
The charge and scope of VA SHIELD is unique.3 As an entity, this program leverages the strengths of the diverse VHA network, has a broad potential impact on national health care, is positioned to respond rapidly to national and international health-related events, and substantially contributes to clinical research and development. In addition, VA SHIELD upholds VA’s Fourth Mission, which is to contribute to national emergencies and support emergency management, public health, safety, and homeland security efforts.
VA SHIELD is part of the VA Office of Research and Development (ORD). The coordinating center (CC), headquartered in Cleveland, Ohio, is the central operational partner, leading VA SHIELD and interacting with other important VA programs, including laboratory, clinical science, rehabilitation, and health services. The VA SHIELD CC oversees all aspects of operations, including biospecimen collection, creating and enforcing of standard operating procedures, ensuring the quality of the samples, processing research applications, distribution of samples, financing, and progress reports. The CC also initiates and maintains interagency collaborations, convenes stakeholders, and develops strategic plans to address emerging diseases.
The VA SHIELD Executive Steering Committee (ESC) is composed of infectious disease, biorepository, and public health specialists. The ESC provides scientific and programmatic direction to the CC, including operational activities and guidance regarding biorepository priorities and scientific agenda, and measuring and reporting on VA SHIELD accomplishments.
The primary function of the Programmatic and Scientific Review Board (PSRB) is to evaluate incoming research proposals for specimen and data use for feasibility and make recommendations to the VA SHIELD CC. The PSRB evaluates and ensures that data and specimen use align with VA SHIELD ethical, clinical, and scientific objectives.
VA SHIELD IN PRACTICE
VA SHIELD consisted of 11 specimen collection sites (Atlanta, GA; Boise, ID; Bronx, NY; Cincinnati, OH; Cleveland, OH; Durham, NC; Houston, TX; Los Angeles, CA; Mountain Home, TN; Palo Alto, CA; and Tucson, AZ), a data processing center in Boston, MA, and 2 central biorepositories in Palo Alto, CA, and Tucson, AZ. Information flow is a coordinated process among specimen collection sites, data processing centers, and the biorepositories. Initially, each local collection site identifies residual specimens that would have been discarded after clinical laboratory testing. These samples currently account for the majority of biological material within VA SHIELD via a novel collection protocol known as “Sweep,” which allows residual clinical discarded samples as well as samples from patients with new emerging infectious and noninfectious diseases of concern to be collected at the time of first emergence and submitted to VA SHIELD during the course of routine veteran health care.3 These clinical discarded samples are de-identified and transferred from the clinical laboratory to VA SHIELD. The VA Central Institutional Review Board (cIRB) has approved the use of these samples as nonhuman subject research. Biological samples are collected, processed, aliquoted, shipped to, and stored at the central biorepository sites.
The Umbrella amendment to Sweep that has been approved also by the VA cIRB, will allow VA SHIELD sites to prospectively consent veterans and collect biospecimens and additional clinical and self-reported information. The implementation of Umbrella could significantly enhance collection and research. Although Sweep is a onetime collection of samples, the Umbrella protocol will allow the longitudinal collection of samples from the same patient. Additionally, the Umbrella amendment will allow VA SHIELD to accept samples from other preexisting biorepositories or specimen collections.
Central Biorepositories
VA SHIELD has a federated organization with 2 central specimen biorepositories (Palo Alto, CA and Tucson, AZ), and an enterprise data processing center (Boston, MA). The specimen biorepositories receive de-identified specimens that are stored until distribution to approved research projects. The samples and data are linked using an electronic honest broker system to protect privacy, which integrates de-identified specimens with requested clinical and demographic data as needed for approved projects. The honest broker system is operated by independent personnel and does not have vested interest in any studies being performed under VA SHIELD. The integration of sample and associated data is done only as needed when characterization of the donor/participant is necessary byresearch aims or project outcomes. The process is facilitated by a nationally supported laboratory information management system (LIMS), managed by the VA SHIELD data center, that assists with all data requests. The clinical and demographic data are collected from VA electronic health record (EHR), available through VA Corporate Data Warehouse (CDW) and VA Informatics and Computing Infrastructure (VINCI) as needed and integrated with the biorepository samples information for approved VA SHIELD studies. The CDW is the largest longitudinal EHR data collection in the US and has the ability to provide access to national clinical and demographic data.
VA SHIELD interacts with multiple VA programs and other entities (Figure). For example, Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) is a network of 5 VA medical centers supported by the Centers for Disease Control and Prevention.4 Its initial goal was to perform surveillance for acute gastroenteritis. In 2020, SUPERNOVA shifted to conduct surveillance for COVID-19 variants among veterans.5 VA SHIELD also interacts with VHA genomic surveillance and sequencing programs: the VA Sequencing Collaborations United for Research and Epidemiology (SeqCURE) and VA Sequencing for Research Clinical and Epidemiology (SeqFORCE), described by Krishnan and colleagues.6
Working Groups
To encourage research projects that use biospecimens, VA SHIELD developed content-oriented research working groups. The goal is to inspire collaborations between VA scientists and prevent redundant or overlapping projects. Currently working groups are focused on long COVID, and COVID-19 neurology, pathogen host response, epidemiology and sequencing, cancer and cancer biomarkers, antimicrobial resistance, and vector-borne diseases. Working groups meet regularly to discuss projects and report progress. Working groups also may consider samples that might benefit VA health research and identify potential veteran populations for future research. Working groups connect VA SHIELD and investigators and guide the collection and use of resources.
Ethical Considerations
We recognize the significant ethical concerns for biobanking of specimens. However, there is no general consensus or guideline that addresses all of the complex ethical issues regarding biobanking.7 To address these ethical concerns, we applied the VA Ethical Framework Principles for Access to and Use of Veteran Data principles to VA SHIELD, including all parties who oversee the access to, sharing of, or the use of data, or who access or use its data.8
Conclusions
The VA has assembled a scientific enterprise dedicated to combating emerging infectious diseases and other threats to our patients. This enterprise has been modeled in its structure and oversight to support VA SHIELD. The establishment of a real-time biorepository and data procurement system linked to clinical samples is a bold step forward to address current and future challenges. Similarly, the integration and cooperation of multiple arms within the VA that transcend disciplines and boundaries promise to shepherd a new era of system-wide investigation. In the future, VA SHIELD will integrate with other existing government agencies to advance mutual scientific agendas. VA SHIELD has established the data and biorepository infrastructure to develop innovative and novel technologies to address future challenges. The alignment of basic science, clinical, and translational research goals under one governance is a significant advancement compared with previous models of research coordination.
VA SHIELD was developed to meet an immediate need; it was also framed to be a research enterprise that harnesses the robust clinical and research environment in VHA. The VA SHIELD infrastructure was conceptualized to harmonize specimen and data collection across the VA, allowing researchers to leverage broader collection efforts. Building a biorepository and data collection system within the largest integrated health care system has the potential to provide a lasting impact on VHA and on our nation’s health.
Acknowledgments
The authors wish to acknowledge Ms. Daphne Swancutt for her contribution as copywriter for this manuscript. The authors wish to acknowledge the VA SHIELD investigators: Mary Cloud Ammons, David Beenhouwer, Sheldon T. Brown, Victoria Davey, Abhinav Diwan, John B. Harley, Mark Holodniy, Vincent C. Marconi, Jonathan Moorman, Emerson B. Padiernos, Ian F. Robey, Maria Rodriguez-Barradas, Jason Wertheim, Christopher W. Woods.
The Veterans Health Administration (VHA) clinicians, clinician-investigators, and investigators perform basic and translational research for the benefit of our nation and are widely recognized for treating patients and discovering cures.1,2 In May 2020, the US Department of Veterans Affairs (VA) launched the VA Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD). The goal of this novel enterprise was to assemble a comprehensive specimen and data repository for emerging life-threatening diseases and to address future challenges. VA SHIELD was specifically charged with creating a biorepository to advance research, improve diagnostic and therapeutic capabilities, and develop strategies for immediate deployment to VA clinical environments. One main objective of VA SHIELD is to harness the clinical and scientific strengths of the VA in order to create a more cohesive collaboration between preexisting clinical research efforts within the VA.
ANATOMY OF VA SHIELD
The charge and scope of VA SHIELD is unique.3 As an entity, this program leverages the strengths of the diverse VHA network, has a broad potential impact on national health care, is positioned to respond rapidly to national and international health-related events, and substantially contributes to clinical research and development. In addition, VA SHIELD upholds VA’s Fourth Mission, which is to contribute to national emergencies and support emergency management, public health, safety, and homeland security efforts.
VA SHIELD is part of the VA Office of Research and Development (ORD). The coordinating center (CC), headquartered in Cleveland, Ohio, is the central operational partner, leading VA SHIELD and interacting with other important VA programs, including laboratory, clinical science, rehabilitation, and health services. The VA SHIELD CC oversees all aspects of operations, including biospecimen collection, creating and enforcing of standard operating procedures, ensuring the quality of the samples, processing research applications, distribution of samples, financing, and progress reports. The CC also initiates and maintains interagency collaborations, convenes stakeholders, and develops strategic plans to address emerging diseases.
The VA SHIELD Executive Steering Committee (ESC) is composed of infectious disease, biorepository, and public health specialists. The ESC provides scientific and programmatic direction to the CC, including operational activities and guidance regarding biorepository priorities and scientific agenda, and measuring and reporting on VA SHIELD accomplishments.
The primary function of the Programmatic and Scientific Review Board (PSRB) is to evaluate incoming research proposals for specimen and data use for feasibility and make recommendations to the VA SHIELD CC. The PSRB evaluates and ensures that data and specimen use align with VA SHIELD ethical, clinical, and scientific objectives.
VA SHIELD IN PRACTICE
VA SHIELD consisted of 11 specimen collection sites (Atlanta, GA; Boise, ID; Bronx, NY; Cincinnati, OH; Cleveland, OH; Durham, NC; Houston, TX; Los Angeles, CA; Mountain Home, TN; Palo Alto, CA; and Tucson, AZ), a data processing center in Boston, MA, and 2 central biorepositories in Palo Alto, CA, and Tucson, AZ. Information flow is a coordinated process among specimen collection sites, data processing centers, and the biorepositories. Initially, each local collection site identifies residual specimens that would have been discarded after clinical laboratory testing. These samples currently account for the majority of biological material within VA SHIELD via a novel collection protocol known as “Sweep,” which allows residual clinical discarded samples as well as samples from patients with new emerging infectious and noninfectious diseases of concern to be collected at the time of first emergence and submitted to VA SHIELD during the course of routine veteran health care.3 These clinical discarded samples are de-identified and transferred from the clinical laboratory to VA SHIELD. The VA Central Institutional Review Board (cIRB) has approved the use of these samples as nonhuman subject research. Biological samples are collected, processed, aliquoted, shipped to, and stored at the central biorepository sites.
The Umbrella amendment to Sweep that has been approved also by the VA cIRB, will allow VA SHIELD sites to prospectively consent veterans and collect biospecimens and additional clinical and self-reported information. The implementation of Umbrella could significantly enhance collection and research. Although Sweep is a onetime collection of samples, the Umbrella protocol will allow the longitudinal collection of samples from the same patient. Additionally, the Umbrella amendment will allow VA SHIELD to accept samples from other preexisting biorepositories or specimen collections.
Central Biorepositories
VA SHIELD has a federated organization with 2 central specimen biorepositories (Palo Alto, CA and Tucson, AZ), and an enterprise data processing center (Boston, MA). The specimen biorepositories receive de-identified specimens that are stored until distribution to approved research projects. The samples and data are linked using an electronic honest broker system to protect privacy, which integrates de-identified specimens with requested clinical and demographic data as needed for approved projects. The honest broker system is operated by independent personnel and does not have vested interest in any studies being performed under VA SHIELD. The integration of sample and associated data is done only as needed when characterization of the donor/participant is necessary byresearch aims or project outcomes. The process is facilitated by a nationally supported laboratory information management system (LIMS), managed by the VA SHIELD data center, that assists with all data requests. The clinical and demographic data are collected from VA electronic health record (EHR), available through VA Corporate Data Warehouse (CDW) and VA Informatics and Computing Infrastructure (VINCI) as needed and integrated with the biorepository samples information for approved VA SHIELD studies. The CDW is the largest longitudinal EHR data collection in the US and has the ability to provide access to national clinical and demographic data.
VA SHIELD interacts with multiple VA programs and other entities (Figure). For example, Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) is a network of 5 VA medical centers supported by the Centers for Disease Control and Prevention.4 Its initial goal was to perform surveillance for acute gastroenteritis. In 2020, SUPERNOVA shifted to conduct surveillance for COVID-19 variants among veterans.5 VA SHIELD also interacts with VHA genomic surveillance and sequencing programs: the VA Sequencing Collaborations United for Research and Epidemiology (SeqCURE) and VA Sequencing for Research Clinical and Epidemiology (SeqFORCE), described by Krishnan and colleagues.6
Working Groups
To encourage research projects that use biospecimens, VA SHIELD developed content-oriented research working groups. The goal is to inspire collaborations between VA scientists and prevent redundant or overlapping projects. Currently working groups are focused on long COVID, and COVID-19 neurology, pathogen host response, epidemiology and sequencing, cancer and cancer biomarkers, antimicrobial resistance, and vector-borne diseases. Working groups meet regularly to discuss projects and report progress. Working groups also may consider samples that might benefit VA health research and identify potential veteran populations for future research. Working groups connect VA SHIELD and investigators and guide the collection and use of resources.
Ethical Considerations
We recognize the significant ethical concerns for biobanking of specimens. However, there is no general consensus or guideline that addresses all of the complex ethical issues regarding biobanking.7 To address these ethical concerns, we applied the VA Ethical Framework Principles for Access to and Use of Veteran Data principles to VA SHIELD, including all parties who oversee the access to, sharing of, or the use of data, or who access or use its data.8
Conclusions
The VA has assembled a scientific enterprise dedicated to combating emerging infectious diseases and other threats to our patients. This enterprise has been modeled in its structure and oversight to support VA SHIELD. The establishment of a real-time biorepository and data procurement system linked to clinical samples is a bold step forward to address current and future challenges. Similarly, the integration and cooperation of multiple arms within the VA that transcend disciplines and boundaries promise to shepherd a new era of system-wide investigation. In the future, VA SHIELD will integrate with other existing government agencies to advance mutual scientific agendas. VA SHIELD has established the data and biorepository infrastructure to develop innovative and novel technologies to address future challenges. The alignment of basic science, clinical, and translational research goals under one governance is a significant advancement compared with previous models of research coordination.
VA SHIELD was developed to meet an immediate need; it was also framed to be a research enterprise that harnesses the robust clinical and research environment in VHA. The VA SHIELD infrastructure was conceptualized to harmonize specimen and data collection across the VA, allowing researchers to leverage broader collection efforts. Building a biorepository and data collection system within the largest integrated health care system has the potential to provide a lasting impact on VHA and on our nation’s health.
Acknowledgments
The authors wish to acknowledge Ms. Daphne Swancutt for her contribution as copywriter for this manuscript. The authors wish to acknowledge the VA SHIELD investigators: Mary Cloud Ammons, David Beenhouwer, Sheldon T. Brown, Victoria Davey, Abhinav Diwan, John B. Harley, Mark Holodniy, Vincent C. Marconi, Jonathan Moorman, Emerson B. Padiernos, Ian F. Robey, Maria Rodriguez-Barradas, Jason Wertheim, Christopher W. Woods.
1. Lipshy KA, Itani K, Chu D, et al. Sentinel contributions of US Department of Veterans Affairs surgeons in shaping the face of health care. JAMA Surg. 2021;156(4):380-386. doi:10.1001/jamasurg.2020.6372
2. Zucker S, Crabbe JC, Cooper G 4th, et al. Veterans Administration support for medical research: opinions of the endangered species of physician-scientists. FASEB J. 2004;18(13):1481-1486. doi:10.1096/fj.04-1573lfe
3. Harley JB, Pyarajan S, Partan ES, et al. The US Department of Veterans Affairs Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD): a biorepository addressing national health threats. Open Forum Infect Dis. 2022;9(12):ofac641. doi:10.1093/ofid/ofac641
4. Meites E, Bajema KL, Kambhampati A, et al; SUPERNOVA COVID-19 Surveillance Group. Adapting the Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) for COVID-19 among hospitalized adults: surveillance protocol. Front Public Health. 2021;9:739076. doi:10.3389/fpubh.2021.739076
5. Bajema KL, Dahl RM, Evener SL, et al; SUPERNOVA COVID-19 Surveillance Group; Surveillance Platform for Enteric and Respiratory Infectious Organisms at the VA (SUPERNOVA) COVID-19 Surveillance Group. Comparative effectiveness and antibody responses to Moderna and Pfizer-BioNTech COVID-19 vaccines among hospitalized veterans–five Veterans Affairs Medical Centers, United States, February 1-September 30, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(49):1700-1705. doi:10.15585/mmwr.mm7049a2external icon
6. Krishnan J, Woods C, Holodniy M, et al. Nationwide genomic surveillance and response to coronavirus disease 2019 (COVID-19): SeqCURE and SeqFORCE consortiums. Fed Pract. 2023;40(suppl 5):S44-S47. doi:10.12788/fp.0417
7. Ashcroft JW, Macpherson CC. The complex ethical landscape of biobanking. Lancet Public Health. 2019;(6):e274-e275. doi:10.1016/S2468-2667(19)30081-7
8. Principle-Based Ethics Framework for Access to and Use of Veteran Data. Fed Regist. 2022;87(129):40451-40452.
1. Lipshy KA, Itani K, Chu D, et al. Sentinel contributions of US Department of Veterans Affairs surgeons in shaping the face of health care. JAMA Surg. 2021;156(4):380-386. doi:10.1001/jamasurg.2020.6372
2. Zucker S, Crabbe JC, Cooper G 4th, et al. Veterans Administration support for medical research: opinions of the endangered species of physician-scientists. FASEB J. 2004;18(13):1481-1486. doi:10.1096/fj.04-1573lfe
3. Harley JB, Pyarajan S, Partan ES, et al. The US Department of Veterans Affairs Science and Health Initiative to Combat Infectious and Emerging Life-Threatening Diseases (VA SHIELD): a biorepository addressing national health threats. Open Forum Infect Dis. 2022;9(12):ofac641. doi:10.1093/ofid/ofac641
4. Meites E, Bajema KL, Kambhampati A, et al; SUPERNOVA COVID-19 Surveillance Group. Adapting the Surveillance Platform for Enteric and Respiratory Infectious Organisms at United States Veterans Affairs Medical Centers (SUPERNOVA) for COVID-19 among hospitalized adults: surveillance protocol. Front Public Health. 2021;9:739076. doi:10.3389/fpubh.2021.739076
5. Bajema KL, Dahl RM, Evener SL, et al; SUPERNOVA COVID-19 Surveillance Group; Surveillance Platform for Enteric and Respiratory Infectious Organisms at the VA (SUPERNOVA) COVID-19 Surveillance Group. Comparative effectiveness and antibody responses to Moderna and Pfizer-BioNTech COVID-19 vaccines among hospitalized veterans–five Veterans Affairs Medical Centers, United States, February 1-September 30, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(49):1700-1705. doi:10.15585/mmwr.mm7049a2external icon
6. Krishnan J, Woods C, Holodniy M, et al. Nationwide genomic surveillance and response to coronavirus disease 2019 (COVID-19): SeqCURE and SeqFORCE consortiums. Fed Pract. 2023;40(suppl 5):S44-S47. doi:10.12788/fp.0417
7. Ashcroft JW, Macpherson CC. The complex ethical landscape of biobanking. Lancet Public Health. 2019;(6):e274-e275. doi:10.1016/S2468-2667(19)30081-7
8. Principle-Based Ethics Framework for Access to and Use of Veteran Data. Fed Regist. 2022;87(129):40451-40452.