User login
Imaging techniques will revolutionize cancer detection, expert predicts
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
PHOENIX –
In a lecture during a multispecialty roundup of cutting-edge energy-based device applications at the annual conference of the American Society for Laser Medicine and Surgery, Dr. Barton, a biomedical engineer who directs the BIO5 Institute at the University of Arizona, Tucson, said that while no current modality exists to enable physicians in dermatology and other specialties to view internal structures throughout the entire body with cellular resolution, refining existing technologies is a good way to start.
In 2011, renowned cancer researchers Douglas Hanahan, PhD, and Robert A. Weinberg, PhD, proposed six hallmarks of cancer, which include sustaining proliferative signaling, evading growth suppressors, resisting cell death, enabling replicative immortality, inducing angiogenesis, and activating invasion and metastasis. Each hallmark poses unique imaging challenges. For example, enabling replicative immortality “means that the cell nuclei change size and shape; they change their position,” said Dr. Barton, who is also professor of biomedical engineering and optical sciences at the university. “If we want to see that, we’re going to need an imaging modality that’s subcellular in resolution.”
Similarly, if clinicians want to view how proliferative signaling is changing, “that means being able to visualize the cell surface receptors; those are even smaller to actually visualize,” she said. “But we have technologies where we can target those receptors with fluorophores. And then we can look at large areas very quickly.” Meanwhile, the ability of cancer cells to resist cell death and evade growth suppressors often results in thickening of epithelium throughout the body. “So, if we can measure the thickness of the epithelium, we can see that there’s something wrong with that tissue,” she said.
As for cancer’s propensity for invasion and metastasis, “here, we’re looking at how the collagen structure [between the cells] has changed and whether there’s layer breakdown or not. Optical imaging can detect cancer. However, high resolution optical techniques can only image about 1 mm deep, so unless you’re looking at the skin or the eye, you’re going to have to develop an endoscope to be able to view these hallmarks.”
OCT images the tissue microstructure, generally in a resolution of 2-20 microns, at a depth of 1-2 mm, and it measures reflected light. When possible, Dr. Barton combines OCT with laser-induced fluorescence for enhanced accuracy of detection of cancer. Induced fluorescence senses molecular information with the natural fluorophores in the body or with targeted exogenous agents. Then there’s multiphoton microscopy, an advanced imaging technique that enables clinicians to view cellular and subcellular events within living tissue. Early models of this technology “took up entire benches” in physics labs, Dr. Barton said, but she and other investigators are designing smaller devices for use in clinics. “This is exciting, because not only do we [view] subcellular structure with this modality, but it can also be highly sensitive to collagen structure,” she said.
Ovarian cancer model
In a model of ovarian cancer, she and colleagues externalized the ovaries of a mouse, imaged the organs, put them back in, and reassessed them at 8 weeks. “This model develops cancer very quickly,” said Dr. Barton, who once worked for McDonnell Douglas on the Space Station program. At 8 weeks, using fluorescence and targeted agents with a tabletop multiphoton microscopy system, they observed that the proliferation signals of cancer had begun. “So, with an agent targeted to the folate receptor or to other receptors that are implicated in cancer development, we can see that ovaries and fallopian tubes are lighting up,” she said.
With proof of concept established with the mouse study, she and other researchers are drawing from technological advances to create tiny laser systems for use in the clinic to image a variety of structures in the human body. Optics advances include bulk optics and all-fiber designs where engineers can create an imaging probe that’s only 125 microns in diameter, “or maybe even as small as 70 microns in diameter,” she said. “We can do fabrications on the tips of endoscopes to redirect the light and focus it. We can also do 3-D printing and spiral scanning to create miniature devices to make new advances. That means that instead of just white light imaging of the colon or the lung like we have had in the past, we can start moving into smaller structures, such as the eustachian tube, the fallopian tube, the bile ducts, or making miniature devices for brain biopsies, lung biopsies, and maybe being able to get into bronchioles and arterioles.”
According to Dr. Barton, prior research has demonstrated that cerebral vasculature can be imaged with a catheter 400 microns in diameter, the spaces in the lungs can be imaged with a needle that is 310 microns in diameter, and the inner structures of the eustachian tube can be viewed with an endoscope 1 mm in diameter.
She and her colleagues are developing an OCT/fluorescence imaging falloposcope that is 0.8 mm in diameter, flexible, and steerable, as a tool for early detection of ovarian cancer in humans. “It’s now known that most ovarian cancer starts in the fallopian tubes,” Dr. Barton said. “It’s metastatic disease when those cells break off from the fallopian tubes and go to the ovaries. We wanted to create an imaging system where we created a fiber bundle that we could navigate with white light and with fluorescence so that we can see these early stages of cancer [and] how they fluoresce differently. We also wanted to have an OCT system so that we could image through the wall of the fallopian tube and look for that layer thickening and other precursors to ovarian cancer.”
To date, in vivo testing in healthy women has demonstrated that the miniature endoscope is able to reach the fallopian tubes through the natural orifice of the vagina and uterus. “That is pretty exciting,” she said. “The images may not be of the highest quality, but we are advancing.”
Dr. Barton reported having no relevant financial disclosures.
AT ASLMS 2023
FDA approves ritlecitinib for ages 12 and up for alopecia areata
Taken as a once-daily pill, ritlecitinib is a dual inhibitor of the TEC family of tyrosine kinases and of Janus kinase 3 (JAK3). The recommended dose of ritlecitinib, which will be marketed as Litfulo, is 50 mg once a day, according to the statement announcing the approval from Pfizer.
It is the second JAK inhibitor approved for treating alopecia areata, following approval of baricitinib (Olumiant) in June 2022 for AA in adults. Ritlecitinib is the first JAK inhibitor approved for children ages 12 and older with AA.
The European Medicines Agency has also accepted the Marketing Authorization Application for ritlecitinib in the same population and a decision is expected in the fourth quarter of this year.
Approval based on ALLEGRO trials
Approval was based on previously announced results from trials, including the phase 2b/3 ALLEGRO study of ritlecitinib in 718 patients aged 12 years and older with alopecia areata, with 50% of more scalp hair loss, as measured by the Severity of Alopecia Tool (SALT), including patients with alopecia totalis (complete scalp hair loss) and alopecia universalis (complete scalp, face, and body hair loss).
Patients in the trial were experiencing a current episode of alopecia areata that had lasted between 6 months and 10 years. They were randomized to receive once-daily ritlecitinib at doses of 30 mg or 50 mg (with or without 1 month of initial treatment with once-daily ritlecitinib 200 mg), ritlecitinib 10 mg, or placebo.
Statistically significantly higher proportions of patients treated with ritlecitinib 30 mg and 50 mg (with or without the loading dose) had 80% or more scalp hair coverage, as measured by a SALT score of 20 or less after 6 months of treatment versus placebo. After 6 months of treatment, among those on the 50-mg dose, 23% had achieved a SALT score of 20 or less, compared with 2% of those on placebo. The results were published in The Lancet.
According to the company release, efficacy and safety of ritlecitinib was consistent between those ages 12-17 and adults, and the most common adverse events reported in the study, in at least 4% of patients treated with ritlecitinib, were headache (10.8%), diarrhea (10%), acne (6.2%), rash (5.4%), and urticaria (4.6%).
Ritlecitinib labeling includes the boxed warning about the risk for serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis, which is included in the labels for other JAK inhibitors.
Ritlecitinib evaluated for other diseases
In addition to alopecia areata, ritlecitinib has shown efficacy and acceptable safety in treating ulcerative colitis and is being evaluated for treating vitiligo, Crohn’s disease, and rheumatoid arthritis.
In the statement, the company says that ritlecitinib will be available “in the coming weeks.” The manufacturer says it also has completed regulatory submissions for ritlecitinib in the United Kingdom, China, and Japan, and expects decisions this year.
Alopecia areata affects about 6.8 million people in the United States and 147 million globally.
In a statement, Nicole Friedland, president and CEO of the National Alopecia Areata Foundation, said that NAAF “is thrilled to have a second FDA-approved treatment for alopecia areata, which is the first approved for adolescents.”
A version of this article first appeared on Medscape.com.
Taken as a once-daily pill, ritlecitinib is a dual inhibitor of the TEC family of tyrosine kinases and of Janus kinase 3 (JAK3). The recommended dose of ritlecitinib, which will be marketed as Litfulo, is 50 mg once a day, according to the statement announcing the approval from Pfizer.
It is the second JAK inhibitor approved for treating alopecia areata, following approval of baricitinib (Olumiant) in June 2022 for AA in adults. Ritlecitinib is the first JAK inhibitor approved for children ages 12 and older with AA.
The European Medicines Agency has also accepted the Marketing Authorization Application for ritlecitinib in the same population and a decision is expected in the fourth quarter of this year.
Approval based on ALLEGRO trials
Approval was based on previously announced results from trials, including the phase 2b/3 ALLEGRO study of ritlecitinib in 718 patients aged 12 years and older with alopecia areata, with 50% of more scalp hair loss, as measured by the Severity of Alopecia Tool (SALT), including patients with alopecia totalis (complete scalp hair loss) and alopecia universalis (complete scalp, face, and body hair loss).
Patients in the trial were experiencing a current episode of alopecia areata that had lasted between 6 months and 10 years. They were randomized to receive once-daily ritlecitinib at doses of 30 mg or 50 mg (with or without 1 month of initial treatment with once-daily ritlecitinib 200 mg), ritlecitinib 10 mg, or placebo.
Statistically significantly higher proportions of patients treated with ritlecitinib 30 mg and 50 mg (with or without the loading dose) had 80% or more scalp hair coverage, as measured by a SALT score of 20 or less after 6 months of treatment versus placebo. After 6 months of treatment, among those on the 50-mg dose, 23% had achieved a SALT score of 20 or less, compared with 2% of those on placebo. The results were published in The Lancet.
According to the company release, efficacy and safety of ritlecitinib was consistent between those ages 12-17 and adults, and the most common adverse events reported in the study, in at least 4% of patients treated with ritlecitinib, were headache (10.8%), diarrhea (10%), acne (6.2%), rash (5.4%), and urticaria (4.6%).
Ritlecitinib labeling includes the boxed warning about the risk for serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis, which is included in the labels for other JAK inhibitors.
Ritlecitinib evaluated for other diseases
In addition to alopecia areata, ritlecitinib has shown efficacy and acceptable safety in treating ulcerative colitis and is being evaluated for treating vitiligo, Crohn’s disease, and rheumatoid arthritis.
In the statement, the company says that ritlecitinib will be available “in the coming weeks.” The manufacturer says it also has completed regulatory submissions for ritlecitinib in the United Kingdom, China, and Japan, and expects decisions this year.
Alopecia areata affects about 6.8 million people in the United States and 147 million globally.
In a statement, Nicole Friedland, president and CEO of the National Alopecia Areata Foundation, said that NAAF “is thrilled to have a second FDA-approved treatment for alopecia areata, which is the first approved for adolescents.”
A version of this article first appeared on Medscape.com.
Taken as a once-daily pill, ritlecitinib is a dual inhibitor of the TEC family of tyrosine kinases and of Janus kinase 3 (JAK3). The recommended dose of ritlecitinib, which will be marketed as Litfulo, is 50 mg once a day, according to the statement announcing the approval from Pfizer.
It is the second JAK inhibitor approved for treating alopecia areata, following approval of baricitinib (Olumiant) in June 2022 for AA in adults. Ritlecitinib is the first JAK inhibitor approved for children ages 12 and older with AA.
The European Medicines Agency has also accepted the Marketing Authorization Application for ritlecitinib in the same population and a decision is expected in the fourth quarter of this year.
Approval based on ALLEGRO trials
Approval was based on previously announced results from trials, including the phase 2b/3 ALLEGRO study of ritlecitinib in 718 patients aged 12 years and older with alopecia areata, with 50% of more scalp hair loss, as measured by the Severity of Alopecia Tool (SALT), including patients with alopecia totalis (complete scalp hair loss) and alopecia universalis (complete scalp, face, and body hair loss).
Patients in the trial were experiencing a current episode of alopecia areata that had lasted between 6 months and 10 years. They were randomized to receive once-daily ritlecitinib at doses of 30 mg or 50 mg (with or without 1 month of initial treatment with once-daily ritlecitinib 200 mg), ritlecitinib 10 mg, or placebo.
Statistically significantly higher proportions of patients treated with ritlecitinib 30 mg and 50 mg (with or without the loading dose) had 80% or more scalp hair coverage, as measured by a SALT score of 20 or less after 6 months of treatment versus placebo. After 6 months of treatment, among those on the 50-mg dose, 23% had achieved a SALT score of 20 or less, compared with 2% of those on placebo. The results were published in The Lancet.
According to the company release, efficacy and safety of ritlecitinib was consistent between those ages 12-17 and adults, and the most common adverse events reported in the study, in at least 4% of patients treated with ritlecitinib, were headache (10.8%), diarrhea (10%), acne (6.2%), rash (5.4%), and urticaria (4.6%).
Ritlecitinib labeling includes the boxed warning about the risk for serious infections, mortality, malignancy, major adverse cardiovascular events, and thrombosis, which is included in the labels for other JAK inhibitors.
Ritlecitinib evaluated for other diseases
In addition to alopecia areata, ritlecitinib has shown efficacy and acceptable safety in treating ulcerative colitis and is being evaluated for treating vitiligo, Crohn’s disease, and rheumatoid arthritis.
In the statement, the company says that ritlecitinib will be available “in the coming weeks.” The manufacturer says it also has completed regulatory submissions for ritlecitinib in the United Kingdom, China, and Japan, and expects decisions this year.
Alopecia areata affects about 6.8 million people in the United States and 147 million globally.
In a statement, Nicole Friedland, president and CEO of the National Alopecia Areata Foundation, said that NAAF “is thrilled to have a second FDA-approved treatment for alopecia areata, which is the first approved for adolescents.”
A version of this article first appeared on Medscape.com.
Study supports new NCCN classification for cutaneous squamous cell carcinoma
, according to new findings.
In addition, regardless of the NCCN risk group, the study found that Mohs surgery or peripheral and deep en face margin assessment (PDEMA) conferred a lower risk of developing LR, DM, and disease-related death.
“Although the NCCN included this new high-risk group in the last iteration of the guidelines, there were no studies that identified whether the high-risk group achieved the goal of identifying riskier tumors,” said senior author Emily Ruiz, MD, MPH, associate physician at the Mohs and Dermatologic Surgery Center at Brigham and Women’s Faulkner Hospital, Boston. “Based on the data in our study, the risk groups did risk stratify tumors and so clinicians can utilize the high-risk group risk factors to identify which tumors may require additional surveillance or treatment.”
The study was published online in JAMA Dermatology.
Most patients with CSCC are successfully treated with Mohs micrographic surgery or wide local excision (WLE) alone, but a subset will experience more severe and aggressive disease. While useful for prognostication, current staging systems do not incorporate patient factors or other high-risk tumor features that influence outcomes, which led to the NCCN reclassifying CSCC into low-, high-, and very high-risk groups. The NCCN guidelines also made a new recommendation that Mohs or PDEMA be the preferred method for tissue processing for high- and very-high-risk tumors, based on this new stratification.
However, these changes to the NCCN guidelines have not been validated. The goal of this study was to compare outcomes in very-high-, high-, and low-risk NCCN groups as well as comparing outcomes of CSCCs stratified by Mohs and WLE.
Dr. Ruiz and colleagues conducted a retrospective cohort study using patient data from two tertiary care academic medical centers. Their analysis included 10,196 tumors from 8,727 patients that were then stratified into low-risk (3,054 tumors [30.0%]), high-risk (6,269 tumors [61.5%]), and very-high-risk (873 tumors [8.6%]) groups.
Tumors in the very-high-risk group were more likely to have high-risk tumor and histologic features, such as large-caliber perineural invasion, large diameter, invasion beyond the subcutaneous fat or bone, poor differentiation, and lymphovascular invasion.
The authors found that, compared with the low-risk group, the high- and very-high-risk groups demonstrated a greater risk of LR (high-risk subhazard ratio, 1.99; P = .007; very-high-risk SHR, 12.66; P < .001); NM (high-risk SHR, 4.26; P = .02; very-high-risk SHR, 62.98; P < .001); DM (high-risk SHR, 2.2 × 107; P < .001; very-high-risk SHR, 6.3 × 108; P < .001); and DSD (high-risk SHR, 4.02; P = .03; very-high-risk SHR, 93.87; P < .001).
Adjusted 5-year cumulative incidence was also significantly higher in very-high- vs. high- and low-risk groups for all endpoints.
They next compared the procedures used to treat the tumors. Compared with WLE, patients treated with Mohs or PDEMA had a lower risk of LR (SHR, 0.65; P = .009), DM (SHR, 0.38; P = .02), and DSD (SHR, 0.55; P = .006).
Mohs and PDEMA have already became preferred surgical modalities for high- and very-high-risk tumors, and Dr. Ruiz pointed out that their analysis was for the entire cohort.
“We did not stratify this by risk group,” she said. “So our results do not change anything clinically at this time, but support prior studies that have found Mohs/PDEMA to have improved outcomes, compared to WLE. Further studies are needed evaluating surgical approach by risk-group.”
However, she emphasized, “our studies further validate prior evidence showing Mohs/PDEMA to have the lowest rates of recurrence and in this study, even disease-related death.”
Approached for an independent comment, Jeffrey M. Farma, MD, codirector of the melanoma and skin cancer program, and interim chair, department of surgical oncology, Fox Chase Cancer Center, Philadelphia, noted that this study supports the new reclassification of CSCC tumors by the NCCN, and confirms that the high-risk and very-high-risk tumors surely have a higher propensity for worse outcomes overall.
“That being said, the notion for type of resection and margin assessment is still an area of controversy in the dermatology, surgical oncology, and pathology community,” said Dr. Farma, who is also on the NCCN panel. “I believe we need further studies to truly understand the role of the type of resection and the pathologic evaluation play in this disease process.”
He also pointed out that it is unclear in this dataset if patients initially had any imaging to evaluate for local or regional metastatic disease. “It would be helpful to have a further understanding of which type of provider was performing the excisions, the type of excision decided upon, and if there was a standardized approach to [decide] which patients had MOHS or PDEMA and what was the surveillance for these patients both with imaging and physical examinations,” said Dr. Farma. “This data also evaluated patients over a long time period where practice patterns have evolved.”
Finally, he noted that the number of local and metastatic events subjectively seems low in this cohort. “We also do not know any information about the initial workup of the patients, patterns of recurrence, and adjuvant or palliative treatment after recurrence,” he added. “It is unclear from this manuscript how the type of resection or pathologic evaluation of margins leads to improved outcomes and further prospective studies are warranted.”
Dr. Ruiz reports reported serving as a coinvestigator and principal investigator for Regeneron Pharmaceuticals and as a coinvestigator for Merck and consulting for Checkpoint Therapeutics, BDO, and Genentech outside the submitted work. Dr. Farma has no disclosures other than the NCCN panel. The study was supported by Harvard Catalyst and the Harvard University Clinical and Translational Science Center and by Harvard University and its affiliated academic health care centers and partially supported by the Melvin Markey Discovery Fund at Cleveland Clinic Foundation.
, according to new findings.
In addition, regardless of the NCCN risk group, the study found that Mohs surgery or peripheral and deep en face margin assessment (PDEMA) conferred a lower risk of developing LR, DM, and disease-related death.
“Although the NCCN included this new high-risk group in the last iteration of the guidelines, there were no studies that identified whether the high-risk group achieved the goal of identifying riskier tumors,” said senior author Emily Ruiz, MD, MPH, associate physician at the Mohs and Dermatologic Surgery Center at Brigham and Women’s Faulkner Hospital, Boston. “Based on the data in our study, the risk groups did risk stratify tumors and so clinicians can utilize the high-risk group risk factors to identify which tumors may require additional surveillance or treatment.”
The study was published online in JAMA Dermatology.
Most patients with CSCC are successfully treated with Mohs micrographic surgery or wide local excision (WLE) alone, but a subset will experience more severe and aggressive disease. While useful for prognostication, current staging systems do not incorporate patient factors or other high-risk tumor features that influence outcomes, which led to the NCCN reclassifying CSCC into low-, high-, and very high-risk groups. The NCCN guidelines also made a new recommendation that Mohs or PDEMA be the preferred method for tissue processing for high- and very-high-risk tumors, based on this new stratification.
However, these changes to the NCCN guidelines have not been validated. The goal of this study was to compare outcomes in very-high-, high-, and low-risk NCCN groups as well as comparing outcomes of CSCCs stratified by Mohs and WLE.
Dr. Ruiz and colleagues conducted a retrospective cohort study using patient data from two tertiary care academic medical centers. Their analysis included 10,196 tumors from 8,727 patients that were then stratified into low-risk (3,054 tumors [30.0%]), high-risk (6,269 tumors [61.5%]), and very-high-risk (873 tumors [8.6%]) groups.
Tumors in the very-high-risk group were more likely to have high-risk tumor and histologic features, such as large-caliber perineural invasion, large diameter, invasion beyond the subcutaneous fat or bone, poor differentiation, and lymphovascular invasion.
The authors found that, compared with the low-risk group, the high- and very-high-risk groups demonstrated a greater risk of LR (high-risk subhazard ratio, 1.99; P = .007; very-high-risk SHR, 12.66; P < .001); NM (high-risk SHR, 4.26; P = .02; very-high-risk SHR, 62.98; P < .001); DM (high-risk SHR, 2.2 × 107; P < .001; very-high-risk SHR, 6.3 × 108; P < .001); and DSD (high-risk SHR, 4.02; P = .03; very-high-risk SHR, 93.87; P < .001).
Adjusted 5-year cumulative incidence was also significantly higher in very-high- vs. high- and low-risk groups for all endpoints.
They next compared the procedures used to treat the tumors. Compared with WLE, patients treated with Mohs or PDEMA had a lower risk of LR (SHR, 0.65; P = .009), DM (SHR, 0.38; P = .02), and DSD (SHR, 0.55; P = .006).
Mohs and PDEMA have already became preferred surgical modalities for high- and very-high-risk tumors, and Dr. Ruiz pointed out that their analysis was for the entire cohort.
“We did not stratify this by risk group,” she said. “So our results do not change anything clinically at this time, but support prior studies that have found Mohs/PDEMA to have improved outcomes, compared to WLE. Further studies are needed evaluating surgical approach by risk-group.”
However, she emphasized, “our studies further validate prior evidence showing Mohs/PDEMA to have the lowest rates of recurrence and in this study, even disease-related death.”
Approached for an independent comment, Jeffrey M. Farma, MD, codirector of the melanoma and skin cancer program, and interim chair, department of surgical oncology, Fox Chase Cancer Center, Philadelphia, noted that this study supports the new reclassification of CSCC tumors by the NCCN, and confirms that the high-risk and very-high-risk tumors surely have a higher propensity for worse outcomes overall.
“That being said, the notion for type of resection and margin assessment is still an area of controversy in the dermatology, surgical oncology, and pathology community,” said Dr. Farma, who is also on the NCCN panel. “I believe we need further studies to truly understand the role of the type of resection and the pathologic evaluation play in this disease process.”
He also pointed out that it is unclear in this dataset if patients initially had any imaging to evaluate for local or regional metastatic disease. “It would be helpful to have a further understanding of which type of provider was performing the excisions, the type of excision decided upon, and if there was a standardized approach to [decide] which patients had MOHS or PDEMA and what was the surveillance for these patients both with imaging and physical examinations,” said Dr. Farma. “This data also evaluated patients over a long time period where practice patterns have evolved.”
Finally, he noted that the number of local and metastatic events subjectively seems low in this cohort. “We also do not know any information about the initial workup of the patients, patterns of recurrence, and adjuvant or palliative treatment after recurrence,” he added. “It is unclear from this manuscript how the type of resection or pathologic evaluation of margins leads to improved outcomes and further prospective studies are warranted.”
Dr. Ruiz reports reported serving as a coinvestigator and principal investigator for Regeneron Pharmaceuticals and as a coinvestigator for Merck and consulting for Checkpoint Therapeutics, BDO, and Genentech outside the submitted work. Dr. Farma has no disclosures other than the NCCN panel. The study was supported by Harvard Catalyst and the Harvard University Clinical and Translational Science Center and by Harvard University and its affiliated academic health care centers and partially supported by the Melvin Markey Discovery Fund at Cleveland Clinic Foundation.
, according to new findings.
In addition, regardless of the NCCN risk group, the study found that Mohs surgery or peripheral and deep en face margin assessment (PDEMA) conferred a lower risk of developing LR, DM, and disease-related death.
“Although the NCCN included this new high-risk group in the last iteration of the guidelines, there were no studies that identified whether the high-risk group achieved the goal of identifying riskier tumors,” said senior author Emily Ruiz, MD, MPH, associate physician at the Mohs and Dermatologic Surgery Center at Brigham and Women’s Faulkner Hospital, Boston. “Based on the data in our study, the risk groups did risk stratify tumors and so clinicians can utilize the high-risk group risk factors to identify which tumors may require additional surveillance or treatment.”
The study was published online in JAMA Dermatology.
Most patients with CSCC are successfully treated with Mohs micrographic surgery or wide local excision (WLE) alone, but a subset will experience more severe and aggressive disease. While useful for prognostication, current staging systems do not incorporate patient factors or other high-risk tumor features that influence outcomes, which led to the NCCN reclassifying CSCC into low-, high-, and very high-risk groups. The NCCN guidelines also made a new recommendation that Mohs or PDEMA be the preferred method for tissue processing for high- and very-high-risk tumors, based on this new stratification.
However, these changes to the NCCN guidelines have not been validated. The goal of this study was to compare outcomes in very-high-, high-, and low-risk NCCN groups as well as comparing outcomes of CSCCs stratified by Mohs and WLE.
Dr. Ruiz and colleagues conducted a retrospective cohort study using patient data from two tertiary care academic medical centers. Their analysis included 10,196 tumors from 8,727 patients that were then stratified into low-risk (3,054 tumors [30.0%]), high-risk (6,269 tumors [61.5%]), and very-high-risk (873 tumors [8.6%]) groups.
Tumors in the very-high-risk group were more likely to have high-risk tumor and histologic features, such as large-caliber perineural invasion, large diameter, invasion beyond the subcutaneous fat or bone, poor differentiation, and lymphovascular invasion.
The authors found that, compared with the low-risk group, the high- and very-high-risk groups demonstrated a greater risk of LR (high-risk subhazard ratio, 1.99; P = .007; very-high-risk SHR, 12.66; P < .001); NM (high-risk SHR, 4.26; P = .02; very-high-risk SHR, 62.98; P < .001); DM (high-risk SHR, 2.2 × 107; P < .001; very-high-risk SHR, 6.3 × 108; P < .001); and DSD (high-risk SHR, 4.02; P = .03; very-high-risk SHR, 93.87; P < .001).
Adjusted 5-year cumulative incidence was also significantly higher in very-high- vs. high- and low-risk groups for all endpoints.
They next compared the procedures used to treat the tumors. Compared with WLE, patients treated with Mohs or PDEMA had a lower risk of LR (SHR, 0.65; P = .009), DM (SHR, 0.38; P = .02), and DSD (SHR, 0.55; P = .006).
Mohs and PDEMA have already became preferred surgical modalities for high- and very-high-risk tumors, and Dr. Ruiz pointed out that their analysis was for the entire cohort.
“We did not stratify this by risk group,” she said. “So our results do not change anything clinically at this time, but support prior studies that have found Mohs/PDEMA to have improved outcomes, compared to WLE. Further studies are needed evaluating surgical approach by risk-group.”
However, she emphasized, “our studies further validate prior evidence showing Mohs/PDEMA to have the lowest rates of recurrence and in this study, even disease-related death.”
Approached for an independent comment, Jeffrey M. Farma, MD, codirector of the melanoma and skin cancer program, and interim chair, department of surgical oncology, Fox Chase Cancer Center, Philadelphia, noted that this study supports the new reclassification of CSCC tumors by the NCCN, and confirms that the high-risk and very-high-risk tumors surely have a higher propensity for worse outcomes overall.
“That being said, the notion for type of resection and margin assessment is still an area of controversy in the dermatology, surgical oncology, and pathology community,” said Dr. Farma, who is also on the NCCN panel. “I believe we need further studies to truly understand the role of the type of resection and the pathologic evaluation play in this disease process.”
He also pointed out that it is unclear in this dataset if patients initially had any imaging to evaluate for local or regional metastatic disease. “It would be helpful to have a further understanding of which type of provider was performing the excisions, the type of excision decided upon, and if there was a standardized approach to [decide] which patients had MOHS or PDEMA and what was the surveillance for these patients both with imaging and physical examinations,” said Dr. Farma. “This data also evaluated patients over a long time period where practice patterns have evolved.”
Finally, he noted that the number of local and metastatic events subjectively seems low in this cohort. “We also do not know any information about the initial workup of the patients, patterns of recurrence, and adjuvant or palliative treatment after recurrence,” he added. “It is unclear from this manuscript how the type of resection or pathologic evaluation of margins leads to improved outcomes and further prospective studies are warranted.”
Dr. Ruiz reports reported serving as a coinvestigator and principal investigator for Regeneron Pharmaceuticals and as a coinvestigator for Merck and consulting for Checkpoint Therapeutics, BDO, and Genentech outside the submitted work. Dr. Farma has no disclosures other than the NCCN panel. The study was supported by Harvard Catalyst and the Harvard University Clinical and Translational Science Center and by Harvard University and its affiliated academic health care centers and partially supported by the Melvin Markey Discovery Fund at Cleveland Clinic Foundation.
FROM JAMA DERMATOLOGY
FDA passes on olorofim despite critical need for antifungals
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
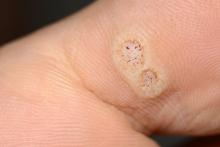
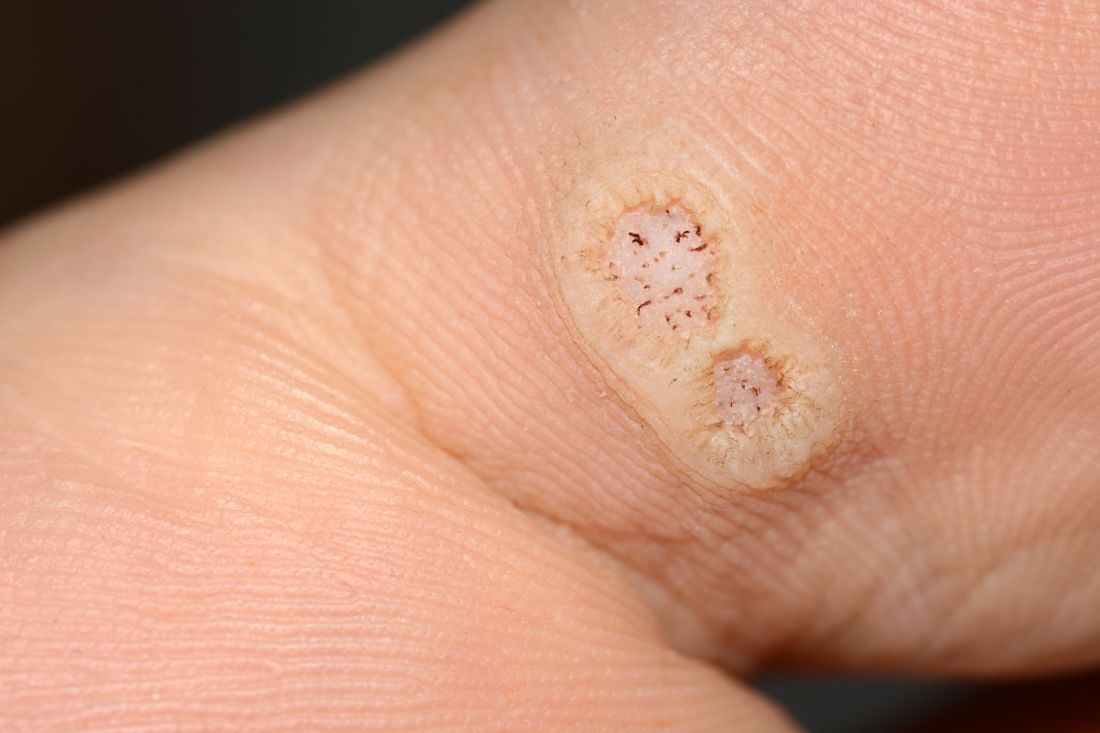
Warts difficult to eradicate in immunocompromised children
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
.
Only a quarter of patients (24%) who were undergoing active cancer treatment experienced complete resolution of their warts, compared with 63.3% of patients who were not on active treatment.
In addition, warts persisted or worsened in 56.0% of patients receiving active treatment compared with 13.4% of those who were not receiving it.
“These data enable providers treating warts in children with cancer to have an educated discussion regarding the expected clinical progression of warts and the likelihood of response to wart therapy while on and off anti-cancer treatment,” the authors wrote in the study, published in Pediatric Dermatology.
In immunocompromised children, warts are more common than in the general pediatric population, and more resistant to treatment. But as the authors noted, data on the course and prognosis of warts in pediatric patients who are actively receiving anti-cancer therapy compared with patients who have completed treatment are limited.
Tina Ho, MD, PhD, of the department of dermatology, and colleagues from Boston Children’s Hospital, sought to analyze the clinical course of warts treated in this patient population at their institution over a 10-year period. They conducted a retrospective study of 72 children who were treated for cancer between 2011 and 2021, and who had also been treated for warts.
The median age of the cohort was 12 years, and they were followed for a median of 2 years following their diagnosis of warts. Within this group, more than half (55%) had hematologic malignancies, while 27% had a history of bone marrow transplantation.
Of note, the authors pointed out, 54% of the patients had plantar warts, and 60% of patients (38 of 63) with a documented number of warts had more than five at the time of presentation.
The treatment regimens that the children had received varied, with 81% of patients receiving cytotoxic chemotherapy and 23% of patients on targeted therapies that included immunotherapy.
The warts were most commonly treated with cryotherapy and topical salicylic acid; this was the case for those actively receiving oncology treatment or those who had completed their treatment regimens.
Outcomes of wart treatments were available in 25 of the patients undergoing active cancer treatment and in 30 of those who had completed treatment. For children on active oncology treatment, 5 (20%) achieved partial resolution, 6 (24%) achieved complete resolution, and 14 (56%) experienced persistence or worsening of their warts following therapy. Those who had completed treatment had better outcomes: Seven (23.3%) had a partial response, 19 (63.3%) had complete resolution, and 4 (13.4%) had persistence or worsening of warts after treatment of warts.
The authors also pointed out the treatment of warts can be painful, expensive, and time-consuming. “It is thus imperative that the risks and benefits of these treatments are carefully considered before proceeding with treatment,” wrote Dr. Ho and colleagues. “This is especially true in medically complex children with cancer who may be fearful of procedures and spend significant portions of their young lives within the medical system.”
Limitations to the study include its retrospective design and small sample size. Clinical data were not uniformly complete, and follow-up intervals varied among the participants. Also, it was conducted at a single-institution and at a large tertiary center, so the results may not be fully generalizable.
The authors declared no conflict of interest. No outside funding source was listed.
FROM PEDIATRIC DERMATOLOGY
Prognostic factors of SCCs in organ transplant recipients worse compared with general population
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
, results from a dual cohort study demonstrated.
The findings build on previous research and underscore the need for early diagnosis and aggressive surveillance in this patient population, corresponding author Adele C. Green, MBBS, PhD, professor and senior scientist at the QIMR Berghofer Medical Research Institute, Brisbane, Queensland, Australia, and colleagues wrote in the study, which was published online in JAMA Dermatology. “Squamous cell carcinomas (SCCs) of the skin develop up to 77 times more frequently in immunosuppressed organ transplant recipients (OTRs) than the general population,” they wrote. “Because SCCs cause substantially more morbidity and death in the former, they are postulated to be innately more aggressive than in immunocompetent patients, but OTRs’ higher SCC mortality may simply reflect greater SCC tumor burdens per patient.”
In what is believed to be the first study of its kind, Dr. Green and colleagues drew data from two cohort studies to evaluate five key clinicopathologic indicators of poor SCC outcomes in organ transplant recipients, and in those from the general population in Queensland, Australia: cephalic location, perineural invasion, invasion to/beyond subcutaneous fat, poor differentiation, and tumor size greater than 20 mm. The study population included organ transplant recipients at high risk of skin cancer, who were enrolled in the Skin Tumours in Allograft Recipients (STAR) study, and those from a population-based cohort, the QSkin Sun and Health Study. STAR consisted of lung transplant recipients and kidney and liver transplant recipients at high risk of skin cancer who were recruited from tertiary centers and diagnosed with histopathologically confirmed SCC from 2012 to 2015. QSkin consisted of individuals from Queensland’s general adult population diagnosed with SCCs from 2012 to 2015.
SCC cases in QSkin were ascertained through Australia’s universal health insurance agency and linked with histopathology records. Next, the researchers performed data analysis from both cohort studies to determine the prevalence ratio (PR) of head/neck location, perineural invasion, tumor invasion to/beyond subcutaneous fat, poor cellular differentiation, and tumor diameter greater than 20 mm among SCCs among organ transplant recipients compared with the general population.
After combining the two studies, the researchers compared 741 SCCs excised from 191 organ transplant recipients and 2,558 SCCs excised from 1,507 individuals in the general population. Their median ages were similar (62.7 and 63.7 years, respectively) and most were male (78% and 63.4%, respectively).
As for site of involvement, SCCs developed most often on the head and neck in the transplant recipients (38.6%) and on the arms and hands in the general population (35.2%). After adjustment for age and sex, perineural invasion of SCCs was more than twice as common in transplant recipients than among cases in the general population, as was invasion to/beyond subcutaneous fat (PR of 2.37 for both associations).
In other findings, compared with SCCs in the general population, poorly vs. well-differentiated SCCs were more than threefold more common in transplant recipients (PR, 3.45), while the prevalence of tumors greater than 20 mm vs. 20 mm or smaller was moderately higher in transplant recipients (PR, 1.52).
“These findings are considered generalizable, confirming that OTRs’ poorer SCC outcomes are associated with not only their sheer numbers of SCC tumors, but also with a strong shift toward more invasive, less differentiated, and larger SCC tumors, in agreement with previous findings,” the researchers wrote. “This shift is likely associated with decreased immunosurveillance resulting from immunosuppressive therapy (since carcinogenesis decelerates with therapy cessation) interacting with effects of high UV radiation exposure.”
They acknowledged certain limitations of their analysis, chiefly the lack of central review of SCCs to ensure standard assessment of histopathologic features “including caliber of nerves with perineural invasion and cell differentiation; such a review would not have been feasible logistically.”
The study was supported by grants from the National Health and Medical Research Council of Australia. The researchers reported having no disclosures related to the submitted work.
FROM JAMA DERMATOLOGY
A 63-year-old male presented for evaluation of worsening genital lesions and associated swelling
.1 Clinically, ENV presents as verrucous, hyperkeratotic, cobblestone-like patches, plaques, and nodules with associated nonpitting edema of the affected body area.1 Secondary bacterial infections are common and often worsen the clinical course. The etiology of ENV involves chronic lymphatic obstruction and venous insufficiency, with additional risk factors including obesity, chronic lymphedema, bacterial infection, surgery or trauma, neoplasia, radiation, congestive heart failure, or scleroderma.2,3 While most commonly presenting on the lower extremities, cases have been reported involving the abdomen, sacrum, ears, buttocks, and penoscrotal area.1,2
Regardless of location, the pathogenesis of ENV remains the same. Chronic lymphatic obstruction results in accumulation and lymphostasis of protein-rich dermal fluid, which subsequently precipitates fibroblast proliferation and activation, suppression of the local immune response and development of recurrent lymphangitis, chronic inflammation, and potential secondary bacterial infection.2,4
There is no standard of care for the treatment and management of ENV and recurrence is common. Interventions often involve those used for chronic lymphedema – including leg elevation, compression stockings or devices, skin hygiene, and lymphatic pumping.2,3 Medical management with topical and oral retinoids has been reported, as well as emphasis on weight loss and infection control.1,4 Surgical intervention is often reserved for refractory cases that fail to respond to more conservative management, or severe presentations resulting in extensive functional and aesthetic impairment. Less commonly reported treatment modalities include lymphaticovenular anastomosis and ablative carbon dioxide laser use, although this latter intervention demonstrated minimal improvement in this patient.5,6
Penoscrotal ENV is a rare form of ENV affecting the genital region of males, often resulting in significant disfigurement, functional impairment, and psychosocial distress. Penoscrotal elephantiasis can be idiopathic, due to filarial infections, scleroinflammatory stricture of the urethra, Chlamydia trachomatis infection, and lymphostasis secondary to chronic inflammatory conditions such as streptococcal infections, radiotherapy, surgery, chronic venous stasis, or Kaposi sarcoma.7
In addition, hidradenitis suppurativa (HS) has been documented multiple times in the literature in association with the development of ENV, detailing lymphatic scarring secondary to chronic inguinal HS as the main pathogenic factor.8,9
Surgery is the mainstay of treatment for penoscrotal ENV, which not only improves functionality and cosmesis, but also aids in prevention of rare malignant sequelae, such as lymphangiosarcoma.10 Such interventions can involve lymphangioplasty to aid in lymphatic drainage or excision of the mass and subcutaneous tissue with full-thickness skin grafting for reconstruction.7 Collaboration between urology, plastic surgery, and dermatology is often essential to obtain adequate care with satisfactory outcomes and minimal recurrence for patients with this uncommon condition.
This case and photo were submitted by Marlee Hill, a medical student at the University of Oklahoma, Oklahoma City; and Michael Franzetti, MD, and Jeffrey McBride, MD, department of dermatology, University of Oklahoma Health Sciences Center. The column was edited by Donna Bilu Martin, MD.
Dr. Donna Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hadian Y et al. Dermatol Online J. 2019 Dec 15;25(12):13030/qt6rn1s8ff.
2. Judge N and Kilic A. J Dermatol Case Rep. 2016 Nov 13;10(2):32-4.
3. Dean SM et al. J Am Acad Dermatol. 2011 Jun;64(6):1104-10.
4. Sisto K and Khachemoune A. Am J Clin Dermatol. 2008;9(3):141-6.
5. Motegi S et al. Dermatology. 2007;215(2):147-51.
6. Robinson CG et al. J Cutan Med Surg. 2018;22(6):611-3.
7. Koualla S et al. Ann Chir Plast Esthet. 2023 Apr 10;S0294-1260(23)00035-3.
8. Lelonek E et al. Acta Derm Venereol. 2021 Feb 11;101(2):adv00389.
9. Good LM et al. J Am Acad Dermatol. 2011 May;64(5):993-4.
10. Cerri A et al. Eur J Dermatol. 1998 Oct-Nov;8(7):511-4.
.1 Clinically, ENV presents as verrucous, hyperkeratotic, cobblestone-like patches, plaques, and nodules with associated nonpitting edema of the affected body area.1 Secondary bacterial infections are common and often worsen the clinical course. The etiology of ENV involves chronic lymphatic obstruction and venous insufficiency, with additional risk factors including obesity, chronic lymphedema, bacterial infection, surgery or trauma, neoplasia, radiation, congestive heart failure, or scleroderma.2,3 While most commonly presenting on the lower extremities, cases have been reported involving the abdomen, sacrum, ears, buttocks, and penoscrotal area.1,2
Regardless of location, the pathogenesis of ENV remains the same. Chronic lymphatic obstruction results in accumulation and lymphostasis of protein-rich dermal fluid, which subsequently precipitates fibroblast proliferation and activation, suppression of the local immune response and development of recurrent lymphangitis, chronic inflammation, and potential secondary bacterial infection.2,4
There is no standard of care for the treatment and management of ENV and recurrence is common. Interventions often involve those used for chronic lymphedema – including leg elevation, compression stockings or devices, skin hygiene, and lymphatic pumping.2,3 Medical management with topical and oral retinoids has been reported, as well as emphasis on weight loss and infection control.1,4 Surgical intervention is often reserved for refractory cases that fail to respond to more conservative management, or severe presentations resulting in extensive functional and aesthetic impairment. Less commonly reported treatment modalities include lymphaticovenular anastomosis and ablative carbon dioxide laser use, although this latter intervention demonstrated minimal improvement in this patient.5,6
Penoscrotal ENV is a rare form of ENV affecting the genital region of males, often resulting in significant disfigurement, functional impairment, and psychosocial distress. Penoscrotal elephantiasis can be idiopathic, due to filarial infections, scleroinflammatory stricture of the urethra, Chlamydia trachomatis infection, and lymphostasis secondary to chronic inflammatory conditions such as streptococcal infections, radiotherapy, surgery, chronic venous stasis, or Kaposi sarcoma.7
In addition, hidradenitis suppurativa (HS) has been documented multiple times in the literature in association with the development of ENV, detailing lymphatic scarring secondary to chronic inguinal HS as the main pathogenic factor.8,9
Surgery is the mainstay of treatment for penoscrotal ENV, which not only improves functionality and cosmesis, but also aids in prevention of rare malignant sequelae, such as lymphangiosarcoma.10 Such interventions can involve lymphangioplasty to aid in lymphatic drainage or excision of the mass and subcutaneous tissue with full-thickness skin grafting for reconstruction.7 Collaboration between urology, plastic surgery, and dermatology is often essential to obtain adequate care with satisfactory outcomes and minimal recurrence for patients with this uncommon condition.
This case and photo were submitted by Marlee Hill, a medical student at the University of Oklahoma, Oklahoma City; and Michael Franzetti, MD, and Jeffrey McBride, MD, department of dermatology, University of Oklahoma Health Sciences Center. The column was edited by Donna Bilu Martin, MD.
Dr. Donna Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hadian Y et al. Dermatol Online J. 2019 Dec 15;25(12):13030/qt6rn1s8ff.
2. Judge N and Kilic A. J Dermatol Case Rep. 2016 Nov 13;10(2):32-4.
3. Dean SM et al. J Am Acad Dermatol. 2011 Jun;64(6):1104-10.
4. Sisto K and Khachemoune A. Am J Clin Dermatol. 2008;9(3):141-6.
5. Motegi S et al. Dermatology. 2007;215(2):147-51.
6. Robinson CG et al. J Cutan Med Surg. 2018;22(6):611-3.
7. Koualla S et al. Ann Chir Plast Esthet. 2023 Apr 10;S0294-1260(23)00035-3.
8. Lelonek E et al. Acta Derm Venereol. 2021 Feb 11;101(2):adv00389.
9. Good LM et al. J Am Acad Dermatol. 2011 May;64(5):993-4.
10. Cerri A et al. Eur J Dermatol. 1998 Oct-Nov;8(7):511-4.
.1 Clinically, ENV presents as verrucous, hyperkeratotic, cobblestone-like patches, plaques, and nodules with associated nonpitting edema of the affected body area.1 Secondary bacterial infections are common and often worsen the clinical course. The etiology of ENV involves chronic lymphatic obstruction and venous insufficiency, with additional risk factors including obesity, chronic lymphedema, bacterial infection, surgery or trauma, neoplasia, radiation, congestive heart failure, or scleroderma.2,3 While most commonly presenting on the lower extremities, cases have been reported involving the abdomen, sacrum, ears, buttocks, and penoscrotal area.1,2
Regardless of location, the pathogenesis of ENV remains the same. Chronic lymphatic obstruction results in accumulation and lymphostasis of protein-rich dermal fluid, which subsequently precipitates fibroblast proliferation and activation, suppression of the local immune response and development of recurrent lymphangitis, chronic inflammation, and potential secondary bacterial infection.2,4
There is no standard of care for the treatment and management of ENV and recurrence is common. Interventions often involve those used for chronic lymphedema – including leg elevation, compression stockings or devices, skin hygiene, and lymphatic pumping.2,3 Medical management with topical and oral retinoids has been reported, as well as emphasis on weight loss and infection control.1,4 Surgical intervention is often reserved for refractory cases that fail to respond to more conservative management, or severe presentations resulting in extensive functional and aesthetic impairment. Less commonly reported treatment modalities include lymphaticovenular anastomosis and ablative carbon dioxide laser use, although this latter intervention demonstrated minimal improvement in this patient.5,6
Penoscrotal ENV is a rare form of ENV affecting the genital region of males, often resulting in significant disfigurement, functional impairment, and psychosocial distress. Penoscrotal elephantiasis can be idiopathic, due to filarial infections, scleroinflammatory stricture of the urethra, Chlamydia trachomatis infection, and lymphostasis secondary to chronic inflammatory conditions such as streptococcal infections, radiotherapy, surgery, chronic venous stasis, or Kaposi sarcoma.7
In addition, hidradenitis suppurativa (HS) has been documented multiple times in the literature in association with the development of ENV, detailing lymphatic scarring secondary to chronic inguinal HS as the main pathogenic factor.8,9
Surgery is the mainstay of treatment for penoscrotal ENV, which not only improves functionality and cosmesis, but also aids in prevention of rare malignant sequelae, such as lymphangiosarcoma.10 Such interventions can involve lymphangioplasty to aid in lymphatic drainage or excision of the mass and subcutaneous tissue with full-thickness skin grafting for reconstruction.7 Collaboration between urology, plastic surgery, and dermatology is often essential to obtain adequate care with satisfactory outcomes and minimal recurrence for patients with this uncommon condition.
This case and photo were submitted by Marlee Hill, a medical student at the University of Oklahoma, Oklahoma City; and Michael Franzetti, MD, and Jeffrey McBride, MD, department of dermatology, University of Oklahoma Health Sciences Center. The column was edited by Donna Bilu Martin, MD.
Dr. Donna Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
1. Hadian Y et al. Dermatol Online J. 2019 Dec 15;25(12):13030/qt6rn1s8ff.
2. Judge N and Kilic A. J Dermatol Case Rep. 2016 Nov 13;10(2):32-4.
3. Dean SM et al. J Am Acad Dermatol. 2011 Jun;64(6):1104-10.
4. Sisto K and Khachemoune A. Am J Clin Dermatol. 2008;9(3):141-6.
5. Motegi S et al. Dermatology. 2007;215(2):147-51.
6. Robinson CG et al. J Cutan Med Surg. 2018;22(6):611-3.
7. Koualla S et al. Ann Chir Plast Esthet. 2023 Apr 10;S0294-1260(23)00035-3.
8. Lelonek E et al. Acta Derm Venereol. 2021 Feb 11;101(2):adv00389.
9. Good LM et al. J Am Acad Dermatol. 2011 May;64(5):993-4.
10. Cerri A et al. Eur J Dermatol. 1998 Oct-Nov;8(7):511-4.
FDA warns of tattoo ink tied to dangerous infections
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
The Food and Drug Administration draft guidance released recently on possible contamination of tattoo ink was not concerning Whitney Donohue, 34, owner of Forget Me Not Tattoo in Billings, Mont.
“I get our ink directly through the manufacturer – not at a store or through Amazon or eBay,” she said. “You never know if it’s going to be repackaged.”
Tattoo artists themselves, she said, regulate the quality of ink they use.
Still, the threat is real, said Bruce Brod, MD, a clinical professor of dermatology at the University of Pennsylvania Health System. “I’ve seen several different infections from tattooing, and they are from organisms that tend to contaminate things in damp, liquid-type environments.”
, dermatologists said.
“Tattooing involves puncturing the epidermis about 100 times per second with needles and depositing ink 1.5 to 2 millimeters below the surface of the skin, deep into the dermis,” the guidance states. “Contaminated tattoo ink can cause infections and serious injuries. Because these inks are injected, pathogens or other harmful substances in these inks can travel from the injection site through the blood and lymphatic systems to other parts of the body.”
The guidance comes as body art continues to get more popular. According to a 2019 poll, 30% of Americans had at least one tattoo – up from 21% in 2012. Forty percent of people 18-34 and 36% of those ages 35-54 had at least one tattoo. And though they are commonplace, tattoos come with medical risks that should be known beforehand, doctors said.
Commonly reported symptoms of tattoo ink–associated infections include rashes, blisters, painful nodules, and severe abscesses. One of the most common bacteria found in contaminated tattoo ink is nontuberculous mycobacteria, which is related to the bacteria that causes tuberculosis and can be found in soil and water.
The guidance lists several unsanitary manufacturing conditions that may lead to ink contamination, including:
- Preparing or packing of tattoo inks in facilities that are hard to sanitize, such as carpeted areas
- Ink or ink components left uncovered, especially near open air ducts
- Unsanitary mixing of tattoo inks, including with unclean utensils or containers
- Lack of appropriate attire by staff, failure to use hairnets, lab coats, aprons, gowns, masks, or gloves
“Infections will often spread along the drainage channels in the skin and create squiggly, uneven lines of big red, lumpy nodules,” Dr. Brod said.
Between 2003 and 2023, there were 18 recalls of tattoo inks that were contaminated with various microorganisms, according to the FDA. In May 2019, the FDA issued a safety alert advising consumers, tattoo artists, and retailers to avoid using or selling certain tattoo inks contaminated with microorganisms.
Reputable ink manufacturers use a process called gamma radiation, which refers to electromagnetic radiation of high frequencies to kill microorganisms in the ink and its packaging.
Most of the trustworthy, high-quality ink manufacturers are well-known among tattoo artists, Ms. Donohue said.
While she has seen customers with sensitive skin have allergic reactions, she has not seen someone come back with an infection in her 9 years working in the tattoo industry.
Because tattoo ink is considered a cosmetic product, there is not much regulatory oversight involved, which means the sterility and quality of ingredients vary, said Teo Soleymani, MD, an assistant clinical professor of dermatology and dermatological surgery at the UCLA David Geffen School of Medicine.
“Cosmeceuticals aren’t regulated by the FDA like prescription medication,” he said. “What we’ve seen many times is inadvertent contamination during the application process or contamination while the inks are being made.”
In years past, unclean needles spreading hepatitis and HIV were more of a concern, but those rates have dropped significantly, Dr. Soleymani said.
The infections that have increased are from rare bacteria that exist in stagnant water. And they are injected into a part of the body that allows them to evade the immune system, he said: shallow enough that there aren’t many associated blood vessels, but not still below the layer of skin that gets sloughed off every 28 days.
Sometimes, antibiotics alone won’t cut it, and the tattoo will require surgical removal.
“The aesthetic you were going for has to be not only removed, but you’re left with a surgical scar,” Dr. Soleymani said. “Tattoos can be beautiful, but they can come with unwanted visitors that can cause months of misery.”
A version of this article first appeared on WebMD.com.
After Yusimry’s steep discount, little clarity on future adalimumab biosimilar pricing
Adalimumab, sold under the brand name Humira, enjoyed a long run as one of the world’s best-selling medicines. But its 20-year, competition-free period has ended, and despite its best efforts to delay their arrival, drug manufacturer AbbVie now faces increasing competition from biosimilars entering the marketplace.
But one biosimilar about to be launched may be something of a game changer. Coherus BioSciences has announced plans to market its biosimilar Yusimry (adalimumab-aqvh) at a cost of $995 for two autoinjectors. This represents an approximate 85% discount over Humira’s sale list price of $6922.
This price, however, is slated to plunge even further as Coherus has also revealed that it will work with the Mark Cuban Cost Plus Drug Company (MCCPDC) to offer an even lower price. When Yusimry launches in July, it will sell for about $579 for two autoinjectors, making it the lowest-priced adalimumab biosimilar on the market.
“Coherus and Cost Plus Drug Company share a common mission, to increase access to high-quality medicine for patients at an affordable price,” said Dennis Lanfear, MBA, president, CEO and chairman of Coherus. “Mark Cuban and his team offer innovative solutions to health care problems, and Coherus is also a highly innovative company focused on unmet patient needs.”
He noted that, with adalimumab biosimilar pricing, this translates to a low list price approach. “We are pleased that Yusimry will be a part of that, as the first biologic they carry,” Mr. Lanfear said.
MCCPDC prices are based on the cost of ingredients and manufacturing plus 15% margin, a $3 pharmacy dispensing fee, and a $5 shipping fee. The company has expanded its inventory from 100 generics to more than 350 medications since it launched in January 2022. While MCCPDC is primarily directed to people who are paying cash for drugs, it does take insurance from select plans. And even for people who are covered by other insurers, the cost of drugs from Mr. Cuban’s company may be less than their out-of-pocket costs if they did go through their payer.
The low pricing of Yusimry is welcome, said Marcus Snow, MD, an assistant professor in the division of rheumatology at the University of Nebraska Medical Center, Omaha, but he pointed out that it is still a very expensive drug. “For patients who can’t afford Humira due to poor insurance coverage and high out-of-pocket costs, it is a welcome option. But it’s also unclear how many patients who lack adequate health insurance coverage can afford to pay $579 a month out of their own pockets.”
The biosimilars are coming
By early December 2022, the Food and Drug Administration had approved seven Humira biosimilars, and Amgen launched the first biosimilar to come on the market, Amjevita, soon afterward. By July 2023, half a dozen more are expected to enter the marketplace, said Steven Horvitz, managing director of EMC Analytics Group, a pharmaceutical research firm.
Mr. Horvitz agrees that the system is out of control, but it is unclear how much of an effect the low price tag on the Coherus product will have. “Some insurers may say, ‘we want the lowest price, and we don’t care about rebates,’ and will go with it,” he said. “PBMs [pharmacy benefit managers] are all about economics, so we have to see how many of their major clients will ask for the lowest price.”
Amgen has more or less followed the status quo on pricing for its biosimilar, but with a twist. It›s being offered at two different prices: $85,494 a year, which is only a 5% discount from Humira’s list price, or at $40,497 a year, a 55% discount. However, to date, the lower price has generally not been granted favorable formulary placement by PBMs. The plans that adopt the higher-priced biosimilar will get bigger rebates, but patients with coinsurance and deductibles will pay more out of pocket.
It is yet unknown how the pricing on Yusimry will affect the biosimilars ready to launch. “Will it give them pause for thought or not make any difference?” Mr. Horvitz said. “The companies do not reveal their pricing before the fact, so we have to wait and see.”
Large PBMs have not jumped at the opportunity to offer the Coherus biosimilar, but SmithRx, which bills itself as “next-generation pharmacy benefits management,” announced that it will offer Yusimry to its members at a discount of more than 90%.
“Unlike traditional PBMs, SmithRx prioritizes transparency and up-front cost savings. Humira is often an employer’s top drug expense so offering a low-cost alternative will have significant impact,” Jake Frenz, CEO and founder of SmithRx, said in a statement. “We’re excited to work with Cost Plus Drugs to bring this biosimilar to our members – and significantly reduce costs for them and their employers.”
A version of this article first appeared on Medscape.com.
Adalimumab, sold under the brand name Humira, enjoyed a long run as one of the world’s best-selling medicines. But its 20-year, competition-free period has ended, and despite its best efforts to delay their arrival, drug manufacturer AbbVie now faces increasing competition from biosimilars entering the marketplace.
But one biosimilar about to be launched may be something of a game changer. Coherus BioSciences has announced plans to market its biosimilar Yusimry (adalimumab-aqvh) at a cost of $995 for two autoinjectors. This represents an approximate 85% discount over Humira’s sale list price of $6922.
This price, however, is slated to plunge even further as Coherus has also revealed that it will work with the Mark Cuban Cost Plus Drug Company (MCCPDC) to offer an even lower price. When Yusimry launches in July, it will sell for about $579 for two autoinjectors, making it the lowest-priced adalimumab biosimilar on the market.
“Coherus and Cost Plus Drug Company share a common mission, to increase access to high-quality medicine for patients at an affordable price,” said Dennis Lanfear, MBA, president, CEO and chairman of Coherus. “Mark Cuban and his team offer innovative solutions to health care problems, and Coherus is also a highly innovative company focused on unmet patient needs.”
He noted that, with adalimumab biosimilar pricing, this translates to a low list price approach. “We are pleased that Yusimry will be a part of that, as the first biologic they carry,” Mr. Lanfear said.
MCCPDC prices are based on the cost of ingredients and manufacturing plus 15% margin, a $3 pharmacy dispensing fee, and a $5 shipping fee. The company has expanded its inventory from 100 generics to more than 350 medications since it launched in January 2022. While MCCPDC is primarily directed to people who are paying cash for drugs, it does take insurance from select plans. And even for people who are covered by other insurers, the cost of drugs from Mr. Cuban’s company may be less than their out-of-pocket costs if they did go through their payer.
The low pricing of Yusimry is welcome, said Marcus Snow, MD, an assistant professor in the division of rheumatology at the University of Nebraska Medical Center, Omaha, but he pointed out that it is still a very expensive drug. “For patients who can’t afford Humira due to poor insurance coverage and high out-of-pocket costs, it is a welcome option. But it’s also unclear how many patients who lack adequate health insurance coverage can afford to pay $579 a month out of their own pockets.”
The biosimilars are coming
By early December 2022, the Food and Drug Administration had approved seven Humira biosimilars, and Amgen launched the first biosimilar to come on the market, Amjevita, soon afterward. By July 2023, half a dozen more are expected to enter the marketplace, said Steven Horvitz, managing director of EMC Analytics Group, a pharmaceutical research firm.
Mr. Horvitz agrees that the system is out of control, but it is unclear how much of an effect the low price tag on the Coherus product will have. “Some insurers may say, ‘we want the lowest price, and we don’t care about rebates,’ and will go with it,” he said. “PBMs [pharmacy benefit managers] are all about economics, so we have to see how many of their major clients will ask for the lowest price.”
Amgen has more or less followed the status quo on pricing for its biosimilar, but with a twist. It›s being offered at two different prices: $85,494 a year, which is only a 5% discount from Humira’s list price, or at $40,497 a year, a 55% discount. However, to date, the lower price has generally not been granted favorable formulary placement by PBMs. The plans that adopt the higher-priced biosimilar will get bigger rebates, but patients with coinsurance and deductibles will pay more out of pocket.
It is yet unknown how the pricing on Yusimry will affect the biosimilars ready to launch. “Will it give them pause for thought or not make any difference?” Mr. Horvitz said. “The companies do not reveal their pricing before the fact, so we have to wait and see.”
Large PBMs have not jumped at the opportunity to offer the Coherus biosimilar, but SmithRx, which bills itself as “next-generation pharmacy benefits management,” announced that it will offer Yusimry to its members at a discount of more than 90%.
“Unlike traditional PBMs, SmithRx prioritizes transparency and up-front cost savings. Humira is often an employer’s top drug expense so offering a low-cost alternative will have significant impact,” Jake Frenz, CEO and founder of SmithRx, said in a statement. “We’re excited to work with Cost Plus Drugs to bring this biosimilar to our members – and significantly reduce costs for them and their employers.”
A version of this article first appeared on Medscape.com.
Adalimumab, sold under the brand name Humira, enjoyed a long run as one of the world’s best-selling medicines. But its 20-year, competition-free period has ended, and despite its best efforts to delay their arrival, drug manufacturer AbbVie now faces increasing competition from biosimilars entering the marketplace.
But one biosimilar about to be launched may be something of a game changer. Coherus BioSciences has announced plans to market its biosimilar Yusimry (adalimumab-aqvh) at a cost of $995 for two autoinjectors. This represents an approximate 85% discount over Humira’s sale list price of $6922.
This price, however, is slated to plunge even further as Coherus has also revealed that it will work with the Mark Cuban Cost Plus Drug Company (MCCPDC) to offer an even lower price. When Yusimry launches in July, it will sell for about $579 for two autoinjectors, making it the lowest-priced adalimumab biosimilar on the market.
“Coherus and Cost Plus Drug Company share a common mission, to increase access to high-quality medicine for patients at an affordable price,” said Dennis Lanfear, MBA, president, CEO and chairman of Coherus. “Mark Cuban and his team offer innovative solutions to health care problems, and Coherus is also a highly innovative company focused on unmet patient needs.”
He noted that, with adalimumab biosimilar pricing, this translates to a low list price approach. “We are pleased that Yusimry will be a part of that, as the first biologic they carry,” Mr. Lanfear said.
MCCPDC prices are based on the cost of ingredients and manufacturing plus 15% margin, a $3 pharmacy dispensing fee, and a $5 shipping fee. The company has expanded its inventory from 100 generics to more than 350 medications since it launched in January 2022. While MCCPDC is primarily directed to people who are paying cash for drugs, it does take insurance from select plans. And even for people who are covered by other insurers, the cost of drugs from Mr. Cuban’s company may be less than their out-of-pocket costs if they did go through their payer.
The low pricing of Yusimry is welcome, said Marcus Snow, MD, an assistant professor in the division of rheumatology at the University of Nebraska Medical Center, Omaha, but he pointed out that it is still a very expensive drug. “For patients who can’t afford Humira due to poor insurance coverage and high out-of-pocket costs, it is a welcome option. But it’s also unclear how many patients who lack adequate health insurance coverage can afford to pay $579 a month out of their own pockets.”
The biosimilars are coming
By early December 2022, the Food and Drug Administration had approved seven Humira biosimilars, and Amgen launched the first biosimilar to come on the market, Amjevita, soon afterward. By July 2023, half a dozen more are expected to enter the marketplace, said Steven Horvitz, managing director of EMC Analytics Group, a pharmaceutical research firm.
Mr. Horvitz agrees that the system is out of control, but it is unclear how much of an effect the low price tag on the Coherus product will have. “Some insurers may say, ‘we want the lowest price, and we don’t care about rebates,’ and will go with it,” he said. “PBMs [pharmacy benefit managers] are all about economics, so we have to see how many of their major clients will ask for the lowest price.”
Amgen has more or less followed the status quo on pricing for its biosimilar, but with a twist. It›s being offered at two different prices: $85,494 a year, which is only a 5% discount from Humira’s list price, or at $40,497 a year, a 55% discount. However, to date, the lower price has generally not been granted favorable formulary placement by PBMs. The plans that adopt the higher-priced biosimilar will get bigger rebates, but patients with coinsurance and deductibles will pay more out of pocket.
It is yet unknown how the pricing on Yusimry will affect the biosimilars ready to launch. “Will it give them pause for thought or not make any difference?” Mr. Horvitz said. “The companies do not reveal their pricing before the fact, so we have to wait and see.”
Large PBMs have not jumped at the opportunity to offer the Coherus biosimilar, but SmithRx, which bills itself as “next-generation pharmacy benefits management,” announced that it will offer Yusimry to its members at a discount of more than 90%.
“Unlike traditional PBMs, SmithRx prioritizes transparency and up-front cost savings. Humira is often an employer’s top drug expense so offering a low-cost alternative will have significant impact,” Jake Frenz, CEO and founder of SmithRx, said in a statement. “We’re excited to work with Cost Plus Drugs to bring this biosimilar to our members – and significantly reduce costs for them and their employers.”
A version of this article first appeared on Medscape.com.
Experts share their sun protection tips for children
“I basically say, ‘sun protection means clothing, shade, [considering the] time of day of exposure, and sunscreen if you are going to be otherwise exposed,’ ” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego, said during a panel discussion about sunscreen use at the Hawaii Dermatology Seminar provided by MedscapeLIVE! He recommends photoprotective gear such as rash guards for surfers and other water sport enthusiasts. When patients ask him if they should use sunscreen, he often replies with a question of his own.
“Do you brush your teeth?” he’ll ask.
“Yes, I do.”
“Well, you should put sunscreen on every day.”
Another panelist, Adelaide A. Hebert, MD, professor of dermatology and pediatrics and chief of pediatric dermatology at the University of Texas, Houston, said that she advises new parents to start sun protection efforts early. “Most sunscreens are not approved for use in children under the age of 6 months because testing has not been done in this age group, but I do recommend protective clothing. I also recommend wrap-around sunglasses, which offer 5% more protection from the sun than regular sunglasses.”
In her opinion, stick sunscreens are “a good add-on,” especially for under the eyes and the backs of the hands, but she is not a fan of spray sunscreens, which can leave large areas of skin unprotected if not applied properly.
Fellow panelist Jennifer Huang, MD, a pediatric dermatologist at Boston Children’s Hospital, who has a special interest in taking care of dermatologic conditions of children with cancer, generally recommends mineral-based sunscreens. “There is data to suggest that nonmineral sunscreens are less safe than mineral sunscreens for humans, and mineral sunscreens are considered to be better for the environment,” Dr. Huang said. “Plus, there are more elegant versions of mineral sunscreens that don’t make your skin pasty white.” However, for patients with darker skin tones, “it can be hard to apply a pasty white sunscreen, so I lean on some recommendations for tinted sunscreens, too, so there are options. I specifically recommend sunscreens that have iron oxides in them so that it can block physical rays and help with the cosmetic appearance.”
Moise Levy, MD, professor of internal medicine and pediatrics at the University of Texas at Austin, said that his approach to imparting sunscreen advice to children and their parents involves a mix of spoken information, printed information, and sunscreen samples for children to try in the office, in the presence of a parent. To help patients choose among different samples, be they ointments, gels, or lotions, he will often ask the child: “‘What do you like the feel of better?’ If the child says, ‘I like this one,’ I make sure the parent hears that,” Dr. Levy said.
Next, Dr. Eichenfield, who moderated the discussion, asked his fellow panelists how they would counsel someone who comes to their practice for evaluation of moles and has a family history of nonmelanoma skin cancer. “I think this is one of the easier counseling sessions, because there are enough kids who are asked about the moles on their skin when they’re at school,” Dr. Hebert said. “I think they’re very ready to wear sun protective clothing and I certainly don’t want any sun exposure that would pose an increased risk for their child.”
In addition to routine sun protection, Dr. Huang recommends annual mole checks for children who have a first-degree relative with a history of malignant melanoma. Other high-risk groups that should undergo annual skin exams include anyone who has received high doses of radiation, bone marrow transplants, prolonged use of voriconazole, or prolonged systemic immunosuppression. Without a known genetic predisposition syndrome, a family history of nonmelanoma skin cancer would not raise concern for melanoma in an otherwise healthy child.
Dr. Eichenfield added that freckling used to be the secondary risk factor for melanoma, “but it’s flipped over to a primary risk factor. A history of immunosuppression or prior cancer is a major risk factor in childhood and teenage years.”
Dr. Eichenfield disclosed that he is a consultant or adviser for numerous pharmaceutical companies. He has also received research funding from AbbVie, Bausch & Lomb, Galderma Laboratories, and Pfizer. Dr. Hebert disclosed that she is a consultant or adviser for AbbVie, Almirall, Amryt Pharma, Arcutis Biotherapeutics, Beiersdorf, Dermavant Sciences, Galderma Laboratories, L’Oreal, Novan, Ortho Dermatologics, Pfizer, and Verrica. Dr. Levy disclosed that he is consultant or adviser for Abeona, Castle Creek, Dusa Pharma, Krystal Bio, Novan, Regeneron, and Sanofi Genzyme. Dr. Huang disclosed that she is an adviser for EllaOla.
MedscapeLive! and this news organization are owned by the same parent company.
“I basically say, ‘sun protection means clothing, shade, [considering the] time of day of exposure, and sunscreen if you are going to be otherwise exposed,’ ” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego, said during a panel discussion about sunscreen use at the Hawaii Dermatology Seminar provided by MedscapeLIVE! He recommends photoprotective gear such as rash guards for surfers and other water sport enthusiasts. When patients ask him if they should use sunscreen, he often replies with a question of his own.
“Do you brush your teeth?” he’ll ask.
“Yes, I do.”
“Well, you should put sunscreen on every day.”
Another panelist, Adelaide A. Hebert, MD, professor of dermatology and pediatrics and chief of pediatric dermatology at the University of Texas, Houston, said that she advises new parents to start sun protection efforts early. “Most sunscreens are not approved for use in children under the age of 6 months because testing has not been done in this age group, but I do recommend protective clothing. I also recommend wrap-around sunglasses, which offer 5% more protection from the sun than regular sunglasses.”
In her opinion, stick sunscreens are “a good add-on,” especially for under the eyes and the backs of the hands, but she is not a fan of spray sunscreens, which can leave large areas of skin unprotected if not applied properly.
Fellow panelist Jennifer Huang, MD, a pediatric dermatologist at Boston Children’s Hospital, who has a special interest in taking care of dermatologic conditions of children with cancer, generally recommends mineral-based sunscreens. “There is data to suggest that nonmineral sunscreens are less safe than mineral sunscreens for humans, and mineral sunscreens are considered to be better for the environment,” Dr. Huang said. “Plus, there are more elegant versions of mineral sunscreens that don’t make your skin pasty white.” However, for patients with darker skin tones, “it can be hard to apply a pasty white sunscreen, so I lean on some recommendations for tinted sunscreens, too, so there are options. I specifically recommend sunscreens that have iron oxides in them so that it can block physical rays and help with the cosmetic appearance.”
Moise Levy, MD, professor of internal medicine and pediatrics at the University of Texas at Austin, said that his approach to imparting sunscreen advice to children and their parents involves a mix of spoken information, printed information, and sunscreen samples for children to try in the office, in the presence of a parent. To help patients choose among different samples, be they ointments, gels, or lotions, he will often ask the child: “‘What do you like the feel of better?’ If the child says, ‘I like this one,’ I make sure the parent hears that,” Dr. Levy said.
Next, Dr. Eichenfield, who moderated the discussion, asked his fellow panelists how they would counsel someone who comes to their practice for evaluation of moles and has a family history of nonmelanoma skin cancer. “I think this is one of the easier counseling sessions, because there are enough kids who are asked about the moles on their skin when they’re at school,” Dr. Hebert said. “I think they’re very ready to wear sun protective clothing and I certainly don’t want any sun exposure that would pose an increased risk for their child.”
In addition to routine sun protection, Dr. Huang recommends annual mole checks for children who have a first-degree relative with a history of malignant melanoma. Other high-risk groups that should undergo annual skin exams include anyone who has received high doses of radiation, bone marrow transplants, prolonged use of voriconazole, or prolonged systemic immunosuppression. Without a known genetic predisposition syndrome, a family history of nonmelanoma skin cancer would not raise concern for melanoma in an otherwise healthy child.
Dr. Eichenfield added that freckling used to be the secondary risk factor for melanoma, “but it’s flipped over to a primary risk factor. A history of immunosuppression or prior cancer is a major risk factor in childhood and teenage years.”
Dr. Eichenfield disclosed that he is a consultant or adviser for numerous pharmaceutical companies. He has also received research funding from AbbVie, Bausch & Lomb, Galderma Laboratories, and Pfizer. Dr. Hebert disclosed that she is a consultant or adviser for AbbVie, Almirall, Amryt Pharma, Arcutis Biotherapeutics, Beiersdorf, Dermavant Sciences, Galderma Laboratories, L’Oreal, Novan, Ortho Dermatologics, Pfizer, and Verrica. Dr. Levy disclosed that he is consultant or adviser for Abeona, Castle Creek, Dusa Pharma, Krystal Bio, Novan, Regeneron, and Sanofi Genzyme. Dr. Huang disclosed that she is an adviser for EllaOla.
MedscapeLive! and this news organization are owned by the same parent company.
“I basically say, ‘sun protection means clothing, shade, [considering the] time of day of exposure, and sunscreen if you are going to be otherwise exposed,’ ” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady’s Children’s Hospital, San Diego, said during a panel discussion about sunscreen use at the Hawaii Dermatology Seminar provided by MedscapeLIVE! He recommends photoprotective gear such as rash guards for surfers and other water sport enthusiasts. When patients ask him if they should use sunscreen, he often replies with a question of his own.
“Do you brush your teeth?” he’ll ask.
“Yes, I do.”
“Well, you should put sunscreen on every day.”
Another panelist, Adelaide A. Hebert, MD, professor of dermatology and pediatrics and chief of pediatric dermatology at the University of Texas, Houston, said that she advises new parents to start sun protection efforts early. “Most sunscreens are not approved for use in children under the age of 6 months because testing has not been done in this age group, but I do recommend protective clothing. I also recommend wrap-around sunglasses, which offer 5% more protection from the sun than regular sunglasses.”
In her opinion, stick sunscreens are “a good add-on,” especially for under the eyes and the backs of the hands, but she is not a fan of spray sunscreens, which can leave large areas of skin unprotected if not applied properly.
Fellow panelist Jennifer Huang, MD, a pediatric dermatologist at Boston Children’s Hospital, who has a special interest in taking care of dermatologic conditions of children with cancer, generally recommends mineral-based sunscreens. “There is data to suggest that nonmineral sunscreens are less safe than mineral sunscreens for humans, and mineral sunscreens are considered to be better for the environment,” Dr. Huang said. “Plus, there are more elegant versions of mineral sunscreens that don’t make your skin pasty white.” However, for patients with darker skin tones, “it can be hard to apply a pasty white sunscreen, so I lean on some recommendations for tinted sunscreens, too, so there are options. I specifically recommend sunscreens that have iron oxides in them so that it can block physical rays and help with the cosmetic appearance.”
Moise Levy, MD, professor of internal medicine and pediatrics at the University of Texas at Austin, said that his approach to imparting sunscreen advice to children and their parents involves a mix of spoken information, printed information, and sunscreen samples for children to try in the office, in the presence of a parent. To help patients choose among different samples, be they ointments, gels, or lotions, he will often ask the child: “‘What do you like the feel of better?’ If the child says, ‘I like this one,’ I make sure the parent hears that,” Dr. Levy said.
Next, Dr. Eichenfield, who moderated the discussion, asked his fellow panelists how they would counsel someone who comes to their practice for evaluation of moles and has a family history of nonmelanoma skin cancer. “I think this is one of the easier counseling sessions, because there are enough kids who are asked about the moles on their skin when they’re at school,” Dr. Hebert said. “I think they’re very ready to wear sun protective clothing and I certainly don’t want any sun exposure that would pose an increased risk for their child.”
In addition to routine sun protection, Dr. Huang recommends annual mole checks for children who have a first-degree relative with a history of malignant melanoma. Other high-risk groups that should undergo annual skin exams include anyone who has received high doses of radiation, bone marrow transplants, prolonged use of voriconazole, or prolonged systemic immunosuppression. Without a known genetic predisposition syndrome, a family history of nonmelanoma skin cancer would not raise concern for melanoma in an otherwise healthy child.
Dr. Eichenfield added that freckling used to be the secondary risk factor for melanoma, “but it’s flipped over to a primary risk factor. A history of immunosuppression or prior cancer is a major risk factor in childhood and teenage years.”
Dr. Eichenfield disclosed that he is a consultant or adviser for numerous pharmaceutical companies. He has also received research funding from AbbVie, Bausch & Lomb, Galderma Laboratories, and Pfizer. Dr. Hebert disclosed that she is a consultant or adviser for AbbVie, Almirall, Amryt Pharma, Arcutis Biotherapeutics, Beiersdorf, Dermavant Sciences, Galderma Laboratories, L’Oreal, Novan, Ortho Dermatologics, Pfizer, and Verrica. Dr. Levy disclosed that he is consultant or adviser for Abeona, Castle Creek, Dusa Pharma, Krystal Bio, Novan, Regeneron, and Sanofi Genzyme. Dr. Huang disclosed that she is an adviser for EllaOla.
MedscapeLive! and this news organization are owned by the same parent company.
FROM THE MEDSCAPELIVE! HAWAII DERMATOLOGY SEMINAR