User login
What to Know About Sexually Transmitted Ringworm
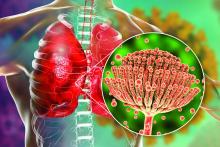
Ringworm (also known as tinea, jock itch, or athlete’s foot) is a common infection caused by dermatophyte fungi, known to affect skin, hair, or nails. It causes skin infections that are typically mild and are often treated with topical antifungals.
However, in recent years, newly emerging dermatophyte strains have been causing more severe and harder-to-treat ringworm. Notably, one emerging strain, Trichophyton mentagrophytes genotype VII(TMVII), is associated with sexual contact. In recent years, TMVII infections linked to sexual contact have been reported among men who have sex with men in Europe and in travelers returning from Southeast Asia. The first US case of TMVII was reported in June 2024, after which public health authorities were alerted to additional cases; all were associated with recent sexual contact. Other dermatophyte species have also been reported to cause ringworm transmitted through sexual contact.
Here are some key points to know about sexually transmitted ringworm.
Tell me more about sexually transmitted ringworm: What is causing it?
Skin-to-skin contact is a common mode of ringworm transmission. Infections with sexually transmitted TMVII commonly cause lesions on anatomical sites that may be exposed during intimate or sexual contact, such as the face, genitals, and perianal region. Sexual transmission of TMVII has been reported in Europe, predominantly among men who have sex with men, for several years. Other dermatophyte strains have been reported in association with sexual contact, including the emerging strain Trichophyton indotineae. However, sexual transmission is not the main mode of transmission for T indotineae and other dermatophyte strains.
When should clinicians suspect a potential case of sexually transmitted ringworm?
Providers should consider sexually transmitted ringworm when seeing ringworm in locations associated with intimate contact (for example, a rash on or around the genitals, perianal area, or mouth).
The typical appearance of ringworm is a raised, ring-like, erythematous rash with a scaly border that grows over time. The rash may appear pink, brown, or gray on different types of skin. Patients may note itching and flaking of the rash. In areas with hair such as the beard area, ringworm can present as pustules and be associated with hair loss.
Emerging ringworm infections can present in atypical or more severe ways, including a highly inflammatory (painful, scarring, or otherwise severe) rash, a rash affecting a large area or multiple sites, nodules, and pustules.
Sexually transmitted ringworm may be considered based on sexual history and recent sexual contact with someone with known TMVII. Recent history of travel to a region with reported sexually transmitted ringworm may increase suspicion of TMVII. In patients with a travel history to South Asia, T indotineae should be considered, especially if the rash does not improve with oral terbinafine.
How can testing help guide the diagnosis of sexually transmitted ringworm infection?
When evaluating a rash that may represent ringworm, providers should use a confirmatory test such as potassium hydroxide (KOH) preparation when possible. KOH prep can confirm the presence of a fungus that causes ringworm, but it does not identify the species or type of ringworm. Testing such as fungal culture and molecular testing can help identify specific types of ringworm, but these tests are not often performed and may take a long time to yield results.
Routine fungal cultures cannot identify TMVII and T indotineae; these tests may identify the genus Trichophyton, but only advanced molecular testing, which is available at selected US laboratories, can identify TMVII and T indotineae.
We recommend confirmatory testing because ringworm can easily be misdiagnosed as skin conditions such as psoriasis or eczema. The use of topical steroids can worsen a ringworm infection, so clinicians should be cautious about treating a rash with topical steroids if the etiology is unclear. Treatment should not be delayed if testing is not available.
Clinicians who suspect a case of TMVII infection or infection with another emerging type of severe or antifungal-resistant ringworm can contact the Centers for Disease Control and Prevention (CDC) at fungaloutbreaks@cdc.gov. More details on how clinicians can pursue testing to identify emerging strains of ringworm can be found on the American Academy of Dermatology (AAD) emerging diseases task force website.
How should clinicians treat and manage sexually transmitted ringworm?
If TMVII infection is suspected, providers can consider starting empirical treatment with oral terbinafine. Although data are limited, experience from case series suggests that TMVII may require oral antifungal treatment because it can cause severe skin infections and often does not improve with topical antifungals. Clinicians should advise patients that they may need prolonged treatment courses until the rash resolves, with possible need for treatment courses of 6-8 weeks or longer.
Any diagnosis of a sexually transmitted infection is an opportunity to engage patients in comprehensive sexual health services. Patients with suspected sexually transmitted ringworm should be evaluated for HIV and other sexually transmitted infections, including syphilis, chlamydia, and gonorrhea; clinicians should discuss and facilitate access to other preventive services, such as HIV pre-exposure prophylaxis if the patient is HIV negative and at risk for HIV. Patients should also notify their partner(s) about the diagnosis.
Is sexually transmitted ringworm a public health concern?
It is important to know that very few cases of TMVII have been reported in the United States thus far. CDC continues to monitor emerging dermatophyte strains because these types of ringworm can cause more severe or difficult-to-treat infections. Clinicians should be aware of the potential severity of sexually transmitted ringworm infections and of how diagnosis and treatment of these infections may differ from typical management of ringworm.
So far, TMVII, the dermatophyte strain most associated with spread through sexual contact, has not been documented to have antifungal resistance. More rarely, sexually transmitted ringworm may be caused by other emerging dermatophyte strains that are antifungal resistant, such as T indotineae. Itraconazole is the recommended first-line treatment for T indotineae infections.
How can clinicians counsel patients with sexually transmitted ringworm?
Ringworm can spread with skin-to-skin contact, so patients should avoid such contact with others while they have a rash. They should also avoid sharing personal items (such as razors or towels) and clothing, and launder their clothing, towels, and bedding in a high heat cycle.
People can reduce their risk of getting all types of ringworm infection by keeping their skin clean and dry, changing their socks and underwear daily, and wearing sandals in public locker rooms and other public spaces. People should avoid skin-to-skin contact with anyone with ringworm or an unexplained rash. Before having sex, people can check in with their partners and be aware of unexplained rashes on their partners’ bodies.
Where can clinicians go to learn more about sexually transmitted and other emerging types of ringworm?
CDC has partnered with the AAD to create set of online resources for clinicians for diagnosing and managing emerging dermatophyte infections. Clinicians who suspect or confirm antimicrobial resistant ringworm infection are also encouraged to submit cases to the AAD’s Emerging Diseases Registry. Clinicians wanting further guidance on how to manage suspected or confirmed ringworm infection with an emerging dermatophyte strain can also contact the CDC at fungaloutbreaks@cdc.gov. Useful information on emerging dermatophyte infections for providers and patients is also available on CDC’s website.
Relevant Reading
Zucker J et al. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.Spivack S et al. Emerg Infect Dis. 2024;30:807-809.Jabet A et al. Emerg Infect Dis. 2023;29:1411-1414.
A version of this article appeared on Medscape.com.
Dr Anand is Epidemic Intelligence Service Officer, Division of STD Prevention, Centers for Disease Control and Prevention, Atlanta, Georgia. Dr Gold is Medical Officer, Mycotic Diseases Branch, Centers for Disease Control and Prevention. Dr Quilter is Medical Officer, Division of STD Prevention, Centers for Disease Control and Prevention. None reported any relevant conflicts of interest.
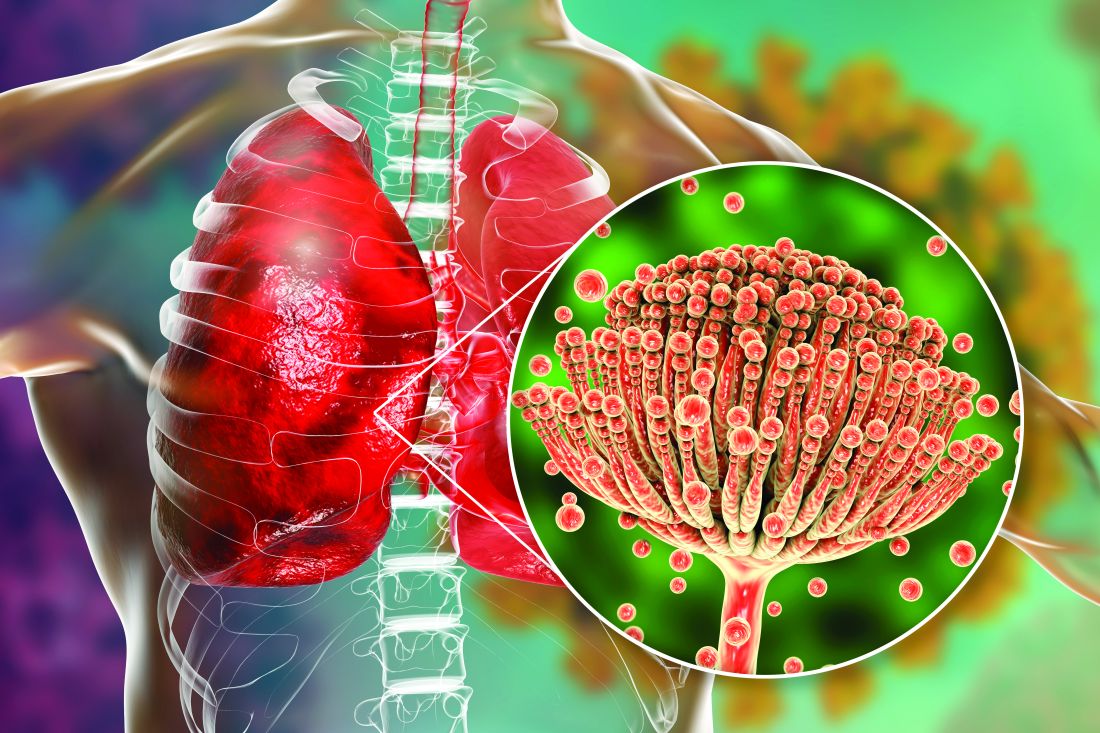
Ringworm (also known as tinea, jock itch, or athlete’s foot) is a common infection caused by dermatophyte fungi, known to affect skin, hair, or nails. It causes skin infections that are typically mild and are often treated with topical antifungals.
However, in recent years, newly emerging dermatophyte strains have been causing more severe and harder-to-treat ringworm. Notably, one emerging strain, Trichophyton mentagrophytes genotype VII(TMVII), is associated with sexual contact. In recent years, TMVII infections linked to sexual contact have been reported among men who have sex with men in Europe and in travelers returning from Southeast Asia. The first US case of TMVII was reported in June 2024, after which public health authorities were alerted to additional cases; all were associated with recent sexual contact. Other dermatophyte species have also been reported to cause ringworm transmitted through sexual contact.
Here are some key points to know about sexually transmitted ringworm.
Tell me more about sexually transmitted ringworm: What is causing it?
Skin-to-skin contact is a common mode of ringworm transmission. Infections with sexually transmitted TMVII commonly cause lesions on anatomical sites that may be exposed during intimate or sexual contact, such as the face, genitals, and perianal region. Sexual transmission of TMVII has been reported in Europe, predominantly among men who have sex with men, for several years. Other dermatophyte strains have been reported in association with sexual contact, including the emerging strain Trichophyton indotineae. However, sexual transmission is not the main mode of transmission for T indotineae and other dermatophyte strains.
When should clinicians suspect a potential case of sexually transmitted ringworm?
Providers should consider sexually transmitted ringworm when seeing ringworm in locations associated with intimate contact (for example, a rash on or around the genitals, perianal area, or mouth).
The typical appearance of ringworm is a raised, ring-like, erythematous rash with a scaly border that grows over time. The rash may appear pink, brown, or gray on different types of skin. Patients may note itching and flaking of the rash. In areas with hair such as the beard area, ringworm can present as pustules and be associated with hair loss.
Emerging ringworm infections can present in atypical or more severe ways, including a highly inflammatory (painful, scarring, or otherwise severe) rash, a rash affecting a large area or multiple sites, nodules, and pustules.
Sexually transmitted ringworm may be considered based on sexual history and recent sexual contact with someone with known TMVII. Recent history of travel to a region with reported sexually transmitted ringworm may increase suspicion of TMVII. In patients with a travel history to South Asia, T indotineae should be considered, especially if the rash does not improve with oral terbinafine.
How can testing help guide the diagnosis of sexually transmitted ringworm infection?
When evaluating a rash that may represent ringworm, providers should use a confirmatory test such as potassium hydroxide (KOH) preparation when possible. KOH prep can confirm the presence of a fungus that causes ringworm, but it does not identify the species or type of ringworm. Testing such as fungal culture and molecular testing can help identify specific types of ringworm, but these tests are not often performed and may take a long time to yield results.
Routine fungal cultures cannot identify TMVII and T indotineae; these tests may identify the genus Trichophyton, but only advanced molecular testing, which is available at selected US laboratories, can identify TMVII and T indotineae.
We recommend confirmatory testing because ringworm can easily be misdiagnosed as skin conditions such as psoriasis or eczema. The use of topical steroids can worsen a ringworm infection, so clinicians should be cautious about treating a rash with topical steroids if the etiology is unclear. Treatment should not be delayed if testing is not available.
Clinicians who suspect a case of TMVII infection or infection with another emerging type of severe or antifungal-resistant ringworm can contact the Centers for Disease Control and Prevention (CDC) at fungaloutbreaks@cdc.gov. More details on how clinicians can pursue testing to identify emerging strains of ringworm can be found on the American Academy of Dermatology (AAD) emerging diseases task force website.
How should clinicians treat and manage sexually transmitted ringworm?
If TMVII infection is suspected, providers can consider starting empirical treatment with oral terbinafine. Although data are limited, experience from case series suggests that TMVII may require oral antifungal treatment because it can cause severe skin infections and often does not improve with topical antifungals. Clinicians should advise patients that they may need prolonged treatment courses until the rash resolves, with possible need for treatment courses of 6-8 weeks or longer.
Any diagnosis of a sexually transmitted infection is an opportunity to engage patients in comprehensive sexual health services. Patients with suspected sexually transmitted ringworm should be evaluated for HIV and other sexually transmitted infections, including syphilis, chlamydia, and gonorrhea; clinicians should discuss and facilitate access to other preventive services, such as HIV pre-exposure prophylaxis if the patient is HIV negative and at risk for HIV. Patients should also notify their partner(s) about the diagnosis.
Is sexually transmitted ringworm a public health concern?
It is important to know that very few cases of TMVII have been reported in the United States thus far. CDC continues to monitor emerging dermatophyte strains because these types of ringworm can cause more severe or difficult-to-treat infections. Clinicians should be aware of the potential severity of sexually transmitted ringworm infections and of how diagnosis and treatment of these infections may differ from typical management of ringworm.
So far, TMVII, the dermatophyte strain most associated with spread through sexual contact, has not been documented to have antifungal resistance. More rarely, sexually transmitted ringworm may be caused by other emerging dermatophyte strains that are antifungal resistant, such as T indotineae. Itraconazole is the recommended first-line treatment for T indotineae infections.
How can clinicians counsel patients with sexually transmitted ringworm?
Ringworm can spread with skin-to-skin contact, so patients should avoid such contact with others while they have a rash. They should also avoid sharing personal items (such as razors or towels) and clothing, and launder their clothing, towels, and bedding in a high heat cycle.
People can reduce their risk of getting all types of ringworm infection by keeping their skin clean and dry, changing their socks and underwear daily, and wearing sandals in public locker rooms and other public spaces. People should avoid skin-to-skin contact with anyone with ringworm or an unexplained rash. Before having sex, people can check in with their partners and be aware of unexplained rashes on their partners’ bodies.
Where can clinicians go to learn more about sexually transmitted and other emerging types of ringworm?
CDC has partnered with the AAD to create set of online resources for clinicians for diagnosing and managing emerging dermatophyte infections. Clinicians who suspect or confirm antimicrobial resistant ringworm infection are also encouraged to submit cases to the AAD’s Emerging Diseases Registry. Clinicians wanting further guidance on how to manage suspected or confirmed ringworm infection with an emerging dermatophyte strain can also contact the CDC at fungaloutbreaks@cdc.gov. Useful information on emerging dermatophyte infections for providers and patients is also available on CDC’s website.
Relevant Reading
Zucker J et al. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.Spivack S et al. Emerg Infect Dis. 2024;30:807-809.Jabet A et al. Emerg Infect Dis. 2023;29:1411-1414.
A version of this article appeared on Medscape.com.
Dr Anand is Epidemic Intelligence Service Officer, Division of STD Prevention, Centers for Disease Control and Prevention, Atlanta, Georgia. Dr Gold is Medical Officer, Mycotic Diseases Branch, Centers for Disease Control and Prevention. Dr Quilter is Medical Officer, Division of STD Prevention, Centers for Disease Control and Prevention. None reported any relevant conflicts of interest.
Ringworm (also known as tinea, jock itch, or athlete’s foot) is a common infection caused by dermatophyte fungi, known to affect skin, hair, or nails. It causes skin infections that are typically mild and are often treated with topical antifungals.
However, in recent years, newly emerging dermatophyte strains have been causing more severe and harder-to-treat ringworm. Notably, one emerging strain, Trichophyton mentagrophytes genotype VII(TMVII), is associated with sexual contact. In recent years, TMVII infections linked to sexual contact have been reported among men who have sex with men in Europe and in travelers returning from Southeast Asia. The first US case of TMVII was reported in June 2024, after which public health authorities were alerted to additional cases; all were associated with recent sexual contact. Other dermatophyte species have also been reported to cause ringworm transmitted through sexual contact.
Here are some key points to know about sexually transmitted ringworm.
Tell me more about sexually transmitted ringworm: What is causing it?
Skin-to-skin contact is a common mode of ringworm transmission. Infections with sexually transmitted TMVII commonly cause lesions on anatomical sites that may be exposed during intimate or sexual contact, such as the face, genitals, and perianal region. Sexual transmission of TMVII has been reported in Europe, predominantly among men who have sex with men, for several years. Other dermatophyte strains have been reported in association with sexual contact, including the emerging strain Trichophyton indotineae. However, sexual transmission is not the main mode of transmission for T indotineae and other dermatophyte strains.
When should clinicians suspect a potential case of sexually transmitted ringworm?
Providers should consider sexually transmitted ringworm when seeing ringworm in locations associated with intimate contact (for example, a rash on or around the genitals, perianal area, or mouth).
The typical appearance of ringworm is a raised, ring-like, erythematous rash with a scaly border that grows over time. The rash may appear pink, brown, or gray on different types of skin. Patients may note itching and flaking of the rash. In areas with hair such as the beard area, ringworm can present as pustules and be associated with hair loss.
Emerging ringworm infections can present in atypical or more severe ways, including a highly inflammatory (painful, scarring, or otherwise severe) rash, a rash affecting a large area or multiple sites, nodules, and pustules.
Sexually transmitted ringworm may be considered based on sexual history and recent sexual contact with someone with known TMVII. Recent history of travel to a region with reported sexually transmitted ringworm may increase suspicion of TMVII. In patients with a travel history to South Asia, T indotineae should be considered, especially if the rash does not improve with oral terbinafine.
How can testing help guide the diagnosis of sexually transmitted ringworm infection?
When evaluating a rash that may represent ringworm, providers should use a confirmatory test such as potassium hydroxide (KOH) preparation when possible. KOH prep can confirm the presence of a fungus that causes ringworm, but it does not identify the species or type of ringworm. Testing such as fungal culture and molecular testing can help identify specific types of ringworm, but these tests are not often performed and may take a long time to yield results.
Routine fungal cultures cannot identify TMVII and T indotineae; these tests may identify the genus Trichophyton, but only advanced molecular testing, which is available at selected US laboratories, can identify TMVII and T indotineae.
We recommend confirmatory testing because ringworm can easily be misdiagnosed as skin conditions such as psoriasis or eczema. The use of topical steroids can worsen a ringworm infection, so clinicians should be cautious about treating a rash with topical steroids if the etiology is unclear. Treatment should not be delayed if testing is not available.
Clinicians who suspect a case of TMVII infection or infection with another emerging type of severe or antifungal-resistant ringworm can contact the Centers for Disease Control and Prevention (CDC) at fungaloutbreaks@cdc.gov. More details on how clinicians can pursue testing to identify emerging strains of ringworm can be found on the American Academy of Dermatology (AAD) emerging diseases task force website.
How should clinicians treat and manage sexually transmitted ringworm?
If TMVII infection is suspected, providers can consider starting empirical treatment with oral terbinafine. Although data are limited, experience from case series suggests that TMVII may require oral antifungal treatment because it can cause severe skin infections and often does not improve with topical antifungals. Clinicians should advise patients that they may need prolonged treatment courses until the rash resolves, with possible need for treatment courses of 6-8 weeks or longer.
Any diagnosis of a sexually transmitted infection is an opportunity to engage patients in comprehensive sexual health services. Patients with suspected sexually transmitted ringworm should be evaluated for HIV and other sexually transmitted infections, including syphilis, chlamydia, and gonorrhea; clinicians should discuss and facilitate access to other preventive services, such as HIV pre-exposure prophylaxis if the patient is HIV negative and at risk for HIV. Patients should also notify their partner(s) about the diagnosis.
Is sexually transmitted ringworm a public health concern?
It is important to know that very few cases of TMVII have been reported in the United States thus far. CDC continues to monitor emerging dermatophyte strains because these types of ringworm can cause more severe or difficult-to-treat infections. Clinicians should be aware of the potential severity of sexually transmitted ringworm infections and of how diagnosis and treatment of these infections may differ from typical management of ringworm.
So far, TMVII, the dermatophyte strain most associated with spread through sexual contact, has not been documented to have antifungal resistance. More rarely, sexually transmitted ringworm may be caused by other emerging dermatophyte strains that are antifungal resistant, such as T indotineae. Itraconazole is the recommended first-line treatment for T indotineae infections.
How can clinicians counsel patients with sexually transmitted ringworm?
Ringworm can spread with skin-to-skin contact, so patients should avoid such contact with others while they have a rash. They should also avoid sharing personal items (such as razors or towels) and clothing, and launder their clothing, towels, and bedding in a high heat cycle.
People can reduce their risk of getting all types of ringworm infection by keeping their skin clean and dry, changing their socks and underwear daily, and wearing sandals in public locker rooms and other public spaces. People should avoid skin-to-skin contact with anyone with ringworm or an unexplained rash. Before having sex, people can check in with their partners and be aware of unexplained rashes on their partners’ bodies.
Where can clinicians go to learn more about sexually transmitted and other emerging types of ringworm?
CDC has partnered with the AAD to create set of online resources for clinicians for diagnosing and managing emerging dermatophyte infections. Clinicians who suspect or confirm antimicrobial resistant ringworm infection are also encouraged to submit cases to the AAD’s Emerging Diseases Registry. Clinicians wanting further guidance on how to manage suspected or confirmed ringworm infection with an emerging dermatophyte strain can also contact the CDC at fungaloutbreaks@cdc.gov. Useful information on emerging dermatophyte infections for providers and patients is also available on CDC’s website.
Relevant Reading
Zucker J et al. MMWR Morb Mortal Wkly Rep. 2024;73:985-988.Spivack S et al. Emerg Infect Dis. 2024;30:807-809.Jabet A et al. Emerg Infect Dis. 2023;29:1411-1414.
A version of this article appeared on Medscape.com.
Dr Anand is Epidemic Intelligence Service Officer, Division of STD Prevention, Centers for Disease Control and Prevention, Atlanta, Georgia. Dr Gold is Medical Officer, Mycotic Diseases Branch, Centers for Disease Control and Prevention. Dr Quilter is Medical Officer, Division of STD Prevention, Centers for Disease Control and Prevention. None reported any relevant conflicts of interest.
The Multipronged Problem of Candida auris
The Multipronged Problem of Candida auris
Candida auris, a yeast-like fungus, is spreading globally, increasing the urgency for enhanced surveillance, new therapies, and more antimicrobial stewardship to combat its multidrug-resistant strains.
Since its discovery in 2009, C auris has been found in more than 50 countries across six continents, including Asia, Africa, and the Americas, according to the World Health Organization. In 2022, CDC reported 2377 clinical cases and 5754 screening cases of C auris in the United States.
In September, The Lancet Microbe reported on three C auris isolates from a Singapore hospital belonging to a new clade (clade six), “which is phenotypically and genotypically distinct” from the first five clades, the authors wrote. In June, Microbiology Spectrum published a study about two unusual C auris isolates from a Bangladesh NICU in 2021. They were also assigned to clade six “with potential for international transmission,” the study authors noted.
C auris has all the hallmarks of “critical pathogen,” as defined by the World Health Organization in 2022. It increases morbidity and mortality for affected patients, is difficult to eradicate in hospitals, and can be treatment resistant.
As a result, infectious disease specialists are raising more awareness and advocating for greater surveillance of C auris colonization and disease in the hospital setting for high-risk patients.
Arturo Casadevall, MD, PhD, MS, is one of them. “C auris could be a problem in your hospital as fungal diseases are getting worse every year,” said Casadevall, chair of Molecular Microbiology and Immunology at Johns Hopkins Bloomberg School of Public Health in Baltimore. The increasing number of cases “is incremental, but when [we] look at the data over years, it is a growing problem. We may see more of these cases in the coming years.”
Expediting Diagnoses
Symptoms of C auris disease vary and can cause invasive infections, such as bloodstream or intra-abdominal infections. This is why Casadevall encourages infectious disease specialists to “always consider fungal disease when you are approaching an individual. The diagnosis is sometimes delayed because you don’t look for it,” he said.
C auris can also be misidentified in the lab “when using traditional biochemical methods for yeast identification. Accurate identification of C auris requires use of sequencing or mass spectrometry,” according to CDC.
C auris is typically found on the skin of colonized patients and can enter the body through invasive devices, incisions, wounds, and during surgery. Mostly, immunosuppressed patients are at risk for serious fungal disease, Casadevall said.
Invasive fungal disease can be life-threatening for hospitalized patients. In one review of 37 studies from 2011 to 2021, researchers found that overall mortality rates for C auris infections ranged from 29% to 62%, with 30-day mortality rates between 23% and 67%, Medical Mycology reported. Patients typically had a median hospital stay of 46-68 days, sometimes extending up to 140 days. Late-onset complications included metastatic septic issues, according to the study.
Overcoming Treatment-Resistant Strains
A resilient yeast, C auris shows higher resistance to antifungal treatments compared to other Candida species, JAMA reported. Echinocandins are the first-line treatment for adults and children over 2 months old “and some of those therapies are already resistant,” said George Thompson, MD, professor of clinical medicine at the University of California Davis School of Medicine, Davis, California. The second line is liposomal amphotericin B (5 mg/kg daily), but it has toxicity problems, Thompson said.
New therapies sans toxicity are needed to treat C auris disease. Thompson, eg, served as the principal investigator in the ReSTORE trial to study a new therapy (rezafungin for injection). In March 2023, the US Food and Drug Administration approved the treatment for candidemia and invasive candidiasis in adults with limited or no alternative treatment options.
Thompson has observed that patients with C auris disease can present with “an infection in the urinary system with burning, pain, and bladder spasms. In the majority of cases of candida sepsis, the patients will have it in their blood stream with fever, chills, and sweats,” he said. The new treatment may clear the infection quickly, said Thompson, who noted results published in The Lancet.
Infection Prevention and Antimicrobial Stewardship
Institutions like University of Michigan Health (U-M Health) in Ann Arbor, Michigan, have increased measures to tackle the issue from different angles.
To address the broader issue of treatment-resistant fungal disease, U-M Health “has a robust antimicrobial stewardship program in place,” said Laraine Lynn Washer, MD, infectious disease physician.
The program includes oversight and restriction of various antifungals to avoid potential for overuse that could lead to increased risk for antifungal resistance. Use of echinocandins, for example, “requires prior approval by our antimicrobial stewardship team members,” said Washer, who is also Clinical Professor of Infectious Diseases and the Medical Director of Infection Prevention of Epidemiology at U-M Health.
Infection prevention measures entail screening hospitalized adult patients for risk factors for C auris, such as:
- Overnight international hospitalization
- Recent stay in a long-term acute care facility
- Recent stay in a ventilator skilled nursing facility.
“If a patient has these risk factors, we perform testing to assess for colonization (presence of C auris without infection) by obtaining skin swabs from the axilla and the groin and asking our lab to perform PCR to identify genetic elements of C auris,” Washer said. “Patients who are transferred directly from another hospital ICU to our ICU also undergo testing for colonization.”
If a patient is identified with C auris, hospitals ought to perform screening tests using cultures or PCR “on other patients who may have overlapped in time and space with the patient such as hospital roommates,” Washer explained.
Once in a hospital environment, the pathogen is hard to eradicate. C auris has a unique ability to be transmitted in the healthcare environment, is relatively heat tolerant, and is resistant to some common disinfectants, Washer added. The yeast can survive for over 2 weeks on plastic and months on skin, JAMA reported.
“Hospitals should partner with local and state level public health authorities in reporting cases of Candida auris and assist in any contact investigations as requested by public health authorities,” Washer advised.
Casadevall and Washer reported no conflicts of interest. Thompson has consulted and received research funding from Astellas, Basilea, Cidara, F2G, GSK, Melinta, Mundipharma, Pfizer, and Scynexis.
A version of this article appeared on Medscape.com.
Candida auris, a yeast-like fungus, is spreading globally, increasing the urgency for enhanced surveillance, new therapies, and more antimicrobial stewardship to combat its multidrug-resistant strains.
Since its discovery in 2009, C auris has been found in more than 50 countries across six continents, including Asia, Africa, and the Americas, according to the World Health Organization. In 2022, CDC reported 2377 clinical cases and 5754 screening cases of C auris in the United States.
In September, The Lancet Microbe reported on three C auris isolates from a Singapore hospital belonging to a new clade (clade six), “which is phenotypically and genotypically distinct” from the first five clades, the authors wrote. In June, Microbiology Spectrum published a study about two unusual C auris isolates from a Bangladesh NICU in 2021. They were also assigned to clade six “with potential for international transmission,” the study authors noted.
C auris has all the hallmarks of “critical pathogen,” as defined by the World Health Organization in 2022. It increases morbidity and mortality for affected patients, is difficult to eradicate in hospitals, and can be treatment resistant.
As a result, infectious disease specialists are raising more awareness and advocating for greater surveillance of C auris colonization and disease in the hospital setting for high-risk patients.
Arturo Casadevall, MD, PhD, MS, is one of them. “C auris could be a problem in your hospital as fungal diseases are getting worse every year,” said Casadevall, chair of Molecular Microbiology and Immunology at Johns Hopkins Bloomberg School of Public Health in Baltimore. The increasing number of cases “is incremental, but when [we] look at the data over years, it is a growing problem. We may see more of these cases in the coming years.”
Expediting Diagnoses
Symptoms of C auris disease vary and can cause invasive infections, such as bloodstream or intra-abdominal infections. This is why Casadevall encourages infectious disease specialists to “always consider fungal disease when you are approaching an individual. The diagnosis is sometimes delayed because you don’t look for it,” he said.
C auris can also be misidentified in the lab “when using traditional biochemical methods for yeast identification. Accurate identification of C auris requires use of sequencing or mass spectrometry,” according to CDC.
C auris is typically found on the skin of colonized patients and can enter the body through invasive devices, incisions, wounds, and during surgery. Mostly, immunosuppressed patients are at risk for serious fungal disease, Casadevall said.
Invasive fungal disease can be life-threatening for hospitalized patients. In one review of 37 studies from 2011 to 2021, researchers found that overall mortality rates for C auris infections ranged from 29% to 62%, with 30-day mortality rates between 23% and 67%, Medical Mycology reported. Patients typically had a median hospital stay of 46-68 days, sometimes extending up to 140 days. Late-onset complications included metastatic septic issues, according to the study.
Overcoming Treatment-Resistant Strains
A resilient yeast, C auris shows higher resistance to antifungal treatments compared to other Candida species, JAMA reported. Echinocandins are the first-line treatment for adults and children over 2 months old “and some of those therapies are already resistant,” said George Thompson, MD, professor of clinical medicine at the University of California Davis School of Medicine, Davis, California. The second line is liposomal amphotericin B (5 mg/kg daily), but it has toxicity problems, Thompson said.
New therapies sans toxicity are needed to treat C auris disease. Thompson, eg, served as the principal investigator in the ReSTORE trial to study a new therapy (rezafungin for injection). In March 2023, the US Food and Drug Administration approved the treatment for candidemia and invasive candidiasis in adults with limited or no alternative treatment options.
Thompson has observed that patients with C auris disease can present with “an infection in the urinary system with burning, pain, and bladder spasms. In the majority of cases of candida sepsis, the patients will have it in their blood stream with fever, chills, and sweats,” he said. The new treatment may clear the infection quickly, said Thompson, who noted results published in The Lancet.
Infection Prevention and Antimicrobial Stewardship
Institutions like University of Michigan Health (U-M Health) in Ann Arbor, Michigan, have increased measures to tackle the issue from different angles.
To address the broader issue of treatment-resistant fungal disease, U-M Health “has a robust antimicrobial stewardship program in place,” said Laraine Lynn Washer, MD, infectious disease physician.
The program includes oversight and restriction of various antifungals to avoid potential for overuse that could lead to increased risk for antifungal resistance. Use of echinocandins, for example, “requires prior approval by our antimicrobial stewardship team members,” said Washer, who is also Clinical Professor of Infectious Diseases and the Medical Director of Infection Prevention of Epidemiology at U-M Health.
Infection prevention measures entail screening hospitalized adult patients for risk factors for C auris, such as:
- Overnight international hospitalization
- Recent stay in a long-term acute care facility
- Recent stay in a ventilator skilled nursing facility.
“If a patient has these risk factors, we perform testing to assess for colonization (presence of C auris without infection) by obtaining skin swabs from the axilla and the groin and asking our lab to perform PCR to identify genetic elements of C auris,” Washer said. “Patients who are transferred directly from another hospital ICU to our ICU also undergo testing for colonization.”
If a patient is identified with C auris, hospitals ought to perform screening tests using cultures or PCR “on other patients who may have overlapped in time and space with the patient such as hospital roommates,” Washer explained.
Once in a hospital environment, the pathogen is hard to eradicate. C auris has a unique ability to be transmitted in the healthcare environment, is relatively heat tolerant, and is resistant to some common disinfectants, Washer added. The yeast can survive for over 2 weeks on plastic and months on skin, JAMA reported.
“Hospitals should partner with local and state level public health authorities in reporting cases of Candida auris and assist in any contact investigations as requested by public health authorities,” Washer advised.
Casadevall and Washer reported no conflicts of interest. Thompson has consulted and received research funding from Astellas, Basilea, Cidara, F2G, GSK, Melinta, Mundipharma, Pfizer, and Scynexis.
A version of this article appeared on Medscape.com.
Candida auris, a yeast-like fungus, is spreading globally, increasing the urgency for enhanced surveillance, new therapies, and more antimicrobial stewardship to combat its multidrug-resistant strains.
Since its discovery in 2009, C auris has been found in more than 50 countries across six continents, including Asia, Africa, and the Americas, according to the World Health Organization. In 2022, CDC reported 2377 clinical cases and 5754 screening cases of C auris in the United States.
In September, The Lancet Microbe reported on three C auris isolates from a Singapore hospital belonging to a new clade (clade six), “which is phenotypically and genotypically distinct” from the first five clades, the authors wrote. In June, Microbiology Spectrum published a study about two unusual C auris isolates from a Bangladesh NICU in 2021. They were also assigned to clade six “with potential for international transmission,” the study authors noted.
C auris has all the hallmarks of “critical pathogen,” as defined by the World Health Organization in 2022. It increases morbidity and mortality for affected patients, is difficult to eradicate in hospitals, and can be treatment resistant.
As a result, infectious disease specialists are raising more awareness and advocating for greater surveillance of C auris colonization and disease in the hospital setting for high-risk patients.
Arturo Casadevall, MD, PhD, MS, is one of them. “C auris could be a problem in your hospital as fungal diseases are getting worse every year,” said Casadevall, chair of Molecular Microbiology and Immunology at Johns Hopkins Bloomberg School of Public Health in Baltimore. The increasing number of cases “is incremental, but when [we] look at the data over years, it is a growing problem. We may see more of these cases in the coming years.”
Expediting Diagnoses
Symptoms of C auris disease vary and can cause invasive infections, such as bloodstream or intra-abdominal infections. This is why Casadevall encourages infectious disease specialists to “always consider fungal disease when you are approaching an individual. The diagnosis is sometimes delayed because you don’t look for it,” he said.
C auris can also be misidentified in the lab “when using traditional biochemical methods for yeast identification. Accurate identification of C auris requires use of sequencing or mass spectrometry,” according to CDC.
C auris is typically found on the skin of colonized patients and can enter the body through invasive devices, incisions, wounds, and during surgery. Mostly, immunosuppressed patients are at risk for serious fungal disease, Casadevall said.
Invasive fungal disease can be life-threatening for hospitalized patients. In one review of 37 studies from 2011 to 2021, researchers found that overall mortality rates for C auris infections ranged from 29% to 62%, with 30-day mortality rates between 23% and 67%, Medical Mycology reported. Patients typically had a median hospital stay of 46-68 days, sometimes extending up to 140 days. Late-onset complications included metastatic septic issues, according to the study.
Overcoming Treatment-Resistant Strains
A resilient yeast, C auris shows higher resistance to antifungal treatments compared to other Candida species, JAMA reported. Echinocandins are the first-line treatment for adults and children over 2 months old “and some of those therapies are already resistant,” said George Thompson, MD, professor of clinical medicine at the University of California Davis School of Medicine, Davis, California. The second line is liposomal amphotericin B (5 mg/kg daily), but it has toxicity problems, Thompson said.
New therapies sans toxicity are needed to treat C auris disease. Thompson, eg, served as the principal investigator in the ReSTORE trial to study a new therapy (rezafungin for injection). In March 2023, the US Food and Drug Administration approved the treatment for candidemia and invasive candidiasis in adults with limited or no alternative treatment options.
Thompson has observed that patients with C auris disease can present with “an infection in the urinary system with burning, pain, and bladder spasms. In the majority of cases of candida sepsis, the patients will have it in their blood stream with fever, chills, and sweats,” he said. The new treatment may clear the infection quickly, said Thompson, who noted results published in The Lancet.
Infection Prevention and Antimicrobial Stewardship
Institutions like University of Michigan Health (U-M Health) in Ann Arbor, Michigan, have increased measures to tackle the issue from different angles.
To address the broader issue of treatment-resistant fungal disease, U-M Health “has a robust antimicrobial stewardship program in place,” said Laraine Lynn Washer, MD, infectious disease physician.
The program includes oversight and restriction of various antifungals to avoid potential for overuse that could lead to increased risk for antifungal resistance. Use of echinocandins, for example, “requires prior approval by our antimicrobial stewardship team members,” said Washer, who is also Clinical Professor of Infectious Diseases and the Medical Director of Infection Prevention of Epidemiology at U-M Health.
Infection prevention measures entail screening hospitalized adult patients for risk factors for C auris, such as:
- Overnight international hospitalization
- Recent stay in a long-term acute care facility
- Recent stay in a ventilator skilled nursing facility.
“If a patient has these risk factors, we perform testing to assess for colonization (presence of C auris without infection) by obtaining skin swabs from the axilla and the groin and asking our lab to perform PCR to identify genetic elements of C auris,” Washer said. “Patients who are transferred directly from another hospital ICU to our ICU also undergo testing for colonization.”
If a patient is identified with C auris, hospitals ought to perform screening tests using cultures or PCR “on other patients who may have overlapped in time and space with the patient such as hospital roommates,” Washer explained.
Once in a hospital environment, the pathogen is hard to eradicate. C auris has a unique ability to be transmitted in the healthcare environment, is relatively heat tolerant, and is resistant to some common disinfectants, Washer added. The yeast can survive for over 2 weeks on plastic and months on skin, JAMA reported.
“Hospitals should partner with local and state level public health authorities in reporting cases of Candida auris and assist in any contact investigations as requested by public health authorities,” Washer advised.
Casadevall and Washer reported no conflicts of interest. Thompson has consulted and received research funding from Astellas, Basilea, Cidara, F2G, GSK, Melinta, Mundipharma, Pfizer, and Scynexis.
A version of this article appeared on Medscape.com.
The Multipronged Problem of Candida auris
The Multipronged Problem of Candida auris
After Rapid Weight Loss, Monitor Antiobesity Drug Dosing
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
A patient who developed atrial fibrillation resulting from the failure to adjust the levothyroxine dose after rapid, significant weight loss while on the antiobesity drug tirzepatide (Zepbound) serves as a key reminder in managing patients experiencing rapid weight loss, either from antiobesity medications or any other means: Patients taking medications with weight-based dosing need to have their doses closely monitored.
“Failing to monitor and adjust dosing of these [and other] medications during a period of rapid weight loss may lead to supratherapeutic — even toxic — levels, as was seen in this [case],” underscore the authors of an editorial regarding the Teachable Moment case, published in JAMA Internal Medicine.
Toxicities from excessive doses can have a range of detrimental effects. In terms of thyroid medicine, the failure to adjust levothyroxine treatment for hypothyroidism in cases of rapid weight loss can lead to thyrotoxicosis, and in older patients in particular, a resulting thyrotropin level < 0.1 mIU/L is associated with as much as a threefold increased risk for atrial fibrillation, as observed in the report.
Case Demonstrates Risks
The case involved a 62-year-old man with obesity, hypothyroidism, and type 1 diabetes who presented to the emergency department with palpitations, excessive sweating, confusion, fever, and hand tremors. Upon being diagnosed with atrial fibrillation, the patient was immediately treated.
His medical history revealed the underlying culprit: Six months earlier, the patient had started treatment with the gastric inhibitory polypeptide (GIP)/glucagon-like peptide (GLP) 1 dual agonist tirzepatide. As is typical with the drug, the patient’s weight quickly plummeted, dropping from a starting body mass index of 44.4 down to 31.2 after 6 months and a decrease in body weight from 132 kg to 93 kg (a loss of 39 kg [approximately 86 lb]).
When he was prescribed tirzepatide, 2.5 mg weekly, for obesity, the patient had been recommended to increase the dose every 4 weeks as tolerated and, importantly, to have a follow-up visit in a month. But because he lived in different states seasonally, the follow-up never occurred.
Upon his emergency department visit, the patient’s thyrotropin level had dropped from 1.9 mIU/L at the first visit 6 months earlier to 0.001 mIU/L (well within the atrial fibrillation risk range), and his free thyroxine level (fT4) was 7.26 ng/ dL — substantially outside of the normal range of about 0.9-1.7 ng/dL for adults.
“The patient had 4-times higher fT4 levels of the upper limit,” first author Kagan E. Karakus, MD, of the Barbara Davis Center for Diabetes, University of Colorado Anschutz Medical Campus, Aurora, told this news organization. “That is why he had experienced the adverse event of atrial fibrillation.”
Thyrotoxicosis Symptoms Can Be ‘Insidious,’ Levothyroxine Should Be Monitored
Although tirzepatide has not been approved by the US Food and Drug Administration for the treatment of type 1 diabetes, obesity is on the rise among patients with this disorder and recent research has shown a more than 10% reduction in body weight in 6 months and significant reductions in A1c with various doses.
Of note, in the current case, although the patient’s levothyroxine dose was not adjusted, his insulin dose was gradually self-decreased during his tirzepatide treatment to prevent hypoglycemia.
“If insulin treatment is excessive in diabetes, it causes hypoglycemia, [and] people with type 1 diabetes will recognize the signs of hypoglycemia related to excessive insulin earlier,” Dr. Karakus said.
If symptoms appear, patients can reduce their insulin doses on their own; however, the symptoms of thyrotoxicosis caused by excessive levothyroxine can be more insidious compared with hypoglycemia, he explained.
“Although patients can change their insulin doses, they cannot change the levothyroxine doses since it requires a blood test [thyroid-stimulating hormone; TSH] and a new prescription of the new dose.”
The key lesson is that “following levothyroxine treatment initiation or dose adjustment, 4-6 weeks is the optimal duration to recheck [the] thyrotropin level and adjust the dose as needed,” Dr. Karakus said.
Key Medications to Monitor
Other common outpatient medications that should be closely monitored in patients experiencing rapid weight loss, by any method, range from anticoagulants, anticonvulsants, and antituberculosis drugs to antibiotics and antifungals, the authors note.
Of note, medications with a narrow therapeutic index include phenytoin, warfarin, lithium carbonate, digoxin theophylline, tacrolimus, valproic acid, carbamazepine, and cyclosporine.
The failure to make necessary dose adjustments “is seen more often since the newer antiobesity drugs reduce a great amount of weight within months, almost as rapidly as bariatric surgery,” Dr. Karakus said.
“It is very important for physicians to be aware of the weight-based medications and narrow therapeutic index medications since their doses should be adjusted carefully, especially during weight loss,” he added.
Furthermore, “the patient should also know that weight reduction medication may cause adverse effects like nausea, vomiting and also may affect metabolism of other medications such that some medication doses should be adjusted regularly.”
In the editorial published with the study, Tyrone A. Johnson, MD, of the Department of Medicine, University of California, San Francisco, and colleagues note that the need for close monitoring is particularly important with older patients, who, in addition to having a higher likelihood of comorbidities, commonly have polypharmacy that could increase the potential for adverse effects.
Another key area concern is the emergence of direct-to-consumer avenues for GLP-1/GIP agonists for the many who either cannot afford or do not have access to the drugs, providing further opportunities for treatment without appropriate clinical oversight, they add.
Overall, the case “highlights the potential dangers underlying under-supervised prescribing of GLP-1/GIP receptor agonists and affirms the need for strong partnerships between patients and their clinicians during their use,” they wrote.
“These medications are best used in collaboration with continuity care teams, in context of a patient’s entire health, and in comprehensive risk-benefit assessment throughout the entire duration of treatment.”
A Caveat: Subclinical Levothyroxine Dosing
Commenting on the study, Matthew Ettleson, MD, a clinical instructor of medicine in the Section of Endocrinology, Diabetes, & Metabolism, University of Chicago, noted the important caveat that patients with hypothyroidism are commonly on subclinical doses, with varying dose adjustment needs.
“The patient in the case was clearly on a replacement level dose. However, many patients are on low doses of levothyroxine (75 µg or lower) for subclinical hypothyroidism, and, in general, I think the risks are lower with patients with subclinical hypothyroidism on lower doses of levothyroxine,” he told this news organization.
Because of that, “frequent TSH monitoring may be excessive in this population,” he said. “I would hesitate to empirically lower the dose with weight loss, unless it was clear that the patient was unlikely to follow up.
“Checking TSH at a more frequent interval and adjusting the dose accordingly should be adequate to prevent situations like this case.”
Dr. Karakus, Dr. Ettleson, and the editorial authors had no relevant disclosures to report.
A version of this article appeared on Medscape.com.
Could a Fungal Infection Cause a Future Pandemic?
The principle of resilience and survival is crucial for medically significant fungi. These microorganisms are far from creating the postapocalyptic scenario depicted in TV series like The Last of Us, and much work is necessary to learn more about them. Accurate statistics on fungal infections, accompanied by clinical histories, simple laboratory tests, new antifungals, and a necessary One Health approach are lacking.
The entomopathogenic fungus Ophiocordyceps unilateralis was made notorious by the TV series, but for now, it only manages to control the brains of some ants at will. Luckily, there are no signs that fungi affecting humans are inclined to create zombies.
What is clear is that the world belongs to the kingdom of fungi and that fungi are everywhere. There are already close to 150,000 described species, but millions remain to be discovered. They abound in decomposing organic matter, soil, or animal excrement, including that of bats and pigeons. Some fungi have even managed to find a home in hospitals. Lastly, we must not forget those that establish themselves in the human microbiome.
Given such diversity, it is legitimate to ask whether any of them could be capable of generating new pandemics. Could the forgotten Cryptococcus neoformans, Aspergillus fumigatus, or Histoplasma species, among others, trigger new health emergencies on the scale of the one generated by SARS-CoV-2?
We cannot forget that a coronavirus has already confirmed that reality can surpass fiction. However, Edith Sánchez Paredes, a biologist, doctor in biomedical sciences, and specialist in medical mycology, provided a reassuring response to Medscape Spanish Edition on this point.
“That would be very difficult to see because the way fungal infections are acquired is not from person to person, in most cases,” said Dr. Sánchez Paredes, from the Mycology Unit of the Faculty of Medicine at the National Autonomous University of Mexico.
Close to 300 species have already been classified as pathogenic in humans. Although the numbers are not precise and are increasing, it is estimated that around 1,500,000 people worldwide die each year of systemic fungal infections.
“However, it is important to emphasize that establishment of an infection depends not only on the causal agent. A crucial factor is the host, in this case, the human. Generally, these types of infections will develop in individuals with some deficiency in their immune system. The more deficient the immune response, the more likely a fungal infection may occur,” stated Dr. Sánchez Paredes.
The possibility of a pandemic like the one experienced with SARS-CoV-2 in the short term is remote, but the threat posed by fungal infections persists.
In 2022, the World Health Organization (WHO) defined a priority list of pathogenic fungi, with the aim of guiding actions to control them. It is mentioned there that invasive fungal diseases are on the rise worldwide, particularly in immunocompromised populations.
“Despite the growing concern, fungal infections receive very little attention and resources, leading to a paucity of quality data on fungal disease distribution and antifungal resistance patterns. Consequently, it is impossible to estimate their exact burden,” as stated in the document.
In line with this, an article published in Mycoses in 2022 concluded that fungal infections are neglected diseases in Latin America. Among other difficulties, deficiencies in access to tests such as polymerase chain reaction or serum detection of beta-1,3-D-glucan have been reported there.
In terms of treatments, most countries encounter problems with access to liposomal amphotericin B and new azoles, such as posaconazole and isavuconazole.
“Unfortunately, in Latin America, we suffer from a poor infrastructure for diagnosing fungal infections; likewise, we have limited access to antifungals available in the global market. What’s more, we lack reliable data on the epidemiology of fungal infections in the region, so many times governments are unaware of the true extent of the problem,” said Rogelio de Jesús Treviño Rangel, PhD, a medical microbiologist and expert in clinical mycology, professor, and researcher at the Faculty of Medicine of the Autonomous University of Nuevo León in Mexico.
Need for More Medical Mycology Training
Dr. Fernando Messina is a medical mycologist with the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital in Buenos Aires, Argentina. He has noted an increase in the number of cases of cryptococcosis, histoplasmosis, and aspergillosis in his daily practice.
“Particularly, pulmonary aspergillosis is steadily increasing. This is because many patients have structural lung alterations that favor the appearance of this mycosis. This is related to the increase in cases of tuberculosis and the rise in life expectancy of patients with chronic obstructive pulmonary disease or other pulmonary or systemic diseases,” Dr. Messina stated.
For Dr. Messina, the main obstacle in current clinical practice is the low level of awareness among nonspecialist physicians regarding the presence of systemic fungal infections, and because these infections are more common than realized, it is vital to consider fungal etiology before starting empirical antibiotic therapy.
“Health professionals usually do not think about mycoses because mycology occupies a very small space in medical education at universities. As the Venezuelan mycologist Gioconda Cunto de San Blas once said, ‘Mycology is the Cinderella of microbiology.’ To change this, we need to give more space to mycoses in undergraduate and postgraduate studies,” Dr. Messina asserted.
He added, “The main challenge is to train professionals with an emphasis on the clinical interpretation of cases. Current medicine has a strong trend toward molecular biology and the use of rapid diagnostic methods, without considering the clinical symptoms or the patient’s history. Determinations are very useful, but it is necessary to interpret the results.”
Dr. Messina sees it as unlikely in the short term for a pandemic to be caused by fungi, but if it were to occur, he believes it would happen in healthcare systems in regions that are not prepared in terms of infrastructure. However, as seen in the health emergency resulting from SARS-CoV-2, he thinks the impact would be mitigated by the performance of healthcare professionals.
“In general, we have the ability to adapt to any adverse situation or change — although it is clear that we need more doctors, biochemists, and microbiologists trained in mycology,” emphasized Dr. Messina.
More than 40 interns pass through Muñiz Hospital each year. They are doctors and biochemists from Argentina, other countries in the region, or even Europe, seeking to enhance their training in mycology. Regarding fungal infection laboratory work, the interest lies in learning to use traditional techniques and innovative molecular methods.
“Rapid diagnostic methods, especially the detection of circulating antigens, have marked a change in the prognosis of deep mycosis in immunocompromised hosts. The possibility of screening and monitoring in this group of patients is very important and has a great benefit,” said Gabriela Santiso, PhD, a biochemist and head of the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital.
According to Dr. Santiso, the current landscape includes the ability to identify genus and species, which can help in understanding resistance to antifungals. Furthermore, conducting sensitivity tests to these drugs, using standardized commercial methods, also provides timely information for treatment.
But Dr. Santiso warns that Latin America is a vast region with great disparity in human and technological resources. Although most countries in the region have networks facilitating access to timely diagnosis, resources are generally more available in major urban centers.
This often clashes with the epidemiology of most fungal infections. “Let’s not forget that many fungal pathologies affect low-income people who have difficulties accessing health centers, which sometimes turns them into chronic diseases that are hard to treat,” Dr. Santiso pointed out.
In mycology laboratories, the biggest cost is incurred by new diagnostic tests, such as those allowing molecular identification. Conventional methods are not usually expensive, but they require time and effort to train human resources to handle them.
Because new methodologies are not always available or easily accessible throughout the region, Dr. Santiso recommended not neglecting traditional mycological techniques. “Molecular methods, rapid diagnostic methods, and conventional mycology techniques are complementary and not mutually exclusive tests. Continuous training and updating are needed in this area,” she emphasized.
Why Are Resistant Fungal Infections Becoming Increasingly Common?
The first barrier for fungi to cause infection in humans is body temperature; most of them cannot withstand 37 °C. However, they also struggle to evade the immune response that is activated when they try to enter the body.
“We are normally exposed to many of these fungi, almost all the time, but if our immune system is adequate, it may not go beyond a mild infection, in most cases subclinical, which will resolve quickly,” Dr. Sánchez Paredes stated.
However, according to Dr. Sánchez Paredes, if the immune response is weak, “the fungus will have no trouble establishing itself in our organs. Some are even part of our microbiota, such as Candida albicans, which in the face of an imbalance or immunocompromise, can lead to serious infections.”
It is clear that the population at risk for immunosuppression has increased. According to the WHO, this is due to the high prevalence of such diseases as tuberculosis, cancer, and HIV infection, among others.
But the WHO also believes that the increase in fungal infections is related to greater population access to critical care units, invasive procedures, chemotherapy, or immunotherapy treatments.
Furthermore, factors related to the fungus itself and the environment play a role. “These organisms have enzymes, proteins, and other molecules that allow them to survive in the environment in which they normally inhabit. When they face a new and stressful one, they must express other molecules that will allow them to survive. All of this helps them evade elements of the immune system, antifungals, and, of course, body temperature,” according to Dr. Sánchez Paredes.
It is possible that climate change is also behind the noticeable increase in fungal infections and that this crisis may have an even greater impact in the future. The temperature of the environment has increased, and fungi will have to adapt to the planet’s temperature, to the point where body temperature may no longer be a significant barrier for them.
Environmental changes would also be responsible for modifications in the distribution of endemic mycoses, and it is believed that fungi will more frequently find new ecological niches, be able to survive in other environments, and alter distribution zones.
This is what is happening between Mexico and the United States with coccidioidomycosis, or valley fever. “We will begin to see cases of some mycoses where they were not normally seen, so we will have to conduct more studies to confirm that the fungus is inhabiting these new areas or is simply appearing in new sites owing to migration and the great mobility of populations,” Dr. Sánchez Paredes said.
Finally, exposure to environmental factors would partly be responsible for the increasing resistance to first-line antifungals observed in these microorganisms. This seems to be the case with A. fumigatus when exposed to azoles used as fungicides in agriculture.
One Health in Fungal Infections
The increasing resistance to antifungals is a clear testament that human, animal, and environmental health are interconnected. This is why a multidisciplinary approach that adopts the perspective of One Health is necessary for its management.
“The use of fungicides in agriculture, structurally similar to the azoles used in clinics, generates resistance in Aspergillus fumigatus found in the environment. These fungi in humans can be associated with infections that do not respond to first-line treatment,” explained Carlos Arturo Álvarez, an infectious diseases physician and professor at the Faculty of Medicine at the National University of Colombia.
According to Dr. Álvarez, the approach to control them should not only focus on the search for diagnostic methods that allow early detection of antifungal resistance or research on new antifungal treatments. He believes that progress must also be made with strategies that allow for the proper use of antifungals in agriculture.
“Unfortunately, the One Health approach is not yet well implemented in the region, and in my view, there is a lack of articulation in the different sectors. That is, there is a need for true coordination between government offices of agriculture, animal and human health, academia, and international organizations. This is not happening yet, and I believe we are in the initial stage of visibility,” Dr. Álvarez opined.
Veterinary public health is another pillar of the aforementioned approach. For various reasons, animals experience a higher frequency of fungal infections. A few carry and transmit true zoonoses that affect human health, but most often, animals act only as sentinels indicating a potential source of transmission.
Carolina Segundo Zaragoza, PhD, has worked in veterinary mycology for 30 years. She currently heads the veterinary mycology laboratory at the Animal Production Teaching, Research, and Extension Center in Altiplano, under the Faculty of Veterinary Medicine and Animal Husbandry at the National Autonomous University of Mexico. Because she has frequent contact with specialists in human mycology, during her professional career she has received several patient consultations, most of which were for cutaneous mycoses.
“They detect some dermatomycosis and realize that the common factor is owning a companion animal or a production animal with which the patient has contact. Both animals and humans present the same type of lesions, and then comes the question: Who infected whom? I remind them that the main source of infection is the soil and that animals should not be blamed in the first instance,” Dr. Segundo Zaragoza clarified.
She is currently collaborating on a research project analyzing the presence of Coccidioides immitis in the soil. This pathogen is responsible for coccidioidomycosis in dogs and humans, and she sees with satisfaction how these types of initiatives, which include some components of the One Health vision, are becoming more common in Mexico.
“Fortunately, human mycologists are increasingly providing more space for the dissemination of veterinary mycology. So I have had the opportunity to be invited to different forums on medical mycology to present the clinical cases we can have in animals and talk about the research projects we carry out. I have more and more opportunities to conduct joint research with human mycologists and veterinary doctors,” she said.
Dr. Segundo Zaragoza believes that to better implement the One Health vision, standardizing the criteria for detecting, diagnosing, and treating mycoses is necessary. She considers that teamwork will be key to achieving the common goal of safeguarding the well-being and health of humans and animals.
Alarms Sound for Candida auris
The WHO included the yeast Candida auris in its group of pathogens with critical priority, and since 2009, it has raised alarm owing to the ease with which it grows in hospitals. In that setting, C auris is known for its high transmissibility, its ability to cause outbreaks, and the high mortality rate from disseminated infections.
“It has been a concern for the mycological community because it shows resistance to most antifungals used clinically, mainly azoles, but also for causing epidemic outbreaks,” emphasized Dr. Sánchez Paredes.
Its mode of transmission is not very clear, but it has been documented to be present on the skin and persist in hospital materials and furniture. It causes nosocomial infections in critically ill patients, such as those in intensive care, and those with cancer or who have received a transplant.
Risk factors for its development include renal insufficiency, hospital stays of more than 15 days, mechanical ventilation, central lines, use of parenteral nutrition, and presence of sepsis.
As for other mycoses, there are no precise studies reporting global incidence rates, but the trend indicates an increase in the detection of outbreaks in various countries lately — something that began to be visible during the COVID-19 pandemic.
In Mexico, Dr. Treviño Rangel and colleagues from Nuevo León reported the first case of candidemia caused by this agent. It occurred in May 2020 and involved a 58-year-old woman with a history of severe endometriosis and multiple complications in the gastrointestinal tract. The patient’s condition improved favorably thanks to antifungal therapy with caspofungin and liposomal amphotericin B.
However, 3 months after that episode, the group reported an outbreak of C. auris at the same hospital in 12 critically ill patients co-infected with SARS-CoV-2. All were on mechanical ventilation, had peripherally inserted central catheters and urinary catheters, and had a prolonged hospital stay (20-70 days). The mortality in patients with candidemia in this cohort was 83.3%.
Open Ending
As seen in some science fiction series, fungal infections in the region still have an open ending, and Global Action For Fungal Infections (GAFFI) has estimated that with better diagnostics and treatments, deaths caused by fungi could decrease to less than 750,000 per year worldwide.
But if everything continues as is, some aspects of what is to come may resemble the dystopia depicted in The Last of Us. No zombies, but emerging and reemerging fungi in a chaotic distribution, and resistant to all established treatments.
“The risk factors of patients and their immune status, combined with the behavior of mycoses, bring a complicated scenario. But therapeutic failure resulting from multidrug resistance to antifungals could make it catastrophic,” Dr. Sánchez Paredes summarized.
At the moment, there are only four families of drugs capable of counteracting fungal infections — and as mentioned, some are already scarce in Latin America’s hospital pharmacies.
“Historically, fungal infections have been given less importance than those caused by viruses or bacteria. Even in some developed countries, the true extent of morbidity and mortality they present is unknown. This results in less investment in the development of new antifungal molecules because knowledge is lacking about the incidence and prevalence of these diseases,” Dr. Treviño Rangel pointed out.
He added that the main limitation for the development of new drugs is economic. “Unfortunately, not many pharmaceutical companies are willing to invest in the development of new antifungals, and there are no government programs specifically promoting and supporting research into new therapeutic options against these neglected diseases,” he asserted.
Development of vaccines to prevent fungal infections faces the same barriers. Although, according to Dr. Treviño Rangel, the difficulties are compounded by the great similarity between fungal cells and human cells. This makes it possible for harmful cross-reactivity to occur. In addition, because most severe fungal infections occur in individuals with immunosuppression, a vaccine would need to trigger an adequate immune response despite this issue.
Meanwhile, fungi quietly continue to do what they do best: resist and survive. For millions of years, they have mutated and adapted to new environments. Some theories even blame them for the extinction of dinosaurs and the subsequent rise of mammals. They exist on the edge of life and death, decomposing and creating. There is consensus that at the moment, it does not seem feasible for them to generate a pandemic like the one due to SARS-CoV-2, given their transmission mechanism. But who is willing to rule out that this may not happen in the long or medium term?
Dr. Sánchez Paredes, Dr. Treviño Rangel, Dr. Messina, Dr. Santiso, Dr. Álvarez, and Dr. Segundo Zaragoza have declared no relevant financial conflicts of interest.
This story was translated from Medscape Spanish Edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The principle of resilience and survival is crucial for medically significant fungi. These microorganisms are far from creating the postapocalyptic scenario depicted in TV series like The Last of Us, and much work is necessary to learn more about them. Accurate statistics on fungal infections, accompanied by clinical histories, simple laboratory tests, new antifungals, and a necessary One Health approach are lacking.
The entomopathogenic fungus Ophiocordyceps unilateralis was made notorious by the TV series, but for now, it only manages to control the brains of some ants at will. Luckily, there are no signs that fungi affecting humans are inclined to create zombies.
What is clear is that the world belongs to the kingdom of fungi and that fungi are everywhere. There are already close to 150,000 described species, but millions remain to be discovered. They abound in decomposing organic matter, soil, or animal excrement, including that of bats and pigeons. Some fungi have even managed to find a home in hospitals. Lastly, we must not forget those that establish themselves in the human microbiome.
Given such diversity, it is legitimate to ask whether any of them could be capable of generating new pandemics. Could the forgotten Cryptococcus neoformans, Aspergillus fumigatus, or Histoplasma species, among others, trigger new health emergencies on the scale of the one generated by SARS-CoV-2?
We cannot forget that a coronavirus has already confirmed that reality can surpass fiction. However, Edith Sánchez Paredes, a biologist, doctor in biomedical sciences, and specialist in medical mycology, provided a reassuring response to Medscape Spanish Edition on this point.
“That would be very difficult to see because the way fungal infections are acquired is not from person to person, in most cases,” said Dr. Sánchez Paredes, from the Mycology Unit of the Faculty of Medicine at the National Autonomous University of Mexico.
Close to 300 species have already been classified as pathogenic in humans. Although the numbers are not precise and are increasing, it is estimated that around 1,500,000 people worldwide die each year of systemic fungal infections.
“However, it is important to emphasize that establishment of an infection depends not only on the causal agent. A crucial factor is the host, in this case, the human. Generally, these types of infections will develop in individuals with some deficiency in their immune system. The more deficient the immune response, the more likely a fungal infection may occur,” stated Dr. Sánchez Paredes.
The possibility of a pandemic like the one experienced with SARS-CoV-2 in the short term is remote, but the threat posed by fungal infections persists.
In 2022, the World Health Organization (WHO) defined a priority list of pathogenic fungi, with the aim of guiding actions to control them. It is mentioned there that invasive fungal diseases are on the rise worldwide, particularly in immunocompromised populations.
“Despite the growing concern, fungal infections receive very little attention and resources, leading to a paucity of quality data on fungal disease distribution and antifungal resistance patterns. Consequently, it is impossible to estimate their exact burden,” as stated in the document.
In line with this, an article published in Mycoses in 2022 concluded that fungal infections are neglected diseases in Latin America. Among other difficulties, deficiencies in access to tests such as polymerase chain reaction or serum detection of beta-1,3-D-glucan have been reported there.
In terms of treatments, most countries encounter problems with access to liposomal amphotericin B and new azoles, such as posaconazole and isavuconazole.
“Unfortunately, in Latin America, we suffer from a poor infrastructure for diagnosing fungal infections; likewise, we have limited access to antifungals available in the global market. What’s more, we lack reliable data on the epidemiology of fungal infections in the region, so many times governments are unaware of the true extent of the problem,” said Rogelio de Jesús Treviño Rangel, PhD, a medical microbiologist and expert in clinical mycology, professor, and researcher at the Faculty of Medicine of the Autonomous University of Nuevo León in Mexico.
Need for More Medical Mycology Training
Dr. Fernando Messina is a medical mycologist with the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital in Buenos Aires, Argentina. He has noted an increase in the number of cases of cryptococcosis, histoplasmosis, and aspergillosis in his daily practice.
“Particularly, pulmonary aspergillosis is steadily increasing. This is because many patients have structural lung alterations that favor the appearance of this mycosis. This is related to the increase in cases of tuberculosis and the rise in life expectancy of patients with chronic obstructive pulmonary disease or other pulmonary or systemic diseases,” Dr. Messina stated.
For Dr. Messina, the main obstacle in current clinical practice is the low level of awareness among nonspecialist physicians regarding the presence of systemic fungal infections, and because these infections are more common than realized, it is vital to consider fungal etiology before starting empirical antibiotic therapy.
“Health professionals usually do not think about mycoses because mycology occupies a very small space in medical education at universities. As the Venezuelan mycologist Gioconda Cunto de San Blas once said, ‘Mycology is the Cinderella of microbiology.’ To change this, we need to give more space to mycoses in undergraduate and postgraduate studies,” Dr. Messina asserted.
He added, “The main challenge is to train professionals with an emphasis on the clinical interpretation of cases. Current medicine has a strong trend toward molecular biology and the use of rapid diagnostic methods, without considering the clinical symptoms or the patient’s history. Determinations are very useful, but it is necessary to interpret the results.”
Dr. Messina sees it as unlikely in the short term for a pandemic to be caused by fungi, but if it were to occur, he believes it would happen in healthcare systems in regions that are not prepared in terms of infrastructure. However, as seen in the health emergency resulting from SARS-CoV-2, he thinks the impact would be mitigated by the performance of healthcare professionals.
“In general, we have the ability to adapt to any adverse situation or change — although it is clear that we need more doctors, biochemists, and microbiologists trained in mycology,” emphasized Dr. Messina.
More than 40 interns pass through Muñiz Hospital each year. They are doctors and biochemists from Argentina, other countries in the region, or even Europe, seeking to enhance their training in mycology. Regarding fungal infection laboratory work, the interest lies in learning to use traditional techniques and innovative molecular methods.
“Rapid diagnostic methods, especially the detection of circulating antigens, have marked a change in the prognosis of deep mycosis in immunocompromised hosts. The possibility of screening and monitoring in this group of patients is very important and has a great benefit,” said Gabriela Santiso, PhD, a biochemist and head of the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital.
According to Dr. Santiso, the current landscape includes the ability to identify genus and species, which can help in understanding resistance to antifungals. Furthermore, conducting sensitivity tests to these drugs, using standardized commercial methods, also provides timely information for treatment.
But Dr. Santiso warns that Latin America is a vast region with great disparity in human and technological resources. Although most countries in the region have networks facilitating access to timely diagnosis, resources are generally more available in major urban centers.
This often clashes with the epidemiology of most fungal infections. “Let’s not forget that many fungal pathologies affect low-income people who have difficulties accessing health centers, which sometimes turns them into chronic diseases that are hard to treat,” Dr. Santiso pointed out.
In mycology laboratories, the biggest cost is incurred by new diagnostic tests, such as those allowing molecular identification. Conventional methods are not usually expensive, but they require time and effort to train human resources to handle them.
Because new methodologies are not always available or easily accessible throughout the region, Dr. Santiso recommended not neglecting traditional mycological techniques. “Molecular methods, rapid diagnostic methods, and conventional mycology techniques are complementary and not mutually exclusive tests. Continuous training and updating are needed in this area,” she emphasized.
Why Are Resistant Fungal Infections Becoming Increasingly Common?
The first barrier for fungi to cause infection in humans is body temperature; most of them cannot withstand 37 °C. However, they also struggle to evade the immune response that is activated when they try to enter the body.
“We are normally exposed to many of these fungi, almost all the time, but if our immune system is adequate, it may not go beyond a mild infection, in most cases subclinical, which will resolve quickly,” Dr. Sánchez Paredes stated.
However, according to Dr. Sánchez Paredes, if the immune response is weak, “the fungus will have no trouble establishing itself in our organs. Some are even part of our microbiota, such as Candida albicans, which in the face of an imbalance or immunocompromise, can lead to serious infections.”
It is clear that the population at risk for immunosuppression has increased. According to the WHO, this is due to the high prevalence of such diseases as tuberculosis, cancer, and HIV infection, among others.
But the WHO also believes that the increase in fungal infections is related to greater population access to critical care units, invasive procedures, chemotherapy, or immunotherapy treatments.
Furthermore, factors related to the fungus itself and the environment play a role. “These organisms have enzymes, proteins, and other molecules that allow them to survive in the environment in which they normally inhabit. When they face a new and stressful one, they must express other molecules that will allow them to survive. All of this helps them evade elements of the immune system, antifungals, and, of course, body temperature,” according to Dr. Sánchez Paredes.
It is possible that climate change is also behind the noticeable increase in fungal infections and that this crisis may have an even greater impact in the future. The temperature of the environment has increased, and fungi will have to adapt to the planet’s temperature, to the point where body temperature may no longer be a significant barrier for them.
Environmental changes would also be responsible for modifications in the distribution of endemic mycoses, and it is believed that fungi will more frequently find new ecological niches, be able to survive in other environments, and alter distribution zones.
This is what is happening between Mexico and the United States with coccidioidomycosis, or valley fever. “We will begin to see cases of some mycoses where they were not normally seen, so we will have to conduct more studies to confirm that the fungus is inhabiting these new areas or is simply appearing in new sites owing to migration and the great mobility of populations,” Dr. Sánchez Paredes said.
Finally, exposure to environmental factors would partly be responsible for the increasing resistance to first-line antifungals observed in these microorganisms. This seems to be the case with A. fumigatus when exposed to azoles used as fungicides in agriculture.
One Health in Fungal Infections
The increasing resistance to antifungals is a clear testament that human, animal, and environmental health are interconnected. This is why a multidisciplinary approach that adopts the perspective of One Health is necessary for its management.
“The use of fungicides in agriculture, structurally similar to the azoles used in clinics, generates resistance in Aspergillus fumigatus found in the environment. These fungi in humans can be associated with infections that do not respond to first-line treatment,” explained Carlos Arturo Álvarez, an infectious diseases physician and professor at the Faculty of Medicine at the National University of Colombia.
According to Dr. Álvarez, the approach to control them should not only focus on the search for diagnostic methods that allow early detection of antifungal resistance or research on new antifungal treatments. He believes that progress must also be made with strategies that allow for the proper use of antifungals in agriculture.
“Unfortunately, the One Health approach is not yet well implemented in the region, and in my view, there is a lack of articulation in the different sectors. That is, there is a need for true coordination between government offices of agriculture, animal and human health, academia, and international organizations. This is not happening yet, and I believe we are in the initial stage of visibility,” Dr. Álvarez opined.
Veterinary public health is another pillar of the aforementioned approach. For various reasons, animals experience a higher frequency of fungal infections. A few carry and transmit true zoonoses that affect human health, but most often, animals act only as sentinels indicating a potential source of transmission.
Carolina Segundo Zaragoza, PhD, has worked in veterinary mycology for 30 years. She currently heads the veterinary mycology laboratory at the Animal Production Teaching, Research, and Extension Center in Altiplano, under the Faculty of Veterinary Medicine and Animal Husbandry at the National Autonomous University of Mexico. Because she has frequent contact with specialists in human mycology, during her professional career she has received several patient consultations, most of which were for cutaneous mycoses.
“They detect some dermatomycosis and realize that the common factor is owning a companion animal or a production animal with which the patient has contact. Both animals and humans present the same type of lesions, and then comes the question: Who infected whom? I remind them that the main source of infection is the soil and that animals should not be blamed in the first instance,” Dr. Segundo Zaragoza clarified.
She is currently collaborating on a research project analyzing the presence of Coccidioides immitis in the soil. This pathogen is responsible for coccidioidomycosis in dogs and humans, and she sees with satisfaction how these types of initiatives, which include some components of the One Health vision, are becoming more common in Mexico.
“Fortunately, human mycologists are increasingly providing more space for the dissemination of veterinary mycology. So I have had the opportunity to be invited to different forums on medical mycology to present the clinical cases we can have in animals and talk about the research projects we carry out. I have more and more opportunities to conduct joint research with human mycologists and veterinary doctors,” she said.
Dr. Segundo Zaragoza believes that to better implement the One Health vision, standardizing the criteria for detecting, diagnosing, and treating mycoses is necessary. She considers that teamwork will be key to achieving the common goal of safeguarding the well-being and health of humans and animals.
Alarms Sound for Candida auris
The WHO included the yeast Candida auris in its group of pathogens with critical priority, and since 2009, it has raised alarm owing to the ease with which it grows in hospitals. In that setting, C auris is known for its high transmissibility, its ability to cause outbreaks, and the high mortality rate from disseminated infections.
“It has been a concern for the mycological community because it shows resistance to most antifungals used clinically, mainly azoles, but also for causing epidemic outbreaks,” emphasized Dr. Sánchez Paredes.
Its mode of transmission is not very clear, but it has been documented to be present on the skin and persist in hospital materials and furniture. It causes nosocomial infections in critically ill patients, such as those in intensive care, and those with cancer or who have received a transplant.
Risk factors for its development include renal insufficiency, hospital stays of more than 15 days, mechanical ventilation, central lines, use of parenteral nutrition, and presence of sepsis.
As for other mycoses, there are no precise studies reporting global incidence rates, but the trend indicates an increase in the detection of outbreaks in various countries lately — something that began to be visible during the COVID-19 pandemic.
In Mexico, Dr. Treviño Rangel and colleagues from Nuevo León reported the first case of candidemia caused by this agent. It occurred in May 2020 and involved a 58-year-old woman with a history of severe endometriosis and multiple complications in the gastrointestinal tract. The patient’s condition improved favorably thanks to antifungal therapy with caspofungin and liposomal amphotericin B.
However, 3 months after that episode, the group reported an outbreak of C. auris at the same hospital in 12 critically ill patients co-infected with SARS-CoV-2. All were on mechanical ventilation, had peripherally inserted central catheters and urinary catheters, and had a prolonged hospital stay (20-70 days). The mortality in patients with candidemia in this cohort was 83.3%.
Open Ending
As seen in some science fiction series, fungal infections in the region still have an open ending, and Global Action For Fungal Infections (GAFFI) has estimated that with better diagnostics and treatments, deaths caused by fungi could decrease to less than 750,000 per year worldwide.
But if everything continues as is, some aspects of what is to come may resemble the dystopia depicted in The Last of Us. No zombies, but emerging and reemerging fungi in a chaotic distribution, and resistant to all established treatments.
“The risk factors of patients and their immune status, combined with the behavior of mycoses, bring a complicated scenario. But therapeutic failure resulting from multidrug resistance to antifungals could make it catastrophic,” Dr. Sánchez Paredes summarized.
At the moment, there are only four families of drugs capable of counteracting fungal infections — and as mentioned, some are already scarce in Latin America’s hospital pharmacies.
“Historically, fungal infections have been given less importance than those caused by viruses or bacteria. Even in some developed countries, the true extent of morbidity and mortality they present is unknown. This results in less investment in the development of new antifungal molecules because knowledge is lacking about the incidence and prevalence of these diseases,” Dr. Treviño Rangel pointed out.
He added that the main limitation for the development of new drugs is economic. “Unfortunately, not many pharmaceutical companies are willing to invest in the development of new antifungals, and there are no government programs specifically promoting and supporting research into new therapeutic options against these neglected diseases,” he asserted.
Development of vaccines to prevent fungal infections faces the same barriers. Although, according to Dr. Treviño Rangel, the difficulties are compounded by the great similarity between fungal cells and human cells. This makes it possible for harmful cross-reactivity to occur. In addition, because most severe fungal infections occur in individuals with immunosuppression, a vaccine would need to trigger an adequate immune response despite this issue.
Meanwhile, fungi quietly continue to do what they do best: resist and survive. For millions of years, they have mutated and adapted to new environments. Some theories even blame them for the extinction of dinosaurs and the subsequent rise of mammals. They exist on the edge of life and death, decomposing and creating. There is consensus that at the moment, it does not seem feasible for them to generate a pandemic like the one due to SARS-CoV-2, given their transmission mechanism. But who is willing to rule out that this may not happen in the long or medium term?
Dr. Sánchez Paredes, Dr. Treviño Rangel, Dr. Messina, Dr. Santiso, Dr. Álvarez, and Dr. Segundo Zaragoza have declared no relevant financial conflicts of interest.
This story was translated from Medscape Spanish Edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
The principle of resilience and survival is crucial for medically significant fungi. These microorganisms are far from creating the postapocalyptic scenario depicted in TV series like The Last of Us, and much work is necessary to learn more about them. Accurate statistics on fungal infections, accompanied by clinical histories, simple laboratory tests, new antifungals, and a necessary One Health approach are lacking.
The entomopathogenic fungus Ophiocordyceps unilateralis was made notorious by the TV series, but for now, it only manages to control the brains of some ants at will. Luckily, there are no signs that fungi affecting humans are inclined to create zombies.
What is clear is that the world belongs to the kingdom of fungi and that fungi are everywhere. There are already close to 150,000 described species, but millions remain to be discovered. They abound in decomposing organic matter, soil, or animal excrement, including that of bats and pigeons. Some fungi have even managed to find a home in hospitals. Lastly, we must not forget those that establish themselves in the human microbiome.
Given such diversity, it is legitimate to ask whether any of them could be capable of generating new pandemics. Could the forgotten Cryptococcus neoformans, Aspergillus fumigatus, or Histoplasma species, among others, trigger new health emergencies on the scale of the one generated by SARS-CoV-2?
We cannot forget that a coronavirus has already confirmed that reality can surpass fiction. However, Edith Sánchez Paredes, a biologist, doctor in biomedical sciences, and specialist in medical mycology, provided a reassuring response to Medscape Spanish Edition on this point.
“That would be very difficult to see because the way fungal infections are acquired is not from person to person, in most cases,” said Dr. Sánchez Paredes, from the Mycology Unit of the Faculty of Medicine at the National Autonomous University of Mexico.
Close to 300 species have already been classified as pathogenic in humans. Although the numbers are not precise and are increasing, it is estimated that around 1,500,000 people worldwide die each year of systemic fungal infections.
“However, it is important to emphasize that establishment of an infection depends not only on the causal agent. A crucial factor is the host, in this case, the human. Generally, these types of infections will develop in individuals with some deficiency in their immune system. The more deficient the immune response, the more likely a fungal infection may occur,” stated Dr. Sánchez Paredes.
The possibility of a pandemic like the one experienced with SARS-CoV-2 in the short term is remote, but the threat posed by fungal infections persists.
In 2022, the World Health Organization (WHO) defined a priority list of pathogenic fungi, with the aim of guiding actions to control them. It is mentioned there that invasive fungal diseases are on the rise worldwide, particularly in immunocompromised populations.
“Despite the growing concern, fungal infections receive very little attention and resources, leading to a paucity of quality data on fungal disease distribution and antifungal resistance patterns. Consequently, it is impossible to estimate their exact burden,” as stated in the document.
In line with this, an article published in Mycoses in 2022 concluded that fungal infections are neglected diseases in Latin America. Among other difficulties, deficiencies in access to tests such as polymerase chain reaction or serum detection of beta-1,3-D-glucan have been reported there.
In terms of treatments, most countries encounter problems with access to liposomal amphotericin B and new azoles, such as posaconazole and isavuconazole.
“Unfortunately, in Latin America, we suffer from a poor infrastructure for diagnosing fungal infections; likewise, we have limited access to antifungals available in the global market. What’s more, we lack reliable data on the epidemiology of fungal infections in the region, so many times governments are unaware of the true extent of the problem,” said Rogelio de Jesús Treviño Rangel, PhD, a medical microbiologist and expert in clinical mycology, professor, and researcher at the Faculty of Medicine of the Autonomous University of Nuevo León in Mexico.
Need for More Medical Mycology Training
Dr. Fernando Messina is a medical mycologist with the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital in Buenos Aires, Argentina. He has noted an increase in the number of cases of cryptococcosis, histoplasmosis, and aspergillosis in his daily practice.
“Particularly, pulmonary aspergillosis is steadily increasing. This is because many patients have structural lung alterations that favor the appearance of this mycosis. This is related to the increase in cases of tuberculosis and the rise in life expectancy of patients with chronic obstructive pulmonary disease or other pulmonary or systemic diseases,” Dr. Messina stated.
For Dr. Messina, the main obstacle in current clinical practice is the low level of awareness among nonspecialist physicians regarding the presence of systemic fungal infections, and because these infections are more common than realized, it is vital to consider fungal etiology before starting empirical antibiotic therapy.
“Health professionals usually do not think about mycoses because mycology occupies a very small space in medical education at universities. As the Venezuelan mycologist Gioconda Cunto de San Blas once said, ‘Mycology is the Cinderella of microbiology.’ To change this, we need to give more space to mycoses in undergraduate and postgraduate studies,” Dr. Messina asserted.
He added, “The main challenge is to train professionals with an emphasis on the clinical interpretation of cases. Current medicine has a strong trend toward molecular biology and the use of rapid diagnostic methods, without considering the clinical symptoms or the patient’s history. Determinations are very useful, but it is necessary to interpret the results.”
Dr. Messina sees it as unlikely in the short term for a pandemic to be caused by fungi, but if it were to occur, he believes it would happen in healthcare systems in regions that are not prepared in terms of infrastructure. However, as seen in the health emergency resulting from SARS-CoV-2, he thinks the impact would be mitigated by the performance of healthcare professionals.
“In general, we have the ability to adapt to any adverse situation or change — although it is clear that we need more doctors, biochemists, and microbiologists trained in mycology,” emphasized Dr. Messina.
More than 40 interns pass through Muñiz Hospital each year. They are doctors and biochemists from Argentina, other countries in the region, or even Europe, seeking to enhance their training in mycology. Regarding fungal infection laboratory work, the interest lies in learning to use traditional techniques and innovative molecular methods.
“Rapid diagnostic methods, especially the detection of circulating antigens, have marked a change in the prognosis of deep mycosis in immunocompromised hosts. The possibility of screening and monitoring in this group of patients is very important and has a great benefit,” said Gabriela Santiso, PhD, a biochemist and head of the Mycology Unit of the Francisco Javier Muñiz Infectious Diseases Hospital.
According to Dr. Santiso, the current landscape includes the ability to identify genus and species, which can help in understanding resistance to antifungals. Furthermore, conducting sensitivity tests to these drugs, using standardized commercial methods, also provides timely information for treatment.
But Dr. Santiso warns that Latin America is a vast region with great disparity in human and technological resources. Although most countries in the region have networks facilitating access to timely diagnosis, resources are generally more available in major urban centers.
This often clashes with the epidemiology of most fungal infections. “Let’s not forget that many fungal pathologies affect low-income people who have difficulties accessing health centers, which sometimes turns them into chronic diseases that are hard to treat,” Dr. Santiso pointed out.
In mycology laboratories, the biggest cost is incurred by new diagnostic tests, such as those allowing molecular identification. Conventional methods are not usually expensive, but they require time and effort to train human resources to handle them.
Because new methodologies are not always available or easily accessible throughout the region, Dr. Santiso recommended not neglecting traditional mycological techniques. “Molecular methods, rapid diagnostic methods, and conventional mycology techniques are complementary and not mutually exclusive tests. Continuous training and updating are needed in this area,” she emphasized.
Why Are Resistant Fungal Infections Becoming Increasingly Common?
The first barrier for fungi to cause infection in humans is body temperature; most of them cannot withstand 37 °C. However, they also struggle to evade the immune response that is activated when they try to enter the body.
“We are normally exposed to many of these fungi, almost all the time, but if our immune system is adequate, it may not go beyond a mild infection, in most cases subclinical, which will resolve quickly,” Dr. Sánchez Paredes stated.
However, according to Dr. Sánchez Paredes, if the immune response is weak, “the fungus will have no trouble establishing itself in our organs. Some are even part of our microbiota, such as Candida albicans, which in the face of an imbalance or immunocompromise, can lead to serious infections.”
It is clear that the population at risk for immunosuppression has increased. According to the WHO, this is due to the high prevalence of such diseases as tuberculosis, cancer, and HIV infection, among others.
But the WHO also believes that the increase in fungal infections is related to greater population access to critical care units, invasive procedures, chemotherapy, or immunotherapy treatments.
Furthermore, factors related to the fungus itself and the environment play a role. “These organisms have enzymes, proteins, and other molecules that allow them to survive in the environment in which they normally inhabit. When they face a new and stressful one, they must express other molecules that will allow them to survive. All of this helps them evade elements of the immune system, antifungals, and, of course, body temperature,” according to Dr. Sánchez Paredes.
It is possible that climate change is also behind the noticeable increase in fungal infections and that this crisis may have an even greater impact in the future. The temperature of the environment has increased, and fungi will have to adapt to the planet’s temperature, to the point where body temperature may no longer be a significant barrier for them.
Environmental changes would also be responsible for modifications in the distribution of endemic mycoses, and it is believed that fungi will more frequently find new ecological niches, be able to survive in other environments, and alter distribution zones.
This is what is happening between Mexico and the United States with coccidioidomycosis, or valley fever. “We will begin to see cases of some mycoses where they were not normally seen, so we will have to conduct more studies to confirm that the fungus is inhabiting these new areas or is simply appearing in new sites owing to migration and the great mobility of populations,” Dr. Sánchez Paredes said.
Finally, exposure to environmental factors would partly be responsible for the increasing resistance to first-line antifungals observed in these microorganisms. This seems to be the case with A. fumigatus when exposed to azoles used as fungicides in agriculture.
One Health in Fungal Infections
The increasing resistance to antifungals is a clear testament that human, animal, and environmental health are interconnected. This is why a multidisciplinary approach that adopts the perspective of One Health is necessary for its management.
“The use of fungicides in agriculture, structurally similar to the azoles used in clinics, generates resistance in Aspergillus fumigatus found in the environment. These fungi in humans can be associated with infections that do not respond to first-line treatment,” explained Carlos Arturo Álvarez, an infectious diseases physician and professor at the Faculty of Medicine at the National University of Colombia.
According to Dr. Álvarez, the approach to control them should not only focus on the search for diagnostic methods that allow early detection of antifungal resistance or research on new antifungal treatments. He believes that progress must also be made with strategies that allow for the proper use of antifungals in agriculture.
“Unfortunately, the One Health approach is not yet well implemented in the region, and in my view, there is a lack of articulation in the different sectors. That is, there is a need for true coordination between government offices of agriculture, animal and human health, academia, and international organizations. This is not happening yet, and I believe we are in the initial stage of visibility,” Dr. Álvarez opined.
Veterinary public health is another pillar of the aforementioned approach. For various reasons, animals experience a higher frequency of fungal infections. A few carry and transmit true zoonoses that affect human health, but most often, animals act only as sentinels indicating a potential source of transmission.
Carolina Segundo Zaragoza, PhD, has worked in veterinary mycology for 30 years. She currently heads the veterinary mycology laboratory at the Animal Production Teaching, Research, and Extension Center in Altiplano, under the Faculty of Veterinary Medicine and Animal Husbandry at the National Autonomous University of Mexico. Because she has frequent contact with specialists in human mycology, during her professional career she has received several patient consultations, most of which were for cutaneous mycoses.
“They detect some dermatomycosis and realize that the common factor is owning a companion animal or a production animal with which the patient has contact. Both animals and humans present the same type of lesions, and then comes the question: Who infected whom? I remind them that the main source of infection is the soil and that animals should not be blamed in the first instance,” Dr. Segundo Zaragoza clarified.
She is currently collaborating on a research project analyzing the presence of Coccidioides immitis in the soil. This pathogen is responsible for coccidioidomycosis in dogs and humans, and she sees with satisfaction how these types of initiatives, which include some components of the One Health vision, are becoming more common in Mexico.
“Fortunately, human mycologists are increasingly providing more space for the dissemination of veterinary mycology. So I have had the opportunity to be invited to different forums on medical mycology to present the clinical cases we can have in animals and talk about the research projects we carry out. I have more and more opportunities to conduct joint research with human mycologists and veterinary doctors,” she said.
Dr. Segundo Zaragoza believes that to better implement the One Health vision, standardizing the criteria for detecting, diagnosing, and treating mycoses is necessary. She considers that teamwork will be key to achieving the common goal of safeguarding the well-being and health of humans and animals.
Alarms Sound for Candida auris
The WHO included the yeast Candida auris in its group of pathogens with critical priority, and since 2009, it has raised alarm owing to the ease with which it grows in hospitals. In that setting, C auris is known for its high transmissibility, its ability to cause outbreaks, and the high mortality rate from disseminated infections.
“It has been a concern for the mycological community because it shows resistance to most antifungals used clinically, mainly azoles, but also for causing epidemic outbreaks,” emphasized Dr. Sánchez Paredes.
Its mode of transmission is not very clear, but it has been documented to be present on the skin and persist in hospital materials and furniture. It causes nosocomial infections in critically ill patients, such as those in intensive care, and those with cancer or who have received a transplant.
Risk factors for its development include renal insufficiency, hospital stays of more than 15 days, mechanical ventilation, central lines, use of parenteral nutrition, and presence of sepsis.
As for other mycoses, there are no precise studies reporting global incidence rates, but the trend indicates an increase in the detection of outbreaks in various countries lately — something that began to be visible during the COVID-19 pandemic.
In Mexico, Dr. Treviño Rangel and colleagues from Nuevo León reported the first case of candidemia caused by this agent. It occurred in May 2020 and involved a 58-year-old woman with a history of severe endometriosis and multiple complications in the gastrointestinal tract. The patient’s condition improved favorably thanks to antifungal therapy with caspofungin and liposomal amphotericin B.
However, 3 months after that episode, the group reported an outbreak of C. auris at the same hospital in 12 critically ill patients co-infected with SARS-CoV-2. All were on mechanical ventilation, had peripherally inserted central catheters and urinary catheters, and had a prolonged hospital stay (20-70 days). The mortality in patients with candidemia in this cohort was 83.3%.
Open Ending
As seen in some science fiction series, fungal infections in the region still have an open ending, and Global Action For Fungal Infections (GAFFI) has estimated that with better diagnostics and treatments, deaths caused by fungi could decrease to less than 750,000 per year worldwide.
But if everything continues as is, some aspects of what is to come may resemble the dystopia depicted in The Last of Us. No zombies, but emerging and reemerging fungi in a chaotic distribution, and resistant to all established treatments.
“The risk factors of patients and their immune status, combined with the behavior of mycoses, bring a complicated scenario. But therapeutic failure resulting from multidrug resistance to antifungals could make it catastrophic,” Dr. Sánchez Paredes summarized.
At the moment, there are only four families of drugs capable of counteracting fungal infections — and as mentioned, some are already scarce in Latin America’s hospital pharmacies.
“Historically, fungal infections have been given less importance than those caused by viruses or bacteria. Even in some developed countries, the true extent of morbidity and mortality they present is unknown. This results in less investment in the development of new antifungal molecules because knowledge is lacking about the incidence and prevalence of these diseases,” Dr. Treviño Rangel pointed out.
He added that the main limitation for the development of new drugs is economic. “Unfortunately, not many pharmaceutical companies are willing to invest in the development of new antifungals, and there are no government programs specifically promoting and supporting research into new therapeutic options against these neglected diseases,” he asserted.
Development of vaccines to prevent fungal infections faces the same barriers. Although, according to Dr. Treviño Rangel, the difficulties are compounded by the great similarity between fungal cells and human cells. This makes it possible for harmful cross-reactivity to occur. In addition, because most severe fungal infections occur in individuals with immunosuppression, a vaccine would need to trigger an adequate immune response despite this issue.
Meanwhile, fungi quietly continue to do what they do best: resist and survive. For millions of years, they have mutated and adapted to new environments. Some theories even blame them for the extinction of dinosaurs and the subsequent rise of mammals. They exist on the edge of life and death, decomposing and creating. There is consensus that at the moment, it does not seem feasible for them to generate a pandemic like the one due to SARS-CoV-2, given their transmission mechanism. But who is willing to rule out that this may not happen in the long or medium term?
Dr. Sánchez Paredes, Dr. Treviño Rangel, Dr. Messina, Dr. Santiso, Dr. Álvarez, and Dr. Segundo Zaragoza have declared no relevant financial conflicts of interest.
This story was translated from Medscape Spanish Edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Are You Unwittingly Aiding the Rise of Superfungi?
Unnecessary or incorrect use of topical antifungal medications is driving the spread of fungal infections like ringworm, which are becoming more difficult to treat, according to a January 11 study published in Morbidity and Mortality Weekly Report.
If a patient’s condition is not caused by a fungus but is treated as such, treatment will be ineffective.
such as clotrimazole or combinations of antifungals and corticosteroids. And because many topical treatments are also available over-the-counter, doctors should advise patients about how to use them correctly.
“In the last few years, there have been many antifungal resistant cases of tinea corporisand onychomycosisreported,” or ringworm and finger or toenail infections, respectively, said Shari Lipner, MD, PhD, a dermatologist at Weill Cornell Medicine in New York, and an author of the study.
Many of these cases originated in South Asia and have also been reported in Europe and Canada. In 2023, the first cases of a new strain of antifungal-resistant ringworm were reported in the United States. This species, Trichophyton indotineae, does not respond to topical medications, requiring oral treatment instead.
“It’s really a serious problem and a huge public health concern,” Dr. Lipner said.
For the new study, Dr. Lipner and colleagues examined prescription patterns from 2021 Medicare Part D claims of topical antifungals. They report that 6.5 million topical antifungal prescriptions were filled that year, some of which included steroids in the formulation. Primary care clinicians wrote 40% of these prescriptions, the most for any clinician group. The estimate is almost certainly an undercount of topical antifungal use because the database did not include over-the-counter purchases or data from other insurance payers.
The number of prescriptions equate to 1 in every 8 Medicare Part D beneficiary receiving an antifungal, the researchers reported.
“If I think about the patients that come into my office, I’m certainly not giving an antifungal to 1 in 8 of them, and I see a lot of fungal infections,” Dr. Lipner said. The findings suggest to Dr. Lipner that some clinicians are diagnosing ringworm by eyesight alone rather than confirming the diagnosis with techniques such as microscopy, fungal culture testing, or polymerase chain reaction testing.
Sometimes what looks like ringworm may actually be eczema, in which case, the topical antifungal would not be appropriate, according to Avrom Caplan, MD, a dermatologist at NYU Langone Health in New York.
“If you’re prescribing something to somebody that they don’t need, you’re basically exposing them to the side effects without the benefit,” Dr. Caplan, who was not part of the study, said.
Dr. Caplan, who reported the first cases of ringworm that only responded to oral medications in the United States, stressed that topical treatments work fine for many ringworm cases today. But if indiscriminate prescribing spurs the development of more resilient fungi, more situations may arise in which only oral medications work in the future, Dr. Caplan said. In addition, oral medications are inherently more demanding on a patient than something they can rub on their skin, Dr. Caplan added.
“We hope that physicians will really think hard about this study and change their practices if they’re not confirming the diagnosis,” Dr. Lipner said.
Dr. Lipner and Dr. Caplan report no relevant financial relationships.
A version of this article appeared on Medscape.com.
Unnecessary or incorrect use of topical antifungal medications is driving the spread of fungal infections like ringworm, which are becoming more difficult to treat, according to a January 11 study published in Morbidity and Mortality Weekly Report.
If a patient’s condition is not caused by a fungus but is treated as such, treatment will be ineffective.
such as clotrimazole or combinations of antifungals and corticosteroids. And because many topical treatments are also available over-the-counter, doctors should advise patients about how to use them correctly.
“In the last few years, there have been many antifungal resistant cases of tinea corporisand onychomycosisreported,” or ringworm and finger or toenail infections, respectively, said Shari Lipner, MD, PhD, a dermatologist at Weill Cornell Medicine in New York, and an author of the study.
Many of these cases originated in South Asia and have also been reported in Europe and Canada. In 2023, the first cases of a new strain of antifungal-resistant ringworm were reported in the United States. This species, Trichophyton indotineae, does not respond to topical medications, requiring oral treatment instead.
“It’s really a serious problem and a huge public health concern,” Dr. Lipner said.
For the new study, Dr. Lipner and colleagues examined prescription patterns from 2021 Medicare Part D claims of topical antifungals. They report that 6.5 million topical antifungal prescriptions were filled that year, some of which included steroids in the formulation. Primary care clinicians wrote 40% of these prescriptions, the most for any clinician group. The estimate is almost certainly an undercount of topical antifungal use because the database did not include over-the-counter purchases or data from other insurance payers.
The number of prescriptions equate to 1 in every 8 Medicare Part D beneficiary receiving an antifungal, the researchers reported.
“If I think about the patients that come into my office, I’m certainly not giving an antifungal to 1 in 8 of them, and I see a lot of fungal infections,” Dr. Lipner said. The findings suggest to Dr. Lipner that some clinicians are diagnosing ringworm by eyesight alone rather than confirming the diagnosis with techniques such as microscopy, fungal culture testing, or polymerase chain reaction testing.
Sometimes what looks like ringworm may actually be eczema, in which case, the topical antifungal would not be appropriate, according to Avrom Caplan, MD, a dermatologist at NYU Langone Health in New York.
“If you’re prescribing something to somebody that they don’t need, you’re basically exposing them to the side effects without the benefit,” Dr. Caplan, who was not part of the study, said.
Dr. Caplan, who reported the first cases of ringworm that only responded to oral medications in the United States, stressed that topical treatments work fine for many ringworm cases today. But if indiscriminate prescribing spurs the development of more resilient fungi, more situations may arise in which only oral medications work in the future, Dr. Caplan said. In addition, oral medications are inherently more demanding on a patient than something they can rub on their skin, Dr. Caplan added.
“We hope that physicians will really think hard about this study and change their practices if they’re not confirming the diagnosis,” Dr. Lipner said.
Dr. Lipner and Dr. Caplan report no relevant financial relationships.
A version of this article appeared on Medscape.com.
Unnecessary or incorrect use of topical antifungal medications is driving the spread of fungal infections like ringworm, which are becoming more difficult to treat, according to a January 11 study published in Morbidity and Mortality Weekly Report.
If a patient’s condition is not caused by a fungus but is treated as such, treatment will be ineffective.
such as clotrimazole or combinations of antifungals and corticosteroids. And because many topical treatments are also available over-the-counter, doctors should advise patients about how to use them correctly.
“In the last few years, there have been many antifungal resistant cases of tinea corporisand onychomycosisreported,” or ringworm and finger or toenail infections, respectively, said Shari Lipner, MD, PhD, a dermatologist at Weill Cornell Medicine in New York, and an author of the study.
Many of these cases originated in South Asia and have also been reported in Europe and Canada. In 2023, the first cases of a new strain of antifungal-resistant ringworm were reported in the United States. This species, Trichophyton indotineae, does not respond to topical medications, requiring oral treatment instead.
“It’s really a serious problem and a huge public health concern,” Dr. Lipner said.
For the new study, Dr. Lipner and colleagues examined prescription patterns from 2021 Medicare Part D claims of topical antifungals. They report that 6.5 million topical antifungal prescriptions were filled that year, some of which included steroids in the formulation. Primary care clinicians wrote 40% of these prescriptions, the most for any clinician group. The estimate is almost certainly an undercount of topical antifungal use because the database did not include over-the-counter purchases or data from other insurance payers.
The number of prescriptions equate to 1 in every 8 Medicare Part D beneficiary receiving an antifungal, the researchers reported.
“If I think about the patients that come into my office, I’m certainly not giving an antifungal to 1 in 8 of them, and I see a lot of fungal infections,” Dr. Lipner said. The findings suggest to Dr. Lipner that some clinicians are diagnosing ringworm by eyesight alone rather than confirming the diagnosis with techniques such as microscopy, fungal culture testing, or polymerase chain reaction testing.
Sometimes what looks like ringworm may actually be eczema, in which case, the topical antifungal would not be appropriate, according to Avrom Caplan, MD, a dermatologist at NYU Langone Health in New York.
“If you’re prescribing something to somebody that they don’t need, you’re basically exposing them to the side effects without the benefit,” Dr. Caplan, who was not part of the study, said.
Dr. Caplan, who reported the first cases of ringworm that only responded to oral medications in the United States, stressed that topical treatments work fine for many ringworm cases today. But if indiscriminate prescribing spurs the development of more resilient fungi, more situations may arise in which only oral medications work in the future, Dr. Caplan said. In addition, oral medications are inherently more demanding on a patient than something they can rub on their skin, Dr. Caplan added.
“We hope that physicians will really think hard about this study and change their practices if they’re not confirming the diagnosis,” Dr. Lipner said.
Dr. Lipner and Dr. Caplan report no relevant financial relationships.
A version of this article appeared on Medscape.com.
FDA passes on olorofim despite critical need for antifungals
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration is declining to approve the investigational antifungal olorofim and is asking for more data, according to a news release from the manufacturer, F2G.
Olorofim, (formerly known as F901318) is the first in the orotomide class of antifungals to be evaluated clinically for the treatment of invasive mold infections. Its maker, F2G, is a biotech company based in Manchester, England, that focuses on developing drugs for rare fungal diseases.
The company says it remains optimistic and will address the FDA’s requirements and continue to seek approval.
The FDA’s denial comes as fungal infections are becoming increasingly common and resistant to treatment. There are only four antifungal classes currently available, and there are few new candidates in the pipeline. No new classes of antifungals have been developed in 2 decades.
David Andes, MD, chief of the division of infectious diseases at the University of Wisconsin–Madison, told this news organization he shares the hope that the company can meet the requirements to gain approval.
“Some of the early results were really exciting,” he said. “People are enthusiastic about the compound because it has a novel mechanism of action, and it is active against a group of fungi that we have limited to no options for.”
Early results ‘exciting’
Dr. Andes said several physicians have been able to prescribe olorofim under the compassionate use program “and have witnessed success.”
Olorofim is the first antifungal agent to be granted breakthrough therapy designation, which the FDA granted in November 2019 for the treatment of invasive mold infections for patients with limited or no treatment options, including patients with refractory aspergillosis or those who are intolerant of currently available therapy. It is also indicated for infections due to Lomentospora prolificans, Scedosporium, and Scopulariopsis species.
Olorofim received a second breakthrough therapy designation in October 2020. The second designation was granted for treatment of central nervous system coccidioidomycosis that is refractory or for cases that cannot be treated with standard-of-care therapy.
It is very difficult for patients to be approved to receive compassionate use medicines, Dr. Andes pointed out. “I’d like to have access sooner rather than later,” he added.
Dr. Andes says the drugs are expensive and are time consuming to produce. And with antifungals, it is difficult to demonstrate safety in comparison with other antimicrobial agents because “it’s hard to hurt a fungus without having toxicity with human cells.”
Complete response letter issued
F2G received a complete response letter from the FDA regarding its new drug application for olorofim, according to the news release issued by the company. “While F2G is disappointed with this outcome, we remain optimistic about olorofim’s potential to address an unmet need for patients with invasive fungal infections who have exhausted their treatment alternatives,” Francesco Maria Lavino, chief executive officer, said in the release. “We are assessing the details of the Complete Response Letter, and we plan to meet with the FDA to discuss it further.”
Dr. Andes says few other antifungals have made it as far as olorofim in clinical trials.
Lance B. Price, PhD, codirector of the Antibiotic Resistance Action Center at George Washington University in Washington, told this news organization that despite the lack of antifungals in the pipeline, “We can’t allow our desperation to override the checkpoints that ensure that antifungals are safe to use in people.”
In the meantime, he said, it is important to preserve the utility of current antifungals by avoiding overusing them in medicine and agriculture.
“Sadly,” he said, “a drug called ipflufenoquin, which works by a similar mode of action as olorofim, has already been approved by the U.S. Environmental Protection Agency for use in plant agriculture. This could weaken the effectiveness of olorofim for treating things like Aspergillus infections even before the drug has been approved for use in humans.”
Plant drug undermining olorofim efficacy in humans
“While I’m sure this makes financial sense for the makers of ipflufenoquin, it borders on insanity from a public health perspective,” Dr. Price said.
Meanwhile, the global threat of fungal infections grows. The World Health Organization has launched its first-ever list of health-threatening fungi. Authors of a WHO report that contains the list write, “The invasive forms of these fungal infections often affect severely ill patients and those with significant underlying immune system–related conditions.”
F2G will continue to expand olorofim’s clinical trial program, according to the company’s statement. Along with its partner, Shionogi, it is enrolling patients with proven or probable invasive aspergillosis in a global phase 3 trial (OASIS), which will compare outcomes after treatment with olorofim in comparison with amphotericin B liposome (AmBisome) followed by standard of care.
A version of this article first appeared on Medscape.com.
Pound of flesh buys less prison time
Pound of flesh buys less prison time
We should all have more Shakespeare in our lives. Yeah, yeah, Shakespeare is meant to be played, not read, and it can be a struggle to herd teenagers through the Bard’s interesting and bloody tragedies, but even a perfunctory reading of “The Merchant of Venice” would hopefully have prevented the dystopian nightmare Massachusetts has presented us with today.
The United States has a massive shortage of donor organs. This is an unfortunate truth. So, to combat this issue, a pair of Massachusetts congresspeople have proposed HD 3822, which would allow prisoners to donate organs and/or bone marrow (a pound of flesh, so to speak) in exchange for up to a year in reduced prison time. Yes, that’s right. Give up pieces of yourself and the state of Massachusetts will deign to reduce your long prison sentence.
Oh, and before you dismiss this as typical Republican antics, the bill was sponsored by two Democrats, and in a statement one of them hoped to address racial disparities in organ donation, as people of color are much less likely to receive organs. Never mind that Black people are imprisoned at a much higher rate than Whites.
Yeah, this whole thing is what people in the business like to call an ethical disaster.
Fortunately, the bill will likely never be passed and it’s probably illegal anyway. A federal law from 1984 (how’s that for a coincidence) prevents people from donating organs for use in human transplantation in exchange for “valuable consideration.” In other words, you can’t sell your organs for profit, and in this case, reducing prison time would probably count as valuable consideration in the eyes of the courts.
Oh, and in case you’ve never read Merchant of Venice, Shylock, the character looking for the pound of flesh as payment for a debt? He’s the villain. In fact, it’s pretty safe to say that anyone looking to extract payment from human dismemberment is probably the bad guy of the story. Apparently that wasn’t clear.
How do you stop a fungi? With a deadly guy
Thanks to the new HBO series “The Last of Us,” there’s been a lot of talk about the upcoming fungi-pocalypse, as the show depicts the real-life “zombie fungus” Cordyceps turning humans into, you know, zombies.
No need to worry, ladies and gentleman, because science has discovered a way to turn back the fungal horde. A heroic, and environmentally friendly, alternative to chemical pesticides “in the fight against resistant fungi [that] are now resistant to antimycotics – partly because they are used in large quantities in agricultural fields,” investigators at the Leibniz Institute for Natural Product Research and Infection Biology in Jena, Germany, said in a written statement.
We are, of course, talking about Keanu Reeves. Wait a second. He’s not even in “The Last of Us.” Sorry folks, we are being told that it really is Keanu Reeves. Our champion in the inevitable fungal pandemic is movie star Keanu Reeves. Sort of. It’s actually keanumycin, a substance produced by bacteria of the genus Pseudomonas.
Really? Keanumycin? “The lipopeptides kill so efficiently that we named them after Keanu Reeves because he, too, is extremely deadly in his roles,” lead author Sebastian Götze, PhD, explained.
Dr. Götze and his associates had been working with pseudomonads for quite a while before they were able to isolate the toxins responsible for their ability to kill amoebae, which resemble fungi in some characteristics. When then finally tried the keanumycin against gray mold rot on hydrangea leaves, the intensely contemplative star of “The Matrix” and “John Wick” – sorry, wrong Keanu – the bacterial derivative significantly inhibited growth of the fungus, they said.
Additional testing has shown that keanumycin is not highly toxic to human cells and is effective against fungi such as Candida albicans in very low concentrations, which makes it a good candidate for future pharmaceutical development.
To that news there can be only one response from the substance’s namesake.
High fat, bye parasites
Fat. Fat. Fat. Seems like everyone is trying to avoid it these days, but fat may be good thing when it comes to weaseling out a parasite.
The parasite in this case is the whipworm, aka Trichuris trichiura. You can find this guy in the intestines of millions of people, where it causes long-lasting infections. Yikes … Researchers have found that the plan of attack to get rid of this invasive species is to boost the immune system, but instead of vitamin C and zinc it’s fat they’re pumping in. Yes, fat.
The developing countries with poor sewage that are at the highest risk for contracting parasites such as this also are among those where people ingest cheaper diets that are generally higher in fat. The investigators were interested to see how a high-fat diet would affect immune responses to the whipworms.
And, as with almost everything else, the researchers turned to mice, which were introduced to a closely related species, Trichuris muris.
A high-fat diet, rather than obesity itself, increases a molecule on T-helper cells called ST2, and this allows an increased T-helper 2 response, effectively giving eviction notices to the parasites in the intestinal lining.
To say the least, the researchers were surprised since “high-fat diets are mostly associated with increased pathology during disease,” said senior author Richard Grencis, PhD, of the University of Manchester (England), who noted that ST2 is not normally triggered with a standard diet in mice but the high-fat diet gave it a boost and an “alternate pathway” out.
Now before you start ordering extra-large fries at the drive-through to keep the whipworms away, the researchers added that they “have previously published that weight loss can aid the expulsion of a different gut parasite worm.” Figures.
Once again, though, signs are pointing to the gut for improved health.
Pound of flesh buys less prison time
We should all have more Shakespeare in our lives. Yeah, yeah, Shakespeare is meant to be played, not read, and it can be a struggle to herd teenagers through the Bard’s interesting and bloody tragedies, but even a perfunctory reading of “The Merchant of Venice” would hopefully have prevented the dystopian nightmare Massachusetts has presented us with today.
The United States has a massive shortage of donor organs. This is an unfortunate truth. So, to combat this issue, a pair of Massachusetts congresspeople have proposed HD 3822, which would allow prisoners to donate organs and/or bone marrow (a pound of flesh, so to speak) in exchange for up to a year in reduced prison time. Yes, that’s right. Give up pieces of yourself and the state of Massachusetts will deign to reduce your long prison sentence.
Oh, and before you dismiss this as typical Republican antics, the bill was sponsored by two Democrats, and in a statement one of them hoped to address racial disparities in organ donation, as people of color are much less likely to receive organs. Never mind that Black people are imprisoned at a much higher rate than Whites.
Yeah, this whole thing is what people in the business like to call an ethical disaster.
Fortunately, the bill will likely never be passed and it’s probably illegal anyway. A federal law from 1984 (how’s that for a coincidence) prevents people from donating organs for use in human transplantation in exchange for “valuable consideration.” In other words, you can’t sell your organs for profit, and in this case, reducing prison time would probably count as valuable consideration in the eyes of the courts.
Oh, and in case you’ve never read Merchant of Venice, Shylock, the character looking for the pound of flesh as payment for a debt? He’s the villain. In fact, it’s pretty safe to say that anyone looking to extract payment from human dismemberment is probably the bad guy of the story. Apparently that wasn’t clear.
How do you stop a fungi? With a deadly guy
Thanks to the new HBO series “The Last of Us,” there’s been a lot of talk about the upcoming fungi-pocalypse, as the show depicts the real-life “zombie fungus” Cordyceps turning humans into, you know, zombies.
No need to worry, ladies and gentleman, because science has discovered a way to turn back the fungal horde. A heroic, and environmentally friendly, alternative to chemical pesticides “in the fight against resistant fungi [that] are now resistant to antimycotics – partly because they are used in large quantities in agricultural fields,” investigators at the Leibniz Institute for Natural Product Research and Infection Biology in Jena, Germany, said in a written statement.
We are, of course, talking about Keanu Reeves. Wait a second. He’s not even in “The Last of Us.” Sorry folks, we are being told that it really is Keanu Reeves. Our champion in the inevitable fungal pandemic is movie star Keanu Reeves. Sort of. It’s actually keanumycin, a substance produced by bacteria of the genus Pseudomonas.
Really? Keanumycin? “The lipopeptides kill so efficiently that we named them after Keanu Reeves because he, too, is extremely deadly in his roles,” lead author Sebastian Götze, PhD, explained.
Dr. Götze and his associates had been working with pseudomonads for quite a while before they were able to isolate the toxins responsible for their ability to kill amoebae, which resemble fungi in some characteristics. When then finally tried the keanumycin against gray mold rot on hydrangea leaves, the intensely contemplative star of “The Matrix” and “John Wick” – sorry, wrong Keanu – the bacterial derivative significantly inhibited growth of the fungus, they said.
Additional testing has shown that keanumycin is not highly toxic to human cells and is effective against fungi such as Candida albicans in very low concentrations, which makes it a good candidate for future pharmaceutical development.
To that news there can be only one response from the substance’s namesake.
High fat, bye parasites
Fat. Fat. Fat. Seems like everyone is trying to avoid it these days, but fat may be good thing when it comes to weaseling out a parasite.
The parasite in this case is the whipworm, aka Trichuris trichiura. You can find this guy in the intestines of millions of people, where it causes long-lasting infections. Yikes … Researchers have found that the plan of attack to get rid of this invasive species is to boost the immune system, but instead of vitamin C and zinc it’s fat they’re pumping in. Yes, fat.
The developing countries with poor sewage that are at the highest risk for contracting parasites such as this also are among those where people ingest cheaper diets that are generally higher in fat. The investigators were interested to see how a high-fat diet would affect immune responses to the whipworms.
And, as with almost everything else, the researchers turned to mice, which were introduced to a closely related species, Trichuris muris.
A high-fat diet, rather than obesity itself, increases a molecule on T-helper cells called ST2, and this allows an increased T-helper 2 response, effectively giving eviction notices to the parasites in the intestinal lining.
To say the least, the researchers were surprised since “high-fat diets are mostly associated with increased pathology during disease,” said senior author Richard Grencis, PhD, of the University of Manchester (England), who noted that ST2 is not normally triggered with a standard diet in mice but the high-fat diet gave it a boost and an “alternate pathway” out.
Now before you start ordering extra-large fries at the drive-through to keep the whipworms away, the researchers added that they “have previously published that weight loss can aid the expulsion of a different gut parasite worm.” Figures.
Once again, though, signs are pointing to the gut for improved health.
Pound of flesh buys less prison time
We should all have more Shakespeare in our lives. Yeah, yeah, Shakespeare is meant to be played, not read, and it can be a struggle to herd teenagers through the Bard’s interesting and bloody tragedies, but even a perfunctory reading of “The Merchant of Venice” would hopefully have prevented the dystopian nightmare Massachusetts has presented us with today.
The United States has a massive shortage of donor organs. This is an unfortunate truth. So, to combat this issue, a pair of Massachusetts congresspeople have proposed HD 3822, which would allow prisoners to donate organs and/or bone marrow (a pound of flesh, so to speak) in exchange for up to a year in reduced prison time. Yes, that’s right. Give up pieces of yourself and the state of Massachusetts will deign to reduce your long prison sentence.
Oh, and before you dismiss this as typical Republican antics, the bill was sponsored by two Democrats, and in a statement one of them hoped to address racial disparities in organ donation, as people of color are much less likely to receive organs. Never mind that Black people are imprisoned at a much higher rate than Whites.
Yeah, this whole thing is what people in the business like to call an ethical disaster.
Fortunately, the bill will likely never be passed and it’s probably illegal anyway. A federal law from 1984 (how’s that for a coincidence) prevents people from donating organs for use in human transplantation in exchange for “valuable consideration.” In other words, you can’t sell your organs for profit, and in this case, reducing prison time would probably count as valuable consideration in the eyes of the courts.
Oh, and in case you’ve never read Merchant of Venice, Shylock, the character looking for the pound of flesh as payment for a debt? He’s the villain. In fact, it’s pretty safe to say that anyone looking to extract payment from human dismemberment is probably the bad guy of the story. Apparently that wasn’t clear.
How do you stop a fungi? With a deadly guy
Thanks to the new HBO series “The Last of Us,” there’s been a lot of talk about the upcoming fungi-pocalypse, as the show depicts the real-life “zombie fungus” Cordyceps turning humans into, you know, zombies.
No need to worry, ladies and gentleman, because science has discovered a way to turn back the fungal horde. A heroic, and environmentally friendly, alternative to chemical pesticides “in the fight against resistant fungi [that] are now resistant to antimycotics – partly because they are used in large quantities in agricultural fields,” investigators at the Leibniz Institute for Natural Product Research and Infection Biology in Jena, Germany, said in a written statement.
We are, of course, talking about Keanu Reeves. Wait a second. He’s not even in “The Last of Us.” Sorry folks, we are being told that it really is Keanu Reeves. Our champion in the inevitable fungal pandemic is movie star Keanu Reeves. Sort of. It’s actually keanumycin, a substance produced by bacteria of the genus Pseudomonas.
Really? Keanumycin? “The lipopeptides kill so efficiently that we named them after Keanu Reeves because he, too, is extremely deadly in his roles,” lead author Sebastian Götze, PhD, explained.
Dr. Götze and his associates had been working with pseudomonads for quite a while before they were able to isolate the toxins responsible for their ability to kill amoebae, which resemble fungi in some characteristics. When then finally tried the keanumycin against gray mold rot on hydrangea leaves, the intensely contemplative star of “The Matrix” and “John Wick” – sorry, wrong Keanu – the bacterial derivative significantly inhibited growth of the fungus, they said.
Additional testing has shown that keanumycin is not highly toxic to human cells and is effective against fungi such as Candida albicans in very low concentrations, which makes it a good candidate for future pharmaceutical development.
To that news there can be only one response from the substance’s namesake.
High fat, bye parasites
Fat. Fat. Fat. Seems like everyone is trying to avoid it these days, but fat may be good thing when it comes to weaseling out a parasite.
The parasite in this case is the whipworm, aka Trichuris trichiura. You can find this guy in the intestines of millions of people, where it causes long-lasting infections. Yikes … Researchers have found that the plan of attack to get rid of this invasive species is to boost the immune system, but instead of vitamin C and zinc it’s fat they’re pumping in. Yes, fat.
The developing countries with poor sewage that are at the highest risk for contracting parasites such as this also are among those where people ingest cheaper diets that are generally higher in fat. The investigators were interested to see how a high-fat diet would affect immune responses to the whipworms.
And, as with almost everything else, the researchers turned to mice, which were introduced to a closely related species, Trichuris muris.
A high-fat diet, rather than obesity itself, increases a molecule on T-helper cells called ST2, and this allows an increased T-helper 2 response, effectively giving eviction notices to the parasites in the intestinal lining.
To say the least, the researchers were surprised since “high-fat diets are mostly associated with increased pathology during disease,” said senior author Richard Grencis, PhD, of the University of Manchester (England), who noted that ST2 is not normally triggered with a standard diet in mice but the high-fat diet gave it a boost and an “alternate pathway” out.
Now before you start ordering extra-large fries at the drive-through to keep the whipworms away, the researchers added that they “have previously published that weight loss can aid the expulsion of a different gut parasite worm.” Figures.
Once again, though, signs are pointing to the gut for improved health.
Rise of the fungi: Pandemic tied to increasing fungal infections
COVID-19 has lifted the lid on the risks of secondary pulmonary fungal infections in patients with severe respiratory viral illness – even previously immunocompetent individuals – and highlighted the importance of vigilant investigation to achieve early diagnoses, leading experts say.
Most fungi are not under surveillance in the United States, leaving experts without a national picture of the true burden of infection through the pandemic. However, a collection of published case series, cohort studies, and reviews from Europe, the United States, and throughout the world – mainly pre-Omicron – show that fungal disease has affected a significant portion of critically ill patients with COVID-19, with concerning excess mortality, these experts say.
COVID-associated pulmonary aspergillosis (CAPA) has been the predominant fungal coinfection in the United States and internationally. But COVID-associated mucormycosis (CAM) – the infection that surged in India in early 2021 – has also affected some patients in the United States, published data show. So have Pneumocystitis pneumonia, cryptococcosis, histoplasmosis, and Candida infections (which mainly affect the bloodstream and abdomen), say the experts who were interviewed.
“We had predicted [a rise in] aspergillosis, but we saw more than we thought we’d see. Most fungal infections became more common with COVID-19,” said George Thompson, MD, professor of clinical medicine at the University of California, Davis, and cochair of the University of Alabama–based Mycoses Study Group Education Committee, a group of experts in medical mycology. Pneumocystitis, for instance, “has historically been associated with AIDS or different types of leukemia or lymphoma, and is not an infection we’ve typically seen in our otherwise healthy ICU patients,” he noted. “But we did see more of it [with COVID-19].”
More recently, with fewer patients during the Omicron phase in intensive care units with acute respiratory failure, the profile of fungal disease secondary to COVID-19 has changed. Increasing proportions of patients have traditional risk factors for aspergillosis, such as hematologic malignancies and longer-term, pre-COVID use of systemic corticosteroids – a change that makes the contribution of the viral illness harder to distinguish.
Moving forward, the lessons of the COVID era – the fungal risks to patients with serious viral infections and the persistence needed to diagnose aspergillosis and other pulmonary fungal infections using bronchoscopy and imperfect noninvasive tests – should be taken to heart, experts say.
“Fungal diseases are not rare. They’re just not diagnosed because no one thinks to look for them,” said Dr. Thompson, a contributor to a recently released World Health Organization report naming a “fungal priority pathogens” list.
“We’re going to continue to see [secondary fungal infections] with other respiratory viruses,” he said. And overall, given environmental and other changes, “we’re going to see more and more fungal disease in the patients we take care of.”
CAPA not a surprise
CAPA is “not an unfamiliar story” in the world of fungal disease, given a history of influenza-associated pulmonary aspergillosis (IAPA), said Kieren A. Marr, MD, MBA, adjunct professor of medicine and past director of the transplant and oncology infectious diseases program at Johns Hopkins University, Baltimore, who has long researched invasive fungal disease.
European researchers, she said, have led the way in describing a high incidence of IAPA in patients admitted to ICUs with influenza. In a retrospective multicenter cohort study reported in 2018 by the Dutch-Belgian Mycosis Study group, for instance, almost 20% of 432 influenza patients admitted to the ICU, including patients who were otherwise healthy and not immunocompromised, had the diagnosis a median of 3 days after ICU admission. (Across other cohort studies, rates of IAPA have ranged from 7% to 30%.)
Mortality was significant: 51% of patients with influenza and invasive pulmonary aspergillosis died within 90 days, compared with 28% of patients with influenza and no invasive pulmonary aspergillosis.
Reports from Europe early in the pandemic indicated that CAPA was a similarly serious problem, prompting establishment at Johns Hopkins University of an aggressive screening program utilizing biomarker-based testing of blood and bronchoalveolar lavage (BAL) fluid. Of 396 mechanically ventilated COVID-19 patients admitted to Johns Hopkins University hospitals between March and August 2020, 39 met the institution’s criteria for CAPA, Dr. Marr and her colleagues reported this year in what might be the largest U.S. cohort study of CAPA published to date.
“We now know definitively that people with severe influenza and with severe COVID also have high risks for both invasive and airway disease caused by airborne fungi, most commonly aspergilliosis,” Dr. Marr said.
More recent unpublished analyses of patients from the start of the pandemic to June 2021 show persistent risk, said Nitipong Permpalung, MD, MPH, assistant professor in transplant and oncology infectious diseases at Johns Hopkins University and lead author of the cohort study. Among 832 patients with COVID-19 who were mechanically ventilated in Johns Hopkins University hospitals, 11.8% had CAPA, he said. (Also, 3.2% had invasive candidiasis, and 1.1% had other invasive fungal infections.)
Other sources said in interviews that these CAPA prevalence rates generally mirror reports from Europe, though some investigators in Europe have reported CAPA rates more toward 15%.
(The Mycoses Study Group recently collected data from its consortium of U.S. medical centers on the prevalence of CAPA, with funding support from the CDC, but at press time the data had not yet been released. Dr. Thompson said he suspected the prevalence will be lower than earlier papers have suggested, “but still will reflect a significant burden of disease.”)
Patients in the published Johns Hopkins University study who had CAPA were more likely than those with COVID-19 but no CAPA to have underlying pulmonary disease, liver disease, coagulopathy, solid tumors, multiple myeloma, and COVID-19–directed corticosteroids. And they had uniformly worse outcomes with regards to severity of illness and length of intubation.
How much of CAPA is driven by the SARS-CoV-2 virus itself and how much is a consequence of COVID-19 treatments is a topic of active discussion and research. Martin Hoenigl, MD, of the University of Graz, Austria, a leading researcher in medical mycology, said research shows corticosteroids and anti–IL-6 treatments, such as tocilizumab, used to treat COVID-19–driven acute respiratory failure clearly have contributed to CAPA. But he contends that “a number of other mechanisms” are involved as well.
“The immunologic mechanisms are definitely different in these patients with viral illness than in other ICU patients [who develop aspergilliosis]. It’s not just the corticosteroids. The more we learn, we see the virus plays a role as well, suppressing the interferon pathway,” for example, said Dr. Hoenigl, associate professor in the division of infectious diseases and the European Confederation of Medical Mycology (ECMM) Center of Excellence at the university. The earliest reports of CAPA came “when ICUs weren’t using dexamethasone or tocilizumab,” he noted.
In a paper published recently in Lancet Respiratory Medicine that Dr. Hoenigl and others point to, Belgian researchers reported a “three-level breach” in innate antifungal immunity in both IAPA and CAPA, affecting the integrity of the epithelial barrier, the capacity to phagocytose and kill Aspergillus spores, and the ability to destroy Aspergillus hyphae, which is mainly mediated by neutrophils.
The researchers ran a host of genetic and protein analyses on lung samples (most collected via BAL) of 169 patients with influenza or COVID-19, with and without aspergillosis. They found that patients with CAPA had significantly lower neutrophil cell fractions than patients with COVID-19 only, and patients with IAPA or CAPA had reduced type II IFN signaling and increased concentrations of fibrosis-associated growth factors in the lower respiratory tracts (Lancet Respir Med. 2022 Aug 24).
Tom Chiller, MD, MPH, chief of the Center for Disease Control and Prevention’s Mycotic Disease Branch, said he’s watching such research with interest. For now, he said, it’s important to also consider that “data on COVID show that almost all patients going into the ICUs with pneumonia and COVID are getting broad-spectrum antibiotics” in addition to corticosteroids.
By wiping out good bacteria, the antibiotics could be “creating a perfect niche for fungi to grow,” he said.
Diagnostic challenges
Aspergillus that has invaded the lung tissue in patients with COVID-19 appears to grow there for some time – around 8-10 days, much longer than in IAPA – before becoming angioinvasive, said Dr. Hoenigl. Such a pathophysiology “implicates that we should try to diagnose it while it’s in the lung tissue, using the BAL fluid, and not yet in the blood,” he said.
Some multicenter studies, including one from Europe on Aspergillus test profiles in critically ill COVID-19 patients, have shown mortality rates of close to 90% in patients with CAPA who have positive serum biomarkers, despite appropriate antifungal therapy. “If diagnosed while confined to the lung, however, mortality rates are more like 40%-50% with antifungal therapy,” Dr. Hoenigl said. (Cohort studies published thus far have fairly consistently reported mortality rates in patients with CAPA greater than 40%, he said.)
Bronchoscopy isn’t always pragmatic or possible, however, and is variably used. Some patients with severe COVID-19 may be too unstable for any invasive procedure, said Dr. Permpalung.
Dr. Permpalung looks for CAPA using serum (1-3) beta-D-glucan (BDG, a generic fungal test not specific to Aspergillus), serum galactomannan (GM, specific for Aspergillus), and respiratory cultures (sputum or endotracheal aspirate if intubated) as initial screening tests in the ICU. If there are concerns for CAPA – based on these tests and/or the clinical picture – “a thoughtful risk-benefit discussion is required to determine if patients would benefit from a bronchoscopy or if we should just start them on empiric antifungal therapy.”
Unfortunately, the sensitivity of serum GM is relatively low in CAPA – lower than with classic invasive aspergillosis in the nonviral setting, sources said. BDG, on the other hand, can be falsely positive in the setting of antimicrobials and within the ICU. And the utility of imaging for CAPA is limited. Both the clinical picture and radiological findings of CAPA have resembled those of severe COVID – with the caveat of cavitary lung lesions visible on imaging.
“Cavities or nodules are a highly suspicious finding that could indicate possible fungal infection,” said pulmonologist Amir A. Zeki, MD, MAS, professor of medicine at the University of California, Davis, and codirector of the UC Davis Asthma Network Clinic, who has cared for patients with CAPA.
Cavitation has been described in only a proportion of patients with CAPA, however. So in patients not doing well, “your suspicion has to be raised if you’re not seeing cavities,” he said.
Early in the pandemic, when patients worsened or failed to progress on mechanical ventilation, clinicians at the University of California, Davis, quickly learned not to pin blame too quickly on COVID-19 alone. This remains good advice today, Dr. Zeki said.
“If you have a patient who’s not doing well on a ventilator, not getting better [over weeks], has to be reintubated, has infiltrates or lung nodules that are evolving, or certainly, if they have a cavity, you have to suspect fungal infection,” said Dr. Zeki, who also practices at the Veterans Affairs Medical Center in San Diego. “Think about it for those patients who just aren’t moving forward and are continuing to struggle. Have a high index of suspicion, and consult with your infectious disease colleagues.”
Empiric treatment is warranted in some cases if a patient is doing poorly and suspicion for fungal infection is high based on clinical, radiographic, and/or laboratory evidence, he said.
The CDC’s Dr. Chiller said that screening and diagnostic algorithms currently vary from institution to institution, and that diagnostic challenges likely dissuade clinicians from thinking about fungi. “Clinicians often don’t want to deal with fungi – they’re difficult to diagnose, the treatments are limited and can be toxic. But fungi get pushed back until it’s too late,” he said.
“Fungal diagnostics is an area we all need a lot more help with,” and new diagnostics are in the pipeline, he said. In the meantime, he said, “there are tools out there, and we just need to use them more, and improve how they’re used.”
While reported CAPA thus far has typically occurred in the setting of ICU care and mechanical ventilation, it’s not always the case, Dr. Permpalung said. Lung and other solid organ transplant (SOT) recipients with COVID-19 are developing CAPA and other invasive secondary invasive fungal infections despite not being intubated, he said.
Of 276 SOT recipients with COVID-19 who required inpatient treatment at Johns Hopkins University hospitals from the beginning of the pandemic to March 2022, 23 patients developed invasive fungal infections (13 CAPA). Only a fraction – 38 of the 276 – had been intubated, he said.
Mucormycosis resistance
After CAPA, candidiasis and COVID-19-associated mucormycosis (CAM) – most frequently, rhino-orbital-cerebral disease or pulmonary disease – have been the leading reported fungal coinfections in COVID-19, said Dr. Hoenigl, who described the incidence, timeline, risk factors, and pathogenesis of these infections in a review published this year in Nature Microbiology. .
In India, where there has long been high exposure to Mucorales spores and a greater burden of invasive fungal disease, the rate of mucormycosis doubled in 2021, with rhino-orbital-cerebral disease reported almost exclusively, he said. Pulmonary disease has occurred almost exclusively in the ICU setting and has been present in about 50% of cases outside of India, including Europe and the United States.
A preprint meta-analysis of CAM cases posted by the Lancet in July 2022, in which investigators analyzed individual data of 556 reported cases of COVID-19–associated CAM, shows diabetes and history of corticosteroid use present in most patients, and an overall mortality rate of 44.4%, most of which stems from cases of pulmonary or disseminated disease. Thirteen of the 556 reported cases were from the United States.
An important take-away from the analysis, Dr. Hoenigl said, is that Aspergillus coinfection was seen in 7% of patients and was associated with higher mortality. “It’s important to consider that coinfections [of Aspergillus and Mucorales] can exist,” Dr. Hoenigl said, noting that like CAPA, pulmonary CAM is likely underdiagnosed and underreported.
As with CAPA, the clinical and radiological features of pulmonary CAM largely overlap with those associated with COVID-19, and bronchoscopy plays a central role in definitive diagnosis. In the United States, a Mucorales PCR test for blood and BAL fluid is commercially available and used at some centers, Dr. Hoenigl said.
“Mucormycosis is always difficult to treat ... a lot of the treatments don’t work particularly well,” said Dr. Thompson. “With aspergillosis, we have better treatment options.”
Dr. Thompson worries, however, about treatment resistance becoming widespread. Resistance to azole antifungal agents “is already pretty widespread in northern Europe, particularly in the Netherlands and part of the U.K.” because of injudicious use of antifungals in agriculture, he said. “We’ve started to see a few cases [of azole-resistant aspergillosis in the United States] and know it will be more widespread soon.”
Treatment resistance is a focus of the new WHO fungal priority pathogens list – the first such report from the organization. Of the 19 fungi on the list, 4 were ranked as critical: Cryptococcus neoformans, Candida auris, Aspergillus fumigatus, and Candida albicans. Like Dr. Thompson, Dr. Hoenigl contributed to the WHO report.
Dr. Hoenigl reported grant/research support from Astellas, Merck, F2G, Gilread, Pfizer, and Scynexis. Dr. Marr disclosed employment and equity in Pearl Diagnostics and Sfunga Therapeutics. Dr. Thompson, Dr. Permpalung, and Dr. Zeki reported that they have no relevant financial disclosures.
COVID-19 has lifted the lid on the risks of secondary pulmonary fungal infections in patients with severe respiratory viral illness – even previously immunocompetent individuals – and highlighted the importance of vigilant investigation to achieve early diagnoses, leading experts say.
Most fungi are not under surveillance in the United States, leaving experts without a national picture of the true burden of infection through the pandemic. However, a collection of published case series, cohort studies, and reviews from Europe, the United States, and throughout the world – mainly pre-Omicron – show that fungal disease has affected a significant portion of critically ill patients with COVID-19, with concerning excess mortality, these experts say.
COVID-associated pulmonary aspergillosis (CAPA) has been the predominant fungal coinfection in the United States and internationally. But COVID-associated mucormycosis (CAM) – the infection that surged in India in early 2021 – has also affected some patients in the United States, published data show. So have Pneumocystitis pneumonia, cryptococcosis, histoplasmosis, and Candida infections (which mainly affect the bloodstream and abdomen), say the experts who were interviewed.
“We had predicted [a rise in] aspergillosis, but we saw more than we thought we’d see. Most fungal infections became more common with COVID-19,” said George Thompson, MD, professor of clinical medicine at the University of California, Davis, and cochair of the University of Alabama–based Mycoses Study Group Education Committee, a group of experts in medical mycology. Pneumocystitis, for instance, “has historically been associated with AIDS or different types of leukemia or lymphoma, and is not an infection we’ve typically seen in our otherwise healthy ICU patients,” he noted. “But we did see more of it [with COVID-19].”
More recently, with fewer patients during the Omicron phase in intensive care units with acute respiratory failure, the profile of fungal disease secondary to COVID-19 has changed. Increasing proportions of patients have traditional risk factors for aspergillosis, such as hematologic malignancies and longer-term, pre-COVID use of systemic corticosteroids – a change that makes the contribution of the viral illness harder to distinguish.
Moving forward, the lessons of the COVID era – the fungal risks to patients with serious viral infections and the persistence needed to diagnose aspergillosis and other pulmonary fungal infections using bronchoscopy and imperfect noninvasive tests – should be taken to heart, experts say.
“Fungal diseases are not rare. They’re just not diagnosed because no one thinks to look for them,” said Dr. Thompson, a contributor to a recently released World Health Organization report naming a “fungal priority pathogens” list.
“We’re going to continue to see [secondary fungal infections] with other respiratory viruses,” he said. And overall, given environmental and other changes, “we’re going to see more and more fungal disease in the patients we take care of.”
CAPA not a surprise
CAPA is “not an unfamiliar story” in the world of fungal disease, given a history of influenza-associated pulmonary aspergillosis (IAPA), said Kieren A. Marr, MD, MBA, adjunct professor of medicine and past director of the transplant and oncology infectious diseases program at Johns Hopkins University, Baltimore, who has long researched invasive fungal disease.
European researchers, she said, have led the way in describing a high incidence of IAPA in patients admitted to ICUs with influenza. In a retrospective multicenter cohort study reported in 2018 by the Dutch-Belgian Mycosis Study group, for instance, almost 20% of 432 influenza patients admitted to the ICU, including patients who were otherwise healthy and not immunocompromised, had the diagnosis a median of 3 days after ICU admission. (Across other cohort studies, rates of IAPA have ranged from 7% to 30%.)
Mortality was significant: 51% of patients with influenza and invasive pulmonary aspergillosis died within 90 days, compared with 28% of patients with influenza and no invasive pulmonary aspergillosis.
Reports from Europe early in the pandemic indicated that CAPA was a similarly serious problem, prompting establishment at Johns Hopkins University of an aggressive screening program utilizing biomarker-based testing of blood and bronchoalveolar lavage (BAL) fluid. Of 396 mechanically ventilated COVID-19 patients admitted to Johns Hopkins University hospitals between March and August 2020, 39 met the institution’s criteria for CAPA, Dr. Marr and her colleagues reported this year in what might be the largest U.S. cohort study of CAPA published to date.
“We now know definitively that people with severe influenza and with severe COVID also have high risks for both invasive and airway disease caused by airborne fungi, most commonly aspergilliosis,” Dr. Marr said.
More recent unpublished analyses of patients from the start of the pandemic to June 2021 show persistent risk, said Nitipong Permpalung, MD, MPH, assistant professor in transplant and oncology infectious diseases at Johns Hopkins University and lead author of the cohort study. Among 832 patients with COVID-19 who were mechanically ventilated in Johns Hopkins University hospitals, 11.8% had CAPA, he said. (Also, 3.2% had invasive candidiasis, and 1.1% had other invasive fungal infections.)
Other sources said in interviews that these CAPA prevalence rates generally mirror reports from Europe, though some investigators in Europe have reported CAPA rates more toward 15%.
(The Mycoses Study Group recently collected data from its consortium of U.S. medical centers on the prevalence of CAPA, with funding support from the CDC, but at press time the data had not yet been released. Dr. Thompson said he suspected the prevalence will be lower than earlier papers have suggested, “but still will reflect a significant burden of disease.”)
Patients in the published Johns Hopkins University study who had CAPA were more likely than those with COVID-19 but no CAPA to have underlying pulmonary disease, liver disease, coagulopathy, solid tumors, multiple myeloma, and COVID-19–directed corticosteroids. And they had uniformly worse outcomes with regards to severity of illness and length of intubation.
How much of CAPA is driven by the SARS-CoV-2 virus itself and how much is a consequence of COVID-19 treatments is a topic of active discussion and research. Martin Hoenigl, MD, of the University of Graz, Austria, a leading researcher in medical mycology, said research shows corticosteroids and anti–IL-6 treatments, such as tocilizumab, used to treat COVID-19–driven acute respiratory failure clearly have contributed to CAPA. But he contends that “a number of other mechanisms” are involved as well.
“The immunologic mechanisms are definitely different in these patients with viral illness than in other ICU patients [who develop aspergilliosis]. It’s not just the corticosteroids. The more we learn, we see the virus plays a role as well, suppressing the interferon pathway,” for example, said Dr. Hoenigl, associate professor in the division of infectious diseases and the European Confederation of Medical Mycology (ECMM) Center of Excellence at the university. The earliest reports of CAPA came “when ICUs weren’t using dexamethasone or tocilizumab,” he noted.
In a paper published recently in Lancet Respiratory Medicine that Dr. Hoenigl and others point to, Belgian researchers reported a “three-level breach” in innate antifungal immunity in both IAPA and CAPA, affecting the integrity of the epithelial barrier, the capacity to phagocytose and kill Aspergillus spores, and the ability to destroy Aspergillus hyphae, which is mainly mediated by neutrophils.
The researchers ran a host of genetic and protein analyses on lung samples (most collected via BAL) of 169 patients with influenza or COVID-19, with and without aspergillosis. They found that patients with CAPA had significantly lower neutrophil cell fractions than patients with COVID-19 only, and patients with IAPA or CAPA had reduced type II IFN signaling and increased concentrations of fibrosis-associated growth factors in the lower respiratory tracts (Lancet Respir Med. 2022 Aug 24).
Tom Chiller, MD, MPH, chief of the Center for Disease Control and Prevention’s Mycotic Disease Branch, said he’s watching such research with interest. For now, he said, it’s important to also consider that “data on COVID show that almost all patients going into the ICUs with pneumonia and COVID are getting broad-spectrum antibiotics” in addition to corticosteroids.
By wiping out good bacteria, the antibiotics could be “creating a perfect niche for fungi to grow,” he said.
Diagnostic challenges
Aspergillus that has invaded the lung tissue in patients with COVID-19 appears to grow there for some time – around 8-10 days, much longer than in IAPA – before becoming angioinvasive, said Dr. Hoenigl. Such a pathophysiology “implicates that we should try to diagnose it while it’s in the lung tissue, using the BAL fluid, and not yet in the blood,” he said.
Some multicenter studies, including one from Europe on Aspergillus test profiles in critically ill COVID-19 patients, have shown mortality rates of close to 90% in patients with CAPA who have positive serum biomarkers, despite appropriate antifungal therapy. “If diagnosed while confined to the lung, however, mortality rates are more like 40%-50% with antifungal therapy,” Dr. Hoenigl said. (Cohort studies published thus far have fairly consistently reported mortality rates in patients with CAPA greater than 40%, he said.)
Bronchoscopy isn’t always pragmatic or possible, however, and is variably used. Some patients with severe COVID-19 may be too unstable for any invasive procedure, said Dr. Permpalung.
Dr. Permpalung looks for CAPA using serum (1-3) beta-D-glucan (BDG, a generic fungal test not specific to Aspergillus), serum galactomannan (GM, specific for Aspergillus), and respiratory cultures (sputum or endotracheal aspirate if intubated) as initial screening tests in the ICU. If there are concerns for CAPA – based on these tests and/or the clinical picture – “a thoughtful risk-benefit discussion is required to determine if patients would benefit from a bronchoscopy or if we should just start them on empiric antifungal therapy.”
Unfortunately, the sensitivity of serum GM is relatively low in CAPA – lower than with classic invasive aspergillosis in the nonviral setting, sources said. BDG, on the other hand, can be falsely positive in the setting of antimicrobials and within the ICU. And the utility of imaging for CAPA is limited. Both the clinical picture and radiological findings of CAPA have resembled those of severe COVID – with the caveat of cavitary lung lesions visible on imaging.
“Cavities or nodules are a highly suspicious finding that could indicate possible fungal infection,” said pulmonologist Amir A. Zeki, MD, MAS, professor of medicine at the University of California, Davis, and codirector of the UC Davis Asthma Network Clinic, who has cared for patients with CAPA.
Cavitation has been described in only a proportion of patients with CAPA, however. So in patients not doing well, “your suspicion has to be raised if you’re not seeing cavities,” he said.
Early in the pandemic, when patients worsened or failed to progress on mechanical ventilation, clinicians at the University of California, Davis, quickly learned not to pin blame too quickly on COVID-19 alone. This remains good advice today, Dr. Zeki said.
“If you have a patient who’s not doing well on a ventilator, not getting better [over weeks], has to be reintubated, has infiltrates or lung nodules that are evolving, or certainly, if they have a cavity, you have to suspect fungal infection,” said Dr. Zeki, who also practices at the Veterans Affairs Medical Center in San Diego. “Think about it for those patients who just aren’t moving forward and are continuing to struggle. Have a high index of suspicion, and consult with your infectious disease colleagues.”
Empiric treatment is warranted in some cases if a patient is doing poorly and suspicion for fungal infection is high based on clinical, radiographic, and/or laboratory evidence, he said.
The CDC’s Dr. Chiller said that screening and diagnostic algorithms currently vary from institution to institution, and that diagnostic challenges likely dissuade clinicians from thinking about fungi. “Clinicians often don’t want to deal with fungi – they’re difficult to diagnose, the treatments are limited and can be toxic. But fungi get pushed back until it’s too late,” he said.
“Fungal diagnostics is an area we all need a lot more help with,” and new diagnostics are in the pipeline, he said. In the meantime, he said, “there are tools out there, and we just need to use them more, and improve how they’re used.”
While reported CAPA thus far has typically occurred in the setting of ICU care and mechanical ventilation, it’s not always the case, Dr. Permpalung said. Lung and other solid organ transplant (SOT) recipients with COVID-19 are developing CAPA and other invasive secondary invasive fungal infections despite not being intubated, he said.
Of 276 SOT recipients with COVID-19 who required inpatient treatment at Johns Hopkins University hospitals from the beginning of the pandemic to March 2022, 23 patients developed invasive fungal infections (13 CAPA). Only a fraction – 38 of the 276 – had been intubated, he said.
Mucormycosis resistance
After CAPA, candidiasis and COVID-19-associated mucormycosis (CAM) – most frequently, rhino-orbital-cerebral disease or pulmonary disease – have been the leading reported fungal coinfections in COVID-19, said Dr. Hoenigl, who described the incidence, timeline, risk factors, and pathogenesis of these infections in a review published this year in Nature Microbiology. .
In India, where there has long been high exposure to Mucorales spores and a greater burden of invasive fungal disease, the rate of mucormycosis doubled in 2021, with rhino-orbital-cerebral disease reported almost exclusively, he said. Pulmonary disease has occurred almost exclusively in the ICU setting and has been present in about 50% of cases outside of India, including Europe and the United States.
A preprint meta-analysis of CAM cases posted by the Lancet in July 2022, in which investigators analyzed individual data of 556 reported cases of COVID-19–associated CAM, shows diabetes and history of corticosteroid use present in most patients, and an overall mortality rate of 44.4%, most of which stems from cases of pulmonary or disseminated disease. Thirteen of the 556 reported cases were from the United States.
An important take-away from the analysis, Dr. Hoenigl said, is that Aspergillus coinfection was seen in 7% of patients and was associated with higher mortality. “It’s important to consider that coinfections [of Aspergillus and Mucorales] can exist,” Dr. Hoenigl said, noting that like CAPA, pulmonary CAM is likely underdiagnosed and underreported.
As with CAPA, the clinical and radiological features of pulmonary CAM largely overlap with those associated with COVID-19, and bronchoscopy plays a central role in definitive diagnosis. In the United States, a Mucorales PCR test for blood and BAL fluid is commercially available and used at some centers, Dr. Hoenigl said.
“Mucormycosis is always difficult to treat ... a lot of the treatments don’t work particularly well,” said Dr. Thompson. “With aspergillosis, we have better treatment options.”
Dr. Thompson worries, however, about treatment resistance becoming widespread. Resistance to azole antifungal agents “is already pretty widespread in northern Europe, particularly in the Netherlands and part of the U.K.” because of injudicious use of antifungals in agriculture, he said. “We’ve started to see a few cases [of azole-resistant aspergillosis in the United States] and know it will be more widespread soon.”
Treatment resistance is a focus of the new WHO fungal priority pathogens list – the first such report from the organization. Of the 19 fungi on the list, 4 were ranked as critical: Cryptococcus neoformans, Candida auris, Aspergillus fumigatus, and Candida albicans. Like Dr. Thompson, Dr. Hoenigl contributed to the WHO report.
Dr. Hoenigl reported grant/research support from Astellas, Merck, F2G, Gilread, Pfizer, and Scynexis. Dr. Marr disclosed employment and equity in Pearl Diagnostics and Sfunga Therapeutics. Dr. Thompson, Dr. Permpalung, and Dr. Zeki reported that they have no relevant financial disclosures.
COVID-19 has lifted the lid on the risks of secondary pulmonary fungal infections in patients with severe respiratory viral illness – even previously immunocompetent individuals – and highlighted the importance of vigilant investigation to achieve early diagnoses, leading experts say.
Most fungi are not under surveillance in the United States, leaving experts without a national picture of the true burden of infection through the pandemic. However, a collection of published case series, cohort studies, and reviews from Europe, the United States, and throughout the world – mainly pre-Omicron – show that fungal disease has affected a significant portion of critically ill patients with COVID-19, with concerning excess mortality, these experts say.
COVID-associated pulmonary aspergillosis (CAPA) has been the predominant fungal coinfection in the United States and internationally. But COVID-associated mucormycosis (CAM) – the infection that surged in India in early 2021 – has also affected some patients in the United States, published data show. So have Pneumocystitis pneumonia, cryptococcosis, histoplasmosis, and Candida infections (which mainly affect the bloodstream and abdomen), say the experts who were interviewed.
“We had predicted [a rise in] aspergillosis, but we saw more than we thought we’d see. Most fungal infections became more common with COVID-19,” said George Thompson, MD, professor of clinical medicine at the University of California, Davis, and cochair of the University of Alabama–based Mycoses Study Group Education Committee, a group of experts in medical mycology. Pneumocystitis, for instance, “has historically been associated with AIDS or different types of leukemia or lymphoma, and is not an infection we’ve typically seen in our otherwise healthy ICU patients,” he noted. “But we did see more of it [with COVID-19].”
More recently, with fewer patients during the Omicron phase in intensive care units with acute respiratory failure, the profile of fungal disease secondary to COVID-19 has changed. Increasing proportions of patients have traditional risk factors for aspergillosis, such as hematologic malignancies and longer-term, pre-COVID use of systemic corticosteroids – a change that makes the contribution of the viral illness harder to distinguish.
Moving forward, the lessons of the COVID era – the fungal risks to patients with serious viral infections and the persistence needed to diagnose aspergillosis and other pulmonary fungal infections using bronchoscopy and imperfect noninvasive tests – should be taken to heart, experts say.
“Fungal diseases are not rare. They’re just not diagnosed because no one thinks to look for them,” said Dr. Thompson, a contributor to a recently released World Health Organization report naming a “fungal priority pathogens” list.
“We’re going to continue to see [secondary fungal infections] with other respiratory viruses,” he said. And overall, given environmental and other changes, “we’re going to see more and more fungal disease in the patients we take care of.”
CAPA not a surprise
CAPA is “not an unfamiliar story” in the world of fungal disease, given a history of influenza-associated pulmonary aspergillosis (IAPA), said Kieren A. Marr, MD, MBA, adjunct professor of medicine and past director of the transplant and oncology infectious diseases program at Johns Hopkins University, Baltimore, who has long researched invasive fungal disease.
European researchers, she said, have led the way in describing a high incidence of IAPA in patients admitted to ICUs with influenza. In a retrospective multicenter cohort study reported in 2018 by the Dutch-Belgian Mycosis Study group, for instance, almost 20% of 432 influenza patients admitted to the ICU, including patients who were otherwise healthy and not immunocompromised, had the diagnosis a median of 3 days after ICU admission. (Across other cohort studies, rates of IAPA have ranged from 7% to 30%.)
Mortality was significant: 51% of patients with influenza and invasive pulmonary aspergillosis died within 90 days, compared with 28% of patients with influenza and no invasive pulmonary aspergillosis.
Reports from Europe early in the pandemic indicated that CAPA was a similarly serious problem, prompting establishment at Johns Hopkins University of an aggressive screening program utilizing biomarker-based testing of blood and bronchoalveolar lavage (BAL) fluid. Of 396 mechanically ventilated COVID-19 patients admitted to Johns Hopkins University hospitals between March and August 2020, 39 met the institution’s criteria for CAPA, Dr. Marr and her colleagues reported this year in what might be the largest U.S. cohort study of CAPA published to date.
“We now know definitively that people with severe influenza and with severe COVID also have high risks for both invasive and airway disease caused by airborne fungi, most commonly aspergilliosis,” Dr. Marr said.
More recent unpublished analyses of patients from the start of the pandemic to June 2021 show persistent risk, said Nitipong Permpalung, MD, MPH, assistant professor in transplant and oncology infectious diseases at Johns Hopkins University and lead author of the cohort study. Among 832 patients with COVID-19 who were mechanically ventilated in Johns Hopkins University hospitals, 11.8% had CAPA, he said. (Also, 3.2% had invasive candidiasis, and 1.1% had other invasive fungal infections.)
Other sources said in interviews that these CAPA prevalence rates generally mirror reports from Europe, though some investigators in Europe have reported CAPA rates more toward 15%.
(The Mycoses Study Group recently collected data from its consortium of U.S. medical centers on the prevalence of CAPA, with funding support from the CDC, but at press time the data had not yet been released. Dr. Thompson said he suspected the prevalence will be lower than earlier papers have suggested, “but still will reflect a significant burden of disease.”)
Patients in the published Johns Hopkins University study who had CAPA were more likely than those with COVID-19 but no CAPA to have underlying pulmonary disease, liver disease, coagulopathy, solid tumors, multiple myeloma, and COVID-19–directed corticosteroids. And they had uniformly worse outcomes with regards to severity of illness and length of intubation.
How much of CAPA is driven by the SARS-CoV-2 virus itself and how much is a consequence of COVID-19 treatments is a topic of active discussion and research. Martin Hoenigl, MD, of the University of Graz, Austria, a leading researcher in medical mycology, said research shows corticosteroids and anti–IL-6 treatments, such as tocilizumab, used to treat COVID-19–driven acute respiratory failure clearly have contributed to CAPA. But he contends that “a number of other mechanisms” are involved as well.
“The immunologic mechanisms are definitely different in these patients with viral illness than in other ICU patients [who develop aspergilliosis]. It’s not just the corticosteroids. The more we learn, we see the virus plays a role as well, suppressing the interferon pathway,” for example, said Dr. Hoenigl, associate professor in the division of infectious diseases and the European Confederation of Medical Mycology (ECMM) Center of Excellence at the university. The earliest reports of CAPA came “when ICUs weren’t using dexamethasone or tocilizumab,” he noted.
In a paper published recently in Lancet Respiratory Medicine that Dr. Hoenigl and others point to, Belgian researchers reported a “three-level breach” in innate antifungal immunity in both IAPA and CAPA, affecting the integrity of the epithelial barrier, the capacity to phagocytose and kill Aspergillus spores, and the ability to destroy Aspergillus hyphae, which is mainly mediated by neutrophils.
The researchers ran a host of genetic and protein analyses on lung samples (most collected via BAL) of 169 patients with influenza or COVID-19, with and without aspergillosis. They found that patients with CAPA had significantly lower neutrophil cell fractions than patients with COVID-19 only, and patients with IAPA or CAPA had reduced type II IFN signaling and increased concentrations of fibrosis-associated growth factors in the lower respiratory tracts (Lancet Respir Med. 2022 Aug 24).
Tom Chiller, MD, MPH, chief of the Center for Disease Control and Prevention’s Mycotic Disease Branch, said he’s watching such research with interest. For now, he said, it’s important to also consider that “data on COVID show that almost all patients going into the ICUs with pneumonia and COVID are getting broad-spectrum antibiotics” in addition to corticosteroids.
By wiping out good bacteria, the antibiotics could be “creating a perfect niche for fungi to grow,” he said.
Diagnostic challenges
Aspergillus that has invaded the lung tissue in patients with COVID-19 appears to grow there for some time – around 8-10 days, much longer than in IAPA – before becoming angioinvasive, said Dr. Hoenigl. Such a pathophysiology “implicates that we should try to diagnose it while it’s in the lung tissue, using the BAL fluid, and not yet in the blood,” he said.
Some multicenter studies, including one from Europe on Aspergillus test profiles in critically ill COVID-19 patients, have shown mortality rates of close to 90% in patients with CAPA who have positive serum biomarkers, despite appropriate antifungal therapy. “If diagnosed while confined to the lung, however, mortality rates are more like 40%-50% with antifungal therapy,” Dr. Hoenigl said. (Cohort studies published thus far have fairly consistently reported mortality rates in patients with CAPA greater than 40%, he said.)
Bronchoscopy isn’t always pragmatic or possible, however, and is variably used. Some patients with severe COVID-19 may be too unstable for any invasive procedure, said Dr. Permpalung.
Dr. Permpalung looks for CAPA using serum (1-3) beta-D-glucan (BDG, a generic fungal test not specific to Aspergillus), serum galactomannan (GM, specific for Aspergillus), and respiratory cultures (sputum or endotracheal aspirate if intubated) as initial screening tests in the ICU. If there are concerns for CAPA – based on these tests and/or the clinical picture – “a thoughtful risk-benefit discussion is required to determine if patients would benefit from a bronchoscopy or if we should just start them on empiric antifungal therapy.”
Unfortunately, the sensitivity of serum GM is relatively low in CAPA – lower than with classic invasive aspergillosis in the nonviral setting, sources said. BDG, on the other hand, can be falsely positive in the setting of antimicrobials and within the ICU. And the utility of imaging for CAPA is limited. Both the clinical picture and radiological findings of CAPA have resembled those of severe COVID – with the caveat of cavitary lung lesions visible on imaging.
“Cavities or nodules are a highly suspicious finding that could indicate possible fungal infection,” said pulmonologist Amir A. Zeki, MD, MAS, professor of medicine at the University of California, Davis, and codirector of the UC Davis Asthma Network Clinic, who has cared for patients with CAPA.
Cavitation has been described in only a proportion of patients with CAPA, however. So in patients not doing well, “your suspicion has to be raised if you’re not seeing cavities,” he said.
Early in the pandemic, when patients worsened or failed to progress on mechanical ventilation, clinicians at the University of California, Davis, quickly learned not to pin blame too quickly on COVID-19 alone. This remains good advice today, Dr. Zeki said.
“If you have a patient who’s not doing well on a ventilator, not getting better [over weeks], has to be reintubated, has infiltrates or lung nodules that are evolving, or certainly, if they have a cavity, you have to suspect fungal infection,” said Dr. Zeki, who also practices at the Veterans Affairs Medical Center in San Diego. “Think about it for those patients who just aren’t moving forward and are continuing to struggle. Have a high index of suspicion, and consult with your infectious disease colleagues.”
Empiric treatment is warranted in some cases if a patient is doing poorly and suspicion for fungal infection is high based on clinical, radiographic, and/or laboratory evidence, he said.
The CDC’s Dr. Chiller said that screening and diagnostic algorithms currently vary from institution to institution, and that diagnostic challenges likely dissuade clinicians from thinking about fungi. “Clinicians often don’t want to deal with fungi – they’re difficult to diagnose, the treatments are limited and can be toxic. But fungi get pushed back until it’s too late,” he said.
“Fungal diagnostics is an area we all need a lot more help with,” and new diagnostics are in the pipeline, he said. In the meantime, he said, “there are tools out there, and we just need to use them more, and improve how they’re used.”
While reported CAPA thus far has typically occurred in the setting of ICU care and mechanical ventilation, it’s not always the case, Dr. Permpalung said. Lung and other solid organ transplant (SOT) recipients with COVID-19 are developing CAPA and other invasive secondary invasive fungal infections despite not being intubated, he said.
Of 276 SOT recipients with COVID-19 who required inpatient treatment at Johns Hopkins University hospitals from the beginning of the pandemic to March 2022, 23 patients developed invasive fungal infections (13 CAPA). Only a fraction – 38 of the 276 – had been intubated, he said.
Mucormycosis resistance
After CAPA, candidiasis and COVID-19-associated mucormycosis (CAM) – most frequently, rhino-orbital-cerebral disease or pulmonary disease – have been the leading reported fungal coinfections in COVID-19, said Dr. Hoenigl, who described the incidence, timeline, risk factors, and pathogenesis of these infections in a review published this year in Nature Microbiology. .
In India, where there has long been high exposure to Mucorales spores and a greater burden of invasive fungal disease, the rate of mucormycosis doubled in 2021, with rhino-orbital-cerebral disease reported almost exclusively, he said. Pulmonary disease has occurred almost exclusively in the ICU setting and has been present in about 50% of cases outside of India, including Europe and the United States.
A preprint meta-analysis of CAM cases posted by the Lancet in July 2022, in which investigators analyzed individual data of 556 reported cases of COVID-19–associated CAM, shows diabetes and history of corticosteroid use present in most patients, and an overall mortality rate of 44.4%, most of which stems from cases of pulmonary or disseminated disease. Thirteen of the 556 reported cases were from the United States.
An important take-away from the analysis, Dr. Hoenigl said, is that Aspergillus coinfection was seen in 7% of patients and was associated with higher mortality. “It’s important to consider that coinfections [of Aspergillus and Mucorales] can exist,” Dr. Hoenigl said, noting that like CAPA, pulmonary CAM is likely underdiagnosed and underreported.
As with CAPA, the clinical and radiological features of pulmonary CAM largely overlap with those associated with COVID-19, and bronchoscopy plays a central role in definitive diagnosis. In the United States, a Mucorales PCR test for blood and BAL fluid is commercially available and used at some centers, Dr. Hoenigl said.
“Mucormycosis is always difficult to treat ... a lot of the treatments don’t work particularly well,” said Dr. Thompson. “With aspergillosis, we have better treatment options.”
Dr. Thompson worries, however, about treatment resistance becoming widespread. Resistance to azole antifungal agents “is already pretty widespread in northern Europe, particularly in the Netherlands and part of the U.K.” because of injudicious use of antifungals in agriculture, he said. “We’ve started to see a few cases [of azole-resistant aspergillosis in the United States] and know it will be more widespread soon.”
Treatment resistance is a focus of the new WHO fungal priority pathogens list – the first such report from the organization. Of the 19 fungi on the list, 4 were ranked as critical: Cryptococcus neoformans, Candida auris, Aspergillus fumigatus, and Candida albicans. Like Dr. Thompson, Dr. Hoenigl contributed to the WHO report.
Dr. Hoenigl reported grant/research support from Astellas, Merck, F2G, Gilread, Pfizer, and Scynexis. Dr. Marr disclosed employment and equity in Pearl Diagnostics and Sfunga Therapeutics. Dr. Thompson, Dr. Permpalung, and Dr. Zeki reported that they have no relevant financial disclosures.
Emerging invasive fungal infections call for multidisciplinary cooperation
BUENOS AIRES – Emerging invasive fungal infections represent a new diagnostic and therapeutic challenge. To address their growing clinical impact on immunocompromised patients requires better local epidemiologic records, said a specialist at the XXII Congress of the Argentine Society of Infectology.
“To know that these fungal infections exist, I believe that in this respect we are falling short,” said Javier Afeltra, PhD, a mycologist at the Ramos Mejía Hospital in Buenos Aires, professor of microbiology at the School of Medicine of the University of Buenos Aires, and coordinator of the commission of immunocompromised patients of the Argentine Society of Infectious Diseases.
“There is some change in mentality that encourages professionals to report the cases they detect – for example, in scientific meetings,” Dr. Afeltra told this news orgnization. “But the problem is that there is no unified registry.
“That’s what we lack: a place to record all those isolated cases. Records where clinical and microbiological data are together within a click. Perhaps the microbiologists report their findings to the Malbrán Institute, an Argentine reference center for infectious disease research, but we do not know what the patients had. And we doctors may get together to make records of what happens clinically with the patient, but the germ data are elsewhere. We need a common registry,” he stressed.
“The main importance of a registry of this type is that it would allow a diagnostic and therapeutic decision to be made that is appropriate to the epidemiological profile of the country and the region, not looking at what they do in the North. Most likely, the best antifungal treatment for our country differs from what is indicated in the guidelines written elsewhere,” said Dr. Afeltra.
Dr. Afeltra pointed out that in the United States, when an oncohematology patient does not respond to antimicrobial treatment, the first thing that doctors think is that the patient has aspergillosis or mucormycosis, in which the fungal infection is caused by filamentous fungi.
But an analysis of data from the REMINI registry – the only prospective, observational, multicenter surveillance registry for invasive mycoses in immunocompromised patients (excluding HIV infection) in Argentina, which has been in existence since 2010 – tells a different story. The most prevalent fungal infections turned out to be those caused by Aspergillus species, followed by Fusarium species. Together, they account for more than half of cases. Mucoral infections (mucormycosis) account for less than 6%. And the initial treatments for these diseases could be different.
Changes in the local epidemiology can occur because the behavior of phytopathogenic fungi found in the environment can be modified. For example, cases of chronic mucormycosis can be detected in China but are virtually nonexistent on this side of the Greenwich meridian, Dr. Afeltra said.
“Nature is not the same in geographical areas, and the fungi … we breathe are completely different, so patients have different infections and require different diagnostic and treatment approaches,” he stressed.
Dr. Afeltra mentioned different fungi that are emerging locally and globally, including yeasts, septate, dimorphic, and pigmented hyaline fungi, that have a variable response to antifungal drugs and are associated with high mortality, “which has a lot to do with a later diagnosis,” he said, noting that reports have increased worldwide. A barrier to sharing this information more widely with the professional community, in addition to the lack of records, is the difficulty in publishing cases or series of cases in indexed journals.
Another challenge in characterizing the phenomenon is in regard to taxonomic reclassifications of fungi. Such reclassifications can mean that “perhaps we are speaking of the same pathogen in similar situations, believing that we are referring to different pathogens,” said Dr. Afeltra.
Clinical pearls related to emerging fungal pathogens
Candida auris. This organism has emerged simultaneously on several continents. It has pathogenicity factors typical of the genus, such as biofilm formation and production of phospholipases and proteinases, although it has greater thermal tolerance. In hospitals, it colonizes for weeks and months. In Argentina, it is resistant to multiple antifungal agents. Sensitivity is variable in different geographical regions. Most strains are resistant to fluconazole, and there is variable resistance to the other triazoles [which are not normally used to treat candidemia]. In the United States, in vitro resistance to amphotericin B is up to 30%, and resistance to echinocandins is up to 5%. New drugs such as rezafungin and ibrexafungerp are being studied. Infection control is similar to that used to control Clostridium difficile.
Fusarium. This genus affects immunocompromised patients, including transplant recipients of solid organs and hematopoietic progenitor cells and patients with neutropenia. The genus has various species, included within complexes, such as F. solani SC, F. oxysporum SC, and F. fujikuroi SC, with clinical manifestations similar to those of aspergillosis. In addition to the pulmonary and disseminated forms, there may be skin involvement attributable to dissemination from a respiratory focus or by contiguity from a focus of onychomycosis. In general, mortality is high, and responses to antifungal agents are variable. Some species are more sensitive to voriconazole or posaconazole, and others less so. All show in vitro resistance to itraconazole. In Argentina, voriconazole is usually used as initial treatment, and in special cases, liposomal amphotericin B or combinations. Fosmanogepix is being evaluated for the future.
Azole-resistant aspergillosis. This infection has shown resistance to itraconazole and third-generation azole drugs. In immunocompromised patients, mortlaity is high. Early detection is key. It is sensitive to amphotericin B and echinocandins. It is generally treated with liposomal amphotericin B. Olorofim and fosmanogepix are being studied.
Pulmonary aspergillosis associated with COVID-19. This infection is associated with high mortality among intubated patients. Signs and symptoms include fever, pleural effusion, hemoptysis, and chest pain, with infiltrates or cavitations on imaging. Determining the diagnosis is difficult. “We couldn’t perform lung biopsies, and it was difficult for us to get patients out of intensive care units for CT scans. We treated the proven cases. We treated the probable cases, and those that had a very low certainty of disease were also treated. We came across this emergency and tried to do the best we could,” said Dr. Afeltra. A digital readout lateral flow trial (Sona Aspergillus Galactomannan LFA) for the quantification of galactomannan, a cell wall component of the Aspergillus genus, proved to be a useful tool for screening and diagnosing patients with probable pulmonary aspergillosis associated with COVID-19. The incidence of invasive mycosis was around 10% among 185 seriously ill COVID-19 patients, according to an Argentine multicenter prospective study in which Dr. Afeltra participated.
Scedosporium and Lomentospora. These genera are rarer septate hyaline fungi. Scedosporium is a complex of species. One species, S. apiospermum, can colonize pediatric patients with cystic fibrosis. Lomentospora prolificans is a multiresistant fungus. It produces pulmonary compromise or disseminated infection. The response to antifungal agents is variable, with a high minimum inhibitory concentration for amphotericin B and isavuconazole. Patients are usually treated with voriconazole alone or in combination with terbinafine or micafungin. Olorofim is emerging as a promising treatment.
Dr. Afeltra has received fees from Biotoscana, Gador, Pfizer, Merck, and Sandoz.
This article was translated from the Medscape Spanish edition, a version appeared on Medscape.com.
BUENOS AIRES – Emerging invasive fungal infections represent a new diagnostic and therapeutic challenge. To address their growing clinical impact on immunocompromised patients requires better local epidemiologic records, said a specialist at the XXII Congress of the Argentine Society of Infectology.
“To know that these fungal infections exist, I believe that in this respect we are falling short,” said Javier Afeltra, PhD, a mycologist at the Ramos Mejía Hospital in Buenos Aires, professor of microbiology at the School of Medicine of the University of Buenos Aires, and coordinator of the commission of immunocompromised patients of the Argentine Society of Infectious Diseases.
“There is some change in mentality that encourages professionals to report the cases they detect – for example, in scientific meetings,” Dr. Afeltra told this news orgnization. “But the problem is that there is no unified registry.
“That’s what we lack: a place to record all those isolated cases. Records where clinical and microbiological data are together within a click. Perhaps the microbiologists report their findings to the Malbrán Institute, an Argentine reference center for infectious disease research, but we do not know what the patients had. And we doctors may get together to make records of what happens clinically with the patient, but the germ data are elsewhere. We need a common registry,” he stressed.
“The main importance of a registry of this type is that it would allow a diagnostic and therapeutic decision to be made that is appropriate to the epidemiological profile of the country and the region, not looking at what they do in the North. Most likely, the best antifungal treatment for our country differs from what is indicated in the guidelines written elsewhere,” said Dr. Afeltra.
Dr. Afeltra pointed out that in the United States, when an oncohematology patient does not respond to antimicrobial treatment, the first thing that doctors think is that the patient has aspergillosis or mucormycosis, in which the fungal infection is caused by filamentous fungi.
But an analysis of data from the REMINI registry – the only prospective, observational, multicenter surveillance registry for invasive mycoses in immunocompromised patients (excluding HIV infection) in Argentina, which has been in existence since 2010 – tells a different story. The most prevalent fungal infections turned out to be those caused by Aspergillus species, followed by Fusarium species. Together, they account for more than half of cases. Mucoral infections (mucormycosis) account for less than 6%. And the initial treatments for these diseases could be different.
Changes in the local epidemiology can occur because the behavior of phytopathogenic fungi found in the environment can be modified. For example, cases of chronic mucormycosis can be detected in China but are virtually nonexistent on this side of the Greenwich meridian, Dr. Afeltra said.
“Nature is not the same in geographical areas, and the fungi … we breathe are completely different, so patients have different infections and require different diagnostic and treatment approaches,” he stressed.
Dr. Afeltra mentioned different fungi that are emerging locally and globally, including yeasts, septate, dimorphic, and pigmented hyaline fungi, that have a variable response to antifungal drugs and are associated with high mortality, “which has a lot to do with a later diagnosis,” he said, noting that reports have increased worldwide. A barrier to sharing this information more widely with the professional community, in addition to the lack of records, is the difficulty in publishing cases or series of cases in indexed journals.
Another challenge in characterizing the phenomenon is in regard to taxonomic reclassifications of fungi. Such reclassifications can mean that “perhaps we are speaking of the same pathogen in similar situations, believing that we are referring to different pathogens,” said Dr. Afeltra.
Clinical pearls related to emerging fungal pathogens
Candida auris. This organism has emerged simultaneously on several continents. It has pathogenicity factors typical of the genus, such as biofilm formation and production of phospholipases and proteinases, although it has greater thermal tolerance. In hospitals, it colonizes for weeks and months. In Argentina, it is resistant to multiple antifungal agents. Sensitivity is variable in different geographical regions. Most strains are resistant to fluconazole, and there is variable resistance to the other triazoles [which are not normally used to treat candidemia]. In the United States, in vitro resistance to amphotericin B is up to 30%, and resistance to echinocandins is up to 5%. New drugs such as rezafungin and ibrexafungerp are being studied. Infection control is similar to that used to control Clostridium difficile.
Fusarium. This genus affects immunocompromised patients, including transplant recipients of solid organs and hematopoietic progenitor cells and patients with neutropenia. The genus has various species, included within complexes, such as F. solani SC, F. oxysporum SC, and F. fujikuroi SC, with clinical manifestations similar to those of aspergillosis. In addition to the pulmonary and disseminated forms, there may be skin involvement attributable to dissemination from a respiratory focus or by contiguity from a focus of onychomycosis. In general, mortality is high, and responses to antifungal agents are variable. Some species are more sensitive to voriconazole or posaconazole, and others less so. All show in vitro resistance to itraconazole. In Argentina, voriconazole is usually used as initial treatment, and in special cases, liposomal amphotericin B or combinations. Fosmanogepix is being evaluated for the future.
Azole-resistant aspergillosis. This infection has shown resistance to itraconazole and third-generation azole drugs. In immunocompromised patients, mortlaity is high. Early detection is key. It is sensitive to amphotericin B and echinocandins. It is generally treated with liposomal amphotericin B. Olorofim and fosmanogepix are being studied.
Pulmonary aspergillosis associated with COVID-19. This infection is associated with high mortality among intubated patients. Signs and symptoms include fever, pleural effusion, hemoptysis, and chest pain, with infiltrates or cavitations on imaging. Determining the diagnosis is difficult. “We couldn’t perform lung biopsies, and it was difficult for us to get patients out of intensive care units for CT scans. We treated the proven cases. We treated the probable cases, and those that had a very low certainty of disease were also treated. We came across this emergency and tried to do the best we could,” said Dr. Afeltra. A digital readout lateral flow trial (Sona Aspergillus Galactomannan LFA) for the quantification of galactomannan, a cell wall component of the Aspergillus genus, proved to be a useful tool for screening and diagnosing patients with probable pulmonary aspergillosis associated with COVID-19. The incidence of invasive mycosis was around 10% among 185 seriously ill COVID-19 patients, according to an Argentine multicenter prospective study in which Dr. Afeltra participated.
Scedosporium and Lomentospora. These genera are rarer septate hyaline fungi. Scedosporium is a complex of species. One species, S. apiospermum, can colonize pediatric patients with cystic fibrosis. Lomentospora prolificans is a multiresistant fungus. It produces pulmonary compromise or disseminated infection. The response to antifungal agents is variable, with a high minimum inhibitory concentration for amphotericin B and isavuconazole. Patients are usually treated with voriconazole alone or in combination with terbinafine or micafungin. Olorofim is emerging as a promising treatment.
Dr. Afeltra has received fees from Biotoscana, Gador, Pfizer, Merck, and Sandoz.
This article was translated from the Medscape Spanish edition, a version appeared on Medscape.com.
BUENOS AIRES – Emerging invasive fungal infections represent a new diagnostic and therapeutic challenge. To address their growing clinical impact on immunocompromised patients requires better local epidemiologic records, said a specialist at the XXII Congress of the Argentine Society of Infectology.
“To know that these fungal infections exist, I believe that in this respect we are falling short,” said Javier Afeltra, PhD, a mycologist at the Ramos Mejía Hospital in Buenos Aires, professor of microbiology at the School of Medicine of the University of Buenos Aires, and coordinator of the commission of immunocompromised patients of the Argentine Society of Infectious Diseases.
“There is some change in mentality that encourages professionals to report the cases they detect – for example, in scientific meetings,” Dr. Afeltra told this news orgnization. “But the problem is that there is no unified registry.
“That’s what we lack: a place to record all those isolated cases. Records where clinical and microbiological data are together within a click. Perhaps the microbiologists report their findings to the Malbrán Institute, an Argentine reference center for infectious disease research, but we do not know what the patients had. And we doctors may get together to make records of what happens clinically with the patient, but the germ data are elsewhere. We need a common registry,” he stressed.
“The main importance of a registry of this type is that it would allow a diagnostic and therapeutic decision to be made that is appropriate to the epidemiological profile of the country and the region, not looking at what they do in the North. Most likely, the best antifungal treatment for our country differs from what is indicated in the guidelines written elsewhere,” said Dr. Afeltra.
Dr. Afeltra pointed out that in the United States, when an oncohematology patient does not respond to antimicrobial treatment, the first thing that doctors think is that the patient has aspergillosis or mucormycosis, in which the fungal infection is caused by filamentous fungi.
But an analysis of data from the REMINI registry – the only prospective, observational, multicenter surveillance registry for invasive mycoses in immunocompromised patients (excluding HIV infection) in Argentina, which has been in existence since 2010 – tells a different story. The most prevalent fungal infections turned out to be those caused by Aspergillus species, followed by Fusarium species. Together, they account for more than half of cases. Mucoral infections (mucormycosis) account for less than 6%. And the initial treatments for these diseases could be different.
Changes in the local epidemiology can occur because the behavior of phytopathogenic fungi found in the environment can be modified. For example, cases of chronic mucormycosis can be detected in China but are virtually nonexistent on this side of the Greenwich meridian, Dr. Afeltra said.
“Nature is not the same in geographical areas, and the fungi … we breathe are completely different, so patients have different infections and require different diagnostic and treatment approaches,” he stressed.
Dr. Afeltra mentioned different fungi that are emerging locally and globally, including yeasts, septate, dimorphic, and pigmented hyaline fungi, that have a variable response to antifungal drugs and are associated with high mortality, “which has a lot to do with a later diagnosis,” he said, noting that reports have increased worldwide. A barrier to sharing this information more widely with the professional community, in addition to the lack of records, is the difficulty in publishing cases or series of cases in indexed journals.
Another challenge in characterizing the phenomenon is in regard to taxonomic reclassifications of fungi. Such reclassifications can mean that “perhaps we are speaking of the same pathogen in similar situations, believing that we are referring to different pathogens,” said Dr. Afeltra.
Clinical pearls related to emerging fungal pathogens
Candida auris. This organism has emerged simultaneously on several continents. It has pathogenicity factors typical of the genus, such as biofilm formation and production of phospholipases and proteinases, although it has greater thermal tolerance. In hospitals, it colonizes for weeks and months. In Argentina, it is resistant to multiple antifungal agents. Sensitivity is variable in different geographical regions. Most strains are resistant to fluconazole, and there is variable resistance to the other triazoles [which are not normally used to treat candidemia]. In the United States, in vitro resistance to amphotericin B is up to 30%, and resistance to echinocandins is up to 5%. New drugs such as rezafungin and ibrexafungerp are being studied. Infection control is similar to that used to control Clostridium difficile.
Fusarium. This genus affects immunocompromised patients, including transplant recipients of solid organs and hematopoietic progenitor cells and patients with neutropenia. The genus has various species, included within complexes, such as F. solani SC, F. oxysporum SC, and F. fujikuroi SC, with clinical manifestations similar to those of aspergillosis. In addition to the pulmonary and disseminated forms, there may be skin involvement attributable to dissemination from a respiratory focus or by contiguity from a focus of onychomycosis. In general, mortality is high, and responses to antifungal agents are variable. Some species are more sensitive to voriconazole or posaconazole, and others less so. All show in vitro resistance to itraconazole. In Argentina, voriconazole is usually used as initial treatment, and in special cases, liposomal amphotericin B or combinations. Fosmanogepix is being evaluated for the future.
Azole-resistant aspergillosis. This infection has shown resistance to itraconazole and third-generation azole drugs. In immunocompromised patients, mortlaity is high. Early detection is key. It is sensitive to amphotericin B and echinocandins. It is generally treated with liposomal amphotericin B. Olorofim and fosmanogepix are being studied.
Pulmonary aspergillosis associated with COVID-19. This infection is associated with high mortality among intubated patients. Signs and symptoms include fever, pleural effusion, hemoptysis, and chest pain, with infiltrates or cavitations on imaging. Determining the diagnosis is difficult. “We couldn’t perform lung biopsies, and it was difficult for us to get patients out of intensive care units for CT scans. We treated the proven cases. We treated the probable cases, and those that had a very low certainty of disease were also treated. We came across this emergency and tried to do the best we could,” said Dr. Afeltra. A digital readout lateral flow trial (Sona Aspergillus Galactomannan LFA) for the quantification of galactomannan, a cell wall component of the Aspergillus genus, proved to be a useful tool for screening and diagnosing patients with probable pulmonary aspergillosis associated with COVID-19. The incidence of invasive mycosis was around 10% among 185 seriously ill COVID-19 patients, according to an Argentine multicenter prospective study in which Dr. Afeltra participated.
Scedosporium and Lomentospora. These genera are rarer septate hyaline fungi. Scedosporium is a complex of species. One species, S. apiospermum, can colonize pediatric patients with cystic fibrosis. Lomentospora prolificans is a multiresistant fungus. It produces pulmonary compromise or disseminated infection. The response to antifungal agents is variable, with a high minimum inhibitory concentration for amphotericin B and isavuconazole. Patients are usually treated with voriconazole alone or in combination with terbinafine or micafungin. Olorofim is emerging as a promising treatment.
Dr. Afeltra has received fees from Biotoscana, Gador, Pfizer, Merck, and Sandoz.
This article was translated from the Medscape Spanish edition, a version appeared on Medscape.com.
AT SADI 2022
FDA approves oteseconazole for chronic yeast infections
The Food and Drug Administration has approved oteseconazole capsules (Vivjoa), an azole antifungal agent, for the prevention of recurrent yeast infections in women who are not of reproductive potential.
Oteseconazole inhibits CYP51, an enzyme fungi require to preserve the integrity of their cell walls and to grow properly, according to Mycovia, the drug’s manufacturer. It is the first FDA-approved product for the treatment of recurrent vulvovaginal candidiasis (RVVC).
Recurrent vulvovaginal candidiasis, or chronic yeast infection, affects an estimated 138 million women worldwide annually. The condition is defined as three or more symptomatic acute episodes of yeast infection within a 12-month period. The primary symptoms of RVVC include vaginal itching, burning, irritation, and inflammation. Some patients may also experience abnormal vaginal discharge and pain during sex or urination.
“A medicine with Vivjoa’s sustained efficacy combined with the clinical safety profile has been long needed, as until now, physicians and their patients have had no FDA-approved medications for RVVC,” Stephen Brand, PhD, chief development officer of Mycovia, said in a statement. “We are excited to be the first to offer a medication designed specifically for RVVC, a challenging and chronic condition that is expected to increase in prevalence over the next decade.”
Approval for oteseconazole was based on results of three phase 3 trials involving 875 patients at 232 sites across 11 countries. In the U.S.-only ultraVIOLET trial, 89.7% of women with RVVC who received oteseconazole cleared their initial yeast infection and did not experience a recurrence during the 50-week maintenance period, compared with 57.1% of those who received fluconazole (Diflucan) followed by placebo (P < .001), according to Mycovia.
The most common side effects reported in phase 3 clinical studies were headache (7.4%) and nausea (3.6%), the company said. Patients with a hypersensitivity to oteseconazole should not take the drug, nor should those who are of reproductive potential, pregnant, or lactating.
Mycovia said it plans to launch the drug in the second quarter of 2022.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved oteseconazole capsules (Vivjoa), an azole antifungal agent, for the prevention of recurrent yeast infections in women who are not of reproductive potential.
Oteseconazole inhibits CYP51, an enzyme fungi require to preserve the integrity of their cell walls and to grow properly, according to Mycovia, the drug’s manufacturer. It is the first FDA-approved product for the treatment of recurrent vulvovaginal candidiasis (RVVC).
Recurrent vulvovaginal candidiasis, or chronic yeast infection, affects an estimated 138 million women worldwide annually. The condition is defined as three or more symptomatic acute episodes of yeast infection within a 12-month period. The primary symptoms of RVVC include vaginal itching, burning, irritation, and inflammation. Some patients may also experience abnormal vaginal discharge and pain during sex or urination.
“A medicine with Vivjoa’s sustained efficacy combined with the clinical safety profile has been long needed, as until now, physicians and their patients have had no FDA-approved medications for RVVC,” Stephen Brand, PhD, chief development officer of Mycovia, said in a statement. “We are excited to be the first to offer a medication designed specifically for RVVC, a challenging and chronic condition that is expected to increase in prevalence over the next decade.”
Approval for oteseconazole was based on results of three phase 3 trials involving 875 patients at 232 sites across 11 countries. In the U.S.-only ultraVIOLET trial, 89.7% of women with RVVC who received oteseconazole cleared their initial yeast infection and did not experience a recurrence during the 50-week maintenance period, compared with 57.1% of those who received fluconazole (Diflucan) followed by placebo (P < .001), according to Mycovia.
The most common side effects reported in phase 3 clinical studies were headache (7.4%) and nausea (3.6%), the company said. Patients with a hypersensitivity to oteseconazole should not take the drug, nor should those who are of reproductive potential, pregnant, or lactating.
Mycovia said it plans to launch the drug in the second quarter of 2022.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved oteseconazole capsules (Vivjoa), an azole antifungal agent, for the prevention of recurrent yeast infections in women who are not of reproductive potential.
Oteseconazole inhibits CYP51, an enzyme fungi require to preserve the integrity of their cell walls and to grow properly, according to Mycovia, the drug’s manufacturer. It is the first FDA-approved product for the treatment of recurrent vulvovaginal candidiasis (RVVC).
Recurrent vulvovaginal candidiasis, or chronic yeast infection, affects an estimated 138 million women worldwide annually. The condition is defined as three or more symptomatic acute episodes of yeast infection within a 12-month period. The primary symptoms of RVVC include vaginal itching, burning, irritation, and inflammation. Some patients may also experience abnormal vaginal discharge and pain during sex or urination.
“A medicine with Vivjoa’s sustained efficacy combined with the clinical safety profile has been long needed, as until now, physicians and their patients have had no FDA-approved medications for RVVC,” Stephen Brand, PhD, chief development officer of Mycovia, said in a statement. “We are excited to be the first to offer a medication designed specifically for RVVC, a challenging and chronic condition that is expected to increase in prevalence over the next decade.”
Approval for oteseconazole was based on results of three phase 3 trials involving 875 patients at 232 sites across 11 countries. In the U.S.-only ultraVIOLET trial, 89.7% of women with RVVC who received oteseconazole cleared their initial yeast infection and did not experience a recurrence during the 50-week maintenance period, compared with 57.1% of those who received fluconazole (Diflucan) followed by placebo (P < .001), according to Mycovia.
The most common side effects reported in phase 3 clinical studies were headache (7.4%) and nausea (3.6%), the company said. Patients with a hypersensitivity to oteseconazole should not take the drug, nor should those who are of reproductive potential, pregnant, or lactating.
Mycovia said it plans to launch the drug in the second quarter of 2022.
Full prescribing information is available online.
A version of this article first appeared on Medscape.com.