User login
What will pediatrics look like in 2022?
In 1966 I was struggling with the decision of whether to become an art historian or go to medical school. I decided corporate ladder climbs and tenure chases were not for me. I wanted to be my own boss. I reckoned that medicine would offer me rock-solid job security and a comfortable income that I could adjust to my needs simply by working harder. In my Norman Rockwell–influenced view of the world, there would always be sick children. There would never be a quiet week or even a day when I would have to worry about not having an income.
So it was an idyllic existence for decades, tarnished only slightly when corporate entities began gobbling up owner-operator practices. But I never envisioned a pandemic that would turn the world – including its pediatricians – upside down. For the last several weeks as I pedal past my old office, I am dumbstruck by the empty parking lot. For the present I appear to be buffered by my retirement, but know that many of you are under serious financial pressure as a result of the pandemic.
We are all yearning to return to business as usual, but we know that it isn’t going to happen because everything has changed. The usual has yet to be defined. When you finally reopen your offices, you will be walking into a strange and eerie new normal. Initially you may struggle to make it feel like nothing has changed, but very quickly the full force of the postpandemic tsunami will hit us all broadside. In 2 years, the ship may still be rocking but what will clinical pediatrics look like in the late spring of 2022?
Will the patient mix have shifted even more toward behavioral and mental health complaints as a ripple effect of the pandemic’s emotional turmoil? Will your waiting room have become a maze of plexiglass barriers to separate the sick from the well? Has the hospital invested hundreds of thousands of dollars in a ventilation system in hopes of minimizing contagion in your exam rooms? Maybe you will have instituted an appointment schedule with sick visits in the morning and well checks in the afternoon. Or you may no longer have a waiting room because patients are queuing in their cars in the parking lot. Your support staff may be rollerskating around like carhops at a drive-in recording histories and taking vital signs.
Telemedicine will hopefully have gone mainstream with more robust guidelines for billing and quality control. Medical schools may be devoting more attention to teaching student how to assess remotely. Parents may now be equipped with a tool kit of remote sensors so that you can assess their child’s tympanic membranes, pulse rate, oxygen saturation, and blood pressure on your office computer screen.
Will the EHR finally have begun to emerge from its awkward and at times painful adolescence into an easily accessible and transportable nationwide data bank that includes immunization records for all ages? Patients may have been asked or ordered to allow their cell phones to be used as tracking devices for serious communicable diseases. How many vaccine-resistant people will have responded to the pandemic by deciding that immunizations are worth the minimal risks? I fear not many.
How many of your colleagues will have left pediatrics and heeded the call for more epidemiologists? Will you be required to take a CME course in ventilation management? The good news may be that to keep the pediatric workforce robust the government has decided to forgive your student loans.
None of these changes may have come to pass because we have notoriously short memories. But I am sure that we will all still bear the deep scars of this world changing event.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
In 1966 I was struggling with the decision of whether to become an art historian or go to medical school. I decided corporate ladder climbs and tenure chases were not for me. I wanted to be my own boss. I reckoned that medicine would offer me rock-solid job security and a comfortable income that I could adjust to my needs simply by working harder. In my Norman Rockwell–influenced view of the world, there would always be sick children. There would never be a quiet week or even a day when I would have to worry about not having an income.
So it was an idyllic existence for decades, tarnished only slightly when corporate entities began gobbling up owner-operator practices. But I never envisioned a pandemic that would turn the world – including its pediatricians – upside down. For the last several weeks as I pedal past my old office, I am dumbstruck by the empty parking lot. For the present I appear to be buffered by my retirement, but know that many of you are under serious financial pressure as a result of the pandemic.
We are all yearning to return to business as usual, but we know that it isn’t going to happen because everything has changed. The usual has yet to be defined. When you finally reopen your offices, you will be walking into a strange and eerie new normal. Initially you may struggle to make it feel like nothing has changed, but very quickly the full force of the postpandemic tsunami will hit us all broadside. In 2 years, the ship may still be rocking but what will clinical pediatrics look like in the late spring of 2022?
Will the patient mix have shifted even more toward behavioral and mental health complaints as a ripple effect of the pandemic’s emotional turmoil? Will your waiting room have become a maze of plexiglass barriers to separate the sick from the well? Has the hospital invested hundreds of thousands of dollars in a ventilation system in hopes of minimizing contagion in your exam rooms? Maybe you will have instituted an appointment schedule with sick visits in the morning and well checks in the afternoon. Or you may no longer have a waiting room because patients are queuing in their cars in the parking lot. Your support staff may be rollerskating around like carhops at a drive-in recording histories and taking vital signs.
Telemedicine will hopefully have gone mainstream with more robust guidelines for billing and quality control. Medical schools may be devoting more attention to teaching student how to assess remotely. Parents may now be equipped with a tool kit of remote sensors so that you can assess their child’s tympanic membranes, pulse rate, oxygen saturation, and blood pressure on your office computer screen.
Will the EHR finally have begun to emerge from its awkward and at times painful adolescence into an easily accessible and transportable nationwide data bank that includes immunization records for all ages? Patients may have been asked or ordered to allow their cell phones to be used as tracking devices for serious communicable diseases. How many vaccine-resistant people will have responded to the pandemic by deciding that immunizations are worth the minimal risks? I fear not many.
How many of your colleagues will have left pediatrics and heeded the call for more epidemiologists? Will you be required to take a CME course in ventilation management? The good news may be that to keep the pediatric workforce robust the government has decided to forgive your student loans.
None of these changes may have come to pass because we have notoriously short memories. But I am sure that we will all still bear the deep scars of this world changing event.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
In 1966 I was struggling with the decision of whether to become an art historian or go to medical school. I decided corporate ladder climbs and tenure chases were not for me. I wanted to be my own boss. I reckoned that medicine would offer me rock-solid job security and a comfortable income that I could adjust to my needs simply by working harder. In my Norman Rockwell–influenced view of the world, there would always be sick children. There would never be a quiet week or even a day when I would have to worry about not having an income.
So it was an idyllic existence for decades, tarnished only slightly when corporate entities began gobbling up owner-operator practices. But I never envisioned a pandemic that would turn the world – including its pediatricians – upside down. For the last several weeks as I pedal past my old office, I am dumbstruck by the empty parking lot. For the present I appear to be buffered by my retirement, but know that many of you are under serious financial pressure as a result of the pandemic.
We are all yearning to return to business as usual, but we know that it isn’t going to happen because everything has changed. The usual has yet to be defined. When you finally reopen your offices, you will be walking into a strange and eerie new normal. Initially you may struggle to make it feel like nothing has changed, but very quickly the full force of the postpandemic tsunami will hit us all broadside. In 2 years, the ship may still be rocking but what will clinical pediatrics look like in the late spring of 2022?
Will the patient mix have shifted even more toward behavioral and mental health complaints as a ripple effect of the pandemic’s emotional turmoil? Will your waiting room have become a maze of plexiglass barriers to separate the sick from the well? Has the hospital invested hundreds of thousands of dollars in a ventilation system in hopes of minimizing contagion in your exam rooms? Maybe you will have instituted an appointment schedule with sick visits in the morning and well checks in the afternoon. Or you may no longer have a waiting room because patients are queuing in their cars in the parking lot. Your support staff may be rollerskating around like carhops at a drive-in recording histories and taking vital signs.
Telemedicine will hopefully have gone mainstream with more robust guidelines for billing and quality control. Medical schools may be devoting more attention to teaching student how to assess remotely. Parents may now be equipped with a tool kit of remote sensors so that you can assess their child’s tympanic membranes, pulse rate, oxygen saturation, and blood pressure on your office computer screen.
Will the EHR finally have begun to emerge from its awkward and at times painful adolescence into an easily accessible and transportable nationwide data bank that includes immunization records for all ages? Patients may have been asked or ordered to allow their cell phones to be used as tracking devices for serious communicable diseases. How many vaccine-resistant people will have responded to the pandemic by deciding that immunizations are worth the minimal risks? I fear not many.
How many of your colleagues will have left pediatrics and heeded the call for more epidemiologists? Will you be required to take a CME course in ventilation management? The good news may be that to keep the pediatric workforce robust the government has decided to forgive your student loans.
None of these changes may have come to pass because we have notoriously short memories. But I am sure that we will all still bear the deep scars of this world changing event.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Email him at pdnews@mdedge.com.
The cataclysmic COVID-19 pandemic: THIS CHANGES EVERYTHING!
It was unthinkable, but it has happened. A virulent and invisible virus, 10 microns in size, with no vaccine or cure yet, shut down our nation, the third largest country in the world with 330 million people. Overnight, our thriving cities became ghost towns. Schools were closed. Millions of businesses, restaurants, and stores were abruptly shuttered. Sporting events were instantly canceled. Air travel came to a halt.
The largest economy in the world started to tank. Millions of people lost their jobs and were forced to stay home. The vital structures of society were dismantled. Our vibrant culture came to a screeching halt. It’s a nightmare scenario that even the most imaginative science fiction writers could not have envisioned. By any measure, the coronavirus disease 2019 (COVID-19) pandemic unraveled everything, and became a human catastrophe and a social calamity reminiscent of the deadly 1918 influenza pandemic, or the devastating plagues that decimated Europe during the Middle Ages.
The human toll in death and suffering was the real disaster. Emergency departments and hospitals filled up with victims of the scourge, sickly and unable to breathe as the virus hijacked their red blood cells and lungs, and destroyed their alveoli. Compounding the disaster was a lack of medical supplies. The country was clearly caught off-guard, completely unprepared for the scale of the pandemic and the massive onslaught of desperately ill people requiring intensive care and special equipment. In addition, health care staff became stretched beyond the limit, and entire hospitals were transformed overnight into highly specialized ICUs. Medical care for millions with non-COVID-19 conditions was put on hold so that vital resources could be diverted to the desperately ill victims of these infections. Many physicians, nurses, and respiratory therapists—laudable heroes—succumbed to the perverse virus exhaled by their patients.
Insidious social effects
COVID-19 is not only a murderer, but also a thief. It stole our Spring; our religious holidays (Easter and Passover); classroom education in schools and colleges; the Prom; weddings; graduation ceremonies; proper funerals; concerts; football, basketball, hockey, and baseball games; Broadway shows; and even data from animal research. More important, it robbed us of our peace of mind, our liberty, and our pursuit of small pleasures such as family gatherings or schmoozing with friends at a nice restaurant. COVID-19 is a cruel, dastardly scoundrel.
I write this editorial as I sit at home, which I have not left for several weeks, like hundreds of millions in our country and around the world. We were all glued to TV news or the internet to learn about the latest updates, including the grim news of those who got infected, hospitalized, or passed away. Fear of dying permeated all age groups, especially those who were older and infirmed.
Making it worse was the relentless uncertainty. When will it end? Gradually or suddenly? When is it going to be safe to go to work again, or to visit our loved ones and our friends? When can we see our patients face-to-face instead of remotely by phone or video conferencing? When can we have live meetings instead of virtual video conferences? When will stores open so we can shop? When can we take our children or grandchildren to a baseball game or a show? Will the virus return next winter for another cycle of mayhem and social paralysis? When will the economy start to rebound, and how long will that take? Will our retirement accounts recoup their losses? So many questions with no clear answers. A malignant uncertainty, indeed.
And there are our patients who live with anxiety and depression, whose anguish is intensifying as they sit alone in their apartments or homes, struggling to cope with this sudden, overwhelming stress. How will they react to this pandemic? Obviously, a life-threatening event such as a deadly pandemic with no cure is likely to produce an acute stress reaction and, ultimately, posttraumatic stress disorder (PTSD). And if COVID-19 returns next year for another unwelcome visit, PTSD symptoms will get a booster shot and lead to severe anxiety, depression, or suicide. Psychiatrists and other mental health professionals, who were already stretched thin, must contend with another crisis that has destabilized millions of patients receiving psychiatric care, or new patients who seek help for themselves or their family members.
Continue to: One intervention that is emerging...
One intervention that is emerging on a large scale is online therapy. This includes reassurance and supportive therapy, cognitive-behavioral therapy, relaxation techniques, stress management, resilience training, mindfulness, and online group therapy. Those therapies can be effective for stress-induced anxiety and dysphoria when pharmacotherapy is not available, and can provide patients with tools and techniques that can be implemented by the patients themselves in the absence of a physician or nurse practitioner to prescribe a medication.
Lessons learned
This pandemic has taught us many lessons: that life as we know it should not be taken for granted, and can change drastically overnight; that human life is fragile and can be destroyed rapidly and ruthlessly on an unimaginable scale by an invisible enemy; that scientific drug development research by the often maligned pharmaceutical industry is indispensable to our well-being; that policymakers must always prepare for the worst and must have a well-designed disaster plan; that modifying human behavior and full compliance with public health measures are vital and can be the most effective way to prevent the spread of catastrophic pandemics, viral or otherwise; that we must all learn how to be resilient to cope with solitude and restricted mobility or socialization; that the human ingenuity and innovation that created technologies to enable virtual connectivity among us, even when we are isolated, has been a lifesaver during health crises such as the COVID-19 pandemic; that the clinicians and health care workers treating highly infectious and desperately ill patients are genuine heroes who deserve our respect and gratitude; and that magnificent altruism outstrips and outshines the selfish hoarding and profiteering that may emerge during life-threatening pandemics.
And that we shall overcome this horrid pandemic, a ghastly tribulation that changed everything.
It was unthinkable, but it has happened. A virulent and invisible virus, 10 microns in size, with no vaccine or cure yet, shut down our nation, the third largest country in the world with 330 million people. Overnight, our thriving cities became ghost towns. Schools were closed. Millions of businesses, restaurants, and stores were abruptly shuttered. Sporting events were instantly canceled. Air travel came to a halt.
The largest economy in the world started to tank. Millions of people lost their jobs and were forced to stay home. The vital structures of society were dismantled. Our vibrant culture came to a screeching halt. It’s a nightmare scenario that even the most imaginative science fiction writers could not have envisioned. By any measure, the coronavirus disease 2019 (COVID-19) pandemic unraveled everything, and became a human catastrophe and a social calamity reminiscent of the deadly 1918 influenza pandemic, or the devastating plagues that decimated Europe during the Middle Ages.
The human toll in death and suffering was the real disaster. Emergency departments and hospitals filled up with victims of the scourge, sickly and unable to breathe as the virus hijacked their red blood cells and lungs, and destroyed their alveoli. Compounding the disaster was a lack of medical supplies. The country was clearly caught off-guard, completely unprepared for the scale of the pandemic and the massive onslaught of desperately ill people requiring intensive care and special equipment. In addition, health care staff became stretched beyond the limit, and entire hospitals were transformed overnight into highly specialized ICUs. Medical care for millions with non-COVID-19 conditions was put on hold so that vital resources could be diverted to the desperately ill victims of these infections. Many physicians, nurses, and respiratory therapists—laudable heroes—succumbed to the perverse virus exhaled by their patients.
Insidious social effects
COVID-19 is not only a murderer, but also a thief. It stole our Spring; our religious holidays (Easter and Passover); classroom education in schools and colleges; the Prom; weddings; graduation ceremonies; proper funerals; concerts; football, basketball, hockey, and baseball games; Broadway shows; and even data from animal research. More important, it robbed us of our peace of mind, our liberty, and our pursuit of small pleasures such as family gatherings or schmoozing with friends at a nice restaurant. COVID-19 is a cruel, dastardly scoundrel.
I write this editorial as I sit at home, which I have not left for several weeks, like hundreds of millions in our country and around the world. We were all glued to TV news or the internet to learn about the latest updates, including the grim news of those who got infected, hospitalized, or passed away. Fear of dying permeated all age groups, especially those who were older and infirmed.
Making it worse was the relentless uncertainty. When will it end? Gradually or suddenly? When is it going to be safe to go to work again, or to visit our loved ones and our friends? When can we see our patients face-to-face instead of remotely by phone or video conferencing? When can we have live meetings instead of virtual video conferences? When will stores open so we can shop? When can we take our children or grandchildren to a baseball game or a show? Will the virus return next winter for another cycle of mayhem and social paralysis? When will the economy start to rebound, and how long will that take? Will our retirement accounts recoup their losses? So many questions with no clear answers. A malignant uncertainty, indeed.
And there are our patients who live with anxiety and depression, whose anguish is intensifying as they sit alone in their apartments or homes, struggling to cope with this sudden, overwhelming stress. How will they react to this pandemic? Obviously, a life-threatening event such as a deadly pandemic with no cure is likely to produce an acute stress reaction and, ultimately, posttraumatic stress disorder (PTSD). And if COVID-19 returns next year for another unwelcome visit, PTSD symptoms will get a booster shot and lead to severe anxiety, depression, or suicide. Psychiatrists and other mental health professionals, who were already stretched thin, must contend with another crisis that has destabilized millions of patients receiving psychiatric care, or new patients who seek help for themselves or their family members.
Continue to: One intervention that is emerging...
One intervention that is emerging on a large scale is online therapy. This includes reassurance and supportive therapy, cognitive-behavioral therapy, relaxation techniques, stress management, resilience training, mindfulness, and online group therapy. Those therapies can be effective for stress-induced anxiety and dysphoria when pharmacotherapy is not available, and can provide patients with tools and techniques that can be implemented by the patients themselves in the absence of a physician or nurse practitioner to prescribe a medication.
Lessons learned
This pandemic has taught us many lessons: that life as we know it should not be taken for granted, and can change drastically overnight; that human life is fragile and can be destroyed rapidly and ruthlessly on an unimaginable scale by an invisible enemy; that scientific drug development research by the often maligned pharmaceutical industry is indispensable to our well-being; that policymakers must always prepare for the worst and must have a well-designed disaster plan; that modifying human behavior and full compliance with public health measures are vital and can be the most effective way to prevent the spread of catastrophic pandemics, viral or otherwise; that we must all learn how to be resilient to cope with solitude and restricted mobility or socialization; that the human ingenuity and innovation that created technologies to enable virtual connectivity among us, even when we are isolated, has been a lifesaver during health crises such as the COVID-19 pandemic; that the clinicians and health care workers treating highly infectious and desperately ill patients are genuine heroes who deserve our respect and gratitude; and that magnificent altruism outstrips and outshines the selfish hoarding and profiteering that may emerge during life-threatening pandemics.
And that we shall overcome this horrid pandemic, a ghastly tribulation that changed everything.
It was unthinkable, but it has happened. A virulent and invisible virus, 10 microns in size, with no vaccine or cure yet, shut down our nation, the third largest country in the world with 330 million people. Overnight, our thriving cities became ghost towns. Schools were closed. Millions of businesses, restaurants, and stores were abruptly shuttered. Sporting events were instantly canceled. Air travel came to a halt.
The largest economy in the world started to tank. Millions of people lost their jobs and were forced to stay home. The vital structures of society were dismantled. Our vibrant culture came to a screeching halt. It’s a nightmare scenario that even the most imaginative science fiction writers could not have envisioned. By any measure, the coronavirus disease 2019 (COVID-19) pandemic unraveled everything, and became a human catastrophe and a social calamity reminiscent of the deadly 1918 influenza pandemic, or the devastating plagues that decimated Europe during the Middle Ages.
The human toll in death and suffering was the real disaster. Emergency departments and hospitals filled up with victims of the scourge, sickly and unable to breathe as the virus hijacked their red blood cells and lungs, and destroyed their alveoli. Compounding the disaster was a lack of medical supplies. The country was clearly caught off-guard, completely unprepared for the scale of the pandemic and the massive onslaught of desperately ill people requiring intensive care and special equipment. In addition, health care staff became stretched beyond the limit, and entire hospitals were transformed overnight into highly specialized ICUs. Medical care for millions with non-COVID-19 conditions was put on hold so that vital resources could be diverted to the desperately ill victims of these infections. Many physicians, nurses, and respiratory therapists—laudable heroes—succumbed to the perverse virus exhaled by their patients.
Insidious social effects
COVID-19 is not only a murderer, but also a thief. It stole our Spring; our religious holidays (Easter and Passover); classroom education in schools and colleges; the Prom; weddings; graduation ceremonies; proper funerals; concerts; football, basketball, hockey, and baseball games; Broadway shows; and even data from animal research. More important, it robbed us of our peace of mind, our liberty, and our pursuit of small pleasures such as family gatherings or schmoozing with friends at a nice restaurant. COVID-19 is a cruel, dastardly scoundrel.
I write this editorial as I sit at home, which I have not left for several weeks, like hundreds of millions in our country and around the world. We were all glued to TV news or the internet to learn about the latest updates, including the grim news of those who got infected, hospitalized, or passed away. Fear of dying permeated all age groups, especially those who were older and infirmed.
Making it worse was the relentless uncertainty. When will it end? Gradually or suddenly? When is it going to be safe to go to work again, or to visit our loved ones and our friends? When can we see our patients face-to-face instead of remotely by phone or video conferencing? When can we have live meetings instead of virtual video conferences? When will stores open so we can shop? When can we take our children or grandchildren to a baseball game or a show? Will the virus return next winter for another cycle of mayhem and social paralysis? When will the economy start to rebound, and how long will that take? Will our retirement accounts recoup their losses? So many questions with no clear answers. A malignant uncertainty, indeed.
And there are our patients who live with anxiety and depression, whose anguish is intensifying as they sit alone in their apartments or homes, struggling to cope with this sudden, overwhelming stress. How will they react to this pandemic? Obviously, a life-threatening event such as a deadly pandemic with no cure is likely to produce an acute stress reaction and, ultimately, posttraumatic stress disorder (PTSD). And if COVID-19 returns next year for another unwelcome visit, PTSD symptoms will get a booster shot and lead to severe anxiety, depression, or suicide. Psychiatrists and other mental health professionals, who were already stretched thin, must contend with another crisis that has destabilized millions of patients receiving psychiatric care, or new patients who seek help for themselves or their family members.
Continue to: One intervention that is emerging...
One intervention that is emerging on a large scale is online therapy. This includes reassurance and supportive therapy, cognitive-behavioral therapy, relaxation techniques, stress management, resilience training, mindfulness, and online group therapy. Those therapies can be effective for stress-induced anxiety and dysphoria when pharmacotherapy is not available, and can provide patients with tools and techniques that can be implemented by the patients themselves in the absence of a physician or nurse practitioner to prescribe a medication.
Lessons learned
This pandemic has taught us many lessons: that life as we know it should not be taken for granted, and can change drastically overnight; that human life is fragile and can be destroyed rapidly and ruthlessly on an unimaginable scale by an invisible enemy; that scientific drug development research by the often maligned pharmaceutical industry is indispensable to our well-being; that policymakers must always prepare for the worst and must have a well-designed disaster plan; that modifying human behavior and full compliance with public health measures are vital and can be the most effective way to prevent the spread of catastrophic pandemics, viral or otherwise; that we must all learn how to be resilient to cope with solitude and restricted mobility or socialization; that the human ingenuity and innovation that created technologies to enable virtual connectivity among us, even when we are isolated, has been a lifesaver during health crises such as the COVID-19 pandemic; that the clinicians and health care workers treating highly infectious and desperately ill patients are genuine heroes who deserve our respect and gratitude; and that magnificent altruism outstrips and outshines the selfish hoarding and profiteering that may emerge during life-threatening pandemics.
And that we shall overcome this horrid pandemic, a ghastly tribulation that changed everything.
Global registry collects data on pediatric cancer patients with COVID-19
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
Angiotensin drugs and COVID-19: More reassuring data
Initial data from one Chinese center on the use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) in patients hospitalized with COVID-19 appear to give some further reassurance about continued use of these drugs.
The report from one hospital in Wuhan found that among patients with hypertension hospitalized with the COVID-19 virus, there was no difference in disease severity or death rate in patients taking ACE inhibitors or ARBs and those not taking such medications.
The data were published online April 23 in JAMA Cardiology.
The study adds to another recent report in a larger number of COVID-19 patients from nine Chinese hospitals that suggested a beneficial effect of ACE inhibitors or ARBs on mortality.
Additional studies
Two other similar studies have also been recently released. Another study from China, published online March 31 in Emerging Microbes & Infections, included a small sample of 42 hospitalized patients with COVID-19 on antihypertensive therapy. Those on ACE inhibitor/ARB therapy had a lower rate of severe disease and a trend toward a lower level of IL-6 in peripheral blood. In addition, patients on ACE inhibitor/ARB therapy had increased CD3+ and CD8+ T-cell counts in peripheral blood and decreased peak viral load compared with other antihypertensive drugs.
And a preliminary study from the UK, which has not yet been peer reviewed, found that treatment with ACE inhibitors was associated with a reduced risk of rapidly deteriorating severe COVID-19 disease.
The study, available online on MedRxiv, a preprint server for health sciences, reports on 205 acute inpatients with COVID-19 at King’s College Hospital and Princess Royal University Hospital, London.
Of these, 51.2% had hypertension, 30.2% had diabetes, and 14.6% had ischemic heart disease or heart failure. Of the 37 patients on ACE inhibitors, five (14%) died or required critical care support compared with 29% (48/168) of patients not taking an ACE inhibitor.
New Wuhan study
The authors of the new article published in JAMA Cardiology, led by Juyi Li, MD, reported on a case series of 1,178 patients hospitalized with COVID-19 at the Central Hospital of Wuhan, Hubei, China, between Jan. 15 and March 15, 2020.
Patients were a median age of 55 years, and 46% were men. They had an overall in-hospital mortality rate of 11%.
Of the 1,178 patients, 362 (30.7%) had a diagnosis of hypertension. These patients were older (median age, 66 years) and had a greater prevalence of chronic diseases. Patients with hypertension also had more severe manifestations of COVID-19 compared to those without hypertension, including higher rates of acute respiratory distress syndrome and in-hospital mortality (21.3% vs. 6.5%).
Of the 362 patients with hypertension, 31.8% were taking ACE inhibitors or ARBs.
Apart from a greater prevalence of coronary artery disease, patients taking ACE inhibitors or ARBs had similar comorbidities to those not taking these medications, and also similar laboratory profile results including blood counts, inflammatory markers, renal and liver function tests, and cardiac biomarkers, although those taking ACE inhibitors/ARBs had higher levels of alkaline phosphatase.
The most commonly used antihypertensive drugs were calcium blockers. The percentage of patients with hypertension taking any drug or drug combination did not differ between those with severe and nonsevere infections and between those who survived and those who died.
Specifically regarding ACE inhibitors/ARBs, there was no difference between those with severe versus nonsevere illness in the use of ACE inhibitors (9.2% vs. 10.1%; P = .80), ARBs (24.9% vs. 21.2%; P = .40), or the composite of ACE inhibitors or ARBs (32.9% vs. 30.7%; P = .65).
Similarly, there were no differences in nonsurvivors and survivors in the use of ACE inhibitors (9.1% vs. 9.8%; P = .85); ARBs (19.5% vs. 23.9%; P = .42), or the composite of ACE inhibitors or ARBs (27.3% vs. 33.0%; P = .34).
The frequency of severe illness and death also did not differ between those treated with and without ACE inhibitors/ARBs in patients with hypertension and other various chronic conditions including coronary heart disease, cerebrovascular disease, diabetes, neurological disease, and chronic renal disease.
The authors noted that these data confirm previous reports showing that patients with hypertension have more severe illness and higher mortality rates associated with COVID-19 than those without hypertension.
But they added: “Our data provide some reassurance that ACE inhibitors/ARBs are not associated with the progression or outcome of COVID-19 hospitalizations in patients with hypertension.”
They also noted that these results support the recommendations from almost all major cardiovascular societies that patients do not discontinue ACE inhibitors or ARBs because of worries about COVID-19.
However, the authors did point out some limitations of their study, which included a small number of patients with hypertension taking ACE inhibitors or ARBs and the fact that a nonsevere disease course was still severe enough to require hospitalization. In addition, it was not clear whether ACE inhibitor/ARB treatment at baseline was maintained throughout hospitalization for all patients.
This was also an observational comparison and may be biased by differences in patients taking versus not taking ACE inhibitors or ARBs at the time of hospitalization, although the measured baseline characteristics were similar in both groups.
But the authors also highlighted the finding that, in this cohort, patients with hypertension had three times the mortality rate of all other patients hospitalized with COVID-19.
“Hypertension combined with cardiovascular and cerebrovascular disease, diabetes, and chronic kidney disease would predispose patients to an increased risk of severity and mortality of COVID-19. Therefore, patients with these underlying conditions who develop COVID-19 require particularly intensive surveillance and care,” they wrote.
Experts cautiously optimistic
Some cardiovascular experts were cautiously optimistic about these latest results.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, and editor-in-chief of the Journal of Clinical Hypertension, said: “This new report from Wuhan, China, gives modest reassurance that the use of ACE inhibitors or ARBs in hypertensive patients with COVID-19 disease does not increase the risk of clinical deterioration or death.
“Ongoing, more definitive studies should help resolve competing hypotheses regarding the effects of these agents: whether the increased ACE2 enzyme levels they produce can worsen outcomes by increasing access of the COVID virus to lung tissue; or whether there is a benefit linked to a protective effect of increased ACE2 on alveolar cell function,” Dr. Weber noted.
“Though the number of patients included in this new report is small, it is startling that hypertensive patients were three times as likely as nonhypertensives to have a fatal outcome, presumably reflecting vulnerability due to the cardiovascular and metabolic comorbidities associated with hypertension,” he added.
“In any case, for now, clinicians should continue treating hypertensive patients with whichever drugs, including ACE inhibitors and ARBs, best provide protection from adverse outcomes,” Dr. Weber concluded.
John McMurray, MD, professor of medical cardiology, University of Glasgow, Scotland, commented: “This study from Wuhan provides some reassurance about one of the two questions about ACEI/ARBs: Do these drugs increase susceptibility to infection? And if [the patient is] infected, do they increase the severity of infection? This study addresses the latter question and appears to suggest no increased severity.”
However, Dr. McMurray pointed out that the study had many limitations. There were only small patient numbers and the data were unadjusted, “although it looks like the ACE inhibitor/ARB treated patients were higher risk to start with.” It was an observational study, and patients were not randomized and were predominantly treated with ARBs, and not ACE inhibitors, so “we don’t know if the concerns apply equally to these two classes of drug.
“Other data published and unpublished supporting this (even showing better outcomes in patients treated with an ACE inhibitor/ARB), and, to date, any concerns about these drugs remain unsubstantiated and the guidance from medical societies to continue treatment with these agents in patients prescribed them seems wise,” Dr. McMurray added.
Franz H. Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “The study from Wuhan is not a great study. They didn’t even do a multivariable analysis. They could have done a bit more with the data, but it still gives some reassurance.”
Dr. Messerli said it was “interesting” that 30% of the patients hospitalized with COVID-19 in the sample had hypertension. “That corresponds to the general population, so does not suggest that having hypertension increases susceptibility to infection – but it does seem to increase the risk of a bad outcome.”
Dr. Messerli noted that there are two more similar studies due to be published soon, both said to suggest either a beneficial or neutral effect of ACE inhibitors/ARBs on COVID-19 outcomes in hospitalized patients.
“This does help with confidence in prescribing these agents and reinforces the recommendations for patients to stay on these drugs,” he said.
“However, none of these studies address the infectivity issue – whether their use upregulates the ACE2 receptor, which the virus uses to gain entry to cells, thereby increasing susceptibility to the infection,” Dr. Messerli cautioned. “But the similar or better outcomes on these drugs are encouraging,” he added.
The Wuhan study was supported by the Health and Family Planning Commission of Wuhan City, China. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Initial data from one Chinese center on the use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) in patients hospitalized with COVID-19 appear to give some further reassurance about continued use of these drugs.
The report from one hospital in Wuhan found that among patients with hypertension hospitalized with the COVID-19 virus, there was no difference in disease severity or death rate in patients taking ACE inhibitors or ARBs and those not taking such medications.
The data were published online April 23 in JAMA Cardiology.
The study adds to another recent report in a larger number of COVID-19 patients from nine Chinese hospitals that suggested a beneficial effect of ACE inhibitors or ARBs on mortality.
Additional studies
Two other similar studies have also been recently released. Another study from China, published online March 31 in Emerging Microbes & Infections, included a small sample of 42 hospitalized patients with COVID-19 on antihypertensive therapy. Those on ACE inhibitor/ARB therapy had a lower rate of severe disease and a trend toward a lower level of IL-6 in peripheral blood. In addition, patients on ACE inhibitor/ARB therapy had increased CD3+ and CD8+ T-cell counts in peripheral blood and decreased peak viral load compared with other antihypertensive drugs.
And a preliminary study from the UK, which has not yet been peer reviewed, found that treatment with ACE inhibitors was associated with a reduced risk of rapidly deteriorating severe COVID-19 disease.
The study, available online on MedRxiv, a preprint server for health sciences, reports on 205 acute inpatients with COVID-19 at King’s College Hospital and Princess Royal University Hospital, London.
Of these, 51.2% had hypertension, 30.2% had diabetes, and 14.6% had ischemic heart disease or heart failure. Of the 37 patients on ACE inhibitors, five (14%) died or required critical care support compared with 29% (48/168) of patients not taking an ACE inhibitor.
New Wuhan study
The authors of the new article published in JAMA Cardiology, led by Juyi Li, MD, reported on a case series of 1,178 patients hospitalized with COVID-19 at the Central Hospital of Wuhan, Hubei, China, between Jan. 15 and March 15, 2020.
Patients were a median age of 55 years, and 46% were men. They had an overall in-hospital mortality rate of 11%.
Of the 1,178 patients, 362 (30.7%) had a diagnosis of hypertension. These patients were older (median age, 66 years) and had a greater prevalence of chronic diseases. Patients with hypertension also had more severe manifestations of COVID-19 compared to those without hypertension, including higher rates of acute respiratory distress syndrome and in-hospital mortality (21.3% vs. 6.5%).
Of the 362 patients with hypertension, 31.8% were taking ACE inhibitors or ARBs.
Apart from a greater prevalence of coronary artery disease, patients taking ACE inhibitors or ARBs had similar comorbidities to those not taking these medications, and also similar laboratory profile results including blood counts, inflammatory markers, renal and liver function tests, and cardiac biomarkers, although those taking ACE inhibitors/ARBs had higher levels of alkaline phosphatase.
The most commonly used antihypertensive drugs were calcium blockers. The percentage of patients with hypertension taking any drug or drug combination did not differ between those with severe and nonsevere infections and between those who survived and those who died.
Specifically regarding ACE inhibitors/ARBs, there was no difference between those with severe versus nonsevere illness in the use of ACE inhibitors (9.2% vs. 10.1%; P = .80), ARBs (24.9% vs. 21.2%; P = .40), or the composite of ACE inhibitors or ARBs (32.9% vs. 30.7%; P = .65).
Similarly, there were no differences in nonsurvivors and survivors in the use of ACE inhibitors (9.1% vs. 9.8%; P = .85); ARBs (19.5% vs. 23.9%; P = .42), or the composite of ACE inhibitors or ARBs (27.3% vs. 33.0%; P = .34).
The frequency of severe illness and death also did not differ between those treated with and without ACE inhibitors/ARBs in patients with hypertension and other various chronic conditions including coronary heart disease, cerebrovascular disease, diabetes, neurological disease, and chronic renal disease.
The authors noted that these data confirm previous reports showing that patients with hypertension have more severe illness and higher mortality rates associated with COVID-19 than those without hypertension.
But they added: “Our data provide some reassurance that ACE inhibitors/ARBs are not associated with the progression or outcome of COVID-19 hospitalizations in patients with hypertension.”
They also noted that these results support the recommendations from almost all major cardiovascular societies that patients do not discontinue ACE inhibitors or ARBs because of worries about COVID-19.
However, the authors did point out some limitations of their study, which included a small number of patients with hypertension taking ACE inhibitors or ARBs and the fact that a nonsevere disease course was still severe enough to require hospitalization. In addition, it was not clear whether ACE inhibitor/ARB treatment at baseline was maintained throughout hospitalization for all patients.
This was also an observational comparison and may be biased by differences in patients taking versus not taking ACE inhibitors or ARBs at the time of hospitalization, although the measured baseline characteristics were similar in both groups.
But the authors also highlighted the finding that, in this cohort, patients with hypertension had three times the mortality rate of all other patients hospitalized with COVID-19.
“Hypertension combined with cardiovascular and cerebrovascular disease, diabetes, and chronic kidney disease would predispose patients to an increased risk of severity and mortality of COVID-19. Therefore, patients with these underlying conditions who develop COVID-19 require particularly intensive surveillance and care,” they wrote.
Experts cautiously optimistic
Some cardiovascular experts were cautiously optimistic about these latest results.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, and editor-in-chief of the Journal of Clinical Hypertension, said: “This new report from Wuhan, China, gives modest reassurance that the use of ACE inhibitors or ARBs in hypertensive patients with COVID-19 disease does not increase the risk of clinical deterioration or death.
“Ongoing, more definitive studies should help resolve competing hypotheses regarding the effects of these agents: whether the increased ACE2 enzyme levels they produce can worsen outcomes by increasing access of the COVID virus to lung tissue; or whether there is a benefit linked to a protective effect of increased ACE2 on alveolar cell function,” Dr. Weber noted.
“Though the number of patients included in this new report is small, it is startling that hypertensive patients were three times as likely as nonhypertensives to have a fatal outcome, presumably reflecting vulnerability due to the cardiovascular and metabolic comorbidities associated with hypertension,” he added.
“In any case, for now, clinicians should continue treating hypertensive patients with whichever drugs, including ACE inhibitors and ARBs, best provide protection from adverse outcomes,” Dr. Weber concluded.
John McMurray, MD, professor of medical cardiology, University of Glasgow, Scotland, commented: “This study from Wuhan provides some reassurance about one of the two questions about ACEI/ARBs: Do these drugs increase susceptibility to infection? And if [the patient is] infected, do they increase the severity of infection? This study addresses the latter question and appears to suggest no increased severity.”
However, Dr. McMurray pointed out that the study had many limitations. There were only small patient numbers and the data were unadjusted, “although it looks like the ACE inhibitor/ARB treated patients were higher risk to start with.” It was an observational study, and patients were not randomized and were predominantly treated with ARBs, and not ACE inhibitors, so “we don’t know if the concerns apply equally to these two classes of drug.
“Other data published and unpublished supporting this (even showing better outcomes in patients treated with an ACE inhibitor/ARB), and, to date, any concerns about these drugs remain unsubstantiated and the guidance from medical societies to continue treatment with these agents in patients prescribed them seems wise,” Dr. McMurray added.
Franz H. Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “The study from Wuhan is not a great study. They didn’t even do a multivariable analysis. They could have done a bit more with the data, but it still gives some reassurance.”
Dr. Messerli said it was “interesting” that 30% of the patients hospitalized with COVID-19 in the sample had hypertension. “That corresponds to the general population, so does not suggest that having hypertension increases susceptibility to infection – but it does seem to increase the risk of a bad outcome.”
Dr. Messerli noted that there are two more similar studies due to be published soon, both said to suggest either a beneficial or neutral effect of ACE inhibitors/ARBs on COVID-19 outcomes in hospitalized patients.
“This does help with confidence in prescribing these agents and reinforces the recommendations for patients to stay on these drugs,” he said.
“However, none of these studies address the infectivity issue – whether their use upregulates the ACE2 receptor, which the virus uses to gain entry to cells, thereby increasing susceptibility to the infection,” Dr. Messerli cautioned. “But the similar or better outcomes on these drugs are encouraging,” he added.
The Wuhan study was supported by the Health and Family Planning Commission of Wuhan City, China. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Initial data from one Chinese center on the use of angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) in patients hospitalized with COVID-19 appear to give some further reassurance about continued use of these drugs.
The report from one hospital in Wuhan found that among patients with hypertension hospitalized with the COVID-19 virus, there was no difference in disease severity or death rate in patients taking ACE inhibitors or ARBs and those not taking such medications.
The data were published online April 23 in JAMA Cardiology.
The study adds to another recent report in a larger number of COVID-19 patients from nine Chinese hospitals that suggested a beneficial effect of ACE inhibitors or ARBs on mortality.
Additional studies
Two other similar studies have also been recently released. Another study from China, published online March 31 in Emerging Microbes & Infections, included a small sample of 42 hospitalized patients with COVID-19 on antihypertensive therapy. Those on ACE inhibitor/ARB therapy had a lower rate of severe disease and a trend toward a lower level of IL-6 in peripheral blood. In addition, patients on ACE inhibitor/ARB therapy had increased CD3+ and CD8+ T-cell counts in peripheral blood and decreased peak viral load compared with other antihypertensive drugs.
And a preliminary study from the UK, which has not yet been peer reviewed, found that treatment with ACE inhibitors was associated with a reduced risk of rapidly deteriorating severe COVID-19 disease.
The study, available online on MedRxiv, a preprint server for health sciences, reports on 205 acute inpatients with COVID-19 at King’s College Hospital and Princess Royal University Hospital, London.
Of these, 51.2% had hypertension, 30.2% had diabetes, and 14.6% had ischemic heart disease or heart failure. Of the 37 patients on ACE inhibitors, five (14%) died or required critical care support compared with 29% (48/168) of patients not taking an ACE inhibitor.
New Wuhan study
The authors of the new article published in JAMA Cardiology, led by Juyi Li, MD, reported on a case series of 1,178 patients hospitalized with COVID-19 at the Central Hospital of Wuhan, Hubei, China, between Jan. 15 and March 15, 2020.
Patients were a median age of 55 years, and 46% were men. They had an overall in-hospital mortality rate of 11%.
Of the 1,178 patients, 362 (30.7%) had a diagnosis of hypertension. These patients were older (median age, 66 years) and had a greater prevalence of chronic diseases. Patients with hypertension also had more severe manifestations of COVID-19 compared to those without hypertension, including higher rates of acute respiratory distress syndrome and in-hospital mortality (21.3% vs. 6.5%).
Of the 362 patients with hypertension, 31.8% were taking ACE inhibitors or ARBs.
Apart from a greater prevalence of coronary artery disease, patients taking ACE inhibitors or ARBs had similar comorbidities to those not taking these medications, and also similar laboratory profile results including blood counts, inflammatory markers, renal and liver function tests, and cardiac biomarkers, although those taking ACE inhibitors/ARBs had higher levels of alkaline phosphatase.
The most commonly used antihypertensive drugs were calcium blockers. The percentage of patients with hypertension taking any drug or drug combination did not differ between those with severe and nonsevere infections and between those who survived and those who died.
Specifically regarding ACE inhibitors/ARBs, there was no difference between those with severe versus nonsevere illness in the use of ACE inhibitors (9.2% vs. 10.1%; P = .80), ARBs (24.9% vs. 21.2%; P = .40), or the composite of ACE inhibitors or ARBs (32.9% vs. 30.7%; P = .65).
Similarly, there were no differences in nonsurvivors and survivors in the use of ACE inhibitors (9.1% vs. 9.8%; P = .85); ARBs (19.5% vs. 23.9%; P = .42), or the composite of ACE inhibitors or ARBs (27.3% vs. 33.0%; P = .34).
The frequency of severe illness and death also did not differ between those treated with and without ACE inhibitors/ARBs in patients with hypertension and other various chronic conditions including coronary heart disease, cerebrovascular disease, diabetes, neurological disease, and chronic renal disease.
The authors noted that these data confirm previous reports showing that patients with hypertension have more severe illness and higher mortality rates associated with COVID-19 than those without hypertension.
But they added: “Our data provide some reassurance that ACE inhibitors/ARBs are not associated with the progression or outcome of COVID-19 hospitalizations in patients with hypertension.”
They also noted that these results support the recommendations from almost all major cardiovascular societies that patients do not discontinue ACE inhibitors or ARBs because of worries about COVID-19.
However, the authors did point out some limitations of their study, which included a small number of patients with hypertension taking ACE inhibitors or ARBs and the fact that a nonsevere disease course was still severe enough to require hospitalization. In addition, it was not clear whether ACE inhibitor/ARB treatment at baseline was maintained throughout hospitalization for all patients.
This was also an observational comparison and may be biased by differences in patients taking versus not taking ACE inhibitors or ARBs at the time of hospitalization, although the measured baseline characteristics were similar in both groups.
But the authors also highlighted the finding that, in this cohort, patients with hypertension had three times the mortality rate of all other patients hospitalized with COVID-19.
“Hypertension combined with cardiovascular and cerebrovascular disease, diabetes, and chronic kidney disease would predispose patients to an increased risk of severity and mortality of COVID-19. Therefore, patients with these underlying conditions who develop COVID-19 require particularly intensive surveillance and care,” they wrote.
Experts cautiously optimistic
Some cardiovascular experts were cautiously optimistic about these latest results.
Michael A. Weber, MD, professor of medicine at the State University of New York, Brooklyn, and editor-in-chief of the Journal of Clinical Hypertension, said: “This new report from Wuhan, China, gives modest reassurance that the use of ACE inhibitors or ARBs in hypertensive patients with COVID-19 disease does not increase the risk of clinical deterioration or death.
“Ongoing, more definitive studies should help resolve competing hypotheses regarding the effects of these agents: whether the increased ACE2 enzyme levels they produce can worsen outcomes by increasing access of the COVID virus to lung tissue; or whether there is a benefit linked to a protective effect of increased ACE2 on alveolar cell function,” Dr. Weber noted.
“Though the number of patients included in this new report is small, it is startling that hypertensive patients were three times as likely as nonhypertensives to have a fatal outcome, presumably reflecting vulnerability due to the cardiovascular and metabolic comorbidities associated with hypertension,” he added.
“In any case, for now, clinicians should continue treating hypertensive patients with whichever drugs, including ACE inhibitors and ARBs, best provide protection from adverse outcomes,” Dr. Weber concluded.
John McMurray, MD, professor of medical cardiology, University of Glasgow, Scotland, commented: “This study from Wuhan provides some reassurance about one of the two questions about ACEI/ARBs: Do these drugs increase susceptibility to infection? And if [the patient is] infected, do they increase the severity of infection? This study addresses the latter question and appears to suggest no increased severity.”
However, Dr. McMurray pointed out that the study had many limitations. There were only small patient numbers and the data were unadjusted, “although it looks like the ACE inhibitor/ARB treated patients were higher risk to start with.” It was an observational study, and patients were not randomized and were predominantly treated with ARBs, and not ACE inhibitors, so “we don’t know if the concerns apply equally to these two classes of drug.
“Other data published and unpublished supporting this (even showing better outcomes in patients treated with an ACE inhibitor/ARB), and, to date, any concerns about these drugs remain unsubstantiated and the guidance from medical societies to continue treatment with these agents in patients prescribed them seems wise,” Dr. McMurray added.
Franz H. Messerli, MD, professor of medicine at the University of Bern, Switzerland, commented: “The study from Wuhan is not a great study. They didn’t even do a multivariable analysis. They could have done a bit more with the data, but it still gives some reassurance.”
Dr. Messerli said it was “interesting” that 30% of the patients hospitalized with COVID-19 in the sample had hypertension. “That corresponds to the general population, so does not suggest that having hypertension increases susceptibility to infection – but it does seem to increase the risk of a bad outcome.”
Dr. Messerli noted that there are two more similar studies due to be published soon, both said to suggest either a beneficial or neutral effect of ACE inhibitors/ARBs on COVID-19 outcomes in hospitalized patients.
“This does help with confidence in prescribing these agents and reinforces the recommendations for patients to stay on these drugs,” he said.
“However, none of these studies address the infectivity issue – whether their use upregulates the ACE2 receptor, which the virus uses to gain entry to cells, thereby increasing susceptibility to the infection,” Dr. Messerli cautioned. “But the similar or better outcomes on these drugs are encouraging,” he added.
The Wuhan study was supported by the Health and Family Planning Commission of Wuhan City, China. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Hydroxychloroquine ineffective for COVID-19, VA study suggests
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).
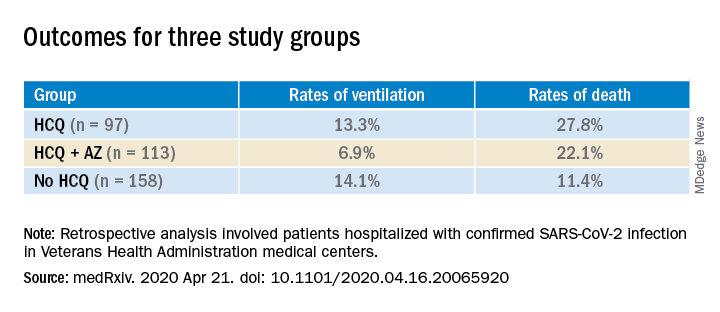
Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).

Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Hydroxychloroquine (HCQ) with or without azithromycin (AZ) is not associated with a lower risk of requiring mechanical ventilation, according to a retrospective study of Veterans Affairs patients hospitalized with COVID-19.
The study, which was posted on a preprint server April 21 and has not been peer reviewed, also showed an increased risk of death associated with COVID-19 patients treated with HCQ alone.
“These findings highlight the importance of awaiting the results of ongoing prospective, randomized controlled studies before widespread adoption of these drugs,” write Joseph Magagnoli with Dorn Research Institute at the Columbia (S.C.) VA Health Care System and the department of clinical pharmacy & outcomes sciences, University of South Carolina, and colleagues.
A spokesperson with the University of Virginia, Charlottesville, where several of coauthors practice, said that the authors declined to comment for this article before peer review is completed.
The new data are not the first to suggest no benefit with HCQ among patients with COVID-19. A randomized trial showed no benefit and more side effects among 75 patients in China treated with HCQ, compared with 75 who received standard of care alone, according to a preprint posted online April 14.
No benefit in ventilation, death rates
The current analysis included data from all 368 male patients hospitalized with confirmed COVID-19 and treated at Veterans Health Administration medical centers in the United States through April 11.
Patients were categorized into three groups: those treated with HCQ in addition to standard of care (n = 97); those treated with HCQ and the antibiotic azithromycin plus standard of care (n = 113); and those who received standard supportive care only (n = 158).

Compared with the no HCQ group, the risk of death from any cause was higher in the HCQ group (adjusted hazard ratio, 2.61; 95% confidence interval, 1.10-6.17; P = .03) but not in the HCQ+AZ group (aHR, 1.14; 95% CI, 0.56-2.32; P = .72).
The risk of ventilation was similar in the HCQ group (aHR, 1.43; 95% CI, 0.53-3.79; P = .48) and in the HCQ+AZ group (aHR, 0.43; 95% CI, 0.16-1.12; P = .09), compared with the no-HCQ group.
This study provides another counterbalance to claims of HCQ efficacy, David R. Wessner, PhD, professor of biology and chair of the department of health and human values at Davidson (N.C.) College, said in an interview.
Interest in HCQ spiked after an open-label, nonrandomized, single-center study of COVID-19 patients in France suggested that hydroxychloroquine helped clear the virus and had a potential enhanced effect when combined with azithromycin.
But the 36-patient trial has since been called into question.
Wait for convincing data
Dr. Wessner, whose research focuses on viral pathogenesis, says that, although the current data don’t definitively answer the question of whether HCQ is effective in treating COVID-19, taking a “let’s try it and see” approach is not reasonable.
“Until we have good, prospective randomized trials, it’s hard to know what to make of this. But this is more evidence that there’s not a good reason to use [HCQ],” Dr. Wessner said. He points out that the small randomized trial from China shows that HCQ comes with potential harms.
Anecdotal evidence is often cited by those who promote HCQ as a potential treatment, but “those are one-off examples,” Wessner continued. “That doesn’t really tell us anything.”
Some HCQ proponents have said that trials finding no benefit are flawed in that the drug is given too late. However, Dr. Wessner says, there’s no way to prove or disprove that claim without randomized controlled trials.
Conflicting messages
Despite lack of clear evidence of benefit for patients with COVID-19, HCQ is recommended off-label by the Chinese National guideline, and the U.S. Food and Drug Administration has issued an emergency-use authorization for the treatment of adult patients with COVID-19.
Conversely, the Infectious Diseases Society of America and a guideline panel convened by the National Institutes of Health each concluded recently that because of insufficient data, they could not recommend any specific treatments for patients with COVID-19.
The VA data for the current study came from the Veterans Affairs Informatics and Computing Infrastructure, which includes inpatient, outpatient and laboratory data and pharmacy claims.
The authors acknowledge some limitations, “including those inherent to all retrospective analyses such as nonrandomization of treatments.”
However, they note that they did adjust for potential confounders, including comorbidities, medications, and clinical and laboratory factors.
A coauthor, Jayakrishna Ambati, MD, is a cofounder of iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and has received consultancy fees from Allergan, Biogen, Boehringer Ingelheim, Immunovant, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences, all unrelated to this work. Dr. Ambati is named as an inventor on a patent application filed by the University of Virginia relating to COVID-19 but unrelated to this work. Another coauthor has received research grants from Boehringer Ingelheim, Gilead Sciences, Portola Pharmaceuticals, and United Therapeutics, all unrelated to this work. The other authors and Dr. Wessner have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Prioritizing ambulatory gynecology care during COVID-19: The latest guidance
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
What exactly constitutes appropriate ambulatory gynecology during this time of social distancing?
On March 30, 2020, the American College of Obstetricians and Gynecologists (ACOG) weighed in, releasing COVID-19 FAQs for Obstetrician-Gynecologists. These recommendations, which include information about obstetric and gynecologic surgery, are available to everyone, including the general public. They are intended to supplement guidance from the Centers for Disease Control and Prevention, as well as previously released ACOG guidance.
The recommendations include examples of patients needing in-person appointments, telehealth visits, or visits that should be deferred.
In-person appointments. Examples of patients for whom in-person appointments are appropriate include those with suspected ectopic pregnancy or profuse vaginal bleeding. With respect to contraceptive services, ACOG suggests that placement of IUDs and implants should continue whenever possible. If placement of the contraceptive device is deferred, use of self-administered hormonal contraceptives (including subcutaneous injections, oral, transdermal patch, and vaginal ring) should be encouraged as a bridge to later initiation of long-acting methods.
Telehealth visits. Video or telephone visits are advised for women desiring counseling and prescribing for contraception or menopausal symptoms.
Deferred. Deferral of office visits until after COVID-19 lockdowns is advised for average-risk women wishing routine well-woman visits. Other situations in which deferral should be considered include the following:
- For patients with abnormal cervical cancer screening results, ACOG suggests that colposcopy with cervical biopsies could be deferred for 6-12 months for patients with low-grade test results. In contrast, for patients with high-grade results, ACOG recommends that evaluation be performed within 3 months.
- For women who wish to discontinue their contraceptive, ACOG advises that removal of IUDs and implants be postponed when possible. These women should be counseled regarding extended use of these devices.
ACOG emphasizes that decisions regarding ambulatory gynecology should be individualized and take into consideration such issues as availability of local and regional resources, staffing, personal protective equipment, and the local prevalence of COVID-19.
As a gynecologist focused on ambulatory care, I believe that many clinicians will welcome this guidance from ACOG, which helps us provide optimal care during these challenging times.
Dr. Kaunitz is professor and associate chairman in the department of obstetrics and gynecology at the University of Florida, Jacksonville. He has disclosed receiving royalties from UpToDate, serving on the safety monitoring board for Femasys, and serving as a consultant for AMAG Pharmaceuticals, Merck & Co, Mithra, and Pfizer. His institution has received funding from pharmaceutical companies and nonprofits.
A version of this article originally appeared on Medscape.com.
Will coronavirus restrictions lead to more advanced cancers?
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
European COVID-19 insights: Try helmet CPAP
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Noninvasive ventilation with helmet continuous positive air pressure (CPAP) deserves to be embraced as an effective strategy in preventing self-induced lung injury, often a key factor in progression from the early milder expression of COVID-19 disease to classic severe acute respiratory distress syndrome, according to European physicians who have been through what they hope are the worst days of the pandemic in the Lombardy region of Northern Italy.
Helmet CPAP is a relatively inexpensive, convenient, well-tolerated intervention. It allows patients to remain conscious and responsive to commands such as “Time to roll over,” which in turn frees up nursing staff. The purpose of helmet CPAP is to curb the huge inspiratory drive that’s a defining feature of this disease and which, unchecked, can lead to self-induced lung injury (SILI), Luciano Gattinoni, MD, explained at a webinar hosted by the European Society of Anaesthesiology.
“Paranoid attention to inspiratory effort – checking it and correcting it – is something where we can make the difference between death and life. It’s extremely important,” said Dr. Gattinoni, guest professor of anesthesiology and intensive care at the University of Gottingen (Germany).
He and his fellow panelists were in accord regarding the merits of helmet CPAP as the premier method of noninvasive ventilatory assistance. They also addressed the importance of monitoring for hypercoagulation, as well as what they’ve come to see as the essential role of pronation in what they define as Type H disease, and the need to have detailed respiratory physiotherapy protocols in place.
“COVID-19 doesn’t like physiotherapy,” explained Paolo Pelosi, MD, professor of anesthesiology and intensive care medicine at the University of Genoa (Italy).
Dr. Gattinoni is credited for identification of two polar phenotypes of what he considers to be a single COVID-19 disease. Early on, many patients present with an atypical form of acute respiratory distress syndrome (ARDS), distinguished by an often-unexpected degree of hypoxia accompanied by high pulmonary compliance and surprisingly little shortness of breath. Dr. Gattinoni and colleagues call this Type L disease, which stands for low elastane, low ventilation to perfusion ratio, low lung weight on CT, and low lung recruitability, which means the patient has a high proportion of aerated lung tissue. Over time, because of either the natural history of the disease or SILI, this may shift to Type H disease, marked by high elastane, high right-to-left shunt, high lung weight, and high recruitability.
“If the pulmonary compliance is above 60 [mL/cm H2O], I’m pretty sure it’s Type L. If it’s 30 [mL/cm H2O] or less, I’m pretty sure it’s Type H. Don’t ask me about 45-55 [mL/cm H2O]; it’s a grey zone,” Dr. Gattinoni said.
Giuseppe Foti, MD, said helmet CPAP in patients with COVID-19 should be free flow, not attached to a ventilator, and the gas flow should be set high – at least 50 L/min – in order to prevent CO2 rebreathing. Although noninvasive ventilation is well accepted for patients with chronic obstructive pulmonary disease or acute cardiogenic pulmonary edema, it hasn’t been extensively studied in the setting of ARDS. A notable exception is a single-center randomized trial in which 83 patients with ARDS at the University of Chicago were assigned to noninvasive ventilation delivered by helmet or face mask (JAMA. 2016 Jun 14;315[22]:2435-41). The endotracheal intubation rate was just 18% in the helmet group, compared with 62% in the face mask group. The 90-day mortality rate was significantly lower in the helmet group as well, noted Dr. Foti, director of the department of anesthesia and intensive care at Monza University Hospital in Milan.
Christian Putensen, MD, said he views intubation for mechanical ventilation as wise in moderate or severe ARDS with an arterial oxygen partial pressure/fraction of inspired oxygen (PaO2/FiO2) ratio below 150. But in milder, Type L COVID-19 disease, he also likes helmet CPAP. It spares the patient from the traumatic compressive stress to the lung induced by mechanical ventilation, which may cause alveolar edema and SILI.
There is, however, a caveat: “Watch carefully and do not delay intubation if you see helmet CPAP is not working; that is, if the blood gas analysis doesn’t improve, the respiratory rate increases, tidal volume increases, and there is still increased respiratory drive,” advised Dr. Putensen, an anesthesiologist at the University of Bonn (Germany).
There is no agreed-upon practical quantitative measure of respiratory drive. A clinical evaluation of the patient’s depth of inspiration is the best guide, he added.
Dr. Gattinoni said that, when helmet CPAP can’t control respiratory drive in a patient with early-stage disease, he feels the only way to interrupt this destructive process is through early intubation and what he termed “gentle mechanical ventilation,” not with a positive end expiratory pressure of 20 cm H2O, but more like 4-5.
Watch for hypercoagulation
Thromboembolic complications are a common feature in COVID-19 disease.
“I’ve had occasion to see the autopsy results in more than 100 patients. It’s devastating to see the number of thromboses and microthromboses in the lungs, the liver, the kidney, and in the brain,” Dr. Gattinoni said.
“COVID-19 is a serial killer, no doubt,” Dr. Pelosi agreed. “He has no mercy for anyone. And he has two bullets: The first one is for the lung, the second is on the vascular side.”
Dr. Putensen is aggressive in utilizing prophylactic high-dose anticoagulation with heparin. He carefully monitors levels of fibrinogen, Factors V and VIII, and d-dimers. In the setting of COVID-19, he has found thromboelastography to be more reliable than partial thromboplastin time in guiding heparin titration.
Pronation
Panelists agreed that pronation is an especially valuable means of enhancing oxygenation in patients with Type H disease. Dr. Putensen tries for more than 16 hours per day. Dr. Foti is preparing a study of the impact of pronation in 50 awake, nonintubated patients, most of whom were on helmet CPAP. Seven of them couldn’t tolerate pronation for even an hour at a time; for the others, the median duration was 3.5 hours at a time.
“We saw a dramatic improvement, a nearly doubling in the PaO2/FiO2 ratio,” Dr. Foti said.
The helmet CPAP study was done outside of the ICU because, in March 2020, the Milan hospital was utterly overwhelmed by COVID-19. The university hospital ordinarily has 25 ICU beds. This was expanded to 100 ICU beds in an effort to meet the emergency, but that still wasn’t sufficient. Indeed, COVID-19 patients occupied 600 of the hospital’s 650 beds. Physicians were forced to do something formerly unthinkable: triage patients for intubation and mechanical ventilation based upon age, comorbidities, and survival prospects.
“We felt schizophrenic. I completely agree with Luciano’s idea to intubate early when we cannot control the respiratory drive that’s due to the disease. But we couldn’t do it because we had too many patients. So we had to triage,” Dr. Foti recalled, breaking off with a sob as other panelists wiped away their own tears during the webcast.
Respiratory physical therapy
Dr. Pelosi said he believes that optimal care of patients with COVID-19 disease requires a major commitment to physical therapy. He strongly recommends having thoughtfully designed separate written protocols in place for respiratory physiotherapy during mechanical ventilation, weaning, and postextubation. COVID-19 patients typically require 7-10 days of assisted ventilation before weaning, and weaning is a protracted process as well.
“I like to say COVID-19 always requires patience. You have to be very, very patient with this disease,” he emphasized. “These patients have a long and difficult weaning. If the patient isn’t improving during weaning, look at two issues: superinfection and thrombembolism, macro and micro.” The physical therapy measures routinely utilized at his hospital during mechanical ventilation include elevation of the bed head greater than 30 degrees, neuromuscular electrical stimulation, subglottic secretion suctioning, tracheal and oral aspiration, and cough assistance. Separate physical therapy menus are used during before and after extubation.
Dr. Gattinoni offered a final word: “We can do almost nothing with this disease. We try our best to keep the patient alive. What we can do is avoid excessive ventilation of the patient. Applying the typical treatment of ARDS in atypical [Type L] ARDS does not make sense and may be extremely harmful.”
Are patients with epilepsy at increased risk of COVID-19 infection?
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
Chronic conditions such as lung disease, diabetes, and heart disease frequently receive attention for increasing the risk of complications for people who contract the coronavirus. Meanwhile, many members of the epilepsy community continue to wonder how the virus affects them. To address these concerns, the Epilepsy Foundation has released information that answers many common questions that people with epilepsy have about how COVID-19 can impact their health.
Perhaps the most pressing of these questions is: Does epilepsy increase the risk or severity of the coronavirus?
“The most common thing we’re hearing from patients in my practice is their proactive concern for being at increased risk for getting the coronavirus,” confirmed Selim Benbadis, MD, division director, epilepsy, EEG, and sleep medicine at the University of South Florida in Tampa. “Epilepsy patients are not at increased risk for complications from the coronavirus because epilepsy does not affect the immune system.”
In other words, people who have epilepsy face the same health challenges as people who do not have the condition and are otherwise healthy. For this reason, people who have epilepsy should exercise the same habits and preventative measures that healthy people would typically take, such as social distancing; avoiding contact with sick people; washing hands regularly; disinfecting surfaces regularly; and avoiding touching hands, eyes, nose and mouth.
However, as Dr. Benbadis explained, the high fever associated with coronavirus can trigger seizures. The increased risk is another reason people who have epilepsy should do their best to avoid getting sick.
Seizure medications do not increase COVID-19 risk but other conditions can
Similarly, epilepsy medications do not increase the risk of contracting the disease.
“The medications patients take to treat their epilepsy do not affect their immune system,” said Andrew Wilner, MD, associate professor of neurology at the University of Tennessee Health Science Center, Memphis. There are a few exceptions – such as adrenocorticotropic hormone and everolimus – but doctors rarely use these drugs to treat epilepsy.
However, there are some situations and conditions that may pose a risk for people who contact the coronavirus. For instance, people who have problems swallowing their food and tend to suck food down their windpipes are more likely to develop pneumonia. Also, much like the general population, having diabetes, heart disease, or lung problems increase the chances of developing complications from the virus.
The best ways to avoid additional risks in epilepsy
Because of the pandemic, people who have epilepsy may have found that many of their doctors’ appointments have been canceled. Many clinics and medical practices have done this in order minimize exposing people who have acute illnesses to the virus. By focusing more on patients with acute conditions, doctors and nurses can better tend to patients with acute problems. As a result, practices have shifted to providing patient care using telemedicine as much as possible.
“Telemedicine services have surged, and I’ve been saying for years that telemedicine was going to grow,” Dr. Benbadis said. “It’s more convenient, and I believe that we’re going to see increased use of telemedicine long after the coronavirus pandemic is over.”
Aside from communicating with their doctors, the Epilepsy Foundation and Dr. Wilner stress that the best way for people who have epilepsy to stay healthy is by taking their medications on a regular basis exactly as prescribed.
“Taking mediation correctly and regularly is the best strategy for epilepsy patients to avoid unnecessary hospitalizations,” Dr. Wilner said. “If they have breakthrough seizures and get sent to the emergency room, then they risk being exposed to the virus in the ER.”
Also, because ERs are more crowded than usual, the Epilepsy Foundation encourages people who suspect they have the coronavirus to call their doctor’s office first. The goal is to try to make sure that people who have severe or life-threatening symptoms have access to treatment in the ER.
As with the general population, the first thing that epilepsy patients who suspect they have the coronavirus should do is call his or her doctor’s office. The health care professional taking the call will ask the patient a series of questions to determine whether the patient has COVID-19 or another condition or needs to seek emergency medical attention.
Fever, cough, and trouble breathing fall among the most commonly reported symptoms of the coronavirus. In many cases, health care providers recommend that people with mild versions of these symptoms stay at home.
Helpful tips
The Epilepsy Foundation offers tips on signs to look for when trying to figure out when a seizure requires an ER visit. These are:
- Seizures in which awareness is lost for more than 5 minutes and no reversal medications are available.
- Seizures with an unusual pattern or duration.
- Seizures that cannot be treated safely at home or are not responding to rescue medication even after the medication has had enough time to work.
- Seizures that occur after a severe blow to the head.
Additionally, while COVID-19 can cause death and sudden death in patients, the virus does not cause sudden unexpected death in epilepsy (SUDEP). Because SUDEP is extremely rare, Dr. Benbadis said that there is no information to suggest that contracting the coronavirus will increase the risk,
Finally, no shortages of seizures medications have been reported as a result of COVID-19. However, there were shortages of generic levetiracetam immediate-release and levetiracetam extended-release medications prior to and during COVID-19. Experts expect the shortage to continue.
Overall, people who have epilepsy should be able to stay healthy – provided they exercise healthy and preventative habits.
“The majority of epilepsy patients should be reassured that if they continue their usual care, take their meds as directed, get adequate sleep, nutritious diet, they’re not at any increased risk compared to the general population,” said Dr. Wilner.
Dr. Benbadis reported the following disclosures: consultant for Bioserenity (DigiTrace), Brain Sentinel, Cavion, Ceribell, Eisai, Greenwich, LivaNova, Neuropace, SK biopharmaceuticals, Sunovion; speakers bureau for Eisai, Greenwich, LivaNova, Sunovion; Florida Medical Director of Stratus/Alliance; Member: Epilepsy Study Consortium; grant support from Cavion, LivaNova, Greenwich, SK biopharmaceuticals, Sunovion, Takeda, UCB, Xenon; royalties as an author or editor for Emedicine-Medscape-WebMD, UpToDate; editorial board for the Epilepsy.com (Epilepsy Foundation) controversy section, Emedicine-Medscape-WebMD, Epileptic Disorders, Epilepsy and Behavior, and Expert Review of Neurotherapeutics. Dr. Wilner reports Medical Advisory Board of Accordant Health Services, Greensboro, S.C., and book royalties: “The Locum Life: A Physician’s Guide to Locum Tenens,” Lulu Press.
COVID-19 antibody tests proliferate, but what do they show?
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.
Noopur Raje, MD, has been sitting at home for 5 weeks waiting for her COVID-19 test to turn negative so she can get back to work. She’s a cancer specialist – head of the Massachusetts General Hospital’s Center for Multiple Myeloma – but Raje says as soon as she’s allowed back to the hospital, she’ll head straight to the front line of COVID-19 caregivers.
“It’s people like us who have to get back in the trenches and do the work now,” she told Medscape Medical News.
“I still will be at risk,” she said. But, having nursed her physician husband through COVID-19 at home until he was admitted to an intensive care unit, she is determined to help in the COVID-19 wards.
“I will be the first one to volunteer to take care of these patients,” she said. “I can’t wait, as I want to give these folks hope. They are so scared.”
Around the world, it’s assumed that she and others like her who’ve recovered from COVID-19 will be immune to the infection.
Some have suggested that with antibodies to the virus coursing through their veins, these survivors might be given immunity passports. They could be the ones to jump-start people’s lives again ― the first to be let out from lockdown, and in healthcare, the ones to head the ongoing battle against this pandemic.
So, there has been a race to develop COVID-19 antibody tests to identify these people.
Circumventing the Usual Clearance Process
To speed up the process, the US Food and Drug Administration (FDA) made a much-criticized move to allow a free-for-all for developers to begin marketing antibody tests that had not gone through the agency’s usual evaluation process. The result was a flood of more than 90 unapproved tests “that have, frankly, dubious quality,” said Scott Becker, CEO of the Association of Public Health Laboratories (APHL), which represents local and state public laboratories.
The APHL spoke out in dismay – its chief program officer, Eric Blank, decried the “Wild West” of tests unleashed on the public.
“These tests create more uncertainty than before,” said Kelly Wroblewski, APHL’s director of infectious diseases, in a news conference on April 14. “Having many inaccurate tests is worse than having no tests at all.”
The APHL and the FDA, working with the Centers for Disease Control and Prevention and the National Institutes of Health (NIH), have moved quickly into damage control, conducting evaluations of the tests in an effort to distinguish the potentially useful from the useless.
So far, they have succeeded in issuing emergency use authorizations (EUAs) to only four tests, those marketed by Cellex, Ortho Clinical Diagnostics, Chembio Diagnostic Systems, and the Mount Sinai Laboratory.
For all the other antibody tests on the market that do not have an EUA, “They’re trusting that the test developer has done a good job in validation,” Becker said. But there are worrying anecdotes. “Our members have reported that they’ve seen fraudulent marketing.... We’ve seen the FDA clamp down on some companies... [and] a number of cities and health departments have issued warnings because of what they’ve seen,” he added.
In particular, Wroblewski said, some companies are marketing tests for use in physicians’ offices or pharmacies. “Today, there are no serology tests approved for point-of-care settings,” she warned. “We don’t know how to interpret the test results, if the presence of antibodies indicates immunity, how long it will last, or what titer might be sufficient.”
Uncertainty Emphasized
The FDA emphasized the uncertainty about antibody tests in a statement released on April 18.
Although the tests can identify people who have been exposed and who developed an immune response to the virus, the agency noted, “we don’t yet know that just because someone has developed antibodies, that they are fully protected from reinfection, or how long any immunity lasts.”
The FDA says that the role of these antibody tests, at present, lies in providing information to “help us track the spread of the virus nationwide and assess the impact of our public health efforts now, while also informing our COVID-19 response as we continue to move forward.”
The World Health Organization (WHO) also emphasized the current uncertainty over antibody tests at a press briefing on April 17. “Nobody is sure about the length of protection that antibodies may give and whether they fully protect against ... the disease,” said Mike Ryan, MD, executive director of the WHO’s emergencies program. There is also a concern that such tests may give false assurance or be misused. “There is still a lot of work that needs to be done to validate these antibody tests,” he added.
“The WHO are right to highlight that any antibody test, if we get one, won’t be able to definitely say whether someone is immune to the infection, because we just don’t know enough yet about how immunity works with COVID-19,” commented Prof. Chris Dye, Oxford Martin School, University of Oxford, in reaction on the UK Science Media Center.
Expanding on this point on the same site, Andrew Easton PhD, professor of virology at the University of Warwick, noted that “a serology test does not discriminate between neutralising and non-neutralising antibodies; a discriminatory test is much more complex and slow.”
Only the neutralizing antibodies have the ability to inactivate the invading virus, he noted.
“When people are infected, the proportions of neutralising and non-neutralising antibodies can differ. It is not always understood what makes an antibody neutralising and another non-neutralising, or why an infection leads to production of more of one of these types of antibodies,” he explained. “The initial immune response immediately following infection sets the memory of the immune system, so if the person had generated mostly non-neutralising antibodies, the next time that person encounters the same virus, they may not be able to prevent an infection.”
So at present, the information from antibody testing is largely unhelpful to individuals, but it could be valuable to epidemiologists and policy makers.
“States are looking at ways they can integrate reliable serologic tests for surveillance,” explained APHL’s Blank.
Knowing how widespread the infection has been within a community could guide research and possibly public health decisions, Wroblewski said at the APHL press conference. But she’s hesitant here, too. “I know there has been a lot of talk about using this testing to ease restrictions, but I do think we need to be cautious on how quickly we move in that direction.” If people don’t have antibodies, it means they haven’t been exposed and that they’re still vulnerable, she noted. “If nothing else, that still informs policy decisions, even if they’re not the policy decisions we want.”
Trials Recruiting, Medical Centers Develop Own Tests
Despite the uncertainties over antibody testing, many efforts are still being guided by this strategy.
The NIH is recruiting volunteers to its antibody testing study and suggests that immunity is “likely” for those who test positive.
In addition, several large medical centers have developed their own antibody tests, including Stanford, the Yale New Haven Hospital, and the Mayo Clinic.
The Stanford test detects two types of antibodies: IgM, which is made early in an immune response and usually wanes quickly, and IgG, which rises more slowly after infection but usually persists longer.
“There’s limited data out of China and Europe showing that this appears to be the response pattern followed with this virus,” commented Thomas Montine, MD, PhD, professor and chair of pathology at Stanford University. “But no one has had this long enough to know how long after infection the antibodies persist,” he added.
“There is enormous demand for serologic testing,” said William Morice, MD, PhD, president of Mayo Clinic Laboratories. “At this time, serology testing needs to be prioritized for efforts to identify individuals in areas where potential immunity is key ― supporting healthcare workers, screening for potential plasma donors, and helping advance the most promising vaccine candidates.”
During a recent webinar with the Association for Value-Based Cancer Care, the largest physician-owned oncology-hematology practice in the country, the president, Lucio Gordan, MD, said his organization was looking into antibody testing for staff. “They wanted to see how many have been exposed,” he said, although “what it means is uncertain.”
When Medscape Medical News checked back with him a few weeks later, Gordan, president of Florida Cancer Specialists and Research Institute, reported that no progress had been made.
“We unfortunately have not been able to test yet, due to concerns with reliability of kits. We are waiting for a better solution so we can reassess our strategy,” he said.
This article first appeared on Medscape.com.







