User login
Delaying RT for higher-risk prostate cancer found safe
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
A study of more than 60,000 prostate cancer patients suggests it is safe to delay radiation therapy (RT) for at least 6 months for localized higher-risk disease being treated with androgen deprivation therapy.
These findings are relevant to oncology care in the COVID-19 era, as the pandemic has complicated delivery of radiation therapy (RT) in several ways, the study authors wrote in JAMA Oncology.
“Daily hospital trips for RT create many possible points of COVID-19 transmission, and patients with cancer are at high risk of COVID-19 mortality,” Edward Christopher Dee, a research fellow at Dana-Farber Cancer Institute in Boston, and colleagues wrote.
To assess the safety of delaying RT, the investigators analyzed National Cancer Database data for 63,858 men with localized but unfavorable intermediate-risk, high-risk, or very-high-risk prostate cancer diagnosed during 2004-2014 and managed with external beam RT and androgen deprivation therapy (ADT).
Only 5.6% of patients (n = 3,572) initiated their RT 0-60 days before starting ADT. Another 36.3% (n = 23,207) initiated RT 1-60 days after starting ADT, 47.4% (n = 30,285) initiated RT 61-120 days after starting ADT, and 10.6% (n = 6,794) initiated RT 121-180 days after starting ADT.
The investigators found that 10-year overall survival rates were similar regardless of when patients started RT.
Multivariate analysis in the unfavorable intermediate-risk group showed that, relative to peers who started RT before ADT, men initiating RT later did not have significantly poorer overall survival, regardless of whether RT was initiated 1-60 days after starting ADT (hazard ratio for death, 1.03; P = .64), 61-120 days after (HR, 0.95; P = .42), or 121-180 days after (HR, 0.99; P = .90).
Findings were similar in the combined high-risk and very-high-risk group, with no significant elevation of mortality risk for patients initiating RT 1-60 days after starting ADT (HR, 1.07; P = .12), 61-120 days after (HR, 1.04; P = .36), or 121-180 days after (HR, 1.07; P = .17).
“These results validate the findings of two prior randomized trials and possibly justify the delay of prostate RT for patients currently receiving ADT until COVID-19 infection rates in the community and hospitals are lower,” the authors wrote.
Despite the fairly short follow-up period and other study limitations, “if COVID-19 outbreaks continue to occur sporadically during the coming months to years, these data could allow future flexibility about the timing of RT initiation,” the authors concluded.
Experts weigh in
“Overall, this study is asking a good question given the COVID situation and the fact that many providers are delaying RT due to COVID concerns of patients and providers,” Colleen A. Lawton, MD, of the Medical College of Wisconsin, Milwaukee, commented in an interview.
At the same time, Dr. Lawton cautioned about oversimplifying the issue, noting that results of the Radiation Therapy Oncology Group (RTOG) 9413 trial suggest important interactions between the anatomic extent of RT and the timing of ADT on outcomes (Int J Radiat Oncol Biol Phys. 2007 Nov 1;69[3]:646-55).
“I have certainly delayed some of my own patients with ADT during the COVID pandemic,” she reported. “No one knows what the maximum acceptable delay should be. A few months is likely not a problem, and a year is probably too much, but scientifically, we just don’t know.”
The interplay of volume irradiated and ADT timing is relevant here, agreed Mack Roach III, MD, of University of California, San Francisco.
In addition, the study did not address why ADT was given when it was, the duration of this therapy, and endpoints other than overall survival (such as prostate-specific antigen failure rate) that may better reflect the effectiveness of cancer treatment.
“Yes, delays are safe for patients on ADT, but not for the reasons stated. A more appropriate source of data is RTOG 9910, which compared 28 versus 8 weeks of ADT prior to RT for mostly intermediate-risk prostate cancer patients with comparable results,” Dr. Roach noted (J Clin Oncol. 2015 Feb 1;33[4]:332-9).
“Delay duration should be based on the risk of disease, but 6 months is probably safe, especially if on ADT,” he said.
Michael J. Zelefsky, MD, of Memorial Sloan Kettering Cancer Center in New York, said he agreed with the investigators’ main conclusions. “Once ADT suppression is achieved, maintaining patients on this regimen for 6 months would not likely lead to the development of a castrate-resistant state where radiotherapy would be less effective,” he elaborated.
However, limitations of the database used preclude conclusions about the safety of longer delays or the impact on other outcomes, he cautioned.
“This study provides further support to the accepted notion that delays of up to 6 months prior to initiation of planned prostate radiation would be safe and appropriate, especially where concerns of COVID outbreaks may present significant logistic challenges and concerns for the patient, who needs to commit to a course of daily radiation treatments, which could span for 5-8 weeks,” Dr. Zelefsky said.
“We have, in fact, adopted this approach in our clinics during the COVID outbreaks in New York,” he reported. “Most of our patients with unfavorable intermediate- or high-risk disease were initiated on ADT planned for at least 4-6 months before the radiotherapy was initiated. In addition, for these reasons, our preference has been to also offer such patients, if feasible, an ultrahypofractionated treatment course where the radiotherapy course is completed in five fractions over 1-2 weeks.”
This research was funded by the National Institutes of Health. The authors disclosed various grants and personal fees outside the submitted work. Dr. Lawton disclosed that she was a coauthor on RTOG 9413. Dr. Roach and Dr. Zelefsky disclosed no relevant conflicts of interest.
SOURCE: Dee EC et al. JAMA Oncol. 2020 Aug 13. doi: 10.1001/jamaoncol.2020.3545.
FROM JAMA ONCOLOGY
Latest report adds almost 44,000 child COVID-19 cases in 1 week
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
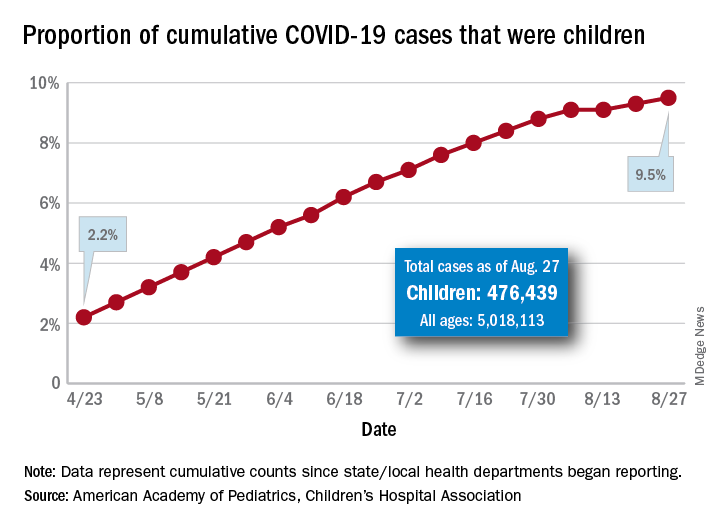
The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The new cases bring the cumulative number of infected children to over 476,000, and that figure represents 9.5% of the over 5 million COVID-19 cases reported among all ages, the AAP and the CHA said in their weekly report. The cumulative number of children covers 49 states (New York is not reporting age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
From lowest to highest, the states occupying opposite ends of the cumulative proportion spectrum are New Jersey at 3.4% – New York City was lower with a 3.2% figure but is not a state – and Wyoming at 18.3%, the report showed.
Children represent more than 15% of all reported COVID-19 cases in five other states: Tennessee (17.1%), North Dakota (16.0%), Alaska (15.9%), New Mexico (15.7%), and Minnesota (15.1%). The states just above New Jersey are Florida (5.8%), Connecticut (5.9%), and Massachusetts (6.7%). Texas has a rate of 5.6% but has reported age for only 8% of confirmed cases, the AAP and CHA noted.
Children make up a much lower share of COVID-19 hospitalizations – 1.7% of the cumulative number for all ages – although that figure has been slowly rising over the course of the pandemic: it was 1.2% on July 9 and 0.9% on May 8. Arizona (4.1%) is the highest of the 22 states reporting age for hospitalizations and Hawaii (0.6%) is the lowest, based on the AAP/CHA data.
Mortality figures for children continue to be even lower. Nationwide, 0.07% of all COVID-19 deaths occurred in children, and 19 of the 43 states reporting age distributions have had no deaths yet. Pediatric deaths totaled 101 as of Aug. 27, the two groups reported.
First randomized trial reassures on ACEIs, ARBs in COVID-19
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-19: In-hospital mortality data miss bigger picture of racial inequality
A recent study that reported no association between race and in-hospital mortality among patients with COVID-19 failed to capture broader health care inequities, according to a leading expert.
During an AGA FORWARD Program webinar, Darrell Gray II, MD, deputy director of the Center for Cancer Health Equity at Ohio State University in Columbus, noted that the study by Baligh R. Yehia, MD, and colleagues had several important limitations: specifically, a lack of data from before or after hospitalization, flawed neighborhood deprivation indices, and poorly characterized comorbidities.
While Dr. Yehia and colleagues described these limitations in their publication, Dr. Gray suggested that future studies evaluating race and health outcomes need to be “deliberate and intentional with collecting data.”
According to Dr. Gray, statistics from the Centers for Disease Control and Prevention and the APM Research Lab paint a more accurate picture of health care inequities. The CDC, for instance, reports that people who are Black are nearly five times as likely to be hospitalized for COVID-19, and approximately twice as likely to die from the disease, compared with those who are White. The APM Research Lab reports an even more striking relative mortality rate for Black Americans – almost four times higher than that of White Americans.
“People of color have been disproportionately impacted by COVID-19, whether it be by cases, hospitalizations, or deaths,” Dr. Gray said. “We have to think about why that is, and what has led to this.”
Dr. Gray emphasized that poorer outcomes among people of color are “not necessarily biological.”
“It’s the environment and social constructs that contribute to why there’s a disproportionate burden of chronic disease and why there’s a disproportionate burden of COVID-19,” he said.
According to Dr. Gray, disparate health care outcomes can be traced back to social determinants of health, which he and his colleagues highlighted in a June comment published in Nature Reviews Gastroenterology & Hepatology.
“Although much attention has focused on the high burden of chronic disease among [people of color], which predisposes them to poor outcomes if they acquire COVID-19, there is less recognition of the nonmedical health-related social needs and social determinants of health that represent the root causes of such health disparities,” they wrote.
Social determinants of health include an array of population factors, including economic stability, social and community context, neighborhood and environment, education, and access to health care.
For each, Dr. Gray encouraged comprehensive and nuanced assessment.
“Is there access to health care?” Dr. Gray asked. “Not just access in the sense of having insurance – certainly that’s a benefit – but if someone has insurance, can they get to where the health center is? Or is that something they might have to catch three buses and a cab to get to?”
Dr. Gray said that such obstacles are not outside the scope of the medical community.
“This is not beyond our responsibility ... to address social determinants of health,” Dr. Gray said.
When asked by a webinar attendee how the medical community can tackle racism, Dr. Gray offered several practical steps to move forward.
First, he suggested that clinicians and researchers listen to affected patient populations.
“Many of us, including clinicians, have been privileged to have their blinders on, if you will, to issues of racism that have been affecting our patients for a long time,” he said.
Second, Dr. Gray encouraged those who have learned to teach others.
“You need to start teaching your peers, your colleagues, your family, and friends about how racism affects patient outcomes.”
Third, he recommended that clinicians incorporate these lessons into routine practice, whether in a private or an academic setting.
“Are there ways in which you can refer patients to address social determinants of health? Are you capturing that information in your check-in materials?” Dr. Gray asked. “If you’re an investigator, when you’re doing research – whether it’s health disparities research or other – are you looking at your research through a health equity lens? Are you asking questions about social determinants of health?”
Finally, Dr. Gray called for stronger community engagement during design and conduction of clinical trials.
“People don’t care how much you know until they know how much you care,” he said. “And they won’t know how much you care unless you’re visible, and unless you’re there, and these are sustainable relationships.”
The FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health.
A recent study that reported no association between race and in-hospital mortality among patients with COVID-19 failed to capture broader health care inequities, according to a leading expert.
During an AGA FORWARD Program webinar, Darrell Gray II, MD, deputy director of the Center for Cancer Health Equity at Ohio State University in Columbus, noted that the study by Baligh R. Yehia, MD, and colleagues had several important limitations: specifically, a lack of data from before or after hospitalization, flawed neighborhood deprivation indices, and poorly characterized comorbidities.
While Dr. Yehia and colleagues described these limitations in their publication, Dr. Gray suggested that future studies evaluating race and health outcomes need to be “deliberate and intentional with collecting data.”
According to Dr. Gray, statistics from the Centers for Disease Control and Prevention and the APM Research Lab paint a more accurate picture of health care inequities. The CDC, for instance, reports that people who are Black are nearly five times as likely to be hospitalized for COVID-19, and approximately twice as likely to die from the disease, compared with those who are White. The APM Research Lab reports an even more striking relative mortality rate for Black Americans – almost four times higher than that of White Americans.
“People of color have been disproportionately impacted by COVID-19, whether it be by cases, hospitalizations, or deaths,” Dr. Gray said. “We have to think about why that is, and what has led to this.”
Dr. Gray emphasized that poorer outcomes among people of color are “not necessarily biological.”
“It’s the environment and social constructs that contribute to why there’s a disproportionate burden of chronic disease and why there’s a disproportionate burden of COVID-19,” he said.
According to Dr. Gray, disparate health care outcomes can be traced back to social determinants of health, which he and his colleagues highlighted in a June comment published in Nature Reviews Gastroenterology & Hepatology.
“Although much attention has focused on the high burden of chronic disease among [people of color], which predisposes them to poor outcomes if they acquire COVID-19, there is less recognition of the nonmedical health-related social needs and social determinants of health that represent the root causes of such health disparities,” they wrote.
Social determinants of health include an array of population factors, including economic stability, social and community context, neighborhood and environment, education, and access to health care.
For each, Dr. Gray encouraged comprehensive and nuanced assessment.
“Is there access to health care?” Dr. Gray asked. “Not just access in the sense of having insurance – certainly that’s a benefit – but if someone has insurance, can they get to where the health center is? Or is that something they might have to catch three buses and a cab to get to?”
Dr. Gray said that such obstacles are not outside the scope of the medical community.
“This is not beyond our responsibility ... to address social determinants of health,” Dr. Gray said.
When asked by a webinar attendee how the medical community can tackle racism, Dr. Gray offered several practical steps to move forward.
First, he suggested that clinicians and researchers listen to affected patient populations.
“Many of us, including clinicians, have been privileged to have their blinders on, if you will, to issues of racism that have been affecting our patients for a long time,” he said.
Second, Dr. Gray encouraged those who have learned to teach others.
“You need to start teaching your peers, your colleagues, your family, and friends about how racism affects patient outcomes.”
Third, he recommended that clinicians incorporate these lessons into routine practice, whether in a private or an academic setting.
“Are there ways in which you can refer patients to address social determinants of health? Are you capturing that information in your check-in materials?” Dr. Gray asked. “If you’re an investigator, when you’re doing research – whether it’s health disparities research or other – are you looking at your research through a health equity lens? Are you asking questions about social determinants of health?”
Finally, Dr. Gray called for stronger community engagement during design and conduction of clinical trials.
“People don’t care how much you know until they know how much you care,” he said. “And they won’t know how much you care unless you’re visible, and unless you’re there, and these are sustainable relationships.”
The FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health.
A recent study that reported no association between race and in-hospital mortality among patients with COVID-19 failed to capture broader health care inequities, according to a leading expert.
During an AGA FORWARD Program webinar, Darrell Gray II, MD, deputy director of the Center for Cancer Health Equity at Ohio State University in Columbus, noted that the study by Baligh R. Yehia, MD, and colleagues had several important limitations: specifically, a lack of data from before or after hospitalization, flawed neighborhood deprivation indices, and poorly characterized comorbidities.
While Dr. Yehia and colleagues described these limitations in their publication, Dr. Gray suggested that future studies evaluating race and health outcomes need to be “deliberate and intentional with collecting data.”
According to Dr. Gray, statistics from the Centers for Disease Control and Prevention and the APM Research Lab paint a more accurate picture of health care inequities. The CDC, for instance, reports that people who are Black are nearly five times as likely to be hospitalized for COVID-19, and approximately twice as likely to die from the disease, compared with those who are White. The APM Research Lab reports an even more striking relative mortality rate for Black Americans – almost four times higher than that of White Americans.
“People of color have been disproportionately impacted by COVID-19, whether it be by cases, hospitalizations, or deaths,” Dr. Gray said. “We have to think about why that is, and what has led to this.”
Dr. Gray emphasized that poorer outcomes among people of color are “not necessarily biological.”
“It’s the environment and social constructs that contribute to why there’s a disproportionate burden of chronic disease and why there’s a disproportionate burden of COVID-19,” he said.
According to Dr. Gray, disparate health care outcomes can be traced back to social determinants of health, which he and his colleagues highlighted in a June comment published in Nature Reviews Gastroenterology & Hepatology.
“Although much attention has focused on the high burden of chronic disease among [people of color], which predisposes them to poor outcomes if they acquire COVID-19, there is less recognition of the nonmedical health-related social needs and social determinants of health that represent the root causes of such health disparities,” they wrote.
Social determinants of health include an array of population factors, including economic stability, social and community context, neighborhood and environment, education, and access to health care.
For each, Dr. Gray encouraged comprehensive and nuanced assessment.
“Is there access to health care?” Dr. Gray asked. “Not just access in the sense of having insurance – certainly that’s a benefit – but if someone has insurance, can they get to where the health center is? Or is that something they might have to catch three buses and a cab to get to?”
Dr. Gray said that such obstacles are not outside the scope of the medical community.
“This is not beyond our responsibility ... to address social determinants of health,” Dr. Gray said.
When asked by a webinar attendee how the medical community can tackle racism, Dr. Gray offered several practical steps to move forward.
First, he suggested that clinicians and researchers listen to affected patient populations.
“Many of us, including clinicians, have been privileged to have their blinders on, if you will, to issues of racism that have been affecting our patients for a long time,” he said.
Second, Dr. Gray encouraged those who have learned to teach others.
“You need to start teaching your peers, your colleagues, your family, and friends about how racism affects patient outcomes.”
Third, he recommended that clinicians incorporate these lessons into routine practice, whether in a private or an academic setting.
“Are there ways in which you can refer patients to address social determinants of health? Are you capturing that information in your check-in materials?” Dr. Gray asked. “If you’re an investigator, when you’re doing research – whether it’s health disparities research or other – are you looking at your research through a health equity lens? Are you asking questions about social determinants of health?”
Finally, Dr. Gray called for stronger community engagement during design and conduction of clinical trials.
“People don’t care how much you know until they know how much you care,” he said. “And they won’t know how much you care unless you’re visible, and unless you’re there, and these are sustainable relationships.”
The FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health.
FROM THE AGA FORWARD PROGRAM
Pandemic worsens disparities in GI and liver disease
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
FROM THE AGA FORWARD PROGRAM
Obesity boosts risks in COVID-19 from diagnosis to death
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
FROM OBESITY REVIEWS
COVID-19 and patients with serious mental illness
“This whole thing is not about heroism. It’s about decency. It may seem a ridiculous idea, but the only way to fight the plague is with decency . ”
– Albert Camus, La Peste (1947)1
Severe acute respiratory syndrome (SARS), H1N1 swine flu, Ebola, Zika, and Middle East respiratory syndrome (MERS): the 21st century has already been witness to several serious infectious outbreaks and pandemics,2 but none has been as deadly and consequential as the current one. The ongoing SARS-coronavirus-2 (SARS-CoV-2) pandemic is shaping not only current psychiatric care but the future of psychiatry. Now that we are beyond the initial stages of the coronavirus disease 2019 (COVID-19) pandemic, when psychiatrists had a crash course in disaster psychiatry, our attention must shift to rebuilding and managing disillusionment and other psychological fallout of the intense early days.3
In this article, we offer guidance to psychiatrists caring for patients with serious mental illness (SMI) during the SARS-CoV-2 pandemic. Patients with SMI are easily forgotten as other issues (eg, preserving ICU capacity) overshadow the already historically neglected needs of this impoverished group.4 From both human and public-health perspectives, this inattention is a mistake. Assuring psychiatric stability is critically important to prevent the spread of COVID-19 in marginalized communities comprised of individuals who are poor, members of racial minorities, and others who already experience health disparities.5 Without controlling transmission in these groups, the pandemic will not be sufficiently contained.
We begin by highlighting general principles of pandemic management because caring for patients with SMI does not occur in a vacuum. Infectious outbreaks require not only helping those who need direct medical care because they are infected, but also managing populations that are at risk of getting infected, including health care and other essential workers.
Principles of pandemic management
Delivery of medical care during a pandemic differs from routine care. An effective disaster response requires collaboration and coordination among public-health, treatment, and emergency systems. Many institutions shift to an incident management system and crisis leadership, with clear lines of authority to coordinate responders and build medical surge capacity. Such a top-down leadership approach must plan and allow for the emergence of other credible leaders and for the restoration of people’s agency.
Unfortunately, adaptive capacity may be limited, especially in the public sector and psychiatric care system, where resources are already poor. Particularly early in a pandemic, services considered non-essential—which includes most psychiatric outpatient care—can become unavailable. A major effort is needed to prevent the psychiatric care system from contracting further, as happened during 9/11.6 Additionally, “essential” cannot be conflated with “emergent,” as can easily occur in extreme circumstances. Early and sustained efforts are required to ensure that patients with SMI who may be teetering on the edge of emergency status do not slip off that edge, especially when the emergency medical system is operating over capacity.
A comprehensive outbreak response must consider that a pandemic is not only a medical crisis but a mental health crisis and a communication emergency.7 Mental health clinicians need to provide accurate information and help patients cope with their fears.
Continue to: Psychological aspects of pandemics
Psychological aspects of pandemics. Previous infectious outbreaks have reaffirmed that mental health plays an outsized role during epidemics. Chaos, uncertainty, fear of death, and loss of income and housing cause prolonged stress and exact a psychological toll.
Adverse psychological impacts include expectable, normal reactions such as stress-induced anxiety or insomnia. In addition, new-onset psychiatric illnesses or exacerbations of existing ones may emerge.8 As disillusionment and demoralization appear in the wake of the acute phase, with persistently high unemployment, suicide prevention becomes an important goal.9
Pandemics lead to expectable behavioral responses (eg, increases in substance use and interpersonal conflict). Fear-based decisions may result in unhelpful behavior, such as hoarding medications (which may result in shortages) or dangerous, unsupervised use of unproven medications (eg, hydroxychloroquine). Trust is needed to accept public-health measures, and recommendations (eg, wearing masks) must be culturally informed to be credible and effective.
Because people are affected differently, at individual, cultural, and socioeconomic levels, they will view the situation differently. For many people, secondary stressors (eg, job loss) may be more disastrous than the primary medical event (ie, the pandemic). This distinction is critical because concrete financial help, not psychiatric care, is needed. Sometimes, even when a psychiatric disorder such as SMI or major neurocognitive disorder is present, the illusion of an acute decompensation can be created by the loss of social and structural supports that previously scaffolded a person’s life.
Mental illness prevention. Community mental-health surveillance is important to monitor for distress, psychiatric symptoms, health-risk behaviors, risk and safety perception, and preparedness. Clinicians must be ready to normalize expectable and temporary distress, while recognizing when that distress becomes pathological. This may be difficult in patients with SMI who often already have reduced stress tolerance or problem-based coping skills.10
Continue to: Psychological first aid...
Psychological first aid (PFA) is a standard intervention recommended by the World Health Organization for most individuals following a disaster; it is evidence-informed and has face validity.11 Intended to relieve distress by creating an environment that is safe, calm, and connected, PFA fosters self-efficacy and hope. While PFA is a form of universal prevention, it is not designed for patients with SMI, is not a psychiatric intervention, and is not provided by clinicians. Its principles, however, can easily be applied to patients with SMI to prevent distressing symptoms from becoming a relapse.
Communication. Good risk and crisis communication are critical because individual and population behavior will be governed by the perception of risk and fear, and not by facts. Failure to manage the “infodemic”7—with its misinformation, contradictory messages, and rumors—jeopardizes infection control if patients become paralyzed by uncertainty and fear. Scapegoating occurs easily during times of threat, and society must contain the parallel epidemic of xenophobia based on stigma and misinformation.12
Decision-making under uncertainty is not perfect and subject to revision as better information becomes available. Pointing this out to the public is delicate but essential to curtail skepticism and mistrust when policies are adjusted in response to new circumstances and knowledge.
Mistrust of an authority’s legitimacy and fear-based decisions lead to lack of cooperation with public-health measures, which can undermine an effective response to the pandemic. Travel restrictions or quarantine measures will not be followed if individuals question their importance. Like the general public, patients need education and clear communication to address their fear of contagion, dangers posed to family (and pets), and mistrust of authority and government. A lack of appreciation of the seriousness of the pandemic and individual responsibility may need to be addressed. Two important measures to accomplish this are steering patients to reputable sources of information and advising that they limit media exposure.
Resilience-building. Community and workplace resilience are important aspects of making it through a disaster as best as possible. Resilience is not innate and fixed; it must be deliberately built.13 Choosing an attitude of post-traumatic growth over the victim narrative is a helpful stance. Practicing self-care (rest, nutrition, exercise) and self-compassion (self-kindness, common humanity, mindfulness) is good advice for patients and caregivers alike.
Continue to: Workforce protection
Workforce protection. Compared to other disasters, infectious outbreaks disproportionally affect the medical community, and care delivery is at stake. While psychological and psychiatric needs may increase during a pandemic, services often contract, day programs and clinics close, teams are reduced to skeleton crews, and only emergency psychiatric care is available. Workforce protection is critical to avoid illness or simple absenteeism due to mistrust of protective measures.
Only a well-briefed, well-led, well-supported, and adequately resourced workforce is going to be effective in managing this public-health emergency. Burnout and moral injury are feared long-term consequences for health care workers that need to be proactively addressed.14 As opposed to other forms of disasters, managing your own fears about safety is important. Clinicians and their patients sit in the proverbial same boat.
Ethics. The anticipated need to ration life-saving care (eg, ventilators) has been at the forefront of ethical concerns.15 In psychiatry, the question of involuntary public-health interventions for uncooperative psychiatric patients sits uncomfortably between public-health ethics and human rights, and is an opportunity for collaboration with public-health and infectious-disease colleagues.
Redeployed clinicians and those working under substandard conditions may be concerned about civil liability due to a modified standard of care during a crisis. Some clinicians may ask if their duty to care must override their natural instinct to protect themselves. There is a lot of room for resentment in these circumstances. Redeployed or otherwise “conscripted” clinicians may resent administrators, especially those administering from the safety of their homes. Those “left behind” to work in potentially precarious circumstances may resent their absent colleagues. Moreover, these front-line clinicians may have been forced to make ethical decisions for which they were not prepared.16 Maintaining morale is far from trivial, not just during the pandemic, but afterward, when (and if) the entire workforce is reunited. All parties need to be mindful of how their actions and decisions impact and are perceived by others, both in the hospital and at home.
Managing patients with SMI during COVID-19
Patients with SMI are potentially hard hit by COVID-19 due to a “tragic” epidemiologic triad of agent-host-environment: SARS-CoV-2 is a highly infectious agent affecting patients with SMI who are vulnerable hosts in permissive environments (Figure).
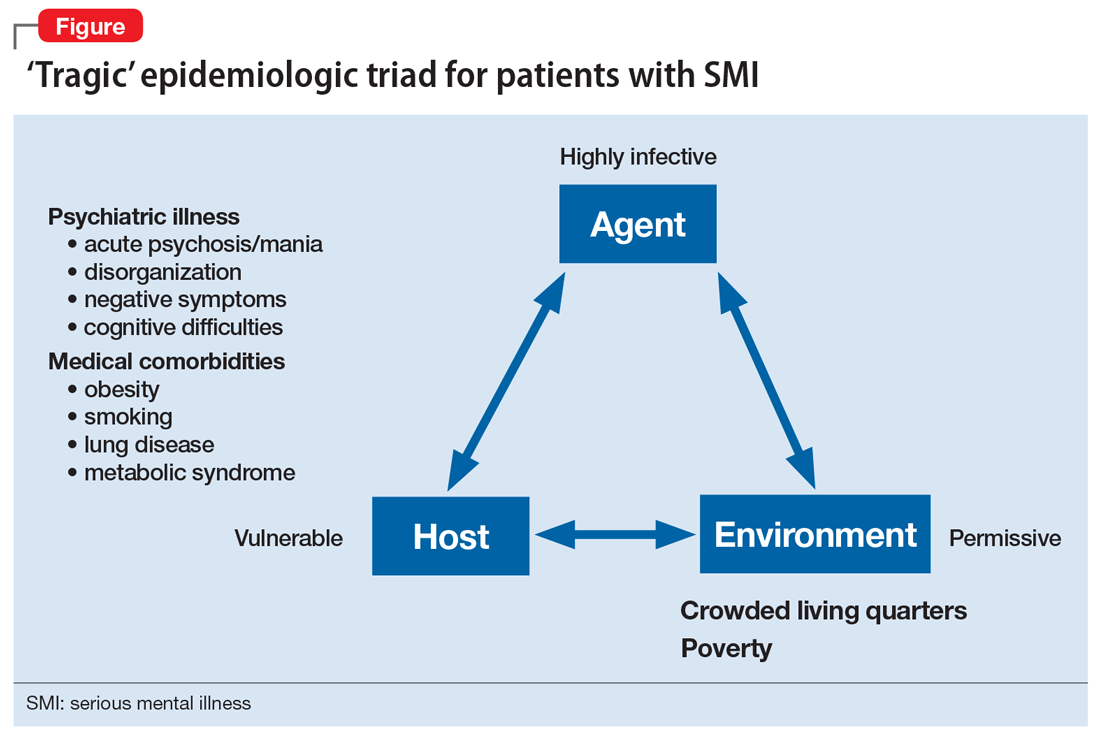
Continue to: While not as infectious as measles...
While not as infectious as measles, COVID-19 is more infectious than the seasonal flu virus.17 It can lead to uncontrolled infection within a short period of time, particularly in enclosed settings. Outbreaks have occurred readily on cruise ships and aircraft carriers as well as in nursing homes, homeless shelters, prisons, and group homes.
Patients with SMI are vulnerable hosts because they have many of the medical risk factors18 that portend a poor prognosis if they become infected, including pre-existing lung conditions and heart disease19 as well as diabetes and obesity.20 Obesity likely creates a hyperinflammatory state and a decrease in vital capacity. Patient-related behavioral factors include poor early-symptom reporting and ineffective infection control.
Unfavorable social determinants of health include not only poverty but crowded housing that is a perfect incubator for COVID-19.
Priority treatment goals. The overarching goal during a pandemic is to keep patients with SMI in psychiatric treatment and prevent them from disengaging from care in the service of infection control. Urgent tasks include infection control, relapse prevention, and preventing treatment disengagement and loneliness.
Infection control. As trusted sources of information, psychiatrists can play an important role in infection control in several important ways:
- educating patients about infection-control measures and public-health recommendations
- helping patients understand what testing can accomplish and when to pursue it
- encouraging protective health behaviors (eg, hand washing, mask wearing, physical distancing)
- assessing patients’ risk appreciation
- assessing for and addressing obstacles to implementing and complying with infection-control measures
- explaining contact tracing
- providing reassurance.
Continue to: Materials and explanations...
Materials and explanations must be adapted for patient understanding.
Patients with disorganization or cognitive disturbances may have difficulties cooperating or problem-solving. Patients with negative symptoms may be inappropriately unconcerned and also inaccurately report symptoms that suggest COVID-19. Acute psychosis or mania can prevent patients from complying with public-health efforts. Some measures may be difficult to implement if the means are simply not there (eg, physical distancing in a crowded apartment). Previously open settings (eg, group homes) have had to develop new mechanisms under the primacy of infection control. Inpatient units—traditionally places where community, shared healing, and group therapy are prized—have had to decrease maximum occupancy, limit the number of patients attending groups, and discourage or outrightly prohibit social interaction (eg, dining together).
Relapse prevention. Patients who take maintenance medications need to be supported. A manic or psychotic relapse during a pandemic puts patients at risk of acquiring and spreading COVID-19. “Treatment as prevention” is a slogan from human immunodeficiency virus (HIV) care that captures the importance of antiretroviral treatment to prevent medical complications from HIV, and also to reduce infecting other people. By analogy, psychiatric treatment for patients with SMI can prevent psychiatric instability and thereby control viral transmission. Avoiding sending psychiatric patients to a potentially stressed acute-care system is important.
Psychosocial support. Clinics need to ensure that patients continue to engage in care beyond medication-taking to proactively prevent psychiatric exacerbations. Healthful, resilience-building behaviors should be encouraged while monitoring and counseling against maladaptive ones (eg, increased substance use). Supporting patients emotionally and helping them solve problems are critical, particularly for those who are subjected to quarantine or isolation. Obviously, in these latter situations, outreach will be necessary and may require creative delivery systems and dedicated clinicians for patients who lack access to the technology necessary for virtual visits. Havens and Ghaemi21 have suggested that a good therapeutic alliance can be viewed as a mood stabilizer. Helping patients grieve losses (loved ones, jobs, sense of safety) may be an important part of support.
Even before COVID-19, loneliness was a major factor for patients with schizophrenia.22 A psychiatric clinic is one aspect of a person with SMI’s social network; during the initial phase of the pandemic, many clinics and treatment programs closed. Patients for whom clinics structure and anchor their activities are at high risk of disconnecting from treatment, staying at home, and becoming lonely.
Continue to: Caregivers are always important...
Caregivers are always important to SMI patients, but they may assume an even bigger role during this pandemic. Some patients may have moved in with a relative, after years of living on their own. In other cases, stable caregiver relationships may be disrupted due to COVID-19–related sickness in the caregiver; if not addressed, this can result in a patient’s clinical decompensation. Clinicians should take the opportunity to understand who a patient’s caregivers are (group home staff, families) and rekindle clinical contact with them. Relationships with caregivers that may have been on “autopilot” during normal times are opportunities for welcome support and guidance, to the benefit of both patients and caregivers.
Table 1 summarizes clinical tasks that need to be kept in mind when conducting clinic visits during COVID-19 in order to achieve the high-priority treatment goals of infection control, relapse prevention, and psychosocial support.
Differential diagnosis. Neuropsychiatric syndromes have long been observed in influenza pandemics,23 due both to direct viral effects and to the effects of critical illness on the brain. Two core symptoms of COVID-19—anosmia and ageusia—suggest that COVID-19 can directly affect the brain. While neurologic manifestations are common,24 it remains unclear to what extent COVID-19 can directly “cause” psychiatric symptoms, or if such symptoms are the result of cytokines25 or other medical processes (eg, thromboembolism).26 Psychosis due to COVID-19 may, in some cases, represent a stress-related brief psychotic disorder.27
Hospitalized patients who have recovered from COVID-19 may have experienced prolonged sedation and severe delirium in an ICU.28 Complications such as posttraumatic stress disorder,29 hypoperfusion-related brain injuries, or other long-term cognitive difficulties may result. In previous flu epidemics, patients developed serious neurologic complications such as post-encephalitic Parkinson’s disease.30
Any person subjected to isolation or quarantine is at risk for psychiatric complications.31 Patients with SMI who live in group homes may be particularly susceptible to new rules, including no-visitor policies.
Continue to: Outpatients whose primary disorder...
Outpatients whose primary disorder is well controlled may, like anyone else, struggle with the effects of the pandemic. It is necessary to carefully differentiate non-specific symptoms associated with stress from the emergence of a new disorder resulting from stress.32 For some patients, grief or adjustment disorders should be considered. Prolonged stress and uncertainty may eventually lead to an exacerbation of a primary disorder, particularly if the situation (eg, financial loss) does not improve or worsens. Demoralization and suicidal thinking need to be monitored. Relapse or increased use of alcohol or other substances as a response to stress may also complicate the clinical picture.33 Last, smoking cessation as a major treatment goal in general should be re-emphasized and not ignored during the ongoing pandemic.34
Table 2 summarizes psychiatric symptoms that need to be considered when managing a patient with SMI during this pandemic.
Treatment tools
Psychopharmacology. Even though crisis-mode prescribing may be necessary, the safe use of psychotropics remains the goal of psychiatric prescribing. Access to medications becomes a larger consideration; for many patients, a 90-day supply may be indicated. Review of polypharmacy, including for pneumonia risk, should be undertaken. Preventing drooling (eg, from sedation, clozapine, extrapyramidal symptoms [EPS]) will decrease aspiration risk.
In general, treatment of psychiatric symptoms in a patient with COVID-19 follows usual guidelines. The best treatment for COVID-19 patients with delirium, however, remains to be established, particularly how to manage severe agitation.28 Pharmacodynamic and pharmacokinetic drug–drug interactions between psychotropics and antiviral treatments for COVID-19 (eg, QTc prolongation) can be expected and need to be reviewed.35 For stress-related anxiety, judicious pharmacotherapy can be helpful. Diazepam given at the earliest signs of a psychotic relapse may stave off a relapse for patients with schizophrenia.36 Even if permitted under relaxed prescribing rules during a public-health emergency, prescribing controlled substances without seeing patients in person requires additional thought. In some cases, adjusting the primary medication to buffer against stress may be preferred (eg, adjusting an antipsychotic in a patient on maintenance treatment for schizophrenia, particularly if a low-dose strategy is pursued).
Clozapine requires registry-based prescribing and bloodwork (“no blood, no drug”). The use of clozapine during this public-health emergency has been made easier because of FDA guidance that allows clozapine to be dispensed without blood work if obtaining blood work is not possible (eg, a patient is quarantined) or can be accomplished only at substantial risk to patients and the population at large. Under certain conditions, clozapine can be dispensed safely and in a way that is consistent with infection prevention. Clozapine-treated patients admitted with COVID-19 should be monitored for clozapine toxicity and the clozapine dose adjusted.37 A consensus statement consistent with the FDA and clinical considerations for using clozapine during COVID-19 is summarized in Table 3.38
Continue to: Long-acting injectable antipsychotics...
Long-acting injectable antipsychotics (LAIs) pose a problem because they require in-person visits. Ideally, during a pandemic, patients should be seen in person as frequently as medically necessary but as infrequently as possible to limit exposure of both patients and staff. Table 4 provides some clinical recommendations on how to use LAIs during the pandemic.39
Supportive psychotherapy may be the most important tool we have in helping patients with loss and uncertainty during these challenging months.40 Simply staying in contact with patients plays a major role in preventing care discontinuity. Even routine interactions have become stressful, with everyone wearing a mask that partially obscures the face. People with impaired hearing may find it even more difficult to understand you.
Education, problem-solving, and a directive, encouraging style are major tools of supportive psychotherapy to reduce symptoms and increase adaptive skills. Clarify that social distancing refers to physical, not emotional, distancing. The judicious and temporary use of anxiolytics is appropriate to reduce anxiety. Concrete help and problem-solving (eg, filling out forms) are examples of proactive crisis intervention.
Telepsychiatry emerged in the pandemic’s early days as the default mode of practice in order to limit in-person contacts.41 Like all new technology, telepsychiatry brings progress and peril.42 While it has gone surprisingly well for most, the “digital divide” does not afford all patients access to the needed technology. The long-term effectiveness and acceptance of telehealth remain to be seen. (Editor’s Note: For more about this topic, see “Telepsychiatry: What you need to know.”
Lessons learned and outlook
Infectious outbreaks have historically inflicted long-term disruptions on societies and altered the course of history. However, each disaster is unique, and lessons from previous disasters may only partially apply.43 We do not yet know how this one will end, including how long it will take for the world’s economies to recover. If nothing else, the current public-health emergency has brought to the forefront what psychiatrists have always known: health disparities are partially responsible for different disease risks (in this case, the risk of getting infected with SARS-CoV-2).5 It may not be a coincidence that the Black Lives Matter movement is becoming a major impetus for social change at a time when the pandemic is exposing health-care inequalities.
Continue to: Some areas of the country...
Some areas of the country succeeded in reducing infections and limiting community spread, which ushered in an uneasy sense of normalcy even while the pandemic continues. At least for now, these locales can focus on rebuilding and preparing for expectable fluctuations in disease activity, including the arrival of the annual flu season on top of COVID-19.44 Recovery is not a return to the status quo ante but building stronger communities—“building back better.”45 Unless there is a continuum of care, shortcomings in one sector will have ripple effects through the entire system, particularly for psychiatric care for patients with SMI, which was inadequate before the pandemic.
Ensuring access to critical care was a priority during the pandemic’s early phase but came at the price of deferring other types of care, such as routine primary care; the coming months will see the downstream consequences of this approach,46 including for patients with SMI.
In the meantime, doing our job as clinicians, as Camus’s fictitious Dr. Bernard Rieux from the epigraph responds when asked how to define decency, may be the best we can do in these times. This includes contributing to and molding our field’s future and fostering a sense of agency in our patients and in ourselves. Major goals will be to preserve lessons learned, maintain flexibility, and avoid a return to unhelpful overregulation and payment models that do not reflect the flexible, person-centered care so important for patients with SMI.47
Bottom Line
During a pandemic, patients with serious mental illness may be easily forgotten as other issues overshadow the needs of this impoverished group. During a pandemic, the priority treatment goals for these patients are infection control, relapse prevention, and preventing treatment disengagement and loneliness. A pandemic requires changes in how patients with serious mental illness will receive psychopharmacology and psychotherapy.
Related Resources
- Huremović D (ed). Psychiatry of pandemics: a mental health response to infection outbreak. Cham, Switzerland: Springer Nature Switzerland AG; 2019.
- Ursano RJ, Fullerton CS, Weisaeth L, et al (eds). Textbook of disaster psychiatry. 2nd ed. Cambridge, UK: Cambridge University Press; 2017.
- Centers for Disease Control and Prevention. Coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/index.html.
- American Psychiatric Association. Coronavirus resources. https://www.psychiatry.org/psychiatrists/covid-19-coronavirus.
- SMI Adviser. Make informed decisions related to COVID-19 and mental health. https://smiadviser.org/about/covid.
Drug Brand Names
Clozapine • Clozaril
Diazepam • Valium
Hydroxychloroquine • Plaquenil
1. Camus A. La peste. Paris, France: Éditions Gallimard; 1947.
2. Huremovic
3. Substance Abuse and Mental Health Services Administration. Phases of disaster. https://www.samhsa.gov/dtac/recovering-disasters/phases-disaster. Updated June 17, 2020. Accessed August 7, 2020.
4. Geller J. COVID-19 and advocacy—the good and the unacceptable. Psychiatric News. https://psychnews.psychiatryonline.org/doi/10.1176/appi.pn.2020.5b13. Published May 7, 2020. Accessed August 7, 2020.
5. Webb Hooper M, Nápoles AM, Perez-Stable EJ. COVID-19 and racial/ethnic disparities. JAMA. 2020;323(24):2466-2467.
6. Sederer LI, Lanzara CB, Essock SM, et al. Lessons learned from the New York State mental health response to the September 11, 2001, attacks. Psychiatr Serv. 2011;62(9):1085-1089.
7. World Health Organization. Infodemic management – infodemiology. https://www.who.int/teams/risk-communication/infodemic-management. Accessed August 7, 2020.
8. Zhou J, Liu L, Xue P, et al. Mental health response to the COVID-19 outbreak in China. Am J Psychiatry. 2020;117(7):574-575.
9. Kawohl W, Nordt C. COVID-19, unemployment, and suicide. Lancet Psychiatry. 2020;7(5):389-390.
10. Yao H, Chen JH, Xu YF. Patients with mental health disorders in the COVID-19 epidemic. Lancet Psychiatry. 2020;7(4):e21. doi: 10.1016/S2215-0366(20)30090-0.
11. Minihan E, Gavin B, Kelly BD, et al. Covid-19, mental health and psychological first aid. Ir J Psychol Med. 2020:1-12.
12. Adja KYC, Golinelli D, Lenzi J, et al. Pandemics and social stigma: who’s next? Italy’s experience with COVID-19. Public Health. 2020;185:39-41.
13. Rosenberg AR. Cultivating deliberate resilience during the coronavirus disease 2019 pandemic [published online April 14, 2020]. JAMA Pediatr. doi: 10.1001/jamapediatrics.2020.1436.
14. Dean W, Talbot SG, Caplan A. Clarifying the language of clinician distress [published online January 31, 2020]. JAMA. doi: 10.1001/jama.2019.21576.
15. Emanuel EJ, Persad G, Upshur R, et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055.
16. Rosenbaum L. Facing Covid-19 in Italy - ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020;382(20):1873-1875.
17. Viceconte G, Petrosillo N. COVID-19 R0: magic number or conundrum? Infect Dis Rep. 2020;12(1):8516.
18. de Hert M, Schreurs V, Vancampfort D, van Winkel R. Metabolic syndrome in people with schizophrenia: a review. World Psychiatry. 2009;8(1):15-22.
19. Chen R, Liang W, Jiang M, et al. Risk factors of fatal outcome in hospitalized subjects with coronavirus disease 2019 from a nationwide analysis in China. Chest. 2020;158(1):97-105.
20. Finer N, Garnett SP, Bruun JM. COVID-19 and obesity. Clin Obes. 2020;10(3):e12365. doi: 10.1111/cob.12365.
21. Havens LL, Ghaemi SN. Existential despair and bipolar disorder: the therapeutic alliance as a mood stabilizer. Am J Psychother. 2005;59(2):137-147.
22. Trémeau F, Antonius D, Malaspina D, et al. Loneliness in schizophrenia and its possible correlates. An exploratory study. Psychiatry Res. 2016;246:211-217.
23. Menninger KA. Psychoses associated with influenza: I. General data: statistical analysis. JAMA. 1919;72(4):235-241.
24. Asadi-Pooya AA, Simani L. Central nervous system manifestations of COVID-19: a systematic review. J Neurol Sci. 2020;413:116832. doi: 10.1016/j.jns.2020.116832.
25. Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.012.
26. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun. 2020;87:34-39.
27. Martin Jr. EB. Brief psychotic disorder triggered by fear of coronavirus? Psychiatric Times. https://www.psychiatrictimes.com/view/brief-psychotic-disorder-triggered-fear-coronavirus-small-case-series. Published May 8, 2020. Accessed August 7, 2020.
28. Sher Y, Rabkin B, Maldonado JR, et al. COVID-19-associated hyperactive intensive care unit delirium with proposed pathophysiology and treatment: a case report [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.007.
29. Wolters AE, Peelen LM, Welling MC, et al. Long-term mental health problems after delirium in the ICU. Crit Care Med. 2016;44(10):1808-1813.
30. Toovey S. Influenza-associated central nervous system dysfunction: a literature review. Travel Med Infect Dis. 2008;6(3):114-124.
31. Brooks SK, Webster RK, Smith LE, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. Lancet. 2020;395(10227):912-920.
32. Maercker A, Brewin CR, Bryant RA, et al. Diagnosis and classification of disorders specifically associated with stress: proposals for ICD-11. World Psychiatry. 2013;12(3):198-206.
33. Ornell F, Moura HF, Scherer JN, et al. The COVID-19 pandemic and its impact on substance use: implications for prevention and treatment. Psychiatry Res. 2020;289:113096. doi: 10.1016/j.psychres.2020.113096.
34. Berlin I, Thomas D, Le Faou AL, Cornuz J. COVID-19 and smoking [published online April 3, 2020]. Nicotine Tob Res. https://doi.org/10.1093/ntr/ntaa059.
35. Back D, Marzolini C, Hodge C, et al. COVID-19 treatment in patients with comorbidities: awareness of drug-drug interactions [published online May 8, 2020]. Br J Clin Pharmacol. doi: 10.1111/bcp.14358.
36. Carpenter WT Jr., Buchanan RW, Kirkpatrick B, et al. Diazepam treatment of early signs of exacerbation in schizophrenia. Am J Psychiatry. 1999;156(2):299-303.
37. Dotson S, Hartvigsen N, Wesner T, et al. Clozapine toxicity in the setting of COVID-19 [published online May 30, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.025.
38. Siskind D, Honer WG, Clark S, et al. Consensus statement on the use of clozapine during the COVID-19 pandemic. J Psychiatry Neurosci. 2020;45(3):222-223.
39. Schnitzer K, MacLaurin S, Freudenreich O. Long-acting injectable antipsychotics during the COVID-19 pandemic. Current Psychiatry. In press.
40. Winston A, Rosenthal RN, Pinsker H. Learning supportive psychotherapy: an illustrated guide. Washington, DC: American Psychiatric Publishing; 2012.
41. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
42. Jordan A, Dixon LB. Considerations for telepsychiatry service implementation in the era of COVID-19. Psychiatr Serv. 2020;71(6):643-644.
43. DePierro J, Lowe S, Katz C. Lessons learned from 9/11: mental health perspectives on the COVID-19 pandemic. Psychiatry Res. 2020;288:113024.
44. Hussain S. Immunization and vaccination. In: Huremovic
45. Epping-Jordan JE, van Ommeren M, Ashour HN, et al. Beyond the crisis: building back better mental health care in 10 emergency-affected areas using a longer-term perspective. Int J Ment Health Syst. 2015;9:15.
46. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382(24):2368-2371.
47. Bartels SJ, Baggett TP, Freudenreich O, et al. COVID-19 emergency reforms in Massachusetts to support behavioral health care and reduce mortality of people with serious mental illness [published online June 3, 2020]. Psychiatr Serv. doi: 10.1176/appi.ps.202000244.
“This whole thing is not about heroism. It’s about decency. It may seem a ridiculous idea, but the only way to fight the plague is with decency . ”
– Albert Camus, La Peste (1947)1
Severe acute respiratory syndrome (SARS), H1N1 swine flu, Ebola, Zika, and Middle East respiratory syndrome (MERS): the 21st century has already been witness to several serious infectious outbreaks and pandemics,2 but none has been as deadly and consequential as the current one. The ongoing SARS-coronavirus-2 (SARS-CoV-2) pandemic is shaping not only current psychiatric care but the future of psychiatry. Now that we are beyond the initial stages of the coronavirus disease 2019 (COVID-19) pandemic, when psychiatrists had a crash course in disaster psychiatry, our attention must shift to rebuilding and managing disillusionment and other psychological fallout of the intense early days.3
In this article, we offer guidance to psychiatrists caring for patients with serious mental illness (SMI) during the SARS-CoV-2 pandemic. Patients with SMI are easily forgotten as other issues (eg, preserving ICU capacity) overshadow the already historically neglected needs of this impoverished group.4 From both human and public-health perspectives, this inattention is a mistake. Assuring psychiatric stability is critically important to prevent the spread of COVID-19 in marginalized communities comprised of individuals who are poor, members of racial minorities, and others who already experience health disparities.5 Without controlling transmission in these groups, the pandemic will not be sufficiently contained.
We begin by highlighting general principles of pandemic management because caring for patients with SMI does not occur in a vacuum. Infectious outbreaks require not only helping those who need direct medical care because they are infected, but also managing populations that are at risk of getting infected, including health care and other essential workers.
Principles of pandemic management
Delivery of medical care during a pandemic differs from routine care. An effective disaster response requires collaboration and coordination among public-health, treatment, and emergency systems. Many institutions shift to an incident management system and crisis leadership, with clear lines of authority to coordinate responders and build medical surge capacity. Such a top-down leadership approach must plan and allow for the emergence of other credible leaders and for the restoration of people’s agency.
Unfortunately, adaptive capacity may be limited, especially in the public sector and psychiatric care system, where resources are already poor. Particularly early in a pandemic, services considered non-essential—which includes most psychiatric outpatient care—can become unavailable. A major effort is needed to prevent the psychiatric care system from contracting further, as happened during 9/11.6 Additionally, “essential” cannot be conflated with “emergent,” as can easily occur in extreme circumstances. Early and sustained efforts are required to ensure that patients with SMI who may be teetering on the edge of emergency status do not slip off that edge, especially when the emergency medical system is operating over capacity.
A comprehensive outbreak response must consider that a pandemic is not only a medical crisis but a mental health crisis and a communication emergency.7 Mental health clinicians need to provide accurate information and help patients cope with their fears.
Continue to: Psychological aspects of pandemics
Psychological aspects of pandemics. Previous infectious outbreaks have reaffirmed that mental health plays an outsized role during epidemics. Chaos, uncertainty, fear of death, and loss of income and housing cause prolonged stress and exact a psychological toll.
Adverse psychological impacts include expectable, normal reactions such as stress-induced anxiety or insomnia. In addition, new-onset psychiatric illnesses or exacerbations of existing ones may emerge.8 As disillusionment and demoralization appear in the wake of the acute phase, with persistently high unemployment, suicide prevention becomes an important goal.9
Pandemics lead to expectable behavioral responses (eg, increases in substance use and interpersonal conflict). Fear-based decisions may result in unhelpful behavior, such as hoarding medications (which may result in shortages) or dangerous, unsupervised use of unproven medications (eg, hydroxychloroquine). Trust is needed to accept public-health measures, and recommendations (eg, wearing masks) must be culturally informed to be credible and effective.
Because people are affected differently, at individual, cultural, and socioeconomic levels, they will view the situation differently. For many people, secondary stressors (eg, job loss) may be more disastrous than the primary medical event (ie, the pandemic). This distinction is critical because concrete financial help, not psychiatric care, is needed. Sometimes, even when a psychiatric disorder such as SMI or major neurocognitive disorder is present, the illusion of an acute decompensation can be created by the loss of social and structural supports that previously scaffolded a person’s life.
Mental illness prevention. Community mental-health surveillance is important to monitor for distress, psychiatric symptoms, health-risk behaviors, risk and safety perception, and preparedness. Clinicians must be ready to normalize expectable and temporary distress, while recognizing when that distress becomes pathological. This may be difficult in patients with SMI who often already have reduced stress tolerance or problem-based coping skills.10
Continue to: Psychological first aid...
Psychological first aid (PFA) is a standard intervention recommended by the World Health Organization for most individuals following a disaster; it is evidence-informed and has face validity.11 Intended to relieve distress by creating an environment that is safe, calm, and connected, PFA fosters self-efficacy and hope. While PFA is a form of universal prevention, it is not designed for patients with SMI, is not a psychiatric intervention, and is not provided by clinicians. Its principles, however, can easily be applied to patients with SMI to prevent distressing symptoms from becoming a relapse.
Communication. Good risk and crisis communication are critical because individual and population behavior will be governed by the perception of risk and fear, and not by facts. Failure to manage the “infodemic”7—with its misinformation, contradictory messages, and rumors—jeopardizes infection control if patients become paralyzed by uncertainty and fear. Scapegoating occurs easily during times of threat, and society must contain the parallel epidemic of xenophobia based on stigma and misinformation.12
Decision-making under uncertainty is not perfect and subject to revision as better information becomes available. Pointing this out to the public is delicate but essential to curtail skepticism and mistrust when policies are adjusted in response to new circumstances and knowledge.
Mistrust of an authority’s legitimacy and fear-based decisions lead to lack of cooperation with public-health measures, which can undermine an effective response to the pandemic. Travel restrictions or quarantine measures will not be followed if individuals question their importance. Like the general public, patients need education and clear communication to address their fear of contagion, dangers posed to family (and pets), and mistrust of authority and government. A lack of appreciation of the seriousness of the pandemic and individual responsibility may need to be addressed. Two important measures to accomplish this are steering patients to reputable sources of information and advising that they limit media exposure.
Resilience-building. Community and workplace resilience are important aspects of making it through a disaster as best as possible. Resilience is not innate and fixed; it must be deliberately built.13 Choosing an attitude of post-traumatic growth over the victim narrative is a helpful stance. Practicing self-care (rest, nutrition, exercise) and self-compassion (self-kindness, common humanity, mindfulness) is good advice for patients and caregivers alike.
Continue to: Workforce protection
Workforce protection. Compared to other disasters, infectious outbreaks disproportionally affect the medical community, and care delivery is at stake. While psychological and psychiatric needs may increase during a pandemic, services often contract, day programs and clinics close, teams are reduced to skeleton crews, and only emergency psychiatric care is available. Workforce protection is critical to avoid illness or simple absenteeism due to mistrust of protective measures.
Only a well-briefed, well-led, well-supported, and adequately resourced workforce is going to be effective in managing this public-health emergency. Burnout and moral injury are feared long-term consequences for health care workers that need to be proactively addressed.14 As opposed to other forms of disasters, managing your own fears about safety is important. Clinicians and their patients sit in the proverbial same boat.
Ethics. The anticipated need to ration life-saving care (eg, ventilators) has been at the forefront of ethical concerns.15 In psychiatry, the question of involuntary public-health interventions for uncooperative psychiatric patients sits uncomfortably between public-health ethics and human rights, and is an opportunity for collaboration with public-health and infectious-disease colleagues.
Redeployed clinicians and those working under substandard conditions may be concerned about civil liability due to a modified standard of care during a crisis. Some clinicians may ask if their duty to care must override their natural instinct to protect themselves. There is a lot of room for resentment in these circumstances. Redeployed or otherwise “conscripted” clinicians may resent administrators, especially those administering from the safety of their homes. Those “left behind” to work in potentially precarious circumstances may resent their absent colleagues. Moreover, these front-line clinicians may have been forced to make ethical decisions for which they were not prepared.16 Maintaining morale is far from trivial, not just during the pandemic, but afterward, when (and if) the entire workforce is reunited. All parties need to be mindful of how their actions and decisions impact and are perceived by others, both in the hospital and at home.
Managing patients with SMI during COVID-19
Patients with SMI are potentially hard hit by COVID-19 due to a “tragic” epidemiologic triad of agent-host-environment: SARS-CoV-2 is a highly infectious agent affecting patients with SMI who are vulnerable hosts in permissive environments (Figure).

Continue to: While not as infectious as measles...
While not as infectious as measles, COVID-19 is more infectious than the seasonal flu virus.17 It can lead to uncontrolled infection within a short period of time, particularly in enclosed settings. Outbreaks have occurred readily on cruise ships and aircraft carriers as well as in nursing homes, homeless shelters, prisons, and group homes.
Patients with SMI are vulnerable hosts because they have many of the medical risk factors18 that portend a poor prognosis if they become infected, including pre-existing lung conditions and heart disease19 as well as diabetes and obesity.20 Obesity likely creates a hyperinflammatory state and a decrease in vital capacity. Patient-related behavioral factors include poor early-symptom reporting and ineffective infection control.
Unfavorable social determinants of health include not only poverty but crowded housing that is a perfect incubator for COVID-19.
Priority treatment goals. The overarching goal during a pandemic is to keep patients with SMI in psychiatric treatment and prevent them from disengaging from care in the service of infection control. Urgent tasks include infection control, relapse prevention, and preventing treatment disengagement and loneliness.
Infection control. As trusted sources of information, psychiatrists can play an important role in infection control in several important ways:
- educating patients about infection-control measures and public-health recommendations
- helping patients understand what testing can accomplish and when to pursue it
- encouraging protective health behaviors (eg, hand washing, mask wearing, physical distancing)
- assessing patients’ risk appreciation
- assessing for and addressing obstacles to implementing and complying with infection-control measures
- explaining contact tracing
- providing reassurance.
Continue to: Materials and explanations...
Materials and explanations must be adapted for patient understanding.
Patients with disorganization or cognitive disturbances may have difficulties cooperating or problem-solving. Patients with negative symptoms may be inappropriately unconcerned and also inaccurately report symptoms that suggest COVID-19. Acute psychosis or mania can prevent patients from complying with public-health efforts. Some measures may be difficult to implement if the means are simply not there (eg, physical distancing in a crowded apartment). Previously open settings (eg, group homes) have had to develop new mechanisms under the primacy of infection control. Inpatient units—traditionally places where community, shared healing, and group therapy are prized—have had to decrease maximum occupancy, limit the number of patients attending groups, and discourage or outrightly prohibit social interaction (eg, dining together).
Relapse prevention. Patients who take maintenance medications need to be supported. A manic or psychotic relapse during a pandemic puts patients at risk of acquiring and spreading COVID-19. “Treatment as prevention” is a slogan from human immunodeficiency virus (HIV) care that captures the importance of antiretroviral treatment to prevent medical complications from HIV, and also to reduce infecting other people. By analogy, psychiatric treatment for patients with SMI can prevent psychiatric instability and thereby control viral transmission. Avoiding sending psychiatric patients to a potentially stressed acute-care system is important.
Psychosocial support. Clinics need to ensure that patients continue to engage in care beyond medication-taking to proactively prevent psychiatric exacerbations. Healthful, resilience-building behaviors should be encouraged while monitoring and counseling against maladaptive ones (eg, increased substance use). Supporting patients emotionally and helping them solve problems are critical, particularly for those who are subjected to quarantine or isolation. Obviously, in these latter situations, outreach will be necessary and may require creative delivery systems and dedicated clinicians for patients who lack access to the technology necessary for virtual visits. Havens and Ghaemi21 have suggested that a good therapeutic alliance can be viewed as a mood stabilizer. Helping patients grieve losses (loved ones, jobs, sense of safety) may be an important part of support.
Even before COVID-19, loneliness was a major factor for patients with schizophrenia.22 A psychiatric clinic is one aspect of a person with SMI’s social network; during the initial phase of the pandemic, many clinics and treatment programs closed. Patients for whom clinics structure and anchor their activities are at high risk of disconnecting from treatment, staying at home, and becoming lonely.
Continue to: Caregivers are always important...
Caregivers are always important to SMI patients, but they may assume an even bigger role during this pandemic. Some patients may have moved in with a relative, after years of living on their own. In other cases, stable caregiver relationships may be disrupted due to COVID-19–related sickness in the caregiver; if not addressed, this can result in a patient’s clinical decompensation. Clinicians should take the opportunity to understand who a patient’s caregivers are (group home staff, families) and rekindle clinical contact with them. Relationships with caregivers that may have been on “autopilot” during normal times are opportunities for welcome support and guidance, to the benefit of both patients and caregivers.
Table 1 summarizes clinical tasks that need to be kept in mind when conducting clinic visits during COVID-19 in order to achieve the high-priority treatment goals of infection control, relapse prevention, and psychosocial support.
Differential diagnosis. Neuropsychiatric syndromes have long been observed in influenza pandemics,23 due both to direct viral effects and to the effects of critical illness on the brain. Two core symptoms of COVID-19—anosmia and ageusia—suggest that COVID-19 can directly affect the brain. While neurologic manifestations are common,24 it remains unclear to what extent COVID-19 can directly “cause” psychiatric symptoms, or if such symptoms are the result of cytokines25 or other medical processes (eg, thromboembolism).26 Psychosis due to COVID-19 may, in some cases, represent a stress-related brief psychotic disorder.27
Hospitalized patients who have recovered from COVID-19 may have experienced prolonged sedation and severe delirium in an ICU.28 Complications such as posttraumatic stress disorder,29 hypoperfusion-related brain injuries, or other long-term cognitive difficulties may result. In previous flu epidemics, patients developed serious neurologic complications such as post-encephalitic Parkinson’s disease.30
Any person subjected to isolation or quarantine is at risk for psychiatric complications.31 Patients with SMI who live in group homes may be particularly susceptible to new rules, including no-visitor policies.
Continue to: Outpatients whose primary disorder...
Outpatients whose primary disorder is well controlled may, like anyone else, struggle with the effects of the pandemic. It is necessary to carefully differentiate non-specific symptoms associated with stress from the emergence of a new disorder resulting from stress.32 For some patients, grief or adjustment disorders should be considered. Prolonged stress and uncertainty may eventually lead to an exacerbation of a primary disorder, particularly if the situation (eg, financial loss) does not improve or worsens. Demoralization and suicidal thinking need to be monitored. Relapse or increased use of alcohol or other substances as a response to stress may also complicate the clinical picture.33 Last, smoking cessation as a major treatment goal in general should be re-emphasized and not ignored during the ongoing pandemic.34
Table 2 summarizes psychiatric symptoms that need to be considered when managing a patient with SMI during this pandemic.
Treatment tools
Psychopharmacology. Even though crisis-mode prescribing may be necessary, the safe use of psychotropics remains the goal of psychiatric prescribing. Access to medications becomes a larger consideration; for many patients, a 90-day supply may be indicated. Review of polypharmacy, including for pneumonia risk, should be undertaken. Preventing drooling (eg, from sedation, clozapine, extrapyramidal symptoms [EPS]) will decrease aspiration risk.
In general, treatment of psychiatric symptoms in a patient with COVID-19 follows usual guidelines. The best treatment for COVID-19 patients with delirium, however, remains to be established, particularly how to manage severe agitation.28 Pharmacodynamic and pharmacokinetic drug–drug interactions between psychotropics and antiviral treatments for COVID-19 (eg, QTc prolongation) can be expected and need to be reviewed.35 For stress-related anxiety, judicious pharmacotherapy can be helpful. Diazepam given at the earliest signs of a psychotic relapse may stave off a relapse for patients with schizophrenia.36 Even if permitted under relaxed prescribing rules during a public-health emergency, prescribing controlled substances without seeing patients in person requires additional thought. In some cases, adjusting the primary medication to buffer against stress may be preferred (eg, adjusting an antipsychotic in a patient on maintenance treatment for schizophrenia, particularly if a low-dose strategy is pursued).
Clozapine requires registry-based prescribing and bloodwork (“no blood, no drug”). The use of clozapine during this public-health emergency has been made easier because of FDA guidance that allows clozapine to be dispensed without blood work if obtaining blood work is not possible (eg, a patient is quarantined) or can be accomplished only at substantial risk to patients and the population at large. Under certain conditions, clozapine can be dispensed safely and in a way that is consistent with infection prevention. Clozapine-treated patients admitted with COVID-19 should be monitored for clozapine toxicity and the clozapine dose adjusted.37 A consensus statement consistent with the FDA and clinical considerations for using clozapine during COVID-19 is summarized in Table 3.38
Continue to: Long-acting injectable antipsychotics...
Long-acting injectable antipsychotics (LAIs) pose a problem because they require in-person visits. Ideally, during a pandemic, patients should be seen in person as frequently as medically necessary but as infrequently as possible to limit exposure of both patients and staff. Table 4 provides some clinical recommendations on how to use LAIs during the pandemic.39
Supportive psychotherapy may be the most important tool we have in helping patients with loss and uncertainty during these challenging months.40 Simply staying in contact with patients plays a major role in preventing care discontinuity. Even routine interactions have become stressful, with everyone wearing a mask that partially obscures the face. People with impaired hearing may find it even more difficult to understand you.
Education, problem-solving, and a directive, encouraging style are major tools of supportive psychotherapy to reduce symptoms and increase adaptive skills. Clarify that social distancing refers to physical, not emotional, distancing. The judicious and temporary use of anxiolytics is appropriate to reduce anxiety. Concrete help and problem-solving (eg, filling out forms) are examples of proactive crisis intervention.
Telepsychiatry emerged in the pandemic’s early days as the default mode of practice in order to limit in-person contacts.41 Like all new technology, telepsychiatry brings progress and peril.42 While it has gone surprisingly well for most, the “digital divide” does not afford all patients access to the needed technology. The long-term effectiveness and acceptance of telehealth remain to be seen. (Editor’s Note: For more about this topic, see “Telepsychiatry: What you need to know.”
Lessons learned and outlook
Infectious outbreaks have historically inflicted long-term disruptions on societies and altered the course of history. However, each disaster is unique, and lessons from previous disasters may only partially apply.43 We do not yet know how this one will end, including how long it will take for the world’s economies to recover. If nothing else, the current public-health emergency has brought to the forefront what psychiatrists have always known: health disparities are partially responsible for different disease risks (in this case, the risk of getting infected with SARS-CoV-2).5 It may not be a coincidence that the Black Lives Matter movement is becoming a major impetus for social change at a time when the pandemic is exposing health-care inequalities.
Continue to: Some areas of the country...
Some areas of the country succeeded in reducing infections and limiting community spread, which ushered in an uneasy sense of normalcy even while the pandemic continues. At least for now, these locales can focus on rebuilding and preparing for expectable fluctuations in disease activity, including the arrival of the annual flu season on top of COVID-19.44 Recovery is not a return to the status quo ante but building stronger communities—“building back better.”45 Unless there is a continuum of care, shortcomings in one sector will have ripple effects through the entire system, particularly for psychiatric care for patients with SMI, which was inadequate before the pandemic.
Ensuring access to critical care was a priority during the pandemic’s early phase but came at the price of deferring other types of care, such as routine primary care; the coming months will see the downstream consequences of this approach,46 including for patients with SMI.
In the meantime, doing our job as clinicians, as Camus’s fictitious Dr. Bernard Rieux from the epigraph responds when asked how to define decency, may be the best we can do in these times. This includes contributing to and molding our field’s future and fostering a sense of agency in our patients and in ourselves. Major goals will be to preserve lessons learned, maintain flexibility, and avoid a return to unhelpful overregulation and payment models that do not reflect the flexible, person-centered care so important for patients with SMI.47
Bottom Line
During a pandemic, patients with serious mental illness may be easily forgotten as other issues overshadow the needs of this impoverished group. During a pandemic, the priority treatment goals for these patients are infection control, relapse prevention, and preventing treatment disengagement and loneliness. A pandemic requires changes in how patients with serious mental illness will receive psychopharmacology and psychotherapy.
Related Resources
- Huremović D (ed). Psychiatry of pandemics: a mental health response to infection outbreak. Cham, Switzerland: Springer Nature Switzerland AG; 2019.
- Ursano RJ, Fullerton CS, Weisaeth L, et al (eds). Textbook of disaster psychiatry. 2nd ed. Cambridge, UK: Cambridge University Press; 2017.
- Centers for Disease Control and Prevention. Coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/index.html.
- American Psychiatric Association. Coronavirus resources. https://www.psychiatry.org/psychiatrists/covid-19-coronavirus.
- SMI Adviser. Make informed decisions related to COVID-19 and mental health. https://smiadviser.org/about/covid.
Drug Brand Names
Clozapine • Clozaril
Diazepam • Valium
Hydroxychloroquine • Plaquenil
“This whole thing is not about heroism. It’s about decency. It may seem a ridiculous idea, but the only way to fight the plague is with decency . ”
– Albert Camus, La Peste (1947)1
Severe acute respiratory syndrome (SARS), H1N1 swine flu, Ebola, Zika, and Middle East respiratory syndrome (MERS): the 21st century has already been witness to several serious infectious outbreaks and pandemics,2 but none has been as deadly and consequential as the current one. The ongoing SARS-coronavirus-2 (SARS-CoV-2) pandemic is shaping not only current psychiatric care but the future of psychiatry. Now that we are beyond the initial stages of the coronavirus disease 2019 (COVID-19) pandemic, when psychiatrists had a crash course in disaster psychiatry, our attention must shift to rebuilding and managing disillusionment and other psychological fallout of the intense early days.3
In this article, we offer guidance to psychiatrists caring for patients with serious mental illness (SMI) during the SARS-CoV-2 pandemic. Patients with SMI are easily forgotten as other issues (eg, preserving ICU capacity) overshadow the already historically neglected needs of this impoverished group.4 From both human and public-health perspectives, this inattention is a mistake. Assuring psychiatric stability is critically important to prevent the spread of COVID-19 in marginalized communities comprised of individuals who are poor, members of racial minorities, and others who already experience health disparities.5 Without controlling transmission in these groups, the pandemic will not be sufficiently contained.
We begin by highlighting general principles of pandemic management because caring for patients with SMI does not occur in a vacuum. Infectious outbreaks require not only helping those who need direct medical care because they are infected, but also managing populations that are at risk of getting infected, including health care and other essential workers.
Principles of pandemic management
Delivery of medical care during a pandemic differs from routine care. An effective disaster response requires collaboration and coordination among public-health, treatment, and emergency systems. Many institutions shift to an incident management system and crisis leadership, with clear lines of authority to coordinate responders and build medical surge capacity. Such a top-down leadership approach must plan and allow for the emergence of other credible leaders and for the restoration of people’s agency.
Unfortunately, adaptive capacity may be limited, especially in the public sector and psychiatric care system, where resources are already poor. Particularly early in a pandemic, services considered non-essential—which includes most psychiatric outpatient care—can become unavailable. A major effort is needed to prevent the psychiatric care system from contracting further, as happened during 9/11.6 Additionally, “essential” cannot be conflated with “emergent,” as can easily occur in extreme circumstances. Early and sustained efforts are required to ensure that patients with SMI who may be teetering on the edge of emergency status do not slip off that edge, especially when the emergency medical system is operating over capacity.
A comprehensive outbreak response must consider that a pandemic is not only a medical crisis but a mental health crisis and a communication emergency.7 Mental health clinicians need to provide accurate information and help patients cope with their fears.
Continue to: Psychological aspects of pandemics
Psychological aspects of pandemics. Previous infectious outbreaks have reaffirmed that mental health plays an outsized role during epidemics. Chaos, uncertainty, fear of death, and loss of income and housing cause prolonged stress and exact a psychological toll.
Adverse psychological impacts include expectable, normal reactions such as stress-induced anxiety or insomnia. In addition, new-onset psychiatric illnesses or exacerbations of existing ones may emerge.8 As disillusionment and demoralization appear in the wake of the acute phase, with persistently high unemployment, suicide prevention becomes an important goal.9
Pandemics lead to expectable behavioral responses (eg, increases in substance use and interpersonal conflict). Fear-based decisions may result in unhelpful behavior, such as hoarding medications (which may result in shortages) or dangerous, unsupervised use of unproven medications (eg, hydroxychloroquine). Trust is needed to accept public-health measures, and recommendations (eg, wearing masks) must be culturally informed to be credible and effective.
Because people are affected differently, at individual, cultural, and socioeconomic levels, they will view the situation differently. For many people, secondary stressors (eg, job loss) may be more disastrous than the primary medical event (ie, the pandemic). This distinction is critical because concrete financial help, not psychiatric care, is needed. Sometimes, even when a psychiatric disorder such as SMI or major neurocognitive disorder is present, the illusion of an acute decompensation can be created by the loss of social and structural supports that previously scaffolded a person’s life.
Mental illness prevention. Community mental-health surveillance is important to monitor for distress, psychiatric symptoms, health-risk behaviors, risk and safety perception, and preparedness. Clinicians must be ready to normalize expectable and temporary distress, while recognizing when that distress becomes pathological. This may be difficult in patients with SMI who often already have reduced stress tolerance or problem-based coping skills.10
Continue to: Psychological first aid...
Psychological first aid (PFA) is a standard intervention recommended by the World Health Organization for most individuals following a disaster; it is evidence-informed and has face validity.11 Intended to relieve distress by creating an environment that is safe, calm, and connected, PFA fosters self-efficacy and hope. While PFA is a form of universal prevention, it is not designed for patients with SMI, is not a psychiatric intervention, and is not provided by clinicians. Its principles, however, can easily be applied to patients with SMI to prevent distressing symptoms from becoming a relapse.
Communication. Good risk and crisis communication are critical because individual and population behavior will be governed by the perception of risk and fear, and not by facts. Failure to manage the “infodemic”7—with its misinformation, contradictory messages, and rumors—jeopardizes infection control if patients become paralyzed by uncertainty and fear. Scapegoating occurs easily during times of threat, and society must contain the parallel epidemic of xenophobia based on stigma and misinformation.12
Decision-making under uncertainty is not perfect and subject to revision as better information becomes available. Pointing this out to the public is delicate but essential to curtail skepticism and mistrust when policies are adjusted in response to new circumstances and knowledge.
Mistrust of an authority’s legitimacy and fear-based decisions lead to lack of cooperation with public-health measures, which can undermine an effective response to the pandemic. Travel restrictions or quarantine measures will not be followed if individuals question their importance. Like the general public, patients need education and clear communication to address their fear of contagion, dangers posed to family (and pets), and mistrust of authority and government. A lack of appreciation of the seriousness of the pandemic and individual responsibility may need to be addressed. Two important measures to accomplish this are steering patients to reputable sources of information and advising that they limit media exposure.
Resilience-building. Community and workplace resilience are important aspects of making it through a disaster as best as possible. Resilience is not innate and fixed; it must be deliberately built.13 Choosing an attitude of post-traumatic growth over the victim narrative is a helpful stance. Practicing self-care (rest, nutrition, exercise) and self-compassion (self-kindness, common humanity, mindfulness) is good advice for patients and caregivers alike.
Continue to: Workforce protection
Workforce protection. Compared to other disasters, infectious outbreaks disproportionally affect the medical community, and care delivery is at stake. While psychological and psychiatric needs may increase during a pandemic, services often contract, day programs and clinics close, teams are reduced to skeleton crews, and only emergency psychiatric care is available. Workforce protection is critical to avoid illness or simple absenteeism due to mistrust of protective measures.
Only a well-briefed, well-led, well-supported, and adequately resourced workforce is going to be effective in managing this public-health emergency. Burnout and moral injury are feared long-term consequences for health care workers that need to be proactively addressed.14 As opposed to other forms of disasters, managing your own fears about safety is important. Clinicians and their patients sit in the proverbial same boat.
Ethics. The anticipated need to ration life-saving care (eg, ventilators) has been at the forefront of ethical concerns.15 In psychiatry, the question of involuntary public-health interventions for uncooperative psychiatric patients sits uncomfortably between public-health ethics and human rights, and is an opportunity for collaboration with public-health and infectious-disease colleagues.
Redeployed clinicians and those working under substandard conditions may be concerned about civil liability due to a modified standard of care during a crisis. Some clinicians may ask if their duty to care must override their natural instinct to protect themselves. There is a lot of room for resentment in these circumstances. Redeployed or otherwise “conscripted” clinicians may resent administrators, especially those administering from the safety of their homes. Those “left behind” to work in potentially precarious circumstances may resent their absent colleagues. Moreover, these front-line clinicians may have been forced to make ethical decisions for which they were not prepared.16 Maintaining morale is far from trivial, not just during the pandemic, but afterward, when (and if) the entire workforce is reunited. All parties need to be mindful of how their actions and decisions impact and are perceived by others, both in the hospital and at home.
Managing patients with SMI during COVID-19
Patients with SMI are potentially hard hit by COVID-19 due to a “tragic” epidemiologic triad of agent-host-environment: SARS-CoV-2 is a highly infectious agent affecting patients with SMI who are vulnerable hosts in permissive environments (Figure).

Continue to: While not as infectious as measles...
While not as infectious as measles, COVID-19 is more infectious than the seasonal flu virus.17 It can lead to uncontrolled infection within a short period of time, particularly in enclosed settings. Outbreaks have occurred readily on cruise ships and aircraft carriers as well as in nursing homes, homeless shelters, prisons, and group homes.
Patients with SMI are vulnerable hosts because they have many of the medical risk factors18 that portend a poor prognosis if they become infected, including pre-existing lung conditions and heart disease19 as well as diabetes and obesity.20 Obesity likely creates a hyperinflammatory state and a decrease in vital capacity. Patient-related behavioral factors include poor early-symptom reporting and ineffective infection control.
Unfavorable social determinants of health include not only poverty but crowded housing that is a perfect incubator for COVID-19.
Priority treatment goals. The overarching goal during a pandemic is to keep patients with SMI in psychiatric treatment and prevent them from disengaging from care in the service of infection control. Urgent tasks include infection control, relapse prevention, and preventing treatment disengagement and loneliness.
Infection control. As trusted sources of information, psychiatrists can play an important role in infection control in several important ways:
- educating patients about infection-control measures and public-health recommendations
- helping patients understand what testing can accomplish and when to pursue it
- encouraging protective health behaviors (eg, hand washing, mask wearing, physical distancing)
- assessing patients’ risk appreciation
- assessing for and addressing obstacles to implementing and complying with infection-control measures
- explaining contact tracing
- providing reassurance.
Continue to: Materials and explanations...
Materials and explanations must be adapted for patient understanding.
Patients with disorganization or cognitive disturbances may have difficulties cooperating or problem-solving. Patients with negative symptoms may be inappropriately unconcerned and also inaccurately report symptoms that suggest COVID-19. Acute psychosis or mania can prevent patients from complying with public-health efforts. Some measures may be difficult to implement if the means are simply not there (eg, physical distancing in a crowded apartment). Previously open settings (eg, group homes) have had to develop new mechanisms under the primacy of infection control. Inpatient units—traditionally places where community, shared healing, and group therapy are prized—have had to decrease maximum occupancy, limit the number of patients attending groups, and discourage or outrightly prohibit social interaction (eg, dining together).
Relapse prevention. Patients who take maintenance medications need to be supported. A manic or psychotic relapse during a pandemic puts patients at risk of acquiring and spreading COVID-19. “Treatment as prevention” is a slogan from human immunodeficiency virus (HIV) care that captures the importance of antiretroviral treatment to prevent medical complications from HIV, and also to reduce infecting other people. By analogy, psychiatric treatment for patients with SMI can prevent psychiatric instability and thereby control viral transmission. Avoiding sending psychiatric patients to a potentially stressed acute-care system is important.
Psychosocial support. Clinics need to ensure that patients continue to engage in care beyond medication-taking to proactively prevent psychiatric exacerbations. Healthful, resilience-building behaviors should be encouraged while monitoring and counseling against maladaptive ones (eg, increased substance use). Supporting patients emotionally and helping them solve problems are critical, particularly for those who are subjected to quarantine or isolation. Obviously, in these latter situations, outreach will be necessary and may require creative delivery systems and dedicated clinicians for patients who lack access to the technology necessary for virtual visits. Havens and Ghaemi21 have suggested that a good therapeutic alliance can be viewed as a mood stabilizer. Helping patients grieve losses (loved ones, jobs, sense of safety) may be an important part of support.
Even before COVID-19, loneliness was a major factor for patients with schizophrenia.22 A psychiatric clinic is one aspect of a person with SMI’s social network; during the initial phase of the pandemic, many clinics and treatment programs closed. Patients for whom clinics structure and anchor their activities are at high risk of disconnecting from treatment, staying at home, and becoming lonely.
Continue to: Caregivers are always important...
Caregivers are always important to SMI patients, but they may assume an even bigger role during this pandemic. Some patients may have moved in with a relative, after years of living on their own. In other cases, stable caregiver relationships may be disrupted due to COVID-19–related sickness in the caregiver; if not addressed, this can result in a patient’s clinical decompensation. Clinicians should take the opportunity to understand who a patient’s caregivers are (group home staff, families) and rekindle clinical contact with them. Relationships with caregivers that may have been on “autopilot” during normal times are opportunities for welcome support and guidance, to the benefit of both patients and caregivers.
Table 1 summarizes clinical tasks that need to be kept in mind when conducting clinic visits during COVID-19 in order to achieve the high-priority treatment goals of infection control, relapse prevention, and psychosocial support.
Differential diagnosis. Neuropsychiatric syndromes have long been observed in influenza pandemics,23 due both to direct viral effects and to the effects of critical illness on the brain. Two core symptoms of COVID-19—anosmia and ageusia—suggest that COVID-19 can directly affect the brain. While neurologic manifestations are common,24 it remains unclear to what extent COVID-19 can directly “cause” psychiatric symptoms, or if such symptoms are the result of cytokines25 or other medical processes (eg, thromboembolism).26 Psychosis due to COVID-19 may, in some cases, represent a stress-related brief psychotic disorder.27
Hospitalized patients who have recovered from COVID-19 may have experienced prolonged sedation and severe delirium in an ICU.28 Complications such as posttraumatic stress disorder,29 hypoperfusion-related brain injuries, or other long-term cognitive difficulties may result. In previous flu epidemics, patients developed serious neurologic complications such as post-encephalitic Parkinson’s disease.30
Any person subjected to isolation or quarantine is at risk for psychiatric complications.31 Patients with SMI who live in group homes may be particularly susceptible to new rules, including no-visitor policies.
Continue to: Outpatients whose primary disorder...
Outpatients whose primary disorder is well controlled may, like anyone else, struggle with the effects of the pandemic. It is necessary to carefully differentiate non-specific symptoms associated with stress from the emergence of a new disorder resulting from stress.32 For some patients, grief or adjustment disorders should be considered. Prolonged stress and uncertainty may eventually lead to an exacerbation of a primary disorder, particularly if the situation (eg, financial loss) does not improve or worsens. Demoralization and suicidal thinking need to be monitored. Relapse or increased use of alcohol or other substances as a response to stress may also complicate the clinical picture.33 Last, smoking cessation as a major treatment goal in general should be re-emphasized and not ignored during the ongoing pandemic.34
Table 2 summarizes psychiatric symptoms that need to be considered when managing a patient with SMI during this pandemic.
Treatment tools
Psychopharmacology. Even though crisis-mode prescribing may be necessary, the safe use of psychotropics remains the goal of psychiatric prescribing. Access to medications becomes a larger consideration; for many patients, a 90-day supply may be indicated. Review of polypharmacy, including for pneumonia risk, should be undertaken. Preventing drooling (eg, from sedation, clozapine, extrapyramidal symptoms [EPS]) will decrease aspiration risk.
In general, treatment of psychiatric symptoms in a patient with COVID-19 follows usual guidelines. The best treatment for COVID-19 patients with delirium, however, remains to be established, particularly how to manage severe agitation.28 Pharmacodynamic and pharmacokinetic drug–drug interactions between psychotropics and antiviral treatments for COVID-19 (eg, QTc prolongation) can be expected and need to be reviewed.35 For stress-related anxiety, judicious pharmacotherapy can be helpful. Diazepam given at the earliest signs of a psychotic relapse may stave off a relapse for patients with schizophrenia.36 Even if permitted under relaxed prescribing rules during a public-health emergency, prescribing controlled substances without seeing patients in person requires additional thought. In some cases, adjusting the primary medication to buffer against stress may be preferred (eg, adjusting an antipsychotic in a patient on maintenance treatment for schizophrenia, particularly if a low-dose strategy is pursued).
Clozapine requires registry-based prescribing and bloodwork (“no blood, no drug”). The use of clozapine during this public-health emergency has been made easier because of FDA guidance that allows clozapine to be dispensed without blood work if obtaining blood work is not possible (eg, a patient is quarantined) or can be accomplished only at substantial risk to patients and the population at large. Under certain conditions, clozapine can be dispensed safely and in a way that is consistent with infection prevention. Clozapine-treated patients admitted with COVID-19 should be monitored for clozapine toxicity and the clozapine dose adjusted.37 A consensus statement consistent with the FDA and clinical considerations for using clozapine during COVID-19 is summarized in Table 3.38
Continue to: Long-acting injectable antipsychotics...
Long-acting injectable antipsychotics (LAIs) pose a problem because they require in-person visits. Ideally, during a pandemic, patients should be seen in person as frequently as medically necessary but as infrequently as possible to limit exposure of both patients and staff. Table 4 provides some clinical recommendations on how to use LAIs during the pandemic.39
Supportive psychotherapy may be the most important tool we have in helping patients with loss and uncertainty during these challenging months.40 Simply staying in contact with patients plays a major role in preventing care discontinuity. Even routine interactions have become stressful, with everyone wearing a mask that partially obscures the face. People with impaired hearing may find it even more difficult to understand you.
Education, problem-solving, and a directive, encouraging style are major tools of supportive psychotherapy to reduce symptoms and increase adaptive skills. Clarify that social distancing refers to physical, not emotional, distancing. The judicious and temporary use of anxiolytics is appropriate to reduce anxiety. Concrete help and problem-solving (eg, filling out forms) are examples of proactive crisis intervention.
Telepsychiatry emerged in the pandemic’s early days as the default mode of practice in order to limit in-person contacts.41 Like all new technology, telepsychiatry brings progress and peril.42 While it has gone surprisingly well for most, the “digital divide” does not afford all patients access to the needed technology. The long-term effectiveness and acceptance of telehealth remain to be seen. (Editor’s Note: For more about this topic, see “Telepsychiatry: What you need to know.”
Lessons learned and outlook
Infectious outbreaks have historically inflicted long-term disruptions on societies and altered the course of history. However, each disaster is unique, and lessons from previous disasters may only partially apply.43 We do not yet know how this one will end, including how long it will take for the world’s economies to recover. If nothing else, the current public-health emergency has brought to the forefront what psychiatrists have always known: health disparities are partially responsible for different disease risks (in this case, the risk of getting infected with SARS-CoV-2).5 It may not be a coincidence that the Black Lives Matter movement is becoming a major impetus for social change at a time when the pandemic is exposing health-care inequalities.
Continue to: Some areas of the country...
Some areas of the country succeeded in reducing infections and limiting community spread, which ushered in an uneasy sense of normalcy even while the pandemic continues. At least for now, these locales can focus on rebuilding and preparing for expectable fluctuations in disease activity, including the arrival of the annual flu season on top of COVID-19.44 Recovery is not a return to the status quo ante but building stronger communities—“building back better.”45 Unless there is a continuum of care, shortcomings in one sector will have ripple effects through the entire system, particularly for psychiatric care for patients with SMI, which was inadequate before the pandemic.
Ensuring access to critical care was a priority during the pandemic’s early phase but came at the price of deferring other types of care, such as routine primary care; the coming months will see the downstream consequences of this approach,46 including for patients with SMI.
In the meantime, doing our job as clinicians, as Camus’s fictitious Dr. Bernard Rieux from the epigraph responds when asked how to define decency, may be the best we can do in these times. This includes contributing to and molding our field’s future and fostering a sense of agency in our patients and in ourselves. Major goals will be to preserve lessons learned, maintain flexibility, and avoid a return to unhelpful overregulation and payment models that do not reflect the flexible, person-centered care so important for patients with SMI.47
Bottom Line
During a pandemic, patients with serious mental illness may be easily forgotten as other issues overshadow the needs of this impoverished group. During a pandemic, the priority treatment goals for these patients are infection control, relapse prevention, and preventing treatment disengagement and loneliness. A pandemic requires changes in how patients with serious mental illness will receive psychopharmacology and psychotherapy.
Related Resources
- Huremović D (ed). Psychiatry of pandemics: a mental health response to infection outbreak. Cham, Switzerland: Springer Nature Switzerland AG; 2019.
- Ursano RJ, Fullerton CS, Weisaeth L, et al (eds). Textbook of disaster psychiatry. 2nd ed. Cambridge, UK: Cambridge University Press; 2017.
- Centers for Disease Control and Prevention. Coronavirus (COVID-19). https://www.cdc.gov/coronavirus/2019-ncov/index.html.
- American Psychiatric Association. Coronavirus resources. https://www.psychiatry.org/psychiatrists/covid-19-coronavirus.
- SMI Adviser. Make informed decisions related to COVID-19 and mental health. https://smiadviser.org/about/covid.
Drug Brand Names
Clozapine • Clozaril
Diazepam • Valium
Hydroxychloroquine • Plaquenil
1. Camus A. La peste. Paris, France: Éditions Gallimard; 1947.
2. Huremovic
3. Substance Abuse and Mental Health Services Administration. Phases of disaster. https://www.samhsa.gov/dtac/recovering-disasters/phases-disaster. Updated June 17, 2020. Accessed August 7, 2020.
4. Geller J. COVID-19 and advocacy—the good and the unacceptable. Psychiatric News. https://psychnews.psychiatryonline.org/doi/10.1176/appi.pn.2020.5b13. Published May 7, 2020. Accessed August 7, 2020.
5. Webb Hooper M, Nápoles AM, Perez-Stable EJ. COVID-19 and racial/ethnic disparities. JAMA. 2020;323(24):2466-2467.
6. Sederer LI, Lanzara CB, Essock SM, et al. Lessons learned from the New York State mental health response to the September 11, 2001, attacks. Psychiatr Serv. 2011;62(9):1085-1089.
7. World Health Organization. Infodemic management – infodemiology. https://www.who.int/teams/risk-communication/infodemic-management. Accessed August 7, 2020.
8. Zhou J, Liu L, Xue P, et al. Mental health response to the COVID-19 outbreak in China. Am J Psychiatry. 2020;117(7):574-575.
9. Kawohl W, Nordt C. COVID-19, unemployment, and suicide. Lancet Psychiatry. 2020;7(5):389-390.
10. Yao H, Chen JH, Xu YF. Patients with mental health disorders in the COVID-19 epidemic. Lancet Psychiatry. 2020;7(4):e21. doi: 10.1016/S2215-0366(20)30090-0.
11. Minihan E, Gavin B, Kelly BD, et al. Covid-19, mental health and psychological first aid. Ir J Psychol Med. 2020:1-12.
12. Adja KYC, Golinelli D, Lenzi J, et al. Pandemics and social stigma: who’s next? Italy’s experience with COVID-19. Public Health. 2020;185:39-41.
13. Rosenberg AR. Cultivating deliberate resilience during the coronavirus disease 2019 pandemic [published online April 14, 2020]. JAMA Pediatr. doi: 10.1001/jamapediatrics.2020.1436.
14. Dean W, Talbot SG, Caplan A. Clarifying the language of clinician distress [published online January 31, 2020]. JAMA. doi: 10.1001/jama.2019.21576.
15. Emanuel EJ, Persad G, Upshur R, et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055.
16. Rosenbaum L. Facing Covid-19 in Italy - ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020;382(20):1873-1875.
17. Viceconte G, Petrosillo N. COVID-19 R0: magic number or conundrum? Infect Dis Rep. 2020;12(1):8516.
18. de Hert M, Schreurs V, Vancampfort D, van Winkel R. Metabolic syndrome in people with schizophrenia: a review. World Psychiatry. 2009;8(1):15-22.
19. Chen R, Liang W, Jiang M, et al. Risk factors of fatal outcome in hospitalized subjects with coronavirus disease 2019 from a nationwide analysis in China. Chest. 2020;158(1):97-105.
20. Finer N, Garnett SP, Bruun JM. COVID-19 and obesity. Clin Obes. 2020;10(3):e12365. doi: 10.1111/cob.12365.
21. Havens LL, Ghaemi SN. Existential despair and bipolar disorder: the therapeutic alliance as a mood stabilizer. Am J Psychother. 2005;59(2):137-147.
22. Trémeau F, Antonius D, Malaspina D, et al. Loneliness in schizophrenia and its possible correlates. An exploratory study. Psychiatry Res. 2016;246:211-217.
23. Menninger KA. Psychoses associated with influenza: I. General data: statistical analysis. JAMA. 1919;72(4):235-241.
24. Asadi-Pooya AA, Simani L. Central nervous system manifestations of COVID-19: a systematic review. J Neurol Sci. 2020;413:116832. doi: 10.1016/j.jns.2020.116832.
25. Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.012.
26. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun. 2020;87:34-39.
27. Martin Jr. EB. Brief psychotic disorder triggered by fear of coronavirus? Psychiatric Times. https://www.psychiatrictimes.com/view/brief-psychotic-disorder-triggered-fear-coronavirus-small-case-series. Published May 8, 2020. Accessed August 7, 2020.
28. Sher Y, Rabkin B, Maldonado JR, et al. COVID-19-associated hyperactive intensive care unit delirium with proposed pathophysiology and treatment: a case report [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.007.
29. Wolters AE, Peelen LM, Welling MC, et al. Long-term mental health problems after delirium in the ICU. Crit Care Med. 2016;44(10):1808-1813.
30. Toovey S. Influenza-associated central nervous system dysfunction: a literature review. Travel Med Infect Dis. 2008;6(3):114-124.
31. Brooks SK, Webster RK, Smith LE, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. Lancet. 2020;395(10227):912-920.
32. Maercker A, Brewin CR, Bryant RA, et al. Diagnosis and classification of disorders specifically associated with stress: proposals for ICD-11. World Psychiatry. 2013;12(3):198-206.
33. Ornell F, Moura HF, Scherer JN, et al. The COVID-19 pandemic and its impact on substance use: implications for prevention and treatment. Psychiatry Res. 2020;289:113096. doi: 10.1016/j.psychres.2020.113096.
34. Berlin I, Thomas D, Le Faou AL, Cornuz J. COVID-19 and smoking [published online April 3, 2020]. Nicotine Tob Res. https://doi.org/10.1093/ntr/ntaa059.
35. Back D, Marzolini C, Hodge C, et al. COVID-19 treatment in patients with comorbidities: awareness of drug-drug interactions [published online May 8, 2020]. Br J Clin Pharmacol. doi: 10.1111/bcp.14358.
36. Carpenter WT Jr., Buchanan RW, Kirkpatrick B, et al. Diazepam treatment of early signs of exacerbation in schizophrenia. Am J Psychiatry. 1999;156(2):299-303.
37. Dotson S, Hartvigsen N, Wesner T, et al. Clozapine toxicity in the setting of COVID-19 [published online May 30, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.025.
38. Siskind D, Honer WG, Clark S, et al. Consensus statement on the use of clozapine during the COVID-19 pandemic. J Psychiatry Neurosci. 2020;45(3):222-223.
39. Schnitzer K, MacLaurin S, Freudenreich O. Long-acting injectable antipsychotics during the COVID-19 pandemic. Current Psychiatry. In press.
40. Winston A, Rosenthal RN, Pinsker H. Learning supportive psychotherapy: an illustrated guide. Washington, DC: American Psychiatric Publishing; 2012.
41. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
42. Jordan A, Dixon LB. Considerations for telepsychiatry service implementation in the era of COVID-19. Psychiatr Serv. 2020;71(6):643-644.
43. DePierro J, Lowe S, Katz C. Lessons learned from 9/11: mental health perspectives on the COVID-19 pandemic. Psychiatry Res. 2020;288:113024.
44. Hussain S. Immunization and vaccination. In: Huremovic
45. Epping-Jordan JE, van Ommeren M, Ashour HN, et al. Beyond the crisis: building back better mental health care in 10 emergency-affected areas using a longer-term perspective. Int J Ment Health Syst. 2015;9:15.
46. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382(24):2368-2371.
47. Bartels SJ, Baggett TP, Freudenreich O, et al. COVID-19 emergency reforms in Massachusetts to support behavioral health care and reduce mortality of people with serious mental illness [published online June 3, 2020]. Psychiatr Serv. doi: 10.1176/appi.ps.202000244.
1. Camus A. La peste. Paris, France: Éditions Gallimard; 1947.
2. Huremovic
3. Substance Abuse and Mental Health Services Administration. Phases of disaster. https://www.samhsa.gov/dtac/recovering-disasters/phases-disaster. Updated June 17, 2020. Accessed August 7, 2020.
4. Geller J. COVID-19 and advocacy—the good and the unacceptable. Psychiatric News. https://psychnews.psychiatryonline.org/doi/10.1176/appi.pn.2020.5b13. Published May 7, 2020. Accessed August 7, 2020.
5. Webb Hooper M, Nápoles AM, Perez-Stable EJ. COVID-19 and racial/ethnic disparities. JAMA. 2020;323(24):2466-2467.
6. Sederer LI, Lanzara CB, Essock SM, et al. Lessons learned from the New York State mental health response to the September 11, 2001, attacks. Psychiatr Serv. 2011;62(9):1085-1089.
7. World Health Organization. Infodemic management – infodemiology. https://www.who.int/teams/risk-communication/infodemic-management. Accessed August 7, 2020.
8. Zhou J, Liu L, Xue P, et al. Mental health response to the COVID-19 outbreak in China. Am J Psychiatry. 2020;117(7):574-575.
9. Kawohl W, Nordt C. COVID-19, unemployment, and suicide. Lancet Psychiatry. 2020;7(5):389-390.
10. Yao H, Chen JH, Xu YF. Patients with mental health disorders in the COVID-19 epidemic. Lancet Psychiatry. 2020;7(4):e21. doi: 10.1016/S2215-0366(20)30090-0.
11. Minihan E, Gavin B, Kelly BD, et al. Covid-19, mental health and psychological first aid. Ir J Psychol Med. 2020:1-12.
12. Adja KYC, Golinelli D, Lenzi J, et al. Pandemics and social stigma: who’s next? Italy’s experience with COVID-19. Public Health. 2020;185:39-41.
13. Rosenberg AR. Cultivating deliberate resilience during the coronavirus disease 2019 pandemic [published online April 14, 2020]. JAMA Pediatr. doi: 10.1001/jamapediatrics.2020.1436.
14. Dean W, Talbot SG, Caplan A. Clarifying the language of clinician distress [published online January 31, 2020]. JAMA. doi: 10.1001/jama.2019.21576.
15. Emanuel EJ, Persad G, Upshur R, et al. Fair allocation of scarce medical resources in the time of Covid-19. N Engl J Med. 2020;382(21):2049-2055.
16. Rosenbaum L. Facing Covid-19 in Italy - ethics, logistics, and therapeutics on the epidemic’s front line. N Engl J Med. 2020;382(20):1873-1875.
17. Viceconte G, Petrosillo N. COVID-19 R0: magic number or conundrum? Infect Dis Rep. 2020;12(1):8516.
18. de Hert M, Schreurs V, Vancampfort D, van Winkel R. Metabolic syndrome in people with schizophrenia: a review. World Psychiatry. 2009;8(1):15-22.
19. Chen R, Liang W, Jiang M, et al. Risk factors of fatal outcome in hospitalized subjects with coronavirus disease 2019 from a nationwide analysis in China. Chest. 2020;158(1):97-105.
20. Finer N, Garnett SP, Bruun JM. COVID-19 and obesity. Clin Obes. 2020;10(3):e12365. doi: 10.1111/cob.12365.
21. Havens LL, Ghaemi SN. Existential despair and bipolar disorder: the therapeutic alliance as a mood stabilizer. Am J Psychother. 2005;59(2):137-147.
22. Trémeau F, Antonius D, Malaspina D, et al. Loneliness in schizophrenia and its possible correlates. An exploratory study. Psychiatry Res. 2016;246:211-217.
23. Menninger KA. Psychoses associated with influenza: I. General data: statistical analysis. JAMA. 1919;72(4):235-241.
24. Asadi-Pooya AA, Simani L. Central nervous system manifestations of COVID-19: a systematic review. J Neurol Sci. 2020;413:116832. doi: 10.1016/j.jns.2020.116832.
25. Ferrando SJ, Klepacz L, Lynch S, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.012.
26. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun. 2020;87:34-39.
27. Martin Jr. EB. Brief psychotic disorder triggered by fear of coronavirus? Psychiatric Times. https://www.psychiatrictimes.com/view/brief-psychotic-disorder-triggered-fear-coronavirus-small-case-series. Published May 8, 2020. Accessed August 7, 2020.
28. Sher Y, Rabkin B, Maldonado JR, et al. COVID-19-associated hyperactive intensive care unit delirium with proposed pathophysiology and treatment: a case report [published online May 19, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.007.
29. Wolters AE, Peelen LM, Welling MC, et al. Long-term mental health problems after delirium in the ICU. Crit Care Med. 2016;44(10):1808-1813.
30. Toovey S. Influenza-associated central nervous system dysfunction: a literature review. Travel Med Infect Dis. 2008;6(3):114-124.
31. Brooks SK, Webster RK, Smith LE, et al. The psychological impact of quarantine and how to reduce it: rapid review of the evidence. Lancet. 2020;395(10227):912-920.
32. Maercker A, Brewin CR, Bryant RA, et al. Diagnosis and classification of disorders specifically associated with stress: proposals for ICD-11. World Psychiatry. 2013;12(3):198-206.
33. Ornell F, Moura HF, Scherer JN, et al. The COVID-19 pandemic and its impact on substance use: implications for prevention and treatment. Psychiatry Res. 2020;289:113096. doi: 10.1016/j.psychres.2020.113096.
34. Berlin I, Thomas D, Le Faou AL, Cornuz J. COVID-19 and smoking [published online April 3, 2020]. Nicotine Tob Res. https://doi.org/10.1093/ntr/ntaa059.
35. Back D, Marzolini C, Hodge C, et al. COVID-19 treatment in patients with comorbidities: awareness of drug-drug interactions [published online May 8, 2020]. Br J Clin Pharmacol. doi: 10.1111/bcp.14358.
36. Carpenter WT Jr., Buchanan RW, Kirkpatrick B, et al. Diazepam treatment of early signs of exacerbation in schizophrenia. Am J Psychiatry. 1999;156(2):299-303.
37. Dotson S, Hartvigsen N, Wesner T, et al. Clozapine toxicity in the setting of COVID-19 [published online May 30, 2020]. Psychosomatics. doi: 10.1016/j.psym.2020.05.025.
38. Siskind D, Honer WG, Clark S, et al. Consensus statement on the use of clozapine during the COVID-19 pandemic. J Psychiatry Neurosci. 2020;45(3):222-223.
39. Schnitzer K, MacLaurin S, Freudenreich O. Long-acting injectable antipsychotics during the COVID-19 pandemic. Current Psychiatry. In press.
40. Winston A, Rosenthal RN, Pinsker H. Learning supportive psychotherapy: an illustrated guide. Washington, DC: American Psychiatric Publishing; 2012.
41. Hollander JE, Carr BG. Virtually perfect? Telemedicine for Covid-19. N Engl J Med. 2020;382(18):1679-1681.
42. Jordan A, Dixon LB. Considerations for telepsychiatry service implementation in the era of COVID-19. Psychiatr Serv. 2020;71(6):643-644.
43. DePierro J, Lowe S, Katz C. Lessons learned from 9/11: mental health perspectives on the COVID-19 pandemic. Psychiatry Res. 2020;288:113024.
44. Hussain S. Immunization and vaccination. In: Huremovic
45. Epping-Jordan JE, van Ommeren M, Ashour HN, et al. Beyond the crisis: building back better mental health care in 10 emergency-affected areas using a longer-term perspective. Int J Ment Health Syst. 2015;9:15.
46. Rosenbaum L. The untold toll - the pandemic’s effects on patients without Covid-19. N Engl J Med. 2020;382(24):2368-2371.
47. Bartels SJ, Baggett TP, Freudenreich O, et al. COVID-19 emergency reforms in Massachusetts to support behavioral health care and reduce mortality of people with serious mental illness [published online June 3, 2020]. Psychiatr Serv. doi: 10.1176/appi.ps.202000244.
Coronavirus vaccine: The contenders, the potential controversy
References
- CDC. Coronavirus Disease 2019 (COVID-19): Cases in the US. www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html. Accessed August 18, 2020.
- US Department of Health and Human Services. Fact Sheet: explaining Operation Warp Speed. www.hhs.gov/coronavirus/explaining-operation-warp-speed/index.html. Accessed August 18, 2020.
- O’Callahan KP, Blatz AM, Offit PA. Developing a SARS-CoV-2 vaccine at warp speed. JAMA. 2020;324:437-438.
- Pardi N, Hogan MJ, Porter FW, et al. mRNA vaccines—a new era in vaccinology. Nat Rev Drug Discov. 2018;17:261-279.
- Lurie N, Sharfstein JM, Goodman JL. The development of COVID-19 vaccines: safeguards needed [commentary]. JAMA. 2020;324:439-440.
- Salman DA, Akhtar A, Mergler MJ, et al; H1N1 Working Group of Federal Immunization Safety Task Force. Immunization safety monitoring systems for the 2009 H1N1 monovalent influenza vaccination program. Pediatrics. 2011;127(suppl 1):S78-S86.
References
- CDC. Coronavirus Disease 2019 (COVID-19): Cases in the US. www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html. Accessed August 18, 2020.
- US Department of Health and Human Services. Fact Sheet: explaining Operation Warp Speed. www.hhs.gov/coronavirus/explaining-operation-warp-speed/index.html. Accessed August 18, 2020.
- O’Callahan KP, Blatz AM, Offit PA. Developing a SARS-CoV-2 vaccine at warp speed. JAMA. 2020;324:437-438.
- Pardi N, Hogan MJ, Porter FW, et al. mRNA vaccines—a new era in vaccinology. Nat Rev Drug Discov. 2018;17:261-279.
- Lurie N, Sharfstein JM, Goodman JL. The development of COVID-19 vaccines: safeguards needed [commentary]. JAMA. 2020;324:439-440.
- Salman DA, Akhtar A, Mergler MJ, et al; H1N1 Working Group of Federal Immunization Safety Task Force. Immunization safety monitoring systems for the 2009 H1N1 monovalent influenza vaccination program. Pediatrics. 2011;127(suppl 1):S78-S86.
References
- CDC. Coronavirus Disease 2019 (COVID-19): Cases in the US. www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html. Accessed August 18, 2020.
- US Department of Health and Human Services. Fact Sheet: explaining Operation Warp Speed. www.hhs.gov/coronavirus/explaining-operation-warp-speed/index.html. Accessed August 18, 2020.
- O’Callahan KP, Blatz AM, Offit PA. Developing a SARS-CoV-2 vaccine at warp speed. JAMA. 2020;324:437-438.
- Pardi N, Hogan MJ, Porter FW, et al. mRNA vaccines—a new era in vaccinology. Nat Rev Drug Discov. 2018;17:261-279.
- Lurie N, Sharfstein JM, Goodman JL. The development of COVID-19 vaccines: safeguards needed [commentary]. JAMA. 2020;324:439-440.
- Salman DA, Akhtar A, Mergler MJ, et al; H1N1 Working Group of Federal Immunization Safety Task Force. Immunization safety monitoring systems for the 2009 H1N1 monovalent influenza vaccination program. Pediatrics. 2011;127(suppl 1):S78-S86.
FDA expands remdesivir use for all COVID-19 hospitalized patients
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
VTE, sepsis risk increased among COVID-19 patients with cancer
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
, according to data from a registry study.
Researchers analyzed data on 5,556 patients with COVID-19 who had an inpatient or emergency encounter at Mount Sinai Health System (MSHS) in New York between March 1 and May 27, 2020. Patients were included in an anonymous MSHS COVID-19 registry.
There were 421 patients who had cancer: 96 with a hematologic malignancy and 325 with solid tumors.
After adjustment for age, gender, and number of comorbidities, the odds ratios for acute VTE and sepsis for patients with cancer (versus those without cancer) were 1.77 and 1.34, respectively. The adjusted odds ratio for mortality in cancer patients was 1.02.
The results remained “relatively consistent” after stratification by solid and nonsolid cancer types, with no significant difference in outcomes between those two groups, and results remained consistent in a propensity-matched model, according to Naomi Alpert, a biostatistician at Icahn School of Medicine at Mount Sinai, New York.
Ms. Alpert reported these findings at the AACR virtual meeting: COVID-19 and Cancer.
She noted that the cancer patients were older than the noncancer patients (mean age, 69.2 years vs. 63.8 years), and cancer patients were more likely to have two or more comorbid conditions (48.2% vs. 30.4%). Cancer patients also had significantly lower hemoglobin levels and red blood cell, platelet, and white blood cell counts (P < .01 for all).
“Low white blood cell count may be one of the reasons for higher risk of sepsis in cancer patients, as it may lead to a higher risk of infection,” Ms. Alpert said. “However, it’s not clear what role cancer therapies play in the risks of COVID-19 morbidity and mortality, so there is still quite a bit to learn.”
In fact, the findings are limited by a lack of information about cancer treatment, as the registry was not designed for that purpose, she noted.
Another study limitation is the short follow-up of a month or less in most patients, due, in part, to the novelty of COVID-19, but also to the lack of information on patients after they left the hospital.
“However, we had a very large sample size, with more than 400 cancer patients included, and, to our knowledge, this is the largest analysis of its kind to be done so far,” Ms. Alpert said. “In the future, it’s going to be very important to assess the effect of cancer therapies on COVID-19 complications and to see if prior therapies had any effect on outcomes.”
Longer follow-up would also be helpful for assessing the chronic effects of COVID-19 on cancer patients over time, she said. “It would be important to see whether some of these elevated risks of venous thromboembolism and sepsis are associated with longer-term mortality risks than what we were able to measure here,” she added.
Asked about the discrepancy between mortality in this study and those of larger registries, such as the COVID-19 and Cancer Consortium (CCC19) and TERAVOLT, Ms. Alpert noted that the current study included only patients who required hospitalization or emergency care.
“Our mortality rate was actually a bit higher than what was reported in some of the other studies,” she said. “We had about a 30% mortality rate in the cancer patients and about 25% for the noncancer patients, so ... we’re sort of looking at a subset of patients who we know are the sickest of the sick, which may explain some of the higher mortality that we’re seeing.”
Ms. Alpert reported having no disclosures.
SOURCE: Alpert N et al. AACR COVID-19 and Cancer, Abstract S12-02.
FROM AACR: COVID-19 AND CANCER