User login
Add AFib to noncardiac surgery risk evaluation: New support
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practice has gone back and forth on whether atrial fibrillation (AFib) should be considered in the preoperative cardiovascular risk (CV) evaluation of patients slated for noncardiac surgery, and the Revised Cardiac Risk Index (RCRI), currently widely used as an assessment tool, doesn’t include the arrhythmia.
But consideration of preexisting AFib along with the RCRI predicted 30-day mortality more sharply than the RCRI alone in an analysis of data covering several million patients slated for such procedures.
Indeed, AFib emerged as a significant, independent risk factor for a number of bad postoperative outcomes. Mortality within a month of the procedure climbed about 30% for patients with AFib before the noncardiac surgery. Their 30-day risks for stroke and for heart failure hospitalization went up similarly.
The addition of AFib to the RCRI significantly improved its ability to discriminate 30-day postoperative risk levels regardless of age, sex, and type of noncardiac surgery, Amgad Mentias, MD, Cleveland Clinic, told this news organization. And “it was able to correctly up-classify patients to high risk, if AFib was there, and it was able to down-classify some patients to lower risk if it wasn’t there.”
“I think [the findings] are convincing evidence that atrial fib should at least be part of the thought process for the surgical team and the medical team taking care of the patient,” said Dr. Mentias, who is senior author on the study published in the Journal of the American College of Cardiology, with lead author Sameer Prasada, MD, also of the Cleveland Clinic.
The results “call for incorporating AFib as a risk factor in perioperative risk scores for cardiovascular morbidity and mortality,” the published report states.
Supraventricular arrhythmias had been part of the Goldman Risk Index once widely used preoperatively to assess cardiac risk before practice adopted the RCRI in the past decade, observe Anne B. Curtis, MD, and Sai Krishna C. Korada, MD, University at Buffalo, New York, in an accompanying editorial.
The current findings “demonstrate improved prediction of adverse postsurgical outcomes” from supplementing the RCRI with AFib, they write. Given associations between preexisting AFib and serious cardiac events, “it is time to ‘re-revise’ the RCRI and acknowledge the importance of AFib in predicting adverse outcomes” after noncardiac surgery.
The new findings, however, aren’t all straightforward. In one result that remains a bit of a head-scratcher, postoperative risk of myocardial infarction (MI) in patients with preexisting AFib went in the opposite direction of risk for death and other CV outcomes, falling by almost 20%.
That is “hard to explain with the available data,” the report states, but “the use of anticoagulation, whether oral or parenteral (as a bridge therapy in the perioperative period), is a plausible explanation” given the frequent role of thrombosis in triggering MIs.
Consistent with such a mechanism, the group argues, the MI risk reduction was seen primarily among patients with AFib and a CHA2DS2-VASc score of 2 or higher – that is, those at highest risk for stroke and therefore most likely to be on oral anticoagulation. The MI risk reduction wasn’t seen in such patients with a CHA2DS2-VASc score of 0 or 1.
“I think that’s part of the explanation, that anticoagulation can reduce risk of MI. But it’s not the whole explanation,” Dr. Mentias said in an interview. If it were the sole mechanism, he said, then the same oral anticoagulation that protected against MI should have also cut the postoperative stroke risk. Yet that risk climbed 40% among patients with preexisting AFib.
The analysis started with 8.6 million Medicare patients with planned noncardiac surgery, seen from 2015 to 2019, of whom 16.4% had preexisting AFib. Propensity matching for demographics, urgency and type of surgery, CHA2DS2-VASc score, and RCRI index created two cohorts for comparison: 1.13 million patients with and 1.92 million without preexisting AFib.
Preexisting AFib was associated with a higher 30-day risk for death from any cause, the primary endpoint being 8.3% versus 5.8% for those without such AFib (P < .001), for an odds ratio of 1.31 (95% confidence interval, 1.30-1.32).
Corresponding 30-day ORs for other events, all significant at P < .001, were:
- 1.31 (95% CI, 1.30-1.33) for heart failure
- 1.40 (95% CI, 1.37-1.43) for stroke
- 1.59 (95% CI, 1.43-1.75) for systemic embolism
- 1.14 (95% CI, 1.13-1.16) for major bleeding
- 0.81 (95% CI, 0.79-0.82) for MI
Those with preexisting AFib also had longer hospitalizations at a median 5 days, compared with 4 days for those without such AFib (P < .001).
The study has the limitations of most any retrospective cohort analysis. Other limitations, the report notes, include lack of information on any antiarrhythmic meds given during hospitalization or type of AFib.
For example, AFib that is permanent – compared with paroxysmal or persistent – may be associated with more atrial fibrosis, greater atrial dilatation, “and probably higher pressures inside the heart,” Dr. Mentias observed.
“That’s not always the case, but that’s the notion. So presumably people with persistent or permanent atrial fib would have more advanced heart disease, and that could imply more risk. But we did not have that kind of data.”
Dr. Mentias and Dr. Prasada report no relevant financial relationships; disclosures for the other authors are in the report. Dr. Curtis discloses serving on advisory boards for Abbott, Janssen Pharmaceuticals, Sanofi, and Milestone Pharmaceuticals; receiving honoraria for speaking from Medtronic and Zoll; and serving on a data-monitoring board for Medtronic. Dr. Korada reports he has no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Updated AHA/ASA guideline changes care for spontaneous intracerebral hemorrhage
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
Many strategies widely considered “standard care” for managing spontaneous intracerebral hemorrhage (ICH) are not as effective as previously thought and are no longer recommended in updated guidelines from the American Heart Association/American Stroke Association (ASA).
Compression stockings, antiseizure medication, and steroid treatment are among the treatments with uncertain effectiveness, the writing group says.
The 2022 Guideline for the Management of Patients With Spontaneous ICH was published online in Stroke. The 80-page document contains major changes and refinements to the 2015 guideline on ICH management.
“Advances have been made in an array of fields related to ICH, including the organization of regional health care systems, reversal of the negative effects of blood thinners, minimally invasive surgical procedures, and the underlying disease in small blood vessels,” Steven M. Greenberg, MD, PhD, chair of the guideline writing group with Harvard Medical School and Massachusetts General Hospital, both in Boston, said in a news release.
“We’ve updated sections across the board. There’s probably no area that went untouched with some tweaking and new evidence added that led to some changes in level of evidence or strength of a recommendation,” Dr. Greenberg added in an interview with this news organization.
“Each section comes with knowledge gaps, and it wasn’t hard to come up with knowledge gaps in every section,” Dr. Greenberg acknowledged.
Time-honored treatments no more?
Among the key updates are changes to some “time-honored” treatments that continue to be used with some “regularity” for patients with ICH, yet appear to confer either no benefit or harm, Dr. Greenberg said.
For example, for emergency or critical care treatment of ICH, prophylactic corticosteroids or continuous hyperosmolar therapy is not recommended, because it appears to have no benefit for outcome, while use of platelet transfusions outside the setting of emergency surgery or severe thrombocytopenia appears to worsen outcome, the authors say.
Use of graduated knee- or thigh-high compression stockings alone is not an effective prophylactic therapy for prevention of deep vein thrombosis (DVT). Instead, intermittent pneumatic compression (IPC) starting on the day of diagnosis is now recommended for DVT prophylaxis.
“This is an area where we still have a lot of exploration to do. It is unclear whether even specialized compression devices reduce the risks of deep vein thrombosis or improve the overall health of people with a brain bleed,” Dr. Greenberg said in the release.
The new guidance advises against use of antiseizure or antidepressant medications for ICH patients in whom there is no evidence of seizures or depression.
In clinical trials, antiseizure medication did not contribute to improvements in functionality or long-term seizure control, and the use of antidepressants increased the chance of bone fractures, the authors say.
The guideline also provides updated recommendations for acute reversal of anticoagulation after ICH. It highlights the use of protein complex concentrate for reversal of vitamin K antagonists, such as warfarin; idarucizumab for reversal of the thrombin inhibitor dabigatran; and andexanet alfa for reversal of factor Xa inhibitors, such as rivaroxaban, apixaban, and edoxaban.
For acute blood pressure lowering after mild to moderate ICH, treatment regimens that limit blood pressure variability and achieve smooth, sustained blood pressure control appear to reduce hematoma expansion and yield better functional outcome, the guideline says.
It also notes that minimally invasive approaches for hematoma evacuation, compared with medical management alone‚ have been shown to reduce mortality.
For patients with cerebellar hemorrhage, indications for immediate surgical evacuation with or without an external ventricular drain to reduce mortality now include larger volume (> 15 mL) in addition to previously recommended indications of neurologic deterioration, brainstem compression, and hydrocephalus, the authors note.
However, a “major knowledge gap is whether we can improve functional outcome with hematoma evacuation,” Dr. Greenberg said.
Multidisciplinary care
For rehabilitation after ICH, the guideline reinforces the importance of having a multidisciplinary team to develop a comprehensive plan for recovery.
Starting rehabilitation activities such as stretching and functional task training may be considered 24 to 48 hours following mild or moderate ICH. However, early aggressive mobilization within the first 24 hours has been linked to an increased risk of death within 14 days after an ICH, the guideline says.
Knowledge gaps include how soon it’s safe to return to work, drive, and participate in other social engagements. Recommendations on sexual activity and exercise levels that are safe after a stroke are also needed.
“People need additional help with these lifestyle changes, whether it’s moving around more, curbing their alcohol use, or eating healthier foods. This all happens after they leave the hospital, and we need to be sure we are empowering families with the information they may need to be properly supportive,” Dr. Greenberg says in the release.
The guideline points to the patient’s home caregiver as a “key and sometimes overlooked” member of the care team. It recommends psychosocial education, practical support, and training for the caregiver to improve the patient’s balance, activity level, and overall quality of life.
Opportunity for prevention?
The guideline also suggests there may be an opportunity to prevent ICH in some people through neuroimaging markers.
While neuroimaging is not routinely performed as a part of risk stratification for primary ICH risk, damage to small blood vessels that is associated with ICH may be evident on MRI that could signal future ICH risk, the guideline says.
“We added to the guidelines for the first time a section on mostly imaging markers of risk for having a first-ever hemorrhage,” Dr. Greenberg said in an interview.
“We don’t make any recommendations as to how to act on these markers because there is a knowledge gap. The hope is that we’ll see growth in our ability to predict first-ever hemorrhage and be able to do things to prevent first-ever hemorrhage,” he said.
“We believe the wide range of knowledge set forth in the new guideline will translate into meaningful improvements in ICH care,” Dr. Greenberg adds in the release.
The updated guideline has been endorsed by the American Association of Neurological Surgeons and Congress of Neurological Surgeons, the Society of Vascular and Interventional Neurology, and the Neurocritical Care Society. The American Academy of Neurology has affirmed the value of this statement as an educational tool for neurologists.
This research had no commercial funding. Dr. Greenberg has disclosed no relevant financial relationships. A complete list of disclosures for the guideline group is available with the original article.
A version of this article first appeared on Medscape.com.
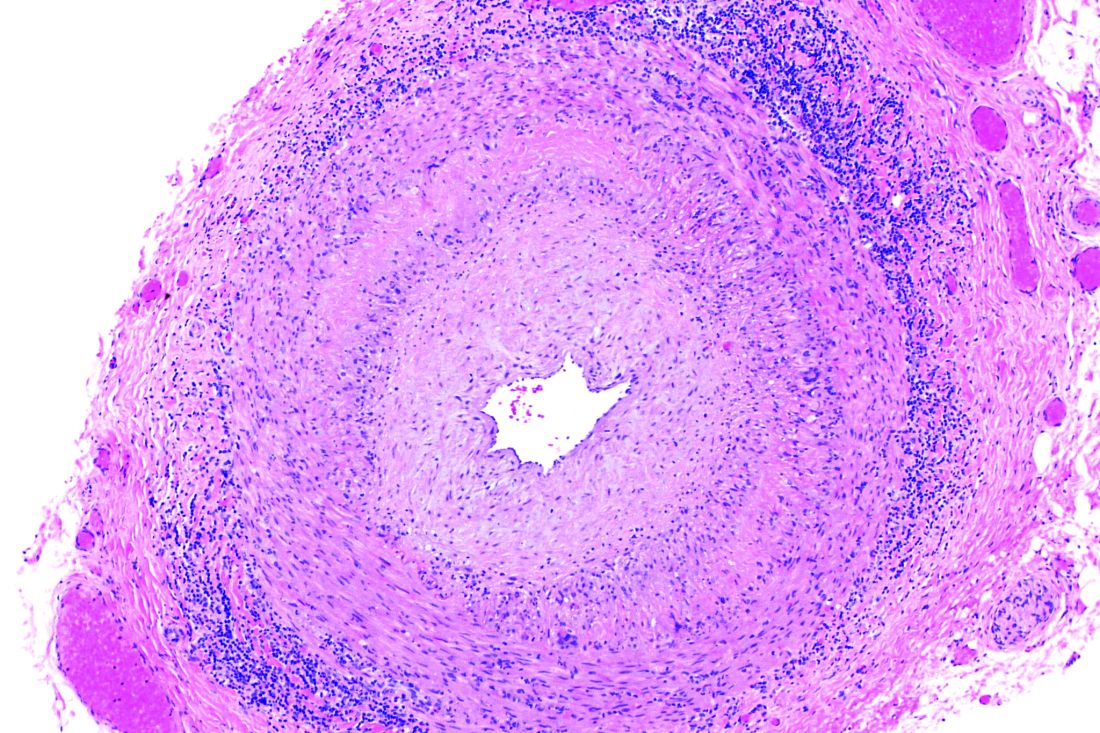
ACST-2: Carotid stenting, surgery on par in asymptomatic patients
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Carotid artery stenting (CAS) and carotid endarterectomy (CEA) provided comparable outcomes over time in asymptomatic patients receiving good medical therapy in the largest trial to date of what to do with severe carotid artery narrowing that is yet to cause a stroke.
Among more than 3,600 patients, stenting and surgery performed by experienced physicians involved a 1.0% risk for causing disabling stroke or death within 30 days.
The annual rate of fatal or disabling strokes was about 0.5% with either procedure over an average 5 years’ follow-up – essentially halving the annual stroke risk had neither procedure been performed, according to Alison Halliday, MD, principal investigator of the Asymptomatic Carotid Surgery Trial-2 (ACST-2).
The results were reported Aug. 29 in a Hot Line session at the virtual annual congress of the European Society of Cardiology and published simultaneously online in The Lancet.
Session chair Gilles Montalescot, MD, Sorbonne University, Paris, noted that ACST-2 doubled the number of randomly assigned patients with asymptomatic carotid stenosis studied in previous trials, “so, a huge contribution to the evidence base in this field and apparently good news for both revascularization techniques.”
Thirty-day and 5-year outcomes
The trial was conducted in 33 countries between January 2008 and December 2020, enrolling 3,625 patients (70% were male; mean age, 70 years) with carotid stenosis of at least 60% on ultrasonography, in whom stenting or surgery was suitable but both the doctor and patient were “substantially uncertain” which procedure to prefer.
Among the 1,811 patients assigned to stenting, 87% underwent the procedure at a median of 14 days; 6% crossed over to surgery, typically because of a highly calcified lesion or a more tortuous carotid than anticipated; and 6% had no intervention.
Among the 1,814 patients assigned to surgery, 92% had the procedure at a median of 14 days; 3% crossed over to stenting, typically because of patient or doctor preference or reluctance to undergo general anesthesia; and 4% had no intervention.
Patients without complications who had stenting stayed on average 1 day less than did those undergoing surgery.
During an earlier press briefing, Dr. Halliday highlighted the need for procedural competency and said doctors had to submit a record of their CEA or CAS experience and, consistent with current guidelines, had to demonstrate an independently verified stroke or death rate of 6% or less for symptomatic patients and 3% or lower for asymptomatic patients.
The results showed the 30-day risk for death, myocardial infarction (MI), or any stroke was 3.9% with carotid stenting and 3.2% with surgery (P = .26).
But with stenting, there was a slightly higher risk for procedural nondisabling strokes (48 vs. 29; P = .03), including 15 strokes vs. 5 strokes, respectively, that left patients with no residual symptoms. This is “consistent with large, recent nationally representative registry data,” observed Dr. Halliday, of the University of Oxford (England).
For those undergoing surgery, cranial nerve palsies were reported in 5.4% vs. no patients undergoing stenting.
At 5 years, the nonprocedural fatal or disabling stroke rate was 2.5% in each group (rate ratio [RR], 0.98; P = .91), with any nonprocedural stroke occurring in 5.3% of patients with stenting vs. 4.5% with surgery (RR, 1.16; P = .33).
The investigators performed a meta-analysis combining the ACST-2 results with those of eight prior trials (four in asymptomatic and four in symptomatic patients) that yielded a similar nonsignificant result for any nonprocedural stroke (RR, 1.11; P = .21).
Based on the results from ACST-2 plus the major trials, stenting and surgery involve “similar risks and similar benefits,” Dr. Halliday concluded.
Discussant Marco Roffi, MD, University Hospital of Geneva, said, “In centers with documented expertise, carotid artery stenting should be offered as an alternative to carotid endarterectomy in patients with asymptomatic stenosis and suitable anatomy.”
While the trial provides “good news” for patients, he pointed out that a reduction in the sample size from 5,000 to 3,625 limited the statistical power and that enrollment over a long period of time may have introduced confounders, such as changes in equipment technique, and medical therapy.
Also, many centers enrolled few patients, raising the concern over low-volume centers and operators, Dr. Roffi said. “We know that 8% of the centers enrolled 39% of the patients,” and “information on the credentialing and experience of the interventionalists was limited.”
Further, a lack of systematic MI assessment may have favored the surgery group, and more recent developments in stenting with the potential of reducing periprocedural stroke were rarely used, such as proximal emboli protection in only 15% and double-layer stents in 11%.
Friedhelm Beyersdorf, MD, University Hospital of Freiburg, Germany, said that, as a vascular surgeon, he finds it understandable that there might be a higher incidence of nonfatal strokes when treating carotid stenosis with stents, given the vulnerability of these lesions.
“Nevertheless, the main conclusion from the entire study is that carotid artery treatment is extremely safe, it has to be done in order to avoid strokes, and, obviously, there seems to be an advantage for surgery in terms of nondisabling stroke,” he said.
Session chair Dr. Montalescot, however, said that what the study cannot address – and what was the subject of many online audience comments – is whether either intervention should be performed in these patients.
Unlike earlier trials comparing interventions to medical therapy, Dr. Halliday said ACST-2 enrolled patients for whom the decision had been made that revascularization was needed. In addition, 99%-100% were receiving antithrombotic therapy at baseline, 85%-90% were receiving antihypertensives, and about 85% were taking statins.
Longer-term follow-up should provide a better picture of the nonprocedural stroke risk, with patients asked annually about exactly what medications and doses they are taking, she said.
“We will have an enormous list of exactly what’s gone on and the intensity of that therapy, which is, of course, much more intense than when we carried out our first trial. But these were people in whom a procedure was thought to be necessary,” she noted.
When asked during the press conference which procedure she would choose, Dr. Halliday, a surgeon, observed that patient preference is important but that the nature of the lesion itself often determines the optimal choice.
“If you know the competence of the people doing it is equal, then the less invasive procedure – providing it has good long-term viability, and that’s why we’re following for 10 years – is the more important,” she added.
The study was funded by the UK Medical Research Council and Health Technology Assessment Programme. Dr. Halliday reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
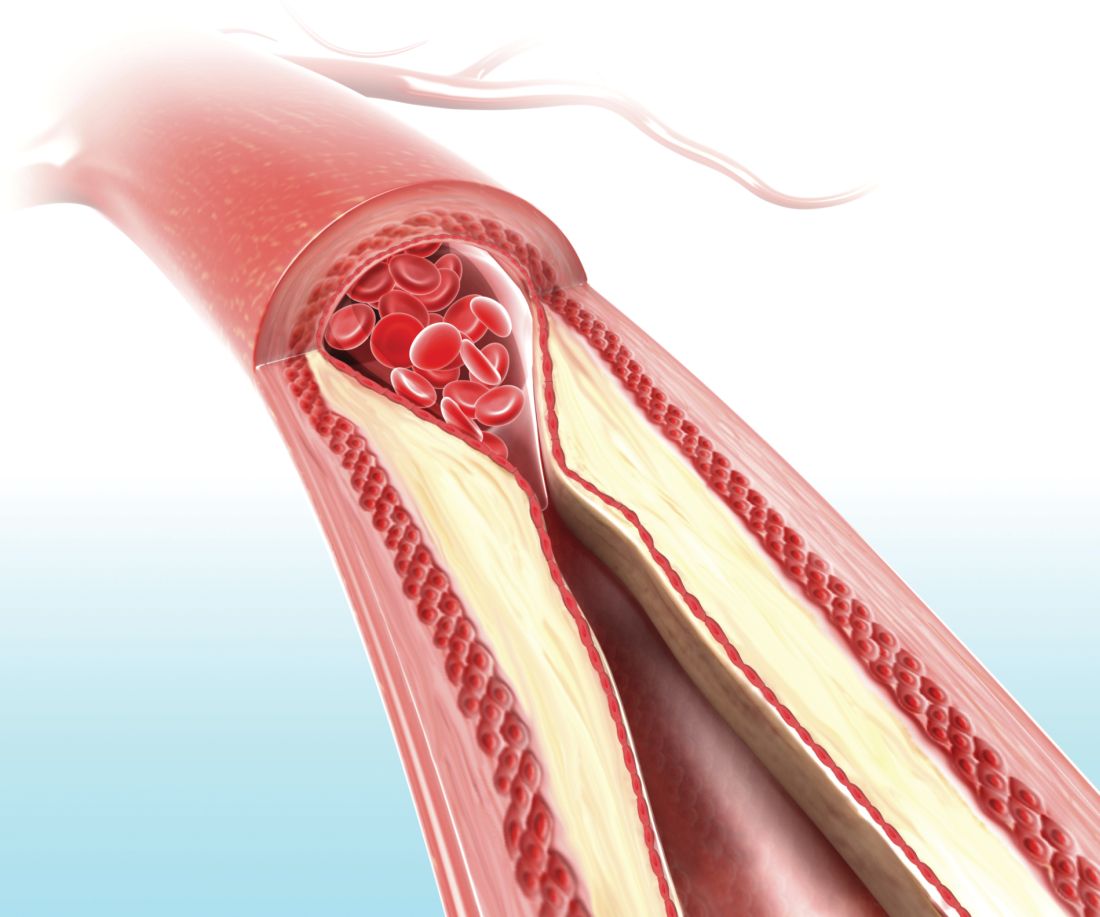
FDA panel supports Vascepa expanded indication for CVD reduction
Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid, received unanimous backing from a Food and Drug Administration advisory panel for a new indication for reducing cardiovascular event risk.
Icosapent ethyl (Vascepa) received initial agency approval in 2012 for the indication of cutting triglyceride levels once they reached at least 500 mg/dL.
The target patient population for this new, cardiovascular-event protection role will reflect some or all of the types of patients enrolled in REDUCE-IT (Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial), which tested icosapent ethyl in 8,179 patients with either established cardiovascular disease or diabetes and at least one additional cardiovascular disease risk factor. This study provided the bulk of the data considered by the FDA panel.
REDUCE-IT showed that, during a median of 4.9 years, patients who received icosapent ethyl had a statistically significant 25% relative risk reduction in the trial’s primary, composite endpoint (New Engl J Med. 2019 Jan 3;380[1]:11-22).
Icosapent ethyl “appeared effective and safe,” and would be a “useful, new, added agent for treating patients” like those enrolled in the trial, said Kenneth D. Burman, MD, professor and chief of endocrinology at Medstar Washington (D.C.) Hospital Center and chair of the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee.
The advisory panel members appeared uniformly comfortable with recommending that the FDA add a cardiovascular disease indication based on the REDUCE-IT findings.
But while they agreed that icosapent ethyl should receive some type of indication for cardiovascular event reduction, the committee split over which patients the indication should include. Specifically, they diverged on the issue of primary prevention.
Some said that the patient enrollment that produced a positive result in REDUCE-IT should not be retrospectively subdivided, while others said that combining secondary- and primary-prevention patients in a single large trial inappropriately lumped together patients who would be better considered separately.
Committee members also expressed uncertainty over the appropriate triglyceride level to warrant treatment. The REDUCE-IT trial was designed to enroll patients with triglycerides of 135 mg/dL or greater, but several panel members suggested that, for labeling, the threshold should be at least 150 mg/dL, or even 200 mg/dL.
Safety was another aspect that generated a lot of panel discussion throughout their day-long discussion, with particular focus on a signal of a small but concerning increased rate of incident atrial fibrillation among patients who received icosapent ethyl, as well as a small but nearly significant increase in the rate of serious bleeds.
Further analyses presented during the meeting showed that an increased bleeding rate linked with icosapent ethyl was focused in patients who concurrently received one or more antiplatelet drugs or an anticoagulant.
However, panel members rejected the notion that these safety concerns warranted a boxed warning, agreeing that it could be managed with appropriate labeling information.
Clinician reaction
Clinicians who manage these types of patients viewed the prospect of an expanded indication for icosapent ethyl as an important advance.
The REDUCE-IT results by themselves “were convincing” for patients with established cardiovascular disease without need for a confirmatory trial, Robert H. Eckel, MD, an endocrinologist and professor of medicine at the University of Colorado at Denver, Aurora, said in an interview. But he remained unconvinced about efficacy for primary-prevention patients, or even for secondary-prevention patients with a triglyceride level below 150 mg/dL.
“Icosapent ethyl will clearly be a mainstay for managing high-risk patients. It gives us another treatment option,” Yehuda Handelsman, MD, an endocrinologist and medical director and principal investigator of the Metabolic Institute of America in Tarzana, Calif., said in an interview. “I do not see the atrial fibrillation or bleeding effects as reasons not to approve this drug. It should be a precaution. Overall, icosapent ethyl is one of the easier drugs for patients to take.”
Dr. Handelsman said it would be unethical to run a confirmatory trial and randomize patients to placebo. “Another trial makes no sense,” he said.
But the data from REDUCE-IT were “not as convincing” for primary-prevention patients, suggesting a need for caution about using icosapent ethyl for patients without established cardiovascular disease, Paul S. Jellinger, MD, an endocrinologist in Fort Lauderdale, Fla., said in an interview.
Cost-effectiveness
An analysis of the cost-effectiveness of icosapent ethyl as used in REDUCE-IT showed that the drug fell into the rare category of being a “dominant” treatment, meaning that it both improved patient outcomes and reduced medical costs. William S. Weintraub, MD, will report findings from this analysis on Nov. 16, 2019, at the annual scientific sessions of the American Heart Association.
The analysis used a wholesale acquisition cost for a 1-day dosage of icosapent ethyl of $4.16, derived from a commercial source for prescription-drug pricing and actual hospitalization costs for the patients in the trial.
Based on the REDUCE-IT outcomes, treatment with icosapent ethyl was linked with a boost in quality-adjusted life-years that extrapolated to an average 0.26 increase during the full lifetime of REDUCE-IT participants, at a cost that averaged $1,284 less per treated patient over their lifetime, according to Dr. Weintraub, director of Outcomes Research at Medstar Washington Hospital Center, Washington.
Although the 0.26 lifetime increase in quality-adjusted life-years may sound modest, “in the cost-effectiveness world, 0.26 is actually significant,” Dr. Weintraub said. He also highlighted how unusual it is to find a patented drug that improves quality of life and longevity while also saving money.
“I know of no other on-patent, branded pharmaceutical that is dominant,” he said.
Off-patent pharmaceuticals, like statins, can be quite inexpensive and may also be dominant, he noted. Being dominant for cost-effectiveness means that icosapent ethyl “provides good value and is worth what we pay for it, well within social thresholds of willingness to pay,” Dr. Weintraub said.
REDUCE-IT was sponsored by Amarin, the company that markets icosapent ethyl (Vascepa). Dr. Burman has received research funding from AstraZeneca, Eisai, and IBSA. Dr. Eckel has received personal fees from Kowa Pharmaceuticals, Merck, Novartis, and Sanofi/Regeneron, as well as research funding from Endece, Ionis Pharmaceuticals, and UniQure. Dr. Handelsman has been a consultant to and received research funding from Amarin and several other companies. Dr. Jellinger has been a speaker on behalf of Amarin, Amgen, and Regeneron. Dr. Weintraub has received honoraria and research support from Amarin, and honoraria from AstraZeneca.
Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid, received unanimous backing from a Food and Drug Administration advisory panel for a new indication for reducing cardiovascular event risk.
Icosapent ethyl (Vascepa) received initial agency approval in 2012 for the indication of cutting triglyceride levels once they reached at least 500 mg/dL.
The target patient population for this new, cardiovascular-event protection role will reflect some or all of the types of patients enrolled in REDUCE-IT (Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial), which tested icosapent ethyl in 8,179 patients with either established cardiovascular disease or diabetes and at least one additional cardiovascular disease risk factor. This study provided the bulk of the data considered by the FDA panel.
REDUCE-IT showed that, during a median of 4.9 years, patients who received icosapent ethyl had a statistically significant 25% relative risk reduction in the trial’s primary, composite endpoint (New Engl J Med. 2019 Jan 3;380[1]:11-22).
Icosapent ethyl “appeared effective and safe,” and would be a “useful, new, added agent for treating patients” like those enrolled in the trial, said Kenneth D. Burman, MD, professor and chief of endocrinology at Medstar Washington (D.C.) Hospital Center and chair of the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee.
The advisory panel members appeared uniformly comfortable with recommending that the FDA add a cardiovascular disease indication based on the REDUCE-IT findings.
But while they agreed that icosapent ethyl should receive some type of indication for cardiovascular event reduction, the committee split over which patients the indication should include. Specifically, they diverged on the issue of primary prevention.
Some said that the patient enrollment that produced a positive result in REDUCE-IT should not be retrospectively subdivided, while others said that combining secondary- and primary-prevention patients in a single large trial inappropriately lumped together patients who would be better considered separately.
Committee members also expressed uncertainty over the appropriate triglyceride level to warrant treatment. The REDUCE-IT trial was designed to enroll patients with triglycerides of 135 mg/dL or greater, but several panel members suggested that, for labeling, the threshold should be at least 150 mg/dL, or even 200 mg/dL.
Safety was another aspect that generated a lot of panel discussion throughout their day-long discussion, with particular focus on a signal of a small but concerning increased rate of incident atrial fibrillation among patients who received icosapent ethyl, as well as a small but nearly significant increase in the rate of serious bleeds.
Further analyses presented during the meeting showed that an increased bleeding rate linked with icosapent ethyl was focused in patients who concurrently received one or more antiplatelet drugs or an anticoagulant.
However, panel members rejected the notion that these safety concerns warranted a boxed warning, agreeing that it could be managed with appropriate labeling information.
Clinician reaction
Clinicians who manage these types of patients viewed the prospect of an expanded indication for icosapent ethyl as an important advance.
The REDUCE-IT results by themselves “were convincing” for patients with established cardiovascular disease without need for a confirmatory trial, Robert H. Eckel, MD, an endocrinologist and professor of medicine at the University of Colorado at Denver, Aurora, said in an interview. But he remained unconvinced about efficacy for primary-prevention patients, or even for secondary-prevention patients with a triglyceride level below 150 mg/dL.
“Icosapent ethyl will clearly be a mainstay for managing high-risk patients. It gives us another treatment option,” Yehuda Handelsman, MD, an endocrinologist and medical director and principal investigator of the Metabolic Institute of America in Tarzana, Calif., said in an interview. “I do not see the atrial fibrillation or bleeding effects as reasons not to approve this drug. It should be a precaution. Overall, icosapent ethyl is one of the easier drugs for patients to take.”
Dr. Handelsman said it would be unethical to run a confirmatory trial and randomize patients to placebo. “Another trial makes no sense,” he said.
But the data from REDUCE-IT were “not as convincing” for primary-prevention patients, suggesting a need for caution about using icosapent ethyl for patients without established cardiovascular disease, Paul S. Jellinger, MD, an endocrinologist in Fort Lauderdale, Fla., said in an interview.
Cost-effectiveness
An analysis of the cost-effectiveness of icosapent ethyl as used in REDUCE-IT showed that the drug fell into the rare category of being a “dominant” treatment, meaning that it both improved patient outcomes and reduced medical costs. William S. Weintraub, MD, will report findings from this analysis on Nov. 16, 2019, at the annual scientific sessions of the American Heart Association.
The analysis used a wholesale acquisition cost for a 1-day dosage of icosapent ethyl of $4.16, derived from a commercial source for prescription-drug pricing and actual hospitalization costs for the patients in the trial.
Based on the REDUCE-IT outcomes, treatment with icosapent ethyl was linked with a boost in quality-adjusted life-years that extrapolated to an average 0.26 increase during the full lifetime of REDUCE-IT participants, at a cost that averaged $1,284 less per treated patient over their lifetime, according to Dr. Weintraub, director of Outcomes Research at Medstar Washington Hospital Center, Washington.
Although the 0.26 lifetime increase in quality-adjusted life-years may sound modest, “in the cost-effectiveness world, 0.26 is actually significant,” Dr. Weintraub said. He also highlighted how unusual it is to find a patented drug that improves quality of life and longevity while also saving money.
“I know of no other on-patent, branded pharmaceutical that is dominant,” he said.
Off-patent pharmaceuticals, like statins, can be quite inexpensive and may also be dominant, he noted. Being dominant for cost-effectiveness means that icosapent ethyl “provides good value and is worth what we pay for it, well within social thresholds of willingness to pay,” Dr. Weintraub said.
REDUCE-IT was sponsored by Amarin, the company that markets icosapent ethyl (Vascepa). Dr. Burman has received research funding from AstraZeneca, Eisai, and IBSA. Dr. Eckel has received personal fees from Kowa Pharmaceuticals, Merck, Novartis, and Sanofi/Regeneron, as well as research funding from Endece, Ionis Pharmaceuticals, and UniQure. Dr. Handelsman has been a consultant to and received research funding from Amarin and several other companies. Dr. Jellinger has been a speaker on behalf of Amarin, Amgen, and Regeneron. Dr. Weintraub has received honoraria and research support from Amarin, and honoraria from AstraZeneca.
Icosapent ethyl, a highly purified form of the ethyl ester of eicosapentaenoic acid, received unanimous backing from a Food and Drug Administration advisory panel for a new indication for reducing cardiovascular event risk.
Icosapent ethyl (Vascepa) received initial agency approval in 2012 for the indication of cutting triglyceride levels once they reached at least 500 mg/dL.
The target patient population for this new, cardiovascular-event protection role will reflect some or all of the types of patients enrolled in REDUCE-IT (Reduction of Cardiovascular Events with Icosapent Ethyl–Intervention Trial), which tested icosapent ethyl in 8,179 patients with either established cardiovascular disease or diabetes and at least one additional cardiovascular disease risk factor. This study provided the bulk of the data considered by the FDA panel.
REDUCE-IT showed that, during a median of 4.9 years, patients who received icosapent ethyl had a statistically significant 25% relative risk reduction in the trial’s primary, composite endpoint (New Engl J Med. 2019 Jan 3;380[1]:11-22).
Icosapent ethyl “appeared effective and safe,” and would be a “useful, new, added agent for treating patients” like those enrolled in the trial, said Kenneth D. Burman, MD, professor and chief of endocrinology at Medstar Washington (D.C.) Hospital Center and chair of the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee.
The advisory panel members appeared uniformly comfortable with recommending that the FDA add a cardiovascular disease indication based on the REDUCE-IT findings.
But while they agreed that icosapent ethyl should receive some type of indication for cardiovascular event reduction, the committee split over which patients the indication should include. Specifically, they diverged on the issue of primary prevention.
Some said that the patient enrollment that produced a positive result in REDUCE-IT should not be retrospectively subdivided, while others said that combining secondary- and primary-prevention patients in a single large trial inappropriately lumped together patients who would be better considered separately.
Committee members also expressed uncertainty over the appropriate triglyceride level to warrant treatment. The REDUCE-IT trial was designed to enroll patients with triglycerides of 135 mg/dL or greater, but several panel members suggested that, for labeling, the threshold should be at least 150 mg/dL, or even 200 mg/dL.
Safety was another aspect that generated a lot of panel discussion throughout their day-long discussion, with particular focus on a signal of a small but concerning increased rate of incident atrial fibrillation among patients who received icosapent ethyl, as well as a small but nearly significant increase in the rate of serious bleeds.
Further analyses presented during the meeting showed that an increased bleeding rate linked with icosapent ethyl was focused in patients who concurrently received one or more antiplatelet drugs or an anticoagulant.
However, panel members rejected the notion that these safety concerns warranted a boxed warning, agreeing that it could be managed with appropriate labeling information.
Clinician reaction
Clinicians who manage these types of patients viewed the prospect of an expanded indication for icosapent ethyl as an important advance.
The REDUCE-IT results by themselves “were convincing” for patients with established cardiovascular disease without need for a confirmatory trial, Robert H. Eckel, MD, an endocrinologist and professor of medicine at the University of Colorado at Denver, Aurora, said in an interview. But he remained unconvinced about efficacy for primary-prevention patients, or even for secondary-prevention patients with a triglyceride level below 150 mg/dL.
“Icosapent ethyl will clearly be a mainstay for managing high-risk patients. It gives us another treatment option,” Yehuda Handelsman, MD, an endocrinologist and medical director and principal investigator of the Metabolic Institute of America in Tarzana, Calif., said in an interview. “I do not see the atrial fibrillation or bleeding effects as reasons not to approve this drug. It should be a precaution. Overall, icosapent ethyl is one of the easier drugs for patients to take.”
Dr. Handelsman said it would be unethical to run a confirmatory trial and randomize patients to placebo. “Another trial makes no sense,” he said.
But the data from REDUCE-IT were “not as convincing” for primary-prevention patients, suggesting a need for caution about using icosapent ethyl for patients without established cardiovascular disease, Paul S. Jellinger, MD, an endocrinologist in Fort Lauderdale, Fla., said in an interview.
Cost-effectiveness
An analysis of the cost-effectiveness of icosapent ethyl as used in REDUCE-IT showed that the drug fell into the rare category of being a “dominant” treatment, meaning that it both improved patient outcomes and reduced medical costs. William S. Weintraub, MD, will report findings from this analysis on Nov. 16, 2019, at the annual scientific sessions of the American Heart Association.
The analysis used a wholesale acquisition cost for a 1-day dosage of icosapent ethyl of $4.16, derived from a commercial source for prescription-drug pricing and actual hospitalization costs for the patients in the trial.
Based on the REDUCE-IT outcomes, treatment with icosapent ethyl was linked with a boost in quality-adjusted life-years that extrapolated to an average 0.26 increase during the full lifetime of REDUCE-IT participants, at a cost that averaged $1,284 less per treated patient over their lifetime, according to Dr. Weintraub, director of Outcomes Research at Medstar Washington Hospital Center, Washington.
Although the 0.26 lifetime increase in quality-adjusted life-years may sound modest, “in the cost-effectiveness world, 0.26 is actually significant,” Dr. Weintraub said. He also highlighted how unusual it is to find a patented drug that improves quality of life and longevity while also saving money.
“I know of no other on-patent, branded pharmaceutical that is dominant,” he said.
Off-patent pharmaceuticals, like statins, can be quite inexpensive and may also be dominant, he noted. Being dominant for cost-effectiveness means that icosapent ethyl “provides good value and is worth what we pay for it, well within social thresholds of willingness to pay,” Dr. Weintraub said.
REDUCE-IT was sponsored by Amarin, the company that markets icosapent ethyl (Vascepa). Dr. Burman has received research funding from AstraZeneca, Eisai, and IBSA. Dr. Eckel has received personal fees from Kowa Pharmaceuticals, Merck, Novartis, and Sanofi/Regeneron, as well as research funding from Endece, Ionis Pharmaceuticals, and UniQure. Dr. Handelsman has been a consultant to and received research funding from Amarin and several other companies. Dr. Jellinger has been a speaker on behalf of Amarin, Amgen, and Regeneron. Dr. Weintraub has received honoraria and research support from Amarin, and honoraria from AstraZeneca.
Low LDL-C and blood pressure can reduce lifetime CVD risk by 80%
PARIS – Over the course of years and decades, lower LDL cholesterol levels and lower systolic blood pressure can reduce the lifetime risk of cardiovascular disease by up to 80%, according to a new study.
“What we found is that lifetime exposure to the combination of lower LDL and lower systolic blood pressure is associated with independent, additive, and dose-dependent effects on the lifetime risk of cardiovascular disease,” said the study’s senior author, Brian Ference, MD, speaking at the annual congress of the European Society of Cardiology. “The data seem to confirm that most cardiovascular events are preventable, and suggest that most cardiovascular events can be prevented, with prolonged exposure to modestly lower LDL cholesterol and systolic blood pressure.”
Any reduction of LDL-C and systolic blood pressure (SBP), in any combination, was associated with a lower lifetime risk of cardiovascular disease (CVD) in the study, which took advantage of the United Kingdom’s large Biobank to identify individuals with genetically lower LDL-C and blood pressure levels. The relationship was dose-dependent and showed a log-linear relationship to the combined absolute LDL-C and SBP differences, said Dr. Ference, professor and executive director of the Centre for Naturally Randomised Trials at the University of Cambridge, England.
The results validate current guidelines that focus on a lifetime approach to cardiovascular risk reduction and support a focus on therapeutic lifestyle interventions for individuals at all levels of risk for cardiovascular events, said Dr. Ference. He foresees the results shaping new risk-estimating algorithms and informing the next round of prevention guidelines.
Previous studies had suggested that long-term exposure to lower levels of LDL-C and lower systolic blood pressure reduced cardiovascular risk, but the association hadn’t been fully quantified. Ideally, said Dr. Ference, the question would be answered by a long-term randomized controlled trial, but it would be decades before meaningful data would accrue, and such a trial is unlikely to be conducted.
Using data from 438,952 Biobank participants, Dr. Ference and coinvestigators sought to quantify the association between LDL-C, systolic blood pressure, and atherosclerotic CVD. Taking advantage of genetic variants known to be associated with both lower LDL-C and lower systolic blood pressure, the researchers constructed a “natural randomization” trial. This trial design is also known as Mendelian randomization.
First, the entire study population was randomized into those with exome variants associated with higher or lower LDL-C, which resulted in a mean 15-mg/dL difference between the arms. Then, each LDL-C arm was randomized into groups with exome variants associated with higher or lower SBP, resulting in a difference of 2.9-3 mm Hg between the blood pressure arms within each LDL arm. This randomization yielded a reference group, a group with lower LDL-C, a group with lower SBP, and a group with lower LDL-C and SBP.
For the total population, the mean LDL-C was 138 mg/dL, and the mean SBP was 137.8 mm Hg.
A total of 24,980 participants had coronary revascularization, a nonfatal myocardial infarction (MI), or coronary death – the composite primary outcome measure of major coronary events.
“What we found is that long-term exposure to the combination of 1 mmol/L [about 39 mg/dL] lower LDL and 10 mm/Hg lower blood pressure is associated with an 80% lifetime reduction in risk of cardiovascular events, a 75% reduction in the risk of MI, and 68% reduction in the long-term risk of cardiovascular death,” said Dr. Ference.
By breaking participants out into separate quartiles of LDL-C and SBP levels, and examining outcomes for each quartile independently, Dr. Ference and collaborators were able to ascertain that the salutary effects of lower LDL-C and SBP were independent of each other.
Looking at individual cardiovascular outcomes, “The effect of combined exposure to both lower LDL and lower systolic blood pressure appear to be quite similar across multiple composite cardiovascular outcomes,” said Dr. Ference; benefit was seen in risk of MI, stroke, and other vascular events.
Plotting out the amount of risk reduction against the genetic scores for LDL-C and SBP reduction showed a proportional relationship that was logarithmically linear. “These large proportional reductions in risk really suggest that, for LDL, systolic blood pressure, and their combination, the benefit really depends both on the magnitude and the duration of the exposure,” said Dr. Ference. The effect was seen regardless of age, gender, body mass index, and diabetes status; being a smoker slightly attenuated the effects of LDL-C and SBP.
The mean participant age was 65 years, and women made up 54% of the study population. Aside from lipid values and systolic blood pressure, there were no significant between-group differences.
From these findings, what message can clinicians take to their patients? “Benefit is a much greater motivator, rather than the nebulous concept of risk,” said Dr. Ference. “So if we begin to crystallize and give an estimate of how much someone can benefit – either from adhering to a healthy lifestyle, with specific goals for LDL and blood pressure reductions, or from encouraging them to remain compliant with their therapies, achieving those corresponding goals – we can quantify their expected clinical benefit and encourage them to invest in their health over the long term.”
Dr. Ference said that the actual mechanism by which lipids and blood pressure are lowered matters less than the amount and duration of lowering: “These data are really agnostic as to the mechanism by which either blood pressure or LDL – or apo-B–containing lipoproteins generally – and blood pressure are reduced. It really suggests that whatever mechanism by which an individual person can most effectively lower their LDL and blood pressure, that’s the best one for that person, if they can maintain that over time.”
Dr. Ference reported financial relationships, including research contracts, consulting arrangements, receipt of royalties, and being an owner or stockholder of more than a dozen pharmaceutical companies. The study was funded by the United Kingdom’s National Institute of Health Research and Medical Research Council, and by the British Heart Foundation.
SOURCE: Ference B. et al. ESC Congress 2019, Hot Line Session 3.
Jemma Hopewell, PhD, was the assigned discussant for the Mendelian randomization study of LDL-C and SBP’s effects on cardiovascular health. She placed the genetic epidemiological study within the framework of other short- and medium-term studies that have examined the effects of LDL-C and SBP on cardiovascular health.
“Let’s think about this in the context of other studies,” said Dr. Hopewell, asking what the study adds to what’s known about exposure to LDL-C and systolic blood pressure levels. Shorter-term clinical trials that tracked differences in LDL-C over about 5 years have shown a 20%-25% drop in cardiovascular risk, while medium-term observational studies have shown a decrease of about 30%.
Now, she said, Mendelian randomization studies such as this analysis of the UK Biobank data are showing larger effects with the lifelong exposure to lower LDL levels that genetic variants confer. “As you can see, a pattern emerges ... of larger effects on risk than might be anticipated from the short-term clinical trials.”
A similar pattern can be seen with SBP, with shorter-term clinical trials showing smaller reductions in CVD. Observational studies show more reduction in risk when participants are followed for longer periods, and studies such as the present one show the larger effects of a lifetime of lower blood pressure, said Dr. Hopewell.
In terms of the combined effects, “It’s for the first time today that we see these nice results in a Mendelian randomization framework. This is a very well conducted analysis.”
Still, she cited potential limitations that can inform interpretation of the study results. These include the fact that Biobank participants have been followed for just about 10 years at this point, with most participants still alive. “Therefore, it is unclear whether this truly reflects the lifetime risk of coronary events.”
Also, the paucity of ethnic variation in the Biobank cohort means generalization is problematic until studies are conducted across different ethnic groups, she said.
The study design leaves open the possibility for reverse causality given the fact that participant characteristics captured at the time of recruitment may be influenced by prior disease, said Dr. Hopewell.
She also cited the complication of pleiotropy that’s a known limitation of Mendelian randomization studies. Importantly, the study’s reliance on genetic variation means that results may not directly translate to long-term use of lipid-lowering medication and antihypertensives, she said.
Still, the effects seen with the Biobank population bolster the importance of prevention efforts. “This really is quite encouraging,” said Dr. Hopewell. “Small differences over a long period of time have a material impact on risk.”
Dr. Hopewell is associate professor and senior scientist in genetic epidemiology and clinical trials at Oxford Cardiovascular Science, University of Oxford, England. She disclosed research contracts from unspecified pharmaceutical companies, and she has a fellowship from the British Heart Foundation.
Jemma Hopewell, PhD, was the assigned discussant for the Mendelian randomization study of LDL-C and SBP’s effects on cardiovascular health. She placed the genetic epidemiological study within the framework of other short- and medium-term studies that have examined the effects of LDL-C and SBP on cardiovascular health.
“Let’s think about this in the context of other studies,” said Dr. Hopewell, asking what the study adds to what’s known about exposure to LDL-C and systolic blood pressure levels. Shorter-term clinical trials that tracked differences in LDL-C over about 5 years have shown a 20%-25% drop in cardiovascular risk, while medium-term observational studies have shown a decrease of about 30%.
Now, she said, Mendelian randomization studies such as this analysis of the UK Biobank data are showing larger effects with the lifelong exposure to lower LDL levels that genetic variants confer. “As you can see, a pattern emerges ... of larger effects on risk than might be anticipated from the short-term clinical trials.”
A similar pattern can be seen with SBP, with shorter-term clinical trials showing smaller reductions in CVD. Observational studies show more reduction in risk when participants are followed for longer periods, and studies such as the present one show the larger effects of a lifetime of lower blood pressure, said Dr. Hopewell.
In terms of the combined effects, “It’s for the first time today that we see these nice results in a Mendelian randomization framework. This is a very well conducted analysis.”
Still, she cited potential limitations that can inform interpretation of the study results. These include the fact that Biobank participants have been followed for just about 10 years at this point, with most participants still alive. “Therefore, it is unclear whether this truly reflects the lifetime risk of coronary events.”
Also, the paucity of ethnic variation in the Biobank cohort means generalization is problematic until studies are conducted across different ethnic groups, she said.
The study design leaves open the possibility for reverse causality given the fact that participant characteristics captured at the time of recruitment may be influenced by prior disease, said Dr. Hopewell.
She also cited the complication of pleiotropy that’s a known limitation of Mendelian randomization studies. Importantly, the study’s reliance on genetic variation means that results may not directly translate to long-term use of lipid-lowering medication and antihypertensives, she said.
Still, the effects seen with the Biobank population bolster the importance of prevention efforts. “This really is quite encouraging,” said Dr. Hopewell. “Small differences over a long period of time have a material impact on risk.”
Dr. Hopewell is associate professor and senior scientist in genetic epidemiology and clinical trials at Oxford Cardiovascular Science, University of Oxford, England. She disclosed research contracts from unspecified pharmaceutical companies, and she has a fellowship from the British Heart Foundation.
Jemma Hopewell, PhD, was the assigned discussant for the Mendelian randomization study of LDL-C and SBP’s effects on cardiovascular health. She placed the genetic epidemiological study within the framework of other short- and medium-term studies that have examined the effects of LDL-C and SBP on cardiovascular health.
“Let’s think about this in the context of other studies,” said Dr. Hopewell, asking what the study adds to what’s known about exposure to LDL-C and systolic blood pressure levels. Shorter-term clinical trials that tracked differences in LDL-C over about 5 years have shown a 20%-25% drop in cardiovascular risk, while medium-term observational studies have shown a decrease of about 30%.
Now, she said, Mendelian randomization studies such as this analysis of the UK Biobank data are showing larger effects with the lifelong exposure to lower LDL levels that genetic variants confer. “As you can see, a pattern emerges ... of larger effects on risk than might be anticipated from the short-term clinical trials.”
A similar pattern can be seen with SBP, with shorter-term clinical trials showing smaller reductions in CVD. Observational studies show more reduction in risk when participants are followed for longer periods, and studies such as the present one show the larger effects of a lifetime of lower blood pressure, said Dr. Hopewell.
In terms of the combined effects, “It’s for the first time today that we see these nice results in a Mendelian randomization framework. This is a very well conducted analysis.”
Still, she cited potential limitations that can inform interpretation of the study results. These include the fact that Biobank participants have been followed for just about 10 years at this point, with most participants still alive. “Therefore, it is unclear whether this truly reflects the lifetime risk of coronary events.”
Also, the paucity of ethnic variation in the Biobank cohort means generalization is problematic until studies are conducted across different ethnic groups, she said.
The study design leaves open the possibility for reverse causality given the fact that participant characteristics captured at the time of recruitment may be influenced by prior disease, said Dr. Hopewell.
She also cited the complication of pleiotropy that’s a known limitation of Mendelian randomization studies. Importantly, the study’s reliance on genetic variation means that results may not directly translate to long-term use of lipid-lowering medication and antihypertensives, she said.
Still, the effects seen with the Biobank population bolster the importance of prevention efforts. “This really is quite encouraging,” said Dr. Hopewell. “Small differences over a long period of time have a material impact on risk.”
Dr. Hopewell is associate professor and senior scientist in genetic epidemiology and clinical trials at Oxford Cardiovascular Science, University of Oxford, England. She disclosed research contracts from unspecified pharmaceutical companies, and she has a fellowship from the British Heart Foundation.
PARIS – Over the course of years and decades, lower LDL cholesterol levels and lower systolic blood pressure can reduce the lifetime risk of cardiovascular disease by up to 80%, according to a new study.
“What we found is that lifetime exposure to the combination of lower LDL and lower systolic blood pressure is associated with independent, additive, and dose-dependent effects on the lifetime risk of cardiovascular disease,” said the study’s senior author, Brian Ference, MD, speaking at the annual congress of the European Society of Cardiology. “The data seem to confirm that most cardiovascular events are preventable, and suggest that most cardiovascular events can be prevented, with prolonged exposure to modestly lower LDL cholesterol and systolic blood pressure.”
Any reduction of LDL-C and systolic blood pressure (SBP), in any combination, was associated with a lower lifetime risk of cardiovascular disease (CVD) in the study, which took advantage of the United Kingdom’s large Biobank to identify individuals with genetically lower LDL-C and blood pressure levels. The relationship was dose-dependent and showed a log-linear relationship to the combined absolute LDL-C and SBP differences, said Dr. Ference, professor and executive director of the Centre for Naturally Randomised Trials at the University of Cambridge, England.
The results validate current guidelines that focus on a lifetime approach to cardiovascular risk reduction and support a focus on therapeutic lifestyle interventions for individuals at all levels of risk for cardiovascular events, said Dr. Ference. He foresees the results shaping new risk-estimating algorithms and informing the next round of prevention guidelines.
Previous studies had suggested that long-term exposure to lower levels of LDL-C and lower systolic blood pressure reduced cardiovascular risk, but the association hadn’t been fully quantified. Ideally, said Dr. Ference, the question would be answered by a long-term randomized controlled trial, but it would be decades before meaningful data would accrue, and such a trial is unlikely to be conducted.
Using data from 438,952 Biobank participants, Dr. Ference and coinvestigators sought to quantify the association between LDL-C, systolic blood pressure, and atherosclerotic CVD. Taking advantage of genetic variants known to be associated with both lower LDL-C and lower systolic blood pressure, the researchers constructed a “natural randomization” trial. This trial design is also known as Mendelian randomization.
First, the entire study population was randomized into those with exome variants associated with higher or lower LDL-C, which resulted in a mean 15-mg/dL difference between the arms. Then, each LDL-C arm was randomized into groups with exome variants associated with higher or lower SBP, resulting in a difference of 2.9-3 mm Hg between the blood pressure arms within each LDL arm. This randomization yielded a reference group, a group with lower LDL-C, a group with lower SBP, and a group with lower LDL-C and SBP.
For the total population, the mean LDL-C was 138 mg/dL, and the mean SBP was 137.8 mm Hg.
A total of 24,980 participants had coronary revascularization, a nonfatal myocardial infarction (MI), or coronary death – the composite primary outcome measure of major coronary events.
“What we found is that long-term exposure to the combination of 1 mmol/L [about 39 mg/dL] lower LDL and 10 mm/Hg lower blood pressure is associated with an 80% lifetime reduction in risk of cardiovascular events, a 75% reduction in the risk of MI, and 68% reduction in the long-term risk of cardiovascular death,” said Dr. Ference.
By breaking participants out into separate quartiles of LDL-C and SBP levels, and examining outcomes for each quartile independently, Dr. Ference and collaborators were able to ascertain that the salutary effects of lower LDL-C and SBP were independent of each other.
Looking at individual cardiovascular outcomes, “The effect of combined exposure to both lower LDL and lower systolic blood pressure appear to be quite similar across multiple composite cardiovascular outcomes,” said Dr. Ference; benefit was seen in risk of MI, stroke, and other vascular events.
Plotting out the amount of risk reduction against the genetic scores for LDL-C and SBP reduction showed a proportional relationship that was logarithmically linear. “These large proportional reductions in risk really suggest that, for LDL, systolic blood pressure, and their combination, the benefit really depends both on the magnitude and the duration of the exposure,” said Dr. Ference. The effect was seen regardless of age, gender, body mass index, and diabetes status; being a smoker slightly attenuated the effects of LDL-C and SBP.
The mean participant age was 65 years, and women made up 54% of the study population. Aside from lipid values and systolic blood pressure, there were no significant between-group differences.
From these findings, what message can clinicians take to their patients? “Benefit is a much greater motivator, rather than the nebulous concept of risk,” said Dr. Ference. “So if we begin to crystallize and give an estimate of how much someone can benefit – either from adhering to a healthy lifestyle, with specific goals for LDL and blood pressure reductions, or from encouraging them to remain compliant with their therapies, achieving those corresponding goals – we can quantify their expected clinical benefit and encourage them to invest in their health over the long term.”
Dr. Ference said that the actual mechanism by which lipids and blood pressure are lowered matters less than the amount and duration of lowering: “These data are really agnostic as to the mechanism by which either blood pressure or LDL – or apo-B–containing lipoproteins generally – and blood pressure are reduced. It really suggests that whatever mechanism by which an individual person can most effectively lower their LDL and blood pressure, that’s the best one for that person, if they can maintain that over time.”
Dr. Ference reported financial relationships, including research contracts, consulting arrangements, receipt of royalties, and being an owner or stockholder of more than a dozen pharmaceutical companies. The study was funded by the United Kingdom’s National Institute of Health Research and Medical Research Council, and by the British Heart Foundation.
SOURCE: Ference B. et al. ESC Congress 2019, Hot Line Session 3.
PARIS – Over the course of years and decades, lower LDL cholesterol levels and lower systolic blood pressure can reduce the lifetime risk of cardiovascular disease by up to 80%, according to a new study.
“What we found is that lifetime exposure to the combination of lower LDL and lower systolic blood pressure is associated with independent, additive, and dose-dependent effects on the lifetime risk of cardiovascular disease,” said the study’s senior author, Brian Ference, MD, speaking at the annual congress of the European Society of Cardiology. “The data seem to confirm that most cardiovascular events are preventable, and suggest that most cardiovascular events can be prevented, with prolonged exposure to modestly lower LDL cholesterol and systolic blood pressure.”
Any reduction of LDL-C and systolic blood pressure (SBP), in any combination, was associated with a lower lifetime risk of cardiovascular disease (CVD) in the study, which took advantage of the United Kingdom’s large Biobank to identify individuals with genetically lower LDL-C and blood pressure levels. The relationship was dose-dependent and showed a log-linear relationship to the combined absolute LDL-C and SBP differences, said Dr. Ference, professor and executive director of the Centre for Naturally Randomised Trials at the University of Cambridge, England.
The results validate current guidelines that focus on a lifetime approach to cardiovascular risk reduction and support a focus on therapeutic lifestyle interventions for individuals at all levels of risk for cardiovascular events, said Dr. Ference. He foresees the results shaping new risk-estimating algorithms and informing the next round of prevention guidelines.
Previous studies had suggested that long-term exposure to lower levels of LDL-C and lower systolic blood pressure reduced cardiovascular risk, but the association hadn’t been fully quantified. Ideally, said Dr. Ference, the question would be answered by a long-term randomized controlled trial, but it would be decades before meaningful data would accrue, and such a trial is unlikely to be conducted.
Using data from 438,952 Biobank participants, Dr. Ference and coinvestigators sought to quantify the association between LDL-C, systolic blood pressure, and atherosclerotic CVD. Taking advantage of genetic variants known to be associated with both lower LDL-C and lower systolic blood pressure, the researchers constructed a “natural randomization” trial. This trial design is also known as Mendelian randomization.
First, the entire study population was randomized into those with exome variants associated with higher or lower LDL-C, which resulted in a mean 15-mg/dL difference between the arms. Then, each LDL-C arm was randomized into groups with exome variants associated with higher or lower SBP, resulting in a difference of 2.9-3 mm Hg between the blood pressure arms within each LDL arm. This randomization yielded a reference group, a group with lower LDL-C, a group with lower SBP, and a group with lower LDL-C and SBP.
For the total population, the mean LDL-C was 138 mg/dL, and the mean SBP was 137.8 mm Hg.
A total of 24,980 participants had coronary revascularization, a nonfatal myocardial infarction (MI), or coronary death – the composite primary outcome measure of major coronary events.
“What we found is that long-term exposure to the combination of 1 mmol/L [about 39 mg/dL] lower LDL and 10 mm/Hg lower blood pressure is associated with an 80% lifetime reduction in risk of cardiovascular events, a 75% reduction in the risk of MI, and 68% reduction in the long-term risk of cardiovascular death,” said Dr. Ference.
By breaking participants out into separate quartiles of LDL-C and SBP levels, and examining outcomes for each quartile independently, Dr. Ference and collaborators were able to ascertain that the salutary effects of lower LDL-C and SBP were independent of each other.
Looking at individual cardiovascular outcomes, “The effect of combined exposure to both lower LDL and lower systolic blood pressure appear to be quite similar across multiple composite cardiovascular outcomes,” said Dr. Ference; benefit was seen in risk of MI, stroke, and other vascular events.
Plotting out the amount of risk reduction against the genetic scores for LDL-C and SBP reduction showed a proportional relationship that was logarithmically linear. “These large proportional reductions in risk really suggest that, for LDL, systolic blood pressure, and their combination, the benefit really depends both on the magnitude and the duration of the exposure,” said Dr. Ference. The effect was seen regardless of age, gender, body mass index, and diabetes status; being a smoker slightly attenuated the effects of LDL-C and SBP.
The mean participant age was 65 years, and women made up 54% of the study population. Aside from lipid values and systolic blood pressure, there were no significant between-group differences.
From these findings, what message can clinicians take to their patients? “Benefit is a much greater motivator, rather than the nebulous concept of risk,” said Dr. Ference. “So if we begin to crystallize and give an estimate of how much someone can benefit – either from adhering to a healthy lifestyle, with specific goals for LDL and blood pressure reductions, or from encouraging them to remain compliant with their therapies, achieving those corresponding goals – we can quantify their expected clinical benefit and encourage them to invest in their health over the long term.”
Dr. Ference said that the actual mechanism by which lipids and blood pressure are lowered matters less than the amount and duration of lowering: “These data are really agnostic as to the mechanism by which either blood pressure or LDL – or apo-B–containing lipoproteins generally – and blood pressure are reduced. It really suggests that whatever mechanism by which an individual person can most effectively lower their LDL and blood pressure, that’s the best one for that person, if they can maintain that over time.”
Dr. Ference reported financial relationships, including research contracts, consulting arrangements, receipt of royalties, and being an owner or stockholder of more than a dozen pharmaceutical companies. The study was funded by the United Kingdom’s National Institute of Health Research and Medical Research Council, and by the British Heart Foundation.
SOURCE: Ference B. et al. ESC Congress 2019, Hot Line Session 3.
REPORTING FROM ESC CONGRESS 2019
White and black patients have similar rates of giant cell arteritis
To determine the incidence of biopsy-proven GCA (BP-GCA) in a racially diverse cohort, Anna M. Gruener of Nottingham (England) University Hospitals NHS Trust and coauthors analyzed the medical records of more than 10 years of patients who underwent temporal artery biopsy at Johns Hopkins Wilmer Eye Institute in Baltimore. Of the 586 patients in the study, 167 (28.5%) were black, 382 (65.2%) were white, and 37 (6.3%) were other or unknown. The mean age was 70.5 years.
Of the 573 patients who were aged 50 years and older, 92 (16.1%) had a positive biopsy for BP-GCA; 14 were black (8.4% of all black patients), 75 were white (19.6% of all white patients), and 3 were other or unknown. The population-adjusted, age- and sex-standardized incidence rates per 100,000 were 3.1 (95% confidence interval, 1.0-5.2) for black patients and 3.6 (95% CI, 2.5-4.7) for white patients.
Overall, BP-GCA occurred more frequently in women than in men (incidence rate ratio, 1.9; 95% CI, 1.1-3.4; P = .03) but at similar levels in white and black patients (IRR, 1.2; 95% CI, 0.6-2.4; P = .66).
In an accompanying editorial, Michael K. Yoon, MD, and Joseph F. Rizzo III, MD, of Harvard Medical School, Boston, praised the researchers for conducting their study in a population with a large percentage of black patients, a noted weakness of earlier studies in this area (JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2933). That said, the two doctors also recognized the limitations of the work done by Gruener et al., including relying on U.S. Census data to calculate adjusted incidence rates instead of local racial distribution and also the potentially problematic choice to count patients with healed arteritis as having BP-GCA.
Still, Dr. Yoon and Dr. Rizzo commended Gruener et al. for questioning previous findings on GCA rates. “Although the authors’ methods are imperfect,” they wrote, “the studies that had previously established a low incidence of GCA in black patients were also flawed in design.”
The study had no outside funding source, and no conflicts of interest were reported.
SOURCE: Gruener AM et al. JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2919.
To determine the incidence of biopsy-proven GCA (BP-GCA) in a racially diverse cohort, Anna M. Gruener of Nottingham (England) University Hospitals NHS Trust and coauthors analyzed the medical records of more than 10 years of patients who underwent temporal artery biopsy at Johns Hopkins Wilmer Eye Institute in Baltimore. Of the 586 patients in the study, 167 (28.5%) were black, 382 (65.2%) were white, and 37 (6.3%) were other or unknown. The mean age was 70.5 years.
Of the 573 patients who were aged 50 years and older, 92 (16.1%) had a positive biopsy for BP-GCA; 14 were black (8.4% of all black patients), 75 were white (19.6% of all white patients), and 3 were other or unknown. The population-adjusted, age- and sex-standardized incidence rates per 100,000 were 3.1 (95% confidence interval, 1.0-5.2) for black patients and 3.6 (95% CI, 2.5-4.7) for white patients.
Overall, BP-GCA occurred more frequently in women than in men (incidence rate ratio, 1.9; 95% CI, 1.1-3.4; P = .03) but at similar levels in white and black patients (IRR, 1.2; 95% CI, 0.6-2.4; P = .66).
In an accompanying editorial, Michael K. Yoon, MD, and Joseph F. Rizzo III, MD, of Harvard Medical School, Boston, praised the researchers for conducting their study in a population with a large percentage of black patients, a noted weakness of earlier studies in this area (JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2933). That said, the two doctors also recognized the limitations of the work done by Gruener et al., including relying on U.S. Census data to calculate adjusted incidence rates instead of local racial distribution and also the potentially problematic choice to count patients with healed arteritis as having BP-GCA.
Still, Dr. Yoon and Dr. Rizzo commended Gruener et al. for questioning previous findings on GCA rates. “Although the authors’ methods are imperfect,” they wrote, “the studies that had previously established a low incidence of GCA in black patients were also flawed in design.”
The study had no outside funding source, and no conflicts of interest were reported.
SOURCE: Gruener AM et al. JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2919.
To determine the incidence of biopsy-proven GCA (BP-GCA) in a racially diverse cohort, Anna M. Gruener of Nottingham (England) University Hospitals NHS Trust and coauthors analyzed the medical records of more than 10 years of patients who underwent temporal artery biopsy at Johns Hopkins Wilmer Eye Institute in Baltimore. Of the 586 patients in the study, 167 (28.5%) were black, 382 (65.2%) were white, and 37 (6.3%) were other or unknown. The mean age was 70.5 years.
Of the 573 patients who were aged 50 years and older, 92 (16.1%) had a positive biopsy for BP-GCA; 14 were black (8.4% of all black patients), 75 were white (19.6% of all white patients), and 3 were other or unknown. The population-adjusted, age- and sex-standardized incidence rates per 100,000 were 3.1 (95% confidence interval, 1.0-5.2) for black patients and 3.6 (95% CI, 2.5-4.7) for white patients.
Overall, BP-GCA occurred more frequently in women than in men (incidence rate ratio, 1.9; 95% CI, 1.1-3.4; P = .03) but at similar levels in white and black patients (IRR, 1.2; 95% CI, 0.6-2.4; P = .66).
In an accompanying editorial, Michael K. Yoon, MD, and Joseph F. Rizzo III, MD, of Harvard Medical School, Boston, praised the researchers for conducting their study in a population with a large percentage of black patients, a noted weakness of earlier studies in this area (JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2933). That said, the two doctors also recognized the limitations of the work done by Gruener et al., including relying on U.S. Census data to calculate adjusted incidence rates instead of local racial distribution and also the potentially problematic choice to count patients with healed arteritis as having BP-GCA.
Still, Dr. Yoon and Dr. Rizzo commended Gruener et al. for questioning previous findings on GCA rates. “Although the authors’ methods are imperfect,” they wrote, “the studies that had previously established a low incidence of GCA in black patients were also flawed in design.”
The study had no outside funding source, and no conflicts of interest were reported.
SOURCE: Gruener AM et al. JAMA Ophthalmol. 2019 Aug 8. doi: 10.1001/jamaophthalmol.2019.2919.
FROM JAMA OPHTHALMOLOGY
Carotid ultrasound may aid cardiovascular risk stratification of patients with psoriatic disease
according to findings from a retrospective study.
When added to the Framingham risk score, the measurement significantly improved its predictive ability, Curtis Sobchak, MD, and colleagues wrote in Arthritis & Rheumatology.
The findings indicate that carotid ultrasound could be a useful addition to cardiovascular risk stratification among these patients.
“Traditional algorithms do not consider other factors that may contribute to increased cardiovascular risk in rheumatic disease patients and tend to underestimate cardiovascular risk,” wrote Dr. Sobchak of the University of Toronto and coauthors.
“The advantage of ultrasound over other modalities for vascular imaging includes lack of radiation, low cost of the examination, and its widespread use in rheumatology for joint evaluation. Thus, this assessment could potentially be performed ‘at the bedside’ during consultation to provide immediate valuable information to complement clinical data from history, physical examination, and laboratory data,” they added.
The study retrospectively examined a prospective, observational cohort of 559 patients with psoriasis alone or psoriasis and psoriatic arthritis enrolled in the University of Toronto Psoriatic Disease Program. The investigators evaluated five ultrasound measures of atherosclerosis, including total plaque area (TPA), mean carotid intima-media thickness (cIMT), maximal cIMT, plaque category, and TPA category. Then they analyzed the risk relationship with major cardiovascular events (CVEs) classified as myocardial infarction, unstable angina, ischemic stroke, revascularization procedures, or cardiovascular-related death. Minor CVEs included stable angina, exacerbation of congestive heart failure, and transient ischemic attack over a mean follow-up close to 4 years.
The mean baseline TPA was 0.18 cm2 and mean cIMT was 639 mcm. Most patients had plaques, including 27.0% with unilateral and 31.5% with bilateral plaques.
The rate of a first CVE during the study period was 1.11 per 100 patient-years, and the rate of a first major CVE was 0.91 per 100 patient-years. The risk of each was significantly related to a higher baseline burden of atherosclerosis.
A multivariate analysis determined that increased TPA at baseline increased the risk of an event by nearly 200% (hazard ratio, 2.85). Mean cIMT was not an independent predictor in the final analysis, “suggesting that TPA is a stronger predictor for CVE than cIMT,” the authors wrote.
Finally, they examined the predictive value of atherosclerosis alone, as well as combined with the Framingham risk score. The 5-year model indicated that the bivariate model was slightly more accurate than the Framingham score alone (area under the curve, 0.84 vs. 0.81), although this was not a significant difference. The predictive value of the Framingham risk score plus maximal cIMT, mean cIMT, or TPA all significantly improved when they were calculated using only high-risk patients (those above the treatment threshold for dyslipidemia).
“To the best of our knowledge this is the first study to assess the utility of various measures of carotid atherosclerosis to predict CVE in patients with psoriasis and PsA [psoriatic arthritis]. ... Combining vascular imaging data with clinical and laboratory measures of traditional cardiovascular risk factors could improve accuracy of cardiovascular risk stratification in patients with psoriatic disease and facilitate earlier initiation of appropriate treatment to reduce CVE in this population,” the investigators wrote.
The study was supported in part by a Young Investigator Operating Grant from the Arthritis Society. Dr. Sobchak had no financial disclosures.
SOURCE: Sobchak C et al. Arthritis Rheumatol. 2019 Jun 5. doi: 10.1002/art.40925.
according to findings from a retrospective study.
When added to the Framingham risk score, the measurement significantly improved its predictive ability, Curtis Sobchak, MD, and colleagues wrote in Arthritis & Rheumatology.
The findings indicate that carotid ultrasound could be a useful addition to cardiovascular risk stratification among these patients.
“Traditional algorithms do not consider other factors that may contribute to increased cardiovascular risk in rheumatic disease patients and tend to underestimate cardiovascular risk,” wrote Dr. Sobchak of the University of Toronto and coauthors.
“The advantage of ultrasound over other modalities for vascular imaging includes lack of radiation, low cost of the examination, and its widespread use in rheumatology for joint evaluation. Thus, this assessment could potentially be performed ‘at the bedside’ during consultation to provide immediate valuable information to complement clinical data from history, physical examination, and laboratory data,” they added.
The study retrospectively examined a prospective, observational cohort of 559 patients with psoriasis alone or psoriasis and psoriatic arthritis enrolled in the University of Toronto Psoriatic Disease Program. The investigators evaluated five ultrasound measures of atherosclerosis, including total plaque area (TPA), mean carotid intima-media thickness (cIMT), maximal cIMT, plaque category, and TPA category. Then they analyzed the risk relationship with major cardiovascular events (CVEs) classified as myocardial infarction, unstable angina, ischemic stroke, revascularization procedures, or cardiovascular-related death. Minor CVEs included stable angina, exacerbation of congestive heart failure, and transient ischemic attack over a mean follow-up close to 4 years.
The mean baseline TPA was 0.18 cm2 and mean cIMT was 639 mcm. Most patients had plaques, including 27.0% with unilateral and 31.5% with bilateral plaques.
The rate of a first CVE during the study period was 1.11 per 100 patient-years, and the rate of a first major CVE was 0.91 per 100 patient-years. The risk of each was significantly related to a higher baseline burden of atherosclerosis.
A multivariate analysis determined that increased TPA at baseline increased the risk of an event by nearly 200% (hazard ratio, 2.85). Mean cIMT was not an independent predictor in the final analysis, “suggesting that TPA is a stronger predictor for CVE than cIMT,” the authors wrote.
Finally, they examined the predictive value of atherosclerosis alone, as well as combined with the Framingham risk score. The 5-year model indicated that the bivariate model was slightly more accurate than the Framingham score alone (area under the curve, 0.84 vs. 0.81), although this was not a significant difference. The predictive value of the Framingham risk score plus maximal cIMT, mean cIMT, or TPA all significantly improved when they were calculated using only high-risk patients (those above the treatment threshold for dyslipidemia).
“To the best of our knowledge this is the first study to assess the utility of various measures of carotid atherosclerosis to predict CVE in patients with psoriasis and PsA [psoriatic arthritis]. ... Combining vascular imaging data with clinical and laboratory measures of traditional cardiovascular risk factors could improve accuracy of cardiovascular risk stratification in patients with psoriatic disease and facilitate earlier initiation of appropriate treatment to reduce CVE in this population,” the investigators wrote.
The study was supported in part by a Young Investigator Operating Grant from the Arthritis Society. Dr. Sobchak had no financial disclosures.
SOURCE: Sobchak C et al. Arthritis Rheumatol. 2019 Jun 5. doi: 10.1002/art.40925.
according to findings from a retrospective study.
When added to the Framingham risk score, the measurement significantly improved its predictive ability, Curtis Sobchak, MD, and colleagues wrote in Arthritis & Rheumatology.
The findings indicate that carotid ultrasound could be a useful addition to cardiovascular risk stratification among these patients.
“Traditional algorithms do not consider other factors that may contribute to increased cardiovascular risk in rheumatic disease patients and tend to underestimate cardiovascular risk,” wrote Dr. Sobchak of the University of Toronto and coauthors.
“The advantage of ultrasound over other modalities for vascular imaging includes lack of radiation, low cost of the examination, and its widespread use in rheumatology for joint evaluation. Thus, this assessment could potentially be performed ‘at the bedside’ during consultation to provide immediate valuable information to complement clinical data from history, physical examination, and laboratory data,” they added.
The study retrospectively examined a prospective, observational cohort of 559 patients with psoriasis alone or psoriasis and psoriatic arthritis enrolled in the University of Toronto Psoriatic Disease Program. The investigators evaluated five ultrasound measures of atherosclerosis, including total plaque area (TPA), mean carotid intima-media thickness (cIMT), maximal cIMT, plaque category, and TPA category. Then they analyzed the risk relationship with major cardiovascular events (CVEs) classified as myocardial infarction, unstable angina, ischemic stroke, revascularization procedures, or cardiovascular-related death. Minor CVEs included stable angina, exacerbation of congestive heart failure, and transient ischemic attack over a mean follow-up close to 4 years.
The mean baseline TPA was 0.18 cm2 and mean cIMT was 639 mcm. Most patients had plaques, including 27.0% with unilateral and 31.5% with bilateral plaques.
The rate of a first CVE during the study period was 1.11 per 100 patient-years, and the rate of a first major CVE was 0.91 per 100 patient-years. The risk of each was significantly related to a higher baseline burden of atherosclerosis.
A multivariate analysis determined that increased TPA at baseline increased the risk of an event by nearly 200% (hazard ratio, 2.85). Mean cIMT was not an independent predictor in the final analysis, “suggesting that TPA is a stronger predictor for CVE than cIMT,” the authors wrote.
Finally, they examined the predictive value of atherosclerosis alone, as well as combined with the Framingham risk score. The 5-year model indicated that the bivariate model was slightly more accurate than the Framingham score alone (area under the curve, 0.84 vs. 0.81), although this was not a significant difference. The predictive value of the Framingham risk score plus maximal cIMT, mean cIMT, or TPA all significantly improved when they were calculated using only high-risk patients (those above the treatment threshold for dyslipidemia).
“To the best of our knowledge this is the first study to assess the utility of various measures of carotid atherosclerosis to predict CVE in patients with psoriasis and PsA [psoriatic arthritis]. ... Combining vascular imaging data with clinical and laboratory measures of traditional cardiovascular risk factors could improve accuracy of cardiovascular risk stratification in patients with psoriatic disease and facilitate earlier initiation of appropriate treatment to reduce CVE in this population,” the investigators wrote.
The study was supported in part by a Young Investigator Operating Grant from the Arthritis Society. Dr. Sobchak had no financial disclosures.
SOURCE: Sobchak C et al. Arthritis Rheumatol. 2019 Jun 5. doi: 10.1002/art.40925.
FROM ARTHRITIS & RHEUMATOLOGY
Alirocumab gains indication to reduce cardiovascular risks
Alirocumab has received an updated indication from the Food and Drug Administration for reducing the overall risk of major adverse cardiovascular events in patients with a recent acute coronary event.
Alirocumab is designed to inhibit the binding of PCSK9 (proprotein convertase subtilisin/kexin type 9) to LDL receptors, thereby lowering LDL cholesterol, according to manufacturer Regeneron, which is developing alirocumab in partnership with Sanofi.
The drug was previously approved in the United States as an adjunct treatment along with diet and maximally tolerated statin therapy to help lower LDL cholesterol in adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease.
The approval of the supplemental Biologics License Application was supported by data from the ODYSSEY Outcomes trial in which 18,924 patients who had an acute coronary syndrome were randomized to alirocumab or placebo plus background high-intensity statin therapy starting at a median of 2.6 months after the event. Over 3 years’ follow-up, a composite endpoint outcome including death from coronary heart disease, nonfatal myocardial infarction, ischemic stroke, or unstable angina occurred in 9.5% of alirocumab patients and 11.1% of placebo patients.
In the study, patients received subcutaneous dose of 75 mg of alirocumab every 2 weeks, which was adjusted to achieve an LDL cholesterol level of 25-50 mg/dL. The most significant benefits occurred among patients with a baseline LDL cholesterol of 100 mg/dL or higher who were taking high-intensity statins, which supports the role of LDL cholesterol reduction in improving outcomes for coronary syndrome patients, according to study investigators.
Alirocumab is given as a subcutaneous injection. The most common side effects include pain and tenderness at the injection site, and redness, itching, or swelling; some patients have reported symptoms of a common cold or flu.
More details of the ODYSSEY Outcomes trial were presented at the annual meeting of the American College of Cardiology.
Alirocumab has received an updated indication from the Food and Drug Administration for reducing the overall risk of major adverse cardiovascular events in patients with a recent acute coronary event.
Alirocumab is designed to inhibit the binding of PCSK9 (proprotein convertase subtilisin/kexin type 9) to LDL receptors, thereby lowering LDL cholesterol, according to manufacturer Regeneron, which is developing alirocumab in partnership with Sanofi.
The drug was previously approved in the United States as an adjunct treatment along with diet and maximally tolerated statin therapy to help lower LDL cholesterol in adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease.
The approval of the supplemental Biologics License Application was supported by data from the ODYSSEY Outcomes trial in which 18,924 patients who had an acute coronary syndrome were randomized to alirocumab or placebo plus background high-intensity statin therapy starting at a median of 2.6 months after the event. Over 3 years’ follow-up, a composite endpoint outcome including death from coronary heart disease, nonfatal myocardial infarction, ischemic stroke, or unstable angina occurred in 9.5% of alirocumab patients and 11.1% of placebo patients.
In the study, patients received subcutaneous dose of 75 mg of alirocumab every 2 weeks, which was adjusted to achieve an LDL cholesterol level of 25-50 mg/dL. The most significant benefits occurred among patients with a baseline LDL cholesterol of 100 mg/dL or higher who were taking high-intensity statins, which supports the role of LDL cholesterol reduction in improving outcomes for coronary syndrome patients, according to study investigators.
Alirocumab is given as a subcutaneous injection. The most common side effects include pain and tenderness at the injection site, and redness, itching, or swelling; some patients have reported symptoms of a common cold or flu.
More details of the ODYSSEY Outcomes trial were presented at the annual meeting of the American College of Cardiology.
Alirocumab has received an updated indication from the Food and Drug Administration for reducing the overall risk of major adverse cardiovascular events in patients with a recent acute coronary event.
Alirocumab is designed to inhibit the binding of PCSK9 (proprotein convertase subtilisin/kexin type 9) to LDL receptors, thereby lowering LDL cholesterol, according to manufacturer Regeneron, which is developing alirocumab in partnership with Sanofi.
The drug was previously approved in the United States as an adjunct treatment along with diet and maximally tolerated statin therapy to help lower LDL cholesterol in adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease.
The approval of the supplemental Biologics License Application was supported by data from the ODYSSEY Outcomes trial in which 18,924 patients who had an acute coronary syndrome were randomized to alirocumab or placebo plus background high-intensity statin therapy starting at a median of 2.6 months after the event. Over 3 years’ follow-up, a composite endpoint outcome including death from coronary heart disease, nonfatal myocardial infarction, ischemic stroke, or unstable angina occurred in 9.5% of alirocumab patients and 11.1% of placebo patients.
In the study, patients received subcutaneous dose of 75 mg of alirocumab every 2 weeks, which was adjusted to achieve an LDL cholesterol level of 25-50 mg/dL. The most significant benefits occurred among patients with a baseline LDL cholesterol of 100 mg/dL or higher who were taking high-intensity statins, which supports the role of LDL cholesterol reduction in improving outcomes for coronary syndrome patients, according to study investigators.
Alirocumab is given as a subcutaneous injection. The most common side effects include pain and tenderness at the injection site, and redness, itching, or swelling; some patients have reported symptoms of a common cold or flu.
More details of the ODYSSEY Outcomes trial were presented at the annual meeting of the American College of Cardiology.
Deep sleep decreases, Alzheimer’s increases
Also today, physician groups are pushing back on Part B of the drug reimbursement proposal, dabigatran matches aspirin for second stroke prevention, and reassurance for pregnancy in atopic dermatitis.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, physician groups are pushing back on Part B of the drug reimbursement proposal, dabigatran matches aspirin for second stroke prevention, and reassurance for pregnancy in atopic dermatitis.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
Also today, physician groups are pushing back on Part B of the drug reimbursement proposal, dabigatran matches aspirin for second stroke prevention, and reassurance for pregnancy in atopic dermatitis.
Amazon Alexa
Apple Podcasts
Google Podcasts
Spotify
In Medicare population, carotid revascularization has declined
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
NEW YORK – The rates of carotid artery revascularization with either endarterectomy or stenting declined precipitously over a recent 15-year period, at least among Medicare fee-for-service beneficiaries, according to data presented at a symposium on vascular and endovascular issues sponsored by the Cleveland Clinic Foundation.
A reduction in carotid endarterectomies (CEA) largely accounted for the decline during 1999-2014 although there was a cumulative decline in all carotid revascularization procedures when rates of CEA and stenting were combined, according to Brajesh K. Lal, MD, professor of surgery, University of Maryland Medical System, Baltimore.
In 1999, when enthusiasm for CEA appears to have peaked, 81,306 patients received this procedure, but a steady decline was observed until 2014, when 36,325 patients were being treated annually in the Medicare database. When calculated as endarterectomies per 100,000 beneficiaries, the rate declined from 298 to 128 (57%; P less than .001) over this 15-year period.
The number of stenting procedures had not reached its peak in 1999, when 10,416 were performed. Rather, the number performed annually nearly doubled to, 22.865 by 2006. However, it then began to decline and reached 10,208 by 2014, which was slightly fewer than in 1999, according to Dr. Lal.
These trends have been observed even though outcomes are getting better, at least for CEA, according to Dr. Lal. From the same pool of data, there was a 31% (1.1% vs. 1.6%) reduction from 1999 to 2014 in mortality at 30 days following CEA. For a composite of ischemic stroke and all-cause mortality, the rate fell 29.5% (3.1% vs. 4.4%). Both reductions were called statistically significant by Dr. Lal.
The improvements in CEA outcomes were observed even though “the treated patients got sicker when looking at comorbidities and risk factors, particularly hypertension, renal insufficiency, and diabetes,” Dr. Lal said.
Outcomes also improved among patients undergoing carotid stenting in general, although the patterns were described as “more complex.” In general, there was steady improvement on outcomes during 1999-2006, but there was no further gain and some lost ground during 2006-2014. For example, ischemic stroke or death fell from 7.0% in 1999 to 4.8% in 2006, but it had climbed back to 7.0% by 2014 with no net change when the first and last year were compared.
However, with risk adjustment, there was a reduction in in-hospital mortality (1.13% vs. 2.78%) over the study period for patients undergoing carotid stenting, according to Dr. Lal, who said this reached statistical significance. Like the CEA group, there was more comorbidity among those treated with stenting at the end, relative to the early part of the study period.
In the stenting group, patients with symptomatic carotid disease rose from 14.4% in 1999 to 25.9% in 2014. This tracks with Medicare policy, which required patients after 2005 to have symptomatic disease for reimbursement, according to Dr. Lal. Prior to 2005, reimbursement was granted for patients participating in clinical trials only.
The rates of carotid revascularization are not evenly distributed geographically in the United States, according to the Medicare data. Endarterectomy in particular has been more common in the south and Midwest than on either coast. This was true in 1999 and remained so in 2014. The distribution was similar for stenting, although it was also relatively common in the southwest in the early part of the study period.
In the beginning of the study, the increased rate of stenting might have contributed to the decline in endarterectomy, but there are several other factors that are implicated in the observed trends, according to Dr. Lal. He suggested that decreasing reimbursement for the performance of these procedures, better clinical management of risk factors, and advances in medical therapy. He cited a physician survey that showed a growing preference for medical management over invasive procedures in patients with high-grade stenosis and indicated that this last factor might be a particularly important driver of the decline in revascularization referrals for asymptomatic carotid disease.
The degree to which these Medicare data are representative of overall trends in the United States is unclear, but Dr. Lal called for further work to understand the forces that these data suggest are driving the changing patterns of carotid revascularization.
REPORTING FROM VEITHSYMPOSIUM
Key clinical point:
Major finding: During 1999-2014, the rate of carotid endarterectomy per 100,000 beneficiaries fell from 291 to 128 (57%; P less than .001).
Study details: Retrospective database review.
Disclosures: Dr. Lal reported having no financial conflicts relevant to the study.