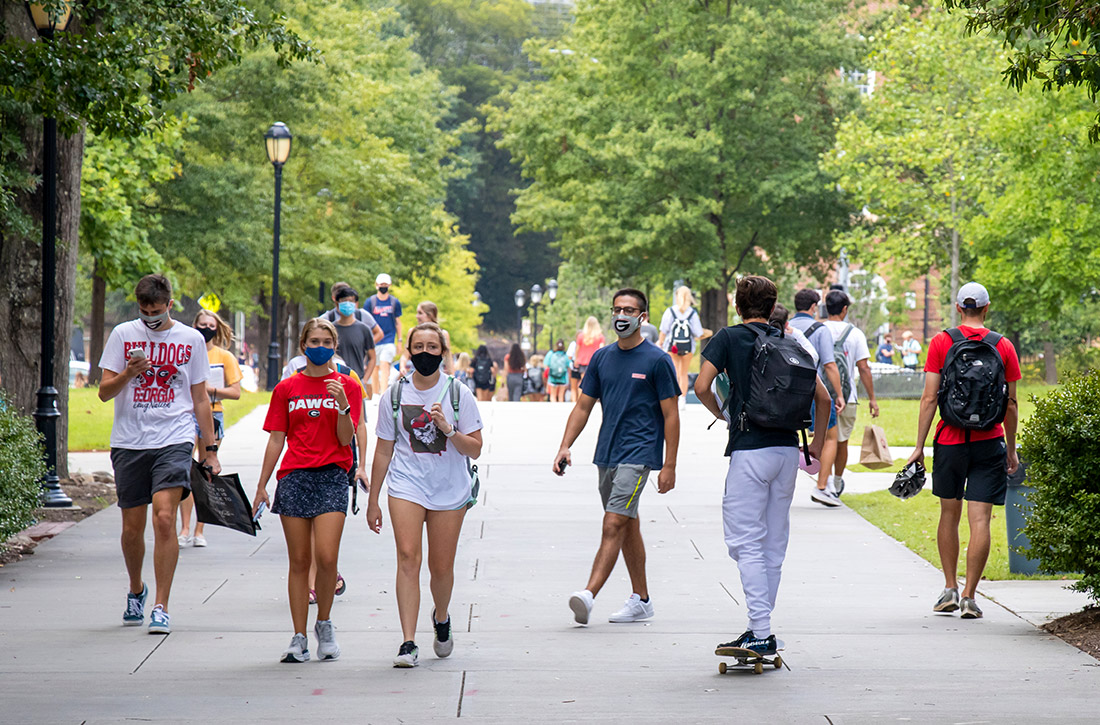User login
A high proportion of SARS-CoV-2–infected university students are asymptomatic
Many individuals infected with SARS-CoV-2 never become symptomatic. In a South Korean study, these infected individuals remained asymptomatic for a prolonged period while maintaining the same viral load as symptomatic patients, suggesting that they are just as infectious.1 A narrative review found high rates of asymptomatic disease in several younger populations, including women in an obstetric ward (88%), the crew of an aircraft carrier (58%), and prisoners (96%).2 However, there is no published research on the percentage of university students who are asymptomatic.
Methods
The University of Georgia (UGA) began classes on August 20, 2020. Shortly before the beginning of classes, UGA implemented a surveillance program for asymptomatic students, faculty, and staff, testing 300 to 450 people per day. Initially, during Weeks 1 and 2 of data collection, anyone could choose to be tested. In Weeks 3 and 4, students, faculty, and staff were randomly invited to participate.
Over the 4-week period beginning on August 17, we calculated the percent of positive cases in surveillance testing and applied this percentage to the entire UGA student population (n = 38,920) to estimate the total number of asymptomatic COVID-19 students each week.3 Data for symptomatic cases were also reported by the university on a weekly basis. This included positive tests from the University Health Center, as well as voluntary reporting using a smartphone app from other sites.
Positive tests in symptomatic individuals were not stratified by student vs nonstudent until Week 3; students comprised 95% of positive symptomatic reports in Week 3 and 99% in Week 4, so we conservatively estimated that 95% of symptomatic cases in Weeks 1 and 2 were students. These data were used to estimate the percentage of SARS-CoV-2–positive students who were asymptomatic.
Results
Our results are summarized in the table. The percentage of asymptomatic students testing positive in surveillance testing was 3.4% in Week 1 and rose steadily to 9% by Week 4. We estimated that there were 1303 asymptomatic cases among students in Week 1, increasing to 3487 asymptomatic positive students on campus by Week 4. The estimated percentage of asymptomatic students infected with SARS-CoV-2 ranged from 73% to 92.5% by week and was 81.1% overall.

Discussion
During the reporting period from August 17 to September 13, the 7-day moving average of new cases in Clarke County (home of UGA) increased from 30 to 83 per 100,000 persons/day (https://dph.georgia.gov/covid-19-daily-status-report). During this period, there were large increases in the number of infected students, more than 80% of whom were asymptomatic. With the assumption that anyone could be infected even if asymptomatic, these numbers highlight the importance for infection control to prevent potential spread within a community by taking universal precautions such as wearing a mask, following physical distancing guidelines, and handwashing.
Limitations. First, reporting of positive tests in symptomatic individuals is highly encouraged but not required. The large drop in symptomatic positive test reports between Weeks 3 and 4, with no change in test positivity in surveillance of asymptomatic students (8.9% vs 9%), suggests that students may have chosen to be tested elsewhere in conjunction with evaluation of their symptoms and/or not reported positive tests, possibly to avoid mandatory isolation and other restrictions on their activities. Further evidence to support no change in actual infection rates comes from testing for virus in wastewater, which also remained unchanged.4
Continue to: Second, each week's surveillance...
Second, each week’s surveillance population is not a true random sample, so extrapolating this estimate to the full student population could over- or undercount asymptomatic cases depending on the direction of bias (ie, healthy volunteer bias vs test avoidance by those with high-risk behaviors).
Finally, some students who were positive in surveillance testing may have been presymptomatic, rather than asymptomatic.
In conclusion, we estimate that approximately 80% of students infected with SARS-CoV-2 are asymptomatic. This is consistent with other studies in young adult populations.2
Mark H. Ebell, MD, MS
Cassie Chupp, MPH
Michelle Bentivegna, MPH
Department of Epidemiology and Biostatistics, College of Public Health, University of Georgia, Athens
ebell@uga.edu
The authors reported no potential conflict of interest relevant to this article.
1. Lee S, Kim T, Lee E, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 infection in a community treatment center in the Republic of Korea [published online ahead of print August 6, 2020]. JAMA Intern Med. doi:10.1001/jamainternmed.2020.3862
2. Oran DP, Topol EJ. Prevalence of asymptomatic SARS-CoV-2 infection : a narrative review. Ann Intern Med. 2020;173:362-367.
3. UGA by the Numbers. University of Georgia Web site. www.uga.edu/facts.php. Updated August 2020. Accessed October 20, 2020.
4. Lott M, Norfolk W, Robertson M, et al. Wastewater surveillance for SARS-CoV-2 in Athens, GA. COVID-19 Portal: Center for the Ecology of Infectious Diseases, University of Georgia Web site. www.covid19.uga.edu/wastewater-athens.html. Updated October 15, 2020. Accessed October 20, 2020.
Many individuals infected with SARS-CoV-2 never become symptomatic. In a South Korean study, these infected individuals remained asymptomatic for a prolonged period while maintaining the same viral load as symptomatic patients, suggesting that they are just as infectious.1 A narrative review found high rates of asymptomatic disease in several younger populations, including women in an obstetric ward (88%), the crew of an aircraft carrier (58%), and prisoners (96%).2 However, there is no published research on the percentage of university students who are asymptomatic.
Methods
The University of Georgia (UGA) began classes on August 20, 2020. Shortly before the beginning of classes, UGA implemented a surveillance program for asymptomatic students, faculty, and staff, testing 300 to 450 people per day. Initially, during Weeks 1 and 2 of data collection, anyone could choose to be tested. In Weeks 3 and 4, students, faculty, and staff were randomly invited to participate.
Over the 4-week period beginning on August 17, we calculated the percent of positive cases in surveillance testing and applied this percentage to the entire UGA student population (n = 38,920) to estimate the total number of asymptomatic COVID-19 students each week.3 Data for symptomatic cases were also reported by the university on a weekly basis. This included positive tests from the University Health Center, as well as voluntary reporting using a smartphone app from other sites.
Positive tests in symptomatic individuals were not stratified by student vs nonstudent until Week 3; students comprised 95% of positive symptomatic reports in Week 3 and 99% in Week 4, so we conservatively estimated that 95% of symptomatic cases in Weeks 1 and 2 were students. These data were used to estimate the percentage of SARS-CoV-2–positive students who were asymptomatic.
Results
Our results are summarized in the table. The percentage of asymptomatic students testing positive in surveillance testing was 3.4% in Week 1 and rose steadily to 9% by Week 4. We estimated that there were 1303 asymptomatic cases among students in Week 1, increasing to 3487 asymptomatic positive students on campus by Week 4. The estimated percentage of asymptomatic students infected with SARS-CoV-2 ranged from 73% to 92.5% by week and was 81.1% overall.

Discussion
During the reporting period from August 17 to September 13, the 7-day moving average of new cases in Clarke County (home of UGA) increased from 30 to 83 per 100,000 persons/day (https://dph.georgia.gov/covid-19-daily-status-report). During this period, there were large increases in the number of infected students, more than 80% of whom were asymptomatic. With the assumption that anyone could be infected even if asymptomatic, these numbers highlight the importance for infection control to prevent potential spread within a community by taking universal precautions such as wearing a mask, following physical distancing guidelines, and handwashing.
Limitations. First, reporting of positive tests in symptomatic individuals is highly encouraged but not required. The large drop in symptomatic positive test reports between Weeks 3 and 4, with no change in test positivity in surveillance of asymptomatic students (8.9% vs 9%), suggests that students may have chosen to be tested elsewhere in conjunction with evaluation of their symptoms and/or not reported positive tests, possibly to avoid mandatory isolation and other restrictions on their activities. Further evidence to support no change in actual infection rates comes from testing for virus in wastewater, which also remained unchanged.4
Continue to: Second, each week's surveillance...
Second, each week’s surveillance population is not a true random sample, so extrapolating this estimate to the full student population could over- or undercount asymptomatic cases depending on the direction of bias (ie, healthy volunteer bias vs test avoidance by those with high-risk behaviors).
Finally, some students who were positive in surveillance testing may have been presymptomatic, rather than asymptomatic.
In conclusion, we estimate that approximately 80% of students infected with SARS-CoV-2 are asymptomatic. This is consistent with other studies in young adult populations.2
Mark H. Ebell, MD, MS
Cassie Chupp, MPH
Michelle Bentivegna, MPH
Department of Epidemiology and Biostatistics, College of Public Health, University of Georgia, Athens
ebell@uga.edu
The authors reported no potential conflict of interest relevant to this article.
Many individuals infected with SARS-CoV-2 never become symptomatic. In a South Korean study, these infected individuals remained asymptomatic for a prolonged period while maintaining the same viral load as symptomatic patients, suggesting that they are just as infectious.1 A narrative review found high rates of asymptomatic disease in several younger populations, including women in an obstetric ward (88%), the crew of an aircraft carrier (58%), and prisoners (96%).2 However, there is no published research on the percentage of university students who are asymptomatic.
Methods
The University of Georgia (UGA) began classes on August 20, 2020. Shortly before the beginning of classes, UGA implemented a surveillance program for asymptomatic students, faculty, and staff, testing 300 to 450 people per day. Initially, during Weeks 1 and 2 of data collection, anyone could choose to be tested. In Weeks 3 and 4, students, faculty, and staff were randomly invited to participate.
Over the 4-week period beginning on August 17, we calculated the percent of positive cases in surveillance testing and applied this percentage to the entire UGA student population (n = 38,920) to estimate the total number of asymptomatic COVID-19 students each week.3 Data for symptomatic cases were also reported by the university on a weekly basis. This included positive tests from the University Health Center, as well as voluntary reporting using a smartphone app from other sites.
Positive tests in symptomatic individuals were not stratified by student vs nonstudent until Week 3; students comprised 95% of positive symptomatic reports in Week 3 and 99% in Week 4, so we conservatively estimated that 95% of symptomatic cases in Weeks 1 and 2 were students. These data were used to estimate the percentage of SARS-CoV-2–positive students who were asymptomatic.
Results
Our results are summarized in the table. The percentage of asymptomatic students testing positive in surveillance testing was 3.4% in Week 1 and rose steadily to 9% by Week 4. We estimated that there were 1303 asymptomatic cases among students in Week 1, increasing to 3487 asymptomatic positive students on campus by Week 4. The estimated percentage of asymptomatic students infected with SARS-CoV-2 ranged from 73% to 92.5% by week and was 81.1% overall.

Discussion
During the reporting period from August 17 to September 13, the 7-day moving average of new cases in Clarke County (home of UGA) increased from 30 to 83 per 100,000 persons/day (https://dph.georgia.gov/covid-19-daily-status-report). During this period, there were large increases in the number of infected students, more than 80% of whom were asymptomatic. With the assumption that anyone could be infected even if asymptomatic, these numbers highlight the importance for infection control to prevent potential spread within a community by taking universal precautions such as wearing a mask, following physical distancing guidelines, and handwashing.
Limitations. First, reporting of positive tests in symptomatic individuals is highly encouraged but not required. The large drop in symptomatic positive test reports between Weeks 3 and 4, with no change in test positivity in surveillance of asymptomatic students (8.9% vs 9%), suggests that students may have chosen to be tested elsewhere in conjunction with evaluation of their symptoms and/or not reported positive tests, possibly to avoid mandatory isolation and other restrictions on their activities. Further evidence to support no change in actual infection rates comes from testing for virus in wastewater, which also remained unchanged.4
Continue to: Second, each week's surveillance...
Second, each week’s surveillance population is not a true random sample, so extrapolating this estimate to the full student population could over- or undercount asymptomatic cases depending on the direction of bias (ie, healthy volunteer bias vs test avoidance by those with high-risk behaviors).
Finally, some students who were positive in surveillance testing may have been presymptomatic, rather than asymptomatic.
In conclusion, we estimate that approximately 80% of students infected with SARS-CoV-2 are asymptomatic. This is consistent with other studies in young adult populations.2
Mark H. Ebell, MD, MS
Cassie Chupp, MPH
Michelle Bentivegna, MPH
Department of Epidemiology and Biostatistics, College of Public Health, University of Georgia, Athens
ebell@uga.edu
The authors reported no potential conflict of interest relevant to this article.
1. Lee S, Kim T, Lee E, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 infection in a community treatment center in the Republic of Korea [published online ahead of print August 6, 2020]. JAMA Intern Med. doi:10.1001/jamainternmed.2020.3862
2. Oran DP, Topol EJ. Prevalence of asymptomatic SARS-CoV-2 infection : a narrative review. Ann Intern Med. 2020;173:362-367.
3. UGA by the Numbers. University of Georgia Web site. www.uga.edu/facts.php. Updated August 2020. Accessed October 20, 2020.
4. Lott M, Norfolk W, Robertson M, et al. Wastewater surveillance for SARS-CoV-2 in Athens, GA. COVID-19 Portal: Center for the Ecology of Infectious Diseases, University of Georgia Web site. www.covid19.uga.edu/wastewater-athens.html. Updated October 15, 2020. Accessed October 20, 2020.
1. Lee S, Kim T, Lee E, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 infection in a community treatment center in the Republic of Korea [published online ahead of print August 6, 2020]. JAMA Intern Med. doi:10.1001/jamainternmed.2020.3862
2. Oran DP, Topol EJ. Prevalence of asymptomatic SARS-CoV-2 infection : a narrative review. Ann Intern Med. 2020;173:362-367.
3. UGA by the Numbers. University of Georgia Web site. www.uga.edu/facts.php. Updated August 2020. Accessed October 20, 2020.
4. Lott M, Norfolk W, Robertson M, et al. Wastewater surveillance for SARS-CoV-2 in Athens, GA. COVID-19 Portal: Center for the Ecology of Infectious Diseases, University of Georgia Web site. www.covid19.uga.edu/wastewater-athens.html. Updated October 15, 2020. Accessed October 20, 2020.
The Effect of Radium-223 Therapy in Agent Orange-Related Prostate Carcinoma
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
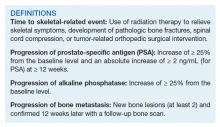
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
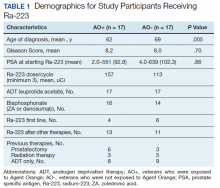
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
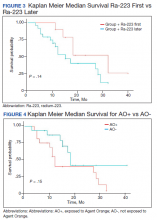
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
Patients with metastatic castrate resistant prostate carcinoma (CRPC) have several treatment options, including radium-223 dichloride (Ra-223) radionuclide therapy, abiraterone, enzalutamide, and cabazitaxel. Ra-223 therapy has been reported to increase median survival in patients with bone metastatic prostate carcinoma.1,2 However, ERA 223 trial data showed an increase of bone fractures with combination of Ra-223 and abiraterone.3
Agent Orange (AO) exposure has been studied as a potential risk factor for development of prostate carcinoma. AO was a commercially manufactured defoliate that was sprayed extensively during the Vietnam War. Due to a side product of chemical manufacturing, AO was contaminated with the toxin 2,3,7,8-tetrachlorodibenzo-p-dioxin, a putative carcinogen. These dioxins can enter the food chain through soil contamination. There is enough evidence to link AO to hematologic malignancies and several solid tumors, including prostate carcinoma.4 Although no real estimates exist for what percentage of Vietnam veterans experienced AO exposure, Surveillance, Epidemiology, and End Results data showed that about 3 million veterans served in Southeast Asia where AO was used extensively in the combat theater. AO has been reported to be positively associated with a 52% increase in risk of prostate carcinoma detection at initial prostate biopsy.5
There has been no reported study of treatment efficacy in veterans with AO-related prostate carcinoma. We present a retrospective study of Ra-223 and other therapies in metastatic CRPC. The purpose of this study was to compare response to therapy and survival in veterans exposed to agent orange (AO+) vs veterans who were not exposed to (AO-) in a single US Department of Veteran Affairs (VA) medical center.
Methods
This was a retrospective study of veterans with metastatic CRPC to bones who received Ra-223 radionuclide therapy with standard dose of 50 kBq per kg of body weight and other sequential therapies at VA Pittsburgh Healthcare System (VAPHS) from January 2014 to January 2019. The purpose of this study was to measure difference in treatment outcome between AO+ veterans and AO- veterans.
Eligibility Criteria
All veterans had a history that included bone metastasis CRPC. They could have 2 to 3 small lymphadenopathies but not visceral metastasis. They received a minimum of 3 cycles and a maximum of 6 cycles of Ra-223 therapy, which was given in 4-week intervals. Pretreatment criteria was hemoglobin > 10 g/dL, platelet > 100
Statistics
Time to study was calculated from the initiation of Ra-223 therapy. Time to skeletal-related events (SRE), progression of prostate specific antigen (PSA), bone metastasis, and alkaline phosphatase (ALP) were calculated in months, using unpaired t test with 2-tailed P value. Median survival was calculated in months by Kaplan Meier R log-rank test Definition).
Results
Forty-eight veterans with bone metastasis CRPC received Ra-223 therapy. Of those, 34 veterans were eligible for this retrospective study: 17 AO+ veterans and 17 AO- veterans. Mean age of diagnosis was 62 years (AO+) and 69 years (AO-) (P = .005). Mean Gleason score was 8.2 (AO+) and 8.0 (AO-) (P = .705). Veterans received initial therapy at diagnosis of prostate carcinoma, including radical prostatectomy (6 AO+ and 3 AO-), localized radiation therapy (3 AO+ and 5 AO-), and ADT (8 AO+ and 9 AO-) (Table 1).
Mean PSA at the initiation of Ra-223 therapy for AO+ was 92.8 (range, 2-551) and for AO- was 102.3 (range, 4-639; P = .86). Mean Ra-223 dose per cycle for AO+ and AO- was 157 uCi and 113 uCi, respectively. All 34 veterans received ADT (leuprolide acetate), and 30 veterans (16 AO+ and 14 AO-) received bisphosphonates (zoledronic acid or denosumab). A total of 10 veterans (29%) received Ra-223 as a first-line therapy (4 AO+ and 6 AO-), and 24 veterans (71%) received Ra-223 after hormonal or chemotherapy (13 AO+ and 11 AO-).
There were 12 SRE (8 AO+ and 4 AO-). Mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Three veterans received concurrent Ra-223 and abiraterone (participated in ERA 223 trial). Two AO+ veterans experienced SRE at 7 months and 11 months, respectively. Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months, respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months, respectively (P = .05). (Table 2). The treatment pattern of AO+ and AO- is depicted on a swimmer plot (Figures 1 and 2).
Twenty veterans (58%) had died: 13 AO+ and 7 AO- veterans. Median survival for Ra-223 first and Ra-223 later was was 32 months and 15 months, respectively (P = .14; hazard ratio [HR], 0.48). Overall median survival for AO+ veterans and AO- veterans were 12 months and 18 months, respectively (P = .15; HR, 2.0) (Figures 3 and 4).
Discussions
There has been no reported VA study of using Ra-223 and other therapies (hormonal and chemotherapy) in veterans exposed to AO. This is the first retrospective study to compare the response and survival between AO+ and AO- veterans. Even though this study featured a small sample, it is interesting to note the difference between those 2 populations. There was 1 prior study in veterans with prostate carcinoma using radiotherapy (brachytherapy) in early-stage disease. Everly and colleagues reported that AO+ veterans were less likely to remain biochemically controlled compared with AO- and nonveteran patients with prostate carcinoma.4
Ansbaugh and colleagues reported that AO was associated with a 75% increase in the risk of Gleason ≥ 7 and a 110% increase in Gleason ≥ 8. AO+ veterans are at risk for the detection of high-grade prostate carcinoma. They also tend to have an average age of diagnosis that is 4 to 5 years younger than AO- veterans.6
Our study revealed that AO+ veterans were diagnosed at a younger age (mean 62 years) compared with that of AO- veterans (mean 69 years, P = .005). We also proved that AO veterans have a higher mean Gleason score (8.2) compared with that of AO- veterans (8.0). Veterans received therapy at the time of diagnosis of prostate carcinoma with either radical prostatectomy, radiation therapy, or ADT with leuprolide acetate. Mean PSA at the start of Ra-223 therapy for AO+ was 92.8 (range, 2-551); for AO- was 102.3 (range, 4-639), which is not statistically significant.
Ra-223, an
In a phase 3, randomized, double-blind, placebo-controlled study by Parker and colleagues (ALSYMPCA study), 921 patients who had received, were not eligible to receive, or declined docetaxel, in a 2:1 ratio, were randomized to receive 6 injections of Ra-223 or matching placebo.2 Ra-223 significantly improved overall survival (OS) (median, 14.9 months vs 11.3 months) compared with that of placebo. Ra-223 also prolonged the time to the first symptomatic SRE (median, 15.6 months vs 9.8 months), the time to an increase in the total ALP level (median 7.4 months vs 3.8 months), and the time to an increase in the PSA level (median 3.6 months vs 3.4 months).2
In our study, the mean time to SRE for AO+ was 10.6 months and AO- was 10.3 months (P = .93). Mean time to PSA progression for AO+ was 5.4 months and for AO- was 6.8 months (P = .28). Mean time to bone progression for AO+ and for AO- were 7.6 months and 10.1 months respectively (P = .16). Mean time to ALP progression for AO+ and AO- were 6.3 months and 8.7 months respectively (P = .05). There is a trend of shorter PSA progression, bone progression, and ALP progression in AO+ veterans, though these were not statistically significant due to small sample population. In our study the median survival in for AO- was 18 months and for AO+ was 12 months, which is comparable with median survival of 14.9 months from the ALSYMPCA study.
There were 12 veterans who developed SREs. All received radiation therapy due to bone progression or impending fracture. AO+ veterans developed more SREs (n = 8) when compared with AO- veterans (n = 4). There were more AO- veterans alive (n = 10) than there were AO+ veterans (n = 4). The plausible explanation of this may be due to the aggressive pattern of prostate carcinoma in AO+ veterans (younger age and higher Gleason score).
VAPHS participated in the ERA trial between 2014 and 2016. The trial enrolled 806 patients who were randomly assigned to receive first-line Ra-223 or placebo in addition to abiraterone acetate plus prednisone.3 The study was unblinded prematurely after more fractures and deaths were noted in the Ra-223 and abiraterone group than there were in the placebo and abiraterone group. Median symptomatic SRE was 22.3 months in the Ra-223 group and 26.0 months in the placebo group. Fractures (any grade) occurred in 29% in the Ra-223 group and 11% in the placebo group. It was suggested that Ra-223 could contribute to the risk of osteoporotic fractures in patients with bone metastatic prostate carcinoma. Median OS was 30.7 months in the Ra-223 group and 33.3 months in the placebo group.
We enrolled 3 veterans in the ERA clinical trial. Two AO+ veterans had SREs at 7 months and 11 months. In our study, the median OS for Ra-223 first line was 32 months, which is comparable with median survival of 30.7 months from ERA-223 study. Median survival for Ra-223 later was only 15 months. We recommend veterans with at least 2 to 3-bone metastasis receive Ra-223 in the first-line setting rather than second- or third-line setting. In this retrospective study with Ra-223 and other therapies, we proved that AO+ veterans have a worse response and OS when compared with that of AO- veterans.
Conclusions
This is the first VA study to compare the efficacy of Ra-223 and other therapies in metastatic CRPC between AO+ and AO- veterans. AO+ veterans were diagnosed at a younger age and had higher Gleason scores. There was no statistical difference between AO+ and AO- veterans in terms of time to SRE, PSA progression, and bone and ALP progression even though there was a trend of shorter duration in AO+ veterans. There was no median survival difference between veterans who received Ra-223 first vs Ra-223 later as well as between AO+ and AO- veterans, but there was a trend of worse survival in veteran who received Ra-223 later and AO+ veterans.
This study showed that AO+ veterans have a shorter duration of response to therapy and shorter median survival compared with that of AO- veterans. We recommend that veterans should get Ra-223 in the first-line setting rather than after hormonal therapy and chemotherapy because their marrows are still intact. We need to investigate further whether veterans that exposed to carcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) may have different molecular biology and as such may cause inferior efficacy in the treatment of prostate carcinoma.
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
1. Shore ND. Radium-223 dichloride for metastatic castration-resistant prostate cancer: the urologist’s perspective. Urology. 2015;85(4):717-724. doi:10.1016/j.urology.2014.11.031
2. Parker C, Nilsson S, Heinrich D, et al; ALSYMPCA Investigators. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369(3):213-223. doi:10.1056/NEJMoa1213755
3. Smith M, Parker C, Saad F, et al. Addition of radium-223 to abiraterone acetate and prednisone or prednisolone in patients with castration-resistant prostate cancer and bone metastases (ERA 223): a randomized, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet Oncol. 2019 Oct;20(10):e559]. Lancet Oncol. 2019;20(3):408-419. doi:10.1016/S1470-2045(18)30860-X
4. Everly L, Merrick GS, Allen ZA, et al. Prostate cancer control and survival in Vietnam veterans exposed to Agent Orange. Brachytherapy. 2009;8(1):57-62. doi: 10.1016/j.brachy.2008.08.001
5. Altekruse S. SEER Cancer Statistics Review, 1975-2017 Bethesda, MD: National Cancer Institute. 2009. 6. Ansbaugh N, Shannon J, Mori M, Farris PE, Garzotto M. Agent Orange as a risk factor for high-grade prostate cancer. Cancer. 2013;119(13):2399-2404. doi:10.1002/cncr.27941
7. Jadvar H, Quinn DI. Targeted α-particle therapy of bone metastases in prostate cancer. Clin Nucl Med. 2013;38(12):966-971. doi:10.1097/RLU.0000000000000290
Guideline Concordance with Durvalumab in Unresectable Stage III Non-Small Cell Lung Cancer: A Single Center Veterans Hospital Experience
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
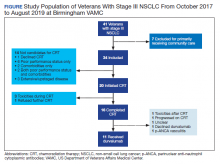
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
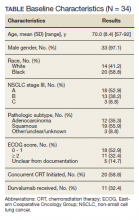
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
COVID-19 and Venous Thromboembolism Pharmacologic Thromboprophylaxis
The novel coronavirus SARS-CoV-2 and resulting viral syndrome (COVID-19) was first reported in China during December 2019 and within weeks emerged in the US.1 Since it is a rapidly evolving situation, clinicians must remain current on best practices—a challenging institutional responsibility. According to LitCovid, a curated literature hub for tracking scientific information on COVID-19, there are > 54,000 articles on the subject in PubMed. Among these include venous thromboembolism (VTE) prophylaxis guidance from 4 respected thrombosis organizations/societies and the US National Institutes of Health.1-5
Observations
COVID-19 predisposes patients with and without a history of cardiovascular disease to thrombotic complications, occurring in either the venous or arterial circulation system.2,6 Early observational studies suggest that thrombotic rates may be in excess of 20 to 30%; however, the use of prophylactic anticoagulation was inconsistent among studies that were rushed to publication.6
Autopsy data have demonstrated the presence of fibrin thrombi within distended small vessels and capillaries and extensive extracellular fibrin deposition.6 Investigators compared the characteristics of acute pulmonary embolism in 23 cases with COVID-19 but with no clinical signs of deep vein thrombosis with 100 controls without COVID-19.7 They observed that thrombotic lesions had a greater distribution in peripheral lung segments (ie, peripheral arteries) and were less extensive for those with COVID-19 vs without COVID-19 infection. Thus, experts currently hypothesize that COVID-19 has a distinct “pathomechanism.” As a unique phenotype, thrombotic events represent a combination of thromboembolic disease influenced by components of the Virchow triad (eg, acute illness and immobility) and in situ immunothrombosis, a local inflammatory response.6,7
Well-established surgical and nonsurgical VTE thromboprophylaxis guidelines serve as the foundation for current COVID-19 thromboprophylaxis guidance.8,9 Condition specific guidance is extrapolated from small, retrospective observational studies or based on expert opinion, representing levels 2 and 3 evidence, respectively.1-5 Table 1 captures similarities and differences among COVID-19 VTE thromboprophylaxis recommendations which vary by time to publication and by society member expertise gained from practice in the field.
Three thrombosis societies recommend universal pharmacologic thromboprophylaxis for acutely ill COVID-19 patients who lack contraindications.3-5 Others recommend use of risk stratification scoring tools, such as the Padua risk assessment model (RAM) for medical patients or Caprini RAM for surgical patients, the disseminated intravascular coagulation (DIC) score, or the sepsis-induced coagulopathy score to determine therapeutic appropriateness (Tables 2 and 3).1,2 Since most patients hospitalized for COVID-19 will present with a pathognomonic pneumonia and an oxygen requirement, they will generally achieve a score of ≥ 4 when the Padua RAM is applied; thus, representing a clear indication for pharmacologic thromboprophylaxis.8,9 If the patient is pregnant, the Anticoagulation Forum recommends pharmacologic prophylaxis, consultation with an obstetrician, and use of obstetrical thromboprophylaxis guidelines.3,10,11
Most thrombosis experts prefer parenteral thromboprophylaxis, specifically low molecular-weight heparin (LMWH) or fondaparinux, for inpatients over use of direct oral anticoagulants (DOACs) in order to minimize the potential for drug interactions particularly when investigational antivirals are in use.4 Once-daily agents (eg, rivaroxaban, fondaparinux, and enoxaparin) are preferred over multiple daily doses to minimize staff contact with patients infected with COVID-19.4,5 Fondaparinux and DOACs should preferentially be used in patients with a recent history of heparin-induced thrombocytopenia with and without thrombosis (HIT/HITTS). Subcutaneous heparin is reserved for patients who are scheduled for invasive procedures or have reduced renal function (eg, creatinine clearance < 30 mL/min).1,3-5 In line with existing pharmacologic thromboprophylaxis guidance, standard prophylactic LMWH doses are recommended unless patients are obese (body mass index [BMI] > 30) or morbidly obese (BMI > 40) necessitating selection of intermediate doses.4
Since early non-US studies demonstrated high thrombotic risk without signaling a potential for harm from pharmacologic thromboprophylaxis, some organizations recommend empiric escalation of anticoagulation doses for critical illness.3,4,6 Thus, it may be reasonable to advance to either intermediate pharmacologic thromboprophylaxis dosing or therapeutic doses.3 However, observational studies question this aggressive practice unless a clear indication exists for intensification (ie, atrial fibrillation, known VTE).
A large multi-institutional registry study that included 400 subjects from 5 centers demonstrated a radiographically confirmed VTE rate of 4.8% and an arterial thrombosis rate of 2.8%.6 When limiting to the critically ill setting, VTE and arterial thrombosis occurred at slightly higher rates (7.6% and 5.6%, respectively). Patients also were at risk for nonvessel thrombotic complications (eg, CVVH circuit, central venous catheters, and arterial lines). Subsequently, the overall thrombotic complication rate was 9.5%. All thrombotic events except one arose in patients who were receiving standard doses of pharmacologic thromboprophylaxis. Unfortunately, D-dimer elevation at admission was not only predictive of thrombosis and death, but portended bleeding. The overall bleeding rate was 4.8%, with a major bleeding rate of 2.3%. In the context of observing thromboses at normally expected rates during critical illness in association with a significant bleeding risk, the authors recommended further investigation into the net clinical benefit.
Similarly, a National Institutes of Health funded, observational, single center US study evaluated 4,389 inpatients infected with COVID-19 and determined that therapeutic and prophylactic anticoagulation reduced inpatient mortality (adjusted hazard ratio [aHR], 0.53 and 0.50, respectively for the primary outcome) and intubation (aHR, 0.69 and 0.72, respectively) over no anticoagulation.12 Notably, use of inpatient therapeutic anticoagulation commonly represented a continuation of preadmission therapy or progressive COVID-19. A subanalysis demonstrated that timely use (eg, within 48 hours of admission) of prophylactic or therapeutic anticoagulation, resulted in no difference (P < .08) in the primary outcome. Bleeding rates were low overall: 3%, 1.7%, and 1.9% for therapeutic, prophylactic, and no anticoagulation groups, respectively. Furthermore, selection of DOACs seems to be associated with lower bleeding rates when compared with that of LMWH heparin (1.3% vs 2.6%, respectively). In those where site of bleeding could be ascertained, the most common sites were the gastrointestinal tract (50.7%) followed by mucocutaneous (19.4%), bronchopulmonary (14.9%), and intracranial (6%). In summary, prophylactic thromboprophylaxis doses seem to be associated with positive net clinical benefit.
As of October 30, 2020, the US Department of Veterans Affairs (VA) had reported 75,156 COVID-19 cases and 3,961 deaths.13 Since the VA Pharmacy Benefits Management (PBM) does not disseminate nationally prepared anticoagulation order sets to the field, facility anticoagulation leads should be encouraged to develop local guidance-based policies to help standardize care and minimize further variations in practice, which would likely lack evidential support. Per the VA Tennessee Valley Healthcare System (TVHS)- Nashville/Murfreesboro anticoagulation policy, we limit the ordering of parenteral anticoagulation to Computerized Patient Record System (CPRS) order sets in order to provide decision support (eFigure 1, available at doi:10.12788/fp.0063). Other facilities have shown that embedded clinical decision support tools increase adherence to guideline VTE prophylaxis recommendations within the VA.14
In April 2020, the TVHS anticoagulation clinical pharmacy leads developed a COVID-19 specific order set based on review of societal guidance and the evolving, supportive literature summarized in this review with consideration of provider familiarity (eFigure 2, available at doi:10.12788/fp.0063)). Between April and June 2020, the COVID-19 order set content consistently evolved with publication of each COVID-19 thromboprophylaxis guideline.1-5
Since TVHS is a high-complexity facility, we elected to use universal pharmacologic thromboprophylaxis for patients with COVID-19. This construct bypasses the use of scoring tools (eg, RAM), although we use Padua and Caprini RAMS for medical and surgical patients, respectively, who are not diagnosed with COVID-19. The order set displays all acceptable guideline recommended options, delineated by location of care (eg, medical ward vs intensive care unit), prior history of heparin-induced thrombocytopenia, and renal function. Subsequently, all potential agents, doses, and dosing interval options are offered so that the provider autonomously determines how to individualize the clinical care. Since TVHS has only diagnosed 932 ambulatory/inpatient COVID-19 cases combined, our plans are to complete a future observational analysis to determine the effectiveness of the inpatient COVID-19 thromboprophylaxis order set for our internal customers.
Conclusions
The COVID-19 pandemic has resulted in arguably the most challenging medical climate in the evidence-based medicine era. Until high-quality randomized controlled trials are published, the medical community is, in a sense, operating within a crucible of crisis having to navigate therapeutic policy with little certainty. This principle holds true for thromboprophylaxis in patients with COVID-19 despite the numerous advancements in this field over the past decade.
A review of societal guidance shows there is universal agreement with regards to supporting standard doses of pharmacologicalprophylaxis in acutely ill patients either when universally applied or guided by a RAM as well as the use of universal thromboprophylaxis in critically ill patients. All societies discourage the use of antiplatelet therapy for arterial thrombosis prevention and advocate for mechanical compression in patients with contraindications to pharmacologic anticoagulation. Beyond this, divergence between guidance statements begins to appear. For example, societies do not currently agree on the role and approach for extended pharmacologic prophylaxis postdischarge. The differences between societal guidance speaks to the degree of uncertainty among leading experts, which is considered to be the logical outworking of the current level of evidence. Regardless, these guidance documents should be considered the best resource currently available.
The medical community is fortunate to have robust societies that have published guidance on thromboprophylaxis in patients with COVID-19. The novelty of COVID-19 precludes these societal guidance publications from being based on high-quality evidence, but at the very least, they provide insight into how leading experts in the field of thrombosis and hemostasis are currently navigating the therapeutic landscape.
While this paper provides a summary of the current guidance, evidence is evolving at an unprecedented pace. Facilities and anticoagulation leads should be actively and frequently evaluating literature and guidance to ensure their practices and policies remain current.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville/Murfreesboro.
1. National Institutes of Health. Coronavirus disease 2019 (COVID-19) treatment guidelines. https://www.covid19treatmentguidelines.nih.gov/whats-new/. Updated October 9, 2020. Accessed October 15, 2020.
2. Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(23):2950-2973. doi:10.1016/j.jacc.2020.04.031
3. Barnes GD, Burnett A, Allen A, et al. Thromboembolism and anticoagulant therapy during the COVID-19 pandemic: interim clinical guidance from the anticoagulation forum. J Thromb Thrombolysis. 2020;50(1):72-81. doi:10.1007/s11239-020-02138-z
4. Spyropoulos AC, Levy JH, Ageno W, et al. Scientific and Standardization Committee communication: Clinical guidance on the diagnosis, prevention, and treatment of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18(8):1859-1865. doi:10.1111/jth.14929
5. Moores LK, Tritschler T, Brosnahan S, et al. Prevention, Diagnosis, and Treatment of VTE in Patients With Coronavirus Disease 2019: CHEST Guideline and Expert Panel Report. Chest. 2020;158(3):1143-1163. doi:10.1016/j.chest.2020.05.559
6. Al-Samkari H, Karp Leaf RS, Dzik WH, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136(4):489-500. doi:10.1182/blood.2020006520.
7. van Dam LF, Kroft LJM, van der Wal LI, et al. Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: a different phenotype of thrombotic disease?. Thromb Res. 2020;193:86-89. doi:10.1016/j.thromres.2020.06.010
8. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e195S-e226S. doi:10.1378/chest.11-2296
9. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines [published correction appears in Chest. 2012 May;141(5):1369]. Chest. 2012;141(suppl 2):e227S-e277S. doi:10.1378/chest.11-2297
10. ACOG Practice Bulletin No. 196 Summary: thromboembolism in pregnancy. Obstet Gynecol. 2018;132(1):243-248. doi:10.1097/AOG.0000000000002707
11. Royal College of Obstetricians and Gynaecologists. Reducing the risk of venous thromboembolism during pregnancy and the puerperium. Green-top Guideline. No. 37a. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-37a.pdf. Published April 2015. Accessed October 15, 2020.
12. Nadkarni GN, Lala A, Bagiella E, et al. Anticoagulation, mortality, bleeding and pathology among patients hospitalized with COVID-19: a single health system study [published online ahead of print, 2020 Aug 24]. J Am Coll Cardiol. 2020;76(16):1815-1826. doi:10.1016/j.jacc.2020.08.041
13. US Department of Veterans Affairs. Department of Veterans Affairs COVID-19 national summary. https://www.accesstocare.va.gov/Healthcare/COVID19NationalSummary. Updated November 1, 2020. Accessed November 1, 2020.
14. George B, Gonzales S, Patel K, Petit S, Franck AJ, Bovio Franck J. Impact of a clinical decision-support tool on venous thromboembolism prophylaxis in acutely ill medical patients. J Pharm Technol. 2020;36(4):141-147. doi:10.1177/8755122520930288
The novel coronavirus SARS-CoV-2 and resulting viral syndrome (COVID-19) was first reported in China during December 2019 and within weeks emerged in the US.1 Since it is a rapidly evolving situation, clinicians must remain current on best practices—a challenging institutional responsibility. According to LitCovid, a curated literature hub for tracking scientific information on COVID-19, there are > 54,000 articles on the subject in PubMed. Among these include venous thromboembolism (VTE) prophylaxis guidance from 4 respected thrombosis organizations/societies and the US National Institutes of Health.1-5
Observations
COVID-19 predisposes patients with and without a history of cardiovascular disease to thrombotic complications, occurring in either the venous or arterial circulation system.2,6 Early observational studies suggest that thrombotic rates may be in excess of 20 to 30%; however, the use of prophylactic anticoagulation was inconsistent among studies that were rushed to publication.6
Autopsy data have demonstrated the presence of fibrin thrombi within distended small vessels and capillaries and extensive extracellular fibrin deposition.6 Investigators compared the characteristics of acute pulmonary embolism in 23 cases with COVID-19 but with no clinical signs of deep vein thrombosis with 100 controls without COVID-19.7 They observed that thrombotic lesions had a greater distribution in peripheral lung segments (ie, peripheral arteries) and were less extensive for those with COVID-19 vs without COVID-19 infection. Thus, experts currently hypothesize that COVID-19 has a distinct “pathomechanism.” As a unique phenotype, thrombotic events represent a combination of thromboembolic disease influenced by components of the Virchow triad (eg, acute illness and immobility) and in situ immunothrombosis, a local inflammatory response.6,7
Well-established surgical and nonsurgical VTE thromboprophylaxis guidelines serve as the foundation for current COVID-19 thromboprophylaxis guidance.8,9 Condition specific guidance is extrapolated from small, retrospective observational studies or based on expert opinion, representing levels 2 and 3 evidence, respectively.1-5 Table 1 captures similarities and differences among COVID-19 VTE thromboprophylaxis recommendations which vary by time to publication and by society member expertise gained from practice in the field.
Three thrombosis societies recommend universal pharmacologic thromboprophylaxis for acutely ill COVID-19 patients who lack contraindications.3-5 Others recommend use of risk stratification scoring tools, such as the Padua risk assessment model (RAM) for medical patients or Caprini RAM for surgical patients, the disseminated intravascular coagulation (DIC) score, or the sepsis-induced coagulopathy score to determine therapeutic appropriateness (Tables 2 and 3).1,2 Since most patients hospitalized for COVID-19 will present with a pathognomonic pneumonia and an oxygen requirement, they will generally achieve a score of ≥ 4 when the Padua RAM is applied; thus, representing a clear indication for pharmacologic thromboprophylaxis.8,9 If the patient is pregnant, the Anticoagulation Forum recommends pharmacologic prophylaxis, consultation with an obstetrician, and use of obstetrical thromboprophylaxis guidelines.3,10,11
Most thrombosis experts prefer parenteral thromboprophylaxis, specifically low molecular-weight heparin (LMWH) or fondaparinux, for inpatients over use of direct oral anticoagulants (DOACs) in order to minimize the potential for drug interactions particularly when investigational antivirals are in use.4 Once-daily agents (eg, rivaroxaban, fondaparinux, and enoxaparin) are preferred over multiple daily doses to minimize staff contact with patients infected with COVID-19.4,5 Fondaparinux and DOACs should preferentially be used in patients with a recent history of heparin-induced thrombocytopenia with and without thrombosis (HIT/HITTS). Subcutaneous heparin is reserved for patients who are scheduled for invasive procedures or have reduced renal function (eg, creatinine clearance < 30 mL/min).1,3-5 In line with existing pharmacologic thromboprophylaxis guidance, standard prophylactic LMWH doses are recommended unless patients are obese (body mass index [BMI] > 30) or morbidly obese (BMI > 40) necessitating selection of intermediate doses.4
Since early non-US studies demonstrated high thrombotic risk without signaling a potential for harm from pharmacologic thromboprophylaxis, some organizations recommend empiric escalation of anticoagulation doses for critical illness.3,4,6 Thus, it may be reasonable to advance to either intermediate pharmacologic thromboprophylaxis dosing or therapeutic doses.3 However, observational studies question this aggressive practice unless a clear indication exists for intensification (ie, atrial fibrillation, known VTE).
A large multi-institutional registry study that included 400 subjects from 5 centers demonstrated a radiographically confirmed VTE rate of 4.8% and an arterial thrombosis rate of 2.8%.6 When limiting to the critically ill setting, VTE and arterial thrombosis occurred at slightly higher rates (7.6% and 5.6%, respectively). Patients also were at risk for nonvessel thrombotic complications (eg, CVVH circuit, central venous catheters, and arterial lines). Subsequently, the overall thrombotic complication rate was 9.5%. All thrombotic events except one arose in patients who were receiving standard doses of pharmacologic thromboprophylaxis. Unfortunately, D-dimer elevation at admission was not only predictive of thrombosis and death, but portended bleeding. The overall bleeding rate was 4.8%, with a major bleeding rate of 2.3%. In the context of observing thromboses at normally expected rates during critical illness in association with a significant bleeding risk, the authors recommended further investigation into the net clinical benefit.
Similarly, a National Institutes of Health funded, observational, single center US study evaluated 4,389 inpatients infected with COVID-19 and determined that therapeutic and prophylactic anticoagulation reduced inpatient mortality (adjusted hazard ratio [aHR], 0.53 and 0.50, respectively for the primary outcome) and intubation (aHR, 0.69 and 0.72, respectively) over no anticoagulation.12 Notably, use of inpatient therapeutic anticoagulation commonly represented a continuation of preadmission therapy or progressive COVID-19. A subanalysis demonstrated that timely use (eg, within 48 hours of admission) of prophylactic or therapeutic anticoagulation, resulted in no difference (P < .08) in the primary outcome. Bleeding rates were low overall: 3%, 1.7%, and 1.9% for therapeutic, prophylactic, and no anticoagulation groups, respectively. Furthermore, selection of DOACs seems to be associated with lower bleeding rates when compared with that of LMWH heparin (1.3% vs 2.6%, respectively). In those where site of bleeding could be ascertained, the most common sites were the gastrointestinal tract (50.7%) followed by mucocutaneous (19.4%), bronchopulmonary (14.9%), and intracranial (6%). In summary, prophylactic thromboprophylaxis doses seem to be associated with positive net clinical benefit.
As of October 30, 2020, the US Department of Veterans Affairs (VA) had reported 75,156 COVID-19 cases and 3,961 deaths.13 Since the VA Pharmacy Benefits Management (PBM) does not disseminate nationally prepared anticoagulation order sets to the field, facility anticoagulation leads should be encouraged to develop local guidance-based policies to help standardize care and minimize further variations in practice, which would likely lack evidential support. Per the VA Tennessee Valley Healthcare System (TVHS)- Nashville/Murfreesboro anticoagulation policy, we limit the ordering of parenteral anticoagulation to Computerized Patient Record System (CPRS) order sets in order to provide decision support (eFigure 1, available at doi:10.12788/fp.0063). Other facilities have shown that embedded clinical decision support tools increase adherence to guideline VTE prophylaxis recommendations within the VA.14
In April 2020, the TVHS anticoagulation clinical pharmacy leads developed a COVID-19 specific order set based on review of societal guidance and the evolving, supportive literature summarized in this review with consideration of provider familiarity (eFigure 2, available at doi:10.12788/fp.0063)). Between April and June 2020, the COVID-19 order set content consistently evolved with publication of each COVID-19 thromboprophylaxis guideline.1-5
Since TVHS is a high-complexity facility, we elected to use universal pharmacologic thromboprophylaxis for patients with COVID-19. This construct bypasses the use of scoring tools (eg, RAM), although we use Padua and Caprini RAMS for medical and surgical patients, respectively, who are not diagnosed with COVID-19. The order set displays all acceptable guideline recommended options, delineated by location of care (eg, medical ward vs intensive care unit), prior history of heparin-induced thrombocytopenia, and renal function. Subsequently, all potential agents, doses, and dosing interval options are offered so that the provider autonomously determines how to individualize the clinical care. Since TVHS has only diagnosed 932 ambulatory/inpatient COVID-19 cases combined, our plans are to complete a future observational analysis to determine the effectiveness of the inpatient COVID-19 thromboprophylaxis order set for our internal customers.
Conclusions
The COVID-19 pandemic has resulted in arguably the most challenging medical climate in the evidence-based medicine era. Until high-quality randomized controlled trials are published, the medical community is, in a sense, operating within a crucible of crisis having to navigate therapeutic policy with little certainty. This principle holds true for thromboprophylaxis in patients with COVID-19 despite the numerous advancements in this field over the past decade.
A review of societal guidance shows there is universal agreement with regards to supporting standard doses of pharmacologicalprophylaxis in acutely ill patients either when universally applied or guided by a RAM as well as the use of universal thromboprophylaxis in critically ill patients. All societies discourage the use of antiplatelet therapy for arterial thrombosis prevention and advocate for mechanical compression in patients with contraindications to pharmacologic anticoagulation. Beyond this, divergence between guidance statements begins to appear. For example, societies do not currently agree on the role and approach for extended pharmacologic prophylaxis postdischarge. The differences between societal guidance speaks to the degree of uncertainty among leading experts, which is considered to be the logical outworking of the current level of evidence. Regardless, these guidance documents should be considered the best resource currently available.
The medical community is fortunate to have robust societies that have published guidance on thromboprophylaxis in patients with COVID-19. The novelty of COVID-19 precludes these societal guidance publications from being based on high-quality evidence, but at the very least, they provide insight into how leading experts in the field of thrombosis and hemostasis are currently navigating the therapeutic landscape.
While this paper provides a summary of the current guidance, evidence is evolving at an unprecedented pace. Facilities and anticoagulation leads should be actively and frequently evaluating literature and guidance to ensure their practices and policies remain current.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville/Murfreesboro.
The novel coronavirus SARS-CoV-2 and resulting viral syndrome (COVID-19) was first reported in China during December 2019 and within weeks emerged in the US.1 Since it is a rapidly evolving situation, clinicians must remain current on best practices—a challenging institutional responsibility. According to LitCovid, a curated literature hub for tracking scientific information on COVID-19, there are > 54,000 articles on the subject in PubMed. Among these include venous thromboembolism (VTE) prophylaxis guidance from 4 respected thrombosis organizations/societies and the US National Institutes of Health.1-5
Observations
COVID-19 predisposes patients with and without a history of cardiovascular disease to thrombotic complications, occurring in either the venous or arterial circulation system.2,6 Early observational studies suggest that thrombotic rates may be in excess of 20 to 30%; however, the use of prophylactic anticoagulation was inconsistent among studies that were rushed to publication.6
Autopsy data have demonstrated the presence of fibrin thrombi within distended small vessels and capillaries and extensive extracellular fibrin deposition.6 Investigators compared the characteristics of acute pulmonary embolism in 23 cases with COVID-19 but with no clinical signs of deep vein thrombosis with 100 controls without COVID-19.7 They observed that thrombotic lesions had a greater distribution in peripheral lung segments (ie, peripheral arteries) and were less extensive for those with COVID-19 vs without COVID-19 infection. Thus, experts currently hypothesize that COVID-19 has a distinct “pathomechanism.” As a unique phenotype, thrombotic events represent a combination of thromboembolic disease influenced by components of the Virchow triad (eg, acute illness and immobility) and in situ immunothrombosis, a local inflammatory response.6,7
Well-established surgical and nonsurgical VTE thromboprophylaxis guidelines serve as the foundation for current COVID-19 thromboprophylaxis guidance.8,9 Condition specific guidance is extrapolated from small, retrospective observational studies or based on expert opinion, representing levels 2 and 3 evidence, respectively.1-5 Table 1 captures similarities and differences among COVID-19 VTE thromboprophylaxis recommendations which vary by time to publication and by society member expertise gained from practice in the field.
Three thrombosis societies recommend universal pharmacologic thromboprophylaxis for acutely ill COVID-19 patients who lack contraindications.3-5 Others recommend use of risk stratification scoring tools, such as the Padua risk assessment model (RAM) for medical patients or Caprini RAM for surgical patients, the disseminated intravascular coagulation (DIC) score, or the sepsis-induced coagulopathy score to determine therapeutic appropriateness (Tables 2 and 3).1,2 Since most patients hospitalized for COVID-19 will present with a pathognomonic pneumonia and an oxygen requirement, they will generally achieve a score of ≥ 4 when the Padua RAM is applied; thus, representing a clear indication for pharmacologic thromboprophylaxis.8,9 If the patient is pregnant, the Anticoagulation Forum recommends pharmacologic prophylaxis, consultation with an obstetrician, and use of obstetrical thromboprophylaxis guidelines.3,10,11
Most thrombosis experts prefer parenteral thromboprophylaxis, specifically low molecular-weight heparin (LMWH) or fondaparinux, for inpatients over use of direct oral anticoagulants (DOACs) in order to minimize the potential for drug interactions particularly when investigational antivirals are in use.4 Once-daily agents (eg, rivaroxaban, fondaparinux, and enoxaparin) are preferred over multiple daily doses to minimize staff contact with patients infected with COVID-19.4,5 Fondaparinux and DOACs should preferentially be used in patients with a recent history of heparin-induced thrombocytopenia with and without thrombosis (HIT/HITTS). Subcutaneous heparin is reserved for patients who are scheduled for invasive procedures or have reduced renal function (eg, creatinine clearance < 30 mL/min).1,3-5 In line with existing pharmacologic thromboprophylaxis guidance, standard prophylactic LMWH doses are recommended unless patients are obese (body mass index [BMI] > 30) or morbidly obese (BMI > 40) necessitating selection of intermediate doses.4
Since early non-US studies demonstrated high thrombotic risk without signaling a potential for harm from pharmacologic thromboprophylaxis, some organizations recommend empiric escalation of anticoagulation doses for critical illness.3,4,6 Thus, it may be reasonable to advance to either intermediate pharmacologic thromboprophylaxis dosing or therapeutic doses.3 However, observational studies question this aggressive practice unless a clear indication exists for intensification (ie, atrial fibrillation, known VTE).
A large multi-institutional registry study that included 400 subjects from 5 centers demonstrated a radiographically confirmed VTE rate of 4.8% and an arterial thrombosis rate of 2.8%.6 When limiting to the critically ill setting, VTE and arterial thrombosis occurred at slightly higher rates (7.6% and 5.6%, respectively). Patients also were at risk for nonvessel thrombotic complications (eg, CVVH circuit, central venous catheters, and arterial lines). Subsequently, the overall thrombotic complication rate was 9.5%. All thrombotic events except one arose in patients who were receiving standard doses of pharmacologic thromboprophylaxis. Unfortunately, D-dimer elevation at admission was not only predictive of thrombosis and death, but portended bleeding. The overall bleeding rate was 4.8%, with a major bleeding rate of 2.3%. In the context of observing thromboses at normally expected rates during critical illness in association with a significant bleeding risk, the authors recommended further investigation into the net clinical benefit.
Similarly, a National Institutes of Health funded, observational, single center US study evaluated 4,389 inpatients infected with COVID-19 and determined that therapeutic and prophylactic anticoagulation reduced inpatient mortality (adjusted hazard ratio [aHR], 0.53 and 0.50, respectively for the primary outcome) and intubation (aHR, 0.69 and 0.72, respectively) over no anticoagulation.12 Notably, use of inpatient therapeutic anticoagulation commonly represented a continuation of preadmission therapy or progressive COVID-19. A subanalysis demonstrated that timely use (eg, within 48 hours of admission) of prophylactic or therapeutic anticoagulation, resulted in no difference (P < .08) in the primary outcome. Bleeding rates were low overall: 3%, 1.7%, and 1.9% for therapeutic, prophylactic, and no anticoagulation groups, respectively. Furthermore, selection of DOACs seems to be associated with lower bleeding rates when compared with that of LMWH heparin (1.3% vs 2.6%, respectively). In those where site of bleeding could be ascertained, the most common sites were the gastrointestinal tract (50.7%) followed by mucocutaneous (19.4%), bronchopulmonary (14.9%), and intracranial (6%). In summary, prophylactic thromboprophylaxis doses seem to be associated with positive net clinical benefit.
As of October 30, 2020, the US Department of Veterans Affairs (VA) had reported 75,156 COVID-19 cases and 3,961 deaths.13 Since the VA Pharmacy Benefits Management (PBM) does not disseminate nationally prepared anticoagulation order sets to the field, facility anticoagulation leads should be encouraged to develop local guidance-based policies to help standardize care and minimize further variations in practice, which would likely lack evidential support. Per the VA Tennessee Valley Healthcare System (TVHS)- Nashville/Murfreesboro anticoagulation policy, we limit the ordering of parenteral anticoagulation to Computerized Patient Record System (CPRS) order sets in order to provide decision support (eFigure 1, available at doi:10.12788/fp.0063). Other facilities have shown that embedded clinical decision support tools increase adherence to guideline VTE prophylaxis recommendations within the VA.14
In April 2020, the TVHS anticoagulation clinical pharmacy leads developed a COVID-19 specific order set based on review of societal guidance and the evolving, supportive literature summarized in this review with consideration of provider familiarity (eFigure 2, available at doi:10.12788/fp.0063)). Between April and June 2020, the COVID-19 order set content consistently evolved with publication of each COVID-19 thromboprophylaxis guideline.1-5
Since TVHS is a high-complexity facility, we elected to use universal pharmacologic thromboprophylaxis for patients with COVID-19. This construct bypasses the use of scoring tools (eg, RAM), although we use Padua and Caprini RAMS for medical and surgical patients, respectively, who are not diagnosed with COVID-19. The order set displays all acceptable guideline recommended options, delineated by location of care (eg, medical ward vs intensive care unit), prior history of heparin-induced thrombocytopenia, and renal function. Subsequently, all potential agents, doses, and dosing interval options are offered so that the provider autonomously determines how to individualize the clinical care. Since TVHS has only diagnosed 932 ambulatory/inpatient COVID-19 cases combined, our plans are to complete a future observational analysis to determine the effectiveness of the inpatient COVID-19 thromboprophylaxis order set for our internal customers.
Conclusions
The COVID-19 pandemic has resulted in arguably the most challenging medical climate in the evidence-based medicine era. Until high-quality randomized controlled trials are published, the medical community is, in a sense, operating within a crucible of crisis having to navigate therapeutic policy with little certainty. This principle holds true for thromboprophylaxis in patients with COVID-19 despite the numerous advancements in this field over the past decade.
A review of societal guidance shows there is universal agreement with regards to supporting standard doses of pharmacologicalprophylaxis in acutely ill patients either when universally applied or guided by a RAM as well as the use of universal thromboprophylaxis in critically ill patients. All societies discourage the use of antiplatelet therapy for arterial thrombosis prevention and advocate for mechanical compression in patients with contraindications to pharmacologic anticoagulation. Beyond this, divergence between guidance statements begins to appear. For example, societies do not currently agree on the role and approach for extended pharmacologic prophylaxis postdischarge. The differences between societal guidance speaks to the degree of uncertainty among leading experts, which is considered to be the logical outworking of the current level of evidence. Regardless, these guidance documents should be considered the best resource currently available.
The medical community is fortunate to have robust societies that have published guidance on thromboprophylaxis in patients with COVID-19. The novelty of COVID-19 precludes these societal guidance publications from being based on high-quality evidence, but at the very least, they provide insight into how leading experts in the field of thrombosis and hemostasis are currently navigating the therapeutic landscape.
While this paper provides a summary of the current guidance, evidence is evolving at an unprecedented pace. Facilities and anticoagulation leads should be actively and frequently evaluating literature and guidance to ensure their practices and policies remain current.
Acknowledgments
This material is the result of work supported with resources and the use of facilities at the VA Tennessee Valley Healthcare System in Nashville/Murfreesboro.
1. National Institutes of Health. Coronavirus disease 2019 (COVID-19) treatment guidelines. https://www.covid19treatmentguidelines.nih.gov/whats-new/. Updated October 9, 2020. Accessed October 15, 2020.
2. Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(23):2950-2973. doi:10.1016/j.jacc.2020.04.031
3. Barnes GD, Burnett A, Allen A, et al. Thromboembolism and anticoagulant therapy during the COVID-19 pandemic: interim clinical guidance from the anticoagulation forum. J Thromb Thrombolysis. 2020;50(1):72-81. doi:10.1007/s11239-020-02138-z
4. Spyropoulos AC, Levy JH, Ageno W, et al. Scientific and Standardization Committee communication: Clinical guidance on the diagnosis, prevention, and treatment of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18(8):1859-1865. doi:10.1111/jth.14929
5. Moores LK, Tritschler T, Brosnahan S, et al. Prevention, Diagnosis, and Treatment of VTE in Patients With Coronavirus Disease 2019: CHEST Guideline and Expert Panel Report. Chest. 2020;158(3):1143-1163. doi:10.1016/j.chest.2020.05.559
6. Al-Samkari H, Karp Leaf RS, Dzik WH, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136(4):489-500. doi:10.1182/blood.2020006520.
7. van Dam LF, Kroft LJM, van der Wal LI, et al. Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: a different phenotype of thrombotic disease?. Thromb Res. 2020;193:86-89. doi:10.1016/j.thromres.2020.06.010
8. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e195S-e226S. doi:10.1378/chest.11-2296
9. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines [published correction appears in Chest. 2012 May;141(5):1369]. Chest. 2012;141(suppl 2):e227S-e277S. doi:10.1378/chest.11-2297
10. ACOG Practice Bulletin No. 196 Summary: thromboembolism in pregnancy. Obstet Gynecol. 2018;132(1):243-248. doi:10.1097/AOG.0000000000002707
11. Royal College of Obstetricians and Gynaecologists. Reducing the risk of venous thromboembolism during pregnancy and the puerperium. Green-top Guideline. No. 37a. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-37a.pdf. Published April 2015. Accessed October 15, 2020.
12. Nadkarni GN, Lala A, Bagiella E, et al. Anticoagulation, mortality, bleeding and pathology among patients hospitalized with COVID-19: a single health system study [published online ahead of print, 2020 Aug 24]. J Am Coll Cardiol. 2020;76(16):1815-1826. doi:10.1016/j.jacc.2020.08.041
13. US Department of Veterans Affairs. Department of Veterans Affairs COVID-19 national summary. https://www.accesstocare.va.gov/Healthcare/COVID19NationalSummary. Updated November 1, 2020. Accessed November 1, 2020.
14. George B, Gonzales S, Patel K, Petit S, Franck AJ, Bovio Franck J. Impact of a clinical decision-support tool on venous thromboembolism prophylaxis in acutely ill medical patients. J Pharm Technol. 2020;36(4):141-147. doi:10.1177/8755122520930288
1. National Institutes of Health. Coronavirus disease 2019 (COVID-19) treatment guidelines. https://www.covid19treatmentguidelines.nih.gov/whats-new/. Updated October 9, 2020. Accessed October 15, 2020.
2. Bikdeli B, Madhavan MV, Jimenez D, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(23):2950-2973. doi:10.1016/j.jacc.2020.04.031
3. Barnes GD, Burnett A, Allen A, et al. Thromboembolism and anticoagulant therapy during the COVID-19 pandemic: interim clinical guidance from the anticoagulation forum. J Thromb Thrombolysis. 2020;50(1):72-81. doi:10.1007/s11239-020-02138-z
4. Spyropoulos AC, Levy JH, Ageno W, et al. Scientific and Standardization Committee communication: Clinical guidance on the diagnosis, prevention, and treatment of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18(8):1859-1865. doi:10.1111/jth.14929
5. Moores LK, Tritschler T, Brosnahan S, et al. Prevention, Diagnosis, and Treatment of VTE in Patients With Coronavirus Disease 2019: CHEST Guideline and Expert Panel Report. Chest. 2020;158(3):1143-1163. doi:10.1016/j.chest.2020.05.559
6. Al-Samkari H, Karp Leaf RS, Dzik WH, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136(4):489-500. doi:10.1182/blood.2020006520.
7. van Dam LF, Kroft LJM, van der Wal LI, et al. Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: a different phenotype of thrombotic disease?. Thromb Res. 2020;193:86-89. doi:10.1016/j.thromres.2020.06.010
8. Kahn SR, Lim W, Dunn AS, et al. Prevention of VTE in nonsurgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e195S-e226S. doi:10.1378/chest.11-2296
9. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines [published correction appears in Chest. 2012 May;141(5):1369]. Chest. 2012;141(suppl 2):e227S-e277S. doi:10.1378/chest.11-2297
10. ACOG Practice Bulletin No. 196 Summary: thromboembolism in pregnancy. Obstet Gynecol. 2018;132(1):243-248. doi:10.1097/AOG.0000000000002707
11. Royal College of Obstetricians and Gynaecologists. Reducing the risk of venous thromboembolism during pregnancy and the puerperium. Green-top Guideline. No. 37a. https://www.rcog.org.uk/globalassets/documents/guidelines/gtg-37a.pdf. Published April 2015. Accessed October 15, 2020.
12. Nadkarni GN, Lala A, Bagiella E, et al. Anticoagulation, mortality, bleeding and pathology among patients hospitalized with COVID-19: a single health system study [published online ahead of print, 2020 Aug 24]. J Am Coll Cardiol. 2020;76(16):1815-1826. doi:10.1016/j.jacc.2020.08.041
13. US Department of Veterans Affairs. Department of Veterans Affairs COVID-19 national summary. https://www.accesstocare.va.gov/Healthcare/COVID19NationalSummary. Updated November 1, 2020. Accessed November 1, 2020.
14. George B, Gonzales S, Patel K, Petit S, Franck AJ, Bovio Franck J. Impact of a clinical decision-support tool on venous thromboembolism prophylaxis in acutely ill medical patients. J Pharm Technol. 2020;36(4):141-147. doi:10.1177/8755122520930288
The Veterans Affairs Patient Safety Center of Inquiry—Suicide Prevention Collaborative: Creating Novel Approaches to Suicide Prevention Among Veterans Receiving Community Services
Since 2008, suicide has ranked as the tenth leading cause of death for all ages in the US, with rates of suicide continuing to rise.1-3 Suicide is even more urgent to address in veteran populations. The age- and sex-adjusted suicide rate in 2017 was more than 1.5 times greater for veterans than it was for nonveteran adults.2 Of importance, rates of suicide are increasing at a faster rate in veterans who are not connected to Veterans Health Administration (VHA) care.4,5 These at-risk veterans include individuals who are eligible for VHA care yet have not had a VHA appointment within the year before death; veterans who may be ineligible to receive VHA care due to complex rules set by legislation; and veterans who are eligible but not enrolled in VHA care. Notably, between 2005 and 2016, the number of veterans not enrolled in VHA care rose more quickly than did the number of veterans enrolled in VHA care.5,6 Thus, to impact the high veteran suicide rates, an emergent challenge for VHA is to prevent suicide among unenrolled veterans and veterans receiving community care, while continuing to increase access to mental health services for veterans enrolled in VHA health care.
In response to the high rates of veteran suicide deaths, the US Department of Veterans Affairs (VA) has developed a broad, multicomponent suicide prevention program that is unparalleled in private US health care systems.4,7 Suicide prevention efforts are led and implemented by both the VHA National Center for Patient Safety and the VHA Office of Mental Health and Suicide Prevention. Program components are numerous and multifaceted, falling within the broad promotion and prevention strategies outlined by the National Academy of Medicine (NAM).1,8-11 The NAM continuum of prevention model encompassing multiple strategies is also referred to as the Universal, Selective, Indicated (USI) Model.7,8,10 The VHA suicide prevention program contains a wide spread of program components, making it both comprehensive and innovative (Table 1).
Although significant momentum and progress has been made within the VHA, policy set by legislation has historically limited access to VHA health care services to VHA-eligible veterans. This is particularly concerning given the rising suicide rates among veterans not engaged in VHA care.2 Adding to this complexity, recent legislation has increased veterans’ access to non-VHA health care, in addition to their existing access through Medicare, Medicaid, and other health care programs.12-14 Best practices for suicide prevention are not often implemented in the private sector; thus, these systems are ill prepared to adequately meet the suicide prevention care needs of veterans.4,15-18 Furthermore, VHA and non-VHA services generally are not well coordinated, and private sector health care providers (HCPs) are not required to complete a commensurate level of suicide prevention training as are VHA HCPs.16-18 Most non-VHA HCPs do not receive military cultural competence training.19 These issues create a significant gap in suicide prevention services and may contribute to the increases in suicide rates in veterans who do not receive VHA care. Thus, changes in policy to increase access through private sector care may have paradoxical effects on veteran suicide deaths. To impact the veteran suicide rate, VHA must develop and disseminate best practices for veterans who use non-VHA services.
A Roadmap to Suicide Prevention
There is significant momentum at the federal level regarding this issue. The President’s Roadmap to Empower Veterans and End the National Tragedy of Suicide (Executive Order 13,861) directs the VHA to work closely with community organizations to improve veteran suicide prevention.20 The VHA and partners, such as the Substance Abuse and Mental Health Services Administration (SAMHSA), are bridging this gap with collaborative efforts that increase suicide prevention resources for veterans living in the community through programs such as the Governor’s Challenges to Prevent Suicide Among Service Members, Veterans, and their Families. These programs intend to empower communities to develop statewide, strategic action plans to prevent veteran suicide.7,21-24
In addition to partnerships, VHA has built other aspects of outreach and intervention into its programming. A key VHA initiative is to “know all veterans” by committing to identifying and reaching out to all veterans who may be at risk for suicide.22 The VHA has committed to offering “emergency stabilization care for former service members who present at the facility with an emergent mental health need” regardless of eligibility.25 The intent is to provide temporary emergent mental health care to veterans who are otherwise ineligible for care, such as those who were discharged under other-than-honorable conditions while the VHA determines eligibility status.26 However, veterans must meet certain criteria, and there is a limit on services.
Although services are being expanded to reach veterans who do not access VHA health care, how to best implement these new directives with regard to suicide prevention is unclear. Strategic development and innovations to expand suicide prevention care to veterans outside the current reach of VHA are desperately needed.
Program Overview
VHA Patient Safety Center of Inquiry-Suicide Prevention Collaborative (PSCI-SPC), funded by the VHA National Center for Patient Safety, aims to help fill the gap in community-based suicide prevention for veterans. PSCI-SPC is located within the VHA Rocky Mountain Mental Illness Research, Education, and Clinical Center in Aurora, Colorado. The overarching mission of PSCI-SPC is to develop, implement, and evaluate practical solutions to reduce suicide among veterans not receiving VHA care. PSCI-SPC serves as a national clinical innovation and dissemination center for best practices in suicide prevention for organizations that serve veterans who receive care in the community. PSCI-SPC creates products to support dissemination of these practices to other VAMCs and works to ensure these programs are sustainable. PSCI-SPC focuses on 3 primary objectives. All PSCI-SPC projects are currently underway.
Objective 1: Growing a Community Learning Collaborative
Acknowledging that nearly two-thirds of veterans who die by suicide do not use VHA services, PSCI-SPC aims to reduce suicide among all veterans by expanding the reach of best practices for suicide prevention to veterans who receive myriad services in the community.27 Community organizations are defined here as organizations that may in some way serve, interact with, or work with veterans, and/or employ veterans. Examples include non-VHA health care systems, public services such as police and fire departments, nonprofit organizations, mental health clinics, and veterans’ courts. As veterans increasingly seek health care and other services within their communities, the success of suicide prevention will be influenced by the capability of non-VHA public and private organizations. Objective 1, therefore, seeks to develop a VHA-community collaborative that can be leveraged to improve systems of suicide prevention.
Current programs in the VHA have focused on implementation of suicide prevention awareness and prevention education campaigns instead of grassroots partnerships that are intended to be sustainable. Additionally, these programs typically lack the capacity and systems to sustain numerous meaningful community partnerships. Traditionally, community organizations have been hesitant to partner with government agencies, such as the VHA, due to histories of institutional mistrust and bureaucracy.28
The PSCI-SPC model for developing a VHA-community collaborative partnership draws from the tradition of community-based participatory research. The best community-based participatory research practices are to build on strengths and resources within the local community; develop collaborative, equitable partnerships that involve an empowering and power-sharing process; foster colearning, heuristics, and capacity building among partners; and focus on systems development using an iterative process. These practices also are consistent with the literature on learning collaboratives.29-31
The premise for a learning collaborative is to bridge the gap between knowledge and practice in health care.31 Figure 1 depicts how this collaborative was developed, and how it supports Objectives 2 and 3. To achieve Objective 1, we developed a VHA-learning collaborative of 13 influential community partners in the Denver and Colorado Springs region of Colorado. The VHA team consists of a learning collaborative leader, a program manager, and a program support assistant. The principal investigator attends and contributes to all meetings. Learning collaborative partners include a university psychology clinic that focuses on veterans’ care, 3 veterans service organizations, a mental health private practice, a university school of nursing, a community mental health center, veterans’ courts, and 5 city departments.
These partners participated in qualitative interviews to identify where gaps and breakdowns were occurring. With this information, the PSCI-SPC team and VHA-learning collaborative held a kickoff event. At this meeting the team discussed the qualitative findings, provided veteran suicide prevention information, and basic information regarding suicide prevention program building and implementation science.
Throughout quarterly learning collaborative meetings and monthly facilitation calls, we have worked to develop a blueprint and an action plan for each partner to develop best practices for dissemination to aid in providing consistency in the standard of care. A postimplementation event will be held to identify successes and challenges encountered while operationalizing project action plans.
Currently, activities of the learning collaborative are making a large impact on the community. Not all collaborative members track information regarding their populations served, nor specific metrics on veterans encountered. Even so, of those who do capture metrics, suicide prevention program components implemented by the collaborative will impact more than 21,000 individuals and at least 2,500 known veterans. In addition, 52 new connections have been made between community organizations or between community organizations and the VHA, and > 300 individuals have been trained.The learning collaborative support team has assisted in the dissemination of a large resource list for veterans. As the learning collaborative is ongoing and we are working with organizations to improve their data collection and analytics, we expect these numbers to increase. We anticipate that the learning collaborative will develop a stronger suicide prevention safety net within the community. In addition, we expect increased referrals of at-risk veterans to the VHA and enhance the long-term continuity of care between community and VHA services.
Objective 2: Implementation Toolkit
The second PSCI-SPC objective is to develop a toolkit for the implementation of best practices within a VHA-community suicide prevention learning collaborative. Lessons from the development of a successful suicide prevention learning collaborative will be shared through an online guide that other VHA facilities can use to support similar collaborative efforts within their communities. The toolkit will be disseminated across the VHA to assist suicide prevention coordinators and other staff in developing a suicide prevention learning collaborative at their facilities.
PSCI-SPC uses the Zero Suicide framework and the VA/US Department of Defense (DoD) Clinical Practice Guideline for the Assessment and Management of Patients at Risk for Suicide as models for preventing suicide in veterans not enrolled in VHA care.11,32 This implementation toolkit focuses on how to implement suicide prevention best practices into organizations that serve veterans. This toolkit differs from clinical practice guidelines in that it focuses on implementation strategies to promote success and effectively address challenges.
In order to provide a menu of available options for the learning collaborative and resulting toolkit, PSCI-SPC uses a logic model to compare the components of the VHA suicide prevention program, as well as other similar veteran and military suicide prevention programs.7,12,14,21,33,34 These programs are categorized into 2 types of prevention frameworks, the USI model as described above, and the SAMHSA Strategic Prevention Framework (Table 2).35 The SAMHSA framework was designed to promote mental health and prevent substance abuse, yet the derived classification is also applicable to suicide prevention programs.35 The results of the logic model comparison form the basis of the best practice interventions for the learning collaborative and initial toolkit. In addition to the best practice interventions, the toolkit consists of documents describing how to develop a veteran suicide prevention learning collaborative, as well as tools for learning collaborative members. Current tool development includes workbooks to guide collaborative members through the implementation process, guides for community organizations in implementing suicide prevention screening and risk assessment, a standard operating procedure for suicide prevention in a veterans court, and peer support training for veteran suicide prevention.
The methods to achieve Objective 2 include regular discussions with the VHA-learning collaborative on current best practices, identifying gaps and overlap of community programs. Successes and challenges of implementing suicide prevention best practices into learning collaborative organizations will be documented and incorporated into the toolkit. The learning collaborative will work iteratively as a team to improve the toolkit. Once complete, the toolkit will be disseminated to other VHA health care systems nationally, as well as to other state or regional partners that the learning collaborative identifies. A plan is under development for national suicide prevention entities to also disseminate the toolkit to lessen the burden of veteran suicide through their stakeholder base.
Objective 3: High-Risk Veterans Not Receiving VHA Care
Although veterans not receiving VHA care account for a number of veteran deaths by suicide, we are not aware of any current VHA programs that provide temporary psychotherapy and intensive case management to at-risk veterans ineligible for VHA care who are in need of immediate care while an appropriate permanent community placement is identified. In the current system, veterans in the community can present to VHA suicide prevention services through several different systems, including referrals to VHA and the Veterans Crisis Line (VCL). However, a portion of VCL calls are from veterans whose VHA eligibility is unknown or who are ineligible for services. If veterans are at imminent risk for suicide, emergency care is coordinated for them. However, if veterans are not at imminent suicide risk they are referred to the local suicide prevention coordinator and instructed to independently work toward determining their VHA eligibility.
It is currently unknown how many veterans follow through with these instructions. Nonetheless, if veterans are deemed eligible, they may present to VHA to obtain a same-day appointment. If not eligible, a suicide prevention coordinator may give them the phone number of a community referral. However, this practice is not standardized across VA medical centers, and the provided resources are up to the suicide prevention coordinator to research. Additionally, when a VHA suicide prevention coordinator leaves the position, knowledge of these community resources and rapport with community HCPs are often lost, leaving the next coordinator to develop these again, which reduces the efficiency and effectiveness of limited resources. It is also unknown how many veterans complete this contact and receive evidence-based treatment following referral. This is a complex system to navigate, particularly when at risk for suicide and in need of immediate but not emergency services.
Suicide prevention in such circumstances may be improved by adapting current suicide prevention practices, including evidence-based interventions, and the new VHA intensive case management program,11,36 within a Zero Suicide framework. PSCI-SPC has developed a brief intervention to transition ineligible veterans to permanent community treatment and provide them with additional resources to meet their varied needs. The brief 1 to 3 session intervention combines practices from brief cognitive behavioral therapy (BCBT) for suicide prevention, crisis response planning (CRP), and intensive case management within a Zero Suicide framework. Both the 2019 VA/DoD suicide prevention clinical practice guidelines and Zero Suicide recommend using cognitive behavioral therapy (CBT)-based interventions for suicide prevention.11,32 These interventions are packaged into a single intervention delivered by a PSCI-SPC therapist, typically a licensed clinical social worker, a licensed clinical psychologist, or an unlicensed psychologist under the supervision of a licensed clinical psychologist.
BCBT is one type of CBT that has shown initial efficacy in reducing suicide attempts.37 BCBT reduces the risk for suicide attempts both at the conclusion of treatment and at 24-month follow-up.37 BCBT is boiled down to its most essential components so it can be delivered in a distilled format. An essential element of BCBT that will remain is the CRP. A CRP11,37,38 entails collaboratively identifying effective, appropriate coping strategies and specific individuals to contact during a crisis. CRPs demonstrated efficacy as a stand-alone intervention to existing suicide prevention methods in a randomized clinical trial, such that individuals who received CRP had faster reductions in suicidal ideation and were 76% less likely to make a suicide attempt during the 6-month follow-up period.39 These results demonstrate that use of a CRP is connected to a decrease in suicidal behavior among suicidal patients.
The VHA has developed and is piloting a new initiative focused on restructuring its intensive case management services. RACETIME to Integrated Care (eg, Risk stratification, Assessment of complexity, Coordinator of lead assignment, Evaluate whole health needs, Trusting partnerships, Integrate care, Monitor progress, Experience of the veteran and employee) is a framework that assists VHA case managers in transitioning from a traditional case management mind-set to a more integrated and holistic method of care.36 RACETIME intensive case management practices will be incorporated into the intervention. However, RACETIME focuses on case management internally to the VHA. A modification for this treatment will be to focus on intensive case management from a mental health perspective and connecting to external community resources. Community referrals are mapped within a structured process and stored on a shared drive. This improves continuity between suicide prevention coordinators when they leave for a new position.
This intervention is conducted within a Zero Suicide framework. Pertinent to PSCI-SPC innovation to enhance care for non-VHA veterans is the care transitions element within the Zero Suicide framework, which has developed comprehensive suicide prevention guidance, including a pathway to care.32 This pathway refers a process to conduct follow-up supportive contacts that are tracked and recorded.
The PSCI-SPC pilot program incorporates the elements of CRP and brief CBT within a Zero Suicide framework. The PSCI-SPC team is developing and testing a protocol for providing brief treatment and community referrals to ineligible veterans that integrates these programming elements (Figure 2). A PSCI-SPC social worker will coordinate with the eligibility office to determine VHA eligibility. Ineligible veterans are referred to community partners and nonenrolled, eligible veterans are linked to VHA HCPs if they desire. These transitions will be coordinated, closely monitored, and verified.
Once the eligibility is determined, and the veteran’s preference is assessed, the team will perform a warm handoff to a VHA representative for enrollment into VHA care or to a community placement. We have sourced multiple community placements at varied payor levels. All veterans who are ineligible for care or who do not desire to be enrolled in VHA care will be provided the intervention package described above. After the veteran is placed in community care, PSCI-SPC team will follow up regularly with the veteran for 3 months to ensure continuity of care. If additional sessions are needed while the veteran needs a community placement, the PSCI team can accommodate this and will track the number of participants who needed additional sessions. If the veteran is deemed to be at imminent risk for suicide at any time during the pilot, he/she is transferred to emergency care. The veteran is allowed to participate once stabilized.
Findings from the pilot program will inform a manual intended for dissemination to patient safety and suicide prevention coordinators nationally across the VHA. The products from this objective will be integrated with the other objectives in training that will be provided on how to develop a local collaborative (Objective 1), disseminate the toolkit (Objective 2), and receive referrals into VHA or refer veterans to community clinical partners through the pilot program (Objective 3). Products developed and disseminated throughout PSCI-SPC aim to create momentum to reduce rates of suicide in veterans who are not connected to the VHA at the local community level.
Discussion
Improving suicide prevention for veterans who receive non-VHA health care is essential to significantly reduce veteran suicide rates. For the past decade, VHA suicide prevention initiatives have largely focused on veterans eligible for care, although the fastest increase in veteran suicide rates has occurred among veterans not connected to VHA services. Currently, if a veteran is deemed ineligible for care, it is up to the veteran to find other health care services in his or her community. There is not always a clear next step for the veteran to take, nor clear guidance provided to the VHA registration staff to assist with this care transition. This is particularly concerning for veterans at high risk for suicide as this could further thwart the veteran’s sense of belongingness and increase perceived burdensomeness, both suicide risk factors, and discourage them from attaining help.40 Overall, while the VHA has successfully implemented diverse suicide prevention initiatives and services, the need for continued system improvement focused on non-VHA veterans remains. PSCI-SPC was developed for this purpose.
By creating a collaborative that will connect VHA and community organizations, there will be better utilization of resources and more appropriate referrals throughout systems that interact with veterans. Sharing suicide prevention best practices between VHA and community partners is expected to increase access to mental health treatment to all veterans. Finally, by allowing best practices for suicide prevention in the VHA to serve as a guide in the development of best practices for suicide prevention between the VHA and the local health and behavioral health care community, PSCI-SPC will create a new suicide prevention intervention for veterans with mental health needs. Through these initiatives, PSCI-SPC will support providers’ and concerned citizens’ efforts to ensure that fewer veterans fall through the cracks of disjointed systems and will promote healthier communities where, regardless of VHA enrollment status, veterans receive suicide prevention care.
Conclusions
PSCI-SPC is a novel center for the innovation and dissemination of the nation’s best practices in suicide prevention for veterans who are ineligible for or otherwise not engaged in VHA services and who turn to their community for health care. PSCI-SPC not only seeks to create, develop, and measure various solutions to reduce suicide among veterans who receive non-VHA care, but also seeks to facilitate the overall quality of existing practices for suicide prevention and care coordination for enrolled veterans who use community resources. By bridging the gap between the VHA, civilian health care systems, and other community partners striving to prevent veteran suicides, we can create better access to care and a more seamless path of communication among these important entities that impact the lives of our veterans daily
1. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published September 2018. Accessed October 14, 2020.
2. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National veteran suicide prevention annual report. https://www.mentalhealth.va.gov/suicide_prevention/data.asp. Accessed October 14, 2020.
3. Centers for Disease Control and Prevention. Web-based injury statistics query and reporting system (WISQARS). 2014. https://www.cdc.gov/injury/wisqars/index.html. Updated July 1, 2020. Accessed October 14, 2020.
4. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health services. Fed Pract. 2018;35(3):18-24.
5. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. VA national suicide data report 2005-2016. https://www.mentalhealth.va.gov/docs/data-sheets/OMHSP_National_Suicide_Data_Report_2005-2016_508.pdf. Published September 2018. Accessed October 14, 2020.
6. US Department of Veterans Affairs, Office of Suicide Prevention. VA national suicide data report 2005-2015. https://www.mentalhealth.va.gov/docs/data-sheets/2015/OMHSP_National_Suicide_Data_Report_2005-2015_06-14-18_508.pdf. Published June 2018. Accessed October 14, 2020.
7. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published 2018. Accessed October 30, 2020.
8. Gordon RS. An operational classification of disease prevention. Public Health Rep. 1983;98(2):107-109.
9. National Research Council and Institute of Medicine. O’Connell ME, Boat T, Warner KE, eds. Preventing Mental, Emotional, and Behavioral Disorders Among Young People: Progress and Possibilities. Washington, DC: National Academies Press; 2009.
10. US Department of Health and Human Services, National Institute on Drug Abuse, Office of Science Policy and Communications. Drug Abuse Prevention: What Works. Rockville, MD: National Institutes of Health; 1997.
11. US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the assessment and management of patients at risk for suicide. https://www.healthquality.va.gov/guidelines/MH/srb. Published 2019. Updated July 30, 2020. Accessed October 14, 2020.
12. US Department of Veterans Affairs. Veterans Access, Choice and Accountability Act of 2014. https://www.govinfo.gov/content/pkg/PLAW-113publ146/pdf/PLAW-113publ146.pdf. Accessed October 14, 2020.
13. US Department of Veterans Affairs. Strategic Health Care. VA MISSION Act: Extension of the VA choice program. https://strategichealthcare.net/wp-content/uploads/2018/05/052718-VA-MISSION-Act-Summary.pdf. Accessed October 14, 2020.
14. US Congress. H.R.5674—VA MISSION Act of 2018. https://www.congress.gov/bill/115th-congress/house-bill/5674. Accessed October 15, 2020.
15. Grumet JG, Hogan MF, Chu A, Covington DW, Johnson KE. Compliance standards pave the way for reducing suicide in health care systems. J Health Care Compliance. 2019;17-26.
16. Hillestad R, Bigelow J, Bower A, et al. Can electronic medical record systems transform health care? Potential health benefits, savings, and costs. Health Aff (Millwood). 2005;24(5):1103-1117. doi:10.1377/hlthaff.24.5.110
17. Porter ME, Pabo EA, Lee TH. Redesigning primary care: a strategic vision to improve value by organizing around patients’ needs. Health Aff (Millwood). 2013;32(3):516-525. doi:10.1377/hlthaff.2012.0961
18. Nevedal AL, Wagner TH, Ellerbe LS, Asch SM, Koenig CJ. A qualitative study of primary care providers’ experiences with the Veterans Choice Program. J Gen Intern Med. 2019;34:598-603. doi:10.1007/s11606-018-4810-2
19. Tanielian T, Farris C, Epley C, et al. Ready to serve: community-based provider capacity to deliver culturally competent, quality mental health care to veterans and their families. https://www.rand.org/content/dam/rand/pubs/research_reports/RR800/RR806/RAND_RR806.pdf. Published 2014. Accessed October 15, 2020.
20. White House. Executive Order 13861: President’s roadmap to empower veterans and end the national tragedy of suicide (PREVENTS). https://www.va.gov/PREVENTS/docs/PRE-007-The-PREVENTS-Roadmap-1-2_508.pdf. Published June 17, 2020. Accessed October 15, 2020.
21. US Department of Veterans Affairs, Office of the Under Secretary for Health. Memorandum. VA continues community suicide prevention challenge at another mayor’s challenge policy academy. https://www.blogs.va.gov/VAntage/58468/va-continues-community-suicide-prevention-challenge-another-mayors-challenge-policy-academy. Published April 4, 2019. Accessed October 15, 2020.
22. US Department of Veterans Affairs, Veterans Health Administration (VHA) Veterans Health Administration (VHA) Fiscal Year (FY) 2018-2019 Operational Plan. www.navao.org/wp-content/uploads/2018/06/USH-Memo-10-2018-03-VHA_FY_2018-2019_Operational_Plan-4-19-18.pdf. Published April 19, 2018. Accessed October 15, 2020.
23. US Department of Veterans Affairs. VA, Health and Human Services announce governor’s challenge to prevent suicide. https://www.blogs.va.gov/VAntage/55707/va-health-human-services-announce-governors-challenge-prevent-suicide. Published January 17, 2019. Accessed October 15, 2020.
24. VA’s suicide prevention campaign “Be There’’ kicks off in Times Square. https://www.blogs.va.gov/VAntage/57272/vas-suicide-prevention-campaign-kicks-off-times-square. Published March 6, 2019. Accessed October 15, 2020.
25. US Department of Veterans Affairs, Office of Public Affairs Media Relations. Emergent mental health care for former service members. https://www.mentalhealth.va.gov/docs/Fact_Sheet-Emergent_Mental_Health_Care_Former_Service_Members.pdf. Published 2017. Accessed October 15, 2020.
26. US Department of Veterans Affairs. VA secretary formalizes expansion of emergency mental health care to former service members with other-than-honorable discharges. https://www.blogs.va.gov/VAntage/39092/va-secretary-formalizes-expansion-emergency-mental-health-care-former-service-members-honorable-discharges. Published June 27, 2017. Accessed October 15, 2020.
27. Shane L. New veteran suicide numbers raise concerns among experts hoping for positive news. Military Times. https://www.militarytimes.com/news/pentagon-congress/2019/10/09/new-veteran-suicide-numbers-raise-concerns-among-experts-hoping-for-positive-news. Published October 9, 2019. Accessed October 15, 2020.
28. US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 Strategic Plan. https://www.va.gov/oei/docs/VA2018-2024strategicPlan.pdf. Updated May 31, 2019. Accessed October 15, 2020.
29. Komaie G, Goodman M, McCall A, et al. Training community members in public health research: development and implementation of a community participatory research pilot project. Health Equity. 2018;2(1):282-287. doi:10.1089/heq.2018.0043
30. Wang KH, Ray NJ, Berg DN, et al. Using community-based participatory research and organizational diagnosis to characterize relationships between community leaders and academic researchers. Prev Med Reports. 2017;7:180-186. doi:10.1016/j.pmedr.2017.06.007
31. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed October 15, 2020.
32. Zero Suicide in Health and Behavioral Health Care Institute. Zero suicide toolkit. https://zerosuicide.sprc.org/toolkit. Accessed October 15, 2020.

33. Preventing suicide: a technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicideTechnicalPackage.pdf. Published 2017. Accessed October 30, 2020.
34. US Department of Veterans Affairs. VA Office of Mental Health and Suicide Prevention guidebook. https://www.mentalhealth.va.gov/docs/VA-Office-of-Mental-Health-and-Suicide-Prevention-Guidebook-June-2018-FINAL-508.pdf. Published June 2018. Accessed October 15, 2020.
35. US Department of Health and Human Services, Substance Abuse and Mental Health Services, Administration Center for Substance Abuse Prevention. Focus on prevention: strategies and programs to prevent substance abuse. HHS Publication No. (SMA) 10-4120. https://helpandhopewv.org/docs/Focus%20on%20Prevention.pdf. Revised 2017. Accessed October 15, 2020.
36. Misiti R, Risinger A. RACETIME: an organizational care coordination approach to improving patient outcomes in a complex healthcare setting. Presentation at the Military Social Work & Behavioral Health Conference Austin, Texas. July 23, 2020. https://sites.utexas.edu/military-social-work-conference/misiti-risinger. Accessed October 23, 2020.
37. Rudd MD, Bryan CJ, Wertenberger EG, et al. Brief cognitive-behavioral therapy effects on post-treatment suicide attempts in a military sample: results of a randomized clinical trial with 2-year follow-up. Am J Psychiatry. 2015;172(5):441-449. doi:10.1176/appi.ajp.2014.14070843
38. Rudd MD, Mandrusiak M, Joiner TE Jr. The case against no-suicide contracts: The commitment to treatment statement as a practice alternative. Clin Psychol. 2006;62(2):243-251.
39. Bryan CJ, Mintz J, Clemans TA, et al. Effect of crisis response planning vs. contracts for safety on suicide risk in US Army soldiers: a randomized clinical trial. J Affect Disord. 2017;212:64-72. doi:10.1016/j.jad.2017.01.028
40. Joiner TE. Why People Die by Suicide. Cambridge, MA: First Harvard University Press; 2005:27.
41. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.16: Defense suicide prevention program. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649016p.pdf?ver=2020-09-11-122632-850. Published November 6, 2017. Updated September 11, 2020. Accessed October 14, 2020.
42. Zero Suicide in Health and Behavioral Health Care Institute. Engage: engaging patients. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=0. Accessed October 14, 2020.
43. Stone D, Holland K, Bartholow B, Crosby A, Davis S, Wilkins N. Preventing Suicide: A technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicidetechnicalpackage.pdf. Published 2017. Accessed October 14, 2020.
44. Mills PD, Watts BV, Miller S, et al. A checklist to identify inpatient suicide hazards in veterans affairs hospitals. Jt Comm J Qual Patient Saf. 2010;36(2):87-93. doi:10.1016/s1553-7250(10)36015-6
45. US Department of Veterans Affairs, Office of Research and Development. Crisis prevention: study evaluates VA program that identifies vets at highest risk for suicide. https://www.research.va.gov/currents/0918-Study-evaluates-VA-program-that-identifies-Vets-at-highest-risk-for-suicide.cfm. Updated September, 20, 2018. Accessed October 14, 2020.
46. Zero Suicide in Health and Behavioral Health Care Institute. Engage: active engagement for safer suicide care--reducing access to lethal means. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Published 2017. Accessed October 14, 2020.
47. Zero Suicide in Health and Behavioral Health Care Institute. Train: training for all. http://zerosuicide.sprc.org/toolkit/train. Accessed October 14, 2020.
48. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention (OMHSP). Facts about veteran suicide. https://www.mentalhealth.va.gov/docs/FINAL_VA_OMHSP_Suicide_Prevention_Fact_Sheet_508.pdf. Published June 2018. Accessed October 14, 2020.
49. Zero Suicide in Health and Behavioral Health Care Institute. Treat: direct treatment. http://zerosuicide.edc.org/toolkit/treat. Accessed October 14, 2020.
50. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.04: Mental health evaluations of members of the military services. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649004p.pdf. Published March 4, 2013. Updated April 22, 2020. Accessed October 14, 2020.
51. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention Frequently asked questions: VA National Suicide Data Report. https://www.mentalhealth.va.gov/docs/data-sheets/2015_Data_Release_FAQs_508.pdf. Published September 2018. Updated June 22, 2018. Accessed October 14, 2020.
52. Zero Suicide in Health and Behavioral Health Care Institute. ZERO Suicide engage: active engagement for safer suicide care safety planning. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Accessed October 14, 2020.
Since 2008, suicide has ranked as the tenth leading cause of death for all ages in the US, with rates of suicide continuing to rise.1-3 Suicide is even more urgent to address in veteran populations. The age- and sex-adjusted suicide rate in 2017 was more than 1.5 times greater for veterans than it was for nonveteran adults.2 Of importance, rates of suicide are increasing at a faster rate in veterans who are not connected to Veterans Health Administration (VHA) care.4,5 These at-risk veterans include individuals who are eligible for VHA care yet have not had a VHA appointment within the year before death; veterans who may be ineligible to receive VHA care due to complex rules set by legislation; and veterans who are eligible but not enrolled in VHA care. Notably, between 2005 and 2016, the number of veterans not enrolled in VHA care rose more quickly than did the number of veterans enrolled in VHA care.5,6 Thus, to impact the high veteran suicide rates, an emergent challenge for VHA is to prevent suicide among unenrolled veterans and veterans receiving community care, while continuing to increase access to mental health services for veterans enrolled in VHA health care.
In response to the high rates of veteran suicide deaths, the US Department of Veterans Affairs (VA) has developed a broad, multicomponent suicide prevention program that is unparalleled in private US health care systems.4,7 Suicide prevention efforts are led and implemented by both the VHA National Center for Patient Safety and the VHA Office of Mental Health and Suicide Prevention. Program components are numerous and multifaceted, falling within the broad promotion and prevention strategies outlined by the National Academy of Medicine (NAM).1,8-11 The NAM continuum of prevention model encompassing multiple strategies is also referred to as the Universal, Selective, Indicated (USI) Model.7,8,10 The VHA suicide prevention program contains a wide spread of program components, making it both comprehensive and innovative (Table 1).
Although significant momentum and progress has been made within the VHA, policy set by legislation has historically limited access to VHA health care services to VHA-eligible veterans. This is particularly concerning given the rising suicide rates among veterans not engaged in VHA care.2 Adding to this complexity, recent legislation has increased veterans’ access to non-VHA health care, in addition to their existing access through Medicare, Medicaid, and other health care programs.12-14 Best practices for suicide prevention are not often implemented in the private sector; thus, these systems are ill prepared to adequately meet the suicide prevention care needs of veterans.4,15-18 Furthermore, VHA and non-VHA services generally are not well coordinated, and private sector health care providers (HCPs) are not required to complete a commensurate level of suicide prevention training as are VHA HCPs.16-18 Most non-VHA HCPs do not receive military cultural competence training.19 These issues create a significant gap in suicide prevention services and may contribute to the increases in suicide rates in veterans who do not receive VHA care. Thus, changes in policy to increase access through private sector care may have paradoxical effects on veteran suicide deaths. To impact the veteran suicide rate, VHA must develop and disseminate best practices for veterans who use non-VHA services.
A Roadmap to Suicide Prevention
There is significant momentum at the federal level regarding this issue. The President’s Roadmap to Empower Veterans and End the National Tragedy of Suicide (Executive Order 13,861) directs the VHA to work closely with community organizations to improve veteran suicide prevention.20 The VHA and partners, such as the Substance Abuse and Mental Health Services Administration (SAMHSA), are bridging this gap with collaborative efforts that increase suicide prevention resources for veterans living in the community through programs such as the Governor’s Challenges to Prevent Suicide Among Service Members, Veterans, and their Families. These programs intend to empower communities to develop statewide, strategic action plans to prevent veteran suicide.7,21-24
In addition to partnerships, VHA has built other aspects of outreach and intervention into its programming. A key VHA initiative is to “know all veterans” by committing to identifying and reaching out to all veterans who may be at risk for suicide.22 The VHA has committed to offering “emergency stabilization care for former service members who present at the facility with an emergent mental health need” regardless of eligibility.25 The intent is to provide temporary emergent mental health care to veterans who are otherwise ineligible for care, such as those who were discharged under other-than-honorable conditions while the VHA determines eligibility status.26 However, veterans must meet certain criteria, and there is a limit on services.
Although services are being expanded to reach veterans who do not access VHA health care, how to best implement these new directives with regard to suicide prevention is unclear. Strategic development and innovations to expand suicide prevention care to veterans outside the current reach of VHA are desperately needed.
Program Overview
VHA Patient Safety Center of Inquiry-Suicide Prevention Collaborative (PSCI-SPC), funded by the VHA National Center for Patient Safety, aims to help fill the gap in community-based suicide prevention for veterans. PSCI-SPC is located within the VHA Rocky Mountain Mental Illness Research, Education, and Clinical Center in Aurora, Colorado. The overarching mission of PSCI-SPC is to develop, implement, and evaluate practical solutions to reduce suicide among veterans not receiving VHA care. PSCI-SPC serves as a national clinical innovation and dissemination center for best practices in suicide prevention for organizations that serve veterans who receive care in the community. PSCI-SPC creates products to support dissemination of these practices to other VAMCs and works to ensure these programs are sustainable. PSCI-SPC focuses on 3 primary objectives. All PSCI-SPC projects are currently underway.
Objective 1: Growing a Community Learning Collaborative
Acknowledging that nearly two-thirds of veterans who die by suicide do not use VHA services, PSCI-SPC aims to reduce suicide among all veterans by expanding the reach of best practices for suicide prevention to veterans who receive myriad services in the community.27 Community organizations are defined here as organizations that may in some way serve, interact with, or work with veterans, and/or employ veterans. Examples include non-VHA health care systems, public services such as police and fire departments, nonprofit organizations, mental health clinics, and veterans’ courts. As veterans increasingly seek health care and other services within their communities, the success of suicide prevention will be influenced by the capability of non-VHA public and private organizations. Objective 1, therefore, seeks to develop a VHA-community collaborative that can be leveraged to improve systems of suicide prevention.
Current programs in the VHA have focused on implementation of suicide prevention awareness and prevention education campaigns instead of grassroots partnerships that are intended to be sustainable. Additionally, these programs typically lack the capacity and systems to sustain numerous meaningful community partnerships. Traditionally, community organizations have been hesitant to partner with government agencies, such as the VHA, due to histories of institutional mistrust and bureaucracy.28
The PSCI-SPC model for developing a VHA-community collaborative partnership draws from the tradition of community-based participatory research. The best community-based participatory research practices are to build on strengths and resources within the local community; develop collaborative, equitable partnerships that involve an empowering and power-sharing process; foster colearning, heuristics, and capacity building among partners; and focus on systems development using an iterative process. These practices also are consistent with the literature on learning collaboratives.29-31
The premise for a learning collaborative is to bridge the gap between knowledge and practice in health care.31 Figure 1 depicts how this collaborative was developed, and how it supports Objectives 2 and 3. To achieve Objective 1, we developed a VHA-learning collaborative of 13 influential community partners in the Denver and Colorado Springs region of Colorado. The VHA team consists of a learning collaborative leader, a program manager, and a program support assistant. The principal investigator attends and contributes to all meetings. Learning collaborative partners include a university psychology clinic that focuses on veterans’ care, 3 veterans service organizations, a mental health private practice, a university school of nursing, a community mental health center, veterans’ courts, and 5 city departments.
These partners participated in qualitative interviews to identify where gaps and breakdowns were occurring. With this information, the PSCI-SPC team and VHA-learning collaborative held a kickoff event. At this meeting the team discussed the qualitative findings, provided veteran suicide prevention information, and basic information regarding suicide prevention program building and implementation science.
Throughout quarterly learning collaborative meetings and monthly facilitation calls, we have worked to develop a blueprint and an action plan for each partner to develop best practices for dissemination to aid in providing consistency in the standard of care. A postimplementation event will be held to identify successes and challenges encountered while operationalizing project action plans.
Currently, activities of the learning collaborative are making a large impact on the community. Not all collaborative members track information regarding their populations served, nor specific metrics on veterans encountered. Even so, of those who do capture metrics, suicide prevention program components implemented by the collaborative will impact more than 21,000 individuals and at least 2,500 known veterans. In addition, 52 new connections have been made between community organizations or between community organizations and the VHA, and > 300 individuals have been trained.The learning collaborative support team has assisted in the dissemination of a large resource list for veterans. As the learning collaborative is ongoing and we are working with organizations to improve their data collection and analytics, we expect these numbers to increase. We anticipate that the learning collaborative will develop a stronger suicide prevention safety net within the community. In addition, we expect increased referrals of at-risk veterans to the VHA and enhance the long-term continuity of care between community and VHA services.
Objective 2: Implementation Toolkit
The second PSCI-SPC objective is to develop a toolkit for the implementation of best practices within a VHA-community suicide prevention learning collaborative. Lessons from the development of a successful suicide prevention learning collaborative will be shared through an online guide that other VHA facilities can use to support similar collaborative efforts within their communities. The toolkit will be disseminated across the VHA to assist suicide prevention coordinators and other staff in developing a suicide prevention learning collaborative at their facilities.
PSCI-SPC uses the Zero Suicide framework and the VA/US Department of Defense (DoD) Clinical Practice Guideline for the Assessment and Management of Patients at Risk for Suicide as models for preventing suicide in veterans not enrolled in VHA care.11,32 This implementation toolkit focuses on how to implement suicide prevention best practices into organizations that serve veterans. This toolkit differs from clinical practice guidelines in that it focuses on implementation strategies to promote success and effectively address challenges.
In order to provide a menu of available options for the learning collaborative and resulting toolkit, PSCI-SPC uses a logic model to compare the components of the VHA suicide prevention program, as well as other similar veteran and military suicide prevention programs.7,12,14,21,33,34 These programs are categorized into 2 types of prevention frameworks, the USI model as described above, and the SAMHSA Strategic Prevention Framework (Table 2).35 The SAMHSA framework was designed to promote mental health and prevent substance abuse, yet the derived classification is also applicable to suicide prevention programs.35 The results of the logic model comparison form the basis of the best practice interventions for the learning collaborative and initial toolkit. In addition to the best practice interventions, the toolkit consists of documents describing how to develop a veteran suicide prevention learning collaborative, as well as tools for learning collaborative members. Current tool development includes workbooks to guide collaborative members through the implementation process, guides for community organizations in implementing suicide prevention screening and risk assessment, a standard operating procedure for suicide prevention in a veterans court, and peer support training for veteran suicide prevention.
The methods to achieve Objective 2 include regular discussions with the VHA-learning collaborative on current best practices, identifying gaps and overlap of community programs. Successes and challenges of implementing suicide prevention best practices into learning collaborative organizations will be documented and incorporated into the toolkit. The learning collaborative will work iteratively as a team to improve the toolkit. Once complete, the toolkit will be disseminated to other VHA health care systems nationally, as well as to other state or regional partners that the learning collaborative identifies. A plan is under development for national suicide prevention entities to also disseminate the toolkit to lessen the burden of veteran suicide through their stakeholder base.
Objective 3: High-Risk Veterans Not Receiving VHA Care
Although veterans not receiving VHA care account for a number of veteran deaths by suicide, we are not aware of any current VHA programs that provide temporary psychotherapy and intensive case management to at-risk veterans ineligible for VHA care who are in need of immediate care while an appropriate permanent community placement is identified. In the current system, veterans in the community can present to VHA suicide prevention services through several different systems, including referrals to VHA and the Veterans Crisis Line (VCL). However, a portion of VCL calls are from veterans whose VHA eligibility is unknown or who are ineligible for services. If veterans are at imminent risk for suicide, emergency care is coordinated for them. However, if veterans are not at imminent suicide risk they are referred to the local suicide prevention coordinator and instructed to independently work toward determining their VHA eligibility.
It is currently unknown how many veterans follow through with these instructions. Nonetheless, if veterans are deemed eligible, they may present to VHA to obtain a same-day appointment. If not eligible, a suicide prevention coordinator may give them the phone number of a community referral. However, this practice is not standardized across VA medical centers, and the provided resources are up to the suicide prevention coordinator to research. Additionally, when a VHA suicide prevention coordinator leaves the position, knowledge of these community resources and rapport with community HCPs are often lost, leaving the next coordinator to develop these again, which reduces the efficiency and effectiveness of limited resources. It is also unknown how many veterans complete this contact and receive evidence-based treatment following referral. This is a complex system to navigate, particularly when at risk for suicide and in need of immediate but not emergency services.
Suicide prevention in such circumstances may be improved by adapting current suicide prevention practices, including evidence-based interventions, and the new VHA intensive case management program,11,36 within a Zero Suicide framework. PSCI-SPC has developed a brief intervention to transition ineligible veterans to permanent community treatment and provide them with additional resources to meet their varied needs. The brief 1 to 3 session intervention combines practices from brief cognitive behavioral therapy (BCBT) for suicide prevention, crisis response planning (CRP), and intensive case management within a Zero Suicide framework. Both the 2019 VA/DoD suicide prevention clinical practice guidelines and Zero Suicide recommend using cognitive behavioral therapy (CBT)-based interventions for suicide prevention.11,32 These interventions are packaged into a single intervention delivered by a PSCI-SPC therapist, typically a licensed clinical social worker, a licensed clinical psychologist, or an unlicensed psychologist under the supervision of a licensed clinical psychologist.
BCBT is one type of CBT that has shown initial efficacy in reducing suicide attempts.37 BCBT reduces the risk for suicide attempts both at the conclusion of treatment and at 24-month follow-up.37 BCBT is boiled down to its most essential components so it can be delivered in a distilled format. An essential element of BCBT that will remain is the CRP. A CRP11,37,38 entails collaboratively identifying effective, appropriate coping strategies and specific individuals to contact during a crisis. CRPs demonstrated efficacy as a stand-alone intervention to existing suicide prevention methods in a randomized clinical trial, such that individuals who received CRP had faster reductions in suicidal ideation and were 76% less likely to make a suicide attempt during the 6-month follow-up period.39 These results demonstrate that use of a CRP is connected to a decrease in suicidal behavior among suicidal patients.
The VHA has developed and is piloting a new initiative focused on restructuring its intensive case management services. RACETIME to Integrated Care (eg, Risk stratification, Assessment of complexity, Coordinator of lead assignment, Evaluate whole health needs, Trusting partnerships, Integrate care, Monitor progress, Experience of the veteran and employee) is a framework that assists VHA case managers in transitioning from a traditional case management mind-set to a more integrated and holistic method of care.36 RACETIME intensive case management practices will be incorporated into the intervention. However, RACETIME focuses on case management internally to the VHA. A modification for this treatment will be to focus on intensive case management from a mental health perspective and connecting to external community resources. Community referrals are mapped within a structured process and stored on a shared drive. This improves continuity between suicide prevention coordinators when they leave for a new position.
This intervention is conducted within a Zero Suicide framework. Pertinent to PSCI-SPC innovation to enhance care for non-VHA veterans is the care transitions element within the Zero Suicide framework, which has developed comprehensive suicide prevention guidance, including a pathway to care.32 This pathway refers a process to conduct follow-up supportive contacts that are tracked and recorded.
The PSCI-SPC pilot program incorporates the elements of CRP and brief CBT within a Zero Suicide framework. The PSCI-SPC team is developing and testing a protocol for providing brief treatment and community referrals to ineligible veterans that integrates these programming elements (Figure 2). A PSCI-SPC social worker will coordinate with the eligibility office to determine VHA eligibility. Ineligible veterans are referred to community partners and nonenrolled, eligible veterans are linked to VHA HCPs if they desire. These transitions will be coordinated, closely monitored, and verified.
Once the eligibility is determined, and the veteran’s preference is assessed, the team will perform a warm handoff to a VHA representative for enrollment into VHA care or to a community placement. We have sourced multiple community placements at varied payor levels. All veterans who are ineligible for care or who do not desire to be enrolled in VHA care will be provided the intervention package described above. After the veteran is placed in community care, PSCI-SPC team will follow up regularly with the veteran for 3 months to ensure continuity of care. If additional sessions are needed while the veteran needs a community placement, the PSCI team can accommodate this and will track the number of participants who needed additional sessions. If the veteran is deemed to be at imminent risk for suicide at any time during the pilot, he/she is transferred to emergency care. The veteran is allowed to participate once stabilized.
Findings from the pilot program will inform a manual intended for dissemination to patient safety and suicide prevention coordinators nationally across the VHA. The products from this objective will be integrated with the other objectives in training that will be provided on how to develop a local collaborative (Objective 1), disseminate the toolkit (Objective 2), and receive referrals into VHA or refer veterans to community clinical partners through the pilot program (Objective 3). Products developed and disseminated throughout PSCI-SPC aim to create momentum to reduce rates of suicide in veterans who are not connected to the VHA at the local community level.
Discussion
Improving suicide prevention for veterans who receive non-VHA health care is essential to significantly reduce veteran suicide rates. For the past decade, VHA suicide prevention initiatives have largely focused on veterans eligible for care, although the fastest increase in veteran suicide rates has occurred among veterans not connected to VHA services. Currently, if a veteran is deemed ineligible for care, it is up to the veteran to find other health care services in his or her community. There is not always a clear next step for the veteran to take, nor clear guidance provided to the VHA registration staff to assist with this care transition. This is particularly concerning for veterans at high risk for suicide as this could further thwart the veteran’s sense of belongingness and increase perceived burdensomeness, both suicide risk factors, and discourage them from attaining help.40 Overall, while the VHA has successfully implemented diverse suicide prevention initiatives and services, the need for continued system improvement focused on non-VHA veterans remains. PSCI-SPC was developed for this purpose.
By creating a collaborative that will connect VHA and community organizations, there will be better utilization of resources and more appropriate referrals throughout systems that interact with veterans. Sharing suicide prevention best practices between VHA and community partners is expected to increase access to mental health treatment to all veterans. Finally, by allowing best practices for suicide prevention in the VHA to serve as a guide in the development of best practices for suicide prevention between the VHA and the local health and behavioral health care community, PSCI-SPC will create a new suicide prevention intervention for veterans with mental health needs. Through these initiatives, PSCI-SPC will support providers’ and concerned citizens’ efforts to ensure that fewer veterans fall through the cracks of disjointed systems and will promote healthier communities where, regardless of VHA enrollment status, veterans receive suicide prevention care.
Conclusions
PSCI-SPC is a novel center for the innovation and dissemination of the nation’s best practices in suicide prevention for veterans who are ineligible for or otherwise not engaged in VHA services and who turn to their community for health care. PSCI-SPC not only seeks to create, develop, and measure various solutions to reduce suicide among veterans who receive non-VHA care, but also seeks to facilitate the overall quality of existing practices for suicide prevention and care coordination for enrolled veterans who use community resources. By bridging the gap between the VHA, civilian health care systems, and other community partners striving to prevent veteran suicides, we can create better access to care and a more seamless path of communication among these important entities that impact the lives of our veterans daily
Since 2008, suicide has ranked as the tenth leading cause of death for all ages in the US, with rates of suicide continuing to rise.1-3 Suicide is even more urgent to address in veteran populations. The age- and sex-adjusted suicide rate in 2017 was more than 1.5 times greater for veterans than it was for nonveteran adults.2 Of importance, rates of suicide are increasing at a faster rate in veterans who are not connected to Veterans Health Administration (VHA) care.4,5 These at-risk veterans include individuals who are eligible for VHA care yet have not had a VHA appointment within the year before death; veterans who may be ineligible to receive VHA care due to complex rules set by legislation; and veterans who are eligible but not enrolled in VHA care. Notably, between 2005 and 2016, the number of veterans not enrolled in VHA care rose more quickly than did the number of veterans enrolled in VHA care.5,6 Thus, to impact the high veteran suicide rates, an emergent challenge for VHA is to prevent suicide among unenrolled veterans and veterans receiving community care, while continuing to increase access to mental health services for veterans enrolled in VHA health care.
In response to the high rates of veteran suicide deaths, the US Department of Veterans Affairs (VA) has developed a broad, multicomponent suicide prevention program that is unparalleled in private US health care systems.4,7 Suicide prevention efforts are led and implemented by both the VHA National Center for Patient Safety and the VHA Office of Mental Health and Suicide Prevention. Program components are numerous and multifaceted, falling within the broad promotion and prevention strategies outlined by the National Academy of Medicine (NAM).1,8-11 The NAM continuum of prevention model encompassing multiple strategies is also referred to as the Universal, Selective, Indicated (USI) Model.7,8,10 The VHA suicide prevention program contains a wide spread of program components, making it both comprehensive and innovative (Table 1).
Although significant momentum and progress has been made within the VHA, policy set by legislation has historically limited access to VHA health care services to VHA-eligible veterans. This is particularly concerning given the rising suicide rates among veterans not engaged in VHA care.2 Adding to this complexity, recent legislation has increased veterans’ access to non-VHA health care, in addition to their existing access through Medicare, Medicaid, and other health care programs.12-14 Best practices for suicide prevention are not often implemented in the private sector; thus, these systems are ill prepared to adequately meet the suicide prevention care needs of veterans.4,15-18 Furthermore, VHA and non-VHA services generally are not well coordinated, and private sector health care providers (HCPs) are not required to complete a commensurate level of suicide prevention training as are VHA HCPs.16-18 Most non-VHA HCPs do not receive military cultural competence training.19 These issues create a significant gap in suicide prevention services and may contribute to the increases in suicide rates in veterans who do not receive VHA care. Thus, changes in policy to increase access through private sector care may have paradoxical effects on veteran suicide deaths. To impact the veteran suicide rate, VHA must develop and disseminate best practices for veterans who use non-VHA services.
A Roadmap to Suicide Prevention
There is significant momentum at the federal level regarding this issue. The President’s Roadmap to Empower Veterans and End the National Tragedy of Suicide (Executive Order 13,861) directs the VHA to work closely with community organizations to improve veteran suicide prevention.20 The VHA and partners, such as the Substance Abuse and Mental Health Services Administration (SAMHSA), are bridging this gap with collaborative efforts that increase suicide prevention resources for veterans living in the community through programs such as the Governor’s Challenges to Prevent Suicide Among Service Members, Veterans, and their Families. These programs intend to empower communities to develop statewide, strategic action plans to prevent veteran suicide.7,21-24
In addition to partnerships, VHA has built other aspects of outreach and intervention into its programming. A key VHA initiative is to “know all veterans” by committing to identifying and reaching out to all veterans who may be at risk for suicide.22 The VHA has committed to offering “emergency stabilization care for former service members who present at the facility with an emergent mental health need” regardless of eligibility.25 The intent is to provide temporary emergent mental health care to veterans who are otherwise ineligible for care, such as those who were discharged under other-than-honorable conditions while the VHA determines eligibility status.26 However, veterans must meet certain criteria, and there is a limit on services.
Although services are being expanded to reach veterans who do not access VHA health care, how to best implement these new directives with regard to suicide prevention is unclear. Strategic development and innovations to expand suicide prevention care to veterans outside the current reach of VHA are desperately needed.
Program Overview
VHA Patient Safety Center of Inquiry-Suicide Prevention Collaborative (PSCI-SPC), funded by the VHA National Center for Patient Safety, aims to help fill the gap in community-based suicide prevention for veterans. PSCI-SPC is located within the VHA Rocky Mountain Mental Illness Research, Education, and Clinical Center in Aurora, Colorado. The overarching mission of PSCI-SPC is to develop, implement, and evaluate practical solutions to reduce suicide among veterans not receiving VHA care. PSCI-SPC serves as a national clinical innovation and dissemination center for best practices in suicide prevention for organizations that serve veterans who receive care in the community. PSCI-SPC creates products to support dissemination of these practices to other VAMCs and works to ensure these programs are sustainable. PSCI-SPC focuses on 3 primary objectives. All PSCI-SPC projects are currently underway.
Objective 1: Growing a Community Learning Collaborative
Acknowledging that nearly two-thirds of veterans who die by suicide do not use VHA services, PSCI-SPC aims to reduce suicide among all veterans by expanding the reach of best practices for suicide prevention to veterans who receive myriad services in the community.27 Community organizations are defined here as organizations that may in some way serve, interact with, or work with veterans, and/or employ veterans. Examples include non-VHA health care systems, public services such as police and fire departments, nonprofit organizations, mental health clinics, and veterans’ courts. As veterans increasingly seek health care and other services within their communities, the success of suicide prevention will be influenced by the capability of non-VHA public and private organizations. Objective 1, therefore, seeks to develop a VHA-community collaborative that can be leveraged to improve systems of suicide prevention.
Current programs in the VHA have focused on implementation of suicide prevention awareness and prevention education campaigns instead of grassroots partnerships that are intended to be sustainable. Additionally, these programs typically lack the capacity and systems to sustain numerous meaningful community partnerships. Traditionally, community organizations have been hesitant to partner with government agencies, such as the VHA, due to histories of institutional mistrust and bureaucracy.28
The PSCI-SPC model for developing a VHA-community collaborative partnership draws from the tradition of community-based participatory research. The best community-based participatory research practices are to build on strengths and resources within the local community; develop collaborative, equitable partnerships that involve an empowering and power-sharing process; foster colearning, heuristics, and capacity building among partners; and focus on systems development using an iterative process. These practices also are consistent with the literature on learning collaboratives.29-31
The premise for a learning collaborative is to bridge the gap between knowledge and practice in health care.31 Figure 1 depicts how this collaborative was developed, and how it supports Objectives 2 and 3. To achieve Objective 1, we developed a VHA-learning collaborative of 13 influential community partners in the Denver and Colorado Springs region of Colorado. The VHA team consists of a learning collaborative leader, a program manager, and a program support assistant. The principal investigator attends and contributes to all meetings. Learning collaborative partners include a university psychology clinic that focuses on veterans’ care, 3 veterans service organizations, a mental health private practice, a university school of nursing, a community mental health center, veterans’ courts, and 5 city departments.
These partners participated in qualitative interviews to identify where gaps and breakdowns were occurring. With this information, the PSCI-SPC team and VHA-learning collaborative held a kickoff event. At this meeting the team discussed the qualitative findings, provided veteran suicide prevention information, and basic information regarding suicide prevention program building and implementation science.
Throughout quarterly learning collaborative meetings and monthly facilitation calls, we have worked to develop a blueprint and an action plan for each partner to develop best practices for dissemination to aid in providing consistency in the standard of care. A postimplementation event will be held to identify successes and challenges encountered while operationalizing project action plans.
Currently, activities of the learning collaborative are making a large impact on the community. Not all collaborative members track information regarding their populations served, nor specific metrics on veterans encountered. Even so, of those who do capture metrics, suicide prevention program components implemented by the collaborative will impact more than 21,000 individuals and at least 2,500 known veterans. In addition, 52 new connections have been made between community organizations or between community organizations and the VHA, and > 300 individuals have been trained.The learning collaborative support team has assisted in the dissemination of a large resource list for veterans. As the learning collaborative is ongoing and we are working with organizations to improve their data collection and analytics, we expect these numbers to increase. We anticipate that the learning collaborative will develop a stronger suicide prevention safety net within the community. In addition, we expect increased referrals of at-risk veterans to the VHA and enhance the long-term continuity of care between community and VHA services.
Objective 2: Implementation Toolkit
The second PSCI-SPC objective is to develop a toolkit for the implementation of best practices within a VHA-community suicide prevention learning collaborative. Lessons from the development of a successful suicide prevention learning collaborative will be shared through an online guide that other VHA facilities can use to support similar collaborative efforts within their communities. The toolkit will be disseminated across the VHA to assist suicide prevention coordinators and other staff in developing a suicide prevention learning collaborative at their facilities.
PSCI-SPC uses the Zero Suicide framework and the VA/US Department of Defense (DoD) Clinical Practice Guideline for the Assessment and Management of Patients at Risk for Suicide as models for preventing suicide in veterans not enrolled in VHA care.11,32 This implementation toolkit focuses on how to implement suicide prevention best practices into organizations that serve veterans. This toolkit differs from clinical practice guidelines in that it focuses on implementation strategies to promote success and effectively address challenges.
In order to provide a menu of available options for the learning collaborative and resulting toolkit, PSCI-SPC uses a logic model to compare the components of the VHA suicide prevention program, as well as other similar veteran and military suicide prevention programs.7,12,14,21,33,34 These programs are categorized into 2 types of prevention frameworks, the USI model as described above, and the SAMHSA Strategic Prevention Framework (Table 2).35 The SAMHSA framework was designed to promote mental health and prevent substance abuse, yet the derived classification is also applicable to suicide prevention programs.35 The results of the logic model comparison form the basis of the best practice interventions for the learning collaborative and initial toolkit. In addition to the best practice interventions, the toolkit consists of documents describing how to develop a veteran suicide prevention learning collaborative, as well as tools for learning collaborative members. Current tool development includes workbooks to guide collaborative members through the implementation process, guides for community organizations in implementing suicide prevention screening and risk assessment, a standard operating procedure for suicide prevention in a veterans court, and peer support training for veteran suicide prevention.
The methods to achieve Objective 2 include regular discussions with the VHA-learning collaborative on current best practices, identifying gaps and overlap of community programs. Successes and challenges of implementing suicide prevention best practices into learning collaborative organizations will be documented and incorporated into the toolkit. The learning collaborative will work iteratively as a team to improve the toolkit. Once complete, the toolkit will be disseminated to other VHA health care systems nationally, as well as to other state or regional partners that the learning collaborative identifies. A plan is under development for national suicide prevention entities to also disseminate the toolkit to lessen the burden of veteran suicide through their stakeholder base.
Objective 3: High-Risk Veterans Not Receiving VHA Care
Although veterans not receiving VHA care account for a number of veteran deaths by suicide, we are not aware of any current VHA programs that provide temporary psychotherapy and intensive case management to at-risk veterans ineligible for VHA care who are in need of immediate care while an appropriate permanent community placement is identified. In the current system, veterans in the community can present to VHA suicide prevention services through several different systems, including referrals to VHA and the Veterans Crisis Line (VCL). However, a portion of VCL calls are from veterans whose VHA eligibility is unknown or who are ineligible for services. If veterans are at imminent risk for suicide, emergency care is coordinated for them. However, if veterans are not at imminent suicide risk they are referred to the local suicide prevention coordinator and instructed to independently work toward determining their VHA eligibility.
It is currently unknown how many veterans follow through with these instructions. Nonetheless, if veterans are deemed eligible, they may present to VHA to obtain a same-day appointment. If not eligible, a suicide prevention coordinator may give them the phone number of a community referral. However, this practice is not standardized across VA medical centers, and the provided resources are up to the suicide prevention coordinator to research. Additionally, when a VHA suicide prevention coordinator leaves the position, knowledge of these community resources and rapport with community HCPs are often lost, leaving the next coordinator to develop these again, which reduces the efficiency and effectiveness of limited resources. It is also unknown how many veterans complete this contact and receive evidence-based treatment following referral. This is a complex system to navigate, particularly when at risk for suicide and in need of immediate but not emergency services.
Suicide prevention in such circumstances may be improved by adapting current suicide prevention practices, including evidence-based interventions, and the new VHA intensive case management program,11,36 within a Zero Suicide framework. PSCI-SPC has developed a brief intervention to transition ineligible veterans to permanent community treatment and provide them with additional resources to meet their varied needs. The brief 1 to 3 session intervention combines practices from brief cognitive behavioral therapy (BCBT) for suicide prevention, crisis response planning (CRP), and intensive case management within a Zero Suicide framework. Both the 2019 VA/DoD suicide prevention clinical practice guidelines and Zero Suicide recommend using cognitive behavioral therapy (CBT)-based interventions for suicide prevention.11,32 These interventions are packaged into a single intervention delivered by a PSCI-SPC therapist, typically a licensed clinical social worker, a licensed clinical psychologist, or an unlicensed psychologist under the supervision of a licensed clinical psychologist.
BCBT is one type of CBT that has shown initial efficacy in reducing suicide attempts.37 BCBT reduces the risk for suicide attempts both at the conclusion of treatment and at 24-month follow-up.37 BCBT is boiled down to its most essential components so it can be delivered in a distilled format. An essential element of BCBT that will remain is the CRP. A CRP11,37,38 entails collaboratively identifying effective, appropriate coping strategies and specific individuals to contact during a crisis. CRPs demonstrated efficacy as a stand-alone intervention to existing suicide prevention methods in a randomized clinical trial, such that individuals who received CRP had faster reductions in suicidal ideation and were 76% less likely to make a suicide attempt during the 6-month follow-up period.39 These results demonstrate that use of a CRP is connected to a decrease in suicidal behavior among suicidal patients.
The VHA has developed and is piloting a new initiative focused on restructuring its intensive case management services. RACETIME to Integrated Care (eg, Risk stratification, Assessment of complexity, Coordinator of lead assignment, Evaluate whole health needs, Trusting partnerships, Integrate care, Monitor progress, Experience of the veteran and employee) is a framework that assists VHA case managers in transitioning from a traditional case management mind-set to a more integrated and holistic method of care.36 RACETIME intensive case management practices will be incorporated into the intervention. However, RACETIME focuses on case management internally to the VHA. A modification for this treatment will be to focus on intensive case management from a mental health perspective and connecting to external community resources. Community referrals are mapped within a structured process and stored on a shared drive. This improves continuity between suicide prevention coordinators when they leave for a new position.
This intervention is conducted within a Zero Suicide framework. Pertinent to PSCI-SPC innovation to enhance care for non-VHA veterans is the care transitions element within the Zero Suicide framework, which has developed comprehensive suicide prevention guidance, including a pathway to care.32 This pathway refers a process to conduct follow-up supportive contacts that are tracked and recorded.
The PSCI-SPC pilot program incorporates the elements of CRP and brief CBT within a Zero Suicide framework. The PSCI-SPC team is developing and testing a protocol for providing brief treatment and community referrals to ineligible veterans that integrates these programming elements (Figure 2). A PSCI-SPC social worker will coordinate with the eligibility office to determine VHA eligibility. Ineligible veterans are referred to community partners and nonenrolled, eligible veterans are linked to VHA HCPs if they desire. These transitions will be coordinated, closely monitored, and verified.
Once the eligibility is determined, and the veteran’s preference is assessed, the team will perform a warm handoff to a VHA representative for enrollment into VHA care or to a community placement. We have sourced multiple community placements at varied payor levels. All veterans who are ineligible for care or who do not desire to be enrolled in VHA care will be provided the intervention package described above. After the veteran is placed in community care, PSCI-SPC team will follow up regularly with the veteran for 3 months to ensure continuity of care. If additional sessions are needed while the veteran needs a community placement, the PSCI team can accommodate this and will track the number of participants who needed additional sessions. If the veteran is deemed to be at imminent risk for suicide at any time during the pilot, he/she is transferred to emergency care. The veteran is allowed to participate once stabilized.
Findings from the pilot program will inform a manual intended for dissemination to patient safety and suicide prevention coordinators nationally across the VHA. The products from this objective will be integrated with the other objectives in training that will be provided on how to develop a local collaborative (Objective 1), disseminate the toolkit (Objective 2), and receive referrals into VHA or refer veterans to community clinical partners through the pilot program (Objective 3). Products developed and disseminated throughout PSCI-SPC aim to create momentum to reduce rates of suicide in veterans who are not connected to the VHA at the local community level.
Discussion
Improving suicide prevention for veterans who receive non-VHA health care is essential to significantly reduce veteran suicide rates. For the past decade, VHA suicide prevention initiatives have largely focused on veterans eligible for care, although the fastest increase in veteran suicide rates has occurred among veterans not connected to VHA services. Currently, if a veteran is deemed ineligible for care, it is up to the veteran to find other health care services in his or her community. There is not always a clear next step for the veteran to take, nor clear guidance provided to the VHA registration staff to assist with this care transition. This is particularly concerning for veterans at high risk for suicide as this could further thwart the veteran’s sense of belongingness and increase perceived burdensomeness, both suicide risk factors, and discourage them from attaining help.40 Overall, while the VHA has successfully implemented diverse suicide prevention initiatives and services, the need for continued system improvement focused on non-VHA veterans remains. PSCI-SPC was developed for this purpose.
By creating a collaborative that will connect VHA and community organizations, there will be better utilization of resources and more appropriate referrals throughout systems that interact with veterans. Sharing suicide prevention best practices between VHA and community partners is expected to increase access to mental health treatment to all veterans. Finally, by allowing best practices for suicide prevention in the VHA to serve as a guide in the development of best practices for suicide prevention between the VHA and the local health and behavioral health care community, PSCI-SPC will create a new suicide prevention intervention for veterans with mental health needs. Through these initiatives, PSCI-SPC will support providers’ and concerned citizens’ efforts to ensure that fewer veterans fall through the cracks of disjointed systems and will promote healthier communities where, regardless of VHA enrollment status, veterans receive suicide prevention care.
Conclusions
PSCI-SPC is a novel center for the innovation and dissemination of the nation’s best practices in suicide prevention for veterans who are ineligible for or otherwise not engaged in VHA services and who turn to their community for health care. PSCI-SPC not only seeks to create, develop, and measure various solutions to reduce suicide among veterans who receive non-VHA care, but also seeks to facilitate the overall quality of existing practices for suicide prevention and care coordination for enrolled veterans who use community resources. By bridging the gap between the VHA, civilian health care systems, and other community partners striving to prevent veteran suicides, we can create better access to care and a more seamless path of communication among these important entities that impact the lives of our veterans daily
1. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published September 2018. Accessed October 14, 2020.
2. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National veteran suicide prevention annual report. https://www.mentalhealth.va.gov/suicide_prevention/data.asp. Accessed October 14, 2020.
3. Centers for Disease Control and Prevention. Web-based injury statistics query and reporting system (WISQARS). 2014. https://www.cdc.gov/injury/wisqars/index.html. Updated July 1, 2020. Accessed October 14, 2020.
4. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health services. Fed Pract. 2018;35(3):18-24.
5. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. VA national suicide data report 2005-2016. https://www.mentalhealth.va.gov/docs/data-sheets/OMHSP_National_Suicide_Data_Report_2005-2016_508.pdf. Published September 2018. Accessed October 14, 2020.
6. US Department of Veterans Affairs, Office of Suicide Prevention. VA national suicide data report 2005-2015. https://www.mentalhealth.va.gov/docs/data-sheets/2015/OMHSP_National_Suicide_Data_Report_2005-2015_06-14-18_508.pdf. Published June 2018. Accessed October 14, 2020.
7. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published 2018. Accessed October 30, 2020.
8. Gordon RS. An operational classification of disease prevention. Public Health Rep. 1983;98(2):107-109.
9. National Research Council and Institute of Medicine. O’Connell ME, Boat T, Warner KE, eds. Preventing Mental, Emotional, and Behavioral Disorders Among Young People: Progress and Possibilities. Washington, DC: National Academies Press; 2009.
10. US Department of Health and Human Services, National Institute on Drug Abuse, Office of Science Policy and Communications. Drug Abuse Prevention: What Works. Rockville, MD: National Institutes of Health; 1997.
11. US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the assessment and management of patients at risk for suicide. https://www.healthquality.va.gov/guidelines/MH/srb. Published 2019. Updated July 30, 2020. Accessed October 14, 2020.
12. US Department of Veterans Affairs. Veterans Access, Choice and Accountability Act of 2014. https://www.govinfo.gov/content/pkg/PLAW-113publ146/pdf/PLAW-113publ146.pdf. Accessed October 14, 2020.
13. US Department of Veterans Affairs. Strategic Health Care. VA MISSION Act: Extension of the VA choice program. https://strategichealthcare.net/wp-content/uploads/2018/05/052718-VA-MISSION-Act-Summary.pdf. Accessed October 14, 2020.
14. US Congress. H.R.5674—VA MISSION Act of 2018. https://www.congress.gov/bill/115th-congress/house-bill/5674. Accessed October 15, 2020.
15. Grumet JG, Hogan MF, Chu A, Covington DW, Johnson KE. Compliance standards pave the way for reducing suicide in health care systems. J Health Care Compliance. 2019;17-26.
16. Hillestad R, Bigelow J, Bower A, et al. Can electronic medical record systems transform health care? Potential health benefits, savings, and costs. Health Aff (Millwood). 2005;24(5):1103-1117. doi:10.1377/hlthaff.24.5.110
17. Porter ME, Pabo EA, Lee TH. Redesigning primary care: a strategic vision to improve value by organizing around patients’ needs. Health Aff (Millwood). 2013;32(3):516-525. doi:10.1377/hlthaff.2012.0961
18. Nevedal AL, Wagner TH, Ellerbe LS, Asch SM, Koenig CJ. A qualitative study of primary care providers’ experiences with the Veterans Choice Program. J Gen Intern Med. 2019;34:598-603. doi:10.1007/s11606-018-4810-2
19. Tanielian T, Farris C, Epley C, et al. Ready to serve: community-based provider capacity to deliver culturally competent, quality mental health care to veterans and their families. https://www.rand.org/content/dam/rand/pubs/research_reports/RR800/RR806/RAND_RR806.pdf. Published 2014. Accessed October 15, 2020.
20. White House. Executive Order 13861: President’s roadmap to empower veterans and end the national tragedy of suicide (PREVENTS). https://www.va.gov/PREVENTS/docs/PRE-007-The-PREVENTS-Roadmap-1-2_508.pdf. Published June 17, 2020. Accessed October 15, 2020.
21. US Department of Veterans Affairs, Office of the Under Secretary for Health. Memorandum. VA continues community suicide prevention challenge at another mayor’s challenge policy academy. https://www.blogs.va.gov/VAntage/58468/va-continues-community-suicide-prevention-challenge-another-mayors-challenge-policy-academy. Published April 4, 2019. Accessed October 15, 2020.
22. US Department of Veterans Affairs, Veterans Health Administration (VHA) Veterans Health Administration (VHA) Fiscal Year (FY) 2018-2019 Operational Plan. www.navao.org/wp-content/uploads/2018/06/USH-Memo-10-2018-03-VHA_FY_2018-2019_Operational_Plan-4-19-18.pdf. Published April 19, 2018. Accessed October 15, 2020.
23. US Department of Veterans Affairs. VA, Health and Human Services announce governor’s challenge to prevent suicide. https://www.blogs.va.gov/VAntage/55707/va-health-human-services-announce-governors-challenge-prevent-suicide. Published January 17, 2019. Accessed October 15, 2020.
24. VA’s suicide prevention campaign “Be There’’ kicks off in Times Square. https://www.blogs.va.gov/VAntage/57272/vas-suicide-prevention-campaign-kicks-off-times-square. Published March 6, 2019. Accessed October 15, 2020.
25. US Department of Veterans Affairs, Office of Public Affairs Media Relations. Emergent mental health care for former service members. https://www.mentalhealth.va.gov/docs/Fact_Sheet-Emergent_Mental_Health_Care_Former_Service_Members.pdf. Published 2017. Accessed October 15, 2020.
26. US Department of Veterans Affairs. VA secretary formalizes expansion of emergency mental health care to former service members with other-than-honorable discharges. https://www.blogs.va.gov/VAntage/39092/va-secretary-formalizes-expansion-emergency-mental-health-care-former-service-members-honorable-discharges. Published June 27, 2017. Accessed October 15, 2020.
27. Shane L. New veteran suicide numbers raise concerns among experts hoping for positive news. Military Times. https://www.militarytimes.com/news/pentagon-congress/2019/10/09/new-veteran-suicide-numbers-raise-concerns-among-experts-hoping-for-positive-news. Published October 9, 2019. Accessed October 15, 2020.
28. US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 Strategic Plan. https://www.va.gov/oei/docs/VA2018-2024strategicPlan.pdf. Updated May 31, 2019. Accessed October 15, 2020.
29. Komaie G, Goodman M, McCall A, et al. Training community members in public health research: development and implementation of a community participatory research pilot project. Health Equity. 2018;2(1):282-287. doi:10.1089/heq.2018.0043
30. Wang KH, Ray NJ, Berg DN, et al. Using community-based participatory research and organizational diagnosis to characterize relationships between community leaders and academic researchers. Prev Med Reports. 2017;7:180-186. doi:10.1016/j.pmedr.2017.06.007
31. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed October 15, 2020.
32. Zero Suicide in Health and Behavioral Health Care Institute. Zero suicide toolkit. https://zerosuicide.sprc.org/toolkit. Accessed October 15, 2020.

33. Preventing suicide: a technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicideTechnicalPackage.pdf. Published 2017. Accessed October 30, 2020.
34. US Department of Veterans Affairs. VA Office of Mental Health and Suicide Prevention guidebook. https://www.mentalhealth.va.gov/docs/VA-Office-of-Mental-Health-and-Suicide-Prevention-Guidebook-June-2018-FINAL-508.pdf. Published June 2018. Accessed October 15, 2020.
35. US Department of Health and Human Services, Substance Abuse and Mental Health Services, Administration Center for Substance Abuse Prevention. Focus on prevention: strategies and programs to prevent substance abuse. HHS Publication No. (SMA) 10-4120. https://helpandhopewv.org/docs/Focus%20on%20Prevention.pdf. Revised 2017. Accessed October 15, 2020.
36. Misiti R, Risinger A. RACETIME: an organizational care coordination approach to improving patient outcomes in a complex healthcare setting. Presentation at the Military Social Work & Behavioral Health Conference Austin, Texas. July 23, 2020. https://sites.utexas.edu/military-social-work-conference/misiti-risinger. Accessed October 23, 2020.
37. Rudd MD, Bryan CJ, Wertenberger EG, et al. Brief cognitive-behavioral therapy effects on post-treatment suicide attempts in a military sample: results of a randomized clinical trial with 2-year follow-up. Am J Psychiatry. 2015;172(5):441-449. doi:10.1176/appi.ajp.2014.14070843
38. Rudd MD, Mandrusiak M, Joiner TE Jr. The case against no-suicide contracts: The commitment to treatment statement as a practice alternative. Clin Psychol. 2006;62(2):243-251.
39. Bryan CJ, Mintz J, Clemans TA, et al. Effect of crisis response planning vs. contracts for safety on suicide risk in US Army soldiers: a randomized clinical trial. J Affect Disord. 2017;212:64-72. doi:10.1016/j.jad.2017.01.028
40. Joiner TE. Why People Die by Suicide. Cambridge, MA: First Harvard University Press; 2005:27.
41. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.16: Defense suicide prevention program. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649016p.pdf?ver=2020-09-11-122632-850. Published November 6, 2017. Updated September 11, 2020. Accessed October 14, 2020.
42. Zero Suicide in Health and Behavioral Health Care Institute. Engage: engaging patients. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=0. Accessed October 14, 2020.
43. Stone D, Holland K, Bartholow B, Crosby A, Davis S, Wilkins N. Preventing Suicide: A technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicidetechnicalpackage.pdf. Published 2017. Accessed October 14, 2020.
44. Mills PD, Watts BV, Miller S, et al. A checklist to identify inpatient suicide hazards in veterans affairs hospitals. Jt Comm J Qual Patient Saf. 2010;36(2):87-93. doi:10.1016/s1553-7250(10)36015-6
45. US Department of Veterans Affairs, Office of Research and Development. Crisis prevention: study evaluates VA program that identifies vets at highest risk for suicide. https://www.research.va.gov/currents/0918-Study-evaluates-VA-program-that-identifies-Vets-at-highest-risk-for-suicide.cfm. Updated September, 20, 2018. Accessed October 14, 2020.
46. Zero Suicide in Health and Behavioral Health Care Institute. Engage: active engagement for safer suicide care--reducing access to lethal means. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Published 2017. Accessed October 14, 2020.
47. Zero Suicide in Health and Behavioral Health Care Institute. Train: training for all. http://zerosuicide.sprc.org/toolkit/train. Accessed October 14, 2020.
48. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention (OMHSP). Facts about veteran suicide. https://www.mentalhealth.va.gov/docs/FINAL_VA_OMHSP_Suicide_Prevention_Fact_Sheet_508.pdf. Published June 2018. Accessed October 14, 2020.
49. Zero Suicide in Health and Behavioral Health Care Institute. Treat: direct treatment. http://zerosuicide.edc.org/toolkit/treat. Accessed October 14, 2020.
50. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.04: Mental health evaluations of members of the military services. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649004p.pdf. Published March 4, 2013. Updated April 22, 2020. Accessed October 14, 2020.
51. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention Frequently asked questions: VA National Suicide Data Report. https://www.mentalhealth.va.gov/docs/data-sheets/2015_Data_Release_FAQs_508.pdf. Published September 2018. Updated June 22, 2018. Accessed October 14, 2020.
52. Zero Suicide in Health and Behavioral Health Care Institute. ZERO Suicide engage: active engagement for safer suicide care safety planning. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Accessed October 14, 2020.
1. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published September 2018. Accessed October 14, 2020.
2. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. National veteran suicide prevention annual report. https://www.mentalhealth.va.gov/suicide_prevention/data.asp. Accessed October 14, 2020.
3. Centers for Disease Control and Prevention. Web-based injury statistics query and reporting system (WISQARS). 2014. https://www.cdc.gov/injury/wisqars/index.html. Updated July 1, 2020. Accessed October 14, 2020.
4. Lemle RB. Choice program expansion jeopardizes high-quality VHA mental health services. Fed Pract. 2018;35(3):18-24.
5. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention. VA national suicide data report 2005-2016. https://www.mentalhealth.va.gov/docs/data-sheets/OMHSP_National_Suicide_Data_Report_2005-2016_508.pdf. Published September 2018. Accessed October 14, 2020.
6. US Department of Veterans Affairs, Office of Suicide Prevention. VA national suicide data report 2005-2015. https://www.mentalhealth.va.gov/docs/data-sheets/2015/OMHSP_National_Suicide_Data_Report_2005-2015_06-14-18_508.pdf. Published June 2018. Accessed October 14, 2020.
7. US Department of Veterans Affairs. National strategy for preventing veteran suicide 2018-2028. https://www.mentalhealth.va.gov/suicide_prevention/docs/Office-of-Mental-Health-and-Suicide-Prevention-National-Strategy-for-Preventing-Veterans-Suicide.pdf. Published 2018. Accessed October 30, 2020.
8. Gordon RS. An operational classification of disease prevention. Public Health Rep. 1983;98(2):107-109.
9. National Research Council and Institute of Medicine. O’Connell ME, Boat T, Warner KE, eds. Preventing Mental, Emotional, and Behavioral Disorders Among Young People: Progress and Possibilities. Washington, DC: National Academies Press; 2009.
10. US Department of Health and Human Services, National Institute on Drug Abuse, Office of Science Policy and Communications. Drug Abuse Prevention: What Works. Rockville, MD: National Institutes of Health; 1997.
11. US Department of Veterans Affairs, US Department of Defense. VA/DoD clinical practice guideline for the assessment and management of patients at risk for suicide. https://www.healthquality.va.gov/guidelines/MH/srb. Published 2019. Updated July 30, 2020. Accessed October 14, 2020.
12. US Department of Veterans Affairs. Veterans Access, Choice and Accountability Act of 2014. https://www.govinfo.gov/content/pkg/PLAW-113publ146/pdf/PLAW-113publ146.pdf. Accessed October 14, 2020.
13. US Department of Veterans Affairs. Strategic Health Care. VA MISSION Act: Extension of the VA choice program. https://strategichealthcare.net/wp-content/uploads/2018/05/052718-VA-MISSION-Act-Summary.pdf. Accessed October 14, 2020.
14. US Congress. H.R.5674—VA MISSION Act of 2018. https://www.congress.gov/bill/115th-congress/house-bill/5674. Accessed October 15, 2020.
15. Grumet JG, Hogan MF, Chu A, Covington DW, Johnson KE. Compliance standards pave the way for reducing suicide in health care systems. J Health Care Compliance. 2019;17-26.
16. Hillestad R, Bigelow J, Bower A, et al. Can electronic medical record systems transform health care? Potential health benefits, savings, and costs. Health Aff (Millwood). 2005;24(5):1103-1117. doi:10.1377/hlthaff.24.5.110
17. Porter ME, Pabo EA, Lee TH. Redesigning primary care: a strategic vision to improve value by organizing around patients’ needs. Health Aff (Millwood). 2013;32(3):516-525. doi:10.1377/hlthaff.2012.0961
18. Nevedal AL, Wagner TH, Ellerbe LS, Asch SM, Koenig CJ. A qualitative study of primary care providers’ experiences with the Veterans Choice Program. J Gen Intern Med. 2019;34:598-603. doi:10.1007/s11606-018-4810-2
19. Tanielian T, Farris C, Epley C, et al. Ready to serve: community-based provider capacity to deliver culturally competent, quality mental health care to veterans and their families. https://www.rand.org/content/dam/rand/pubs/research_reports/RR800/RR806/RAND_RR806.pdf. Published 2014. Accessed October 15, 2020.
20. White House. Executive Order 13861: President’s roadmap to empower veterans and end the national tragedy of suicide (PREVENTS). https://www.va.gov/PREVENTS/docs/PRE-007-The-PREVENTS-Roadmap-1-2_508.pdf. Published June 17, 2020. Accessed October 15, 2020.
21. US Department of Veterans Affairs, Office of the Under Secretary for Health. Memorandum. VA continues community suicide prevention challenge at another mayor’s challenge policy academy. https://www.blogs.va.gov/VAntage/58468/va-continues-community-suicide-prevention-challenge-another-mayors-challenge-policy-academy. Published April 4, 2019. Accessed October 15, 2020.
22. US Department of Veterans Affairs, Veterans Health Administration (VHA) Veterans Health Administration (VHA) Fiscal Year (FY) 2018-2019 Operational Plan. www.navao.org/wp-content/uploads/2018/06/USH-Memo-10-2018-03-VHA_FY_2018-2019_Operational_Plan-4-19-18.pdf. Published April 19, 2018. Accessed October 15, 2020.
23. US Department of Veterans Affairs. VA, Health and Human Services announce governor’s challenge to prevent suicide. https://www.blogs.va.gov/VAntage/55707/va-health-human-services-announce-governors-challenge-prevent-suicide. Published January 17, 2019. Accessed October 15, 2020.
24. VA’s suicide prevention campaign “Be There’’ kicks off in Times Square. https://www.blogs.va.gov/VAntage/57272/vas-suicide-prevention-campaign-kicks-off-times-square. Published March 6, 2019. Accessed October 15, 2020.
25. US Department of Veterans Affairs, Office of Public Affairs Media Relations. Emergent mental health care for former service members. https://www.mentalhealth.va.gov/docs/Fact_Sheet-Emergent_Mental_Health_Care_Former_Service_Members.pdf. Published 2017. Accessed October 15, 2020.
26. US Department of Veterans Affairs. VA secretary formalizes expansion of emergency mental health care to former service members with other-than-honorable discharges. https://www.blogs.va.gov/VAntage/39092/va-secretary-formalizes-expansion-emergency-mental-health-care-former-service-members-honorable-discharges. Published June 27, 2017. Accessed October 15, 2020.
27. Shane L. New veteran suicide numbers raise concerns among experts hoping for positive news. Military Times. https://www.militarytimes.com/news/pentagon-congress/2019/10/09/new-veteran-suicide-numbers-raise-concerns-among-experts-hoping-for-positive-news. Published October 9, 2019. Accessed October 15, 2020.
28. US Department of Veterans Affairs. Department of Veterans Affairs FY 2018-2024 Strategic Plan. https://www.va.gov/oei/docs/VA2018-2024strategicPlan.pdf. Updated May 31, 2019. Accessed October 15, 2020.
29. Komaie G, Goodman M, McCall A, et al. Training community members in public health research: development and implementation of a community participatory research pilot project. Health Equity. 2018;2(1):282-287. doi:10.1089/heq.2018.0043
30. Wang KH, Ray NJ, Berg DN, et al. Using community-based participatory research and organizational diagnosis to characterize relationships between community leaders and academic researchers. Prev Med Reports. 2017;7:180-186. doi:10.1016/j.pmedr.2017.06.007
31. Institute for Healthcare Improvement. The breakthrough series: IHI’s collaborative model for achieving breakthrough improvement. http://www.ihi.org/resources/Pages/IHIWhitePapers/TheBreakthroughSeriesIHIsCollaborativeModelforAchievingBreakthroughImprovement.aspx. Published 2003. Accessed October 15, 2020.
32. Zero Suicide in Health and Behavioral Health Care Institute. Zero suicide toolkit. https://zerosuicide.sprc.org/toolkit. Accessed October 15, 2020.

33. Preventing suicide: a technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicideTechnicalPackage.pdf. Published 2017. Accessed October 30, 2020.
34. US Department of Veterans Affairs. VA Office of Mental Health and Suicide Prevention guidebook. https://www.mentalhealth.va.gov/docs/VA-Office-of-Mental-Health-and-Suicide-Prevention-Guidebook-June-2018-FINAL-508.pdf. Published June 2018. Accessed October 15, 2020.
35. US Department of Health and Human Services, Substance Abuse and Mental Health Services, Administration Center for Substance Abuse Prevention. Focus on prevention: strategies and programs to prevent substance abuse. HHS Publication No. (SMA) 10-4120. https://helpandhopewv.org/docs/Focus%20on%20Prevention.pdf. Revised 2017. Accessed October 15, 2020.
36. Misiti R, Risinger A. RACETIME: an organizational care coordination approach to improving patient outcomes in a complex healthcare setting. Presentation at the Military Social Work & Behavioral Health Conference Austin, Texas. July 23, 2020. https://sites.utexas.edu/military-social-work-conference/misiti-risinger. Accessed October 23, 2020.
37. Rudd MD, Bryan CJ, Wertenberger EG, et al. Brief cognitive-behavioral therapy effects on post-treatment suicide attempts in a military sample: results of a randomized clinical trial with 2-year follow-up. Am J Psychiatry. 2015;172(5):441-449. doi:10.1176/appi.ajp.2014.14070843
38. Rudd MD, Mandrusiak M, Joiner TE Jr. The case against no-suicide contracts: The commitment to treatment statement as a practice alternative. Clin Psychol. 2006;62(2):243-251.
39. Bryan CJ, Mintz J, Clemans TA, et al. Effect of crisis response planning vs. contracts for safety on suicide risk in US Army soldiers: a randomized clinical trial. J Affect Disord. 2017;212:64-72. doi:10.1016/j.jad.2017.01.028
40. Joiner TE. Why People Die by Suicide. Cambridge, MA: First Harvard University Press; 2005:27.
41. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.16: Defense suicide prevention program. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649016p.pdf?ver=2020-09-11-122632-850. Published November 6, 2017. Updated September 11, 2020. Accessed October 14, 2020.
42. Zero Suicide in Health and Behavioral Health Care Institute. Engage: engaging patients. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=0. Accessed October 14, 2020.
43. Stone D, Holland K, Bartholow B, Crosby A, Davis S, Wilkins N. Preventing Suicide: A technical package of policy, programs, and practices. https://www.cdc.gov/violenceprevention/pdf/suicidetechnicalpackage.pdf. Published 2017. Accessed October 14, 2020.
44. Mills PD, Watts BV, Miller S, et al. A checklist to identify inpatient suicide hazards in veterans affairs hospitals. Jt Comm J Qual Patient Saf. 2010;36(2):87-93. doi:10.1016/s1553-7250(10)36015-6
45. US Department of Veterans Affairs, Office of Research and Development. Crisis prevention: study evaluates VA program that identifies vets at highest risk for suicide. https://www.research.va.gov/currents/0918-Study-evaluates-VA-program-that-identifies-Vets-at-highest-risk-for-suicide.cfm. Updated September, 20, 2018. Accessed October 14, 2020.
46. Zero Suicide in Health and Behavioral Health Care Institute. Engage: active engagement for safer suicide care--reducing access to lethal means. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Published 2017. Accessed October 14, 2020.
47. Zero Suicide in Health and Behavioral Health Care Institute. Train: training for all. http://zerosuicide.sprc.org/toolkit/train. Accessed October 14, 2020.
48. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention (OMHSP). Facts about veteran suicide. https://www.mentalhealth.va.gov/docs/FINAL_VA_OMHSP_Suicide_Prevention_Fact_Sheet_508.pdf. Published June 2018. Accessed October 14, 2020.
49. Zero Suicide in Health and Behavioral Health Care Institute. Treat: direct treatment. http://zerosuicide.edc.org/toolkit/treat. Accessed October 14, 2020.
50. Office of the Under Secretary of Defense for Personnel and Readiness. DoD Instruction 6490.04: Mental health evaluations of members of the military services. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/649004p.pdf. Published March 4, 2013. Updated April 22, 2020. Accessed October 14, 2020.
51. US Department of Veterans Affairs, Office of Mental Health and Suicide Prevention Frequently asked questions: VA National Suicide Data Report. https://www.mentalhealth.va.gov/docs/data-sheets/2015_Data_Release_FAQs_508.pdf. Published September 2018. Updated June 22, 2018. Accessed October 14, 2020.
52. Zero Suicide in Health and Behavioral Health Care Institute. ZERO Suicide engage: active engagement for safer suicide care safety planning. http://zerosuicide.edc.org/toolkit/engage#quicktabs-engage=1. Accessed October 14, 2020.
Evaluation of Metabolic Monitoring and Diabetes Incidence in the First Year for Veterans Newly Started on an Atypical Antipsychotic
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
Atypical antipsychotic use may result in metabolic abnormalities, such as hyperglycemia, dyslipidemia, weight gain, and metabolic syndrome. These adverse effects (AEs) can cause progression to type 2 diabetes mellitus (T2DM) as well as increased risk of cardiovascular disease and cardiac mortality. Individuals diagnosed with T2DM have medical expenses that are about 2.3 times higher than individuals without diabetes.1,2 The risk of experiencing metabolic abnormalities is likely elevated for patients who were antipsychotic-naïve prior to initiation.3
In response to an increased awareness of atypical antipsychotic-related AEs, the American Diabetes Association (ADA) and American Psychiatric Association (APA) released a consensus statement in 2004 with a metabolic monitoring protocol for patients initiating or changing to a new antipsychotic medication.4 Within the first year after initiation, the ADA/APA consensus statements recommends that clinicians acquire a personal and family history, weight, body mass index (BMI), waist circumference, blood pressure (BP), fasting plasma glucose, and fasting lipid profile at the initial patient visit. Patient weight is recommended to be collected at 4 weeks and again 8 weeks later. Twelve weeks after the initial visit, weight, BMI, BP, fasting plasma glucose and a fasting lipid profile are recommended to be collected and assessed for abnormalities. Weight is then recommended to be assessed every 3 months thereafter. Review of personal and family history, waist circumference, BP, and a fasting plasma glucose is recommended to occur annually. Finally, a fasting lipid profile is to be collected every 5 years.
Since the initial consensus statement release, metabolic monitoring of patients prescribed antipsychotic medications has been found to be inadequate within several large health care organizations.5,6 Mittal and colleagues reviewed metabolic monitoring practices occurring in 32 facilities within the Veterans Health Administration (VHA) and found that monitoring practices in the first 90 days after antipsychotic initiation were largely nonadherent to the ADA/APA consensus statement recommendations.6 Medical staff in Veterans Integrated Service Network 21 (VISN 21) currently serve about 268,000 veterans actively receiving care across California, Nevada, and the Pacific Islands.To support veteran care in the fields of mental health and medication safety, the VISN 21 pharmacy benefits manager office created a clinical dashboard that identifies veterans who are currently prescribed an antipsychotic and have not completed at least 1 annual blood glucose test. While this dashboard is a valuable tool for tracking patient care for those who have been prescribed an antipsychotic > 1 year, it does not consider the ADA/APA recommendations for more frequent monitoring in the first year after initiation. A literature review found no citations of a systematic evaluation of adherence to ADA/APA monitoring recommendations or patient progression to T2DM in the first year after antipsychotic initiation for an antipsychotic-naïve veteran population. The goal of this quality improvement project is to assess VHA health care provider and patient adherence to the 2004 consensus statement recommendations within the first year after initiation for previously antipsychotic-naïve patients receiving an atypical antipsychotic and determine rate of progression to T2DM.
Methods
The project was reviewed by the University of Nevada-Reno Institutional Review Board and determined to be a nonresearch quality improvement project. This was a retrospective chart analysis that included patients receiving their first-ever atypical antipsychotic across 8 US Department of Veterans Affairs (VA) medical centers within VISN 21. Clinical patient data, including prescription, vital sign, and laboratory information, were extracted from the VA Corporate Data Warehouse using transact sequential query language.
Veterans were included in the final cohort if they met the following criteria: aged ≥ 18 years at antipsychotic initiation, initiated their first-ever atypical antipsychotic within the VHA between February 2014 and February 2019, continued the antipsychotic for ≥ 1 year, had a medication possession ratio (MPR) > 80%, and had previously established care within VHA as evidenced by having ≥ 1 primary care or outpatient mental health visit in the 6 months prior to initiation. The MPR is defined as the sum of the day’s supply of all dispensed medications in the project time frame divided by the total number of days in the project time frame.
Veterans were excluded if they initiated any other antipsychotic during the first course, had a prior diagnosis of T2DM, had any prior use of antidiabetic medications, or had a hemoglobin A1c (HbA1c) > 6.4 in the year prior to initiation.
The primary outcome was completion of all recommended metabolic monitoring time points in the first year after atypical antipsychotic initiation. The secondary outcome was incidence of T2DM as evidenced by either a HbA1c > 6.4 or diagnosis of T2DM entered into the electronic health record. Baseline monitoring for BP, blood glucose, and lipids were considered complete if a data point was collected between 3 months prior and 1 month after atypical antipsychotic initiation. Baseline monitoring for weight was considered complete if a data point was collected between 3 months prior and 2 weeks after initiation. Follow-up monitoring for BP, blood glucose, and lipids were considered completed if a data point was collected at 3 and 12 months (mean, 1 month). Follow-up monitoring for weight was considered completed if collected at 1, 2, and 3 months (mean, 2 weeks) and at 6, 9, and 12 months (mean, 1 month). Waist circumference data and patient and family history are not collected as capturable data points. Therefore, the authors were unable to include these in the final data extraction.
Results
The final cohort consisted of 1,651 veterans who met the inclusion criteria. Overall, at antipsychotic initiation the cohort had a mean (SD) age of 55 (14.6) years, was largely male (88%), and was considered overweight with a mean (SD) BMI of 29.1 (6.4) (Table 1).
Appropriate BP monitoring was completed most often with 492 patients (30%) meeting ADA/APA recommendations followed by HbA1c and/or blood glucose monitoring with 203 patients (12%) completing all time points. Recommended lipid monitoring was completed by 96 patients (6%). Weight monitoring was completed least often with 47 patients (3%) completing all recommended time points. Regarding completion of all metabolic monitoring time points, 3 (0.2%) patients in the final cohort were found to have completed all recommended monitoring. Ninety-nine patients (6%) were found to have progressed to T2DM as indicated by an HbA1c > 6.4 and/or entry of a T2DM ninth or tenth edition International Statistical Classification of Diseases code into the chart (Table 2).
Discussion
No previous literature exists that reviews adherence to recommended metabolic monitoring guidance up to 1 year after antipsychotic initiation in a previously antipsychotic-naïve cohort within the VHA. Metabolic monitoring was overall incomplete with 0.2% of the cohort completing all recommended monitoring time points. Weight was the parameter that was least completed. Based on these findings, the authors concluded that efforts are needed to improve completion rates of atypical antipsychotic metabolic monitoring. In the final cohort, 6% of patients were noted to have progressed to T2DM in the first year after atypical antipsychotic initiation. The actual number of patients progressing to T2DM may be larger because not all received adequate blood glucose monitoring. For comparison, the Centers for Disease Control and Prevention released information in 2015 that stated that the US population has an annual T2DM incidence of about 1% for adults aged 45 to 64 years.7
We understand that individuals with mental health disorders are at increased risk of T2DM compared with that of the general population and hope that this comparison only serves to drive home the point that appropriate metabolic monitoring is vital for this subgroup. The strengths of this project include identification of an area for improvement and encouraging evidence-based monitoring. Utilization of clinical data is a cost-effective and efficient method to improve patient care.
Limitations
Limitations of this study include the data’s dependence on accuracy of entry by the end-user and a lack of available data regarding prescriptions dispensed outside of the VHA. Vital signs data may have been entered into patient notes and not documented in the vitals section of the current medical record causing the appearance of missing data. Access to VHA health services and patient adherence to follow-up appointments were not assessed in this project and could affect patient ability to complete follow-up. The final analysis included only patients who remained on 1 atypical antipsychotic for a year and were considered adherent with an MPR > 80% and did not consider less adherent patients. It is also possible that health care providers who closely monitor metabolic parameters after atypical antipsychotic initiation more frequently switch patients to an alternative atypical antipsychotic while others who monitor less also switch medications less frequently. This could lead to selection of patients with health care providers who are less adherent to metabolic monitoring recommendations.
Conclusions
As a result of this study, in VISN 21 several strategies will be implemented to improve monitoring. First, the results of this project will be shared with the subject matter experts of the VISN 21 Mental Health Task Force. This task force serves as a venue for clinicians to meet virtually, discuss clinical topics, as well as to create and distribute strategies to improve patient care. Clinicians at this forum will be encouraged to implement monitoring protocols into routine practice, share best practices with colleagues, and increase patient awareness about the importance of metabolic monitoring. Second, modifications may be applied to the electronic health record to guide metabolic monitoring order entry at the time of prescription entry, which includes development of clinical reminders and laboratory order sets. Third, the clinical data manager team may be leveraged to create an electronic report identifying patients currently receiving suboptimal monitoring in the first year after antipsychotic initiation. The patients identified in this report will be discussed at the recurring VISN 21 Mental Health Task Force meeting, and strong practices will be shared with the medical centers across VISN 21. Other strategies under consideration include requiring proof of metabolic monitoring completion prior to allowing further atypical antipsychotic refills and providing direct provider education regarding the ADA/APA metabolic monitoring recommendations via the academic detailing service in effort to standardize clinical care.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
1. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr Res. 2005;80(1):45-53. doi:10.1016/j.schres.2005.08.010
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2012. Diabetes Care. 2013;36(4):1033-1046. doi:10.2337/dc12-2625
3. Rajkumar AP, Horsdal HT, Wimberley T, et al. Endogenous and antipsychotic-related risks for diabetes mellitus in young people with schizophrenia: a Danish population-based cohort study. Am J Psychiatry. 2017;174(7):686-694. doi:10.1176/appi.ajp.2016.16040442
4. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27(2):596-601. doi:10.2337/diacare.27.2.596
5. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67(1):17-24. doi:10.1001/archgenpsychiatry.2009.179
6. Mittal D, Li C, Williams JS, Viverito K, Landes RD, Owen RR. Monitoring veterans for metabolic side effects when prescribing antipsychotics. Psychiatr Serv. 2013;64(1):28-35. doi:10.1176/appi.ps.201100445
7. Centers for Disease Control and Prevention. National diabetes statistics report, 2020. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Updated August 28, 2020. Accessed October 9, 2020.
The Gips Procedure for Pilonidal Disease: A Retrospective Review of Adolescent Patients
Pilonidal disease (PD) is common in Turkey. In a study in Turkey, 19,013 young patients aged 17 to 28 years were examined; PD was detected in 6.6% of patients (0.37% of females in the cohort and 6.23% of males).1 The incidence of PD in military personnel (women 18 years and older; men 22 years and older) is remarkably higher, with an incidence of 9% reported in Turkish soldiers.2
Pilonidal disease has become common in Turkish adolescents, who now experience an increase in desk time because of computer use and a long duration of preparation for high school and university entrance examinations. In adolescent and adult population studies, Yildiz et al3 and Harlak et al4 reported that sitting for 6 hours or more per day was found to significantly increase the risk for PD compared to the control group (P=.028 and P<.001, respectively).
Surgery for PD often is followed by a considerable and unpleasant postoperative course, with a long period of limited physical activity, loss of school time, and reduced social relationships. The recurrence rate of PD is reported to be as high as 40% to 50% after incision and drainage, 40% to 55% with rigorous hygiene and weekly shaving, and as high as 30% following operative intervention. Drawbacks of operative intervention include associated morbidity; lost work and school time; and prolonged wound healing, which can take days to months.5-7
For these reasons, minimally invasive surgical techniques have become popular for treating PD in adolescents, as surgery can cause less disruption of the school and examination schedule and provide an earlier return to normal activities. Gips et al8—who operated on 1358 adults using skin trephines to extirpate pilonidal pits and the underlying fistulous tract and hair debris—reported a low recurrence rate and good postoperative functional outcomes with this technique. Herein, we present our short-duration experience with the Gips procedure of minimally invasive sinusectomy in adolescent PD.
Methods
Patients
We performed a retrospective medical record review of patients with symptomatic PD who were treated in our clinic between January 2018 and February 2019 using the Gips procedure of minimally invasive sinusectomy. We identified 19 patients younger than 17 years. Patients with acute inflammation and an acute undrained collection of pits were treated with incision and drainage, with close clinical follow-up until inflammation resolved. We also recommended that patients take a warm sitz bath at least once daily and chemically epilate the hair in the affected area if they were hirsute.
Gips Procedure
For all patients, the Gips procedure was performed in the left lateral position under general anesthesia using a laryngeal mask airway for anesthesia. Patients were closely shaved (if hirsute) then prepared with povidone-iodine solution. First, each fistulous opening was probed to assess depth and direction of underlying tracts using a thin (0.5–1.0 mm), round-tipped probe. Next, a trephine—comprising a cylindrical blade on a handle—was used to remove cylindrical cores of tissue. All visible median pits and lateral fistulous skin openings were excised using skin trephines of various diameters (Figure, A and B). Once the pilonidal cavity was reached, attention was directed to removing all residual underlying tissue—granulation tissue, debris, and hair—through all available accesses. The cavity was cleaned with hydrogen peroxide and normal saline. Then, all trephine-made openings were left unpacked or were packed for only a few hours and were not sutured (Figure, C and D); a light gauze bandage was eventually applied with a minimum of tape and skin traction. Patients were kept supine during a 1- or 2-hour clinical observation period before they were discharged.
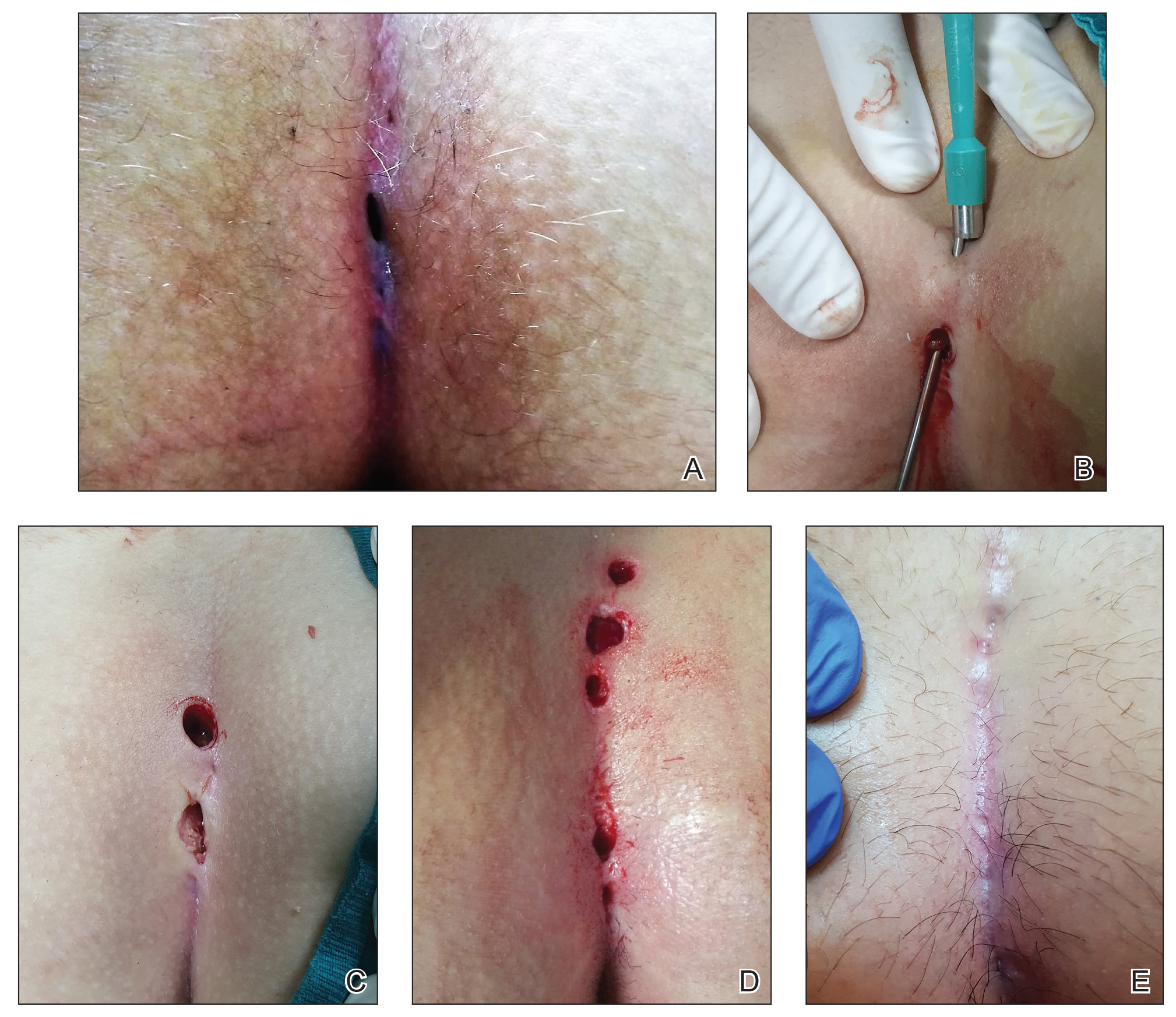
Postoperatively, no regular medications other than analgesics were recommended; routine daily activities were allowed. Patients were encouraged to sleep supine and wash the sacrococcygeal region with running water several times a day after the second postoperative day. Frequent showering, application of povidone-iodine to the wound after defecation, and regular epilation of the sacrococcygeal area also were recommended to all patients.
All patients were routinely followed by the same surgical group weekly until wound healing was complete (Figure, E).
Medical Record Review
Patients’ electronic medical records were reviewed retrospectively, and parameters including age at surgery, surgical history, symptoms, duration of operation and hospital stay, time to return to activity, wound healing time, and recurrence were recorded.
Results
Of the 19 patients who underwent the Gips procedure, 17 (90%) were male; 2 (10%) were female. The mean (standard deviation [SD]) body mass index was 25 (3.7). (Body mass index was calculated as weight in kilograms divided by height in meters squared.) The mean age (SD) of patients was 15 (1.1) years (range, 12–17 years). The most common symptom at presentation was purulent discharge (11/19 [58%]). Other common symptoms included pain (8/19 [42%]), pilonidal abscess (6/19 [32%]), and bleeding (4/19 [21%]). Nine patients (47%) had prior abscess drainage at presentation; 1 (5%) had previously undergone surgery, and 5 (26%) previously had phenol injections.
The median (SD) length of stay in the hospital was 15 (3.2) hours (range, 11–22 hours). The mean (SD) time before returning to daily activities and school was 2 (0.6) days (range, 1–3 days). In our patients, the Gips procedure was performed on either a Thursday or more often a Friday; therefore, patients could be scheduled to be discharged from the hospital and return to home the next day, and then return to school on Monday. All patients were advised to take an oral analgesic for 2 days following the procedure.
The mean (SD) duration of the operative procedure was 14 (3) minutes (range, 10–20 minutes). One patient (5%) developed bleeding that ceased spontaneously. The mean (SD) complete wound healing time was 3 (0.6) weeks (range, 2–4 weeks).
Postoperative clinical examination and telephone interviews were performed for follow-up. The mean follow-up period was 5 months (range, 1–13 months); 17 of 19 patients (89%) made a complete recovery. Two patients (11%) reported recurrence in the third and fourth months following the procedure and were treated with a repeat Gips procedure 6 months after the first treatment. Improvement was noted after a second Gips procedure in 1 of 2 patients who had recurrence, leaving the success rate of the procedure in our practice at 95% (18/19).
Comment
Various treatment methods for PD have been postulated,5-7 including incision and drainage, hair removal and hygiene alone, excision and primary wound closure, excision and secondary wound closure, and various flap techniques. More recently, there has been a dramatic shift to management of patients with PD in an outpatient setting. The Gips procedure, an innovative minimally surgical technique for PD, was introduced in 2008 based on a large consecutive series of more than 1300 patients.8 Studies have shown promising results and minimal recovery time for the Gips procedure in adult and pediatric patients.8-10
Nevertheless, conventional excision down to the sacral fascia, with or without midline or asymmetrical closure, is still the procedure performed most often for PD worldwide.
Advantages of the Gips Procedure
Advantages of the Gips procedure are numerous. It is easily applicable, inexpensive, well tolerated, and requires minimal postoperative care. Placing the patient in the lateral position for the procedure—rather than the prone position that is required for more extensive surgical procedures—is highly feasible, permitting the easy application of a laryngeal mask for anesthesia. The Gips procedure can be performed on patients with severe PD after a period of improved hygiene and hair control and allows for less morbidity than older surgical techniques. Overall, results are satisfactory.
Health services and the hospital admissions process are less costly in university hospitals in Turkey. This procedure costs an average of 400 Turkish liras (<US $50). For that reason, patients in our review were discharged the next day; however, patients could be discharged within a few hours. In the future, it is possible for appropriate cases to be managed in an outpatient setting with sedation and local anesthesia only. Because their postoperative courses are eventless, these patients can be managed without hospitalization.
Recovery is quick and allows for early return to school and other physical activities. Because the procedure was most often performed on the last school day of the week, we did not see any restriction of physical or social activities in our patients.
Lastly, this procedure can be applied to PD patients who have previously undergone extensive surgery or phenol injection, as was the case in our patients.
Conclusion
The Gips procedure is an easy-to-use technique in children and adolescents with PD. It has a high success rate and places fewer restrictions on school and social activities than traditional surgical therapies.
- Duman K, Gırgın M, Harlak A. Prevalence of sacrococcygeal pilonidal disease in Turkey. Asian J Surg. 2017;40:434-437.
- Akinci OF, Bozer M, Uzunköy A, et al. Incidence and aetiological factors in pilonidal sinus among Turkish soldiers. Eur J Surg. 1999;165:339-342.
- Yildiz T, Elmas B, Yucak A, et al. Risk factors for pilonidal sinus disease in teenagers. Indian J Pediatr. 2017;84:134-138.
- Harlak A, Mentes O, Kilic S, et al. Sacrococcygeal pilonidal disease: analysis of previously proposed risk factors. Clinics (Sao Paulo). 2010;65:125-131.
- Delshad HR, Dawson M, Melvin P, et al. Pit-picking resolves pilonidal disease in adolescents. J Pediatr Surg. 2019;54:174-176.
- Humphries AE, Duncan JE. Evaluation and management of pilonidal disease. Surg Clin North Am. 2010;90:113-124.
- Bascom J. Pilonidal disease: origin from follicles of hairs and results of follicle removal as treatment. Surgery. 1980;87:567-572.
- Gips M, Melki Y, Salem L, et al. Minimal surgery for pilonidal disease using trephines: description of a new technique and long-term outcomes in 1,358 patients. Dis Colon Rectum. 2008;51:1656-1662; discussion, 1662-1663.
- Speter C, Zmora O, Nadler R, et al. Minimal incision as a promising technique for resection of pilonidal sinus in children. J Pediatr Surg. 2017;52:1484-1487.
- Di Castro A, Guerra F, Levi Sandri GB, et al. Minimally invasive surgery for the treatment of pilonidal disease. the Gips procedure on 2347 patients. Int J Surg. 2016;36:201-205.
- Guerra F, Giuliani G, Amore Bonapasta S, et al. Cleft lift versus standard excision with primary midline closure for the treatment of pilonidal disease. a snapshot of worldwide current practice. Eur Surg. 2016;48:269-272.
Pilonidal disease (PD) is common in Turkey. In a study in Turkey, 19,013 young patients aged 17 to 28 years were examined; PD was detected in 6.6% of patients (0.37% of females in the cohort and 6.23% of males).1 The incidence of PD in military personnel (women 18 years and older; men 22 years and older) is remarkably higher, with an incidence of 9% reported in Turkish soldiers.2
Pilonidal disease has become common in Turkish adolescents, who now experience an increase in desk time because of computer use and a long duration of preparation for high school and university entrance examinations. In adolescent and adult population studies, Yildiz et al3 and Harlak et al4 reported that sitting for 6 hours or more per day was found to significantly increase the risk for PD compared to the control group (P=.028 and P<.001, respectively).
Surgery for PD often is followed by a considerable and unpleasant postoperative course, with a long period of limited physical activity, loss of school time, and reduced social relationships. The recurrence rate of PD is reported to be as high as 40% to 50% after incision and drainage, 40% to 55% with rigorous hygiene and weekly shaving, and as high as 30% following operative intervention. Drawbacks of operative intervention include associated morbidity; lost work and school time; and prolonged wound healing, which can take days to months.5-7
For these reasons, minimally invasive surgical techniques have become popular for treating PD in adolescents, as surgery can cause less disruption of the school and examination schedule and provide an earlier return to normal activities. Gips et al8—who operated on 1358 adults using skin trephines to extirpate pilonidal pits and the underlying fistulous tract and hair debris—reported a low recurrence rate and good postoperative functional outcomes with this technique. Herein, we present our short-duration experience with the Gips procedure of minimally invasive sinusectomy in adolescent PD.
Methods
Patients
We performed a retrospective medical record review of patients with symptomatic PD who were treated in our clinic between January 2018 and February 2019 using the Gips procedure of minimally invasive sinusectomy. We identified 19 patients younger than 17 years. Patients with acute inflammation and an acute undrained collection of pits were treated with incision and drainage, with close clinical follow-up until inflammation resolved. We also recommended that patients take a warm sitz bath at least once daily and chemically epilate the hair in the affected area if they were hirsute.
Gips Procedure
For all patients, the Gips procedure was performed in the left lateral position under general anesthesia using a laryngeal mask airway for anesthesia. Patients were closely shaved (if hirsute) then prepared with povidone-iodine solution. First, each fistulous opening was probed to assess depth and direction of underlying tracts using a thin (0.5–1.0 mm), round-tipped probe. Next, a trephine—comprising a cylindrical blade on a handle—was used to remove cylindrical cores of tissue. All visible median pits and lateral fistulous skin openings were excised using skin trephines of various diameters (Figure, A and B). Once the pilonidal cavity was reached, attention was directed to removing all residual underlying tissue—granulation tissue, debris, and hair—through all available accesses. The cavity was cleaned with hydrogen peroxide and normal saline. Then, all trephine-made openings were left unpacked or were packed for only a few hours and were not sutured (Figure, C and D); a light gauze bandage was eventually applied with a minimum of tape and skin traction. Patients were kept supine during a 1- or 2-hour clinical observation period before they were discharged.

Postoperatively, no regular medications other than analgesics were recommended; routine daily activities were allowed. Patients were encouraged to sleep supine and wash the sacrococcygeal region with running water several times a day after the second postoperative day. Frequent showering, application of povidone-iodine to the wound after defecation, and regular epilation of the sacrococcygeal area also were recommended to all patients.
All patients were routinely followed by the same surgical group weekly until wound healing was complete (Figure, E).
Medical Record Review
Patients’ electronic medical records were reviewed retrospectively, and parameters including age at surgery, surgical history, symptoms, duration of operation and hospital stay, time to return to activity, wound healing time, and recurrence were recorded.
Results
Of the 19 patients who underwent the Gips procedure, 17 (90%) were male; 2 (10%) were female. The mean (standard deviation [SD]) body mass index was 25 (3.7). (Body mass index was calculated as weight in kilograms divided by height in meters squared.) The mean age (SD) of patients was 15 (1.1) years (range, 12–17 years). The most common symptom at presentation was purulent discharge (11/19 [58%]). Other common symptoms included pain (8/19 [42%]), pilonidal abscess (6/19 [32%]), and bleeding (4/19 [21%]). Nine patients (47%) had prior abscess drainage at presentation; 1 (5%) had previously undergone surgery, and 5 (26%) previously had phenol injections.
The median (SD) length of stay in the hospital was 15 (3.2) hours (range, 11–22 hours). The mean (SD) time before returning to daily activities and school was 2 (0.6) days (range, 1–3 days). In our patients, the Gips procedure was performed on either a Thursday or more often a Friday; therefore, patients could be scheduled to be discharged from the hospital and return to home the next day, and then return to school on Monday. All patients were advised to take an oral analgesic for 2 days following the procedure.
The mean (SD) duration of the operative procedure was 14 (3) minutes (range, 10–20 minutes). One patient (5%) developed bleeding that ceased spontaneously. The mean (SD) complete wound healing time was 3 (0.6) weeks (range, 2–4 weeks).
Postoperative clinical examination and telephone interviews were performed for follow-up. The mean follow-up period was 5 months (range, 1–13 months); 17 of 19 patients (89%) made a complete recovery. Two patients (11%) reported recurrence in the third and fourth months following the procedure and were treated with a repeat Gips procedure 6 months after the first treatment. Improvement was noted after a second Gips procedure in 1 of 2 patients who had recurrence, leaving the success rate of the procedure in our practice at 95% (18/19).
Comment
Various treatment methods for PD have been postulated,5-7 including incision and drainage, hair removal and hygiene alone, excision and primary wound closure, excision and secondary wound closure, and various flap techniques. More recently, there has been a dramatic shift to management of patients with PD in an outpatient setting. The Gips procedure, an innovative minimally surgical technique for PD, was introduced in 2008 based on a large consecutive series of more than 1300 patients.8 Studies have shown promising results and minimal recovery time for the Gips procedure in adult and pediatric patients.8-10
Nevertheless, conventional excision down to the sacral fascia, with or without midline or asymmetrical closure, is still the procedure performed most often for PD worldwide.
Advantages of the Gips Procedure
Advantages of the Gips procedure are numerous. It is easily applicable, inexpensive, well tolerated, and requires minimal postoperative care. Placing the patient in the lateral position for the procedure—rather than the prone position that is required for more extensive surgical procedures—is highly feasible, permitting the easy application of a laryngeal mask for anesthesia. The Gips procedure can be performed on patients with severe PD after a period of improved hygiene and hair control and allows for less morbidity than older surgical techniques. Overall, results are satisfactory.
Health services and the hospital admissions process are less costly in university hospitals in Turkey. This procedure costs an average of 400 Turkish liras (<US $50). For that reason, patients in our review were discharged the next day; however, patients could be discharged within a few hours. In the future, it is possible for appropriate cases to be managed in an outpatient setting with sedation and local anesthesia only. Because their postoperative courses are eventless, these patients can be managed without hospitalization.
Recovery is quick and allows for early return to school and other physical activities. Because the procedure was most often performed on the last school day of the week, we did not see any restriction of physical or social activities in our patients.
Lastly, this procedure can be applied to PD patients who have previously undergone extensive surgery or phenol injection, as was the case in our patients.
Conclusion
The Gips procedure is an easy-to-use technique in children and adolescents with PD. It has a high success rate and places fewer restrictions on school and social activities than traditional surgical therapies.
Pilonidal disease (PD) is common in Turkey. In a study in Turkey, 19,013 young patients aged 17 to 28 years were examined; PD was detected in 6.6% of patients (0.37% of females in the cohort and 6.23% of males).1 The incidence of PD in military personnel (women 18 years and older; men 22 years and older) is remarkably higher, with an incidence of 9% reported in Turkish soldiers.2
Pilonidal disease has become common in Turkish adolescents, who now experience an increase in desk time because of computer use and a long duration of preparation for high school and university entrance examinations. In adolescent and adult population studies, Yildiz et al3 and Harlak et al4 reported that sitting for 6 hours or more per day was found to significantly increase the risk for PD compared to the control group (P=.028 and P<.001, respectively).
Surgery for PD often is followed by a considerable and unpleasant postoperative course, with a long period of limited physical activity, loss of school time, and reduced social relationships. The recurrence rate of PD is reported to be as high as 40% to 50% after incision and drainage, 40% to 55% with rigorous hygiene and weekly shaving, and as high as 30% following operative intervention. Drawbacks of operative intervention include associated morbidity; lost work and school time; and prolonged wound healing, which can take days to months.5-7
For these reasons, minimally invasive surgical techniques have become popular for treating PD in adolescents, as surgery can cause less disruption of the school and examination schedule and provide an earlier return to normal activities. Gips et al8—who operated on 1358 adults using skin trephines to extirpate pilonidal pits and the underlying fistulous tract and hair debris—reported a low recurrence rate and good postoperative functional outcomes with this technique. Herein, we present our short-duration experience with the Gips procedure of minimally invasive sinusectomy in adolescent PD.
Methods
Patients
We performed a retrospective medical record review of patients with symptomatic PD who were treated in our clinic between January 2018 and February 2019 using the Gips procedure of minimally invasive sinusectomy. We identified 19 patients younger than 17 years. Patients with acute inflammation and an acute undrained collection of pits were treated with incision and drainage, with close clinical follow-up until inflammation resolved. We also recommended that patients take a warm sitz bath at least once daily and chemically epilate the hair in the affected area if they were hirsute.
Gips Procedure
For all patients, the Gips procedure was performed in the left lateral position under general anesthesia using a laryngeal mask airway for anesthesia. Patients were closely shaved (if hirsute) then prepared with povidone-iodine solution. First, each fistulous opening was probed to assess depth and direction of underlying tracts using a thin (0.5–1.0 mm), round-tipped probe. Next, a trephine—comprising a cylindrical blade on a handle—was used to remove cylindrical cores of tissue. All visible median pits and lateral fistulous skin openings were excised using skin trephines of various diameters (Figure, A and B). Once the pilonidal cavity was reached, attention was directed to removing all residual underlying tissue—granulation tissue, debris, and hair—through all available accesses. The cavity was cleaned with hydrogen peroxide and normal saline. Then, all trephine-made openings were left unpacked or were packed for only a few hours and were not sutured (Figure, C and D); a light gauze bandage was eventually applied with a minimum of tape and skin traction. Patients were kept supine during a 1- or 2-hour clinical observation period before they were discharged.

Postoperatively, no regular medications other than analgesics were recommended; routine daily activities were allowed. Patients were encouraged to sleep supine and wash the sacrococcygeal region with running water several times a day after the second postoperative day. Frequent showering, application of povidone-iodine to the wound after defecation, and regular epilation of the sacrococcygeal area also were recommended to all patients.
All patients were routinely followed by the same surgical group weekly until wound healing was complete (Figure, E).
Medical Record Review
Patients’ electronic medical records were reviewed retrospectively, and parameters including age at surgery, surgical history, symptoms, duration of operation and hospital stay, time to return to activity, wound healing time, and recurrence were recorded.
Results
Of the 19 patients who underwent the Gips procedure, 17 (90%) were male; 2 (10%) were female. The mean (standard deviation [SD]) body mass index was 25 (3.7). (Body mass index was calculated as weight in kilograms divided by height in meters squared.) The mean age (SD) of patients was 15 (1.1) years (range, 12–17 years). The most common symptom at presentation was purulent discharge (11/19 [58%]). Other common symptoms included pain (8/19 [42%]), pilonidal abscess (6/19 [32%]), and bleeding (4/19 [21%]). Nine patients (47%) had prior abscess drainage at presentation; 1 (5%) had previously undergone surgery, and 5 (26%) previously had phenol injections.
The median (SD) length of stay in the hospital was 15 (3.2) hours (range, 11–22 hours). The mean (SD) time before returning to daily activities and school was 2 (0.6) days (range, 1–3 days). In our patients, the Gips procedure was performed on either a Thursday or more often a Friday; therefore, patients could be scheduled to be discharged from the hospital and return to home the next day, and then return to school on Monday. All patients were advised to take an oral analgesic for 2 days following the procedure.
The mean (SD) duration of the operative procedure was 14 (3) minutes (range, 10–20 minutes). One patient (5%) developed bleeding that ceased spontaneously. The mean (SD) complete wound healing time was 3 (0.6) weeks (range, 2–4 weeks).
Postoperative clinical examination and telephone interviews were performed for follow-up. The mean follow-up period was 5 months (range, 1–13 months); 17 of 19 patients (89%) made a complete recovery. Two patients (11%) reported recurrence in the third and fourth months following the procedure and were treated with a repeat Gips procedure 6 months after the first treatment. Improvement was noted after a second Gips procedure in 1 of 2 patients who had recurrence, leaving the success rate of the procedure in our practice at 95% (18/19).
Comment
Various treatment methods for PD have been postulated,5-7 including incision and drainage, hair removal and hygiene alone, excision and primary wound closure, excision and secondary wound closure, and various flap techniques. More recently, there has been a dramatic shift to management of patients with PD in an outpatient setting. The Gips procedure, an innovative minimally surgical technique for PD, was introduced in 2008 based on a large consecutive series of more than 1300 patients.8 Studies have shown promising results and minimal recovery time for the Gips procedure in adult and pediatric patients.8-10
Nevertheless, conventional excision down to the sacral fascia, with or without midline or asymmetrical closure, is still the procedure performed most often for PD worldwide.
Advantages of the Gips Procedure
Advantages of the Gips procedure are numerous. It is easily applicable, inexpensive, well tolerated, and requires minimal postoperative care. Placing the patient in the lateral position for the procedure—rather than the prone position that is required for more extensive surgical procedures—is highly feasible, permitting the easy application of a laryngeal mask for anesthesia. The Gips procedure can be performed on patients with severe PD after a period of improved hygiene and hair control and allows for less morbidity than older surgical techniques. Overall, results are satisfactory.
Health services and the hospital admissions process are less costly in university hospitals in Turkey. This procedure costs an average of 400 Turkish liras (<US $50). For that reason, patients in our review were discharged the next day; however, patients could be discharged within a few hours. In the future, it is possible for appropriate cases to be managed in an outpatient setting with sedation and local anesthesia only. Because their postoperative courses are eventless, these patients can be managed without hospitalization.
Recovery is quick and allows for early return to school and other physical activities. Because the procedure was most often performed on the last school day of the week, we did not see any restriction of physical or social activities in our patients.
Lastly, this procedure can be applied to PD patients who have previously undergone extensive surgery or phenol injection, as was the case in our patients.
Conclusion
The Gips procedure is an easy-to-use technique in children and adolescents with PD. It has a high success rate and places fewer restrictions on school and social activities than traditional surgical therapies.
- Duman K, Gırgın M, Harlak A. Prevalence of sacrococcygeal pilonidal disease in Turkey. Asian J Surg. 2017;40:434-437.
- Akinci OF, Bozer M, Uzunköy A, et al. Incidence and aetiological factors in pilonidal sinus among Turkish soldiers. Eur J Surg. 1999;165:339-342.
- Yildiz T, Elmas B, Yucak A, et al. Risk factors for pilonidal sinus disease in teenagers. Indian J Pediatr. 2017;84:134-138.
- Harlak A, Mentes O, Kilic S, et al. Sacrococcygeal pilonidal disease: analysis of previously proposed risk factors. Clinics (Sao Paulo). 2010;65:125-131.
- Delshad HR, Dawson M, Melvin P, et al. Pit-picking resolves pilonidal disease in adolescents. J Pediatr Surg. 2019;54:174-176.
- Humphries AE, Duncan JE. Evaluation and management of pilonidal disease. Surg Clin North Am. 2010;90:113-124.
- Bascom J. Pilonidal disease: origin from follicles of hairs and results of follicle removal as treatment. Surgery. 1980;87:567-572.
- Gips M, Melki Y, Salem L, et al. Minimal surgery for pilonidal disease using trephines: description of a new technique and long-term outcomes in 1,358 patients. Dis Colon Rectum. 2008;51:1656-1662; discussion, 1662-1663.
- Speter C, Zmora O, Nadler R, et al. Minimal incision as a promising technique for resection of pilonidal sinus in children. J Pediatr Surg. 2017;52:1484-1487.
- Di Castro A, Guerra F, Levi Sandri GB, et al. Minimally invasive surgery for the treatment of pilonidal disease. the Gips procedure on 2347 patients. Int J Surg. 2016;36:201-205.
- Guerra F, Giuliani G, Amore Bonapasta S, et al. Cleft lift versus standard excision with primary midline closure for the treatment of pilonidal disease. a snapshot of worldwide current practice. Eur Surg. 2016;48:269-272.
- Duman K, Gırgın M, Harlak A. Prevalence of sacrococcygeal pilonidal disease in Turkey. Asian J Surg. 2017;40:434-437.
- Akinci OF, Bozer M, Uzunköy A, et al. Incidence and aetiological factors in pilonidal sinus among Turkish soldiers. Eur J Surg. 1999;165:339-342.
- Yildiz T, Elmas B, Yucak A, et al. Risk factors for pilonidal sinus disease in teenagers. Indian J Pediatr. 2017;84:134-138.
- Harlak A, Mentes O, Kilic S, et al. Sacrococcygeal pilonidal disease: analysis of previously proposed risk factors. Clinics (Sao Paulo). 2010;65:125-131.
- Delshad HR, Dawson M, Melvin P, et al. Pit-picking resolves pilonidal disease in adolescents. J Pediatr Surg. 2019;54:174-176.
- Humphries AE, Duncan JE. Evaluation and management of pilonidal disease. Surg Clin North Am. 2010;90:113-124.
- Bascom J. Pilonidal disease: origin from follicles of hairs and results of follicle removal as treatment. Surgery. 1980;87:567-572.
- Gips M, Melki Y, Salem L, et al. Minimal surgery for pilonidal disease using trephines: description of a new technique and long-term outcomes in 1,358 patients. Dis Colon Rectum. 2008;51:1656-1662; discussion, 1662-1663.
- Speter C, Zmora O, Nadler R, et al. Minimal incision as a promising technique for resection of pilonidal sinus in children. J Pediatr Surg. 2017;52:1484-1487.
- Di Castro A, Guerra F, Levi Sandri GB, et al. Minimally invasive surgery for the treatment of pilonidal disease. the Gips procedure on 2347 patients. Int J Surg. 2016;36:201-205.
- Guerra F, Giuliani G, Amore Bonapasta S, et al. Cleft lift versus standard excision with primary midline closure for the treatment of pilonidal disease. a snapshot of worldwide current practice. Eur Surg. 2016;48:269-272.
Practice Points
- The Gips procedure is an easy-to-use outpatient procedure for adolescents with pilonidal disease.
- This procedure has a high success rate and does not restrict school or social activities.
Nivolumab Use for First-Line Management of Hepatocellular Carcinoma: Results of a Real-World Cohort of Patients
Hepatocellular carcinoma (HCC) has a poor prognosis and remains an important cause of cancer-related morbidity and mortality.1,2 Potentially curative interventions include surgical resection, radiofrequency ablation, and liver transplantation. However, the majority of patients are not eligible for these procedures because they are diagnosed at an advanced stage, when locoregional therapies are much more limited.3,4 Although the kinase inhibitors sorafenib and lenvatinib are approved as first-line systemic treatment, at the US Department of Veterans Affairs (VA) Kansas City VA Medical Center (KCVAMC) in Missouri, nivolumab was used instead because of concerns for the tolerability of the kinase inhibitors. Locoregional therapies, resection, and transplantation options were either not appropriate or had been exhausted for these patients. The objective of this retrospective study was to determine the outcomes of those veteran patients in a small cohort.
Methods
The KCVAMC Institutional Review Board approved this retrospective chart review. Patients were selected from pharmacy records at KCVAMC. We identified all patients with a diagnosis of HCC who received nivolumab from January 2016 to December 2019. We then included only the patients that had nivolumab in the front-line setting for our final analysis. At the time of initiation of treatment, all patients were informed that immunotherapy was not approved for front-line treatment, but available evidence suggested that it would be easier to tolerate than sorafenib or lenvatinib. These patients were determined to be either ineligible for sorafenib or lenvatinib therapy or expected to tolerate it poorly, and hence they consented to the use of nivolumab. Tumor response and progression were assessed by the investigator according to iRECIST (Immune Response Evaluation Criteria in Solid Tumors) criteria.5 Data were obtained from retrospective health record review.
Results
Fourteen men received nivolumab in the front-line systemic therapy setting from January 2016 to December 2019 at KCVAMC. The median age was 63.5 years (range, 58-72 years), and the median Eastern Cooperative Oncology Group score was 1. The Table highlights patient characteristics.
Of the 14 patients included in the review, 2 patients had a response to nivolumab (14.3%) and 1 patient had a complete response (7.1%). The median duration of immunotherapy was 4.5 months. Immunotherapy was discontinued due to disease progression in 10 patients and toxicity in 3 patients.
The median progression-free survival (PFS) from initiation of immunotherapy was 4 months; median overall survival (OS) was 8 months. The median time from diagnosis to survival was 41 months. Only 1 patient received a second-line treatment.
Incidence of grade 3 or higher toxicity was 35%. Three deaths resulted from auto-immune hepatitis (grade 5 toxicity), as well as 1 grade 3 skin toxicity, and 1 grade 4 liver toxicity.
Discussion
Immunotherapy has shown promise in patients with HCC based on the results of the KEYNOTE-224 and Checkmate-040 studies,6,7 which led to an accelerated US Food and Drug Administration approval of nivolumab and pembrolizumab for HCC following failure of first-line sorafenib.8,9
Several clinical trials are evaluating front-line immunotherapy for HCC. The Checkmate 459 study demonstrated the median OS to be 16.4 months for nivolumab vs 14.7 months for sorafenib, a difference that was not statistically significant. However, tolerability of nivolumab was better than it was for sorafenib, thus positioning it as a potentially attractive first-line option.10 The GO30140 study evaluated
The results from our study differed from the previous studies and raise concern for the applicability of these trials to a real-world population. For example, both the GO30140 and IMbrave150 excluded patients with untreated varices.11,12 Both IMbrave150 and Checkmate 459 limited enrollment only to patients with a Child-Pugh A score for liver disease; 36% of the KCVAMC patients had a Child-Pugh B score. Three patients (21.4%) were homeless, 6 patients (42.8%) had substance abuse history and 5 patients (35.7%) had mental illness. Several psychosocial factors present in our patients, such as substance abuse, mental illness, and homelessness, would have excluded them from clinical trials. Our small cohort of patients, thus, represents a frail real-world population due to multiple medical and psychosocial comorbidities. Real-world experience with immunotherapy as second-line therapy after treatment with sorafenib has been reported, but this is the first reported real-world experience of immunotherapy in the front-line setting for HCC.13,14
Large differences in sociodemographic status and health status exist between the veteran population and typical clinical trial populations. Veterans are predominantly male and older than a clinical trial population. Veterans are more likely to belong to a minority group, more likely to have lower level education and more likely to be poor than a clinical trial population. They are more likely to have poorer health status with higher number of medical conditions and psychosocial conditions.15
Limitations
We acknowledge several limitations to our study, such as the small number of patients and the retrospective single center nature of this study. Patients were older men with multiple psychosocial comorbitities like mental illness, substance abuse, and homelessness. This cohort may not represent the non-VA population, but is an excellent representation of a frail, real-world veteran population.
Conclusions
Despite clinical trials showing the promise of immunotherapy as an attractive front-line systemic treatment option for HCC, our results show poor outcomes in a frail real-world population. In a cohort of patients who received immunotherapy as a front-line systemic treatment for HCC, results were poor with a response rate of 14.3%, a median PFS of 4 months, and a median OS of 8 months. We noted a significantly higher number of adverse effects, including 21% incidence of grade 5 hepatotoxicity. There remains an urgent need to develop more effective and safer therapies for this patient population as well as validation from larger real-world studies.
1. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127. doi:10.1056/NEJMra1001683
2. Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359-E386. doi:10.1002/ijc.29210
3. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362(9399):1907-1917. doi:10.1016/S0140-6736(03)14964-1
4. Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol. 2013;47 Suppl(0):S2-S6. doi:10.1097/MCG.0b013e3182872f29
5. Seymour L, Bogaerts J, Perrone A, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics [published correction appears in Lancet Oncol. 2019 May;20(5):e242]. Lancet Oncol. 2017;18(3):e143-e152. doi:10.1016/S1470-2045(17)30074-8
6. El-Khoueiry AB, Sangro B, Yau T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389(10088):2492-2502.doi:10.1016/S0140-6736(17)31046-2
7. Zhu AX, Finn RS, Edeline J, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial [published correction appears in Lancet Oncol. 2018 Sep;19(9):e440]. Lancet Oncol. 2018;19(7):940-952. doi:10.1016/S1470-2045(18)30351-6
8. US Food and Drug Administration. FDA grants accelerated approval to nivolumab for HCC previously treated with sorafenib. Updated September 25, 2017. Accessed October 7, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-hcc-previously-treated-sorafenib.
9. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for hepatocellular carcinoma. Updated December 14, 2018. Accessed October 7, 2020. https://www.fda.gov/drugs/fda-grants-accelerated-approval-pembrolizumab-hepatocellular-carcinoma.
10. Yau T, Park JW, Finn RS, et al. CheckMate 459: A randomized, multi-center phase 3 study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019. Ann Onc. 2019;30(suppl_5):v851-v934. doi:10.1093/annonc/mdz394
11. Lee M, Ryoo BY, Hsu CH, et al. Randomised efficacy and safety results for atezolizumab (atezo) + bevacizumab (bev) in patients (pts) with previously untreated, unresectable hepatocellular carcinoma (HCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019.
12. Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894-1905.doi:10.1056/NEJMoa1915745
13. Scheiner B, Kirstein MM, Hucke F, et al. Programmed cell death protein-1 (PD-1)-targeted immunotherapy in advanced hepatocellular carcinoma: efficacy and safety data from an international multicentre real-world cohort. Aliment Pharmacol Ther. 2019;49(10):1323-1333. doi:10.1111/apt.15245
14. Yoon SE, Hur JY, Lee KK, et al. Real-world data on nivolumab treatment in Asian patients with advanced hepatocellular carcinoma. Presented at: ESMO 2018 Congress. Munich, Germany: October 21, 2018. Ann Onc. 2018;29(suppl_8):viii205-viii270. doi:10.1093/annonc/mdy282
15. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252
Hepatocellular carcinoma (HCC) has a poor prognosis and remains an important cause of cancer-related morbidity and mortality.1,2 Potentially curative interventions include surgical resection, radiofrequency ablation, and liver transplantation. However, the majority of patients are not eligible for these procedures because they are diagnosed at an advanced stage, when locoregional therapies are much more limited.3,4 Although the kinase inhibitors sorafenib and lenvatinib are approved as first-line systemic treatment, at the US Department of Veterans Affairs (VA) Kansas City VA Medical Center (KCVAMC) in Missouri, nivolumab was used instead because of concerns for the tolerability of the kinase inhibitors. Locoregional therapies, resection, and transplantation options were either not appropriate or had been exhausted for these patients. The objective of this retrospective study was to determine the outcomes of those veteran patients in a small cohort.
Methods
The KCVAMC Institutional Review Board approved this retrospective chart review. Patients were selected from pharmacy records at KCVAMC. We identified all patients with a diagnosis of HCC who received nivolumab from January 2016 to December 2019. We then included only the patients that had nivolumab in the front-line setting for our final analysis. At the time of initiation of treatment, all patients were informed that immunotherapy was not approved for front-line treatment, but available evidence suggested that it would be easier to tolerate than sorafenib or lenvatinib. These patients were determined to be either ineligible for sorafenib or lenvatinib therapy or expected to tolerate it poorly, and hence they consented to the use of nivolumab. Tumor response and progression were assessed by the investigator according to iRECIST (Immune Response Evaluation Criteria in Solid Tumors) criteria.5 Data were obtained from retrospective health record review.
Results
Fourteen men received nivolumab in the front-line systemic therapy setting from January 2016 to December 2019 at KCVAMC. The median age was 63.5 years (range, 58-72 years), and the median Eastern Cooperative Oncology Group score was 1. The Table highlights patient characteristics.
Of the 14 patients included in the review, 2 patients had a response to nivolumab (14.3%) and 1 patient had a complete response (7.1%). The median duration of immunotherapy was 4.5 months. Immunotherapy was discontinued due to disease progression in 10 patients and toxicity in 3 patients.
The median progression-free survival (PFS) from initiation of immunotherapy was 4 months; median overall survival (OS) was 8 months. The median time from diagnosis to survival was 41 months. Only 1 patient received a second-line treatment.
Incidence of grade 3 or higher toxicity was 35%. Three deaths resulted from auto-immune hepatitis (grade 5 toxicity), as well as 1 grade 3 skin toxicity, and 1 grade 4 liver toxicity.
Discussion
Immunotherapy has shown promise in patients with HCC based on the results of the KEYNOTE-224 and Checkmate-040 studies,6,7 which led to an accelerated US Food and Drug Administration approval of nivolumab and pembrolizumab for HCC following failure of first-line sorafenib.8,9
Several clinical trials are evaluating front-line immunotherapy for HCC. The Checkmate 459 study demonstrated the median OS to be 16.4 months for nivolumab vs 14.7 months for sorafenib, a difference that was not statistically significant. However, tolerability of nivolumab was better than it was for sorafenib, thus positioning it as a potentially attractive first-line option.10 The GO30140 study evaluated
The results from our study differed from the previous studies and raise concern for the applicability of these trials to a real-world population. For example, both the GO30140 and IMbrave150 excluded patients with untreated varices.11,12 Both IMbrave150 and Checkmate 459 limited enrollment only to patients with a Child-Pugh A score for liver disease; 36% of the KCVAMC patients had a Child-Pugh B score. Three patients (21.4%) were homeless, 6 patients (42.8%) had substance abuse history and 5 patients (35.7%) had mental illness. Several psychosocial factors present in our patients, such as substance abuse, mental illness, and homelessness, would have excluded them from clinical trials. Our small cohort of patients, thus, represents a frail real-world population due to multiple medical and psychosocial comorbidities. Real-world experience with immunotherapy as second-line therapy after treatment with sorafenib has been reported, but this is the first reported real-world experience of immunotherapy in the front-line setting for HCC.13,14
Large differences in sociodemographic status and health status exist between the veteran population and typical clinical trial populations. Veterans are predominantly male and older than a clinical trial population. Veterans are more likely to belong to a minority group, more likely to have lower level education and more likely to be poor than a clinical trial population. They are more likely to have poorer health status with higher number of medical conditions and psychosocial conditions.15
Limitations
We acknowledge several limitations to our study, such as the small number of patients and the retrospective single center nature of this study. Patients were older men with multiple psychosocial comorbitities like mental illness, substance abuse, and homelessness. This cohort may not represent the non-VA population, but is an excellent representation of a frail, real-world veteran population.
Conclusions
Despite clinical trials showing the promise of immunotherapy as an attractive front-line systemic treatment option for HCC, our results show poor outcomes in a frail real-world population. In a cohort of patients who received immunotherapy as a front-line systemic treatment for HCC, results were poor with a response rate of 14.3%, a median PFS of 4 months, and a median OS of 8 months. We noted a significantly higher number of adverse effects, including 21% incidence of grade 5 hepatotoxicity. There remains an urgent need to develop more effective and safer therapies for this patient population as well as validation from larger real-world studies.
Hepatocellular carcinoma (HCC) has a poor prognosis and remains an important cause of cancer-related morbidity and mortality.1,2 Potentially curative interventions include surgical resection, radiofrequency ablation, and liver transplantation. However, the majority of patients are not eligible for these procedures because they are diagnosed at an advanced stage, when locoregional therapies are much more limited.3,4 Although the kinase inhibitors sorafenib and lenvatinib are approved as first-line systemic treatment, at the US Department of Veterans Affairs (VA) Kansas City VA Medical Center (KCVAMC) in Missouri, nivolumab was used instead because of concerns for the tolerability of the kinase inhibitors. Locoregional therapies, resection, and transplantation options were either not appropriate or had been exhausted for these patients. The objective of this retrospective study was to determine the outcomes of those veteran patients in a small cohort.
Methods
The KCVAMC Institutional Review Board approved this retrospective chart review. Patients were selected from pharmacy records at KCVAMC. We identified all patients with a diagnosis of HCC who received nivolumab from January 2016 to December 2019. We then included only the patients that had nivolumab in the front-line setting for our final analysis. At the time of initiation of treatment, all patients were informed that immunotherapy was not approved for front-line treatment, but available evidence suggested that it would be easier to tolerate than sorafenib or lenvatinib. These patients were determined to be either ineligible for sorafenib or lenvatinib therapy or expected to tolerate it poorly, and hence they consented to the use of nivolumab. Tumor response and progression were assessed by the investigator according to iRECIST (Immune Response Evaluation Criteria in Solid Tumors) criteria.5 Data were obtained from retrospective health record review.
Results
Fourteen men received nivolumab in the front-line systemic therapy setting from January 2016 to December 2019 at KCVAMC. The median age was 63.5 years (range, 58-72 years), and the median Eastern Cooperative Oncology Group score was 1. The Table highlights patient characteristics.
Of the 14 patients included in the review, 2 patients had a response to nivolumab (14.3%) and 1 patient had a complete response (7.1%). The median duration of immunotherapy was 4.5 months. Immunotherapy was discontinued due to disease progression in 10 patients and toxicity in 3 patients.
The median progression-free survival (PFS) from initiation of immunotherapy was 4 months; median overall survival (OS) was 8 months. The median time from diagnosis to survival was 41 months. Only 1 patient received a second-line treatment.
Incidence of grade 3 or higher toxicity was 35%. Three deaths resulted from auto-immune hepatitis (grade 5 toxicity), as well as 1 grade 3 skin toxicity, and 1 grade 4 liver toxicity.
Discussion
Immunotherapy has shown promise in patients with HCC based on the results of the KEYNOTE-224 and Checkmate-040 studies,6,7 which led to an accelerated US Food and Drug Administration approval of nivolumab and pembrolizumab for HCC following failure of first-line sorafenib.8,9
Several clinical trials are evaluating front-line immunotherapy for HCC. The Checkmate 459 study demonstrated the median OS to be 16.4 months for nivolumab vs 14.7 months for sorafenib, a difference that was not statistically significant. However, tolerability of nivolumab was better than it was for sorafenib, thus positioning it as a potentially attractive first-line option.10 The GO30140 study evaluated
The results from our study differed from the previous studies and raise concern for the applicability of these trials to a real-world population. For example, both the GO30140 and IMbrave150 excluded patients with untreated varices.11,12 Both IMbrave150 and Checkmate 459 limited enrollment only to patients with a Child-Pugh A score for liver disease; 36% of the KCVAMC patients had a Child-Pugh B score. Three patients (21.4%) were homeless, 6 patients (42.8%) had substance abuse history and 5 patients (35.7%) had mental illness. Several psychosocial factors present in our patients, such as substance abuse, mental illness, and homelessness, would have excluded them from clinical trials. Our small cohort of patients, thus, represents a frail real-world population due to multiple medical and psychosocial comorbidities. Real-world experience with immunotherapy as second-line therapy after treatment with sorafenib has been reported, but this is the first reported real-world experience of immunotherapy in the front-line setting for HCC.13,14
Large differences in sociodemographic status and health status exist between the veteran population and typical clinical trial populations. Veterans are predominantly male and older than a clinical trial population. Veterans are more likely to belong to a minority group, more likely to have lower level education and more likely to be poor than a clinical trial population. They are more likely to have poorer health status with higher number of medical conditions and psychosocial conditions.15
Limitations
We acknowledge several limitations to our study, such as the small number of patients and the retrospective single center nature of this study. Patients were older men with multiple psychosocial comorbitities like mental illness, substance abuse, and homelessness. This cohort may not represent the non-VA population, but is an excellent representation of a frail, real-world veteran population.
Conclusions
Despite clinical trials showing the promise of immunotherapy as an attractive front-line systemic treatment option for HCC, our results show poor outcomes in a frail real-world population. In a cohort of patients who received immunotherapy as a front-line systemic treatment for HCC, results were poor with a response rate of 14.3%, a median PFS of 4 months, and a median OS of 8 months. We noted a significantly higher number of adverse effects, including 21% incidence of grade 5 hepatotoxicity. There remains an urgent need to develop more effective and safer therapies for this patient population as well as validation from larger real-world studies.
1. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127. doi:10.1056/NEJMra1001683
2. Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359-E386. doi:10.1002/ijc.29210
3. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362(9399):1907-1917. doi:10.1016/S0140-6736(03)14964-1
4. Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol. 2013;47 Suppl(0):S2-S6. doi:10.1097/MCG.0b013e3182872f29
5. Seymour L, Bogaerts J, Perrone A, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics [published correction appears in Lancet Oncol. 2019 May;20(5):e242]. Lancet Oncol. 2017;18(3):e143-e152. doi:10.1016/S1470-2045(17)30074-8
6. El-Khoueiry AB, Sangro B, Yau T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389(10088):2492-2502.doi:10.1016/S0140-6736(17)31046-2
7. Zhu AX, Finn RS, Edeline J, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial [published correction appears in Lancet Oncol. 2018 Sep;19(9):e440]. Lancet Oncol. 2018;19(7):940-952. doi:10.1016/S1470-2045(18)30351-6
8. US Food and Drug Administration. FDA grants accelerated approval to nivolumab for HCC previously treated with sorafenib. Updated September 25, 2017. Accessed October 7, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-hcc-previously-treated-sorafenib.
9. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for hepatocellular carcinoma. Updated December 14, 2018. Accessed October 7, 2020. https://www.fda.gov/drugs/fda-grants-accelerated-approval-pembrolizumab-hepatocellular-carcinoma.
10. Yau T, Park JW, Finn RS, et al. CheckMate 459: A randomized, multi-center phase 3 study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019. Ann Onc. 2019;30(suppl_5):v851-v934. doi:10.1093/annonc/mdz394
11. Lee M, Ryoo BY, Hsu CH, et al. Randomised efficacy and safety results for atezolizumab (atezo) + bevacizumab (bev) in patients (pts) with previously untreated, unresectable hepatocellular carcinoma (HCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019.
12. Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894-1905.doi:10.1056/NEJMoa1915745
13. Scheiner B, Kirstein MM, Hucke F, et al. Programmed cell death protein-1 (PD-1)-targeted immunotherapy in advanced hepatocellular carcinoma: efficacy and safety data from an international multicentre real-world cohort. Aliment Pharmacol Ther. 2019;49(10):1323-1333. doi:10.1111/apt.15245
14. Yoon SE, Hur JY, Lee KK, et al. Real-world data on nivolumab treatment in Asian patients with advanced hepatocellular carcinoma. Presented at: ESMO 2018 Congress. Munich, Germany: October 21, 2018. Ann Onc. 2018;29(suppl_8):viii205-viii270. doi:10.1093/annonc/mdy282
15. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252
1. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127. doi:10.1056/NEJMra1001683
2. Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359-E386. doi:10.1002/ijc.29210
3. Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362(9399):1907-1917. doi:10.1016/S0140-6736(03)14964-1
4. Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol. 2013;47 Suppl(0):S2-S6. doi:10.1097/MCG.0b013e3182872f29
5. Seymour L, Bogaerts J, Perrone A, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics [published correction appears in Lancet Oncol. 2019 May;20(5):e242]. Lancet Oncol. 2017;18(3):e143-e152. doi:10.1016/S1470-2045(17)30074-8
6. El-Khoueiry AB, Sangro B, Yau T, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389(10088):2492-2502.doi:10.1016/S0140-6736(17)31046-2
7. Zhu AX, Finn RS, Edeline J, et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial [published correction appears in Lancet Oncol. 2018 Sep;19(9):e440]. Lancet Oncol. 2018;19(7):940-952. doi:10.1016/S1470-2045(18)30351-6
8. US Food and Drug Administration. FDA grants accelerated approval to nivolumab for HCC previously treated with sorafenib. Updated September 25, 2017. Accessed October 7, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-nivolumab-hcc-previously-treated-sorafenib.
9. US Food and Drug Administration. FDA grants accelerated approval to pembrolizumab for hepatocellular carcinoma. Updated December 14, 2018. Accessed October 7, 2020. https://www.fda.gov/drugs/fda-grants-accelerated-approval-pembrolizumab-hepatocellular-carcinoma.
10. Yau T, Park JW, Finn RS, et al. CheckMate 459: A randomized, multi-center phase 3 study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019. Ann Onc. 2019;30(suppl_5):v851-v934. doi:10.1093/annonc/mdz394
11. Lee M, Ryoo BY, Hsu CH, et al. Randomised efficacy and safety results for atezolizumab (atezo) + bevacizumab (bev) in patients (pts) with previously untreated, unresectable hepatocellular carcinoma (HCC). Presented at: ESMO 2019 Congress. Barcelona, Spain: September 27, 2019.
12. Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382(20):1894-1905.doi:10.1056/NEJMoa1915745
13. Scheiner B, Kirstein MM, Hucke F, et al. Programmed cell death protein-1 (PD-1)-targeted immunotherapy in advanced hepatocellular carcinoma: efficacy and safety data from an international multicentre real-world cohort. Aliment Pharmacol Ther. 2019;49(10):1323-1333. doi:10.1111/apt.15245
14. Yoon SE, Hur JY, Lee KK, et al. Real-world data on nivolumab treatment in Asian patients with advanced hepatocellular carcinoma. Presented at: ESMO 2018 Congress. Munich, Germany: October 21, 2018. Ann Onc. 2018;29(suppl_8):viii205-viii270. doi:10.1093/annonc/mdy282
15. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252