User login
Should treatment be initiated for mild chronic hypertension in pregnancy to improve outcomes?
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Tita AT, Szychowski JM, Boggess K, et al. Treatment for mild chronic hypertension during pregnancy. N Engl J Med. 2022;386:1781-1792. doi: 10.1056/NEJMoa2201295.
Expert Commentary
In the nonpregnant population, medical management of hypertension >140/90 mm Hg is standard practice. By contrast, much higher blood pressures (BPs; up to 160/110 mm Hg) traditionally have been tolerated in pregnant patients with chronic hypertension without initiating treatment, and existing medications are often discontinued during pregnancy. Concern for impaired fetal growth as well as lack of data on improved outcomes have led to different recommendations for the management of mild chronic hypertension in pregnancy. However, chronic hypertension affects a substantial number of pregnant patients and is known to be a risk factor for severe short-term pregnancy and long-term health complications. With preliminary data suggesting that BPs >140/90 mm Hg prior to 20 weeks’ gestation are associated with an increase in adverse outcomes, Tita and colleagues sought to determine the effects of decreasing BP in pregnant patients with mild chronic hypertension.
Details about the study
This is an investigator-initiated, multicenter, pragmatic, open-label, randomized control trial of 2,408 patients with mild chronic hypertension. The active treatment group was treated with antihypertensive medication (including titration of existing medication), targeting a BP of <140/90 mm Hg. The control group only received medication for severe hypertension (≥160 mm Hg systolic or ≥105 mm Hg diastolic). The primary outcome of the study was a composite of preeclampsia with severe features, medically indicated preterm delivery prior to 35 weeks’ gestation (not spontaneous labor or rupture of membranes), placental abruption, and fetal or neonatal death. Birthweight that was less than the 10th percentile was used as a safety outcome. The hypothesis was that treatment would decrease the rate of adverse pregnancy and fetal/neonatal outcomes.
The patient population of singleton pregnancies at a gestational age of less than 23 weeks included 56% with known chronic hypertension on medications, 22% with known chronic hypertension without medications, and 22% with newly diagnosed (during pregnancy) chronic hypertension. The treatment group primarily received labetalol (61.7%) or nifedipine (35.6%); the maximum dose of a single agent was used as tolerated prior to adding a second agent. The control group only received an antihypertensive medication for severe hypertension.
Treatment of chronic hypertension demonstrated a decreased risk of the composite adverse outcome with an adjusted risk ratio of 0.82 (95% confidence interval [CI], 0.74 to 0.92; P<.001) and a number needed to treat (NNT) of 14.7. When analyzed separately, a similar risk reduction was noted for both preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation. There was no statistical difference between the groups for birth weight <10th percentile and <5th percentile (adjusted risk ratio, 1.04 [0.82–1.31] vs 0.89 [0.62–1.26], respectively).
Planned subgroup analysis by type of chronic hypertension, race/ethnic group, diabetes status, gestational age at baseline, and body mass index (BMI) demonstrated a similar treatment effect to the overall composite primary outcome, with the exception of patients with newly diagn
Study strengths and weaknesses
The study strengths cited are a large sample size, multiple study sites, an independent data and safety monitoring board with close oversight, and centralized blinded confirmation of outcomes. Another strength is that the patient population of the study was similar to the overall population of pregnant patients in the United States with chronic hypertension in terms of age, race, and ethnicity.
The weaknesses of the study include the open-label design and the high ratio of screened to enrolled patients. Both of these issues appear related to the study design (ethics and logistics of a blinded treatment and gestational age cutoff) and the physiology of pregnancy (expected decrease in BP in the second trimester rendering patients ineligible due to lower BP). The study was also not powered to assess treatment effect in all of the subgroups, and further evaluation of patients with newly diagnosed chronic hypertension and BMI ≥ 40 kg/m2 is needed. ●
Pregnant patients with chronic hypertension should continue or initiate antihypertensive medication to target a BP goal of <140/90 mm Hg. This substantial practice change is supported by the significant decrease demonstrated in this study in adverse outcomes such as preeclampsia with severe features and medically indicated preterm birth <35 weeks’ gestation without an increase in small-for-gestational-age newborns.
Misoprostol: Clinical pharmacology in obstetrics and gynecology
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
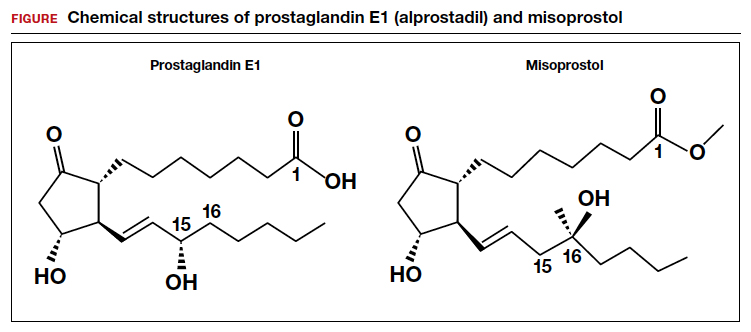
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.
Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.

Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
Oxytocin and prostaglandins are critically important regulators of uterine contraction. Obstetrician-gynecologists commonly prescribe oxytocin and prostaglandin agonists (misoprostol, dinoprostone) to stimulate uterine contraction for the induction of labor, prevention and treatment of postpartum hemorrhage, and treatment of miscarriage and fetal demise. The focus of this editorial is the clinical pharmacology of misoprostol.
Misoprostol is approved by the US Food and Drug Administration (FDA) for the prevention and treatment of nonsteroidal anti-inflammatory drug–induced gastric ulcers and for patients at high risk for gastric ulcers, including those with a history of gastric ulcers. The approved misoprostol route and dose for this indication is oral administration of 200 µg four times daily with food.1 Recent food intake and antacid use reduces the absorption of orally administered misoprostol. There are no FDA-approved indications for the use of misoprostol as a single agent in obstetrics and gynecology. The FDA has approved the combination of mifepristone and misoprostol for medication abortion in the first trimester. In contrast to misoprostol, PGE2 (dinoprostone) is approved by the FDA as a vaginal insert containing 10 mg of dinoprostone for the initiation and/or continuation of cervical ripening in patients at or near term in whom there is a medical or obstetric indication for induction of labor (Cervidil; Ferring Pharmaceuticals Inc, Parsippany, New Jersey).2
Pharmacology of misoprostol
Misoprostol is a prostaglandin E1 (PGE1) agonist analogue. Prostaglandin E1 (alprostadil) is rapidly metabolized, has a half-life in the range of minutes and is not orally active, requiring administration by intravenous infusion or injection. It is indicated to maintain a patent ductus arteriosus in newborns with ductal-dependent circulation and to treat erectile dysfunction.3 In contrast to PGE1, misoprostol has a methyl ester group at carbon-1 (C-1) that increases potency and duration of action. Misoprostol also has no hydroxyl group at C-15, replacing that moiety with the addition of both a methyl- and hydroxyl- group at C-16 (FIGURE). These molecular changes improve oral activity and increase duration of action.4 Pure misoprostol is a viscous oil. It is formulated into tables by dispersing the oil on hydroxypropyl methyl cellulose before compounding into tablets. Unlike naturally occurring prostaglandins (PGE1), misoprostol tablets are stabile at room temperature for years.4
Following absorption, the methyl ester at C-1 is enzymatically cleaved, yielding misoprostol acid, the active drug.4 Misoprostol binds to the E prostanoid receptor 3 (EP-3).5 Activation of myometrial EP-3 receptor induces an increase in intracellular phosphoinositol turnover and calcium mobilization, resulting in an increase in intracellular-free calcium, triggering actin-myosin contractility.6 The increase in free calcium is propagated cell-to-cell through gap junctions that link the myometrial cells to facilitate the generation of a coordinated contraction.

Misoprostol: Various routes of administration are not equal
Misoprostol can be given by an oral, buccal, vaginal, or rectal route of administration. To study the effect of the route of administration on uterine tone and contractility, investigators randomly assigned patients at 8 to 11 weeks’ gestation to receive misoprostol 400 µg as a single dose by the oral or vaginal route. Uterine tone and contractility were measured using an intrauterine pressure transducer. Compared to vaginal administration, oral administration of misprostol was associated with rapid attainment of peak plasma level at 30 minutes, followed by a decline in concentration by 60 minutes. This rapid onset and rapid offset of plasma concentration was paralleled by the onset of uterine tone within 8 minutes, but surprisingly no sustained uterine contractions.7 By contrast, following vaginal administration of misoprostol, serum levels rose slowly and peaked in 1 to 2 hours. Uterine tone increased within 21 minutes, and sustained uterine contractions were recorded for 4 hours.7 The rapid rise and fall in plasma misoprostol following oral administration and the more sustained plasma misoprostol concentration over 4 hours has been previously reported.8 In a second study involving patients 8 to 11 weeks’ gestation, the effect of a single dose of misoprostol 400 µg by an oral or vaginal route on uterine contractility was compared using an intrauterine pressure transducer.9 Confirming previous results, the time from misoprostol administration to increased uterine tone was more rapid with oral than with vaginal administration (8 min vs 19 min). Over the course of 4 hours, uterine contraction activity was greater with vaginal than with oral administration (454 vs 166 Montevideo units).9
Both studies reported that oral administration of misoprostol resulted in more rapid onset and offset of action than vaginal administration. Oral administration of a single dose of misoprostol 400 µg did not result in sustained uterine contractions in most patients in the first trimester. Vaginal administration produced a slower onset of increased uterine tone but sustained uterine contractions over 4 hours. Compared with vaginal administration of misoprostol, the rapid onset and offset of action of oral misoprostol may reduce the rate of tachysystole and changes in fetal heart rate observed with vaginal administration.10
An important finding is that buccal and vaginal administration of misoprostol have similar effects on uterine tone in the first trimester.11 To study the effect of buccal and vaginal administration of misoprostol on uterine tone, patients 6 to 13 weeks’ gestation were randomly allocated to receive a single dose of misoprostol 400 µg by a buccal or vaginal route.11 Uterine activity over 5 hours following administration was assessed using an intrauterine pressure transducer. Uterine tone 20 to 30 minutes after buccal or vaginal administration of misoprostol (400 µg) was 27 and 28 mm Hg, respectively. Peak uterine tone, as measured by an intrauterine pressure transducer, for buccal and vaginal administration of misoprostol was 49 mm Hg and 54 mm Hg, respectively. Total Alexandria units (AU) over 5 hours following buccal or vaginal administration was 6,537 AU and 6,090 AU, respectively.11
An AU is calculated as the average amplitude of the contractions (mm Hg) multiplied by the average duration of the contractions (min) multiplied by average frequency of contraction over 10 minutes.12 By contrast, a Montevideo unit does not include an assessment of contraction duration and is calculated as average amplitude of contractions (mm Hg) multiplied by frequency of uterine contractions over 10 minutes.12
In contrast to buccal or vaginal administration, rectal administration of misoprostol resulted in much lower peak uterine tone and contractility as measured by a pressure transducer. Uterine tone 20 to 30 minutes after vaginal and rectal administration of misoprostol (400 µg) was 28 and 19 mm Hg, respectively.11 Peak uterine tone, as measured by an intrauterine pressure transducer, for vaginal and rectal administration of misoprostol was 54 and 31 mm Hg, respectively. AUs over 5 hours following vaginal and rectal administration was 6,090 AU and 2,768 AU, respectively.11 Compared with buccal and vaginal administration of misoprostol, rectal administration produced less sustained uterine contractions in the first trimester of pregnancy. To achieve maximal sustained uterine contractions, buccal and vaginal routes of administration are superior to oral and rectal administration.
Continue to: Misoprostol and cervical ripening...
Misoprostol and cervical ripening
Misoprostol is commonly used to soften and ripen the cervix. Some of the cervical ripening effects of misoprostol are likely due to increased uterine tone. In addition, misoprostol may have a direct effect on the collagen structure of the cervix. To study the effect of misoprostol on the cervix, pregnant patients in the first trimester were randomly assigned to receive misoprostol 200 µg by vaginal self-administration, isosorbide mononitrate (IMN) 40 mg by vaginal self-administration or no treatment the evening prior to pregnancy termination.13 The following day, before uterine evacuation, a cervical biopsy was obtained for electron microscopy studies and immunohistochemistry to assess the presence of enzymes involved in collagen degradation, including matrix metalloproteinase 1 (MMP-1) and matrix metalloproteinase 9 (MMP-9). Electron microscopy demonstrated that pretreatment with misoprostol resulted in a pronounced splitting and disorganization of collagen fibers.13 Compared with misoprostol treatment, IMN produced less splitting and disorganization of collagen fibers, and in the no treatment group, no marked changes in the collagen framework were observed.
Compared with no treatment, misoprostol and IMN pretreatment were associated with marked increases in MMP-1 and MMP-9 as assessed by immunohistochemistry. Misoprostol pretreatment also resulted in a significant increase in interleukin-8 concentration compared with IMN pretreatment and no treatment (8.8 vs 2.7 vs 2.4 pg/mg tissue), respectively.13 Other investigators have also reported that misoprostol increased cervical leukocyte influx and collagen disrupting enzymes MMP-8 and MMP-9.14,15
An open-label clinical trial compared the efficacy of misoprostol versus Foley catheter for labor induction at term in 1,859 patients ≥ 37 weeks’ gestation with a Bishop score <6.16 Patients were randomly allocated to misoprostol (50 µg orally every 4 hours up to 3 times in 24 hours) versus placement of a 16 F or 18 F Foley catheter introduced through the cervix, filled with 30 mL of sodium chloride or water. The investigators reported that oral misoprostol and Foley catheter cervical ripening had similar safety and effectiveness for cervical ripening as a prelude to induction of labor, including no statistically significant differences in 5-minute Apgar score <7, umbilical cord artery pH ≤ 7.05, postpartum hemorrhage, or cesarean birth rate.16
Bottom line
Misoprostol and oxytocin are commonly prescribed in obstetric practice for cervical ripening and induction of labor, respectively. The dose and route of administration of misoprostol influences the effect on the uterus. For cervical ripening, where rapid onset and offset may help to reduce the risk of uterine tachysystole and worrisome fetal heart rate changes, low-dose (50 µg) oral administration of misoprostol may be a preferred dose and route. For the treatment of miscarriage and fetal demise, to stimulate sustained uterine contractions over many hours, buccal and vaginal administration of misoprostol are preferred. Rectal administration is generally inferior to buccal and vaginal administration for stimulating sustained uterine contractions and its uses should be limited. ●
Common side effects of misoprostol are abdominal cramping, diarrhea, nausea, vomiting, headache, and fever. Elevated temperature following misoprostol administration is a concerning side effect that may require further investigation to rule out an infection, especially if the elevated temperature persists for > 4 hours. The preoptic area of the anterior hypothalamus (POAH) plays a major role in thermoregulation. When an infection causes an increase in endogenous pyrogens, including interleukin-1β, interleukin-6 and tumor necrosis factor, prostaglandins are generated in the region of the POAH, increasing the thermoregulatory set point, triggering cutaneous vasoconstriction and shivering and non-shivering thermogenesis.1 Misoprostol, especially at doses >400 µg commonly causes both patient-reported chills and temperature elevation >38° C.
In a study comparing misoprostol and oxytocin for the management of the third stage of labor, 597 patients were randomly allocated to receive oxytocin 10 units by intramuscular injection or misoprostol 400 µg or 600 µg by the oral route.2 Patient-reported shivering occurred in 13%, 19%, and 28% of patients receiving oxytocin, misoprostol 400 µg and misoprostol 800 µg, respectively. A recorded temperature >38° C occurred within 1 hour of medication administration in approximately 3%, 2%, and 7.5% of patients receiving oxytocin, misoprostol 400 µg, and misoprostol 800 µg, respectively. In another study, 453 patients scheduled for a cesarean birth were randomly allocated to receive 1 of 3 doses of rectal misoprostol 200 μg, 400 μg, or 600 μg before incision. Fever was detected in 2.6%, 9.9%, and 5.1% of the patients receiving misoprostol 200 μg, 400 μg, or 600 μg, respectively.3
References
1. Aronoff DM, Neilson EG. Antipyretics: mechanisms of action and clinical use in fever suppression. Am J Med. 2001;111:304-315. doi: 10.1016/s0002-9343(01)00834-8.
2. Lumbiganon P, Hofmeyr J, Gumezoglu AM, et al. Misoprostol dose-related shivering and pyrexia in the third stage of labor. WHO Collaborative Trial of Misoprostol in the Management of the Third Stage of Labor. Br J Obstet Gynaecol. 1999;106:304-308. doi: 10.1111/j.1471-0528.1999.tb08266.x.
3. Sweed M, El-Said M, Abou-Gamrah AA, et al. Comparison between 200, 400 and 600 microgram rectal misoprostol before cesarean section: a randomized clinical trial. J Obstet Gynaecol Res. 2019;45:585-591. doi: 10.1111 /jog.13883.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.
- Cytotec [package insert]. Chicago, IL: GD Searle & Co. https://www.accessdata.fda.gov/drugsatfda_docs/label/2002/19268slr037.pdf. Accessed June 20, 2022.
- Cervidil [package insert]. St Louis, MO: Forrest Pharmaceuticals Inc.; May 2006. Accessed June 20, 2022.
- Caverject [package insert]. New York, NY: Pfizer Inc.; March 2014. Accessed June 20, 2022.
- Collins PW. Misoprostol: discovery, development and clinical applications. Med Res Rev. 1990;10:149-172. doi: 10.1002/med.2610100202.
- Audit M, White KI, Breton B, et al. Crystal structure of misoprostol bound to the labor inducer prostaglandin E2 receptor. Nat Chem Biol. 2019;15:11-17. doi: 10.1038/s41589-018-0160-y.
- Pallliser KH, Hirst JJ, Ooi G, et al. Prostaglandin E and F receptor expression and myometrial sensitivity in labor onset in the sheep. Biol Reprod. 2005;72:937-943. doi: 10.1095/biolreprod.104.035311.
- Gemzell-Danilesson K, Marions L, Rodriguez A, et al. Comparison between oral and vaginal administration of misoprostol on uterine contractility. Obstet Gynecol. 1999;93:275-280. doi: 10.1016/s0029-7844(98)00436-0.
- Zieman M, Fong SK, Benowitz NL, et al. Absorption kinetics of misoprostol with oral or vaginal administration. Obstet Gynecol. 1997;90:88-92. doi: 10.1016/S0029-7844(97)00111-7.
- Aronsson A, Bygdeman M, Gemzell-Danielsson K. Effects of misoprostol on uterine contractility following different routes of administration. Hum Reprod. 2004;19:81-84. doi: 10.1093/humrep/deh005.
- Young DC, Delaney T, Armson BA, et al. Oral misoprostol, low dose vaginal misoprostol and vaginal dinoprostone for labor induction: randomized controlled trial. PLOS One. 2020;15:e0227245. doi: 10.1371/journal.pone.0227245.
- Meckstroth KR, Whitaker AK, Bertisch S, et al. Misoprostol administered by epithelial routes. Drug absorption and uterine response. Obstet Gynecol. 2006;108:582-590. doi: 10.1097/01.AOG.0000230398.32794.9d.
- el-Sahwi S, Gaafar AA, Toppozada HK. A new unit for evaluation of uterine activity. Am J Obstet Gynecol. 1967;98:900-903. doi: 10.1016/0002-9378(67)90074-9.
- Vukas N, Ekerhovd E, Abrahamsson G, et al. Cervical priming in the first trimester: morphological and biochemical effects of misoprostol and isosorbide mononitrate. Acta Obstet Gyecol. 2009;88:43-51. doi: 10.1080/00016340802585440.
- Aronsson A, Ulfgren AK, Stabi B, et al. The effect of orally and vaginally administered misoprostol on inflammatory mediators and cervical ripening during early pregnancy. Contraception. 2005;72:33-39. doi: 10.1016/j.contraception.2005.02.012.
- Denison FC, Riley SC, Elliott CL, et al. The effect of mifepristone administration on leukocyte populations, matrix metalloproteinases and inflammatory mediators in the first trimester cervix. Mol Hum Reprod. 2000;6:541-548. doi: 10.1093/molehr/6.6.541.
- ten Eikelder MLG, Rengerink KO, Jozwiak M, et al. Induction of labour at term with oral misoprostol versus a Foley catheter (PROBAAT-II): a multicentre randomised controlled non-inferiority trial. Lancet. 2016;387:1619-1628. doi: 10.1016 /S0140-6736(16)00084-2.
What are the perinatal risks of SARS-CoV-2 infection in pregnancy?
Ferrara A, Hedderson MM, Zhu Y, et al. Perinatal complications in individuals in California with or without SARS-CoV-2 infection during pregnancy. JAMA Intern Med. 2022;182:503-512. doi:10.1001/jamainternmed.2022.0330
Expert Commentary
SARS-CoV-2 infection is associated with several adverse outcomes, with the magnitude of specific risks varying by population studied and study design used. Early Centers for Disease Control and Prevention (CDC) data demonstrated that pregnant women were at increased risk for severe illness, including risks of intensive care unit (ICU) admission, invasive ventilation, and extracorporeal membrane oxygenation, compared with non–pregnant women.1 Since then, other groups have confirmed the increased risks of severe COVID-19, and also identified pregnancy-specific risks, such as preeclampsia, cesarean delivery (CD), prematurity, venous thromboembolic (VTE) disease, and stillbirth.2-6
The recent study by Ferrara and colleagues adds more granular data to help refine understanding of COVID-19 in pregnancy and counsel patients.
Details of the study
The authors conducted a retrospective cohort study between March 1, 2020, and March 16, 2021, using the electronic health records (EHRs) from Kaiser Permanente Northern California, an integrated managed care organization that serves 4.5 million patients annually. Universal testing for SARS-CoV-2 upon admission for delivery began December 1, 2020; prior to this date, asymptomatic pregnant women were tested only for certain criteria (such as being a health care worker or having high-risk medical conditions).
Pregnant women were identified with SARS-CoV-2 based on 1) a positive polymerase chain reaction test result between 30 days prior to the last menstrual period up to 7 days after delivery or 2) an ICD-10 diagnosis of SARS-CoV-2 infection. Pregnant women not meeting these criteria were classified as SARS-CoV-2 negative. Women were followed through pregnancy to understand if they experienced preterm birth (spontaneous and medically indicated), gestational hypertension, preeclampsia/eclampsia, VTE disease, gestational diabetes, severe maternal morbidity (as defined by the CDC), hospitalization, and livebirth (or stillbirth), in order to consider the timing of SARS-CoV-2 infection relative to each of these outcomes (and ascertain whether SARS-CoV-2 infection preceded any of these outcomes more commonly than not). Management of pregnancies with COVID-19 across this large organization and multiple hospitals was not specified.
Identified perinatal risks
Among 43,886 pregnant women included in the cohort, 1,332 (3.0%) were diagnosed with SARS-CoV-2, with the vast majority of positive tests in the third trimester. Significant sociodemographic differences were noted between those with and without SARS-CoV-2, including differences in age, self-reported race/ethnicity, neighborhood deprivation index, and pre-pregnancy body mass index; no differences were noted for other pre-existing comorbidities, gestational week at delivery, or smoking in pregnancy.
In multivariable models, SARS-CoV-2 infection in pregnancy was associated with severe maternal morbidity, preterm birth, and VTE disease. It was not associated with stillbirth, any hypertensive disorder of pregnancy, CD, or any neonatal complication.
The prevalence of SARS-CoV-2 was 1.3% prior to and 8.0% after implementation of universal testing in pregnancy
A total of 307 of the 1,332 pregant women with SARS-CoV-2 were admitted to the hospital for symptomatic infection; 3 required noninvasive positive-pressure ventilation, and 1 required mechanical ventilation.
Continue to: Study strengths and weaknesses...
Study strengths and weaknesses
Strengths include the large, EHR-based dataset from a single organization, allowing for granular analysis on patient comorbidities and outcomes (rather than only based on diagnosis codes, as is true of many other large databases), as well as focus on relevant perinatal outcomes and thoughtful statistical modeling. However, a significant challenge with this, and many other studies, is ascertainment of SARS-CoV-2 infections throughout pregnancy. Asymptomatic and mildly symptomatic women, who may not be as likely to have adverse pregnancy outcomes, can often be counted in the unaffected population, biasing study results toward increased risks of SARS-CoV-2. Although the findings stratified by implementation of universal testing (which captures a greater fraction of asymptomatic patients at admission for delivery), do not suggest risk mitigation with asymptomatic status, this analysis did not capture asymptomatic infections earlier in pregnancy, many of which might not be associated with perinatal risk.
Another challenge with such a dataset is that one cannot determine the severity of illness of each patient without manual review of each chart; however, other data that are easily abstracted from the EHR may serve as a proxy. For instance, of the 307 women with symptomatic COVID-19, 4 required respiratory support above nasal cannula. This suggests a low rate of severely ill women, and may explain some of the findings in the study, such as no differences in the rate of CD, hypertensive disorders of pregnancy, or stillbirth, but does not explain the increased risk of both medically indicated and spontaneous preterm birth, or the rates of acute respiratory distress syndrome and sepsis that drive the increased risk of severe maternal morbidity.
The CDC has published data on the risks of stillbirth from a large hospital-based administrative database for COVID-19 from Premier Healthcare.2 In a cohort of over 1.2 million women admitted for delivery, including the timeframe of Ferrara et al’s study, COVID-19 was associated with a 2-fold increased risk of stillbirth, with higher risks noted with the delta variant. A rare outcome, stillbirth occurs in 6/1,000 births,7 which was the rate seen in Ferrara’s publication for both women with and without SARS-CoV-2 infection. The rare nature of the outcome may explain why a signal was not noted in the article of interest.
Translating data to patient counseling
Ferrara and colleagues’ study clearly confirms that COVID-19 infection has risks. Although many women with a COVID-19 infection in pregnancy may have an uncomplicated course, a favorable outcome is hard to predict with certainty. Risks of prematurity, VTE, organ dysfunction, and stillbirth from COVID-19 are rare but devastating complications. However, vaccinated women tend to incur far fewer adverse outcomes of COVID-19 in pregnancy, namely a 90% risk reduction in severe or critical COVID-19, with lower rates of ICU admissions and stillbirths.8 While these data strongly favor vaccination, we remain ill-advised on management strategies specifically to mitigate risk for the pregnancy once affected by COVID-19 infection. Thus, prevention with vaccination, mask wearing, and physical distancing remains a cornerstone of prenatal care in the current day. ●
These data continue to support that SARS-CoV-2 infection is associated with prematurity, VTE, and severe maternal adverse outcomes. As sports fanatics often state, the best defense is a good offense. In the case of SARS-CoV-2, COVID-19 vaccination, mask wearing, and physical distancing are likely the best offense against COVID-19 infection in pregnancy.
- Zambrano LD, Ellington S, Strid P, et al. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status - United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3.
- DeSisto CL. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morb Mortal Wkly Rep. 2021;70. doi:10.15585/mmwr.mm7047e1.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and metaanalysis. BMJ. 2020;370:m3320. doi:10.1136/bmj.m3320.
- Jering KS, Claggett BL, Cunningham JW, et al. Clinical characteristics and outcomes of hospitalized women giving birth with and without COVID-19. JAMA Intern Med. 2021;181:714-717. doi:10.1001/jamainternmed.2020.9241.
- Katz D, Bateman BT, Kjaer K, et al. The Society for Obstetric Anesthesia and Perinatology Coronavirus Disease 2019 Registry: an analysis of outcomes among pregnant women delivering during the initial severe acute respiratory syndrome Coronavirus-2 outbreak in the United States. Anesth Analg. 2021;133:462-473. doi:10.1213/ANE.0000000000005592.
- Metz TD, Clifton RG, Hughes BL, et al. Association of SARS-CoV-2 infection with serious maternal morbidity and mortality from obstetric complications. JAMA. 2022;327:748759. doi:10.1001/jama.2022.1190.
- Management of stillbirth. https ://www.acog.org/en/clinical/clinical-guidance/obstetric-care-consensus/articles/2020/03/management-of-stillbirth. Accessed May 23, 2022.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109. doi:10.1097/AOG.0000000000004621.
Ferrara A, Hedderson MM, Zhu Y, et al. Perinatal complications in individuals in California with or without SARS-CoV-2 infection during pregnancy. JAMA Intern Med. 2022;182:503-512. doi:10.1001/jamainternmed.2022.0330
Expert Commentary
SARS-CoV-2 infection is associated with several adverse outcomes, with the magnitude of specific risks varying by population studied and study design used. Early Centers for Disease Control and Prevention (CDC) data demonstrated that pregnant women were at increased risk for severe illness, including risks of intensive care unit (ICU) admission, invasive ventilation, and extracorporeal membrane oxygenation, compared with non–pregnant women.1 Since then, other groups have confirmed the increased risks of severe COVID-19, and also identified pregnancy-specific risks, such as preeclampsia, cesarean delivery (CD), prematurity, venous thromboembolic (VTE) disease, and stillbirth.2-6
The recent study by Ferrara and colleagues adds more granular data to help refine understanding of COVID-19 in pregnancy and counsel patients.
Details of the study
The authors conducted a retrospective cohort study between March 1, 2020, and March 16, 2021, using the electronic health records (EHRs) from Kaiser Permanente Northern California, an integrated managed care organization that serves 4.5 million patients annually. Universal testing for SARS-CoV-2 upon admission for delivery began December 1, 2020; prior to this date, asymptomatic pregnant women were tested only for certain criteria (such as being a health care worker or having high-risk medical conditions).
Pregnant women were identified with SARS-CoV-2 based on 1) a positive polymerase chain reaction test result between 30 days prior to the last menstrual period up to 7 days after delivery or 2) an ICD-10 diagnosis of SARS-CoV-2 infection. Pregnant women not meeting these criteria were classified as SARS-CoV-2 negative. Women were followed through pregnancy to understand if they experienced preterm birth (spontaneous and medically indicated), gestational hypertension, preeclampsia/eclampsia, VTE disease, gestational diabetes, severe maternal morbidity (as defined by the CDC), hospitalization, and livebirth (or stillbirth), in order to consider the timing of SARS-CoV-2 infection relative to each of these outcomes (and ascertain whether SARS-CoV-2 infection preceded any of these outcomes more commonly than not). Management of pregnancies with COVID-19 across this large organization and multiple hospitals was not specified.
Identified perinatal risks
Among 43,886 pregnant women included in the cohort, 1,332 (3.0%) were diagnosed with SARS-CoV-2, with the vast majority of positive tests in the third trimester. Significant sociodemographic differences were noted between those with and without SARS-CoV-2, including differences in age, self-reported race/ethnicity, neighborhood deprivation index, and pre-pregnancy body mass index; no differences were noted for other pre-existing comorbidities, gestational week at delivery, or smoking in pregnancy.
In multivariable models, SARS-CoV-2 infection in pregnancy was associated with severe maternal morbidity, preterm birth, and VTE disease. It was not associated with stillbirth, any hypertensive disorder of pregnancy, CD, or any neonatal complication.
The prevalence of SARS-CoV-2 was 1.3% prior to and 8.0% after implementation of universal testing in pregnancy
A total of 307 of the 1,332 pregant women with SARS-CoV-2 were admitted to the hospital for symptomatic infection; 3 required noninvasive positive-pressure ventilation, and 1 required mechanical ventilation.
Continue to: Study strengths and weaknesses...
Study strengths and weaknesses
Strengths include the large, EHR-based dataset from a single organization, allowing for granular analysis on patient comorbidities and outcomes (rather than only based on diagnosis codes, as is true of many other large databases), as well as focus on relevant perinatal outcomes and thoughtful statistical modeling. However, a significant challenge with this, and many other studies, is ascertainment of SARS-CoV-2 infections throughout pregnancy. Asymptomatic and mildly symptomatic women, who may not be as likely to have adverse pregnancy outcomes, can often be counted in the unaffected population, biasing study results toward increased risks of SARS-CoV-2. Although the findings stratified by implementation of universal testing (which captures a greater fraction of asymptomatic patients at admission for delivery), do not suggest risk mitigation with asymptomatic status, this analysis did not capture asymptomatic infections earlier in pregnancy, many of which might not be associated with perinatal risk.
Another challenge with such a dataset is that one cannot determine the severity of illness of each patient without manual review of each chart; however, other data that are easily abstracted from the EHR may serve as a proxy. For instance, of the 307 women with symptomatic COVID-19, 4 required respiratory support above nasal cannula. This suggests a low rate of severely ill women, and may explain some of the findings in the study, such as no differences in the rate of CD, hypertensive disorders of pregnancy, or stillbirth, but does not explain the increased risk of both medically indicated and spontaneous preterm birth, or the rates of acute respiratory distress syndrome and sepsis that drive the increased risk of severe maternal morbidity.
The CDC has published data on the risks of stillbirth from a large hospital-based administrative database for COVID-19 from Premier Healthcare.2 In a cohort of over 1.2 million women admitted for delivery, including the timeframe of Ferrara et al’s study, COVID-19 was associated with a 2-fold increased risk of stillbirth, with higher risks noted with the delta variant. A rare outcome, stillbirth occurs in 6/1,000 births,7 which was the rate seen in Ferrara’s publication for both women with and without SARS-CoV-2 infection. The rare nature of the outcome may explain why a signal was not noted in the article of interest.
Translating data to patient counseling
Ferrara and colleagues’ study clearly confirms that COVID-19 infection has risks. Although many women with a COVID-19 infection in pregnancy may have an uncomplicated course, a favorable outcome is hard to predict with certainty. Risks of prematurity, VTE, organ dysfunction, and stillbirth from COVID-19 are rare but devastating complications. However, vaccinated women tend to incur far fewer adverse outcomes of COVID-19 in pregnancy, namely a 90% risk reduction in severe or critical COVID-19, with lower rates of ICU admissions and stillbirths.8 While these data strongly favor vaccination, we remain ill-advised on management strategies specifically to mitigate risk for the pregnancy once affected by COVID-19 infection. Thus, prevention with vaccination, mask wearing, and physical distancing remains a cornerstone of prenatal care in the current day. ●
These data continue to support that SARS-CoV-2 infection is associated with prematurity, VTE, and severe maternal adverse outcomes. As sports fanatics often state, the best defense is a good offense. In the case of SARS-CoV-2, COVID-19 vaccination, mask wearing, and physical distancing are likely the best offense against COVID-19 infection in pregnancy.
Ferrara A, Hedderson MM, Zhu Y, et al. Perinatal complications in individuals in California with or without SARS-CoV-2 infection during pregnancy. JAMA Intern Med. 2022;182:503-512. doi:10.1001/jamainternmed.2022.0330
Expert Commentary
SARS-CoV-2 infection is associated with several adverse outcomes, with the magnitude of specific risks varying by population studied and study design used. Early Centers for Disease Control and Prevention (CDC) data demonstrated that pregnant women were at increased risk for severe illness, including risks of intensive care unit (ICU) admission, invasive ventilation, and extracorporeal membrane oxygenation, compared with non–pregnant women.1 Since then, other groups have confirmed the increased risks of severe COVID-19, and also identified pregnancy-specific risks, such as preeclampsia, cesarean delivery (CD), prematurity, venous thromboembolic (VTE) disease, and stillbirth.2-6
The recent study by Ferrara and colleagues adds more granular data to help refine understanding of COVID-19 in pregnancy and counsel patients.
Details of the study
The authors conducted a retrospective cohort study between March 1, 2020, and March 16, 2021, using the electronic health records (EHRs) from Kaiser Permanente Northern California, an integrated managed care organization that serves 4.5 million patients annually. Universal testing for SARS-CoV-2 upon admission for delivery began December 1, 2020; prior to this date, asymptomatic pregnant women were tested only for certain criteria (such as being a health care worker or having high-risk medical conditions).
Pregnant women were identified with SARS-CoV-2 based on 1) a positive polymerase chain reaction test result between 30 days prior to the last menstrual period up to 7 days after delivery or 2) an ICD-10 diagnosis of SARS-CoV-2 infection. Pregnant women not meeting these criteria were classified as SARS-CoV-2 negative. Women were followed through pregnancy to understand if they experienced preterm birth (spontaneous and medically indicated), gestational hypertension, preeclampsia/eclampsia, VTE disease, gestational diabetes, severe maternal morbidity (as defined by the CDC), hospitalization, and livebirth (or stillbirth), in order to consider the timing of SARS-CoV-2 infection relative to each of these outcomes (and ascertain whether SARS-CoV-2 infection preceded any of these outcomes more commonly than not). Management of pregnancies with COVID-19 across this large organization and multiple hospitals was not specified.
Identified perinatal risks
Among 43,886 pregnant women included in the cohort, 1,332 (3.0%) were diagnosed with SARS-CoV-2, with the vast majority of positive tests in the third trimester. Significant sociodemographic differences were noted between those with and without SARS-CoV-2, including differences in age, self-reported race/ethnicity, neighborhood deprivation index, and pre-pregnancy body mass index; no differences were noted for other pre-existing comorbidities, gestational week at delivery, or smoking in pregnancy.
In multivariable models, SARS-CoV-2 infection in pregnancy was associated with severe maternal morbidity, preterm birth, and VTE disease. It was not associated with stillbirth, any hypertensive disorder of pregnancy, CD, or any neonatal complication.
The prevalence of SARS-CoV-2 was 1.3% prior to and 8.0% after implementation of universal testing in pregnancy
A total of 307 of the 1,332 pregant women with SARS-CoV-2 were admitted to the hospital for symptomatic infection; 3 required noninvasive positive-pressure ventilation, and 1 required mechanical ventilation.
Continue to: Study strengths and weaknesses...
Study strengths and weaknesses
Strengths include the large, EHR-based dataset from a single organization, allowing for granular analysis on patient comorbidities and outcomes (rather than only based on diagnosis codes, as is true of many other large databases), as well as focus on relevant perinatal outcomes and thoughtful statistical modeling. However, a significant challenge with this, and many other studies, is ascertainment of SARS-CoV-2 infections throughout pregnancy. Asymptomatic and mildly symptomatic women, who may not be as likely to have adverse pregnancy outcomes, can often be counted in the unaffected population, biasing study results toward increased risks of SARS-CoV-2. Although the findings stratified by implementation of universal testing (which captures a greater fraction of asymptomatic patients at admission for delivery), do not suggest risk mitigation with asymptomatic status, this analysis did not capture asymptomatic infections earlier in pregnancy, many of which might not be associated with perinatal risk.
Another challenge with such a dataset is that one cannot determine the severity of illness of each patient without manual review of each chart; however, other data that are easily abstracted from the EHR may serve as a proxy. For instance, of the 307 women with symptomatic COVID-19, 4 required respiratory support above nasal cannula. This suggests a low rate of severely ill women, and may explain some of the findings in the study, such as no differences in the rate of CD, hypertensive disorders of pregnancy, or stillbirth, but does not explain the increased risk of both medically indicated and spontaneous preterm birth, or the rates of acute respiratory distress syndrome and sepsis that drive the increased risk of severe maternal morbidity.
The CDC has published data on the risks of stillbirth from a large hospital-based administrative database for COVID-19 from Premier Healthcare.2 In a cohort of over 1.2 million women admitted for delivery, including the timeframe of Ferrara et al’s study, COVID-19 was associated with a 2-fold increased risk of stillbirth, with higher risks noted with the delta variant. A rare outcome, stillbirth occurs in 6/1,000 births,7 which was the rate seen in Ferrara’s publication for both women with and without SARS-CoV-2 infection. The rare nature of the outcome may explain why a signal was not noted in the article of interest.
Translating data to patient counseling
Ferrara and colleagues’ study clearly confirms that COVID-19 infection has risks. Although many women with a COVID-19 infection in pregnancy may have an uncomplicated course, a favorable outcome is hard to predict with certainty. Risks of prematurity, VTE, organ dysfunction, and stillbirth from COVID-19 are rare but devastating complications. However, vaccinated women tend to incur far fewer adverse outcomes of COVID-19 in pregnancy, namely a 90% risk reduction in severe or critical COVID-19, with lower rates of ICU admissions and stillbirths.8 While these data strongly favor vaccination, we remain ill-advised on management strategies specifically to mitigate risk for the pregnancy once affected by COVID-19 infection. Thus, prevention with vaccination, mask wearing, and physical distancing remains a cornerstone of prenatal care in the current day. ●
These data continue to support that SARS-CoV-2 infection is associated with prematurity, VTE, and severe maternal adverse outcomes. As sports fanatics often state, the best defense is a good offense. In the case of SARS-CoV-2, COVID-19 vaccination, mask wearing, and physical distancing are likely the best offense against COVID-19 infection in pregnancy.
- Zambrano LD, Ellington S, Strid P, et al. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status - United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3.
- DeSisto CL. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morb Mortal Wkly Rep. 2021;70. doi:10.15585/mmwr.mm7047e1.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and metaanalysis. BMJ. 2020;370:m3320. doi:10.1136/bmj.m3320.
- Jering KS, Claggett BL, Cunningham JW, et al. Clinical characteristics and outcomes of hospitalized women giving birth with and without COVID-19. JAMA Intern Med. 2021;181:714-717. doi:10.1001/jamainternmed.2020.9241.
- Katz D, Bateman BT, Kjaer K, et al. The Society for Obstetric Anesthesia and Perinatology Coronavirus Disease 2019 Registry: an analysis of outcomes among pregnant women delivering during the initial severe acute respiratory syndrome Coronavirus-2 outbreak in the United States. Anesth Analg. 2021;133:462-473. doi:10.1213/ANE.0000000000005592.
- Metz TD, Clifton RG, Hughes BL, et al. Association of SARS-CoV-2 infection with serious maternal morbidity and mortality from obstetric complications. JAMA. 2022;327:748759. doi:10.1001/jama.2022.1190.
- Management of stillbirth. https ://www.acog.org/en/clinical/clinical-guidance/obstetric-care-consensus/articles/2020/03/management-of-stillbirth. Accessed May 23, 2022.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109. doi:10.1097/AOG.0000000000004621.
- Zambrano LD, Ellington S, Strid P, et al. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status - United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3.
- DeSisto CL. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morb Mortal Wkly Rep. 2021;70. doi:10.15585/mmwr.mm7047e1.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and metaanalysis. BMJ. 2020;370:m3320. doi:10.1136/bmj.m3320.
- Jering KS, Claggett BL, Cunningham JW, et al. Clinical characteristics and outcomes of hospitalized women giving birth with and without COVID-19. JAMA Intern Med. 2021;181:714-717. doi:10.1001/jamainternmed.2020.9241.
- Katz D, Bateman BT, Kjaer K, et al. The Society for Obstetric Anesthesia and Perinatology Coronavirus Disease 2019 Registry: an analysis of outcomes among pregnant women delivering during the initial severe acute respiratory syndrome Coronavirus-2 outbreak in the United States. Anesth Analg. 2021;133:462-473. doi:10.1213/ANE.0000000000005592.
- Metz TD, Clifton RG, Hughes BL, et al. Association of SARS-CoV-2 infection with serious maternal morbidity and mortality from obstetric complications. JAMA. 2022;327:748759. doi:10.1001/jama.2022.1190.
- Management of stillbirth. https ://www.acog.org/en/clinical/clinical-guidance/obstetric-care-consensus/articles/2020/03/management-of-stillbirth. Accessed May 23, 2022.
- Morgan JA, Biggio JRJ, Martin JK, et al. Maternal outcomes after severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection in vaccinated compared with unvaccinated pregnant patients. Obstet Gynecol. 2022;139:107-109. doi:10.1097/AOG.0000000000004621.
Defending access to reproductive health care
The 1973 Supreme Court of the United States (SCOTUS) decision in Roe v Wade was a landmark ruling,1 establishing that the United States Constitution provides a fundamental “right to privacy,” protecting pregnant people’s freedom to access all available reproductive health care options. Recognizing that the right to abortion was not absolute, the majority of justices supported a trimester system. In the first trimester, decisions about abortion care are fully controlled by patients and clinicians, and no government could place restrictions on access to abortion. In the second trimester, SCOTUS ruled that states may choose to regulate abortion to protect maternal health. (As an example of such state restrictions, in Massachusetts, for many years, but no longer, the state required that abortions occur in a hospital when the patient was between 18 and 24 weeks’ gestation in order to facilitate comprehensive emergency care for complications.) Beginning in the third trimester, a point at which a fetus could be viable, the Court ruled that a government could prohibit abortion except when an abortion was necessary to protect the life or health of the pregnant person. In 1992, the SCOTUS decision in Planned Parenthood v Casey2 rejected the trimester system, reaffirming the right to an abortion before fetal viability, and adopting a new standard that states may not create an undue burden on a person seeking an abortion b
If, as anticipated, the 2022 SCOTUS decision in Dobbs v Jackson Women’s Health Organization3 overturns the precedents set in Roe v Wade and Planned Parenthood v Casey, decisions on abortion law will be relegated to elected legislators and state courts.4 It is expected that at least 26 state legislatures and governors will enact stringent new restrictions on access to abortion. This cataclysmic reversal of judicial opinion creates a historic challenge to obstetrician-gynecologists and their patients and could threaten access to other vital reproductive services beyond abortion, like contraception. We will be fighting, state by state, for people’s right to access all available reproductive health procedures. This will also significantly affect the ability for providers in women’s reproductive health to obtain appropriate and necessary education and training in a critical skills. If access to safe abortion is restricted, we fear patients may be forced to consider unsafe abortion, raising the specter of a return to the 1960s, when an epidemic of unsafe abortion caused countless injuries and deaths.5,6
How do we best prepare for these challenges?
- We will need to be flexible and continually evolve our clinical practices to be adherent with state and local legislation and regulation.
- To reduce unintended pregnancies, we need to strengthen our efforts to ensure that every patient has ready access to all available contraceptive options with no out-of-pocket cost.
- When a contraceptive is desired, we will focus on educating people about effectiveness, and offering them highly reliable contraception, such as the implant or intrauterine devices.
- We need to ensure timely access to abortion if state-based laws permit abortion before 6 or 7 weeks’ gestation. Providing medication abortion without an in-person visit using a telehealth option would be one option to expand rapid access to early first trimester abortion.
- Clinicians in states with access to abortion services will need to collaborate with colleagues in states with restrictions on abortion services to improve patient access across state borders.
On a national level, advancing our effective advocacy in Congress may lead to national legislation passed and signed by the President. This could supersede most state laws prohibiting access to comprehensive women’s reproductive health and create a unified, national approach to abortion care, allowing for the appropriate training of all obstetrician-gynecologists. We will also need to develop teams in every state capable of advocating for laws that ensure access to all reproductive health care options. The American College of Obstetricians and Gynecologists has leaders trained and tasked with legislative advocacy in every state.7 This network will be a foundation upon which to build additional advocacy efforts.
As women’s health care professionals, our responsibility to our patients, is to work to ensure universal access to safe and effective comprehensive reproductive options, and to ensure that our workforce is prepared to meet the needs of our patients by defending the patient-clinician relationship. Abortion care saves lives of pregnant patients and reduces maternal morbidity.8 Access to safe abortion care as part of comprehensive reproductive services is an important component of health care. ●
- Roe v Wade, 410 U.S. 113 (1973).
- Planned Parenthood v Casey, 505 U.S. 833 (1992).
- Dobbs v Jackson Women’s Health Organization, 19-1392. https://www.supremecourt.gov/search .aspx?filename=/docket/docketfiles/html /public/19-1392.html. Accessed May 18, 2022.
- Gerstein J, Ward A. Supreme Court has voted to overturn abortion rights, draft opinion shows. Politico. May 5, 2022. Updated May 3, 2022.
- Gold RB. Lessons from before Roe: will past be prologue? Guttmacher Institute. March 1, 2003. https://www.guttmacher.org/gpr/2003/03 /lessons-roe-will-past-be-prologue. Accessed May 18, 2022.
- Edelin KC. Broken Justice: A True Story of Race, Sex and Revenge in a Boston Courtroom. Pond View Press; 2007.
- The American College of Obstetricians and Gynecologists. Get involved in your state. ACOG web site. https://www.acog.org/advocacy /get-involved/get-involved-in-your-state. Accessed May 18, 2022.
- Institute of Medicine (US) Committee on Improving Birth Outcomes. Bale JR, Stoll BJ, Lucas AO, eds. Reducing maternal mortality and morbidity. In: Improving Birth Outcomes: Meeting the Challenge in the Developing World. Washington, DC: National Academies Press (US); 2003.
The 1973 Supreme Court of the United States (SCOTUS) decision in Roe v Wade was a landmark ruling,1 establishing that the United States Constitution provides a fundamental “right to privacy,” protecting pregnant people’s freedom to access all available reproductive health care options. Recognizing that the right to abortion was not absolute, the majority of justices supported a trimester system. In the first trimester, decisions about abortion care are fully controlled by patients and clinicians, and no government could place restrictions on access to abortion. In the second trimester, SCOTUS ruled that states may choose to regulate abortion to protect maternal health. (As an example of such state restrictions, in Massachusetts, for many years, but no longer, the state required that abortions occur in a hospital when the patient was between 18 and 24 weeks’ gestation in order to facilitate comprehensive emergency care for complications.) Beginning in the third trimester, a point at which a fetus could be viable, the Court ruled that a government could prohibit abortion except when an abortion was necessary to protect the life or health of the pregnant person. In 1992, the SCOTUS decision in Planned Parenthood v Casey2 rejected the trimester system, reaffirming the right to an abortion before fetal viability, and adopting a new standard that states may not create an undue burden on a person seeking an abortion b
If, as anticipated, the 2022 SCOTUS decision in Dobbs v Jackson Women’s Health Organization3 overturns the precedents set in Roe v Wade and Planned Parenthood v Casey, decisions on abortion law will be relegated to elected legislators and state courts.4 It is expected that at least 26 state legislatures and governors will enact stringent new restrictions on access to abortion. This cataclysmic reversal of judicial opinion creates a historic challenge to obstetrician-gynecologists and their patients and could threaten access to other vital reproductive services beyond abortion, like contraception. We will be fighting, state by state, for people’s right to access all available reproductive health procedures. This will also significantly affect the ability for providers in women’s reproductive health to obtain appropriate and necessary education and training in a critical skills. If access to safe abortion is restricted, we fear patients may be forced to consider unsafe abortion, raising the specter of a return to the 1960s, when an epidemic of unsafe abortion caused countless injuries and deaths.5,6
How do we best prepare for these challenges?
- We will need to be flexible and continually evolve our clinical practices to be adherent with state and local legislation and regulation.
- To reduce unintended pregnancies, we need to strengthen our efforts to ensure that every patient has ready access to all available contraceptive options with no out-of-pocket cost.
- When a contraceptive is desired, we will focus on educating people about effectiveness, and offering them highly reliable contraception, such as the implant or intrauterine devices.
- We need to ensure timely access to abortion if state-based laws permit abortion before 6 or 7 weeks’ gestation. Providing medication abortion without an in-person visit using a telehealth option would be one option to expand rapid access to early first trimester abortion.
- Clinicians in states with access to abortion services will need to collaborate with colleagues in states with restrictions on abortion services to improve patient access across state borders.
On a national level, advancing our effective advocacy in Congress may lead to national legislation passed and signed by the President. This could supersede most state laws prohibiting access to comprehensive women’s reproductive health and create a unified, national approach to abortion care, allowing for the appropriate training of all obstetrician-gynecologists. We will also need to develop teams in every state capable of advocating for laws that ensure access to all reproductive health care options. The American College of Obstetricians and Gynecologists has leaders trained and tasked with legislative advocacy in every state.7 This network will be a foundation upon which to build additional advocacy efforts.
As women’s health care professionals, our responsibility to our patients, is to work to ensure universal access to safe and effective comprehensive reproductive options, and to ensure that our workforce is prepared to meet the needs of our patients by defending the patient-clinician relationship. Abortion care saves lives of pregnant patients and reduces maternal morbidity.8 Access to safe abortion care as part of comprehensive reproductive services is an important component of health care. ●
The 1973 Supreme Court of the United States (SCOTUS) decision in Roe v Wade was a landmark ruling,1 establishing that the United States Constitution provides a fundamental “right to privacy,” protecting pregnant people’s freedom to access all available reproductive health care options. Recognizing that the right to abortion was not absolute, the majority of justices supported a trimester system. In the first trimester, decisions about abortion care are fully controlled by patients and clinicians, and no government could place restrictions on access to abortion. In the second trimester, SCOTUS ruled that states may choose to regulate abortion to protect maternal health. (As an example of such state restrictions, in Massachusetts, for many years, but no longer, the state required that abortions occur in a hospital when the patient was between 18 and 24 weeks’ gestation in order to facilitate comprehensive emergency care for complications.) Beginning in the third trimester, a point at which a fetus could be viable, the Court ruled that a government could prohibit abortion except when an abortion was necessary to protect the life or health of the pregnant person. In 1992, the SCOTUS decision in Planned Parenthood v Casey2 rejected the trimester system, reaffirming the right to an abortion before fetal viability, and adopting a new standard that states may not create an undue burden on a person seeking an abortion b
If, as anticipated, the 2022 SCOTUS decision in Dobbs v Jackson Women’s Health Organization3 overturns the precedents set in Roe v Wade and Planned Parenthood v Casey, decisions on abortion law will be relegated to elected legislators and state courts.4 It is expected that at least 26 state legislatures and governors will enact stringent new restrictions on access to abortion. This cataclysmic reversal of judicial opinion creates a historic challenge to obstetrician-gynecologists and their patients and could threaten access to other vital reproductive services beyond abortion, like contraception. We will be fighting, state by state, for people’s right to access all available reproductive health procedures. This will also significantly affect the ability for providers in women’s reproductive health to obtain appropriate and necessary education and training in a critical skills. If access to safe abortion is restricted, we fear patients may be forced to consider unsafe abortion, raising the specter of a return to the 1960s, when an epidemic of unsafe abortion caused countless injuries and deaths.5,6
How do we best prepare for these challenges?
- We will need to be flexible and continually evolve our clinical practices to be adherent with state and local legislation and regulation.
- To reduce unintended pregnancies, we need to strengthen our efforts to ensure that every patient has ready access to all available contraceptive options with no out-of-pocket cost.
- When a contraceptive is desired, we will focus on educating people about effectiveness, and offering them highly reliable contraception, such as the implant or intrauterine devices.
- We need to ensure timely access to abortion if state-based laws permit abortion before 6 or 7 weeks’ gestation. Providing medication abortion without an in-person visit using a telehealth option would be one option to expand rapid access to early first trimester abortion.
- Clinicians in states with access to abortion services will need to collaborate with colleagues in states with restrictions on abortion services to improve patient access across state borders.
On a national level, advancing our effective advocacy in Congress may lead to national legislation passed and signed by the President. This could supersede most state laws prohibiting access to comprehensive women’s reproductive health and create a unified, national approach to abortion care, allowing for the appropriate training of all obstetrician-gynecologists. We will also need to develop teams in every state capable of advocating for laws that ensure access to all reproductive health care options. The American College of Obstetricians and Gynecologists has leaders trained and tasked with legislative advocacy in every state.7 This network will be a foundation upon which to build additional advocacy efforts.
As women’s health care professionals, our responsibility to our patients, is to work to ensure universal access to safe and effective comprehensive reproductive options, and to ensure that our workforce is prepared to meet the needs of our patients by defending the patient-clinician relationship. Abortion care saves lives of pregnant patients and reduces maternal morbidity.8 Access to safe abortion care as part of comprehensive reproductive services is an important component of health care. ●
- Roe v Wade, 410 U.S. 113 (1973).
- Planned Parenthood v Casey, 505 U.S. 833 (1992).
- Dobbs v Jackson Women’s Health Organization, 19-1392. https://www.supremecourt.gov/search .aspx?filename=/docket/docketfiles/html /public/19-1392.html. Accessed May 18, 2022.
- Gerstein J, Ward A. Supreme Court has voted to overturn abortion rights, draft opinion shows. Politico. May 5, 2022. Updated May 3, 2022.
- Gold RB. Lessons from before Roe: will past be prologue? Guttmacher Institute. March 1, 2003. https://www.guttmacher.org/gpr/2003/03 /lessons-roe-will-past-be-prologue. Accessed May 18, 2022.
- Edelin KC. Broken Justice: A True Story of Race, Sex and Revenge in a Boston Courtroom. Pond View Press; 2007.
- The American College of Obstetricians and Gynecologists. Get involved in your state. ACOG web site. https://www.acog.org/advocacy /get-involved/get-involved-in-your-state. Accessed May 18, 2022.
- Institute of Medicine (US) Committee on Improving Birth Outcomes. Bale JR, Stoll BJ, Lucas AO, eds. Reducing maternal mortality and morbidity. In: Improving Birth Outcomes: Meeting the Challenge in the Developing World. Washington, DC: National Academies Press (US); 2003.
- Roe v Wade, 410 U.S. 113 (1973).
- Planned Parenthood v Casey, 505 U.S. 833 (1992).
- Dobbs v Jackson Women’s Health Organization, 19-1392. https://www.supremecourt.gov/search .aspx?filename=/docket/docketfiles/html /public/19-1392.html. Accessed May 18, 2022.
- Gerstein J, Ward A. Supreme Court has voted to overturn abortion rights, draft opinion shows. Politico. May 5, 2022. Updated May 3, 2022.
- Gold RB. Lessons from before Roe: will past be prologue? Guttmacher Institute. March 1, 2003. https://www.guttmacher.org/gpr/2003/03 /lessons-roe-will-past-be-prologue. Accessed May 18, 2022.
- Edelin KC. Broken Justice: A True Story of Race, Sex and Revenge in a Boston Courtroom. Pond View Press; 2007.
- The American College of Obstetricians and Gynecologists. Get involved in your state. ACOG web site. https://www.acog.org/advocacy /get-involved/get-involved-in-your-state. Accessed May 18, 2022.
- Institute of Medicine (US) Committee on Improving Birth Outcomes. Bale JR, Stoll BJ, Lucas AO, eds. Reducing maternal mortality and morbidity. In: Improving Birth Outcomes: Meeting the Challenge in the Developing World. Washington, DC: National Academies Press (US); 2003.
Can US “pattern recognition” of classic adnexal lesions reduce surgery, and even referrals for other imaging, in average-risk women?
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
Gupta A, Jha P, Baran TM, et al. Ovarian cancer detection in average-risk women: classic- versus nonclassic-appearing adnexal lesions at US. Radiology. 2022;212338. doi: 10.1148/radiol.212338.
Expert commentary
Gupta and colleagues conducted a multicenter, retrospective review of 970 adnexal lesions among 878 women—75% were premenopausal and 25% were postmenopausal.
Imaging details
The lesions were characterized by pattern recognition as “classic” (simple cysts, endometriomas, hemorrhagic cysts, or dermoids) or “nonclassic.” Out of 673 classic lesions, there were 4 malignancies (0.6%), of which 1 was an endometrioma and 3 were classified as simple cysts. However, out of 297 nonclassic lesions (multilocular, unilocular with solid areas or wall irregularity, or mostly solid), 32% (33/103) were malignant when vascularity was present, while 8% (16/184) were malignant when no intralesional vascularity was appreciated.
The authors pointed out that, especially because their study was retrospective, there was no standardization of scan technique or equipment employed. However, this point adds credibility to the “real world” nature of such imaging.
Other data corroborate findings
Other studies have looked at pattern recognition in efforts to optimize a conservative approach to benign masses and referral to oncology for suspected malignant masses, as described above. This was the main cornerstone of the International Consensus Conference,2 which also identified next steps for indeterminate masses, including evidence-based risk assessment algorithms and referral (to an expert imager or gynecologic oncologist). A multicenter trial in Europe3 found that ultrasound experience substantially impacts on diagnostic performance when adnexal masses are classified using pattern recognition. This occurred in a stepwise fashion with increasing accuracy directly related to the level of expertise. Shetty and colleagues4 found that pattern recognition performed better than the risk of malignancy index (sensitivities of 95% and 79%, respectively). ●
While the concept of pattern recognition for some “classic” benign ovarian masses has been around for some time, this is the first time a large United States–based study (albeit retrospective) has corroborated that when ultrasonography reveals a classic, or “almost certainly benign” finding, patients can be reassured that the lesion is benign, thereby avoiding extensive further workup. When a lesion is “nonclassic” in appearance and without any blood flow, further imaging with follow-up magnetic resonance imaging or repeat ultrasound could be considered. In women with a nonclassic lesion with blood flow, particularly in older women, referral to a gynecologic oncologic surgeon will help ensure expeditious treatment of possible ovarian cancer.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
- Boll D, Geomini PM, Brölmann HA. The pre-operative assessment of the adnexal mass: the accuracy of clinical estimates versus clinical prediction rules. BJOG. 2003;110:519-523.
- Glanc P, Benacerraf B, Bourne T, et al. First International Consensus Report on adnexal masses: management recommendations. J Ultrasound Med. 2017;36:849-863. doi: 10.1002/jum.14197.
- Van Holsbeke C, Daemen A, Yazbek J, et al. Ultrasound experience substantially impacts on diagnostic performance and confidence when adnexal masses are classified using pattern recognition. Gynecol Obstet Invest. 2010;69:160-168. doi: 10.1159/000265012.
- Shetty J, Reddy G, Pandey D. Role of sonographic grayscale pattern recognition in the diagnosis of adnexal masses. J Clin Diagn Res. 2017;11:QC12-QC15. doi: 10.7860 /JCDR/2017/28533.10614.
What placental mechanisms protect the fetus from harm in the setting of maternal COVID-19 infection?
Taglauer ES, Wachman EM, Juttukonda L, et al. Acute severe acute respiratory syndrome coronavirus 2 infection in pregnancy is associated with placental angiotensin-converting enzyme 2 shedding. Am J Pathol. 2022;192:595-603. doi.org/10.1016/j.ajpath.2021.12.011
EXPERT COMMENTARY
Although transmission of SARS-CoV-2 virus from an infected mother to her fetus is rare, placental infection with SARS-CoV-2 can occur and has been observed in association with placental damage and adverse pregnancy outcomes, including stillbirth.1 Understanding what mechanisms of defense protect the placenta and fetus from direct SARS-CoV-2 infection at the maternal-fetal interface, as well as the factors that might disturb or enhance that protection, is critical to gaining a deeper understanding of the potential impact of maternal COVID-19 on fetal well-being.
Details of the study
In a cohort of 24 pregnant individuals, Taglauer and colleagues investigated levels of placental angiotensin-converting enzyme (ACE)-2, placental ADAM17 (a disintegrin and metalloprotease domain 17) activity, and maternal serum soluble ACE2 in samples obtained at delivery from individuals with a history of second trimester COVID-19 infection, early third trimester COVID-19 infection, and no history of COVID-19 infection.
Results. Maternal COVID-19 infection in the early third trimester of pregnancy resulted in lower ACE2 protein levels in the placenta at delivery, higher ACE2 gene expression, and an increase in ADAM17 activity, compared with infection in the second trimester of pregnancy and compared with noninfected controls.
The authors postulated that increased ADAM17 activity—the enzyme responsible for ACE2 cleavage and shedding—may be responsible for lower ACE2 protein levels. Soluble ACE2 levels in maternal blood at delivery were increased in individuals with third trimester COVID-19 infection, although the source of soluble ACE2 (placental or otherwise) could not be determined with the methods employed. Levels of placental estrogen were no different between groups, which suggests that estrogen is not responsible for the observed differences.
Study strengths and limitations
ACE2 is the main receptor for the SARS-CoV-2 virus and facilitates viral entry into the cell.2 Placental villous cells that are in direct contact with maternal blood express the ACE2 protein, rendering them potentially vulnerable to SARS-CoV-2 infection.3 In this study, the authors observed lower placental ACE2 protein in term placentas from recent (early third trimester) but not remote (second trimester) maternal SARS-CoV-2 infection, arguably the result of the observed increase in ADAM17 cleavage activity. Prior studies have shown conflicting results, with equal or higher ACE2 levels noted in the setting of maternal COVID-19 infection, which may be related to differences in COVID-19 disease severity, gestational age of infection, and/or fetal sex in these cohorts.4-6
The concept that increased placental ACE2 shedding represents a protective defense mechanism that might last weeks beyond the acute infectious period is intriguing, but it requires further study. Observed differences in third but not second trimester COVID-19 infections could indicate either 1) an effect of maternal COVID-19 infection that lasts for several weeks but eventually normalizes over time, in the case of a remote infection; or 2) that second trimester maternal COVID-19 infection does not have the same pronounced effect on ACE2 levels as does third trimester infection. Observational studies of the human placenta are not able to answer this question, as directly sampling the placenta at the time of the exposure (or repeated sampling over time) in ongoing pregnancies is neither practical nor ethical. Further studies using animal or cellular models of SARS-CoV-2 infection in pregnancy may be necessary to fully understand the clinical relevance of these findings.
The study by Taglauer and colleagues provides a compelling argument for exploring how immune defenses at the maternal-fetal interface evolve over time and vary by trimester of exposure. ●
As the number of pregnancies exposed to COVID-19 continues to grow worldwide, how immune defenses at the maternal-fetal interface protect against fetal infection remains an important area of investigation.
LYDIA L. SHOOK, MD
- Pregnant people are at increased risk of more severe COVID-19 illness.
- The risk of stillbirth is 2- to 4-fold higher in women with COVID-19 infection during pregnancy.1
- COVID-19 vaccination is recommended for all people who are pregnant, lactating, or considering pregnancy.
- Pregnant and recently pregnant people up to 6 weeks postpartum should receive a third “booster” dose of a COVID-19 mRNA vaccine following completion of their initial COVID-19 vaccine or vaccine series.
- The mRNA COVID-19 vaccines are preferred over the Johnson & Johnson/Janssen COVID-19 vaccine for pregnant and lactating individuals for primary series and booster vaccination.
- Completion of a 2-dose mRNA COVID-19 vaccination series during pregnancy might help prevent COVID-19 hospitalization among infants <6 months.2
aVaccine recommendations adapted from: ACOG practice advisory: COVID-19 vaccination considerations for obstetric-gynecologic care. Last updated March 2, 2022. https://www.acog.org/clinical/ clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetricgynecologic-care. Accessed March 21, 2022.
References
1. DeSisto CL, Wallace B, Simeone RM, et al. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morbid Mortal Wkly Rep. 2021;70:1640-1645.
2. Halasa NB, Olson SM, Staat MA, et al; Overcoming COVID-19 Investigators; Overcoming COVID-19 Network. Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:264-270.
- Schwartz DA, Avvad-Portari E, Babál, et al. Placental tissue destruction and insufficiency from COVID-19 causes stillbirth and neonatal death from hypoxic-ischemic injury: a study of 68 cases with SARS-CoV-2 placentitis from 12 countries. Arch Pathol Lab Med. February 10, 2022. doi:10.5858/arpa.2022- 0029-SA.
- Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181: 271-280.e8.
- Hecht JL, Quade B, Deshpande V, et al. SARS-CoV-2 can infect the placenta and is not associated with specific placental histopathology: a series of 19 placentas from COVID-19- positive mothers. Mod Pathol. 2020;33:2092-2103.
- Mourad M, Jacob T, Sadovsky E, et al. Placental response to maternal SARS-CoV-2 infection. Sci Rep. 2021;11:14390.
- Lu-Culligan A, Chavan AR, Vijayakumar P, et al. Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface. Med (N Y). 2021;2:591-610.e10.
- Shook LL, Bordt EA, Meinsohn MC, et al. Placental expression of ACE2 and TMPRSS2 in maternal severe acute respiratory syndrome coronavirus 2 infection: are placental defenses mediated by fetal sex? J Infect Dis. 2021;224(suppl 6):S659.
Taglauer ES, Wachman EM, Juttukonda L, et al. Acute severe acute respiratory syndrome coronavirus 2 infection in pregnancy is associated with placental angiotensin-converting enzyme 2 shedding. Am J Pathol. 2022;192:595-603. doi.org/10.1016/j.ajpath.2021.12.011
EXPERT COMMENTARY
Although transmission of SARS-CoV-2 virus from an infected mother to her fetus is rare, placental infection with SARS-CoV-2 can occur and has been observed in association with placental damage and adverse pregnancy outcomes, including stillbirth.1 Understanding what mechanisms of defense protect the placenta and fetus from direct SARS-CoV-2 infection at the maternal-fetal interface, as well as the factors that might disturb or enhance that protection, is critical to gaining a deeper understanding of the potential impact of maternal COVID-19 on fetal well-being.
Details of the study
In a cohort of 24 pregnant individuals, Taglauer and colleagues investigated levels of placental angiotensin-converting enzyme (ACE)-2, placental ADAM17 (a disintegrin and metalloprotease domain 17) activity, and maternal serum soluble ACE2 in samples obtained at delivery from individuals with a history of second trimester COVID-19 infection, early third trimester COVID-19 infection, and no history of COVID-19 infection.
Results. Maternal COVID-19 infection in the early third trimester of pregnancy resulted in lower ACE2 protein levels in the placenta at delivery, higher ACE2 gene expression, and an increase in ADAM17 activity, compared with infection in the second trimester of pregnancy and compared with noninfected controls.
The authors postulated that increased ADAM17 activity—the enzyme responsible for ACE2 cleavage and shedding—may be responsible for lower ACE2 protein levels. Soluble ACE2 levels in maternal blood at delivery were increased in individuals with third trimester COVID-19 infection, although the source of soluble ACE2 (placental or otherwise) could not be determined with the methods employed. Levels of placental estrogen were no different between groups, which suggests that estrogen is not responsible for the observed differences.
Study strengths and limitations
ACE2 is the main receptor for the SARS-CoV-2 virus and facilitates viral entry into the cell.2 Placental villous cells that are in direct contact with maternal blood express the ACE2 protein, rendering them potentially vulnerable to SARS-CoV-2 infection.3 In this study, the authors observed lower placental ACE2 protein in term placentas from recent (early third trimester) but not remote (second trimester) maternal SARS-CoV-2 infection, arguably the result of the observed increase in ADAM17 cleavage activity. Prior studies have shown conflicting results, with equal or higher ACE2 levels noted in the setting of maternal COVID-19 infection, which may be related to differences in COVID-19 disease severity, gestational age of infection, and/or fetal sex in these cohorts.4-6
The concept that increased placental ACE2 shedding represents a protective defense mechanism that might last weeks beyond the acute infectious period is intriguing, but it requires further study. Observed differences in third but not second trimester COVID-19 infections could indicate either 1) an effect of maternal COVID-19 infection that lasts for several weeks but eventually normalizes over time, in the case of a remote infection; or 2) that second trimester maternal COVID-19 infection does not have the same pronounced effect on ACE2 levels as does third trimester infection. Observational studies of the human placenta are not able to answer this question, as directly sampling the placenta at the time of the exposure (or repeated sampling over time) in ongoing pregnancies is neither practical nor ethical. Further studies using animal or cellular models of SARS-CoV-2 infection in pregnancy may be necessary to fully understand the clinical relevance of these findings.
The study by Taglauer and colleagues provides a compelling argument for exploring how immune defenses at the maternal-fetal interface evolve over time and vary by trimester of exposure. ●
As the number of pregnancies exposed to COVID-19 continues to grow worldwide, how immune defenses at the maternal-fetal interface protect against fetal infection remains an important area of investigation.
LYDIA L. SHOOK, MD
- Pregnant people are at increased risk of more severe COVID-19 illness.
- The risk of stillbirth is 2- to 4-fold higher in women with COVID-19 infection during pregnancy.1
- COVID-19 vaccination is recommended for all people who are pregnant, lactating, or considering pregnancy.
- Pregnant and recently pregnant people up to 6 weeks postpartum should receive a third “booster” dose of a COVID-19 mRNA vaccine following completion of their initial COVID-19 vaccine or vaccine series.
- The mRNA COVID-19 vaccines are preferred over the Johnson & Johnson/Janssen COVID-19 vaccine for pregnant and lactating individuals for primary series and booster vaccination.
- Completion of a 2-dose mRNA COVID-19 vaccination series during pregnancy might help prevent COVID-19 hospitalization among infants <6 months.2
aVaccine recommendations adapted from: ACOG practice advisory: COVID-19 vaccination considerations for obstetric-gynecologic care. Last updated March 2, 2022. https://www.acog.org/clinical/ clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetricgynecologic-care. Accessed March 21, 2022.
References
1. DeSisto CL, Wallace B, Simeone RM, et al. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morbid Mortal Wkly Rep. 2021;70:1640-1645.
2. Halasa NB, Olson SM, Staat MA, et al; Overcoming COVID-19 Investigators; Overcoming COVID-19 Network. Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:264-270.
Taglauer ES, Wachman EM, Juttukonda L, et al. Acute severe acute respiratory syndrome coronavirus 2 infection in pregnancy is associated with placental angiotensin-converting enzyme 2 shedding. Am J Pathol. 2022;192:595-603. doi.org/10.1016/j.ajpath.2021.12.011
EXPERT COMMENTARY
Although transmission of SARS-CoV-2 virus from an infected mother to her fetus is rare, placental infection with SARS-CoV-2 can occur and has been observed in association with placental damage and adverse pregnancy outcomes, including stillbirth.1 Understanding what mechanisms of defense protect the placenta and fetus from direct SARS-CoV-2 infection at the maternal-fetal interface, as well as the factors that might disturb or enhance that protection, is critical to gaining a deeper understanding of the potential impact of maternal COVID-19 on fetal well-being.
Details of the study
In a cohort of 24 pregnant individuals, Taglauer and colleagues investigated levels of placental angiotensin-converting enzyme (ACE)-2, placental ADAM17 (a disintegrin and metalloprotease domain 17) activity, and maternal serum soluble ACE2 in samples obtained at delivery from individuals with a history of second trimester COVID-19 infection, early third trimester COVID-19 infection, and no history of COVID-19 infection.
Results. Maternal COVID-19 infection in the early third trimester of pregnancy resulted in lower ACE2 protein levels in the placenta at delivery, higher ACE2 gene expression, and an increase in ADAM17 activity, compared with infection in the second trimester of pregnancy and compared with noninfected controls.
The authors postulated that increased ADAM17 activity—the enzyme responsible for ACE2 cleavage and shedding—may be responsible for lower ACE2 protein levels. Soluble ACE2 levels in maternal blood at delivery were increased in individuals with third trimester COVID-19 infection, although the source of soluble ACE2 (placental or otherwise) could not be determined with the methods employed. Levels of placental estrogen were no different between groups, which suggests that estrogen is not responsible for the observed differences.
Study strengths and limitations
ACE2 is the main receptor for the SARS-CoV-2 virus and facilitates viral entry into the cell.2 Placental villous cells that are in direct contact with maternal blood express the ACE2 protein, rendering them potentially vulnerable to SARS-CoV-2 infection.3 In this study, the authors observed lower placental ACE2 protein in term placentas from recent (early third trimester) but not remote (second trimester) maternal SARS-CoV-2 infection, arguably the result of the observed increase in ADAM17 cleavage activity. Prior studies have shown conflicting results, with equal or higher ACE2 levels noted in the setting of maternal COVID-19 infection, which may be related to differences in COVID-19 disease severity, gestational age of infection, and/or fetal sex in these cohorts.4-6
The concept that increased placental ACE2 shedding represents a protective defense mechanism that might last weeks beyond the acute infectious period is intriguing, but it requires further study. Observed differences in third but not second trimester COVID-19 infections could indicate either 1) an effect of maternal COVID-19 infection that lasts for several weeks but eventually normalizes over time, in the case of a remote infection; or 2) that second trimester maternal COVID-19 infection does not have the same pronounced effect on ACE2 levels as does third trimester infection. Observational studies of the human placenta are not able to answer this question, as directly sampling the placenta at the time of the exposure (or repeated sampling over time) in ongoing pregnancies is neither practical nor ethical. Further studies using animal or cellular models of SARS-CoV-2 infection in pregnancy may be necessary to fully understand the clinical relevance of these findings.
The study by Taglauer and colleagues provides a compelling argument for exploring how immune defenses at the maternal-fetal interface evolve over time and vary by trimester of exposure. ●
As the number of pregnancies exposed to COVID-19 continues to grow worldwide, how immune defenses at the maternal-fetal interface protect against fetal infection remains an important area of investigation.
LYDIA L. SHOOK, MD
- Pregnant people are at increased risk of more severe COVID-19 illness.
- The risk of stillbirth is 2- to 4-fold higher in women with COVID-19 infection during pregnancy.1
- COVID-19 vaccination is recommended for all people who are pregnant, lactating, or considering pregnancy.
- Pregnant and recently pregnant people up to 6 weeks postpartum should receive a third “booster” dose of a COVID-19 mRNA vaccine following completion of their initial COVID-19 vaccine or vaccine series.
- The mRNA COVID-19 vaccines are preferred over the Johnson & Johnson/Janssen COVID-19 vaccine for pregnant and lactating individuals for primary series and booster vaccination.
- Completion of a 2-dose mRNA COVID-19 vaccination series during pregnancy might help prevent COVID-19 hospitalization among infants <6 months.2
aVaccine recommendations adapted from: ACOG practice advisory: COVID-19 vaccination considerations for obstetric-gynecologic care. Last updated March 2, 2022. https://www.acog.org/clinical/ clinical-guidance/practice-advisory/articles/2020/12/covid-19-vaccination-considerations-for-obstetricgynecologic-care. Accessed March 21, 2022.
References
1. DeSisto CL, Wallace B, Simeone RM, et al. Risk for stillbirth among women with and without COVID-19 at delivery hospitalization—United States, March 2020–September 2021. MMWR Morbid Mortal Wkly Rep. 2021;70:1640-1645.
2. Halasa NB, Olson SM, Staat MA, et al; Overcoming COVID-19 Investigators; Overcoming COVID-19 Network. Effectiveness of maternal vaccination with mRNA COVID-19 vaccine during pregnancy against COVID-19–associated hospitalization in infants aged <6 months—17 states, July 2021–January 2022. MMWR Morbid Mortal Wkly Rep. 2022;71:264-270.
- Schwartz DA, Avvad-Portari E, Babál, et al. Placental tissue destruction and insufficiency from COVID-19 causes stillbirth and neonatal death from hypoxic-ischemic injury: a study of 68 cases with SARS-CoV-2 placentitis from 12 countries. Arch Pathol Lab Med. February 10, 2022. doi:10.5858/arpa.2022- 0029-SA.
- Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181: 271-280.e8.
- Hecht JL, Quade B, Deshpande V, et al. SARS-CoV-2 can infect the placenta and is not associated with specific placental histopathology: a series of 19 placentas from COVID-19- positive mothers. Mod Pathol. 2020;33:2092-2103.
- Mourad M, Jacob T, Sadovsky E, et al. Placental response to maternal SARS-CoV-2 infection. Sci Rep. 2021;11:14390.
- Lu-Culligan A, Chavan AR, Vijayakumar P, et al. Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface. Med (N Y). 2021;2:591-610.e10.
- Shook LL, Bordt EA, Meinsohn MC, et al. Placental expression of ACE2 and TMPRSS2 in maternal severe acute respiratory syndrome coronavirus 2 infection: are placental defenses mediated by fetal sex? J Infect Dis. 2021;224(suppl 6):S659.
- Schwartz DA, Avvad-Portari E, Babál, et al. Placental tissue destruction and insufficiency from COVID-19 causes stillbirth and neonatal death from hypoxic-ischemic injury: a study of 68 cases with SARS-CoV-2 placentitis from 12 countries. Arch Pathol Lab Med. February 10, 2022. doi:10.5858/arpa.2022- 0029-SA.
- Hoffmann M, Kleine-Weber H, Schroeder S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181: 271-280.e8.
- Hecht JL, Quade B, Deshpande V, et al. SARS-CoV-2 can infect the placenta and is not associated with specific placental histopathology: a series of 19 placentas from COVID-19- positive mothers. Mod Pathol. 2020;33:2092-2103.
- Mourad M, Jacob T, Sadovsky E, et al. Placental response to maternal SARS-CoV-2 infection. Sci Rep. 2021;11:14390.
- Lu-Culligan A, Chavan AR, Vijayakumar P, et al. Maternal respiratory SARS-CoV-2 infection in pregnancy is associated with a robust inflammatory response at the maternal-fetal interface. Med (N Y). 2021;2:591-610.e10.
- Shook LL, Bordt EA, Meinsohn MC, et al. Placental expression of ACE2 and TMPRSS2 in maternal severe acute respiratory syndrome coronavirus 2 infection: are placental defenses mediated by fetal sex? J Infect Dis. 2021;224(suppl 6):S659.